GENERAL BIOLOGY I TEST I
|
|
|
- Brent Higgins
- 3 years ago
- Views:
From this document you will learn the answers to the following questions:
What is an example of an example of a plant?
What part of a cell does a prokarryote have?
What is the purpose of this paper?
Transcription
1 GENERAL BIOLOGY I TEST I R E V I E W The Academic Support Daytona State College (Science 59, Page 1 of 15)
2 BE ABLE TO DEFINE Biology The study of life Hypothesis A proposed explanation of a natural phenomenon, must be testable and falsifiable Alternative Hypothesis The opposite of the hypothesis Theory A hypothesis that has stood the test of time, and has a large amount of supporting evidence Testable Quantifiable, measurable Falsifiable Can be disproven Eukaryote Has cells with a nucleus and membrane-bound organelles Prokaryote Cells have no nucleus or membrane bound organelles The Academic Support Daytona State College (Science 59, Page 2 of 15)
3 BE ABLE TO DEFINE Proton Positively charged particle in the nucleus of an atom. (Mass = 1 amu) Neutron Non charged particle in the nucleus of an atom. (Mass = 1 amu) Electron Negatively charged particle that orbits the nucleus of an atom. Electron Orbital The area where an electron may be found Hydrophilic Will mix with water (polar) Hydrophobic Repels water (non-polar) DNA Deoxyribonucleic Acid, used for storing information in a cell RNA Ribonucleic Acid, used to carry information to be processed and turned into protein The Academic Support Daytona State College (Science 59, Page 3 of 15)
4 BE ABLE TO DEFINE Nucleotide A nitrogenous base acid + A 5-carbon sugar (deoxyribose or ribose) + a phosphate group, building blocks of DNA and RNA Positive Feedback A cycle in which one signal triggers increase in another, which increases the original signal. System output increases until system crashes. i.e. childbirth Negative Feedback One signal triggers increase of another, which decreases the original signal, Maintains balance. i.e. insulin and glucagon Artificial Selection Selection by humans for specific traits in animals or plants Natural Selection Selection by the environment for certain traits in animals or plants Common Descent All things are descended from a common ancestor, i.e. all species evolved from one organism Solvent What a chemical is dissolved in, usually water Solute What is being dissolved The Academic Support Daytona State College (Science 59, Page 4 of 15)
5 BE ABLE TO DEFINE Photosynthetic Uses photons (sunlight) to make food, i.e. makes glucose via photosynthesis Heterotrophic Uses existing sources of food, i.e. eats other things Isotope An atom of a given element with extra neutrons (extra mass) in its nucleus Trace Element Elements that make up less than.01% of human body weight Atomic Number The number of protons in an atom s nucleus, defines the element Atomic Mass The mass of protons and neutrons in an atom Calorie The heat required to raise one gram of water one degree centigrade. Metric unit of Energy. Kilocalorie 1000 Calories The Academic Support Daytona State College (Science 59, Page 5 of 15)
6 CONCEPT QUESTIONS What are the characteristics that distinguish a living organism from a non-living substance? Order, Evolutionary adaptation, Response to environment, Regulation (or homeostasis), Energy processing, Growth, Development, Reproduction Know the scientific method. Observation, Question, Hypothesis, Prediction, Experiment What did the Pasteur experiment prove? No spontaneous generation Know the order of the hierarchy of life Atom, molecule, organelle, cell, tissue, organ, organism, population, community, ecosystem, and biosphere. Be able to define each term and give examples The Academic Support Daytona State College (Science 59, Page 6 of 15)
7 THE HIERARCHY OF LIFE Examples (where relevant use plant and animal examples) Define Atom Molecule Organelle Cell Tissue Organ Organism Population Community Ecosystem Biosphere The Academic Support Daytona State College (Science 59, Page 7 of 15)
8 THE HIERARCHY OF LIFE Examples (where relevant use plant and animal examples) Define Atom Oxygen, Hydrogen, Nitrogen Smallest unit of matter Molecule Water (H 2 0) Carbon Dioxide (C0 2 ) Two or more atoms held together by covalent bond(s) Organelle Mitochondria, chloroplast a membrane bound structure; like nucleus, mitochondria, or chloroplast Cell animal cell, plant cell Basic or smallest unit of life Tissue skin or heart muscle tissue A group of cells with one function Organ heart A group of tissues with one function. Organism a single person, a single Zebra a single organism Population number of Zebras in a certain area Organisms of one species in an area. Community All the animals and plants in a forest all populations of all species in one particular area Ecosystem The Ocean, Artic, or Desert Ecosystem all biotic (living) and abiotic (nonliving) factors that interact with the biotic in an area. Biosphere The Earth or maybe northern and southern hemisphere all ecosystems on earth or global ecosystem The Academic Support Daytona State College (Science 59, Page 8 of 15)
9 CONCEPT QUESTIONS How does energy flow through an ecosystem? One way What is the ultimate source of energy for almost all ecosystems? The sun What are an ion, element, and a molecule? Ion: an atom with a different number of electrons, a charged atom Element: A pure substance, made of only one type of atom Molecule: Two or more atoms bound together What part of the atom defines an element? The number of protons Which four elements comprise 96% of the human body? Carbon, Hydrogen, Oxygen, Nitrogen What is a hydrogen bond? A dipole-dipole interaction between Hydrogen and either Oxygen, Nitrogen, or Fluorine. What is an ionic bond? A bond between two atoms, where electrons are transferred from one atom to the other What is a covalent bond? A bond between two atoms, where electrons are shared between both atoms The Academic Support Daytona State College (Science 59, Page 9 of 15)
10 CONCEPT QUESTIONS What is a polar covalent and nonpolar covalent bond? Polar covalent bonds are bonds between two non-metals of different electronegativities. Nonpolar covalent bonds are bonds between two non-metals of the same electronegativities. When differences in electronegativities is very small the bond is considered to be nonpolar. Which carries the partial negative and which carries the partial positive charge in water? Oxygen carries the partial negative, Hydrogen carries the partial positive In addition to oxygen-hydrogen bond, nitrogenhydrogen bond is polar The carbon-hydrogen bond is non-polar The Academic Support Daytona State College (Science 59, Page 10 of 15)
11 CONCEPT QUESTIONS What is the ph scale? A measure of the concentration of hydrogen ions present in a substance, it represents the acidity of the solution. What magnitude of [H] or [OH] does each number represent? ph = -log[h] Therefore each number is a magnitude of ten. How much more acidic is ph 6 than ph 7? 10 times more acidic How much more acidic is ph 4 than ph 7? 1000 times more acidic The Academic Support Daytona State College (Science 59, Page 11 of 15)
12 CONCEPT QUESTIONS Be able to identify whether a molecule is positive or negative based on whether oxygen or hydrogen is attracted to it. If Oxygen is attracted to a molecule then the molecule is positive If Hydrogen is attracted to a molecule then the molecule is negative. What is the order of taxonomy from kingdom to species? Kingdom, Phylum, Class, Order, Family, Genus, Species; keep piling chocolate on for goodness sake What are the characteristics of the three domains and the characteristics of kingdoms of Eukarya? The Academic Support Daytona State College (Science 59, Page 12 of 15)
