Pacemaker Counter and Histogram Operation and Interpretation. Percentage of total count. Figure 2. Detailed ALTRUA Paced and Sensed Event Counters
|
|
|
- Victoria Arnold
- 9 years ago
- Views:
Transcription
1 A Closer Look SUMMARY This article provides information regarding the operation and interpretation of Event Counters and Histograms available in ALTRUA, INSIGNIA, PULSAR MAX II, and DISCOVERY II pacemakers. Products Referenced All referenced Boston Scientific Pulse Generators and the ZOOM LATITUDE Programming System. ALTRUA, INSIGNIA, PULSAR, DISCOVERY, ZOOM, and LATITUDE are registered trademarks of Boston Scientific Corporation. Products referenced herein may not be approved in all geographies. For comprehensive information on device operation, reference the full instructions for use found at: CAUTION: Federal (U.S.) law restricts this device to sale by or on the order of a physician. All graphics produced by Boston Scientific Corporation, unless otherwise noted. Pacemaker Counter and Histogram Operation and Interpretation The Event Counters feature in Boston Scientific pacemakers feature counts and records, and display a variety of data, including the total number of paced and sensed events. The Histograms feature displays a graphical representation of the counter data and provides the amount of time that the device has spent pacing and sensing at various rates. Event Counters and Histograms are available by selecting Therapy History on the ZOOM LATITUDE Programmer. Event Counters The Paced and Sensed Event Counters (Figure 1) record the number of intrinsic and paced events that occur during an event recording period. This period begins with the last time the Counters were reset by the clinician and ends when the data set is retrieved from the pacemaker during a telemetry session. Three sets of event counters are available for review (Figure 2) by pressing the Paced and Sensed Details icon (Figure 1): 1. Atrial-only events 2. Ventricular-only events 3. Combination Atrial-Ventricular (A-V) events NOTE: These data are provided as both a percentage of the total events and a total count since the last reset. CRT-D: Cardiac Resynchronization Therapy Defibrillator CRT-P: Cardiac Resynchronization Therapy Pacemaker ICD: Implantable Cardioverter Defibrillator S-ICD: Subcutaneous Implantable Defibrillator Contact Information Americas (Caribbean, and Central, North, and South America) LATITUDE Customer Support CARDIAC ( ) Patient Services Europe, Middle East, Africa [email protected] LATITUDE Clinician Support [email protected] Asia Pacific [email protected] [email protected] LATITUDE Customer Support [email protected] [email protected] (Japan) 2013 by Boston Scientific Corporation or its affiliates. All rights reserved. Details Icon Figure 1. ALTRUA Event Counters Percentage of total count Total count since last reset K total atrial events K total ventricular events K total A-V Events Figure 2. Detailed ALTRUA Paced and Sensed Event Counters September 5, , Rev. B, US Page 1 of 4
2 Events Counted The events that are counted are: Atrial-only events: Counted from the atrial channel and assigned to either the paced or sensed bin. Ventricular-only events: Counted from the ventricular channel and assigned to either the paced or sensed bin. Combination A-V events: Counted from the ventricular channel, and then categorized in the following sequence based upon the preceding atrial event: 1. The ventricular event is designated as either paced or sensed. 2. The atrial event immediately preceding the ventricular event is classified as either paced or sensed. 3. The overall A-V combination is assigned to one of four possible A-V pace/sense bins (Figure 2). Comparing Counter Data When evaluating counter data, the following should be considered: The total of the combination A-V event counters will always match the total of the ventricular-only event counters as both are counted from the same ventricular events (Figure 2). The total of the combination A-V event counters will not match the total of the atrial-only event counters unless there is perfect 1:1 A-V conduction in dual-chamber mode. In this scenario, every ventricular event is preceded by one and only one atrial event, resulting in matching counters. Occasionally, the total of the combination A-V event counters will not match the total of the atrial-only counters (Figure 2) because the atrium and the ventricle often work independently of each other. Examples of common situations resulting in dissociation of the counters include: - Atrial arrhythmias (more than one atrial event for each ventricular event) - Ventricular arrhythmias (more than one ventricular event for each atrial event) NOTE: PVCs are not included in the ventricular pace/sense counters; they have their own counters. - Device programming to a single-chamber mode AAI(R) or VVI(R) Table 1 contains examples of counter results that may be observed in the clinical setting. Table 1. Examples of Counter Results in Various Programmed Modes Example 1 A dual-chamber device programmed to DDI(R) mode in a patient with frequent atrial fibrillation A large number of atrial-sensed events are recorded as a result of the patient s atrial fibrillation. These data are not represented in the combination A-V event counters since those counters only recognizes the single atrial event immediately preceding the associated ventricular event K K K K K K K 0 77 Counters do not match due to atrial fibrillation. September 5, , Rev. B, US Page 2 of 4
3 Example 2 A dual-chamber device programmed to VVI(R) mode While the device is operating in VVI mode, even though a lack of atrial sensing prevents classification of combination A-V events, the A-V event counters are populated. This occurs because both V and AV counters count ventricular-only events. By default, the ventricular events are considered to have been preceded by atrial sensed events and are classified accordingly, even though no atrial data have been collected. Nevertheless, both totals are fully accurate. In addition, note that the totals are also equal K K K K In VVI mode, the V counters match the Combination A-V counters since the source data are the same. Example 3 A dual-chamber device programmed to AAI(R) mode While the device is operating in AAI mode, the A-V event counters are not populated, because they count ventricular activity K 9 18K V counters and Combination A-V counters are not populated (i.e., set at 0 ) when the device is programmed to AAI Mode. Histograms Histograms provide a graphical representation of the atrial and ventricular paced and sensed events collected during the recording period. This information may facilitate diagnostic interpretation of cardiac activity. Events are sorted into rate bins on the Histograms, with two different options for displaying the data, AV Histograms and Pace/Sense Histograms. A-V Histograms show the type of ventricular events (paced or sensed) that follow atrial activity (Figure 3). The maximum Y-axis scale value (50% or 100%) is automatically determined by the maximum value in either the Ventricular Paced/ Ventricular Sensed bins (displayed on the Pace/Sense Histograms) or the Atrial Paced VP/VS bins (displayed on the A-V Histograms). Pace/Sense Histograms show atrial paced and sensed events and ventricular paced and sensed events (Figure 4). The maximum Y-axis scale value (50% or 100%) is automatically determined by the maximum value in any of the Atrial or Ventricular bins (displayed on the Pace/Sense Histograms). September 5, , Rev. B, US Page 3 of 4
4 Figure 3. ALTRUA A-V Histograms Figure 4. ALTRUA Pace/Sense Histograms When the A-V Histograms are printed, sometimes an Atrial Sensed histogram bar that extends beyond the chart boundaries (Figure 5) is present. This will occur only if: The highest value of Ventricular Paced/Ventricular Sensed or Atrial Paced VP/VS events at any given rate is less than 50%, setting the maximum Y-axis scale value to 50% (rather than 100%), AND Atrial sensed VP/VS events occurred with a frequency greater than 50% at any given rate. Because this value is greater than the set Y-axis scale value of 50%, the histogram bar will appear above the previously scaled Y-axis. In this situation, the printed histogram differs from the histogram displayed on the programmer in the following manner: Figure 5. A-V Histograms Printout Programmer screen: the bar noted above is automatically truncated and does not extend beyond the top of the grid. Printout: the actual pacing percentage will be displayed, by going beyond 50%. Counters will also display the actual pacing percentage. Also, the bar will extend beyond the top of the grid as shown in Figure 5. NOTE: This Y-axis scaling will not occur with the Pace/Sense Histograms because scale selection is based solely on a single maximum count (not on combinations). September 5, , Rev. B, US Page 4 of 4
