HYBRIDOMA TECHNOLOGY FOR PRODUCTION OF MONOCLONAL ANTIBODIES
|
|
|
- Bonnie Pierce
- 10 years ago
- Views:
Transcription
1 HYBRIDOMA TECHNOLOGY FOR PRODUCTION OF MONOCLONAL ANTIBODIES Shivanand Pandey Smt. R. B. P. M. Pharmacy College, Atkot , Rajkot, Gujarat. India ABSTRACT Hybridomas are cells that have been engineered to produce a desired antibody in large amounts. To produce monoclonal antibodies, B- cells are removed from the spleen of an animal that has been challenged with the relevant antigen. These B-cells are then fused with myeloma tumor cells that can grow indefinitely in culture (myeloma is a B-cell cancer). This fusion is performed by making the cell membranes more permeable. The fused hybrid cells (called hybridomas), being cancer cells, will multiply rapidly and indefinitely and will produce large amounts of the desired antibodies. They have to be selected and subsequently cloned by limiting dilution. Supplemental media containing Interleukin-6 (such as briclone) are essential for this step.the production of monoclonal anti-bodies was first invented by Cesar Milstein, Georges J. F. Köhler and Niels Kaj Jerne in Selection occurs via culturing the newly fused primary hybridoma cells in selective-media, specifically media containing 1x concentration HAT for roughly days. After using HAT it is often desirable to use HT containing media. Cloning occurs after identification of positive primary hybridoma cells. Clone by limited dilution. While some may believe that IL-6 is essential for this step, it is not necessary to add that expensive supplement, rather use 50% heat-inactivated FBS for the first week. Add 10% FBS DMEM to the clone culture plate after screening for single colony wells. Keywords: Hybridomas, monoclonal antibodies, Interleukin-6, Supplemental media INTRODUCTION Hybridomas are cells that have been engineered to produce a desired antibody in large amounts, to produce monoclonal antibodies. (1, 2) Monoclonal antibodies can be produced in specialized cells through a technique now popularly known as hybridoma technology. 1 Hybridoma technology was discovered in 1975 by two scientists, Georges Kohler of West Germany and Cesar Milstein of Argentina (now working in U.K.), who jointly with Niels Jerne of Denmark (now working in Germany) were awarded the 1984 Noble prize for physiology and medicine. 1 Generally, the production of one MAb, using the hybridoma technology, costs between $8,000 and $12,000. The average reasonably SK can generate only 15 to 30 hybridoma fusions per year, but in an environment where the focus is on diagnostic- or therapeutic-quality MAbs, there are additional significant limitations than can further decrease throughput. Monoclonal antibodies is valuable for the analysis of parasites antigen and appropriate that WHO should have organized a symposium (held at the national university of Singapore, October 1981) which brought together those who have establish and refined the technology and those who are using it, or intending to use it for the study of organism responsible for some of the major diseases affecting mankind. Such monoclonal antibodies, as they are known, have opened remarkable new approaches to preventing, diagnosing, and treating disease. Monoclonal antibodies are used, for instance, to distinguish subsets of B cells and T cells. This knowledge is helpful not only for basic research but also for identifying different types of leukemias and lymphomas and allowing physicians to tailor treatment accordingly. Quantitating the number of B cells and helper T cells is all-important in immune disorders such as AIDS. Monoclonal antibodies are being used to track cancer antigens and, alone or linked to anticancer agents, to attack cancer metastases. The monoclonal antibody known as OKT3 is saving organ transplants threatened with rejection, and preventing bone marrow transplants from setting off graft-versus-host disease (immune system series) METHADOLOGY A hybridoma, which can be considered as a harry cell, is produced by the injection of a specific antigen into a mouse, procuring the antigen-specific plasma cells (antibody-producing cell) from the mouse's spleen and the subsequent fusion of this cell with a cancerous immune cell called a myeloma cell. The hybrid cell, which is thus produced, can be cloned to produce many identical daughter clones. These daughter clones then secrete the immune cell product. Since these antibodies come from only one type of cell (the hybridoma cell) they are called monoclonal antibodies. The advantage of this process is that it can combine the qualities of the two different types of cells; the ability to grow continually, and to produce large amounts of pure antibody. HAT medium (Hypoxanthine Aminopetrin Thymidine) is used for preparation of monoclonal antibodies. Laboratory animals (eg. mice) are first exposed to an antigen to which we are interested in isolating an antibody against. Once splenocytes are isolated from the mammal, the B cells are fused with immortalized myeloma cells - which lack the HGPRT(hypoxanthine-guanine phosphoribosyltransferase) gene - using polyethylene glycol or the Sendai virus. Fused cells are incubated in the HAT (Hypoxanthine Aminopetrin Thymidine) medium. Aminopterin in the myeloma cells die, as they cannot produce nucleotides by the de novo or salvage medium blocks the pathway that allows for nucleotide synthesis. Hence, unfused D cell die. Unfused B cells die as they have a short life span. Only the International Journal of Pharmaceutical Sciences Review and Research Page 88
2 B cell-myeloma hybrids survive, since the HGPRT gene coming from the B cells is functional. These cells produce antibodies (a property of B cells) and are immortal (a property of myeloma cells). 2 The incubated medium is then diluted into multiwell plates to such an extent that each well contains only 1 cell. Then the supernatant in each well can be checked for desired antibody. Since the antibodies in a well are produced by the same B cell, they will be directed towards the same epitope, and are known as monoclonal antibodies. 3 Once a hybridoma colony is established, it will continually grow in culture medium like RPMI-1640 (with antibiotics and foetal bovine serum) and produce antibody (Nelson et al., 2000.) 3 The next stage is a rapid primary screening process, which identifies and selects only those hybridomas that produce antibodies of appropriate specificity. The hybridoma culture supernatant, secondary enzyme labelled conjugate, and chromogenic substrate, is then incubated, and the formation of a colored product indicates a positive hybridoma. Alternatively, immunocytochemical screening can also be used (Nelson et al., Multiwell plates are used initially to grow the hybridomas and after selection, are changed to larger tissue culture flasks. This maintains the well being of the hybridomas and provides enough cells for cryopreservation and supernatant for subsequent investigations. The culture supernatant can yield 1to 60 ug/ml of monoclonal antibody, which is maintained at 20 C or lower until required (Nelson et al., 2000.)By using culture supernatant or a purified immunoglobulin preparation, further analysis of a potential monoclonal antibody producing hybridoma can be made in terms of reactivity, specificity, and crossreactivity (Nelson et al., 2000.) 3 Advancements OR Improvements in Hybridoma Technology Considerable efforts during the last years have been made to improve the yield of monoclonal antibodies using hybridoma technology 4, 5. These efforts included the following: (1) The substitution of a chemical fusion promoter (P.E.G.) for the Sendai virus initially used to promote fusion, and (2) The use of myelomas that do not secrete their own antibodies and that therefore do not interfere with the production of the required antibody (3) A continuous cell line (Sp 2/0) was used as a fusion partner for the antibody producing B cells. (4) Feeder layers consisting of extra cells to feed newly formed hybridomas were used for optimal growth and hybridoma production. The most common feeder layers consisted of murine peritoneal cells, marcrophages derived from mouse, rat or guinea pig International Journal of Pharmaceutical Sciences Review and Research Page 89
3 extra non immunized spleen cells, human fibroblasts, human peripheral blood monocytes or thymus cells; these feeder cells had some limitations like depletion of nutrients meant for hybridoma and contamination, so that other sources of hybridoma growth factors (HGF) like interleukin-6 (II-6) derived from human cells were used. Purification of Antibodies Monoclonal antibodies may need to be purified before they are used for a variety of purposes. Before final purification, the cultures may be subjected to cell fractionation for enrichment of the antibody protein. In E. coli, the antibodies may be secreted in the periplasm, which may be used for enrichment of antibody, so that further purification is simplified. Alternatively the antibodies may be purified from cell homogenate or cell debris obtained from the medium. Antibodies can be purified by anyone of the following techniques (I) ion-exchange chromatography; (ii) antigen affinity chromatography. Serum Free Media for Bulk Culture of Hybridoma Cells The media for culturing a variety of animal cells and discussed the significance of adding serum to basal nutrient media. Serum is a highly complex and poorly defined mixture of components like albumin, transferrin, lipoproteins and various hormones/growth factors. Nevertheless, serum makes an essential component of media for culturing animal cells. The use of serum, however, leads to difficulties in purification of antibodies. Furthers, it is an expensive technology for large scale production of hybridoma cells for industrial production of monoclonal antibodies. In view of these difficulties, serum free media are being increasingly used for culturing hybridoma cells. Advantages of Serum Free Media in Hybridoma Cell Culture and Preparation of Monoclonal Antibodies: 1. Greatly simplified purification of antibodies due to increased 1.initial purity and absence of contaminating immunoglobulin. 