Structure, Function, and Ecology in the Goatfishes (Family Mullidae)!
|
|
|
- Baldwin Stone
- 9 years ago
- Views:
Transcription
1 Pacific Science (1984), vol. 38, no by the University of Hawaii Press. All rights reserved Structure, Function, and Ecology in the Goatfishes (Family Mullidae)! WILLIAM A. GOSLINE 2 ABSTRACT: The Mullidae differ from other percoid families in a number of structural features. Th e most notable ofthese is a pair ofhighly developed hyoid barbels used in feeding. Many ofthe other goatfish specializations seem to be associated in one way or another with the use made of these barbels. Structural peculiarities of the Mullidae are described and their functional and ecological implications suggested where possible. The hypothesis is made that the goatfishes have evolved a distincti ve ecological niche for themselves based on their use of the barbels in hunting. MOST OF THE50 ORSOfamilies of percoid fishes are separated from one another by minor structural features (Regan 1913). The goatfishes (Family Mullidae) differ dra stically from this general pattern. They have developed a structur al peculiarity unique among percoids, the hyoid barbels, and a number of other specializations, several of which appear to be functionally or structurally associated with the barbels.the adaptations of the Mullid ae seem to have been rather successful, for goatfi shes are represented circumtropically in inshore areas, often by large numbers of individual s. Within the family, there appears to have been a minor adaptive radi ation based on the exploitation of the barbels for various types of feeding because the principal differences between the six otherwise quite similar genera of the family are in dentition. A pair of well-developed barbels is present in all adult goatfishes. Th e two members of each pair are independently movable and bear numerou s sense organs (Sato 1937). When a goatfish is not feeding, the barbels are folded back under the rims of the chin and gill covers. In feeding, the barbels are lowered to touch the bottom under the fish. Probably all goatfishes use their barbels to detect food items on or slightly below the surface of the substrate (Figu re 1). When an area with such items is located, different goatfishes adopt various methods for obtaining the prey. Some, such as I Manu script accepted I July University of Michigan, Museum of Zoology, Ann Arbor, Michigan Upeneus tragula, shown in Figure 2, use their barbels as an excavating device (see also the comments of Hiatt and Strasburg [1960: 84] concerning Parupeneus cyclostomus). In Hawaii Parupeneus cyclostomus (formerly P. chryserydros) pokes its barbels into holes and crevices in hard substrates to dislodge small an imals (Hobson 1974). Mulloides fiavolineatus (Mulloidichthys samoensis auctorum) may back off from an item discovered by the barbel s (Figure 1) and " blow" away sand to uncover it (pers. obs.); the same species may also burrow into the sand with its snout to retrieve a food item (Hobson, pers. comm.). Mullus surmuletus on occasion ploughs up the mud with its snout in feeding and appa rently expels water from its mouth while doing so (LoBianco 1907). These feeding methods are only used by half-grown and adult goatfishes, for all mullids so far as known have pelagic eggs, larvae, and ju veniles (Caldwell 1962). Because of the use that goatfishes make of their barbels and functionally associated characters, the interrelationship between structure, function, and ecology in the Mullid ae is particularly close and is stre ssed here. On the oth er hand, the distinctiveness of the family seems to have obscured evidence for relationship between the Mullidae and other percoid s, and an attempt to determine these relationships has not been very successful. In the present pap er the various peculiarities of the goa tfishes are described beginning with the barbels. The se peculia rities are compared with similar developments elsewhere in teleosts and where possible are interpreted in terms of
2 Structure, Function, and Ecology in Goatfishes-GosLINE 313 FIGURE 1. Mulloidesflavolineatus probing for food (Kona, Hawaii). Photograph by E. S. Hobson. FIGURE 2. Upeneus tragula using its barbelsas an excavatingtool (UjungoffPandang, Sulawesi, formerly Makassar, Celebes). Photograph by J. E. Randall. function. Comments on mullid genera, family relationships, and ecology conclude the paper. The material on which the article is based consists of partly dissected and whole specimens ofall six mullid genera in the University of Michigan collections. The scientific names used in the paper are those suggested by Randall (pers. comm., March 4, 1984). The name changes of concern here are as follows: Parupeneus chryserydros is placed in the synonymy ofp. cyclostomus and Upeneus arge in that of U. taeniopterus; Mulloides is considered a valid generic name, replacing the more widely used substitute Mulloidichthys. The question ofwhether the genus Parupeneus should be synonymized with the closely related Pseudupeneus is left open. THE BARBEL APPARATUS There are two mechanically separate components to barbel manipulation in goatfishes (Figure 3 A, B). One has to do with the movements of the barbels (A, AI' and A z) relative to the hyoid bars with which they articulate. Each of the two barbels has its own musculature that controls such movements, thus enabling the two to move independently. The second mechanical component (B, B I, and
3 314 PACIFIC SCIENCE, Volume 38, October FIGURE 3. Diagram to illustrate goatfish barbel mechanics, Lateral view from right side. Stippled areas represent bone. Arrows indicate direction of movement. A, barbel with the ligaments that (A I) retract and (At) lower it. B, hyoid bar with the urohyal (B I ) that lowers and the M. prot ractor hyoideus (B t ) that raises it. B z) cau ses the lowering and raising of the anterior ends ofthe pair of hyoid bars relative to the head of the fish. The barbel itselfconsists ofa fleshy, sensory part that enclo ses a long, tapering, distally flexible branchiostegal ray. A large nerve enters the base of the fleshy part and innervates the numerous sense organs on the surface of the barbel. The modified branchiostegal ray (La Bianco 1907) provides a strengthening support for the barbel. It originates in a cap of fibro-cartilage which forms a socket over the tip of a forward projection from the anterior ceratohyal of the hyoid bar (Figure 4). Two ligaments insert on the cap of fibro-cartilage at the base of the barbel, one above the ceratohyal projection and one below. These ligaments lead back into musculature originating along the hyoid bar (not shown in Figure 3). Contraction of the muscle to the upper ligament (Figure 3A z) lowers the barbel whereas contractionofthe muscle to the lower ligament (A 1) retracts the barbel. The muscles to both ligaments appear to be modified parts of the hyohyoideus system (Winterbottom 1974a). The second component of the barbel apparatus governs the movement of the anterior ends of the hyoid bars. Generally in teleosts the hyoid bars are movable at both ends. An teriorly they are connected to the symphyseal area of the chin by musculature (the M. protractor hyoideus of Winterbottom 1974a; the M. genioh yoideus of Osse 1969); posteriorly to the inner surface of the suspensorium by the intercalation of a movable interhyal bone. In the Mullidae the freedom of movement ofthe anterior ends ofthe hyoid of the hyoid bars has been increased and that of their posterior ends reduced to rot ation in a firm socket. The structural changes associated with the
4 Structure, Function, and Ecology in Goatfishes-e-Go suxs 315 Ac Uh B2Hy B1 Uh Lh Ac FIGURE 4. Bones associated with the forward end of the hyoid apparatus in Pseudupeneus macula/ us. A, lateral and B, dorsal view. Only the basal part of the branchiostegal ray is shown. Ac, anterior ceratohyal; BI, first basibranchial; B2, second basibranchial; Br, branchiostegal; GI, glossohyal; Hy, first hypobranchial; Lh, lower hypoh yal; and Uh, upper hypohyai. hyoid bar movements of goatfishes are as follows. The posterior end of each hyoid bar is restricted to a socket formed primarily by a semicircular, medianly projecting flange on the inner surface of the interopercle (Figures 3, 5). In front of this flange, the posterior end of the bar is propped from above by the interhyal bone. Anteriorly, the forward end of the pair of hyoid bars is connected with the chin only by flexible membrane. The protractor hyoideus muscle that is usually in this area has moved its posterior attachment ' well back along the shaft ofthe hyoid bar (Figure 3B 2 ). It still serves to raise the hyoid bar, but the posterior displacement of its attachment permits considerably greater movement of the anteriorends ofthe bars than in other teleosts. The cause of such lowering is, as usual, contraction of the sternohyoideus muscles acting via the urohyal bone (Figure 3B 1 ). Lh A GI Teleosts have evolved ventrally directed projections ofvarious types, usually or always equipped with sense organs. Such projections range from barbels on the chin to extensions ofthe pectoral rays. However, sensory probes developed in association with branchiostegal rays are rare and occur, to my knowledge, only in the Mullidae and in the beryciform genus Polymixia. That the hyoid barbels in these two taxa have evolved independently is clear from a number ofstructural differences between them. Furthermore, the barbel apparatus of the Mullidae is much more specialized than that ofpolymixia. In Polymixia three modified branchiostegal rays support the barbel rather than the one ray in the Mullidae (Starks 1904). In Polymixia the hyoid bar has the usualmovable attachment at both ends rather than the mod ifications described above in the Mullidae. There is also a considerable difference between the two taxa in the basal articulation of the barbel and in the bones at the anterior end of the hyoid bar. In Polymixia the hypohyal bones hold their usual position, one under the other, at the anterior end of the hyoid bar, and the three branchiostegal rays supporting the barbel have simply moved forward to an articulation with the under surface of the lower hypohyal (Starks 1904: fig. 4). In the larval goatfish Mullus surmuletus the branchiostegal rays have their normal serial arrangement (LoBianco 1907), but during growth the anterior branchiostegal moves forward along the hyoid bar and becomes widely separated from the other branchiostegal rays. This forward movement ofthe anterior branchiostegal ray is apparently accompanied by the development of a forward projection from the lower part ofthe anterior ceratohyal, because in the adultgoatfish, this projectionextends anterior to the two hypohyals which themselves have become horizontally aligned (Figure 4; see also Starks 1904: fig. 5). The result of these changesis that in goatfishes the barbel articulation has moved forward to about the level of the tip of the glossohyal (concealed under the membrane of the floor of the mouth) and forward of the hypohyals (Figure 4).
5 316 PACIFIC SCIENCE, Volume 38, October t--0 p ---,~--\--,t p 0 /I..;.-----,,~ j'-- Ih Qu 8y Hy 10 F IGURE 5. Internal view of the suspensorium of Mullus barbatus with the posterior end of the hyoid bar and the interhyal included. A w, part of the A w section of the M. adducto r mandi bulae; ca, cartilage; Ec, ectopterygoid; Hm, hyomandibular; Hy, posterior part of the hyoid bar; Ih, interhyal; 10, interopercle; Ms, mesopterygoid; M t, metapterygoid; Op, opercle; Pa, palatine; Po, preope rcle; Qu, quadrate; So, subopercle; st, strut on the mesopterygoid that abuts against the back of the lateral ethmoid; and Sy, symplectic. There are two points to be made about the forward articulation of the barbel in goatfishes. First, the greater the distance of the articulation from the fulcrum at the posterior end of the hyoid bar, the greater the amount the barbel will be lowered when the bar itself is rot ated through a given angle. Presumably, then, the extent to which the hyoid bar can move the barbel do wnward has been increased by the forward displacement of the barbel articulation. The second point concerns the possible structural relationship between the anterior elongation ofthe hyoid bar and the lengthening of the preorbital part of the head that appears to have occurred in the Mullidae. In goatfishes the snout region above the mouth has apparently become extended as have the hyoid bars below the mouth, and the possibility seems high that the two features are related. THE LENGTHENIN G OF THE SNOUT REGION Th e snout length in various goatfishes is quite different, ranging from short and blunt in Mullus to con siderably extended in Parupeneus and Pseudup eneus. Nevertheless two osteological features present in all goatfishes suggest that the preorbital part of the head has been lengthened in the mullid ancestral stock. One is in the superficial circumorbital series of bon es. Usually in teleosts, the anteriormost member of the series, the lacrimal, articulates with the lateral ethmoid in fron t of the orbit, and the infr aorbital bone behind it is well separa ted from the lateral ethmoid. In the Mullidae, the lacrimal seems to have shifted forward, for it has only a slight contact with the lateral ethmoid, wherea s the infraorbital behind it has the main contact with the lateral ethmoid (Smith and Bailey 1962).
