Chapter 7 Complex Ocular Motor Disorders in Children
|
|
|
- Arleen Williamson
- 10 years ago
- Views:
Transcription
1 Chapter 7 Complex Ocular Motor Disorders in Children Introduction A number of complex ocular motility disorders are discussed in this chapter. The diversity of these conditions reflects the need for the ophthalmologist to maintain a broad working knowledge of pediatric neurologic disorders along with their ocular motor manifestations. Some clinical features of these conditions (e.g., congenital ocular motor apraxia, congenital fibrosis syndrome) are sufficiently unique that the diagnosis can be established solely on the basis of the clinical appearance. Other disorders either show overlapping manifestations or effectively masquerade as other entities. Unique features of some conditions, such as conjugate ocular torsion in patients with skew deviation, have been recently recognized and are considered worthy of emphasis because they significantly expand the differential diagnosis. Indeed, assessment of objective torsion (and subjective torsion when possible) is a necessary component of any comprehensive strabismological evaluation. 323 Correction of coexisting torsion can be integrated into the surgical plan, except when the torsion serves a compensatory function, as in patients with skew deviation. Although the clinical history and physical examinations remain the gold standard for establishing the diagnosis, neuroimaging has become an integral part of the diagnostic evaluation. High-resolution neuroimaging can now depict the presence or absence of most ocular motor nerves and the size, position, and dynamic function of the extraocular muscles. In patients with craniosynostosis, for example, heterotopic extraocular muscles within the orbit can produce a motility disorder indistinguishable from innervational inferior oblique muscle overaction, 736 absence of the isolated extraocular muscles can produce complex ocular motility dysfunction, and retrodisplacement of the trochlea can produce a motility pattern indistinguishable from congenital superior oblique palsy. 580 Optimal surgical treatment depends on accurate recognition of the specific cause of a given patient s motility disturbance. 204 Thus, a detailed know ledge of clinical findings that characterize each condition is necessary to perform the diagnostic workup in an efficient, timely, and cost-effective manner. A knowledgeable clinician is less likely to embark on fishing expeditions. The emphasis of this chapter is on ocular motility disorders of neurologic origin and their differential diagnosis. The most current pathophysiologic concepts of the disorders are summarized. A section at the end of the chapter is devoted to a few common eyelid and pupillary abnormalities encountered in children. Some of these disorders, such as excessive blinking in children, commonly represent benign transient tics that receive very little attention in the ophthalmologic literature, but are not rare in clinical practice. These bear only superficial resemblance to the more chronic benign essential blepharospasm of adults although, rarely, childhood tics and adult blepharospasm show clustering in the same family, suggesting a possible link. Occasionally, underlying ocular surface abnormalities and seizure disorders may be uncovered in children with excessive blinking. Other disorders, such as hemifacial spasm, which is more common in adults, may be the harbingers of more serious central nervous system (CNS) disorders if encountered in very early childhood. Pediatric Horner syndrome is treated in some detail at the end of the chapter, owing to its potentially ominous association with certain neoplasms, especially neuroblastoma. The human immunodeficiency virus (HIV) infection has joined the ranks of other great mimickers (e.g., myasthenia gravis, syphilis), with an ever-expanding list of neuro-ophthalmologic manifestations. No part of the nervous system is spared. Even though it is not specifically covered in this chapter, HIV-related neurological disease should be included in the differential diagnosis of childhood ocular motor disorders of cortical, brain stem, 343 cerebellar, or peripheral nervous system origin. Strabismus in Children with Neurological Dysfunction Common neurologic disorders of children are frequently associated with strabismus. These include cerebral palsy, Down syndrome, myelomeningocele, and hydrocephalus. M.C. Brodsky, Pediatric Neuro-Ophthalmology, DOI / _7, Springer Science+Business Media, LLC
2 310 7 Complex Ocular Motor Disorders in Children The features of the associated strabismus are often indistinguishable from the varieties found in otherwise normal children, but sufficient differences exist in a distinct minority of neurologically affected children to warrant separate consideration. Children with neurologic disorders, who have horizontal strabismus, have a higher prevalence of constant exotropia and primary superior oblique muscle overaction than otherwise healthy strabismic children (Fig. 7.1). 334,341 Fig.7.1 (a) Bilateral superior oblique overaction in child with lumbar myelomeningocele. Note resemblance to alternating skew deviation. (b) 1MR imaging characteristically associated Chiari II malformation with pronounced peaking of tectum and herniation of cerebellar vermis into cervical spinal canal
3 Strabismus in Children with Neurological Dysfunction Cerebral palsy is perhaps one of the most common conditions seen in a pediatric neuro-ophthalmology practice. 32 The term cerebral palsy defines a group of chronic neurologic disorders resulting from damage to the immature brain. Most cases nowadays are due to prematurity. Care should be taken to distinguish the static nature of cerebral palsy from the inexorably progressive neurodegenerative disorders such as Pelizaeus Merzbacher disease. Cerebral palsy is characterized by the onset of neurologic deficits in the neonatal period and absence of progression. In addition to motor disorders, such as paralysis, weakness, and incoordination, children with cerebral palsy may display sensory deficits, as exemplified by the frequent findings of optic atrophy, deafness, and cortical visual impairment. Patients with cerebral palsy show ophthalmologic abnormalities with a frequency ranging from 50 to 90%. These include optic atrophy, amblyopia, refractive errors, visual field defects, congenital cataracts, corneal leukomas, retinal dysplasia, choroiditis, macular or iris colobomas, retinopathy of prematurity, ptosis, spastic eyelids, abnormal head postures, and ocular motility disorders. 395,740 The latter include concomitant strabismus, ocular motor nerve palsy, nystagmus, gaze palsy, and other supranuclear disturbances of ocular movements. 489 Children with cerebral palsy have a markedly increased incidence of strabismus. 489 The strabismic deviations are usually horizontal and nonparalytic. Associated vertical incomitance is often seen, with A-pattern strabismus being particularly common. In one series, % of strabismic children with cerebral palsy showed A pattern, and 46% showed V pattern. Variability of the magnitude and direction of the strabismus are commonly noted in cerebral palsy. 40 Some studies note a predominance of esotropia, 40,489 while others have found exotropia to be more common. Momentary fluctuation from esotropia to exotropia has been termed dyskinetic strabismus and is considered unique to cerebral palsy. 129 Dyskinetic strabismus is unrelated to accommodative effort or attention. In addition to the dyskinetic strabismus of cerebral palsy, the differential diagnosis of a variable strabismus shifting from exotropia to esotropia includes exotropia with a high AC/A ratio, as well as surgically overcorrected accommodative esotropia with high AC/A ratio, and exotropia with dissociated horizontal deviation. Patients with oculomotor palsy with cyclic spasm may also appear to switch from exotropia to esotropia during a spasm phase, but the associated features are unique enough to obviate diagnostic confusion. Generally, the subset of children with cerebral palsy who exhibit dyskinetic strabismus are poor candidates for surgical correction. Children with stable deviations respond favorably to strabismus surgery, although their overall outcome is not as good as in strabismic children without cerebral palsy. 198,364 Unlike children with infantile esotropia, some 311 esotropic children with cerebral palsy convert spontaneously to an exotropia over several years. For this reasons, current trends toward early surgery for infantile esotropia should not be loosely applied to these patients, and it is important to rule out occult neurologic disease when early strabismus surgery is contemplated in seemingly normal children. As children with cerebral palsy and esotropia seem to have a strong predilection for surgical overcorrection, 371 standard surgical doses for treating esotropia should be reduced. In our experience, however, it is not necessary to reduce doses for bilateral lateral rectus muscle recession in neurologic exotropia. As detailed in Chap. 1, children with cerebral palsy may have abnormalities of gaze affecting pursuit and saccadic movements and vestibulo-ocular reflex suppression by fixation. 417,618 The craniosynostosis syndromes often present with complex forms of horizontal and vertical strabismus that are both neurologic and anatomical in origin. 144,206,280,541,558,580,646 Although hypertelorism is classically associated with exotropia with a V pattern, esotropia can also be seen. 431 Good stereopsis is rare in the syndromic craniosynostosis and may sometimes be confined to certain positions of gaze. 576 Some patients develop esotropia following craniofacial surgery, which can disrupt stereopsis. 788 The frequent finding of overelevation of the adducting eye in children with bilateral craniosynostosis was previously attributed to recession of the frontal processes, creating a mechanical disadvantage for the superior oblique muscle. It now appears that excyclorotation of both orbits alters the horizontal rectus muscle positions, positioning the lateral rectus muscles lower than normal and the medial rectus muscles higher than normal (Fig. 7.2). 156 This orbital excyclorotation is associated with marked excyclorotation of the globes, as is evident on retinal examination. 736 Although the resulting motility pattern is indistinguishable from that produced by primary inferior oblique muscle overaction, inferior oblique weakening procedures produce negligible improvement. Rectus muscle transposition, 172 in addition to superior oblique muscle strengthening (Harado Ito procedure), is necessary to treat this condition. Other mechanisms for vertical deviations in the craniosynostosis include absent or anomalous superior rectus (or other) muscles. 517,576,710 Preoperative orbital imaging allows the surgeon to anticipate the multitude of orbital anatomical disturbances that contribute to strabismus in children with craniosynostosis. 580 Visuovestibular Disorders Some forms of esotropia and their associated ocular motility signs signify benignity. From a neuro-ophthalmological perspective, horizontal strabismus can be subdivided into the
4 312 7 Complex Ocular Motor Disorders in Children Fig. 7.2 Craniosynostosis. Left: Coronal orbital MR image in a girl with Crouzon s disease and bilateral inferior oblique muscle overaction, V-pattern, and fundus extorsion showing excyclorotated extraocular muscle positions. Inferior oblique recession produced no improvement. Right: Schematic diagram showing affect of extraocular muscle heterotopia on binocular rotations Table 7.1 Visuovestibular disorders Dissociated vertical divergence Infantile esotropia (dissociated esotonus) Latent nystagmus Primary Inferior Oblique Overaction common conditions that presumably arise from unequal visual input to the two eyes and the less common conditions associated with neurologic disease. The former conditions constitute the visuovestibular disorders (Table 7.1), which may present as acquired and thereby may simulate neurologic disease. As these conditions arise from unbalanced binocular visual input to the central vestibular system, they signify benignity, although it can be argued that they constitute a physiologic form of neurologic disease. Although they are more commonly associated with esotropia, patients with early-onset exotropia present with the same constellation of dissociated ocular motor signs discussed in the following lines. Brodsky and Fray 117 have theorized that infantile esotropia may itself arise from a dissociated deviation. This occurs when unequal visual input generates dissociated esotonus, which leads to the gradual development of infantile esotropia, and explains the preponderance of infantile esotropia over infantile exotropia in nonneurologic infants. 117 Visuovestibular eye movements produce binocular rotations that are evoked by unequal visual input to the two eyes. 110 These consist of latent nystagmus, dissociated vertical
5 Strabismus in Children with Neurological Dysfunction 313 divergence, and primary oblique muscle overaction. These preternatural ocular rotations recapitulate the primitive eye movements induced by light or optokinetic stimulation in lateral-eyed animals. They are mediated by subcortical pathways extending from the optic nerves to the contralateral vestibular nucleus and cerebellum. Although present to a small degree in normals, they develop almost exclusively when infantile strabismus precludes the development of normal binocular cortical connections. As these visual reflexes are corrective for a central vestibular imbalance, they are not associated with dizziness, oscillopsia, or other neurologic symptoms that characterize vestibular disease. They are evoked by physiologic stimuli rather than by neurologic lesions. In the clinic, mechanical occlusion of one eye and cortical suppression of one eye can both activate in the subcortical visuovestibular pathways that evoke this visuovestibular response. The resurgence of visuovestibular movements in humans with infantile strabismus shows that our eyes function not only as visual organs, but as balance organs. Because they recapitulate visual reflexes in lateral-eyed animals, the absence of early cortical binocular development is a permissive factor that allows them to develop, the retention of primitive subcortical reflexes is the proximate cause of these movements. 110 Infantile strabismus provides a natural experiment to uncover the primitive visual reflexes that lie buried within us. By disrupting the development of frontal binocular vision, infantile strabismus allows these reflexes to bubble to the surface. The resulting ocular motor intrusions correspond to an imbalance of binocular visual input in three planes of physical space. Latent nystagmus is characterized by conjugate horizontal nystagmus induced by monocular occlusion or cortical suppression. The fast phase (in both eyes) is directed toward the side of the fixating eye. Latent nystagmus corresponds to the optokinetic component of ocular rotation that is driven monocularly by nasal optic flow during a turning movement of the body in lateral-eyed animals. 123 When infantile esotropia disrupts the establishment of binocular visual connections, visual input from the fixating eye to the contralateral nucleus of the optic tract evokes a counterrotation of the eyes which corresponds to a turning movement of the body toward the object of regard. The clinical expression of this visual reflex is also evident in the monocular nasotemporal asymmetry to horizontal optokinetic stimulation that characterizes infantile strabismus. 123 Dissociated vertical divergence is characterized by an upward drift (sometimes accompanied by a dynamic extorsional movement) of a covered or cortically suppressed eye. 105 Dissociated vertical divergence corresponds to the dorsal light reflex that has been observed in fish and other lateral-eyed animals when unequal luminance to the two eyes evokes a body tilt or vertical divergence of the eyes toward the side with greater luminance. 105 In humans, dissociated vertical divergence is a visual balancing reflex that uses weighted binocular visual input to orient eye position to the perceived vertical. 105 The exaptation of a cycloversional movement into the human dorsal light reflex permits active modulation of perceived visual tilt when the eyes are frontally positioned. 105,108,109 Primary oblique muscle overaction consists of bilateral overelevation or overdepression of the adducting eye. Primary inferior oblique muscle overaction corresponds to a similar dorsal light reflex that is induced in fish when a forward or backward shift in overhead luminance evokes an ipsidirectional body pitch or torsional rotation of the eyes backward to reorient the body with respect to the light. 107 These binocular torsional rotations of the eyes constitute a physiologic form of primary oblique muscle overaction that can be induced by altering binocular visual input. In humans, a forward or backward rotation relative to overhead light sends excitatory innervation to each of the elevators or each of the depressors. Because the vestibular system segregates innervation to its target extraocular muscles, the vertical actions of the human oblique muscles can summate in adduction with those of the rectus muscles to produce the innervational overelevation of the adducting eye that defines primary inferior oblique muscle overaction. The fundamental association of primary oblique muscle overaction with loss of binocular vision in humans suggests that the brain registers loss of binocular visual input as forward rotation of the body. 107 Although primary superior oblique muscle overaction is considered as a sign of neurologic disease (see section on Skew Deviation ) in the United States and Europe, it is reported to accompany infantile esotropia more commonly than primary inferior oblique overaction in Argentina and South Africa, 139,140,167 suggesting that it may also arise from a visuovestibular imbalance. 107 Neurologic Esotropia A variety of prenuclear neurologic diseases are characterized by an esotropic deviation of the eyes 717 (Tables 7.2 and 7.3). Spielmann has suggested that organic spasm of the near reflex, (Parinaud syndrome with convergence-retraction nystagmus or convergence excess), and thalamic esotropia Table 7.2 Complex neurologic disorders predisposing to esotropia Acute comitant esotropia Congenital cranial dysinnervation syndromes Convergence-retraction nystagmus Exercise-induced diplopia Periventricular leukomalacia (with or without cerebral palsy) Spasm of the near reflex Thalamic esotropia
6 314 7 Complex Ocular Motor Disorders in Children Table 7.3 Exotropia as a sign of neurologic disease Congenital cranial dysinnervation disorders (congenital fibrosis syndrome) Constant exotropia (cortical visual insufficiency, congenital homonymous hemianopia) Convergence insufficiency Intermittent exotropia greater at near fixation than distance fixation fall into a group of neurological diseases characterized by convergence excess. 717 Many of these disorders have been detailed in their specific clinical context in other chapters. As many neurologic patients take medications with strong anticholinergic side effects, esotropia of neurologic origin must be distinguished from anticholinergic esotropia, a condition in which excessive accommodative effort leads to accommodative esotropia. Several recent cases of esotropia induced by systemic anticholinergic medications have recently been described. 23,478 Anticholinergic esotropia seems to be precipitated by increased convergence of the eyes due to the increased accommodative effort in patients with an inherent tendency to develop esotropia. Spasm of the Near Reflex Spasm of the near reflex is characterized by intermittent episodes of miosis, convergence, and accommodation. Patients exhibit variable esotropia and varying pupillary size. Unlike in other patients with intermittent esotropia, monocular refixation during alternate occlusion is often slow and inaccurate. The name spasm of the near reflex should not be used interchangeably with either accommodation spasm or convergence spasm, each of which may present separately as a distinct entity. 67 Patients with spasm of the near reflex may present with diplopia, blurred vision (especially for distant objects), fluctuating vision, or nonspecific ocular discomfort. Due to its rarity, affected children may be misdiagnosed as having childhood esotropia, and unilateral or bilateral sixth nerve palsy, and, less commonly, divergence insufficiency, horizontal gaze palsy, ocular motor apraxia, convergence retraction nystagmus, true myopia, or Tensilon-negative myasthenia gravis. 657 Careful attention to pupillary size, retinoscopy during episodes of spasm to confirm the fluctuating induced myopia, and the variable esotropia should help confirm the diagnosis. The disorder is most commonly considered to be functional in nature, possibly due to underlying emotional conflict, with some patients showing signs of hysteria, malingering, or personality disturbances. 51 However, cases associated with organic disorders are occasionally reported, including head trauma, stroke, pretectal lesions, neurosyphilis, labyrinthine dysfunction, diphenylhydantoin intoxication, Wernicke s encephalopathy, metabolic encephalopathy, Fisher s syndrome (two cases reported so far), Arnold Chiari malformation, and photosensitive epilepsy. 81,195,442,566,636,694 However, isolated spasm of accommodation can also follow neurologic injury. 151 Several patients with intracranial tumors and spasm of the near reflex have been reported. 178,195 It should be emphasized that most patients with spasm of the near reflex in association with other neurologic disease have other symptoms and signs that can be readily uncovered during a careful neurologic and neuro-ophthalmologic examination. Spasm of the near reflex in infants and children should be distinguished from nystagmus blockage syndrome and from convergence substitution. The latter is seen in patients with congenital or acquired horizontal gaze paralysis (e.g., Möbius sequence, multiple sclerosis), in which the patient substitutes a convergence movement for a lateral version movement when attempting to fixate an eccentric target. Spasm of the near reflex can manifest in patients with significant hyperopia who are unable to relax accommodation even when plus lenses are used. 299 In this context, spasm of the near reflex can mimic deteriorating accommodative esotropia. 259,661 We examined a 12-year-old boy who had undergone left medial rectus muscle resection (4 mm) for presumed convergence insufficiency 4 months before. Shortly after the surgery, he complained of fluctuating vision and varying ocular alignment ranging from orthotropia to 80 prism diopters of esotropia. He was noted to have episodic, pronounced spasm of the near reflex associated with severe spasms involving the eyelids and the facial musculature (Fig. 7.3). His neurological evaluation and MR imaging of the head were totally unremarkable otherwise. His symptoms continued unchanged during a follow-up period of 8 months. Patients with isolated spasm of the near reflex may improve with reassurance. Some patients require psychiatric counseling. Current ophthalmologic treatment of spasm of the near reflex involves administering cycloplegic eye drops and providing bifocal glasses for reading. Historically, some patients have shown improvement with miotic drops, placebo drops, benzodiazepines, special glasses with occlusion of the inner third of each lens, monocular occlusion, or narcosuggestion during an amobarbital sodium interview. In patients with underlying organic disease, spasm of the near reflex (like essential blepharospasm) may be a type of dystonia. Like other types of dystonia, some cases respond to botulinum injection. 525,585 In patients without organic disease, this problem usually resolves spontaneously over months to years. Exercise-Induced Diplopia Exercise-induced diplopia has rarely been reported in children. 681,763 This condition may represent a breakdown of an esophoria, 763 but warrants neuroimaging to rule out a midline cerebellar tumor. 681
7 Strabismus in Children with Neurological Dysfunction 315 Fig. 7.3 Spasm of near reflex. Twelve-year-old boy had undergone small left medial rectus muscle resection for presumed convergence insufficiency. Four months later, he was noted to have episodic, pronounced spasm of near reflex (large esotropia, miosis, induced myopia with blurred vision) associated with severe spasms involving eyelids and facial musculature (a). In between spasms, only small esotropia was variably present (b) Neurologic Exotropia The differential diagnosis of constant exotropia includes cortical visual insufficiency, craniofacial synostosis, and congenital fibrosis syndrome. Isolated infantile exotropia is a rare but well-recognized nonneurologic condition in which a large-angle exotropia is associated with the same constellation of visuovestibular disorders that accompany infantile esotropia. Some early-onset cases of intermittent exotropia also fall into this group. 384 Some cases of infantile exotropia are hereditary, showing an autosomal dominant inheritance pattern. 119 Nevertheless, the clinician must maintain a high index of suspicion for neurologic disease in the child with infantile exotropia. In our experience, constant exotropia is most commonly seen in the setting of cortical visual insufficiency secondary to hypoxic-ischemic encephalopathy. 118 Associated signs include horizontal conjugate gaze deviation with little or no nystagmus and mild or sectoral pallor of the optic discs. 118 Because children with congenital homonymous hemianopia may develop a constant exotropia that serves to expand the existing visual field, confrontation visual fields should be checked in this setting. Signs of craniofacial disease are usually apparent on initial examination. As detailed below, fixed downgaze, associated ptosis, sparing of pupillary function, and similar findings in other family members provide clinical clues to the diagnosis of congenital fibrosis syndrome. Convergence Insufficiency Convergence insufficiency is a common condition characterized by:(1) diplopia or blurring at near, (2) decreased convergence amplitudes, and (3) a recessed near point of accommodation. 401 It seems to afflict intelligent patients and is considered to be the one form of strabismus that is treatable with eye exercises. Convergence training has been shown to improve proximal and tonic convergence, but not accommodative or fusional convergence. 33 Convergence insufficiency is most commonly a primary condition presenting in young adults. In this setting, it is presumably caused by an inborn deficiency or acquired imbalance of vergence eye movements that has yet to be identified. 33 In this setting, patients may present with sleeping on reading or near work, tearing, heavy lids, uncomfortable eyes, asthenopic symptoms, double vision, or blepharitis (from frequent rubbing of the eyes). These patients perform their near work optimally immediately after awakening. Convergence insufficiency is also a prominent accompaniment of neurologic disease, having been reported in association with head trauma, 19,130,181,450,452,483,719 neurodegenerative disorders, 75,125,631 infarction, 390,593 thyroid ophthalmopathy, 126 myasthenia gravis, toxic agents, medications, 125,738 inflammation, 125,620 decompression sickness, 493 whiplash injuries, 134 and attention-deficit hyperactivity disorder (ADHD). 310 In the last mentioned condition, it is not always clear whether convergence insufficiency is the cause or effect, whether they are comorbid conditions, or whether medications used to treat the ADHD may contribute to the convergence insufficiency. Some believe that accommodative insufficiency is the primary source of symptoms in children diagnosed with convergence insufficiency, 520 although this issue continues to be debated. 680 Therefore, accommodative amplitudes and dynamic retinoscopy should be examined to rule out an underlying or associated accommodative paresis. 653 It is also important to check for undetected ocular torsion, which can secondarily impair convergence. The potential pharmacological role of any medications in impairing accommodation should always be considered. 130 Convergence insufficiency should not be confused with convergence paralysis, another neurologic disorder in which there is a constant exotropia at near. Although convergence exercises are the mainstay of treatment for convergence insufficiency, 613 treatment with base in prisms has also been
8 316 7 Complex Ocular Motor Disorders in Children reported to be effective. 721 In patients with convergence paralysis or convergence insufficiency that does not respond to these measures, bimedial resections 325,360,464,786 and superior transpositions of the medial rectus muscles (to induce a physiologic V pattern and allow for single binocular near vision in downgaze) (Buckley EG: verbal communication) have been used. Because convergence insufficiency represents a kinetic problem rather than a static deviation, strabismus surgery may not provide an optimal approach. The superimposition of convergence insufficiency on a baseline exodeviation produces an exodeviation that is greater at near, which is often a soft sign of neurologic disease. 616 Neurologic forms of exotropia are summarized in Table 7.3. Skew Deviation Skew deviation of the eyes is a prenuclear vertical strabismus that is usually associated with posterior fossa lesions, particularly those affecting the brainstem tegmentum or the cerebellum. 419 While causative lesions are usually structural, skew deviation does rarely result from elevated intracranial pressure due to idiopathic intracranial hypertension. 281 The misalignment may be comitant or incomitant, simulate a paresis of an extraocular muscle, and alternate with time (slowly alternating skew deviation) or on lateral gaze (alternating skew on lateral gaze). As discussed below, it is usually accompanied by binocular torsion and torticollis. Although skew deviation has been historically considered to be a nonlocalizing sign, simply indicating involvement of the posterior fossa without further reference to a specific region, our understanding of its value as a clinical localizing sign has been revised in recent years. While early studies reported the absence of torsion in patients with skew deviation, 94 this myth has now been dispelled. 92,93 The degree of ocular torsion may or may not be equal in the two eyes. Skew deviation usually presents in the more general context of the ocular tilt reaction. The ocular tilt reaction is caused by unilateral injury to the otolithic pathways, which causes the brain to perceive the visual world as tilted. 116 The ocular tilt reaction is a righting reflex that results from alteration in the otolithic and/or vertical semicircular canal pathways that occurs in patients with lesions of either peripheral or central vestibular system. The ocular tilt reaction rotates the eyes and head toward the tilted visual world to restore vertical orientation (Fig. 7.4). The result is an oculocephalic synkinesis consisting of vertical divergence of the eyes (skew deviation), head tilt in the direction of the lower eye, and ocular torsion in the direction of head tilt. 92 These compensatory roll rotations strive to realign the interpupillary axis, head, and torsional position of the eyes, respectively, to the orientation that the patient erroneously perceives as vertical. The ocular tilt Fig. 7.4 Ocular tilt reaction. Illustration depicts graviceptive pathways from the otoliths and vertical semicircular canals mediating the vestibular reactions in the roll plane. The projections from the otoliths and the vertical semicircular canals to the ocular motor nuclei (trochlear nucleus IV, oculomotor nucleus III, abducens nucleus VI), and the supranuclear centers of the interstitial nucleus of Cajal (INC), and the rostral interstitial nucleus of the medial longitudinal fasciculus (rimlf) are shown. They subserve vestibuloocular reflex (VOR) in three planes. The VOR is part of a more complex vestibular reaction that also involves vestibulospinal connections via the medial and lateral vestibulospinal tracts for head and body posture control. Note that graviceptive vestibular pathways for the roll plane cross at the pontine level. Ocular tilt reaction is depicted schematically on the right in relation to the level of the lesion (i.e., ipsiversive with peripheral and pontomedullary lesions, and a contraversive with pontomesencephalic lesions). In vestibular thalamus lesions, the tilts of the subjective visual vertical may be contraversive or ipsiversive. With permission from Brandt et al 95 reaction has been reported in patients with intra-axial brainstem lesions (midbrain tegmentum, dorso-lateral medulla oblongata), unilateral vestibular neurectomy, and labyrinthectomy (performed in patients with severe Meniere s disease or acoustic neuroma). 812 The skew deviation that accompanies the ocular tilt reaction may be subdivided into at least three distinct types, each with distinct clinical features, ocular torsion, and localizing significance. 93 Because all three components of the ocular tilt reaction are compensatory for a subjective visual tilt, the head tilt does not serve to promote vertical fusion as it does in superior oblique palsy. 116 Consequently, prismatic or surgical correction of the vertical deviation does not eliminate the associated head tilt. Patients with the ocular tilt reaction may be misdiagnosed as having a superior oblique muscle palsy, because the skew deviation may be incomitant, and both disorders can show a positive Bielschowsky Head Tilt test. 284
9 Strabismus in Children with Neurological Dysfunction The diagnosis of ocular tilt reaction should be considered in patients with suspected superior oblique muscle palsy who show intorsion of the higher eye. Donahue et al 217 described five patients with incomitant skew deviation that simulated superior oblique muscle palsy on the Bielschowsky Three Step test, and intorsion of the higher eye and extorsion of the lower eye. In the patient with superior oblique palsy, the finding of intorsion (rather than the expected extorsion) of the higher eye (on both Double Maddox rod testing and indirect ophthalmoscopy) should therefore signal the diagnosis of skew deviation. In other patients, skew deviation can simulate isolated inferior oblique palsy. 217 Recently, Pareluker et al found that the vertical and torsional deviations associated with skew deviation resolve in the reclined position, a finding that provides another useful clue to the diagnosis. 610 For reasons that are not understood, the utricles become functionally deafferented in the reclined position, 113 which explains the disappearance of skew deviation and the disappearance of compensatory head tilt in supine patients with superior oblique palsy 732 (once asymmetrical utricular output can no longer be accessed to neutralize the vertical deviation). The localizing value of skew deviation is now well established. 93,94 In a study of 56 adults with unilateral brainstem infarctions and skew deviations, clinical and neuroimaging 317 analysis revealed the following: (1) The ipsilateral eye was hypotropic with caudal pontomedullary lesions and higher with rostral pontomesencephalic lesions. (2) All patients with skew deviation showed simultaneous conjugate bilateral ocular torsion toward the hypotropic eye. Ocular torsion was evaluated from fundus photographs. This and other studies demonstrate that skew deviation is a sensitive brainstem sign of localizing and lateralizing value. 93,94 While the skew deviation in adults results primarily from infarction, skew deviation in children may be associated with a wide variety of conditions, including tumors, 158 Chiari malformation, 173 autoimmune disease, 632 paroxysmal hemiparesis of childhood, 214,357 and increased intracranial pressure. 52,281,530 In one case, skew deviation was diagnosed on prenatal MR images in a fetus with a glioblastoma involving the brainstem. 158 Incomitant skew deviation may mimic a primary overaction of an oblique extraocular muscle. One type of skew deviation, termed alternating skew on lateral gaze or bilateral abducting hypertropia, closely resembles bilateral superior oblique overaction (Fig. 7.5). 332,339,341,567 Affected patients typically display a right hypotropia on downgaze and to the left and left hypotropia on downgaze and to the right. One study of children with brainstem tumors 341 showed that alternating skew deviation on lateral gaze localizes to the lower Fig. 7.5 Alternating skew on lateral gaze: Patient presented with new onset of downbeat nystagmus and oscillopsia. He was diagnosed with spinocerebellar degeneration. He was orthotropic in primary position (b) but displayed left hypotropia on gaze down and to right (a) and right hypotropia on gaze down and to left (c). Note similarity to bilateral overaction of superior oblique muscles
10 318 7 Complex Ocular Motor Disorders in Children brainstem or cerebellum. Given the similar localization of neuroanatomic lesions in patients with myelomeningocele (most whom have Chiari II malformation) and the observation that these patients show a predilection for superior oblique overaction (Fig. 7.1), Hamed et al have argued that primary superior oblique overaction and skew deviation are phenomenologically indistinguishable. 334,339,341 This hypothesis is further supported by the observation that strabismic children who have superior oblique overaction show a higher frequency of associated neurologic disorders compared with a control population consisting of strabismic children without superior oblique overaction. 339 Superior oblique overaction is commonly seen in conjunction with more generalized neurological disease such as cerebral palsy, myelomeningocele, and hydrocephalus. Brandt and Dieterich 96 later suggested that overlapping pathways modulate roll and pitch function of the vestibuloocular reflex, making efficient use of the vestibular network. According to their hypothesis, a unilateral skew deviation reflects a central graviceptive imbalance in the roll plane, while bilateral paramedian lesions or bilateral dysfunction of the cerebellar flocculus produces a tone imbalance in the pitch plane. The principle behind this operation resembles the guidance system of airplanes, wherein unilateral activation of a brake flap causes the plane to roll, while bilateral activation results in downward pitch. In a bilateral ocular tilt reaction, the vertical components summate to produce the slow phase vertical drift of both eyes while the torsional components cancel each other out. Thus, a roll imbalance manifests as an ocular tilt reaction, while bilateral otolithic imbalance produces upbeat or downbeat nystagmus in conjunction with an alternating skew deviation on lateral gaze. 96 Zee 835 presented an interesting model in lateral-eyed animals to demonstrate how a pitch disturbance (forward or backward) would generate the patterns of vertical deviation in lateral gaze that are seen in humans with bilateral alternating skew deviation. Thus, while unilateral skew deviation corresponds to central graviceptive dysfunction of otolithic pathways in the roll (tilt) plane, lateral alternating skew deviation corresponds to a central graviceptive dysfunction of otolithic pathways in the pitch plane. 107 By viewing a pencil slanted forward or backward in the pitch plane, then closing one eye and then the other, the reader can see that each image appears tilted in opposite directions when viewed monocularly. 109 Guyton and Weingarten 323 hypothesized that primary oblique muscle overaction and A- and V-pattern strabismus are a result of sensory torsion that arises when fusion is absent. However, it now appears that true oblique muscle overaction, caused by premotor disinhibition in the presence of early binocular visual loss, is the cause, rather than the effect, of primary oblique muscle overaction. 107 While most primary oblique muscle overaction is innervational in origin, heterotopia of the extraocular muscles and their surrounding connective tissues has been recognized as an anatomical cause of apparent primary oblique muscle overaction. 169,203,204 Other conditions causing overdepression of the adducting eye (mimicking primary superior oblique muscle overaction) include inferior rectus muscle palsy, apparent overaction of the oblique muscles in exotropia, downshoot of the eye in Duane syndrome, physiologic overaction of the oblique muscles in eccentric gaze, and other restrictive or paretic conditions. 334 Overelevation or overdepression in adduction is therefore an ocular sign that may reflect a variety of conditions. It is now possible to conceptualize oblique muscle overaction as comprising a visuovestibular type (primary inferior oblique muscle overaction), a bilateral vestibular injury (primary superior oblique muscle overaction), a leash-effect type (Duane syndrome), and a muscle heterotopia type. Gaze Palsies, Gaze Deviations, and Ophthalmoplegia This section discusses conditions causing ophthalmoplegia (i.e., mixed vertical and horizontal ocular paresis), or gaze palsies (horizontal or vertical). Gaze palsy may reflect abnormalities of saccadic eye movements, as may result from frontal lobe or frontomesencephalic saccadic pathway damage, pursuit eye movements, as may result from occipito-parieto-mesencephalic damage, or both, as may result from damage to the brainstem gaze center. It is sometimes difficult to distinguish a horizontal gaze palsy from a unilateral oculomotor or abducens nerve paresis with a compensatory head turn. 63 An infant or toddler with an abducens nerve palsy may appear to have a gaze palsy when he or she adopts a compensatory head position to achieve binocularity and resists any gaze shift out of the zone of binocularity. In such cases, one must place a patch on the suspected paretic eye, which leads to resolution of the head turn in ocular motor nerve paresis, but not in gaze palsy. Complete resolution of such a head turn may occasionally require several days of patching. 63 Horizontal Gaze Palsy in Children The causes of horizontal gaze palsy in children are shown in Table 7.4. Generally speaking, inability to conjugately move the eyes horizontally to one side is caused by a lesion in the contralateral frontal eye field or the ipsilateral paramedian pontine reticular formation (PPRF) or abducens nucleus.
11 Gaze Palsies, Gaze Deviations, and Ophthalmoplegia Table 7.4 Causes of horizontal gaze palsy in children Brainstem arteriovenous malformation Bilateral Duane syndrome Congenital horizontal gaze palsy and scoliosis Congenital horizontal gaze paralysis and ear dysplasia Familial, congenital paralysis of horizontal gaze Leigh syndrome Möbius sequence Neuronopathic Gaucher disease Pontine glioma Wildervanck syndrome Such a lesion may be distinguishable with reflex maneuvers (e.g., caloric stimulation, Doll s head maneuver) that would drive the eyes into the paretic gaze if the PPRF and abducens nucleus are intact. Neuronopathic Gaucher disease with hepatosplenomegaly can be associated with isolated horizontal gaze palsy. Children with type 2 Gaucher disease may also show a fixed esotropia, while those with type 3 may be accompanied by head thrusts suggestive of COMA. 611 Causes of horizontal gaze palsy that are now classified as congenital cranial dysinnervation syndromes (such as congenital horizontal gaze palsy with scoliosis) are detailed later in this chapter. Congenital bilateral paralysis of horizontal gaze has been reported in association with facial paralysis, most likely representing a form of Möbius sequence (facial diplegia and sixth nerve palsy). It has also been described without facial paralysis. In the latter cases, the most typical findings are total absence of conjugate horizontal gaze, both volitionally and after stimulation of optokinetic and vestibular systems; preserved convergence with substitution of convergence movements for conjugate eye movements upon attempting horizontal lateral gaze; cross-fixation; and apparently normal vertical eye movements. 61 These cases, which show overlap with the Wildervanck syndrome, have occurred either as an isolated abnormality or in association with other findings that included kyphoscoliosis, facial contracture and myokymia, and the Klippel Feil syndrome (fusion of cervical and upper thoracic vertebrae) in one patient. 311 The precise cause of these cases is unknown, but the underlying defect has been speculated to represent selective maldevelopment affecting either the horizontal gaze center in the PPRF or the motor neurons and interneurons in the abducens nuclei. Yee et al 824 reviewed the evidence and concluded that a developmental anomaly affecting the abducens nucleus, but not the horizontal gaze center in the PPRF, is most consistent with the clinical findings in this syndrome. They further speculated that the involvement of the facial musculature associated with some cases may result from a similar developmental anomaly of the facial nucleus. Some cases of bilateral horizontal gaze paralysis are familial. 824 In children with pontine gliomas, involvement of the abducens nuclei and/or the PPRF may lead to horizontal gaze palsy (Fig. 7.6). Congenital Ocular Motor Apraxia 319 Apraxia (Greek: inaction) literally denotes the inability to perform volitional, purposeful motor activity despite the absence of paralysis. In the context of eye movements, the term apraxia should be limited to describe conditions in which volitional saccades are defective, but reflex and random movements are preserved (i.e., the quick phases of vestibular or optokinetic nystagmus), underscoring the presence of intact lower motor neuron pathways. This apraxic defect becomes more readily understandable by noting the presence of at least two major classes of cerebrally triggered saccades, namely, intrinsically triggered (volitional) saccades and extrinsically triggered (reflexive) saccades. Children with true saccadic apraxia show a defect in the first class of saccades, but not in the second. 695 COMA is characterized by the selective absence of horizontal saccades with preservation of vertical saccades. 174 COMA was so named because it was thought to fulfill the strict definition of apraxia outlined above, namely, the reflex and random eye movements as well as the fast phases of optokinetic nystagmus are preserved. However, there is no clear evidence for a cortical etiology and it is now recognized that the quick reflexive fast phases are also defective in these patients. 354 For these reasons, Harris et al 354 advocate use of the term intermittent saccadic failure. Ocular motor apraxia is divided into congenital and acquired varieties. The acquired form is usually encountered in adults, following bilateral basal ganglia or cerebral hemispheric lesions, 623 although some acquired cases have been reported in children. For example, two children developed isolated ocular motor apraxia following cardiac surgery. 829 In children, the idiopathic form of COMA must be differentiated from the forms seen in Gaucher disease, ataxia telangiectasia, and Leigh disease (discussed later). Acquired ocular motor apraxia seldom fulfills the strict definition of an apraxic disorder. 695 Most acquired cases more closely reflect a global saccadic dysfunction rather than an apraxic disorder and may be better designated as horizontal saccadic palsies or gaze palsies, depending on whether or not smooth pursuit is also affected. 354 The natural history is one of gradual resolution over several years (with persisting subclinical defects), which may reflect the normal maturation process or the development of blink-induced saccades. 354 A prominent feature of this syndrome is large-amplitude horizontal head thrusting to achieve visual fixation. 268 However, head thrusting need not always be present. The quick phases of induced vestibular nystagmus and optokinetic nystagmus are intermittently or completely absent, causing the eyes to deviate and lock up at the mechanical limit of gaze at the end of the slow phase. 354 The classical explanation of the head thrusts is that they are compensatory,
12 320 7 Complex Ocular Motor Disorders in Children Fig. 7.6 Horizontal gaze palsy. Child with pontine glioma shows paralysis of (a) right gaze and (b) left gaze but intact (c) upgaze and (d) downgaze. (e) MR imaging shows diffuse enlargement of pons from glioma which explains why the head is thrust in the direction of an eccentric target, and the eyes rotate conjugately in the opposite direction under the influence of the resulting vestibuloocular reflex. The head excursion must then overshoot the intended target to allow the controversively rotated eyes to fixate the desired target. Once fixation is achieved, the head slowly reassumes its neutral position to allow a direct, straight gaze. The child is usually noted to blink at the onset of head thrusts. The concept that head thrusts are always compensatory has been challenged by evidence suggesting that a thrustsaccade synkinesis is the explanation for head thrusts; in that, the thrusts may actually facilitate the initiation of saccades in a subgroup of patients. 837 Eustace et al 255 questioned whether, rather than being compensatory, the head thrusts in COMA may be a defect that reflects an inability to unlock or cancel the effects of the vestibulo-ocular reflex. Interestingly, some children with COMA display occasional head thrusts
13 Gaze Palsies, Gaze Deviations, and Ophthalmoplegia or a low-amplitude horizontal head nodding, even when fixating a stable target. The head thrusts so characteristic of COMA appear when the baby acquires head and neck control (usually at 6 months). Therefore, although congenital, the disorder is rarely diagnosed until late infancy. At an earlier age, blindness may be suspected due to failure to follow objects. This failure to visually pursue objects is understandable given the defective saccadic system, because infants follow with a series of hypometric saccades. In this context, failure to pursue may be misconstrued as a visual deficit. Evaluation of the vestibuloocular response in such infants by spinning them elicits a slow but not a fast component, which helps establish the diagnosis. Although isolated COMA is usually sporadic, familial cases have been documented. 84,175,293,320,628,775 Phillips et al 615 described COMA in four children of an affected father, indicating probable autosomal dominant transmission. COMA has been associated with a wide variety of conditions ranging from the relatively benign to the rapidly progressive neurometabolic degenerations. COMA may be congenital or acquired. 519 It may also be idiopathic or associated with structural brain abnormalities (e.g., cerebellar hypoplasia, agenesis of the corpus callosum), neurodegenerative disorders (e.g., ataxia telangectasia, Gaucher disease), perinatal problems (e.g., hypoxia, cerebral palsy, hydrocephalus), or acquired disease (e.g., herpes encephalitis, posterior fossa tumors, ischemia) (Table 7.5). 354,519 These disorders should be suspected when vertical saccades are affected or when saccades are slow. 354 While affected children are often judged to be otherwise healthy, most have associated deficits in other motor spheres, resulting in problems in oral-motor planning affecting speech output (speech apraxia), as well as truncal ataxia, hypotonia, developmental delay, or perceptual visual-spatial difficulties. 312,396,519,635,674,725,726 In a recent study, of 14 patients with no structural abnormality on neuroimaging showed abnormal neurodevelopment, although it has been stated that speech delay in ocular motor apraxia is expressive and due to verbal apraxia. 396,635 These included delayed speech and reading, and abnormalities of language development involving both receptive (comprehension) and expressive (language) Table 7.5 Neurometabolic causes of congenital ocular motor apraxia (Adapted from Harris et al 354 ) Ataxia telangectasia Gaucher disease Joubert syndrome Krabbe disease Niemann Pick syndrome Cockayne syndrome Pelizeus Merzbacher disease Recessive ataxia with ocular apraxia Spinocerebellar ataxia type development. One mother of a 3-year-old girl stated, Her speech has been good in terms of word acquisition, but she has an odd linking of phrases lots of backwards phrases. These studies indicate that COMA is the salient feature of a more generalized neurological disorder, 312 and that isolated cases are at risk for mild-to-moderate educational difficulties. The association of COMA with CNS lesions has long been recognized. 232,268,599 Neuroimaging often discloses vermian hypoplasia and dilatation of the fourth ventricle at the upper brainstem level. 448 Sargent et al 674 found hypoplasia of the cerebellar vermis in approximately 50% of patients with COMA, with preferential involvement of the inferior portion. Additional involvement of the superior portion correlated with more severe feeding difficulties, speech apraxia, or more severe generalized motor problems. Kim et al 437 reported COMA with spasmus nutans in a child with cerebellar vermian hypoplasia. They speculated that hypoplasia of the vermal cortex could disinhibit the caudal fastigial nucleus, leading to an abnormally increased activity in the fixation region of the superior colliculus and results in the inhibition of saccadic generation. On the other hand, Harris et al found vermian hypoplasia in only one of two siblings with COMA, suggesting that this defect may not be causative, but a marker of some other underlying pathology. 369 Shawkat et al found abnormal scans in 61% of patients with saccadic initiation failure. 697 Most abnormalities involved the brainstem and cerebellar vermis, although other abnormalities of the cerebral cortex and basal ganglia were also detected. Other neuroimaging abnormalities have been reported less frequently, including porencephalic cyst, agenesis or hypoplasia of the corpus callosum, 84 posterior fossa tumors such as medulloblastoma or lipoma, and gray matter heterotopias. 27,505,725,731,833 Recently, a subgroup of autosomal recessive cerebellar ataxias was identified. 473 It includes four distinct subtypes: ataxia-telangectasia, ataxia-telangectasia-like disorder, and ataxia with ocular motor ataxia types 1 and 2. The phenotypes share similarities, and the responsible genes ATM, MRE11, APTX, and SETX, respectively, are all implicated in DNA break repair. As in many other DNA repair deficiencies, neurodegeneration is a hallmark of these diseases. 473 The first two conditions are detailed in Chap. 10. The third, ataxia with ocular motor apraxia type 1 (AOA type 1) is a recently described autosomal-recessive condition of childhood onset that is caused by mutations in the APTX gene, which encodes the protein aprataxin. 826 Onset is usually 2 10 years (mean, 6.8 years), with gait and limb ataxia, dysarthria, ocular motor apraxia, mild peripheral neuropathy, and progression of neurological deficits. 473,481 MR imaging may show cortical and cerebellar atrophy that is most prominent in the mid-vermis. 267 Aprataxin mutations are associated with low coenzyme Q10 levels in muscle, which is not associated with duration, severity, or progression of the disease. 473
14 322 7 Complex Ocular Motor Disorders in Children Ataxia with ocular motor apraxia type 2 (AOA type 2) is an autosomal recessive disorder associated with mutations in the Senataxin (SETX) gene. 189,506,559 The age of onset is later (11 20 years), with sensory motor neuropathy, primary ovarian failure, chorea, and ocular motor apraxia in about half of affected patients. An elevated level of serum alpha-fetoprotein is a consistent finding. 481 Ocular motor apraxia can also be a clinical manifestation of other well-known neurologic or systemic diseases. The association of COMA and a molar tooth sign should suggest the diagnosis of Joubert syndrome and Related Disorders (JRSD). First described in 1969, Joubert syndrome is characterized by the variable combination of episodic neonatal tachpynea and apnea, rhythmic protrusion of the tongue, ataxia, hypotonia, and a variable degree of psychomotor retardation. 405 The episodic tachpynea presents in the neonatal period and alternates with periods of apnea, resembling the panting of a dog, and usually resolves or improves over time. Typical facial features include high rounded eyebrows, broad nasal bridge, anteverted nostrils, and low-set ears. 89,513,609 The associated congenital retinal dystrophy was at first labeled as a variant of Leber congenital amaurosis but subsequently considered different because the visual loss is not as profound (20/60 to 20/200) in JRSD, compared with counting fingers or worse in Leber congenital amaurosis. In addition, the visual evoked potentials (VEPs) are relatively spared (mild-to-moderate reduction in amplitudes, compared with absent or highly attenuated signals). Both conditions show flat or highly attenuated electroretinograms (ERGs). 516 Other ophthalmologic findings include ptosis, congenital ocular fibrosis, and colobomas. 468 Systemic manifestations have been infrequently reported, including meningoencephalocele, microcephaly, polydactyly, kidney abnormalities, soft tissue tumors of the tongue, liver disease, and duodenal atresia. 513 Ocular motor disorders described in JSRD include slow, hypometric saccades, ocular motor apraxia, strabismus, periodic alternating gaze deviation, pendular torsional nystagmus, seesaw nystagmus, skew deviation, and defective smooth pursuit, as well as optokinetic and vestibular responses. 468,515,797a The ocular motor apraxia in JRSD differs from COMA in that both volitional saccades and quick phases of nystagmus are impaired in both the horizontal and vertical directions, and the problem does not resolve with time. 757 The most common ocular motor abnormalities are saccadic dysfunction with head thrusts and primary position nystagmus (typically, seesaw nystagmus). 432 Joubert syndrome is characterized by hypoplasia of the cerebellar vermis and a particular midbrain hindbrain molar tooth sign, a finding shared by a group of Joubert syndrome-related disorders with a wide phenotypic variability. 432 The molar tooth sign is a complex malformation of the hindbrain midbrain junction (brainstem isthmus) characterized by cerebellar vermis hypoplasia, thick and Fig. 7.7 Axial T1-weighted MR image in patient with Joubert disease showing molar tooth sign maloriented superior cerebellar peduncles, and an abnormally deep interpeduncular fossa. As first described by Maria, axial MR imaging at the pontomesencephalic level has shown that this malformation produces a peculiar appearance resembling a molar tooth (Fig. 7.7). 456,515,516,651 The pathological findings include vermian hypoplasia or dysplasia, elongation of the caudal midbrain tegmentum, and marked dysplasia of the caudal medulla. The molar tooth sign (and the absence of associated hydrocephalus) distinguishes Joubert syndrome from the vermian agenesis that occurs with the Dandy Walker variant, but 10% of patients with a Dandy Walker-like cyst have the molar tooth sign and are clinically similar to classic Joubert syndrome patients. Such patients were referred to as Dandy Walker Plus by Maria et al 514 Importantly, the molar tooth sign is not seen in isolated vermian hypoplasia because the cerebellar peduncles and interpeduncular fossa can be normal in several vermian hypoplasia syndromes. The deep interpeduncular fossa and decreased anteroposterior diameter of the brainstem isthmus in the molar tooth sign can best be explained by nondecussation of the ascending superior cerebellar peduncles. The coincident finding of asymmetrical VEPs in some patients with seesaw nystagmus suggests failure of chiasmal decussation. 432 This finding, together with the absence of decussation in both the superior cerebellar peduncles and the corticospinal tracts at the medullary pyramids,
15 Gaze Palsies, Gaze Deviations, and Ophthalmoplegia suggests that patients with Joubert syndrome have a defect of axon guidance in the motor circuits. 265,432,626,715 Although the molar tooth sign is still considered essential to the diagnosis of Joubert syndrome, there are a number of Joubert syndrome-related disorders such as COACH, Arima, Debakan, and Senior-Løken that are much more rare and which may also show a molar tooth sign on MR imaging. In these conditions, there are numerous other abnormalities in addition to the neurological features of the Joubert syndrome; involvement of other organs such as eye, kidney, and liver, have been described. 673,675 The Arima syndrome exhibits pigmentary degeneration suggestive of Leber congenital amaurosis, severe psychomotor retardation, hypotonia, characteristic facies, polycystic kidneys, and absent cerebellar vermis. Joubert and Arima syndromes may be distinguished by such clinical features as neonatal tachypnea, which is one of the cardinal features of Joubert syndrome. The identification of mutations in seven different cilium genes in up to 20% of patients with Joubert syndrome suggests that a certain percentage of Joubert syndrome is a celiopathy that can affect various organ systems, including the connecting cilium of photoreceptors. 432,609 Although patients with the NPHP1 deletion often have juvenile nephronophthisis and rarely have retinal dystrophy, patients with AH1 mutations seem less likely to develop retinal dystrophy. 609,767 Mutations in the AHI1 gene can also be associated with other CNS malformations (in non-joubert patients) such as corpus callosal abnormalities, polymicrogyria, hydrocephalus, and encephalomeningocele. 292 Patients with the CEP290 mutation seem to have both nephronophthisis and retinal dystrophy. 91,679,772 Other phenotypes have included nonspecific renal cortical cysts, ocular colobomas, and encephaloceles. 91,432,679,772 Joubert syndrome may be sporadic or familial, with familial cases inherited in an autosomal recessive pattern. Recent genetic analyses have suggested at least three loci, JBTS1 (9q34.3), JBTS2 (11.2-q12.3), and JBTS3 (6q23). 770 Only the JBTS3 gene, AHI1, encoding Jouberin, has been cloned. JBTS1 and 3 primarily show features restricted to the CNS, with JBTS1 showing largely pure cerebellar and midbrain hindbrain junction involvement, and JBTS3 displaying cerebellar, midbrain hindbrain junction, and cerebral cortical features, most notably polymicrogyria. Conversely, JBTS2 is associated with multiorgan involvement of kidney, retina, and liver, in addition to the CNS features, and results in extreme phenotypic variability. 771 Occasionally, COMA is one of the manifestations of a degenerative disorder such as ataxia telangiectasia. 53,727 Some children with ataxia telangiectasia may pose a diagnostic quandary by showing no overt immune dysfunction, inconspicuous oculocutaneous telangiectasia, and an atypical neurological presentation with dystonia predominating over cerebellar ataxia. 166 Vertical saccades are usually involved to some degree when COMA is associated with systemic disease. 323 Although ataxia telangiectasia is said to be associated with slow saccades, eye movement recordings in patients with ataxia telangiectasia have showed saccadic velocities to be faster than normal, but followed by slow eye movements that were not clearly saccadic in origin. 492 A syndrome mimicking ataxia telangiectasia with slowly progressive ataxia, choreoathetosis, and ocular motor apraxia in the horizontal and vertical plane has been described. 12 Although the neurological findings are indistinguishable from those of ataxia telangiectasia, the onset tends to be later and patients have not shown evidence of multisystem involvement. Ocular motor apraxia is often found in patients with Gaucher disease, 253 a lysosomal storage disorder, and may even be the presenting feature of the disease. 316 Other children with Gaucher disease show a supranuclear horizontal gaze palsy. 611 In most patients with COMA, the ability to generate saccades improves over the first decade, with better eye movements and less noticeable head thrusts. 177 This spontaneous improvement led Cogan to favor a delayed maturation of the ocular motor pathways rather than congenitally absent initiation pathway for horizontal saccades as the underlying cause. While the ocular motor abnormalities tend to improve with age, most affected children show some degree of delayed motor, speech, or cognitive development. 726 Notwithstanding the benign course in most cases, it is advisable to obtain MR imaging to rule out intracranial pathology (tumor, congenital structural malformations), the occurrence of which has been well documented in a minority of children. Treatment of underlying pathology may ameliorate or cure the apraxia in some cases, 833 but not in all. 731 For instance, Zaret et al 833 described a case of COMA that showed rapid improvement after surgical evacuation of a large cystic tumor in the rostral brainstem, while complete resection of a posterior fossa lipoma in a 10-month-old girl had no effect on the apraxia. Although typical COMA involves a bilateral palsy of volitional horizontal saccades and unilateral cases, 146,434 cases involving only vertical saccades have been described. 383,645 Vertical Gaze Palsies in Children Vertical gaze disturbances are generally less common than horizontal ones and, among vertical disorders, upgaze palsy and combined upgaze and downgaze paralysis are more common than downgaze palsy (Fig. 7.8). Most vertical gaze palsies are supranuclear and conjugate (i.e., upgaze palsy, downgaze palsy, and vertical gaze palsy). Supranuclear disconjugate vertical gaze syndromes are rare, and their topographic correlation is less precisely determined. They include skew deviation and variants (e.g., slowly alternating skew deviation), seesaw nystagmus, monocular upgaze palsy,
16 324 7 Complex Ocular Motor Disorders in Children Fig. 7.8 Supranuclear vertical gaze palsy. Five-year-old boy with cerebral palsy shows total paralysis of (a) upgaze and (b) downgaze. (c) Right gaze and (d) left gaze are intact. (e) Elevation of eye with Bell s phenomenon indicates that upgaze paralysis is supranuclear ocular tilt reaction, vertical one-and-a-half syndrome, and V-pattern pseudobobbing. Some of these disorders are not only location-specific, but also point to a specific mechanism of injury (e.g., acute downgaze palsy suggests bilateral infarction of the posterior thalamo-subthalamic paramedian territory). Occasionally, cases involving complete vertical ophthalmoplegia are described that remain unexplained despite thorough evaluation. Nightingale and Barton 578 described a 6-year-old girl who had episodes of severe ataxia and vertical supranuclear ophthalmoplegia. Horizontal eye movements were not affected, and the patient was normal in between attacks. Congenital vertical ocular motor apraxia may occur as a benign nonprogressive condition similar to the horizontal variety or may signal the presence of intracranial tumors, especially if other neurological signs are present. 231 Ebner reported a case of purely vertical ocular motor apraxia in a 4-year-old boy whose MR imaging demonstrated bilateral subthalamic lesions. 231 Rare cases of isolated vertical COMA have also been associated with birth asphyxia. 27,383,645 The association of this disorder with perinatal hypoxic encephalopathy implicates injury to the frontal eye fields and posterior parietal cortex or their descending projections through the internal capsule and basal ganglia to the rostral interstitial nucleus of the medial longitudinal fasciculus (rimlf). Downgaze Palsy in Children Isolated downgaze palsy usually results from a bilateral lesion (usually infarction) involving the midbrain reticular formation and affecting the lateral parts of the rimlf bilaterally.
