Big Bang Nucleosynthesis. Suggested Reading: Ryden, Chapter 10 Fields, 2006: The European Physical Journal A, Volume 27, Issue 1, pp.
|
|
|
- Myron Ralph Higgins
- 7 years ago
- Views:
Transcription
1 Big Bang Nucleosynthesis Suggested Reading: Ryden, Chapter Fields, 2006: The European Physical Journal A, Volume 27, Issue 1, pp.314
2 Elemental Abundances
3 Binding Energy/Nucleon 8 4 He B/A (MeV) H 2 0 from Ryden s book D 3 He 1 0 Figure.1: The binding energy per nucleon (B/A) as a function of the number of nucleons (protons and neutrons) in an atomic nucleus. Note the absence of nuclei at A = 5 and A = 8. A =Z+N
4 Main BBN Network Fields (2006)
5 BBN Calculation Ω B h 2 =WMAP Mass fraction n p 2 H 4 He 3 H 3 He Li Be Li C 11 B Be Li B Li 13 B 12 B 8 B Be Coc et al. (arxiv: ) Time (s) 25
6 Dependence on BaryontoPhoton Ratio Fields (2006)
7 Observation of D Abundance Ferlet et al. (ISM) Q Q Q PKS Burles & Tytler (1998) Fields (2006) Fig. 6. D/H abundances shown as a function of [Si/H]. Labels denote the background QSO, except for the local interstellar value (LISM; [28]).
8 Observation of 4 He Abundance Steigman (arxiv: ) Figure 2: Helium abundance (mass fractions, Y) determinations from the sample of extragalactic H II regions studied by Izotov & Thuan (20) [29] as a function of the corresponding oxygen abundances (O/H by number). The solid line is the Izotov & Thuan best fit to a linear Y versus O/H correlation.
9 Evolution of the primordial helium abundance Steigman, arxiv:
10 Observation of 7 Li Abundance Steigman (arxiv: ) Figure 2: The lithium abundances ([Li] 12+log(Li/H)) as a function of metallicity ([Fe/H]) as derived from observations of very metalpoor stars in the Galaxy. See thetextforreferences.thehorizontalband shows the ±1σ SBBN prediction.
11 y d s e c e ) s e n y o s f g r s n 2 y.] k d r s Ma BBN 0.22 vs CMB on η 3 He/H, D/H 7 Li/H Li 4 He D 3 He Coc (arxiv: ) Planck 1 η Fig. 1. (Color online) Abundances of 4 He D, 3 He and 7 Li (blue) as a function of the baryon over photon ratio (bottom) or baryonic density (top). The vertical areas corresponds to the WMAP (dot, black) and Planck (solid, yellow) baryonic densities while the horizontal areas (green) represent the adopted observational abundances; see text. The (red) dot dashed lines correspond to the extreme values of the effective neutrino families coming from CMB Planck study, N eff =(3.02, 3.70); see text. l. (2012); (g) Ade et al. (2013) 7 3 Li/H He/H, D/H Mass fraction He D 3 He 7 Li WMAP Planck Ω B h 2 1 η
Topic 3. Evidence for the Big Bang
 Topic 3 Primordial nucleosynthesis Evidence for the Big Bang! Back in the 1920s it was generally thought that the Universe was infinite! However a number of experimental observations started to question
Topic 3 Primordial nucleosynthesis Evidence for the Big Bang! Back in the 1920s it was generally thought that the Universe was infinite! However a number of experimental observations started to question
thermal history of the universe and big bang nucleosynthesis
 thermal history of the universe and big bang nucleosynthesis Kosmologie für Nichtphysiker Markus Pössel (vertreten durch Björn Malte Schäfer) Fakultät für Physik und Astronomie, Universität Heidelberg
thermal history of the universe and big bang nucleosynthesis Kosmologie für Nichtphysiker Markus Pössel (vertreten durch Björn Malte Schäfer) Fakultät für Physik und Astronomie, Universität Heidelberg
arxiv:astro-ph/0208186v1 8 Aug 2002
 Primordial Alchemy: From The Big Bang To The Present Universe By GARY STEIGMAN arxiv:astro-ph/0208186v1 8 Aug 2002 Departments of Physics and Astronomy, The Ohio State University, Columbus, OH 43210, USA
Primordial Alchemy: From The Big Bang To The Present Universe By GARY STEIGMAN arxiv:astro-ph/0208186v1 8 Aug 2002 Departments of Physics and Astronomy, The Ohio State University, Columbus, OH 43210, USA
Institut für Kern- und Teilchenphysik Neutrinos & Cosmology
 Neutrinos & Cosmology 1 Cosmology: WHY??? From laboratory experiment limits can be set ONLY in neutrino mass difference No information if neutrino masses are degenerated From kinematic experiment limits
Neutrinos & Cosmology 1 Cosmology: WHY??? From laboratory experiment limits can be set ONLY in neutrino mass difference No information if neutrino masses are degenerated From kinematic experiment limits
The Birth of the Universe Newcomer Academy High School Visualization One
 The Birth of the Universe Newcomer Academy High School Visualization One Chapter Topic Key Points of Discussion Notes & Vocabulary 1 Birth of The Big Bang Theory Activity 4A the How and when did the universe
The Birth of the Universe Newcomer Academy High School Visualization One Chapter Topic Key Points of Discussion Notes & Vocabulary 1 Birth of The Big Bang Theory Activity 4A the How and when did the universe
Chapter 23 The Beginning of Time
 Chapter 23 The Beginning of Time 23.1 The Big Bang Our goals for learning What were conditions like in the early universe? What is the history of the universe according to the Big Bang theory? What were
Chapter 23 The Beginning of Time 23.1 The Big Bang Our goals for learning What were conditions like in the early universe? What is the history of the universe according to the Big Bang theory? What were
Particle Soup: Big Bang Nucleosynthesis
 Name: Partner(s): Lab #7 Particle Soup: Big Bang Nucleosynthesis Purpose The student explores how helium was made in the Big Bang. Introduction Very little helium is made in stars. Yet the universe is
Name: Partner(s): Lab #7 Particle Soup: Big Bang Nucleosynthesis Purpose The student explores how helium was made in the Big Bang. Introduction Very little helium is made in stars. Yet the universe is
Big Bang Nucleosynthesis
 Big Bang Nucleosynthesis The emergence of elements in the universe Benjamin Topper Abstract. In this paper, I will first give a brief overview of what general relativity has to say about cosmology, getting
Big Bang Nucleosynthesis The emergence of elements in the universe Benjamin Topper Abstract. In this paper, I will first give a brief overview of what general relativity has to say about cosmology, getting
