A Tool To Visualize and Evaluate Data Obtained by Liquid Chromatography-Electrospray Ionization-Mass Spectrometry
|
|
|
- Winfred Green
- 10 years ago
- Views:
Transcription
1 Anal. Chem. 2004, 76, A Tool To Visualize and Evaluate Data Obtained by Liquid Chromatography-Electrospray Ionization-Mass Spectrometry Xiao-jun Li,* Patrick G. A. Pedrioli, Jimmy Eng, Dan Martin, Eugene C. Yi, Hookeun Lee, and Ruedi Aebersold The Institute for Systems Biology, 1441 North 34th Street, Seattle, Washington We present a software tool for visualizing data obtained from analyzing complex peptide mixtures by liquid chromatography (LC) electrospray ionization (ESI) mass spectrometry (MS). The data are represented as a twodimensional density plot. For experiments employing collision-induced dissociation (CID), links are embedded in the image to the CID spectra and the corresponding peptide sequences that are represented by the respective feature. The image provides an intuitive method to evaluate sample quality and the performance of an LC-ESI-MS system and can be used to optimize experimental conditions. Local patterns of the image can also be used to identify chemical contaminants and specific peptide features. Therefore, this software tool may have broad application in MS-based proteomics. * Corresponding author. [email protected]. Fax: (206) (1) Aebersold, R.; Mann, M. Nature 2003, 422, (2) Han, D. K.; Eng, J.; Zhou, H.; Aebersold, R. Nat. Biotechnol. 2001, 19, (3) Washburn, M. P.; Wolters, D.; Yates, J. R., 3rd. Nat. Biotechnol. 2001, 19, (4) Ong, S. E.; Blagoev, B.; Kratchmarova, I.; Kristensen, D. B.; Steen, H.; Pandey, A.; Mann, M. Mol. Cell. Proteomics 2002, 1, (5) Hansen, K. C.; Schmitt-Ulms, G.; Chalkley, R. J.; Hirsch, J.; Baldwin, M. A.; Burlingame, A. L. Mol. Cell. Proteomics 2003, 2, (6) Von Haller, P. D.; Yi, E.; Donohoe, S.; Vaughn, K.; Keller, A.; Nesvizhskii, A. I.; Eng, J.; Li, X. J.; Goodlett, D. R.; Aebersold, R.; Watts, J. D. Mol. Cell. Proteomics 2003, 2, (7) Link, A. J.; Eng, J.; Schieltz, D. M.; Carmack, E.; Mize, G. J.; Morris, D. R.; Garvik, B. M.; Yates, J. R., 3rd. Nat. Biotechnol. 1999, 17, (8) Gygi, S. P.; Rist, B.; Gerber, S. A.; Turecek, F.; Gelb, M. H.; Aebersold, R. Nat. Biotechnol. 1999, 17, Mass spectrometry (MS)-based proteomics has the capacity to identify and quantify hundreds to thousands of proteins in a single experiment and has become a powerful analytical tool in biological and medical research. 1-6 In a typical MS-based proteomics experiment, proteins are harvested from cells, tissues, or body fluids, dissolved and denatured in solution, and enzymatically digested into peptide mixtures. These peptide mixtures are then separated by multidimensional liquid chromatography (LC) and identified by methods of MS. 3,7 In cases in which the analytes are labeled with stable isotopes, accurate quantitative information can also be obtained in the same operation. 2,4,8 In a common implementation, a reversed-phase microcapillary LC system is coupled online with an electrospray ionization (ESI) tandem mass spectrometer. Peptides eluting from the LC column are immediately ionized by ESI and subjected to mass measurement (survey scan). The instrument, under computer control, then automatically selects specific precursor ion(s) for fragmentation in a collision cell, thus generating collision-induced dissociation (CID) or tandem mass (MS/MS) spectra. These CID spectra are then searched against sequence databases using software tools such as SEQUEST, 9 Mascot, 10 or ProteinProspector, 11 which leads to the identification of the peptide sequences. Spectra collected in survey scanning mode can be used to reconstruct peptide chromatograms using software tools such as XPRESS 2 or ASAPRatio, 12 from which the accurate quantities of isotopically labeled peptides can be derived. In such cases, proteins in the original samples are therefore both identified and quantified by assembling data obtained from peptides that represent each specific protein. Despite its success, this technology remains challenging and several factors are crucial for the success of LC-MS/MS experiments. These include the sample quality, the quality of the separation system used, and the performance of the mass spectrometer. Because the outcome of an LC-MS/MS experiment depends on many variables, it is almost impossible to optimize the system by systematically optimizing individual variables. Troubleshooting in the case of suboptimal performance is difficult and time-consuming. Visualization tools play an important role in evaluating the performance of many instruments and experiments. To monitor MS performance or the performance of an LC-MS experiment, mass spectrometer manufacturers provide several visualization tools to display individual MS spectra, the total ion current, baseline peaks, etc. While these tools provide detailed insight into a specific performance characteristic of an LC-MS/MS experiment, such as the quality of CID spectra, mass resolution, or chromatographic peak resolution, they make it difficult to extract an overview of the overall success of the experiment. Therefore, a visualization tool that is easy to use and offers transparent diagnostic features for common problems encountered in LC- MS/MS analyses would be useful. (9) Eng, J. K.; McCormack, A. L.; Yates, J. R., III. J. Am. Soc. Mass Spectrum. 1994, 5, (10) Perkins, D. N.; Pappin, D. J.; Creasy, D. M.; Cottrell, J. S. Electrophoresis 1999, 20, (11) Clauser, K. R.; Baker, P.; Burlingame, A. L. Anal. Chem. 1999, 71, (12) Li, X. J.; Zhang, H.; Ranish, J. A.; Aebersold, R. Anal. Chem 2003, 75, Analytical Chemistry, Vol. 76, No. 13, July 1, /ac035375s CCC: $ American Chemical Society Published on Web 05/18/2004
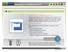
2 We present here a visualization tool, called Pep3D, which displays LC-ESI-MS data in a two-dimensional (retention time (RT) vs m/z) density plot. A Pep3D image graphically resembles a twodimensional (2D) polyacrylamide gel electrophoresis (PAGE) image, 13 in that the darkness of individual features displayed in the 2D array reflects the intensity of the detected signals. While similar images have been reported in the MS literature using the Xcalibur software (ThermoFinnigan) or other tools, a number of unique features set the Pep3D image apart from those tools: (1) Images are created very quickly. (2) Pep3D allows users to zoom into any interesting range of time and m/z. (3) Pep3D allows users to display those precursor ions selected for CID, all peptides successfully identified in the MS analysis, or both, with boxes whose color varies across a gradient from green to red to display the corresponding identification scores. (4) All CID boxes and peptide boxes have embedded common gateway interface (CGI) links allowing immediate display of the corresponding CID spectrum and its peptide identification. Pep3D can therefore be used to evaluate LC-ESI-MS system performance and identify features of the separated peptides. EXPERIMENTAL SECTION Data presented here were collected from various projects in our laboratory. Experimental protocols used to generate these data have been described in other publications. 2,6,8 Protocols at were used to label samples with ICAT reagent. 8 Peptide samples were analyzed on an LCQ Classic or an LCQ Deca (ThermoFinnigan) ion trap mass spectrometer or on an Q-Tof Ultima (Waters) quadrupole timeof-flight mass spectrometer. CID spectra were searched against corresponding databases using SEQUEST. 9 Peptide identifications were verified with PeptideProphet 18 and organized by INTERACT. 2 RESULTS Pep3D Algorithm. The Pep3D software tool is written in C, and the current version runs on a Linux operating system. Pep3D images are generated on the fly in PNG file format using a GD graphics library ( It normally takes a few seconds on a Pentium III personal computer to generate a Pep3D image for a 3-h LC-ESI-MS run. The Pep3D software tool is capable of displaying LC-ESI-MS data from all types of mass spectrometers for which the data can be represented in the mzxml data format. 