Absolute Structure Absolute Configuration
|
|
|
- Aron Carson
- 8 years ago
- Views:
Transcription
1 Absolute Structure Absolute Configuration
2 Some definitions Absolute Configuration -> spatial arrangement of the atoms for a chiral molecule (R/S, P/M or D/L assignment). Absolute Structure -> spatial arrangement of atoms in a noncentrosymmetric crystal structure (unit-cell, space group)
3 Chiral molecules Molecules that cannot be superimposed with their mirror image Two mirror images of a chiral molecule are called enantiomers, they are optical isomers Determination of absolute configuration -> handness of the molecule
4 Asymmetric carbon atoms (R/S) Axial chirality Origin of chirality R-Binol S-Binol Chiral Propeller Arrangement (P/M or / ) Helicenes
5 In the solid state Chiral molecules can crystallize as an enantiopure bulk sample or as a racemic mixture. For enantiopure crystals Space group restriction: Only 65 space groups allowed for chiral molecules: No Inversion Center / No Mirror / No Glide Plane These include 11 pairs of eniantomorph space groups (screw axes of opposite handedness) eg: P4 1 /P4 3 or P6 1 /P6 5
6 Space Group Restrictions 85 %
7 Racemic mixture In the solid state 1) Conglomerate: a mixture of well-resolved crystals of both enantiomers Chiral space group Individual crystals have an optical activity 5-10 % 2) Racemates: One type of crystal containing the two enantiommers in a well defined stoechiometry. No optical activity Usually centrosymmetric space group %
8 Conglomerate
9 Racemates
10
11 Racemic mixture In the solid state 3) Inversion twin : twinned crystals of both enantiomers Chiral space group 4) Disordered solid-solution: Crystal containing the two enantiomers in a disordered arrangement. Usually centrosymmetric space group
12 Louis Pasteur Experiment (1848): Separation of the two enantiomers by the visual sorting of crystals of a conglomerate D-(-) levotartaric acid L-(+) dextrotartaric acid Sodium ammonium tartrate cristals
13 P2 1 P2 1 /c
14 Intensity (u.a.) Powder diffraction Ammonium Hydrogen Tartrate P (degrees) The two eniantopure samples and the conglomerate give the same powder X-ray diffraction pattern
15 Intensity (u.a.) Intensity (u.a.) Powder diffraction Ammonium Hydrogen Tartrate P (degrees) Ammonium Hydrogen Tartrate P2 1 /c (degrees)
16 Polar structures Achiral molecules can crystallize in chiral and non-centrosymmetric space groups Helical arrangement of SiO 4 tetrahedra in a-quartz (P3 1 21)
17 How we determine Absolute Configuration? Method 1 : Internal Chiral Reference Method 2 : From Absolute Structure
18 Absolute configuration from an internal chiral reference Co-crystallisation with a compound of known absolute configuration HOOC S N S COOMe H 3 C H NH 2 O O Iding et al., Tetrahedron Asymmetry 14 (2003)
19 Absolute configuration from Absolute Structure Method based on the anomalous scattering Friedel s Law is not respected: F( h, k, l) F( h, k, l) Heavy atom should be present: Light atoms usually show only a small anomalous dispersion effect MoKa: element > P (15) CuKa: element > O (8)
20 Absolute Configuration from Absolute Structure Refine the structure and its inverse, compare the R- factors Calculated pairs of reflections hkl vs hkl (Friedel /Bijvoet Pairs) can be examined Refinement of the Flack Parameter x I calc ( h, k, l) (1 x) F ( h, k, l) x F ( h, k, l) calc 2 calc 2 0 x 1 Diffraction from a crystal twinned by inversion
21 Reporting absolute structure Report x and its esd u If u > 0.3 : absolute structure cannot be determined reliably. 0 < x < 1 x 0: correct absolute structure x 1: wrong absolute structure invert structure! x 0.5: inversion twin
22 Refinement in Shelx
23 Refinement in Shelx L.S. 4 BOND $H TWIN BASF 0.1 FMAP 2 PLAN 5 ACTA WGHT FVAR MOLE 1 C = C = AFIX 13 H AFIX 0
24 Refinement in Shelx L.S. 4 BOND $H TWIN BASF FMAP 2 PLAN 5 ACTA WGHT FVAR MOLE 1 C = C = AFIX 13 H AFIX 0
25 Refinement in Shelx
26 Hooft parameter Hooft R. B. W., Straver L. H., Spek A. L. Determination of absolute structure using Bayesian statistics on Bijvoet differences. J. Applied Cryst. (2008) 41, Run SHELX without refining the Flack parameter. Generate the CIF file and the corresponding.fcf reflection file (ACTA instruction). Run PLATON and open the CIF File. On the Graphical Menu, select the ByVoetPair option.
27 Hooft parameter
28
29 Absolute Configuration from Absolute Structure Once you have determined the absolute structure and the absolute configuration of the molecule, you should ensure that the bulk sample is eniantopure i.e. contains only the eniantomer found. (eg. circular dichroism, chromatograpy) Useful References: H.D. Flack, G. Bernardinelli, Absolute structure and absolute configuration. Acta Cryst, (1999) A55, H.D. Flack, G. Bernardinelli, Reporting and evaluating absolute structure and absolute configuration determinations. J. Applied Cryst. (2000) 33, Thompson A. L., Watkin D. J. X-ray crystallography and chiralty: understanding the limitations. Tetrahedron: Asymmetry (2009) 20,
Molecule Projections
 Key Definitions ü Stereochemistry refers to the chemistry in 3 dimensions (greek stereos = solid). This science was created by Pasteur (1860), van Hoff et LeBel (1874). ü Stereisomers are isomeric molecules
Key Definitions ü Stereochemistry refers to the chemistry in 3 dimensions (greek stereos = solid). This science was created by Pasteur (1860), van Hoff et LeBel (1874). ü Stereisomers are isomeric molecules
Isomers Have same molecular formula, but different structures
 Isomers ave same molecular formula, but different structures Constitutional Isomers Differ in the order of attachment of atoms (different bond connectivity) Stereoisomers Atoms are connected in the same
Isomers ave same molecular formula, but different structures Constitutional Isomers Differ in the order of attachment of atoms (different bond connectivity) Stereoisomers Atoms are connected in the same
Electron density is complex!
