To Determine which Antacid could neutralize Stomach acid the most
|
|
|
- Bruno Knight
- 7 years ago
- Views:
Transcription
1 To Determine which Antacid could neutralize Stomach acid the most
2 CONTENTS Objective Introduction Acids Stomach Acid Some foods containing acids Antacid Action mechanism Indication Side Effects Problems with reduced stomach acidity Experiment Design Material Required Procedure Observation And Calculation Result Precautions OBJECTIVE The purpose of this experiment was to determine which antacid could neutralize the most stomach acid. I became interested in this idea when I saw some experiments on medicines and wanted to find out some scientific facts about medicines. The information gained from this experiment will help people know which antacid they should look for in the stores. It will also let them know which antacid will give them the most comfort. This could also save consumers money and provide better health. INTRODUCTION Digestion in the stomach results from the action of gastric fluid, which includes secretions of digestive enzymes, mucous, and hydrochloric acid. The acidic environment of the stomach makes it possible for inactive forms of digestive enzymes to be converted into active forms (i.e. pepsinogen into pepsin), and acid is also needed to dissolve minerals and kill bacteria that may enter the stomach along with food. However, excessive acid production (hyperacidity) results in the unpleasant symptoms of heartburn and may contribute to ulcer formation in the stomach lining. Antacids are weak bases (most commonly bicarbonates, hydroxides, and carbonates) that neutralize excess stomach acid and thus alleviate symptoms of heartburn. The general neutralization reaction is: Antacid (weak base) + HCl (stomach acid) salts + H 2 O + CO 2 The hydrochloric acid solution used in this experiment (0.1 M) approximates the acid conditions of the human stomach, which is typically 0.4 to 0.5% HCl by mass (ph ~ 1).Antacids help people who have or get heartburn. The following information will help people understand how stomach acid works and what antacid will help those most.
3 ACIDS Acids are a group of chemicals, usually in liquid form. They can be recognized by their sour taste and their ability to react with other substances. Acids are confirmed as an acid by their ph. The ph of acids ranges from (below 7). The two main acids are: mineral acid and organic acid. The three well known acids that are sulphuric acid (H2SO4), nitric acid (HNO3), and hydrochloric acid (HCl). ANTACID An antacid is any substance that can neutralize an acid. All antacids are bases. A base is any substance that can neutralize an acid. The ph of a base is (above 7). All antacids have chemical in them called a buffer. When an antacid is mixed with an acid the buffer tries to even out the acidity and that is how stomach acid gets neutralized. In an antacid it is not the name brand that tells how well it works it is something called an active ingredient. Not all antacids have a different active ingredient. Some have one of the same active ingredients and some have all of the same active ingredients. Almost all the antacids that have the same active ingredient work the same amount as the other. The active ingredient of most of the antacids is bases of calcium, magnesium, aluminium. ACTION MECHANISM Antacids perform neutralization reaction, i.e. they buffer gastric acid, raising the ph to reduce acidity in the stomach. When gastric hydrochloric acid reaches the nerves in the gastrointestinal mucosa, they signal pain to the central nervous system. This happens when these nerves are exposed, as in peptic ulcers. The gastric acid may also reach ulcers in the esophagus or the duodenum. Other mechanisms may contribute, such as the effect of aluminium ions inhibiting smooth muscle cell contraction and delaying gastric emptying. Antacids are commonly used to help neutralize stomach acid. Antacids are bases with a ph above 7.0 that chemically react with acids to neutralize them. The action of antacids is based on the fact that a base reacts with acid to form salt and water. INDICATIONS Antacids are taken by mouth to relieve heartburn, the major symptom of gastro esophageal reflux disease, or acid indigestion. Treatment with antacids alone is symptotic and only justified for minor symptoms. Peptic ulcers may require H2- receptor antagonists or proton pump inhibitors. The usefulness of many combinations of antacids is not clear, although the combination of magnesium and aluminium salts may prevent alteration of bowel habits. SIDE EFFECTS Aluminium hydroxide: may lead to the formation of insoluble aluminium phosphate complexes, with a risk for hypophosphate and osteomalacia. Although aluminium has a low gastrointestinal absorption, accumulation may occur in the presence of renal insufficiency. Aluminium containing drugs may cause constipation. Magnesium hydroxide: has a laxative property. Magnesium may accumulate in patients with renal failure leading to hypo magnesia, with cardiovascular and neurological complications. Calcium: compounds containing calcium may increase calcium output in the urine, which might be associated to renal stones. Calcium salts may cause constipation.
4 Carbonate: regular high doses may cause alkalosis, which in turn may result in altered excretion of other drugs, and kidney stones. PROBLEMS WITH REDUCED STOMACH ACIDITY Reduced stomach acidity may result in an impaired ability to digest and absorb certain nutrients, such as iron and the B vitamins. Since the low Ph of the stomach normally kills ingested bacteria, antacids increase the vulnerability to infection. It could also result in the reduced bioavailability of some drugs. For example, the bioavailability of ketocanazole (antifungal), is reduced at high intragastric ph (low acid content). EXPERIMENT The constants in this study were: - Type of acid - Consistency of procedures The variables in the study were: -Different types of antacid used The responding variable was: -The amount of stomach acid each antacid could neutralize measured in ml MATERIAL REQUIRED Burette, pipette, titration flask, measuring flask, beaker, weighing machine, concentrated sulphuric acid, methyl orange, antacid samples. PROCEDURE - Prepare half litre of N/10 HCl solution by diluting 10 ml of the concentrated acid to 1 litre. - Prepare N/10 sodium carbonate solution by weighing exactly g of anhydrous sodium carbonate and then dissolving it in water to prepare exactly 0.25 litre of solution. - Standardize the HCl solution by titrating it against the standard sodium carbonate solution using methyl orange as indicator. - Take 20 ml of standardized HCl in the conical flask, use methyl orange as indicator and see the amount of base used for neutralization. - Powder the various sample of antacids tablets and weigh 10 mg of each. - Take 20 ml of standardized HCl solution in the conical flask; add the weighed samples to it. - Add two drops of methyl orange and warm the flask till most of the powder dissolves. Filter off the insoluble material. - Titrate the solution against the standardized Na2CO3 solution till a permanent red tinge appears. permanent red tinge appears. - Note the amount of base used for titration and note the reduction in the amount of base used. - Repeat the experiment with different antacids.
5 OBSERVATIONS AND CALCULATIONS 1. Standardization of HCl solution Concordant reading=15.0 ml Applying normality equation N1V1(acid) = N2V2(base) N1 (15) = (1/10) 20 Normality of HCl solution, N1 = N 2. Neutralization of standardized HCl solution used Volume of HCl taken= 20.0ml 3. Analysis of antacid tablets Weight of the antacid tablet powder= 10 mg Volume of HCl solution added= 20.0 ml RESULT The most effective antacid out of the taken samples is acid 10.
