Ion Formation Grade Nine
|
|
|
- Andrea McLaughlin
- 9 years ago
- Views:
Transcription
1 Ohio Standards Connection: Physical Sciences Benchmark A Describe that matter is made of minute particles called atoms and atoms are comprised of even smaller components. Explain the structure and properties of atoms. Indicator 5 Describe how ions are formed when an atom or a group of atoms acquire an unbalanced charge by gaining or losing one or more electrons. Lesson Summary: In this lesson, students will review atomic structure using two-color counters (plastic disks that are red on one side and yellow on the other) to represent positive and negative charges in the formation of ions. Estimated Duration: One hour Commentary: This lesson was designed to introduce students to an abstract concept by working with concrete models. Students may have encountered two-color counters in mathematics when studying positive and negative numbers, so it may be an easy transition to use them to represent protons and electrons. After using the counters to represent several examples of ion formation, students should find it easier to mentally determine the charge on an ion without using the models. This lesson was reviewed by educators across the state. Some of their comments were: Having students draw diagrams and then use their writing skills to describe ion formation is an excellent learning activity. Ion formation is an abstract idea. Any hands-on activity will help. 1
2 Pre-Assessment: Based on what students have previously learned, select and ask appropriate questions from the following questions. 1. What is an atom? 2. What are its parts? 3. What is an electron? 4. What is a proton? 5. What is a neutron? 6. What does it mean to charge an atom? 7. Describe the structure of atoms. 8. What does the atomic number of an atom represent? 9. What does the mass number of an atom represent? 10. What does the periodic table tell us about the atoms of various elements? 11. Can atoms lose electrons? Gain electrons? 12. What are compounds? 13. How are compounds formed? 14. What is a valence electron? 15. What is the Bohr electronic structure of an atom? 16. How is the Lewis dot configuration used to represent atoms? 17. How are ions formed? 18. What is a positive ion? 19. What is a negative ion? 20. What is a polyatomic ion? Ask some questions to ensure that students know how to add negative numbers. Provide a review as needed. Scoring Guideline: This pre-assessment is intended for the teacher s use in determining students knowledge of background information about atoms, compounds, charging and related concepts and deciding which topics need to be reviewed before or during this lesson. If students do not understand the parts of an atom, atomic number, mass number, valence electrons and electronic structure, then these topics should be taught before going into the instructional procedures of this lesson. Post-Assessment: Ask students to prepare essays and related diagrams or other graphics that describe how atoms become ions (both cations and anions). Have students include in their discussions the types and numbers of subatomic particles (electron, protons and neutrons) that affect the charge on the ion. See Attachment B, Post-Assessment, for a student handout. 2
3 Scoring Guidelines: A good response should include the following key points about each ion: 1. The number of electrons, protons and neutrons in the neutral atom; 2. The number of electrons that are lost or gained; 3. A justification for the charge on the ion; 4. A statement that indicates the neutrons have no effect on the charge. Instructional Procedures: Part I: Introductory prompt 1. Tell students the following joke. Two atoms have been traveling separately and when they meet at an intersection, they have this conversation: First atom: I think I just lost an electron. Second atom: Are you sure? First atom: Yes, I m rather positive. Second atom: I m getting a negative feeling about this! 2. Ask the students, What causes some atoms to respond this way? 3. After pausing to see how many students have the background to understand the humor, cover some scientific topics related to the simple story that extend their knowledge. Part II: Modeling Ions 4. Have students use two-color counters (plastic disks that are red on one side and yellow on the other) to represent positive and negative charges. (Two-color counters are commonly used mathematics manipulatives and may be borrowed from the mathematics department.) Similar disks that can be used on the overhead are available. Some students may benefit by marking the counters with positive or negative signs to represent positive and negative charges. 5. Ask students, What does the atomic number represent? (The number of protons or the number of electrons in a neutral atom) Then ask, What is meant when an atom is considered neutral? (There is no net charge; the number of positively charged particles is equal to the number of negatively charged particles.) 6. Have the students complete the following steps: Select an element with a small atomic number, such as lithium. Find its atomic number on the periodic table. Represent the electrons and protons with the two-color counters. (For lithium, students will use six two-color counters, three with the red side up to represent the protons and three with the yellow side up to represent the electrons.) 7. Check to see that the counters are being used correctly, and then ask, Why is lithium neutral? 3
4 8. After receiving correct answers or guiding students to the correct answers have the students complete the following steps: Have students remove one electron (one yellow disk). Ask them, Is the atom still neutral? Why or why not? What is the net charge on the atom after it lost one electron? (Positive one) What is the net charge when the neutrons of the atom are also considered? (The charge does not change because the neutrons are neutral and do not add any charge to the atom.) 9. Have students pair up the protons and electrons and point out that when the protons and electrons are paired, there is no net charge on the atom. If there are excess protons or electrons, there is a net charge and it is equal to the number of excess protons (positive) or excess electrons (negative). At this point, consider providing information about the number of electrons that lithium is most likely to lose, telling the students that in most compounds, lithium has lost only one electron. 10. Have the students complete the following steps: Select beryllium, another element with a small atomic number. Find its atomic number on the periodic table. Represent the electrons and protons with the two-color counters. (For beryllium, students will use eight two-color counters, four with the red side up to represent the protons and four with the yellow side up to represent the electrons.) 11. After learning that the students have represented beryllium correctly and view it as neutral, have students remove one electron from the beryllium atom and predict the resulting charge, and then remove a second electron and predict the charge. (Positive one; then positive two). If timely and appropriate, inform students that beryllium is most likely to lose two electrons, and that in most compounds, beryllium has lost two electrons. 12. Ask, What are charged atoms called? (Ions) Tell students that positively charged ions are called cations. 13. When students have mastered the concept of positive ion formation, shift the activity to negative ion formation by completing the following steps: Have students represent fluorine with the two-color counters. Have students add one electron to fluorine. Ask them Is the fluorine atom still neutral? Why or why not? What is the net charge on the atom after it gained one electron? (Negative one) 4
5 If timely and appropriate, inform students that fluorine atoms seek to gain electrons and, in compounds, fluorine gains one electron. You can be assured that fluorine will nearly always take an electron from any atom it meets at the corner. 14. Ask students to find the atomic number of oxygen, an element whose atoms are likely to gain electrons, and represent its number of electrons and protons with the two-color counters. 15. Take these four actions: Tell students to add one electron to oxygen. Ask them Is the oxygen atom still neutral? Why or why not? What is the net charge on the atom after it gained one electron? (Negative one) Tell them to add another electron and predict the net charge. (Negative two) If timely and appropriate, let students know that oxygen is most likely to gain two electrons and in most compounds of oxygen, the oxygen atom has gained two electrons. 16. Continue the activity with hydrogen by completing the following steps: Have students represent hydrogen with the two-color counters. Have students remove one electron from hydrogen. Ask them Is the hydrogen atom still neutral? Why or why not? What is the net charge on the atom after it lost one electron? (Positive one) 17. Have students restore hydrogen to its neutral state and then add an additional electron. Ask them Is the hydrogen atom still neutral? Why or why not? What is the net charge on the hydrogen atom after it gained one electron? (Negative one) Tell the students that hydrogen, like some other elements, may form ions by gaining an electron or by losing an electron. To add real-life examples to the lesson, ask What happens when hydrogen burns? (Two hydrogen atoms each give up an electron and one oxygen atom gains two electrons to form water.) 18. Ask, What are negatively charged atoms called? (All charged atoms are called ions, and negatively charged ions are called anions. ) 19. Point out that, so far, the class has used only elements of Periods 1 and 2 of the Periodic Table (first and second horizontal rows), and then say: How do we represent ion formation by the many other elements that are known to exist? What is the atomic number of iodine? (53) 5
