S8 CD studies. Fig. S5 CD spectra recorded at 20 C of mir-509-3p/pna1 and mir-509-3p/pna2 S9. Cell line, Construct and Transfections
|
|
|
- Andrea McGee
- 8 years ago
- Views:
Transcription
1 Design, synthesis and biochemical investigation, by in vitro luciferase reporter system, of peptide nucleic acids as new inhibitors of mir-509-3p involved in the regulation of cystic fibrosis disease-gene expression Felice Amato, a,b Rossella Tomaiuolo, a,b icola Borbone, c Ausilia Elce, a,b,d Jussara Amato, c Stefano D Errico, c Giuseppe De Rosa, c Laura Mayol, c Gennaro Piccialli, c Giorgia liviero*,c and Giuseppe Castaldo a,b a Dipartimento di Medicina Molecolare e Biotecnologie Mediche, Via Pansini 5, 80131, apoli, Italy b CEIGE-Biotecnologie avanzate, apoli, Italy c Dipartimento di Farmacia, Università degli Studi di apoli Federico II, Via D. Montesano 49, 80131, apoli, Italy. d Università Telematica Pegaso, apoli, Italy Contents Page umber Synthesis of mir-509-3p and PA1-3 S2 Synthesis of PA1 and PA3 S3 Synthesis of PA2 S5 Preparation of mir-509-3p/pa heteroduplexes S6 UV and UV melting studies S6 Fig. S1 UV profiles of both mir-509-3p and PA1 alone and in 1:1.5 molar ratio S6 Fig. S2 UV profiles of both mir-509-3p and PA2 alone and in 1:1.5 molar ratio S7 Fig. S3 UV melting profile of mir-509-3p/pa1 (1:1.5) heteroduplex S7 Fig. S4 UV melting profile of mir-509-3p/pa2 (1:1.5) heteroduplex S8 CD studies S9 Fig. S5 CD spectra recorded at 20 C of mir-509-3p/pa1 and mir-509-3p/pa2 S9 Cell line, Construct and Transfections S10 Fig. S6 Representative uptake of FITC-labeled PA1 (panel A) and PA2 (panel B) by A549 cells and their effect as mira inhibitors (panel C) in the absence of any transfection reagent. S11 Fig. S7 FITC-labeled PA1 (panel A) and PA2 (panel B) transfected in A549 cells. S12 Electrophoretic Mobility Shift Assay S13 Fig. S8 Ethidium Bromide staining of the EMSA. S13 S1
2 Synthesis of mir-509-3p and PA1-3 The mir-509-3p mimic (2 -Me modified) was purchased and purified by the oligonucleotide synthesis facility at CEIGE-Biotecnologie Avanzate. The sequences of PAs used in this study are listed in Table 1. Trifluoroacetic acid (TFA), dimethylformamide (DMF), dichloromethane (DCM),,-diisopropylethylamine (DIPEA), - methyl-pyrrolidone (MP), 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU), 2-(6-hydroxy-3-oxo-3xanthen-9-yl)-5-isothiocyanate-benzoic acid (FITC), phosphoric acid 3 P 4, 4-methylbenzhydrylamine-resin (MBA resin, 0.5 mmol/g), and all standard protected Fmoc-amino acid derivatives were purchased from Sigma-Aldrich. Fmoc-L-Ser[P(Bzl)]- building block, 2- (1--benzotriazol-1-yl)-1,1,3,3-tetramethyluronium hexafluorophosphate (BTU) 2-(1--7- azabenzotriazole-1-yl)-1,1,3,3-tetramethyluronium hexafluorophosphate (ATU) and 1- hydroxybenzotriazole (Bt) were purchased from ovabiochem. For PA synthesis, all Fmoc/Boc protected monomers and the 2-[2- (fluorenylmethoxycarbonylamino)ethoxy]ethoxyacetyc (AEEA) spacer-linker were purchased from Link Technologies. Analytical and semipreparative RP-PLC were performed at room temperature on a Jasco PU-2089 equipped with a Jasco UV-2070 UV/Vis detector. A Thermo C18 column ( mm, particle size 5 μm, particle pore diameter 100 Å) was used for analytical RP-PLC. For semipreparative PLC, a Merck C18 column ( mm, 5 μm, 100 Å) was used. The concentration of each PA has been evaluated by UV measurement using the calculated molar extinction coefficient value at 260 nm (see Table 1). Table 1. PA and mir-509-3p sequences used in this study. PA sequences are written from C to terminus. Molecule Sequence Molar extinction coefficient (M -1 * cm -1 ) PA-1 G-S(P)-S(P)-G-actaaccatgcaga-Linker-FITC 149,600 PA-2 PA-3 mira K-K-actaaccatgcaga-Linker-FITC G-S(P)-S(P)-G-tgccgagtaggcac-Linker-FITC UGAUUGGUACGUCUGUGGGUAG 149, , ,000 S2
3 P C 2 C PA 1,3 S P From C to terminus Synthesis of the bis-phosphate-pas (PA1 and PA3) The bis-phosphate-pa1,3 were prepared by solid-phase synthesis with Fmoc-strategy on a polystyrene-based MBA resin. The MBA resin (50 mg) was swollen in DCM for 30 min and washed four times with DMF. The resin was treated twice with a solution of 20% piperidine (2 ml) in DMF for 10 min, and subsequently washed four times with DMF. The resin was then functionalized with a Fmoc-glycine monomer using the following coupling conditions: Fmoc-Gly (5 eq., in MP 0.25M), ATU (3.6 eq. in DMF 0.2M) and DIPEA (5 eq.)/lutidine (6 eq.), 1 h at room temperature. In the synthesis the Fmoc group was removed by a treatment with a 5% DBU in DMF solution (5 min). In the case of Fmoc-Ser amino acids the basic treatment was prolonged (20 min). After each coupling step a capping procedure was performed using acetic anhydride (5%)/lutidine (6%) in DMF solution. Couplings of Fmoc-L-Ser[P(Bzl)]- were achieved using the following conditions: Fmoc- Ser monomer (8 eq. in MP 0.4M), ATU (8 eq. in DMF 0.4M) and DIPEA (8 eq.)/lutidine (12 eq.), 15 h at room temperature. After the serine couplings, a further glycine residue was attached on the -terminal of the serine tract following the previously described coupling with glycine monomer. The couplings with the PA monomers were carried out using the following conditions: Fmoc- PA monomer (8 eq. in MP 0.4M), ATU (8 eq. in DMF 0.4M) and DIPEA (8 eq.)/lutidine (12 eq.), 4 h at room temperature. The coupling with AEEA-C monomer (8 eq.) was performed using the same conditions described for PA couplings. S3
4 The FITC conjugation was performed using the following conditions: FITC monomer (5 eq., 0.2M) was dissolved in DMF/DIPEA (2.5:97.5 v/v) and the solution was added to the resin. The mixture was gently shaken in the dark for 15 h. The functionalized PA-FITC oligomer was detached from the resin using a treatment with TFA/anisole/ethanedithiol (9:0.5:0.5; v/v/v) for 3.5 h and precipitated with cold diethyl ether. This treatment assures the complete deprotection of the phosphate groups. The precipitated was collected by centrifugation and resuspended in ether twice. The pellet was dissolved in 0.1% 3 P 4 in water and lyophilized. The crude sample was purified by semipreparative RP-PLC using a linear gradient from 10% to 90 % of eluent B in eluent A in 30 min. Eluent A: 0.05% 3 P 4 in water; Eluent B: 0.05% 3 P 4 in acetonitrile; UV/VIS-detector was set at 495 nm, corresponding to the maximum of absorption of FITC. The collected yellow fractions were lyophilized and stored at -20 C in the dark. The isolated PA1 oligomer was characterized by MALDI-TF spectrometry on a Bruker Autoflex I instrument using α-cyano-4-hydroxycinnamic acid, 10 mg/ml in acetonitrile-3% aqueous TFA (1:1, v/v) as the matrix. m/z found The isolated PA3 oligomer was characterized by MALDI-TF spectrometry on a Bruker Autoflex I instrument using using α-cyano-4-hydroxycinnamic acid, 10 mg/ml in acetonitrile-3% aqueous TFA (1:1, v/v) as the matrix. m/z found S4