19 The mzxml file format was developed recently to represent MS data from all types of mass spectrometers in a standard XML file format. A detailed description of mzxml (13) Rabilloud, T. Proteomics 2002, 2, (14) Binz, P. A.; Muller, M.; Walther, D.; Bienvenut, W. V.; Gras, R.; Hoogland, C.; Bouchet, G.; Gasteiger, E.; Fabbretti, R.; Gay, S.; Palagi, P.; Wilkins, M. R.; Rouge, V.; Tonella, L.; Paesano, S.; Rossellat, G.; Karmime, A.; Bairoch, A.; Sanchez, J. C.; Appel, R. D.; Hochstrasser, D. F. Anal. Chem 1999, 71, (15) Berger, S. J.; Lee, S. W.; Anderson, G. A.; Pasa-Tolic, L.; Tolic, N.; Shen, Y.; Zhao, R.; Smith, R. D. Anal. Chem 2002, 74, (16) Palmblad, M.; Ramstrom, M.; Markides, K. E.; Hakansson, P.; Bergquist, J. Anal. Chem 2002, 74, (17) Griffin, T. J.; Lock, C. M.; Li, X. J.; Patel, A.; Chervetsova, I.; Lee, H.; Wright, M. E.; Ranish, J. A.; Chen, S. S.; Aebersold, R. Anal. Chem 2003, 75, (18) Keller, A.; Nesvizhskii, A. I.; Kolker, E.; Aebersold, R. Anal. Chem. 2002, 74, (19) Pedrioli, P.; Eng, J.; Hubley, R.; Pratt, B.; Nilsson, E.; Aebersold, R., submitted. Figure 1. Screen capture image of Pep3D web interface. schema can be found at software.php. Currently, data generated from LCQ (ThermoFinnigan) ion trap, Q-Tof Ultima (Waters), and QSTAR (ABI/Sciex) mass spectrometers can be represented in the mzxml file format and displayed by Pep3D images. Embedded links to CID spectra or peptide identities are developed by using server side graphics. CID spectra are plotted by using the gnuplot software tool ( The Pep3D source code and compiled executable are available under an open source license at The Pep3D software tool has a web interface, developed in CGI programming, for users to specify the contents of Pep3D images. A screen capture image of the interface was plotted in Figure 1. The options allowed by the software tool include the following. (1) Input file: INTERACT data file interact-data.htm or mzxml files are accepted. Data in interact-data.htm can be filtered by using INTERACT filtering tools. 2 Multiple mzxml files can be selected simultaneously. (2) M/Z range: With the option Full, the whole m/z range of an experiment is displayed; with the option Selected, only the m/z range specified by >) and <) is displayed. Empty input in >) or <) is replaced by the lower or upper boundary of the whole m/z range. The m/z range is divided into pixels of size specified by Grid. A pixel is plotted as a rectangular of width specified by Image grid. (3) Elution time range (in min): This option is similar to the m/z range option. (4) Intensity range: With the option unit in background, the software automatically estimates the average intensity of background ions within the image range. The Intensity range is then specified in unit of this average intensity by >) and <). With the option Absolute, Intensity range is specified directly by >) and <). Empty input in >) or <) is replaced by the lowest or highest ion intensity within the image range. (5) Display peptides: With the option None, only raw LC-ESI-MS data are displayed; with the option CID, all precursor ions are displayed along with raw data; with the Analytical Chemistry, Vol. 76, No. 13, July 1,
3 Figure 2. (A) A Pep3D image displaying raw LC-ESI-MS data. The whole image is discretized into identical rectangular pixels. The shading of each pixel is determined by the signal intensity in the respective pixel. The intensity scale is denoted to the right of the image. (B) A Pep3D image displaying LC-ESI-MS data and highlighting all precursor ions selected for CID. Precursor ions are marked with blue boxes with centers specified by retention times and m/z values. (C) A Pep3D image displaying LC-ESI-MS data and highlighting the positively identified peptides. Peptides are marked with boxes in the same way as precursor ions in (B) except for their color. The hue of a peptide box on a gradient of green to red is determined by the corresponding PeptideProphet probability and displayed below the image. (D) A Pep3D image displaying LC-ESI- MS data, all precursor ions selected for CID, and all identified peptides. Precursor ions and peptides are marked in the same way as in (B) and (C). option Peptide, all peptides identified with identification scores within a range specified by Score >) and <) are displayed along with raw data. Currently SEQUEST Xcorr scores 9 and PeptideProphet probabilities 18 are accepted as identification scores. The type of scores can be easily extended to include scores generated by other sequence searching engines; the option All 3858 Analytical Chemistry, Vol. 76, No. 13, July 1, 2004
4 Figure 3. Screen capture image of a CGI-generated web page displaying peptide information including the scan number, the m/z value, and the retention time of the precursor ion and the corresponding CID spectrum. Information on peptide identification was copied from the INTERACT data file. If the web page is for an unidentified precursor ion, information on peptide identification will be replaced by None. is equivalent to the combination of options CID and Peptide. CID and/or peptide boxes are plotted in a size specified by Image Grid. Empty input in Score >) or <) is replaced by the lowest or highest score with the data. Peptides are displayed only if the input file is interact-data.htm. (6) Mapping function: With the option Linear, the darkness of a pixel is linear to its ion intensity; with the option Log (base 10), the darkness of a pixel is linear to the logarithm (base 10) of its ion intensity. (7) Image type: With the option Full, pixels with ion intensity greater than the upper boundary of Intensity range are plotted in completely dark while pixels with ion intensity less than the lower boundary of Intensity range are plotted in completely white; with the option Section, pixels with ion intensity outside Intensity range are plotted in completely white. (8) Save option: In addition to viewing Pep3D images on a screen, one may also save them in an html file. By varying these options, one can zoom in any time or m/z range of a Pep3D image, visualize only pixels within a certain intensity range, change scales between ion intensity and pixel darkness, and display peptides of various identification scores, etc. These options add flexibility and power to the visualization tool. Pep3D Features. The Pep3D software tool can display LC- ESI-MS experimental data in four different types of images; an example of each type is shown in Figure 2. In Figure 2A, raw LC-ESI-MS data are plotted as a density plot with the RT in minutes on the abscissa and the m/z value of the detected signals on the ordinate. The whole image is discretized into rectangular pixels of identical size with time width and m/z height specified by the user. After discretization, ion signals in all MS spectra are assigned to individual pixels. All ion intensities within a pixel are summed together and averaged by the number of MS spectra collected over the duration of the pixel; the result is the ion intensity of the pixel. If a pixel has no ion signals, its ion intensity is set to zero. In the Pep3D image, the ion intensity of each pixel is specified by the shade of the pixel over a gray scale. The relationship between the ion intensity of a pixel and its darkness is denoted along the image. The intensity scale is specified by the user. The Pep3D image visualizes LC-ESI-MS ion signals of the whole experiment in a very transparent manner. In Figure 2B, all precursor ions selected for CID are highlighted with blue boxes on the density plot in Figure 2A. The center of each CID box is specified by the RT and the m/z value of the corresponding precursor ion. There is a CGI link embedded behind each CID box. When a CID box is clicked, a CGI program opens a new window that displays the scan number, the m/z value, and the RT of the corresponding precursor ion, as well as the CID spectrum with its major fragment ions labeled with the respective m/z values (see Figure 3 for an example). This feature of the Pep3D image allows users to easily connect individual Analytical Chemistry, Vol. 76, No. 13, July 1,
5 features in the Pep3D image with corresponding CID spectra. It also provides users with a method to discover and interpret certain patterns in the Pep3D image and to survey the performance of automated precursor ion selection. In the data displayed in Figure 2B, there were 1196 precursor ions selected for CID, a fact that is automatically reported by the software tool. It is apparent from the figure that the majority of high-intensity MS signals were selected for CID during this experiment while the majority of lowintensity MS signals were not selected. As demonstrated in Supporting Information, this information can be used to evaluate sample loading and sample separation. In Figure 2C, all the peptides identified in the experiment and assigned a PeptideProphet 18 probability of no less than 0.5 are highlighted with color boxes. The color varies across a gradient from green to red and is determined by the corresponding PeptideProphet probability. Except for their color, these boxes are plotted in the same fashion as those boxes indicating CID attempts (Figure 2B) and they also include embedded CGI links behind them. By clicking on a peptide box, the corresponding peptide identity entry in the INTERACT data file is displayed, along with all information for that precursor ion. Through the peptide identity entry within the link, the corresponding protein identity, peptide identity, peptide mass, peptide charge state, etc., are all displayed (see Figure 3 for an example). The display in Figure 2C indicates the distribution and the number of the peptides that were successfully identified in the experiment. Among 1196 precursor ions selected for CID, 221 were successfully identified. The number of identified peptides is automatically reported. For unknown reasons, a large proportion of highintensity ions were not