Respiratory Clinical Trial Services. the difference is clear. Your respiratory clinical trials partner
|
|
|
- Silas Moore
- 8 years ago
- Views:
Transcription
1 Respiratory Clinical Trial Services the difference is clear Your respiratory clinical trials partner
2 Our heritage We at Vitalograph are proud of our 50 year heritage as a world leading respiratory specialist. Our pioneering spirit has led us to develop a number of innovative respiratory devices, including the first ever spirometer designed for use in primary care. This same commitment to innovation is reflected in our wide range of clinical trials services. Vitalograph has been owned by the Garbe family since the day the company was founded. This continuity of management has resulted in a clear and consistent corporate vision; it has also created the reassuring stability and speed of response that comes from a financially strong, privately owned company with no influence from large corporate shareholders or venture capitalists. Our people Our expertise Our people are committed to delivering the highest possible standards of customer care and are constantly seeking new ways to improve our service. In addition to friendliness, professionalism and flexibility our highly trained clinical trials team provides a breadth of experience in the delivery of multi-centred global trials, on time and within budget. They also have an in-depth understanding of ICH-GCP and the ATS/ERS requirements for pulmonary function testing in clinical trials. Vitalograph is a leading global provider of respiratory products and services with offices and a network of expert distributors around the world. Vitalograph works in close collaboration with a wide range of international respiratory experts and is involved with CEN and ISO on the development of performance standards for respiratory measuring devices. In the last 5 years Vitalograph has provided 1 million clean test sessions from studies involving 1,110 sites and 40 countries. Our services and equipment for clinical trials We are committed to providing the highest level of service and equipment to meet the endpoints and measurement outcomes of your respiratory study, including: A Centralized PFTs and data capture services: Equipment rental or purchase for clinic and home based measurements Spirotrac spirometry and In2itive e-diary Integrated challenge testing Integrated ECG services with over-reading by expert cardiologists Integrated FeNO data capture A VitaloJAK Cough Monitor Our rigorous systems and processes A QA Review by world-leading PFT experts to maximise data quality and provide detailed feedback on all tests A Vitalograph PFT Web Capture System A Complete data reporting including our secure web-based, on-demand, VIEWER Reports Portal A Project and Data Management, including Protocol Review, Software Customization, 24/7 Support and Training Services A Stand alone equipment for non-centralized studies We have developed a wide range of sophisticated systems and processes to protect the security and success of your trial including: A Secure data transfer of trial data from clinic or subject s home for home based spirometry recording and Patient Reported Outcomes A Vitalograph recently passed its first routine inspection by The Medicines and Healthcare products Regulatory Agency (MHRA). The Agency reported zero critical findings and zero major findings. Vitalograph was one of only 5% of CROs to pass with this degree of success. In addition to this audit success, the latest FDA routine inspection of the Vitalograph research and manufacturing facility in Ireland was also a great success, again with zero deficiencies {483s} raised Project management A Mirrored data management system at a second Vitalograph location everything is automatically backed up and continues to operate seamlessly in the unlikely event that the primary system fails A Automatic data backup memory card in all Site Systems and in the In2itive e-diary making it simple to change in the event of a fault and to re-start with all data intact A Encrypted Internet data transmission system highly secure, fast and cost effective via a local broadband connection A Web-based, on-demand access to data and reports a powerful tool for monitoring the progress of your study using VIEWER Our project management expertise ensures that studies run to specification and schedule and that all steps involved in successful completion are conducted with ongoing analysis and management of risks to GCP. Each study is assigned a dedicated project manager with several years experience in respiratory clinical trials. Project managers ensure that the following activities are tailored to suit the needs of each study: A Study Management: to include project management, project analysis and reports to the sponsor A Spirometry Consultation: to include protocol review and electronic quality review of pulmonary function tests and query resolution A Data Management: based at Vitalograph Ltd, Buckingham, UK, this includes secure daily back-ups and a mirrored system at Vitalograph (Ireland) Ltd, Ennis; data edit checking and corrections; on-line (Web) monitoring and reporting
3 Spirotrac specialized centralized data services The Vitalograph Spirotrac Centralized Spirometry System has been designed so that on receipt into our central database tests may be rapidly and regularly QA reviewed by expert over-readers. The database is then made available for edit checking, data transfers to the sponsor, and subsequent archiving. Different methods of connection can be made to the Internet to transmit encrypted data from investigator sites to the Vitalograph Centralized Data Management System, with all data collected at sites transmitted in this way. The intuitive Spirotrac software provides an easy to use, step-bystep work flow to collect data and is tailored for each study with protocol feedback. We also centralize full pulmonary function test services from diagnostic equipment for gas diffusion (DLCO) and lung volumes (TLC, FRC and RV), FeNO (exhaled nitric oxide) and other measurements. Vitalograph reporting services from the data management system Regular reports are provided from the central database via the Vitalograph Project Team, including: A Pulmonary Function QA review A Subject recruitment progress A Subject alerts A Site technician performance A Subject visit status A Subject compliance Our reporting services are delivered either by or via our VIEWER web portal. Reports of pulmonary function and subject diary data are provided to the sponsor so that monitoring of subject enrolment, progress of the study, subject compliance and alerts can be performed by CRAs and other registered personnel at any time, by country, by site, by subject. Home based spirometry recording and epro Vitalograph s pioneering In2itive e-diary combines an integrated diagnostic quality spirometer, featuring a removable flow head, with an easy to use touch screen. This versatile e-pro system is the first of its kind and can be customized for use in a clinical trial in a wide range of therapeutic areas and in any language. The device itself has a high resolution colour touch-screen, text based interface that accommodates validated questionnaires and is designed to comply with FDA s PRO Guidance. It is manufactured as a Class II medical device to a robust design that allows wireless data transmission using mobile phone technology to send data securely from the subject s home for instant population of the study database. This system enables the study team to monitor and report upon the data in real-time using our on-demand reporting website, VIEWER. Exhaled Nitric Oxide (FeNO) In addition to our Centralized PFT Testing and Data Capture Services, we were the first company to integrate Aerocrine s NiOX MINO into our centralized spirometry system. This integrated solution transmits FeNO data together with spirometry, e-diary and any other measurements collected via Spirotrac as a single dataset, to Vitalograph s Centralized Data Management System and from there to the sponsor. As well as capturing the data electronically, thus avoiding transcription errors, sponsors also like this integrated solution as they only require one vendor contract and one set of shipping costs and associated logistics. Spirotrac ECG VitaloJAK cough monitor This unique product from Vitalograph embodies pioneering technology using dedicated HiFi recording, signal processing and expert algorithms to provide a 48 hour objective measure of cough in clinical trials. It provides an objective measure of cough in subjects with COPD, asthma and chronic cough, making it unnecessary to rely on time-consuming video, tape recordings or inaccurate subject diaries. Worn like a Holter monitor, with a single sensor affixed to the subject s chest wall, the VitaloJAK makes 48 hour cough monitoring simple. An optional second channel via a conventional microphone allows quality assurance with human intervention. With the Spirotrac ECG software from Vitalograph, ECG measurements are captured and displayed in real-time, saved, recalled and printed. The software provides ECG analysis and interpretation based on the scientifically recognized Glasgow algorithm. With the Bluetooth ECG BT12 device the 12-channel ECG data is wirelessly transmitted to the Spirotrac ECG Software. The built-in display provides information on electrode contacts, heart rate, battery status and telemetry reception. All results captured by the Spirotrac ECG software are transmitted from clinical trial sites to the central server for expert review/over-reading.
