Principle of Formation of Magnetic Field of Iron
|
|
|
- Brittney Bruce
- 7 years ago
- Views:
Transcription
1 Principle of Formation of Magnetic Field of Iron Anatoli Bedritsky Abstract. This article discovers physical phenomena which occur at the formation of the magnetic field of iron and the reason of an attraction and pushing away of two constant magnets. Given article is a part of Real theoretical physics, which I have done on basis of existence of an ether. 1 Action of Magnetic Field on Proton Orbits of Atoms of Iron The electric current cannot create a magnetic field of such strength as the constant magnets. Therefore, the orbital electrons of atoms also cannot create the magnetic field of magnetized iron. The magnetic field of magnetized iron can be created only by rotated protons of atoms of iron. But this magnetic field can be created only in case, if greater amount of protons have the magnetic fields in identical direction than in opposite direction. The atom of iron consists of a nucleus, which consists of 30 neutrons and 2 protons. Around of a nucleus 24 orbital protons on three orbits are moving. The first proton orbit consist of 8 protons, the second orbit consist of 14 protons and the third orbit consist of 2 protons. On the first orbit around of the nucleus move two orbital electrons, which have an attraction directly to the nucleus. Other orbital electrons also move around of the nucleus, but everyone electron is attracted to the nucleus through one of orbital protons. Thus above everyone orbital proton, one orbital electron moves synchronously. Crystals of iron have a cubic volumetric-aligned lattice. The cell of a crystal of iron consists of eight angular atoms, which are forming a cube, and of one central atom. The angular atoms are general for the nearby cells 1
2 of a crystal. The orbital protons have a attraction not only to the nucleus of their atom, but also to the nucleuses of the nearby atoms. Therefore the proton and electron orbits located in a plane which are passing through the nucleuses of the nearby atoms, but not between them. As orbital protons of atom have the left direction of rotation, and orbital electrons have the right direction of rotation, their magnetic fields are directed counter, but due to that that the strength of magnetic field of protons is much more than the strength of magnetic field of electrons, then the general magnetic field of separate atom has a great strength. But the orbital protons of next atoms of a crystal have a miscellaneous and opposite direction of orbital motion, therefore the magnetic fields of these atoms have opposite direction and are mutually destroyed. In amorphous bodies the molecules are located miscellaneous in the relation to each other and consequently the general magnetic field also is not formed. If iron to place in a magnetic field, then the proton orbits and the electronic orbits of the central atoms deviate in a direction of a magnetic field and will have identical orientation. In this case the magnetic field, formed by orbital protons of the central atoms will have an identical direction, forming the general magnetic field of iron. If to remove the iron from the magnetic field, then the orbits of the central atoms will not be located equally and due to that the general magnetic field will disappear. The iron body having a constant magnetic field represent itself a constant magnet. The constant magnets are made of a special alloy of iron with additives of other element. This iron in a magnetic field gets a magnetic properties which remain after an exit of iron from the magnetic field. On fig.?? above is shown the connection of atoms in a crystal of iron before action of a magnetic field on this iron, and on fig. below is shown the connection of atoms in a crystal of iron after action on him of a magnetic field. Apparently, at action of a magnetic field on iron, the orbits with protons and electrons of central atoms turn on 90 degrees and they are established by on a new diagonal of the cubic cell. But the orbits of angular atoms of the cubic cell do not turn, because the orbital protons of angular atoms have stable position. Stability of position of orbits is formed due to that that on a way of orbital motion of protons is formed rarefied ether, and also it occurs due to that that other possible positions of orbital are occupied by orbits of the next atoms. Turn of orbits of central atoms is possible due to that what the orbits of the central atom has an identical opportunity of location on all diagonals 2
3 Proton and electron orbits of atoms Nucleuses of atoms Diagonal orbits of central atoms Denotation of nucleus and orbits of separate atom Figure 1: Connection of atoms in a crystal of iron. 3
4 of a cubic cell. Turn of orbits of the central atoms occurs due to the action of a magnetic field on the rotated orbital protons. An axis of rotation of orbital protons is the line of their orbital motion. Apparently, as a result of action of a magnetic field on iron, the orbits of the central atoms of cells of a crystal are established in parallel with an identical direction of motion, due to that the orbital protons of these orbits are forming a magnetic field which is not compensated. If iron to not keep in the certain position during action on him a magnetic field, this iron will rotate. 2 Interaction of Magnets If two magnets are in a magnetic field each of other, then the ovalmats of the magnetic field of each magnet gets on the rotating sphere of protons of another magnet. As a result of it, the protons which have a identical direction of orbital motion and rotation, are moving together with the all body under action of magnetic field, if this field is directed perpendicularly to axises of rotation of these orbital protons. The magnetic fields move the magnets to one another or repulses one from the other depending on the location of their proton orbits, which are forming the magnetic field. Because at magnetization of iron the orbits of protons of the central atom can be directed in two crossed planes, then the magnetized iron can have one pair or two pairs of magnetic poles. If we bring a magnet closer to a unmagnetized iron, then this iron is attracted to the magnet as this iron is magnetized in magnetic field of the constant magnet. If an electric current passes through a solenoid, this solenoid creates a weak magnetic field, but if inside of this solenoid we place a iron core, then the strength of a magnetic field on the ends of the solenoid considerably increases. It occurs because the metal core becomes magnetized, and the magnetic diclons of its atoms create a magnetic field which has much a greater strength than the electric field created by the electric current. Conclusions 1. The magnetic field of magnetized iron represents the magnetic field formed by orbital protons of central atoms of cells of crystals of iron. Under action of external magnetic field, the orbit of these protons is turned in a 4
5 direction of it magnetic field, and due to that these protons form magnetic fields in identical direction. 2. The Attraction of one constant magnet with another constant magnet and the pushing away of one constant magnet from another constant magnet occurs because of the action of the magnetic field of one constant magnet on the magnetic diclons of another constant magnet. 5
Physical Principle of Formation and Essence of Radio Waves
 Physical Principle of Formation and Essence of Radio Waves Anatoli Bedritsky Abstract. This article opens physical phenomena which occur at the formation of the radio waves, and opens the essence of the
Physical Principle of Formation and Essence of Radio Waves Anatoli Bedritsky Abstract. This article opens physical phenomena which occur at the formation of the radio waves, and opens the essence of the
Physics 112 Homework 5 (solutions) (2004 Fall) Solutions to Homework Questions 5
 Solutions to Homework Questions 5 Chapt19, Problem-2: (a) Find the direction of the force on a proton (a positively charged particle) moving through the magnetic fields in Figure P19.2, as shown. (b) Repeat
Solutions to Homework Questions 5 Chapt19, Problem-2: (a) Find the direction of the force on a proton (a positively charged particle) moving through the magnetic fields in Figure P19.2, as shown. (b) Repeat
Magnetism. d. gives the direction of the force on a charge moving in a magnetic field. b. results in negative charges moving. clockwise.