assigned a sequence with a high degree of confidence, a fact that is immediately apparent from the Pep3D image. This observation is rather common in most LC-MS/MS analyses, and possible explanations include peptide modifications, database deficiencies, and difficulties of generating quality CID spectra from the precursor ions. Instead of PeptideProphet probability, SEQUEST Xcorr score can also be used for displaying identified peptides. Figure 2D combines the features described in Figure 2B and C, where all precursor ions selected for CID and all peptides assigned a PeptideProphet probability of no less than 0.5 are highlighted with color boxes. This image provides an entry point to investigate why certain CID spectra yielded identifications while others failed to do so. For example, one may reanalyze unidentified high-quality CID spectra by de novo sequencing or database searching with additional peptide modifications allowed. As demonstrated in Supporting Information, this display also makes it easy to identify chemical contaminants within the sample. Figure 2D indicates 1196 CID attempts and 221 peptide identifications; 18.5% of all CID spectra were positively identified. The likelihood of a CID attempt producing positive peptide identification varied significantly across the plot. By comparing this percentage in different areas, a researcher may identify high-percentage areas and adjust LC-MS settings accordingly. This type of information (20) Fenn, J. B.; Mann, M.; Meng, C. K.; Wong, S. F.; Whitehouse, C. M. Science 1989, 246, can be valuable for optimizing LC-MS settings to improve the quality of CID identifications. DISCUSSION A Pep3D image graphically resembles a 2D-PAGE image. However, most spots in a Pep3D image represent peptides, not proteins as in a 2D-PAGE image. A small percentage of spots in a Pep3D image may belong to chemical contaminants such as solvent molecules, lipids, or polymers. Since a peptide may be ionized into more than one charge state during ESI, 20 more than one spot may represent the same peptide. The darkness of a peptide spot is not indicative of its absolute concentration because different peptides have different ionization efficiencies during ESI. An exception to this statement is when the peptides are identical yet in different isotopic forms. The relative darkness of a particular peptide spot across different LC-MS samples may reflect the relative abundance of the peptide in the corresponding samples if appropriate abundance normalization can be carried out. We describe here a visualization tool, Pep3D, to display LC- ESI-MS data. The tool is transparent and easy to interpret and provides informative, comprehensive, and sophisticated insights into the performance of an LC-MS system and the analytes detected. As illustrated in Supporting Information, a Pep3D image can be used to evaluate sample quality, diagnose system performance, and discover peptide features. Pep3D images can also be used to check the reproducibility and consistency of different sample analyses. Furthermore, by comparing peptide spots across parallel samples, one can potentially use Pep3D images to identify unique peptide spots capable of distinguishing different samples, a feature that has potential usage for biomarker discovery. One can also use the tool to collect unidentified high-quality CID spectra and subject them to further analysis, from which some posttranslationally modified peptides may be identified. Owing to these unique features, the Pep3D software tool may have a broad application in LC-MS-based proteomics. The tool is freely available via an open source license and is compatible with any type of mass spectrometer for which the data can be translated into the mzxml data format. A new version of the software tool is planned for Windows operating system and will be available to the public as soon as it is finished. ACKNOWLEDGMENT This project has been funded in part with federal funds from the National Heart, Lung, and Blood Institute, National Institutes of Health, under Contract N01-HV and from National Institutes of Health Grant R33 CA The ISB is the recipient of a generous gift from Merck and Co. D.M. was supported by NIH Grant CA SUPPORTING INFORMATION AVAILABLE Examples illustrating the application of Pep3D software tool. This material is available free of charge via the Internet at pubs.acs.org. Received for review November 20, Accepted April 1, AC035375S 3860 Analytical Chemistry, Vol. 76, No. 13, July 1, 2004
泛 用 蛋 白 質 體 學 之 質 譜 儀 資 料 分 析 平 台 的 建 立 與 應 用 Universal Mass Spectrometry Data Analysis Platform for Quantitative and Qualitative Proteomics
 泛 用 蛋 白 質 體 學 之 質 譜 儀 資 料 分 析 平 台 的 建 立 與 應 用 Universal Mass Spectrometry Data Analysis Platform for Quantitative and Qualitative Proteomics 2014 Training Course Wei-Hung Chang ( 張 瑋 宏 ) ABRC, Academia
泛 用 蛋 白 質 體 學 之 質 譜 儀 資 料 分 析 平 台 的 建 立 與 應 用 Universal Mass Spectrometry Data Analysis Platform for Quantitative and Qualitative Proteomics 2014 Training Course Wei-Hung Chang ( 張 瑋 宏 ) ABRC, Academia
Aiping Lu. Key Laboratory of System Biology Chinese Academic Society [email protected]
 Aiping Lu Key Laboratory of System Biology Chinese Academic Society [email protected] Proteome and Proteomics PROTEin complement expressed by genome Marc Wilkins Electrophoresis. 1995. 16(7):1090-4. proteomics
Aiping Lu Key Laboratory of System Biology Chinese Academic Society [email protected] Proteome and Proteomics PROTEin complement expressed by genome Marc Wilkins Electrophoresis. 1995. 16(7):1090-4. proteomics
The Scheduled MRM Algorithm Enables Intelligent Use of Retention Time During Multiple Reaction Monitoring
 The Scheduled MRM Algorithm Enables Intelligent Use of Retention Time During Multiple Reaction Monitoring Delivering up to 2500 MRM Transitions per LC Run Christie Hunter 1, Brigitte Simons 2 1 AB SCIEX,
The Scheduled MRM Algorithm Enables Intelligent Use of Retention Time During Multiple Reaction Monitoring Delivering up to 2500 MRM Transitions per LC Run Christie Hunter 1, Brigitte Simons 2 1 AB SCIEX,
Application Note # LCMS-81 Introducing New Proteomics Acquisiton Strategies with the compact Towards the Universal Proteomics Acquisition Method
 Application Note # LCMS-81 Introducing New Proteomics Acquisiton Strategies with the compact Towards the Universal Proteomics Acquisition Method Introduction During the last decade, the complexity of samples
Application Note # LCMS-81 Introducing New Proteomics Acquisiton Strategies with the compact Towards the Universal Proteomics Acquisition Method Introduction During the last decade, the complexity of samples
Introduction to Proteomics 1.0
 Introduction to Proteomics 1.0 CMSP Workshop Tim Griffin Associate Professor, BMBB Faculty Director, CMSP Objectives Why are we here? For participants: Learn basics of MS-based proteomics Learn what s
Introduction to Proteomics 1.0 CMSP Workshop Tim Griffin Associate Professor, BMBB Faculty Director, CMSP Objectives Why are we here? For participants: Learn basics of MS-based proteomics Learn what s
Mass Spectrometry Based Proteomics
 Mass Spectrometry Based Proteomics Proteomics Shared Research Oregon Health & Science University Portland, Oregon This document is designed to give a brief overview of Mass Spectrometry Based Proteomics
Mass Spectrometry Based Proteomics Proteomics Shared Research Oregon Health & Science University Portland, Oregon This document is designed to give a brief overview of Mass Spectrometry Based Proteomics
Increasing the Multiplexing of High Resolution Targeted Peptide Quantification Assays
 Increasing the Multiplexing of High Resolution Targeted Peptide Quantification Assays Scheduled MRM HR Workflow on the TripleTOF Systems Jenny Albanese, Christie Hunter AB SCIEX, USA Targeted quantitative
Increasing the Multiplexing of High Resolution Targeted Peptide Quantification Assays Scheduled MRM HR Workflow on the TripleTOF Systems Jenny Albanese, Christie Hunter AB SCIEX, USA Targeted quantitative
ProteinScape. Innovation with Integrity. Proteomics Data Analysis & Management. Mass Spectrometry
 ProteinScape Proteomics Data Analysis & Management Innovation with Integrity Mass Spectrometry ProteinScape a Virtual Environment for Successful Proteomics To overcome the growing complexity of proteomics
ProteinScape Proteomics Data Analysis & Management Innovation with Integrity Mass Spectrometry ProteinScape a Virtual Environment for Successful Proteomics To overcome the growing complexity of proteomics
MultiQuant Software 2.0 for Targeted Protein / Peptide Quantification
 MultiQuant Software 2.0 for Targeted Protein / Peptide Quantification Gold Standard for Quantitative Data Processing Because of the sensitivity, selectivity, speed and throughput at which MRM assays can
MultiQuant Software 2.0 for Targeted Protein / Peptide Quantification Gold Standard for Quantitative Data Processing Because of the sensitivity, selectivity, speed and throughput at which MRM assays can
In-Depth Qualitative Analysis of Complex Proteomic Samples Using High Quality MS/MS at Fast Acquisition Rates
