The Final Learning Experience
|
|
|
- Naomi Davidson
- 7 years ago
- Views:
Transcription
1 Chemistry 212 and 300 rganic Chemistry II Winter Semester 2000 Dr. Rainer Glaser Examination #5 Carbon-Carbon Bond Forming Reactions, Sugars & Nucleic Acids The Final Learning Experience Friday, May 5, 2000, 3:30-5:30 Your Name: Question 1. verview 40 Question 2. Fancy Aldehydes & Ketones & Acids 30 Question 3. Syntheses of Carbocycles 25 Question 4. Aromatic Chemistry 20 Question 5. Carbohydrates 55 Question 6. DNA & RNA Structure 30 Question 7. Wittig Reaction (News #18) 40 Total 240 Exam #4, The Final, Chemistry 212, WS00, Dr. Glaser 1
2 Question 1. verview. (40 points) There you are, you made it through the second semester of rganic Chemistry! While the first semester of organic chemistry basically was an introduction to bonding, stereochemistry and the chemistry of a few important functional groups, in the second semester of organic chemistry you learned how to change the carbon skeleton, the center issue of synthesis. ence, carbon-carbon bond forming reactions are in the focal point of this final. The chapter on carbanion chemistry contains many of the carbon-carbon bond forming reactions. ne of the standard CC bond forming reactions is the aldol reaction. The aldol reaction of an aldehyde can lead to by way of the aldol addition or it can go further and lead to after condensation of. This reaction basically involves the nucleophilic addition of a carbanion to a carbonyl group. The Claisen reaction is analogous to the Aldol reaction in that it is a dimerization of sorts. The Claisen condensation of an ester leads to. But there is a major mechanistic difference: While carbanions add to the carbonyl compound in the aldol reaction, the carbanion does a nucleophilic reaction with the neutral ester. This reaction goes through a intermediate and the reactions mechanism is designated as. The Michael additions are the third big group of C-C bond formations involving carbanions. In a Michael addition a carbanion is added to an electron- alkene. This is an example of a nucleophilic addition to an alkene. You learned about electrophilic additions to alkenes in the first semester. The nucleophilic substitution reaction of carboxylic acids and of their derivatives are nucleophilic substitutions at an unsaturated carbon-atom. Aside from these reactions, there also are many nucleophilic substitution reactions at saturated carbons that can be used in carbon-carbon bond forming reactions. A simple reaction of this type would be the nucleophilic substitution of chlorine in an alkyl chloride by cyanide ion. More complicated carbon nucleophiles of course also can be used and we will return to this chemistry below. These reactions involve alkylations of As & Ks and the alkylations of dicarbonyls. These kinds of reactions play an important role in the malonic acid synthesis Exam #4, The Final, Chemistry 212, WS00, Dr. Glaser 2
3 and the acetoacetic ester synthesis. All of these reactions form products that basically are fancy aldehydes, fancy ketones, fancy esters or acids, fancy alkenes or bigger molecules with several functionalities. The carbanion plus carbonyl approach to carbon-carbon bonds may involve two types of carbanions: reagents and other organometallic reagents, on the one hand, and, on the other hand, the carbanions generated from carbon- compounds, that is, compounds that contain -atoms in the alpha-position to a carbonyl group. Aside from the carbanion plus carbonyl approach, carbon-carbon bond formations also can be achieved by way of the Wittig reaction. Wittig reactions contains carbanionic carbons in the overall (neutral, cationic, anionic) ylides. We will take a look at a few Wittig reactions in the last question. Finally, aromatic chemistry was a big part of this course and we will review some of that chemistry again with a focus on carbon-carbon bond formation. ere we will try to make C-C bonds between a C-atom that is part of a benzene ring and another C-atom that is part of an aliphatic system. This kind of bond formation can be accomplished by way of a nucleophilic aromatic substitution. In this case a carbanion is generated and reacted with a benzene derivative that contains a very good nucleofug. Benzonitrile, for example can be readily obtained by nucleophilic aromatic substitution of the dinitrogen of benzene ion by the nucleophile which is employed as its salt. This reaction is called the reaction. The Friedel- Crafts reactions are C-C bond formations of a different type. In the FC-alkylation, it is the benzene that serves as the (electrophile, nucleophile) seeking to replace the halide of an alkyl halide with some help by a -acid. If one looks at this reaction from the perspective that something happens to the benzene, then one classifies this Friedel-Crafts alkylation reaction as an (electrophilic, nucleophilic) aromatic substitution where the electrophile is a. (40 points total, 2 points for each correct entry). Exam #4, The Final, Chemistry 212, WS00, Dr. Glaser 3
4 Question 2. Fancy Aldehydes & Ketones & Acids. (30 points) (a) Beta-ketoesters undergo decarboxylation after hydrolysis and upon heating. This decarboxylation proceeds via a 6-membered ring transition state. For the beta-ketoacid shown, draw the conformation that contains the 6-membered ring and draw curved arrows that indicate the electron flow in the decarboxylation. Also draw the products of the decarboxylation. (8 points) (b) Draw the product obtained after hydrolysis and decarboxylation of the diester shown. Provide an acceptable IUPAC name of the product. (6 points) Et Et Exam #4, The Final, Chemistry 212, WS00, Dr. Glaser 4
5 (c) Malonic Ester Synthesis. The ethyl diester of malonic acid, EtC-C 2 -CEt, is treated with sodium ethoxide followed by ethyl bromide. The product is treated with sodium ethoxide again and then again with ethyl bromide. Draw the product obtained after the twofold application of the NaEt/EtBr sequence. Then draw the product that is obtained after hydrolysis and draw the final product obtain after heating. Name the final product. (8 points) (d) Acetoacetic Ester Synthesis. The methyl ketone 3 C-C-C 2 -C 2 -C 3 can be prepared via an acetoacetic ester synthesis. Draw the structure of acetoacetic ester. Give reagents and product of the alkylation. Draw the structure of the product obtained after hydrolysis. Draw the structure after decarboxylation. Name the final product, the methyl ketone. (8 points) Exam #4, The Final, Chemistry 212, WS00, Dr. Glaser 5
6 Question 3. Synthesis of Carbocycles. (25 points) (a) The Robinson annulation is a classic method for the synthesis of 6-membered rings. The Robinson annulation is a base-catalyzed tandem reaction and involves a -reaction and an -condensation. Draw the substrates used to prepare alpha,beta-unsaturated cyclohexene shown. Also draw the intermediate obtained by the first reaction of the sequence. (9 points) (b) Suggest a synthesis of cyclohexanecarboxylic acid via a Diels-Alder reaction. Show substrates of the Diels-Alder reaction. Show the electron flow of the DAR using curved arrows. Suggest a method for the conversion of the product of the DAR into the cyclohexane derivative. (8 points) (c) Suggest a synthesis of cyclopentanone by way of a Dieckmann condensation of an acyclic diester. Show the structures of the substrate diester and of the product of the Dieckmann condensation. Give conditions for the conversion of the beta-ketoester to the final product. (8 points) Exam #4, The Final, Chemistry 212, WS00, Dr. Glaser 6
