-1- BIOS Fall, 2009 Exam I, 18 Sept, 2008 Michael Muller, Instructor
|
|
|
- Gary Leonard Day
- 10 years ago
- Views:
Transcription
1 BIOS Fall, 2009 Exam I, 18 Sept, 2008 Michael Muller, Instructor Name: TA: This exam consists of 42 questions over 7 pages (the last page of which has the periodic table and Bloom s hierarchy). Please check to see that all the pages are present before you begin. There are some useful diagrams on the last page - check them out before you start taking the exam. Use a #2 pencil and bubble in all answers. Your score will be posted on the UIC Blackboard site as soon as they are in. You will gain four extra credit points if you bubble in your last name first. Good Luck! 1. How does the word theory in science differ from its use in everyday English? A. There is no difference the usages are interchangeable. B. A scientific theory is always right and never changes. C. Scientific theories are testable explanations, not speculative guesses. D. A scientific theory can be confirmed by a single experiment designed to prove its accuracy. 2. A friend of yours calls to say that his car would not start this morning. He asks for your help. You tell him the battery must be dead, and that if so, then jump-starting the car from a good battery should solve the problem. In doing so, you are. A. summarizing observations about why the car won t start. B. stating a scientific theory for why the car won t start. C. offering a specific hypothesis and associated prediction to explain why the car won t start. D. performing an experimental test of a hypothesis for why the car won t start. 3. When the atoms involved in a covalent bond have the same electronegativity, what type of bond results? A. hydrogen bond B. ionic bond C. polar covalent bond D. nonpolar covalent bond 4. Which of the levels of protein organization is correctly paired with the interaction that might be responsible for producing the structure? A. primary structure - ionic bonding between different amino acids B. secondary structure - hydrogen bonding between elements of the protein backbone C. tertiary structure - polar interactions between hydrophobic amino acids D. quaternary structure - disulfide bonds between different cysteine residues in the same polypeptide 5. What chemical property of RNA makes it much more likely to degrade than DNA? A. RNA molecules use the base uracil instead of thymine. B. RNA molecules cannot form secondary structures with antiparallel strands. C. RNA nucleotides have additional phosphate groups between the 5-carbon sugars. D. RNA nucleotides have a 2' hydroxyl (-OH) group. -1-
2 6. Which one of the following predictions follows from the sexual selection hypothesis for why giraffes have long necks? A. In contests over females, the best-nourished male should always, or almost always, win. B. In contests over females, the male with the longest neck should have an advantage over the other males. C. In natural populations, female neck length should decline over time. D. Young males that are given extra amounts of high-quality food should grow particularly long, strong necks. 7. Which of the following is a powerful way to test a hypothesis? A. Incorporate the hypothesis into a more general theory. B. Formulate a competing or alternative hypothesis. C. Formulate a null hypothesis. D. Perform an experiment that tests a prediction that follows from the hypothesis. 8. An atom has six electrons in its valence shell. How many single covalent bonds would you expect it to form in most circumstances? A. one B. two C. three D. six 9. The difference between a polar covalent bond and an ionic bond is that electrons are shared unequally in a polar covalent bond but completely transferred (i.e., not shared) in an ionic bond. A. True B. False 10. Which of the following true statements can be attributed to water's high specific heat? A. Oil and water do not mix well. B. Our body temperature takes a long time to change because our body is composed mostly of water. C. Ice floats on water. D. Sugar dissolves in hot tea faster than in iced tea. 11. The cities of Portland, Oregon, and Minneapolis, Minnesota, are at about the same latitude, but Minneapolis has much hotter summers and much colder winters than Portland does. Why? (Portland is near the Pacific Ocean; Minneapolis is near a number of large lakes.) A. They are not at exactly the same latitude. B. The ocean is so large that it has a highly moderating influence on temperature. C. Freshwater is more likely to freeze than saltwater. D. Minneapolis is much windier, due to its location in the middle of a continent. 12. The molecule diagramed below is an example of what type of biological molecule? A. Phospholipid B. Steroid C. Amino acid D. Nucleotide E. ATP -2-
3 13. Question #12 is asking for what type of learning? A. Lower-order learning (Knowledge & Understanding) B. Higher-order learning (Applying & Analyzing) C. Higher-order learning (Evaluating & Creating) D. None of the above 14. In which of the following molecules is the Carbon the most reduced? A. CO B. CH OH C. C H D. HCN E. C H O Why are most biologically important molecules polymers? A. Polymers are simple molecules B. Each polymer has a common bond type joining each monomer. This makes it easy to mix and match to create a large number of different molecules C. The monomers are energetically cheap to produce D. Polymers are very unstable, which makes them highly reactive Because you demanded it, matching on a diagram! Use the key on the right to properly identify the features on the diagram. A. Mitochondria - 18 B. Rough ER - 16 C. Smooth ER D. Golgi Apparatus - 17 E. Chloroplast 19. Which of the following statements (A-D) about enzymes is FALSE? If statements A-D are true, then choose E. A. Enzymes are not consumed in a reaction B. Enzymes lower the activation energy of both the forward and the reverse reactions C. Human enzymes typically operate under a narrow range of temperatures and ph s D. Substrates bind to the enzyme forming a temporary enzyme-substrate complex E. All of the above statements about enzymes are TRUE 20. Plant cells contain functional mitochondria A. True B. False -3-
4 21. The reaction of A + B >> C + D is exothermic. There is an enzyme which speeds up the reaction, but the reaction can proceed without the aid of a catalyst. Which statement below is TRUE? A. The enzyme-catalyzed reaction will release more energy than non-enzyme-catalyzed reaction B. The non-enzyme-catalyzed reaction will release more energy than enzyme catalyzed reaction C. Both the enzyme-catalyzed and the non-enzyme-catalyzed reactions will release the same amount of energy 22. The graph below on the left shows the relationship between the natural log of a mammals body mass and its temperature. Basically, as a mammals mass increases, its body temperature decreases. All mammals possess the enzyme alcohol dehydrogenase. Based on this information, predict which enzyme corresponds to each organism in the graph on the below left. A. X = mouse, Y = human, Z = elephant B. X = mouse, Y = elephant, Z = human C. X = elephant, Y = mouse, Z = human D. X = elephant, Y = human, Z = mouse 23. Why do most biological enzymes have a ph optimum? A. The ph affects the substrate s ability to react and form the product B. Changes in ph affect the enzyme s secondary and tertiary structures, which affect enzyme activity C. ATP is destroyed when the ph is too high or too low D. Changes in ph often affect the temperature, so there is a ph optimum because there is a temperature optimum 24. Which of the following is not found in all cells? A. Nucleus B. Ribosomes C. Cytoplasm D. Plasma membrane E. All of the above are found in all cells -4-