6 Structure, Function, and Ecology in Goatfishes-GosLINE 317 The other apparently forward shift in the anterior head structures occurs in the suspensorium. Normally, the anteriormost bone in the suspensorium, the palatine, provides the only articulation between the forward part of the suspensorium and the skull. As with the circumorbital series, in the Mullidae a bone behind the palatine, the mesopterygoid, has developed an articulation with the skull (Figure 5). The apparent forward dislocation of the lacrimal and palatine bones in the Mullidae is almost exactly duplicated in another very different group of percoids, the Pentacerotidae (Quinquarius examined). Some pentacerotids, like mullids, also have barbels, but those of the pentacerotids are on the chin, not the hyoid bars. In Quinquarius the anterior (palatine) part of the suspensorium appears to have a normal, firm attachment to the rest of the suspensorium, and the whole compound structure seems to move as a single unit. In the Mullidae, however, the upper part of the palatine is joined to the mesopterygoid strut behind it by a flexible area of cartilage (Figure 5), providing ajoint between the anterior and posterior components of the suspensorium. Such a joint is common in percoids. Furthermore, in the Mullidae the M. adductor arcus palatini, or a more or less separate section of it, extends forward along the inner surface of the palatine. I have not seen such a forward extension elsewhere. It may have to do with the expulsion ofajet of water from the mouth in these fishes. OTHER STRU CTURES ASSOCIATED WITH THE MOUTH In addition to the goatfish specializations already discussed, there are a numberof other peculiarities associated with the mouth that range in distribution from one which is widely held among percoid fishes to at least one feature that appears to be entirely restricted to the Mullidae. Adult goatfishes feed on benthic animals and their mouths open forward and downward. Some ofthe specializations in the snout region appear to be related to their bottom feeding. The lacrimal bone, as in many bottomfeeding percoids, (e.g., the lutjanoid-sparoidhaemuloid series, Johnson 1980), is extended downward so as to overlap a large part of the maxillary shaft. Also, as in the same and other percoid groups, the originally movable nasal bone has in goatfishes a rigid posterior attachment to the skull. LoBianco (1907) suggests that in Mullus this increased rigidity of the anterior part ofthe skull is associated with the habit in this genus of ploughing up the bottom with its snout. Goatfishes have gone one step further along this same line of development than many fishes with rigidly attached nasal bones. They have lost the usual ethmoid-maxillary ligament and have functionally replaced it with a ligament that passes anteriorly from the lateral border of the nasal bone over the palatine prong to an attachment on the forward rim of the maxillary (Figure 6A). This same substitution of ligaments occurs elsewhere in percoids in such fishes as the pentacerotid Quinquarius and in lethrinids (pers. obs. ; see also Johnson 1980). In goatfishes the premaxillae protrude in a more or less downward direction, and a number of specializations seem to be associated with this. The anterior face ofthe vomer has a raised, median, cartilaginous ridge (Starks 1926) over which the ascending premaxillary processes slide. There is a similar ridge in, for example, Menticirrhus, a sciaenid with a downwardly projecting mouth opening. In goatfishes the shaft of the maxilla is about equally deep throughout, the primordial ligament does not pass across it to an attachment on its outer rim, and the palatine prong extends down over the maxilla and has a more or less expanded tip (Figure 6A). These features are all present in the sparoid genus Lethrinus. Other peculiarities associated with the mouth ofgoatfishes cannot readily, if at all, be functionally associated with the downward protrusion of the premaxillae. The most distinctive of these is what will here be called a supramaxillary scale (Figure 6A). Like the supramaxillary bone of many lower percoids this specialized scale, present in all Mullidae,
7 318 PACIFIC SCIENCE, Volume 38, October nm --\-\ Po Ay---lr- --~~-'--- M x - -:::j",-...,...:r-----,--7' sm Md- ~ ~_~=::::..--- :;~= rb --rm Mx----_+_ Md----1~ ph cp.figure 6. The adductor mandibulae muscle and topographically associated features in Upeneus moluccensis. A, externa l and B, interna l view. A I ' A 3, and A w ' thre e of the sections of the M. adductor mand ibula e; Ax and A y, the two part s of the usual A 2 section; ap, aponeurosis; Md, mandible; Mx, maxilla; nm, nasal-maxillary ligament; Pa, palatine; ph, upper end of the M. protractor hyoideus; rb, lower branch of the ramu s mand ibularis nerve; rm, ramus mandibularis; and sm, supramaxillary scale. extends upward from the distal end of the maxillary bone. It frequently takes on some of the characteristics of the distal end of the maxillary bone (e.g., sculpturing). Furthermore, it may underlie other, more normally developed scales. It differs from the supramaxillary bone in that its base overlaps the maxilla rather than riding on its upper rim. Furthermore, in Upeneus par vus, which has a particularly large supramaxillary scale, the re are circuli around its anterior surface. Usually in goatfishes there are a number of scales on the maxilla, but in some species of Upeneus only the supramaxillary scale ap pears to be present. I do not know ofa scale of this type elsewhere, but in a number of percoids with a scaleless maxilla there is a cartilagelike upward extension along the dorsal rim of the
8 Structure, Function, and Ecology in Goatfishes-GosLINE 319 distal end of the maxilla (e.g., in the sciaenid Menticirrhus). The configuration of the adductor mandibulae muscle, though inadequately investigated in percoids up to now, can be a good systematic character (see, for example, Johnson 1980); it certainly is within the Mullidae. As in so many small-mouthed higher teleosts (see, for example, Winterbottom 1974b) the M. adductor mandibulae of all goatfishes is complex (Figure 6). In Upeneus molueeensis, which seems to represent the basal goatfish condition in this feature, there are five more or less separate sections of this muscle passing between the suspensorium arid the jaws. The uppermost section in external view (Figure 6A) is undoubtedly an extended maxillaris (A 1) component. It originates on the posterior part of the suspensorium and passes forward to an insertion on the inner surface of the maxilla just behind the proximal head of that bone. The usual A 2 component of the M. adductor mandibulae appears to be represented by two sections (Figure 6 Ax, A y). The upper of the two in external view passes forward to an insertion along the inside of the mandible above A w (Figure 6B). The lower of the A 2 sections in external view almost completely underlies the upper. It passes forward into the aponeurosis with A w and then is continued forward by A w on the inside of the mandible. The mandibular ramus of nerve V passes forward medial to A 1 and the upper section of A, 2 then forward into the mandible between the two sections of A 2, sometimes, as in Figure 6A, with a branch that passes down across the base of the lower section of A 2 ' A 3 is the usual small muscle visible only in internal view. In addition, A w, normally limited to the mandible, has a part attached by ligament to the inner surface of the quadrate bone of the suspensorium (Figure 5). Pseudupeneus (see Smith and Bailey 1962) and Parupeneus differ notably from other goatfishes in that most or all of the A 1 section ofthe adductor mandibulae is attached to the under surface of the infraorbital bones. Such an attachment also occurs in the percoid genus Lutjanus (Smith and Bailey 1962) and certain other members of the Lutjanidae (Johnson 1980), but I do not know of such an attachment elsewhere. THE SPH ENOTI C PROJECTION Goatfishes all have one peculiarity in the bony postorbital rim. In percoids with more or less cylindrical heads (e.g., Perea, Osse 1969), the M. levator arcus palatini originates. on the under surface of the postorbital part of the skull. In fishes with high, compressed heads this muscle usually originates on the posteroventral surface of a lateral projection from the sphenotic which, with the frontal bone above it, forms a continuous rim around the posterodorsal part of the eye. In goatfishes there is a gap, crossed externally by the dermosphenotic, between the sphenotic projection and the frontal borderofthe orbit above it. In some goatfishes (e.g., Mullus, Mulloides, Upeneiehthy s) the M. dilatatlor operculi passes forward through this gap to an attachment on the innersurface ofthe orbitalwall. A similar gap in the postorbital rim occurs in some sparoid fishes (e.g., Calamus, Archosargus) but by no means all. The functional significance ofthis gap is unclear, and its systematic significance, if any, has been too little investigated to determine. STRUCTURES ASSOCIATED WITH TH E FINS Goatfishes always have two, well-separated dorsal fins. They also have a single, minute anal spine and a well-developed scaly proces s in the axil of the pelvic fin. These features, held in common with other percoids, mayor may not have value as indicators of relationship. In two other features associated with the fins, the goatfishes are highly peculiar. The scapula of the pectoral girdle contains either two or three foramina (Starks 1930). Most other teleosts ha ve only one (Starks, 1930, record s two in cirrhitids) through which a major nerve passes (see, for example, Freihofer 1963). Examination of the scapularforamina of goatfishes indicates that the anteriormost corresponds to that of other fishes, and that the other one or two transmit nothing. Their significance is unclear.
