Anatomy and Physiology Placement Exam 2 Practice with Answers at End!
|
|
|
- Francine Stevens
- 10 years ago
- Views:
Transcription
1 Anatomy and Physiology Placement Exam 2 Practice with Answers at End! General Chemical Principles 1. bonds are characterized by the sharing of electrons between the participating atoms. a. hydrogen b. ionic c. covalent d. electrical 2. A chemical bond in which electrons are gained or lost is called a(n) bond. a. ionic b. covalent c. hydrogen d. electrical 3. An electron is a(n) a. negatively charged subatomic particle b. molecule with a neutral charge c. neutral atom of an element d. uncharged subatomic particle 4. A subatomic particle possessing no electrical charge is called a(n) a. ion b. neutron c. atom d. electron 5. All isotopes of a particular element possess a. identical atomic weights b. the same number of neutrons, but different numbers of electrons c. the same number of protons, but different numbers of neutrons d. identical atomic weights and numbers 6. An atom or a group of atoms possessing a positive or negative electrical charge is called a(n): a. molecule b. compound c. element d. ion Carbon has an atomic number of 6, but has 8 neutrons. How many electrons are present in each atom? a. 6 b. 8 c. 14 d. some have 6 and some have 8
2 8. Chlorine must gain one electron to fill its outer energy level completely. Potassium must lose one electron in order to possess a completely filled outer energy level. What type of bond would you expect to find in potassium chloride? a. covalent b. ionic c. hydrogen d. it cannot be determined from the information given 9. Atomic number is determined by the number of in each atom. a. protons only b. protons and electrons c. protons and neutrons d. protons, neutrons, and electrons 10. Atoms of the element carbon have four electrons in the outermost shell. What is the maximum number of single covalent bonds that can form between one atom of carbon and other atoms? a. 1 b. 2 c. 3 d. 4 Water, Acids, Bases and ph 11. What determines the ph of a solution? a. the number of ionic bonds available b. the hydrogen ion concentration c. the strength of the hydrogen bonds present d. the number of water molecules available 12. Identify the mismatched pair: a. hydrophilic polar molecules b. hydrogen ion proton c. acid solution ph more than 7 d. water universal solvent 13. Which of the following qualities of water is largely responsible for the phenomenon known as capillary action? a. Water is relatively transparent. b. Water expands slightly as it freezes. c. Water is strongly cohesive. d. Water has a high heat capacity. 14. Which of the following qualities of water is largely responsible for the ability of small insects to walk directly on the surface of water? a. Water is relatively transparent. b. Water displays a measurable surface tension. c. Water expands as it freezes. d. Water demonstrates an observable phase transition. 2
3 15. Which arrow points to a hydrogen bond? 16. Which arrow points to a covalent bond? 17. Among solutions with the following ph values, the one that has the greatest concentration of protons is the one with a ph value of a. 3 b. 7 c. 10 d What is the ph of baking soda? a. 10 b. 9 c. 8 d Which of the following statements is true about a solution with a ph of 9, one with a ph of 2 and one with a ph of 7? a. The ph 2 solution has a higher hydrogen (H + ) ion concentration than the ph 7 solution. b. The ph 2 solution has a higher hydroxyl (OH - ) ion concentration than the ph 7 solution. c. The ph 9 solution has a higher hydrogen (H + ) ion concentration than the ph 2 solution. d. The ph 9 solution has a hydrogen (H + ) ion concentration that is seven times higher than that of the ph 2 solution. 3
4 Organic Molecules 20. Organic chemistry is the chemistry of a. oxygen molecules b. isotopes c. carbon compounds d. ionic bonds 21. Organic molecules contain particular elements. Based on what you know of organic chemistry, find the mismatched pair. a. carbohydrate - CHO b. lipid - CHNOP c. protein - CHNOPS d. nucleic acid - CHNOP 22. A molecule with the formula C 5 H 10 O 5 is a(n) a. carbohydrate b. steroid c. fat d. amino acid 23. Simple sugars are also known as a. polymers b. monosaccharides c. disaccharides d. polysaccharides 24. Which of the following is a condensation (dehydration synthesis) reaction? a. the linking together of two glucose molecules b. the breakdown of a polymer into monomers c. a reactions that consumes a water molecule d. an enzyme reaction that divides water into two subunits 25. Each amino acid has a different a. amino group b. carboxyl group c. R group d. peptide bond 26. A pentose sugar, a phosphate group and a nitrogen-containing base are found in a(n) a. amino acid b. nucleotide c. lipid d. carbohydrate 27. In a theoretical monosaccharide, there are 18 atoms of hydrogen. How many atoms of oxygen are present? a. 6 b. 9 c. 11 d. 22 4
5 28. Amino acids are the building blocks or monomers for a. nucleic acids b. proteins c. carbohydrates d. lipids Cell Structure 29. Aerobic cellular respiration occurs in the a. mitochondria b. ribosomes c. nucleolus d. cytoplasm 30. Detoxification of certain potentially poisonous molecules occurs in the a. peroxisomes b. lysosomes c. rough ER d. Golgi complex 31. The site of protein formation in the cell is the a. mitochondria b. Golgi complex c. lysosomes d. ribosomes 32. This organelle is the command and control center of the cell a. mitochondria b. nucleus c. Golgi complex d. centrioles 33. Identify the mismatched pair a. ribosome synthesis nucleolus b. rough endoplasmic reticulum attached ribosomes c. Golgi apparatus lytic enzymes d. cytoskeleton microtubules 34. Proteins that are secreted outside the cell in the process of exocytosis are released by a. secretory vesicles b. Golgi complex c. vacuoles d. rough ER 35. Cilia and flagella are specialized structures used a. in mitosis b. for locomotion c. to maintain cell shape d. as cellular supports 5
6 36. The mitochondria are organelles that a. convert their DNA into food for the cell b. produce ATP from the chemical energy of food c. produce ATP from the chemical energy of their DNA d. convert the chemical energy of food into DNA 37. Why are ribosomes attached to the endoplasmic reticulum (ER)? a. Newly synthesized polypeptides can move directly through the ER membrane. b. The new protein is more easily released directly into the cytoplasm. c. Ribosomes help prevent the ER from leaking material out of the cell. d. Ribosomes are involved in steroid synthesis and chemical detoxification. Cellular Transport 38. Diffusion is the process by which a substances moves from an area of a. lower temperature to an area of higher temperature b. higher temperature to an area of lower temperature c. lower concentration to an area of higher concentration d. higher concentration to an area of lower concentration 39. A diffusion equilibrium is reached when a. individual molecules stop moving b. molecular motion is not random c. there is no net movement between two areas d. a concentration gradient is established 40. Osmosis is a special case of a. diffusion that involves the movement of water across a membrane b. facilitated diffusion that involves the movement of sodium c. active transport that involves the movement of water d. diffusion that involves the movement of water via the sodium-potassium pump 41. The net movement of water across a selectively permeable membrane is always a. from the hypertonic solution to the hypotonic one b. from the hypotonic solution to the hypertonic one c. from the equilibrium solution to the hypertonic one d. from the equilibrium solution to the isotonic one 42. The direction of osmosis is determined by the a. ph of both solutions b. temperature of the fluid and that of the membrane c. concentration of dissolved solutes on both sides of the membrane. d. temperature of the solutions 43. is the process by which the undigested contents of food vacuoles are removed from the cell. a. Phagocytosis b. Pinocytosis c. Exocytosis d. Vacuolation 6
