Fructan: More Than a Reserve Carbohydrate? 1
|
|
|
- Adelia Gaines
- 9 years ago
- Views:
Transcription
1 Plant Physiology, June 1999, Vol. 120, pp , American Society of Plant Physiologists Update on Biochemistry Fructan: More Than a Reserve Carbohydrate? 1 Irma Vijn 2 and Sjef Smeekens* Department of Botanical Ecology and Evolutionary Biology, University of Utrecht, Padualaan 8, 3584 CH Utrecht, The Netherlands Most plants store starch or Suc as reserve carbohydrates, but about 15% of all flowering plant species store fructans, which are linear and branched polymers of Fru. Among the plants that store fructans are many of significant economic importance, such as cereals (e.g. barley, wheat, and oat), vegetables (e.g. chicory, onion, and lettuce), ornamentals (e.g. dahlia and tulip), and forage grasses (e.g. Lolium and Festuca) (Hendry and Wallace, 1993). Fructans isolated from these plants have a variety of applications. Small fructans have a sweet taste, whereas longer fructan chains form emulsions with a fat-like texture and a neutral taste. The human digestive tract does not contain enzymes able to degrade fructans; therefore, there is strong interest from the food industry to use them as low-calorie food ingredients. In plants, fructans may have functions other than carbon storage; they have been implicated in protecting plants against water deficit caused by drought or low temperatures (Hendry and Wallace, 1993; Pilon-Smits et al., 1995). The substrate for fructan synthesis is Suc, and like Suc, fructans are stored in the vacuole. Although Suc is synthesized in the cytoplasm, fructans are produced in the vacuole by the action of specific enzymes (fructosyltransferases) that transfer Fru from Suc to the growing fructan chain. Fructan synthesis is modulated by light, which changes the availability of Suc in the cell (Fig. 1). The biosynthetic enzymes are evolutionarily related to invertases, enzymes that hydrolyze Suc. The biochemistry of fructan synthesis has been determined, and the first genes encoding these biosynthetic enzymes have recently been cloned, opening new biotechnological opportunities for the use of fructans. Until now the major obstacles have been the limited availability of long-chain fructans and the heterogeneity of harvested fructans. It will now be possible to genetically engineer plants to produce large quantities of fructans of defined structure and size. Furthermore, fructan accumulation in 1 This research was funded in part by the Netherlands Foundation for Chemical Research and the Netherlands Technology Foundation. 2 Present address: Department of Molecular Genetics, Institute for Molecular Biological Sciences, BioCentrum Amsterdam, Vrije Universiteit, De Boelelaan 1087, 1081 HV Amsterdam, The Netherlands. * Corresponding author; j.c.m.smeekens@bio.uu.nl; fax plants that normally do not produce them may contribute to protection from water stress in these plants. A number of research groups have studied fructan accumulation in plants in an attempt to understand fructan synthesis and the physiological role of fructan accumulation in plants and to improve the commercial availability of fructans. In this Update we give an overview of these attempts and discuss their impact on our insight into fructan production in plants. First, a few words on fructan synthesis in bacteria, which is simpler than plant fructan biosynthesis because only a single biosynthetic enzyme is involved. BACTERIAL FRUCTAN BIOSYNTHESIS Fructan-producing bacteria can be found in a wide range of taxa, including plant pathogens and the bacteria present in oral and gut floras of animals and humans. Examples of bacterial genera in which fructan-producing strains can be found are Bacillus, Streptococcus, Pseudomonas, Erwinia, and Actinomyces (Hendry and Wallace, 1993). In general, bacteria produce fructan molecules consisting mainly of (2-6)- linked fructosyl residues, occasionally containing (2-1)- linked branches (Dedonder, 1966). Such fructans are called levans and can reach a DP of more than 100,000 Fru units. Bacterial levan is produced extracellularly by a single enzyme, levansucrase, which produces levan directly from Suc. In addition to fructosyltransferase activity, several of these bacterial levansucrases can transfer fructosyl units to water (invertase activity) and to other sugars such as Glu, Fru, and raffinose (Cote and Ahlgren, 1993) as a side reaction. Although most of the bacteria produce levans, a few strains of Streptococcus mutans known for their involvement in dental caries produce fructan consisting mostly of (2-1)-linked fructosyl units (for review, see Uchiyama, 1993). For the degradation of levan, bacteria produce specific enzymes called levanases, which are divided into endoand exo-levanases. Exo-levanases hydrolyze only levan and the product is usually levanbiose, meaning that a terminal di-fructosyl unit is removed. Endo-levanases hydrolyze levan and levan oligomers consisting of more than three fructosyl units. These levan molecules are randomly Abbreviations: 1-FFT, fructan:fructan 1-fructosyltransferase; 1-SST, Suc:Suc 1-fructosyltransferase; 6G-FFT, fructan:fructan 6Gfructosyltransferase; 6-SFT, Suc:fructan 6-fructosyltransferase; DP, degree of polymerization.
2 352 Vijn and Smeekens Plant Physiol. Vol. 120, 1999 Figure 1. Schematic representation of carbohydrate metabolism in a plant cell. High photosynthetic activity is associated with high rates of carbon export from the chloroplast to the cytoplasm, resulting in an increase of intermediates for Suc synthesis. The synthesized Suc is either distributed to the vacuole (storage) or to the apoplast (export). In the vacuole, Suc can be converted into fructan by fructosyltransferases (1) or hydrolyzed into Glu and Fru by invertase (2). split into short-chain levans. Nonspecific -fructosidases are produced to split off the terminal Fru residue (Uchiyama, 1993). STRUCTURAL DIVERSITY OF FRUCTANS In contrast to the seemingly uniform structure of bacterial fructans, plant fructans show much more structural diversity (Pollock and Cairns, 1991). The fructosyl chain length in plants varies greatly and is much shorter than that of bacterial fructan. In general, DPs of 30 to 50 fructosyl residues are found, but occasionally DPs can exceed 200. Furthermore, plant fructans have a greater variety in the linkage of the fructosyl residues. In higher plants five major classes of structurally different fructans can be distinguished: inulin, levan, mixed levan, inulin neoseries, and levan neoseries. Inulin consists of linear (2-1)-linked -d-fructosyl units (G1-2F1-2Fn) and is usually found in plant species belonging to the order Asterales, such as chicory and Jerusalem artichoke (Bonnett et al., 1994; Koops and Jonker, 1996). The shortest inulin molecule is the trisaccharide 1-kestose, also called isokestose (Fig. 2A). Levan consists of linear (2-6)-linked -d-fructosyl units (G1-2F6-2Fn) and is found in some grasses (e.g. Dactylis glomerata; Bonnett et al., 1997). Mixed levan is composed of both (2-1)- and (2-6)-linked -d-fructosyl units. This type of fructan is found in most plant species belonging to the Poales, such as wheat and barley (Carpita et al., 1989; Bonnett et al., 1997). An example of this type of fructan is the molecule bifurcose (Fig. 2B). The inulin neoseries are linear (2-1)-linked -d-fructosyl units linked to both C1 and C6 of the Glu moiety of the Suc molecule. This results in a fructan polymer with a Fru chain (mf2-1f2-6g1-2f1-2fn) on both ends of the Glu molecule. These fructans are found in plants belonging to the Liliaceae (e.g. onion and asparagus; Shiomi, 1989). The smallest inulin neoseries molecule is neokestose (Fig. 2C). The levan neoseries are polymers of predominantly (2-6)-linked fructosyl residues on either end of the Glu moiety of the Suc molecule. These fructans are found in a few plant species belonging to the Poales (e.g. oat; Livingstone et al., 1993). Although most fructan molecules consist of fructosyl residues linked to Suc, fructan molecules have also been isolated from species of the Asteraceae that contain only (2-1)-linked Fru molecules (Ernst et al., 1996). PLANT FRUCTOSYLTRANSFERASES In plants, fructan is synthesized from Suc by the action of two or more different fructosyltransferases. According to the classical model of Edelman and Jefford (1968), two enzymes are involved in the synthesis of the most simple form of fructan, inulin. The first enzyme, 1-SST, initiates de novo fructan synthesis by catalyzing the transfer of a fructosyl residue from Suc to another Suc molecule, resulting in the formation of the trisaccharide, 1-kestose (G1-2F1-2F;
3 Fructan: More Than a Reserve Carbohydrate? 353 Figure 2. Some examples of structurally different fructan molecules found in plants. A, The trisaccharide 1-kestose consists of a (2-1)- linked -D-fructosyl unit to Suc and is the shortest inulin molecule. B, The tetrasaccharide bifurcose is an example of a mixed-type levan and consists of a (2-1)- and a (2-6)-linked -D-fructosyl unit to Suc. C, Neokestose is the smallest inulin neoseries molecule, and in this molecule a -D-fructosyl unit is linked to the C6 of the Glu moiety of Suc. The numbers encircled in black represent the numbers of the carbon atoms in the sugar molecules. For a detailed description, see text. In addition to the enzymes, the cdnas encoding 1-SST and 1-FFT have also been recently isolated from several plant species. 1-SST has been cloned from Jerusalem artichoke (Van der Meer et al., 1998), chicory (de Halleux and Van Cutsem, 1997), artichoke (Hellwege et al., 1997), and onion (Vijn et al., 1998), and 1-FFT has been cloned from Jerusalem artichoke (Van der Meer et al., 1998) and artichoke (Hellwege et al., 1998). Transformation of 1-SST cdnas to crops such as sugarbeet and potato has shown that this enzyme is capable of inducing synthesis of 1-kestose and nystose (G1-2F1-2F1-2F) (Hellwege et al., 1997; Sévenier et al., 1998). Remarkably, transgenic sugarbeet plants were obtained that converted 90% of the taproot vacuolar Suc into 1-kestose and nystose. A long-standing question has been how fructan chain length is determined. For example, in Jerusalem artichoke inulin has an average DP of 8 to 10, whereas in blossom discs of artichoke, inulin has an average DP of 65 (Praznik and Beck, 1985). Transformation of tobacco protoplasts with cdnas encoding the 1-FFTs from these plants and in vitro incubation of protein extracts from the transformed protoplasts with low-dp (3 5) inulin as the substrate showed that the 1-FFT of artichoke produces fructan molecules with a higher DP (up to 20) than the 1-FFT of Jerusalem artichoke (up to 12) under the same conditions. This suggests that the size of the fructosyl polymers produced by a plant depends mainly on the enzymatic activity of their 1-FFTs, although a role for exohydrolases in defining the final size of the fructosyl chain in the original host plant cannot be excluded (Hellwege et al., 1998). Therefore, cloning of the cdnas encoding 1-FFT from artichoke and Jerusalem artichoke and their expression in a heterologous system revealed more about the intrinsic properties of these enzymes. In barley the fructan molecule bifurcose is produced. This molecule is composed of both (2-1)- and (2-6)-linked -d-fructosyl units linked to Suc (Fig. 2B). Duchateau et al. Fig. 3). The second enzyme, 1-FFT, transfers fructosyl residues from a fructan molecule with a DP of 3 to another fructan molecule or to Suc (Fig. 3). The action of 1-SST and 1-FFT results in the formation of a mixture of fructan molecules with different chain lengths. Although this model for fructan synthesis was proposed by Edelman and Jefford in 1968, it took nearly 30 years before it was shown to be correct. In 1996, several research groups published results of the purification to homogeneity of the enzymes 1-SST and 1-FFT and showed that incubation of these purified fructosyltransferases with Suc resulted in the formation of inulin with a polymer length of up to 20 fructosyl residues (Koops and Jonker, 1996; Lüscher et al., 1996; Van den Ende and Van Laere, 1996). Both 1-SST and 1-FFT are unusual enzymes in that they do not show simple Michaelis-Menten kinetics; their activity depends on both the substrate and the enzyme concentration and is essentially nonsaturable (Koops and Jonker, 1996). Figure 3. Enzymatic activities of different plant fructosyltransferases involved in fructan synthesis and of fructan exohydrolase, an enzyme involved in fructan degradation. FEH, Fructan exohydrolase.