6 PRECAUTIONS - All apparatus should be clean and washed properly. - Burette and pipette must be rinsed with the respective solution to - be put in them. - Air bubbles must be removed from the burette and jet. - Last drop from the pipette should not be removed by blowing. - The flask should not be rinsed with any of the solution, which are being titrated.
7 This document was created with Win2PDF available at The unregistered version of Win2PDF is for evaluation or non-commercial use only. This page will not be added after purchasing Win2PDF.
Syllabus OC18 Use litmus or a universal indicator to test a variety of solutions, and classify these as acidic, basic or neutral
 Chemistry: 9. Acids and Bases Please remember to photocopy 4 pages onto one sheet by going A3 A4 and using back to back on the photocopier Syllabus OC18 Use litmus or a universal indicator to test a variety
Chemistry: 9. Acids and Bases Please remember to photocopy 4 pages onto one sheet by going A3 A4 and using back to back on the photocopier Syllabus OC18 Use litmus or a universal indicator to test a variety
Experiment 7: Titration of an Antacid
 1 Experiment 7: Titration of an Antacid Objective: In this experiment, you will standardize a solution of base using the analytical technique known as titration. Using this standardized solution, you will
1 Experiment 7: Titration of an Antacid Objective: In this experiment, you will standardize a solution of base using the analytical technique known as titration. Using this standardized solution, you will
Dissolving of sodium hydroxide generates heat. Take care in handling the dilution container.
 TITRATION: STANDARDIZATION OF A BASE AND ANALYSIS OF STOMACH ANTACID TABLETS 2009, 1996, 1973 by David A. Katz. All rights reserved. Reproduction permitted for education use provided original copyright
TITRATION: STANDARDIZATION OF A BASE AND ANALYSIS OF STOMACH ANTACID TABLETS 2009, 1996, 1973 by David A. Katz. All rights reserved. Reproduction permitted for education use provided original copyright
Digestive System (continued) Digestive System. Stomach. Peptic Ulcer Disease
 Digestive System Digestive System (continued) Responsible for breaking down food, absorbing nutrients, eliminating wastes Alimentary canal Also known as gastrointestinal tract Reaches from mouth to anus
Digestive System Digestive System (continued) Responsible for breaking down food, absorbing nutrients, eliminating wastes Alimentary canal Also known as gastrointestinal tract Reaches from mouth to anus
Digestion, Absorption. How & where?
 Digestion, Absorption How & where? What happens to food? Three processes Digestion Absorption Elimination Where do they occur? GI tract Overview of Digestion GI tract Gastrointestinal (GI) tract: series
Digestion, Absorption How & where? What happens to food? Three processes Digestion Absorption Elimination Where do they occur? GI tract Overview of Digestion GI tract Gastrointestinal (GI) tract: series
PART I: PREPARATION OF SOLUTIONS AND STANDARDIZATION OF A BASE
 TITRATION: STANDARDIZATION OF A BASE AND ANALYSIS OF STOMACH ANTACID TABLETS 2009, 1996, 1973 by David A. Katz. All rights reserved. Reproduction permitted for education use provided original copyright
TITRATION: STANDARDIZATION OF A BASE AND ANALYSIS OF STOMACH ANTACID TABLETS 2009, 1996, 1973 by David A. Katz. All rights reserved. Reproduction permitted for education use provided original copyright
EXPERIMENT 10: TITRATION AND STANDARDIZATION
 EXPERIMENT 10: TITRATION AND STANDARDIZATION PURPOSE To determine the molarity of a NaOH solution by titrating it with a standard HCl solution. To determine the molarity of acetic acid in vinegar using
EXPERIMENT 10: TITRATION AND STANDARDIZATION PURPOSE To determine the molarity of a NaOH solution by titrating it with a standard HCl solution. To determine the molarity of acetic acid in vinegar using
Properties of Acids and Bases
 Lab 22 Properties of Acids and Bases TN Standard 4.2: The student will investigate the characteristics of acids and bases. Have you ever brushed your teeth and then drank a glass of orange juice? What
Lab 22 Properties of Acids and Bases TN Standard 4.2: The student will investigate the characteristics of acids and bases. Have you ever brushed your teeth and then drank a glass of orange juice? What
ACIDS AND BASES SAFETY PRECAUTIONS
 ACIDS AND BASES Mild acids and bases are used in cooking (their reaction makes biscuits and bread rise). Acids such as those in our stomachs eat away at food or digest it. Strong acids and bases are used
ACIDS AND BASES Mild acids and bases are used in cooking (their reaction makes biscuits and bread rise). Acids such as those in our stomachs eat away at food or digest it. Strong acids and bases are used
Experiment 16-Acids, Bases and ph
 Definitions acid-an ionic compound that releases or reacts with water to form hydrogen ion (H + ) in aqueous solution. They taste sour and turn litmus red. Acids react with certain metals such as zinc,
Definitions acid-an ionic compound that releases or reacts with water to form hydrogen ion (H + ) in aqueous solution. They taste sour and turn litmus red. Acids react with certain metals such as zinc,
DRUG TESTING The Analysis of Some Over-the-Counter Drugs 2004 by David A. Katz. All rights reserved.
 DRUG TESTING The Analysis of Some ver-the-counter Drugs 2004 by David A. Katz. All rights reserved. A major part of forensic testing is the identification of drugs, both licit and illicit. Due to restrictions
DRUG TESTING The Analysis of Some ver-the-counter Drugs 2004 by David A. Katz. All rights reserved. A major part of forensic testing is the identification of drugs, both licit and illicit. Due to restrictions
5.0 EXPERIMENT ON DETERMINATION OF TOTAL HARDNESS
 5.0 EXPERIMENT ON DETERMINATION OF TOTAL HARDNESS Sl. No. Contents Preamble 5.1 Aim 5.2 Introduction 5.2.1 Environmental Significance 5.3 Principle 5.4 Materials Required 5.4.1 Apparatus Required 5.4.2
5.0 EXPERIMENT ON DETERMINATION OF TOTAL HARDNESS Sl. No. Contents Preamble 5.1 Aim 5.2 Introduction 5.2.1 Environmental Significance 5.3 Principle 5.4 Materials Required 5.4.1 Apparatus Required 5.4.2
GI TRACT ORGANS ACCESSORY ORGANS
 Digestive System GI TRACT ORGANS Oral cavity Oropharynx Esophagus Stomach Small intestine Large Intestine Anus ACCESSORY ORGANS Salivary glands Pancreas Liver Gall bladder GI TRACT LAYERS Mucosa Submucosa
Digestive System GI TRACT ORGANS Oral cavity Oropharynx Esophagus Stomach Small intestine Large Intestine Anus ACCESSORY ORGANS Salivary glands Pancreas Liver Gall bladder GI TRACT LAYERS Mucosa Submucosa
Digestion, Absorption. How & where?
