Medical Policy An independent licensee of the Blue Cross Blue Shield Association
|
|
|
- Marlene Carpenter
- 7 years ago
- Views:
Transcription
1 Interspinous Fixation (Fusion) Devices Page 1 of 6 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: See Also: Interspinous Fixation (Fusion) Devices Lumbar Spine Fusion Professional Institutional Original Effective Date: January 23, 2015 Original Effective Date: January 23, 2015 Revision Date(s): January 23, 2015; November 24, 2015 Revision Date(s): January 23, 2015; November 24, 2015 Current Effective Date: January 23, 2015 Current Effective Date: January 23, 2015 State and Federal mandates and health plan member contract language, including specific provisions/exclusions, take precedence over Medical Policy and must be considered first in determining eligibility for coverage. To verify a member's benefits, contact Blue Cross and Blue Shield of Kansas Customer Service. The BCBSKS Medical Policies contained herein are for informational purposes and apply only to members who have health insurance through BCBSKS or who are covered by a self-insured group plan administered by BCBSKS. Medical Policy for FEP members is subject to FEP medical policy which may differ from BCBSKS Medical Policy. The medical policies do not constitute medical advice or medical care. Treating health care providers are independent contractors and are neither employees nor agents of Blue Cross and Blue Shield of Kansas and are solely responsible for diagnosis, treatment and medical advice. If your patient is covered under a different Blue Cross and Blue Shield plan, please refer to the Medical Policies of that plan. Populations Interventions Comparators Outcomes Interventions of interest Comparators of interest Interspinous fixation Pedicle screw devices with interbody constructs fusion Individuals: Who are undergoing spinal fusion Individuals: With spinal stenosis and/or spondylolisthesis Interventions of interest Interspinous fixation devices alone Comparators of interest Decompression Posterolateral fusion Interbody fusion Relevant outcomes include: Symptoms Functional outcomes Quality of life Resource utilization Treatment-related morbidity Relevant outcomes include: Symptoms Functional outcomes Quality of life Resource utilization Treatment-related morbidity
2 Interspinous Fixation (Fusion) Devices Page 2 of 6 DESCRIPTION Interspinous fixation (fusion) devices are being developed to aid in the stabilization of the spine. They are being evaluated as alternatives to pedicle screw and rod constructs in combination with interbody fusion. Interspinous fixation devices are also being evaluated for stand-alone use in patients with spinal stenosis and/or spondylolisthesis. Background Contemporary models of interspinous fixation devices have evolved from spinous process wiring with bone blocks and early device designs (e.g., Wilson plate, Meurig-Williams system, Daab plate). The newer devices range from paired plates with teeth to U-shaped devices with wings that are attached to the spinous process. They are intended to be an alternative to pedicle screw and rod constructs to aid in the stabilization of the spine with interbody fusion. Interspinous fixation devices are placed under direct visualization, while screw and rod systems may be placed either under direct visualization or percutaneously. Use of an interspinous fixation device in combination with a unilateral pedicle screw system has also been proposed. Interspinous fixation devices are not intended for standalone use. Interspinous fixation (fusion) devices contrast with interspinous distraction devices (spacers), which are used alone for decompression and are typically not fixed to the spinous process. In addition, whereas interspinous distraction devices may use dynamic stabilization, interspinous fixation devices are rigid. However, the fixation devices might also be used to distract the spinous processes and decrease lordosis. Thus, the fixation devices might be used off-label without interbody fusion as decompression (distraction) devices in patients with spinal stenosis. If fixation devices are used alone as a spacer, there is a risk of spinous process fracture. For use in combination with fusion, it is proposed that interspinous fixation systems are less invasive and present fewer risks than pedicle or facet screws. However, while biomechanical studies indicate that interspinous fixation devices may be similar to pedicle screw-rod constructs in limiting the range of flexion-extension, they may be less effective than bilateral pedicle screw-rod fixation for limiting axial rotation and lateral bending. 1 There is a potential for a negative impact on the interbody cage and bone graft due to focal kyphosis resulting from the interspinous device. There is also a potential for spinous process fracture. Given these uncertainties, studies are needed that compare health outcomes between interspinous fixation devices and pedicle screw-rod fixation. Regulatory Status The following interspinous fixation devices have received clearance to market by the U.S. Food and Drug Administration (FDA). This may not be an exhaustive list. Affix (NuVasive) Aileron (Life Spine) Aspen (Lanx, acquired by BioMet) Axle (X-Spine)
3 Interspinous Fixation (Fusion) Devices Page 3 of 6 BacFuse (Pioneer Surgical) BridgePoint (Alphatec Spine) coflex-f (Paradigm Spine) Inspan (Spine Frontier) Interbridge Interspinous Posterior Fixation System (LDR Spine) Minuteman (Spinal Simplicity) PrimaLOK (OsteoMed) Octave (Life Spine) Spire (Medtronic) SP-Fix (Globus) Zip Mis Interspinous Fusion System Interspinous fixation devices are intended to be used as an adjunct to interbody fusion. For example, the indication for use of the coflex-f implant: is a posterior, nonpedicle supplemental fixation device intended for use with an interbody cage as an adjunct to fusion at a single level in the lumbar spine (L1-S1). It is intended for attachment to the spinous processes for the purpose of achieving stabilization to promote fusion in patients with degenerative disc disease defined as back pain of discogenic origin with degeneration of the disc confirmed by history and radiographic studies with up to Grade 1 spondylolisthesis. Use of an interspinous fixation device for a stand-alone procedure would be considered off-label. FDA Product code: PEK. POLICY A. Interspinous fixation (fusion) devices are considered experimental / investigational for any indication, including, but not limited to, use: 1. in combination with interbody fusion, OR 2. alone for decompression in patients with spinal stenosis. Policy Guidelines Clinical input has identified potential exceptions where the devices might be considered medically necessary, such as patients with small pedicles where pedicle screws could not be safely placed. RATIONALE This policy was created with a search of the literature through July 2012 and updated periodically using the MEDLINE database. The most recent update was performed though August 12, Use of the Aspen interspinous fixation device as a stand-alone interspinous spacer was reported in a prospective series of 6 cases (an additional 32 patients received the X-stop interspinous