6 How many colored counters would we need to represent the electrons and protons of iodine? (106) Wow! Do we have enough to deal with bromine and other elements with high atomic numbers? (Tell the students that we usually represent only those electrons that are valence electrons.) 20. Review or guide a class discussion of the following topics: Atomic structure; Bohr electronic structures; Valence electrons. Depending on the background of the students, it may be appropriate to: Have students predict how many electrons an atom is most likely to gain or lose. Guide students to an understanding of the relationships with the periodic table and the electrons an element is likely to gain or lose. 21. Choose some elements from the third, fourth and fifth periods and have the students go through abbreviated steps similar to those used above, using valence electrons. 22. Encourage students to determine the charges of the resulting ions without relying on the counters: Iodine gaining one electron (negative one); Calcium losing two electrons (positive two); Sodium losing one electron (positive one). 23. At some point, begin to ask students to use the chemical symbols for the atoms and the ions, as they continue to form the following ions: Sulfur gaining two electrons (negative two); Aluminum losing three electrons (positive three); Iron losing two electrons, then losing another electron; (positive two, then positive three); Argon What would Argon do? (Argon and the other noble gases normally do not form ions and compounds, hence the terms, noble or inert gases!) 24. To add other real-life examples to the lesson, ask What happens when sodium reacts with chlorine to form salt? (A sodium atom gives up an electron to become a Na +1 ion and a chlorine atom gains an electron to become a Cl -1 ion. The two oppositely charged ions are attracted to each other (ionic bond), forming NaCl.) Other examples are iron rusting to become iron oxide, silver tarnishing into silver sulfide and copper tarnishing (oxidizing) into cupric oxide. 25. Conclude the lesson with a review of the concepts and general points covered in class about atoms, electrons, neutral atoms, formation of ions by loss or gain of electrons, etc. 26. Give students some practice questions and ask them to review their notes and their texts; discuss what they learned and what they may not understand with fellow students; ask 6
7 questions of the teacher as needed; and prepare for an exam on a later class day. See Attachment A, Homework for Ion Formation. 27. As the class ends, give this advice to students; Be careful out there and watch your electrons! You never know what kind of atoms you may meet! 28. During the next class, collect and review the homework. Review the information and concepts as needed. Ensure that students know: Electronic structure of atoms; Valence electrons; Process by which ions are formed by atoms or groups of atoms; Ions may be positively or negatively charged; Symbols used for atoms and ions; Ions typically have charges of 3, -2, -1, +1, +2 or +3; Some elements form positive and negative ions; Noble gases normally do not form ions and compounds. 29. Ask students to reflect on how this lesson increased their understanding of atoms and ions and how they would explain to a family member how atoms become ions. Differentiated Instructional Support: Instruction is differentiated according to learner needs, to help all learners either meet the intent of the specified indicator(s) or, if the indicator is already met, to advance beyond the specified indicator(s). Students may work independently, in pairs or heterogeneous groups. Reinforce understanding of lesson vocabulary by providing accurate definitions of terms. Utilize graphic organizers to categorize information about atomic structure, Bohr electronic structures and valence electrons. Challenge students to identify an ion of interest and conduct research to share with the class. Extensions: Discuss chemical reactivity in relation to the ease of formation of ions. Discuss ionic character and the properties of ionic compounds, like solubility. Show the relationship between the periodic table and types of ions formed. Include the naming of ions in the lesson, especially the more common ones. Discuss the formation and naming of polyatomic ions. Homework Options Examine labels in home or in stores to see which ions may be in common substances. 7
8 Materials and Resources: The inclusion of a specific resource in any lesson formulated by the Ohio Department of Education should not be interpreted as an endorsement of that particular resource, or any of its contents, by the Ohio Department of Education. The Ohio Department of Education does not endorse any particular resource. The Web addresses listed are for a given site s main page, therefore, it may be necessary to search within that site to find the specific information required for a given lesson. Please note that information published on the Internet changes over time, therefore the links provided may no longer contain the specific information related to a given lesson. Teachers are advised to preview all sites before using them with students. For the teacher: Colored plastic disks for the overhead. For the students: Two-color counters (plastic disks that are red on one side and yellow on the other), periodic table. Vocabulary: atoms molecules ions elements chemical symbols cations anions electrons compounds protons neutrons nucleus orbit reactions charges inert polyatomic atomic structure valence periodic table groups periods Technology Connections: Have students visit Internet sites for practice, similar lessons, film clips, etc. Use computer animations to illustrate the process of ion formation and bonding. 8
9 Research Connections: Marzano, R. et al. Classroom Instruction that Works: Research-Based Strategies for Increasing Student Achievement. Alexandria: Association for Supervision and Curriculum Development, Nonlinguistic representations help students think about and recall knowledge. They include the following: Creating graphic representations (organizers); Making physical models; Generating mental pictures; Drawing pictures and pictographs; Engaging in kinesthetic activity. General Tips: Commercial materials are available with shapes that help students match the ions. These materials may help students learn how to form polyatomic ions and, later, to balance ionic compounds. Attachments: Attachment A, Homework for Ion Formation Attachment B, Post-Assessment 9
10 Attachment A Homework for Ion Formation 1. Use your textbook and notes to review these terms and concepts: atoms, molecules, ions, elements, chemical symbols, cations, anions, electrons, compounds, protons, neutrons, nucleus, orbit, reactions, charges, inert, polyatomic, atomic structure, valence, periodic table, groups and periods. 2. In your own words, explain to your relatives, how ions are formed by the loss or gain of electrons. 3. Write a short description of how each of the following ions might have been formed. Use any diagrams and symbols that will help. K +1, H -1, Ca +2, Fe +3, Br -1, O -2, N -3 10
11 Attachment B Post-Assessment Prepare an essay with related diagrams or other graphics that describe how the following became ions (both cations and anions). Include in your discussion the types and numbers of subatomic particles (electron, protons and neutrons) that affect the charge on the ion. Na +1, Mg +2, Fe +3, Br -1, S -2 11
7.4. Using the Bohr Theory KNOW? Using the Bohr Theory to Describe Atoms and Ions
 7.4 Using the Bohr Theory LEARNING TIP Models such as Figures 1 to 4, on pages 218 and 219, help you visualize scientific explanations. As you examine Figures 1 to 4, look back and forth between the diagrams
7.4 Using the Bohr Theory LEARNING TIP Models such as Figures 1 to 4, on pages 218 and 219, help you visualize scientific explanations. As you examine Figures 1 to 4, look back and forth between the diagrams
Untitled Document. 1. Which of the following best describes an atom? 4. Which statement best describes the density of an atom s nucleus?
 Name: Date: 1. Which of the following best describes an atom? A. protons and electrons grouped together in a random pattern B. protons and electrons grouped together in an alternating pattern C. a core
Name: Date: 1. Which of the following best describes an atom? A. protons and electrons grouped together in a random pattern B. protons and electrons grouped together in an alternating pattern C. a core
Chapter 5 TEST: The Periodic Table name
 Chapter 5 TEST: The Periodic Table name HPS # date: Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The order of elements in the periodic table is based
Chapter 5 TEST: The Periodic Table name HPS # date: Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The order of elements in the periodic table is based
Name Class Date. What is ionic bonding? What happens to atoms that gain or lose electrons? What kinds of solids are formed from ionic bonds?