 Electron density is complex! Göttingen, November 13 th 2008 George M. Sheldrick, Göttingen University http://shelx.uni-ac.gwdg.de/shelx/ Friedel s Law F h,k,l = F h, k, l and φ h,k,l = φ h, k, l Friedel
Electron density is complex! Göttingen, November 13 th 2008 George M. Sheldrick, Göttingen University http://shelx.uni-ac.gwdg.de/shelx/ Friedel s Law F h,k,l = F h, k, l and φ h,k,l = φ h, k, l Friedel
X-ray Structure Determination. Crystal Structure Solution
 X-ray Structure Determination Crystal Structure Solution Intro: Much of what you will have learned about crystal structures so far has been fairly abstract and far removed from what is actually done in
X-ray Structure Determination Crystal Structure Solution Intro: Much of what you will have learned about crystal structures so far has been fairly abstract and far removed from what is actually done in
LMB Crystallography Course, 2013. Crystals, Symmetry and Space Groups Andrew Leslie
 LMB Crystallography Course, 2013 Crystals, Symmetry and Space Groups Andrew Leslie Many of the slides were kindly provided by Erhard Hohenester (Imperial College), several other illustrations are from
LMB Crystallography Course, 2013 Crystals, Symmetry and Space Groups Andrew Leslie Many of the slides were kindly provided by Erhard Hohenester (Imperial College), several other illustrations are from
Phasing & Substructure Solution
 CCP4 Workshop Chicago 2011 Phasing & Substructure Solution Tim Grüne Dept. of Structural Chemistry, University of Göttingen June 2011 http://shelx.uni-ac.gwdg.de tg@shelx.uni-ac.gwdg.de CCP4 Chicago 2011:
CCP4 Workshop Chicago 2011 Phasing & Substructure Solution Tim Grüne Dept. of Structural Chemistry, University of Göttingen June 2011 http://shelx.uni-ac.gwdg.de tg@shelx.uni-ac.gwdg.de CCP4 Chicago 2011:
Quick Start Users Guide for Bruker SMART APEX II Diffractometer
 Quick Start Users Guide for Bruker SMART APEX II Diffractometer APENDIX IV INSTRUCTION FOR USING SQUEEZE J. Tanski 2/12/06 1 Suppose your files are f10.* (0) SQUEEZE should only be used for good reason;
Quick Start Users Guide for Bruker SMART APEX II Diffractometer APENDIX IV INSTRUCTION FOR USING SQUEEZE J. Tanski 2/12/06 1 Suppose your files are f10.* (0) SQUEEZE should only be used for good reason;
X-ray Powder Diffraction Pattern Indexing for Pharmaceutical Applications
 The published version of this manuscript may be found at the following webpage: http://www.pharmtech.com/pharmtech/peer-reviewed+research/x-ray-powder-diffraction-pattern-indexing-for- Phar/ArticleStandard/Article/detail/800851
The published version of this manuscript may be found at the following webpage: http://www.pharmtech.com/pharmtech/peer-reviewed+research/x-ray-powder-diffraction-pattern-indexing-for- Phar/ArticleStandard/Article/detail/800851
Assessment Schedule 2013 Chemistry: Demonstrate understanding of the properties of organic compounds (91391)
 NCEA Level 3 Chemistry (91391) 2013 page 1 of 8 Assessment Schedule 2013 Chemistry: Demonstrate understanding of the properties of organic compounds (91391) Evidence Statement Q Evidence Achievement Achievement
NCEA Level 3 Chemistry (91391) 2013 page 1 of 8 Assessment Schedule 2013 Chemistry: Demonstrate understanding of the properties of organic compounds (91391) Evidence Statement Q Evidence Achievement Achievement
Write the acid-base equilibria connecting all components in the aqueous solution. Now list all of the species present.
 Chapter 16 Acids and Bases Concept Check 16.1 Chemists in the seventeenth century discovered that the substance that gives red ants their irritating bite is an acid with the formula HCHO 2. They called
Chapter 16 Acids and Bases Concept Check 16.1 Chemists in the seventeenth century discovered that the substance that gives red ants their irritating bite is an acid with the formula HCHO 2. They called
Crystal Structure Determination I
 Crystal Structure Determination I Dr. Falak Sher Pakistan Institute of Engineering and Applied Sciences National Workshop on Crystal Structure Determination using Powder XRD, organized by the Khwarzimic
Crystal Structure Determination I Dr. Falak Sher Pakistan Institute of Engineering and Applied Sciences National Workshop on Crystal Structure Determination using Powder XRD, organized by the Khwarzimic
Basics of X-ray diffraction: From symmetry to structure determination
 Basics of X-ray diffraction: From symmetry to structure determination T. N. Guru Row Solid State and Structural Chemistry Unit Indian Institute of Science Bangalore-560012 Email: ssctng@sscu.iisc.ernet.in
Basics of X-ray diffraction: From symmetry to structure determination T. N. Guru Row Solid State and Structural Chemistry Unit Indian Institute of Science Bangalore-560012 Email: ssctng@sscu.iisc.ernet.in
Stereochemistry Tutorial: Drawing Enantiomers and Diastereomers
 tereochemistry Tutorial: Drawing Enantiomers and Diastereomers Definitions for vocabulary words can be found in the Illustrated Glossary of rganic Chemistry, available on the course web site. A. Discussion
tereochemistry Tutorial: Drawing Enantiomers and Diastereomers Definitions for vocabulary words can be found in the Illustrated Glossary of rganic Chemistry, available on the course web site. A. Discussion
especially the coplanarity of the conjugated amide system, are as required
 VOL. 37, 1951 CHEMISTRY: PA ULING AND COREY 241 THE STRUCTURE OF SYNTHETIC POLYPEPTIDES BY LINUS PAULING AND ROBERT B. COREY GATES AND CRELLIN LABORATORIES OF CHEMISTRY,* CALIFORNIA INSTITUTE OF TECH-
VOL. 37, 1951 CHEMISTRY: PA ULING AND COREY 241 THE STRUCTURE OF SYNTHETIC POLYPEPTIDES BY LINUS PAULING AND ROBERT B. COREY GATES AND CRELLIN LABORATORIES OF CHEMISTRY,* CALIFORNIA INSTITUTE OF TECH-
Chapter 11 Homework and practice questions Reactions of Alkyl Halides: Nucleophilic Substitutions and Eliminations
 Chapter 11 Homework and practice questions Reactions of Alkyl Halides: Nucleophilic Substitutions and Eliminations SHORT ANSWER Exhibit 11-1 Circle your response in each set below. 1. Circle the least
Chapter 11 Homework and practice questions Reactions of Alkyl Halides: Nucleophilic Substitutions and Eliminations SHORT ANSWER Exhibit 11-1 Circle your response in each set below. 1. Circle the least
methyl RX example primary RX example secondary RX example secondary RX example tertiary RX example
 ucleophilic Substitution & Elimination hemistry 1 eginning patterns to knowfor S and E eactions - horizontal and vertical templates for practice Example 1 - two possible perspectives (deuterium and tritium
ucleophilic Substitution & Elimination hemistry 1 eginning patterns to knowfor S and E eactions - horizontal and vertical templates for practice Example 1 - two possible perspectives (deuterium and tritium
