Laboratory Studies and Numerical Modeling of using Natural Micro beads for Environmental Applications
|
|
|
- Brittany Morrison
- 10 years ago
- Views:
Transcription
1 Int. J. Electrochem. Sci., 5 (2010) International Journal of ELECTROCHEMICAL SCIENCE Laboratory Studies and Numerical Modeling of using Natural Micro beads for Environmental Applications M. Elsayed Youssef 2, E. A. Soliman 1, M. A. Abu-Saied 1, 3, M. S. Mohy Eldin 1, Salem S. Al-Deyab 3,El- Refaie Kenawy 3, A. A. Elzatahry 1,* 1 Polymer Materials Research Department, Advanced Technology and New Materials Research Institute, MuCSAT, New Boarg El-Arab City 21934, Alexandria, Egypt 2 Computer-Based Engineering Applications Department, Informatics Research Institute, MuCSAT, New Boarg El-Arab City 21934, Alexandria, Egypt 3 Department of chemistry, Petrochemical research chair, King Saud University, PO Box: 2455 Riyadh 11451, Riyadh, Kingdom of Saudi Arabia * [email protected]; [email protected]. Received: 4 October 2010 / Accepted: 30 October 2010 / Published: 1 December 2010 Micro-beads have been prepared from naturally occurring polymers, sodium alginate (an anionic polysaccharide). Beads forming technique applied in this study is based on the principle that a laminar liquid jet is broken into equally sized droplets by a superimposed vibration. The efficiency of alginate beads for removal of methylene blue (MB) dye from effluents in dynamic batch mode has been investigated. Parameters affecting dye removal such as beads size and ph, and thus adsorption kinetics have been studied. The prepared alginate beads showed a high adsorption capacity for MB, where they removed up to 85% of the initial dye concentration throughout twenty minutes. Moreover, a mathematical model has been developed to explore mechanism removal of dye by these porous polymeric beads. The flow pattern through such porous beads has been resolved using Navier Stock equations and couple the equations of motion with mass balance equations through the multiphysics module (COMSOL). Keywords: Alginate micro-beads, encapsulation, methylene blue, dye removal, mathematical modeling 1. INTRODUCTION Various techniques are being used to remove pollutants from aqueous waste, where sorption is one of the most promising technologies for water purification. The most used adsorbents in this concern including clay minerals [1, 2], activated carbon [3] and polymers [4,5]. Thus, there is a
2 Int. J. Electrochem. Sci., Vol. 5, growing interest in the use of bio-based materials such as polymers obtained from natural resources due to their environmental friendly properties and renewable abundance [6-8]. Alginate is a natural polysaccharide extracted from brown seaweeds. This linear polymer is composed of β-d-mannuronate (M) and α-l-guluronate (G) units linked by β-1, 4 and α-1, 4 glycosidic bonds. M and G units are organized in MM, GG and MG blocks, the proportion of these blocks varying with the source of the polymer. This natural polymer is one of the most extensively studied biopolymers because of being inexpensive, non-toxic and efficient for removal of pollutants from aqueous solution. Therefore, its beads are a common and well-known support material used in bioscience application for immobilization of enzymes [9] and living cells [10]. In the environmental field, alginate beads have been used for the removal of some heavy metal ions and organic pollutants [11-13]. In this work, the impact of various factors controlling the adsorption process of dye on alginate beads such as particle size, time, and ph have been investigated. In this study, the encapsulation process was used to form alginate micro-beads. This technology that developed by Inotech company is based on the principle that a laminar liquid jet is broken into equally sized droplets by a superimposed vibration. In the late 19th century, Lord Rayleigh theoretically analyzed the instability of liquid jets. He showed that the frequency for maximum instability is related to the velocity of the jet and the nozzle diameter. The optimal vibration parameters have to be determined in the light of the incorporated stroboscope. Once determined, the parameters can be reset in the future, making the process highly reproducible. Generally, by using this type of encapsulators, the bead diameter can be set in the range of mm [14]. The adsorption capacity of the prepared alginate micro-beads for methylene blue has been measured, and also mathematical adsorption modelling has been studied. In earlier studies, each of film diffusion, intra-particle pore diffusion, and pore surface diffusion mechanisms have been examined individually or in a combination [15, 16]. This study aimed to study the efficiency of alginate micro-beads for removal of methylene blue dye, optimize the adsorption process, and to verify a mathematical model fitting with the results of the experimental studies. 2. EXPERIMENNTAL PART 2.1. Reagents and Chemicals Cationic MB dye, C 16 H 18 N 3 SCl.3H 2 O, supplied from (NICE CHEMICALS Pvt. Ltd., COCHIN). Sodium alginate (Na-ALG) with a medium viscosity was purchased from Sigma-Aldrich Company. Calcium Chloride (CaCl 2 ) was supplied from Riedel-de Hean Company Apparatus and Procedure 2.3. Beads preparation Sodium alginate solution (1.5%) was prepared by dissolving 1.5 gm of sodium alginate in 100 ml of hot distilled water with stirring until the solution become homogenous. For preparation of beads,
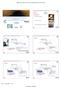
3 Int. J. Electrochem. Sci., Vol. 5, the prepared alginate solution was injected into the encapsulator, where it has the ability to charge the surface of the beads. The voltage applied lies in the range of V. This surface charge transforms the one-dimensional droplet chain in a funnel-like multiline stream. This will prevent beads from hitting each other during flight, or when fall into the cross-linking solution. The process has been described in Figure Dye adsorption Aqueous dye solution was prepared by dissolving 50 mg of methylene blue in 1000 ml distilled water to obtain 50 mg L -1 initial dye concentrations. Adsorption experiments were taken place using an aliquot of 100 ml MB dye solution containing 50 mg L -1 that was mixed with certain amount of alginate micro-beads at room temperature with mechanical stirring at 250 rpm. Samples of 0.5 ml were drawn at time interval 5 min. for 60 min. and placed in test tubes with adding 1.5 ml distilled water to be further analyzed by spectrophotometer. Figure 1: Schematic diagram represents the formation of alginate beads using encapsulator The concentration of MB remaining in the supernatant after and before adsorption was determined with a 1.0 cm light path quartz cells using spectrophotometer at λ max of 665 nm. Prior to the measurement, a calibration curve was obtained by using the standard MB solutions series with known concentrations. The % dye removal is calculated for each sample according to the following relation: % dyes removal = (1 - C/C o ) 100 (1)
4 Int. J. Electrochem. Sci., Vol. 5, Where; C o is the initial concentration of the dye. C is the concentration at time (t). To study the effect of a certain variable on the uptake of the dye, all the other variables are fixed throughout the experiments Beads size measurements The particle size distribution of beads was determined by Beckman Coulter Laser Diffraction Analyzers using photon correlation spectroscopy (PCS), which determines particle size by measuring the rate of fluctuations in laser light intensity scattered by particles as they diffuse through a fluid operating at a 25 mw laser. A constant amount of beads was dispersed in distilled water to prepare a concentration of 1% and was kept at 37 o C under agitation at 150 rpm. 3. RESULTS AND DISCUSSION 3.1. Beads size Alginate has the ability of quick gelling on contact with CaCl 2 solution. The size of the formed beads depends on many factors; Nozzle diameter, charge applied to the beads surface, viscosity and concentration of the used polymeric material. In encapsulation system, the Process allows a very narrow bead size distribution. The desired diameter can be set and controlled by choosing the right nozzle diameter, mass flow and superimposed frequency. It is well known that the beads having small size will possess the high surface area that is associated with high adsorption capacity. For this reason, before dye removal test, the effect of factors affecting beads size distribution were examined to determine the optimum formation conditions resulting beads with smaller size and narrow size distribution. The encapsulation conditions have been settled to produce micro-beads with size average of 500 µm as shown in Figure 2. Figure 3 illustrates the effect of beads size on the dye removal was very prominent, showing a continuous increase of removal efficiency with lowering size of the bead. This of course related to high surface area of the beads with much smaller size and high availability of function groups, which responsible for dye adsorption over beads surface.
