7.1 Atoms and Isotopes
|
|
|
- Brianna Harrison
- 9 years ago
- Views:
Transcription
1 7.1 Atoms and Isotopes The history of atomic discovery begins with the ancient Greeks, when, around 400 BCE, philosopher Democritus asserted that all material things are composed of extremely small irreducible particles called atoms. His theory was rejected and ignored for almost 2000 years. John Dalton resurrected the atomic theory of matter in the early nineteenth century by characterizing elements by their atomic structure and weight. Over the next hundred years or so, scientists continued to refine their understanding of the atom, until the advances of Niels Bohr and Ernest Rutherford. Bohr Rutherford Model of the Atom Niels Bohr, a Danish scientist, and Ernest Rutherford, a New Zealand scientist, are credited with a number of discoveries that led to the development of the Bohr Rutherford atomic model. Rutherford found that when a beam of positively charged particles was fired at a thin gold foil, most particles passed through the foil, as expected, but some were scattered in all directions. To explain this, it was proposed that the atom consists of a dense, positively charged nucleus surrounded by tiny negatively charged electrons and a relatively vast region of empty space. Bohr also discovered that the electrons could only occupy certain energy levels. When these discoveries were combined, the Bohr Rutherford model of the atom was created. This model has the following key features: The dense nucleus contains the atom s protons and neutrons. The relatively tiny electrons orbit the nucleus. The electrons only occupy certain energy levels. Most of the atom consists of empty space. The model provides a visual method for describing the atomic structure of an element. The atomic structure refers to the number of protons, neutrons, and electrons in an atom and their organization within the atom. Simplified Bohr Rutherford diagrams for helium and fluorine are shown in Figure 1. 2p + 2n 0 9p + 10n 0 proton a positively charged particle in the neutron an uncharged particle in the nucleons particles in the nucleus of an atom; protons and neutrons electron a negatively charged particle found in the space surrounding the ground state state in which all electrons are at their lowest possible energy levels excited state state in which one or more electrons are at higher energy levels than in the ground state He (b) Figure 1 Bohr Rutherford diagrams for helium and (b) fl uorine. Notice that a helium atom has two protons, two neutrons, and two electrons. A fl uorine atom has nine protons, ten neutrons, and nine electrons. The nucleus is the centre of the atom and consists of protons and neutrons. A proton is a positively charged particle, and a neutron is an uncharged particle. In nuclear physics, neutrons and protons are often referred to collectively as nucleons. Protons and neutrons have approximately the same mass. An electron is a negatively charged particle that moves in the space surrounding the nucleus and is extremely small compared to nucleons. In general, an atom in its normal state has the same number of electrons as protons. In a Bohr Rutherford diagram, electrons are placed in the lower energy levels, or shells, first, until these shells are filled. Atoms that have electrons placed in this way are said to be in their ground state: the electrons are all at the lowest possible energy levels. An atom is said to be in an excited state if it absorbs energy that causes an electron to have more energy and move to a higher energy level. An excited atom returns F 318 Chapter 7 Nuclear Energy and Society
2 to its ground state by releasing energy as the electron drops back to its lowest available energy level. Figure 2 shows a hydrogen atom in the ground state and in an excited state. The first energy level is the innermost shell in the Bohr Rutherford diagram. 1p + 1p + H (b) H Figure 2 Hydrogen in an excited state and (b) its ground state According to the Bohr Rutherford model, each energy level or shell can hold a certain number of electrons. Table 1 gives the maximum number of electrons for each shell. Atomic Number, Mass Number, and the Periodic Table The periodic table of elements lists all the elements known today. It can be used to determine the atomic structure of an element (Figure 3). Table 1 Electron Distribution in a Bohr Rutherford Model Shell number Maximum number of electrons F fluorine atomic number chemical symbol mass number Figure 3 Identifying mass number and atomic number for the element fl uorine on the periodic table Th e atomic number is the number of protons in an atom of an element. Each element has a different number of protons. The mass number is equal to the number of nucleons in an atom. The periodic table entry shown in Figure 3 indicates that fluorine has nine protons and nine electrons. The number of neutrons is determined by subtracting the atomic number from the mass number: 19 nucleons 1mass number2 2 9 protons 1atomic number neutrons Isotopes Carbon-12 consists of six protons and six neutrons (Figure 4). Most naturally occurring carbon has this atomic structure. There is, however, another form of carbon called carbon-14. Carbon-14 has six protons and eight neutrons. Carbon-14 is a different isotope than carbon-12. Different isotopes of an element have the same number of protons, but different numbers of neutrons (Figure 5). The mass number of 14 indicates that an atom of carbon-14 has two more neutrons than an atom of carbon-12. number of neutrons 5 mass number 2 atomic number atomic number the number of protons in the nucleus mass number the number of protons and neutrons in the nucleus 12 6 C chemical mass number symbol atomic number Figure 4 The standard notation for carbon-12 isotope a form of an element that has the same atomic number, but a different mass number than all other forms of that element 6p + 6n 0 n n 5 6 6p + 8n 0 n n 5 8 carbon-12 carbon C (b) 14 6 C Figure 5 Bohr Rutherford models for carbon-12 and (b) carbon Atoms and Isotopes 319
3 Carbon-14 has some interesting properties that are useful to archaeologists and scientists. A process called carbon dating provides a reasonably accurate method to determine the age of fossils and objects made of things that were once alive. You will learn more about carbon dating in Section 7.3. Most samples of elements consist of a number of different isotopes, some occurring naturally and others produced in laboratories. The most common isotope of hydrogen has a nucleus consisting of only one proton. There are, however, two other isotopes of hydrogen. These isotopes are important in nuclear science, so they have been given their own names: deuterium and tritium. Deuterium, which has one proton and one neutron, is a naturally occurring substance. Tritium, which has one proton and two neutrons, is only produced as a by-product of human-made nuclear reactions. The periodic table identifies the most commonly occurring isotopes for each element. A more general table that lists atomic information for all known isotopes is called a chart of the nuclides. In the following Tutorial, you will draw Bohr Rutherford diagrams for various isotopes. LEarNiNg TIP Mass Numbers The mass numbers of most elements have decimal values associated with them. Carbon-12 has a mass of exactly 12 atomic units because it is the substance to which all other elements are compared by atomic mass. The mass number that appears in the periodic table for carbon is slightly higher than 12 because of the small amounts of carbon-14 that exist in nature. Tutorial 1 Constructing a Bohr Rutherford Diagram Sample Problem 1 Draw the Bohr Rutherford diagram for silicon-31. Step 1. Locate silicon on the periodic table. The chemical symbol for silicon is Si. Step 2. The mass of this isotope is given: 31. Use a periodic table to identify the atomic number. The atomic number is 14. Step 3. Since the atomic number is 14, there are 14 protons and 14 electrons. The number of neutrons is found by subtracting the atomic number from the mass number: There are 17 neutrons in an atom of silicon. Use this information to draw a Bohr Rutherford diagram for silicon (Figure 6). 14p + 17n 0 Si Figure 6 Practice radioisotope an unstable isotope that spontaneously changes its nuclear structure and releases energy in the form of radiation radiation energy released when the nucleus of an unstable isotope undergoes a change in structure 1. Sketch a Bohr Rutherford diagram for each element. T/I C aluminum, Al (mass number 28) (b) silver, Ag (mass number 110) (c) two other elements of your choice from the periodic table or Appendix B (page 662) Some isotopes, called radioisotopes, are unstable; that is, they spontaneously change their nuclear structure. Radiation is energy released in the form of waves when a radioisotope undergoes a structural change. This radiation can be harmful if not properly controlled. In some cases, however, these radioisotopes are beneficial. You will examine the process by which isotopes spontaneously change later in this chapter. 320 Chapter 7 Nuclear Energy and Society