13 WHAT ARE THE CHARACTERISTICS OF THE THREE DOMAINS AND THE CHARACTERISTICS OF KINGDOMS OF EUKARYA? Domain = Archaea Domain = Bacteria Example organism Characteristics of Domain (and then kingdom of Eukarya) Kingdom = Protista Kingdom =Animalia Kingdom =Plantae Kingdom =Fungi The Academic Support Daytona State College (Science 59, Page 13 of 15)
14 WHAT ARE THE CHARACTERISTICS OF THE THREE DOMAINS AND THE CHARACTERISTICS OF KINGDOMS OF EUKARYA? Example organism Characteristics of Domain (and then kingdom of Eukarya) Domain = Archaea Thermophile or Halophile Live in extreme environments (Salt and Heat for example) Domain = Bacteria E. Coli Prokaryotes, meaning they do not have membrane bound organelles. Also true bacteria have a peptidoglycan cell wall. Paramecium, Human, Sunflower, Mushrooms Paramecium multicellular organisms single celled organisms Kingdom = Protista Kingdom =Animalia Kingdom =Plantae Kingdom =Fungi Cheetah, Whale, Human Oak Tree, Sunflower Mushrooms multicellular eukaryotic heterotrophic organisms that ingest organic materials. multicellular eukaryotic photosynthetic organisms multicellular heterotroph that digests externally The Academic Support Daytona State College (Science 59, Page 14 of 15)
15 Questions Prepared by K. Martin & A. Bhatia (Peer Tutors) & D. Leonard (Learning Specialist) The Academic Support Daytona State College The Academic Support Daytona State College (Science 59, Page 15 of 15)
4. Why are common names not good to use when classifying organisms? Give an example.
 1. Define taxonomy. Classification of organisms 2. Who was first to classify organisms? Aristotle 3. Explain Aristotle s taxonomy of organisms. Patterns of nature: looked like 4. Why are common names not
1. Define taxonomy. Classification of organisms 2. Who was first to classify organisms? Aristotle 3. Explain Aristotle s taxonomy of organisms. Patterns of nature: looked like 4. Why are common names not
How To Understand The Human Body
 Introduction to Biology and Chemistry Outline I. Introduction to biology A. Definition of biology - Biology is the study of life. B. Characteristics of Life 1. Form and size are characteristic. e.g. A
Introduction to Biology and Chemistry Outline I. Introduction to biology A. Definition of biology - Biology is the study of life. B. Characteristics of Life 1. Form and size are characteristic. e.g. A
Topic 3: Nutrition, Photosynthesis, and Respiration
 1. Base your answer to the following question on the chemical reaction represented below and on your knowledge of biology. If this reaction takes place in an organism that requires sunlight to produce
1. Base your answer to the following question on the chemical reaction represented below and on your knowledge of biology. If this reaction takes place in an organism that requires sunlight to produce
Name: Class: Date: Multiple Choice Identify the choice that best completes the statement or answers the question.
 Name: Class: Date: Chapter 17 Practice Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The correct order for the levels of Linnaeus's classification system,
Name: Class: Date: Chapter 17 Practice Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The correct order for the levels of Linnaeus's classification system,
List the 3 main types of subatomic particles and indicate the mass and electrical charge of each.
 Basic Chemistry Why do we study chemistry in a biology course? All living organisms are composed of chemicals. To understand life, we must understand the structure, function, and properties of the chemicals
Basic Chemistry Why do we study chemistry in a biology course? All living organisms are composed of chemicals. To understand life, we must understand the structure, function, and properties of the chemicals
Examination One. Biology 101. Dr. Jaeson T. Fournier
 Examination One Biology 101 Dr. Jaeson T. Fournier Examination Instructions: Answers are to be indicated on a scantron. Keep your work protected! This helps prevent dishonesty. The instructor will not
Examination One Biology 101 Dr. Jaeson T. Fournier Examination Instructions: Answers are to be indicated on a scantron. Keep your work protected! This helps prevent dishonesty. The instructor will not
Cells & Cell Organelles
 Cells & Cell Organelles The Building Blocks of Life H Biology Types of cells bacteria cells Prokaryote - no organelles Eukaryotes - organelles animal cells plant cells Cell size comparison Animal cell
Cells & Cell Organelles The Building Blocks of Life H Biology Types of cells bacteria cells Prokaryote - no organelles Eukaryotes - organelles animal cells plant cells Cell size comparison Animal cell
Unit 5 Photosynthesis and Cellular Respiration
 Unit 5 Photosynthesis and Cellular Respiration Advanced Concepts What is the abbreviated name of this molecule? What is its purpose? What are the three parts of this molecule? Label each part with the
Unit 5 Photosynthesis and Cellular Respiration Advanced Concepts What is the abbreviated name of this molecule? What is its purpose? What are the three parts of this molecule? Label each part with the
Break down material outside their body and then absorb the nutrients. Most are single-celled organisms Usually green. Do not have nuclei
 Name Date Class CHAPTER 9 REINFORCEMENT WORKSHEET Keys to the Kingdom Complete this worksheet after you have finished reading Chapter 9, Section 2. Patty dropped her notes while she was studying the six
Name Date Class CHAPTER 9 REINFORCEMENT WORKSHEET Keys to the Kingdom Complete this worksheet after you have finished reading Chapter 9, Section 2. Patty dropped her notes while she was studying the six
Cytology. Living organisms are made up of cells. Either PROKARYOTIC or EUKARYOTIC cells.
 CYTOLOGY Cytology Living organisms are made up of cells. Either PROKARYOTIC or EUKARYOTIC cells. A. two major cell types B. distinguished by structural organization See table on handout for differences.
CYTOLOGY Cytology Living organisms are made up of cells. Either PROKARYOTIC or EUKARYOTIC cells. A. two major cell types B. distinguished by structural organization See table on handout for differences.
The animals at higher levels are more competitive, so fewer animals survive. B.
 Energy Flow in Ecosystems 1. The diagram below shows an energy pyramid. Which of the following best explains why the number of organisms at each level decreases while moving up the energy pyramid? The
Energy Flow in Ecosystems 1. The diagram below shows an energy pyramid. Which of the following best explains why the number of organisms at each level decreases while moving up the energy pyramid? The
Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display
 The Living World Chapter 1: The Science of Biology Specific Learning Outcomes: 1.1 List the major properties of life. 1.2 Explain how science is distinguished from other ways of seeking understanding of
The Living World Chapter 1: The Science of Biology Specific Learning Outcomes: 1.1 List the major properties of life. 1.2 Explain how science is distinguished from other ways of seeking understanding of
Organic Compounds. Essential Questions: What is Organic? What are the 4 major Organic Compounds? How are they made? What are they used for?