 Digestion, Absorption How & where? What happens to food? Three processes Digestion Absorption Elimination Where do they occur? GI tract Overview of Digestion GI tract Gastrointestinal (GI) tract: series
Digestion, Absorption How & where? What happens to food? Three processes Digestion Absorption Elimination Where do they occur? GI tract Overview of Digestion GI tract Gastrointestinal (GI) tract: series
Chemistry 52. Reacts with active metals to produce hydrogen gas. Have a slippery, soapy feeling. React with carbonates to produce CO 2
 ACID AND BASE STRENGTH Experiment #2 PURPOSE: 1. To distinguish between acids, bases and neutral substances, by observing their effect on some common indicators. 2. To distinguish between strong and weak
ACID AND BASE STRENGTH Experiment #2 PURPOSE: 1. To distinguish between acids, bases and neutral substances, by observing their effect on some common indicators. 2. To distinguish between strong and weak
ACID-BASE TITRATIONS: DETERMINATION OF CARBONATE BY TITRATION WITH HYDROCHLORIC ACID BACKGROUND
 #3. Acid - Base Titrations 27 EXPERIMENT 3. ACID-BASE TITRATIONS: DETERMINATION OF CARBONATE BY TITRATION WITH HYDROCHLORIC ACID BACKGROUND Carbonate Equilibria In this experiment a solution of hydrochloric
#3. Acid - Base Titrations 27 EXPERIMENT 3. ACID-BASE TITRATIONS: DETERMINATION OF CARBONATE BY TITRATION WITH HYDROCHLORIC ACID BACKGROUND Carbonate Equilibria In this experiment a solution of hydrochloric
Neutralizing an Acid and a Base
 Balancing Act Teacher Information Objectives In this activity, students neutralize a base with an acid. Students determine the point of neutralization of an acid mixed with a base while they: Recognize
Balancing Act Teacher Information Objectives In this activity, students neutralize a base with an acid. Students determine the point of neutralization of an acid mixed with a base while they: Recognize
The Determination of Acid Content in Vinegar
 The Determination of Acid Content in Vinegar Reading assignment: Chang, Chemistry 10 th edition, pages 153-156. Goals We will use a titration to determine the concentration of acetic acid in a sample of
The Determination of Acid Content in Vinegar Reading assignment: Chang, Chemistry 10 th edition, pages 153-156. Goals We will use a titration to determine the concentration of acetic acid in a sample of
4.0 EXPERIMENT ON DETERMINATION OF CHLORIDES
 4.0 EXPERIMENT ON DETERMINATION OF CHLORIDES Sl. No. Contents Preamble 4.1 Aim 4.2 Introduction 4.2.1 Environmental Significance 4.3 Principle 4.4 Materials Required 4.4.1 Apparatus Required 4.4.2 Chemicals
4.0 EXPERIMENT ON DETERMINATION OF CHLORIDES Sl. No. Contents Preamble 4.1 Aim 4.2 Introduction 4.2.1 Environmental Significance 4.3 Principle 4.4 Materials Required 4.4.1 Apparatus Required 4.4.2 Chemicals
Chapter 17 Digestive System. Alimentary Canal. Movements of the Tube
 Chapter 17 Digestive System Functions of Digestive System ingestion mechanical digestion chemical digestion propulsion absorption defecation Consists of the alimentary canal and accessory organs 1 Alimentary
Chapter 17 Digestive System Functions of Digestive System ingestion mechanical digestion chemical digestion propulsion absorption defecation Consists of the alimentary canal and accessory organs 1 Alimentary
10.2 The Human Digestive System pg. 411
 10.2 The Human Digestive System pg. 411 The human digestive system is made up of a group of organs working together. The digestive tract is made up of the mouth, esophagus, stomach, small intestine, and
10.2 The Human Digestive System pg. 411 The human digestive system is made up of a group of organs working together. The digestive tract is made up of the mouth, esophagus, stomach, small intestine, and
The Excretory and Digestive Systems
 The Excretory and Digestive Systems 38.2 The Process of Digestion Organs of the Digestive System The digestive system includes the: Mouth Pharynx Esophagus Stomach Small and large intestine. Other structures
The Excretory and Digestive Systems 38.2 The Process of Digestion Organs of the Digestive System The digestive system includes the: Mouth Pharynx Esophagus Stomach Small and large intestine. Other structures
Chemistry Unit Test Review
 SNC 2DI Chemistry Unit Test Review Note: this review sheet only covers the second half of our chemistry unit. You should use our mid-unit test review sheet to recall concepts from the first half of the
SNC 2DI Chemistry Unit Test Review Note: this review sheet only covers the second half of our chemistry unit. You should use our mid-unit test review sheet to recall concepts from the first half of the
PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement)
 1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
Acid 7 Base. 1. Describe two things hydrochloric acid does in your body system. 2. What does sodium hydrogencarbonate do in your body system?
 Acids and Bases acid: a compound that, when dissolved in water, forms a solution with a ph less than 7 base: a compound that, when dissolved in water, forms a solution with a ph greater than 7 ph: the
Acids and Bases acid: a compound that, when dissolved in water, forms a solution with a ph less than 7 base: a compound that, when dissolved in water, forms a solution with a ph greater than 7 ph: the
ACIDS, BASES AND SALTS
 Acids are those chemical substances which have a sour taste. Bases are those chemical substances which have a bitter taste. A salt is an ionic compound which dissociates to yield a positive ion other than
Acids are those chemical substances which have a sour taste. Bases are those chemical substances which have a bitter taste. A salt is an ionic compound which dissociates to yield a positive ion other than
Acids, Bases, and ph
 CHAPTER 9 1 SECTION Acids, Bases, and Salts Acids, Bases, and ph KEY IDEAS As you read this section, keep these questions in mind: What properties do acids have? What properties do bases have? How can
CHAPTER 9 1 SECTION Acids, Bases, and Salts Acids, Bases, and ph KEY IDEAS As you read this section, keep these questions in mind: What properties do acids have? What properties do bases have? How can
Medical Nutrition Therapy for Upper Gastrointestinal Tract Disorders. By: Jalal Hejazi PhD, MSc.
 Medical Nutrition Therapy for Upper Gastrointestinal Tract Disorders By: Jalal Hejazi PhD, MSc. Digestive Disorders Common problem; more than 50 million outpatient visits per year Dietary habits and nutrition
Medical Nutrition Therapy for Upper Gastrointestinal Tract Disorders By: Jalal Hejazi PhD, MSc. Digestive Disorders Common problem; more than 50 million outpatient visits per year Dietary habits and nutrition
Determination of the amount of sodium carbonate and sodium hydroxide in a mixture by titration.