5 25. Which cell structure is incorrectly matched with its function? A. Nucleolis storage of genetic information B. Rough ER protein synthesis C. Smooth ER lipid syntheis D. Lysosome destruction and recycling of old organelles and macromolecules E. Plant Central Vacuole storage 26. Propose a function for cells that contain extensive rough endoplasmic reticulum A. Production and processing of fatty acids and steroids B. Movement of the cell by cell crawling C. Storage of large amounts of starch D. Production of large amounts of muscle protein after exercise 27. What would happen to a cell that had a mutation in the gene which produces nuclear localization signals (NLS)? A. It would not be able to distribute proteins in the Golgi Apparatus B. It would not be able to produce lipids C. It would not be able to synthesize ribosomes D. It would not be able to produce the cytoskeleton E. It would not be able to move molecules into or out of the nucleus 28. Which of the following is NOT evidence in support of the endosymbiosis theory of the origin of the mitochondria and chloroplast? A. Mitochondria and chloroplasts have archaean ribosomes B. Mitochondria and chloroplasts divide in a process very similar to binary fission C. Mitochondria and chloroplasts are approximately the size of a prokaryotic cell D. Mitochondria and chloroplasts have naked DNA E. All of the above are evidence in support of the endosymbiosis theory of the origin of the mitochondria and chloroplasts. 29. Cooking oil and gasoline (a hydrocarbon) are not amphipathic molecules. Why? A. They do not have a polar or charged region. B. They do not have a hydrophobic region. C. They are highly reduced molecules. D. They spontaneously form micelles or liposomes in solution. 30. Which of the following best describes a biological membrane? A. A lipid bilayer with peripheral proteins on the outside and inside surfaces B. A lipid monolayer with asymmetrical distribution of integral and peripheral proteins C. A lipid bilayer with a symmetrical distribution of integral and peripheral proteins D. A lipid bilayer with an asymmetrical distribution of integral and peripheral proteins -5-
6 31. You have just discovered an organism that lives in extremely cold environments. Which of the following would you predict to be true about the phospholipids in its membranes, compared to phospholipids in the membranes of organisms that live in warmer environments? A. The membrane phospholipids of cold-adapted organisms will have longer hydrocarbon tails. B. The membrane phospholipids of cold-adapted organisms will have more saturated hydrocarbon tails. C. The membrane phospholipids of cold-adapted organisms will have more unsaturated hydrocarbon tails. 32. What do you predict about the cholesterol levels in the membranes in the above organism when compared to a similar organism that lives in the tropics? A. The cholesterol levels will be greater in the organism that lives in the colder environments than in the organism that lives in the tropics B. The cholesterol levels will be lower in the organism that lives in the colder environments than in the organism that lives in the tropics C. The cholesterol levels will be the same in the organism that lives in the colder environments and in the organism that lives in the tropics 33. If a plasma membrane is punctured with a small object, the hole will close up and the membrane will be as good as new. Why? A. Proteins on the outer surface are responsible for membrane healing B. The cytoskeleton is responsible for membrane healing C. The amphipathic phospholipids will be moved by the water molecules and heal the membrane wound D. The Golgi apparatus will secrete versicles that will heal the wound 34. Insulin is a protein that will be produced and secreted by a pancreatic cell. Actin is a protein produced by a pancreatic cell used in formation of the the cytoskeleton. Which of the following statements about the production of these two proteins in a pancreatic cell is TRUE? A. Both the actin and the insulin proteins will be glycolated when first produced B. Both the actin and the insulin proteins will be produced in ribosomes floating free in the cytoplasm, not on ribosomes bound to the ER C. Both actin and insulin will be sorted in the Golgi Apparatus D. Only the insulin will be glycolated, the actin will not be glycolated are TRUE. 35. Which of the reasons given below was NOT given by the judge in the Kitzmiller v Dover court case for why Intelligent Design (ID) is not science? A. ID violated the centuries-old ground rules of science by invoking and permitting supernatural causation for natural phenomena B. the argument of irreducible complexity, central to ID, employs the same flawed and illogical contrived dualism that doomed creation science C. ID s negative attacks on evolution have been refuted by the scientific community D. All of ID s claims have not been scientifically demonstrated E. All of the above reasons were cited by the judge in the Kitzmiller v Dover case -6-
7 36. One reason why supernatural arguments for phenomena have not been accepted by most scientists is because you cannot see, replicate, control or test supernatural phenomena, so they lie outside of the realms of science. A. True B. False 37. What is a major difference in the extracellular matrix (ECM) between plant and animal cells? A. Plant ECM is composed primarily of proteins, whereas animal ECM is mainly carbohydrates. B. Plant ECM is primarily carbohydrate in nature, whereas animal ECM is largely protein based. C. Plant and animal ECMs are quite similar in structure and function. D. Plant ECM components are released extracellularly by the Golgi stacks, whereas lysosomes function in this capacity in animal cells. 38. Flagella and cilia bend or move, imparting mobility to cells. How do these structures move? A. The basal body at the base of the structure hydrolyzes ATP, causing a conformational change that results in movement of the cilium or flagellum. B. Two microtubules at the core of the structure serve as motor proteins. C. Axonemes are structured such that movement is constant. D. Dynein is a motor protein that hydrolyzes ATP and is responsible for movement of the cilium or flagellum. 39. Question #38 is asking for what type of learning? A. Lower-order learning (Knowledge & Understanding) B. Higher-order learning (Applying & Analyzing) C. Higher-order learning (Evaluating & Creating) D. None of the above 40. In lab, you had to prepare a series of known concentrations, measure their absorbency, and then prepare a standard curve. You could then use this standard curve to answer questions about the concentration of your unknown. This process utilizes what kind of learning? A. Lower-order learning (Knowledge & Understanding) B. Higher-order learning (Applying & Analyzing) C. Higher-order learning (Evaluating & Creating) D. None of the above 41. The FBI continues to study the behavior of serial killers and add the data to their profiles for use in predicting the behavior of future killers. The generation of this database is what kind of process? A. An inductive process B. A deductive process 42. Everyone will get credit for this one, but only if you answer (and be honest - I m collecting data). How many different lecture captures had you listened to BEFORE lecture on Wednesday? A. 0 B. 1 C. 2 D. 3-4 E
Anatomy and Physiology Placement Exam 2 Practice with Answers at End!
 Anatomy and Physiology Placement Exam 2 Practice with Answers at End! General Chemical Principles 1. bonds are characterized by the sharing of electrons between the participating atoms. a. hydrogen b.
Anatomy and Physiology Placement Exam 2 Practice with Answers at End! General Chemical Principles 1. bonds are characterized by the sharing of electrons between the participating atoms. a. hydrogen b.
2007 7.013 Problem Set 1 KEY
 2007 7.013 Problem Set 1 KEY Due before 5 PM on FRIDAY, February 16, 2007. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. Where in a eukaryotic cell do you
2007 7.013 Problem Set 1 KEY Due before 5 PM on FRIDAY, February 16, 2007. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. Where in a eukaryotic cell do you
A disaccharide is formed when a dehydration reaction joins two monosaccharides. This covalent bond is called a glycosidic linkage.
 CH 5 Structure & Function of Large Molecules: Macromolecules Molecules of Life All living things are made up of four classes of large biological molecules: carbohydrates, lipids, proteins, and nucleic
CH 5 Structure & Function of Large Molecules: Macromolecules Molecules of Life All living things are made up of four classes of large biological molecules: carbohydrates, lipids, proteins, and nucleic
4. Which carbohydrate would you find as part of a molecule of RNA? a. Galactose b. Deoxyribose c. Ribose d. Glucose
 1. How is a polymer formed from multiple monomers? a. From the growth of the chain of carbon atoms b. By the removal of an OH group and a hydrogen atom c. By the addition of an OH group and a hydrogen
1. How is a polymer formed from multiple monomers? a. From the growth of the chain of carbon atoms b. By the removal of an OH group and a hydrogen atom c. By the addition of an OH group and a hydrogen
NO CALCULATORS OR CELL PHONES ALLOWED
 Biol 205 Exam 1 TEST FORM A Spring 2008 NAME Fill out both sides of the Scantron Sheet. On Side 2 be sure to indicate that you have TEST FORM A The answers to Part I should be placed on the SCANTRON SHEET.