5 2 C 2 C PA 2 S 2 From C to terminus Synthesis of the bis-lysine-pa (PA2) The polystyrene-based MBA resin (50 mg) was swollen in DCM for 30 min and washed four times with DMF. The resin was treated twice with a solution of 20% piperidine (3 ml) in DMF for 10 min, and subsequently washed four times with DMF. Couplings with Fmoc-L-Lysine(Boc)- amino acid were achieved using the following conditions: Fmoc-L-Lysine(Boc)- (4 eq.), BTU (3.6 eq.), Bt (3.6 eq) and DIEA (8 eq.) were dissolved in DMF (2 ml) and the resulting solution was added to the resin and allowed to react for 30 min. The PA tract was synthesized using the same procedure described for PA1,3. The couplings with AEEA linker and FITC were performed using the same procedure described for PA1,3. After each coupling step a capping procedure was performed using acetic anhydride (5%)/lutidine (6%) in DMF solution. In the synthesis the Fmoc group was removed by treatment with 20% piperidine in DMF solution (10 min). Successively, the synthesized Lys-Lys-PA-AEEA-FITC was detached from the resin and precipitated in diethyl ether using the same conditions described for PA1,3. The crude sample was purified by semipreparative RP-PLC using a linear gradient from 10% to 90 % of eluent B in eluent A in 30 min. Eluent A: 0.05% 3 P 4 in water; Eluent B: 0.05% 3 P 4 in acetonitrile. The isolated PA2 was characterized by MALDI-TF spectrometry on a Bruker Autoflex I instrument using using α-cyano-4-hydroxycinnamic acid, 10 mg/ml in acetonitrile-3% aqueous TFA (1:1, v/v) as the matrix. m/z found S5
6 Preparation of mira/pa heteroduplexes (annealing) The mir-509-3p/pas heteroduplexes (1:1.5 or 1:5) were formed by heating the mixture of the samples dissolved in 100 mm KCl, 10 mm K 2 P 4, at 90 C for 5 min and slowly cooling at room temperature for 12 h. UV and UV melting studies The UV spectra were recorded on a Jasco V-530 UV spectrophotometer equipped with a Peltiertype temperature control system (model PTC-348WI). Thermal denaturation experiments were carried out in the temperature range C by monitoring the absorbance at 260 nm at a heating rate of 0.5 C/min. The apparent Tm was estimated from the maximum in the first derivative of the melting profile Abs Wavelength [nm] Fig. S1 UV spectra of mir-509-3p (blue), PA1 (green), mir-509-3p/pa1 (1:1.5) (cyan) and the arithmetical sum of blue and green lines (red). S6
7 2.2 2 Abs Wavelength [nm] Fig. S2 UV spectra of mir-509-3p (blue), PA2 (green), mir-509-3p/pa2 (1:1.5) (cyan) and arithmetic sum of blue and green lines (red) Abs Temperature [C] Fig. S3 UV melting profile of mir-509-3p/pa1 (1:1.5) heteroduplex. S7
8 Abs Temperature [C] Fig. S4 UV melting profile of mir-509-3p/pa2 (1:1.5) heteroduplex. S8
9 CD studies CD spectra were recorded with a Jasco J-715 spectropolarimeter equipped with a Peltier Thermostat Jasco ETC-505T using 0.1 cm path length cuvettes and calibrated with an aqueous solution of 0.06% d-10-(1)- 2 dmol -1 ) was l C C is the oligonucleotide molar concentration, and l is the optical path length of the cell (cm). CD measurements ( nm) were carried out at a scan rate of 100 nm/min with a 2 nm bandwidth. The concentration of the mira/pa1 and mira/pa2 complexes was M. The spectra were signal averaged over at least three scans and baseline corrected by subtracting the buffer spectrum. Fig. S5 CD spectra recorded at 20 C of mir-509-3p/pa1 (solid line) and mir-509-3p/pa2 (dotted line). S9
10 Cell line, Construct and Transfections A549 cell line, human lung carcinoma cells, was purchased from ATCC (Manassas, USA). Cells were maintained in Dulbecco s modified Eagle s medium (Gibco Invitrogen, USA) with 10% heatinactivated fetal bovine serum (yclone, USA) without the addition of antibiotics. Luciferase construct bearing the 3 UTR of CFTR gene (8) was used as mir-509-3p sensitive. Transfection of A549 cells with mira-mimics (Qiagen, Germany, EU) or PA was performed with Attractene Transfection Reagent (Qiagen). Cells were seeded in 96-well plates and were cotransfected 12 hrs after seeding with the firefly reporter constructs described above (20 ng), the Renilla reporter plasmid pcmv-rl (5 ng), and the mir-509-3p mimic. Twelve hours after transfection of cells with constructs and mir-509-3p mix, cells were transfected with PA anti-mir-509-3p. The transfection efficiency (75% for PA1 and 25% for PA2) was assessed by measuring the percentage of fluorescent cells relative to the total numer of cells. Twenty-four hours after transfection, cells were lysed and Firefly and Renilla luciferase activities were determined using the Dual-Glo Luciferase Assay System (Promega Corporation). The relative reporter activity was obtained by normalization to the Renilla luciferase activity. S10
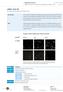
11 Luciferase activity (cps) Electronic Supplementary Material (ESI) for Medicinal Chemistry Communications A B C pcmv-rl 50ng pluc-cftr-3'utr-wt mir-509-3p PA 1 50µM + PA 2 50µM + Fig. S6 Representative uptake of FITC-labeled PA1 (panel A) and PA2 (panel B) by A549 cells and their effect as mira inhibitors (panel C) in the absence of any transfection reagent. S11
12 A B Fig. S7 FITC-labeled PA1 (panel A) and PA2 (panel B) transfected in A549 cells. S12
13 Electrophoretic Mobility Shift Assay The mir-509-3p mimic (2 Me modified) was synthesized by the oligonucleotide synthesis facility at CEIGE-Biotecnologie Avanzate (aples Italy). The mira and PA1 or PA2 were annealed in 1X EBuffer 2 (50 mm acl, 10 mm Tris-Cl, 10 mm MgCl2, 1 mm DTT, p7.9 at 25 C) for 2 hrs at room temperature. All the reactions were loaded into 20% polyacrylamide gels in 0.5X Tris-Borate-EDTA (TBE) buffer and run at 140 V for 3 hrs. The fluorescence signal was acquired placing the wet gel directly on the platen of Typhoon 8600 scanner. After fluorescence acquisition the gel was EtBr stained, destained in distilled water and the image acquired with a Gel Doc 2000 (Bio-Rad). PA2 mir-509-3p+pa2 (1:5) mir-509-3p+pa2 (1:1.5) PA1 mir-509-3p mir-509-3p+pa1 (1:5) mir-509-3p+pa1 (1:1.5 Fig. S8 Ethidium Bromide staining of the EMSA. The green arrows show the position of mir-509-3p migration; the blue arrow shows the position of the mir-509-3p/pa1 heteroduplex in a 1:5 ratio; the yellow arrow shows the position of the mir-509-3p/pa2 heteroduplex in a 1:5 ratio. S13
Experimental procedures. Solid phase peptide synthesis (SPPS)
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry This journal is The Royal Society of Chemistry 214 Experimental procedures Solid phase peptide synthesis (SPPS) Solid phase
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry This journal is The Royal Society of Chemistry 214 Experimental procedures Solid phase peptide synthesis (SPPS) Solid phase
Small μmol Scale Synthesis of a Labeled Antimicrobial Peptide using Biotage
 Application ote A098 Small μmol Scale Synthesis of a Labeled Antimicrobial Peptide Page 1 Small μmol Scale Synthesis of a Labeled Antimicrobial Peptide using Biotage Initiator+ Alstra Introduction Labeled
Application ote A098 Small μmol Scale Synthesis of a Labeled Antimicrobial Peptide Page 1 Small μmol Scale Synthesis of a Labeled Antimicrobial Peptide using Biotage Initiator+ Alstra Introduction Labeled
1) Technical informations. - a) How does it work? - b) Purification - c) Quality Control. 2) Standard synthesis
 1) Technical informations - a) How does it work? - b) Purification - c) Quality Control 2) Standard synthesis - a) Standard peptides - b) Modified peptides - c) Shipment and Delivery Time - d) How to order?
1) Technical informations - a) How does it work? - b) Purification - c) Quality Control 2) Standard synthesis - a) Standard peptides - b) Modified peptides - c) Shipment and Delivery Time - d) How to order?