7 44. A cell placed in a beaker of sea water will a. actively transport water out b. shrink due to the loss of water by osmosis c. swell due to gain of water by osmosis d. be largely unaffected and not changed appreciably in size or appearance 45. A hypothetical microbullet shot through a phospholipid bilayer would pass the components in which order? a. polar >>> nonpolar>>>polar>>>nonpolar b. polar>>>polar>>>nonpolar>>>nonpolar c. nonpolar>>>polar>>>polar>>>nonpolar d. polar>>>nonpolar>>>nonpolar>>>polar Use the following picture of a neuron (nerve cell) to answer questions Na+ = 12 Na+ = The concentration of Na + is outside the cell than inside; therefore, the Na+ will move the cell. a. greater into b. lower out of c. greater out of d. lower into 47. When a neuron is stimulated and Na + ions move through an open Na + channel, the process is a. endocytosis b. osmosis c. facilitated diffusion d. active transport Enzymes and Energy 48. Enzymes are important as a. raw materials for building body parts b. a source of energy for metabolic processes c. hormones d. catalysts for chemical reactions 49. Nearly all of the energy available on the earth comes from e. the core of the planet b. the sun c. volcanoes d. the oceans 50. What is activation energy? a. chemical energy released from any reaction b. the energy required to start a chemical reaction c. energy released from a reaction that does not involve molecular oxygen d. a tremendous amount of energy that is spontaneously generated during a chemical reaction 7
8 51. Enzymes a. frequently violate both the first and second law of thermodynamics b. lower activation energy requirements c. can affect the energy released from a chemical reaction d. slow metabolic reactions 52. The reactants in an enzyme catalyzed reaction are called a. substrates b. products c. complex d. active site 53. The information about the synthesis of enzymes comes from a. ATP b. DNA c. lipid molecules d. specific complex carbohydrates 54. With few exceptions, all enzymes that have been identified are a. high energy phosphate molecules b. nucleic acids c. complex lipids or carbohydrates d. proteins Organization within the Human Body 55. Lungs, heart, or brain are examples of a. systems b. organs c. cell tissues d. organelles 56. Pick the answer that ranks the following from smallest to largest. a. atoms, organs, cells, organisms, organ systems b. atoms, cells, tissues, organs, organisms c. cells, atoms, organ systems, organs, organisms d. organism, organ systems, organs, atoms, cells 57. Circulatory, respiratory, or digestive are examples of a. systems b. organs c. cell tissues d. organelles 8
9 Mass (g) Graphs Use the graph below for questions Volume (ml) 58. An appropriate title for this graph would be a. Volume and mass b. How volume effects mass c. The increasing relationship between volume and mass d. Mass as a function of volume 59. If the volume of a liquid is 40 ml, the mass is a. 25 g b. 37 g c. 40 g d. 45 g 60. The relationship between mass and volume could best be described as a. Increasing mass decreases volume b. Increasing volume results in a decrease of mass c. Increasing volume results in an increase of mass d. There is no relationship between mass and volume ANSWERS: 1. C 2. A 3.A 4.B 5.C 6.D 7.A 8.B 9.A 10.D 11.B 12.C 13.C 14.B 15.C 16.A 17.A 18.B 19.A 20.C 21.B 22.A 23.B 24.A 25.C 26.B 27.B 28.B 29.A 30.A 31.D 32.B 33.C 34.A 35.B 36.B 37.A 38.D 39.C 40.A 41.B 42.C 43.C 44.B 45.D 46.A 47.C 48.D 49.B 50.B 51.B 52.A 53.B 54.D 55.B 56.B 57.A 58.D 59.B 60.C 9
Keystone Review Practice Test Module A Cells and Cell Processes. 1. Which characteristic is shared by all prokaryotes and eukaryotes?
 Keystone Review Practice Test Module A Cells and Cell Processes 1. Which characteristic is shared by all prokaryotes and eukaryotes? a. Ability to store hereditary information b. Use of organelles to control
Keystone Review Practice Test Module A Cells and Cell Processes 1. Which characteristic is shared by all prokaryotes and eukaryotes? a. Ability to store hereditary information b. Use of organelles to control
Date: Student Name: Teacher Name: Jared George. Score: 1) A cell with 1% solute concentration is placed in a beaker with a 5% solute concentration.
 Biology Keystone (PA Core) Quiz Homeostasis and Transport - (BIO.A.4.1.1 ) Plasma Membrane, (BIO.A.4.1.2 ) Transport Mechanisms, (BIO.A.4.1.3 ) Transport Facilitation Student Name: Teacher Name: Jared
Biology Keystone (PA Core) Quiz Homeostasis and Transport - (BIO.A.4.1.1 ) Plasma Membrane, (BIO.A.4.1.2 ) Transport Mechanisms, (BIO.A.4.1.3 ) Transport Facilitation Student Name: Teacher Name: Jared
Review of the Cell and Its Organelles
 Biology Learning Centre Review of the Cell and Its Organelles Tips for most effective learning of this material: Memorize the names and structures over several days. This will help you retain what you
Biology Learning Centre Review of the Cell and Its Organelles Tips for most effective learning of this material: Memorize the names and structures over several days. This will help you retain what you
1. When applying the process of science, which of these is tested? a. an observation b. a result c. a hypothesis d. a question e.
 BCOR 11 Exam 1, 2004 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1. When applying the process of science, which of these is tested? a. an observation
BCOR 11 Exam 1, 2004 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1. When applying the process of science, which of these is tested? a. an observation
Chemical Basis of Life Module A Anchor 2
 Chemical Basis of Life Module A Anchor 2 Key Concepts: - Water is a polar molecule. Therefore, it is able to form multiple hydrogen bonds, which account for many of its special properties. - Water s polarity
Chemical Basis of Life Module A Anchor 2 Key Concepts: - Water is a polar molecule. Therefore, it is able to form multiple hydrogen bonds, which account for many of its special properties. - Water s polarity
Biological cell membranes
 Unit 14: Cell biology. 14 2 Biological cell membranes The cell surface membrane surrounds the cell and acts as a barrier between the cell s contents and the environment. The cell membrane has multiple
Unit 14: Cell biology. 14 2 Biological cell membranes The cell surface membrane surrounds the cell and acts as a barrier between the cell s contents and the environment. The cell membrane has multiple
Name: Hour: Elements & Macromolecules in Organisms
 Name: Hour: Elements & Macromolecules in Organisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds
Name: Hour: Elements & Macromolecules in Organisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds
FIGURE 2.18. A. The phosphate end of the molecule is polar (charged) and hydrophilic (attracted to water).