4 Interspinous Fixation (Fusion) Devices Page 4 of 6 distraction device). 2 The study population consisted of consecutive patients with a primary diagnosis of lumbar spinal stenosis with pain that was relieved by sitting or lumbar flexion. Of the 6 patients implanted with the Aspen device, 2 (33%) had spinous process fractures observable on computed tomography. Of the entire group of 38 patients, 55% of those with spondylolisthesis (n=20) had a fracture within 6 months of surgery. None of the 18 patients without spondylolisthesis experienced a fracture. In 2013, use of an interspinous fusion plate was reported in a series of 4 patients in conjunction with surgery for recurrent lumbar disc herniation. 3 Vokshoor et al reported a retrospective series of 86 patients who had a spinous process device implanted. 4 After adjusting for age and sex, there was a 3.6-point decrease in the visual analog scale for pain that was maintained over the 12-month follow-up. In the 50 patients who had computed tomography scans, interspinous process fusion was observed in 94%. Presence of an interbody cage did not affect the fusion rate. Two patients (2.3%) had the devices removed due to pain secondary to spinous process and/or lamina fracture. In 2014, Sclafani et al reported an industry-sponsored retrospective series on the polyaxial PrimaLOK interspinous fusion device. 5 Thirty four patients were implanted with the interspinous fusion device alone, 16 patients received the PrimaLOK together with an interbody cage, and 3 patients received the PrimaLOK together with pedicle screw instrumentation and an interbody cage. Evaluation at 6 weeks found no cases of fracture or migration of the device, although there were 4 cases of hardware removal and 2 cases of reoperation for adjacent level disease during the follow-up. At a mean 22 months after the index surgery, the average pain score had improved from 7.2 to 4.5 on a 10-point scale. There was a statistically significant improvement in pain score for patients with degenerative disc disease with lumbar stenosis (2.8, n=25, p<0.001) or spondylolisthesis (4.6, n=6, p=0.01), but not for patients with lumbar disc herniation (2.2, n=10, p>0.05). Ongoing and Unpublished Clinical Trials Some currently unpublished trials that might influence this evidence review are listed in Table 1. Table 1. Summary of Key Trials NCT No. Trial Name Planned Ongoing NCT a NCT a NCT a Unpublished NCT a NCT a A Multi-Center Prospective Randomized Study Comparing Supplemental Posterior Instrumentation, Aspen Spinous Process System Versus Pedicle Screw Fixation, in Lateral Lumbar Interbody Fusion (LLIF) or Anterior Lumbar Interbody Fusion (ALIF) A Multi-Center Prospective Randomized Study to Evaluate the Efficacy of the Aspen Spinous Process System for Use in Anterior Lumbar Interbody Fusion (ALIF) Efficacy and Quality of Life Following Treatment of Lumbar Spinal Stenosis, Spondylolisthesis or Degenerative Disc Disease With the Minuteman Interspinous Interlaminar Fusion Implant Versus Surgical Decompression A Prospective, Non-randomized, Multi-Center Evaluation of Interlaminar Lumbar Instrumented Fusion (ILIF ) A Multi-Center Prospective Study Evaluation Aspen Spinous Process Fixation System for Use in Posterolateral Fusion (PLF) in Patients With Spondylolisthesis NCT: national clinical trial. a Denotes industry-sponsored or cosponsored trial. Enrollment Completion Date 144 Dec Dec Dec Completed Jun Suspended
5 Interspinous Fixation (Fusion) Devices Page 5 of 6 Summary of Evidence The evidence on interspinous fixation (fusion) devices in patients who are undergoing interbody fusion or who have spinal stenosis and/or spondylolisthesis includes a small prospective series and retrospective series. Relevant outcomes are symptoms, functional outcomes, quality of life, resource utilization, and treatment-related morbidity. There is a lack of evidence on the efficacy of interspinous fixation devices, both for use in combination with interbody fusion and for use as a stand-alone procedure. Randomized controlled trials are needed that evaluate health outcomes following use of interspinous fixation (fusion) devices in comparison with the established standard of pedicle screw-rod fixation. Clinical trials are also needed to evaluate these devices when used alone for decompression. The evidence is insufficient to determine the effects of the technology on health outcomes. Clinical Input Received From Physician Specialty Societies and Academic Medical Centers While the various physician specialty societies and academic medical centers may collaborate with and make recommendations during this process through the provision of appropriate reviewers, input received does not represent an endorsement or position statement by the physician specialty societies or academic medical centers, unless otherwise noted. In response to requests, input was received from 3 physician specialty societies (2 reviewers) and 2 academic medical centers while this policy was under review in The input was mixed. Some cases where the devices might be medically necessary were noted, such as patients with small pedicles where pedicle screws could not be safely placed. Practice Guidelines and Position Statements The North American Spine Society (NASS) issued a coverage position on the use of interspinous devices with lumbar fusion. 6 NASS recommends that interspinous fixation with fusion for stabilization is currently not indicated as an alternative to pedicle screw fixation with lumbar fusion procedures. U.S. Preventive Services Task Force Recommendations Use of interspinous fixation devices is not a preventive service. CODING The following codes for treatment and procedures applicable to this policy are included below for informational purposes. Inclusion or exclusion of a procedure, diagnosis or device code(s) does not constitute or imply member coverage or provider reimbursement. Please refer to the member's contract benefits in effect at the time of service to determine coverage or noncoverage of these services as it applies to an individual member. CPT/HCPCS Posterior non-segmental instrumentation (eg, Harrington rod technique, pedicle fixation across 1 interspace, atlantoaxial transarticular screw fixation, sublaminar wiring at C1, facet screw fixation) (List separately in addition to code for primary procedure) Application of intervertebral biomechanical device(s) (eg, synthetic cage(s), methylmethacrylate) to vertebral defect or interspace (List separately in addition to code for primary procedure)
6 Interspinous Fixation (Fusion) Devices Page 6 of 6 There are no specific CPT codes for insertion of these devices. Diagnoses Experimental / Investigational for all diagnoses related to this medical policy. REVISIONS Policy added to the bcbsks.com web site on ; effective , 30 days after posting Updated Description section. Updated Rationale section. Updated References section. REFERENCES 1. Wu JC, Mummaneni PV. Using lumbar interspinous anchor with transforaminal lumbar interbody fixation. World Neurosurg. May 2010;73(5): PMID Kim DH, Shanti N, Tantorski ME, et al. Association between degenerative spondylolisthesis and spinous process fracture after interspinous process spacer surgery. Spine J. Jun 2012;12(6): PMID Oppenheim JS, Mills J. Recurrent lumbar disc herniation treated with interspinous fusion and instrumentation: a case series. Surg Technol Int. Sep 2013;23: PMID Vokshoor A, Khurana S, Wilson D, et al. Clinical and radiographic outcomes after spinous process fixation and posterior fusion in an elderly cohort. Surg Technol Int. Nov 2014;25: PMID Sclafani JA, Liang K, Ohnmeiss DD, et al. Clinical outcomes of a polyaxial interspinous fusion system. Int J Spine Surg. 2014;8. PMID North American Spine Society (NASS). NASS coverage policy recommendations: Interspinous fixation with fusion. ationwithfusion.pdf. Accessed July 6, 2015.
visualized. The correct level is then identified again. With the use of a microscope and
 SURGERY FOR SPINAL STENOSIS Laminectomy A one inch (or longer for extensive stenosis) incision is made in the middle of the back over the effected region of the spine. The muscles over the bone are moved
SURGERY FOR SPINAL STENOSIS Laminectomy A one inch (or longer for extensive stenosis) incision is made in the middle of the back over the effected region of the spine. The muscles over the bone are moved
Reimbursement Overview Supporting Adjunctive Use of the coflex-f Implant During Lumbar Fusion Procedures
 Interlaminar Technology Reimbursement Overview Supporting Adjunctive Use of the coflex-f Implant During Lumbar Fusion Procedures Coding Recommendations Overview Implant Description & Device Type Differentiation
Interlaminar Technology Reimbursement Overview Supporting Adjunctive Use of the coflex-f Implant During Lumbar Fusion Procedures Coding Recommendations Overview Implant Description & Device Type Differentiation
Minimally Invasive Lumbar Fusion
 Minimally Invasive Lumbar Fusion Biomechanical Evaluation (1) coflex-f screw Biomechanical Evaluation (1) coflex-f intact Primary Stability intact Primary Stability Extension Neutral Position Flexion Coflex
Minimally Invasive Lumbar Fusion Biomechanical Evaluation (1) coflex-f screw Biomechanical Evaluation (1) coflex-f intact Primary Stability intact Primary Stability Extension Neutral Position Flexion Coflex
Patient Information. Lateral Lumbar Interbody Fusion Surgery (LLIF).