9 320 Goatfishes have 13 instead of the usual 15 branchedcaudal rays ofpercoids. A reduction in branched ray count occurs sporadically elsewhere in higher teleosts, for example in anabantoids and Scatophagus. More notable is the pattern of fusion in the caudal skeleton. Externally the caudal fin ofgoatfishes is essentially symmetrical but the caudal skeleton is more than usually asymmetrical (see figures in Monod 1968). The hypurals to the upper caudal lobe are fused to one another and to the urostylar process, but the hypurals to the lower lobe remain separate from one another and from the urostyle. I have not seen this type of fusion in the caudal skeleton elsewhere. The reasons behindthe differentialfusion in the upper and lower caudal lobes of goatfishes can only be a matter for speculation. However, the following possibility seems worth suggesting. Goatfishes have the forked tails of strong swimmers, which they undoubtedly are. Both lobes of the caudal fin are swung from side to side during rapid locomotion, and the fusion in the parts of the caudal skeleton associated with the upper caudal lobe may increase the strengthof the tail beat. However, during hunting (Figure 1) or feeding (Figure 2) it is necessary to adjust the head precisely with reference to the bottom. The relative flexibility of the lower caudal lobe may be used for this purpose by enabling differential movements between the upper and lower caudal lobes during slow forward movements or for lowering or raising the head during feeding. COMMENTS ON GENERA Dentition is the principal character used for differentiating goatfish genera (see, for example, Lachner 1960). Certain rectifications in descriptions of this feature may be noted here. Upeneus has the most extensive dentition among goatfishes and one that is of a generalized percoid type. The genus is said to have teeth on the jaws, vomer, and palatines (e.g., Lachner 1954). Though there are normally teeth on the vomer, they may be few. In a specimen of U. sundaicus I cannot find any at PACIFIC SCIENCE, Volume 38, October 1984 all. The teeth on the palatines, on the other hand, are always well represented. Furthermore, the palatine patch seems always to be continued back on to the ectopterygoid to a greater or lesser extent. Mullus is usually described as lacking teeth in the upper jaw. This, as Caldwell (1962) notes, is only true of adults. The vomer of Mullus is expanded into a broad, medially grooved plate bearing pebblelike teeth. Though the palatines are sometimes said to provide part of the patch of teeth on the roof of the mouth (e.g., Lachner 1960), they are toothless and quite separate from the vomer. Lachner (1960: 3)states that Pseudupeneus differs from Parupeneus in the presence on the upper jaw of a second, outer row of one to several teeth. These may be somewhat reverted and tusklike. What apparently happens here is that the teeth in the single row of juveniles develop irregularly as the fish grows, for I can find no second row of teeth in available specimens smaller than 164mm in standard length. Specimens available seem to have a single row of teeth in the lower jaw, not two or more as stated by Lachner (1960). Two other aspects of differentiation between goatfish genera deserve comment. Upeneichthys, by comparison with other genera, appears to have emphasized perception by the lateralis system, as the old name porosus applied to one of the species indicates. In U. lineatus the whole snout region is covered by a series of moderately large pores and lacks the embedded scales of other goatfish genera. Parupeneus and Pseudupeneus, two closely related genera (or subgenera), differ more radically from other goatfishes and appear to be the most specialized genera in the family. The following specializations may all be associated with their relatively elongate snouts. In addition to the attachment ofthe A 1 section ofm. adductor mandibulae to the infraorbital bones noted above, they have the epaxial body musculature extending farther forward over the top of the head than in other goatfishes (except possibly for some species of Upeneus) and have lost the superficial connection between the preopercular and temporallateralis canals present in other mullids.
10 Structure, Function, and Ecology in Goatfishcs-e-Gostrxs 321 FAMILY RELATIONSHIPS The systematic position of the Mullidae among percoid families is obscure. As already noted, several goatfish specializations appear to be unique among percoids. One of these, the increase in the number of scapular foramina, seems to offer no indication ofrelationship. Two others suggest something about mullid ancestry by implication; Morphologically, the hyoid barbels have no counterpart, rudimentary or well-developed, elsewhere among percoids, but their position and the use made of them suggest that they originated in a bottom-feeding fish. The supramaxillary scale again seems to have no counterpart elsewhere, but its position suggests a substitute development for the supramaxillary bone of many lower percoids and lower teleosts generally. If this interpretation is correct, two implications are at least worth considering: that goatfishes evolved from a stock that (1) had lost its supramaxillary bone and (2) had scales on the maxilla. Other goatfish specializations, or even suites of them, seem to have evolved independently in other percoid groups. This is true of the attachment of the A 1 section of the M. adductor mandibulae to the infraorbital bones in certain mullids and lutjanids. It also appears to be true ofthe several similarities in the anterior part of the head between mullids and the pentacerotid genus Quinquarius: the articulations of both the first infraorbital and the mesopterygoid with the lateral ethmoid, and the loss of the ethmoid-maxillary ligament. Two suggestions concerning mullid relationships have been made. One is with the Lutjanidae by Regan (1913) and the other with the Sparidae by Boulenger (1904). There are far more similarities between goatfishes and the sparoid fishes than between mullids and lutjanids. These similarities are listed below for what they are worth, but to me they do not furnish very convincing evidence of relationship. The possibility of their independent development in two different bottomfeeding percoid groups seems strong. A caveat in this regard is that some of the more notable similarities do not occur between goatfishes and all sparoids, or even with the most generalized sparoids, but rather with now one segment and now another of the sparoid group (Johnson 1980). To start with the least diagnostic, the features held in common are: 1. a well-developed scaly process in the axil of the pelvic fin; vertebrae; 3. two widely separated nostrils on each side ofthe head with the posterior one a valved slit near the orbit as in such sparids as Calamus; 4. the lacrimal bone broadly overlapping the maxilla; 5. the ethmoid-maxillary ligament functionally replaced by a ligament from the nasal bone to the maxilla as in many sparoids; 6. the maxillary blade ofabout equal width for most of its length and overlapped anteriorly by a palatine prong that tends to be expanded at its tip as in the lethrinid sparoids; 7. a gap between the sphenotic projection and the frontal border of the orbit above it as in such sparid genera as Calamus; and 8. the distally expanded symplectic. There are of course numerous differences between the mullids and the sparoids. Perhaps particularly significant among these, so far as possible relationships are concerned, are two characters of the Mullidae that might be expected in a related group but which do not occur in sparoids, namely the two wellseparated dorsal fins and the squamation of the maxilla. To me, a real relationship between mull ids and sparoids seems far from established, but I am unable to suggest a more convincing relative for the Mullidae. STRUcrUR E AND ECOLOGY The structural peculiarities of goatfishes seem to be reflected at several points in their ecology, or vice versa. Goatfishes appear to have evolved a particular feeding niche for themselves. This niche depends on a specialized method of hunting, not on the exploita-
11 322 tion of any particular food source. To judge from reports of goatfish stomach contents (see, for example, Hiatt and Strasburg 1960, Hobson 1974), mullids eat a wide variety of small to medium-sized animals. No goatfish species appears to be a food specialist. (In this. regard the gill rakers and pharyngeal dentition of all goatfishes seem to be of normal, percoid type.) The highly developed hyoid barbels seem to provide the base from which goatfishes have evolved their ecological peculiarities. Man y fishes have sensory probes by means ofwhich they locate food on or in the bottom. The goatfi sh barbel apparatus, however, has evolved beyond analagous structures in other fishes in a number of respects. The hyoid barbels of the Mullid ae are relatively long, strong, and highly movable. The y can be and are used as an excavating device (Figure 2) or for harassing and dislodging prey from holes and crevices in hard substrates. Ind eed, other fishes often take advantage of these mullid barbel activities. Hobson (1974: 962) notes that in H awaii Parupeneus chryse rydros (now P. cyclostom us) is frequently followed around by the jack, Caran x melampygus, "probably as a tactic to capture pre y driven from cover as the foraging goatfi sh disturb th e substratum," and Randall has a photograph ofajack investigating the possibilities of Mulloides foraging in the West Indie s. Equally or more important is the goatfish abilit y to raise or lower the barbel appa ratus (Figure 3) while the fish maintains a hori zontal position abo ve the substrate (Figures 1, 2). Though members of the relatively specialized Parupeneus forage in reef ar eas, the more generalized Upeneus, Mullus, and Mulloides feed over sand or mud. Sandy or muddy bottoms may occur as pockets in hard substrates but are more frequently present as bro ad, unprotected areas. There appear to be two aspects of the relationship between the goatfish barbel apparatus and feeding in such areas. One has to do with escape from predators. Goatfishes do not have effective spines; they do not bury themselve s in the substrate; and they are often far from holes in which they can take refuge. To escape, they simply swim off into open water, a system that PACIFIC SCIENCE, Volume 38, October 1984 depends on rapid acceleration and susta ined speed among other things. The horizontal position of the body above the substrate while hunting or excavating appears to be advantageous here. In moving off, goatfishes do not have first to leave the substrate or to shift from a head-down bod y position (except when they are actually feeding rather than hunting). The same horizontal position of the body above the substra te while locating food also appears advantageous as a method ofhunting over wide areas with occasional sampling of the bottom for food. Indeed, on e goatfi sh uses, at least on occasion, a roving method of hunting in which the barbels are not used at all. Hiatt and Strasburg (1960 : 87) state regarding Upeneus arge (now U. taeniopterus): " It does not probe the sand with its barbels as do oth er goatfishes but rather seeks out benthic crustaceans living exposed on the bottom. Perhaps because it does not probe, it seems swifter than oth er goatfishes." However, U. taeniop terus has weli-developed barbels which it probably uses at least on occasion, as U. tragula (Figure 2) certainl y does. To summarize briefly, goatfish feeding over sand or mud seems to combine attributes of roving predators such as the bon efish (A lbula) and the pompano (Trachinotus) with some of those of excavators such as ophichthid eels and flatfishes. As noted in the introduction, there appears to have been a minor adaptive radiation within the Mullidae. Morphological differences between gener a, together with what is known of their ecology, indicate variations in feeding methods. The relatively extensive dentition of Upeneus suggests a generalized carnivore from which the genera Mullus on the one hand and Parupeneus and Pseudupeneus on the other have become specialized in two different directions. In Mullus the absence of teeth in the adult upper jaw suggests a suction feeder, and its habit of ploughing up the mud is in line with such a feeding method. At the other extreme, the strong jaw teeth near the end of a relati vely long snout in Parupeneus and Pseudup eneus suggest that their teeth are used for immediately seizing active prey that might otherwise escape or be eaten by other fishes. That at least
12 Structure, Function, and Ecology in Goatfi shes-gosline 323 some of the prey of these two mullid genera are relatively large, active animals is indicated by the higher proportion offishes in the stomachs of Parup eneus chryserydros (now P. eyclostomus) than in the stomachs of any of the Hawaiian goatfishes Hobson (1974) investigated. ACKNOWLEDGMENTS I am greatly indebted to three people for help with this paper: John Paxton supplied specimens of Upeneichthys and Quinquarius hendeeaeanthus from the Australian Museum; Jack Randall brought me up to date on mullid nomenclature and provided Figure 2 as well as general information; and to Ted Hobson I am indebted for Figure I and a discussion of goatfish ecology. LITERATURE CITED BOULENGER, G. A Fishes (Systematic account of Teleostei). Pages , figs in The Cambridge natural history, vol. vii. Macmillan, London. CALDWELL, M. K Development and distribution oflarval and juvenile fishes ofthe family Mullidae of the western North Atlantic. Fish. Bull. Fish and Wildl. Ser. 62: , 43 figs. FREIHOFER, W. C Patterns ofthe ramus lateralis accessorius and their systema tic significance in teleostean fishes. Stanford Ichthyol. Bull. 8: , 29 figs. HIATT, R. W., and D. W. STRASBURG Ecological relationships ofthe fish fauna on coral reefs of the Marshall Islands. Eco!' Monogr. 30:65-127, 7 figs. HOBSON, E. S Feeding relationships of teleostean fishes on coral reefs in Kona, Hawaii. Fish. Bull. Fish and Wildl. Ser. 72: ,42 figs. JOHNSON, G. D The limits and relationships of the Lutjanidae and associated families. Bull. Scripps In st. Oceanogr. 24 : I-Il4, 37 figs. LACHNER, E. S A revision of the goatfish genus Upeneus with descriptions of two new species. Proc. U.S. Natl. Mus. 103: ,pls.13, Family Mullidae: Goatfishes. Pages 1-46, pis in L. P. Schultz et ai., Fishes of the Marshall and Marianas Islands, vol. 2. U.S. Natl. Mus. Bull LoBIANCO, S L'origine dei barbigli tattili nel genere Mullus. Atti della Reale Accademia dei Lincei, Ser. 5, Rendiconti, Classe di Scienzi fisiche, matematiche e naturali, 16: , 8 figs. MONOD, T Le complexe urophore des poissons teleosteens, Memoires de l'institut fondamental d'afrique noire 81: 1-702, 989 figs. OSSE, J. W. M Functional morphology of the head of the perch (Perea fluviatilis L.): An electromyographic study. Netherlands. J. Zool. 19: ,15 pis., 24 text figs. R EGAN, C. T The classification of the percoid fishes. Ann. Mag. Nat. Hist., ser. 8, 12 : SATO, M Preliminary report on the barbels ofa Japanese goatfish, Upeneoides bensasi (Temminck & Schlegel). Sci. Repts. Tohoku Imperial University, 4th Ser. (Biology), II: , 3 figs. SMITH, C. L., and R. M. BAILEY The subocular shelf of fishes. J. Morph. 110: 1-18,3 pis. STARKS, E. C The osteology of some berycoid fishes. Proc. U.S. Nat. Mus. 27: ,10 figs Bones of the ethmoid region of the fish skull. Stanford Univ. Pub!., Univ. Series, BioI. Sci. 4: , 58 figs The primary shoulder girdle of the bony fishes. Stanford Univ. Publ., Univ. Series, BioI. Sci. 6 : ,38 figs. WINTERBOTTOM, R. 1974a. A descriptive synonymy of the striated muscles of the Teleostei. Proc. Acad. Nat. Sci. Philadelphia 125: , 56 figs b. The familial phylogeny of the Tetraodontiformes (Acanthopterygii: Pisces) as evidenced by their comparative myology. Smithsonian Contr. Zool. 155, 201 pp., 185 figs.