Solid-phase Synthesis of Homodimeric Peptides: Preparation of Covalently-linked Dimers of Amyloid-beta Peptide
 Electronic Supplementary Information Solid-phase Synthesis of Homodimeric Peptides: Preparation of Covalently-linked Dimers of Amyloid-beta Peptide W. Mei Kok, a,b,c Denis B. Scanlon, b John A. Karas,
Electronic Supplementary Information Solid-phase Synthesis of Homodimeric Peptides: Preparation of Covalently-linked Dimers of Amyloid-beta Peptide W. Mei Kok, a,b,c Denis B. Scanlon, b John A. Karas,
Novel Method for Solid Phase Peptide Synthesis Using Microwave Energy
 Novel Method for Solid Phase Peptide Synthesis Using Microwave Energy Jonathan M. Collins, Michael J. Collins, Rebecca C. Steorts CEM Corporation, Matthews, NC 28106-0200, U.S.A. Presented at American
Novel Method for Solid Phase Peptide Synthesis Using Microwave Energy Jonathan M. Collins, Michael J. Collins, Rebecca C. Steorts CEM Corporation, Matthews, NC 28106-0200, U.S.A. Presented at American
Supporting Information. The G-triplex DNA could function as a new variety of DNA peroxidase
 Supporting Information The G-triplex DNA could function as a new variety of DNA peroxidase Shaoru Wang +, Boshi Fu +, Shuang Peng, Xiaoe Zhang, Tian Tian*, Xiang Zhou* College of Chemistry and Molecular
Supporting Information The G-triplex DNA could function as a new variety of DNA peroxidase Shaoru Wang +, Boshi Fu +, Shuang Peng, Xiaoe Zhang, Tian Tian*, Xiang Zhou* College of Chemistry and Molecular
Peptide Library Synthesis
 Peptide Library Synthesis Jamie M. R. Moore Guy Laboratory UCSF I. verview.. page 2 II. Reagents and Apparatus. page 4 III. Flow Chart. page 6 IV. Protocol. page 7 IV. Tables A. List of Fmoc Amino. page
Peptide Library Synthesis Jamie M. R. Moore Guy Laboratory UCSF I. verview.. page 2 II. Reagents and Apparatus. page 4 III. Flow Chart. page 6 IV. Protocol. page 7 IV. Tables A. List of Fmoc Amino. page
Supplemental data. A simple and effective cleavable linker for chemical proteomics applications
 Supplemental data A simple and effective cleavable linker for chemical proteomics applications Yinliang Yang, annes ahne, Bernhard Kuster, and Steven. L. Verhelst * Figure S1 Figure S2 Figure S3 Table
Supplemental data A simple and effective cleavable linker for chemical proteomics applications Yinliang Yang, annes ahne, Bernhard Kuster, and Steven. L. Verhelst * Figure S1 Figure S2 Figure S3 Table
Supporting Information
 Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2013 More than Meets the Eye: Conformational Switching of a Stacked Dialkoxynaphthalene Naphthalenetetracarboxylic diimide
Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2013 More than Meets the Eye: Conformational Switching of a Stacked Dialkoxynaphthalene Naphthalenetetracarboxylic diimide
Peptide Synthesis Zheng Miao* and Zhen Cheng
 Peptide Synthesis Zheng Miao* and Zhen Cheng 1 Department of Radiology, Molecular Imaging Program at Stanford, Stanford University School of Medicine, Stanford, USA *For correspondence: zmiao@stanford.edu
Peptide Synthesis Zheng Miao* and Zhen Cheng 1 Department of Radiology, Molecular Imaging Program at Stanford, Stanford University School of Medicine, Stanford, USA *For correspondence: zmiao@stanford.edu
Peptide synthesis, radiolabelling and radiochemical analysis
 SUPPLEMENTAL DATA MATERIALS AND METHODS Peptide synthesis, radiolabelling and radiochemical analysis Solid phase synthesis of peptides was carried out on using ABI 433A peptide synthesizer, on a preloaded
SUPPLEMENTAL DATA MATERIALS AND METHODS Peptide synthesis, radiolabelling and radiochemical analysis Solid phase synthesis of peptides was carried out on using ABI 433A peptide synthesizer, on a preloaded
DNA Assembly and Enzymatic Cutting in Solutions: A Gold Nanoparticle Based SERS Detection Strategy
 Supporting Information DNA Assembly and Enzymatic Cutting in Solutions: A Gold Nanoparticle Based SERS Detection Strategy Elizabeth Crew 1, Hong Yan 1, Liqin Lin 1, Jun Yin 1, Zakiya Skeete 1, Timur Kotlyar
Supporting Information DNA Assembly and Enzymatic Cutting in Solutions: A Gold Nanoparticle Based SERS Detection Strategy Elizabeth Crew 1, Hong Yan 1, Liqin Lin 1, Jun Yin 1, Zakiya Skeete 1, Timur Kotlyar
TECHNICAL BULLETIN. FluoroTag FITC Conjugation Kit. Product Number FITC1 Storage Temperature 2 8 C
 FluoroTag FITC Conjugation Kit Product Number FITC1 Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description The FluoroTag FITC Conjugation Kit is suitable for the conjugation of polyclonal and
FluoroTag FITC Conjugation Kit Product Number FITC1 Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description The FluoroTag FITC Conjugation Kit is suitable for the conjugation of polyclonal and
LUMEFANTRINE Draft proposal for The International Pharmacopoeia (October 2006)
 October 2006 RESTRICTED LUMEFANTRINE Draft proposal for The International Pharmacopoeia (October 2006) DRAFT FOR DISCUSSION World Health Organization 2006 All rights reserved. This draft is intended for
October 2006 RESTRICTED LUMEFANTRINE Draft proposal for The International Pharmacopoeia (October 2006) DRAFT FOR DISCUSSION World Health Organization 2006 All rights reserved. This draft is intended for
Standard practices for Fmoc-based solid-phase. peptide synthesis in the Nowick laboratory. (Version 1.6.1)
 Standard practices for Fmoc-based solid-phase peptide synthesis in the Nowick laboratory (Version 1.6.1) Adam G. Kreutzer and Patrick J. Salveson E-mail: Contents Contributions to this guide 3 General
Standard practices for Fmoc-based solid-phase peptide synthesis in the Nowick laboratory (Version 1.6.1) Adam G. Kreutzer and Patrick J. Salveson E-mail: Contents Contributions to this guide 3 General
Structure-Based Design of Covalent Siah Inhibitors
 Chemistry & Biology, Volume 20 Supplemental Information Structure-Based Design of Covalent Siah Inhibitors John L. Stebbins, Eugenio Santelli, Yongmei Feng, Surya K. De, Angela Purves, Khatereh Motamedchaboki,
Chemistry & Biology, Volume 20 Supplemental Information Structure-Based Design of Covalent Siah Inhibitors John L. Stebbins, Eugenio Santelli, Yongmei Feng, Surya K. De, Angela Purves, Khatereh Motamedchaboki,
Overview'of'Solid-Phase'Peptide'Synthesis'(SPPS)'and'Secondary'Structure'Determination'by'FTIR'
 verviewofsolid-phasepeptidesynthesis(spps)andsecondarystructuredeterminationbyftir Introduction Proteinsareubiquitousinlivingorganismsandcells,andcanserveavarietyoffunctions.Proteinscanactas enzymes,hormones,antibiotics,receptors,orserveasstructuralsupportsintissuessuchasmuscle,hair,and
verviewofsolid-phasepeptidesynthesis(spps)andsecondarystructuredeterminationbyftir Introduction Proteinsareubiquitousinlivingorganismsandcells,andcanserveavarietyoffunctions.Proteinscanactas enzymes,hormones,antibiotics,receptors,orserveasstructuralsupportsintissuessuchasmuscle,hair,and
Supplemental Material: peptide synthesis
 Supplemental Material: peptide synthesis Synthesis of MMAE containing ACPPs (Scheme 1) The peptides were synthesized by using regular solid phase Fmoc peptide synthesis. Peptides that were used for fluorescence
Supplemental Material: peptide synthesis Synthesis of MMAE containing ACPPs (Scheme 1) The peptides were synthesized by using regular solid phase Fmoc peptide synthesis. Peptides that were used for fluorescence
Biological Stability and Activity of sirna in Ionic Liquids**
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supporting Information Biological Stability and Activity of sirna in Ionic Liquids** Romiza R.
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supporting Information Biological Stability and Activity of sirna in Ionic Liquids** Romiza R.