Nuclear Physics. Nuclear Physics comprises the study of:
 Nuclear Physics Nuclear Physics comprises the study of: The general properties of nuclei The particles contained in the nucleus The interaction between these particles Radioactivity and nuclear reactions
Nuclear Physics Nuclear Physics comprises the study of: The general properties of nuclei The particles contained in the nucleus The interaction between these particles Radioactivity and nuclear reactions
THE COSMIC PRODUCTION OF HELIUM
 TURUN YLIOPISTON JULKAISUJA ANNALES UNIVERSITATIS TURKUENSIS SARJA - SER. A I OSA - TOM. 382 ASTRONOMICA - CHEMICA - PHYSICA - MATHEMATICA THE COSMIC PRODUCTION OF HELIUM by Luca Casagrande TURUN YLIOPISTO
TURUN YLIOPISTON JULKAISUJA ANNALES UNIVERSITATIS TURKUENSIS SARJA - SER. A I OSA - TOM. 382 ASTRONOMICA - CHEMICA - PHYSICA - MATHEMATICA THE COSMIC PRODUCTION OF HELIUM by Luca Casagrande TURUN YLIOPISTO
Bohr s Model of the Atom
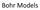 Bohr Models Bohr s Model of the Atom Focuses on electrons and their arrangement. Bohr stated that electrons move with constant speed in fixed orbits around the nucleus, like planets around a sun. Bohr
Bohr Models Bohr s Model of the Atom Focuses on electrons and their arrangement. Bohr stated that electrons move with constant speed in fixed orbits around the nucleus, like planets around a sun. Bohr
2 ATOMIC SYSTEMATICS AND NUCLEAR STRUCTURE
 2 ATOMIC SYSTEMATICS AND NUCLEAR STRUCTURE In this chapter the principles and systematics of atomic and nuclear physics are summarised briefly, in order to introduce the existence and characteristics of
2 ATOMIC SYSTEMATICS AND NUCLEAR STRUCTURE In this chapter the principles and systematics of atomic and nuclear physics are summarised briefly, in order to introduce the existence and characteristics of
............... [2] At the time of purchase of a Strontium-90 source, the activity is 3.7 10 6 Bq.
![............... [2] At the time of purchase of a Strontium-90 source, the activity is 3.7 10 6 Bq. ............... [2] At the time of purchase of a Strontium-90 source, the activity is 3.7 10 6 Bq.](/thumbs/40/21837699.jpg) 1 Strontium-90 decays with the emission of a β-particle to form Yttrium-90. The reaction is represented by the equation 90 38 The decay constant is 0.025 year 1. 90 39 0 1 Sr Y + e + 0.55 MeV. (a) Suggest,
1 Strontium-90 decays with the emission of a β-particle to form Yttrium-90. The reaction is represented by the equation 90 38 The decay constant is 0.025 year 1. 90 39 0 1 Sr Y + e + 0.55 MeV. (a) Suggest,
Main sequence stars. Haris Ðapo. Antalya Lecture 3. 1 Akdeniz University, Antalya
 Main sequence stars Haris Ðapo 1 Akdeniz University, Antalya Antalya Lecture 3. Haris Ðapo (Akdeniz University) Main sequence stars Main sequence stars 1 / 22 Outline 1 Introduction 2 Hydrogen burning
Main sequence stars Haris Ðapo 1 Akdeniz University, Antalya Antalya Lecture 3. Haris Ðapo (Akdeniz University) Main sequence stars Main sequence stars 1 / 22 Outline 1 Introduction 2 Hydrogen burning
Physics 1104 Midterm 2 Review: Solutions
 Physics 114 Midterm 2 Review: Solutions These review sheets cover only selected topics from the chemical and nuclear energy chapters and are not meant to be a comprehensive review. Topics covered in these
Physics 114 Midterm 2 Review: Solutions These review sheets cover only selected topics from the chemical and nuclear energy chapters and are not meant to be a comprehensive review. Topics covered in these
WHERE DID ALL THE ELEMENTS COME FROM??
 WHERE DID ALL THE ELEMENTS COME FROM?? In the very beginning, both space and time were created in the Big Bang. It happened 13.7 billion years ago. Afterwards, the universe was a very hot, expanding soup
WHERE DID ALL THE ELEMENTS COME FROM?? In the very beginning, both space and time were created in the Big Bang. It happened 13.7 billion years ago. Afterwards, the universe was a very hot, expanding soup
Atoms and Elements. Outline Atoms Orbitals and Energy Levels Periodic Properties Homework
 Atoms and the Periodic Table The very hot early universe was a plasma with cationic nuclei separated from negatively charged electrons. Plasmas exist today where the energy of the particles is very high,
Atoms and the Periodic Table The very hot early universe was a plasma with cationic nuclei separated from negatively charged electrons. Plasmas exist today where the energy of the particles is very high,
1 Introduction. 1 There may, of course, in principle, exist other universes, but they are not accessible to our
 1 1 Introduction Cosmology is the study of the universe as a whole, its structure, its origin, and its evolution. Cosmology is soundly based on observations, mostly astronomical, and laws of physics. These
1 1 Introduction Cosmology is the study of the universe as a whole, its structure, its origin, and its evolution. Cosmology is soundly based on observations, mostly astronomical, and laws of physics. These
Solar Energy Production
 Solar Energy Production We re now ready to address the very important question: What makes the Sun shine? Why is this such an important topic in astronomy? As humans, we see in the visible part of the
Solar Energy Production We re now ready to address the very important question: What makes the Sun shine? Why is this such an important topic in astronomy? As humans, we see in the visible part of the
Atoms and Elements. Atoms: Learning Goals. Chapter 3. Atoms and Elements; Isotopes and Ions; Minerals and Rocks. Clicker 1. Chemistry Background?
 Chapter 3 Atoms Atoms and Elements; Isotopes and Ions; Minerals and Rocks A Review of Chemistry: What geochemistry tells us Clicker 1 Chemistry Background? A. No HS or College Chemistry B. High School
Chapter 3 Atoms Atoms and Elements; Isotopes and Ions; Minerals and Rocks A Review of Chemistry: What geochemistry tells us Clicker 1 Chemistry Background? A. No HS or College Chemistry B. High School
The Universe Inside of You: Where do the atoms in your body come from?