4 Vitalograph USB, Serial and Bluetooth respiratory monitors The Vitalograph range of USB, Serial and Bluetooth respiratory monitors is designed to enable transmission of respiratory data to your e-diary, telephone, PDA, computer or telemedicine hub, making the home monitoring of those with chronic respiratory diseases, including asthma, cystic fibrosis, COPD and transplant patients, easier than ever. These respiratory monitoring and screening devices are ideal for use in clinical trials where low cost yet accurate and reliable home respiratory monitoring is required. Spirotrac Pulse Oximetry Integrate pulse oximetry, 6 minute walk test and spirometry data. PDA Mobile Global 24/7 support Global 24/7 support is provided for sites and study monitors. Support can be provided in any language through the use of an expert interpretation service. Vitalograph maintains trouble-shooting procedures with a brief compiled for customer support personnel trained on the specific systems and procedures used by a given study. We have recently expanded Vitalograph s help desk and support network to allow provision of 24/7 support directly to subjects as well as study sites for all time-zones in over 140 languages. This new system utilises a global network of Support Technicians trained in both Vitalograph s devices and our sponsors clinical trials. Vitalograph also offers a range of on-site equipment servicing options and calibration checks for longer studies. Device and software customization Devices and software can be customized to meet the needs of a particular trial. The stages of customization include: A Specification development and approval by the sponsor A Software/device development A Software/device testing/qa/validation A Documentation A Study documentation By customizing the software, site personnel follow a workflow that takes them through each subject visit test type in accordance with the study protocol. This ensures that the protocol is correctly followed and provides a simple step by step approach to minimize site errors. Spirotrac Pulse Oximetry The Spirotrac Pulse Oximetry Software Module allows you to plug an SpO2 sensor directly into any USB port on your PC to give accurate and high quality oximetry alongside spirometry. This optional module calculates, trends and reports all primary parameters including minimum, maximum and median SpO2/pulse rate. Capable of recording quick assessments or longer term recordings, with up to four transmitted values selectable by the user. 6 Minute Walk Test A Spirometry and pulse oximetry data is automatically or manually integrated by the 6 minute walk test protocol A 6 minute walk test Wizard runs in 10 logical stages Training A 6 minute walk test is a measure of disability e.g. in COPD studies A The BODE* Index predicts COPD mortality based on FEV₁, 6 MWT, BMI, MMRC and Borg Scales (*Body Mass Index, Obstructive Index, Dyspnoea Score, Exercise Tolerance) The Vitalograph team provides training at investigator and monitor meetings with hands-on sessions and certification. Training can also be provided onsite with top-up training face-to-face or web-based. A range of training videos are also available via the VIEWER Web portal. Vitalograph offers full training of your project team to cover all relevant guidelines/regulations, protocol feedback and data quality prior to the study starting. Web services PFT Web Capture System This innovative web portal and electronic data capture system provides web services for effective monitoring of the study by study managers and CRAs. PFT Web Capture is designed to provide fast, secure data capture with centralized QA review without the need to invest in standardized high cost capital equipment or lengthy training. The system can be tailored to suit your exact needs and its versatility can meet the requirements of any primary or secondary endpoint. Vitalograph provides custom step-by-step software for each site to quickly complete a web page either on or off line. This data can include reports, graphs and images produced by equipment already in use at the site, whether electronic or on paper. The Vitalograph team in Buckingham, UK, checks that data entry from each site is correct before the new data is accepted into the central database on the web server. In this way clean data is captured throughout the study with fast data-lock after LPLV. Using the site s existing equipment gives a big advantage for the study, including: low capital cost; no shipping logistics; faster FPFV; minimal training; higher quality data with automated transmission and error handling. This new system is particularly valuable in trials where: A You wish to monitor the quality of the site data without the need to provide diagnostic equipment or additional training to each site A There is no requirement to standardize equipment at sites in those studies where safety data or perhaps home-based measurements or QOL are the primary endpoints
5 Regulatory compliance Vitalograph systems and methodology conform to: A ICH GCP A FDA 21CFR Part 11 A FDA PRO Guidance A HIPAA and 95/46/EC Vitalograph s design and manufacture conform to: A ISO13485: 2003 QMS for Medical Devices A 21 CFR 820 FDA Quality System Regulation A 93/42/EEC - L169 European Medical Devices Directive, as amended Vitalograph is a world leading provider of outstanding quality cardio-respiratory diagnostic devices, clinical trial services and medical equipment servicing. With a pioneering heritage of excellence spanning half a century Vitalograph continues to make valuable contributions to effective medical care and enhanced quality of life. Vitalograph clinical project management, customer support and device services conform to: A ICH GCP A ISO13485:2003 A ISO9001:2008 A FDA 21 CFR Part 11 A FDA PRO Guidance Vitalograph works to detailed SOPs and study specific procedures in every clinical trial. Vitalograph Ltd. Maids Moreton Buckingham MK18 1SW England Tel: Fax: pharma@vitalograph.co.uk Vitalograph GmbH Rellinger Straße 64a Hamburg Germany Tel: (040) Fax: (040) info@vitalograph.de Vitalograph Inc West 99th Street Lenexa Kansas USA Tel: (913) Fax: (913) vitcs@vitalograph.com Vitalograph (Ireland) Ltd. Gort Road Business Park Ennis Co Clare Ireland Tel: (065) Fax: (065) sales@vitalograph.ie Vitalograph, Spirotrac, VIEWER, VitaloJAK and In2itive are trademarks or registered trademarks of Vitalograph Ltd. Bluetooth is a trademark of Bluetooth SIG. PRINT REF: 14771_1
Vitalograph Products and Services Guide
 Vitalograph Products and Services Guide Vitalograph is a world leading provider of outstanding quality cardio-respiratory devices, clinical trial services and medical equipment servicing. With a pioneering
Vitalograph Products and Services Guide Vitalograph is a world leading provider of outstanding quality cardio-respiratory devices, clinical trial services and medical equipment servicing. With a pioneering
1993-2013 Twenty years of innovative solutions in Spirometry, Oximetry, Telemedicine
 1993-2013 Twenty years of innovative solutions in Spirometry, Oximetry, Telemedicine Established in 1993 MIR has become a landmark for innovation in portable diagnostic devices. High quality and ease of
1993-2013 Twenty years of innovative solutions in Spirometry, Oximetry, Telemedicine Established in 1993 MIR has become a landmark for innovation in portable diagnostic devices. High quality and ease of
Telehealth. Putting the patient at the heart of the journey
 Telehealth Putting the patient at the heart of the journey Why telehealth? 1 Telehealth is the remote monitoring of a patient s vital signs and symptoms in their own home proven to enhance the quality
Telehealth Putting the patient at the heart of the journey Why telehealth? 1 Telehealth is the remote monitoring of a patient s vital signs and symptoms in their own home proven to enhance the quality
Spirodoc. One Touch Laboratory for respiratory analysis two functional modes: doctor and patient. Spirometer with Touch Screen display
 MEDICAL INTERNATIONAL RESEARCH Spirodoc One Touch Laboratory for respiratory analysis two functional modes: doctor and patient Spirometer with Touch Screen display Pulse Oximeter intelligent with on screen
MEDICAL INTERNATIONAL RESEARCH Spirodoc One Touch Laboratory for respiratory analysis two functional modes: doctor and patient Spirometer with Touch Screen display Pulse Oximeter intelligent with on screen
Spirodoc. 3D Laboratory for respiratory analysis two functional modes: doctor and patient. Four devices in one. Spirometer with Touch Screen display
 Spirodoc 3D Laboratory for respiratory analysis two functional modes: doctor and patient Four devices in one Spirometer with Touch Screen display Pulse Oximeter intelligent with on screen results 3D Accelerometer
Spirodoc 3D Laboratory for respiratory analysis two functional modes: doctor and patient Four devices in one Spirometer with Touch Screen display Pulse Oximeter intelligent with on screen results 3D Accelerometer
Vmax Vyntus SPIRO. Advance your lung function testing experience
 Vmax Vyntus SPIRO Advance your lung function testing experience The Vmax Vyntus SPIRO with SentrySuite software is designed to advance the lung function testing experience for both the patient and the
Vmax Vyntus SPIRO Advance your lung function testing experience The Vmax Vyntus SPIRO with SentrySuite software is designed to advance the lung function testing experience for both the patient and the
Smart. Scalable. Simple.