 Patient Information. Lateral Lumbar Interbody Fusion Surgery (LLIF). Understanding your spine Disc Between each pair of vertebrae there is a disc that acts as a cushion to protect the vertebra, allows
Patient Information. Lateral Lumbar Interbody Fusion Surgery (LLIF). Understanding your spine Disc Between each pair of vertebrae there is a disc that acts as a cushion to protect the vertebra, allows
Subject: Implanted Devices for Spinal Stenosis Policy #: SURG.00092 Current Effective Date: 07/13/2011 Status: Reviewed Last Review Date: 05/19/2011
 1 of 5 6/18/2012 11:08 AM Medical Policy Subject: Implanted Devices for Spinal Stenosis Policy #: SURG.00092 Current Effective Date: 07/13/2011 Status: Reviewed Last Review Date: 05/19/2011 Description/Scope
1 of 5 6/18/2012 11:08 AM Medical Policy Subject: Implanted Devices for Spinal Stenosis Policy #: SURG.00092 Current Effective Date: 07/13/2011 Status: Reviewed Last Review Date: 05/19/2011 Description/Scope
Presented by Zoran Maric, M.D. Orthopaedic Spine Surgeon May 22, 2010
 Presented by Zoran Maric, M.D. Orthopaedic Spine Surgeon May 22, 2010 1 cervical area thoracic area lumbar area sacrum coccyx Mayfield Clinic 2 3 4 5 Zoran Maric, MD Spine Surgery Procedures How to Document
Presented by Zoran Maric, M.D. Orthopaedic Spine Surgeon May 22, 2010 1 cervical area thoracic area lumbar area sacrum coccyx Mayfield Clinic 2 3 4 5 Zoran Maric, MD Spine Surgery Procedures How to Document
Issued and entered this _6th_ day of October 2010 by Ken Ross Commissioner ORDER I PROCEDURAL BACKGROUND
 STATE OF MICHIGAN DEPARTMENT OF ENERGY, LABOR & ECONOMIC GROWTH OFFICE OF FINANCIAL AND INSURANCE REGULATION Before the Commissioner of Financial and Insurance Regulation In the matter of XXXXX Petitioner
STATE OF MICHIGAN DEPARTMENT OF ENERGY, LABOR & ECONOMIC GROWTH OFFICE OF FINANCIAL AND INSURANCE REGULATION Before the Commissioner of Financial and Insurance Regulation In the matter of XXXXX Petitioner
CERVICAL PROCEDURES PHYSICIAN CODING
 CERVICAL PROCEDURES PHYSICIAN CODING Anterior Cervical Discectomy with Interbody Fusion (ACDF) Anterior interbody fusion, with discectomy and decompression; cervical below C2 22551 first interspace 22552
CERVICAL PROCEDURES PHYSICIAN CODING Anterior Cervical Discectomy with Interbody Fusion (ACDF) Anterior interbody fusion, with discectomy and decompression; cervical below C2 22551 first interspace 22552
Motion Preservation. Hansen Yuan, MD President, Spine Arthroplasty Society
 Motion Preservation Procedure Codes Hansen Yuan, MD President, Spine Arthroplasty Society Who are we? The Spine Arthroplasty Society (SAS) is a group of medical and associated specialists devoted to the
Motion Preservation Procedure Codes Hansen Yuan, MD President, Spine Arthroplasty Society Who are we? The Spine Arthroplasty Society (SAS) is a group of medical and associated specialists devoted to the
Dynamic stabilization of the lumbar spine Robert W. Molinari
 Dynamic stabilization of the lumbar spine Robert W. Molinari Purpose of review This is a review of the recent literature involving dynamic posterior stabilization in the lumbar spine. Recent findings As
Dynamic stabilization of the lumbar spine Robert W. Molinari Purpose of review This is a review of the recent literature involving dynamic posterior stabilization in the lumbar spine. Recent findings As
CPT Changes in Spine 2012
 CPT Changes in Spine 2012 Are you prepared? Presented by Barbara Cataletto, MBA, CPC Disclaimer The following presentations are not to be considered a replacement for the Current Procedural Terminology
CPT Changes in Spine 2012 Are you prepared? Presented by Barbara Cataletto, MBA, CPC Disclaimer The following presentations are not to be considered a replacement for the Current Procedural Terminology
SURGICAL TREATMENT FOR SPINE PAIN
 Lots of MEDICAL POLICY SURGICAL TREATMENT FOR SPINE PAIN Policy Number: 2015T0547J Effective Date: April 1, 2015 Table of Contents BENEFIT CONSIDERATIONS COVERAGE RATIONALE APPLICABLE CODES.. DESCRIPTION
Lots of MEDICAL POLICY SURGICAL TREATMENT FOR SPINE PAIN Policy Number: 2015T0547J Effective Date: April 1, 2015 Table of Contents BENEFIT CONSIDERATIONS COVERAGE RATIONALE APPLICABLE CODES.. DESCRIPTION
Low Back Pain (LBP) Prevalence. Low Back Pain (LBP) Prevalence. Lumbar Fusion: Where is the Evidence?
 15 th Annual Cleveland Clinic Pain Management Symposium Sarasota, Florida Lumbar Fusion: Where is the Evidence? Gordon R. Bell, M.D. Director, Cleveland Clinic Low Back Pain (LBP) Prevalence Lifetime prevalence:
15 th Annual Cleveland Clinic Pain Management Symposium Sarasota, Florida Lumbar Fusion: Where is the Evidence? Gordon R. Bell, M.D. Director, Cleveland Clinic Low Back Pain (LBP) Prevalence Lifetime prevalence:
Degenerative Spine Solutions
 Degenerative Spine Solutions The Backbone for Your Surgical Needs Aesculap Spine Backbone for Your Degenerative Spine Needs Comprehensive operative solutions, unique product technology and world-class
Degenerative Spine Solutions The Backbone for Your Surgical Needs Aesculap Spine Backbone for Your Degenerative Spine Needs Comprehensive operative solutions, unique product technology and world-class
LUMBAR SPINAL STENOSIS OBSERVATIONS, EVIDENCE, AND TRENDS FULILLING THE UNMET CLINICAL NEED WRITTEN BY: HALLETT MATHEWS, MD, MBA
 LUMBAR SPINAL STENOSIS OBSERVATIONS, EVIDENCE, AND TRENDS FULILLING THE UNMET CLINICAL NEED WRITTEN BY: HALLETT MATHEWS, MD, MBA Overview of Lumbar Spinal Stenosis Spine stabilization, which has equated
LUMBAR SPINAL STENOSIS OBSERVATIONS, EVIDENCE, AND TRENDS FULILLING THE UNMET CLINICAL NEED WRITTEN BY: HALLETT MATHEWS, MD, MBA Overview of Lumbar Spinal Stenosis Spine stabilization, which has equated
Value Analysis Brief MIS Lateral Approach to Interbody Fusion
 Value Analysis Brief MIS Lateral Approach to Interbody Fusion Methods This value analysis brief presents information on the clinical and economic benefits of the minimally invasive lateral approach to
Value Analysis Brief MIS Lateral Approach to Interbody Fusion Methods This value analysis brief presents information on the clinical and economic benefits of the minimally invasive lateral approach to
Surgical Treatment for Lumbar Spinal Stenosis Dynamic Interspinous Distraction Interlaminar Stabilization Implant - Coflex
 International 31st Course For Percutaneous Endoscopic Spinal Surgery And Complementary Minimal Invasive Techniques Zurich, Switzerland January 24-25, 2013 Surgical Treatment for Lumbar Spinal Stenosis
International 31st Course For Percutaneous Endoscopic Spinal Surgery And Complementary Minimal Invasive Techniques Zurich, Switzerland January 24-25, 2013 Surgical Treatment for Lumbar Spinal Stenosis
How To Understand The Anatomy Of A Lumbar Spine
 Sciatica: Low back and Leg Pain Diagnosis and Treatment Options Presented by Devesh Ramnath, MD Orthopaedic Associates Of Dallas Baylor Spine Center Sciatica Compression of the spinal nerves in the back
Sciatica: Low back and Leg Pain Diagnosis and Treatment Options Presented by Devesh Ramnath, MD Orthopaedic Associates Of Dallas Baylor Spine Center Sciatica Compression of the spinal nerves in the back
ISPI Newsletter Archive Lumbar Spine Surgery
 ISPI Newsletter Archive Lumbar Spine Surgery January 2005 Effects of Charite Artificial Disc on the Implanted and Adjacent Spinal Segments Mechanics Using a Hybrid Testing Protocol Spine. 30(24):2755-2764,
ISPI Newsletter Archive Lumbar Spine Surgery January 2005 Effects of Charite Artificial Disc on the Implanted and Adjacent Spinal Segments Mechanics Using a Hybrid Testing Protocol Spine. 30(24):2755-2764,
1 REVISOR 5223.0070. (4) Pain associated with rigidity (loss of motion or postural abnormality) or
 1 REVISOR 5223.0070 5223.0070 MUSCULOSKELETAL SCHEDULE; BACK. Subpart 1. Lumbar spine. The spine rating is inclusive of leg symptoms except for gross motor weakness, bladder or bowel dysfunction, or sexual
1 REVISOR 5223.0070 5223.0070 MUSCULOSKELETAL SCHEDULE; BACK. Subpart 1. Lumbar spine. The spine rating is inclusive of leg symptoms except for gross motor weakness, bladder or bowel dysfunction, or sexual
Patient Information. Anterior Cervical Discectomy and Fusion Surgery (ACDF).