4 Medical Applications of Radioisotopes Nuclear medical imaging is a diagnostic technique that involves injecting a patient with a small dose of a radioisotope, such as technetium-99m. These materials, sometimes called radioactive tracers, emit radiation that can be detected and converted into an image. By comparing radiation patterns of an unhealthy organ to those of a healthy one, doctors are better able to pinpoint a malignancy, or tumour (Figure 7). One of the advantages of nuclear imaging over traditional X-rays is that it provides a detailed account of both hard tissues like bone and softer tissues like the liver and kidneys. X-rays are primarily useful for detecting bone fractures. Figure 7 A radioactive tracer provides a detailed image of a diseased organ. This image is detecting the spread of lung cancer to the skeleton. Research This Technetium-99m Skills: Researching, Analyzing, Communicating Technetium-99m is an unusual isotope for which medical scientists have discovered several important uses. The m in its name identifi es it as a meta-stable isotope. 1. Research Technetium-99m (Tc-99m) on the Internet or at the library. Write a brief report of your fi ndings that includes answers to the following questions: A. What is a meta-stable isotope? T/I B. How is Tc-99m obtained? T/I SKILLS HANDBOOK A5.1 C. What is it about Tc-99m that makes it particularly useful in medicine? T/I D. Discuss some of the applications of Tc-99m in the medical A fi eld. E. Are there any drawbacks to using nuclear imaging, such as A health risks, costs, or wait times? go to son science Medical Treatments One of the earliest medical applications of radioisotopes began in the 1950s, when iodine-131 was used to diagnose and treat thyroid disease. Sufferers of hyperthyroidism have an overactive thyroid gland: it releases more thyroid hormone than the body requires. Iodine-131 can be used to both identify a diseased thyroid gland and halt production of the hormone. Radionuclide therapy (RNT) is a rapidly growing medical field in which the properties of certain radioactive substances are used to treat various ailments. RNT is currently used to treat certain types of tumours, bone pain, and other conditions. In cancer treatments, the fundamental idea behind RNT is to bombard rapidly dividing harmful cells with radiation. These cells tend to absorb the radiation, which prevents them from dividing further. career link A radiation therapist works with doctors, other medical staff, and patients to design and administer radiation health treatment plans. To learn more about careers in radiation therapy and related fi elds, go to son science 7.1 Atoms and Isotopes 321
5 7.1 Summary The Bohr Rutherford model of the atom illustrates the atomic structure of an element. You can identify the number of protons, neutrons, and electrons of an element from its Bohr Rutherford model. You can identify the mass number and atomic number of an element from the periodic table. Isotopes of an element have the same number of protons but different numbers of neutrons. Radioactive isotopes are unstable and will spontaneously undergo a change in their nuclear structure. Some radioactive isotopes have useful applications, such as medical diagnosis and therapy. 7.1 Questions 1. Draw a Bohr Rutherford diagram for each isotope. K/U C oxygen-16 (b) potassium Draw Bohr Rutherford diagrams for hydrogen, deuterium, and tritium. (b) Identify their similarities and differences. K/U C 3. For each Bohr Rutherford model shown in Figure 8, determine the atomic number and the mass number (b) write the chemical name of the isotope K/U (b) Describe how each isotope compares with its most commonly occurring isotope. K/U C 6. Identify each isotope shown in Figure 9 given its Bohr Rutherford diagram. K/U 14p + 14n 0 10p + 12n 0 (i) Figure 8 7p + 13p + 5n 0 13n 0 (ii) 4. Draw a Bohr Rutherford diagram for each isotope of beryllium. (i) 7 4 Be (ii) 9 4 Be (iii) 11 4 Be (b) Explain the similarities and differences between these models. (c) Which isotope of beryllium is the most common in nature? Explain how you know. K/U C 5. Draw a Bohr Rutherford model for each isotope. (i) lithium-5, 5 3Li (ii) oxygen-20, 20 8O Figure 9 (ii) (b) 7. An isotope has 16 protons and 22 neutrons. Identify the element. K/U 8. Draw a Bohr Rutherford model for each isotope of argon. (i) Ar-40 (ii) Ar-44 (iii) Ar-47 (b) Explain how these isotopes are (i) alike (ii) different K/U C 9. Neon has three stable isotopes: Ne-20, Ne-21, and Ne-22. K/U C Draw a Bohr Rutherford model for each isotope. (b) How are these models alike? How are they different? 10. Draw Bohr Rutherford models for lithium-10, beryllium-10, and boron-10. (b) How are these models alike? How are they different? K/U C 322 Chapter 7 Nuclear Energy and Society
SCH 3UI Unit 2 Outline Up to Quiz #1 Atomic Theory and the Periodic Table
 Lesson Topics Covered SCH 3UI Unit 2 Outline Up to Quiz #1 Atomic Theory and the Periodic Table 1 Note: History of Atomic Theory progression of understanding of composition of matter; ancient Greeks and
Lesson Topics Covered SCH 3UI Unit 2 Outline Up to Quiz #1 Atomic Theory and the Periodic Table 1 Note: History of Atomic Theory progression of understanding of composition of matter; ancient Greeks and
NOTES ON The Structure of the Atom
 NOTES ON The Structure of the Atom Chemistry is the study of matter and its properties. Those properties can be explained by examining the atoms that compose the matter. An atom is the smallest particle
NOTES ON The Structure of the Atom Chemistry is the study of matter and its properties. Those properties can be explained by examining the atoms that compose the matter. An atom is the smallest particle
6.7: Explaining the Periodic Table pg. 234
 Unit C: Atoms, elements, and Compounds 6.7: Explaining the Periodic Table pg. 234 Key Concepts: 3. Elements are organized according to their atomic number and electron arrangement on the periodic table.
Unit C: Atoms, elements, and Compounds 6.7: Explaining the Periodic Table pg. 234 Key Concepts: 3. Elements are organized according to their atomic number and electron arrangement on the periodic table.
9/13/2013. However, Dalton thought that an atom was just a tiny sphere with no internal parts. This is sometimes referred to as the cannonball model.
 John Dalton was an English scientist who lived in the early 1800s. Dalton s atomic theory served as a model for how matter worked. The principles of Dalton s atomic theory are: 1. Elements are made of
John Dalton was an English scientist who lived in the early 1800s. Dalton s atomic theory served as a model for how matter worked. The principles of Dalton s atomic theory are: 1. Elements are made of
Atomic Calculations. 2.1 Composition of the Atom. number of protons + number of neutrons = mass number
 2.1 Composition of the Atom Atomic Calculations number of protons + number of neutrons = mass number number of neutrons = mass number - number of protons number of protons = number of electrons IF positive
2.1 Composition of the Atom Atomic Calculations number of protons + number of neutrons = mass number number of neutrons = mass number - number of protons number of protons = number of electrons IF positive
Chapter Five: Atomic Theory and Structure
 Chapter Five: Atomic Theory and Structure Evolution of Atomic Theory The ancient Greek scientist Democritus is often credited with developing the idea of the atom Democritus proposed that matter was, on
Chapter Five: Atomic Theory and Structure Evolution of Atomic Theory The ancient Greek scientist Democritus is often credited with developing the idea of the atom Democritus proposed that matter was, on
1. In the general symbol cleus, which of the three letters. 2. What is the mass number of an alpha particle?
 1. In the general symbol cleus, which of the three letters Z A X for a nu represents the atomic number? 2. What is the mass number of an alpha particle? 3. What is the mass number of a beta particle? 4.