2. Decreased variability of culture medium. 3. Reduced risk of infectious agents. 4. Fewer variables for quality control/quality assurance. 5. Increased control over bioreactor conditions. 6. Potential for increased antibody secretion. 7. Low or no dependence on animals. 8. Cost effective. 9. Overall enhanced efficiency Disadvantages of Serum Free Media in Hybridoma Cell Culture and Preparation of Monoclonal Antibodies- 1. Not all serum free media are applicable to all cell lines. 2. Cells may not grow to as high densities and may be more fragile than cells in serum 3. Media may take longer to prepare. Bypassing Hybridomas and Cloning of mab Genes The VH and VL genes for antibodies can be amplified through polymerase chain reaction (PCR) using 'universal primers' (universal primers will carry conserved sequences for most antibodies). By building restriction sites in the above primers, the amplified VH and VL genes can also be cloned directly for expression in mammalian cells or bacteria. The raw material for PCR may be hybridomas or B cells, which may be homogeneous (if derived from single cells) or heterogeneous. In the latter case, a variety of VH and VL genes will be amplified and will combine at random to produce as many as 106 clones for antibody genes (from 1000 different VH and 1000 different VL genes). These genes will be cloned in a phage and their products (particularly Fab fragments) can be screened for antigen binding activities. From such a large number of combinations in a combatorial library, it is very difficult to recover the original pairs of V genes (e.g. VHa.VLa or VHx.VLx is an original pair: VLy is a new combination VHa). However, the complexity may be reduced by using antigen-selected B lymphocytes (filters coated with antigen can be used for screening). (7) Designing and Building of mab Genes The antigen binding sites of antibodies have been studied in some detail in recent years. This led to modelling of entirely new antibodies, sometimes for their use as enzymes. This modelling through computer graphics can be used for alteration of antibody genes or for synthesis of entirely genes. These genes can be cloned and expressed in bacteria. The antibodies produced can be tested for their specificity and affinity for specific antigen. (7) Primary and Secondary Libraries for Antibody Genes In this method a repertoire of antibody genes can be prepared by using genes that can be obtained from a number of different sources including the following (i) Rearranged V genes from animals obtained through the use of PCR (with universal primers) (ii) New V genes obtained through gene conversion, a process adopted in birds (iii) Rearranged genes obtained from mrna through reverse transcription (iv) Designing entirely new V genes or D segments. The next step is to allow the expression of library in bacteria and screen antibodies for antigen binding activities. The International Journal of Pharmaceutical Sciences Review and Research Page 90
4 screening can be done on membrane filters coated with antigen. In future, the screening procedures may be replaced by methods of selection. In either case the selected VH and VL genes can be subjected to mutations to increase the affinity of an antibody for a specific antigen. A variety of methods for the above strategy are being developed, so that in future monoclonal antibodies will be produced without hybridomas and lymphocytes gens. MabCure hybridoma technology Classic Hybridoma technology is based on the generation of immortal hybrid cells (hybridoma), which follows the fusion of antibody-producing B-cells with myeloma tumor cells. Each hybridoma continuously manufactures a single type of ( monoclonal ) antibody. MabCure has "re-engineered" the classic hybridoma technology into a highly efficient and optimized one, overcoming the inherent limitations of the former, yet exploits its benefits. MabCure hybridoma technology exhibits several important advantages over competing technologies: 8 Prior identity of cancer antigen(s) is not required for generating Mabs against these antigens. MabCure's approach to creating anticancer Mabs is unbiased and enhances the discovery of new cancer markers. Antigens are preserved in their natural conformations; the resulting Mabs are able to better recognize and bind more selectively to tumor cells. Unlimited quantities of antibodies produced against a selected cancer target. A large number of antibodies is the key factor for obtaining an antibody repertoire which would exhibit both tumor specificity and tumor "universality". Cancer cells have an abundance of normal molecules (antigens) on their surface which are often "over-expressed" compared to normal cells and are also quite immunogenic, i.e. they can trigger robust immune response in the injected animal. In contrast, tumor-specific antigens (TSA) on the surface of the cancer cells are thought to be much less numerous and are often poorly immunogenic (i.e. triggering weak immune response).consequently, the vast majority of Mabs produced by the injected animals is directed against normal antigens while only a few target the TSA. Therefore, one has to be able to produce copious amounts of Mabs in order to find and select those few which are not only tumor-specific, but also "universal" for that type of cancer. 8 MabCure's proprietary technology is capable of generating specific monoclonal antibodies ( Mabs ) against a broad spectrum of antigens, including rare and poorly immunogenic antigens, such as those known as cancer markers, or tumor specific antigens ( TSA ), presumed to be present on the surface of cancer cells. 8 Mabs Monoclonal antibodies Drawing by Galia Karpol Peptide Synthesis for Monoclonal form. Custom monoclonal antibody production at Cell Essentials normally takes 4-6 months and is divided into 3 phases. The client has the option of halting the hybridoma production prior to the beginning of the next phase. Cell Essentials will not proceed to the next phase of the project without written ( ) confirmation from the client. The client is invoiced for each phase at its initiation. The amount of material needed for immunizations of 5 mice is 1mg to 1.5mg and an additional 0.8mg to 1mg is needed for ELISA screening. 9 Phase I: Immunization of 5 Balb/c mice including preinjection tail bleeds, antigen/adjuvant injections, all antigen/adjuvant boosts, tail bleeds and ELISA to determine highest titer mouse to be used for cell fusion. Time for this Phase is ~ 2 months and costs $1,800(USD). Phase II: Fusion of spleen cells with myeloma cell line, plating the fusion product into six 96 well plates, and determination of those wells expressing antibodies to the antigen by ELISA assay. All antibody-secreting colonies are isolated and transferred to a 24 well plate, expanded and a portion frozen. Antibody containing medium (3ml) of up to 12 ELISA-positive preclones* will be shipped to the client for evaluation in the client's specific application. Time for this Phase is ~ 3-4 weeks and costs $2,900(USD). 9 Phase III: Cloning of positive (IgG secreting) wells. A maximum of 5** of the positives selected by the client will be cloned by limiting dilution and isotyped. Clones will be expanded, and 10 vials of each selected clone will be frozen. Medium (10ml) from each clone as well as the frozen vials will be shipped to the client. Time for this Phase is ~ 1.5 to 3 months and costs $2,800(USD). 9 Total Cost per project is $7,500(USD). Additional clones can be isolated and processed as in Phase III for $350(USD)/Clone. The success of a custom hybridoma project is dependent upon the antigenicity of the material supplied by the client. Cell Essentials cannot guarantee that the material supplied by the client will produce an immune response sufficient to warrant proceeding to Phase II. Cell Essentials only guarantees that the antibodies produced by hybridomas developed recognize International Journal of Pharmaceutical Sciences Review and Research Page 91
5 the immunogen as assayed by ELISA. The client is responsible for determining the utility of these antibodies in their specific applications. All resulting hybridomas, their antibodies, and reagents supplied are solely the property of the client. At the completion of the project, these items will be returned to the client.9 Hybridoma cells are grown at high density in culture flasks or roller bottles. The antibody-containing conditioned medium is recovered, sterile filtered and frozen. Depending on the particular hybridoma, antibody concentration can vary greatly, but averages between 20 and 40 ug/ml. The cost is $1/ml (minimum order 100 ml). 10 Monoclonal production: CellCulture Hybridoma cells are grown at high density in culture flasks or roller bottles. The antibody-containing conditioned medium is recovered, sterile filtered and frozen. Depending on the particular hybridoma, antibody concentration can vary greatly, but averages between 20 and 40 ug/ml. The cost is $1/ml (minimum order 100 ml). 9 Bioreactor: Hybridoma cells are grown in dialysisbased mini-fermentors. This technology produces high density cultures that can be 20 to 30 times that achieved in static cell culture systems. Depending on the particular hybridoma these high density cultures on average produce antibody concentrations of 1 mg/ml. We harvest antibody rich medium over a 5 week period. The cost is $600(USD). 9 Price per amino Peptide Purity acid residue: Peptide Quantity >90% >95% 98% 20mg to 49mg $40 $45 Inquire 50mg to 100mg $50 $60 Inquire >100mg inquire inquire Inquire Minimum cost for peptide synthesis and purification is $400. For other amounts of peptides and/or purities, please contact us as listed above. 11 Included in the prices listed above are: 1.Synthesis set-up costs and synthesis of the peptide, 2. Reverse-phase HPLC 3. purification of all the crude peptide, 4. Peptide identification by Maldi- TOF mass spectrometer. 