5 ALTRUA and INSIGNIA Pacing Systems from Boston Scientific Indications Pacemaker indications include: symptomatic paroxysmal or permanent second- or third-degree AV block; symptomatic bilateral bundle branch block; symptomatic paroxysmal or transient sinus node dysfunction with or without associated AV conduction disorders; bradycardia-tachycardia syndrome, to prevent symptomatic bradycardia or some forms of symptomatic tachyarrhythmias; neurovascular (vaso-vagal) syndromes or hypersensitive carotid sinus syndromes. Adaptive-rate pacing is indicated for patients who may benefit from increased pacing rates concurrent with increases in minute ventilation and/or level of physical activity. Pacemakers dual-chamber and atrial tracking modes are also indicated for patients who may benefit from maintenance of AV synchrony. Dual-chamber modes are specifically indicated for: conduction disorders that require restoration of AV synchrony, including varying degrees of AV block; VVI intolerance (e.g., pacemaker syndrome) in the presence of persistent sinus rhythm. Contraindications Pacemakers are contraindicated for the following patients under the circumstances listed: patients with unipolar pacing leads or in MV mode with an implanted ICD because it may cause unwanted delivery or inhibition of ICD therapy; use of the MV sensor in patients with only unipolar leads, because a bipolar lead is required in either the atrium or the ventricle for MV detection (INSIGNIA Plus, ALTRUA 20/40); MV mode in patients with both unipolar atrial and ventricular leads (INSIGNIA Ultra, ALTRUA 60); single-chamber atrial pacing in patients with impaired AV nodal conduction; atrial tracking modes for patients with chronic refractory atrial tachyarrhythmias, which might trigger ventricular pacing; dual-chamber and single-chamber atrial pacing in patients with chronic refractory atrial tachyarrhythmias; asynchronous pacing in the presence (or likelihood) of competition between paced and intrinsic rhythms. Warnings Read the product labeling thoroughly before implanting the pulse generator to avoid damage to the system. For single use only-do not resterilize devices. Inappropriate sustained high-rate pacing occurred in the PULSAR MAX clinical study in 5 out of 130 patients with MV ON, 4 to 14 days after implant. If sustained high-rate pacing could be of concern, consider programming a reduced Max Sensor Rate or MV to Passive. These programming recommendations are intended to assure that MV calibration is evaluated and, if necessary, recalibrated (4 ON) when the patient and pacing system have stabilized post implant. Continued monitoring of the MV sensor performance should be performed at all follow-up visits until implant stabilization has occurred. Precautions For specific information on precautions, refer to the following sections of the product labeling: MV sensor calibration at implant; clinical considerations; sterilization, storage and handling; lead evaluation and connection; implantation; programming and pacemaker operation; MV initialization; environmental and medical therapy hazards; elevated pressure; explanted pacemakers. Advise patients to avoid sources of electric or magnetic interference (EMI). If the pacemaker inhibits or reverts to asynchronous operation at the programmed pacing rate or at the magnet rate while in the presence of the EMI, moving away from the source or turning it off will usually allow the pulse generator to return to its normal mode of operation. Potential Adverse Events Potential adverse events include, but are not limited to, the following: allergic/physical/physiologic reaction, death, erosion/migration, lead or accessory breakage (fracture/insulation/lead tip), hematoma/seroma, inappropriate or inability to provide therapy (pacing/sensing), infection, procedure related, and component failure. In rare cases severe complications or device failures can occur. Refer to the product labeling for specific indications, contraindications, warnings/precautions and adverse events. Rx only. (Rev. R) ZOOM LATITUDE Programming System from Boston Scientific Intended Use The Model 3120 Programmer/Recorder/Monitor (PRM) is intended to be used as a complete system to communicate with Guidant or Boston Scientific implantable pulse generators. The software in use controls all communication functions for the pulse generator. For detailed software application instructions, refer to the System Guide for the Guidant or Boston Scientific pulse generator being interrogated. Contraindications The Model 3120 PRM is contraindicated for use with any pulse generator other than a Guidant or Boston Scientific device. For contraindications for use related to the Guidant or Boston Scientific pulse generator, refer to the System Guide for the Guidant or Boston Scientific pulse generator being interrogated. Warnings There are no warnings associated with this programming system. Precautions For specific information on precautions, read the following sections of the product labeling: General, Preparation for Use, Maintenance and Handling. Adverse Effects None known. Refer to the product labeling for specific indications, contraindications, warnings/precautions and adverse effects. Rx only. (Rev. E) ADDENDUM TO: September 5, 2013 Most Current Brief Summaries www.
Basics of Pacing. Ruth Hickling, RN-BSN Tasha Conley, RN-BSN
 Basics of Pacing Ruth Hickling, RN-BSN Tasha Conley, RN-BSN The Cardiac Conduction System Cardiac Conduction System Review Normal Conduction Conduction QRS QRS Complex Complex RR PP ST ST segment segment
Basics of Pacing Ruth Hickling, RN-BSN Tasha Conley, RN-BSN The Cardiac Conduction System Cardiac Conduction System Review Normal Conduction Conduction QRS QRS Complex Complex RR PP ST ST segment segment
MRI Case Study MRI Cervical Spine
 MRI Case Study MRI Cervical Spine Jewish Hospital Louisville, KY The Revo MRI SureScan pacing system is MR Conditional designed to allow patients to undergo MRI under the specified conditions for use.
MRI Case Study MRI Cervical Spine Jewish Hospital Louisville, KY The Revo MRI SureScan pacing system is MR Conditional designed to allow patients to undergo MRI under the specified conditions for use.
Worldwide Experience with S-ICD: Early results from the EFFORTLESS Registry
 Worldwide Experience with S-ICD: Early results from the EFFORTLESS Registry Results published in European Heart Journal (March 2014) Pier D. Lambiase, Craig Barr, Dominic A.M.J. Theuns, Reinoud Knops,
Worldwide Experience with S-ICD: Early results from the EFFORTLESS Registry Results published in European Heart Journal (March 2014) Pier D. Lambiase, Craig Barr, Dominic A.M.J. Theuns, Reinoud Knops,
Introduction. Planned surgical procedures
 Guidelines for the perioperative management of patients with implantable pacemakers or implantable cardioverter defibrillators, where the use of surgical diathermy/electrocautery is anticipated. Introduction
Guidelines for the perioperative management of patients with implantable pacemakers or implantable cardioverter defibrillators, where the use of surgical diathermy/electrocautery is anticipated. Introduction
CLINICIAN MANUAL. LATITUDE Patient Management System
 CLINICIAN MANUAL LATITUDE Patient Management System CAUTION: Federal law restricts this device to sale by or on the order of a physician trained or experienced in device implant and follow-up procedures.
CLINICIAN MANUAL LATITUDE Patient Management System CAUTION: Federal law restricts this device to sale by or on the order of a physician trained or experienced in device implant and follow-up procedures.