 Organic Compounds Essential Questions: What is Organic? What are the 4 major Organic Compounds? How are they made? What are they used for? Aristotle: Francesco Redi: What do we already know? Spontaneous
Organic Compounds Essential Questions: What is Organic? What are the 4 major Organic Compounds? How are they made? What are they used for? Aristotle: Francesco Redi: What do we already know? Spontaneous
Given these characteristics of life, which of the following objects is considered a living organism? W. X. Y. Z.
 Cell Structure and Organization 1. All living things must possess certain characteristics. They are all composed of one or more cells. They can grow, reproduce, and pass their genes on to their offspring.
Cell Structure and Organization 1. All living things must possess certain characteristics. They are all composed of one or more cells. They can grow, reproduce, and pass their genes on to their offspring.
ATOMS AND BONDS. Bonds
 ATOMS AND BONDS Atoms of elements are the simplest units of organization in the natural world. Atoms consist of protons (positive charge), neutrons (neutral charge) and electrons (negative charge). The
ATOMS AND BONDS Atoms of elements are the simplest units of organization in the natural world. Atoms consist of protons (positive charge), neutrons (neutral charge) and electrons (negative charge). The
IDENTIFICATION OF ORGANISMS
 reflect Take a look at the pictures on the right. Think about what the two organisms have in common. They both need food and water to survive. They both grow and reproduce. They both have similar body
reflect Take a look at the pictures on the right. Think about what the two organisms have in common. They both need food and water to survive. They both grow and reproduce. They both have similar body
D.U.C. Assist. Lec. Faculty of Dentistry Medical Biology Ihsan Dhari. Kingdoms of life
 Kingdoms of life The earliest classification system recognized only two kingdoms : plants and animals but the use of microscope led to discovery of microorganisms, so the two kingdoms system was no longer
Kingdoms of life The earliest classification system recognized only two kingdoms : plants and animals but the use of microscope led to discovery of microorganisms, so the two kingdoms system was no longer
Chapter 2 Chemical Principles
 Chapter 2 Chemical Principles I. Chemistry. [Students should read this section on their own]. a. Chemistry is the study of the interactions between atoms and molecules. b. The atom is the smallest unit
Chapter 2 Chemical Principles I. Chemistry. [Students should read this section on their own]. a. Chemistry is the study of the interactions between atoms and molecules. b. The atom is the smallest unit
The Origin of Life. The Origin of Life. Reconstructing the history of life: What features define living systems?
 The Origin of Life I. Introduction: What is life? II. The Primitive Earth III. Evidence of Life s Beginning on Earth A. Fossil Record: a point in time B. Requirements for Chemical and Cellular Evolution:
The Origin of Life I. Introduction: What is life? II. The Primitive Earth III. Evidence of Life s Beginning on Earth A. Fossil Record: a point in time B. Requirements for Chemical and Cellular Evolution:
THE HISTORY OF CELL BIOLOGY
 SECTION 4-1 REVIEW THE HISTORY OF CELL BIOLOGY Define the following terms. 1. cell 2. cell theory Write the correct letter in the blank. 1. One early piece of evidence supporting the cell theory was the
SECTION 4-1 REVIEW THE HISTORY OF CELL BIOLOGY Define the following terms. 1. cell 2. cell theory Write the correct letter in the blank. 1. One early piece of evidence supporting the cell theory was the
Which of the following can be determined based on this model? The atmosphere is the only reservoir on Earth that can store carbon in any form. A.
 Earth s Cycles 1. Models are often used to explain scientific knowledge or experimental results. A model of the carbon cycle is shown below. Which of the following can be determined based on this model?
Earth s Cycles 1. Models are often used to explain scientific knowledge or experimental results. A model of the carbon cycle is shown below. Which of the following can be determined based on this model?
Anatomy and Physiology Placement Exam 2 Practice with Answers at End!
 Anatomy and Physiology Placement Exam 2 Practice with Answers at End! General Chemical Principles 1. bonds are characterized by the sharing of electrons between the participating atoms. a. hydrogen b.
Anatomy and Physiology Placement Exam 2 Practice with Answers at End! General Chemical Principles 1. bonds are characterized by the sharing of electrons between the participating atoms. a. hydrogen b.
1.1.2. thebiotutor. AS Biology OCR. Unit F211: Cells, Exchange & Transport. Module 1.2 Cell Membranes. Notes & Questions.
 thebiotutor AS Biology OCR Unit F211: Cells, Exchange & Transport Module 1.2 Cell Membranes Notes & Questions Andy Todd 1 Outline the roles of membranes within cells and at the surface of cells. The main
thebiotutor AS Biology OCR Unit F211: Cells, Exchange & Transport Module 1.2 Cell Membranes Notes & Questions Andy Todd 1 Outline the roles of membranes within cells and at the surface of cells. The main
MCAS Biology. Review Packet
 MCAS Biology Review Packet 1 Name Class Date 1. Define organic. THE CHEMISTRY OF LIFE 2. All living things are made up of 6 essential elements: SPONCH. Name the six elements of life. S N P C O H 3. Elements
MCAS Biology Review Packet 1 Name Class Date 1. Define organic. THE CHEMISTRY OF LIFE 2. All living things are made up of 6 essential elements: SPONCH. Name the six elements of life. S N P C O H 3. Elements
Chapter 3: Water and Life
 Name Period Chapter 3: Water and Life Concept 3.1 Polar covalent bonds in water result in hydrogen bonding 1. Study the water molecules at the right. On the central molecule, label oxygen (O) and hydrogen
Name Period Chapter 3: Water and Life Concept 3.1 Polar covalent bonds in water result in hydrogen bonding 1. Study the water molecules at the right. On the central molecule, label oxygen (O) and hydrogen
Chapter 2: The Chemical Context of Life
 Chapter 2: The Chemical Context of Life Name Period This chapter covers the basics that you may have learned in your chemistry class. Whether your teacher goes over this chapter, or assigns it for you
Chapter 2: The Chemical Context of Life Name Period This chapter covers the basics that you may have learned in your chemistry class. Whether your teacher goes over this chapter, or assigns it for you
Name Date Class. energy phosphate adenine charged ATP chemical bonds work ribose
 Energy in a Cell Reinforcement and Study Guide Section.1 The Need for Energy In your textbook, read about cell energy. Use each of the terms below just once to complete the passage. energy phosphate adenine
Energy in a Cell Reinforcement and Study Guide Section.1 The Need for Energy In your textbook, read about cell energy. Use each of the terms below just once to complete the passage. energy phosphate adenine
5.1 Ecosystems, Energy, and Nutrients
 CHAPTER 5 ECOSYSTEMS 5.1 Ecosystems, Energy, and Nutrients Did anyone ever ask you the question: Where do you get your energy? Energy enters our world from the Sun but how does the Sun s energy become
CHAPTER 5 ECOSYSTEMS 5.1 Ecosystems, Energy, and Nutrients Did anyone ever ask you the question: Where do you get your energy? Energy enters our world from the Sun but how does the Sun s energy become
Keystone Review Practice Test Module A Cells and Cell Processes. 1. Which characteristic is shared by all prokaryotes and eukaryotes?
