Methods, tools, and pipelines for analysis of Ion PGM Sequencer mirna and gene expression data
|
|
|
- Theodora Knight
- 10 years ago
- Views:
Transcription
1 WHITE PAPER Ion RNA-Seq Methods, tools, and pipelines for analysis of Ion PGM Sequencer mirna and gene expression data Introduction High-resolution measurements of transcriptional activity and organization have been made possible in recent years through massively parallel RNA sequencing (RNA-Seq) technologies. The level of precision enabled by this technology has been demonstrated to be superior to traditional platforms for measuring gene expression in a cell type or tissue [1,2]. Furthermore, with increased sequencing depth, other more specialized analyses such as identification of novel splice variants [3], transcript fusions in cancer [4], and micrornas [5] (mirna) are possible. The depth needed for a given experiment is informed by the biological question at hand and complexity of the transcriptome being measured. RNA-Seq experiments typically produce vast data files that introduce potential bottlenecks when attempting to extract and interpret gene expression information from a given sample. This problem is exacerbated when bioinformatics personnel are few or not available. Much of this can be mitigated through the use of standardized file formats and automated analytical pipelines that are modular and flexible. In this white paper, we describe methods and bioinformatics tools that have been logically connected into automated pipelines used in the analysis of mirna and whole transcriptome (WT) sequencing data generated on the Ion PGM Sequencer. Dataflow for these pipelines begins with raw sequences from the Ion PGM Sequencer and ends with statistical reporting and mapped counts assigned to each gene or mirna. Methods for quality assessment and quantification are discussed along with descriptions of individual mapping programs and standard file formats, which are used to help ensure interoperability between known and tested third-party tools. Count outputs from these pipelines enable downstream applications such as differential gene expression analysis and mirna profiling. Through the use of these types of pipelines, a reproducible and accurate representation of RNA-Seq library content can be achieved. Implementation At a high level, both the WT and mirna pipelines have five fundamental tasks to perform once sequences (reads) are acquired from a dedicated Ion PGM Torrent Server quality/adapter trimming preprocessing 2. Quality control/assessment 3. Mapping to a reference genome and or transcripts 4. Counting mapped reads 5. Generation of global mapping statistics Whole transcriptome RNA-Seq analysis pipeline Attached to each Ion PGM Sequencer is a dedicated Torrent Server running the Torrent Suite software that processes raw signal produced by the PGM Sequencer and calls bases with associated per-base quality values [6]. These data values are written to a standard [7] file, then copied to an analysis server where it serves as the only input required by the WT pipeline. At this point, reads have already been trimmed at their 3 ends of regions of trailing low quality, and adapter (P1) sequences have been clipped by the Torrent Suite software [8]. With larger insert libraries, adapter clipping is not often necessary as the read does not extend far enough given the number of cycles configured for a given run. While the Torrent Suite is very effective for 3 quality trimming and adapter clipping using default parameters, occasionally residual adapter and low-quality bases remain at 3 ends of reads. For this reason, we add one additional 3 end trimming function as an initial step in the WT pipeline (Figure 1). It should be noted that adapter and quality trimming are critical since the proxy for mrna and mirna quantitation is alignment to a known sequence reference and, ideally, each RNA fragment should map to a genomic location from which it was transcribed. Low quality and portions of adapter sequence flanking an RNA insert can lead to mapping errors such as a misalignment, or can prevent the read from mapping completely.
2 Quality Counting/statistics Mapping assessment QC/QA -positional quality profile -K-mer biases -plot results ERCC matches count reads matching ERCCs ERCC counts ERCC analysis -detection level -dose response ERCC stats PGM reads 3 end trimming Alignment align reads to genome, ERCCs, rrna, exon junctions Gene matches count number of reads that map to known exon locations on chromosomes -alignment stats -rrna matches -exon matches -min. QV: 17 -min. length: 35 nt -remove too short reads trimmed SAM file gene counts -ERCC R 2 /scatterplot -RefSeq genes detected -top expressed genes Figure 1. Whole transciptome workflow for quantifying RefSeq genes and ERCC spike-in controls. Final outputs include counts tables and pertinent global statistics. This preprocessing step is accomplished through the use of the FASTX-toolkit [9] available from the Hannon lab (CSHL). This software suite includes a set of tools for processing and evaluating files. Specifically, the fastq_quality_trimmer tool is applied such that sequences below a minimum Phred [10] quality value (QV) 17 scanning from 5 to the 3 end of the read are trimmed. If the read falls below 35 bases after trimming, it is removed from further analysis to ensure higher specificity when aligning to a genome reference. Quality control and assessment After this preprocessing step, trimmed reads in format are used as input to generate plots that may be utilized for future quality control/assessment. A program called FastQC [11] uses a file as input, calculates global statistics based on QVs and nucleotide composition, and subsequently produces several plots that provide a high-level view of the sequence quality and potential biases detectable in a RNA-Seq library. Specific elements in this tool set include plots of positional QV across the length of all reads, QV distribution, positional nucleotide composition, and relative k-mer enrichment over read length. A scenario highlighting how this analysis could prove very useful is when fewer reads are mapping to the reference genome than expected. After reviewing the FastQC report, it is observed that the distribution of QVs is shifted much lower than normal indicating a problem may have occurred at steps upstream from the pipeline. Mapping Running in parallel to the FastQC step, the trimmed reads are aligned to human reference genome using the TMAP mapping program [12]. This mapper is ideal for aligning reads of variable length and includes three algorithms that may be run individually (map1, map2, and map3) or together (mapall). For the purposes of the WT pipeline, we use the mapall parameter with seed lengths of 18 nucleotides and employ the default number of allowable mismatches per seed. We have found that for an entire alignment of a read to a reference, allowing for approximately 10% mismatches will preserve alignability and specificity. Run time for this step is typically just over two hours for a file containing 2 million reads and utilizing 8 threads (-n 8 TMAP parameter) on a multi-core analysis server. The output of TMAP (and most other aligners for RNA-Seq) is a Sequence Alignment Map or SAM [13] formatted file. This file contains pertinent information about the alignment of each read such as chromosomal location, chromosomal strand, number of mismatches, alignment score, and insertions/ deletions that occurred between the read and a reference sequence. A binary and more compact version of a SAM file, or BAM file, may be generated from a SAM file using SamTools [14]. The genome reference contains the chromosomal sequences available from UCSC [15], ribosomal RNAs (rrna), known and putative exon-exon junctions, and the sequences of 92 synthetic RNA spike-in controls (ERCC) [16], which will be discussed later. Ribosomal RNA sequences are included in the reference so that rrna in a library may be quantified for the purposes of evaluating 2
3 log 2 ERCC counts (present calls: 1 count) R 2 = N = log 2 Relative ERCC concentration Figure 2. ERCC spike-in control dose-response plot generated from the whole transcriptome pipeline. A sample ERCC dose-response scatter plot and linear regression statistics from an RNA-Seq library prepared from HeLa cell poly(a)-enriched RNA with Ambion ERCC RNA Spike-In Mix ( ) added and sequenced on an Ion 318 Chip. This plot is automatically generated by the whole transcriptome pipeline. sensitivity and/or the efficiency of rrna depletion or poly(a) selection techniques. RNA-Seq reads that map to span splice junctions in the genome may result in suboptimal partial alignments or not map at all due to the presence of introns. To mitigate this issue, a sequence reference of all possible combinations of known and putative exon-exon junctions was constructed and included in the mapping reference. The number of reads mapping to this reference tends to increase with mean alignment length suggesting that the likelihood of crossing a splice junction increases with read length. Counting After the alignment step, mapped read counts per gene and ERCC are obtained through the use of the htseq-count.py script included the HTSeq [17] Python package. This script will extract chromosomal coordinates of each mapped read in a SAM file and detect overlaps with known RefSeq 3