5 Int. J. Electrochem. Sci., Vol. 5, Figure 2: Size distribution of the beads Figure 3: Effect of alginate beads size variation on dye removal 3.2. Adsorption Kinetics Figure 4 shows the effect of contact time on the removal of MB of initial concentration (1 mmol L 1). Fast uptake of the dyes has been observed within the first minutes of the adsorption process. Since 50% of dye amount has been approximately removed after 15 min from the experiment
6 Int. J. Electrochem. Sci., Vol. 5, start point and the equilibrium time has been measured to be 150 min of 24 h, the time setup for the experiment. The fast adsorption stage was followed by slower stage to reach its equilibrium value. Figure 4: Adsorbed amount of MB by composite beads (Ct in mmol g-1 wet beads) as a function of time (C0=1 mmol L-1) Several kinetic models are available to study the mechanisms of the sorption process and to fit the experimental data. Among them the pseudo-second-order equation is often successfully used to describe the kinetics of the fixation reaction of pollutants on the adsorbent. dqt k dt Q Q 2 eq t (2) Where k (gmmol 1 min 1 ) is the second-order rate constant, Qeq (mmol g 1, wet bead) is the amount of dye adsorbed at equilibrium and Qt (mmol g 1, wet bead) is the amount of dye adsorbed at any time t (min). Integrating this equation with the boundary conditions t=0 (Qt =0) to teq (Qt =Qeq) and then linearizing leads to t Q t 1 kq 2 eq 1 Q eq t (3) Where constant, k and Qeq values were calculated from the slope and intercepts of the t/q t versus t plot. A linear relationship with high correlation coefficients was observed between t/q t and t, indicating the applicability of the pseudo-second-order model to describe the sorption process. The
7 Int. J. Electrochem. Sci., Vol. 5, correlation coefficients R 2, the second-order constants k and the calculated and experimental equilibrium sorption capacities Q eq has been shown in Table 1. Table 1: Adsorption kinetic constants, modelled by a pseudo-second-order equation C 0 t eq Q eq, exp Q eq, calc k (mmol L _1 ) (min) (mmol g _1 ) (mmol g _1 ) (gmmol _1 min _1 ) (min) X X t 50 R Effect of ph Figure 5 shows the effect of ph on the removal of dye by composite beads, the results shows that no apparent effect of ph on the removal of dye, this observation reflects the fact that the removal of dye is mainly due to the presence of AC which is not a ph sensitive. Figure 5: Effect of ph on the removal of MB dyes by composite beads (C0=1 mmol L-1) 3.4. Performance modeling and analysis Porous particles are widely used in the chemical industry and are extensively treated in the chemical engineering. The beads are in most cases fluidized the action of gas or liquid flowing through a reactor. The fluid enters at the bottom of a bed of particles and the particles are fluidized or made to
8 Int. J. Electrochem. Sci., Vol. 5, (float) by the shear force that the fluid exerts on their surface, at the same time, species dissolved in the fluid reaction on the surface and inside the porous particles. In this study, cross-section of spherical polymer particle has been modeled. The spherical polymer particles are subjected to the dye which will be removed. The flow pattern and concentration field surrounding the spherical polymer particle and concentration field inside and outside the particle will be resolved Model Definition The spherical polymer particles transposed to dye which will be removed is usually solved by assuming a uniform thick diffusion layer around the porous particle, which requires amass transfer coefficient to describe transport through the diffusion layer. The mass transfer coefficient as determined from the wall structure of the particle and transport properties of the fluid. The flow pattern around the porous particle will be resolved using Navier Stock equations and couple the equations of motion and the mass balance equations through the multiphysics module (COMSOL)[17]. The structure of the diffusion layer will be known from the simultaneous solution of these equations. Symmetry is assumed at the vertical boundaries, implying symmetry in the y-axis and that the modeled unit cell is typical for the system.the fluid density will be assumed constant. The set of equation which will be used in the fluid sub domain denoted by Φ f are µ 2 u+ρ (u. )u+ p=0 in Φ f. (4) u=0. in Φ f (5) Where µ denotes the dynamic viscosity (kg/m.s), u is the velocity vector (m/s),ρ is the fluid density(kg/m 3 ) and p denotes pressure (pa). The coupling of motion equation to the mass transport equation, for the reactant in he fluid at steady-state, through:. (-D C+CU)=0. in Φ f (6) Where D denotes the diffusion coefficient (m 2 /s) and C the concentration (mol/ m 3 ) The mass balance in the pellet by using an effective diffusion coefficient in the porous media is defined as following:.(d eff C) -KC 2 )=0 in Φ pl (7) Where, D eff denotes the effective diffusion coefficient (m 2 /s) and K the rate constant fir the second order reaction (m 3 /s.mol). D eff is related to the porosity and tortuosity of the pellet.
9 Int. J. Electrochem. Sci., Vol. 5, The boundary conditions of the fluid flow as following: u.n=u o at inlet (8) u.n=0 at δφ fl (9) u =(0,0) at δφ pl.1 (10) p=0 at δφ outlet (11) Where δφ pl is sub domain boundary, since the Navier Stockes equation and continuity equations are not defined in the pellet domain. The boundary conditions for the mass transport equation can be defined by assuming that the inlet concentration of the die in water is known C = C o at δφ inlet (12) (-D C+CU). N = 0 at δφ f,1 and δφ f,2 (13) (D eff C).n = 0 at δφ pl (14) (-D C+CU).n = Cu.n (15) Figure 6: Schematic diagram of the domain that will be modeled. Figure 6 shows a schematic representation of the domain that will be modeled. The dye is introduced at the bottom of the picture and exits at the top. Symmetry is assumed at the vertical boundaries, implying symmetry in the y-axis and that the modeled unit cell is typical for the system. Furthermore, It will be assumed that the density of the fluid does not change due to the sorption process on the beads.
10 Int. J. Electrochem. Sci., Vol. 5, Figure 7 shows the diffusion layer, in the free fluid surrounding the particle, is of non uniform thickness. This results in a non-symmetrical concentration distribution along the outer surface of the particle. The diffusion layer at the particle surface, facing the inlet of the dye, is far thinner than that obtained behind the particle. Moreover, depletion of the dye, along the main direction of flow, influences the concentration around the surface of the particle. It is seen from figure 8 that the maximum velocity occurs in the contraction between the particle and the symmetry line on the right side. There are two stagnation zones in front and behind the particle. Figure 7: Contour lines of concentration Figure 8: Flow pattern of dye around the beads
11 Int. J. Electrochem. Sci., Vol. 5, CONCLUSION In the last two decades, many researches have been taken place on the removal of environmental pollutants using adsorption on various absorbents either those from agro-waste or those extracted from renewable resources such as biopolymers because of being inexpensive and efficient items in this respect. Our study has focused on forming alginate micro-beads using encapsulation technique, the prepared beads exhibited high adsorption capacity for MB where removed 85% of the dye content within twenty minutes. Significant effect has been exhibited for the size of alginate beads on adsorption capacity for MB. However, there is no significant effect has been shown for ph. Notwithstanding, little researches have been carried out on the modeling and simulation of these processes. The mathematical model developed in this study indicated that the dye removal by these micro-beads hasn t just based on the surface adsorption, but MB dye interpenetrates the internal layers of the beads. References 1. M. Dogan et al., Journal of Hazardous Materials 109 (1 3) (2004), pp P. Liu and L. Zhang, Separation and Purification Technology 58 (1) (2007), pp J.M. Dias et al., Waste materials for activated carbon preparation and its use in aqueous-phase treatment: a review, Journal of Environmental Management 85 (4) (2007), pp N.K. Lazaridis et al., Chitosan derivatives as biosorbents for basic dyes, Langmuir (2007). 5. S. Mondal, Methods of dye removal from dye house effluents. An overview, Environmental Engineering Science 25 (3) (2008), pp G. Crini, Recent developments in polysaccharide-based materials used as adsorbents in wastewater treatment, Progress in Polymer Science 30 (1) (2005), pp G. Crini, Non-conventional low-cost adsorbents for dye removal: a review, Bioresource Technology 97 (9) (2006), pp F. Delval et al., Preparation, characterization and sorption properties of crosslinked starch-based exchangers, Carbohydrate Polymers 60 (1) (2005), pp Ribeiro, C.C., Barrias, C.C., et al., Biomaterials 25 (18), Smidsrod, O., Skjak-Brk, G., Trends Biotechnol. 8, Aravindhan, R., Fathima, N.N., et al., Colloids Surf. A: Physicochem. Eng. Aspects 299 (1 3), Nasr, M.F., El-Ola, S.M.A., et al., Polymer-Plastics Technol. Eng. 45 (3), Vincent Rocher, Jean-Michel Siaugue, Vale rie Cabuil, Agne`s Bee, Water Research 42 (2008) INOTECH Biotechnologies Ltd, Breuher, L.E., Frantz, D.C., and Kostecki, J.A., Combined Diffusion in Batch Adsorption Systems Displaying BET Isotherm Part II, American institute of chemical Engineering Symposium Series, Vol. 63, p. 251, Crittenden, J.C., Mathematical Modeling of Absorber Dynamics-Single Components and Multi- Components, Ph.D. Thesis, Michigan University, USA, Elzatahry,A.A., Hassan, H.M., Yousse,M.E, Int. J. Electrochem. Sci., 5 (2010) by ESG (
