Revised: 9/2014. LUMIGAN (bimatoprost ophthalmic solution) 0.01% for topical ophthalmic use. Initial U.S. Approval: 2001
|
|
|
- Grant Hart
- 9 years ago
- Views:
Transcription
1 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LUMIGAN (bimatoprost ophthalmic solution) 0.01% safely and effectively. See full prescribing information for LUMIGAN 0.01%. LUMIGAN (bimatoprost ophthalmic solution) 0.01% for topical ophthalmic use Initial U.S. Approval: 2001 RECENT MAJOR CHANGES Warnings and Precautions, Intraocular Inflammation (5.3) 9/2014 INDICATIONS AND USAGE LUMIGAN 0.01% is a prostaglandin analog indicated for the reduction of elevated intraocular pressure in patients with open angle glaucoma or ocular hypertension. (1) DOSAGE AND ADMINISTRATION One drop in the affected eye(s) once daily in the evening. (2) DOSAGE FORMS AND STRENGTHS Solution containing 0.1 mg/ml bimatoprost. (3) CONTRAINDICATIONS None (4) WARNINGS AND PRECAUTIONS Pigmentation. Pigmentation of the iris, periorbital tissue (eyelid) and eyelashes can occur. Iris pigmentation is likely to be permanent. (5.1) Eyelash Changes. Gradual change to eyelashes including increased length, thickness and number of lashes. Usually reversible. (5.2) ADVERSE REACTIONS Most common adverse reaction is conjunctival hyperemia (31%). (6.1) To report SUSPECTED ADVERSE REACTIONS, contact Allergan at or FDA at FDA-1088 or USE IN SPECIFIC POPULATIONS Use in pediatric patients below the age of 16 years is not recommended because of potential safety concerns related to increased pigmentation following long-term chronic use. (8.4) See 17 for PATIENT COUNSELING INFORMATION. Revised: 9/2014 FULL PRESCRIBING INFORMATION: CONTENTS* 1 INDICATIONS AND USAGE 2 DOSAGE AND ADMINISTRATION 3 DOSAGE FORMS AND STRENGTHS 4 CONTRAINDICATIONS 5 WARNINGS AND PRECAUTIONS 5.1 Pigmentation 5.2 Eyelash Changes 5.3 Intraocular Inflammation 5.4 Macular Edema 5.5 Bacterial Keratitis 5.6 Use with Contact Lenses 6 ADVERSE REACTIONS 6.1 Clinical Studies Experience 6.2 Postmarketing Experience 8 USE IN SPECIFIC POPULATIONS 8.1 Pregnancy 8.3 Nursing Mothers 8.4 Pediatric Use 8.5 Geriatric Use 8.6 Hepatic Impairment 10 OVERDOSAGE 11 DESCRIPTION 12 CLINICAL PHARMACOLOGY 12.1 Mechanism of Action 12.3 Pharmacokinetics 13 NONCLINICAL TOXICOLOGY 13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility 14 CLINICAL STUDIES 16 HOW SUPPLIED/STORAGE AND HANDLING 17 PATIENT COUNSELING INFORMATION 17.1 Potential for Pigmentation 17.2 Potential for Eyelash Changes 17.3 Handling the Container 17.4 When to Seek Physician Advice 17.5 Use with Contact Lenses 17.6 Use with Other Ophthalmic Drugs * Sections or subsections omitted from the full prescribing information are not listed.
2 FULL PRESCRIBING INFORMATION 1 INDICATIONS AND USAGE LUMIGAN (bimatoprost ophthalmic solution) 0.01% is indicated for the reduction of elevated intraocular pressure in patients with open angle glaucoma or ocular hypertension. 2 DOSAGE AND ADMINISTRATION The recommended dosage is one drop in the affected eye(s) once daily in the evening. LUMIGAN (bimatoprost ophthalmic solution) 0.01% should not be administered more than once daily since it has been shown that more frequent administration of prostaglandin analogs may decrease the intraocular pressure lowering effect. Reduction of the intraocular pressure starts approximately 4 hours after the first administration with maximum effect reached within approximately 8 to 12 hours. LUMIGAN 0.01% may be used concomitantly with other topical ophthalmic drug products to lower intraocular pressure. If more than one topical ophthalmic drug is being used, the drugs should be administered at least five (5) minutes apart. 3 DOSAGE FORMS AND STRENGTHS Ophthalmic solution containing bimatoprost 0.1 mg/ml. 4 CONTRAINDICATIONS None 5 WARNINGS AND PRECAUTIONS 5.1 Pigmentation Bimatoprost ophthalmic solution has been reported to cause changes to pigmented tissues. The most frequently reported changes have been increased pigmentation of the iris, periorbital tissue (eyelid) and eyelashes. Pigmentation is expected to increase as long as bimatoprost is administered. The pigmentation change is due to increased melanin content in the melanocytes rather than to an increase in the number of melanocytes. After discontinuation of bimatoprost, pigmentation of the iris is likely to be permanent, while pigmentation of the periorbital tissue and eyelash changes have been reported to be reversible in some patients. Patients who receive treatment should be informed of the possibility of increased pigmentation. The long term effects of increased pigmentation are not known. Iris color change may not be noticeable for several months to years. Typically, the brown pigmentation around the pupil spreads concentrically towards the periphery of the iris and the entire iris or parts of the iris become more brownish. Neither nevi nor freckles of the iris appear to be affected by treatment. While treatment with LUMIGAN (bimatoprost ophthalmic solution) 0.01% can be continued in patients who develop noticeably increased iris pigmentation, these patients should be examined regularly [see Patient Counseling Information (17.1)]. 5.2 Eyelash Changes LUMIGAN 0.01% may gradually change eyelashes and vellus hair in the treated eye. These changes include increased length, thickness, and number of lashes. Eyelash changes are usually reversible upon discontinuation of treatment.
3 5.3 Intraocular Inflammation Prostaglandin analogs, including bimatoprost, have been reported to cause intraocular inflammation. In addition, because these products may exacerbate inflammation, caution should be used in patients with active intraocular inflammation (e.g., uveitis). 5.4 Macular Edema Macular edema, including cystoid macular edema, has been reported during treatment with bimatoprost ophthalmic solution. LUMIGAN 0.01% should be used with caution in aphakic patients, in pseudophakic patients with a torn posterior lens capsule, or in patients with known risk factors for macular edema. 5.5 Bacterial Keratitis There have been reports of bacterial keratitis associated with the use of multiple-dose containers of topical ophthalmic products. These containers had been inadvertently contaminated by patients who, in most cases, had a concurrent corneal disease or a disruption of the ocular epithelial surface [see Patient Counseling Information (17.3)]. 5.6 Use with Contact Lenses Contact lenses should be removed prior to instillation of LUMIGAN 0.01% and may be reinserted 15 minutes following its administration. 6 ADVERSE REACTIONS 6.1 Clinical Studies Experience Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in practice. In a 12-month clinical study with bimatoprost ophthalmic solutions 0.01%, the most common adverse reaction was conjunctival hyperemia (31%). Approximately 1.6% of patients discontinued therapy due to conjunctival hyperemia. Other adverse drug reactions (reported in 1 to 4% of patients) with LUMIGAN 0.01% in this study included conjunctival edema, conjunctival hemorrhage, eye irritation, eye pain, eye pruritus, erythema of eyelid, eyelids pruritus, growth of eyelashes, hypertrichosis, instillation site irritation, punctate keratitis, skin hyperpigmentation, vision blurred, and visual acuity reduced. 6.2 Postmarketing Experience The following reaction has been identified during postmarketing use of LUMIGAN 0.01% in clinical practice. Because it was reported voluntarily from a population of unknown size, estimates of frequency cannot be made. The reaction, which has been chosen for inclusion due to either its seriousness, frequency of reporting, possible causal connection to LUMIGAN 0.01%, or a combination of these factors, includes headache. In postmarketing use with prostaglandin analogs, periorbital and lid changes including deepening of the eyelid sulcus have been observed. 8 USE IN SPECIFIC POPULATIONS 8.1 Pregnancy Pregnancy Category C Teratogenic effects: In embryo/fetal developmental studies in pregnant mice and rats, abortion was observed at oral doses of bimatoprost which achieved at least 33 or 97 times, respectively, the maximum intended human exposure based on blood AUC levels.