 Keystone Review Practice Test Module A Cells and Cell Processes 1. Which characteristic is shared by all prokaryotes and eukaryotes? a. Ability to store hereditary information b. Use of organelles to control
Keystone Review Practice Test Module A Cells and Cell Processes 1. Which characteristic is shared by all prokaryotes and eukaryotes? a. Ability to store hereditary information b. Use of organelles to control
7.1 What Are Cells? You are made of cells. A cell is the basic unit of structure and function in a living thing. CHAPTER 7
 CELL STRUCTURE AND FUNCTION 7.1 What Are Cells? Look closely at the skin on your arm. Can you see that it is made of cells? Of course not! Your skin cells are much too small to see with your eyes. Now
CELL STRUCTURE AND FUNCTION 7.1 What Are Cells? Look closely at the skin on your arm. Can you see that it is made of cells? Of course not! Your skin cells are much too small to see with your eyes. Now
Cellular Energy. 1. Photosynthesis is carried out by which of the following?
 Cellular Energy 1. Photosynthesis is carried out by which of the following? A. plants, but not animals B. animals, but not plants C. bacteria, but neither animals nor plants D. all living organisms 2.
Cellular Energy 1. Photosynthesis is carried out by which of the following? A. plants, but not animals B. animals, but not plants C. bacteria, but neither animals nor plants D. all living organisms 2.
4. Which carbohydrate would you find as part of a molecule of RNA? a. Galactose b. Deoxyribose c. Ribose d. Glucose
 1. How is a polymer formed from multiple monomers? a. From the growth of the chain of carbon atoms b. By the removal of an OH group and a hydrogen atom c. By the addition of an OH group and a hydrogen
1. How is a polymer formed from multiple monomers? a. From the growth of the chain of carbon atoms b. By the removal of an OH group and a hydrogen atom c. By the addition of an OH group and a hydrogen
BIOLOGICAL MOLECULES OF LIFE
 BIOLOGICAL MOLECULES OF LIFE C A R B O H Y D R A T E S, L I P I D S, P R O T E I N S, A N D N U C L E I C A C I D S The Academic Support Center @ Daytona State College (Science 115, Page 1 of 29) Carbon
BIOLOGICAL MOLECULES OF LIFE C A R B O H Y D R A T E S, L I P I D S, P R O T E I N S, A N D N U C L E I C A C I D S The Academic Support Center @ Daytona State College (Science 115, Page 1 of 29) Carbon
Chemical Bonds. Chemical Bonds. The Nature of Molecules. Energy and Metabolism < < Covalent bonds form when atoms share 2 or more valence electrons.
 The Nature of Molecules Chapter 2 Energy and Metabolism Chapter 6 Chemical Bonds Molecules are groups of atoms held together in a stable association. Compounds are molecules containing more than one type
The Nature of Molecules Chapter 2 Energy and Metabolism Chapter 6 Chemical Bonds Molecules are groups of atoms held together in a stable association. Compounds are molecules containing more than one type
Chapter 2 The Chemical Context of Life
 Chapter 2 The Chemical Context of Life Multiple-Choice Questions 1) About 25 of the 92 natural elements are known to be essential to life. Which four of these 25 elements make up approximately 96% of living
Chapter 2 The Chemical Context of Life Multiple-Choice Questions 1) About 25 of the 92 natural elements are known to be essential to life. Which four of these 25 elements make up approximately 96% of living
2. Which type of macromolecule contains high-energy bonds and is used for long-term energy storage?
 Energy Transport Study Island 1. During the process of photosynthesis, plants use energy from the Sun to convert carbon dioxide and water into glucose and oxygen. These products are, in turn, used by the
Energy Transport Study Island 1. During the process of photosynthesis, plants use energy from the Sun to convert carbon dioxide and water into glucose and oxygen. These products are, in turn, used by the
Chapter 2. The Chemistry of Life Worksheets
 Chapter 2 The Chemistry of Life Worksheets (Opening image courtesy of David Iberri, http://en.wikipedia.org/wiki/file:camkii.png, and under the Creative Commons license CC-BY-SA 3.0.) Lesson 2.1: Matter
Chapter 2 The Chemistry of Life Worksheets (Opening image courtesy of David Iberri, http://en.wikipedia.org/wiki/file:camkii.png, and under the Creative Commons license CC-BY-SA 3.0.) Lesson 2.1: Matter
Biological Science, 5e (Freeman) Chapter 1 Biology and the Tree of Life
 Biological Science, 5e (Freeman) Chapter 1 Biology and the Tree of Life 1) Pasteur s experiments proved that A) Cells cannot survive in swan necked flasks B) In order to grow, cells need to be supplied
Biological Science, 5e (Freeman) Chapter 1 Biology and the Tree of Life 1) Pasteur s experiments proved that A) Cells cannot survive in swan necked flasks B) In order to grow, cells need to be supplied
Cellular Respiration: Practice Questions #1
 Cellular Respiration: Practice Questions #1 1. Which statement best describes one of the events taking place in the chemical reaction? A. Energy is being stored as a result of aerobic respiration. B. Fermentation
Cellular Respiration: Practice Questions #1 1. Which statement best describes one of the events taking place in the chemical reaction? A. Energy is being stored as a result of aerobic respiration. B. Fermentation
Test Bank - Chapter 4 Multiple Choice
 Test Bank - Chapter 4 The questions in the test bank cover the concepts from the lessons in Chapter 4. Select questions from any of the categories that match the content you covered with students. The
Test Bank - Chapter 4 The questions in the test bank cover the concepts from the lessons in Chapter 4. Select questions from any of the categories that match the content you covered with students. The
Bioenergetics Module A Anchor 3
 Bioenergetics Module A Anchor 3 Key Concepts: - ATP can easily release and store energy by breaking and re-forming the bonds between its phosphate groups. This characteristic of ATP makes it exceptionally
Bioenergetics Module A Anchor 3 Key Concepts: - ATP can easily release and store energy by breaking and re-forming the bonds between its phosphate groups. This characteristic of ATP makes it exceptionally
How To Understand The Chemistry Of Organic Molecules
 CHAPTER 3 THE CHEMISTRY OF ORGANIC MOLECULES 3.1 Organic Molecules The chemistry of carbon accounts for the diversity of organic molecules found in living things. Carbon has six electrons, four of which
CHAPTER 3 THE CHEMISTRY OF ORGANIC MOLECULES 3.1 Organic Molecules The chemistry of carbon accounts for the diversity of organic molecules found in living things. Carbon has six electrons, four of which
Chapter 4. Photosynthesis and Cellular Respiration Worksheets. 63 www.ck12.org
 Chapter 4 Photosynthesis and Cellular Respiration Worksheets (Opening image copyright by Derek Ramsey, http://en.wikipedia.org/wiki/file:monarch_butterfly_ Danaus_plexippus_Feeding_Down_3008px.jpg, and
Chapter 4 Photosynthesis and Cellular Respiration Worksheets (Opening image copyright by Derek Ramsey, http://en.wikipedia.org/wiki/file:monarch_butterfly_ Danaus_plexippus_Feeding_Down_3008px.jpg, and
Name Class Date. binomial nomenclature. MAIN IDEA: Linnaeus developed the scientific naming system still used today.