 Module 9 : Experiments in Chemistry Lecture 38 : Titrations : Acid-Base, Redox and Complexometric Objectives In this lecture you will learn the techniques to do following Determination of the amount of
Module 9 : Experiments in Chemistry Lecture 38 : Titrations : Acid-Base, Redox and Complexometric Objectives In this lecture you will learn the techniques to do following Determination of the amount of
Determination of the Amount of Acid Neutralized by an Antacid Tablet Using Back Titration
 Determination of the Amount of Acid Neutralized by an Antacid Tablet Using Back Titration GOAL AND OVERVIEW Antacids are bases that react stoichiometrically with acid. The number of moles of acid that
Determination of the Amount of Acid Neutralized by an Antacid Tablet Using Back Titration GOAL AND OVERVIEW Antacids are bases that react stoichiometrically with acid. The number of moles of acid that
THE DIGESTIVE SYSTEM
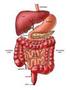 THE DIGESTIVE SYSTEM What is digestion? Digestion is the process of breaking down food so that it's small enough to be absorbed and used by the body for energy or in other bodily functions. Digestion involves
THE DIGESTIVE SYSTEM What is digestion? Digestion is the process of breaking down food so that it's small enough to be absorbed and used by the body for energy or in other bodily functions. Digestion involves
Standardization of Potassium Permanganate solution with Standard Sodium Oxalate Solution.
 Experiment Number: 07 Name of the experiment: Standardization of Potassium Permanganate solution with Standard Sodium Oxalate Solution. Course: Chem-114 Name: Noor Nashid Islam Roll: 0105044 Group: A2
Experiment Number: 07 Name of the experiment: Standardization of Potassium Permanganate solution with Standard Sodium Oxalate Solution. Course: Chem-114 Name: Noor Nashid Islam Roll: 0105044 Group: A2
Chapter 48. Nutrients in Food. Carbohydrates, Proteins, and Lipids. Carbohydrates, Proteins, and Lipids, continued
 Carbohydrates, Proteins, and Lipids The three nutrients needed by the body in the greatest amounts are carbohydrates, proteins, and lipids. Nutrients in Food All of these nutrients are called organic compounds,
Carbohydrates, Proteins, and Lipids The three nutrients needed by the body in the greatest amounts are carbohydrates, proteins, and lipids. Nutrients in Food All of these nutrients are called organic compounds,
Properties of Acids and Bases
 Properties of Acids and Bases (Adapted from Flinn Scientific Acid Base Test Kit I #AP4567) Introduction Battery acid, stomach acid, acid rain just a few acids in our everyday life! What does it mean when
Properties of Acids and Bases (Adapted from Flinn Scientific Acid Base Test Kit I #AP4567) Introduction Battery acid, stomach acid, acid rain just a few acids in our everyday life! What does it mean when
ANALYSIS OF VITAMIN C
 Purpose To learn how to analyze food for vitamin C content and to examine various sources for vitamin C content. Caution Handle the glassware with caution to prevent breakage. When using a burner in the
Purpose To learn how to analyze food for vitamin C content and to examine various sources for vitamin C content. Caution Handle the glassware with caution to prevent breakage. When using a burner in the
Long Term Use of Antacid Medications Can Cause an Increased Risk for Osteoporosis and Much More
 Long Term Use of Antacid Medications Can Cause an Increased Risk for Osteoporosis and Much More By: Jeremie Pederson D.C., C.S.C.S. Many people are concerned about the FDA news release dated May 25, 2010
Long Term Use of Antacid Medications Can Cause an Increased Risk for Osteoporosis and Much More By: Jeremie Pederson D.C., C.S.C.S. Many people are concerned about the FDA news release dated May 25, 2010
Estimation of Hardness of Water by EDTA Method
 Estimation of Hardness of Water by EDTA Method 1 EXPERIMENT 1 Estimation of Hardness of Water by EDTA Method INTRODUCTION Water hardness is the traditional measure of the capacity of water to precipitate
Estimation of Hardness of Water by EDTA Method 1 EXPERIMENT 1 Estimation of Hardness of Water by EDTA Method INTRODUCTION Water hardness is the traditional measure of the capacity of water to precipitate
Preparation of frequently used solutions
 Preparation of frequently used solutions Content 1. Diluting Concentrated Acids (Last Login: 08/08/2009) 2. Indicators (Last Login: 27/07/2009) 3. Standard Buffer Solutions (Last Login: 27/07/2009) 4.
Preparation of frequently used solutions Content 1. Diluting Concentrated Acids (Last Login: 08/08/2009) 2. Indicators (Last Login: 27/07/2009) 3. Standard Buffer Solutions (Last Login: 27/07/2009) 4.
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 CHE 1400 - Spring 2015 - Chapter 7 Homework 7 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1)According to Arrhenius, an acid and a base will react
CHE 1400 - Spring 2015 - Chapter 7 Homework 7 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1)According to Arrhenius, an acid and a base will react
Ascorbic Acid Titration of Vitamin C Tablets This lab will be completed individually! Make sure you come prepared!
 Ascorbic Acid Titration of Vitamin C Tablets This lab will be completed individually! Make sure you come prepared! Introduction Vitamin C (also known as ascorbic acid, HC6H7O6) is a necessary ingredient
Ascorbic Acid Titration of Vitamin C Tablets This lab will be completed individually! Make sure you come prepared! Introduction Vitamin C (also known as ascorbic acid, HC6H7O6) is a necessary ingredient
5 ACIDS, BASES AND SALTS
 5 ACIDS, BASES AND SALTS TEXTBOOK QUESTIONS AND ANSWERS Q.1. Taste the following substances and enter the result in the following table : Substance Taste (Sour / bitter / any other) Lemon juice Orange
5 ACIDS, BASES AND SALTS TEXTBOOK QUESTIONS AND ANSWERS Q.1. Taste the following substances and enter the result in the following table : Substance Taste (Sour / bitter / any other) Lemon juice Orange
Experiment 8 - Double Displacement Reactions
 Experiment 8 - Double Displacement Reactions A double displacement reaction involves two ionic compounds that are dissolved in water. In a double displacement reaction, it appears as though the ions are
Experiment 8 - Double Displacement Reactions A double displacement reaction involves two ionic compounds that are dissolved in water. In a double displacement reaction, it appears as though the ions are
CHM1 Review for Exam 12
 Topics Solutions 1. Arrhenius Acids and bases a. An acid increases the H + concentration in b. A base increases the OH - concentration in 2. Strong acids and bases completely dissociate 3. Weak acids and
Topics Solutions 1. Arrhenius Acids and bases a. An acid increases the H + concentration in b. A base increases the OH - concentration in 2. Strong acids and bases completely dissociate 3. Weak acids and
Lab 25. Acid-Base Titration and Neutralization Reactions: What Is the Concentration of Acetic Acid in Each Sample of Vinegar?