 CHAPTER 1 2 Ionic Bonds SECTION Chemical Bonding BEFORE YOU READ After you read this section, you should be able to answer these questions: What is ionic bonding? What happens to atoms that gain or lose
CHAPTER 1 2 Ionic Bonds SECTION Chemical Bonding BEFORE YOU READ After you read this section, you should be able to answer these questions: What is ionic bonding? What happens to atoms that gain or lose
Test Bank - Chapter 4 Multiple Choice
 Test Bank - Chapter 4 The questions in the test bank cover the concepts from the lessons in Chapter 4. Select questions from any of the categories that match the content you covered with students. The
Test Bank - Chapter 4 The questions in the test bank cover the concepts from the lessons in Chapter 4. Select questions from any of the categories that match the content you covered with students. The
Ionic and Metallic Bonding
 Ionic and Metallic Bonding BNDING AND INTERACTINS 71 Ions For students using the Foundation edition, assign problems 1, 3 5, 7 12, 14, 15, 18 20 Essential Understanding Ions form when atoms gain or lose
Ionic and Metallic Bonding BNDING AND INTERACTINS 71 Ions For students using the Foundation edition, assign problems 1, 3 5, 7 12, 14, 15, 18 20 Essential Understanding Ions form when atoms gain or lose
Unit 3 Study Guide: Electron Configuration & The Periodic Table
 Name: Teacher s Name: Class: Block: Date: Unit 3 Study Guide: Electron Configuration & The Periodic Table 1. For each of the following elements, state whether the element is radioactive, synthetic or both.
Name: Teacher s Name: Class: Block: Date: Unit 3 Study Guide: Electron Configuration & The Periodic Table 1. For each of the following elements, state whether the element is radioactive, synthetic or both.
UNIT (2) ATOMS AND ELEMENTS
 UNIT (2) ATOMS AND ELEMENTS 2.1 Elements An element is a fundamental substance that cannot be broken down by chemical means into simpler substances. Each element is represented by an abbreviation called
UNIT (2) ATOMS AND ELEMENTS 2.1 Elements An element is a fundamental substance that cannot be broken down by chemical means into simpler substances. Each element is represented by an abbreviation called
SCPS Chemistry Worksheet Periodicity A. Periodic table 1. Which are metals? Circle your answers: C, Na, F, Cs, Ba, Ni
 SCPS Chemistry Worksheet Periodicity A. Periodic table 1. Which are metals? Circle your answers: C, Na, F, Cs, Ba, Ni Which metal in the list above has the most metallic character? Explain. Cesium as the
SCPS Chemistry Worksheet Periodicity A. Periodic table 1. Which are metals? Circle your answers: C, Na, F, Cs, Ba, Ni Which metal in the list above has the most metallic character? Explain. Cesium as the
Periodic Table Questions
 Periodic Table Questions 1. The elements characterized as nonmetals are located in the periodic table at the (1) far left; (2) bottom; (3) center; (4) top right. 2. An element that is a liquid at STP is
Periodic Table Questions 1. The elements characterized as nonmetals are located in the periodic table at the (1) far left; (2) bottom; (3) center; (4) top right. 2. An element that is a liquid at STP is
Part I: Principal Energy Levels and Sublevels
 Part I: Principal Energy Levels and Sublevels As you already know, all atoms are made of subatomic particles, including protons, neutrons, and electrons. Positive protons and neutral neutrons are found
Part I: Principal Energy Levels and Sublevels As you already know, all atoms are made of subatomic particles, including protons, neutrons, and electrons. Positive protons and neutral neutrons are found
******* KEY ******* Atomic Structure & Periodic Table Test Study Guide
 Atomic Structure & Periodic Table Test Study Guide VOCABULARY: Write a brief definition of each term in the space provided. 1. Atoms: smallest unit of an element that has all of the properties of that
Atomic Structure & Periodic Table Test Study Guide VOCABULARY: Write a brief definition of each term in the space provided. 1. Atoms: smallest unit of an element that has all of the properties of that
List the 3 main types of subatomic particles and indicate the mass and electrical charge of each.
 Basic Chemistry Why do we study chemistry in a biology course? All living organisms are composed of chemicals. To understand life, we must understand the structure, function, and properties of the chemicals
Basic Chemistry Why do we study chemistry in a biology course? All living organisms are composed of chemicals. To understand life, we must understand the structure, function, and properties of the chemicals
47374_04_p25-32.qxd 2/9/07 7:50 AM Page 25. 4 Atoms and Elements
 47374_04_p25-32.qxd 2/9/07 7:50 AM Page 25 4 Atoms and Elements 4.1 a. Cu b. Si c. K d. N e. Fe f. Ba g. Pb h. Sr 4.2 a. O b. Li c. S d. Al e. H f. Ne g. Sn h. Au 4.3 a. carbon b. chlorine c. iodine d.
47374_04_p25-32.qxd 2/9/07 7:50 AM Page 25 4 Atoms and Elements 4.1 a. Cu b. Si c. K d. N e. Fe f. Ba g. Pb h. Sr 4.2 a. O b. Li c. S d. Al e. H f. Ne g. Sn h. Au 4.3 a. carbon b. chlorine c. iodine d.
ANSWER KEY : BUILD AN ATOM PART I: ATOM SCREEN Build an Atom simulation ( http://phet.colorado.edu/en/simulation/build an atom )
 ANSWER KEY : PART I: ATOM SCREEN Build an Atom simulation ( http://phet.colorado.edu/en/simulation/build an atom ) 1. Explore the Build an Atom simulation with your group. As you explore, talk about what
ANSWER KEY : PART I: ATOM SCREEN Build an Atom simulation ( http://phet.colorado.edu/en/simulation/build an atom ) 1. Explore the Build an Atom simulation with your group. As you explore, talk about what
Chemistry CP Unit 2 Atomic Structure and Electron Configuration. Learning Targets (Your exam at the end of Unit 2 will assess the following:)
 Chemistry CP Unit 2 Atomic Structure and Electron Learning Targets (Your exam at the end of Unit 2 will assess the following:) 2. Atomic Structure and Electron 2-1. Give the one main contribution to the
Chemistry CP Unit 2 Atomic Structure and Electron Learning Targets (Your exam at the end of Unit 2 will assess the following:) 2. Atomic Structure and Electron 2-1. Give the one main contribution to the
Molecular Models in Biology
 Molecular Models in Biology Objectives: After this lab a student will be able to: 1) Understand the properties of atoms that give rise to bonds. 2) Understand how and why atoms form ions. 3) Model covalent,
Molecular Models in Biology Objectives: After this lab a student will be able to: 1) Understand the properties of atoms that give rise to bonds. 2) Understand how and why atoms form ions. 3) Model covalent,
9/13/2013. However, Dalton thought that an atom was just a tiny sphere with no internal parts. This is sometimes referred to as the cannonball model.