Biol 205 Exam 1 TEST FORM A Spring 2008 NAME Fill out both sides of the Scantron Sheet. On Side 2 be sure to indicate that you have TEST FORM A The answers to Part I should be placed on the SCANTRON SHEET.
Carbohydrates, proteins and lipids
 Carbohydrates, proteins and lipids Chapter 3 MACROMOLECULES Macromolecules: polymers with molecular weights >1,000 Functional groups THE FOUR MACROMOLECULES IN LIFE Molecules in living organisms: proteins,
Carbohydrates, proteins and lipids Chapter 3 MACROMOLECULES Macromolecules: polymers with molecular weights >1,000 Functional groups THE FOUR MACROMOLECULES IN LIFE Molecules in living organisms: proteins,
Review of the Cell and Its Organelles
 Biology Learning Centre Review of the Cell and Its Organelles Tips for most effective learning of this material: Memorize the names and structures over several days. This will help you retain what you
Biology Learning Centre Review of the Cell and Its Organelles Tips for most effective learning of this material: Memorize the names and structures over several days. This will help you retain what you
Lecture Overview. Hydrogen Bonds. Special Properties of Water Molecules. Universal Solvent. ph Scale Illustrated. special properties of water
 Lecture Overview special properties of water > water as a solvent > ph molecules of the cell > properties of carbon > carbohydrates > lipids > proteins > nucleic acids Hydrogen Bonds polarity of water
Lecture Overview special properties of water > water as a solvent > ph molecules of the cell > properties of carbon > carbohydrates > lipids > proteins > nucleic acids Hydrogen Bonds polarity of water
Keystone Review Practice Test Module A Cells and Cell Processes. 1. Which characteristic is shared by all prokaryotes and eukaryotes?
 Keystone Review Practice Test Module A Cells and Cell Processes 1. Which characteristic is shared by all prokaryotes and eukaryotes? a. Ability to store hereditary information b. Use of organelles to control
Keystone Review Practice Test Module A Cells and Cell Processes 1. Which characteristic is shared by all prokaryotes and eukaryotes? a. Ability to store hereditary information b. Use of organelles to control
Chemical Basis of Life Module A Anchor 2
 Chemical Basis of Life Module A Anchor 2 Key Concepts: - Water is a polar molecule. Therefore, it is able to form multiple hydrogen bonds, which account for many of its special properties. - Water s polarity
Chemical Basis of Life Module A Anchor 2 Key Concepts: - Water is a polar molecule. Therefore, it is able to form multiple hydrogen bonds, which account for many of its special properties. - Water s polarity
Chapter 2: Cell Structure and Function pg. 70-107
 UNIT 1: Biochemistry Chapter 2: Cell Structure and Function pg. 70-107 Organelles are internal structures that carry out specialized functions, interacting and complementing each other. Animal and plant
UNIT 1: Biochemistry Chapter 2: Cell Structure and Function pg. 70-107 Organelles are internal structures that carry out specialized functions, interacting and complementing each other. Animal and plant
1. When applying the process of science, which of these is tested? a. an observation b. a result c. a hypothesis d. a question e.
 BCOR 11 Exam 1, 2004 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1. When applying the process of science, which of these is tested? a. an observation
BCOR 11 Exam 1, 2004 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1. When applying the process of science, which of these is tested? a. an observation
Chapter 3 Molecules of Cells
 Bio 100 Molecules of cells 1 Chapter 3 Molecules of Cells Compounds containing carbon are called organic compounds Molecules such as methane that are only composed of carbon and hydrogen are called hydrocarbons
Bio 100 Molecules of cells 1 Chapter 3 Molecules of Cells Compounds containing carbon are called organic compounds Molecules such as methane that are only composed of carbon and hydrogen are called hydrocarbons
Disaccharides consist of two monosaccharide monomers covalently linked by a glycosidic bond. They function in sugar transport.
 1. The fundamental life processes of plants and animals depend on a variety of chemical reactions that occur in specialized areas of the organism s cells. As a basis for understanding this concept: 1.
1. The fundamental life processes of plants and animals depend on a variety of chemical reactions that occur in specialized areas of the organism s cells. As a basis for understanding this concept: 1.
Chapter 5. The Structure and Function of Macromolecule s
 Chapter 5 The Structure and Function of Macromolecule s Most Macromolecules are polymers: Polymer: (poly: many; mer: part) Large molecules consisting of many identical or similar subunits connected together.
Chapter 5 The Structure and Function of Macromolecule s Most Macromolecules are polymers: Polymer: (poly: many; mer: part) Large molecules consisting of many identical or similar subunits connected together.
Cell Structure & Function!
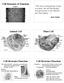 Cell Structure & Function! Chapter 3! The most exciting phrase to hear in science, the one that heralds new discoveries, is not 'Eureka!' but 'That's funny.! -- Isaac Asimov Animal Cell Plant Cell Cell
Cell Structure & Function! Chapter 3! The most exciting phrase to hear in science, the one that heralds new discoveries, is not 'Eureka!' but 'That's funny.! -- Isaac Asimov Animal Cell Plant Cell Cell
Plasma Membrane hydrophilic polar heads
 The Parts of the Cell 3 main parts in ALL cells: plasma membrane, cytoplasm, genetic material this is about the parts of a generic eukaryotic cell Plasma Membrane -is a fluid mosaic model membrane is fluid
The Parts of the Cell 3 main parts in ALL cells: plasma membrane, cytoplasm, genetic material this is about the parts of a generic eukaryotic cell Plasma Membrane -is a fluid mosaic model membrane is fluid
How To Understand The Chemistry Of Organic Molecules
 CHAPTER 3 THE CHEMISTRY OF ORGANIC MOLECULES 3.1 Organic Molecules The chemistry of carbon accounts for the diversity of organic molecules found in living things. Carbon has six electrons, four of which
CHAPTER 3 THE CHEMISTRY OF ORGANIC MOLECULES 3.1 Organic Molecules The chemistry of carbon accounts for the diversity of organic molecules found in living things. Carbon has six electrons, four of which
Lecture 4 Cell Membranes & Organelles
 Lecture 4 Cell Membranes & Organelles Structure of Animal Cells The Phospholipid Structure Phospholipid structure Encases all living cells Its basic structure is represented by the fluidmosaic model Phospholipid
Lecture 4 Cell Membranes & Organelles Structure of Animal Cells The Phospholipid Structure Phospholipid structure Encases all living cells Its basic structure is represented by the fluidmosaic model Phospholipid
Name Date Period. 2. When a molecule of double-stranded DNA undergoes replication, it results in
 DNA, RNA, Protein Synthesis Keystone 1. During the process shown above, the two strands of one DNA molecule are unwound. Then, DNA polymerases add complementary nucleotides to each strand which results
DNA, RNA, Protein Synthesis Keystone 1. During the process shown above, the two strands of one DNA molecule are unwound. Then, DNA polymerases add complementary nucleotides to each strand which results
Preliminary MFM Quiz
 Preliminary MFM Quiz 1. The major carrier of chemical energy in all cells is: A) adenosine monophosphate B) adenosine diphosphate C) adenosine trisphosphate D) guanosine trisphosphate E) carbamoyl phosphate
Preliminary MFM Quiz 1. The major carrier of chemical energy in all cells is: A) adenosine monophosphate B) adenosine diphosphate C) adenosine trisphosphate D) guanosine trisphosphate E) carbamoyl phosphate
Chapter 5: The Structure and Function of Large Biological Molecules
 Name Period Concept 5.1 Macromolecules are polymers, built from monomers 1. The large molecules of all living things fall into just four main classes. Name them. 2. Circle the three classes that are called
Name Period Concept 5.1 Macromolecules are polymers, built from monomers 1. The large molecules of all living things fall into just four main classes. Name them. 2. Circle the three classes that are called
Unit 2: Cells, Membranes and Signaling CELL MEMBRANE. Chapter 5 Hillis Textbook
 Unit 2: Cells, Membranes and Signaling CELL MEMBRANE Chapter 5 Hillis Textbook HOW DOES THE LAB RELATE TO THE NEXT CHAPTER? SURFACE AREA: the entire outer covering of a cell that enables materials pass.