7 Question 4. Aromatic Chemistry. (20 points) (a) Draw the structure of the major product of this Friedel-Crafts. (4 points) C C 3 -C-Cl 2. AlCl 3 (b) Suggest a way to convert acetophenone, Ph-C-Me, to ethyl benzene. (2 points). (c) Suggest a synthesis of benzoic acid starting from toluene. Draw toluene, draw benzoic acid, and provide the reagent needed. (6 points) (d) Suggest a synthesis of benzoic acid starting from aniline. Draw aniline. Provide the reagent needed for diazotization and provide the reagents needed for all subsequent transformations. (8 points) Exam #4, The Final, Chemistry 212, WS00, Dr. Glaser 7
8 Question 5. Carbohydrates. (55 points) (a) Glycerinaldehyde is the smallest (aldose, ketose). Draw the Fischer projection of D- glycerinaldehyde and also produce a perspective drawing of this same molecule. Mark the chiral carbon with a star. In the R/S-nomenclature, the absolute configuration of the chiral carbon is. (9 points) (b) D-glycerinaldehyde is subjected to a chain elongation. This chain elongation involves a cyanohydrin formation, then a hydrolysis, and then a reduction with sodium amalgam. This chain elongation is referred to as the synthesis. The cyanohydrin formation involves the reaction of the reagent with the aldehyde function of the sugar. The hydrolysis of the cyanohydrin produces an acid which is then reduced to the aldose. The C1-carbon atom of D-glycerinaldehyde is converted into a chiral center in this chain elongation reaction and two products are formed. Draw the Fischer projections of these two products. For each of these products, mark whether it is D or L. These two products are (enantiomers, diastereoisomers). These two products also are referred to as (epimers, anomers) because they differ in the configuration of (give number) chiral center(s). ne of these products features a stereochmistry described as erythro and the other features a stereochemistry that is referred to as threo. Assign the correct label to the two products. (20 points, 6*2 for blanks, 8 for structures and labels.) Exam #4, The Final, Chemistry 212, WS00, Dr. Glaser 8
9 (c) D-glycerinaldehyde is treated with periodic acid, I 4. Show what products are formed in this reaction. When applied to a cyclic carbohydrate, the periodic acid cleavage provides information about the of the sugar. (6 points) (d) The molecule shown is a (mono, di, oligo, poly) saccharide. This sugar contains (0, 1, 2) glucose molecule(s) and (0, 1, 2) fructose molecule(s). Both rings are (pyranoses, furanoses). The molecule (is, is not) a reducing sugar. The molecule is (alpha, beta) at the central anomeric carbon-atom. The stereochemistry of the hemiacetal is shown as (alpha, beta) and interconversion (is, is not) possible at that position. This sugar contains one (1,4-, 1,6-, 4,6-) glycosidic bond. The trivial name of this sugar is and this sugar is contained in (cellulose, amylose). (20 points) Exam #4, The Final, Chemistry 212, WS00, Dr. Glaser 9
10 Question 6. DNA & RNA Structure. (30 points) N N N N N 2 - P - (1, 2 p.) Label the C-atoms of the sugar moiety (e.g. assign the numbers 1 to 5 to the correct atoms). (2, 2 p.) The sugar is a (pyranose, furanose). (3, 2 p.) The fragment shown can be found in (RNA, DNA). (4, 2 p.) The sugar is (give the name of the free sugar). (5, 2 p.) The anomeric carbon is (alpha, beta). (6, 2 p.) The fragment shown is a (nucleoside, nucleotide). (7, 2 p.) Circle the alcohol function that would be used for another phosphate ester formation of the fragment shown would be incorporated into a single strand. (8, 4 p.) The nucleobase in the fragment shown is or (C, T, U, A, G) for short. (9, 2 p.) This nucleobase is a (purine, pyrimidine) base. (10, 2 p.) The name of the fragment shown is. (11, 2 p.) The fragment shown is abbreviated as (UMP, dump, AMP, damp, etc). (12, 2 p.) In the Watson-Crick base pair, this nucleobase would -bond with (C, T, U, A, G). (13, 2 p.) The Watson-Crick base pair involving the fragment shown contains -bonds. (14, 2 p.) The hybridization of the N-atom involved in the N-glycosidic bond is. Exam #4, The Final, Chemistry 212, WS00, Dr. Glaser 10
11 Question 7. Wittig Reaction and Vitamin A. (40 points, News #18) The newspaper article NATURAL VITAMINS AVE MRE ANTIXIDANTS TAN MAN- MADE featured vitamin A and beta-carotenes as important antioxidants. A good article to review some of the features of the Wittig reaction and of its important applications. (a) In a Wittig reaction, a compound and an ylide react to form an (name of the class of compounds) and a phosphine. The ylide is formed by treatment of a phosphonium halide with a strong base. The phosphonium halide is generated by way of a (nucleophilic, electrophilic) substitution reaction of an (name of the class of compounds) with a phoshine. (10 points) (b) Suppose you convert benzyl bromide into an ylide using triphenylphosphine and Na as the base. Suppose further that you react this ylide with acetone. Draw both of the important resonance forms of the ylide (pay attention to formal charges), draw the structure of the oxaphosphetane formed by reaction with acetone, and draw the structure of the product of the Wittig reaction. Using curved arrows show how the oxaphosphetane is formed and how it is cleaved. (14 points) Exam #4, The Final, Chemistry 212, WS00, Dr. Glaser 11
12 (c) The phosphonium chloride shown can be deprotonated with sodium methoxide to form an ylide. Draw this ylide. This ylide can couple with an appropriate aldehyde X to obtain the vitamin A acetate shown. Draw the structure of the aldehyde X. (8 points) Cl - + PPh 3 (d) The ylide derived from the phosphonium chloride shown in (c) serves as the starting material for a synthesis of the beta-carotene shown below. The beta-carotene is constructed with two Wittig reactions between two molecules of the same ylide and one di-aldehyde. Indicate which two C=C bonds are formed in these two Wittig reactions and provide the structure of the required di-aldehyde. (8 points) Exam #4, The Final, Chemistry 212, WS00, Dr. Glaser 12
13 Exam #4, The Final, Chemistry 212, WS00, Dr. Glaser 13
Chapter 22 Carbonyl Alpha-Substitution Reactions
 John E. McMurry www.cengage.com/chemistry/mcmurry Chapter 22 Carbonyl Alpha-Substitution Reactions The α Position The carbon next to the carbonyl group is designated as being in the α position Electrophilic
John E. McMurry www.cengage.com/chemistry/mcmurry Chapter 22 Carbonyl Alpha-Substitution Reactions The α Position The carbon next to the carbonyl group is designated as being in the α position Electrophilic
Carboxylic Acid Derivatives and Nitriles
 Carboxylic Acid Derivatives and itriles Carboxylic Acid Derivatives: There are really only four things to worry about under this heading; acid chlorides, anhydrides, esters and amides. We ll start with
Carboxylic Acid Derivatives and itriles Carboxylic Acid Derivatives: There are really only four things to worry about under this heading; acid chlorides, anhydrides, esters and amides. We ll start with
Chapter 11 Homework and practice questions Reactions of Alkyl Halides: Nucleophilic Substitutions and Eliminations
 Chapter 11 Homework and practice questions Reactions of Alkyl Halides: Nucleophilic Substitutions and Eliminations SHORT ANSWER Exhibit 11-1 Circle your response in each set below. 1. Circle the least
Chapter 11 Homework and practice questions Reactions of Alkyl Halides: Nucleophilic Substitutions and Eliminations SHORT ANSWER Exhibit 11-1 Circle your response in each set below. 1. Circle the least
But in organic terms: Oxidation: loss of H 2 ; addition of O or O 2 ; addition of X 2 (halogens).