 Thermo Scientific Wireless Monitoring Solution Smart. Scalable. Simple. providing the protection you need with the simplicity you deserve Thermo Scientific Wireless Monitoring Solution Optimized Sample
Thermo Scientific Wireless Monitoring Solution Smart. Scalable. Simple. providing the protection you need with the simplicity you deserve Thermo Scientific Wireless Monitoring Solution Optimized Sample
GCP INSPECTORS WORKING GROUP <DRAFT> REFLECTION PAPER ON EXPECTATIONS FOR ELECTRONIC SOURCE DOCUMENTS USED IN CLINICAL TRIALS
 European Medicines Agency London, 17 October 2007 Doc. Ref. EMEA/505620/2007 GCP INSPECTORS WORKING GROUP REFLECTION PAPER ON EXPECTATIONS FOR ELECTRONIC SOURCE DOCUMENTS USED IN CLINICAL TRIALS
European Medicines Agency London, 17 October 2007 Doc. Ref. EMEA/505620/2007 GCP INSPECTORS WORKING GROUP REFLECTION PAPER ON EXPECTATIONS FOR ELECTRONIC SOURCE DOCUMENTS USED IN CLINICAL TRIALS
A Telehomecare monitoring project in Cystic Fibrosis
 ICEHTM 2011 1st International Conference on E-HEALTH and TELEMEDICINE 10-12 October 2011 Nicosia - North Cyprus A Telehomecare monitoring project in Cystic Fibrosis Murgia F, Alghisi F, Majo F, Montemitro
ICEHTM 2011 1st International Conference on E-HEALTH and TELEMEDICINE 10-12 October 2011 Nicosia - North Cyprus A Telehomecare monitoring project in Cystic Fibrosis Murgia F, Alghisi F, Majo F, Montemitro
How to Run Clinical Trials in Private Practice
 How to Run Clinical Trials in Private Practice Thomas M. Siler, MD Midwest Chest Consultants, PC Saint Charles, MO DISCLOSURE Dr. Siler has received research grants from GlaxoSmithKline, Forest, Boehringer
How to Run Clinical Trials in Private Practice Thomas M. Siler, MD Midwest Chest Consultants, PC Saint Charles, MO DISCLOSURE Dr. Siler has received research grants from GlaxoSmithKline, Forest, Boehringer
Infoset builds software and services to advantage business operations and improve patient s life
 Infoset builds software and services to advantage business operations and improve patient s life Clinical Data Management ecrf & EDC Patient Support Programs Medication Adherence Mobile e-health Big Data
Infoset builds software and services to advantage business operations and improve patient s life Clinical Data Management ecrf & EDC Patient Support Programs Medication Adherence Mobile e-health Big Data
Electronic patient diaries in clinical research
 Topics Electronic diaries in Clinical Trials Electronic diaries versus Paper Electronic patient diaries in clinical research Case Study: Novel detection of exacerbations of COPD with patient reported outcome
Topics Electronic diaries in Clinical Trials Electronic diaries versus Paper Electronic patient diaries in clinical research Case Study: Novel detection of exacerbations of COPD with patient reported outcome
INSERT COMPANY LOGO HERE BEST PRACTICES RESEARCH
 INSERT COMPANY LOGO HERE BEST PRACTICES RESEARCH Background and Company Performance Industry Challenges esource: Electronic Clinical Trial Solution Clinical trial sponsors and clinical research organizations
INSERT COMPANY LOGO HERE BEST PRACTICES RESEARCH Background and Company Performance Industry Challenges esource: Electronic Clinical Trial Solution Clinical trial sponsors and clinical research organizations
Agenda. ARITHMOS Presents: Introduction to epro. EDC and epro integration General Overview. epro and ecrf Integration: a case study
 Agenda ARITHMOS Presents: epro and ecrf Integration: a case study Introduction to epro EDC and epro integration General Overview From epro to ipro: Technology trend in Pharma epro EDC integration: A case
Agenda ARITHMOS Presents: epro and ecrf Integration: a case study Introduction to epro EDC and epro integration General Overview From epro to ipro: Technology trend in Pharma epro EDC integration: A case
An information platform that delivers clinical studies better, faster, safer and more cost effectively
 An information platform that delivers clinical studies better, faster, safer and more cost effectively Powering Process & Performance Proactively manage study start-up and execution Risk profile new sites
An information platform that delivers clinical studies better, faster, safer and more cost effectively Powering Process & Performance Proactively manage study start-up and execution Risk profile new sites
Innovation in Spirometry Oximetry Telemedicine
 English Innovation in Spirometry Oximetry Telemedicine Contact MIR at 844-4MIRUSA for MIR USA Sales Support & Service, Monday to Friday 8:00am to 5:00pm (central time) MIR Group Organization MIR Medical
English Innovation in Spirometry Oximetry Telemedicine Contact MIR at 844-4MIRUSA for MIR USA Sales Support & Service, Monday to Friday 8:00am to 5:00pm (central time) MIR Group Organization MIR Medical
4Sight Calibration Management Software
 GE Measurement & Control Solutions 4Sight Calibration Management Software 4Sight calibration and maintenance management software provides visibility to the assets, data, and resources that affect maintenance,
GE Measurement & Control Solutions 4Sight Calibration Management Software 4Sight calibration and maintenance management software provides visibility to the assets, data, and resources that affect maintenance,
Reflection paper on expectations for electronic source data and data transcribed to electronic data collection tools in clinical trials
 09 June 2010 EMA/INS/GCP/454280/2010 GCP Inspectors Working Group (GCP IWG) Reflection paper on expectations for electronic source data and data transcribed to electronic data collection tools in clinical
09 June 2010 EMA/INS/GCP/454280/2010 GCP Inspectors Working Group (GCP IWG) Reflection paper on expectations for electronic source data and data transcribed to electronic data collection tools in clinical
Clinical Training Management
 Clinical Training Management Learning and Compliance for Clinical Research Helping to Fuel the Growth of CROs and Service Providers According to clinical researcher CenterWatch, the FDA and global regulatory
Clinical Training Management Learning and Compliance for Clinical Research Helping to Fuel the Growth of CROs and Service Providers According to clinical researcher CenterWatch, the FDA and global regulatory
IP Video Surveillance
 IP Video Surveillance Solutions and Services Wireless IP PDA Administration Wireless Access Point Viewer Remote Control IP Video Server IP Network Alarm Management Video Recording Analog Video Server Access
IP Video Surveillance Solutions and Services Wireless IP PDA Administration Wireless Access Point Viewer Remote Control IP Video Server IP Network Alarm Management Video Recording Analog Video Server Access
How can I protect valuable assets from fire and ensure business continuity?