 Lab 25. Acid-Base Titration and Neutralization Reactions: What Is the Concentration of Acetic Acid in Each Sample of Vinegar? Introduction Vinegar is basically a solution of acetic acid (CH3COOH). It is
Lab 25. Acid-Base Titration and Neutralization Reactions: What Is the Concentration of Acetic Acid in Each Sample of Vinegar? Introduction Vinegar is basically a solution of acetic acid (CH3COOH). It is
OXIDATION-REDUCTION TITRATIONS-Permanganometry
 Experiment No. Date OXIDATION-REDUCTION TITRATIONS-Permanganometry INTRODUCTION Potassium permanganate, KMnO 4, is probably the most widely used of all volumetric oxidizing agents. It is a powerful oxidant
Experiment No. Date OXIDATION-REDUCTION TITRATIONS-Permanganometry INTRODUCTION Potassium permanganate, KMnO 4, is probably the most widely used of all volumetric oxidizing agents. It is a powerful oxidant
Upper Gastrointestinal Tract KNH 406
 Upper Gastrointestinal Tract KNH 406 Upper GI A&P GI tract long tube ~ 15 ft. Upper GI mouth, pharynx, esophagus, stomach Accessory organs pancreas, biliary system, liver Four basic functions: motility,
Upper Gastrointestinal Tract KNH 406 Upper GI A&P GI tract long tube ~ 15 ft. Upper GI mouth, pharynx, esophagus, stomach Accessory organs pancreas, biliary system, liver Four basic functions: motility,
Digestive system Review
 Digestive system Review 1. Distinguish between chemical digestion and mechanical digestion. The physical breakdown of food begins in the mouth with two types of processes. The mouth is a complex structure
Digestive system Review 1. Distinguish between chemical digestion and mechanical digestion. The physical breakdown of food begins in the mouth with two types of processes. The mouth is a complex structure
Nomenclature and Household Items
 Nomenclature and Household Items NAME: DATE: PERIOD: Background: Many household items contain chemicals that are commonly used in everyday life. Some chemicals are edible while other compounds would be
Nomenclature and Household Items NAME: DATE: PERIOD: Background: Many household items contain chemicals that are commonly used in everyday life. Some chemicals are edible while other compounds would be
Calcium Analysis by EDTA Titration
 Calcium Analysis by EDTA Titration ne of the factors that establish the quality of a water supply is its degree of hardness. The hardness of water is defined in terms of its content of calcium and magnesium
Calcium Analysis by EDTA Titration ne of the factors that establish the quality of a water supply is its degree of hardness. The hardness of water is defined in terms of its content of calcium and magnesium
Chapter 17. How are acids different from bases? Acid Physical properties. Base. Explaining the difference in properties of acids and bases
 Chapter 17 Acids and Bases How are acids different from bases? Acid Physical properties Base Physical properties Tastes sour Tastes bitter Feels slippery or slimy Chemical properties Chemical properties
Chapter 17 Acids and Bases How are acids different from bases? Acid Physical properties Base Physical properties Tastes sour Tastes bitter Feels slippery or slimy Chemical properties Chemical properties
Acetic Acid Content of Vinegar: An Acid-Base Titration E10-1
 Experiment 10 Acetic Acid Content of Vinegar: An Acid-Base Titration E10-1 E10-2 The task The goal of this experiment is to determine accurately the concentration of acetic acid in vinegar via volumetric
Experiment 10 Acetic Acid Content of Vinegar: An Acid-Base Titration E10-1 E10-2 The task The goal of this experiment is to determine accurately the concentration of acetic acid in vinegar via volumetric
Acid-Base Titrations. Setup for a Typical Titration. Titration 1
 Titration 1 Acid-Base Titrations Molarities of acidic and basic solutions can be used to convert back and forth between moles of solutes and volumes of their solutions, but how are the molarities of these
Titration 1 Acid-Base Titrations Molarities of acidic and basic solutions can be used to convert back and forth between moles of solutes and volumes of their solutions, but how are the molarities of these
Acids, Bases and Salts
 Acids, Bases and Salts 2 HAPTER Tips and Tricks Acids are sour in taste and bases are bitter in taste. Indicators are chemical substances which give different colours in acidic and basic solutions. If
Acids, Bases and Salts 2 HAPTER Tips and Tricks Acids are sour in taste and bases are bitter in taste. Indicators are chemical substances which give different colours in acidic and basic solutions. If
QUESTION (2012:3) (a) (i) Complete the table below showing the conjugate acids and bases. CO 3 H 2 O OH HCN CN -
 QUESTION (2012:3) (i) Complete the table below showing the conjugate acids and bases. Conjugate acid Conjugate base - HCO 3 2 CO 3 H 2 O OH HCN CN - (ii) HPO 4 2 (aq) Write equations for the reactions
QUESTION (2012:3) (i) Complete the table below showing the conjugate acids and bases. Conjugate acid Conjugate base - HCO 3 2 CO 3 H 2 O OH HCN CN - (ii) HPO 4 2 (aq) Write equations for the reactions
PHARMACOLOGICAL PROPERTIES
 ANTİ-FOSFAT ca 700 mg Film Tablet COMPOSITION 1 film tablet contains 700 mg Calcium acetate, as an active ingredient. Excipients are Microcrystalline cellulose, Povidone K30, Crosspovidone, Magnesium stearate,
ANTİ-FOSFAT ca 700 mg Film Tablet COMPOSITION 1 film tablet contains 700 mg Calcium acetate, as an active ingredient. Excipients are Microcrystalline cellulose, Povidone K30, Crosspovidone, Magnesium stearate,
Digestive System Notes
 Digestive System Notes Structure Function Relation Mouth cavity Mechanical digestion by teeth; chemical digestion of starch by saliva. Salivary glands Three pairs of glands which secrete saliva containing
Digestive System Notes Structure Function Relation Mouth cavity Mechanical digestion by teeth; chemical digestion of starch by saliva. Salivary glands Three pairs of glands which secrete saliva containing
Letter to the Student... 5 Test-Taking Checklist... 6 Next Generation Sunshine State Standards Correlation Chart... 7
 Table of Contents Letter to the Student..................................... 5 Test-Taking Checklist.................................... 6 Next Generation Sunshine State Standards Correlation Chart...
Table of Contents Letter to the Student..................................... 5 Test-Taking Checklist.................................... 6 Next Generation Sunshine State Standards Correlation Chart...