4 354 Vijn and Smeekens Plant Physiol. Vol. 120, 1999 (1995) succeeded in purifying to homogeneity 6-SFT, the enzyme that catalyzes the production of the tetrasaccharide bifurcose (G1-2F1[6-2F]-2F) from Suc and 1-kestose. If only Suc is available as a substrate for 6-SFT, it acts mainly as an invertase, resulting in the hydrolysis of Suc to Glc and Fru. Moreover, 6-SFT can also catalyze the formation of the trisaccharide 6-kestose (G1-2F6-2; Fig. 3). The barley cdna encoding 6-SFT has been cloned and its catalytic activities have been verified by transient expression assays in tobacco protoplasts (Sprenger et al., 1995). Liliaceous species such as onion and asparagus produce fructan of the inulin neoseries in addition to the normal inulin. These species harbor an additional fructosyltransferase, 6G-FFT, which catalyzes the formation of the trisaccharide neokestose (F2-6G1-2F; Fig. 2C) by the transfer of a Fru residue from 1-kestose to the C6 of the Glu moiety of Suc (Fig. 3; Shiomi, 1989; Wiemken et al., 1995). Extension of the Fru chains on either end of the Glu molecule is catalyzed by the action of 1-FFT. A cdna encoding such a 6G-FFT has been cloned from onion, and its functionality has been proven (Vijn et al., 1997). Next to fructan-synthesizing enzymes, fructan-degrading exohydrolytic enzymes have been purified from several fructan-accumulating plants. An exohydrolase with a -(2-6)-linkage-specific fructan- -fructosidase activity has been purified from the grass Lolium perenne, and a -(2-1)-linkagespecific exohydrolase has been purified from Jerusalem artichoke (Marx et al., 1997a, 1997b). These enzymes degrade the fructan polymers by removing the terminal Fru residue, resulting in the release of free Fru (Fig. 3; Henson and Livingston, 1996; Marx et al., 1997a, 1997b). To our knowledge, no endo-inulinases have yet been purified from plants, but they are present in fungi. Also, no reports have yet appeared regarding the cloning of cdnas encoding plant exohydrolases, but this is likely to happen in the near future. MODEL FOR PLANT FRUCTAN BIOSYNTHESIS Based on the activities observed for the above-mentioned fructosyltransferases, a model can be proposed for biosynthesis of the structurally different fructan molecules found in plants (Fig. 4). Starting from Suc, 1-SST produces 1-kestose, which can be elongated by 1-FFT, resulting in the formation of inulin. From Suc and 1-kestose, 6G-FFT produces neokestose, which can be elongated by 1-FFT or 6-SFT, resulting in the production of the inulin or levan neoseries, respectively. From Suc and 1-kestose, 6-SFT produces bifurcose, which can also be elongated by either 1-FFT or 6-SFT, resulting in branched, mixed-type levans. When only Suc is available as a substrate, 6-SFT produces 6-kestose, which can also be elongated by 6-SFT to produce levans. Another possibility for the production of levans, which was proposed by Wiemken et al. (1995), involves the removal of the (2-1)-linked fructosyl residue from bifurcose by either 1-FFT or exohydrolase (Fig. 4). Looking at the proposed model (Fig. 4), we must keep in mind that fructan synthesis in plants might be much more complex. All of the different fructosyltransferases tested so far are able to produce several fructan molecules depending on the available substrate or on incubation conditions. For example, in addition to 1-kestose, 1-SST is able to produce tetra- and even pentasaccharides from Suc (Koops and Jonker, 1996; Lüscher et al., 1996; Van den Ende and Van Laere, 1996), and in addition to neokestose, 6G-FFT produces the inulin tetrasaccharide nystose (Vijn et al., 1997). This suggests that as-yet-unidentified physiological conditions of the plant influence the substrate affinities of the different fructosyltransferases. Although the general model for fructan synthesis suggests that Suc is the only substrate for de novo fructan synthesis, fructan molecules consisting only of Fru are found in plants (Ernst et al., 1996; Van den Ende et al., Figure 4. Model of fructan biosynthesis in plants. Starting from Suc, structurally different fructan molecules can be produced by the concerted action of different fructosyltransferases. For a detailed description, see text. The dotted arrow shows an alternative route for the production of levan, as suggested by Wiemken et al. (1995). FEH, Fructan exohydrolase.
5 Fructan: More Than a Reserve Carbohydrate? ). These reducing fructofuranosyl-only fructan molecules lack the terminal Glu residue. Although such fructan molecules can be produced by the action of endo-inulinases or, possibly, -glucosidases that remove the Glu moiety, it has recently been shown that 1-FFT is able to produce fructofuranosyl-only oligosaccharides from Fru and inulin (Van den Ende et al., 1996). DID FRUCTOSYLTRANSFERASES EVOLVE FROM INVERTASES? The cloning of cdnas encoding fructosyltransferase enables us to study the properties of these enzymes in detail. It also allows us to take a closer look at their molecular relations and evolutionary origin. Biochemical analyses of fructosyltransferases have already shown that some of these enzymes also have Suc-hydrolytic (invertase) activity (e.g. barley 6-SFT; Sprenger et al., 1995). Furthermore, it has been observed that at high Suc concentrations most invertases have 1-SST activity (Obenland et al., 1993; Vijn et al., 1998). Not surprisingly, comparison of the amino acid sequences of fructosyltransferases and plant invertases reveals a high degree of identity. For example, at the amino acid level the onion acid invertase shows 61% identity to the onion 1-SST and 63% identity to the onion 6G-FFT (Vijn et al., 1998). The highest homology is found between the fructosyltransferases and the acid invertases, but identity with cell wall invertases is also substantial (approximately 39% similarity) (Fig. 5A). Recent studies of yeast invertase have revealed the catalytic mechanism for Suc hydrolysis (Reddy and Maley, 1996). The amino acids involved in Suc hydrolysis are the Asp (D) in the Suc-binding box NDPNG (Fig. 5A, region A) and the Glu (E) in box G (Fig. 5A, region G). These amino acids are also conserved among the plant invertases and fructosyltransferases, suggesting an analogous mechanism for Suc hydrolysis (Fig. 5A). However, which amino acids are involved in fructosyltransferase activity is not yet known. For bacterial levansucrases it has been shown that a single point mutation can convert the enzymes into invertases (Chambert and Petit-Glatron, 1991). The availability of the genes encoding plant invertases and fructosyltransferases now allows us to address the question of which domains of the enzymes carry the specificity for hydrolysis and Fru transfer. The close relationship between invertases and fructosyltransferases at the biochemical and molecular levels is strong evidence for the notion that fructosyltransferases evolved from invertases by relatively few mutational changes. This is especially evident in the close relationship between acid invertases and fructosyltransferases (Fig. 5B). The different fructosyltransferases cluster together with the acid or vacuolar invertases, whereas the cell wall invertases form a separate cluster (Fig. 5B). Several unrelated fructanaccumulating plant families, e.g. the Poales and Asteraceae, acquired fructosyltransferases during evolution. This polyphyletic origin of the fructan-accumulating trait raises questions concerning the nature of the selective forces that have led to the evolution of invertases to fructosyltransferases. PHYSIOLOGICAL ROLE OF FRUCTAN IN PLANTS The physiological role of fructans in plants is not fully understood. The presence of fructosyltransferases and fructans in the vacuole has been shown convincingly; fructans are probably synthesized there as well (Wagner et al., 1983; Vijn et al., 1997). However, fructan synthesis in compartments other than vacuoles, such as prevacuolar compartments, cannot yet be ruled out. In addition to its role as a major reserve carbohydrate, fructan synthesis might control Suc concentration in the vacuole. Vacuolar fructan synthesis lowers the Suc concentration in the cell and prevents sugar-induced feedback inhibition of photosynthesis (for review, see Pollock, 1986). Continuous illumination or feeding Suc to excised leaves of fructan-accumulating plants induces fructan synthesis, suggesting a correlation between high Suc levels and the induction of fructan synthesis (Wagner et al., 1983; Vijn et al., 1997). In the vacuole, fructan accumulation can reach levels in excess of 70% of dry weight without inhibiting photosynthesis. Moreover, unlike starch, fructans are soluble. Another reason for plants to use Suc or fructan as major storage carbohydrates is related to climate. The global distribution of fructan-accumulating plants shows that they are especially abundant in temperate climate zones with seasonal drought or frost, but they are almost absent in tropical regions (Hendry and Wallace, 1993). Although starch biosynthesis decreases dramatically when the temperature drops below 10 C, photosynthesis and fructan production are much less sensitive to low temperature (Pollock, 1986). For example, 1-SST of Jerusalem artichoke retains 50% of its activity at 5 C compared with its activity at optimal temperatures from 20 C to 25 C (Koops and Jonker, 1996). For the above-mentioned reasons fructan storage would be advantageous for plants that are photosynthetically active during the winter or early spring. The protection of the photosynthetic apparatus and the mobilization of stored fructan reserves for rapid growth when temperatures rise are strong factors influencing the evolution of the fructan-accumulation trait. The involvement of fructans in drought and cold tolerance has been suggested repeatedly (for review, see Wiemken et al., 1995). However, studies of environmental stress resistance are complex, and it is difficult to show a direct correlation between stress and fructan accumulation. In one recent attempt, Puebla and colleagues (1997) compared fructan synthesis in two Bromus species adapted to different climatic conditions. These two species were subjected to cold stress and water deficit, and it was found that the species adapted to a cold desert climate exhibited constitutive fructan synthesis, whereas the species adapted to a warmer climate produced fructan only under cold stress. In this study, drought did not influence fructan synthesis. Such an increased tolerance to drought was also reported by Pilon-Smits et al. (1995), who introduced a bacterial levansucrase into tobacco plants, a species normally incapable of forming fructan. These fructan-producing transgenic tobacco plants were found to be more resistant to PEG-induced drought stress, as determined by their growth properties and biomass accumulation.