 PLASMA MEMBRANE 1. The plasma membrane is the outermost part of a cell. 2. The main component of the plasma membrane is phospholipids. FIGURE 2.18 A. The phosphate end of the molecule is polar (charged)
PLASMA MEMBRANE 1. The plasma membrane is the outermost part of a cell. 2. The main component of the plasma membrane is phospholipids. FIGURE 2.18 A. The phosphate end of the molecule is polar (charged)
Modes of Membrane Transport
 Modes of Membrane Transport Transmembrane Transport movement of small substances through a cellular membrane (plasma, ER, mitochondrial..) ions, fatty acids, H 2 O, monosaccharides, steroids, amino acids
Modes of Membrane Transport Transmembrane Transport movement of small substances through a cellular membrane (plasma, ER, mitochondrial..) ions, fatty acids, H 2 O, monosaccharides, steroids, amino acids
Plasma Membrane hydrophilic polar heads
 The Parts of the Cell 3 main parts in ALL cells: plasma membrane, cytoplasm, genetic material this is about the parts of a generic eukaryotic cell Plasma Membrane -is a fluid mosaic model membrane is fluid
The Parts of the Cell 3 main parts in ALL cells: plasma membrane, cytoplasm, genetic material this is about the parts of a generic eukaryotic cell Plasma Membrane -is a fluid mosaic model membrane is fluid
Lecture Overview. Hydrogen Bonds. Special Properties of Water Molecules. Universal Solvent. ph Scale Illustrated. special properties of water
 Lecture Overview special properties of water > water as a solvent > ph molecules of the cell > properties of carbon > carbohydrates > lipids > proteins > nucleic acids Hydrogen Bonds polarity of water
Lecture Overview special properties of water > water as a solvent > ph molecules of the cell > properties of carbon > carbohydrates > lipids > proteins > nucleic acids Hydrogen Bonds polarity of water
Carbon-organic Compounds
 Elements in Cells The living substance of cells is made up of cytoplasm and the structures within it. About 96% of cytoplasm and its included structures are composed of the elements carbon, hydrogen, oxygen,
Elements in Cells The living substance of cells is made up of cytoplasm and the structures within it. About 96% of cytoplasm and its included structures are composed of the elements carbon, hydrogen, oxygen,
1. The diagram below represents a biological process
 1. The diagram below represents a biological process 5. The chart below indicates the elements contained in four different molecules and the number of atoms of each element in those molecules. Which set
1. The diagram below represents a biological process 5. The chart below indicates the elements contained in four different molecules and the number of atoms of each element in those molecules. Which set
Six major functions of membrane proteins: Transport Enzymatic activity
 CH 7 Membranes Cellular Membranes Phospholipids are the most abundant lipid in the plasma membrane. Phospholipids are amphipathic molecules, containing hydrophobic and hydrophilic regions. The fluid mosaic
CH 7 Membranes Cellular Membranes Phospholipids are the most abundant lipid in the plasma membrane. Phospholipids are amphipathic molecules, containing hydrophobic and hydrophilic regions. The fluid mosaic
AP Biology-Chapter #6 & 7 Review
 DO NOT WRITE ON THIS TEST- USE ANSWER DOCUMENT AP Biology-Chapter #6 & 7 Review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. All of the following are
DO NOT WRITE ON THIS TEST- USE ANSWER DOCUMENT AP Biology-Chapter #6 & 7 Review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. All of the following are
Osmosis, Diffusion and Cell Transport
 Osmosis, Diffusion and Cell Transport Types of Transport There are 3 types of transport in cells: 1. Passive Transport: does not use the cell s energy in bringing materials in & out of the cell 2. Active
Osmosis, Diffusion and Cell Transport Types of Transport There are 3 types of transport in cells: 1. Passive Transport: does not use the cell s energy in bringing materials in & out of the cell 2. Active
Homeostasis and Transport Module A Anchor 4
 Homeostasis and Transport Module A Anchor 4 Key Concepts: - Buffers play an important role in maintaining homeostasis in organisms. - To maintain homeostasis, unicellular organisms grow, respond to the
Homeostasis and Transport Module A Anchor 4 Key Concepts: - Buffers play an important role in maintaining homeostasis in organisms. - To maintain homeostasis, unicellular organisms grow, respond to the
Cell Structure & Function!
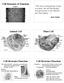 Cell Structure & Function! Chapter 3! The most exciting phrase to hear in science, the one that heralds new discoveries, is not 'Eureka!' but 'That's funny.! -- Isaac Asimov Animal Cell Plant Cell Cell
Cell Structure & Function! Chapter 3! The most exciting phrase to hear in science, the one that heralds new discoveries, is not 'Eureka!' but 'That's funny.! -- Isaac Asimov Animal Cell Plant Cell Cell
Chapter 4: A Tour of the Cell. 1. Cell Basics. Limits to Cell Size. 1. Cell Basics. 2. Prokaryotic Cells. 3. Eukaryotic Cells
 Chapter 4: A Tour of the Cell 1. Cell Basics 2. Prokaryotic Cells 3. Eukaryotic Cells 1. Cell Basics Limits to Cell Size There are 2 main reasons why cells are so small: If cells get too large: 1) there
Chapter 4: A Tour of the Cell 1. Cell Basics 2. Prokaryotic Cells 3. Eukaryotic Cells 1. Cell Basics Limits to Cell Size There are 2 main reasons why cells are so small: If cells get too large: 1) there
tissues are made of cells that work together, organs are )
 Study Guide Cells Unit Test Matching. Write the letter of the correct response on the line. You may use the responses more than once. A. proteins B. simple carbohydrates C. complex carbohydrates D. lipids
Study Guide Cells Unit Test Matching. Write the letter of the correct response on the line. You may use the responses more than once. A. proteins B. simple carbohydrates C. complex carbohydrates D. lipids
Membrane Structure and Function
 Membrane Structure and Function -plasma membrane acts as a barrier between cells and the surrounding. -plasma membrane is selective permeable -consist of lipids, proteins and carbohydrates -major lipids
Membrane Structure and Function -plasma membrane acts as a barrier between cells and the surrounding. -plasma membrane is selective permeable -consist of lipids, proteins and carbohydrates -major lipids
Membrane Structure and Function
 Membrane Structure and Function Part A Multiple Choice 1. The fluid mosaic model describes membranes as having A. a set of protein channels separated by phospholipids. B. a bilayer of phospholipids in
Membrane Structure and Function Part A Multiple Choice 1. The fluid mosaic model describes membranes as having A. a set of protein channels separated by phospholipids. B. a bilayer of phospholipids in
Ch. 8 - The Cell Membrane
 Ch. 8 - The Cell Membrane 2007-2008 Phospholipids Phosphate head hydrophilic Fatty acid tails hydrophobic Arranged as a bilayer Phosphate attracted to water Fatty acid repelled by water Aaaah, one of those
Ch. 8 - The Cell Membrane 2007-2008 Phospholipids Phosphate head hydrophilic Fatty acid tails hydrophobic Arranged as a bilayer Phosphate attracted to water Fatty acid repelled by water Aaaah, one of those
Cellular Structure and Function
 Chapter Test A CHAPTER 7 Cellular Structure and Function Part A: Multiple Choice In the space at the left, write the letter of the term or phrase that best answers each question. 1. Which defines a cell?