REMOTE CARE ALERT MANAGEMENT REFERENCE GUIDE Managing Merlin.net
 REMOTE CARE ALERT MANAGEMENT REFERENCE GUIDE Managing Merlin.net Patient Care Network (PCN) Alerts Merlin.net PCN offers a powerful range of alert capabilities to give your practice the control, flexibility
REMOTE CARE ALERT MANAGEMENT REFERENCE GUIDE Managing Merlin.net Patient Care Network (PCN) Alerts Merlin.net PCN offers a powerful range of alert capabilities to give your practice the control, flexibility
MRI SureScan Pacing System
 MRI SureScan Pacing System Step-by-step Instructions For A Successful MRI Scan Timothy S. E. Albert, MD Medical Director Advanced Diagnostic Imaging Center Monterey, CA Lisa L. Hungate, BS, RN clinical
MRI SureScan Pacing System Step-by-step Instructions For A Successful MRI Scan Timothy S. E. Albert, MD Medical Director Advanced Diagnostic Imaging Center Monterey, CA Lisa L. Hungate, BS, RN clinical
CCAD Training Manual. Cardiac Rhythm Management (CRM)
 CCAD Training Manual Cardiac Rhythm Management (CRM) Version 1.0 A D Cunningham 19/3/2008 Introduction This manual is intended to assist users of the Notes Client version of the CCAD Cardiac Rhythm Management
CCAD Training Manual Cardiac Rhythm Management (CRM) Version 1.0 A D Cunningham 19/3/2008 Introduction This manual is intended to assist users of the Notes Client version of the CCAD Cardiac Rhythm Management
ACCOLADE PROPONENT ESSENTIO ALTRUA 2 FORMIO VITALIO INGENIO ADVANTIO CAUTION:
 REFERENCE GUIDE ACCOLADE Model L300, L301, L321 PROPONENT Model L200, L201, L221 ESSENTIO Model L100, L101, L121 ALTRUA 2 Model S701, S702, S722 FORMIO Model K278 VITALIO Model K272, K273, K274 INGENIO
REFERENCE GUIDE ACCOLADE Model L300, L301, L321 PROPONENT Model L200, L201, L221 ESSENTIO Model L100, L101, L121 ALTRUA 2 Model S701, S702, S722 FORMIO Model K278 VITALIO Model K272, K273, K274 INGENIO
Dual Chamber Temporary Pacemaker
 5388 Dual Chamber Temporary Pacemaker Technical Manual Caution: Federal Law (USA) restricts this device to sale by c or on the order of a physician. 0123 MODEL 5388 0 Technical Manual 0 Dual Chamber Temporary
5388 Dual Chamber Temporary Pacemaker Technical Manual Caution: Federal Law (USA) restricts this device to sale by c or on the order of a physician. 0123 MODEL 5388 0 Technical Manual 0 Dual Chamber Temporary
SW012. Installation software for the Right Ventricular Lead Integrity Alert feature. Reference Manual
 Installation software for the Right Ventricular Lead Integrity Alert feature Reference Manual Caution: Federal law (USA) restricts this device to sale by or on the order of a physician. The following list
Installation software for the Right Ventricular Lead Integrity Alert feature Reference Manual Caution: Federal law (USA) restricts this device to sale by or on the order of a physician. The following list
UCSF Anesthesia Resident Pearls: Cardiac Implantable Electronic Devices: Pacemakers and ICDs Table of Contents
 UCSF Anesthesia Resident Pearls: Cardiac Implantable Electronic Devices: Pacemakers and ICDs Table of Contents Page 1 Intro Indications for Pacers and ICDs Page 2 Differentiating between Pacers and ICDs
UCSF Anesthesia Resident Pearls: Cardiac Implantable Electronic Devices: Pacemakers and ICDs Table of Contents Page 1 Intro Indications for Pacers and ICDs Page 2 Differentiating between Pacers and ICDs
Atrial Fibrillation and Cardiac Device Therapy RAKESH LATCHAMSETTY, MD DIVISION OF ELECTROPHYSIOLOGY UNIVERSITY OF MICHIGAN HOSPITAL ANN ARBOR, MI
 Atrial Fibrillation and Cardiac Device Therapy RAKESH LATCHAMSETTY, MD DIVISION OF ELECTROPHYSIOLOGY UNIVERSITY OF MICHIGAN HOSPITAL ANN ARBOR, MI Outline Atrial Fibrillation What is it? What are the associated
Atrial Fibrillation and Cardiac Device Therapy RAKESH LATCHAMSETTY, MD DIVISION OF ELECTROPHYSIOLOGY UNIVERSITY OF MICHIGAN HOSPITAL ANN ARBOR, MI Outline Atrial Fibrillation What is it? What are the associated
Management of Pacing Wires After Cardiac Surgery
 Management of Pacing Wires After Cardiac Surgery David E. Lizotte, Jr. PA C, MPAS, FAPACVS President, Association of Physician Assistants in Cardiovascular Surgery Conflicts: None Indications 2008 Journal
Management of Pacing Wires After Cardiac Surgery David E. Lizotte, Jr. PA C, MPAS, FAPACVS President, Association of Physician Assistants in Cardiovascular Surgery Conflicts: None Indications 2008 Journal
New CPT Codes for Cardiac Device Monitoring SIDE-BY-SIDE COMPARISON 2008-2009
 New CPT Codes for Cardiac Device Monitoring SIDE-BY-SIDE COMPARISON 2008-2009 Effective January 1, 2009 NEW CPT 1 CODES FOR CARDIAC DEVICE MONITORING Twenty-three new CPT codes for Cardiac Device Monitoring
New CPT Codes for Cardiac Device Monitoring SIDE-BY-SIDE COMPARISON 2008-2009 Effective January 1, 2009 NEW CPT 1 CODES FOR CARDIAC DEVICE MONITORING Twenty-three new CPT codes for Cardiac Device Monitoring
CareLink Network. Clinic Setup Guide WORKFLOWS. FAQs
 INTRODUCTION CareLink Network Clinic Setup Guide All patient and clinical data displayed on the screenshots are fictitious and for demonstration purposes only. Introduction INTRODUCTION Transforming cardiac
INTRODUCTION CareLink Network Clinic Setup Guide All patient and clinical data displayed on the screenshots are fictitious and for demonstration purposes only. Introduction INTRODUCTION Transforming cardiac
IMAGEREADY MR CONDITIONAL DEFIBRILLATION SYSTEM
 MRI TECHNICAL GUIDE IMAGEREADY MR CONDITIONAL DEFIBRILLATION SYSTEM REF D000, D002, D010, D012, D020, D022, D044, D046, D050, D052, D140, D142, D150, D152, D174, D176, G058, G148, G158, G179, 0265, 0266,
MRI TECHNICAL GUIDE IMAGEREADY MR CONDITIONAL DEFIBRILLATION SYSTEM REF D000, D002, D010, D012, D020, D022, D044, D046, D050, D052, D140, D142, D150, D152, D174, D176, G058, G148, G158, G179, 0265, 0266,
Tachyarrhythmias (fast heart rhythms)
 Patient information factsheet Tachyarrhythmias (fast heart rhythms) The normal electrical system of the heart The heart has its own electrical conduction system. The conduction system sends signals throughout
Patient information factsheet Tachyarrhythmias (fast heart rhythms) The normal electrical system of the heart The heart has its own electrical conduction system. The conduction system sends signals throughout
STANDARDS FOR IMPLANTATION AND FOLLOW-UP OF CARDIAC RHYTHM MANAGEMENT DEVICES IN ADULTS January 2013
 1. INTRODUCTION STANDARDS FOR IMPLANTATION AND FOLLOW-UP OF CARDIAC RHYTHM MANAGEMENT DEVICES IN ADULTS January 2013 This document replaces the previous Heart Rhythm UK documents Clinical Guidance by Consensus
1. INTRODUCTION STANDARDS FOR IMPLANTATION AND FOLLOW-UP OF CARDIAC RHYTHM MANAGEMENT DEVICES IN ADULTS January 2013 This document replaces the previous Heart Rhythm UK documents Clinical Guidance by Consensus
Diagnosis Code Crosswalk : ICD-9-CM to ICD-10-CM Cardiac Rhythm and Heart Failure Diagnoses
 Diagnosis Code Crosswalk : to 402.01 Hypertensive heart disease, malignant, with heart failure 402.11 Hypertensive heart disease, benign, with heart failure 402.91 Hypertensive heart disease, unspecified,
Diagnosis Code Crosswalk : to 402.01 Hypertensive heart disease, malignant, with heart failure 402.11 Hypertensive heart disease, benign, with heart failure 402.91 Hypertensive heart disease, unspecified,
DEPARTMENT OF HEALTH AND HUMAN SERVICES Centers for Medicare & Medicaid Services
 DEPARTMENT OF HEALTH AND HUMAN SERVICES Centers for Medicare & Medicaid Services NEW product from the Medicare Learning Network (MLN) Provider Compliance Tips for Computed Tomography (CT) Scans Podcast,
DEPARTMENT OF HEALTH AND HUMAN SERVICES Centers for Medicare & Medicaid Services NEW product from the Medicare Learning Network (MLN) Provider Compliance Tips for Computed Tomography (CT) Scans Podcast,
Michael Hartman, CRNA, DNP, MSN, BSN, BA
 Michael Hartman, CRNA, DNP, MSN, BSN, BA Intrinsic Conduction Automaticity-cardiac cells ability to spontaneously depolarize and initiate impulse Depolarization-cardiac cells go to a+ intracellular and
Michael Hartman, CRNA, DNP, MSN, BSN, BA Intrinsic Conduction Automaticity-cardiac cells ability to spontaneously depolarize and initiate impulse Depolarization-cardiac cells go to a+ intracellular and
LATITUDE Patient Management. Purpose. Objectives
 Contents Purpose...1 Objectives...1 Application Overview...2 Component Definitions...3 Follow-up Information Retrieval...4 Patient Record Dismissal...6 Follow-up Information Transfer..10 Patient Record
Contents Purpose...1 Objectives...1 Application Overview...2 Component Definitions...3 Follow-up Information Retrieval...4 Patient Record Dismissal...6 Follow-up Information Transfer..10 Patient Record
LATITUDE NXT 4.0 Quick Reference Guide
 LATITUDE NXT 4.0 Quick Reference Guide LATITUDE NXT is compatible with Boston Scientific s most advanced pacemaker and defibrillator families 1, and now also with the EMBLEM sub-cutaneous defibrillator.