 Reactions of Alcohols Alcohols are versatile organic compounds since they undergo a wide variety of transformations the majority of which are either oxidation or reduction type reactions. Normally: Oxidation
Reactions of Alcohols Alcohols are versatile organic compounds since they undergo a wide variety of transformations the majority of which are either oxidation or reduction type reactions. Normally: Oxidation
Lecture Overview. Hydrogen Bonds. Special Properties of Water Molecules. Universal Solvent. ph Scale Illustrated. special properties of water
 Lecture Overview special properties of water > water as a solvent > ph molecules of the cell > properties of carbon > carbohydrates > lipids > proteins > nucleic acids Hydrogen Bonds polarity of water
Lecture Overview special properties of water > water as a solvent > ph molecules of the cell > properties of carbon > carbohydrates > lipids > proteins > nucleic acids Hydrogen Bonds polarity of water
California State Polytechnic University, Pomona. Exam Points 1. Nomenclature (1) 30
 Chem 316 Final Exam Winter, 2008 Beauchamp ame: Topic Total Points Exam Points 1. omenclature (1) 30 Credit 2. Explanation of elative eactivities of Aromatic 20 Compounds or Carbonyl Compounds 3. eactions
Chem 316 Final Exam Winter, 2008 Beauchamp ame: Topic Total Points Exam Points 1. omenclature (1) 30 Credit 2. Explanation of elative eactivities of Aromatic 20 Compounds or Carbonyl Compounds 3. eactions
Mass Spec - Fragmentation
 Mass Spec - Fragmentation An extremely useful result of EI ionization in particular is a phenomenon known as fragmentation. The radical cation that is produced when an electron is knocked out of a neutral
Mass Spec - Fragmentation An extremely useful result of EI ionization in particular is a phenomenon known as fragmentation. The radical cation that is produced when an electron is knocked out of a neutral
Introduction to Biodiesel Chemistry Terms and Background Information
 Introduction to Biodiesel Chemistry Terms and Background Information Basic rganic Chemistry rganic chemistry is the branch of chemistry that deals with organic compounds. rganic compounds are compounds
Introduction to Biodiesel Chemistry Terms and Background Information Basic rganic Chemistry rganic chemistry is the branch of chemistry that deals with organic compounds. rganic compounds are compounds
3) How many monosaccharides are connected to each other in a disaccharide? A) 1 B) 2 C) 3 D) 4
 General, Organic, and Biochemistry, 2e (Frost) HOMEWORK Chapter 6 Carbohydrates Life s Sweet Molecules 6.1 Multiple-Choice 1) Which of the following is a polysaccharide? Glucose Sucrose C) Starch D) Maltose
General, Organic, and Biochemistry, 2e (Frost) HOMEWORK Chapter 6 Carbohydrates Life s Sweet Molecules 6.1 Multiple-Choice 1) Which of the following is a polysaccharide? Glucose Sucrose C) Starch D) Maltose
4/18/2011. 9.8 Substituent Effects in Electrophilic Substitutions. Substituent Effects in Electrophilic Substitutions
 9.8 Substituent effects in the electrophilic substitution of an aromatic ring Substituents affect the reactivity of the aromatic ring Some substituents activate the ring, making it more reactive than benzene
9.8 Substituent effects in the electrophilic substitution of an aromatic ring Substituents affect the reactivity of the aromatic ring Some substituents activate the ring, making it more reactive than benzene
Disaccharides consist of two monosaccharide monomers covalently linked by a glycosidic bond. They function in sugar transport.
 1. The fundamental life processes of plants and animals depend on a variety of chemical reactions that occur in specialized areas of the organism s cells. As a basis for understanding this concept: 1.
1. The fundamental life processes of plants and animals depend on a variety of chemical reactions that occur in specialized areas of the organism s cells. As a basis for understanding this concept: 1.
I. Chapter 5 Summary. II. Nucleotides & Nucleic Acids. III. Lipids
 I. Chapter 5 Summary A. Simple Sugars (CH 2 O) n : 1. One C contains a carbonyl (C=O) rest contain - 2. Classification by functional group: aldoses & ketoses 3. Classification by number of C's: trioses,
I. Chapter 5 Summary A. Simple Sugars (CH 2 O) n : 1. One C contains a carbonyl (C=O) rest contain - 2. Classification by functional group: aldoses & ketoses 3. Classification by number of C's: trioses,
1. The functional group present in carboxylic acids is called a A) carbonyl group. B) carboxyl group. C) carboxylate group. D) carbohydroxyl group.
 Name: Date: 1. The functional group present in carboxylic acids is called a A) carbonyl group. B) carboxyl group. C) carboxylate group. D) carbohydroxyl group. 2. Which of the following statements concerning
Name: Date: 1. The functional group present in carboxylic acids is called a A) carbonyl group. B) carboxyl group. C) carboxylate group. D) carbohydroxyl group. 2. Which of the following statements concerning
Q.1 Draw out some suitable structures which fit the molecular formula C 6 H 6
 Aromatic compounds GE 1 BENZENE Structure Primary analysis revealed benzene had an... empirical formula of and a molecular formula of 6 6 Q.1 Draw out some suitable structures which fit the molecular formula
Aromatic compounds GE 1 BENZENE Structure Primary analysis revealed benzene had an... empirical formula of and a molecular formula of 6 6 Q.1 Draw out some suitable structures which fit the molecular formula
Writing a Correct Mechanism
 Chapter 2 1) Balancing Equations Writing a Correct Mechanism 2) Using Arrows to show Electron Movement 3) Mechanisms in Acidic and Basic Media 4) Electron rich Species: Nucleophile or Base? 5) Trimolecular
Chapter 2 1) Balancing Equations Writing a Correct Mechanism 2) Using Arrows to show Electron Movement 3) Mechanisms in Acidic and Basic Media 4) Electron rich Species: Nucleophile or Base? 5) Trimolecular
Chapter 5: The Structure and Function of Large Biological Molecules
 Name Period Concept 5.1 Macromolecules are polymers, built from monomers 1. The large molecules of all living things fall into just four main classes. Name them. 2. Circle the three classes that are called
Name Period Concept 5.1 Macromolecules are polymers, built from monomers 1. The large molecules of all living things fall into just four main classes. Name them. 2. Circle the three classes that are called
Aldehydes can react with alcohols to form hemiacetals. 340 14. Nucleophilic substitution at C=O with loss of carbonyl oxygen
 340 14. Nucleophilic substitution at C= with loss of carbonyl oxygen Ph In Chapter 13 we saw this way of making a reaction go faster by raising the energy of the starting material. We also saw that the
340 14. Nucleophilic substitution at C= with loss of carbonyl oxygen Ph In Chapter 13 we saw this way of making a reaction go faster by raising the energy of the starting material. We also saw that the
Identification of Unknown Organic Compounds
 Identification of Unknown Organic Compounds Introduction The identification and characterization of the structures of unknown substances are an important part of organic chemistry. Although it is often
Identification of Unknown Organic Compounds Introduction The identification and characterization of the structures of unknown substances are an important part of organic chemistry. Although it is often
CORK INSTITUTE OF TECHNOLOGY INSTITIÚID TEICNEOLAÍOCHTA CHORCAÍ
 CORK INSTITUTE OF TECHNOLOGY INSTITIÚID TEICNEOLAÍOCHTA CHORCAÍ Module Title: Topics in Organic Chemistry Module Code: CHEO 7003 School : Science Programme Title: Bachelor of Science in Analytical & Pharmaceutical
CORK INSTITUTE OF TECHNOLOGY INSTITIÚID TEICNEOLAÍOCHTA CHORCAÍ Module Title: Topics in Organic Chemistry Module Code: CHEO 7003 School : Science Programme Title: Bachelor of Science in Analytical & Pharmaceutical
A disaccharide is formed when a dehydration reaction joins two monosaccharides. This covalent bond is called a glycosidic linkage.
 CH 5 Structure & Function of Large Molecules: Macromolecules Molecules of Life All living things are made up of four classes of large biological molecules: carbohydrates, lipids, proteins, and nucleic
CH 5 Structure & Function of Large Molecules: Macromolecules Molecules of Life All living things are made up of four classes of large biological molecules: carbohydrates, lipids, proteins, and nucleic
A Grignard reagent formed would deprotonate H of the ethyl alcohol OH.
 216 S11-E2 Page 2 Name Key I. (9 points) Answer in the boxes below the following questions for the Grignard reagent C 3 -Mg. (1) (2 points) Is the carbon atom associated with magnesium electrophilic or
216 S11-E2 Page 2 Name Key I. (9 points) Answer in the boxes below the following questions for the Grignard reagent C 3 -Mg. (1) (2 points) Is the carbon atom associated with magnesium electrophilic or
Catalysis by Enzymes. Enzyme A protein that acts as a catalyst for a biochemical reaction.