 Section 1: The Linnaean System of Classification 17.1 Reading Guide KEY CONCEPT Organisms can be classified based on physical similarities. VOCABULARY taxonomy taxon binomial nomenclature genus MAIN IDEA:
Section 1: The Linnaean System of Classification 17.1 Reading Guide KEY CONCEPT Organisms can be classified based on physical similarities. VOCABULARY taxonomy taxon binomial nomenclature genus MAIN IDEA:
Chapter 5 Student Reading
 Chapter 5 Student Reading THE POLARITY OF THE WATER MOLECULE Wonderful water Water is an amazing substance. We drink it, cook and wash with it, swim and play in it, and use it for lots of other purposes.
Chapter 5 Student Reading THE POLARITY OF THE WATER MOLECULE Wonderful water Water is an amazing substance. We drink it, cook and wash with it, swim and play in it, and use it for lots of other purposes.
Cell and Membrane Practice. A. chromosome B. gene C. mitochondrion D. vacuole
 Name: ate: 1. Which structure is outside the nucleus of a cell and contains N?. chromosome. gene. mitochondrion. vacuole 2. potato core was placed in a beaker of water as shown in the figure below. Which
Name: ate: 1. Which structure is outside the nucleus of a cell and contains N?. chromosome. gene. mitochondrion. vacuole 2. potato core was placed in a beaker of water as shown in the figure below. Which
Energy Flow Through an Ecosystem. Food Chains, Food Webs, and Ecological Pyramids
 Energy Flow Through an Ecosystem Food Chains, Food Webs, and Ecological Pyramids What is Ecology? ECOLOGY is a branch of biology that studies ecosystems. Ecological Terminology Environment Ecology Biotic
Energy Flow Through an Ecosystem Food Chains, Food Webs, and Ecological Pyramids What is Ecology? ECOLOGY is a branch of biology that studies ecosystems. Ecological Terminology Environment Ecology Biotic
pathway that involves taking in heat from the environment at each step. C.
 Study Island Cell Energy Keystone Review 1. Cells obtain energy by either capturing light energy through photosynthesis or by breaking down carbohydrates through cellular respiration. In both photosynthesis
Study Island Cell Energy Keystone Review 1. Cells obtain energy by either capturing light energy through photosynthesis or by breaking down carbohydrates through cellular respiration. In both photosynthesis
DNA is found in all organisms from the smallest bacteria to humans. DNA has the same composition and structure in all organisms!
 Biological Sciences Initiative HHMI DNA omponents and Structure Introduction Nucleic acids are molecules that are essential to, and characteristic of, life on Earth. There are two basic types of nucleic
Biological Sciences Initiative HHMI DNA omponents and Structure Introduction Nucleic acids are molecules that are essential to, and characteristic of, life on Earth. There are two basic types of nucleic
Unit I: Introduction To Scientific Processes
 Unit I: Introduction To Scientific Processes This unit is an introduction to the scientific process. This unit consists of a laboratory exercise where students go through the QPOE2 process step by step
Unit I: Introduction To Scientific Processes This unit is an introduction to the scientific process. This unit consists of a laboratory exercise where students go through the QPOE2 process step by step
UNIT 2 PRACTICE EXAM (Part 1: General Chemistry)
 UIT 2 PRACTICE EXAM (Part 1: General Chemistry) 1. Which would be the best definition of an ionic bond? a. The attraction between the partial positive region of one molecule and the partial negative region
UIT 2 PRACTICE EXAM (Part 1: General Chemistry) 1. Which would be the best definition of an ionic bond? a. The attraction between the partial positive region of one molecule and the partial negative region
2. What kind of energy is stored in food? A. chemical energy B. heat energy C. kinetic energy D. light energy
 Assessment Bank Matter and Energy in Living Things SC.8.L.18.4 1. What is energy? A. anything that takes up space B. anything that has mass C. the ability to conduct current D. the ability to do work 2.
Assessment Bank Matter and Energy in Living Things SC.8.L.18.4 1. What is energy? A. anything that takes up space B. anything that has mass C. the ability to conduct current D. the ability to do work 2.
McDougal Littell Middle School Science
 McDougal Littell Middle School Science correlated to the Assessed Indicators, and the Assessed Indicators CC2 1/2004 2004 KS 49 correlated to the, and the Science Correlation Chart Strand Inquiry (INQ
McDougal Littell Middle School Science correlated to the Assessed Indicators, and the Assessed Indicators CC2 1/2004 2004 KS 49 correlated to the, and the Science Correlation Chart Strand Inquiry (INQ
Lecture Overview. Hydrogen Bonds. Special Properties of Water Molecules. Universal Solvent. ph Scale Illustrated. special properties of water
 Lecture Overview special properties of water > water as a solvent > ph molecules of the cell > properties of carbon > carbohydrates > lipids > proteins > nucleic acids Hydrogen Bonds polarity of water
Lecture Overview special properties of water > water as a solvent > ph molecules of the cell > properties of carbon > carbohydrates > lipids > proteins > nucleic acids Hydrogen Bonds polarity of water
A disaccharide is formed when a dehydration reaction joins two monosaccharides. This covalent bond is called a glycosidic linkage.