I. ACID-BASE NEUTRALIZATION, TITRATION
 LABORATORY 3 I. ACID-BASE NEUTRALIZATION, TITRATION Acid-base neutralization is a process in which acid reacts with base to produce water and salt. The driving force of this reaction is formation of a
LABORATORY 3 I. ACID-BASE NEUTRALIZATION, TITRATION Acid-base neutralization is a process in which acid reacts with base to produce water and salt. The driving force of this reaction is formation of a
Tutorial 4 SOLUTION STOICHIOMETRY. Solution stoichiometry calculations involve chemical reactions taking place in solution.
 T-27 Tutorial 4 SOLUTION STOICHIOMETRY Solution stoichiometry calculations involve chemical reactions taking place in solution. Of the various methods of expressing solution concentration the most convenient
T-27 Tutorial 4 SOLUTION STOICHIOMETRY Solution stoichiometry calculations involve chemical reactions taking place in solution. Of the various methods of expressing solution concentration the most convenient
MRP-No. DE/H/0279/001/P/002 Dr. Scheffler Vitamin C, 1000mg, effervescent tablets
 PACKAGE LEAFLET: INFORMATION FOR THE USER Dr. Scheffler Vitamin C, 1000 mg, effervescent tablets Ascorbic Acid Read all of this leaflet carefully because it contains important information for you. This
PACKAGE LEAFLET: INFORMATION FOR THE USER Dr. Scheffler Vitamin C, 1000 mg, effervescent tablets Ascorbic Acid Read all of this leaflet carefully because it contains important information for you. This
Vitamin C Content of Foods
 Vitamin C Content of Foods Experiment #11 Objective: To measure the heat and alkaline stability of vitamin C and its quantity in juices or tablets. Introduction Vitamin C is an essential component of the
Vitamin C Content of Foods Experiment #11 Objective: To measure the heat and alkaline stability of vitamin C and its quantity in juices or tablets. Introduction Vitamin C is an essential component of the
Chapter 16: Tests for ions and gases
 The position of hydrogen in the reactivity series Hydrogen, although not a metal, is included in the reactivity series because it, like metals, can be displaced from aqueous solution, only this time the
The position of hydrogen in the reactivity series Hydrogen, although not a metal, is included in the reactivity series because it, like metals, can be displaced from aqueous solution, only this time the
Medical Physiology Z.H.Al-Zubaydi
 Lec.13 Medical Physiology Z.H.Al-Zubaydi Functions of the Digestive System The major functions of the digestive tract include the following six processes, summarized in Figure 1: 1. Ingestion Food must
Lec.13 Medical Physiology Z.H.Al-Zubaydi Functions of the Digestive System The major functions of the digestive tract include the following six processes, summarized in Figure 1: 1. Ingestion Food must
B12 & Cobalamin. Learning objectives
 Learning objectives B12 & Cobalamin Define vitamins Classify fat soluble and water soluble vitamins. Study chemical structure and biological active coenzyme form of vitamin B12. List the dietary sources
Learning objectives B12 & Cobalamin Define vitamins Classify fat soluble and water soluble vitamins. Study chemical structure and biological active coenzyme form of vitamin B12. List the dietary sources
ANSWER KEY. Acids, Bases, and Solutions. Chapter Project Worksheet 1 1. Answers will vary. Sample: cherries, blueberries,
 Chapter Project Worksheet 1 1. Answers will vary. Sample: cherries, blueberries, and grass 2. Answers will vary. Sample: Cut 5 g of cherries into small pieces and place in blender. Blend for two minutes,
Chapter Project Worksheet 1 1. Answers will vary. Sample: cherries, blueberries, and grass 2. Answers will vary. Sample: Cut 5 g of cherries into small pieces and place in blender. Blend for two minutes,
Determination of Aspirin using Back Titration
 Determination of Aspirin using Back Titration This experiment is designed to illustrate techniques used in a typical indirect or back titration. You will use the NaH you standardized last week to back
Determination of Aspirin using Back Titration This experiment is designed to illustrate techniques used in a typical indirect or back titration. You will use the NaH you standardized last week to back
CHAPTER 16: ACIDS AND BASES
 CHAPTER 16: ACIDS AND BASES Active Learning: 4, 6, 14; End-of-Chapter Problems: 2-25, 27-58, 66-68, 70, 75-77, 83, 90-91, 93-104 Chapter 15 End-of-Chapter Problems: 69-74, 125, 129, 133 16.1 ACIDS AND
CHAPTER 16: ACIDS AND BASES Active Learning: 4, 6, 14; End-of-Chapter Problems: 2-25, 27-58, 66-68, 70, 75-77, 83, 90-91, 93-104 Chapter 15 End-of-Chapter Problems: 69-74, 125, 129, 133 16.1 ACIDS AND
The Digestive System. You are what you eat!