 In-Depth Qualitative Analysis of Complex Proteomic Samples Using High Quality MS/MS at Fast Acquisition Rates Using the Explore Workflow on the AB SCIEX TripleTOF 5600 System A major challenge in proteomics
In-Depth Qualitative Analysis of Complex Proteomic Samples Using High Quality MS/MS at Fast Acquisition Rates Using the Explore Workflow on the AB SCIEX TripleTOF 5600 System A major challenge in proteomics
Pep-Miner: A Novel Technology for Mass Spectrometry-Based Proteomics
 Pep-Miner: A Novel Technology for Mass Spectrometry-Based Proteomics Ilan Beer Haifa Research Lab Dec 10, 2002 Pep-Miner s Location in the Life Sciences World The post-genome era - the age of proteome
Pep-Miner: A Novel Technology for Mass Spectrometry-Based Proteomics Ilan Beer Haifa Research Lab Dec 10, 2002 Pep-Miner s Location in the Life Sciences World The post-genome era - the age of proteome
Global and Discovery Proteomics Lecture Agenda
 Global and Discovery Proteomics Christine A. Jelinek, Ph.D. Johns Hopkins University School of Medicine Department of Pharmacology and Molecular Sciences Middle Atlantic Mass Spectrometry Laboratory Global
Global and Discovery Proteomics Christine A. Jelinek, Ph.D. Johns Hopkins University School of Medicine Department of Pharmacology and Molecular Sciences Middle Atlantic Mass Spectrometry Laboratory Global
AB SCIEX TOF/TOF 4800 PLUS SYSTEM. Cost effective flexibility for your core needs
 AB SCIEX TOF/TOF 4800 PLUS SYSTEM Cost effective flexibility for your core needs AB SCIEX TOF/TOF 4800 PLUS SYSTEM It s just what you expect from the industry leader. The AB SCIEX 4800 Plus MALDI TOF/TOF
AB SCIEX TOF/TOF 4800 PLUS SYSTEM Cost effective flexibility for your core needs AB SCIEX TOF/TOF 4800 PLUS SYSTEM It s just what you expect from the industry leader. The AB SCIEX 4800 Plus MALDI TOF/TOF
Quantitative proteomics background
 Proteomics data analysis seminar Quantitative proteomics and transcriptomics of anaerobic and aerobic yeast cultures reveals post transcriptional regulation of key cellular processes de Groot, M., Daran
Proteomics data analysis seminar Quantitative proteomics and transcriptomics of anaerobic and aerobic yeast cultures reveals post transcriptional regulation of key cellular processes de Groot, M., Daran
LC-MS/MS for Chromatographers
 LC-MS/MS for Chromatographers An introduction to the use of LC-MS/MS, with an emphasis on the analysis of drugs in biological matrices LC-MS/MS for Chromatographers An introduction to the use of LC-MS/MS,
LC-MS/MS for Chromatographers An introduction to the use of LC-MS/MS, with an emphasis on the analysis of drugs in biological matrices LC-MS/MS for Chromatographers An introduction to the use of LC-MS/MS,
MRMPilot Software: Accelerating MRM Assay Development for Targeted Quantitative Proteomics
 MRMPilot Software: Accelerating MRM Assay Development for Targeted Quantitative Proteomics With Unique QTRAP and TripleTOF 5600 System Technology Targeted peptide quantification is a rapidly growing application
MRMPilot Software: Accelerating MRM Assay Development for Targeted Quantitative Proteomics With Unique QTRAP and TripleTOF 5600 System Technology Targeted peptide quantification is a rapidly growing application
Effects of Intelligent Data Acquisition and Fast Laser Speed on Analysis of Complex Protein Digests
 Effects of Intelligent Data Acquisition and Fast Laser Speed on Analysis of Complex Protein Digests AB SCIEX TOF/TOF 5800 System with DynamicExit Algorithm and ProteinPilot Software for Robust Protein
Effects of Intelligent Data Acquisition and Fast Laser Speed on Analysis of Complex Protein Digests AB SCIEX TOF/TOF 5800 System with DynamicExit Algorithm and ProteinPilot Software for Robust Protein
MarkerView Software 1.2.1 for Metabolomic and Biomarker Profiling Analysis
 MarkerView Software 1.2.1 for Metabolomic and Biomarker Profiling Analysis Overview MarkerView software is a novel program designed for metabolomics applications and biomarker profiling workflows 1. Using
MarkerView Software 1.2.1 for Metabolomic and Biomarker Profiling Analysis Overview MarkerView software is a novel program designed for metabolomics applications and biomarker profiling workflows 1. Using
GENERAL UNKNOWN SCREENING FOR DRUGS IN BIOLOGICAL SAMPLES BY LC/MS Luc Humbert1, Michel Lhermitte 1, Frederic Grisel 2 1
 GENERAL UNKNOWN SCREENING FOR DRUGS IN BIOLOGICAL SAMPLES BY LC/MS Luc Humbert, Michel Lhermitte, Frederic Grisel Laboratoire de Toxicologie & Génopathologie, CHRU Lille, France Waters Corporation, Guyancourt,
GENERAL UNKNOWN SCREENING FOR DRUGS IN BIOLOGICAL SAMPLES BY LC/MS Luc Humbert, Michel Lhermitte, Frederic Grisel Laboratoire de Toxicologie & Génopathologie, CHRU Lille, France Waters Corporation, Guyancourt,
Learning Objectives:
 Proteomics Methodology for LC-MS/MS Data Analysis Methodology for LC-MS/MS Data Analysis Peptide mass spectrum data of individual protein obtained from LC-MS/MS has to be analyzed for identification of
Proteomics Methodology for LC-MS/MS Data Analysis Methodology for LC-MS/MS Data Analysis Peptide mass spectrum data of individual protein obtained from LC-MS/MS has to be analyzed for identification of
Accurate Mass Screening Workflows for the Analysis of Novel Psychoactive Substances
 Accurate Mass Screening Workflows for the Analysis of Novel Psychoactive Substances TripleTOF 5600 + LC/MS/MS System with MasterView Software Adrian M. Taylor AB Sciex Concord, Ontario (Canada) Overview
Accurate Mass Screening Workflows for the Analysis of Novel Psychoactive Substances TripleTOF 5600 + LC/MS/MS System with MasterView Software Adrian M. Taylor AB Sciex Concord, Ontario (Canada) Overview
Bruker ToxScreener TM. Innovation with Integrity. A Comprehensive Screening Solution for Forensic Toxicology UHR-TOF MS
 Bruker ToxScreener TM A Comprehensive Screening Solution for Forensic Toxicology Innovation with Integrity UHR-TOF MS ToxScreener - Get the Complete Picture Forensic laboratories are frequently required
Bruker ToxScreener TM A Comprehensive Screening Solution for Forensic Toxicology Innovation with Integrity UHR-TOF MS ToxScreener - Get the Complete Picture Forensic laboratories are frequently required
Application Note # LCMS-62 Walk-Up Ion Trap Mass Spectrometer System in a Multi-User Environment Using Compass OpenAccess Software
 Application Note # LCMS-62 Walk-Up Ion Trap Mass Spectrometer System in a Multi-User Environment Using Compass OpenAccess Software Abstract Presented here is a case study of a walk-up liquid chromatography
Application Note # LCMS-62 Walk-Up Ion Trap Mass Spectrometer System in a Multi-User Environment Using Compass OpenAccess Software Abstract Presented here is a case study of a walk-up liquid chromatography
Using Natural Products Application Solution with UNIFI for the Identification of Chemical Ingredients of Green Tea Extract
 Using Natural Products Application Solution with UNIFI for the Identification of Chemical Ingredients of Green Tea Extract Lirui Qiao, 1 Rob Lewis, 2 Alex Hooper, 2 James Morphet, 2 Xiaojie Tan, 1 Kate
Using Natural Products Application Solution with UNIFI for the Identification of Chemical Ingredients of Green Tea Extract Lirui Qiao, 1 Rob Lewis, 2 Alex Hooper, 2 James Morphet, 2 Xiaojie Tan, 1 Kate
Overview. Triple quadrupole (MS/MS) systems provide in comparison to single quadrupole (MS) systems: Introduction
 Advantages of Using Triple Quadrupole over Single Quadrupole Mass Spectrometry to Quantify and Identify the Presence of Pesticides in Water and Soil Samples André Schreiber AB SCIEX Concord, Ontario (Canada)
Advantages of Using Triple Quadrupole over Single Quadrupole Mass Spectrometry to Quantify and Identify the Presence of Pesticides in Water and Soil Samples André Schreiber AB SCIEX Concord, Ontario (Canada)
Challenges and Opportunities in Proteomics Data Analysis*
 Perspectives Challenges and Opportunities in Proteomics Data Analysis* Bruno Domon and Ruedi Aebersold Accurate, consistent, and transparent data processing and analysis are integral and critical parts
Perspectives Challenges and Opportunities in Proteomics Data Analysis* Bruno Domon and Ruedi Aebersold Accurate, consistent, and transparent data processing and analysis are integral and critical parts
Chapter 14. Modeling Experimental Design for Proteomics. Jan Eriksson and David Fenyö. Abstract. 1. Introduction
 Chapter Modeling Experimental Design for Proteomics Jan Eriksson and David Fenyö Abstract The complexity of proteomes makes good experimental design essential for their successful investigation. Here,
Chapter Modeling Experimental Design for Proteomics Jan Eriksson and David Fenyö Abstract The complexity of proteomes makes good experimental design essential for their successful investigation. Here,
Proteomic data analysis for Orbitrap datasets using Resources available at MSI. September 28 th 2011 Pratik Jagtap
 Proteomic data analysis for Orbitrap datasets using Resources available at MSI. September 28 th 2011 Pratik Jagtap The Minnesota http://www.mass.msi.umn.edu/ Proteomics workflow Trypsin Protein Peptides
