NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate.
|
|
|
- Joy Bridges
- 9 years ago
- Views:
Transcription
1 NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate. PRESENTATION Eye Drops: NAPHCON-A Eye Drops are a combination of an antihistamine (pheniramine maleate) and a decongestant (naphazoline hydrochloride) prepared as a sterile solution for ophthalmic use. NAPHCON-A Eye Drops contain 0.25 mg/ml naphazoline hydrochloride and 3.0 mg/ml pheniramine maleate, together with the excipients disodium edetate dihydrate, boric acid, sodium borate decahydrate, sodium chloride, hydrochloric acid, sodium hydroxide and purified water. The solution is preserved with benzalkonium chloride (0.1 mg/ml). USES Actions NAPHCON-A Eye Drops combine the effects of the antihistamine, pheniramine maleate, and the decongestant, naphazoline hydrochloride. Naphazoline hydrochloride is a direct acting sympathomimetic amine. It acts on alpha-adrenergic receptors in the arterioles of the conjunctiva to produce vasoconstriction, resulting in decreased conjunctival congestion. Pheniramine maleate is an alkylamine derivative with antimuscarinic and central sedative properties which is used for the symptomatic relief of hypersensitivity reactions including conjunctivitis. Indications For the symptomatic treatment of allergic conjunctivitis. DOSAGE AND ADMINISTRATION One to two drops of NAPHCON-A Eye Drops should be instilled into the affected eye(s) three to four times daily. CONTRAINDICATIONS The use of NAPHCON-A Eye Drops is contraindicated in patients who are known to experience narrow-angle glaucoma or who have known hypersensitivities to one or more of the components of this preparation.
2 WARNINGS AND PRECAUTIONS Patients being treated with monoamine oxidase inhibitors (MAOIs) may experience a severe hypertensive crisis if given a sympathomimetic drug. Patients already using an eye product obtained on prescription should use NAPHCON-A only after consultation with a doctor or pharmacist. Patients known to be sensitive to other ophthalmic sympathomimetic preparations may also experience sensitivity reactions to NAPHCON-A Eye Drops. This preparation should be used with caution in children, the elderly, patients with cardiovascular disease including cardiac arrhythmia, patients with poorly controlled hypertension, patients with diabetes (especially when the diabetes is not adequately controlled) and patients who have urinary retention or prostate hypertrophy. Use with caution in patients with sympathetic denervation (e.g. patients with insulin dependent diabetes, orthostatic hypotension, hypertension, hyperthyroidism) due to the risk for possible systemic effects. Caution should also be taken if patients are known to suffer from pyloroduodenal obstruction or epilepsy. Prolonged and / or excessive use may lead to rebound ocular vasodilatation or congestion. NAPHCON-A Eye Drops contain benzalkonium chloride which may cause eye irritation and is known to discolour soft contact lenses. Avoid contact with soft contact lenses. Patients must be instructed to remove contact lenses prior to application of NAPHCON-A and wait at least 15 minutes before reinsertion. If a diminution of symptoms is not seen or the condition worsens in the first 72 hours of treatment with NAPHCON-A Eye Drops, treatment should be discontinued and the advice of a physician sought. NAPHCON-A Eye Drops should not be used for longterm treatment (i.e. for more than 14 days) without further evaluation of the patient. Fertility Studies have not been performed to evaluate the effect of topical ocular administration of NAPHCON-A Eye Drops on human fertility. Use in pregnancy - CATEGORY A Drugs which have been taken by a large number of pregnant women and women of childbearing age without any proven increase in the frequency of malformations or other direct or indirect harmful effects on the foetus having been observed. There are no or a limited amount of data from the use of topical ophthalmic naphazoline or pheniramine in pregnant women; naphazoline hydrochloride may be absorbed systemically following ophthalmic administration. NAPHCON-A Eye Drops should only be used by a pregnant woman if clearly needed. Animal studies are insufficient with respect to reproductive toxicity.
3 Use in lactation There are no well-controlled studies in breast-feeding women; naphazoline hydrochloride may be absorbed systemically following ophthalmic administration. It is unknown whether topical naphazoline / metabolites are excreted in human milk. However, a risk to the breastfed child cannot be excluded. NAPHCON-A Eye Drops should only be used by a breast-feeding woman if clearly needed. Use in children Safety and effectiveness in children under 12 years of age have not been established. Use in the elderly No well-controlled studies in elderly populations have been conducted, however, no potential issues have been identified since marketing the product. Effects of ability to drive and use machines NAPHCON-A Eye Drops may cause transient mydriasis, temporary blurred vision or other visual disturbances that may affect the ability to drive or use machines. If there is mydriasis or if blurred vision occurs after instillation, the patient must wait until the vision clears before driving or using machinery. Patient instructions Patients should be advised: to avoid touching the dropper tip to the eyelids, surrounding areas or any other surface as this may contaminate the product; to contact a doctor or pharmacist if the symptoms persist for more than 72 hours or worsen; not to use NAPHCON-A Eye Drops while soft (hydrophilic) contact lenses are in place. Soft contact lenses can be inserted 15 minutes after instillation of NAPHCON-A Eye Drops; to read the Patient Information Leaflet supplied with NAPHCON-A Eye Drops before using the product. ADVERSE REACTIONS The following adverse reactions have been reported during clinical trials with NAPHCON-A Eye Drops. They are classified according to the subsequent convention: very common ( 1/10), common ( 1/100 to <1/10), uncommon ( 1/1,000 to <1/100), rare ( 1/10,000 to <1/1,000) and very rare (<1/10,000). Within each frequency-grouping, adverse reactions are presented in order of decreasing seriousness. System Organ Classification MedDRA Preferred term (v. 14.1) Eye disorders Common: ocular discomfort Uncommon: keratitis, eye pain, eye oedema, ocular hyperaemia Additional adverse reactions identified from post-marketing surveillance include the following. Frequencies cannot be estimated from the available data.