 The Digestive System You are what you eat! Try to label the diagram (PENCIL!!) What is Digestion? Digestion: the breakdown of large macromolecules (proteins, fats, carbohydrates) into smaller molecules
The Digestive System You are what you eat! Try to label the diagram (PENCIL!!) What is Digestion? Digestion: the breakdown of large macromolecules (proteins, fats, carbohydrates) into smaller molecules
CHEMICAL PRECIPITATION: WATER SOFTENING
 CHEMICAL PRECIPITATION: WATER SOFTENING Submitted to: Dr. Hashsham Research Complex Engineering Department of Civil and Environmental Engineering Michigan State University East Lansing, MI 4884 Authors
CHEMICAL PRECIPITATION: WATER SOFTENING Submitted to: Dr. Hashsham Research Complex Engineering Department of Civil and Environmental Engineering Michigan State University East Lansing, MI 4884 Authors
EXPERIMENT 4 Acid Strength
 EXPERIMENT 4 Acid Strength Introduction Many common substances are either acids or bases. Some acids, like stomach acid are necessary for our health, while others, like sulfuric acid are dangerous and
EXPERIMENT 4 Acid Strength Introduction Many common substances are either acids or bases. Some acids, like stomach acid are necessary for our health, while others, like sulfuric acid are dangerous and
We use in our daily life a large
 5 Acids, Bases and Salts We use in our daily life a large number of substances such as lemon, tamarind, common salt, sugar and vinegar. Do they have the same taste? Let us recall tastes of some edible
5 Acids, Bases and Salts We use in our daily life a large number of substances such as lemon, tamarind, common salt, sugar and vinegar. Do they have the same taste? Let us recall tastes of some edible
Digestive System Functions
 Digestive System Functions A. Gastrointestinal Processes 1. Ingestion: placing food in mouth (voluntary) 2. Propulsion: moving food through GI tract a. Peristalsis: alternating waves of contraction and
Digestive System Functions A. Gastrointestinal Processes 1. Ingestion: placing food in mouth (voluntary) 2. Propulsion: moving food through GI tract a. Peristalsis: alternating waves of contraction and
2. ATOMIC, MOLECULAR AND EQUIVALENT MASSES
 2. ATOMIC, MOLECULAR AND EQUIVALENT MASSES INTRODUCTION: EQUIVALENT WEIGHT Since hydrogen is the lightest of all elements, it was chosen as a standard for determination of equivalent weights. On this basis,
2. ATOMIC, MOLECULAR AND EQUIVALENT MASSES INTRODUCTION: EQUIVALENT WEIGHT Since hydrogen is the lightest of all elements, it was chosen as a standard for determination of equivalent weights. On this basis,
Note: (H 3 O + = hydronium ion = H + = proton) Example: HS - + H 2 O H 3 O + + S 2-
 AcidBase Chemistry Arrhenius acid: Substance that dissolves in water and provides H + ions Arrhenius base: Substance that dissolves in water and provides OH ions Examples: HCl H + and Cl Acid NaOH Na +
AcidBase Chemistry Arrhenius acid: Substance that dissolves in water and provides H + ions Arrhenius base: Substance that dissolves in water and provides OH ions Examples: HCl H + and Cl Acid NaOH Na +
Q1: What is the ph Scale? Q6: As acids become more acidic, their ph values
 Q1: What is the ph Scale? Q6: As acids become more acidic, their ph values increase or decrease? Q2: The range of values of the ph scale is: Q7: As bases become more alkaline, their ph values increase
Q1: What is the ph Scale? Q6: As acids become more acidic, their ph values increase or decrease? Q2: The range of values of the ph scale is: Q7: As bases become more alkaline, their ph values increase
DULCOLAX Tablets and Suppositories Bisacodyl
 New Zealand Consumer Medicine Information DULCOLAX Tablets and Suppositories Bisacodyl What is in this leaflet This leaflet answers some common questions about DULCOLAX. It does not contain all available
New Zealand Consumer Medicine Information DULCOLAX Tablets and Suppositories Bisacodyl What is in this leaflet This leaflet answers some common questions about DULCOLAX. It does not contain all available
ph Alkalinity of Water
 ph Alkalinity of Water DOC316.52.93085 Based on ISO standard 9963-1:1994 ph-metric Titration 0.4 to 20 mmol/l of Total Alkalinity 1. Introduction Alkalinity of water is its acid-neutralizing capacity.
ph Alkalinity of Water DOC316.52.93085 Based on ISO standard 9963-1:1994 ph-metric Titration 0.4 to 20 mmol/l of Total Alkalinity 1. Introduction Alkalinity of water is its acid-neutralizing capacity.
BLOWING UP BALLOONS, chemically
 BLOWING UP BALLOONS, chemically PRE LAB DISCUSSION: Today we will be using a closed system. A closed system does not permit matter to enter or exit the apparatus. Lavoisier's classic 12-day experiment,
BLOWING UP BALLOONS, chemically PRE LAB DISCUSSION: Today we will be using a closed system. A closed system does not permit matter to enter or exit the apparatus. Lavoisier's classic 12-day experiment,
Chapter 19: Acids and Bases Homework Packet (50 pts) Name: Score: / 50
 Chapter 19: Acids and Bases Homework Packet (50 pts) Topic pg Section 19.1 1-3 Section 19.2 3-6 Section 19.3 6-7 Section 19.4 8 Naming Acids 9 Properties of Acids/Bases 10-11 Conjugate Acid/Base Pairs
Chapter 19: Acids and Bases Homework Packet (50 pts) Topic pg Section 19.1 1-3 Section 19.2 3-6 Section 19.3 6-7 Section 19.4 8 Naming Acids 9 Properties of Acids/Bases 10-11 Conjugate Acid/Base Pairs
Additional Lecture: TITRATION BASICS
 Additional Lecture: TITRATION BASICS 1 Definition and Applications Titration is the incremental addition of a reagent solution (called titrant) to the analyte until the reaction is complete Common applications:
Additional Lecture: TITRATION BASICS 1 Definition and Applications Titration is the incremental addition of a reagent solution (called titrant) to the analyte until the reaction is complete Common applications:
Acids and Bases. Chapter 16
 Acids and Bases Chapter 16 The Arrhenius Model An acid is any substance that produces hydrogen ions, H +, in an aqueous solution. Example: when hydrogen chloride gas is dissolved in water, the following
Acids and Bases Chapter 16 The Arrhenius Model An acid is any substance that produces hydrogen ions, H +, in an aqueous solution. Example: when hydrogen chloride gas is dissolved in water, the following
15. Acid-Base Titration. Discover the concentration of an unknown acid solution using acid-base titration.
 S HIFT INTO NEUTRAL 15. Acid-Base Titration Shift into Neutral Student Instruction Sheet Challenge Discover the concentration of an unknown acid solution using acid-base titration. Equipment and Materials
S HIFT INTO NEUTRAL 15. Acid-Base Titration Shift into Neutral Student Instruction Sheet Challenge Discover the concentration of an unknown acid solution using acid-base titration. Equipment and Materials
Vitamin C Content of Fruit Juice
 1 Vitamin C Content of Fruit Juice Introduction Vitamin C Vitamins are organic compounds that have important biological functions. For instance, in humans they enable a variety of enzymes in the body to
1 Vitamin C Content of Fruit Juice Introduction Vitamin C Vitamins are organic compounds that have important biological functions. For instance, in humans they enable a variety of enzymes in the body to
Problems of the Digestive System
 The American College of Obstetricians and Gynecologists f AQ FREQUENTLY ASKED QUESTIONS FAQ120 WOMEN S HEALTH Problems of the Digestive System What are some common digestive problems? What is constipation?
The American College of Obstetricians and Gynecologists f AQ FREQUENTLY ASKED QUESTIONS FAQ120 WOMEN S HEALTH Problems of the Digestive System What are some common digestive problems? What is constipation?