 Magnetism 1. An electron which moves with a speed of 3.0 10 4 m/s parallel to a uniform magnetic field of 0.40 T experiences a force of what magnitude? (e = 1.6 10 19 C) a. 4.8 10 14 N c. 2.2 10 24 N b.
Magnetism 1. An electron which moves with a speed of 3.0 10 4 m/s parallel to a uniform magnetic field of 0.40 T experiences a force of what magnitude? (e = 1.6 10 19 C) a. 4.8 10 14 N c. 2.2 10 24 N b.
Eðlisfræði 2, vor 2007
 [ Assignment View ] [ Pri Eðlisfræði 2, vor 2007 28. Sources of Magnetic Field Assignment is due at 2:00am on Wednesday, March 7, 2007 Credit for problems submitted late will decrease to 0% after the deadline
[ Assignment View ] [ Pri Eðlisfræði 2, vor 2007 28. Sources of Magnetic Field Assignment is due at 2:00am on Wednesday, March 7, 2007 Credit for problems submitted late will decrease to 0% after the deadline
Section 11.3 Atomic Orbitals Objectives
 Objectives 1. To learn about the shapes of the s, p and d orbitals 2. To review the energy levels and orbitals of the wave mechanical model of the atom 3. To learn about electron spin A. Electron Location
Objectives 1. To learn about the shapes of the s, p and d orbitals 2. To review the energy levels and orbitals of the wave mechanical model of the atom 3. To learn about electron spin A. Electron Location
Force on Moving Charges in a Magnetic Field
 [ Assignment View ] [ Eðlisfræði 2, vor 2007 27. Magnetic Field and Magnetic Forces Assignment is due at 2:00am on Wednesday, February 28, 2007 Credit for problems submitted late will decrease to 0% after
[ Assignment View ] [ Eðlisfræði 2, vor 2007 27. Magnetic Field and Magnetic Forces Assignment is due at 2:00am on Wednesday, February 28, 2007 Credit for problems submitted late will decrease to 0% after
Making an Electromagnet Grade 4
 TEACHING LEARNING COLLABORATIVE (TLC) PHYSICAL SCIENCE Making an Electromagnet Grade 4 Created by: Maria Schetter (Terrace Heights Elementary School), Stella Winckler (Lucerne Elementary School), Karen
TEACHING LEARNING COLLABORATIVE (TLC) PHYSICAL SCIENCE Making an Electromagnet Grade 4 Created by: Maria Schetter (Terrace Heights Elementary School), Stella Winckler (Lucerne Elementary School), Karen
Introduction to Nuclear Physics
 Introduction to Nuclear Physics 1. Atomic Structure and the Periodic Table According to the Bohr-Rutherford model of the atom, also called the solar system model, the atom consists of a central nucleus
Introduction to Nuclear Physics 1. Atomic Structure and the Periodic Table According to the Bohr-Rutherford model of the atom, also called the solar system model, the atom consists of a central nucleus
Electron Arrangements
 Section 3.4 Electron Arrangements Objectives Express the arrangement of electrons in atoms using electron configurations and Lewis valence electron dot structures New Vocabulary Heisenberg uncertainty
Section 3.4 Electron Arrangements Objectives Express the arrangement of electrons in atoms using electron configurations and Lewis valence electron dot structures New Vocabulary Heisenberg uncertainty
Magnetism. Magnetism. Magnetic Fields and Magnetic Domains. Magnetic Fields and Magnetic Domains. Creating and Destroying a Magnet
 Magnetism Magnetism Opposite poles attract and likes repel Opposite poles attract and likes repel Like electric force, but magnetic poles always come in pairs (North, South) Like electric force, but magnetic
Magnetism Magnetism Opposite poles attract and likes repel Opposite poles attract and likes repel Like electric force, but magnetic poles always come in pairs (North, South) Like electric force, but magnetic
Part I: Principal Energy Levels and Sublevels
 Part I: Principal Energy Levels and Sublevels As you already know, all atoms are made of subatomic particles, including protons, neutrons, and electrons. Positive protons and neutral neutrons are found
Part I: Principal Energy Levels and Sublevels As you already know, all atoms are made of subatomic particles, including protons, neutrons, and electrons. Positive protons and neutral neutrons are found
The unifying field Theory
 The unifying field Theory M T Keshe 2000-2009, all rights reserved Date of release 28.10.2009 Abstract In this paper the origin of electromagnetic fields or electromagnetism and how they are created within
The unifying field Theory M T Keshe 2000-2009, all rights reserved Date of release 28.10.2009 Abstract In this paper the origin of electromagnetic fields or electromagnetism and how they are created within
1. The diagram below represents magnetic lines of force within a region of space.
 1. The diagram below represents magnetic lines of force within a region of space. 4. In which diagram below is the magnetic flux density at point P greatest? (1) (3) (2) (4) The magnetic field is strongest
1. The diagram below represents magnetic lines of force within a region of space. 4. In which diagram below is the magnetic flux density at point P greatest? (1) (3) (2) (4) The magnetic field is strongest
Chapter 27 Magnetic Field and Magnetic Forces
 Chapter 27 Magnetic Field and Magnetic Forces - Magnetism - Magnetic Field - Magnetic Field Lines and Magnetic Flux - Motion of Charged Particles in a Magnetic Field - Applications of Motion of Charged
Chapter 27 Magnetic Field and Magnetic Forces - Magnetism - Magnetic Field - Magnetic Field Lines and Magnetic Flux - Motion of Charged Particles in a Magnetic Field - Applications of Motion of Charged
Elements in the periodic table are indicated by SYMBOLS. To the left of the symbol we find the atomic mass (A) at the upper corner, and the atomic num
 . ATOMIC STRUCTURE FUNDAMENTALS LEARNING OBJECTIVES To review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. This material is essential
. ATOMIC STRUCTURE FUNDAMENTALS LEARNING OBJECTIVES To review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. This material is essential
List the 3 main types of subatomic particles and indicate the mass and electrical charge of each.
 Basic Chemistry Why do we study chemistry in a biology course? All living organisms are composed of chemicals. To understand life, we must understand the structure, function, and properties of the chemicals
Basic Chemistry Why do we study chemistry in a biology course? All living organisms are composed of chemicals. To understand life, we must understand the structure, function, and properties of the chemicals
STUDY GUIDE: ELECTRICITY AND MAGNETISM
 319 S. Naperville Road Wheaton, IL 60187 www.questionsgalore.net Phone: (630) 580-5735 E-Mail: info@questionsgalore.net Fax: (630) 580-5765 STUDY GUIDE: ELECTRICITY AND MAGNETISM An atom is made of three
319 S. Naperville Road Wheaton, IL 60187 www.questionsgalore.net Phone: (630) 580-5735 E-Mail: info@questionsgalore.net Fax: (630) 580-5765 STUDY GUIDE: ELECTRICITY AND MAGNETISM An atom is made of three
Crystalline solids. A solid crystal consists of different atoms arranged in a periodic structure.