6 356 Vijn and Smeekens Plant Physiol. Vol. 120, 1999 Figure 5. Comparison of amino acid sequences of plant fructosyltransferases with those of acid invertases and cell wall invertases. A, Alignment of well-conserved regions of invertases and fructosyltransferases. Letters in bold type show almost perfectly conserved amino acids. Numbers above the comparisons represent the amino acid sequence of onion (Allium cepa) 1-SST (Ac1-SST). Region A contains the so-called Suc-binding box NDPNG with the well-conserved Asp. Region G contains the well-conserved Glu, which, together with the Asp, is involved in Suc hydrolysis in invertases. B, Dendrogram showing evolutionary relatedness of sequences. The following sequences were included: 1-SST from onion (accession no. AJ ), artichoke (accession no. Y09662), Jerusalem artichoke (accession no. AJ009757), and chicory (accession no. U81520); 1-FFT from Jerusalem artichoke (accession no. AJ009756) and artichoke (accession no. AJ000481); 6G-FFT from onion (accession no. Y07838); 6-SFT from barley (accession no. X83233); acid/vacuolar invertases of onion (accession no. AJ006067), asparagus (accession no. AF002656), carrot (accession no. A67163 [DcINV] and accession no. X75351 [DcINV1-1]), tulip (accession no. X95651), tomato (accession no. D22350), bean (accession no. U92438), mung bean (accession no. D10265), potato (accession no. X70368); and cell wall invertases from Arabidopsis (accession no. X78424), tobacco (accession no. X81834), carrot (accession no. X78424), tomato (accession no. AB004558), wheat (accession no. AJ224681), and fava bean (accession no. Z35162). Abbreviations for the source plants are as follows: Nt, Nicotiana tabacum; Le, Lycopersicon esculentum; Dc, Daucus carota; Vf, Vicia faba; Ta, Triticum aestivum; At, Arabidopsis thaliana; Pv, Phaseolus vulgaris; Vr, Vigna radiata; Ci, Cichorium intybus; Ht, Helianthus tuberosus; Cs, Cynara scolymus; St, Solanum tuberosum; Ac, Allium cepa; Ao, Asparagus officinales; Hv, Hordeum vulgare; Tg, Tulipa gesneriana.
7 Fructan: More Than a Reserve Carbohydrate? 357 It should be noted that in these studies a direct correlation between the observed fructan accumulation and drought tolerance was not unequivocally shown. In the latter study (Pilon-Smits et al., 1995) the introduction of a bacterial levansucrase resulted in the production of fructans with a DP of more than 100,000. Such high-dp fructan molecules are normally not found in plants and may induce stress responses. For studies of the physiological role of fructan metabolism in plants it is preferable to use plant fructosyltransferase genes. Such genes are now available and have been transformed into several different plant species that do not normally accumulate fructans, and the performance of these transgenic plants under different environmental stresses can now be studied in detail. Another approach to studying the physiological role of fructan accumulation is by inhibiting its synthesis in plants that normally accumulate fructan. This can be achieved by silencing the endogenous fructosyltransferase genes with the introduction of additional fructosyltransferase genes, either in the antisense or in the sense orientation (cosuppression). FRUCTAN UTILITY Since the mid 1930s, fructans have been used in tests for human kidney function (Shannon and Smith, 1935), and interest in potential medical uses for inulin and inulin derivatives is growing (Fuchs, 1993). Recently, it was found that a fructan-rich diet may have health-promoting effects (Roberfroid, 1993). Fructans are a low-calorie food because they cannot be digested by humans but are instead used efficiently as a carbon source by beneficial bifidobacteria in the colon (Gibson et al., 1995). These bifidobacteria ferment fructans to short-chain fatty acids that have a positive effect on systemic lipid metabolism. Small fructans with DPs of 3 to 6 are sweet tasting and therefore constitute natural lowcaloric sweeteners. At this time the most agronomically acceptable crop for fructan production is chicory; however, the function of the fructan isolated from chicory is limited because of the degradation of long fructan chains by fructan exohydrolase upon harvesting. High-DP fructans are now being used in alimentary products where they can replace fat. Emulsions of long-chain fructans in water have organoleptic properties similar to fat. High-DP fructans also hold great promise for a variety of nonfood applications (for review, see Fuchs, 1993). However, the difficulty in obtaining long-chain and complex-branched fructans has thus far limited their application. FRUCTAN BIOTECHNOLOGY The availability of cdna clones encoding fructosyltransferases with different enzymatic activities from several plant species allows the biotechnological exploitation of fructan metabolism. Transformation of fructosyltransferases in agronomically important crops may improve the commercial availability of fructan. The introduction of 1-SST into sugarbeet and potato has shown that large amounts of short-chain fructan molecules are produced (Hellwege, 1998; Sévenier, 1998). The advantage of using crops such as sugarbeet or potato for fructan accumulation is that they lack fructan-hydrolyzing enzymes such as exohydrolase for breaking down the accumulated fructan upon harvesting. Fructan-accumulating sugarbeet is an especially promising crop because of its excellent agronomic performance in temperate zones and its natural Suc storage capacity. In another approach, novel fructan accumulation was enhanced by the introduction of heterologous fructosyltransferase genes in existing fructan-accumulating crops (Sprenger et al., 1997; Vijn et al., 1997). For example, transformation of onion 6G-FFT into chicory resulted in the production of fructan molecules of the inulin neoseries in addition to normally produced inulin, showing that the type of fructan molecule produced by a plant can be adapted (Vijn et al., 1997). In the near future it will also be possible to synthesize homogeneous pools of structurally defined fructan molecules in transgenic plants by the introduction of specific sets of fructosyltransferase genes. Pilot experiments in which combinations of protein extracts of tobacco protoplasts transformed with different fructosyltransferases were incubated with Suc produced the expected type of fructan molecules (Vijn et al., 1998). Other important strides have included the successful expression of an active barley 6-SFT in the yeast Pichia pastoris by Hochstrasser and colleagues (1998) and the expression of onion invertase and 1-SST in a mutant Saccharomyces cerevisiae strain lacking invertase (I. Vijn and S. Smeekens, unpublished data). The enzymatic activity of the cloned plant fructosyltransferases has so far been studied mainly by transient expression assays in tobacco protoplasts or by stable transformations of plants that normally do not accumulate fructan. These systems are time-consuming and laborious, contain endogenous invertase activities that compete with fructosyltransferases for the substrate, Suc, and also hide possible invertase activity of the introduced fructosyltransferases. The development of an efficient heterologous expression system for plant fructosyltransferases would allow a much more detailed biochemical characterization of these enzymes. In heterologous expression systems it will be possible to study the contribution of specific amino acids to enzymatic activity and to acceptor specificity by site-directed mutagenesis. The influence of posttranslational processing could also be studied. CONCLUSIONS AND PERSPECTIVES Since the cloning of the first fructosyltransferase cdna in 1995 (Sprenger et al., 1995), many others have been isolated. Transformation of these cdnas into plants that normally do not accumulate fructan and detailed biochemical characterization of the expressed fructosyltransferases have resulted in better insights into fructan biosynthesis in plants. Future studies of fructan synthesis in plants, especially grasses, will probably result in the identification of other fructosyltransferases with additional enzymatic activities, which will lead to a better understanding of the complexity of plant fructan biosynthesis.