1. What is the hybridization of the indicated atom in the following molecule?
 Practice Final Exam, Chemistry 2210, rganic Chem I 1. What is the hybridization of the indicated atom in the following molecule? A. sp 3 B. sp 2 C. sp D. not hybridized 2. Name the functional groups in
Practice Final Exam, Chemistry 2210, rganic Chem I 1. What is the hybridization of the indicated atom in the following molecule? A. sp 3 B. sp 2 C. sp D. not hybridized 2. Name the functional groups in
Bonding & Molecular Shape Ron Robertson
 Bonding & Molecular Shape Ron Robertson r2 n:\files\courses\1110-20\2010 possible slides for web\00bondingtrans.doc The Nature of Bonding Types 1. Ionic 2. Covalent 3. Metallic 4. Coordinate covalent Driving
Bonding & Molecular Shape Ron Robertson r2 n:\files\courses\1110-20\2010 possible slides for web\00bondingtrans.doc The Nature of Bonding Types 1. Ionic 2. Covalent 3. Metallic 4. Coordinate covalent Driving
Molecular Models in Biology
 Molecular Models in Biology Objectives: After this lab a student will be able to: 1) Understand the properties of atoms that give rise to bonds. 2) Understand how and why atoms form ions. 3) Model covalent,
Molecular Models in Biology Objectives: After this lab a student will be able to: 1) Understand the properties of atoms that give rise to bonds. 2) Understand how and why atoms form ions. 3) Model covalent,
Publication of small-unit-cell structures in Acta Crystallographica Michael Hoyland ECM28 University of Warwick, 2013
 Publication of small-unit-cell structures in Acta Crystallographica Michael Hoyland ECM28 University of Warwick, 2013 International Union of Crystallography 5 Abbey Square Chester CH1 2HU UK International
Publication of small-unit-cell structures in Acta Crystallographica Michael Hoyland ECM28 University of Warwick, 2013 International Union of Crystallography 5 Abbey Square Chester CH1 2HU UK International
Communicated March 31, 1951 CHR CHR CHR. *H* zz '" *H _ 0.-...H / k C,.. CHR CNR CHR CHR CHR *HN/' N 'H_N/' H_./ - H-(H.
 VOL. 37, 1951 CHEMISTR Y: PA ULING AND COREY 251 THE PLEATED SHEET, A NEW LAYER CONFIGURATION OF POL YPEPTIDE CHAINS BY LINUS PAULING AND ROBERT B. COREY GATES AND CRELLIN LABORATORIES OF CHEMISTRY,* CALIFORNIA
VOL. 37, 1951 CHEMISTR Y: PA ULING AND COREY 251 THE PLEATED SHEET, A NEW LAYER CONFIGURATION OF POL YPEPTIDE CHAINS BY LINUS PAULING AND ROBERT B. COREY GATES AND CRELLIN LABORATORIES OF CHEMISTRY,* CALIFORNIA
Classical direct methods
 Classical direct methods Göttingen, December 4 th 2008 George M. Sheldrick http://shelx.uni-ac.gwdg.de/shelx/ The crystallographic phase problem In order to calculate an electron density map, we require
Classical direct methods Göttingen, December 4 th 2008 George M. Sheldrick http://shelx.uni-ac.gwdg.de/shelx/ The crystallographic phase problem In order to calculate an electron density map, we require
Properties of a molecule. Absolute configuration, Cahn-Ingold Prelog. Planar, axial, topological chirality and chirality at atoms other than carbon
 Key stereochemical terminology Stereochemical terminology in organic chemistry can refer to structure of a molecule or to properties of a physical sample of the molecule. It is very important to recognize
Key stereochemical terminology Stereochemical terminology in organic chemistry can refer to structure of a molecule or to properties of a physical sample of the molecule. It is very important to recognize
In the box below, draw the Lewis electron-dot structure for the compound formed from magnesium and oxygen. [Include any charges or partial charges.
 Name: 1) Which molecule is nonpolar and has a symmetrical shape? A) NH3 B) H2O C) HCl D) CH4 7222-1 - Page 1 2) When ammonium chloride crystals are dissolved in water, the temperature of the water decreases.
Name: 1) Which molecule is nonpolar and has a symmetrical shape? A) NH3 B) H2O C) HCl D) CH4 7222-1 - Page 1 2) When ammonium chloride crystals are dissolved in water, the temperature of the water decreases.
Quick Guide for Data Collection on the NIU Bruker Smart CCD
 Quick Guide for Data Collection on the NIU Bruker Smart CCD 1. Create a new project 2. Optically align the crystal 3. Take rotation picture 4. Collect matrix to determine unit cell 5. Refine unit cell
Quick Guide for Data Collection on the NIU Bruker Smart CCD 1. Create a new project 2. Optically align the crystal 3. Take rotation picture 4. Collect matrix to determine unit cell 5. Refine unit cell
Practical suggestions for better crystal structures
 Crystallography Reviews Vol. 15, No. 1, January March 2009, 57 83 Practical suggestions for better crystal structures Peter Mu ller* Department of Chemistry, Massachusetts Institute of Technology, Cambridge,
Crystallography Reviews Vol. 15, No. 1, January March 2009, 57 83 Practical suggestions for better crystal structures Peter Mu ller* Department of Chemistry, Massachusetts Institute of Technology, Cambridge,
CHEM 121. Chapter 18. Name: Date: 1. Which of the following compounds is both an aldose and a hexose? A) Page 1
 CEM 121. Chapter 18. Name: Date: 1. Which of the following compounds is both an aldose and a hexose? A) B) C) D) Page 1 2. Which of the following structures is that of an L-monosaccharide? A) B) C) D)
CEM 121. Chapter 18. Name: Date: 1. Which of the following compounds is both an aldose and a hexose? A) B) C) D) Page 1 2. Which of the following structures is that of an L-monosaccharide? A) B) C) D)
Enantiomers: Synthesis, characterization, and resolution of tris(ethylenediamine)cobalt(iii) chloride Introduction:
 Enantiomers: Synthesis, characterization, and resolution of tris(ethylenediamine)cobalt(iii) chloride Introduction: The development of coordination chemistry prior to 1950 involved the synthesis and characterization
Enantiomers: Synthesis, characterization, and resolution of tris(ethylenediamine)cobalt(iii) chloride Introduction: The development of coordination chemistry prior to 1950 involved the synthesis and characterization
CHAPTER 10: INTERMOLECULAR FORCES: THE UNIQUENESS OF WATER Problems: 10.2, 10.6,10.15-10.33, 10.35-10.40, 10.56-10.60, 10.101-10.
