Inhibition of CYFIP2 promotes gastric cancer cell proliferation and chemoresistance to 5 fluorouracil through activation of the Akt signaling pathway
|
|
|
- Sabina Henderson
- 7 years ago
- Views:
Transcription
1 ONCOLOGY LETTERS Inhibition of CYFIP2 promotes gastric cancer cell proliferation and chemoresistance to 5 fluorouracil through activation of the Akt signaling pathway SHUHUA JIAO, NUO LI, SHUANG CAI, HAIMEI GUO and YANHUI WEN Department of Gastroenterology, The Fourth Affiliated Hospital of China Medical University, Shenyang, Liaoning , P.R. China Received January 25, 2016; Accepted December 6, 2016 DOI: /ol Abstract. Gastric cancer is a common gastrointestinal malignancy that accounts for a notable proportion of cancer associated mortalities worldwide. Cytoplasmic fragile X mental retardation 1 interacting protein 2 (CYFIP2) is a novel p53 mediated pro apoptotic protein whose expression is decreased in gastric cancer. However, whether decreased expression of CYFIP2 contributes to gastric carcinogenesis remains unclear. In order to mimic in vivo gastric tumor CYFIP2 expression levels, the present study used short hairpin RNA targeting CYFIP2 mrna to silence CYFIP2 expression in MGC803 and SGC7901 gastric cancer cells. Gastric cancer cells with constitutively decreased CYFIP2 expression levels were successfully established. It was observed that CYFIP2 knockdown promoted proliferation and colony formation, and inhibited apoptosis in these cells. Furthermore, 5 fluorouracil (5 FU) induced apoptosis was decreased following inhibition of CYFIP2 expression. In SGC7901 cells, protein expression of active caspase 3 and cleaved poly (ADP ribose) polymerase was increased following treatment with 5 FU, while phosphorylated Akt serine/threonine kinase 1 (Akt) levels were decreased. These 5 FU induced effects were reduced following CYFIP2 knockdown. In addition, inhibition of the Akt signaling pathway using the Akt inhibitor LY restored CYFIP2 knockdown SGC7901 cell chemosensitivity to 5 FU. The results of the present study demonstrate that decreased CYFIP2 expression is associated with increased gastric tumor growth in vitro and that CYFIP2 knockdown induced activation of the Akt pro survival signaling pathway confers Correspondence to: Dr Yanhui Wen, Department of Gastroenterology, The Fourth Affiliated Hospital of China Medical University, 4 East Chongshan Road, Shenyang, Liaoning , P.R. China E mail: yanhuiwen1122@163.com Key words: cytoplasmic fragile X mental retardation 1-interacting protein 2, gastric cancer, proliferation, chemoresistance, Akt serine/threonine kinase 1 signaling pathway resistance to 5 FU based chemotherapy in gastric cancer cells. Therefore, combined treatment with an Akt inhibitor and chemotherapeutic drugs may improve the efficacy of gastric cancer therapy in patients with low CYFIP2 expression. Introduction Gastric cancer accounts for a considerable proportion of cancer associated morbidity and mortality worldwide (1), and the incidence of this malignancy is higher in developing countries (2). Gastric cancer incidence and mortality has declined in previous decades, which is due in part to a reduction in smoking and the improved treatment of Helicobacter pylori infection (1,3). However, the prognosis of patients with advanced stage gastric cancer remains poor (4). The identification of novel molecules associated with gastric carcinogenesis is required to improve gastric cancer treatments. Cytoplasmic fragile X mental retardation (FMR) 1 interacting protein 2 (CYFIP2) is a homologue of CYFIP1, a protein that is capable of interacting with all FMR proteins (5). CYFIP2, also known as p53 inducible protein 121, is a protein with unknown functional domains and structural motifs. The majority of CYFIP2 studies focused on its functional role in fragile X syndrome, a common form of inherited intellectual disability and autism (5,6). However, whether CYFIP2 is associated with carcinogenesis remains to be elucidated. CYFIP2 was reported to be a candidate p53 target gene by Saller et al (7) in H1299 lung cancer cells and this was validated by Jackson et al (8) in DLD1 colorectal adenocarcinoma cells. CYFIP2 overexpression was not demonstrated to induce apoptosis in lung cancer cells (7); however, CYFIP2 overexpression promoted apoptotic like death in colorectal cancer cells (8). The former study (7) suggested that CYFIP2 induced apoptosis is part of a coordinated p53 dependent response in cancer cells, while the latter study (8) suggested that CYFIP2 itself functions as a pro apoptotic protein. CYFIP2 mrna expression has been demonstrated to be significantly decreased in gastric cancer tissues compared with non cancerous tissues (9). In the present study, the association between decreased CYFIP2 expression and gastric carcinogenesis was investigated, and the potential underlying mechanisms were examined.
2 2 JIAO et al: INHIBITION OF CYFIP2 PROMOTES CHEMORESISTANCE IN GASTRIC CANCER CELLS In order to mimic the gastric tumor microenvironment in vivo, RNA interference (RNAi) was used to suppress endogenous CYFIP2 expression in the gastric cancer cell lines MGC 803 and SGC Antitumor drug based cytotoxic chemotherapy is routinely administered to the majority of patients with advanced gastric cancer (10); however, the development of chemoresistance is common, which leads to treatment failure (11). To investigate the potential association between decreased CYFIP2 expression and chemoresistance in gastric cancer cells, gastric cancer cell proliferation and apoptosis following treatment with or without 5 fluorouracil (5 FU) was determined. Materials and methods Cell culture. The human gastric cancer cell lines MGC803 and SGC7901 were obtained from the Type Culture Collection of the Chinese Academy of Sciences (Shanghai, China). Cells were cultured in Dulbecco's modified Eagle's medium (Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA) containing 10% fetal bovine serum (HyClone; GE Healthcare, Logan, UT, USA), 100 µg/ml streptomycin and 100 U/ml penicillin. Cells were cultured at 37 C in a humidified atmosphere containing 5% CO 2. Stable transfection of gastric cancer cells with CYFIP2 tagerting short hairpin (sh)rna. In order to inhibit CYFIP2 protein expression in the gastric cancer cells, the prna H1.1 vector was purchased from GenScript (Nanjing, China) and used generate CYFIP2 targeting shrna expressing plasmids. A pair of oligonucleotides (sense, 5' GAT CCC CGG ACC TGC TTG AAG AGC TTT TCA AGA GAA AGC TCT TCA AGC AGG TCC TTT TT 3' and antisense, 5' AGC TAA AAA GGA CCT GCT TGA AGA GCT TTC TCT TGA AAA GCT CTT CAA GCA GGT CCG GG 3') targeting CYFIP2 mrna were synthesized by Sangon Biotech Co., Ltd. (Shanghai, China) and inserted into the prna H1.1 expression vector. The CYFIP2 targeting shrna targeted the sequence 5' GGA CCT GCT TGA AGA GCT T 3'. The oligonucleotides were initially allowed to anneal to form a DNA duplex, with cohesive ends containing a BamHI and HindIII site. A total of 2 µg empty prna H1.1 vector was digested using 2.5 µl BamHI and 2.5 µl HindIII (both Fermentas; Thermo Fisher Scientific, Inc.) in a 50 µl system for 30 min at 37 C. The plasmids were subsequently incubated with the CYFIP2 shrna and 1 µl T4 DNA ligase (Takara Biotechnology Co., Ltd., Dalian, China) at 16 C overnight, according to the manufacturer's protocol. Plasmids containing scrambled shrna (sense, 5' GAT CCC CTT CTC CGA ACG TGT CAC GTT TCA AGA GAA CGT GAC ACG TTC GGA GAA TTT TT 3' and antisense, 5' AGC TAA AAA TTC TCC GAA CGT GTC ACG TTC TCT TGA AAC GTG ACA CGT TCG GAG AAG GG 3') were used as a negative control. The constructed plasmids were transfected into gastric cancer cells using Lipofectamine 2000 (Invitrogen; Thermo Fisher Scientific, Inc.), according to the manufacturer's protocol. Following transfection, cells were treated with 400 µg/ml G418 (Invitrogen; Thermo Fisher Scientific, Inc.) for between 1 and 2 weeks to select for successfully transfected cells expressing CYFIP2 shrna or scrambled shrna. Reverse transcription quantitative polymerase chain reaction (RT qpcr) and western blot analyses were performed using the G418 resistant clones, as described below, to determine CYFIP2 mrna and protein expression, respectively. The cell clone from each gastric cancer cell line with the lowest CYFIP2 expression was used for subsequent analysis. Cell Counting Kit 8 (CCK 8) and colony formation assay. The CCK 8 was purchased from Beyotime Institute of Biotechnology (Haimen, China) and was used to determine cell proliferation rates. Following trypsinization (Sigma Aldrich; Merck Millipore, Darmstadt, Germany), cells were seeded into 96 well plates at a density of 2x10 3 cells/well (n=5), and cultured for 24, 48, 72 and 96 h at 37 C. A total of 10 µl CCK 8 solution in 100 µl culture medium was added into each well at each time point for 1 h and the absorbance was measured at a wavelength of 450 nm. For the colony formation assay, 100 cells were seeded into sterile 35 mm dishes and cultured at 37 C for 14 days. The culture medium was replaced every 3 days. Cell colonies were subsequently fixed using 4% paraformaldehyde for 20 min at room temperature (RT), incubated with Wright Giemsa stain (Nanjung Jiancheng Bioengineering Institute, Nanjing, China) for between 5 8 min at RT, and counted using a light microscope (n=3). Colony formation was determined as the ratio of the number of colonies:number of cells seeded. Apoptosis assay. Cell apoptosis was assessed using a flow cytometer. Certain gastric cancer cells were treated with 25 µm LY (Beyotime Institute of Biotechnology). Cells (1x10 5 ) were treated with 15 µg/ml 5 FU (Sigma Aldrich; Merck Millipore) for 24 h at 37 C. Following treatment with 5 FU, cells were trypsinized (0.2%; Thermo Fisher Scientific, Inc.) and centrifuged at 309 x g for 5 min at 4 C, prior to the addition of 500 µl binding buffer (Nanjing KeyGen Biotech Co., Ltd., Nanjing, China). Cells were subsequently cultured with 5 µl Annexin V fluorescein isothiocyanate (FITC) and 5 µl propidium iodide (PI) (both Nanjing KeyGen Biotech Co., Ltd.) for 15 min in the dark at RT. Apoptotic cells were identified using flow cytometry and BD Accuri C6 software (BD Biosciences, San Jose, CA, USA). RT qpcr. Total cellular RNA was prepared using a Fast RNApure Extraction kit (BioTek Instruments, Inc., Winooski, VT, USA). Extracted RNA was reverse transcribed into first strand cdna using Super Moloney murine leukemia virus reverse transcriptase (BioTek Instruments, Inc.) under the following temperature conditions: 25 C for 10 min; 42 C for 50 min; and 95 C for 5 min to terminate the reaction. The following CYFIP2 primers were used in PCR analysis: Forward, 5' TCC GTA TCC ACC GTC CA A T 3'; and reverse, 5' AAT CTC CAG CAG CCA CTCC 3'. qpcr was performed using the SYBR Green Master mix (Beijing Solarbio Science & Technology Co., Ltd., Beijing, China). The following thermocycling conditions were used: 94 C for 10 min; 40 cycles of 94 C for 10 sec, 60 C for 20 sec and 72 C for 30 sec; 72 C for 2.5 min; 40 C for 5.5 min; temperature was increased from 60 to 94 C at a rate of 1 C/sec; and 25 C for 1 min. The relative expression levels of CYFIP2 were calculated using the 2 ΔΔCq method (12), with β actin used as the endogenous control. The β actin primer sequences were as follows: Forward, 5' CCA TCG TCC ACC GCA AAT 3' and reverse, 5' GCT GTC ACC TTC ACC GTTC 3'.