 The Universe Inside of You: Where do the atoms in your body come from? Matthew Mumpower University of Notre Dame Thursday June 27th 2013 Nucleosynthesis nu cle o syn the sis The formation of new atomic
The Universe Inside of You: Where do the atoms in your body come from? Matthew Mumpower University of Notre Dame Thursday June 27th 2013 Nucleosynthesis nu cle o syn the sis The formation of new atomic
Big Bang Cosmology. Big Bang vs. Steady State
 Big Bang vs. Steady State Big Bang Cosmology Perfect cosmological principle: universe is unchanging in space and time => Steady-State universe - Bondi, Hoyle, Gold. True? No! Hubble s Law => expansion
Big Bang vs. Steady State Big Bang Cosmology Perfect cosmological principle: universe is unchanging in space and time => Steady-State universe - Bondi, Hoyle, Gold. True? No! Hubble s Law => expansion
Nuclear Physics and Radioactivity
 Nuclear Physics and Radioactivity 1. The number of electrons in an atom of atomic number Z and mass number A is 1) A 2) Z 3) A+Z 4) A-Z 2. The repulsive force between the positively charged protons does
Nuclear Physics and Radioactivity 1. The number of electrons in an atom of atomic number Z and mass number A is 1) A 2) Z 3) A+Z 4) A-Z 2. The repulsive force between the positively charged protons does
Untitled Document. 1. Which of the following best describes an atom? 4. Which statement best describes the density of an atom s nucleus?
 Name: Date: 1. Which of the following best describes an atom? A. protons and electrons grouped together in a random pattern B. protons and electrons grouped together in an alternating pattern C. a core
Name: Date: 1. Which of the following best describes an atom? A. protons and electrons grouped together in a random pattern B. protons and electrons grouped together in an alternating pattern C. a core
Nuclear fusion in stars. Collapse of primordial density fluctuations into galaxies and stars, nucleosynthesis in stars
 Nuclear fusion in stars Collapse of primordial density fluctuations into galaxies and stars, nucleosynthesis in stars The origin of structure in the Universe Until the time of formation of protogalaxies,
Nuclear fusion in stars Collapse of primordial density fluctuations into galaxies and stars, nucleosynthesis in stars The origin of structure in the Universe Until the time of formation of protogalaxies,
EXPERIMENT 4 The Periodic Table - Atoms and Elements
 EXPERIMENT 4 The Periodic Table - Atoms and Elements INTRODUCTION Primary substances, called elements, build all the materials around you. There are more than 109 different elements known today. The elements
EXPERIMENT 4 The Periodic Table - Atoms and Elements INTRODUCTION Primary substances, called elements, build all the materials around you. There are more than 109 different elements known today. The elements
Section 1: Arranging the Elements Pages 106-112
 Study Guide Chapter 5 Periodic Table Section 1: Arranging the Elements Pages 106-112 DISCOVERING A PATTERN 1. How did Mendeleev arrange the elements? a. by increasing density b. by increasing melting point
Study Guide Chapter 5 Periodic Table Section 1: Arranging the Elements Pages 106-112 DISCOVERING A PATTERN 1. How did Mendeleev arrange the elements? a. by increasing density b. by increasing melting point
Basic Concepts in Nuclear Physics
 Basic Concepts in Nuclear Physics Paolo Finelli Corso di Teoria delle Forze Nucleari 2011 Literature/Bibliography Some useful texts are available at the Library: Wong, Nuclear Physics Krane, Introductory
Basic Concepts in Nuclear Physics Paolo Finelli Corso di Teoria delle Forze Nucleari 2011 Literature/Bibliography Some useful texts are available at the Library: Wong, Nuclear Physics Krane, Introductory
Interstellar Cosmic-Ray Spectrum from Gamma Rays and Synchrotron
 Interstellar Cosmic-Ray Spectrum from Gamma Rays and Synchrotron Chuck Naval Research Laboratory, Washington, DC charles.dermer@nrl.navy.mil Andy Strong Max-Planck-Institut für extraterrestrische Physik,
Interstellar Cosmic-Ray Spectrum from Gamma Rays and Synchrotron Chuck Naval Research Laboratory, Washington, DC charles.dermer@nrl.navy.mil Andy Strong Max-Planck-Institut für extraterrestrische Physik,
Your years of toil Said Ryle to Hoyle Are wasted years, believe me. The Steady State Is out of date Unless my eyes deceive me.
 Your years of toil Said Ryle to Hoyle Are wasted years, believe me. The Steady State Is out of date Unless my eyes deceive me. My telescope Has dashed your hope; Your tenets are refuted. Let me be terse:
Your years of toil Said Ryle to Hoyle Are wasted years, believe me. The Steady State Is out of date Unless my eyes deceive me. My telescope Has dashed your hope; Your tenets are refuted. Let me be terse:
Atoms Absorb & Emit Light
 Atoms Absorb & Emit Light Spectra The wavelength of the light that an element emits or absorbs is its fingerprint. Atoms emit and absorb light First Test is Thurs, Feb 1 st About 30 multiple choice questions
Atoms Absorb & Emit Light Spectra The wavelength of the light that an element emits or absorbs is its fingerprint. Atoms emit and absorb light First Test is Thurs, Feb 1 st About 30 multiple choice questions
THE PERIODIC TABLE O F T H E E L E M E N T S. The Academic Support Center @ Daytona State College (Science 117, Page 1 of 27)
 THE PERIODIC TABLE O F T H E E L E M E N T S The Academic Support Center @ Daytona State College (Science 117, Page 1 of 27) THE PERIODIC TABLE In 1872, Dmitri Mendeleev created the periodic table arranged
THE PERIODIC TABLE O F T H E E L E M E N T S The Academic Support Center @ Daytona State College (Science 117, Page 1 of 27) THE PERIODIC TABLE In 1872, Dmitri Mendeleev created the periodic table arranged
3 Atomic Structure 15
 3 Atomic Structure 15 3.1 Atoms You need to be familiar with the terms in italics The diameter of the nucleus is approximately 10-15 m and an atom 10-10 m. All matter consists of atoms. An atom can be
3 Atomic Structure 15 3.1 Atoms You need to be familiar with the terms in italics The diameter of the nucleus is approximately 10-15 m and an atom 10-10 m. All matter consists of atoms. An atom can be
The Universe. The Solar system, Stars and Galaxies
 The Universe The Universe is everything. All us, the room, the U.S. the earth, the solar system, all the other stars in the Milky way galaxy, all the other galaxies... everything. How big and how old is
The Universe The Universe is everything. All us, the room, the U.S. the earth, the solar system, all the other stars in the Milky way galaxy, all the other galaxies... everything. How big and how old is
Chapter Five: Atomic Theory and Structure
 Chapter Five: Atomic Theory and Structure Evolution of Atomic Theory The ancient Greek scientist Democritus is often credited with developing the idea of the atom Democritus proposed that matter was, on
Chapter Five: Atomic Theory and Structure Evolution of Atomic Theory The ancient Greek scientist Democritus is often credited with developing the idea of the atom Democritus proposed that matter was, on
Unit 3 Study Guide: Electron Configuration & The Periodic Table
 Name: Teacher s Name: Class: Block: Date: Unit 3 Study Guide: Electron Configuration & The Periodic Table 1. For each of the following elements, state whether the element is radioactive, synthetic or both.