Objectives AXIAL SKELETON. 1. Frontal Bone. 2. Parietal Bones. 3. Temporal Bones. CRANIAL BONES (8 total flat bones w/ 2 paired)
 Objectives AXIAL SKELETON SKULL 1. On a skull or diagram, identify and name the bones of the skull 2. Identify the structure and function of the bones of the skull 3. Describe how a fetal skull differs
Objectives AXIAL SKELETON SKULL 1. On a skull or diagram, identify and name the bones of the skull 2. Identify the structure and function of the bones of the skull 3. Describe how a fetal skull differs
Clarification of Terms
 Shoulder Girdle Clarification of Terms Shoulder girdle = scapula and clavicle Shoulder joint (glenohumeral joint) = scapula and humerus What is the purpose (or function) of the shoulder and entire upper
Shoulder Girdle Clarification of Terms Shoulder girdle = scapula and clavicle Shoulder joint (glenohumeral joint) = scapula and humerus What is the purpose (or function) of the shoulder and entire upper
Lab 4: Shark Myology. Other Terms Muscle fiber Origin/Insertion
 Lab 4: Shark Myology Lab Objectives 1. To gain an understanding of the muscular system of the shark. 2. To learn the insertions, origins, and actions of key muscles you study. 3. To study how the muscles
Lab 4: Shark Myology Lab Objectives 1. To gain an understanding of the muscular system of the shark. 2. To learn the insertions, origins, and actions of key muscles you study. 3. To study how the muscles
Bony Fish Anatomy Worksheet
 Educational Material Bony Fish Anatomy Worksheet Teacher Information This activity is designed to be team-taught by the classroom and art teacher. Use this guide in conjunction with fish-related art to
Educational Material Bony Fish Anatomy Worksheet Teacher Information This activity is designed to be team-taught by the classroom and art teacher. Use this guide in conjunction with fish-related art to
Classification of bones Any bone may be classified into one of the following groups:
 Skeletal system This system is made up of hard tissues like bone and cartilages. This system gives form and shape to animal body The skeleton of a living animal is made up living structures of bones. The
Skeletal system This system is made up of hard tissues like bone and cartilages. This system gives form and shape to animal body The skeleton of a living animal is made up living structures of bones. The
ON A NEW SPECIES OF DENISONIA (REPTILIA, SERPENTES) FROM NEW GUINEA
 ON A NEW SPECIES OF DENISONIA (REPTILIA, SERPENTES) FROM NEW GUINEA by L. D. BRONGERSMA and M. S. KNAAP-VAN MEEUWEN Until now the Elapid genus Denisonia had not been recorded from New Guinea, and this
ON A NEW SPECIES OF DENISONIA (REPTILIA, SERPENTES) FROM NEW GUINEA by L. D. BRONGERSMA and M. S. KNAAP-VAN MEEUWEN Until now the Elapid genus Denisonia had not been recorded from New Guinea, and this
The Axial Skeleton Eighty bones segregated into three regions
 The Axial Skeleton Eighty bones segregated into three regions Skull Vertebral column Bony thorax Bones of the Axial Skeleton Figure 7.1 The Skull The skull, the body s most complex bony structure, is formed
The Axial Skeleton Eighty bones segregated into three regions Skull Vertebral column Bony thorax Bones of the Axial Skeleton Figure 7.1 The Skull The skull, the body s most complex bony structure, is formed
DEVELOPMENT AND GROWTH OF THE MANDIBLE
 2012-2013 ORAL BIOLOGY DEVELOPMENT AND GROWTH OF THE MANDIBLE Ass. Prof. Dr. Heba M. Elsabaa Development and Growth of the Mandible DEVELOPMENT OF THE MANDIBLE The Mandible Is the largest and strongest
2012-2013 ORAL BIOLOGY DEVELOPMENT AND GROWTH OF THE MANDIBLE Ass. Prof. Dr. Heba M. Elsabaa Development and Growth of the Mandible DEVELOPMENT OF THE MANDIBLE The Mandible Is the largest and strongest
Diagnostic characters: Lance- or tongue-shaped flatfishes with eyes on left side of body; body highly
 click for previous page 1934 Bony Fishes CYNOGLOSSIDAE Tonguefishes and tongue soles T.A. Munroe, National Marine Fisheries Service, National Museum of Natural History, Washington D.C., USA Diagnostic
click for previous page 1934 Bony Fishes CYNOGLOSSIDAE Tonguefishes and tongue soles T.A. Munroe, National Marine Fisheries Service, National Museum of Natural History, Washington D.C., USA Diagnostic
THE SHOULDER JOINT T H E G L E N O H U M E R A L ( G H ) J O I N T
 THE SHOULDER JOINT T H E G L E N O H U M E R A L ( G H ) J O I N T CLARIFICATION OF TERMS Shoulder girdle = scapula and clavicle Shoulder joint (glenohumeral joint) = scapula and humerus Lippert, p115
THE SHOULDER JOINT T H E G L E N O H U M E R A L ( G H ) J O I N T CLARIFICATION OF TERMS Shoulder girdle = scapula and clavicle Shoulder joint (glenohumeral joint) = scapula and humerus Lippert, p115
Lab 1: External Anatomy & Taxonomy
 Lab 1: External Anatomy & Taxonomy Goals: 1. Learn the basic external anatomy of fishes. 2. Learn how to identify fish species. Tasks: 1. Choose a fish. 2. Identify all major external structures of your
Lab 1: External Anatomy & Taxonomy Goals: 1. Learn the basic external anatomy of fishes. 2. Learn how to identify fish species. Tasks: 1. Choose a fish. 2. Identify all major external structures of your
Perciformes: Percoidei: Nemipteridae 3051 NEMIPTERIDAE
 click for previous page Perciformes: Percoidei: Nemipteridae 3051 NEMIPTERIDAE Threadfin breams (also whiptail breams, monocle breams, dwarf monocle breams, and coral breams) by B.C. Russell Diagnostic
click for previous page Perciformes: Percoidei: Nemipteridae 3051 NEMIPTERIDAE Threadfin breams (also whiptail breams, monocle breams, dwarf monocle breams, and coral breams) by B.C. Russell Diagnostic
Classification of Malocclusion
 Classification of Malocclusion What s going on here? How would you describe this? Dr. Robert Gallois REFERENCE: Where Do We Begin? ESSENTIALS FOR ORTHODONTIC PRACTICE By Riolo and Avery Chapter 6 pages
Classification of Malocclusion What s going on here? How would you describe this? Dr. Robert Gallois REFERENCE: Where Do We Begin? ESSENTIALS FOR ORTHODONTIC PRACTICE By Riolo and Avery Chapter 6 pages
Human Anatomy & Physiology
 PowerPoint Lecture Slides prepared by Barbara Heard, Atlantic Cape Community College Ninth Edition Human Anatomy & Physiology C H A P T E R 7 The Skeleton: Part B Annie Leibovitz/Contact Press Images Vertebral
PowerPoint Lecture Slides prepared by Barbara Heard, Atlantic Cape Community College Ninth Edition Human Anatomy & Physiology C H A P T E R 7 The Skeleton: Part B Annie Leibovitz/Contact Press Images Vertebral
Chapter 5. The Shoulder Joint. The Shoulder Joint. Bones. Bones. Bones
 Copyright The McGraw-Hill Companies, Inc. Reprinted by permission. Chapter 5 The Shoulder Joint Structural Kinesiology R.T. Floyd, Ed.D, ATC, CSCS Structural Kinesiology The Shoulder Joint 5-1 The Shoulder
Copyright The McGraw-Hill Companies, Inc. Reprinted by permission. Chapter 5 The Shoulder Joint Structural Kinesiology R.T. Floyd, Ed.D, ATC, CSCS Structural Kinesiology The Shoulder Joint 5-1 The Shoulder
its lifetime. The skeletal system is divided into: 1. Axial Division: bones of the body s axis (skulll, ribs, vertebrae)
 The Axial Skeleton The basic features of the human skeleton have been shaped by evolution, but the detailed characteristics of each bone reflect the stresses put on it. As a result, the skeleton changes
The Axial Skeleton The basic features of the human skeleton have been shaped by evolution, but the detailed characteristics of each bone reflect the stresses put on it. As a result, the skeleton changes
LABORATORY EXERCISE 12 BONE STRUCTURE AND CLASSIFICATION
 LABORATORY EXERCISE 12 BONE STRUCTURE AND CLASSIFICATION FIG. 12.1 1. Articular cartilage (hyaline cartilage) 6. Periosteum 2. Spongy bone (red marrow) 7. Proximal epiphysis 3. Medullary cavity 8. Diaphysis
LABORATORY EXERCISE 12 BONE STRUCTURE AND CLASSIFICATION FIG. 12.1 1. Articular cartilage (hyaline cartilage) 6. Periosteum 2. Spongy bone (red marrow) 7. Proximal epiphysis 3. Medullary cavity 8. Diaphysis
specimen, a practically complete cranium, was found by the author's
 Article 56.9,33N XLI.- PRELIMINARY DIAGNOSIS OF AN APPARENTLY NEW FAMILY OF INSECTIVORES. BY H. E. ANTHONY. PLATE XXIII. In July, 1916, while searching for remains of fossil mammals in Porto Rico, in accordance
Article 56.9,33N XLI.- PRELIMINARY DIAGNOSIS OF AN APPARENTLY NEW FAMILY OF INSECTIVORES. BY H. E. ANTHONY. PLATE XXIII. In July, 1916, while searching for remains of fossil mammals in Porto Rico, in accordance
Fishy Adaptations. Adapted from: Fashion a Fish in Project Wild Aquatic Education Activity Guide. The Council for Environmental Education, 1992
 Fishy Adaptations Adapted from: Fashion a Fish in Project Wild Aquatic Education Activity Guide. The Council for Environmental Education, 1992 Physical Structure Grade Level: Basic Duration: 45 minutes
Fishy Adaptations Adapted from: Fashion a Fish in Project Wild Aquatic Education Activity Guide. The Council for Environmental Education, 1992 Physical Structure Grade Level: Basic Duration: 45 minutes
reptile (Synapsida: Sphenacodontidae) from the early Permian of Texas
 zoological Journal of the Linnean Society (1992), 10.1: 127-184. With 17 figures The cranial anatomy and relationships of Secodontosaurus, an unusual mammal-like reptile (Synapsida: Sphenacodontidae) from
zoological Journal of the Linnean Society (1992), 10.1: 127-184. With 17 figures The cranial anatomy and relationships of Secodontosaurus, an unusual mammal-like reptile (Synapsida: Sphenacodontidae) from
Human, Male, White. Bone Clones Osteological Evaluation Report. Product Number: 1 intact mandible. General observations:
 Human, Male, White Product Number: Specimen Evaluated: Skeletal Inventory: BC-107 Bone Clones replica 1 intact cranium 1 intact mandible General observations: In general, the molding process has preserved
Human, Male, White Product Number: Specimen Evaluated: Skeletal Inventory: BC-107 Bone Clones replica 1 intact cranium 1 intact mandible General observations: In general, the molding process has preserved
Definition: A joint or articulation is a place in the body where two bones come together.