4 At doses at least 41 times the maximum intended human exposure based on blood AUC levels, the gestation length was reduced in the dams, the incidence of dead fetuses, late resorptions, peri- and postnatal pup mortality was increased, and pup body weights were reduced. There are no adequate and well-controlled studies of LUMIGAN (bimatoprost ophthalmic solution) 0.01% administration in pregnant women. Because animal reproductive studies are not always predictive of human response LUMIGAN 0.01% should be administered during pregnancy only if the potential benefit justifies the potential risk to the fetus. 8.3 Nursing Mothers It is not known whether LUMIGAN 0.01% is excreted in human milk, although in animal studies, bimatoprost has been shown to be excreted in breast milk. Because many drugs are excreted in human milk, caution should be exercised when LUMIGAN 0.01% is administered to a nursing woman. 8.4 Pediatric Use Use in pediatric patients below the age of 16 years is not recommended because of potential safety concerns related to increased pigmentation following long-term chronic use. 8.5 Geriatric Use No overall clinical differences in safety or effectiveness have been observed between elderly and other adult patients. 8.6 Hepatic Impairment In patients with a history of liver disease or abnormal ALT, AST and/or bilirubin at baseline, bimatoprost 0.03% had no adverse effect on liver function over 48 months. 10 OVERDOSAGE No information is available on overdosage in humans. If overdose with LUMIGAN (bimatoprost ophthalmic solution) 0.01% occurs, treatment should be symptomatic. In oral (by gavage) mouse and rat studies, doses up to 100 mg/kg/day did not produce any toxicity. This dose expressed as mg/m 2 is at least 210 times higher than the accidental dose of one bottle of LUMIGAN 0.01% for a 10 kg child. 11 DESCRIPTION LUMIGAN (bimatoprost ophthalmic solution) 0.01% is a synthetic prostamide analog with ocular hypotensive activity. Its chemical name is (Z)-7-[(1R,2R,3R,5S)-3,5-Dihydroxy-2-[(1E,3S)-3-hydroxy-5-phenyl- 1-pentenyl]cyclopentyl]-5-N-ethylheptenamide, and its molecular weight is Its molecular formula is C 25 H 37 NO 4. Its chemical structure is:
5 Bimatoprost is a powder, which is very soluble in ethyl alcohol and methyl alcohol and slightly soluble in water. LUMIGAN 0.01% is a clear, isotonic, colorless, sterile ophthalmic solution with an osmolality of approximately 290 mosmol/kg. LUMIGAN 0.01% contains Active: bimatoprost 0.1 mg/ml; Inactives: benzalkonium chloride 0.2 mg/ml; sodium chloride; sodium phosphate, dibasic; citric acid; and purified water. Sodium hydroxide and/or hydrochloric acid may be added to adjust ph. The ph during its shelf life ranges from CLINICAL PHARMACOLOGY 12.1 Mechanism of Action Bimatoprost, a prostaglandin analog, is a synthetic structural analog of prostaglandin with ocular hypotensive activity. It selectively mimics the effects of naturally occurring substances, prostamides. Bimatoprost is believed to lower intraocular pressure (IOP) in humans by increasing outflow of aqueous humor through both the trabecular meshwork and uveoscleral routes. Elevated IOP presents a major risk factor for glaucomatous field loss. The higher the level of IOP, the greater the likelihood of optic nerve damage and visual field loss Pharmacokinetics Absorption: After one drop of bimatoprost ophthalmic solution 0.03% was administered once daily to both eyes of 15 healthy subjects for two weeks, blood concentrations peaked within 10 minutes after dosing and were below the lower limit of detection (0.025 ng/ml) in most subjects within 1.5 hours after dosing. Mean C max and AUC 0-24hr values were similar on days 7 and 14 at approximately 0.08 ng/ml and 0.09 ng hr/ml, respectively, indicating that steady state was reached during the first week of ocular dosing. There was no significant systemic drug accumulation over time. Distribution: Bimatoprost is moderately distributed into body tissues with a steady-state volume of distribution of 0.67 L/kg. In human blood, bimatoprost resides mainly in the plasma. Approximately 12% of bimatoprost remains unbound in human plasma. Metabolism: Bimatoprost is the major circulating species in the blood once it reaches the systemic circulation following ocular dosing. Bimatoprost then undergoes oxidation, N-deethylation and glucuronidation to form a diverse variety of metabolites. Elimination: Following an intravenous dose of radiolabeled bimatoprost (3.12 mcg/kg) to six healthy subjects, the maximum blood concentration of unchanged drug was 12.2 ng/ml and decreased rapidly with an elimination half-life of approximately 45 minutes. The total blood clearance of bimatoprost was 1.5 L/hr/kg. Up to 67% of the administered dose was excreted in the urine while 25% of the dose was recovered in the feces. 13 NONCLINICAL TOXICOLOGY 13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility Bimatoprost was not carcinogenic in either mice or rats when administered by oral gavage at doses of up to 2 mg/kg/day and 1 mg/kg/day respectively (at least 192 and 291 times the recommended human exposure based on blood AUC levels respectively) for 104 weeks. Bimatoprost was not mutagenic or clastogenic in the Ames test, in the mouse lymphoma test, or in the in vivo mouse micronucleus tests. Bimatoprost did not impair fertility in male or female rats up to doses of 0.6 mg/kg/day (at least 103 times the recommended human exposure based on blood AUC levels).
6 14 CLINICAL STUDIES In a 12-month clinical study of patients with open angle glaucoma or ocular hypertension with an average baseline IOP of 23.5 mmhg, the IOP-lowering effect of LUMIGAN 0.01% once daily (in the evening) was up to 7.5 mmhg. 16 HOW SUPPLIED/STORAGE AND HANDLING LUMIGAN (bimatoprost ophthalmic solution) 0.01% is supplied sterile in opaque white low density polyethylene ophthalmic dispenser bottles and tips with turquoise polystyrene caps in the following sizes: 2.5 ml fill in a 5 ml container - NDC ml fill in a 10 ml container - NDC ml fill in a 10 ml container - NDC Storage: Store at 2-25 C (36-77 F). 17 PATIENT COUNSELING INFORMATION 17.1 Potential for Pigmentation Advise patients about the potential for increased brown pigmentation of the iris, which may be permanent. Also inform patients about the possibility of eyelid skin darkening, which may be reversible after discontinuation of LUMIGAN (bimatoprost ophthalmic solution) 0.01% Potential for Eyelash Changes Inform patients of the possibility of eyelash and vellus hair changes in the treated eye during treatment with LUMIGAN 0.01%. These changes may result in a disparity between eyes in length, thickness, pigmentation, number of eyelashes or vellus hairs, and/or direction of eyelash growth. Eyelash changes are usually reversible upon discontinuation of treatment Handling the Container Instruct patients to avoid allowing the tip of the dispensing container to contact the eye, surrounding structures, fingers, or any other surface in order to avoid contamination of the solution by common bacteria known to cause ocular infections. Serious damage to the eye and subsequent loss of vision may result from using contaminated solutions When to Seek Physician Advice Advise patients that if they develop an intercurrent ocular condition (e.g., trauma or infection), have ocular surgery, or develop any ocular reactions, particularly conjunctivitis and eyelid reactions, they should immediately seek their physician s advice concerning the continued use of LUMIGAN 0.01% Use with Contact Lenses Advise patients that LUMIGAN 0.01% contains benzalkonium chloride, which may be absorbed by soft contact lenses. Contact lenses should be removed prior to instillation of LUMIGAN 0.01% and may be reinserted 15 minutes following its administration Use with Other Ophthalmic Drugs Advise patients that if more than one topical ophthalmic drug is being used, the drugs should be administered at least five (5) minutes between applications Allergan, Inc. Irvine, CA marks owned by Allergan, Inc.