 Crystalline solids A solid crystal consists of different atoms arranged in a periodic structure. Crystals can be formed via various bonding mechanisms: Ionic bonding Covalent bonding Metallic bonding Van
Crystalline solids A solid crystal consists of different atoms arranged in a periodic structure. Crystals can be formed via various bonding mechanisms: Ionic bonding Covalent bonding Metallic bonding Van
Relevant Reading for this Lecture... Pages 83-87.
 LECTURE #06 Chapter 3: X-ray Diffraction and Crystal Structure Determination Learning Objectives To describe crystals in terms of the stacking of planes. How to use a dot product to solve for the angles
LECTURE #06 Chapter 3: X-ray Diffraction and Crystal Structure Determination Learning Objectives To describe crystals in terms of the stacking of planes. How to use a dot product to solve for the angles
Atomic Structure Ron Robertson
 Atomic Structure Ron Robertson r2 n:\files\courses\1110-20\2010 possible slides for web\atomicstructuretrans.doc I. What is Light? Debate in 1600's: Since waves or particles can transfer energy, what is
Atomic Structure Ron Robertson r2 n:\files\courses\1110-20\2010 possible slides for web\atomicstructuretrans.doc I. What is Light? Debate in 1600's: Since waves or particles can transfer energy, what is
Chapter 18: The Structure of the Atom
 Chapter 18: The Structure of the Atom 1. For most elements, an atom has A. no neutrons in the nucleus. B. more protons than electrons. C. less neutrons than electrons. D. just as many electrons as protons.
Chapter 18: The Structure of the Atom 1. For most elements, an atom has A. no neutrons in the nucleus. B. more protons than electrons. C. less neutrons than electrons. D. just as many electrons as protons.
Determination of Molecular Structure by MOLECULAR SPECTROSCOPY
 Determination of Molecular Structure by MOLEULAR SPETROSOPY hemistry 3 B.Z. Shakhashiri Fall 29 Much of what we know about molecular structure has been learned by observing and analyzing how electromagnetic
Determination of Molecular Structure by MOLEULAR SPETROSOPY hemistry 3 B.Z. Shakhashiri Fall 29 Much of what we know about molecular structure has been learned by observing and analyzing how electromagnetic
Chemical misconceptions 115. Ionisation energy. Ionisation energy, structure of the atom, intra-atomic forces.
 Chemical misconceptions 115 Ionisation energy Target level Topics Rationale This is a diagnostic probe designed for post-16 students studying chemistry. Ionisation energy, structure of the atom, intra-atomic
Chemical misconceptions 115 Ionisation energy Target level Topics Rationale This is a diagnostic probe designed for post-16 students studying chemistry. Ionisation energy, structure of the atom, intra-atomic
1. Units of a magnetic field might be: A. C m/s B. C s/m C. C/kg D. kg/c s E. N/C m ans: D
 Chapter 28: MAGNETIC FIELDS 1 Units of a magnetic field might be: A C m/s B C s/m C C/kg D kg/c s E N/C m 2 In the formula F = q v B: A F must be perpendicular to v but not necessarily to B B F must be
Chapter 28: MAGNETIC FIELDS 1 Units of a magnetic field might be: A C m/s B C s/m C C/kg D kg/c s E N/C m 2 In the formula F = q v B: A F must be perpendicular to v but not necessarily to B B F must be
3 Atomic Structure 15
 3 Atomic Structure 15 3.1 Atoms You need to be familiar with the terms in italics The diameter of the nucleus is approximately 10-15 m and an atom 10-10 m. All matter consists of atoms. An atom can be
3 Atomic Structure 15 3.1 Atoms You need to be familiar with the terms in italics The diameter of the nucleus is approximately 10-15 m and an atom 10-10 m. All matter consists of atoms. An atom can be
Atoms and Elements. Outline Atoms Orbitals and Energy Levels Periodic Properties Homework
 Atoms and the Periodic Table The very hot early universe was a plasma with cationic nuclei separated from negatively charged electrons. Plasmas exist today where the energy of the particles is very high,
Atoms and the Periodic Table The very hot early universe was a plasma with cationic nuclei separated from negatively charged electrons. Plasmas exist today where the energy of the particles is very high,
Nuclear Physics. Nuclear Physics comprises the study of:
 Nuclear Physics Nuclear Physics comprises the study of: The general properties of nuclei The particles contained in the nucleus The interaction between these particles Radioactivity and nuclear reactions
Nuclear Physics Nuclear Physics comprises the study of: The general properties of nuclei The particles contained in the nucleus The interaction between these particles Radioactivity and nuclear reactions
NaCl Lattice Science Activities
 NaCl Lattice Science Activities STEM: The Science of Salt Using a Salt Lattice Model Teacher Notes Science Activities A Guided-Inquiry Approach Using the 3D Molecular Designs NaCl Lattice Model Classroom
NaCl Lattice Science Activities STEM: The Science of Salt Using a Salt Lattice Model Teacher Notes Science Activities A Guided-Inquiry Approach Using the 3D Molecular Designs NaCl Lattice Model Classroom
Conceptual: 1, 3, 5, 6, 8, 16, 18, 19. Problems: 4, 6, 8, 11, 16, 20, 23, 27, 34, 41, 45, 56, 60, 65. Conceptual Questions
 Conceptual: 1, 3, 5, 6, 8, 16, 18, 19 Problems: 4, 6, 8, 11, 16, 20, 23, 27, 34, 41, 45, 56, 60, 65 Conceptual Questions 1. The magnetic field cannot be described as the magnetic force per unit charge
Conceptual: 1, 3, 5, 6, 8, 16, 18, 19 Problems: 4, 6, 8, 11, 16, 20, 23, 27, 34, 41, 45, 56, 60, 65 Conceptual Questions 1. The magnetic field cannot be described as the magnetic force per unit charge
The purposes of this experiment are to test Faraday's Law qualitatively and to test Lenz's Law.
 260 17-1 I. THEORY EXPERIMENT 17 QUALITATIVE STUDY OF INDUCED EMF Along the extended central axis of a bar magnet, the magnetic field vector B r, on the side nearer the North pole, points away from this
260 17-1 I. THEORY EXPERIMENT 17 QUALITATIVE STUDY OF INDUCED EMF Along the extended central axis of a bar magnet, the magnetic field vector B r, on the side nearer the North pole, points away from this
............... [2] At the time of purchase of a Strontium-90 source, the activity is 3.7 10 6 Bq.