Unit 2: Cells, Membranes and Signaling CELL MEMBRANE Chapter 5 Hillis Textbook HOW DOES THE LAB RELATE TO THE NEXT CHAPTER? SURFACE AREA: the entire outer covering of a cell that enables materials pass.
Student name ID # 2. (4 pts) What is the terminal electron acceptor in respiration? In photosynthesis? O2, NADP+
 1. Membrane transport. A. (4 pts) What ion couples primary and secondary active transport in animal cells? What ion serves the same function in plant cells? Na+, H+ 2. (4 pts) What is the terminal electron
1. Membrane transport. A. (4 pts) What ion couples primary and secondary active transport in animal cells? What ion serves the same function in plant cells? Na+, H+ 2. (4 pts) What is the terminal electron
7.2 Cell Structure. Lesson Objectives. Lesson Summary. Cell Organization Eukaryotic cells contain a nucleus and many specialized structures.
 7.2 Cell Structure Lesson Objectives Describe the structure and function of the cell nucleus. Describe the role of vacuoles, lysosomes, and the cytoskeleton. Identify the role of ribosomes, endoplasmic
7.2 Cell Structure Lesson Objectives Describe the structure and function of the cell nucleus. Describe the role of vacuoles, lysosomes, and the cytoskeleton. Identify the role of ribosomes, endoplasmic
The Molecules of Cells
 The Molecules of Cells I. Introduction A. Most of the world s population cannot digest milk-based foods. 1. These people are lactose intolerant because they lack the enzyme lactase. 2. This illustrates
The Molecules of Cells I. Introduction A. Most of the world s population cannot digest milk-based foods. 1. These people are lactose intolerant because they lack the enzyme lactase. 2. This illustrates
Carbon-organic Compounds
 Elements in Cells The living substance of cells is made up of cytoplasm and the structures within it. About 96% of cytoplasm and its included structures are composed of the elements carbon, hydrogen, oxygen,
Elements in Cells The living substance of cells is made up of cytoplasm and the structures within it. About 96% of cytoplasm and its included structures are composed of the elements carbon, hydrogen, oxygen,
THE HISTORY OF CELL BIOLOGY
 SECTION 4-1 REVIEW THE HISTORY OF CELL BIOLOGY Define the following terms. 1. cell 2. cell theory Write the correct letter in the blank. 1. One early piece of evidence supporting the cell theory was the
SECTION 4-1 REVIEW THE HISTORY OF CELL BIOLOGY Define the following terms. 1. cell 2. cell theory Write the correct letter in the blank. 1. One early piece of evidence supporting the cell theory was the
Proteins and Nucleic Acids
 Proteins and Nucleic Acids Chapter 5 Macromolecules: Proteins Proteins Most structurally & functionally diverse group of biomolecules. : o Involved in almost everything o Enzymes o Structure (keratin,
Proteins and Nucleic Acids Chapter 5 Macromolecules: Proteins Proteins Most structurally & functionally diverse group of biomolecules. : o Involved in almost everything o Enzymes o Structure (keratin,
Biological molecules:
 Biological molecules: All are organic (based on carbon). Monomers vs. polymers: Monomers refer to the subunits that, when polymerized, make up a larger polymer. Monomers may function on their own in some
Biological molecules: All are organic (based on carbon). Monomers vs. polymers: Monomers refer to the subunits that, when polymerized, make up a larger polymer. Monomers may function on their own in some
The Molecules of Life - Overview. The Molecules of Life. The Molecules of Life. The Molecules of Life
 The Molecules of Life - Overview The Molecules of Life The Importance of Carbon Organic Polymers / Monomers Functions of Organic Molecules Origin of Organic Molecules The Molecules of Life Water is the
The Molecules of Life - Overview The Molecules of Life The Importance of Carbon Organic Polymers / Monomers Functions of Organic Molecules Origin of Organic Molecules The Molecules of Life Water is the
Biochemistry of Cells
 Biochemistry of Cells 1 Carbon-based Molecules Although a cell is mostly water, the rest of the cell consists mostly of carbon-based molecules Organic chemistry is the study of carbon compounds Carbon
Biochemistry of Cells 1 Carbon-based Molecules Although a cell is mostly water, the rest of the cell consists mostly of carbon-based molecules Organic chemistry is the study of carbon compounds Carbon
Cells & Cell Organelles
 Cells & Cell Organelles The Building Blocks of Life H Biology Types of cells bacteria cells Prokaryote - no organelles Eukaryotes - organelles animal cells plant cells Cell size comparison Animal cell
Cells & Cell Organelles The Building Blocks of Life H Biology Types of cells bacteria cells Prokaryote - no organelles Eukaryotes - organelles animal cells plant cells Cell size comparison Animal cell
Cell Structure and Function. Eukaryotic Cell: Neuron
 Cell Structure and Function Eukaryotic Cell: Neuron Cell Structure and Function Eukaryotic Cells: Blood Cells Cell Structure and Function Prokaryotic Cells: Bacteria Cell Structure and Function All living
Cell Structure and Function Eukaryotic Cell: Neuron Cell Structure and Function Eukaryotic Cells: Blood Cells Cell Structure and Function Prokaryotic Cells: Bacteria Cell Structure and Function All living
Chapter 5 Organelles. Lesson Objectives List the organelles of the cell and their functions. Distinguish between plant and animal cells.
 Chapter 5 Organelles Lesson Objectives List the organelles of the cell and their functions. Distinguish between plant and animal cells. Check Your Understanding What is a cell? How do we visualize cells?
Chapter 5 Organelles Lesson Objectives List the organelles of the cell and their functions. Distinguish between plant and animal cells. Check Your Understanding What is a cell? How do we visualize cells?