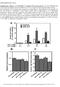
3 ONCOLOGY LETTERS 3 Figure 1. Evaluation of the knockdown efficiency of CYFIP2 following shrna transfection. CYFIP2 mrna expression levels in (A) MGC803 and (B) SGC7901 cells. CYFIP2 protein expression levels in (C) MGC803 and (D) SGC7901 cells. Values were normalized to endogenous β actin mrna and protein expression, respectively. Results are presented as the mean ± standard deviation (n=3). CYFIP2, cytoplasmic fragile X mental retardation 1 interacting protein 2; shrna, short hairpin RNA. Western blot analysis. Total cellular protein was prepared using radioimmunoprecipitation assay buffer and quantified using the BCA Protein Assay kit (both Beyotime Institute of Biotechnology). Total protein samples (30 µg) were separated using SDS PAGE on a 10% gel and transferred onto polyvinylidene difluoride membranes. Membranes were blocked with 5% (w/v) non fat milk for 1 h at RT. Membranes were subsequently incubated with primary rabbit polyclonal antibodies against CYFIP2 (1:500; cat. no. bs 7606R; Bioss, Beijing, China), active caspase 3 (1:1,000; cat. no. ab2302), cleaved poly (ADP ribose) polymerase (PARP; 1:1,000; cat. no. ab32561 (both Abcam, Cambridge, UK), phosphorylated Akt serine/threonine kinase 1 (p Akt; 1:200; cat. no. sc ), Akt (1:200; cat. no. sc 8312) and β actin (1:1,000; cat. no. sc 47778; all Santa Cruz Biotechnology, Inc., Dallas, TX, USA) overnight at 4 C. Membranes were then incubated with goat anti rabbit horseradish peroxidase (HRP) labeled immunoglobulin (Ig)G (1:5,000; cat. no. A0208; Beyotime Institute of Biotechnology) or goat anti mouse HRP labeled IgG (1:5,000; cat. no. A0216; Beyotime Institute of Biotechnology) for 45 min at 37 C. Protein bands were visualized using an enhanced chemiluminescence kit (Shanghai Seven Sea Pharmtech Company, Shanghai, China) and band densities were normalized to endogenous β actin expression. Bands were quantified using Gel Pro Analyzer software (version 4.0; Media Cybernetics, Inc., Rockville, MD, USA). Statistical analysis. Values are presented as the mean ± standard deviation of triplicate experiments. All data were analyzed using SPSS software (version 20.0; IBM SPSS, Armonk, NY, USA). Where appropriate, an unpaired t test was performed to compare data between two groups. A nonparametric test was performed to compare datasets with unequal variances. P<0.05 was considered to indicate a statistically significant difference. Results Evaluation of the knockdown efficiency of CYFIP2 shrna in gastric cancer cells. To evaluate the role of CYFIP2 in gastric
4 4 JIAO et al: INHIBITION OF CYFIP2 PROMOTES CHEMORESISTANCE IN GASTRIC CANCER CELLS Figure 2. CYFIP2 inhibition promotes gastric cancer cell growth in vitro. (A) MGC803 and (B) SGC7901 cell proliferation was significantly increased following CYFIP2 knockdown (n=5). (C) MGC803 and (D) SGC7901 colony formation was significantly increased following CYFIP2 knockdown (n=8). Results are presented as the mean ± standard deviation. CYFIP2, cytoplasmic fragile X mental retardation 1 interacting protein 2; shrna, short hairpin RNA. tumorigenesis, RNAi experiments using a CYFIP2 shrna were performed in two gastric cancer cell lines. The knockdown efficiency of the CYFIP2 shrna was determined by evaluating CYFIP2 mrna and protein expression using RT qpcr and western blotting, respectively. CYFIP2 mrna expression was significantly decreased by 74% in MGC803 cells following CYFIP2 shrna transfection compared with the scrambled shrna group (P=0.0008; Fig. 1A). CYFIP2 mrna expression was significantly decreased by 72% in SGC7901 cells following CYFIP2 shrna transfection compared with the scrambled shrna group (P<0.0001; Fig. 1B). CYFIP2 protein expression was significantly decreased by 79% in MGC803 cells following CYFIP2 shrna transfection compared with the scrambled shrna group (P<0.0001; Fig. 1A). CYFIP2 protein expression was significantly decreased by 69% in SGC7901 cells following CYFIP2 shrna transfection compared with the scrambled shrna group (P=0.0004; Fig. 1A). These results suggest that gastric cancer cells with decreased CYFIP2 expression levels were successfully established. Silencing of CYFIP2 suppresses gastric cancer cell proliferation and colony formation. Cell proliferation was determined using the CCK 8 assay. Cell proliferation rates were significantly increased in MGC803 (P=0.0016; Fig. 2A) and SGC7901 (P=0.0005; Fig. 2B) cells from 48 h following CYFIP2 shrna transfection compared with their respective scrambled shrna transfected groups. Furthermore, rates of MGC803 (P=0.038) and SGC7901 (P=0.0478) cell colony formation were significantly increased following CYFIP2 shrna transfection compared with the scrambled shrna transfected groups (Fig. 2C and D). These results demonstrate that CYFIP2 silencing contributes to gastric cancer cell proliferation in vitro. Knockdown of CYFIP2 inhibits gastric cancer cell apoptosis and induces resistance to 5 FU. Cell apoptosis was evaluated using flow cytometric analysis of Annexin V FITC/PI stained cells. In the absence of 5 FU, CYFIP2 knockdown significantly decreased the apoptotic percentage of MGC803 cells from 15.00±1.71 in the scrambled shrna transfected control cells to 6.13±0.58% (P=0.001; Fig. 3A and B). Similarly, CYFIP2 knockdown significantly decreased the apoptotic percentage of SGC7901 cells from 14.26±1.53 in the scrambled shrna transfected control cells to 3.54±0.51% (P=0.0003; Fig. 3A and C). Evasion of apoptosis is an essential component
5 ONCOLOGY LETTERS 5 Figure 3. CYFIP2 knockdown induces 5 FU resistance in gastric cancer cells in vitro. (A) MGC803 and SGC7901 cell apoptosis was quantified using flow cytometric analysis. The apoptotic cell ratio was calculated as the sum of the lower and upper right quadrants of the flow cytometric data for (B) MGC803 and (C) SGC 7901 cells. Results are presented as the mean ± standard deviation (n=3). CYFIP2, cytoplasmic fragile X mental retardation 1 interacting protein 2; 5 FU, 5 fluorouracil; shrna, short hairpin RNA; FL, fluorescence; A, area; PI, propidium iodide; FITC, fluorescein isothiocyanate. of the chemoresistance of cancer cells (13). Subsequently, the effect of CYFIP2 knockdown on the response of gastric cancer cells to 5 FU was subsequently investigated. Treatment with 5 FU induced significantly increased apoptosis in MGC803 (P=0.0073; Fig. 3B) and SGC7901 (P=0.0118; Fig. 3C) cells compared with the cells that did not receive 5 FU treatment. However, the cytotoxic effect of treatment with 5 FU was significantly decreased by CYFIP2 knockdown in MGC803 (P=0.0353; Fig. 3B) and SGC7901 (P=0.04; Fig. 3C) cells compared with the scrambled shrna transfected group. These data indicate that inhibition of CYFIP2 contributes to the development of resistance to 5 FU in gastric cancer cells. Akt serine/threonine kinase 1 (Akt) inhibition restores the 5 FU chemosensitivity of CYFIP2 knockdown SGC 7901 cells. Aberrant activation of the Akt signaling pathway is associated with tumorigenesis and chemoresistance in various types of cancer (14). The phosphorylation status of Akt in SGC 7901 cells was evaluated using western blotting following CYFIP2 knockdown and treatment with 5 FU. While total Akt protein expression remained unchanged in the SGC 7901 cells treated with the scrambled shrna, phosphorylated (p) Akt expression was decreased 12 h following treatment with 5 FU in a time dependent manner (Fig. 4A and B). CYFIP2 knockdown SGC 7901 cells exhibited significantly increased p Akt expression following treatment with 5 FU compared with the scrambled shrna transfected group at all time points (all P<0.05; Fig. 4A and B). Furthermore, the expression of cleaved caspase 3 and cleaved PARP were determined in SGC 7901 cells. 5 FU induced upregulation of cleaved caspase 3 and
6 6 JIAO et al: INHIBITION OF CYFIP2 PROMOTES CHEMORESISTANCE IN GASTRIC CANCER CELLS Figure 4. Akt inhibitor LY restores the chemosensitivity of CYFIP2 deficient SGC7901 cells to 5 FU. (A) Western blot analysis of p Akt, Akt, active caspase 3 and cleaved PARP protein expression SGC7901 cells following CYFIP2 knockdown and treatment with 5 FU. Quantification of (B) p Akt, (C) cleaved caspase 3 and (D) cleaved PARP protein bands. Values were normalized to β actin. (E) SGC7901 cell apoptosis was quantified using flow cytometric analysis following treatment with LY Results are presented as the mean ± standard deviation (n=3). Akt, Akt serine/threonine kinase 1; CYFIP2, cytoplasmic fragile X mental retardation 1 interacting protein 2; 5 FU, 5 fluorouracil; p, phosphorylated; PARP, poly (ADP ribose) polymerase; shrna, short hairpin RNA; FL, fluorescence; A, area; PI, propidium iodide; FITC, fluorescein isothiocyanate. PARP protein expression was significantly inhibited by CYFIP2 knockdown (P<0.05 vs. the scrambled shrna transfected group; Fig. 4A, C and D). To confirm whether activation of the Akt signaling pathway was associated with CYFIP2 knockdown induced chemoresistance in SGC 7901 cells, the potent Akt inhibitor LY was used. The results of the apoptosis assay performed demonstrated that treatment with LY significantly increased the apoptosis of CYFIP2 knockdown cells in the presence (P=0.0002) and absence (P=0.0007) of 5 FU (Fig. 4E). These results indicate that inhibition of the Akt signaling pathway restores the chemosensitivity of CYFIP2 knockdown gastric cancer cells to 5 FU.