 CH 5 Structure & Function of Large Molecules: Macromolecules Molecules of Life All living things are made up of four classes of large biological molecules: carbohydrates, lipids, proteins, and nucleic
CH 5 Structure & Function of Large Molecules: Macromolecules Molecules of Life All living things are made up of four classes of large biological molecules: carbohydrates, lipids, proteins, and nucleic
The Cell Teaching Notes and Answer Keys
 The Cell Teaching Notes and Answer Keys Subject area: Science / Biology Topic focus: The Cell: components, types of cells, organelles, levels of organization Learning Aims: describe similarities and differences
The Cell Teaching Notes and Answer Keys Subject area: Science / Biology Topic focus: The Cell: components, types of cells, organelles, levels of organization Learning Aims: describe similarities and differences
Carbon-organic Compounds
 Elements in Cells The living substance of cells is made up of cytoplasm and the structures within it. About 96% of cytoplasm and its included structures are composed of the elements carbon, hydrogen, oxygen,
Elements in Cells The living substance of cells is made up of cytoplasm and the structures within it. About 96% of cytoplasm and its included structures are composed of the elements carbon, hydrogen, oxygen,
The Living Cell from the Biology: The Science of Life Series. Pre-Test
 1 Pre-Test Directions: Answer each question TRUE OR FALSE. 1. The instructions for making proteins are stored in molecules of DNA. 2. Proteins are made in the nucleus. 3. All cells are surrounded by a
1 Pre-Test Directions: Answer each question TRUE OR FALSE. 1. The instructions for making proteins are stored in molecules of DNA. 2. Proteins are made in the nucleus. 3. All cells are surrounded by a
Biological molecules:
 Biological molecules: All are organic (based on carbon). Monomers vs. polymers: Monomers refer to the subunits that, when polymerized, make up a larger polymer. Monomers may function on their own in some
Biological molecules: All are organic (based on carbon). Monomers vs. polymers: Monomers refer to the subunits that, when polymerized, make up a larger polymer. Monomers may function on their own in some
A Correlation of Pearson Miller & Levine Biology 2014 To the Utah Core State Standards for Biology Grades 9-12
 A Correlation of Pearson To the Utah Core State Standards Resource Title: Publisher: Pearson Education publishing as Prentice Hall ISBN (10 or 13 digit unique identifier is required): SE: 9780133242003
A Correlation of Pearson To the Utah Core State Standards Resource Title: Publisher: Pearson Education publishing as Prentice Hall ISBN (10 or 13 digit unique identifier is required): SE: 9780133242003
PHOTOSYNTHESIS AND CELLULAR RESPIRATION
 reflect Wind turbines shown in the photo on the right are large structures with blades that move in response to air movement. When the wind blows, the blades rotate. This motion generates energy that is
reflect Wind turbines shown in the photo on the right are large structures with blades that move in response to air movement. When the wind blows, the blades rotate. This motion generates energy that is
Chapter 9 Review Worksheet Cellular Respiration
 1 of 5 11/9/2011 8:11 PM Name: Hour: Chapter 9 Review Worksheet Cellular Respiration Energy in General 1. Differentiate an autotroph from a hetertroph as it relates to obtaining energy and the processes
1 of 5 11/9/2011 8:11 PM Name: Hour: Chapter 9 Review Worksheet Cellular Respiration Energy in General 1. Differentiate an autotroph from a hetertroph as it relates to obtaining energy and the processes
Chemical Basis of Life Module A Anchor 2
 Chemical Basis of Life Module A Anchor 2 Key Concepts: - Water is a polar molecule. Therefore, it is able to form multiple hydrogen bonds, which account for many of its special properties. - Water s polarity
Chemical Basis of Life Module A Anchor 2 Key Concepts: - Water is a polar molecule. Therefore, it is able to form multiple hydrogen bonds, which account for many of its special properties. - Water s polarity
Chapter 4: A Tour of the Cell. 1. Cell Basics. Limits to Cell Size. 1. Cell Basics. 2. Prokaryotic Cells. 3. Eukaryotic Cells
 Chapter 4: A Tour of the Cell 1. Cell Basics 2. Prokaryotic Cells 3. Eukaryotic Cells 1. Cell Basics Limits to Cell Size There are 2 main reasons why cells are so small: If cells get too large: 1) there
Chapter 4: A Tour of the Cell 1. Cell Basics 2. Prokaryotic Cells 3. Eukaryotic Cells 1. Cell Basics Limits to Cell Size There are 2 main reasons why cells are so small: If cells get too large: 1) there
But what about the prokaryotic cells?
 Chapter 32: Page 318 In the past two chapters, you have explored the organelles that can be found in both plant and animal s. You have also learned that plant s contain an organelle that is not found in
Chapter 32: Page 318 In the past two chapters, you have explored the organelles that can be found in both plant and animal s. You have also learned that plant s contain an organelle that is not found in
Mississippi SATP Biology I Student Review Guide
 Mississippi Student Review Guide Author: Cecilia L. Boles Published by Enrichment Plus, LLC PO Box 2755 Acworth, GA 30102 Toll Free: 1-800-745-4706 Fax 678-445-6702 Web site: www.enrichmentplus.com Mississippi
Mississippi Student Review Guide Author: Cecilia L. Boles Published by Enrichment Plus, LLC PO Box 2755 Acworth, GA 30102 Toll Free: 1-800-745-4706 Fax 678-445-6702 Web site: www.enrichmentplus.com Mississippi
Name: LAB SECTION: Circle your answer on the test sheet: completely erase or block out unwanted answers.