Synthesis of Leucine Zippers and Leucine Zipper Dendrimers
 Supplementary Materials Helical Supramolecules and Fibers Utilizing Leucine-Zipper Displaying Dendrimers Min Zhou, David Bentley, Indraneel Ghosh Contribution from the Department of Chemistry, University
Supplementary Materials Helical Supramolecules and Fibers Utilizing Leucine-Zipper Displaying Dendrimers Min Zhou, David Bentley, Indraneel Ghosh Contribution from the Department of Chemistry, University
FAST AND EFFICIENT PURIFICATION OF SYNTHETIC PEPTIDES BY SOLID-PHASE EXTRACTION
 ACTA CHROMATOGRAPHICA, NO. 14, 2004 FAST AND EFFICIENT PURIFICATION OF SYNTHETIC PEPTIDES BY SOLID-PHASE EXTRACTION W. Kamysz 1,*, M. Okrój 2, E. Łempicka 3, T. Ossowski 3, and J. Łukasiak 1 1 Faculty
ACTA CHROMATOGRAPHICA, NO. 14, 2004 FAST AND EFFICIENT PURIFICATION OF SYNTHETIC PEPTIDES BY SOLID-PHASE EXTRACTION W. Kamysz 1,*, M. Okrój 2, E. Łempicka 3, T. Ossowski 3, and J. Łukasiak 1 1 Faculty
Lab #11: Determination of a Chemical Equilibrium Constant
 Lab #11: Determination of a Chemical Equilibrium Constant Objectives: 1. Determine the equilibrium constant of the formation of the thiocyanatoiron (III) ions. 2. Understand the application of using a
Lab #11: Determination of a Chemical Equilibrium Constant Objectives: 1. Determine the equilibrium constant of the formation of the thiocyanatoiron (III) ions. 2. Understand the application of using a
Real-time monitoring of rolling circle amplification using aggregation-induced emission: applications for biological detection
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 215 Supplementary Information Real-time monitoring of rolling circle amplification using aggregation-induced
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 215 Supplementary Information Real-time monitoring of rolling circle amplification using aggregation-induced
Labelling of peptides with CyDye fluors for fluorescent applications on the LEADseeker Homogeneous Imaging System
 Issue No. 7 abelling of peptides with CyDye fluors for fluorescent applications on the EADseeker Homogeneous Imaging System EADseeker TM homogeneous imaging system is a digital imager comprising a chargecoupled
Issue No. 7 abelling of peptides with CyDye fluors for fluorescent applications on the EADseeker Homogeneous Imaging System EADseeker TM homogeneous imaging system is a digital imager comprising a chargecoupled
Supporting Information
 Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2014 A General Strategy for Site-Directed Enzyme Immobilization by Using NiO Nanoparticle Decorated Mesoporous Silica
Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2014 A General Strategy for Site-Directed Enzyme Immobilization by Using NiO Nanoparticle Decorated Mesoporous Silica
lung cancer targeted photodynamic therapy and imaging
 99m Tc-Hematoporphyrin linked albumin nanoparticles for lung cancer targeted photodynamic therapy and imaging Su-Geun Yang, Ji-Eun Chang, Byungchul Shin, Sanghyun Park, Kun Na and Chang-Koo Shim* *Corresponding
99m Tc-Hematoporphyrin linked albumin nanoparticles for lung cancer targeted photodynamic therapy and imaging Su-Geun Yang, Ji-Eun Chang, Byungchul Shin, Sanghyun Park, Kun Na and Chang-Koo Shim* *Corresponding
Structural basis for the enhanced activity of cyclic antimicrobial peptides: the case of BPC194
 SUPPLEMENTARY DATA Structural basis for the enhanced activity of cyclic antimicrobial peptides: the case of BPC194 Jacek T. Mika,a, Gemma Moiset,a, Anna D. Cirac a,b, Lidia Feliu c, Eduard Bardají c, Marta
SUPPLEMENTARY DATA Structural basis for the enhanced activity of cyclic antimicrobial peptides: the case of BPC194 Jacek T. Mika,a, Gemma Moiset,a, Anna D. Cirac a,b, Lidia Feliu c, Eduard Bardají c, Marta
Spatial Screening of Cyclic Neoglycopeptides: Identification of Multivalent Wheat Germ Agglutinin Ligands**
 1 Spatial Screening of Cyclic Neoglycopeptides: Identification of Multivalent Wheat Germ Agglutinin Ligands** Valentin Wittmann* and Sonja Seeberger Experimental Section General. Solid-phase peptide synthesis
1 Spatial Screening of Cyclic Neoglycopeptides: Identification of Multivalent Wheat Germ Agglutinin Ligands** Valentin Wittmann* and Sonja Seeberger Experimental Section General. Solid-phase peptide synthesis
Lab 10: Bacterial Transformation, part 2, DNA plasmid preps, Determining DNA Concentration and Purity
 Lab 10: Bacterial Transformation, part 2, DNA plasmid preps, Determining DNA Concentration and Purity Today you analyze the results of your bacterial transformation from last week and determine the efficiency
Lab 10: Bacterial Transformation, part 2, DNA plasmid preps, Determining DNA Concentration and Purity Today you analyze the results of your bacterial transformation from last week and determine the efficiency
Guide to Reverse Phase SpinColumns Chromatography for Sample Prep
 Guide to Reverse Phase SpinColumns Chromatography for Sample Prep www.harvardapparatus.com Contents Introduction...2-3 Modes of Separation...4-6 Spin Column Efficiency...7-8 Fast Protein Analysis...9 Specifications...10
Guide to Reverse Phase SpinColumns Chromatography for Sample Prep www.harvardapparatus.com Contents Introduction...2-3 Modes of Separation...4-6 Spin Column Efficiency...7-8 Fast Protein Analysis...9 Specifications...10
Supplementary Information for
 Electronic Supplementary Material (ESI) for Polymer Chemistry. This journal is The Royal Society of Chemistry 2015 Supplementary Information for Doubly Responsive Polymersomes towards Monosaccharides and
Electronic Supplementary Material (ESI) for Polymer Chemistry. This journal is The Royal Society of Chemistry 2015 Supplementary Information for Doubly Responsive Polymersomes towards Monosaccharides and
Peptide Nucleic Acid Microarrays
 PNA Microarrays 283 21 Peptide Nucleic Acid Microarrays Anette Jacob, Ole Brandt, Achim Stephan, and Jörg D. Hoheisel Summary A fast and economical procedure for the production of peptide nucleic acid
PNA Microarrays 283 21 Peptide Nucleic Acid Microarrays Anette Jacob, Ole Brandt, Achim Stephan, and Jörg D. Hoheisel Summary A fast and economical procedure for the production of peptide nucleic acid
Supporting information. Cyclic peptide-polymer conjugates: grafting to VS grafting from
 Supporting information Cyclic peptide-polymer conjugates: grafting to VS grafting from Sophie C. Larnaudie, a Johannes C. Brendel, a,c Katrina A. Jolliffe* b and Sébastien Perrier* a,c a Department of
Supporting information Cyclic peptide-polymer conjugates: grafting to VS grafting from Sophie C. Larnaudie, a Johannes C. Brendel, a,c Katrina A. Jolliffe* b and Sébastien Perrier* a,c a Department of
Plant Genomic DNA Extraction using CTAB
 Plant Genomic DNA Extraction using CTAB Introduction The search for a more efficient means of extracting DNA of both higher quality and yield has lead to the development of a variety of protocols, however
Plant Genomic DNA Extraction using CTAB Introduction The search for a more efficient means of extracting DNA of both higher quality and yield has lead to the development of a variety of protocols, however
Supporting Information. Minimum active structure of insulin-like. peptide 5 (INSL5)
 Supporting Information Minimum active structure of insulin-like peptide 5 (INSL5) Alessia Belgi 1,2, Ross A.D. Bathgate *1,2,3, Martina Kocan *4, Nitin Patil 1,5, Suode Zhang 1, Geoffrey W. Tregear 1,2,
Supporting Information Minimum active structure of insulin-like peptide 5 (INSL5) Alessia Belgi 1,2, Ross A.D. Bathgate *1,2,3, Martina Kocan *4, Nitin Patil 1,5, Suode Zhang 1, Geoffrey W. Tregear 1,2,
An In-Gel Digestion Protocol
 An In-Gel Digestion Protocol This protocol describes the digestion of a protein present in an SDS-PAGE gel band with trypsin. The band can be taken from either a 1D or 2D electrophoresis gel. Reagents
An In-Gel Digestion Protocol This protocol describes the digestion of a protein present in an SDS-PAGE gel band with trypsin. The band can be taken from either a 1D or 2D electrophoresis gel. Reagents
Nucleic Acid Purity Assessment using A 260 /A 280 Ratios
 Nucleic Acid Purity Assessment using A 260 /A 280 Ratios A common practice in molecular biology is to perform a quick assessment of the purity of nucleic acid samples by determining the ratio of spectrophotometric
Nucleic Acid Purity Assessment using A 260 /A 280 Ratios A common practice in molecular biology is to perform a quick assessment of the purity of nucleic acid samples by determining the ratio of spectrophotometric
A prochelator with a modular masking group featuring hydrogen peroxide activation with concurrent fluorescent reporting
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supporting Information A prochelator with a modular masking group featuring hydrogen peroxide activation
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supporting Information A prochelator with a modular masking group featuring hydrogen peroxide activation
Chem 405 Biochemistry Lab I Experiment 2 Quantitation of an unknown protein solution.