Chapter Test A CHAPTER 7 Cellular Structure and Function Part A: Multiple Choice In the space at the left, write the letter of the term or phrase that best answers each question. 1. Which defines a cell?
Biology I. Chapter 7
 Biology I Chapter 7 Interest Grabber NOTEBOOK #1 Are All Cells Alike? All living things are made up of cells. Some organisms are composed of only one cell. Other organisms are made up of many cells. 1.
Biology I Chapter 7 Interest Grabber NOTEBOOK #1 Are All Cells Alike? All living things are made up of cells. Some organisms are composed of only one cell. Other organisms are made up of many cells. 1.
Cells & Cell Organelles
 Cells & Cell Organelles The Building Blocks of Life H Biology Types of cells bacteria cells Prokaryote - no organelles Eukaryotes - organelles animal cells plant cells Cell size comparison Animal cell
Cells & Cell Organelles The Building Blocks of Life H Biology Types of cells bacteria cells Prokaryote - no organelles Eukaryotes - organelles animal cells plant cells Cell size comparison Animal cell
THE HISTORY OF CELL BIOLOGY
 SECTION 4-1 REVIEW THE HISTORY OF CELL BIOLOGY Define the following terms. 1. cell 2. cell theory Write the correct letter in the blank. 1. One early piece of evidence supporting the cell theory was the
SECTION 4-1 REVIEW THE HISTORY OF CELL BIOLOGY Define the following terms. 1. cell 2. cell theory Write the correct letter in the blank. 1. One early piece of evidence supporting the cell theory was the
7. A selectively permeable membrane only allows certain molecules to pass through.
 CHAPTER 2 GETTING IN & OUT OF CELLS PASSIVE TRANSPORT Cell membranes help organisms maintain homeostasis by controlling what substances may enter or leave cells. Some substances can cross the cell membrane
CHAPTER 2 GETTING IN & OUT OF CELLS PASSIVE TRANSPORT Cell membranes help organisms maintain homeostasis by controlling what substances may enter or leave cells. Some substances can cross the cell membrane
Biochemistry of Cells
 Biochemistry of Cells 1 Carbon-based Molecules Although a cell is mostly water, the rest of the cell consists mostly of carbon-based molecules Organic chemistry is the study of carbon compounds Carbon
Biochemistry of Cells 1 Carbon-based Molecules Although a cell is mostly water, the rest of the cell consists mostly of carbon-based molecules Organic chemistry is the study of carbon compounds Carbon
Biology Chapter 7 Practice Test
 Biology Chapter 7 Practice Test Multiple Choice Write the letter that best answers the question or completes the statement on the line provided. 1. The work of Schleiden and Schwann can be summarized by
Biology Chapter 7 Practice Test Multiple Choice Write the letter that best answers the question or completes the statement on the line provided. 1. The work of Schleiden and Schwann can be summarized by
Carbohydrates, proteins and lipids
 Carbohydrates, proteins and lipids Chapter 3 MACROMOLECULES Macromolecules: polymers with molecular weights >1,000 Functional groups THE FOUR MACROMOLECULES IN LIFE Molecules in living organisms: proteins,
Carbohydrates, proteins and lipids Chapter 3 MACROMOLECULES Macromolecules: polymers with molecular weights >1,000 Functional groups THE FOUR MACROMOLECULES IN LIFE Molecules in living organisms: proteins,
The Molecules of Life - Overview. The Molecules of Life. The Molecules of Life. The Molecules of Life
 The Molecules of Life - Overview The Molecules of Life The Importance of Carbon Organic Polymers / Monomers Functions of Organic Molecules Origin of Organic Molecules The Molecules of Life Water is the
The Molecules of Life - Overview The Molecules of Life The Importance of Carbon Organic Polymers / Monomers Functions of Organic Molecules Origin of Organic Molecules The Molecules of Life Water is the
Chapter 2 Chemical Principles
 Chapter 2 Chemical Principles I. Chemistry. [Students should read this section on their own]. a. Chemistry is the study of the interactions between atoms and molecules. b. The atom is the smallest unit
Chapter 2 Chemical Principles I. Chemistry. [Students should read this section on their own]. a. Chemistry is the study of the interactions between atoms and molecules. b. The atom is the smallest unit
PRESTWICK ACADEMY NATIONAL 5 BIOLOGY CELL BIOLOGY SUMMARY
 Name PRESTWICK ACADEMY NATIONAL 5 BIOLOGY CELL BIOLOGY SUMMARY Cell Structure Identify animal, plant, fungal and bacterial cell ultrastructure and know the structures functions. Plant cell Animal cell
Name PRESTWICK ACADEMY NATIONAL 5 BIOLOGY CELL BIOLOGY SUMMARY Cell Structure Identify animal, plant, fungal and bacterial cell ultrastructure and know the structures functions. Plant cell Animal cell
2007 7.013 Problem Set 1 KEY
 2007 7.013 Problem Set 1 KEY Due before 5 PM on FRIDAY, February 16, 2007. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. Where in a eukaryotic cell do you
2007 7.013 Problem Set 1 KEY Due before 5 PM on FRIDAY, February 16, 2007. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. Where in a eukaryotic cell do you
4. Biology of the Cell
 4. Biology of the Cell Our primary focus in this chapter will be the plasma membrane and movement of materials across the plasma membrane. You should already be familiar with the basic structures and roles
4. Biology of the Cell Our primary focus in this chapter will be the plasma membrane and movement of materials across the plasma membrane. You should already be familiar with the basic structures and roles
Elements & Macromolecules in Organisms
 Name: Date: Per: Table # Elements & Macromolecules in rganisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight.
Name: Date: Per: Table # Elements & Macromolecules in rganisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight.
1.1.2. thebiotutor. AS Biology OCR. Unit F211: Cells, Exchange & Transport. Module 1.2 Cell Membranes. Notes & Questions.
 thebiotutor AS Biology OCR Unit F211: Cells, Exchange & Transport Module 1.2 Cell Membranes Notes & Questions Andy Todd 1 Outline the roles of membranes within cells and at the surface of cells. The main
thebiotutor AS Biology OCR Unit F211: Cells, Exchange & Transport Module 1.2 Cell Membranes Notes & Questions Andy Todd 1 Outline the roles of membranes within cells and at the surface of cells. The main
A disaccharide is formed when a dehydration reaction joins two monosaccharides. This covalent bond is called a glycosidic linkage.
