DO YOU HAVE ITCHY ALLERGY EYES? Find out about lasting relief
|
|
|
- Lewis Pitts
- 9 years ago
- Views:
Transcription
1 DO YOU HAVE ITCHY ALLERGY EYES? Find out about lasting relief
2 Common causes of itching due to eye allergies include: Pollen from trees, grasses, and ragweed Dust mites Cat dander Approximately 10 million people are allergic to cat dander, the most common pet allergy.
3 How common is it? About 20% of people have eye allergies. For those who do, itchy eyes is a very common symptom. APPROVED USE LASTACAFT (alcaftadine ophthalmic solution) 0.25% is a prescription medicine approved for the prevention of itching associated with eye allergies. IMPORTANT SAFETY INFORMATION WARNINGS AND PRECAUTIONS To minimize contaminating the dropper tip of the bottle and solution, do not touch your eyelids or the areas around your eyes with the dropper tip. Keep bottle tightly closed when not in use. Please see more Important Safety Information on the following pages. OF PEOPLE WITH THIS TYPE OF EYE ALLERGY EXPERIENCE ITCHY EYES.
4 UNDERSTANDING ALLERGENS THAT CAUSE ITCHY EYES What are allergens? Allergens are what trigger your eye allergies. To the body, allergens are considered foreign substances that threaten or irritate the body, such as pollen from ragweed or cat dander. If you react to certain allergens, this means your eyes have become sensitized at some point. Therefore, when your eyes come into contact with specific allergens, an allergic response results. INDOOR ALLERGENS Examples of common indoor allergens that may cause itchy eyes are airborne cat dander and dust mites. OUTDOOR ALLERGENS Examples of common outdoor allergens that often trigger itchy eyes are grass, tree, and ragweed pollens. Eye allergies are grouped in various ways, one of which is where the allergens are located indoors or outdoors. Some allergens are more likely to be found indoors. Others you re more likely to be exposed to outdoors.
5 POLLEN MAP Common pollens in your area Pollens affect people at different times during the year. Visit Lastacaft.com to explore the interactive US Pollen Map. You will find out when common pollens are most likely to thrive in your area. IMPORTANT SAFETY INFORMATION (continued) WARNINGS AND PRECAUTIONS (continued) Do not wear a contact lens if your eye is red. LASTACAFT (alcaftadine ophthalmic solution) 0.25% should not be used to treat contact lens-related irritation. Please see more Important Safety Information on the following pages. SCAN CODE
6 ALLERGEN MANAGEMENT TIPS In the home When the outdoor air is filled with pollen, people with eye allergies often try to control their symptoms by staying indoors. But for people who are sensitive to indoor allergens, there are things you can do in the home that may help. IMPORTANT SAFETY INFORMATION (continued) WARNINGS AND PRECAUTIONS (continued) Remove contact lenses before putting LASTACAFT (alcaftadine ophthalmic solution) 0.25% in your eyes. The preservative in LASTACAFT may be absorbed by soft contact lenses. Lenses may be put back in your eyes 10 minutes after using LASTACAFT. LASTACAFT is for topical use in your eyes only. Please see more Important Safety Information on the following pages.
7 Clean your house Get rid of the allergen sources. Wearing a dust mask while cleaning can help reduce exposure to allergens Rid your bedroom of allergen sources Pay special attention to cleaning the bedroom you sleep in Vacuum at least once a week Use a vacuum with a HEPA filter or special double filter bags to avoid releasing allergens into the air. Use a damp or treated cloth for dusting and leave the house for several hours after cleaning it Keep windows and doors closed Prevent pollen entry Tips to reduce indoor allergens Avoid cat dander If you are allergic to cat dander, it is suggested not to keep pets in the house, and if you do, to keep pets out of the bedroom Eliminate dust mites Make sure surfaces in the home stay clean and tidy. Uncarpeted, hard floors are best throughout the home and especially in the bedroom. Put allergen-impermeable covers on all pillows and mattresses, which is even more effective than using air cleaners. Wash bedding weekly in hot water (130 F) to kill dust mites
8 How to treat itchy allergy eyes When your eyes itch because of eye allergies, one thing that matters is getting lasting relief. You can get that with LASTACAFT (alcaftadine ophthalmic solution) 0.25% a prescription eyedrop that works at 3 minutes and lasts all day; just one drop, once a day, helps you prevent itchy allergy eyes through a full 16 hours.
9 HOW LASTACAFT WORKS Itch prevention: LASTACAFT (alcaftadine ophthalmic solution) 0.25% interferes with a specific type of chemical process that causes itchy eyes in people with allergies Histamine blocking: LASTACAFT (alcaftadine ophthalmic solution) 0.25% blocks the release of histamine from certain cells IMPORTANT SAFETY INFORMATION (continued) SIDE EFFECTS The most common eye-related side effects that were reported in less than 4% of LASTACAFT (alcaftadine ophthalmic solution) 0.25% treated eyes were: eye irritation, burning and/or stinging in the eyes after use, eye redness, and eye itching. Please see more Important Safety Information on the following pages.
10 CLINICAL STUDY RESULTS LASTACAFT (alcaftadine ophthalmic solution) 0.25% effectively prevents itching due to eye allergies LASTACAFT ophthalmic solution was also evaluated for safety in a separate clinical study of 909 patients over 6 weeks. The most common eye-related side effects were reported in less than 4% of patients treated with LASTACAFT and included: Eye irritation Burning and/or stinging in the eyes after use Effectiveness Side Effects LASTACAFT (alcaftadine ophthalmic solution) 0.25% works fast (at 3 minutes) and prevents itchy eyes all day (through 16 hours) Eye redness Eye itching The most common non eye-related side effects were reported in less than 3% of patients treated with LASTACAFT ophthalmic solution and included: Inflammation of the nose and the upper part of the throat Headache The flu
11 Visit Lastacaft.com to learn about current savings offers IMPORTANT SAFETY INFORMATION (continued) SIDE EFFECTS (continued) The most common non eye-related side effects that were reported in less than 3% of patients with LASTACAFT (alcaftadine ophthalmic solution) 0.25% treated eyes were: inflammation of the nose and the upper part of the throat, headache, and the flu. Some of these side effects were similar to the symptoms of eye allergies. Please see accompanying full Prescribing Information.