 Catalysis by Enzymes Enzyme A protein that acts as a catalyst for a biochemical reaction. Enzymatic Reaction Specificity Enzyme Cofactors Many enzymes are conjugated proteins that require nonprotein portions
Catalysis by Enzymes Enzyme A protein that acts as a catalyst for a biochemical reaction. Enzymatic Reaction Specificity Enzyme Cofactors Many enzymes are conjugated proteins that require nonprotein portions
HOMEWORK PROBLEMS: IR SPECTROSCOPY AND 13C NMR. The peak at 1720 indicates a C=O bond (carbonyl). One possibility is acetone:
 HMEWRK PRBLEMS: IR SPECTRSCPY AND 13C NMR 1. You find a bottle on the shelf only labeled C 3 H 6. You take an IR spectrum of the compound and find major peaks at 2950, 1720, and 1400 cm -1. Draw a molecule
HMEWRK PRBLEMS: IR SPECTRSCPY AND 13C NMR 1. You find a bottle on the shelf only labeled C 3 H 6. You take an IR spectrum of the compound and find major peaks at 2950, 1720, and 1400 cm -1. Draw a molecule
Organic Chemistry, 5e (Bruice) Chapter 17: Carbonyl Compounds II
 Organic Chemistry, 5e (Bruice) Chapter 17: Carbonyl Compounds II 1) Which of the following compounds is an aldehyde? A) I B) II C) III D) IV E) V D Section: 17-1 2) Which of the following compounds is
Organic Chemistry, 5e (Bruice) Chapter 17: Carbonyl Compounds II 1) Which of the following compounds is an aldehyde? A) I B) II C) III D) IV E) V D Section: 17-1 2) Which of the following compounds is
Name. Department of Chemistry and Biochemistry SUNY/Oneonta. Chem 322 - Organic Chemistry II Examination #2 - March 14, 2005 ANSWERS
 Name INSTRUTINS --- Department of hemistry and Biochemistry SUNY/neonta hem 322 - rganic hemistry II Examination #2 - March 14, 2005 ANSWERS This examination has two parts. Part I is in multiple choice
Name INSTRUTINS --- Department of hemistry and Biochemistry SUNY/neonta hem 322 - rganic hemistry II Examination #2 - March 14, 2005 ANSWERS This examination has two parts. Part I is in multiple choice
methyl RX example primary RX example secondary RX example secondary RX example tertiary RX example
 ucleophilic Substitution & Elimination hemistry 1 eginning patterns to knowfor S and E eactions - horizontal and vertical templates for practice Example 1 - two possible perspectives (deuterium and tritium
ucleophilic Substitution & Elimination hemistry 1 eginning patterns to knowfor S and E eactions - horizontal and vertical templates for practice Example 1 - two possible perspectives (deuterium and tritium
Chapter 3: Biological Molecules. 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids
 Chapter 3: Biological Molecules 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids Elements in Biological Molecules Biological macromolecules are made almost entirely of just 6 elements: Carbon (C)
Chapter 3: Biological Molecules 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids Elements in Biological Molecules Biological macromolecules are made almost entirely of just 6 elements: Carbon (C)
Elements in Biological Molecules
 Chapter 3: Biological Molecules 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids Elements in Biological Molecules Biological macromolecules are made almost entirely of just 6 elements: Carbon (C)
Chapter 3: Biological Molecules 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids Elements in Biological Molecules Biological macromolecules are made almost entirely of just 6 elements: Carbon (C)
pk a Values for Selected Compounds
 Appendix A pk a Values for Selected ompounds ompound pk a ompound pk a I 10 Br 9 2 S 4 9 + 3 3 7.3 3 S 3 7 Br 4.0 4.2 3 4.3 2 N l 7 [( 3 ) 2 ] + 3.8 [ 3 2 ] + 2.5 3 + 1.7 3 S 3 1.2 + 3 N2 0.0 F 3 0.2 l
Appendix A pk a Values for Selected ompounds ompound pk a ompound pk a I 10 Br 9 2 S 4 9 + 3 3 7.3 3 S 3 7 Br 4.0 4.2 3 4.3 2 N l 7 [( 3 ) 2 ] + 3.8 [ 3 2 ] + 2.5 3 + 1.7 3 S 3 1.2 + 3 N2 0.0 F 3 0.2 l
Name Lab #3: Solubility of Organic Compounds Objectives: Introduction: soluble insoluble partially soluble miscible immiscible
 Lab #3: Solubility of rganic Compounds bjectives: - Understanding the relative solubility of organic compounds in various solvents. - Exploration of the effect of polar groups on a nonpolar hydrocarbon
Lab #3: Solubility of rganic Compounds bjectives: - Understanding the relative solubility of organic compounds in various solvents. - Exploration of the effect of polar groups on a nonpolar hydrocarbon
CH 102 Practice Exam 2 PCC-Sylvania
 CH 102 Practice Exam 2 PCC-Sylvania True/False Indicate if the statement is true or false. 1.Tertiary alcohols are not easily oxidized. 2.Secondary alcohols can be oxidized to aldehydes. 3.Primary alcohols
CH 102 Practice Exam 2 PCC-Sylvania True/False Indicate if the statement is true or false. 1.Tertiary alcohols are not easily oxidized. 2.Secondary alcohols can be oxidized to aldehydes. 3.Primary alcohols
AROMATIC COMPOUNDS A STUDENT SHOULD BE ABLE TO:
 A STUDENT SHULD BE ABLE T: ARMATIC CMPUNDS 1. Name benzene derivatives given the structures, and draw the structures given the names. This includes: Monosubstituted benzenes named as derivatives of benzene:
A STUDENT SHULD BE ABLE T: ARMATIC CMPUNDS 1. Name benzene derivatives given the structures, and draw the structures given the names. This includes: Monosubstituted benzenes named as derivatives of benzene:
CHEM 211 CHAPTER 16 - Homework
 CHEM 211 CHAPTER 16 - Homework SHORT ANSWER Consider the Friedel-Crafts alkylation reaction below to answer the following question(s): 1. Refer to the reaction above. Draw the structure of the electrophilic
CHEM 211 CHAPTER 16 - Homework SHORT ANSWER Consider the Friedel-Crafts alkylation reaction below to answer the following question(s): 1. Refer to the reaction above. Draw the structure of the electrophilic
The Chemistry of Carbohydrates
 The Chemistry of Carbohydrates Experiment #5 Objective: To determine the carbohydrate class of an unknown by carrying out a series of chemical reactions with the unknown and known compounds in each class
The Chemistry of Carbohydrates Experiment #5 Objective: To determine the carbohydrate class of an unknown by carrying out a series of chemical reactions with the unknown and known compounds in each class
Acids and Bases: Molecular Structure and Acidity
 Acids and Bases: Molecular Structure and Acidity Review the Acids and Bases Vocabulary List as needed. Tutorial Contents A. Introduction B. Resonance C. Atomic Radius D. Electronegativity E. Inductive
Acids and Bases: Molecular Structure and Acidity Review the Acids and Bases Vocabulary List as needed. Tutorial Contents A. Introduction B. Resonance C. Atomic Radius D. Electronegativity E. Inductive
IUPAC System of Nomenclature
 IUPAC System of Nomenclature The IUPAC (International Union of Pure and Applied Chemistry) is composed of chemists representing the national chemical societies of several countries. ne committee of the
IUPAC System of Nomenclature The IUPAC (International Union of Pure and Applied Chemistry) is composed of chemists representing the national chemical societies of several countries. ne committee of the
CHM220 Addition lab. Experiment: Reactions of alkanes, alkenes, and cycloalkenes*
 CM220 Addition lab Experiment: Reactions of alkanes, alkenes, and cycloalkenes* Purpose: To investigate the physical properties, solubility, and density of some hydrocarbon. To compare the chemical reactivity
CM220 Addition lab Experiment: Reactions of alkanes, alkenes, and cycloalkenes* Purpose: To investigate the physical properties, solubility, and density of some hydrocarbon. To compare the chemical reactivity
CHEM 208(Organic Chemistry I) Instructor: Dr. Niranjan Goswami. Tel: (618)545-3361. Email: Ngoswami@kaskaskia.edu. Web: www.kc.cc.il.