7 ONCOLOGY LETTERS 7 Discussion Gastric cancer cells exhibit increased proliferation during carcinogenesis (15). A previous study revealed that CYFIP2 expression is decreased in gastric cancer tissues (9). However, whether this low expression of CYFIP2 is associated with gastric carcinogenesis remains to be elucidated. In the present study, CYFIP2 expression was suppressed in two gastric cancer cell lines in vitro through shrna mediated knockdown. It was observed that CYFIP2 knockdown promoted the proliferation and survival of malignant clones in the MGC 803 and SGC 7901 cell lines. Constitutive activation of the oncogenic protein GTPase KRas (K Ras) occurs frequently in human cancer, including gastric cancer (16,17). A previous study identified that inducible and conditional activation of K Ras resulted in the hyperproliferation of squamous epithelial cells in the forestomach of a mouse model and induced precancerous gastric lesions (18,19). Another study demonstrated that CYFIP2 inhibition is able to increase K Ras expression in colon cancer cells (20). The results of these studies and the present study indicate that CYFIP2 knockdown induced gastric cancer cell proliferation is associated with increased K Ras expression. Subsequently, the authors of the present study aim to investigate the in vivo association between CYFIP2 expression and K Ras activation in gastric tumor samples in the future. Evasion of apoptosis is an essential mechanism underlying carcinogenesis (21); however, stress induced apoptosis has also been suggested to lead to tumor formation under certain circumstances (22). The ability to activate caspases, a group of apoptosis inducing proteases, is a key mechanism used by potent anticancer drugs (23). The release of intermembrane proteins from the mitochondria, including cytochrome c, is able to promote the cleavage of caspase 9 and subsequently induce activation of the essential apoptotic protein caspase 3, which leads to cell apoptosis (23). Overexpression of CYFIP2 can lead to enhanced activation of caspase 3 and its downstream target PARP in colorectal cancer cells (8), suggesting that CYFIP2 is stimulates caspase activation in tumor cells. The results of the present study were consistent with the results of previous studies, in demonstrating that gastric SGC7901 cancer cells expressing CYFIP2 targeting shrna exhibited decreased levels of active caspase 3 and cleaved PARP. Patients with gastric cancer are typically treated with DNA damage inducing drugs (10). Chemotherapeutic regimes frequently induce cancer cell apoptosis through the activation of caspases (24). The present study demonstrated that treatment with 5 FU induced apoptosis, and promoted caspase 3 and PARP cleavage in gastric cancer cells. However, 5 FU induced cytotoxicity was decreased in cells with decreased CYFIP2 expression. These results suggest that 5 FU chemoresistance in gastric cancer cells is associated with decreased CYFIP2 expression. Akt is an essential effector of multiple signaling pathways, transducing downstream survival signals (25). Akt signaling is activated by phosphoinositide 3 kinase (26) and inhibited by the tumor suppressors p53 (27) and phosphatase and tensin homolog (28). CYFIP2 is a novel proapoptotic factor that is regulated by p53 (8). 5 FU initiates apoptosis in gastric tumor cells by activating the p53 signaling pathway (29) and inactivating the Akt signaling pathway (30). To the best of our knowledge, the present study was the first to demonstrate that 5 FU induced Akt signaling inhibition was decreased by CYFIP2 knockdown in gastric cancers in vitro. The role of the Akt signaling pathway in the promotion of tumorigenesis has been studied previously; however, there is no evidence of functional crosstalk between CYFIP2 and Akt in gastric cancer. Notably, another p53 inducible protein, pleckstrin homology like domain family A member 3, has been reported to compete with Akt for the binding of membrane lipids and therefore inhibit Akt activation (27). Thus, the association between CYFIP2 and Akt during gastric tumorigenesis requires further investigation in the future, via the ectopic overexpression of CYFIP2. The authors of the present study have also identified that BGC 823 gastric cancer cells exhibit decreased CYFIP2 expression (Jiao et al, unpublished data). CYFIP2 gain of function experiments in BGC 823 cells may assist in the elucidation of the role of CYFIP2 in gastric cancer development and progression. In conclusion, the results of the present study demonstrate that inhibition of CYFIP2 contributes to the proliferation of gastric cancer cells in vitro. In addition, CYFIP2 knockdown induced activation of the Akt pro survival signaling pathway was revealed to confer resistance to 5 FU based chemotherapy in gastric cancer cells. These results indicate that combination therapy with an Akt inhibitor and 5 FU may improve the efficacy of treatments for patients with gastric cancer who exhibit decreased CYFIP2 expression. Acknowledgements The present study was supported by the Science and Technology Project of Liaoning Province (grant no ). References 1. McLean MH and El Omar EM: Genetics of gastric cancer. Nat Rev Gastroenterol Hepatol 11: , Herrero R, Park JY and Forman D: The fight against gastric cancer the IARC working group report. Best Pract Res Clin Gastroenterol 28: , Peleteiro B, Bastos A, Ferro A and Lunet N: Prevalence of Helicobacter pylori infection worldwide: A systematic review of studies with national coverage. Dig Dis Sci 59: , Shah MA and Ajani JA: Gastric cancer an enigmatic and heterogeneous disease. JAMA 303: , Schenck A, Bardoni B, Moro A, Bagni C and Mandel JL: A highly conserved protein family interacting with the fragile X mental retardation protein (FMRP) and displaying selective interactions with FMRP related proteins FXR1P and FXR2P. Proc Natl Acad Sci USA 98: , Hoeffer CA, Sanchez E, Hagerman RJ, Mu Y, Nguyen DV, Wong H, Whelan AM, Zukin RS, Klann E and Tassone F: Altered mtor signaling and enhanced CYFIP2 expression levels in subjects with fragile X syndrome. Genes Brain Behav 11: , Saller E, Tom E, Brunori M, Otter M, Estreicher A, Mack DH and Iggo R: Increased apoptosis induction by 121F mutant p53. EMBO J 18: , Jackson RS II, Cho YJ, Stein S and Liang P: CYFIP2, a direct p53 target, is leptomycin B sensitive. Cell Cycle 6: , Cheng AS, Li MS, Kang W, Cheng VY, Chou JL, Lau SS, Go MY, Lee CC, Ling TK, Ng EK, et al: Helicobacter pylori causes epigenetic dysregulation of FOXD3 to promote gastric carcinogenesis. Gastroenterology 144: , e9, Liu R, Huang M, Zhao X, Peng W, Sun S, Cao J, Ji D, Wang C, Guo W and Li J, et al: Neutropenia predicts better prognosis in patients with metastatic gastric cancer on a combined epirubicin, oxaliplatin and 5 fluorouracil regimen. Oncotarget 6: , 2015.
8 8 JIAO et al: INHIBITION OF CYFIP2 PROMOTES CHEMORESISTANCE IN GASTRIC CANCER CELLS 11. Bain GH, Collie Duguid E, Murray GI, Gilbert FJ, Denison A, McKiddie F, Ahearn T, Fleming I, Leeds J, Phull P, et al: Tumour expression of leptin is associated with chemotherapy resistance and therapy independent prognosis in gastro oesophageal adenocarcinomas. Br J Cancer 110: , Livak KJ and Schmittgen TD: Analysis of relative gene expression data using real time quantitative PCR and the 2( Delta Delta C(T)) Method. Methods 25: , Delbridge AR and Strasser A: The BCL 2 protein family, BH3 mimetics and cancer therapy. Cell Death Differ 22: , Mayer IA and Arteaga CL: The PI3K/AKT pathway as a target for cancer treatment. Annu Rev Med 67: 11 28, Xia HH and Talley NJ: Apoptosis in gastric epithelium induced by Helicobacter pylori infection: Implications in gastric carcinogenesis. Am J Gastroenterol 96: 16 26, Park MT, Kim MJ, Suh Y, Kim RK, Kim H, Lim EJ, Yoo KC, Lee GH, Kim YH, Hwang SG, et al: Novel signaling axis for ROS generation during K Ras induced cellular transformation. Cell Death Differ 21: , Song IS, Oh NS, Kim HT, Ha GH, Jeong SY, Kim JM, Kim DI, Yoo HS, Kim CH and Kim NS: Human ZNF312b promotes the progression of gastric cancer by transcriptional activation of the K ras gene. Cancer Res 69: , Matkar SS, Durham A, Brice A, Wang TC, Rustgi AK and Hua X: Systemic activation of K ras rapidly induces gastric hyperplasia and metaplasia in mice. Am J Cancer Res 1: , Lee TI, Jenner RG, Boyer LA, Guenther MG, Levine SS, Kumar RM, Chevalier B, Johnstone SE, Cole MF, Isono K, et al: Control of developmental regulators by Polycomb in human embryonic stem cells. Cell 125: , Mongroo PS, Noubissi FK, Cuatrecasas M, Kalabis J, King CE, Johnstone CN, Bowser MJ, Castells A, Spiegelman VS and Rustgi AK: IMP 1 displays cross talk with K Ras and modulates colon cancer cell survival through the novel proapoptotic protein CYFIP2. Cancer Res 71: , Hanahan D and Weinberg RA: The hallmarks of cancer. Cell 100: 57 70, Labi V and Erlacher M: How cell death shapes cancer. Cell Death Dis 6: e1675, Hensley P, Mishra M and Kyprianou N: Targeting caspases in cancer therapeutics. Biol Chem 394: , Galluzzi L, Kepp O and Kroemer G: Caspase 3 and prostaglandins signal for tumor regrowth in cancer therapy. Oncogene 31: , Xue G, Zippelius A, Wicki A, Mandalà M, Tang F, Massi D and Hemmings BA: Integrated Akt/PKB signaling in immunomodulation and its potential role in cancer immunotherapy. J Natl Cancer Inst 107: pii: djv171, Dibble CC and Cantley LC: Regulation of mtorc1 by PI3K signaling. Trends Cell Biol 25: , Kawase T, Ohki R, Shibata T, Tsutsumi S, Kamimura N, Inazawa J, Ohta T, Ichikawa H, Aburatani H, Tashiro F and Taya Y: PH domain only protein PHLDA3 is a p53 regulated repressor of Akt. Cell 136: , Worby CA and Dixon JE: Pten. Annu Rev Biochem 83: , Li B, Wang L and Chi B: Upregulation of periostin prevents P53 mediated apoptosis in SGC 7901 gastric cancer cells. Mol Biol Rep 40: , Hong SW, Jung KH, Park BH, Zheng HM, Lee HS, Choi MJ, Yun JI, Kang NS, Lee J and Hong SS: KRC 408, a novel c Met inhibitor, suppresses cell proliferation and angiogenesis of gastric cancer. Cancer Lett 332: 74 82, 2013.