12 Go to Lastacaft.com for more information APPROVED USE LASTACAFT (alcaftadine ophthalmic solution) 0.25% is a prescription medicine approved for the prevention of itching associated with eye allergies. IMPORTANT SAFETY INFORMATION WARNINGS AND PRECAUTIONS To minimize contaminating the dropper tip of the bottle and solution, do not touch your eyelids or the areas around your eyes with the dropper tip. Keep bottle tightly closed when not in use. Please see inside for more Important Safety Information. Please see accompanying full Prescribing Information Allergan, Inc., Irvine, CA marks owned by Allergan, Inc. APC42EQ
13 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LASTACAFT safely and effectively. See full prescribing information for LASTACAFT. LASTACAFT (alcaftadine ophthalmic solution) 0.25% Initial U.S. Approval: 2010 INDICATIONS AND USAGE LASTACAFT is an H 1 histamine receptor antagonist indicated for the prevention of itching associated with allergic conjunctivitis. (1) DOSAGE AND ADMINISTRATION Instill one drop in each eye once daily. (2) DOSAGE FORMS AND STRENGTHS Ophthalmic solution containing alcaftadine, 0.25% (2.5 mg/ml) (3) WARNINGS AND PRECAUTIONS To minimize the risk of contamination, do not touch dropper tip to any surface. Keep bottle tightly closed when not in use. (5.1) LASTACAFT should not be used to treat contact lens-related irritation. (5.2) Remove contact lenses prior to instillation of LASTACAFT. (5.2) ADVERSE REACTIONS The most common ocular adverse reactions, occurring in < 4% of LASTACAFT treated eyes, were eye irritation, burning and/or stinging on instillation, eye redness, and eye pruritus. (6.1) The most common non-ocular adverse reactions, occurring in < 3% of subjects with LASTACAFT treated eyes, were nasopharyngitis, headache and influenza. (6.2) To report SUSPECTED ADVERSE REACTIONS, contact Allergan at or FDA at FDA-1088 or See 17 for PATIENT COUNSELING INFORMATION Revised: 09/2011 FULL PRESCRIBING INFORMATION: CONTENTS* 1 INDICATIONS AND USAGE 2 DOSAGE AND ADMINISTRATION 3 DOSAGE FORMS AND STRENGTHS 4 CONTRAINDICATIONS 5 WARNINGS AND PRECAUTIONS 5.1 Contamination of Tip and Solution 5.2 Contact Lens Use 5.3 Topical Ophthalmic Use Only 6 ADVERSE REACTIONS 6.1 Ocular Adverse Reactions 6.2 Non-ocular Adverse Reactions 8 USE IN SPECIFIC POPULATIONS 8.1 Pregnancy 8.3 Nursing Mothers 8.4 Pediatric Use 8.5 Geriatric Use 11 DESCRIPTION 12 CLINICAL PHARMACOLOGY 12.1 Mechanism of Action 12.3 Pharmacokinetics 13 NONCLINICAL TOXICOLOGY 13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility 14 CLINICAL STUDIES 16 HOW SUPPLIED/STORAGE AND HANDLING 17 PATIENT COUNSELING INFORMATION 17.1 Sterility of Dropper Tip 17.2 Concomitant Use of Contact Lenses 17.3 Topical Ophthalmic Use Only * Sections or subsections omitted from the full prescribing information are not listed
14 FULL PRESCRIBING INFORMATION 1 INDICATIONS AND USAGE LASTACAFT is an H 1 histamine receptor antagonist indicated for the prevention of itching associated with allergic conjunctivitis. 2 DOSAGE AND ADMINISTRATION Instill one drop in each eye once daily. 3 DOSAGE FORMS AND STRENGTHS Topical ophthalmic solution containing alcaftadine, 0.25% (2.5 mg/ml). 4 CONTRAINDICATIONS None. 5 WARNINGS AND PRECAUTIONS 5.1 Contamination of Tip and Solution To minimize contaminating the dropper tip and solution, care should be taken not to touch the eyelids or surrounding areas with the dropper tip of the bottle. Keep bottle tightly closed when not in use. 5.2 Contact Lens Use Patients should be advised not to wear a contact lens if their eye is red. LASTACAFT should not be used to treat contact lens-related irritation. LASTACAFT should not be instilled while wearing contact lenses. Remove contact lenses prior to instillation of LASTACAFT. The preservative in LASTACAFT, benzalkonium chloride, may be absorbed by soft contact lenses. Lenses may be reinserted after 10 minutes following administration of LASTACAFT. 5.3 Topical Ophthalmic Use Only LASTACAFT is for topical ophthalmic use only. 6 ADVERSE REACTIONS Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice. 6.1 Ocular Adverse Reactions The most frequent ocular adverse reactions, occurring in < 4% of LASTACAFT treated eyes, were eye irritation, burning and/or stinging upon instillation, eye redness and eye pruritus. 6.2 Non-ocular Adverse Reactions The most frequent non-ocular adverse reactions, occurring in < 3% of subjects with LASTACAFT treated eyes, were nasopharyngitis, headache and influenza. Some of these events were similar to the underlying disease being studied. 8 USE IN SPECIFIC POPULATIONS 8.1 Pregnancy Pregnancy Category B. Reproduction studies performed in rats and rabbits revealed no evidence of impaired female reproduction or harm to the fetus due to alcaftadine. Oral doses in rats and rabbits of 20 and 80 mg/kg/day, respectively, produced plasma exposure levels approximately 200 and 9000 times the plasma exposure at the recommended human ocular dose. There are however, no adequate and well controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed. 8.3 Nursing Mothers It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when LASTACAFT is administered to a nursing woman. 8.4 Pediatric Use Safety and effectiveness in pediatric patients below the age of 2 years have not been established. 8.5 Geriatric Use No overall differences in safety or effectiveness were observed between elderly and younger subjects. 11 DESCRIPTION LASTACAFT is a sterile, topically administered H 1 receptor antagonist containing alcaftadine for ophthalmic use. Alcaftadine is a white to yellow powder with an empirical formula of C 19 H 21 N 3 O and a molecular weight of Contains: Active: alcaftadine 0.25% (2.5 mg/ml). Preservative: benzalkonium chloride 0.005%. Inactives: edetate disodium; sodium phosphate, monobasic; purified water; sodium chloride; sodium hydroxide; and/or hydrochloric acid to adjust ph Chemical Name: 6,11-dihydro-11-(1-methyl-4-piperidinylidene)-5H-imidazo[2,1-b] [3] benzazepine-3-carboxaldehyde Structural Formula: The drug product has a ph of approximately 7 and an osmolality of approximately 290 mosm/kg. 12 CLINICAL PHARMACOLOGY 12.1 Mechanism of Action Alcaftadine is an H 1 histamine receptor antagonist and inhibitor of the release of histamine from mast cells. Decreased chemotaxis and inhibition of eosinophil activation has also been demonstrated Pharmacokinetics Absorption Following bilateral topical ocular administration of alcaftadine ophthalmic solution, 0.25%, the mean plasma Cmax of alcaftadine was approximately 60 pg/ml and the median Tmax occurred at 15 minutes. Plasma concentrations of alcaftadine were below the lower limit of quantification (10 pg/ml) by 3 hours after dosing. The mean C max of the active carboxylic acid metabolite was approximately 3 ng/ml and occurred at 1 hour after dosing. Plasma concentrations of the carboxylic acid metabolite were below the lower limit of quantification (100 pg/ml) by 12 hours after dosing. There was no indication of systemic accumulation or changes in plasma exposure of alcaftadine or the active metabolite following daily topical ocular administration. Distribution The protein binding of alcaftadine and the active metabolite are 39.2% and 62.7%, respectively. Metabolism The metabolism of alcaftadine is mediated by non-cyp450 cytosolic enzymes to the active carboxylic acid metabolite. Excretion The elimination half-life of the carboxylic acid metabolite is approximately 2 hours following topical ocular administration. Based on data following oral administration of alcaftadine, the carboxylic acid metabolite is primarily eliminated unchanged in the urine. In vitro studies showed that neither alcaftadine nor the carboxylic acid metabolite substantially inhibited reactions catalyzed by major CYP450 enzymes. 13 NONCLINICAL TOXICOLOGY 13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility Alcaftadine was not mutagenic or genotoxic in the Ames test, the mouse lymphoma assay or the mouse micronucleus assay. Alcaftadine was found to have no effect on fertility of male and female rats at oral doses up to 20 mg/kg/day (approximately 200 times the plasma exposure at the recommended human ocular dose). 14 CLINICAL STUDIES Clinical efficacy was evaluated in conjunctival allergen challenge (CAC) studies. LASTACAFT was more effective than its vehicle in preventing ocular itching in patients with allergic conjunctivitis induced by an ocular allergen challenge, both at 3 minutes post-dosing and at 16 hours post-dosing of LASTACAFT. The safety of LASTACAFT was evaluated in a randomized clinical study of 909 subjects over a period of 6 weeks. 16 HOW SUPPLIED/STORAGE AND HANDLING LASTACAFT (alcaftadine ophthalmic solution) 0.25% is supplied in an opaque, white low-density polyethylene bottle with a white polypropylene cap. 3 ml fill in 5 ml bottle NDC Storage: Store at C (59-77 F). 17 PATIENT COUNSELING INFORMATION 17.1 Sterility of Dropper Tip Patients should be advised to not touch dropper tip to any surface, as this may contaminate the contents Concomitant Use of Contact Lenses Patients should be advised not to wear a contact lens if their eye is red. Patients should be advised that LASTACAFT should not be used to treat contact lens-related irritation. Patients should also be advised to remove contact lenses prior to instillation of LASTACAFT. The preservative in LASTACAFT, benzalkonium chloride, may be absorbed by soft contact lenses. Lenses may be reinserted after 10 minutes following administration of LASTACAFT Topical Ophthalmic Use only For topical ophthalmic administration only. Manufactured for Allergan, Inc., Irvine, CA 92612, U.S.A Allergan, Inc. marks owned by Allergan, Inc. Made in the U.S.A. Based on 72409US11C APC14EI11
Revised: 9/2014. LUMIGAN (bimatoprost ophthalmic solution) 0.01% for topical ophthalmic use. Initial U.S. Approval: 2001
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LUMIGAN (bimatoprost ophthalmic solution) 0.01% safely and effectively. See full prescribing information
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LUMIGAN (bimatoprost ophthalmic solution) 0.01% safely and effectively. See full prescribing information
NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate.
 NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate. PRESENTATION Eye Drops: NAPHCON-A Eye Drops are a combination of an antihistamine (pheniramine maleate) and a decongestant
NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate. PRESENTATION Eye Drops: NAPHCON-A Eye Drops are a combination of an antihistamine (pheniramine maleate) and a decongestant
FULL PRESCRIBING INFORMATION: CONTENTS*
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PHOTREXA VISCOUS and PHOTREXA safely and effectively. See full prescribing information for PHOTREXA
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PHOTREXA VISCOUS and PHOTREXA safely and effectively. See full prescribing information for PHOTREXA
See 17 for PATIENT COUNSELING INFORMATION. Revised: 3/2016 FULL PRESCRIBING INFORMATION: CONTENTS* WARNING: ADRENAL CRISIS IN THE SETTING OF SHOCK OR
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LYSODREN safely and effectively. See full prescribing information for LYSODREN. LYSODREN (mitotane)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LYSODREN safely and effectively. See full prescribing information for LYSODREN. LYSODREN (mitotane)
Conjunctivitis - Pink Eye
 Conjunctivitis - Pink Eye Introduction Conjunctivitis is often called pink eye. It causes inflammation of the thin, clear lining inside the eyelid and on the white of the eye. The inflammation gives the
Conjunctivitis - Pink Eye Introduction Conjunctivitis is often called pink eye. It causes inflammation of the thin, clear lining inside the eyelid and on the white of the eye. The inflammation gives the
Learn More About Product Labeling
 Learn More About Product Labeling Product label The product label is developed during the formal process of review and approval by regulatory agencies of any medicine or medical product. There are specific
Learn More About Product Labeling Product label The product label is developed during the formal process of review and approval by regulatory agencies of any medicine or medical product. There are specific
If you forget to use Catacrom Eye Drops If you forget to use this medicine, use it as soon as you remember.
 Package leaflet: Information for the user Catacrom 2 % w/v Eye Drops, Solution Sodium cromoglicate Read all of this leaflet carefully before you start using this medicine because it contains important
Package leaflet: Information for the user Catacrom 2 % w/v Eye Drops, Solution Sodium cromoglicate Read all of this leaflet carefully before you start using this medicine because it contains important
Guide to. Allergies 020 8742 7042. A guide to allergies
 If you need advice on taking over-the-counter medicines speak to a pharmacist or call the Consumer Health Information Centre s Over-the-Counter Medicines Advice Line on 020 8742 7042. The advice line is
If you need advice on taking over-the-counter medicines speak to a pharmacist or call the Consumer Health Information Centre s Over-the-Counter Medicines Advice Line on 020 8742 7042. The advice line is
Hypromellose Eye Drops BP 0.3% w/v PL 23097/0006
 Hypromellose Eye Drops BP 0.3% w/v PL 23097/0006 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
Hypromellose Eye Drops BP 0.3% w/v PL 23097/0006 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
Allergies: ENT and Allergy Center of Missouri YOUR GUIDE TO TESTING AND TREATMENT. University of Missouri Health Care
 Allergies: YOUR GUIDE TO TESTING AND TREATMENT ENT and Allergy Center of Missouri University of Missouri Health Care 812 N. Keene St., Columbia, MO 65201 (573) 817-3000 www.muhealth.org WHAT CAUSES ALLERGIES
Allergies: YOUR GUIDE TO TESTING AND TREATMENT ENT and Allergy Center of Missouri University of Missouri Health Care 812 N. Keene St., Columbia, MO 65201 (573) 817-3000 www.muhealth.org WHAT CAUSES ALLERGIES
Public Assessment Report. Decentralised Procedure
 Public Assessment Report Decentralised Procedure Olopatadine Zentiva 1 mg/ml eye drops, solution (olopatadine hydrochloride) Procedure No: UK Licence No: PL 17780/0568 Winthrop Pharmaceuticals UK Limited
Public Assessment Report Decentralised Procedure Olopatadine Zentiva 1 mg/ml eye drops, solution (olopatadine hydrochloride) Procedure No: UK Licence No: PL 17780/0568 Winthrop Pharmaceuticals UK Limited
Selected Requirements of Prescribing Information
 The Selected Requirements of Prescribing Information (SRPI) is a checklist of 42 important format prescribing information (PI) items based on labeling regulations [21 CFR 201.56(d) and 201.57] and guidances.
The Selected Requirements of Prescribing Information (SRPI) is a checklist of 42 important format prescribing information (PI) items based on labeling regulations [21 CFR 201.56(d) and 201.57] and guidances.
Allergy Shots and Allergy Drops for Adults and Children. A Review of the Research
 Allergy Shots and Allergy Drops for Adults and Children A Review of the Research Is This Information Right for Me? This information may be helpful to you if: Your doctor* has said that you or your child
Allergy Shots and Allergy Drops for Adults and Children A Review of the Research Is This Information Right for Me? This information may be helpful to you if: Your doctor* has said that you or your child
Teriflunomide is the active metabolite of Leflunomide, a drug employed since 1994 for the treatment of rheumatoid arthritis (Baselt, 2011).