 CHAPTER 10: INTERMOLECULAR FORCES: THE UNIQUENESS OF WATER Problems: 10.2, 10.6,10.15-10.33, 10.35-10.40, 10.56-10.60, 10.101-10.102 10.1 INTERACTIONS BETWEEN IONS Ion-ion Interactions and Lattice Energy
CHAPTER 10: INTERMOLECULAR FORCES: THE UNIQUENESS OF WATER Problems: 10.2, 10.6,10.15-10.33, 10.35-10.40, 10.56-10.60, 10.101-10.102 10.1 INTERACTIONS BETWEEN IONS Ion-ion Interactions and Lattice Energy
Diffraction Course Series 2015
 Diffraction Course Series 2015 Mark Wainwright Analytical Centre Kensington Campus, Chemical Sciences Building F10, Room G37 The Mark Wainwright Analytical Centre is offering a new series of courses covering
Diffraction Course Series 2015 Mark Wainwright Analytical Centre Kensington Campus, Chemical Sciences Building F10, Room G37 The Mark Wainwright Analytical Centre is offering a new series of courses covering
Structure Check. Authors: Eduard Schreiner Leonardo G. Trabuco. February 7, 2012
 University of Illinois at Urbana-Champaign NIH Resource for Macromolecular Modeling and Bioinformatics Beckman Institute Computational Biophysics Workshop Structure Check Authors: Eduard Schreiner Leonardo
University of Illinois at Urbana-Champaign NIH Resource for Macromolecular Modeling and Bioinformatics Beckman Institute Computational Biophysics Workshop Structure Check Authors: Eduard Schreiner Leonardo
MCAT Organic Chemistry - Problem Drill 23: Amino Acids, Peptides and Proteins
 MCAT rganic Chemistry - Problem Drill 23: Amino Acids, Peptides and Proteins Question No. 1 of 10 Question 1. Which amino acid does not contain a chiral center? Question #01 (A) Serine (B) Proline (C)
MCAT rganic Chemistry - Problem Drill 23: Amino Acids, Peptides and Proteins Question No. 1 of 10 Question 1. Which amino acid does not contain a chiral center? Question #01 (A) Serine (B) Proline (C)
IR Applied to Isomer Analysis
 DiscovIR-LC TM Application Note 025 April 2008 Deposition and Detection System IR Applied to Isomer Analysis Infrared spectra provide valuable information about local configurations of atoms in molecules.
DiscovIR-LC TM Application Note 025 April 2008 Deposition and Detection System IR Applied to Isomer Analysis Infrared spectra provide valuable information about local configurations of atoms in molecules.
Chemical and Structural Classifications
 Chemical and Structural Classifications What? Materials can be classified by their chemistry and structure. There are three main types of structural classifications: 1. Prototype Structure Structure prototyping
Chemical and Structural Classifications What? Materials can be classified by their chemistry and structure. There are three main types of structural classifications: 1. Prototype Structure Structure prototyping
2.4 Enantiomeric Isomers
 2.4 Enantiomeric Isomers There is an additional type of isomer that can exist when we have an atom with 4 different groups attached to that atom in tetrahedral geometry. Consider a methane molecule with
2.4 Enantiomeric Isomers There is an additional type of isomer that can exist when we have an atom with 4 different groups attached to that atom in tetrahedral geometry. Consider a methane molecule with
Features of the formation of hydrogen bonds in solutions of polysaccharides during their use in various industrial processes. V.Mank a, O.
 Features of the formation of hydrogen bonds in solutions of polysaccharides during their use in various industrial processes. V.Mank a, O. Melnyk b a National University of life and environmental sciences
Features of the formation of hydrogen bonds in solutions of polysaccharides during their use in various industrial processes. V.Mank a, O. Melnyk b a National University of life and environmental sciences
VSEPR Model. The Valence-Shell Electron Pair Repulsion Model. Predicting Molecular Geometry
 VSEPR Model The structure around a given atom is determined principally by minimizing electron pair repulsions. The Valence-Shell Electron Pair Repulsion Model The valence-shell electron pair repulsion
VSEPR Model The structure around a given atom is determined principally by minimizing electron pair repulsions. The Valence-Shell Electron Pair Repulsion Model The valence-shell electron pair repulsion
Amino Acids. Amino acids are the building blocks of proteins. All AA s have the same basic structure: Side Chain. Alpha Carbon. Carboxyl. Group.
 Protein Structure Amino Acids Amino acids are the building blocks of proteins. All AA s have the same basic structure: Side Chain Alpha Carbon Amino Group Carboxyl Group Amino Acid Properties There are
Protein Structure Amino Acids Amino acids are the building blocks of proteins. All AA s have the same basic structure: Side Chain Alpha Carbon Amino Group Carboxyl Group Amino Acid Properties There are
Structure Factors 59-553 78
 78 Structure Factors Until now, we have only typically considered reflections arising from planes in a hypothetical lattice containing one atom in the asymmetric unit. In practice we will generally deal
78 Structure Factors Until now, we have only typically considered reflections arising from planes in a hypothetical lattice containing one atom in the asymmetric unit. In practice we will generally deal
research papers Single-crystal structure validation with the program PLATON
 Journal of Applied Crystallography ISSN 0021-8898 Received 27 September 2002 Accepted 28 November 2002 Single-crystal structure validation with the program PLATON A. L. Spek Crystal and Structural Chemistry,
Journal of Applied Crystallography ISSN 0021-8898 Received 27 September 2002 Accepted 28 November 2002 Single-crystal structure validation with the program PLATON A. L. Spek Crystal and Structural Chemistry,
Chapter 12 Organic Compounds with Oxygen and Sulfur
 Chapter 12 Organic Compounds with Oxygen and Sulfur 1 Alcohols An alcohol contains a hydroxyl group ( OH) that replaces a hydrogen atom in a hydrocarbon. A phenol contains a hydroxyl group ( OH) attached
Chapter 12 Organic Compounds with Oxygen and Sulfur 1 Alcohols An alcohol contains a hydroxyl group ( OH) that replaces a hydrogen atom in a hydrocarbon. A phenol contains a hydroxyl group ( OH) attached
Guide to Understanding X-ray Crystallography
 Guide to Understanding X-ray Crystallography What is X-ray Crystallography and why do I need to learn it? X-ray Crystallography is a scientific method of determining the precise positions/arrangements
Guide to Understanding X-ray Crystallography What is X-ray Crystallography and why do I need to learn it? X-ray Crystallography is a scientific method of determining the precise positions/arrangements
Solid Phase Peptide Synthesis
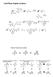 Solid Phase Peptide Synthesis Fischer Projections revisited C C C Remember that group rotations are always allowed C C 80 rotations of entire molecule are allowed C 80 C 90 rotations of entire molecule