10 µg lyophilized plasmid DNA (store lyophilized plasmid at 20 C)
 TECHNICAL DATA SHEET BIOLUMINESCENCE RESONANCE ENERGY TRANSFER RENILLA LUCIFERASE FUSION PROTEIN EXPRESSION VECTOR Product: prluc-c Vectors Catalog number: Description: Amount: The prluc-c vectors contain
TECHNICAL DATA SHEET BIOLUMINESCENCE RESONANCE ENERGY TRANSFER RENILLA LUCIFERASE FUSION PROTEIN EXPRESSION VECTOR Product: prluc-c Vectors Catalog number: Description: Amount: The prluc-c vectors contain
GENEWIZ, Inc. DNA Sequencing Service Details for USC Norris Comprehensive Cancer Center DNA Core
 DNA Sequencing Services Pre-Mixed o Provide template and primer, mixed into the same tube* Pre-Defined o Provide template and primer in separate tubes* Custom o Full-service for samples with unknown concentration
DNA Sequencing Services Pre-Mixed o Provide template and primer, mixed into the same tube* Pre-Defined o Provide template and primer in separate tubes* Custom o Full-service for samples with unknown concentration
pcas-guide System Validation in Genome Editing
 pcas-guide System Validation in Genome Editing Tagging HSP60 with HA tag genome editing The latest tool in genome editing CRISPR/Cas9 allows for specific genome disruption and replacement in a flexible
pcas-guide System Validation in Genome Editing Tagging HSP60 with HA tag genome editing The latest tool in genome editing CRISPR/Cas9 allows for specific genome disruption and replacement in a flexible
(http://genomes.urv.es/caical) TUTORIAL. (July 2006)
 (http://genomes.urv.es/caical) TUTORIAL (July 2006) CAIcal manual 2 Table of contents Introduction... 3 Required inputs... 5 SECTION A Calculation of parameters... 8 SECTION B CAI calculation for FASTA
(http://genomes.urv.es/caical) TUTORIAL (July 2006) CAIcal manual 2 Table of contents Introduction... 3 Required inputs... 5 SECTION A Calculation of parameters... 8 SECTION B CAI calculation for FASTA
(A) Microarray analysis was performed on ATM and MDM isolated from 4 obese donors.
 Legends of supplemental figures and tables Figure 1: Overview of study design and results. (A) Microarray analysis was performed on ATM and MDM isolated from 4 obese donors. After raw data gene expression
Legends of supplemental figures and tables Figure 1: Overview of study design and results. (A) Microarray analysis was performed on ATM and MDM isolated from 4 obese donors. After raw data gene expression
DNA Sample preparation and Submission Guidelines
 DNA Sample preparation and Submission Guidelines Requirements: Please submit samples in 1.5ml microcentrifuge tubes. Fill all the required information in the Eurofins DNA sequencing order form and send
DNA Sample preparation and Submission Guidelines Requirements: Please submit samples in 1.5ml microcentrifuge tubes. Fill all the required information in the Eurofins DNA sequencing order form and send
Inverse PCR & Cycle Sequencing of P Element Insertions for STS Generation
 BDGP Resources Inverse PCR & Cycle Sequencing of P Element Insertions for STS Generation For recovery of sequences flanking PZ, PlacW and PEP elements E. Jay Rehm Berkeley Drosophila Genome Project I.
BDGP Resources Inverse PCR & Cycle Sequencing of P Element Insertions for STS Generation For recovery of sequences flanking PZ, PlacW and PEP elements E. Jay Rehm Berkeley Drosophila Genome Project I.
ONLINE SUPPLEMENTAL MATERIAL. Allele-Specific Expression of Angiotensinogen in Human Subcutaneous Adipose Tissue
 ONLINE SUPPLEMENTAL MATERIAL Allele-Specific Expression of Angiotensinogen in Human Subcutaneous Adipose Tissue Sungmi Park 1, Ko-Ting Lu 1, Xuebo Liu 1, Tapan K. Chatterjee 2, Steven M. Rudich 3, Neal
ONLINE SUPPLEMENTAL MATERIAL Allele-Specific Expression of Angiotensinogen in Human Subcutaneous Adipose Tissue Sungmi Park 1, Ko-Ting Lu 1, Xuebo Liu 1, Tapan K. Chatterjee 2, Steven M. Rudich 3, Neal
Effect of STK17A on the sensitivity of ovarian cancer cells to paclitaxel and carboplatin
 ONCOLOGY LETTERS Effect of STK17A on the sensitivity of ovarian cancer cells to paclitaxel and carboplatin JIANHUA GAO, DAN LIU, JIE LI, QIANLIN SONG and QI WANG Department of Obstetrics and Gynecology,
ONCOLOGY LETTERS Effect of STK17A on the sensitivity of ovarian cancer cells to paclitaxel and carboplatin JIANHUA GAO, DAN LIU, JIE LI, QIANLIN SONG and QI WANG Department of Obstetrics and Gynecology,
UNIVERSITETET I OSLO Det matematisk-naturvitenskapelige fakultet
 1 UNIVERSITETET I OSLO Det matematisk-naturvitenskapelige fakultet Exam in: MBV4010 Arbeidsmetoder i molekylærbiologi og biokjemi I MBV4010 Methods in molecular biology and biochemistry I Day of exam:.
1 UNIVERSITETET I OSLO Det matematisk-naturvitenskapelige fakultet Exam in: MBV4010 Arbeidsmetoder i molekylærbiologi og biokjemi I MBV4010 Methods in molecular biology and biochemistry I Day of exam:.
Next Generation Sequencing
 Next Generation Sequencing 38. Informationsgespräch der Blutspendezentralefür Wien, Niederösterreich und Burgenland Österreichisches Rotes Kreuz 22. November 2014, Parkhotel Schönbrunn Die Zukunft hat
Next Generation Sequencing 38. Informationsgespräch der Blutspendezentralefür Wien, Niederösterreich und Burgenland Österreichisches Rotes Kreuz 22. November 2014, Parkhotel Schönbrunn Die Zukunft hat
Notch 1 -dependent regulation of cell fate in colorectal cancer
 Notch 1 -dependent regulation of cell fate in colorectal cancer Referees: PD Dr. Tobias Dick Prof. Dr. Wilfried Roth http://d-nb.info/1057851272 CONTENTS Summary 1 Zusammenfassung 2 1 INTRODUCTION 3 1.1
Notch 1 -dependent regulation of cell fate in colorectal cancer Referees: PD Dr. Tobias Dick Prof. Dr. Wilfried Roth http://d-nb.info/1057851272 CONTENTS Summary 1 Zusammenfassung 2 1 INTRODUCTION 3 1.1
Table S1. Related to Figure 4
 Table S1. Related to Figure 4 Final Diagnosis Age PMD Control Control 61 15 Control 67 6 Control 68 10 Control 49 15 AR-PD PD 62 15 PD 65 4 PD 52 18 PD 68 10 AR-PD cingulate cortex used for immunoblot
Table S1. Related to Figure 4 Final Diagnosis Age PMD Control Control 61 15 Control 67 6 Control 68 10 Control 49 15 AR-PD PD 62 15 PD 65 4 PD 52 18 PD 68 10 AR-PD cingulate cortex used for immunoblot
Understanding the immune response to bacterial infections
 Understanding the immune response to bacterial infections A Ph.D. (SCIENCE) DISSERTATION SUBMITTED TO JADAVPUR UNIVERSITY SUSHIL KUMAR PATHAK DEPARTMENT OF CHEMISTRY BOSE INSTITUTE 2008 CONTENTS Page SUMMARY
Understanding the immune response to bacterial infections A Ph.D. (SCIENCE) DISSERTATION SUBMITTED TO JADAVPUR UNIVERSITY SUSHIL KUMAR PATHAK DEPARTMENT OF CHEMISTRY BOSE INSTITUTE 2008 CONTENTS Page SUMMARY
micrornas Non protein coding, endogenous RNAs of 21-22nt length Evolutionarily conserved
 microrna 2 micrornas Non protein coding, endogenous RNAs of 21-22nt length Evolutionarily conserved Regulate gene expression by binding complementary regions at 3 regions of target mrnas Act as negative
microrna 2 micrornas Non protein coding, endogenous RNAs of 21-22nt length Evolutionarily conserved Regulate gene expression by binding complementary regions at 3 regions of target mrnas Act as negative
Gene Expression Assays
 APPLICATION NOTE TaqMan Gene Expression Assays A mpl i fic ationef ficienc yof TaqMan Gene Expression Assays Assays tested extensively for qpcr efficiency Key factors that affect efficiency Efficiency
APPLICATION NOTE TaqMan Gene Expression Assays A mpl i fic ationef ficienc yof TaqMan Gene Expression Assays Assays tested extensively for qpcr efficiency Key factors that affect efficiency Efficiency
The cell lines used in this study were obtained from the American Type Culture
 Supplementary materials and methods Cell culture and drug treatments The cell lines used in this study were obtained from the American Type Culture Collection (ATCC) and grown as previously described.
Supplementary materials and methods Cell culture and drug treatments The cell lines used in this study were obtained from the American Type Culture Collection (ATCC) and grown as previously described.