5. Analytical reverse-phase HPLC determination of peptide purity Copies of synthesis, HPLC and mass spectrometer reports, Additional costs: Shipping Peptide modifications (i.e., phosphopeptides, biotinylation, or other labels) Crude Sample Analysis Made Eseasy At Attana, we have developed a system that makes this possible. This system describe how it can be used in screening of antibodies and determininomateg their off rate in serum containing hybridoma supernant. The attana200 is dual channel biosensor for the automated analysis of biomolecular interactions. Incorporating quartz crystal microbalance (QCM) core technology.the system can be used to determining specificity, off rate, kinetics, affinity, active concentrations and thermodynamics using crude samples. The QCM core technology enables not only the study of biomolecules of varying species such as protein, nucleic acid and carbohydrate but also vastly different sizes, ranging from peptide to cells. QCM technology An antigen is initially immobilized on the sample surface, while an appropriate reference surface constructed. A new resonance frequency is registered. The antibodies then injected and binding of the antibodies to the surface bound antigen increases the mass further, whereupon a new shift in the resonance frequency is registered. If only a measurement of affinity (kd) is required, the interaction is allowed to reach equilibrium. To determine the off rate constant (kd), pure buffer is allowed to flow over the sensor surface, washing away unbound antibody.the surface is then regenerated leaving only immobilized antigen and is ready for injected of a new antibody. (12-18) Production of human antibodies: Human antibodies are currently produced by the following methods: (i) fusion of mouse myeloma cells with human lymphocytes (blast cells in peripheral blood lymphocytes) (ii) Immortalization of human cells by Epstein Barr virus. Both the methods have limitations. Human mouse hybrid cells have a tendency for preferential loss of human chromosomes, making them unstable. Similarly, Epstein-Barr virus does not allow preferential immortalization of blasts engaged in antibody response. In view of these difficulties, humanizing of rodent monoclonal antibodies through genetic engineering is the most practical approach, which is being evolved and used for the production of mouse human chimeric monoclonal antibodies to be used for tumour therapy or for manipulation of human immune system or against cell surface antigens. The humanized chimeric antibodies combine the rodent variable regions with the human constant (or constant + variable) framework regions. To further reduce the immunogenicity of rodent elements, humanized antibodies have been produced, which retain only the antigen binding complementarity determining regions (CDRs) from the rodent in association with human framework regions. In other approach transgenic mice carrying human genes for V, D, J and C regions have been produced. These can be used for the production of human antibodies directly by hyperimmunization Kenta s technology Kenta s technology for the generation and selection of an advanced class of fully human monoclonal antibodies International Journal of Pharmaceutical Sciences Review and Research Page 92
6 (MAbs) combines the advantages of classical hybridoma technology with a unique specific heteromyeloma fusion cell line. (24) Advantages (24) 1. Kenta has isolated and cultured human hybridomas secreting antigen specific antibodies of IgM, IgG, IgA, and IgE isotypes. 2. High affinity IgG antibodies are far more efficient in neutralising viruses and preventing infections. In contrast gram-negative bacteria are most efficiently attacked with IgM antibodies targeting the bacterial surface polysaccharides. 3. gm antibodies not only destroy bacterial cells but also promote the removal of bacterial debris from inflammed tissue, which will speed up healing of the affected tissue. Bound IgM antibodies activate the human complement system, which leads to the destruction of bacterial cells. Antibiotics used in hybridoma technology In order to evaluate the antigenic contribution of different regions of the penicillin molecule, monoclonal antibodies were raised against amoxicillin-protein conjugates and their specificities analysed in detail. A random sample of the clones produced was analysed by a quantitative inhibition-elisa, using, as inhibitors, monomeric conjugates of the following antibiotics to butylamine (BA), amoxicillin (AX), ampicillin (AMP), benzylpenicillin (BP). There was a high degree of cross reactivity with aminopenicillins, but low or absent cross reactivity with BP. None of the antibodies recognized the thiazolidine ring or the conjugated nuclear region of the penicillins. Non Hodgkin s lymphoma- Rituxan Brest cancer- herceptine. Inflammatory disease- Infliximab. Allergic asthma omalizumab In type -1 diabetes- muromunab. Clumping of platelet-abiciximab. Hybridoma secreting MAbs against 17 hydroxyprogesterone (17OHP) have been generated. Uses of monoclonal antibodies Monoclonal antibodies or specific antibodies, arc now an essential tool of much biomedical research and are of great commercial and medical value. For instance, ABO blood groups could be earlier identified with the help of human sera carrying antibodies of known specificity. These human sera in U.K. have been replaced by monoclonal antibodies produced by hybridomas, for the identification of ABO blood groups. Thus the diagnostic and screening value of the monoclonal antibodies through serological tests has been demonstrated. Besides the use of monoclonal in identification of blood groups in UK (UK blood typing), following three uses for monoclonal are described, although, only the first two of these make a definite market at present: (I) Diagnosis (including ELISA test for detection of viruses and imaging), (ii) Immunopurification (iii) Therapy. CONCLUSION In diagnosis, pregnancy can be detected by assaying of hormones with monoclonal. Similarly, pathogens can be detected in a few hours sparing several days of culturing of cells earlier needed. Immunopurification involves separation of one substance from a mixture of very similar molecules. The Company s patented technology has the capacity to address major industrial needs for a faster, lower cost and better quality process. Many of the steps in making hybridomas are similar to those involved with NeoClone s ABL-MYC technology. Antibodies are proteins synthesized in blood against specific antigens just to combat and give immunity in blood. They can be collected from the blood serum of an animal. Such antibodies are heterogeneous and contain a mixture of antibodies (i.e., monoclonal antibodies). Therefore, they do not have characteristics of specificity. If a specific lymphocyte, after the isolation and culture in vitro, the becomes capable of producing a single type of antibody which bears specificity against specific antigen. It is known as monoclonal antibodies. Due to the presence of desired immunity, monoclonal antibodies are used in the diagnosis of diseases. International Journal of Pharmaceutical Sciences Review and Research Page 93
7 REFERENCES 1. Bretton, PR, Melamed, MR, Fair, WR, Cote, RJ (1994). Detection of occult micrometastases in the bone marrow of patients with prostate carcinoma. Prostate. 25(2), #Method 3. Franklin, WA, Shpall, EJ, Archer, P, Johnston, CS, Garza-Williams, S, Hami, L, Bitter MA, Bast RC, Jones, RB (1996). Immunocytochemical detection of breast cancer cells in marrow and peripheral blood of patients undergoing high dose chemotherapy with autologous stem cell support. Breast Cancer Res Treat. 41(1), Ghosh, AK, Spriggs, Al, Taylor-papadimitriou, J and Mason, DY(1983). Immunocytochemical staining of cells in pleural and peritoneal effusions with a panel of monoclonal antibodies. J Clin Pathol, 36, Kvalheim, G (1996). Detection of occult tumour cells in bone marrow and blood in breast cancer patients methods and clinical significance. Acta Oncol, 35, doma-and-monoclonal-antibodies-mabs/ raising-theantibodies.htm 7. Elements of biotechnology, P.K. GUPTA (1 st EDITION): , ( ) 8. Laboratory for Plague Microbiology, Department of Infectious Diseases, State Research Center for Applied Microbiology and Biotechnology, Obolensk, Serpukhov District, Moscow Region, Russia 9. Roilides E, Pizzo PA. Modulation of host defenses by cytokines: evolving adjuncts in prevention and treatment of serious infections in immuno compromised hosts. Clin Infect Dis 1992;15: Hybridoma Techniques 1980 EMBO, SKMB Course, Basel. Cold Spring Harbor, NY: Cold Spring Harbor Laboratory. 11. Mackenzie, Michael, Alberto Cambrosio, and Peter Keating 1987 Scientific information and intellectual property: the case of the hybndoma/monoclonal antibody technique. Paper presented at the 1987 meeting of the Society for Social Studies of Science, Worcester, MA. 12. Knorr-Cetina, Karin D the Manufacture of Knowledge. New York: Pergamon Press. 13. Knorr.Cetina, Karin D. and Michael Mulkay, eds Science Observed: Perspectives on the Social Study of Science. London: Sage. 14. Latour, Bruno and Steve Woolgar 1979 Laboratory Life: The Social Construction of Facts. Beverly Hills, CA: Sage 15. Callon, Michel and Bruno Latour 1986 Les paradoxes de la modernité: comment concevoir les innovation? Prospective et Sante Hiver: Lynch, Michael 1985 Art and Artifact in Laboratory Science. London: Routledge & Kegan Paul. 17. Lynch, Michael 1982 Technical Work and Critical Inquiry: Investigations in a Scientific Laboratory. Social Studies of Science 12: Art To Science In Tissue Culture 1983 Cell fusion with P.E.G.: is it reproducible? Art to Science in Tissue Culture 2: Mumford RS, Murphy TV. Antimicrobial resistance in Streptococcus pneumoniae: can immunization prevent its spread. J Invest Med 1994; 42: Canadian Food Inspection Agency, Animal Diseases Research Institute, P.O. 640, Township Road 9-1, Lethbridge, AB T1J 3Z4, Canada 21. C:\Documents and Settings\Admin\My Documents \Cell Essentials.mht IPT_29.pdf 23. C:\Documents and Settings\Destiny\My Documents \History.htm 24. C:\Documents and Settings\Destiny\My Documents \humane hybridoma technology Kenta Biotech.htm *********** International Journal of Pharmaceutical Sciences Review and Research Page 94