 Definition: A joint or articulation is a place in the body where two bones come together. CLASSES OF JOINTS. 1. Joints are classified according to how the bones are held together. 2. The three types of
Definition: A joint or articulation is a place in the body where two bones come together. CLASSES OF JOINTS. 1. Joints are classified according to how the bones are held together. 2. The three types of
Chapter 4 The Shoulder Girdle
 Chapter 4 The Shoulder Girdle Key Manubrium Clavicle Coracoidprocess Acromionprocess bony landmarks Glenoid fossa Bones Lateral Inferior Medial border angle McGraw-Hill Higher Education. All rights reserved.
Chapter 4 The Shoulder Girdle Key Manubrium Clavicle Coracoidprocess Acromionprocess bony landmarks Glenoid fossa Bones Lateral Inferior Medial border angle McGraw-Hill Higher Education. All rights reserved.
Temporo-Mandibular Joint Complex Exercise Suggestions
 Temporo-Mandibular Joint Complex Exercise Suggestions I. Exercise Generalizations: A. Patients with post-traumatic TMJ problems or with recent-onset dysfunction that is largely posture-related will generally
Temporo-Mandibular Joint Complex Exercise Suggestions I. Exercise Generalizations: A. Patients with post-traumatic TMJ problems or with recent-onset dysfunction that is largely posture-related will generally
Thoracic Spine Anatomy
 A Patient s Guide to Thoracic Spine Anatomy 228 West Main, Suite C Missoula, MT 59802 Phone: info@spineuniversity.com DISCLAIMER: The information in this booklet is compiled from a variety of sources.
A Patient s Guide to Thoracic Spine Anatomy 228 West Main, Suite C Missoula, MT 59802 Phone: info@spineuniversity.com DISCLAIMER: The information in this booklet is compiled from a variety of sources.
Divisions of the Skeletal System
 OpenStax-CNX module: m46344 1 Divisions of the Skeletal System OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this
OpenStax-CNX module: m46344 1 Divisions of the Skeletal System OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this
Anatomy and Physiology 121: Muscles of the Human Body
 Epicranius Anatomy and Physiology 121: Muscles of the Human Body Covers upper cranium Raises eyebrows, surprise, headaches Parts Frontalis Occipitalis Epicranial aponeurosis Orbicularis oculi Ring (sphincter)
Epicranius Anatomy and Physiology 121: Muscles of the Human Body Covers upper cranium Raises eyebrows, surprise, headaches Parts Frontalis Occipitalis Epicranial aponeurosis Orbicularis oculi Ring (sphincter)
Vertebral anatomy study guide. Human Structure Summer 2015. Prepared by Daniel Schmitt, Angel Zeininger, and Karyne Rabey.
 Vertebral anatomy study guide. Human Structure Summer 2015 Prepared by Daniel Schmitt, Angel Zeininger, and Karyne Rabey. 1. Plan of Action: In this guide you will learn to identify these structures: Cervical
Vertebral anatomy study guide. Human Structure Summer 2015 Prepared by Daniel Schmitt, Angel Zeininger, and Karyne Rabey. 1. Plan of Action: In this guide you will learn to identify these structures: Cervical
Chapter 11. What are the functions of the skeletal system? More detail on bone
 Skeletal System Chapter 11 11.1 Overview of the skeletal system What are the functions of the skeletal system? 1. Supports the body 2. Protects the soft body parts 3. Produces blood cells 4. Stores minerals
Skeletal System Chapter 11 11.1 Overview of the skeletal system What are the functions of the skeletal system? 1. Supports the body 2. Protects the soft body parts 3. Produces blood cells 4. Stores minerals
CHAPTER 9 BODY ORGANIZATION
 CHAPTER 9 BODY ORGANIZATION Objectives Identify the meaning of 10 or more terms relating to the organization of the body Describe the properties of life Describe the function for the structures of the
CHAPTER 9 BODY ORGANIZATION Objectives Identify the meaning of 10 or more terms relating to the organization of the body Describe the properties of life Describe the function for the structures of the
UNIVERSITY OF MICHIGAN THE FIRST ICNOWN BLIND FISH OF THE FAMILY CHARACIDAE: A NEW GENUS FROM MEXICO
 OCCASIONAL PAPERS OF THE MUSEUM OF ZOOLOGY UNIVERSITY OF MICHIGAN THE FIRST ICNOWN BLIND FISH OF THE FAMILY CHARACIDAE: A NEW GENUS FROM MEXICO AMONG the interesting fish novelties being introduced from
OCCASIONAL PAPERS OF THE MUSEUM OF ZOOLOGY UNIVERSITY OF MICHIGAN THE FIRST ICNOWN BLIND FISH OF THE FAMILY CHARACIDAE: A NEW GENUS FROM MEXICO AMONG the interesting fish novelties being introduced from
The Anatomy of Spinal Cord Injury (SCI)
 The Anatomy of Spinal Cord Injury (SCI) What is the Spinal Cord? The spinal cord is that part of your central nervous system that transmits messages between your brain and your body. The spinal cord has
The Anatomy of Spinal Cord Injury (SCI) What is the Spinal Cord? The spinal cord is that part of your central nervous system that transmits messages between your brain and your body. The spinal cord has
Anatomy PHL 212. By Dr Tajdar Husain Khan
 Anatomy PHL 212 By Dr Tajdar Husain Khan Overview of Anatomy Anatomy(from the Greek word anatome,"dissection") is a branch of natural science dealing with the structural organization of living things The
Anatomy PHL 212 By Dr Tajdar Husain Khan Overview of Anatomy Anatomy(from the Greek word anatome,"dissection") is a branch of natural science dealing with the structural organization of living things The
Activity: Can You Identify the Age?
 Activity: Can You Identify the Age? Skeletons are good age markers because teeth and bones mature at fairly predictable rates. How Teeth Reveal Age For toddler to age 21, teeth are the most accurate age
Activity: Can You Identify the Age? Skeletons are good age markers because teeth and bones mature at fairly predictable rates. How Teeth Reveal Age For toddler to age 21, teeth are the most accurate age
Diagnostic characters: Small to medium-sized fishes recognized by their shortened, eel-like shape;
 click for previous page 1740 Bony Fishes Suborder ZOARCOIDEI ZOARCIDAE Eelpouts by M.E. Anderson, South African Institute for Aquatic Biodiversity, South Africa Diagnostic characters: Small to medium-sized
click for previous page 1740 Bony Fishes Suborder ZOARCOIDEI ZOARCIDAE Eelpouts by M.E. Anderson, South African Institute for Aquatic Biodiversity, South Africa Diagnostic characters: Small to medium-sized
Laerdal' Human Anatomy Manual The Skeleton
 Human Anatomy Manual The Skeleton Laerdal Texas P.O. Box 38.226 EM. 116 Gatesville,Texas U.S.A.76528 U.S.A.1-800-433-5539 IntemationaI1-254-865-7221 24 Hour Fax 254-865-8011 ~ Laerdal' TABLE OF CONTENTS
Human Anatomy Manual The Skeleton Laerdal Texas P.O. Box 38.226 EM. 116 Gatesville,Texas U.S.A.76528 U.S.A.1-800-433-5539 IntemationaI1-254-865-7221 24 Hour Fax 254-865-8011 ~ Laerdal' TABLE OF CONTENTS
THE SKELETAL SYSTEM - THE AXIAL SKELETON
 THE SKELETAL SYSTEM - THE AXIAL SKELETON Chapter 7 Anatomy and Physiology Lecture 1 THE SKELETAL SYSTEM THE AXIAL SKELETON Skeletal System forms the framework of the body. TYPES OF BONES: FOUR PRINCIPAL
THE SKELETAL SYSTEM - THE AXIAL SKELETON Chapter 7 Anatomy and Physiology Lecture 1 THE SKELETAL SYSTEM THE AXIAL SKELETON Skeletal System forms the framework of the body. TYPES OF BONES: FOUR PRINCIPAL
Researcher 2013;5(12) http://www.sciencepub.net/researcher
 The Mouth and Gastro-Intestinal Tract of Pomadasys Jubelini (Cuvier, 1830) In the New Calabar-Bonny River, Rivers State, Nigeria Agbugui, M. O. Department of Biological Sciences, Ahmadu Bello University
The Mouth and Gastro-Intestinal Tract of Pomadasys Jubelini (Cuvier, 1830) In the New Calabar-Bonny River, Rivers State, Nigeria Agbugui, M. O. Department of Biological Sciences, Ahmadu Bello University
Lumbar Spine Anatomy. eorthopod.com 228 West Main St., Suite D Missoula, MT 59802-4345 Phone: 406-721-3072 Fax: 406-721-2619 info@eorthopod.