Relative Quantification of mirna Target mrnas by Real-Time qpcr. 1 Introduction. Gene Expression Application Note No. 4
 Gene Expression Application Note No. 4 Relative Quantification of mirna Target mrnas by Real-Time qpcr Ute Ernst, Jitao David Zhang, Anja Irsigler, Stefan Wiemann, Ulrich Tschulena Division: Molecular
Gene Expression Application Note No. 4 Relative Quantification of mirna Target mrnas by Real-Time qpcr Ute Ernst, Jitao David Zhang, Anja Irsigler, Stefan Wiemann, Ulrich Tschulena Division: Molecular
Investigating the role of a Cryptosporidium parum apyrase in infection
 Investigating the role of a Cryptosporidium parum apyrase in infection David Riccardi and Patricio Manque Abstract This project attempted to characterize the function of a Cryptosporidium parvum apyrase
Investigating the role of a Cryptosporidium parum apyrase in infection David Riccardi and Patricio Manque Abstract This project attempted to characterize the function of a Cryptosporidium parvum apyrase
Effects of Herceptin on circulating tumor cells in HER2 positive early breast cancer
 Effects of Herceptin on circulating tumor cells in HER2 positive early breast cancer J.-L. Zhang, Q. Yao, J.-H. Chen,Y. Wang, H. Wang, Q. Fan, R. Ling, J. Yi and L. Wang Xijing Hospital Vascular Endocrine
Effects of Herceptin on circulating tumor cells in HER2 positive early breast cancer J.-L. Zhang, Q. Yao, J.-H. Chen,Y. Wang, H. Wang, Q. Fan, R. Ling, J. Yi and L. Wang Xijing Hospital Vascular Endocrine
岑 祥 股 份 有 限 公 司 技 術 專 員 費 軫 尹 20100803
 技 術 專 員 費 軫 尹 20100803 Overview of presentation Basic Biology of RNA interference Application of sirna for gene function? How to study mirna? How to deliver sirna and mirna? New prospects on RNAi research
技 術 專 員 費 軫 尹 20100803 Overview of presentation Basic Biology of RNA interference Application of sirna for gene function? How to study mirna? How to deliver sirna and mirna? New prospects on RNAi research
Supplementary Online Material for Morris et al. sirna-induced transcriptional gene
 Supplementary Online Material for Morris et al. sirna-induced transcriptional gene silencing in human cells. Materials and Methods Lentiviral vector and sirnas. FIV vector pve-gfpwp was prepared as described
Supplementary Online Material for Morris et al. sirna-induced transcriptional gene silencing in human cells. Materials and Methods Lentiviral vector and sirnas. FIV vector pve-gfpwp was prepared as described
PART 3.3: MicroRNA and Cancer
 BIBM 2010 Tutorial: Epigenomics and Cancer PART 3.3: MicroRNA and Cancer Dec 18, 2010 Sun Kim at Indiana University Outline of Part 3.3 Background on microrna Role of microrna in cancer MicroRNA pathway
BIBM 2010 Tutorial: Epigenomics and Cancer PART 3.3: MicroRNA and Cancer Dec 18, 2010 Sun Kim at Indiana University Outline of Part 3.3 Background on microrna Role of microrna in cancer MicroRNA pathway
The Need for a PARP in vivo Pharmacodynamic Assay
 The Need for a PARP in vivo Pharmacodynamic Assay Jay George, Ph.D., Chief Scientific Officer, Trevigen, Inc., Gaithersburg, MD For further infomation, please contact: William Booth, Ph.D. Tel: +44 (0)1235
The Need for a PARP in vivo Pharmacodynamic Assay Jay George, Ph.D., Chief Scientific Officer, Trevigen, Inc., Gaithersburg, MD For further infomation, please contact: William Booth, Ph.D. Tel: +44 (0)1235
Cell Cycle Phase Determination Kit
 Cell Cycle Phase Determination Kit Item No. 10009349 Customer Service 800.364.9897 * Technical Support 888.526.5351 www.caymanchem.com TABLE OF CONTENTS GENERAL INFORMATION 3 Materials Supplied 3 Safety
Cell Cycle Phase Determination Kit Item No. 10009349 Customer Service 800.364.9897 * Technical Support 888.526.5351 www.caymanchem.com TABLE OF CONTENTS GENERAL INFORMATION 3 Materials Supplied 3 Safety
RT-PCR: Two-Step Protocol
 RT-PCR: Two-Step Protocol We will provide both one-step and two-step protocols for RT-PCR. We recommend the twostep protocol for this class. In the one-step protocol, the components of RT and PCR are mixed
RT-PCR: Two-Step Protocol We will provide both one-step and two-step protocols for RT-PCR. We recommend the twostep protocol for this class. In the one-step protocol, the components of RT and PCR are mixed
Molecular analyses of EGFR: mutation and amplification detection
 Molecular analyses of EGFR: mutation and amplification detection Petra Nederlof, Moleculaire Pathologie NKI Amsterdam Henrique Ruijter, Ivon Tielen, Lucie Boerrigter, Aafke Ariaens Outline presentation
Molecular analyses of EGFR: mutation and amplification detection Petra Nederlof, Moleculaire Pathologie NKI Amsterdam Henrique Ruijter, Ivon Tielen, Lucie Boerrigter, Aafke Ariaens Outline presentation
Hands on Simulation of Mutation
 Hands on Simulation of Mutation Charlotte K. Omoto P.O. Box 644236 Washington State University Pullman, WA 99164-4236 omoto@wsu.edu ABSTRACT This exercise is a hands-on simulation of mutations and their
Hands on Simulation of Mutation Charlotte K. Omoto P.O. Box 644236 Washington State University Pullman, WA 99164-4236 omoto@wsu.edu ABSTRACT This exercise is a hands-on simulation of mutations and their
CONTRACTING ORGANIZATION: University of Alabama at Birmingham Birmingham, AL 35294
 AD Award Number: W81XWH-08-1-0030 TITLE: Regulation of Prostate Cancer Bone Metastasis by DKK1 PRINCIPAL INVESTIGATOR: Gregory A. Clines, M.D., Ph.D. CONTRACTING ORGANIZATION: University of Alabama at
AD Award Number: W81XWH-08-1-0030 TITLE: Regulation of Prostate Cancer Bone Metastasis by DKK1 PRINCIPAL INVESTIGATOR: Gregory A. Clines, M.D., Ph.D. CONTRACTING ORGANIZATION: University of Alabama at
mirnaselect pep-mir Cloning and Expression Vector
 Product Data Sheet mirnaselect pep-mir Cloning and Expression Vector CATALOG NUMBER: MIR-EXP-C STORAGE: -80ºC QUANTITY: 2 vectors; each contains 100 µl of bacterial glycerol stock Components 1. mirnaselect
Product Data Sheet mirnaselect pep-mir Cloning and Expression Vector CATALOG NUMBER: MIR-EXP-C STORAGE: -80ºC QUANTITY: 2 vectors; each contains 100 µl of bacterial glycerol stock Components 1. mirnaselect
Mutations and Genetic Variability. 1. What is occurring in the diagram below?
 Mutations and Genetic Variability 1. What is occurring in the diagram below? A. Sister chromatids are separating. B. Alleles are independently assorting. C. Genes are replicating. D. Segments of DNA are
Mutations and Genetic Variability 1. What is occurring in the diagram below? A. Sister chromatids are separating. B. Alleles are independently assorting. C. Genes are replicating. D. Segments of DNA are
Annexin V-EGFP Apoptosis Detection Kit
 ab14153 Annexin V-EGFP Apoptosis Detection Kit Instructions for Use For the rapid, sensitive and accurate measurement of apoptosis in various samples This product is for research use only and is not intended
ab14153 Annexin V-EGFP Apoptosis Detection Kit Instructions for Use For the rapid, sensitive and accurate measurement of apoptosis in various samples This product is for research use only and is not intended
pcmv6-neo Vector Application Guide Contents
 pcmv6-neo Vector Application Guide Contents Package Contents and Storage Conditions... 2 Product Description... 2 Introduction... 2 Production and Quality Assurance... 2 Methods... 3 Other required reagents...
pcmv6-neo Vector Application Guide Contents Package Contents and Storage Conditions... 2 Product Description... 2 Introduction... 2 Production and Quality Assurance... 2 Methods... 3 Other required reagents...
The heart specimens were fixed with formalin, embedded in paraffin, and sectioned at 6-
 Hsu et al. 2010/958033 R2 SUPPLEMENTAL MATERIALS Supplemental methods: Immunohistochemistry The heart specimens were fixed with formalin, embedded in paraffin, and sectioned at 6- μm thickness. Formalin-fixed
Hsu et al. 2010/958033 R2 SUPPLEMENTAL MATERIALS Supplemental methods: Immunohistochemistry The heart specimens were fixed with formalin, embedded in paraffin, and sectioned at 6- μm thickness. Formalin-fixed
SERVICES CATALOGUE WITH SUBMISSION GUIDELINES
 SERVICES CATALOGUE WITH SUBMISSION GUIDELINES 3921 Montgomery Road Cincinnati, Ohio 45212 513-841-2428 www.agctsequencing.com CONTENTS Welcome Dye Terminator Sequencing DNA Sequencing Services - Full Service
SERVICES CATALOGUE WITH SUBMISSION GUIDELINES 3921 Montgomery Road Cincinnati, Ohio 45212 513-841-2428 www.agctsequencing.com CONTENTS Welcome Dye Terminator Sequencing DNA Sequencing Services - Full Service
No Disclosures. Learning Objectives 10/25/13
 No Disclosures Gregory A. Brent, MD Departments of Medicine and Physiology David Geffen School of Medicine at UCLA VA Greater Los Angeles Healthcare System Learning Objectives Describe the pathways that
No Disclosures Gregory A. Brent, MD Departments of Medicine and Physiology David Geffen School of Medicine at UCLA VA Greater Los Angeles Healthcare System Learning Objectives Describe the pathways that
OriGene Technologies, Inc. MicroRNA analysis: Detection, Perturbation, and Target Validation
 OriGene Technologies, Inc. MicroRNA analysis: Detection, Perturbation, and Target Validation -Optimal strategies to a successful mirna research project Optimal strategies to a successful mirna research
OriGene Technologies, Inc. MicroRNA analysis: Detection, Perturbation, and Target Validation -Optimal strategies to a successful mirna research project Optimal strategies to a successful mirna research
Instructions. Torpedo sirna. Material. Important Guidelines. Specifications. Quality Control
 is a is a state of the art transfection reagent, specifically designed for the transfer of sirna and mirna into a variety of eukaryotic cell types. is a state of the art transfection reagent, specifically
is a is a state of the art transfection reagent, specifically designed for the transfer of sirna and mirna into a variety of eukaryotic cell types. is a state of the art transfection reagent, specifically
Inhibition of MicroRNA Sensitizes 3D Breast Cancer Microtissues to Radiation Therapy
 CASE STUDY High Content Image Analysis Inhibition of MicroRNA Sensitizes 3D Breast Cancer Microtissues to Radiation Therapy Dr. Nataša Anastasov leads the Personalized Radiation Therapy Group within the
CASE STUDY High Content Image Analysis Inhibition of MicroRNA Sensitizes 3D Breast Cancer Microtissues to Radiation Therapy Dr. Nataša Anastasov leads the Personalized Radiation Therapy Group within the
Gene Synthesis 191. Mutagenesis 194. Gene Cloning 196. AccuGeneBlock Service 198. Gene Synthesis FAQs 201. User Protocol 204
 Gene Synthesis 191 Mutagenesis 194 Gene Cloning 196 AccuGeneBlock Service 198 Gene Synthesis FAQs 201 User Protocol 204 Gene Synthesis Overview Gene synthesis is the most cost-effective way to enhance
Gene Synthesis 191 Mutagenesis 194 Gene Cloning 196 AccuGeneBlock Service 198 Gene Synthesis FAQs 201 User Protocol 204 Gene Synthesis Overview Gene synthesis is the most cost-effective way to enhance
Supplemental Data. Short Article. PPARγ Activation Primes Human Monocytes. into Alternative M2 Macrophages. with Anti-inflammatory Properties
 Cell Metabolism, Volume 6 Supplemental Data Short Article PPARγ Activation Primes Human Monocytes into Alternative M2 Macrophages with Anti-inflammatory Properties M. Amine Bouhlel, Bruno Derudas, Elena
Cell Metabolism, Volume 6 Supplemental Data Short Article PPARγ Activation Primes Human Monocytes into Alternative M2 Macrophages with Anti-inflammatory Properties M. Amine Bouhlel, Bruno Derudas, Elena
First Strand cdna Synthesis
 380PR 01 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name First Strand cdna Synthesis (Cat. # 786 812) think proteins! think G-Biosciences
380PR 01 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name First Strand cdna Synthesis (Cat. # 786 812) think proteins! think G-Biosciences
The p53 MUTATION HANDBOOK
 The p MUTATION HANDBOOK Version 1. /7 Thierry Soussi Christophe Béroud, Dalil Hamroun Jean Michel Rubio Nevado http://p/free.fr The p Mutation HandBook By T Soussi, J.M. Rubio-Nevado, D. Hamroun and C.