LATITUDE NXT 4.0 Quick Reference Guide LATITUDE NXT is compatible with Boston Scientific s most advanced pacemaker and defibrillator families 1, and now also with the EMBLEM sub-cutaneous defibrillator.
What Are Arrhythmias?
 What Are Arrhythmias? Many people have questions about what the word arrhythmia means, and arrhythmias can be a difficult subject to understand. The text below should give you a better understanding of
What Are Arrhythmias? Many people have questions about what the word arrhythmia means, and arrhythmias can be a difficult subject to understand. The text below should give you a better understanding of
«Δυσλειτουργία βηματοδότη. Πως μπορούμε να την εκτιμήσουμε στο ιατρείο.» Koσσυβάκης Χάρης Καρδιολογικό Τμήμα Γ.Ν.Α. «Γ. ΓΕΝΝΗΜΑΤΑΣ
 «Δυσλειτουργία βηματοδότη. Πως μπορούμε να την εκτιμήσουμε στο ιατρείο.» Koσσυβάκης Χάρης Καρδιολογικό Τμήμα Γ.Ν.Α. «Γ. ΓΕΝΝΗΜΑΤΑΣ Diagnostic tools History: symptoms, physical examination 12 leads ECG,
«Δυσλειτουργία βηματοδότη. Πως μπορούμε να την εκτιμήσουμε στο ιατρείο.» Koσσυβάκης Χάρης Καρδιολογικό Τμήμα Γ.Ν.Α. «Γ. ΓΕΝΝΗΜΑΤΑΣ Diagnostic tools History: symptoms, physical examination 12 leads ECG,
CLINICIAN MANUAL. LATITUDE NXT Patient Management System
 CLINICIAN MANUAL LATITUDE NXT Patient Management System CAUTION: Federal (USA) law restricts this device to distribution and use by or on the lawful order of a physician. Table of Contents INTRODUCTION:
CLINICIAN MANUAL LATITUDE NXT Patient Management System CAUTION: Federal (USA) law restricts this device to distribution and use by or on the lawful order of a physician. Table of Contents INTRODUCTION:
Reimbursement Information For Electrophysiology and Arrhythmia Service Procedures 1
 GE Healthcare Information For Electrophysiology and Arrhythmia Procedures 1 2011 Update www.gehealthcare.com/reimbursement This overview addresses coding, coverage, and payment for electrophysiology procedures
GE Healthcare Information For Electrophysiology and Arrhythmia Procedures 1 2011 Update www.gehealthcare.com/reimbursement This overview addresses coding, coverage, and payment for electrophysiology procedures
COVERAGE GUIDANCE: ABLATION FOR ATRIAL FIBRILLATION
 COVERAGE GUIDANCE: ABLATION FOR ATRIAL FIBRILLATION Question: How should the EGBS Coverage Guidance regarding ablation for atrial fibrillation be applied to the Prioritized List? Question source: Evidence
COVERAGE GUIDANCE: ABLATION FOR ATRIAL FIBRILLATION Question: How should the EGBS Coverage Guidance regarding ablation for atrial fibrillation be applied to the Prioritized List? Question source: Evidence
INTRODUCTORY GUIDE TO IDENTIFYING ECG IRREGULARITIES
 INTRODUCTORY GUIDE TO IDENTIFYING ECG IRREGULARITIES NOTICE: This is an introductory guide for a user to understand basic ECG tracings and parameters. The guide will allow user to identify some of the
INTRODUCTORY GUIDE TO IDENTIFYING ECG IRREGULARITIES NOTICE: This is an introductory guide for a user to understand basic ECG tracings and parameters. The guide will allow user to identify some of the
INSERTABLE CARDIAC MONITORING SYSTEM. UNLOCK the ANSWER. Your heart and long-term monitoring
 INSERTABLE CARDIAC MONITORING SYSTEM UNLOCK the ANSWER Your heart and long-term monitoring UNLOCK the ANSWER Irregular heartbeats can be related to a variety of conditions, including unexplained fainting,
INSERTABLE CARDIAC MONITORING SYSTEM UNLOCK the ANSWER Your heart and long-term monitoring UNLOCK the ANSWER Irregular heartbeats can be related to a variety of conditions, including unexplained fainting,
By the end of this continuing education module the clinician will be able to:
 EKG Interpretation WWW.RN.ORG Reviewed March, 2015, Expires April, 2017 Provider Information and Specifics available on our Website Unauthorized Distribution Prohibited 2015 RN.ORG, S.A., RN.ORG, LLC Developed
EKG Interpretation WWW.RN.ORG Reviewed March, 2015, Expires April, 2017 Provider Information and Specifics available on our Website Unauthorized Distribution Prohibited 2015 RN.ORG, S.A., RN.ORG, LLC Developed
Pacers use a 5-letter code: first 3 letters most important
 PACEMAKERS 2 Pacemakers: Nomenclature Pacers use a 5-letter code: first 3 letters most important t First Letter: Chamber Paced A= Atrium V= Ventricle D= Dual (A+V) 2nd Letter: Chamber Sensed A= Atrium
PACEMAKERS 2 Pacemakers: Nomenclature Pacers use a 5-letter code: first 3 letters most important t First Letter: Chamber Paced A= Atrium V= Ventricle D= Dual (A+V) 2nd Letter: Chamber Sensed A= Atrium
Implantable Cardioverter Defibrillator Therapy
 Implantable Cardioverter Defibrillator Therapy A Message to Patients Boston Scientific Corporation acquired Guidant Corporation in April 2006. During our transition period, you may see both the Boston
Implantable Cardioverter Defibrillator Therapy A Message to Patients Boston Scientific Corporation acquired Guidant Corporation in April 2006. During our transition period, you may see both the Boston
Billing and Coding Guide
 2014 Billing and Coding Guide Contents Links to sections are clickable. To return to the place last viewed: alt+left arrow or right click mouse and select previous view. Introduction Letter to the User
2014 Billing and Coding Guide Contents Links to sections are clickable. To return to the place last viewed: alt+left arrow or right click mouse and select previous view. Introduction Letter to the User
VIVA QUAD XT CRT-D DTBA1QQ
 VIVA QUAD XT CRT-D DTBA1QQ Digital implantable cardioverter defibrillator with cardiac resynchronization therapy (DDE-DDDR) PhysioCurve Design, AdaptivCRT Algorithm, CardioSync Optimization, VectorExpress
VIVA QUAD XT CRT-D DTBA1QQ Digital implantable cardioverter defibrillator with cardiac resynchronization therapy (DDE-DDDR) PhysioCurve Design, AdaptivCRT Algorithm, CardioSync Optimization, VectorExpress
CLINICIAN MANUAL. LATITUDE TM Patient Management System
 CLINICIAN MANUAL LATITUDE TM Patient Management System Table of Contents LATITUDE NXT PATIENT MANAGEMENT INTRODUCTION....................... 5 Intended Use..........................................................