Removal of Sulfate from Waste Water by Activated Carbon. Mohammed Sadeq Salman Computer Centre/ University of Baghdad
 Al-Khwarizmi Engineering Journal, Vol. 5, No. 3, PP 72-76 (29) Al-Khwarizmi Engineering Journal Removal of Sulfate from Waste Water by Activated Carbon Mohammed Sadeq Salman Computer Centre/ University
Al-Khwarizmi Engineering Journal, Vol. 5, No. 3, PP 72-76 (29) Al-Khwarizmi Engineering Journal Removal of Sulfate from Waste Water by Activated Carbon Mohammed Sadeq Salman Computer Centre/ University
REMOVAL OF PHOSPHATE FROM WASTEWATER USING LOW-COST ADSORBENTS
 International Journal of Engineering Inventions ISSN: 2278-7461, www.ijeijournal.com Volume 1, Issue 7 (October2012) PP: 44-50 REMOVAL OF PHOSPHATE FROM WASTEWATER USING LOW-COST ADSORBENTS Dr. C.R.Ramakrishnaiah
International Journal of Engineering Inventions ISSN: 2278-7461, www.ijeijournal.com Volume 1, Issue 7 (October2012) PP: 44-50 REMOVAL OF PHOSPHATE FROM WASTEWATER USING LOW-COST ADSORBENTS Dr. C.R.Ramakrishnaiah
Theoretical and Experimental Modeling of Multi-Species Transport in Soils Under Electric Fields
 United States National Risk Management Environmental Protection Research Laboratory Agency Cincinnati, OH 45268 Research and Development EPA/6/SR-97/54 August 997 Project Summary Theoretical and Experimental
United States National Risk Management Environmental Protection Research Laboratory Agency Cincinnati, OH 45268 Research and Development EPA/6/SR-97/54 August 997 Project Summary Theoretical and Experimental
GRANULAR ACTIVATED CARBON FOR WATER & WASTEWATER TREATMENT
 GRANULAR ACTIVATED CARBON FOR WATER & WASTEWATER TREATMENT CARBTROL Corporation September 1992 Rev. 10/92 TP-3 INTRODUCTION Carbon has been used as an adsorbent for centuries. Early uses of carbon were
GRANULAR ACTIVATED CARBON FOR WATER & WASTEWATER TREATMENT CARBTROL Corporation September 1992 Rev. 10/92 TP-3 INTRODUCTION Carbon has been used as an adsorbent for centuries. Early uses of carbon were
POLLUTED EMISSION TREATMENTS FROM INCINERATOR GASES
 POLLUTED EMISSION TREATMENTS FROM INCINERATOR GASES Ecole Nationale Supérieure de Chimie Avenue du Général Leclerc, Campus de Beaulieu 35700 Rennes, France Tel 33 (0)2 23 23 80 02 Fax 33 (0)2 23 23 81
POLLUTED EMISSION TREATMENTS FROM INCINERATOR GASES Ecole Nationale Supérieure de Chimie Avenue du Général Leclerc, Campus de Beaulieu 35700 Rennes, France Tel 33 (0)2 23 23 80 02 Fax 33 (0)2 23 23 81
CHEMICAL ENGINEERING AND CHEMICAL PROCESS TECHNOLOGY - Vol. I - Interphase Mass Transfer - A. Burghardt
 INTERPHASE MASS TRANSFER A. Burghardt Institute of Chemical Engineering, Polish Academy of Sciences, Poland Keywords: Turbulent flow, turbulent mass flux, eddy viscosity, eddy diffusivity, Prandtl mixing
INTERPHASE MASS TRANSFER A. Burghardt Institute of Chemical Engineering, Polish Academy of Sciences, Poland Keywords: Turbulent flow, turbulent mass flux, eddy viscosity, eddy diffusivity, Prandtl mixing
DEVELOPMENT OF HIGH SPEED RESPONSE LAMINAR FLOW METER FOR AIR CONDITIONING
 DEVELOPMENT OF HIGH SPEED RESPONSE LAMINAR FLOW METER FOR AIR CONDITIONING Toshiharu Kagawa 1, Yukako Saisu 2, Riki Nishimura 3 and Chongho Youn 4 ABSTRACT In this paper, we developed a new laminar flow
DEVELOPMENT OF HIGH SPEED RESPONSE LAMINAR FLOW METER FOR AIR CONDITIONING Toshiharu Kagawa 1, Yukako Saisu 2, Riki Nishimura 3 and Chongho Youn 4 ABSTRACT In this paper, we developed a new laminar flow
Texture characteristic of membrane materials ASAP, BET
 Texture characteristic of membrane materials ASAP, BET Theory Specific surface (surface area) is used for the characterization of many materials. There are various techniques how measure the specific surface
Texture characteristic of membrane materials ASAP, BET Theory Specific surface (surface area) is used for the characterization of many materials. There are various techniques how measure the specific surface
Diffusion and Fluid Flow
 Diffusion and Fluid Flow What determines the diffusion coefficient? What determines fluid flow? 1. Diffusion: Diffusion refers to the transport of substance against a concentration gradient. ΔS>0 Mass
Diffusion and Fluid Flow What determines the diffusion coefficient? What determines fluid flow? 1. Diffusion: Diffusion refers to the transport of substance against a concentration gradient. ΔS>0 Mass
SORPTION ISOTHERM STUDY ON TWO POLYAMIDE NANOFIBEROUS MEMBRANES. Yan WANG, Jakub WIENER, Guocheng ZHU
 SORPTION ISOTHERM STUDY ON TWO POLYAMIDE NANOFIBEROUS MEMBRANES Yan WANG, Jakub WIENER, Guocheng ZHU Technical University of Liberec, Faculty of Textile Engineering, Department of Textile Materials, Liberec,
SORPTION ISOTHERM STUDY ON TWO POLYAMIDE NANOFIBEROUS MEMBRANES Yan WANG, Jakub WIENER, Guocheng ZHU Technical University of Liberec, Faculty of Textile Engineering, Department of Textile Materials, Liberec,
NUMERICAL ANALYSIS OF THE EFFECTS OF WIND ON BUILDING STRUCTURES
 Vol. XX 2012 No. 4 28 34 J. ŠIMIČEK O. HUBOVÁ NUMERICAL ANALYSIS OF THE EFFECTS OF WIND ON BUILDING STRUCTURES Jozef ŠIMIČEK email: [email protected] Research field: Statics and Dynamics Fluids mechanics
Vol. XX 2012 No. 4 28 34 J. ŠIMIČEK O. HUBOVÁ NUMERICAL ANALYSIS OF THE EFFECTS OF WIND ON BUILDING STRUCTURES Jozef ŠIMIČEK email: [email protected] Research field: Statics and Dynamics Fluids mechanics
Paper No. 4071 APPLICATION OF EQCM TO THE STUDY OF CO2 CORROSION
 Paper No. 471 APPLICATION OF EQCM TO THE STUDY OF CO2 CORROSION Yang Yang, Bruce Brown and Srdjan Nešić Institute for Corrosion and Multiphase Technology, Department of Chemical and Biomolecular Engineering
Paper No. 471 APPLICATION OF EQCM TO THE STUDY OF CO2 CORROSION Yang Yang, Bruce Brown and Srdjan Nešić Institute for Corrosion and Multiphase Technology, Department of Chemical and Biomolecular Engineering
7. DYNAMIC LIGHT SCATTERING 7.1 First order temporal autocorrelation function.
 7. DYNAMIC LIGHT SCATTERING 7. First order temporal autocorrelation function. Dynamic light scattering (DLS) studies the properties of inhomogeneous and dynamic media. A generic situation is illustrated
7. DYNAMIC LIGHT SCATTERING 7. First order temporal autocorrelation function. Dynamic light scattering (DLS) studies the properties of inhomogeneous and dynamic media. A generic situation is illustrated
TDS. Dirk Rosenthal Department of Inorganic Chemistry Fritz-Haber-Institut der MPG Faradayweg 4-6, DE 14195 Berlin [email protected].
 Modern Methods in Heterogeneous Catalysis Research TDS Dirk Rosenthal Department of Inorganic Chemistry Fritz-Haber-Institut der MPG Faradayweg 4-6, DE 14195 Berlin [email protected] TDS = TPD
Modern Methods in Heterogeneous Catalysis Research TDS Dirk Rosenthal Department of Inorganic Chemistry Fritz-Haber-Institut der MPG Faradayweg 4-6, DE 14195 Berlin [email protected] TDS = TPD
HEAT TRANSFER ANALYSIS IN A 3D SQUARE CHANNEL LAMINAR FLOW WITH USING BAFFLES 1 Vikram Bishnoi
 HEAT TRANSFER ANALYSIS IN A 3D SQUARE CHANNEL LAMINAR FLOW WITH USING BAFFLES 1 Vikram Bishnoi 2 Rajesh Dudi 1 Scholar and 2 Assistant Professor,Department of Mechanical Engineering, OITM, Hisar (Haryana)
HEAT TRANSFER ANALYSIS IN A 3D SQUARE CHANNEL LAMINAR FLOW WITH USING BAFFLES 1 Vikram Bishnoi 2 Rajesh Dudi 1 Scholar and 2 Assistant Professor,Department of Mechanical Engineering, OITM, Hisar (Haryana)
AN EFFECT OF GRID QUALITY ON THE RESULTS OF NUMERICAL SIMULATIONS OF THE FLUID FLOW FIELD IN AN AGITATED VESSEL
 14 th European Conference on Mixing Warszawa, 10-13 September 2012 AN EFFECT OF GRID QUALITY ON THE RESULTS OF NUMERICAL SIMULATIONS OF THE FLUID FLOW FIELD IN AN AGITATED VESSEL Joanna Karcz, Lukasz Kacperski
14 th European Conference on Mixing Warszawa, 10-13 September 2012 AN EFFECT OF GRID QUALITY ON THE RESULTS OF NUMERICAL SIMULATIONS OF THE FLUID FLOW FIELD IN AN AGITATED VESSEL Joanna Karcz, Lukasz Kacperski
SYNERGISTIC APPLICATION OF ADVANCED PRIMARY AND SECONDARY WASTEWATER TREATMENT SYSTEMS
 SYNERGISTIC APPLICATION OF ADVANCED PRIMARY AND SECONDARY WASTEWATER TREATMENT SYSTEMS Published in Water and Waste Digest membrane issue, November 2008 Miroslav Colic; Chief Scientist, Clean Water Technology
SYNERGISTIC APPLICATION OF ADVANCED PRIMARY AND SECONDARY WASTEWATER TREATMENT SYSTEMS Published in Water and Waste Digest membrane issue, November 2008 Miroslav Colic; Chief Scientist, Clean Water Technology
CFD SIMULATION OF SDHW STORAGE TANK WITH AND WITHOUT HEATER
 International Journal of Advancements in Research & Technology, Volume 1, Issue2, July-2012 1 CFD SIMULATION OF SDHW STORAGE TANK WITH AND WITHOUT HEATER ABSTRACT (1) Mr. Mainak Bhaumik M.E. (Thermal Engg.)