 CHEM 208(Organic Chemistry I) Instructor: Dr. Niranjan Goswami Tel: (618)545-3361 Email: Ngoswami@kaskaskia.edu Web: www.kc.cc.il.us/ngoswami CHEM 208 COURSE SYLLABUS KASKASKIA COLLEGE NAME TERM YEAR TEXT:
CHEM 208(Organic Chemistry I) Instructor: Dr. Niranjan Goswami Tel: (618)545-3361 Email: Ngoswami@kaskaskia.edu Web: www.kc.cc.il.us/ngoswami CHEM 208 COURSE SYLLABUS KASKASKIA COLLEGE NAME TERM YEAR TEXT:
IDENTIFICATION OF ALCOHOLS
 IDENTIFICATION OF ALCOHOLS Alcohols are organic compounds that which considered as derivatives of water. One of the hydrogen atoms of water molecule (H-O-H) has been replaced by an alkyl or substituted
IDENTIFICATION OF ALCOHOLS Alcohols are organic compounds that which considered as derivatives of water. One of the hydrogen atoms of water molecule (H-O-H) has been replaced by an alkyl or substituted
Electrophilic Aromatic Substitution Reactions
 Electrophilic Aromatic Substitution Reactions, Course Notes Archive, 1 Electrophilic Aromatic Substitution Reactions An organic reaction in which an electrophile substitutes a hydrogen atom in an aromatic
Electrophilic Aromatic Substitution Reactions, Course Notes Archive, 1 Electrophilic Aromatic Substitution Reactions An organic reaction in which an electrophile substitutes a hydrogen atom in an aromatic
CHE 232 - Organic Chemistry Exam 1, February 10, 2004
 CE 232 - rganic Chemistry Exam 1, February 10, 2004 ame Student ID o. Before you begin this exam: First: You are allowed to have a simple model set at your seat. Please put away all other materials. Second:
CE 232 - rganic Chemistry Exam 1, February 10, 2004 ame Student ID o. Before you begin this exam: First: You are allowed to have a simple model set at your seat. Please put away all other materials. Second:
Suggested solutions for Chapter 3
 s for Chapter PRBLEM Assuming that the molecular ion is the base peak (00% abundance) what peaks would appear in the mass spectrum of each of these molecules: (a) C5Br (b) C60 (c) C64Br In cases (a) and
s for Chapter PRBLEM Assuming that the molecular ion is the base peak (00% abundance) what peaks would appear in the mass spectrum of each of these molecules: (a) C5Br (b) C60 (c) C64Br In cases (a) and
Nucleophilic Substitution and Elimination
 Nucleophilic Substitution and Elimination What does the term "nucleophilic substitution" imply? A nucleophile is an the electron rich species that will react with an electron poor species A substitution
Nucleophilic Substitution and Elimination What does the term "nucleophilic substitution" imply? A nucleophile is an the electron rich species that will react with an electron poor species A substitution
EXPERIMENT 5: DIPEPTIDE RESEARCH PROJECT
 EXPERIMENT 5: DIPEPTIDE RESEARCH PROJECT Pre-Lab Questions: None. 64 I. Background Information DIPEPTIDE RESEARCH PROJECT Methods developed by organic chemists for the synthesis of biopolymers have had
EXPERIMENT 5: DIPEPTIDE RESEARCH PROJECT Pre-Lab Questions: None. 64 I. Background Information DIPEPTIDE RESEARCH PROJECT Methods developed by organic chemists for the synthesis of biopolymers have had
Ch17_PT MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Ch17_PT MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which molecule is a carboxylic acid? A) 1) B) C) D) E) CH3 CH2 CH2 NH2 2) Which molecule
Ch17_PT MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which molecule is a carboxylic acid? A) 1) B) C) D) E) CH3 CH2 CH2 NH2 2) Which molecule
Everything You Need to Know About Mechanisms. First rule: Arrows are used to indicate movement of electrons
 Everything You eed to Know About Mechanisms A) The orrect Use of Arrows to Indicate Electron Movement The ability to write an organic reaction mechanism properly is key to success in organic chemistry
Everything You eed to Know About Mechanisms A) The orrect Use of Arrows to Indicate Electron Movement The ability to write an organic reaction mechanism properly is key to success in organic chemistry
Unit Vocabulary: o Organic Acid o Alcohol. o Ester o Ether. o Amine o Aldehyde
 Unit Vocabulary: Addition rxn Esterification Polymer Alcohol Ether Polymerization Aldehyde Fermentation Primary Alkane Functional group Saponification Alkene Halide (halocarbon) Saturated hydrocarbon Alkyne
Unit Vocabulary: Addition rxn Esterification Polymer Alcohol Ether Polymerization Aldehyde Fermentation Primary Alkane Functional group Saponification Alkene Halide (halocarbon) Saturated hydrocarbon Alkyne
BIOLOGICAL MOLECULES OF LIFE
 BIOLOGICAL MOLECULES OF LIFE C A R B O H Y D R A T E S, L I P I D S, P R O T E I N S, A N D N U C L E I C A C I D S The Academic Support Center @ Daytona State College (Science 115, Page 1 of 29) Carbon
BIOLOGICAL MOLECULES OF LIFE C A R B O H Y D R A T E S, L I P I D S, P R O T E I N S, A N D N U C L E I C A C I D S The Academic Support Center @ Daytona State College (Science 115, Page 1 of 29) Carbon
Electrophilic Addition Reactions
 Electrophilic Addition Reactions Electrophilic addition reactions are an important class of reactions that allow the interconversion of C=C and C C into a range of important functional groups. Conceptually,
Electrophilic Addition Reactions Electrophilic addition reactions are an important class of reactions that allow the interconversion of C=C and C C into a range of important functional groups. Conceptually,
2. Couple the two protected amino acids.
 General Considerations The Strategy of Peptide Synthesis Making peptide bonds between amino acids is not difficult. The challenge is connecting amino acids in the correct sequence. andom peptide bond formation
General Considerations The Strategy of Peptide Synthesis Making peptide bonds between amino acids is not difficult. The challenge is connecting amino acids in the correct sequence. andom peptide bond formation
Reactions of Fats and Fatty Acids
 Reactions of Fats and Fatty Acids Outline Fats and Oils Fatty Acid Biosynthesis Biodiesel Homework We hear quite a lot about the place of fats and oils in human nutrition. Foods high in fat are at the
Reactions of Fats and Fatty Acids Outline Fats and Oils Fatty Acid Biosynthesis Biodiesel Homework We hear quite a lot about the place of fats and oils in human nutrition. Foods high in fat are at the
Chemistry Notes for class 12 Chapter 13 Amines
 1 P a g e Chemistry Notes for class 12 Chapter 13 Amines Amines constitute an important class of organic compounds derived by replacing one or more hydrogen atoms ofnh 3 molecule by alkyl/aryl group(s).
1 P a g e Chemistry Notes for class 12 Chapter 13 Amines Amines constitute an important class of organic compounds derived by replacing one or more hydrogen atoms ofnh 3 molecule by alkyl/aryl group(s).