CLINICIAN MANUAL LATITUDE TM Patient Management System Table of Contents LATITUDE NXT PATIENT MANAGEMENT INTRODUCTION....................... 5 Intended Use..........................................................
Establishing a Remote Monitoring Program. Martha Ferrara, FNP
 Establishing a Remote Monitoring Program Martha Ferrara, FNP Establishing a Remote Monitoring Program What is Remote Monitoring? Martha Ferrara, FNP, CCDS November 2012 CIED Timeline: Cardiovascular Implantable
Establishing a Remote Monitoring Program Martha Ferrara, FNP Establishing a Remote Monitoring Program What is Remote Monitoring? Martha Ferrara, FNP, CCDS November 2012 CIED Timeline: Cardiovascular Implantable
2013 Medicare Physician Coding and Reimbursement Changes
 2013 Medicare Physician Coding and Reimbursement Changes Disclaimer This presentation is intended for educational use. Any duplication is prohibited without written consent of Medtronic s Economic Strategies
2013 Medicare Physician Coding and Reimbursement Changes Disclaimer This presentation is intended for educational use. Any duplication is prohibited without written consent of Medtronic s Economic Strategies
Automatic External Defibrillators
 Last Review Date: May 27, 2016 Number: MG.MM.DM.10dC2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth
Last Review Date: May 27, 2016 Number: MG.MM.DM.10dC2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth
Reporting of Devices and Leads When a Credit is Received
 Reporting of Devices and Leads When a Credit is Received Cardiac Rhythm Management and Electrophysiology Updated January 2014 Medicare Reporting Requirements For Full or Partial Credits of Devices and
Reporting of Devices and Leads When a Credit is Received Cardiac Rhythm Management and Electrophysiology Updated January 2014 Medicare Reporting Requirements For Full or Partial Credits of Devices and
Reocor D External Pacemaker
 Cardiac Rhythm Management External Devices Quick Reference Guide Reocor D External Pacemaker Quick Reference Guide Reocor D Note: Before using Reocor D, read its user manual, including all safety instructions.
Cardiac Rhythm Management External Devices Quick Reference Guide Reocor D External Pacemaker Quick Reference Guide Reocor D Note: Before using Reocor D, read its user manual, including all safety instructions.
 Epimed Would Like To Congratulate The 20th Annual Budapest Conference The New Shape of Pain Relief SmalleST. ThiNNeST. CoNTouRed design. Precision Novi is the world s smallest, thinnest 16 contact primary
Epimed Would Like To Congratulate The 20th Annual Budapest Conference The New Shape of Pain Relief SmalleST. ThiNNeST. CoNTouRed design. Precision Novi is the world s smallest, thinnest 16 contact primary
Atrial Fibrillation: Drugs, Ablation, or Benign Neglect. Robert Kennedy, MD October 10, 2015
 Atrial Fibrillation: Drugs, Ablation, or Benign Neglect Robert Kennedy, MD October 10, 2015 Definitions 2014 AHA/ACC/HRS Guideline for the Management of Patients With Atrial Fibrillation: Executive Summary.
Atrial Fibrillation: Drugs, Ablation, or Benign Neglect Robert Kennedy, MD October 10, 2015 Definitions 2014 AHA/ACC/HRS Guideline for the Management of Patients With Atrial Fibrillation: Executive Summary.
Catheter Ablation. A Guided Approach for Treating Atrial Arrhythmias
 Catheter Ablation A Guided Approach for Treating Atrial Arrhythmias A P A T I E N T H A N D B O O K This brochure will provide an overview of atrial arrhythmias (heart rhythm problems affecting the upper
Catheter Ablation A Guided Approach for Treating Atrial Arrhythmias A P A T I E N T H A N D B O O K This brochure will provide an overview of atrial arrhythmias (heart rhythm problems affecting the upper
Basic principles of pacing
 CHAPTER 1 Basic principles of pacing Malcolm Kirk The aim of this chapter is to give sufficient background and information about cardiac pacemakers to allow interpretation of electrocardiograms (ECGs)
CHAPTER 1 Basic principles of pacing Malcolm Kirk The aim of this chapter is to give sufficient background and information about cardiac pacemakers to allow interpretation of electrocardiograms (ECGs)
Medical management of CHF: A New Class of Medication. Al Timothy, M.D. Cardiovascular Institute of the South
 Medical management of CHF: A New Class of Medication Al Timothy, M.D. Cardiovascular Institute of the South Disclosures Speakers Bureau for Amgen Background Chronic systolic congestive heart failure remains
Medical management of CHF: A New Class of Medication Al Timothy, M.D. Cardiovascular Institute of the South Disclosures Speakers Bureau for Amgen Background Chronic systolic congestive heart failure remains
Guidelines for Perioperative Management of Pacemakers and Defibrillators
 Guidelines for Perioperative Management of Pacemakers and Defibrillators Developed By: Deborah Wolbrette, MD, Medical Director, Electrophysiology Lab, Dept of Cardiology Kane High, MD, Department of Anesthesiology
Guidelines for Perioperative Management of Pacemakers and Defibrillators Developed By: Deborah Wolbrette, MD, Medical Director, Electrophysiology Lab, Dept of Cardiology Kane High, MD, Department of Anesthesiology
Versione obsoleta. Non utilizzare. Verouderde versie. Niet gebruiken. Föråldrad version. Använd ej. Versão obsoleta. Não utilize.
 Q-TECH PROGRAMMER A COMPONENT OF THE S-ICD SYSTEM USER S MANUAL 2020EU Copyright 2013 Cameron Health, Inc., San Clemente, CA USA. All rights reserved. Limited Software License and Equipment Use. S-ICD,
Q-TECH PROGRAMMER A COMPONENT OF THE S-ICD SYSTEM USER S MANUAL 2020EU Copyright 2013 Cameron Health, Inc., San Clemente, CA USA. All rights reserved. Limited Software License and Equipment Use. S-ICD,
2015 CPT Codes for Cardiac Device Monitoring
 2015 CPT Codes for Cardiac Device Monitoring CPT 1 copyright 2014. American Medical Association. All rights reserved. CPT is a registered trademark of the American Medical Association. CPT Code IMPLANTABLE
2015 CPT Codes for Cardiac Device Monitoring CPT 1 copyright 2014. American Medical Association. All rights reserved. CPT is a registered trademark of the American Medical Association. CPT Code IMPLANTABLE
2015 Billing and Coding Guide. Rhythm Management
 2015 Billing and Coding Guide Rhythm Management Links to sections are clickable. Introduction Letter to the User i Disclaimer ii GuidePoint Reimbursement Resources at a Glance Intro 1 Medicare Payment
2015 Billing and Coding Guide Rhythm Management Links to sections are clickable. Introduction Letter to the User i Disclaimer ii GuidePoint Reimbursement Resources at a Glance Intro 1 Medicare Payment
Special Topics in Security and Privacy of Medical Information. Reminders. Medical device security. Sujata Garera
 Special Topics in Security and Privacy of Medical Information Sujata Garera Reminders Assignment due today Project part 1 due on next Tuesday Assignment 2 will be online today evening 2nd Discussion session
Special Topics in Security and Privacy of Medical Information Sujata Garera Reminders Assignment due today Project part 1 due on next Tuesday Assignment 2 will be online today evening 2nd Discussion session
GuidePoint. Physician Reimbursement Primer for Cardiac Rhythm Management. With Clinical Case Examples. GuidePoint is simplifying reimbursement.