The production of monoclonal antibodies using the hybridoma technology
 The production of monoclonal antibodies using the hybridoma technology Sbonelo Hadebe 211554629 October 16, 2015 Contents 1 Introduction 1 2 Theory 2 2.1 Monoclonal antibodies...................... 2 2.2
The production of monoclonal antibodies using the hybridoma technology Sbonelo Hadebe 211554629 October 16, 2015 Contents 1 Introduction 1 2 Theory 2 2.1 Monoclonal antibodies...................... 2 2.2
SCANTIBODIES Laboratory, Inc. Contract Monoclonal Antibody Production
 A Technical Publication of SCANTIBODIES Laboratory, Inc. Volume 1 Number 4 9336 Abraham Way Santee, CA 92071 USA (619) 258-9300 fax (619) 258-9366 www.scantibodies.com SCANTIBODIES Laboratory, Inc. Contract
A Technical Publication of SCANTIBODIES Laboratory, Inc. Volume 1 Number 4 9336 Abraham Way Santee, CA 92071 USA (619) 258-9300 fax (619) 258-9366 www.scantibodies.com SCANTIBODIES Laboratory, Inc. Contract
Chapter 18: Applications of Immunology
 Chapter 18: Applications of Immunology 1. Vaccinations 2. Monoclonal vs Polyclonal Ab 3. Diagnostic Immunology 1. Vaccinations What is Vaccination? A method of inducing artificial immunity by exposing
Chapter 18: Applications of Immunology 1. Vaccinations 2. Monoclonal vs Polyclonal Ab 3. Diagnostic Immunology 1. Vaccinations What is Vaccination? A method of inducing artificial immunity by exposing
Applications of Ab Molecules. Chapter 4 Monoclonal Ab (p.99) Chapter 5 Ab genes and Ab Engineering (p.128)
 Applications of Ab Molecules Chapter 4 Monoclonal Ab (p.99) Chapter 5 Ab genes and Ab Engineering (p.128) Monoclonal Antibodies Clonal Selection of B Lymphocytes Hybridoma Köhler and Milsten (1975) - continuous
Applications of Ab Molecules Chapter 4 Monoclonal Ab (p.99) Chapter 5 Ab genes and Ab Engineering (p.128) Monoclonal Antibodies Clonal Selection of B Lymphocytes Hybridoma Köhler and Milsten (1975) - continuous
Custom Antibody Services
 Custom Antibody Services Custom service offerings DNA sequence Plasmid Peptide Structure Protein Peptide Small molecule Cells Spleen Lymphocytes Antigen Preparation Immunization Fusion & Subcloning Expansion
Custom Antibody Services Custom service offerings DNA sequence Plasmid Peptide Structure Protein Peptide Small molecule Cells Spleen Lymphocytes Antigen Preparation Immunization Fusion & Subcloning Expansion
Chapter 3.2» Custom Monoclonal
 198 3 3.2 Custom Monoclonal 199 Mouse monoclonal antibody development Chapter 3.2» Custom Monoclonal 200 In vitro monoclonals expression service 201 Mouse monoclonal antibody additional services 202 Magnetic
198 3 3.2 Custom Monoclonal 199 Mouse monoclonal antibody development Chapter 3.2» Custom Monoclonal 200 In vitro monoclonals expression service 201 Mouse monoclonal antibody additional services 202 Magnetic
MONOCLONAL ANTIBODY PRODUCTION
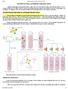 MONOCLONAL ANTIBODY PRODUCTION Antibodies having single specificity produced from a single clone of B cell are referred as Mono clonal antibodies (MAbs). In 1975, Georges Köhler and Cesar Milstein devised
MONOCLONAL ANTIBODY PRODUCTION Antibodies having single specificity produced from a single clone of B cell are referred as Mono clonal antibodies (MAbs). In 1975, Georges Köhler and Cesar Milstein devised
Antibody Function & Structure
 Antibody Function & Structure Specifically bind to antigens in both the recognition phase (cellular receptors) and during the effector phase (synthesis and secretion) of humoral immunity Serology: the
Antibody Function & Structure Specifically bind to antigens in both the recognition phase (cellular receptors) and during the effector phase (synthesis and secretion) of humoral immunity Serology: the
3 months 1.5 months 1.5 months. 1 month
 Rabbit monoclonal antibody (Mab) is secreted by the plasma B-cell of the rabbit. Traditional generation of rabbit Mab relies on a rabbit myeloma for B- cell fusion (
Rabbit monoclonal antibody (Mab) is secreted by the plasma B-cell of the rabbit. Traditional generation of rabbit Mab relies on a rabbit myeloma for B- cell fusion (
Antigens & Antibodies II. Polyclonal antibodies vs Monoclonal antibodies
 A Brief Review of Antibody Structure A Brief Review of Antibody Structure The basic antibody is a dimer of dimer (2 heavy chain-light chain pairs) composed of repeats of a single structural unit known
A Brief Review of Antibody Structure A Brief Review of Antibody Structure The basic antibody is a dimer of dimer (2 heavy chain-light chain pairs) composed of repeats of a single structural unit known
MAB Solut. MABSolys Génopole Campus 1 5 rue Henri Desbruères 91030 Evry Cedex. www.mabsolut.com. is involved at each stage of your project
 Mabsolus-2015-UK:Mise en page 1 03/07/15 14:13 Page1 Services provider Department of MABSolys from conception to validation MAB Solut is involved at each stage of your project Creation of antibodies Production
Mabsolus-2015-UK:Mise en page 1 03/07/15 14:13 Page1 Services provider Department of MABSolys from conception to validation MAB Solut is involved at each stage of your project Creation of antibodies Production
Antibody Services. Best Guarantees in the Industry! Monoclonal Antibody Services. Polyclonal Antibody Services. Express Antibody TM Services
 Antibody Services Best Guarantees in the Industry! Monoclonal Antibody Services Polyclonal Antibody Services Express Antibody TM Services Guaranteed Antibody Services Antibody Purification Services Your
Antibody Services Best Guarantees in the Industry! Monoclonal Antibody Services Polyclonal Antibody Services Express Antibody TM Services Guaranteed Antibody Services Antibody Purification Services Your
KMS-Specialist & Customized Biosimilar Service
 KMS-Specialist & Customized Biosimilar Service 1. Polyclonal Antibody Development Service KMS offering a variety of Polyclonal Antibody Services to fit your research and production needs. we develop polyclonal
KMS-Specialist & Customized Biosimilar Service 1. Polyclonal Antibody Development Service KMS offering a variety of Polyclonal Antibody Services to fit your research and production needs. we develop polyclonal
1.Gene Synthesis. 2.Peptide & Phospho-P. Assembly PCR. Design & Synthesis. Advantages. Specifications. Advantages
 1.Gene Synthesis Assembly PCR Looking for a cdna for your research but could not fish out the gene through traditional cloning methods or a supplier? Abnova provides a gene synthesis service via assembly
1.Gene Synthesis Assembly PCR Looking for a cdna for your research but could not fish out the gene through traditional cloning methods or a supplier? Abnova provides a gene synthesis service via assembly
specific B cells Humoral immunity lymphocytes antibodies B cells bone marrow Cell-mediated immunity: T cells antibodies proteins
 Adaptive Immunity Chapter 17: Adaptive (specific) Immunity Bio 139 Dr. Amy Rogers Host defenses that are specific to a particular infectious agent Can be innate or genetic for humans as a group: most microbes
Adaptive Immunity Chapter 17: Adaptive (specific) Immunity Bio 139 Dr. Amy Rogers Host defenses that are specific to a particular infectious agent Can be innate or genetic for humans as a group: most microbes
Aviva Systems Biology
 Aviva Custom Antibody Service and Price Mouse Monoclonal Antibody Service Package Number Description Package Contents Time Price Customer provides antigen protein $6,174 Monoclonal package1 (From protein
Aviva Custom Antibody Service and Price Mouse Monoclonal Antibody Service Package Number Description Package Contents Time Price Customer provides antigen protein $6,174 Monoclonal package1 (From protein
Your partner in immunology
 Your partner in immunology Expertise Expertise Reactivity Reactivity Quality Quality Advice Advice Who are we? Specialist of antibody engineering Covalab is a French biotechnology company, specialised
Your partner in immunology Expertise Expertise Reactivity Reactivity Quality Quality Advice Advice Who are we? Specialist of antibody engineering Covalab is a French biotechnology company, specialised
Advanced BioDesign Outlines Solutions. Antibody Overview. by Advanced BioDesign. Project Start. Immunogenicity. Selecting Your Antigen
 Advanced BioDesign Outlines Solutions by Advanced BioDesign Antibody Overview Launching an immunisation programme is an important experimental step that needs care. With Advanced BioDesign, you may develop
Advanced BioDesign Outlines Solutions by Advanced BioDesign Antibody Overview Launching an immunisation programme is an important experimental step that needs care. With Advanced BioDesign, you may develop
Microbiology AN INTRODUCTION EIGHTH EDITION
 TORTORA FUNKE CASE Microbiology AN INTRODUCTION EIGHTH EDITION Differentiate between innate and acquired immunity. Chapter 17 Specific Defenses of the Host: The Immune Response B.E Pruitt & Jane J. Stein
TORTORA FUNKE CASE Microbiology AN INTRODUCTION EIGHTH EDITION Differentiate between innate and acquired immunity. Chapter 17 Specific Defenses of the Host: The Immune Response B.E Pruitt & Jane J. Stein
Basics of Immunology
 Basics of Immunology 2 Basics of Immunology What is the immune system? Biological mechanism for identifying and destroying pathogens within a larger organism. Pathogens: agents that cause disease Bacteria,
Basics of Immunology 2 Basics of Immunology What is the immune system? Biological mechanism for identifying and destroying pathogens within a larger organism. Pathogens: agents that cause disease Bacteria,
CUSTOM ANTIBODIES. Fully customised services: rat and murine monoclonals, rat and rabbit polyclonals, antibody characterisation, antigen preparation
 CUSTOM ANTIBODIES Highly competitive pricing without compromising quality. Rat monoclonal antibodies for the study of gene expression and proteomics in mice and in mouse models of human diseases available.
CUSTOM ANTIBODIES Highly competitive pricing without compromising quality. Rat monoclonal antibodies for the study of gene expression and proteomics in mice and in mouse models of human diseases available.