8 358 Vijn and Smeekens Plant Physiol. Vol. 120, 1999 Another consequence of the cloning of fructosyltransferases is that their physiological role can now be studied in plants that normally do not accumulate fructans and in plants that inhibit fructan synthesis. Studying the responses of these transgenic plants to environmental stresses such as drought and cold may lead to greater insight into the physiological role of fructans in stress resistance. Furthermore, the transformation of the cloned fructosyltransferases into agronomically important crops shows that such crops have great potential as fructan sources and that it may soon be possible to produce a range of structurally different fructan molecules. Received February 2, 1999; accepted March 4, LITERATURE CITED Bonnett GD, Sims IM, John JAS, Simpson RJ (1994) Purification and characterization of fructans with -2,1- and -2,6-glycosidic linkages suitable for enzyme studies. New Phytol 127: Bonnett GD, Sims IM, Simpson RJ, Cairns AJ (1997) Structural diversity of fructan in relation to the taxonomy of the Poaceae. New Phytol 136: Carpita NC, Kanabus J, Housley TL (1989) Linkage structure of fructans and fructan oligomers from Triticum aestivum and Festuca arundinaceae leaves. J Plant Physiol 134: Chambert R, Petit-Glatron MF (1991) Polymerase and hydrolase activities of Bacillus subtilis levansucrase can be separately modulated by site-directed mutagenesis. J Biochem 279: Cote GL, Ahlgren J (1993) Metabolism in microorganisms, Part I. Levan and levansucrase. In M Suzuki, NJ Chatterton, eds, Science and Technology of Fructans. CRC Press, Boca Raton, FL, pp Dedonder R (1966) Levansucrase from Bacillus subtilis. Methods Enzymol 8: de Halleux S, Van Cutsem P (1997) Cloning and sequencing of the 1-SST cdna from chicory root (accession no. U81520) (PGR ). Plant Physiol 113: 1003 Duchateau N, Bortlik K, Simmen U, Wiemken A, Bancal P (1995) Sucrose:fructan 6-fructosyltransferase, a key enzyme for diverting carbon from sucrose to fructan in barley leaves. Plant Physiol 107: Edelman J, Jefford TG (1968) The mechanism of fructosan metabolism in plants as exemplified in Helianthus tuberosis. New Phytol 67: Ernst M, Chatterton NJ, Harrison PA (1996) Purification and characterization of a new fructan series from species of Asteraceae. New Phytol 132: Fuchs A, ed (1993) Inulin and Inulin Containing Crops: Studies in Plant Science. Elsevier, Amsterdam Gibson GR, Beatty ER, Wang X, Cummings JH (1995) Selective stimulation of bifidobacteria in the human colon by oligofructose and inulin. Gastroenterology 108: Hellwege EM, Gritscher D, Willmitzer L, Heyer AG (1997) Transgenic potato tubers accumulate high levels of 1-kestose and nystose: functional identification of a sucrose:sucrose 1-fructosyltransferase of artichoke (Cynara scolymus) blossom discs. Plant J 12: Hellwege EM, Raap M, Gritscher D, Willmitzer L, Heyer AG (1998) Differences in chain length distribution of inulin from Cynara scolymus and Helianthus tuberosus are reflected in a transient plant expression system using the respective 1-FFT cdnas. FEBS Lett 427: Hendry GAF, Wallace RK (1993) The origin, distribution, and evolutionary significance of fructans. In M Suzuki, NJ Chatterton, eds, Science and Technology of Fructans. CRC Press, Boca Raton, FL, pp Henson CA, Livingstone DP III (1996) Purification and characterization of an oat fructan exohydrolase that preferentially hydrolyzes -2,6-fructans. Plant Physiol 110: Hochstrasser U, Lüscher M, De Virgilio C, Boller T, Wiemken A (1998) Expression of a functional 6-fructosyltransferase in the methylotrophic yeast Pichia pastoris. FEBS Lett 440: Koops AJ, Jonker HH (1996) Purification and characterization of the enzymes of fructan biosynthesis in tubers of Helianthus tuberosus Colombia. II. Purification of sucrose:sucrose 1-fructosyltransferase and reconstitution of fructan synthesis in vitro with purified sucrose:sucrose 1-fructosyltransferase and fructan:fructan 1-fructosyltransferase. Plant Physiol 110: Livingstone DP III, Chatterton NJ, Harrison PA (1993) Structure and quantity of fructan oligomers in oat (Avena spp.). New Phytol 123: Lüscher M, Erdin C, Sprenger N, Hochstrasser U, Boller T, Wiemken A (1996) Inulin synthesis by a combination of purified fructosyltransferases from tubers of Helianthus tuberosus. FEBS Lett 385: Marx SP, Nösberger J, Frehner M (1997a) Seasonal variation of fructan- -fructosidase (FEH) activity and characterization of a -(2-1)-linkage-specific FEH from tubers of Jerusalem artichoke (Helianthus tuberosus). New Phytol 135: Marx SP, Nösberger J, Frehner M (1997b) Hydrolysis of fructan in grasses: a -(2-6)-linkage-specific fructan- -fructosidase from stubble of Lolium perenne. New Phytol 135: Obenland DM, Simmen U, Boller T, Wiemken A (1993) Purification and characterization of three soluble invertases from barley (Hordeum vulgare L.) leaves. Plant Physiol 89: Pilon-Smits EAH, Ebskamp MJM, Paul MJ, Jeuken MJW, Weisbeek PJ, Smeekens SCM (1995) Improved performance of transgenic fructan-accumulating tobacco under drought stress. Plant Physiol 107: Pollock CJ (1986) Fructans and the metabolism of sucrose in vascular plants. New Phytol 104: 1 24 Pollock CJ, Cairns AJ (1991) Fructan metabolism in grasses and cereals. Annu Rev Plant Physiol Plant Mol Biol 42: Praznik W, Beck RHF (1985) Application of gel permeation chromatographic systems to the determination of the molecular weight of inulin. J Chromatogr 348: Puebla AF, Salerno GL, Pontis HG (1997) Fructan metabolism in two species of Bromus subjected to chilling and water stress. New Phytol 136: Reddy A, Maley F (1996) Studies on identifying the catalytic role of Glu-204 in the active site of yeast invertase. J Biol Chem 271: Roberfroid M (1993) Dietary fiber, inulin and oligofructose: a review comparing their physiological effects. Crit Rev Food Sci Nutr 33: Sévenier R, Hall RD, Van der Meer IM, Hakkert HJC, Van Tunen AJ, Koops AJ (1998) High level fructan accumulation in a transgenic sugarbeet. Nat Biotechnol 16: Shannon JA, Smith HW (1935) The excretion of inulin, xylose and urea by normal and phlorizinized man. J Clin Invest 14: Shiomi N (1989) Properties of fructosyltransferases involved in the synthesis of fructan in liliaceous plants. J Plant Physiol 134: Sprenger N, Bortlik K, Brandt A, Boller T, Wiemken A (1995) Purification, cloning, and functional expression of sucrose:fructan 6-fructosyltransferase, a key enzyme of fructan synthesis in barley. Proc Natl Acad Sci USA 92: Sprenger N, Schellenbaum L, van Dun K, Boller T, Wiemken A (1997) Fructan synthesis in transgenic tobacco and chicory plants expressing barley sucrose:fructan 6-fructosyltransferase. FEBS Lett 400: Uchiyama T (1993) Metabolism in microorganisms, Part II. Biosynthesis and degradation of fructans by microbial enzymes other than levansucrase. In M Suzuki, NJ Chatterton, eds, Science and Technology of Fructans. CRC Press, Boca Raton, FL, pp Van den Ende W, De Roover J, Van Laere A (1996) In vitro synthesis of fructofuranosyl-only oligosaccharides from inulin and fructose by purified chicory root fructan:fructan fructosyltransferase. Physiol Plant 97:
9 Fructan: More Than a Reserve Carbohydrate? 359 Van den Ende W, Van Laere A (1996) De-novo synthesis of fructans from sucrose in vitro by a combination of two purified enzymes (sucrose:sucrose 1-fructosyltransferase and fructan: fructan 1-fructosyltransferase) from chicory roots (Cichorium intybus L.). Planta 200: Van der Meer IM, Koops AJ, Hakkert JC, Van Tunen AJ (1998) Cloning of the fructan biosynthesis pathway of Jerusalem artichoke. Plant J 15: Vijn I, van Dijken A, Lüscher M, Bos A, Smeets E, Weisbeek P, Wiemken A, Smeekens S (1998) Cloning of sucrose:sucrose 1-fructosyltransferase from onion and synthesis of structurally defined fructan molecules from sucrose. Plant Physiol 117: Vijn I, van Dijken A, Sprenger N, van Dun K, Weisbeek P, Wiemken A, Smeekens S (1997) Fructan of the inulin neoseries is synthesized in transgenic chicory plants (Cichorium intybus L.) harbouring onion (Allium cepa L.) fructan:fructan 6Gfructosyltransferase. Plant J 11: Wagner W, Keller F, Wiemken A (1983) Fructan metabolism in cereals: induction in leaves and compartmentation in protoplasts and vacuoles. Z Pflanzenphysiol 112: Wiemken A, Sprenger N, Boller T (1995) Fructan: an extension of sucrose by sucrose. In HG Pontis, GL Salerno, EJ Echeverria, eds, Sucrose Metabolism, Biochemistry, Physiology and Molecular Biology. American Society of Plant Physiologists, Rockville, MD, pp
Sugars, Starches, and Fibers Are All Carbohydrates
 Sugars, Starches, and Fibers Are All Carbohydrates What are carbohydrates? Today's food advertisements call them carbs, but they are not all the same. They are a group of compounds that have some similarities
Sugars, Starches, and Fibers Are All Carbohydrates What are carbohydrates? Today's food advertisements call them carbs, but they are not all the same. They are a group of compounds that have some similarities
1. The diagram below represents a biological process
 1. The diagram below represents a biological process 5. The chart below indicates the elements contained in four different molecules and the number of atoms of each element in those molecules. Which set
1. The diagram below represents a biological process 5. The chart below indicates the elements contained in four different molecules and the number of atoms of each element in those molecules. Which set
GENE CLONING AND RECOMBINANT DNA TECHNOLOGY
 GENE CLONING AND RECOMBINANT DNA TECHNOLOGY What is recombinant DNA? DNA from 2 different sources (often from 2 different species) are combined together in vitro. Recombinant DNA forms the basis of cloning.
GENE CLONING AND RECOMBINANT DNA TECHNOLOGY What is recombinant DNA? DNA from 2 different sources (often from 2 different species) are combined together in vitro. Recombinant DNA forms the basis of cloning.
The Structure and Function of Macromolecules: Carbohydrates, Lipids & Phospholipids
 The Structure and Function of Macromolecules: Carbohydrates, Lipids & Phospholipids The FOUR Classes of Large Biomolecules All living things are made up of four classes of large biological molecules: Carbohydrates
The Structure and Function of Macromolecules: Carbohydrates, Lipids & Phospholipids The FOUR Classes of Large Biomolecules All living things are made up of four classes of large biological molecules: Carbohydrates
Enzymes: Practice Questions #1
 Enzymes: Practice Questions #1 1. Compound X increases the rate of the reaction below. Compound X is most likely A. an enzyme B. a lipid molecule C. an indicator D. an ADP molecule 2. The equation below
Enzymes: Practice Questions #1 1. Compound X increases the rate of the reaction below. Compound X is most likely A. an enzyme B. a lipid molecule C. an indicator D. an ADP molecule 2. The equation below
DOE Office of Biological & Environmental Research: Biofuels Strategic Plan
 DOE Office of Biological & Environmental Research: Biofuels Strategic Plan I. Current Situation The vast majority of liquid transportation fuel used in the United States is derived from fossil fuels. In
DOE Office of Biological & Environmental Research: Biofuels Strategic Plan I. Current Situation The vast majority of liquid transportation fuel used in the United States is derived from fossil fuels. In
Cloning of the fructan biosynthesis pathway of Jerusalem artichoke
 The Plant Journal (1998) 15(4), 489 500 Cloning of the fructan biosynthesis pathway of Jerusalem artichoke Ingrid M. van der Meer*, Andries J. Koops, Johanna C. Hakkert and Arjen J. van Tunen DLO-Centre
The Plant Journal (1998) 15(4), 489 500 Cloning of the fructan biosynthesis pathway of Jerusalem artichoke Ingrid M. van der Meer*, Andries J. Koops, Johanna C. Hakkert and Arjen J. van Tunen DLO-Centre
Metabolism Poster Questions
 Metabolism Poster Questions Answer the following questions concerning respiration. 1. Consider the mitochondrial electron transport chain. a. How many hydrogen ions can be pumped for every NADH? b. How
Metabolism Poster Questions Answer the following questions concerning respiration. 1. Consider the mitochondrial electron transport chain. a. How many hydrogen ions can be pumped for every NADH? b. How
Carbon-organic Compounds
 Elements in Cells The living substance of cells is made up of cytoplasm and the structures within it. About 96% of cytoplasm and its included structures are composed of the elements carbon, hydrogen, oxygen,
Elements in Cells The living substance of cells is made up of cytoplasm and the structures within it. About 96% of cytoplasm and its included structures are composed of the elements carbon, hydrogen, oxygen,
Unit I: Introduction To Scientific Processes
 Unit I: Introduction To Scientific Processes This unit is an introduction to the scientific process. This unit consists of a laboratory exercise where students go through the QPOE2 process step by step
Unit I: Introduction To Scientific Processes This unit is an introduction to the scientific process. This unit consists of a laboratory exercise where students go through the QPOE2 process step by step
Methods of Grading S/N Style of grading Percentage Score 1 Attendance, class work and assignment 10 2 Test 20 3 Examination 70 Total 100
 COURSE: MIB 303 Microbial Physiology and Metabolism (3 Units- Compulsory) Course Duration: Three hours per week for 15 weeks (45 hours). Lecturer: Jimoh, S.O. B.Sc., M.Sc, Ph.D Microbiology (ABU, Zaria)
COURSE: MIB 303 Microbial Physiology and Metabolism (3 Units- Compulsory) Course Duration: Three hours per week for 15 weeks (45 hours). Lecturer: Jimoh, S.O. B.Sc., M.Sc, Ph.D Microbiology (ABU, Zaria)
Regents Biology REGENTS REVIEW: PROTEIN SYNTHESIS
 Period Date REGENTS REVIEW: PROTEIN SYNTHESIS 1. The diagram at the right represents a portion of a type of organic molecule present in the cells of organisms. What will most likely happen if there is
Period Date REGENTS REVIEW: PROTEIN SYNTHESIS 1. The diagram at the right represents a portion of a type of organic molecule present in the cells of organisms. What will most likely happen if there is
Human Physiology Lab (Biol 236L) Digestive Physiology: Amylase hydrolysis of starch
 Human Physiology Lab (Biol 236L) Digestive Physiology: Amylase hydrolysis of starch Introduction Enzymes are proteins composed of amino acid building blocks. Enzymes catalyze or increase the rate of metabolic
Human Physiology Lab (Biol 236L) Digestive Physiology: Amylase hydrolysis of starch Introduction Enzymes are proteins composed of amino acid building blocks. Enzymes catalyze or increase the rate of metabolic
Disaccharides consist of two monosaccharide monomers covalently linked by a glycosidic bond. They function in sugar transport.