 CH 5 Structure & Function of Large Molecules: Macromolecules Molecules of Life All living things are made up of four classes of large biological molecules: carbohydrates, lipids, proteins, and nucleic
CH 5 Structure & Function of Large Molecules: Macromolecules Molecules of Life All living things are made up of four classes of large biological molecules: carbohydrates, lipids, proteins, and nucleic
Chapter 2: Cell Structure and Function pg. 70-107
 UNIT 1: Biochemistry Chapter 2: Cell Structure and Function pg. 70-107 Organelles are internal structures that carry out specialized functions, interacting and complementing each other. Animal and plant
UNIT 1: Biochemistry Chapter 2: Cell Structure and Function pg. 70-107 Organelles are internal structures that carry out specialized functions, interacting and complementing each other. Animal and plant
Examination One. Biology 101. Dr. Jaeson T. Fournier
 Examination One Biology 101 Dr. Jaeson T. Fournier Examination Instructions: Answers are to be indicated on a scantron. Keep your work protected! This helps prevent dishonesty. The instructor will not
Examination One Biology 101 Dr. Jaeson T. Fournier Examination Instructions: Answers are to be indicated on a scantron. Keep your work protected! This helps prevent dishonesty. The instructor will not
Disaccharides consist of two monosaccharide monomers covalently linked by a glycosidic bond. They function in sugar transport.
 1. The fundamental life processes of plants and animals depend on a variety of chemical reactions that occur in specialized areas of the organism s cells. As a basis for understanding this concept: 1.
1. The fundamental life processes of plants and animals depend on a variety of chemical reactions that occur in specialized areas of the organism s cells. As a basis for understanding this concept: 1.
Introduction to the Cell: Plant and Animal Cells
 Introduction to the Cell: Plant and Animal Cells Tissues, Organs, and Systems of Living Things Cells, Cell Division, and Animal Systems and Plant Systems Cell Specialization Human Systems All organisms
Introduction to the Cell: Plant and Animal Cells Tissues, Organs, and Systems of Living Things Cells, Cell Division, and Animal Systems and Plant Systems Cell Specialization Human Systems All organisms
Todays Outline. Metabolism. Why do cells need energy? How do cells acquire energy? Metabolism. Concepts & Processes. The cells capacity to:
 and Work Metabolic Pathways Enzymes Features Factors Affecting Enzyme Activity Membrane Transport Diffusion Osmosis Passive Transport Active Transport Bulk Transport Todays Outline -Releasing Pathways
and Work Metabolic Pathways Enzymes Features Factors Affecting Enzyme Activity Membrane Transport Diffusion Osmosis Passive Transport Active Transport Bulk Transport Todays Outline -Releasing Pathways
How To Understand The Chemistry Of Organic Molecules
 CHAPTER 3 THE CHEMISTRY OF ORGANIC MOLECULES 3.1 Organic Molecules The chemistry of carbon accounts for the diversity of organic molecules found in living things. Carbon has six electrons, four of which
CHAPTER 3 THE CHEMISTRY OF ORGANIC MOLECULES 3.1 Organic Molecules The chemistry of carbon accounts for the diversity of organic molecules found in living things. Carbon has six electrons, four of which
Chapter 5. The Structure and Function of Macromolecule s
 Chapter 5 The Structure and Function of Macromolecule s Most Macromolecules are polymers: Polymer: (poly: many; mer: part) Large molecules consisting of many identical or similar subunits connected together.
Chapter 5 The Structure and Function of Macromolecule s Most Macromolecules are polymers: Polymer: (poly: many; mer: part) Large molecules consisting of many identical or similar subunits connected together.
Comparing Plant And Animal Cells
 Comparing Plant And Animal Cells http://khanacademy.org/video?v=hmwvj9x4gny Plant Cells shape - most plant cells are squarish or rectangular in shape. amyloplast (starch storage organelle)- an organelle
Comparing Plant And Animal Cells http://khanacademy.org/video?v=hmwvj9x4gny Plant Cells shape - most plant cells are squarish or rectangular in shape. amyloplast (starch storage organelle)- an organelle
PART I: Neurons and the Nerve Impulse
 PART I: Neurons and the Nerve Impulse Identify each of the labeled structures of the neuron below. A. B. C. D. E. F. G. Identify each of the labeled structures of the neuron below. A. dendrites B. nucleus
PART I: Neurons and the Nerve Impulse Identify each of the labeled structures of the neuron below. A. B. C. D. E. F. G. Identify each of the labeled structures of the neuron below. A. dendrites B. nucleus
Chapter 8. Movement across the Cell Membrane. AP Biology
 Chapter 8. Movement across the Cell Membrane More than just a barrier Expanding our view of cell membrane beyond just a phospholipid bilayer barrier phospholipids plus Fluid Mosaic Model In 1972, S.J.
Chapter 8. Movement across the Cell Membrane More than just a barrier Expanding our view of cell membrane beyond just a phospholipid bilayer barrier phospholipids plus Fluid Mosaic Model In 1972, S.J.
Chapter 2. The Chemistry of Life Worksheets
 Chapter 2 The Chemistry of Life Worksheets (Opening image courtesy of David Iberri, http://en.wikipedia.org/wiki/file:camkii.png, and under the Creative Commons license CC-BY-SA 3.0.) Lesson 2.1: Matter
Chapter 2 The Chemistry of Life Worksheets (Opening image courtesy of David Iberri, http://en.wikipedia.org/wiki/file:camkii.png, and under the Creative Commons license CC-BY-SA 3.0.) Lesson 2.1: Matter
Cell and Membrane Practice. A. chromosome B. gene C. mitochondrion D. vacuole
 Name: ate: 1. Which structure is outside the nucleus of a cell and contains N?. chromosome. gene. mitochondrion. vacuole 2. potato core was placed in a beaker of water as shown in the figure below. Which
Name: ate: 1. Which structure is outside the nucleus of a cell and contains N?. chromosome. gene. mitochondrion. vacuole 2. potato core was placed in a beaker of water as shown in the figure below. Which
Section 7-3 Cell Boundaries
 Note: For the past several years, I ve been puzzling how to integrate new discoveries on the nature of water movement through cell membranes into Chapter 7. The Section below is a draft of my first efforts
Note: For the past several years, I ve been puzzling how to integrate new discoveries on the nature of water movement through cell membranes into Chapter 7. The Section below is a draft of my first efforts
Cell Membrane Structure (and How to Get Through One)
 Cell Membrane Structure (and How to Get Through One) A cell s membrane is a wall of sorts that defines the boundaries of a cell. The membrane provides protection and structure for the cell and acts as
Cell Membrane Structure (and How to Get Through One) A cell s membrane is a wall of sorts that defines the boundaries of a cell. The membrane provides protection and structure for the cell and acts as
10.1 The function of Digestion pg. 402
 10.1 The function of Digestion pg. 402 Macromolecules and Living Systems The body is made up of more than 60 % water. The water is found in the cells cytoplasm, the interstitial fluid and the blood (5
10.1 The function of Digestion pg. 402 Macromolecules and Living Systems The body is made up of more than 60 % water. The water is found in the cells cytoplasm, the interstitial fluid and the blood (5
Chapter 3. Cellular Structure and Function Worksheets. 39 www.ck12.org