International Journal of Advancements in Research & Technology, Volume 1, Issue2, July-2012 1 CFD SIMULATION OF SDHW STORAGE TANK WITH AND WITHOUT HEATER ABSTRACT (1) Mr. Mainak Bhaumik M.E. (Thermal Engg.)
Removal of Liquid Water Droplets in Gas Channels of Proton Exchange Membrane Fuel Cell
 第 五 届 全 球 华 人 航 空 科 技 研 讨 会 Removal of Liquid Water Droplets in Gas Channels of Proton Exchange Membrane Fuel Cell Chin-Hsiang Cheng 1,*, Wei-Shan Han 1, Chun-I Lee 2, Huan-Ruei Shiu 2 1 Department of
第 五 届 全 球 华 人 航 空 科 技 研 讨 会 Removal of Liquid Water Droplets in Gas Channels of Proton Exchange Membrane Fuel Cell Chin-Hsiang Cheng 1,*, Wei-Shan Han 1, Chun-I Lee 2, Huan-Ruei Shiu 2 1 Department of
Integration of a fin experiment into the undergraduate heat transfer laboratory
 Integration of a fin experiment into the undergraduate heat transfer laboratory H. I. Abu-Mulaweh Mechanical Engineering Department, Purdue University at Fort Wayne, Fort Wayne, IN 46805, USA E-mail: [email protected]
Integration of a fin experiment into the undergraduate heat transfer laboratory H. I. Abu-Mulaweh Mechanical Engineering Department, Purdue University at Fort Wayne, Fort Wayne, IN 46805, USA E-mail: [email protected]
Removing Thallium from Industrial FGD Scrubber Water with Sorbster Adsorbent Media
 Case History MAR Systems Inc. Removing Thallium from Industrial FGD Scrubber Water with Sorbster Adsorbent Media Trace thallium levels in process and wastewater streams pose a human toxicity threat. Tidwell
Case History MAR Systems Inc. Removing Thallium from Industrial FGD Scrubber Water with Sorbster Adsorbent Media Trace thallium levels in process and wastewater streams pose a human toxicity threat. Tidwell
Heterogeneous Catalysis and Catalytic Processes Prof. K. K. Pant Department of Chemical Engineering Indian Institute of Technology, Delhi
 Heterogeneous Catalysis and Catalytic Processes Prof. K. K. Pant Department of Chemical Engineering Indian Institute of Technology, Delhi Module - 03 Lecture 10 Good morning. In my last lecture, I was
Heterogeneous Catalysis and Catalytic Processes Prof. K. K. Pant Department of Chemical Engineering Indian Institute of Technology, Delhi Module - 03 Lecture 10 Good morning. In my last lecture, I was
Universidade Fernando Pessoa - CIAGEB, Praça de 9 de Abril 349, 4249-004 Porto, Portugal 2
 1 Universidade Fernando Pessoa - CIAGEB, Praça de 9 de Abril 349, 4249-004 Porto, Portugal 2 Academia das Ciências de Lisboa, Rua da Academia das Ciências 19, 1249-122 Lisboa, Portugal 16-18 Sept 2009
1 Universidade Fernando Pessoa - CIAGEB, Praça de 9 de Abril 349, 4249-004 Porto, Portugal 2 Academia das Ciências de Lisboa, Rua da Academia das Ciências 19, 1249-122 Lisboa, Portugal 16-18 Sept 2009
Coupling Forced Convection in Air Gaps with Heat and Moisture Transfer inside Constructions
 Coupling Forced Convection in Air Gaps with Heat and Moisture Transfer inside Constructions M. Bianchi Janetti 1, F. Ochs 1 and R. Pfluger 1 1 University of Innsbruck, Unit for Energy Efficient Buildings,
Coupling Forced Convection in Air Gaps with Heat and Moisture Transfer inside Constructions M. Bianchi Janetti 1, F. Ochs 1 and R. Pfluger 1 1 University of Innsbruck, Unit for Energy Efficient Buildings,
Effect of the Hydrodynamic Conditions on the Electrochemical Degradation of Phenol on a BDD Anode
 Portugaliae Electrochimica Acta 2011, 29(5), 343-348 DOI: 1152/pea.201105343 PORTUGALIAE ELECTROCHIMICA ACTA ISSN 1647-1571 Effect of the Hydrodynamic Conditions on the Electrochemical Degradation of Phenol
Portugaliae Electrochimica Acta 2011, 29(5), 343-348 DOI: 1152/pea.201105343 PORTUGALIAE ELECTROCHIMICA ACTA ISSN 1647-1571 Effect of the Hydrodynamic Conditions on the Electrochemical Degradation of Phenol
DETERMINATION OF THE HEAT STORAGE CAPACITY OF PCM AND PCM-OBJECTS AS A FUNCTION OF TEMPERATURE. E. Günther, S. Hiebler, H. Mehling
 DETERMINATION OF THE HEAT STORAGE CAPACITY OF PCM AND PCM-OBJECTS AS A FUNCTION OF TEMPERATURE E. Günther, S. Hiebler, H. Mehling Bavarian Center for Applied Energy Research (ZAE Bayern) Walther-Meißner-Str.
DETERMINATION OF THE HEAT STORAGE CAPACITY OF PCM AND PCM-OBJECTS AS A FUNCTION OF TEMPERATURE E. Günther, S. Hiebler, H. Mehling Bavarian Center for Applied Energy Research (ZAE Bayern) Walther-Meißner-Str.