7 Patented. See: Made in the U.S.A US14
DO YOU HAVE ITCHY ALLERGY EYES? Find out about lasting relief
 DO YOU HAVE ITCHY ALLERGY EYES? Find out about lasting relief Common causes of itching due to eye allergies include: Pollen from trees, grasses, and ragweed Dust mites Cat dander Approximately 10 million
DO YOU HAVE ITCHY ALLERGY EYES? Find out about lasting relief Common causes of itching due to eye allergies include: Pollen from trees, grasses, and ragweed Dust mites Cat dander Approximately 10 million
Keep Up With Your Treatment and Keep Track of Your Eye Pressure
 Keep Up With Your Treatment and Keep Track of Your Eye Pressure Get into the habit of applying your XAATAN (latanoprost ophthalmic solution) eye drops at the same time every day DAY 1 DAY 2 DAY 3 DAY 4
Keep Up With Your Treatment and Keep Track of Your Eye Pressure Get into the habit of applying your XAATAN (latanoprost ophthalmic solution) eye drops at the same time every day DAY 1 DAY 2 DAY 3 DAY 4
See 17 for PATIENT COUNSELING INFORMATION. Revised: 3/2016 FULL PRESCRIBING INFORMATION: CONTENTS* WARNING: ADRENAL CRISIS IN THE SETTING OF SHOCK OR
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LYSODREN safely and effectively. See full prescribing information for LYSODREN. LYSODREN (mitotane)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LYSODREN safely and effectively. See full prescribing information for LYSODREN. LYSODREN (mitotane)
NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate.
 NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate. PRESENTATION Eye Drops: NAPHCON-A Eye Drops are a combination of an antihistamine (pheniramine maleate) and a decongestant
NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate. PRESENTATION Eye Drops: NAPHCON-A Eye Drops are a combination of an antihistamine (pheniramine maleate) and a decongestant
FULL PRESCRIBING INFORMATION: CONTENTS*
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PHOTREXA VISCOUS and PHOTREXA safely and effectively. See full prescribing information for PHOTREXA
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PHOTREXA VISCOUS and PHOTREXA safely and effectively. See full prescribing information for PHOTREXA
Selected Requirements of Prescribing Information
 The Selected Requirements of Prescribing Information (SRPI) is a checklist of 42 important format prescribing information (PI) items based on labeling regulations [21 CFR 201.56(d) and 201.57] and guidances.
The Selected Requirements of Prescribing Information (SRPI) is a checklist of 42 important format prescribing information (PI) items based on labeling regulations [21 CFR 201.56(d) and 201.57] and guidances.
Teriflunomide is the active metabolite of Leflunomide, a drug employed since 1994 for the treatment of rheumatoid arthritis (Baselt, 2011).
 Page 1 of 10 ANALYTE NAME AND STRUCTURE TERIFLUNOMIDE Teriflunomide TRADE NAME Aubagio CATEGORY Antimetabolite TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND sclerosis. The
Page 1 of 10 ANALYTE NAME AND STRUCTURE TERIFLUNOMIDE Teriflunomide TRADE NAME Aubagio CATEGORY Antimetabolite TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND sclerosis. The
ZOVIRAX Cold Sore Cream
 Data Sheet ZOVIRAX Cold Sore Cream Aciclovir 5% w/w Presentation Topical cream Indications ZOVIRAX Cold Sore Cream is indicated for the treatment of Herpes simplex virus infections of the lips and face
Data Sheet ZOVIRAX Cold Sore Cream Aciclovir 5% w/w Presentation Topical cream Indications ZOVIRAX Cold Sore Cream is indicated for the treatment of Herpes simplex virus infections of the lips and face
Learn More About Product Labeling
 Learn More About Product Labeling Product label The product label is developed during the formal process of review and approval by regulatory agencies of any medicine or medical product. There are specific
Learn More About Product Labeling Product label The product label is developed during the formal process of review and approval by regulatory agencies of any medicine or medical product. There are specific
DOSAGE FORMS AND STRENGTHS Intravitreal implant containing dexamethasone 0.7 mg in the NOVADUR solid polymer drug delivery system.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use OZURDEX safely and effectively. See full prescribing information for OZURDEX. OZURDEX (dexamethasone
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use OZURDEX safely and effectively. See full prescribing information for OZURDEX. OZURDEX (dexamethasone
EXPANDING YOUR HORIZONS
 Glaucoma Therapy EXPANDING YOUR HORIZONS Learn how to control glaucoma with SLT therapy. SLTPATIENT EDUCATION PROGRAM Glaucoma Glaucoma is a degenerative disease that if left untreated can cause permanent
Glaucoma Therapy EXPANDING YOUR HORIZONS Learn how to control glaucoma with SLT therapy. SLTPATIENT EDUCATION PROGRAM Glaucoma Glaucoma is a degenerative disease that if left untreated can cause permanent
FOR ORAL OR RECTAL ADMINISTRATION FOR THE PREVENTION AND TREATMENT OF PORTAL-SYSTEMIC ENCEPHALOPATHY
 FORM NO. 1360 VC3074 Rev.2/08 LACTULOSE SOLUTION, USP 10 g/15 ml FOR ORAL OR RECTAL ADMINISTRATION FOR THE PREVENTION AND TREATMENT OF PORTAL-SYSTEMIC ENCEPHALOPATHY Rx Only DESCRIPTION Lactulose is a
FORM NO. 1360 VC3074 Rev.2/08 LACTULOSE SOLUTION, USP 10 g/15 ml FOR ORAL OR RECTAL ADMINISTRATION FOR THE PREVENTION AND TREATMENT OF PORTAL-SYSTEMIC ENCEPHALOPATHY Rx Only DESCRIPTION Lactulose is a
8.5 Geriatric Use * Sections or subsections omitted from the full prescribing information are not listed.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Injectafer safely and effectively. See full prescribing information for Injectafer. INJECTAFER (ferric
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Injectafer safely and effectively. See full prescribing information for Injectafer. INJECTAFER (ferric
INITIATING ORAL AUBAGIO (teriflunomide) THERAPY
 FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
2 DOSAGE AND ADMINISTRATION 2.1 Dosing and Dose Adjustment
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Auryxia safely and effectively. See full prescribing information for Auryxia. Auryxia (ferric citrate)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Auryxia safely and effectively. See full prescribing information for Auryxia. Auryxia (ferric citrate)