The p MUTATION HANDBOOK Version 1. /7 Thierry Soussi Christophe Béroud, Dalil Hamroun Jean Michel Rubio Nevado http://p/free.fr The p Mutation HandBook By T Soussi, J.M. Rubio-Nevado, D. Hamroun and C.
Annexin V-FITC Apoptosis Detection Kit
 ab14085 Annexin V-FITC Apoptosis Detection Kit Instructions for Use For the rapid, sensitive and accurate measurement of Apoptosis in living cells (adherent and suspension). This product is for research
ab14085 Annexin V-FITC Apoptosis Detection Kit Instructions for Use For the rapid, sensitive and accurate measurement of Apoptosis in living cells (adherent and suspension). This product is for research
Mir-X mirna First-Strand Synthesis Kit User Manual
 User Manual Mir-X mirna First-Strand Synthesis Kit User Manual United States/Canada 800.662.2566 Asia Pacific +1.650.919.7300 Europe +33.(0)1.3904.6880 Japan +81.(0)77.543.6116 Clontech Laboratories, Inc.
User Manual Mir-X mirna First-Strand Synthesis Kit User Manual United States/Canada 800.662.2566 Asia Pacific +1.650.919.7300 Europe +33.(0)1.3904.6880 Japan +81.(0)77.543.6116 Clontech Laboratories, Inc.
Recombinant DNA and Biotechnology
 Recombinant DNA and Biotechnology Chapter 18 Lecture Objectives What Is Recombinant DNA? How Are New Genes Inserted into Cells? What Sources of DNA Are Used in Cloning? What Other Tools Are Used to Study
Recombinant DNA and Biotechnology Chapter 18 Lecture Objectives What Is Recombinant DNA? How Are New Genes Inserted into Cells? What Sources of DNA Are Used in Cloning? What Other Tools Are Used to Study
PNA BRAF Mutation Detection Kit
 - PNA BRAF Mutation Detection Kit Catalog Number KA2102 50 tests/kit Version: 01 Intended for research use only www.abnova.com Introduction and Background Intended use The PNA BRAF Mutation Detection Kit
- PNA BRAF Mutation Detection Kit Catalog Number KA2102 50 tests/kit Version: 01 Intended for research use only www.abnova.com Introduction and Background Intended use The PNA BRAF Mutation Detection Kit
Annexin V-FITC Apoptosis Detection Kit
 ab14085 Annexin V-FITC Apoptosis Detection Kit Instructions for Use For the rapid, sensitive and accurate measurement of Apoptosis in living cells (adherent and suspension). This product is for research
ab14085 Annexin V-FITC Apoptosis Detection Kit Instructions for Use For the rapid, sensitive and accurate measurement of Apoptosis in living cells (adherent and suspension). This product is for research
Introduction To Real Time Quantitative PCR (qpcr)
 Introduction To Real Time Quantitative PCR (qpcr) SABiosciences, A QIAGEN Company www.sabiosciences.com The Seminar Topics The advantages of qpcr versus conventional PCR Work flow & applications Factors
Introduction To Real Time Quantitative PCR (qpcr) SABiosciences, A QIAGEN Company www.sabiosciences.com The Seminar Topics The advantages of qpcr versus conventional PCR Work flow & applications Factors
INTERPRETATION INFORMATION SHEET
 Creative Testing Solutions 2424 West Erie Dr. 2205 Highway 121 10100 Martin Luther King Jr. St. No. Tempe, AZ 85282 Bedford, TX 76021 St. Petersburg, FL 33716 INTERPRETATION INFORMATION SHEET Human Immunodeficiency
Creative Testing Solutions 2424 West Erie Dr. 2205 Highway 121 10100 Martin Luther King Jr. St. No. Tempe, AZ 85282 Bedford, TX 76021 St. Petersburg, FL 33716 INTERPRETATION INFORMATION SHEET Human Immunodeficiency
APOT - Assay. Protocol for HPV16 and 18. Amplification of Papilloma Virus Oncogene Transcripts HPV. E6 E7 E1 Zelluläre DNA poly(a)
 E5 E2 E1 APOT - Assay Amplification of Papilloma Virus Oncogene Transcripts URR E6 E7 L1 HPV L2 E4 E6 E7 E1 Zelluläre DNA poly(a) Protocol for HPV16 and 18 Brief summary of the APOT assay Fig.1A shows
E5 E2 E1 APOT - Assay Amplification of Papilloma Virus Oncogene Transcripts URR E6 E7 L1 HPV L2 E4 E6 E7 E1 Zelluläre DNA poly(a) Protocol for HPV16 and 18 Brief summary of the APOT assay Fig.1A shows
Application Note No. 2 / July 2012. Quantitative Assessment of Cell Quality, Viability and Proliferation. System
 Application Note No. 2 / July 2012 Quantitative Assessment of Cell Quality, Viability and Proliferation System Quantitative Assessment of Cell Quality, Viability and Proliferation Introduction In vitro
Application Note No. 2 / July 2012 Quantitative Assessment of Cell Quality, Viability and Proliferation System Quantitative Assessment of Cell Quality, Viability and Proliferation Introduction In vitro
Transfection reagent for visualizing lipofection with DNA. For ordering information, MSDS, publications and application notes see www.biontex.
 METAFECTENE FluoR Transfection reagent for visualizing lipofection with DNA For ordering information, MSDS, publications and application notes see www.biontex.com Description Cat. No. Size METAFECTENE
METAFECTENE FluoR Transfection reagent for visualizing lipofection with DNA For ordering information, MSDS, publications and application notes see www.biontex.com Description Cat. No. Size METAFECTENE
Supplementary Figure 1.
 Supplementary Figure 1. (A) MicroRNA 212 enhances IS from pancreatic β-cells. INS-1 832/3 β-cells were transfected with precursors for mirnas 212, 375, or negative control oligonucleotides. 48 hrs after
Supplementary Figure 1. (A) MicroRNA 212 enhances IS from pancreatic β-cells. INS-1 832/3 β-cells were transfected with precursors for mirnas 212, 375, or negative control oligonucleotides. 48 hrs after
Thermo Scientific DyNAmo cdna Synthesis Kit for qrt-pcr Technical Manual
 Thermo Scientific DyNAmo cdna Synthesis Kit for qrt-pcr Technical Manual F- 470S 20 cdna synthesis reactions (20 µl each) F- 470L 100 cdna synthesis reactions (20 µl each) Table of contents 1. Description...
Thermo Scientific DyNAmo cdna Synthesis Kit for qrt-pcr Technical Manual F- 470S 20 cdna synthesis reactions (20 µl each) F- 470L 100 cdna synthesis reactions (20 µl each) Table of contents 1. Description...