 A Patient s Guide to Lumbar Spine Anatomy 228 West Main St., Suite D Missoula, MT 59802-4345 Phone: 406-721-3072 Fax: 406-721-2619 info@eorthopod.com DISCLAIMER: The information in this booklet is compiled
A Patient s Guide to Lumbar Spine Anatomy 228 West Main St., Suite D Missoula, MT 59802-4345 Phone: 406-721-3072 Fax: 406-721-2619 info@eorthopod.com DISCLAIMER: The information in this booklet is compiled
Structure & Function of the Knee. One of the most complex simple structures in the human body. The middle child of the lower extremity.
 Structure & Function of the Knee One of the most complex simple structures in the human body. The middle child of the lower extremity. Osteology of the Knee Distal femur (ADDuctor tubercle) Right Femur
Structure & Function of the Knee One of the most complex simple structures in the human body. The middle child of the lower extremity. Osteology of the Knee Distal femur (ADDuctor tubercle) Right Femur
II. Axial Skeleton (Skull, Thoracic Cage, and Vertebral Column)
 THE SKELETAL SYSTEM Lab Objectives Students should be able to: 1. Recognize bones and bone markings for the axial and appendicular skeleton 2. Recognize bones disarticulated and/or articulated 3. Identify
THE SKELETAL SYSTEM Lab Objectives Students should be able to: 1. Recognize bones and bone markings for the axial and appendicular skeleton 2. Recognize bones disarticulated and/or articulated 3. Identify
PHYLETIC STUDIES OF TELEOSTEAN FISHES, - WITH A PROVISIONAL CLASSIFICATION OF LIVING FORMS
 PHYLETIC STUDIES OF TELEOSTEAN FISHES, - WITH A PROVISIONAL CLASSIFICATION OF LIVING FORMS P. HUMPHRY GREENWOOD, DONN E. ROSEN, STANLEY H. WEITZMAN, AND GEORGE S MYERS VOLUME,-.,~ ~~~~~~~~~~~~~~~~~~,mlt
PHYLETIC STUDIES OF TELEOSTEAN FISHES, - WITH A PROVISIONAL CLASSIFICATION OF LIVING FORMS P. HUMPHRY GREENWOOD, DONN E. ROSEN, STANLEY H. WEITZMAN, AND GEORGE S MYERS VOLUME,-.,~ ~~~~~~~~~~~~~~~~~~,mlt
Lateral pterygoid muscle Medial pterygoid muscle
 PATIENT INFORMATION BOOKLET Trismus Normal Jaw Function The jaw is a pair of bones that form the framework of the mouth and teeth. The upper jaw is called the maxilla. The lower jaw is called the mandible.
PATIENT INFORMATION BOOKLET Trismus Normal Jaw Function The jaw is a pair of bones that form the framework of the mouth and teeth. The upper jaw is called the maxilla. The lower jaw is called the mandible.
Lab 5 Overview of the Skeleton: Classification and Structure of Bones and Cartilages Exercise 9 The Axial Skeleton Exercise 10
 Lab 5 Overview of the Skeleton: Classification and Structure of Bones and Cartilages Exercise 9 The Axial Skeleton Exercise 10 Overview of the Skeleton Locate the important cartilages in the human skeleton
Lab 5 Overview of the Skeleton: Classification and Structure of Bones and Cartilages Exercise 9 The Axial Skeleton Exercise 10 Overview of the Skeleton Locate the important cartilages in the human skeleton
CSE511 Brain & Memory Modeling. Lect04: Brain & Spine Neuroanatomy
 CSE511 Brain & Memory Modeling CSE511 Brain & Memory Modeling Lect02: BOSS Discrete Event Simulator Lect04: Brain & Spine Neuroanatomy Appendix of Purves et al., 4e Larry Wittie Computer Science, StonyBrook
CSE511 Brain & Memory Modeling CSE511 Brain & Memory Modeling Lect02: BOSS Discrete Event Simulator Lect04: Brain & Spine Neuroanatomy Appendix of Purves et al., 4e Larry Wittie Computer Science, StonyBrook
Anatomy and Pathomechanics of the Sacrum and Pelvis. Charles R. Thompson Head Athletic Trainer Princeton University
 Anatomy and Pathomechanics of the Sacrum and Pelvis Charles R. Thompson Head Athletic Trainer Princeton University Simplify Everything There are actually only three bones: Two innominates, one sacrum.
Anatomy and Pathomechanics of the Sacrum and Pelvis Charles R. Thompson Head Athletic Trainer Princeton University Simplify Everything There are actually only three bones: Two innominates, one sacrum.
Transverse Sections of the Spinal Cord
 Transverse Sections of the Spinal Cord The spinal cord is perhaps the most simply arranged part of the CNS. Its basic structure, indicated in a schematic drawing of the eighth cervical segment (Figure
Transverse Sections of the Spinal Cord The spinal cord is perhaps the most simply arranged part of the CNS. Its basic structure, indicated in a schematic drawing of the eighth cervical segment (Figure
Anatomy and Terminology of the Spine. Bones of the Spine (Vertebrae)
 Anatomy and Terminology of the Spine The spine, also called the spinal column, vertebral column or backbone, consists of bones, intervertebral discs, ligaments, and joints. In addition, the spine serves
Anatomy and Terminology of the Spine The spine, also called the spinal column, vertebral column or backbone, consists of bones, intervertebral discs, ligaments, and joints. In addition, the spine serves
The Shoulder Complex & Shoulder Girdle
 The Shoulder Complex & Shoulder Girdle The shoulder complex 4 articulations involving The sternum The clavicle The ribs The scapula and The humerus Bony Landmarks provide attachment points for muscles
The Shoulder Complex & Shoulder Girdle The shoulder complex 4 articulations involving The sternum The clavicle The ribs The scapula and The humerus Bony Landmarks provide attachment points for muscles
Laboratory 1 Anatomical Planes and Regions
 Laboratory 1 Anatomical Planes and Regions Goals: Define the anatomical position, including the application of the terms right and left. List and correctly use the major directional terms used in anatomy.
Laboratory 1 Anatomical Planes and Regions Goals: Define the anatomical position, including the application of the terms right and left. List and correctly use the major directional terms used in anatomy.
Biomechanical Analysis of the Deadlift (aka Spinal Mechanics for Lifters) Tony Leyland
 Biomechanical Analysis of the Deadlift (aka Spinal Mechanics for Lifters) Tony Leyland Mechanical terminology The three directions in which forces are applied to human tissues are compression, tension,
Biomechanical Analysis of the Deadlift (aka Spinal Mechanics for Lifters) Tony Leyland Mechanical terminology The three directions in which forces are applied to human tissues are compression, tension,
In Class IV arch: Fulcrum line passes through two abutments adjacent to single edentulous space.
 It is that part of removable partial denture which assists the direct retainers in preventing displacement of distal extension denture bases by resisting lever action from the opposite side of the fulcrum
It is that part of removable partial denture which assists the direct retainers in preventing displacement of distal extension denture bases by resisting lever action from the opposite side of the fulcrum
9/3/2013 JOINTS. Joints. Axial Skeleton STRUCTURE AND FUNCTION:
 STRUCTURE AND FUNCTION: JOINTS Joints A connection between 2 or more bones A pivot point for bony motion The features of the joint help determine The ROM freedom Functional potential of the joint Axial
STRUCTURE AND FUNCTION: JOINTS Joints A connection between 2 or more bones A pivot point for bony motion The features of the joint help determine The ROM freedom Functional potential of the joint Axial
Expansion screws. Standard expansion screws. Expansion screw Mini. Expansion screw Mini
 Standard expansion screws Expansion screw Mini holes in body of screws for better retention in acrylic built-in stops prevent screws from coming apart Indication: for transversal expansion and distalization
Standard expansion screws Expansion screw Mini holes in body of screws for better retention in acrylic built-in stops prevent screws from coming apart Indication: for transversal expansion and distalization
Headgear Appliances. Dentofacial Orthopedics and Orthodontics. A Common Misconception. What is Headgear? Ideal Orthodontic Treatment Sequence
 Ideal Orthodontic Treatment Sequence Headgear Appliances Natalie A. Capan, D.M.D. 580 Sylvan Avenue, Suite 1M Englewood Cliffs, New Jersey 07632 (201)569-9055 www.capanorthodontics.com CapanOrtho@nj.rr.com
Ideal Orthodontic Treatment Sequence Headgear Appliances Natalie A. Capan, D.M.D. 580 Sylvan Avenue, Suite 1M Englewood Cliffs, New Jersey 07632 (201)569-9055 www.capanorthodontics.com CapanOrtho@nj.rr.com
There When You Need Them: 10 Principles of Successful RPD Treatment
 There When You Need Them: 10 Principles of Successful RPD Treatment Jeff Scott, DMD Jeff.Scott@jeffscottdentistry.com 239 2 nd Ave South Suite 100 St. Petersburg, FL 33701 The West Coast District Dental
There When You Need Them: 10 Principles of Successful RPD Treatment Jeff Scott, DMD Jeff.Scott@jeffscottdentistry.com 239 2 nd Ave South Suite 100 St. Petersburg, FL 33701 The West Coast District Dental
Lectures of Human Anatomy
 Lectures of Human Anatomy Vertebral Column-I By DR. ABDEL-MONEM AWAD HEGAZY M.B. with honor 1983, Dipl."Gynecology and Obstetrics "1989, Master "Anatomy and Embryology" 1994, M.D. "Anatomy and Embryology"
Lectures of Human Anatomy Vertebral Column-I By DR. ABDEL-MONEM AWAD HEGAZY M.B. with honor 1983, Dipl."Gynecology and Obstetrics "1989, Master "Anatomy and Embryology" 1994, M.D. "Anatomy and Embryology"
Introduction. I. Objectives. II. Introduction. A. To become familiar with the terms of direction and location.