Solid Phase Peptide Synthesis Fischer Projections revisited C C C Remember that group rotations are always allowed C C 80 rotations of entire molecule are allowed C 80 C 90 rotations of entire molecule
Polarization Dependence in X-ray Spectroscopy and Scattering. S P Collins et al Diamond Light Source UK
 Polarization Dependence in X-ray Spectroscopy and Scattering S P Collins et al Diamond Light Source UK Overview of talk 1. Experimental techniques at Diamond: why we care about x-ray polarization 2. How
Polarization Dependence in X-ray Spectroscopy and Scattering S P Collins et al Diamond Light Source UK Overview of talk 1. Experimental techniques at Diamond: why we care about x-ray polarization 2. How
21 Crystal Packing. by Angelo Gavezzotti and Howard Flack
 21 Crystal Packing by Angelo Gavezzotti and Howard Flack This electronic edition may be freely copied and redistributed for educational or research purposes only. It may not be sold for profit nor incorporated
21 Crystal Packing by Angelo Gavezzotti and Howard Flack This electronic edition may be freely copied and redistributed for educational or research purposes only. It may not be sold for profit nor incorporated
Solid State Theory Physics 545
 Solid State Theory Physics 545 CRYSTAL STRUCTURES Describing periodic structures Terminology Basic Structures Symmetry Operations Ionic crystals often have a definite habit which gives rise to particular
Solid State Theory Physics 545 CRYSTAL STRUCTURES Describing periodic structures Terminology Basic Structures Symmetry Operations Ionic crystals often have a definite habit which gives rise to particular
The CIF file, refinement details and validation of the structure
 The CIF file, refinement details and validation of the structure CCCW 2011 Artefacts versus errors Artefacts in crystallography: An error which cannot be avoided, since it is inherent in the method. Examples
The CIF file, refinement details and validation of the structure CCCW 2011 Artefacts versus errors Artefacts in crystallography: An error which cannot be avoided, since it is inherent in the method. Examples
Science Standard Articulated by Grade Level Strand 5: Physical Science
 Concept 1: Properties of Objects and Materials Classify objects and materials by their observable properties. Kindergarten Grade 1 Grade 2 Grade 3 Grade 4 PO 1. Identify the following observable properties
Concept 1: Properties of Objects and Materials Classify objects and materials by their observable properties. Kindergarten Grade 1 Grade 2 Grade 3 Grade 4 PO 1. Identify the following observable properties
Combinatorial Biochemistry and Phage Display
 Combinatorial Biochemistry and Phage Display Prof. Valery A. Petrenko Director - Valery Petrenko Instructors Galina Kouzmitcheva and I-Hsuan Chen Auburn 2006, Spring semester COMBINATORIAL BIOCHEMISTRY
Combinatorial Biochemistry and Phage Display Prof. Valery A. Petrenko Director - Valery Petrenko Instructors Galina Kouzmitcheva and I-Hsuan Chen Auburn 2006, Spring semester COMBINATORIAL BIOCHEMISTRY
CHEM 208(Organic Chemistry I) Instructor: Dr. Niranjan Goswami. Tel: (618)545-3361. Email: Ngoswami@kaskaskia.edu. Web: www.kc.cc.il.
 CHEM 208(Organic Chemistry I) Instructor: Dr. Niranjan Goswami Tel: (618)545-3361 Email: Ngoswami@kaskaskia.edu Web: www.kc.cc.il.us/ngoswami CHEM 208 COURSE SYLLABUS KASKASKIA COLLEGE NAME TERM YEAR TEXT:
CHEM 208(Organic Chemistry I) Instructor: Dr. Niranjan Goswami Tel: (618)545-3361 Email: Ngoswami@kaskaskia.edu Web: www.kc.cc.il.us/ngoswami CHEM 208 COURSE SYLLABUS KASKASKIA COLLEGE NAME TERM YEAR TEXT:
Crystal Structure Determination of 5,6-Dibromoacenaphthene from X-ray Powder Diffraction Data
 ScienceAsia 28 (2002) : 271-275 Crystal Structure Determination of 5,6-Dibromoacenaphthene from X-ray Powder Diffraction Data Nongnuj Jaiboon* Department of Chemistry, Faculty of Science, Chulalongkorn
ScienceAsia 28 (2002) : 271-275 Crystal Structure Determination of 5,6-Dibromoacenaphthene from X-ray Powder Diffraction Data Nongnuj Jaiboon* Department of Chemistry, Faculty of Science, Chulalongkorn
On Factoring Chirality and Stereoisomerism
 Topics in Stereochemisty, Volume 14 Edited by Ernest L. Eliel, Norman L. Allinger, Samuel H. Wilen Copyright 1983 by John Wiley & Sons, Inc. On Factoring Chirality and Stereoisomerism HANS HIRSCHMANN Department
Topics in Stereochemisty, Volume 14 Edited by Ernest L. Eliel, Norman L. Allinger, Samuel H. Wilen Copyright 1983 by John Wiley & Sons, Inc. On Factoring Chirality and Stereoisomerism HANS HIRSCHMANN Department
Ionic Bonding Pauling s Rules and the Bond Valence Method
 Ionic Bonding Pauling s Rules and the Bond Valence Method Chemistry 754 Solid State Chemistry Dr. Patrick Woodward Lecture #14 Pauling Rules for Ionic Structures Linus Pauling,, J. Amer. Chem. Soc. 51,,
Ionic Bonding Pauling s Rules and the Bond Valence Method Chemistry 754 Solid State Chemistry Dr. Patrick Woodward Lecture #14 Pauling Rules for Ionic Structures Linus Pauling,, J. Amer. Chem. Soc. 51,,
Matter, Materials, Crystal Structure and Bonding. Chris J. Pickard
 Matter, Materials, Crystal Structure and Bonding Chris J. Pickard Why should a theorist care? Where the atoms are determines what they do Where the atoms can be determines what we can do Overview of Structure
Matter, Materials, Crystal Structure and Bonding Chris J. Pickard Why should a theorist care? Where the atoms are determines what they do Where the atoms can be determines what we can do Overview of Structure
Survival Organic Chemistry Part I: Molecular Models
 Survival Organic Chemistry Part I: Molecular Models The goal in this laboratory experience is to get you so you can easily and quickly move between empirical formulas, molecular formulas, condensed formulas,
Survival Organic Chemistry Part I: Molecular Models The goal in this laboratory experience is to get you so you can easily and quickly move between empirical formulas, molecular formulas, condensed formulas,
Chem101: General Chemistry Lecture 9 Acids and Bases
 : General Chemistry Lecture 9 Acids and Bases I. Introduction A. In chemistry, and particularly biochemistry, water is the most common solvent 1. In studying acids and bases we are going to see that water
: General Chemistry Lecture 9 Acids and Bases I. Introduction A. In chemistry, and particularly biochemistry, water is the most common solvent 1. In studying acids and bases we are going to see that water
Introduction to X-Ray Powder Diffraction Data Analysis
 Introduction to X-Ray Powder Diffraction Data Analysis Center for Materials Science and Engineering at MIT http://prism.mit.edu/xray An X-ray diffraction pattern is a plot of the intensity of X-rays scattered
Introduction to X-Ray Powder Diffraction Data Analysis Center for Materials Science and Engineering at MIT http://prism.mit.edu/xray An X-ray diffraction pattern is a plot of the intensity of X-rays scattered
C has 4 valence electrons, O has six electrons. The total number of electrons is 4 + 2(6) = 16.