4 Quality Counting/statistics Mapping assessment QC/QA -positional quality profile -K-mer biases -plot results PGM reads 3 end trimming -min. QV: 17 -min length: 35 nt -remove too short reads Alignment align reads to mirbase hairpins, rrna, & trna Small-RNA matches count number of reads mapping to known mirna hairpins, rrna, & trna trimmed SAM file Small RNA counts -basic alignment stats -mirnas detected -mirna matches -rrna matches -top expressed mirnas -trna matches Figure 3. Small RNA workflow for quantifying mirna transcripts. Final outputs include counts tables and pertinent global statistics. exon features that are annotated in a GTF (gene transfer format) [18] file referred to as a RefGene GTF. This may be downloaded as a table from the UCSC Genome Browser website. For counting read matches to ERCCs, we simply added 92 entries to the RefGene GTF one for each transcript then again run htseq-count.py to count reads mapping to each ERCC. Counts to genes and ERCCs are generated in two distinct steps, each run in parallel and resulting in a table of counts. Analysis of external RNA spike-in controls Dynamic range, sensitivity, and variability may be examined with the use of 92 ERCC external RNA spike-in controls. These transcripts are polyadenylated, unlabeled RNAs which have been certified and tested by the National Institute of Standards and Technology (NIST) as a means to evaluate RNA measurement systems for performance and control sources of variability. These transcripts have been balanced for GC content to closely represent characteristics of endogenous eukaryotic mrnas and lengths range from 250 to 2,000 nucleotides. The ERCC pool of transcripts is configured in known titrations designed to represent a large dynamic range of expression levels. The table of ERCC counts from the HTSeq step along with known relative concentrations are sent to an R [19] script which then generates a linear regression model. From this, a dose-response curve is plotted in log 2 space (Figure 2) and R 2 slope and sample size are extracted. A typical, high-quality sequenced library will have an R 2 above 0.9 and a sample size between 60 and 70. The sample size is defined as the number of ERCC transcripts detected with 1 or more counts. At this point, all major analysis components in the WT pipeline have completed and need to be summarized. A Perl script is used to parse the SAM file and calculate basic mapping statistics such as total number of mapped reads, reads mapping to exons, exon junctions, rrnas, and reads which map to the genome but do not overlap with known feature annotations. In addition, the number RefSeq genes detected at several minimum count thresholds are reported as a measure of sensitivity and library complexity along with the genes with the highest expression are extracted. These summary statistics are formatted for clarity and written to single report file which resides inside an Ion PGM Sequencer specific directory along with all plots, program output files and counts tables. mirna-seq analysis pipeline There are many workflow similarities between the WT and mirna analysis pipelines, and they share the basic high-level functionality. The following sections describing individual components of the mirna-seq analysis pipeline (Figure 3) will focus on where the two pipelines differ and why. The 3 end trimming based on quality uses a QV threshold of 17 like the WT pipeline. However, the mean length of mature mirnas that comprise a small RNA library is 22 with a range between 18 and 32 nucleotides. Thus, minimum sequence length after trimming is set to 4
5 17 bases. At this minimum length, high mapping specificity is achievable, since the mapping reference is relatively minuscule (178 kb) and fewer locations exist where reads can ambiguously align. If the read does happen to fall below 17 bases after trimming, it is removed from further analysis. The detection and trimming of adapter sequences is more crucial with small RNA libraries. For a typical small RNA run on the Ion PGM Sequencer, reads are usually longer than the mature mirna insert and thus the P1 adapter constitutes the last several bases in a read. As mentioned before, this can complicate and or inhibit mappability of these reads, but is well handled by the Torrent Suite in most cases. The fastq_quality_trimmer tool includes a parameter to set the maximum length of each read. We set this to 35 bases, thereby maximizing the number alignments possible to the small RNA reference and leaving little or no adapter sequence to compromise mapability. Moreover, the aligner used in this pipeline has a local alignment step which will find the best alignment of the read to the reference even if it is not a contiguous match. Mapping Once FastQC has been run on the trimmed file for mirna-seq libraries, reads are mapped using the SHRiMP [20] short read aligner to a small RNA reference containing mirna hairpin sequences, rrnas, trnas, and 3 adapter sequence. SHRiMP was used since it has been tested and optimized for mirna mapping and can be run using a special mirna mode. As with TMAP, each alignment is initialized with a seeding step. Additionally, a recent paper [21] comparing several aligners for the purposes of quantitative mirna expression analysis found that SHRiMP was one of two aligners that showed the highest sensitivity in mapping a published synthetic mirna dataset. While TMAP has shown promising results in this area, extensive testing and optimization has yet to be performed. The primary mapping reference sequences for this pipeline are mirna hairpins. These are precursor mirnas nucleotides in length and are available from the RFAM mirbase [22] database in FASTA format. This repository is frequently updated and includes supporting evidence from RNA-Seq experiments. Precursor sequences are chosen for mapping rather than mature sequences since non-canonical mature mirna sequences, termed isomirs (first described by Morin et al. [23]), may be present in a sample due to variability in biogenesis. Also included in the mapping reference are trnas and rrnas. While mappings to these RNAs are typically less frequent than mirnas, the level of these RNA species can provide insight into library preparation procedures and optimizations; especially in terms of how the RNA sample was size selected. Adapter sequences are included to accommodate the situation where the entire read is only 3 adapter, indicating that there is no RNA insert attached to a bead and the rare event that the sequence was not effectively removed by Torrent Suite preprocessing. Output from SHRiMP can either be in the larger pretty alignment format or can be written directly to a SAM file. The pretty format is reminiscent of default BLAST alignment format [24] and is useful if one intends to view alignments nucleotide by nucleotide. Given the vastness of these files and incompatibility with downstream applications, this format is generally regarded as impractical. Counting Since all mapping is to a reference of RNA transcripts and not a genome, counts to individual transcripts may be calculated directly from the SAM file. This is accomplished by a simple Perl script, which parses the SAM header for transcript identifiers and then scans the remainder of the file to tally counts to each of those RNAs. The resulting counts file contains a column of IDs and counts for each mirna precursor, trna and rrna. As with the WT pipeline, counts and basic mapping statistics are compiled with a Perl summary script that processes the SAM file. In addition, counts of matches to adapter only (no insert) and number of mirbase mirnas detected are reported. Discussion Downstream applications of RNA-Seq count data Counts tables serve as quantitative measure of gene/ mirna expression. Many methods for examining differential expression, normalization, and profiling have emerged as RNA-Seq has increased in popularity and with technological advances of deep-sequencing platforms. Specifically for comparing gene expression, a table of gene counts from two experiments having multiple biological replicates can be compared to estimate differential expression while taking into account of biological variability using the R package DESeq [25]. While DESeq has generally been applied to whole transciptome count data, Cordero et al. [21] reports that DESeq also shows very good sensitivity and specificity when examining mirna-seq count data. Since the SAM (and BAM) formats have become so versatile, many tools are now available for visualization of read coverage across genomes and transcripts that use this format for input. The Integrative Genomics Viewer (IGV) [26] and Tablet [27] are among the most popular. 5
6 Commercial software packages from Partek [28] and CLC bio [29], which combine some of the pipeline steps described above with various types of visualizations and in-depth transcriptome analysis, are also available. Pipeline automation and jobs scheduling Analysis pipelines such as these lend themselves well to additional automation to easily integrate into a high-throughput environment where several Ion PGM Sequencer runs are to be processed in a particular day. A simple wrapper Perl or Python script can easily execute each pipeline step in series. An even more efficient option, if a UNIX-based compute cluster environment is available, would be to submit each step as task in a job scheduler such as the Portable Batch System (PBS) [30]. In this system, steps may be configured to run in parallel, and jobs will run from a queue as computational resources are available. Processing time and hardware requirements For either pipeline, the rate-limiting step is the alignment component. Processing times are approximately one hour and ten minutes for the WT and small RNA pipelines respectively, per million reads. Each pipeline is run such that aligner uses 8 threads allowing for parallel processing on a multi-core CPU. Memory requirements scale with the size of the reference genome to which is being mapped to. For the WT pipeline described here (which includes the human genome, ERCC and rrna transcripts, plus putative and novel exons junction sequences), approximately 16 GB of RAM is required by TMAP while only 1.5 GB is required for SHRiMP. For the description of the hardware configuration used for running these pipelines, please see Appendix A. References 1. Wang Z, Gerstein M, Snyder M. (2009) RNA-Seq: a revolutionary tool for transcriptomics. Nat Rev Genet 10: Mortazavi A, Williams BA, McCue K, et al. (2008) Mapping and quantifying mammalian transcriptomes by RNA-Seq. Nat Methods 5: Sultan M, Schulz MH, Richard H, et al. (2008) A global view of gene activity and alternative splicing by deep sequencing of the human transcriptome. Science 321: Smith TM, Olson NE, Smith D (2010) Making cancer transcriptome sequencing assays practical for the research and clinical scientist. Genome Biology (Supp 1), p Ryu S, Joshi N, McDonnell K, et al. (2011) Discovery of novel human breast cancer micrornas from deep sequencing data by analysis of pri-microrna secondary structures. PLoS One 6:e doi: /journal.pone Torrent Suite technical note: The Per-Base Quality Score System ( 7. format description from Wikipedia: ( 8. Torrent Suite technical note: Filtering and Trimming ( 9. FASTX-Toolkit: ( 10. Phred quality score description from Wikipedia: ( 11. FastQC sequencing quality control program: ( 12. TMAP: The Ion Torrent flow sequence mapping program: ( 13. SAM Format Specification (v1.4-r985): ( 14. Li H, Handsaker B, Wysoker A, et al. (2009) 1000 Genome Project Data Processing Subgroup. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25: UCSC human genome reference: ( 16. Jiang L, Schlesinger F, Davis CA, et al. (2011) Synthetic spike-in standards for RNA-seq experiments. Genome Res 21: HTSeq overview: ( 18. GTF/GFF file format: ( 19. R Development Core Team (2011). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN : ( 20. David M, Dzamba M, Lister D, et al. (2011) SHRiMP2: sensitive yet practical SHort Read Mapping. Bioinformatics 27: Cordero F, Beccuti M, Arigoni M, et al. (2012) Optimizing a massive parallel sequencing workflow for quantitative mirna expression analysis. PLoS One 7:e doi: /journal.pone Kozomara A, Griffiths-Jones S. (2011) mirbase: integrating microrna annotation and deep-sequencing data. Nucleic Acids Res (database issue) 39:D152 D Morin RD, O Connor MD, Griffith M, et al. (2008) Application of massively parallel sequencing to microrna profiling and discovery in human embryonic stem cells. Genome Res 18: BLASTN alignment format: ( 25. Anders S., Huber W. (2010) Differential expression analysis for sequence count data. Genome Biol 11:R Robinson JT, Thorvaldsdóttir H, Winckler W, et al. (2011) Integrative genomics viewer. Nat Biotechnol 29: Milne I, Bayer M, Cardle L, et al. (2010) Tablet--next generation sequence assembly visualization. Bioinformatics 26: Partek Genomics Suite: ( 29. CLC bio: ( 30. Portable Batch System (PBS): ( 6
7 Appendix A Pipeline software versions utilized Torrent Server v2.2.1 FASTX-Toolkit v FastQC v0.9.5 (requires Java Runtime Environment) TMAP v0.1.3 SHRiMP v2.1.0 R v HTSeq v0.5.3p1 Perl v5.8.8 Python v2.6 Pipeline hardware configuration CPU: Intel Xeon E GHz, 4 cores per chip RAM: 64 GB Storage array: 72 hard drives, 1 TB each For Research Use Only. Not intended for any animal or human therapeutic or diagnostic use Life Technologies Corporation. All rights reserved. The trademarks mentioned herein are the property of Life Technologies Corporation or their respective owners. Printed in the USA. CO Headquarters 5791 Van Allen Way Carlsbad, CA USA Phone Toll Free in the USA
Standards, Guidelines and Best Practices for RNA-Seq V1.0 (June 2011) The ENCODE Consortium
 Standards, Guidelines and Best Practices for RNA-Seq V1.0 (June 2011) The ENCODE Consortium I. Introduction: Sequence based assays of transcriptomes (RNA-seq) are in wide use because of their favorable
Standards, Guidelines and Best Practices for RNA-Seq V1.0 (June 2011) The ENCODE Consortium I. Introduction: Sequence based assays of transcriptomes (RNA-seq) are in wide use because of their favorable
FlipFlop: Fast Lasso-based Isoform Prediction as a Flow Problem
 FlipFlop: Fast Lasso-based Isoform Prediction as a Flow Problem Elsa Bernard Laurent Jacob Julien Mairal Jean-Philippe Vert September 24, 2013 Abstract FlipFlop implements a fast method for de novo transcript
FlipFlop: Fast Lasso-based Isoform Prediction as a Flow Problem Elsa Bernard Laurent Jacob Julien Mairal Jean-Philippe Vert September 24, 2013 Abstract FlipFlop implements a fast method for de novo transcript
PreciseTM Whitepaper
 Precise TM Whitepaper Introduction LIMITATIONS OF EXISTING RNA-SEQ METHODS Correctly designed gene expression studies require large numbers of samples, accurate results and low analysis costs. Analysis
Precise TM Whitepaper Introduction LIMITATIONS OF EXISTING RNA-SEQ METHODS Correctly designed gene expression studies require large numbers of samples, accurate results and low analysis costs. Analysis
LifeScope Genomic Analysis Software 2.5
 USER GUIDE LifeScope Genomic Analysis Software 2.5 Graphical User Interface DATA ANALYSIS METHODS AND INTERPRETATION Publication Part Number 4471877 Rev. A Revision Date November 2011 For Research Use
USER GUIDE LifeScope Genomic Analysis Software 2.5 Graphical User Interface DATA ANALYSIS METHODS AND INTERPRETATION Publication Part Number 4471877 Rev. A Revision Date November 2011 For Research Use
Using Illumina BaseSpace Apps to Analyze RNA Sequencing Data
 Using Illumina BaseSpace Apps to Analyze RNA Sequencing Data The Illumina TopHat Alignment and Cufflinks Assembly and Differential Expression apps make RNA data analysis accessible to any user, regardless
Using Illumina BaseSpace Apps to Analyze RNA Sequencing Data The Illumina TopHat Alignment and Cufflinks Assembly and Differential Expression apps make RNA data analysis accessible to any user, regardless
Introduction to NGS data analysis
 Introduction to NGS data analysis Jeroen F. J. Laros Leiden Genome Technology Center Department of Human Genetics Center for Human and Clinical Genetics Sequencing Illumina platforms Characteristics: High
Introduction to NGS data analysis Jeroen F. J. Laros Leiden Genome Technology Center Department of Human Genetics Center for Human and Clinical Genetics Sequencing Illumina platforms Characteristics: High
Shouguo Gao Ph. D Department of Physics and Comprehensive Diabetes Center
 Computational Challenges in Storage, Analysis and Interpretation of Next-Generation Sequencing Data Shouguo Gao Ph. D Department of Physics and Comprehensive Diabetes Center Next Generation Sequencing
Computational Challenges in Storage, Analysis and Interpretation of Next-Generation Sequencing Data Shouguo Gao Ph. D Department of Physics and Comprehensive Diabetes Center Next Generation Sequencing
Analysis of ChIP-seq data in Galaxy
 Analysis of ChIP-seq data in Galaxy November, 2012 Local copy: https://galaxy.wi.mit.edu/ Joint project between BaRC and IT Main site: http://main.g2.bx.psu.edu/ 1 Font Conventions Bold and blue refers
Analysis of ChIP-seq data in Galaxy November, 2012 Local copy: https://galaxy.wi.mit.edu/ Joint project between BaRC and IT Main site: http://main.g2.bx.psu.edu/ 1 Font Conventions Bold and blue refers
BIOL 3200 Spring 2015 DNA Subway and RNA-Seq Data Analysis
 BIOL 3200 Spring 2015 DNA Subway and RNA-Seq Data Analysis By the end of this lab students should be able to: Describe the uses for each line of the DNA subway program (Red/Yellow/Blue/Green) Describe
BIOL 3200 Spring 2015 DNA Subway and RNA-Seq Data Analysis By the end of this lab students should be able to: Describe the uses for each line of the DNA subway program (Red/Yellow/Blue/Green) Describe
Data Analysis & Management of High-throughput Sequencing Data. Quoclinh Nguyen Research Informatics Genomics Core / Medical Research Institute
 Data Analysis & Management of High-throughput Sequencing Data Quoclinh Nguyen Research Informatics Genomics Core / Medical Research Institute Current Issues Current Issues The QSEQ file Number files per
Data Analysis & Management of High-throughput Sequencing Data Quoclinh Nguyen Research Informatics Genomics Core / Medical Research Institute Current Issues Current Issues The QSEQ file Number files per
Introduction to transcriptome analysis using High Throughput Sequencing technologies (HTS)
 Introduction to transcriptome analysis using High Throughput Sequencing technologies (HTS) A typical RNA Seq experiment Library construction Protocol variations Fragmentation methods RNA: nebulization,
Introduction to transcriptome analysis using High Throughput Sequencing technologies (HTS) A typical RNA Seq experiment Library construction Protocol variations Fragmentation methods RNA: nebulization,
Frequently Asked Questions Next Generation Sequencing
 Frequently Asked Questions Next Generation Sequencing Import These Frequently Asked Questions for Next Generation Sequencing are some of the more common questions our customers ask. Questions are divided
Frequently Asked Questions Next Generation Sequencing Import These Frequently Asked Questions for Next Generation Sequencing are some of the more common questions our customers ask. Questions are divided
Challenges associated with analysis and storage of NGS data
 Challenges associated with analysis and storage of NGS data Gabriella Rustici Research and training coordinator Functional Genomics Group [email protected] Next-generation sequencing Next-generation sequencing
Challenges associated with analysis and storage of NGS data Gabriella Rustici Research and training coordinator Functional Genomics Group [email protected] Next-generation sequencing Next-generation sequencing
Comparing Methods for Identifying Transcription Factor Target Genes
 Comparing Methods for Identifying Transcription Factor Target Genes Alena van Bömmel (R 3.3.73) Matthew Huska (R 3.3.18) Max Planck Institute for Molecular Genetics Folie 1 Transcriptional Regulation TF
Comparing Methods for Identifying Transcription Factor Target Genes Alena van Bömmel (R 3.3.73) Matthew Huska (R 3.3.18) Max Planck Institute for Molecular Genetics Folie 1 Transcriptional Regulation TF
Delivering the power of the world s most successful genomics platform
 Delivering the power of the world s most successful genomics platform NextCODE Health is bringing the full power of the world s largest and most successful genomics platform to everyday clinical care NextCODE
Delivering the power of the world s most successful genomics platform NextCODE Health is bringing the full power of the world s largest and most successful genomics platform to everyday clinical care NextCODE
SeqScape Software Version 2.5 Comprehensive Analysis Solution for Resequencing Applications
 Product Bulletin Sequencing Software SeqScape Software Version 2.5 Comprehensive Analysis Solution for Resequencing Applications Comprehensive reference sequence handling Helps interpret the role of each
Product Bulletin Sequencing Software SeqScape Software Version 2.5 Comprehensive Analysis Solution for Resequencing Applications Comprehensive reference sequence handling Helps interpret the role of each
-> Integration of MAPHiTS in Galaxy
 Enabling NGS Analysis with(out) the Infrastructure, 12:0512 Development of a workflow for SNPs detection in grapevine From Sets to Graphs: Towards a Realistic Enrichment Analy species: MAPHiTS -> Integration
Enabling NGS Analysis with(out) the Infrastructure, 12:0512 Development of a workflow for SNPs detection in grapevine From Sets to Graphs: Towards a Realistic Enrichment Analy species: MAPHiTS -> Integration
New Technologies for Sensitive, Low-Input RNA-Seq. Clontech Laboratories, Inc.