Enhanced Organic Precursor Removals Using Aged Filter Media Page 1. Enhanced Organic Precursor Removals Using Aged Filter Media
 Enhanced Organic Precursor Removals Using Aged Media Page 1 Enhanced Organic Precursor Removals Using Aged Media O b j e c t i v e s The main goals of this project was to evaluate the dissolved organic
Enhanced Organic Precursor Removals Using Aged Media Page 1 Enhanced Organic Precursor Removals Using Aged Media O b j e c t i v e s The main goals of this project was to evaluate the dissolved organic
Introduction to COMSOL. The Navier-Stokes Equations
 Flow Between Parallel Plates Modified from the COMSOL ChE Library module rev 10/13/08 Modified by Robert P. Hesketh, Chemical Engineering, Rowan University Fall 2008 Introduction to COMSOL The following
Flow Between Parallel Plates Modified from the COMSOL ChE Library module rev 10/13/08 Modified by Robert P. Hesketh, Chemical Engineering, Rowan University Fall 2008 Introduction to COMSOL The following
EXPERIMENTAL METHODS IN COLLOIDS AND SURFACES
 EXPERIMENTAL METHODS IN COLLOIDS AND SURFACES PARTICLE SURFACE AREA FROM GAS ADSORPTION TYPES OF ADSORPTION Physical adsorption: rapid, depends on adsorbate bulk concentration, multiple molecular layers
EXPERIMENTAL METHODS IN COLLOIDS AND SURFACES PARTICLE SURFACE AREA FROM GAS ADSORPTION TYPES OF ADSORPTION Physical adsorption: rapid, depends on adsorbate bulk concentration, multiple molecular layers
Radial-axial Radial mixing is based on the premise that the fluids to be mixed enter the mixer in the correct proportions simultaneously
 Brochure E-0501 1 Introduction. Static mixers are used for a wide range of applications including mixing, heat exchange and dispersion, due to numerous unique innovations our products are especially suitable
Brochure E-0501 1 Introduction. Static mixers are used for a wide range of applications including mixing, heat exchange and dispersion, due to numerous unique innovations our products are especially suitable
Physical & Chemical Properties. Properties
 Physical & Chemical Properties Properties Carbon black can be broadly defined as very fine particulate aggregates of carbon possessing an amorphous quasi-graphitic molecular structure. The most significant
Physical & Chemical Properties Properties Carbon black can be broadly defined as very fine particulate aggregates of carbon possessing an amorphous quasi-graphitic molecular structure. The most significant
Lecture 24 - Surface tension, viscous flow, thermodynamics
 Lecture 24 - Surface tension, viscous flow, thermodynamics Surface tension, surface energy The atoms at the surface of a solid or liquid are not happy. Their bonding is less ideal than the bonding of atoms
Lecture 24 - Surface tension, viscous flow, thermodynamics Surface tension, surface energy The atoms at the surface of a solid or liquid are not happy. Their bonding is less ideal than the bonding of atoms
The atomic packing factor is defined as the ratio of sphere volume to the total unit cell volume, or APF = V S V C. = 2(sphere volume) = 2 = V C = 4R
 3.5 Show that the atomic packing factor for BCC is 0.68. The atomic packing factor is defined as the ratio of sphere volume to the total unit cell volume, or APF = V S V C Since there are two spheres associated
3.5 Show that the atomic packing factor for BCC is 0.68. The atomic packing factor is defined as the ratio of sphere volume to the total unit cell volume, or APF = V S V C Since there are two spheres associated
Chemical Waves in the Belousov-Zhabotinsky Reaction: Determining a Rate Constant with a Ruler
 Chemical Waves in the Belousov-Zhabotinsky Reaction: Determining a Rate Constant with a Ruler John A. Pojman Department of Chemistry and Biochemistry, The University of Southern Mississippi, Hattiesburg,
Chemical Waves in the Belousov-Zhabotinsky Reaction: Determining a Rate Constant with a Ruler John A. Pojman Department of Chemistry and Biochemistry, The University of Southern Mississippi, Hattiesburg,
Chapter 12 IVP Practice Problems
 PRACTICE PROBLEMS 43 Chapter IVP Practice Problems Use Excel and VBA to solve the following problems. Document your solutions using the Expert Problem Solving steps outlined in Table... Find an approximate
PRACTICE PROBLEMS 43 Chapter IVP Practice Problems Use Excel and VBA to solve the following problems. Document your solutions using the Expert Problem Solving steps outlined in Table... Find an approximate
Numerical Analysis of the Resin Transfer Molding Process via PAM- RTM Software
 Defect and Diffusion Forum Vol 365 (2015) pp 88-93 (2015) Trans Tech Publications, Switzerland doi:10.4028/www.scientific.net/ddf.365.88 Numerical Analysis of the Resin Transfer Molding Process via PAM-
Defect and Diffusion Forum Vol 365 (2015) pp 88-93 (2015) Trans Tech Publications, Switzerland doi:10.4028/www.scientific.net/ddf.365.88 Numerical Analysis of the Resin Transfer Molding Process via PAM-
Effect of surface area, pore volume and particle size of P25 titania on the phase transformation of anatase to rutile
 Indian Journal of Chemistry Vol. 48A, October 2009, pp. 1378-1382 Notes Effect of surface area, pore volume and particle size of P25 titania on the phase transformation of anatase to rutile K Joseph Antony
Indian Journal of Chemistry Vol. 48A, October 2009, pp. 1378-1382 Notes Effect of surface area, pore volume and particle size of P25 titania on the phase transformation of anatase to rutile K Joseph Antony
DOWEX Resins as Organic Solvent Desiccants
 Product Information DOWEX Resins as Organic Solvent Desiccants DOWEX* ion exchange resins can be used as desiccants for organic solvents, after having been dried to a low moisture level, in a manner similar
Product Information DOWEX Resins as Organic Solvent Desiccants DOWEX* ion exchange resins can be used as desiccants for organic solvents, after having been dried to a low moisture level, in a manner similar
Problem Set 6 UV-Vis Absorption Spectroscopy. 13-1. Express the following absorbances in terms of percent transmittance:
 Problem Set 6 UV-Vis Absorption Spectroscopy 13-1. Express the following absorbances in terms of percent transmittance: a 0.051 b 0.918 c 0.379 d 0.261 e 0.485 f 0.072 A = log P o /P = log1/t = - log T
Problem Set 6 UV-Vis Absorption Spectroscopy 13-1. Express the following absorbances in terms of percent transmittance: a 0.051 b 0.918 c 0.379 d 0.261 e 0.485 f 0.072 A = log P o /P = log1/t = - log T
Guide to Reverse Phase SpinColumns Chromatography for Sample Prep
 Guide to Reverse Phase SpinColumns Chromatography for Sample Prep www.harvardapparatus.com Contents Introduction...2-3 Modes of Separation...4-6 Spin Column Efficiency...7-8 Fast Protein Analysis...9 Specifications...10
Guide to Reverse Phase SpinColumns Chromatography for Sample Prep www.harvardapparatus.com Contents Introduction...2-3 Modes of Separation...4-6 Spin Column Efficiency...7-8 Fast Protein Analysis...9 Specifications...10
Experiment 5: Phase diagram for a three-component system (Dated: April 12, 2010)
 Experiment 5: Phase diagram for a three-component system (Dated: April 12, 2010) I. INTRODUCTION It is sometimes necessary to know the mutual solubilities of liquids in a two-phase system. For example,
Experiment 5: Phase diagram for a three-component system (Dated: April 12, 2010) I. INTRODUCTION It is sometimes necessary to know the mutual solubilities of liquids in a two-phase system. For example,
Chemical Engineering - CHEN
 Auburn University 1 Chemical Engineering - CHEN Courses CHEN 2100 PRINCIPLES OF CHEMICAL ENGINEERING (4) LEC. 3. LAB. 3. Pr. (CHEM 1110 or CHEM 1117 or CHEM 1030) and (MATH 1610 or MATH 1613 or MATH 1617
Auburn University 1 Chemical Engineering - CHEN Courses CHEN 2100 PRINCIPLES OF CHEMICAL ENGINEERING (4) LEC. 3. LAB. 3. Pr. (CHEM 1110 or CHEM 1117 or CHEM 1030) and (MATH 1610 or MATH 1613 or MATH 1617
Adsorption. December 2014
 Adsorption December 2014 1 Nanosized objects have a large surface area Dividing the size of an object by 2... doubles the accessible surface 2 Adsorption Absorption Absorption is a phenomenon that occurs
Adsorption December 2014 1 Nanosized objects have a large surface area Dividing the size of an object by 2... doubles the accessible surface 2 Adsorption Absorption Absorption is a phenomenon that occurs
IMWA 2010 Sydney, Nova Scotia Mine Water & Innovative Thinking. 3 main divisions : Slide 2 DCO - 08/09/2004 - titre - 2. Context
 INERIS 3 main divisions : Since 1990 Budget : 70 M 600 employees Mechanisms of gas migration in flooding post- INERIS mining context Experimental approach and modelling Maîtriser le risque pour un développement
INERIS 3 main divisions : Since 1990 Budget : 70 M 600 employees Mechanisms of gas migration in flooding post- INERIS mining context Experimental approach and modelling Maîtriser le risque pour un développement
Solar Photocatalytic Oxidation (PCO) Process with a Flat-plate Reactor
 Solar Photocatalytic Oxidation (PCO) Process with a Flat-plate Reactor Y., L. Zou and E. Hu School of Engineering and Technology Deain University Geelong, Victoria 317 Australia E-mail: [email protected]
Solar Photocatalytic Oxidation (PCO) Process with a Flat-plate Reactor Y., L. Zou and E. Hu School of Engineering and Technology Deain University Geelong, Victoria 317 Australia E-mail: [email protected]
presented by Neal Leddy CMA Analytical Workshop 2012 SURFACE AREA AND POROSITY
 presented by Neal Leddy CMA Analytical Workshop 2012 SURFACE AREA AND POROSITY Adsorption When a gas or vapour phase is brought into contact with a solid, part of it is taken up and remains on the outside
presented by Neal Leddy CMA Analytical Workshop 2012 SURFACE AREA AND POROSITY Adsorption When a gas or vapour phase is brought into contact with a solid, part of it is taken up and remains on the outside
KINETIC DETERMINATION OF SELENIUM BY VISIBLE SPECTROSCOPY (VERSION 1.8)
 Selenium Determination, Page 1 KINETIC DETERMINATION OF SELENIUM BY VISIBLE SPECTROSCOPY I. BACKGROUND. (VERSION 1.8) The majority of reactions used in analytical chemistry possess the following characteristics:
Selenium Determination, Page 1 KINETIC DETERMINATION OF SELENIUM BY VISIBLE SPECTROSCOPY I. BACKGROUND. (VERSION 1.8) The majority of reactions used in analytical chemistry possess the following characteristics:
Modelling the Drying of Porous Coal Particles in Superheated Steam
 B. A. OLUFEMI and I. F. UDEFIAGBON, Modelling the Drying of Porous Coal, Chem. Biochem. Eng. Q. 24 (1) 29 34 (2010) 29 Modelling the Drying of Porous Coal Particles in Superheated Steam B. A. Olufemi *
B. A. OLUFEMI and I. F. UDEFIAGBON, Modelling the Drying of Porous Coal, Chem. Biochem. Eng. Q. 24 (1) 29 34 (2010) 29 Modelling the Drying of Porous Coal Particles in Superheated Steam B. A. Olufemi *
A LAMINAR FLOW ELEMENT WITH A LINEAR PRESSURE DROP VERSUS VOLUMETRIC FLOW. 1998 ASME Fluids Engineering Division Summer Meeting
 TELEDYNE HASTINGS TECHNICAL PAPERS INSTRUMENTS A LAMINAR FLOW ELEMENT WITH A LINEAR PRESSURE DROP VERSUS VOLUMETRIC FLOW Proceedings of FEDSM 98: June -5, 998, Washington, DC FEDSM98 49 ABSTRACT The pressure
TELEDYNE HASTINGS TECHNICAL PAPERS INSTRUMENTS A LAMINAR FLOW ELEMENT WITH A LINEAR PRESSURE DROP VERSUS VOLUMETRIC FLOW Proceedings of FEDSM 98: June -5, 998, Washington, DC FEDSM98 49 ABSTRACT The pressure
BET Surface Area Analysis of Nanoparticles
 OpenStax-CNX module: m38278 1 BET Surface Area Analysis of Nanoparticles Nina Hwang Andrew R. Barron This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0
OpenStax-CNX module: m38278 1 BET Surface Area Analysis of Nanoparticles Nina Hwang Andrew R. Barron This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0
Modeling and Simulation of Oil-Water Flows with Viscous Fingering in Heterogeneous Porous Media.