17 Gaze Palsies, Gaze Deviations, and Ophthalmoplegia In general, an acquired isolated downgaze paralysis requires bilateral lesions at the level of the upper midbrain tegmentum, while a unilateral lesion may be sufficient to produce an upgaze palsy or a combined upgaze and downgaze palsy. 79 Green et al 311 reported a 9-year-old girl with selective downgaze paralysis following pneumococcal meningitis. MR imaging showed bilateral lesions in the rimlf. In the setting of a bilateral midbrain lesion, the examiner can often use horizontal eye movements to assess which side of the midbrain is more severely affected. If the right side of the midbrain is more severely affected, the patient will have a saccadic deficit to the left and a pursuit deficit to the right, because saccadic innervation from the hemispheres is crossed and pursuit innervation is uncrossed. In adults, downgaze paresis is most commonly an early sign of progressive supranuclear palsy. Kumagai et al 455 described a patient with selective downgaze paresis who had a pineal germinoma with bilateral involvement of the thalamo-mesencephalic junction. Rhythmic vergence eye movements (alternating convergence and divergence) were observed at a rate of 3 Hz during eyelid closure. Downgaze palsy in children is a well-known feature of DAF (downgaze palsy, ataxia or athetosis, and foam cells in the bone marrow) syndrome, a neurovisceral storage disease considered to be a variant of Niemann Pick disease type C. It is characterized by supranuclear gaze palsy in the vertical plane (typically downgaze palsy), hepatosplenomegaly, slowly progressive ataxia, mental deterioration, and other CNS disorders. Foamy cells or sea-blue histiocytes in the bone marrow as well as accumulation of sphingomyelin, cholesterol, and other glycosphingolipids are characteristic histopathologic findings. The acronym DAF was coined by Cogan to denote this triad of findings. 176 Rarely, patients show predominantly horizontal supranuclear gaze palsy. 373 An autosomal recessive inheritance pattern is suspected. 42 The age of onset of neurologic symptoms is between 5 and 15 years. In addition to abnormalities indicated by the acronym, affected patients have hepatosplenomegaly, dementia, and widespread CNS dysfunction. Other neurological symptoms and signs include poor coordination, slurred speech, dysphagia, seizures, cerebellar dysfunction, hyperreflexia, and involuntary movements (dystonia, chorea, or athetosis). 100 The DAF variant of Niemann Pick disease should be suspected when evaluating school-aged children who have suffered a recent decline in intelligence or school performance and who show progressive neurologic disease and vertical supranuclear ophthalmoplegia. An acquired condition of bilateral downgaze palsy with monocular elevation palsy, termed the vertical one-and-onehalf syndrome, has been reported in a patient with bilateral infarction in the mesodiencephalic region. 201 The authors speculated that the lesions may have affected the efferent fibers of the rimlf bilaterally and the supranuclear fibers to 325 the contralateral superior rectus subnucleus and ipsilateral inferior oblique subnucleus. This disorder should be distinguished from a different condition, also termed a one-andone-half syndrome, consisting of bilateral upgaze palsy and monocular depression deficit due to thalamomesencephalic infarction ipsilateral to the downgaze paresis. 80 Upgaze Palsy in Children The most common causes of upgaze palsy in children include hydrocephalus (congenital and acquired) and various conditions associated with the dorsal midbrain syndrome, such as a tumor in the pineal region, arteriovenous malformations, encephalitis, and third ventricular tumors. 421 Midbrain infarction, multiple sclerosis, and syphilis are rare causes in children. The eye signs of hydrocephalus are detailed in the chapter on neuro-ophthalmologic signs of intracranial disease. Ocular motility disorders reported in hydrocephalus are listed in Table 7.6. Upgaze paresis is the hallmark of the dorsal midbrain syndrome. 326 Mild cases involve only upward saccades, but severe cases show paralysis of all upward movements. Attempts to produce upward saccades, best elicited by fixating a downward rotating optokinetic nystagmus drum, evoke convergence-retraction nystagmus. The pupils are usually mid-dilated and show light-near dissociation. The upper eyelid shows pathologic lid retraction (Collier s sign) and lid lag, due to disruption of inhibitory fibers from the posterior commissure to the central caudal nucleus. 182 The setting sun sign is unique to children and is suggestive of congenital hydrocephalus. It is not clear why a similar sign is not seen in adults with acquired hydrocephalus. The setting sun sign may be thought of as a combination of Collier s sign and tonic downgaze, which has been reported in some children with hydrocephalus, with or without associated intraventricular hemorrhage. 735 Table 7.6 Ocular motility disorders in hydrocephalus 59,167 A-pattern esotropia with superior oblique muscle overaction Convergence insufficiency Convergence-retraction nystagmus Convergence spasm Fixation instability Lid retraction Mydriasis with light-near dissociation Pseudo-abducens palsy Setting sun sign (in infants) Skew deviation Superior oblique palsy (unilateral or bilateral) Upgaze paralysis (affecting saccades more than pursuit) V-pattern pseudobobbing (with shunt failure)
18 326 7 Complex Ocular Motor Disorders in Children The upgaze palsy of congenital hydrocephalus usually improves dramatically and quickly after shunt placement. Incomplete, delayed improvement over months to years suggests damage to the upgaze pathway either as a result of thalamic hemorrhage and infarction or by the hydrocephalus itself. 735 Children with severe, congenital visual loss may have difficulty moving their eyes volitionally. In this context, upward gaze is the most severely affected. 393 Jan et al 393 theorized that a selective upgaze deficit exists because children with marked visual impairment rarely look up, because they see much more with their limited vision when viewing closer objects sideways or downward. Isolated paralysis of upgaze may be the presenting sign of the Miller Fisher syndrome. 423 It may also result from vitamin B 1 or B 12 deficiency. 671 Isolated bilateral elevation deficiency may be due to congenital restriction of the inferior rectus muscles in the congenital fibrosis syndrome. 759 Diagnostic confusion arises because congenital fibrosis syndrome can produce convergent movements of the eyes in attempted upgaze, which simulates convergence retraction nystagmus. The distinguishing clinical feature is that patients with congenital fibrosis syndrome display ptosis rather than lid retraction. As discussed below, these restrictive phenomena can also be differentiated from the dorsal midbrain syndrome by the positive family history, positive forced ductions, and the finding of hypoplasia of the intracranial nerves and extraocular muscles in the congenital fibrosis syndrome, but not in dorsal midbrain syndrome. In adult patients, upgaze palsy is usually less disabling than downgaze palsy, because the superior visual space is comparatively less important. This is in contrast to downgaze palsies, which cause significant visual deficits due to the importance of downgaze for such tasks as reading, walking, and eating. However, the visual significance of upgaze palsy is much more profound in children who, by virtue of their short stature, spend a considerable amount of their time looking up. Diffuse Ophthalmoplegia in Children The numerous manifestations of diffuse ophthalmoplegia in children can usually be differentiated by clinical history and associated findings (Table 7.7). 305 Mitochondrial disorders and the spinocerebellar ataxias (SCAs) should be considered when the onset is gradual and the eye signs are progressive, and when myasthenia gravis has been ruled out. Ocular myasthenia gravis is associated with variability and fatigability, with or without muscle systemic weakness. Congenital cranial dysinnervation disorders, such as congenital fibrosis syndrome, are usually present at birth and often associated with ptosis and fixed downgaze. Many other conditions that Table 7.7 Diffuse ophthalmoplegia in children Bickerstaff brainstem encephalitis Botulism Chronic progressive external ophthalmoplegia Intrinsic brain stem tumors Kearns Sayre syndrome Maple syrup urine disease Medications (e.g., toxic doses of phenytoin, amitriptylene) Miller Fisher syndrome Mitochondrial encephalopathy (CPEO, MELAS, MERRF, MNGIE, MDS, Pearson syndrome) Myasthenia gravis Olivopontocerebellar degeneration Tick fever Toxicity of chemotherapeutic agents (e.g., vincristine) present with acute ophthalmoplegia, including Fisher syndrome, botulinum, and tick fever, can cause usually have telltale historical clues. Each disorder has a characteristic, if overlapping, clinical profile, making it possible for the diagnosis to be established on clinical grounds in most cases. Chronic Progressive External Ophthalmoplegia Chronic progressive external ophthalmoplegia (CPEO) is an umbrella term that includes a number of diverse conditions having in common insidious onset of slowly progressive, typically symmetric, multidirectional external ophthalmoplegia. The various conditions encompassing CPEO range from disorders limited to the eyelids and extraocular muscles to ones that include systemic and encephalopathic features. Extraocular muscles have smaller motor unit sizes, higher motor neuron discharge rates, higher blood flow, and higher mitochondrial volume fractions than skeletal muscles. 688 These differences suggest that the energy demands and therefore the susceptibility to mitochondrial dysfunction are greater in extraocular muscle. Because of the often gradual and symmetric involvement of the extraocular muscles, patients with CPEO generally do not experience diplopia. 59,614,639,713 Ptosis and ophthalmoplegia generally occur together, but each can occur alone, and the ptosis can be asymmetrical. When strabismus does occur, the most common deviation is exotropia, with or without a vertical deviation. 639,700 Confirmation of the diagnosis usually requires fresh muscle biopsy for histopathological examination (using cytochrome oxidase stain with electron microscopy to look for parking lot inclusions) and Southern blot analysis to look for deletions. Polymerase chain reaction (PCR) has recently been used to detect the mutation in swabbed buccal cells. 386 Mitochondrial encephalopathy is divided into several clinical phenotypes, including Kearns Sayre syndrome; the
19 Diffuse Ophthalmoplegia in Children syndrome of mitochondrial encephalopathy, lactic acidosis, and stroke (MELAS); mitochondrial neurointestinal encephalopathy (MNGIE); and the syndrome of myoclonus, epilepsy, and ragged red fibers (MERRF). 688 The later syndrome is not generally associated with external ophthalmoplegia. Kearns Sayre syndrome is characterized by the triad of progressive external ophthalmoplegia, pigmentary degeneration of the retina, and heart block. Most cases are sporadic. The onset of the disorder occurs before age 20. Affected patients have short stature, other neurologic disorders, and an elevated protein concentration (>100 mg/dl) in the cerebrospinal fluid (CSF). Other findings may include hearing loss, cerebellar signs, mental retardation, delayed puberty, vestibular abnormalities, and ragged red fibers on muscle biopsy, 688 mental deterioration, pyramidal signs, and diabetes. 59,212,424,472,705,838 In addition to complete heart block, cardiac conduction abnormalities include bundle branch block, bifascicular disease, and intraventricular conduction defects and are thought to result from an associated cardiomyopathy. The heart block generally occurs years after onset of the ocular signs and may cause sudden death. The retinal pigmentary degeneration progresses slowly and may be too subtle early on to detect ophthalmoscopically. The associated ptosis may become visually significant, but surgical correction should be approached with caution because the limited eye movements and the absence of Bell s phenomenon render patients prone to exposure keratopathy. Orbital neuroimaging is probably of little value in CPEO. One study found marked atrophy of the extraocular muscles, 608 while another showed little or no reduction in extraocular muscle volume. 601 Furthermore, myasthenia gravis may also show atrophic extraocular muscles late in the course of the disease, making it difficult to distinguish from CPEO. 595 It has been suggested that the myopathic process that results in chronic progressive external ophthalmoplegia renders rectus muscle recessions less effective than resections for correcting the associated strabismus that occasionally develops in these patients. 507,713 Postoperative prisms are occasionally necessary to fine-tune single binocular vision. 791 CPEO may be sporadic, show Mendelian inheritance, or show maternal inheritance. 386 Almost half of CPEO cases are sporadic and associated with single deletions of mtdna. 74,314,315,476,617 In sporadic cases of CPEO and Kearns Sayre syndrome, all the mutant mtdnas in an individual have the same deletion. Because Kearns Sayre syndrome is an artificial construct of the most severe cases of the CPEO syndromes, it is always more likely to have deletions (90%) than CPEO in general, and it is more likely to be sporadic. However, some members of severe autosomal dominant CPEO pedigrees have had a high deletion load express the full-blown Kearns Sayre syndrome phenotype, while other family members do not. 74 Autosomal dominant or recessive inheritance is noted in about 15% of patients with CPEO. Autosomal dominant 327 inheritance is characterized by accumulation of multiple deletions of mtdna in the patient s tissues. Most single deletions are not inherited, but occur spontaneously after fertilization of the oocyte. Individuals with multiple different deletions likely come from pedigrees in which there is a nuclear mutation in a gene encoding a protein that regulates mitochondrial DNA replication and therefore is inherited mendelianly (usually autosomal dominant or autosomal recessive). 59,74,315,838 No clinical difference between hereditary and sporadic CPEO can be demonstrated. 59,838 Many patients without structural abnormalities of mitochondrial DNA have point mutations at many different nucleotide positions, which are inherited maternally. 386 Point mutations causing CPEO are often homoplasmic. Large-scale deletions resulting in CPEO are almost always heteroplasmic; the more tissue with the deletions (i.e., the greater the degree of heteroplasmy), the more likely the phenotype will be severe (i.e., more toward Kearns Sayre syndrome phenotype rather than the simple CPEO phenotype). 47,783 While mitochondrial disorders that have both dominant and recessive modes of inheritance are usually caused by mutations at different loci, both can be caused by mutations in the polymerase gamma (POLG) gene. 184,267a The risk of developing a severe phenotype (i.e., additional CNS symptoms with neurological manifestations) is higher when the age of onset is before age 9 and lower when the onset is after age While mitochondrial disorders associated with ophthalmoplegia tend to affect adults, early symptoms are often experienced in childhood. MELAS is the mitochondrial disease that is most consistently associated with retrochiasmal visual loss. MELAS is characterized by recurrent abrupt attacks of headache, vomiting, focal and generalized seizures, and focal neurologic symptoms and signs lasting hours to days. 74 There is a posterior cerebral predilection for damage, and visual disturbances have been reported in more than half of patients. It is not uncommon for patients with MELAS to have CPEO, pigmentary retinopathy, optic atrophy, or homonymous hemianopia. Patients may present with stroke-like episodes, seizures, headaches, progressive dementia, and exercise intolerance. 539 Classically, the brain MRI of patients with MELAS demonstrates lesions that may mimic ischemia, except that they usually do not respect vascular territories and are often restricted to the cortex with relative sparing of deep white matter. 366 However, lesions spanning both gray and white matter have been reported. 554 The diagnosis of MELAS is established made from the characteristic neuroimaging findings, elevated lactic acid levels, ragged red fibers on muscle biopsy, or genetic sequencing. MELAS is often maternally inherited, with 80% of patients harboring the A3243G transition within the mitochondrial genome, although many other mutations have also been reported. 162 Patients with MERRF syndrome present with combinations of myoclonic epilepsy, ataxia, spasticity, generalized seizures, optic atrophy, and/or dementia. 315 They may
20 328 7 Complex Ocular Motor Disorders in Children also have muscle weakness, myopathy, neuropathy, ophthalmoplegia, ptosis, headache, foot deformity, cervical lipoma, or sensorineural hearing loss. Myoclonus is usually the presenting symptom and is often precipitated by noise, photic stimulation, or action. Myopathy is generally subclinical or mild. Biochemical aberrations may include elevations of serum pyruvate, or pyruvate and lactate, and reduced activities of complexes I and IV. The most common mutation, accounting for 80% of MERRF cases, is at np 8344 within the trna. 346 Other mutations in the same trna gene, t8356c and G8363C, are also found in association with this phenotype. 606 Mitochondrial Neurogastrointestinal Encephalomyopathy (MNGIE) syndrome is an autosomal recessive syndrome characterized by chronic progressive external ophthlmoplegia intestinal hypomotility, and neuropathy. 795 Clinical presentation is usually in childhood, although gastrointestinal symptoms may be problematic long before other features of the disease appear. Some patients have diabetes mellitus, heart block, or cardiomyopathy. Long-term prognosis is poor, with most patients dying in early-to-mid adulthood from weight loss and other gastrointestinal complications. 795 MR imaging may show an unsuspected leukoencephalopathy of the brain. 184,365,542,581 Most patients have mutations in the nuclear-encoded thymidine phosphorylase gene. 795 In the child with progressive external ophthalmoplegia, a history of marrow failure with transfusion-dependent sideroblastic anemia, and exocrine pancreatic dysfunction causing malabsorption or diarrhea should raise suspicion for the Pearson syndrome. This disorder is characterized by a progressive pigmentary retinopathy that initially spares the posterior pole. Older patients may develop symptoms of Kearn Sayre syndrome. It is caused by deletions in mitochondrial DNA. 659a Myasthenia Gravis Myasthenia gravis was the first recognized autoimmune neurologic disease. Myasthenia gravis is a disorder of neuromuscular transmission characterized by fatiguability and fluctuating muscular weakness, with a predilection for the extraocular muscles. 48 In severe cases, acute respiratory failure and death may occur. About half of all patients with myasthenia present with ophthalmologic symptoms. These include ptosis, strabismus, and limited ocular ductions, all with a tendency to be highly variable. No pupillary involvement is clinically discerned in myasthenia, and the presence of pupillary signs effectively excludes the diagnosis. Myasthenia gravis has a well-known predilection for extraocular muscle involvement. Factors that may predispose the extraocular muscles to preferential involvement in myasthenia gravis include their higher discharge rates, differences in complement inhibitor proteins, and lack of action potentials in the tonic fibers. The more constant activity also makes the orbital extraocular muscle fibers more susceptible to fatigue ,481 It has also been suggested that antibodies directed against the fetal form of the acetylcholine receptor, which may be found at synapses on extraocular but not skeletal muscles, may be an important factor that predisposes the extraocular muscles to involvement by myasthenia. 407 Nearly 90% of patients with myasthenia develop ocular involvement at some point during their illness. Most patients with ocular myasthenia who develop systemic symptoms and signs do so within 2 years of onset. 69,459 Three distinct myasthenic syndromes may be encountered in the pediatric age group: transient neonatal, congenital, and juvenile. This classification is not based on age at presentation but on pathophysiology. Both congenital myasthenia and juvenile myasthenia may present any time between infancy and adulthood, but are distinguished primarily by the fact that congenital myasthenia is not immune-mediated. However, this distinguishing feature is not absolute because juvenile- and adult-onset myasthenia may also be antibodynegative. Also, the defect in acquired myasthenia is postsynaptic, while the defect in congenital myasthenia may be either at the presynaptic or postsynaptic level. Transient Neonatal Myasthenia Transient neonatal myasthenia gravis is due to transfer of antibody from the mother, tends to spare eye movements. 285 Approximately 12% of newborn infants of myasthenic mothers develop transient myasthenic symptoms, presumably due to passive transplacental transfer of anti-achr IgGs. Serum AChR antibody titers of affected neonates follow the same pattern as their mothers. Neonatal disease does not appear to correlate with the severity of maternal symptoms; affected mothers commonly have active myasthenia, but they may be in remission or, rarely, have undiagnosed subclinical disease. The onset of transient neonatal myasthenia occurs within a few hours after birth in two-thirds of patients, and within the first 3 days in all of them. Affected infants present with temporary skeletal muscle weakness producing hypotonia, a feeble cry, difficulty sucking and swallowing, facial diparesis, and mild respiratory distress. Occasionally, they may suffer respiratory depression that requires mechanical ventilation. Transient neonatal myasthenia tends to spare eye movements. 285 Ocular involvement, including ptosis, limited eye movements, and orbicularis weakness, affects 15% of infants. An atypical, more severe form of transient neonatal myasthenia includes the above-mentioned manifestations in addition to multiple joint contractures and occasional prenatal difficulties such as polyhydramnios or decreased fetal movement. Unlike the typical variety, response to oral or parenteral
21 Diffuse Ophthalmoplegia in Children anticholinesterase agents is poor. Severe cases may rarely require assisted mechanical ventilation for up to 1 year. The pathogenesis of the disorder is increasingly understood. It is controversial and uncertain whether the maternal and neonatal antibodies are similar and passively transferred, or different, with the neonate synthesizing their own antibodies, or whether both mechanisms are in play. 479,760 Transient neonatal myasthenia is not fully explained by passive transplacental IgG transfer because these antibodies are also found in the serum of many newborns who are asymptomatic. Other infants are symptomatic without detectable antibodies in their maternal serum. Also, high IgG levels have been found in a few asymptomatic infants. There is now a report of transient neonatal myasthenia in an infant with anti-musk antibodies. 579 Clinical symptoms usually last 2 or 3 weeks, 560 but may resolve in 1 week or linger on for 2 months before complete recovery. Response to oral (e.g., pyridostigmine bromide) or parenteral anticholinesterase agents is very good, and these agents should be administered until spontaneous resolution occurs. No permanent neuromuscular sequelae are detectable after resolution. If this condition is not recognized and promptly treated, some affected infants may deteriorate from respiratory depression to respiratory arrest and death. The characteristic clinical features and the history of maternal myasthenia should be enough to confirm the diagnosis, but the diagnosis may be delayed if the mother s disease has not been previously detected. Further verification of the diagnosis may be derived from a favorable response to neostigmine methylsulfate, with improvement of symptoms min after intramuscular injection of 0.15 mg/kg of body weight. Alternatively, intramuscular edrophonium chloride (Tensilon) may be administered intramuscularly or subcutaneously (0.15 mg/kg) or intravenously (0.10 mg/kg). Sufficient differences exist between transient neonatal myasthenia and the congenital myasthenic syndromes (see below) to render diagnostic confusion rare. Congenital myasthenia does not occur in infants born to mothers with acquired myasthenia. The other condition affecting neuromuscular transmission in this age group, infant botulism, is readily ruled out because it occurs after the second week of life, 5 days after the infant ingests food contaminated with Clostridium botulinum, whereas the onset of transient neonatal myasthenia is within the first 3 days of life only. Congenital Myasthenic Syndromes Congenital myasthenia constitutes a heterogeneous group of genetic disorders affecting neuromuscular transmission that presents in the first year of life, producing mainly ptosis, and usually has a benign course. 239,240, ,351 Although rare, congenital myasthenic syndromes are an important cause of 329 seronegative myasthenia gravis. Congenital myasthenic syndromes occur in infants born to nonmyasthenic mothers and that are associated with hypotonia and weakness. 60,239,240 Congenital myasthenic syndromes should be suspected in patients with seronegative myasthenia gravis, in infants with hypotonia and poor development of motor milestones, and in patients with a childhood history of neuromuscular difficulties affecting cranial, respiratory, truncal, or limb muscles. 351 In some patients, however, symptoms may not appear until later childhood or even adulthood. Forty-two percent of the cases present before the age of 2 years, and over 60% before 20 years. 574 In contradistinction to juvenile and adult-onset myasthenia, which have an autoimmune basis and are attributable to antibodies that bind to the acetylcholine receptor and cause increased turnover and destruction of the receptor, congenital myasthenia is not immune-mediated. 351 The congenital myasthenia syndromes are caused by structural or functional alterations at the myoneural junction. 246,550,699 Most cases present in the neonatal period or shortly thereafter with poor feeding, failure to thrive, and weakness. Three congenital myasthenia syndromes (slow-channel syndrome, prolonged channel open time syndrome, and limb girdle myasthenia) may present later in childhood or adolescence and represent a specific, rare group of potential differential diagnoses. 351 A distinction between congenital and acquired myasthenia cannot be made with certainty on the basis of the age of onset because both types may manifest during the neonatal period, infancy, or childhood, and acquired myasthenia may also be antibody-negative. 288 Congenital myasthenia is usually characterized by ocular musculature with ophthalmoparesis, orbicularis weakness, and ptosis. We described a unique child who had a congenital oculomotor nerve palsy with aberrant regeneration and fatigable ptosis associated with congental myasthenia gravis. During periods of levator fatigability, gaze activation of the ipsilateral superior or medial rectus muscles produced instantaneous levator retraction with no fatigability (Fig. 7.9). The ability of this child s synkinetic neuromuscular connections to override her myasthenia suggested that there was selective sensitivity of the myasthenic levator muscle to the misdirected oculomotor axons, which presumably provided fresh synaptic reserve of acetylcholine to their target fibers. 111 Congenital myasthenic syndromes can arise from presynaptic, synaptic, or postsynaptic defects. 244 The major syndromes in which the defect is presynaptic are related to defects in Ach synthesis, mobilization, or release. 351 Presynaptic congenital myasthenic syndromes were previously called familial infantile myasthenia. 355,647 Postsynaptic syndromes are mainly caused by endplate acetylcholinesterase deficiency and Ach receptor kinetics, including the fast- and slow-channel syndromes. 785 The array of disorders and involved genes identified has increased significantly. 351 While genes have been identified for many
22 330 7 Complex Ocular Motor Disorders in Children Fig. 7.9 Myasthenic ptosis in a child with congenital left oculomotor nerve palsy. Upper panel: Facial photographs depict the clinical variability of the left upper lid ptosis and the compensatory head turn before treatment. This child s congenital myasthenia was worse on the day of the right photograph, as evidenced by the increased ptosis and left facial weakness. Lower panel: Primary and secondary positions of gaze shing retraction of the left upper eyelid in adduction and supraduction due to oculomotor synkinesis. Even with prolonged fixation, fatigable ptosis could not be elicitied in these positions of gaze. With permission from Brodsky 111 forms of congenital myasthenic syndromes, mutation analysis is not yet available on a commercial basis. 60,239,240 Some cases may be familial, with other siblings affected, supporting a genetic basis for the disorder. 733 Most commonly, congenital myasthenic syndromes are caused by mutations that reduce the expression or alter the kinetics of the acetylecholine receptor. 245 Reduced receptor expression is also caused by mutations in rapsyn, a molecule that is produced by muscle and is important for acetylcholine receptor aggregation on the postsynaptic membrane. 245 The second most common congenital myasthenic syndrome is caused by mutations of the acetylcholinesterase molecule. 242 Mutations of choline acetyltransferase, the ratelimiting enzyme in the synthesis of acetylcholine, cause the congenital myasthenic syndrome associated with episodic apnea (previously known as familial infantile myasthenia ). 591 Recently, a unique congenital myasthenic syndrome caused by mutations of a perijunctional skeletal muscle sodium channel has been discovered. 521 Congenital myasthenia is not immune-mediated, in contradistinction to juvenile- and adult-onset acquired myasthenia, which have an autoimmune basis and are attributed to antibodies that bind to the acetylcholine receptor and cause increased turnover and destruction of the receptor. 26 Serum antiacetylcholine receptor antibodies and other autoantibodies are absent. Congenital myasthenia is not associated with any autoimmune
23 Diffuse Ophthalmoplegia in Children Table 7.8 Classification of congenital myasthenic syndromes (based on 271 index cases evaluated at Mayo Clinic) Presynaptic defects (7%) Choline acetyltransferase (ChAT) deficiency Paucity of synaptic vesicles Congenital Lambert Eaton-like syndrome Other unclassified presynaptic defects Synaptic defect (basal lamina) (14%) Endplate AChE deficiency Postsynaptic defects (79%) Reduced AChR expression (with or without minor AChR kinetic abnormality) Rapsyn mutations MuSK mutations DOK-7 mutations (formerly Limb-Girdle myasthenia) expression) Slow-channel syndrome Fast-channel syndrome AChE acetylcholinesterase; AChR acetylcholine receptor; ChAT choline acetyl-transferase; MuSK muscle specific kinase disease or any particular human leukocyte antigen genotype, and cytohistochemical studies fail to reveal immune complexes at the myoneural junction. The condition is generally nonremitting. Once thought to be a single disorder, it is now known to represent a group of diverse disorders distinguishable by the specific site of dysfunction at the myoneural junction (Table 7.8). Multiple inherited defects of neuromuscular transmission at presynaptic or postsynaptic levels are known, but some defects have not yet been characterized. The putative inheritance of most of these defects is autosomal recessive, with the exception of the slow-channel syndrome, which is autosomal dominant. The precise characterization of these defects requires the combined use of clinical, electromyographic, in vitro electrophysiological, and morphological data. It is probably impractical to perform all tests needed for accurate characterization of the myoneural defect on each infant suspected with the diagnosis, especially when the infant is ill. However, determination of the specific subtype of congenital myasthenia would be helpful for therapeutic purposes and would enhance our understanding of the heretofore incompletely understood disorders that constitute congenital myasthenia. Certainly, a detailed history should be obtained on each child regarding the onset and severity of feeding difficulty, breathing dysfunction, choking episodes, drooling, facial weakness, ophthalmoparesis, ptosis, hypotonia, and muscular fatiguability. This would start the process of differentiation between the different syndromes involved. For example, a syndrome characterized by defective AChR shows neonatal respiratory difficulty, feeding difficulty, and ophthalmoparesis, 331 while a syndrome characterized by impaired ACh release shows few, if any, of these features. The developmental milestones, progression or regression of symptoms and signs during infancy and childhood, response to any therapeutic modalities, and a complete family pedigree should be recorded. In some congenital myasthenia syndromes, a specific diagnosis can be made by simple histological or EMG studies while, in others, a complex panel of in vitro electrophysiological, ultrastructural, and immunocytochenical investigations are needed for accurate diagnosis. 244 Because the congenital myasthenic syndromes are not immune-mediated, neither plasmapheresis nor immunosuppression has any beneficial effect. Thymectomy usually produces negligible benefits, although a transient improvement has been reported in two patients, one of whom had an abnormal thymus. 170,803 Because of the diversity of the underlying abnormalities, no specific conclusion can be drawn regarding the efficacy of the anticholinesterase preparations. Some types of congenital myasthenia (e.g., congenital acetylcholine receptor deficiency) respond favorably to anticholinesterase preparations, while other types (e.g., acetylcholinesterase deficiency, slow-channel syndrome) are refractory to such treatment and may even be made worse by it. Therefore, an effort to differentiate juvenile myasthenia from congenital myasthenia and to specifically identify the type of congenital myasthenic syndrome has important therapeutic implications. On the basis of the identified pathophysiology of the congenital myasthenic syndrome, a number of other pharmacologic interventions are tried in individual children, including fluoxetine, ephedrine, albuterol, and 3,4-diaminopyridine. Juvenile Myasthenia Juvenile myasthenia is said to be similar to the adult form, but more frequently familial, showing slower progression, characterized by more severe ophthalmoplegia, and having a higher rate of spontaneous remissions. 778 Children with myasthenia often first present to the ophthalmologist with symptoms such as ptosis, diplopia, strabismus, and ophthalmoplegia. 90,285 Juvenile myasthenia is otherwise similar to the adult variety in presentation, pathogenesis, clinical course, and response to therapy. While it has been observed that there is an increased incidence of autoimmune disorders in children with acquired autoimmune myasthenia gravis and their first-degree relatives, acquired autoimmune myasthenia gravis is probably not a familial disease. However, some studies reveal the juvenile variety to be more frequently familial, 571,574 to show more severe ophthalmoplegia, and to have slower progression and a higher rate of spontaneous remissions. An acute fulminating form of myasthenia gravis has been described, with onset between 2 and 10 years of age, with respiratory crisis as the presenting feature of the disorder. 264
24 332 7 Complex Ocular Motor Disorders in Children Many medications have been reported to incite or worsen myasthenia. 796,811 These drugs may interfere with neuromuscular transmission pre or postsynaptically. 811 Cardiovascular drugs (antiarrhythmics and beta adrenergic receptor-blocking agents), anticholinergics, anticonvulsants, antirheumatics, immunosuppressives, psychotropics, and antibiotics have all been reported in association with myasthenia. 811 Many antibiotics, including kanamycin, ampicillin, imipenem with cilastatin, ciprofloxacin, erythromycin, and clarithromycin, have also been reported to cause myasthenia. 624,656 Interferon-alpha, a drug frequently used in the treatment of malignancies and viral hepatitis, has been found to exacerbate myasthenia. 83,768 Wasserman reported ocular myasthenia in a 10-year-old girl taking nitrofurantoin that resolved after discontinuation of the drug. 796 Rarely, myasthenia gravis in children can be precipitated by a viral illness. 260,530 Molecular mimicry between the acetylcholine receptor and viral proteins may be involved in the pathogenesis of postinfections myasthenia gravis. Autoimmune manifestations of hyperthyroidism and myasthenia gravis occasionally overlap. 682 The finding of lid retraction in a myasthenic child should lead to suspicion of this phenomenon. A large proportion of young patients with juvenile myasthenia gravis are seronegative. 25,26,256,708 Careful differentiation from late-onset congenital myasthenic syndromes is necessary in these cases. Children younger 5 years of age, who present with ocular symptoms of myasthenia gravis, frequently respond to anticholinesterase medications without requiring immunomodulating therapy and have low rates of progressing to generalized involvement. 569,600 However, one retrospective study found a 43% prevalence of systemic involvement. 524 Several studies have found that pediatric myasthenia gravis shows a female-to-male ratio of about 4:1. 5,361,524 Myasthenia can often be diagnosed on clinical grounds when ptosis or ophthalmoplegia is accompanied by certain neuro-ophthalmologic signs. These include fatiguable ptosis, orbicularis weakness, variable strabismus, quiver-like eye movements, and a Cogan lid twitch sign. A Cogan lid twitch is elicited by having a patient rapidly refixate the eyes from a depressed position to the primary position, with a positive lid twitch sign indicated by the lids overshooting briefly upward before settling in their usual ptotic position. If the lid is first fatigued by sustained upward gaze, the lid twitch sign becomes more exaggerated. Apparently, the short relaxation of the upper lid allowed by fixating an object in downgaze allows for transient recovery of strength by the myasthenic levator muscle. Although ptosis is the most common sign, lid retraction may occasionally be encountered, especially unilaterally in patients with contralateral ptosis. This seemingly paradoxical finding is explained by Hering s law of equal innervation, as the patient attempts to elevate the contralateral ptotic lid. Bilateral lid retraction, which is also rarely reported in myasthenia, is not readily explained by Hering s law. In such cases, the possibility of concurrent thyroid eye disease must be excluded. 412 Children with diffuse myasthenic ophthalmoplegia rarely present with myopia. 652 Myopia occurs when medial rectus weakness leads to exotropia, which necessitates excessive accommodative convergence to maintain single binocular vision. These children are caught in the unpleasant situation of having to sacrifice clear vision to avoid diplopia. Jacqueline Winterkorn, M.D., has observed another unique ocular motor sign of myasthenia that can be elicited in patients with diffuse ophthalmoplegia. An optokinetic drum is spun horizontally for approximately 30 s, as the patient attempts to follow the targets. The drum is then spun diagonally and the resultant optokinetic movements are observed. In myasthenic ophthalmoplegia, the resultant movements are vertical rather than diagonal because the horizontal component has been prefatigued. Conversely, if the drum is first spun vertically for 30 s and then diagonally, the resultant optokinetic movements are horizontal (Winterkorn sign). In nonmyasthenic ophthalmoplegia, optokinetic preconditioning does not alter the trajectory of the patient s diagonal optokinetic responses. Systemic myasthenia is often first suspected when weakness is demonstrated to be fatigable (worsening with use or exercise and relieved by periods of rest). The diagnosis of myasthenia may be confirmed with certain ancillary tests, including serologic testing for antibodies, clinical neurophysiologic testing (repetitive stimulation of motor nerves, needle electromyography, including single fiber testing), and pharmacologic testing. Because children with acquired autoimmune ocular myasthenia gravis have a higher incidence of seronegativity and normal clinical neurophysiologic testing, the diagnosis often depends on other measures. There are three main confirmatory procedures for myasthenia: response to acetylcholinesterase inhibitors, electrophysiologic testing, and antibody assays. In general, all share the same problem of a lower sensitivity in ocular myasthenia gravis than in generalized myasthenia gravis. 58 The sensitivity of the edrophonium test is approximately 95% in generalized myasthenia gravis 602 and is similar in patients with ocular myasthenia gravis with ptosis. 257 However, diplopia fails to respond in approximately one-third of patients, and long-standing myasthenia may not respond at all. The repetitive nerve stimulation test is positive in most patients with generalized myasthenia gravis, but may be normal in well over 50% of patients with ocular myasthenia gravis. 256,257 Single-fiber EMG of the orbicularis oculi is currently the most sensitive test for ocular myasthenia. 481 When coupled with the standard EMG, it usually distinguishes myasthenia from other disorders such as mitochondrial myopathy or oculopharyngeal dystrophy. 643 The sensitivity of single-fiber EMG is more than 90% in generalized myasthenia gravis 669 and approximately 85% in ocular myasthenia gravis. 764 Since needle
25 Diffuse Ophthalmoplegia in Children EMG and repetitive nerve stimulation are usually positive in systemic myasthenia, these tests should first be performed to rule out denervation or atrophy in patients with limb weakness. Only when these tests are negative, or when there are purely ocular signs, is single-fiber EMG indicated. 481 Because it is painful and requires patient cooperation, it may be difficult or impossible to perform in young children. Positive acetylcholinesterase antibodies are found in 90% of cases of systemic myasthenia, but in only 50% of cases of ocular myasthenia. Because myasthenic children have a higher frequency of negative anticholinesterase antibodies, a negative test is not especially useful in this setting. The recent identification of muscle-specific kinase (MuSK) as a target of the autoimmune process in a subset of patients indicates that autoimmune myasthenia gravis includes at least two distinct immunologic disorders directed against different antigens in the postsynaptic membrane of the neuromuscular junction. 24 Autoantibodies targeting MuSK have been demonstrated in approximately 40% of seronegative patients. 257,368,668,784 Although the numbers are small, our experience is that the anti-musk variant tends to be more severe and less responsive to therapy. The Tensilon (edrophonium hydrochloride) test is the most commonly utilized. For children less than 34 kg, a test bolus of intravenous edrophonium should be 0.1 cc or 1 mg, followed by repeated boluses at 1-min intervals to the point of positive response or maximum injection of 5 mg total dose. Cardiac monitoring is advisable. The dose of atropine given for excess parasympathetic response (primarily bradycardia or bradyarrhythmias) should be 0.02 mg/kg/dose, with a maximum of 0.5 mg/dose in younger children and a maximum of 1 mg/dose in adolescents (Fig. 7.10). Results of the Tensilon test may be equivocal, falsely negative, or falsely 333 positive. False-negative results are more common than falsepositive ones. Therefore, if the clinical findings are suggestive of myasthenia, repeating an initially negative Tensilon test is recommended. False-positive results have been reported in patients with compressive lesions (brain tumors, intracranial aneurysm), botulism, Eaton Lambert syndrome, amyotrophic lateral sclerosis, poliomyelitis, transverse myelitis, Guillain Barré syndrome, and myositis. 211,557 Because severe bradyarrhythmias, conduction pauses, and cardiac arrests have been reported, albeit rarely, 389 cardiac monitoring and pretreatment with atropine should be considered in at-risk patients. In young children, neostigmine (Prostigmine) is administered intramuscularly, which produces a more prolonged response in myasthenic patients than does edrophonium chloride. Neostigmine can produce a more prolonged clinical response (onset, 10 min; duration, 60 min), which can allow for more leisurely assessment of the response. Doses are 0.04 mg/kg/dose administered intramuscularly or subcutaneously, with a maximum of 0.5 mg/dose. Side effects related to excess parasympathetic activity can also last up to 60 min and, as clinically indicated, are treated with atropine in a dose of 0.02 mg/kg/dose, intravenously, with a maximum 0.05 mg/kg/dose in younger children and 1 mg/dose in adolescents. To avoid the complications of pharmacological testing, a sleep test has been devised wherein the patient is evaluated for improvement of ocular signs immediately after a 30-min period of sleep (or eye closure) and for worsening of these signs shortly thereafter (Fig. 7.11). 588 The ice test, performed in an outpatient setting, can similarly differentiate a neuromuscular transmission defect from other causes of ptosis. 234,235,254,300,453 In this test, a surgical glove filled with ice Fig Positive Tensilon test in juvenile myasthenia gravis. Child presented with variable ptosis and exotropia (a) that showed resolution upon administration of intravenous Tensilon (b)
26 334 7 Complex Ocular Motor Disorders in Children Fig Sleep test. Left: Child with myasthenic ptosis. Right: Resolution of ptosis following 5 min of gentle eye closure. Good WV, Hoyt CS. Strabismus Management. Boston, MA: Butterworth-Heinemann; 1996 chips is placed over the ptotic eyelid for 2 min after the eyelid has been fatigued by prolonged upgaze. Measurement of the millimeters of ptosis before and after the cooling is used to objectively measure response with lessening of the ptosis, suggesting the presence of myasthenia. The ice test is based on the observation that increased temperature has a detrimental effect, and decreased temperature a beneficial effect, on muscle force generation in myasthenia gravis. 85 Both tests and the sleep test often produce dramatic resolution of myasthenic ptosis, and some of the observed effect of the ice test may be attributable to resting of the closed lid. The medical treatment of juvenile myasthenia is similar to that of the adult variety in that the ptosis shows a good therapeutic response while the strabismus often does not. 436 In a recent retrospective study, children with ocular myasthenia were found to have a high incidence of ptosis (96%) and strabismus (88%), most commonly with exotropia and vertical heterotropia, and amblyopia (21%). The ptosis showed a good therapeutic response in 87%, but the strabismus improved in only 29%. 436 Combined pyridostigmine and prednisone were most commonly used. Treatment with a short course of prednisone and long-term azathioprine is reported to reduce the risk and severity of generalized symptoms and to promote remission of the disease. Although thymomas are rare in myasthenic children, 7 thymectomy has been observed to be associated with an increased rate of clinical remission in children with autoimmune myasthenia gravis. 497,649 Early surgery (within 2 years of clinical onset), bulbar involvement without ocular signs of generalized weakness, clinical onset during adolescence (12 16 years of age) and presence of other autoimmune disorders were associated with increased probability of clinical remission after thymectomy. 649 Surgeons have developed a technique of using video-assisted thorascopic thymectomy that has significantly decreased operative morbidity compared to transsternal thymectomy. 749 Unanswered questions, regarding the impact of thymectomy early in life on the developing immune system, have kept thymectomy limited to children with generalized weakness and will likely lead to additional review of the effectiveness of this operation as a therapeutic option. Because the disease is immune-mediated, unlike in congenital myasthenia, immunosuppression and plasmapheresis have a therapeutic role. Systemic therapy of myasthenia helps control ocular symptoms in most patients. 220,221,711 In addition to producing superior resolution of ocular misalignment and diplopia, 460 treatment with oral prednisone appears to reduce the conversion of ocular to systemic myasthenia gravis ,528,556 After an initial period with immune suppressing dose of corticosteroids, the benefit may be maintained with doses that do not suppress the immune system and appear to cause few major systemic adverse effects. 458 There is a theoretical basis for using corticosteriod treatment to prevent generalized myasthenia, even at doses that do not cause immunosuppression. 457 Corticosteroid treatment of in vitro human muscle cultures increases the number of acetylcholine receptors 98,414 and prevents receptor loss induced by the serum from generalized myasthenia gravis patients. Neuromuscular junctions increase in size, and the length, number, and depths of postsynaptic folds increase after weeks of corticosteroid exposure. 43 In patients with ocular misalignment, strabismus surgery has been used to manage both stable 3,64,197,335,594 and unstable 563 diplopia with long-standing success. Brain tumors rarely produce clinical findings that are indistinguishable from myasthenia. 413,633,730 These findings are attributed to a putative compression of the central caudal nucleus in the dorsal midbrain. 413 Ragge and Hoyt 633 described an adolescent girl with neurofibromatosis type I and a dorsal midbrain astrocytoma who had fatiguable ptosis, upgaze paresis, and a positive lid twitch sign. These findings improved significantly following radiotherapy of the astrocytoma, confirming that the muscular fatiguability was central in origin. Straube and Witt 730 described four patients with posterior fossa tumors who presented only with fluctuating weakness of the external ocular muscles and/or the pharyngeal muscles, leading to an incorrect diagnosis of ocular myasthenia. Moreover, as noted earlier, false-positive results of Tensilon testing may