 How can I protect valuable assets from fire and ensure business continuity? XC10 compact panels combine fire detection and extinguishing control for comprehensive safety. Answers for infrastructure. Compact
How can I protect valuable assets from fire and ensure business continuity? XC10 compact panels combine fire detection and extinguishing control for comprehensive safety. Answers for infrastructure. Compact
Topics. From paper to EDC. From paper to EDC. From paper to EDC. From paper to EDC
 Topics Infermed MACRO - Electronic Data Capture according to GCP Dipl. Inf. A. Fischer I. II. Requirements by GCP III. IV. Heidelberg / January 28, 2008 2 History Idea born in 1970s 1980 - early 1990s:
Topics Infermed MACRO - Electronic Data Capture according to GCP Dipl. Inf. A. Fischer I. II. Requirements by GCP III. IV. Heidelberg / January 28, 2008 2 History Idea born in 1970s 1980 - early 1990s:
Needs, Providing Solutions
 Identifying Needs, Providing Solutions 1 I n d u s t r y The growth of medical research and the countless innovations coming from the pharmaceutical, biotechnology and medical device industry, has improved
Identifying Needs, Providing Solutions 1 I n d u s t r y The growth of medical research and the countless innovations coming from the pharmaceutical, biotechnology and medical device industry, has improved
Managing & Validating Research Data
 Research Management Standard Operating Procedure ISOP-H02 VERSION / REVISION: 2.0 EFFECTIVE DATE: 01 03 12 REVIEW DATE: 01 03 14 AUTHOR(S): CONTROLLER(S): APPROVED BY: Information Officer; NBT Clinical
Research Management Standard Operating Procedure ISOP-H02 VERSION / REVISION: 2.0 EFFECTIVE DATE: 01 03 12 REVIEW DATE: 01 03 14 AUTHOR(S): CONTROLLER(S): APPROVED BY: Information Officer; NBT Clinical
Smart wayside management software
 Smart wayside management software FleetWise is innovative wayside data management software specifically designed to help transit authorities achieve superior fleet performance by collecting, distributing
Smart wayside management software FleetWise is innovative wayside data management software specifically designed to help transit authorities achieve superior fleet performance by collecting, distributing
Operations Manager Comprehensive, secure remote monitoring and management of your entire digital signage network infrastructure
 Network monitoring, management and maintenance 3M TM Network Operations Manager Comprehensive, secure remote monitoring and management of your entire digital signage network infrastructure Provides real-time
Network monitoring, management and maintenance 3M TM Network Operations Manager Comprehensive, secure remote monitoring and management of your entire digital signage network infrastructure Provides real-time
Interactive Response Technologies
 Interactive Response Technologies Increasing accuracy and efficiency in clinical trials To increase the accuracy and efficiency of conducting your global clinical trials, ICON s Interactive Response Technologies
Interactive Response Technologies Increasing accuracy and efficiency in clinical trials To increase the accuracy and efficiency of conducting your global clinical trials, ICON s Interactive Response Technologies
KCR Data Management: Designed for Full Data Transparency
 KCR Data Management: Designed for Full Data Transparency 6% Observational 4% Phase IV 2% Device 11% Phase I 28% Phase II 49% Phase III The quality of study data relies first and foremost on the quality
KCR Data Management: Designed for Full Data Transparency 6% Observational 4% Phase IV 2% Device 11% Phase I 28% Phase II 49% Phase III The quality of study data relies first and foremost on the quality
Wireless Patient Monitoring
 Wireless Patient Monitoring Live ECG streaming for mobile users and in-hospital ICU units. Full network integration with WAP Access point technology and Wi-Fi. Optional web server upload for telemedicine
Wireless Patient Monitoring Live ECG streaming for mobile users and in-hospital ICU units. Full network integration with WAP Access point technology and Wi-Fi. Optional web server upload for telemedicine
LEADING INNOVATION IN DATA AND TELEMATICS. www.iris-technology.co.uk
 LEADING INNOVATION IN DATA AND TELEMATICS About IRIS IRIS Technology was formed in 2001 to address offshore subsea telemetry markets and is now one of the leading specialist telematics companies in Europe
LEADING INNOVATION IN DATA AND TELEMATICS About IRIS IRIS Technology was formed in 2001 to address offshore subsea telemetry markets and is now one of the leading specialist telematics companies in Europe
MOBILE HEALTHCARE APPDRAGON SMARTMED
 APPDRAGON SMARTMED With a rapid increase of chronic conditions such as diabetes, obesity, heart disease and respiratory problems, healthcare providers are under increasing pressure to deliver quality care,
APPDRAGON SMARTMED With a rapid increase of chronic conditions such as diabetes, obesity, heart disease and respiratory problems, healthcare providers are under increasing pressure to deliver quality care,
24/7 Monitoring Pro-Active Support High Availability Hardware & Software Helpdesk. itg CloudBase
 24/7 Monitoring Pro-Active Support High Availability Hardware & Software Helpdesk Onsite Support itg CloudBase Pro-Active managed it support services for one single cost per month covers all aspects of
24/7 Monitoring Pro-Active Support High Availability Hardware & Software Helpdesk Onsite Support itg CloudBase Pro-Active managed it support services for one single cost per month covers all aspects of
Aligning Quality Management Processes to Compliance Goals
 Aligning Quality Management Processes to Compliance Goals MetricStream.com Smart Consulting Group Joint Webinar February 23 rd 2012 Nigel J. Smart, Ph.D. Smart Consulting Group 20 E. Market Street West
Aligning Quality Management Processes to Compliance Goals MetricStream.com Smart Consulting Group Joint Webinar February 23 rd 2012 Nigel J. Smart, Ph.D. Smart Consulting Group 20 E. Market Street West
GE Healthcare. The new MAC * 800
 GE Healthcare The new MAC * 800 Fits your needs wherever you are. Advanced ECG technology can make a real difference in patient care. As long as it s working for you and not the other way around. The new
GE Healthcare The new MAC * 800 Fits your needs wherever you are. Advanced ECG technology can make a real difference in patient care. As long as it s working for you and not the other way around. The new
eclinical Services Predictable Pricing Full Service EDC Phase I-IV Sophisticated Edit Checks Drug Supply Chain Forms Library Data Collection Services
 eclinical Services Predictable Pricing Full Service EDC Phase I-IV Sophisticated Edit Checks Data Collection Services Drug Supply Chain Forms Library Real-time Data Access Clinical Data Management Electronic
eclinical Services Predictable Pricing Full Service EDC Phase I-IV Sophisticated Edit Checks Data Collection Services Drug Supply Chain Forms Library Real-time Data Access Clinical Data Management Electronic
AssurX Makes Quality & Compliance a Given Not Just a Goal
 AssurX Makes Quality & Compliance a Given Not Just a Goal TRACK. MANAGE. AUTOMATE. IMPROVE. AssurX s powerfully flexible software unites and coordinates information, activities and documentation in one
AssurX Makes Quality & Compliance a Given Not Just a Goal TRACK. MANAGE. AUTOMATE. IMPROVE. AssurX s powerfully flexible software unites and coordinates information, activities and documentation in one
Captus 3000 Thyroid Uptake And Well System
 (201) 825-9500 / (800) 631-3826 THRYOID UPTAKE SYSTEMS 51 Captus 3000 Thyroid Uptake And Well System The Ultimate... User Friendly... Intuitive Performing Thyroid Uptake measurements has never been easier.
