msmseda LC-MS/MS Exploratory Data Analysis
|
|
|
- Bernadette Chase
- 8 years ago
- Views:
Transcription
1 msmseda LC-MS/MS Exploratory Data Analysis Josep Gregori, Alex Sanchez, and Josep Villanueva Vall Hebron Institute of Oncology & Statistics Dept. Barcelona University October 13, Introduction In biomarker discovery, label-free differential proteomics is based on comparing the expression of proteins between different biological conditions [1] [2]. The experimental design involved for these experiments requires of randomization and blocking [12]. Working with balanced blocks in which all biological conditions are measured by a number of technical and/or biological replicates, and within the shortest time window possible, helps to reduce experimental bias. Unfortunately, there are many factors that may bias the results in a systematic manner: different operators, different chromatographic columns, different protein digestions, an eventual repair of the LC-MS system, and different laboratory environment conditions. Blocking and randomization help to control to some extent these factors. However, even using the best experimental design some uncontrolled variables may still interfere with differential proteomics experiments. These uncontrolled variables may be responsible for the manifestation of batch effects. Effects which are usually evidenced when samples do not cluster by their biological condition using multidimensional unsupervised techniques such as Principal Components Analysis (PCA) or Hierarchical Clustering (HC) [3]. The most benign consequence of batch effects is an increase in the observed variability with a decreased sensitivity to detect biological differences. In the worst scenario, batch effects can mask completely the underlying biology in the experiment. Exploratory Data Analysis (EDA) helps in evidencing confounding factors and eventual outliers. In front of any -omics experiment it is always wise to perfom an EDA before any differential expression statistical analysis [4] [5]. The results of this exploratory analysis, visualized by PCA maps, HC trees, and heatmaps, will inform about the extent of batch effects and the presence of putative outliers. As a result the analyst may decide on the inclusion of a blocking factor to take into account the batch effects, or about the exclusion of a bad conditioned sample. 1
2 2 An example LC-MS/MS dataset The dataset of this example [5] is the result of an spiking experiment, showing LC- MS/MS data obtained in ideal conditions, and optimal to detect batch effects. Samples of 500 micrograms of a standard yeast lysate are spiked either with 200fm or 600fm of a complex mix of 48 human proteins (UPS1, Sigma-Aldrich ). The measures were done in two different runs, separated by a year time. The first run consisted of four replicates of each condition, and the second run consisted of three replicates of each condition. The dataset consists in an instance of the MSnSet class, defined in the MSnbase package [6], a S4 class [7] [8]. This MSnSet object contains a spectral counts (SpC) matrix in the assaydata slot, and factors treatment and batch in the phenodata slot. (See also the expressionset vignette [9]) > library(msmseda) > data(msms.dataset) > msms.dataset MSnSet (storagemode: lockedenvironment) assaydata: 697 features, 14 samples element names: exprs protocoldata: none phenodata samplenames: U (14 total) varlabels: treat batch varmetadata: labeldescription featuredata: none experimentdata: use 'experimentdata(object)' pubmedids: Annotation: Processing information MSnbase version: > dim(msms.dataset) [1] > head(pdata(msms.dataset)) treat batch U U U U U U > table(pdata(msms.dataset)$treat) 2
3 U200 U > table(pdata(msms.dataset)$batch) > table(pdata(msms.dataset)$treat, pdata(msms.dataset)$batch) U U The aim of the exploratory data analysis, in this case, is to evidence the existence of batch effects between the two runs. And eventually to check the opportunity of a simple batch effects correction. Before proceeding to the EDA, a data pre-processing is required to solve NAs and to remove improper rows in the spectral counts matrix. The NAs, common when joining datasets in which not exactly the same proteins are identified, should be substituted by 0. By improper rows to be removed, we mean: i) the rows with all zeroes, which could come from the subsetting from a bigger SpC matrix. ii) The rows belonging to artefactual identifications of -R proteins. > e <- pp.msms.data(msms.dataset) > processingdata(e) Processing information Subset [697,14][675,14] Mon Oct 13 20:46: Applied pp.msms.data preprocessing: Mon Oct 13 20:46: MSnbase version: > dim(e) [1] > setdiff(featurenames(msms.dataset), featurenames(e)) [1] "YER160C-R" "YJR066W-R" "YEL061C-R" "YJL190C (1)" "YLL057C" [6] "YBR189W" "YDR227W" "YER103W" "YGR029W" "YNR032C-A" [11] "YMR172W" "YJL200C" "YDL126C" "YGL173C" "YML037C" [16] "YHR102W" "YMR165C" "YJL138C (1)" "YBL046W" "YJL123C" [21] "YOR123C" "YGR276C" 3
4 3 SpC distribution A first glance to the contents of the spectral counts matrix is given by the distribution of SpC by sample, including the number of proteins identified and the total spectral counts by sample. > tfvnm <- count.stats(e) > { cat("\nsample statistics after removing NAs and -R:\n\n") cat("spc matrix dimension:",dim(e),"\n\n") print(tfvnm) } Sample statistics after removing NAs and -R: SpC matrix dimension: proteins counts min lwh med hgh max U U U U U U Three graphical means will contribute to visualize this distribution: i) A barplot of total SpC by sample scaled to the median. Ideally when the same amount of total protein is measured in each sample, the same total number of spectral counts will be observed. A good quality experiment will show all the bars near 1. ii) A set of SpC boxplots by sample. As most proteins in a sample may show very low signal, 0 or 1 SpC, resulting in sparce SpC vectors, more informative boxplots are obtained when removing the values below a given threshold. Here the proteins with SpC below minspc are excluded of a sample. The boxplots show the distribution of the remaining log 2 transformed SpC values by sample. iii) Superposed SpC density plots by sample. As before the proteins with SpC below minspc are excluded of each sample and the SpC are log 2 transformed. 4
5 > layout(mat=matrix(1:2,ncol=1),widths=1,heights=c(0.35,0.65)) > spc.barplots(exprs(e),fact=pdata(e)$treat) > spc.boxplots(exprs(e),fact=pdata(e)$treat,minspc=2, main="ups1 200fm vs 600fm") U U U U U U UPS1 200fm vs 600fm log2 SpC min SpC >= 2 U200 U U U U U U U Figure 1: A) Total SpC by sample scaled to the median. B) Samples SpC boxplots. 5
6 > spc.densityplots(exprs(e),fact=pdata(e)$treat,minspc=2, main="ups1 200fm vs 600fm") UPS1 200fm vs 600fm Density min SpC >= 2 U200 U log2 SpC Figure 2: Samples SpC density plots. 6
7 4 Principal Components Analysis A plot on the two principal components of the SpC matrix visualizes the clustering of samples. Ideally the samples belonging to the same condition should cluster together. Any mixing of samples of different conditions may indicate the influence of some confounding factors. PC % var PC1 PCA batch % var PC % var PC % var Figure 3: PCA plot on PC1/PC2, showing confounding. The labels are colored by treatment level on top, and by batch number bellow. Labels themselves are selfexplicative of treatment condition and batch. > facs <- pdata(e) > snms <- substr(as.character(facs$treat),1,2) > snms <- paste(snms,as.integer(facs$batch),sep=".") > pcares <- counts.pca(e) 7
8 > smpl.pca <- pcares$pca > { cat("principal components analisis on the raw SpC matrix\n") cat("variance of the first four principal components:\n\n") print(summary(smpl.pca)$importance[,1:4]) } Principal components analisis on the raw SpC matrix Variance of the first four principal components: PC1 PC2 PC3 PC4 Standard deviation Proportion of Variance Cumulative Proportion Note how in these plots the samples tend to cluster by batch instead of by treatment, this is evidence of a confounding factor. Something uncontrolled contributes globally to the results in a higer extend than the treatment itself. 5 Hierarchical clustering The hierarchical clustering of samples offers another view of the same phenomenon: > counts.hc(e,facs=pdata(e)[, "treat", drop = FALSE]) HC treat U U U U U U Figure 4: Hirearchical clustering of samples, showing confounding. The labels are colored by treatment level. 8
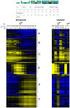
9 > counts.hc(e,facs=pdata(e)[, "batch", drop = FALSE]) HC batch U U U U U U Figure 5: Hirearchical clustering of samples, showing confounding. The labels are colored by batch number. 6 Heatmap A heatmap may be more informative than the dendrogram of samples, in the sense that it allows to identify the proteins most sensitive to this confounding. In this case we need a heatmap heigh enough to allow room for all the protein names. The function counts.heatmap provides two heatmaps, the first one is fitted on an A4 page and offers a general view, the second one is provided in a separate pdf file and is drawn with 3mm high rows to allow a confortable identification of each protein and its expression profile in the experiment. 9
10 > counts.heatmap(e,etit="ups1",fac=pdata(e)[, "treat"]) U U U U U U Figure 6: Global view heatmap. The column color bar is colored as per treatment levels. 7 Batch effects correction When counfounding is detected due to batch effects, as in this case, and the runs are balanced in the two conditions to be compared, blocking may help in the reduction of the residual variance, improving the sensitivity of the statistical tests. When dealing with spectral counts the usual model for the differential expression tests is a Generalized Linear Model (GLM) [10] based on the Poisson distribution, the negative binomial, or the quasi-likelihood, and these models admit blocking as the usual ANOVA [11] when dealing with normally distributed continous data. The visualization of the influence of a batch effects correction, in the exploratory data analysis step, is easily carried out by the so called mean centering approach [4], by which the centers of each batch are made to coincide concealing the observed bias between the different batches. The PCA on the batch mean centered expression matrix will then show the level of improvement. > data(msms.dataset) 10
11 > msnset <- pp.msms.data(msms.dataset) > spcm <- exprs(msnset) > fbatch <- pdata(msnset)$batch > spcm2 <- batch.neutralize(spcm, fbatch, half=true, sqrt.trans=true) > ### Plot the PCA on the two first PC, and colour by treatment level > ### to visualize the improvement. > exprs(msnset) <- spcm2 > facs <- pdata(e) > snms <- substr(as.character(facs$treat),1,2) > snms <- paste(snms,as.integer(facs$batch),sep=".") > par(mar=c(4,4,0.5,2)0.1) > counts.pca(msnset, facs=facs$treat, do.plot=true, snms=snms) PC % var PC % var Figure 7: PCA plot on the batch mean centered expression matrix, showing now a better clustering by treatment level. This plots shows a clear improvement in the clustering of samples by treatment condition, and suggest that a model with batch as block factor will give better results than a model just including the treatment factor. The incidence of the correction may be evaluated as: > ### Incidence of the correction > summary(as.vector(spcm-spcm2)) Min. 1st Qu. Median Mean 3rd Qu. Max > plot(density(as.vector(spcm-spcm2))) 11
12 8 Dispersion The simplest distribution used to explain the observed counts in sampling is the Poisson distribution. With this distribution the variance is equal to the mean, so that the dispersion coefficient -the ratio variance to mean- is one. When there are other sources of variation, apart of the sampling, such as the usual variability among biological replicates, we talk of overdispersion. In this situation the coefficient of dispersion is greater than one and the Poisson distribution is unable to explain this extra source of variance. Alternative GLM models able to explain overdispersion are based on the negative binomial distribution, or on the quasilikelihood [10]. An EDA of a dataset based on counts should include an exploration of the residual coefficients of dispersion for each of the factors in the experimental design. This will help in deciding the model to use in the inference step. The disp.estimates function plots the distribution of residual dispersion coefficients, and the scatterplot of residual variances vs mean SpC for each of the factors in the parameter facs, if this parameter is NULL then the factors are taken as default from the phenodata slot of the MSnSet object. > dsp <- disp.estimates(e) > signif(dsp,4) treat batch This function returns silently the quartiles and the quantiles at 0.9, 0.95 and 0.99 of the residual dispersion of each factor. With technical replicates it is not uncommon to observe most of the dispersion coefficients lower that one. This is a situation of underdispersion, most likely due to the competitive sampling at the MS system entrance. 12
13 Density Log SpC variance N = 675 Bandwidth = Residual dispersion factor batch Log SpC mean Figure 8: Residual dispersion density plot, and residual variance vs mean scatterplot in log10 scale, of the batch factor. 13
14 9 Informative features In the border between EDA and inference we may explore the number and distribution of informative features. We mean by informative features those proteins showing a high enough signal in the most abundant condition and with an absolute log fold change between treatment levels above a given threshold. The following plot shows in red the features with signal not bellow 2 SpC in the most abundant condition, and with minlfc of 1. To improve the plot two transformations are available, either log2 or sqrt, although none is also accepted. > spc.scatterplot(spcm2,facs$treat,trans="sqrt",minspc=2,minlfc=1, main="ups1 200fm vs 600fm") UPS1 200fm vs 600fm mean SpC(U600) mean SpC(U200) Figure 9: Scatterplot with informative features in red, showing the borders of fold change 2 and 0.5 as blue dash-dot lines. 14
15 References [1] Mallick P., Kuster B. Proteomics: a pragmatic perspective. Nat Biotechnol 2010;28: [2] Neilson K.A., Ali N.A., Muralidharan S., Mirzaei M., Mariani M., Assadourian G., et al. Less label, more free: approaches in label-free quantitative mass spectrometry. Proteomics 2011;11: [3] Husson F., Le S., Pages J. Exploratory Multivariate Analysis by Example Using R. CRC Press; [4] Luo J., Schumacher M., Scherer A., Sanoudou D., Megherbi D., Davison T., et al. A comparison of batch effect removal methods for enhancement of prediction performance using MAQC-II microarray gene expression data. Pharmacogenomics J 2010, 10(4): [5] Gregori J., Villareal L., Mendez O., Sanchez A., Baselga J., Villanueva J., Batch effects correction improves the sensitivity of significance tests in spectral countingbased comparative discovery proteomics, Journal of Proteomics, 2012, 75, [6] Laurent Gatto and Kathryn S. Lilley, MSnbase - an R/Bioconductor package for isobaric tagged mass spectrometry data visualization, processing and quantitation, Bioinformatics 28(2), (2012). [7] Chambers J.M. Software for data analysis: programming with R, 2008 Springer [8] Genolini C. A (Not So) Short Introduction to S4 (2008) [9] Falcon S., Morgan M., Gentleman R. An Introduction to Bioconductor s Expression- Set Class (2007) [10] Agresti A., Categorical Data Analysis, Wiley-Interscience, Hoboken NJ, 2002 [11] Kutner M.H., Nachtsheim C.J., Neter J., Li W., Applied Linear Statistical Models, Fifth Edition, McGraw-Hill Intl. Edition 2005 [12] Quinn G.P., Keough M.J. Experimental Design and Data Analysis for Biologists Cambridge University Press; 1st Edition, Cambridge
Tutorial for proteome data analysis using the Perseus software platform
 Tutorial for proteome data analysis using the Perseus software platform Laboratory of Mass Spectrometry, LNBio, CNPEM Tutorial version 1.0, January 2014. Note: This tutorial was written based on the information
Tutorial for proteome data analysis using the Perseus software platform Laboratory of Mass Spectrometry, LNBio, CNPEM Tutorial version 1.0, January 2014. Note: This tutorial was written based on the information
Lecture 2: Descriptive Statistics and Exploratory Data Analysis
 Lecture 2: Descriptive Statistics and Exploratory Data Analysis Further Thoughts on Experimental Design 16 Individuals (8 each from two populations) with replicates Pop 1 Pop 2 Randomly sample 4 individuals
Lecture 2: Descriptive Statistics and Exploratory Data Analysis Further Thoughts on Experimental Design 16 Individuals (8 each from two populations) with replicates Pop 1 Pop 2 Randomly sample 4 individuals
Gene Expression Analysis
 Gene Expression Analysis Jie Peng Department of Statistics University of California, Davis May 2012 RNA expression technologies High-throughput technologies to measure the expression levels of thousands
Gene Expression Analysis Jie Peng Department of Statistics University of California, Davis May 2012 RNA expression technologies High-throughput technologies to measure the expression levels of thousands
DeCyder Extended Data Analysis module Version 1.0
 GE Healthcare DeCyder Extended Data Analysis module Version 1.0 Module for DeCyder 2D version 6.5 User Manual Contents 1 Introduction 1.1 Introduction... 7 1.2 The DeCyder EDA User Manual... 9 1.3 Getting
GE Healthcare DeCyder Extended Data Analysis module Version 1.0 Module for DeCyder 2D version 6.5 User Manual Contents 1 Introduction 1.1 Introduction... 7 1.2 The DeCyder EDA User Manual... 9 1.3 Getting
Quantitative proteomics background
 Proteomics data analysis seminar Quantitative proteomics and transcriptomics of anaerobic and aerobic yeast cultures reveals post transcriptional regulation of key cellular processes de Groot, M., Daran
Proteomics data analysis seminar Quantitative proteomics and transcriptomics of anaerobic and aerobic yeast cultures reveals post transcriptional regulation of key cellular processes de Groot, M., Daran
STATISTICA Formula Guide: Logistic Regression. Table of Contents
 : Table of Contents... 1 Overview of Model... 1 Dispersion... 2 Parameterization... 3 Sigma-Restricted Model... 3 Overparameterized Model... 4 Reference Coding... 4 Model Summary (Summary Tab)... 5 Summary
: Table of Contents... 1 Overview of Model... 1 Dispersion... 2 Parameterization... 3 Sigma-Restricted Model... 3 Overparameterized Model... 4 Reference Coding... 4 Model Summary (Summary Tab)... 5 Summary
Hierarchical Clustering Analysis
 Hierarchical Clustering Analysis What is Hierarchical Clustering? Hierarchical clustering is used to group similar objects into clusters. In the beginning, each row and/or column is considered a cluster.
Hierarchical Clustering Analysis What is Hierarchical Clustering? Hierarchical clustering is used to group similar objects into clusters. In the beginning, each row and/or column is considered a cluster.
Statistical Analysis. NBAF-B Metabolomics Masterclass. Mark Viant
 Statistical Analysis NBAF-B Metabolomics Masterclass Mark Viant 1. Introduction 2. Univariate analysis Overview of lecture 3. Unsupervised multivariate analysis Principal components analysis (PCA) Interpreting
Statistical Analysis NBAF-B Metabolomics Masterclass Mark Viant 1. Introduction 2. Univariate analysis Overview of lecture 3. Unsupervised multivariate analysis Principal components analysis (PCA) Interpreting
Exploratory data analysis (Chapter 2) Fall 2011
 Exploratory data analysis (Chapter 2) Fall 2011 Data Examples Example 1: Survey Data 1 Data collected from a Stat 371 class in Fall 2005 2 They answered questions about their: gender, major, year in school,
Exploratory data analysis (Chapter 2) Fall 2011 Data Examples Example 1: Survey Data 1 Data collected from a Stat 371 class in Fall 2005 2 They answered questions about their: gender, major, year in school,
Multivariate Analysis of Ecological Data
 Multivariate Analysis of Ecological Data MICHAEL GREENACRE Professor of Statistics at the Pompeu Fabra University in Barcelona, Spain RAUL PRIMICERIO Associate Professor of Ecology, Evolutionary Biology
Multivariate Analysis of Ecological Data MICHAEL GREENACRE Professor of Statistics at the Pompeu Fabra University in Barcelona, Spain RAUL PRIMICERIO Associate Professor of Ecology, Evolutionary Biology
Statistical Analysis Strategies for Shotgun Proteomics Data
 Statistical Analysis Strategies for Shotgun Proteomics Data Ming Li, Ph.D. Cancer Biostatistics Center Vanderbilt University Medical Center Ayers Institute Biomarker Pipeline normal shotgun proteome analysis
Statistical Analysis Strategies for Shotgun Proteomics Data Ming Li, Ph.D. Cancer Biostatistics Center Vanderbilt University Medical Center Ayers Institute Biomarker Pipeline normal shotgun proteome analysis
Normalization of RNA-Seq
 Normalization of RNA-Seq Davide Risso Modified: April 27, 2012. Compiled: April 27, 2012 1 Retrieving the data Usually, an RNA-Seq data analysis from scratch starts with a set of FASTQ files (see e.g.
Normalization of RNA-Seq Davide Risso Modified: April 27, 2012. Compiled: April 27, 2012 1 Retrieving the data Usually, an RNA-Seq data analysis from scratch starts with a set of FASTQ files (see e.g.
EDASeq: Exploratory Data Analysis and Normalization for RNA-Seq
 EDASeq: Exploratory Data Analysis and Normalization for RNA-Seq Davide Risso Modified: May 22, 2012. Compiled: October 14, 2013 1 Introduction In this document, we show how to conduct Exploratory Data
EDASeq: Exploratory Data Analysis and Normalization for RNA-Seq Davide Risso Modified: May 22, 2012. Compiled: October 14, 2013 1 Introduction In this document, we show how to conduct Exploratory Data
OplAnalyzer: A Toolbox for MALDI-TOF Mass Spectrometry Data Analysis
 OplAnalyzer: A Toolbox for MALDI-TOF Mass Spectrometry Data Analysis Thang V. Pham and Connie R. Jimenez OncoProteomics Laboratory, Cancer Center Amsterdam, VU University Medical Center De Boelelaan 1117,
OplAnalyzer: A Toolbox for MALDI-TOF Mass Spectrometry Data Analysis Thang V. Pham and Connie R. Jimenez OncoProteomics Laboratory, Cancer Center Amsterdam, VU University Medical Center De Boelelaan 1117,
WebFOCUS RStat. RStat. Predict the Future and Make Effective Decisions Today. WebFOCUS RStat
 Information Builders enables agile information solutions with business intelligence (BI) and integration technologies. WebFOCUS the most widely utilized business intelligence platform connects to any enterprise
Information Builders enables agile information solutions with business intelligence (BI) and integration technologies. WebFOCUS the most widely utilized business intelligence platform connects to any enterprise
Why Taking This Course? Course Introduction, Descriptive Statistics and Data Visualization. Learning Goals. GENOME 560, Spring 2012
 Why Taking This Course? Course Introduction, Descriptive Statistics and Data Visualization GENOME 560, Spring 2012 Data are interesting because they help us understand the world Genomics: Massive Amounts
Why Taking This Course? Course Introduction, Descriptive Statistics and Data Visualization GENOME 560, Spring 2012 Data are interesting because they help us understand the world Genomics: Massive Amounts
Principal Component Analysis
 Principal Component Analysis ERS70D George Fernandez INTRODUCTION Analysis of multivariate data plays a key role in data analysis. Multivariate data consists of many different attributes or variables recorded
Principal Component Analysis ERS70D George Fernandez INTRODUCTION Analysis of multivariate data plays a key role in data analysis. Multivariate data consists of many different attributes or variables recorded
STATS8: Introduction to Biostatistics. Data Exploration. Babak Shahbaba Department of Statistics, UCI
 STATS8: Introduction to Biostatistics Data Exploration Babak Shahbaba Department of Statistics, UCI Introduction After clearly defining the scientific problem, selecting a set of representative members
STATS8: Introduction to Biostatistics Data Exploration Babak Shahbaba Department of Statistics, UCI Introduction After clearly defining the scientific problem, selecting a set of representative members
From Reads to Differentially Expressed Genes. The statistics of differential gene expression analysis using RNA-seq data
 From Reads to Differentially Expressed Genes The statistics of differential gene expression analysis using RNA-seq data experimental design data collection modeling statistical testing biological heterogeneity
From Reads to Differentially Expressed Genes The statistics of differential gene expression analysis using RNA-seq data experimental design data collection modeling statistical testing biological heterogeneity
AGILENT S BIOINFORMATICS ANALYSIS SOFTWARE
 ACCELERATING PROGRESS IS IN OUR GENES AGILENT S BIOINFORMATICS ANALYSIS SOFTWARE GENESPRING GENE EXPRESSION (GX) MASS PROFILER PROFESSIONAL (MPP) PATHWAY ARCHITECT (PA) See Deeper. Reach Further. BIOINFORMATICS
ACCELERATING PROGRESS IS IN OUR GENES AGILENT S BIOINFORMATICS ANALYSIS SOFTWARE GENESPRING GENE EXPRESSION (GX) MASS PROFILER PROFESSIONAL (MPP) PATHWAY ARCHITECT (PA) See Deeper. Reach Further. BIOINFORMATICS
How To Cluster
 Data Clustering Dec 2nd, 2013 Kyrylo Bessonov Talk outline Introduction to clustering Types of clustering Supervised Unsupervised Similarity measures Main clustering algorithms k-means Hierarchical Main
Data Clustering Dec 2nd, 2013 Kyrylo Bessonov Talk outline Introduction to clustering Types of clustering Supervised Unsupervised Similarity measures Main clustering algorithms k-means Hierarchical Main
Identification of rheumatoid arthritis and osteoarthritis patients by transcriptome-based rule set generation
 Identification of rheumatoid arthritis and osterthritis patients by transcriptome-based rule set generation Bering Limited Report generated on September 19, 2014 Contents 1 Dataset summary 2 1.1 Project
Identification of rheumatoid arthritis and osterthritis patients by transcriptome-based rule set generation Bering Limited Report generated on September 19, 2014 Contents 1 Dataset summary 2 1.1 Project
Un (bref) aperçu des méthodes et outils de fouilles et de visualisation de données «omics»
 Un (bref) aperçu des méthodes et outils de fouilles et de visualisation de données «omics» Workshop «Protéomique & Maladies rares» 25 th September 2012, Paris yves.vandenbrouck@cea.fr CEA Grenoble irtsv
Un (bref) aperçu des méthodes et outils de fouilles et de visualisation de données «omics» Workshop «Protéomique & Maladies rares» 25 th September 2012, Paris yves.vandenbrouck@cea.fr CEA Grenoble irtsv
Automated Biosurveillance Data from England and Wales, 1991 2011
 Article DOI: http://dx.doi.org/10.3201/eid1901.120493 Automated Biosurveillance Data from England and Wales, 1991 2011 Technical Appendix This online appendix provides technical details of statistical
Article DOI: http://dx.doi.org/10.3201/eid1901.120493 Automated Biosurveillance Data from England and Wales, 1991 2011 Technical Appendix This online appendix provides technical details of statistical
A Handbook of Statistical Analyses Using R. Brian S. Everitt and Torsten Hothorn
 A Handbook of Statistical Analyses Using R Brian S. Everitt and Torsten Hothorn CHAPTER 6 Logistic Regression and Generalised Linear Models: Blood Screening, Women s Role in Society, and Colonic Polyps
A Handbook of Statistical Analyses Using R Brian S. Everitt and Torsten Hothorn CHAPTER 6 Logistic Regression and Generalised Linear Models: Blood Screening, Women s Role in Society, and Colonic Polyps
MarkerView Software 1.2.1 for Metabolomic and Biomarker Profiling Analysis
 MarkerView Software 1.2.1 for Metabolomic and Biomarker Profiling Analysis Overview MarkerView software is a novel program designed for metabolomics applications and biomarker profiling workflows 1. Using
MarkerView Software 1.2.1 for Metabolomic and Biomarker Profiling Analysis Overview MarkerView software is a novel program designed for metabolomics applications and biomarker profiling workflows 1. Using
Analyzing the Effect of Treatment and Time on Gene Expression in Partek Genomics Suite (PGS) 6.6: A Breast Cancer Study
 Analyzing the Effect of Treatment and Time on Gene Expression in Partek Genomics Suite (PGS) 6.6: A Breast Cancer Study The data for this study is taken from experiment GSE848 from the Gene Expression
Analyzing the Effect of Treatment and Time on Gene Expression in Partek Genomics Suite (PGS) 6.6: A Breast Cancer Study The data for this study is taken from experiment GSE848 from the Gene Expression
Data, Measurements, Features
 Data, Measurements, Features Middle East Technical University Dep. of Computer Engineering 2009 compiled by V. Atalay What do you think of when someone says Data? We might abstract the idea that data are
Data, Measurements, Features Middle East Technical University Dep. of Computer Engineering 2009 compiled by V. Atalay What do you think of when someone says Data? We might abstract the idea that data are
Practical Differential Gene Expression. Introduction
 Practical Differential Gene Expression Introduction In this tutorial you will learn how to use R packages for analysis of differential expression. The dataset we use are the gene-summarized count data
Practical Differential Gene Expression Introduction In this tutorial you will learn how to use R packages for analysis of differential expression. The dataset we use are the gene-summarized count data
Assumptions. Assumptions of linear models. Boxplot. Data exploration. Apply to response variable. Apply to error terms from linear model
 Assumptions Assumptions of linear models Apply to response variable within each group if predictor categorical Apply to error terms from linear model check by analysing residuals Normality Homogeneity
Assumptions Assumptions of linear models Apply to response variable within each group if predictor categorical Apply to error terms from linear model check by analysing residuals Normality Homogeneity
Tutorial 3: Graphics and Exploratory Data Analysis in R Jason Pienaar and Tom Miller
 Tutorial 3: Graphics and Exploratory Data Analysis in R Jason Pienaar and Tom Miller Getting to know the data An important first step before performing any kind of statistical analysis is to familiarize
Tutorial 3: Graphics and Exploratory Data Analysis in R Jason Pienaar and Tom Miller Getting to know the data An important first step before performing any kind of statistical analysis is to familiarize
Descriptive statistics Statistical inference statistical inference, statistical induction and inferential statistics
 Descriptive statistics is the discipline of quantitatively describing the main features of a collection of data. Descriptive statistics are distinguished from inferential statistics (or inductive statistics),
Descriptive statistics is the discipline of quantitatively describing the main features of a collection of data. Descriptive statistics are distinguished from inferential statistics (or inductive statistics),
MASCOT Search Results Interpretation
 The Mascot protein identification program (Matrix Science, Ltd.) uses statistical methods to assess the validity of a match. MS/MS data is not ideal. That is, there are unassignable peaks (noise) and usually
The Mascot protein identification program (Matrix Science, Ltd.) uses statistical methods to assess the validity of a match. MS/MS data is not ideal. That is, there are unassignable peaks (noise) and usually
MTH 140 Statistics Videos
 MTH 140 Statistics Videos Chapter 1 Picturing Distributions with Graphs Individuals and Variables Categorical Variables: Pie Charts and Bar Graphs Categorical Variables: Pie Charts and Bar Graphs Quantitative
MTH 140 Statistics Videos Chapter 1 Picturing Distributions with Graphs Individuals and Variables Categorical Variables: Pie Charts and Bar Graphs Categorical Variables: Pie Charts and Bar Graphs Quantitative
Statistical issues in the analysis of microarray data
 Statistical issues in the analysis of microarray data Daniel Gerhard Institute of Biostatistics Leibniz University of Hannover ESNATS Summerschool, Zermatt D. Gerhard (LUH) Analysis of microarray data
Statistical issues in the analysis of microarray data Daniel Gerhard Institute of Biostatistics Leibniz University of Hannover ESNATS Summerschool, Zermatt D. Gerhard (LUH) Analysis of microarray data
ProteinScape. Innovation with Integrity. Proteomics Data Analysis & Management. Mass Spectrometry
 ProteinScape Proteomics Data Analysis & Management Innovation with Integrity Mass Spectrometry ProteinScape a Virtual Environment for Successful Proteomics To overcome the growing complexity of proteomics
ProteinScape Proteomics Data Analysis & Management Innovation with Integrity Mass Spectrometry ProteinScape a Virtual Environment for Successful Proteomics To overcome the growing complexity of proteomics
Business Statistics. Successful completion of Introductory and/or Intermediate Algebra courses is recommended before taking Business Statistics.
 Business Course Text Bowerman, Bruce L., Richard T. O'Connell, J. B. Orris, and Dawn C. Porter. Essentials of Business, 2nd edition, McGraw-Hill/Irwin, 2008, ISBN: 978-0-07-331988-9. Required Computing
Business Course Text Bowerman, Bruce L., Richard T. O'Connell, J. B. Orris, and Dawn C. Porter. Essentials of Business, 2nd edition, McGraw-Hill/Irwin, 2008, ISBN: 978-0-07-331988-9. Required Computing
Service courses for graduate students in degree programs other than the MS or PhD programs in Biostatistics.
 Course Catalog In order to be assured that all prerequisites are met, students must acquire a permission number from the education coordinator prior to enrolling in any Biostatistics course. Courses are
Course Catalog In order to be assured that all prerequisites are met, students must acquire a permission number from the education coordinator prior to enrolling in any Biostatistics course. Courses are
How To Run Statistical Tests in Excel
 How To Run Statistical Tests in Excel Microsoft Excel is your best tool for storing and manipulating data, calculating basic descriptive statistics such as means and standard deviations, and conducting
How To Run Statistical Tests in Excel Microsoft Excel is your best tool for storing and manipulating data, calculating basic descriptive statistics such as means and standard deviations, and conducting
Choices, choices, choices... Which sequence database? Which modifications? What mass tolerance?
 Optimization 1 Choices, choices, choices... Which sequence database? Which modifications? What mass tolerance? Where to begin? 2 Sequence Databases Swiss-prot MSDB, NCBI nr dbest Species specific ORFS
Optimization 1 Choices, choices, choices... Which sequence database? Which modifications? What mass tolerance? Where to begin? 2 Sequence Databases Swiss-prot MSDB, NCBI nr dbest Species specific ORFS
Alignment and Preprocessing for Data Analysis
 Alignment and Preprocessing for Data Analysis Preprocessing tools for chromatography Basics of alignment GC FID (D) data and issues PCA F Ratios GC MS (D) data and issues PCA F Ratios PARAFAC Piecewise
Alignment and Preprocessing for Data Analysis Preprocessing tools for chromatography Basics of alignment GC FID (D) data and issues PCA F Ratios GC MS (D) data and issues PCA F Ratios PARAFAC Piecewise
Fairfield Public Schools
 Mathematics Fairfield Public Schools AP Statistics AP Statistics BOE Approved 04/08/2014 1 AP STATISTICS Critical Areas of Focus AP Statistics is a rigorous course that offers advanced students an opportunity
Mathematics Fairfield Public Schools AP Statistics AP Statistics BOE Approved 04/08/2014 1 AP STATISTICS Critical Areas of Focus AP Statistics is a rigorous course that offers advanced students an opportunity
Session 1. Course Presentation: Mass spectrometry-based proteomics for molecular and cellular biologists
 Program Overview Session 1. Course Presentation: Mass spectrometry-based proteomics for molecular and cellular biologists Session 2. Principles of Mass Spectrometry Session 3. Mass spectrometry based proteomics
Program Overview Session 1. Course Presentation: Mass spectrometry-based proteomics for molecular and cellular biologists Session 2. Principles of Mass Spectrometry Session 3. Mass spectrometry based proteomics
The Scheduled MRM Algorithm Enables Intelligent Use of Retention Time During Multiple Reaction Monitoring
 The Scheduled MRM Algorithm Enables Intelligent Use of Retention Time During Multiple Reaction Monitoring Delivering up to 2500 MRM Transitions per LC Run Christie Hunter 1, Brigitte Simons 2 1 AB SCIEX,
The Scheduled MRM Algorithm Enables Intelligent Use of Retention Time During Multiple Reaction Monitoring Delivering up to 2500 MRM Transitions per LC Run Christie Hunter 1, Brigitte Simons 2 1 AB SCIEX,
BioVisualization: Enhancing Clinical Data Mining
 BioVisualization: Enhancing Clinical Data Mining Even as many clinicians struggle to give up their pen and paper charts and spreadsheets, some innovators are already shifting health care information technology
BioVisualization: Enhancing Clinical Data Mining Even as many clinicians struggle to give up their pen and paper charts and spreadsheets, some innovators are already shifting health care information technology
Exercise 1.12 (Pg. 22-23)
 Individuals: The objects that are described by a set of data. They may be people, animals, things, etc. (Also referred to as Cases or Records) Variables: The characteristics recorded about each individual.
Individuals: The objects that are described by a set of data. They may be people, animals, things, etc. (Also referred to as Cases or Records) Variables: The characteristics recorded about each individual.
Analysis of Bead-summary Data using beadarray
 Analysis of Bead-summary Data using beadarray Mark Dunning October 13, 2015 Contents 1 Introduction 2 2 feature and pheno data 3 3 Subsetting the data 4 4 Exploratory analysis using boxplots 7 4.1 A note
Analysis of Bead-summary Data using beadarray Mark Dunning October 13, 2015 Contents 1 Introduction 2 2 feature and pheno data 3 3 Subsetting the data 4 4 Exploratory analysis using boxplots 7 4.1 A note
Exploratory Data Analysis
 Exploratory Data Analysis Johannes Schauer johannes.schauer@tugraz.at Institute of Statistics Graz University of Technology Steyrergasse 17/IV, 8010 Graz www.statistics.tugraz.at February 12, 2008 Introduction
Exploratory Data Analysis Johannes Schauer johannes.schauer@tugraz.at Institute of Statistics Graz University of Technology Steyrergasse 17/IV, 8010 Graz www.statistics.tugraz.at February 12, 2008 Introduction
ProteinPilot Report for ProteinPilot Software
 ProteinPilot Report for ProteinPilot Software Detailed Analysis of Protein Identification / Quantitation Results Automatically Sean L Seymour, Christie Hunter SCIEX, USA Pow erful mass spectrometers like
ProteinPilot Report for ProteinPilot Software Detailed Analysis of Protein Identification / Quantitation Results Automatically Sean L Seymour, Christie Hunter SCIEX, USA Pow erful mass spectrometers like
Increasing the Multiplexing of High Resolution Targeted Peptide Quantification Assays
 Increasing the Multiplexing of High Resolution Targeted Peptide Quantification Assays Scheduled MRM HR Workflow on the TripleTOF Systems Jenny Albanese, Christie Hunter AB SCIEX, USA Targeted quantitative
Increasing the Multiplexing of High Resolution Targeted Peptide Quantification Assays Scheduled MRM HR Workflow on the TripleTOF Systems Jenny Albanese, Christie Hunter AB SCIEX, USA Targeted quantitative
Exiqon Array Software Manual. Quick guide to data extraction from mircury LNA microrna Arrays
 Exiqon Array Software Manual Quick guide to data extraction from mircury LNA microrna Arrays March 2010 Table of contents Introduction Overview...................................................... 3 ImaGene
Exiqon Array Software Manual Quick guide to data extraction from mircury LNA microrna Arrays March 2010 Table of contents Introduction Overview...................................................... 3 ImaGene
Analyzing microrna Data and Integrating mirna with Gene Expression Data in Partek Genomics Suite 6.6
 Analyzing microrna Data and Integrating mirna with Gene Expression Data in Partek Genomics Suite 6.6 Overview This tutorial outlines how microrna data can be analyzed within Partek Genomics Suite. Additionally,
Analyzing microrna Data and Integrating mirna with Gene Expression Data in Partek Genomics Suite 6.6 Overview This tutorial outlines how microrna data can be analyzed within Partek Genomics Suite. Additionally,
Quality Assessment of Exon and Gene Arrays
 Quality Assessment of Exon and Gene Arrays I. Introduction In this white paper we describe some quality assessment procedures that are computed from CEL files from Whole Transcript (WT) based arrays such
Quality Assessment of Exon and Gene Arrays I. Introduction In this white paper we describe some quality assessment procedures that are computed from CEL files from Whole Transcript (WT) based arrays such
Proteomic Analysis using Accurate Mass Tags. Gordon Anderson PNNL January 4-5, 2005
 Proteomic Analysis using Accurate Mass Tags Gordon Anderson PNNL January 4-5, 2005 Outline Accurate Mass and Time Tag (AMT) based proteomics Instrumentation Data analysis Data management Challenges 2 Approach
Proteomic Analysis using Accurate Mass Tags Gordon Anderson PNNL January 4-5, 2005 Outline Accurate Mass and Time Tag (AMT) based proteomics Instrumentation Data analysis Data management Challenges 2 Approach
Design of Experiments (DOE)
 MINITAB ASSISTANT WHITE PAPER This paper explains the research conducted by Minitab statisticians to develop the methods and data checks used in the Assistant in Minitab 17 Statistical Software. Design
MINITAB ASSISTANT WHITE PAPER This paper explains the research conducted by Minitab statisticians to develop the methods and data checks used in the Assistant in Minitab 17 Statistical Software. Design
Example: Credit card default, we may be more interested in predicting the probabilty of a default than classifying individuals as default or not.
 Statistical Learning: Chapter 4 Classification 4.1 Introduction Supervised learning with a categorical (Qualitative) response Notation: - Feature vector X, - qualitative response Y, taking values in C
Statistical Learning: Chapter 4 Classification 4.1 Introduction Supervised learning with a categorical (Qualitative) response Notation: - Feature vector X, - qualitative response Y, taking values in C
Diagrams and Graphs of Statistical Data
 Diagrams and Graphs of Statistical Data One of the most effective and interesting alternative way in which a statistical data may be presented is through diagrams and graphs. There are several ways in
Diagrams and Graphs of Statistical Data One of the most effective and interesting alternative way in which a statistical data may be presented is through diagrams and graphs. There are several ways in
Software and Methods for the Analysis of Affymetrix GeneChip Data. Rafael A Irizarry Department of Biostatistics Johns Hopkins University
 Software and Methods for the Analysis of Affymetrix GeneChip Data Rafael A Irizarry Department of Biostatistics Johns Hopkins University Outline Overview Bioconductor Project Examples 1: Gene Annotation
Software and Methods for the Analysis of Affymetrix GeneChip Data Rafael A Irizarry Department of Biostatistics Johns Hopkins University Outline Overview Bioconductor Project Examples 1: Gene Annotation
Course Text. Required Computing Software. Course Description. Course Objectives. StraighterLine. Business Statistics
 Course Text Business Statistics Lind, Douglas A., Marchal, William A. and Samuel A. Wathen. Basic Statistics for Business and Economics, 7th edition, McGraw-Hill/Irwin, 2010, ISBN: 9780077384470 [This
Course Text Business Statistics Lind, Douglas A., Marchal, William A. and Samuel A. Wathen. Basic Statistics for Business and Economics, 7th edition, McGraw-Hill/Irwin, 2010, ISBN: 9780077384470 [This
Data Exploration Data Visualization
 Data Exploration Data Visualization What is data exploration? A preliminary exploration of the data to better understand its characteristics. Key motivations of data exploration include Helping to select
Data Exploration Data Visualization What is data exploration? A preliminary exploration of the data to better understand its characteristics. Key motivations of data exploration include Helping to select
A Streamlined Workflow for Untargeted Metabolomics
 A Streamlined Workflow for Untargeted Metabolomics Employing XCMS plus, a Simultaneous Data Processing and Metabolite Identification Software Package for Rapid Untargeted Metabolite Screening Baljit K.
A Streamlined Workflow for Untargeted Metabolomics Employing XCMS plus, a Simultaneous Data Processing and Metabolite Identification Software Package for Rapid Untargeted Metabolite Screening Baljit K.
GENEGOBI : VISUAL DATA ANALYSIS AID TOOLS FOR MICROARRAY DATA
 COMPSTAT 2004 Symposium c Physica-Verlag/Springer 2004 GENEGOBI : VISUAL DATA ANALYSIS AID TOOLS FOR MICROARRAY DATA Eun-kyung Lee, Dianne Cook, Eve Wurtele, Dongshin Kim, Jihong Kim, and Hogeun An Key
COMPSTAT 2004 Symposium c Physica-Verlag/Springer 2004 GENEGOBI : VISUAL DATA ANALYSIS AID TOOLS FOR MICROARRAY DATA Eun-kyung Lee, Dianne Cook, Eve Wurtele, Dongshin Kim, Jihong Kim, and Hogeun An Key
Learning Objectives:
 Proteomics Methodology for LC-MS/MS Data Analysis Methodology for LC-MS/MS Data Analysis Peptide mass spectrum data of individual protein obtained from LC-MS/MS has to be analyzed for identification of
Proteomics Methodology for LC-MS/MS Data Analysis Methodology for LC-MS/MS Data Analysis Peptide mass spectrum data of individual protein obtained from LC-MS/MS has to be analyzed for identification of
Exploratory data analysis for microarray data
 Eploratory data analysis for microarray data Anja von Heydebreck Ma Planck Institute for Molecular Genetics, Dept. Computational Molecular Biology, Berlin, Germany heydebre@molgen.mpg.de Visualization
Eploratory data analysis for microarray data Anja von Heydebreck Ma Planck Institute for Molecular Genetics, Dept. Computational Molecular Biology, Berlin, Germany heydebre@molgen.mpg.de Visualization
2. Filling Data Gaps, Data validation & Descriptive Statistics
 2. Filling Data Gaps, Data validation & Descriptive Statistics Dr. Prasad Modak Background Data collected from field may suffer from these problems Data may contain gaps ( = no readings during this period)
2. Filling Data Gaps, Data validation & Descriptive Statistics Dr. Prasad Modak Background Data collected from field may suffer from these problems Data may contain gaps ( = no readings during this period)
Application Note # LCMS-81 Introducing New Proteomics Acquisiton Strategies with the compact Towards the Universal Proteomics Acquisition Method
 Application Note # LCMS-81 Introducing New Proteomics Acquisiton Strategies with the compact Towards the Universal Proteomics Acquisition Method Introduction During the last decade, the complexity of samples
Application Note # LCMS-81 Introducing New Proteomics Acquisiton Strategies with the compact Towards the Universal Proteomics Acquisition Method Introduction During the last decade, the complexity of samples
ALLEN Mouse Brain Atlas
 TECHNICAL WHITE PAPER: QUALITY CONTROL STANDARDS FOR HIGH-THROUGHPUT RNA IN SITU HYBRIDIZATION DATA GENERATION Consistent data quality and internal reproducibility are critical concerns for high-throughput
TECHNICAL WHITE PAPER: QUALITY CONTROL STANDARDS FOR HIGH-THROUGHPUT RNA IN SITU HYBRIDIZATION DATA GENERATION Consistent data quality and internal reproducibility are critical concerns for high-throughput
430 Statistics and Financial Mathematics for Business
 Prescription: 430 Statistics and Financial Mathematics for Business Elective prescription Level 4 Credit 20 Version 2 Aim Students will be able to summarise, analyse, interpret and present data, make predictions
Prescription: 430 Statistics and Financial Mathematics for Business Elective prescription Level 4 Credit 20 Version 2 Aim Students will be able to summarise, analyse, interpret and present data, make predictions
SAS Software to Fit the Generalized Linear Model
 SAS Software to Fit the Generalized Linear Model Gordon Johnston, SAS Institute Inc., Cary, NC Abstract In recent years, the class of generalized linear models has gained popularity as a statistical modeling
SAS Software to Fit the Generalized Linear Model Gordon Johnston, SAS Institute Inc., Cary, NC Abstract In recent years, the class of generalized linear models has gained popularity as a statistical modeling
Geostatistics Exploratory Analysis
 Instituto Superior de Estatística e Gestão de Informação Universidade Nova de Lisboa Master of Science in Geospatial Technologies Geostatistics Exploratory Analysis Carlos Alberto Felgueiras cfelgueiras@isegi.unl.pt
Instituto Superior de Estatística e Gestão de Informação Universidade Nova de Lisboa Master of Science in Geospatial Technologies Geostatistics Exploratory Analysis Carlos Alberto Felgueiras cfelgueiras@isegi.unl.pt
Microarray Data Analysis. A step by step analysis using BRB-Array Tools
 Microarray Data Analysis A step by step analysis using BRB-Array Tools 1 EXAMINATION OF DIFFERENTIAL GENE EXPRESSION (1) Objective: to find genes whose expression is changed before and after chemotherapy.
Microarray Data Analysis A step by step analysis using BRB-Array Tools 1 EXAMINATION OF DIFFERENTIAL GENE EXPRESSION (1) Objective: to find genes whose expression is changed before and after chemotherapy.
Scatter Plots with Error Bars
 Chapter 165 Scatter Plots with Error Bars Introduction The procedure extends the capability of the basic scatter plot by allowing you to plot the variability in Y and X corresponding to each point. Each
Chapter 165 Scatter Plots with Error Bars Introduction The procedure extends the capability of the basic scatter plot by allowing you to plot the variability in Y and X corresponding to each point. Each
Classification and Regression by randomforest
 Vol. 2/3, December 02 18 Classification and Regression by randomforest Andy Liaw and Matthew Wiener Introduction Recently there has been a lot of interest in ensemble learning methods that generate many
Vol. 2/3, December 02 18 Classification and Regression by randomforest Andy Liaw and Matthew Wiener Introduction Recently there has been a lot of interest in ensemble learning methods that generate many
NCSS Statistical Software Principal Components Regression. In ordinary least squares, the regression coefficients are estimated using the formula ( )
 Chapter 340 Principal Components Regression Introduction is a technique for analyzing multiple regression data that suffer from multicollinearity. When multicollinearity occurs, least squares estimates
Chapter 340 Principal Components Regression Introduction is a technique for analyzing multiple regression data that suffer from multicollinearity. When multicollinearity occurs, least squares estimates
The Open2Dprot Proteomics Project for n-dimensional Protein Expression Data Analysis
 The Open2Dprot Proteomics Project for n-dimensional Protein Expression Data Analysis http://open2dprot.sourceforge.net/ Revised 2-05-2006 * (cf. 2D-LC) Introduction There is a need for integrated proteomics
The Open2Dprot Proteomics Project for n-dimensional Protein Expression Data Analysis http://open2dprot.sourceforge.net/ Revised 2-05-2006 * (cf. 2D-LC) Introduction There is a need for integrated proteomics
Introduction. Chapter 1. 1.1 Before you start. 1.1.1 Formulation
 Chapter 1 Introduction 1.1 Before you start Statistics starts with a problem, continues with the collection of data, proceeds with the data analysis and finishes with conclusions. It is a common mistake
Chapter 1 Introduction 1.1 Before you start Statistics starts with a problem, continues with the collection of data, proceeds with the data analysis and finishes with conclusions. It is a common mistake
ASSURING THE QUALITY OF TEST RESULTS
 Page 1 of 12 Sections Included in this Document and Change History 1. Purpose 2. Scope 3. Responsibilities 4. Background 5. References 6. Procedure/(6. B changed Division of Field Science and DFS to Office
Page 1 of 12 Sections Included in this Document and Change History 1. Purpose 2. Scope 3. Responsibilities 4. Background 5. References 6. Procedure/(6. B changed Division of Field Science and DFS to Office
Protein Protein Interaction Networks
 Functional Pattern Mining from Genome Scale Protein Protein Interaction Networks Young-Rae Cho, Ph.D. Assistant Professor Department of Computer Science Baylor University it My Definition of Bioinformatics
Functional Pattern Mining from Genome Scale Protein Protein Interaction Networks Young-Rae Cho, Ph.D. Assistant Professor Department of Computer Science Baylor University it My Definition of Bioinformatics
BNG 202 Biomechanics Lab. Descriptive statistics and probability distributions I
 BNG 202 Biomechanics Lab Descriptive statistics and probability distributions I Overview The overall goal of this short course in statistics is to provide an introduction to descriptive and inferential
BNG 202 Biomechanics Lab Descriptive statistics and probability distributions I Overview The overall goal of this short course in statistics is to provide an introduction to descriptive and inferential
Environmental Remote Sensing GEOG 2021
 Environmental Remote Sensing GEOG 2021 Lecture 4 Image classification 2 Purpose categorising data data abstraction / simplification data interpretation mapping for land cover mapping use land cover class
Environmental Remote Sensing GEOG 2021 Lecture 4 Image classification 2 Purpose categorising data data abstraction / simplification data interpretation mapping for land cover mapping use land cover class
Analysis of Variance. MINITAB User s Guide 2 3-1
 3 Analysis of Variance Analysis of Variance Overview, 3-2 One-Way Analysis of Variance, 3-5 Two-Way Analysis of Variance, 3-11 Analysis of Means, 3-13 Overview of Balanced ANOVA and GLM, 3-18 Balanced
3 Analysis of Variance Analysis of Variance Overview, 3-2 One-Way Analysis of Variance, 3-5 Two-Way Analysis of Variance, 3-11 Analysis of Means, 3-13 Overview of Balanced ANOVA and GLM, 3-18 Balanced
Medical Information Management & Mining. You Chen Jan,15, 2013 You.chen@vanderbilt.edu
 Medical Information Management & Mining You Chen Jan,15, 2013 You.chen@vanderbilt.edu 1 Trees Building Materials Trees cannot be used to build a house directly. How can we transform trees to building materials?
Medical Information Management & Mining You Chen Jan,15, 2013 You.chen@vanderbilt.edu 1 Trees Building Materials Trees cannot be used to build a house directly. How can we transform trees to building materials?
Data Visualization in R
 Data Visualization in R L. Torgo ltorgo@fc.up.pt Faculdade de Ciências / LIAAD-INESC TEC, LA Universidade do Porto Oct, 2014 Introduction Motivation for Data Visualization Humans are outstanding at detecting
Data Visualization in R L. Torgo ltorgo@fc.up.pt Faculdade de Ciências / LIAAD-INESC TEC, LA Universidade do Porto Oct, 2014 Introduction Motivation for Data Visualization Humans are outstanding at detecting
 T O P I C 1 2 Techniques and tools for data analysis Preview Introduction In chapter 3 of Statistics In A Day different combinations of numbers and types of variables are presented. We go through these
T O P I C 1 2 Techniques and tools for data analysis Preview Introduction In chapter 3 of Statistics In A Day different combinations of numbers and types of variables are presented. We go through these
Statistics I for QBIC. Contents and Objectives. Chapters 1 7. Revised: August 2013
 Statistics I for QBIC Text Book: Biostatistics, 10 th edition, by Daniel & Cross Contents and Objectives Chapters 1 7 Revised: August 2013 Chapter 1: Nature of Statistics (sections 1.1-1.6) Objectives
Statistics I for QBIC Text Book: Biostatistics, 10 th edition, by Daniel & Cross Contents and Objectives Chapters 1 7 Revised: August 2013 Chapter 1: Nature of Statistics (sections 1.1-1.6) Objectives
Tutorial for Proteomics Data Submission. Katalin F. Medzihradszky Robert J. Chalkley UCSF
 Tutorial for Proteomics Data Submission Katalin F. Medzihradszky Robert J. Chalkley UCSF Why Have Guidelines? Large-scale proteomics studies create huge amounts of data. It is impossible/impractical to
Tutorial for Proteomics Data Submission Katalin F. Medzihradszky Robert J. Chalkley UCSF Why Have Guidelines? Large-scale proteomics studies create huge amounts of data. It is impossible/impractical to
Introduction to Proteomics
 Introduction to Proteomics Åsa Wheelock, Ph.D. Division of Respiratory Medicine & Karolinska Biomics Center asa.wheelock@ki.se In: Systems Biology and the Omics Cascade, Karolinska Institutet, June 9-13,
Introduction to Proteomics Åsa Wheelock, Ph.D. Division of Respiratory Medicine & Karolinska Biomics Center asa.wheelock@ki.se In: Systems Biology and the Omics Cascade, Karolinska Institutet, June 9-13,
Paper No 19. FINALTERM EXAMINATION Fall 2009 MTH302- Business Mathematics & Statistics (Session - 2) Ref No: Time: 120 min Marks: 80
 Paper No 19 FINALTERM EXAMINATION Fall 2009 MTH302- Business Mathematics & Statistics (Session - 2) Ref No: Time: 120 min Marks: 80 Question No: 1 ( Marks: 1 ) - Please choose one Scatterplots are used
Paper No 19 FINALTERM EXAMINATION Fall 2009 MTH302- Business Mathematics & Statistics (Session - 2) Ref No: Time: 120 min Marks: 80 Question No: 1 ( Marks: 1 ) - Please choose one Scatterplots are used
Step-by-Step Guide to Basic Expression Analysis and Normalization
 Step-by-Step Guide to Basic Expression Analysis and Normalization Page 1 Introduction This document shows you how to perform a basic analysis and normalization of your data. A full review of this document
Step-by-Step Guide to Basic Expression Analysis and Normalization Page 1 Introduction This document shows you how to perform a basic analysis and normalization of your data. A full review of this document
Predictive Gene Signature Selection for Adjuvant Chemotherapy in Non-Small Cell Lung Cancer Patients
 Predictive Gene Signature Selection for Adjuvant Chemotherapy in Non-Small Cell Lung Cancer Patients by Li Liu A practicum report submitted to the Department of Public Health Sciences in conformity with
Predictive Gene Signature Selection for Adjuvant Chemotherapy in Non-Small Cell Lung Cancer Patients by Li Liu A practicum report submitted to the Department of Public Health Sciences in conformity with
Package GEOquery. August 18, 2015
 Type Package Package GEOquery August 18, 2015 Title Get data from NCBI Gene Expression Omnibus (GEO) Version 2.34.0 Date 2014-09-28 Author Maintainer BugReports
Type Package Package GEOquery August 18, 2015 Title Get data from NCBI Gene Expression Omnibus (GEO) Version 2.34.0 Date 2014-09-28 Author Maintainer BugReports
How To Use Statgraphics Centurion Xvii (Version 17) On A Computer Or A Computer (For Free)
 Statgraphics Centurion XVII (currently in beta test) is a major upgrade to Statpoint's flagship data analysis and visualization product. It contains 32 new statistical procedures and significant upgrades
Statgraphics Centurion XVII (currently in beta test) is a major upgrade to Statpoint's flagship data analysis and visualization product. It contains 32 new statistical procedures and significant upgrades
Mass Spectrometry Signal Calibration for Protein Quantitation
 Cambridge Isotope Laboratories, Inc. www.isotope.com Proteomics Mass Spectrometry Signal Calibration for Protein Quantitation Michael J. MacCoss, PhD Associate Professor of Genome Sciences University of
Cambridge Isotope Laboratories, Inc. www.isotope.com Proteomics Mass Spectrometry Signal Calibration for Protein Quantitation Michael J. MacCoss, PhD Associate Professor of Genome Sciences University of
Statistics in Medicine Research Lecture Series CSMC Fall 2014
 Catherine Bresee, MS Senior Biostatistician Biostatistics & Bioinformatics Research Institute Statistics in Medicine Research Lecture Series CSMC Fall 2014 Overview Review concept of statistical power
Catherine Bresee, MS Senior Biostatistician Biostatistics & Bioinformatics Research Institute Statistics in Medicine Research Lecture Series CSMC Fall 2014 Overview Review concept of statistical power
Institute of Actuaries of India Subject CT3 Probability and Mathematical Statistics
 Institute of Actuaries of India Subject CT3 Probability and Mathematical Statistics For 2015 Examinations Aim The aim of the Probability and Mathematical Statistics subject is to provide a grounding in
Institute of Actuaries of India Subject CT3 Probability and Mathematical Statistics For 2015 Examinations Aim The aim of the Probability and Mathematical Statistics subject is to provide a grounding in
Analyzing data. Thomas LaToza. 05-899D: Human Aspects of Software Development (HASD) Spring, 2011. (C) Copyright Thomas D. LaToza
 Analyzing data Thomas LaToza 05-899D: Human Aspects of Software Development (HASD) Spring, 2011 (C) Copyright Thomas D. LaToza Today s lecture Last time Why would you do a study? Which type of study should
Analyzing data Thomas LaToza 05-899D: Human Aspects of Software Development (HASD) Spring, 2011 (C) Copyright Thomas D. LaToza Today s lecture Last time Why would you do a study? Which type of study should
Analysis of gene expression data. Ulf Leser and Philippe Thomas
 Analysis of gene expression data Ulf Leser and Philippe Thomas This Lecture Protein synthesis Microarray Idea Technologies Applications Problems Quality control Normalization Analysis next week! Ulf Leser:
Analysis of gene expression data Ulf Leser and Philippe Thomas This Lecture Protein synthesis Microarray Idea Technologies Applications Problems Quality control Normalization Analysis next week! Ulf Leser:
Exploratory Data Analysis with MATLAB
 Computer Science and Data Analysis Series Exploratory Data Analysis with MATLAB Second Edition Wendy L Martinez Angel R. Martinez Jeffrey L. Solka ( r ec) CRC Press VV J Taylor & Francis Group Boca Raton
Computer Science and Data Analysis Series Exploratory Data Analysis with MATLAB Second Edition Wendy L Martinez Angel R. Martinez Jeffrey L. Solka ( r ec) CRC Press VV J Taylor & Francis Group Boca Raton
Exploratory Data Analysis
 Exploratory Data Analysis Paul Cohen ISTA 370 Spring, 2012 Paul Cohen ISTA 370 () Exploratory Data Analysis Spring, 2012 1 / 46 Outline Data, revisited The purpose of exploratory data analysis Learning
Exploratory Data Analysis Paul Cohen ISTA 370 Spring, 2012 Paul Cohen ISTA 370 () Exploratory Data Analysis Spring, 2012 1 / 46 Outline Data, revisited The purpose of exploratory data analysis Learning
Survey, Statistics and Psychometrics Core Research Facility University of Nebraska-Lincoln. Log-Rank Test for More Than Two Groups
 Survey, Statistics and Psychometrics Core Research Facility University of Nebraska-Lincoln Log-Rank Test for More Than Two Groups Prepared by Harlan Sayles (SRAM) Revised by Julia Soulakova (Statistics)
Survey, Statistics and Psychometrics Core Research Facility University of Nebraska-Lincoln Log-Rank Test for More Than Two Groups Prepared by Harlan Sayles (SRAM) Revised by Julia Soulakova (Statistics)
