Mayo Alliance for Clinical Trials
|
|
|
- Chastity Dean
- 10 years ago
- Views:
Transcription
1 Mayo Alliance for Clinical Trials A Division within Mayo Clinical Trial Services PROFESSIONAL CLINICAL TRIALS SERVICES ADVANCING PATIENT CARE THROUGH SCIENTIFIC DISCOVERY It is a great thing to make scientific discoveries of rare value, but it is even greater to be willing to share these discoveries and to encourage other workers in the same field of scientific research. William J. Mayo, M.D.
2 Mayo Alliance for Clinical Trials Mayo Alliance for Clinical Trials (Mayo ACT) was formed to offer the best of both worlds that is, the operational efficiencies of contract research organizations and the clinical and scientific excellence of an academic research organization to provide professional clinical trial management services committed to: Academic Center Mayo Trials n Quality Timeliness Personal Attention Stability Quality Scientific and data integrity are ensured through the integration of clinical excellence with a proven operational infrastructure. We ve responded to your needs by merging the operational efficiencies of CROs with the clinical excellence of AROs to provide science and service excellence. Timeliness Faster regulatory approval is supported through our validated, reliable processes and measures. Personal Attention Dedicated Mayo project management teams proactively collaborate with your management teams to monitor study progress and respond to issues quickly and decisively. Stability Our long term focus to clinical excellence provides you with a stable team of professionals during the entire duration of your study. Mayo Alliance for Clinical Trials is a fully self-contained group bringing dedicated contract, regulatory, quality and data management expertise through a service provision committed to excellence and timeliness.
3 SERVICES FEATURES Protocol Consultation is available and includes scientific review and clinical/biostatistical input for sponsor protocols. Mayo ACT works closely with many Mayo Clinic research professionals involved in over 5,000 active protocols. Site Recruitment and Qualification efforts are ongoing and selective. Recruitment sources are national in scope and target investigators from among the most active specialties. Trial Management services are centralized for efficiency and responsiveness. Our teams feature a single point of contact who manages processes to help integrate your needs with our team members. Regulatory Management is committed to proactively identify and resolve any regulatory issues that stand in the way of a quality and timely study outcome. Legal/Contract Management is dedicated to industry-sponsored research and provides rapid, responsive service for expedited contract execution. Data Management begins with a validated system, monitored by quality and productivity rules, and designed to ensure data consistency. Biostatistics expertise is provided by experienced professionals focused on the accurate collection, organization and analysis of clinical data for faster regulatory review and study completion. Quality Assurance measures such as document management, personnel competency and training and our Quality System Essentials (QSE) program ensure the reliability you expect. Mayo Alliance for Clinical Trials is dedicated to quality study results that can only come about through personalized service conducted in a timely, cost effective manner. Our mission is to help you advance the science of medicine through the merging of scientific and service excellence.
4 SERVICES OVERVIEW Mayo Alliance for Clinical Trials provides a full range of integrated services designed to complete your study on time and on budget. These services include: Protocol Consultation Site Recruitment and Qualification Trial Management Regulatory Management Legal/Contract Management Data Management Biostatistics Quality Assurance Additional Services Supporting Clinical Trials Mayo Clinical Trial Services can provide important service line support in addition to Mayo ACT. Mayo Central Laboratory for Clinical Trials can provide medical and scientific attention to rapid assay development for the diagnostic test component of your studies. Our 58 specialized laboratories are among the most recognized in the world in terms of quality and reliability. M I Elevation Mayo Angio and ECG Core Laboratories are designed for the rigors of clinical trials and are under the direction of Mayo Clinic staff cardiologists providing computer-assisted analysis in a validated setting.
5 Mayo Alliance for Clinical Trials Advancing patient care through scientific discovery. For more information on Mayo Alliance for Clinical Trials please contact: Phone: Fax: Stabile Building 150 Third Street SW Rochester, Minnesota , Mayo Foundation for Medical Education and Research (MFMER). All rights reserved. MAYO, Mayo Clinical Trial Services and the triple-shield Mayo logo are trademarks and/or service marks of MFMER. MC2285/R0602
Mayo Central Laboratory for Clinical Trials A Division within Mayo Clinical Trial Services
 Mayo Central Laboratory for Clinical Trials A Division within Mayo Clinical Trial Services LABORATORY TESTING SERVICES YOUR PARTNER IN LABORATORY MEDICINE The key note of progress in the 20th century is
Mayo Central Laboratory for Clinical Trials A Division within Mayo Clinical Trial Services LABORATORY TESTING SERVICES YOUR PARTNER IN LABORATORY MEDICINE The key note of progress in the 20th century is
m 4 Biobank Alliance & m 4 Trial Service Center
 m 4 Biobank Alliance & m 4 Trial Service Center Services & Consulting in (non-)clinical development www.m4.de m 4 Biobank Alliance Central Access to High Quality Human Biospecimens for R&D The m 4 Biobank
m 4 Biobank Alliance & m 4 Trial Service Center Services & Consulting in (non-)clinical development www.m4.de m 4 Biobank Alliance Central Access to High Quality Human Biospecimens for R&D The m 4 Biobank
and Regulatory Aspects
 Good Clinical Practice and Regulatory Aspects Nora Espiritu MD, MPh, PhD (c) Former Executive Director of Research and Technology Transfer. Peruvian National Institute t of Health. Member of the Ethics
Good Clinical Practice and Regulatory Aspects Nora Espiritu MD, MPh, PhD (c) Former Executive Director of Research and Technology Transfer. Peruvian National Institute t of Health. Member of the Ethics
exactly. The need for efficiency in developing effective new therapeutics has never been greater.