Topic 4: Digestion and Nutrition
 Topic 4: Digestion and Nutrition THE CONTENTS OF FOOD Food contains nutrients: Nutrients include: 1. 2. 3. 4. 5. Nutrients must be small enough to enter our cells. If they are too large they must be digested
Topic 4: Digestion and Nutrition THE CONTENTS OF FOOD Food contains nutrients: Nutrients include: 1. 2. 3. 4. 5. Nutrients must be small enough to enter our cells. If they are too large they must be digested
hij Teacher Resource Bank GCE Chemistry PSA10: A2 Inorganic Chemistry Carry out a redox titration
 hij Teacher Resource Bank GCE Chemistry : A2 Inorganic Chemistry Copyright 2009 AQA and its licensors. All rights reserved. The Assessment and Qualifications Alliance (AQA) is a company limited by guarantee
hij Teacher Resource Bank GCE Chemistry : A2 Inorganic Chemistry Copyright 2009 AQA and its licensors. All rights reserved. The Assessment and Qualifications Alliance (AQA) is a company limited by guarantee
Continuous process of sodium bicarbonate production by Solvay method
 Continuous process of sodium bicarbonate production by Solvay method Manual to experiment nr 10 Instructor: Dr Tomasz S. Pawłowski 1 Goal of the experiment The goal of the experiment is introduction of
Continuous process of sodium bicarbonate production by Solvay method Manual to experiment nr 10 Instructor: Dr Tomasz S. Pawłowski 1 Goal of the experiment The goal of the experiment is introduction of
Determination of Citric Acid in Powdered Drink Mixes
 Determination of Citric Acid in Powdered Drink Mixes Citric acid and its salts (sodium citrate and potassium citrate) are found in many foods, drinks, pharmaceuticals, shampoos, and cosmetics. The tartness
Determination of Citric Acid in Powdered Drink Mixes Citric acid and its salts (sodium citrate and potassium citrate) are found in many foods, drinks, pharmaceuticals, shampoos, and cosmetics. The tartness
Water Softening for Hardness Removal. Hardness in Water. Methods of Removing Hardness 5/1/15. WTRG18 Water Softening and Hardness
 Water Softening for Removal 1 in Water High concentration of calcium (Ca2+) and magnesium (Mg2+) ions in water cause hardness Generally, water containing more than 100 mg/l of hardness expressed as calcium
Water Softening for Removal 1 in Water High concentration of calcium (Ca2+) and magnesium (Mg2+) ions in water cause hardness Generally, water containing more than 100 mg/l of hardness expressed as calcium
PECTINS. SYNONYMS INS No. 440 DEFINITION DESCRIPTION. FUNCTIONAL USES Gelling agent, thickener, stabilizer, emulsifier CHARACTERISTICS
 PECTINS SYNONYMS INS No. 440 Prepared at the 71 st JECFA (2009) and published in FAO JECFA Monographs 7 (2009), superseding specifications prepared at the 68 th JECFA (2007) and published in FAO JECFA
PECTINS SYNONYMS INS No. 440 Prepared at the 71 st JECFA (2009) and published in FAO JECFA Monographs 7 (2009), superseding specifications prepared at the 68 th JECFA (2007) and published in FAO JECFA
Eating, pooping, and peeing THE DIGESTIVE SYSTEM
 THE DIGESTIVE SYSTEM Ingested food is not technically in the body until it is absorbed so it needs to be: Mechanically and chemically reduced Transported by the blood to the cells Large portions are not
THE DIGESTIVE SYSTEM Ingested food is not technically in the body until it is absorbed so it needs to be: Mechanically and chemically reduced Transported by the blood to the cells Large portions are not
National Digestive Diseases Information Clearinghouse
 Gastritis National Digestive Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is gastritis? Gastritis is a condition in which the stomach
Gastritis National Digestive Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is gastritis? Gastritis is a condition in which the stomach
Simulation of the determination of lead azide content in waste water from explosives manufacture
 Simulation of the determination of lead azide content in waste water from explosives manufacture Lead azide ranks in the category of intensive explosives, which may, even in an insignificant amount, initiate
Simulation of the determination of lead azide content in waste water from explosives manufacture Lead azide ranks in the category of intensive explosives, which may, even in an insignificant amount, initiate
Q1. A student studied the reaction between dilute hydrochloric acid and an excess of calcium carbonate.
 Q. A student studied the reaction between dilute hydrochloric acid and an excess of calcium carbonate. calcium carbonate + hydrochloric acid calcium chloride + water + carbon dioxide The student measured
Q. A student studied the reaction between dilute hydrochloric acid and an excess of calcium carbonate. calcium carbonate + hydrochloric acid calcium chloride + water + carbon dioxide The student measured
Digestive System Why is digestion important? How is food digested? Physical Digestion and Movement
 Digestive System The digestive system is made up of the digestive tract a series of hollow organs joined in a long, twisting tube from the mouth to the anus and other organs that help the body break down
Digestive System The digestive system is made up of the digestive tract a series of hollow organs joined in a long, twisting tube from the mouth to the anus and other organs that help the body break down
Determination of calcium by Standardized EDTA Solution
 Determination of calcium by Standardized EDTA Solution Introduction The classic method of determining calcium and other suitable cations is titration with a standardized solution of ethylenediaminetetraacetic
Determination of calcium by Standardized EDTA Solution Introduction The classic method of determining calcium and other suitable cations is titration with a standardized solution of ethylenediaminetetraacetic
Determining the Quantity of Iron in a Vitamin Tablet. Evaluation copy
 Determining the Quantity of Iron in a Vitamin Tablet Computer 34 As biochemical research becomes more sophisticated, we are learning more about the role of metallic elements in the human body. For example,
Determining the Quantity of Iron in a Vitamin Tablet Computer 34 As biochemical research becomes more sophisticated, we are learning more about the role of metallic elements in the human body. For example,
DEIONIZATION IN A "NUT SHELL"
 Deionized Water (DI) DEIONIZATION IN A "NUT SHELL" City water is passed through dark amber colored, caviar sized plastic beads called cation ion exchange resin. The cation resin is in the hydrogen form
Deionized Water (DI) DEIONIZATION IN A "NUT SHELL" City water is passed through dark amber colored, caviar sized plastic beads called cation ion exchange resin. The cation resin is in the hydrogen form
1. Read P. 368-375, P. 382-387 & P. 429-436; P. 375 # 1-11 & P. 389 # 1,7,9,12,15; P. 436 #1, 7, 8, 11
 SCH3U- R.H.KING ACADEMY SOLUTION & ACID/BASE WORKSHEET Name: The importance of water - MAKING CONNECTION READING 1. Read P. 368-375, P. 382-387 & P. 429-436; P. 375 # 1-11 & P. 389 # 1,7,9,12,15; P. 436
SCH3U- R.H.KING ACADEMY SOLUTION & ACID/BASE WORKSHEET Name: The importance of water - MAKING CONNECTION READING 1. Read P. 368-375, P. 382-387 & P. 429-436; P. 375 # 1-11 & P. 389 # 1,7,9,12,15; P. 436
Experiment 18: ph Measurements of Common Substances. Experiment 17: Reactions of Acids with Common Substances
 Experiment 18: ph Measurements of Common Substances and Experiment 17: Reactions of Acids with Common Substances What is this lab about? You mean what ARE THESE labs about? Ok, so what are THESE labs about?
Experiment 18: ph Measurements of Common Substances and Experiment 17: Reactions of Acids with Common Substances What is this lab about? You mean what ARE THESE labs about? Ok, so what are THESE labs about?