![............... [2] At the time of purchase of a Strontium-90 source, the activity is 3.7 10 6 Bq. ............... [2] At the time of purchase of a Strontium-90 source, the activity is 3.7 10 6 Bq.](/thumbs/40/21837699.jpg) 1 Strontium-90 decays with the emission of a β-particle to form Yttrium-90. The reaction is represented by the equation 90 38 The decay constant is 0.025 year 1. 90 39 0 1 Sr Y + e + 0.55 MeV. (a) Suggest,
1 Strontium-90 decays with the emission of a β-particle to form Yttrium-90. The reaction is represented by the equation 90 38 The decay constant is 0.025 year 1. 90 39 0 1 Sr Y + e + 0.55 MeV. (a) Suggest,
Chapter 2 The Chemical Context of Life
 Chapter 2 The Chemical Context of Life Multiple-Choice Questions 1) About 25 of the 92 natural elements are known to be essential to life. Which four of these 25 elements make up approximately 96% of living
Chapter 2 The Chemical Context of Life Multiple-Choice Questions 1) About 25 of the 92 natural elements are known to be essential to life. Which four of these 25 elements make up approximately 96% of living
PS-6.2 Explain the factors that determine potential and kinetic energy and the transformation of one to the other.
 PS-6.1 Explain how the law of conservation of energy applies to the transformation of various forms of energy (including mechanical energy, electrical energy, chemical energy, light energy, sound energy,
PS-6.1 Explain how the law of conservation of energy applies to the transformation of various forms of energy (including mechanical energy, electrical energy, chemical energy, light energy, sound energy,
Review Questions PHYS 2426 Exam 2
 Review Questions PHYS 2426 Exam 2 1. If 4.7 x 10 16 electrons pass a particular point in a wire every second, what is the current in the wire? A) 4.7 ma B) 7.5 A C) 2.9 A D) 7.5 ma E) 0.29 A Ans: D 2.
Review Questions PHYS 2426 Exam 2 1. If 4.7 x 10 16 electrons pass a particular point in a wire every second, what is the current in the wire? A) 4.7 ma B) 7.5 A C) 2.9 A D) 7.5 ma E) 0.29 A Ans: D 2.
PROTONS AND ELECTRONS
 reflect Imagine that you have a bowl of oranges, bananas, pineapples, berries, pears, and watermelon. How do you identify each piece of fruit? Most likely, you are familiar with the characteristics of
reflect Imagine that you have a bowl of oranges, bananas, pineapples, berries, pears, and watermelon. How do you identify each piece of fruit? Most likely, you are familiar with the characteristics of
Basic Nuclear Concepts
 Section 7: In this section, we present a basic description of atomic nuclei, the stored energy contained within them, their occurrence and stability Basic Nuclear Concepts EARLY DISCOVERIES [see also Section
Section 7: In this section, we present a basic description of atomic nuclei, the stored energy contained within them, their occurrence and stability Basic Nuclear Concepts EARLY DISCOVERIES [see also Section
Name Class Date. What is ionic bonding? What happens to atoms that gain or lose electrons? What kinds of solids are formed from ionic bonds?
 CHAPTER 1 2 Ionic Bonds SECTION Chemical Bonding BEFORE YOU READ After you read this section, you should be able to answer these questions: What is ionic bonding? What happens to atoms that gain or lose
CHAPTER 1 2 Ionic Bonds SECTION Chemical Bonding BEFORE YOU READ After you read this section, you should be able to answer these questions: What is ionic bonding? What happens to atoms that gain or lose
Chapter Outline. How do atoms arrange themselves to form solids?
 Chapter Outline How do atoms arrange themselves to form solids? Fundamental concepts and language Unit cells Crystal structures Simple cubic Face-centered cubic Body-centered cubic Hexagonal close-packed
Chapter Outline How do atoms arrange themselves to form solids? Fundamental concepts and language Unit cells Crystal structures Simple cubic Face-centered cubic Body-centered cubic Hexagonal close-packed
Chapter 2: Forms of Energy
 Chapter 2: Forms of Energy Goals of Period 2 Section 2.1: To describe the forms of energy Section 2.2: To illustrate conversions from one form of energy to another Section 2.3 To describe energy storage
Chapter 2: Forms of Energy Goals of Period 2 Section 2.1: To describe the forms of energy Section 2.2: To illustrate conversions from one form of energy to another Section 2.3 To describe energy storage
ANSWER KEY. Energy Levels, Electrons and IONIC Bonding It s all about the Give and Take!
 ANSWER KEY Energy Levels, Electrons and IONIC Bonding It s all about the Give and Take! From American Chemical Society Middle School Chemistry Unit: Chapter 4 Content Statements: Distinguish the difference
ANSWER KEY Energy Levels, Electrons and IONIC Bonding It s all about the Give and Take! From American Chemical Society Middle School Chemistry Unit: Chapter 4 Content Statements: Distinguish the difference
18.2 Comparing Atoms. Atomic number. Chapter 18
 As you know, some substances are made up of only one kind of atom and these substances are called elements. You already know something about a number of elements you ve heard of hydrogen, helium, silver,
As you know, some substances are made up of only one kind of atom and these substances are called elements. You already know something about a number of elements you ve heard of hydrogen, helium, silver,
Atomic Structure: Chapter Problems
 Atomic Structure: Chapter Problems Bohr Model Class Work 1. Describe the nuclear model of the atom. 2. Explain the problems with the nuclear model of the atom. 3. According to Niels Bohr, what does n stand
Atomic Structure: Chapter Problems Bohr Model Class Work 1. Describe the nuclear model of the atom. 2. Explain the problems with the nuclear model of the atom. 3. According to Niels Bohr, what does n stand
Physics 121 Sample Common Exam 3 NOTE: ANSWERS ARE ON PAGE 6. Instructions: 1. In the formula F = qvxb:
 Physics 121 Sample Common Exam 3 NOTE: ANSWERS ARE ON PAGE 6 Signature Name (Print): 4 Digit ID: Section: Instructions: Answer all questions 24 multiple choice questions. You may need to do some calculation.
Physics 121 Sample Common Exam 3 NOTE: ANSWERS ARE ON PAGE 6 Signature Name (Print): 4 Digit ID: Section: Instructions: Answer all questions 24 multiple choice questions. You may need to do some calculation.