PHENYLEPHRINE HYDROCHLORIDE INJECTION USP
 PRESCRIBING INFORMATION PHENYLEPHRINE HYDROCHLORIDE INJECTION USP 10 mg/ml Sandoz Canada Inc. Date of Preparation: September 1992 145 Jules-Léger Date of Revision : January 13, 2011 Boucherville, QC, Canada
PRESCRIBING INFORMATION PHENYLEPHRINE HYDROCHLORIDE INJECTION USP 10 mg/ml Sandoz Canada Inc. Date of Preparation: September 1992 145 Jules-Léger Date of Revision : January 13, 2011 Boucherville, QC, Canada
Hypromellose Eye Drops BP 0.3% w/v PL 23097/0006
 Hypromellose Eye Drops BP 0.3% w/v PL 23097/0006 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
Hypromellose Eye Drops BP 0.3% w/v PL 23097/0006 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
Reference ID: 3482803
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use EVZIO safely and effectively. See full prescribing information for EVZIO. EVZIO (naloxone hydrochloride
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use EVZIO safely and effectively. See full prescribing information for EVZIO. EVZIO (naloxone hydrochloride
VOLTAREN OPHTHA EYE DROPS
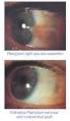 פורמט עלון זה נקבע ע"ימשרד הבריאות ותוכנו נבדק ואושר על ידו באפריל 2008 PRESCRIBING INFORMATION VOLTAREN OPHTHA EYE DROPS 1 Name of the medicinal product Voltaren Ophtha Eye Drops. 2 Qualitative and quantitative
פורמט עלון זה נקבע ע"ימשרד הבריאות ותוכנו נבדק ואושר על ידו באפריל 2008 PRESCRIBING INFORMATION VOLTAREN OPHTHA EYE DROPS 1 Name of the medicinal product Voltaren Ophtha Eye Drops. 2 Qualitative and quantitative
FAQ. Possible Allergic Reaction
 FAQ Possible Allergic Reaction First, please be sure you are using RevitaLash as directed on the package insert. Remember: only a small quantity of RevitaLash is needed to enhance the beauty of your natural
FAQ Possible Allergic Reaction First, please be sure you are using RevitaLash as directed on the package insert. Remember: only a small quantity of RevitaLash is needed to enhance the beauty of your natural
FRENCH AGENCY FOR VETERINARY MEDICINAL PRODUCTS DECENTRALISED PROCEDURE PUBLICLY AVAILABLE ASSESSMENT REPORT FOR A VETERINARY MEDICINAL PRODUCT
 FRENCH AGENCY FOR VETERINARY MEDICINAL PRODUCTS DECENTRALISED PROCEDURE FOR A VETERINARY MEDICINAL PRODUCT EMEDOG, 1 mg/ml, solution for injection for dogs Date: 20/08/2015 French agency for food, environnemental
FRENCH AGENCY FOR VETERINARY MEDICINAL PRODUCTS DECENTRALISED PROCEDURE FOR A VETERINARY MEDICINAL PRODUCT EMEDOG, 1 mg/ml, solution for injection for dogs Date: 20/08/2015 French agency for food, environnemental
Billing and Coding Guide
 Billing and Coding Guide INDICATIONS Injectafer is an iron replacement product indicated for the treatment of iron deficiency anemia (IDA) in adult patients: who have intolerance to or have had unsatisfactory
Billing and Coding Guide INDICATIONS Injectafer is an iron replacement product indicated for the treatment of iron deficiency anemia (IDA) in adult patients: who have intolerance to or have had unsatisfactory
Re: LUMIGAN 0.03% (bimatoprost ophthalmic solution 0.03%) and LUMIGAN 0.01% (bimatoprost ophthalmic solution 0.01%)
 Allergan Ltd, 1st Floor, Marlow International, Parkway, Marlow, Bucks SL7 1YL Tel: (01628) 494444 Facsimile: (01628) 494449 Ref No: UK15-000677 Mandeep Allingham NW Surrey CCG February 18, 2015 Dear Ms
Allergan Ltd, 1st Floor, Marlow International, Parkway, Marlow, Bucks SL7 1YL Tel: (01628) 494444 Facsimile: (01628) 494449 Ref No: UK15-000677 Mandeep Allingham NW Surrey CCG February 18, 2015 Dear Ms
0.9% Sodium Chloride Injection, USP In VIAFLEX Plastic Container
 Page 1 of 8 PRESCRIBING INFORMATION 0.9% Sodium Chloride Injection, USP In VIAFLEX Plastic Container IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of
Page 1 of 8 PRESCRIBING INFORMATION 0.9% Sodium Chloride Injection, USP In VIAFLEX Plastic Container IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of
VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension
 VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension DESCRIPTION Hydroxyzine pamoate is designated chemically as 1-(p-chlorobenzhydryl) 4- [2-(2-hydroxyethoxy) ethyl] diethylenediamine salt of 1,1
VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension DESCRIPTION Hydroxyzine pamoate is designated chemically as 1-(p-chlorobenzhydryl) 4- [2-(2-hydroxyethoxy) ethyl] diethylenediamine salt of 1,1
PACKAGE LEAFLET: INFORMATION FOR THE USER. VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution. Cyanocobalamin
 PACKAGE LEAFLET: INFORMATION FOR THE USER VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution Cyanocobalamin Read all of this leaflet carefully before you start using this medicine because
PACKAGE LEAFLET: INFORMATION FOR THE USER VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution Cyanocobalamin Read all of this leaflet carefully before you start using this medicine because
FULL PRESCRIBING INFORMATION
 HIGHLIGHTS F PRESCRIBING INFRMATIN These highlights do not include all the information needed to use AzaSite safely and effectively. See full prescribing information for AzaSite. AzaSite (azithromycin
HIGHLIGHTS F PRESCRIBING INFRMATIN These highlights do not include all the information needed to use AzaSite safely and effectively. See full prescribing information for AzaSite. AzaSite (azithromycin
3% Sodium Chloride Injection, USP 5% Sodium Chloride Injection, USP
 PRESCRIBING INFORMATION 3% Sodium Chloride Injection, USP 5% Sodium Chloride Injection, USP IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of Revision:
PRESCRIBING INFORMATION 3% Sodium Chloride Injection, USP 5% Sodium Chloride Injection, USP IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of Revision:
Nursing 113. Pharmacology Principles
 Nursing 113 Pharmacology Principles 1. The study of how drugs enter the body, reach the site of action, and are removed from the body is called a. pharmacotherapeutics b. pharmacology c. pharmacodynamics
Nursing 113 Pharmacology Principles 1. The study of how drugs enter the body, reach the site of action, and are removed from the body is called a. pharmacotherapeutics b. pharmacology c. pharmacodynamics
used to treat inflammation, corneal injury and bacterial infections in the external part of the eye.