Name: Teacher s Name: Class: Block: Date: Unit 3 Study Guide: Electron Configuration & The Periodic Table 1. For each of the following elements, state whether the element is radioactive, synthetic or both.
UNIT (2) ATOMS AND ELEMENTS
 UNIT (2) ATOMS AND ELEMENTS 2.1 Elements An element is a fundamental substance that cannot be broken down by chemical means into simpler substances. Each element is represented by an abbreviation called
UNIT (2) ATOMS AND ELEMENTS 2.1 Elements An element is a fundamental substance that cannot be broken down by chemical means into simpler substances. Each element is represented by an abbreviation called
Basic Nuclear Concepts
 Section 7: In this section, we present a basic description of atomic nuclei, the stored energy contained within them, their occurrence and stability Basic Nuclear Concepts EARLY DISCOVERIES [see also Section
Section 7: In this section, we present a basic description of atomic nuclei, the stored energy contained within them, their occurrence and stability Basic Nuclear Concepts EARLY DISCOVERIES [see also Section
47374_04_p25-32.qxd 2/9/07 7:50 AM Page 25. 4 Atoms and Elements
 47374_04_p25-32.qxd 2/9/07 7:50 AM Page 25 4 Atoms and Elements 4.1 a. Cu b. Si c. K d. N e. Fe f. Ba g. Pb h. Sr 4.2 a. O b. Li c. S d. Al e. H f. Ne g. Sn h. Au 4.3 a. carbon b. chlorine c. iodine d.
47374_04_p25-32.qxd 2/9/07 7:50 AM Page 25 4 Atoms and Elements 4.1 a. Cu b. Si c. K d. N e. Fe f. Ba g. Pb h. Sr 4.2 a. O b. Li c. S d. Al e. H f. Ne g. Sn h. Au 4.3 a. carbon b. chlorine c. iodine d.
Lecture 3: (Lec3A) Atomic Theory
 Lecture 3: (Lec3A) Atomic Theory Mass of Atoms Sections (Zumdahl 6 th Edition) 3.1-3.4 The Concept of the Mole Outline: The mass of a mole of atoms and the mass of a mole of molecules The composition of
Lecture 3: (Lec3A) Atomic Theory Mass of Atoms Sections (Zumdahl 6 th Edition) 3.1-3.4 The Concept of the Mole Outline: The mass of a mole of atoms and the mass of a mole of molecules The composition of
Name Date Class ELECTRONS IN ATOMS. Standard Curriculum Core content Extension topics
 13 ELECTRONS IN ATOMS Conceptual Curriculum Concrete concepts More abstract concepts or math/problem-solving Standard Curriculum Core content Extension topics Honors Curriculum Core honors content Options
13 ELECTRONS IN ATOMS Conceptual Curriculum Concrete concepts More abstract concepts or math/problem-solving Standard Curriculum Core content Extension topics Honors Curriculum Core honors content Options
MASS DEFECT AND BINDING ENERGY
 MASS DEFECT AND BINDING ENERGY The separate laws of Conservation of Mass and Conservation of Energy are not applied strictly on the nuclear level. It is possible to convert between mass and energy. Instead
MASS DEFECT AND BINDING ENERGY The separate laws of Conservation of Mass and Conservation of Energy are not applied strictly on the nuclear level. It is possible to convert between mass and energy. Instead
PERIODIC TABLE OF GROUPS OF ELEMENTS Elements can be classified using two different schemes.
 1 PERIODIC TABLE OF GROUPS OF ELEMENTS Elements can be classified using two different schemes. Metal Nonmetal Scheme (based on physical properties) Metals - most elements are metals - elements on left
1 PERIODIC TABLE OF GROUPS OF ELEMENTS Elements can be classified using two different schemes. Metal Nonmetal Scheme (based on physical properties) Metals - most elements are metals - elements on left
Name Block Date Ch 17 Atomic Nature of Matter Notes Mrs. Peck. atoms- the smallest particle of an element that can be identified with that element
 Name Block Date Ch 17 Atomic Nature of Matter Notes Mrs. Peck atoms- the smallest particle of an element that can be identified with that element are the building blocks of matter consists of protons and
Name Block Date Ch 17 Atomic Nature of Matter Notes Mrs. Peck atoms- the smallest particle of an element that can be identified with that element are the building blocks of matter consists of protons and
Photons. ConcepTest 27.1. 1) red light 2) yellow light 3) green light 4) blue light 5) all have the same energy. Which has more energy, a photon of:
 ConcepTest 27.1 Photons Which has more energy, a photon of: 1) red light 2) yellow light 3) green light 4) blue light 5) all have the same energy 400 nm 500 nm 600 nm 700 nm ConcepTest 27.1 Photons Which
ConcepTest 27.1 Photons Which has more energy, a photon of: 1) red light 2) yellow light 3) green light 4) blue light 5) all have the same energy 400 nm 500 nm 600 nm 700 nm ConcepTest 27.1 Photons Which
PROTONS AND ELECTRONS
 reflect Imagine that you have a bowl of oranges, bananas, pineapples, berries, pears, and watermelon. How do you identify each piece of fruit? Most likely, you are familiar with the characteristics of
reflect Imagine that you have a bowl of oranges, bananas, pineapples, berries, pears, and watermelon. How do you identify each piece of fruit? Most likely, you are familiar with the characteristics of
WELCOME to Aurorae In the Solar System. J.E. Klemaszewski
 WELCOME to Aurorae In the Solar System Aurorae in the Solar System Sponsoring Projects Galileo Europa Mission Jupiter System Data Analysis Program ACRIMSAT Supporting Projects Ulysses Project Outer Planets
WELCOME to Aurorae In the Solar System Aurorae in the Solar System Sponsoring Projects Galileo Europa Mission Jupiter System Data Analysis Program ACRIMSAT Supporting Projects Ulysses Project Outer Planets
(b) find the force of repulsion between a proton at the surface of a 12. 6 C nucleus and the remaining five protons.