Introduction to Electricity & Magnetism. Dr Lisa Jardine-Wright Cavendish Laboratory
 Introduction to Electricity & Magnetism Dr Lisa Jardine-Wright Cavendish Laboratory Examples of uses of electricity Christmas lights Cars Electronic devices Human body Electricity? Electricity is the presence
Introduction to Electricity & Magnetism Dr Lisa Jardine-Wright Cavendish Laboratory Examples of uses of electricity Christmas lights Cars Electronic devices Human body Electricity? Electricity is the presence
Molecular Geometry and Chemical Bonding Theory
 Chapter 10 Molecular Geometry and Chemical Bonding Theory Concept Check 10.1 An atom in a molecule is surrounded by four pairs of electrons, one lone pair and three bonding pairs. Describe how the four
Chapter 10 Molecular Geometry and Chemical Bonding Theory Concept Check 10.1 An atom in a molecule is surrounded by four pairs of electrons, one lone pair and three bonding pairs. Describe how the four
Copyrighted by Gabriel Tang B.Ed., B.Sc.
 Chapter 8: The Periodic Table 8.1: Development of the Periodic Table Johann Dobereiner: - first to discover a pattern of a group of elements like Cl, Br, and I (called triads). John Newland: - suggested
Chapter 8: The Periodic Table 8.1: Development of the Periodic Table Johann Dobereiner: - first to discover a pattern of a group of elements like Cl, Br, and I (called triads). John Newland: - suggested
As you learned in the previous activity, energy is either potential energy or kinetic energy. Each can take many forms.
 Topic 6: Forms of Potential Energy As you learned in the previous activity, energy is either potential energy or kinetic energy. Each can take many forms. Forms of potential energy include Stored Mechanical
Topic 6: Forms of Potential Energy As you learned in the previous activity, energy is either potential energy or kinetic energy. Each can take many forms. Forms of potential energy include Stored Mechanical
Material Deformations. Academic Resource Center
 Material Deformations Academic Resource Center Agenda Origin of deformations Deformations & dislocations Dislocation motion Slip systems Stresses involved with deformation Deformation by twinning Origin
Material Deformations Academic Resource Center Agenda Origin of deformations Deformations & dislocations Dislocation motion Slip systems Stresses involved with deformation Deformation by twinning Origin
ElectroMagnetic Induction. AP Physics B
 ElectroMagnetic Induction AP Physics B What is E/M Induction? Electromagnetic Induction is the process of using magnetic fields to produce voltage, and in a complete circuit, a current. Michael Faraday
ElectroMagnetic Induction AP Physics B What is E/M Induction? Electromagnetic Induction is the process of using magnetic fields to produce voltage, and in a complete circuit, a current. Michael Faraday
LCAO-MO Correlation Diagrams
 LCAO-MO Correlation Diagrams (Linear Combination of Atomic Orbitals to yield Molecular Orbitals) For (Second Row) Homonuclear Diatomic Molecules (X 2 ) - the following LCAO-MO s are generated: LCAO MO
LCAO-MO Correlation Diagrams (Linear Combination of Atomic Orbitals to yield Molecular Orbitals) For (Second Row) Homonuclear Diatomic Molecules (X 2 ) - the following LCAO-MO s are generated: LCAO MO
Bohr s Model of the Atom
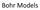 Bohr Models Bohr s Model of the Atom Focuses on electrons and their arrangement. Bohr stated that electrons move with constant speed in fixed orbits around the nucleus, like planets around a sun. Bohr
Bohr Models Bohr s Model of the Atom Focuses on electrons and their arrangement. Bohr stated that electrons move with constant speed in fixed orbits around the nucleus, like planets around a sun. Bohr
Precession of spin and Precession of a top
 6. Classical Precession of the Angular Momentum Vector A classical bar magnet (Figure 11) may lie motionless at a certain orientation in a magnetic field. However, if the bar magnet possesses angular momentum,
6. Classical Precession of the Angular Momentum Vector A classical bar magnet (Figure 11) may lie motionless at a certain orientation in a magnetic field. However, if the bar magnet possesses angular momentum,
AP CHEMISTRY 2009 SCORING GUIDELINES
 AP CHEMISTRY 2009 SCORING GUIDELINES Question 6 (8 points) Answer the following questions related to sulfur and one of its compounds. (a) Consider the two chemical species S and S 2. (i) Write the electron
AP CHEMISTRY 2009 SCORING GUIDELINES Question 6 (8 points) Answer the following questions related to sulfur and one of its compounds. (a) Consider the two chemical species S and S 2. (i) Write the electron
Department of Physics and Geology The Elements and the Periodic Table
 Department of Physics and Geology The Elements and the Periodic Table Physical Science 1422 Equipment Needed Qty Periodic Table 1 Part 1: Background In 1869 a Russian chemistry professor named Dmitri Mendeleev
Department of Physics and Geology The Elements and the Periodic Table Physical Science 1422 Equipment Needed Qty Periodic Table 1 Part 1: Background In 1869 a Russian chemistry professor named Dmitri Mendeleev
2, 8, 20, 28, 50, 82, 126.
 Chapter 5 Nuclear Shell Model 5.1 Magic Numbers The binding energies predicted by the Liquid Drop Model underestimate the actual binding energies of magic nuclei for which either the number of neutrons
Chapter 5 Nuclear Shell Model 5.1 Magic Numbers The binding energies predicted by the Liquid Drop Model underestimate the actual binding energies of magic nuclei for which either the number of neutrons
Background A nucleus with an odd atomic number or an odd mass number has a nuclear spin that can be observed by NMR spectrometers.
 NMR Spectroscopy I Reading: Wade chapter, sections -- -7 Study Problems: -, -7 Key oncepts and Skills: Given an structure, determine which protons are equivalent and which are nonequivalent, predict the
NMR Spectroscopy I Reading: Wade chapter, sections -- -7 Study Problems: -, -7 Key oncepts and Skills: Given an structure, determine which protons are equivalent and which are nonequivalent, predict the
Four Different Kinds of Magnetism
 Four Different Kinds of Magnetism 1.) Diamagnetism A phenomenon in some materials in which the susceptibility is negative, i.e. the magnetization opposed the magnetizing force. It arises from the precession
Four Different Kinds of Magnetism 1.) Diamagnetism A phenomenon in some materials in which the susceptibility is negative, i.e. the magnetization opposed the magnetizing force. It arises from the precession
12.524 2003 Lec 17: Dislocation Geometry and Fabric Production 1. Crystal Geometry
 12.524 2003 Lec 17: Dislocation Geometry and Fabric Production 1. Bibliography: Crystal Geometry Assigned Reading: [Poirier, 1985]Chapter 2, 4. General References: [Kelly and Groves, 1970] Chapter 1. [Hirth
12.524 2003 Lec 17: Dislocation Geometry and Fabric Production 1. Bibliography: Crystal Geometry Assigned Reading: [Poirier, 1985]Chapter 2, 4. General References: [Kelly and Groves, 1970] Chapter 1. [Hirth
19.1 Bonding and Molecules
 Most of the matter around you and inside of you is in the form of compounds. For example, your body is about 80 percent water. You learned in the last unit that water, H 2 O, is made up of hydrogen and
Most of the matter around you and inside of you is in the form of compounds. For example, your body is about 80 percent water. You learned in the last unit that water, H 2 O, is made up of hydrogen and
LECTURE SUMMARY September 30th 2009
 LECTURE SUMMARY September 30 th 2009 Key Lecture Topics Crystal Structures in Relation to Slip Systems Resolved Shear Stress Using a Stereographic Projection to Determine the Active Slip System Slip Planes
LECTURE SUMMARY September 30 th 2009 Key Lecture Topics Crystal Structures in Relation to Slip Systems Resolved Shear Stress Using a Stereographic Projection to Determine the Active Slip System Slip Planes
Force on a square loop of current in a uniform B-field.