 New Technologies for Sensitive, Low-Input RNA-Seq Clontech Laboratories, Inc. Outline Introduction Single-Cell-Capable mrna-seq Using SMART Technology SMARTer Ultra Low RNA Kit for the Fluidigm C 1 System
New Technologies for Sensitive, Low-Input RNA-Seq Clontech Laboratories, Inc. Outline Introduction Single-Cell-Capable mrna-seq Using SMART Technology SMARTer Ultra Low RNA Kit for the Fluidigm C 1 System
GeneSifter: Next Generation Data Management and Analysis for Next Generation Sequencing
 for Next Generation Sequencing Dale Baskin, N. Eric Olson, Laura Lucas, Todd Smith 1 Abstract Next generation sequencing technology is rapidly changing the way laboratories and researchers approach the
for Next Generation Sequencing Dale Baskin, N. Eric Olson, Laura Lucas, Todd Smith 1 Abstract Next generation sequencing technology is rapidly changing the way laboratories and researchers approach the
RETRIEVING SEQUENCE INFORMATION. Nucleotide sequence databases. Database search. Sequence alignment and comparison
 RETRIEVING SEQUENCE INFORMATION Nucleotide sequence databases Database search Sequence alignment and comparison Biological sequence databases Originally just a storage place for sequences. Currently the
RETRIEVING SEQUENCE INFORMATION Nucleotide sequence databases Database search Sequence alignment and comparison Biological sequence databases Originally just a storage place for sequences. Currently the
NGS Data Analysis: An Intro to RNA-Seq
 NGS Data Analysis: An Intro to RNA-Seq March 25th, 2014 GST Colloquim: March 25th, 2014 1 / 1 Workshop Design Basics of NGS Sample Prep RNA-Seq Analysis GST Colloquim: March 25th, 2014 2 / 1 Experimental
NGS Data Analysis: An Intro to RNA-Seq March 25th, 2014 GST Colloquim: March 25th, 2014 1 / 1 Workshop Design Basics of NGS Sample Prep RNA-Seq Analysis GST Colloquim: March 25th, 2014 2 / 1 Experimental
17 July 2014 WEB-SERVER MANUAL. Contact: Michael Hackenberg ([email protected])
 WEB-SERVER MANUAL Contact: Michael Hackenberg ([email protected]) 1 1 Introduction srnabench is a free web-server tool and standalone application for processing small- RNA data obtained from next generation
WEB-SERVER MANUAL Contact: Michael Hackenberg ([email protected]) 1 1 Introduction srnabench is a free web-server tool and standalone application for processing small- RNA data obtained from next generation
RNA Express. Introduction 3 Run RNA Express 4 RNA Express App Output 6 RNA Express Workflow 12 Technical Assistance
 RNA Express Introduction 3 Run RNA Express 4 RNA Express App Output 6 RNA Express Workflow 12 Technical Assistance ILLUMINA PROPRIETARY 15052918 Rev. A February 2014 This document and its contents are
RNA Express Introduction 3 Run RNA Express 4 RNA Express App Output 6 RNA Express Workflow 12 Technical Assistance ILLUMINA PROPRIETARY 15052918 Rev. A February 2014 This document and its contents are
GeneProf and the new GeneProf Web Services
 GeneProf and the new GeneProf Web Services Florian Halbritter [email protected] Stem Cell Bioinformatics Group (Simon R. Tomlinson) [email protected] December 10, 2012 Florian Halbritter
GeneProf and the new GeneProf Web Services Florian Halbritter [email protected] Stem Cell Bioinformatics Group (Simon R. Tomlinson) [email protected] December 10, 2012 Florian Halbritter
Next Generation Sequencing
 Next Generation Sequencing Technology and applications 10/1/2015 Jeroen Van Houdt - Genomics Core - KU Leuven - UZ Leuven 1 Landmarks in DNA sequencing 1953 Discovery of DNA double helix structure 1977
Next Generation Sequencing Technology and applications 10/1/2015 Jeroen Van Houdt - Genomics Core - KU Leuven - UZ Leuven 1 Landmarks in DNA sequencing 1953 Discovery of DNA double helix structure 1977
Go where the biology takes you. Genome Analyzer IIx Genome Analyzer IIe
 Go where the biology takes you. Genome Analyzer IIx Genome Analyzer IIe Go where the biology takes you. To published results faster With proven scalability To the forefront of discovery To limitless applications
Go where the biology takes you. Genome Analyzer IIx Genome Analyzer IIe Go where the biology takes you. To published results faster With proven scalability To the forefront of discovery To limitless applications
Systematic discovery of regulatory motifs in human promoters and 30 UTRs by comparison of several mammals
 Systematic discovery of regulatory motifs in human promoters and 30 UTRs by comparison of several mammals Xiaohui Xie 1, Jun Lu 1, E. J. Kulbokas 1, Todd R. Golub 1, Vamsi Mootha 1, Kerstin Lindblad-Toh
Systematic discovery of regulatory motifs in human promoters and 30 UTRs by comparison of several mammals Xiaohui Xie 1, Jun Lu 1, E. J. Kulbokas 1, Todd R. Golub 1, Vamsi Mootha 1, Kerstin Lindblad-Toh
GenBank, Entrez, & FASTA
 GenBank, Entrez, & FASTA Nucleotide Sequence Databases First generation GenBank is a representative example started as sort of a museum to preserve knowledge of a sequence from first discovery great repositories,
GenBank, Entrez, & FASTA Nucleotide Sequence Databases First generation GenBank is a representative example started as sort of a museum to preserve knowledge of a sequence from first discovery great repositories,
SGI. High Throughput Computing (HTC) Wrapper Program for Bioinformatics on SGI ICE and SGI UV Systems. January, 2012. Abstract. Haruna Cofer*, PhD
 White Paper SGI High Throughput Computing (HTC) Wrapper Program for Bioinformatics on SGI ICE and SGI UV Systems Haruna Cofer*, PhD January, 2012 Abstract The SGI High Throughput Computing (HTC) Wrapper
White Paper SGI High Throughput Computing (HTC) Wrapper Program for Bioinformatics on SGI ICE and SGI UV Systems Haruna Cofer*, PhD January, 2012 Abstract The SGI High Throughput Computing (HTC) Wrapper
mirnaselect pep-mir Cloning and Expression Vector
 Product Data Sheet mirnaselect pep-mir Cloning and Expression Vector CATALOG NUMBER: MIR-EXP-C STORAGE: -80ºC QUANTITY: 2 vectors; each contains 100 µl of bacterial glycerol stock Components 1. mirnaselect
Product Data Sheet mirnaselect pep-mir Cloning and Expression Vector CATALOG NUMBER: MIR-EXP-C STORAGE: -80ºC QUANTITY: 2 vectors; each contains 100 µl of bacterial glycerol stock Components 1. mirnaselect
RNAseq / ChipSeq / Methylseq and personalized genomics
 RNAseq / ChipSeq / Methylseq and personalized genomics 7711 Lecture Subhajyo) De, PhD Division of Biomedical Informa)cs and Personalized Biomedicine, Department of Medicine University of Colorado School
RNAseq / ChipSeq / Methylseq and personalized genomics 7711 Lecture Subhajyo) De, PhD Division of Biomedical Informa)cs and Personalized Biomedicine, Department of Medicine University of Colorado School
Analyzing microrna Data and Integrating mirna with Gene Expression Data in Partek Genomics Suite 6.6