Proteomic data analysis for Orbitrap datasets using Resources available at MSI. September 28 th 2011 Pratik Jagtap The Minnesota http://www.mass.msi.umn.edu/ Proteomics workflow Trypsin Protein Peptides
Simultaneous qualitative and quantitative analysis using the Agilent 6540 Accurate-Mass Q-TOF
 Simultaneous qualitative and quantitative analysis using the Agilent 654 Accurate-Mass Q-TOF Technical Overview Authors Pat Perkins Anabel Fandino Lester Taylor Agilent Technologies, Inc. Santa Clara,
Simultaneous qualitative and quantitative analysis using the Agilent 654 Accurate-Mass Q-TOF Technical Overview Authors Pat Perkins Anabel Fandino Lester Taylor Agilent Technologies, Inc. Santa Clara,
Pesticide Analysis by Mass Spectrometry
 Pesticide Analysis by Mass Spectrometry Purpose: The purpose of this assignment is to introduce concepts of mass spectrometry (MS) as they pertain to the qualitative and quantitative analysis of organochlorine
Pesticide Analysis by Mass Spectrometry Purpose: The purpose of this assignment is to introduce concepts of mass spectrometry (MS) as they pertain to the qualitative and quantitative analysis of organochlorine
Session 1. Course Presentation: Mass spectrometry-based proteomics for molecular and cellular biologists
 Program Overview Session 1. Course Presentation: Mass spectrometry-based proteomics for molecular and cellular biologists Session 2. Principles of Mass Spectrometry Session 3. Mass spectrometry based proteomics
Program Overview Session 1. Course Presentation: Mass spectrometry-based proteomics for molecular and cellular biologists Session 2. Principles of Mass Spectrometry Session 3. Mass spectrometry based proteomics
Mass Spectrometry Signal Calibration for Protein Quantitation
 Cambridge Isotope Laboratories, Inc. www.isotope.com Proteomics Mass Spectrometry Signal Calibration for Protein Quantitation Michael J. MacCoss, PhD Associate Professor of Genome Sciences University of
Cambridge Isotope Laboratories, Inc. www.isotope.com Proteomics Mass Spectrometry Signal Calibration for Protein Quantitation Michael J. MacCoss, PhD Associate Professor of Genome Sciences University of
Analysis of the Vitamin B Complex in Infant Formula Samples by LC-MS/MS
 Analysis of the Vitamin B Complex in Infant Formula Samples by LC-MS/MS Stephen Lock 1 and Matthew Noestheden 2 1 AB SCIEX Warrington, Cheshire (UK), 2 AB SCIEX Concord, Ontario (Canada) Overview A rapid,
Analysis of the Vitamin B Complex in Infant Formula Samples by LC-MS/MS Stephen Lock 1 and Matthew Noestheden 2 1 AB SCIEX Warrington, Cheshire (UK), 2 AB SCIEX Concord, Ontario (Canada) Overview A rapid,
Tutorial for Proteomics Data Submission. Katalin F. Medzihradszky Robert J. Chalkley UCSF
 Tutorial for Proteomics Data Submission Katalin F. Medzihradszky Robert J. Chalkley UCSF Why Have Guidelines? Large-scale proteomics studies create huge amounts of data. It is impossible/impractical to
Tutorial for Proteomics Data Submission Katalin F. Medzihradszky Robert J. Chalkley UCSF Why Have Guidelines? Large-scale proteomics studies create huge amounts of data. It is impossible/impractical to
Introduction to mass spectrometry (MS) based proteomics and metabolomics
 Introduction to mass spectrometry (MS) based proteomics and metabolomics Tianwei Yu Department of Biostatistics and Bioinformatics Rollins School of Public Health Emory University September 10, 2015 Background
Introduction to mass spectrometry (MS) based proteomics and metabolomics Tianwei Yu Department of Biostatistics and Bioinformatics Rollins School of Public Health Emory University September 10, 2015 Background
Mass Frontier 7.0 Quick Start Guide
 Mass Frontier 7.0 Quick Start Guide The topics in this guide briefly step you through key features of the Mass Frontier application. Editing a Structure Working with Spectral Trees Building a Library Predicting
Mass Frontier 7.0 Quick Start Guide The topics in this guide briefly step you through key features of the Mass Frontier application. Editing a Structure Working with Spectral Trees Building a Library Predicting
Protein Prospector and Ways of Calculating Expectation Values
 Protein Prospector and Ways of Calculating Expectation Values 1/16 Aenoch J. Lynn; Robert J. Chalkley; Peter R. Baker; Mark R. Segal; and Alma L. Burlingame University of California, San Francisco, San
Protein Prospector and Ways of Calculating Expectation Values 1/16 Aenoch J. Lynn; Robert J. Chalkley; Peter R. Baker; Mark R. Segal; and Alma L. Burlingame University of California, San Francisco, San
La Protéomique : Etat de l art et perspectives
 La Protéomique : Etat de l art et perspectives Odile Schiltz Institut de Pharmacologie et de Biologie Structurale CNRS, Université de Toulouse, [email protected] Protéomique et Spectrométrie de Masse
La Protéomique : Etat de l art et perspectives Odile Schiltz Institut de Pharmacologie et de Biologie Structurale CNRS, Université de Toulouse, [email protected] Protéomique et Spectrométrie de Masse
ProteinPilot Report for ProteinPilot Software
 ProteinPilot Report for ProteinPilot Software Detailed Analysis of Protein Identification / Quantitation Results Automatically Sean L Seymour, Christie Hunter SCIEX, USA Pow erful mass spectrometers like
ProteinPilot Report for ProteinPilot Software Detailed Analysis of Protein Identification / Quantitation Results Automatically Sean L Seymour, Christie Hunter SCIEX, USA Pow erful mass spectrometers like
Proteomics in Practice
 Reiner Westermeier, Torn Naven Hans-Rudolf Höpker Proteomics in Practice A Guide to Successful Experimental Design 2008 Wiley-VCH Verlag- Weinheim 978-3-527-31941-1 Preface Foreword XI XIII Abbreviations,
Reiner Westermeier, Torn Naven Hans-Rudolf Höpker Proteomics in Practice A Guide to Successful Experimental Design 2008 Wiley-VCH Verlag- Weinheim 978-3-527-31941-1 Preface Foreword XI XIII Abbreviations,
Unique Software Tools to Enable Quick Screening and Identification of Residues and Contaminants in Food Samples using Accurate Mass LC-MS/MS
 Unique Software Tools to Enable Quick Screening and Identification of Residues and Contaminants in Food Samples using Accurate Mass LC-MS/MS Using PeakView Software with the XIC Manager to Get the Answers
Unique Software Tools to Enable Quick Screening and Identification of Residues and Contaminants in Food Samples using Accurate Mass LC-MS/MS Using PeakView Software with the XIC Manager to Get the Answers
Improving the Metabolite Identification Process with Efficiency and Speed: LightSight Software for Metabolite Identification
 Improving the Metabolite Identification Process with Efficiency and Speed: LightSight Software for Metabolite Identification Overview LightSight Software for Metabolite Identification is a complete environment
Improving the Metabolite Identification Process with Efficiency and Speed: LightSight Software for Metabolite Identification Overview LightSight Software for Metabolite Identification is a complete environment
Choices, choices, choices... Which sequence database? Which modifications? What mass tolerance?
 Optimization 1 Choices, choices, choices... Which sequence database? Which modifications? What mass tolerance? Where to begin? 2 Sequence Databases Swiss-prot MSDB, NCBI nr dbest Species specific ORFS
Optimization 1 Choices, choices, choices... Which sequence database? Which modifications? What mass tolerance? Where to begin? 2 Sequence Databases Swiss-prot MSDB, NCBI nr dbest Species specific ORFS
Challenges in Computational Analysis of Mass Spectrometry Data for Proteomics
 Ma B. Challenges in computational analysis of mass spectrometry data for proteomics. SCIENCE AND TECHNOLOGY 25(1): 1 Jan. 2010 JOURNAL OF COMPUTER Challenges in Computational Analysis of Mass Spectrometry
Ma B. Challenges in computational analysis of mass spectrometry data for proteomics. SCIENCE AND TECHNOLOGY 25(1): 1 Jan. 2010 JOURNAL OF COMPUTER Challenges in Computational Analysis of Mass Spectrometry
Overview. Introduction. AB SCIEX MPX -2 High Throughput TripleTOF 4600 LC/MS/MS System
 Investigating the use of the AB SCIEX TripleTOF 4600 LC/MS/MS System for High Throughput Screening of Synthetic Cannabinoids/Metabolites in Human Urine AB SCIEX MPX -2 High Throughput TripleTOF 4600 LC/MS/MS
Investigating the use of the AB SCIEX TripleTOF 4600 LC/MS/MS System for High Throughput Screening of Synthetic Cannabinoids/Metabolites in Human Urine AB SCIEX MPX -2 High Throughput TripleTOF 4600 LC/MS/MS
MASCOT Search Results Interpretation
 The Mascot protein identification program (Matrix Science, Ltd.) uses statistical methods to assess the validity of a match. MS/MS data is not ideal. That is, there are unassignable peaks (noise) and usually
The Mascot protein identification program (Matrix Science, Ltd.) uses statistical methods to assess the validity of a match. MS/MS data is not ideal. That is, there are unassignable peaks (noise) and usually
PeptidomicsDB: a new platform for sharing MS/MS data.