 129 Lewis Structures G. N. Lewis hypothesized that electron pair bonds between unlike elements in the second (and sometimes the third) row occurred in a way that electrons were shared such that each element
129 Lewis Structures G. N. Lewis hypothesized that electron pair bonds between unlike elements in the second (and sometimes the third) row occurred in a way that electrons were shared such that each element
Chem 11 Oa-Section 2. Exam #2. 1 October 2004
 " Chem 11 Oa-Section 2 Exam #2 1 October 2004 Name: K~ Instructions Please read each question carefully and answer it completely and clearly. Do the problems in the order that is easiest for you. Point
" Chem 11 Oa-Section 2 Exam #2 1 October 2004 Name: K~ Instructions Please read each question carefully and answer it completely and clearly. Do the problems in the order that is easiest for you. Point
STANDARD ANSWERS AND DEFINITIONS
 Evidence for Kekule s model to be wrong: STANDARD ANSWERS AND DEFINITIONS All C-C bond lengths are the same length, between C-C and C=C. Only reacts with Br2 with a halogen carrier Benzene is lower in
Evidence for Kekule s model to be wrong: STANDARD ANSWERS AND DEFINITIONS All C-C bond lengths are the same length, between C-C and C=C. Only reacts with Br2 with a halogen carrier Benzene is lower in
 2814 hains, Rings and Spectroscopy June 2003 Mark Scheme 2814 Mark Scheme June 2003 The following annotations may be used when marking: X = incorrect response (errors may also be underlined) ^ = omission
2814 hains, Rings and Spectroscopy June 2003 Mark Scheme 2814 Mark Scheme June 2003 The following annotations may be used when marking: X = incorrect response (errors may also be underlined) ^ = omission
MEMORANDUM GRADE 11. PHYSICAL SCIENCES: CHEMISTRY Paper 2
 MEMORANDUM GRADE 11 PHYSICAL SCIENCES: CHEMISTRY Paper 2 MARKS: 150 TIME: 3 hours Learning Outcomes and Assessment Standards LO1 LO2 LO3 AS 11.1.1: Plan and conduct a scientific investigation to collect
MEMORANDUM GRADE 11 PHYSICAL SCIENCES: CHEMISTRY Paper 2 MARKS: 150 TIME: 3 hours Learning Outcomes and Assessment Standards LO1 LO2 LO3 AS 11.1.1: Plan and conduct a scientific investigation to collect
3) How many monosaccharides are connected to each other in a disaccharide? A) 1 B) 2 C) 3 D) 4
 General, Organic, and Biochemistry, 2e (Frost) HOMEWORK Chapter 6 Carbohydrates Life s Sweet Molecules 6.1 Multiple-Choice 1) Which of the following is a polysaccharide? Glucose Sucrose C) Starch D) Maltose
General, Organic, and Biochemistry, 2e (Frost) HOMEWORK Chapter 6 Carbohydrates Life s Sweet Molecules 6.1 Multiple-Choice 1) Which of the following is a polysaccharide? Glucose Sucrose C) Starch D) Maltose
Getting the most from this book...4 About this book...5
 Contents Getting the most from this book...4 About this book....5 Content Guidance Topic 1 Atomic structure and the periodic table...8 Topic 2 Bonding and structure...14 Topic 2A Bonding....14 Topic 2B
Contents Getting the most from this book...4 About this book....5 Content Guidance Topic 1 Atomic structure and the periodic table...8 Topic 2 Bonding and structure...14 Topic 2A Bonding....14 Topic 2B
Chapter 6 Assessment. Name: Class: Date: ID: A. Multiple Choice Identify the choice that best completes the statement or answers the question.
 Name: Class: Date: ID: A Chapter 6 Assessment Multiple Choice Identify the choice that best completes the statement or answers the question. 1. When an atom loses an electron, it forms a(n) a. anion. c.
Name: Class: Date: ID: A Chapter 6 Assessment Multiple Choice Identify the choice that best completes the statement or answers the question. 1. When an atom loses an electron, it forms a(n) a. anion. c.
The Mole Concept and Atoms
 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Chapter 4 24 September 2013 Calculations and the Chemical Equation The Mole Concept and Atoms Atoms are exceedingly
Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Chapter 4 24 September 2013 Calculations and the Chemical Equation The Mole Concept and Atoms Atoms are exceedingly
Amount of Substance. http://www.avogadro.co.uk/definitions/elemcompmix.htm
 Page 1 of 14 Amount of Substance Key terms in this chapter are: Element Compound Mixture Atom Molecule Ion Relative Atomic Mass Avogadro constant Mole Isotope Relative Isotopic Mass Relative Molecular
Page 1 of 14 Amount of Substance Key terms in this chapter are: Element Compound Mixture Atom Molecule Ion Relative Atomic Mass Avogadro constant Mole Isotope Relative Isotopic Mass Relative Molecular
EXPERIMENT 1: Survival Organic Chemistry: Molecular Models
 EXPERIMENT 1: Survival Organic Chemistry: Molecular Models Introduction: The goal in this laboratory experience is for you to easily and quickly move between empirical formulas, molecular formulas, condensed
EXPERIMENT 1: Survival Organic Chemistry: Molecular Models Introduction: The goal in this laboratory experience is for you to easily and quickly move between empirical formulas, molecular formulas, condensed
GCE. Chemistry A. Mark Scheme for June 2010. Advanced GCE F325 Equilibria, Energetics and Elements. Oxford Cambridge and RSA Examinations
 GCE Chemistry A Advanced GCE F325 Equilibria, Energetics and Elements Mark Scheme for June 2010 Oxford Cambridge and RSA Examinations OCR (Oxford Cambridge and RSA) is a leading UK awarding body, providing
GCE Chemistry A Advanced GCE F325 Equilibria, Energetics and Elements Mark Scheme for June 2010 Oxford Cambridge and RSA Examinations OCR (Oxford Cambridge and RSA) is a leading UK awarding body, providing
* Dr. Jyotsna Chauhan, * Dr. Rachna Tiwari, ** Dr. R.K Tiwari
 Comparative X-Ray Structure analysis of systemic fungicides (N-(2,6 dimethyl phenyl)-n-(2-keto-1- methyl butyl) 3-hydroxypropanamide) AND cis N-(1,1,2,2-tetrachloroethylthio)-4-cyclohexene-1,2- dicarboximide)
Comparative X-Ray Structure analysis of systemic fungicides (N-(2,6 dimethyl phenyl)-n-(2-keto-1- methyl butyl) 3-hydroxypropanamide) AND cis N-(1,1,2,2-tetrachloroethylthio)-4-cyclohexene-1,2- dicarboximide)
Chapter 2 The Chemical Context of Life
 Chapter 2 The Chemical Context of Life Multiple-Choice Questions 1) About 25 of the 92 natural elements are known to be essential to life. Which four of these 25 elements make up approximately 96% of living
Chapter 2 The Chemical Context of Life Multiple-Choice Questions 1) About 25 of the 92 natural elements are known to be essential to life. Which four of these 25 elements make up approximately 96% of living
Extension of COSMO-RS to interfacial phenomena
 Extension of COSMO-RS to interfacial phenomena Martin P. Andersson et al. Nano-Science Center Department of Chemistry University of Copenhagen Denmark Presentation at the 4th COSMO-RS symposium Mar 2015
Extension of COSMO-RS to interfacial phenomena Martin P. Andersson et al. Nano-Science Center Department of Chemistry University of Copenhagen Denmark Presentation at the 4th COSMO-RS symposium Mar 2015
hij GCSE Additional Science Chemistry 2 Higher Tier Chemistry 2H SPECIMEN MARK SCHEME Version 1.0
 hij GCSE Additional Science Chemistry 2 Higher Tier Chemistry 2H SPECIMEN MARK SCHEME Version.0 Copyright 20 AQA and its licensors. All rights reserved. The Assessment and Qualifications Alliance (AQA)
hij GCSE Additional Science Chemistry 2 Higher Tier Chemistry 2H SPECIMEN MARK SCHEME Version.0 Copyright 20 AQA and its licensors. All rights reserved. The Assessment and Qualifications Alliance (AQA)
How much does a single atom weigh? Different elements weigh different amounts related to what makes them unique.