 TOBRADEX * Eye Drops Tobramycin and Dexamethasone CONSUMER MEDICINE INFORMATION What is in this Leaflet This leaflet answers some common questions about TOBRADEX* Eye Drops. It does not contain all the
TOBRADEX * Eye Drops Tobramycin and Dexamethasone CONSUMER MEDICINE INFORMATION What is in this Leaflet This leaflet answers some common questions about TOBRADEX* Eye Drops. It does not contain all the
Guidance for Industry Safety Testing of Drug Metabolites
 Guidance for Industry Safety Testing of Drug Metabolites U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2008 Pharmacology
Guidance for Industry Safety Testing of Drug Metabolites U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2008 Pharmacology
EFFIMET 1000 XR Metformin Hydrochloride extended release tablet
 BRAND NAME: Effimet XR. THERAPEUTIC CATEGORY: Anti-Diabetic PHARMACOLOGIC CLASS: Biguanides EFFIMET 1000 XR Metformin Hydrochloride extended release tablet COMPOSITION AND PRESENTATION Composition Each
BRAND NAME: Effimet XR. THERAPEUTIC CATEGORY: Anti-Diabetic PHARMACOLOGIC CLASS: Biguanides EFFIMET 1000 XR Metformin Hydrochloride extended release tablet COMPOSITION AND PRESENTATION Composition Each
NIZORAL (KETOCONAZOLE) 2% SHAMPOO DESCRIPTION
 NIZORAL (KETOCONAZOLE) 2% SHAMPOO DESCRIPTION NIZORAL (ketoconazole) 2% Shampoo is a red-orange liquid for topical application, containing the broad spectrum synthetic antifungal agent ketoconazole in
NIZORAL (KETOCONAZOLE) 2% SHAMPOO DESCRIPTION NIZORAL (ketoconazole) 2% Shampoo is a red-orange liquid for topical application, containing the broad spectrum synthetic antifungal agent ketoconazole in
Drug Coding and CPT (Current Procedural Terminology) Guidance
 Aridol (mannitol inhalation powder) Bronchial Challenge Test Kit NDC 44178-0552-1 Drug Coding and CPT (Current Procedural Terminology) Guidance Product Reimbursement Code: HCPCS Code 1 Description 1 J7665
Aridol (mannitol inhalation powder) Bronchial Challenge Test Kit NDC 44178-0552-1 Drug Coding and CPT (Current Procedural Terminology) Guidance Product Reimbursement Code: HCPCS Code 1 Description 1 J7665
NONCLINICAL EVALUATION FOR ANTICANCER PHARMACEUTICALS
 INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE NONCLINICAL EVALUATION FOR ANTICANCER PHARMACEUTICALS
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE NONCLINICAL EVALUATION FOR ANTICANCER PHARMACEUTICALS
PARACETAMOL REXIDOL. 600 mg Tablet. Analgesic-Antipyretic. Paracetamol 600 mg
 (Insert Text) UL Consumer Health PARACETAMOL REXIDOL 600 mg Tablet Analgesic-Antipyretic FORMULATION Each tablet contains: Paracetamol 600 mg PRODUCT DESCRIPTION Rexidol is a round, yellow, flat, bevel-edged
(Insert Text) UL Consumer Health PARACETAMOL REXIDOL 600 mg Tablet Analgesic-Antipyretic FORMULATION Each tablet contains: Paracetamol 600 mg PRODUCT DESCRIPTION Rexidol is a round, yellow, flat, bevel-edged
VOLTAREN OPHTHA EYE DROPS
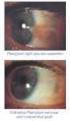 2013 במאי משרד הבריאות ותוכנו נבדק ואושר על ידו ע"י עלון זה נקבע ע פורמט PRESCRIBING INFORMATION VOLTAREN OPHTHA EYE DROPS 1 Name of the medicinal product Voltaren Ophtha Eye Drops. 2 Qualitative and quantitative
2013 במאי משרד הבריאות ותוכנו נבדק ואושר על ידו ע"י עלון זה נקבע ע פורמט PRESCRIBING INFORMATION VOLTAREN OPHTHA EYE DROPS 1 Name of the medicinal product Voltaren Ophtha Eye Drops. 2 Qualitative and quantitative
PLEGRIDY (peginterferon beta-1a) injection, for subcutaneous injection Initial U.S. Approval: 2014
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PLEGRIDY safely and effectively. See full prescribing information for PLEGRIDY. PLEGRIDY (peginterferon
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PLEGRIDY safely and effectively. See full prescribing information for PLEGRIDY. PLEGRIDY (peginterferon
MATERIAL SAFETY DATA SHEET
 Page 1 of 5 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 5 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Section 4. Toxicology
 Section 4 Toxicology Occupational Health Any chemical you use incorrectly can result in over-exposure leading to adverse health effects immediately or in the future. All hazardous materials can be handled
Section 4 Toxicology Occupational Health Any chemical you use incorrectly can result in over-exposure leading to adverse health effects immediately or in the future. All hazardous materials can be handled
LEFLUNOMIDE (Adults)
 Shared Care Guideline DRUG: Introduction: LEFLUNOMIDE (Adults) Indication: Disease modifying drug for rheumatoid arthritis and psoriatic arthritis Licensing Information: Disease modifying drug for active
Shared Care Guideline DRUG: Introduction: LEFLUNOMIDE (Adults) Indication: Disease modifying drug for rheumatoid arthritis and psoriatic arthritis Licensing Information: Disease modifying drug for active
Material Safety data sheet
 EMERGENCY OVERVIEW METFORMIN HYDROCHLORIDE TABLET contain Metformin and excipients generally considered to be non- toxic and non-hazardous in small quantities and under conditions of normal occupational
EMERGENCY OVERVIEW METFORMIN HYDROCHLORIDE TABLET contain Metformin and excipients generally considered to be non- toxic and non-hazardous in small quantities and under conditions of normal occupational
Calcium Folinate Ebewe Data Sheet
 NAME OF THE MEDICINE Calcium folinate injection Composition Active: Calcium folinate (equivalent to 10 mg folinic acid per ml) Inactive: Sodium chloride (7.7mg/mL), qs Water for Injections. Preservative
NAME OF THE MEDICINE Calcium folinate injection Composition Active: Calcium folinate (equivalent to 10 mg folinic acid per ml) Inactive: Sodium chloride (7.7mg/mL), qs Water for Injections. Preservative
Public Assessment Report Scientific discussion. Tenofovir disoproxil Teva (tenofovir disoproxil) SE/H/1432/01/DC
 Public Assessment Report Scientific discussion Tenofovir disoproxil Teva (tenofovir disoproxil) SE/H/1432/01/DC This module reflects the scientific discussion for the approval of Tenofovir disoproxil Teva.
Public Assessment Report Scientific discussion Tenofovir disoproxil Teva (tenofovir disoproxil) SE/H/1432/01/DC This module reflects the scientific discussion for the approval of Tenofovir disoproxil Teva.
MATERIAL SAFETY DATA SHEET
 Page 1 of 5 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 5 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Naloxone Hydrochloride Injection PRODUCT INFORMATION
 Naloxone Hydrochloride Injection PRODUCT INFORMATION DESCRIPTION Naloxone hydrochloride is 17-allyl-4,5α-epoxy-3,14-dihydroxymorphinan-6-one hydrochloride; C 19 H 21 NO 4.HCl. It is an off-white powder
Naloxone Hydrochloride Injection PRODUCT INFORMATION DESCRIPTION Naloxone hydrochloride is 17-allyl-4,5α-epoxy-3,14-dihydroxymorphinan-6-one hydrochloride; C 19 H 21 NO 4.HCl. It is an off-white powder
RADIOPHARMACEUTICALS BASED ON MONOCLONAL ANTIBODIES
 RADIOPHARMACEUTICALS BASED ON MONOCLONAL ANTIBODIES Guideline Title Radiopharmaceuticals based on Monoclonal Antibodies Legislative basis Directives 65/65/EEC, 75/318/EEC as amended, Directive 89/343/EEC
RADIOPHARMACEUTICALS BASED ON MONOCLONAL ANTIBODIES Guideline Title Radiopharmaceuticals based on Monoclonal Antibodies Legislative basis Directives 65/65/EEC, 75/318/EEC as amended, Directive 89/343/EEC
ESBRIET (pirfenidone) capsules, for oral use Initial U.S. Approval: 2014
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ESBRIET safely and effectively. See full prescribing information for ESBRIET. ESBRIET (pirfenidone)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ESBRIET safely and effectively. See full prescribing information for ESBRIET. ESBRIET (pirfenidone)
RISK EVALUATION AND MITIGATION STRATEGY (REMS)
 Initial REMS Approval: 9/2010 Most Recent Modification: 05/2015 0.5mg capsules Sphingosine 1-phosphate Receptor Modulator Novartis Pharmaceuticals Corporation One Health Plaza East Hanover, NJ 07936 RISK
Initial REMS Approval: 9/2010 Most Recent Modification: 05/2015 0.5mg capsules Sphingosine 1-phosphate Receptor Modulator Novartis Pharmaceuticals Corporation One Health Plaza East Hanover, NJ 07936 RISK
Mucodyne-Clear 250 mg/5 ml Syrup PL 04425/0665
 Mucodyne-Clear 250 mg/5 ml Syrup PL 04425/0665 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
Mucodyne-Clear 250 mg/5 ml Syrup PL 04425/0665 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
ORAL MEDICATIONS FOR MS! Gilenya and Aubagio
 ORAL MEDICATIONS FOR MS! Gilenya and Aubagio Champions against MS 4/20/13 Alexandra Goodyear, MD Stanford University Oral Medications Since 2010, 3 new oral medications for MS: Gilenya 2010 Aubagio 2012
ORAL MEDICATIONS FOR MS! Gilenya and Aubagio Champions against MS 4/20/13 Alexandra Goodyear, MD Stanford University Oral Medications Since 2010, 3 new oral medications for MS: Gilenya 2010 Aubagio 2012
SR 5. W2364A_NT.XATRAL SR5 PAK/SA 5/12/05 9:47 Page 1
 W2364A_NT.XATRAL SR5 PAK/SA 5/12/05 9:47 Page 1 alfuzosin SR 5 mg (Alfuzosin HCI) COMPOSITION Each sustained-release, film-coated tablet contains: Alfuzosin hydrochloride......... 5 mg Excipients... q.s.