 exactly. The need for efficiency in developing effective new therapeutics has never been greater. As demands on the global healthcare system increase and treating disease becomes more complex, the research,
exactly. The need for efficiency in developing effective new therapeutics has never been greater. As demands on the global healthcare system increase and treating disease becomes more complex, the research,
General Clinical Research Center at Stony Brook
 General Clinical Research Center at Stony Brook General Clinical Research Center at Stony Brook National Institute of Health funded for over $1 million per year for center s operation to help improve health
General Clinical Research Center at Stony Brook General Clinical Research Center at Stony Brook National Institute of Health funded for over $1 million per year for center s operation to help improve health
Organization and Job Profile
 Organization and Job Profile New Orleans, Louisiana Position: Reports to: Location: System Vice President for Research New Orleans, Louisiana CORPORATE BACKGROUND is a non-profit, academic, multi-specialty,
Organization and Job Profile New Orleans, Louisiana Position: Reports to: Location: System Vice President for Research New Orleans, Louisiana CORPORATE BACKGROUND is a non-profit, academic, multi-specialty,
PPD LABORATORIES CENTRAL LAB: SUPERIOR SERVICE, QUALITY DATA WITHOUT COMPROMISES
 PPD LABORATORIES CENTRAL LAB: SUPERIOR SERVICE, QUALITY DATA WITHOUT COMPROMISES PPD Laboratories provides world-class scientific expertise with state-of-the-art technologies supported by a commitment
PPD LABORATORIES CENTRAL LAB: SUPERIOR SERVICE, QUALITY DATA WITHOUT COMPROMISES PPD Laboratories provides world-class scientific expertise with state-of-the-art technologies supported by a commitment
Office of Clinical Research and the. Institute (CTSI) Patricia Emmanuel, MD
 Office of Clinical Research and the Clinical and Translational Research Institute (CTSI) Patricia Emmanuel, MD Office of Clinical Research- Priorities Review processes for clinical study implementation,
Office of Clinical Research and the Clinical and Translational Research Institute (CTSI) Patricia Emmanuel, MD Office of Clinical Research- Priorities Review processes for clinical study implementation,
Regulatory Issues in Genetic Testing and Targeted Drug Development
 Regulatory Issues in Genetic Testing and Targeted Drug Development Janet Woodcock, M.D. Deputy Commissioner for Operations Food and Drug Administration October 12, 2006 Genetic and Genomic Tests are Types
Regulatory Issues in Genetic Testing and Targeted Drug Development Janet Woodcock, M.D. Deputy Commissioner for Operations Food and Drug Administration October 12, 2006 Genetic and Genomic Tests are Types
How To Use Centricity Emr
 Centricity Electronic Medical Record Complete information, one location Turn information into insight Imagine being able to raise the bar on patient care safety and quality. And find the information you
Centricity Electronic Medical Record Complete information, one location Turn information into insight Imagine being able to raise the bar on patient care safety and quality. And find the information you
Annex 7 Guidelines for the preparation of a contract research organization master file
 World Health Organization WHO Technical Report Series, No. 957, 2010 Annex 7 Guidelines for the preparation of a contract research organization master file Background 1. General information 2. Quality
World Health Organization WHO Technical Report Series, No. 957, 2010 Annex 7 Guidelines for the preparation of a contract research organization master file Background 1. General information 2. Quality
American College of Medical Genetics and Genomics Strategic Plan
 American College of Medical Genetics and Genomics Strategic Plan The American College of Medical Genetics and Genomics (ACMG) is the specialty society for diplomates certified by the American Board of
American College of Medical Genetics and Genomics Strategic Plan The American College of Medical Genetics and Genomics (ACMG) is the specialty society for diplomates certified by the American Board of
Sharon H. Johnson, BS, MS 123 Main Street Capital City, VA 00000 Phone: 434-555-1234 Email: [email protected]
 SAMPLE CRA CV Sharon H. Johnson, BS, MS 123 Main Street Capital City, VA 00000 Phone: 434-555-1234 Email: [email protected] Education: Masters of Science, Healthcare Administration, Capital City University,
SAMPLE CRA CV Sharon H. Johnson, BS, MS 123 Main Street Capital City, VA 00000 Phone: 434-555-1234 Email: [email protected] Education: Masters of Science, Healthcare Administration, Capital City University,
August 2011. www.ppdi.com
 Innovative Technology Provides Seamless Data Integration Linking Clinical Trial Patient Data to Central Laboratory Data Via an Oracle -Based Exchange Platform August 2011 www.ppdi.com Introduction Drug
Innovative Technology Provides Seamless Data Integration Linking Clinical Trial Patient Data to Central Laboratory Data Via an Oracle -Based Exchange Platform August 2011 www.ppdi.com Introduction Drug
A Comparison of Two Commonly Used CRO Resourcing Models for SAS/ Statistical Programmers R. Mouly Satyavarapu, PharmaNet/ i3, Ann Arbor, MI
 PharmaSUG 2012 - Paper MS11 A Comparison of Two Commonly Used CRO Resourcing Models for SAS/ R. Mouly Satyavarapu, PharmaNet/ i3, Ann Arbor, MI ABSTRACT: Why do we have Contract Research Organizations
PharmaSUG 2012 - Paper MS11 A Comparison of Two Commonly Used CRO Resourcing Models for SAS/ R. Mouly Satyavarapu, PharmaNet/ i3, Ann Arbor, MI ABSTRACT: Why do we have Contract Research Organizations