1. In the general symbol cleus, which of the three letters Z A X for a nu represents the atomic number? 2. What is the mass number of an alpha particle? 3. What is the mass number of a beta particle? 4.
For convenience, we may consider an atom in two parts: the nucleus and the electrons.
 Atomic structure A. Introduction: In 1808, an English scientist called John Dalton proposed an atomic theory based on experimental findings. (1) Elements are made of extremely small particles called atoms.
Atomic structure A. Introduction: In 1808, an English scientist called John Dalton proposed an atomic theory based on experimental findings. (1) Elements are made of extremely small particles called atoms.
7.4. Using the Bohr Theory KNOW? Using the Bohr Theory to Describe Atoms and Ions
 7.4 Using the Bohr Theory LEARNING TIP Models such as Figures 1 to 4, on pages 218 and 219, help you visualize scientific explanations. As you examine Figures 1 to 4, look back and forth between the diagrams
7.4 Using the Bohr Theory LEARNING TIP Models such as Figures 1 to 4, on pages 218 and 219, help you visualize scientific explanations. As you examine Figures 1 to 4, look back and forth between the diagrams
CHEM 1411 Chapter 5 Homework Answers
 1 CHEM 1411 Chapter 5 Homework Answers 1. Which statement regarding the gold foil experiment is false? (a) It was performed by Rutherford and his research group early in the 20 th century. (b) Most of
1 CHEM 1411 Chapter 5 Homework Answers 1. Which statement regarding the gold foil experiment is false? (a) It was performed by Rutherford and his research group early in the 20 th century. (b) Most of
History of the Atom & Atomic Theory
 Chapter 5 History of the Atom & Atomic Theory You re invited to a Thinking Inside the Box Conference Each group should nominate a: o Leader o Writer o Presenter You have 5 minutes to come up with observations
Chapter 5 History of the Atom & Atomic Theory You re invited to a Thinking Inside the Box Conference Each group should nominate a: o Leader o Writer o Presenter You have 5 minutes to come up with observations
Chemistry CP Unit 2 Atomic Structure and Electron Configuration. Learning Targets (Your exam at the end of Unit 2 will assess the following:)
 Chemistry CP Unit 2 Atomic Structure and Electron Learning Targets (Your exam at the end of Unit 2 will assess the following:) 2. Atomic Structure and Electron 2-1. Give the one main contribution to the
Chemistry CP Unit 2 Atomic Structure and Electron Learning Targets (Your exam at the end of Unit 2 will assess the following:) 2. Atomic Structure and Electron 2-1. Give the one main contribution to the
5.1 Evolution of the Atomic Model
 5.1 Evolution of the Atomic Model Studying the atom has been a fascination of scientists for hundreds of years. Even Greek philosophers, over 2500 years ago, discussed the idea of there being a smallest
5.1 Evolution of the Atomic Model Studying the atom has been a fascination of scientists for hundreds of years. Even Greek philosophers, over 2500 years ago, discussed the idea of there being a smallest
2. John Dalton did his research work in which of the following countries? a. France b. Greece c. Russia d. England
 CHAPTER 3 1. Which combination of individual and contribution is not correct? a. Antoine Lavoisier - clarified confusion over cause of burning b. John Dalton - proposed atomic theory c. Marie Curie - discovered
CHAPTER 3 1. Which combination of individual and contribution is not correct? a. Antoine Lavoisier - clarified confusion over cause of burning b. John Dalton - proposed atomic theory c. Marie Curie - discovered
Atomic Structure OBJECTIVES SCHEDULE PREPARATION VOCABULARY MATERIALS. For each team of four. The students. For the class.
 activity 4 Atomic Structure OBJECTIVES Students are introduced to the structure of the atom and the nature of subatomic particles. The students are introduced to the properties of protons, neutrons, and
activity 4 Atomic Structure OBJECTIVES Students are introduced to the structure of the atom and the nature of subatomic particles. The students are introduced to the properties of protons, neutrons, and
ANSWER KEY : BUILD AN ATOM PART I: ATOM SCREEN Build an Atom simulation ( http://phet.colorado.edu/en/simulation/build an atom )
 ANSWER KEY : PART I: ATOM SCREEN Build an Atom simulation ( http://phet.colorado.edu/en/simulation/build an atom ) 1. Explore the Build an Atom simulation with your group. As you explore, talk about what
ANSWER KEY : PART I: ATOM SCREEN Build an Atom simulation ( http://phet.colorado.edu/en/simulation/build an atom ) 1. Explore the Build an Atom simulation with your group. As you explore, talk about what
Department of Physics and Geology The Elements and the Periodic Table
 Department of Physics and Geology The Elements and the Periodic Table Physical Science 1422 Equipment Needed Qty Periodic Table 1 Part 1: Background In 1869 a Russian chemistry professor named Dmitri Mendeleev
Department of Physics and Geology The Elements and the Periodic Table Physical Science 1422 Equipment Needed Qty Periodic Table 1 Part 1: Background In 1869 a Russian chemistry professor named Dmitri Mendeleev
Unit 3 Study Guide: Electron Configuration & The Periodic Table
 Name: Teacher s Name: Class: Block: Date: Unit 3 Study Guide: Electron Configuration & The Periodic Table 1. For each of the following elements, state whether the element is radioactive, synthetic or both.
Name: Teacher s Name: Class: Block: Date: Unit 3 Study Guide: Electron Configuration & The Periodic Table 1. For each of the following elements, state whether the element is radioactive, synthetic or both.
Instructors Guide: Atoms and Their Isotopes
 Instructors Guide: Atoms and Their Isotopes Standards Connections Connections to NSTA Standards for Science Teacher Preparation C.3.a.1 Fundamental structures of atoms and molecules. C.3.b.27 Applications
Instructors Guide: Atoms and Their Isotopes Standards Connections Connections to NSTA Standards for Science Teacher Preparation C.3.a.1 Fundamental structures of atoms and molecules. C.3.b.27 Applications
ATOMS A T O M S, I S O T O P E S, A N D I O N S. The Academic Support Center @ Daytona State College (Science 120, Page 1 of 39)
 ATOMS A T O M S, I S O T O P E S, A N D I O N S The Academic Support Center @ Daytona State College (Science 120, Page 1 of 39) THE ATOM All elements listed on the periodic table are made up of atoms.
ATOMS A T O M S, I S O T O P E S, A N D I O N S The Academic Support Center @ Daytona State College (Science 120, Page 1 of 39) THE ATOM All elements listed on the periodic table are made up of atoms.