27 Olivopontocerebellar Atrophy occur in some instances involving tumors or even aneurysms. Branley et al 97 reported a 7-year-old girl who developed an oculomotor nerve palsy of subacute onset due to a cerebral artery aneurysm. The condition was initially confused with myasthenia gravis because the ptosis improved after Tensilon administration (false-positive result), but the key clinical distinguishing findings were the presence of aberrant innervation and pupillary involvement, which are not features of myasthenia. Chemotherapeutic agents, such as, vincristine, may also cause neurological findings (ptosis, ophthalmoplegia, jaw pain) that can confound the diagnostic picture in children with intracranial tumors. 15 Olivopontocerebellar Atrophy The nomenclature regarding olivopontocerebellar atrophy and the related hereditary spinocerebellar ataxias has undergone revision, first to the clinically based autosomal dominant cerebellar ataxias and, more recently, to the genetically defined spinocerebellar ataxias. 611a Olivopontocerebellar atrophy (OPCA) is a pathological label implying not only olivopontocerebellar changes, but also cases with more widespread lesions involving the CNS. 65 OPCA may also be part of the pathological hallmark of other disorders such as mitochondrial encephalomyopathies, prion disorders, and hereditary metabolic diseases. In 1983, Harding 347 developed a clinicogenetic classification, which has been subsequently modified according to molecular genetic advances. Older diagnostic categories, such as olivopontocerebellar atrophy, are now known to carry mutations for spinocerebellar ataxia (SCA1, SCA2, and SCA3). 65 The hereditary ataxias and loci now comprise around 80 loci, with 28 belonging to spinocerebellar ataxia and designated as SCA1 28 in order of discovery. Several SCA subtypes (SCA1, SCA2, SCA3, SCA6, SCA7) are caused by CAG trinucleotide expansions in their respective genes. Autosomal dominant cerebellar atrophy 1 (ADCA1), which presents with a range of findings, including ataxia, pyrimadal and extrapyramidal signs and ophthalmoplegia, corresponds to the current classifications SCA1 4. ADCA2, which is similar but includes retinal degeneration, corresponds to SCA7. ADCA3, which involves relatively pure cerebellar signs, corresponds to SCA6 and Clinically, patients diagnosed with OPCA correspond to those with SCA1, SCA2, and SCA3. SCA1 maps to 6p23, SCA2 to 12q24, and SCA3 to 14q24.3-q31. Macular and retinal degeneration are seen predominantly in SCA7, but occasionally in SCA Patients with SCA7 have abnormal electroretinograms showing predominantly cone dysfuntion. 547a MR imaging shows severe OPCA in SCA2, similar but milder changes in SCA1, and very mild atrophy with sparing of the olives in SCA An SCA panel can be ordered when the one of the spinocerebellar ataxias are suspected. 335 Supranuclear ophthalmoplegia may be accompanied by gaze-evoked nystagmus in patients with SCA1, SCA3, and SCA6, and by rebound nystagmus in SCA1 and SCA Impaired saccadic velocity and latency are characteristic of SCA2 and SCA2. In SCA1, saccadic amplitude is significantly increased, resulting in hypermetria. 132 A supranuclear ophthalmoplegia with severe saccadic slowing is highly characteristic of SCA2, 133 which may show selective involvement of vertical saccades, impaired VOR, and gaze-evoked nystagmus. 132,136 The constellation of impaired VOR, saccadic dysmetria, and slow-wave jerks are common to both SCA3 and Friedreich s ataxia. 565 The association of these ocular motor disorders with dystonia, spasticity, facial and lingual fasciculations, diplopia, and parkinsonism should suggest the diagnosis of Machado Joseph disease (SCA3). 187,687 Many reports have described a striking ocular stare or prominent eyes, which are thought to result from lid retraction rather than proptosis. 229,734 SCA1 4 (previously known as ADCA type 1) are associated with optic atrophy and ophthalmoplegia. SCA7 (previously known as ADCA II) is distinguished by the presence of pigmentation macular dystrophy that includes early granularity and mottling, and is later associated with pigmentation changes that gradually spread to the periphery, with late optic atrophy and attenuation of retinal vessels. 238,308,309,372,392,663,752 The ADCA type II locus, which corresponds to SCA7, has been mapped to chromosome 3p12-p21. SCA7 (or ADCA type II) is caused by an unstable CAG repeat in the SCA gene. Larger expansions are associated with earlier onset, a more severe and rapid clinical course, and a higher frequency of decreased vision, ophthalmoplegia, extensor plantar responses, and scoliosis. The mutation is highly unstable, with an increase in repeats with paternal transmission correlating with marked anticipation. This instability of transmission is more marked than with other SCA subtypes. A supranuclear ophthalmoplegia is commonly seen in more than half of SCA patients. The responsible gene for SCA7 has been cloned. Molecular testing of DNA from whole blood is now available to detect the SCA7 CAG repeat to confirm the diagnosis genetically. The fundamental neurologic lesions of the spinocerebellar ataxias are localized to the cerebellum, and the pontine, inferior olivery, arcuate, and pontobulbare nuclei. 445 Cerebellar atrophy fundamentally involves Purkinje cells and predominates in the neocerebellum. CT scanning and MR imaging show the characteristic brainstem and cerebellar atrophy, with the most sensitive diagnostic feature being the diameter of the middle cerebellar peduncle. 596 Intermediate and T2-weighted MR images may show cruciform hyperintensity ( hot cross bun sign) of the ventral part of the pons, accounted for by demyelination and gliosis of transverse fibers of the pons secondary to atrophy of nuclei pontis. 65
28 336 7 Complex Ocular Motor Disorders in Children It is important to remember that several metabolic disorders that present in infancy or childhood (abetalipoproteinemia, hexosaminidase A deficiency, cholestanolosis, the leukodystrophies, [metachromatic, Krabbe s disease, and adrenoleukomyeloneuropathy], and Refsum disease) can be associated with a progressive and unremitting ataxia that can first appear in early adulthood. 598 Of the vitamin deficiencies, vitamin E deficiency is most often associated with the development of progressive ataxia, associated with areflexia, and loss of proprioception and vibration sensation (abetaliproteinemia or Bassen Kornzweig disease). 348 Affected patients develop pigmentary retinopathy and progressive gaze restriction. Some patients have vitamin E deficiency without fat malabsorption, inherited in an autosomal recessive manner, with symptoms similar to Freidreich s ataxia. Botulism Signs of total (internal and external) ophthalmoplegia, dry mouth, descending paralysis, obstipation, absence of fever, and lucid sensorium, as cardinal symptoms should raise suspicion of botulism. Clostridium botulinum produces seven distinct protein toxins, of which A, B, and E are the most commonly responsible for human botulism. 815 The botulism toxin causes neural paralysis of skeletal muscle by disruption of both spontaneous and stimulus-induced release of acetylcholine from the presynaptic nerve terminal and by interfering with exocytosis of vesicle contents. The toxin enters the body via the following four routes: (1) ingestion of preformed toxin, as in the case of food poisoning; (2) toxin production by Clostridium spores or bacteria infecting a wound; (3) colonization of the gastrointestinal tract by Clostridium botulinum, with subsequent production of toxin; it is by this mechanism that infant botulism and the infant form of botulism that affects some adults occurs; and (4) the hidden form in which no identifiable route of entry of toxin or bacteria into the body can be identified. Botulism spores are ubiquitous, and have been isolated from yard and houseplant soil and vacuum cleaner dus. 38 In adults, the disease usually results from ingestion of preformed botulinum toxin in contaminated foods such as poorly sterilized canned goods or raw fish. Infant botulism, first described in 1976, 619 is caused by ingestion of live Clostridium botulinum organisms or spores, which subsequently release toxin in the intestine. Most pediatricians advise against feeding honey to infants younger than 1 year of age, as it has been documented as a source of spores causing infant botulism. 38,745 Infant botulism was first recognized in 1976 and has since become the most frequently reported form of botulism. 684,689,804 Most reported cases have been from North America. A history of honey ingestion or soil eating is frequently obtained. Because Clostridium botulinum is ubiquitous and is commonly ingested without adverse effects, a number of host factors may render a particular individual predisposed. Constipation, immune dysfunction, gastric ph, and unusual gut flora, among others, may permit the colonization and germination of Clostridium spores and subsequent local production of toxin. Infant botulism has been described in infants ranging from 10 days to 8.5 months old, but 98% of those affected are between 2 weeks and 6 months old. 35 The earliest clinical sign of disease is usually constipation, which can be present for several days before other signs appear. 34,745 Bulbar signs later predominate and include impaired sucking with poor feeding, a feeble cry, tachycardia due to loss of vagal tone. 34 Isolated internal ophthalmoplegia may evolve into diffuse ophthalmoplegia with ptosis. Crouch et al reported an infant who had bilateral sixth nerve palsies following resolution of infant botulinum. 192 Other signs of cranial nerve palsy include drooling, diminished gag reflex, and facial weakness. Generalized muscular weakness progresses in a descending fashion from the cranial nerves to the limbs. Generalized weakness, hypotonia, hyporeflexia, and cranial nerve weakness constitute the floppy infant syndrome. Respiratory arrest and death may follow, but most infants recover completely in 1 5 months. While mild cases are often managed on an outpatient basis, the age distribution of infant botulism closely mirrors that of the Sudden Infant Death syndrome (SIDS), and botulism toxin has been recovered from 5% of autopsy specimens of infants with the diagnosis of SIDS. 37 Cases of infant botulism may also be mistakenly diagnosed as crib death, failure to thrive, sepsis, dehydration, viral infection, idiopathic hypotonia, poliomyelitis, meningitis, brainstem encephalitis, or other neuromuscular disorders such as myasthenia. To add to the diagnostic quandary, some patients with botulism may give false-positive response to intravenous edrophonium chloride. 190 One important differentiating feature of botulism is the dilated, poorly reactive pupils, which are not seen in ocular myasthenia. The differential diagnosis also includes Reye s syndrome, Guillain Barré syndrome, hypothyroidism, tick paralysis, and toxins. The diagnosis is made when toxin or organisms are recovered from stool samples. However, the level of botulinum toxin is too low to be detected in the sera of patients with infant botulism. Electromyographic (EMG) studies are highly useful in the diagnosis of botulism, with a characteristic EMG pattern that has been given the acronym BSAP, for brief duration, small-amplitude, overly abundant, motorunit action potentials. Fibrillation potentials suggesting functional denervation are observed in about half the cases. Repetitive fast rates of stimulation (20 50 Hz) result in marked incremental response, a finding not present in older
29 Olivopontocerebellar Atrophy children and adults with botulism, probably due to the large amount of toxin present. Other conditions showing an incremental EMG response, such as Eaton Lambert (not seen in infants), hypermagnesemia, and aminoglycoside toxicity, can be readily excluded on clinical and laboratory grounds. Logistical problems in terms of application of EMG to the investigation of infant botulism include the need to transport the cumbersome equipment to the intensive care unit and electrical interference there with the EMG recording. Spontaneous recovery occurs through sprouting of new nerve endings with formation of new myoneural junctions. Treatment of affected patients includes insertion of a nasogastric tube with suction, enemas, antitoxin administration (benefit controversial), and mechanical ventilation, if indicated. The recently introduced botulinum immune globulin (BIG) appears promising. In a recent clinical trial, it reduced the severity of illness and reduced the mean length of hospital stay from 5.7 to 2.6 weeks without complications. 39 Fisher Syndrome: A Variant of Guillain Barré Syndrome The Guillain Barré syndrome (acute infectious polyneuropathy) consists of progressive, usually symmetric muscular weakness that appears several days after a nonspecific infectious prodrome. Mild sensory disturbances, such as pain and parasthesias, are commonly present. The paralysis usually affects the lower extremities and then ascends. Cranial nerve palsy may appear at any time during the clinical course. Results of EMG studies are usually consistent with involvement of the lower motor neurons or peripheral nerves. Fisher syndrome is a variant of Guillain Barré syndrome with the distinct triad of ataxia, areflexia, and ophthalmoplegia without concurrent peripheral neuropathy. Fisher syndrome constitutes about 5% of all reported cases of Guillain Barré syndrome. There appears to be a gender predilection, with the male/female ratio being 2:1. Of the 223 cases reported before 1993, the average age was 43.6 years (range, 14 months to 80 years), with 14.3% of these being children. 66 Most patients with Fisher syndrome suffer a preceding viral prodrome, usually respiratory, 1 3 weeks prior to onset of the syndrome. 66 Most patients reach maximum neurologic deficit within 1 week of onset. Diplopia and ataxia are the most common presenting symptoms. Associated ophthalmoplegia is complete (including the parasympathetic fibers to the pupillary sphincter muscle) in about half of patients with Fisher syndrome, 66 but pure external ophthalmoplegia or internal ophthalmoplegia may occur. Isolated ocular motor nerve palsy and combinations of horizontal and vertical gaze palsies may be noted. Internuclear ophthalmoplegia, oneand-a-half syndrome, gaze-evoked lid nystagmus, convergence 337 spasm, and a dorsal midbrain syndrome with upward gaze paralysis, but intact Bell s phenomenon, have also been reported. 16,66,529 The presence of these latter disorders and similar findings pointing to brainstem involvement fuels the controversy about the nature of the disease as a peripheral (infranuclear, due to involvement of sensory fibers in the peripheral nerves and dorsal roots) or CNS (supranuclear) disorder or as a combination of both. 529 Other cranial nerves may be affected, the most common being the facial nerve. Dysphasia and dysarthria may result when the lower cranial nerves are involved. Monoparesis, hemiparesis, or quadraparesis have been described. Patients may have sensory symptoms, including parasthesias, dysesthesia, and headaches. Other symptoms and signs include disturbance of consciousness, seizures, myoclonus, tremors, fever, vomiting, irritability, positive Babinski sign, and respiratory insufficiency. The CSF, if examined 2 3 weeks or later after onset, usually shows mild elevation in protein, with about 10% of cases in the literature showing pleocytosis. Some authors consider the Fisher variant to be due to pathologic changes exclusively within the peripheral nervous system. This concept is supported by reports of normal MR imaging scans in affected patients. 655 While pure cases of Fisher syndrome are apparently attributable to peripheral neuropathy and are not uncommon, overlapping cases occur that bridge the spectrum from the benign Fisher variant to the more virulent Guillain Barré syndrome. For instance, cases that include limb paralysis do not, strictly speaking, fall within the original definition of the Fisher syndrome and are transitional forms to the Guillain Barré syndrome. On the basis of current evidence, some authors believe that Fisher syndrome represents an encephalomyeloneuritis. 66 The triad of severe external ophthalmoplegia, ataxia, and areflexia in an otherwise alert child is fairly unique. The characteristic history and physical findings are usually sufficient to establish the presumptive diagnosis, but differentiation from posterior fossa tumors may be difficult without neuroimaging. Wernicke syndrome and phenytoin intoxication can show a similar syndrome complex, but these entities can be excluded by the clinical history. Both Fisher syndrome and botulism may show a similar early presentation consisting of extraocular muscle weakness and mydriasis. Other conditions included in the differential diagnosis of Fisher syndrome are brainstem stroke, pituitary apoplexy, cerebral sinus thrombosis, tick fever, and diphtheria. While some cases may follow a more virulent course, most cases of Fisher syndrome resolve spontaneously within 1 3 months. Corticosteroids as well as plasmapheresis have been used, but there is little evidence to support their efficacy in the limited form of the condition without brainstem signs. Intravenous immunoglobulin may be helpful in severe cases. 30
30 338 7 Complex Ocular Motor Disorders in Children Serological, immunological, and pathological studies all implicated a role for complement-mediated damage to neuronal and Schwann cell membranes in Guillain Barré syndromes. 329 In vivo and in vitro models of Fisher syndrome have shown that anti-gq1b ganglioside antibodies target the presynaptic motor nerve terminal axon and surrounding peripsynaptic Schwann cells, thereby mediating destructive injury through deposition of membrane attack complex (MAC). The anti-gq1b antibody has been detected in the sera from patients during the acute phases of Fisher syndrome, Bickerstaff brainstem encephalitis, and Guillain Barré syndrome with ophthalmoparesis. 828 These cases seem to demonstrate a close relationship between ophthalmoparesis and serum anti-gq1b antibody. 159 It has been hypothesized that ganglioside epitopes on Campylobacter jejuni are the key to the development and characterization of Guillain Barré syndrome. 446 The presence of these bacterial epitopes in neuropathy patients correspond to autoantibody reactivity. In one study, patients infected with C. jejuni (Asn51) regularly expressed the GQ1b epitope (83%), whereas those with cst-ii (Thr51) had the GM1 (92%) and GD1a epitopes. Patients infected with C. jejuni (Asn51) more often were positive for anti-gq1b IgG (56% vs. 8%), and had ophthalmoparesis (64% vs. 13%) and ataxia (42% vs. 11%). Patients who had C. jejuni (Thr51) more frequently were positive for anti-gm1 (88% vs. 35%) and anti-gd1a IgG (52% vs. 24%) and had limb weakness. Thus, the genetic polymorphism of C. jejuni appears to determine autoantibody reactivity as well as the clinical presentation of Guillain Barré syndrome, possibly through modification of the host-mimicking molecule. Bickerstaff Brainstem Encephalitis Bickerstaff s brainstem encephalitis is a clinical syndrome of ophthalmoplegia, cerebellar ataxia, and central nervous system signs. 16,72 Diagnostic criteria include a progressive, relatively symmetric external ophthalmoplegia and ataxia with disturbance of consciousness or hyperreflexia. 724 Positive anti- GQ1b antibodies support the diagnosis. 587,724 Conditions that mimic the disorder, including brainstem tumors, must be excluded. There is a clinical continuum between Bickerstaff brainstem encephalitis and Fisher syndrome. 724 However, CNS signs, such as disturbance of consciousness and cogwheel rigidity, are more in keeping with brainstem encephalitis. In general, Bickerstaff brainstem encephalitis is a disease of the CNS, while Fisher syndrome is primarily a disease of the peripheral nerves. 587 However, some clinical and pathologic overlap is found. It is interesting to note, for example, that one in three patients in the original description of Fisher syndrome had drowsiness, whereas Bickerstaff described four cases of Bickerstaff brainstem encephalitis in which the patients had areflexia. Both conditions have been described following Mycoplasma pneumoniae infection. 724 Treatment with intravenous immunoglobulin or use of plasmapharesis has been reported to produce clinical improvement, although it is unproven that they affect the natural history of this disorder. Tick Paralysis Tick paralysis should be considered in the differential diagnosis of Fisher syndrome in children in tick-infested areas who develop ophthalmoplegia and ascending paralysis. 149 The diagnosis is confirmed by finding a tick embedded in the skin and observing for signs of improvement after tick removal. No other tests for confirming tick paralysis exist. Tick paralysis is thought to be caused by a toxin secreted in tick saliva that reduces motor neuron action potentials and the action of acetylcholine. 263,307 Symptoms usually begin 4 7 days after tick feeding. Ascending flaccid paralysis progresses over several hours or days; sensory loss does not usually occur, and pain is absent. 714 When the tick is not removed, the mortality rate resulting from respiratory paralysis is approximately 10%. 575,686 Wernicke Encephalopathy Although classically considered an adult disorder, in 2003, an epidemic of Wernicke encephalopathy developed in Israeli infants fed a thiamine-deficient soy-based formula. 427 These infants showed no neurologic signs for the first 6 months on the thiamine-deficient diet. 627 Apathy, vomiting, and diffuse ophthalmoplegia brought these infants to medical attention and helped to establish the diagnosis. Wernicke encephalopathy preferentially affects neurons in particular areas of the nervous system such as the putamen, parts of the thalamus, and the periaqueductal gray matter. Thiamine turnover is higher in these areas and these structures have high rates of oxidative metabolism in neonates, which could make them more vulnerable. 748 Miscellaneous Causes of Ophthalmoplegia Involvement of the respiratory and renal systems in the child with diffuse ophthalmoplegia should suggest the diagnosis of Wegener granulomatosis. 488 A new autosomal recessive ophthalmoplegic disorder affecting highly inbred Arab families in Israel produces limited upgaze, slow saccades, and impaired
31 Transient Ocular Motor Disturbances of Infancy 339 Fig Transient esotropia in neonate. Infant showed 30 prism diopters of left esotropia (a) with mild limitation of abduction of left eye that totally resolved by 5 months of age (b). Estropia was caused by perinatal trauma to abducens nerve convergence without ptosis or pupillary abnormalities. In addition, there is involvement of the face, orbicularis, and neck muscles, giving the appearance of a long, thin face. Onset is probably in childhood. Linkage studies identified markers on chromosome 17p13.1-p12, an interval to which several genes for sarcomeric heavy chain map. 500 Transient Ocular Motor Disturbances of Infancy Healthy neonates may exhibit a variety of benign, transient supranuclear eye movement disturbances. These include horizontal heterophorias, tonic downgaze and upgaze, opsoclonus, skew deviations, and transient idiopathic nystagmus in infants. As each of these disorders may forebode serious neurological disease, especially in older children, their benign nature in healthy neonates must be recognized to avoid unnecessary diagnostic testing. Transient Neonatal Strabismus Several nursery studies have shown that the eyes of otherwise healthy, full-term neonates are commonly misaligned. In one study of 1,219 neonates, % had orthotropia, 32.7% had exotropia, 3.2% had esotropia, and 15.4% were not sufficiently alert to allow determination of ocular alignment. No cases of congenital esotropia were found in any neonate, supporting the concept that congenital esotropia does not manifest at birth. Follow-up studies of these patients have shown that the vast majority of children with transient heterophorias become orthotropic between 2 and 3 months of age, a stage coincident with development of stereoscopic vision. 31,712 Unlike exotropia, which may be found transiently in the neonatal period, the finding of large esotropia in the first several weeks of life should not be classified as congenital esotropia (which usually first appears after 6 weeks of age) until other conditions such as sixth nerve palsy and Möbius syndrome are ruled out (Fig. 7.12). It should also be recognized that the eyes of premature infants may transiently display exotropia with limited adduction. These findings have been speculated to result from immaturity of the medial longitudinal fasciculus in the premature infant. Large convergent movements of the eyes, producing esotropia, are often observed in the first 2 months of life. 374,375 These convergent eye movements resolve spontaneously, giving way to normal ocular alignment. The fact that these large-angle convergent eye movements are predictive of the normal binocular alignment 374,375 belies the notion that infantile esotropia results from excessive convergence. 112 Transient Idiopathic Nystagmus Good et al described six infants with transient idiopathic nystagmus. Four of these infants had other visual abnormalities, including regressed retinopathy of prematurity, coloboma, and delayed visual maturation, which may have precipitated the nystagmus. They interpreted the transient nystagmus as indicative of a fragile period of postnatal maturation of the ocular motor system. 304 It is also recognized that some infants with delayed visual maturation may exhibit transient nystagmus. 71 Tonic Downgaze Tonic downward deviation of the eyes may occur as a transient phenomenon in neonates and does not necessarily indicate underlying neurologic dysfunction. 275,761,813 Both eyes are tonically deviated downward while the infant is
32 340 7 Complex Ocular Motor Disorders in Children awake, but can be maneuvered upward with oculocephalic or vestibulo-ocular stimulation; the condition resolves during sleep, with both eyes returning to the midline. It usually resolves within the first 6 months of life. Two infants who displayed this transient tonic downgaze also showed associated upbeat nystagmus, with complete resolution of both findings within the first few months of life. 295 The authors proposed that immaturity of the vestibular system was the cause. In some cases, these transient episodes of downgaze can be followed by abnormal body movements. 813 The benign, transient form of tonic downgaze differs from the setting sun sign associated with hydrocephalus by the absence of eyelid retraction. Yokochi 825 described downward deviations of the eyes of neurologically affected infants that were paroxysmal rather than constant, as in the foregoing benign variety. The paroxysms were not associated with seizure activity. He considered this to be a sign found in braindamaged infants with cortical visual impairment. The paroxysms spontaneously resolved with time in many patients. We examined a blind infant with profound bilateral optic nerve hypoplasia, absent septum pellucidum, and developmental delay who showed sudden episodic downward deviations of the eyes occurring every 1 2 min and lasting 5 10 s (Fig. 7.13). The downward deviation was associated with lid lag. The patient also showed intermittent side-to-side head shaking. Episodic downgaze may be one of the presenting signs of Leigh subacute necrotizing encephalomyelopathy. 511 Mak et al 511 reported a previously healthy infant who, at 6 months of age, showed episodic downgaze with limited horizontal movements that resolved after 5 days, then recurred several times along with other abnormalities before a diagnosis of Leigh disease was made. It is not clear whether congenital hydrocephalus alone results in tonic downgaze. The setting sun sign, a feature of congenital hydrocephalus (Fig. 7.14), may be considered a Fig Episodic tonic downgaze. Blind infant with profound bilateral optic nerve hypoplasia, absent septum pellucidum, and developmental delay showed sudden episodic downward deviations of eyes occurring every 1 2 min and lasting 5 10 s. These have occurred since birth. Downward deviation was associated with lid lag. Infant also showed intermittent side-to-side head shaking Fig Setting sun sign in hydrocephalus. Child presented with pronounced lid retraction and mild to moderate tonic downward deviation of eyes. Disproportionately large head and dilated ventricles on neuroimaging confirmed diagnosis of hydrocephalus combination of lid retraction (Collier s sign) and tonic downgaze. Acquired hydrocephalus in older patients does not produce tonic downgaze, suggesting a specific susceptibility of the neonatal brain to the mass effect of hydrocephalus on midbrain structures responsible for downgaze or suggesting a higher sensitivity of the pretectal area in infants to hydrocephalus, leading to more severe upgaze palsy (causing the eyes to deviate downward) than is seen in older patients. Tamura and Hoyt 735 reported 11 premature infants with intraventricular hemorrhages who showed acute tonic downward deviation of the eyes, esotropia, and upgaze palsy. All patients showed associated hydrocephalus, shunting of which resulted in gradual improvement in upgaze, with persistence of the large-angle esotropia. The authors suggested that the gradual recovery of upgaze indicates that the upgaze palsy may not be simply due to the acute effects of the hydrocephalus. Rather, they suggested that the associated intraventricular hemorrhages act as a mass lesion, compressing (and hence paralyzing) the upgaze centers or irritating (and hence stimulating) the downgaze centers within the mesencephalon. They speculated that injury to these mesencephalic structures may contribute to the delay in recovery of upgaze after shunting. We most commonly see tonic downgaze in conjunction with esotropia in infants with periventricular leukomalacia, where it can occur in the absence of intraventricular hemorrhage. 118 As with intraventricular hemorrhage, the tonic downgaze spontaneously resolves, and infants are usually left with an A-pattern esotropia with superior oblique muscle overaction. The telltale ocular intorsion can easily be missed if the retinas are examined in the esotropic position, which places the eyes in the vertical rather than the torsional field of action of the overacting superior oblique muscles. We have proposed that a posterior canal predominance caused by injury to bilateral central brainstem pathways mediating upward pitch could explain this constellation of
33 Transient Ocular Motor Disturbances of Infancy finding. 115 Neuroimaging often shows associated thalamic injury which may explain this combination of findings. 118 In congenital hydrocephalus that is not caused by intraventricular hemorrhage, however, the upgaze palsy and/or the setting sun sign responds quickly to reduction of intracranial pressure. For instance, Hoyt and Daroff 379 described a 3-month-old infant with intermittent hydrocephalus secondary to a tumor of the thalamus and septum pellucidum. The infant displayed tonic downgaze and esotropia whenever the ventricular pressure increased; eye movements normalized when the ventricular pressure became normal. Such cases demonstrate that a mass effect, independent of the effect of the hydrocephalus itself, need not be present for tonic downgaze to develop in infants with hydrocephalus. Children with hydrocephalus are predisposed to esotropia through several different mechanisms: (1) early-onset childhood esotropia, which is more common in neurologically impaired children; (2) unilateral or bilateral sixth nerve palsy; and (3) intraventricular hemorrhage in premature infants, which may involve the thalamus and mesencephalon and produce neuro-ophthalmologic signs of a thalamic infarction (tonic downward deviation of the eyes, upgaze palsy, and esotropia). The esotropia in these patients usually persists even after the vertical gaze deficits resolve. Visually significant upgaze limitation can be relieved with bilateral inferior rectus recessions in Parinaud s syndrome. 128 Tonic downgaze may be observed in very ill patients with impaired consciousness who have medial thalamic hemorrhage, severe encephalopathy, acute obstructive hydrocephalus, or severe subarachnoid hemorrhage. In this setting, it should be distinguished from V-pattern pseudobobbing, wherein the eyes show an abrupt downward jerk followed by a slow, upward drift to primary position. Keane 420 reported this finding in five patients with acute obstructive hydrocephalus who had arrhythmic, repetitive, downward and inward ocular deviations at a rate of 1 per 3 s to 2 per second. The fast downward movements render the condition similar to ocular bobbing, but it differs by the presence of a V pattern, a generally faster rate, and associated pretectal, rather than pontine, signs. Keane speculated that V-pattern pseudobobbing represents a variant of convergence-retraction nystagmus that signals the need for prompt shunt placement or revision. Tonic Upgaze Tonic upgaze is less common than tonic downgaze. It also tends to be more episodic. In 1988, Ouvrier and Billson 605 described four patients with a new condition they termed benign paroxysmal tonic upgaze of childhood. This condition was characterized by onset during infancy of episodic 341 tonic conjugate upward deviation of the eyes that was relieved by sleep. The children had impaired downgaze below the primary position, with downbeating nystagmus on attempted downgaze and apparently normal horizontal movements. The patients were otherwise neurologically intact, with the exception of mild ataxia. Results of metabolic, electroencephalographic (EEG), and neuroradiologic investigations were unremarkable. All eventually improved, with one child showing a favorable therapeutic response to levodopa. The following year, Ahn et al 11 described three infants who had tonic upgaze with no associated seizure activity or neurologic disease. The vestibulo-ocular reflexes were intact, revealing a full range of vertical movements. These episodes were initially noted in the first month or two of life and were most conspicuous when the infant was ill or fatigued. These episodes diminished with time. 11 Mets 532 described a 9-month-old otherwise healthy infant with largeangle esotropia who displayed extreme sustained spasms of upgaze. This infant was noted to have full vertical range of ocular motion at times and could fixate in primary gaze. No EEG abnormality was found. These episodes resolved at age 3% months, but the esotropia with associated amblyopia required patching therapy and strabismus surgery. Tonic upgaze is not an epileptic phenomenon, and paroxysmal tonic upgaze may be exacerbated by treating coincident epilepsy with valproate. 502 Phenomenologically, benign paroxysmal upgaze and congenital downbeat nystagmus are closely related conditions. Congenital downbeat nystagmus is distinguishable only by the presence of corrective downward refixational saccades. 115a It is likely that benign hereditary downbeat nystagmus and the syndrome of benign tonic upward deviation of the eyes with ataxia are variants of the same disorder. In several affected children, tonic upgaze has evolved into downbeating nystagmus. 605,605a We have proposed that, tonic upgaze an anterior canal predominance. 115,116 Tonic upgaze is difficult to distinguish from the overlooking that was reported by Taylor 741 in patients with neuronal ceroid lipofuscinosis. Instead of looking at the object of regard directly, affected children look above the object. Initially attributed to relative preservation of the inferior visual field associated with certain retinal disorders, it was later reported not to be disease-specific, but rather to represent a sign of bilateral central scotomas (and vision of 20/200 or worse) in children from a variety of causes. 303 Recently, Cruysberg reported that some patients with overlooking display a tonic upgaze and a dystonic neck extension, the two conditions are not always distinct. 194 When not associated with overlooking, tonic upgaze is more often associated with neck flexion than neck extension. 502 Intermittent episodes of upward ocular deviation may be a manifestation of oculogyric crisis or of a seizure disorder,
34 342 7 Complex Ocular Motor Disorders in Children typically petit mal. Oculogyric crisis denotes an extreme, episodic upward rotation of the eyes, often obliquely to the right or to the left. The deviation is usually sustained and is often associated with rhythmical jerking or twitching of the eyelid. Each oculogyric movement lasts several seconds, after which the eyes return to the horizontal position before deviating again a few seconds later until the crisis passes, typically in 1 or 2 h. Patients may have associated thought disorders. Dopamine-blocking agents (neuroleptics) are the most common cause of drug-induced acute dystonic reactions such as oculogyric crisis, but other drugs have been incriminated, including carbamazepine, lithium carbonate, metoclopramide, and sulpiride, among others. Oculogyric crisis has also been reported after Tensilon administration. 586 Similar eye movements have been observed in patients with various CNS disorders, such as herpetic encephalitis, pantothenate kinase-associated neurodegeneration, and Rett syndrome, 270 and in one patient with cystic glioma in whom the onset of crisis was positional. 722 Oculogyric crisis can be aborted with use of anticholinergics (e.g., Cogentin) and subsequently controlled by reducing the dosage of the offending medication or changing it altogether. Patients with petit mal seizures may show eye movements similar to oculogyric crisis, usually with concurrent eyelid flutter. 28 Eye movement tics (see later in this chapter) may also superficially resemble intermittent episodic tonic upgaze. Barontini 56 concluded that either dystonia or downgaze palsy can underlie the phenomenon of tonic upgaze. Acquired tonic upward deviation is also seen in comatose patients, wherein it indicates a very poor prognosis, or in patients with brainstem disease causing downgaze paralysis. Cherubism, a rare inherited disorder that involves the facial bones and produces facial fullness because of boney enlargement occurring between 2 and 4 years of age should also be considered in the differential diagnosis of tonic upgaze. 273 This disorder was so named by Jones in 1933 because the marked fullness of the jaws and cheeks and the upward displacement of the eyes in patients were thought to resemble the heavenward gaze and round faces of Renaissance cherubs. 404 Although this lesion was originally considered to be a form of fibrous dysplasia, cherubism was eventually accepted as a clinicopathologic entity, with features identical to giant cell reparative granuloma. 161 This condition can be complicated by proptosis and optic neuropathy. 358 Neonatal Opsoclonus Opsoclonus denotes bursts of chaotic repetitive back-to-back saccades in different directions (see Chap. 8). Although usually indicative of an underlying viral encephalitis or neuroblastoma, opsoclonus has been reported as a benign transient finding in healthy neonates. It usually resolves in 1 3 months, passing through a phase of ocular flutter. Given the apparent rarity of this observation, it is probably prudent to consider this benign transient variant a diagnosis of exclusion, especially in light of the serious nature of other potentially causative lesions such as neuroblastoma. When opsoclonus accompanies neuroblastoma, it imparts a poor long-term neurological prognosis, 551 but a highly favorable prognosis for survival. 572 Transient Vertical Strabismus in Infancy A transient vertical deviation of the eyes with or without a horizontal component has been reported in the neonatal period without associated evidence of posterior fossa dysfunction and is referred to as skew deviation. 381 Some infants exhibiting this sign may subsequently develop large-angle esotropia typical for congenital esotropia or nystagmus compensation syndrome. 381 Whether this transient vertical deviation of the eyes is an early manifestation of dissociated vertical divergence and whether it is a reliable premonitory sign for the subsequent development of congenital esotropia is not known. Congenital Cranial Dysinnervation Syndromes The congenital cranial dysinnervation syndromes comprise a group of disorders characterized by deficient innervation to the extraocular muscles and facial musculature. 321,621,750 These congenital neuromuscular disorders result from developmental errors in innervation of the ocular and facial muscles. 821 They result from mutations in a number of genes, including ROBO3, 400 PHOX2A, 573 SALL4, 14 HOXA1, 747 and KIF21A, 821 that are essential to the normal development of brainstem motor neurons or axons. 87 Recognized disorders congenital ptosis, congenital fibrosis of the extraocular muscles, Duane s syndrome and its variants, Möbius sequence, horizontal gaze palsy with progressive scoliosis, and congenital facial palsy. 321 They present with varying degrees of ptosis and ophthalmoplegia from birth, together with signs of aberrant innervation. Many of these disorders were previously considered to be myopathic in origin. However, the congenital cranial dysinnervation disorders also include sensory disorders due to trigeminal nerve maldevelopment that cause congenital corneal anesthesia.
35 Congenital Cranial Dysinnervation Syndromes Congenital Ptosis Our long-standing understanding of congenital ptosis as a primary myopathy has been overturned, as clinical signs of congenital cranial dysinnervation have been demonstrated. 106 The notion of congenital ptosis as a primary myopathy was first challenged in 1996 when Steel and Harrad found an unrecognized form of oculomotor synkinesis in 44% of patients with unilateral congenital ptosis. 723 Affected patients displayed excessive elevation of the ipsilateral eye beneath the ptotic eyelid in upgaze, but no vertical deviation in primary gaze. This upshoot disappeared when the ptotic eyelid was manually elevated. The authors insightfully attributed this phenomenon to a misdirection of levator innervation to the ipsilateral superior rectus muscle. They proposed that manual elevation of the ptotic eyelid reduced innervation to the levator muscles, thereby extinguishing the aberrant superior rectus innervation. In 2000, Harrad and Shuttleworth 352 described another group of patients with long-standing unilateral ptosis that resolved in upgaze and increased in downgaze, suggesting that superior rectus innervation had become misdirected to a paretic levator muscle. These clinical observations suggest that prenatal and postnatal routing of the superior division of the oculomotor nerve is a delicate process that may be subject to directional modification, perhaps on the basis of the relative innervational recruitment by its two target muscles. 106 For reasons that are unclear, an inverse Bell s phenomenon (unilateral or bilateral) is occasionally observed postoperatively following levator resection. This phenomenon reverts to normal within 2 weeks. 68 Congenital ptosis may be associated with other forms of ocular motor synkinesis. Khan et al 429 described a large family in which two siblings exhibited ptosis with abnormal synkinetic elevation on ipsilateral abduction. One was bilaterally affected, while the other had unilateral findings. A third demonstrated classic bilateral congenital ptosis, while a fourth demonstrated Duane s syndrome. Its association with congenital fibrosis syndrome is detailed below. Indeed, congenital ptosis can now be viewed as a limited form of congenital fibrosis syndrome selectively affecting the superior division of the third nerve. Several genes for isolated congenital ptosis have now been identified. 248,527 Marcus Gunn Jaw Winking (Trigemino-Oculomotor Synkinesis) A normal synkinesis denotes simultaneous contraction of muscles normally innervated by different peripheral nerves or 343 different branches of the same nerve. A pathologic synkinesis occurs when muscles are reinnervated by nerves other than their own following a nerve injury. Pathologic synkinesis may be congenital (Duane s syndrome, synergistic divergence, Marcus Gunn jaw winking) or acquired (facial synkinesis or oculomotor synkinesis after trauma). The Marcus Gunn jaw winking (MGJW) synkinesis usually presents as variable unilateral ptosis noted at birth or shortly thereafter. Unlike ordinary congenital ptosis, when the infant nurses, the ptotic lid jerks upward with each suckling movement. In a series of nearly 1,500 cases of congenital ptosis, 70 the Marcus Gunn phenomenon accounted for 80 patients (5%). Of these 80 patients, 42 showed right eye involvement, 35 showed left eye involvement, and three had bilateral synkinesis. No gender preponderance was identified, and only two cases were familial. Fifty-four percent of patients showed amblyopia, and 26% showed anisometropia. Some form of strabismus was found in 56%, including 19 cases of superior rectus palsy, 19 cases of double elevator palsy, and two cases of Duane s retraction syndrome. The natural history of the disorder remains unsettled, with some authors noting that the synkinetic movement becomes less conspicuous with age, although patients may learn to camouflage the lid excursions. The pathogenesis of the MGJW synkinesis is controversial. The prevalent concept is that the disorder results from aberrant innervation of the levator palpebrae muscle by a branch of the motor division of the trigeminal nerve that supplies the muscles of mastication, hence the designation trigemino-oculomotor synkinesis. However, Sano 672 has presented EMG evidence that the jaw winking phenomenon is a release phenomenon representing an exaggeration of a normally found but clinically undetectable physiologic cocontraction. Utilizing EMG studies of normal subjects, he demonstrated cofiring of the oculomotor-innervated extraocular muscles and the muscles of mastication innervated by the motor branch of the trigeminal nerve. He argues that congenital brainstem lesions may release phylogenetically older neural mechanisms, such as synkinetic movements from higher central control, which are similar to other synkinetic movements such as the palmomental and primitive grasp-feeding reflexes. Muller s muscle is doubly innervated by an efferent sympathetic nerve and an afferent proprioceptive nerve. 828a Muller s muscle acts as a serial muscle spindle (similar to that seen in jaw closing periodontal receptors). 828a Stretching of Muller s muscle has been found to produce involuntary levator muscle contraction (the Hoffman reflex). These proprioceptive fibers function as mechanoreceptors to induce reflex contracture of the levator muscle against gravity as a type of length servomechanism. The central modulation of these levator and jaw closing pathways by motor trigeminal
36 344 7 Complex Ocular Motor Disorders in Children nucleus may give rise to the innervational crossover that manifests in the form of Marcus Gunn jaw winking. 53a Sano 672 has classified the trigemino-ocular motor synkinesis into two major groups: (1) external pterygoid-levator synkinesis (the most common type) with lid elevation upon thrusting the jaw to the opposite side (ipsilateral external pterygoid firing), upon projecting the jaw forward (both external pterygoids firing), or upon opening the mouth widely; and (2) internal pterygoid-levator synkinesis (relatively rare) with lid elevation upon teeth clinching. In the typical case of MGJW, firing of the motor branch of the trigeminal nerve is associated with firing of the oculomotor branch to the levator. A rare variant of this phenomenon, termed the inverse Marcus Gunn phenomenon, 504 shows firing of the motor branch of the trigeminal nerve synkinetically, with inhibition of the oculomotor branch to the levator. Here, the affected eyelid falls as the mouth opens or as the jaw moves to the opposite side, without associated activity of the orbicularis oculi. Pathologic synkineses may cluster together. For instance, the MGJW synkinesis (congenital trigemino-oculomotor synkinesis) has been reported in association with Duane s syndrome (congenital oculomotor-abducens synkinesis) and with synergistic divergence. 120,337 This association generally occurs in the setting of congenital fibrosis syndrome. 104,120,337 Other forms of ocular motor synkinesis have also been reported in patients with Marcus Gunn jaw winking. Eye bobbing was attributed to coactivation of the superior rectus muscle in one patient with Marcus Gunn jaw winking. 603 Trigemino-abducens synkinesis 444,466 and pseudo-inferior oblique muscle overaction have also been described. 387 A frontalis suspension operation is usually recommended for cases severe enough to come to surgery. Some controversy exists regarding whether to disinsert the ipsilateral levator and whether to also disinsert the contralateral levator and perform a frontalis suspension on the opposite side to achieve a greater degree of symmetry. Congenital Fibrosis Syndrome Congenital fibrosis of the extraocular muscles (CFEOM) is characterized by the presence of congenital restrictive ophthalmoplegia, fixed downgaze, horizontal strabismus, ptosis, and a compensatory backward tilting of the head. 751 In this disorder, the extraocular muscles and the levator muscles are replaced by fibrous tissue to a variable extent. 350 The disorder may be unilateral or bilateral and may be clinically limited to specific muscles in a given individual. The resultant ocular motility deficits depend on which of the extraocular muscles are involved. The inferior rectus muscle is most commonly involved, followed by the levator muscle and the lateral rectus muscle. In the presence of ptosis, inferior and lateral rectus muscle involvement may mimic unilateral or bilateral congenital oculomotor palsy. Rarely, all of the extraocular muscles, including the levator, are affected (generalized fibrosis syndrome). Historically, investigators have divided congenital ocular fibrosis into different subtypes that include congenital fibrosis of the inferior rectus with ptosis, strabismus fixus, congenital unilateral enophthalmos with ocular muscle fibrosis and ptosis, 362 and the vertical retraction syndrome. Fixed downgaze and associated ptosis with sparing of pupillary function and similar findings in other family members provide critical clues to the diagnosis. Patients with CFEOM may show rapid convergent movements of the eyes on attempted upgaze, simulating convergence retraction nystagmus. 104 The diagnosis of CFEOM should therefore be suspected when convergence-retraction nystagmus is accompanied by ptosis rather than lid retraction. Although this phenomenon could theoretically result from the mechanical tethering effect of tight inferior rectus muscles in attempted upgaze, the fact that these convergent movements persist following recession of the inferior rectus muscles suggests that they arise from a supranuclear deficiency of elevation, as suggested by several investigators. 1,78,168,262 CFEOM has traditionally been regarded as a primary myopathy localized to the extraocular muscles on the basis of biopsy studies of affected muscles. Both light and electron microscopy studies show replacement of extraocular muscles by dense fibrous connective tissue and collagen. 350 Histology of an affected levator showed reduction or absence of striations and Z bands and vacuolation of muscle cells. Muller muscle fibers appeared intact. Anomalous insertions of affected muscles may occur, presumably resulting from a maturational defect at or before the seventh week of gestation. 29 Involved eyes may also have anomalous adhesions between the muscles, Tenon s capsule and globe, and inelasticity or fragility of the conjunctiva so that conjunctival recession may also be necessary. 266,487 Like in Duane syndrome, the involved extraocular muscles in congenital fibrosis syndrome are simultaneously tight and hypoplastic. Affected muscles often appear small and atrophic on orbital imaging studies. 805 On the basis of the presence of multiple synkinetic eye movements in a patient with CFEOM, Brodsky et al, in 1989, proposed that is caused by failure of normal neuronal connections with the extraocular muscles to become established early in embryogenesis. 120 Assaf independently concluded that congenital fibrosis syndrome must arise from a CNS abnormality. 44,45 Our subsequent report of additional patients with CFEOM, who displayed both synergistic divergence and MGJW, 120,337 provided clear evidence that congenital fibrosis of the extraocular muscles can result from the absence of normal innervation to orbital striated muscles early in development (Fig. 7.15). Unlike the synergistic divergence of Duane syndrome, the paradoxically abducting eye simultaneously abducted and depressed, suggesting that the synergistic divergence was attributable to aberrant innervation of the superior oblique muscle. A primary failure to establish normal neuronal connections with the extraocular muscles and levator muscle
37 Congenital Cranial Dysinnervation Syndromes 345 Fig Congenital fibrosis syndrome with synergistic divergence (simultaneous abduction during attempted lateral gaze) and Marcus Gunn jaw winking. (a) Bilateral ptosis, exotropia, and fixed downgaze. (b) Retraction of the right upper eyelid with mouth opening. (c) Position of the eyes with eyelids manually elevated. (d) In attempted left gaze, the right eye abducts and depresses. From Brodsky 104 with permission would predispose to neuronal misdirection, which allows for limited preservation of innervated muscle fibers and replacement of the remaining muscle by fibrous tissue. 120,702 Clinically, only deficient innervation could explain the superimposition of synkinetic eye movements on a diffuse congenital ophthalmoplegia, 291 Other forms of aberrant innervation have since been documented in CFEOM. 428 A neuropathologic study by Engle et al 249 demonstrated that congenital fibrosis of the extraocular muscles 1 is associated with abnormal development of the oculomotor axis, primarily affecting the superior division of the oculomotor nerve, corresponding to alpha motoneurons in the midbrain, the target extraocular muscles, and the levator and superior rectus muscles. This study established that CFEOM can result from the failure of developing cranial nerves to form appropriate neuromuscular connections with their target extraocular muscles, a finding supported by subsequent genetic studies. 8 10,104,120,247,823 Because most patients with CFEOM do not display signs of neuronal misdirection, and because anomalous fibrous bands and adhesions are sometimes discovered to be attached to the globe during strabismus surgery, 266,487 we now suspect that the genetically determined dysgenesis of CFEOM can directly influence the extraocular muscles, in addition to developing cranial nerves. It is therefore likely that the underlying pathophysiology of congenital fibrosis syndrome must lie along a spectrum from a primary absence of normal innervation to localized orbital dysgenesis of the extraocular muscles. Neuroanatomic abnormalities are often missed on routine intracranial MR imaging, which frequently shows no overt