An information platform that delivers clinical studies better, faster, safer and more cost effectively
 An information platform that delivers clinical studies better, faster, safer and more cost effectively Powering Process & Performance Proactively manage study start-up and execution Risk profile new sites
An information platform that delivers clinical studies better, faster, safer and more cost effectively Powering Process & Performance Proactively manage study start-up and execution Risk profile new sites
THE BIOTECH & PHARMACEUTICAL INDUSTRY
 THE BIOTECH & PHARMACEUTICAL INDUSTRY ESSENTIAL CAREERS INFORMATION CALUM LECKIE KATIE BISARO CAREERS CONSULTANTS What we will cover Sector overview Types of role Graduate recruitment trends and issues
THE BIOTECH & PHARMACEUTICAL INDUSTRY ESSENTIAL CAREERS INFORMATION CALUM LECKIE KATIE BISARO CAREERS CONSULTANTS What we will cover Sector overview Types of role Graduate recruitment trends and issues
Technology. Accenture Infrastructure Outsourcing Services
 Technology Accenture Infrastructure Outsourcing Services 2 Accenture Infrastructure Outsourcing Services enable organizations to create a more cost-effective and responsive IT infrastructure one that not
Technology Accenture Infrastructure Outsourcing Services 2 Accenture Infrastructure Outsourcing Services enable organizations to create a more cost-effective and responsive IT infrastructure one that not
Hollie Goddard Sr. IRB Coordinator McKesson Specialty Health
 Hollie Goddard Sr. IRB Coordinator McKesson Specialty Health We are responsible for acquiring, analyzing, and protecting medical information vital to providing quality patient care HIM professionals ensure
Hollie Goddard Sr. IRB Coordinator McKesson Specialty Health We are responsible for acquiring, analyzing, and protecting medical information vital to providing quality patient care HIM professionals ensure
Global healthcare solutions
 Global healthcare solutions clinical trials pharma/biotech medical technology & supplies Global healthcare solutions for your peace-of-mind A global network with specialist options Global presence with
Global healthcare solutions clinical trials pharma/biotech medical technology & supplies Global healthcare solutions for your peace-of-mind A global network with specialist options Global presence with
To Certify or Not to Certify
 To Certify or Not to Certify Sandra Halvorson, BA, CCRP Clinical Research Coordinator II CIBMTR Minneapolis Campus Sue Logan, BS, CCRP Clinical Research Coordinator II CIBMTR Minneapolis Campus November
To Certify or Not to Certify Sandra Halvorson, BA, CCRP Clinical Research Coordinator II CIBMTR Minneapolis Campus Sue Logan, BS, CCRP Clinical Research Coordinator II CIBMTR Minneapolis Campus November
To Certify or Not to Certify Sandra Halvorson, BA, CCRP
 To Certify or Not to Certify Sandra Halvorson, BA, CCRP Clinical Research Coordinator II CIBMTR Minneapolis Campus Sue Logan, BS, CCRP Clinical Research Coordinator II We have no financial relationships
To Certify or Not to Certify Sandra Halvorson, BA, CCRP Clinical Research Coordinator II CIBMTR Minneapolis Campus Sue Logan, BS, CCRP Clinical Research Coordinator II We have no financial relationships
2015 Initiatives for Proactive Quality Management. Progress through Collaboration
 2015 Initiatives for Proactive Quality Management Progress through Collaboration 1 2015 AQC Members 2 The Avoca Quality Consortium Accelerating the development of leading practices and industry standards
2015 Initiatives for Proactive Quality Management Progress through Collaboration 1 2015 AQC Members 2 The Avoca Quality Consortium Accelerating the development of leading practices and industry standards
ROLE DESCRIPTIONS BY COMPETENCY LEVEL
 Description of Work: Positions in this class provide patient evaluation and care in area of assignment. Duties include but are not limited to development and presentation of educational programs, assessment,
Description of Work: Positions in this class provide patient evaluation and care in area of assignment. Duties include but are not limited to development and presentation of educational programs, assessment,
Health Care Job Information Sheet #20. Clinical Research
 Health Care Job Information Sheet #20 Clinical Research A. Background B. Occupations 1) Clinical Research Associate (Study Monitor) 2) Clinical Research Coordinator 3) Other positions in the field C. Labour
Health Care Job Information Sheet #20 Clinical Research A. Background B. Occupations 1) Clinical Research Associate (Study Monitor) 2) Clinical Research Coordinator 3) Other positions in the field C. Labour
A Greater Understanding. A Practical Guide to Clinical Trials
 A Greater Understanding A Practical Guide to Clinical Trials Solving Insurance and Healthcare AccessProblems I since 1996 A Greater Understanding Patient Advocate Foundation MISSION STATEMENT Patient Advocate
A Greater Understanding A Practical Guide to Clinical Trials Solving Insurance and Healthcare AccessProblems I since 1996 A Greater Understanding Patient Advocate Foundation MISSION STATEMENT Patient Advocate
Technology and Expertise Add Operational Value to Medical Device Trials
 Technology and Expertise Add Operational Value to Medical Device Trials Copyright 2015 Medidata Solutions. Medidata Solutions and other trademarks reserved in the US and globally. Medidata and other marks
Technology and Expertise Add Operational Value to Medical Device Trials Copyright 2015 Medidata Solutions. Medidata Solutions and other trademarks reserved in the US and globally. Medidata and other marks
Role of IRB/IEC in GCP. Benjamin Kuo, MD, Dr.PH, CIP.