Chapter 2 Atoms, Ions, and the Periodic Table
 Chapter 2 Atoms, Ions, and the Periodic Table 2.1 (a) neutron; (b) law of conservation of mass; (c) proton; (d) main-group element; (e) relative atomic mass; (f) mass number; (g) isotope; (h) cation; (i)
Chapter 2 Atoms, Ions, and the Periodic Table 2.1 (a) neutron; (b) law of conservation of mass; (c) proton; (d) main-group element; (e) relative atomic mass; (f) mass number; (g) isotope; (h) cation; (i)
Untitled Document. 1. Which of the following best describes an atom? 4. Which statement best describes the density of an atom s nucleus?
 Name: Date: 1. Which of the following best describes an atom? A. protons and electrons grouped together in a random pattern B. protons and electrons grouped together in an alternating pattern C. a core
Name: Date: 1. Which of the following best describes an atom? A. protons and electrons grouped together in a random pattern B. protons and electrons grouped together in an alternating pattern C. a core
The Models of the Atom
 The Models of the Atom All life, whether in the form of trees, whales, mushrooms, bacteria or amoebas, consists of cells. Similarly, all matter, whether in the form of aspirin, gold, vitamins, air or minerals,
The Models of the Atom All life, whether in the form of trees, whales, mushrooms, bacteria or amoebas, consists of cells. Similarly, all matter, whether in the form of aspirin, gold, vitamins, air or minerals,
2 The Structure of Atoms
 CHAPTER 4 2 The Structure of Atoms SECTION Atoms KEY IDEAS As you read this section, keep these questions in mind: What do atoms of the same element have in common? What are isotopes? How is an element
CHAPTER 4 2 The Structure of Atoms SECTION Atoms KEY IDEAS As you read this section, keep these questions in mind: What do atoms of the same element have in common? What are isotopes? How is an element
PROTONS AND ELECTRONS
 reflect Imagine that you have a bowl of oranges, bananas, pineapples, berries, pears, and watermelon. How do you identify each piece of fruit? Most likely, you are familiar with the characteristics of
reflect Imagine that you have a bowl of oranges, bananas, pineapples, berries, pears, and watermelon. How do you identify each piece of fruit? Most likely, you are familiar with the characteristics of
Objectives. PAM1014 Introduction to Radiation Physics. Constituents of Atoms. Atoms. Atoms. Atoms. Basic Atomic Theory
 PAM1014 Introduction to Radiation Physics Basic Atomic Theory Objectives Introduce and Molecules The periodic Table Electronic Energy Levels Atomic excitation & de-excitation Ionisation Molecules Constituents
PAM1014 Introduction to Radiation Physics Basic Atomic Theory Objectives Introduce and Molecules The periodic Table Electronic Energy Levels Atomic excitation & de-excitation Ionisation Molecules Constituents
Objectives 404 CHAPTER 9 RADIATION
 Objectives Explain the difference between isotopes of the same element. Describe the force that holds nucleons together. Explain the relationship between mass and energy according to Einstein s theory
Objectives Explain the difference between isotopes of the same element. Describe the force that holds nucleons together. Explain the relationship between mass and energy according to Einstein s theory
Level 3 Achievement Scale
 Unit 1: Atoms Level 3 Achievement Scale Can state the key results of the experiments associated with Dalton, Rutherford, Thomson, Chadwick, and Bohr and what this lead each to conclude. Can explain that
Unit 1: Atoms Level 3 Achievement Scale Can state the key results of the experiments associated with Dalton, Rutherford, Thomson, Chadwick, and Bohr and what this lead each to conclude. Can explain that
Name: Worksheet: Electron Configurations. I Heart Chemistry!
 1. Which electron configuration represents an atom in an excited state? 1s 2 2s 2 2p 6 3p 1 1s 2 2s 2 2p 6 3s 2 3p 2 1s 2 2s 2 2p 6 3s 2 3p 1 1s 2 2s 2 2p 6 3s 2 Worksheet: Electron Configurations Name:
1. Which electron configuration represents an atom in an excited state? 1s 2 2s 2 2p 6 3p 1 1s 2 2s 2 2p 6 3s 2 3p 2 1s 2 2s 2 2p 6 3s 2 3p 1 1s 2 2s 2 2p 6 3s 2 Worksheet: Electron Configurations Name:
Chapter NP-1. Nuclear Physics. Atomic Nature of Matter TABLE OF CONTENTS INTRODUCTION OBJECTIVES 1.0 PROPERTIES OF SUBSTANCES
 Chapter NP-1 Nuclear Physics Atomic Nature of Matter TABLE OF CONTENTS INTRODUCTION OBJECTIVES 1.0 PROPERTIES OF SUBSTANCES 1.1 CHEMICAL AND PHYSICAL PROPERTIES 2.0 COMPOSITION OF ATOMS 2.1 ATOMIC STRUCTURE
Chapter NP-1 Nuclear Physics Atomic Nature of Matter TABLE OF CONTENTS INTRODUCTION OBJECTIVES 1.0 PROPERTIES OF SUBSTANCES 1.1 CHEMICAL AND PHYSICAL PROPERTIES 2.0 COMPOSITION OF ATOMS 2.1 ATOMIC STRUCTURE
Development of the Atomic Theory
 Development of the Atomic Theory Atom The smallest particle into which an element can be divided and still be the same substance. Element A pure substance that cannot be separated into simpler substances
Development of the Atomic Theory Atom The smallest particle into which an element can be divided and still be the same substance. Element A pure substance that cannot be separated into simpler substances
Nuclear medicine. Answering your questions
 Nuclear medicine Answering your questions ANSTO s OPAL research reactor in Sydney What is nuclear medicine? This is a branch of medicine that uses radiation from radioactive tracers to provide information
Nuclear medicine Answering your questions ANSTO s OPAL research reactor in Sydney What is nuclear medicine? This is a branch of medicine that uses radiation from radioactive tracers to provide information
Radioactivity & Particles
 Radioactivity & Particles Introduction... 2 Atomic structure... 2 How are these particles arranged?... 2 Atomic notation... 4 Isotopes... 4 What is radioactivity?... 5 Types of Radiation: alpha, beta and
Radioactivity & Particles Introduction... 2 Atomic structure... 2 How are these particles arranged?... 2 Atomic notation... 4 Isotopes... 4 What is radioactivity?... 5 Types of Radiation: alpha, beta and
Unit 1 Practice Test. Matching
 Unit 1 Practice Test Matching Match each item with the correct statement below. a. proton d. electron b. nucleus e. neutron c. atom 1. the smallest particle of an element that retains the properties of
Unit 1 Practice Test Matching Match each item with the correct statement below. a. proton d. electron b. nucleus e. neutron c. atom 1. the smallest particle of an element that retains the properties of
47374_04_p25-32.qxd 2/9/07 7:50 AM Page 25. 4 Atoms and Elements
 47374_04_p25-32.qxd 2/9/07 7:50 AM Page 25 4 Atoms and Elements 4.1 a. Cu b. Si c. K d. N e. Fe f. Ba g. Pb h. Sr 4.2 a. O b. Li c. S d. Al e. H f. Ne g. Sn h. Au 4.3 a. carbon b. chlorine c. iodine d.
47374_04_p25-32.qxd 2/9/07 7:50 AM Page 25 4 Atoms and Elements 4.1 a. Cu b. Si c. K d. N e. Fe f. Ba g. Pb h. Sr 4.2 a. O b. Li c. S d. Al e. H f. Ne g. Sn h. Au 4.3 a. carbon b. chlorine c. iodine d.