 1. The fundamental life processes of plants and animals depend on a variety of chemical reactions that occur in specialized areas of the organism s cells. As a basis for understanding this concept: 1.
1. The fundamental life processes of plants and animals depend on a variety of chemical reactions that occur in specialized areas of the organism s cells. As a basis for understanding this concept: 1.
Cellular Respiration: Practice Questions #1
 Cellular Respiration: Practice Questions #1 1. Which statement best describes one of the events taking place in the chemical reaction? A. Energy is being stored as a result of aerobic respiration. B. Fermentation
Cellular Respiration: Practice Questions #1 1. Which statement best describes one of the events taking place in the chemical reaction? A. Energy is being stored as a result of aerobic respiration. B. Fermentation
O 2. What is anaerobic digestion?
 What is anaerobic digestion? Microbial degradation of organic material under anaerobic conditions Ubiquitous, naturally-occurring process Occurs in swamps, hydric soils, landfills, digestive tracks of
What is anaerobic digestion? Microbial degradation of organic material under anaerobic conditions Ubiquitous, naturally-occurring process Occurs in swamps, hydric soils, landfills, digestive tracks of
Biochemistry of Cells
 Biochemistry of Cells 1 Carbon-based Molecules Although a cell is mostly water, the rest of the cell consists mostly of carbon-based molecules Organic chemistry is the study of carbon compounds Carbon
Biochemistry of Cells 1 Carbon-based Molecules Although a cell is mostly water, the rest of the cell consists mostly of carbon-based molecules Organic chemistry is the study of carbon compounds Carbon
* Is chemical energy potential or kinetic energy? The position of what is storing energy?
 Biology 1406 Exam 2 - Metabolism Chs. 5, 6 and 7 energy - capacity to do work 5.10 kinetic energy - energy of motion : light, electrical, thermal, mechanical potential energy - energy of position or stored
Biology 1406 Exam 2 - Metabolism Chs. 5, 6 and 7 energy - capacity to do work 5.10 kinetic energy - energy of motion : light, electrical, thermal, mechanical potential energy - energy of position or stored
Course Curriculum for Master Degree in Food Science and Technology/ Department of Nutrition and Food Technology
 Course Curriculum for Master Degree in Food Science and Technology/ Department of Nutrition and Food Technology The Master Degree in Food Science and Technology / Department of Nutrition and Food Technology,
Course Curriculum for Master Degree in Food Science and Technology/ Department of Nutrition and Food Technology The Master Degree in Food Science and Technology / Department of Nutrition and Food Technology,
The correct answer is d C. Answer c is incorrect. Reliance on the energy produced by others is a characteristic of heterotrophs.
 1. An autotroph is an organism that a. extracts energy from organic sources b. converts energy from sunlight into chemical energy c. relies on the energy produced by other organisms as an energy source
1. An autotroph is an organism that a. extracts energy from organic sources b. converts energy from sunlight into chemical energy c. relies on the energy produced by other organisms as an energy source
What affects an enzyme s activity? General environmental factors, such as temperature and ph. Chemicals that specifically influence the enzyme.
 CH s 8-9 Respiration & Metabolism Metabolism A catalyst is a chemical agent that speeds up a reaction without being consumed by the reaction. An enzyme is a catalytic protein. Hydrolysis of sucrose by
CH s 8-9 Respiration & Metabolism Metabolism A catalyst is a chemical agent that speeds up a reaction without being consumed by the reaction. An enzyme is a catalytic protein. Hydrolysis of sucrose by
GUIDELINES FOR THE REGISTRATION OF BIOLOGICAL PEST CONTROL AGENTS FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS
 GUIDELINES FOR THE REGISTRATION OF BIOLOGICAL PEST CONTROL AGENTS FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS -ii- GUIDELINES ON THE REGISTRATION OF BIOLOGICAL PEST CONTROL AGENTS FOOD AND
GUIDELINES FOR THE REGISTRATION OF BIOLOGICAL PEST CONTROL AGENTS FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS -ii- GUIDELINES ON THE REGISTRATION OF BIOLOGICAL PEST CONTROL AGENTS FOOD AND
CCR Biology - Chapter 9 Practice Test - Summer 2012
 Name: Class: Date: CCR Biology - Chapter 9 Practice Test - Summer 2012 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Genetic engineering is possible
Name: Class: Date: CCR Biology - Chapter 9 Practice Test - Summer 2012 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Genetic engineering is possible
Student name ID # 2. (4 pts) What is the terminal electron acceptor in respiration? In photosynthesis? O2, NADP+
 1. Membrane transport. A. (4 pts) What ion couples primary and secondary active transport in animal cells? What ion serves the same function in plant cells? Na+, H+ 2. (4 pts) What is the terminal electron
1. Membrane transport. A. (4 pts) What ion couples primary and secondary active transport in animal cells? What ion serves the same function in plant cells? Na+, H+ 2. (4 pts) What is the terminal electron
8/20/2012 H C OH H R. Proteins
 Proteins Rubisco monomer = amino acids 20 different amino acids polymer = polypeptide protein can be one or more polypeptide chains folded & bonded together large & complex 3-D shape hemoglobin Amino acids
Proteins Rubisco monomer = amino acids 20 different amino acids polymer = polypeptide protein can be one or more polypeptide chains folded & bonded together large & complex 3-D shape hemoglobin Amino acids
Chapter 5: The Structure and Function of Large Biological Molecules
 Name Period Concept 5.1 Macromolecules are polymers, built from monomers 1. The large molecules of all living things fall into just four main classes. Name them. 2. Circle the three classes that are called
Name Period Concept 5.1 Macromolecules are polymers, built from monomers 1. The large molecules of all living things fall into just four main classes. Name them. 2. Circle the three classes that are called
Photosynthesis (Life from Light)
 Photosynthesis Photosynthesis (Life from Light) Energy needs of life All life needs a constant input of energy o Heterotrophs (consumers) Animals, fungi, most bacteria Get their energy from other organisms
Photosynthesis Photosynthesis (Life from Light) Energy needs of life All life needs a constant input of energy o Heterotrophs (consumers) Animals, fungi, most bacteria Get their energy from other organisms
Keystone Review Practice Test Module A Cells and Cell Processes. 1. Which characteristic is shared by all prokaryotes and eukaryotes?
 Keystone Review Practice Test Module A Cells and Cell Processes 1. Which characteristic is shared by all prokaryotes and eukaryotes? a. Ability to store hereditary information b. Use of organelles to control
Keystone Review Practice Test Module A Cells and Cell Processes 1. Which characteristic is shared by all prokaryotes and eukaryotes? a. Ability to store hereditary information b. Use of organelles to control
Anabolic and Catabolic Reactions are Linked by ATP in Living Organisms
 Chapter 5: Microbial Metabolism Microbial Metabolism Metabolism refers to all chemical reactions that occur within a living a living organism. These chemical reactions are generally of two types: Catabolic:
Chapter 5: Microbial Metabolism Microbial Metabolism Metabolism refers to all chemical reactions that occur within a living a living organism. These chemical reactions are generally of two types: Catabolic:
AP BIOLOGY CHAPTER 7 Cellular Respiration Outline
 AP BIOLOGY CHAPTER 7 Cellular Respiration Outline I. How cells get energy. A. Cellular Respiration 1. Cellular respiration includes the various metabolic pathways that break down carbohydrates and other
AP BIOLOGY CHAPTER 7 Cellular Respiration Outline I. How cells get energy. A. Cellular Respiration 1. Cellular respiration includes the various metabolic pathways that break down carbohydrates and other
Lipids in Photosynthesis: Structure, Function and Genetics
 -5Z-/7- Lipids in Photosynthesis: Structure, Function and Genetics Edited by Paul-Andre Siegenthaler Laboratoire de Physiologie Vegetale, Universite de Neuchatel, Neuchatel, Switzerland and Norio Murata
-5Z-/7- Lipids in Photosynthesis: Structure, Function and Genetics Edited by Paul-Andre Siegenthaler Laboratoire de Physiologie Vegetale, Universite de Neuchatel, Neuchatel, Switzerland and Norio Murata
LAB 3: DIGESTION OF ORGANIC MACROMOLECULES
 LAB 3: DIGESTION OF ORGANIC MACROMOLECULES INTRODUCTION Enzymes are a special class of proteins that lower the activation energy of biological reactions. These biological catalysts change the rate of chemical
LAB 3: DIGESTION OF ORGANIC MACROMOLECULES INTRODUCTION Enzymes are a special class of proteins that lower the activation energy of biological reactions. These biological catalysts change the rate of chemical
Just the Facts: A Basic Introduction to the Science Underlying NCBI Resources
 1 of 8 11/7/2004 11:00 AM National Center for Biotechnology Information About NCBI NCBI at a Glance A Science Primer Human Genome Resources Model Organisms Guide Outreach and Education Databases and Tools
1 of 8 11/7/2004 11:00 AM National Center for Biotechnology Information About NCBI NCBI at a Glance A Science Primer Human Genome Resources Model Organisms Guide Outreach and Education Databases and Tools
INDUSTRIAL BIOTECHNOLOGY. Production hosts for real-life feedstock utilization
 Selection of production hosts for real-life feedstock utilization Karl Rumbold (karl.rumbold@tno.nl) INDUSTRIAL BIOTECHNOLOGY Industrial Biotechnology is the application of biotechnology for the processing
Selection of production hosts for real-life feedstock utilization Karl Rumbold (karl.rumbold@tno.nl) INDUSTRIAL BIOTECHNOLOGY Industrial Biotechnology is the application of biotechnology for the processing
Biological molecules:
 Biological molecules: All are organic (based on carbon). Monomers vs. polymers: Monomers refer to the subunits that, when polymerized, make up a larger polymer. Monomers may function on their own in some
Biological molecules: All are organic (based on carbon). Monomers vs. polymers: Monomers refer to the subunits that, when polymerized, make up a larger polymer. Monomers may function on their own in some
Visualizing Cell Processes
 Visualizing Cell Processes A Series of Five Programs produced by BioMEDIA ASSOCIATES Content Guide for Program 3 Photosynthesis and Cellular Respiration Copyright 2001, BioMEDIA ASSOCIATES www.ebiomedia.com
Visualizing Cell Processes A Series of Five Programs produced by BioMEDIA ASSOCIATES Content Guide for Program 3 Photosynthesis and Cellular Respiration Copyright 2001, BioMEDIA ASSOCIATES www.ebiomedia.com
Chair of Chemistry of Biogenic Resources TU München - R&D activities -
 Chair of Chemistry of Biogenic Resources TU München - R&D activities - Chair of Chemistry of Biogenic Resources Schulgasse 18, 94315 Straubing, Germany Sieber@tum.de www.rohstoffwandel.de Locations Freising
Chair of Chemistry of Biogenic Resources TU München - R&D activities - Chair of Chemistry of Biogenic Resources Schulgasse 18, 94315 Straubing, Germany Sieber@tum.de www.rohstoffwandel.de Locations Freising
Catalysis by Enzymes. Enzyme A protein that acts as a catalyst for a biochemical reaction.