 GuidePoint GuidePoint is simplifying reimbursement. Physician Reimbursement Primer for Cardiac Rhythm Management With Clinical Case Examples DISCLAIMER Reimbursement information provided by Boston Scientific
GuidePoint GuidePoint is simplifying reimbursement. Physician Reimbursement Primer for Cardiac Rhythm Management With Clinical Case Examples DISCLAIMER Reimbursement information provided by Boston Scientific
Summary of: P890003/S285, P090013/S112, P010031/S397, P010015/S215, P980016/S436, P980035/S343. Background and Summary
 Summary of: P890003/S285, P090013/S112, P010031/S397, P010015/S215, P980016/S436, P980035/S343 Background and Summary This PMA Supplement is to obtain approval for updates to the Medtronic MyCareLink Patient
Summary of: P890003/S285, P090013/S112, P010031/S397, P010015/S215, P980016/S436, P980035/S343 Background and Summary This PMA Supplement is to obtain approval for updates to the Medtronic MyCareLink Patient
Boston Scientific Spinal Cord Stimulation (SCS) Systems
 Boston Scientific Spinal Cord Stimulation (SCS) Systems 91005942-03 REV B Boston Scientific Spinal Cord Stimulator Systems - Guarantees Boston Scientific Corporation reserves the right to modify, without
Boston Scientific Spinal Cord Stimulation (SCS) Systems 91005942-03 REV B Boston Scientific Spinal Cord Stimulator Systems - Guarantees Boston Scientific Corporation reserves the right to modify, without
Purpose: To outline the care of patients with permanent or temporary pacemakers.
 University of Kentucky / UK HealthCare Policy and Procedure Policy # NR08-03 Title/Description: Care of Patients with Pacemakers Purpose: To outline the care of patients with permanent or temporary pacemakers.
University of Kentucky / UK HealthCare Policy and Procedure Policy # NR08-03 Title/Description: Care of Patients with Pacemakers Purpose: To outline the care of patients with permanent or temporary pacemakers.
Activity 4.2.3: EKG. Introduction. Equipment. Procedure
 Activity 4.2.3: EKG The following is used with permission of Vernier Software and Technology. This activity is based on the experiment Analyzing the Heart with EKG from the book Human Physiology with Vernier,
Activity 4.2.3: EKG The following is used with permission of Vernier Software and Technology. This activity is based on the experiment Analyzing the Heart with EKG from the book Human Physiology with Vernier,
CARDIAC ELECTROPHYSIOLOGY, ARRHYTHMIAS AND PACING. Medical Knowledge. Goals and Objectives PF EF MF LF Aspirational
 Know the histology and gross anatomy of the normal sinoatrial node, atrial conduction pathways, atrioventricular (AV) junction and nod, His bundle, conduction fascicles and terminal intra-ventricular conduction
Know the histology and gross anatomy of the normal sinoatrial node, atrial conduction pathways, atrioventricular (AV) junction and nod, His bundle, conduction fascicles and terminal intra-ventricular conduction
Electrophysiology Heart Study - EPS -
 Electrophysiology Heart Study - EPS - What is an EPS? EPS is short for ElectroPhysiology heart Study. This procedure looks at the electrical system of your heart. An EPS will show if you have a heart rhythm
Electrophysiology Heart Study - EPS - What is an EPS? EPS is short for ElectroPhysiology heart Study. This procedure looks at the electrical system of your heart. An EPS will show if you have a heart rhythm
Medtronic Cardiac Rhythm and Heart Failure ICD-10 Coding for Physicians
 Medtronic Cardiac Rhythm and Heart Failure ICD-10 Coding for Physicians May 19, 2015 Disclaimer This presentation is intended for educational use. Any duplication is prohibited without written consent
Medtronic Cardiac Rhythm and Heart Failure ICD-10 Coding for Physicians May 19, 2015 Disclaimer This presentation is intended for educational use. Any duplication is prohibited without written consent
Electrophysiology Daymar College. Lisa H. Young, RN, BSN, MAE 2011
 Electrophysiology Daymar College Lisa H. Young, RN, BSN, MAE 2011 Electrical Conduction Pathway Chemical Basis for Impulse Formation Cardiac Action Potential Phases http://www.youtube.com/watch?v=oqpffilde0e
Electrophysiology Daymar College Lisa H. Young, RN, BSN, MAE 2011 Electrical Conduction Pathway Chemical Basis for Impulse Formation Cardiac Action Potential Phases http://www.youtube.com/watch?v=oqpffilde0e
Implantable pacemakers and implantable cardioverter
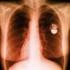 BRIEF REPORT EAS SYSTEMS AND INTERACTIONS WITH IMPLANTABLE CARDIAC DEVICES Electronic Article Surveillance Systems and Interactions With Implantable Cardiac Devices: Risk of Adverse Interactions in Public
BRIEF REPORT EAS SYSTEMS AND INTERACTIONS WITH IMPLANTABLE CARDIAC DEVICES Electronic Article Surveillance Systems and Interactions With Implantable Cardiac Devices: Risk of Adverse Interactions in Public
Reimbursement Assistance: For Reveal ICM products, please call the Medtronic CRHF Coding Hotline at 1 (866) 877-4102
 Product Support Patient Registration Services: Patient Registration Services will be able to answer implant registration-related questions and change a submitted registration: 1 (877) 527-3426 Portal Account
Product Support Patient Registration Services: Patient Registration Services will be able to answer implant registration-related questions and change a submitted registration: 1 (877) 527-3426 Portal Account
PHENYLEPHRINE HYDROCHLORIDE INJECTION USP
 PRESCRIBING INFORMATION PHENYLEPHRINE HYDROCHLORIDE INJECTION USP 10 mg/ml Sandoz Canada Inc. Date of Preparation: September 1992 145 Jules-Léger Date of Revision : January 13, 2011 Boucherville, QC, Canada
PRESCRIBING INFORMATION PHENYLEPHRINE HYDROCHLORIDE INJECTION USP 10 mg/ml Sandoz Canada Inc. Date of Preparation: September 1992 145 Jules-Léger Date of Revision : January 13, 2011 Boucherville, QC, Canada
The Emerging Atrial Fibrillation Epidemic: Treat It, Leave It or Burn It?