W2364A_NT.XATRAL SR5 PAK/SA 5/12/05 9:47 Page 1 alfuzosin SR 5 mg (Alfuzosin HCI) COMPOSITION Each sustained-release, film-coated tablet contains: Alfuzosin hydrochloride......... 5 mg Excipients... q.s.
PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement)
 1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
MATERIAL SAFETY DATA SHEET
 MATERIAL SAFETY DATA SHEET 1. IDENTIFICATION OF THE SUBSTANCE AND THE COMPANY Material Manufacturer Distributor Losartan Potassium and Hydrochlorothiazide Tablets 100 mg/12.5 mg Lupin Limited Goa 403 722
MATERIAL SAFETY DATA SHEET 1. IDENTIFICATION OF THE SUBSTANCE AND THE COMPANY Material Manufacturer Distributor Losartan Potassium and Hydrochlorothiazide Tablets 100 mg/12.5 mg Lupin Limited Goa 403 722
SECONDARY GLAUCOMA: Pseudoexfoliation (PXF), Pigmentary Dispersion Syndrome (PDS), Neovascular (NV), Uveitic
 SECONDARY GLAUCOMA: Pseudoexfoliation (PXF), Pigmentary Dispersion Syndrome (PDS), Neovascular (NV), Uveitic Many roads can lead to glaucoma. With the exception of primary open-angle glaucoma (POAG) and
SECONDARY GLAUCOMA: Pseudoexfoliation (PXF), Pigmentary Dispersion Syndrome (PDS), Neovascular (NV), Uveitic Many roads can lead to glaucoma. With the exception of primary open-angle glaucoma (POAG) and
Perrigo Pharmaceuticals Co.
 Ibuprofen Page 1 of 7 Perrigo Pharmaceuticals Co. U.S. Pharmaceuticals Division MATERIAL SAFETY DATA SHEET NDC Product(s) Code(s): 0121-4774-05 0121-4774-10 Product Name: Ibuprofen Oral Suspension 100mg/5mL
Ibuprofen Page 1 of 7 Perrigo Pharmaceuticals Co. U.S. Pharmaceuticals Division MATERIAL SAFETY DATA SHEET NDC Product(s) Code(s): 0121-4774-05 0121-4774-10 Product Name: Ibuprofen Oral Suspension 100mg/5mL
Current version dated 5 March 2012
 M3(R2) Implementation Working Group M3(R2) Guideline: Guidance on Nonclinical Safety Studies for the Conduct of Human Clinical Trials and Marketing Authorization for Pharmaceuticals Questions & Answers
M3(R2) Implementation Working Group M3(R2) Guideline: Guidance on Nonclinical Safety Studies for the Conduct of Human Clinical Trials and Marketing Authorization for Pharmaceuticals Questions & Answers
Public Assessment Report. Scientific discussion. Apotel 10 mg/ml, solution for infusion. (paracetamol) NL/H/2857/001/DC
 Public Assessment Report Scientific discussion Apotel 10 mg/ml, solution for infusion (paracetamol) NL/H/2857/001/DC Date: 16 December 2014 This module reflects the scientific discussion for the approval
Public Assessment Report Scientific discussion Apotel 10 mg/ml, solution for infusion (paracetamol) NL/H/2857/001/DC Date: 16 December 2014 This module reflects the scientific discussion for the approval
45813E/Revised: April 2008 CYANOCOBALAMIN INJECTION, USP
 45813E/Revised: April 2008 CYANOCOBALAMIN INJECTION, USP DESCRIPTION: Cyanocobalamin Injection, USP is a sterile solution of cyanocobalamin for intramuscular or subcutaneous use. Each ml contains 1000
45813E/Revised: April 2008 CYANOCOBALAMIN INJECTION, USP DESCRIPTION: Cyanocobalamin Injection, USP is a sterile solution of cyanocobalamin for intramuscular or subcutaneous use. Each ml contains 1000
Drug Excretion. Renal Drug Clearance. Drug Clearance and Half-Life. Glomerular Filtration II. Glomerular Filtration I. Drug Excretion and Clearance
 t/.drugexcretion AINTRAVENOUSDOSE 36848765430TIME(hours) t/ Drug Excretion Dr. Robert G. Lamb Professor Pharmacology & Toxicology Drug Excretion and Clearance Drug Excretion: is the movement of drug from
t/.drugexcretion AINTRAVENOUSDOSE 36848765430TIME(hours) t/ Drug Excretion Dr. Robert G. Lamb Professor Pharmacology & Toxicology Drug Excretion and Clearance Drug Excretion: is the movement of drug from
ALLERGENIC EXTRACT. Prescription Set of Serial Dilutions (or Maintenance Vial (s)) INSTRUCTIONS FOR USE. U.S. Government License No.
 ALLERGENIC EXTRACT Prescription Set of Serial Dilutions (or Maintenance Vial (s)) INSTRUCTIONS FOR USE U.S. Government License No. 308 Revised 07/04 PO Box 800 Lenoir, NC 28645 USA DESCRIPTION This set
ALLERGENIC EXTRACT Prescription Set of Serial Dilutions (or Maintenance Vial (s)) INSTRUCTIONS FOR USE U.S. Government License No. 308 Revised 07/04 PO Box 800 Lenoir, NC 28645 USA DESCRIPTION This set
Who are Levonorgestrel Tablets, 0.75mg for? Other things your patients should know: When are Levonorgestrel Tablets, 0.75mg effective?
 One-hour ACPE accredited continuing education program is available at no charge for pharmacists provided by the University of Illinois-Chicago College of Pharmacy at www.pharmacyce.uic.edu Accidents happen...
One-hour ACPE accredited continuing education program is available at no charge for pharmacists provided by the University of Illinois-Chicago College of Pharmacy at www.pharmacyce.uic.edu Accidents happen...