4 System Organ Classification Eye disorders MedDRA Preferred Term (v.14.1) Mydriasis, eye irritation, vision blurred Paediatric population Excessive use of naphazoline-pheniramine in infants and young children may cause depression of the central nervous system and significant reduction in body temperature. INTERACTIONS An increased risk of arrhythmias may be seen if NAPHCON-A Eye Drops are administered concomitantly with cardiac glycosides, quinidine or tricyclic antidepressants. The sedative effects of central nervous system depressants such as alcohol, barbiturates, hypnotics, opioid analgesics, anxiolytic sedatives and neuroleptics may be potentiated if administered concomitantly with NAPHCON-A Eye Drops. Patients being treated with monoamine oxidase inhibitors may experience a severe hypertensive reaction if given a sympathomimetic drug. Although this reaction has not specifically been reported with naphazoline, the possibility of such an interaction should be considered. OVERDOSAGE A topical overdose of NAPHCON-A Eye Drops may be flushed from the eyes with warm tap water. In case of overdosage or accidental ingestion, naphazoline can cause the following, particularly in children: depression of the central nervous system with a clear fall in body temperature and symptoms of bradycardia, excessive sweating, drowsiness and coma; hypertension followed by hypotension. Treatment of an oral overdose is symptomatic and supportive. PHARMACEUTICAL PRECAUTIONS Store below 25 C. Protect from light and excessive heat. Discard container 4 weeks after opening. Keep NAPHCON-A Eye Drops and all other medicines out of the reach of children. Remove contact lenses before using (see WARNINGS AND PRECAUTIONS). MEDICINE CLASSIFICATION Pharmacy Only Medicine.
5 PACKAGE QUANTITIES 15 ml DROP-TAINER dispenser. NAME AND ADDRESS Pharmaco (NZ) Ltd 4 Fisher Crescent Auckland 1060 New Zealand DATE OF PREPARATION July 2014
Hypromellose Eye Drops BP 0.3% w/v PL 23097/0006
 Hypromellose Eye Drops BP 0.3% w/v PL 23097/0006 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
Hypromellose Eye Drops BP 0.3% w/v PL 23097/0006 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
used to treat inflammation, corneal injury and bacterial infections in the external part of the eye.
 TOBRADEX * Eye Drops Tobramycin and Dexamethasone CONSUMER MEDICINE INFORMATION What is in this Leaflet This leaflet answers some common questions about TOBRADEX* Eye Drops. It does not contain all the
TOBRADEX * Eye Drops Tobramycin and Dexamethasone CONSUMER MEDICINE INFORMATION What is in this Leaflet This leaflet answers some common questions about TOBRADEX* Eye Drops. It does not contain all the
PHENYLEPHRINE HYDROCHLORIDE INJECTION USP
 PRESCRIBING INFORMATION PHENYLEPHRINE HYDROCHLORIDE INJECTION USP 10 mg/ml Sandoz Canada Inc. Date of Preparation: September 1992 145 Jules-Léger Date of Revision : January 13, 2011 Boucherville, QC, Canada
PRESCRIBING INFORMATION PHENYLEPHRINE HYDROCHLORIDE INJECTION USP 10 mg/ml Sandoz Canada Inc. Date of Preparation: September 1992 145 Jules-Léger Date of Revision : January 13, 2011 Boucherville, QC, Canada
DO YOU HAVE ITCHY ALLERGY EYES? Find out about lasting relief
 DO YOU HAVE ITCHY ALLERGY EYES? Find out about lasting relief Common causes of itching due to eye allergies include: Pollen from trees, grasses, and ragweed Dust mites Cat dander Approximately 10 million
DO YOU HAVE ITCHY ALLERGY EYES? Find out about lasting relief Common causes of itching due to eye allergies include: Pollen from trees, grasses, and ragweed Dust mites Cat dander Approximately 10 million
UBISTESIN 1:200,000 and UBISTESIN FORTE 1:100,000
 UBISTESIN 1:200,000 and UBISTESIN FORTE 1:100,000 Articaine hydrochloride and adrenaline hydrochloride Consumer Medicine Information WHAT IS IN THIS LEAFLET Please read this leaflet carefully before you
UBISTESIN 1:200,000 and UBISTESIN FORTE 1:100,000 Articaine hydrochloride and adrenaline hydrochloride Consumer Medicine Information WHAT IS IN THIS LEAFLET Please read this leaflet carefully before you
ZOVIRAX Cold Sore Cream
 Data Sheet ZOVIRAX Cold Sore Cream Aciclovir 5% w/w Presentation Topical cream Indications ZOVIRAX Cold Sore Cream is indicated for the treatment of Herpes simplex virus infections of the lips and face
Data Sheet ZOVIRAX Cold Sore Cream Aciclovir 5% w/w Presentation Topical cream Indications ZOVIRAX Cold Sore Cream is indicated for the treatment of Herpes simplex virus infections of the lips and face
PACKAGE LEAFLET: INFORMATION FOR THE USER. ADRENALINE (HCl) STEROP 0,8mg/1ml. Solution for injection. Adrenaline (Levorenine, Epinephrine)
 PACKAGE LEAFLET: INFORMATION FOR THE USER ADRENALINE (HCl) STEROP 0,4mg/1ml ADRENALINE (HCl) STEROP 0,8mg/1ml Solution for injection Adrenaline (Levorenine, Epinephrine) Read all of this leaflet carefully
PACKAGE LEAFLET: INFORMATION FOR THE USER ADRENALINE (HCl) STEROP 0,4mg/1ml ADRENALINE (HCl) STEROP 0,8mg/1ml Solution for injection Adrenaline (Levorenine, Epinephrine) Read all of this leaflet carefully
Summary of the risk management plan (RMP) for Rasagiline ratiopharm (rasagiline)
 EMA/744222/2014 Summary of the risk management plan (RMP) for Rasagiline ratiopharm (rasagiline) This is a summary of the risk management plan (RMP) for Rasagiline ratiopharm, which details the measures