REACTIONS OF AROMATIC COMPOUNDS
 A STUDENT SHOULD BE ABLE TO: REACTIONS OF AROMATIC COMPOUNDS 1. Predict the product(s) of Electrophilic Aromatic Substitution (EAS), Nucleophilic Aromatic Substitution (S N Ar) and Elimination-Addition
A STUDENT SHOULD BE ABLE TO: REACTIONS OF AROMATIC COMPOUNDS 1. Predict the product(s) of Electrophilic Aromatic Substitution (EAS), Nucleophilic Aromatic Substitution (S N Ar) and Elimination-Addition
22.7 ALKYLATION OF ESTER ENOLATE IONS
 1084 CHAPTER THE CHEMITRY F ENLATE IN, ENL, AND a,b-unaturated CARBNYL CMPUND H H CA CL CoA + enol form of acetyl-coa _ C N NH acetyl-coa carboxylase H H R H carboxybiotin HN NH _ LC LCH LCLCoA + H H malonyl-coa
1084 CHAPTER THE CHEMITRY F ENLATE IN, ENL, AND a,b-unaturated CARBNYL CMPUND H H CA CL CoA + enol form of acetyl-coa _ C N NH acetyl-coa carboxylase H H R H carboxybiotin HN NH _ LC LCH LCLCoA + H H malonyl-coa
Assessment Schedule 2013 Chemistry: Demonstrate understanding of the properties of organic compounds (91391)
 NCEA Level 3 Chemistry (91391) 2013 page 1 of 8 Assessment Schedule 2013 Chemistry: Demonstrate understanding of the properties of organic compounds (91391) Evidence Statement Q Evidence Achievement Achievement
NCEA Level 3 Chemistry (91391) 2013 page 1 of 8 Assessment Schedule 2013 Chemistry: Demonstrate understanding of the properties of organic compounds (91391) Evidence Statement Q Evidence Achievement Achievement
How To Understand The Chemistry Of Organic Molecules
 CHAPTER 3 THE CHEMISTRY OF ORGANIC MOLECULES 3.1 Organic Molecules The chemistry of carbon accounts for the diversity of organic molecules found in living things. Carbon has six electrons, four of which
CHAPTER 3 THE CHEMISTRY OF ORGANIC MOLECULES 3.1 Organic Molecules The chemistry of carbon accounts for the diversity of organic molecules found in living things. Carbon has six electrons, four of which
Avg. 16.4 / 25 Stnd. Dev. 8.2
 QUIZ TREE Avg. 16.4 / 25 Stnd. Dev. 8.2 xidation of Alcohols with Chromium (VI): Jones xidation 2 Alcohols are oxidized by a solution of chromium trioxide in aqueous acetone (2), in the presence of an
QUIZ TREE Avg. 16.4 / 25 Stnd. Dev. 8.2 xidation of Alcohols with Chromium (VI): Jones xidation 2 Alcohols are oxidized by a solution of chromium trioxide in aqueous acetone (2), in the presence of an
Page 1. 6. Which hydrocarbon is a member of the alkane series? (1) 1. Which is the structural formula of methane? (1) (2) (2) (3) (3) (4) (4)
 1. Which is the structural formula of methane? 6. Which hydrocarbon is a member of the alkane series? 7. How many carbon atoms are contained in an ethyl group? 1 3 2 4 2. In the alkane series, each molecule
1. Which is the structural formula of methane? 6. Which hydrocarbon is a member of the alkane series? 7. How many carbon atoms are contained in an ethyl group? 1 3 2 4 2. In the alkane series, each molecule
2. Rank the following three compounds in decreasing order of basicity. O NHCCH 3 NH 2
 1. To convert a nitrile to a primary amine you must: A) hydrolyze it with water. B) oxidize it with chromic acid. C) reduce it with hydrogen or lithium aluminum hydride. D) substitute it with an alkyl
1. To convert a nitrile to a primary amine you must: A) hydrolyze it with water. B) oxidize it with chromic acid. C) reduce it with hydrogen or lithium aluminum hydride. D) substitute it with an alkyl
ORGANIC CHEMISTRY I PRACTICE EXERCISE Sn1 and Sn2 Reactions
 ORGANIC CEMISTRY I PRACTICE EXERCISE Sn1 and Sn2 Reactions 1) Which of the following best represents the carbon-chlorine bond of methyl chloride? d d - d - d d d d - d - I II III IV V 2) Provide a detailed,
ORGANIC CEMISTRY I PRACTICE EXERCISE Sn1 and Sn2 Reactions 1) Which of the following best represents the carbon-chlorine bond of methyl chloride? d d - d - d d d d - d - I II III IV V 2) Provide a detailed,
SULFONATE AND INORGANIC ESTER DERIVATIVES OF ALCOHOLS
 0. ULFNATE AND INRGANIC ETER DERIVATIVE F ALCL 44 R 2 C L CR 2 carbocation Lewis acid base association X (halide ion) 2 $ R 2 C L CR 2 R R X C A C $ alkyl halide R R alkene $ $ Brønsted acid base reaction
0. ULFNATE AND INRGANIC ETER DERIVATIVE F ALCL 44 R 2 C L CR 2 carbocation Lewis acid base association X (halide ion) 2 $ R 2 C L CR 2 R R X C A C $ alkyl halide R R alkene $ $ Brønsted acid base reaction
EXPERIMENT 6 (Organic Chemistry II) Identification of Ketones and Aldehydes
 EXPERIMENT 6 (rganic hemistry II) Identification of Ketones and Aldehydes Pahlavan/herif hemicals 2,4-Dinitrophenylhydrazine(DNP) 0.10 M AgN 3 2-propanol Benzaldehyde 10% Na 2-butanol Methyl ethyl Ketone
EXPERIMENT 6 (rganic hemistry II) Identification of Ketones and Aldehydes Pahlavan/herif hemicals 2,4-Dinitrophenylhydrazine(DNP) 0.10 M AgN 3 2-propanol Benzaldehyde 10% Na 2-butanol Methyl ethyl Ketone
Chapter 5 Classification of Organic Compounds by Solubility
 Chapter 5 Classification of Organic Compounds by Solubility Deductions based upon interpretation of simple solubility tests can be extremely useful in organic structure determination. Both solubility and
Chapter 5 Classification of Organic Compounds by Solubility Deductions based upon interpretation of simple solubility tests can be extremely useful in organic structure determination. Both solubility and
UNIT (9) CARBOXYLIC ACIDS, ESTERS, AMINES, AND AMIDES
 UNIT (9) CARBXYLIC ACIDS, ESTERS, AMINES, AND AMIDES 9.1 Carboxylic Acids The functional group in carboxylic acids is called the carboxyl group. A carboxyl group is a carbonyl group (C = ) with a hydroxyl
UNIT (9) CARBXYLIC ACIDS, ESTERS, AMINES, AND AMIDES 9.1 Carboxylic Acids The functional group in carboxylic acids is called the carboxyl group. A carboxyl group is a carbonyl group (C = ) with a hydroxyl
Suggested solutions for Chapter 7
 s for Chapter 7 7 PRBLEM 1 Are these molecules conjugated? Explain your answer in any reasonable way. C Et C Et C Et Revision of the basic kinds of conjugation and how to show conjugation with curly arrows.
s for Chapter 7 7 PRBLEM 1 Are these molecules conjugated? Explain your answer in any reasonable way. C Et C Et C Et Revision of the basic kinds of conjugation and how to show conjugation with curly arrows.
Chapter 13 Carboxylic Acids, Esters, Amines, and Amides. Carboxylic Acids. Names and Sources of Some Carboxylic Acids. IUPAC Names
 Chapter 13 Carboxylic Acids, Esters, Amines, and Amides 13.1 Carboxylic Acids Carboxylic Acids A carboxylic acid contains a carboxyl group, which is a carbonyl group (C=) attached to a hydroxyl group (
Chapter 13 Carboxylic Acids, Esters, Amines, and Amides 13.1 Carboxylic Acids Carboxylic Acids A carboxylic acid contains a carboxyl group, which is a carbonyl group (C=) attached to a hydroxyl group (
Chapter 12 Organic Compounds with Oxygen and Sulfur
 Chapter 12 Organic Compounds with Oxygen and Sulfur 1 Alcohols An alcohol contains a hydroxyl group ( OH) that replaces a hydrogen atom in a hydrocarbon. A phenol contains a hydroxyl group ( OH) attached
Chapter 12 Organic Compounds with Oxygen and Sulfur 1 Alcohols An alcohol contains a hydroxyl group ( OH) that replaces a hydrogen atom in a hydrocarbon. A phenol contains a hydroxyl group ( OH) attached
1. What is the hybridization of the indicated atom in the following molecule?
 Practice Final Exam, Chemistry 2210, rganic Chem I 1. What is the hybridization of the indicated atom in the following molecule? A. sp 3 B. sp 2 C. sp D. not hybridized 2. Name the functional groups in
Practice Final Exam, Chemistry 2210, rganic Chem I 1. What is the hybridization of the indicated atom in the following molecule? A. sp 3 B. sp 2 C. sp D. not hybridized 2. Name the functional groups in
NOMENCLATURE OF ORGANIC COMPOUNDS 2010, 2003, 1980, by David A. Katz. All rights reserved.