 Analyzing microrna Data and Integrating mirna with Gene Expression Data in Partek Genomics Suite 6.6 Overview This tutorial outlines how microrna data can be analyzed within Partek Genomics Suite. Additionally,
Analyzing microrna Data and Integrating mirna with Gene Expression Data in Partek Genomics Suite 6.6 Overview This tutorial outlines how microrna data can be analyzed within Partek Genomics Suite. Additionally,
Introduction to next-generation sequencing data
 Introduction to next-generation sequencing data David Simpson Centre for Experimental Medicine Queens University Belfast http://www.qub.ac.uk/research-centres/cem/ Outline History of DNA sequencing NGS
Introduction to next-generation sequencing data David Simpson Centre for Experimental Medicine Queens University Belfast http://www.qub.ac.uk/research-centres/cem/ Outline History of DNA sequencing NGS
MultiQuant Software 2.0 for Targeted Protein / Peptide Quantification
 MultiQuant Software 2.0 for Targeted Protein / Peptide Quantification Gold Standard for Quantitative Data Processing Because of the sensitivity, selectivity, speed and throughput at which MRM assays can
MultiQuant Software 2.0 for Targeted Protein / Peptide Quantification Gold Standard for Quantitative Data Processing Because of the sensitivity, selectivity, speed and throughput at which MRM assays can
Exiqon Array Software Manual. Quick guide to data extraction from mircury LNA microrna Arrays
 Exiqon Array Software Manual Quick guide to data extraction from mircury LNA microrna Arrays March 2010 Table of contents Introduction Overview...................................................... 3 ImaGene
Exiqon Array Software Manual Quick guide to data extraction from mircury LNA microrna Arrays March 2010 Table of contents Introduction Overview...................................................... 3 ImaGene
Data Analysis for Ion Torrent Sequencing
 IFU022 v140202 Research Use Only Instructions For Use Part III Data Analysis for Ion Torrent Sequencing MANUFACTURER: Multiplicom N.V. Galileilaan 18 2845 Niel Belgium Revision date: August 21, 2014 Page
IFU022 v140202 Research Use Only Instructions For Use Part III Data Analysis for Ion Torrent Sequencing MANUFACTURER: Multiplicom N.V. Galileilaan 18 2845 Niel Belgium Revision date: August 21, 2014 Page
G E N OM I C S S E RV I C ES
 GENOMICS SERVICES THE NEW YORK GENOME CENTER NYGC is an independent non-profit implementing advanced genomic research to improve diagnosis and treatment of serious diseases. capabilities. N E X T- G E
GENOMICS SERVICES THE NEW YORK GENOME CENTER NYGC is an independent non-profit implementing advanced genomic research to improve diagnosis and treatment of serious diseases. capabilities. N E X T- G E
8/7/2012. Experimental Design & Intro to NGS Data Analysis. Examples. Agenda. Shoe Example. Breast Cancer Example. Rat Example (Experimental Design)
 Experimental Design & Intro to NGS Data Analysis Ryan Peters Field Application Specialist Partek, Incorporated Agenda Experimental Design Examples ANOVA What assays are possible? NGS Analytical Process
Experimental Design & Intro to NGS Data Analysis Ryan Peters Field Application Specialist Partek, Incorporated Agenda Experimental Design Examples ANOVA What assays are possible? NGS Analytical Process
Searching Nucleotide Databases
 Searching Nucleotide Databases 1 When we search a nucleic acid databases, Mascot always performs a 6 frame translation on the fly. That is, 3 reading frames from the forward strand and 3 reading frames
Searching Nucleotide Databases 1 When we search a nucleic acid databases, Mascot always performs a 6 frame translation on the fly. That is, 3 reading frames from the forward strand and 3 reading frames
Scalable Cloud Computing Solutions for Next Generation Sequencing Data
 Scalable Cloud Computing Solutions for Next Generation Sequencing Data Matti Niemenmaa 1, Aleksi Kallio 2, André Schumacher 1, Petri Klemelä 2, Eija Korpelainen 2, and Keijo Heljanko 1 1 Department of
Scalable Cloud Computing Solutions for Next Generation Sequencing Data Matti Niemenmaa 1, Aleksi Kallio 2, André Schumacher 1, Petri Klemelä 2, Eija Korpelainen 2, and Keijo Heljanko 1 1 Department of
Discovery and Quantification of RNA with RNASeq Roderic Guigó Serra Centre de Regulació Genòmica (CRG) [email protected]
 Bioinformatique et Séquençage Haut Débit, Discovery and Quantification of RNA with RNASeq Roderic Guigó Serra Centre de Regulació Genòmica (CRG) [email protected] 1 RNA Transcription to RNA and subsequent
Bioinformatique et Séquençage Haut Débit, Discovery and Quantification of RNA with RNASeq Roderic Guigó Serra Centre de Regulació Genòmica (CRG) [email protected] 1 RNA Transcription to RNA and subsequent
Final Project Report
 CPSC545 by Introduction to Data Mining Prof. Martin Schultz & Prof. Mark Gerstein Student Name: Yu Kor Hugo Lam Student ID : 904907866 Due Date : May 7, 2007 Introduction Final Project Report Pseudogenes
CPSC545 by Introduction to Data Mining Prof. Martin Schultz & Prof. Mark Gerstein Student Name: Yu Kor Hugo Lam Student ID : 904907866 Due Date : May 7, 2007 Introduction Final Project Report Pseudogenes
SUBJECT: New Features in Version 5.3
 User Bulletin Sequencing Analysis Software v5.3 July 2007 SUBJECT: New Features in Version 5.3 This user bulletin includes the following topics: New Features in v5.3.....................................
User Bulletin Sequencing Analysis Software v5.3 July 2007 SUBJECT: New Features in Version 5.3 This user bulletin includes the following topics: New Features in v5.3.....................................
Chapter 2. imapper: A web server for the automated analysis and mapping of insertional mutagenesis sequence data against Ensembl genomes
 Chapter 2. imapper: A web server for the automated analysis and mapping of insertional mutagenesis sequence data against Ensembl genomes 2.1 Introduction Large-scale insertional mutagenesis screening in
Chapter 2. imapper: A web server for the automated analysis and mapping of insertional mutagenesis sequence data against Ensembl genomes 2.1 Introduction Large-scale insertional mutagenesis screening in
The world of non-coding RNA. Espen Enerly
 The world of non-coding RNA Espen Enerly ncrna in general Different groups Small RNAs Outline mirnas and sirnas Speculations Common for all ncrna Per def.: never translated Not spurious transcripts Always/often
The world of non-coding RNA Espen Enerly ncrna in general Different groups Small RNAs Outline mirnas and sirnas Speculations Common for all ncrna Per def.: never translated Not spurious transcripts Always/often
CRAC: An integrated approach to analyse RNA-seq reads Additional File 3 Results on simulated RNA-seq data.