 Chapter 3 Cellular Structure and Function Worksheets (Opening image copyright by Sebastian Kaulitzki, 2010. Used under license from Shutterstock.com.) Lesson 3.1: Introduction to Cells Lesson 3.2: Cell
Chapter 3 Cellular Structure and Function Worksheets (Opening image copyright by Sebastian Kaulitzki, 2010. Used under license from Shutterstock.com.) Lesson 3.1: Introduction to Cells Lesson 3.2: Cell
ATOMS AND BONDS. Bonds
 ATOMS AND BONDS Atoms of elements are the simplest units of organization in the natural world. Atoms consist of protons (positive charge), neutrons (neutral charge) and electrons (negative charge). The
ATOMS AND BONDS Atoms of elements are the simplest units of organization in the natural world. Atoms consist of protons (positive charge), neutrons (neutral charge) and electrons (negative charge). The
Cell Biology - Part 2 Membranes
 Cell Biology - Part 2 Membranes The organization of cells is made possible by membranes. Membranes isolate, partition, and compartmentalize cells. 1 Membranes isolate the inside of the cell from the outside
Cell Biology - Part 2 Membranes The organization of cells is made possible by membranes. Membranes isolate, partition, and compartmentalize cells. 1 Membranes isolate the inside of the cell from the outside
MCAS Biology. Review Packet
 MCAS Biology Review Packet 1 Name Class Date 1. Define organic. THE CHEMISTRY OF LIFE 2. All living things are made up of 6 essential elements: SPONCH. Name the six elements of life. S N P C O H 3. Elements
MCAS Biology Review Packet 1 Name Class Date 1. Define organic. THE CHEMISTRY OF LIFE 2. All living things are made up of 6 essential elements: SPONCH. Name the six elements of life. S N P C O H 3. Elements
Lecture 4 Cell Membranes & Organelles
 Lecture 4 Cell Membranes & Organelles Structure of Animal Cells The Phospholipid Structure Phospholipid structure Encases all living cells Its basic structure is represented by the fluidmosaic model Phospholipid
Lecture 4 Cell Membranes & Organelles Structure of Animal Cells The Phospholipid Structure Phospholipid structure Encases all living cells Its basic structure is represented by the fluidmosaic model Phospholipid
Chapter 3 Molecules of Cells
 Bio 100 Molecules of cells 1 Chapter 3 Molecules of Cells Compounds containing carbon are called organic compounds Molecules such as methane that are only composed of carbon and hydrogen are called hydrocarbons
Bio 100 Molecules of cells 1 Chapter 3 Molecules of Cells Compounds containing carbon are called organic compounds Molecules such as methane that are only composed of carbon and hydrogen are called hydrocarbons
RAD 223. Radiography physiology. Lecture Notes. First lecture: Cell and Tissue
 RAD 223 Radiography physiology Lecture Notes First lecture: Cell and Tissue Physiology: the word physiology derived from a Greek word for study of nature. It is the study of how the body and its part work
RAD 223 Radiography physiology Lecture Notes First lecture: Cell and Tissue Physiology: the word physiology derived from a Greek word for study of nature. It is the study of how the body and its part work
Cell Structure and Function. Eukaryotic Cell: Neuron
 Cell Structure and Function Eukaryotic Cell: Neuron Cell Structure and Function Eukaryotic Cells: Blood Cells Cell Structure and Function Prokaryotic Cells: Bacteria Cell Structure and Function All living
Cell Structure and Function Eukaryotic Cell: Neuron Cell Structure and Function Eukaryotic Cells: Blood Cells Cell Structure and Function Prokaryotic Cells: Bacteria Cell Structure and Function All living
CELLS: PLANT CELLS 20 FEBRUARY 2013
 CELLS: PLANT CELLS 20 FEBRUARY 2013 Lesson Description In this lesson we will discuss the following: The Cell Theory Terminology Parts of Plant Cells: Organelles Difference between plant and animal cells
CELLS: PLANT CELLS 20 FEBRUARY 2013 Lesson Description In this lesson we will discuss the following: The Cell Theory Terminology Parts of Plant Cells: Organelles Difference between plant and animal cells
List the 3 main types of subatomic particles and indicate the mass and electrical charge of each.
 Basic Chemistry Why do we study chemistry in a biology course? All living organisms are composed of chemicals. To understand life, we must understand the structure, function, and properties of the chemicals
Basic Chemistry Why do we study chemistry in a biology course? All living organisms are composed of chemicals. To understand life, we must understand the structure, function, and properties of the chemicals
Cell Unit Practice Test #1
 ell Unit Practice Test #1 Name: ate: 1. Which organelle is primarily concerned with the conversion of potential energy of organic compounds into suitable form for immediate use by the cell?. mitochondria.
ell Unit Practice Test #1 Name: ate: 1. Which organelle is primarily concerned with the conversion of potential energy of organic compounds into suitable form for immediate use by the cell?. mitochondria.
Lesson Aim To explain the human body at a microscopic level, including the structure and function of cells, tissues and membranes.
 LESSON 1. CELLS & TISSUES Lesson Aim To explain the human body at a microscopic level, including the structure and function of cells, tissues and membranes. THE CELL All living matter is composed of functional
LESSON 1. CELLS & TISSUES Lesson Aim To explain the human body at a microscopic level, including the structure and function of cells, tissues and membranes. THE CELL All living matter is composed of functional
Lab 3 Organic Molecules of Biological Importance
 Name Biology 3 ID Number Lab 3 Organic Molecules of Biological Importance Section 1 - Organic Molecules Section 2 - Functional Groups Section 3 - From Building Blocks to Macromolecules Section 4 - Carbohydrates
Name Biology 3 ID Number Lab 3 Organic Molecules of Biological Importance Section 1 - Organic Molecules Section 2 - Functional Groups Section 3 - From Building Blocks to Macromolecules Section 4 - Carbohydrates
Chapter 5: The Structure and Function of Large Biological Molecules
 Name Period Concept 5.1 Macromolecules are polymers, built from monomers 1. The large molecules of all living things fall into just four main classes. Name them. 2. Circle the three classes that are called
Name Period Concept 5.1 Macromolecules are polymers, built from monomers 1. The large molecules of all living things fall into just four main classes. Name them. 2. Circle the three classes that are called
Biology I New Summit School High School Diploma Program
 Course Description: Biology I Syllabus Biology I is a two semester course. Students completing this course will earn 1.0 unit upon completion. Required Materials: 1. Student Text: Biology-Principles and
Course Description: Biology I Syllabus Biology I is a two semester course. Students completing this course will earn 1.0 unit upon completion. Required Materials: 1. Student Text: Biology-Principles and
How To Understand The Human Body
 Introduction to Biology and Chemistry Outline I. Introduction to biology A. Definition of biology - Biology is the study of life. B. Characteristics of Life 1. Form and size are characteristic. e.g. A
Introduction to Biology and Chemistry Outline I. Introduction to biology A. Definition of biology - Biology is the study of life. B. Characteristics of Life 1. Form and size are characteristic. e.g. A
Organic Compounds. Essential Questions: What is Organic? What are the 4 major Organic Compounds? How are they made? What are they used for?