EMA/744222/2014 Summary of the risk management plan (RMP) for Rasagiline ratiopharm (rasagiline) This is a summary of the risk management plan (RMP) for Rasagiline ratiopharm, which details the measures
PACKAGE LEAFLET: INFORMATION FOR THE USER. ADRENALINE (TARTRATE) STEROP 1 mg/1 ml Solution for injection. Adrenaline (Levorenine, Epinephrine)
 PACKAGE LEAFLET: INFORMATION FOR THE USER ADRENALINE (TARTRATE) STEROP 1 mg/1 ml Solution for injection Adrenaline (Levorenine, Epinephrine) Read all of this leaflet carefully before you start using this
PACKAGE LEAFLET: INFORMATION FOR THE USER ADRENALINE (TARTRATE) STEROP 1 mg/1 ml Solution for injection Adrenaline (Levorenine, Epinephrine) Read all of this leaflet carefully before you start using this
Revised: 9/2014. LUMIGAN (bimatoprost ophthalmic solution) 0.01% for topical ophthalmic use. Initial U.S. Approval: 2001
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LUMIGAN (bimatoprost ophthalmic solution) 0.01% safely and effectively. See full prescribing information
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LUMIGAN (bimatoprost ophthalmic solution) 0.01% safely and effectively. See full prescribing information
VOLTAREN OPHTHA EYE DROPS
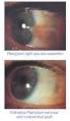 פורמט עלון זה נקבע ע"ימשרד הבריאות ותוכנו נבדק ואושר על ידו באפריל 2008 PRESCRIBING INFORMATION VOLTAREN OPHTHA EYE DROPS 1 Name of the medicinal product Voltaren Ophtha Eye Drops. 2 Qualitative and quantitative
פורמט עלון זה נקבע ע"ימשרד הבריאות ותוכנו נבדק ואושר על ידו באפריל 2008 PRESCRIBING INFORMATION VOLTAREN OPHTHA EYE DROPS 1 Name of the medicinal product Voltaren Ophtha Eye Drops. 2 Qualitative and quantitative
White, circular, biconvex, uncoated tablets with a score line on one side, plain on the other.
 Nausicalm Cyclizine hydrochloride Ph. Eur. 50 mg Presentation White, circular, biconvex, uncoated tablets with a score line on one side, plain on the other. Uses Actions The active ingredient-cyclizine
Nausicalm Cyclizine hydrochloride Ph. Eur. 50 mg Presentation White, circular, biconvex, uncoated tablets with a score line on one side, plain on the other. Uses Actions The active ingredient-cyclizine
Glycopyrronium Bromide 0.5mg/mL and Neostigmine Metilsulfate 2.5mg/mL Solution for Injection
 NEW ZEALAND DATA SHEET Glycopyrronium Bromide 0.5mg/mL and Neostigmine Metilsulfate 2.5mg/mL Solution for Injection Each 1mL contains Glycopyrrolate USP 0.5mg (Glycopyrronium Bromide) and Neostigmine Metilsulfate
NEW ZEALAND DATA SHEET Glycopyrronium Bromide 0.5mg/mL and Neostigmine Metilsulfate 2.5mg/mL Solution for Injection Each 1mL contains Glycopyrrolate USP 0.5mg (Glycopyrronium Bromide) and Neostigmine Metilsulfate
1 What Anapen is and what it is used for?
 PACKAGE LEAFLET: INFORMATION FOR THE USER Anapen 500 micrograms in 0.3 ml solution for injection (pre-filled syringe) Adrenaline (Epinephrine) Auto-Injector Read all of this leaflet carefully before you
PACKAGE LEAFLET: INFORMATION FOR THE USER Anapen 500 micrograms in 0.3 ml solution for injection (pre-filled syringe) Adrenaline (Epinephrine) Auto-Injector Read all of this leaflet carefully before you
Summary of the risk management plan (RMP) for Ionsys (fentanyl)
 EMA/764409/2015 Summary of the risk management plan (RMP) for Ionsys (fentanyl) This is a summary of the risk management plan (RMP) for Ionsys, which details the measures to be taken in order to ensure
EMA/764409/2015 Summary of the risk management plan (RMP) for Ionsys (fentanyl) This is a summary of the risk management plan (RMP) for Ionsys, which details the measures to be taken in order to ensure
What Codeine Phosphate Tablets are used for
 New Zealand Consumer Medicine Information CODEINE PHOSPHATE 15mg, 30mg & 60mg Tablets What is in this leaflet Please read this leaflet carefully before you start using Codeine Phosphate Tablets. This leaflet
New Zealand Consumer Medicine Information CODEINE PHOSPHATE 15mg, 30mg & 60mg Tablets What is in this leaflet Please read this leaflet carefully before you start using Codeine Phosphate Tablets. This leaflet
1. What Xylocaine with adrenaline is and what it is used for
 Package leaflet: Information for the user Xylocaine 1% and 2% with adrenaline (epinephrine) 1:200,000 Solution for Injection lidocaine, adrenaline (epinephrine) Read all of this leaflet carefully before
Package leaflet: Information for the user Xylocaine 1% and 2% with adrenaline (epinephrine) 1:200,000 Solution for Injection lidocaine, adrenaline (epinephrine) Read all of this leaflet carefully before
Elements for a public summary. VI.2.1 Overview of disease epidemiology. VI.2.2 Summary of treatment benefits
 VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Pain is one of the most common reasons for a patient to seek medical attention. Moderate or severe intensity pain can be acute
VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Pain is one of the most common reasons for a patient to seek medical attention. Moderate or severe intensity pain can be acute
PACKAGE LEAFLET: INFORMATION FOR THE USER. PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion. Paracetamol
 PACKAGE LEAFLET: INFORMATION FOR THE USER PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion Paracetamol Read all of this leaflet carefully before you start using this medicine. Keep this leaflet.