 E X E R C I S E Introduction I. Objectives A. To become familiar with the terms of direction and location. B. To become familiar with different types of planes and sections. C. To learn the names and locations
E X E R C I S E Introduction I. Objectives A. To become familiar with the terms of direction and location. B. To become familiar with different types of planes and sections. C. To learn the names and locations
Fundus Photograph Reading Center
 Modified 7-Standard Field Digital Color Fundus Photography (7M-D) 8010 Excelsior Drive, Suite 100, Madison WI 53717 Telephone: (608) 410-0560 Fax: (608) 410-0566 Table of Contents 1. 7M-D Overview... 2
Modified 7-Standard Field Digital Color Fundus Photography (7M-D) 8010 Excelsior Drive, Suite 100, Madison WI 53717 Telephone: (608) 410-0560 Fax: (608) 410-0566 Table of Contents 1. 7M-D Overview... 2
The etiology of orthodontic problems Fifth session
 بنام خداوند جان و خرد The etiology of orthodontic problems Fifth session دکتر مھتاب نوری دانشيار گروه ارتدنسی Course Outline( 5 sessions) Specific causes of malocclusion Genetic Influences Environmental
بنام خداوند جان و خرد The etiology of orthodontic problems Fifth session دکتر مھتاب نوری دانشيار گروه ارتدنسی Course Outline( 5 sessions) Specific causes of malocclusion Genetic Influences Environmental
AUSTRALIAN MUSEUM SCIENTIFIC PUBLICATIONS
 AUSTRALIAN MUSEUM SCIENTIFIC PUBLICATIONS Bayly, I. A. E., 1971. A new species of Kelleria (Copepoda: Cyclopoida) from brackish water in Victoria. Records of the Australian Museum 28(6): 111 116. [27 September
AUSTRALIAN MUSEUM SCIENTIFIC PUBLICATIONS Bayly, I. A. E., 1971. A new species of Kelleria (Copepoda: Cyclopoida) from brackish water in Victoria. Records of the Australian Museum 28(6): 111 116. [27 September
Shapely Shoulders. Bodysculpting Routine
 8 Shapely Shoulders The shoulders are one of the most aesthetically pleasing muscle groups; when properly developed, they can literally redefine your physique. The importance of nicely rounded shoulders
8 Shapely Shoulders The shoulders are one of the most aesthetically pleasing muscle groups; when properly developed, they can literally redefine your physique. The importance of nicely rounded shoulders
Thyroid eye disease (TED) Synonyms: Graves ophthalmopathy, thyroid ophthalmopathy, thyroid associated ophthalmopathy
 Thyroid eye disease (TED) Synonyms: Graves ophthalmopathy, thyroid ophthalmopathy, thyroid associated ophthalmopathy This information leaflet briefly covers the following issues in TED: What is TED? When
Thyroid eye disease (TED) Synonyms: Graves ophthalmopathy, thyroid ophthalmopathy, thyroid associated ophthalmopathy This information leaflet briefly covers the following issues in TED: What is TED? When
TWO NEW FRESHWATER SPECIES OF THE GENUS JESOGAMMARUS (CRUSTACEA: AMPHIPODA: ANISOGAMMARIDAE) FROM CHINA
 THE RAFFLES BULLETIN OF ZOOLOGY 2004 THE RAFFLES BULLETIN OF ZOOLOGY 2004 52(2): 455-466 National University of Singapore TWO NEW FRESHWATER SPECIES OF THE GENUS JESOGAMMARUS (CRUSTACEA: AMPHIPODA: ANISOGAMMARIDAE)
THE RAFFLES BULLETIN OF ZOOLOGY 2004 THE RAFFLES BULLETIN OF ZOOLOGY 2004 52(2): 455-466 National University of Singapore TWO NEW FRESHWATER SPECIES OF THE GENUS JESOGAMMARUS (CRUSTACEA: AMPHIPODA: ANISOGAMMARIDAE)
Anatomy of Skeletal System
 Anatomy of Skeletal System two main subdivisions of skeletal system: axial : skull, vertebral column, rib cage appendicular: arms and legs and girdles Bone Markings: Foramen: opening in bone passageway
Anatomy of Skeletal System two main subdivisions of skeletal system: axial : skull, vertebral column, rib cage appendicular: arms and legs and girdles Bone Markings: Foramen: opening in bone passageway
Introduction to Dental Anatomy
 Introduction to Dental Anatomy Vickie P. Overman, RDH, MEd Continuing Education Units: N/A This continuing education course is intended for dental students and dental hygiene students. Maintaining the
Introduction to Dental Anatomy Vickie P. Overman, RDH, MEd Continuing Education Units: N/A This continuing education course is intended for dental students and dental hygiene students. Maintaining the
CHAPTER 8: JOINTS OF THE SKELETAL SYSTEM. 4. Name the three types of fibrous joints and give an example of each.
 OBJECTIVES: 1. Define the term articulation. 2. Distinguish between the functional and structural classification of joints, and relate the terms that are essentially synonymous. 3. Compare and contrast
OBJECTIVES: 1. Define the term articulation. 2. Distinguish between the functional and structural classification of joints, and relate the terms that are essentially synonymous. 3. Compare and contrast
THE SKELETAL & ARTICULAR SYSTEMS. The Bones & Joints
 THE SKELETAL & ARTICULAR SYSTEMS The Bones & Joints CLOSE YOUR POWERPOINT HANDOUTS!! Think-Pair-Share: Why do we need bones? Try to think of 3 reasons. THE SKELETAL SYSTEM Is made up of numerous bones
THE SKELETAL & ARTICULAR SYSTEMS The Bones & Joints CLOSE YOUR POWERPOINT HANDOUTS!! Think-Pair-Share: Why do we need bones? Try to think of 3 reasons. THE SKELETAL SYSTEM Is made up of numerous bones
Parts of the Brain. Chapter 1
 Chapter 1 Parts of the Brain Living creatures are made up of cells. Groups of cells, similar in appearance and with the same function, form tissue. The brain is a soft mass of supportive tissues and nerve
Chapter 1 Parts of the Brain Living creatures are made up of cells. Groups of cells, similar in appearance and with the same function, form tissue. The brain is a soft mass of supportive tissues and nerve
Surgical Art. Formulaic Drawing Method. DRAWING WORKSHOP Learning to sketch for patient notes
 DRAWING WORKSHOP Learning to sketch for patient notes Surgical Art Formulaic Drawing Method Formulaic figure drawing systems involve using abstract rhythms and interlocking shapes to construct the human
DRAWING WORKSHOP Learning to sketch for patient notes Surgical Art Formulaic Drawing Method Formulaic figure drawing systems involve using abstract rhythms and interlocking shapes to construct the human
Observing Vertebrate Skeletons
 Name Class Date Chapter 33 Comparing Chordates Observing Vertebrate Skeletons Introduction One characteristic common to all vertebrates is the presence of a skeleton. The endoskeleton provides support,
Name Class Date Chapter 33 Comparing Chordates Observing Vertebrate Skeletons Introduction One characteristic common to all vertebrates is the presence of a skeleton. The endoskeleton provides support,
click for previous page CHIMAERAS
 click for previous page CHIMAERAS by D.A. Didier, Academy of Natural Sciences, Philadelphia, Pennsylvania, USA 592 Chimaeras TECHNICAL TERMS AND MEASUREMENTS total length spine frontal tenaculum lateral-line
click for previous page CHIMAERAS by D.A. Didier, Academy of Natural Sciences, Philadelphia, Pennsylvania, USA 592 Chimaeras TECHNICAL TERMS AND MEASUREMENTS total length spine frontal tenaculum lateral-line
Upper Limb QUESTIONS UPPER LIMB: QUESTIONS
 1 Upper Limb QUESTIONS 1.1 Which of the following statements best describes the scapula? a. It usually overlies the 2nd to 9th ribs. b. The spine continues laterally as the coracoid process. c. The suprascapular
1 Upper Limb QUESTIONS 1.1 Which of the following statements best describes the scapula? a. It usually overlies the 2nd to 9th ribs. b. The spine continues laterally as the coracoid process. c. The suprascapular
DISSECTION OF THE SHEEP'S BRAIN
 DISSECTION OF THE SHEEP'S BRAIN Introduction The purpose of the sheep brain dissection is to familiarize you with the threedimensional structure of the brain and teach you one of the great methods of studying
DISSECTION OF THE SHEEP'S BRAIN Introduction The purpose of the sheep brain dissection is to familiarize you with the threedimensional structure of the brain and teach you one of the great methods of studying
FORENSIC ANTHROPOLOGY NOTES
 FORENSIC ANTHROPOLOGY NOTES Forensic Anthropology = the examinations of human skeletal remains for law enforcement agencies to determine the identity of unidentified bones. Questions asked: 1. Are the
FORENSIC ANTHROPOLOGY NOTES Forensic Anthropology = the examinations of human skeletal remains for law enforcement agencies to determine the identity of unidentified bones. Questions asked: 1. Are the
Longfin Mako Shark. Isurus paucus NE ATL LMA. Lateral View ( ) Ventral View ( ) APPEARANCE Longfin Mako Shark, Petit Taupe (Fr), Marrajo Carite (Es).