 Role of IRB/IEC in GCP Benjamin Kuo, MD, Dr.PH, CIP. Institutional Review Board (IRB) An independent body constituted of medical, scientific and non scientific members Responsible for ensuring protection
Role of IRB/IEC in GCP Benjamin Kuo, MD, Dr.PH, CIP. Institutional Review Board (IRB) An independent body constituted of medical, scientific and non scientific members Responsible for ensuring protection
GOVERNANCE AND THE EHR4CR INSTITUTE
 GOVERNANCE AND THE EHR4CR INSTITUTE Dipak Kalra EuroRec, University College London Christian Ohmann, European Clinical Research Infrastructure Network (ECRIN) Electronic Health Records for Clinical Research
GOVERNANCE AND THE EHR4CR INSTITUTE Dipak Kalra EuroRec, University College London Christian Ohmann, European Clinical Research Infrastructure Network (ECRIN) Electronic Health Records for Clinical Research
The NIH Roadmap: Re-Engineering the Clinical Research Enterprise
 NIH BACKGROUNDER National Institutes of Health The NIH Roadmap: Re-Engineering the Clinical Research Enterprise Clinical research is the linchpin of the nation s biomedical research enterprise. Before
NIH BACKGROUNDER National Institutes of Health The NIH Roadmap: Re-Engineering the Clinical Research Enterprise Clinical research is the linchpin of the nation s biomedical research enterprise. Before
SERVICE PLANS. More Productivity. Less Worry. SERVICE PLANS
 SERVICE PLANS More Productivity. Less Worry. SERVICE PLANS Flexible service plans to help you maximize the value of your technology investment. Let us help you improve your laboratory productivity and
SERVICE PLANS More Productivity. Less Worry. SERVICE PLANS Flexible service plans to help you maximize the value of your technology investment. Let us help you improve your laboratory productivity and
Date : Date of start of procedure: Authorisation/ positive opinion :
 Substantial Amendment Notification Form (Cf. Section 3.7.b of the Detailed guidance on the request to the competent authorities for authorisation of a clinical trial on a medicinal product for human use,
Substantial Amendment Notification Form (Cf. Section 3.7.b of the Detailed guidance on the request to the competent authorities for authorisation of a clinical trial on a medicinal product for human use,
CDISC standards and data management The essential elements for Advanced Review with Electronic Data
 Session 6: Toward Electronic Submission of Study Data for New Drug Applications CDISC standards and data management The essential elements for Advanced Review with Electronic Data Yuki Ando Senior Scientist
Session 6: Toward Electronic Submission of Study Data for New Drug Applications CDISC standards and data management The essential elements for Advanced Review with Electronic Data Yuki Ando Senior Scientist
RECOMMENDATION ON THE CONTENT OF THE TRIAL MASTER FILE AND ARCHIVING
 RECOMMENDATION ON THE CONTENT OF THE TRIAL MASTER FILE AND ARCHIVING July 2006 TABLE OF CONTENTS Page 1. Introduction 2 2. Scope 2 3. Documents to be archived 2 4. Quality of essential documents 10 5.
RECOMMENDATION ON THE CONTENT OF THE TRIAL MASTER FILE AND ARCHIVING July 2006 TABLE OF CONTENTS Page 1. Introduction 2 2. Scope 2 3. Documents to be archived 2 4. Quality of essential documents 10 5.