 Chem 405 Biochemistry Lab I Experiment 2 Quantitation of an unknown protein solution. Introduction: The determination of protein concentration is frequently required in biochemical work. Several methods
Chem 405 Biochemistry Lab I Experiment 2 Quantitation of an unknown protein solution. Introduction: The determination of protein concentration is frequently required in biochemical work. Several methods
TransformAid Bacterial Transformation Kit
 Home Contacts Order Catalog Support Search Alphabetical Index Numerical Index Restriction Endonucleases Modifying Enzymes PCR Kits Markers Nucleic Acids Nucleotides & Oligonucleotides Media Transfection
Home Contacts Order Catalog Support Search Alphabetical Index Numerical Index Restriction Endonucleases Modifying Enzymes PCR Kits Markers Nucleic Acids Nucleotides & Oligonucleotides Media Transfection
Covalent Conjugation to Cytodiagnostics Carboxylated Gold Nanoparticles Tech Note #105
 Covalent Conjugation to Cytodiagnostics Carboxylated Gold Nanoparticles Tech Note #105 Background Gold nanoparticle conjugates have been widely used in biological research and biosensing applications.
Covalent Conjugation to Cytodiagnostics Carboxylated Gold Nanoparticles Tech Note #105 Background Gold nanoparticle conjugates have been widely used in biological research and biosensing applications.
TECHNICAL BULLETIN. HIS-Select Nickel Affinity Gel. Catalog Number P6611 Storage Temperature 2 8 C
 HIS-Select Nickel Affinity Gel Catalog Number P6611 Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description HIS-Select Nickel Affinity Gel is an immobilized metalion affinity chromatography (IMAC)
HIS-Select Nickel Affinity Gel Catalog Number P6611 Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description HIS-Select Nickel Affinity Gel is an immobilized metalion affinity chromatography (IMAC)
LC-MS/MS Method for the Determination of Docetaxel in Human Serum for Clinical Research
 LC-MS/MS Method for the Determination of Docetaxel in Human Serum for Clinical Research J. Jones, J. Denbigh, Thermo Fisher Scientific, Runcorn, Cheshire, UK Application Note 20581 Key Words SPE, SOLA,
LC-MS/MS Method for the Determination of Docetaxel in Human Serum for Clinical Research J. Jones, J. Denbigh, Thermo Fisher Scientific, Runcorn, Cheshire, UK Application Note 20581 Key Words SPE, SOLA,
How To Make A Nucleic Acid
 Protocol Received: 31 May 2010 Revised: 3 August 2010 Accepted: 26 August 2010 Published online in Wiley nline Library: 26 ctober 2010 (wileyonlinelibrary.com) DI 10.1002/psc.1305 Facile synthesis of peptide
Protocol Received: 31 May 2010 Revised: 3 August 2010 Accepted: 26 August 2010 Published online in Wiley nline Library: 26 ctober 2010 (wileyonlinelibrary.com) DI 10.1002/psc.1305 Facile synthesis of peptide
HiPer Ion Exchange Chromatography Teaching Kit
 HiPer Ion Exchange Chromatography Teaching Kit Product Code: HTC001 Number of experiments that can be performed: 5 Duration of Experiment: Protocol: 5-6 hours Storage Instructions: The kit is stable for
HiPer Ion Exchange Chromatography Teaching Kit Product Code: HTC001 Number of experiments that can be performed: 5 Duration of Experiment: Protocol: 5-6 hours Storage Instructions: The kit is stable for
Troubleshooting Guide for DNA Electrophoresis
 Troubleshooting Guide for Electrophoresis. ELECTROPHORESIS Protocols and Recommendations for Electrophoresis electrophoresis problem 1 Low intensity of all or some bands 2 Smeared bands 3 Atypical banding
Troubleshooting Guide for Electrophoresis. ELECTROPHORESIS Protocols and Recommendations for Electrophoresis electrophoresis problem 1 Low intensity of all or some bands 2 Smeared bands 3 Atypical banding
PROTOCOL. Immunocytochemistry (ICC) MATERIALS AND EQUIPMENT REQUIRED
 PROTOCOL Immunocytochemistry (ICC) 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 11-07 MATERIALS AND EQUIPMENT REQUIRED Materials: MitoSciences primary monoclonal antibody/antibodies Fluorophore-conjugated
PROTOCOL Immunocytochemistry (ICC) 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 11-07 MATERIALS AND EQUIPMENT REQUIRED Materials: MitoSciences primary monoclonal antibody/antibodies Fluorophore-conjugated
Classic Immunoprecipitation
 292PR 01 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Classic Immunoprecipitation Utilizes Protein A/G Agarose for Antibody Binding (Cat.
292PR 01 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Classic Immunoprecipitation Utilizes Protein A/G Agarose for Antibody Binding (Cat.
Monoclonal Antibody Fragment Separation and Characterization Using Size Exclusion Chromatography Coupled with Mass Spectrometry
 Monoclonal ntibody Fragment Separation and haracterization Using Size Exclusion hromatography oupled with Mass Spectrometry uthors Haiying hen Katherine McLaughlin Sepax Technologies, Inc. 5 Innovation
Monoclonal ntibody Fragment Separation and haracterization Using Size Exclusion hromatography oupled with Mass Spectrometry uthors Haiying hen Katherine McLaughlin Sepax Technologies, Inc. 5 Innovation
How To Make A Molecular Encoder And Decoder
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supporting information Molecular Encoder - Decoder Based on Assembly of Graphene with Dye-labeled
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supporting information Molecular Encoder - Decoder Based on Assembly of Graphene with Dye-labeled
Supplemental Information. Converting a Staphylococcus aureus Toxin. into Effective Cyclic Pseudopeptide Antibiotics
 Chemistry & Biology, Volume 22 Supplemental Information Converting a Staphylococcus aureus Toxin into Effective Cyclic Pseudopeptide Antibiotics Olivia Solecki, Amor Mosbah, Michèle Baudy Floc'h, and Brice
Chemistry & Biology, Volume 22 Supplemental Information Converting a Staphylococcus aureus Toxin into Effective Cyclic Pseudopeptide Antibiotics Olivia Solecki, Amor Mosbah, Michèle Baudy Floc'h, and Brice
Materials and Methods. Protocol for Fmoc- based solid- phase peptide synthesis
 Materials and Methods All commercially available materials (Aldrich, TCI, ovabiochem, Fluka ) were used without further purification. All solvents were reagent grade or PLC grade (Fisher ). Anhydrous TF,
Materials and Methods All commercially available materials (Aldrich, TCI, ovabiochem, Fluka ) were used without further purification. All solvents were reagent grade or PLC grade (Fisher ). Anhydrous TF,
Peptide Antibody Production
 Peptide Antibody Production A) Peptide BioSynthesis (http://www.biosyn.com, 800-227-0627) B) Conjugation of peptide to KLH (Imject Maleimide Activated KLH, PIERCE=Thermo #77605, 10 mg) C) Peptide affinity
Peptide Antibody Production A) Peptide BioSynthesis (http://www.biosyn.com, 800-227-0627) B) Conjugation of peptide to KLH (Imject Maleimide Activated KLH, PIERCE=Thermo #77605, 10 mg) C) Peptide affinity
6 Characterization of Casein and Bovine Serum Albumin
 6 Characterization of Casein and Bovine Serum Albumin (BSA) Objectives: A) To separate a mixture of casein and bovine serum albumin B) to characterize these proteins based on their solubilities as a function
6 Characterization of Casein and Bovine Serum Albumin (BSA) Objectives: A) To separate a mixture of casein and bovine serum albumin B) to characterize these proteins based on their solubilities as a function
How To Use An Acquity Qda Detector
 Mass-Directed Isolation of a Synthetic Peptide Using the ACQUITY QDa Detector Jo-Ann M. Jablonski and Andrew J. Aubin Waters Corporation, Milford, MA, USA APPLICATION BENEFITS The ACQUITY QDa Detector
Mass-Directed Isolation of a Synthetic Peptide Using the ACQUITY QDa Detector Jo-Ann M. Jablonski and Andrew J. Aubin Waters Corporation, Milford, MA, USA APPLICATION BENEFITS The ACQUITY QDa Detector
Supplementary Information. Primary amino acid lithium salt as a catalyst for asymmetric Michael addition of isobutyraldehyde with β-nitroalkenes.