 Lateral View ( ) Ventral View ( ) COMMON NAMES APPEARANCE, Petit Taupe (Fr), Marrajo Carite (Es). Moderately long, conical snout with relatively large eyes. Pectoral fins at least as long as head with
Lateral View ( ) Ventral View ( ) COMMON NAMES APPEARANCE, Petit Taupe (Fr), Marrajo Carite (Es). Moderately long, conical snout with relatively large eyes. Pectoral fins at least as long as head with
Thyroid eye disease (TED)
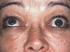 Thyroid eye disease (TED) Mr David H Verity, MD MA FRCOphth Consultant Ophthalmic Surgeon Synonyms: Graves ophthalmopathy, thyroid ophthalmopathy, thyroid associated ophthalmopathy This information leaflet
Thyroid eye disease (TED) Mr David H Verity, MD MA FRCOphth Consultant Ophthalmic Surgeon Synonyms: Graves ophthalmopathy, thyroid ophthalmopathy, thyroid associated ophthalmopathy This information leaflet
Simplified Positioning for Dental Radiology
 Simplified Positioning for Dental Radiology Prepared by: Animal Dental Care Tony M. Woodward DVM, Dipl. AVDC 5520 N. Nevada Ave. Suite 150 Colorado Springs, CO 80918 (719) 536-9949 tw@wellpets.com www.wellpets.com
Simplified Positioning for Dental Radiology Prepared by: Animal Dental Care Tony M. Woodward DVM, Dipl. AVDC 5520 N. Nevada Ave. Suite 150 Colorado Springs, CO 80918 (719) 536-9949 tw@wellpets.com www.wellpets.com
Five New Fish Species of the Genus Alabes (Gobiesocidae: Cheilobranchinae)
 Copyright Australian Museum, 2004 Records of the Australian Museum (2004) Vol. 56: 147 158. ISSN 0067-1975 Five New Fish Species of the Genus Alabes (Gobiesocidae: Cheilobranchinae) BARRY HUTCHINS* AND
Copyright Australian Museum, 2004 Records of the Australian Museum (2004) Vol. 56: 147 158. ISSN 0067-1975 Five New Fish Species of the Genus Alabes (Gobiesocidae: Cheilobranchinae) BARRY HUTCHINS* AND
by joseph e. muscolino, DO photography by yanik chauvin
 by joseph e. muscolino, DO photography by yanik chauvin body mechanics palpation of the anterior neck ESOUCES For more information go to www.medlineplus.gov and search under anterior neck. The anterior
by joseph e. muscolino, DO photography by yanik chauvin body mechanics palpation of the anterior neck ESOUCES For more information go to www.medlineplus.gov and search under anterior neck. The anterior
UNIVERSITY OF MICHIGAN PRESS
 OCCASIONAL PAPERS OF THE MUSEUM OF ZOOLOGY UNIVERSITY OF MICHIGAN ANN ARBOR, MICHIGAN UNIVERSITY OF MICHIGAN PRESS NOTES ON THE GENUS PLECTROHYLA, WITH DESCRIPTIONS OF NEW SPECIES TIIE generic and specific
OCCASIONAL PAPERS OF THE MUSEUM OF ZOOLOGY UNIVERSITY OF MICHIGAN ANN ARBOR, MICHIGAN UNIVERSITY OF MICHIGAN PRESS NOTES ON THE GENUS PLECTROHYLA, WITH DESCRIPTIONS OF NEW SPECIES TIIE generic and specific
Anterior Superior Iliac Spine. Anterior Inferior Iliac Spine. head neck greater trochanter intertrochanteric line lesser trochanter
 Ilium Bones The Skeleton Ischium Pubis Sacro-iliac Joint Iliac Crest Anterior Superior Superior Pubic Ramus Anterior Inferior Acetabulum Obturator Foramen Ischio-pubic ramus Ischial tuberosity Pubic Crest
Ilium Bones The Skeleton Ischium Pubis Sacro-iliac Joint Iliac Crest Anterior Superior Superior Pubic Ramus Anterior Inferior Acetabulum Obturator Foramen Ischio-pubic ramus Ischial tuberosity Pubic Crest
Sexual Dimorphism in the Tooth-Crown Diameters of the Deciduous Teeth
 Sexual Dimorphism in the Tooth-Crown Diameters of the Deciduous Teeth THOMAS K. BLACK 111 Museum ofanthropology, The university of Michigan, Ann Arbor, Michigan 48109 KEY WORDS Deciduous dentition criminant
Sexual Dimorphism in the Tooth-Crown Diameters of the Deciduous Teeth THOMAS K. BLACK 111 Museum ofanthropology, The university of Michigan, Ann Arbor, Michigan 48109 KEY WORDS Deciduous dentition criminant
Muscular System. Student Learning Objectives: Identify the major muscles of the body Identify the action of major muscles of the body
 Muscular System Student Learning Objectives: Identify the major muscles of the body Identify the action of major muscles of the body Structures to be identified: Muscle actions: Extension Flexion Abduction
Muscular System Student Learning Objectives: Identify the major muscles of the body Identify the action of major muscles of the body Structures to be identified: Muscle actions: Extension Flexion Abduction
A new fish, Peristedion nesium (Scorpaeniformes: Peristediidae) from Isla del Coco, Costa Rica
 A new fish, Peristedion nesium (Scorpaeniformes: Peristediidae) from Isla del Coco, Costa Rica William A. Bussing Escuela de Biología & Centro de Investigaciones en Ciencias del Mar y Limnología (CIMAR),
A new fish, Peristedion nesium (Scorpaeniformes: Peristediidae) from Isla del Coco, Costa Rica William A. Bussing Escuela de Biología & Centro de Investigaciones en Ciencias del Mar y Limnología (CIMAR),
DSM Spine+Sport - Mobility
 To set yourself up for success, practice keeping a neutral spine throughout all of these movements. This will ensure the tissue mobilization is being applied to the correct area, and make the techniques
To set yourself up for success, practice keeping a neutral spine throughout all of these movements. This will ensure the tissue mobilization is being applied to the correct area, and make the techniques
Internal Anatomy. Figure 2. The bones of the avian skull (a) lateral view (b) posterior view (c) sclerotic ring.
 Internal Anatomy Skeletal System The skeletal system (Figure 1) is highly modified to meet the structural and energetic demands of flight. Throughout the skeleton are examples of three types of adaptive
Internal Anatomy Skeletal System The skeletal system (Figure 1) is highly modified to meet the structural and energetic demands of flight. Throughout the skeleton are examples of three types of adaptive
UNIT 4 - SKELETAL SYSTEM LECTURE NOTES
 UNIT 4 - SKELETAL SYSTEM LECTURE NOTES 4.01 FUNCTIONS OF THE SKELETAL SYSTEM A. Support 1. Provides a framework for the body. 2. Supports soft tissue. 3. Serves as a point of attachment for ligaments,
UNIT 4 - SKELETAL SYSTEM LECTURE NOTES 4.01 FUNCTIONS OF THE SKELETAL SYSTEM A. Support 1. Provides a framework for the body. 2. Supports soft tissue. 3. Serves as a point of attachment for ligaments,
SYSTEMATIC APPROACH TO ORTHODONTIC DIAGNOSIS DENT 656
 SYSTEMATIC APPROACH TO ORTHODONTIC DIAGNOSIS DENT 656 ORTHODONTIC CLASSIFICATION / DIAGNOSIS Goal of diagnosis: An orderly reduction of the data base to a useful list of the patient s problems Useful??
SYSTEMATIC APPROACH TO ORTHODONTIC DIAGNOSIS DENT 656 ORTHODONTIC CLASSIFICATION / DIAGNOSIS Goal of diagnosis: An orderly reduction of the data base to a useful list of the patient s problems Useful??
SHOULDER PULL DOWNS. To learn efficient use of the shoulder blades and arms while maintaining a neutral spine position.
 SHOULDER INTRODUCT ION Welcome to your shoulder exercise program The exercises in the program are designed to improve your shoulder mobility, posture and the control of the muscles in your neck and shoulder
SHOULDER INTRODUCT ION Welcome to your shoulder exercise program The exercises in the program are designed to improve your shoulder mobility, posture and the control of the muscles in your neck and shoulder
WHIPLASH INJURIES By Prof RP Grabe, Department of Orthopaedics, University of Pretoria
 1 WHIPLASH INJURIES By Prof RP Grabe, Department of Orthopaedics, University of Pretoria In a recent publication in Spine the Quebec task force mentions that very little is available in the literature
1 WHIPLASH INJURIES By Prof RP Grabe, Department of Orthopaedics, University of Pretoria In a recent publication in Spine the Quebec task force mentions that very little is available in the literature
Unit 4: Skeletal System Test Review Test Review
 Name: Period: Unit 4: Skeletal System Test Review Test Review 1. List four functions of the skeletal system: a. b. c. d. 2. Define ossification and identify the roles of the osteoblasts, osteocytes, and
Name: Period: Unit 4: Skeletal System Test Review Test Review 1. List four functions of the skeletal system: a. b. c. d. 2. Define ossification and identify the roles of the osteoblasts, osteocytes, and
ON TWO NEW SPECIES OF THE GENUS ACANTHOCHONDRIA OAKLEY (CRUSTACEA COPEPODA) FOUND IN JAPAN
 ON TWO NEW SPECIES OF THE GENUS ACANTHOCHONDRIA OAKLEY (CRUSTACEA COPEPODA) FOUND IN JAPAN by SUEO M. SHIINO Faculty of Fisheries, Prefectural University of Mie, Tsu, Japan Acanthochondria Oakley is a
ON TWO NEW SPECIES OF THE GENUS ACANTHOCHONDRIA OAKLEY (CRUSTACEA COPEPODA) FOUND IN JAPAN by SUEO M. SHIINO Faculty of Fisheries, Prefectural University of Mie, Tsu, Japan Acanthochondria Oakley is a
Elbow & Forearm H O W V I T A L I S T H E E L B O W T O O U R D A I L Y L I V E S?
 Elbow & Forearm H O W V I T A L I S T H E E L B O W T O O U R D A I L Y L I V E S? Clarification of Terms The elbow includes: 3 bones (humerus, radius, and ulna) 2 joints (humeroulnar and humeroradial)
Elbow & Forearm H O W V I T A L I S T H E E L B O W T O O U R D A I L Y L I V E S? Clarification of Terms The elbow includes: 3 bones (humerus, radius, and ulna) 2 joints (humeroulnar and humeroradial)
LOCOMOTION AND MOVEMENT
 UNIT - HUMAN PHYSIOLOGY Chapter 18 LOCOMOTION AND MOVEMENT Movement is an important feature of living organism. Both the microbes and macrobes show wide range of movements. The movements results in change
UNIT - HUMAN PHYSIOLOGY Chapter 18 LOCOMOTION AND MOVEMENT Movement is an important feature of living organism. Both the microbes and macrobes show wide range of movements. The movements results in change
a guide to understanding crouzon syndrome a publication of children s craniofacial association
 a guide to understanding crouzon syndrome a publication of children s craniofacial association a guide to understanding crouzon syndrome this parent s guide to Crouzon syndrome is designed to answer questions
a guide to understanding crouzon syndrome a publication of children s craniofacial association a guide to understanding crouzon syndrome this parent s guide to Crouzon syndrome is designed to answer questions
Merluccius Rafinesque, 1810
 click for previous page 327 Local Names : AUSTRALIA: Blue grenadier; GERMANY: Langschwanz-Seehecht; ITALY : Nasello azzurro; JAPAN : Hoki; NEW ZEALAND : Blue hake, Hoki, Whiptail; SPAIN : Merluza azul;
click for previous page 327 Local Names : AUSTRALIA: Blue grenadier; GERMANY: Langschwanz-Seehecht; ITALY : Nasello azzurro; JAPAN : Hoki; NEW ZEALAND : Blue hake, Hoki, Whiptail; SPAIN : Merluza azul;
FCI-Standard N 105 / 29. 03. 2006 /GB FRENCH WATER DOG. (Barbet)
 FCI-Standard N 105 / 29. 03. 2006 /GB FRENCH WATER DOG (Barbet) 2 TRANSLATION : Jennifer Mulholland. ORIGIN : France DATE OF PUBLICATION OF ORIGINAL VALID STANDARD : 21.02.2006. UTILIZATION: Water dog
FCI-Standard N 105 / 29. 03. 2006 /GB FRENCH WATER DOG (Barbet) 2 TRANSLATION : Jennifer Mulholland. ORIGIN : France DATE OF PUBLICATION OF ORIGINAL VALID STANDARD : 21.02.2006. UTILIZATION: Water dog
Structure and Function of the Hip
 Structure and Function of the Hip Objectives Identify the bones and bony landmarks of the hip and pelvis Identify and describe the supporting structures of the hip joint Describe the kinematics of the
Structure and Function of the Hip Objectives Identify the bones and bony landmarks of the hip and pelvis Identify and describe the supporting structures of the hip joint Describe the kinematics of the
METHOD OF STATEMENT FOR STATIC LOADING TEST
 Compression Test, METHOD OF STATEMENT FOR STATIC LOADING TEST Tension Test and Lateral Test According to the American Standards ASTM D1143 07, ASTM D3689 07, ASTM D3966 07 and Euro Codes EC7 Table of Contents
Compression Test, METHOD OF STATEMENT FOR STATIC LOADING TEST Tension Test and Lateral Test According to the American Standards ASTM D1143 07, ASTM D3689 07, ASTM D3966 07 and Euro Codes EC7 Table of Contents