 Page 1 of 10 ANALYTE NAME AND STRUCTURE TERIFLUNOMIDE Teriflunomide TRADE NAME Aubagio CATEGORY Antimetabolite TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND sclerosis. The
Page 1 of 10 ANALYTE NAME AND STRUCTURE TERIFLUNOMIDE Teriflunomide TRADE NAME Aubagio CATEGORY Antimetabolite TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND sclerosis. The
Seasonal Allergies. 1995-2012 The Patient Education Institute, Inc. www.x-plain.com im010101 Last reviewed: 05/30/2012 1
 Seasonal Allergies Introduction Seasonal allergies are allergies that develop during certain times of the year. Seasonal allergies are usually a response to pollen from trees, grasses, and weeds. Constant
Seasonal Allergies Introduction Seasonal allergies are allergies that develop during certain times of the year. Seasonal allergies are usually a response to pollen from trees, grasses, and weeds. Constant
ZOVIRAX Cold Sore Cream
 Data Sheet ZOVIRAX Cold Sore Cream Aciclovir 5% w/w Presentation Topical cream Indications ZOVIRAX Cold Sore Cream is indicated for the treatment of Herpes simplex virus infections of the lips and face
Data Sheet ZOVIRAX Cold Sore Cream Aciclovir 5% w/w Presentation Topical cream Indications ZOVIRAX Cold Sore Cream is indicated for the treatment of Herpes simplex virus infections of the lips and face
used to treat inflammation, corneal injury and bacterial infections in the external part of the eye.
 TOBRADEX * Eye Drops Tobramycin and Dexamethasone CONSUMER MEDICINE INFORMATION What is in this Leaflet This leaflet answers some common questions about TOBRADEX* Eye Drops. It does not contain all the
TOBRADEX * Eye Drops Tobramycin and Dexamethasone CONSUMER MEDICINE INFORMATION What is in this Leaflet This leaflet answers some common questions about TOBRADEX* Eye Drops. It does not contain all the
Take Action on Asthma. Environmental triggers of asthma and allergies
 Take Action on Asthma Environmental triggers of asthma and allergies What are asthma and allergies? They are both conditions where the body reacts to substances commonly found in the air. Asthma is a very
Take Action on Asthma Environmental triggers of asthma and allergies What are asthma and allergies? They are both conditions where the body reacts to substances commonly found in the air. Asthma is a very
Yoon Nofsinger, M.D. Tampa ENT Associates, 3450 East Fletcher Avenue, Tampa, FL 33613 Phone (813) 972-3353, Fax (813) 978 3667
 Yoon Nofsinger, M.D. Tampa ENT Associates, 3450 East Fletcher Avenue, Tampa, FL 33613 Phone (813) 972-3353, Fax (813) 978 3667 General Information: Allergies and Injection Therapy 1. General: You have
Yoon Nofsinger, M.D. Tampa ENT Associates, 3450 East Fletcher Avenue, Tampa, FL 33613 Phone (813) 972-3353, Fax (813) 978 3667 General Information: Allergies and Injection Therapy 1. General: You have
FULL PRESCRIBING INFORMATION
 HIGHLIGHTS F PRESCRIBING INFRMATIN These highlights do not include all the information needed to use AzaSite safely and effectively. See full prescribing information for AzaSite. AzaSite (azithromycin
HIGHLIGHTS F PRESCRIBING INFRMATIN These highlights do not include all the information needed to use AzaSite safely and effectively. See full prescribing information for AzaSite. AzaSite (azithromycin
FLIXONASE ALLERGY Non Drowsy Nasal Spray 24 hour Effective Relief and Prevention Available in 60 & 150 sprays
 FLIXONASE ALLERGY Non Drowsy Nasal Spray 24 hour Effective Relief and Prevention Available in 60 & 150 sprays CONSUMER MEDICINE INFORMATION WHAT IS IN THIS LEAFLET? Please read this leaflet carefully before
FLIXONASE ALLERGY Non Drowsy Nasal Spray 24 hour Effective Relief and Prevention Available in 60 & 150 sprays CONSUMER MEDICINE INFORMATION WHAT IS IN THIS LEAFLET? Please read this leaflet carefully before
A ragweed pollen as a treatment for a ragweed allergy? It s called immunotherapy.
 A ragweed pollen as a treatment for a ragweed allergy? It s called immunotherapy. RAGWITEK is a prescription medicine used for sublingual (under the tongue) immunotherapy to treat ragweed pollen allergies
A ragweed pollen as a treatment for a ragweed allergy? It s called immunotherapy. RAGWITEK is a prescription medicine used for sublingual (under the tongue) immunotherapy to treat ragweed pollen allergies
MATERIAL SAFETY DATA SHEET
 MATERIAL SAFETY DATA SHEET 1. IDENTIFICATION OF THE SUBSTANCE AND THE COMPANY Material Manufacturer Distributor Losartan Potassium and Hydrochlorothiazide Tablets 100 mg/12.5 mg Lupin Limited Goa 403 722
MATERIAL SAFETY DATA SHEET 1. IDENTIFICATION OF THE SUBSTANCE AND THE COMPANY Material Manufacturer Distributor Losartan Potassium and Hydrochlorothiazide Tablets 100 mg/12.5 mg Lupin Limited Goa 403 722
RAGWITEK TM (Short Ragweed Pollen Allergen Extract) Tablet for Sublingual Use Initial U.S. Approval: 2014
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use RAGWITEK safely and effectively. See full prescribing information for RAGWITEK. RAGWITEK TM (Short
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use RAGWITEK safely and effectively. See full prescribing information for RAGWITEK. RAGWITEK TM (Short
8.5 Geriatric Use * Sections or subsections omitted from the full prescribing information are not listed.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Injectafer safely and effectively. See full prescribing information for Injectafer. INJECTAFER (ferric
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Injectafer safely and effectively. See full prescribing information for Injectafer. INJECTAFER (ferric
FAQs on Influenza A (H1N1-2009) Vaccine
 FAQs on Influenza A (H1N1-2009) Vaccine 1) What is Influenza A (H1N1-2009) (swine flu) 1? Influenza A (H1N1-2009), previously known as "swine flu", is a new strain of influenza virus that spreads from
FAQs on Influenza A (H1N1-2009) Vaccine 1) What is Influenza A (H1N1-2009) (swine flu) 1? Influenza A (H1N1-2009), previously known as "swine flu", is a new strain of influenza virus that spreads from
PACKAGE LEAFLET: INFORMATION FOR THE USER. VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution. Cyanocobalamin
 PACKAGE LEAFLET: INFORMATION FOR THE USER VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution Cyanocobalamin Read all of this leaflet carefully before you start using this medicine because
PACKAGE LEAFLET: INFORMATION FOR THE USER VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution Cyanocobalamin Read all of this leaflet carefully before you start using this medicine because
Public Assessment Report. Decentralised Procedure
 Public Assessment Report Decentralised Procedure Opticrom Allergy Single Dose 2% w/v Eye Drops, Solution (sodium cromoglicate) Procedure No: UK Licence No: PL 04425/0688 Aventis Pharma Limited (T/A Sanofi-aventis
Public Assessment Report Decentralised Procedure Opticrom Allergy Single Dose 2% w/v Eye Drops, Solution (sodium cromoglicate) Procedure No: UK Licence No: PL 04425/0688 Aventis Pharma Limited (T/A Sanofi-aventis
Environmental Allergens. Allergies to Dust, Mold and Pollen. A Patient s Guide
 Environmental Allergens Allergies to Dust, Mold and Pollen A Patient s Guide Our environment, at home or outdoors, can be a potent source of allergens causing itchy, puffy and watery eyes, sneezing, a
Environmental Allergens Allergies to Dust, Mold and Pollen A Patient s Guide Our environment, at home or outdoors, can be a potent source of allergens causing itchy, puffy and watery eyes, sneezing, a
Pesticide Harmful Effects And Emergency Response
 Pesticide Harmful Effects And Emergency Response Most pesticides are designed to harm or kill pests. Because some pests have systems similar to the human system, some pesticides also can harm or kill humans.