 ACMA 2014 Modeling and Simulation of Oil-Water Flows with Viscous Fingering in Heterogeneous Porous Media. H. DJEBOURI 1, S. ZOUAOUI 1, K. MOHAMMEDI 2, and A. AIT AIDER 1 1 Laboratoire de Mécanique Structure
ACMA 2014 Modeling and Simulation of Oil-Water Flows with Viscous Fingering in Heterogeneous Porous Media. H. DJEBOURI 1, S. ZOUAOUI 1, K. MOHAMMEDI 2, and A. AIT AIDER 1 1 Laboratoire de Mécanique Structure
Abaqus/CFD Sample Problems. Abaqus 6.10
 Abaqus/CFD Sample Problems Abaqus 6.10 Contents 1. Oscillatory Laminar Plane Poiseuille Flow 2. Flow in Shear Driven Cavities 3. Buoyancy Driven Flow in Cavities 4. Turbulent Flow in a Rectangular Channel
Abaqus/CFD Sample Problems Abaqus 6.10 Contents 1. Oscillatory Laminar Plane Poiseuille Flow 2. Flow in Shear Driven Cavities 3. Buoyancy Driven Flow in Cavities 4. Turbulent Flow in a Rectangular Channel
HEXAVALENT CHROMIUM REMOVAL FROM INDUSTRIAL WATSEWATER BY CHEMICAL PRECIPITATION METHOD
 HEXAVALENT CHROMIUM REMOVAL FROM INDUSTRIAL WATSEWATER BY CHEMICAL PRECIPITATION METHOD Dr. C.R.Ramakrishnaiah P.G-Environmental Engineering Dept of Civil Engineering, B.M.S. College of Engineering Bull
HEXAVALENT CHROMIUM REMOVAL FROM INDUSTRIAL WATSEWATER BY CHEMICAL PRECIPITATION METHOD Dr. C.R.Ramakrishnaiah P.G-Environmental Engineering Dept of Civil Engineering, B.M.S. College of Engineering Bull
Pore size and BET surface area. analysis at your fingertips. SA 3100. Gas Adsorption Analyzer
 Pore size and BET surface area analysis at your fingertips. SA 3100 Gas Adsorption Analyzer The History of Beckman Coulter Particle Characterization Introducing The SA 3100 Surface Area and Pore Size Analyzer
Pore size and BET surface area analysis at your fingertips. SA 3100 Gas Adsorption Analyzer The History of Beckman Coulter Particle Characterization Introducing The SA 3100 Surface Area and Pore Size Analyzer
SIZE OF A MOLECULE FROM A VISCOSITY MEASUREMENT
 Experiment 8, page 1 Version of April 25, 216 Experiment 446.8 SIZE OF A MOLECULE FROM A VISCOSITY MEASUREMENT Theory Viscous Flow. Fluids attempt to minimize flow gradients by exerting a frictional force,
Experiment 8, page 1 Version of April 25, 216 Experiment 446.8 SIZE OF A MOLECULE FROM A VISCOSITY MEASUREMENT Theory Viscous Flow. Fluids attempt to minimize flow gradients by exerting a frictional force,
Numerical Analysis of Independent Wire Strand Core (IWSC) Wire Rope
 Numerical Analysis of Independent Wire Strand Core (IWSC) Wire Rope Rakesh Sidharthan 1 Gnanavel B K 2 Assistant professor Mechanical, Department Professor, Mechanical Department, Gojan engineering college,
Numerical Analysis of Independent Wire Strand Core (IWSC) Wire Rope Rakesh Sidharthan 1 Gnanavel B K 2 Assistant professor Mechanical, Department Professor, Mechanical Department, Gojan engineering college,
Characterization of surfaces by AFM topographical, mechanical and chemical properties
 Characterization of surfaces by AFM topographical, mechanical and chemical properties Jouko Peltonen Department of physical chemistry Åbo Akademi University Atomic Force Microscopy (AFM) Contact mode AFM
Characterization of surfaces by AFM topographical, mechanical and chemical properties Jouko Peltonen Department of physical chemistry Åbo Akademi University Atomic Force Microscopy (AFM) Contact mode AFM
Comparison of Heat Transfer between a Helical and Straight Tube Heat Exchanger
 International Journal of Engineering Research and Technology. ISSN 0974-3154 Volume 6, Number 1 (2013), pp. 33-40 International Research Publication House http://www.irphouse.com Comparison of Heat Transfer
International Journal of Engineering Research and Technology. ISSN 0974-3154 Volume 6, Number 1 (2013), pp. 33-40 International Research Publication House http://www.irphouse.com Comparison of Heat Transfer
Surface Area and Porosity
 Surface Area and Porosity 1 Background Techniques Surface area Outline Total - physical adsorption External Porosity meso micro 2 Length 1 Å 1 nm 1 µm 1 1 1 1 1 mm macro meso micro metal crystallite 1-1
Surface Area and Porosity 1 Background Techniques Surface area Outline Total - physical adsorption External Porosity meso micro 2 Length 1 Å 1 nm 1 µm 1 1 1 1 1 mm macro meso micro metal crystallite 1-1
Low Temperature Adsorption Versus Pore Size in Activated Carbons
 Low Temperature Adsorption Versus Pore Size in Activated Carbons D. Martins 1, I. Catarino 1, D. Lopes 1, I. Esteves 2, J.P. Mota 2, G. Bonfait 1 1 CEFITEC Departamento de Física, Faculdade de Ciências
Low Temperature Adsorption Versus Pore Size in Activated Carbons D. Martins 1, I. Catarino 1, D. Lopes 1, I. Esteves 2, J.P. Mota 2, G. Bonfait 1 1 CEFITEC Departamento de Física, Faculdade de Ciências
Journal bearings/sliding bearings
 Journal bearings/sliding bearings Operating conditions: Advantages: - Vibration damping, impact damping, noise damping - not sensitive for vibrations, low operating noise level - dust tight (if lubricated
Journal bearings/sliding bearings Operating conditions: Advantages: - Vibration damping, impact damping, noise damping - not sensitive for vibrations, low operating noise level - dust tight (if lubricated
PhD Theses STUDY OF THE SOLVENT GRADIENT SIMULATED MOVING BED PREPARATIVE LIQUID CHROMATOGRAPHIC PROCESS. Written by Melinda Nagy
 PhD Theses STUDY OF THE SOLVENT GRADIENT SIMULATED MOVING BED PREPARATIVE LIQUID CHROMATOGRAPHIC PROCESS Written by Melinda Nagy Consultants Tibor Szánya Géza Horváth University of Pannonia Department
PhD Theses STUDY OF THE SOLVENT GRADIENT SIMULATED MOVING BED PREPARATIVE LIQUID CHROMATOGRAPHIC PROCESS Written by Melinda Nagy Consultants Tibor Szánya Géza Horváth University of Pannonia Department
Hydrogen Adsorption on Nanoporous Biocarbon
 Hydrogen Adsorption on Nanoporous Biocarbon Mikael Wood, Jacob Burress, Cintia Lapilli, Peter Pfeifer, Parag Shah, Galen Suppes University of Missouri-Columbia Phillip Parilla, Anne Dillon National Renewable
Hydrogen Adsorption on Nanoporous Biocarbon Mikael Wood, Jacob Burress, Cintia Lapilli, Peter Pfeifer, Parag Shah, Galen Suppes University of Missouri-Columbia Phillip Parilla, Anne Dillon National Renewable
TEMPERATURE, CONCENTRATION, AND PUMPING EFFECTS ON PAM VISCOSITY
 TEMPERATURE, CONCENTRATION, AND PUMPING EFFECTS ON PAM VISCOSITY D. L. Bjorneberg ABSTRACT. As polyacrylamide (PAM) use in irrigated agriculture increases, new methods are being sought to accurately and
TEMPERATURE, CONCENTRATION, AND PUMPING EFFECTS ON PAM VISCOSITY D. L. Bjorneberg ABSTRACT. As polyacrylamide (PAM) use in irrigated agriculture increases, new methods are being sought to accurately and
Lecture 6 - Boundary Conditions. Applied Computational Fluid Dynamics
 Lecture 6 - Boundary Conditions Applied Computational Fluid Dynamics Instructor: André Bakker http://www.bakker.org André Bakker (2002-2006) Fluent Inc. (2002) 1 Outline Overview. Inlet and outlet boundaries.