 Chapter 13 Nuclear Structure. Home Work s 13.1 Problem 13.10 (a) find the radius of the 12 6 C nucleus. (b) find the force of repulsion between a proton at the surface of a 12 6 C nucleus and the remaining
Chapter 13 Nuclear Structure. Home Work s 13.1 Problem 13.10 (a) find the radius of the 12 6 C nucleus. (b) find the force of repulsion between a proton at the surface of a 12 6 C nucleus and the remaining
Masses in Atomic Units
 Nuclear Composition - the forces binding protons and neutrons in the nucleus are much stronger (binding energy of MeV) than the forces binding electrons to the atom (binding energy of ev) - the constituents
Nuclear Composition - the forces binding protons and neutrons in the nucleus are much stronger (binding energy of MeV) than the forces binding electrons to the atom (binding energy of ev) - the constituents
2014 Spring CHEM101 Ch1-2 Review Worksheet Modified by Dr. Cheng-Yu Lai,
 Ch1 1) Which of the following underlined items is not an intensive property? A) A chemical reaction requires 3.00 g of oxygen. B) The density of helium at 25 C is 1.64 10-4 g/cm3. C) The melting point
Ch1 1) Which of the following underlined items is not an intensive property? A) A chemical reaction requires 3.00 g of oxygen. B) The density of helium at 25 C is 1.64 10-4 g/cm3. C) The melting point
Main properties of atoms and nucleus
 Main properties of atoms and nucleus. Atom Structure.... Structure of Nuclei... 3. Definition of Isotopes... 4. Energy Characteristics of Nuclei... 5. Laws of Radioactive Nuclei Transformation... 3. Atom
Main properties of atoms and nucleus. Atom Structure.... Structure of Nuclei... 3. Definition of Isotopes... 4. Energy Characteristics of Nuclei... 5. Laws of Radioactive Nuclei Transformation... 3. Atom
Structure and Properties of Atoms
 PS-2.1 Compare the subatomic particles (protons, neutrons, electrons) of an atom with regard to mass, location, and charge, and explain how these particles affect the properties of an atom (including identity,
PS-2.1 Compare the subatomic particles (protons, neutrons, electrons) of an atom with regard to mass, location, and charge, and explain how these particles affect the properties of an atom (including identity,
2, 8, 20, 28, 50, 82, 126.
 Chapter 5 Nuclear Shell Model 5.1 Magic Numbers The binding energies predicted by the Liquid Drop Model underestimate the actual binding energies of magic nuclei for which either the number of neutrons
Chapter 5 Nuclear Shell Model 5.1 Magic Numbers The binding energies predicted by the Liquid Drop Model underestimate the actual binding energies of magic nuclei for which either the number of neutrons
Periodic Table. 1. In the modern Periodic Table, the elements are arranged in order of increasing. A. atomic number B. mass number
 Name: ate: 1. In the modern, the elements are arranged in order of increasing. atomic number. mass number. oxidation number. valence number 5. s the elements in Group I are considered in order of increasing
Name: ate: 1. In the modern, the elements are arranged in order of increasing. atomic number. mass number. oxidation number. valence number 5. s the elements in Group I are considered in order of increasing
THESIS PROJECT PROPOSAL FORM
 INSTITUTO DE ASTROFÍSICA DE CANARIAS GRADUATE STUDIES DIVISION THESIS PROJECT PROPOSAL FORM NAME OF THE PROPONENT: Jorge Sánchez Almeida Casiana Muñoz Tuñón Alfonso López Aguerri AFFILIATION: (Please indicate
INSTITUTO DE ASTROFÍSICA DE CANARIAS GRADUATE STUDIES DIVISION THESIS PROJECT PROPOSAL FORM NAME OF THE PROPONENT: Jorge Sánchez Almeida Casiana Muñoz Tuñón Alfonso López Aguerri AFFILIATION: (Please indicate
Name: Worksheet: Electron Configurations. I Heart Chemistry!
 1. Which electron configuration represents an atom in an excited state? 1s 2 2s 2 2p 6 3p 1 1s 2 2s 2 2p 6 3s 2 3p 2 1s 2 2s 2 2p 6 3s 2 3p 1 1s 2 2s 2 2p 6 3s 2 Worksheet: Electron Configurations Name:
1. Which electron configuration represents an atom in an excited state? 1s 2 2s 2 2p 6 3p 1 1s 2 2s 2 2p 6 3s 2 3p 2 1s 2 2s 2 2p 6 3s 2 3p 1 1s 2 2s 2 2p 6 3s 2 Worksheet: Electron Configurations Name:
Chapter NP-5. Nuclear Physics. Nuclear Reactions TABLE OF CONTENTS INTRODUCTION OBJECTIVES 1.0 NUCLEAR REACTIONS 2.0 NEUTRON INTERACTIONS
 Chapter NP-5 Nuclear Physics Nuclear Reactions TABLE OF CONTENTS INTRODUCTION OBJECTIVES 1.0 2.0 NEUTRON INTERACTIONS 2.1 ELASTIC SCATTERING 2.2 INELASTIC SCATTERING 2.3 RADIATIVE CAPTURE 2.4 PARTICLE
Chapter NP-5 Nuclear Physics Nuclear Reactions TABLE OF CONTENTS INTRODUCTION OBJECTIVES 1.0 2.0 NEUTRON INTERACTIONS 2.1 ELASTIC SCATTERING 2.2 INELASTIC SCATTERING 2.3 RADIATIVE CAPTURE 2.4 PARTICLE
23. The Beginning of Time. Agenda. Agenda. ESA s Venus Express. Conditions in the Early Universe. 23.1 Running the Expansion Backward
 23. The Beginning of Time Somewhere, something incredible is waiting to be known. Agenda Announce: Solar Altitude Lab (#2) due today Read Ch. 24 for Thursday Observation make-up next week Project Presentations
23. The Beginning of Time Somewhere, something incredible is waiting to be known. Agenda Announce: Solar Altitude Lab (#2) due today Read Ch. 24 for Thursday Observation make-up next week Project Presentations
Transcript 22 - Universe
 Transcript 22 - Universe A few introductory words of explanation about this transcript: This transcript includes the words sent to the narrator for inclusion in the latest version of the associated video.
Transcript 22 - Universe A few introductory words of explanation about this transcript: This transcript includes the words sent to the narrator for inclusion in the latest version of the associated video.