Pesticide Harmful Effects And Emergency Response Most pesticides are designed to harm or kill pests. Because some pests have systems similar to the human system, some pesticides also can harm or kill humans.
Asthma Triggers. What are they and what can be done about them?
 Asthma Triggers What are they and what can be done about them? This brochure has been developed for the community by Asthma Australia It provides information about: Asthma triggers What you can do about
Asthma Triggers What are they and what can be done about them? This brochure has been developed for the community by Asthma Australia It provides information about: Asthma triggers What you can do about
MINISTRY OF HEALTH PANDEMIC INFLUENZA A / H1N1 2009 VACCINE FREQUENTLY ASKED QUESTIONS
 Government of the Republic of Trinidad and Tobago MINISTRY OF HEALTH PANDEMIC INFLUENZA A / H1N1 2009 VACCINE FREQUENTLY ASKED QUESTIONS Influenza vaccines are one of the most effective ways to protect
Government of the Republic of Trinidad and Tobago MINISTRY OF HEALTH PANDEMIC INFLUENZA A / H1N1 2009 VACCINE FREQUENTLY ASKED QUESTIONS Influenza vaccines are one of the most effective ways to protect
VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension
 VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension DESCRIPTION Hydroxyzine pamoate is designated chemically as 1-(p-chlorobenzhydryl) 4- [2-(2-hydroxyethoxy) ethyl] diethylenediamine salt of 1,1
VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension DESCRIPTION Hydroxyzine pamoate is designated chemically as 1-(p-chlorobenzhydryl) 4- [2-(2-hydroxyethoxy) ethyl] diethylenediamine salt of 1,1
Anaphylaxis: a severe, life threatening allergic reaction usually involving swelling, trouble breathing, and can progress to shock
 Allergy is a condition in which the immune system causes sneezing, itching, rashes, and wheezing, or sometimes even life-threatening allergic reactions. The more you know about allergies, the better prepared
Allergy is a condition in which the immune system causes sneezing, itching, rashes, and wheezing, or sometimes even life-threatening allergic reactions. The more you know about allergies, the better prepared
ALLERGENIC EXTRACT. Prescription Set of Serial Dilutions (or Maintenance Vial (s)) INSTRUCTIONS FOR USE. U.S. Government License No.
 ALLERGENIC EXTRACT Prescription Set of Serial Dilutions (or Maintenance Vial (s)) INSTRUCTIONS FOR USE U.S. Government License No. 308 Revised 07/04 PO Box 800 Lenoir, NC 28645 USA DESCRIPTION This set
ALLERGENIC EXTRACT Prescription Set of Serial Dilutions (or Maintenance Vial (s)) INSTRUCTIONS FOR USE U.S. Government License No. 308 Revised 07/04 PO Box 800 Lenoir, NC 28645 USA DESCRIPTION This set
IMPORTANT: PLEASE READ
 PART III: CONSUMER INFORMATION combined hepatitis A (inactivated) and hepatitis B (recombinant) vaccine This leaflet is part III of a three-part "Product Monograph" published when was approved for sale
PART III: CONSUMER INFORMATION combined hepatitis A (inactivated) and hepatitis B (recombinant) vaccine This leaflet is part III of a three-part "Product Monograph" published when was approved for sale
EMERGENCY TREATMENT OF ANAPHYLAXIS EPINEPHRINE AUTO-INJECTOR
 I. GENERAL GUIDELINES EMERGENCY TREATMENT OF ANAPHYLAXIS EPINEPHRINE AUTO-INJECTOR A. PURPOSE To counteract a severe allergic reaction (anaphylaxis) to a foreign substance as prescribed by the licensed
I. GENERAL GUIDELINES EMERGENCY TREATMENT OF ANAPHYLAXIS EPINEPHRINE AUTO-INJECTOR A. PURPOSE To counteract a severe allergic reaction (anaphylaxis) to a foreign substance as prescribed by the licensed
Who are Levonorgestrel Tablets, 0.75mg for? Other things your patients should know: When are Levonorgestrel Tablets, 0.75mg effective?
 One-hour ACPE accredited continuing education program is available at no charge for pharmacists provided by the University of Illinois-Chicago College of Pharmacy at www.pharmacyce.uic.edu Accidents happen...
One-hour ACPE accredited continuing education program is available at no charge for pharmacists provided by the University of Illinois-Chicago College of Pharmacy at www.pharmacyce.uic.edu Accidents happen...
Reference ID: 3482803
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use EVZIO safely and effectively. See full prescribing information for EVZIO. EVZIO (naloxone hydrochloride
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use EVZIO safely and effectively. See full prescribing information for EVZIO. EVZIO (naloxone hydrochloride
BROCKTON AREA MULTI-SERVICES, INC. MEDICAL PROCEDURE GUIDE. Date(s) Reviewed/Revised:
 Page 1 of 5 PROCEDURE FOR: MAP-certified staff and RN/LPN MAP-certified staff are to be trained in the use of epinephrine administration via pre-filled autoinjector devices(s) annually. Certified staff
Page 1 of 5 PROCEDURE FOR: MAP-certified staff and RN/LPN MAP-certified staff are to be trained in the use of epinephrine administration via pre-filled autoinjector devices(s) annually. Certified staff
Patient Information Leaflet for Flixonase Aqueous Nasal Spray (fluticasone propionate)
 Patient Information Leaflet for Flixonase Aqueous Nasal Spray (fluticasone propionate) Your doctor has decided to prescribe Flixonase Aqueous Nasal Spray as part of your treatment. This leaflet tells you
Patient Information Leaflet for Flixonase Aqueous Nasal Spray (fluticasone propionate) Your doctor has decided to prescribe Flixonase Aqueous Nasal Spray as part of your treatment. This leaflet tells you
Chemical Name: (±)-9-Fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7H-pyrido[1,2,3-de]-1,4-benzoxazine-6- carboxylic acid.