(201) 825-9500 / (800) 631-3826 THRYOID UPTAKE SYSTEMS 51 Captus 3000 Thyroid Uptake And Well System The Ultimate... User Friendly... Intuitive Performing Thyroid Uptake measurements has never been easier.
epro Integration with Oracle Remote Data Capture
 epro Integration with Oracle Remote Data Capture BIAS Congress 27-28 May 2010 Rome (IT) Davide Di Giovine, CROS NT Senior DM & CSV Manager davide.digiovine@cros.it AGENDA Changes in handling of clinical
epro Integration with Oracle Remote Data Capture BIAS Congress 27-28 May 2010 Rome (IT) Davide Di Giovine, CROS NT Senior DM & CSV Manager davide.digiovine@cros.it AGENDA Changes in handling of clinical
On Demand Customer Feedback at the Point of Experience
 On Demand Customer Feedback at the Point of Experience For further information, please contact: Morgan Strickland, CEO Opinionmeter International, Ltd. 510-352-4943, x101 morgan@opinionmeter.com www.opinionmeter.com
On Demand Customer Feedback at the Point of Experience For further information, please contact: Morgan Strickland, CEO Opinionmeter International, Ltd. 510-352-4943, x101 morgan@opinionmeter.com www.opinionmeter.com
Intelligent Infrastructure Solutions. imvision. Infrastructure Management. Made Easy.
 TM Intelligent Infrastructure Solutions imvision. Infrastructure Management. Made Easy. Every network must be managed. The question is HOW? The vision: Business success that starts with better-managed
TM Intelligent Infrastructure Solutions imvision. Infrastructure Management. Made Easy. Every network must be managed. The question is HOW? The vision: Business success that starts with better-managed
Introduction. The Evolution of the Data Management Role: The Clinical Data Liaison
 Introduction The CDL is a new role that will become a standard in the industry for companies that want to make more efficient use of limited resources: time and money. A CDL is key in that he or she conducts
Introduction The CDL is a new role that will become a standard in the industry for companies that want to make more efficient use of limited resources: time and money. A CDL is key in that he or she conducts
Guidance for Industry Computerized Systems Used in Clinical Investigations
 Guidance for Industry Computerized Systems Used in Clinical Investigations U.S. Department of Health and Human Services Food and Drug Administration (FDA) Office of the Commissioner (OC) May 2007 Guidance
Guidance for Industry Computerized Systems Used in Clinical Investigations U.S. Department of Health and Human Services Food and Drug Administration (FDA) Office of the Commissioner (OC) May 2007 Guidance
Genesis II. revolutionise monitoring. Why Tutela Genesis II. delivering medical monitoring worldwide
 Genesis II revolutionise monitoring Why Tutela Genesis II Simply because Tutela Genesis II is the most complete, robust, flexible and future-proof solution to data monitoring in medical and scientific
Genesis II revolutionise monitoring Why Tutela Genesis II Simply because Tutela Genesis II is the most complete, robust, flexible and future-proof solution to data monitoring in medical and scientific
Execution in Clinical Research
 TM TM Execution in Clinical Research Unrivaled CRA training program drives success for Sponsors. Sponsors have many choices in choosing development partners at the CRO level. The need for a documented
TM TM Execution in Clinical Research Unrivaled CRA training program drives success for Sponsors. Sponsors have many choices in choosing development partners at the CRO level. The need for a documented
Skill Levels for Delivering High Quality Asthma and COPD Respiratory Care by Nurses in Primary Care
 Skill Levels for Delivering High Quality Asthma and COPD Respiratory Care by Nurses in Primary Care September 2007 Revised December 2009, April 2014 Author: Ruth McArthur In conjunction with the PCRS-UK
Skill Levels for Delivering High Quality Asthma and COPD Respiratory Care by Nurses in Primary Care September 2007 Revised December 2009, April 2014 Author: Ruth McArthur In conjunction with the PCRS-UK
Cellular Wireless technology: Creating a link between people and the healthcare community
 Cellular Wireless technology: Creating a link between people and the healthcare community Introduction Demands on health-care systems worldwide have increased to the point where the delivery and cost of
Cellular Wireless technology: Creating a link between people and the healthcare community Introduction Demands on health-care systems worldwide have increased to the point where the delivery and cost of
GE Healthcare. Avance Carestation. Innovating with you, shaping exceptional care
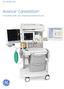 GE Healthcare Avance Carestation Innovating with you, shaping exceptional care Clinician inspired perioperative solutions GE s Avance Carestation was developed using an approach to perioperative solutions
GE Healthcare Avance Carestation Innovating with you, shaping exceptional care Clinician inspired perioperative solutions GE s Avance Carestation was developed using an approach to perioperative solutions
What We Are..! www.ardent-cro.com
 Your Trusted CRO! Regus, Level-2, Connaught Place, Bund Garden Road, Pune-411001, MH, India. Phone: 020-65-31-31-71 Email: ardent@ardent-cro.com Web: What We Are..! Ardent Clinical Research Services is
Your Trusted CRO! Regus, Level-2, Connaught Place, Bund Garden Road, Pune-411001, MH, India. Phone: 020-65-31-31-71 Email: ardent@ardent-cro.com Web: What We Are..! Ardent Clinical Research Services is
Results Q2 &H1. August 24, 2001
 Results Q2 &H1 August 24, 2001 Contents Section 1 Company overview and highlights Q2 2001 Erez Alroy, Co-President Section 2 Insight Information Technology Irit Alroy, VP Technology Development Section
Results Q2 &H1 August 24, 2001 Contents Section 1 Company overview and highlights Q2 2001 Erez Alroy, Co-President Section 2 Insight Information Technology Irit Alroy, VP Technology Development Section
CARE HOME MANAGEMENT SOFTWARE
 CARE HOME MANAGEMENT SOFTWARE // CONTENTS 02 About Us 03 Introducing CoolCare4 05 Dashboard 07 Caring for Residents 11 Care Home Staff 15 Home Management 17 Virtual Notice Board 18 Performance Analysis
CARE HOME MANAGEMENT SOFTWARE // CONTENTS 02 About Us 03 Introducing CoolCare4 05 Dashboard 07 Caring for Residents 11 Care Home Staff 15 Home Management 17 Virtual Notice Board 18 Performance Analysis
EHR 101. A Guide to Successfully Implementing Electronic Health Records
 EHR 101 A Guide to Successfully Implementing Electronic Health Records Electronic health records are the inevitable next step in the continued progress of U.S. healthcare. Medicine may be the most information-intensive
EHR 101 A Guide to Successfully Implementing Electronic Health Records Electronic health records are the inevitable next step in the continued progress of U.S. healthcare. Medicine may be the most information-intensive
How to implement a Quality Management System
 How to implement a Quality Management System This whitepaper will help you to implement a Quality Management System (QMS), based on Good Manufacturing Practice (GMP), ISO 9001 or ISO 13485 within your
How to implement a Quality Management System This whitepaper will help you to implement a Quality Management System (QMS), based on Good Manufacturing Practice (GMP), ISO 9001 or ISO 13485 within your
QUMAS Remote Assist Program
 QUMAS Remote Assist Program A tailored approach to day-to-day application, server and change management, which is integrated into your existing support and SLA requirements to ensure optimal performance
QUMAS Remote Assist Program A tailored approach to day-to-day application, server and change management, which is integrated into your existing support and SLA requirements to ensure optimal performance
INTRODUCING... COMPLETE SERVICE. The Professional Service Desk Solution
 INTRODUCING... COMPLETE SERVICE The Professional Service Desk Solution COMPLETE SERVICE OVERVIEW Complete Service is the next generation of cost-effective service desk and field engineer co-ordination
INTRODUCING... COMPLETE SERVICE The Professional Service Desk Solution COMPLETE SERVICE OVERVIEW Complete Service is the next generation of cost-effective service desk and field engineer co-ordination
DATA MANAGEMENT IN CLINICAL TRIALS: GUIDELINES FOR RESEARCHERS
 Reference Number: UHB 139 Version Number: 2 Date of Next Review: 14 Apr 2018 Previous Trust/LHB Reference Number: N/A DATA MANAGEMENT IN CLINICAL TRIALS: GUIDELINES FOR RESEARCHERS Introduction and Aim
Reference Number: UHB 139 Version Number: 2 Date of Next Review: 14 Apr 2018 Previous Trust/LHB Reference Number: N/A DATA MANAGEMENT IN CLINICAL TRIALS: GUIDELINES FOR RESEARCHERS Introduction and Aim
Mayo Alliance for Clinical Trials
 Mayo Alliance for Clinical Trials A Division within Mayo Clinical Trial Services PROFESSIONAL CLINICAL TRIALS SERVICES ADVANCING PATIENT CARE THROUGH SCIENTIFIC DISCOVERY It is a great thing to make scientific
Mayo Alliance for Clinical Trials A Division within Mayo Clinical Trial Services PROFESSIONAL CLINICAL TRIALS SERVICES ADVANCING PATIENT CARE THROUGH SCIENTIFIC DISCOVERY It is a great thing to make scientific
PARTNER SEARCH FORM. Aware Engineering. Newton USA. Robert CEO. 1. Company Profile 1.1 General Profile Full Name of Company.