 This journal is (c) The Royal Society of Chemistry 8 Supplementary Information Primary amino acid lithium salt as a catalyst for asymmetric Michael addition of isobutyraldehyde with β-nitroalkenes. Atsushi
This journal is (c) The Royal Society of Chemistry 8 Supplementary Information Primary amino acid lithium salt as a catalyst for asymmetric Michael addition of isobutyraldehyde with β-nitroalkenes. Atsushi
Guidance for Industry
 Guidance for Industry for the Submission of Chemistry, Manufacturing, and Controls Information for Synthetic Peptide Substances Center for Drug Evaluation and Research (CDER) Center for Biologics Evaluation
Guidance for Industry for the Submission of Chemistry, Manufacturing, and Controls Information for Synthetic Peptide Substances Center for Drug Evaluation and Research (CDER) Center for Biologics Evaluation
CypExpress 3A4 Catalyzed Conversion of Testosterone (TE) to 6β- Hydroxytestosterone (HT)
 TM CASE STUDY CypExpress 3A4 Catalyzed Conversion of to Shuvendu Das, 1 Enrique Martez, 2 and Mani Subramanian 1 1 Center for Biocatalysis and Bioprocessg, University of Iowa 2 Oxford Biomedical Research,
TM CASE STUDY CypExpress 3A4 Catalyzed Conversion of to Shuvendu Das, 1 Enrique Martez, 2 and Mani Subramanian 1 1 Center for Biocatalysis and Bioprocessg, University of Iowa 2 Oxford Biomedical Research,
Mouse ES Cell Nucleofector Kit
 page 1 of 7 Mouse ES Cell Nucleofector Kit for Mouse Embryonic Stem Cells Cell type Origin Cells derived from mouse blastocysts. Morphology Round cells growing in clumps. Important remarks! 1. This protocol
page 1 of 7 Mouse ES Cell Nucleofector Kit for Mouse Embryonic Stem Cells Cell type Origin Cells derived from mouse blastocysts. Morphology Round cells growing in clumps. Important remarks! 1. This protocol
The cell lines used in this study were obtained from the American Type Culture
 Supplementary materials and methods Cell culture and drug treatments The cell lines used in this study were obtained from the American Type Culture Collection (ATCC) and grown as previously described.
Supplementary materials and methods Cell culture and drug treatments The cell lines used in this study were obtained from the American Type Culture Collection (ATCC) and grown as previously described.
Creatine Kinase Activity Colorimetric Assay Kit ABE5487 100 assays; Store at -20 C
 Creatine Kinase Activity Colorimetric Assay Kit ABE5487 100 assays; Store at -20 C I. Introduction: Creatine Kinase (CK) also known as creatine phosphokinase (CPK) and ATP: creatine N- phosphotransferase
Creatine Kinase Activity Colorimetric Assay Kit ABE5487 100 assays; Store at -20 C I. Introduction: Creatine Kinase (CK) also known as creatine phosphokinase (CPK) and ATP: creatine N- phosphotransferase
Studying Photosynthetic Electron Transport through the Hill Reaction
 Studying Photosynthetic Electron Transport through the Hill Reaction In this lab exercise you will learn techniques for studying electron transport. While our research system will be chloroplasts and photosynthetic
Studying Photosynthetic Electron Transport through the Hill Reaction In this lab exercise you will learn techniques for studying electron transport. While our research system will be chloroplasts and photosynthetic
Human serum albumin (HSA) nanoparticles stabilized with. intermolecular disulfide bonds. Supporting Information
 Human serum albumin (HSA) nanoparticles stabilized with intermolecular disulfide bonds Wentan Wang, Yanbin Huang*, Shufang Zhao, Ting Shao and Yi Cheng* Department of Chemical Engineering, Tsinghua University,
Human serum albumin (HSA) nanoparticles stabilized with intermolecular disulfide bonds Wentan Wang, Yanbin Huang*, Shufang Zhao, Ting Shao and Yi Cheng* Department of Chemical Engineering, Tsinghua University,
Optimized Protocol sirna Test Kit for Cell Lines and Adherent Primary Cells
 page 1 of 8 sirna Test Kit for Cell Lines and Adherent Primary Cells Kit principle Co-transfection of pmaxgfp, encoding the green fluorescent protein (GFP) from Pontellina p. with an sirna directed against
page 1 of 8 sirna Test Kit for Cell Lines and Adherent Primary Cells Kit principle Co-transfection of pmaxgfp, encoding the green fluorescent protein (GFP) from Pontellina p. with an sirna directed against
In vitro analysis of pri-mirna processing. by Drosha-DGCR8 complex. (Narry Kim s lab)
 In vitro analysis of pri-mirna processing by Drosha-DGCR8 complex (Narry Kim s lab) 1-1. Preparation of radiolabeled pri-mirna transcript The RNA substrate for a cropping reaction can be prepared by in
In vitro analysis of pri-mirna processing by Drosha-DGCR8 complex (Narry Kim s lab) 1-1. Preparation of radiolabeled pri-mirna transcript The RNA substrate for a cropping reaction can be prepared by in
CLEAN-UP PROCESS FOR MASS SPECTRAL STUDY OF AMPHETAMINES IN PUTREFIED BODY MATERIALS
 CLEAN-UP PROCESS FOR MASS SPECTRAL STUDY OF AMPHETAMINES IN PUTREFIED BODY MATERIALS Kenji HARA, Seiichi KASHIMURA, Masayuki KASHIWAGI, Tomoko HAMANAKA, Mitsuyoshi KAGEURA Department of Forensic Medicine,
CLEAN-UP PROCESS FOR MASS SPECTRAL STUDY OF AMPHETAMINES IN PUTREFIED BODY MATERIALS Kenji HARA, Seiichi KASHIMURA, Masayuki KASHIWAGI, Tomoko HAMANAKA, Mitsuyoshi KAGEURA Department of Forensic Medicine,
Supplementary Figure 1.
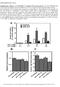 Supplementary Figure 1. (A) MicroRNA 212 enhances IS from pancreatic β-cells. INS-1 832/3 β-cells were transfected with precursors for mirnas 212, 375, or negative control oligonucleotides. 48 hrs after
Supplementary Figure 1. (A) MicroRNA 212 enhances IS from pancreatic β-cells. INS-1 832/3 β-cells were transfected with precursors for mirnas 212, 375, or negative control oligonucleotides. 48 hrs after
Technical Note. Roche Applied Science. No. LC 19/2004. Color Compensation
 Roche Applied Science Technical Note No. LC 19/2004 Purpose of this Note Color The LightCycler System is able to simultaneously detect and analyze more than one color in each capillary. Due to overlap
Roche Applied Science Technical Note No. LC 19/2004 Purpose of this Note Color The LightCycler System is able to simultaneously detect and analyze more than one color in each capillary. Due to overlap
EZ-Run Protein Gel Solution. EZ-Run Protein Standards. EZ-Run Gel Staining Solution. Traditional SDS-Page Reagents. Protein Electrophoresis
 EZ-Run Protein Gel Solution EZ-Run Protein Standards EZ-Run Gel Staining Solution Traditional SDS-Page Reagents Protein Electrophoresis protein electrophoresis Introduction Sodium dodecyl sulfate polyacrylamide
EZ-Run Protein Gel Solution EZ-Run Protein Standards EZ-Run Gel Staining Solution Traditional SDS-Page Reagents Protein Electrophoresis protein electrophoresis Introduction Sodium dodecyl sulfate polyacrylamide
Peptides: Synthesis and Biological Interest
 Peptides: Synthesis and Biological Interest Therapeutic Agents Therapeutic peptides approved by the FDA (2009-2011) 3 Proteins Biopolymers of α-amino acids. Amino acids are joined by peptide bond. They
Peptides: Synthesis and Biological Interest Therapeutic Agents Therapeutic peptides approved by the FDA (2009-2011) 3 Proteins Biopolymers of α-amino acids. Amino acids are joined by peptide bond. They
HighPure Maxi Plasmid Kit
 HighPure Maxi Plasmid Kit For purification of high pure plasmid DNA with high yields www.tiangen.com PP120109 HighPure Maxi Plasmid Kit Kit Contents Storage Cat.no. DP116 Contents RNaseA (100 mg/ml) Buffer
HighPure Maxi Plasmid Kit For purification of high pure plasmid DNA with high yields www.tiangen.com PP120109 HighPure Maxi Plasmid Kit Kit Contents Storage Cat.no. DP116 Contents RNaseA (100 mg/ml) Buffer
Fluorescent dyes for use with the
 Detection of Multiple Reporter Dyes in Real-time, On-line PCR Analysis with the LightCycler System Gregor Sagner, Cornelia Goldstein, and Rob van Miltenburg Roche Molecular Biochemicals, Penzberg, Germany
Detection of Multiple Reporter Dyes in Real-time, On-line PCR Analysis with the LightCycler System Gregor Sagner, Cornelia Goldstein, and Rob van Miltenburg Roche Molecular Biochemicals, Penzberg, Germany
EXPERIMENT 11 UV/VIS Spectroscopy and Spectrophotometry: Spectrophotometric Analysis of Potassium Permanganate Solutions.