 Catalysis by Enzymes Enzyme A protein that acts as a catalyst for a biochemical reaction. Enzymatic Reaction Specificity Enzyme Cofactors Many enzymes are conjugated proteins that require nonprotein portions
Catalysis by Enzymes Enzyme A protein that acts as a catalyst for a biochemical reaction. Enzymatic Reaction Specificity Enzyme Cofactors Many enzymes are conjugated proteins that require nonprotein portions
Carbohydrates, proteins and lipids
 Carbohydrates, proteins and lipids Chapter 3 MACROMOLECULES Macromolecules: polymers with molecular weights >1,000 Functional groups THE FOUR MACROMOLECULES IN LIFE Molecules in living organisms: proteins,
Carbohydrates, proteins and lipids Chapter 3 MACROMOLECULES Macromolecules: polymers with molecular weights >1,000 Functional groups THE FOUR MACROMOLECULES IN LIFE Molecules in living organisms: proteins,
Production of prebiotics from PATRICK ADLERCREUTZ, DIV. OF BIOTECHNOLOGY
 Production of prebiotics from hemicellulose PATRICK ADLERCREUTZ, DIV. OF BIOTECHNOLOGY Gut microbiota Gut microbiota microorganisms in our gastrointestinal tract The gut microbiota has a large influence
Production of prebiotics from hemicellulose PATRICK ADLERCREUTZ, DIV. OF BIOTECHNOLOGY Gut microbiota Gut microbiota microorganisms in our gastrointestinal tract The gut microbiota has a large influence
Site-Directed Nucleases and Cisgenesis Maria Fedorova, Ph.D.
 Site-Directed Nucleases and Cisgenesis Maria Fedorova, Ph.D. Regulatory Strategy Lead Enabling Technologies DuPont-Pioneer, USA 1 New Plant Breeding Techniques 2007 New Techniques Working Group established
Site-Directed Nucleases and Cisgenesis Maria Fedorova, Ph.D. Regulatory Strategy Lead Enabling Technologies DuPont-Pioneer, USA 1 New Plant Breeding Techniques 2007 New Techniques Working Group established
4. Which carbohydrate would you find as part of a molecule of RNA? a. Galactose b. Deoxyribose c. Ribose d. Glucose
 1. How is a polymer formed from multiple monomers? a. From the growth of the chain of carbon atoms b. By the removal of an OH group and a hydrogen atom c. By the addition of an OH group and a hydrogen
1. How is a polymer formed from multiple monomers? a. From the growth of the chain of carbon atoms b. By the removal of an OH group and a hydrogen atom c. By the addition of an OH group and a hydrogen
Twincore - Zentrum für Experimentelle und Klinische Infektionsforschung Institut für Molekulare Bakteriologie
 Twincore - Zentrum für Experimentelle und Klinische Infektionsforschung Institut für Molekulare Bakteriologie 0 HELMHOLTZ I ZENTRUM FÜR INFEKTIONSFORSCHUNG Technische Universität Braunschweig Institut
Twincore - Zentrum für Experimentelle und Klinische Infektionsforschung Institut für Molekulare Bakteriologie 0 HELMHOLTZ I ZENTRUM FÜR INFEKTIONSFORSCHUNG Technische Universität Braunschweig Institut
pathway that involves taking in heat from the environment at each step. C.
 Study Island Cell Energy Keystone Review 1. Cells obtain energy by either capturing light energy through photosynthesis or by breaking down carbohydrates through cellular respiration. In both photosynthesis
Study Island Cell Energy Keystone Review 1. Cells obtain energy by either capturing light energy through photosynthesis or by breaking down carbohydrates through cellular respiration. In both photosynthesis
Working With Enzymes. a world of learning. Introduction. How Enzymes Work. Types and Sources of Enzymes
 Working With Enzymes a world of learning Presented by Peter J Ball, Southern Biological. For further information, please contact the author by phone (03) 9877-4597 or by email peterjball@southernbiological.com.
Working With Enzymes a world of learning Presented by Peter J Ball, Southern Biological. For further information, please contact the author by phone (03) 9877-4597 or by email peterjball@southernbiological.com.
Regulation of enzyme activity
 1 Regulation of enzyme activity Regulation of enzyme activity is important to coordinate the different metabolic processes. It is also important for homeostasis i.e. to maintain the internal environment
1 Regulation of enzyme activity Regulation of enzyme activity is important to coordinate the different metabolic processes. It is also important for homeostasis i.e. to maintain the internal environment
Protein Expression. A Practical Approach J. HIGGIN S
 Protein Expression A Practical Approach S. J. HIGGIN S B. D. HAMES List of contributors Abbreviations xv Xvi i 1. Protein expression in mammalian cell s Marlies Otter-Nilsson and Tommy Nilsso n 1. Introduction
Protein Expression A Practical Approach S. J. HIGGIN S B. D. HAMES List of contributors Abbreviations xv Xvi i 1. Protein expression in mammalian cell s Marlies Otter-Nilsson and Tommy Nilsso n 1. Introduction
"Fingerprinting" Vegetables DNA-based Marker Assisted Selection
 "Fingerprinting" Vegetables DNA-based Marker Assisted Selection Faster, Cheaper, More Reliable; These are some of the goals that vegetable breeders at seed companies and public institutions desire for
"Fingerprinting" Vegetables DNA-based Marker Assisted Selection Faster, Cheaper, More Reliable; These are some of the goals that vegetable breeders at seed companies and public institutions desire for
10.1 The function of Digestion pg. 402
 10.1 The function of Digestion pg. 402 Macromolecules and Living Systems The body is made up of more than 60 % water. The water is found in the cells cytoplasm, the interstitial fluid and the blood (5
10.1 The function of Digestion pg. 402 Macromolecules and Living Systems The body is made up of more than 60 % water. The water is found in the cells cytoplasm, the interstitial fluid and the blood (5
Copyright 2000-2003 Mark Brandt, Ph.D. 54
 Pyruvate Oxidation Overview of pyruvate metabolism Pyruvate can be produced in a variety of ways. It is an end product of glycolysis, and can be derived from lactate taken up from the environment (or,
Pyruvate Oxidation Overview of pyruvate metabolism Pyruvate can be produced in a variety of ways. It is an end product of glycolysis, and can be derived from lactate taken up from the environment (or,
MULTIPLE CHOICE QUESTIONS
 MULTIPLE CHOICE QUESTIONS 1. Most components of energy conversion systems evolved very early; thus, the most fundamental aspects of energy metabolism tend to be: A. quite different among a diverse group
MULTIPLE CHOICE QUESTIONS 1. Most components of energy conversion systems evolved very early; thus, the most fundamental aspects of energy metabolism tend to be: A. quite different among a diverse group
Overview on EFSA data requirements for the safety evaluation of food enzymes applications
 Overview on EFSA data requirements for the safety evaluation of food enzymes applications Fidel Toldrá and Klaus-Dieter Jany EFSA CEF Panel Info session on Food Enzymes applications Parma, 27 May 2014
Overview on EFSA data requirements for the safety evaluation of food enzymes applications Fidel Toldrá and Klaus-Dieter Jany EFSA CEF Panel Info session on Food Enzymes applications Parma, 27 May 2014
Introduction to Proteins and Enzymes
 Introduction to Proteins and Enzymes Basics of protein structure and composition The life of a protein Enzymes Theory of enzyme function Not all enzymes are proteins / not all proteins are enzymes Enzyme
Introduction to Proteins and Enzymes Basics of protein structure and composition The life of a protein Enzymes Theory of enzyme function Not all enzymes are proteins / not all proteins are enzymes Enzyme
Rubisco; easy Purification and Immunochemical Determination
 Rubisco; easy Purification and Immunochemical Determination Ulrich Groß Justus-Liebig-Universität Gießen, Institute of Plant Nutrition, Department of Tissue Culture, Südanlage 6, D-35390 Giessen e-mail:
Rubisco; easy Purification and Immunochemical Determination Ulrich Groß Justus-Liebig-Universität Gießen, Institute of Plant Nutrition, Department of Tissue Culture, Südanlage 6, D-35390 Giessen e-mail:
Nucleotides and Nucleic Acids
 Nucleotides and Nucleic Acids Brief History 1 1869 - Miescher Isolated nuclein from soiled bandages 1902 - Garrod Studied rare genetic disorder: Alkaptonuria; concluded that specific gene is associated
Nucleotides and Nucleic Acids Brief History 1 1869 - Miescher Isolated nuclein from soiled bandages 1902 - Garrod Studied rare genetic disorder: Alkaptonuria; concluded that specific gene is associated
Production of antigens and antibodies in plants: alternative technology?