 1. Company Profile 1.1 General Profile Full Name of Company Aware Engineering Registration # Prior name of Company Type of Company HT R&D Research Institute University Other Stage Seed R&D Initial Revenues
1. Company Profile 1.1 General Profile Full Name of Company Aware Engineering Registration # Prior name of Company Type of Company HT R&D Research Institute University Other Stage Seed R&D Initial Revenues
Guidance for Industry
 Guidance for Industry Electronic Source Data in Clinical Investigations U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) Center for
Guidance for Industry Electronic Source Data in Clinical Investigations U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) Center for
Printgroove JT Suite. The complete print process fast, simple and reliable. Applications Printgroove JT Suite
 Printgroove JT Suite The complete print process fast, simple and reliable Applications Printgroove JT Suite Printgroove JT Suite Automation for the print workflow Both in-house print facilities and external
Printgroove JT Suite The complete print process fast, simple and reliable Applications Printgroove JT Suite Printgroove JT Suite Automation for the print workflow Both in-house print facilities and external
The MedStar Remote Patient Monitoring System
 The MedStar Remote Patient Monitoring System Cybernet Medical Contact Information: Dr. Charles Jacobus Phone: 734-668-2567 Web: www.cybernet.com www.cybernetmedical.com Mail: 3885 Research Park Drive E-Mail:
The MedStar Remote Patient Monitoring System Cybernet Medical Contact Information: Dr. Charles Jacobus Phone: 734-668-2567 Web: www.cybernet.com www.cybernetmedical.com Mail: 3885 Research Park Drive E-Mail:
SmartBox-V A Comprehensive range of new functionalities.
 SMARTBOX-V A COMPREHENSIVE RANGE OF NEW FUNCTIONALITIES. Issue 1 SmartBox-V A Comprehensive range of new functionalities. January 2014 NETWORK INTELIGENE DEVICE (NID) FOR THE MARITIME INDUSTRY IN THIS
SMARTBOX-V A COMPREHENSIVE RANGE OF NEW FUNCTIONALITIES. Issue 1 SmartBox-V A Comprehensive range of new functionalities. January 2014 NETWORK INTELIGENE DEVICE (NID) FOR THE MARITIME INDUSTRY IN THIS
Whole Systems Demonstrators An Overview of Telecare and Telehealth
 Whole Systems Demonstrators An Overview of Telecare and Telehealth The WSD Programme The WSD Programme The Whole System Demonstrator (WSD) programme is a two year research project funded by the Department
Whole Systems Demonstrators An Overview of Telecare and Telehealth The WSD Programme The WSD Programme The Whole System Demonstrator (WSD) programme is a two year research project funded by the Department
FDA Regulation of Electronic Source Data in Clinical Investigations
 FDA Regulation of Electronic Source Data in Clinical Investigations Q1 Productions Second Annual Innovations in Clinical Data Management Conference, Arlington, VA Mahnu Davar Presented on Oct. 28, 2014
FDA Regulation of Electronic Source Data in Clinical Investigations Q1 Productions Second Annual Innovations in Clinical Data Management Conference, Arlington, VA Mahnu Davar Presented on Oct. 28, 2014
510(k) Summary May 7, 2012
 510(k) Summary Medicalgorithmics 510(k) Premarket Notification 510(k) Summary May 7, 2012 1. Submitter Name and Address Medicalgorithmics LLC 245 West 107th St., Suite 11A New York, NY 10025, USA Contact
510(k) Summary Medicalgorithmics 510(k) Premarket Notification 510(k) Summary May 7, 2012 1. Submitter Name and Address Medicalgorithmics LLC 245 West 107th St., Suite 11A New York, NY 10025, USA Contact
Computerised Systems. Seeing the Wood from the Trees
 Computerised Systems Seeing the Wood from the Trees Scope WHAT IS A COMPUTERISED SYSTEM? WHY DO WE NEED VALIDATED SYSTEMS? WHAT NEEDS VALIDATING? HOW DO WE PERFORM CSV? WHO DOES WHAT? IT S VALIDATED -
Computerised Systems Seeing the Wood from the Trees Scope WHAT IS A COMPUTERISED SYSTEM? WHY DO WE NEED VALIDATED SYSTEMS? WHAT NEEDS VALIDATING? HOW DO WE PERFORM CSV? WHO DOES WHAT? IT S VALIDATED -
Role of eclinical Systems in FDA Regulated Studies. Jonathan S. Helfgott
 Role of eclinical Systems in FDA Regulated Studies Presented by: Jonathan S. Helfgott Operations Research Analyst Office of Scientific Investigations Disclaimer The contents of this presentation are my
Role of eclinical Systems in FDA Regulated Studies Presented by: Jonathan S. Helfgott Operations Research Analyst Office of Scientific Investigations Disclaimer The contents of this presentation are my
The Pension Portal. Helping you take your pension business into the paperless age
 The Pension Portal Helping you take your pension business into the paperless age When you ve been helping pension professionals implement client portals for as long as we have, you understand that the
The Pension Portal Helping you take your pension business into the paperless age When you ve been helping pension professionals implement client portals for as long as we have, you understand that the
PACS A WEB-BASED APPLICATION DESIGNED TO AUTOMATE YOUR WORKFLOW.