 Force on a square loop of current in a uniform B-field. F top = 0 θ = 0; sinθ = 0; so F B = 0 F bottom = 0 F left = I a B (out of page) F right = I a B (into page) Assume loop is on a frictionless axis
Force on a square loop of current in a uniform B-field. F top = 0 θ = 0; sinθ = 0; so F B = 0 F bottom = 0 F left = I a B (out of page) F right = I a B (into page) Assume loop is on a frictionless axis
CHAPTER 5: MAGNETIC PROPERTIES
 CHAPTER 5: MAGNETIC PROPERTIES and Magnetic Materials ISSUES TO ADDRESS... Why do we study magnetic properties? What is magnetism? How do we measure magnetic properties? What are the atomic reasons for
CHAPTER 5: MAGNETIC PROPERTIES and Magnetic Materials ISSUES TO ADDRESS... Why do we study magnetic properties? What is magnetism? How do we measure magnetic properties? What are the atomic reasons for
SUPERCONDUCTIVITY. PH 318- Introduction to superconductors 1
 SUPERCONDUCTIVITY property of complete disappearance of electrical resistance in solids when they are cooled below a characteristic temperature. This temperature is called transition temperature or critical
SUPERCONDUCTIVITY property of complete disappearance of electrical resistance in solids when they are cooled below a characteristic temperature. This temperature is called transition temperature or critical
Ch. 9 - Electron Organization. The Bohr Model [9.4] Orbitals [9.5, 9.6] Counting Electrons, configurations [9.7]
![Ch. 9 - Electron Organization. The Bohr Model [9.4] Orbitals [9.5, 9.6] Counting Electrons, configurations [9.7] Ch. 9 - Electron Organization. The Bohr Model [9.4] Orbitals [9.5, 9.6] Counting Electrons, configurations [9.7]](/thumbs/27/11797291.jpg) Ch. 9 - Electron Organization The Bohr Model [9.4] Orbitals [9.5, 9.6] Counting Electrons, configurations [9.7] Predicting ion charges from electron configurations. CHEM 100 F07 1 Organization of Electrons
Ch. 9 - Electron Organization The Bohr Model [9.4] Orbitals [9.5, 9.6] Counting Electrons, configurations [9.7] Predicting ion charges from electron configurations. CHEM 100 F07 1 Organization of Electrons
Magnetic Fields. I. Magnetic Field and Magnetic Field Lines
 Magnetic Fields I. Magnetic Field and Magnetic Field Lines A. The concept of the magnetic field can be developed in a manner similar to the way we developed the electric field. The magnitude of the magnetic
Magnetic Fields I. Magnetic Field and Magnetic Field Lines A. The concept of the magnetic field can be developed in a manner similar to the way we developed the electric field. The magnitude of the magnetic
Chapter 29: Magnetic Fields
 Chapter 29: Magnetic Fields Magnetism has been known as early as 800C when people realized that certain stones could be used to attract bits of iron. Experiments using magnets hae shown the following:
Chapter 29: Magnetic Fields Magnetism has been known as early as 800C when people realized that certain stones could be used to attract bits of iron. Experiments using magnets hae shown the following:
7.4. Using the Bohr Theory KNOW? Using the Bohr Theory to Describe Atoms and Ions
 7.4 Using the Bohr Theory LEARNING TIP Models such as Figures 1 to 4, on pages 218 and 219, help you visualize scientific explanations. As you examine Figures 1 to 4, look back and forth between the diagrams
7.4 Using the Bohr Theory LEARNING TIP Models such as Figures 1 to 4, on pages 218 and 219, help you visualize scientific explanations. As you examine Figures 1 to 4, look back and forth between the diagrams
CHAPTER 7 DISLOCATIONS AND STRENGTHENING MECHANISMS PROBLEM SOLUTIONS
 7-1 CHAPTER 7 DISLOCATIONS AND STRENGTHENING MECHANISMS PROBLEM SOLUTIONS Basic Concepts of Dislocations Characteristics of Dislocations 7.1 The dislocation density is just the total dislocation length
7-1 CHAPTER 7 DISLOCATIONS AND STRENGTHENING MECHANISMS PROBLEM SOLUTIONS Basic Concepts of Dislocations Characteristics of Dislocations 7.1 The dislocation density is just the total dislocation length
Chapter 21. Magnetic Forces and Magnetic Fields
 Chapter 21 Magnetic Forces and Magnetic Fields 21.1 Magnetic Fields The needle of a compass is permanent magnet that has a north magnetic pole (N) at one end and a south magnetic pole (S) at the other.
Chapter 21 Magnetic Forces and Magnetic Fields 21.1 Magnetic Fields The needle of a compass is permanent magnet that has a north magnetic pole (N) at one end and a south magnetic pole (S) at the other.