 Organic Compounds Essential Questions: What is Organic? What are the 4 major Organic Compounds? How are they made? What are they used for? Aristotle: Francesco Redi: What do we already know? Spontaneous
Organic Compounds Essential Questions: What is Organic? What are the 4 major Organic Compounds? How are they made? What are they used for? Aristotle: Francesco Redi: What do we already know? Spontaneous
pathway that involves taking in heat from the environment at each step. C.
 Study Island Cell Energy Keystone Review 1. Cells obtain energy by either capturing light energy through photosynthesis or by breaking down carbohydrates through cellular respiration. In both photosynthesis
Study Island Cell Energy Keystone Review 1. Cells obtain energy by either capturing light energy through photosynthesis or by breaking down carbohydrates through cellular respiration. In both photosynthesis
7.2 Cell Structure. Lesson Objectives. Lesson Summary. Cell Organization Eukaryotic cells contain a nucleus and many specialized structures.
 7.2 Cell Structure Lesson Objectives Describe the structure and function of the cell nucleus. Describe the role of vacuoles, lysosomes, and the cytoskeleton. Identify the role of ribosomes, endoplasmic
7.2 Cell Structure Lesson Objectives Describe the structure and function of the cell nucleus. Describe the role of vacuoles, lysosomes, and the cytoskeleton. Identify the role of ribosomes, endoplasmic
The correct answer is d C. Answer c is incorrect. Reliance on the energy produced by others is a characteristic of heterotrophs.
 1. An autotroph is an organism that a. extracts energy from organic sources b. converts energy from sunlight into chemical energy c. relies on the energy produced by other organisms as an energy source
1. An autotroph is an organism that a. extracts energy from organic sources b. converts energy from sunlight into chemical energy c. relies on the energy produced by other organisms as an energy source
CHAPTER 5.1 5.2: Plasma Membrane Structure
 CHAPTER 5.1 5.2: Plasma Membrane Structure 1. Describe the structure of a phospholipid molecule. Be sure to describe their behavior in relationship to water. 2. What happens when a collection of phospholipids
CHAPTER 5.1 5.2: Plasma Membrane Structure 1. Describe the structure of a phospholipid molecule. Be sure to describe their behavior in relationship to water. 2. What happens when a collection of phospholipids
Cell Structure and Function
 Bio 100 - Cells 1 Cell Structure and Function Tenets of Cell Theory 1. All living things are made up of one or more cells 2. Cells are the basic living units within organisms, and the chemical reactions
Bio 100 - Cells 1 Cell Structure and Function Tenets of Cell Theory 1. All living things are made up of one or more cells 2. Cells are the basic living units within organisms, and the chemical reactions
Investigating cells. Cells are the basic units of living things (this means that all living things are made up of one or more cells).
 SG Biology Summary notes Investigating cells Sub-topic a: Investigating living cells Cells are the basic units of living things (this means that all living things are made up of one or more cells). Cells
SG Biology Summary notes Investigating cells Sub-topic a: Investigating living cells Cells are the basic units of living things (this means that all living things are made up of one or more cells). Cells
-1- BIOS 100 - Fall, 2009 Exam I, 18 Sept, 2008 Michael Muller, Instructor
 BIOS 100 - Fall, 2009 Exam I, 18 Sept, 2008 Michael Muller, Instructor Name: TA: This exam consists of 42 questions over 7 pages (the last page of which has the periodic table and Bloom s hierarchy). Please
BIOS 100 - Fall, 2009 Exam I, 18 Sept, 2008 Michael Muller, Instructor Name: TA: This exam consists of 42 questions over 7 pages (the last page of which has the periodic table and Bloom s hierarchy). Please
Organelles and Their Functions
 Organelles and Their Functions The study of cell organelles and their functions is a fascinating part of biology. The current article provides a brief description of the structure of organelles and their
Organelles and Their Functions The study of cell organelles and their functions is a fascinating part of biology. The current article provides a brief description of the structure of organelles and their
Cells. Structure, Function and Homeostasis
 Cells Structure, Function and Homeostasis Characteristics of Cells Basic unit of life anything alive is made of cells Plasma membrane (skin) that separates them from the environment. Skeletonsfor protection
Cells Structure, Function and Homeostasis Characteristics of Cells Basic unit of life anything alive is made of cells Plasma membrane (skin) that separates them from the environment. Skeletonsfor protection
COMPARISON OF PLANT AND ANIMAL CELLS SIMILARITIES IN PLANT & ANIMAL CELLS
 COMPARISON OF PLANT AND ANIMAL CELLS Cells vary widely in structure and function, even within the same organism. The human body, for example, has more than 200 different types of cells, each with a specialized
COMPARISON OF PLANT AND ANIMAL CELLS Cells vary widely in structure and function, even within the same organism. The human body, for example, has more than 200 different types of cells, each with a specialized
Transmembrane proteins span the bilayer. α-helix transmembrane domain. Multiple transmembrane helices in one polypeptide
 Transmembrane proteins span the bilayer α-helix transmembrane domain Hydrophobic R groups of a.a. interact with fatty acid chains Multiple transmembrane helices in one polypeptide Polar a.a. Hydrophilic
Transmembrane proteins span the bilayer α-helix transmembrane domain Hydrophobic R groups of a.a. interact with fatty acid chains Multiple transmembrane helices in one polypeptide Polar a.a. Hydrophilic
Biological molecules:
 Biological molecules: All are organic (based on carbon). Monomers vs. polymers: Monomers refer to the subunits that, when polymerized, make up a larger polymer. Monomers may function on their own in some
Biological molecules: All are organic (based on carbon). Monomers vs. polymers: Monomers refer to the subunits that, when polymerized, make up a larger polymer. Monomers may function on their own in some
Proteins and Nucleic Acids
 Proteins and Nucleic Acids Chapter 5 Macromolecules: Proteins Proteins Most structurally & functionally diverse group of biomolecules. : o Involved in almost everything o Enzymes o Structure (keratin,
Proteins and Nucleic Acids Chapter 5 Macromolecules: Proteins Proteins Most structurally & functionally diverse group of biomolecules. : o Involved in almost everything o Enzymes o Structure (keratin,
The Molecules of Cells
 The Molecules of Cells I. Introduction A. Most of the world s population cannot digest milk-based foods. 1. These people are lactose intolerant because they lack the enzyme lactase. 2. This illustrates
The Molecules of Cells I. Introduction A. Most of the world s population cannot digest milk-based foods. 1. These people are lactose intolerant because they lack the enzyme lactase. 2. This illustrates
Carbon Hydrogen Oxygen Nitrogen
 Concept 1 - Thinking Practice 1. If the following molecules were to undergo a dehydration synthesis reaction, what molecules would result? Circle the parts of each amino acid that will interact and draw
Concept 1 - Thinking Practice 1. If the following molecules were to undergo a dehydration synthesis reaction, what molecules would result? Circle the parts of each amino acid that will interact and draw
Organic Molecules of Life - Exercise 2
 Organic Molecules of Life - Exercise 2 Objectives -Know the difference between a reducing sugar and a non-reducing sugar. -Distinguish Monosaccharides from Disaccharides and Polysaccharides -Understand
Organic Molecules of Life - Exercise 2 Objectives -Know the difference between a reducing sugar and a non-reducing sugar. -Distinguish Monosaccharides from Disaccharides and Polysaccharides -Understand
BIOLOGICAL MOLECULES OF LIFE
 BIOLOGICAL MOLECULES OF LIFE C A R B O H Y D R A T E S, L I P I D S, P R O T E I N S, A N D N U C L E I C A C I D S The Academic Support Center @ Daytona State College (Science 115, Page 1 of 29) Carbon
BIOLOGICAL MOLECULES OF LIFE C A R B O H Y D R A T E S, L I P I D S, P R O T E I N S, A N D N U C L E I C A C I D S The Academic Support Center @ Daytona State College (Science 115, Page 1 of 29) Carbon
3120-1 - Page 1. Name:
 Name: 1) Which series is arranged in correct order according to decreasing size of structures? A) DNA, nucleus, chromosome, nucleotide, nitrogenous base B) chromosome, nucleus, nitrogenous base, nucleotide,
Name: 1) Which series is arranged in correct order according to decreasing size of structures? A) DNA, nucleus, chromosome, nucleotide, nitrogenous base B) chromosome, nucleus, nitrogenous base, nucleotide,
7.2 Cells: A Look Inside
 CHAPTER 7 CELL STRUCTURE AND FUNCTION 7.2 Cells: A Look Inside Imagine a factory that makes thousands of cookies a day. Ingredients come into the factory, get mixed and baked, then the cookies are packaged.