![Chemical Name: (±)-9-Fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7H-pyrido[1,2,3-de]-1,4-benzoxazine-6- carboxylic acid. Chemical Name: (±)-9-Fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7H-pyrido[1,2,3-de]-1,4-benzoxazine-6- carboxylic acid.](/thumbs/40/20977501.jpg) OCUFLOX (ofloxacin ophthalmic solution) 0.3% sterile DESCRIPTION OCUFLOX (ofloxacin ophthalmic solution) 0.3% is a sterile ophthalmic solution. It is a fluorinated carboxyquinolone anti-infective for topical
OCUFLOX (ofloxacin ophthalmic solution) 0.3% sterile DESCRIPTION OCUFLOX (ofloxacin ophthalmic solution) 0.3% is a sterile ophthalmic solution. It is a fluorinated carboxyquinolone anti-infective for topical
Consumers. Allergic rhinitis. and your. Asthma. What you should know
 Consumers Allergic rhinitis and your Asthma What you should know Contents Allergic rhinitis facts...3 How allergic rhinitis can affect your asthma...3 What are the symptoms of allergic rhinitis?...4 What
Consumers Allergic rhinitis and your Asthma What you should know Contents Allergic rhinitis facts...3 How allergic rhinitis can affect your asthma...3 What are the symptoms of allergic rhinitis?...4 What
2 DOSAGE AND ADMINISTRATION 2.1 Dosing and Dose Adjustment
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Auryxia safely and effectively. See full prescribing information for Auryxia. Auryxia (ferric citrate)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Auryxia safely and effectively. See full prescribing information for Auryxia. Auryxia (ferric citrate)
EVERYTHING YOU WANTED TO KNOW ABOUT ALLERGIES
 EVERYTHING YOU WANTED TO KNOW ABOUT ALLERGIES I. ABOUT ALLERGIES A. What is an allergy? Allergies are hypersensitivity reactions of the body s immune system to specific substances we come in contact with
EVERYTHING YOU WANTED TO KNOW ABOUT ALLERGIES I. ABOUT ALLERGIES A. What is an allergy? Allergies are hypersensitivity reactions of the body s immune system to specific substances we come in contact with
Environmental Asthma Triggers Kit
 Environmental Asthma Triggers Kit Overview This is an activity used to demonstrate environmental asthma triggers and solutions 1-on-1 or with small audiences in the home or clinical environment. Learning
Environmental Asthma Triggers Kit Overview This is an activity used to demonstrate environmental asthma triggers and solutions 1-on-1 or with small audiences in the home or clinical environment. Learning
READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION. [new-ka la]
![READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION. [new-ka la] READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION. [new-ka la]](/thumbs/35/17235501.jpg) READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION Pr NUCALA [new-ka la] mepolizumab lyophilized powder for subcutaneous injection Read this carefully before you start
READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION Pr NUCALA [new-ka la] mepolizumab lyophilized powder for subcutaneous injection Read this carefully before you start
Stachybotrys chartarum a mold that may be found in water-damaged homes
 Stachybotrys chartarum a mold that may be found in water-damaged homes November 2000 Environmental Health Investigations Branch California Department of Health Services Stachybotrys chartarum ecology Stachybotrys
Stachybotrys chartarum a mold that may be found in water-damaged homes November 2000 Environmental Health Investigations Branch California Department of Health Services Stachybotrys chartarum ecology Stachybotrys
PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement)
 1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
Emergency Telephone (800) 551-7176
 MATERIAL SAFETY DATA SHEET SECTION 1 - PRODUCT & COMPANY IDENTIFICATION 1501 East Woodfield Road Suite 300 East Schaumburg, IL 60173 (847) 969-2700 Product Identifier: Chemical Name: Category: Mesna Detoxifying
MATERIAL SAFETY DATA SHEET SECTION 1 - PRODUCT & COMPANY IDENTIFICATION 1501 East Woodfield Road Suite 300 East Schaumburg, IL 60173 (847) 969-2700 Product Identifier: Chemical Name: Category: Mesna Detoxifying
Nursing 113. Pharmacology Principles
 Nursing 113 Pharmacology Principles 1. The study of how drugs enter the body, reach the site of action, and are removed from the body is called a. pharmacotherapeutics b. pharmacology c. pharmacodynamics
Nursing 113 Pharmacology Principles 1. The study of how drugs enter the body, reach the site of action, and are removed from the body is called a. pharmacotherapeutics b. pharmacology c. pharmacodynamics
Public Assessment Report. Pharmacy to General Sales List Reclassification. Pirinase Hayfever Relief for Adults 0.05% Nasal Spray.
 Public Assessment Report Pharmacy to General Sales List Reclassification Pirinase Hayfever Relief for Adults 0.05% Nasal Spray (Fluticasone) PL 00079/0688 Glaxo Wellcome UK Limited TABLE OF CONTENTS Introduction
Public Assessment Report Pharmacy to General Sales List Reclassification Pirinase Hayfever Relief for Adults 0.05% Nasal Spray (Fluticasone) PL 00079/0688 Glaxo Wellcome UK Limited TABLE OF CONTENTS Introduction
Food Allergies and. Food allergies and the immune system. Food allergies and the immune system
 Food Allergies and Intolerances Food allergies and the immune system Food allergies and True allergies involve an adverse reaction/ response by the body s immune system to a usually harmless substance
Food Allergies and Intolerances Food allergies and the immune system Food allergies and True allergies involve an adverse reaction/ response by the body s immune system to a usually harmless substance
Treatment of Asthma. Talk to your doctor about the various medications available to treat asthma.
 Please call 911 if you think you have a medical emergency. Treatment of Asthma The goals of asthma therapy are to prevent your child from having chronic and troublesome symptoms, to maintain your child's
Please call 911 if you think you have a medical emergency. Treatment of Asthma The goals of asthma therapy are to prevent your child from having chronic and troublesome symptoms, to maintain your child's
Immunotherapy For Control Of Allergies In Your Pets
 Immunotherapy For Control Of Allergies In Your Pets Pet owner information from The Quality Clinical Pathology Service What is Immunotherapy? Immunotherapy is a medical treatment where allergens (pollens,
Immunotherapy For Control Of Allergies In Your Pets Pet owner information from The Quality Clinical Pathology Service What is Immunotherapy? Immunotherapy is a medical treatment where allergens (pollens,
FOR ORAL OR RECTAL ADMINISTRATION FOR THE PREVENTION AND TREATMENT OF PORTAL-SYSTEMIC ENCEPHALOPATHY
 FORM NO. 1360 VC3074 Rev.2/08 LACTULOSE SOLUTION, USP 10 g/15 ml FOR ORAL OR RECTAL ADMINISTRATION FOR THE PREVENTION AND TREATMENT OF PORTAL-SYSTEMIC ENCEPHALOPATHY Rx Only DESCRIPTION Lactulose is a
FORM NO. 1360 VC3074 Rev.2/08 LACTULOSE SOLUTION, USP 10 g/15 ml FOR ORAL OR RECTAL ADMINISTRATION FOR THE PREVENTION AND TREATMENT OF PORTAL-SYSTEMIC ENCEPHALOPATHY Rx Only DESCRIPTION Lactulose is a
FDA Update on the H1N1 Flu Vaccine and Antiviral Medications
 FDA Update on the H1N1 Flu Vaccine and Antiviral Medications Beth Fabian Fritsch, R.Ph., M.B.A. Commander, U.S. Public Health Service Health Programs Coordinator Office of Special Health Issues Food and
FDA Update on the H1N1 Flu Vaccine and Antiviral Medications Beth Fabian Fritsch, R.Ph., M.B.A. Commander, U.S. Public Health Service Health Programs Coordinator Office of Special Health Issues Food and
MATERIAL SAFETY DATA SHEET
 Page 1 of 5 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 5 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
MATERIAL SAFETY DATA SHEET
 Page 1 of 6 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 6 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
VITAMIN C AND INFECTIOUS DISEASE: A REVIEW OF THE LITERATURE AND THE RESULTS OF A RANDOMIZED, DOUBLE-BLIND, PROSPECTIVE STUDY OVER 8 YEARS
 39 Chapter 3 VITAMIN C AND INFECTIOUS DISEASE: A REVIEW OF THE LITERATURE AND THE RESULTS OF A RANDOMIZED, DOUBLE-BLIND, PROSPECTIVE STUDY OVER 8 YEARS Maxine Briggs TABLE OF CONTENTS I. Review of the