PACKAGE LEAFLET: INFORMATION FOR THE USER PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion Paracetamol Read all of this leaflet carefully before you start using this medicine. Keep this leaflet.
NEUROTONE THR 00904/0005 UKPAR
 NEUROTONE THR 00904/0005 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 10 Summary of Product Characteristics Page 11 Product Information Leaflet
NEUROTONE THR 00904/0005 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 10 Summary of Product Characteristics Page 11 Product Information Leaflet
VOLTAREN OPHTHA EYE DROPS
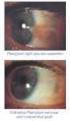 2013 במאי משרד הבריאות ותוכנו נבדק ואושר על ידו ע"י עלון זה נקבע ע פורמט PRESCRIBING INFORMATION VOLTAREN OPHTHA EYE DROPS 1 Name of the medicinal product Voltaren Ophtha Eye Drops. 2 Qualitative and quantitative
2013 במאי משרד הבריאות ותוכנו נבדק ואושר על ידו ע"י עלון זה נקבע ע פורמט PRESCRIBING INFORMATION VOLTAREN OPHTHA EYE DROPS 1 Name of the medicinal product Voltaren Ophtha Eye Drops. 2 Qualitative and quantitative
4 Clinical Particulars
 SUMMARY OF PRODUCT CHARACTERISTICS 1 Name of the Medicinal Product Procyclidine Syrup 5mg/5ml 2. Qualitative and Quantitative Composition Each 5ml dose contains 5mg Procyclidine Hydrochloride BP. 3. Pharmaceutical
SUMMARY OF PRODUCT CHARACTERISTICS 1 Name of the Medicinal Product Procyclidine Syrup 5mg/5ml 2. Qualitative and Quantitative Composition Each 5ml dose contains 5mg Procyclidine Hydrochloride BP. 3. Pharmaceutical
Naloxone Hydrochloride Injection PRODUCT INFORMATION
 Naloxone Hydrochloride Injection PRODUCT INFORMATION DESCRIPTION Naloxone hydrochloride is 17-allyl-4,5α-epoxy-3,14-dihydroxymorphinan-6-one hydrochloride; C 19 H 21 NO 4.HCl. It is an off-white powder
Naloxone Hydrochloride Injection PRODUCT INFORMATION DESCRIPTION Naloxone hydrochloride is 17-allyl-4,5α-epoxy-3,14-dihydroxymorphinan-6-one hydrochloride; C 19 H 21 NO 4.HCl. It is an off-white powder
Thioctacid 600 T Solution for Injection contains 600 mg alpha-lipoic acid
 Package Leaflet: Information for the User Thioctacid 600 T Solution for Injection contains 600 mg alpha-lipoic acid For use in adults Active substance: Alpha-lipoic acid, Trometamol salt (1:1) Read all
Package Leaflet: Information for the User Thioctacid 600 T Solution for Injection contains 600 mg alpha-lipoic acid For use in adults Active substance: Alpha-lipoic acid, Trometamol salt (1:1) Read all
CODEINE PHOSPHATE 15 MG TABLETS CODEINE PHOSPHATE 30 MG TABLETS CODEINE PHOSPHATE 60 MG TABLETS PL 24837/0023-5 UK PAR TABLE OF CONTENTS
 CODEINE PHOSPHATE 15 MG TABLETS CODEINE PHOSPHATE 30 MG TABLETS CODEINE PHOSPHATE 60 MG TABLETS PL 24837/0023-5 UK PAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for
CODEINE PHOSPHATE 15 MG TABLETS CODEINE PHOSPHATE 30 MG TABLETS CODEINE PHOSPHATE 60 MG TABLETS PL 24837/0023-5 UK PAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for
If you forget to use Catacrom Eye Drops If you forget to use this medicine, use it as soon as you remember.
 Package leaflet: Information for the user Catacrom 2 % w/v Eye Drops, Solution Sodium cromoglicate Read all of this leaflet carefully before you start using this medicine because it contains important
Package leaflet: Information for the user Catacrom 2 % w/v Eye Drops, Solution Sodium cromoglicate Read all of this leaflet carefully before you start using this medicine because it contains important
PRESCRIBING INFORMATION PRODUCT MONOGRAPH ADRENALIN* Adrenalin* Chloride Solution (Epinephrine Injection U.S.P) 1:1000 (1 mg/ml)
 PRESCRIBING INFORMATION PRODUCT MONOGRAPH ADRENALIN* Adrenalin* Chloride Solution (Epinephrine Injection U.S.P) 1:1000 (1 mg/ml) Adrenalin* Chloride Injection (Epinephrine Injection U.S.P.) 1:1000, 30
PRESCRIBING INFORMATION PRODUCT MONOGRAPH ADRENALIN* Adrenalin* Chloride Solution (Epinephrine Injection U.S.P) 1:1000 (1 mg/ml) Adrenalin* Chloride Injection (Epinephrine Injection U.S.P.) 1:1000, 30
New Zealand Consumer Medicine Information
 New Zealand Consumer Medicine Information Actrapid Penfill 100 IU/ml solution for injection in cartridge Insulin human (rdna) Read all of this leaflet carefully before you start using your insulin Keep
New Zealand Consumer Medicine Information Actrapid Penfill 100 IU/ml solution for injection in cartridge Insulin human (rdna) Read all of this leaflet carefully before you start using your insulin Keep
PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement)
 1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
Perfalgan 10 mg/ml, solution for infusion
 PACKAGE LEAFLET: INFORMATION FOR THE USER Perfalgan 10 mg/ml, solution for infusion Paracetamol Read all of this leaflet carefully before you start using this medicine. Keep this leaflet. You may need
PACKAGE LEAFLET: INFORMATION FOR THE USER Perfalgan 10 mg/ml, solution for infusion Paracetamol Read all of this leaflet carefully before you start using this medicine. Keep this leaflet. You may need
Remeron (mirtazapine)