Types, production of antibodies and Antibody/antigen interaction
 Types, production of antibodies and Antibody/antigen interaction Antibodies Secreted by B lymphocytes Great diversity and specificity: >109 different antibodies; can distinguish between very similar molecules
Types, production of antibodies and Antibody/antigen interaction Antibodies Secreted by B lymphocytes Great diversity and specificity: >109 different antibodies; can distinguish between very similar molecules
HuCAL Custom Monoclonal Antibodies
 HuCAL Custom Monoclonal HuCAL Custom Monoclonal Antibodies Highly Specific, Recombinant Antibodies in 8 Weeks Highly Specific Monoclonal Antibodies in Just 8 Weeks HuCAL PLATINUM (Human Combinatorial Antibody
HuCAL Custom Monoclonal HuCAL Custom Monoclonal Antibodies Highly Specific, Recombinant Antibodies in 8 Weeks Highly Specific Monoclonal Antibodies in Just 8 Weeks HuCAL PLATINUM (Human Combinatorial Antibody
Activation and effector functions of HMI
 Activation and effector functions of HMI Hathairat Thananchai, DPhil Department of Microbiology Faculty of Medicine Chiang Mai University 25 August 2015 ว ตถ ประสงค หล งจากช วโมงบรรยายน แล วน กศ กษาสามารถ
Activation and effector functions of HMI Hathairat Thananchai, DPhil Department of Microbiology Faculty of Medicine Chiang Mai University 25 August 2015 ว ตถ ประสงค หล งจากช วโมงบรรยายน แล วน กศ กษาสามารถ
STANDARD OPERATING PROCEDURE
 STANDARD OPERATING PROCEDURE Title: Antibody Production at Strategic Diagnostics Inc. SOP#: M-119 Version #: 1 Date Approved: August 6, 2009 Author: Strategic Diagnostic Inc. Date Modified: 1. PURPOSE
STANDARD OPERATING PROCEDURE Title: Antibody Production at Strategic Diagnostics Inc. SOP#: M-119 Version #: 1 Date Approved: August 6, 2009 Author: Strategic Diagnostic Inc. Date Modified: 1. PURPOSE
GenScript Antibody Services
 GenScript Antibody Services Scientific experts, innovative technologies, proven performance We specialize in custom antibody production to empower your research Custom Polyclonal Antibody Production Custom
GenScript Antibody Services Scientific experts, innovative technologies, proven performance We specialize in custom antibody production to empower your research Custom Polyclonal Antibody Production Custom
Chapter 2 Antibodies. Contents. Introduction
 Chapter 2 Antibodies Keywords Immunohistochemistry Antibody labeling Fluorescence microscopy Fluorescent immunocytochemistry Fluorescent immunohistochemistry Indirect immunocytochemistry Immunostaining
Chapter 2 Antibodies Keywords Immunohistochemistry Antibody labeling Fluorescence microscopy Fluorescent immunocytochemistry Fluorescent immunohistochemistry Indirect immunocytochemistry Immunostaining
HuCAL Custom Monoclonal Antibodies
 HuCAL Custom Monoclonal Antibodies Highly Specific Monoclonal Antibodies in just 8 Weeks PROVEN, HIGHLY SPECIFIC, HIGH AFFINITY ANTIBODIES IN 8 WEEKS WITHOUT HuCAL PLATINUM IMMUNIZATION (Human Combinatorial
HuCAL Custom Monoclonal Antibodies Highly Specific Monoclonal Antibodies in just 8 Weeks PROVEN, HIGHLY SPECIFIC, HIGH AFFINITY ANTIBODIES IN 8 WEEKS WITHOUT HuCAL PLATINUM IMMUNIZATION (Human Combinatorial
How To Make A Hybridoma
 Heddy Zola, Child Health Research Institute, Adelaide, Australia Monoclonal antibodies are antibodies with a unique specificity, generally made by cloning cells containing a particular antibody gene to
Heddy Zola, Child Health Research Institute, Adelaide, Australia Monoclonal antibodies are antibodies with a unique specificity, generally made by cloning cells containing a particular antibody gene to
Geniron. Custom Antibody Services. Serum services Antibody Monoclonal. Purification Antibody Mono Y Genetic Immunization Genbody Polyclonal Antibody
 Geniron Custom Antibody Services Serum services Antibody Monoclonal Purification Antibody Mono Y Genetic Immunization Genbody Polyclonal Antibody Geniron Poly Y WE PROVIDE OUR SERVICES TO With Expertise
Geniron Custom Antibody Services Serum services Antibody Monoclonal Purification Antibody Mono Y Genetic Immunization Genbody Polyclonal Antibody Geniron Poly Y WE PROVIDE OUR SERVICES TO With Expertise
In Vitro And In Vivo Production Of Antibodies
 In Vitro And In Vivo Production Of Antibodies OCTOBER 27, 2015 BY ADMIN Overview An antibody is a protein normally produced by the B cells of the immune system. Their original purpose is to identify and
In Vitro And In Vivo Production Of Antibodies OCTOBER 27, 2015 BY ADMIN Overview An antibody is a protein normally produced by the B cells of the immune system. Their original purpose is to identify and
ELISA BIO 110 Lab 1. Immunity and Disease
 ELISA BIO 110 Lab 1 Immunity and Disease Introduction The principal role of the mammalian immune response is to contain infectious disease agents. This response is mediated by several cellular and molecular
ELISA BIO 110 Lab 1 Immunity and Disease Introduction The principal role of the mammalian immune response is to contain infectious disease agents. This response is mediated by several cellular and molecular
Name (print) Name (signature) Period. (Total 30 points)
 AP Biology Worksheet Chapter 43 The Immune System Lambdin April 4, 2011 Due Date: Thurs. April 7, 2011 You may use the following: Text Notes Power point Internet One other person in class "On my honor,
AP Biology Worksheet Chapter 43 The Immune System Lambdin April 4, 2011 Due Date: Thurs. April 7, 2011 You may use the following: Text Notes Power point Internet One other person in class "On my honor,
About Our Products. Blood Products. Purified Infectious/Inactivated Agents. Native & Recombinant Viral Proteins. DNA Controls and Primers for PCR
 About Our Products Purified Infectious/Inactivated Agents ABI produces a variety of specialized reagents, allowing researchers to choose the best preparations for their studies. Available reagents include
About Our Products Purified Infectious/Inactivated Agents ABI produces a variety of specialized reagents, allowing researchers to choose the best preparations for their studies. Available reagents include
Elabscience Biotechnology Co.,Ltd
 Biotechnology Co.,Ltd Your one-stop custom antibody service Antibody is a kind of protein that is the most widely used in the fields of biology and medicine. Antibodies, as the preparations for disease
Biotechnology Co.,Ltd Your one-stop custom antibody service Antibody is a kind of protein that is the most widely used in the fields of biology and medicine. Antibodies, as the preparations for disease
Monoclonal Antibody. By Dr. Adel Gabr
 Monoclonal Antibody By Dr. Adel Gabr Terminology Immunotherapy Monoclonal antibody Biotherapy Biological therapy Target therapy TKIs Cancer vaccine Types of targeted therapy used today Today many different
Monoclonal Antibody By Dr. Adel Gabr Terminology Immunotherapy Monoclonal antibody Biotherapy Biological therapy Target therapy TKIs Cancer vaccine Types of targeted therapy used today Today many different
The Use of Antibodies in Immunoassays
 TECHNICAL NOTE The Use of Antibodies in Immunoassays Introduction Structure of an IgG Antibody Immunological reagents are the backbone of every immunoassay system. Immunoassays can be utilized to quantitatively
TECHNICAL NOTE The Use of Antibodies in Immunoassays Introduction Structure of an IgG Antibody Immunological reagents are the backbone of every immunoassay system. Immunoassays can be utilized to quantitatively
Antibody Production Price List
 Antibody Production Price List Presenting Insight Biotechnology s price list for custom polyclonal and monoclonal antibody production services. We are happy to tailor individual packages towards the specific
Antibody Production Price List Presenting Insight Biotechnology s price list for custom polyclonal and monoclonal antibody production services. We are happy to tailor individual packages towards the specific
DELFIA assays bring convenience in monoclonal antibody development
 DELFIA DELFIA assays bring convenience in monoclonal development INTRODUCTION The DELFIA system provides a method of discovering specific, high-affinity monoclonal antibodies (Mabs) even in low concentrations
DELFIA DELFIA assays bring convenience in monoclonal development INTRODUCTION The DELFIA system provides a method of discovering specific, high-affinity monoclonal antibodies (Mabs) even in low concentrations
CHAPTER 8 IMMUNOLOGICAL IMPLICATIONS OF PEPTIDE CARBOHYDRATE MIMICRY
 CHAPTER 8 IMMUNOLOGICAL IMPLICATIONS OF PEPTIDE CARBOHYDRATE MIMICRY Immunological Implications of Peptide-Carbohydrate Mimicry 8.1 Introduction The two chemically dissimilar molecules, a peptide (12mer)
CHAPTER 8 IMMUNOLOGICAL IMPLICATIONS OF PEPTIDE CARBOHYDRATE MIMICRY Immunological Implications of Peptide-Carbohydrate Mimicry 8.1 Introduction The two chemically dissimilar molecules, a peptide (12mer)
GenScript Antibody Services
 GenScript Antibody Services Scientific experts, innovative technologies, proven performance We specialize in custom antibody production to empower your research Custom Polyclonal Antibody Production Custom
GenScript Antibody Services Scientific experts, innovative technologies, proven performance We specialize in custom antibody production to empower your research Custom Polyclonal Antibody Production Custom
Chapter 3. Immunity and how vaccines work
 Chapter 3 Immunity and how vaccines work 3.1 Objectives: To understand and describe the immune system and how vaccines produce immunity To understand the differences between Passive and Active immunity
Chapter 3 Immunity and how vaccines work 3.1 Objectives: To understand and describe the immune system and how vaccines produce immunity To understand the differences between Passive and Active immunity
Bio-manufacturing of antigens, antibodies and bioresearch materials
 Bio-manufacturing of antigens, antibodies and bioresearch materials Foetal Bovine Serum (FBS) Human AB Serum Chick Embryo Extract (CEE) Custom polyclonal antibody production Custom monoclonal antibody
Bio-manufacturing of antigens, antibodies and bioresearch materials Foetal Bovine Serum (FBS) Human AB Serum Chick Embryo Extract (CEE) Custom polyclonal antibody production Custom monoclonal antibody
CCR Biology - Chapter 9 Practice Test - Summer 2012
 Name: Class: Date: CCR Biology - Chapter 9 Practice Test - Summer 2012 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Genetic engineering is possible
Name: Class: Date: CCR Biology - Chapter 9 Practice Test - Summer 2012 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Genetic engineering is possible
Interim Progress Report R&D Project 348. Development of a Field Test Kit for Detection of Blue-Green Algal Toxins
 Interim Progress Report R&D Project 348 Development of a Field Test Kit for Detection of Blue-Green Algal Toxins Biocode Limited November 1992 R&D 348/04/A ENVIRONMENT AGENCY 135357 CONTENTS SUMMARY KEYWORDS
Interim Progress Report R&D Project 348 Development of a Field Test Kit for Detection of Blue-Green Algal Toxins Biocode Limited November 1992 R&D 348/04/A ENVIRONMENT AGENCY 135357 CONTENTS SUMMARY KEYWORDS
Chapter 5: Organization and Expression of Immunoglobulin Genes
 Chapter 5: Organization and Expression of Immunoglobulin Genes I. Genetic Model Compatible with Ig Structure A. Two models for Ab structure diversity 1. Germ-line theory: maintained that the genome contributed
Chapter 5: Organization and Expression of Immunoglobulin Genes I. Genetic Model Compatible with Ig Structure A. Two models for Ab structure diversity 1. Germ-line theory: maintained that the genome contributed
Some terms: An antigen is a molecule or pathogen capable of eliciting an immune response
 Overview of the immune system We continue our discussion of protein structure by considering the structure of antibodies. All organisms are continually subject to attack by microorganisms and viruses.