2.1.2 Characterization of antiviral effect of cytokine expression on HBV replication in transduced mouse hepatocytes line
 i 1 INTRODUCTION 1.1 Human Hepatitis B virus (HBV) 1 1.1.1 Pathogenesis of Hepatitis B 1 1.1.2 Genome organization of HBV 3 1.1.3 Structure of HBV virion 5 1.1.4 HBV life cycle 5 1.1.5 Experimental models
i 1 INTRODUCTION 1.1 Human Hepatitis B virus (HBV) 1 1.1.1 Pathogenesis of Hepatitis B 1 1.1.2 Genome organization of HBV 3 1.1.3 Structure of HBV virion 5 1.1.4 HBV life cycle 5 1.1.5 Experimental models
HiPer RT-PCR Teaching Kit
 HiPer RT-PCR Teaching Kit Product Code: HTBM024 Number of experiments that can be performed: 5 Duration of Experiment: Protocol: 4 hours Agarose Gel Electrophoresis: 45 minutes Storage Instructions: The
HiPer RT-PCR Teaching Kit Product Code: HTBM024 Number of experiments that can be performed: 5 Duration of Experiment: Protocol: 4 hours Agarose Gel Electrophoresis: 45 minutes Storage Instructions: The
Title : Parallel DNA Synthesis : Two PCR product from one DNA template
 Title : Parallel DNA Synthesis : Two PCR product from one DNA template Bhardwaj Vikash 1 and Sharma Kulbhushan 2 1 Email: vikashbhardwaj@ gmail.com 1 Current address: Government College Sector 14 Gurgaon,
Title : Parallel DNA Synthesis : Two PCR product from one DNA template Bhardwaj Vikash 1 and Sharma Kulbhushan 2 1 Email: vikashbhardwaj@ gmail.com 1 Current address: Government College Sector 14 Gurgaon,
Protein kinase C alpha expression and resistance to neo-adjuvant gemcitabine-containing chemotherapy in non-small cell lung cancer
 Protein kinase C alpha expression and resistance to neo-adjuvant gemcitabine-containing chemotherapy in non-small cell lung cancer Dan Vogl Lay Abstract Early stage non-small cell lung cancer can be cured
Protein kinase C alpha expression and resistance to neo-adjuvant gemcitabine-containing chemotherapy in non-small cell lung cancer Dan Vogl Lay Abstract Early stage non-small cell lung cancer can be cured
TITRATION OF raav (VG) USING QUANTITATIVE REAL TIME PCR
 Page 1 of 5 Materials DNase digestion buffer [13 mm Tris-Cl, ph7,5 / 5 mm MgCl2 / 0,12 mm CaCl2] RSS plasmid ptr-uf11 SV40pA Forward primer (10µM) AGC AAT AGC ATC ACA AAT TTC ACA A SV40pA Reverse Primer
Page 1 of 5 Materials DNase digestion buffer [13 mm Tris-Cl, ph7,5 / 5 mm MgCl2 / 0,12 mm CaCl2] RSS plasmid ptr-uf11 SV40pA Forward primer (10µM) AGC AAT AGC ATC ACA AAT TTC ACA A SV40pA Reverse Primer
Taqman TCID50 for AAV Vector Infectious Titer Determination
 Page 1 of 8 Purpose: To determine the concentration of infectious particles in an AAV vector sample. This process involves serial dilution of the vector in a TCID50 format and endpoint determination through
Page 1 of 8 Purpose: To determine the concentration of infectious particles in an AAV vector sample. This process involves serial dilution of the vector in a TCID50 format and endpoint determination through
Supplementary Information. Binding region and interaction properties of sulfoquinovosylacylglycerol (SQAG) with human
 Supplementary Information Binding region and interaction properties of sulfoquinovosylacylglycerol (SQAG) with human vascular endothelial growth factor 165 revealed by biosensor based assays Yoichi Takakusagi
Supplementary Information Binding region and interaction properties of sulfoquinovosylacylglycerol (SQAG) with human vascular endothelial growth factor 165 revealed by biosensor based assays Yoichi Takakusagi
Fast, easy and effective transfection reagent for mammalian cells
 METAFECTENE EASY + Fast, easy and effective transfection reagent for mammalian cells For ordering information, MSDS, publications and application notes see www.biontex.com Description Cat. No. Size METAFECTENE
METAFECTENE EASY + Fast, easy and effective transfection reagent for mammalian cells For ordering information, MSDS, publications and application notes see www.biontex.com Description Cat. No. Size METAFECTENE
Introduction to Perl Programming Input/Output, Regular Expressions, String Manipulation. Beginning Perl, Chap 4 6. Example 1
 Introduction to Perl Programming Input/Output, Regular Expressions, String Manipulation Beginning Perl, Chap 4 6 Example 1 #!/usr/bin/perl -w use strict; # version 1: my @nt = ('A', 'C', 'G', 'T'); for
Introduction to Perl Programming Input/Output, Regular Expressions, String Manipulation Beginning Perl, Chap 4 6 Example 1 #!/usr/bin/perl -w use strict; # version 1: my @nt = ('A', 'C', 'G', 'T'); for
Contact Information Korea Health Industry Development Institute
 AGM-130, a novel CDK inhibitor with a marked anti-tumor activity and reduced toxicity, targeting the triple negative breast cancer(tnbc) and tamoxifen-resistant estrogen receptor (ER) positive breast cancer
AGM-130, a novel CDK inhibitor with a marked anti-tumor activity and reduced toxicity, targeting the triple negative breast cancer(tnbc) and tamoxifen-resistant estrogen receptor (ER) positive breast cancer
Heraeus Sepatech, Kendro Laboratory Products GmbH, Berlin. Becton Dickinson,Heidelberg. Biozym, Hessisch Oldendorf. Eppendorf, Hamburg
 13 4. MATERIALS 4.1 Laboratory apparatus Biofuge A Centrifuge 5804R FACScan Gel electrophoresis chamber GPR Centrifuge Heraeus CO-AUTO-ZERO Light Cycler Microscope Motopipet Neubauer Cell Chamber PCR cycler
13 4. MATERIALS 4.1 Laboratory apparatus Biofuge A Centrifuge 5804R FACScan Gel electrophoresis chamber GPR Centrifuge Heraeus CO-AUTO-ZERO Light Cycler Microscope Motopipet Neubauer Cell Chamber PCR cycler
Targeted Therapy What the Surgeon Needs to Know
 Targeted Therapy What the Surgeon Needs to Know AATS Focus in Thoracic Surgery 2014 David R. Jones, M.D. Professor & Chief, Thoracic Surgery Memorial Sloan Kettering Cancer Center I have no disclosures
Targeted Therapy What the Surgeon Needs to Know AATS Focus in Thoracic Surgery 2014 David R. Jones, M.D. Professor & Chief, Thoracic Surgery Memorial Sloan Kettering Cancer Center I have no disclosures
Supplemental Fig. S1. The schematic diagrams of the expression constructs used in this study.
 1 Supplemental data Supplemental Fig. S1. The schematic diagrams of the expression constructs used in this study. Supplemental Fig. S2. Ingenuity Pathway Analysis (IPA) of the 56 putative caspase substrates
1 Supplemental data Supplemental Fig. S1. The schematic diagrams of the expression constructs used in this study. Supplemental Fig. S2. Ingenuity Pathway Analysis (IPA) of the 56 putative caspase substrates
MystiCq microrna cdna Synthesis Mix Catalog Number MIRRT Storage Temperature 20 C
 microrna cdna Synthesis Mix Catalog Number MIRRT Storage Temperature 20 C Product Description The microrna cdna Synthesis Mix has been designed to easily convert micrornas into cdna templates for qpcr
microrna cdna Synthesis Mix Catalog Number MIRRT Storage Temperature 20 C Product Description The microrna cdna Synthesis Mix has been designed to easily convert micrornas into cdna templates for qpcr
TECHNICAL INSIGHTS TECHNOLOGY ALERT
 TECHNICAL INSIGHTS TECHNOLOGY ALERT UNLOCKING BREAST CANCER TUMORS' DRUG RESISTANCE A troubling phenomenon in breast cancer treatment is that some patients tumors become resistant to treatments over time.
TECHNICAL INSIGHTS TECHNOLOGY ALERT UNLOCKING BREAST CANCER TUMORS' DRUG RESISTANCE A troubling phenomenon in breast cancer treatment is that some patients tumors become resistant to treatments over time.
Preliminary Results from a Phase 2 Study of ARQ 197 in Patients with Microphthalmia Transcription Factor Family (MiT) Associated Tumors
 Preliminary Results from a Phase 2 Study of ARQ 197 in Patients with Microphthalmia Transcription Factor Family (MiT) Associated Tumors John Goldberg 1 *, George Demetri 2, Edwin Choy 3, Lee Rosen 4, Alberto
Preliminary Results from a Phase 2 Study of ARQ 197 in Patients with Microphthalmia Transcription Factor Family (MiT) Associated Tumors John Goldberg 1 *, George Demetri 2, Edwin Choy 3, Lee Rosen 4, Alberto
use. 3,5 Apoptosis Viability Titration Assays
 Introduction Monitoring cell viability can provide critical insights into the effectiveness of biological protocols and the stability of biological systems including: the efficacy of cell harvesting procedures,
Introduction Monitoring cell viability can provide critical insights into the effectiveness of biological protocols and the stability of biological systems including: the efficacy of cell harvesting procedures,
hydrocortisone (5 mg/ml), EGF (10 µg/ml) and Heparin (5000 U/ml). Antibodies against the N-terminal peptide of MEK1 (MEK1-N) and against Flotillin 1
 SUPPLEMENTARY METHODS Cells HUVEC (Human Umbilical Vein Endothelial Cells) were grown in complete Medium 199 (Gibco) supplemented with glutamax, 10% foetal calf serum, BBE (9 mg/ml), hydrocortisone (5
SUPPLEMENTARY METHODS Cells HUVEC (Human Umbilical Vein Endothelial Cells) were grown in complete Medium 199 (Gibco) supplemented with glutamax, 10% foetal calf serum, BBE (9 mg/ml), hydrocortisone (5
Dot Blot Analysis. Teacher s Guidebook. (Cat. # BE 502) think proteins! think G-Biosciences www.gbiosciences.com
 PR110 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Dot Blot Analysis Teacher s Guidebook (Cat. # BE 502) think proteins! think G-Biosciences
PR110 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Dot Blot Analysis Teacher s Guidebook (Cat. # BE 502) think proteins! think G-Biosciences
MEF Nucleofector Kit 1 and 2
 page 1 of 7 MEF Nucleofector Kit 1 and 2 for Mouse Embryonic Fibroblasts (MEF) MEF display significant phenotypic variations which depend on the strain, the genetic background of the they are isolated
page 1 of 7 MEF Nucleofector Kit 1 and 2 for Mouse Embryonic Fibroblasts (MEF) MEF display significant phenotypic variations which depend on the strain, the genetic background of the they are isolated
High-quality genomic DNA isolation and sensitive mutation analysis
 Application Note High-quality genomic DNA isolation and sensitive mutation analysis Izabela Safin, Ivonne Schröder-Stumberger and Peter Porschewski Introduction A major objective of cancer research is
Application Note High-quality genomic DNA isolation and sensitive mutation analysis Izabela Safin, Ivonne Schröder-Stumberger and Peter Porschewski Introduction A major objective of cancer research is
Amaxa Mouse T Cell Nucleofector Kit
 Amaxa Mouse T Cell Nucleofector Kit For T cells isolated from C57BL/6 & BALB/c mice Evaluated for murine T cells isolated from C57BL/6 & BALB/c mice This protocol is designed for murine lymphocytes or
Amaxa Mouse T Cell Nucleofector Kit For T cells isolated from C57BL/6 & BALB/c mice Evaluated for murine T cells isolated from C57BL/6 & BALB/c mice This protocol is designed for murine lymphocytes or
Supporting Information
 Supporting Information Kondo et al. 1.173/pnas.787415 SI Methods Conventional and Quantitative RT-PCR. Total RNA was extracted from cultured ES cells or ES-derived cells using the RNeasy Minikit (Qiagen).
Supporting Information Kondo et al. 1.173/pnas.787415 SI Methods Conventional and Quantitative RT-PCR. Total RNA was extracted from cultured ES cells or ES-derived cells using the RNeasy Minikit (Qiagen).