Clinical Training Management
 Clinical Training Management Learning and Compliance for Clinical Research Helping to Fuel the Growth of CROs and Service Providers According to clinical researcher CenterWatch, the FDA and global regulatory
Clinical Training Management Learning and Compliance for Clinical Research Helping to Fuel the Growth of CROs and Service Providers According to clinical researcher CenterWatch, the FDA and global regulatory
Mayo School of Health Sciences. Surgical Technology (Affiliated Training Rotation) Rochester, Minnesota. www.mayo.edu
 Mayo School of Health Sciences Surgical Technology (Affiliated Training Rotation) Rochester, Minnesota www.mayo.edu Surgical Technology (Affiliated Training Rotation) PROGRAM DESCRIPTION Students enrolled
Mayo School of Health Sciences Surgical Technology (Affiliated Training Rotation) Rochester, Minnesota www.mayo.edu Surgical Technology (Affiliated Training Rotation) PROGRAM DESCRIPTION Students enrolled
TALENT MANAGEMENT SOLUTIONS TO THE CRA SHORTAGE: Investing in a Global Talent Pool
 TALENT MANAGEMENT SOLUTIONS TO THE CRA SHORTAGE: Investing in a Global Talent Pool inventivhealth.com/clinical Over the past few years, the field of clinical research has been hampered by a shortage of
TALENT MANAGEMENT SOLUTIONS TO THE CRA SHORTAGE: Investing in a Global Talent Pool inventivhealth.com/clinical Over the past few years, the field of clinical research has been hampered by a shortage of
Speed to Market Abbott Nutrition Experience in Streamlining the Pediatric Clinical Research Process
 Speed to Market Abbott Nutrition Experience in Streamlining the Pediatric Clinical Research Process Geraldine Baggs, Suzanne Monnin, Deborah Shye, Sue Zhang Abbott Nutrition Research & Development Abbott
Speed to Market Abbott Nutrition Experience in Streamlining the Pediatric Clinical Research Process Geraldine Baggs, Suzanne Monnin, Deborah Shye, Sue Zhang Abbott Nutrition Research & Development Abbott
Joint Position on the Disclosure of Clinical Trial Information via Clinical Trial Registries and Databases 1 Updated November 10, 2009
 Joint Position on the Disclosure of Clinical Trial Information via Clinical Trial Registries and Databases 1 Updated November 10, 2009 The innovative pharmaceutical industry 2 is committed to the transparency
Joint Position on the Disclosure of Clinical Trial Information via Clinical Trial Registries and Databases 1 Updated November 10, 2009 The innovative pharmaceutical industry 2 is committed to the transparency
Global Alzheimer s Platform. Luc Truyen, [email protected] George Vradenburg, [email protected] Debra Lappin, debra.lappin@faegrebd.
 Global Alzheimer s Platform 2015 Luc Truyen, [email protected] George Vradenburg, [email protected] Debra Lappin, [email protected] 1 Presentation 1. Today s Reality 2. Global Momentum 3. GAP
Global Alzheimer s Platform 2015 Luc Truyen, [email protected] George Vradenburg, [email protected] Debra Lappin, [email protected] 1 Presentation 1. Today s Reality 2. Global Momentum 3. GAP
Clinical Research Associate (CRA) - A Growing Career Path in Biotechnology / Pharmaceutical Industry
 Clinical Research Associate (CRA) - A Growing Career Path in Biotechnology / Pharmaceutical Industry By http://www.clinicalresearchassociate cra.com/studyguide/ A clinical research associate (CRA) is a
Clinical Research Associate (CRA) - A Growing Career Path in Biotechnology / Pharmaceutical Industry By http://www.clinicalresearchassociate cra.com/studyguide/ A clinical research associate (CRA) is a
RBM as Competitive Advantage for Mid-sized CROs
 RBM as Competitive Advantage for Mid-sized CROs Cyntegrity s White Paper Applying RBM analytics to improve patient safety and preventive care Table of Contents Introduction...3 What is Data-driven RBM?...4
RBM as Competitive Advantage for Mid-sized CROs Cyntegrity s White Paper Applying RBM analytics to improve patient safety and preventive care Table of Contents Introduction...3 What is Data-driven RBM?...4
Job Profile Clinical Research Associate III (CRA)
 PART 1 - PROFILE You are an experienced CRA with a strong background in Clinical Research who is passionate about drug development and are seeking a challenging and rewarding career in a quality focussed
PART 1 - PROFILE You are an experienced CRA with a strong background in Clinical Research who is passionate about drug development and are seeking a challenging and rewarding career in a quality focussed
CLINICAL RESEARCH GENERIC TASK DESCRIPTIONS
 Purpose Purpose Purpose Primary responsibility for implementation, coordination, evaluation, communication and/or management of research studies. May involve more than one study or multiple sites within
Purpose Purpose Purpose Primary responsibility for implementation, coordination, evaluation, communication and/or management of research studies. May involve more than one study or multiple sites within
EMA Clinical Laboratory Guidance - Key points and Clarifications PAUL STEWART
 EMA Clinical Laboratory Guidance - Key points and Clarifications PAUL STEWART Framework Labs generate data that are used to make decisions on the safety and efficacy of medicinal products; consequently,
EMA Clinical Laboratory Guidance - Key points and Clarifications PAUL STEWART Framework Labs generate data that are used to make decisions on the safety and efficacy of medicinal products; consequently,
PharmaSUG 2013 - Paper IB05
 PharmaSUG 2013 - Paper IB05 The Value of an Advanced Degree in Statistics as a Clinical Statistical SAS Programmer Mark Matthews, inventiv Health Clinical, Indianapolis, IN Ying (Evelyn) Guo, PAREXEL International,