Quick Hit Activity Using UIL Science Contests For Formative and Summative Assessments of Pre-AP and AP Biology Students
 Quick Hit Activity Using UIL Science Contests For Formative and Summative Assessments of Pre-AP and AP Biology Students Activity Title: Quick Hit Goal of Activity: To perform formative and summative assessments
Quick Hit Activity Using UIL Science Contests For Formative and Summative Assessments of Pre-AP and AP Biology Students Activity Title: Quick Hit Goal of Activity: To perform formative and summative assessments
Name: Hour: Elements & Macromolecules in Organisms
 Name: Hour: Elements & Macromolecules in Organisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds
Name: Hour: Elements & Macromolecules in Organisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds
Elements in Biological Molecules
 Chapter 3: Biological Molecules 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids Elements in Biological Molecules Biological macromolecules are made almost entirely of just 6 elements: Carbon (C)
Chapter 3: Biological Molecules 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids Elements in Biological Molecules Biological macromolecules are made almost entirely of just 6 elements: Carbon (C)
Bacterial (Prokaryotic) Cell. Common features of all cells. Tour of the Cell. Eukaryotic Cell. Plasma Membrane defines inside from outside
 www.denniskunkel.com Tour of the Cell www.denniskunkel.com Today s Topics Properties of all cells Prokaryotes and Eukaryotes Functions of Major Cellular Organelles Information, Synthesis&Transport,, Vesicles
www.denniskunkel.com Tour of the Cell www.denniskunkel.com Today s Topics Properties of all cells Prokaryotes and Eukaryotes Functions of Major Cellular Organelles Information, Synthesis&Transport,, Vesicles
Chapter 4: A Tour of the Cell. 1. Cell Basics. Limits to Cell Size. 1. Cell Basics. 2. Prokaryotic Cells. 3. Eukaryotic Cells
 Chapter 4: A Tour of the Cell 1. Cell Basics 2. Prokaryotic Cells 3. Eukaryotic Cells 1. Cell Basics Limits to Cell Size There are 2 main reasons why cells are so small: If cells get too large: 1) there
Chapter 4: A Tour of the Cell 1. Cell Basics 2. Prokaryotic Cells 3. Eukaryotic Cells 1. Cell Basics Limits to Cell Size There are 2 main reasons why cells are so small: If cells get too large: 1) there
BSC 2010 - Exam I Lectures and Text Pages. The Plasma Membrane Structure and Function. Phospholipids. I. Intro to Biology (2-29) II.
 BSC 2010 - Exam I Lectures and Text Pages I. Intro to Biology (2-29) II. Chemistry of Life Chemistry review (30-46) Water (47-57) Carbon (58-67) Macromolecules (68-91) III. Cells and Membranes Cell structure
BSC 2010 - Exam I Lectures and Text Pages I. Intro to Biology (2-29) II. Chemistry of Life Chemistry review (30-46) Water (47-57) Carbon (58-67) Macromolecules (68-91) III. Cells and Membranes Cell structure
ATOMS AND BONDS. Bonds
 ATOMS AND BONDS Atoms of elements are the simplest units of organization in the natural world. Atoms consist of protons (positive charge), neutrons (neutral charge) and electrons (negative charge). The
ATOMS AND BONDS Atoms of elements are the simplest units of organization in the natural world. Atoms consist of protons (positive charge), neutrons (neutral charge) and electrons (negative charge). The
1. The diagram below represents a biological process
 1. The diagram below represents a biological process 5. The chart below indicates the elements contained in four different molecules and the number of atoms of each element in those molecules. Which set
1. The diagram below represents a biological process 5. The chart below indicates the elements contained in four different molecules and the number of atoms of each element in those molecules. Which set
The Structure and Function of Macromolecules: Carbohydrates, Lipids & Phospholipids
 The Structure and Function of Macromolecules: Carbohydrates, Lipids & Phospholipids The FOUR Classes of Large Biomolecules All living things are made up of four classes of large biological molecules: Carbohydrates
The Structure and Function of Macromolecules: Carbohydrates, Lipids & Phospholipids The FOUR Classes of Large Biomolecules All living things are made up of four classes of large biological molecules: Carbohydrates
Cell and Membrane Practice. A. chromosome B. gene C. mitochondrion D. vacuole
 Name: ate: 1. Which structure is outside the nucleus of a cell and contains N?. chromosome. gene. mitochondrion. vacuole 2. potato core was placed in a beaker of water as shown in the figure below. Which
Name: ate: 1. Which structure is outside the nucleus of a cell and contains N?. chromosome. gene. mitochondrion. vacuole 2. potato core was placed in a beaker of water as shown in the figure below. Which
Chapter 3: Biological Molecules. 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids
 Chapter 3: Biological Molecules 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids Elements in Biological Molecules Biological macromolecules are made almost entirely of just 6 elements: Carbon (C)
Chapter 3: Biological Molecules 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids Elements in Biological Molecules Biological macromolecules are made almost entirely of just 6 elements: Carbon (C)
Biology Chapter 7 Practice Test
 Biology Chapter 7 Practice Test Multiple Choice Write the letter that best answers the question or completes the statement on the line provided. 1. The work of Schleiden and Schwann can be summarized by
Biology Chapter 7 Practice Test Multiple Choice Write the letter that best answers the question or completes the statement on the line provided. 1. The work of Schleiden and Schwann can be summarized by
I. Chapter 5 Summary. II. Nucleotides & Nucleic Acids. III. Lipids
 I. Chapter 5 Summary A. Simple Sugars (CH 2 O) n : 1. One C contains a carbonyl (C=O) rest contain - 2. Classification by functional group: aldoses & ketoses 3. Classification by number of C's: trioses,
I. Chapter 5 Summary A. Simple Sugars (CH 2 O) n : 1. One C contains a carbonyl (C=O) rest contain - 2. Classification by functional group: aldoses & ketoses 3. Classification by number of C's: trioses,
Elements & Macromolecules in Organisms
 Name: Date: Per: Table # Elements & Macromolecules in rganisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight.
Name: Date: Per: Table # Elements & Macromolecules in rganisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight.