 : An integrated approach to analyse RNA-seq reads Additional File 3 Results on simulated RNA-seq data. Nicolas Philippe and Mikael Salson and Thérèse Commes and Eric Rivals February 13, 2013 1 Results
: An integrated approach to analyse RNA-seq reads Additional File 3 Results on simulated RNA-seq data. Nicolas Philippe and Mikael Salson and Thérèse Commes and Eric Rivals February 13, 2013 1 Results
When you install Mascot, it includes a copy of the Swiss-Prot protein database. However, it is almost certain that you and your colleagues will want
 1 When you install Mascot, it includes a copy of the Swiss-Prot protein database. However, it is almost certain that you and your colleagues will want to search other databases as well. There are very
1 When you install Mascot, it includes a copy of the Swiss-Prot protein database. However, it is almost certain that you and your colleagues will want to search other databases as well. There are very
Mir-X mirna First-Strand Synthesis Kit User Manual
 User Manual Mir-X mirna First-Strand Synthesis Kit User Manual United States/Canada 800.662.2566 Asia Pacific +1.650.919.7300 Europe +33.(0)1.3904.6880 Japan +81.(0)77.543.6116 Clontech Laboratories, Inc.
User Manual Mir-X mirna First-Strand Synthesis Kit User Manual United States/Canada 800.662.2566 Asia Pacific +1.650.919.7300 Europe +33.(0)1.3904.6880 Japan +81.(0)77.543.6116 Clontech Laboratories, Inc.
CD-HIT User s Guide. Last updated: April 5, 2010. http://cd-hit.org http://bioinformatics.org/cd-hit/
 CD-HIT User s Guide Last updated: April 5, 2010 http://cd-hit.org http://bioinformatics.org/cd-hit/ Program developed by Weizhong Li s lab at UCSD http://weizhong-lab.ucsd.edu [email protected] 1. Introduction
CD-HIT User s Guide Last updated: April 5, 2010 http://cd-hit.org http://bioinformatics.org/cd-hit/ Program developed by Weizhong Li s lab at UCSD http://weizhong-lab.ucsd.edu [email protected] 1. Introduction
Analysis of gene expression data. Ulf Leser and Philippe Thomas
 Analysis of gene expression data Ulf Leser and Philippe Thomas This Lecture Protein synthesis Microarray Idea Technologies Applications Problems Quality control Normalization Analysis next week! Ulf Leser:
Analysis of gene expression data Ulf Leser and Philippe Thomas This Lecture Protein synthesis Microarray Idea Technologies Applications Problems Quality control Normalization Analysis next week! Ulf Leser:
Subread/Rsubread Users Guide
 Subread/Rsubread Users Guide Subread v1.5.0-p1/rsubread v1.20.3 1 February 2016 Wei Shi and Yang Liao Bioinformatics Division The Walter and Eliza Hall Institute of Medical Research The University of Melbourne
Subread/Rsubread Users Guide Subread v1.5.0-p1/rsubread v1.20.3 1 February 2016 Wei Shi and Yang Liao Bioinformatics Division The Walter and Eliza Hall Institute of Medical Research The University of Melbourne
Next Generation Sequencing: Technology, Mapping, and Analysis
 Next Generation Sequencing: Technology, Mapping, and Analysis Gary Benson Computer Science, Biology, Bioinformatics Boston University [email protected] http://tandem.bu.edu/ The Human Genome Project took
Next Generation Sequencing: Technology, Mapping, and Analysis Gary Benson Computer Science, Biology, Bioinformatics Boston University [email protected] http://tandem.bu.edu/ The Human Genome Project took
Introduction to Bioinformatics 3. DNA editing and contig assembly
 Introduction to Bioinformatics 3. DNA editing and contig assembly Benjamin F. Matthews United States Department of Agriculture Soybean Genomics and Improvement Laboratory Beltsville, MD 20708 [email protected]
Introduction to Bioinformatics 3. DNA editing and contig assembly Benjamin F. Matthews United States Department of Agriculture Soybean Genomics and Improvement Laboratory Beltsville, MD 20708 [email protected]
Quality Assessment of Exon and Gene Arrays
 Quality Assessment of Exon and Gene Arrays I. Introduction In this white paper we describe some quality assessment procedures that are computed from CEL files from Whole Transcript (WT) based arrays such
Quality Assessment of Exon and Gene Arrays I. Introduction In this white paper we describe some quality assessment procedures that are computed from CEL files from Whole Transcript (WT) based arrays such
Micro RNAs: potentielle Biomarker für das. Blutspenderscreening
 Micro RNAs: potentielle Biomarker für das Blutspenderscreening micrornas - Background Types of RNA -Coding: messenger RNA (mrna) -Non-coding (examples): Ribosomal RNA (rrna) Transfer RNA (trna) Small nuclear
Micro RNAs: potentielle Biomarker für das Blutspenderscreening micrornas - Background Types of RNA -Coding: messenger RNA (mrna) -Non-coding (examples): Ribosomal RNA (rrna) Transfer RNA (trna) Small nuclear
A Tutorial in Genetic Sequence Classification Tools and Techniques
 A Tutorial in Genetic Sequence Classification Tools and Techniques Jake Drew Data Mining CSE 8331 Southern Methodist University [email protected] www.jakemdrew.com Sequence Characters IUPAC nucleotide
A Tutorial in Genetic Sequence Classification Tools and Techniques Jake Drew Data Mining CSE 8331 Southern Methodist University [email protected] www.jakemdrew.com Sequence Characters IUPAC nucleotide
Deep Sequencing Data Analysis
 Deep Sequencing Data Analysis Ross Whetten Professor Forestry & Environmental Resources Background Who am I, and why am I teaching this topic? I am not an expert in bioinformatics I started as a biologist
Deep Sequencing Data Analysis Ross Whetten Professor Forestry & Environmental Resources Background Who am I, and why am I teaching this topic? I am not an expert in bioinformatics I started as a biologist
Gene Expression Analysis
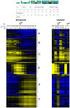 Gene Expression Analysis Jie Peng Department of Statistics University of California, Davis May 2012 RNA expression technologies High-throughput technologies to measure the expression levels of thousands
Gene Expression Analysis Jie Peng Department of Statistics University of California, Davis May 2012 RNA expression technologies High-throughput technologies to measure the expression levels of thousands
From DNA to Protein. Proteins. Chapter 13. Prokaryotes and Eukaryotes. The Path From Genes to Proteins. All proteins consist of polypeptide chains
 Proteins From DNA to Protein Chapter 13 All proteins consist of polypeptide chains A linear sequence of amino acids Each chain corresponds to the nucleotide base sequence of a gene The Path From Genes
Proteins From DNA to Protein Chapter 13 All proteins consist of polypeptide chains A linear sequence of amino acids Each chain corresponds to the nucleotide base sequence of a gene The Path From Genes
Version 5.0 Release Notes
 Version 5.0 Release Notes 2011 Gene Codes Corporation Gene Codes Corporation 775 Technology Drive, Ann Arbor, MI 48108 USA 1.800.497.4939 (USA) +1.734.769.7249 (elsewhere) +1.734.769.7074 (fax) www.genecodes.com
Version 5.0 Release Notes 2011 Gene Codes Corporation Gene Codes Corporation 775 Technology Drive, Ann Arbor, MI 48108 USA 1.800.497.4939 (USA) +1.734.769.7249 (elsewhere) +1.734.769.7074 (fax) www.genecodes.com
Basic processing of next-generation sequencing (NGS) data
 Basic processing of next-generation sequencing (NGS) data Getting from raw sequence data to expression analysis! 1 Reminder: we are measuring expression of protein coding genes by transcript abundance
Basic processing of next-generation sequencing (NGS) data Getting from raw sequence data to expression analysis! 1 Reminder: we are measuring expression of protein coding genes by transcript abundance
UGENE Quick Start Guide
 Quick Start Guide This document contains a quick introduction to UGENE. For more detailed information, you can find the UGENE User Manual and other special manuals in project website: http://ugene.unipro.ru.
Quick Start Guide This document contains a quick introduction to UGENE. For more detailed information, you can find the UGENE User Manual and other special manuals in project website: http://ugene.unipro.ru.
Focusing on results not data comprehensive data analysis for targeted next generation sequencing
 Focusing on results not data comprehensive data analysis for targeted next generation sequencing Daniel Swan, Jolyon Holdstock, Angela Matchan, Richard Stark, John Shovelton, Duarte Mohla and Simon Hughes
Focusing on results not data comprehensive data analysis for targeted next generation sequencing Daniel Swan, Jolyon Holdstock, Angela Matchan, Richard Stark, John Shovelton, Duarte Mohla and Simon Hughes
Gene Models & Bed format: What they represent.