38 346 7 Complex Ocular Motor Disorders in Children Fig CFEOM. Axial MR imaging showing bilateral hypoplasia of the ocular motor nerves. Courtesy of Joseph Demer, M.D. structural malformations of the brain. 104 However, highresolution images of the ocular motor nerve commonly show hypoplasia or absence of the oculomotor nerves and involved extraocular muscles (Fig. 7.16). 205,432,494 To date, three genetic CFEOM loci have been identified and three clinical phenotypes have been delineated. 252,508,753 is the most common form. The key clinical findings include bilateral nonprogressive ophthalmoplegia, bilateral ptosis, and an infraducted (downward) primary position of each eye with limited supraduction. 252 CFEOM1 pedigrees demonstrate autosomal dominant inheritance with full penetrance and minimal variation in expression. 820 CFEOM1 has been mapped to the centromeric region of chromosome ,769 It has now been demonstrated that congenital fibrosis of the extraocular muscles type 1 results from heterozygous mutations in the KIF21A gene encoding a kinesin motor protein. 820 Although the specific function of the KIF21A protein and its stalk are yet to be determined, the mouse ortholog, KIF21a, was found to be an anterograde microtubule-based motor protein expressed predominantly in neuronal tissues. Engle et al have found that human KIF21A is expressed most abundantly in developing neuronal tissues, suggesting that it plays an important role in neuronal development consistent with the congenital fibrosis of the extraocular muscles 1 phenotype. 150,430,820 As kinesins affect axonal transport after development (microtubule function), it remains unclear how a kinesin mutation could lead to neural agenesis. Because KIF21A has also been observed in other tissues, including skeletal muscle, it remains to be determined whether the primary pathology of congenital fibrosis of the extraocular muscles, resulting from KIF21A mutations, is in the nervous system (neurogenic), the extraocular muscles (myopathic), or both. For example, the frequent finding of fibrous bands in the orbits of patients with congenital fibrosis of the extraocular muscles is clearly incompatible with a purely neurogenic mechanism. CFEOM2 is autosomal recessive, and its gene is localized on chromosome 11q Affected individuals have bilateral ptosis and restrictive ophthalmoplegia, with eyes partially or completely fixed in an exotropic position. 750 Patients have severely limited ability to depress or adduct either globe. ARIX, previously called PHOX2A, is the gene that is mutated in CFEOM2 550,793,822 ARIX is a transcription factor essential for the development of oculomotor and trochlear nuclei in mice and zebra fish. 318 It is therefore believed that CFEOM2 results from hypoplasia of the oculomotor and trochlear nerve nuclei as a result of mutations in both copies of ARIX. CFEOM3 is an autosomal dominant disorder with variable expression and probably incomplete penetrance. 750 The gene maps to markers on 16p24.2-q Severely affected patients have ptosis, with eyes fixed in a downward and exotropic position and bilateral severe restriction of eye movements (a phenotype resembling that of congenital fibrosis of the extraocular muscles 1). Mildly affected patients have normally positioned globes, with limitation of vertical gaze. Moderately affected patients may have asymmetric involvement, with one eye severely affected and the other mildly affected. Mackey and colleagues 508 reported a family that maps to the CFEOM3, with involvement primarily of the vertically acting extraocular muscles. Because the involved extraocular muscles are both tight and hypoplastic, the surgical management of congenital fibrosis syndrome presents a unique set of challenges. 103 Free tenotomies obviate the need for suture placement into the rectus muscles, which can be technically difficult due to severe restriction. 103 Free tenotomies of tight rectus muscles are technically simple and rarely produce surgical overcorrection. 103 The need for resections can be determined by examining the position of the eyes under nondepolarizing paralyzing anesthesias. 823 It is also well to remember that surgical weakening of tight inferior rectus muscles can reduce their secondary adducting action and produce exotropia. 103 In our experience, raising the globe does not produce improvement in associated ptosis, so surgical correction of ptosis often needs to be performed in conjunction with inferior rectus tenotomy. Surgical correction of ptosis should be modest to avoid exposure keratopathy. Despite multiple surgeries, some degree of ocular misalignment usually persists. 103 Congenital Horizontal Gaze Palsy with Scoliosis In 1974, Dretakis and Kondoyannis described the autosomal dominant syndrome of horizontal gaze palsy and progressive scoliosis. 223 In 2004, Jen et al 400 described 11 patients with autosomal recessive congenital horizontal gaze palsy caused by a mutation of the ROBO3 gene on chromosome 11. Affected children are born with absent horizontal eye move-
39 Congenital Cranial Dysinnervation Syndromes 347 Fig Congenital horizontal gaze palsy with scoliosis. T2-weighted fast spin echo axial MR imaging shows butterfly configuration of the medulla with flattening of the ventral surface, reduced anterior posterior diameter and midline medullary cleft. Courtesy of Joseph Demer, M.D. ments and had variable strabismus, horizontal nystagmus, and defective vertical smooth pursuit. Convergence can be preserved, and some people use convergence substitution as a substitute for horizontal gaze. 241 All patients developed progressive scoliosis during early childhood. The disorder is found in both consanguineous and nonconsaguinous families. 241 Heterozygotes were unaffected. This disorder produces a pathognomonic brainstem malformation. MR imaging shows a hypoplastic pons and medulla, 622 with overall diminished anterior posterior dimension (Fig. 7.17). The ventral surface of the pons is flat. The medulla fans out both dorsally and ventrally in a butterfly configuration. There is a prominent midline cleft in the medulla that extends down to the cervicomedullary junction. Affected patients had electrophysiologic evidence of ipsilateral corticospinal and dorsal column-medial lemniscus tract innervation. In contradistinction to most other congenital cranial dysinnervation disorders, the abducens nerves are normal in size and configuration. 241 ROBO3 mutations may disturb brainstem morphogenesis by failing to promote decussation of long motor and sensory tracts in the pons and medulla. Impaired decussation of pontine ocular motor pathways may explain the absence of horizontal eye movements in this disorder. 88 Möbius Sequence Fig Möbius syndrome. Note characteristic flattening of lower facial features. Courtesy of Joseph Demer, M.D. Möbius sequence is a rare congenital disorder characterized by congenital facial weakness with horizontal gaze palsy 296 or impairment of ocular abduction. 781 Dysfunction of other cranial nerves, orofacial malformations, limb malformations, and musculoskeletal system defects are common associated features, but they are not obligatory for the diagnosis. Möbius sequence is a sporadic multiple-malformation complex that affects the face and horizontal gaze mechanisms bilaterally. 566 Affected children have mask-like facies, with the mouth constantly held open (Fig. 7.18). The upper facial nerves are affected more than the lower facial nerves, and facial asymmetry is common due to asymmetric facial strength. The eyes may be straight, esotropic or, rarely, exotropic. 191,200,518a,555 Those with straight eyes tend to have horizontal gaze palsy, 200 and some use convergence substitution to look to the side. Such children may be thought to have isolated bilateral sixth nerve paresis if the slow convergence movement and the associated pupillary constriction are not recognized. Another subset exhibits retraction of the globe on attempted adduction. 518a As in Duane syndrome, congenital ocular motor synkinesis may contribute to the horizontal conjugate gaze paresis in some cases. Additional deficits affecting other cranial nerves, particularly V, IX, and XII, may produce feeding and sucking difficulties in the neonatal period and subsequent speech difficulties, with or without atrophy of the tongue. 16 Rarely, the sixth cranial nerves may be spared. 754 Möbius sequence may be associated with a wide variety of associated limb malformations (talipes equinovarus, brachydactyly, syndactyly, congenital amputations) as well as hypoplasia or absence of the branchial musculature, particularly the pectoralis muscle (Poland anomaly). 16,454 Miller et al 553 refer to the subgroup with associated limb anomalies as terminal transverse defects with orofacial malformations (TTV-OFM), a term originally used by Temtamy and McKusick. 16 Endotracheal intubation may be especially
40 348 7 Complex Ocular Motor Disorders in Children difficult in children with Möbius sequence, owing to structural abnormalities of the mandible and palate. 22 Cardiovascular abnormalities (most commonly dextrocardia, patent ductus arteriosus, and ventricular septal defects), micrognathia, structural abnormalities of the pinna, and mild mental retardation and autism are also occasionally present. 16,54,193,403,545,547 Other abnormalities can include an A-shaped mouth, lagophthalmos, a hidden smile and laughter, syndactyly, a hypoplastic, asymmetric tongue that cannot be protruded over the lips, and a groove over the midline of the tongue. The ocular abduction deficit may be caused by congenital horizontal gaze paresis, Duane s syndrome, simple abducens palsy, or congenital fibrosis. 781 Children with Möbius sequence have difficulty relating to people in their environment because of an inability to convey their reaction of joy or sorrow. They are often incorrectly assumed to be mentally retarded and are predisposed to social and psychiatric problems. 21a,547a,555 The successful management of Möbius sequence entails a multidisciplinary approach that includes the medical, speech, education, and mental health disciplines. 16 Strabismus surgery can restore ocular alignment, 718,777 and regional muscle transfer and microvascular free tissue transfer have been used to provide innervated dynamic muscle to restore facial movement. 164,547,742 Because of their orofacial abnormalities, children with Möbius sequence are at higher risk of postoperative respiratory failure following general anesthesia. 302 Although Möbius sequence is now classified as one of the congenital cranial dysinnervation disorders, it is more than a cranial nerve or nuclear developmental disorder. Möbius sequence is a rhombencephalic developmental disorder with hypoplasia of the entire brainstem, including the traversing long tracts, and signs of neuronal degeneration and other congenital brain abnormalities. 782 Neurophysiologic studies suggest dysfunction at infranuclear, nuclear, and supranuclear levels. 148,779 Aberrant regeneration may contribute to the horizontal conjugate gaze paresis in some cases of Möbius sequence. The associated feeding and respiratory problems, along with poor motor development, are consistent with this hypothesis. 781 Often, the clinical features (facial weakness and impairment of ocular abduction) are only the most salient clinical features of a more extensive developmental disorder with probably diverse pathogenetic mechanisms. Neither etiology nor pathogenesis of this syndrome have yet been elucidated and are likely to be diverse in nature. 780 Two major pathogenetic explanations have been suggested: a primary genetic cause, implying a maldevelopment of the brainstem, and a primary ischemic cause, possibly due to embryological or environmental toxic factors mediating an interruption in the blood supply of the brainstem during early embryologic development. 781 These postulated pathogenetic mechanisms are based on a few postmortem observations, which include hypoplasia of the cranial nerve nuclei with or without active neuronal degeneration and focal necrosis with neuronal loss, gliosis, and calcifications in the brainstem. 781 MR imaging shows brainstem hypoplasia when measuring the pons (Fig. 7.17), supporting the hypothesis that Möbius sequence is a developmental disorder of the entire lower brainstem 604,612,780 with other cranial nerve aplasia. 225 Absence of the facial nerves despite residual function in some facial muscles suggests that other cranial nerves may aberrantly innervate some of the facial muscles. 780 The finding of crocodile tears in patients with Möbius sequence also supports this association. A variant of Möbius sequence with normal brainstem anatomy has recently been recognized. 225 Some investigators have suggested that ischemia of the lower cranial nuclei due to an insufficient blood supply in the pontine branches is the cause of Möbius sequence. 80,226 Intrauterine brainstem infarction could result from premature regression or obstruction of the primitive trigeminal arteries before the establishment of a sufficient blood supply from the vertebral arteries, which may explain the variability in clinical expression. 16 Brainstem calcifications, which have been attributed to vascular insufficiency, 16,219 are not indicative of a specific mechanism and could equally support a maldevelopmental pathogenesis. 782 Neuropathologic studies have emphasized the presence of brainstem atrophy and/or necrosis in Möbius sequence. 188a Patients with Möbius sequence are predisposed to primary respiratory failure and recurrent apnea, which correlates with both tegmental necrosis with calcifications and marked brainstem hypoplasia at autopsy. The association of Möbius sequence with thalidomide embryopathy and misoprostol exposure 191,518,774 demonstrates that early embryonic exposure to teratogens may produce a similar malformation complex. 543a Möbius sequence is usually considered sporadic, however, rare instances of autosomal dominant, autosomal recessive, and X-linked recessive inheritance have been reported. Cytogenetic studies have suggested two loci for Möbius sequence: 1p22 and 13q Monocular Elevation Deficiency, or Double Elevator Palsy Double elevator palsy is a descriptive term denoting a congenital deficiency of monocular elevation that is equal in abduction and adduction. Generally speaking, an inability to elevate one eye may occur on a restrictive or paretic basis and may be congenital or acquired. The patient or his family frequently reports that one eye shoots up and disappears under the upper eyelid while, in fact, the contralateral eye is the one with abnormal motility. The patient frequently has associated hypotropia and ptosis or pseudoptosis. The term double elevator palsy was originally coined to reflect what was then thought to be the basis for the disorder, namely, con-
41 Congenital Cranial Dysinnervation Syndromes genital palsy of the ipsilateral inferior oblique and superior rectus muscles, 801 a concept that has since been abandoned. It has since become apparent that the concomitant limited elevation, very characteristic of double elevator palsy, may result from at least three disparate pathophysiologic disorders, namely, inferior rectus restriction, superior rectus paresis, and a supranuclear disturbance of monocular elevation. 261 In truly paretic cases, dual palsy of the inferior oblique and superior rectus muscles does not occur; paresis of the superior rectus muscle alone (the dominant elevator of the globe) is sufficient to produce the clinical picture. The term monocular elevation deficiency is a more accurate descriptive term. The monocular elevation deficiencies undoubtedly overlap with the congenital cranial dysinnervation syndromes (discussed below). 577 Several large series have shown that most patients with monocular elevation deficiency have a restrictive abnormality to elevation. 533,534,690 Some of these cases represent unilateral inferior rectus muscle fibrosis, which can present with a tight inferior rectus muscle, hypotropia, and secondary ptosis in CFEOM3. 207,350,750 Congenital inferior rectus fibrosis and neurogenic double elevator palsy may closely mimic each other because both may exhibit defective elevation associated with ptosis or pseudoptosis. Conversely, a long-standing hypotropia associated with neurogenic double elevator palsy may result in secondary contracture of the inferior rectus muscle and cause a positive forced duction test. Similar cases of inferior rectus tightness may result from perinatal orbital trauma. 338 The diagnosis of contralateral superior oblique palsy should be considered in any child who has a monocular elevation deficiency, because fixation with the paretic eye can produce a contralateral inferior rectus contracture. Other conditions to be considered include orbital blowout fractures and orbital fat adherence syndrome. Patients with inferior rectus restriction due to orbital blowout fracture may also show a component of inferior rectus paresis. 461 On the basis of studies utilizing scleral search coil techniques, Ziffer et al 839 suggested the existence of at least three distinct etiologic categories: primary inferior rectus restriction, primary superior rectus palsy, and congenital supranuclear elevation defects. The inferior rectus restriction may be present primarily or may secondarily result from long-standing superior rectus weakness. Examination for the status of Bell s phenomenon and other reflex upward movements is very useful. An intact Bell s phenomenon, or the ability to produce an upward movement of the eye with the oculocephalic maneuver, suggests a supranuclear disturbance. An absent Bell s phenomenon would indicate either inferior rectus restriction or superior rectus palsy. The two can be distinguished by saccadic velocity analysis, forced ductions, and active force generation. If restriction is absent and the eyes are orthotropic in the primary position, a superior rectus paresis is most unlikely, and a supranuclear disturbance is 349 inferred. 534 Scott and Jackson 690 have noted that the appearance of an accentuated lower lid fold on attempted upgaze predicts the presence of an inferior rectus contracture. While the location of the lesion in the restrictive variety of the monocular elevation deficiency is readily apparent, generally pointing to tightness in the inferior rectus muscle complex, the location of a lesion in the other two classes of the monocular elevation deficiency is not as well determined. 535 Cases resulting from isolated superior rectus weakness could theoretically result from disorders, affecting the muscle or its nerve supply anywhere from the superior rectus subnucleus to the orbit. Congenital absence of the superior oculomotor division could produce double elevator palsy with hypotropia and neurogenic ptosis. Hoyt described a patient who developed sudden monocular elevation deficit and features of superior rectus paresis, which he attributed to superior rectus subnucleus infarction precipitated by coexisting polycythemia vera. 378 Mather and Saunders reported a case of bilaterally absent superior rectus muscles. 522 As oculomotor fascicular fibers destined to the elevators of the eye and eyelid are believed to course laterally in the fascicle as it traverses the midbrain, a midbrain infarction involving a lateral portion of the oculomotor fascicle can cause an acquired unilateral ptosis and elevation deficit. 145,382 A congenital lesion at this location could theoretically explain the paretic infranuclear form of monocular elevation deficiency. MR imaging has shown hypoplasia of the involved superior rectus muscle in one case, 138 and focal thickening of the inferior rectus in another. 435 However, the absence of these findings on neuroimaging in the great majority of cases support a prenuclear deficit in the unilateral center for upgaze. 435,607 Olson and Scott noted that 9 of 31 patients with monocular elevation deficiency had dissociated vertical divergence manifesting in the affected eye. 597 The findings of Bell s phenomenon, dissociated vertical divergence, normal velocity of upgaze saccades when moving from a downward to primary position, negative Bielschowsky Head Tilt test, and elevation during stage II of general anesthesia provide evidence of a supranuclear etiology. 548 Supranuclear disturbances of monocular eye movements are rare. The neuroanatomic substrate of unilateral supranuclear upgaze deficiency is controversial. It has been attributed to either lesions of the contralateral pretectum or lesions involving the upgaze efferents from the ipsilateral rostral interstitial nucleus of the medial longitudinal fasciculus. Most reports have been in adults in association with metastatic tumors. Lessell 485 reported a man with bronchogenic carcinoma who developed left monocular elevation deficit, but with orthotropia in primary position and an intact Bell s phenomenon. A metastatic tumor was found in the right pretectum at autopsy, which was speculated to have caused the double elevator palsy by interrupting axons destined for the ipsilateral superior rectus subnucleus and the contralateral
42 350 7 Complex Ocular Motor Disorders in Children inferior oblique subnucleus. Ford et al 274 described a 52-yearold woman who developed right monocular elevation paresis who was demonstrated to have a focal, right-sided tumor of the mesodiencephalic junction in the region of the rimlf. Acquired unilateral double elevator palsy has been described in a child with a pineocytoma. 570 The most recent evidence incriminates a lesion of the vertical saccadic burst or pause neurons of the rimlf. A genetic factor is suggested by the finding of identical twins concordant for supranuclear double elevator palsy on the same side, with preservation of Bell s phenomenon. 62 The possible neuroanatomic origins of monocular elevation deficit are summarized in Table 7.9. Patients with superior oblique palsy who habitually fixate with the paretic eye may present with limited monocular elevation of the contralateral (hypotropic) eye due to inhibitional palsy of the contralateral antagonist. 208 This so-called fallen eye syndrome may lead to a contralateral inferior rectus contracture and thereby cause a monocular elevation deficiency. The correct diagnosis may be inferred by the three-step test and the comparison of ductions to versions. Cases of vertical retraction syndrome, wherein congenital unilateral restriction of elevation is associated with retraction of the globe and narrowing of the palpebral fissure, 683 have been speculated to arise from a vertical innervational misdirection similar to that observed in Duane s syndrome and in some cases of congenital fibrosis syndrome. 291 The association of an isolated monocular elevation deficiency with congenital ptosis in general, and MGJW in particular, implies that aberrant misdirection may play a role in the pathogenesis of some cases of double elevator palsy. 819 Undoubtedly, a subgroup of patients with monocular elevation deficiency fall into the classification of congenital cranial dysinnervation syndromes (discussed below). This view is supported by reports showing that the superior rectus muscle itself may be involved in the aberrant phenomenon. For example, Oesterle et al 603 described a 9-month-old infant with congenital ptosis without jaw winking who showed an up-and-down movement of the left eye synchronous with nursing movements of the jaw. A 5-year-old girl with left ptosis, jaw winking, and left double elevator palsy showed up-and-down movements of the left upper lid and the left eye Table 7.9 Neuroanatomic differential diagnosis of monocular elevation deficiency Absence or hypoplasia of the superior rectus (e.g., Crouzon s disease) Inferior rectus contracture (e.g., congenital fibrosis syndrome, fallen eye syndrome ) Lateral oculomotor fascicular lesion Superior division oculomotor paresis Superior rectus myoneural junction disease (e.g., myasthenia gravis) Superior rectus subnucleus lesion Supranuclear lesion synchronous with chewing. The eye movements persisted after levator excision and fascia lata sling procedures. The authors speculated that the up-and-down eye movements probably represented aberrant innervation of the superior rectus muscle in a manner analogous to the abnormal innervation of the levator muscle in MGJW. Surgical treatment consists of inferior rectus recession (in cases with a positive forced duction test), 690 partial or full tendon width transpositions of the horizontal rectus muscles (in cases with a negative forced duction test), 141,227,349,441 or innervation surgery with large recession of the contralateral superior rectus muscle (in cases with a negative forced duction test and some residual supraduction). 629 Newer modifications of this procedure may include posterior fixation sutures to the transposed muscles and placement posterior fixation sutures on the normal superior rectus muscle. 709 Inferior rectus recession alone or in combination with other procedures ameliorates the restrictive variety of double elevator palsy. Treatment of neurogenic double elevator palsy includes conventional vertical rectus muscle surgery (recess and/or resect) and vertical transposition of the tendons of the medial rectus-lateral rectus superiorly (Knapp procedure). 135 The pseudoptosis disappears on correction of the vertical deviation, and any residual true ptosis can be addressed after ocular alignment is optimized. 142 Brown Syndrome Brown syndrome can be distinguished from double elevator palsy by its increasing limitation of elevation in the adducted position. 808a Brown syndrome must also be distinguished from the much less common inferior oblique palsy by the presence of little or no associated superior oblique overaction, a Y-pattern producing exotropia in extreme upgaze, and a positive forced duction test. Brown syndrome results from a congenital or, less commonly, an acquired dysfunction involving the trochlear-superior oblique tendon complex. 808a Either form may be constant or intermittent (Fig. 7.19). Intraoperative forced duction testing can help distinguish an inferior rectus restriction from a severe Brown syndrome. Retropulsion of the globe into the orbit while performing forced supraductions reduces or eliminates an inferior rectus restriction, but exacerbates tightness due to Brown syndrome. Pulling on the globe does the opposite. Many acquired and some congenital cases of Brown syndrome improve spontaneously so that a conservative approach is reasonable. It has recently been argued that congenital superior oblique palsy with misinnervation to the medial or inferior rectus muscle can produce a motility pattern that is indistinguishable from Brown s syndrome due to a tight superior oblique tendon. 447
43 Other Pathologic Synkineses 351 Fig Intermittent Brown syndrome: Teenager complained of intermittent diplopia. On attempted gaze up and to his left, full ocular ductions and versions were noted on some trials (a), but intermittently, right eye failed to elevate in adduction (b) Although classically viewed as a restriction syndrome, Neugebauer and colleagues have argued that Brown syndrome may be a congenital cranial dysinnervation syndrome. 577 Using high-resolution dynamic MR imaging, Kolling and colleagues demonstrated absence of the fourth nerve within the orbit and hypoplasia of the superior oblique muscle on the affected side in patients with Brown syndrome. 447 They have suggested that reinnervation of a paretic superior oblique muscle by branches from the medial or inferior rectus muscles can produce a motility pattern that is indistinguishable from Brown s syndrome due to a tight superior oblique tendon. 447 Treatment is undertaken for persistent cases with significant strabismus in the primary position and/or a significant compensatory anomalous head position. Superior oblique weakening (tenotomy or placement of a silicone spacer to lengthen the tendon), 818 with or without inferior oblique weakening, is the treatment of choice in persistent congenital or idiopathic acquired cases with symptomatic diplopia or marked compensatory chin elevation. Local corticosteroid injections around the trochlea may be helpful in acquired inflammatory cases. The occurrence of Brown s syndrome in multiple siblings 331 and in monozygotic twins 817 supports a genetic basis in some cases. Causes of acquired Brown syndrome include trauma, a rheumatoid nodule in the vicinity of the trochlea, peribulbar anesthesia, blepharoplasty, pansinusitis, frontal sinus osteoma, metastatic lesions, localized inflammation (orbital pseudotumor), surgical manipulation in the area of the trochlea, and surgical tucking of the superior oblique tendon. Other Pathologic Synkineses Physiological synkineses are well-recognized. Opening the mouth causes the eyes to simultaneously open, a reflex that is useful in the ocular examination of children. In the less known oculo-auricular phenomenon, horizontal gaze evokes a synkinetic retraction of the external ear muscles. 808 Curling of both auricles during strong lateral gaze is said to be present in 40% of the normal population. 765,808 This reflex is seen more easily in people who have prominent ears. 765 As discussed in Chap. 6, other forms of ocular motor synkineses can occur in isolation or cluster together. A trigemino-abducens synkinesis may occur after trauma, and facial synkinesis is a common finding after facial nerve palsy. It should be noted that various forms of aberrant innervation have been reported in the thalidomide embryopathy, especially Duane s retraction syndrome and aberrant lacrimation 544 (crocodile tears). Platysma-levator synkinesis has been documented in a child with congenital third nerve palsy, demonstrating the potential for aberrant regeneration from a portion of the facial nerve. 102 Rarely, deglutition-trochlear synkinesis may be noted, 526 with the affected patient showing torsional diplopia associated with swallowing. This phenomenon suggests a synkinetic movement coupling the trochlear nerve with the bulbar musculature that is innervated by the trigeminal, facial, and hypoglossal nerves. Boehme and Graef described a 10-yearold boy with a right-sided Horner s syndrome and a paresis of the recurrent laryngeal nerve, following neuroblastoma resection, whose right pupil distorted into an oval shape when he swallowed. 77 They postulated that aberrant vagal sprouts had established connections with the cervical sympathetic chain. Some phenomena resemble synkinetic movements, but are difficult to explain on the basis of aberrant reinnervation. In some patients, voluntary gaze may evoke such phenomena as vertigo, tinnitus, blepharoclonus, eyelid nystagmus, eyelid closure, facial twitching, arm movements, or seizures. 226,693,789,790 The pathogenesis of these synkinetic movements is unclear, but has been speculated to involve ephaptic transmission. 693 Gaze-evoked upper lid jerks (lid nystagmus) may also be seen in patients with Fisher s syndrome. 666 and in patients with midbrain pseudomyasthenia (e.g., due to mesencephalic astrocytoma). 114
44 352 7 Complex Ocular Motor Disorders in Children Internuclear Ophthalmoplegia The hallmark of internuclear ophthalmoplegia (INO) is weakness or absence of adduction in one or both eyes on lateral gaze, with nystagmus in the abducting eye. 481,706,834 Despite this adduction limitation, the eyes remain orthotropic in the primary position. The adduction weakness may be profound and quite noticeable on testing of ocular ductions and versions or may be so subtle as to be discernible only by noting slow adducting saccades in the affected eye. Refixation from abduction to primary position causes the contralateral eye to momentarily overshoot the target. 707 The slow, floating nature of the adducting saccades can be effectively elicited by having the patient fixate an optokinetic nystagmus target moving temporally with respect to the eye with limited adduction, which elicits a dysconjugate horizontal nystagmus of greater intensity in the abducting eye. 707 Convergence is usually preserved, except in rostral cases that simultaneously affect the medial rectus subnucleus. However, decreased convergence in the setting of bilateral INO with multiple sclerosis may be due to central scotomas caused by optic atrophy, as opposed to a primary convergence disorder. Older children may complain of diplopia. A skew deviation often accompanies unilateral INO, while vertical upbeating nystagmus in upgaze often accompanies bilateral INO. Unlike most forms of skew deviation in which the lower eye is ipsilateral to the lesion, the higher eye is usually on the same side of the lesion in patients with INO. As INO resolves, the clinical picture evolves from absent or decreased adduction to slow adduction to normal-appearing adduction with retention of the abducting nystagmus (i.e., abducting nystagmus is the last to resolve). Internuclear ophthalmoplegia signifies intrinsic brainstem disease involving the pons or midbrain. 706 It results from injury to axons that originate from interneurons in the abducens nucleus and project via the medial longitudinal fasciculus to the contralateral medial rectus subnucleus of the oculomotor nuclear complex. A number of mechanisms have been suggested to explain the dissociated nystagmus in the abducting eye. 836 One popular explanation suggests that increased innervation to the medial rectus to overcome the adduction weakness is accompanied, under the influence of Hering s law of equal innervation, by a commensurate increase in the innervation to the normal lateral rectus muscle of the contralateral eye. While increased innervation to the paretic medial rectus muscle would improve adduction, the increased innervation to the normal lateral rectus of the other eye would result in abducting saccadic overshoot followed by backward postsaccadic drift. This mechanism is supported by the observation that abducting nystagmus may also be noted in patients with adduction weakness resulting from medial rectus muscle recession. 786 Other mechanisms invoke the possibility that the abducting nystagmus may result from: (1) increased convergence tone to improve adduction of the weak eye; (2) interruption of descending internuclear neurons that run in the medial longitudinal fasciculus from the oculomotor internuclear neurons to the abducens nucleus; (3) associated injury to fibers other than those of the medial longitudinal fasciculus; and (4) an associated gaze-evoked nystagmus, with the component of the nystagmus expected in the adducted eye being dampened by the coexisting adduction paresis. 481,834 In unilateral cases, diplopia in the primary position is often vertical rather than horizontal and is attributable to a concurrent skew deviation. It can be eliminated with two to three prism diopters of vertical prism incorporated into glasses. Nystagmus and oscillopsia may complicate bilateral cases. The oscillopsia is a result of deficient vertical vestibuloocular reflex and pursuit movements or the abduction nystagmus. The role of strabismus surgery in selected patients has been recently analyzed. 463 A lesion, encompassing both the medial longitudinal fasciculus and the ipsilateral PPRF or the abducens nucleus, results in an ipsilateral horizontal gaze palsy and an INO. The only preserved horizontal movement is abduction of the contralateral eye. This constellation of findings is called the one-and-one-half syndrome. Bilateral INO with unilateral abducens paresis gives a similar motility deficit. The one-and-one-half syndrome has a similar spectrum of causes as INO. In premature infants, one occasionally notes large-angle exotropia with decreased or no response of the medial recti to vestibular manipulations with rotation or calorics. This has been interpreted as possible bilateral INO, suggesting a maturational delay in the development of the medial longitudinal fasciculus. Such exotropia has a good prognosis for spontaneous ocular alignment as the medial longitudinal fasciculus matures, compared to the constant large-angle exotropia of infancy that shows no evidence of adduction weakness. 381 This latter variety is usually observed in the setting of neurologic disease, but a subset is seen in otherwise healthy infants in a manner analogous to congenital esotropia. Duane type 2 syndrome, medial rectus entrapment in a nasal orbital wall fracture with associated paresis, myasthenia gravis, and Fisher syndrome may produce a similar motility pattern and must be excluded. Causes of INO in infants and children include demyelinating disease, stroke, brainstem tumors (particularly pontine glioma), 180 vasculitis, 439 inborn errors of metabolism, parainfectious encephalitis, structural malformations (Chiari malformation), 816 drug intoxication, 218,644 B 12 deficiency, 13 and trauma. Bilateral INO may be accompanied by exotropia, bilateral ptosis, and supraduction deficits in the midline mesencephalic cleft syndrome. 131,465 Another increasingly
45 Cyclic, Periodic, or Aperiodic Disorders Affecting Ocular Structures important cause of INO is HIV encephalitis. Myasthenia gravis is a recognized cause of pseudo-ino. 422 Those with bilateral and intermittent exotropia (WEBINO syndrome) can be treated surgically with large bimedial resections (successful in our hands) or bilateral lateral rectus recessions. 4 However, most cases without exotropia remit spontaneously. In adults, recovery is more likely in demyelinating than in vascular lesions and slower when there are other signs of brainstem injury. 82,233 The relative prognosis for INO in children has not been established. Cyclic, Periodic, or Aperiodic Disorders Affecting Ocular Structures A number of heterogeneous ocular disorders have in common a cyclic, periodic, or aperiodic pattern, that is, an involuntary process that repeats over time (Table 7.10). A major lesson Table 7.10 Cyclic, periodic, or aperiodic disorders affecting ocular structures Alternating anisocoria Cyclic esotropia Cyclic superior oblique palsy Cyclic vertical deviation Migrating pupil Oculomotor palsy with cyclic spasm Periodic alternating esotropia Periodic alternating gaze deviation Periodic alternating lid retraction Periodic alternating nystagmus Periodic alternating skew deviation Periodic head turns in congenital nystagmus Periodic mydriasis Ping-pong gaze Rhythmic pupillary oscillations (periodic pupillary phenomenon) 353 to be learned from these cyclic phenomena is the importance of observing certain ocular disorders over time. The nature of the cyclic phenomenon and the duration of a complete cycle define each disorder. Cyclic esotropia is a relatively rare condition that is also referred to as alternate day, circadian, or clock-mechanism esotropia. 333 The designation circadian, which denotes a 24-h cycle, is a misnomer because the usual cycle duration is 48 h. The typical case shows a 24-h period of manifest esotropia measuring prism diopters, alternating with a 24-h period of normal ocular alignment (Fig. 7.20). Less common are cycles of 24, 72, or 96 h in duration. The esotropia is nonaccommodative and nonparalytic, with normal eye movements in both eyes when straight and also when esotropic. On days when the eyes are crossed, diplopia is rare, fusional amplitudes are defective or absent, and sensory anomalies are frequent. Whether other cyclic phenomena occur in association with this condition is debatable. Friendly et al 279 monitored numerous psychological and physiologic functions and found no associated cyclical phenomenon. In contrast, Roper-Hall and Yapp 654 described cyclical changes in behavior, frequency of micturition, and EEG activity. Cyclic esotropia may first appear in infancy, but the diagnosis may be delayed months to years, with the average age at diagnosis ranging from 2 to 4 years, although some cases may occur abruptly in adulthood. Generally, the condition occurs spontaneously without apparent cause, but may be precipitated by strabismus surgery, retinal reattachment surgery, ocular trauma, optic atrophy, or CNS disease. 333 The natural history of this condition is not definitively known, but the cyclicity is thought to diminish with time, leading to a constant esotropia after several months to years. The mechanism of cyclic esotropia remains unknown, 737 and no correlative abnormalities of the hypothalamic pituitary axis have been found. 282 Some cases are associated with neurologic disease, 282,625 one case was triggered by traumatic sixth nerve palsy, 385 and one by surgery for intermittent exotropia. 762 These reports suggest that patients Fig Cyclic esotropia. Patient presented with history of intermittent esotropia but was found on further followup to have cyclic esotropia. She showed orthotropia (a) and esotropia (b) alternating daily
46 354 7 Complex Ocular Motor Disorders in Children with cyclic esotropia are basically strabismic with cycles of remission. 536 This abolition of cyclic esotropia after visual improvement appears analogous to previously reported cases of periodic alternating nystagmus that resolved after visual rehabilitation by removing a cataract or clearing a vitreous hemorrhage. It is therefore advisable to provide maximal optical/refractive correction prior to considering extraocular muscle surgery. The treatment of cyclic esotropia consists of strabismus surgery to correct the maximum deviation that stops the overt cycles. Richter 641 likened the effect of surgery to removing the hand of the clock without altering its motor. Cyclic esotropia has been reported in the aftermath of cataract, retinal detachment, intracranial surgery, 625,642,756 intermittent exotropia, 568 infantile esotropia, 536 and accommodative esotropia. 224 Cyclic esotropia should not be confused with periodic alternating esotropia. 344 The latter is a rare condition with cycles of similar duration to periodic alternating nystagmus (about 200 s). The few reported cases occurred in association with congenital periodic alternating gaze deviation. 342 Such patients probably had the neuroanatomic substrate of periodic alternating nystagmus, but with superimposed saccadic palsy leading to absent fast phases of nystagmus. Periodic alternating gaze deviation (PAGD) is a rare disorder consisting of a slow, conjugate horizontal deviation of the eyes from one side to the other. 344,480,720 It may occur at any age (some cases have been reported in infancy) and is associated with concurrent, controversive, cyclic face turning to maintain fixation. During a complete cycle, the eyes rotate conjugately toward one side for 1 2 min, usually with compensatory head turning to the opposite side; return to the midline for a changeover period lasting s; then conjugately deviate to the other side for 1 2 min, with compensatory head turning to the side opposite the direction of ocular rotation (Fig. 7.21). The patient may be orthotropic or esotropic. During eye closure, the alternating turning of the head ceases, even though the eyes continue the rhythmic movement. While the eyes are deviated, they exhibit abnormal voluntary movements, convergence, optokinetic nystagmus, and oculocephalic responses, but all of these normalize during the changeover phase. Caloric testing can override the eye and head movements, and the cycling stops during sleep. Periodic alternating gaze deviation may occur on a congenital basis (Fig. 7.20) or may be acquired. Most documented cases reported so far have been due to posterior fossa abnormalities. It has been reported in association with pontine vascular lesions, Arnold Chiari malformation, Dandy Walker malformation, congenital hydrocephalus, occipital encephalocele, cerebellar medulloblastoma, spinocerebellar degeneration, and Joubert s syndrome with cerebellar vermis hypoplasia. 342,425,480,720,728 In congenital or early onset PAGD, cerebellar vermis atrophy is the most common neuroradiologic abnormality. The duration of a complete cycle of PAGD (3 4 min) is similar to that of periodic alternating nystagmus (PAN), suggesting that the two conditions may have a similar neuroanatomic substrate, the difference being an added saccadic palsy in the case of PAGD. This concept is supported by the findings in a patient with recurrent cerebellar medulloblastoma who sequentially demonstrated PAN, bilateral gaze palsy, PAGD, and then resumption of PAN. 425 When encountered in a comatose patient, PAGD is usually considered a sign of bilateral cerebral hemispheric dysfunction with a relatively intact brainstem. 728 In this setting, the total duration of the abnormal movements is usually short, a few hours to days, disappearing a few hours before death. Ping-pong gaze superficially resembles PAGDs, but the movements are much faster, although some authors have used the two terms interchangeably. 638,692 It consists of conjugate horizontal ocular deviations that alternate rapidly every few seconds. Ping-pong gaze is usually seen in comatose or semicomatose patients and typically denotes cerebellar or bilateral cerebral hemispheric lesions, but a relatively intact brainstem. Ping-pong gaze has been speculatively ascribed to damage of the descending supranuclear inhibitory input on the horizontal gaze centers. Periodic alternating skew deviation is a rare condition in which the patient develops right hypertropia lasting one to several minutes, alternating with left hypertropia in a cyclic fashion. In some patients, the condition may be aperiodic or intermittent. Downbeat nystagmus may also be present. In one patient, a focal lesion affecting the interstitial nucleus of Cajal was demonstrated with computed tomography. In contrast to seesaw nystagmus, periodic alternating skew deviation has slower movements, larger excursions, and no torsional component. Periodic eye movement disorders with a cycle duration of about 200 s, which include PAN, PAGD, and periodic alternating skew deviation, may have similar pathophysiologic mechanisms and have been reported to occur in combinations in the same individual. 425,491 One case of cyclic superior oblique palsy developed in a 10-year-old patient following trauma to the left trochlear area. 809 Typical left superior oblique palsy alternated daily with orthotropia. A patient with cyclic vertical deviation of an unspecified nature, with a cyclic duration of 48 h, has been reported following craniofacial surgery. 537 Other cyclic ocular motor disorders include paroxysmal ocular tilt reaction and oculomotor palsy with cyclic spasm. In patients with oculomotor palsy with cyclic spasm, the pupil may reportedly be the only structure to cycle although, more commonly, the extraocular muscles and levator muscle are also involved in the cyclic spasms. Cyclic or alternating phenomena may affect the pupils. Keane described a patient with traumatic oculomotor palsy in whom the ipsilateral pupil, although unreactive to light, showed continuous rhythmic involuntary oscillations.
47 Cyclic, Periodic, or Aperiodic Disorders Affecting Ocular Structures 355 Fig Congenital PAGD. Two-year-old girl with Joubert syndrome displayed PAGD with cycle duration of about 200 s. During complete cycle, (a) eyes rotated conjugately to right over 90 s, usually with compensatory head turning to opposite side, (b) returned to midline for a He speculated that dysfunction of the central parasympathetic nervous system may have been responsible. Alternating anisocoria is a rare condition characterized by alternating changeover period lasting seconds, then (c) conjugately deviated to other side over 90 s, with compensatory head turning to side opposite direction of ocular rotation. (d) MR imaging showed profound cerebellar vermis hypoplasia pupillary dilatation, with the dilated pupil showing little or no light reactivity in some cases, but normal reactivity in others. 121 This may occur in otherwise neurologically normal children,
48 356 7 Complex Ocular Motor Disorders in Children but a similar phenomenon has been observed in a quadriplegic patient with a posttraumatic syringomyelic cyst. 440 The pathophysiology of this phenomenon is unclear, but it may be due to either episodic oculosympathetic spasm producing alternating Claude Bernard Horner s syndrome or episodic oculosympathetic interruption producing Horner s syndrome. Some other pupillary disorders have been intermittent rather than rhythmic and include intermittent mydriasis, cyclic sympathetic spasm with concentric dilatation lasting s, and tadpole pupils, in which the sympathetic spasm is sectoral. 746 Conditions with cycle duration of a few minutes sometimes become apparent when the clinician notices a reversal of a previously noted finding for example, observation of conjugately turned eyes to the right in a patient whose eyes were previously noted to be conjugately turned to the left brings forth the diagnosis of PAGD. Conditions with a cycle duration of 24 h or longer cannot be definitively diagnosed from data collected during a single office visit. For instance, a patient with cyclic esotropia may be initially evaluated on a crossed day and be labeled as congenital or acquired esotrope. If the subsequent visit occurs on a crossed day, it may only consolidate the earlier false diagnosis. If it occurs on a straight day, the clinician may suspect spontaneous resolution or an accommodative element with variable angle and either question the observation of esotropia in earlier visits or correctly suspect cyclic esotropia. Conversely, initial evaluation of a patient with cyclic esotropia on a straight day may lead to missing the diagnosis, dubbing the condition pseudostrabismus. Cyclic phenomena in children are not limited to the neuroophthalmologic disorders discussed here, but involve other organ systems as well. Other biologic phenomena, such as sweating, salivation, and pulse rate, also have intrinsic periodicity, as do many manifestations of psychiatric dysfunction. Periodicity appears to be the norm in many biologic phenomena, and a normal person displays a complex array of biorhythms involving the various body systems. 640 Numerous periodic or rhythmic disorders have been described for example, periodic recurrence of fever, swelling of joints, fluctuations of circulating blood cells (periodic hematopoiesis), and edema. Accumulating evidence points to the existence of a biologic clock mechanism that keeps time with extraordinary accuracy and is independent of internal and external stimuli. discharge in a peripheral nerve. Neuromyotonia describes neurotonia accompanied by fibrillations, fasciculations, myokymia, or sustained contraction of a muscle group. Ocular neuromyotonia describes sustained contraction of one or more extraocular muscles due to involuntary firing of the supplying ocular motor nerve. Ocular neuromyotonia is a relatively rare ocular motility disorder that manifests with brief paroxysmal monocular deviations with associated diplopia. 701 Episodes generally last s (range, 5 s to 3 min) and may recur 20 or more times per day. Some episodes occur spontaneously, while others are triggered by gaze in the direction of the involved muscle. Between attacks, affected patients usually show normal ocular motility, although some may show subtle evidence of aberrant innervation manifesting as minimal lid retraction in downgaze. The paroxysms result from tonic involuntary contraction of extraocular muscles innervated by the third (all muscles supplied by the nerve or any combination thereof), fourth, or sixth cranial nerves. 57,486 Spontaneous discharges from axons with unstable cell membranes are presumed to underlie this condition. In some cases, ocular neuromyotonia may coexist with primary aberrant regeneration. 160 Histopathologic studies of peripheral nerves in nonocular cases of neuromyotonia have shown segmental demyelination as well as axonal degeneration, sprouting, and remyelination. 794 Most patients have a history of brain irradiation, typically for the treatment of tumors of the skull base, such as pituitary tumors or craniopharyngiomas. 486,798 Idiopathic cases with no specific cause have been reported. 798 The interval between radiotherapy and onset of neuromyotonia may be months to years. Two cases have been reported in teenagers following radiation therapy for parasellar lesions. 199,562 A favorable response to membrane-stabilizing medications, such as carbamazepine is reported in many patients, supporting pathoetiology consisting of spontaneous or impulse-induced repetitive discharge of hyperexcitable trigger zones in ocular motor nerves. In some instances, the neuromyotonia has not recurred on cessation of the medication. Some cases may remit spontaneously. Several conditions not associated with radiation therapy may come to be classified as ocular neuromyotonia. Adie s pupil and superior oblique myokymia are two common forms of ocular neuromyotonia seen in neuro-ophthalmologic practice. Ocular Neuromyotonia Myotonia denotes delayed muscle relaxation after sustained contraction as a result of muscle membrane dysfunction. In contrast, neurotonia or pseudomyotonia represents delayed muscle relaxation as a result of impulse-induced repetitive Ocular Motor Adaptations and Disorders in Patients with Hemispheric Abnormalities Cerebral hemispheric abnormalities are often associated with ocular motor abnormalities that may be subtle or profound depending upon the size, location, and other characteristics of the lesion. These are reviewed in detail elsewhere.