PharmaSUG 2013 - Paper IB05 The Value of an Advanced Degree in Statistics as a Clinical Statistical SAS Programmer Mark Matthews, inventiv Health Clinical, Indianapolis, IN Ying (Evelyn) Guo, PAREXEL International,
Become a Dynamics Business
 Managed Services Become a Dynamics Business faster through the business value of Avanade Managed Services Executive Summary In an always-on digital world, enterprises are challenged to capitalize on new
Managed Services Become a Dynamics Business faster through the business value of Avanade Managed Services Executive Summary In an always-on digital world, enterprises are challenged to capitalize on new
Infoset builds software and services to advantage business operations and improve patient s life
 Infoset builds software and services to advantage business operations and improve patient s life Clinical Data Management ecrf & EDC Patient Support Programs Medication Adherence Mobile e-health Big Data
Infoset builds software and services to advantage business operations and improve patient s life Clinical Data Management ecrf & EDC Patient Support Programs Medication Adherence Mobile e-health Big Data
Reduce Trial Costs While Increasing Study Speed and Data Quality with Oracle Siebel CTMS Cloud Service
 Reduce Trial Costs While Increasing Study Speed and Data Quality with Oracle Siebel CTMS Cloud Service Comprehensive Enterprise Trial Management in the Cloud Oracle Siebel CTMS Cloud Service lets you effectively
Reduce Trial Costs While Increasing Study Speed and Data Quality with Oracle Siebel CTMS Cloud Service Comprehensive Enterprise Trial Management in the Cloud Oracle Siebel CTMS Cloud Service lets you effectively
ClinPlus. Clinical Trial Management System (CTMS) Technology Consulting Outsourcing. Expedite trial design and study set-up
 Technology Consulting Outsourcing ClinPlus Clinical Trial Management System (CTMS) Expedite trial design and study set-up Efficiently collect and manage patient and trial administration data Benefit from
Technology Consulting Outsourcing ClinPlus Clinical Trial Management System (CTMS) Expedite trial design and study set-up Efficiently collect and manage patient and trial administration data Benefit from
Operational aspects of a clinical trial
 Operational aspects of a clinical trial Carlo Tomino Pharm.D. Coordinator Pre-authorization Department Head of Research and Clinical Trial Italian Medicines Agency Mwanza (Tanzania), June 11, 2012 1 Declaration
Operational aspects of a clinical trial Carlo Tomino Pharm.D. Coordinator Pre-authorization Department Head of Research and Clinical Trial Italian Medicines Agency Mwanza (Tanzania), June 11, 2012 1 Declaration
Biotechnology and Life Science Marketing Services Mailing List and Data Card Order Form
 C H I Cambridge Healthtech Institute s Biotechnology and Life Science Marketing Services Mailing List and Data Card Order Form Over 800,000 names segmented by scientific interest Featuring U.S and International
C H I Cambridge Healthtech Institute s Biotechnology and Life Science Marketing Services Mailing List and Data Card Order Form Over 800,000 names segmented by scientific interest Featuring U.S and International
Data Center Infrastructure Management
 Data Center Infrastructure Management Helping IT Empower the Business Luis M Burgos, HP Services BDM Arrow, ECS Proactive Care Advanced Presented under Non-Disclosure A New Style of IT Driven by Four New
Data Center Infrastructure Management Helping IT Empower the Business Luis M Burgos, HP Services BDM Arrow, ECS Proactive Care Advanced Presented under Non-Disclosure A New Style of IT Driven by Four New
BOOSTING THE COMMERCIAL RETURNS FROM RESEARCH
 BOOSTING THE COMMERCIAL RETURNS FROM RESEARCH Submission in response to the Discussion Paper November 2014 Page 1 ABOUT RESEARCH AUSTRALIA is an alliance of 160 members and supporters advocating for health
BOOSTING THE COMMERCIAL RETURNS FROM RESEARCH Submission in response to the Discussion Paper November 2014 Page 1 ABOUT RESEARCH AUSTRALIA is an alliance of 160 members and supporters advocating for health
PharmaSUG 2016 Paper IB10
 ABSTRACT PharmaSUG 2016 Paper IB10 Moving from Data Collection to Data Visualization and Analytics: Leveraging CDISC SDTM Standards to Support Data Marts Steve Kirby, JD, MS, Chiltern, King of Prussia,
ABSTRACT PharmaSUG 2016 Paper IB10 Moving from Data Collection to Data Visualization and Analytics: Leveraging CDISC SDTM Standards to Support Data Marts Steve Kirby, JD, MS, Chiltern, King of Prussia,
COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) GUIDELINE ON DATA MONITORING COMMITTEES
 European Medicines Agency Pre-authorisation Evaluation of Medicines for Human Use London, 27 July 2005 Doc. Ref. EMEA/CHMP/EWP/5872/03 Corr COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) GUIDELINE
European Medicines Agency Pre-authorisation Evaluation of Medicines for Human Use London, 27 July 2005 Doc. Ref. EMEA/CHMP/EWP/5872/03 Corr COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) GUIDELINE
The Role of the CRO in Effective Risk-Based Monitoring
 New Whitepaper The Role of the CRO in Effective Risk-Based Monitoring The clinical trial industry is evolving. In an effort to improve participant safety and data integrity, regulators are encouraging
New Whitepaper The Role of the CRO in Effective Risk-Based Monitoring The clinical trial industry is evolving. In an effort to improve participant safety and data integrity, regulators are encouraging
Becoming Agile: a getting started guide for Agile project management in Marketing, Customer Service, HR and other business teams.