 GeneModels&Bedformat:Whattheyrepresent. Gene models are hypotheses about the structure of transcripts produced by a gene. Like all models, they may be correct, partly correct, or entirely wrong. Typically,
GeneModels&Bedformat:Whattheyrepresent. Gene models are hypotheses about the structure of transcripts produced by a gene. Like all models, they may be correct, partly correct, or entirely wrong. Typically,
Bioruptor NGS: Unbiased DNA shearing for Next-Generation Sequencing
 STGAAC STGAACT GTGCACT GTGAACT STGAAC STGAACT GTGCACT GTGAACT STGAAC STGAAC GTGCAC GTGAAC Wouter Coppieters Head of the genomics core facility GIGA center, University of Liège Bioruptor NGS: Unbiased DNA
STGAAC STGAACT GTGCACT GTGAACT STGAAC STGAACT GTGCACT GTGAACT STGAAC STGAAC GTGCAC GTGAAC Wouter Coppieters Head of the genomics core facility GIGA center, University of Liège Bioruptor NGS: Unbiased DNA
Removing Sequential Bottlenecks in Analysis of Next-Generation Sequencing Data
 Removing Sequential Bottlenecks in Analysis of Next-Generation Sequencing Data Yi Wang, Gagan Agrawal, Gulcin Ozer and Kun Huang The Ohio State University HiCOMB 2014 May 19 th, Phoenix, Arizona 1 Outline
Removing Sequential Bottlenecks in Analysis of Next-Generation Sequencing Data Yi Wang, Gagan Agrawal, Gulcin Ozer and Kun Huang The Ohio State University HiCOMB 2014 May 19 th, Phoenix, Arizona 1 Outline
Module 1. Sequence Formats and Retrieval. Charles Steward
 The Open Door Workshop Module 1 Sequence Formats and Retrieval Charles Steward 1 Aims Acquaint you with different file formats and associated annotations. Introduce different nucleotide and protein databases.
The Open Door Workshop Module 1 Sequence Formats and Retrieval Charles Steward 1 Aims Acquaint you with different file formats and associated annotations. Introduce different nucleotide and protein databases.
AGILENT S BIOINFORMATICS ANALYSIS SOFTWARE
 ACCELERATING PROGRESS IS IN OUR GENES AGILENT S BIOINFORMATICS ANALYSIS SOFTWARE GENESPRING GENE EXPRESSION (GX) MASS PROFILER PROFESSIONAL (MPP) PATHWAY ARCHITECT (PA) See Deeper. Reach Further. BIOINFORMATICS
ACCELERATING PROGRESS IS IN OUR GENES AGILENT S BIOINFORMATICS ANALYSIS SOFTWARE GENESPRING GENE EXPRESSION (GX) MASS PROFILER PROFESSIONAL (MPP) PATHWAY ARCHITECT (PA) See Deeper. Reach Further. BIOINFORMATICS
Geospiza s Finch-Server: A Complete Data Management System for DNA Sequencing
 KOO10 5/31/04 12:17 PM Page 131 10 Geospiza s Finch-Server: A Complete Data Management System for DNA Sequencing Sandra Porter, Joe Slagel, and Todd Smith Geospiza, Inc., Seattle, WA Introduction The increased
KOO10 5/31/04 12:17 PM Page 131 10 Geospiza s Finch-Server: A Complete Data Management System for DNA Sequencing Sandra Porter, Joe Slagel, and Todd Smith Geospiza, Inc., Seattle, WA Introduction The increased
GenomeStudio Data Analysis Software
 GenomeStudio Analysis Software Illumina has created a comprehensive suite of data analysis tools to support a wide range of genetic analysis assays. This single software package provides data visualization
GenomeStudio Analysis Software Illumina has created a comprehensive suite of data analysis tools to support a wide range of genetic analysis assays. This single software package provides data visualization
Core Facility Genomics
 Core Facility Genomics versatile genome or transcriptome analyses based on quantifiable highthroughput data ascertainment 1 Topics Collaboration with Harald Binder and Clemens Kreutz Project: Microarray
Core Facility Genomics versatile genome or transcriptome analyses based on quantifiable highthroughput data ascertainment 1 Topics Collaboration with Harald Binder and Clemens Kreutz Project: Microarray
New solutions for Big Data Analysis and Visualization
 New solutions for Big Data Analysis and Visualization From HPC to cloud-based solutions Barcelona, February 2013 Nacho Medina [email protected] http://bioinfo.cipf.es/imedina Head of the Computational Biology
New solutions for Big Data Analysis and Visualization From HPC to cloud-based solutions Barcelona, February 2013 Nacho Medina [email protected] http://bioinfo.cipf.es/imedina Head of the Computational Biology
SeqPig: simple and scalable scripting for large sequencing data sets in Hadoop
 SeqPig: simple and scalable scripting for large sequencing data sets in Hadoop André Schumacher, Luca Pireddu, Matti Niemenmaa, Aleksi Kallio, Eija Korpelainen, Gianluigi Zanetti and Keijo Heljanko Abstract
SeqPig: simple and scalable scripting for large sequencing data sets in Hadoop André Schumacher, Luca Pireddu, Matti Niemenmaa, Aleksi Kallio, Eija Korpelainen, Gianluigi Zanetti and Keijo Heljanko Abstract
Bioinformatics Resources at a Glance
 Bioinformatics Resources at a Glance A Note about FASTA Format There are MANY free bioinformatics tools available online. Bioinformaticists have developed a standard format for nucleotide and protein sequences
Bioinformatics Resources at a Glance A Note about FASTA Format There are MANY free bioinformatics tools available online. Bioinformaticists have developed a standard format for nucleotide and protein sequences
Outline. MicroRNA Bioinformatics. microrna biogenesis. short non-coding RNAs not considered in this lecture. ! Introduction
 Outline MicroRNA Bioinformatics Rickard Sandberg Dept. of Cell and Molecular Biology (CMB) Karolinska Institutet! Introduction! microrna target site prediction! Useful resources 2 short non-coding RNAs
Outline MicroRNA Bioinformatics Rickard Sandberg Dept. of Cell and Molecular Biology (CMB) Karolinska Institutet! Introduction! microrna target site prediction! Useful resources 2 short non-coding RNAs
Data Processing of Nextera Mate Pair Reads on Illumina Sequencing Platforms
 Data Processing of Nextera Mate Pair Reads on Illumina Sequencing Platforms Introduction Mate pair sequencing enables the generation of libraries with insert sizes in the range of several kilobases (Kb).
Data Processing of Nextera Mate Pair Reads on Illumina Sequencing Platforms Introduction Mate pair sequencing enables the generation of libraries with insert sizes in the range of several kilobases (Kb).
PrimePCR Assay Validation Report
 Gene Information Gene Name Gene Symbol Organism Gene Summary Gene Aliases RefSeq Accession No. UniGene ID Ensembl Gene ID papillary renal cell carcinoma (translocation-associated) PRCC Human This gene
Gene Information Gene Name Gene Symbol Organism Gene Summary Gene Aliases RefSeq Accession No. UniGene ID Ensembl Gene ID papillary renal cell carcinoma (translocation-associated) PRCC Human This gene
OpenCB development - A Big Data analytics and visualisation platform for the Omics revolution
 OpenCB development - A Big Data analytics and visualisation platform for the Omics revolution Ignacio Medina, Paul Calleja, John Taylor (University of Cambridge, UIS, HPC Service (HPCS)) Abstract The advent
OpenCB development - A Big Data analytics and visualisation platform for the Omics revolution Ignacio Medina, Paul Calleja, John Taylor (University of Cambridge, UIS, HPC Service (HPCS)) Abstract The advent
Real-time PCR: Understanding C t
 APPLICATION NOTE Real-Time PCR Real-time PCR: Understanding C t Real-time PCR, also called quantitative PCR or qpcr, can provide a simple and elegant method for determining the amount of a target sequence
APPLICATION NOTE Real-Time PCR Real-time PCR: Understanding C t Real-time PCR, also called quantitative PCR or qpcr, can provide a simple and elegant method for determining the amount of a target sequence
Leading Genomics. Diagnostic. Discove. Collab. harma. Shanghai Cambridge, MA Reykjavik
 Leading Genomics Diagnostic harma Discove Collab Shanghai Cambridge, MA Reykjavik Global leadership for using the genome to create better medicine WuXi NextCODE provides a uniquely proven and integrated
Leading Genomics Diagnostic harma Discove Collab Shanghai Cambridge, MA Reykjavik Global leadership for using the genome to create better medicine WuXi NextCODE provides a uniquely proven and integrated
Development of Monitoring and Analysis Tools for the Huawei Cloud Storage
 Development of Monitoring and Analysis Tools for the Huawei Cloud Storage September 2014 Author: Veronia Bahaa Supervisors: Maria Arsuaga-Rios Seppo S. Heikkila CERN openlab Summer Student Report 2014
Development of Monitoring and Analysis Tools for the Huawei Cloud Storage September 2014 Author: Veronia Bahaa Supervisors: Maria Arsuaga-Rios Seppo S. Heikkila CERN openlab Summer Student Report 2014
Thermo Scientific DyNAmo cdna Synthesis Kit for qrt-pcr Technical Manual
 Thermo Scientific DyNAmo cdna Synthesis Kit for qrt-pcr Technical Manual F- 470S 20 cdna synthesis reactions (20 µl each) F- 470L 100 cdna synthesis reactions (20 µl each) Table of contents 1. Description...