 A WEB-BASED APPLICATION DESIGNED TO AUTOMATE YOUR WORKFLOW. Our Company Our Mission is Simple Our company is Canadian and was established in 1994. After 20 years, we continue to be a leading IT software
A WEB-BASED APPLICATION DESIGNED TO AUTOMATE YOUR WORKFLOW. Our Company Our Mission is Simple Our company is Canadian and was established in 1994. After 20 years, we continue to be a leading IT software
SERVICE PLANS. More Productivity. Less Worry. SERVICE PLANS
 SERVICE PLANS More Productivity. Less Worry. SERVICE PLANS Flexible service plans to help you maximize the value of your technology investment. Let us help you improve your laboratory productivity and
SERVICE PLANS More Productivity. Less Worry. SERVICE PLANS Flexible service plans to help you maximize the value of your technology investment. Let us help you improve your laboratory productivity and
FDA Issues Final Guidance on Mobile Medical Apps
 ADVISORY September 2013 FDA Issues Final Guidance on Mobile Medical Apps On September 23, 2013, the U.S. Food & Drug Administration (FDA or the Agency) issued its final Guidance for Industry and Food and
ADVISORY September 2013 FDA Issues Final Guidance on Mobile Medical Apps On September 23, 2013, the U.S. Food & Drug Administration (FDA or the Agency) issued its final Guidance for Industry and Food and
Get EnABELed with ABELMed
 Get EnABELed with ABELMed Electronic Health Record and Practice Management Solutions We focus on your Practice... So you can focus on your Patients Start reaping benefits of ABELMed EHR from day one: Eliminate
Get EnABELed with ABELMed Electronic Health Record and Practice Management Solutions We focus on your Practice... So you can focus on your Patients Start reaping benefits of ABELMed EHR from day one: Eliminate
Data Capture Solutions for Manufacturing Applications
 Manufacturing Data Capture Solutions for Manufacturing Applications Create a Non-Stop Production Line and Cost-Effective Solutions for Manufacturing The growing complexity of manufacturing requires an
Manufacturing Data Capture Solutions for Manufacturing Applications Create a Non-Stop Production Line and Cost-Effective Solutions for Manufacturing The growing complexity of manufacturing requires an
LIFENET System PRODUCT BROCHURE
 LIFENET System PRODUCT BROCHURE LIFENET System In the rig or in the ED, more efficiency with your equipment, your information, and your decisions means working better. And that means helping save more
LIFENET System PRODUCT BROCHURE LIFENET System In the rig or in the ED, more efficiency with your equipment, your information, and your decisions means working better. And that means helping save more
The evolution of data connectivity
 Leveraging the Benefits of IP and the Cloud in the Security Sector The CCTV and alarm industry has relied on analogue or Integrated Services Digital Network (ISDN) communications to provide data connectivity
Leveraging the Benefits of IP and the Cloud in the Security Sector The CCTV and alarm industry has relied on analogue or Integrated Services Digital Network (ISDN) communications to provide data connectivity
Bring Your Own Device (BYOD) Approaches To The Collection Of Electronic Patient Reported Outcome Data
 Bring Your Own Device (BYOD) Approaches To The Collection Of Electronic Patient Reported Outcome Data Chad Gwaltney, Ph.D. Chief Scientist and Regulatory Advisor, Endpoints 1 Disclaimer The views and opinions
Bring Your Own Device (BYOD) Approaches To The Collection Of Electronic Patient Reported Outcome Data Chad Gwaltney, Ph.D. Chief Scientist and Regulatory Advisor, Endpoints 1 Disclaimer The views and opinions
Guidance for CTUs on Assessing the Suitability of Laboratories Processing Research Samples
 Guidance for CTUs on Assessing the Suitability of Laboratories Processing Research Samples UKCRC Registered CTUs Network Guidance for CTUs on Assessing the Suitability of Laboratories Processing Research
Guidance for CTUs on Assessing the Suitability of Laboratories Processing Research Samples UKCRC Registered CTUs Network Guidance for CTUs on Assessing the Suitability of Laboratories Processing Research
Meeting Priorities of Biotech & Small Pharma Companies
 Meeting Priorities of Biotech & Small Pharma Companies Dr. Andreas Orfanos, MBBCH,FFPM,MBA Thrasos Therapeutics Inc. Vice President Clinical Research & Development 28 Oct 2015 Montreal Technoparc Private,
Meeting Priorities of Biotech & Small Pharma Companies Dr. Andreas Orfanos, MBBCH,FFPM,MBA Thrasos Therapeutics Inc. Vice President Clinical Research & Development 28 Oct 2015 Montreal Technoparc Private,
digital Vitals BY MIDMARK vitals
 digital Vitals BY MIDMARK IQ IQ Midmark IQ is an affordable, Windows based, touchscreen device that provides automated blood pressure, temperature, SpO 2 and pulse rate readings with a touch of the screen.
digital Vitals BY MIDMARK IQ IQ Midmark IQ is an affordable, Windows based, touchscreen device that provides automated blood pressure, temperature, SpO 2 and pulse rate readings with a touch of the screen.
10 QUESTIONS TO ASK ECG SOLUTION PROVIDERS
 HOW TO SELECT THE BEST ECG MANAGEMENT SOLUTION 10 QUESTIONS TO ASK ECG SOLUTION PROVIDERS BY STEPHEN CHERNOFF October 15, 2009 Inside 1 The Challenge 2 Testing Equipment 3 Customization 4 Interfacing 5
HOW TO SELECT THE BEST ECG MANAGEMENT SOLUTION 10 QUESTIONS TO ASK ECG SOLUTION PROVIDERS BY STEPHEN CHERNOFF October 15, 2009 Inside 1 The Challenge 2 Testing Equipment 3 Customization 4 Interfacing 5
Device Lifecycle Management
 Device Lifecycle Management 1 (8) Table of Contents 1. Executive summary... 3 2. Today's challenges in adapting to lifecycle management... 3 3. How is Miradore different?... 5 4. Conclusion... 8 2 (8)
Device Lifecycle Management 1 (8) Table of Contents 1. Executive summary... 3 2. Today's challenges in adapting to lifecycle management... 3 3. How is Miradore different?... 5 4. Conclusion... 8 2 (8)
GE Energy. Solutions
 GE Energy Wind Service Solutions Speed, competency and technology... Faster return to service GE s wind turbine fleet is one of the fastest growing and most reliable in the world. Building on a strong
GE Energy Wind Service Solutions Speed, competency and technology... Faster return to service GE s wind turbine fleet is one of the fastest growing and most reliable in the world. Building on a strong
Product. Velocity Gain Efficiencies and Improve Loan Quality with a Comprehensive, Open Architecture Loan Origination Solution
 Product Velocity Gain Efficiencies and Improve Loan Quality with a Comprehensive, Open Architecture Loan Origination Solution Product To compete successfully for quality loans, credit unions need a process
Product Velocity Gain Efficiencies and Improve Loan Quality with a Comprehensive, Open Architecture Loan Origination Solution Product To compete successfully for quality loans, credit unions need a process
Workflow Solutions Data Collection, Data Review and Data Management
 Data Collection, Data Review and Data Management Workflow Finding more efficient ways to support patient needs begins with better workflow management. MGC Diagnostics has developed a complete workflow
Data Collection, Data Review and Data Management Workflow Finding more efficient ways to support patient needs begins with better workflow management. MGC Diagnostics has developed a complete workflow
Mobile Medical Application Development: FDA Regulation
 Mobile Medical Application Development: FDA Regulation Mobile Medical Applications: Current Environment Currently over 100,000 mobile health related applications are available for download; there is an
Mobile Medical Application Development: FDA Regulation Mobile Medical Applications: Current Environment Currently over 100,000 mobile health related applications are available for download; there is an
Data Management Unit Research Institute for Health Sciences, Chiang Mai University
 Data Management Unit Research Institute for Health Sciences, Chiang Mai University Clinical Data Management is the process of handling data from clinical trials. The inherent goal of any clinical data
Data Management Unit Research Institute for Health Sciences, Chiang Mai University Clinical Data Management is the process of handling data from clinical trials. The inherent goal of any clinical data
BT Cloud Phone for your business. A guide to our IP Phones portfolio.