 Becoming Agile: a getting started guide for Agile project management in Marketing, Customer Service, HR and other business teams. Agile for Business www.agilefluent.com Summary The success of Agile project
Becoming Agile: a getting started guide for Agile project management in Marketing, Customer Service, HR and other business teams. Agile for Business www.agilefluent.com Summary The success of Agile project
Synergizing global best practices in the CRO industry
 Synergizing global best practices in the CRO industry KREARA - India Established on April 24 th 2004 Contract Research Services to Pharmaceutical companies Clinical Data management and Biometrics Interactive
Synergizing global best practices in the CRO industry KREARA - India Established on April 24 th 2004 Contract Research Services to Pharmaceutical companies Clinical Data management and Biometrics Interactive
Clinical Trial Management Software
 Clinical Trial Management Software For single-site and multi-site organizations, Maptrial is a cloud-hosted clinical trial management system (CTMS) designed to track the operational data of clinical trials
Clinical Trial Management Software For single-site and multi-site organizations, Maptrial is a cloud-hosted clinical trial management system (CTMS) designed to track the operational data of clinical trials
PROCEDURE FOR PREPARING GCP INSPECTIONS REQUESTED BY THE EMEA. GCP Inspectors Working Group
 European Medicines Agency Inspections London, 20 September 2007 EMEA/INS/GCP/197228/2005 Procedure no.: INS/GCP/2 PROCEDURE FOR PREPARING GCP INSPECTIONS REQUESTED BY THE EMEA GCP Inspectors Working Group
European Medicines Agency Inspections London, 20 September 2007 EMEA/INS/GCP/197228/2005 Procedure no.: INS/GCP/2 PROCEDURE FOR PREPARING GCP INSPECTIONS REQUESTED BY THE EMEA GCP Inspectors Working Group
Position Statement. Education and Certification for Nurse Life Care Planners. Status: Original Statement
 Position Statement Education and Certification for Nurse Life Care Planners Status: Original Statement Authored by: American Association of Nurse Life Care Planners (AANLCP *) and the Certified Nurse Life
Position Statement Education and Certification for Nurse Life Care Planners Status: Original Statement Authored by: American Association of Nurse Life Care Planners (AANLCP *) and the Certified Nurse Life
CRITERIA FOR ACCREDITING COMPUTING PROGRAMS
 CRITERIA FOR ACCREDITING COMPUTING PROGRAMS Effective for Evaluations During the 2009-2010 Accreditation Cycle Incorporates all changes approved by the ABET Board of Directors as of November 1, 2008 Computing
CRITERIA FOR ACCREDITING COMPUTING PROGRAMS Effective for Evaluations During the 2009-2010 Accreditation Cycle Incorporates all changes approved by the ABET Board of Directors as of November 1, 2008 Computing
MD/PhD MISSION OBJECTIVES GOALS
 MD/PhD MISSION OBJECTIVES GOALS Mission & Objectives Overarching Objective: Train physician-scientists to be an integral component of the healthcare system, now and into the future. Expectations for Graduates:
MD/PhD MISSION OBJECTIVES GOALS Mission & Objectives Overarching Objective: Train physician-scientists to be an integral component of the healthcare system, now and into the future. Expectations for Graduates:
Professional Standards and Guidelines
 College of Physicians and Surgeons of British Columbia Professional Standards and Guidelines Conflict of Interest Arising from Clinical Research Preamble This document is a guideline of the Board of the
College of Physicians and Surgeons of British Columbia Professional Standards and Guidelines Conflict of Interest Arising from Clinical Research Preamble This document is a guideline of the Board of the
Diagnostic Tests. Brad Spring Director, Regulatory Affairs
 Regulatory Challenges in the Development of Diagnostic Tests Industry Perspective Brad Spring Director, Regulatory Affairs September 28, 2011 Balancing Evidence of Safety & Effectiveness and Time to Market
Regulatory Challenges in the Development of Diagnostic Tests Industry Perspective Brad Spring Director, Regulatory Affairs September 28, 2011 Balancing Evidence of Safety & Effectiveness and Time to Market
How to Become a Cardiovascular Investigator. Careers in Electrophysiology Research Michael E. Cain, MD
 How to Become a Cardiovascular Investigator Careers in Electrophysiology Research Michael E. Cain, MD Background Reduce incidence cardiac arrhythmias is desirable Accurate identification of patients at
How to Become a Cardiovascular Investigator Careers in Electrophysiology Research Michael E. Cain, MD Background Reduce incidence cardiac arrhythmias is desirable Accurate identification of patients at