 John Dalton was an English scientist who lived in the early 1800s. Dalton s atomic theory served as a model for how matter worked. The principles of Dalton s atomic theory are: 1. Elements are made of
John Dalton was an English scientist who lived in the early 1800s. Dalton s atomic theory served as a model for how matter worked. The principles of Dalton s atomic theory are: 1. Elements are made of
Lewis Dot Structures of Atoms and Ions
 Why? The chemical properties of an element are based on the number of electrons in the outer shell of its atoms. We use Lewis dot structures to map these valence electrons in order to identify stable electron
Why? The chemical properties of an element are based on the number of electrons in the outer shell of its atoms. We use Lewis dot structures to map these valence electrons in order to identify stable electron
Chapter 2 Atoms, Ions, and the Periodic Table
 Chapter 2 Atoms, Ions, and the Periodic Table 2.1 (a) neutron; (b) law of conservation of mass; (c) proton; (d) main-group element; (e) relative atomic mass; (f) mass number; (g) isotope; (h) cation; (i)
Chapter 2 Atoms, Ions, and the Periodic Table 2.1 (a) neutron; (b) law of conservation of mass; (c) proton; (d) main-group element; (e) relative atomic mass; (f) mass number; (g) isotope; (h) cation; (i)
Electrons in Atoms & Periodic Table Chapter 13 & 14 Assignment & Problem Set
 Electrons in Atoms & Periodic Table Name Warm-Ups (Show your work for credit) Date 1. Date 2. Date 3. Date 4. Date 5. Date 6. Date 7. Date 8. Electrons in Atoms & Periodic Table 2 Study Guide: Things You
Electrons in Atoms & Periodic Table Name Warm-Ups (Show your work for credit) Date 1. Date 2. Date 3. Date 4. Date 5. Date 6. Date 7. Date 8. Electrons in Atoms & Periodic Table 2 Study Guide: Things You
Chapter 2 Atoms, Molecules, and Ions
 Chapter 2 Atoms, Molecules, and Ions 1. Methane and ethane are both made up of carbon and hydrogen. In methane, there are 12.0 g of carbon for every 4.00 g of hydrogen, a ration of 3:1 by mass. In ethane,
Chapter 2 Atoms, Molecules, and Ions 1. Methane and ethane are both made up of carbon and hydrogen. In methane, there are 12.0 g of carbon for every 4.00 g of hydrogen, a ration of 3:1 by mass. In ethane,
Chapter 2 The Chemical Context of Life
 Chapter 2 The Chemical Context of Life Multiple-Choice Questions 1) About 25 of the 92 natural elements are known to be essential to life. Which four of these 25 elements make up approximately 96% of living
Chapter 2 The Chemical Context of Life Multiple-Choice Questions 1) About 25 of the 92 natural elements are known to be essential to life. Which four of these 25 elements make up approximately 96% of living
The Periodic Table: Periodic trends
 Unit 1 The Periodic Table: Periodic trends There are over one hundred different chemical elements. Some of these elements are familiar to you such as hydrogen, oxygen, nitrogen and carbon. Each one has
Unit 1 The Periodic Table: Periodic trends There are over one hundred different chemical elements. Some of these elements are familiar to you such as hydrogen, oxygen, nitrogen and carbon. Each one has
( + and - ) ( - and - ) ( + and + ) Atoms are mostly empty space. = the # of protons in the nucleus. = the # of protons in the nucleus
 Atoms are mostly empty space Atomic Structure Two regions of every atom: Nucleus - is made of protons and neutrons - is small and dense Electron cloud -is a region where you might find an electron -is
Atoms are mostly empty space Atomic Structure Two regions of every atom: Nucleus - is made of protons and neutrons - is small and dense Electron cloud -is a region where you might find an electron -is
Atoms and Molecules. Preparation. Objectives. Standards. Materials. Grade Level: 5-8 Group Size: 20-30 Time: 60 90 Minutes Presenters: 2-4
 Atoms and Molecules Preparation Grade Level: 5-8 Group Size: 20-30 Time: 60 90 Minutes Presenters: 2-4 Objectives This lesson will enable students to: Describe how atoms are the building blocks of matter
Atoms and Molecules Preparation Grade Level: 5-8 Group Size: 20-30 Time: 60 90 Minutes Presenters: 2-4 Objectives This lesson will enable students to: Describe how atoms are the building blocks of matter
Chapter 2: The Chemical Context of Life
 Chapter 2: The Chemical Context of Life Name Period This chapter covers the basics that you may have learned in your chemistry class. Whether your teacher goes over this chapter, or assigns it for you
Chapter 2: The Chemical Context of Life Name Period This chapter covers the basics that you may have learned in your chemistry class. Whether your teacher goes over this chapter, or assigns it for you
PROTONS AND ELECTRONS
 reflect Imagine that you have a bowl of oranges, bananas, pineapples, berries, pears, and watermelon. How do you identify each piece of fruit? Most likely, you are familiar with the characteristics of
reflect Imagine that you have a bowl of oranges, bananas, pineapples, berries, pears, and watermelon. How do you identify each piece of fruit? Most likely, you are familiar with the characteristics of
Elements, Atoms & Ions
 Introductory Chemistry: A Foundation FOURTH EDITION by Steven S. Zumdahl University of Illinois Elements, Atoms & Ions Chapter 4 1 2 Elements Aims: To learn about the relative abundances of the elements,
Introductory Chemistry: A Foundation FOURTH EDITION by Steven S. Zumdahl University of Illinois Elements, Atoms & Ions Chapter 4 1 2 Elements Aims: To learn about the relative abundances of the elements,
19.1 Bonding and Molecules
 Most of the matter around you and inside of you is in the form of compounds. For example, your body is about 80 percent water. You learned in the last unit that water, H 2 O, is made up of hydrogen and
Most of the matter around you and inside of you is in the form of compounds. For example, your body is about 80 percent water. You learned in the last unit that water, H 2 O, is made up of hydrogen and
Trends of the Periodic Table Diary
 Trends of the Periodic Table Diary Trends are patterns of behaviors that atoms on the periodic table of elements follow. Trends hold true most of the time, but there are exceptions, or blips, where the
Trends of the Periodic Table Diary Trends are patterns of behaviors that atoms on the periodic table of elements follow. Trends hold true most of the time, but there are exceptions, or blips, where the
Ions & Their Charges Worksheet
 Ions & Their Charges Worksheet Name Date Teacher Diagram of charges based on groups on the periodic table including transition metals and noble gases: IA IIA Transition IIIA IVA VA VIA VIIA VIIIA metals
Ions & Their Charges Worksheet Name Date Teacher Diagram of charges based on groups on the periodic table including transition metals and noble gases: IA IIA Transition IIIA IVA VA VIA VIIA VIIIA metals
EXPERIMENT 4 The Periodic Table - Atoms and Elements
 EXPERIMENT 4 The Periodic Table - Atoms and Elements INTRODUCTION Primary substances, called elements, build all the materials around you. There are more than 109 different elements known today. The elements
EXPERIMENT 4 The Periodic Table - Atoms and Elements INTRODUCTION Primary substances, called elements, build all the materials around you. There are more than 109 different elements known today. The elements
ATOMS A T O M S, I S O T O P E S, A N D I O N S. The Academic Support Center @ Daytona State College (Science 120, Page 1 of 39)
 ATOMS A T O M S, I S O T O P E S, A N D I O N S The Academic Support Center @ Daytona State College (Science 120, Page 1 of 39) THE ATOM All elements listed on the periodic table are made up of atoms.
ATOMS A T O M S, I S O T O P E S, A N D I O N S The Academic Support Center @ Daytona State College (Science 120, Page 1 of 39) THE ATOM All elements listed on the periodic table are made up of atoms.