Overview of the immune system We continue our discussion of protein structure by considering the structure of antibodies. All organisms are continually subject to attack by microorganisms and viruses.
Custom Antibody Services
 prosci-inc.com Custom Antibody Services High Performance Antibodies and More Broad Antibody Catalog Extensive Antibody Services CUSTOM ANTIBODY SERVICES Established in 1998, ProSci Incorporated is a leading
prosci-inc.com Custom Antibody Services High Performance Antibodies and More Broad Antibody Catalog Extensive Antibody Services CUSTOM ANTIBODY SERVICES Established in 1998, ProSci Incorporated is a leading
Mouse IFN-gamma ELISpot Kit
 Page 1 of 8 Mouse IFN-gamma ELISpot Kit Without Plates With Plates With Sterile Plates Quantity Catalog Nos. 862.031.001 862.031.001P 862.031.001S 1 x 96 tests 862.031.005 862.031.005P 862.031.005S 5 x
Page 1 of 8 Mouse IFN-gamma ELISpot Kit Without Plates With Plates With Sterile Plates Quantity Catalog Nos. 862.031.001 862.031.001P 862.031.001S 1 x 96 tests 862.031.005 862.031.005P 862.031.005S 5 x
Chapter 6: Antigen-Antibody Interactions
 Chapter 6: Antigen-Antibody Interactions I. Strength of Ag-Ab interactions A. Antibody Affinity - strength of total noncovalent interactions between single Ag-binding site on an Ab and a single epitope
Chapter 6: Antigen-Antibody Interactions I. Strength of Ag-Ab interactions A. Antibody Affinity - strength of total noncovalent interactions between single Ag-binding site on an Ab and a single epitope
Human hybridoma technology for the production of monoclonal antibodies
 Human hybridoma technology for the production of monoclonal antibodies A new method of creating cell lines has been developed for the production of "totally human" monoclonal antibodies. Professor Hans
Human hybridoma technology for the production of monoclonal antibodies A new method of creating cell lines has been developed for the production of "totally human" monoclonal antibodies. Professor Hans
Immuno-Oncology Therapies to Treat Lung Cancer
 Immuno-Oncology Therapies to Treat Lung Cancer What you need to know ONCHQ14NP07519 Introduction: Immuno-oncology represents an innovative approach to cancer research that seeks to harness the body s own
Immuno-Oncology Therapies to Treat Lung Cancer What you need to know ONCHQ14NP07519 Introduction: Immuno-oncology represents an innovative approach to cancer research that seeks to harness the body s own
Generating Monoclonal Antibodies
 CHAPTER 7 Generating Monoclonal Antibodies Edward A. Greenfield Dana-Farber Cancer Institute, Boston, Massachusetts 02115 INTRODUCTION Characteristics of Monoclonal Antibodies, 203 Production of Monoclonal
CHAPTER 7 Generating Monoclonal Antibodies Edward A. Greenfield Dana-Farber Cancer Institute, Boston, Massachusetts 02115 INTRODUCTION Characteristics of Monoclonal Antibodies, 203 Production of Monoclonal
Recombinant DNA and Biotechnology
 Recombinant DNA and Biotechnology Chapter 18 Lecture Objectives What Is Recombinant DNA? How Are New Genes Inserted into Cells? What Sources of DNA Are Used in Cloning? What Other Tools Are Used to Study
Recombinant DNA and Biotechnology Chapter 18 Lecture Objectives What Is Recombinant DNA? How Are New Genes Inserted into Cells? What Sources of DNA Are Used in Cloning? What Other Tools Are Used to Study
ELITE Custom Antibody Services
 ELITE Custom Antibody Services ELITE Custom Antibody Services Experience, confidence, and understanding As a manufacturer and service provider, we have the experience, confidence, and understanding to
ELITE Custom Antibody Services ELITE Custom Antibody Services Experience, confidence, and understanding As a manufacturer and service provider, we have the experience, confidence, and understanding to
Custom Polyclonal Anti-Peptide Antibody, Brochure
 Custom Polyclonal Anti-Peptide Antibody, Brochure Interest in any of the products, request or order them at Bio-Connect. Bio-Connect B.V. T NL +31 (0)26 326 44 50 T BE +32 (0)2 503 03 48 Begonialaan 3a
Custom Polyclonal Anti-Peptide Antibody, Brochure Interest in any of the products, request or order them at Bio-Connect. Bio-Connect B.V. T NL +31 (0)26 326 44 50 T BE +32 (0)2 503 03 48 Begonialaan 3a
The Body s Defenses CHAPTER 24
 CHAPTER 24 The Body s Defenses PowerPoint Lectures for Essential Biology, Third Edition Neil Campbell, Jane Reece, and Eric Simon Essential Biology with Physiology, Second Edition Neil Campbell, Jane Reece,
CHAPTER 24 The Body s Defenses PowerPoint Lectures for Essential Biology, Third Edition Neil Campbell, Jane Reece, and Eric Simon Essential Biology with Physiology, Second Edition Neil Campbell, Jane Reece,
OBTAINING MONOCLONAL ANTIBODIES TO EPITOPES OF IVERMECTIN
 С.Сейфуллин атындағы Қазақ агротехникалық университетінің Ғылым жаршысы / Вестник науки Казахского агротехнического университета им. С. Сейфуллина. 2013. - 4 (79). C.8-14 OBTAINING MONOCLONAL ANTIBODIES
С.Сейфуллин атындағы Қазақ агротехникалық университетінің Ғылым жаршысы / Вестник науки Казахского агротехнического университета им. С. Сейфуллина. 2013. - 4 (79). C.8-14 OBTAINING MONOCLONAL ANTIBODIES
TABLE OF CONTENT. Page ACKNOWLEDGEMENTS. iii ENGLISH ABSTRACT THAI ABSTRACT. vii LIST OF TABLES LIST OF FIGURES. xvi ABBREVIATIONS.