 Production of antigens and antibodies in plants: alternative technology? George Lomonossoff John Innes Centre Norwich, UK ECOPA, Alicante 29 th Sept. 2006 Why use Plants as Biofactories? Produce large
Production of antigens and antibodies in plants: alternative technology? George Lomonossoff John Innes Centre Norwich, UK ECOPA, Alicante 29 th Sept. 2006 Why use Plants as Biofactories? Produce large
Curriculum Policy of the Graduate School of Agricultural Science, Graduate Program
 Curriculum Policy of the Graduate School of Agricultural Science, Graduate Program Agricultural Science plans to conserve natural and artificial ecosystems and its ideal of "Sustainable coexistence science"
Curriculum Policy of the Graduate School of Agricultural Science, Graduate Program Agricultural Science plans to conserve natural and artificial ecosystems and its ideal of "Sustainable coexistence science"
CURRICULUM VITAE. Yong-Cheol Park, Ph.D.
 CURRICULUM VITAE Yong-Cheol Park, Ph.D. Postdoctoral Research Associate Department of Biochemistry and Cell Biology, Rice University, Houston, TX 77005-1892, U.S.A. Tel) 1-713-348-3304, Fax) 1-713-348-5154,
CURRICULUM VITAE Yong-Cheol Park, Ph.D. Postdoctoral Research Associate Department of Biochemistry and Cell Biology, Rice University, Houston, TX 77005-1892, U.S.A. Tel) 1-713-348-3304, Fax) 1-713-348-5154,
Mammalian digestive tracts
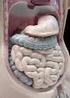 Mammalian digestive tracts Mouth: mastication, some digestive enzymes Esophagus: simple transport tube Stomach: most initial digestion, some physical processing Small intestine: digestion continues, some
Mammalian digestive tracts Mouth: mastication, some digestive enzymes Esophagus: simple transport tube Stomach: most initial digestion, some physical processing Small intestine: digestion continues, some
Chapter 5. The Structure and Function of Macromolecule s
 Chapter 5 The Structure and Function of Macromolecule s Most Macromolecules are polymers: Polymer: (poly: many; mer: part) Large molecules consisting of many identical or similar subunits connected together.
Chapter 5 The Structure and Function of Macromolecule s Most Macromolecules are polymers: Polymer: (poly: many; mer: part) Large molecules consisting of many identical or similar subunits connected together.
Lecture Overview. Hydrogen Bonds. Special Properties of Water Molecules. Universal Solvent. ph Scale Illustrated. special properties of water
 Lecture Overview special properties of water > water as a solvent > ph molecules of the cell > properties of carbon > carbohydrates > lipids > proteins > nucleic acids Hydrogen Bonds polarity of water
Lecture Overview special properties of water > water as a solvent > ph molecules of the cell > properties of carbon > carbohydrates > lipids > proteins > nucleic acids Hydrogen Bonds polarity of water
Understanding Feed Analysis Terminology
 Understanding Feed Analysis Terminology One of the most important steps in developing a ration suitable for dairy animals is feed testing. It is essential to have a starting point in order to formulate
Understanding Feed Analysis Terminology One of the most important steps in developing a ration suitable for dairy animals is feed testing. It is essential to have a starting point in order to formulate
Essentials of Human Anatomy & Physiology 11 th Edition, 2015 Marieb
 A Correlation of Essentials of Human Anatomy Marieb To the Next Generation Science Standards Life A Correlation of, HS-LS1 From Molecules to Organisms: Structures and Processes HS-LS1-1. Construct an explanation
A Correlation of Essentials of Human Anatomy Marieb To the Next Generation Science Standards Life A Correlation of, HS-LS1 From Molecules to Organisms: Structures and Processes HS-LS1-1. Construct an explanation
Chapter 2. The Chemistry of Life Worksheets
 Chapter 2 The Chemistry of Life Worksheets (Opening image courtesy of David Iberri, http://en.wikipedia.org/wiki/file:camkii.png, and under the Creative Commons license CC-BY-SA 3.0.) Lesson 2.1: Matter
Chapter 2 The Chemistry of Life Worksheets (Opening image courtesy of David Iberri, http://en.wikipedia.org/wiki/file:camkii.png, and under the Creative Commons license CC-BY-SA 3.0.) Lesson 2.1: Matter
Amino Acid Metabolism (Chapter 20) Lecture 8:
 Amino Acid Metabolism (Chapter 20) Lecture 8: Nitrogen Fixation (20.7); Nitrite Assimilation (not in text?); Protein Digestion in the Gut (5.3b, 11.5, 20.2); Amino Acid Degradation in Cells (20.2); Next:
Amino Acid Metabolism (Chapter 20) Lecture 8: Nitrogen Fixation (20.7); Nitrite Assimilation (not in text?); Protein Digestion in the Gut (5.3b, 11.5, 20.2); Amino Acid Degradation in Cells (20.2); Next:
Chemistry 20 Chapters 15 Enzymes
 Chemistry 20 Chapters 15 Enzymes Enzymes: as a catalyst, an enzyme increases the rate of a reaction by changing the way a reaction takes place, but is itself not changed at the end of the reaction. An
Chemistry 20 Chapters 15 Enzymes Enzymes: as a catalyst, an enzyme increases the rate of a reaction by changing the way a reaction takes place, but is itself not changed at the end of the reaction. An
Gene Regulation -- The Lac Operon
 Gene Regulation -- The Lac Operon Specific proteins are present in different tissues and some appear only at certain times during development. All cells of a higher organism have the full set of genes:
Gene Regulation -- The Lac Operon Specific proteins are present in different tissues and some appear only at certain times during development. All cells of a higher organism have the full set of genes:
Name Date Period. 2. When a molecule of double-stranded DNA undergoes replication, it results in
 DNA, RNA, Protein Synthesis Keystone 1. During the process shown above, the two strands of one DNA molecule are unwound. Then, DNA polymerases add complementary nucleotides to each strand which results
DNA, RNA, Protein Synthesis Keystone 1. During the process shown above, the two strands of one DNA molecule are unwound. Then, DNA polymerases add complementary nucleotides to each strand which results
A disaccharide is formed when a dehydration reaction joins two monosaccharides. This covalent bond is called a glycosidic linkage.
 CH 5 Structure & Function of Large Molecules: Macromolecules Molecules of Life All living things are made up of four classes of large biological molecules: carbohydrates, lipids, proteins, and nucleic
CH 5 Structure & Function of Large Molecules: Macromolecules Molecules of Life All living things are made up of four classes of large biological molecules: carbohydrates, lipids, proteins, and nucleic
Novel physiological functions of oligosaccharides*
 Pure Appl. Chem., Vol. 74, No. 7, pp. 1271 1279, 2002. 2002 IUPAC Novel physiological functions of oligosaccharides* Masao Hirayama Health and Bioscience Laboratories, Meiji Seika Kaisha, Ltd., 5-3-1 Chiyoda,
Pure Appl. Chem., Vol. 74, No. 7, pp. 1271 1279, 2002. 2002 IUPAC Novel physiological functions of oligosaccharides* Masao Hirayama Health and Bioscience Laboratories, Meiji Seika Kaisha, Ltd., 5-3-1 Chiyoda,
Department of Food and Nutrition
 Department of Food and Nutrition Faculties Professors Lee-Kim, Yang Cha, Ph.D. (M.I.T., 1973) Nutritional biochemistry, Antioxidant vitamins, Fatty acid metabolism, Brain development, and Hyperlipidemia
Department of Food and Nutrition Faculties Professors Lee-Kim, Yang Cha, Ph.D. (M.I.T., 1973) Nutritional biochemistry, Antioxidant vitamins, Fatty acid metabolism, Brain development, and Hyperlipidemia
BIOLOGICAL MOLECULES OF LIFE
 BIOLOGICAL MOLECULES OF LIFE C A R B O H Y D R A T E S, L I P I D S, P R O T E I N S, A N D N U C L E I C A C I D S The Academic Support Center @ Daytona State College (Science 115, Page 1 of 29) Carbon
BIOLOGICAL MOLECULES OF LIFE C A R B O H Y D R A T E S, L I P I D S, P R O T E I N S, A N D N U C L E I C A C I D S The Academic Support Center @ Daytona State College (Science 115, Page 1 of 29) Carbon
Nutrients: Carbohydrates, Proteins, and Fats. Chapter 5 Lesson 2
 Nutrients: Carbohydrates, Proteins, and Fats Chapter 5 Lesson 2 Carbohydrates Definition- the starches and sugars found in foods. Carbohydrates are the body s preferred source of energy providing four
Nutrients: Carbohydrates, Proteins, and Fats Chapter 5 Lesson 2 Carbohydrates Definition- the starches and sugars found in foods. Carbohydrates are the body s preferred source of energy providing four
Photo Cell Resp Practice. A. ATP B. oxygen C. DNA D. water. The following equation represents the process of photosynthesis in green plants.
 Name: ate: 1. Which molecule supplies the energy for cellular functions?. TP. oxygen. N. water 2. Photosynthesis The following equation represents the process of photosynthesis in green plants. What happens
Name: ate: 1. Which molecule supplies the energy for cellular functions?. TP. oxygen. N. water 2. Photosynthesis The following equation represents the process of photosynthesis in green plants. What happens
1. A covalent bond between two atoms represents what kind of energy? a. Kinetic energy b. Potential energy c. Mechanical energy d.
 1. A covalent bond between two atoms represents what kind of energy? a. Kinetic energy b. Potential energy c. Mechanical energy d. Solar energy A. Answer a is incorrect. Kinetic energy is the energy of
1. A covalent bond between two atoms represents what kind of energy? a. Kinetic energy b. Potential energy c. Mechanical energy d. Solar energy A. Answer a is incorrect. Kinetic energy is the energy of
The Need for a PARP in vivo Pharmacodynamic Assay
 The Need for a PARP in vivo Pharmacodynamic Assay Jay George, Ph.D., Chief Scientific Officer, Trevigen, Inc., Gaithersburg, MD For further infomation, please contact: William Booth, Ph.D. Tel: +44 (0)1235
The Need for a PARP in vivo Pharmacodynamic Assay Jay George, Ph.D., Chief Scientific Officer, Trevigen, Inc., Gaithersburg, MD For further infomation, please contact: William Booth, Ph.D. Tel: +44 (0)1235
STATE UNIVERSITY OF NEW YORK COLLEGE OF TECHNOLOGY CANTON, NEW YORK COURSE OUTLINE. BIOL 101 Introduction to Biology
 STATE UNIVERSITY OF NEW YORK COLLEGE OF TECHNOLOGY CANTON, NEW YORK COURSE OUTLINE BIOL 101 Introduction to Biology Prepared By: W. David Barnes SCHOOL OF SCIENCE, HEALTH & PROFESSIONAL STUDIES SCIENCE
STATE UNIVERSITY OF NEW YORK COLLEGE OF TECHNOLOGY CANTON, NEW YORK COURSE OUTLINE BIOL 101 Introduction to Biology Prepared By: W. David Barnes SCHOOL OF SCIENCE, HEALTH & PROFESSIONAL STUDIES SCIENCE
Cellular Energy. 1. Photosynthesis is carried out by which of the following?