Local Government and Leaders Grade Three
 Ohio Standards Connection: Government Benchmark A Identify the responsibilities of the branches of the U.S. government and explain why they are necessary. Indicator 2 Explain the structure of local governments
Ohio Standards Connection: Government Benchmark A Identify the responsibilities of the branches of the U.S. government and explain why they are necessary. Indicator 2 Explain the structure of local governments
Exam 2 Chemistry 65 Summer 2015. Score:
 Name: Exam 2 Chemistry 65 Summer 2015 Score: Instructions: Clearly circle the one best answer 1. Valence electrons are electrons located A) in the outermost energy level of an atom. B) in the nucleus of
Name: Exam 2 Chemistry 65 Summer 2015 Score: Instructions: Clearly circle the one best answer 1. Valence electrons are electrons located A) in the outermost energy level of an atom. B) in the nucleus of
B) atomic number C) both the solid and the liquid phase D) Au C) Sn, Si, C A) metal C) O, S, Se C) In D) tin D) methane D) bismuth B) Group 2 metal
 1. The elements on the Periodic Table are arranged in order of increasing A) atomic mass B) atomic number C) molar mass D) oxidation number 2. Which list of elements consists of a metal, a metalloid, and
1. The elements on the Periodic Table are arranged in order of increasing A) atomic mass B) atomic number C) molar mass D) oxidation number 2. Which list of elements consists of a metal, a metalloid, and
19.2 Chemical Formulas
 In the previous section, you learned how and why atoms form chemical bonds with one another. You also know that atoms combine in certain ratios with other atoms. These ratios determine the chemical formula
In the previous section, you learned how and why atoms form chemical bonds with one another. You also know that atoms combine in certain ratios with other atoms. These ratios determine the chemical formula
2. John Dalton did his research work in which of the following countries? a. France b. Greece c. Russia d. England
 CHAPTER 3 1. Which combination of individual and contribution is not correct? a. Antoine Lavoisier - clarified confusion over cause of burning b. John Dalton - proposed atomic theory c. Marie Curie - discovered
CHAPTER 3 1. Which combination of individual and contribution is not correct? a. Antoine Lavoisier - clarified confusion over cause of burning b. John Dalton - proposed atomic theory c. Marie Curie - discovered
6.5 Periodic Variations in Element Properties
 324 Chapter 6 Electronic Structure and Periodic Properties of Elements 6.5 Periodic Variations in Element Properties By the end of this section, you will be able to: Describe and explain the observed trends
324 Chapter 6 Electronic Structure and Periodic Properties of Elements 6.5 Periodic Variations in Element Properties By the end of this section, you will be able to: Describe and explain the observed trends
Chapter 6 Assessment. Name: Class: Date: ID: A. Multiple Choice Identify the choice that best completes the statement or answers the question.
 Name: Class: Date: ID: A Chapter 6 Assessment Multiple Choice Identify the choice that best completes the statement or answers the question. 1. When an atom loses an electron, it forms a(n) a. anion. c.
Name: Class: Date: ID: A Chapter 6 Assessment Multiple Choice Identify the choice that best completes the statement or answers the question. 1. When an atom loses an electron, it forms a(n) a. anion. c.
Chapter 7 Periodic Properties of the Elements
 Chapter 7 Periodic Properties of the Elements 1. Elements in the modern version of the periodic table are arranged in order of increasing. (a). oxidation number (b). atomic mass (c). average atomic mass
Chapter 7 Periodic Properties of the Elements 1. Elements in the modern version of the periodic table are arranged in order of increasing. (a). oxidation number (b). atomic mass (c). average atomic mass
Elements in the periodic table are indicated by SYMBOLS. To the left of the symbol we find the atomic mass (A) at the upper corner, and the atomic num
 . ATOMIC STRUCTURE FUNDAMENTALS LEARNING OBJECTIVES To review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. This material is essential
. ATOMIC STRUCTURE FUNDAMENTALS LEARNING OBJECTIVES To review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. This material is essential
Bonding Practice Problems
 NAME 1. When compared to H 2 S, H 2 O has a higher 8. Given the Lewis electron-dot diagram: boiling point because H 2 O contains stronger metallic bonds covalent bonds ionic bonds hydrogen bonds 2. Which
NAME 1. When compared to H 2 S, H 2 O has a higher 8. Given the Lewis electron-dot diagram: boiling point because H 2 O contains stronger metallic bonds covalent bonds ionic bonds hydrogen bonds 2. Which
Laboratory 11: Molecular Compounds and Lewis Structures
 Introduction Laboratory 11: Molecular Compounds and Lewis Structures Molecular compounds are formed by sharing electrons between non-metal atoms. A useful theory for understanding the formation of molecular
Introduction Laboratory 11: Molecular Compounds and Lewis Structures Molecular compounds are formed by sharing electrons between non-metal atoms. A useful theory for understanding the formation of molecular
Objectives. PAM1014 Introduction to Radiation Physics. Constituents of Atoms. Atoms. Atoms. Atoms. Basic Atomic Theory
 PAM1014 Introduction to Radiation Physics Basic Atomic Theory Objectives Introduce and Molecules The periodic Table Electronic Energy Levels Atomic excitation & de-excitation Ionisation Molecules Constituents
PAM1014 Introduction to Radiation Physics Basic Atomic Theory Objectives Introduce and Molecules The periodic Table Electronic Energy Levels Atomic excitation & de-excitation Ionisation Molecules Constituents
CHEMISTRY BONDING REVIEW
 Answer the following questions. CHEMISTRY BONDING REVIEW 1. What are the three kinds of bonds which can form between atoms? The three types of Bonds are Covalent, Ionic and Metallic. Name Date Block 2.
Answer the following questions. CHEMISTRY BONDING REVIEW 1. What are the three kinds of bonds which can form between atoms? The three types of Bonds are Covalent, Ionic and Metallic. Name Date Block 2.
neutrons are present?
 AP Chem Summer Assignment Worksheet #1 Atomic Structure 1. a) For the ion 39 K +, state how many electrons, how many protons, and how many 19 neutrons are present? b) Which of these particles has the smallest
AP Chem Summer Assignment Worksheet #1 Atomic Structure 1. a) For the ion 39 K +, state how many electrons, how many protons, and how many 19 neutrons are present? b) Which of these particles has the smallest
Changes in Our Sky Grade Two
 Ohio Standards Connection: Earth and Space Sciences Benchmark A Observe constant and changing patterns of objects in the day and night sky. Indicator 2 Observe and describe how the sun, moon and stars
Ohio Standards Connection: Earth and Space Sciences Benchmark A Observe constant and changing patterns of objects in the day and night sky. Indicator 2 Observe and describe how the sun, moon and stars
Find a pair of elements in the periodic table with atomic numbers less than 20 that are an exception to the original periodic law.