Thermo Scientific DyNAmo cdna Synthesis Kit for qrt-pcr Technical Manual F- 470S 20 cdna synthesis reactions (20 µl each) F- 470L 100 cdna synthesis reactions (20 µl each) Table of contents 1. Description...
DNA Replication & Protein Synthesis. This isn t a baaaaaaaddd chapter!!!
 DNA Replication & Protein Synthesis This isn t a baaaaaaaddd chapter!!! The Discovery of DNA s Structure Watson and Crick s discovery of DNA s structure was based on almost fifty years of research by other
DNA Replication & Protein Synthesis This isn t a baaaaaaaddd chapter!!! The Discovery of DNA s Structure Watson and Crick s discovery of DNA s structure was based on almost fifty years of research by other
Genetic information (DNA) determines structure of proteins DNA RNA proteins cell structure 3.11 3.15 enzymes control cell chemistry ( metabolism )
 Biology 1406 Exam 3 Notes Structure of DNA Ch. 10 Genetic information (DNA) determines structure of proteins DNA RNA proteins cell structure 3.11 3.15 enzymes control cell chemistry ( metabolism ) Proteins
Biology 1406 Exam 3 Notes Structure of DNA Ch. 10 Genetic information (DNA) determines structure of proteins DNA RNA proteins cell structure 3.11 3.15 enzymes control cell chemistry ( metabolism ) Proteins
HADOOP ON ORACLE ZFS STORAGE A TECHNICAL OVERVIEW
 HADOOP ON ORACLE ZFS STORAGE A TECHNICAL OVERVIEW 757 Maleta Lane, Suite 201 Castle Rock, CO 80108 Brett Weninger, Managing Director [email protected] Dave Smelker, Managing Principal [email protected]
HADOOP ON ORACLE ZFS STORAGE A TECHNICAL OVERVIEW 757 Maleta Lane, Suite 201 Castle Rock, CO 80108 Brett Weninger, Managing Director [email protected] Dave Smelker, Managing Principal [email protected]
Just the Facts: A Basic Introduction to the Science Underlying NCBI Resources
 1 of 8 11/7/2004 11:00 AM National Center for Biotechnology Information About NCBI NCBI at a Glance A Science Primer Human Genome Resources Model Organisms Guide Outreach and Education Databases and Tools
1 of 8 11/7/2004 11:00 AM National Center for Biotechnology Information About NCBI NCBI at a Glance A Science Primer Human Genome Resources Model Organisms Guide Outreach and Education Databases and Tools
Using Galaxy for NGS Analysis. Daniel Blankenberg Postdoctoral Research Associate The Galaxy Team http://usegalaxy.org
 Using Galaxy for NGS Analysis Daniel Blankenberg Postdoctoral Research Associate The Galaxy Team http://usegalaxy.org Overview NGS Data Galaxy tools for NGS Data Galaxy for Sequencing Facilities Overview
Using Galaxy for NGS Analysis Daniel Blankenberg Postdoctoral Research Associate The Galaxy Team http://usegalaxy.org Overview NGS Data Galaxy tools for NGS Data Galaxy for Sequencing Facilities Overview
BioHPC Web Computing Resources at CBSU
 BioHPC Web Computing Resources at CBSU 3CPG workshop Robert Bukowski Computational Biology Service Unit http://cbsu.tc.cornell.edu/lab/doc/biohpc_web_tutorial.pdf BioHPC infrastructure at CBSU BioHPC Web
BioHPC Web Computing Resources at CBSU 3CPG workshop Robert Bukowski Computational Biology Service Unit http://cbsu.tc.cornell.edu/lab/doc/biohpc_web_tutorial.pdf BioHPC infrastructure at CBSU BioHPC Web
Processing Genome Data using Scalable Database Technology. My Background
 Johann Christoph Freytag, Ph.D. [email protected] http://www.dbis.informatik.hu-berlin.de Stanford University, February 2004 PhD @ Harvard Univ. Visiting Scientist, Microsoft Res. (2002)
Johann Christoph Freytag, Ph.D. [email protected] http://www.dbis.informatik.hu-berlin.de Stanford University, February 2004 PhD @ Harvard Univ. Visiting Scientist, Microsoft Res. (2002)
How-To: SNP and INDEL detection
 How-To: SNP and INDEL detection April 23, 2014 Lumenogix NGS SNP and INDEL detection Mutation Analysis Identifying known, and discovering novel genomic mutations, has been one of the most popular applications
How-To: SNP and INDEL detection April 23, 2014 Lumenogix NGS SNP and INDEL detection Mutation Analysis Identifying known, and discovering novel genomic mutations, has been one of the most popular applications
NEXT GENERATION SEQUENCING
 NEXT GENERATION SEQUENCING Dr. R. Piazza SANGER SEQUENCING + DNA NEXT GENERATION SEQUENCING Flowcell NEXT GENERATION SEQUENCING Library di DNA Genomic DNA NEXT GENERATION SEQUENCING NEXT GENERATION SEQUENCING
NEXT GENERATION SEQUENCING Dr. R. Piazza SANGER SEQUENCING + DNA NEXT GENERATION SEQUENCING Flowcell NEXT GENERATION SEQUENCING Library di DNA Genomic DNA NEXT GENERATION SEQUENCING NEXT GENERATION SEQUENCING
Name Class Date. Figure 13 1. 2. Which nucleotide in Figure 13 1 indicates the nucleic acid above is RNA? a. uracil c. cytosine b. guanine d.
 13 Multiple Choice RNA and Protein Synthesis Chapter Test A Write the letter that best answers the question or completes the statement on the line provided. 1. Which of the following are found in both
13 Multiple Choice RNA and Protein Synthesis Chapter Test A Write the letter that best answers the question or completes the statement on the line provided. 1. Which of the following are found in both
Next Generation Sequencing: Adjusting to Big Data. Daniel Nicorici, Dr.Tech. Statistikot Suomen Lääketeollisuudessa 29.10.2013
 Next Generation Sequencing: Adjusting to Big Data Daniel Nicorici, Dr.Tech. Statistikot Suomen Lääketeollisuudessa 29.10.2013 Outline Human Genome Project Next-Generation Sequencing Personalized Medicine
Next Generation Sequencing: Adjusting to Big Data Daniel Nicorici, Dr.Tech. Statistikot Suomen Lääketeollisuudessa 29.10.2013 Outline Human Genome Project Next-Generation Sequencing Personalized Medicine
Data Analytics. Sequence Exosome RNAs
 Sequence Exosome RNAs The Exo-NGS service provides the exosome researcher with a comprehensive, expert service to isolate and identify exosome-associated RNA biomarkers - leveraging the throughput and
Sequence Exosome RNAs The Exo-NGS service provides the exosome researcher with a comprehensive, expert service to isolate and identify exosome-associated RNA biomarkers - leveraging the throughput and
How Sequencing Experiments Fail
 How Sequencing Experiments Fail v1.0 Simon Andrews [email protected] Classes of Failure Technical Tracking Library Contamination Biological Interpretation Something went wrong with a machine
How Sequencing Experiments Fail v1.0 Simon Andrews [email protected] Classes of Failure Technical Tracking Library Contamination Biological Interpretation Something went wrong with a machine
Disease gene identification with exome sequencing
 Disease gene identification with exome sequencing Christian Gilissen Dept. of Human Genetics Radboud University Nijmegen Medical Centre [email protected] Contents Infrastructure Exome sequencing
Disease gene identification with exome sequencing Christian Gilissen Dept. of Human Genetics Radboud University Nijmegen Medical Centre [email protected] Contents Infrastructure Exome sequencing
Translation Study Guide
 Translation Study Guide This study guide is a written version of the material you have seen presented in the replication unit. In translation, the cell uses the genetic information contained in mrna to
Translation Study Guide This study guide is a written version of the material you have seen presented in the replication unit. In translation, the cell uses the genetic information contained in mrna to