 Cellular Energy 1. Photosynthesis is carried out by which of the following? A. plants, but not animals B. animals, but not plants C. bacteria, but neither animals nor plants D. all living organisms 2.
Cellular Energy 1. Photosynthesis is carried out by which of the following? A. plants, but not animals B. animals, but not plants C. bacteria, but neither animals nor plants D. all living organisms 2.
Manufacturing process of biologics
 Manufacturing process of biologics K. Ho Afssaps, France 2011 ICH International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use 2011 ICH 1 Disclaimer:
Manufacturing process of biologics K. Ho Afssaps, France 2011 ICH International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use 2011 ICH 1 Disclaimer:
PRESTWICK ACADEMY NATIONAL 5 BIOLOGY CELL BIOLOGY SUMMARY
 Name PRESTWICK ACADEMY NATIONAL 5 BIOLOGY CELL BIOLOGY SUMMARY Cell Structure Identify animal, plant, fungal and bacterial cell ultrastructure and know the structures functions. Plant cell Animal cell
Name PRESTWICK ACADEMY NATIONAL 5 BIOLOGY CELL BIOLOGY SUMMARY Cell Structure Identify animal, plant, fungal and bacterial cell ultrastructure and know the structures functions. Plant cell Animal cell
General Properties Protein Nature of Enzymes Folded Shape of Enzymes H-bonds complementary
 Proteins that function as biological catalysts are called enzymes. Enzymes speed up specific metabolic reactions. Low contamination, low temperature and fast metabolism are only possible with enzymes.
Proteins that function as biological catalysts are called enzymes. Enzymes speed up specific metabolic reactions. Low contamination, low temperature and fast metabolism are only possible with enzymes.
Review of the Cell and Its Organelles
 Biology Learning Centre Review of the Cell and Its Organelles Tips for most effective learning of this material: Memorize the names and structures over several days. This will help you retain what you
Biology Learning Centre Review of the Cell and Its Organelles Tips for most effective learning of this material: Memorize the names and structures over several days. This will help you retain what you
Engineering of Yellow Mosaic Virus Resistance (YMVR) in Blackgram. Project ID: 1 April 2000 to 31 August 2004. Project Duration:
 Engineering of Yellow Mosaic Virus Resistance (YMVR) in Blackgram ID: Duration: Coordinator in Switzerland: PS1 1 April 2000 to 31 August 2004 Prof. Thomas Hohn University of Basel Botanisches Institut
Engineering of Yellow Mosaic Virus Resistance (YMVR) in Blackgram ID: Duration: Coordinator in Switzerland: PS1 1 April 2000 to 31 August 2004 Prof. Thomas Hohn University of Basel Botanisches Institut
BIOMOLECULES. reflect
 reflect A child s building blocks are relatively simple structures. When they come together, however, they can form magnifi cent structures. The elaborate city scene to the right is made of small, simple
reflect A child s building blocks are relatively simple structures. When they come together, however, they can form magnifi cent structures. The elaborate city scene to the right is made of small, simple
Organelles and Their Functions
 Organelles and Their Functions The study of cell organelles and their functions is a fascinating part of biology. The current article provides a brief description of the structure of organelles and their
Organelles and Their Functions The study of cell organelles and their functions is a fascinating part of biology. The current article provides a brief description of the structure of organelles and their
Detailed Course Descriptions for the Human Nutrition Program
 1 Detailed Course Descriptions for the Human Nutrition Program Major Required Courses NUTR221 Principles of Food Science and Nutrition Credit (Contact) Hours 2 CH(2 Theory) Prerequisites Course Description
1 Detailed Course Descriptions for the Human Nutrition Program Major Required Courses NUTR221 Principles of Food Science and Nutrition Credit (Contact) Hours 2 CH(2 Theory) Prerequisites Course Description
III. THE MICROBIAL BIOMASS
 III. THE MICROBIAL BIOMASS Required Readings: Ley, R.E., D.A. Lipson and S.K. Schmidt. 2001. Microbial biomass levels in barren and vegetated high altitude talus soils. Soil Sci. Soc. Am. J. 65:111 117.
III. THE MICROBIAL BIOMASS Required Readings: Ley, R.E., D.A. Lipson and S.K. Schmidt. 2001. Microbial biomass levels in barren and vegetated high altitude talus soils. Soil Sci. Soc. Am. J. 65:111 117.
Cell and Membrane Practice. A. chromosome B. gene C. mitochondrion D. vacuole
 Name: ate: 1. Which structure is outside the nucleus of a cell and contains N?. chromosome. gene. mitochondrion. vacuole 2. potato core was placed in a beaker of water as shown in the figure below. Which
Name: ate: 1. Which structure is outside the nucleus of a cell and contains N?. chromosome. gene. mitochondrion. vacuole 2. potato core was placed in a beaker of water as shown in the figure below. Which
Chapter 8: An Introduction to Metabolism
 Chapter 8: An Introduction to Metabolism Name Period Concept 8.1 An organism s metabolism transforms matter and energy, subject to the laws of thermodynamics 1. Define metabolism. The totality of an organism
Chapter 8: An Introduction to Metabolism Name Period Concept 8.1 An organism s metabolism transforms matter and energy, subject to the laws of thermodynamics 1. Define metabolism. The totality of an organism
ENZYMES. Serine Proteases Chymotrypsin, Trypsin, Elastase, Subtisisin. Principle of Enzyme Catalysis
 ENZYMES Serine Proteases Chymotrypsin, Trypsin, Elastase, Subtisisin Principle of Enzyme Catalysis Linus Pauling (1946) formulated the first basic principle of enzyme catalysis Enzyme increase the rate
ENZYMES Serine Proteases Chymotrypsin, Trypsin, Elastase, Subtisisin Principle of Enzyme Catalysis Linus Pauling (1946) formulated the first basic principle of enzyme catalysis Enzyme increase the rate
PHOTOSYNTHESIS AND CELLULAR RESPIRATION
 reflect Wind turbines shown in the photo on the right are large structures with blades that move in response to air movement. When the wind blows, the blades rotate. This motion generates energy that is
reflect Wind turbines shown in the photo on the right are large structures with blades that move in response to air movement. When the wind blows, the blades rotate. This motion generates energy that is
Chapter 2: Cell Structure and Function pg. 70-107
 UNIT 1: Biochemistry Chapter 2: Cell Structure and Function pg. 70-107 Organelles are internal structures that carry out specialized functions, interacting and complementing each other. Animal and plant
UNIT 1: Biochemistry Chapter 2: Cell Structure and Function pg. 70-107 Organelles are internal structures that carry out specialized functions, interacting and complementing each other. Animal and plant
Functional properties of fructans and their potential role in management of intestinal inflammatory pathologies
 Functional properties of fructans and their potential role in management of intestinal inflammatory pathologies Dr.ssa Valentina Pasqualetti Unità di Ricerca di Scienze degli Alimenti e della Nutrizione
Functional properties of fructans and their potential role in management of intestinal inflammatory pathologies Dr.ssa Valentina Pasqualetti Unità di Ricerca di Scienze degli Alimenti e della Nutrizione
AP Bio Photosynthesis & Respiration
 AP Bio Photosynthesis & Respiration Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 1. What is the term used for the metabolic pathway in which
AP Bio Photosynthesis & Respiration Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 1. What is the term used for the metabolic pathway in which
Chapter 6 DNA Replication
 Chapter 6 DNA Replication Each strand of the DNA double helix contains a sequence of nucleotides that is exactly complementary to the nucleotide sequence of its partner strand. Each strand can therefore
Chapter 6 DNA Replication Each strand of the DNA double helix contains a sequence of nucleotides that is exactly complementary to the nucleotide sequence of its partner strand. Each strand can therefore
Fields of Application / Industry:
 = Fields of Application / Industry: Chemistry / Polymer Industry Clinical Chemistry / Medicine / Hygiene / Health Care Cosmetics Electronics Energy Environment / Water / Waste Food / Agriculture Geology
= Fields of Application / Industry: Chemistry / Polymer Industry Clinical Chemistry / Medicine / Hygiene / Health Care Cosmetics Electronics Energy Environment / Water / Waste Food / Agriculture Geology
The Cell Teaching Notes and Answer Keys
 The Cell Teaching Notes and Answer Keys Subject area: Science / Biology Topic focus: The Cell: components, types of cells, organelles, levels of organization Learning Aims: describe similarities and differences
The Cell Teaching Notes and Answer Keys Subject area: Science / Biology Topic focus: The Cell: components, types of cells, organelles, levels of organization Learning Aims: describe similarities and differences
BIOVALUE PROJECT 2 ANDERS PETER S. ADAMSEN AARHUS UNIVERSITY HEALTH 04 JUNE 2015
 BIOVALUE PROJECT 2 ANDERS PETER ADAMSEN CONTENT Protein work KU Effekt of time lag between harvest and processing Flow sheeting Pilot plant OLIGOSACCHARIDES - A Upgrading of C5 oligomers Structural composition
BIOVALUE PROJECT 2 ANDERS PETER ADAMSEN CONTENT Protein work KU Effekt of time lag between harvest and processing Flow sheeting Pilot plant OLIGOSACCHARIDES - A Upgrading of C5 oligomers Structural composition
Chemical Basis of Life Module A Anchor 2
 Chemical Basis of Life Module A Anchor 2 Key Concepts: - Water is a polar molecule. Therefore, it is able to form multiple hydrogen bonds, which account for many of its special properties. - Water s polarity
Chemical Basis of Life Module A Anchor 2 Key Concepts: - Water is a polar molecule. Therefore, it is able to form multiple hydrogen bonds, which account for many of its special properties. - Water s polarity
A Review of Factors Affecting Carbohydrate Levels in Forage. Kathryn A. Watts, B.S., N. Jerry Chatterton, Ph. D.
 Kathryn A. Watts, B.S., N. Jerry Chatterton, Ph. D. Take home message: Carbohydrate concentration of forages should be taken into account when formulating rations for carbohydrate intolerant equines. Carbohydrate
Kathryn A. Watts, B.S., N. Jerry Chatterton, Ph. D. Take home message: Carbohydrate concentration of forages should be taken into account when formulating rations for carbohydrate intolerant equines. Carbohydrate