Comparing Plant And Animal Cells
 Comparing Plant And Animal Cells http://khanacademy.org/video?v=hmwvj9x4gny Plant Cells shape - most plant cells are squarish or rectangular in shape. amyloplast (starch storage organelle)- an organelle
Comparing Plant And Animal Cells http://khanacademy.org/video?v=hmwvj9x4gny Plant Cells shape - most plant cells are squarish or rectangular in shape. amyloplast (starch storage organelle)- an organelle
PRESTWICK ACADEMY NATIONAL 5 BIOLOGY CELL BIOLOGY SUMMARY
 Name PRESTWICK ACADEMY NATIONAL 5 BIOLOGY CELL BIOLOGY SUMMARY Cell Structure Identify animal, plant, fungal and bacterial cell ultrastructure and know the structures functions. Plant cell Animal cell
Name PRESTWICK ACADEMY NATIONAL 5 BIOLOGY CELL BIOLOGY SUMMARY Cell Structure Identify animal, plant, fungal and bacterial cell ultrastructure and know the structures functions. Plant cell Animal cell
Lab 3 Organic Molecules of Biological Importance
 Name Biology 3 ID Number Lab 3 Organic Molecules of Biological Importance Section 1 - Organic Molecules Section 2 - Functional Groups Section 3 - From Building Blocks to Macromolecules Section 4 - Carbohydrates
Name Biology 3 ID Number Lab 3 Organic Molecules of Biological Importance Section 1 - Organic Molecules Section 2 - Functional Groups Section 3 - From Building Blocks to Macromolecules Section 4 - Carbohydrates
BIOMOLECULES. reflect
 reflect A child s building blocks are relatively simple structures. When they come together, however, they can form magnifi cent structures. The elaborate city scene to the right is made of small, simple
reflect A child s building blocks are relatively simple structures. When they come together, however, they can form magnifi cent structures. The elaborate city scene to the right is made of small, simple
AP BIOLOGY 2006 SCORING GUIDELINES. Question 1
 AP BIOLOGY 2006 SCORING GUIDELINES Question 1 A major distinction between prokaryotes and eukaryotes is the presence of membrane-bound organelles in eukaryotes. (a) Describe the structure and function
AP BIOLOGY 2006 SCORING GUIDELINES Question 1 A major distinction between prokaryotes and eukaryotes is the presence of membrane-bound organelles in eukaryotes. (a) Describe the structure and function
BIOLOGICAL MOLECULES OF LIFE
 BIOLOGICAL MOLECULES OF LIFE C A R B O H Y D R A T E S, L I P I D S, P R O T E I N S, A N D N U C L E I C A C I D S The Academic Support Center @ Daytona State College (Science 115, Page 1 of 29) Carbon
BIOLOGICAL MOLECULES OF LIFE C A R B O H Y D R A T E S, L I P I D S, P R O T E I N S, A N D N U C L E I C A C I D S The Academic Support Center @ Daytona State College (Science 115, Page 1 of 29) Carbon
ISTEP+: Biology I End-of-Course Assessment Released Items and Scoring Notes
 ISTEP+: Biology I End-of-Course Assessment Released Items and Scoring Notes Page 1 of 22 Introduction Indiana students enrolled in Biology I participated in the ISTEP+: Biology I Graduation Examination
ISTEP+: Biology I End-of-Course Assessment Released Items and Scoring Notes Page 1 of 22 Introduction Indiana students enrolled in Biology I participated in the ISTEP+: Biology I Graduation Examination
Microscopes. Eukaryotes Eukaryotic cells are characterized by having: DNA in a nucleus that is bounded by a membranous nuclear envelope
 CH 6 The Cell Microscopy Scientists use microscopes to visualize cells too small to see with the naked eye. In a light microscope (LM), visible light is passed through a specimen and then through glass
CH 6 The Cell Microscopy Scientists use microscopes to visualize cells too small to see with the naked eye. In a light microscope (LM), visible light is passed through a specimen and then through glass
Chapter 2 Chemical Principles
 Chapter 2 Chemical Principles I. Chemistry. [Students should read this section on their own]. a. Chemistry is the study of the interactions between atoms and molecules. b. The atom is the smallest unit
Chapter 2 Chemical Principles I. Chemistry. [Students should read this section on their own]. a. Chemistry is the study of the interactions between atoms and molecules. b. The atom is the smallest unit
The Steps. 1. Transcription. 2. Transferal. 3. Translation
 Protein Synthesis Protein synthesis is simply the "making of proteins." Although the term itself is easy to understand, the multiple steps that a cell in a plant or animal must go through are not. In order
Protein Synthesis Protein synthesis is simply the "making of proteins." Although the term itself is easy to understand, the multiple steps that a cell in a plant or animal must go through are not. In order
3120-1 - Page 1. Name:
 Name: 1) Which series is arranged in correct order according to decreasing size of structures? A) DNA, nucleus, chromosome, nucleotide, nitrogenous base B) chromosome, nucleus, nitrogenous base, nucleotide,
Name: 1) Which series is arranged in correct order according to decreasing size of structures? A) DNA, nucleus, chromosome, nucleotide, nitrogenous base B) chromosome, nucleus, nitrogenous base, nucleotide,
Six major functions of membrane proteins: Transport Enzymatic activity
 CH 7 Membranes Cellular Membranes Phospholipids are the most abundant lipid in the plasma membrane. Phospholipids are amphipathic molecules, containing hydrophobic and hydrophilic regions. The fluid mosaic
CH 7 Membranes Cellular Membranes Phospholipids are the most abundant lipid in the plasma membrane. Phospholipids are amphipathic molecules, containing hydrophobic and hydrophilic regions. The fluid mosaic
Plant and Animal Cells
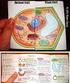 Plant and Animal Cells a. Explain that cells take in nutrients in order to grow, divide and to make needed materials. S7L2a b. Relate cell structures (cell membrane, nucleus, cytoplasm, chloroplasts, and
Plant and Animal Cells a. Explain that cells take in nutrients in order to grow, divide and to make needed materials. S7L2a b. Relate cell structures (cell membrane, nucleus, cytoplasm, chloroplasts, and
The Cell Teaching Notes and Answer Keys
 The Cell Teaching Notes and Answer Keys Subject area: Science / Biology Topic focus: The Cell: components, types of cells, organelles, levels of organization Learning Aims: describe similarities and differences
The Cell Teaching Notes and Answer Keys Subject area: Science / Biology Topic focus: The Cell: components, types of cells, organelles, levels of organization Learning Aims: describe similarities and differences
Cytology. Living organisms are made up of cells. Either PROKARYOTIC or EUKARYOTIC cells.
 CYTOLOGY Cytology Living organisms are made up of cells. Either PROKARYOTIC or EUKARYOTIC cells. A. two major cell types B. distinguished by structural organization See table on handout for differences.
CYTOLOGY Cytology Living organisms are made up of cells. Either PROKARYOTIC or EUKARYOTIC cells. A. two major cell types B. distinguished by structural organization See table on handout for differences.
Given these characteristics of life, which of the following objects is considered a living organism? W. X. Y. Z.
 Cell Structure and Organization 1. All living things must possess certain characteristics. They are all composed of one or more cells. They can grow, reproduce, and pass their genes on to their offspring.
Cell Structure and Organization 1. All living things must possess certain characteristics. They are all composed of one or more cells. They can grow, reproduce, and pass their genes on to their offspring.
MCAS Biology. Review Packet
 MCAS Biology Review Packet 1 Name Class Date 1. Define organic. THE CHEMISTRY OF LIFE 2. All living things are made up of 6 essential elements: SPONCH. Name the six elements of life. S N P C O H 3. Elements
MCAS Biology Review Packet 1 Name Class Date 1. Define organic. THE CHEMISTRY OF LIFE 2. All living things are made up of 6 essential elements: SPONCH. Name the six elements of life. S N P C O H 3. Elements
Cellular Structure and Function
 Chapter Test A CHAPTER 7 Cellular Structure and Function Part A: Multiple Choice In the space at the left, write the letter of the term or phrase that best answers each question. 1. Which defines a cell?
Chapter Test A CHAPTER 7 Cellular Structure and Function Part A: Multiple Choice In the space at the left, write the letter of the term or phrase that best answers each question. 1. Which defines a cell?
Introduction to the Cell: Plant and Animal Cells
 Introduction to the Cell: Plant and Animal Cells Tissues, Organs, and Systems of Living Things Cells, Cell Division, and Animal Systems and Plant Systems Cell Specialization Human Systems All organisms
Introduction to the Cell: Plant and Animal Cells Tissues, Organs, and Systems of Living Things Cells, Cell Division, and Animal Systems and Plant Systems Cell Specialization Human Systems All organisms
7.2 Cells: A Look Inside
 CHAPTER 7 CELL STRUCTURE AND FUNCTION 7.2 Cells: A Look Inside Imagine a factory that makes thousands of cookies a day. Ingredients come into the factory, get mixed and baked, then the cookies are packaged.
CHAPTER 7 CELL STRUCTURE AND FUNCTION 7.2 Cells: A Look Inside Imagine a factory that makes thousands of cookies a day. Ingredients come into the factory, get mixed and baked, then the cookies are packaged.