 x TABLE OF CONTENT ACKNOWLEDGEMENTS ENGLISH ABSTRACT THAI ABSTRACT LIST OF TABLES LIST OF FIGURES ABBREVIATIONS iii iv vii xv xvi xviii CHAPTER I: INTRODUCTION 1.1 Statement of problems 1 1.2 Literature
x TABLE OF CONTENT ACKNOWLEDGEMENTS ENGLISH ABSTRACT THAI ABSTRACT LIST OF TABLES LIST OF FIGURES ABBREVIATIONS iii iv vii xv xvi xviii CHAPTER I: INTRODUCTION 1.1 Statement of problems 1 1.2 Literature
Dot Blot Analysis. Teacher s Guidebook. (Cat. # BE 502) think proteins! think G-Biosciences www.gbiosciences.com
 PR110 G-Biosciences 1-800-628-7730 1-314-991-6034 [email protected] A Geno Technology, Inc. (USA) brand name Dot Blot Analysis Teacher s Guidebook (Cat. # BE 502) think proteins! think G-Biosciences
PR110 G-Biosciences 1-800-628-7730 1-314-991-6034 [email protected] A Geno Technology, Inc. (USA) brand name Dot Blot Analysis Teacher s Guidebook (Cat. # BE 502) think proteins! think G-Biosciences
27E10: A unique monoclonal antibody against MRP8/14 (S100A8/A9, Calprotectin)
 BMA BIOMEDICALS Rheinstrasse 28-32 CH-4302 Augst (Switzerland) Phone:+41 61 811 6222 Fax: +41 61 811 6006 [email protected] www.bma.ch 27E10: A unique monoclonal antibody against MRP8/14 (S100A8/A9, Calprotectin)
BMA BIOMEDICALS Rheinstrasse 28-32 CH-4302 Augst (Switzerland) Phone:+41 61 811 6222 Fax: +41 61 811 6006 [email protected] www.bma.ch 27E10: A unique monoclonal antibody against MRP8/14 (S100A8/A9, Calprotectin)
CHAPTER 6 ANTIBODY GENETICS: ISOTYPES, ALLOTYPES, IDIOTYPES
 CHAPTER 6 ANTIBODY GENETICS: ISOTYPES, ALLOTYPES, IDIOTYPES See APPENDIX: (3) OUCHTERLONY; (4) AFFINITY CHROMATOGRAPHY Human immunoglobulins are made up of LIGHT and HEAVY chains encoded by a total of
CHAPTER 6 ANTIBODY GENETICS: ISOTYPES, ALLOTYPES, IDIOTYPES See APPENDIX: (3) OUCHTERLONY; (4) AFFINITY CHROMATOGRAPHY Human immunoglobulins are made up of LIGHT and HEAVY chains encoded by a total of
Aviva Systems Biology
 Aviva Custom Antibody Services and Prices Rabbit Polyclonal Antibody Service Package Number Description Package Contents Time Price Polyclonal package 1 (From protein to antiserum) Polyclonal package 2
Aviva Custom Antibody Services and Prices Rabbit Polyclonal Antibody Service Package Number Description Package Contents Time Price Polyclonal package 1 (From protein to antiserum) Polyclonal package 2
From cells to therapeuticsvivalis. Humalex Fully human antibody discovery platform An integrated therapeutic development offering
 From cells to therapeuticsvivalis Humalex Fully human antibody discovery platform An integrated therapeutic development offering Humalex from bench-top to clinical development The Humalex platform enables
From cells to therapeuticsvivalis Humalex Fully human antibody discovery platform An integrated therapeutic development offering Humalex from bench-top to clinical development The Humalex platform enables
Custom Antibodies Services. GeneCust Europe. GeneCust Europe
 GeneCust Europe Laboratoire de Biotechnologie du Luxembourg S.A. 6 rue Dominique Lang L-3505 Dudelange Luxembourg Tél. : +352 27620411 Fax : +352 27620412 Email : [email protected] Web : www.genecust.com
GeneCust Europe Laboratoire de Biotechnologie du Luxembourg S.A. 6 rue Dominique Lang L-3505 Dudelange Luxembourg Tél. : +352 27620411 Fax : +352 27620412 Email : [email protected] Web : www.genecust.com
Serology: Fluorescent antibody tests and other tests employing conjugated antibodies
 Serology: Fluorescent antibody tests and other tests employing conjugated antibodies Authors: Adapted by Prof M van Vuuren. Originally compiled by Dr RW Worthington. (Retired) Licensed under a Creative
Serology: Fluorescent antibody tests and other tests employing conjugated antibodies Authors: Adapted by Prof M van Vuuren. Originally compiled by Dr RW Worthington. (Retired) Licensed under a Creative
Bio-Reagents Gene synthesis Peptide Synthesis Protein Expression Antibody Production. Life Science Products and Services
 Bio-Reagents Gene synthesis Peptide Synthesis Protein Expression Antibody Production Life Science Products and Services Since 2002, Biomatik has provided worldwide researchers in life science discovery
Bio-Reagents Gene synthesis Peptide Synthesis Protein Expression Antibody Production Life Science Products and Services Since 2002, Biomatik has provided worldwide researchers in life science discovery
THE His Tag Antibody, mab, Mouse
 THE His Tag Antibody, mab, Mouse Cat. No. A00186 Technical Manual No. TM0243 Update date 01052011 I Description.... 1 II Key Features. 2 III Storage 2 IV Applications.... 2 V Examples - ELISA..... 2 VI
THE His Tag Antibody, mab, Mouse Cat. No. A00186 Technical Manual No. TM0243 Update date 01052011 I Description.... 1 II Key Features. 2 III Storage 2 IV Applications.... 2 V Examples - ELISA..... 2 VI
Topic: Serological reactions: the purpose and a principle of reactions. Agglutination test. Precipitation test. CFT, IFT, ELISA, RIA.
 Topic: Serological reactions: the purpose and a principle of reactions. Agglutination test. Precipitation test. CFT, IFT, ELISA, RIA. Serology is the study and use of immunological tests to diagnose and
Topic: Serological reactions: the purpose and a principle of reactions. Agglutination test. Precipitation test. CFT, IFT, ELISA, RIA. Serology is the study and use of immunological tests to diagnose and
1) Siderophores are bacterial proteins that compete with animal A) Antibodies. B) Red blood cells. C) Transferrin. D) White blood cells. E) Receptors.
 Prof. Lester s BIOL 210 Practice Exam 4 (There is no answer key. Please do not email or ask me for answers.) Chapters 15, 16, 17, 19, HIV/AIDS, TB, Quorum Sensing 1) Siderophores are bacterial proteins
Prof. Lester s BIOL 210 Practice Exam 4 (There is no answer key. Please do not email or ask me for answers.) Chapters 15, 16, 17, 19, HIV/AIDS, TB, Quorum Sensing 1) Siderophores are bacterial proteins
LAB 14 ENZYME LINKED IMMUNOSORBENT ASSAY (ELISA)
 STUDENT GUIDE LAB 14 ENZYME LINKED IMMUNOSORBENT ASSAY (ELISA) GOAL The goal of this laboratory lesson is to explain the concepts and technique of enzyme linked immunosorbent assay (ELISA). OBJECTIVES
STUDENT GUIDE LAB 14 ENZYME LINKED IMMUNOSORBENT ASSAY (ELISA) GOAL The goal of this laboratory lesson is to explain the concepts and technique of enzyme linked immunosorbent assay (ELISA). OBJECTIVES
From Biology to Discovery
 POLYCLONAL ANTIBODY PRODUCTION SERVICES Abbiotec offers custom polyclonal antibody production services to the scientific community to accelerate a project From Biology to Discovery. When the antibody for
POLYCLONAL ANTIBODY PRODUCTION SERVICES Abbiotec offers custom polyclonal antibody production services to the scientific community to accelerate a project From Biology to Discovery. When the antibody for
Optimal Conditions for F(ab ) 2 Antibody Fragment Production from Mouse IgG2a
 Optimal Conditions for F(ab ) 2 Antibody Fragment Production from Mouse IgG2a Ryan S. Stowers, 1 Jacqueline A. Callihan, 2 James D. Bryers 2 1 Department of Bioengineering, Clemson University, Clemson,
Optimal Conditions for F(ab ) 2 Antibody Fragment Production from Mouse IgG2a Ryan S. Stowers, 1 Jacqueline A. Callihan, 2 James D. Bryers 2 1 Department of Bioengineering, Clemson University, Clemson,
Antibody Services from GenScript
 Services from GenScript www.genscript.com GenScript USA Inc. 860 Centennial Ave., Piscataway, NJ 08854 USA Toll-Free: 1-877-436-7274 Fax: 1-732-210-0262 1-732-885-5878 Academic Services Pharmaceutical
Services from GenScript www.genscript.com GenScript USA Inc. 860 Centennial Ave., Piscataway, NJ 08854 USA Toll-Free: 1-877-436-7274 Fax: 1-732-210-0262 1-732-885-5878 Academic Services Pharmaceutical
High Throughput Robotic Production of Monoclonal Antibodies at MATF. Ed Nice, Daniel Layton, Caroline Laverty, Natasha Dodge,
 High Throughput Robotic Production of Monoclonal Antibodies at MATF Ed Nice, Daniel Layton, Caroline Laverty, Natasha Dodge, Bronwyn Briers and Jean Tang Monash Antibody Technology Facility Clayton Victoria
High Throughput Robotic Production of Monoclonal Antibodies at MATF Ed Nice, Daniel Layton, Caroline Laverty, Natasha Dodge, Bronwyn Briers and Jean Tang Monash Antibody Technology Facility Clayton Victoria
Analyzing antibody sequence for recombinant antibody expression. Hangxing Yu, Ph.D Senior Scientist, GenScript May 20, 2015
 Analyzing antibody sequence for recombinant antibody expression Hangxing Yu, Ph.D Senior Scientist, GenScript May 20, 2015 Presentation Outline 1 2 3 4 Antibody basics, structure and function Antibody
Analyzing antibody sequence for recombinant antibody expression Hangxing Yu, Ph.D Senior Scientist, GenScript May 20, 2015 Presentation Outline 1 2 3 4 Antibody basics, structure and function Antibody
The Immune System: A Tutorial
 The Immune System: A Tutorial Modeling and Simulation of Biological Systems 21-366B Shlomo Ta asan Images taken from http://rex.nci.nih.gov/behindthenews/uis/uisframe.htm http://copewithcytokines.de/ The
The Immune System: A Tutorial Modeling and Simulation of Biological Systems 21-366B Shlomo Ta asan Images taken from http://rex.nci.nih.gov/behindthenews/uis/uisframe.htm http://copewithcytokines.de/ The
Antibody Structure, and the Generation of B-cell Diversity CHAPTER 4 04/05/15. Different Immunoglobulins
 Antibody Structure, and the Generation of B-cell Diversity B cells recognize their antigen without needing an antigen presenting cell CHAPTER 4 Structure of Immunoglobulin G Different Immunoglobulins Differences
Antibody Structure, and the Generation of B-cell Diversity B cells recognize their antigen without needing an antigen presenting cell CHAPTER 4 Structure of Immunoglobulin G Different Immunoglobulins Differences
Fulfilling the Promise
 Fulfilling the Promise Advancing the Fight Against Cancer: America s Medical Schools and Teaching Hospitals For more than a century, the nation s medical schools and teaching hospitals have worked to understand,
Fulfilling the Promise Advancing the Fight Against Cancer: America s Medical Schools and Teaching Hospitals For more than a century, the nation s medical schools and teaching hospitals have worked to understand,
Antibody Services from GenScript, Brochure
 Services from GenScript, Brochure Interest in any of the products, request or order them at Bio-Connect. Bio-Connect B.V. T NL +31 (0)26 326 44 50 T BE +32 (0)2 503 03 48 Begonialaan 3a F NL +31 (0)26
Services from GenScript, Brochure Interest in any of the products, request or order them at Bio-Connect. Bio-Connect B.V. T NL +31 (0)26 326 44 50 T BE +32 (0)2 503 03 48 Begonialaan 3a F NL +31 (0)26
Basic Immunologic Procedures. Complex Serological Tests
 Basic Immunologic Procedures Complex Serological Tests Amal Alghamdi 2014-2015 1 Classification of antigen-antibody interactions: 1. Primary serological tests: (Marker techniques) e.g. Enzyme linked immuonosorben
Basic Immunologic Procedures Complex Serological Tests Amal Alghamdi 2014-2015 1 Classification of antigen-antibody interactions: 1. Primary serological tests: (Marker techniques) e.g. Enzyme linked immuonosorben
Final Review. Aptamers. Making Aptamers: SELEX 6/3/2011. sirna and mirna. Central Dogma. RNAi: A translation regulation mechanism.