Lecture 6 - Boundary Conditions Applied Computational Fluid Dynamics Instructor: André Bakker http://www.bakker.org André Bakker (2002-2006) Fluent Inc. (2002) 1 Outline Overview. Inlet and outlet boundaries.
Part IV. Conclusions
 Part IV Conclusions 189 Chapter 9 Conclusions and Future Work CFD studies of premixed laminar and turbulent combustion dynamics have been conducted. These studies were aimed at explaining physical phenomena
Part IV Conclusions 189 Chapter 9 Conclusions and Future Work CFD studies of premixed laminar and turbulent combustion dynamics have been conducted. These studies were aimed at explaining physical phenomena
Modeling and Simulation of Complex Multiphase Flows in the Pharmaceutical Industry
 SIMNET Days 2010 Februar 10, 2010 Modeling and Simulation of Complex Multiphase Flows in the Pharmaceutical Industry D. Suzzi a, G. Toschkoff a, S. Radl a,b, Th. Hörmann a, M. Schaffer a, D. Machold a,
SIMNET Days 2010 Februar 10, 2010 Modeling and Simulation of Complex Multiphase Flows in the Pharmaceutical Industry D. Suzzi a, G. Toschkoff a, S. Radl a,b, Th. Hörmann a, M. Schaffer a, D. Machold a,
Turbulence Modeling in CFD Simulation of Intake Manifold for a 4 Cylinder Engine
 HEFAT2012 9 th International Conference on Heat Transfer, Fluid Mechanics and Thermodynamics 16 18 July 2012 Malta Turbulence Modeling in CFD Simulation of Intake Manifold for a 4 Cylinder Engine Dr MK
HEFAT2012 9 th International Conference on Heat Transfer, Fluid Mechanics and Thermodynamics 16 18 July 2012 Malta Turbulence Modeling in CFD Simulation of Intake Manifold for a 4 Cylinder Engine Dr MK
A Comparison of Analytical and Finite Element Solutions for Laminar Flow Conditions Near Gaussian Constrictions
 A Comparison of Analytical and Finite Element Solutions for Laminar Flow Conditions Near Gaussian Constrictions by Laura Noelle Race An Engineering Project Submitted to the Graduate Faculty of Rensselaer
A Comparison of Analytical and Finite Element Solutions for Laminar Flow Conditions Near Gaussian Constrictions by Laura Noelle Race An Engineering Project Submitted to the Graduate Faculty of Rensselaer
Understanding Analytical Chemistry (Weighing, Mixing, Measuring and Evaluating)
 Name: Date: Understanding Analytical Chemistry (Weighing, Mixing, Measuring and Evaluating) High School Environmental Science AP Module 1 Environmental Lab NGSSS Big Ideas: This module is a laboratory-based
Name: Date: Understanding Analytical Chemistry (Weighing, Mixing, Measuring and Evaluating) High School Environmental Science AP Module 1 Environmental Lab NGSSS Big Ideas: This module is a laboratory-based
Investigation of the Effect of Dynamic Capillary Pressure on Waterflooding in Extra Low Permeability Reservoirs
 Copyright 013 Tech Science Press SL, vol.9, no., pp.105-117, 013 Investigation of the Effect of Dynamic Capillary Pressure on Waterflooding in Extra Low Permeability Reservoirs Tian Shubao 1, Lei Gang
Copyright 013 Tech Science Press SL, vol.9, no., pp.105-117, 013 Investigation of the Effect of Dynamic Capillary Pressure on Waterflooding in Extra Low Permeability Reservoirs Tian Shubao 1, Lei Gang
Dynamic Process Modeling. Process Dynamics and Control
 Dynamic Process Modeling Process Dynamics and Control 1 Description of process dynamics Classes of models What do we need for control? Modeling for control Mechanical Systems Modeling Electrical circuits
Dynamic Process Modeling Process Dynamics and Control 1 Description of process dynamics Classes of models What do we need for control? Modeling for control Mechanical Systems Modeling Electrical circuits
Performance prediction of a centrifugal pump working in direct and reverse mode using Computational Fluid Dynamics
 European Association for the Development of Renewable Energies, Environment and Power Quality (EA4EPQ) International Conference on Renewable Energies and Power Quality (ICREPQ 10) Granada (Spain), 23rd
European Association for the Development of Renewable Energies, Environment and Power Quality (EA4EPQ) International Conference on Renewable Energies and Power Quality (ICREPQ 10) Granada (Spain), 23rd
Spectrophotometry and the Beer-Lambert Law: An Important Analytical Technique in Chemistry
 Spectrophotometry and the Beer-Lambert Law: An Important Analytical Technique in Chemistry Jon H. Hardesty, PhD and Bassam Attili, PhD Collin College Department of Chemistry Introduction: In the last lab
Spectrophotometry and the Beer-Lambert Law: An Important Analytical Technique in Chemistry Jon H. Hardesty, PhD and Bassam Attili, PhD Collin College Department of Chemistry Introduction: In the last lab
VALIDATION, MODELING, AND SCALE-UP OF CHEMICAL LOOPING COMBUSTION WITH OXYGEN UNCOUPLING
 VALIDATION, MODELING, AND SCALE-UP OF CHEMICAL LOOPING COMBUSTION WITH OXYGEN UNCOUPLING A research program funded by the University of Wyoming School of Energy Resources Executive Summary Principal Investigator:
VALIDATION, MODELING, AND SCALE-UP OF CHEMICAL LOOPING COMBUSTION WITH OXYGEN UNCOUPLING A research program funded by the University of Wyoming School of Energy Resources Executive Summary Principal Investigator:
Shake gels based on Laponite PEO mixtures: effect of polymer molecular weight
 Designed Monomers and Polymers, Vol. 8, No. 5, pp. 453 462 (2005) VSP 2005. Also available online - www.vsppub.com Shake gels based on Laponite PEO mixtures: effect of polymer molecular weight VOLKAN CAN
Designed Monomers and Polymers, Vol. 8, No. 5, pp. 453 462 (2005) VSP 2005. Also available online - www.vsppub.com Shake gels based on Laponite PEO mixtures: effect of polymer molecular weight VOLKAN CAN
Some generalization of Langmuir adsorption isotherm
 Internet Journal of Chemistry, 2000, 3, 14 [ISSN: 1099-8292]. Article 14 Some generalization of Langmuir adsorption isotherm Leszek Czepirski, Mieczyslaw R. Balys, Ewa Komorowska-Czepirska University of
Internet Journal of Chemistry, 2000, 3, 14 [ISSN: 1099-8292]. Article 14 Some generalization of Langmuir adsorption isotherm Leszek Czepirski, Mieczyslaw R. Balys, Ewa Komorowska-Czepirska University of
In order to solve this problem it is first necessary to use Equation 5.5: x 2 Dt. = 1 erf. = 1.30, and x = 2 mm = 2 10-3 m. Thus,
 5.3 (a) Compare interstitial and vacancy atomic mechanisms for diffusion. (b) Cite two reasons why interstitial diffusion is normally more rapid than vacancy diffusion. Solution (a) With vacancy diffusion,
5.3 (a) Compare interstitial and vacancy atomic mechanisms for diffusion. (b) Cite two reasons why interstitial diffusion is normally more rapid than vacancy diffusion. Solution (a) With vacancy diffusion,
Measurement of Soil Parameters by Using Penetrometer Needle Apparatus
 Vol.3, Issue.1, Jan-Feb. 2013 pp-284-290 ISSN: 2249-6645 Measurement of Soil Parameters by Using Penetrometer Needle Apparatus Mahmoud M. Abu zeid, 1 Amr M. Radwan, 2 Emad A. Osman, 3 Ahmed M.Abu-bakr,
Vol.3, Issue.1, Jan-Feb. 2013 pp-284-290 ISSN: 2249-6645 Measurement of Soil Parameters by Using Penetrometer Needle Apparatus Mahmoud M. Abu zeid, 1 Amr M. Radwan, 2 Emad A. Osman, 3 Ahmed M.Abu-bakr,
Steady Heat Conduction
 Steady Heat Conduction In thermodynamics, we considered the amount of heat transfer as a system undergoes a process from one equilibrium state to another. hermodynamics gives no indication of how long
Steady Heat Conduction In thermodynamics, we considered the amount of heat transfer as a system undergoes a process from one equilibrium state to another. hermodynamics gives no indication of how long
Activity Sheets Enzymes and Their Functions
 Name: Date: Activity Sheets Enzymes and Their Functions amylase What are Enzymes? starch glucose Enzymes are compounds that assist chemical reactions by increasing the rate at which they occur. For example,
Name: Date: Activity Sheets Enzymes and Their Functions amylase What are Enzymes? starch glucose Enzymes are compounds that assist chemical reactions by increasing the rate at which they occur. For example,
Reaction Stoichiometry and the Formation of a Metal Ion Complex
 Reaction Stoichiometry and the Formation of a Metal Ion Complex Objectives The objectives of this laboratory are as follows: To use the method of continuous variation to determine the reaction stoichiometry
Reaction Stoichiometry and the Formation of a Metal Ion Complex Objectives The objectives of this laboratory are as follows: To use the method of continuous variation to determine the reaction stoichiometry
Spectrophotometer - Milton Roy Spectronic 21D or equivalent.