September 19, 1984 FOOD PRODUCTION AND DIRECTION GÉNÉRALE, SECTION INSPECTION BRANCH PRODUCTION ET INSPECTION PESTICIDES DES ALIMENTS TRADE MEMORANDUM
 Agriculture Canada September 19, 1984 T-1-245 FOOD PRODUCTION AND DIRECTION GÉNÉRALE, SECTION INSPECTION BRANCH PRODUCTION ET INSPECTION PESTICIDES DES ALIMENTS TRADE MEMORANDUM RE: Guidelines for Developing
Agriculture Canada September 19, 1984 T-1-245 FOOD PRODUCTION AND DIRECTION GÉNÉRALE, SECTION INSPECTION BRANCH PRODUCTION ET INSPECTION PESTICIDES DES ALIMENTS TRADE MEMORANDUM RE: Guidelines for Developing
Beaumont Hospital Department of Nephrology and Renal Nursing. Guideline for administering Ferinject
 Beaumont Hospital Department of Nephrology and Renal Nursing Guideline Name: Guideline for administering Ferinject Guideline Number: 18 Guideline Version: a Developed By: Louise Kelly CNM 1 Renal Day Care
Beaumont Hospital Department of Nephrology and Renal Nursing Guideline Name: Guideline for administering Ferinject Guideline Number: 18 Guideline Version: a Developed By: Louise Kelly CNM 1 Renal Day Care
MATERIAL SAFETY DATA SHEET
 Page 1 of 6 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 6 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Guidance for Industry
 Guidance for Industry S9 Nonclinical Evaluation for Anticancer Pharmaceuticals U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) Center
Guidance for Industry S9 Nonclinical Evaluation for Anticancer Pharmaceuticals U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) Center
Clinical Study Synopsis for Public Disclosure
 abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis - which is part of the clinical
abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis - which is part of the clinical
IMPORTANT DRUG WARNING Regarding Mycophenolate-Containing Products
 Dear Healthcare Provider: Mycophenolate REMS (Risk Evaluation and Mitigation Strategy) has been mandated by the FDA (Food and Drug Administration) due to postmarketing reports showing that exposure to
Dear Healthcare Provider: Mycophenolate REMS (Risk Evaluation and Mitigation Strategy) has been mandated by the FDA (Food and Drug Administration) due to postmarketing reports showing that exposure to
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Prednicare Tablets 5mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains: Active Substance(s) Prednisolone
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Prednicare Tablets 5mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains: Active Substance(s) Prednisolone
SUMMARY OF PRODUCT CHARACTERISTICS. Albuman 200 g/l is a solution containing 200 g/l (20%) of total protein of which at least 95% is human albumin.
 Albuman 200 g/l SPC 01 December 2015 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Albuman 200 g/l solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Albuman 200 g/l
Albuman 200 g/l SPC 01 December 2015 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Albuman 200 g/l solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Albuman 200 g/l
How To Treat Eye Problems With A Laser
 1550 Oak St., Suite 5 1515 Oak St., St Eugene, OR 97401 Eugene, OR 97401 (541) 687-2110 (541) 344-2010 INFORMED CONSENT FOR PHOTOREFRACTIVE KERATECTOMY (PRK) This information is to help you make an informed
1550 Oak St., Suite 5 1515 Oak St., St Eugene, OR 97401 Eugene, OR 97401 (541) 687-2110 (541) 344-2010 INFORMED CONSENT FOR PHOTOREFRACTIVE KERATECTOMY (PRK) This information is to help you make an informed
Emergency Telephone (800) 551-7176
 MATERIAL SAFETY DATA SHEET SECTION 1 - PRODUCT & COMPANY IDENTIFICATION 1501 East Woodfield Road Suite 300 East Schaumburg, IL 60173 (847) 969-2700 Product Identifier: Chemical Name: Category: Mesna Detoxifying
MATERIAL SAFETY DATA SHEET SECTION 1 - PRODUCT & COMPANY IDENTIFICATION 1501 East Woodfield Road Suite 300 East Schaumburg, IL 60173 (847) 969-2700 Product Identifier: Chemical Name: Category: Mesna Detoxifying
Summary of the risk management plan (RMP) for Orkambi (lumacaftor and ivacaftor)
 EMA/662624/2015 Summary of the risk management plan (RMP) for Orkambi (lumacaftor and ivacaftor) This is a summary of the risk management plan (RMP) for Orkambi, which details the measures to be taken
EMA/662624/2015 Summary of the risk management plan (RMP) for Orkambi (lumacaftor and ivacaftor) This is a summary of the risk management plan (RMP) for Orkambi, which details the measures to be taken
CAPASTAT SULFATE CAPREOMYCIN FOR INJECTION, USP
 1 CAPASTAT SULFATE CAPREOMYCIN FOR INJECTION, USP PV 4801 AMP FOR INTRAMUSCULAR AND INTRAVENOUS INFUSION ONLY NOT FOR PEDIATRIC USE WARNINGS The use of Capastat Sulfate (Capreomycin for Injection, USP)
1 CAPASTAT SULFATE CAPREOMYCIN FOR INJECTION, USP PV 4801 AMP FOR INTRAMUSCULAR AND INTRAVENOUS INFUSION ONLY NOT FOR PEDIATRIC USE WARNINGS The use of Capastat Sulfate (Capreomycin for Injection, USP)
Decentralised Procedure. Public Assessment Report. Paracetamol-ratiopharm 10 mg/ml Infusionslösung Paracetamol DE/H/1908/001/DC
 Bundesinstitut für Arzneimittel und Medizinprodukte Decentralised Procedure Public Assessment Report Paracetamol-ratiopharm 10 mg/ml Infusionslösung Paracetamol DE/H/1908/001/DC Applicant: TEVA Pharma
Bundesinstitut für Arzneimittel und Medizinprodukte Decentralised Procedure Public Assessment Report Paracetamol-ratiopharm 10 mg/ml Infusionslösung Paracetamol DE/H/1908/001/DC Applicant: TEVA Pharma
Revised 04/2015 FULL PRESCRIBING INFORMATION: CONTENTS*
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use KYBELLA safely and effectively. See full prescribing information for KYBELLA. KYBELLA (deoxycholic
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use KYBELLA safely and effectively. See full prescribing information for KYBELLA. KYBELLA (deoxycholic
Package leaflet: Information for the patient. Dorzolamide Mylan 20 mg/ml Eye drops, Solution Dorzolamide
 Package leaflet: Information for the patient Dorzolamide Mylan 20 mg/ml Eye drops, Solution Dorzolamide Read all of this leaflet carefully before you start using this medicine because it contains important
Package leaflet: Information for the patient Dorzolamide Mylan 20 mg/ml Eye drops, Solution Dorzolamide Read all of this leaflet carefully before you start using this medicine because it contains important
ATROVENT HFA (ipratropium bromide HFA) Inhalation Aerosol FOR ORAL INHALATION ONLY. Initial U.S. Approval: 2004
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ATROVENT HFA safely and effectively. See full prescribing information for ATROVENT HFA. ATROVENT
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ATROVENT HFA safely and effectively. See full prescribing information for ATROVENT HFA. ATROVENT
Cataracts. Cataract and Primary Eye Care Service...215-928-3041. Main Number...215-928-3000. Physician Referral...1-877-AT-WILLS 1-877-289-4557
 Main Number...215-928-3000 Physician Referral...1-877-AT-WILLS 1-877-289-4557 Emergency Service...215-503-8080 Cataract and Primary Eye Care Service...215-928-3041 Retina Service... 215-928-3300 Cataract
Main Number...215-928-3000 Physician Referral...1-877-AT-WILLS 1-877-289-4557 Emergency Service...215-503-8080 Cataract and Primary Eye Care Service...215-928-3041 Retina Service... 215-928-3300 Cataract
SUMMARY OF PRODUCT CHARACTERISTICS. Paracetamol...10.00 mg for 1 ml of solution for infusion
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Paracetamol...10.00 mg for 1 ml of
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Paracetamol...10.00 mg for 1 ml of
Chemical Name: (±)-9-Fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7H-pyrido[1,2,3-de]-1,4-benzoxazine-6- carboxylic acid.