Fundamentals of grain boundaries and grain boundary migration
 1. Fundamentals of grain boundaries and grain boundary migration 1.1. Introduction The properties of crystalline metallic materials are determined by their deviation from a perfect crystal lattice, which
1. Fundamentals of grain boundaries and grain boundary migration 1.1. Introduction The properties of crystalline metallic materials are determined by their deviation from a perfect crystal lattice, which
Interaction at a Distance
 Interaction at a Distance Lesson Overview: Students come in contact with and use magnets every day. They often don t consider that there are different types of magnets and that they are made for different
Interaction at a Distance Lesson Overview: Students come in contact with and use magnets every day. They often don t consider that there are different types of magnets and that they are made for different
Chapter 3 Student Reading
 Chapter 3 Student Reading If you hold a solid piece of lead or iron in your hand, it feels heavy for its size. If you hold the same size piece of balsa wood or plastic, it feels light for its size. The
Chapter 3 Student Reading If you hold a solid piece of lead or iron in your hand, it feels heavy for its size. If you hold the same size piece of balsa wood or plastic, it feels light for its size. The
Chapter 1 Structure and Bonding. Modified by Dr. Daniela Radu
 John E. McMurry www.cengage.com/chemistry/mcmurry Chapter 1 Structure and Bonding Modified by Dr. Daniela Radu What is Organic Chemistry? Living things are made of organic chemicals Proteins that make
John E. McMurry www.cengage.com/chemistry/mcmurry Chapter 1 Structure and Bonding Modified by Dr. Daniela Radu What is Organic Chemistry? Living things are made of organic chemicals Proteins that make
Atomic structure. Resources and methods for learning about these subjects (list a few here, in preparation for your research):
 Atomic structure This worksheet and all related files are licensed under the Creative Commons Attribution License, version 1.0. To view a copy of this license, visit http://creativecommons.org/licenses/by/1.0/,
Atomic structure This worksheet and all related files are licensed under the Creative Commons Attribution License, version 1.0. To view a copy of this license, visit http://creativecommons.org/licenses/by/1.0/,
Practice TEST 2. Explain your reasoning
 Practice TEST 2 1. Imagine taking an elevator ride from the1 st floor to the 10 th floor of a building. While moving between the 1 st and 2 nd floors the elevator speeds up, but then moves at a constant
Practice TEST 2 1. Imagine taking an elevator ride from the1 st floor to the 10 th floor of a building. While moving between the 1 st and 2 nd floors the elevator speeds up, but then moves at a constant
ELECTRICAL FUNDAMENTALS
 General Electricity is a form of energy called electrical energy. It is sometimes called an "unseen" force because the energy itself cannot be seen, heard, touched, or smelled. However, the effects of
General Electricity is a form of energy called electrical energy. It is sometimes called an "unseen" force because the energy itself cannot be seen, heard, touched, or smelled. However, the effects of
Objectives. Capacitors 262 CHAPTER 5 ENERGY
 Objectives Describe a capacitor. Explain how a capacitor stores energy. Define capacitance. Calculate the electrical energy stored in a capacitor. Describe an inductor. Explain how an inductor stores energy.
Objectives Describe a capacitor. Explain how a capacitor stores energy. Define capacitance. Calculate the electrical energy stored in a capacitor. Describe an inductor. Explain how an inductor stores energy.
Electricity. Atoms. Protons, Neutrons, and Electrons. Electricity is Moving Electrons. Atom
 Electricity is a mysterious force. We can t see it like we see the sun. We can t hold it like we hold coal. We know when it is working, but it is hard to know exactly what it is. Before we can understand
Electricity is a mysterious force. We can t see it like we see the sun. We can t hold it like we hold coal. We know when it is working, but it is hard to know exactly what it is. Before we can understand
KINDERGARTEN PHYSICS
 KINDERGARTEN PHYSICS 3 WEEKS LESSON PLANS AND ACTIVITIES APPLIED SCIENCE OVERVIEW OF KINDERGARTEN SCIENCE AND MATH WEEK 1. PRE: Describing and comparing nests, birds, and eggs. LAB: Describing different
KINDERGARTEN PHYSICS 3 WEEKS LESSON PLANS AND ACTIVITIES APPLIED SCIENCE OVERVIEW OF KINDERGARTEN SCIENCE AND MATH WEEK 1. PRE: Describing and comparing nests, birds, and eggs. LAB: Describing different
MEASURING INSTRUMENTS. By: Nafees Ahmed, Asstt, Prof, EE Deptt, DIT, Dehradun
 MEASURING INSTRUMENTS By: Nafees Ahmed, Asstt, Prof, EE Deptt, DIT, Dehradun MEASURING INSTRUMENTS The device used for comparing the unknown quantity with the unit of measurement or standard quantity is
MEASURING INSTRUMENTS By: Nafees Ahmed, Asstt, Prof, EE Deptt, DIT, Dehradun MEASURING INSTRUMENTS The device used for comparing the unknown quantity with the unit of measurement or standard quantity is
This topic explores the key concepts of magnetism as they relate to: the phenomenon of magnetism magnetic forces and fields a theory of magnetism.
 Magnetism Introduction This topic explores the key concepts of magnetism as they relate to: the phenomenon of magnetism magnetic forces and fields a theory of magnetism. Key concepts of magnetism The activities
Magnetism Introduction This topic explores the key concepts of magnetism as they relate to: the phenomenon of magnetism magnetic forces and fields a theory of magnetism. Key concepts of magnetism The activities
A Practical Guide to Free Energy Devices
 A Practical Guide to Free Energy Devices Electrolysis Patents No 14: Last updated: 28th January 2006 Author: Patrick J. Kelly Please note that this is a re-worded excerpt from this patent. If the content
A Practical Guide to Free Energy Devices Electrolysis Patents No 14: Last updated: 28th January 2006 Author: Patrick J. Kelly Please note that this is a re-worded excerpt from this patent. If the content
Mechanics lecture 7 Moment of a force, torque, equilibrium of a body
 G.1 EE1.el3 (EEE1023): Electronics III Mechanics lecture 7 Moment of a force, torque, equilibrium of a body Dr Philip Jackson http://www.ee.surrey.ac.uk/teaching/courses/ee1.el3/ G.2 Moments, torque and
G.1 EE1.el3 (EEE1023): Electronics III Mechanics lecture 7 Moment of a force, torque, equilibrium of a body Dr Philip Jackson http://www.ee.surrey.ac.uk/teaching/courses/ee1.el3/ G.2 Moments, torque and
ATOMS A T O M S, I S O T O P E S, A N D I O N S. The Academic Support Center @ Daytona State College (Science 120, Page 1 of 39)
 ATOMS A T O M S, I S O T O P E S, A N D I O N S The Academic Support Center @ Daytona State College (Science 120, Page 1 of 39) THE ATOM All elements listed on the periodic table are made up of atoms.
ATOMS A T O M S, I S O T O P E S, A N D I O N S The Academic Support Center @ Daytona State College (Science 120, Page 1 of 39) THE ATOM All elements listed on the periodic table are made up of atoms.