3 CHEMICAL FOUNDATIONS: ELEMENTS, ATOMS AND IONS
 3 CHEMICAL FOUNDATIONS: ELEMENTS, ATOMS AND IONS All matter is built up from chemical combinations of elements. As of 2003, there are 114 known elements, of which 88 are naturally occurring; the remaining
3 CHEMICAL FOUNDATIONS: ELEMENTS, ATOMS AND IONS All matter is built up from chemical combinations of elements. As of 2003, there are 114 known elements, of which 88 are naturally occurring; the remaining
18.2 Comparing Atoms. Atomic number. Chapter 18
 As you know, some substances are made up of only one kind of atom and these substances are called elements. You already know something about a number of elements you ve heard of hydrogen, helium, silver,
As you know, some substances are made up of only one kind of atom and these substances are called elements. You already know something about a number of elements you ve heard of hydrogen, helium, silver,
Introduction to Nuclear Physics
 Introduction to Nuclear Physics 1. Atomic Structure and the Periodic Table According to the Bohr-Rutherford model of the atom, also called the solar system model, the atom consists of a central nucleus
Introduction to Nuclear Physics 1. Atomic Structure and the Periodic Table According to the Bohr-Rutherford model of the atom, also called the solar system model, the atom consists of a central nucleus
Atoms and Molecules. Preparation. Objectives. Standards. Materials. Grade Level: 5-8 Group Size: 20-30 Time: 60 90 Minutes Presenters: 2-4
 Atoms and Molecules Preparation Grade Level: 5-8 Group Size: 20-30 Time: 60 90 Minutes Presenters: 2-4 Objectives This lesson will enable students to: Describe how atoms are the building blocks of matter
Atoms and Molecules Preparation Grade Level: 5-8 Group Size: 20-30 Time: 60 90 Minutes Presenters: 2-4 Objectives This lesson will enable students to: Describe how atoms are the building blocks of matter
******* KEY ******* Atomic Structure & Periodic Table Test Study Guide
 Atomic Structure & Periodic Table Test Study Guide VOCABULARY: Write a brief definition of each term in the space provided. 1. Atoms: smallest unit of an element that has all of the properties of that
Atomic Structure & Periodic Table Test Study Guide VOCABULARY: Write a brief definition of each term in the space provided. 1. Atoms: smallest unit of an element that has all of the properties of that
Atomic Structure: Chapter Problems
 Atomic Structure: Chapter Problems Bohr Model Class Work 1. Describe the nuclear model of the atom. 2. Explain the problems with the nuclear model of the atom. 3. According to Niels Bohr, what does n stand
Atomic Structure: Chapter Problems Bohr Model Class Work 1. Describe the nuclear model of the atom. 2. Explain the problems with the nuclear model of the atom. 3. According to Niels Bohr, what does n stand
Basics of Nuclear Physics and Fission
 Basics of Nuclear Physics and Fission A basic background in nuclear physics for those who want to start at the beginning. Some of the terms used in this factsheet can be found in IEER s on-line glossary.
Basics of Nuclear Physics and Fission A basic background in nuclear physics for those who want to start at the beginning. Some of the terms used in this factsheet can be found in IEER s on-line glossary.
Chapter 18: The Structure of the Atom
 Chapter 18: The Structure of the Atom 1. For most elements, an atom has A. no neutrons in the nucleus. B. more protons than electrons. C. less neutrons than electrons. D. just as many electrons as protons.
Chapter 18: The Structure of the Atom 1. For most elements, an atom has A. no neutrons in the nucleus. B. more protons than electrons. C. less neutrons than electrons. D. just as many electrons as protons.
Main properties of atoms and nucleus
 Main properties of atoms and nucleus. Atom Structure.... Structure of Nuclei... 3. Definition of Isotopes... 4. Energy Characteristics of Nuclei... 5. Laws of Radioactive Nuclei Transformation... 3. Atom
Main properties of atoms and nucleus. Atom Structure.... Structure of Nuclei... 3. Definition of Isotopes... 4. Energy Characteristics of Nuclei... 5. Laws of Radioactive Nuclei Transformation... 3. Atom
Part I: Principal Energy Levels and Sublevels
 Part I: Principal Energy Levels and Sublevels As you already know, all atoms are made of subatomic particles, including protons, neutrons, and electrons. Positive protons and neutral neutrons are found
Part I: Principal Energy Levels and Sublevels As you already know, all atoms are made of subatomic particles, including protons, neutrons, and electrons. Positive protons and neutral neutrons are found
Elements, Atoms & Ions
 Introductory Chemistry: A Foundation FOURTH EDITION by Steven S. Zumdahl University of Illinois Elements, Atoms & Ions Chapter 4 1 2 Elements Aims: To learn about the relative abundances of the elements,
Introductory Chemistry: A Foundation FOURTH EDITION by Steven S. Zumdahl University of Illinois Elements, Atoms & Ions Chapter 4 1 2 Elements Aims: To learn about the relative abundances of the elements,
( + and - ) ( - and - ) ( + and + ) Atoms are mostly empty space. = the # of protons in the nucleus. = the # of protons in the nucleus
 Atoms are mostly empty space Atomic Structure Two regions of every atom: Nucleus - is made of protons and neutrons - is small and dense Electron cloud -is a region where you might find an electron -is
Atoms are mostly empty space Atomic Structure Two regions of every atom: Nucleus - is made of protons and neutrons - is small and dense Electron cloud -is a region where you might find an electron -is
4.1 Studying Atom. Early evidence used to develop models of atoms.
 4.1 Studying Atom Early evidence used to develop models of atoms. Democritus said that all matter consisted of extremely small particles that could NOT be divided called these particles atoms from the
4.1 Studying Atom Early evidence used to develop models of atoms. Democritus said that all matter consisted of extremely small particles that could NOT be divided called these particles atoms from the
The Structure of the Atom
 The Structure of the Atom Copyright Glencoe/McGraw-Hill, a division of the McGraw-Hill Companies, Inc. Section 4. Early Ideas About Matter pages 02 05 Section 4. Assessment page 05. Contrast the methods
The Structure of the Atom Copyright Glencoe/McGraw-Hill, a division of the McGraw-Hill Companies, Inc. Section 4. Early Ideas About Matter pages 02 05 Section 4. Assessment page 05. Contrast the methods
Electrons in Atoms & Periodic Table Chapter 13 & 14 Assignment & Problem Set
 Electrons in Atoms & Periodic Table Name Warm-Ups (Show your work for credit) Date 1. Date 2. Date 3. Date 4. Date 5. Date 6. Date 7. Date 8. Electrons in Atoms & Periodic Table 2 Study Guide: Things You
Electrons in Atoms & Periodic Table Name Warm-Ups (Show your work for credit) Date 1. Date 2. Date 3. Date 4. Date 5. Date 6. Date 7. Date 8. Electrons in Atoms & Periodic Table 2 Study Guide: Things You
Question: Do all electrons in the same level have the same energy?