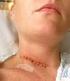 Patient Information. Anterior Cervical Discectomy and Fusion Surgery (ACDF). Understanding your spine Disc Between each pair of vertebrae there is a disc that acts as a cushion to protect the vertebra,
Patient Information. Anterior Cervical Discectomy and Fusion Surgery (ACDF). Understanding your spine Disc Between each pair of vertebrae there is a disc that acts as a cushion to protect the vertebra,
Complications in Adult Deformity Surgery
 Complications in Adult Deformity Surgery Proximal Junctional Kyphosis: Thoracolumbar and Cervicothoracic Sigurd Berven, MD Professor in Residence UC San Francisco Disclosures Research/Institutional Support:
Complications in Adult Deformity Surgery Proximal Junctional Kyphosis: Thoracolumbar and Cervicothoracic Sigurd Berven, MD Professor in Residence UC San Francisco Disclosures Research/Institutional Support:
Does the pain radiating down your legs, buttocks or lower back prevent you from walking long distances?
 Does the pain radiating down your legs, buttocks or lower back prevent you from walking long distances? Do you experience weakness, tingling, numbness, stiffness, or cramping in your legs, buttocks or
Does the pain radiating down your legs, buttocks or lower back prevent you from walking long distances? Do you experience weakness, tingling, numbness, stiffness, or cramping in your legs, buttocks or
Biomechanics of the Lumbar Spine
 Biomechanics of the Lumbar Spine Biomechanics of the Spine 6 degrees of freedom Extension & Flexion Translation Rotation Compression & Distraction The disc/annulus/all/pll complex is the major restraint
Biomechanics of the Lumbar Spine Biomechanics of the Spine 6 degrees of freedom Extension & Flexion Translation Rotation Compression & Distraction The disc/annulus/all/pll complex is the major restraint
Spinal Surgery Functional Status and Quality of Life Outcome Specifications 2015 (01/01/2013 to 12/31/2013 Dates of Procedure) September 2014
 Description Methodology For patients ages 18 years and older who undergo a lumbar discectomy/laminotomy or lumbar spinal fusion procedure during the measurement year, the following measures will be calculated:
Description Methodology For patients ages 18 years and older who undergo a lumbar discectomy/laminotomy or lumbar spinal fusion procedure during the measurement year, the following measures will be calculated:
Information for the Patient About Surgical
 Information for the Patient About Surgical Decompression and Stabilization of the Spine Aging and the Spine Daily wear and tear, along with disc degeneration due to aging and injury, are common causes
Information for the Patient About Surgical Decompression and Stabilization of the Spine Aging and the Spine Daily wear and tear, along with disc degeneration due to aging and injury, are common causes
Advances In Spine Care. James D. Bruffey M.D. Scripps Clinic Division of Orthopaedic Surgery Section of Spinal Surgery
 Advances In Spine Care James D. Bruffey M.D. Scripps Clinic Division of Orthopaedic Surgery Section of Spinal Surgery Introduction The Spine - A common source of problems Back pain is the #2 presenting
Advances In Spine Care James D. Bruffey M.D. Scripps Clinic Division of Orthopaedic Surgery Section of Spinal Surgery Introduction The Spine - A common source of problems Back pain is the #2 presenting
Minimally Invasive Spine Surgery What is it and how will it benefit patients?
 Minimally Invasive Spine Surgery What is it and how will it benefit patients? Dr Raoul Pope MBChB, FRACS, Neurosurgeon and Minimally Invasive Spine Surgeon Concord Hospital and Mater Private Hospital Sydney
Minimally Invasive Spine Surgery What is it and how will it benefit patients? Dr Raoul Pope MBChB, FRACS, Neurosurgeon and Minimally Invasive Spine Surgeon Concord Hospital and Mater Private Hospital Sydney
Medical Policy Manual. Topic: Interspinous and Interlaminar Stabilization/Distraction Devices (Spacers) Date of Origin: October 2006.
 Medical Policy Manual Topic: Interspinous and Interlaminar Stabilization/Distraction Devices (Spacers) Section: Surgery Date of Origin: October 2006 Last Reviewed Date: December 2015 Policy No: 155 Effective
Medical Policy Manual Topic: Interspinous and Interlaminar Stabilization/Distraction Devices (Spacers) Section: Surgery Date of Origin: October 2006 Last Reviewed Date: December 2015 Policy No: 155 Effective
ATLANTIS Anterior Cervical Plate System ATLANTIS Translational Plate. 510(k) Summary. February 2007
 rk,1 10{2 ATLANTIS Anterior Cervical Plate System ATLANTIS Translational Plate 510(k) Summary FEB 2 3 2[007 February 2007 I. Company: Medtronic Sofamor Danek USA 1800 Pyramid Place Memphis, Tennessee 38132
rk,1 10{2 ATLANTIS Anterior Cervical Plate System ATLANTIS Translational Plate 510(k) Summary FEB 2 3 2[007 February 2007 I. Company: Medtronic Sofamor Danek USA 1800 Pyramid Place Memphis, Tennessee 38132
SURGICAL TREATMENT FOR SPINE PAIN
 CLINICAL POLICY SURGICAL TREATMENT FOR SPINE PAIN Policy Number: SURGERY 075.11 T2 Effective Date: April 1, 2015 Table of Contents CONDITIONS OF COVERAGE... COVERAGE RATIONALE... BACKGROUND... CLINICAL
CLINICAL POLICY SURGICAL TREATMENT FOR SPINE PAIN Policy Number: SURGERY 075.11 T2 Effective Date: April 1, 2015 Table of Contents CONDITIONS OF COVERAGE... COVERAGE RATIONALE... BACKGROUND... CLINICAL
coligne treatment technology
 coligne treatment technology coligne a strategy in spine Leadership begins before the patient enters the surgical theater, among the spine care professionals forming the coalliance. This may be a small
coligne treatment technology coligne a strategy in spine Leadership begins before the patient enters the surgical theater, among the spine care professionals forming the coalliance. This may be a small
X Stop Spinal Stenosis Decompression
 X Stop Spinal Stenosis Decompression Am I a candidate for X Stop spinal surgery? You may be a candidate for the X Stop spinal surgery if you have primarily leg pain rather than mostly back pain and your
X Stop Spinal Stenosis Decompression Am I a candidate for X Stop spinal surgery? You may be a candidate for the X Stop spinal surgery if you have primarily leg pain rather than mostly back pain and your
DUKE ORTHOPAEDIC SURGERY GOALS AND OBJECTIVES SPINE SERVICE
 GOALS AND OBJECTIVES PATIENT CARE Able to perform a complete musculoskeletal and neurologic examination on the patient including cervical spine, thoracic spine, and lumbar spine. The neurologic examination
GOALS AND OBJECTIVES PATIENT CARE Able to perform a complete musculoskeletal and neurologic examination on the patient including cervical spine, thoracic spine, and lumbar spine. The neurologic examination
Moderator and Panelists
 Panelists Moderator and Panelists Moderator Dr. Hallett Mathews Executive Vice President Chief Medical Officer Paradigm Spine, LLC Panelist Dr. John Peloza Orthopedic Surgeon The Center for Spine Care
Panelists Moderator and Panelists Moderator Dr. Hallett Mathews Executive Vice President Chief Medical Officer Paradigm Spine, LLC Panelist Dr. John Peloza Orthopedic Surgeon The Center for Spine Care
September 25, 2014. Dear Ms. McCoy:
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 September 25, 2014 Biomet
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 September 25, 2014 Biomet
https://www.laserspineinstitute.com/back_problems/foraminal_stenosis/e...