 Central Dogma Final Review Section Week 10 DNA RNA Protein DNA DNA replication DNA RNA transcription RNA Protein translation **RNA DNA reverse transcription http://bass.bio.uci.edu/~hudel/bs99a/lecture20/lecture1_1.html
Central Dogma Final Review Section Week 10 DNA RNA Protein DNA DNA replication DNA RNA transcription RNA Protein translation **RNA DNA reverse transcription http://bass.bio.uci.edu/~hudel/bs99a/lecture20/lecture1_1.html
10. T and B cells are types of a. endocrine cells. c. lymphocytes. b. platelets. d. complement cells.
 Virus and Immune System Review Directions: Write your answers on a separate piece of paper. 1. Why does a cut in the skin threaten the body s nonspecific defenses against disease? a. If a cut bleeds, disease-fighting
Virus and Immune System Review Directions: Write your answers on a separate piece of paper. 1. Why does a cut in the skin threaten the body s nonspecific defenses against disease? a. If a cut bleeds, disease-fighting
DNA Fingerprinting. Unless they are identical twins, individuals have unique DNA
 DNA Fingerprinting Unless they are identical twins, individuals have unique DNA DNA fingerprinting The name used for the unambiguous identifying technique that takes advantage of differences in DNA sequence
DNA Fingerprinting Unless they are identical twins, individuals have unique DNA DNA fingerprinting The name used for the unambiguous identifying technique that takes advantage of differences in DNA sequence
Grundlagen und Anwendung der Genom- und Proteomforschung
 Grundlagen und Anwendung der Genom- und Proteomforschung Profilmodul (Vorlesung/Seminar WS 2012/13) Philipps-Universität Marburg Fachbereich Biologie Professor Dr. Egon Amann & Dr. Frank Vitzthum Gastbeiträge
Grundlagen und Anwendung der Genom- und Proteomforschung Profilmodul (Vorlesung/Seminar WS 2012/13) Philipps-Universität Marburg Fachbereich Biologie Professor Dr. Egon Amann & Dr. Frank Vitzthum Gastbeiträge
Guide to Purification of Polyclonal Antibodies
 Guide to Purification of Polyclonal Antibodies When choosing a polyclonal antibody, either as a primary or secondary antibody in an immunoassay, researchers are often inundated with an array of antibody
Guide to Purification of Polyclonal Antibodies When choosing a polyclonal antibody, either as a primary or secondary antibody in an immunoassay, researchers are often inundated with an array of antibody
Genes to Proteins to Antibodies
 Genes to Proteins to Antibodies About Us Fusion Antibodies is a CRO established in 2001 as a spin-out from Queen s University Belfast. The company building is situated in a charming area of Springbank
Genes to Proteins to Antibodies About Us Fusion Antibodies is a CRO established in 2001 as a spin-out from Queen s University Belfast. The company building is situated in a charming area of Springbank
WHO Prequalification of Diagnostics Programme PUBLIC REPORT. Product: Genscreen ULTRA HIV Ag-Ab Number: PQDx 0096-031-00. Abstract
 WHO Prequalification of Diagnostics Programme PUBLIC REPORT Product: Genscreen ULTRA HIV Ag-Ab Number: PQDx 0096-031-00 Abstract Genscreen ULTRA HIV Ag-Ab with product codes 72386 and 72388, manufactured
WHO Prequalification of Diagnostics Programme PUBLIC REPORT Product: Genscreen ULTRA HIV Ag-Ab Number: PQDx 0096-031-00 Abstract Genscreen ULTRA HIV Ag-Ab with product codes 72386 and 72388, manufactured
Principles of Vaccination
 Immunology and Vaccine-Preventable Diseases Immunology is a complicated subject, and a detailed discussion of it is beyond the scope of this text. However, an understanding of the basic function of the
Immunology and Vaccine-Preventable Diseases Immunology is a complicated subject, and a detailed discussion of it is beyond the scope of this text. However, an understanding of the basic function of the
Some Immunological Test. Presented by Alaa Faeiz Ashwaaq Dyaa Aseel Abd AL-Razaq Supervised by D.Feras
 Some Immunological Test Presented by Alaa Faeiz Ashwaaq Dyaa Aseel Abd AL-Razaq Supervised by D.Feras Alaa Faeiz Antigen -Antibody Reactions. Antigen antibody reactions are performed to determine the presence
Some Immunological Test Presented by Alaa Faeiz Ashwaaq Dyaa Aseel Abd AL-Razaq Supervised by D.Feras Alaa Faeiz Antigen -Antibody Reactions. Antigen antibody reactions are performed to determine the presence
UNDERSTANDING MULTIPLE MYELOMA AND LABORATORY VALUES Benjamin Parsons, DO [email protected] Gundersen Health System Center for Cancer and
 UNDERSTANDING MULTIPLE MYELOMA AND LABORATORY VALUES Benjamin Parsons, DO [email protected] Gundersen Health System Center for Cancer and Blood Disorders La Crosse, WI UNDERSTANDING MULTIPLE
UNDERSTANDING MULTIPLE MYELOMA AND LABORATORY VALUES Benjamin Parsons, DO [email protected] Gundersen Health System Center for Cancer and Blood Disorders La Crosse, WI UNDERSTANDING MULTIPLE
Custom Antibodies & Recombinant Proteins
 Custom Antibodies & Recombinant Proteins INTRODUCTION Custom services to meet your research and development requirements Improvements in health, medicine and diagnostics over the past century can be largely
Custom Antibodies & Recombinant Proteins INTRODUCTION Custom services to meet your research and development requirements Improvements in health, medicine and diagnostics over the past century can be largely
T Cell Maturation,Activation and Differentiation
 T Cell Maturation,Activation and Differentiation Positive Selection- In thymus, permits survival of only those T cells whose TCRs recognize self- MHC molecules (self-mhc restriction) Negative Selection-
T Cell Maturation,Activation and Differentiation Positive Selection- In thymus, permits survival of only those T cells whose TCRs recognize self- MHC molecules (self-mhc restriction) Negative Selection-
Transgenic technology in the production of therapeutic proteins
 Transgenic technology in the production of therapeutic proteins Transgenic technology represents a new generation of biopharmaceutical production system to meet the medical needs of the new millennium.
Transgenic technology in the production of therapeutic proteins Transgenic technology represents a new generation of biopharmaceutical production system to meet the medical needs of the new millennium.
B Cells and Antibodies
 B Cells and Antibodies Andrew Lichtman, MD PhD Brigham and Women's Hospital Harvard Medical School Lecture outline Functions of antibodies B cell activation; the role of helper T cells in antibody production
B Cells and Antibodies Andrew Lichtman, MD PhD Brigham and Women's Hospital Harvard Medical School Lecture outline Functions of antibodies B cell activation; the role of helper T cells in antibody production