 NMENCLATURE F RGANIC CMPUNDS 2010, 2003, 1980, by David A. Katz. All rights reserved. rganic chemistry is the chemistry of carbon compounds. Carbon has the ability to bond with itself to form long chains
NMENCLATURE F RGANIC CMPUNDS 2010, 2003, 1980, by David A. Katz. All rights reserved. rganic chemistry is the chemistry of carbon compounds. Carbon has the ability to bond with itself to form long chains
Course Prerequisite: Chemistry 141 or 143.
 Instructor: Matthias Brewer; Office: Cook A316; email: Matthias.Brewer@uvm.edu BlackBoard Site: bb.uvm.edu Lecture: 10:40am 11:30am MWF, Angell B106 Review Sessions: 5:30pm Thur., Angell B106 Laboratory
Instructor: Matthias Brewer; Office: Cook A316; email: Matthias.Brewer@uvm.edu BlackBoard Site: bb.uvm.edu Lecture: 10:40am 11:30am MWF, Angell B106 Review Sessions: 5:30pm Thur., Angell B106 Laboratory
Conjugation is broken completely by the introduction of saturated (sp3) carbon:
 Chapter 16 Conjugation, resonance, and dienes Conjugation relies on the partial overlap of p-orbitals on adjacent double or triple bonds. A common conjugated system involves 1,3-dienes, such as 1,3-butadiene.
Chapter 16 Conjugation, resonance, and dienes Conjugation relies on the partial overlap of p-orbitals on adjacent double or triple bonds. A common conjugated system involves 1,3-dienes, such as 1,3-butadiene.
3. Semester Completion date: May 9, 2016. a. You can finish early, and you can start early (or late), but you MUST FINISH BY MAY 9
 4 Dates, Flexible Schedules: Go-At-Your-Own-Pace Asynchronous. 1. FLEXIBILITY. You can schedule your own test dates (so long as you finish all by May 9, 2016) 2. The Official semester start date is January
4 Dates, Flexible Schedules: Go-At-Your-Own-Pace Asynchronous. 1. FLEXIBILITY. You can schedule your own test dates (so long as you finish all by May 9, 2016) 2. The Official semester start date is January
Synthesis of Isopentyl Acetate
 Experiment 8 Synthesis of Isopentyl Acetate Objectives To prepare isopentyl acetate from isopentyl alcohol and acetic acid by the Fischer esterification reaction. Introduction Esters are derivatives of
Experiment 8 Synthesis of Isopentyl Acetate Objectives To prepare isopentyl acetate from isopentyl alcohol and acetic acid by the Fischer esterification reaction. Introduction Esters are derivatives of
Chapter 3 Molecules of Cells
 Bio 100 Molecules of cells 1 Chapter 3 Molecules of Cells Compounds containing carbon are called organic compounds Molecules such as methane that are only composed of carbon and hydrogen are called hydrocarbons
Bio 100 Molecules of cells 1 Chapter 3 Molecules of Cells Compounds containing carbon are called organic compounds Molecules such as methane that are only composed of carbon and hydrogen are called hydrocarbons
Chapter 15 Radical Reactions. Radicals are reactive species with a single unpaired electron, formed by
 Chapter 15 Radical Reactions Radicals are reactive species with a single unpaired electron, formed by homolysis of a covalent bond; a radical contains an atom that does not have an octet of electrons,
Chapter 15 Radical Reactions Radicals are reactive species with a single unpaired electron, formed by homolysis of a covalent bond; a radical contains an atom that does not have an octet of electrons,
3.4 BRØNSTED LOWRY ACIDS AND BASES
 96 CAPTER 3 ACIDS AND BASES. TE CURVED-ARROW NOTATION and that the unshared electron pair (and negative charge) is shared equally by the two terminal carbons. C L C A C 1 allyl anion (c) Using the curved-arrow
96 CAPTER 3 ACIDS AND BASES. TE CURVED-ARROW NOTATION and that the unshared electron pair (and negative charge) is shared equally by the two terminal carbons. C L C A C 1 allyl anion (c) Using the curved-arrow
Figure 8. Example of simple benzene naming with chlorine and NO 2 as substituents.
 BENZENE NAMING EXPLAINED. This was excerpted from CHEM WIKI and is used with appreciation to the authors. http://chemwiki.ucdavis.edu/organic_chemistry/hydrocarbons/aromatics/naming_the_benzenes. Simple
BENZENE NAMING EXPLAINED. This was excerpted from CHEM WIKI and is used with appreciation to the authors. http://chemwiki.ucdavis.edu/organic_chemistry/hydrocarbons/aromatics/naming_the_benzenes. Simple
Experiment #8 properties of Alcohols and Phenols
 Introduction Experiment #8 properties of Alcohols and Phenols As has been mentioned before, over 20 million organic compounds have been identified. If each substance had to be studied as an entity completely
Introduction Experiment #8 properties of Alcohols and Phenols As has been mentioned before, over 20 million organic compounds have been identified. If each substance had to be studied as an entity completely
17.2 REACTIONS INVOLVING ALLYLIC AND BENZYLIC RADICALS
 17. REACTINS INVLVING ALLYLIC AND BENZYLIC RADICALS 793 As Eq. 17. shows, the products derived from the reaction of water at the ring carbons are not formed. The reason is that these products are not aromatic
17. REACTINS INVLVING ALLYLIC AND BENZYLIC RADICALS 793 As Eq. 17. shows, the products derived from the reaction of water at the ring carbons are not formed. The reason is that these products are not aromatic
Name: Hour: Elements & Macromolecules in Organisms
 Name: Hour: Elements & Macromolecules in Organisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds
Name: Hour: Elements & Macromolecules in Organisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds
Biochemistry of Cells
 Biochemistry of Cells 1 Carbon-based Molecules Although a cell is mostly water, the rest of the cell consists mostly of carbon-based molecules Organic chemistry is the study of carbon compounds Carbon
Biochemistry of Cells 1 Carbon-based Molecules Although a cell is mostly water, the rest of the cell consists mostly of carbon-based molecules Organic chemistry is the study of carbon compounds Carbon
Chapter 5. The Structure and Function of Macromolecule s
 Chapter 5 The Structure and Function of Macromolecule s Most Macromolecules are polymers: Polymer: (poly: many; mer: part) Large molecules consisting of many identical or similar subunits connected together.
Chapter 5 The Structure and Function of Macromolecule s Most Macromolecules are polymers: Polymer: (poly: many; mer: part) Large molecules consisting of many identical or similar subunits connected together.
Aromaticity and Reactions of Benzene
 Aromaticity and eactions of Benzene ark College Benzene is a unique molecule it is highly unsaturated with 6 carbons and 6 hydrogens, it is planar, and has a high degree of symmetry. These features explain
Aromaticity and eactions of Benzene ark College Benzene is a unique molecule it is highly unsaturated with 6 carbons and 6 hydrogens, it is planar, and has a high degree of symmetry. These features explain
Experiment 11. Infrared Spectroscopy
 Chem 22 Spring 2010 Experiment 11 Infrared Spectroscopy Pre-lab preparation. (1) In Ch 5 and 12 of the text you will find examples of the most common functional groups in organic molecules. In your notebook,
Chem 22 Spring 2010 Experiment 11 Infrared Spectroscopy Pre-lab preparation. (1) In Ch 5 and 12 of the text you will find examples of the most common functional groups in organic molecules. In your notebook,
Chemistry 1110 Organic Chemistry IUPAC Nomenclature
 hemistry 1110 rganic hemistry IUPA Nomenclature 1 f the approximately 32 million unique chemical compounds presently known, over 95% of them can be classified as organic; i.e., containing carbon. The IUPA
hemistry 1110 rganic hemistry IUPA Nomenclature 1 f the approximately 32 million unique chemical compounds presently known, over 95% of them can be classified as organic; i.e., containing carbon. The IUPA
Final Examination, Organic Chemistry 1 (CHEM 2210) December 2000 Version *A* A. B. C. D.
