Cancer Biostatistics Workshop Science of Doing Science - Biostatistics
|
|
|
- Jessica Washington
- 10 years ago
- Views:
Transcription
1 Cancer Biostatistics Workshop Science of Doing Science - Biostatistics Yu Shyr, PhD Jan. 18, 2008 Cancer Biostatistics Center Vanderbilt-Ingram Cancer Center [email protected]
2 Aims Cancer Biostatistics Workshop Introduction. Issues of the experiment design of biomedical research adaptive, Bayesian. P-value, type I error, FDR and methods of multiple comparison. Topics of the high-dimensional data experiment including the assessment of the quality control. Tools of the visualization.
3 Series One: What You Probably Don t Know About Biostatistics January 18 th - Yu Shyr, PhD The Science of Doing Science Biostatistics February 15 th Dan Ayers, MS Skittles, An Ounce of Measurement, and Intentional Science March 21 st Ayumi Shintani, MPH, PhD Introduction to Power and Sample Size Estimation
4 Series Two: Essentials of Efficient Experimental Design April 18 th - Tatsuki Koyama, PhD Phase II Clinical Trials May 16 th - Leena Choi, PhD Adaptive Designs in Dose-Finding (Phase I) Studies: A Bayesian Approach June 20 th - Fei Ye, PhD Phase III Clinical Trials
5 Series Three: High-Dimensional Data August 15 th - Chun Li, PhD Genome-wide Association Analysis for the Shanghai Breast Cancer Study September 19 th - Irene Feurer, PhD An Introduction to Principal Component Analysis and Data Reduction Methodologies October 17 th - Ming Li, PhD Statistical Analysis Strategies for MALDI-TOF and Shotgun Proteomics Data
6 Special Topics: Observational Data and Epidemiology November 21 st - Ayumi Shintani, MPH, PhD Introduction to Observational Study Design ~~---~ ~---~~ Please call or [email protected] if you have any questions or feedback.
7 Type of the Biomedical Studies in vitro cell lines in vivo animal models Clinical Trials Phase I, II, III, IV Cohort Study Prospective, Retrospective Case-Control Study Nested Case-Control
8 Experiment Design Primary objective of the study Primary hypothesis of the study Primary endpoint of the study How many groups? How many time points? Preliminary data
9 Adaptive Experiment Design Adaptive trial design refers to a clinical trial methodology that allows trial design modifications to be made after patients have been enrolled in a study, without compromising the scientific method. In order to maintain the integrity of the trial, these modifications should be clearly defined in the protocol. When designed well, an adaptive trial empowers sponsors to respond to data collected during the trial. This is achieved by re-focusing the trial in a way that maximizes the impact of each subject s contribution. Examples of adaptive trial designs include dropping a treatment arm, modifying the sample size, balancing treatment assignments using adaptive randomization or simply stopping a study early for success or failure.
10 Two Stage Design One of the primary objectives of a phase II clinical trial of a new drug or regimen is to determine whether it has sufficient biological activity against the disease under study to warrant more extensive development. Several two-stage Phase II designs will be discussed here Gehan, Fleming, Simon s Optimal, Simons s Minimax, Balanced Design
11 Gehan Design Gehan s (1961) design is the oldest design. It is a twostage design for estimating the response rate but providing for early termination if the drug shows insufficient antitumor activity. The design is most commonly used with a first stage of 14 patients. If no responses (completed response or partial response) are observed, the trial is terminated. The rationale for stopping is that if the true response probability were at least 20%, at least one response would very likely have been observed in the first 14 patients. If no responses are seen, it is unlikely that the true response probability is at least 20%.
12 If at least one response is observed in the first 14 patients, then a second stage of accrual is carried out to obtain an estimate of the response rate. The number of patients to accrue in the second stage depends upon the number of responses observed in the first stage and upon the precision desired for the final estimate of response rate. If the first stage consists of 14 patients, the second stage will consist of between one and 11 patients if a standard error of 10% is desired. The second stage will consist of between 45 and 86 patients if a standard error of 5% is desired.
13 Simon s Optimal Design The optimal two-stage designs (Simon, 1989) are optimal in the sense that the expected sample size is minimized if the regimen has low activity subject to constraints upon the size of the type I ( ) and type II ( ) errors. Example of Simon s Optimal Design: Clinical uninteresting level = 5% response rate, Clinical interesting level = 20% response rate, type I error ( ) = 0.05 type II error ( ) = 0.20 or Power = 0.80 (Power = 1 type II error). Stage I Reject drug if response rate 0/10 Stage II Reject drug if response rate 3/29
14 Frequentist versus Bayesians Frequentists: α and β errors Bayesians: Quantify designs with other properties General philosophy Start with prior information ( prior distribution ) Observe data ( likelihood function ) Combine prior and data to get posterior distribution Make inferences based on posterior
15 Bayesian inference No p-values and confidence intervals From the posterior distribution: Posterior probabilities Prediction intervals Credible intervals Bayesian designs Can look at data as often as you like (!) Use information as it accumulates Make what if? calculations Helps decide whether to stop now or not
16 Bayesian Designs Requires prior Reflects uncertainty about the response rate Can be vague, uninformative Can be controversial: inference may change
17 Bayesian Designs
18 Posterior Probabilities
19 Other priors What if we had used a different prior? Assume informative orange prior
20 Experiment Design Variability of In Situ Proteomic Profiling and Implications for Study Design in Colorectal Tumors International Journal of Oncology (2007)
21 Overview The comprehension of intrinsic tumor heterogeneity is vital for understanding of tumor progression mechanisms as well as for providing efficient treatments. In situ proteomic profiling of tumors is a powerful technology with potential to enhance our understanding of tumor biology, but sources of variability due to patient and tumor heterogeneity are poorly understood and are the topic of this investigation.
22 Questions Is proteomic profiling in adenomas more homogenous across patients than in normal mucosa specimens? Does primary carcinoma exhibit greater heterogeneity than normal mucosa and adenomas? Is Inter- and intra-case variability approximately equal for protein peaks? What is the optimal number of sub-samples per case that will reduce the total number of cases required? How to characterize intra- and inter-case variability of highthroughput protein expression in colorectal tumors?
23 Table IV Power = 80% Type I error = 5% Intra-Case Variance Subsample Inter-Case Variance Inter-Case Variance Inter-Case Variance Number (m)
24 P-value, Type I error, FDR, and multiple comparison Type I error: For testing a null hypothesis, H0, against an alternative hypothesis, the type I error of the test is the rejection of the null hypothesis when it is true. The p value of a statistical test is the probability of observing a sample outcome as extreme or more extreme as the one observed. This probability is computed under the assumption that the null hypothesis is true.
25 y Linear Regression 15 group=1 group= group=3 group= x Graphs by group
26 Linear Regression Regression Results: Slope 0.5 (p=0.0022) Parameter Standard T for H0: Variable DF Estimate Error Parameter=0 Prob > T INTERCEP X Which graph on the previous slide corresponds to this analysis?