 Example Exercise 6.1 Periodic Law Find the two elements in the fifth row of the periodic table that violate the original periodic law proposed by Mendeleev. Mendeleev proposed that elements be arranged
Example Exercise 6.1 Periodic Law Find the two elements in the fifth row of the periodic table that violate the original periodic law proposed by Mendeleev. Mendeleev proposed that elements be arranged
Instructors Guide: Atoms and Their Isotopes
 Instructors Guide: Atoms and Their Isotopes Standards Connections Connections to NSTA Standards for Science Teacher Preparation C.3.a.1 Fundamental structures of atoms and molecules. C.3.b.27 Applications
Instructors Guide: Atoms and Their Isotopes Standards Connections Connections to NSTA Standards for Science Teacher Preparation C.3.a.1 Fundamental structures of atoms and molecules. C.3.b.27 Applications
Lewis Dot Notation Ionic Bonds Covalent Bonds Polar Covalent Bonds Lewis Dot Notation Revisited Resonance
 Lewis Dot Notation Ionic Bonds Covalent Bonds Polar Covalent Bonds Lewis Dot Notation Revisited Resonance Lewis Dot notation is a way of describing the outer shell (also called the valence shell) of an
Lewis Dot Notation Ionic Bonds Covalent Bonds Polar Covalent Bonds Lewis Dot Notation Revisited Resonance Lewis Dot notation is a way of describing the outer shell (also called the valence shell) of an
Chapter 5. Chapter 5. Naming Ionic Compounds. Objectives. Chapter 5. Chapter 5
 Objectives Name cations, anions, and ionic compounds. Write chemical formulas for ionic compounds such that an overall neutral charge is maintained. Explain how polyatomic ions and their salts are named
Objectives Name cations, anions, and ionic compounds. Write chemical formulas for ionic compounds such that an overall neutral charge is maintained. Explain how polyatomic ions and their salts are named
Honors Chemistry: Unit 6 Test Stoichiometry PRACTICE TEST ANSWER KEY Page 1. A chemical equation. (C-4.4)
 Honors Chemistry: Unit 6 Test Stoichiometry PRACTICE TEST ANSWER KEY Page 1 1. 2. 3. 4. 5. 6. Question What is a symbolic representation of a chemical reaction? What 3 things (values) is a mole of a chemical
Honors Chemistry: Unit 6 Test Stoichiometry PRACTICE TEST ANSWER KEY Page 1 1. 2. 3. 4. 5. 6. Question What is a symbolic representation of a chemical reaction? What 3 things (values) is a mole of a chemical
2 The Structure of Atoms
 CHAPTER 4 2 The Structure of Atoms SECTION Atoms KEY IDEAS As you read this section, keep these questions in mind: What do atoms of the same element have in common? What are isotopes? How is an element
CHAPTER 4 2 The Structure of Atoms SECTION Atoms KEY IDEAS As you read this section, keep these questions in mind: What do atoms of the same element have in common? What are isotopes? How is an element
Naming and Writing Formulas for Ionic Compounds Using IUPAC Rules
 Naming and Writing Formulas for Ionic Compounds Using IUPAC Rules There are three categories of ionic compounds that we will deal with. 1.Binary ionic o simple ions (only single charges) o multivalent
Naming and Writing Formulas for Ionic Compounds Using IUPAC Rules There are three categories of ionic compounds that we will deal with. 1.Binary ionic o simple ions (only single charges) o multivalent
Electrons In Atoms Mr. O Brien (SFHS) Chapter 5 Standard 1D
 Electrons In Atoms Mr. O Brien (SFHS) Chapter 5 Standard 1D Electrons in Atoms (std.1d) What are Bohr Models? planetary model in which the negatively-charged electrons orbit a small, positively-charged
Electrons In Atoms Mr. O Brien (SFHS) Chapter 5 Standard 1D Electrons in Atoms (std.1d) What are Bohr Models? planetary model in which the negatively-charged electrons orbit a small, positively-charged
Investigating Quadrilaterals Grade Four
 Ohio Standards Connection Geometry and Spatial Sense Benchmark A Provide rationale for groupings and comparisons of two-dimensional figures and three-dimensional objects. Indicator 3 Identify similarities
Ohio Standards Connection Geometry and Spatial Sense Benchmark A Provide rationale for groupings and comparisons of two-dimensional figures and three-dimensional objects. Indicator 3 Identify similarities
The Acid Test Grade Nine
 Ohio Standards Connection: Physical Sciences Benchmark B Explain how atoms react with each other to form other substances and how molecules react with each other or other atoms to form even different substances.
Ohio Standards Connection: Physical Sciences Benchmark B Explain how atoms react with each other to form other substances and how molecules react with each other or other atoms to form even different substances.
ANSWER KEY. Energy Levels, Electrons and IONIC Bonding It s all about the Give and Take!
 ANSWER KEY Energy Levels, Electrons and IONIC Bonding It s all about the Give and Take! From American Chemical Society Middle School Chemistry Unit: Chapter 4 Content Statements: Distinguish the difference
ANSWER KEY Energy Levels, Electrons and IONIC Bonding It s all about the Give and Take! From American Chemical Society Middle School Chemistry Unit: Chapter 4 Content Statements: Distinguish the difference
Commutative Property Grade One
 Ohio Standards Connection Patterns, Functions and Algebra Benchmark E Solve open sentences and explain strategies. Indicator 4 Solve open sentences by representing an expression in more than one way using
Ohio Standards Connection Patterns, Functions and Algebra Benchmark E Solve open sentences and explain strategies. Indicator 4 Solve open sentences by representing an expression in more than one way using
Bohr s Model of the Atom
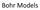 Bohr Models Bohr s Model of the Atom Focuses on electrons and their arrangement. Bohr stated that electrons move with constant speed in fixed orbits around the nucleus, like planets around a sun. Bohr
Bohr Models Bohr s Model of the Atom Focuses on electrons and their arrangement. Bohr stated that electrons move with constant speed in fixed orbits around the nucleus, like planets around a sun. Bohr
PERIODIC TABLE OF GROUPS OF ELEMENTS Elements can be classified using two different schemes.
 1 PERIODIC TABLE OF GROUPS OF ELEMENTS Elements can be classified using two different schemes. Metal Nonmetal Scheme (based on physical properties) Metals - most elements are metals - elements on left
1 PERIODIC TABLE OF GROUPS OF ELEMENTS Elements can be classified using two different schemes. Metal Nonmetal Scheme (based on physical properties) Metals - most elements are metals - elements on left
Chapter 16: Tests for ions and gases
 The position of hydrogen in the reactivity series Hydrogen, although not a metal, is included in the reactivity series because it, like metals, can be displaced from aqueous solution, only this time the
The position of hydrogen in the reactivity series Hydrogen, although not a metal, is included in the reactivity series because it, like metals, can be displaced from aqueous solution, only this time the
Name period AP chemistry Unit 2 worksheet Practice problems
 Name period AP chemistry Unit 2 worksheet Practice problems 1. What are the SI units for a. Wavelength of light b. frequency of light c. speed of light Meter hertz (s -1 ) m s -1 (m/s) 2. T/F (correct
Name period AP chemistry Unit 2 worksheet Practice problems 1. What are the SI units for a. Wavelength of light b. frequency of light c. speed of light Meter hertz (s -1 ) m s -1 (m/s) 2. T/F (correct
Name: Worksheet: Electron Configurations. I Heart Chemistry!