 Final Examination, rganic hemistry 1 (EM 2210) December 2000 Version *A* 1. What are the hybridization of, and the geometrical shape around, the nitrogen atom in the following molecule? N 3 3 A. sp, linear
Final Examination, rganic hemistry 1 (EM 2210) December 2000 Version *A* 1. What are the hybridization of, and the geometrical shape around, the nitrogen atom in the following molecule? N 3 3 A. sp, linear
Willem Elbers. October 9, 2015
 S N 1 and S N 2 reactivity of 3 alkyl bromides Willem Elbers ctober 9, 2015 1 Abstract n this experiment, we investigate the relative reactivities of three alkyl bromides with increasing steric bulk. We
S N 1 and S N 2 reactivity of 3 alkyl bromides Willem Elbers ctober 9, 2015 1 Abstract n this experiment, we investigate the relative reactivities of three alkyl bromides with increasing steric bulk. We
Carboxylic Acid Structure and Chemistry: Part 2
 Principles of Drug Action 1, pring 2005, Carboxylic Acids Part 2 Carboxylic Acid tructure and Chemistry: Part 2 Jack Deuiter IV. eactions of the Carboxylic Acid eactions Depending on their overall structure,
Principles of Drug Action 1, pring 2005, Carboxylic Acids Part 2 Carboxylic Acid tructure and Chemistry: Part 2 Jack Deuiter IV. eactions of the Carboxylic Acid eactions Depending on their overall structure,
MCAT Organic Chemistry - Problem Drill 23: Amino Acids, Peptides and Proteins
 MCAT rganic Chemistry - Problem Drill 23: Amino Acids, Peptides and Proteins Question No. 1 of 10 Question 1. Which amino acid does not contain a chiral center? Question #01 (A) Serine (B) Proline (C)
MCAT rganic Chemistry - Problem Drill 23: Amino Acids, Peptides and Proteins Question No. 1 of 10 Question 1. Which amino acid does not contain a chiral center? Question #01 (A) Serine (B) Proline (C)
Molecules, Compounds, and Chemical Equations (Chapter 3)
 Molecules, Compounds, and Chemical Equations (Chapter 3) Chemical Compounds 1. Classification of Elements and Compounds Types of Pure Substances (Figure 3.4) Elements -- made up of only one type of atom
Molecules, Compounds, and Chemical Equations (Chapter 3) Chemical Compounds 1. Classification of Elements and Compounds Types of Pure Substances (Figure 3.4) Elements -- made up of only one type of atom
These instructions are for a classroom activity which supports OCR A Level Chemistry A.
 Lesson Element Keyword activities Instructions for teachers These instructions are for a classroom activity which supports OCR A Level Chemistry A. Just a minute! To run this activity you will need a set
Lesson Element Keyword activities Instructions for teachers These instructions are for a classroom activity which supports OCR A Level Chemistry A. Just a minute! To run this activity you will need a set
Chapter 2 Polar Covalent Bonds; Acids and Bases
 John E. McMurry http://www.cengage.com/chemistry/mcmurry Chapter 2 Polar Covalent Bonds; Acids and Bases Javier E. Horta, M.D., Ph.D. University of Massachusetts Lowell Polar Covalent Bonds: Electronegativity
John E. McMurry http://www.cengage.com/chemistry/mcmurry Chapter 2 Polar Covalent Bonds; Acids and Bases Javier E. Horta, M.D., Ph.D. University of Massachusetts Lowell Polar Covalent Bonds: Electronegativity
Resonance Structures Arrow Pushing Practice
 Resonance Structures Arrow Pushing Practice The following is a collection of ions and neutral molecules for which several resonance structures can be drawn. For the ions, the charges can be delocalized
Resonance Structures Arrow Pushing Practice The following is a collection of ions and neutral molecules for which several resonance structures can be drawn. For the ions, the charges can be delocalized
Reactions of Aldehydes and Ketones
 Reactions of Aldehydes and Ketones Structure Deduction using lassification Tests 1 Determination of Structure Determining the structure of an unknown organic compound is an exercise in deductive reasoning.
Reactions of Aldehydes and Ketones Structure Deduction using lassification Tests 1 Determination of Structure Determining the structure of an unknown organic compound is an exercise in deductive reasoning.
Austin Peay State University Department of Chemistry CHEM 1021 TESTING FOR ORGANIC FUNCTIONAL GROUPS
 TESTING FOR ORGANIC FUNCTIONAL GROUPS Caution: Chromic acid is hazardous as are many of the organic substances in today s experiment. Treat all unknowns with extreme care. Many organic substances are flammable.
TESTING FOR ORGANIC FUNCTIONAL GROUPS Caution: Chromic acid is hazardous as are many of the organic substances in today s experiment. Treat all unknowns with extreme care. Many organic substances are flammable.
CHEM 121. Chapter 18. Name: Date: 1. Which of the following compounds is both an aldose and a hexose? A) Page 1
 CEM 121. Chapter 18. Name: Date: 1. Which of the following compounds is both an aldose and a hexose? A) B) C) D) Page 1 2. Which of the following structures is that of an L-monosaccharide? A) B) C) D)
CEM 121. Chapter 18. Name: Date: 1. Which of the following compounds is both an aldose and a hexose? A) B) C) D) Page 1 2. Which of the following structures is that of an L-monosaccharide? A) B) C) D)
Please read and sign the Honor Code statement below:
 CHEM 3311 Exam #1 Name Dr. Minger June 8, 2015 Please read and sign the Honor Code statement below: I pledge that on my honor, as a University of Colorado at Boulder student, I have neither given nor received
CHEM 3311 Exam #1 Name Dr. Minger June 8, 2015 Please read and sign the Honor Code statement below: I pledge that on my honor, as a University of Colorado at Boulder student, I have neither given nor received
Solid Phase Peptide Synthesis
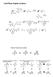 Solid Phase Peptide Synthesis Fischer Projections revisited C C C Remember that group rotations are always allowed C C 80 rotations of entire molecule are allowed C 80 C 90 rotations of entire molecule
Solid Phase Peptide Synthesis Fischer Projections revisited C C C Remember that group rotations are always allowed C C 80 rotations of entire molecule are allowed C 80 C 90 rotations of entire molecule
Alcohols An alcohol contains a hydroxyl group ( OH) attached to a carbon chain. A phenol contains a hydroxyl group ( OH) attached to a benzene ring.
 Chapter : rganic Compounds with xygen Alcohols, Ethers Alcohols An alcohol contains a hydroxyl group ( H) attached to a carbon chain. A phenol contains a hydroxyl group ( H) attached to a benzene ring.
Chapter : rganic Compounds with xygen Alcohols, Ethers Alcohols An alcohol contains a hydroxyl group ( H) attached to a carbon chain. A phenol contains a hydroxyl group ( H) attached to a benzene ring.
21.9 REDUCTION OF CARBOXYLIC ACID DERIVATIVES
 10 APTER 1 TE EMITRY F ARBXYLI AID DERIVATIVE TUDY GUIDE LIK 1.5 Esters and ucleophiles 1.17 Give the structure of the product in the reaction of each of the following esters with isotopically labeled
10 APTER 1 TE EMITRY F ARBXYLI AID DERIVATIVE TUDY GUIDE LIK 1.5 Esters and ucleophiles 1.17 Give the structure of the product in the reaction of each of the following esters with isotopically labeled