39 Chapter 3 VITAMIN C AND INFECTIOUS DISEASE: A REVIEW OF THE LITERATURE AND THE RESULTS OF A RANDOMIZED, DOUBLE-BLIND, PROSPECTIVE STUDY OVER 8 YEARS Maxine Briggs TABLE OF CONTENTS I. Review of the
MATERIAL SAFETY DATA SHEET
 Page 1 of 7 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 7 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
VOLTAREN OPHTHA EYE DROPS
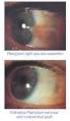 פורמט עלון זה נקבע ע"ימשרד הבריאות ותוכנו נבדק ואושר על ידו באפריל 2008 PRESCRIBING INFORMATION VOLTAREN OPHTHA EYE DROPS 1 Name of the medicinal product Voltaren Ophtha Eye Drops. 2 Qualitative and quantitative
פורמט עלון זה נקבע ע"ימשרד הבריאות ותוכנו נבדק ואושר על ידו באפריל 2008 PRESCRIBING INFORMATION VOLTAREN OPHTHA EYE DROPS 1 Name of the medicinal product Voltaren Ophtha Eye Drops. 2 Qualitative and quantitative
Analytical Specifications RIVAROXABAN
 Page 1 of 9 ANALYTE NAME AND STRUCTURE - RIVAROXABAN SYNONYMS Xarelto CATEGORY Anticoagulant TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND Xarelto (rivaroxaban) is an orally
Page 1 of 9 ANALYTE NAME AND STRUCTURE - RIVAROXABAN SYNONYMS Xarelto CATEGORY Anticoagulant TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND Xarelto (rivaroxaban) is an orally
IMPORTANT DRUG WARNING Regarding Mycophenolate-Containing Products
 Dear Healthcare Provider: Mycophenolate REMS (Risk Evaluation and Mitigation Strategy) has been mandated by the FDA (Food and Drug Administration) due to postmarketing reports showing that exposure to
Dear Healthcare Provider: Mycophenolate REMS (Risk Evaluation and Mitigation Strategy) has been mandated by the FDA (Food and Drug Administration) due to postmarketing reports showing that exposure to
Letter Date March 27, 2007 Stamp Date March 28, 2007 PDUFA Goal Date January 28, 2008
 CLINICAL REVIEW Application Type NDA Submission Number 22-157 Submission Code Letter Date March 27, 2007 Stamp Date March 28, 2007 PDUFA Goal Date January 28, 2008 Reviewer Name Review Completion Date
CLINICAL REVIEW Application Type NDA Submission Number 22-157 Submission Code Letter Date March 27, 2007 Stamp Date March 28, 2007 PDUFA Goal Date January 28, 2008 Reviewer Name Review Completion Date
NONCLINICAL EVALUATION FOR ANTICANCER PHARMACEUTICALS
 INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE NONCLINICAL EVALUATION FOR ANTICANCER PHARMACEUTICALS
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE NONCLINICAL EVALUATION FOR ANTICANCER PHARMACEUTICALS
There are several types of air cleaning devices available, each designed to remove certain types of pollutants.
 Please Note: EPA neither certifies nor recommends particular brands of home air cleaning devices. While some home air cleaning devices may be useful in some circumstances, EPA makes no broad endorsement
Please Note: EPA neither certifies nor recommends particular brands of home air cleaning devices. While some home air cleaning devices may be useful in some circumstances, EPA makes no broad endorsement
Lesson 3 Managing Food Allergies
 Lesson 3 Managing Food Allergies Lesson at a Glance Time Allowed (1 hour) 5 minutes Introduction to Managing Food Allergies 15 minutes Objective 1: Creating a Food Allergy Policy Topic Activity Materials
Lesson 3 Managing Food Allergies Lesson at a Glance Time Allowed (1 hour) 5 minutes Introduction to Managing Food Allergies 15 minutes Objective 1: Creating a Food Allergy Policy Topic Activity Materials
Sodium Sulphite Anhydrous
 Revision date 08-04-2016 1.1 Product identifier Sodium Sulphite Anhydrous Section 1: Identification of the substance/ mixture and of the company/ undertaking. Product name Other names Sodium Sulphite Anhydrous
Revision date 08-04-2016 1.1 Product identifier Sodium Sulphite Anhydrous Section 1: Identification of the substance/ mixture and of the company/ undertaking. Product name Other names Sodium Sulphite Anhydrous
PATIENT INFORMATION ABOUT TREATMENTS FOR ASTHMA AND ALLERGIC RHINITIS, PRESCRIPTIONS & OVER THE COUNTER MEDICINE
 PATIENT INFORMATION ABOUT TREATMENTS FOR ASTHMA AND ALLERGIC RHINITIS, PRESCRIPTIONS & OVER THE COUNTER MEDICINE The content of this booklet was developed by Allergy UK. MSD reviewed this booklet to comment
PATIENT INFORMATION ABOUT TREATMENTS FOR ASTHMA AND ALLERGIC RHINITIS, PRESCRIPTIONS & OVER THE COUNTER MEDICINE The content of this booklet was developed by Allergy UK. MSD reviewed this booklet to comment
Poison Ivy, Oak & Sumac: A Rash of Information About Identification, Treatment and Prevention
 Poison Ivy, Oak & Sumac: A Rash of Information About Identification, Treatment and Prevention The American Academy of Family Physicians Foundation has favorably reviewed this material through 2007. Favorable
Poison Ivy, Oak & Sumac: A Rash of Information About Identification, Treatment and Prevention The American Academy of Family Physicians Foundation has favorably reviewed this material through 2007. Favorable
MATERIAL SAFETY DATA SHEET
 Page 1 of 7 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 7 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
RADIOPHARMACEUTICALS BASED ON MONOCLONAL ANTIBODIES
 RADIOPHARMACEUTICALS BASED ON MONOCLONAL ANTIBODIES Guideline Title Radiopharmaceuticals based on Monoclonal Antibodies Legislative basis Directives 65/65/EEC, 75/318/EEC as amended, Directive 89/343/EEC
RADIOPHARMACEUTICALS BASED ON MONOCLONAL ANTIBODIES Guideline Title Radiopharmaceuticals based on Monoclonal Antibodies Legislative basis Directives 65/65/EEC, 75/318/EEC as amended, Directive 89/343/EEC
Summary of the risk management plan (RMP) for Orkambi (lumacaftor and ivacaftor)
 EMA/662624/2015 Summary of the risk management plan (RMP) for Orkambi (lumacaftor and ivacaftor) This is a summary of the risk management plan (RMP) for Orkambi, which details the measures to be taken
EMA/662624/2015 Summary of the risk management plan (RMP) for Orkambi (lumacaftor and ivacaftor) This is a summary of the risk management plan (RMP) for Orkambi, which details the measures to be taken
Glossary of Terms. Section Glossary. of Terms
 Glossary of Terms Section Glossary of Terms GLOSSARY Acute: Symptoms which can occur suddenly with a short and severe course. Adrenaclick /Generic Adrenaclick : a single use epinephrine auto-injector that
Glossary of Terms Section Glossary of Terms GLOSSARY Acute: Symptoms which can occur suddenly with a short and severe course. Adrenaclick /Generic Adrenaclick : a single use epinephrine auto-injector that
MATERIAL SAFETY DATA SHEET
 Page 1 of 5 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 5 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
MATERIAL SAFETY DATA SHEET
 Page 1 of 6 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 6 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
ASTHMA IN INFANTS AND YOUNG CHILDREN
 ASTHMA IN INFANTS AND YOUNG CHILDREN What is Asthma? Asthma is a chronic inflammatory disease of the airways. Symptoms of asthma are variable. That means that they can be mild to severe, intermittent to
ASTHMA IN INFANTS AND YOUNG CHILDREN What is Asthma? Asthma is a chronic inflammatory disease of the airways. Symptoms of asthma are variable. That means that they can be mild to severe, intermittent to
What You And Your Family Can Do About Asthma
 GLOBAL INITIATIVE FOR ASTHMA What You And Your Family Can Do About Asthma BASED ON THE GLOBAL STRATEGY FOR ASTHMA MANAGEMENT AND PREVENTION NHLBI/WHO WORKSHOP REPORT NATIONAL INSTITUTES OF HEALTH NATIONAL
GLOBAL INITIATIVE FOR ASTHMA What You And Your Family Can Do About Asthma BASED ON THE GLOBAL STRATEGY FOR ASTHMA MANAGEMENT AND PREVENTION NHLBI/WHO WORKSHOP REPORT NATIONAL INSTITUTES OF HEALTH NATIONAL
DRUG INTERACTIONS: WHAT YOU SHOULD KNOW. Council on Family Health
 DRUG INTERACTIONS: WHAT YOU SHOULD KNOW Council on Family Health Drug Interactions There are more opportunities today than ever before to learn about your health and to take better care of yourself. It
DRUG INTERACTIONS: WHAT YOU SHOULD KNOW Council on Family Health Drug Interactions There are more opportunities today than ever before to learn about your health and to take better care of yourself. It
- Woody Kageler, M.D.