27 Solutions for the Multiple Comparison Problem A MCP Solution Must Control False Positives How to measure multiple false positives? Familywise Error Rate (FWER) Chance of any false positives Controlled by Bonferroni & Random Field Methods False Discovery Rate (FDR) Proportion of false positives among rejected tests
28 False Discovery Rate Accept Reject Null True (no diff) V 0A V 0R m 0 Null False V 1A V 1R m 1 N A N R V Observed FDR Obs FDR = V 0R /(V 1R +V 0R ) = V 0R /N R If N R = 0, obsfdr = 0 Only know N R, not how many are true or false Control is on the expected FDR FDR = E(obsFDR)
29 p-value 0 1 Benjamini & Hochberg Procedure Select desired limit q on FDR Order p-values, p (1) p (2)... p (V) Let r be largest i such that JRSS-B (1995) 57: p (i) i / v q / c(v) p (i) Reject all hypotheses corresponding to p (1),..., p (r). i / v i/v q/c(v) 0 1
30 Benjamini & Hochberg: Key Properties FDR is controlled E(obsFDR) q m 0 /V Conservative, if large fraction of nulls false Adaptive Threshold depends on amount of feature More feature, More small p-values, More p (i) less than i/v q/c(v)
31 Benjamini & Hochberg Procedure c(v) = 1 Positive Regression Dependency on Subsets P(X 1 c 1, X 2 c 2,..., X k c k X i =x i ) is non-decreasing in x i Only required of test statistics for which null true Special cases include Independence Multivariate Normal with all positive correlations Same, but studentized with common std. err. c(v) = i=1,...,v 1/i log(v) Arbitrary covariance structure Benjamini & Yekutieli (2001). Ann. Stat. 29:
32 High Dimensional Data The major challenge in high throughput experiments, e.g., microarray data, MALDI-TOF data, Array CGH data, Shotgun Proteomic data, or SNP data, is that the data is often high dimensional. When the number of dimensions reaches thousands or more, the computational time for the pattern recognition algorithms can become unreasonable. This can be a problem, especially when some of the features are not discriminatory.
33 Statistical Determinants of Analytic Uncertainty Contributions to uncertainty can come from a variety of resources: Analytic variation Sampling methods Sample preparation Biological Heterogeneity in Population Specimen Collection/Handling Effects - Tumor: surgical related effects - Cell Line: culture condition Biological Heterogeneity in Specimen
34 Statistical Determinants of Analytic Uncertainty ISO When estimating the uncertainty of measurement, all uncertainty components which are of importance in the given situation shall be taken into account using appropriate methods of analysis. repeatability standard deviation (intra-lab) reproducibility standard deviation (inter-lab) intermediate precision (more factors in the system)
35 Statistical Determinants of Analytic Uncertainty The main objectives in performing an R&R study are to identify and quantify the absolute and relative contribution of each source of variation, to decide if the measurement process is adequate or not and, if not, to correct the errors by recalibrating the instrument, training the operators, other mathematical corrections, etc. In quality control and quality assurance, the goal is to reduce the variability in the system and, consequently, the variability in the biomarker measurement.
36 Quality Control Assessment Reproducibility Correlation of Variation (CV) SD/Mean Intra-class Correlation Coefficient (ICC) Intra / Intra + Inter Variance Component Analysis Mixed/Random Effect Model. The model: investigators, day, spot, machine, lab, etc. Goal Make sure the data is reproducible!!
37 Accuracy WFCCM Class Prediction Model Training data set - 93 vs 91 Testing data set - 52 vs Features
38 Receiver Operating Characteristic (ROC) Curve Training Test
39 WFCCM Class Prediction Model % GP1 % GP2 % correct B % Gp1 B % Gp2 B %
40 Receiver Operating Characteristic (ROC) Curve Training Test
41 Hierarchical clustering of gene expression data
42 Multidimensional scaling (MDS)
43 Multidimensional Scaling
44 Agulnik, M. et al. J Clin Oncol; 25: Multidimensional Scaling
45 Dear Dr. Shyr, Statistical Issues in Clinical Research - Design, Analysis and Interpretation I am a fellow in the Department of...we put together a paper recently... I had performed the statistical analysis incorrectly... A reviewer pointed out that with so many t tests some might give a significant result by chance and suggested we present ANOVA F statistics instead...we will pay the full cost of any such work you may perform... Thanks and Regards Dr...
46 Statistical Issues in Clinical Research - Design, Analysis and Interpretation When Do You Need a Biostatistician? Study Design Implementation Statistical Data Analysis Abstract Writing Paper Writing Letter to the paper reviewer
47 END
False Discovery Rates
 False Discovery Rates John D. Storey Princeton University, Princeton, USA January 2010 Multiple Hypothesis Testing In hypothesis testing, statistical significance is typically based on calculations involving
False Discovery Rates John D. Storey Princeton University, Princeton, USA January 2010 Multiple Hypothesis Testing In hypothesis testing, statistical significance is typically based on calculations involving
Statistical issues in the analysis of microarray data
 Statistical issues in the analysis of microarray data Daniel Gerhard Institute of Biostatistics Leibniz University of Hannover ESNATS Summerschool, Zermatt D. Gerhard (LUH) Analysis of microarray data
Statistical issues in the analysis of microarray data Daniel Gerhard Institute of Biostatistics Leibniz University of Hannover ESNATS Summerschool, Zermatt D. Gerhard (LUH) Analysis of microarray data
Likelihood Approaches for Trial Designs in Early Phase Oncology
 Likelihood Approaches for Trial Designs in Early Phase Oncology Clinical Trials Elizabeth Garrett-Mayer, PhD Cody Chiuzan, PhD Hollings Cancer Center Department of Public Health Sciences Medical University
Likelihood Approaches for Trial Designs in Early Phase Oncology Clinical Trials Elizabeth Garrett-Mayer, PhD Cody Chiuzan, PhD Hollings Cancer Center Department of Public Health Sciences Medical University
Study Design and Statistical Analysis
 Study Design and Statistical Analysis Anny H Xiang, PhD Department of Preventive Medicine University of Southern California Outline Designing Clinical Research Studies Statistical Data Analysis Designing
Study Design and Statistical Analysis Anny H Xiang, PhD Department of Preventive Medicine University of Southern California Outline Designing Clinical Research Studies Statistical Data Analysis Designing
Statistical Analysis Strategies for Shotgun Proteomics Data
 Statistical Analysis Strategies for Shotgun Proteomics Data Ming Li, Ph.D. Cancer Biostatistics Center Vanderbilt University Medical Center Ayers Institute Biomarker Pipeline normal shotgun proteome analysis
Statistical Analysis Strategies for Shotgun Proteomics Data Ming Li, Ph.D. Cancer Biostatistics Center Vanderbilt University Medical Center Ayers Institute Biomarker Pipeline normal shotgun proteome analysis
Service courses for graduate students in degree programs other than the MS or PhD programs in Biostatistics.