 PeptidomicsDB: a new platform for sharing MS/MS data. Federica Viti, Ivan Merelli, Dario Di Silvestre, Pietro Brunetti, Luciano Milanesi, Pierluigi Mauri NETTAB2010 Napoli, 01/12/2010 Mass Spectrometry
PeptidomicsDB: a new platform for sharing MS/MS data. Federica Viti, Ivan Merelli, Dario Di Silvestre, Pietro Brunetti, Luciano Milanesi, Pierluigi Mauri NETTAB2010 Napoli, 01/12/2010 Mass Spectrometry
Error Tolerant Searching of Uninterpreted MS/MS Data
 Error Tolerant Searching of Uninterpreted MS/MS Data 1 In any search of a large LC-MS/MS dataset 2 There are always a number of spectra which get poor scores, or even no match at all. 3 Sometimes, this
Error Tolerant Searching of Uninterpreted MS/MS Data 1 In any search of a large LC-MS/MS dataset 2 There are always a number of spectra which get poor scores, or even no match at all. 3 Sometimes, this
Research-grade Targeted Proteomics Assay Development: PRMs for PTM Studies with Skyline or, How I learned to ditch the triple quad and love the QE
 Research-grade Targeted Proteomics Assay Development: PRMs for PTM Studies with Skyline or, How I learned to ditch the triple quad and love the QE Jacob D. Jaffe Skyline Webinar July 2015 Proteomics and
Research-grade Targeted Proteomics Assay Development: PRMs for PTM Studies with Skyline or, How I learned to ditch the triple quad and love the QE Jacob D. Jaffe Skyline Webinar July 2015 Proteomics and
A Navigation through the Tracefinder Software Structure and Workflow Options. Frans Schoutsen Pesticide Symposium Prague 27 April 2015
 A Navigation through the Tracefinder Software Structure and Workflow Options Frans Schoutsen Pesticide Symposium Prague 27 April 2015 Kings day in The Netherlands 1 Index Introduction Acquisition, Method
A Navigation through the Tracefinder Software Structure and Workflow Options Frans Schoutsen Pesticide Symposium Prague 27 April 2015 Kings day in The Netherlands 1 Index Introduction Acquisition, Method
MassHunter for Agilent GC/MS & GC/MS/MS
 MassHunter for Agilent GC/MS & GC/MS/MS Next Generation Data Analysis Software Presented by : Terry Harper GC/MS Product Specialist 1 Outline of Topics Topic 1: Introduction to MassHunter Topic 2: Data
MassHunter for Agilent GC/MS & GC/MS/MS Next Generation Data Analysis Software Presented by : Terry Harper GC/MS Product Specialist 1 Outline of Topics Topic 1: Introduction to MassHunter Topic 2: Data
Advantages of the LTQ Orbitrap for Protein Identification in Complex Digests
 Application Note: 386 Advantages of the LTQ Orbitrap for Protein Identification in Complex Digests Rosa Viner, Terry Zhang, Scott Peterman, and Vlad Zabrouskov, Thermo Fisher Scientific, San Jose, CA,
Application Note: 386 Advantages of the LTQ Orbitrap for Protein Identification in Complex Digests Rosa Viner, Terry Zhang, Scott Peterman, and Vlad Zabrouskov, Thermo Fisher Scientific, San Jose, CA,
Mass Frontier Version 7.0
 Mass Frontier Version 7.0 User Guide XCALI-97349 Revision A February 2011 2011 Thermo Fisher Scientific Inc. All rights reserved. Mass Frontier, Mass Frontier Server Manager, Fragmentation Library, Spectral
Mass Frontier Version 7.0 User Guide XCALI-97349 Revision A February 2011 2011 Thermo Fisher Scientific Inc. All rights reserved. Mass Frontier, Mass Frontier Server Manager, Fragmentation Library, Spectral
Cliquid ChemoView 3.0 Software Simple automated analysis, from sample to report
 PRODUCT BULLETIN Cliquid ChemoView 3.0 Software for Routine Screening and Quantitation Cliquid ChemoView 3.0 Software Simple automated analysis, from sample to report KEY FEATURES Secure user login that
PRODUCT BULLETIN Cliquid ChemoView 3.0 Software for Routine Screening and Quantitation Cliquid ChemoView 3.0 Software Simple automated analysis, from sample to report KEY FEATURES Secure user login that
Thermo Scientific PepFinder Software A New Paradigm for Peptide Mapping
 Thermo Scientific PepFinder Software A New Paradigm for Peptide Mapping For Conclusive Characterization of Biologics Deep Protein Characterization Is Crucial Pharmaceuticals have historically been small
Thermo Scientific PepFinder Software A New Paradigm for Peptide Mapping For Conclusive Characterization of Biologics Deep Protein Characterization Is Crucial Pharmaceuticals have historically been small
Preprocessing, Management, and Analysis of Mass Spectrometry Proteomics Data
 Preprocessing, Management, and Analysis of Mass Spectrometry Proteomics Data M. Cannataro, P. H. Guzzi, T. Mazza, and P. Veltri Università Magna Græcia di Catanzaro, Italy 1 Introduction Mass Spectrometry
Preprocessing, Management, and Analysis of Mass Spectrometry Proteomics Data M. Cannataro, P. H. Guzzi, T. Mazza, and P. Veltri Università Magna Græcia di Catanzaro, Italy 1 Introduction Mass Spectrometry
Integrated Data Mining Strategy for Effective Metabolomic Data Analysis
 The First International Symposium on Optimization and Systems Biology (OSB 07) Beijing, China, August 8 10, 2007 Copyright 2007 ORSC & APORC pp. 45 51 Integrated Data Mining Strategy for Effective Metabolomic
The First International Symposium on Optimization and Systems Biology (OSB 07) Beijing, China, August 8 10, 2007 Copyright 2007 ORSC & APORC pp. 45 51 Integrated Data Mining Strategy for Effective Metabolomic
Tutorial 9: SWATH data analysis in Skyline
 Tutorial 9: SWATH data analysis in Skyline In this tutorial we will learn how to perform targeted post-acquisition analysis for protein identification and quantitation using a data-independent dataset
Tutorial 9: SWATH data analysis in Skyline In this tutorial we will learn how to perform targeted post-acquisition analysis for protein identification and quantitation using a data-independent dataset
Proteomic Analysis using Accurate Mass Tags. Gordon Anderson PNNL January 4-5, 2005
 Proteomic Analysis using Accurate Mass Tags Gordon Anderson PNNL January 4-5, 2005 Outline Accurate Mass and Time Tag (AMT) based proteomics Instrumentation Data analysis Data management Challenges 2 Approach
Proteomic Analysis using Accurate Mass Tags Gordon Anderson PNNL January 4-5, 2005 Outline Accurate Mass and Time Tag (AMT) based proteomics Instrumentation Data analysis Data management Challenges 2 Approach
A Streamlined Workflow for Untargeted Metabolomics
 A Streamlined Workflow for Untargeted Metabolomics Employing XCMS plus, a Simultaneous Data Processing and Metabolite Identification Software Package for Rapid Untargeted Metabolite Screening Baljit K.
A Streamlined Workflow for Untargeted Metabolomics Employing XCMS plus, a Simultaneous Data Processing and Metabolite Identification Software Package for Rapid Untargeted Metabolite Screening Baljit K.