 The Emerging Atrial Fibrillation Epidemic: Treat It, Leave It or Burn It? Indiana Chapter-ACC 17 th Annual Meeting Indianapolis, Indiana October 19, 2013 Deepak Bhakta MD FACC FACP FAHA FHRS CCDS Associate
The Emerging Atrial Fibrillation Epidemic: Treat It, Leave It or Burn It? Indiana Chapter-ACC 17 th Annual Meeting Indianapolis, Indiana October 19, 2013 Deepak Bhakta MD FACC FACP FAHA FHRS CCDS Associate
Your pacemaker system information
 Pacemaker Therapy Your pacemaker system information Have your doctor or nurse complete these forms before you go home from the hospital. Pacemaker Model Number: Pacemaker Serial Number: Implant Date:
Pacemaker Therapy Your pacemaker system information Have your doctor or nurse complete these forms before you go home from the hospital. Pacemaker Model Number: Pacemaker Serial Number: Implant Date:
Heart Rhythm UK. Standards for Implantation and Follow-up of Cardiac Rhythm Management Devices HRUK, February 2011
 Heart Rhythm UK Standards for Implantation and Follow-up of Cardiac Rhythm Management Devices HRUK, February 2011 1) Introduction 2) Definitions 3) Treatment indications 4) Requirements for performing
Heart Rhythm UK Standards for Implantation and Follow-up of Cardiac Rhythm Management Devices HRUK, February 2011 1) Introduction 2) Definitions 3) Treatment indications 4) Requirements for performing
Overview. LATITUDE Patient Management. EMR Integration Testing Scenarios - 357742-003
 Contents Overview... 1 Testing Scenarios Overview... 2 Live Production Systems Testing... 3 Preparation...3 Process...4 Post Test Cleanup...4 Expected Results...5 Troubleshooting Tips...5 Insert Message
Contents Overview... 1 Testing Scenarios Overview... 2 Live Production Systems Testing... 3 Preparation...3 Process...4 Post Test Cleanup...4 Expected Results...5 Troubleshooting Tips...5 Insert Message
ATRIAL FIBRILLATION (RATE VS RHYTHM CONTROL)
 ATRIAL FIBRILLATION (RATE VS RHYTHM CONTROL) By Prof. Dr. Helmy A. Bakr Mansoura Universirty 2014 AF Classification: Mechanisms of AF : Selected Risk Factors and Biomarkers for AF: WHY AF? 1. Atrial fibrillation
ATRIAL FIBRILLATION (RATE VS RHYTHM CONTROL) By Prof. Dr. Helmy A. Bakr Mansoura Universirty 2014 AF Classification: Mechanisms of AF : Selected Risk Factors and Biomarkers for AF: WHY AF? 1. Atrial fibrillation
Cardiac Rhythm Management Devices (Part II)
 REVIEW ARTICLE Anesthesiology 2001; 95:1492 1506 2001 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Cardiac Rhythm Management Devices (Part II) Perioperative Management
REVIEW ARTICLE Anesthesiology 2001; 95:1492 1506 2001 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Cardiac Rhythm Management Devices (Part II) Perioperative Management
Atrial Fibrillation: The heart of the matter
 Atrial Fibrillation: The heart of the matter This booklet has been written especially for people with atrial fibrillation (AF), a heart condition often described as an irregular heartbeat (also known
Atrial Fibrillation: The heart of the matter This booklet has been written especially for people with atrial fibrillation (AF), a heart condition often described as an irregular heartbeat (also known
Equine Cardiovascular Disease
 Equine Cardiovascular Disease 3 rd most common cause of poor performance in athletic horses (after musculoskeletal and respiratory) Cardiac abnormalities are rare Clinical Signs: Poor performance/exercise
Equine Cardiovascular Disease 3 rd most common cause of poor performance in athletic horses (after musculoskeletal and respiratory) Cardiac abnormalities are rare Clinical Signs: Poor performance/exercise
Introduction to Electrocardiography. The Genesis and Conduction of Cardiac Rhythm
 Introduction to Electrocardiography Munther K. Homoud, M.D. Tufts-New England Medical Center Spring 2008 The Genesis and Conduction of Cardiac Rhythm Automaticity is the cardiac cell s ability to spontaneously
Introduction to Electrocardiography Munther K. Homoud, M.D. Tufts-New England Medical Center Spring 2008 The Genesis and Conduction of Cardiac Rhythm Automaticity is the cardiac cell s ability to spontaneously
PACEMAKERS AND ANTITACHYCARDIA DEVICES WILLIAM BATSFORD, M.D.
 CHAPTER 26 PACEMAKERS AND ANTITACHYCARDIA DEVICES WILLIAM BATSFORD, M.D. INTRODUCTION The heart s natural pacemaker is an electrical timing device that controls the rate of the heart s muscular contractions,
CHAPTER 26 PACEMAKERS AND ANTITACHYCARDIA DEVICES WILLIAM BATSFORD, M.D. INTRODUCTION The heart s natural pacemaker is an electrical timing device that controls the rate of the heart s muscular contractions,
Special Topics in Security and Privacy of Medical Information. Reminders. Last lecture: Recap. Sujata Garera. Project part 1 submission
 Special Topics in Security and Privacy of Medical Information Sujata Garera Reminders Project part 1 submission Assignment 2 is online Last lecture: Recap Medical Telemetry Infrastructure Devices capturing
Special Topics in Security and Privacy of Medical Information Sujata Garera Reminders Project part 1 submission Assignment 2 is online Last lecture: Recap Medical Telemetry Infrastructure Devices capturing
Rome, Italy December 4-7, 2012 Rome Cavalieri TIMETABLE
 Rome, Italy December 4-7, 2012 Rome Cavalieri Monday, December 3 ROOM Terrazza Monte Mario PATIENT MANAGEMENT CONCEPTS AND CONTROVERSIES IN ELECTROPHYSIOLOGY BIOTRONIK International Fellows Program (IFP)
Rome, Italy December 4-7, 2012 Rome Cavalieri Monday, December 3 ROOM Terrazza Monte Mario PATIENT MANAGEMENT CONCEPTS AND CONTROVERSIES IN ELECTROPHYSIOLOGY BIOTRONIK International Fellows Program (IFP)
QRS Complexes. Fast & Easy ECGs A Self-Paced Learning Program
 6 QRS Complexes Fast & Easy ECGs A Self-Paced Learning Program Q I A ECG Waveforms Normally the heart beats in a regular, rhythmic fashion producing a P wave, QRS complex and T wave I Step 4 of ECG Analysis
6 QRS Complexes Fast & Easy ECGs A Self-Paced Learning Program Q I A ECG Waveforms Normally the heart beats in a regular, rhythmic fashion producing a P wave, QRS complex and T wave I Step 4 of ECG Analysis
50 years of CRM Device Therapy Past, Present and Future. Richard Sutton Professor of Cardiology Imperial College, London, UK
 50 years of CRM Device Therapy Past, Present and Future Richard Sutton Professor of Cardiology Imperial College, London, UK The Past First implant 1958 Sweden Dual Chamber pacing (VAT) 1962 US Transvenous
50 years of CRM Device Therapy Past, Present and Future Richard Sutton Professor of Cardiology Imperial College, London, UK The Past First implant 1958 Sweden Dual Chamber pacing (VAT) 1962 US Transvenous
Assessment, diagnosis and specialist referral of adults (>16 years) with an episode of transient loss of consciousness (TLoC) or a blackout.
 Assessment, diagnosis and specialist referral of adults (>16 years) with an episode of transient loss of consciousness (TLoC) or a blackout. TLoC is common huge variation in management range of clinicians
Assessment, diagnosis and specialist referral of adults (>16 years) with an episode of transient loss of consciousness (TLoC) or a blackout. TLoC is common huge variation in management range of clinicians
Living with Your Pacemaker
 Brief Statement Additional Device Information An implantable pacemaker system relieves symptoms of heart rhythm disturbances. They do this by restoring normal heart rates. A normal heart rate provides
Brief Statement Additional Device Information An implantable pacemaker system relieves symptoms of heart rhythm disturbances. They do this by restoring normal heart rates. A normal heart rate provides
Recurrent AF: Choosing the Right Medication.