49 Eyelid Abnormalities in Children Children with congenital hemianopsia develop a compensatory saccadic strategy, wherein they overshoot the intended visual target then find it as the eyes drift back. 380 Some patients with congenital homonymous hemianopsia have been noted to also show concurrent exotropia, usually also of early onset. Some investigators have suggested that the exotropia in these patients is a compensatory phenomenon for the hemianopic field defect, essentially allowing the patient to have panoramic vision. For this to occur, anomalous retinal correspondence has to coexist, otherwise the patient would be diplopic. 306,363 Some investigators raise the possibility that the exotropia in such patients is an epiphenomenon, possibly reflecting the type of strabismus one sees in many children with neurologic disease. 380 It is not clear whether the concurrent exotropia in these children simply represents strabismus precipitated by infantile neurologic disease, or develops as a physiological compensatory phenomenon leading to the development of anomalous retinal correspondence and panoramic vision, or exists as a combination between these two mechanisms. The fact exotropia is so common in these children, who often show an uncanny ability to navigate through visual space without difficulty despite their homonymous hemianopia, suggests that the exotropia serves an important compensatory function. Therefore, the clinician should understand the potentially devasting sensory implications of performing strabismus surgery to straighten the eyes. 286 Eye Movement Tics Tics are quick, jerky, sudden, and repetitive movements of circumscribed groups of muscles without apparent cause or purpose. They occur frequently in children and most often resolve spontaneously. Tics most commonly affect facial musculature, but may rarely affect the extraocular muscles, causing eye movement tics. 696 It is important to recognize eye movement tics as such and not confuse them with more serious disorders. The few cases of eye movement tics reported to date have been mostly associated with facial tics, but isolated ocular tics may occur and may thus mimic nystagmus. Frankel and Cummings 276 reported eye rolling tics in a group of patients with Tourette syndrome. The tics consisted of eye rolling, gaze abnormalities, staring, or forced gaze deviations resembling oculogyric crises. Two of the cases described a corneal sensation preceding their tics. All of these children also had blepharospasm, and most were taking neuroleptic or other medications. Neuroleptics have been implicated in the pathogenesis of a variety of movement disorders, including tics and oculogyric crisis. Binyon and Prendergast 73 reported three cases with conjugate eye movement tics: one was associated with Tourette s syndrome, 357 one was associated with neurological and behavioral disorders, and one was isolated. The latter resolved spontaneously. Shawkat reported opsoclonus-like ocular tics associated with facial tics in an otherwise healthy child that resolved spontaneously. Affected children are sometimes able to imitate their tics in the clinic on request, which is a useful indicator that consciousness is not impaired during the tics, allowing the examiner to rule out epilepsy. Eyelid Abnormalities in Children Congenital Ptosis As detailed above, congenital ptosis occurs most frequently as a congenital cranial dysinervation disorder. Because congenital ptosis can also be the presenting sign of several other neuro-ophthalmologic disorders (Table 7.11), however, specific attention to ocular motility, pupillary examination, and coexisting neurologic signs is imperative. Congenital ptosis must also be differentiated from pseudoptosis due to ipsilateral hypotropia, enophthalmos, or contralateral lid retraction. Motility abnormalities may include congenital oculomotor nerve palsy and double elevator palsy. The latter is a reason to be conservative with ptosis correction to prevent corneal exposure postoperatively due to absent Bell s phenomenon. Pupillary abnormalities associated with unilateral ptosis may include a large unreactive pupil due to oculomotor palsy or a miotic pupil either due to Horner syndrome or to congenital oculomotor palsy with aberrant regeneration involving the pupil. Causes of pupillary abnormalities associated with bilateral early-onset ptosis include Fisher s syndrome and botulism. Rarely, a cerebral hemispheric lesion causes unilateral or bilateral ptosis (termed cerebral ptosis) in the absence of other neurological signs. 452 Lowenstein et al 501 described two children with hemispheric arteriovenous malformations Table 7.11 Differential Diagnosis of Congenital Ptosis Blepharophimosis syndrome Cerebral ptosis Congenital fiber-type disproportion Congenital fibrosis syndrome Congenital myotonia Horner syndrome Hypotropia with pseudoptosis Marcus Gunn jaw winking Microphthalmia, enophthalmos Myasthenia gravis Myotubular myopathy Oculomotor nerve palsy
50 358 7 Complex Ocular Motor Disorders in Children and contralateral ptosis that progressed over several years. In both cases, the ptosis resolved following resection of the arteriovenous malformation. One should consider the rare possibility of cerebral ptosis in the child with progressive nonmyasthenic ptosis, especially when a history of seizures or recurrent unilateral headaches is obtained. The finding of unilateral congenital ptosis with lid swelling should suggest the diagnosis of dural arteriovenous fistula. 313 When accompanied by supraduction deficit, bilateral internuclear ophthalmoplegia, and exotropia, bilateral ptosis should suggest the possibility of midline mesencephalic cleft malformation. 131,465 Tatli et al reported bilateral ptosis with lower extremity muscle weakness and dysautomomia as probable paraneoplastic effects of neuroblastoma in a 28-month-old girl. 739 An extensive workup for myasthenia gravis was negative, and no paraneoplastic autoantibodies were identified. Excessive Blinking in Children The spontaneous blink rate in infants under 18 months of age is low, ranging from 2 to 5 blinks per minute, whereas in older children, it is about 10, reaching approximately 20 blinks by the age of 20 years and remaining relatively constant throughout adulthood. Benign tics, (blinking eyes, twitching mouth, movements of head, jerking shoulders or other body parts) are common in children, with a prevalence as high as 12% between the ages of 6 and 12 years. 471 Benign tics involving the eyes, may manifest as excessive intermittent blinking without associated ocular or systemic disorders. Eye winking or blinking tics in children represent exaggerated contractions of the orbicularis muscle. The tics usually increase in frequency when the child is bored, anxious, or tired. Most these tics are benign and transient, improving spontaneously with time, without any discernible causation. Unlike essential blepharospasm of adults, there is little, if any, functional visual impairment. Although tics have been thought to result from an underlying psychological conflict, 509 recent evidence suggests that they may be of organic origin but triggered or exacerbated by stress. Elston et al 236 described a family in which different members in three generations suffered eye-winking tics, excessive blinking, and/or blepharospasm. The proband in that study had eye-winking tics in childhood, then developed excessive blinking evolving to blepharospasm by the age of 21 years. They also described five patients with adult-onset blepharospasm, or Meige s syndrome, who had a history of excessive blinking dating back to childhood. They speculated that eye-winking tics, excessive blinking, and blepharospasm may represent age-dependent manifestations of a common pathophysiologic disorder. 236 Frequent eye blinking may be a manifestation of ocular surface, tear film, or eyelid disorders at any age. It may also accompany lacrimal drainage obstruction. Children with congenital glaucoma are classically described as presenting with the triad of epiphora, photophopia, and blepharospasm, but may also show frequent blinking, as do children with significant intraocular inflammation. Excessive blinking and blepharospasm also occur in conditions with damage to the basal ganglia, such as Wilson disease and Huntington disease. 127 Seizure activity is associated with a variety of neuroophthalmologic signs and symptoms that include nystagmus, gaze deviations, spasm of the near reflex, hemianopsia, cortical blindness, as well as a variety of eyelid signs. Lid signs may consist of eyelid nystagmus or eyelid fluttering resembling tics and spells of eyelid spasms. For instance, Cogan et al 179 described a 7-year-old girl with methylmalonic aciduria and homocystinuria who exhibited spells of fluttering of the eyelids and elevation of the eyes. The EEG showed 2.5-s polyspike and wave discharge during the spells, characteristic of petit mal seizures. Excessive blinking and eyelid myoclonia has been reported in association with typical absence seizures, 28 and this condition has been reported in monozygotic twins. 202 Finger waving and rapid eyelid blinking is a feature of photoconvulsive epilepsy. Eyelid myoclonia may occur on eye closure in association with electrical status epilepticus without altered consciousness. 776 Patients with occipital lobe epilepsy may exhibit a variety of neuro-ophthalmologic manifestations, including elementary visual hallucinations, ictal or postictal amaurosis, eye movement sensations, eye and/or head deviation, visual field deficits, and early forced blinking or eyelid flutter. 807 Seizure activity may also be associated with abnormal eye movements such as gaze deviations (usually to the contralateral side) and nystagmus. 415,426,758 The terms lid nystagmus, upper lid jerks, and lid hopping have all been applied to a neuro-ophthalmologic phenomenon in which a series of rapid, rhythmic, jerky movements of the upper lids occurs alone or in conjunction with specific movements of the eyes or head. 114 Most previously reported cases have been associated with posterior fossa disease and can be subdivided into lid nystagmus evoked by convergence and lid nystagmus evoked by horizontal gaze. 377,664 Children have been described with signs and symptoms of myasthenic ophthalmoplegia in whom gaze-evoked lid hopping signaled the presence of intrinsic midbrain tumors. 114,633 Other rare causes of lid nystagmus include the lateral medullary syndrome, pretectal lesions, and Fisher syndrome. 666 While horizontal eye movements in myasthenia gravis may be accompanied by occasional twitching or fluttering movements of the upper lids, lid nystagmus is uncharacteristic of neuromuscular disease and should prompt neuroimaging. Lid nystagmus would appear to necessitate a dissociation between the output of the levator and the
51 Eyelid Abnormalities in Children superior rectus subnuclei within the oculomotor nuclear complex. Convergence-evoked lid nystagmus often signifies a structural lesion within the posterior fossa. Howard 377 described a patient with convergence-evoked eyelid nystagmus without ocular upbeating nystagmus, who had a large vascular lesion distorting the blood supply to the pontomesencephalic and pontomedullary junctions. Safran et al 664 described two similar patients with convergence-evoked eyelid nystagmus. Patient 1, a 29-year-old man, developed a cerebellar syndrome after sustaining severe head injuries. Patient 2, a 12-year-old girl, had a tumor of the anterior cerebellar vermis. Treatment with corticosteroids (patient 1) and surgery (patient 2) led to gradual resolution of the eyelid nystagmus, which the authors attributed to cerebellar dysfunction. The cause of tic disorders may lie in the dopaminergic system of the brain, as well as in genetic and other biological factors. 406 Coats et al 171 found the most common etiologies of excessive eye blinking to be anterior segment or lid abnormalities in children. Occasionally, children show frequent eye blinking as a manifestation of Tourette syndrome. 406 Some children may exhibit lower eyelid pulling as a transient behavior lasting weeks to months. 147 These children are otherwise healthy, and the condition is thought to initially be triggered by ocular irritation, but then develops into a bad habit. This functional disorder is readily distinguishable from eye poking (oculodigital sign) encountered in children who are blind secondary to congenital retinal disease (i.e., Leber congenital amaurosis, retinopathy of prematurity, retinal dysplasia) 394 and vigorous eye rubbing seen in patients with Down syndrome. Eyelid myokymia may occur alone or in association with facial myokymia. Facial and eyelid myokymia are characterized by unilateral involuntary fine rippling movements, spreading across the surface of affected muscles. Facial myokymia has been reported in association with multiple sclerosis, Guillain Barré syndrome, Bell s palsy, rattlesnake bites, brainstem tumors, and others. The clinical features of facial myokymia are sufficiently distinctive to avoid confusion with other facial dyskinesias such as essential blepharospasm, tardive dyskinesia, facial tics, focal seizures, aberrant regeneration of the facial nerve, hemifacial spasm, and benign facial fasciculations. Some cases of facial myokymia have occurred in association with spastic paretic facial contracture, which is characterized by tonic spasm of the paretic facial muscles, with typical prominence of the nasolabial fold and deviation of the nose toward the affected side. We have examined a patient with diffuse disseminated encephalomyelitis who showed left facial myokymia, spastic paretic facial contracture, and one-andone-half syndrome and who also showed scattered lesions in the brainstem on MR imaging. Gilles de la Tourette syndrome is a bizarre but relatively common disorder that comprises multiple involuntary motor tics and obscene utterances. There is a predilection for whites 359 and for boys (male-to-female ratio ranges from 3:1 to 5:1). The condition is familial in about one-third of cases, appears to be transmitted as an autosomal dominant trait, and has been mapped to the long arm of chromosome 18. Symptoms usually appear between ages 5 and 10 years, with presenting features consisting of multifocal tics of the face and head. In addition to clonic motor tics (most commonly blinking and facial twitching) and vocal/phonic tics seen in all patients, many patients exhibit one or more dystonic tics that may include oculogyric deviations, blepharospasm, and dystonic neck movements. 398 The globes may also show dystonic tics. For example, Frankel and Cummings 276 reported eye rolling tics in a group of patients with Tourette syndrome who also had blepharospasm. The motor tics are followed by vocal tics such as grunting, sneezing, coughing, barking, coprolalia (compulsive monosyllabic swearing), and echolalia. Tiqueurs may be able to voluntarily suppress the tics, but a resulting mounting tension eventually leads to further discharge of tics. Patients may show associated obsessivecompulsive traits, self-mutilation, attention-deficit disorders, learning disabilities, and serious psychiatric illness. Haloperidol and pimozide are useful in controlling the symptoms. Epileptic disorders and their treatment may cause eye rolling or eyelid myoclonia. 188 When accompanied by absence spells, eyelid myoclonia is highly suggestive of Jeavon s syndrome (i.e., eyelid myoclonia and absences). In this condition, there is marked jerking of the eyelids with upward deviation of the eyes, associated with spike-wave discharges that are often irregular, immediately after eye closure and invariably evoked by intermittent photic stimulation. This diagnosis is likely when the eyelid myoclonia is combined with photosensitivity and pathognomonic when it also occurs after eyelid closure (in some cases, it can look like the child is purposefully closing the eyes to initiate a seizure). EEG shows characteristic eye closure-related discharges and photosensitivity. Rhythmic or random closing of the eyes is often seen in other forms of idiopathic generalized epilepsy, with absences or the eyelid jerking that may occur at the opening or the initial stage of the discharges in typical absence seizures of childhood or juvenile myoclonic epilepsy. Toxicity from the antiepileptic drug clobazam in a 4-yearold boy has been reported to cause episodes of eye rolling, ataxia, and back arching, which were nonepileptic. 50,188 Chung et al found ictal eye closure to be a reliable indicator of psychogenic nonepileptic seizures. 165 The workup of tics is conservative, consisting mainly of observation, unless otherwise dictated by the presence of associated clinical findings. Explanation and reassurance of patients and their families is helpful in steering them away from becoming too focused on the tics. Neuroleptics and other drugs should be reserved for severe cases. The longterm prognosis of tics is good, with about two-thirds of cases spontaneously remitting. 186
52 360 7 Complex Ocular Motor Disorders in Children Hemifacial Spasm Hemifacial spasm is primarily a disorder of middle-aged and older individuals, but a variety of cases have been reported in childhood. Hemifacial spasm is characterized by involuntary, intermittent unilateral twitching of the muscles innervated by the facial nerve, almost uniformly including the orbicularis oculi. They differ from tics in several ways, including the inability of the patient to suppress the twitches or initiate them, and the absence of compulsion to make them. In about half the patients, a particular position of the head (most commonly, the contralateral lateral decubitus position) diminishes or halts the spasms. The patient may hear ipsilateral clicking sounds if the stapedius muscle is involved. Hemifacial spasm should be differentiated from blepharospasm and facial myokymia. Essential blepharospasm consists of bilateral, localized, repetitive spasms of the orbicularis oculi muscle that, in severe cases, leads to visual impairment. The rare case of bilateral hemifacial spasm can be distinguished from essential blepharospasm because the bilateral contractions do not occur synchronously. Essential blepharospasm is not seen in children, but excessive blinking due to tics or blepharospasm due to ocular disorders, such as congenital glaucoma, may bear superficial resemblance to essential blepharospasm. Facial myokymia consists of continuous, fascicular rippling movements of the face that usually begin in the orbicularis muscle. It is not affected by voluntary or reflex activity of the face. The most common causes include multiple sclerosis and brainstem glioma. Most adult cases of hemifacial spasm are caused by vascular compression of the facial nerve at the root exit zone of the brainstem. 397,806 The offending vessels are usually arterial, most commonly the anterior or the posterior inferior cerebellar artery or the posterior inferior cerebellar artery. 667 This neurovascular compression theory was challenged by an MR imaging/mr angiography study that concluded that the development and severity of hemifacial spasm were not associated with a specific blood vessel or multiple neurovascular contact points. 367 Cross talk between the sensory and motor branches of the seventh nerve has been suggested as an underlying pathophysiology. Congenital or acquired cholesteatoma is the most common associated tumor. Vascular malformations of the posterior fossa have also been implicated. Rare familial cases suggest a component of genetic transmission. Friedman et al 278 described a family in which hemifacial spasm occurred in five members through three generations. Aside from neurovascular compression, 490,540 a variety of rare causes of hemifacial spasm have been identified in children. These include venous sinus thrombosis, 271 masses of the fourth ventricle, 271 ganglioneuroma, 470 pilocytic astrocytoma, 538,667 congenital or acquired cholesteotoma, 630 tuberculous meningitis, 670 thickening of the arachnoid membrane, 443 and possible intrauterine facial nerve injury. 831 The diagnosis of epilepsy should also be considered, as hemifacial spasm has been reported as part of a seizure phenomenon. 21,41,356 The presence of hemifacial spasm in an infant should therefore be considered an ominous sign. Langston and Tharp 470 described a case of infantile hemifacial spasm beginning at 6 weeks of age. Surgical exploration at 5½ years of age revealed a ganglioneuroma of the fourth ventricle. Flueler et al 271 described three infants who presented with the onset of hemifacial spasm in the first year of life. One patient had occlusion of the straight sinus and large collateral vessels at the base of the brain, supporting the concept of vascular compression of the facial nerve at its exit from the brainstem as a mechanism for the production of hemifacial spasm. Each of the other two patients had an intrinsic mass compressing the fourth ventricle: one was located in the lower pons and extended into the cerebellar vermis and right cerebellar peduncle; the other involved the cerebellar vermis and right middle cerebellar peduncle. Interestingly, hemifacial spasm has been described in some cases of Joubert syndrome, which includes cerebellar vermis aplasia or hypoplasia. 438 Unlike the usually benign nature of the late childhood and adult variety, it appears that earlyonset hemifacial spasm should raise suspicion of an underlying CNS malignancy. Neuroimaging is therefore warranted in cases of hemifacial spasm with onset in the first 2 years of life. In older children with typical, isolated, hemifacial spasm, neuroimaging should be obtained when atypical features (headaches, facial pain, cranial neuropathy, cerebellar dysfunction) are present. 46 In the toddler with apparent hemifacial spasm, the diagnosis of accommodative esotropia should also be considered. 122 In this setting, a young child may have to exert a hemifacial contraction to close one eye to prevent diplopia. For this reason, a cycloplegic refraction should be included in the diagnostic evaluation of hemifacial spasm. 122 Both botulinum injection and surgical decompression of the facial nerve have been used with good results in both adults and children. 153,704 Microvascular decompression of the facial nerve is considered the definitive treatment by some authors. Jho and Jannetta 402 described improvement of hemifacial spasm with microvascular decompression of the facial nerve in two children and eight adults who had the onset of hemifacial spasm before the age of 20. Eyelid Retraction The margin of the upper eyelid is normally located 1 2 mm below the upper corneoscleral limbus. Upper eyelid retraction is more commonly considered in the adult age group, where it occurs most commonly in patients with thyroid eye disease, dorsal midbrain syndrome, trauma, proptosis,
53 Eyelid Abnormalities in Children and seventh nerve palsy, amongst other conditions. SCAs, such as Machado Joseph disease, are known to produce a peculiar ocular stare that is has been attributed to lid retraction rather than proptosis. The causes of eyelid retraction in children are heterogeneous (Table 7.12). 729 Graves orbitopathy is rare in children. When it occurs, it is less severe than in adults, 317,766,827 and most cases require no treatment. 228 Most children are clinically hyperthyroid at the time of diagnosis. 228 Exophthalmos is notably less common in the prepubescent group, presumably because there is room for the orbit to grow and produce a physical decompression. In our experience, diplopia is rare, Table 7.12 Causes of eyelid retraction in children Setting sun sign/hydrocephalus/dorsal midbrain syndrome (Collier sign) Congenital, cryptogenic Oculomotor palsy with aberrant innervation Oculomotor palsy with cyclic spasm Marcus Gunn jaw winking Eyelid retraction to darkness (normal in first year of life) Neuromyotonia Graves disease/hyperthyroidism Familial periodic paralysis Orbital hemangioma Optic nerve anomalies with vertical nystagmus Contralateral ptosis with compensatory superinnervation of both levators (fixation duress) Myasthenia gravis Hepatic cirrhosis Cushing syndrome Levator muscle fibrosis Inferior rectus muscle restriction with fixation duress Iatrogenic after surgical repair of ptosis Eyelid scarring (e.g., after inflammation from herpes zoster) Claude Bernard syndrome (sympathetic irritation) Volitional lid retraction Startle reflex to dimming light in infants 361 and orbital fat expansion is more common than extraocular muscle enlargement on imaging studies. Compressive optic neuropathy is rare. 228 Secondhand smoke has been implicated as a possible pathogenetic factor. 152 Unless specific clinical or neuroimaging findings suggest the diagnosis of Graves orbitopathy, neurological causes should be primarily considered. Bilateral lid retraction in dorsal midbrain syndrome is attributable to disruption of inhibitory fibers from the posterior commissure to the central caudate nucleus. 182 Rarely, patients with medial midbrain lesions demonstrate a plus minus lid syndrome characterized by ipsilateral ptosis (from injury to the oculomotor fascicle) and contralateral lid retraction (from disruption of bilateral inhibitory fibers to the central caudal nucleus). When the ptosis resolves, the ptotic lid can assume a retracted position. 283,287 Lid retraction in children is often intermittent, as in aberrant reinnervation associated with congenital oculomotor palsy or the MGJW phenomenon. Cases of neuromyotonia affecting only the levator muscle would also be expected to show isolated intermittent lid retraction. Cases of oculomotor palsy with cyclic spasm show intermittent momentary elevation of the eyelid on the affected side, but careful scrutiny of the ocular alignment and the mandatory pupillary involvement in the cyclic process confirm the diagnosis. 499 A single case of posttraumatic, bilateral, pupillary-involving oculomotor palsy has been reported, wherein the patient showed bilateral nonsynchronous episodic eyelid retractions not associated with eye movement or pupillary changes. 467 Patients with unilateral inferior rectus restriction and alternating fixation may show intermittent contralateral eyelid retraction, which occurs as a result of Hering s law when the restricted eye takes up fixation. Healthy infants also frequently display eyelid retraction to darkness, which resembles the setting sun sign (Fig. 7.22). Eyelid retraction to darkness can be elicited by turning off the Fig Pseudo-setting sun sign. Healthy infant showed eyelid retraction immediately after room light was turned off (a) that resolved when ambient illumination was restored (b)
54 362 7 Complex Ocular Motor Disorders in Children room lights while observing the palpebral fissures. It can be a clinically useful sign that an apparently blind infant has at least light perception vision. The phenomenon, which appears to be limited to the first year of life, disappears when ambient illumination is restored and is thought to represent a primitive startle reflex. Apraxia of Eyelid Opening Apraxia of eyelid opening is a nonparalytic motor disorder of the eyelids. It is characterized by the inability to volitionally initiate eyelid opening despite intact reflex lid elevation, lack of concurrent orbicularis oculi muscle contraction, and intact ocular motor nerves. Affected patients open the eyes manually or may employ a head thrusting movement to do so. 484 It may be differentiated from blepharospasm by the absence of Charcot s sign (orbicularis contraction forces the eyebrows to a level lower than the superior orbital margin). The condition usually appears in older patients with extrapyramidal disease, but may be seen in patients with unilateral or bilateral hemispheric dysfunction. It may respond favorably to Botulinum injections. 418 Pupillary Abnormalities Congenital Bilateral Mydriasis Congenital mydriasis is a rare defect that was first described by White and Fulton in Lindberg and Brunvand described a 12-year-old girl with congenital bilateral mydriasis in association with aneurysmal dilatation of a persistent ductus arteriosus. 496 They attributed these findings to a hypoplastic or aplatic sphincter and ciliary body because there was also absent accommodation. Khan et al reported two patients with dilated pupils with hypoplasia of the iris, Moyamoya angiopathy, dolichoectatic internal carotid arteries, and patent ductus arteriosus. The mechanistic link between the ocular and cardiac defects is unclear. 433 Accommodative Paresis A recent report described five children with a syndrome of benign transient loss of accommodation. No child had other ocular, neurological, or systemic abnormalities that could be associated with accommodative paralysis. 18 All had preserved pupillary responses to light and near, and preserved convergence. All did well with bifocals, and accommodation returned to normal in 3 14 months in all cases. Similar cases of isolated accommodative paresis in otherwise healthy young patients have been previously reported. 163,222 Idiopathic paralysis of the near vision triad has also rarely been described. 449 Organic causes of accommodative paresis include head/ neck trauma, pharmacologic agents, systemic diseases (diphtheria, diabetes mellitus, decompression sickness), neuromuscular disease (myasthenia gravis, myotonic dystrophy, botulism, tetanus), neurologic diseases (dorsal midbrain syndrome, encephalitis, Wilson disease, hemispheric hematoma, oculomotor nerve palsy, Adie tonic pupil), ocular disease (uveitis, trauma, metastasis, glaucoma), and presbyopia. Isolated accommodation palsy can also be functional in origin. Accommodative paresis can be one of the earliest symptoms of dorsal midbrain syndrome, resulting from either hydrocephalus or compression by an extrinsic tumor such as a solid pineal tumor. 592 Blurred vision and isolated accommodative palsy have been reported in patients with symptomatic pineal cysts, 86 which are usually considered to be an incidental radiologic finding ( % of all MR studies). 301,477,677 Accommodative paresis is a common component in children with Down syndrome, leading to the frequent need for bifocals. The extra accommodative effort exerted by these patients may explain the development of esotropia and the common undercorrections experienced after strabismus surgery for this condition. Adie Syndrome Isolated internal ophthalmoplegia is commonly due to trauma or pharmacological dilatation of the pupil or is a feature of Adie syndrome. Adie syndrome is rare in childhood, 498 having an average age of onset of approximately 32 years and a predilection for females. It is characterized by unilaterally or bilaterally enlarged, tonic pupils that show markedly slow constriction to either light or near stimulation, followed by a very slow, tonic, redilatation. Patients with Adie s syndrome also show regional corneal hypesthesia due to interruption of fibers of the ophthalmic division of the trigeminal nerve as they traverse the ciliary ganglion. The light-near dissociation found in Adie syndrome differs in pathophysiology from that seen in mesencephalic disease; it may be attributed to either diffusion of acetylcholine from partially innervated or reinnervated ciliary muscle to the supersensitive pupillary sphincter muscle 343 or by aberrant misdirection to the pupillary sphincters of nerve fibers that originally synapsed in the ciliary muscle. 744 An accommodation paresis may also be associated, which is understandable if one considers that the ciliary ganglion has 30 times more neurons destined for the ciliary muscle than for the iris sphincter. Following acute onset, most of the sprouting new axons arise from accommodative neurons, but many of
55 Pupillary Abnormalities these end up in the iris sphincter (i.e., aberrant regeneration). Hence, although the pupils tend to be large at presentation, they become smaller with the passage of time and may eventually be confused with Argyll Robertson pupils if the examiner is not aware of their earlier dilated status. With the slit lamp, segmental vermiform movements of the iris and sectoral palsy are seen. Sectoral palsy produces shifting of the iris stroma toward the area of active contraction, a phenomenon known as iris streaming. 743 An associated sectoral palsy of the ciliary muscle causes lenticular astigmatism that may blur vision during near tasks. The iris vermiform movements noted at the slit lamp represent normal contractile activity of those iris sectors still innervated by light-responsive neurons. Citing two young adults who were found to have long-standing miotic pupils in association with typical features of Adie s syndrome, Rosenberg 658 has argued that some cases of Adie syndrome primarily present with miotic pupils in a manner analogous to primary aberrant regeneration of the oculomotor nerve. However, the observation of Adie syndrome with dilated pupils in children as young as 4 years 6 (with subsequent development of miosis) argues against this hypothesis. Because oculomotor palsy rarely presents with isolated mydriasis, 495,799 it is important to look closely for an exophoria that increases in adduction and for alternating hyperphorias in vertical gaze to rule out subclinical oculomotor nerve palsy. It is now recognized that cholinergic supersensitivity of the iris sphincter may also develop in oculomotor palsy with pupillary involvement. 391 Other clinical signs usually attributed to postganglionic damage (light-near dissociation, segmental sphincter palsy) can also be seen, 391 so it is important to differentiate Adie pupil from an early or resolving oculomotor nerve palsy. One case of Adie syndrome involved a 4-year-old boy who developed bilateral consecutive, idiopathic Adie syndrome over a follow-up period of 6 years. 6,230 At the age of 4 years, he was diagnosed with right Adie syndrome and was found to have amblyopia, because the associated accommodative difficulty unmasked his latent hyperopia. 6 Examination at age 10 revealed additional myotonic involvement of the left pupil and absent or sluggish deep tendon reflexes. 230 Two children with unilateral congenital tonic pupils were found to have ipsilateral orbital neural-glial hamartoma. 124,298 Two children with Adie pupil have recently been found to have an endodermal cyst of the intracranial oculomotor nerve. 561,799 Tonic pupils have also been described in a child with neuroblastoma and attributed to a paraneoplastic process. 800 Lambert et al 469 reported bilateral tonic pupils in two infants with a congenital neuroblastoma, Hirschsprung disease, and central hypoventilation syndrome. Children with congenital hypoventilation syndrome tend to have miotic pupils with light-near dis sociation, convergence insufficiency, and other defects in autonomic control such as absence of normal variability in heart 363 rate. 294a Whether the tonic pupils result from result from a paraneoplastic disorder or from the effects of a generalized neurocristopathy is unclear. Recently, a case of reversible posterior leukoencephalopathy was reported in association with an Adie s tonic pupil in a 9-year-old boy following measles vaccination. 49 Botulism should also be considered in the differential diagnosis of bilateral tonic pupils. In this setting, the tonic pupils may persist as a chronic condition. 277 Thompson 743 classified patients with tonic pupils into three general categories on the basis of their underlying pathophysiologic disorders: (1) Local pathologic disorders within the orbit that involve the ciliary ganglion (e.g., inflammatory processes such as herpes or sarcoid, trauma). These conditions are commonly unilateral. (2) Neuropathic conditions causing diffuse peripheral or autonomic neuropathy. These include syphilis, diabetes, Guillain Barré syndrome, Ross syndrome, 798 and several hereditary neuropathies, such as Shy Drager and Charcot Marie Tooth diseases. These conditions are typically bilateral. The presence of Adie-like pupils in an infant younger than 1 year of age should raise the possibility of familial dysautonomia (Riley Day syndrome). 294 Cryptogenic tonic pupils, or true Adie syndrome, begins unilaterally, but eventually at least 20% of patients develop the syndrome bilaterally. In addition to the ocular signs, the deep tendon reflexes, especially the knee and ankle jerks, may be diminished or absent. Adie syndrome bears some resemblance to Ross syndrome, which is a rare, presumably degenerative peripheral neuropathy characterized by the triad of unilateral or bilateral tonic pupil, hyporeflexia, and segmental anhidrosis. 798 The recent demonstration of subclinical segmental hypohidrosis in patients with Adie syndrome suggests that the two conditions may be related. 327 The diagnosis of Adie syndrome can be confirmed by demonstrating constriction of the dilated pupil in response to a dilute solution of pilocarpine (0.125%), which confirms the presence of pupillary supersensitivity due to parasympathetic denervation. This supersensitivity may not be demonstrable acutely, with some acute cases failing to constrict even to strong solutions of pilocarpine, making a differentiation from pharmacologic blockade difficult. Pharmacological misadventures are a common cause of isolated pupillary dilation, resulting from instillation of mydriatic agents, exposure to certain plants that contain belladonna or atropine-like alkaloids (e.g., jimsonweed ), or exposure to certain perfumes or cosmetics. The mydriatic pupils associated with ophthalmoplegic migraine, oculomotor palsy with cyclic spasm, botulism, Fisher syndrome, and the dilated pupil accompanying the dorsal midbrain syndrome do not usually cause a diagnostic problem due to the other associated features of these disorders. Various infectious diseases have been associated with pupillary mydriasis or tonic pupils, including herpes zoster, measles, diphtheria, syphilis, pertussis, scarlet fever, smallpox, influenza, and
56 364 7 Complex Ocular Motor Disorders in Children hepatitis, but a history of an infectious illness is usually present. Chickenpox may produce a tonic pupil in children, which may present during the incubation period, the active disease stage, or during convalescence. 297,359,584,650,659 The mydriasis and decreased accommodation in the setting are presumed to reflect direct infectious or inflammatory involvement of the ciliary ganglion; however, the coexistent iridocyclitis with iris stromal vasculitis and sphincter necrosis could also contribute in some cases. The finding of mutton-fat keratoprecipitates and/or sectoral iris stromal atrophy would favor the latter mechanism. Traumatic injury, either to the iris sphincter or to the ciliary nerves (e.g., panretinal photocoagulation, orbital surgery), may also produce pupillary mydriasis or tonic pupils. 76 Some children may present with isolated episodic unilateral or bilateral mydriasis accompanied by head pain 328,832 ; these may represent a variant of ophthalmoplegic migraine or represent a migraine equivalent and are usually self-limited. 773 Horner Syndrome Horner syndrome results from a lesion affecting the sympathetic supply to the eye and may be encountered at any age (Fig. 7.23). The clinical features found on the affected side include the following: (1) 1 2 mm of miosis, with greater anisocoria in dim illumination and a dilation lag. The miosis results from denervation of the sympathetically innervated pupillary dilator muscle. Oculosympathetic denervation of the pupillary dilator muscle can be demonstrated by dimming the ambient light and observing an immediate but transient increase in anisocoria, because the affected pupil does not dilate as rapidly as the normal pupil (dilation lag). (2) Mild upper lid ptosis measuring 1 2 mm (due to denervation Fig Congenital Horner syndrome. Note right upper lid ptosis, right miosis, and mild heterochromia. Right lower lid shows mild reverse ptosis, covering more of cornea than its left counterpart of the sympathetically supplied superior tarsal muscle) and corresponding elevation of the lower eyelid (upside-down ptosis) due to denervation of the lower eyelid retractors. The upside-down ptosis may be confirmed by matching the lower limbus to the lower lid margin in each eye; more scleral showing would be found on the normal side. The resulting narrowing of the palpebral fissure leads to an apparent enophthalmos. (3) Anhidrosis if the lesion is proximal to the carotid bifurcation. Lesions distal to the carotid bifurcation do not affect sympathetic innervation of facial sweat glands. Both acquired and congenital Horner s syndrome rarely causes Harlequin syndrome, a neurocutaneous phenomenon in which one half of the face fails to flush during thermal or emotional stress as a result of damage to vasodilator sympathetic fibers. 101,564 In some cases, however, this condition cannot be attributed to straightforward sympathetic injury. 157 Some children show a different pattern of scalp hair growth, with straight hair on the affected side. 698 Congenital or perinatal Horner syndrome results in failure of the iris to become fully pigmented, resulting in heterochromia, with the ipsilateral iris appearing lighter in color. Iris heterochromia takes several months to develop and may be difficult to detect in infants who normally have lightly colored irides. Wallis et al reported the unusual case of a 20-month-old boy with congenital Horner syndrome, with the darker iris on the affected side attributable to concomitant Waardenburg syndrome. 792 Much less commonly, heterochromia may also follow acquired lesions in adults. 209 Subtle iris heterochromia can sometimes be made more visible by examining the child in sunlight. Some congenital cases may present with ipsilateral facial flushing. 665 Patients with acute Horner syndrome may also exhibit decreased Schirmer response, transient myopia, transient hypotony, and transient conjunctival hyperemia. The last three signs are seen only in acquired cases. The location of the causative lesion along the sympathetic pathway may be inferred clinically and confirmed pharmacologically and/or neuroradiologically. 210 For instance, the combination of Horner syndrome and ipsilateral abducens palsy implicates a lesion in the cavernous sinus. Mesencephalic lesions, involving the trochlear nucleus or fascicles before decussation in the superior medullary velum and adjacent sympathetic fibers, may produce an ipsilateral Horner syndrome and contralateral superior oblique muscle paresis. 322 Pharmacological testing consists of topical instillation of autonomically active drugs to confirm oculosympathetic paralysis (cocaine test) and to distinguish a preganglionic from a postganglionic lesion (hydroxyamphetamine test). However, a preganglionic congenital Horner syndrome may show a false-positive hydroxyamphetamine test because of transynaptic degeneration. 216 Recently, apraclonidine 0.5% has been touted as a diagnostic test for Horner syndrome on the basis of its alpha-2
57 Pupillary Abnormalities inhibitory effects in normal eyes and weak alpha-1 adrenergic/excitatory effects in Horner eyes. Apraclonidine makes the Horner pupil dilate and the normal pupil constrict slightly. 154,416 The reversal of anisocoria is more obvious with the room lights on. 155 Apraclonidine is reported to be safe and effective in the pediatric population because it does not cross the blood brain barrier, thereby reducing the incidence of centrally mediated effects. 155 However, a recent report documenting CNS and respiratory depression in three children (one requiring intubation) suggests that its use in children should probably be avoided. 634 Iris pigmentation, which occurs within the first year of life, requires sympathetic stimulation. Interestingly, Mindel et al 549 reported a 21-year-old woman with neurofibromatosis type I who had a unilateral congenital Horner syndrome with heterochromia. The patient showed symmetric Lisch nodules, which are melanocytic hamartomas, in both eyes, suggesting that, unlike iris pigmentation, the formation of Lisch nodules is not influenced by sympathetic innervation of the iris. Other causes of heterochromia, such as iris nevus or melanoma, neurofibromatosis, hemosiderosis, hemochromatosis, Waardenburg syndrome, Fuch heterochromic iridocyclitis, and essential iris atrophy, should be excluded. Most cases of congenital or early acquired Horner syndrome are of a benign nature. 289,399,814 Rarely, congenital Horner syndrome may occur as an autosomal dominant disorder. Hageman et al 324 reported a Dutch family with five cases of congenital Horner syndrome spanning five generations. A history of perinatal trauma, such as brachial plexus injury (Klumpke paralysis), 20,797 neck or cardiothoracic surgery, carotid dissection, 319,648 peritonsilar lesions, and surgical or nonsurgical intraoral trauma, may be elicited in many cases, but some cases remain cryptogenic despite extensive investigations. Permanent postganglionic Horner syndrome can develop in children with a middle ear infection. 370,716 Brachial plexopathies caused by forceps injury at birth are the putative cause in some cases. 101 MR angiography has recently brought to light the association of congenital Horner syndrome with agenesis of the ipsilateral internal carotid artery. 213,388,662 Congenital Horner syndrome has also been reported with hemifacial atrophy, synergistic divergence, basilar impression and Chiari malformation, cervical vertebral anomaly and an enterogenous cyst, and viral infections such as congenital varicella 469 or cytomegalovirus infections, and PHACE syndrome. 531 In acquired pediatric Horner syndrome, it is important to look for Lisch nodules and café au lait spots, because schwannomas, plexiform neurofibromas, or malignant peripheral nerve sheath tumors may affect the sympathetic tract in neurofibromatosis A case of congenital postganglionic Horner syndrome and fibromuscular dysplasia of the ipsilateral internal carotid artery incited speculation that prenatal or neonatal cervical trauma might have been responsible for both findings Congenital tumors, including neuroblastoma of the neck, chest, or abdomen have been reported to underlie some cases of congenital Horner syndrome. 143,676,678,814,830 Most reported cases of neuroblastoma occurred in the neck, 2 where they are frequently mistaken for infectious adenitis, or in the mediastinum, 703 but three patients with abdominal neuroblastoma have also been described. 290,572 Because the cervical sympathetic chain does not descend to the abdominal level, the Horner syndrome in these cases was probably caused by metastatic cervical lesions or a second (undiagnosed) primary lesion. Cervical neuroblastoma causing Horner syndrome can be associated with other paraneoplastic disorders such as cerebellar degeneration. 237,810 Other systemic clues to the presence of neuroblastoma may be present. Nonophthalmologic symptoms of neuroblastoma include pain (due to primary tumor, bone marrow involvement, or abdominal distension), watery diarrhea (due to paraneoplastic secretion of a vasoactive intestinal peptide), 552 and acute cerebellar encephalopathy, 99 which may be related to antineural antibodies such as anti-hu, formed in response to the tumor that react to the normal cerebellum. 269 There is an unusually high rate of spontaneous regression in the neuroblastoma of young infants, even in the disseminated form. 523 Asymptomatic infants may have biologically favorable tumor with higher rate of spontaneous regression. 523 Horner syndrome acquired in early life may be very difficult to distinguish from congenital cases, but close scrutiny of previous photographs may be helpful. In a large retrospective study, Mahoney et al 510 found responsible mass lesions in six of 56 children. Two cases with congenital Horner syndrome were caused by mass lesions (neuroblastoma in one case, neurofibroma in the other). They recommended that the brain, neck, and chest MRI with and without contrast, as well as urinary catecholamine metabolite testing, be performed in any child without a surgical history. This study found direct imaging to be more sensitive than urine testing in this setting. Gibbs and colleagues 290 reported a 2-year-old child with congenital Horner syndrome who was healthy until the age of 2 years, when a remote neuroblastoma of the adrenal gland was diagnosed. They argued that both congenital Horner syndrome and neuroblastoma may represent widespread dysgenesis of the sympathetic system. The rare occurrence of underlying tumors, especially neuroblastoma, in a few congenital cases justifies thorough neurodiagnostic evaluation. Mahoney et al 510 recommended palpation of neck, axilla, and abdomen, spot urine for homovanillic acid and vanillylmandelic acid as a ratio of creatinine (which can be falsely negative in patients with small neuroblastomas), and MR imaging of the brain, neck, and chest, with and without contrast (abdomen is unnecessary because the cervical sympathetic chain does not descend to this level). Because of the recognized association of congenital Horner syndrome with ipsilateral agenesis of the internal
58 366 7 Complex Ocular Motor Disorders in Children carotid artery, we believe that MR angiography should also be included in the evaluation. While it has been suggested that a history of forceful manipulation of the neck during birth may reduce the need for extensive systemic evaluation, 399 the possibility of carotid dissection should now be considered and evaluated by MR angiography in this setting. 319 References 1. Abeloos MC, Cordonnier M, Van-Nechel C, et al. Congenital fibrosis of the ocular muscles: a diagnosis for several clinical pictures. Bull Soc Belge Ophtalmol. 1990;239: Abramson SJ, Berdon WE, Ruzal-Shapiro C, et al. Cervical neuroblastoma in eleven infants a tumor with favorable prognosis. Clinical and radiologic (US, CT, MRI) findings. Pediatr Radiol. 1993;23: Acheson JF, Elston JS, Lee JP, et al. Extraocular muscle surgery in myasthenia gravis. Br J Ophthalmol. 1990;75: Adams WE, Leavitt JA, Holmes JM. Straibmus surgery for internuclear ophthalmoplegia with exotropia in multiple sclerosis. J AAPOS. 2009;13: Adams C, Theodorescu D, Murphy EG, et al. Thymectomy in juvenile myasthenia gravis. J Child Neurol. 1990;5: Agbeja AM, Dutton GN. Adie s syndrome as a cause of amblyopia. J Pediatr Ophthalmol Strabismus. 1987;24: Aghaji MA, Uzuegbunam C. Invasive thymoma and myasthenia gravis in a three-and-a-half-year-old boy: case report and literature review. Cent Afr J Med. 1990;36: Ahmed A. Bilateral congenital vertical gaze disorders: congenital muscle fibrosis or congenital central nervous system abnormality? Neuroophthalmology. 1997;1: Ahmed A. Bilateral congenital vertical gaze disorders: congenital muscle fibrosis or a congenital central nervous system abnormality? In: Transactions 20th Meeting. European Strabismological Association, Brussels; May Ahmed A. Congenital innervation defect syndrome (1998) In: Lennerstrand, G, ed. Advances in Strabismology. Amsterdam: Aeolus Press; Ahn JC, Hoyt WF, Hoyt CS. Tonic upgaze in infancy. A report of three cases. Arch Ophthalmol. 1989;107: Aicardi J, Barbosa C, Andermann E, et al. Ataxia-ocular motor apraxia: a syndrome mimicking ataxia-telangiectasia. Ann Neurol. 1988;24: Akdal G, Yener GG, Ada E, et al. Eye movement disorders in vitamin B12 deficiency: two new cases and a review of the literature. Eur J Neurol. 2007;14: Al-Baradie R, Yamada K, St. Hilaire C, et al. Duane radial ray syndrome (Okihiro syndrome) maps to 20q13 and results from mutations in SALL4, a new member of the SAL family. Am J Hum Genet. 2002;71: Albert DM, Wong VG, Henderson ES. Ocular complications of vincristine therapy. Arch Ophthalmol. 1967;78: Al-Din AN, Anderson M, Bickerstaff ER, et al. Brainstem encephalitis and the syndrome of Miller Fisher: a clinical study. Brain. 1982;105: al-din SN, Anderson M, Eeg-Olofsson O, et al. Neuro-ophthalmic manifestations of the syndrome of ophthalmoplegia, and areflexia: a review. Acta Neurol Scand. 1994;89: Almog Y. A benign syndrome of transient loss of accommodation in young patients. Arch Ophthalmol. 2008;126: Al-Qurainy IA. Convergence insufficiency and failure of accommodation following midfacial trauma. Br J Oral Maxillofac Surg. 1995;33: al-rajeh S, Corea JR, al-sibai MH, et al. Congenital brachial palsy in the eastern province of Saudi Arabia. J Child Neurol. 1990;5: Al-Shahwan SA, Singh B, Riela AR, et al. Hemisomatic spasms in children. Neurology. 1994;44: a. Amaya LG, Walker J, Taylor D. Möbius syndrome: A study and report of 18 cases. Binoc Vis Q 1990;5: Ames WA, Shichor TM, Speakman M, et al. Anesthetic management of children with Möbius sequence. Can J Anaesth. 2005;52: Anderson JM, Brodsky MC. Anticholinergic esotropia: a cautionary note to strabismus surgeons. J Neuroophthalmol. 2008;28: Andrews PI. Autoimmune myasthenia gravis in childhood. Semin Neurol. 2004;24: Andrews PI, Massey JM, Howard JF, et al. Race, sex, and puberty influence onset, severity, and outcome in juvenile myasthenia gravis. Neurology. 1994;44: Andrews PI, Massey JM, Sanders DB. Acetylcholine receptor antibodies in juvenile myasthenia gravis. Neurology. 1993;43: Anteby I, Lee B, Noetzel M, Tychsen L. Variants of congenital ocular motor apraxia: associations with hydrocephalus, pontocerebellar tumor, and a deficit of vertical saccades. J AAPOS. 1997;1: Appleton RE, Panayiotopoulos CP, Acomb BA, et al. Eyelid myoclonia with typical absences: an epilepsy syndrome. J Neurol Neurosurg Psychiatry. 1993;56: Apt L, Axelrod RN. Generalized fibrosis of the extraocular muscles. Am J Ophthalmol. 1978;85: Arakawa Y, Yoshimura M, Kobayashi S, et al. The use of intravenous immunoglobulin in Miller Fisher syndrome. Brain Dev. 1993;15: Archer SM, Helveston EM, Miller KK, et al. Stereopsis in normal infants and infants with congenital esotropia. Am J Ophthalmol. 1986;101: Arnoldi K, Jackson JH. Cerebral palsy for the pediatric eye care team: epidemiology, pathogenesis, and systemic findings. Am Orthopt J. 2005;55: Arnoldi K, Reynolds JD. A review of convergence insufficiency: what are we really accomplishing with exercises? Am Orthopt J. 2007;57: Arnon S, Chin J. The clinical spectrum of infant botulism. Rev Infect Dis. 1979;1: Arnon S, Damus K, Chin J. Infant botulism: epidemiology and relation to sudden infant death syndrome. Epidemiol Rev. 1981;3: Arnon S, Midura T, Clay S, et al. Infant botulism: epidemiological, clinical, and laboratory aspects. JAMA. 1977;237: Arnon SS, Midura TF, Damus K, et al. Intestinal infection and toxin productions by Clostridium botulinum as one cause of sudden infant death syndrome. Lancet. 1978;i: Arnon S, Midura T, Damus K, et al. Honey and other environmental risk factors for infant botulism. J Pediatr. 1979;94: Arnon SS, Schechter R, Maslanka SE, et al. Human botulism immune globulin for the treatment of infant botulinum. N Engl J Med. 2006;354: Arroyo-Yllanes ME, Manzo Villalobos G, Perez-Perez JF, et al. Strabismus in patients with cerebral palsy. Am Orthopt J. 1999;49: Arzimanoglou AA, Salefranque F, Goutieres F, et al. Hemifacial spasm or subcortical epilepsy? Epileptic Disord. 1999;1: Ashwal S, Thrasher TV, Rice DR, et al. A new form of sea-blue histiocytosis associated with progressive anterior horn cell and axonal degeneration. Ann Neurol. 1984;16: Askanas V, McFerrin J, Park-Matsumoto YC, et al. Glucocorticoid increases acetylcholinesterase and organization of the postsynaptic membrane in innervated cultured human muscle. Exp Neurol. 1992;115: Assaf AA. Bilateral vertical gaze disorders: congenital muscle fibrosis or congenital central nervous abnormality? Neuroophthalmology. 1997;1:23 30.
59 References 45. Assaf AA. Bilateral congenital vertical gaze disorders: congenital muscle fibrosis or a congenital central nervous system abnormality? In: Kaufman H, ed. Transactions of the European Strabismological Association, Brussels; May Auger RG, Piepgaras DG. Hemifacial spasm associated with epidermoid tumors of the cerebello-pontine angle. Neurology. 1989;39: Auré K, de Baulny HO, Leforêt P, et al. Chronic progressive ophthalmoplegia with large-scale MtDNA rearrangement: can we predict progression? Brain. 2007;130: Avilla CW. Myasthenia gravis. Am Orthopt J. 2005;55: Aydin K, Elmas S, Guzes EA. Reversible posterior leukoencephalopathy after measles vaccination. J Child Neurol. 2006; 21: Aylett SE, Cross H, Berry D. Eye rolling as a manifestation of clobazam toxicity in a child with epilepsy. Dev Med Child Neurol. 2006;48: Bain KE, Beatty S, Lloyd C. Non-organic visual loss in children. Eye. 2000;5: Baker R, Buncic JR. Vertical ocular motility disturbance in pseudotumor cerebri. J Clin Neuroophthalmol. 1985;5: Baloh RW, Yee RD, Boder E. Eye movements in ataxia-telangiectasia. Neurology. 1978;28: a. Ban R, Matsuo D, Osada Y, et al. Reflexive contraction of the levator palpebrae superioris muscle to involuntarily sustain the effective eyelid retraction through the transverse trigeminal proprioceptive nerve on the proximal Mueller's muscle: verification with evoked electromyography. J Plast Reconstr Aesthet Surg. 2007:Nov 13: Bandim JM, Ventura LO, Miller MT, et al. Autism and Möbius sequence: an exploratory study of children in northeastern Brazil. Arq Neuropsiquiatr. 2003;61: Barbot C, Coutinho P, Chorão R, et al. Recessive ataxia with ocular apraxia: review of 22 patients. Arch Neurol. 2001;58: Barontini F, Simonettia C, Ferranini F, Sita D. Persistent upward eye deviation: report of two cases. Neuroophthalmology. 1983; 3: Barroso L, Hoyt WF. Episodic exotropia from lateral rectus neuromyotonia appearance and remission after radiation therapy for a thalamic glioma. J Pediatr Ophthalmol Strabismus. 1993;30: Barton JJ, Fouladvand M. Ocular aspects of myasthenia gravis. Semin Neurol. 2000;20: Bau V, Zierz S. Update on chronic progressive external ophthalmoplegia. Strabismus. 2005;13: Review. 59a. Bavinck JN, Waver DD. Subclavian artery supply disruption sequence: hypothesis of a vascular etiology for Poland, Klippel- Feil, and Möbius anomalies. Am J Med Genet. 1986;23: Beeson D, Webster R, Cossins J, et al. Congenital myasthenic syndromes and formation of the neuromuscular junction. Ann N Y Acad Sci. 2008;1132: Beigi B, O Keeffe M, Logan P, et al. Convergence substitution for paralyzed horizontal gaze. Br J Ophthalmol. 1995;79: Bell JA, Fielder AR, Viney S. Congenital double elevator palsy in identical twins. J Clin Neuroophthalmol. 1990;10: Benevento WJ, Tychsen L. Distinguishing compensatory head turn from gaze palsy in children with unilateral oculomotor or abducens nerve paresis. Am J Ophthalmol. 1993;115: Letter. 64. Bentley CR, Dawson E, Lee JP. Active management of patients with ocular manifestations of myasthenia gravis. Eye. 2001;15: Berciano J, Boesch S, Pérez-Ramos JM. Olivopontocerebellar atrophy: toward a better nosological definition. Mov Disord. 2006;21: Berlit P, Rakicky J. The Miller Fisher syndrome. Review of the literature. J Clin Neuroophthalmol. 1992;12: Bernasconi O, Borruat FX. Unilateral accommodation spasm: a diagnostic pitfall! Klin Monatsbl Augenheilkd. 1998;212: Betharia SM, Sharma V. Inverse Bell s phenomenon observed following levator resection. Graefes Arch Clin Exp Ophthalmol. 2006;244: Bever CT, Aquino AV, Penn AS, et al. Prognosis of ocular myasthenia gravis. Ann Neurol. 1983;14: Beyer-Machule CK, Johnson CC, Pratt SG, et al. The Marcus Gunn phenomenon. Orbit. 1985;4: Bianchi PE, Salati R, Cavallini A, et al. Transient nystagmus in delayed visual maturation. Dev Med Child Neurol. 1998;40: Bickerstaff E. Brain stem encephalitis. Further observations on a grave syndrome with benign prognosis. BMJ. 1957;ii: Binyon S, Prendergast M. Eye-movement tics in children. Dev Med Child Neurol. 1991;33: Biousse V, Newman NJ. Neuro-ophthalmology of mitochondrial diseases. Curr Opin Ophthalmol. 2003;16: Biousse V, Skibell BC, Watts RL, et al. Ophthalmologic features of Parkinson s disease. Neurology. 2004;62: Bodker FS, Cytryn AS, Putterman AM, et al. Postoperative mydriasis after repair of orbital floor fracture. Am J Ophthalmol. 1993;115: Boehme BI, Graef MH. Acquired segmental iris dilator muscle synkinesis due to deglutition. Arch Ophthalmol. 1998;116: Boergen KP, Lorenz B, Müller-Höcker J. Das Kongenitale Fibrosesyndrome. berlegeungen zur Ätiologie, Genetik und Chirurgischen. Therapie. Klin Monatsbl Augenheilkd. 1990;197: Bogousslaysky J, Miklossy J, Regli F, et al. Vertical gaze palsy and selective unilateral infarction of the rostral interstitial nucleus of the medial longitudinal fasciculus (rimlf). J Neurol Neurosurg Psychiatry. 1990;53: Bogousslaysky J, Regli F. Upgaze palsy and monocular paresis of downward gaze from ipsilateral thalamo-mesencephalic infarction: a vertical one-and-a-half syndrome. J Neurol. 1984;231: Bohlmann BJ, France TD. Persistent accommodative spasm nine years after head trauma. J Clin Neuroophthalmol. 1987;7: Bolanos I, Lozano D, Cantu C. Internuclear ophthalmoplegia: causes and long-term follow-up in 65 patients. Acta Neurol Scand. 2004;110: Bora I, Karh N, Bakar M, et al. Myasthenia gravis following IFNa-2a treatment. Eur Neurol. 1997;38: Borchert MS, Sadun AA, Sommers JD, et al. Congenital ocular motor apraxia in twins: findings with magnetic resonance imaging. J Clin Neuroophthalmol. 1987;7: Borenstein S, Desmedt JE. Temperature and weather correlates of myasthenic fatigue. Lancet. 1974;2: Borraut F-X, Kawasaki A. Isolated accommodation palsy associated with pineal cyst. Report of a case and review of the literature. Neuroophthalmology. 2007;31: Bosley TM, Oystreck DT, Robertson RL, et al. Neurologic features of congenital fibrosis of the extraocular muscles type 2 with mutations in PHOX2A. Brain. 2006;129: Bosley TM, Salih MA, Jen JC, et al. Neurologic features of horizontal gaze palsy and progressive scoliosis with mutations in ROBO3. Neurology. 2005;64: Braddock SR, Henley KM, Maria BL. The face of Joubert syndrome: a study of dysmorphology and antropometry. Am J Med Genet. 2007;143: Brady McCreery KM, Hussein MA, Lee AG, et al. Pediatric myasthenia gravis: blepharoptosis, ophthalmoplegia and strabismus. A report of 14 cases. Binocul Vis Strabismus Q. 2002;17:
60 368 7 Complex Ocular Motor Disorders in Children 91. Brancati F, Barrano G, Silhavy JL, et al. CPE290 mutations are frequently identified in the oculo-renal form of Joubert syndromerelated disorders. Am J Hum Genet. 2007;81: Brandt T, Dieterich M. Pathological eye-head coordination in roll: tonic ocular tilt reaction in mesencephalic and medullary lesions. Brain. 1987;110: Brandt TH, Dieterich M. Different types of skew deviation. J Neurol Neurosurg Psychiatry. 1991;54: Brandt T, Dieterich M. Skew deviation with ocular torsion: a vestibular brain stem sign of topographic diagnostic value. Ann Neurol. 1993;33: Brandt T, Dieterich M. Vestibular syndromes in the roll plane: topographic diagnosis from brainstem to cortex. Ann Neurol. 1994;36: Brandt T, Dieterich M, et al. Central vestibular syndromes in roll, pitch, and yaw planes. Neuroophthalmology. 1995;19: Branley MG, Wright KW, Borchert MS. Third nerve palsy due to cerebral artery aneurysm in a child. Aust N Z J Ophthalmol. 1992;20: Braun S, Askan V, Engel WK, et al. Long-term treatment with glucocorticoids increases synthesis and stability of junctional acetylcholine receptors on innervated cultured human muscle. J Neurochem. 1993;60: Bray PF, Ziter FA, Lahey ME, et al. The coincidence of neuroblastoma and acute cerebellar encephalopathy. J Pediatr. 1969;75: Breen L, Morris HH, Alperin JB, et al. Juvenile Niemann Pick disease with vertical supranuclear ophthalmoplegia. Two case reports and review of the literature. Arch Neurol. 1981;38: Bremner F, Smith S. Pupillographic findings in 39 consecutive cases of Harlequin syndrome. J Neuroophthalmol. 2008;28: Brodsky MC. Platysma-levator synkinesis in congenital third nerve palsy. Arch Ophthalmol. 1991;109: Brodsky MC. Surgical management of the congenital fibrosis syndrome. Am Orthopt J. 1997;47: Brodsky MC. Hereditary external ophthalmoplegia, synergistic divergence, jaw winking, and oculocutaneous hypopigmentation. A congenital fibrosis syndrome caused by deficient innervation to extraocular muscles. Ophthalmology. 1998;105: Brodsky MC. Dissociated vertical divergence: a righting reflex gone wrong. Arch Ophthalmol. 1999;117: Brodsky MC. The Doctor s eye: seeing through the myopathy of congenital ptosis. Ophthalmology. 2000;107: Brodsky MC. Primary oblique muscle overaction: the brain throws a wild pitch. Arch Ophthalmol. 2001;119: Brodsky MC. Dissociated vertical divergence: perceptual correlates of the human dorsal light reflex. Arch Ophthalmol. 2002;102: Brodsky MC. Do you really need your oblique muscles? Adaptations and exaptations. Arch Ophthalmol. 2002;120: Brodsky MC. Visuo-vestibular eye movements. Infantile strabismus in three dimensions. Arch Ophthalmol. 2005;123: Brodsky MC. Myasthenic ptosis with synkinetic override in a child with congenital oculomotor nerve palsy. J AAPOS. 2006;10: Brodsky MC. Does infantile esotropia arise from a dissociated deviation? Arch Ophthalmol. 2008;125: Brodsky MC. Vertical strabismus: diagnosis from the ground up. Arch Ophthalmol. 2008;126: Brodsky MC, Boop FA. Lid nystagmus in diffuse ophthalmoplegia as a sign of intrinsic midbrain disease. J Neuroophthalmol. 1995;15: Brodsky MC, Donahue SP. Primary oblique muscle overaction: the brain throws a wild pitch. Arch Ophthalmol. 2001;119: a. Brodsky MC. Congenital downbeat nystagmus. J Pediatr Ophthamol Strabismus 1996;33: Brodsky MC, Donahue SP, Vaphiades M, et al. Skew deviation revisited. Surv Ophthalmol. 2006;51: Brodsky MC, Fray KJ. Does infantile esotropia arise from a dissociated deviation? Arch Ophthalmol. 2007;125: Brodsky MC, Fray KJ, Glasier CM. Perinatal cortical and subcortical visual loss: mechanisms of injury and associated ophthalmologic signs. Ophthalmology. 2002;109: Brodsky MC, Fritz KJ. Hereditary congenital exotropia: a report of three cases. Binocul Vis Eye Muscle Surg Q. 1993;8: Brodsky MC, Pollock SC, Buckley EG. Neuronal misdirection in congenital ocular fibrosis syndrome: implications and pathogenesis. J Pediatr Ophthalmol Strabismus. 1989;26: Brodsky MC, Sharp GB, Fritz KJ, et al. Idiopathic alternating anisocoria. Am J Ophthalmol. 1992;114: Letter Brodsky MC, Thomas AH. Accommodative esotropia: an unrecognized cause of hemifacial spasm. Dev Med Child Neurol. 2001;43: Brodsky MC, Tusa RJ. Latent nystagmus: vestibular nystagmus with a twist. Arch Ophthalmol. 2004;122: Brooks-Kayal AR, Liu GT, Menacker SJ, et al. Tonic pupil and orbital glial neural hamartoma in infancy. Am J Ophthalmol. 1995;119: Brown B. The convergence insufficiency masquerade. Am Orthopt J. 1990;40: Bruke JP, Shipman TC, Watts MT. Convergence insufficiency in thyroid eye disease. J Pediatr Ophthalmol Strabismus. 1993;30: Bruyn GW. Huntington s chorea. In: Vinker PT, Bruyn GW, eds. Handbook of Clinical Neurology. Diseases of the Basal Ganglia, vol. 6. Amsterdam: NVK-Holland Publishing; 1968: Buckley EG, Holgado S. Surgical treatment of upgaze palsy in Parinaud s syndrome. J AAPOS. 2004;8: Buckley E, Seaber JH. Dyskinetic strabismus as a sign of cerebral palsy. Am J Ophthalmol. 1981;91: Buncic JR. Systemic and pharmacological effects on near vision. Am Orthopt J. 1999;49: Burgett RA, Kawasaki A. Mesencephalic clefts and eye movement disorders. Arch Ophthalmol. 1997;115: Burk K, Abele M, Fetter M, et al. Autosomal dominant cerebellar ataxia type 1: clinical features and MRI in families with SCA1, SCA2, and SCA3. Brain. 1996;119: Burk K, Fetter M, Abele M, et al. Autosomal dominant cerebellar ataxia type I: oculomotor abnormalities in families with SCA1, SCA2, and SCA3. Brain. 1996;246: Burke JP, Orton HP, West J, et al. Whiplash and its effecton the visual system. Graefes Arch Clin Exp Ophthalmol. 1992;230: Burke JP, Ruben JB, Scott WE. Vertical transposition of the horizontal recti (Knapp procedure) for the treatment of double elevator palsy: effectiveness and long-term stability. Br J Ophthalmol. 1992;76: Buttner N, Geschwind D, Jen JC, et al. Oculomotor phenotypes in autosomal dominant ataxias. Arch Neurol. 1998;55: Cackett P, Vallance J, Bennett H. Neurofibromatosis type 1 presenting with Horner s syndrome. Eye. 2005;19: Cadera W, Bloom JN, Karlik S, et al. A magnetic resonance imaging study of double elevator palsy. Can J Ophthalmol. 1997;32: Calcutt C, Murray A. A and V patterns in essential infantile esotropia and their association with DVD. In: Lennerstrand G, ed. Update on Strabismus and Paediatric Ophthalmology. Proceedings of the Seventh Meeting of the International Strabimological Association. Boca Raton, FL: CRC Press; 1995: Calcutt C, Murray AD, Orpen JO, et al. Changes in the functional binocular status of older children and adults with previously untreated infantile esotropia following late surgical realignment. J AAPOS. 2007;11: Caldeira JA. Vertical transposition of the horizontal rectus muscles for congenital/early onset acquired double elevator palsy: a retro-
61 References spective long term study of 10 consecutive patients. Binocul Vis Strabismus Q. 2000;15: Callahan MA. Surgically mismanaged ptosis associated with double elevator palsy. Arch Ophthalmol. 1981;99: Cardesa-Salzmann TM, Mora-Graupera J, Claret G, et al. Congenital cervical neuroblastoma. Pediatr Blood Cancer. 2004;43: Carruthers JD. Strabismus in craniofacial dysostosis. Graefes Arch Clin Exp Ophthalmol. 1988;226: Castro O, Johnson LN, Mamourian AC. Isolated inferior oblique paresis from brain stem infarction: perspective on oculomotor fascicular organization in the ventrual midbrain tegmentum. Arch Ophthalmol. 1990;47: Catalano RA, Calhoun JH, Reinecke RD, Cogan DG. Asymmetry in congenital ocular motor apraxia. Can J Ophthalmol. 1988;23: Catalano RA, Trevisani MG, Simon JW. Functional eyelid pulling in children. Am J Ophthalmol. 1990;110: Cattaneo L, Chieriei E, Bianchi B, et al. The localization of facial motor impairment in sporadic Möbius syndrome. Neurology. 2006;66: Centers for Disease Control and Prevention. Cluster of tick paralysis cases Colorado. MMWR Morb Mortal Wkly Rep. 2006;55: Chan WM, Andrews C, Dragan L, et al. Three novel mutations in KIF21A highlight the importance of the third coiled-coil stalk domain in the etiology of CFEOM1. BMC Genet. 2007;8: Chan RV, Trobe JD. Spasm of accommodation associated with closed head trauma. J Neuroophthalmol. 2002;22: Chan W, Wong GWK, Fan DSP, et al. Ophthalmopathy in childhood Graves disease. Br J Ophthalmol. 2002;86: Chang JW, Chang JH, Park YG, et al. Microvascular decompression of the facial nerve for hemifacial spasm in youth. Childs Nerv Syst. 2001;17: Chen PL, Chen JT, Lu DW, et al. Comparing efficacies of 0.5% apraclonidine with 4% cocaine in the diagnosis of Horner syndrome in pediatric patients. J Ocul Pharmacol Ther. 2006;22: Chen PC, Hsiao C-H, Chen J-T, et al. Efficacy of Apraclonidine 0.5% in the diagnosis of Horner syndrome in pediatric patients under low or high illumination. Am J Ophthalmol. 2006; 142: Cheng H, Burdon MA, Shun-Sin GA, et al. Dissociated eye movements in craniosynostosis: a hypothesis revived. Br J Ophthalmol. 1993;77: Cheshire WP, Low PA. Harlequin syndrome: still only half understood. J Neuroophthalmol. 2008;28: Cheung T, Gou W, Huo DM. Skew ocular deviation: a catastrophic sign on MRI of fetal glioblastoma. Childs Nerv Syst. 2003;19: Chiba A, Kusunoki S, Obata H, et al. Serum anti-gq1b IgG antibody is associated with ophthalmoplegia in Miller Fisher syndrome and Guillain Barré syndrome: clinical and immunohistochemical studies. Neurology. 1993;43: Choi KD, Hwang JM, Park SH, et al. Primary aberrant regeneration and neuromyotonia of the third cranial nerve. J Neuroophthalmol. 2006;26: Chomette G, Auriol M, Guilbert F, et al. Cherubism: histoenzymological and ultrastructural study. Int J Oral Maxillofac Surg. 1988;17: Chomyn A, Enriquez JA, Micol V, et al. The mitochondrial myopathy, encephalopathy, lactic acidosis, and stroke-like episodes syndrome-associated human mitochondrial trnaleu (UUR) mutation causes aminoacylation deficiency and concomitant reduced association of mrna with ribosomes. J Biol Chem. 2000;275: Chrousos GA, O Neill JF, Lueth BD, et al. Accommodation deficiency in healthy young individuals. J Pediatr Ophthalmol Strabismus. 1988;25: Chuang DC. Free tissue transfer for the treatment of facial paralysis. Facial Plast Surg. 2008;24: Chung S, Gerber P, Kirlin KA. Ictal eye closure is a reliable indicator for psychogenic nonepileptic seizures. Neurology. 2006;66: Churchyard A, Stell R, Mastaglia FL. Ataxia telangiectasia presenting as an extrapyramidal movement disorder and ocular motor apraxia without overt telangiectasia. Clin Exp Neurol. 1991;28: Ciancia A, Fino M. Development of A and V anisotropia in surgically treated patients with congenital esotropia. Arch Chil Oftal. 2006;63: Cibis G, Kies R, Lawwill T, et al. Electromyography in congenital famialial ophthalmoplegia. In: Reinecke RD, ed. Strabismus II: Proceedings of the Fourth Meeting at the International Strabismological Association, Oct 25 29, 1982, Asilomar, Calif. Orlando, FL: Grune & Stratton; Clark RA, Miller JM, Demer JL. Heterotopic muscle pulleys or oblique muscle dysfunction? J AAPOS. 1998;2: Clarke RR, Van der Velde RL. Congenital myasthenia gravis. A case report with thymectomy and electron microscopic study of resected thymus. Am J Dis Child. 1971;122: Coats DK, Paysse EA, Kim DS. Excessive blinking in childhood: a prospective evaluation of 99 children. Ophthalmology. 2001;108: Coats DK, Paysse EA, Stager DR. Surgical management of V-pattern strabismus and oblique dysfunction in craniofacial dysostosis. J AAPOS. 2000;4: Cogan DC. Neurology of Eye Muscles. 2nd ed. Springfield, IL: Charles C Thomas; 1958: Cogan DG. Congenital ocular motor apraxia. Can J Ophthalmol. 1966;1: Cogan DG. Heredity of congenital ocular motor apraxia. Trans Am Acad Ophthalmol Otolaryngol. 1972;76: Cogan DG, Chu FC, Bachman DM, et al. The DAF syndrome. Neuroophthalmology. 1981;2: Cogan DG, Chu FC, Reingold D, et al. A long term follow-up of congenital ocular motor apraxia: case report. Neuroophthalmology. 1980;1: Cogan DG, Freese CG. Spasm of the near reflex. Arch Ophthalmol. 1955;54: Cogan DG, Schulman J, Porter RJ, Mudd SH. Epileptiform ocular movements with methylmalonic aciduria and homocystinuria. Am J Ophthalmol. 1980;90: Cogan DG, Wray SH. Internuclear ophthalmoplegia as an early sign of brain stem tumors. Neurology. 1970;20: Cohen M, Groswasser Z, Barchadski R, et al. Convergence insufficiency in brain-injured patients. Brain Inj. 1989;3: Collier J. Nuclear ophthalmoplegia with special reference to retraction of the lids and ptosis and to lesions of the posterior commissure. Brain. 1927;50: Collin JR, Allen L, Castronuovo S. Congenital eyelid retraction. Br J Ophthalmol. 1990;74: Copeland WC. Inherited mitochondrial diseases of DNA replication. Annu Rev Med. 2008;59: Corbett J. Neuro-ophthalmic complications of hydrocephalus and shunting procedures. Semin Neurol. 1986;6: Corbett JA, Mathews AM, Connell PH, et al. Tics and Gilles de la Tourette syndrome: a follow up study and critical review. Br J Psychiatry. 1969;115: Coutinho P, Andrade C. Autosomal dominant system degeneration in Portuguese families of the Azores Islands. A new genetic disorder involving cerebellar, pyramidal, extrapyramidal and spinal cord motor functions. Neurology. 1978;28: Covanis A. Photosensitivity in idiopathic generalized epilepsies. Epilepsia. 2005;46:67 72.