 EXPERIMENT 11 UV/VIS Spectroscopy and Spectrophotometry: Spectrophotometric Analysis of Potassium Permanganate Solutions. Outcomes After completing this experiment, the student should be able to: 1. Prepare
EXPERIMENT 11 UV/VIS Spectroscopy and Spectrophotometry: Spectrophotometric Analysis of Potassium Permanganate Solutions. Outcomes After completing this experiment, the student should be able to: 1. Prepare
EXPERIMENT 5. Molecular Absorption Spectroscopy: Determination of Iron With 1,10-Phenanthroline
 EXPERIMENT 5 Molecular Absorption Spectroscopy: Determination of Iron With 1,10-Phenanthroline UNKNOWN Submit a clean, labeled 100-mL volumetric flask to the instructor so that your unknown iron solution
EXPERIMENT 5 Molecular Absorption Spectroscopy: Determination of Iron With 1,10-Phenanthroline UNKNOWN Submit a clean, labeled 100-mL volumetric flask to the instructor so that your unknown iron solution
Tamsulosin Hydrochloride Capsules
 . nal Revision Bulletin Official October 1, 2011 Tamsulosin 1 standard solution, and shake well. Centrifuge at 1500 rpm for 10 min, and use the supernatant, passing it if Tamsulosin Hydrochloride Capsules
. nal Revision Bulletin Official October 1, 2011 Tamsulosin 1 standard solution, and shake well. Centrifuge at 1500 rpm for 10 min, and use the supernatant, passing it if Tamsulosin Hydrochloride Capsules
Bundesdruckerei Berlin
 Europaisches Patentamt European Patent Office Office europeen des brevets Publication number: 0 289 353 A*2 EUROPEAN PATENT APPLICATION (5) Application number: 88303945.5 @ Date of filing: 29.04.88 @ Int.CI.*:
Europaisches Patentamt European Patent Office Office europeen des brevets Publication number: 0 289 353 A*2 EUROPEAN PATENT APPLICATION (5) Application number: 88303945.5 @ Date of filing: 29.04.88 @ Int.CI.*:
GASTRIC ORGANOID CULTURE PROTOCOL
 GASTRIC ORGANOID CULTURE PROTOCOL THIS PROTOCOL PROVIDES THE PROCEDURE FOR SUBCULTURING NORMAL HUMAN GASTRIC ORGANOIDS WHICH WAS DERIVED FROM THE SUBMERGED METHOD AS DESCRIBED IN BARKER N, ET AL. LGR5+VE
GASTRIC ORGANOID CULTURE PROTOCOL THIS PROTOCOL PROVIDES THE PROCEDURE FOR SUBCULTURING NORMAL HUMAN GASTRIC ORGANOIDS WHICH WAS DERIVED FROM THE SUBMERGED METHOD AS DESCRIBED IN BARKER N, ET AL. LGR5+VE
HRP-Antibody All-in-One Conjugation Kit. (Cat. # A-9002-001)
 HRP-Antibody All-in-One Conjugation Kit (Cat. # A-9002-001) Table of Contents I. Introduction a. Product Description 3 b. All-in-One Technology 3 Conjugation Chemistry 3 Conjugate Purification 5 c. All-in-One
HRP-Antibody All-in-One Conjugation Kit (Cat. # A-9002-001) Table of Contents I. Introduction a. Product Description 3 b. All-in-One Technology 3 Conjugation Chemistry 3 Conjugate Purification 5 c. All-in-One
Melting points (m.p.) were determined using a Reichert hot-stage melting point
 Supporting information 1.0 General experimental 1.1 Instrumentation Melting points (m.p.) were determined using a Reichert hot-stage melting point apparatus and are uncorrected. Infrared spectra (IR) spectra
Supporting information 1.0 General experimental 1.1 Instrumentation Melting points (m.p.) were determined using a Reichert hot-stage melting point apparatus and are uncorrected. Infrared spectra (IR) spectra
EZ Load Molecular Rulers. Catalog Numbers 170-8351 20 bp 170-8352 100 bp 170-8353 100 bp PCR 170-8354 500 bp 170-8355 1 kb 170-8356 Precision Mass
 EZ Load Molecular Rulers Catalog Numbers 170-8351 20 bp 170-8352 100 bp 170-8353 100 bp PCR 170-8354 500 bp 170-8355 1 kb 170-8356 Precision Mass EZ Load Molecular Rulers Quantity DNA sufficient for 100
EZ Load Molecular Rulers Catalog Numbers 170-8351 20 bp 170-8352 100 bp 170-8353 100 bp PCR 170-8354 500 bp 170-8355 1 kb 170-8356 Precision Mass EZ Load Molecular Rulers Quantity DNA sufficient for 100
Investigation of Solid-Phase Peptide Synthesis by the Near-Infrared Multispectral Imaging Technique: A Detection Method for Combinatorial Chemistry
 Anal. Chem. 1999, 71, 2255-2261 Accelerated Articles Investigation of Solid-Phase Peptide Synthesis by the Near-Infrared Multispectral Imaging Technique: A Detection Method for Combinatorial Chemistry
Anal. Chem. 1999, 71, 2255-2261 Accelerated Articles Investigation of Solid-Phase Peptide Synthesis by the Near-Infrared Multispectral Imaging Technique: A Detection Method for Combinatorial Chemistry
α-cyclodextrin SYNONYMS α-schardinger dextrin, α-dextrin, cyclohexaamylose, cyclomaltohexaose, α- cycloamylase
 α-cyclodextrin New specifications prepared at the 57th JECFA (2001) and published in FNP 52 Add 9 (2001). An ADI not specified was established at the 57th JECFA (2001). SYNONYMS α-schardinger dextrin,
α-cyclodextrin New specifications prepared at the 57th JECFA (2001) and published in FNP 52 Add 9 (2001). An ADI not specified was established at the 57th JECFA (2001). SYNONYMS α-schardinger dextrin,
Supporting Information. Self-Folded Redox/Acid Dual-Responsive Nanocarriers for. Anticancer Drug Delivery
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supporting Information Self-Folded Redox/Acid Dual-Responsive Nanocarriers for Anticancer Drug
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supporting Information Self-Folded Redox/Acid Dual-Responsive Nanocarriers for Anticancer Drug
MEF Starter Nucleofector Kit
 page 1 of 7 MEF Starter Nucleofector Kit for Mouse Embryonic Fibroblasts (MEF) MEF display significant phenotypic variations which depend on the strain, the genetic background of the mice they are isolated
page 1 of 7 MEF Starter Nucleofector Kit for Mouse Embryonic Fibroblasts (MEF) MEF display significant phenotypic variations which depend on the strain, the genetic background of the mice they are isolated
PROTOCOL 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 www.mitosciences.com
 PROTOCOL Western Blotting Transfer and Detection Procedure 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 02-11 DESCRIPTION Western Blotting Transfer and Detection Procedure ADDITIONAL MATERIALS REQUIRED
PROTOCOL Western Blotting Transfer and Detection Procedure 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 02-11 DESCRIPTION Western Blotting Transfer and Detection Procedure ADDITIONAL MATERIALS REQUIRED
Calculating Nucleic Acid or Protein Concentration Using the GloMax Multi+ Microplate Instrument
 Calculating Nucleic Acid or Protein Concentration Using the GloMax Multi+ Microplate Instrument Technical Note INTRODUCTION Direct measurements of nucleic acid samples at OD 260 or protein samples at OD
Calculating Nucleic Acid or Protein Concentration Using the GloMax Multi+ Microplate Instrument Technical Note INTRODUCTION Direct measurements of nucleic acid samples at OD 260 or protein samples at OD
Supporting Information
 Copyright WILEY VCH Verlag GmbH & Co. KGaA, 69469 Weinheim, Germany, 2012. Supporting Information for Adv. Funct. Mater., DOI: 10.1002/adfm.201102486 Colorimetric Detection of Warfare Gases by Polydiacetylenes
Copyright WILEY VCH Verlag GmbH & Co. KGaA, 69469 Weinheim, Germany, 2012. Supporting Information for Adv. Funct. Mater., DOI: 10.1002/adfm.201102486 Colorimetric Detection of Warfare Gases by Polydiacetylenes
The fastest spin-column based procedure for purifying up to 10 mg of ultra-pure endotoxin-free transfection-grade plasmid DNA.