AN ITERATIVE ALGORITHM TO QUANTIFY THE FACTORS INFLUENCING PEPTIDE FRAGMENTATION FOR MS/MS SPECTRUM
 353 1 AN ITERATIVE ALGORITHM TO QUANTIFY THE FACTORS INFLUENCING PEPTIDE FRAGMENTATION FOR MS/MS SPECTRUM Chungong Yu, Yu Lin, Shiwei Sun, Jinjin Cai, Jingfen Zhang Institute of Computing Technology, Chinese
353 1 AN ITERATIVE ALGORITHM TO QUANTIFY THE FACTORS INFLUENCING PEPTIDE FRAGMENTATION FOR MS/MS SPECTRUM Chungong Yu, Yu Lin, Shiwei Sun, Jinjin Cai, Jingfen Zhang Institute of Computing Technology, Chinese
Background Information
 1 Gas Chromatography/Mass Spectroscopy (GC/MS/MS) Background Information Instructions for the Operation of the Varian CP-3800 Gas Chromatograph/ Varian Saturn 2200 GC/MS/MS See the Cary Eclipse Software
1 Gas Chromatography/Mass Spectroscopy (GC/MS/MS) Background Information Instructions for the Operation of the Varian CP-3800 Gas Chromatograph/ Varian Saturn 2200 GC/MS/MS See the Cary Eclipse Software
# LCMS-35 esquire series. Application of LC/APCI Ion Trap Tandem Mass Spectrometry for the Multiresidue Analysis of Pesticides in Water
 Application Notes # LCMS-35 esquire series Application of LC/APCI Ion Trap Tandem Mass Spectrometry for the Multiresidue Analysis of Pesticides in Water An LC-APCI-MS/MS method using an ion trap system
Application Notes # LCMS-35 esquire series Application of LC/APCI Ion Trap Tandem Mass Spectrometry for the Multiresidue Analysis of Pesticides in Water An LC-APCI-MS/MS method using an ion trap system
Computational analysis of unassigned high-quality MS/MS spectra in proteomic data sets
 2712 DOI 10.1002/pmic.200900473 Proteomics 2010, 10, 2712 2718 TECHNICAL BRIEF Computational analysis of unassigned high-quality MS/MS spectra in proteomic data sets Kang Ning 1, Damian Fermin 1 and Alexey
2712 DOI 10.1002/pmic.200900473 Proteomics 2010, 10, 2712 2718 TECHNICAL BRIEF Computational analysis of unassigned high-quality MS/MS spectra in proteomic data sets Kang Ning 1, Damian Fermin 1 and Alexey
Introduction to Proteomics
 Introduction to Proteomics Åsa Wheelock, Ph.D. Division of Respiratory Medicine & Karolinska Biomics Center [email protected] In: Systems Biology and the Omics Cascade, Karolinska Institutet, June 9-13,
Introduction to Proteomics Åsa Wheelock, Ph.D. Division of Respiratory Medicine & Karolinska Biomics Center [email protected] In: Systems Biology and the Omics Cascade, Karolinska Institutet, June 9-13,
MultiQuant Software Version 3.0 for Accurate Quantification of Clinical Research and Forensic Samples
 MultiQuant Software Version 3.0 for Accurate Quantification of Clinical Research and Forensic Samples Fast and Efficient Data Review, with Automatic Flagging of Outlier Results Adrian M. Taylor and Michael
MultiQuant Software Version 3.0 for Accurate Quantification of Clinical Research and Forensic Samples Fast and Efficient Data Review, with Automatic Flagging of Outlier Results Adrian M. Taylor and Michael
Already said. Already said. Outlook. Look at LC-MS data. A look at data for quantitative analysis using MSight and Phenyx. What data for quantitation?
 A look at data for quantitative analysis using MSight and Phenyx Pierre-Alain Binz Institut Suisse de Bioinformatique GeneBio SA Atelier Protéomique Quantitative 25-27 Juin 2007 La Grande Motte Already
A look at data for quantitative analysis using MSight and Phenyx Pierre-Alain Binz Institut Suisse de Bioinformatique GeneBio SA Atelier Protéomique Quantitative 25-27 Juin 2007 La Grande Motte Already
using ms based proteomics
 quantification using ms based proteomics lennart martens Computational Omics and Systems Biology Group Department of Medical Protein Research, VIB Department of Biochemistry, Ghent University Ghent, Belgium
quantification using ms based proteomics lennart martens Computational Omics and Systems Biology Group Department of Medical Protein Research, VIB Department of Biochemistry, Ghent University Ghent, Belgium
Statistical Analysis Strategies for Shotgun Proteomics Data
 Statistical Analysis Strategies for Shotgun Proteomics Data Ming Li, Ph.D. Cancer Biostatistics Center Vanderbilt University Medical Center Ayers Institute Biomarker Pipeline normal shotgun proteome analysis
Statistical Analysis Strategies for Shotgun Proteomics Data Ming Li, Ph.D. Cancer Biostatistics Center Vanderbilt University Medical Center Ayers Institute Biomarker Pipeline normal shotgun proteome analysis
Metabolomics Software Tools. Xiuxia Du, Paul Benton, Stephen Barnes
 Metabolomics Software Tools Xiuxia Du, Paul Benton, Stephen Barnes Outline 2 Introduction Software Tools for LC-MS metabolomics Software Tools for GC-MS metabolomics Software Tools for Statistical Analysis
Metabolomics Software Tools Xiuxia Du, Paul Benton, Stephen Barnes Outline 2 Introduction Software Tools for LC-MS metabolomics Software Tools for GC-MS metabolomics Software Tools for Statistical Analysis
Thermo Scientific SIEVE Software for Differential Expression Analysis
 m a s s s p e c t r o m e t r y Thermo Scientific SIEVE Software for Differential Expression Analysis Automated, label-free, semi-quantitative analysis of proteins, peptides, and metabolites based on comparisons
m a s s s p e c t r o m e t r y Thermo Scientific SIEVE Software for Differential Expression Analysis Automated, label-free, semi-quantitative analysis of proteins, peptides, and metabolites based on comparisons
Retrospective Analysis of a Host Cell Protein Perfect Storm: Identifying Immunogenic Proteins and Fixing the Problem
 Retrospective Analysis of a Host Cell Protein Perfect Storm: Identifying Immunogenic Proteins and Fixing the Problem Kevin Van Cott, Associate Professor Dept. of Chemical and Biomolecular Engineering Nebraska
Retrospective Analysis of a Host Cell Protein Perfect Storm: Identifying Immunogenic Proteins and Fixing the Problem Kevin Van Cott, Associate Professor Dept. of Chemical and Biomolecular Engineering Nebraska
Introduction to Proteomics
 Introduction to Proteomics Why Proteomics? Same Genome Different Proteome Black Swallowtail - larvae and butterfly Biological Complexity Yeast - a simple proteome 6,113 proteins = 344,855 tryptic peptides
Introduction to Proteomics Why Proteomics? Same Genome Different Proteome Black Swallowtail - larvae and butterfly Biological Complexity Yeast - a simple proteome 6,113 proteins = 344,855 tryptic peptides
Analysis of Mass Spectrometry. Data for Protein Identification. in Complex Biological Mixtures
 Analysis of Mass Spectrometry Data for Protein Identification in Complex Biological Mixtures by Marina Spivak A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor
Analysis of Mass Spectrometry Data for Protein Identification in Complex Biological Mixtures by Marina Spivak A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor
The Open2Dprot Proteomics Project for n-dimensional Protein Expression Data Analysis
 The Open2Dprot Proteomics Project for n-dimensional Protein Expression Data Analysis http://open2dprot.sourceforge.net/ Revised 2-05-2006 * (cf. 2D-LC) Introduction There is a need for integrated proteomics
The Open2Dprot Proteomics Project for n-dimensional Protein Expression Data Analysis http://open2dprot.sourceforge.net/ Revised 2-05-2006 * (cf. 2D-LC) Introduction There is a need for integrated proteomics
A Quadrupole-Orbitrap Hybrid Mass Spectrometer Offers Highest Benchtop Performance for In-Depth Analysis of Complex Proteomes
 A Quadrupole-Orbitrap Hybrid Mass Spectrometer Offers Highest Benchtop Performance for In-Depth Analysis of Complex Proteomes Zhiqi Hao 1, Yi Zhang 1, Shannon Eliuk 1, Justin Blethrow 1, Dave Horn 1, Vlad
A Quadrupole-Orbitrap Hybrid Mass Spectrometer Offers Highest Benchtop Performance for In-Depth Analysis of Complex Proteomes Zhiqi Hao 1, Yi Zhang 1, Shannon Eliuk 1, Justin Blethrow 1, Dave Horn 1, Vlad
SimGlycan Software*: A New Predictive Carbohydrate Analysis Tool for MS/MS Data
 SimGlycan Software*: A New Predictive Carbohydrate Analysis Tool for MS/MS Data Automated Data Interpretation for Glycan Characterization Jenny Albanese 1, Matthias Glueckmann 2 and Christof Lenz 2 1 AB
SimGlycan Software*: A New Predictive Carbohydrate Analysis Tool for MS/MS Data Automated Data Interpretation for Glycan Characterization Jenny Albanese 1, Matthias Glueckmann 2 and Christof Lenz 2 1 AB
Supplementary Materials and Methods (Metabolomics analysis)
 Supplementary Materials and Methods (Metabolomics analysis) Metabolite extraction and mass spectrometry analysis. 12 Vldlr-/- knock out and 12 wild type (WT) retinas were separately frozen at -80 ºC in
Supplementary Materials and Methods (Metabolomics analysis) Metabolite extraction and mass spectrometry analysis. 12 Vldlr-/- knock out and 12 wild type (WT) retinas were separately frozen at -80 ºC in
API 3200 LC/MS/MS SYSTEM. Performance, productivity and value combined
 API 3200 LC/MS/MS SYSTEM Performance, productivity and value combined API 3200 LC/MS/MS SYSTEM Reliability, reproducibility, and confidence in your data. With an unmatched heritage of technological innovation
API 3200 LC/MS/MS SYSTEM Performance, productivity and value combined API 3200 LC/MS/MS SYSTEM Reliability, reproducibility, and confidence in your data. With an unmatched heritage of technological innovation
Waters Core Chromatography Training (2 Days)
 2015 Page 2 Waters Core Chromatography Training (2 Days) The learning objective of this two day course is to teach the user core chromatography, system and software fundamentals in an outcomes based approach.