 Questions? Call toll free 1-866-249-1627 Contact us today. We're here for you seven days a week. MRI Review Consultation Live help Call 1-866-249-1627 Chat Live Home Laser Spine Institute Laser Spine Institute's
Questions? Call toll free 1-866-249-1627 Contact us today. We're here for you seven days a week. MRI Review Consultation Live help Call 1-866-249-1627 Chat Live Home Laser Spine Institute Laser Spine Institute's
Patient Guide to Lower Back Surgery
 The following is a sampling of products offered by Zimmer Spine for use in Open Lumbar Fusion procedures. Patient Guide to Lower Back Surgery Open Lumbar Fusion Dynesys The Dynesys Dynamic Stabilization
The following is a sampling of products offered by Zimmer Spine for use in Open Lumbar Fusion procedures. Patient Guide to Lower Back Surgery Open Lumbar Fusion Dynesys The Dynesys Dynamic Stabilization
SPINAL FUSION. North American Spine Society Public Education Series
 SPINAL FUSION North American Spine Society Public Education Series WHAT IS SPINAL FUSION? The spine is made up of a series of bones called vertebrae ; between each vertebra are strong connective tissues
SPINAL FUSION North American Spine Society Public Education Series WHAT IS SPINAL FUSION? The spine is made up of a series of bones called vertebrae ; between each vertebra are strong connective tissues
Surgical Guideline for Lumbar Fusion (Arthrodesis)
 I. Introduction Surgical Guideline for Lumbar Fusion (Arthrodesis) The purpose of this guideline is: A. To provide utilization review staff with the information necessary to make recommendations about
I. Introduction Surgical Guideline for Lumbar Fusion (Arthrodesis) The purpose of this guideline is: A. To provide utilization review staff with the information necessary to make recommendations about
SURGICAL TREATMENT FOR SPINE PAIN
 MEDICAL POLICY SURGICAL TREATMENT FOR SPINE PAIN Policy Number: 2012T0547C Effective Date: April 1, 2012 Table of Contents COVERAGE RATIONALE... BACKGROUND... CLINICAL EVIDENCE... U.S. FOOD AND DRUG ADMINISTRATION...
MEDICAL POLICY SURGICAL TREATMENT FOR SPINE PAIN Policy Number: 2012T0547C Effective Date: April 1, 2012 Table of Contents COVERAGE RATIONALE... BACKGROUND... CLINICAL EVIDENCE... U.S. FOOD AND DRUG ADMINISTRATION...
Spine Clinic Neurospine Specialists, Orthopaedics and Neurosurgery
 Spine Clinic Neurospine Specialists, Orthopaedics and Neurosurgery REVISION SPINE SURGERY Revision surgery is a very complex field which requires experience, training and evaluation in a very individual
Spine Clinic Neurospine Specialists, Orthopaedics and Neurosurgery REVISION SPINE SURGERY Revision surgery is a very complex field which requires experience, training and evaluation in a very individual
Title: Interspinous Process Decompression with the X-Stop Device for Lumbar Spinal Stenosis: A Retrospective Review. Authors: Jennifer R.
 Title: Interspinous Process Decompression with the X-Stop Device for Lumbar Spinal Stenosis: A Retrospective Review. Authors: Jennifer R. Madonia-Barr, MS, PA-C and David L. Kramer, MD Institution: Connecticut
Title: Interspinous Process Decompression with the X-Stop Device for Lumbar Spinal Stenosis: A Retrospective Review. Authors: Jennifer R. Madonia-Barr, MS, PA-C and David L. Kramer, MD Institution: Connecticut
4052 Slimplicity Tech final_layout 1 6/29/15 3:29 PM Page 2 Surgical Technique
 Surgical Technique TABLE OF CONTENTS Slimplicity Anterior Cervical Plate System Overview 2 Indications 2 Implants 3 Instruments 4 Surgical Technique 6 1. Patient Positioning and Approach 6 2. Plate Selection
Surgical Technique TABLE OF CONTENTS Slimplicity Anterior Cervical Plate System Overview 2 Indications 2 Implants 3 Instruments 4 Surgical Technique 6 1. Patient Positioning and Approach 6 2. Plate Selection
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: lumbar_spine_fusion_surgery 9/2010 5/2015 5/2016 5/2015 Description of Procedure or Service Low back pain
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: lumbar_spine_fusion_surgery 9/2010 5/2015 5/2016 5/2015 Description of Procedure or Service Low back pain
Surgical Procedures of the Spine
 Surgical Procedures of the Spine Jaideep Chunduri, M.D. Orthopaedic Spine Surgeon Beacon Orthopaedics and Sports Medicine Beacon Spine Center Objectives Discuss the 4 most common procedures performed in
Surgical Procedures of the Spine Jaideep Chunduri, M.D. Orthopaedic Spine Surgeon Beacon Orthopaedics and Sports Medicine Beacon Spine Center Objectives Discuss the 4 most common procedures performed in
Corporate Medical Policy
 Corporate Medical Policy Diagnosis and Treatment of Sacroiliac Joint Pain File Name: Origination: Last CAP Review: Next CAP Review: Last Review: diagnosis_and_treatment_of_sacroiliac_joint_pain 8/2010
Corporate Medical Policy Diagnosis and Treatment of Sacroiliac Joint Pain File Name: Origination: Last CAP Review: Next CAP Review: Last Review: diagnosis_and_treatment_of_sacroiliac_joint_pain 8/2010
ANTERIOR LUMBAR INTERBODY FUSION (ALIF) Basic Anatomical Landmarks: Anterior Lumbar Spine
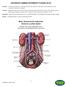 (ALIF) Anterior In human anatomy, referring to the front surface of the body or the position of one structure relative to another Lumbar Relating to the loins or the section of the back and sides between
(ALIF) Anterior In human anatomy, referring to the front surface of the body or the position of one structure relative to another Lumbar Relating to the loins or the section of the back and sides between
Spine Surgery Coding: Don t Break Your Neck Trying to Figure It Out
 Spine Surgery Coding: Don t Break Your Neck Trying to Figure It Out Presented to: AAPC Annual Meeting Orlando, Florida April 15, 2013 Presented by: Kim Pollock, RN, MBA, CPC www.karenzupko.com Kim Pollock,
Spine Surgery Coding: Don t Break Your Neck Trying to Figure It Out Presented to: AAPC Annual Meeting Orlando, Florida April 15, 2013 Presented by: Kim Pollock, RN, MBA, CPC www.karenzupko.com Kim Pollock,
Anterior Lumbar Interbody Fusion (ALIF). Instrument set supports placement of ALIF spacers using anterior or anterolateral approach.