Application for Clinical Privileges Allied Health Professional Specialty: Family Nurse Practitioner (FNP)
 Application for Clinical Privileges Allied Health Professional Specialty: Family Nurse Practitioner (FNP) Qualifications To be eligible to apply for core clinical privileges as a Family Nurse Practitioner
Application for Clinical Privileges Allied Health Professional Specialty: Family Nurse Practitioner (FNP) Qualifications To be eligible to apply for core clinical privileges as a Family Nurse Practitioner
ORACLE SYSTEMS OPTIMIZATION SUPPORT
 ORACLE SYSTEMS OPTIMIZATION SUPPORT Organizations have unique business and IT challenges. With Oracle Systems Optimization Support, part of a flexible portfolio of services offered by Oracle Advanced Customer
ORACLE SYSTEMS OPTIMIZATION SUPPORT Organizations have unique business and IT challenges. With Oracle Systems Optimization Support, part of a flexible portfolio of services offered by Oracle Advanced Customer
EAPCI President-elect Application. M. Haude
 EAPCI President-elect Application M. Haude The European Association of Percutaneous Cardiovascular Interventions (EAPCI) Mission: To reduce the burden of cardiovascular disease in Europe through percutaneous
EAPCI President-elect Application M. Haude The European Association of Percutaneous Cardiovascular Interventions (EAPCI) Mission: To reduce the burden of cardiovascular disease in Europe through percutaneous
ECONOMIC FOOTPRINT OF CTs IN BELGIUM STRATEGIC PLAN TO PROMOTE CTs
 ECONOMIC FOOTPRINT OF CTs IN BELGIUM STRATEGIC PLAN TO PROMOTE CTs Ingrid Maes Director Pharma, Healthcare and Life Sciences PwC on behalf of: PwC on behalf of: PwC on behalf of: PwC PwC on behalf of:
ECONOMIC FOOTPRINT OF CTs IN BELGIUM STRATEGIC PLAN TO PROMOTE CTs Ingrid Maes Director Pharma, Healthcare and Life Sciences PwC on behalf of: PwC on behalf of: PwC on behalf of: PwC PwC on behalf of:
Best Practice Partnership Program
 Best Practice Partnership Program Empowering Community- Based Pathologists Keeping Medicine Local Losing outreach to centralized reference laboratories? Wondering how molecular testing & digital pathology
Best Practice Partnership Program Empowering Community- Based Pathologists Keeping Medicine Local Losing outreach to centralized reference laboratories? Wondering how molecular testing & digital pathology
GE Healthcare. MAC 2000 ECG Analysis System Streamlined for your hospital
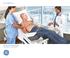 GE Healthcare MAC 2000 ECG Analysis System Streamlined for your hospital smart. MAC 2000 Complex hospital environments demand simple solutions. Equipment that simply works, every time. Technology that
GE Healthcare MAC 2000 ECG Analysis System Streamlined for your hospital smart. MAC 2000 Complex hospital environments demand simple solutions. Equipment that simply works, every time. Technology that
THE COMPLETE GUIDE TO PERFECT PROJECT MANAGEMENT
 THE COMPLETE GUIDE TO PERFECT PROJECT MANAGEMENT 1 CONTENTS Introduction 3 Prince2 5 Every as a project tool 6 Major development and building work 7 Further reading and information..9 2 THE COMPLETE GUIDE
THE COMPLETE GUIDE TO PERFECT PROJECT MANAGEMENT 1 CONTENTS Introduction 3 Prince2 5 Every as a project tool 6 Major development and building work 7 Further reading and information..9 2 THE COMPLETE GUIDE
Full-Service EDC as an Alternative to Outsourcing
 Full-Service EDC as an Alternative to Outsourcing The lifeblood of the entire drug development process is information. Of that information, some of the most challenging, expensive, and unpredictable information
Full-Service EDC as an Alternative to Outsourcing The lifeblood of the entire drug development process is information. Of that information, some of the most challenging, expensive, and unpredictable information
Make the Most of Big Data to Drive Innovation Through Reseach
 White Paper Make the Most of Big Data to Drive Innovation Through Reseach Bob Burwell, NetApp November 2012 WP-7172 Abstract Monumental data growth is a fact of life in research universities. The ability
White Paper Make the Most of Big Data to Drive Innovation Through Reseach Bob Burwell, NetApp November 2012 WP-7172 Abstract Monumental data growth is a fact of life in research universities. The ability
Introduction. The Evolution of the Data Management Role: The Clinical Data Liaison
 Introduction The CDL is a new role that will become a standard in the industry for companies that want to make more efficient use of limited resources: time and money. A CDL is key in that he or she conducts
Introduction The CDL is a new role that will become a standard in the industry for companies that want to make more efficient use of limited resources: time and money. A CDL is key in that he or she conducts