Real-Time PCR Vs. Traditional PCR
 Real-Time PCR Vs. Traditional PCR Description This tutorial will discuss the evolution of traditional PCR methods towards the use of Real-Time chemistry and instrumentation for accurate quantitation. Objectives
Real-Time PCR Vs. Traditional PCR Description This tutorial will discuss the evolution of traditional PCR methods towards the use of Real-Time chemistry and instrumentation for accurate quantitation. Objectives
Head of College Scholars List Scheme. Summer Studentship. Report Form
 Head of College Scholars List Scheme Summer Studentship Report Form This report should be completed by the student with his/her project supervisor. It should summarise the work undertaken during the project
Head of College Scholars List Scheme Summer Studentship Report Form This report should be completed by the student with his/her project supervisor. It should summarise the work undertaken during the project
Chapter 8. Summary and Perspectives
 Chapter 8 Summary and Perspectives 131 Chapter 8 Summary Overexpression of the multidrug resistance protein MRP1 confer multidrug resistance (MDR) to cancer cells. The contents of this thesis describe
Chapter 8 Summary and Perspectives 131 Chapter 8 Summary Overexpression of the multidrug resistance protein MRP1 confer multidrug resistance (MDR) to cancer cells. The contents of this thesis describe
a Beckman Coulter Life Sciences: White Paper
 a Beckman Coulter Life Sciences: White Paper Violet-Excited nim-da Allows Efficient and Reproducible Cell Cycle Analysis on the Gallios Flow Cytometer Authors: Valdez, Ben 1. Carr, Karen 2. Norman, John
a Beckman Coulter Life Sciences: White Paper Violet-Excited nim-da Allows Efficient and Reproducible Cell Cycle Analysis on the Gallios Flow Cytometer Authors: Valdez, Ben 1. Carr, Karen 2. Norman, John
THE His Tag Antibody, mab, Mouse
 THE His Tag Antibody, mab, Mouse Cat. No. A00186 Technical Manual No. TM0243 Update date 01052011 I Description.... 1 II Key Features. 2 III Storage 2 IV Applications.... 2 V Examples - ELISA..... 2 VI
THE His Tag Antibody, mab, Mouse Cat. No. A00186 Technical Manual No. TM0243 Update date 01052011 I Description.... 1 II Key Features. 2 III Storage 2 IV Applications.... 2 V Examples - ELISA..... 2 VI
Essentials of Real Time PCR. About Sequence Detection Chemistries
 Essentials of Real Time PCR About Real-Time PCR Assays Real-time Polymerase Chain Reaction (PCR) is the ability to monitor the progress of the PCR as it occurs (i.e., in real time). Data is therefore collected
Essentials of Real Time PCR About Real-Time PCR Assays Real-time Polymerase Chain Reaction (PCR) is the ability to monitor the progress of the PCR as it occurs (i.e., in real time). Data is therefore collected
PROTOCOL. Immunocytochemistry (ICC) MATERIALS AND EQUIPMENT REQUIRED
 PROTOCOL Immunocytochemistry (ICC) 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 11-07 MATERIALS AND EQUIPMENT REQUIRED Materials: MitoSciences primary monoclonal antibody/antibodies Fluorophore-conjugated
PROTOCOL Immunocytochemistry (ICC) 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 11-07 MATERIALS AND EQUIPMENT REQUIRED Materials: MitoSciences primary monoclonal antibody/antibodies Fluorophore-conjugated
Future Oncology: Technology, Products, Market and Service Opportunities
 Brochure More information from http://www.researchandmarkets.com/reports/296370/ Future Oncology: Technology, Products, Market and Service Opportunities Description: Future Oncology is an analytical newsletter
Brochure More information from http://www.researchandmarkets.com/reports/296370/ Future Oncology: Technology, Products, Market and Service Opportunities Description: Future Oncology is an analytical newsletter
Real-time monitoring of rolling circle amplification using aggregation-induced emission: applications for biological detection
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 215 Supplementary Information Real-time monitoring of rolling circle amplification using aggregation-induced
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 215 Supplementary Information Real-time monitoring of rolling circle amplification using aggregation-induced
MicroRNA formation. 4th International Symposium on Non-Surgical Contraceptive Methods of Pet Population Control
 MicroRNA formation mirna s are processed from several precursor stages Mammalian genomes seem to have 100 s of mirna s Nucleotides in positions 2-8 of an mirna are considered the mirna seed 5 Methyl-G
MicroRNA formation mirna s are processed from several precursor stages Mammalian genomes seem to have 100 s of mirna s Nucleotides in positions 2-8 of an mirna are considered the mirna seed 5 Methyl-G
Transfection-Transfer of non-viral genetic material into eukaryotic cells. Infection/ Transduction- Transfer of viral genetic material into cells.
 Transfection Key words: Transient transfection, Stable transfection, transfection methods, vector, plasmid, origin of replication, reporter gene/ protein, cloning site, promoter and enhancer, signal peptide,
Transfection Key words: Transient transfection, Stable transfection, transfection methods, vector, plasmid, origin of replication, reporter gene/ protein, cloning site, promoter and enhancer, signal peptide,
ab185915 Protein Sumoylation Assay Ultra Kit
 ab185915 Protein Sumoylation Assay Ultra Kit Instructions for Use For the measuring in vivo protein sumoylation in various samples This product is for research use only and is not intended for diagnostic
ab185915 Protein Sumoylation Assay Ultra Kit Instructions for Use For the measuring in vivo protein sumoylation in various samples This product is for research use only and is not intended for diagnostic
CFSE Cell Division Assay Kit
 CFSE Cell Division Assay Kit Item No. 10009853 Customer Service 800.364.9897 * Technical Support 888.526.5351 www.caymanchem.com TABLE OF CONTENTS GENERAL INFORMATION 3 Materials Supplied 4 Precautions
CFSE Cell Division Assay Kit Item No. 10009853 Customer Service 800.364.9897 * Technical Support 888.526.5351 www.caymanchem.com TABLE OF CONTENTS GENERAL INFORMATION 3 Materials Supplied 4 Precautions
CytoSelect LDH Cytotoxicity Assay Kit
 Product Manual CytoSelect LDH Cytotoxicity Assay Kit Catalog Number CBA-241 960 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction The measurement and monitoring of cell cytotoxicity
Product Manual CytoSelect LDH Cytotoxicity Assay Kit Catalog Number CBA-241 960 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction The measurement and monitoring of cell cytotoxicity
sirna Duplexes & RNAi Explorer
 P r o d u c t S p e c i f i c a t i o n s Guaranteed RNAi Explorer kit with Fl/Dabcyl Molecular Beacon Catalog No.:27-6402-01 Guaranteed RNAi Explorer kit with Fluorescein/Tamra TaqMan Catalog No.:27-6402-01
P r o d u c t S p e c i f i c a t i o n s Guaranteed RNAi Explorer kit with Fl/Dabcyl Molecular Beacon Catalog No.:27-6402-01 Guaranteed RNAi Explorer kit with Fluorescein/Tamra TaqMan Catalog No.:27-6402-01
Product: Expression Arrest TM egfp control shrna vector
 Product: Expression Arrest TM egfp control vector Catalog #: RHS1702 Product Description The laboratory of Dr. Greg Hannon at Cold Spring Harbor Laboratory (CSHL) has created an RNAi Clone Library comprised
Product: Expression Arrest TM egfp control vector Catalog #: RHS1702 Product Description The laboratory of Dr. Greg Hannon at Cold Spring Harbor Laboratory (CSHL) has created an RNAi Clone Library comprised
TOOLS sirna and mirna. User guide
 TOOLS sirna and mirna User guide Introduction RNA interference (RNAi) is a powerful tool for suppression gene expression by causing the destruction of specific mrna molecules. Small Interfering RNAs (sirnas)
TOOLS sirna and mirna User guide Introduction RNA interference (RNAi) is a powerful tool for suppression gene expression by causing the destruction of specific mrna molecules. Small Interfering RNAs (sirnas)
CD22 Antigen Is Broadly Expressed on Lung Cancer Cells and Is a Target for Antibody-Based Therapy
 CD22 Antigen Is Broadly Expressed on Lung Cancer Cells and Is a Target for Antibody-Based Therapy Joseph M. Tuscano, Jason Kato, David Pearson, Chengyi Xiong, Laura Newell, Yunpeng Ma, David R. Gandara,
CD22 Antigen Is Broadly Expressed on Lung Cancer Cells and Is a Target for Antibody-Based Therapy Joseph M. Tuscano, Jason Kato, David Pearson, Chengyi Xiong, Laura Newell, Yunpeng Ma, David R. Gandara,
Supplemental Information. McBrayer et al. Supplemental Data
 1 Supplemental Information McBrayer et al. Supplemental Data 2 Figure S1. Glucose consumption rates of MM cell lines exceed that of normal PBMC. (A) Normal PBMC isolated from three healthy donors were
1 Supplemental Information McBrayer et al. Supplemental Data 2 Figure S1. Glucose consumption rates of MM cell lines exceed that of normal PBMC. (A) Normal PBMC isolated from three healthy donors were
Thermo Scientific Phusion Site-Directed Mutagenesis Kit #F-541
 PRODUCT INFORMATION Thermo Scientific Phusion Site-Directed Mutagenesis Kit #F-541 Lot _ Store at -20 C Expiry Date _ www.thermoscientific.com/onebio CERTIFICATE OF ANALYSIS The Phusion Site-Directed Mutagenesis
PRODUCT INFORMATION Thermo Scientific Phusion Site-Directed Mutagenesis Kit #F-541 Lot _ Store at -20 C Expiry Date _ www.thermoscientific.com/onebio CERTIFICATE OF ANALYSIS The Phusion Site-Directed Mutagenesis
CompleteⅡ 1st strand cdna Synthesis Kit
 Instruction Manual CompleteⅡ 1st strand cdna Synthesis Kit Catalog # GM30401, GM30402 Green Mountain Biosystems. LLC Web: www.greenmountainbio.com Tel: 800-942-1160 Sales: Sales@ greenmountainbio.com Support:
Instruction Manual CompleteⅡ 1st strand cdna Synthesis Kit Catalog # GM30401, GM30402 Green Mountain Biosystems. LLC Web: www.greenmountainbio.com Tel: 800-942-1160 Sales: Sales@ greenmountainbio.com Support:
Mir-X mirna First-Strand Synthesis and SYBR qrt-pcr
 User Manual Mir-X mirna First-Strand Synthesis and SYBR qrt-pcr User Manual United States/Canada 800.662.2566 Asia Pacific +1.650.919.7300 Europe +33.(0)1.3904.6880 Japan +81.(0)77.543.6116 Clontech Laboratories,
User Manual Mir-X mirna First-Strand Synthesis and SYBR qrt-pcr User Manual United States/Canada 800.662.2566 Asia Pacific +1.650.919.7300 Europe +33.(0)1.3904.6880 Japan +81.(0)77.543.6116 Clontech Laboratories,