 Biology 100 Practice Final Exam Spring 2011 Name: LAB SECTION: Circle your answer on the test sheet: completely erase or block out unwanted answers. Once you have completed the exam, transfer your answers
Biology 100 Practice Final Exam Spring 2011 Name: LAB SECTION: Circle your answer on the test sheet: completely erase or block out unwanted answers. Once you have completed the exam, transfer your answers
Chapter 3. Cellular Structure and Function Worksheets. 39 www.ck12.org
 Chapter 3 Cellular Structure and Function Worksheets (Opening image copyright by Sebastian Kaulitzki, 2010. Used under license from Shutterstock.com.) Lesson 3.1: Introduction to Cells Lesson 3.2: Cell
Chapter 3 Cellular Structure and Function Worksheets (Opening image copyright by Sebastian Kaulitzki, 2010. Used under license from Shutterstock.com.) Lesson 3.1: Introduction to Cells Lesson 3.2: Cell
8.2 Cells and Energy. What is photosynthesis? Photosynthesis takes place in the chloroplasts. CHAPTER 8. Solar cells and chloroplasts
 CHAPTER 8 CELL PROCESSES 8.2 Cells and Energy To stay alive, you need a constant supply of energy. You need energy to move, think, grow, and even sleep. Where does that energy come from? It all starts
CHAPTER 8 CELL PROCESSES 8.2 Cells and Energy To stay alive, you need a constant supply of energy. You need energy to move, think, grow, and even sleep. Where does that energy come from? It all starts
Plant and Animal Cells
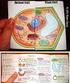 Plant and Animal Cells a. Explain that cells take in nutrients in order to grow, divide and to make needed materials. S7L2a b. Relate cell structures (cell membrane, nucleus, cytoplasm, chloroplasts, and
Plant and Animal Cells a. Explain that cells take in nutrients in order to grow, divide and to make needed materials. S7L2a b. Relate cell structures (cell membrane, nucleus, cytoplasm, chloroplasts, and
2007 7.013 Problem Set 1 KEY
 2007 7.013 Problem Set 1 KEY Due before 5 PM on FRIDAY, February 16, 2007. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. Where in a eukaryotic cell do you
2007 7.013 Problem Set 1 KEY Due before 5 PM on FRIDAY, February 16, 2007. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. Where in a eukaryotic cell do you
Fifth Grade Cells: Structures and Processes Assessment
 Fifth Grade Cells: Structures and Processes Assessment 1a. All living things are made up of. a. cells b. tissues c. organisms d. systems 1b. All living things are made up of. 1c. Explain what cells are
Fifth Grade Cells: Structures and Processes Assessment 1a. All living things are made up of. a. cells b. tissues c. organisms d. systems 1b. All living things are made up of. 1c. Explain what cells are
Introduction to the Cell: Plant and Animal Cells
 Introduction to the Cell: Plant and Animal Cells Tissues, Organs, and Systems of Living Things Cells, Cell Division, and Animal Systems and Plant Systems Cell Specialization Human Systems All organisms
Introduction to the Cell: Plant and Animal Cells Tissues, Organs, and Systems of Living Things Cells, Cell Division, and Animal Systems and Plant Systems Cell Specialization Human Systems All organisms
AP Biology Essential Knowledge Student Diagnostic
 AP Biology Essential Knowledge Student Diagnostic Background The Essential Knowledge statements provided in the AP Biology Curriculum Framework are scientific claims describing phenomenon occurring in
AP Biology Essential Knowledge Student Diagnostic Background The Essential Knowledge statements provided in the AP Biology Curriculum Framework are scientific claims describing phenomenon occurring in
Energy Flow in the Pond Teacher s Guide February 2011
 Energy Flow in the Pond Teacher s Guide February 2011 Grades: 6, 7 & 8 Time: 3 hours With the pond as a model, students explore how energy that originates from the sun keeps changing shape and form as
Energy Flow in the Pond Teacher s Guide February 2011 Grades: 6, 7 & 8 Time: 3 hours With the pond as a model, students explore how energy that originates from the sun keeps changing shape and form as
1. When applying the process of science, which of these is tested? a. an observation b. a result c. a hypothesis d. a question e.
 BCOR 11 Exam 1, 2004 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1. When applying the process of science, which of these is tested? a. an observation
BCOR 11 Exam 1, 2004 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1. When applying the process of science, which of these is tested? a. an observation
Cellular Structure and Function
 Chapter Test A CHAPTER 7 Cellular Structure and Function Part A: Multiple Choice In the space at the left, write the letter of the term or phrase that best answers each question. 1. Which defines a cell?
Chapter Test A CHAPTER 7 Cellular Structure and Function Part A: Multiple Choice In the space at the left, write the letter of the term or phrase that best answers each question. 1. Which defines a cell?
Endocrine System: Practice Questions #1
 Endocrine System: Practice Questions #1 1. Removing part of gland D would most likely result in A. a decrease in the secretions of other glands B. a decrease in the blood calcium level C. an increase in
Endocrine System: Practice Questions #1 1. Removing part of gland D would most likely result in A. a decrease in the secretions of other glands B. a decrease in the blood calcium level C. an increase in
Non-Covalent Bonds (Weak Bond)
 Non-Covalent Bonds (Weak Bond) Weak bonds are those forces of attraction that, in biological situations, do not take a large amount of energy to break. For example, hydrogen bonds are broken by energies
Non-Covalent Bonds (Weak Bond) Weak bonds are those forces of attraction that, in biological situations, do not take a large amount of energy to break. For example, hydrogen bonds are broken by energies
Name Date Period. 2. When a molecule of double-stranded DNA undergoes replication, it results in
 DNA, RNA, Protein Synthesis Keystone 1. During the process shown above, the two strands of one DNA molecule are unwound. Then, DNA polymerases add complementary nucleotides to each strand which results
DNA, RNA, Protein Synthesis Keystone 1. During the process shown above, the two strands of one DNA molecule are unwound. Then, DNA polymerases add complementary nucleotides to each strand which results
Name: Hour: Elements & Macromolecules in Organisms
 Name: Hour: Elements & Macromolecules in Organisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds
Name: Hour: Elements & Macromolecules in Organisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds
In the box below, draw the Lewis electron-dot structure for the compound formed from magnesium and oxygen. [Include any charges or partial charges.
 Name: 1) Which molecule is nonpolar and has a symmetrical shape? A) NH3 B) H2O C) HCl D) CH4 7222-1 - Page 1 2) When ammonium chloride crystals are dissolved in water, the temperature of the water decreases.
Name: 1) Which molecule is nonpolar and has a symmetrical shape? A) NH3 B) H2O C) HCl D) CH4 7222-1 - Page 1 2) When ammonium chloride crystals are dissolved in water, the temperature of the water decreases.
Honors Biology Course Summary Department: Science
 Honors Biology Course Summary Department: Science Semester 1 Learning Objective #1 - Ecology Students will understand how organisms interact with each other and the environment. Target(s) to Meet Learning
Honors Biology Course Summary Department: Science Semester 1 Learning Objective #1 - Ecology Students will understand how organisms interact with each other and the environment. Target(s) to Meet Learning
Chapter 3 Molecules of Cells
 Bio 100 Molecules of cells 1 Chapter 3 Molecules of Cells Compounds containing carbon are called organic compounds Molecules such as methane that are only composed of carbon and hydrogen are called hydrocarbons
Bio 100 Molecules of cells 1 Chapter 3 Molecules of Cells Compounds containing carbon are called organic compounds Molecules such as methane that are only composed of carbon and hydrogen are called hydrocarbons
KEY CONCEPT Organisms can be classified based on physical similarities. binomial nomenclature
 Section 17.1: The Linnaean System of Classification Unit 9 Study Guide KEY CONCEPT Organisms can be classified based on physical similarities. VOCABULARY taxonomy taxon binomial nomenclature genus MAIN
Section 17.1: The Linnaean System of Classification Unit 9 Study Guide KEY CONCEPT Organisms can be classified based on physical similarities. VOCABULARY taxonomy taxon binomial nomenclature genus MAIN
8.3 The Process of Photosynthesis
 8.3 The Process of Photosynthesis Lesson Objectives Describe what happens during the light-dependent reactions. Describe what happens during the light-independent reactions. Identify factors that affect
8.3 The Process of Photosynthesis Lesson Objectives Describe what happens during the light-dependent reactions. Describe what happens during the light-independent reactions. Identify factors that affect
CHAPTER 1 INTRODUCTION: THEMES IN THE STUDY OF LIFE
 CHAPTER 1 INTRODUCTION: THEMES IN THE STUDY OF LIFE OUTLINE I. Life s Hierarchical Order A. The living world is a hierarchy, with each level of biological structure building on the level below it B. Each
CHAPTER 1 INTRODUCTION: THEMES IN THE STUDY OF LIFE OUTLINE I. Life s Hierarchical Order A. The living world is a hierarchy, with each level of biological structure building on the level below it B. Each
The Properties of Water
 1 Matter & Energy: Properties of Water, ph, Chemical Reactions EVPP 110 Lecture GMU Dr. Largen Fall 2003 2 The Properties of Water 3 Water - Its Properties and Its Role in the Fitness of Environment importance
1 Matter & Energy: Properties of Water, ph, Chemical Reactions EVPP 110 Lecture GMU Dr. Largen Fall 2003 2 The Properties of Water 3 Water - Its Properties and Its Role in the Fitness of Environment importance
ATOMS A T O M S, I S O T O P E S, A N D I O N S. The Academic Support Center @ Daytona State College (Science 120, Page 1 of 39)
 ATOMS A T O M S, I S O T O P E S, A N D I O N S The Academic Support Center @ Daytona State College (Science 120, Page 1 of 39) THE ATOM All elements listed on the periodic table are made up of atoms.