 Remeron (mirtazapine) FDA ALERT [07/2005] Suicidal Thoughts or Actions in Children and Adults Patients with depression or other mental illnesses often think about or attempt suicide. Closely watch anyone
Remeron (mirtazapine) FDA ALERT [07/2005] Suicidal Thoughts or Actions in Children and Adults Patients with depression or other mental illnesses often think about or attempt suicide. Closely watch anyone
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Anapen 500 micrograms in 03ml solution for injection in a pre-filled syringe 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each millilitre
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Anapen 500 micrograms in 03ml solution for injection in a pre-filled syringe 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each millilitre
Summary of the risk management plan (RMP) for Aripiprazole Pharmathen (aripiprazole)
 EMA/303592/2015 Summary of the risk management plan (RMP) for Aripiprazole Pharmathen (aripiprazole) This is a summary of the risk management plan (RMP) for Aripiprazole Pharmathen, which details the measures
EMA/303592/2015 Summary of the risk management plan (RMP) for Aripiprazole Pharmathen (aripiprazole) This is a summary of the risk management plan (RMP) for Aripiprazole Pharmathen, which details the measures
3% Sodium Chloride Injection, USP 5% Sodium Chloride Injection, USP
 PRESCRIBING INFORMATION 3% Sodium Chloride Injection, USP 5% Sodium Chloride Injection, USP IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of Revision:
PRESCRIBING INFORMATION 3% Sodium Chloride Injection, USP 5% Sodium Chloride Injection, USP IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of Revision:
Yes This controlled document shall not be copied in part or whole without the express permission of the author or the author s representative.
 Title: Patient Group Direction for the administration of lidocaine hydrochloride 1% injection as infiltration anaesthesia for insertion/removal of central venous catheters by nurses/radiographers working
Title: Patient Group Direction for the administration of lidocaine hydrochloride 1% injection as infiltration anaesthesia for insertion/removal of central venous catheters by nurses/radiographers working
Package Leaflet: Information for the user
 Package Leaflet: Information for the user Actrapid 100 IU/ml solution for injection in vial Insulin human (rdna) Read all of this leaflet carefully before you start using your insulin Keep this leaflet.
Package Leaflet: Information for the user Actrapid 100 IU/ml solution for injection in vial Insulin human (rdna) Read all of this leaflet carefully before you start using your insulin Keep this leaflet.
PACKAGE LEAFLET: INFORMATION FOR THE USER. VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution. Cyanocobalamin
 PACKAGE LEAFLET: INFORMATION FOR THE USER VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution Cyanocobalamin Read all of this leaflet carefully before you start using this medicine because
PACKAGE LEAFLET: INFORMATION FOR THE USER VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution Cyanocobalamin Read all of this leaflet carefully before you start using this medicine because
The format of this leaflet was determined by the Ministry of Health and its content was checked and approved in May 2013.
 The format of this leaflet was determined by the Ministry of Health and its content was checked and approved in May 2013 Havrix 720 Junior 1 NAME OF THE MEDICINAL PRODUCT Havrix 720 Junior 2 QUALITATIVE
The format of this leaflet was determined by the Ministry of Health and its content was checked and approved in May 2013 Havrix 720 Junior 1 NAME OF THE MEDICINAL PRODUCT Havrix 720 Junior 2 QUALITATIVE
Paxil/Paxil-CR (paroxetine)
 Generic name: Paroxetine Available strengths: 10 mg, 20 mg, 30 mg, 40 mg tablets; 10 mg/5 ml oral suspension; 12.5 mg, 25 mg, 37.5 mg controlled-release tablets (Paxil-CR) Available in generic: Yes, except
Generic name: Paroxetine Available strengths: 10 mg, 20 mg, 30 mg, 40 mg tablets; 10 mg/5 ml oral suspension; 12.5 mg, 25 mg, 37.5 mg controlled-release tablets (Paxil-CR) Available in generic: Yes, except
VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension
 VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension DESCRIPTION Hydroxyzine pamoate is designated chemically as 1-(p-chlorobenzhydryl) 4- [2-(2-hydroxyethoxy) ethyl] diethylenediamine salt of 1,1
VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension DESCRIPTION Hydroxyzine pamoate is designated chemically as 1-(p-chlorobenzhydryl) 4- [2-(2-hydroxyethoxy) ethyl] diethylenediamine salt of 1,1
SR 5. W2364A_NT.XATRAL SR5 PAK/SA 5/12/05 9:47 Page 1
 W2364A_NT.XATRAL SR5 PAK/SA 5/12/05 9:47 Page 1 alfuzosin SR 5 mg (Alfuzosin HCI) COMPOSITION Each sustained-release, film-coated tablet contains: Alfuzosin hydrochloride......... 5 mg Excipients... q.s.
W2364A_NT.XATRAL SR5 PAK/SA 5/12/05 9:47 Page 1 alfuzosin SR 5 mg (Alfuzosin HCI) COMPOSITION Each sustained-release, film-coated tablet contains: Alfuzosin hydrochloride......... 5 mg Excipients... q.s.