 BT Cloud Phone for your business. A guide to our IP Phones portfolio. 2 Contents. 3 The IP phone range from BT Business. 3 An overview BT Polycom VVX range 4 An overview BT Yealink W52-P 5 An overview
BT Cloud Phone for your business. A guide to our IP Phones portfolio. 2 Contents. 3 The IP phone range from BT Business. 3 An overview BT Polycom VVX range 4 An overview BT Yealink W52-P 5 An overview
The Strategic Laboratory Portal.
 molis vt molis vt channel molis vt communicator molis vt billing molis vt insight molis vt channel The Strategic Laboratory Portal. 1 The Strategic Laboratory Portal. Product Overview The overall efficiency
molis vt molis vt channel molis vt communicator molis vt billing molis vt insight molis vt channel The Strategic Laboratory Portal. 1 The Strategic Laboratory Portal. Product Overview The overall efficiency
Top5 Considerations for Choosing Practice Information Management System (PIMS)
 Top5 Considerations for Choosing Practice Information Management System (PIMS) Top 5 Considerations for Choosing a Practice Information Management System (PIMS) Software Features page 2 Hardware Requirements
Top5 Considerations for Choosing Practice Information Management System (PIMS) Top 5 Considerations for Choosing a Practice Information Management System (PIMS) Software Features page 2 Hardware Requirements
Full-time or part-time upto 1.0 FTE, 4 year fix term Principal Investigator Department of Cardiology Westmead Hospital
 Position Description POSITION TITLE Project Manager TYPE OF EMPLOYMENT Full-time or part-time upto 1.0 FTE, 4 year fix term REPORTING TO a) Principal Investigator DIVISION Department of Cardiology COMPANY
Position Description POSITION TITLE Project Manager TYPE OF EMPLOYMENT Full-time or part-time upto 1.0 FTE, 4 year fix term REPORTING TO a) Principal Investigator DIVISION Department of Cardiology COMPANY
With HD quality Full transparent networking features And on-demand capacity enhancements
 Always more than you expect Panasonic NS1000 SIP BUSINESS COMMUNICATIONS SERVER With HD quality Full transparent networking features And on-demand capacity enhancements The NS1000 at a glance SIP and IP
Always more than you expect Panasonic NS1000 SIP BUSINESS COMMUNICATIONS SERVER With HD quality Full transparent networking features And on-demand capacity enhancements The NS1000 at a glance SIP and IP
Fully Managed IT Support. Proactive Maintenance. Disaster Recovery. Remote Support. Service Desk. Call Centre. Fully Managed Services Guide July 2007
 Fully Managed IT Support Proactive Maintenance Disaster Recovery Remote Support Service Desk Call Centre London Office ITVET Limited 2 nd Floor 145 157 St John Street London EC1V 4PY t: +44(0)8704 232
Fully Managed IT Support Proactive Maintenance Disaster Recovery Remote Support Service Desk Call Centre London Office ITVET Limited 2 nd Floor 145 157 St John Street London EC1V 4PY t: +44(0)8704 232
WWW.PACOM.COM. Pacom Systems. All rights reserved.
 WWW.PACOM.COM Pacom Systems. All rights reserved. SECURITY SOLUTIONS MANAGED SERVICES Did you know? Research shows that a remotely managed system can reduce false alarms by more than 90%.? 3 ? Managed
WWW.PACOM.COM Pacom Systems. All rights reserved. SECURITY SOLUTIONS MANAGED SERVICES Did you know? Research shows that a remotely managed system can reduce false alarms by more than 90%.? 3 ? Managed
86 C Freezer Accessories. More Choices. Choose from a Wide Selection of Racks, Monitoring Systems and Other New Brunswick Freezer Accessories
 86 C Freezer Accessories More Choices Choose from a Wide Selection of Racks, Monitoring Systems and Other New Brunswick Freezer Accessories 2 A Wide Range of Accessories The perfect complement for your
86 C Freezer Accessories More Choices Choose from a Wide Selection of Racks, Monitoring Systems and Other New Brunswick Freezer Accessories 2 A Wide Range of Accessories The perfect complement for your
A complete, 24-hour HD video EEG solution
 A complete, 24-hour HD video EEG solution PORTABLE & PATIENT FRIENDLY LifeLines ieeg Portable comprises the most critical features requested by leading clinicians and researchers around the globe: Immediate
A complete, 24-hour HD video EEG solution PORTABLE & PATIENT FRIENDLY LifeLines ieeg Portable comprises the most critical features requested by leading clinicians and researchers around the globe: Immediate
Networked versatility. IntelliVue MP60 and MP70 patient monitors
 Networked versatility IntelliVue MP60 and MP70 patient monitors Critical performance The IntelliVue family of networked patient monitors gives care teams throughout the hospital more of the information
Networked versatility IntelliVue MP60 and MP70 patient monitors Critical performance The IntelliVue family of networked patient monitors gives care teams throughout the hospital more of the information
Sensors / Modules / Monitors. Breath Gas Sensing and Monitoring. For life supporting systems in intensive, emergency and ambulant care
 Sensors / Modules / Monitors Breath Gas Sensing and Monitoring For life supporting systems in intensive, emergency and ambulant care Your competent partner for breath gas sensing and monitoring EnviteC
Sensors / Modules / Monitors Breath Gas Sensing and Monitoring For life supporting systems in intensive, emergency and ambulant care Your competent partner for breath gas sensing and monitoring EnviteC
Accenture Accelerated R&D Services:
 Accenture Life Sciences Rethink Reshape Restructure for better patient outcomes Accenture Accelerated R&D Services: Pharmacovigilance Services Overview At our company, we are focused on improving lives
Accenture Life Sciences Rethink Reshape Restructure for better patient outcomes Accenture Accelerated R&D Services: Pharmacovigilance Services Overview At our company, we are focused on improving lives
Verizon Wireless In-Building Distributed Antenna System Featuring ADX Series Fiber DAS By Advanced RF Technologies
 Verizon Wireless In-Building Distributed Antenna System Featuring ADX Series Fiber DAS By Advanced RF Technologies Venue: Hewlett Packard, Windward Campus 5555 Windward Parkway Alpharetta, GA 30004 A White
Verizon Wireless In-Building Distributed Antenna System Featuring ADX Series Fiber DAS By Advanced RF Technologies Venue: Hewlett Packard, Windward Campus 5555 Windward Parkway Alpharetta, GA 30004 A White
Standard Operating Procedure on Training Requirements for staff participating in CTIMPs Sponsored by UCL
 Page 1 of 10 Standard Operating Procedure on Training Requirements for staff participating in CTIMPs Sponsored by UCL SOP ID Number: Effective Date:01/08/2012 Version Number & Date of Authorisation: V02,
Page 1 of 10 Standard Operating Procedure on Training Requirements for staff participating in CTIMPs Sponsored by UCL SOP ID Number: Effective Date:01/08/2012 Version Number & Date of Authorisation: V02,
High-intensity performance. IntelliVue MP90 patient monitor
 High-intensity performance IntelliVue MP90 patient monitor See a higher level The IntelliVue family of networked patient monitors gives care teams throughout the hospital more of the information they need
High-intensity performance IntelliVue MP90 patient monitor See a higher level The IntelliVue family of networked patient monitors gives care teams throughout the hospital more of the information they need