 Anterior Lumbar Interbody Fusion (ALIF). Instrument set supports placement of ALIF spacers using anterior or anterolateral approach. Technique Guide Instruments and implants approved by the AO Foundation
Anterior Lumbar Interbody Fusion (ALIF). Instrument set supports placement of ALIF spacers using anterior or anterolateral approach. Technique Guide Instruments and implants approved by the AO Foundation
Policy #: 111 Latest Review Date: January 2010
 Name of Policy: Co-surgeons and Team Surgeons Policy #: 111 Latest Review Date: January 2010 Category: Administrative Policy Grade: N/A Background: As a general rule, benefits are payable under Blue Cross
Name of Policy: Co-surgeons and Team Surgeons Policy #: 111 Latest Review Date: January 2010 Category: Administrative Policy Grade: N/A Background: As a general rule, benefits are payable under Blue Cross
Spinal Surgery 2. Teaching Aims. Common Spinal Pathologies. Disc Degeneration. Disc Degeneration. Causes of LBP 8/2/13. Common Spinal Conditions
 Teaching Aims Spinal Surgery 2 Mr Mushtaque A. Ishaque BSc(Hons) BChir(Cantab) DM FRCS FRCS(Ed) FRCS(Orth) Hunterian Professor at The Royal College of Surgeons of England Consultant Orthopaedic Spinal
Teaching Aims Spinal Surgery 2 Mr Mushtaque A. Ishaque BSc(Hons) BChir(Cantab) DM FRCS FRCS(Ed) FRCS(Orth) Hunterian Professor at The Royal College of Surgeons of England Consultant Orthopaedic Spinal
Treatment pathways in lumbar spinal stenosis. By Prof. Dr. H. Michael Mayer
 Treatment pathways in lumbar spinal stenosis By Prof. Dr. H. Michael Mayer Content The pathology of spinal stenosis 4 The treatment of spinal stenosis 6 Spinal stenosis treatment options and outcomes 8
Treatment pathways in lumbar spinal stenosis By Prof. Dr. H. Michael Mayer Content The pathology of spinal stenosis 4 The treatment of spinal stenosis 6 Spinal stenosis treatment options and outcomes 8
Spinal Decompression
 Spinal Decompression Spinal decompression is just one more tool we have to treat radiculopathy. With appropriate education and exercises, this modality has been proven to assist in the resolution of symptoms
Spinal Decompression Spinal decompression is just one more tool we have to treat radiculopathy. With appropriate education and exercises, this modality has been proven to assist in the resolution of symptoms
Update to the Treatment of Degenerative Cervical Disc Disease
 Update to the Treatment of Degenerative Cervical Disc Disease Michael Lynn, MD Neurosurgeon, Southeastern Neurosurgical & Spine Institute Adjunct Assistant Clinical Professor of Bioengineering, Clemson
Update to the Treatment of Degenerative Cervical Disc Disease Michael Lynn, MD Neurosurgeon, Southeastern Neurosurgical & Spine Institute Adjunct Assistant Clinical Professor of Bioengineering, Clemson
Spinal Surgery Clinical Coverage Policy No: 1A-30 Revised Date: DRAFT Table of Contents
 Clinical Coverage Policy No: 1A-30 Table of Contents 1.0 Description of the Procedure, Product, or Service... 1 1.1 Definitions... 1 2.0 Eligible Recipients... 2 2.1 General Provisions... 2 2.2 EPSDT Special
Clinical Coverage Policy No: 1A-30 Table of Contents 1.0 Description of the Procedure, Product, or Service... 1 1.1 Definitions... 1 2.0 Eligible Recipients... 2 2.1 General Provisions... 2 2.2 EPSDT Special
Artificial Intervertebral Disc: Cervical Spine
 dapplies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
dapplies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
White Paper: Reducing Utilization Concerns Regarding Spinal Fusion and Artificial Disc Implants
 White Paper: Reducing Utilization Concerns Regarding Spinal Fusion and Artificial Disc Implants For Health Plans, Medical Management Organizations and TPAs Executive Summary Back pain is one of the most
White Paper: Reducing Utilization Concerns Regarding Spinal Fusion and Artificial Disc Implants For Health Plans, Medical Management Organizations and TPAs Executive Summary Back pain is one of the most
Minimally Invasive Spine Surgery For Your Patients
 Minimally Invasive Spine Surgery For Your Patients Lukas P. Zebala, M.D. Assistant Professor Orthopaedic and Neurological Spine Surgery Department of Orthopaedic Surgery Washington University School of
Minimally Invasive Spine Surgery For Your Patients Lukas P. Zebala, M.D. Assistant Professor Orthopaedic and Neurological Spine Surgery Department of Orthopaedic Surgery Washington University School of
Bone Morphogenetic Proteins & Spinal Surgery for Degenarative Disc Disease
 Ontario Health Technology Assessment Series 2004; Vol. 4, No. 4 Bone Morphogenetic Proteins & Spinal Surgery for Degenarative Disc Disease An Evidence Based Analysis March 2004 Medical Advisory Secretariat
Ontario Health Technology Assessment Series 2004; Vol. 4, No. 4 Bone Morphogenetic Proteins & Spinal Surgery for Degenarative Disc Disease An Evidence Based Analysis March 2004 Medical Advisory Secretariat
EXECUTIVE SUMMARY: SPINAL DEVICES SUPPLIED BY PHYSICIAN- OWNED DISTRIBUTORS: OVERVIEW OF PREVALENCE AND USE OEI-01-11-00660
 EXECUTIVE SUMMARY: SPINAL DEVICES SUPPLIED BY PHYSICIAN- OWNED DISTRIBUTORS: OVERVIEW OF PREVALENCE AND USE OEI-01-11-00660 WHY WE DID THIS STUDY This report responds to a congressional request to determine
EXECUTIVE SUMMARY: SPINAL DEVICES SUPPLIED BY PHYSICIAN- OWNED DISTRIBUTORS: OVERVIEW OF PREVALENCE AND USE OEI-01-11-00660 WHY WE DID THIS STUDY This report responds to a congressional request to determine
Instrumented in situ posterolateral fusion for low-grade lytic spondylolisthesis in adults
 Acta Orthop. Belg., 2005, 71, 83-87 ORIGINAL STUDY Instrumented in situ posterolateral fusion for low-grade lytic spondylolisthesis in adults Mohamed A. EL MASRY, Walaa I. EL ASSUITY, Youssry K. EL HAWARY,
Acta Orthop. Belg., 2005, 71, 83-87 ORIGINAL STUDY Instrumented in situ posterolateral fusion for low-grade lytic spondylolisthesis in adults Mohamed A. EL MASRY, Walaa I. EL ASSUITY, Youssry K. EL HAWARY,
SPINAL FUSION SURGERY
 SPINAL FUSION SURGERY Policy Number: 2015M0052A Effective Date: July 1, 2015 Table of Contents: Page: Cross Reference Policy: POLICY DESCRIPTION 2 Spinal Cord Stimulator, 2014M0002B COVERAGE RATIONALE/CLINICAL
SPINAL FUSION SURGERY Policy Number: 2015M0052A Effective Date: July 1, 2015 Table of Contents: Page: Cross Reference Policy: POLICY DESCRIPTION 2 Spinal Cord Stimulator, 2014M0002B COVERAGE RATIONALE/CLINICAL
Direct Lateral Interbody Fusion A Minimally Invasive Approach to Spinal Stabilization
 APPROVED IRN10389-1.1-04 Direct Lateral Interbody Fusion A Minimally Invasive Approach to Spinal Stabilization Because it involves accessing the spine through the patient s side, the Direct Lateral approach
APPROVED IRN10389-1.1-04 Direct Lateral Interbody Fusion A Minimally Invasive Approach to Spinal Stabilization Because it involves accessing the spine through the patient s side, the Direct Lateral approach
Corporate Medical Policy Implantation of Intrastromal Corneal Ring Segments
 Corporate Medical Policy Implantation of Intrastromal Corneal Ring Segments File Name: Origination: Last CAP Review: Next CAP Review: Last Review: implantation_of_intrastromal_corneal_ring_segments 8/2008
Corporate Medical Policy Implantation of Intrastromal Corneal Ring Segments File Name: Origination: Last CAP Review: Next CAP Review: Last Review: implantation_of_intrastromal_corneal_ring_segments 8/2008
Research Article Partial Facetectomy for Lumbar Foraminal Stenosis
 Advances in Orthopedics, Article ID 534658, 4 pages http://dx.doi.org/10.1155/2014/534658 Research Article Partial Facetectomy for Lumbar Foraminal Stenosis Kevin Kang, 1 Juan Carlos Rodriguez-Olaverri,
Advances in Orthopedics, Article ID 534658, 4 pages http://dx.doi.org/10.1155/2014/534658 Research Article Partial Facetectomy for Lumbar Foraminal Stenosis Kevin Kang, 1 Juan Carlos Rodriguez-Olaverri,
Magnetic Resonance Imaging