COGENTIN Injection. (benztropine mesylate)
 COGENTIN Injection (benztropine mesylate) NAME OF THE MEDICINE Benztropine mesylate is a synthetic compound resulting from the combination of the active portions of atropine and diphenhydramine. Chemical
COGENTIN Injection (benztropine mesylate) NAME OF THE MEDICINE Benztropine mesylate is a synthetic compound resulting from the combination of the active portions of atropine and diphenhydramine. Chemical
Adrenaline Hydrochloride Injection ADRENALINE 1:10 000 PRODUCT INFORMATION
 Adrenaline Hydrochloride Injection ADRENALINE 1:10 000 PRODUCT INFORMATION DESCRIPTION Adrenaline is (R)-1-(3,4-dihydroxyphenyl)-2-methylaminoethanol; C 9 H 13 NO 3. It is a white odourless crystalline
Adrenaline Hydrochloride Injection ADRENALINE 1:10 000 PRODUCT INFORMATION DESCRIPTION Adrenaline is (R)-1-(3,4-dihydroxyphenyl)-2-methylaminoethanol; C 9 H 13 NO 3. It is a white odourless crystalline
See 17 for PATIENT COUNSELING INFORMATION. Revised: 3/2016 FULL PRESCRIBING INFORMATION: CONTENTS* WARNING: ADRENAL CRISIS IN THE SETTING OF SHOCK OR
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LYSODREN safely and effectively. See full prescribing information for LYSODREN. LYSODREN (mitotane)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LYSODREN safely and effectively. See full prescribing information for LYSODREN. LYSODREN (mitotane)
Shared Care Agreement Insulin Degludec (Tresiba )
 Licensed Indication Shared Care Agreement Insulin Degludec (Tresiba ) Insulin Degludec is licensed for the treatment of diabetes mellitus in adults. Countess of Chester prescribing guidelines Restricting
Licensed Indication Shared Care Agreement Insulin Degludec (Tresiba ) Insulin Degludec is licensed for the treatment of diabetes mellitus in adults. Countess of Chester prescribing guidelines Restricting
Doxylamine succinate belongs to the ethanolamine class of antihistamines with sedative properties.
 Data Sheet MERSYNDOL Tablet Paracetamol 450mg per tablet Codeine Phosphate 9.75mg per tablet Doxylamine Succinate 5mg per tablet MERSYNDOL FORTE Tablet Paracetamol 450mg per tablet Codeine Phosphate 30mg
Data Sheet MERSYNDOL Tablet Paracetamol 450mg per tablet Codeine Phosphate 9.75mg per tablet Doxylamine Succinate 5mg per tablet MERSYNDOL FORTE Tablet Paracetamol 450mg per tablet Codeine Phosphate 30mg
Public Assessment Report. Table of Contents
 Public Assessment Report Lidocaine Injection BP with preservative 1% Lidocaine Injection BP with preservative 2% PL 01502/0070 PL 01502/0071 Hameln Pharmaceuticals Limited Table of Contents Page Lay Summary
Public Assessment Report Lidocaine Injection BP with preservative 1% Lidocaine Injection BP with preservative 2% PL 01502/0070 PL 01502/0071 Hameln Pharmaceuticals Limited Table of Contents Page Lay Summary
TdaP-Booster (tee-dee-ay-pee boo-ster)
 New Zealand Consumer Medicine Information TdaP-Booster (tee-dee-ay-pee boo-ster) Tetanus, diphtheria and pertussis (acellular mono-component) vaccine (adsorbed, reduced antigen content) CONSUMER MEDICINE
New Zealand Consumer Medicine Information TdaP-Booster (tee-dee-ay-pee boo-ster) Tetanus, diphtheria and pertussis (acellular mono-component) vaccine (adsorbed, reduced antigen content) CONSUMER MEDICINE
Package leaflet : information for the user. Dilute Adrenaline/Epinephrine Injection 1:10,000 adrenaline (epinephrine) (as acid tartrate) 0.
 Package leaflet : information for the user Dilute Adrenaline/Epinephrine Injection 1:10,000 adrenaline (epinephrine) (as acid tartrate) 0.1mg per ml Because of your condition it may not be possible for
Package leaflet : information for the user Dilute Adrenaline/Epinephrine Injection 1:10,000 adrenaline (epinephrine) (as acid tartrate) 0.1mg per ml Because of your condition it may not be possible for
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Methadone 10mg/ml Injection / Physeptone 10mg/ml Injection 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Contains: Methadone Hydrochloride
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Methadone 10mg/ml Injection / Physeptone 10mg/ml Injection 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Contains: Methadone Hydrochloride
The clinical studies have been performed in children, adolescents and adults, from 4 years up to 55 years of age.
 NAME OF THE MEDICINE TdaP-Booster. Diphtheria, tetanus and pertussis (acellular mono-component) vaccine (adsorbed, reduced antigen content). DESCRIPTION TdaP-Booster is a suspension for injection in pre-filled
NAME OF THE MEDICINE TdaP-Booster. Diphtheria, tetanus and pertussis (acellular mono-component) vaccine (adsorbed, reduced antigen content). DESCRIPTION TdaP-Booster is a suspension for injection in pre-filled
Salbutamol 1mg/ml Nebuliser Solution. Salbutamol 2mg/ml Nebuliser Solution PL 36390/0035 PL 36390/0036
 Salbutamol 1mg/ml Nebuliser Solution Salbutamol 2mg/ml Nebuliser Solution PL 36390/0035 PL 36390/0036 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment
Salbutamol 1mg/ml Nebuliser Solution Salbutamol 2mg/ml Nebuliser Solution PL 36390/0035 PL 36390/0036 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 1. Trade Name of the Medicinal Product Dilute Adrenaline (Epinephrine) Injection 1:10,000 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of the solution for injections contains 100 micrograms of Adrenaline
1. Trade Name of the Medicinal Product Dilute Adrenaline (Epinephrine) Injection 1:10,000 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of the solution for injections contains 100 micrograms of Adrenaline
BENZTROP 2 mg tablets are round, flat-faced, cross-scored on one side and embossed PMS- 2 on the other side.