 Question: Do all electrons in the same level have the same energy? From the Shells Activity, one important conclusion we reached based on the first ionization energy experimental data is that electrons
Question: Do all electrons in the same level have the same energy? From the Shells Activity, one important conclusion we reached based on the first ionization energy experimental data is that electrons
UNIT (2) ATOMS AND ELEMENTS
 UNIT (2) ATOMS AND ELEMENTS 2.1 Elements An element is a fundamental substance that cannot be broken down by chemical means into simpler substances. Each element is represented by an abbreviation called
UNIT (2) ATOMS AND ELEMENTS 2.1 Elements An element is a fundamental substance that cannot be broken down by chemical means into simpler substances. Each element is represented by an abbreviation called
About the course GENERAL CHEMISTRY. Recommended literature: Chemistry: science of the matter. Responsible for the course: Dr.
 About the course GENERAL CHEMISTRY University of Pécs Medical School Academic year 2009-2010. Responsible for the course: Dr. Attila AGÓCS Optional course for 2 credit points. To have grade at the and
About the course GENERAL CHEMISTRY University of Pécs Medical School Academic year 2009-2010. Responsible for the course: Dr. Attila AGÓCS Optional course for 2 credit points. To have grade at the and
Chapter 5 TEST: The Periodic Table name
 Chapter 5 TEST: The Periodic Table name HPS # date: Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The order of elements in the periodic table is based
Chapter 5 TEST: The Periodic Table name HPS # date: Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The order of elements in the periodic table is based
ATOMS AND THE PERIODIC TABLE CHAPTER 3 PHYSICAL SCIENCE
 ATOMS AND THE PERIODIC TABLE CHAPTER 3 PHYSICAL SCIENCE Chapter 3 Vocabulary Words (27 words) Nucleus Atomic number Proton Mass number Neutron Isotopes Electron Atomic mass unit (amu) Energy level Average
ATOMS AND THE PERIODIC TABLE CHAPTER 3 PHYSICAL SCIENCE Chapter 3 Vocabulary Words (27 words) Nucleus Atomic number Proton Mass number Neutron Isotopes Electron Atomic mass unit (amu) Energy level Average
22.1 Nuclear Reactions
 In the Middle Ages, individuals called alchemists spent a lot of time trying to make gold. Often, they fooled people into believing that they had made gold. Although alchemists never succeeded in making
In the Middle Ages, individuals called alchemists spent a lot of time trying to make gold. Often, they fooled people into believing that they had made gold. Although alchemists never succeeded in making
Structure and Properties of Atoms
 PS-2.1 Compare the subatomic particles (protons, neutrons, electrons) of an atom with regard to mass, location, and charge, and explain how these particles affect the properties of an atom (including identity,
PS-2.1 Compare the subatomic particles (protons, neutrons, electrons) of an atom with regard to mass, location, and charge, and explain how these particles affect the properties of an atom (including identity,
Medical Physics and Radioactivity
 Medical Physics and Radioactivity Radioactivity Unstable nucleus Electromagnetic wave particle Atoms which emit electromagnetic radiation or a particle by the spontaneous transformation of their nucleus
Medical Physics and Radioactivity Radioactivity Unstable nucleus Electromagnetic wave particle Atoms which emit electromagnetic radiation or a particle by the spontaneous transformation of their nucleus
Atomic Theory Part 1
 Atomic Theory Part 1 Reading: Ch 2 sections 1 6, 8 Homework: Chapter 2: 39, 47, 43, 49, 51*, 53, 55, 57, 71, 73, 77, 99, 103 (optional) * = important homework question The Atomic Theory (John Dalton, 1803)
Atomic Theory Part 1 Reading: Ch 2 sections 1 6, 8 Homework: Chapter 2: 39, 47, 43, 49, 51*, 53, 55, 57, 71, 73, 77, 99, 103 (optional) * = important homework question The Atomic Theory (John Dalton, 1803)
2 ATOMIC SYSTEMATICS AND NUCLEAR STRUCTURE
 2 ATOMIC SYSTEMATICS AND NUCLEAR STRUCTURE In this chapter the principles and systematics of atomic and nuclear physics are summarised briefly, in order to introduce the existence and characteristics of
2 ATOMIC SYSTEMATICS AND NUCLEAR STRUCTURE In this chapter the principles and systematics of atomic and nuclear physics are summarised briefly, in order to introduce the existence and characteristics of
PERIODIC TABLE. reflect
 reflect Suppose you wanted to organize your locker at school. How could you separate and arrange everything in an organized way? You could place the books, notebooks, and folders on a shelf that is separate
reflect Suppose you wanted to organize your locker at school. How could you separate and arrange everything in an organized way? You could place the books, notebooks, and folders on a shelf that is separate
Periodic Table Questions
 Periodic Table Questions 1. The elements characterized as nonmetals are located in the periodic table at the (1) far left; (2) bottom; (3) center; (4) top right. 2. An element that is a liquid at STP is
Periodic Table Questions 1. The elements characterized as nonmetals are located in the periodic table at the (1) far left; (2) bottom; (3) center; (4) top right. 2. An element that is a liquid at STP is
Atomic Structure Chapter 5 Assignment & Problem Set
 Atomic Structure Name Warm-Ups (Show your work for credit) Date 1. Date 2. Date 3. Date 4. Date 5. Date 6. Date 7. Date 8. Atomic Structure 2 Study Guide: Things You Must Know Vocabulary (know the definition
Atomic Structure Name Warm-Ups (Show your work for credit) Date 1. Date 2. Date 3. Date 4. Date 5. Date 6. Date 7. Date 8. Atomic Structure 2 Study Guide: Things You Must Know Vocabulary (know the definition
Chapter 7. Electron Structure of the Atom. Chapter 7 Topics
 Chapter 7 Electron Structure of the Atom Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. 1 Chapter 7 Topics 1. Electromagnetic radiation 2. The Bohr model of
Chapter 7 Electron Structure of the Atom Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. 1 Chapter 7 Topics 1. Electromagnetic radiation 2. The Bohr model of
The Atom and the Periodic Table. Electron Cloud Structure Energy Levels Rows on the Periodic Table Bohr Models Electron Dot Diagrams
 The Atom and the Periodic Table Electron Cloud Structure Energy Levels Rows on the Periodic Table Bohr Models Electron Dot Diagrams Review The vertical columns in the periodic table are called groups.
The Atom and the Periodic Table Electron Cloud Structure Energy Levels Rows on the Periodic Table Bohr Models Electron Dot Diagrams Review The vertical columns in the periodic table are called groups.