Atomic Structure Chapter 5 Assignment & Problem Set
 Atomic Structure Name Warm-Ups (Show your work for credit) Date 1. Date 2. Date 3. Date 4. Date 5. Date 6. Date 7. Date 8. Atomic Structure 2 Study Guide: Things You Must Know Vocabulary (know the definition
Atomic Structure Name Warm-Ups (Show your work for credit) Date 1. Date 2. Date 3. Date 4. Date 5. Date 6. Date 7. Date 8. Atomic Structure 2 Study Guide: Things You Must Know Vocabulary (know the definition
Light sterile neutrinos in the early universe. Cecilia Lunardini Arizona State University
 Light sterile neutrinos in the early universe Cecilia Lunardini Arizona State University ν s and cosmic radiation energy density Motivations and status how much room for ν s in the universe? Contribution
Light sterile neutrinos in the early universe Cecilia Lunardini Arizona State University ν s and cosmic radiation energy density Motivations and status how much room for ν s in the universe? Contribution
Chapter 20 The Big Bang
 Chapter 20 The Big Bang The Universe began life in a very hot, very dense state that we call the big bang. In this chapter we apply the Friedmann equations to the early Universe in an attempt to understand
Chapter 20 The Big Bang The Universe began life in a very hot, very dense state that we call the big bang. In this chapter we apply the Friedmann equations to the early Universe in an attempt to understand
Basics of Nuclear Physics and Fission
 Basics of Nuclear Physics and Fission A basic background in nuclear physics for those who want to start at the beginning. Some of the terms used in this factsheet can be found in IEER s on-line glossary.
Basics of Nuclear Physics and Fission A basic background in nuclear physics for those who want to start at the beginning. Some of the terms used in this factsheet can be found in IEER s on-line glossary.
Name period AP chemistry Unit 2 worksheet Practice problems
 Name period AP chemistry Unit 2 worksheet Practice problems 1. What are the SI units for a. Wavelength of light b. frequency of light c. speed of light Meter hertz (s -1 ) m s -1 (m/s) 2. T/F (correct
Name period AP chemistry Unit 2 worksheet Practice problems 1. What are the SI units for a. Wavelength of light b. frequency of light c. speed of light Meter hertz (s -1 ) m s -1 (m/s) 2. T/F (correct
PHYA5/1. General Certificate of Education Advanced Level Examination June 2011. Unit 5 Nuclear and Thermal Physics Section A
 Centre Number Surname Candidate Number For Examinerʼs Use Other Names Candidate Signature Examinerʼs Initials General Certificate of Education Advanced Level Examination June 2011 Question 1 2 Mark Physics
Centre Number Surname Candidate Number For Examinerʼs Use Other Names Candidate Signature Examinerʼs Initials General Certificate of Education Advanced Level Examination June 2011 Question 1 2 Mark Physics
Evolution of the Universe from 13 to 4 Billion Years Ago
 Evolution of the Universe from 13 to 4 Billion Years Ago Prof. Dr. Harold Geller hgeller@gmu.edu http://physics.gmu.edu/~hgeller/ Department of Physics and Astronomy George Mason University Unity in the
Evolution of the Universe from 13 to 4 Billion Years Ago Prof. Dr. Harold Geller hgeller@gmu.edu http://physics.gmu.edu/~hgeller/ Department of Physics and Astronomy George Mason University Unity in the
2. John Dalton did his research work in which of the following countries? a. France b. Greece c. Russia d. England
 CHAPTER 3 1. Which combination of individual and contribution is not correct? a. Antoine Lavoisier - clarified confusion over cause of burning b. John Dalton - proposed atomic theory c. Marie Curie - discovered
CHAPTER 3 1. Which combination of individual and contribution is not correct? a. Antoine Lavoisier - clarified confusion over cause of burning b. John Dalton - proposed atomic theory c. Marie Curie - discovered
ANSWER KEY : BUILD AN ATOM PART I: ATOM SCREEN Build an Atom simulation ( http://phet.colorado.edu/en/simulation/build an atom )
 ANSWER KEY : PART I: ATOM SCREEN Build an Atom simulation ( http://phet.colorado.edu/en/simulation/build an atom ) 1. Explore the Build an Atom simulation with your group. As you explore, talk about what
ANSWER KEY : PART I: ATOM SCREEN Build an Atom simulation ( http://phet.colorado.edu/en/simulation/build an atom ) 1. Explore the Build an Atom simulation with your group. As you explore, talk about what
Part 1 Composition of Earth Composition of solar system Origin of the elements Part 2 Geochronometry: Age of Earth Formation of Earth and Moon.
 Part 1 Composition of Earth Composition of solar system Origin of the elements Part 2 Geochronometry: Age of Earth Formation of Earth and Moon. Differentiation of core and mantle. Isotope tracing: sequence
Part 1 Composition of Earth Composition of solar system Origin of the elements Part 2 Geochronometry: Age of Earth Formation of Earth and Moon. Differentiation of core and mantle. Isotope tracing: sequence
Modifying Colors and Symbols in ArcMap
 Modifying Colors and Symbols in ArcMap Contents Introduction... 1 Displaying Categorical Data... 3 Creating New Categories... 5 Displaying Numeric Data... 6 Graduated Colors... 6 Graduated Symbols... 9
Modifying Colors and Symbols in ArcMap Contents Introduction... 1 Displaying Categorical Data... 3 Creating New Categories... 5 Displaying Numeric Data... 6 Graduated Colors... 6 Graduated Symbols... 9
GCE Physics A. Mark Scheme for June 2014. Unit G485: Fields, Particles and Frontiers of Physics. Advanced GCE. Oxford Cambridge and RSA Examinations
 GCE Physics A Unit G485: Fields, Particles and Frontiers of Physics Advanced GCE Mark Scheme for June 014 Oxford Cambridge and RSA Examinations OCR (Oxford Cambridge and RSA) is a leading UK awarding body,
GCE Physics A Unit G485: Fields, Particles and Frontiers of Physics Advanced GCE Mark Scheme for June 014 Oxford Cambridge and RSA Examinations OCR (Oxford Cambridge and RSA) is a leading UK awarding body,
NITROGEN-TO-OXYGEN RATIO As a solid tool to ascertain the chemical evolution of star-forming galaxies
 NITROGEN-TO-OXYGEN RATIO As a solid tool to ascertain the chemical evolution of star-forming galaxies Enrique Pérez Montero Instituto de Astrofísica de Andalucía - CSIC (Granada) Proyecto ESTALLIDOS DE
NITROGEN-TO-OXYGEN RATIO As a solid tool to ascertain the chemical evolution of star-forming galaxies Enrique Pérez Montero Instituto de Astrofísica de Andalucía - CSIC (Granada) Proyecto ESTALLIDOS DE
Objectives 404 CHAPTER 9 RADIATION
 Objectives Explain the difference between isotopes of the same element. Describe the force that holds nucleons together. Explain the relationship between mass and energy according to Einstein s theory
Objectives Explain the difference between isotopes of the same element. Describe the force that holds nucleons together. Explain the relationship between mass and energy according to Einstein s theory
Chapter 5 TEST: The Periodic Table name
 Chapter 5 TEST: The Periodic Table name HPS # date: Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The order of elements in the periodic table is based
Chapter 5 TEST: The Periodic Table name HPS # date: Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The order of elements in the periodic table is based
Gravity Testing and Interpreting Cosmological Measurement
 Cosmological Scale Tests of Gravity Edmund Bertschinger MIT Department of Physics and Kavli Institute for Astrophysics and Space Research January 2011 References Caldwell & Kamionkowski 0903.0866 Silvestri