 Magnetic Resonance Imaging North American Spine Society Public Education Series What Is Magnetic Resonance Imaging (MRI)? Magnetic resonance imaging (MRI) is a valuable diagnostic study that has been used
Magnetic Resonance Imaging North American Spine Society Public Education Series What Is Magnetic Resonance Imaging (MRI)? Magnetic resonance imaging (MRI) is a valuable diagnostic study that has been used
POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS DISCLAIMER CODING INFORMATION REFERENCES POLICY HISTORY
 Original Issue Date (Created): 6/14/2004 Most Recent Review Date (Revised): 9/29/2015 Effective Date: 12/1/2015 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS
Original Issue Date (Created): 6/14/2004 Most Recent Review Date (Revised): 9/29/2015 Effective Date: 12/1/2015 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS
How to Overcome the 5 Biggest Reimbursement Challenges in Joint & Spine Coding
 How to Overcome the 5 Biggest Reimbursement Challenges in Joint & Spine Coding Presented by: Carolyn Neumann, CPC Senior Manager Coding and Coverage Access The opinions and codes denoted within are suggestions
How to Overcome the 5 Biggest Reimbursement Challenges in Joint & Spine Coding Presented by: Carolyn Neumann, CPC Senior Manager Coding and Coverage Access The opinions and codes denoted within are suggestions
TABLE OF CONTENTS. Surgical Technique 2. Indications 4. Product Information 5. 1. Patient Positioning and Approach 2
 Surgical Technique TABLE OF CONTENTS Surgical Technique 2 1. Patient Positioning and Approach 2 2. Intervertebral Device Implanted 2 3. Buttress Plate Selection 2 4. Awl Insertion 2 5. Screw Insertion
Surgical Technique TABLE OF CONTENTS Surgical Technique 2 1. Patient Positioning and Approach 2 2. Intervertebral Device Implanted 2 3. Buttress Plate Selection 2 4. Awl Insertion 2 5. Screw Insertion
Lumbar Spinal Stenosis
 Lumbar Spinal Stenosis North American Spine Society Public Education Series What Is Lumbar Spinal Stenosis? The vertebrae are the bones that make up the lumbar spine (low back). The spinal canal runs through
Lumbar Spinal Stenosis North American Spine Society Public Education Series What Is Lumbar Spinal Stenosis? The vertebrae are the bones that make up the lumbar spine (low back). The spinal canal runs through
2013 North American SSL Certificate
 2013 2014 INSERT COMPANY LOGO HERE 2014 North American 2013 North American SSL Certificate Minimally Invasive Spinal Surgical Solutions Product Product Line Leadership Strategy Leadership Award Award Background
2013 2014 INSERT COMPANY LOGO HERE 2014 North American 2013 North American SSL Certificate Minimally Invasive Spinal Surgical Solutions Product Product Line Leadership Strategy Leadership Award Award Background
Patient Guide to Neck Surgery
 The following is a sampling of products offered by Zimmer Spine for use in Anterior Cervical Fusion procedures. Patient Guide to Neck Surgery Anterior Cervical Fusion Trinica Select With the Trinica and
The following is a sampling of products offered by Zimmer Spine for use in Anterior Cervical Fusion procedures. Patient Guide to Neck Surgery Anterior Cervical Fusion Trinica Select With the Trinica and
Do you have Back Pain? Associated with:
 Do you have Back Pain? Associated with: Herniated Discs? Protruding Discs? Degenerative Disk Disease? Posterior Facet Syndrome? Sciatica? You may be a candidate for Decompression Therapy The Dynatronics
Do you have Back Pain? Associated with: Herniated Discs? Protruding Discs? Degenerative Disk Disease? Posterior Facet Syndrome? Sciatica? You may be a candidate for Decompression Therapy The Dynatronics
LOW BACK PAIN. common of these conditions include: muscle strain ( pulled muscle ), weak core muscles
 LOW BACK PAIN Most episodes of low back pain are caused by relatively harmless conditions. The most common of these conditions include: muscle strain ( pulled muscle ), weak core muscles (abdominal and
LOW BACK PAIN Most episodes of low back pain are caused by relatively harmless conditions. The most common of these conditions include: muscle strain ( pulled muscle ), weak core muscles (abdominal and
Employees Compensation Appeals Board
 U. S. DEPARTMENT OF LABOR Employees Compensation Appeals Board In the Matter of DEBORAH R. EVANS and U.S. POSTAL SERVICE, POST OFFICE, Orlando, FL Docket No. 02-1888; Submitted on the Record; Issued December
U. S. DEPARTMENT OF LABOR Employees Compensation Appeals Board In the Matter of DEBORAH R. EVANS and U.S. POSTAL SERVICE, POST OFFICE, Orlando, FL Docket No. 02-1888; Submitted on the Record; Issued December
Jonathan R. Gottlieb, MD 7220 SW 127 Street Pinecrest, FL 33156 jonrgottlieb@gmail.com
 Jonathan R. Gottlieb, MD 7220 SW 127 Street Pinecrest, FL 33156 jonrgottlieb@gmail.com CURRENT PRACTICE September 2009-present University of Miami School of Medicine Assistant Professor, Department of
Jonathan R. Gottlieb, MD 7220 SW 127 Street Pinecrest, FL 33156 jonrgottlieb@gmail.com CURRENT PRACTICE September 2009-present University of Miami School of Medicine Assistant Professor, Department of
EXPERIMENTAL AND THERAPEUTIC MEDICINE 5: 567-571, 2013
 EXPERIMENTAL AND THERAPEUTIC MEDICINE 5: 567-571, 2013 Treatment of multilevel degenerative lumbar spinal stenosis with spondylolisthesis using a combination of microendoscopic discectomy and minimally
EXPERIMENTAL AND THERAPEUTIC MEDICINE 5: 567-571, 2013 Treatment of multilevel degenerative lumbar spinal stenosis with spondylolisthesis using a combination of microendoscopic discectomy and minimally
MINIMAL ACCESS SPINE SURGERY
 MINIMAL ACCESS SPINE SURGERY Spinal ailments are amongst the most common ailments that afflict mankind. Back pain, for example, has achieved epidemic proportions worldwide in the last two decades and is
MINIMAL ACCESS SPINE SURGERY Spinal ailments are amongst the most common ailments that afflict mankind. Back pain, for example, has achieved epidemic proportions worldwide in the last two decades and is
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Imaged-Guided Minimally Invasive Lumbar Page 1 of 9 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Image-Guided Minimally Invasive Lumbar Professional Institutional
Imaged-Guided Minimally Invasive Lumbar Page 1 of 9 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Image-Guided Minimally Invasive Lumbar Professional Institutional
Spine Surgery Coding
 Spine Surgery Coding Lynn M. Anderanin, CPC,CPC-I,COSC 1 Spine Anatomy 7 cervical (neck) vertebrae, 12 thoracic (chest) vertebrae, 5 lumbar (back) vertebrae, and 5 fused vertebra that make up the sacrum
Spine Surgery Coding Lynn M. Anderanin, CPC,CPC-I,COSC 1 Spine Anatomy 7 cervical (neck) vertebrae, 12 thoracic (chest) vertebrae, 5 lumbar (back) vertebrae, and 5 fused vertebra that make up the sacrum
Getting to the Backbone of Spinal Coding in ICD-10-PCS
 Getting to the Backbone of Spinal Coding in ICD-10-PCS Sponsored by 1915 N. Fine Ave #104 Fresno CA 93720-1565 Phone: (559) 251-5038 Fax: (559) 251-5836 www.californiahia.org Program Handouts Tuesday,
Getting to the Backbone of Spinal Coding in ICD-10-PCS Sponsored by 1915 N. Fine Ave #104 Fresno CA 93720-1565 Phone: (559) 251-5038 Fax: (559) 251-5836 www.californiahia.org Program Handouts Tuesday,
Introduction to Non-fusion Technologies in the Lumbar and Cervical Spine
 Introduction to Non-fusion Technologies in the Lumbar and Cervical Spine Introduction The term motion-sparing technology has been used to describe newer surgical techniques designed to preserve spinal
Introduction to Non-fusion Technologies in the Lumbar and Cervical Spine Introduction The term motion-sparing technology has been used to describe newer surgical techniques designed to preserve spinal
National Medical Policy
 National Medical Policy Subject: Policy Number: Vertebral Axial Decompression (VAX-D) NMP42 Effective Date*: October 2003 Updated: November 2015 This National Medical Policy is subject to the terms in