An Atom Apart by Leslie Cargile
 Have you ever walked through a cloud of gnats on a hot summer, only to have them follow you? No matter how you swat at them, or even if you run, they won t leave you alone. If so, then you have something
Have you ever walked through a cloud of gnats on a hot summer, only to have them follow you? No matter how you swat at them, or even if you run, they won t leave you alone. If so, then you have something
13.1 The Nature of Gases. What is Kinetic Theory? Kinetic Theory and a Model for Gases. Chapter 13: States of Matter. Principles of Kinetic Theory
 Chapter 13: States of Matter The Nature of Gases The Nature of Gases kinetic molecular theory (KMT), gas pressure (pascal, atmosphere, mm Hg), kinetic energy The Nature of Liquids vaporization, evaporation,
Chapter 13: States of Matter The Nature of Gases The Nature of Gases kinetic molecular theory (KMT), gas pressure (pascal, atmosphere, mm Hg), kinetic energy The Nature of Liquids vaporization, evaporation,
Chapter 10 Liquids & Solids
 1 Chapter 10 Liquids & Solids * 10.1 Polar Covalent Bonds & Dipole Moments - van der Waals constant for water (a = 5.28 L 2 atm/mol 2 ) vs O 2 (a = 1.36 L 2 atm/mol 2 ) -- water is polar (draw diagram)
1 Chapter 10 Liquids & Solids * 10.1 Polar Covalent Bonds & Dipole Moments - van der Waals constant for water (a = 5.28 L 2 atm/mol 2 ) vs O 2 (a = 1.36 L 2 atm/mol 2 ) -- water is polar (draw diagram)
Matter, Materials, Crystal Structure and Bonding. Chris J. Pickard
 Matter, Materials, Crystal Structure and Bonding Chris J. Pickard Why should a theorist care? Where the atoms are determines what they do Where the atoms can be determines what we can do Overview of Structure
Matter, Materials, Crystal Structure and Bonding Chris J. Pickard Why should a theorist care? Where the atoms are determines what they do Where the atoms can be determines what we can do Overview of Structure
A METHOD OF CALIBRATING HELMHOLTZ COILS FOR THE MEASUREMENT OF PERMANENT MAGNETS
 A METHOD OF CALIBRATING HELMHOLTZ COILS FOR THE MEASUREMENT OF PERMANENT MAGNETS Joseph J. Stupak Jr, Oersted Technology Tualatin, Oregon (reprinted from IMCSD 24th Annual Proceedings 1995) ABSTRACT The
A METHOD OF CALIBRATING HELMHOLTZ COILS FOR THE MEASUREMENT OF PERMANENT MAGNETS Joseph J. Stupak Jr, Oersted Technology Tualatin, Oregon (reprinted from IMCSD 24th Annual Proceedings 1995) ABSTRACT The
physics 112N magnetic fields and forces
 physics 112N magnetic fields and forces bar magnet & iron filings physics 112N 2 bar magnets physics 112N 3 the Earth s magnetic field physics 112N 4 electro -magnetism! is there a connection between electricity
physics 112N magnetic fields and forces bar magnet & iron filings physics 112N 2 bar magnets physics 112N 3 the Earth s magnetic field physics 112N 4 electro -magnetism! is there a connection between electricity
Where Is My Lone Pair?
 Where Is My Lone Pair? Goal: In this tutorial we'll learn how to determine which orbital contains a lone pair. This is important for resonance, conjugation, and aromaticity. To master this subject you'll
Where Is My Lone Pair? Goal: In this tutorial we'll learn how to determine which orbital contains a lone pair. This is important for resonance, conjugation, and aromaticity. To master this subject you'll
States of Matter CHAPTER 10 REVIEW SECTION 1. Name Date Class. Answer the following questions in the space provided.
 CHAPTER 10 REVIEW States of Matter SECTION 1 SHORT ANSWER Answer the following questions in the space provided. 1. Identify whether the descriptions below describe an ideal gas or a real gas. ideal gas
CHAPTER 10 REVIEW States of Matter SECTION 1 SHORT ANSWER Answer the following questions in the space provided. 1. Identify whether the descriptions below describe an ideal gas or a real gas. ideal gas
electron configuration
 electron configuration Electron Configuration Knowing the arrangement of electrons in atoms will better help you understand chemical reactivity and predict an atom s reaction behavior. We know when n=1
electron configuration Electron Configuration Knowing the arrangement of electrons in atoms will better help you understand chemical reactivity and predict an atom s reaction behavior. We know when n=1
Name Partners Date. Energy Diagrams I
 Name Partners Date Visual Quantum Mechanics The Next Generation Energy Diagrams I Goal Changes in energy are a good way to describe an object s motion. Here you will construct energy diagrams for a toy
Name Partners Date Visual Quantum Mechanics The Next Generation Energy Diagrams I Goal Changes in energy are a good way to describe an object s motion. Here you will construct energy diagrams for a toy
Atoms, Ions and Molecules The Building Blocks of Matter
 Atoms, Ions and Molecules The Building Blocks of Matter Chapter 2 1 Chapter Outline 2.1 The Rutherford Model of Atomic Structure 2.2 Nuclides and Their Symbols 2.3 Navigating the Periodic Table 2.4 The
Atoms, Ions and Molecules The Building Blocks of Matter Chapter 2 1 Chapter Outline 2.1 The Rutherford Model of Atomic Structure 2.2 Nuclides and Their Symbols 2.3 Navigating the Periodic Table 2.4 The
NUCLEAR MAGNETIC RESONANCE. Advanced Laboratory, Physics 407, University of Wisconsin Madison, Wisconsin 53706
 (revised 4/21/03) NUCLEAR MAGNETIC RESONANCE Advanced Laboratory, Physics 407, University of Wisconsin Madison, Wisconsin 53706 Abstract This experiment studies the Nuclear Magnetic Resonance of protons
(revised 4/21/03) NUCLEAR MAGNETIC RESONANCE Advanced Laboratory, Physics 407, University of Wisconsin Madison, Wisconsin 53706 Abstract This experiment studies the Nuclear Magnetic Resonance of protons
Chemistry - Elements Electron Configurations The Periodic Table. Ron Robertson
 Chemistry - Elements Electron Configurations The Periodic Table Ron Robertson History of Chemistry Before 16 th Century Alchemy Attempts (scientific or otherwise) to change cheap metals into gold no real
Chemistry - Elements Electron Configurations The Periodic Table Ron Robertson History of Chemistry Before 16 th Century Alchemy Attempts (scientific or otherwise) to change cheap metals into gold no real
Motor Fundamentals. DC Motor
 Motor Fundamentals Before we can examine the function of a drive, we must understand the basic operation of the motor. It is used to convert the electrical energy, supplied by the controller, to mechanical
Motor Fundamentals Before we can examine the function of a drive, we must understand the basic operation of the motor. It is used to convert the electrical energy, supplied by the controller, to mechanical
Centripetal force, rotary motion, angular velocity, apparent force.
 Related Topics Centripetal force, rotary motion, angular velocity, apparent force. Principle and Task A body with variable mass moves on a circular path with ad-justable radius and variable angular velocity.
Related Topics Centripetal force, rotary motion, angular velocity, apparent force. Principle and Task A body with variable mass moves on a circular path with ad-justable radius and variable angular velocity.
UNIT (2) ATOMS AND ELEMENTS
 UNIT (2) ATOMS AND ELEMENTS 2.1 Elements An element is a fundamental substance that cannot be broken down by chemical means into simpler substances. Each element is represented by an abbreviation called
UNIT (2) ATOMS AND ELEMENTS 2.1 Elements An element is a fundamental substance that cannot be broken down by chemical means into simpler substances. Each element is represented by an abbreviation called