Biological cell membranes
 Unit 14: Cell biology. 14 2 Biological cell membranes The cell surface membrane surrounds the cell and acts as a barrier between the cell s contents and the environment. The cell membrane has multiple
Unit 14: Cell biology. 14 2 Biological cell membranes The cell surface membrane surrounds the cell and acts as a barrier between the cell s contents and the environment. The cell membrane has multiple
Examination One. Biology 101. Dr. Jaeson T. Fournier
 Examination One Biology 101 Dr. Jaeson T. Fournier Examination Instructions: Answers are to be indicated on a scantron. Keep your work protected! This helps prevent dishonesty. The instructor will not
Examination One Biology 101 Dr. Jaeson T. Fournier Examination Instructions: Answers are to be indicated on a scantron. Keep your work protected! This helps prevent dishonesty. The instructor will not
Genetic information (DNA) determines structure of proteins DNA RNA proteins cell structure 3.11 3.15 enzymes control cell chemistry ( metabolism )
 Biology 1406 Exam 3 Notes Structure of DNA Ch. 10 Genetic information (DNA) determines structure of proteins DNA RNA proteins cell structure 3.11 3.15 enzymes control cell chemistry ( metabolism ) Proteins
Biology 1406 Exam 3 Notes Structure of DNA Ch. 10 Genetic information (DNA) determines structure of proteins DNA RNA proteins cell structure 3.11 3.15 enzymes control cell chemistry ( metabolism ) Proteins
Biology I. Chapter 7
 Biology I Chapter 7 Interest Grabber NOTEBOOK #1 Are All Cells Alike? All living things are made up of cells. Some organisms are composed of only one cell. Other organisms are made up of many cells. 1.
Biology I Chapter 7 Interest Grabber NOTEBOOK #1 Are All Cells Alike? All living things are made up of cells. Some organisms are composed of only one cell. Other organisms are made up of many cells. 1.
BIOLOGICAL MEMBRANES: FUNCTIONS, STRUCTURES & TRANSPORT
 BIOLOGICAL MEMBRANES: FUNCTIONS, STRUCTURES & TRANSPORT UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY BMLS II / B Pharm II / BDS II VJ Temple
BIOLOGICAL MEMBRANES: FUNCTIONS, STRUCTURES & TRANSPORT UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY BMLS II / B Pharm II / BDS II VJ Temple
Ch. 8 - The Cell Membrane
 Ch. 8 - The Cell Membrane 2007-2008 Phospholipids Phosphate head hydrophilic Fatty acid tails hydrophobic Arranged as a bilayer Phosphate attracted to water Fatty acid repelled by water Aaaah, one of those
Ch. 8 - The Cell Membrane 2007-2008 Phospholipids Phosphate head hydrophilic Fatty acid tails hydrophobic Arranged as a bilayer Phosphate attracted to water Fatty acid repelled by water Aaaah, one of those
Membrane Structure and Function
 Membrane Structure and Function -plasma membrane acts as a barrier between cells and the surrounding. -plasma membrane is selective permeable -consist of lipids, proteins and carbohydrates -major lipids
Membrane Structure and Function -plasma membrane acts as a barrier between cells and the surrounding. -plasma membrane is selective permeable -consist of lipids, proteins and carbohydrates -major lipids
Lecture 8. Protein Trafficking/Targeting. Protein targeting is necessary for proteins that are destined to work outside the cytoplasm.
 Protein Trafficking/Targeting (8.1) Lecture 8 Protein Trafficking/Targeting Protein targeting is necessary for proteins that are destined to work outside the cytoplasm. Protein targeting is more complex
Protein Trafficking/Targeting (8.1) Lecture 8 Protein Trafficking/Targeting Protein targeting is necessary for proteins that are destined to work outside the cytoplasm. Protein targeting is more complex
Carbon Hydrogen Oxygen Nitrogen
 Concept 1 - Thinking Practice 1. If the following molecules were to undergo a dehydration synthesis reaction, what molecules would result? Circle the parts of each amino acid that will interact and draw
Concept 1 - Thinking Practice 1. If the following molecules were to undergo a dehydration synthesis reaction, what molecules would result? Circle the parts of each amino acid that will interact and draw
Introduction, Noncovalent Bonds, and Properties of Water
 Lecture 1 Introduction, Noncovalent Bonds, and Properties of Water Reading: Berg, Tymoczko & Stryer: Chapter 1 problems in textbook: chapter 1, pp. 23-24, #1,2,3,6,7,8,9, 10,11; practice problems at end
Lecture 1 Introduction, Noncovalent Bonds, and Properties of Water Reading: Berg, Tymoczko & Stryer: Chapter 1 problems in textbook: chapter 1, pp. 23-24, #1,2,3,6,7,8,9, 10,11; practice problems at end
Chapter 3. Cellular Structure and Function Worksheets. 39 www.ck12.org
 Chapter 3 Cellular Structure and Function Worksheets (Opening image copyright by Sebastian Kaulitzki, 2010. Used under license from Shutterstock.com.) Lesson 3.1: Introduction to Cells Lesson 3.2: Cell
Chapter 3 Cellular Structure and Function Worksheets (Opening image copyright by Sebastian Kaulitzki, 2010. Used under license from Shutterstock.com.) Lesson 3.1: Introduction to Cells Lesson 3.2: Cell
CELLS: PLANT CELLS 20 FEBRUARY 2013
 CELLS: PLANT CELLS 20 FEBRUARY 2013 Lesson Description In this lesson we will discuss the following: The Cell Theory Terminology Parts of Plant Cells: Organelles Difference between plant and animal cells
CELLS: PLANT CELLS 20 FEBRUARY 2013 Lesson Description In this lesson we will discuss the following: The Cell Theory Terminology Parts of Plant Cells: Organelles Difference between plant and animal cells
Molecular Cell Biology
 Harvey Lodish Arnold Berk Paul Matsudaira Chris A. Kaiser Monty Krieger Matthew P. Scott Lawrence Zipursky James Darnell Molecular Cell Biology Fifth Edition Chapter 2: Chemical Foundations Copyright 2004
Harvey Lodish Arnold Berk Paul Matsudaira Chris A. Kaiser Monty Krieger Matthew P. Scott Lawrence Zipursky James Darnell Molecular Cell Biology Fifth Edition Chapter 2: Chemical Foundations Copyright 2004
Proteins. Proteins. Amino Acids. Most diverse and most important molecule in. Functions: Functions (cont d)
 Proteins Proteins Most diverse and most important molecule in living i organisms Functions: 1. Structural (keratin in hair, collagen in ligaments) 2. Storage (casein in mother s milk) 3. Transport (HAEMOGLOBIN!)
Proteins Proteins Most diverse and most important molecule in living i organisms Functions: 1. Structural (keratin in hair, collagen in ligaments) 2. Storage (casein in mother s milk) 3. Transport (HAEMOGLOBIN!)