 1. Which electron configuration represents an atom in an excited state? 1s 2 2s 2 2p 6 3p 1 1s 2 2s 2 2p 6 3s 2 3p 2 1s 2 2s 2 2p 6 3s 2 3p 1 1s 2 2s 2 2p 6 3s 2 Worksheet: Electron Configurations Name:
1. Which electron configuration represents an atom in an excited state? 1s 2 2s 2 2p 6 3p 1 1s 2 2s 2 2p 6 3s 2 3p 2 1s 2 2s 2 2p 6 3s 2 3p 1 1s 2 2s 2 2p 6 3s 2 Worksheet: Electron Configurations Name:
ATOMS AND THE PERIODIC TABLE CHAPTER 3 PHYSICAL SCIENCE
 ATOMS AND THE PERIODIC TABLE CHAPTER 3 PHYSICAL SCIENCE Chapter 3 Vocabulary Words (27 words) Nucleus Atomic number Proton Mass number Neutron Isotopes Electron Atomic mass unit (amu) Energy level Average
ATOMS AND THE PERIODIC TABLE CHAPTER 3 PHYSICAL SCIENCE Chapter 3 Vocabulary Words (27 words) Nucleus Atomic number Proton Mass number Neutron Isotopes Electron Atomic mass unit (amu) Energy level Average
Sample Exercise 8.1 Magnitudes of Lattice Energies
 Sample Exercise 8.1 Magnitudes of Lattice Energies Without consulting Table 8.2, arrange the ionic compounds NaF, CsI, and CaO in order of increasing lattice energy. Analyze From the formulas for three
Sample Exercise 8.1 Magnitudes of Lattice Energies Without consulting Table 8.2, arrange the ionic compounds NaF, CsI, and CaO in order of increasing lattice energy. Analyze From the formulas for three
Chapter 8 Concepts of Chemical Bonding
 Chapter 8 Concepts of Chemical Bonding Chemical Bonds Three types: Ionic Electrostatic attraction between ions Covalent Sharing of electrons Metallic Metal atoms bonded to several other atoms Ionic Bonding
Chapter 8 Concepts of Chemical Bonding Chemical Bonds Three types: Ionic Electrostatic attraction between ions Covalent Sharing of electrons Metallic Metal atoms bonded to several other atoms Ionic Bonding
Electron Configurations, Isoelectronic Elements, & Ionization Reactions. Chemistry 11
 Electron Configurations, Isoelectronic Elements, & Ionization Reactions Chemistry 11 Note: Of the 3 subatomic particles, the electron plays the greatest role in determining the physical and chemical properties
Electron Configurations, Isoelectronic Elements, & Ionization Reactions Chemistry 11 Note: Of the 3 subatomic particles, the electron plays the greatest role in determining the physical and chemical properties
Chapter 7. Electron Structure of the Atom. Chapter 7 Topics
 Chapter 7 Electron Structure of the Atom Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. 1 Chapter 7 Topics 1. Electromagnetic radiation 2. The Bohr model of
Chapter 7 Electron Structure of the Atom Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. 1 Chapter 7 Topics 1. Electromagnetic radiation 2. The Bohr model of
Plot Connections Grade Five
 Ohio Standards Connection Reading Applications: Literary Text Benchmark C Identify the elements of plot and establish a connection between an element and a future event. Indicator 3 Identify the main incidents
Ohio Standards Connection Reading Applications: Literary Text Benchmark C Identify the elements of plot and establish a connection between an element and a future event. Indicator 3 Identify the main incidents
The Atom and the Periodic Table. Electron Cloud Structure Energy Levels Rows on the Periodic Table Bohr Models Electron Dot Diagrams
 The Atom and the Periodic Table Electron Cloud Structure Energy Levels Rows on the Periodic Table Bohr Models Electron Dot Diagrams Review The vertical columns in the periodic table are called groups.
The Atom and the Periodic Table Electron Cloud Structure Energy Levels Rows on the Periodic Table Bohr Models Electron Dot Diagrams Review The vertical columns in the periodic table are called groups.
APS Science Curriculum Unit Planner
 APS Science Curriculum Unit Planner Grade Level/Subject Chemistry Stage 1: Desired Results Enduring Understanding Topic 1: Elements and the Periodic Table: The placement of elements on the periodic table
APS Science Curriculum Unit Planner Grade Level/Subject Chemistry Stage 1: Desired Results Enduring Understanding Topic 1: Elements and the Periodic Table: The placement of elements on the periodic table
Chapter 3, Elements, Atoms, Ions, and the Periodic Table
 1. Which two scientists in 1869 arranged the elements in order of increasing atomic masses to form a precursor of the modern periodic table of elements? Ans. Mendeleev and Meyer 2. Who stated that the
1. Which two scientists in 1869 arranged the elements in order of increasing atomic masses to form a precursor of the modern periodic table of elements? Ans. Mendeleev and Meyer 2. Who stated that the
Chemistry Diagnostic Questions
 Chemistry Diagnostic Questions Answer these 40 multiple choice questions and then check your answers, located at the end of this document. If you correctly answered less than 25 questions, you need to
Chemistry Diagnostic Questions Answer these 40 multiple choice questions and then check your answers, located at the end of this document. If you correctly answered less than 25 questions, you need to
Geometric Transformations Grade Four
 Ohio Standards Connection Geometry and Spatial Sense Benchmark I Describe, identify and model reflections, rotations and translations, using physical materials. Indicator 7 Identify, describe and use reflections
Ohio Standards Connection Geometry and Spatial Sense Benchmark I Describe, identify and model reflections, rotations and translations, using physical materials. Indicator 7 Identify, describe and use reflections
Urbanization Grade Nine
 Ohio Standards Connection: Geography Benchmark B Analyze geographic changes brought about by human activity using appropriate maps and other geographical data. Indicator 4 Explain the causes and consequences
Ohio Standards Connection: Geography Benchmark B Analyze geographic changes brought about by human activity using appropriate maps and other geographical data. Indicator 4 Explain the causes and consequences
TOPIC 7. CHEMICAL CALCULATIONS I - atomic and formula weights.
 TOPIC 7. CHEMICAL CALCULATIONS I - atomic and formula weights. Atomic structure revisited. In Topic 2, atoms were described as ranging from the simplest atom, H, containing a single proton and usually
TOPIC 7. CHEMICAL CALCULATIONS I - atomic and formula weights. Atomic structure revisited. In Topic 2, atoms were described as ranging from the simplest atom, H, containing a single proton and usually
Chemical Building Blocks: Chapter 3: Elements and Periodic Table
 Name: Class: Date: Chemical Building Blocks: Chapter 3: Elements and Periodic Table Study Guide Multiple Choice Identify the letter of the choice that best completes the statement or answers the question.
Name: Class: Date: Chemical Building Blocks: Chapter 3: Elements and Periodic Table Study Guide Multiple Choice Identify the letter of the choice that best completes the statement or answers the question.
Elements of a Novel and Narrative Writing Grade 10
 Ohio Standards Connection Literary Text Benchmark B Explain and analyze how the context of setting and the author s choice of point of view impact a literary text. Indicator 2 Analyze the features of setting
Ohio Standards Connection Literary Text Benchmark B Explain and analyze how the context of setting and the author s choice of point of view impact a literary text. Indicator 2 Analyze the features of setting
CHEM 150 Exam 1 KEY Name Multiple Choice
 CEM 150 Exam 1 KEY Name Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. _B 1. Which of the following is synonymous with "fact"? a. a hypothesis
CEM 150 Exam 1 KEY Name Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. _B 1. Which of the following is synonymous with "fact"? a. a hypothesis
Atomic Structure. Name Mass Charge Location Protons 1 +1 Nucleus Neutrons 1 0 Nucleus Electrons 1/1837-1 Orbit nucleus in outer shells
 Atomic Structure called nucleons Name Mass Charge Location Protons 1 +1 Nucleus Neutrons 1 0 Nucleus Electrons 1/1837-1 Orbit nucleus in outer shells The number of protons equals the atomic number This
Atomic Structure called nucleons Name Mass Charge Location Protons 1 +1 Nucleus Neutrons 1 0 Nucleus Electrons 1/1837-1 Orbit nucleus in outer shells The number of protons equals the atomic number This
Chapter 5 Periodic Table. Dmitri Mendeleev: Russian Chemist credited with the discovery of the periodic table.