 How much does a single atom weigh? Different elements weigh different amounts related to what makes them unique. What units do we use to define the weight of an atom? amu units of atomic weight. (atomic
How much does a single atom weigh? Different elements weigh different amounts related to what makes them unique. What units do we use to define the weight of an atom? amu units of atomic weight. (atomic
U3-LM2B-WS Molar Mass and Conversions
 U3-LM2B-WS Molar Mass and Conversions Name: KEY 1. The molar mass of chlorine is: 2 x 35.45 g/mol Cl = 70.90 g/mol Cl 2 (Remember that chlorine exists as a diatomic molecule in nature) 2. The molar mass
U3-LM2B-WS Molar Mass and Conversions Name: KEY 1. The molar mass of chlorine is: 2 x 35.45 g/mol Cl = 70.90 g/mol Cl 2 (Remember that chlorine exists as a diatomic molecule in nature) 2. The molar mass
X-ray thin-film measurement techniques
 Technical articles X-ray thin-film measurement techniques II. Out-of-plane diffraction measurements Toru Mitsunaga* 1. Introduction A thin-film sample is two-dimensionally formed on the surface of a substrate,
Technical articles X-ray thin-film measurement techniques II. Out-of-plane diffraction measurements Toru Mitsunaga* 1. Introduction A thin-film sample is two-dimensionally formed on the surface of a substrate,
Sugar Identification using Polarimetry
 Sugar Identification using Polarimetry Safety Concerns: No special safety precautions need to be implemented. Standard organic laboratory safety measures need to be in place. Purpose The purpose of this
Sugar Identification using Polarimetry Safety Concerns: No special safety precautions need to be implemented. Standard organic laboratory safety measures need to be in place. Purpose The purpose of this
Chapter 4: Structure and Properties of Ionic and Covalent Compounds
 Chapter 4: Structure and Properties of Ionic and Covalent Compounds 4.1 Chemical Bonding o Chemical Bond - the force of attraction between any two atoms in a compound. o Interactions involving valence
Chapter 4: Structure and Properties of Ionic and Covalent Compounds 4.1 Chemical Bonding o Chemical Bond - the force of attraction between any two atoms in a compound. o Interactions involving valence
Q.1 Carbonyl compounds are formed by oxidation of alcohols;
 arbonyl compounds 814 1 ARBYL MPUDS - Aldehydes and Ketones Q.1 arbonyl compounds are formed by oxidation of alcohols; a) Which type of alcohol is oxidised to an aldehyde? b) Which type of alcohol is oxidised
arbonyl compounds 814 1 ARBYL MPUDS - Aldehydes and Ketones Q.1 arbonyl compounds are formed by oxidation of alcohols; a) Which type of alcohol is oxidised to an aldehyde? b) Which type of alcohol is oxidised
The Mole Concept. The Mole. Masses of molecules
 The Mole Concept Ron Robertson r2 c:\files\courses\1110-20\2010 final slides for web\mole concept.docx The Mole The mole is a unit of measurement equal to 6.022 x 10 23 things (to 4 sf) just like there
The Mole Concept Ron Robertson r2 c:\files\courses\1110-20\2010 final slides for web\mole concept.docx The Mole The mole is a unit of measurement equal to 6.022 x 10 23 things (to 4 sf) just like there
Chapter 8 Basic Concepts of the Chemical Bonding
 Chapter 8 Basic Concepts of the Chemical Bonding 1. There are paired and unpaired electrons in the Lewis symbol for a phosphorus atom. (a). 4, 2 (b). 2, 4 (c). 4, 3 (d). 2, 3 Explanation: Read the question
Chapter 8 Basic Concepts of the Chemical Bonding 1. There are paired and unpaired electrons in the Lewis symbol for a phosphorus atom. (a). 4, 2 (b). 2, 4 (c). 4, 3 (d). 2, 3 Explanation: Read the question
CHEMISTRY BONDING REVIEW
 Answer the following questions. CHEMISTRY BONDING REVIEW 1. What are the three kinds of bonds which can form between atoms? The three types of Bonds are Covalent, Ionic and Metallic. Name Date Block 2.
Answer the following questions. CHEMISTRY BONDING REVIEW 1. What are the three kinds of bonds which can form between atoms? The three types of Bonds are Covalent, Ionic and Metallic. Name Date Block 2.
Molecular Models Experiment #1
 Molecular Models Experiment #1 Objective: To become familiar with the 3-dimensional structure of organic molecules, especially the tetrahedral structure of alkyl carbon atoms and the planar structure of
Molecular Models Experiment #1 Objective: To become familiar with the 3-dimensional structure of organic molecules, especially the tetrahedral structure of alkyl carbon atoms and the planar structure of
WRITING CHEMICAL FORMULA
 WRITING CHEMICAL FORMULA For ionic compounds, the chemical formula must be worked out. You will no longer have the list of ions in the exam (like at GCSE). Instead you must learn some and work out others.
WRITING CHEMICAL FORMULA For ionic compounds, the chemical formula must be worked out. You will no longer have the list of ions in the exam (like at GCSE). Instead you must learn some and work out others.