IONISATION ENERGY CONTENTS
 IONISATION ENERGY IONISATION ENERGY CONTENTS What is Ionisation Energy? Definition of t Ionisation Energy What affects Ionisation Energy? General variation across periods Variation down groups Variation
IONISATION ENERGY IONISATION ENERGY CONTENTS What is Ionisation Energy? Definition of t Ionisation Energy What affects Ionisation Energy? General variation across periods Variation down groups Variation
EARLY ATOMIC THEORY AND STRUCTURE
 CHAPTER 5 EARLY ATOMIC THEORY AND STRUCTURE SOLUTIONS TO REVIEW QUESTIONS 1. Elements are composed of indivisable particles called atoms. Atoms of the same element have the same properties; atoms of different
CHAPTER 5 EARLY ATOMIC THEORY AND STRUCTURE SOLUTIONS TO REVIEW QUESTIONS 1. Elements are composed of indivisable particles called atoms. Atoms of the same element have the same properties; atoms of different
2014 Spring CHEM101 Ch1-2 Review Worksheet Modified by Dr. Cheng-Yu Lai,
 Ch1 1) Which of the following underlined items is not an intensive property? A) A chemical reaction requires 3.00 g of oxygen. B) The density of helium at 25 C is 1.64 10-4 g/cm3. C) The melting point
Ch1 1) Which of the following underlined items is not an intensive property? A) A chemical reaction requires 3.00 g of oxygen. B) The density of helium at 25 C is 1.64 10-4 g/cm3. C) The melting point
Industrial tracers - Finding leaks and blockages
 Cancer Treatment Alpha radiation is used to treat various forms of cancer. This process, called unsealed source radiotherapy, involves inserting tiny amounts of radium-226 into cancerous organs. The alpha
Cancer Treatment Alpha radiation is used to treat various forms of cancer. This process, called unsealed source radiotherapy, involves inserting tiny amounts of radium-226 into cancerous organs. The alpha
The Periodic Table: Periodic trends
 Unit 1 The Periodic Table: Periodic trends There are over one hundred different chemical elements. Some of these elements are familiar to you such as hydrogen, oxygen, nitrogen and carbon. Each one has
Unit 1 The Periodic Table: Periodic trends There are over one hundred different chemical elements. Some of these elements are familiar to you such as hydrogen, oxygen, nitrogen and carbon. Each one has
Molecular Models & Lewis Dot Structures
 Molecular Models & Lewis Dot Structures Objectives: 1. Draw Lewis structures for atoms, ions and simple molecules. 2. Use Lewis structures as a guide to construct three-dimensional models of small molecules.
Molecular Models & Lewis Dot Structures Objectives: 1. Draw Lewis structures for atoms, ions and simple molecules. 2. Use Lewis structures as a guide to construct three-dimensional models of small molecules.
Physics 1104 Midterm 2 Review: Solutions
 Physics 114 Midterm 2 Review: Solutions These review sheets cover only selected topics from the chemical and nuclear energy chapters and are not meant to be a comprehensive review. Topics covered in these
Physics 114 Midterm 2 Review: Solutions These review sheets cover only selected topics from the chemical and nuclear energy chapters and are not meant to be a comprehensive review. Topics covered in these
3 Atomic Structure 15
 3 Atomic Structure 15 3.1 Atoms You need to be familiar with the terms in italics The diameter of the nucleus is approximately 10-15 m and an atom 10-10 m. All matter consists of atoms. An atom can be
3 Atomic Structure 15 3.1 Atoms You need to be familiar with the terms in italics The diameter of the nucleus is approximately 10-15 m and an atom 10-10 m. All matter consists of atoms. An atom can be
Elements in the periodic table are indicated by SYMBOLS. To the left of the symbol we find the atomic mass (A) at the upper corner, and the atomic num
 . ATOMIC STRUCTURE FUNDAMENTALS LEARNING OBJECTIVES To review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. This material is essential
. ATOMIC STRUCTURE FUNDAMENTALS LEARNING OBJECTIVES To review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. This material is essential
Student Exploration: Electron Configuration
 Name: Date: Student Exploration: Electron Configuration Vocabulary: atomic number, atomic radius, Aufbau principle, chemical family, diagonal rule, electron configuration, Hund s rule, orbital, Pauli exclusion
Name: Date: Student Exploration: Electron Configuration Vocabulary: atomic number, atomic radius, Aufbau principle, chemical family, diagonal rule, electron configuration, Hund s rule, orbital, Pauli exclusion
Chem term # 1 review sheet C. 12 A. 1
 hem term # 1 review sheet Name: ate: 1. n isotope of which element has an atomic number of 6 and a mass number of 14?. carbon. magnesium. nitrogen. silicon 6. Which atoms represent different isotopes of
hem term # 1 review sheet Name: ate: 1. n isotope of which element has an atomic number of 6 and a mass number of 14?. carbon. magnesium. nitrogen. silicon 6. Which atoms represent different isotopes of
Atoms and Elements. Outline Atoms Orbitals and Energy Levels Periodic Properties Homework
 Atoms and the Periodic Table The very hot early universe was a plasma with cationic nuclei separated from negatively charged electrons. Plasmas exist today where the energy of the particles is very high,
Atoms and the Periodic Table The very hot early universe was a plasma with cationic nuclei separated from negatively charged electrons. Plasmas exist today where the energy of the particles is very high,
CHAPTER 4: ATOMS AND ELEMENTS
 CHAPTER 4: ATOMS AND ELEMENTS Problems: 1-70 then after Chapter 9, complete 71-94, 103-104, 107-108, 113-114 4.1 Experiencing Atoms at Tiburon atom: smallest identifiable unit of an element All matter
CHAPTER 4: ATOMS AND ELEMENTS Problems: 1-70 then after Chapter 9, complete 71-94, 103-104, 107-108, 113-114 4.1 Experiencing Atoms at Tiburon atom: smallest identifiable unit of an element All matter
APS Science Curriculum Unit Planner
 APS Science Curriculum Unit Planner Grade Level/Subject Chemistry Stage 1: Desired Results Enduring Understanding Topic 1: Elements and the Periodic Table: The placement of elements on the periodic table
APS Science Curriculum Unit Planner Grade Level/Subject Chemistry Stage 1: Desired Results Enduring Understanding Topic 1: Elements and the Periodic Table: The placement of elements on the periodic table
Chapter 2 The Chemical Context of Life
 Chapter 2 The Chemical Context of Life Multiple-Choice Questions 1) About 25 of the 92 natural elements are known to be essential to life. Which four of these 25 elements make up approximately 96% of living
Chapter 2 The Chemical Context of Life Multiple-Choice Questions 1) About 25 of the 92 natural elements are known to be essential to life. Which four of these 25 elements make up approximately 96% of living
Chapter 17: Radioactivity and Nuclear Chemistry
 Chapter 7: Radioactivity and Nuclear Chemistry Problems: -20, 24-30, 32-46, 49-70, 74-88, 99-0 7.2 THE DISCOVERY OF RADIOACTIVITY In 896, a French physicist named Henri Becquerel discovered that uranium-containing
Chapter 7: Radioactivity and Nuclear Chemistry Problems: -20, 24-30, 32-46, 49-70, 74-88, 99-0 7.2 THE DISCOVERY OF RADIOACTIVITY In 896, a French physicist named Henri Becquerel discovered that uranium-containing
Chapter 4, Lesson 2: The Periodic Table
 Chapter 4, Lesson 2: The Periodic Table Key Concepts The periodic table is a chart containing information about the atoms that make up all matter. An element is a substance made up of only one type of
Chapter 4, Lesson 2: The Periodic Table Key Concepts The periodic table is a chart containing information about the atoms that make up all matter. An element is a substance made up of only one type of
EXPERIMENT 4 The Periodic Table - Atoms and Elements