 Chapter 5 Periodic Table Dmitri Mendeleev: Russian Chemist credited with the discovery of the periodic table. How did he organize the elements? According to similarities in their chemical and physical
Chapter 5 Periodic Table Dmitri Mendeleev: Russian Chemist credited with the discovery of the periodic table. How did he organize the elements? According to similarities in their chemical and physical
5. Structure, Geometry, and Polarity of Molecules
 5. Structure, Geometry, and Polarity of Molecules What you will accomplish in this experiment This experiment will give you an opportunity to draw Lewis structures of covalent compounds, then use those
5. Structure, Geometry, and Polarity of Molecules What you will accomplish in this experiment This experiment will give you an opportunity to draw Lewis structures of covalent compounds, then use those
Energy Chains Grade Seven
 Ohio Standards Connection: Physical Science Benchmark D Describe that energy takes many forms, some forms represent kinetic energy and some forms represent potential energy; and during energy transformations
Ohio Standards Connection: Physical Science Benchmark D Describe that energy takes many forms, some forms represent kinetic energy and some forms represent potential energy; and during energy transformations
3. What would you predict for the intensity and binding energy for the 3p orbital for that of sulfur?
 PSI AP Chemistry Periodic Trends MC Review Name Periodic Law and the Quantum Model Use the PES spectrum of Phosphorus below to answer questions 1-3. 1. Which peak corresponds to the 1s orbital? (A) 1.06
PSI AP Chemistry Periodic Trends MC Review Name Periodic Law and the Quantum Model Use the PES spectrum of Phosphorus below to answer questions 1-3. 1. Which peak corresponds to the 1s orbital? (A) 1.06
Cambridge International Examinations Cambridge International General Certificate of Secondary Education
 Cambridge International Examinations Cambridge International General Certificate of Secondary Education *0123456789* CHEMISTRY 0620/03 Paper 3 Theory (Core) For Examination from 2016 SPECIMEN PAPER 1 hour
Cambridge International Examinations Cambridge International General Certificate of Secondary Education *0123456789* CHEMISTRY 0620/03 Paper 3 Theory (Core) For Examination from 2016 SPECIMEN PAPER 1 hour
Trends of the Periodic Table Basics
 Trends of the Periodic Table Basics Trends are patterns of behaviors that atoms on the periodic table of elements follow. Trends hold true most of the time, but there are exceptions, or blips, where the
Trends of the Periodic Table Basics Trends are patterns of behaviors that atoms on the periodic table of elements follow. Trends hold true most of the time, but there are exceptions, or blips, where the
Chapter 4, Lesson 2: The Periodic Table
 Chapter 4, Lesson 2: The Periodic Table Key Concepts The periodic table is a chart containing information about the atoms that make up all matter. An element is a substance made up of only one type of
Chapter 4, Lesson 2: The Periodic Table Key Concepts The periodic table is a chart containing information about the atoms that make up all matter. An element is a substance made up of only one type of
Chemistry: The Periodic Table and Periodicity
 Chemistry: The Periodic Table and Periodicity Name: per: Date:. 1. By what property did Mendeleev arrange the elements? 2. By what property did Moseley suggest that the periodic table be arranged? 3. What
Chemistry: The Periodic Table and Periodicity Name: per: Date:. 1. By what property did Mendeleev arrange the elements? 2. By what property did Moseley suggest that the periodic table be arranged? 3. What
Molecular Models & Lewis Dot Structures
 Molecular Models & Lewis Dot Structures Objectives: 1. Draw Lewis structures for atoms, ions and simple molecules. 2. Use Lewis structures as a guide to construct three-dimensional models of small molecules.
Molecular Models & Lewis Dot Structures Objectives: 1. Draw Lewis structures for atoms, ions and simple molecules. 2. Use Lewis structures as a guide to construct three-dimensional models of small molecules.
Chemistry Post-Enrolment Worksheet
 Name: Chemistry Post-Enrolment Worksheet The purpose of this worksheet is to get you to recap some of the fundamental concepts that you studied at GCSE and introduce some of the concepts that will be part
Name: Chemistry Post-Enrolment Worksheet The purpose of this worksheet is to get you to recap some of the fundamental concepts that you studied at GCSE and introduce some of the concepts that will be part
Systems of Transportation and Communication Grade Three
 1 Ohio Standards Connection: Geography Benchmark D Analyze ways that transportation and communication relate to patterns of settlement and economic activity. Indicator 8 Identify systems of transportation
1 Ohio Standards Connection: Geography Benchmark D Analyze ways that transportation and communication relate to patterns of settlement and economic activity. Indicator 8 Identify systems of transportation
P. Table & E Configuration Practice TEST
 P. Table & E Configuration Practice TEST Multiple Choice Identify the choice that best completes the statement or answers the question. 1. A line spectrum is produced when an electron moves from one energy
P. Table & E Configuration Practice TEST Multiple Choice Identify the choice that best completes the statement or answers the question. 1. A line spectrum is produced when an electron moves from one energy
Chapter Outline. 3 Elements and Compounds. Elements and Atoms. Elements. Elements. Elements 9/4/2013
 3 Elements and Compounds Chapter Outline 3.1 Elements A. Distribution of Elements Foundations of College Chemistry, 14 th Ed. Morris Hein and Susan Arena Copyright This reclining Buddha in Thailand is
3 Elements and Compounds Chapter Outline 3.1 Elements A. Distribution of Elements Foundations of College Chemistry, 14 th Ed. Morris Hein and Susan Arena Copyright This reclining Buddha in Thailand is
Ionic and Covalent Bonds
 Ionic and Covalent Bonds Ionic Bonds Transfer of Electrons When metals bond with nonmetals, electrons are from the metal to the nonmetal The becomes a cation and the becomes an anion. The between the cation
Ionic and Covalent Bonds Ionic Bonds Transfer of Electrons When metals bond with nonmetals, electrons are from the metal to the nonmetal The becomes a cation and the becomes an anion. The between the cation
Mansa Musa s Pilgrimage Grade Seven
 Ohio Standards Connection: Geography Benchmark A Identify on a map the location of major physical and human features of each continent. Indicator 2 On a map, identify places related to the historical events
Ohio Standards Connection: Geography Benchmark A Identify on a map the location of major physical and human features of each continent. Indicator 2 On a map, identify places related to the historical events
Start: 26e Used: 6e Step 4. Place the remaining valence electrons as lone pairs on the surrounding and central atoms.
 Section 4.1: Types of Chemical Bonds Tutorial 1 Practice, page 200 1. (a) Lewis structure for NBr 3 : Step 1. The central atom for nitrogen tribromide is bromine. 1 N atom: 1(5e ) = 5e 3 Br atoms: 3(7e
Section 4.1: Types of Chemical Bonds Tutorial 1 Practice, page 200 1. (a) Lewis structure for NBr 3 : Step 1. The central atom for nitrogen tribromide is bromine. 1 N atom: 1(5e ) = 5e 3 Br atoms: 3(7e