62 370 7 Complex Ocular Motor Disorders in Children 188a. D Cruz OF, Swisher CN, Jaradeh S, et al. Möbius syndorme: evidence for a vascular etiology. J Child Neurol 1993;8: Criscuolo C, Chessa L, Di Giandomenico S, et al. Ataxia with oculomotor apraxia type 2: a clinical, pathologic, and genetic study. Neurology. 2006;66: Critchley EM, Mitchell JD. Human botulism. Br J Hosp Med. 1990;43: Cronemberger MF, de Castro Moreira JB, Brunoni D, et al. Ocular and clinical manifestations of Möbius syndrome. J Pediatr Ophthalmol Strabismus. 2001;38: Crouch ER, Goodrich-Snyder K, Cunningham P. Permanent sixth nerve paralysis in infantile botulism. Am Orthopt J. 1995;45: Cruysberg JR. Congenital dysinnervation syndromes: new understanding of clinical manifestations. In: EUPO 2008, Geneva, Switzerland; 2008: Cruysberg JR, Willemsen MA, van Moli-Ramirez NG, et al. The overlooking phenomenon of children with neuronal ceroid lipofuscinosis. Neuroophthalmology. 2007;31 [abstract issue] Dagi LR, Chrousos GA, Cogan DC. Spasm of the near reflex associated with organic disease. Am J Ophthalmol. 1987;103: David G, Abbas N, Stevanin G, et al. Cloning of the SCA7 gene reveals a highly unstable CAG repeat expansion. Nat Genet. 1997;17: Davidson JL, Rosenbaum AL, McCall LC. Strabismus surgery in patients with myasthenia. J Pediatr Ophthalmol Strabismus. 1993;30: de Sa LC, Good WV, Hoyt CS. Results of initial surgery for comitant strabismus in 25 neurologically impaired children. Binocul Vis Q. 1992;7: de Saint Sardos A, Vincent A, Aroichane M, et al. Ocular neuromyotonia in a 15-year-old girl after radiation therapy. J AAPOS. 2008;6: de Souza-Dias CR, Goldschmit M. Further considerations about the ophthalmic features of the Möbius sequence, with data of 28 cases. Arq Bras Oftalmol. 2007;70: Deleu D, Buisseret T, Buisseret T. Vertical one-and-a-half syndrome. Supranuclear downgaze paralysis with monocular elevation palsy. Arch Neurol. 1989;46: DeMarco P. Eyelid myoclonia with absences (EMA) in two monovular twins. Clin Electroencephalogr. 1989;20: Demer JL. Clarity of words and thoughts about strabismus. Am J Ophthalmol. 2001;132: Demer JL. Anatomical diagnosis. Br J Ophthalmol. 2007; Demer JL, Clark RA, Engle EC. Magnetic resonance imaging evidence for widespread orbital dysinnervation in congenital fibrosis of extraocular muscles type 1. Invest Ophthalmol Vis Sci. 2005;46: Diamond GR, Whitaker L. Ocular motility in craniofacial reconstruction. Plast Reconstr Surg. 1984;73: Dickey CF. Clinical presentation of congenital fibrosis and double elevator palsy. Am Orthopt J. 1995;43: Dickey CF, Scott WE, Cline RA. Oblique muscle palsies fixating with the paretic eye. Surv Ophthalmol. 1988;33: Diesenhouse MC, Palay DA, Newman NJ, et al. Acquired heterochromia with Homer syndrome in two adults. Ophthalmology. 1992;99: Digre KB, Smoker WR, Johnston P, et al. Selective MR imaging approach for evaluation of patients with Homer s syndrome. AJNR Am J Neuroradiol. 1992;13: Diir LY, Donofrio PD, Patton JF, et al. A false-positive edrophonium test in a patient with a brainstem glioma. Neurology. 1989;39: DiMauro S, Hirano M. Mitochondrial encephalomyopathies: an update. Neuromuscul Disord. 2005;15: Dinc H, Alioglu Z, Erdöl H, et al. Agenesis of the internal carotid artery associated with aortic arch anomaly in a patient with congenital Horner syndrome. Am J Neuroradiol. 2002;23: Dittrich J, Havlová M, Nev&imalova S. Paroxysmal hemiparesis in childhood. Dev Med Child Neurol. 1979;21: Doherty EJ, Macy ME, Wang SM, et al. CFEOM3: a new extraocular congenital fibrosis syndrome that maps to 16q24.2 q24.3. Invest Ophthalmol Vis Sci. 1999;40: Donahue SP, Lavin PJ, Digre K. False-negative hydroxyamphetamine (Paredrine) test in acute Horner s syndrome. Am J Ophthalmol. 1996;122: Donahue SP, Lavin PJ, Hamed LM. Tonic ocular tilt reaction simulating a superior oblique palsy: diagnostic confusion with the 3-step test. Arch Ophthalmol. 1999;117: Donhowe SP. Bilateral internuclear ophthalmoplegia from doxipen overdose. Neurology. 1984;34: Dooley JM, Stewart WA, Hayden JD, et al. Brainstem calcification in Möbius syndrome. Pediatr Neurol. 2004;30: Drachman DB. Present and future treatment of myasthenia gravis. N Engl J Med. 1987;316: Drachman DB. Myasthenia gravis. N Engl J Med. 1994;330: Dralands L, Adaenssens L. Persistent isolated paralysis of accommodation in young people. Bull Soc Belge Ophtalmol. 1978;182: Dretakis EK, Kondoyannis PN. Congenital scoliosis associated with encephalopathy in five children of two families. J Bone Joint Surg Am. 1974;56: Drummond S, Weir C, Buchan D, et al. Cyclical esotropia following surgery for accommodative esotropia. Br J Ophthalmol. 2004;88: Dumars S, Andrews C, Chan WM, et al. Magnetic resonance imaging of the endophenotype of a novel familial Möbius-like syndrome. J AAPOS. 2008;12: Duncan MB, Jabbari B, Rosenberg ML. Gaze evoked visual seizures in nonketotic hyperglycemia. Epilepsia. 1991;32: Dunlap EA. Vertical displacement of the horizontal rectus muscles. In: Symposium on Strabismus: Transactions of the New Orleans Academy of Ophthalmology. St. Louis: Mosby; 1971: Durairaj VD, Bartley GB, Garrity JA. Clinical features and treatment of Graves ophthalmopathy in pediatric patients. Ophthal Plast Reconstr Surg. 2006;22: Durr A, Stevanin G, Cancel G, et al. Spinocerebellar ataxia 3 and Machado Joseph disease: clinical, molecular, and neuropathological features. Ann Neurol. 1996;39: Dutton GN, Paul R. Adie syndrome in a child: a case report. J Pediatr Ophthalmol Strabismus. 1992;29: Ebner R, Lopez L, Ochoa S, et al. Vertical ocular motor apraxia. Neurology. 1990;40: Eda I, Takashim S, Kitahara T, et al. Computed tomography in congenital ocular motor apraxia. Neuroradiology. 1984;26: Eggenberger E, Golnik K, Lee A, et al. Prognosis of ischemic internuclear ophthalmoplegia. Ophthalmology. 2002;109: Ellis FD, Hoyt CS, Ellis FJ, et al. Extraocular muscle responses to orbital cooling (ice test) for ocular myasthenia gravis diagnosis. J AAPOS. 2000;2: Elrod RD, Weinberg DA. Ocular myasthenia. Ophthalmol Clin North Am. 2004;17: Elston JS, Granje FC, Lees AJ. The relationship between eye winking tics, frequent eye blinking, and blepharospasm. J Neurol Neurosurg Psychiatry. 1989;52: Emir S, Kutluk MT, Gogus S, et al. Paraneoplastic cerebellar degeneration and Horner syndrome: association of two uncommon findings in a child with Hodgkin disease. J Pediatr Hematol Oncol. 2000;22:
63 References 238. Enevoldson TP, Sanders MD, Harding AE. Autosomal dominant cerebellar ataxia with pigmentary macular dystrophy (a clinical and genetic study of eight families). Brain. 1994;117: Engel AG. Congenital myasthenic syndromes. Handb Clin Neurol. 2008;91: Engel AG. Further observations in congenital myasthenic syndromes. Ann N Y Acad Sci. 2008;1132: Engel E. New concepts in CFEOM and congenital cranial dysinnervation syndromes. In: Proceedings of the North American Neuro-Ophthalmology Society Meeting, Lake Tahoe, NV; February 26, 2009: Engel AG, Ohno K, Shen XM, et al. Congenital myasthenic syndromes: multiple molecular targets at the neuromuscular junction. Ann N Y Acad Sci. 2003;998: Engel AG, Ohno K, Sine SM. Congenital myasthenic syndromes: recent advances. Arch Neurol. 1999;56: Engel AG, Ohno K, Sine SM. Congenital myasthenic syndromes: progress over the past decade. Muscle Nerve. 2003;27: Engel AG, Ohno K, Sine SM. Sleuthing molecular targets for neurological diseases at the neuromuscular junction. Nat Rev Neurosci. 2003;4: Engel AG, Walls TJ, Nagel A, et al. Newly recognized congenital myasthenic syndromes. I: Congenital paucity of synaptic vesicles and reduced quantal release. II: High-conductance fast-channel syndrome. III: Abnormal acetylcholine receptor (AChR) interaction with acetylcholine. IV: AChR deficiency and short channel-open time. Prog Brain Res. 1990;84: Engle EC. Oculomotility disorders arising from disruptions in brainstem motor neuron development. Arch Neurol. 2007;64: Engle EC, Castro AE, Macy ME, et al. A gene for isolated congenital ptosis maps to a 3-cM region within 1p32 p34.1. Am J Hum Genet. 1997;60: Engle EC, Goumernov B, McKeown CA, et al. Oculomotor nerve and muscle abnormalities in congenital fibrosis of the extraocular muscles. Ann Neurol. 1997;41: Engle EC, Kunkel LM, Specht LA, et al. Mapping a gene for congenital fibrosis of the extraocular muscles to the centromeric region of chromosome 12. Nat Genet. 1994;7: Engle EC, Marondel I, Houtman WA, et al. Congenital fibrosis of the extraocular muscles (autosomal dominant congenital external ophthalmoplegia): genetic homogeneity, linkage refinement, and physical mapping on chromosome 12. Am J Hum Genet. 1995;57: Engle EC, McIntosh N, Yamada K, et al. CFEOM1, the classic familial form of congenital fibrosis of the extraocular muscle, is generally heterogeneous but does not result from mutations in ARIX. BMC Genet. 2002;3: Erikson A, Wahlberg I. Gaucher disease Norrbottnian type. Ocular abnormalities. Acta Ophthalmol. 1985;63: Ertas M, Arac N, Kumral K, et al. Ice test as a simple diagnostic aid for myasthenia gravis. Acta Neurol Scand. 1994;89: Eustace P, Beigi B, Bowell R, et al. Congenital ocular motor apraxia: an inability to unlock the vestibulo-ocular reflex. Neuroophthalmology. 1994;14: Evoli A, Baocchi AP, Bartoccioni E, et al. Juvenile myasthenia gravis with prepubertal onset. Neuromuscul Disord. 1998;8: Evoli A, Tonali P, Bartoccioni E, et al. Ocular myasthenia: diagnostic and therapeutic problems. Acta Neurol Scand. 1988;77: Evoli A, Tonali PA, Padua L, et al. Clinical correlates with anti- MuSK antibodies in generalized seronegative myasthenia gravis. Brain. 2003;126: Faucher C, De Guise D. Spasm of the near reflex triggered by disruption of normal binocular vision. Optom Vis Sci. 2004; 81: Felice KJ, DiMario FJ, Conway SR. Postinfections myasthenia gravis: report of two children. J Child Neurol. 2005;20: Fells P, Jampel RS. Supranuclear factors in monocular elevation palsy. Trans Ophthalmol Soc U K. 1970;90: Fells P, Waddell E, Rodrigues M. Progressive, exaggerated A-pattern strabismus with presumed fibrosis of extraocular muscles. In: Reinecke RD, ed. Strabismus II: Proceedings of the Fourth Meeting at the International Strabismological Association; October 25 29, Asilomar, CA: Grune & Stratton, 1984: Felz MW, Smith CD, Swift TR. A six-year-old girl with tick paralysis. N Engl J Med. 2000;342: Fenichel GM. Clinical syndromes of myasthenia in infancy and childhood. A review. Arch Neurol. 1978;35: Ferland RJ, Eyaid W, Collura RV, et al. Abnormal cerebellar development and axonal decussation due to mutations in AHI1 in Joubert syndrome. Nat Genet. 2004;36: Ferrar JA. General Fibrosis Syndrome. Second Congress of the International Strabismological Association. Paris: Diffus Générale de Librairi; 1976: Ferrarini M, Squintani G, Cavallaro T, et al. A novel mutation of aprataxin associated with ataxia ocular apraxia type I: phenotypical and genotypical characterization. J Neurol Sci. 2007;260: a. Ferraris S, Clark S, Garelli E, et al. Progressive external ophthalmoplegia and vision and hearing loss in a patient with mutations in POLG2 and OPA1. Arch Neurol 2008;65: Fielder AR, Gresty MA, Dodd KL, et al. Congenital ocular motor apraxia. Trans Ophthalmol Soc U K. 1986;105: Fisher PG, Wechsler DS, Singer HS. Anti-Hu antibody in a neuroblastoma-associated paraneoplastic syndrome. Pediatr Neurol. 1994;10: FitzGerald PM, Jankovic J, Glaze DG, et al. Extrapyramidal involvement in Rett s syndrome. Neurology. 1990;40: Flueler U, Taylor D, Hing S, et al. Hemifacial spasm in infancy. Arch Ophthalmol. 1990;108: Flynn JT. Strabismus: A Neurodevelopmental Approach. New York: Springer; Font RL, Blanco G, Soparkar CH, et al. Giant cell reparative granuloma of the orbit associated with cherubism. Ophthalmology. 2003;110: Ford CS, Schwartze GM, Weaver RG, et al. Monocular elevation paresis caused by an ipsilateral lesion. Neurology. 1984;34: Frank JW. Problems with accommodation. Am Orthopt J. 2000;49: Frankel M, Cummings JL. Neuro-ophthalmic abnormalities in Tourette s syndrome: functional and anatomic implications. Neurology. 1984;34: Friedman DI, Fortanasce VN, Sadun AA. Tonic pupils as a result of botulism. Am J Ophthalmol. 1990;109: Friedman A, Jamrozik Z, Bojakowski J. Familial hemifacial spasm. Mov Disord. 1989;4: Friendly DS, Manson RA, Albert DG. Cyclic strabismus a case study. Doc Ophthalmol. 1973;34: Fries PD, Katowitz JA. Congenital craniofacial anomalies of ophthalmic importance. Surv Ophthalmol. 1990;35: Frohman LP, Kupersmith MJ. Reversible vertical ocular deviations associated with raised intracranial pressure. J Clin Neuroophthalmol. 1985;5: Gadoth N, Dickerman Z, Lerman M, et al. Cyclic esotropia with minimal brain dysfunction. J Pediatr Ophthalmol Strabismus. 1981;18: Galetta SL, Gray LC, Raps EC, et al. Unilateral ptosis and contralateral lid retraction from a thalamic-midbrain infarction. Magnetic resonance imaging correlation. J Clin Neuroophtalmol. 1993;4: Galletta SL, Liu GT, Raps EC, et al. Cyclodeviation in skew deviation. Am J Ophthalmol. 1994;118: Gamio S, Garcia-Erro M, Vaccarezza MM, et al. Myasthenia gravis in childhood. Binocul Vis Strabismus Q. 2004;19:
64 372 7 Complex Ocular Motor Disorders in Children 286. Gamio S, Melek N. When the patient says no. Management of exotropia with hemianopic visual field defects. Binocul Vis Strabismus Q. 2003;18: Gaynard B, Lafitte C, Gelot A, et al. Plus minus syndrome. J Neurol Neurosurg Psychiatry. 1992;55: Geh VS, Bradbury JA. Ocular myasthenia presenting in an 11-month-old boy. Eye. 1998;12: George ND, Gonzalez G, Hoyt CS. Does Horner s syndrome in infancy require investigation? Br J Ophthalmol. 1998;82: Gibbs J, Appleton RE, Martin J, Findlay G. Congenital Horner syndrome associated with non cervical neuroblastoma. Dev Med Child Neurol. 1992;34: Gillies WE, Harris AJ, Brooks AM. Congenital fibrosis of the vertically acting extraocular muscles. Ophthalmology. 1995;102: Gleeson JG, Keeler LC, Parisi MA, et al. Molar tooth sign of the midbrain hindbrain junction: occurrence in multiple distinct syndromes. Am J Med Genet. 2004;125A: Godel V, Nemet P, Lazar M. Congenital ocular motor apraxia: familial occurrence. Ophthalmologica. 1979;179: Goldberg MF, Payne JW, Brunt PW. Ophthalmologic studies of familial dysautonomia, the Riley Day syndrome. Am J Ophthalmol. 1968;80: a. Goldberg DS, Ludwig IH. Congenital central hypoventilation syndrome: ocular findings in 37 children. J Pediatr Ophthalmol Strabismus. 1996;33: Goldblum TA, Effron LA. Upbeat nystagmus associated with tonic downward deviation in healthy neonates. J Pediatr Ophthalmol Strabismus. 1994;31: Goldschmit M. Further considerations about the ophthalmic features of the Möbius sequence, with data of 28 cases. Arq Bras Oftalmol. 2007;70: Goldsmith MO. Tonic pupil following varicella. Am J Ophthalmol. 1968;66: Goldstein SM, Liu GT, Edmond JC, et al. Orbital neural glial hamartoma associated with a congenital tonic pupil. J AAPOS. 2002;6: Goldstein JH, Schneekloth BB. Spasm of the near reflex: a spectrum of anomalies. Surv Ophthalmol. 1996;40: Golnik KC, Pena R, Lee AG, et al. An ice test for the diagnosis of myasthenia gravis. Ophthalmology. 1999;106: Golzarian J, Balériaux D, Bank WO, et al. Pineal cyst: normal or pathological? Neuroradiology. 1993;35: Gondipalli P, Tobias JD. Anesthetic implications of Möbius syndrome. J Clin Anesth. 2006;18: Good WV, Crain LS, Quint RD, Koch TK. Overlooking: a sign of bilateral central scotomata in children. Dev Med Child Neurol. 1992;34: Good WV, Hou C, Carden SM. Transient, idiopathic nystagmus in infants. Dev Med Child Neurol. 2003;45: Gordon N. Ophthalmoplegia in childhood. Dev Med Child Neurol. 1994;36: Gote H, Gregersen E, Rindziunski E. Exotropia and panoramic vision compensating for an occult congenital homonymous hemianopia: a case report. Binocul Vis Eye Muscle Surg Q. 1993;8: Gothe R, Kunze K, Hoogstraal H. The mechanisms of pathogenicity in the tick paralyses. J Med Entomol. 1979;16: Gouw LG, Digre KB, Harris CP, et al. Autosomal dominant cerebellar ataxia with retinal degeneration. A clinical and ocular histopathologic study. Neurology. 1994;44: Gouw LG, Kaplan CD, Haines JH, et al. Retinal degeneration characterizes a spinocerebellar ataxia mapping to chromosome 3p. Nat Genet. 1997;17: Granet DB, Gomi CF, Ventura R, et al. The relationship between convergence insufficiency and ADHD. Strabismus. 2005;13: Green JP, Newman NJ, Winterkorn JS. Paralysis of downgaze in two patients with clinical-radiologic correlation. Arch Ophthalmol. 1993;111: Gregorian AP, Phillips P, Brodsky MC. Neurodevelopmental outcome in congenital ocular motor apraxia. In: Presented as a Poster at the North American Neuro-Ophthalmology Society, North Lake Tahoe, Nevada; February 21 26, Griepentrog GJ, Aagaard-Kienitz B, Kushner BJ, et al. Unilateral eyelid swelling and ptosis caused by dural arteriovenous fistula in an infant. Arch Ophthalmol. 2006;124: Griffiths PG, Andrews RM. Chronic progressive external ophthalmoplegia. Br Orthop J. 2000;57: Gropman AL. The neurological presentation of childhood and adult mitochondrial disease: established syndromes and phenotypic variations. Mitochondrion. 2004;4: Gross-Tsur V, Har-Even Y, Gutman I, et al. Oculomotor apraxia: the presenting sign of Gaucher disease. Pediatr Neurol. 1989;5: Gruters A. Ocular manifestations in children and adolescents with thyrotoxicosis. Exp Clin Endocrinol Diabetes. 1999;107: S172 S Guo S, Brush J, Teraoka H, et al. Development of noradrenergic neurons in the zebra fish hindbrain requires BMP, FGFs, and the homeodomain protein soulless/phox2a. Neuron. 1999;24: Gupta M, Dinakaran S, Chan TK. Congenital Horner syndrome and hemiplegia secondary to carotid dissection. J Pediatr Ophthalmol Strabismus. 2005;42: Gurer YK, Kukner S, Kunak B, et al. Congenital ocular motor apraxia in two siblings. Pediatr Neurol. 1995;13: Gutowski NJ, Bosley TM, Engle EC. 110th ENMCC International Workshop. The Congenital Cranial Dysinnervation Disorders (CCDDs). Naarden, The Netherlands, Oct , Neuromuscul Disord. 2003;13: Guy J, Day AL, Mickle JP, et al. Contralateral trochlear nerve paresis and ipsilateral Horner s syndrome. Am J Ophthalmol. 1989;107: Guyton DL, Weingarten PE. Sensory torsion as the cause of primary oblique muscle overaction/underaction and A- and V-pattern strabismus. Binocul Vis Eye Muscle Surg Q. 1994;9: Hageman G, Ippel PF, te-nijenhuis FC. Autosomal dominant congenital Horner s syndrome in a Dutch family. J Neurol Neurosurg Psychiatry. 1992;55: Haildi BA. Surgical management of convergence insufficiency. Am Orthopt J. 1978;28: Hall CJ. Eye movement disorders in hydrocephalus and increased intracranial pressure. Am Orthopt J. 2005;55: Hallermann W. SchweiBsekretionsstorungen beim Adie-syndrom. Eine neuropathia multiplex der peripheren autonomen nerven? Aktuel Neurol. 1990;17: Hallett M, Cogan DG. Episodic unilateral mydriasis in otherwise normal patients. Arch Ophthalmol. 1970;84: Halstead SK, Humphreys PD, Goodfellow JA, et al. Complement inhibition abrogates nerve terminal injury in Miller Fisher syndrome. Ann Neurol. 2005;58: Hamanishi C, Tanaka S, Kasahara Y, et al. Progressive scoliosis associated with lateral gaze palsy. Spine. 1993;18: Hamed LM. Bilateral Brown s syndrome in three siblings. J Pediatr Ophthalmol Strabismus. 1991;28: Hamed LM. Alternating skew on lateral gaze simulating bilateral superior oblique overaction. Binocul Vis Q. 1992;7: Hamed LM. Cyclic, periodic ophthalmic disorders. In: Margo C, Hamed LM, Mames R, eds. Diagnostic Problems in Clinical Ophthalmology. Philadelphia: W.B. Saunders; 1993: Hamed LM. Superior oblique overaction: some nosologic considerations. Am Orthopt J. 1993;43:82 86.
65 Referenes 335. Hamed LM, Chala P, Fanous M, Guy J. Strabismus surgery in selected patients with stable ocular myasthenia gravis. Binocul Vis Q. 1994;9: Hamed LM, Challa P, Fanous MM, et al. Strabismus surgery in selected patients with stable myasthenia gravis. Binocul Vis Eye Muscle Surg. 1994;9: Hamed LM, Dennehy PJ, Lingua RW. Synergistic divergence and jaw-winking phenomenon. J Pediatr Ophthalmol Strabismus. 1990;27: Hamed LM, Fang EN. Inferior rectus muscle contracture resulting from perinatal orbital trauma. J Pediatr Ophthalmol Strabismus. 1992;29: Hamed LM, Fang E, Fanous M, et al. The prevalence of neurological dysfunction in children with strabismus who have superior oblique overaction. Ophthalmology. 1993;100: Hamed LM, Lessner A. Fixation duress in the pathogenesis of upper lid retraction in thyroid orbitopathy. Ophthalmology. 1994;101: Hamed LM, Maria BL, Quisling RG, Mickle JP. Alternating skew on lateral gaze: neuroanatomic pathway and relationship to superior oblique overaction. Ophthalmology. 1993;100: Hamed LM, Maria BL, Tusa R, et al. Periodic alternating gaze deviation in Joubert syndrome. In: Presented as a Poster at the American Academy of Ophthalmology; Hamed LM, Schatz NJ, Galetta SL. Brain stem ocular motility defects and AIDS. Am J Ophthalmol. 1988;106: Hamed LM, Silbiger J. Periodic alternating esotropia. J Pediatr Ophthalmol Strabismus. 1992;29: Hamilton SR, Chatrian GE, Mills RP, Kalina RE, Bird TD. Cone dysfunction in a subgroup of patients with autosomal dominant cerebellar ataxia. Arch Ophthalmol. 1990;108: Hammans SR, Sweeney MG, Brockington M, et al. The mitochondrial DNA transfer RNA-LysA(G(8344) mutation and the syndrome of myoclonic epilepsy with ragged-red fibers (MERRF). Relationship of clinical phenotype to proportion of mutant mitochondrial DNA. Brain. 1991;116: Harding AE. Classification of the hereditary ataxias and paraplegias. Lancet. 1983;1: Harding AE. Vitamin E and the nervous system. Crit Rev Neurobiol. 1987;3: Harley RD. Complete tendon transplantation for ocular motor paralysis. Ann Ophthalmol. 1971;3: Harley RD, Rodriguez MM, Crawford JS. Congenital fibrosis of the extraocular muscles. Trans Am Ophthalmol Soc. 1978;76: Harper CM. Congenital myasthenic syndromes. Semin Neurol. 2004;24: Harrad RA, Shuttleworth GN. Superior-rectus levator synkinesis: a previously unrecognized cause of failure of ptosis surgery. Ophthalmology. 2000;107: Harriman DGF, Garland H. The pathology of Adie s syndrome. Brain. 1968;91: Harris CM, Shawkat F, Russell-Eggitt I, et al. Intermittent horizontal saccadic failure ( ocular motor apraxia ) in children. Br J Ophthalmol. 1996;80: Hart ZH, Sahashi K, Lambert EH, et al. A congenital, familial, myasthenic syndrome caused by a presynaptic defect of transmitter resynthesis or mobilization. Neurology. 1979;29: Harvey AS, Jayakar P, Duchowny M, et al. Hemifacial seizures and cerebellar ganglioglioma: an epilepsy syndrome of infancy with seizures of cerebellar origin. Ann Neurol. 1996;40: Havlová M, Otradovec J, Dittrich J. Hypotonic syndrome accompanied by paroxysmal paralyses and skew deviation. Actat Univ Carol Med Monogr. 1976;75: Hawes MJ. Cherubism and its orbital manifestations. Ophthal Plast Reconstr Surg. 1989;5: Heger T, Kolling GH, Dithmar S. Atypical tonic pupil as a complication of chickenpox infection. Ophthalmologe. 2003;100: Hermann JS. Surgical therapy for convergence insufficiency. J Pediatr Ophthalmol Strabismus. 1981;18: Hermann DN, Carney PR, Wald JJ. Juvenile myasthenia gravis: treatment with immune globulin and thymectomy. Pediatr Neurol. 1998;18: Hertle RW, Katowitz JA, Young TL, et al. Congenital unilateral fibrosis, blepharoptosis, and enophthalmos syndrome. Ophthalmology. 1992;99: Herzau V, Bleher I, Joos-Kratsch E. Infantile exotropia with homonymous hemianopia: a rare contraindication for strabismus surgery. Graefes Arch Clin Exp Ophthalmol. 1988;226: Hiles DA, Walla PH, McFarlane F. Current concepts in the management of strabismus in children with cerebral palsy. Ann Ophthalmol. 1975;7: Hirano M, Nishigaki Y, Marti R. Mitochondrial neurogastrointestinal encephalomyopathy (MNGIE): a disease of two genomes. Neurologist. 2004;10: Hirano M, Pavlakis SG. Mitochondrial myopathy, encephalopathy, lactic acidosis, and strokelike episodes (MELAS): current concepts. J Child Neurol. 1994;9: Ho SL, Cheng PW, Wong WC, et al. A case-controlled MRI/MRA study of neurovascular contact in hemifacial spasm. Neurology. 1999;53(21): Hoch W, McConville J, Helms S, et al. Auto-antibodies to the receptor tyrosine kinase MuSK in patients with myasthenia gravis without acetylcholine receptor antibodies. Nat Med. 2001;7: Hodgkins HCM, PR KA, et al. Familial congenital saccadic initiation and isolated cerebellar vermis hypoplasia. Dev Med Child Neurol. 1998;40: Hoefnagel D, Joseph JB. Oculosympathetic paralysis in otitis media. N Engl J Med. 1961;265: Holman RE, Merrit JC. Infantile esotropia: results in the neurologic impaired and normal child at NCMH (six years). J Pediatr Ophthalmol Strabismus. 1986;23: Holmberg M, Johansson J, Forggren L, et al. Localization of autosomal dominant cerebellar ataxia associated with retinal degeneration and anticipation to chromosome 3p12 p21.1. Hum Mol Genet. 1995;10: Horikawa H, Juo K, Mano Y, et al. A case of neurovisceral storage disease with sea-blue histiocyte and severe horizontal supranuclear ophthalmoplegia. Rinsho Shinkeigaku. 1990;30: Horwood A. Too much or too little: neonatal ocular misalignment frequency can predict lateral abnormality. Br J Ophthalmol. 2003;87: Horwood AM, Riddell PM. Can misalignments in typical infants be used as a model for infantile esotropia? Invest Ophthalmol Vis Sci. 2004;45: Houtman WA, van Weerden TW, Robinson PH, et al. Hereditary congenital external ophthalmoplegia. Ophthalmologica. 1986;193: Howard RS. A case of convergence evoked eyelid nystagmus. J Clin Neuroophthalmol. 1986;6: Hoyt CS. Acquired double elevator palsy and polycythemia vera. J Pediatr Ophthalmol Strabismus. 1978;15: Hoyt WF, Daroff RB. Supranuclear disorders of ocular control systems in man: clinical, anatomical, and physiological correlations In: Bach-Rita P, Collins CC, Hyde JE, eds. The Control of Eye Movements. Orlando, FL: Academic; 1971: Hoyt CS, Good WV. Ocular motor adaptations to congenital hemianopia. Binocul Vis Eye Muscle Surg Q. 1993;8: Hoyt CS, Mousel DK, Weber AA. Transient supranuclear disturbances of gaze in healthy neonates. Am J Ophthalmol. 1980;89: Hriso E, Masdeu JC, Miller A. Monocular elevation weakness and ptosis: an oculomotor fascicular syndrome? J Clin Neuroophthalmol. 1991;11: Hughes JL, O Conner PS, Larsen PD, et al. Congenital vertical ocular motor apraxia. J Clin Neuroophthalmol. 1985;5:
66 374 7 Complex Ocular Motor Disorders in Children 384. Hunter DG, Kelly JB, Ellis FJ. Long-term outcome of uncomplicated infantile exotropia. J AAPOS. 2001;5: Hutcheson KA, Lambert SR. Cyclic esotropia after a traumatic sixth nerve palsy in a child. J AAPOS. 1998;2: Hwang J-M, Choung HK, Ko HS, et al. Ophthalmoplegia diagnosis. Ophthalmology. 2009;116: Hwang JM, Park SH. A case of Marcus Gunn jaw winking and pseudo inferior oblique muscle overaction. Am J Ophthalmol. 2001;131: Ibrahim M, Branson HM, Buncic JR, et al. A case of Horner syndrome with intermittent mydriasis in a patient with hypoplasia of the internal carotid artery. Am J Neuroradiol. 2006;27: Ing EB, Ing SY, Ing T, et al. The complication rate of edrophonium testing for suspected myasthenia gravis. Can J Ophthalmol. 2000;35: Ito S, Hattori T, Kastayama K. Prominent unilateral convergence palsy in a patient with a tiny dorsal midbrain infarction. Eur Neurol. 2005;54: Jacobson DM, Vierkant RA. Comparison of cholinergic supersensitivity of the iris sphincter in patients with oculomotor nerve palsies. Am J Ophthalmol. 1990;40: Jampel RS, Okazaki H, Bernstein H. Ophthalmoplegia and retinal degeneration associated with spinocerebellar ataxia. Arch Ophthalmol. 1961;66: Jan JE, Farrell K, Wong PK, et al. Eye and head movements of visually impaired children. Dev Med Child Neurol. 1986;28: Jan JE, Freeman RD, McCormick AQ, et al. Eye pressing by visually impaired children. Dev Med Child Neurol. 1983;25: Jan JE, Freeman R, Scott E. Visual Impairment in Children and Adolescents. New York: Grune & Stratton; Jan JE, Kearney S, Groenveld M, et al. Speech, cognition, and imaging studies in congenital ocular motor apraxia. Dev Med Child Neurol. 1998;40: Janetta PJ, Abbasy M, Maroon JC, et al. Etiology and definitive microsurgical management of hemifacial spasm: operative techniques and results in 47 patients. J Neurosurg. 1977;47: Jankovic J, Stone L. Dystonic tics in patients with Tourette s syndrome. Mov Disord. 1991;6: Jeffrey AR, Ellis FJ, Repka MX, et al. Pediatric Horner syndrome. J AAPOS. 1998;2: Jen JC, Chan WM, Bosley TM, et al. Mutations in a human ROBO gene disrupt hindbrain axon pathway crossing and morphogenesis. Science. 2004;304: Jenkins RH. Characteristics and diagnosis of convergence insufficiency. Am Orthopt J. 1999;49: Jho HD, Jannetta PJ. Hemifacial spasm in young people treated with microvascular decompression of the facial nerve. Neurosurgery. 1987;20: Johansson M, Wentz E, Fernell E, et al. Autistic spectrum disorders in Möbius sequence: a comprehensive study of 25 individuals. Pediatr Neurol. 2001;24: Jones WA. Familial multilocular cystic disease of the jaw. Am J Cancer. 1933;17: Joubert M, Eisenring J, Robb JP, et al. Familial agenesis of the cerebellar vermis. Neurology. 1989;19: Jung H-Y, Chung S-J, Hwant J-M. Tic disorders in children with frequent eye blinking. J AAPOS. 2004;8: Kaminski JH, Kusner LL, Block CH. Expression of acetylcholine receptor isoforms at extraocular muscle endplates. Invest Ophthalmol Vis Sci. 1996;37: Kaminski JH, Mass E, Spiegel P, et al. Why are the eye muscles frequently involved in myasthenia gravis? Neurology. 1990;40: Kaminski HJ, Richmonds CR, Kusner LL, et al. Differential susceptibility of the ocular motor system to disease. Ann N Y Acad Sci. 2002;956: Kaminski HJ, Richmonds C, Lin F, et al. Complement regulators in extraocular muscle and experimental autoimmune myasthenia gravis. Exp Neurol. 2004;189: Kaminski HJ, Ruff RL. Ocular muscle involvement by myasthenia gravis. Ann Neurol. 1997;41: Kansu T, Subutay N. Lid retraction in myasthenia gravis. J Clin Neuroophthalmol. 1987;7: Kao Y, Lan M, Chou M, Chen W. Intracranial fatigable ptosis. J Neuroophthalmol. 1999;19: Kaplan I, Blakely BT, Pavlath GK, et al. Steroids induce acetylcholine receptors on cultured human muscle: implications for myasthenia gravis. Proc Natl Acad Sci USA. 1990;87: Kaplan PW, Lesser RP. Vertical and horizontal epileptic gaze deviation and nystagmus. Neurology. 1989;39: Kardon R, Lee A. Diagnosis of Horner syndrome using the topical effects of apraclonidine on the eyelids, conjunctivae and pupils. In: NANOS, Tucson, AZ; February 25-March 2, 2006: Katayama M, Tamas LB. Saccadic eye-movements of children with cerebral palsy. Dev Med Child Neurol. 1987;29: Katz B, Rosenberg JH. Botulinum therapy for apraxia of eyelid opening. Am J Ophthalmol. 1987;103: Keane JR. Ocular skew deviation. Analysis of 100 cases. Arch Neurol. 1975;32: Keane JR. Pretectal pseudobobbing. Five patients with V -pattern convergence nystagmus. Arch Neurol. 1985;42: Keane JR. The pretectal syndrome: 206 patients. Neurology. 1990;40: Keane JR. Internuclear ophthalmoplegia. Arch Neurol. 2005;62: Keane JR, Finstead BA. Upward gaze paralysis as the initial sign of Fisher s syndrome. Arch Neurol. 1982;39: Kearns TP, Sayre GP. Retinitis pigmentosa, external ophthalmoplegia, and complete heart block. Arch Ophthalmol. 1958;60: Kennard C, Barger G, Hoyt WF. The association of periodic alternating nystagmus with periodic alternating gaze. J Clin Neuroophthalmol. 1981;1: Kernan JC, Devinsky O, Luciano DJ, et al. Lateralizing significance of head and eye deviation in secondary generalized tonicclonic seizures. Neurology. 1993;43: Kesler A, Stolovitch C, Hoffman C, et al. Acute ophthalmoplegia and nystagmus in infants fed a thiamine-deficient formula: an epidemic of Wernicke encephalopathy. J Neuroophthalmol. 2005;25: Khan AO. A novel form of aberrant innervation in congenital cranial disinnervations disorder. J AAPOS. 2009;13: Khan AO, Al-Hommaidi A, Al-Turkmani S. Familial ptotic lid elevation during ipsilateral abduction. J AAPOS. 2004;8: Khan AO, Khalil DS, Al-Tassan NA. Congenital fibrosis of the extraocular muscles type 1 (CFEOM1) on the Arabian Peninsula. Ophthalmic Genet. 2008;29: Khan S, Nischal KK, Dean F, et al. Ophthalmic morbidity in the syndromic craniosynostoses: a review of 141 cases. Br J Ophthalmol. 2003;87: Khan AO, Oystreck DT, Seidahmed MZ, et al. Ophthalmic features of Joubert syndrome. Ophthalmology. 2008;115: Khan N, Schnizel A, Shuknecht B, et al. Moyamoya angiopathy with dolicoectatic internal carotid arteries. Eur Neurol. 2004;51: Kim WJ, Chang BL. Unilateral congenital ocular motor apraxia: a case report. Korean J Ophthalmol. 1992;6: Kim JH, Hwang JM. Congenital monocular elevation deficiency. Ophthalmology. 2009;116: Kim JH, Hwang JM, Hwang YS, et al. Childhood ocular myasthenia gravis. Ophthalmology. 2003;110: Kim JS, Park S-H, Lee K-W. Spasmus nutans and congenital ocular motor apraxia with cerebellar vermian hypoplasia. Arch Neurol. 2003;60:
67 References 438. King MD, Dudgeon J, Stephenson JBP. Joubert s syndrome with retinal dysplasia; neonatal tachypnea as the clue to the genetic brain eye malformation. Arch Dis Child. 1984;59: Kirkali P, Topaloglu R, Kansu T, et al. Third nerve palsy and internuclear ophthalmoplegia in periarteritis nodosa. J Pediatr Ophthalmol Strabismus. 1991;28: Kline KB, McCleur SM, Boniskowski FP. Oculosympathetic spasm with cervical cord injury. Arch Neurol. 1984;41: Knapp P. The surgical treatment of double elevator paralysis. Trans Am Ophthalmol Soc. 1969;67: Knapp C, Sachdev A, Gottlob I. Spasm of the near reflex associated with head injury. Strabismus. 2002;10: Kobata H, Kondo A, Kinuta Y, et al. Hemifacial spasm in childhood and adolescence. Neurosurgery. 1995;36: Kodsi S. Marcus Gunn jaw winking with trigemino-abducens synkinesis. J AAPOS. 2000;4: Koeppen AH. The pathogenenis of spinocerebellar ataxia. Cerebellum. 2005;4: Koga M, Takahashi M, Masuda M, et al. Campylobacter gene polymorphism as a determinant of clinical features of Guillain Barré syndrome. Neurology. 2005;65: Kolling G, Rohde S, Kress B. Congenital Brown s syndrome is caused by missing fourth cranial nerve in some cases. In: Presented at the 32nd Meeting of the European Strabismological Association, Munich, Germany; September 7 10, Kondo A, Saito Y, Floricel F, et al. Congenital ocular motor apraxia: clinical and neuroradiological findings, and long-term intellectual prognosis. Brain Dev. 2007;29: Kothari M, Mody K, Walinjkar J, et al. Paralysis of the near-vision triad in a child. J AAPOS. 2009;11: Kowal L. Ophthalmic manifestations of head injury. Aust N Z J Ophthalmol. 1992;20: Krohel GB, Griffin JF. Cortical blepharoptosis. Am J Ophthalmol. 1978;85: Krohel GB, Kristan RW, Simon JW, et al. Posttraumatic convergence insufficiency. Ann Ophthalmol. 1986;18: Kubis KC, Danesh-Meyer HV, Savino PJ, et al. The ice test verses the rest test in myasthenia gravis. Ophthalmology. 2000;107: Kuklík M. Poland-öbius syndrome and disruption spectrum affecting the face and extremities: a review paper and presentation of five cases. Acta Chir Plast. 2000;42: Kumagai N, Yuda K, Ohno S. Abnormal vergence eye movement during eyelid closure caused by a pineal tumor. Nippon Ganka Gakkai Zasshi. 1991;95: Kumar J, Kumar A, Saha S. The molar tooth sign of Joubert syndrome. Arch Neurol. 2007;64: Kupersmith MJ. Does early immunotherapy reduce the conversion of ocular myasthenia to generalized myasthenia gravis? J Neuroophthalmol. 2003;23: Kupersmith MJ. Does early treatment of ocular myasthenia gravis with prednisone reduce progression to generalized disease? J Neurol Sci. 2004;217: Kupersmith MJ, Latkany R, Homel P. Development of generalized disease at 2 years in patients with ocular myasthenia gravis. Arch Neurol. 2003;60: Kupersmith MJ, Ying G. Ocular motor dysfunction and ptosis in ocular myasthenia gravis: effects of treatment. Br J Ophthalmol. 2005;89: Kushner BJ. Paresis and restriction of the inferior rectus muscle after orbital floor fracture. Am J Ophthalmol. 1982;94: Kushner BJ. Errors in the three-step test in the diagnosis of vertical strabismus. Ophthalmology. 1989;96: Kushner BJ, ed. Grand rounds #34. A case of exotropia associated with an internuclear ophthalmoplegia. Binocul Vis Eye Muscle Surg Q. 1994;9: Kutschke PJ, Scott WE. The surgical treatment of convergence insufficiency type exodeviation. Am Orthopt J. 2005;71: Lagreze W-DA, Warner JE, Zamani AA, et al. Mesencephalic clefts with associated eye movement disorders. Arch Ophthalmol. 1996;114: Lai T, Chen C, Selva D. Bilateral congenital trigemino-abducens synkinesis. Arch Ophthalmol. 2003;121: Lam BL, Nerad JA, Thompson HS. Paroxysmal eyelid retractions. Am J Ophthalmol. 1992;114(1): Letter Lambert SR, Kriss A, Gresty M, et al. Joubert syndrome. Arch Ophthalmol. 1989;197: Lambert SR, Taylor D, Kriss A, et al. Ocular manifestations of the congenital varicella syndrome. Arch Ophthalmol. 1989;107: Langston JW, Tharp BR. Infantile hemifacial spasm. Arch Neurol. 1976;33: Lapouse R, Monk MA. Behaviour deviation in a representative sample of children. Variation by sex, age, race, social class and family size. Am J Orthopsychiatry. 1964;34: Larrson N-G, Olfords A. Mitochondrial myopathies. Acta Physiol Scand. 2001;171: Le Ber I, Brice A, D rr A. New autosomal recessive cerebellar ataxias with oculomotor apraxia. Curr Neurol Neurosci Rep. 2005;5: Le Ber I, Dubourg O, Benoist JF, et al. Muscle coenzyme Q10 deficiencies in ataxia with oculomotor apraxia 1. Neurology. 2007;68: Le Ber I, Moreira MC, Rivaud-Pechoux S, et al. Cerebellar ataxia with oculomotor apraxia type 1: clinical and genetic studies. Brain. 2003;126: Lee AG, Brazis PW. Chronic progressive external ophthalmoplegia. Curr Neurol Neurosci Rep. 2002;2: Lee DH, Normal D, Newton TH. MR imaging of pineal cysts. J Comput Assist Tomogr. 1987;11: Lee-Y-H LAY, Kim JS. Benzotropine-induced esotropia and mydriasis. J Neuroophthalmol. 2007;27: Lefvert AK, Osterman PO. Newborn infants to myasthenic mothers: a clinical study and an investigation of acetylcholine receptor antibodies in 17 children. Neurology. 1983;33: Legge RH, Weiss HS, Hedges TR III, et al. Periodic alternating gaze deviation in infancy. Neurology. 1992;42: Leigh RJ, Zee DS. The Neurology of Eye Movements. 2nd ed. Philadelphia: F.A. Davis; 1991: Lepore FE. Bilateral cerebral ptosis (Abstract). Neurology. 1986;36(Suppl 1): Lepore FE. Disorders of ocular motility following head trauma. Arch Neurol. 1995;52: Lepore FE, Duvoisin RC. Apraxia of eyelid opening: an involuntary levator inhibition. Neurology. 1985;53: Lessell S. Supranuclear paralysis of monocular elevation. Neurology. 1975;25: Lessell S, Lessell IM, Rizzo JF III. Ocular neuromyotonia after radiation therapy. Am J Ophthalmol. 1986;102: Letson RD. Surgical Management of the ocular congenital fibrosis syndrome. Am Orthopt J. 1980;30: Levi M, Kodsi SR, Rubin SE, et al. Ocular involvement as the initial manifestation of Wegener s granulomatosis in children. J AAPOS. 2008;12: Levy NS, Cassin B, Newman M. Strabismus in children with cerebral palsy. J Pediatr Ophthalmol. 1976;13: Levy EI, Resnick DK, Jannetta PJ, et al. Pediatric hemifacial spasm: the efficacy of microvascular decompression. Pediatr Neurosurg. 1997;27: Lewis JM, Kline LB. Periodic alternating nystagmus associated with periodic alternating skew deviation. J Clin Neuroophthalmol. 1983;3:
68 376 7 Complex Ocular Motor Disorders in Children 492. Lewis RF, Lederman HM, Crawford TO. Ocular motor abnormalities in ataxia telangiectasia. Ann Neurol. 1999;46: Lieppman ME. Accommodation and convergence insufficiency after decompression sickness. Arch Ophthalmol. 1981;99: Lim KH, Engle EC, Demer JL. Abnormalities of the oculomotor nerve in congenital fibrosis of the extraocular muscles and congenital oculomotor palsy. Invest Ophthalmol Vis Sci. 2007;48: Limeira-Soares PH, Rocha EM, Sella W, et al. Tonic pupil due to supranuclear third nerve paresis. J Pediatr Ophthalmol Strabismus. 2003;40: Lindberg K, Brunvand L. Congenital mydriasis combined with aneurysmal dilatation of a persistent ductus arteriosus botalli: a rare syndrome. Acta Ophthalmol Scand. 2005;83: Lindner A, Schalke B, Toyka KV. Outcome in juvenile onset myasthenia gravis: a retrospective study with longterm follow up of 79 patients. Neurology. 1997;244: Loewenfeld IE, Thompson HS. The tonic pupil: a reevaluation. Am J Ophthalmol. 1967;63: Loewenfeld IE, Thompson HS. Ocular paresis with cyclic spasms. A critical review of the literature and a new case. Surv Ophthalmol. 1975;20: Lossos A, Baala L, Soffer D, et al. A novel autosomal recessive myopathy with external ophthalmoplegia linked to chromosome 17p13.1 p12. Brain. 2005;128: Lowenstein DH, Koch TK, Edwards MS. Cerebral ptosis with contralateral arteriovenous malformations: a report of 2 cases. Ann Neurol. 1987;21: Luat AF, Asano E, Chugani HT. Paroxysmal tonic upgaze of childhood with coexistent absence epilepsy. Epileptic Disord. 2007;9: Lubetzki Korn I, Abramsky O. Myasthenia gravis following viral infection. Eur Neurol. 1981;20: Lubkin V. The inverse Marcus Gunn phenomenon: an electromyographic contribution. Arch Neurol. 1978;35: Lyle DJ. Discussion of ocular motor apraxia with a case presentation. Trans Am Ophthalmol Soc. 1961;59: Lynch DR, Braastad CD, Nagan N. Ovarian failure in ataxia with oculomotor apraxia type 2. Am J Med Genet. 2007;143A: MacDonald IM, Johnson ES, Wakeman B. Case report: exotropia surgery in CPEO. Am Orthopt J. 2003;53: Mackey DA, Chan WM, Cyhan C, et al. Congenital fibrosis of the vertically acting extraocular muscles maps to the FEOM3 locus. Hum Genet. 2002;110: Mahler MS, Rangell L. A psychosomatic study of maladie des tics (Gilles de la Tourette s disease). Psychiatr Q. 1943;17: Mahoney NR, Liu GT, Menacker SJ, et al. Pediatric Horner syndrome: etiologies, roles of neuroimaging and urine studies to detect neuroblastoma and other responsible mass lesions. Am J Ophthalmol. 2006;142: Mak SC, Chi CS, Chen CH. Mitochondrial encephalomyopathy presenting with clinical Leigh s disease: report of a case. Chung Hua I Hsueh Tsa Chih Taipei. 1991;47: Marck PA, Kudryk WH. Cherubism. J Otolaryngol. 1992;21: Maria BL, Boltshauser E, Palmer SC, et al. Clinical features and revised diagnostic criteria in Joubert syndrome. J Child Neurol. 1999;14: Maria BL, Bozorgmanesh A, Kimmel K, et al. Quantitative assessment of brainstem development in Joubert syndrome and Dandy- Walker syndrome. J Child Neurol. 2001;16: Maria BL, Hoang KB, Tusa RJ, et al. Joubert syndrome revisited: key ocular motor signs with magnetic resonance imaging correlation. J Child Neurol. 1997;12: Maria BL, Tusa RJ, Hoang KB, et al. Joubert syndrome revisited: key ocular motor signs with MRI correlation. Presented at the 24th Annual Meeting of the Child Neurology Society, Baltimore, Oct Ann Neurol. 1995;38: Marolis S, Pachter BR, Breinin GM. Structural alterations of the extraocular muscle associated with Apert s syndrome. Br J Ophthalmol. 1977;61: Marques-Dias MJ, Gonzalez CH, Rosemberg S. Möbius sequence in children exposed in utero to misoprostol: neuropathological study of three cases. Birth Defects Res A Clin Mol Teratol. 2003;67: a. Miller MT, Ray V, Owens P, Chen P, Chen F. Möbius and Möbiuslike syndromes (TTV-OFM, OMLH). J Pediatr Ophthalmol Strabismus. 1989;26: Marr JE, Green SH, Willshaw HE. Neurodevelopmental implications of ocular motor apraxia. Dev Med Child Neurol. 2005;47: Marran LF, De Land PN, Nguyen AL. Accommodative insufficiency is the primary source of symptoms in children diagnosed with convergence insufficiency. Optom Vis Sci. 2006;83: Maselli RA, Chen D, Mo D, et al. Choline acetyltransferase mutations in myasthenic syndrome due to deficient acetylcholine resynthesis. Muscle Nerve. 2003;27: Mather TR, Saunders RA. Congenital absence of the superior rectus muscle: a case report. J Pediatr Ophthalmol Strabismus. 1987;24: Matthay KK. Neuroblastoma: a clinical challenge and a biological puzzle. CA Cancer J Clin. 1995;45: McCreery KM, Hussein MA, Lee AG, et al. The clinical spectrum of pediatric myasthenia gravis: blepharoptosis, ophthalmoplegia and strabismus: a report of 14 cases. Binocul Vis Strabismus Q. 2002;17: McIntyre A, Lee CJ. The role of botulinum in the management of near reflex spasm. In: de Faber J, ed. Transactions of the 27th Meeting of the ESA, Florence, 2001: McLeod AR, Glaser JS. Deglutition-trochlear synkinesis. Arch Ophthalmol. 1974;92: McMillan TF, Collins AR, Tyers AG, et al. A novel X-linked dominant condition: X-linked congenital isolated ptosis. Am J Hum Genet. 2000;66: Mee J, Paine M, Byrne E, et al. Immunotherapy of ocular myasthenia gravis reduces conversion to generalized myasthenia gravis. J Neuroophthalmol. 2003;23: Meienberg O, Ryffel E. Supranuclear eye movement disorders in Fisher s syndrome of ophthalmoplegia, ataxia, and areflexia. Report of a case and literature review. Arch Neurol. 1983;40: Merihangas JR. Skew deviation in pseudotumor cerebri. Ann Neurol. 1978;4: Metry DW, Dowd CF, Barkovich AJ, et al. The many faces of PHACE syndrome. J Pediatr. 2001;139: Mets M. Tonic upgaze in infancy. Arch Ophthalmol. 1990;108: Metz HS. Double elevator palsy. Arch Ophthalmol. 1979;97: Metz HS. Double elevator palsy. J Pediatr Ophthalmol Strabismus. 1981;18: Metz HS. Double elevator palsy: is there a restriction? Am Orthopt J. 1993;43: Metz HS, Jampolsky A. Alternate day esotropia. J Pediatr Ophthalmol Strabismus. 1979;16: Metz HS, Searl SS. Cyclic vertical deviation. Trans Am Ophthalmol Soc. 1984;82: Mezer E, Nischal KK, Nahjawan N, et al. Hemifacial spasm as the initial manifestation of childhood cerebellar astrocytoma. J AAPOS. 2006;10: Michaelson DJ, Ashwal S. The pathogenesis of stroke in mitochondrial disorders. Mitochondrion. 2004;4: Milani N, Scaioli V, Giomibini S, et al. Hemifacial spasm in a child. Childs Nerv Syst. 1991;7: a. Miller G. Neurological disorders. The mystery of the missing smile. Science 2007;316: Miller MT. Ocular abnormalities in craniofacial dysostosis. Int Ophthalmol Clin. 1984;24:
69 References 542. Miller NR. Walsh and Hoyt: Clinical Neuro-Ophthalmology. 5th ed. Philadelphia: Lippincott Williams & Wilkins; 2005: Miller NR. Walsh and Hoyt s Clinical Neuro-Ophthalmology. 4th ed, vol 2. Baltimore: Williams & Wilkins; 1985: a. Miller MT. Thalidomide embryopathy: a model for the study of congenital incomitant horizontal strabismus. Trans Am Ophthamol Soc. 1991;89: Miller MT, Stromland K. Ocular motility in thalidomide embryopathy. J Pediatr Ophthalmol Strabismus. 1991;28: Miller MT, Strömland K. The Möbius sequence: a relook. J AAPOS. 1999;3: Miller MT, Strömland K, Ventura L, et al. Autism with ophthalmologic malformations: the plot thickens. Trans Am Ophthalmol Soc. 2004;102: Miller MT, Strömland K, Ventura L, et al. Autism associated with conditions characterized by developmental errors in early embryogenesis: a mini review. Int J Dev Neurosci. 2005;23: a. Miller RC, Tewari A, Miller JA, et al. Neuro-ophthalmologic features of spinocerebellar ataxia type 7. J Neuroophthalmol 2009;29: Mims JL. Double Elevator Palsy eye supraducts during stage II general anesthesia supporting hypothesis of (Supra)Nuclear etiology. Binocul Vis Strabismus Q. 2005;20: Mindel JS, Rubenstein AE, Wallace S, et al. Congenital Horner s syndrome does not alter Lisch nodule formation. Ann Neurol. 1994;35: Misulis KE, Fenichel GM. Genetic forms of myasthenia gravis. Pediatr Neurol. 1989;5: Mitchell WG, Davalos-Gonzalez Y, Brumm VL. Opsoclonusataxia-caused by childhood neuroblastoma: developmental and neurological sequelae. Pediatrics. 2002;110: Mitchell CH, Sinatra FR, Crast FW, et al. Intractable watery diarrhea, ganglioneuroblastoma, and vasoactive intestinal peptide. J Pediatr. 1976;89: Miyajima Y, Fukuda M, Kojima S, et al. Wernicke s encephalopathy in a child with acute lymphoblastic leukemia. Am J Pediatr Hematol Oncol. 1993;15: Mizrachi IB, Gomez-Hassan D, Baivas M, et al. Pitfalls in the diagnosis of mitochondrial encephalopathy with lactic acidosis and stroke-like episodes. J Neuroophthalmol. 2006;26: Momtchilova M, Pelosse B, Rocher F, et al. Möbius syndrome: ocular and clinical manifestations. J Fr Ophtalmol. 2007;30: Monsul TN, Patwa HS, Knorr AM, et al. The effect of prednisone on the progression from ocular to generalized myasthenia gravis. J Neurol Sci. 2004;217: Moorthy G, Behrens MM, Drachman DB, et al. Ocular pseudomyasthenia orocular myasthenia plus : a warning to clinicians. Neurology. 1989;39: Moraz S. Oculo-motor disorders in craniofacial malformations. J Maxillofac Surg. 1984;12: Moreira MC, Klur S, Watanabe M, et al. Senataxin, the ortholog of a yeast RNA helicase, is mutant in ataxia-ocular apraxia 2. Nat Genet. 2004;36: Morel E, Eymard B, Vernet-Der Garabedian B, et al. Neonatal myasthenia gravis: a new clinical and immunologic appraisal on 30 cases. Neurology. 1988;38: Morgan MA, Enterline DS, Fukushima T, et al. Endodermal cyst of the oculomotor nerve. Neuroradiology. 2001;43: Morris EB III, Gajjar A, Hoehn ME. Ocular neuromyotonia: video case report. Neurology. 2006;25:E Morris OC, O Day J. Strabismus surgery in the management of diplopia caused by myasthenia gravis. Br J Ophthalmol. 2004;88: Morrison DA, Bibby K, Woodruff G. The harlequin sign and congenital Horner s syndrome. J Neurol Neurosurg Psychiatry. 1997;61: Moschner C, Perlman S, Baloh RW. Oculomotor phenotypes in autosomal dominant ataxias. Arch Neurol. 1998;55: Moster ML, Hoenig EM. Spasm of the near reflex associated with metabolic encephalopathy. Neurology. 1989;39: Moster ML, Schatz NJ, Savino PJ, et al. Alternating skew on lateral gaze (bilateral abducting hypertropia). Ann Neurol. 1988; 23: Muchnick RS, Sanfillippo S, Dunlap EA. Cyclic esotropia developing after strabismus surgery. Arch Ophthalmol. 1976;94: Mullaney P, Vajsar J, Smith R, et al. The natural history and ophthalmic involvement in childhood myasthenia gravis at the hospital for sick children. Ophthalmology. 2000;107: Munoz M, Page LK. Acquired double elevator palsy in a child with a pineocytoma. Am J Ophthalmol. 1995;118: Murphy J, Murphy SF. Myasthenia gravis in identical twins. Neurology. 1986;36: Musarella MA, Ghan HS, DeBoer G, et al. Ocular involvement in neuroblastoma: prognostic implications. Ophthalmology. 1984;91: Nakano M, Yamada K, Fain J, et al. Homozygous mutations in ARIX (PHOS 2A) result in congenital fibrosis of the extraocular muscles type 2. Nat Genet. 2001;118: Namba T, Brunner NG, Brown SB, et al. Familial myasthenia gravis: report of 27 patients in 12 families and review of 164 patients in 73 families. Arch Neurol. 1971;25: Needham GR. Evaluation of five popular methods of tick removal. Pediatrics. 1985;75: Nelson LB, Ingoglia S, Breinin GM. Sensorimotor disturbances in Craniostenosis. J Pediatr Ophthalmol Strabismus. 1981;18: Neugebauer A, Fricke J, Kubisch C. Congenital ocular elevation deficiencies: which are congenital cranial dysinnervation disorders? In: Presented at the 32nd Meeting of the European Strabismological Association, Munich, Germany; September 7 10, Nightingale S, Barton ME. Intermittent vertical supranuclear ophthalmoplegia and ataxia. Mov Disord. 1991;6: Niks EH, Verrips A, Semmekrot BA, et al. A transient neonatal myasthenic syndrome with anti-musk antibodies. Neurology. 2008;70: Nischal KK. Ocular aspects of craniofacial disorders. Am Orthopt J. 2002;52: Nishino I, Spinazzola A, Papadimitriou A, et al. Mitochondrial neurogastrointestinal encephalomyopathy: an autosomal recessive disorder due to thymidine phosphorylase mutations. Ann Neurol. 2000;47: Nishzaki T, Tamaki N, Nishida Y, et al. Bilateral internuclear ophthalmoplegia due to hydrocephalus: a case report. Neurosurgery. 1985;17: Nixon RB, Helveston EM, Miller K, et al. Incidence of strabismus in neonates. Am J Ophthalmol. 1985;100: Noel L-P, Watson AG. Internal ophthalmoplegia following chickenpox. Can J Ophthalmol. 1976;11: Noonan CP, Dillon AB, Swift J, et al. Repeated use of botulinum toxin in the treatment of convergence spasm. In: de Faber J, ed. Transactions of the 27th Meeting of the ESA, Florence, 2001: Nucci P, Brancato R. Oculogyric crisis after the Tensilon test. Graefes Arch Clin Exp Ophthalmol. 1990;228: Odaka M, Yuki N, Yamada M, et al. Bickerstaff s brainstem encephalitis: clinical features of 62 cases and a subgroup associated with Guillian Barre syndrome. Brain. 2003;126: Odel JG, Winterkorn JM, Behrens MM. The sleep test for myasthenia gravis. A safe alternative to Tensilon. J Clin Neuroophthalmol. 1991;11: Oesterle CS, Faulkner WJ, Clay R, et al. Eye bobbing associated with jaw movement. Ophthalmology. 1982;89: Oh SJ, Cho HK. Edrophonium responsiveness not necessarily diagnostic of myasthenia gravis. Neurology. 1979;29:68 76.
70 378 7 Complex Ocular Motor Disorders in Children 591. Ohno K, Tsujino A, Brengman JM, et al. Choline acetyltransferase mutations cause myasthenic syndrome associated with episodic apnea in humans. Proc Natl Acad Sci U S A. 2001;98: Ohtsuka K, Maeda S, Oguri N. Accommodation and convergence palsy caused by lesions in the bilateral superior colliculus. Am J Ophthalmol. 2002;133: Ohtsuka K, Maekawa H, Takeda M, et al. Accommodation and convergence insufficiency with left middle cerebral artery occlusion. Am J Ophthalmol. 1988;106: Ohtsuki H, Hasebe S, Okano M, et al. Strabismus surgery in ocular myasthenia gravis. Ophthalmologica. 1996;210: Okamato K, Ito J, Tokiguchi S, et al. Atrophy of bilateral extraocular muscles. CT and clinical features of seven patients. J Neuroophthalmol. 1996;16: Okamoto K, Tokiguchi S, Furusawa T, et al. MR features of diseases involving bilateral middle cerebellar peduncles. Am J Neuroradiol. 2003;24: Olson RJ, Scott WE. Dissociated phenomena in congenital monocular elevation deficiency. J AAPOS. 1998;2: Opal P, Zoghbi HY. The hereditary ataxias. In: Rimoin DL, Connor JM, Pyeritz RE, eds. Emery and Rimoin s Principles and Practice of Medical Genetics, vol. 3. 5th ed. Philadelphia: Elsevier; 2007: Orrison W, Robertson J. Congenital ocular motor apraxia: a possible disconnection syndrome. Arch Neurol. 1979;36: Ortiz S, Borchert M. Longterm outcomes of pediatric ocular myasthenia gravis. Ophthalmology. 2008;115: Ortube MC, Bhola R, Demer JL. Orbital magnetic resonance imaging of extraocular muscles in chronic progressive external ophthalmoplegia: specific diagnostic findings. J AAPOS. 2006;10: Osserman K, Teng P. Studies in myasthenia-gravis-a rapid diagnostic test. JAMA. 1956;160: Osterele CS, Faulkner WJ, Clay R, et al. Eye bobbing associated with jaw movement. Ophthalmology. 1982;89: Ouanounou S, Saigal G, Birchansky S. Möbius syndrome. Am J Neuroradiol. 2005;26: Ouvrier RA, Billson F. Benign paroxysmal tonic upgaze of childhood. J Child Neurol. 1988;3: a. Ouvrier R, Billson F. Paroxysmal tonic upgaze of childhood a review. Brain Dev. 2005;2;7: Ozawa M, Nishino I, Horai S, et al. Myoclonus epilepsy associated with ragged-red fibers. A G-to-A mutation at nucleotide pair 8363 in mitochondrial trna(lys) in two families. Muscle Nerve. 1997;20: Özkan SB, Aribal ME, Can D, et al. The evaluation of congenital double elevator palsy with magnetic resonance imaging. Neuroophthalmology. 1999;21: Özkan SB, Soylev MF, Karaman ZC, et al. The evaluation of chronic progressive external ophthalmoplegia with computerized tomography. Int Ophthalmol. 2002;24: Parisi MA, Doherty D, Chance PF, et al. Joubert syndrome (and related disorders). Eur J Neurol. 2004;11: Parulekar MV, Dai S, Buncic JR, et al. Head position-dependent changes in ocular torsion and vertical misalignment in skew deviation. Arch Ophthalmol. 2008;126: Patterson MC, Horowitz M, Abel RB, et al. Isolated horizontal supranuclear gaze palsy as a marker of severe systemic involvement in Gaucher s disease. Neurology. 1993;43: a. Manrique RK, Noval S, Aguilar-Amat MJ, et al. Ophthalmic features of spinocerebellar ataxia type 7. J Neuroophthalmol 2009;29: Pedraza S, Gámez J, Rovira A, et al. MRI findings in Möbius syndrome: correlation with clinical features. Neurology. 2000;55: Petrunak JL. The treatment of convergence insufficiency. Am Orthopt J. 1999;49: Petty RK, Harding AE, Morgan-Hughes JA. The clinical features of mitochondrial myopathy. Brain. 1986;109: Phillips PH, Brodsky MC, Henry PM. Congenital ocular motor apraxia with autosomal dominant transmission. Am J Ophthalmol. 2000;129: Phillips PH, Fray KJ, Brodsky MC. Intermittent exotropia increasing with near fixation: a soft sign of neurological disease. Br J Ophthalmol. 2005;89: Phillips PH, Newman NJ. Mitochondrial diseases in pediatric ophthalmology. J AAPOS. 1997;1: Picard A, Lacert P. Disorders of horizontal gaze motility in the cerebral palsy patient. J Fr Ophtalmol. 1984;7: Pickett J, Berg B, Chaplin E, et al. Syndrome of botulism in infancy: clinical and electrophysiologic study. N Engl J Med. 1976;295: Pickwell LD, Hampshire R. Convergence insufficiency in patients taking medicines. Ophthalmic Physiol Opt. 1984;4: Pieh C, Lagreze WA. Congenital cranial dysinnervation disorders (in German). Ophthalmologe. 2007;104: Pieh C, Lengyel D, Neff A, et al. Brainstem hypoplasia in familial horizontal gaze palsy and scoliosis. Neurology. 2002;59: Pierrot-Deseilligny C, Gautier JC, Loron P. Acquired ocular motor apraxia due to bilateral frontoparietal infarcts. Ann Neurol. 1988;23: Pijpers E, van Rijswijk RE, Takx-Kohlen B, et al. A clarithromycin-induced myasthenia gravis. Clin Infect Dis. 1996;22: Pillai P, Dhand UK. Cyclic esotropia with central nervous system disease: report of two cases. J Pediatr Ophthalmol Strabismus. 1987;24: Poretti A, Boltshauser E, Loenneker T, et al. Diffusion tensor imaging in Joubert syndrome. AJNR Am J Neuroradiol. 2007; 28: Prensky AL. Wernicke encephalopathy in infants. J Neuroophthalmol. 2005;25: Presad P, Nair S. Congenital ocular motor apraxia: sporadic and familial: support for natural resolution. J Neuroophthalmol. 1994;14: Prieto-Diaz J, Gamio MS. The surgical innervational effect: utilizing it to treat monocular elevation deficiency strabismus. Binocul Vis Strabismus Q. 2007;22: Pulec JL. Idiopathic hemifacial spasm. Pathogenesis and surgical treatment. Ann Otol Rhinol Laryngol. 1972;81: Racette BA, Golden MS, Tychsen L, et al. Convergence insufficiency in idiopathic Parkinson s disease responsive to levodopa. Strabismus. 1999;7: Ragge NK, Harris CM, Dillon MJ, et al. Ocular tilt reaction due to a mesencephalic lesion in juvenile polyarteritis nodosa. Am J Ophthalmol. 2003;135: Ragge NK, Hoyt WF. Midbrain myasthenia: fatigable ptosis, lid twitch sign, and ophthalmoparesis from a dorsal midbrain glioma. Neurology. 1992;42: Rangan C, Everson G, Cantrell FL. Central alpha-2 adrenergic eye drops: case series of 3 pediatric systemic poisonings. Pediatr Emerg Care. 2008;24: Rappaport L, Urion D, Strand K, et al. Concurrence of congenital ocular motor apraxia and other motor problems: an expanded syndrome. Dev Med Child Neurol. 1987;29: Raymond GL, Crompton JL. Spasm of the near reflex associated with cerebrovascular accident. Aust N Z J Ophthalmol. 1990;18: Reader AL III, Massey EW. Fibromuscular dysplasia of the carotid artery: a cause of congenital Horner s syndrome? Ann Ophthalmol. 1980;12: Reynard M, Wertenbacker C, Behrens M, et al. Ping pong gaze amplified. Neurology. 1979;29: Letter.
71 References 639. Richardson C, Smith T, Schaefer A, et al. Ocular motility findings in chronic progressive external ophthalmoplegia. Eye. 2005;19: Richter CP. Biological Clocks in Medicine and Psychiatry. Springfield, IL: Charles C Thomas; Richter C. Clock-mechanism esotropia in children. Alternate day squint. Johns Hopkins Med J. 1968;122: Riordan-Eva P, Vickers S, McCarry B, et al. Cyclic strabismus without binocular function. J Pediatr Ophthalmol Strabismus. 1993;30: Rivero A, Crovetto L, Lopez L, et al. Single-fiber electromyography of extraocular muscles: a sensitive method for the diagnosis of ocular myasthenia gravis. Muscle Nerve. 1995;18: Rizzo M, Corbett J. Bilateral internuclear ophthalmoplegia reversed by naloxone. Arch Neurol. 1983;40: Ro A, Gummeson B, Orton RB, et al. Vertical congenital ocular motor apraxia. Can J Ophthalmol. 1989;24: Roarty JD, Pron GE, Siegel-Bartelt J, et al. Ocular manifestations of frontonasal dysplasia. Plast Reconstr Surg. 1994;93: Robertson WC, Chun RW, Kornguth SE. Familial infantile myasthenia. Arch Neurol. 1980;37: Robertson WC, Pettigrew LC. Congenital Horner s syndrome and carotid dissection. J Neuroimaging. 2003;13: Rodriguez M, Gomez MR, Howard FM Jr, et al. Myasthenia gravis in children: long-term followup. Ann Neurol. 1983;13: Rogers JW. Internal ophthalmoplegia following chickenpox. Arch Ophthalmol. 1964;71: Romano S, Boddaert N, Desguerre I, et al. Molar tooth sign and superior vermian dysplasia: a radiological, clinical, and genetic study. Neuropediatrics. 2006;37: Romano PE, Stark WJ. Pseudomyopia as a presenting sign in ocular myasthenia gravis. Am J Ophthalmol. 1973;75: Roper-Hall G. Ocular difficulties in the neargaze position. Am Orthopt J. 1980;30: Roper-Hall MJ, Yapp JM. Alternate day squint. In: The First International Congress of Orthoptists, St Louis, MO: CV Mosby; 1968: Ropper AH. Three patients with Fisher s syndrome and normal MRI. Neurology. 1988;38: Roquer J, Cano A, Seoane JL, et al. Myasthenia gravis and ciprofloxacin. Acta Neurol Scand. 1996;94: Rosenberg ML. Spasm of the near reflex mimicking myasthenia gravis. J Clin Neuroophthalmol. 1986;6: Rosenberg ML. Miotic Adie s pupils. J Clin Neuroophthalmol. 1989;9: Ross JV. Ocula varicella with an unusual complication. Am J Ophthalmol. 1961;51: a. Rotig A, Colonna M, Bonnefont JP, et al. Mitochondrial DNA deletion in Pearson s marrow/pancrease syndrome. Lancet 1989;1: Rufa A, Dotti MT, Galli L, et al. Spinocerebellar ataxia type 2 (SCA2) associated with retinal pigmentary degeneration. Eur Neurol. 2002;47: Rutstein RP. Spasm of the near reflex mimicking deteriorating accommodative esotropia. Optom Vis Sci. 2000;77: Ryan FH, Kline LB, Gomez C. Congenital Horner s syndrome resulting from agenesis of the internal carotid artery. Ophthalmology. 2000;107: Ryan SJ, Smith RE. Retinopathy associated with hereditary olivopontocerebellar degeneration. Am J Ophthalmol. 1971;71: Safran AB, Berney J, Safran E. Convergence-evoked eyelid nystagmus. Am J Ophthalmol. 1982;93: Saito H. Congenital Horner s syndrome with unilateral facial flushing. J Neurol Neurosurg Psychiatry. 1990;53: Salisachs P, Lapresle J. Upper lid jerks in the Fisher syndrome. Eur Neurol. 1977;15: Sandberg DI, Souweidane MM. Hemifacial spasm caused by a pilocytic astrocytoma of the fourth ventricle. Pediatr Neurol. 1999;21: Sanders DB, El-Salem K, Massey JM, et al. Clinical aspects of MuSK antibody positive seronegative MG. Neurology. 2003;60: Sanders DB, Howard JF, Johns TR. Single-fiber electromyography in myasthenia gravis. Neurology. 1979;29: Sandyk R. Hemifacial spasm in tuberculous meningitis. Postgrad Med. 1983;59: Sandyk R. Paralysis of upward gaze as a presenting sign of vitamin B 12 deficiency. Eur Neurol. 1984;23: Sano K. Trigemino-oculomotor synkinesis. Neuralgia. 1959;1: Saraiva JM, Baraitser M. Joubert syndrome: a review. Am J Med Genet. 1992;43: Sargent MA, Poskitt KJ, Jan JE. Congenital ocular motor apraxia: imaging findings. AJNR Am J Neuroradiol. 1997;18: Satran D, Pierpoint ME, Dobyns WB. Cerebello-oculo-renal syndromes including Arima, Senior-Løken and COACH syndromes: more than just variants of Joubert syndrome. Am J Med Genet. 1999;86: Sauer C, Levinsohn MN. Horner s syndrome in childhood. Neurology. 1976;26: Sawamura Y, Ikeda J, Ozawa M, et al. Magnetic resonance images reveal a high incidence of asymptomatic pineal cysts in young women. Neurosurgery. 1995;37: Sayed AK, Miller BA, Lack EE, et al. Heterochromia iridis and Horner s syndrome due to paravertebral neurolemmoma. J Surg Oncol. 1983;22: Sayer JA, Otto EA, O Toole JF, et al. The centrosomal protein nephrocystin-6 is mutated in Joubert syndrome and activates transcription factor ATF4. Nat Genet. 2006;38: Scheiman M, Mitchell GL, Cotter S, et al. Accommodative insufficiency is the primary source of symptoms in children diagnosed with convergence insufficiency. Optom Vis Sci. 2006;83: Schinton RA, Jamieson DG. Exercise induced diplopia as a presentation of a midline cerebellar tumor. J Neurol Neurosurg Psychiatry. 1989;52: Schlezinger NS, Corin MS. Myasthenia gravis associated with hyperthyroidism in childhood. Neurology. 1968;18: Schmidt T, Kreibich S. Vertical retraction syndrome. Klin Monatsbl Augenheilkd. 1985;187: Schmidt RD, Schmidt TW. Infant botulism: a case series and review of the literature. J Emerg Med. 1992;10: Schmiedel J, Jackson S, Schäfer J, et al. Mitochondrial cytopathies. J Neurol. 2003;250: Schmitt N, Bowmer EJ, Gregson JD. Tick paralysis in British Columbia. Can Med Assoc J. 1969;100: Schols L, Amoiridis G, Buttner T, et al. Autosomal dominant cerebellar ataxia: phenotypic differences in genetically defined subtypes? Ann Neurol. 1997;42: Schoser BG, Pongratz B. Extraocular mitochondrial myopathies and their differential diagnosis. Strabismus. 2006;14: Schreiner MS, Field E, Ruddy R. Infant botulism: a review of 12 years experience at the Childrens Hospital of Philadelphia. Pediatrics. 1991;87: Scott WE, Jackson OB. Double elevator palsy: the significance of inferior rectus restriction. Am Orthop J. 1977;27: Seaber JH, Chandler AC Jr. A five year study of patients with cerebral palsy and strabismus. In: Moore S, Mein J, Stockbridge L, eds. Orthoptics: Past, Present, and Future. New York: Grune & Stratton; 1976: Senelick RC. Ping pong gaze. Periodic alternating gaze deviation. Neurology. 1976;26:
72 380 7 Complex Ocular Motor Disorders in Children 693. Sethi KD, Hess DC, Harbour RC, et al. Gaze-evoked involuntary movements. Mov Disord. 1990;5: Shahar E, Andraus J. Near reflex accommodative spasm: unusual presentation of generalized photosensitive epilepsy. J Clin Neurosci. 2002;9: Sharpe JA, Johnston JL. Ocular motor paresis versus apraxia. Ann Neurol. 1989;25: Shawkat F, Harris CM, Jacobs M, et al. Eye movement tics. Br J Ophthalmol. 1992;76: Shawkat FS, Kingsley D, Kendall B, et al. Neuroradiological and eye movements correlates in children with intermittent saccade failure: ocular motor apraxia. Neuropediatrics. 1995;26: Shewmon DA. Unilateral straight hair due to stellate ganglion tumour. Ann Neurol. 1983;13: Shillito P, Vincent A, Newsom-Davis J. Congenital myasthenic syndromes. Neuromuscul Disord. 1993;3: Shimko JW. Chronic progressive external ophthalmoplegia. Am Orthopt J. 2005;55: Shults WT, Hoyt WF, Behrens M, et al. Ocular neuromyotonia. A clinical description of six patients. Arch Ophthalmol. 1986;104: Simonsz HJ. Congenital fibrosis syndrome. J Pediatr Ophthalmol Strabismus. 1990;27: Letter Simpson I, Campbell PE. Mediastinal masses in childhood: a review from a paediatric pathologist s point of view. Prog Pediatr Surg. 1991;27: Singer C, Papapetropoulos S, Farronay O. Childhood-onset hemifacial spasm: successful treatment with botulinum toxin. Pediatr Neurol. 2005;33: Smeitink J, Van Den Heuvel L, DiMauro S. The genetics pathology of oxidative phosphorylation. Nat Rev Genet. 2001;2: Smith JL, Cogan DG. Internuclear ophthalmoplegia: a review of fifty-eight cases. Arch Ophthalmol. 1959;61: Smith JL, David NJ. Internuclear ophthalmoplegia: two new clinical signs. Neurology. 1964;14: Snead OC, Benton JW, Dwyer D, et al. Juvenile myasthenia gravis. Neurology. 1980;30: Snir M, Friling R, Kalish Stiebel H, et al. Combined rectus muscle transposition with posterior fixation sutures for the treatment of double-elevator palsy. Ophthalmology. 2005;112: Snir M, Gilad E, Benosira I. An unusual extraocular muscle anomaly in a patient with Crouzon s disease. Br J Ophthalmol. 1982;66: Sommer N, Sigg B, Melms A, et al. Ocular myasthenia gravis: response to long term immunosuppressive treatment. J Neurol Neurosurg Psychiatry. 1997;62: Sondhi N, Archer SM, Helveston EM. Development of normal ocular alignment. J Pediatr Ophthalmol Strabismus. 1988;25: Sorkin JA, Shoffner JM, Grossniklaus HE, et al. Strabismus and mitochondrial defects in chronic progressive external ophthalmoplegia. Am J Ophthalmol. 1997;123: Spach DH, Liles WC, Campbell GL, et al. Tick-borne diseases in the United States. N Engl J Med. 1993;329: Spampinato MV, Kraas J, Maria BL, et al. Absence of decussation of the superior cerebellar peduncles in patients with Joubert syndrome. Am J Med Genet. 2008;146A: Spector RH. Postganglionic Horner syndrome in three patients with coincident middle ear infection. J Neuroophthalmol. 2008;28: Spielmann AC. Convergence excess associated with neurological diseases: surgical treatment. J Fr Ophtalmol. 2006;29: Spierer A, Barak A. Strabismus surgery in children with Möbius syndrome. J AAPOS. 2000;4: Spierer A, Huna R, Rechtman C, et al. Convergence insufficiency secondary to subdural hematoma. Am J Ophthalmol. 1995;120: Staudenmaier C, Buncic JR. Periodic alternating gaze deviation with dissociated secondary face turn. Arch Ophthalmol. 1983;101: Stavis M, Murray M, Jenkins P, et al. Objective improvement from base-in prisms for reading discomfort associated with mini-convergence insufficiency type exophoria in school children. Binocul Vis Strabismus Q. 2002;17: Stechison MT. Cystic glioma with positional oculogyric crisis. J Neurosurg. 1989;71: Letter, comment Steel SH, Harrad RA. Unilateral congenital ptosis with ipsilateral superior rectus muscle overaction. Am J Ophthalmol. 1996;122: Steer AC, Kornberg A. Bickerstaff s brainstem encephalitis associated with Mycoplasma pneumoniae infection. J Child Neurol. 2006;21: Steinlin M, Martin E, Largo R, et al. Congenital ocular motor apraxia: a neurodevelopmental and neuroradiological study. Neuroophthalmology. 1990;10: Steinlin M, Thun-Hohenstein L, Boltshauser E. Congenital oculomotor apraxia. Presentation developmental problems differential diagnosis. Klin Monatsbl Augenheilkd. 1992;200: Stell R, Bronstein AM, Plant GT, et al. Ataxia telangiectasia: a reappraisal of the ocular motor features and their value in the diagnosis of atypical cases. Mov Disord. 1989;4: Stewart JD, Kirkham TH, Mathieson G. Periodic alternating gaze deviation. Neurology. 1979;29: Stout AU, Borchert M. Etiology of eyelid retraction in children: a retrospective study. J Pediatr Ophthalmol Strabismus. 1993;30: Straube A, Witt TN. Oculo-bulbar myasthenic symptoms as the sole sign of tumour involving or compressing the brain stem. J Neurol. 1990;237: Summers CG, MacDonald JT, Wirtschafter JD. Ocular motor apraxia associated with intracranial lipoma. J Pediatr Ophthalmol Strabismus. 1987;24: Sydnor CF, Seaber JH, Buckley EG. Traumatic superior oblique palsies. Ophthalmology. 1982;89: Szobor A. Myasthenia gravis: familial occurrence. A study of 1100 myasthenia gravis patients. Acta Med Hung. 1989;46: Takiyama Y, Nishizawa M, Tanaka H, et al. The gene for Machado Joseph disease maps to human chromosome 14q. Nat Genet. 1993;4: Tamura EE, Hoyt CS. Oculomotor consequences of intraventricular hemorrhages in premature infants. Arch Ophthalmol. 1987;105: Tan KP, Sargent MA, Poskitt KJ, et al. Ocular overelevation in adduction in craniosynostosis: is it the result of excyclorotation of the extraocular muscles? J AAPOS. 2006;9: Tapiero B, Pedespan JM, Rougier MB, et al. Cyclic strabismus: presentation of two new cases and critical review of the literature. J Fr Ophtalmol. 1995;18: Tassinari J. Methyldopa-related convergence insufficiency. J Am Optom Assoc. 1989;60: Tatli B, Saribeyoglu ET, Aydinli N, et al. Neuroblastoma: an unusual presentation with bilateral ptosis. Pediatr Neurol. 2004;30: Taylor D. The visually handicapped baby and family. In: Taylor D et al., eds. Pediatric Ophthalmology. Boston: Blackwell; 1990: Taylor D, Lake BD, Stephens R. Neurolipidoses. In: Wybar K, Taylor D, eds. Pediatric Ophthalmology, Current Aspects. New York: Marcel Dekker; 1983: Terzis JK, Noah EM. Dynamic restoration in Möbius and Möbius-like patients. Plast Reconstr Surg. 2003;111: Thompson HS. Adie s syndrome: some new observations. Trans Am Ophthalmol Soc. 1977;75:
73 References 744. Thompson HS. Light-near dissociation of the pupil. Ophthalmologica. 1984;189: Thompson J, Glasgow L, Warpinski J, et al. Infant botulism: clinical spectrum and epidemiology. Pediatrics. 1980;66: Thompson HS, Zackon DH, Czarnecki JS. Tadpole-shaped pupils caused by segmental spasm of the iris dilator muscle. Am J Ophthalmol. 1983;96: Tischfield MA, Bosley TM, Salih MA, et al. Homozygous HOXA1 mutations disrupt human brainstem, inner ear, cardiovascular and cognitive development. Nat Genet. 2005;37: Todd K, Butterworth RF. Mechanisms of selective neuronal cell death due to thiamine deficiency. Ann N Y Acad Sci. 1999;893: Tomulescu V, Ion V, Kosa A, et al. Thorascopic thymectomy: midterm results. Ann Thorac Surg. 2006;82: Traboulsi EM. Congenital abnormalities of cranial nerve development: overview, molecular mechanisms, and further evidence of heterogeneity and complexity of syndromes with congenital limitation of eye movements. Trans Am Ophthalmol Soc. 2004;102: Traboulsi ET, Jaagar MD, Kattan HM, et al. Congenital fibrosis of the extraocular muscles: report of 24 cases illustrating the clinical spectrum and surgical management. Am Orthopt J. 1993;43: Traboulsi EI, Maumenee IH, Green WR, et al. Olivopontocerebellar atrophy with retinal degeneration. A clinical and ocular histopathologic study. Arch Ophthalmol. 1988;106: Trabousli EI, Lee BA, Mousawi A, et al. Evidence of genetic heterogeneity in autosomal recessive congenital fibrosis of the extraocular muscles. Am J Ophthalmol. 2000;129: Tran DB, Wilson MC, Fox CA, et al. Möbius syndrome with oculomotor nerve paralysis without abducens paralysis. J Neuroophthalmol. 1998;18: Trobe JD. Cyclodeviation in acquired vertical strabismus. Arch Ophthalmol. 1984;102: Trosst BT, Abel L, Noriega J, et al. Acquired cyclic esotropia in an adult. Am J Ophthalmol. 1981;91: Tusa RJ, Hove MT. Ocular and oculomotor signs in Joubert syndrome. J Child Neurol. 1999;14: Tusa RJ, Kaplan PW, Hain TC, et al. Ipsiversive eye deviation and epileptic nystagmus. Neurology. 1990;40: Tychsen L, Imes RK, Hoyt WF. Bilateral congenital restriction of upward eye movement. Arch Neurol. 1986;43: Tzartos SJ, Efthimiadis A, Morel E, et al. Neonatal myasthenia gravis: antigen specificities of antibodies in sera from mothers and their infants. Clin Exp Immunol. 1990;80: Tzoufi M, Sixlimiri P, Makis A, et al. Another case of paroxysmal tonic downgaze in infancy. J Neuroophthalmol. 2009;29: Uemura Y, Tomita M, Tanaka Y. Consecutive cyclic esotropia. J Pediatr Ophthalmol. 1977;14: Uesterle CS. Exercise-induced diplopia. J Pediat Ophthalmol Strabs. 1989;26: Ukachoke C, Ashby P, Basinski A, et al. Usefulness of single fiber EMG for distinguishing neuromuscular from other causes of ocular muscle weakness. Can J Neurol Sci. 1994;21: Urban PP, Marczynski U, Hopf HC. The oculo-auricular phenomenon. Findings in normals and patients with brainstem lesions. Brain. 1993;116: Uretsky SH, Kennerdell JS, Gutai JP. Graves ophthalmopathy in childhood and adolescence. Arch Ophthalmol. 1980;98: Utsch B, Sayer JA, Attanasio M, et al. Identification of the first AHI1 gene mutations in nephronophthisis-associated Joubert syndrome. Pediatr Nephrol. 2006;21: Uyama E, Fujiki N, Uchino M. Exacerbation of myasthenia gravis during interferon-alpha treatment. J Neurol Sci. 1996;144: Uyama E, Yamada K, Kawano H, et al. A Japanese family with FEOM1-linked congenital fibrosis of the extraocular muscles type 1 (CFEOM1) associated with spinal canal stenosis and refinement 381 of the FEOM critical region. Neuromuscul Disord. 2003;13: Valente EM, Brancati F, Silhavy JL, et al. AHI1 gene mutations cause specific forms of Joubert syndrome-related disorders. Ann Neurol. 2006;59: Valente EM, Marsh SE, Castori M, et al. Distinguishing the four genetic causes of Joubert s syndrome-related disorders. Ann Neurol. 2005;57: Valente EM, Silhavy JL, Bracati F, et al. Mutations in CEP290, which encodes a centrosomal protein, cause pleiotropic forms of Joubert syndrome. Nat Genet. 2006;38: van Engelen BG, Renier WO, Gabreels FJ, et al. Bilateral episodic mydriasis as a migraine equivalent in childhood: a case report. Headache. 1991;31: Vargas FR, Schuler-Faccini L, Brunoni D, et al. Prenatal exposure to misoprostol and vascular disruption defects: a case-control study. Am J Hum Genet. 2000;95: Vassella F, Lutschg J, Mumenthaler M. Cogan s congenital ocular motor apraxia in two successive generations. Dev Med Child Neurol. 1972;14: Veggiotti P, Viri M, Lanzi G. Electrical status epilepticus on eye closure: a case report. Neurophysiol Clin. 1992;22: Ventura LO, da Cruz CB, de Almeida HC, et al. Möbius sequence: long-term strabismus surgical outcome. Arq Bras Oftalmol. 2007;70: Verma P, Ogar J. Treatment of acquired autoimmune myasthenia gravis: a topic review. Can J Neurol Sci. 1992;19: Verzijl HT, Padberg GW, Zwarts MJ. The spectrum of Mobius sequence: an electrophysiological study. Brain. 2005;128: Verzijl HT, Valk J, de Vries R, et al. Radiologic evidence for absence of the facial nerve in Möbius syndrome. Neurology. 2005;64: Verzijl HT, van der Zwaag B, Cruysberg JR, et al. Möbius sequence redefined: a syndrome of rhombencephalic maldevelopment. Neurology. 2003;61: Verzijl HT, van der Zwaag B, Lammens HJ, et al. The neuropathology of hereditary congenital facial palsy vs Möbius syndrome. Neurology. 2005;64: Vilarinho L, Santorelli FM, Cardoso ML, et al. Mitochondrial DNA analysis in ocular myopathy. Observations in 29 Portuguese patients. Eur Neurol. 1998;39: Vincent A, Bowen J, Newsom-Davis J, et al. Seronegative generalized myasthenia gravis: clinical features, antibodies, and their targets. Lancet Neurol. 2003;2: Vincent A, Newland C, Croxen R, et al. Genes at the junction: candidates for congenital myasthenic syndromes. Trends Neurosci. 1997;20: von Noorden GK. Resection of both medial rectus muscles in organic convergence insufficiency. Am J Ophthalmol. 1976; 81: von Noorden GK, Tredici TD, Ruttum M. Pseudo-internuclear ophthalmoplegia after surgical paresis of the medial rectus muscle. Am J Ophthalmol. 1984;98: Walker J, Jones BM, Nischal KK. Diplopia post craniofacial surgery for hypertelorism. In: Claes Lsuritzen, ed. Craniofacial Surgery IX. Proceedings of the IX International Congress of the International Society of Craniofacial Surgery. Bologna: Monduzzi Editori; 2001: Wall M, Rosenberg M, Richardson D. Gaze-evoked tinnitus. Neurology. 1987;37: Wallace DC, Lott MT, Brown MD, et al. Report of the Committee on Human Mitochondrial DNA. Baltimore: Johns Hopkins University Press; Wallace DK, Sprunder DT, Helveston EM, et al. Surgical management of strabismus associated with chronic progressive external ophthalmoplegia. Ophthalmology. 1997;104:
74 382 7 Complex Ocular Motor Disorders in Children 792. Wallis DH, Granet DB, Levi L. When the darker eye has the smaller pupil. J AAPOS. 2003;7: Wang SM, Zwaan J, Mullaney PB, et al. Congenital fibrosis of the extraocular muscles type 2, an inherited exotropic strabismus fixus, maps to distal 11q13. Am J Hum Genet. 1998;63: Warmolts JR, Mendell JR. Neurotonia: impulse-induced repetitive discharges in motor nerves in peripheral neuropathy. Ann Neurol. 1980;7: Warner TT, Hammans SR. Practical Neurogenetics. Philadelphia: Saunders Elsevier; Wasserman BN, Chronister TE, Stark BI, et al. Ocular myasthenia and nitrofurantoin. Am J Ophthalmol. 2000;130: Weinstein J, Zweifel TJ, Thompson HS. Congenital Horner s syndrome. Arch Ophthalmol. 1980;98: a. Weiss AH, Doherty D, Parisi M, et al. Eye movement abnormalities in Jpubert syndrome. Invest Ophthalmol Vis Sci 2009;50: Weller M, Wilhelm H, Sommer N, et al. Tonic pupil, areflexia, and segmental anhidrosis: two additional cases of Ross syndrome and review of the literature. J Neurol. 1992;239: Werner M, Bhatti T, Vaishnv H, et al. Isolated anisocoria from an endodermal cyst of the third cranial nerve mimicking an Adie s tonic pupil. J Pediatr Ophthalmol Strabismus. 2005;42: West CE, Repka MX. Tonic pupils associated with neuroblastoma. J Pediatr Ophthalmol Strabismus. 1992;29: White JW. Paralysis of the superior rectus and inferior oblique muscles of the same eye. Arch Ophthalmol. 1942;27: White BV, Fulton MN. A rare pupillary defect inherited by identical twins. J Hered. 1937;28: Whitely AM, Schwartz MS, Sachs JA, Swash M. Congenital myasthenia gravis: clinical and HLA studies in two brothers. J Neurol Neurosurg Psychiatry. 1976;39: Wigginton JM, Thill P. Infant botulism. A review of the literature. Clin Pediatr (Phila). 1993;32: Wilder WM, Williams JP, Hupp SL. Computerized tomographic findings in two cases of congenital fibrosis syndrome. Comput Med Imaging Graph. 1991;15: Wilkins RH. Hemifacial spasm: a review. Surg Neurol. 1991;36: Williamson PD, Thadani VM, Darcey TM, et al. Occipital lobe epilepsy: clinical characteristics, seizure spread patterns, and results of surgery. Ann Neurol. 1992;31: Wilson SA. A note on an associated movement of the eyes and ears in man. Rev Neurol Psychiatry. 1908;6: a. Wilson ME, Eustis HS Jr, Parks MM. Brown s syndrome. Surv Ophthalmol 1989;34: Windsor CE, Berg EF. Circadian heterotropia. Am J Ophthalmol. 1981;91: Winkler K. Paraneoplastic symptoms of neuroblastoma. Monatsschr Kinderheilkd. 1976;124: Wittbrodt ET. Drugs and myasthenia gravis. Arch Intern Med. 1997;157: Wolfe GI, Taylor CL, Flamm ES, et al. Ocular tilt reaction resulting from vestibuloacoustic nerve surgery. Neurosurgery. 1993;32: Discussion Wolsey DH, Warner JE. Paroxysmal downgaze in two healthy infants. J Neuroophthalmol. 2006;26: Woodruff G, Buncic JR, Morin JD. Horner s syndrome in children. J Pediatr Ophthalmol Strabismus. 1988;25: Woodruff BA, Griffin PM, McCroskey LM, et al. Clinical and laboratory comparison of botulinum from toxin types A, B, and E in the United States, J Infect Dis. 1992;166: Woody RC, Reynolds JD. Association of bilateral internuclear ophthalmoplegia and myelomeningocele with Arnold Chiari, type II. J Clin Neuroophthalmol. 1985;5: Wortham EV, Crawford JS. Brown syndrome in twins. Am J Ophthalmol. 1988;25: Wright KW. Superior oblique silicone expander for Brown syndrome and superior oblique overaction. J Pediatr Ophthalmol Strabismus. 1991;28: Wright KW, Liu GY, Murphree AL, et al. Double elevator palsy, ptosis, and jaw winking. Am Orthopt J. 1989;39: Yamada K, Andrews C, Chan WM, et al. Heterozygous mutations of the kinesin KIF21A in congenital fibrosis of the extraocular muscles type 1 (CFEOM1). Nat Genet. 2003;35: Yamada K, Hunter DG, Andrews C, et al. A novel KIF21A mutation in a patient with congenital fibrosis of the extraocular muscles and Marcus Gunn jaw-winking phenomenon. Arch Ophthalmol. 2005;123: Yazdani A, Chung DC, Abbaszadegan MR, et al. A novel PHOX2A/ARIX mutation in an Iranian family with congenital fibrosis of extraocular muscles type 2 (CFEOM2). Am J Ophthalmol. 2003;136: Yazdani A, Traboulsi EI. Classification and surgical management of patients with familial and sporadic forms of congenital fibrosis of the extraocular muscles. Ophthalmology. 2004;111: Yee RD, Duffin RM, Baloh RW, et al. Familial congenital paralysis of horizontal gaze. Arch Ophthalmol. 1982;100: Yokochi K. Paroxysmal ocular downward deviation in neurologically impaired infants. Pediatr Neurol. 1991;7: Yoon G, Westmacott R, MacMillan L, et al. Complete deletion of the aprataxin gene: ataxia with oculomotr apraxia type 1 with severe phenotype and cognitive deficit. J Neurol Neurosurg Psychiatry. 2008;79: Young LA. Dysthyroid ophthalmopathy in children. J Pediatr Ophthalmol Strabismus. 1979;16: Yuki N. Acute paresis of extraocular muscles associated with IgG antigq1b antibody. Ann Neurol. 1996;39: a. Yuzuriha S, Matsuo K, Ishigaki Y, et al. Efferent and afferent innervations of Mueller's muscle related to involuntary contraction of the levator muscle: important for avoiding injury during eyelid surgery. Br J Plast Surg. 2005;58: Zackon DH, Noel LP. Ocular motor apraxia following cardiac surgery. Can J Ophthalmol. 1991;26: Zafeiriou DI, Economou M, Koliouskas D, et al. Congenital Horner s syndrome associated with cervical neuroblastoma. Eur J Paediatr Neurol. 2006;10: Zafeiriou DI, Mauromatis IV, Hatijsevastou HK, et al. Benign congenital hemifacial spasm. Pediatr Neurol. 1997;17: Zak TA. Benign episodic bilateral juvenile internal ophthalmoplegia. J Pediatr Ophthalmol Strabismus. 1983;20: Zaret CR, Behrens MM, Eggers HM. Congenital ocular motor apraxia and brain stem tumors. Arch Ophthalmol. 1980;98: Zee DS. Internuclear ophthalmoplegia: pathophysiology and diagnosis. Baillieres Clin Neurol. 1992;1: Zee DS. Considerations on the mechanisms of alternating skew deviation in patients with cerebellar lesions. J Vestib Res. 1996;6: Zee DS, Hain TC, Carl JR. Abduction nystagmus in internuclear ophthalmoplegia. Ann Neurol. 1987;21: Zee DS, Yee RD, Singer HS. Congenital ocular motor apraxia. Brain. 1977;100: Zeviani M, Di Donato S. Mitochondrial disorders. Brain. 2004; 127: Ziffer AJ, Rosenbaum AL, Demer JL, et al. Congenital double elevator palsy: vertical saccadic velocity utilizing the scleral
75
Fourth Nerve Palsy (a.k.a. Superior Oblique Palsy)
 Hypertropia Hypertropia is a type of strabismus characterized by vertical misalignment of the eyes. Among the many causes of vertical strabismus, one of the most common is a fourth nerve palsy (also known
Hypertropia Hypertropia is a type of strabismus characterized by vertical misalignment of the eyes. Among the many causes of vertical strabismus, one of the most common is a fourth nerve palsy (also known
Esotropia (Crossed Eye(s))
 Esotropia (Crossed Eye(s)) Esotropia is a type of strabismus or eye misalignment in which the eyes are "crossed," that is, while one eye looks straight ahead, the other eye is turned in toward the nose.