Cells. Structure, Function and Homeostasis
 Cells Structure, Function and Homeostasis Characteristics of Cells Basic unit of life anything alive is made of cells Plasma membrane (skin) that separates them from the environment. Skeletonsfor protection
Cells Structure, Function and Homeostasis Characteristics of Cells Basic unit of life anything alive is made of cells Plasma membrane (skin) that separates them from the environment. Skeletonsfor protection
Digestive System Lecture 5 Winter 2014
 Digestive System Lecture 5 Winter 2014 This lecture tells the story of the Flow of Matter from Food to Cells. The pictures are only there to help you visualize structures don t worry about names of structures
Digestive System Lecture 5 Winter 2014 This lecture tells the story of the Flow of Matter from Food to Cells. The pictures are only there to help you visualize structures don t worry about names of structures
cells - relatively simple cells - lack nuclear membrane and many organelles - bacteria and their relatives are all prokaryotic
 Cell Biology A cell is chemical system that is able to maintain its structure and reproduce. Cells are the fundamental unit of life. All living things are cells or composed of cells. 1 The interior contents
Cell Biology A cell is chemical system that is able to maintain its structure and reproduce. Cells are the fundamental unit of life. All living things are cells or composed of cells. 1 The interior contents
Chapter 2. The Chemistry of Life Worksheets
 Chapter 2 The Chemistry of Life Worksheets (Opening image courtesy of David Iberri, http://en.wikipedia.org/wiki/file:camkii.png, and under the Creative Commons license CC-BY-SA 3.0.) Lesson 2.1: Matter
Chapter 2 The Chemistry of Life Worksheets (Opening image courtesy of David Iberri, http://en.wikipedia.org/wiki/file:camkii.png, and under the Creative Commons license CC-BY-SA 3.0.) Lesson 2.1: Matter
AP Biology-Chapter #6 & 7 Review
 DO NOT WRITE ON THIS TEST- USE ANSWER DOCUMENT AP Biology-Chapter #6 & 7 Review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. All of the following are
DO NOT WRITE ON THIS TEST- USE ANSWER DOCUMENT AP Biology-Chapter #6 & 7 Review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. All of the following are
pathway that involves taking in heat from the environment at each step. C.
 Study Island Cell Energy Keystone Review 1. Cells obtain energy by either capturing light energy through photosynthesis or by breaking down carbohydrates through cellular respiration. In both photosynthesis
Study Island Cell Energy Keystone Review 1. Cells obtain energy by either capturing light energy through photosynthesis or by breaking down carbohydrates through cellular respiration. In both photosynthesis
Date: Student Name: Teacher Name: Jared George. Score: 1) A cell with 1% solute concentration is placed in a beaker with a 5% solute concentration.
 Biology Keystone (PA Core) Quiz Homeostasis and Transport - (BIO.A.4.1.1 ) Plasma Membrane, (BIO.A.4.1.2 ) Transport Mechanisms, (BIO.A.4.1.3 ) Transport Facilitation Student Name: Teacher Name: Jared
Biology Keystone (PA Core) Quiz Homeostasis and Transport - (BIO.A.4.1.1 ) Plasma Membrane, (BIO.A.4.1.2 ) Transport Mechanisms, (BIO.A.4.1.3 ) Transport Facilitation Student Name: Teacher Name: Jared
Biology 101 Chapter 4 Cells as the Basic Unit of Life. The Cell Theory Major Contributors: Galileo = first observations made with a microscope
 Biology 101 Chapter 4 Cells as the Basic Unit of Life The Cell Theory Major Contributors: Galileo = first observations made with a microscope Robert Hooke = first to observe small compartments in dead
Biology 101 Chapter 4 Cells as the Basic Unit of Life The Cell Theory Major Contributors: Galileo = first observations made with a microscope Robert Hooke = first to observe small compartments in dead
The Cell: Organelle Diagrams
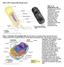 The Cell: Organelle Diagrams Fig 7-4. A prokaryotic cell. Lacking a true nucleus and the other membrane-enclosed organelles of the eukaryotic cell, the prokaryotic cell is much simpler in structure. Only
The Cell: Organelle Diagrams Fig 7-4. A prokaryotic cell. Lacking a true nucleus and the other membrane-enclosed organelles of the eukaryotic cell, the prokaryotic cell is much simpler in structure. Only
RAD 223. Radiography physiology. Lecture Notes. First lecture: Cell and Tissue
 RAD 223 Radiography physiology Lecture Notes First lecture: Cell and Tissue Physiology: the word physiology derived from a Greek word for study of nature. It is the study of how the body and its part work
RAD 223 Radiography physiology Lecture Notes First lecture: Cell and Tissue Physiology: the word physiology derived from a Greek word for study of nature. It is the study of how the body and its part work
Organelles and Their Functions
 Organelles and Their Functions The study of cell organelles and their functions is a fascinating part of biology. The current article provides a brief description of the structure of organelles and their
Organelles and Their Functions The study of cell organelles and their functions is a fascinating part of biology. The current article provides a brief description of the structure of organelles and their
Carbohydrates Lipids Proteins Nucleic Acids
 Carbohydrates Lipids Proteins Nucleic Acids Carbon The element of life! All living things contain the element carbon. Organic means it contains carbon The reason for this is because of carbon s ability
Carbohydrates Lipids Proteins Nucleic Acids Carbon The element of life! All living things contain the element carbon. Organic means it contains carbon The reason for this is because of carbon s ability
Organic Compounds. Essential Questions: What is Organic? What are the 4 major Organic Compounds? How are they made? What are they used for?
 Organic Compounds Essential Questions: What is Organic? What are the 4 major Organic Compounds? How are they made? What are they used for? Aristotle: Francesco Redi: What do we already know? Spontaneous
Organic Compounds Essential Questions: What is Organic? What are the 4 major Organic Compounds? How are they made? What are they used for? Aristotle: Francesco Redi: What do we already know? Spontaneous
Lecture Series 7. From DNA to Protein. Genotype to Phenotype. Reading Assignments. A. Genes and the Synthesis of Polypeptides
 Lecture Series 7 From DNA to Protein: Genotype to Phenotype Reading Assignments Read Chapter 7 From DNA to Protein A. Genes and the Synthesis of Polypeptides Genes are made up of DNA and are expressed
Lecture Series 7 From DNA to Protein: Genotype to Phenotype Reading Assignments Read Chapter 7 From DNA to Protein A. Genes and the Synthesis of Polypeptides Genes are made up of DNA and are expressed
WATER CHAPTER 3 - BIOCHEMISTRY "THE CHEMISTRY OF LIFE" POLARITY HYDROGEN BONDING
 CHAPTER 3 - BIOCHEMISTRY "THE CHEMISTRY OF LIFE" WATER Compare the body of the jellyfish with our own bodies. The jellyfish will die if it is removed from its water environment, yet we can live in the
CHAPTER 3 - BIOCHEMISTRY "THE CHEMISTRY OF LIFE" WATER Compare the body of the jellyfish with our own bodies. The jellyfish will die if it is removed from its water environment, yet we can live in the
Biological Molecules
 Biological Molecules I won t lie. This is probably the most boring topic you have ever done in any science. It s pretty much as simple as this: learn the material deal with it. Enjoy don t say I didn t
Biological Molecules I won t lie. This is probably the most boring topic you have ever done in any science. It s pretty much as simple as this: learn the material deal with it. Enjoy don t say I didn t
The Lipid Bilayer Is a Two-Dimensional Fluid
 The Lipid Bilayer Is a Two-Dimensional Fluid The aqueous environment inside and outside a cell prevents membrane lipids from escaping from bilayer, but nothing stops these molecules from moving about and
The Lipid Bilayer Is a Two-Dimensional Fluid The aqueous environment inside and outside a cell prevents membrane lipids from escaping from bilayer, but nothing stops these molecules from moving about and