 3M COMPANY OCCUPATIONAL HEALTH AND ENVIRONMENTAL SAFETY DIVISION DETERMINATION OF FORMALDEHYDE VAPORS IN AIR USING 3M 3721 FORMALDEHYDE MONITORS May, 2002 SCOPE This procedure covers the method of collecting
3M COMPANY OCCUPATIONAL HEALTH AND ENVIRONMENTAL SAFETY DIVISION DETERMINATION OF FORMALDEHYDE VAPORS IN AIR USING 3M 3721 FORMALDEHYDE MONITORS May, 2002 SCOPE This procedure covers the method of collecting
Reaction of Blue Food Dye with Bleach
 Exercise 2 Reaction of Blue Food Dye with Bleach 2 Introduction In the experiment, you will study the rate of the reaction of FD&C Blue #1 (Blue #1 is denoted by E number E133 in food stuff) with sodium
Exercise 2 Reaction of Blue Food Dye with Bleach 2 Introduction In the experiment, you will study the rate of the reaction of FD&C Blue #1 (Blue #1 is denoted by E number E133 in food stuff) with sodium
Phosphate Recovery from Municipal Wastewater through Crystallization of Calcium Phosphate
 Phosphate Recovery from Municipal Wastewater through Crystallization of Calcium Phosphate A. Alamdari, Ph.D. * and S. Rohani, M.Sc. * Department of Chemical Engineering, School of Engineering, Shiraz University,
Phosphate Recovery from Municipal Wastewater through Crystallization of Calcium Phosphate A. Alamdari, Ph.D. * and S. Rohani, M.Sc. * Department of Chemical Engineering, School of Engineering, Shiraz University,
Improving the yield of soybean oil extraction process by using of microwave system
 Improving the yield of soybean oil extraction process by using of microwave system M. Ghazvehi, M. Nasiri* School of Chemical, Gas and Petroleum Engineering, Semnan University,Semnan, Iran The increasing
Improving the yield of soybean oil extraction process by using of microwave system M. Ghazvehi, M. Nasiri* School of Chemical, Gas and Petroleum Engineering, Semnan University,Semnan, Iran The increasing
DENSITY MEASURING SYSTEMS
 SHIBAYAMA SCIENTIFIC CO., LTD. DENSITY MEASURING SYSTEMS Density Gradient Tube Method Direct Reading Type A / Type B Comply with the standards below: JIS K-0061-1992, K-7112 1980 ASTM D1505 Type A /Left
SHIBAYAMA SCIENTIFIC CO., LTD. DENSITY MEASURING SYSTEMS Density Gradient Tube Method Direct Reading Type A / Type B Comply with the standards below: JIS K-0061-1992, K-7112 1980 ASTM D1505 Type A /Left
vii TABLE OF CONTENTS CHAPTER TITLE PAGE DECLARATION DEDICATION ACKNOWLEDGEMENT ABSTRACT ABSTRAK
 vii TABLE OF CONTENTS CHAPTER TITLE PAGE DECLARATION DEDICATION ACKNOWLEDGEMENT ABSTRACT ABSTRAK TABLE OF CONTENTS LIST OF TABLES LIST OF FIGURES LIST OF SYMBOLS / ABBREVIATIONS LIST OF APPENDICES ii iii
vii TABLE OF CONTENTS CHAPTER TITLE PAGE DECLARATION DEDICATION ACKNOWLEDGEMENT ABSTRACT ABSTRAK TABLE OF CONTENTS LIST OF TABLES LIST OF FIGURES LIST OF SYMBOLS / ABBREVIATIONS LIST OF APPENDICES ii iii
DIESEL ENGINE IN-CYLINDER CALCULATIONS WITH OPENFOAM
 DIESEL ENGINE IN-CYLINDER CALCULATIONS WITH OPENFOAM 1 Ervin Adorean *, 1 Gheorghe-Alexandru Radu 1 Transilvania University of Brasov, Romania KEYWORDS - diesel, engine, CFD, simulation, OpenFOAM ABSTRACT
DIESEL ENGINE IN-CYLINDER CALCULATIONS WITH OPENFOAM 1 Ervin Adorean *, 1 Gheorghe-Alexandru Radu 1 Transilvania University of Brasov, Romania KEYWORDS - diesel, engine, CFD, simulation, OpenFOAM ABSTRACT
Department of Environmental Engineering
 Department of Environmental Engineering Master of Engineering Program in Environmental Engineering (International Program) M.Eng. (Environmental Engineering) Plan A Option 1: (1) Major courses: minimum
Department of Environmental Engineering Master of Engineering Program in Environmental Engineering (International Program) M.Eng. (Environmental Engineering) Plan A Option 1: (1) Major courses: minimum
Upon completion of this lab, the student will be able to:
 1 Learning Outcomes EXPERIMENT B4: CHEMICAL EQUILIBRIUM Upon completion of this lab, the student will be able to: 1) Analyze the absorbance spectrum of a sample. 2) Calculate the equilibrium constant for
1 Learning Outcomes EXPERIMENT B4: CHEMICAL EQUILIBRIUM Upon completion of this lab, the student will be able to: 1) Analyze the absorbance spectrum of a sample. 2) Calculate the equilibrium constant for
HiPer Ion Exchange Chromatography Teaching Kit
 HiPer Ion Exchange Chromatography Teaching Kit Product Code: HTC001 Number of experiments that can be performed: 5 Duration of Experiment: Protocol: 5-6 hours Storage Instructions: The kit is stable for
HiPer Ion Exchange Chromatography Teaching Kit Product Code: HTC001 Number of experiments that can be performed: 5 Duration of Experiment: Protocol: 5-6 hours Storage Instructions: The kit is stable for
CFD Analysis Of Multi-Phase Flow And Its Measurements
 IOSR Journal of Mechanical and Civil Engineering (IOSR-JMCE) e-issn: 2278-1684,p-ISSN: 2320-334X, Volume 9, Issue 4 (Nov. - Dec. 2013), PP 30-37 CFD Analysis Of Multi-Phase Flow And Its Measurements C
IOSR Journal of Mechanical and Civil Engineering (IOSR-JMCE) e-issn: 2278-1684,p-ISSN: 2320-334X, Volume 9, Issue 4 (Nov. - Dec. 2013), PP 30-37 CFD Analysis Of Multi-Phase Flow And Its Measurements C
Supporting Information
 Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2013 A Study of Commercial Nanoparticulate g-al 2 O 3 Catalyst Supports Yahaya Rozita, [a] Rik Brydson,* [a] Tim P. Comyn,
Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2013 A Study of Commercial Nanoparticulate g-al 2 O 3 Catalyst Supports Yahaya Rozita, [a] Rik Brydson,* [a] Tim P. Comyn,
Reversed Phase High Presssure Liquid Chromatograhphic Technique for Determination of Sodium Alginate from Oral Suspension
 International Journal of PharmTech Research CODEN (USA): IJPRIF ISSN : 0974-4304 Vol.2, No.2, pp 1634-1638, April-June 2010 Reversed Phase High Presssure Liquid Chromatograhphic Technique for Determination
International Journal of PharmTech Research CODEN (USA): IJPRIF ISSN : 0974-4304 Vol.2, No.2, pp 1634-1638, April-June 2010 Reversed Phase High Presssure Liquid Chromatograhphic Technique for Determination
ENE 806, Project Report 3 CHEMICAL PRECIPITATION: WATER SOFTENING. Grégoire Seyrig Wenqian Shan
 ENE 806, Project Report 3 CHEMICAL PRECIPITATION: WATER SOFTENING Grégoire Seyrig Wenqian Shan College of Engineering, Michigan State University Spring 2007 ABSTRACT The groundwater with high level initial
ENE 806, Project Report 3 CHEMICAL PRECIPITATION: WATER SOFTENING Grégoire Seyrig Wenqian Shan College of Engineering, Michigan State University Spring 2007 ABSTRACT The groundwater with high level initial
Basic Principles in Microfluidics
 Basic Principles in Microfluidics 1 Newton s Second Law for Fluidics Newton s 2 nd Law (F= ma) : Time rate of change of momentum of a system equal to net force acting on system!f = dp dt Sum of forces
Basic Principles in Microfluidics 1 Newton s Second Law for Fluidics Newton s 2 nd Law (F= ma) : Time rate of change of momentum of a system equal to net force acting on system!f = dp dt Sum of forces
Characteristics of Floc: Interplay between Retention Aid and Hydrodynamic. Forces Effects in Fines Flocculation
 Characteristics of Floc: Interplay between Retention Aid and Hydrodynamic Forces Effects in Fines Flocculation Mohammad. R. Abdallah/Qasaimeh AlBalqa Applied University, AlHuson University College, Chemical
Characteristics of Floc: Interplay between Retention Aid and Hydrodynamic Forces Effects in Fines Flocculation Mohammad. R. Abdallah/Qasaimeh AlBalqa Applied University, AlHuson University College, Chemical