ATOMS A T O M S, I S O T O P E S, A N D I O N S The Academic Support Center @ Daytona State College (Science 120, Page 1 of 39) THE ATOM All elements listed on the periodic table are made up of atoms.
Basic Biological Principles Module A Anchor 1
 Basic Biological Principles Module A Anchor 1 Key Concepts: - Living things are made of units called cells, are based on a universal genetic code, obtain and use materials and energy, grow and develop,
Basic Biological Principles Module A Anchor 1 Key Concepts: - Living things are made of units called cells, are based on a universal genetic code, obtain and use materials and energy, grow and develop,
Preliminary MFM Quiz
 Preliminary MFM Quiz 1. The major carrier of chemical energy in all cells is: A) adenosine monophosphate B) adenosine diphosphate C) adenosine trisphosphate D) guanosine trisphosphate E) carbamoyl phosphate
Preliminary MFM Quiz 1. The major carrier of chemical energy in all cells is: A) adenosine monophosphate B) adenosine diphosphate C) adenosine trisphosphate D) guanosine trisphosphate E) carbamoyl phosphate
XII. Biology, Grade 10
 XII. Biology, Grade 10 Grade 10 Biology Pilot Test The spring 2004 Grade 10 MCAS Biology Test was based on learning standards in the Biology content strand of the Massachusetts Science and Technology/Engineering
XII. Biology, Grade 10 Grade 10 Biology Pilot Test The spring 2004 Grade 10 MCAS Biology Test was based on learning standards in the Biology content strand of the Massachusetts Science and Technology/Engineering
Prentice Hall Biology (Miller/Levine) 2008 Correlated to: Colorado Science Standards and Benchmarks (Grades 9-12)
 1. ask questions and state hypotheses using prior scientific knowledge to help design and guide their development and implementation of a scientific investigation STANDARD 1: Students apply the processes
1. ask questions and state hypotheses using prior scientific knowledge to help design and guide their development and implementation of a scientific investigation STANDARD 1: Students apply the processes
Visualizing Cell Processes
 Visualizing Cell Processes A Series of Five Programs produced by BioMEDIA ASSOCIATES Content Guide for Program 3 Photosynthesis and Cellular Respiration Copyright 2001, BioMEDIA ASSOCIATES www.ebiomedia.com
Visualizing Cell Processes A Series of Five Programs produced by BioMEDIA ASSOCIATES Content Guide for Program 3 Photosynthesis and Cellular Respiration Copyright 2001, BioMEDIA ASSOCIATES www.ebiomedia.com
Chapter 5. The Structure and Function of Macromolecule s
 Chapter 5 The Structure and Function of Macromolecule s Most Macromolecules are polymers: Polymer: (poly: many; mer: part) Large molecules consisting of many identical or similar subunits connected together.
Chapter 5 The Structure and Function of Macromolecule s Most Macromolecules are polymers: Polymer: (poly: many; mer: part) Large molecules consisting of many identical or similar subunits connected together.
7.2 Cell Structure. Lesson Objectives. Lesson Summary. Cell Organization Eukaryotic cells contain a nucleus and many specialized structures.
 7.2 Cell Structure Lesson Objectives Describe the structure and function of the cell nucleus. Describe the role of vacuoles, lysosomes, and the cytoskeleton. Identify the role of ribosomes, endoplasmic
7.2 Cell Structure Lesson Objectives Describe the structure and function of the cell nucleus. Describe the role of vacuoles, lysosomes, and the cytoskeleton. Identify the role of ribosomes, endoplasmic
7.4. Using the Bohr Theory KNOW? Using the Bohr Theory to Describe Atoms and Ions
 7.4 Using the Bohr Theory LEARNING TIP Models such as Figures 1 to 4, on pages 218 and 219, help you visualize scientific explanations. As you examine Figures 1 to 4, look back and forth between the diagrams
7.4 Using the Bohr Theory LEARNING TIP Models such as Figures 1 to 4, on pages 218 and 219, help you visualize scientific explanations. As you examine Figures 1 to 4, look back and forth between the diagrams
Objectives. PAM1014 Introduction to Radiation Physics. Constituents of Atoms. Atoms. Atoms. Atoms. Basic Atomic Theory
 PAM1014 Introduction to Radiation Physics Basic Atomic Theory Objectives Introduce and Molecules The periodic Table Electronic Energy Levels Atomic excitation & de-excitation Ionisation Molecules Constituents
PAM1014 Introduction to Radiation Physics Basic Atomic Theory Objectives Introduce and Molecules The periodic Table Electronic Energy Levels Atomic excitation & de-excitation Ionisation Molecules Constituents
Molecular Models in Biology
 Molecular Models in Biology Objectives: After this lab a student will be able to: 1) Understand the properties of atoms that give rise to bonds. 2) Understand how and why atoms form ions. 3) Model covalent,
Molecular Models in Biology Objectives: After this lab a student will be able to: 1) Understand the properties of atoms that give rise to bonds. 2) Understand how and why atoms form ions. 3) Model covalent,
Cells, tissues and organs
 Chapter 8: Cells, tissues and organs Cells: building blocks of life Living things are made of cells. Many of the chemical reactions that keep organisms alive (metabolic functions) take place in cells.
Chapter 8: Cells, tissues and organs Cells: building blocks of life Living things are made of cells. Many of the chemical reactions that keep organisms alive (metabolic functions) take place in cells.