 Dear Parent/Guardian, We all want our children with asthma to be healthy! Asthma can be a frightening experience for kids, as well as their parents. Let Darby BoIngg be your partner, along with your physician,
Dear Parent/Guardian, We all want our children with asthma to be healthy! Asthma can be a frightening experience for kids, as well as their parents. Let Darby BoIngg be your partner, along with your physician,
Perrigo Pharmaceuticals Co.
 Ibuprofen Page 1 of 7 Perrigo Pharmaceuticals Co. U.S. Pharmaceuticals Division MATERIAL SAFETY DATA SHEET NDC Product(s) Code(s): 0121-4774-05 0121-4774-10 Product Name: Ibuprofen Oral Suspension 100mg/5mL
Ibuprofen Page 1 of 7 Perrigo Pharmaceuticals Co. U.S. Pharmaceuticals Division MATERIAL SAFETY DATA SHEET NDC Product(s) Code(s): 0121-4774-05 0121-4774-10 Product Name: Ibuprofen Oral Suspension 100mg/5mL
Package leaflet: Information for the patient. Dorzolamide Mylan 20 mg/ml Eye drops, Solution Dorzolamide
 Package leaflet: Information for the patient Dorzolamide Mylan 20 mg/ml Eye drops, Solution Dorzolamide Read all of this leaflet carefully before you start using this medicine because it contains important
Package leaflet: Information for the patient Dorzolamide Mylan 20 mg/ml Eye drops, Solution Dorzolamide Read all of this leaflet carefully before you start using this medicine because it contains important
DOSAGE FORMS AND STRENGTHS Intravitreal implant containing dexamethasone 0.7 mg in the NOVADUR solid polymer drug delivery system.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use OZURDEX safely and effectively. See full prescribing information for OZURDEX. OZURDEX (dexamethasone
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use OZURDEX safely and effectively. See full prescribing information for OZURDEX. OZURDEX (dexamethasone
Guidance for Industry Safety Testing of Drug Metabolites
 Guidance for Industry Safety Testing of Drug Metabolites U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2008 Pharmacology
Guidance for Industry Safety Testing of Drug Metabolites U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2008 Pharmacology
FRENCH AGENCY FOR VETERINARY MEDICINAL PRODUCTS DECENTRALISED PROCEDURE PUBLICLY AVAILABLE ASSESSMENT REPORT FOR A VETERINARY MEDICINAL PRODUCT
 FRENCH AGENCY FOR VETERINARY MEDICINAL PRODUCTS DECENTRALISED PROCEDURE FOR A VETERINARY MEDICINAL PRODUCT EMEDOG, 1 mg/ml, solution for injection for dogs Date: 20/08/2015 French agency for food, environnemental
FRENCH AGENCY FOR VETERINARY MEDICINAL PRODUCTS DECENTRALISED PROCEDURE FOR A VETERINARY MEDICINAL PRODUCT EMEDOG, 1 mg/ml, solution for injection for dogs Date: 20/08/2015 French agency for food, environnemental
Guidance for Industry and Review Staff Target Product Profile A Strategic Development Process Tool
 Guidance for Industry and Review Staff Product Profile A Strategic Development Process Tool DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions
Guidance for Industry and Review Staff Product Profile A Strategic Development Process Tool DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions
VOLTAREN OPHTHA EYE DROPS
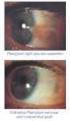 2013 במאי משרד הבריאות ותוכנו נבדק ואושר על ידו ע"י עלון זה נקבע ע פורמט PRESCRIBING INFORMATION VOLTAREN OPHTHA EYE DROPS 1 Name of the medicinal product Voltaren Ophtha Eye Drops. 2 Qualitative and quantitative
2013 במאי משרד הבריאות ותוכנו נבדק ואושר על ידו ע"י עלון זה נקבע ע פורמט PRESCRIBING INFORMATION VOLTAREN OPHTHA EYE DROPS 1 Name of the medicinal product Voltaren Ophtha Eye Drops. 2 Qualitative and quantitative
MATERIAL SAFETY DATA SHEET Description: SAF DRAIN Revision Number: 00
 GRANULAR, ALKALINE DRAIN CLEANER GENERAL SAF-DRAIN is a powerful alkaline drain cleaner designed so that the product reaches the problem area before the reaction takes place. It effectively cleans organic
GRANULAR, ALKALINE DRAIN CLEANER GENERAL SAF-DRAIN is a powerful alkaline drain cleaner designed so that the product reaches the problem area before the reaction takes place. It effectively cleans organic
On completion of this chapter you should be able to: discuss the stepwise approach to the pharmacological management of asthma in children
 7 Asthma Asthma is a common disease in children and its incidence has been increasing in recent years. Between 10-15% of children have been diagnosed with asthma. It is therefore a condition that pharmacists
7 Asthma Asthma is a common disease in children and its incidence has been increasing in recent years. Between 10-15% of children have been diagnosed with asthma. It is therefore a condition that pharmacists
LiAiSON. Formaldehyde. In This Issue. How does Formaldehyde affect human health?
 LiAiSON OCTOBER Marwell Medical Newsletter Formaldehyde How does Formaldehyde affect human health? Formaldehyde is toxic by inhalation, by skin contact, and by swallowing. Breathing Formaldehyde vapours
LiAiSON OCTOBER Marwell Medical Newsletter Formaldehyde How does Formaldehyde affect human health? Formaldehyde is toxic by inhalation, by skin contact, and by swallowing. Breathing Formaldehyde vapours
Teacher s Guide. Chemicals & Human Health Website. Toxicology Activity
 Teacher s Guide Chemicals & Human Health Website www.biology.arizona.edu/chh Toxicology Activity 1. Pre-test - Have the students the questions on the worksheet prior to visiting the website. This is just
Teacher s Guide Chemicals & Human Health Website www.biology.arizona.edu/chh Toxicology Activity 1. Pre-test - Have the students the questions on the worksheet prior to visiting the website. This is just
MEDICATION GUIDE ACTOPLUS MET (ak-tō-plus-met) (pioglitazone hydrochloride and metformin hydrochloride) tablets
 MEDICATION GUIDE (ak-tō-plus-met) (pioglitazone hydrochloride and metformin hydrochloride) tablets Read this Medication Guide carefully before you start taking and each time you get a refill. There may
MEDICATION GUIDE (ak-tō-plus-met) (pioglitazone hydrochloride and metformin hydrochloride) tablets Read this Medication Guide carefully before you start taking and each time you get a refill. There may
MATERIAL SAFETY DATA SHEET
 Page 1 of 6 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 6 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