Esotropia (Crossed Eye(s)) Esotropia is a type of strabismus or eye misalignment in which the eyes are "crossed," that is, while one eye looks straight ahead, the other eye is turned in toward the nose.
Primary Motor Pathway
 Understanding Eye Movements Abdullah Moh. El-Menaisy, MD, FRCS Chief, Neuro-ophthalmology ophthalmology & Investigation Units, Dhahran Eye Specialist Hospital, Dhahran, Saudi Arabia Primary Motor Pathway
Understanding Eye Movements Abdullah Moh. El-Menaisy, MD, FRCS Chief, Neuro-ophthalmology ophthalmology & Investigation Units, Dhahran Eye Specialist Hospital, Dhahran, Saudi Arabia Primary Motor Pathway
Eye movement problems in adults
 Eye movement problems in adults 07/12/2015 A. Dahlmann-Noor Dr med PhD FRCOphth FRCS(Ed) DipMedEd Consultant Ophthalmologist Clinical Trials Lead Paediatric Ophthalmology Horizontal misalignment (Cyclo)vertical
Eye movement problems in adults 07/12/2015 A. Dahlmann-Noor Dr med PhD FRCOphth FRCS(Ed) DipMedEd Consultant Ophthalmologist Clinical Trials Lead Paediatric Ophthalmology Horizontal misalignment (Cyclo)vertical
Exotropias: A Brief Review. Leila M. Khazaeni, MD November 2, 2008
 Exotropias: A Brief Review Leila M. Khazaeni, MD November 2, 2008 Exotropia Myths Myth #1 He/she will grow out of it FALSE 75% of XTs show progression over a 3 year period Myth #2 The only treatment choice
Exotropias: A Brief Review Leila M. Khazaeni, MD November 2, 2008 Exotropia Myths Myth #1 He/she will grow out of it FALSE 75% of XTs show progression over a 3 year period Myth #2 The only treatment choice
Pseudoabducens palsy: When a VI nerve palsy is not a VI nerve palsy
 Pseudoabducens palsy: When a VI nerve palsy is not a VI nerve palsy Emily S. Birkholz, MD, and Michael Wall, MD December 30, 2009 CC: 44 year old male with diplopia HPI: This 44 year old man with a history
Pseudoabducens palsy: When a VI nerve palsy is not a VI nerve palsy Emily S. Birkholz, MD, and Michael Wall, MD December 30, 2009 CC: 44 year old male with diplopia HPI: This 44 year old man with a history
Common visual symptoms and findings in MS: Clues and Identification
 Common visual symptoms and findings in MS: Clues and Identification Teresa C Frohman, PA-C, MSCS Neuro-ophthalmology Research Manager, UT Southwestern Medical Center at Dallas Professor Biomedical Engineering,
Common visual symptoms and findings in MS: Clues and Identification Teresa C Frohman, PA-C, MSCS Neuro-ophthalmology Research Manager, UT Southwestern Medical Center at Dallas Professor Biomedical Engineering,
Care of the Patient with Strabismus: Esotropia and Exotropia
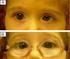 OPTOMETRIC CLINICAL PRACTICE GUIDELINE OPTOMETRY: THE PRIMARY EYE CARE PROFESSION Doctors of optometry are independent primary health care providers who examine, diagnose, treat, and manage diseases and
OPTOMETRIC CLINICAL PRACTICE GUIDELINE OPTOMETRY: THE PRIMARY EYE CARE PROFESSION Doctors of optometry are independent primary health care providers who examine, diagnose, treat, and manage diseases and
The Physiology of the Senses Lecture 11 - Eye Movements www.tutis.ca/senses/
 The Physiology of the Senses Lecture 11 - Eye Movements www.tutis.ca/senses/ Contents Objectives... 2 Introduction... 2 The 5 Types of Eye Movements... 2 The eyes are rotated by 6 extraocular muscles....
The Physiology of the Senses Lecture 11 - Eye Movements www.tutis.ca/senses/ Contents Objectives... 2 Introduction... 2 The 5 Types of Eye Movements... 2 The eyes are rotated by 6 extraocular muscles....
Concussion/MTBI Certification Series. Featuring: Frederick R Carrick, DC, PhD Distinguished Professor of Neurology, Life University
 Concussion/MTBI Certification Series Featuring: Frederick R Carrick, DC, PhD Distinguished Professor of Neurology, Life University Please note that spaces are limited for this specialty certification program.
Concussion/MTBI Certification Series Featuring: Frederick R Carrick, DC, PhD Distinguished Professor of Neurology, Life University Please note that spaces are limited for this specialty certification program.
CONGENITAL NYSTAGMUS WHEN TO RECORD HOW TO TREAT 2009
 CONGENITAL NYSTAGMUS WHEN TO RECORD HOW TO TREAT 2009 LIONEL KOWAL Royal Victorian Eye and Ear Hospital Center for Eye Research Australia Melbourne, Australia TYPES OF CONGENITAL NYSTAGMUS cn cn: any type
CONGENITAL NYSTAGMUS WHEN TO RECORD HOW TO TREAT 2009 LIONEL KOWAL Royal Victorian Eye and Ear Hospital Center for Eye Research Australia Melbourne, Australia TYPES OF CONGENITAL NYSTAGMUS cn cn: any type
GAZE STABILIZATION SYSTEMS Vestibular Ocular Reflex (VOR) Purpose of VOR Chief function is to stabilize gaze during locomotion. Acuity declines if
 GAZE STABILIZATION SYSTEMS Vestibular Ocular Reflex (VOR) Purpose of VOR Chief function is to stabilize gaze during locomotion. Acuity declines if slip exceeds 3-5 deg/sec. Ex: Head bobbing and heel strike
GAZE STABILIZATION SYSTEMS Vestibular Ocular Reflex (VOR) Purpose of VOR Chief function is to stabilize gaze during locomotion. Acuity declines if slip exceeds 3-5 deg/sec. Ex: Head bobbing and heel strike
In normal vision, both eyes are precisely aligned on an object
 3 Binocular Vision and Introduction to Strabismus Kenneth W. Wright In normal vision, both eyes are precisely aligned on an object of regard, so the images from that object fall on the fovea of each eye.
3 Binocular Vision and Introduction to Strabismus Kenneth W. Wright In normal vision, both eyes are precisely aligned on an object of regard, so the images from that object fall on the fovea of each eye.
Management Approach to Isolated Ocular Motor Nerve Palsies
 Management Approach to Isolated Ocular Motor Nerve Palsies Alfredo A. Sadun, MD, PhD Thornton Professor of Vision Doheny Eye Institute Departments of Ophthalmology and Neurosurgery Keck/USC School of Medicine
Management Approach to Isolated Ocular Motor Nerve Palsies Alfredo A. Sadun, MD, PhD Thornton Professor of Vision Doheny Eye Institute Departments of Ophthalmology and Neurosurgery Keck/USC School of Medicine
Institute of Ophthalmology. Thyroid Eye Disease. aka Thyroid Associated Ophthalmopathy
 Institute of Ophthalmology Thyroid Eye Disease aka Thyroid Associated Ophthalmopathy Causes TED/TAO is an eye disease associated with disease of the thyroid gland Most commonly, it occurs with an overactive
Institute of Ophthalmology Thyroid Eye Disease aka Thyroid Associated Ophthalmopathy Causes TED/TAO is an eye disease associated with disease of the thyroid gland Most commonly, it occurs with an overactive
Outcome of Surgery for Bilateral Third Nerve Palsy
 CLINICAL INVESTIGATIONS Outcome of Surgery for Bilateral Third Nerve Palsy Kazuhiro Aoki, Tatsushi Sakaue, Nobue Kubota and Toshio Maruo Department of Ophthalmology, Teikyo University School of Medicine,
CLINICAL INVESTIGATIONS Outcome of Surgery for Bilateral Third Nerve Palsy Kazuhiro Aoki, Tatsushi Sakaue, Nobue Kubota and Toshio Maruo Department of Ophthalmology, Teikyo University School of Medicine,
A Flow Chart For Classification Of Nystagmus
 A Flow Chart For Classification Of Nystagmus Is fixation impaired because of a slow drift, or an intrusive saccade, away from the target? If a slow drift is culprit Jerk Pendular Unidrectional (constant
A Flow Chart For Classification Of Nystagmus Is fixation impaired because of a slow drift, or an intrusive saccade, away from the target? If a slow drift is culprit Jerk Pendular Unidrectional (constant
Thyroid Eye Disease. Anatomy: There are 6 muscles that move your eye.
 Thyroid Eye Disease Your doctor thinks you have thyroid orbitopathy. This is an autoimmune condition where your body's immune system is producing factors that stimulate enlargement of the muscles that
Thyroid Eye Disease Your doctor thinks you have thyroid orbitopathy. This is an autoimmune condition where your body's immune system is producing factors that stimulate enlargement of the muscles that
Doctor, I See Double : Managing Cranial Nerve Palsies
 1 Doctor, I See Double : Managing Cranial Nerve Palsies Joseph W. Sowka, OD, FAAO, Diplomate Professor of Optometry Nova Southeastern University, College of Optometry 3200 South University Drive Fort Lauderdale,
1 Doctor, I See Double : Managing Cranial Nerve Palsies Joseph W. Sowka, OD, FAAO, Diplomate Professor of Optometry Nova Southeastern University, College of Optometry 3200 South University Drive Fort Lauderdale,
Internuclear ophthalmoplegia: recovery and plasticity
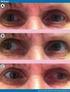 Internuclear ophthalmoplegia: recovery and plasticity M. J. Doslak, L. B. Kline, L. F. Dell'Osso, and R. B. Daroff We studied refixational eye movements of a patient during the gradual resolution of an
Internuclear ophthalmoplegia: recovery and plasticity M. J. Doslak, L. B. Kline, L. F. Dell'Osso, and R. B. Daroff We studied refixational eye movements of a patient during the gradual resolution of an
PUPILS AND NEAR VISION. Akilesh Gokul PhD Research Fellow Department of Ophthalmology
 PUPILS AND NEAR VISION Akilesh Gokul PhD Research Fellow Department of Ophthalmology Iris Anatomy Two muscles: Radially oriented dilator (actually a myo-epithelium) - like the spokes of a wagon wheel Sphincter/constrictor
PUPILS AND NEAR VISION Akilesh Gokul PhD Research Fellow Department of Ophthalmology Iris Anatomy Two muscles: Radially oriented dilator (actually a myo-epithelium) - like the spokes of a wagon wheel Sphincter/constrictor
Less common vestibular disorders presenting with funny turns
 Less common vestibular disorders presenting with funny turns Charlotte Agrup, Department of Neuro-otology, The National Hospital for Neurology and Neurosurgery, London Making the diagnosis Making the diagnosis
Less common vestibular disorders presenting with funny turns Charlotte Agrup, Department of Neuro-otology, The National Hospital for Neurology and Neurosurgery, London Making the diagnosis Making the diagnosis
Surgical Outcome Of Incomitant Exotropia In Patients With Partial Third Nerve Palsy
 Surgical Outcome Of Incomitant Exotropia In Patients With Partial Third Nerve Palsy Amit Mohan, MS ; Sudhir Singh, MS ; Dr V.C. Bhatnagar, MS, DNB Dr Amit Mohan, MS Jr.Consultant Global Hospital Institute
Surgical Outcome Of Incomitant Exotropia In Patients With Partial Third Nerve Palsy Amit Mohan, MS ; Sudhir Singh, MS ; Dr V.C. Bhatnagar, MS, DNB Dr Amit Mohan, MS Jr.Consultant Global Hospital Institute
II. VESTIBULAR SYSTEM OVERVIEW
 HM513 Vertigo and Dizziness; Vestibular System Disorders - Summary Eric Eggenberger*, DO and Kathryn Lovell, PhD *Co-Director, MSU Neuro-Visual Unit; *Director, MSU Ocular Motility Lab Department of Neurology
HM513 Vertigo and Dizziness; Vestibular System Disorders - Summary Eric Eggenberger*, DO and Kathryn Lovell, PhD *Co-Director, MSU Neuro-Visual Unit; *Director, MSU Ocular Motility Lab Department of Neurology
Vestibular Assessment
 Oculomotor Examination A. Tests performed in room light Vestibular Assessment 1. Spontaneous nystagmus 2. Gaze holding nystagmus 3. Skew deviation 4. Vergence 5. Decreased vestibular ocular reflex i. Head
Oculomotor Examination A. Tests performed in room light Vestibular Assessment 1. Spontaneous nystagmus 2. Gaze holding nystagmus 3. Skew deviation 4. Vergence 5. Decreased vestibular ocular reflex i. Head
List of diagnostic flowcharts
 List of diagnostic flowcharts Chapter 3 Visual loss Transient visual loss 43 Sudden or rapidly progressive visual loss 46 Gradual visual loss 61 Chapter 4 The red eye One red eye, decreased vision 83 One
List of diagnostic flowcharts Chapter 3 Visual loss Transient visual loss 43 Sudden or rapidly progressive visual loss 46 Gradual visual loss 61 Chapter 4 The red eye One red eye, decreased vision 83 One
DIPLOPIA - DON T PANIC (JUST PUT ON YOUR THINKING CAP)
 DIPLOPIA - DON T PANIC (JUST PUT ON YOUR THINKING CAP) Lisa Rovick, CO, COMT This article and accompanying quiz are worth.5 JCAHPO Group A continuing education credit. CONTINUING EDUCATION CREDITS ARE
DIPLOPIA - DON T PANIC (JUST PUT ON YOUR THINKING CAP) Lisa Rovick, CO, COMT This article and accompanying quiz are worth.5 JCAHPO Group A continuing education credit. CONTINUING EDUCATION CREDITS ARE
NEUROLOCALIZATION MADE EASY
 NEUROLOCALIZATION MADE EASY Jared B. Galle, DVM, Diplomate ACVIM (Neurology) Dogwood Veterinary Referral Center 4920 Ann Arbor-Saline Road Ann Arbor, MI 48103 Localizing a neurologic problem to an anatomical
NEUROLOCALIZATION MADE EASY Jared B. Galle, DVM, Diplomate ACVIM (Neurology) Dogwood Veterinary Referral Center 4920 Ann Arbor-Saline Road Ann Arbor, MI 48103 Localizing a neurologic problem to an anatomical
Evaluation of Diplopia: An Anatomic and Systematic Approach
 Clinical Review Article Evaluation of Diplopia: An Anatomic and Systematic Approach Victoria S. Pelak, MD Double vision, or diplopia, is a symptom with many potential causes that can involve many different
Clinical Review Article Evaluation of Diplopia: An Anatomic and Systematic Approach Victoria S. Pelak, MD Double vision, or diplopia, is a symptom with many potential causes that can involve many different
Skew deviation is a vertical strabismus caused by
 AAPOS Workshops Understanding skew deviation and a new clinical test to differentiate it from trochlear nerve palsy Agnes M. F. Wong, MD, PhD SUMMARY Skew deviation is a vertical strabismus caused by a
AAPOS Workshops Understanding skew deviation and a new clinical test to differentiate it from trochlear nerve palsy Agnes M. F. Wong, MD, PhD SUMMARY Skew deviation is a vertical strabismus caused by a
Epilepsy 101: Getting Started
 American Epilepsy Society 1 Epilepsy 101 for nurses has been developed by the American Epilepsy Society to prepare professional nurses to understand the general issues, concerns and needs of people with
American Epilepsy Society 1 Epilepsy 101 for nurses has been developed by the American Epilepsy Society to prepare professional nurses to understand the general issues, concerns and needs of people with
Disclosure Statement. I have no financial interest in any of material presented today
 The eyes have it PEARLS IN EVALUATION, TREATMENT, AND REFERRAL OF PEDIATRIC OCULAR CONDITIONS AND TRAUMA MICHAEL G. HUNT, MD PEDIATRIC EYE SPECIALISTS, LLP Disclosure Statement I have no financial interest
The eyes have it PEARLS IN EVALUATION, TREATMENT, AND REFERRAL OF PEDIATRIC OCULAR CONDITIONS AND TRAUMA MICHAEL G. HUNT, MD PEDIATRIC EYE SPECIALISTS, LLP Disclosure Statement I have no financial interest
Basic Cranial Nerve Examination
 Basic Cranial Nerve Examination WIPE Wash hands Introduce yourself Permission Position (Patient sitting facing you, maintain comparable eye level) Exposure (Face exposed only, i.e. remove hats etc) Identify
Basic Cranial Nerve Examination WIPE Wash hands Introduce yourself Permission Position (Patient sitting facing you, maintain comparable eye level) Exposure (Face exposed only, i.e. remove hats etc) Identify
Ptosis. Ptosis and Pupils. Ptosis. Palpebral fissure. Blepharospasm. Blepharospasm or ptosis? Bilateral, longstanding, familial: Palpebral fissure
 Ptosis and Pupils Ptosis Jack CF Chong, MS, MD 99/03/06 Ptosis Bilateral, longstanding, familial: CPEO (chronic progressive external ophthalmoplegia) D/D: Acute, recent, unilateral Palpebral fissure Palpebral
Ptosis and Pupils Ptosis Jack CF Chong, MS, MD 99/03/06 Ptosis Bilateral, longstanding, familial: CPEO (chronic progressive external ophthalmoplegia) D/D: Acute, recent, unilateral Palpebral fissure Palpebral
Developmental delay and Cerebral palsy. Present the differential diagnosis of developmental delay.
 Developmental delay and Cerebral palsy objectives 1. developmental delay Define developmental delay Etiologies of developmental delay Present the differential diagnosis of developmental delay. 2. cerebral
Developmental delay and Cerebral palsy objectives 1. developmental delay Define developmental delay Etiologies of developmental delay Present the differential diagnosis of developmental delay. 2. cerebral
Physical and Mental Conditions Guidelines VISION CONDITIONS AND ACTIONS Page 5.4
 Physical and Mental Conditions Guidelines VISION CONDITIONS AND ACTIONS Page 5.4 AMBLYOPIA (Lazy Eye) A reduction in the acuteness of vision without apparent eye disease. This condition cannot be entirely
Physical and Mental Conditions Guidelines VISION CONDITIONS AND ACTIONS Page 5.4 AMBLYOPIA (Lazy Eye) A reduction in the acuteness of vision without apparent eye disease. This condition cannot be entirely
James H. Hall Eye Center
 James H. Hall Eye Center Established in 1979 Annual Activity Report September, 2010 to September, 2011 COMMUNITY CARE STATISTICS Served 2,000 patients including pre and post-operative visits Our physicians
James H. Hall Eye Center Established in 1979 Annual Activity Report September, 2010 to September, 2011 COMMUNITY CARE STATISTICS Served 2,000 patients including pre and post-operative visits Our physicians
Getting Ready for ICD-10. Dianna Hoskins, OCS Cincinnati Eye Institute
 Dianna Hoskins, OCS Cincinnati Eye Institute Chart Documentation: Will your documentation stand up to ICD-10? Do you always mark which eye, severity or status of the disease (chronic or acute), site, etiology
Dianna Hoskins, OCS Cincinnati Eye Institute Chart Documentation: Will your documentation stand up to ICD-10? Do you always mark which eye, severity or status of the disease (chronic or acute), site, etiology
DIAGNOSTIC CRITERIA OF STROKE
 DIAGNOSTIC CRITERIA OF STROKE Diagnostic criteria are used to validate clinical diagnoses. Here below MONICA diagnostic criteria are reported. MONICA - MONItoring trends and determinants of CArdiovascular
DIAGNOSTIC CRITERIA OF STROKE Diagnostic criteria are used to validate clinical diagnoses. Here below MONICA diagnostic criteria are reported. MONICA - MONItoring trends and determinants of CArdiovascular
To: all optometrists and billing staff
 Number: Opto 27 Date: September 29, 2011 Page: 1 of 1 Subject: Schedule of Optometric Benefits amendments October 1, 2011/New explanatory code list Reference: Schedule of Optometric Benefits To: all optometrists
Number: Opto 27 Date: September 29, 2011 Page: 1 of 1 Subject: Schedule of Optometric Benefits amendments October 1, 2011/New explanatory code list Reference: Schedule of Optometric Benefits To: all optometrists
Diplopia, or double vision, is a frequently encountered. Binocular Diplopia. A Practical Approach ORIGINAL ARTICLE
 ORIGINAL ARTICLE A Practical Approach Janet C. Rucker, MD, and Robert L. Tomsak, MD, PhD Background: Diplopia is a common complaint in both inpatient and outpatient neurologic practice. Its causes are
ORIGINAL ARTICLE A Practical Approach Janet C. Rucker, MD, and Robert L. Tomsak, MD, PhD Background: Diplopia is a common complaint in both inpatient and outpatient neurologic practice. Its causes are
Understanding Nystagmus: Diagnosis, Related Disorders, Treatment, and Research
 Understanding Nystagmus: Diagnosis, Related Disorders, Treatment, and Research Mitra Maybodi,, MD Children s National Medical Center George Washington University School of Medicine and Health Sciences
Understanding Nystagmus: Diagnosis, Related Disorders, Treatment, and Research Mitra Maybodi,, MD Children s National Medical Center George Washington University School of Medicine and Health Sciences
REPORT TO THE STATEWIDE OPHTHALMOLOGY SERVICE (SOS) OF THE GREATER METROPOLITAN CLINICAL TASKFORCE (GMCT)
 REPORT TO THE STATEWIDE OPHTHALMOLOGY SERVICE (SOS) OF THE GREATER METROPOLITAN CLINICAL TASKFORCE (GMCT) The Orthoptist and the management of visual problems in inpatients with stroke: A pilot study to
REPORT TO THE STATEWIDE OPHTHALMOLOGY SERVICE (SOS) OF THE GREATER METROPOLITAN CLINICAL TASKFORCE (GMCT) The Orthoptist and the management of visual problems in inpatients with stroke: A pilot study to
Torsional Diplopia 1,2
 Supplement Handout American Orthoptic Council Workshop: When the Patient Sees Double and the Doctor Sees Nothing A Guide to Double Vision AAPOS 37 th Annual Meeting San Diego, CA April 1, 2011 Torsional
Supplement Handout American Orthoptic Council Workshop: When the Patient Sees Double and the Doctor Sees Nothing A Guide to Double Vision AAPOS 37 th Annual Meeting San Diego, CA April 1, 2011 Torsional
Paralytic Strabismus: Third, Fourth, and Sixth Nerve Palsy
 Paralytic Strabismus: Third, Fourth, and Sixth Nerve Palsy Sashank Prasad, MD a, *, Nicholas J. Volpe, MD b KEYWORDS Paralytic strabismus Nerve palsy Ocular motor nerves Eye movement abnormalities ANATOMY
Paralytic Strabismus: Third, Fourth, and Sixth Nerve Palsy Sashank Prasad, MD a, *, Nicholas J. Volpe, MD b KEYWORDS Paralytic strabismus Nerve palsy Ocular motor nerves Eye movement abnormalities ANATOMY
a guide to understanding facial palsy a publication of children s craniofacial association
 a guide to understanding facial palsy a publication of children s craniofacial association a guide to understanding facial palsy this parent s guide to facial palsy is designed to answer questions that
a guide to understanding facial palsy a publication of children s craniofacial association a guide to understanding facial palsy this parent s guide to facial palsy is designed to answer questions that
Functions of the Brain
 Objectives 0 Participants will be able to identify 4 characteristics of a healthy brain. 0 Participants will be able to state the functions of the brain. 0 Participants will be able to identify 3 types
Objectives 0 Participants will be able to identify 4 characteristics of a healthy brain. 0 Participants will be able to state the functions of the brain. 0 Participants will be able to identify 3 types
AMERICAN ACADEMY OF NEUROLOGY NEURO-OPHTHALMOLOGY/NEURO-OTOLOGY FELLOWSHIP CORE CURRICULUM
 AMERICAN ACADEMY OF NEUROLOGY NEURO-OPHTHALMOLOGY/NEURO-OTOLOGY FELLOWSHIP CORE CURRICULUM A CURRICULUM FOR NEURO-OPHTHALMOLOGY Definition and Scope Neuro-ophthalmology focuses on brain and systemic abnormalities
AMERICAN ACADEMY OF NEUROLOGY NEURO-OPHTHALMOLOGY/NEURO-OTOLOGY FELLOWSHIP CORE CURRICULUM A CURRICULUM FOR NEURO-OPHTHALMOLOGY Definition and Scope Neuro-ophthalmology focuses on brain and systemic abnormalities
Cranial Nerves. Cranial Nerve 1: Olfactory Nerve. Cranial Nerve 1: Olfactory Nerve. Cranial Nerve 2: Optic Nerve. Cranial Nerve 2: Optic Nerve
 Cranial Nerves Examination of Cranial Nerves and Palsies Drs Nathan Kerr and Shenton Chew 1 Olfactory On 2 Optic Old 3 Oculomotor Olympus 4 Trochlear Towering 5 Trigeminal Top 6 Abducens A 7 Facial Finn
Cranial Nerves Examination of Cranial Nerves and Palsies Drs Nathan Kerr and Shenton Chew 1 Olfactory On 2 Optic Old 3 Oculomotor Olympus 4 Trochlear Towering 5 Trigeminal Top 6 Abducens A 7 Facial Finn
MODERN CLINICAL OPTOMETRY BILLING & CODING THE MEDICAL EYE EXAMINATION. Definitions of Eye Examinations. Federal Government Definition
 MODERN CLINICAL OPTOMETRY BILLING & CODING THE MEDICAL EYE EXAMINATION Craig Thomas, O.D. 3900 West Wheatland Road Dallas, Texas 75237 972-780-7199 [email protected] Definitions of Eye Examinations Optometry
MODERN CLINICAL OPTOMETRY BILLING & CODING THE MEDICAL EYE EXAMINATION Craig Thomas, O.D. 3900 West Wheatland Road Dallas, Texas 75237 972-780-7199 [email protected] Definitions of Eye Examinations Optometry
Post-Concussion Syndrome
 Post-Concussion Syndrome Anatomy of the injury: The brain is a soft delicate structure encased in our skull, which protects it from external damage. It is suspended within the skull in a liquid called
Post-Concussion Syndrome Anatomy of the injury: The brain is a soft delicate structure encased in our skull, which protects it from external damage. It is suspended within the skull in a liquid called
Cerebral palsy, neonatal death and stillbirth rates Victoria, 1973-1999
 Cerebral Palsy: Aetiology, Associated Problems and Management Lecture for FRACP candidates July 2010 Definitions and prevalence Risk factors and aetiology Associated problems Management options Cerebral
Cerebral Palsy: Aetiology, Associated Problems and Management Lecture for FRACP candidates July 2010 Definitions and prevalence Risk factors and aetiology Associated problems Management options Cerebral
Guidelines for Obtaining & Reporting CE Credits
 Guidelines for Obtaining & Reporting CE Credits It is understood that continuing education (CE) is the cornerstone of maintenance of professional excellence. All COC members must therefore be able to demonstrate
Guidelines for Obtaining & Reporting CE Credits It is understood that continuing education (CE) is the cornerstone of maintenance of professional excellence. All COC members must therefore be able to demonstrate
CEREBRAL PALSY AND MENTAL RETARDATION DEFINITION
 CEREBRAL PALSY AND MENTAL RETARDATION DEFINITION It is a disorder of posture movement and tone due to a static encephalopathy acquired during brain growth in fetal life infancy or early childhood. Though
CEREBRAL PALSY AND MENTAL RETARDATION DEFINITION It is a disorder of posture movement and tone due to a static encephalopathy acquired during brain growth in fetal life infancy or early childhood. Though
WHAT IS CEREBRAL PALSY?
 WHAT IS CEREBRAL PALSY? Cerebral Palsy is a dysfunction in movement resulting from injury to or poor development of the brain prior to birth or in early childhood. Generally speaking, any injury or disease
WHAT IS CEREBRAL PALSY? Cerebral Palsy is a dysfunction in movement resulting from injury to or poor development of the brain prior to birth or in early childhood. Generally speaking, any injury or disease
THE EYES IN MARFAN SYNDROME
 THE EYES IN MARFAN SYNDROME Marfan syndrome and some related disorders can affect the eyes in many ways, causing dislocated lenses and other eye problems that can affect your sight. Except for dislocated
THE EYES IN MARFAN SYNDROME Marfan syndrome and some related disorders can affect the eyes in many ways, causing dislocated lenses and other eye problems that can affect your sight. Except for dislocated
Proposed Treatment for Vestibular Dysfunction in Dogs By Margaret Kraeling, DPT, CCRT
 Proposed Treatment for Vestibular Dysfunction in Dogs By Margaret Kraeling, DPT, CCRT Vestibular dysfunction in the dog can be a disturbing condition for owners, as well as somewhat confounding for the
Proposed Treatment for Vestibular Dysfunction in Dogs By Margaret Kraeling, DPT, CCRT Vestibular dysfunction in the dog can be a disturbing condition for owners, as well as somewhat confounding for the
Interval: [ ] Baseline [ ] 2 hours post treatment [ ] 24 hours post onset of symptoms ±20 minutes [ ] 7-10 days [ ] 3 months [ ] Other ( )
![Interval: [ ] Baseline [ ] 2 hours post treatment [ ] 24 hours post onset of symptoms ±20 minutes [ ] 7-10 days [ ] 3 months [ ] Other ( ) Interval: [ ] Baseline [ ] 2 hours post treatment [ ] 24 hours post onset of symptoms ±20 minutes [ ] 7-10 days [ ] 3 months [ ] Other ( )](/thumbs/29/13263233.jpg) Patient Identification. - - Pt. Date of Birth / / Hospital ( - ) Date of Exam / / Interval: [ ] Baseline [ ] 2 hours post treatment [ ] 24 hours post onset of symptoms ±20 minutes [ ] 7-10 days [ ] 3 months
Patient Identification. - - Pt. Date of Birth / / Hospital ( - ) Date of Exam / / Interval: [ ] Baseline [ ] 2 hours post treatment [ ] 24 hours post onset of symptoms ±20 minutes [ ] 7-10 days [ ] 3 months
Paediatric Bell s Palsy Paediatric Update November 2014
 Paediatric Bell s Palsy Paediatric Update November 2014 Richard Webster, Paediatric Neurologist Children s Hospital at Westmead Typical history Unilateral LMN facial weakness Acute onset over a day or
Paediatric Bell s Palsy Paediatric Update November 2014 Richard Webster, Paediatric Neurologist Children s Hospital at Westmead Typical history Unilateral LMN facial weakness Acute onset over a day or
Cerebral palsy can be classified according to the type of abnormal muscle tone or movement, and the distribution of these motor impairments.
 The Face of Cerebral Palsy Segment I Discovering Patterns What is Cerebral Palsy? Cerebral palsy (CP) is an umbrella term for a group of non-progressive but often changing motor impairment syndromes, which
The Face of Cerebral Palsy Segment I Discovering Patterns What is Cerebral Palsy? Cerebral palsy (CP) is an umbrella term for a group of non-progressive but often changing motor impairment syndromes, which
Guideline for the Management of Acute Peripheral Facial nerve palsy. Bells Palsy in Children
 Guideline for the Management of Acute Peripheral Facial nerve palsy Definition Bells Palsy in Children Bell palsy is an acute, idiopathic unilateral lower motor neurone facial nerve palsy that is not associated
Guideline for the Management of Acute Peripheral Facial nerve palsy Definition Bells Palsy in Children Bell palsy is an acute, idiopathic unilateral lower motor neurone facial nerve palsy that is not associated
How to test ocular movements in PSP Jan Kassubek
 How to test ocular movements in PSP Jan Kassubek Universitätsklinik für Neurologie, Ulm Bedside Screening: PSP initially slowing of vertical saccades slowing of downward saccades is considered the hallmark
How to test ocular movements in PSP Jan Kassubek Universitätsklinik für Neurologie, Ulm Bedside Screening: PSP initially slowing of vertical saccades slowing of downward saccades is considered the hallmark
BINOCULARITY FOLLOWING SURGICAL CORRECTION OF STRABISMUS IN ADULTS
 17-Mets 12/11/03 12:55 PM Page 201 BINOCULARITY FOLLOWING SURGICAL CORRECTION OF STRABISMUS IN ADULTS BY Marilyn B. Mets MD,* Cynthia Beauchamp MD, AND Betty Anne Haldi CO ABSTRACT Introduction: This is
17-Mets 12/11/03 12:55 PM Page 201 BINOCULARITY FOLLOWING SURGICAL CORRECTION OF STRABISMUS IN ADULTS BY Marilyn B. Mets MD,* Cynthia Beauchamp MD, AND Betty Anne Haldi CO ABSTRACT Introduction: This is
IMAGE ASSISTANT: OPHTHALMOLOGY
 IMAGE ASSISTANT: OPHTHALMOLOGY Summary: The Image Assistant has been developed to provide medical doctors with a software tool to search, display, edit and use medical illustrations of their own specialty,
IMAGE ASSISTANT: OPHTHALMOLOGY Summary: The Image Assistant has been developed to provide medical doctors with a software tool to search, display, edit and use medical illustrations of their own specialty,
CLINICAL NEUROPHYSIOLOGY
 CLINICAL NEUROPHYSIOLOGY Barry S. Oken, MD, Carter D. Wray MD Objectives: 1. Know the role of EMG/NCS in evaluating nerve and muscle function 2. Recognize common EEG findings and their significance 3.
CLINICAL NEUROPHYSIOLOGY Barry S. Oken, MD, Carter D. Wray MD Objectives: 1. Know the role of EMG/NCS in evaluating nerve and muscle function 2. Recognize common EEG findings and their significance 3.
Vision and Rehabilitation After Brain Trauma Eric Singman, MD, PhD, Health.mil
 Vision and Rehabilitation After Brain Trauma Eric Singman, MD, PhD, Health.mil This article was first accessed at Brainline.org. Vision Problems After Brain Injury Visual problems following brain trauma
Vision and Rehabilitation After Brain Trauma Eric Singman, MD, PhD, Health.mil This article was first accessed at Brainline.org. Vision Problems After Brain Injury Visual problems following brain trauma
Modern Techniques of Strabismus Surgery
 Optometry in Practice Vol 2 (2001) 79-88 Modern Techniques of Strabismus Surgery IB Marsh MB ChB FRCS FRCOphth Consultant Ophthalmologist, Walton Eye Unit, University Hospital Aintree, Liverpool, UK Accepted
Optometry in Practice Vol 2 (2001) 79-88 Modern Techniques of Strabismus Surgery IB Marsh MB ChB FRCS FRCOphth Consultant Ophthalmologist, Walton Eye Unit, University Hospital Aintree, Liverpool, UK Accepted
Corporate Medical Policy Reconstructive Eyelid Surgery and Brow Lift
 Corporate Medical Policy Reconstructive Eyelid Surgery and Brow Lift File Name: Origination: Last CAP Review: Next CAP Review: Last Review: reconstructive_eyelid_surgery_and_brow_lift 1/2000 9/2015 9/2016
Corporate Medical Policy Reconstructive Eyelid Surgery and Brow Lift File Name: Origination: Last CAP Review: Next CAP Review: Last Review: reconstructive_eyelid_surgery_and_brow_lift 1/2000 9/2015 9/2016
Clinical guidance for MRI referral
 MRI for cervical radiculopathy Referral by a medical practitioner (excluding a specialist or consultant physician) for a scan of spine for a patient 16 years or older for suspected: cervical radiculopathy
MRI for cervical radiculopathy Referral by a medical practitioner (excluding a specialist or consultant physician) for a scan of spine for a patient 16 years or older for suspected: cervical radiculopathy
2014 Fall CE@SCO. COURSE 3 Doc, I m Seeing Double. COPE Course 40262-NO
 2014 Fall CE@SCO COURSE 3 Doc, I m Seeing Double COPE Course 40262-NO SCO HOMECOMING / FALL CE WEEKEND OCTOBER 9-12, 2014 Levels of disease Dennis Mathews, O.D. Partner, Eye Specialty Group Associate
2014 Fall CE@SCO COURSE 3 Doc, I m Seeing Double COPE Course 40262-NO SCO HOMECOMING / FALL CE WEEKEND OCTOBER 9-12, 2014 Levels of disease Dennis Mathews, O.D. Partner, Eye Specialty Group Associate
How To Diagnose Stroke In Acute Vestibular Syndrome
 Danica Dummer, PT, DPT, University of Utah Abigail Reid, PT, DPT, Kessler Institute for Rehabilitation Online Journal Club-Article Review Article Citation Study Objective/Purpose (hypothesis) Study Design
Danica Dummer, PT, DPT, University of Utah Abigail Reid, PT, DPT, Kessler Institute for Rehabilitation Online Journal Club-Article Review Article Citation Study Objective/Purpose (hypothesis) Study Design
HEADACHES AND THE THIRD OCCIPITAL NERVE
 HEADACHES AND THE THIRD OCCIPITAL NERVE Edward Babigumira M.D. FAAPMR. Interventional Pain Management, Lincoln. B. Pain Clinic, Ltd. Diplomate ABPMR. Board Certified Pain Medicine No disclosures Disclosure
HEADACHES AND THE THIRD OCCIPITAL NERVE Edward Babigumira M.D. FAAPMR. Interventional Pain Management, Lincoln. B. Pain Clinic, Ltd. Diplomate ABPMR. Board Certified Pain Medicine No disclosures Disclosure
Management in the pre-hospital setting
 Management in the pre-hospital setting Inflammation of the joints Two main types: Osteoarthritis - cartilage loss from wear and tear Rheumatoid arthritis - autoimmune disorder Affects all age groups,
Management in the pre-hospital setting Inflammation of the joints Two main types: Osteoarthritis - cartilage loss from wear and tear Rheumatoid arthritis - autoimmune disorder Affects all age groups,
Cerebellum and Basal Ganglia
 Cerebellum and Basal Ganglia 1 Contents Cerebellum and Basal Ganglia... 1 Introduction... 3 A brief review of cerebellar anatomy... 4 Basic Circuit... 4 Parallel and climbing fiber input has a very different
Cerebellum and Basal Ganglia 1 Contents Cerebellum and Basal Ganglia... 1 Introduction... 3 A brief review of cerebellar anatomy... 4 Basic Circuit... 4 Parallel and climbing fiber input has a very different
Pediatric and Binocular Vision Examination and Billing Protocols
 Pediatric and Binocular Vision Examination and Billing Protocols Recommended Eye Examination Frequency for the Pediatric Patient... 3 Routine Comprehensive Pediatric Eye Examination... 4 Billing for Pediatric
Pediatric and Binocular Vision Examination and Billing Protocols Recommended Eye Examination Frequency for the Pediatric Patient... 3 Routine Comprehensive Pediatric Eye Examination... 4 Billing for Pediatric
eyelid position. Treatment of amblyopia is only effective in main- Albert W Biglan, MD
 RESULTS FOLLOWING TREATMENT OF THIRD CRANIAL NERVE PALSY IN CHILDREN* BY Linda A. Schumacher-Feero, MD (BY INVITATION), K W Yoo, MD, (BY INVITATION), Fernando Mendiola Solari, MD (BY INVITATION) AND Albert
RESULTS FOLLOWING TREATMENT OF THIRD CRANIAL NERVE PALSY IN CHILDREN* BY Linda A. Schumacher-Feero, MD (BY INVITATION), K W Yoo, MD, (BY INVITATION), Fernando Mendiola Solari, MD (BY INVITATION) AND Albert
22/02/2015. Possible causes. Decisions decisions decisions. Challenging eye and ear conditions at underwriting and claim stage Dr Maritha van der Walt
 Challenges Underwriting symptoms rather than conditions- tinnitus, vertigo, blurred vision Requests for narrower rather than broad exclusions and consequences Pre existing condition and unrelated claims
Challenges Underwriting symptoms rather than conditions- tinnitus, vertigo, blurred vision Requests for narrower rather than broad exclusions and consequences Pre existing condition and unrelated claims
Graduate Diploma in Optometry. Related modules Pre-requisites Satisfying requirements of second year BSc (Hons) Optometry examination board
 Posterior Eye & General Ophthalmology School and subject group Module code Module title Module type Module replaces (where appropriate) Life and Health Sciences / Optometry OP3PEG Posterior Eye & General
Posterior Eye & General Ophthalmology School and subject group Module code Module title Module type Module replaces (where appropriate) Life and Health Sciences / Optometry OP3PEG Posterior Eye & General
Assessment and Management of Visual Dysfunction Associated with Mild Traumatic Brain Injury. Clinical Recommendation Overview
 Assessment and Management of Visual Dysfunction Associated with Mild Traumatic Brain Injury Clinical Recommendation Overview Learning Objectives Describe an overview of visual dysfunction associated with
Assessment and Management of Visual Dysfunction Associated with Mild Traumatic Brain Injury Clinical Recommendation Overview Learning Objectives Describe an overview of visual dysfunction associated with
A Patient & Parent Guide to Strabismus Surgery. George R. Beauchamp, M.D. Paul R. Mitchell, M.D.
 A Patient & Parent Guide to Strabismus Surgery By George R. Beauchamp, M.D. Paul R. Mitchell, M.D. Table of Contents: Part I: Background Information 1. Basic Anatomy and Functions of the Extra-ocular Muscles
A Patient & Parent Guide to Strabismus Surgery By George R. Beauchamp, M.D. Paul R. Mitchell, M.D. Table of Contents: Part I: Background Information 1. Basic Anatomy and Functions of the Extra-ocular Muscles
Vocabulary & General Concepts of Brain Organization
 Vocabulary & General Concepts of Brain Organization Jeanette J. Norden, Ph.D. Professor Emerita Vanderbilt University School of Medicine Course Outline Lecture 1: Vocabulary & General Concepts of Brain
Vocabulary & General Concepts of Brain Organization Jeanette J. Norden, Ph.D. Professor Emerita Vanderbilt University School of Medicine Course Outline Lecture 1: Vocabulary & General Concepts of Brain
NEUROIMAGING in Parkinsonian Syndromes
 NEUROIMAGING in Parkinsonian Syndromes (Focus on Structural Techniques: CT and MRI) Dr. Roberto Cilia Parkinson Institute, ICP, Milan, Italy OUTLINE Primary Parkinsonism Idiopathic Parkinson s Disease
NEUROIMAGING in Parkinsonian Syndromes (Focus on Structural Techniques: CT and MRI) Dr. Roberto Cilia Parkinson Institute, ICP, Milan, Italy OUTLINE Primary Parkinsonism Idiopathic Parkinson s Disease
Complications of Strabismus Surgery
 Complications of Strabismus Surgery Tjeerd de Faber, Martha Tjon Rutger van Ruyven Alexis Damanakis Ondercorrectie Overcorrectie Wat vind je erger? 1 DELLEN Corneal dellen are small areas of thinning associated
Complications of Strabismus Surgery Tjeerd de Faber, Martha Tjon Rutger van Ruyven Alexis Damanakis Ondercorrectie Overcorrectie Wat vind je erger? 1 DELLEN Corneal dellen are small areas of thinning associated
Management of sixth nerve palsy different approaches
 100 Prună et al Management of sixth nerve palsy Management of sixth nerve palsy different approaches Violeta-Ioana Prună 1,4, Ligia Gabriela Tătăranu 3, V.M. Prună 2, Daniela Cioplean 4, R.M. Gorgan 3
100 Prună et al Management of sixth nerve palsy Management of sixth nerve palsy different approaches Violeta-Ioana Prună 1,4, Ligia Gabriela Tătăranu 3, V.M. Prună 2, Daniela Cioplean 4, R.M. Gorgan 3
Neurology Clerkship Learning Objectives
 Neurology Clerkship Learning Objectives Clinical skills Perform a neurological screening examination of the cranial nerves, motor system, reflexes, and sensory system under the observation and guidance
Neurology Clerkship Learning Objectives Clinical skills Perform a neurological screening examination of the cranial nerves, motor system, reflexes, and sensory system under the observation and guidance
Parts of the Brain. Chapter 1
 Chapter 1 Parts of the Brain Living creatures are made up of cells. Groups of cells, similar in appearance and with the same function, form tissue. The brain is a soft mass of supportive tissues and nerve
Chapter 1 Parts of the Brain Living creatures are made up of cells. Groups of cells, similar in appearance and with the same function, form tissue. The brain is a soft mass of supportive tissues and nerve
Neurofibromatosis Type 2: Information for Patients & Families by Mia MacCollin, M.D., Catherine Bove, R.N. Ed. & M. Priscilla Short, M.D.
 Neurofibromatosis Type 2: Information for Patients & Families by Mia MacCollin, M.D., Catherine Bove, R.N. Ed. & M. Priscilla Short, M.D. Neurofibromatosis Type 2 is a rare genetic disease, which causes
Neurofibromatosis Type 2: Information for Patients & Families by Mia MacCollin, M.D., Catherine Bove, R.N. Ed. & M. Priscilla Short, M.D. Neurofibromatosis Type 2 is a rare genetic disease, which causes
The Visual Cortex 0 http://www.tutis.ca/neuromd/index.htm 20 February 2013
 T he Visual Cortex 0 Chapter contents Contents Chapter 2... 0 T he Visual Cortex... 0 Chapter Contents... 1 Introduction... 2 Optic Chiasm... 2 Where do the eye's ganglion cells project to?... 3 To where
T he Visual Cortex 0 Chapter contents Contents Chapter 2... 0 T he Visual Cortex... 0 Chapter Contents... 1 Introduction... 2 Optic Chiasm... 2 Where do the eye's ganglion cells project to?... 3 To where
Ocular torsion and perceived vertical in oculomotor, trochlear and abducens nerve palsies
 Brain (1993), 116, 1095-1104 Ocular torsion and perceived vertical in oculomotor, trochlear and abducens nerve palsies Marianne Dieterich and Thomas Brandt Department of Neurology, Klinikum Grosshadern,
Brain (1993), 116, 1095-1104 Ocular torsion and perceived vertical in oculomotor, trochlear and abducens nerve palsies Marianne Dieterich and Thomas Brandt Department of Neurology, Klinikum Grosshadern,
Chiari Malformation: Symptoms
 Chiari Malformation: Symptoms SYMPTOMS DIAGNOSIS LIVING WITH CHIARI TREATMENT Rick Labuda, Executive Director [email protected] 724-940-0116 Disclaimer: This presentation is intended for informational
Chiari Malformation: Symptoms SYMPTOMS DIAGNOSIS LIVING WITH CHIARI TREATMENT Rick Labuda, Executive Director [email protected] 724-940-0116 Disclaimer: This presentation is intended for informational
Pediatric Eye Problems When do I refer? Hillary Onan, M.D. Eye Associates of Central Texas Dell Children s Medical Center of Central Texas
 Pediatric Eye Problems When do I refer? Hillary Onan, M.D. Eye Associates of Central Texas Dell Children s Medical Center of Central Texas Red Reflex Testing Check RR at every well child visit until child
Pediatric Eye Problems When do I refer? Hillary Onan, M.D. Eye Associates of Central Texas Dell Children s Medical Center of Central Texas Red Reflex Testing Check RR at every well child visit until child
Brain Tumor 101. Shanna Armstrong, RN Neuro Oncology Nurse Clinician UC Brain Tumor Center
 Brain Tumor 101 Shanna Armstrong, RN Neuro Oncology Nurse Clinician UC Brain Tumor Center Objectives Identify the different parts of the brain Describe how each part of the brain works Connect each part
Brain Tumor 101 Shanna Armstrong, RN Neuro Oncology Nurse Clinician UC Brain Tumor Center Objectives Identify the different parts of the brain Describe how each part of the brain works Connect each part

![Bell s Palsy ]]> <![CDATA[Bell's Palsy]]> Bell s Palsy ]]> <![CDATA[Bell's Palsy]]>](/thumbs/27/10105461.jpg)