Defects Introduction. Bonding + Structure + Defects. Properties
 Defects Introduction Bonding + Structure + Defects Properties The processing determines the defects Composition Bonding type Structure of Crystalline Processing factors Defects Microstructure Types of
Defects Introduction Bonding + Structure + Defects Properties The processing determines the defects Composition Bonding type Structure of Crystalline Processing factors Defects Microstructure Types of
Synthesis and Crystal Structure of Na 2 TiO 3
 27 卷 5 期 结 构 化 学 (JIEGOU HUAXUE) Vol. 27, No.5 2008. 5 Chinese J. Struct. Chem. 553~557 Synthesis and Crystal Structure of Na 2 TiO 3 MAO Shao-Yu 1 REN Xiao-Xia ZHOU Zhao-Hui (State Key Laboratory for
27 卷 5 期 结 构 化 学 (JIEGOU HUAXUE) Vol. 27, No.5 2008. 5 Chinese J. Struct. Chem. 553~557 Synthesis and Crystal Structure of Na 2 TiO 3 MAO Shao-Yu 1 REN Xiao-Xia ZHOU Zhao-Hui (State Key Laboratory for
Chapter 1 Structure and Bonding. Modified by Dr. Daniela Radu
 John E. McMurry www.cengage.com/chemistry/mcmurry Chapter 1 Structure and Bonding Modified by Dr. Daniela Radu What is Organic Chemistry? Living things are made of organic chemicals Proteins that make
John E. McMurry www.cengage.com/chemistry/mcmurry Chapter 1 Structure and Bonding Modified by Dr. Daniela Radu What is Organic Chemistry? Living things are made of organic chemicals Proteins that make
Answer Key, Problem Set 11
 Chemistry 122 Mines, Spring 2010 Answer Key, Problem Set 11 1. Write the electron configuration for each of the following atoms and ions (use the noble gas abbreviation): (a) Mn: [Ar] 4s 2 3d 5 (Note:
Chemistry 122 Mines, Spring 2010 Answer Key, Problem Set 11 1. Write the electron configuration for each of the following atoms and ions (use the noble gas abbreviation): (a) Mn: [Ar] 4s 2 3d 5 (Note:
Phase determination methods in macromolecular X- ray Crystallography
 Phase determination methods in macromolecular X- ray Crystallography Importance of protein structure determination: Proteins are the life machinery and are very essential for the various functions in the
Phase determination methods in macromolecular X- ray Crystallography Importance of protein structure determination: Proteins are the life machinery and are very essential for the various functions in the
Unit 11 Practice. Name: Class: Date: Multiple Choice Identify the choice that best completes the statement or answers the question.
 Name: Class: Date: Unit 11 Practice Multiple Choice Identify the choice that best completes the statement or answers the question. 1) Crystalline solids. A) have their particles arranged randomly B) have
Name: Class: Date: Unit 11 Practice Multiple Choice Identify the choice that best completes the statement or answers the question. 1) Crystalline solids. A) have their particles arranged randomly B) have
Software fayre: raw data hkl file
 Software fayre: raw data hkl file malvaro@vt.edu Matteo Alvaro www.crystal.vt.edu This work was supported by: NSF grant - Earth Sciences Division EAR-0738692 Department of Geosciences, Virginia Tech. College
Software fayre: raw data hkl file malvaro@vt.edu Matteo Alvaro www.crystal.vt.edu This work was supported by: NSF grant - Earth Sciences Division EAR-0738692 Department of Geosciences, Virginia Tech. College
A reinterpretation of the phase transitions in Na 2 CO 3
 Acta Crystallographica Section B Structural Science ISSN 0108-7681 Editor: Carolyn P. Brock A reinterpretation of the phase transitions in Na 2 CO 3 Alla Arakcheeva and Gervais Chapuis Copyright International
Acta Crystallographica Section B Structural Science ISSN 0108-7681 Editor: Carolyn P. Brock A reinterpretation of the phase transitions in Na 2 CO 3 Alla Arakcheeva and Gervais Chapuis Copyright International
CHAPTER 6 Chemical Bonding
 CHAPTER 6 Chemical Bonding SECTION 1 Introduction to Chemical Bonding OBJECTIVES 1. Define Chemical bond. 2. Explain why most atoms form chemical bonds. 3. Describe ionic and covalent bonding.. 4. Explain
CHAPTER 6 Chemical Bonding SECTION 1 Introduction to Chemical Bonding OBJECTIVES 1. Define Chemical bond. 2. Explain why most atoms form chemical bonds. 3. Describe ionic and covalent bonding.. 4. Explain
Chapter 5 Classification of Organic Compounds by Solubility
 Chapter 5 Classification of Organic Compounds by Solubility Deductions based upon interpretation of simple solubility tests can be extremely useful in organic structure determination. Both solubility and
Chapter 5 Classification of Organic Compounds by Solubility Deductions based upon interpretation of simple solubility tests can be extremely useful in organic structure determination. Both solubility and
Element of same atomic number, but different atomic mass o Example: Hydrogen
 Atomic mass: p + = protons; e - = electrons; n 0 = neutrons p + + n 0 = atomic mass o For carbon-12, 6p + + 6n 0 = atomic mass of 12.0 o For chlorine-35, 17p + + 18n 0 = atomic mass of 35.0 atomic mass
Atomic mass: p + = protons; e - = electrons; n 0 = neutrons p + + n 0 = atomic mass o For carbon-12, 6p + + 6n 0 = atomic mass of 12.0 o For chlorine-35, 17p + + 18n 0 = atomic mass of 35.0 atomic mass
Chemistry Post-Enrolment Worksheet
 Name: Chemistry Post-Enrolment Worksheet The purpose of this worksheet is to get you to recap some of the fundamental concepts that you studied at GCSE and introduce some of the concepts that will be part
Name: Chemistry Post-Enrolment Worksheet The purpose of this worksheet is to get you to recap some of the fundamental concepts that you studied at GCSE and introduce some of the concepts that will be part
Type of Chemical Bonds
 Type of Chemical Bonds Covalent bond Polar Covalent bond Ionic bond Hydrogen bond Metallic bond Van der Waals bonds. Covalent Bonds Covalent bond: bond in which one or more pairs of electrons are shared
Type of Chemical Bonds Covalent bond Polar Covalent bond Ionic bond Hydrogen bond Metallic bond Van der Waals bonds. Covalent Bonds Covalent bond: bond in which one or more pairs of electrons are shared
Classification of Chemical Substances
 Classification of Chemical Substances INTRODUCTION: Depending on the kind of bonding present in a chemical substance, the substance may be called ionic, molecular or metallic. In a solid ionic compound
Classification of Chemical Substances INTRODUCTION: Depending on the kind of bonding present in a chemical substance, the substance may be called ionic, molecular or metallic. In a solid ionic compound
X-Ray Diffraction HOW IT WORKS WHAT IT CAN AND WHAT IT CANNOT TELL US. Hanno zur Loye
 X-Ray Diffraction HOW IT WORKS WHAT IT CAN AND WHAT IT CANNOT TELL US Hanno zur Loye X-rays are electromagnetic radiation of wavelength about 1 Å (10-10 m), which is about the same size as an atom. The
X-Ray Diffraction HOW IT WORKS WHAT IT CAN AND WHAT IT CANNOT TELL US Hanno zur Loye X-rays are electromagnetic radiation of wavelength about 1 Å (10-10 m), which is about the same size as an atom. The
Phase diagram of water. Note: for H 2 O melting point decreases with increasing pressure, for CO 2 melting point increases with increasing pressure.
 Phase diagram of water Note: for H 2 O melting point decreases with increasing pressure, for CO 2 melting point increases with increasing pressure. WATER Covers ~ 70% of the earth s surface Life on earth
Phase diagram of water Note: for H 2 O melting point decreases with increasing pressure, for CO 2 melting point increases with increasing pressure. WATER Covers ~ 70% of the earth s surface Life on earth