CHAPTER 7 CELL STRUCTURE AND FUNCTION 7.2 Cells: A Look Inside Imagine a factory that makes thousands of cookies a day. Ingredients come into the factory, get mixed and baked, then the cookies are packaged.
NO CALCULATORS OR CELL PHONES ALLOWED
 Biol 205 Exam 1 TEST FORM A Spring 2008 NAME Fill out both sides of the Scantron Sheet. On Side 2 be sure to indicate that you have TEST FORM A The answers to Part I should be placed on the SCANTRON SHEET.
Biol 205 Exam 1 TEST FORM A Spring 2008 NAME Fill out both sides of the Scantron Sheet. On Side 2 be sure to indicate that you have TEST FORM A The answers to Part I should be placed on the SCANTRON SHEET.
The Structure and Function of Macromolecules: Carbohydrates, Lipids & Phospholipids
 The Structure and Function of Macromolecules: Carbohydrates, Lipids & Phospholipids The FOUR Classes of Large Biomolecules All living things are made up of four classes of large biological molecules: Carbohydrates
The Structure and Function of Macromolecules: Carbohydrates, Lipids & Phospholipids The FOUR Classes of Large Biomolecules All living things are made up of four classes of large biological molecules: Carbohydrates
Molecular Models in Biology
 Molecular Models in Biology Objectives: After this lab a student will be able to: 1) Understand the properties of atoms that give rise to bonds. 2) Understand how and why atoms form ions. 3) Model covalent,
Molecular Models in Biology Objectives: After this lab a student will be able to: 1) Understand the properties of atoms that give rise to bonds. 2) Understand how and why atoms form ions. 3) Model covalent,
CELLS An Introduction to Cell Structure & Function
 CELLS An Introduction to Cell Structure & Function Introduction: In this lab exercise we will be studying three general aspects of cellular structure and function. First, we will observe the anatomical
CELLS An Introduction to Cell Structure & Function Introduction: In this lab exercise we will be studying three general aspects of cellular structure and function. First, we will observe the anatomical
Enzymes: Practice Questions #1
 Enzymes: Practice Questions #1 1. Compound X increases the rate of the reaction below. Compound X is most likely A. an enzyme B. a lipid molecule C. an indicator D. an ADP molecule 2. The equation below
Enzymes: Practice Questions #1 1. Compound X increases the rate of the reaction below. Compound X is most likely A. an enzyme B. a lipid molecule C. an indicator D. an ADP molecule 2. The equation below
Unit 2: Cells, Membranes and Signaling CELL MEMBRANE. Chapter 5 Hillis Textbook
 Unit 2: Cells, Membranes and Signaling CELL MEMBRANE Chapter 5 Hillis Textbook HOW DOES THE LAB RELATE TO THE NEXT CHAPTER? SURFACE AREA: the entire outer covering of a cell that enables materials pass.
Unit 2: Cells, Membranes and Signaling CELL MEMBRANE Chapter 5 Hillis Textbook HOW DOES THE LAB RELATE TO THE NEXT CHAPTER? SURFACE AREA: the entire outer covering of a cell that enables materials pass.
Name Date Period. 2. When a molecule of double-stranded DNA undergoes replication, it results in
 DNA, RNA, Protein Synthesis Keystone 1. During the process shown above, the two strands of one DNA molecule are unwound. Then, DNA polymerases add complementary nucleotides to each strand which results
DNA, RNA, Protein Synthesis Keystone 1. During the process shown above, the two strands of one DNA molecule are unwound. Then, DNA polymerases add complementary nucleotides to each strand which results
4. Which carbohydrate would you find as part of a molecule of RNA? a. Galactose b. Deoxyribose c. Ribose d. Glucose
 1. How is a polymer formed from multiple monomers? a. From the growth of the chain of carbon atoms b. By the removal of an OH group and a hydrogen atom c. By the addition of an OH group and a hydrogen
1. How is a polymer formed from multiple monomers? a. From the growth of the chain of carbon atoms b. By the removal of an OH group and a hydrogen atom c. By the addition of an OH group and a hydrogen
Diffusion, Osmosis, and Membrane Transport
 Diffusion, Osmosis, and Membrane Transport Introduction... 2 Diffusion and osmosis as related to cellular processes... 2 The hotter the medium, the faster the molecules diffuse... 2 TASK 1: TEMPERATURE
Diffusion, Osmosis, and Membrane Transport Introduction... 2 Diffusion and osmosis as related to cellular processes... 2 The hotter the medium, the faster the molecules diffuse... 2 TASK 1: TEMPERATURE
Lecture 8. Protein Trafficking/Targeting. Protein targeting is necessary for proteins that are destined to work outside the cytoplasm.
 Protein Trafficking/Targeting (8.1) Lecture 8 Protein Trafficking/Targeting Protein targeting is necessary for proteins that are destined to work outside the cytoplasm. Protein targeting is more complex
Protein Trafficking/Targeting (8.1) Lecture 8 Protein Trafficking/Targeting Protein targeting is necessary for proteins that are destined to work outside the cytoplasm. Protein targeting is more complex