 In the name of God Shiraz E-Medical Journal Vol. 11, No. 3, July 2010 http://semj.sums.ac.ir/vol11/jul2010/89015.htm Recurrent AF: Choosing the Right Medication. Basamad Z. * Assistant Professor, Department
In the name of God Shiraz E-Medical Journal Vol. 11, No. 3, July 2010 http://semj.sums.ac.ir/vol11/jul2010/89015.htm Recurrent AF: Choosing the Right Medication. Basamad Z. * Assistant Professor, Department
Advantages of Right-Sided Implantation of Permanent Cardiac Pacemakers Performed by a Surgeon
 Advantages of Right-Sided Implantation of Permanent Cardiac Pacemakers Performed by a Surgeon 7 Davor Horvat General Hospital in Karlovac, Croatia 1. Introduction A pacemaker is a medical device which
Advantages of Right-Sided Implantation of Permanent Cardiac Pacemakers Performed by a Surgeon 7 Davor Horvat General Hospital in Karlovac, Croatia 1. Introduction A pacemaker is a medical device which
NAME OF THE HOSPITAL: 1. Coronary Balloon Angioplasty: M7F1.1/ Angioplasty with Stent(PTCA with Stent): M7F1.3
 1. Coronary Balloon Angioplasty: M7F1.1/ Angioplasty with Stent(PTCA with Stent): M7F1.3 1. Name of the Procedure: Coronary Balloon Angioplasty 2. Select the Indication from the drop down of various indications
1. Coronary Balloon Angioplasty: M7F1.1/ Angioplasty with Stent(PTCA with Stent): M7F1.3 1. Name of the Procedure: Coronary Balloon Angioplasty 2. Select the Indication from the drop down of various indications
2015 ST. JUDE MEDICAL THERAPY CODING GUIDE
 2015 ST. JUDE MEDICAL THERAPY CODING GUIDE Physician Coding for Cardiac Device Monitoring This guide provides physician information for cardiac device monitoring. In addition, St. Jude Medical offers a
2015 ST. JUDE MEDICAL THERAPY CODING GUIDE Physician Coding for Cardiac Device Monitoring This guide provides physician information for cardiac device monitoring. In addition, St. Jude Medical offers a
RATE VERSUS RHYTHM CONTROL OF ATRIAL FIBRILLATION: SPECIAL CONSIDERATION IN ELDERLY. Charles Jazra
 RATE VERSUS RHYTHM CONTROL OF ATRIAL FIBRILLATION: SPECIAL CONSIDERATION IN ELDERLY Charles Jazra NO CONFLICT OF INTEREST TO DECLARE Relationship Between Atrial Fibrillation and Age Prevalence, percent
RATE VERSUS RHYTHM CONTROL OF ATRIAL FIBRILLATION: SPECIAL CONSIDERATION IN ELDERLY Charles Jazra NO CONFLICT OF INTEREST TO DECLARE Relationship Between Atrial Fibrillation and Age Prevalence, percent
SUMMARY OF: P910077/S140 BOSTON SCIENTIFIC LATITUDE COMMUNICATOR MODELS 6460, 6280, 6290, 6250, 6498, AND 6476. Executive Summary
 SUMMARY OF: P910077/S140 BOSTON SCIENTIFIC LATITUDE COMMUNICATOR MODELS 6460, 6280, 6290, 6250, 6498, AND 6476 Executive Summary Boston Scientific is requesting approval for modifications to the LATITUDE
SUMMARY OF: P910077/S140 BOSTON SCIENTIFIC LATITUDE COMMUNICATOR MODELS 6460, 6280, 6290, 6250, 6498, AND 6476 Executive Summary Boston Scientific is requesting approval for modifications to the LATITUDE
ACLS RHYTHM TEST. 2. A 74-year-old woman with chest pain. Blood pressure 192/90 and rates her pain 9/10.
 ACLS RHYTHM TEST Name Date Choose the best answer for each of the following questions. Each of the following strips is 6 seconds in length. 1. Identify the following rhythm a. Sinus bradycardia with 2
ACLS RHYTHM TEST Name Date Choose the best answer for each of the following questions. Each of the following strips is 6 seconds in length. 1. Identify the following rhythm a. Sinus bradycardia with 2
ANNE ARUNDEL MEDICAL CENTER CRITICAL CARE MEDICATION MANUAL DEPARTMENT OF NURSING AND PHARMACY. Guidelines for Use of Intravenous Isoproterenol
 ANNE ARUNDEL MEDICAL CENTER CRITICAL CARE MEDICATION MANUAL DEPARTMENT OF NURSING AND PHARMACY Guidelines for Use of Intravenous Isoproterenol Major Indications Status Asthmaticus As a last resort for
ANNE ARUNDEL MEDICAL CENTER CRITICAL CARE MEDICATION MANUAL DEPARTMENT OF NURSING AND PHARMACY Guidelines for Use of Intravenous Isoproterenol Major Indications Status Asthmaticus As a last resort for
What Can I Do about Atrial Fibrillation (AF)?
 Additional Device Information 9529 Reveal XT Insertable Cardiac Monitor The Reveal XT Insertable Cardiac Monitor is an implantable patientactivated and automatically activated monitoring system that records
Additional Device Information 9529 Reveal XT Insertable Cardiac Monitor The Reveal XT Insertable Cardiac Monitor is an implantable patientactivated and automatically activated monitoring system that records
UnitedHealthcare, UnitedHealthcare of the River Valley and Neighborhood Health Partnership Cardiology Notification and Prior Authorization Program:
 UnitedHealthcare, UnitedHealthcare of the River Valley and Neighborhood Health Partnership Cardiology Notification and Prior Authorization Program: Electrophysiology Implant Code Classification Table The
UnitedHealthcare, UnitedHealthcare of the River Valley and Neighborhood Health Partnership Cardiology Notification and Prior Authorization Program: Electrophysiology Implant Code Classification Table The
Novartis Gilenya FDO Program Clinical Protocol and Highlights from Prescribing Information (PI)
 Novartis Gilenya FDO Program Clinical Protocol and Highlights from Prescribing Information (PI) Highlights from Prescribing Information - the link to the full text PI is as follows: http://www.pharma.us.novartis.com/product/pi/pdf/gilenya.pdf
Novartis Gilenya FDO Program Clinical Protocol and Highlights from Prescribing Information (PI) Highlights from Prescribing Information - the link to the full text PI is as follows: http://www.pharma.us.novartis.com/product/pi/pdf/gilenya.pdf
Christopher M. Wright, MD, MBA Pioneer Cardiovascular Consultants Tempe, Arizona
 Christopher M. Wright, MD, MBA Pioneer Cardiovascular Consultants Tempe, Arizona Areas to be covered Historical, current, and future treatments for various cardiovascular disease: Atherosclerosis (Coronary
Christopher M. Wright, MD, MBA Pioneer Cardiovascular Consultants Tempe, Arizona Areas to be covered Historical, current, and future treatments for various cardiovascular disease: Atherosclerosis (Coronary
Guidelines for deactivating implantable cardioverter defibrillators (ICDs) in people nearing the end of their life
 Guidelines for deactivating implantable cardioverter defibrillators (ICDs) in people nearing the end of their life 1 March 2013 Guidelines for deactivating implantable cardioverter defibrillators (ICDs)
Guidelines for deactivating implantable cardioverter defibrillators (ICDs) in people nearing the end of their life 1 March 2013 Guidelines for deactivating implantable cardioverter defibrillators (ICDs)
The increasing use of implanted cardiac pacemakers (ICP) and cardioverter/defibrillators
 Safety of Electronic Apex Locators and Pulp Testers in Patients With Implanted Cardiac Pacemakers or Cardioverter/Defibrillators Brian L. Wilson, DMD,* Craig Broberg, MD, J. Craig Baumgartner, DDS, PhD,*
Safety of Electronic Apex Locators and Pulp Testers in Patients With Implanted Cardiac Pacemakers or Cardioverter/Defibrillators Brian L. Wilson, DMD,* Craig Broberg, MD, J. Craig Baumgartner, DDS, PhD,*
MADIT-II CLINICAL SUMMARY
 CAUTION: Federal law restricts this device to sale by or on the order of a physician trained or experienced in device implant and follow-up procedures. CLINICAL SUMMARY MADIT-II Boston Scientific Corporation
CAUTION: Federal law restricts this device to sale by or on the order of a physician trained or experienced in device implant and follow-up procedures. CLINICAL SUMMARY MADIT-II Boston Scientific Corporation