 INSTRUCTION MANUAL ZymoPURE Plasmid Gigaprep Kit Catalog Nos. D4204 (Patent Pending) Highlights The fastest spin-column based procedure for purifying up to 10 mg of ultra-pure endotoxin-free transfection-grade
INSTRUCTION MANUAL ZymoPURE Plasmid Gigaprep Kit Catalog Nos. D4204 (Patent Pending) Highlights The fastest spin-column based procedure for purifying up to 10 mg of ultra-pure endotoxin-free transfection-grade
Quality. Now Certified to ISO 9001:2008
 Quality Now Certified to ISO 90012008 Quality Policy It is Peptides International's goal is to achieve complete customer satisfaction by addressing customer needs and delivering what we promise. The company
Quality Now Certified to ISO 90012008 Quality Policy It is Peptides International's goal is to achieve complete customer satisfaction by addressing customer needs and delivering what we promise. The company
EdU Flow Cytometry Kit. User Manual
 User Manual Ordering information: (for detailed kit content see Table 2) EdU Flow Cytometry Kits for 50 assays: Product number EdU Used fluorescent dye BCK-FC488-50 10 mg 6-FAM Azide BCK-FC555-50 10 mg
User Manual Ordering information: (for detailed kit content see Table 2) EdU Flow Cytometry Kits for 50 assays: Product number EdU Used fluorescent dye BCK-FC488-50 10 mg 6-FAM Azide BCK-FC555-50 10 mg
Supplemental Material for Jiang et al, Tumor Imaging via Proteolytic Activation of Cell Penetrating Peptides
 Supplemental Material for Jiang et al, Tumor Imaging via Proteolytic Activation of Cell Penetrating Peptides Reagents Fmoc protected amino acids and synthesis resins were purchased from EMD Chemicals Inc.
Supplemental Material for Jiang et al, Tumor Imaging via Proteolytic Activation of Cell Penetrating Peptides Reagents Fmoc protected amino acids and synthesis resins were purchased from EMD Chemicals Inc.
GENOTYPING ASSAYS AT ZIRC
 GENOTYPING ASSAYS AT ZIRC A. READ THIS FIRST - DISCLAIMER Dear ZIRC user, We now provide detailed genotyping protocols for a number of zebrafish lines distributed by ZIRC. These protocols were developed
GENOTYPING ASSAYS AT ZIRC A. READ THIS FIRST - DISCLAIMER Dear ZIRC user, We now provide detailed genotyping protocols for a number of zebrafish lines distributed by ZIRC. These protocols were developed
Northern blot analysis for microrna. (Narry Kim s lab)
 Northern blot analysis for microrna (Narry Kim s lab) Materials 1. 10~50 μg of total RNA extracted from HeLa cells treated with sirna 2. RNA loading buffer 3. Probe: DNA oligonucleotide complementary to
Northern blot analysis for microrna (Narry Kim s lab) Materials 1. 10~50 μg of total RNA extracted from HeLa cells treated with sirna 2. RNA loading buffer 3. Probe: DNA oligonucleotide complementary to
Measuring Protein Concentration through Absorption Spectrophotometry
 Measuring Protein Concentration through Absorption Spectrophotometry In this lab exercise you will learn how to homogenize a tissue to extract the protein, and then how to use a protein assay reagent to
Measuring Protein Concentration through Absorption Spectrophotometry In this lab exercise you will learn how to homogenize a tissue to extract the protein, and then how to use a protein assay reagent to
QUANTITATIVE RT-PCR. A = B (1+e) n. A=amplified products, B=input templates, n=cycle number, and e=amplification efficiency.
 QUANTITATIVE RT-PCR Application: Quantitative RT-PCR is used to quantify mrna in both relative and absolute terms. It can be applied for the quantification of mrna expressed from endogenous genes, and
QUANTITATIVE RT-PCR Application: Quantitative RT-PCR is used to quantify mrna in both relative and absolute terms. It can be applied for the quantification of mrna expressed from endogenous genes, and
TECHNICAL REPORT STUDY CHEMICAL PROPERTIES OF RATTAN SHOOT FROM PLANTATION IN THAILAND
 TECHNICAL REPORT STUDY CHEMICAL PROPERTIES OF RATTAN SHOOT FROM PLANTATION IN THAILAND by Assistant Professor Dr. Noojaree Prasitpan Chemist Analyzer of the ITTO Project on PD 24/00 Rev.1 (I): Promotion
TECHNICAL REPORT STUDY CHEMICAL PROPERTIES OF RATTAN SHOOT FROM PLANTATION IN THAILAND by Assistant Professor Dr. Noojaree Prasitpan Chemist Analyzer of the ITTO Project on PD 24/00 Rev.1 (I): Promotion
Two-Dimensional Gel Electrophoresis (2-DGE)
 - Introduction - Sample preparation - First dimension: Isoelectric focusing - Second dimension: SDS-PAGE - Detection of protein spots: staining - Imaging analysis & 2D Gel databases - Spot handling: excision,
- Introduction - Sample preparation - First dimension: Isoelectric focusing - Second dimension: SDS-PAGE - Detection of protein spots: staining - Imaging analysis & 2D Gel databases - Spot handling: excision,
RayBio Creatine Kinase (CK) Activity Colorimetric Assay Kit
 RayBio Creatine Kinase (CK) Activity Colorimetric Assay Kit User Manual Version 1.0 May 28, 2014 RayBio Creatine Kinase Activity Colorimetric Assay (Cat#: 68CL-CK-S100) RayBiotech, Inc. We Provide You
RayBio Creatine Kinase (CK) Activity Colorimetric Assay Kit User Manual Version 1.0 May 28, 2014 RayBio Creatine Kinase Activity Colorimetric Assay (Cat#: 68CL-CK-S100) RayBiotech, Inc. We Provide You
APPLICATIONS MANUAL. Ifosfamide in blood serum... 2. Ingredients in blood serum... 3. Organophosphorus pesticides in tea leaf... 5
 1 APPLICATIONS MANUAL CONTENTS Ifosfamide in blood serum... 2 Ingredients in blood serum... 3 Organophosphorus pesticides in tea leaf... 5 Sudan i ii iii iv in chilli sauce... 6 Pah in water... 7 Phenols
1 APPLICATIONS MANUAL CONTENTS Ifosfamide in blood serum... 2 Ingredients in blood serum... 3 Organophosphorus pesticides in tea leaf... 5 Sudan i ii iii iv in chilli sauce... 6 Pah in water... 7 Phenols
SmartFlare RNA Detection Probes: Principles, protocols and troubleshooting
 Technical Guide SmartFlare RNA Detection Probes: Principles, protocols and troubleshooting Principles of SmartFlare technology RNA detection traditionally requires transfection, laborious sample prep,
Technical Guide SmartFlare RNA Detection Probes: Principles, protocols and troubleshooting Principles of SmartFlare technology RNA detection traditionally requires transfection, laborious sample prep,