Cosmological Scale Tests of Gravity Edmund Bertschinger MIT Department of Physics and Kavli Institute for Astrophysics and Space Research January 2011 References Caldwell & Kamionkowski 0903.0866 Silvestri
 WIMP dark matter and the isotropic radio signal Roberto A. Lineros R. Instituto de Física Corpuscular - CSIC/U. Valencia @Roberto_Lineros Outline Introduction Cosmic ray propagation Synchrotron emission
WIMP dark matter and the isotropic radio signal Roberto A. Lineros R. Instituto de Física Corpuscular - CSIC/U. Valencia @Roberto_Lineros Outline Introduction Cosmic ray propagation Synchrotron emission
THE BOHR QUANTUM MODEL
 THE BOHR QUANTUM MODEL INTRODUCTION When light from a low-pressure gas is subject to an electric discharge, a discrete line spectrum is emitted. When light from such a low-pressure gas is examined with
THE BOHR QUANTUM MODEL INTRODUCTION When light from a low-pressure gas is subject to an electric discharge, a discrete line spectrum is emitted. When light from such a low-pressure gas is examined with
Test 2 --- Natural Sciences 102, Professors Rieke --- VERSION B March 3, 2010
 Enter your answers on the form provided. Be sure to write your name and student ID number on the first blank at the bottom of the form. Please mark the version (B) in the Key ID space at the top of the
Enter your answers on the form provided. Be sure to write your name and student ID number on the first blank at the bottom of the form. Please mark the version (B) in the Key ID space at the top of the
100 cm 1 m. = 614 cm. 6.14 m. 2.54 cm. 1 m 1 in. 1 m. 2.54 cm 1ft. 1 in = 242 in. 614 cm. 242 in 1 ft. 1 in. 100 cm = 123 m
 Units and Unit Conversions 6. Define the problem: If the nucleus were scaled to a diameter of 4 cm, determine the diameter of the atom. Develop a plan: Find the accepted relationship between the size of
Units and Unit Conversions 6. Define the problem: If the nucleus were scaled to a diameter of 4 cm, determine the diameter of the atom. Develop a plan: Find the accepted relationship between the size of
Unit 1 Practice Test. Matching
 Unit 1 Practice Test Matching Match each item with the correct statement below. a. proton d. electron b. nucleus e. neutron c. atom 1. the smallest particle of an element that retains the properties of
Unit 1 Practice Test Matching Match each item with the correct statement below. a. proton d. electron b. nucleus e. neutron c. atom 1. the smallest particle of an element that retains the properties of
Modified Gravity and the CMB
 Modified Gravity and the CMB Philippe Brax, IphT Saclay, France arxiv:1109.5862 PhB, A.C. Davis Work in progress PhB, ACD, B. Li Minneapolis October 2011 PLANCK will give us very precise information on
Modified Gravity and the CMB Philippe Brax, IphT Saclay, France arxiv:1109.5862 PhB, A.C. Davis Work in progress PhB, ACD, B. Li Minneapolis October 2011 PLANCK will give us very precise information on
2. The Three Pillars of the Big Bang Theory
 2. The Three Pillars of the Big Bang Theory The evolution of the Universe can be compared to a display of fireworks that has just ended: some few wisps, ashes and smoke. Standing on a cooled cinder, we
2. The Three Pillars of the Big Bang Theory The evolution of the Universe can be compared to a display of fireworks that has just ended: some few wisps, ashes and smoke. Standing on a cooled cinder, we
Supporting Information. Fast and Efficient Fragment-Based Lead Generation. by Fully Automated Processing and Analysis of
 Supporting Information Fast and Efficient Fragment-Based Lead Generation by Fully Automated Processing and Analysis of Ligand-Observed NMR Binding Data Chen Peng, Alexandra Frommlet, Manuel Perez, Carlos
Supporting Information Fast and Efficient Fragment-Based Lead Generation by Fully Automated Processing and Analysis of Ligand-Observed NMR Binding Data Chen Peng, Alexandra Frommlet, Manuel Perez, Carlos
Test Bank - Chapter 4 Multiple Choice
 Test Bank - Chapter 4 The questions in the test bank cover the concepts from the lessons in Chapter 4. Select questions from any of the categories that match the content you covered with students. The
Test Bank - Chapter 4 The questions in the test bank cover the concepts from the lessons in Chapter 4. Select questions from any of the categories that match the content you covered with students. The
FOR TEACHERS ONLY The University of the State of New York REGENTS HIGH SCHOOL EXAMINATION
 PS C FOR TEACERS ONLY The University of the State of New York REGENTS IG SCOOL EXAMINATION PYSICAL SETTING/CEMISTRY Wednesday, June, 0 :5 to :5 p.m., only SCORING KEY AND RATING GUIDE Directions to the
PS C FOR TEACERS ONLY The University of the State of New York REGENTS IG SCOOL EXAMINATION PYSICAL SETTING/CEMISTRY Wednesday, June, 0 :5 to :5 p.m., only SCORING KEY AND RATING GUIDE Directions to the
Build Your Own Universe
 Build Your Own Universe You will need: At least 10,000,000,000,000,00 0,000,000,000,000,000,000,00 0,000,000,000,000,000,000,00 0,000,000,000,000,000,000,00 0,000 x Down quarks At least 10,000,000,000,000,000,
Build Your Own Universe You will need: At least 10,000,000,000,000,00 0,000,000,000,000,000,000,00 0,000,000,000,000,000,000,00 0,000,000,000,000,000,000,00 0,000 x Down quarks At least 10,000,000,000,000,000,
Atoms and Molecules. Preparation. Objectives. Standards. Materials. Grade Level: 5-8 Group Size: 20-30 Time: 60 90 Minutes Presenters: 2-4
 Atoms and Molecules Preparation Grade Level: 5-8 Group Size: 20-30 Time: 60 90 Minutes Presenters: 2-4 Objectives This lesson will enable students to: Describe how atoms are the building blocks of matter
Atoms and Molecules Preparation Grade Level: 5-8 Group Size: 20-30 Time: 60 90 Minutes Presenters: 2-4 Objectives This lesson will enable students to: Describe how atoms are the building blocks of matter
Faber-Jackson relation: Fundamental Plane: Faber-Jackson Relation
 Faber-Jackson relation: Faber-Jackson Relation In 1976, Faber & Jackson found that: Roughly, L! " 4 More luminous galaxies have deeper potentials Can show that this follows from the Virial Theorem Why
Faber-Jackson relation: Faber-Jackson Relation In 1976, Faber & Jackson found that: Roughly, L! " 4 More luminous galaxies have deeper potentials Can show that this follows from the Virial Theorem Why
Galaxy Survey data analysis using SDSS-III as an example
 Galaxy Survey data analysis using SDSS-III as an example Will Percival (University of Portsmouth) showing work by the BOSS galaxy clustering working group" Cosmology from Spectroscopic Galaxy Surveys"
Galaxy Survey data analysis using SDSS-III as an example Will Percival (University of Portsmouth) showing work by the BOSS galaxy clustering working group" Cosmology from Spectroscopic Galaxy Surveys"
The Mole. 6.022 x 10 23
 The Mole 6.022 x 10 23 Background: atomic masses Look at the atomic masses on the periodic table. What do these represent? E.g. the atomic mass of Carbon is 12.01 (atomic # is 6) We know there are 6 protons
The Mole 6.022 x 10 23 Background: atomic masses Look at the atomic masses on the periodic table. What do these represent? E.g. the atomic mass of Carbon is 12.01 (atomic # is 6) We know there are 6 protons
Chemical Building Blocks: Chapter 3: Elements and Periodic Table
 Name: Class: Date: Chemical Building Blocks: Chapter 3: Elements and Periodic Table Study Guide Multiple Choice Identify the letter of the choice that best completes the statement or answers the question.
Name: Class: Date: Chemical Building Blocks: Chapter 3: Elements and Periodic Table Study Guide Multiple Choice Identify the letter of the choice that best completes the statement or answers the question.
Atoms, Elements, and the Periodic Table (Chapter 2)
 Atoms, Elements, and the Periodic Table (Chapter 2) Atomic Structure 1. Historical View - Dalton's Atomic Theory Based on empirical observations, formulated as Laws of: Conservation of Mass Definite Proportions
Atoms, Elements, and the Periodic Table (Chapter 2) Atomic Structure 1. Historical View - Dalton's Atomic Theory Based on empirical observations, formulated as Laws of: Conservation of Mass Definite Proportions
18.2 Comparing Atoms. Atomic number. Chapter 18
 As you know, some substances are made up of only one kind of atom and these substances are called elements. You already know something about a number of elements you ve heard of hydrogen, helium, silver,
As you know, some substances are made up of only one kind of atom and these substances are called elements. You already know something about a number of elements you ve heard of hydrogen, helium, silver,
Homework #10 (749508)
 Homework #10 (749508) Current Score: 0 out of 100 Description Homework on quantum physics and radioactivity Instructions Answer all the questions as best you can. 1. Hewitt10 32.E.001. [481697] 0/5 points
Homework #10 (749508) Current Score: 0 out of 100 Description Homework on quantum physics and radioactivity Instructions Answer all the questions as best you can. 1. Hewitt10 32.E.001. [481697] 0/5 points
Properties of Atoms and the Periodic Table
 Properties of Atoms and the Periodic Table Section 3 The Periodic Table Skim Section 3 and write three questions based on your brief preview. 1. Accept all reasonable answers. How are the elements organized
Properties of Atoms and the Periodic Table Section 3 The Periodic Table Skim Section 3 and write three questions based on your brief preview. 1. Accept all reasonable answers. How are the elements organized
PHYA5/1. General Certificate of Education Advanced Level Examination June 2012. Unit 5 Nuclear and Thermal Physics Section A
 Centre Number Surname Candidate Number For Examinerʼs Use Other Names Candidate Signature Examinerʼs Initials General Certificate of Education Advanced Level Examination June 2012 Question 1 2 Mark Physics
Centre Number Surname Candidate Number For Examinerʼs Use Other Names Candidate Signature Examinerʼs Initials General Certificate of Education Advanced Level Examination June 2012 Question 1 2 Mark Physics
Elements in the periodic table are indicated by SYMBOLS. To the left of the symbol we find the atomic mass (A) at the upper corner, and the atomic num
 . ATOMIC STRUCTURE FUNDAMENTALS LEARNING OBJECTIVES To review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. This material is essential
. ATOMIC STRUCTURE FUNDAMENTALS LEARNING OBJECTIVES To review the basics concepts of atomic structure that have direct relevance to the fundamental concepts of organic chemistry. This material is essential
H 2 /HD Molecular Clouds in the Early Universe. An Independent Means of Estimating the Baryon Density of the Universe
 ISSN 163-7729, Astronomy Reports, 215, Vol. 59, No. 2, pp. 1 117. c Pleiades Publishing, Ltd., 215. Original Russian Text c A.V. Ivanchik, S.A. Balashev, D.A. Varshalovich, V.V. Klimenko, 215, published
ISSN 163-7729, Astronomy Reports, 215, Vol. 59, No. 2, pp. 1 117. c Pleiades Publishing, Ltd., 215. Original Russian Text c A.V. Ivanchik, S.A. Balashev, D.A. Varshalovich, V.V. Klimenko, 215, published
******* KEY ******* Atomic Structure & Periodic Table Test Study Guide
 Atomic Structure & Periodic Table Test Study Guide VOCABULARY: Write a brief definition of each term in the space provided. 1. Atoms: smallest unit of an element that has all of the properties of that
Atomic Structure & Periodic Table Test Study Guide VOCABULARY: Write a brief definition of each term in the space provided. 1. Atoms: smallest unit of an element that has all of the properties of that
PANTONE Solid to Process
 PANTONE Solid to Process PANTONE C:0 M:0 Y:100 K:0 Proc. Yellow PC PANTONE C:0 M:0 Y:51 K:0 100 PC PANTONE C:0 M:2 Y:69 K:0 106 PC PANTONE C:0 M:100 Y:0 K:0 Proc. Magen. PC PANTONE C:0 M:0 Y:79 K:0 101
PANTONE Solid to Process PANTONE C:0 M:0 Y:100 K:0 Proc. Yellow PC PANTONE C:0 M:0 Y:51 K:0 100 PC PANTONE C:0 M:2 Y:69 K:0 106 PC PANTONE C:0 M:100 Y:0 K:0 Proc. Magen. PC PANTONE C:0 M:0 Y:79 K:0 101