National Medical Policy Subject: Policy Number: Vertebral Axial Decompression (VAX-D) NMP42 Effective Date*: October 2003 Updated: November 2015 This National Medical Policy is subject to the terms in
Options for Cervical Disc Degeneration A Guide to the Fusion Arm of the M6 -C Artificial Disc Study
 Options for Cervical Disc Degeneration A Guide to the Fusion Arm of the M6 -C Artificial Disc Study Each year, hundreds of thousands of adults are diagnosed with Cervical Disc Degeneration, an upper spine
Options for Cervical Disc Degeneration A Guide to the Fusion Arm of the M6 -C Artificial Disc Study Each year, hundreds of thousands of adults are diagnosed with Cervical Disc Degeneration, an upper spine
Aetna Injection Policy For Back Pain
 Aetna Injection Policy For Back Pain Number: 0016 Policy Aetna considers any of the following injections or procedures medically necessary for the treatment of back pain; provided, however, that only 1
Aetna Injection Policy For Back Pain Number: 0016 Policy Aetna considers any of the following injections or procedures medically necessary for the treatment of back pain; provided, however, that only 1
Utilization and Cost of Surgery for Lumbar Spinal Stenosis in a Commercially Insured Population
 Utilization and Cost of Surgery for Lumbar Spinal Stenosis in a Commercially Insured Population Prepared by Milliman, Inc. Kathryn Fitch, RN, MEd Principal and Healthcare Management Consultant Helen Blumen,
Utilization and Cost of Surgery for Lumbar Spinal Stenosis in a Commercially Insured Population Prepared by Milliman, Inc. Kathryn Fitch, RN, MEd Principal and Healthcare Management Consultant Helen Blumen,
PAUL PARK, M.D., 1 AND KEVIN T. FOLEY, M.D. 2. Methods
 Neurosurg Focus 25 (2):E16, 2008 Minimally invasive transforaminal lumbar interbody fusion with reduction of spondylolisthesis: technique and outcomes after a minimum of 2 years follow-up PAUL PARK, M.D.,
Neurosurg Focus 25 (2):E16, 2008 Minimally invasive transforaminal lumbar interbody fusion with reduction of spondylolisthesis: technique and outcomes after a minimum of 2 years follow-up PAUL PARK, M.D.,
Discogenic Pain: Diagnosis and Treatment
 Discogenic Pain: Diagnosis and Treatment James P. Rathmell, M.D. Professor of Anesthesiology University of Vermont College of Medicine Burlington, Vermont, USA Cottage Sunrise Westford, Vermont 2005 Learning
Discogenic Pain: Diagnosis and Treatment James P. Rathmell, M.D. Professor of Anesthesiology University of Vermont College of Medicine Burlington, Vermont, USA Cottage Sunrise Westford, Vermont 2005 Learning
The goals of modern spinal surgery are to maximize
 SPRING 2013 Robot-Guided Spine Surgery Christopher R. Good, M.D., F.A.C.S. and Blair K. Snyder, P.A.-C. The goals of modern spinal surgery are to maximize patient function and accelerate a return to a
SPRING 2013 Robot-Guided Spine Surgery Christopher R. Good, M.D., F.A.C.S. and Blair K. Snyder, P.A.-C. The goals of modern spinal surgery are to maximize patient function and accelerate a return to a
Spine Trauma: When to Transfer. Alexander Ching, MD Director, Orthopaedic Spine Trauma OHSU
 Spine Trauma: When to Transfer Alexander Ching, MD Director, Orthopaedic Spine Trauma OHSU Disclosures Depuy Spine Consultant (teaching and courses) Department education and research funds Atlas Spine
Spine Trauma: When to Transfer Alexander Ching, MD Director, Orthopaedic Spine Trauma OHSU Disclosures Depuy Spine Consultant (teaching and courses) Department education and research funds Atlas Spine
Degenerative Lumbar Scoliosis with Stenosis Successfully Treated with Cox Distraction Manipulation
 Cox Technic Case Report #92 (sent February 2011 2/8/11 ) 1 Degenerative Lumbar Scoliosis with Stenosis Successfully Treated with Cox Distraction Manipulation Presented By Robert E. Patterson Jr., D.C.
Cox Technic Case Report #92 (sent February 2011 2/8/11 ) 1 Degenerative Lumbar Scoliosis with Stenosis Successfully Treated with Cox Distraction Manipulation Presented By Robert E. Patterson Jr., D.C.
Surgical Spine Care Center
 OUTCOMES DIVISION OF MEDICINE Surgical Spine Care Center About Cleveland Clinic Florida Cleveland Clinic Florida s medical staff are dedicated physicians who have joined the clinic as salaried doctors
OUTCOMES DIVISION OF MEDICINE Surgical Spine Care Center About Cleveland Clinic Florida Cleveland Clinic Florida s medical staff are dedicated physicians who have joined the clinic as salaried doctors
ARTIFICIAL DISC REPLACEMENT FOR DEGENERATIVE DISC DISEASE OF THE LUMBAR SPINE. A Technology Assessment
 ARTIFICIAL DISC REPLACEMENT FOR DEGENERATIVE DISC DISEASE OF THE LUMBAR SPINE A Technology Assessment INTRODUCTION The California Technology Assessment Forum has been asked to update its review of the
ARTIFICIAL DISC REPLACEMENT FOR DEGENERATIVE DISC DISEASE OF THE LUMBAR SPINE A Technology Assessment INTRODUCTION The California Technology Assessment Forum has been asked to update its review of the
Vertebral Axial Decompression For Low Back Pain
 Vertebral Axial Decompression For Low Back Pain By WCB Evidence Based Practice Group Dr. Craig W. Martin, Senior Medical Advisor February 2005 Program Design Division TABLE OF CONTENTS Page Background...1
Vertebral Axial Decompression For Low Back Pain By WCB Evidence Based Practice Group Dr. Craig W. Martin, Senior Medical Advisor February 2005 Program Design Division TABLE OF CONTENTS Page Background...1
STATE OF WEST VIRGINIA SUPREME COURT OF APPEALS MEMORANDUM DECISION
 STATE OF WEST VIRGINIA GARY E. GOSNELL, Claimant Below, Petitioner SUPREME COURT OF APPEALS FILED March 27, 2015 RORY L. PERRY II, CLERK SUPREME COURT OF APPEALS OF WEST VIRGINIA vs.) No. 14-0614 (BOR
STATE OF WEST VIRGINIA GARY E. GOSNELL, Claimant Below, Petitioner SUPREME COURT OF APPEALS FILED March 27, 2015 RORY L. PERRY II, CLERK SUPREME COURT OF APPEALS OF WEST VIRGINIA vs.) No. 14-0614 (BOR
The Effects of Cox Decompression Technic in the Treatment of Low Back Pain and Sciatica in a Golf Professional
 Cox Technic Email Case Report 72, June 2009, James Schantz DC 1 The Effects of Cox Decompression Technic in the Treatment of Low Back Pain and Sciatica in a Golf Professional James E. Schantz, D.C. Leading
Cox Technic Email Case Report 72, June 2009, James Schantz DC 1 The Effects of Cox Decompression Technic in the Treatment of Low Back Pain and Sciatica in a Golf Professional James E. Schantz, D.C. Leading
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Afrezza Page 1 of 6 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Afrezza (human insulin) Prime Therapeutics will review Prior Authorization requests Prior Authorization
Afrezza Page 1 of 6 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Afrezza (human insulin) Prime Therapeutics will review Prior Authorization requests Prior Authorization
Artificial Intervertebral Disc: Lumbar Spine
 MEDICAL POLICY POLICY RELATED POLICIES POLICY GUIDELINES DESCRIPTION SCOPE BENEFIT APPLICATION RATIONALE REFERENCES CODING APPENDIX HISTORY Artificial Intervertebral Disc: Lumbar Spine Number 7.01.87 Effective
MEDICAL POLICY POLICY RELATED POLICIES POLICY GUIDELINES DESCRIPTION SCOPE BENEFIT APPLICATION RATIONALE REFERENCES CODING APPENDIX HISTORY Artificial Intervertebral Disc: Lumbar Spine Number 7.01.87 Effective
TABLE OF CONTENTS. Indications and Contraindications 3. Features and Benefits 4. Ease of connection 4 Stable fixation concept 5
 Surgical Technique TABLE OF CONTENTS Indications and Contraindications 3 Features and Benefits 4 Ease of connection 4 Stable fixation concept 5 Surgical Technique Steps 6 Identification and preparation
Surgical Technique TABLE OF CONTENTS Indications and Contraindications 3 Features and Benefits 4 Ease of connection 4 Stable fixation concept 5 Surgical Technique Steps 6 Identification and preparation