 Benztrop Benzatropine mesilate 2 mg tablets Presentation BENZTROP 2 mg tablets are round, flat-faced, cross-scored on one side and embossed PMS- 2 on the other side. Uses Actions Benzatropine is a centrally
Benztrop Benzatropine mesilate 2 mg tablets Presentation BENZTROP 2 mg tablets are round, flat-faced, cross-scored on one side and embossed PMS- 2 on the other side. Uses Actions Benzatropine is a centrally
BRIMONIDINE TARTRATE 0.2%W/V/EYE DROPS PL 15872/0018 UKPAR TABLE OF CONTENTS
 BRIMONIDINE TARTRATE 0.2%W/V/EYE DROPS PL 15872/0018 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 13 Steps taken after authorisation summary Page
BRIMONIDINE TARTRATE 0.2%W/V/EYE DROPS PL 15872/0018 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 13 Steps taken after authorisation summary Page
SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product)
 PART VI SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product) Format and content of the summary of the RMP The summary of the RMP part VI contains information based on RMP modules SI, SVIII and RMP
PART VI SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product) Format and content of the summary of the RMP The summary of the RMP part VI contains information based on RMP modules SI, SVIII and RMP
Always take this medicine exactly as described in this leaflet or as your doctor, pharmacist or nurse have told you.
 leaflet: Information for the user Macrogol 4000 10 g powder for oral solution in sachet Macrogol 4000
leaflet: Information for the user Macrogol 4000 10 g powder for oral solution in sachet Macrogol 4000
Amitriptyline. Drug information Amitriptyline
 Drug information Amitriptyline Amitriptyline This leaflet provides information on amitriptyline and will answer any questions you have about the treatment. Arthritis Research UK produce and print our booklets
Drug information Amitriptyline Amitriptyline This leaflet provides information on amitriptyline and will answer any questions you have about the treatment. Arthritis Research UK produce and print our booklets
MEDICATION GUIDE. What is Morphine Sulfate Oral Solution?
 MEDICATION GUIDE Morphine Sulfate (mor-pheen) (CII) Oral Solution IMPORTANT: Keep Morphine Sulfate Oral Solution in a safe place away from children. Accidental use by a child is a medical emergency and
MEDICATION GUIDE Morphine Sulfate (mor-pheen) (CII) Oral Solution IMPORTANT: Keep Morphine Sulfate Oral Solution in a safe place away from children. Accidental use by a child is a medical emergency and
3% Citanest Dental with Octapressin PRODUCT INFORMATION
 NAME OF THE MEDICINE 3% Citanest Dental with Octapressin PRODUCT INFORMATION 3% Citanest Dental with Octapressin contains prilocaine hydrochloride and felypressin as the active substances. The chemical
NAME OF THE MEDICINE 3% Citanest Dental with Octapressin PRODUCT INFORMATION 3% Citanest Dental with Octapressin contains prilocaine hydrochloride and felypressin as the active substances. The chemical
1g cream or ointment contains 1 mg methylprednisolone aceponate.
 CONSUMER MEDICINE INFORMATION ADVANTAN 1g cream or ointment contains 1 mg methylprednisolone aceponate. What is in this leaflet Please read this leaflet carefully before you start using ADVANTAN. It will
CONSUMER MEDICINE INFORMATION ADVANTAN 1g cream or ointment contains 1 mg methylprednisolone aceponate. What is in this leaflet Please read this leaflet carefully before you start using ADVANTAN. It will
Package leaflet: Information for the patient. Naloxone Hydrochloride 20 micrograms / ml Solution for Injection Naloxone hydrochloride
 A leaflet will be included in each pack. The leaflet will consist of a Technical Information Leaflet and a Patient Information Leaflet. The two leaflets will be easily separatable. The text of the Technical
A leaflet will be included in each pack. The leaflet will consist of a Technical Information Leaflet and a Patient Information Leaflet. The two leaflets will be easily separatable. The text of the Technical
160S01105, Page 1 of 7. Human Hepatitis B Immunoglobulin, solution for intramuscular injection.
 160S01105, Page 1 of 7 New Zealand Data Sheet Hepatitis B Immunoglobulin-VF NAME OF THE MEDICINE Human Hepatitis B Immunoglobulin, solution for intramuscular injection. DESCRIPTION Hepatitis B Immunoglobulin-VF
160S01105, Page 1 of 7 New Zealand Data Sheet Hepatitis B Immunoglobulin-VF NAME OF THE MEDICINE Human Hepatitis B Immunoglobulin, solution for intramuscular injection. DESCRIPTION Hepatitis B Immunoglobulin-VF
Learn More About Product Labeling
 Learn More About Product Labeling Product label The product label is developed during the formal process of review and approval by regulatory agencies of any medicine or medical product. There are specific
Learn More About Product Labeling Product label The product label is developed during the formal process of review and approval by regulatory agencies of any medicine or medical product. There are specific
Chapter 13. Sympathetic Nervous System. Basic Functions of the Nervous System. Divisions of the Peripheral Nervous System
 Chapter 13 Drugs Affecting the Autonomic Basic Functions of the Recognizing changes in Internal environment External environment Processing and integrating changes Reacting to changes Upper Saddle River,
Chapter 13 Drugs Affecting the Autonomic Basic Functions of the Recognizing changes in Internal environment External environment Processing and integrating changes Reacting to changes Upper Saddle River,
New Zealand Consumer Medicine Information
 New Zealand Consumer Medicine Information PenMix 30 100 IU/ml suspension for injection in cartridge PenMix 40 100 IU/ml suspension for injection in cartridge PenMix 50 100 IU/ml suspension for injection
New Zealand Consumer Medicine Information PenMix 30 100 IU/ml suspension for injection in cartridge PenMix 40 100 IU/ml suspension for injection in cartridge PenMix 50 100 IU/ml suspension for injection
Package leaflet: Information for the patient. Dorzolamide Mylan 20 mg/ml Eye drops, Solution Dorzolamide
 Package leaflet: Information for the patient Dorzolamide Mylan 20 mg/ml Eye drops, Solution Dorzolamide Read all of this leaflet carefully before you start using this medicine because it contains important
Package leaflet: Information for the patient Dorzolamide Mylan 20 mg/ml Eye drops, Solution Dorzolamide Read all of this leaflet carefully before you start using this medicine because it contains important
Epipen Junior 150 micrograms solution for injection in pre-filled pen
 Package leaflet: Information for the user Epipen Junior 150 micrograms solution for injection in pre-filled pen Read all of this leaflet carefully before you start using this medicine because it contains
Package leaflet: Information for the user Epipen Junior 150 micrograms solution for injection in pre-filled pen Read all of this leaflet carefully before you start using this medicine because it contains
COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON HYPERICUM PERFORATUM L., HERBA (TRADITIONAL USE)
 European Medicines Agency Evaluation of Medicines for Human Use London, 12 November 2009 Doc. Ref.: EMEA/HMPC/745582/2009 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH
European Medicines Agency Evaluation of Medicines for Human Use London, 12 November 2009 Doc. Ref.: EMEA/HMPC/745582/2009 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH
Intestinal Permeability Leaky Gut Syndrome Protocol Dr. Kurt Woeller, D.O.