 EXPERIMENT 4 The Periodic Table - Atoms and Elements INTRODUCTION Primary substances, called elements, build all the materials around you. There are more than 109 different elements known today. The elements
EXPERIMENT 4 The Periodic Table - Atoms and Elements INTRODUCTION Primary substances, called elements, build all the materials around you. There are more than 109 different elements known today. The elements
Chapter 2: The Chemical Context of Life
 Chapter 2: The Chemical Context of Life Name Period This chapter covers the basics that you may have learned in your chemistry class. Whether your teacher goes over this chapter, or assigns it for you
Chapter 2: The Chemical Context of Life Name Period This chapter covers the basics that you may have learned in your chemistry class. Whether your teacher goes over this chapter, or assigns it for you
WAVES AND ELECTROMAGNETIC RADIATION
 WAVES AND ELECTROMAGNETIC RADIATION All waves are characterized by their wavelength, frequency and speed. Wavelength (lambda, ): the distance between any 2 successive crests or troughs. Frequency (nu,):
WAVES AND ELECTROMAGNETIC RADIATION All waves are characterized by their wavelength, frequency and speed. Wavelength (lambda, ): the distance between any 2 successive crests or troughs. Frequency (nu,):
Bohr s Model of the Atom
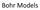 Bohr Models Bohr s Model of the Atom Focuses on electrons and their arrangement. Bohr stated that electrons move with constant speed in fixed orbits around the nucleus, like planets around a sun. Bohr
Bohr Models Bohr s Model of the Atom Focuses on electrons and their arrangement. Bohr stated that electrons move with constant speed in fixed orbits around the nucleus, like planets around a sun. Bohr
B) atomic number C) both the solid and the liquid phase D) Au C) Sn, Si, C A) metal C) O, S, Se C) In D) tin D) methane D) bismuth B) Group 2 metal
 1. The elements on the Periodic Table are arranged in order of increasing A) atomic mass B) atomic number C) molar mass D) oxidation number 2. Which list of elements consists of a metal, a metalloid, and
1. The elements on the Periodic Table are arranged in order of increasing A) atomic mass B) atomic number C) molar mass D) oxidation number 2. Which list of elements consists of a metal, a metalloid, and
Elements and Atoms: The Building Blocks of Matter
 OpenStax-CNX module: m45998 1 Elements and Atoms: The Building Blocks of Matter OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By
OpenStax-CNX module: m45998 1 Elements and Atoms: The Building Blocks of Matter OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By
Nuclear Physics. Nuclear Physics comprises the study of:
 Nuclear Physics Nuclear Physics comprises the study of: The general properties of nuclei The particles contained in the nucleus The interaction between these particles Radioactivity and nuclear reactions
Nuclear Physics Nuclear Physics comprises the study of: The general properties of nuclei The particles contained in the nucleus The interaction between these particles Radioactivity and nuclear reactions
Answers to Review Questions for Atomic Theory Quiz #1
 Answers to Review Questions for Atomic Theory Quiz #1 Multiple Choice Questions: 1. c 7. a 13. c 19. a 25. b 31. b 37. a 43. d 2. d 8. c 14. c 20. c 26. d 32. c 38. d 44. b 3. b 9. a 15. b 21. c 27. b
Answers to Review Questions for Atomic Theory Quiz #1 Multiple Choice Questions: 1. c 7. a 13. c 19. a 25. b 31. b 37. a 43. d 2. d 8. c 14. c 20. c 26. d 32. c 38. d 44. b 3. b 9. a 15. b 21. c 27. b
LEWIS DIAGRAMS. by DR. STEPHEN THOMPSON MR. JOE STALEY
 by DR. STEPHEN THOMPSON MR. JOE STALEY The contents of this module were developed under grant award # P116B-001338 from the Fund for the Improvement of Postsecondary Education (FIPSE), United States Department
by DR. STEPHEN THOMPSON MR. JOE STALEY The contents of this module were developed under grant award # P116B-001338 from the Fund for the Improvement of Postsecondary Education (FIPSE), United States Department
Atomic structure. Resources and methods for learning about these subjects (list a few here, in preparation for your research):
 Atomic structure This worksheet and all related files are licensed under the Creative Commons Attribution License, version 1.0. To view a copy of this license, visit http://creativecommons.org/licenses/by/1.0/,
Atomic structure This worksheet and all related files are licensed under the Creative Commons Attribution License, version 1.0. To view a copy of this license, visit http://creativecommons.org/licenses/by/1.0/,
Basic Nuclear Concepts
 Section 7: In this section, we present a basic description of atomic nuclei, the stored energy contained within them, their occurrence and stability Basic Nuclear Concepts EARLY DISCOVERIES [see also Section
Section 7: In this section, we present a basic description of atomic nuclei, the stored energy contained within them, their occurrence and stability Basic Nuclear Concepts EARLY DISCOVERIES [see also Section
Unit 2 Atomic Structure
 Unit 2 Atomic Structure Big Idea: Atomic structure explains patterns in the behavior of elements and allows us to predict the chemical and physical behavior of a given element. The organization of elements
Unit 2 Atomic Structure Big Idea: Atomic structure explains patterns in the behavior of elements and allows us to predict the chemical and physical behavior of a given element. The organization of elements
Chemistry 1000 Lecture 2: Nuclear reactions and radiation. Marc R. Roussel
 Chemistry 1000 Lecture 2: Nuclear reactions and radiation Marc R. Roussel Nuclear reactions Ordinary chemical reactions do not involve the nuclei, so we can balance these reactions by making sure that
Chemistry 1000 Lecture 2: Nuclear reactions and radiation Marc R. Roussel Nuclear reactions Ordinary chemical reactions do not involve the nuclei, so we can balance these reactions by making sure that
Electrons In Atoms Mr. O Brien (SFHS) Chapter 5 Standard 1D
 Electrons In Atoms Mr. O Brien (SFHS) Chapter 5 Standard 1D Electrons in Atoms (std.1d) What are Bohr Models? planetary model in which the negatively-charged electrons orbit a small, positively-charged
Electrons In Atoms Mr. O Brien (SFHS) Chapter 5 Standard 1D Electrons in Atoms (std.1d) What are Bohr Models? planetary model in which the negatively-charged electrons orbit a small, positively-charged
Atoms, Ions and Molecules The Building Blocks of Matter
 Atoms, Ions and Molecules The Building Blocks of Matter Chapter 2 1 Chapter Outline 2.1 The Rutherford Model of Atomic Structure 2.2 Nuclides and Their Symbols 2.3 Navigating the Periodic Table 2.4 The
Atoms, Ions and Molecules The Building Blocks of Matter Chapter 2 1 Chapter Outline 2.1 The Rutherford Model of Atomic Structure 2.2 Nuclides and Their Symbols 2.3 Navigating the Periodic Table 2.4 The
ATOMS: ATOMIC STRUCTURE QUESTIONS AND ANSWERS
 ATOMS: ATOMIC STRUCTURE QUESTIONS AND ANSWERS QUESTION ONE: MODELS OF THE ATOM (2011;1) At different times scientists have proposed various descriptions or models of the atom to match experimental evidence
ATOMS: ATOMIC STRUCTURE QUESTIONS AND ANSWERS QUESTION ONE: MODELS OF THE ATOM (2011;1) At different times scientists have proposed various descriptions or models of the atom to match experimental evidence
Radiation and the Universe Higher Exam revision questions and answers
 Radiation and the Universe Higher Exam revision questions and answers Madeley High School Q.The names of three different processes are given in List A. Where these processes happen is given in List B.
Radiation and the Universe Higher Exam revision questions and answers Madeley High School Q.The names of three different processes are given in List A. Where these processes happen is given in List B.