2015 Page 2 Waters Core Chromatography Training (2 Days) The learning objective of this two day course is to teach the user core chromatography, system and software fundamentals in an outcomes based approach.
Increasing Quality While Maintaining Efficiency in Drug Chemistry with DART-TOF MS Screening
 Increasing Quality While Maintaining Efficiency in Drug Chemistry with DART-TOF MS Screening Application Note Forensics Author Erin Shonsey Director of Research Alabama Department of Forensic Science Abstract
Increasing Quality While Maintaining Efficiency in Drug Chemistry with DART-TOF MS Screening Application Note Forensics Author Erin Shonsey Director of Research Alabama Department of Forensic Science Abstract
NUVISAN Pharma Services
 NUVISAN Pharma Services CESI MS Now available! 1st CRO in Europe! At the highest levels of quality. LABORATORY SERVICES Equipment update STATE OF THE ART AT NUVISAN CESI MS Now available! 1st CRO in Europe!
NUVISAN Pharma Services CESI MS Now available! 1st CRO in Europe! At the highest levels of quality. LABORATORY SERVICES Equipment update STATE OF THE ART AT NUVISAN CESI MS Now available! 1st CRO in Europe!
A Hybrid Microchip/Capillary Electrophoresis Mass Spectrometry Platform for Rapid and Ultrasensitive Bioanalysis
 A Hybrid Microchip/Capillary Electrophoresis Mass Spectrometry Platform for Rapid and Ultrasensitive Bioanalysis Ryan Kelly 1, Chenchen Wang, Cheng S. Lee, Richard D. Smith, Keqi Tang 1 Environmental Molecular
A Hybrid Microchip/Capillary Electrophoresis Mass Spectrometry Platform for Rapid and Ultrasensitive Bioanalysis Ryan Kelly 1, Chenchen Wang, Cheng S. Lee, Richard D. Smith, Keqi Tang 1 Environmental Molecular
Pinpointing phosphorylation sites using Selected Reaction Monitoring and Skyline
 Pinpointing phosphorylation sites using Selected Reaction Monitoring and Skyline Christina Ludwig group of Ruedi Aebersold, ETH Zürich The challenge of phosphosite assignment Peptides Phosphopeptides MS/MS
Pinpointing phosphorylation sites using Selected Reaction Monitoring and Skyline Christina Ludwig group of Ruedi Aebersold, ETH Zürich The challenge of phosphosite assignment Peptides Phosphopeptides MS/MS
Shotgun Proteomic Analysis. Department of Cell Biology The Scripps Research Institute
 Shotgun Proteomic Analysis Department of Cell Biology The Scripps Research Institute Biological/Functional Resolution of Experiments Organelle Multiprotein Complex Cells/Tissues Function Expression Analysis
Shotgun Proteomic Analysis Department of Cell Biology The Scripps Research Institute Biological/Functional Resolution of Experiments Organelle Multiprotein Complex Cells/Tissues Function Expression Analysis
HRMS in Clinical Research: from Targeted Quantification to Metabolomics
 A sponsored whitepaper. HRMS in Clinical Research: from Targeted Quantification to Metabolomics By: Bertrand Rochat Ph. D., Research Project Leader, Faculté de Biologie et de Médecine of the Centre Hospitalier
A sponsored whitepaper. HRMS in Clinical Research: from Targeted Quantification to Metabolomics By: Bertrand Rochat Ph. D., Research Project Leader, Faculté de Biologie et de Médecine of the Centre Hospitalier
Thermo Scientific Prelude SPLC System FPO. Making LC/MS accessible. to clinical research and toxicology labs
 Thermo Scientific Prelude SPLC System FPO Making LC/MS accessible to clinical research and toxicology labs Specifically designed to make the power of LC/MS accessible Analysis of target compounds in complex
Thermo Scientific Prelude SPLC System FPO Making LC/MS accessible to clinical research and toxicology labs Specifically designed to make the power of LC/MS accessible Analysis of target compounds in complex
Enhancing GCMS analysis of trace compounds using a new dynamic baseline compensation algorithm to reduce background interference
 Enhancing GCMS analysis of trace compounds using a new dynamic baseline compensation algorithm to reduce background interference Abstract The advantages of mass spectrometry (MS) in combination with gas
Enhancing GCMS analysis of trace compounds using a new dynamic baseline compensation algorithm to reduce background interference Abstract The advantages of mass spectrometry (MS) in combination with gas
Step-by-Step Analytical Methods Validation and Protocol in the Quality System Compliance Industry
 Step-by-Step Analytical Methods Validation and Protocol in the Quality System Compliance Industry BY GHULAM A. SHABIR Introduction Methods Validation: Establishing documented evidence that provides a high
Step-by-Step Analytical Methods Validation and Protocol in the Quality System Compliance Industry BY GHULAM A. SHABIR Introduction Methods Validation: Establishing documented evidence that provides a high
SCREENING FOR DRUGS IN SERUM AND URINE BY LC/ESI/CID-MS AND MS/MS WITH LIBRARY SEARCHING *
 SCREENING FOR DRUGS IN SERUM AND URINE BY LC/ESI/CID-MS AND MS/MS WITH LIBRARY SEARCHING * Wolfgang WEINMANN 1, Natalie LEHMANN 1, Michaela RENZ 1, Alexander WIEDEMANN 1, Michal SVOBODA 2 1 Institute of
SCREENING FOR DRUGS IN SERUM AND URINE BY LC/ESI/CID-MS AND MS/MS WITH LIBRARY SEARCHING * Wolfgang WEINMANN 1, Natalie LEHMANN 1, Michaela RENZ 1, Alexander WIEDEMANN 1, Michal SVOBODA 2 1 Institute of
Guide to Reverse Phase SpinColumns Chromatography for Sample Prep
 Guide to Reverse Phase SpinColumns Chromatography for Sample Prep www.harvardapparatus.com Contents Introduction...2-3 Modes of Separation...4-6 Spin Column Efficiency...7-8 Fast Protein Analysis...9 Specifications...10
Guide to Reverse Phase SpinColumns Chromatography for Sample Prep www.harvardapparatus.com Contents Introduction...2-3 Modes of Separation...4-6 Spin Column Efficiency...7-8 Fast Protein Analysis...9 Specifications...10
Analyzing Small Molecules by EI and GC-MS. July 2014
 Analyzing Small Molecules by EI and GC-MS July 2014 Samples Appropriate for GC-MS Volatile and semi-volatile organic compounds Rule of thumb,
Analyzing Small Molecules by EI and GC-MS July 2014 Samples Appropriate for GC-MS Volatile and semi-volatile organic compounds Rule of thumb,
Using NIST Search with Agilent MassHunter Qualitative Analysis Software James Little, Eastman Chemical Company Sept 20, 2012.
 Using NIST Search with Agilent MassHunter Qualitative Analysis Software James Little, Eastman Chemical Company Sept 20, 2012 Introduction Screen captures in this document were taken from MassHunter B.05.00
Using NIST Search with Agilent MassHunter Qualitative Analysis Software James Little, Eastman Chemical Company Sept 20, 2012 Introduction Screen captures in this document were taken from MassHunter B.05.00
Simultaneous Metabolite Identification and Quantitation with UV Data Integration Using LightSight Software Version 2.2
 Technical ote Simultaneous Metabolite Identification and Quantitation with UV Data Integration Using LightSight Software Version 2.2 Alek. Dooley, Carmai Seto, esham Ghobarah, and Elliott B. Jones verview:
Technical ote Simultaneous Metabolite Identification and Quantitation with UV Data Integration Using LightSight Software Version 2.2 Alek. Dooley, Carmai Seto, esham Ghobarah, and Elliott B. Jones verview:
