If you didn't know this was an implant
|
|
|
- Gregory Poole
- 8 years ago
- Views:
Transcription
1 If you didn't know this was an implant New Perspectives. Annual Report 2010.
2 Synthes. LCP Annual Compact Report Hand 2010 you d think it was jewelry. Business Report BR15
3 Spinal Cages To us, no opportunity to contribute to the quality of life is too small. Our products are true gems donned for health.
4 MatrixMIDFACE Although it may sound a bit old fashioned: we like to crown our customers king and queen to be treated with royal dignity.
5 X-Plate and Navicular Plate To innovate high-quality products, we listen carefully to surgeons and OR personnel alike. Our business fundamentals produce elegant simplicity.
6 StenoFix Real innovation often sets off an entire chain: An idea Development Improvement Striving for true excellence.
7 MatrixORBITAL Day in and day out, we search for true clinical benefits and added medical value for surgeons and patients alike. Sometimes the end result is curvaceous beauty.
8 LCP Compact Hand The LCP Compact Hand system consists of different sized stainless steel or titanium plates and screws that are used for the fixation of fractures in the fingers and the mid-hand. The stable fixation achieved with these implants allows early mobilization, which is necessary for the fracture and soft tissue structures to heal. X-Plate and Navicular Plate Foot surgery is complex and often requires metal implants. X-plates and navicular plates are two examples of plates used for the surgical fixation of fractures, but also for connecting bones that have been detached by the surgeon in order to improve the functionality or appearance of the foot. Spinal Cages Cages are used as a placeholder for discs that have been surgically removed because they protrude and impinge upon nerve roots (disc herniation). This causes significant pain and therefore justifies a surgical removal. The type of cage chosen depends on the surgical entry point and the approach the surgeon chooses for a specific intervention. StenoFix StenoFix is an interspinous spacer to be used after surgical decompression of the spine. The device is indicated for moderate to severe spinal lumbar stenosis and is used as a spaceholder between the spinous processes to help treat pain that arises when standing upright or extending. StenoFix controls the segmental extension and distracts the interspinous space. It can be implanted at one or two lumbar levels from L1 to S1. MatrixMIDFACE The MatrixMIDFACE system is part of a whole new plating platform for internal fixation of the craniomaxillofacial skeleton. It is used for trauma and reconstructive surgery of the midface. The titanium plates are available in four thicknesses and color-coded according to their strength. A single screw diameter works for the entire system and all plates so the system is easy to use, but still flexible. MatrixORBITAL These thin preformed plates are used for the surgical fixation of fractures of the orbital base (the bony area underneath the eyeball). The titanium plates are preformed in a threedimensional shape to avoid any bending or cutting by the surgeon during surgery.
9 Significant Facts Sales per region 2010 (US$ millions) North America 2,154.2 Europe Asia Pacific Rest of World ,687.0 Asia Pacific 11.5% Rest of World 7.0% Europe 23.1% North America 58.4% Operating Expenses (US$ millions) Selling and Promotion ,080.1 % of Net Sales 29.5% 29.1% 29.3% 28.8% 29.3% General and Administrative % of Net Sales 11.1% 10.7% 10.9% 11.3% 10.7% Research and Development % of Net Sales 5.3% 5.4% 5.3% 5.0% 4.7% US$ millions 1,100 1, Selling and Promotion General and Administrative Research and Development Profitability (US$ millions) Net Sales 2, , , , ,687.0 Gross Profit Margin 81.9% 81.0% 82.7% 82.6% 82.6% Operating Income Margin 31.9% 32.8% 33.8% 34.3% 34.8% Net Earnings Margin 21.3% 22.2% 23.0% 24.3% 24.6% US$ millions 4,000 3,500 3,000 2,500 2,000 1,500 1, Net Sales Gross Profit Operating Income Net Earnings
10 FCF and Capital Expenditure (US$ millions) Free Cashflow CapEx US$ millions Free Cashflow CapEx Assets 2010 (US$ millions) Current Assets 3,414.9 Property, Plant & Equipment Intangible Assets 2,119.3 Goodwill 1,293.1 Other Assets Other Assets 2.6% Goodwill 16.3% Current Assets 43.1% Intangible Assets 26.7% Property, Plant & Equipment 11.3% Liabilities and Equity 2010 (US$ millions) Current Liabilities Long-term Liabilities Retained Earnings 3,925.2 Other Equity 2,813.5 Other Equity 35.5% Current Liabilities 7.0% Long-term Liabilities 8.0% Retained Earnings 49.5% Employees Total Employees (at year-end) 8,451 9,070 9,947 10,705 11,426 Change in Employees (%) 11% 7% 10% 8% 7% Sales Force (at year-end) 2,134 2,446 2,940 3,319 3,636 Sales Force as % of Total Employees 25% 27% 30% 31% 32% 16,000 16% 14,000 14% 12,000 12% 10,000 8,000 10% 8% 6,000 6% 4,000 4% 2,000 2% 0 0% Total Employees Sales Force Change in Employees (%)
11 Synthes. Annual Report 2010 Letter to Shareholders BR2 Company Profile BR4 Financial Highlights BR4 Global Presence BR5 Business Report Regional Highlights BR7 Corporate Citizenship Educational Initiatives BR12 Employees BR13 Cultivating Synthes Culture of Integrity BR14 Product Highlights Overview Product Groups BR16 Trauma. Variable Angle LCP Forefoot/Midfoot System BR17 Spine. MATRIX BR18 Cranio-Maxillofacial. MatrixORTHOGNATHIC Plating System BR19 Corporate Governance Group Structure and Shareholders CG2 Capital Structure CG3 Board of Directors CG4 Group Management Committee CG9 Compensation, Shareholdings and Loans CG10 Shareholders Participation CG12 Changes of Control and Defense Measures CG14 Auditors CG15 Information Policy CG16 Report of Independent Auditors FR2 Report of Independent Accountants FR3 Consolidated Balance Sheets FR4 Consolidated Statements of Operations FR6 Consolidated Statements of Changes in Stockholders Equity FR7 Consolidated Statements of Cash Flows FR8 Notes to the Consolidated Financial Statements FR10 Note to Directors and Shareholders FR47 Investor Key Data FR48 Business Report BR1
12 Business Report Letter to Shareholders Dear Co-Shareholders and Friends of Synthes In 2010, Synthes sales grew by 7.5% and net earnings by 8.5% (both in constant currency). We launched over fifty new products, created close to 500 new jobs and strengthened our organization in order to master the challenges that lie ahead. As every year, this report provides an overview of the results of the remarkable work undertaken by the more than 11,000 employees of Synthes as well as that of our distributors and partners around the world during a dynamic Strong financial performance despite a challenging year in global orthopaedics 2010 was a year full of challenges and change for both our industry and our company. In that context, Synthes performance and the progress made in most key financial metrics as well as in all major operational activities and all parts of the world are evidence of the strength and resilience of our company. Fortunately, we have the privilege of being deeply integrated into major trauma centers and university hospitals around the globe, with many surgeons using our products on a daily basis. This gave us a wonderful opportunity once again to make a meaningful contribution to improving surgical patient care. We have gained market share in the global Trauma, Cranio-Maxillofacial (CMF) and Power Tools markets, but we also had to cope with changing market conditions in our Spine division with pressure on prices and volume. However, we have finally been able to launch MATRIX Spine, a much-needed new fixation system for the lumbar spine, used in the surgical treatment of back pain and deformity. Synthes sales growth of 7.5% (in constant currency) in markets that are growing more slowly than in the past is testament to the strength of the Synthes brand name, as well as the agility with which we work in sales and in product development. The strength of the Swiss franc and our relatively high share of Swissbased manufacturing operations did not materially impact our financials. Because of cost containment and lower legal expenses, the operating income margin increased by 50 basis points to 34.8%. The tax rate was reduced to 27.7%. All of this resulted in an increase in earnings per share to US$ 7.65 (an increase of 8.5% in constant currency). Synthes is known for being conservative and for acting with a longterm perspective. This was proven again in 2010 by prudently adding to our employee base, which grew by 6.7% (including the acquisition of Anspach in November). In addition, we ended the year with a strong cash balance and the Board of Directors decided to increase the dividend from CHF 1.35 to CHF 1.80 and the payout ratio from 18% to 25%. Highlights from 2010 In May, we formally inaugurated our new manufacturing facility in Suzhou, China, in the presence of local and national representatives from the Chinese government. Production by this small factory will primarily be for the Chinese market and the surrounding area. The facility also comprises a training and product development center. Approval to sell products in China is expected in 2012 following completion of ongoing clinical studies. BR2 Business Report
13 Synthes. Annual Report 2010 In the fourth quarter of 2010, we acquired Anspach, a privately-held company specializing in the development, manufacturing, sale and servicing of high-precision power tools. This was a highly complementary addition to our existing portfolio and organization in power tools and will therefore allow us to rapidly gain market share. We expect double-digit growth in this market that has a value of approximately US$ 1.2 billion. The Board of Directors of Synthes is actively involved in the strategic direction of our company. Dr. Roland Brönnimann retired at the AGM in April 2010 after ten years at Synthes, seven of which serving on the Board. We would once again like to thank him for his valuable service. Daniel Eicher was elected to the Board at the 2010 AGM. He brings us wide-ranging experience on various boards of large Swiss organizations. In 2011, we will consolidate our leadership position in the markets we serve by improving customer experience of our products for the longterm. We will build on the strengths of Trauma, CMF and Power Tools globally, while continuing to address Spine s performance with new product launches. Overall, we expect the market environment to remain fairly challenging and we will continue to counteract with further advances in our productivity gain initiatives to maintain our strong profitability. Cordially, Surgeon education and outlook Much of the success of our company would not be possible without the relationship we have with the AO Foundation, our partner in education for surgeon and operating room personnel. The AO made significant advances in the way that it provided education in 2010, and we expect to continue to benefit from this fruitful collaboration which is now in its 53rd year. Nowadays, if a surgeon wants to attend a course, he or she would probably opt to prepare using elearning or online resources, including videos. The AO (and also Synthes) is therefore adopting a blended learning approach, to address the learner s needs with state-of-the-art technology. Hansjörg Wyss Chairman of the Board Michel Orsinger President and CEO Business Report BR3
14 Business Report Company Profile. Synthes, headquartered in West Chester, PA (USA), is a leading global medical device company, employing over 11,400 people whose mission is to improve patient care around the world. Bone work from head to toe Through its five product groups (Trauma, Spine, Cranio-Maxillofacial, Biomaterials and Power Tools), Synthes develops, produces and markets instruments, implants and biomaterials for the surgical fixation, correction and regeneration of the human skeleton and its soft tissues. We operate in product markets with high growth, driven by the aging population and improvements in technology that enable treatment of more patients with better implants. Present on every continent Our staff works every day toward meeting the needs of surgeons, operating room personnel and patients. We stand for high quality, constant innovation and consistent customer orientation. Over 40 subsidiaries and around 50 distributors are managed by four area head offices: West Chester (USA) for North America; Solothurn (Switzerland) for Europe, Middle East & Africa; Sydney (Australia) for Asia/ Pacific and Miami (USA) for Latin America. We run 13 manufacturing facilities, mainly located in the United States and Switzerland. Reaching for the sky Synthes has set the standard for treating fractures. We are the world leader in traumatology; we rank among the top companies for spinal devices and we are in the leading position in the cranio-maxillofacial business. Synthes is an innovative pioneer in the field of biomaterials such as resorbable implants, bone graft substitutes and coated materials. Synthes also offers a wide range of power tools for surgical use, a business unit that was recently enhanced by a selection of high- speed devices with the acquisition of Anspach in All in all, we offer a comprehensive product portfolio coupled with excellent service through our technical sales force and state-of-the-art educational programs to secure us a leading position for the future. Striving for innovation Synthes establishes the foundations for its excellent market position by continuously developing better solutions. Our goal is to provide the safest and most advanced implants, instruments and technologies that ensure reliable operating procedures, rapid recovery and a pain-free life after surgery. We guarantee high quality, constant innovation and total concentration on the needs of our customers. Guided by our Values & Principles Synthes position as a leading global medical device company is based on our mission of improving patient care and our long-standing threepillar strategy, comprising innovation, sales force expansion and education. Our seven Values & Principles Patient Driven, Surgeon Focus, Innovation, Quality, Education, Partnership and Integrity continuously guide our strategies and actions. Financial facts at a glance In 2010, Synthes generated revenues of US$ 3.7 billion and net earnings of US$ 908 million. Over the last 10 years ( ), Synthes has increased its revenues from US$ 0.9 billion to US$ 3.7 billion, representing a CAGR of 15%, including the acquisition of Mathys in Financial Comparison 2010 vs Net sales in US$ 3,687.0 million 3,394.7 million Sales increase in US$ / local currency 8.6% / 7.5% 6.3 % / 8.9% Gross profit margin 82.6% 82.6% Operating income margin 34.8% 34.3% Net earnings margin 24.6% 24.3% Earnings per share in US$ Free cash flow in US$ million million Approved dividend in CHF Employees at year-end 11,426 10,705 SIX Swiss Exchange Symbol: SYST.VX ISIN: US87162M4096 Market capitalization at year-end 2010: CHF 14,995,969,691 BR4 Business Report
15 Synthes. Annual Report 2010 Global Presence SD Elmira, Horseheads NY VT MA FR DE Umkirch Tuttlingen UT WY Denver Monument CO NE KS PA Brandywine, West Chester West Chester WV MD DE New York NJ CT Balsthal Bettlach/ Grenchen Raron Hägendorf Solothurn Bern CH LI Mezzovico AT Salzburg AZ NM OK VA IT TX Locations U.S.A. Locations Europe NC AL GA SC Suzhou Shanghai CN FL Palm Beach Gardens Hong Kong TW Miami VN LA Locations China Legend Affiliates Distributors Global Corporate Headquarters European Headquarters Manufacturing Sites Business Report BR5
16 Business Report Business Report Regional Highlights BR7 BR6 Business Report
17 Synthes. Annual Report 2010 Business Report. Regional Highlights. Synthes has four reporting regions. North America accounted for almost 60% of global sales, while Europe and the emerging markets of Asia Pacific and Latin America are slowly catching up thanks to our daily contribution to better patient care. Sales per region 2010 (US$ millions) North America 2,154.2 Europe Asia Pacific Rest of World Asia Pacific 11.5% Rest of World 7.0% Europe 23.1% North America 58.4% North America North America attained local currency sales growth of 4.3% for the full year 2010, a result of mixed performances in the various divisons. On the one hand, in Trauma and CMF, Synthes grew at a high singledigit and low double-digit percent rate, respectively, and was able to use its outstanding market position to its benefit in terms of comprehensive product offering and the best-educated and technically proficient sales force. This led to market share gains in these two divisions. On the other hand, the company had to cope with negative growth in Spine, due to market-specific reasons but also to internal reasons related to product-lifecycle management. Strong Trauma position enhanced in 2010 In Trauma, we continued to successfully fight pricing pressures seen in an increasing demand for discounts and an increased rate of discounts offered by the competition. We have a premium offering that justifies a premium price. This is evident from our over 50% market share, which has helped us significantly in our positioning and in our collaboration with our hospital customers. We have a broad product portfolio with unique features and the biggest and best-educated sales force, coupled with a leading educational offering that plays an important role in the decision-making process. We again launched over thirty new Trauma products, mostly expansions of our successful LCP product lines that are now available with variable screw angulations to make the placement of these plate-andscrew constructs even more intuitive and much simpler. In addition, we have launched a large set of implants and instruments for orthopaedic foot interventions, called the Forefoot/Midfoot system, a product line that is important to us and provides an even greater offering to surgeons who operate on feet on an outpatient basis and therefore have historically not been served as much by our sales force as hospital-based surgeons. To address this need, we will start to build a dedicated team of sales force members in 2011 to exclusively take care of these surgeons mainly in outpatient surgery centers. A challenging year for Spine In Spine, Synthes was not immune to the mounting pressures from the U.S. market. Payors and regulators are paying more attention to the volume of spine surgeries being performed and reimbursement has become harder. Insurance companies are tightening the criteria that have to be met for certain types of spinal conditions before agreeing to reimburse for a procedure, particularly spinal fusion with implants. Furthermore, in view of the recent financial crisis and associated job losses, patients are less willing to take time off work for elective surgery or are unable to come up with shared payment or deductible amounts. They tolerate the pain and delay or cancel surgery. In addition, prices have come under downward pressure. Hospitals are starting to dictate prices (at reduced levels) and competitive forces are leading to a single-digit percent decline in the overall price level. Nevertheless, we are seeing some signs of recovery. Our long-awaited launch of MATRIX, a comprehensive pedicle screw fixation system for the lumbar spine (for deformity and for the treatment of lower back pain due to spinal degeneration), began in November in the U.S. and will start in the first quarter of 2011 in Europe and other regions of the world. We feel strongly about MATRIX and will be able to gain back market share that we may have lost over the past few quarters in this important segment in Spine. Together with MATRIX we have also launched T-Pal, a new cage (i.e. placeholder for the disc space after removal of the diseased disc) which allows minimally invasive treatments, a clear trend in the future. In 2011, we will enter the vertebroplasty market with a new cement and delivery system called Vertecem. Vertebroplasties are reconstructions of the vertebral bodies after fractures due to osteoporosis, a growing problem within conditions, sometimes referred to as the aging spine. Business Report BR7
18 Business Report Extraordinary growth in CMF In CMF, growth in 2010 was primarily achieved through the continued expansion of the Matrix Plating Systems, the targeted launch of the MatrixRIB System (for the fixation of rib fractures) and a fixation device to be used in primary sternal closures, i.e. for patients who have a high risk of complications. Our recent creation of the Thorax segment has proven to be successful as evidenced by the fact that this is now our fourth-largest product group within CMF (behind Neuro, Midface and Mandible). The Thorax portfolio includes sternal closure devices (used to fix the sternum after open heart surgeries) and a plating system to fix fractures of the ribs. decades. The settlement with the U.S. government related to the Norian case reinforces our compliance work and we are already vigorously enforcing these rules and regulations, which every member of our team has to abide by. It will make us an even better organization and be valuable beyond the requirements that are part of the settlement. The acquisition of the power tools company Anspach is an excellent addition to our product offering as it brings a complementary product line to the Synthes power tools portfolio. Anspach has a state-ofthe-art service facility and training center and provides us with highspeed power tools used in neurosurgery, ear, nose and throat (ENT) and spinal surgery. In addition, our partnership with Kensey Nash has yielded success. Under the partnership, which was initiated in 2009, Kensey Nash are processing Synthes porcine dermis product, XCM. This acellular dermis is used for soft tissue repair, e.g. as a suture-patch for hernia repair, and we are well positioned to gain market share in this relatively young market. In 2011, we will continue to launch new products within CMF, with a focus on the neuro and midface market. Key initiatives undertaken in 2010 For the U.S. market, the establishment of our Metro Centers has proven to be successful as it reduces required investment into field equipment. Metro Centers are logistics offices that store and manage Synthes surgical instrument and implant sets and dispatch and allocate them as efficiently as possible. The number of sites was expanded within Trauma but also established within the Spine sales organization: there are now almost 40 centers scattered around the U.S. and we will add another 15 by the end of Metro Centers empower our field team to deliver better service and response times. Synthes is always on top of new technologies. The first Synthes iphone and ipad application was launched in 2010 and features a whole array of tools suited to best fit the need of surgeons and supports our sales force in doing their everyday jobs. The Synthes app features product technique guides, surgical approach videos, presentations, and much more. Content update is easily managed within the application. Sales force additions over the last few years have provided us with a unique position with a dense network of technical sales specialists. We will focus on refining territory coverage in 2011 (and beyond) to provide excellent support for the use of our products and to maintain the high level of service that we have achieved over the last several Europe, Middle East and Africa The EMEA region s sales growth of almost 13% (in local currency, excluding acquisitions) is an excellent achievement in the current environment. A strong 10.5% growth in Europe, coupled with over 50% growth in the Middle East and Africa, contributed to this result. Synthes has once again expanded its leadership position and gained market share. Winter weather contributes to growth Europe s strong performance was influenced by severe winter weather, both at the beginning and especially at the end of the year. The early winter in November and December led to icy roads, sidewalks with black ice, and increased winter sport activity: a collection of activities that often leads to increased accidents and falls that involve a higher fracture volume and therefore provided us with record sales months. These record months, however, are not expected to be repeated in Our European Trauma, CMF and Power Tools business has shown strong growth in Synthes has significantly increased its share in these markets. Our Spine business on the other hand as in the U.S. has faced challenges in terms of lower procedure volumes, continued pricing pressure and the entry of new competitors for market share in this challenging market. Additionally, the lack of competitive products in certain segments of our portfolio has undermined our performance. In the Middle East, we benefitted from a large tender order (bulk order from the government of Saudi Arabia), which is made bi-annually on behalf of government-owned hospitals. The economic environment in Europe will continue to soften due to budget cuts and cost constraints in the healthcare sector across most BR8 Business Report
19 Synthes. Annual Report 2010 countries. Capital purchase of equipment has been significantly reduced and Synthes has started an initiative to better manage the effectiveness of the loan service in order to support the increased demand of loan requests. Outlook for 2011 In the middle of 2011, our European headquarters building in Solothurn will be finished and accommodate several hundred employees in the very heart of Switzerland. It is ideally located, close to Zurich and its international airport, the city of Berne, and our Swiss manufacturing sites. In 2011 overall, we expect a continuation of our market share expansion in EMEA. Growth will be lower than in 2010, mainly due to the absence of the Saudi Arabia tender order and due to an expected milder winter. Asia Pacific Asia Pacific attained sales growth of 10.3% (in local currencies) in 2010, a result of mixed performances in the various areas within that region. This growth is above the company s average and a reflection of the potential of emerging markets that Synthes brings to life. Our Asia Pacific business is made up of four distinctive regions. The main share of the business comes from Japan (approximately 40% of sales) and Australia/New Zealand (approximately 20% of sales). These two regions are comparable to Western Europe or the U.S. in terms of the penetration and use of our products. Despite bi-annual price cuts in Japan, the price level in both Japan and Australia is high, allowing for profitable business operations. The third region consists of the larger emerging markets of China, India and Korea. There is a huge demand for our products in these countries. The challenge for us is to keep up with the development of infrastructure (hospital and equipment) as well as trainings of potential new surgeon customers in order to secure the supply to the emerging hospitals that will provide care to the growing population in these countries. The last segment within Asia Pacific consists of a small number of distributor-operated countries in East and Southeast Asia (mainly Taiwan and Thailand), which will eventually also become more meaningful. In 2010, the business continued to expand profitably, securing future growth in all areas. There is an exceptional growth potential in both our Spine and CMF divisions (current market share of 11% and 15%, respectively), primarily because of our relatively recent entry into these businesses in most emerging markets. Manufacturing site in China formally inaugurated A highlight of the year was the formal opening of our new manufacturing site in Suzhou, close to Shanghai, in May. This facility, which is subject to the same rigorous quality standards as our U.S. and European factories, will be coupled with a small product development center and a distribution center that will provide local markets with products appropriate for the Asian population. The factory employs approximately 50 people and manufactures just under 100 line items for India, Taiwan and Malaysia. In order to break into China, we need to undertake clinical trials. These are currently in progress and all approvals are expected in Being a local manufacturer in China enables us to provide Synthes quality implants to a large number of patients who were previously unable to have access to this technology. In addition, we are able to tailor down-sampling product assortments and thereby remain an attractive supplier who understands the specific economic challenges and limitations, and addresses the unique medical needs of patients, in this region. Sales force continues to grow The Asia Pacific sales force grew by 17% in 2010, a result of the commitment to expansion of our service and technical expertise that we want to bring to operating theatres across the region. A specific emphasis was placed on product-specific staff training and education, to strengthen product and technical expertise as well as improve business efficiency and streamline productivity levels. In 2011, the sales force will continue to be expanded as our presence in emerging market grows and the breadth of our product line expands. Education in conjunction with the AO Foundation Together with the AO Foundation (our partner in education and product development), over 130 basic, intermediate and advanced level educational courses were conducted across Asia Pacific in The AO continues to set the standard for surgeon education in trauma, spine and CMF and our continued collaboration with this world-leading organization ensures that we provide surgeons in the region with the best education available. Synthes training and exhibitions A highlight in the training calendar was the first four-day advanced Hand & Wrist cadaveric course, co-presented by the Asia Pacific training team and seven highly respected surgical faculty members. This will become a regular event in the training calendar. Exhibition highlights of the year included high-level sponsorships at a number of Business Report BR9
20 Business Report conferences including the Asian Congress on Oral & Maxillofacial Surgery in Kuala Lumpur, Malaysia, and the triennial International Federation of Societies for Surgery of the Hand (IFSSH in Seoul, South Korea. Outlook for 2011 and beyond In the mid-term future, our goal is to broaden our Asian specific implant portfolio, to provide a dedicated wet lab training site for both surgeons and employees for the region. In addition, we will continue to push for penetration into young markets, the next step being Vietnam. Latin America Over the last few years, Latin America has become more developed and in most countries in the region it is now standard to use internal fixation products for the treatment of orthopaedic trauma, spine and CMF. From this viewpoint, Synthes sales growth of 14.6% (on a constant currency basis) is especially remarkable. Brazil and Mexico large markets in Latin America performed especially well. However, as in other regions of the world, the Spine business had a weaker performance in This can be attributed to a combination of a slowdown in the activity from private hospitals, price pressures in the public hospital segment and the use of more conservative treatment schemes. In addition, the business in countries where distributors are engaged, rather than our own sales force, performed significantly weaker than in other years. Fortunately, our distributorship business represents less than 9% of total Latin American sales. and faster market introductions of the latest technologies that this structure typically entails. In view of the success of logistics points that were set up previously in Brazil and Colombia, this concept was also introduced in Peru and Mexico. These points allow implant and instrument sets to reach the end users in only a few hours, rather than as long as a full day transit time. The AO Foundation remains an important partner in education and continues to be recognized as a valuable training organization to surgeons and operating room personnel in this region. In 2010, 122 AO courses and events in the region were conducted and over 60 are already scheduled for Additional courses will be added as required throughout the year. Allocation of personnel in product development 2010 Asia Pacific 1% Europe 42% North America 57% Allocation of sales personnel 2010 (3,636 people in total) Latin America 13% Various countries with varying performances We were especially encouraged by the positive turn we saw in the middle of 2010 and the upswing that followed in the third and fourth quarter. Asia Pacific 21% North America 45% In Brazil and Mexico, which together represent 50% of our Latin American business, we further progressed with penetration of the markets Europe 21% and posted sales growth of over 20%, on a constant currency basis. In Colombia however, where we generate almost a third of our sales for the region, we had to face various challenges in 2010: on the one hand, the national elections did not allow for growth in public budgets, and on the other, reforms in the national health system caused further uncertainty. Nevertheless, we are confident that 2011 will provide us with better performance in this important market. Sales force growth 2010 North America 3% Europe Asia Pacific 12% 17% For 2011, we are planning to initiate a direct market presence in Chile (where we engaged a distributor until 2010) and we expect to see above average sales growth caused by the increased service efforts Latin America 22% 0% 5% 10% 15% 20% 25% BR10 Business Report
Financial Information
 Financial Information Solid results with in all key financial metrics of 23.6 bn, up 0.4% like-for like Adjusted EBITA margin up 0.3 pt on organic basis Net profit up +4% to 1.9 bn Record Free Cash Flow
Financial Information Solid results with in all key financial metrics of 23.6 bn, up 0.4% like-for like Adjusted EBITA margin up 0.3 pt on organic basis Net profit up +4% to 1.9 bn Record Free Cash Flow
Patient Information. Lateral Lumbar Interbody Fusion Surgery (LLIF).
 Patient Information. Lateral Lumbar Interbody Fusion Surgery (LLIF). Understanding your spine Disc Between each pair of vertebrae there is a disc that acts as a cushion to protect the vertebra, allows
Patient Information. Lateral Lumbar Interbody Fusion Surgery (LLIF). Understanding your spine Disc Between each pair of vertebrae there is a disc that acts as a cushion to protect the vertebra, allows
Advances In Spine Care. James D. Bruffey M.D. Scripps Clinic Division of Orthopaedic Surgery Section of Spinal Surgery
 Advances In Spine Care James D. Bruffey M.D. Scripps Clinic Division of Orthopaedic Surgery Section of Spinal Surgery Introduction The Spine - A common source of problems Back pain is the #2 presenting
Advances In Spine Care James D. Bruffey M.D. Scripps Clinic Division of Orthopaedic Surgery Section of Spinal Surgery Introduction The Spine - A common source of problems Back pain is the #2 presenting
Titanium Wire with Barb and Needle. For canthal tendon procedures.
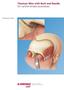 Titanium Wire with Barb and Needle. For canthal tendon procedures. Technique Guide Instruments and implants approved by the AO Foundation Table of Contents Introduction Titanium Wire with Barb and Needle
Titanium Wire with Barb and Needle. For canthal tendon procedures. Technique Guide Instruments and implants approved by the AO Foundation Table of Contents Introduction Titanium Wire with Barb and Needle
Patient Information. Anterior Cervical Discectomy and Fusion Surgery (ACDF).