![Chemical Name: (±)-9-Fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7H-pyrido[1,2,3-de]-1,4-benzoxazine-6- carboxylic acid. Chemical Name: (±)-9-Fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7H-pyrido[1,2,3-de]-1,4-benzoxazine-6- carboxylic acid.](/thumbs/40/20977501.jpg) OCUFLOX (ofloxacin ophthalmic solution) 0.3% sterile DESCRIPTION OCUFLOX (ofloxacin ophthalmic solution) 0.3% is a sterile ophthalmic solution. It is a fluorinated carboxyquinolone anti-infective for topical
OCUFLOX (ofloxacin ophthalmic solution) 0.3% sterile DESCRIPTION OCUFLOX (ofloxacin ophthalmic solution) 0.3% is a sterile ophthalmic solution. It is a fluorinated carboxyquinolone anti-infective for topical
PATIENT INFORMATION BOOKLET
 (060110) VISIONCARE S IMPLANTABLE MINIATURE TELESCOPE ( BY DR. ISAAC LIPSHITZ ) AN INTRAOCULAR TELESCOPE FOR TREATING SEVERE TO PROFOUND VISION IMPAIRMENT DUE TO BILATERAL END-STAGE AGE-RELATED MACULAR
(060110) VISIONCARE S IMPLANTABLE MINIATURE TELESCOPE ( BY DR. ISAAC LIPSHITZ ) AN INTRAOCULAR TELESCOPE FOR TREATING SEVERE TO PROFOUND VISION IMPAIRMENT DUE TO BILATERAL END-STAGE AGE-RELATED MACULAR
Guidance for Industry and Review Staff Target Product Profile A Strategic Development Process Tool
 Guidance for Industry and Review Staff Product Profile A Strategic Development Process Tool DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions
Guidance for Industry and Review Staff Product Profile A Strategic Development Process Tool DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions
Animalgeiscs for Mice & Rats, 3.0 ml Vial Label: Carton Label:
 Animalgeiscs for Mice & Rats, 3.0 ml Vial Label: Carton Label: Animalgesics for Mice & Rats, 1.6 ml Vial Label: Carton Label: Animalgesics for Mice & Rats (buprenorphine extended-release injectable suspension)
Animalgeiscs for Mice & Rats, 3.0 ml Vial Label: Carton Label: Animalgesics for Mice & Rats, 1.6 ml Vial Label: Carton Label: Animalgesics for Mice & Rats (buprenorphine extended-release injectable suspension)
Lung Pathway Group Pemetrexed and Cisplatin in Non-Small Cell Lung Cancer (NSCLC)
 Indication: NICE TA181 First line treatment option in advanced or metastatic non-squamous NSCLC (histology confirmed as adenocarcinoma or large cell carcinoma) Performance status 0-1 Regimen details: Pemetrexed
Indication: NICE TA181 First line treatment option in advanced or metastatic non-squamous NSCLC (histology confirmed as adenocarcinoma or large cell carcinoma) Performance status 0-1 Regimen details: Pemetrexed
Naloxone Hydrochloride Injection, USP Opioid Antagonist
 Naloxone Hydrochloride Injection, USP Opioid Antagonist Rx Only DESCRIPTION Naloxone hydrochloride, an opioid antagonist, is a synthetic congener of oxymorphone. In structure it differs from oxymorphone
Naloxone Hydrochloride Injection, USP Opioid Antagonist Rx Only DESCRIPTION Naloxone hydrochloride, an opioid antagonist, is a synthetic congener of oxymorphone. In structure it differs from oxymorphone
Biological importance of metabolites. Safety and efficacy aspects
 Biological importance of metabolites Safety and efficacy aspects Bernard Walther Technologie Servier Biological importance of metabolites Safety testing of drug metabolites Bioanalytical strategy Structural
Biological importance of metabolites Safety and efficacy aspects Bernard Walther Technologie Servier Biological importance of metabolites Safety testing of drug metabolites Bioanalytical strategy Structural
PRODUCT INFORMATION (This PI contains all registered presentations of Avamys.) AVAMYS Nasal Spray
 PRODUCT INFORMATION (This PI contains all registered presentations of Avamys.) AVAMYS Nasal Spray NAME OF THE MEDICINE: Fluticasone furoate Structure: 21 F O 2 3 28 HO 19 1 10 5 4 O 20 S 18 12 O 11 13
PRODUCT INFORMATION (This PI contains all registered presentations of Avamys.) AVAMYS Nasal Spray NAME OF THE MEDICINE: Fluticasone furoate Structure: 21 F O 2 3 28 HO 19 1 10 5 4 O 20 S 18 12 O 11 13
Doxylamine succinate belongs to the ethanolamine class of antihistamines with sedative properties.
 Data Sheet MERSYNDOL Tablet Paracetamol 450mg per tablet Codeine Phosphate 9.75mg per tablet Doxylamine Succinate 5mg per tablet MERSYNDOL FORTE Tablet Paracetamol 450mg per tablet Codeine Phosphate 30mg
Data Sheet MERSYNDOL Tablet Paracetamol 450mg per tablet Codeine Phosphate 9.75mg per tablet Doxylamine Succinate 5mg per tablet MERSYNDOL FORTE Tablet Paracetamol 450mg per tablet Codeine Phosphate 30mg
GENERAL CONSIDERATIONS FOR CLINICAL TRIALS E8
 INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE GENERAL CONSIDERATIONS FOR CLINICAL TRIALS E8 Current
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE GENERAL CONSIDERATIONS FOR CLINICAL TRIALS E8 Current
PACKAGE LEAFLET: INFORMATION FOR THE USER. ADRENALINE (HCl) STEROP 0,8mg/1ml. Solution for injection. Adrenaline (Levorenine, Epinephrine)
 PACKAGE LEAFLET: INFORMATION FOR THE USER ADRENALINE (HCl) STEROP 0,4mg/1ml ADRENALINE (HCl) STEROP 0,8mg/1ml Solution for injection Adrenaline (Levorenine, Epinephrine) Read all of this leaflet carefully
PACKAGE LEAFLET: INFORMATION FOR THE USER ADRENALINE (HCl) STEROP 0,4mg/1ml ADRENALINE (HCl) STEROP 0,8mg/1ml Solution for injection Adrenaline (Levorenine, Epinephrine) Read all of this leaflet carefully
Vitreo-Retinal and Macular Degeneration Frequently Asked Questions
 Vitreo-Retinal and Macular Degeneration Frequently Asked Questions What is a Vitreo-Retinal specialist? Retinal specialists are eye physicians and surgeons who focus on diseases in the back of the eye
Vitreo-Retinal and Macular Degeneration Frequently Asked Questions What is a Vitreo-Retinal specialist? Retinal specialists are eye physicians and surgeons who focus on diseases in the back of the eye
Session 6 Clinical Trial Assessment Phase I Clinical Trial
 L1 Session 6 Clinical Trial Assessment Phase I Clinical Trial Presentation to APEC Preliminary Workshop on Review of Drug Development in Clinical Trials Celia Lourenco, PhD, Manager, Clinical Group I Office
L1 Session 6 Clinical Trial Assessment Phase I Clinical Trial Presentation to APEC Preliminary Workshop on Review of Drug Development in Clinical Trials Celia Lourenco, PhD, Manager, Clinical Group I Office
MATERIAL SAFETY DATA SHEET
 Page 1 of 7 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 7 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
COMMITTEE FOR VETERINARY MEDICINAL PRODUCTS
 The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Veterinary Use EMEA/MRL/717/99-FINAL December 1999 COMMITTEE FOR VETERINARY MEDICINAL PRODUCTS TOLDIMFOS SUMMARY
The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Veterinary Use EMEA/MRL/717/99-FINAL December 1999 COMMITTEE FOR VETERINARY MEDICINAL PRODUCTS TOLDIMFOS SUMMARY
MATERIAL SAFETY DATA SHEET
 Page 1 of 6 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 6 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product)
 PART VI SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product) Format and content of the summary of the RMP The summary of the RMP part VI contains information based on RMP modules SI, SVIII and RMP
PART VI SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product) Format and content of the summary of the RMP The summary of the RMP part VI contains information based on RMP modules SI, SVIII and RMP