 Course Catalog In order to be assured that all prerequisites are met, students must acquire a permission number from the education coordinator prior to enrolling in any Biostatistics course. Courses are
Course Catalog In order to be assured that all prerequisites are met, students must acquire a permission number from the education coordinator prior to enrolling in any Biostatistics course. Courses are
Endpoint Selection in Phase II Oncology trials
 Endpoint Selection in Phase II Oncology trials Anastasia Ivanova Department of Biostatistics UNC at Chapel Hill [email protected] Primary endpoints in Phase II trials Recently looked at journal articles
Endpoint Selection in Phase II Oncology trials Anastasia Ivanova Department of Biostatistics UNC at Chapel Hill [email protected] Primary endpoints in Phase II trials Recently looked at journal articles
Personalized Predictive Medicine and Genomic Clinical Trials
 Personalized Predictive Medicine and Genomic Clinical Trials Richard Simon, D.Sc. Chief, Biometric Research Branch National Cancer Institute http://brb.nci.nih.gov brb.nci.nih.gov Powerpoint presentations
Personalized Predictive Medicine and Genomic Clinical Trials Richard Simon, D.Sc. Chief, Biometric Research Branch National Cancer Institute http://brb.nci.nih.gov brb.nci.nih.gov Powerpoint presentations
Statistical Analysis. NBAF-B Metabolomics Masterclass. Mark Viant
 Statistical Analysis NBAF-B Metabolomics Masterclass Mark Viant 1. Introduction 2. Univariate analysis Overview of lecture 3. Unsupervised multivariate analysis Principal components analysis (PCA) Interpreting
Statistical Analysis NBAF-B Metabolomics Masterclass Mark Viant 1. Introduction 2. Univariate analysis Overview of lecture 3. Unsupervised multivariate analysis Principal components analysis (PCA) Interpreting
Introduction. Hypothesis Testing. Hypothesis Testing. Significance Testing
 Introduction Hypothesis Testing Mark Lunt Arthritis Research UK Centre for Ecellence in Epidemiology University of Manchester 13/10/2015 We saw last week that we can never know the population parameters
Introduction Hypothesis Testing Mark Lunt Arthritis Research UK Centre for Ecellence in Epidemiology University of Manchester 13/10/2015 We saw last week that we can never know the population parameters
Department/Academic Unit: Public Health Sciences Degree Program: Biostatistics Collaborative Program
 Department/Academic Unit: Public Health Sciences Degree Program: Biostatistics Collaborative Program Department of Mathematics and Statistics Degree Level Expectations, Learning Outcomes, Indicators of
Department/Academic Unit: Public Health Sciences Degree Program: Biostatistics Collaborative Program Department of Mathematics and Statistics Degree Level Expectations, Learning Outcomes, Indicators of
PubH 7470: STATISTICS FOR TRANSLATIONAL & CLINICAL RESEARCH
 PubH 7470: STATISTICS FOR TRANSLATIONAL & CLINICAL RESEARCH CLINICAL TRIALS: TWO-STAGE PHASE II TRIALS This lecture covers a very special form of phase II clinical trials: two-stage design. A small group
PubH 7470: STATISTICS FOR TRANSLATIONAL & CLINICAL RESEARCH CLINICAL TRIALS: TWO-STAGE PHASE II TRIALS This lecture covers a very special form of phase II clinical trials: two-stage design. A small group
Tutorial for proteome data analysis using the Perseus software platform
 Tutorial for proteome data analysis using the Perseus software platform Laboratory of Mass Spectrometry, LNBio, CNPEM Tutorial version 1.0, January 2014. Note: This tutorial was written based on the information
Tutorial for proteome data analysis using the Perseus software platform Laboratory of Mass Spectrometry, LNBio, CNPEM Tutorial version 1.0, January 2014. Note: This tutorial was written based on the information
Analysis Issues II. Mary Foulkes, PhD Johns Hopkins University
 This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike License. Your use of this material constitutes acceptance of that license and the conditions of use of materials on this
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike License. Your use of this material constitutes acceptance of that license and the conditions of use of materials on this
1. What is the critical value for this 95% confidence interval? CV = z.025 = invnorm(0.025) = 1.96
 1 Final Review 2 Review 2.1 CI 1-propZint Scenario 1 A TV manufacturer claims in its warranty brochure that in the past not more than 10 percent of its TV sets needed any repair during the first two years
1 Final Review 2 Review 2.1 CI 1-propZint Scenario 1 A TV manufacturer claims in its warranty brochure that in the past not more than 10 percent of its TV sets needed any repair during the first two years
Statistics Graduate Courses
 Statistics Graduate Courses STAT 7002--Topics in Statistics-Biological/Physical/Mathematics (cr.arr.).organized study of selected topics. Subjects and earnable credit may vary from semester to semester.
Statistics Graduate Courses STAT 7002--Topics in Statistics-Biological/Physical/Mathematics (cr.arr.).organized study of selected topics. Subjects and earnable credit may vary from semester to semester.
Bayesian Phase I/II clinical trials in Oncology
 Bayesian Phase I/II clinical trials in Oncology Pierre Mancini, Sandrine Micallef, Pierre Colin Séminaire JEM-SFES - 26 Janvier 2012 Outline Oncology phase I trials Limitations of traditional phase I designs
Bayesian Phase I/II clinical trials in Oncology Pierre Mancini, Sandrine Micallef, Pierre Colin Séminaire JEM-SFES - 26 Janvier 2012 Outline Oncology phase I trials Limitations of traditional phase I designs
Data, Measurements, Features
 Data, Measurements, Features Middle East Technical University Dep. of Computer Engineering 2009 compiled by V. Atalay What do you think of when someone says Data? We might abstract the idea that data are
Data, Measurements, Features Middle East Technical University Dep. of Computer Engineering 2009 compiled by V. Atalay What do you think of when someone says Data? We might abstract the idea that data are
Descriptive Statistics
 Descriptive Statistics Primer Descriptive statistics Central tendency Variation Relative position Relationships Calculating descriptive statistics Descriptive Statistics Purpose to describe or summarize
Descriptive Statistics Primer Descriptive statistics Central tendency Variation Relative position Relationships Calculating descriptive statistics Descriptive Statistics Purpose to describe or summarize
Fixed-Effect Versus Random-Effects Models
 CHAPTER 13 Fixed-Effect Versus Random-Effects Models Introduction Definition of a summary effect Estimating the summary effect Extreme effect size in a large study or a small study Confidence interval
CHAPTER 13 Fixed-Effect Versus Random-Effects Models Introduction Definition of a summary effect Estimating the summary effect Extreme effect size in a large study or a small study Confidence interval
II. DISTRIBUTIONS distribution normal distribution. standard scores
 Appendix D Basic Measurement And Statistics The following information was developed by Steven Rothke, PhD, Department of Psychology, Rehabilitation Institute of Chicago (RIC) and expanded by Mary F. Schmidt,
Appendix D Basic Measurement And Statistics The following information was developed by Steven Rothke, PhD, Department of Psychology, Rehabilitation Institute of Chicago (RIC) and expanded by Mary F. Schmidt,
Applying Statistics Recommended by Regulatory Documents
 Applying Statistics Recommended by Regulatory Documents Steven Walfish President, Statistical Outsourcing Services [email protected] 301-325 325-31293129 About the Speaker Mr. Steven
Applying Statistics Recommended by Regulatory Documents Steven Walfish President, Statistical Outsourcing Services [email protected] 301-325 325-31293129 About the Speaker Mr. Steven
Regression Analysis: A Complete Example
 Regression Analysis: A Complete Example This section works out an example that includes all the topics we have discussed so far in this chapter. A complete example of regression analysis. PhotoDisc, Inc./Getty
Regression Analysis: A Complete Example This section works out an example that includes all the topics we have discussed so far in this chapter. A complete example of regression analysis. PhotoDisc, Inc./Getty
Statistics in Medicine Research Lecture Series CSMC Fall 2014
 Catherine Bresee, MS Senior Biostatistician Biostatistics & Bioinformatics Research Institute Statistics in Medicine Research Lecture Series CSMC Fall 2014 Overview Review concept of statistical power
Catherine Bresee, MS Senior Biostatistician Biostatistics & Bioinformatics Research Institute Statistics in Medicine Research Lecture Series CSMC Fall 2014 Overview Review concept of statistical power
2013 MBA Jump Start Program. Statistics Module Part 3
 2013 MBA Jump Start Program Module 1: Statistics Thomas Gilbert Part 3 Statistics Module Part 3 Hypothesis Testing (Inference) Regressions 2 1 Making an Investment Decision A researcher in your firm just
2013 MBA Jump Start Program Module 1: Statistics Thomas Gilbert Part 3 Statistics Module Part 3 Hypothesis Testing (Inference) Regressions 2 1 Making an Investment Decision A researcher in your firm just
Introduction to Regression and Data Analysis
 Statlab Workshop Introduction to Regression and Data Analysis with Dan Campbell and Sherlock Campbell October 28, 2008 I. The basics A. Types of variables Your variables may take several forms, and it
Statlab Workshop Introduction to Regression and Data Analysis with Dan Campbell and Sherlock Campbell October 28, 2008 I. The basics A. Types of variables Your variables may take several forms, and it
Introduction to Longitudinal Data Analysis
 Introduction to Longitudinal Data Analysis Longitudinal Data Analysis Workshop Section 1 University of Georgia: Institute for Interdisciplinary Research in Education and Human Development Section 1: Introduction
Introduction to Longitudinal Data Analysis Longitudinal Data Analysis Workshop Section 1 University of Georgia: Institute for Interdisciplinary Research in Education and Human Development Section 1: Introduction
From Reads to Differentially Expressed Genes. The statistics of differential gene expression analysis using RNA-seq data
 From Reads to Differentially Expressed Genes The statistics of differential gene expression analysis using RNA-seq data experimental design data collection modeling statistical testing biological heterogeneity
From Reads to Differentially Expressed Genes The statistics of differential gene expression analysis using RNA-seq data experimental design data collection modeling statistical testing biological heterogeneity
Comparative genomic hybridization Because arrays are more than just a tool for expression analysis
 Microarray Data Analysis Workshop MedVetNet Workshop, DTU 2008 Comparative genomic hybridization Because arrays are more than just a tool for expression analysis Carsten Friis ( with several slides from
Microarray Data Analysis Workshop MedVetNet Workshop, DTU 2008 Comparative genomic hybridization Because arrays are more than just a tool for expression analysis Carsten Friis ( with several slides from
Gene Expression Analysis
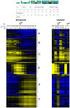 Gene Expression Analysis Jie Peng Department of Statistics University of California, Davis May 2012 RNA expression technologies High-throughput technologies to measure the expression levels of thousands
Gene Expression Analysis Jie Peng Department of Statistics University of California, Davis May 2012 RNA expression technologies High-throughput technologies to measure the expression levels of thousands
Functional Data Analysis of MALDI TOF Protein Spectra
 Functional Data Analysis of MALDI TOF Protein Spectra Dean Billheimer [email protected]. Department of Biostatistics Vanderbilt University Vanderbilt Ingram Cancer Center FDA for MALDI TOF
Functional Data Analysis of MALDI TOF Protein Spectra Dean Billheimer [email protected]. Department of Biostatistics Vanderbilt University Vanderbilt Ingram Cancer Center FDA for MALDI TOF
Validation and Calibration. Definitions and Terminology
 Validation and Calibration Definitions and Terminology ACCEPTANCE CRITERIA: The specifications and acceptance/rejection criteria, such as acceptable quality level and unacceptable quality level, with an
Validation and Calibration Definitions and Terminology ACCEPTANCE CRITERIA: The specifications and acceptance/rejection criteria, such as acceptable quality level and unacceptable quality level, with an
Simple Linear Regression Inference
 Simple Linear Regression Inference 1 Inference requirements The Normality assumption of the stochastic term e is needed for inference even if it is not a OLS requirement. Therefore we have: Interpretation
Simple Linear Regression Inference 1 Inference requirements The Normality assumption of the stochastic term e is needed for inference even if it is not a OLS requirement. Therefore we have: Interpretation
BIOINF 525 Winter 2016 Foundations of Bioinformatics and Systems Biology http://tinyurl.com/bioinf525-w16
 Course Director: Dr. Barry Grant (DCM&B, [email protected]) Description: This is a three module course covering (1) Foundations of Bioinformatics, (2) Statistics in Bioinformatics, and (3) Systems
Course Director: Dr. Barry Grant (DCM&B, [email protected]) Description: This is a three module course covering (1) Foundations of Bioinformatics, (2) Statistics in Bioinformatics, and (3) Systems
Principles of Hypothesis Testing for Public Health
 Principles of Hypothesis Testing for Public Health Laura Lee Johnson, Ph.D. Statistician National Center for Complementary and Alternative Medicine [email protected] Fall 2011 Answers to Questions
Principles of Hypothesis Testing for Public Health Laura Lee Johnson, Ph.D. Statistician National Center for Complementary and Alternative Medicine [email protected] Fall 2011 Answers to Questions
Case Study in Data Analysis Does a drug prevent cardiomegaly in heart failure?
 Case Study in Data Analysis Does a drug prevent cardiomegaly in heart failure? Harvey Motulsky [email protected] This is the first case in what I expect will be a series of case studies. While I mention
Case Study in Data Analysis Does a drug prevent cardiomegaly in heart failure? Harvey Motulsky [email protected] This is the first case in what I expect will be a series of case studies. While I mention
CLINICAL TRIALS: Part 2 of 2
 CLINICAL TRIALS: Part 2 of 2 Lance K. Heilbrun, Ph.D., M.P.H. Professor of Medicine and Oncology Division of Hematology and Oncology Wayne State University School of Medicine Assistant Director, Biostatistics
CLINICAL TRIALS: Part 2 of 2 Lance K. Heilbrun, Ph.D., M.P.H. Professor of Medicine and Oncology Division of Hematology and Oncology Wayne State University School of Medicine Assistant Director, Biostatistics
Adequacy of Biomath. Models. Empirical Modeling Tools. Bayesian Modeling. Model Uncertainty / Selection
 Directions in Statistical Methodology for Multivariable Predictive Modeling Frank E Harrell Jr University of Virginia Seattle WA 19May98 Overview of Modeling Process Model selection Regression shape Diagnostics
Directions in Statistical Methodology for Multivariable Predictive Modeling Frank E Harrell Jr University of Virginia Seattle WA 19May98 Overview of Modeling Process Model selection Regression shape Diagnostics
Guide to Biostatistics
 MedPage Tools Guide to Biostatistics Study Designs Here is a compilation of important epidemiologic and common biostatistical terms used in medical research. You can use it as a reference guide when reading
MedPage Tools Guide to Biostatistics Study Designs Here is a compilation of important epidemiologic and common biostatistical terms used in medical research. You can use it as a reference guide when reading
Statistical Considerations for Phase II Trials
 Statistical Considerations for Phase II Trials Emily Van Meter, PhD Biostatistics Shared Resource Facility, Markey Cancer Center Assistant Professor, Division of Cancer Biostatistics University of Kentucky
Statistical Considerations for Phase II Trials Emily Van Meter, PhD Biostatistics Shared Resource Facility, Markey Cancer Center Assistant Professor, Division of Cancer Biostatistics University of Kentucky
Introduction to Statistics and Quantitative Research Methods
 Introduction to Statistics and Quantitative Research Methods Purpose of Presentation To aid in the understanding of basic statistics, including terminology, common terms, and common statistical methods.
Introduction to Statistics and Quantitative Research Methods Purpose of Presentation To aid in the understanding of basic statistics, including terminology, common terms, and common statistical methods.
Use advanced techniques for summary and visualization of complex data for exploratory analysis and presentation.
 MS Biostatistics MS Biostatistics Competencies Study Development: Work collaboratively with biomedical or public health researchers and PhD biostatisticians, as necessary, to provide biostatistical expertise
MS Biostatistics MS Biostatistics Competencies Study Development: Work collaboratively with biomedical or public health researchers and PhD biostatisticians, as necessary, to provide biostatistical expertise
Chapter 5 Analysis of variance SPSS Analysis of variance
 Chapter 5 Analysis of variance SPSS Analysis of variance Data file used: gss.sav How to get there: Analyze Compare Means One-way ANOVA To test the null hypothesis that several population means are equal,
Chapter 5 Analysis of variance SPSS Analysis of variance Data file used: gss.sav How to get there: Analyze Compare Means One-way ANOVA To test the null hypothesis that several population means are equal,
Section Format Day Begin End Building Rm# Instructor. 001 Lecture Tue 6:45 PM 8:40 PM Silver 401 Ballerini
 NEW YORK UNIVERSITY ROBERT F. WAGNER GRADUATE SCHOOL OF PUBLIC SERVICE Course Syllabus Spring 2016 Statistical Methods for Public, Nonprofit, and Health Management Section Format Day Begin End Building
NEW YORK UNIVERSITY ROBERT F. WAGNER GRADUATE SCHOOL OF PUBLIC SERVICE Course Syllabus Spring 2016 Statistical Methods for Public, Nonprofit, and Health Management Section Format Day Begin End Building
Course Course Name # Summer Courses DCS Clinical Research 5103 Questions & Methods CORE. Credit Hours. Course Description
 Course Course Name # Summer Courses Clinical Research 5103 Questions & Methods Credit Hours Course Description 1 Defining and developing a research question; distinguishing between correlative and mechanistic
Course Course Name # Summer Courses Clinical Research 5103 Questions & Methods Credit Hours Course Description 1 Defining and developing a research question; distinguishing between correlative and mechanistic
Likelihood: Frequentist vs Bayesian Reasoning
 "PRINCIPLES OF PHYLOGENETICS: ECOLOGY AND EVOLUTION" Integrative Biology 200B University of California, Berkeley Spring 2009 N Hallinan Likelihood: Frequentist vs Bayesian Reasoning Stochastic odels and
"PRINCIPLES OF PHYLOGENETICS: ECOLOGY AND EVOLUTION" Integrative Biology 200B University of California, Berkeley Spring 2009 N Hallinan Likelihood: Frequentist vs Bayesian Reasoning Stochastic odels and
NCSS Statistical Software Principal Components Regression. In ordinary least squares, the regression coefficients are estimated using the formula ( )
 Chapter 340 Principal Components Regression Introduction is a technique for analyzing multiple regression data that suffer from multicollinearity. When multicollinearity occurs, least squares estimates
Chapter 340 Principal Components Regression Introduction is a technique for analyzing multiple regression data that suffer from multicollinearity. When multicollinearity occurs, least squares estimates
Section 13, Part 1 ANOVA. Analysis Of Variance
 Section 13, Part 1 ANOVA Analysis Of Variance Course Overview So far in this course we ve covered: Descriptive statistics Summary statistics Tables and Graphs Probability Probability Rules Probability
Section 13, Part 1 ANOVA Analysis Of Variance Course Overview So far in this course we ve covered: Descriptive statistics Summary statistics Tables and Graphs Probability Probability Rules Probability
Statistical Rules of Thumb
 Statistical Rules of Thumb Second Edition Gerald van Belle University of Washington Department of Biostatistics and Department of Environmental and Occupational Health Sciences Seattle, WA WILEY AJOHN
Statistical Rules of Thumb Second Edition Gerald van Belle University of Washington Department of Biostatistics and Department of Environmental and Occupational Health Sciences Seattle, WA WILEY AJOHN
Package ERP. December 14, 2015
 Type Package Package ERP December 14, 2015 Title Significance Analysis of Event-Related Potentials Data Version 1.1 Date 2015-12-11 Author David Causeur (Agrocampus, Rennes, France) and Ching-Fan Sheu
Type Package Package ERP December 14, 2015 Title Significance Analysis of Event-Related Potentials Data Version 1.1 Date 2015-12-11 Author David Causeur (Agrocampus, Rennes, France) and Ching-Fan Sheu
Validation of measurement procedures
 Validation of measurement procedures R. Haeckel and I.Püntmann Zentralkrankenhaus Bremen The new ISO standard 15189 which has already been accepted by most nations will soon become the basis for accreditation
Validation of measurement procedures R. Haeckel and I.Püntmann Zentralkrankenhaus Bremen The new ISO standard 15189 which has already been accepted by most nations will soon become the basis for accreditation
Why Taking This Course? Course Introduction, Descriptive Statistics and Data Visualization. Learning Goals. GENOME 560, Spring 2012
 Why Taking This Course? Course Introduction, Descriptive Statistics and Data Visualization GENOME 560, Spring 2012 Data are interesting because they help us understand the world Genomics: Massive Amounts
Why Taking This Course? Course Introduction, Descriptive Statistics and Data Visualization GENOME 560, Spring 2012 Data are interesting because they help us understand the world Genomics: Massive Amounts
HYPOTHESIS TESTING WITH SPSS:
 HYPOTHESIS TESTING WITH SPSS: A NON-STATISTICIAN S GUIDE & TUTORIAL by Dr. Jim Mirabella SPSS 14.0 screenshots reprinted with permission from SPSS Inc. Published June 2006 Copyright Dr. Jim Mirabella CHAPTER
HYPOTHESIS TESTING WITH SPSS: A NON-STATISTICIAN S GUIDE & TUTORIAL by Dr. Jim Mirabella SPSS 14.0 screenshots reprinted with permission from SPSS Inc. Published June 2006 Copyright Dr. Jim Mirabella CHAPTER
Biostatistics Short Course Introduction to Longitudinal Studies
 Biostatistics Short Course Introduction to Longitudinal Studies Zhangsheng Yu Division of Biostatistics Department of Medicine Indiana University School of Medicine Zhangsheng Yu (Indiana University) Longitudinal
Biostatistics Short Course Introduction to Longitudinal Studies Zhangsheng Yu Division of Biostatistics Department of Medicine Indiana University School of Medicine Zhangsheng Yu (Indiana University) Longitudinal
Study Design. Date: March 11, 2003 Reviewer: Jawahar Tiwari, Ph.D. Ellis Unger, M.D. Ghanshyam Gupta, Ph.D. Chief, Therapeutics Evaluation Branch
 BLA: STN 103471 Betaseron (Interferon β-1b) for the treatment of secondary progressive multiple sclerosis. Submission dated June 29, 1998. Chiron Corp. Date: March 11, 2003 Reviewer: Jawahar Tiwari, Ph.D.
BLA: STN 103471 Betaseron (Interferon β-1b) for the treatment of secondary progressive multiple sclerosis. Submission dated June 29, 1998. Chiron Corp. Date: March 11, 2003 Reviewer: Jawahar Tiwari, Ph.D.
School of Public Health and Health Services Department of Epidemiology and Biostatistics
 School of Public Health and Health Services Department of Epidemiology and Biostatistics Master of Public Health and Graduate Certificate Biostatistics 0-04 Note: All curriculum revisions will be updated
School of Public Health and Health Services Department of Epidemiology and Biostatistics Master of Public Health and Graduate Certificate Biostatistics 0-04 Note: All curriculum revisions will be updated
Example: Credit card default, we may be more interested in predicting the probabilty of a default than classifying individuals as default or not.
 Statistical Learning: Chapter 4 Classification 4.1 Introduction Supervised learning with a categorical (Qualitative) response Notation: - Feature vector X, - qualitative response Y, taking values in C
Statistical Learning: Chapter 4 Classification 4.1 Introduction Supervised learning with a categorical (Qualitative) response Notation: - Feature vector X, - qualitative response Y, taking values in C
Molecular markers and clinical trial design parallels between oncology and rare diseases?
 Molecular markers and clinical trial design parallels between oncology and rare diseases?, Harriet Sommer Institute for Medical Biometry and Statistics, University of Freiburg Medical Center 6. Forum Patientennahe
Molecular markers and clinical trial design parallels between oncology and rare diseases?, Harriet Sommer Institute for Medical Biometry and Statistics, University of Freiburg Medical Center 6. Forum Patientennahe
CONTENTS OF DAY 2. II. Why Random Sampling is Important 9 A myth, an urban legend, and the real reason NOTES FOR SUMMER STATISTICS INSTITUTE COURSE
 1 2 CONTENTS OF DAY 2 I. More Precise Definition of Simple Random Sample 3 Connection with independent random variables 3 Problems with small populations 8 II. Why Random Sampling is Important 9 A myth,
1 2 CONTENTS OF DAY 2 I. More Precise Definition of Simple Random Sample 3 Connection with independent random variables 3 Problems with small populations 8 II. Why Random Sampling is Important 9 A myth,
Sample Size Planning, Calculation, and Justification
 Sample Size Planning, Calculation, and Justification Theresa A Scott, MS Vanderbilt University Department of Biostatistics [email protected] http://biostat.mc.vanderbilt.edu/theresascott Theresa
Sample Size Planning, Calculation, and Justification Theresa A Scott, MS Vanderbilt University Department of Biostatistics [email protected] http://biostat.mc.vanderbilt.edu/theresascott Theresa
Accelerating Development and Approval of Targeted Cancer Therapies
 Accelerating Development and Approval of Targeted Cancer Therapies Anna Barker, NCI David Epstein, Novartis Oncology Stephen Friend, Sage Bionetworks Cindy Geoghegan, Patient and Partners David Kessler,
Accelerating Development and Approval of Targeted Cancer Therapies Anna Barker, NCI David Epstein, Novartis Oncology Stephen Friend, Sage Bionetworks Cindy Geoghegan, Patient and Partners David Kessler,
Analytical Test Method Validation Report Template
 Analytical Test Method Validation Report Template 1. Purpose The purpose of this Validation Summary Report is to summarize the finding of the validation of test method Determination of, following Validation
Analytical Test Method Validation Report Template 1. Purpose The purpose of this Validation Summary Report is to summarize the finding of the validation of test method Determination of, following Validation
Biomarker Discovery and Data Visualization Tool for Ovarian Cancer Screening
 , pp.169-178 http://dx.doi.org/10.14257/ijbsbt.2014.6.2.17 Biomarker Discovery and Data Visualization Tool for Ovarian Cancer Screening Ki-Seok Cheong 2,3, Hye-Jeong Song 1,3, Chan-Young Park 1,3, Jong-Dae
, pp.169-178 http://dx.doi.org/10.14257/ijbsbt.2014.6.2.17 Biomarker Discovery and Data Visualization Tool for Ovarian Cancer Screening Ki-Seok Cheong 2,3, Hye-Jeong Song 1,3, Chan-Young Park 1,3, Jong-Dae
Introduction to Data Mining
 Introduction to Data Mining 1 Why Data Mining? Explosive Growth of Data Data collection and data availability Automated data collection tools, Internet, smartphones, Major sources of abundant data Business:
Introduction to Data Mining 1 Why Data Mining? Explosive Growth of Data Data collection and data availability Automated data collection tools, Internet, smartphones, Major sources of abundant data Business:
Multivariate Normal Distribution
 Multivariate Normal Distribution Lecture 4 July 21, 2011 Advanced Multivariate Statistical Methods ICPSR Summer Session #2 Lecture #4-7/21/2011 Slide 1 of 41 Last Time Matrices and vectors Eigenvalues
Multivariate Normal Distribution Lecture 4 July 21, 2011 Advanced Multivariate Statistical Methods ICPSR Summer Session #2 Lecture #4-7/21/2011 Slide 1 of 41 Last Time Matrices and vectors Eigenvalues
Testing Multiple Secondary Endpoints in Confirmatory Comparative Studies -- A Regulatory Perspective
 Testing Multiple Secondary Endpoints in Confirmatory Comparative Studies -- A Regulatory Perspective Lilly Yue, Ph.D. Chief, Cardiovascular and Ophthalmic Devices Branch Division of Biostatistics CDRH/FDA
Testing Multiple Secondary Endpoints in Confirmatory Comparative Studies -- A Regulatory Perspective Lilly Yue, Ph.D. Chief, Cardiovascular and Ophthalmic Devices Branch Division of Biostatistics CDRH/FDA
BIOS 6660: Analysis of Biomedical Big Data Using R and Bioconductor, Fall 2015 Computer Lab: Education 2 North Room 2201DE (TTh 10:30 to 11:50 am)
 BIOS 6660: Analysis of Biomedical Big Data Using R and Bioconductor, Fall 2015 Computer Lab: Education 2 North Room 2201DE (TTh 10:30 to 11:50 am) Course Instructor: Dr. Tzu L. Phang, Assistant Professor
BIOS 6660: Analysis of Biomedical Big Data Using R and Bioconductor, Fall 2015 Computer Lab: Education 2 North Room 2201DE (TTh 10:30 to 11:50 am) Course Instructor: Dr. Tzu L. Phang, Assistant Professor
AVOIDING BIAS AND RANDOM ERROR IN DATA ANALYSIS
 AVOIDING BIAS AND RANDOM ERROR IN DATA ANALYSIS Susan Ellenberg, Ph.D. Perelman School of Medicine University of Pennsylvania School of Medicine FDA Clinical Investigator Course White Oak, MD November
AVOIDING BIAS AND RANDOM ERROR IN DATA ANALYSIS Susan Ellenberg, Ph.D. Perelman School of Medicine University of Pennsylvania School of Medicine FDA Clinical Investigator Course White Oak, MD November
STATISTICA Formula Guide: Logistic Regression. Table of Contents
 : Table of Contents... 1 Overview of Model... 1 Dispersion... 2 Parameterization... 3 Sigma-Restricted Model... 3 Overparameterized Model... 4 Reference Coding... 4 Model Summary (Summary Tab)... 5 Summary
: Table of Contents... 1 Overview of Model... 1 Dispersion... 2 Parameterization... 3 Sigma-Restricted Model... 3 Overparameterized Model... 4 Reference Coding... 4 Model Summary (Summary Tab)... 5 Summary
Planning sample size for randomized evaluations
 TRANSLATING RESEARCH INTO ACTION Planning sample size for randomized evaluations Simone Schaner Dartmouth College povertyactionlab.org 1 Course Overview Why evaluate? What is evaluation? Outcomes, indicators
TRANSLATING RESEARCH INTO ACTION Planning sample size for randomized evaluations Simone Schaner Dartmouth College povertyactionlab.org 1 Course Overview Why evaluate? What is evaluation? Outcomes, indicators
One-Way Analysis of Variance (ANOVA) Example Problem
 One-Way Analysis of Variance (ANOVA) Example Problem Introduction Analysis of Variance (ANOVA) is a hypothesis-testing technique used to test the equality of two or more population (or treatment) means
One-Way Analysis of Variance (ANOVA) Example Problem Introduction Analysis of Variance (ANOVA) is a hypothesis-testing technique used to test the equality of two or more population (or treatment) means
MTH 140 Statistics Videos
 MTH 140 Statistics Videos Chapter 1 Picturing Distributions with Graphs Individuals and Variables Categorical Variables: Pie Charts and Bar Graphs Categorical Variables: Pie Charts and Bar Graphs Quantitative
MTH 140 Statistics Videos Chapter 1 Picturing Distributions with Graphs Individuals and Variables Categorical Variables: Pie Charts and Bar Graphs Categorical Variables: Pie Charts and Bar Graphs Quantitative
Two-Way ANOVA tests. I. Definition and Applications...2. II. Two-Way ANOVA prerequisites...2. III. How to use the Two-Way ANOVA tool?...
 Two-Way ANOVA tests Contents at a glance I. Definition and Applications...2 II. Two-Way ANOVA prerequisites...2 III. How to use the Two-Way ANOVA tool?...3 A. Parametric test, assume variances equal....4
Two-Way ANOVA tests Contents at a glance I. Definition and Applications...2 II. Two-Way ANOVA prerequisites...2 III. How to use the Two-Way ANOVA tool?...3 A. Parametric test, assume variances equal....4
Data Mining and Machine Learning in Bioinformatics
 Data Mining and Machine Learning in Bioinformatics PRINCIPAL METHODS AND SUCCESSFUL APPLICATIONS Ruben Armañanzas http://mason.gmu.edu/~rarmanan Adapted from Iñaki Inza slides http://www.sc.ehu.es/isg
Data Mining and Machine Learning in Bioinformatics PRINCIPAL METHODS AND SUCCESSFUL APPLICATIONS Ruben Armañanzas http://mason.gmu.edu/~rarmanan Adapted from Iñaki Inza slides http://www.sc.ehu.es/isg
Consider a study in which. How many subjects? The importance of sample size calculations. An insignificant effect: two possibilities.
 Consider a study in which How many subjects? The importance of sample size calculations Office of Research Protections Brown Bag Series KB Boomer, Ph.D. Director, [email protected] A researcher conducts
Consider a study in which How many subjects? The importance of sample size calculations Office of Research Protections Brown Bag Series KB Boomer, Ph.D. Director, [email protected] A researcher conducts
BASIC COURSE IN STATISTICS FOR MEDICAL DOCTORS, SCIENTISTS & OTHER RESEARCH INVESTIGATORS INFORMATION BROCHURE
 INDIAN COUNCIL OF MEDICAL RESEARCH BASIC COURSE IN STATISTICS FOR MEDICAL DOCTORS, SCIENTISTS & OTHER RESEARCH INVESTIGATORS October 13 th 17 th 2014 INFORMATION BROCHURE COURSE OFFERED BY Division of
INDIAN COUNCIL OF MEDICAL RESEARCH BASIC COURSE IN STATISTICS FOR MEDICAL DOCTORS, SCIENTISTS & OTHER RESEARCH INVESTIGATORS October 13 th 17 th 2014 INFORMATION BROCHURE COURSE OFFERED BY Division of
Protein Protein Interaction Networks
 Functional Pattern Mining from Genome Scale Protein Protein Interaction Networks Young-Rae Cho, Ph.D. Assistant Professor Department of Computer Science Baylor University it My Definition of Bioinformatics
Functional Pattern Mining from Genome Scale Protein Protein Interaction Networks Young-Rae Cho, Ph.D. Assistant Professor Department of Computer Science Baylor University it My Definition of Bioinformatics
Unit 31 A Hypothesis Test about Correlation and Slope in a Simple Linear Regression
 Unit 31 A Hypothesis Test about Correlation and Slope in a Simple Linear Regression Objectives: To perform a hypothesis test concerning the slope of a least squares line To recognize that testing for a
Unit 31 A Hypothesis Test about Correlation and Slope in a Simple Linear Regression Objectives: To perform a hypothesis test concerning the slope of a least squares line To recognize that testing for a
Fairfield Public Schools
 Mathematics Fairfield Public Schools AP Statistics AP Statistics BOE Approved 04/08/2014 1 AP STATISTICS Critical Areas of Focus AP Statistics is a rigorous course that offers advanced students an opportunity
Mathematics Fairfield Public Schools AP Statistics AP Statistics BOE Approved 04/08/2014 1 AP STATISTICS Critical Areas of Focus AP Statistics is a rigorous course that offers advanced students an opportunity
Logistic Regression (1/24/13)
 STA63/CBB540: Statistical methods in computational biology Logistic Regression (/24/3) Lecturer: Barbara Engelhardt Scribe: Dinesh Manandhar Introduction Logistic regression is model for regression used
STA63/CBB540: Statistical methods in computational biology Logistic Regression (/24/3) Lecturer: Barbara Engelhardt Scribe: Dinesh Manandhar Introduction Logistic regression is model for regression used
How To Check For Differences In The One Way Anova
 MINITAB ASSISTANT WHITE PAPER This paper explains the research conducted by Minitab statisticians to develop the methods and data checks used in the Assistant in Minitab 17 Statistical Software. One-Way
MINITAB ASSISTANT WHITE PAPER This paper explains the research conducted by Minitab statisticians to develop the methods and data checks used in the Assistant in Minitab 17 Statistical Software. One-Way
COMPARISONS OF CUSTOMER LOYALTY: PUBLIC & PRIVATE INSURANCE COMPANIES.
 277 CHAPTER VI COMPARISONS OF CUSTOMER LOYALTY: PUBLIC & PRIVATE INSURANCE COMPANIES. This chapter contains a full discussion of customer loyalty comparisons between private and public insurance companies
277 CHAPTER VI COMPARISONS OF CUSTOMER LOYALTY: PUBLIC & PRIVATE INSURANCE COMPANIES. This chapter contains a full discussion of customer loyalty comparisons between private and public insurance companies
Analysis and Interpretation of Clinical Trials. How to conclude?
 www.eurordis.org Analysis and Interpretation of Clinical Trials How to conclude? Statistical Issues Dr Ferran Torres Unitat de Suport en Estadística i Metodología - USEM Statistics and Methodology Support
www.eurordis.org Analysis and Interpretation of Clinical Trials How to conclude? Statistical Issues Dr Ferran Torres Unitat de Suport en Estadística i Metodología - USEM Statistics and Methodology Support
Gene expression analysis. Ulf Leser and Karin Zimmermann
 Gene expression analysis Ulf Leser and Karin Zimmermann Ulf Leser: Bioinformatics, Wintersemester 2010/2011 1 Last lecture What are microarrays? - Biomolecular devices measuring the transcriptome of a
Gene expression analysis Ulf Leser and Karin Zimmermann Ulf Leser: Bioinformatics, Wintersemester 2010/2011 1 Last lecture What are microarrays? - Biomolecular devices measuring the transcriptome of a
Measurement Systems Correlation MSC for Suppliers
 Measurement Systems Correlation MSC for Suppliers Copyright 2003-2007 Raytheon Company. All rights reserved. R6σ is a Raytheon trademark registered in the United States and Europe. Raytheon Six Sigma is
Measurement Systems Correlation MSC for Suppliers Copyright 2003-2007 Raytheon Company. All rights reserved. R6σ is a Raytheon trademark registered in the United States and Europe. Raytheon Six Sigma is
Data Mining Techniques Chapter 5: The Lure of Statistics: Data Mining Using Familiar Tools
 Data Mining Techniques Chapter 5: The Lure of Statistics: Data Mining Using Familiar Tools Occam s razor.......................................................... 2 A look at data I.........................................................
Data Mining Techniques Chapter 5: The Lure of Statistics: Data Mining Using Familiar Tools Occam s razor.......................................................... 2 A look at data I.........................................................
August 2012 EXAMINATIONS Solution Part I
 August 01 EXAMINATIONS Solution Part I (1) In a random sample of 600 eligible voters, the probability that less than 38% will be in favour of this policy is closest to (B) () In a large random sample,
August 01 EXAMINATIONS Solution Part I (1) In a random sample of 600 eligible voters, the probability that less than 38% will be in favour of this policy is closest to (B) () In a large random sample,
Study Guide for the Final Exam
 Study Guide for the Final Exam When studying, remember that the computational portion of the exam will only involve new material (covered after the second midterm), that material from Exam 1 will make
Study Guide for the Final Exam When studying, remember that the computational portion of the exam will only involve new material (covered after the second midterm), that material from Exam 1 will make
A POPULATION MEAN, CONFIDENCE INTERVALS AND HYPOTHESIS TESTING
 CHAPTER 5. A POPULATION MEAN, CONFIDENCE INTERVALS AND HYPOTHESIS TESTING 5.1 Concepts When a number of animals or plots are exposed to a certain treatment, we usually estimate the effect of the treatment
CHAPTER 5. A POPULATION MEAN, CONFIDENCE INTERVALS AND HYPOTHESIS TESTING 5.1 Concepts When a number of animals or plots are exposed to a certain treatment, we usually estimate the effect of the treatment
Statistics I for QBIC. Contents and Objectives. Chapters 1 7. Revised: August 2013
 Statistics I for QBIC Text Book: Biostatistics, 10 th edition, by Daniel & Cross Contents and Objectives Chapters 1 7 Revised: August 2013 Chapter 1: Nature of Statistics (sections 1.1-1.6) Objectives
Statistics I for QBIC Text Book: Biostatistics, 10 th edition, by Daniel & Cross Contents and Objectives Chapters 1 7 Revised: August 2013 Chapter 1: Nature of Statistics (sections 1.1-1.6) Objectives
Chapter 9. Two-Sample Tests. Effect Sizes and Power Paired t Test Calculation
 Chapter 9 Two-Sample Tests Paired t Test (Correlated Groups t Test) Effect Sizes and Power Paired t Test Calculation Summary Independent t Test Chapter 9 Homework Power and Two-Sample Tests: Paired Versus
Chapter 9 Two-Sample Tests Paired t Test (Correlated Groups t Test) Effect Sizes and Power Paired t Test Calculation Summary Independent t Test Chapter 9 Homework Power and Two-Sample Tests: Paired Versus
6: Introduction to Hypothesis Testing
 6: Introduction to Hypothesis Testing Significance testing is used to help make a judgment about a claim by addressing the question, Can the observed difference be attributed to chance? We break up significance
6: Introduction to Hypothesis Testing Significance testing is used to help make a judgment about a claim by addressing the question, Can the observed difference be attributed to chance? We break up significance
The Product Review Life Cycle A Brief Overview
 Stat & Quant Mthds Pharm Reg (Spring 2, 2014) Lecture 2,Week 1 1 The review process developed over a 40 year period and has been influenced by 5 Prescription User Fee Act renewals Time frames for review
Stat & Quant Mthds Pharm Reg (Spring 2, 2014) Lecture 2,Week 1 1 The review process developed over a 40 year period and has been influenced by 5 Prescription User Fee Act renewals Time frames for review
Comparison of Non-linear Dimensionality Reduction Techniques for Classification with Gene Expression Microarray Data
 CMPE 59H Comparison of Non-linear Dimensionality Reduction Techniques for Classification with Gene Expression Microarray Data Term Project Report Fatma Güney, Kübra Kalkan 1/15/2013 Keywords: Non-linear
CMPE 59H Comparison of Non-linear Dimensionality Reduction Techniques for Classification with Gene Expression Microarray Data Term Project Report Fatma Güney, Kübra Kalkan 1/15/2013 Keywords: Non-linear
Data Integration. Lectures 16 & 17. ECS289A, WQ03, Filkov
 Data Integration Lectures 16 & 17 Lectures Outline Goals for Data Integration Homogeneous data integration time series data (Filkov et al. 2002) Heterogeneous data integration microarray + sequence microarray
Data Integration Lectures 16 & 17 Lectures Outline Goals for Data Integration Homogeneous data integration time series data (Filkov et al. 2002) Heterogeneous data integration microarray + sequence microarray
Elements of statistics (MATH0487-1)
 Elements of statistics (MATH0487-1) Prof. Dr. Dr. K. Van Steen University of Liège, Belgium December 10, 2012 Introduction to Statistics Basic Probability Revisited Sampling Exploratory Data Analysis -
Elements of statistics (MATH0487-1) Prof. Dr. Dr. K. Van Steen University of Liège, Belgium December 10, 2012 Introduction to Statistics Basic Probability Revisited Sampling Exploratory Data Analysis -