 Patient Information. Anterior Cervical Discectomy and Fusion Surgery (ACDF). Understanding your spine Disc Between each pair of vertebrae there is a disc that acts as a cushion to protect the vertebra,
Patient Information. Anterior Cervical Discectomy and Fusion Surgery (ACDF). Understanding your spine Disc Between each pair of vertebrae there is a disc that acts as a cushion to protect the vertebra,
Mitsubishi Electric Announces Consolidated and Non-consolidated Financial Results for Fiscal 2016
 MITSUBISHI ELECTRIC CORPORATION PUBLIC RELATIONS DIVISION 7-3, Marunouchi 2-chome, Chiyoda-ku, Tokyo, 100-8310 Japan FOR IMMEDIATE RELEASE No. 3023 Investor Relations Inquiries Investor Relations Group,
MITSUBISHI ELECTRIC CORPORATION PUBLIC RELATIONS DIVISION 7-3, Marunouchi 2-chome, Chiyoda-ku, Tokyo, 100-8310 Japan FOR IMMEDIATE RELEASE No. 3023 Investor Relations Inquiries Investor Relations Group,
Half Year 2015 Results
 Half Year 2015 Results Letter to shareholders LifeWatch First Half Highlights Revenue growth of 9.1% to USD 52.5 million Above-market growth of over 12% in core monitoring services resulting in market
Half Year 2015 Results Letter to shareholders LifeWatch First Half Highlights Revenue growth of 9.1% to USD 52.5 million Above-market growth of over 12% in core monitoring services resulting in market
Modular Sternal Cable System. Flexibility and strength in sternal closure and repair.
 Modular Sternal Cable System. Flexibility and strength in sternal closure and repair. Multiple closure options Stable fixation in all configurations Improved strength and flexibility compared to wire Modular
Modular Sternal Cable System. Flexibility and strength in sternal closure and repair. Multiple closure options Stable fixation in all configurations Improved strength and flexibility compared to wire Modular
Benefit Solutions for Federal Employees
 Benefit Solutions for Federal Employees Accident insurance: You re not a kid anymore, even if you still play like one Insurance that helps you act the age you feel Because you re never too old to have
Benefit Solutions for Federal Employees Accident insurance: You re not a kid anymore, even if you still play like one Insurance that helps you act the age you feel Because you re never too old to have
DUKE ORTHOPAEDIC SURGERY GOALS AND OBJECTIVES SPINE SERVICE
 GOALS AND OBJECTIVES PATIENT CARE Able to perform a complete musculoskeletal and neurologic examination on the patient including cervical spine, thoracic spine, and lumbar spine. The neurologic examination
GOALS AND OBJECTIVES PATIENT CARE Able to perform a complete musculoskeletal and neurologic examination on the patient including cervical spine, thoracic spine, and lumbar spine. The neurologic examination
Analysts and press conference for the financial year 2010. March 16, 2011
 Analysts and press conference for the financial year 2010 March 16, 2011 Welcome Walter Gränicher Jost Sigrist Werner Schmidli Chairman of the Board of Directors Chief Executive Officer Chief Financial
Analysts and press conference for the financial year 2010 March 16, 2011 Welcome Walter Gränicher Jost Sigrist Werner Schmidli Chairman of the Board of Directors Chief Executive Officer Chief Financial
WELCOME. to the DePuy Synthes Institute, LLC
 WELCOME to the DePuy Synthes Institute, LLC Healthcare professionals from around the world come to the DePuy Synthes Institute to learn, connect and share knowledge with the common goal of improving patient
WELCOME to the DePuy Synthes Institute, LLC Healthcare professionals from around the world come to the DePuy Synthes Institute to learn, connect and share knowledge with the common goal of improving patient
PERIPROSTHETIC IMPLANTS
 PERIPROSTHETIC IMPLANTS PRODUCT OVERVIEW CLINICAL SOLUTIONS Periprosthetic fractures present unique challenges, such as how to gain fixation when the medullary canal is occupied. Special techniques and
PERIPROSTHETIC IMPLANTS PRODUCT OVERVIEW CLINICAL SOLUTIONS Periprosthetic fractures present unique challenges, such as how to gain fixation when the medullary canal is occupied. Special techniques and
Klöckner & Co SE. FY 2013 Results
 Klöckner & Co SE A Leading Multi Metal Distributor Gisbert Rühl CEO FY 2013 Results Press Conference March 6, 2014 Disclaimer This presentation contains forward-looking statements which reflect the current
Klöckner & Co SE A Leading Multi Metal Distributor Gisbert Rühl CEO FY 2013 Results Press Conference March 6, 2014 Disclaimer This presentation contains forward-looking statements which reflect the current
Does the pain radiating down your legs, buttocks or lower back prevent you from walking long distances?
 Does the pain radiating down your legs, buttocks or lower back prevent you from walking long distances? Do you experience weakness, tingling, numbness, stiffness, or cramping in your legs, buttocks or
Does the pain radiating down your legs, buttocks or lower back prevent you from walking long distances? Do you experience weakness, tingling, numbness, stiffness, or cramping in your legs, buttocks or
Chasing growth in a constrained environment
 Chasing growth in a constrained environment Bernard Fontana CEO June 14, 2012 Agenda 1 Drivers of demand growth 2 Allocating the asset footprint according to demand 3 Growth from customer excellence &
Chasing growth in a constrained environment Bernard Fontana CEO June 14, 2012 Agenda 1 Drivers of demand growth 2 Allocating the asset footprint according to demand 3 Growth from customer excellence &
Management s Discussion and Analysis
 Management s Discussion and Analysis 6 Financial Policy Sysmex regards increasing its market capitalization to maximize corporate value an important management objective and pays careful attention to stable
Management s Discussion and Analysis 6 Financial Policy Sysmex regards increasing its market capitalization to maximize corporate value an important management objective and pays careful attention to stable
Half year results 2011
 Half year results 2011 29 July 2011 Bert De Graeve, Chief Executive Officer Bruno Humblet, Chief Financial Officer Address by Bert De Graeve, Chief Executive Officer Introductory remark The consolidated
Half year results 2011 29 July 2011 Bert De Graeve, Chief Executive Officer Bruno Humblet, Chief Financial Officer Address by Bert De Graeve, Chief Executive Officer Introductory remark The consolidated
The ReThink Group plc ( ReThink Group or the Group ) Unaudited Interim Results. Profits double as strategy delivers continued improved performance
 The ReThink Group plc ( ReThink Group or the Group ) Unaudited Interim Results Profits double as strategy delivers continued improved performance The Group (AIM: RTG), one of the UK s leading recruitment
The ReThink Group plc ( ReThink Group or the Group ) Unaudited Interim Results Profits double as strategy delivers continued improved performance The Group (AIM: RTG), one of the UK s leading recruitment
MEDIA RELEASE SIKA WITH STRONG GROWTH IN EMERGING MARKETS
 DATUM 1 / 6 Zugerstrasse 50 6341 Baar, Switzerland www.sika.com CONTACT TELEPHONE E MAIL Dominik Slappnig Corporate Communications & Investor Relations +41 58 436 68 21 slappnig.dominik@ch.sika.com SIKA
DATUM 1 / 6 Zugerstrasse 50 6341 Baar, Switzerland www.sika.com CONTACT TELEPHONE E MAIL Dominik Slappnig Corporate Communications & Investor Relations +41 58 436 68 21 slappnig.dominik@ch.sika.com SIKA
Verifone Reports Results for the Second Quarter of Fiscal 2016
 Verifone Reports Results for the Second Quarter of Fiscal 2016 SAN JOSE, Calif. (BUSINESS WIRE) Verifone (NYSE: PAY), a world leader in payments and commerce solutions, today announced financial results
Verifone Reports Results for the Second Quarter of Fiscal 2016 SAN JOSE, Calif. (BUSINESS WIRE) Verifone (NYSE: PAY), a world leader in payments and commerce solutions, today announced financial results
Press release Regulated information
 Press release Regulated information 26 February 2010 Annual results 2009 Press Katelijn Bohez T +32 56 23 05 71 Investor Relations Jacques Anckaert T +32 56 23 05 72 www.bekaert.com www.bekaert.mobi Bekaert
Press release Regulated information 26 February 2010 Annual results 2009 Press Katelijn Bohez T +32 56 23 05 71 Investor Relations Jacques Anckaert T +32 56 23 05 72 www.bekaert.com www.bekaert.mobi Bekaert
advantedge Services advantedge Microsoft Dynamics AX Services for ISVs, VARs and existing AX users Africa Asia-Pac Europe North America South America
 advantedge Services SAGlobal advantedge Microsoft Dynamics AX Services for ISVs, VARs and existing AX users Africa Asia-Pac Europe North America South America About SAGlobal SAGlobal is the largest specialist
advantedge Services SAGlobal advantedge Microsoft Dynamics AX Services for ISVs, VARs and existing AX users Africa Asia-Pac Europe North America South America About SAGlobal SAGlobal is the largest specialist
2013 Half Year Results
 2013 Half Year Results Erwin Stoller, Executive Chairman Joris Gröflin, Chief Financial Officer Agenda 1. Introduction and summary of first half year 2013 2. Financial results first half year 2013 3. Outlook
2013 Half Year Results Erwin Stoller, Executive Chairman Joris Gröflin, Chief Financial Officer Agenda 1. Introduction and summary of first half year 2013 2. Financial results first half year 2013 3. Outlook
2013 North American SSL Certificate
 2013 2014 INSERT COMPANY LOGO HERE 2014 North American 2013 North American SSL Certificate Minimally Invasive Spinal Surgical Solutions Product Product Line Leadership Strategy Leadership Award Award Background
2013 2014 INSERT COMPANY LOGO HERE 2014 North American 2013 North American SSL Certificate Minimally Invasive Spinal Surgical Solutions Product Product Line Leadership Strategy Leadership Award Award Background
Statement by Kasper Rorsted Chairman of the Management Board Conference-Call November 11, 2015, 10.30 a.m.
 Statement by Kasper Rorsted Chairman of the Management Board Conference-Call November 11, 2015, 10.30 a.m. Welcome to our conference call today. As you will have seen, this morning we sent out our news
Statement by Kasper Rorsted Chairman of the Management Board Conference-Call November 11, 2015, 10.30 a.m. Welcome to our conference call today. As you will have seen, this morning we sent out our news
Second Quarter Highlights
 Kalamazoo, Michigan - July 23, 2015 - Stryker Corporation (NYSE:SYK) reported operating results for the second quarter of 2015: Second Quarter Highlights Raises full year organic sales growth guidance
Kalamazoo, Michigan - July 23, 2015 - Stryker Corporation (NYSE:SYK) reported operating results for the second quarter of 2015: Second Quarter Highlights Raises full year organic sales growth guidance
Technology Breakthrough in Spinal Implants (Technical Insights)
 Technology Breakthrough in Spinal Implants (Technical Insights) Biomaterial innovations is a growth factor for spinal implant market June 2014 Table of Contents Section Page Number Executive Summary 4
Technology Breakthrough in Spinal Implants (Technical Insights) Biomaterial innovations is a growth factor for spinal implant market June 2014 Table of Contents Section Page Number Executive Summary 4
CITI REPORTS FIRST QUARTER INCOME OF $5.01 BILLION, EPS OF $1.01 RECORD REVENUES OF $25.5 BILLION, UP 15% INTERNATIONAL REVENUES UP 18%
 CITI REPORTS FIRST QUARTER INCOME OF $5.01 BILLION, EPS OF $1.01 RECORD REVENUES OF $25.5 BILLION, UP 15% INTERNATIONAL REVENUES UP 18% RECORD REVENUES AND NET INCOME IN MARKETS & BANKING AND WEALTH MANAGEMENT
CITI REPORTS FIRST QUARTER INCOME OF $5.01 BILLION, EPS OF $1.01 RECORD REVENUES OF $25.5 BILLION, UP 15% INTERNATIONAL REVENUES UP 18% RECORD REVENUES AND NET INCOME IN MARKETS & BANKING AND WEALTH MANAGEMENT
FOSSIL GROUP, INC. REPORTS FOURTH QUARTER AND FISCAL YEAR 2014 RESULTS; Fourth Quarter Net Sales of $1.065 Billion; Diluted EPS Increases 12% to $3.
 FOSSIL GROUP, INC. REPORTS FOURTH QUARTER AND FISCAL YEAR 2014 RESULTS; Fourth Quarter Net Sales of $1.065 Billion; Diluted EPS Increases 12% to $3.00 Fiscal Year 2014 Net Sales Increase 8% to $3.510 Billion;
FOSSIL GROUP, INC. REPORTS FOURTH QUARTER AND FISCAL YEAR 2014 RESULTS; Fourth Quarter Net Sales of $1.065 Billion; Diluted EPS Increases 12% to $3.00 Fiscal Year 2014 Net Sales Increase 8% to $3.510 Billion;
HR - A STRATEGIC PARTNER Evolution in the adoption of Human Capital Management systems
 www.wipro.com HR - A STRATEGIC PARTNER Evolution in the adoption of Human Capital Management systems FUTURE READY SYSTEM FOR AN INSPIRED WORKFORCE Anand Gupta, Director, Oracle Cloud Services, Wipro Table
www.wipro.com HR - A STRATEGIC PARTNER Evolution in the adoption of Human Capital Management systems FUTURE READY SYSTEM FOR AN INSPIRED WORKFORCE Anand Gupta, Director, Oracle Cloud Services, Wipro Table
Investor Presentation
 Investor Presentation June 2015 Safe Harbor Statement Statements in this presentation, which are not historical facts, may be deemed to be "forward looking statements. Actual and anticipated future results
Investor Presentation June 2015 Safe Harbor Statement Statements in this presentation, which are not historical facts, may be deemed to be "forward looking statements. Actual and anticipated future results
FRANKLIN ELECTRIC REPORTS RECORD SECOND QUARTER 2013 SALES AND EARNINGS
 For Immediate Release For Further Information Refer to: John J. Haines 260-824-2900 FRANKLIN ELECTRIC REPORTS RECORD SECOND QUARTER 2013 SALES AND EARNINGS Bluffton, Indiana July 30, 2013 - Franklin Electric
For Immediate Release For Further Information Refer to: John J. Haines 260-824-2900 FRANKLIN ELECTRIC REPORTS RECORD SECOND QUARTER 2013 SALES AND EARNINGS Bluffton, Indiana July 30, 2013 - Franklin Electric
coligne treatment technology
 coligne treatment technology coligne a strategy in spine Leadership begins before the patient enters the surgical theater, among the spine care professionals forming the coalliance. This may be a small
coligne treatment technology coligne a strategy in spine Leadership begins before the patient enters the surgical theater, among the spine care professionals forming the coalliance. This may be a small
Speech of the Board of Management. General Shareholders Meeting, 9 June 2015
 Speech of the Board of Management General Shareholders Meeting, 9 June 2015 AGENDA Overview of the financial year Steven Holland, CEO Financial figures 2014 Georg Müller, CFO ConnectingChemistry Steven
Speech of the Board of Management General Shareholders Meeting, 9 June 2015 AGENDA Overview of the financial year Steven Holland, CEO Financial figures 2014 Georg Müller, CFO ConnectingChemistry Steven
The Petrylaw Lawsuits Settlements and Injury Settlement Report
 The Petrylaw Lawsuits Settlements and Injury Settlement Report BACK INJURIES How Minnesota Juries Decide the Value of Pain and Suffering in Back Injury Cases The Petrylaw Lawsuits Settlements and Injury
The Petrylaw Lawsuits Settlements and Injury Settlement Report BACK INJURIES How Minnesota Juries Decide the Value of Pain and Suffering in Back Injury Cases The Petrylaw Lawsuits Settlements and Injury
Information for the Patient About Surgical
 Information for the Patient About Surgical Decompression and Stabilization of the Spine Aging and the Spine Daily wear and tear, along with disc degeneration due to aging and injury, are common causes
Information for the Patient About Surgical Decompression and Stabilization of the Spine Aging and the Spine Daily wear and tear, along with disc degeneration due to aging and injury, are common causes
Anterior Lumbar Interbody Fusion (ALIF). Instrument set supports placement of ALIF spacers using anterior or anterolateral approach.
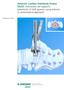 Anterior Lumbar Interbody Fusion (ALIF). Instrument set supports placement of ALIF spacers using anterior or anterolateral approach. Technique Guide Instruments and implants approved by the AO Foundation
Anterior Lumbar Interbody Fusion (ALIF). Instrument set supports placement of ALIF spacers using anterior or anterolateral approach. Technique Guide Instruments and implants approved by the AO Foundation
A Leading Global Health Care Group
 A Leading Global Health Care Group Credit Suisse Global Health Care Conference, March 4, 2015 For detailed financial information please see our annual/quarterly reports and/or conference call materials
A Leading Global Health Care Group Credit Suisse Global Health Care Conference, March 4, 2015 For detailed financial information please see our annual/quarterly reports and/or conference call materials
Summary of Consolidated Financial Statements for the Second Quarter of Fiscal Year Ending March 31, 2012 (Japanese GAAP)
 This document is a translation of the Japanese financial statements and is not in conformity with accounting principles of the United States. Summary of Consolidated Financial Statements for the Second
This document is a translation of the Japanese financial statements and is not in conformity with accounting principles of the United States. Summary of Consolidated Financial Statements for the Second
Patient Guide to Lower Back Surgery
 The following is a sampling of products offered by Zimmer Spine for use in Open Lumbar Fusion procedures. Patient Guide to Lower Back Surgery Open Lumbar Fusion Dynesys The Dynesys Dynamic Stabilization
The following is a sampling of products offered by Zimmer Spine for use in Open Lumbar Fusion procedures. Patient Guide to Lower Back Surgery Open Lumbar Fusion Dynesys The Dynesys Dynamic Stabilization
Patient Guide to Neck Surgery
 The following is a sampling of products offered by Zimmer Spine for use in Anterior Cervical Fusion procedures. Patient Guide to Neck Surgery Anterior Cervical Fusion Trinica Select With the Trinica and
The following is a sampling of products offered by Zimmer Spine for use in Anterior Cervical Fusion procedures. Patient Guide to Neck Surgery Anterior Cervical Fusion Trinica Select With the Trinica and
Lending Club Reports Third Quarter 2015 Results Q3 operating revenue up 104% year-over-year to $115.1 million
 Lending Club Reports Third Quarter Results Q3 operating revenue up 104% year-over-year to $115.1 million SAN FRANCISCO October 29, Lending Club (NYSE:LC), the world s largest online marketplace connecting
Lending Club Reports Third Quarter Results Q3 operating revenue up 104% year-over-year to $115.1 million SAN FRANCISCO October 29, Lending Club (NYSE:LC), the world s largest online marketplace connecting
PATIENT SPECIFIC PLATES FOR MANDIBLE: CUSTOM MADE PLATES FOR TRAUMA AND RECONSTRUCTION
 PATIENT SPECIFIC PLATES FOR MANDIBLE: CUSTOM MADE PLATES FOR TRAUMA AND RECONSTRUCTION Derived from patient CT Data Customizable design features Strength with low profile This publication is not intended
PATIENT SPECIFIC PLATES FOR MANDIBLE: CUSTOM MADE PLATES FOR TRAUMA AND RECONSTRUCTION Derived from patient CT Data Customizable design features Strength with low profile This publication is not intended
40 Years of Color Innovation
 Datacolor 40 Years of Color Innovation Swiss Equity Conference 2010 ALBERT BUSCH & MARK LEUCHTMANN SEPTEMBER 15, 2010 0 Safe Harbor Statement This document has been prepared by the Company solely for the
Datacolor 40 Years of Color Innovation Swiss Equity Conference 2010 ALBERT BUSCH & MARK LEUCHTMANN SEPTEMBER 15, 2010 0 Safe Harbor Statement This document has been prepared by the Company solely for the
Annual Press Conference 2015. 18 March 2015
 Annual Press Conference 2015 18 March 2015 AGENDA Annual Press Conference 2015 Topic Speaker Time 1. Welcome Hubertus Spethmann 10.00 am 2. 2014 at a glance 2.1. Highlights Steven Holland 2.2. Financial
Annual Press Conference 2015 18 March 2015 AGENDA Annual Press Conference 2015 Topic Speaker Time 1. Welcome Hubertus Spethmann 10.00 am 2. 2014 at a glance 2.1. Highlights Steven Holland 2.2. Financial
MAPFRE in 2014. Antonio Huertas. Presentation of Annual Results February 11, 2015. MAPFRE Chairman & CEO
 MAPFRE in 2014 Presentation of Annual Results February 11, 2015 Antonio Huertas MAPFRE Chairman & CEO 2014 Results MAPFRE's results are excellent: 845 million euros in profits 2013 2014 % Consolidated
MAPFRE in 2014 Presentation of Annual Results February 11, 2015 Antonio Huertas MAPFRE Chairman & CEO 2014 Results MAPFRE's results are excellent: 845 million euros in profits 2013 2014 % Consolidated
LENOVO 1 ST QUARTER FY15-16: TOUGH MARKETS, SOLID RESULTS
 LENOVO 1 ST QUARTER FY15-16: TOUGH MARKETS, SOLID RESULTS Decisive actions being taken to meet challenges and accelerate business transformation Revenue was US$10.7 billion, up 3 percent; excluding foreign
LENOVO 1 ST QUARTER FY15-16: TOUGH MARKETS, SOLID RESULTS Decisive actions being taken to meet challenges and accelerate business transformation Revenue was US$10.7 billion, up 3 percent; excluding foreign
Do you have Back Pain? Associated with:
 Do you have Back Pain? Associated with: Herniated Discs? Protruding Discs? Degenerative Disk Disease? Posterior Facet Syndrome? Sciatica? You may be a candidate for Decompression Therapy The Dynatronics
Do you have Back Pain? Associated with: Herniated Discs? Protruding Discs? Degenerative Disk Disease? Posterior Facet Syndrome? Sciatica? You may be a candidate for Decompression Therapy The Dynatronics
FOCUS PROASSURANCE 2005 ANNUAL REPORT
 FOCUS PROASSURANCE 2005 ANNUAL REPORT THE FOCUS OF PROASSURANCE CORPORATION We are a specialty insurer focused on writing medical professional liability insurance and developing insurance solutions which
FOCUS PROASSURANCE 2005 ANNUAL REPORT THE FOCUS OF PROASSURANCE CORPORATION We are a specialty insurer focused on writing medical professional liability insurance and developing insurance solutions which
Diluted net income per share (Yen) 90.03 129.05. Net assets per share assets. Equity
 Summary of Consolidated Financial Results for the Nine months Ended December 31, 2008 February 3, 2009 Listed company name : Sysmex Corporation Code : 6869 Listed stock exchanges : Tokyo Stock Exchange
Summary of Consolidated Financial Results for the Nine months Ended December 31, 2008 February 3, 2009 Listed company name : Sysmex Corporation Code : 6869 Listed stock exchanges : Tokyo Stock Exchange
Degenerative Spine Solutions
 Degenerative Spine Solutions The Backbone for Your Surgical Needs Aesculap Spine Backbone for Your Degenerative Spine Needs Comprehensive operative solutions, unique product technology and world-class
Degenerative Spine Solutions The Backbone for Your Surgical Needs Aesculap Spine Backbone for Your Degenerative Spine Needs Comprehensive operative solutions, unique product technology and world-class
Speech of the Board of Management. General Shareholders Meeting, 17 June 2014
 Speech of the Board of Management General Shareholders Meeting, 17 June 2014 AGENDA Overview of the financial year 2013 Steven Holland, CEO Financial figures 2013 Georg Müller, CFO 24/7 on time worldwide
Speech of the Board of Management General Shareholders Meeting, 17 June 2014 AGENDA Overview of the financial year 2013 Steven Holland, CEO Financial figures 2013 Georg Müller, CFO 24/7 on time worldwide
.org. Fractures of the Thoracic and Lumbar Spine. Cause. Description
 Fractures of the Thoracic and Lumbar Spine Page ( 1 ) Spinal fractures can vary widely in severity. While some fractures are very serious injuries that require emergency treatment, other fractures can
Fractures of the Thoracic and Lumbar Spine Page ( 1 ) Spinal fractures can vary widely in severity. While some fractures are very serious injuries that require emergency treatment, other fractures can
Key figures as of June 30, 2013 1st half
 Never standing still. Interim Report as of June 30, 2013 Contents 2 Key figures as of June 30, 2013 1st half 3 Key figures as of June 30, 2013 2nd quarter 6 Strong revenue growth 12 Consolidated interim
Never standing still. Interim Report as of June 30, 2013 Contents 2 Key figures as of June 30, 2013 1st half 3 Key figures as of June 30, 2013 2nd quarter 6 Strong revenue growth 12 Consolidated interim
www.backandspinefl.com Back and Spine Center 509 Riverside Dr., Suite 203 Stuart, FL 34994 Phone: (772) 288-5862
 Non-Profit ORG. U.S. Postage PAID West Palm BCH. FL. PERMIT NO. 4709 www.backandspinefl.com Back and Spine Center 509 Riverside Dr., Suite 203 Stuart, FL 34994 Phone: (772) 288-5862 The evolution of neurosurgery
Non-Profit ORG. U.S. Postage PAID West Palm BCH. FL. PERMIT NO. 4709 www.backandspinefl.com Back and Spine Center 509 Riverside Dr., Suite 203 Stuart, FL 34994 Phone: (772) 288-5862 The evolution of neurosurgery
Health Care Worldwide. Citi - European Credit Conference September 24, 2015 - London
 Health Care Worldwide Citi - European Credit Conference September 24, 2015 - London Safe Harbor Statement This presentation contains forward-looking statements that are subject to various risks and uncertainties.
Health Care Worldwide Citi - European Credit Conference September 24, 2015 - London Safe Harbor Statement This presentation contains forward-looking statements that are subject to various risks and uncertainties.
Global Investment Trends Survey May 2015. A study into global investment trends and saver intentions in 2015
 May 2015 A study into global investment trends and saver intentions in 2015 Global highlights Schroders at a glance Schroders at a glance At Schroders, asset management is our only business and our goals
May 2015 A study into global investment trends and saver intentions in 2015 Global highlights Schroders at a glance Schroders at a glance At Schroders, asset management is our only business and our goals
China Pacific Insurance (Group) Co., Ltd. Issue of 2010 Interim Results
 China Pacific Insurance (Group) Co., Ltd. Issue of 2010 Interim Results Strong Growth of Insurance Business and Significant Increase in Operating Profit Shanghai, Hong Kong, 30 August 2010 China Pacific
China Pacific Insurance (Group) Co., Ltd. Issue of 2010 Interim Results Strong Growth of Insurance Business and Significant Increase in Operating Profit Shanghai, Hong Kong, 30 August 2010 China Pacific
Planning and Achieving Lumbar Spinal Balance. November 7, 2015 New York, NY. Course Chairmen: Dom Coric, MD Paul McAfee, MD, MBA EW YORK
 Planning and Achieving Lumbar Spinal Balance November 7, 2015 New York, NY Course Chairmen: Dom Coric, MD Paul McAfee, MD, MBA EW YORK Course Dom Coric, MD Co-Chairman Paul McAfee, MD, MBA Co-Chairman
Planning and Achieving Lumbar Spinal Balance November 7, 2015 New York, NY Course Chairmen: Dom Coric, MD Paul McAfee, MD, MBA EW YORK Course Dom Coric, MD Co-Chairman Paul McAfee, MD, MBA Co-Chairman
Midyear Presentation 2013. market strategy
 Midyear Higher order Results intake, Presentation lower profitability 2013 focused market strategy THE SAFE HARBOR STATEMENT UNDER THE US PRIVATE SECURITIES LITIGATION REFORM ACT 1995 This presentation
Midyear Higher order Results intake, Presentation lower profitability 2013 focused market strategy THE SAFE HARBOR STATEMENT UNDER THE US PRIVATE SECURITIES LITIGATION REFORM ACT 1995 This presentation
Investment Counselling Program
 Investment Counselling Program To us there are no foreign markets. N19 70 56 W155 08 58 Golfing with friends, Hawaii Your life has a geography. Where you re from, where you re going, where you aspire to
Investment Counselling Program To us there are no foreign markets. N19 70 56 W155 08 58 Golfing with friends, Hawaii Your life has a geography. Where you re from, where you re going, where you aspire to
Minimally Invasive Spine Surgery What is it and how will it benefit patients?
 Minimally Invasive Spine Surgery What is it and how will it benefit patients? Dr Raoul Pope MBChB, FRACS, Neurosurgeon and Minimally Invasive Spine Surgeon Concord Hospital and Mater Private Hospital Sydney
Minimally Invasive Spine Surgery What is it and how will it benefit patients? Dr Raoul Pope MBChB, FRACS, Neurosurgeon and Minimally Invasive Spine Surgeon Concord Hospital and Mater Private Hospital Sydney
Adult Forearm Fractures
 Adult Forearm Fractures Your forearm is made up of two bones, the radius and ulna. In most cases of adult forearm fractures, both bones are broken. Fractures of the forearm can occur near the wrist at
Adult Forearm Fractures Your forearm is made up of two bones, the radius and ulna. In most cases of adult forearm fractures, both bones are broken. Fractures of the forearm can occur near the wrist at
PRESS RELEASE. Ana Botín: Santander is well positioned to face the challenges. We will lead change GENERAL SHAREHOLDERS MEETING
 PRESS RELEASE GENERAL SHAREHOLDERS MEETING Ana Botín: Santander is well positioned to face the challenges. We will lead change Banco Santander has room for growth within our customer base and in our ten
PRESS RELEASE GENERAL SHAREHOLDERS MEETING Ana Botín: Santander is well positioned to face the challenges. We will lead change Banco Santander has room for growth within our customer base and in our ten
Montana Workers Compensation
 Montana Workers Compensation Part 1 - Ratemaking Overview Copyright 2013 National Council on Compensation Insurance, Inc. All Rights Reserved. Mike_Taylor@ncci.com 503-892-1858 WC Ratemaking Basics Where
Montana Workers Compensation Part 1 - Ratemaking Overview Copyright 2013 National Council on Compensation Insurance, Inc. All Rights Reserved. Mike_Taylor@ncci.com 503-892-1858 WC Ratemaking Basics Where
ADAMA DELIVERS ROBUST VOLUME GROWTH DESPITE DIFFICULT MARKET AND CURRENCY CONDITIONS
 ADAMA DELIVERS ROBUST VOLUME GROWTH DESPITE DIFFICULT MARKET AND CURRENCY CONDITIONS Revenues in constant currencies up estimated 9.5% in the third quarter and 6% year-to-date Strong volume growth in all
ADAMA DELIVERS ROBUST VOLUME GROWTH DESPITE DIFFICULT MARKET AND CURRENCY CONDITIONS Revenues in constant currencies up estimated 9.5% in the third quarter and 6% year-to-date Strong volume growth in all
DISCOVER ASCOM S EXCITING BUSINESS FRITZ MUMENTHALER, CEO
 DISCOVER ASCOM S EXCITING BUSINESS FRITZ MUMENTHALER, CEO ABOUT ASCOM OUR STRATEGY ATTRACTIVE GROWTH OPPORTUNITIES AN APPEALING INVESTMENT CASE ASCOM THE ELEVATOR PITCH Two independent ICT businesses,
DISCOVER ASCOM S EXCITING BUSINESS FRITZ MUMENTHALER, CEO ABOUT ASCOM OUR STRATEGY ATTRACTIVE GROWTH OPPORTUNITIES AN APPEALING INVESTMENT CASE ASCOM THE ELEVATOR PITCH Two independent ICT businesses,
INTERACTIVE DATA CORPORATION REPORTS SECOND-QUARTER RESULTS AND REAFFIRMS 2004 OUTLOOK
 INTERACTIVE DATA CORPORATION REPORTS SECOND-QUARTER RESULTS AND REAFFIRMS 2004 OUTLOOK Second-quarter Revenue Increases 6.7%; Net Income Increases by 7.5% BEDFORD, Mass July 22, 2004 Interactive Data Corporation
INTERACTIVE DATA CORPORATION REPORTS SECOND-QUARTER RESULTS AND REAFFIRMS 2004 OUTLOOK Second-quarter Revenue Increases 6.7%; Net Income Increases by 7.5% BEDFORD, Mass July 22, 2004 Interactive Data Corporation
SPINAL FUSION. North American Spine Society Public Education Series
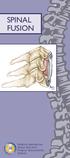 SPINAL FUSION North American Spine Society Public Education Series WHAT IS SPINAL FUSION? The spine is made up of a series of bones called vertebrae ; between each vertebra are strong connective tissues
SPINAL FUSION North American Spine Society Public Education Series WHAT IS SPINAL FUSION? The spine is made up of a series of bones called vertebrae ; between each vertebra are strong connective tissues
Health Care Worldwide
 Health Care Worldwide Goldman Sachs - Leveraged Finance Healthcare Conference March 4, 2014 New York Goldman Sachs Leveraged Finance Conference, Fresenius SE & Co. KGaA Copyright, March 4, 2014 Page 1
Health Care Worldwide Goldman Sachs - Leveraged Finance Healthcare Conference March 4, 2014 New York Goldman Sachs Leveraged Finance Conference, Fresenius SE & Co. KGaA Copyright, March 4, 2014 Page 1
FOSSIL GROUP, INC. REPORTS THIRD QUARTER 2014 RESULTS. Third Quarter Net Sales Increase 10% to $894 Million; Diluted EPS Increases 24% to $1.
 FOSSIL GROUP, INC. REPORTS THIRD QUARTER RESULTS Third Quarter Net Sales Increase 10% to $894 Million; Diluted EPS Increases 24% to $1.96 Updates Full Year Guidance and Provides Fourth Quarter Guidance
FOSSIL GROUP, INC. REPORTS THIRD QUARTER RESULTS Third Quarter Net Sales Increase 10% to $894 Million; Diluted EPS Increases 24% to $1.96 Updates Full Year Guidance and Provides Fourth Quarter Guidance
AmGUARD Insurance Company EastGUARD Insurance Company NorGUARD Insurance Company WestGUARD Insurance Company GUARD
 About Us For over 30 years, we have protected the interests of the small- to mid-sized businesses that insure with us. At Berkshire Hathaway Insurance Companies, we dedicate our efforts in the areas that
About Us For over 30 years, we have protected the interests of the small- to mid-sized businesses that insure with us. At Berkshire Hathaway Insurance Companies, we dedicate our efforts in the areas that
DST SYSTEMS, INC. ANNOUNCES THIRD QUARTER 2015 FINANCIAL RESULTS
 ANNOUNCES THIRD QUARTER 2015 FINANCIAL RESULTS KANSAS CITY, MO - October 22, 2015 DST Systems, Inc. (NYSE: DST) reported consolidated net income of $75.1 million ($2.08 per diluted share) for the third
ANNOUNCES THIRD QUARTER 2015 FINANCIAL RESULTS KANSAS CITY, MO - October 22, 2015 DST Systems, Inc. (NYSE: DST) reported consolidated net income of $75.1 million ($2.08 per diluted share) for the third
Second Quarter and First Half 2015 Trading Update
 Second Quarter and First Half 2015 Trading Update Trading Performance Year-on-Year Gross Profit Reported ( m) Constant 2015 2014 % % Q2 145.3 137.1 +6.0% +10.6% H1 281.0 263.7 +6.6% +10.8% Q2 Gross Profit
Second Quarter and First Half 2015 Trading Update Trading Performance Year-on-Year Gross Profit Reported ( m) Constant 2015 2014 % % Q2 145.3 137.1 +6.0% +10.6% H1 281.0 263.7 +6.6% +10.8% Q2 Gross Profit
CHINA DISTANCE EDUCATION HOLDINGS LIMITED ANNOUNCES FISCAL THIRD QUARTER 2008 RESULTS
 FOR IMMEDIATE RELEASE CHINA DISTANCE EDUCATION HOLDINGS LIMITED ANNOUNCES FISCAL THIRD QUARTER 2008 RESULTS BEIJING, China, August 18, 2008 China Distance Education Holdings Limited (NYSE Arca: DL) ( CDEL,
FOR IMMEDIATE RELEASE CHINA DISTANCE EDUCATION HOLDINGS LIMITED ANNOUNCES FISCAL THIRD QUARTER 2008 RESULTS BEIJING, China, August 18, 2008 China Distance Education Holdings Limited (NYSE Arca: DL) ( CDEL,
If you or a loved one have suffered because of a negligent error during spinal surgery, you will be going through a difficult time.
 If you or a loved one have suffered because of a negligent error during spinal surgery, you will be going through a difficult time. You may be worried about your future, both in respect of finances and
If you or a loved one have suffered because of a negligent error during spinal surgery, you will be going through a difficult time. You may be worried about your future, both in respect of finances and
Staples, Inc. Announces First Quarter 2016 Performance
 Media Contact: Mark Cautela 508-253-3832 Investor Contact: Chris Powers/Scott Tilghman 508-253-4632/1487 Staples, Inc. Announces First Quarter 2016 Performance FRAMINGHAM, Mass., May 18, 2016 Staples,
Media Contact: Mark Cautela 508-253-3832 Investor Contact: Chris Powers/Scott Tilghman 508-253-4632/1487 Staples, Inc. Announces First Quarter 2016 Performance FRAMINGHAM, Mass., May 18, 2016 Staples,
METRO GROUP increases sales 2012 in a challenging consumer environment
 METRO GROUP increases sales in a challenging consumer environment Sales rose by 1.2% to 66.7 billion (adjusted for portfolio measures: +2.3%); EBIT before special items reached around 2.0 billion Operating
METRO GROUP increases sales in a challenging consumer environment Sales rose by 1.2% to 66.7 billion (adjusted for portfolio measures: +2.3%); EBIT before special items reached around 2.0 billion Operating
Jonathan R. Gottlieb, MD 7220 SW 127 Street Pinecrest, FL 33156 jonrgottlieb@gmail.com
 Jonathan R. Gottlieb, MD 7220 SW 127 Street Pinecrest, FL 33156 jonrgottlieb@gmail.com CURRENT PRACTICE September 2009-present University of Miami School of Medicine Assistant Professor, Department of
Jonathan R. Gottlieb, MD 7220 SW 127 Street Pinecrest, FL 33156 jonrgottlieb@gmail.com CURRENT PRACTICE September 2009-present University of Miami School of Medicine Assistant Professor, Department of
Tesla Motors, Inc. Fourth Quarter & Full Year 2013 Shareholder Letter
 Tesla Motors, Inc. Fourth Quarter & Full Year 2013 Shareholder Letter Record 6,892 Model S vehicles sold and delivered in Q4 World s highest Consumer Reports customer satisfaction score of 99/100 Q4 auto
Tesla Motors, Inc. Fourth Quarter & Full Year 2013 Shareholder Letter Record 6,892 Model S vehicles sold and delivered in Q4 World s highest Consumer Reports customer satisfaction score of 99/100 Q4 auto
Sales increased 15 percent to $4.5 billion Earnings per Share increased 37 percent to $0.96 Operating Cash Flow increased 22 percent to $319 million
 Contact: Mark Polzin (314) 982-1758 John Hastings (314) 982-8622 EMERSON REPORTS RECORD FIRST-QUARTER 2006 RESULTS Sales increased 15 percent to $4.5 billion Earnings per Share increased 37 percent to
Contact: Mark Polzin (314) 982-1758 John Hastings (314) 982-8622 EMERSON REPORTS RECORD FIRST-QUARTER 2006 RESULTS Sales increased 15 percent to $4.5 billion Earnings per Share increased 37 percent to
CONFERENCE CALL PRELIMINARY FIGURES FISCAL YEAR 2013
 WELCOME DÜRR AKTIENGESELLSCHAFT CONFERENCE CALL PRELIMINARY FIGURES FISCAL YEAR 2013 Ralf W. Dieter, CEO Ralph Heuwing, CFO Bietigheim-Bissingen, February 25, 2014 www.durr.com DISCLAIMER This presentation
WELCOME DÜRR AKTIENGESELLSCHAFT CONFERENCE CALL PRELIMINARY FIGURES FISCAL YEAR 2013 Ralf W. Dieter, CEO Ralph Heuwing, CFO Bietigheim-Bissingen, February 25, 2014 www.durr.com DISCLAIMER This presentation
A Patient s Guide to Artificial Cervical Disc Replacement
 A Patient s Guide to Artificial Cervical Disc Replacement Each year, hundreds of thousands of adults are diagnosed with Cervical Disc Degeneration, an upper spine condition that can cause pain and numbness
A Patient s Guide to Artificial Cervical Disc Replacement Each year, hundreds of thousands of adults are diagnosed with Cervical Disc Degeneration, an upper spine condition that can cause pain and numbness
FOURTH QUARTER NET INCOME INCREASES 12% TO A RECORD $5.32 BILLION FOURTH QUARTER EPS OF $1.02, UP 12% REVENUES INCREASE 9% TO $21.
 FOURTH QUARTER NET INCOME INCREASES 12% TO A RECORD $5.32 BILLION FOURTH QUARTER EPS OF $1.02, UP 12% REVENUES INCREASE 9% TO $21.9 BILLION CITIGROUP 2004 NET INCOME OF $17.0 BILLION, EPS OF $3.26 REVENUES
FOURTH QUARTER NET INCOME INCREASES 12% TO A RECORD $5.32 BILLION FOURTH QUARTER EPS OF $1.02, UP 12% REVENUES INCREASE 9% TO $21.9 BILLION CITIGROUP 2004 NET INCOME OF $17.0 BILLION, EPS OF $3.26 REVENUES
AIA Delivers Strong Results for Third Quarter 2015. VONB growth of 25 per cent on constant exchange rates
 AIA Group Limited 香 港 中 環 干 諾 道 中 一 號 友 邦 金 融 中 心 三 十 五 樓 35/F, AIA Central 1 Connaught Road Central Hong Kong T: (852) 2832 6166 F: (852) 2838 2005 AIA.com AIA Delivers Strong Results for Third Quarter
AIA Group Limited 香 港 中 環 干 諾 道 中 一 號 友 邦 金 融 中 心 三 十 五 樓 35/F, AIA Central 1 Connaught Road Central Hong Kong T: (852) 2832 6166 F: (852) 2838 2005 AIA.com AIA Delivers Strong Results for Third Quarter
Bank of America Merrill Lynch Banking & Insurance CEO Conference Bob Diamond
 4 October 2011 Bank of America Merrill Lynch Banking & Insurance CEO Conference Bob Diamond Thank you and good morning. It s a pleasure to be here and I d like to thank our hosts for the opportunity to
4 October 2011 Bank of America Merrill Lynch Banking & Insurance CEO Conference Bob Diamond Thank you and good morning. It s a pleasure to be here and I d like to thank our hosts for the opportunity to
Accenture Overview Page 1
 Accenture Overview Page 1 Company Overview Accenture is one of the world s leading management consulting, technology services and outsourcing organizations, with more than 246,000 employees; offices and
Accenture Overview Page 1 Company Overview Accenture is one of the world s leading management consulting, technology services and outsourcing organizations, with more than 246,000 employees; offices and
Kuoni to focus on its core business as a service provider to the global travel industry
 INFORMATION Zurich, 14 January 2015 Kuoni to focus on its core business as a service provider to the global travel industry Exit from tour operating activities Strategic initiatives to accelerate growth
INFORMATION Zurich, 14 January 2015 Kuoni to focus on its core business as a service provider to the global travel industry Exit from tour operating activities Strategic initiatives to accelerate growth
PRESS RELEASE February 4, 2016 SIMPSON MANUFACTURING CO., INC. ANNOUNCES FOURTH QUARTER RESULTS
 PRESS RELEASE February 4, 2016 SIMPSON MANUFACTURING CO., INC. ANNOUNCES FOURTH QUARTER RESULTS Pleasanton, CA - Simpson Manufacturing Co., Inc. (the Company ) (NYSE: SSD) today announced its fourth quarter
PRESS RELEASE February 4, 2016 SIMPSON MANUFACTURING CO., INC. ANNOUNCES FOURTH QUARTER RESULTS Pleasanton, CA - Simpson Manufacturing Co., Inc. (the Company ) (NYSE: SSD) today announced its fourth quarter
PROFILES IN PARTNERSHIP. With the right post-acute care partner, anything is possible.
 PROFILES IN PARTNERSHIP With the right post-acute care partner, anything is possible. Touching the lives of APPROXIMATELY 0,000 patients nationwide every day... in more than 0 inpatient hospitals, in nearly,000
PROFILES IN PARTNERSHIP With the right post-acute care partner, anything is possible. Touching the lives of APPROXIMATELY 0,000 patients nationwide every day... in more than 0 inpatient hospitals, in nearly,000
Herzogenaurach, Germany, July 27, 2004 PUMA AG announces its consolidated nd
 P Quarter P Half-Year For immediate release MEDIA CONTACT: INVESTOR CONTACT: U.S.A.: Lisa Beachy, Tel. +1 617 488 2945 Europe: Ulf Santjer, Tel. +49 9132 81 2489 Dieter Bock, Tel. +49 9132 81 2261 Herzogenaurach,
P Quarter P Half-Year For immediate release MEDIA CONTACT: INVESTOR CONTACT: U.S.A.: Lisa Beachy, Tel. +1 617 488 2945 Europe: Ulf Santjer, Tel. +49 9132 81 2489 Dieter Bock, Tel. +49 9132 81 2261 Herzogenaurach,
2013 GLOBAL PERFORMANCE MANAGEMENT SURVEY REPORT
 2013 GLOBAL PERFORMANCE MANAGEMENT SURVEY REPORT Executive Summary contents Overview Key Findings: Critical Drivers of Performance Management Success Industry Insights Regional and Country Insights Participant
2013 GLOBAL PERFORMANCE MANAGEMENT SURVEY REPORT Executive Summary contents Overview Key Findings: Critical Drivers of Performance Management Success Industry Insights Regional and Country Insights Participant
January 2015. Report on SBLF Participants Small Business Lending Growth Submitted to Congress pursuant to Section 4106(3) of
 January 2015 Report on SBLF Participants Small Business Lending Growth Submitted to Congress pursuant to Section 4106(3) of the Small Business Jobs Act of 2010 OVERVIEW Small businesses are a vital part
January 2015 Report on SBLF Participants Small Business Lending Growth Submitted to Congress pursuant to Section 4106(3) of the Small Business Jobs Act of 2010 OVERVIEW Small businesses are a vital part
Business Entities - Review of the PageGroup: Large, High Potential Markets
 Second Quarter 2014 Trading Update 15 July 2014 PageGroup Second Quarter 2014 Trading Update 2 Overview Group Q2 Gross Profit +9% in constant currency +2% in reported rates Y-o-Y growth in all regions
Second Quarter 2014 Trading Update 15 July 2014 PageGroup Second Quarter 2014 Trading Update 2 Overview Group Q2 Gross Profit +9% in constant currency +2% in reported rates Y-o-Y growth in all regions
A Nielsen Report Global Trust in Advertising and Brand Messages. April 2012
 A Nielsen Report Global Trust in Advertising and Brand Messages April 2012 CONSUMER TRUST IN EARNED ADVERTISING GROWS IN IMPORTANCE Earned media sources remain most credible Trust in traditional paid advertising
A Nielsen Report Global Trust in Advertising and Brand Messages April 2012 CONSUMER TRUST IN EARNED ADVERTISING GROWS IN IMPORTANCE Earned media sources remain most credible Trust in traditional paid advertising
Fundamentals of Spine Surgery and Interventional Pain Management
 The North American Spine Society presents Fundamentals of Spine Surgery and Interventional Pain Management 9.5 Hours of Hands-on Cadaver Lab Sessions 15 AMA PRA Category 1 Credits TM A Surgical/Interventional
The North American Spine Society presents Fundamentals of Spine Surgery and Interventional Pain Management 9.5 Hours of Hands-on Cadaver Lab Sessions 15 AMA PRA Category 1 Credits TM A Surgical/Interventional
FOR IMMEDIATE RELEASE
 FOR IMMEDIATE RELEASE CITIGROUP REPORTS FIRST QUARTER CORE INCOME OF $3.66 BILLION ($0.71 PER SHARE, DILUTED) VS. $3.94 BILLION ($0.76 PER SHARE, DILUTED) IN THE FIRST QUARTER OF 2000 INCOME FROM INVESTMENT
FOR IMMEDIATE RELEASE CITIGROUP REPORTS FIRST QUARTER CORE INCOME OF $3.66 BILLION ($0.71 PER SHARE, DILUTED) VS. $3.94 BILLION ($0.76 PER SHARE, DILUTED) IN THE FIRST QUARTER OF 2000 INCOME FROM INVESTMENT
FOR IMMEDIATE RELEASE
 FOR IMMEDIATE RELEASE O-I REPORTS FULL YEAR AND FOURTH QUARTER 2014 RESULTS O-I generates second highest free cash flow in the Company s history PERRYSBURG, Ohio (February 2, 2015) Owens-Illinois, Inc.
FOR IMMEDIATE RELEASE O-I REPORTS FULL YEAR AND FOURTH QUARTER 2014 RESULTS O-I generates second highest free cash flow in the Company s history PERRYSBURG, Ohio (February 2, 2015) Owens-Illinois, Inc.