 Intestinal Permeability Leaky Gut Syndrome Protocol Dr. Kurt Woeller, D.O. Kurt N. Woeller, DO, is an osteopathic physician specializing in natural medicine. Dr. Woeller began his study of natural medicine
Intestinal Permeability Leaky Gut Syndrome Protocol Dr. Kurt Woeller, D.O. Kurt N. Woeller, DO, is an osteopathic physician specializing in natural medicine. Dr. Woeller began his study of natural medicine
Sativex Cannabis sativa L. extracts (delta-9-tetrahydrocannabinol and cannabidiol) Oromucosal Spray 5.5 / 10 ml
 New Zealand Consumer Medicine Information Sativex Cannabis sativa L. extracts (delta-9-tetrahydrocannabinol and cannabidiol) Oromucosal Spray 5.5 / 10 ml What is in this leaflet Please read all of this
New Zealand Consumer Medicine Information Sativex Cannabis sativa L. extracts (delta-9-tetrahydrocannabinol and cannabidiol) Oromucosal Spray 5.5 / 10 ml What is in this leaflet Please read all of this
DRUG INTERACTIONS: WHAT YOU SHOULD KNOW. Council on Family Health
 DRUG INTERACTIONS: WHAT YOU SHOULD KNOW Council on Family Health Drug Interactions There are more opportunities today than ever before to learn about your health and to take better care of yourself. It
DRUG INTERACTIONS: WHAT YOU SHOULD KNOW Council on Family Health Drug Interactions There are more opportunities today than ever before to learn about your health and to take better care of yourself. It
FULL PRESCRIBING INFORMATION: CONTENTS*
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PHOTREXA VISCOUS and PHOTREXA safely and effectively. See full prescribing information for PHOTREXA
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PHOTREXA VISCOUS and PHOTREXA safely and effectively. See full prescribing information for PHOTREXA
N HUMAN Novo Nordisk Patient Information for Novolin N
 N HUMAN Novo Nordisk Patient Information for Novolin N NOVOLIN N (NO-voe-lin) NPH, Human Insulin Isophane Suspension Injection (recombinant DNA origin) 100 units/ml Important: Know your insulin. Do not
N HUMAN Novo Nordisk Patient Information for Novolin N NOVOLIN N (NO-voe-lin) NPH, Human Insulin Isophane Suspension Injection (recombinant DNA origin) 100 units/ml Important: Know your insulin. Do not
Adrenergic, Adrenergic Blockers, Cholinergic and Cholinergic Blockers
 Adrenergic, Adrenergic Blockers, Cholinergic and Cholinergic Blockers Objective 1: Explain the difference between the sympathetic and parasympathetic nervous systems Autonomic Nervous System Sympathetic
Adrenergic, Adrenergic Blockers, Cholinergic and Cholinergic Blockers Objective 1: Explain the difference between the sympathetic and parasympathetic nervous systems Autonomic Nervous System Sympathetic
0.9% Sodium Chloride Injection, USP In VIAFLEX Plastic Container
 Page 1 of 8 PRESCRIBING INFORMATION 0.9% Sodium Chloride Injection, USP In VIAFLEX Plastic Container IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of
Page 1 of 8 PRESCRIBING INFORMATION 0.9% Sodium Chloride Injection, USP In VIAFLEX Plastic Container IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of
Data Sheet. Paraldehyde
 Data Sheet Paraldehyde Paraldehyde Injection Solution 100% Presentation Paraldehyde Injection BP is a sterile liquid containing paraldehyde BP with hydroquinone 100 micrograms/ml as an antioxidant. It
Data Sheet Paraldehyde Paraldehyde Injection Solution 100% Presentation Paraldehyde Injection BP is a sterile liquid containing paraldehyde BP with hydroquinone 100 micrograms/ml as an antioxidant. It
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Diazepam Tablets BP 2mg 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Diazepam BP 2.00mg 3 PHARMACEUTICAL FORM Tablet 4. CLINICAL PARTICULARS
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Diazepam Tablets BP 2mg 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Diazepam BP 2.00mg 3 PHARMACEUTICAL FORM Tablet 4. CLINICAL PARTICULARS
FLIXONASE ALLERGY Non Drowsy Nasal Spray 24 hour Effective Relief and Prevention Available in 60 & 150 sprays
 FLIXONASE ALLERGY Non Drowsy Nasal Spray 24 hour Effective Relief and Prevention Available in 60 & 150 sprays CONSUMER MEDICINE INFORMATION WHAT IS IN THIS LEAFLET? Please read this leaflet carefully before
FLIXONASE ALLERGY Non Drowsy Nasal Spray 24 hour Effective Relief and Prevention Available in 60 & 150 sprays CONSUMER MEDICINE INFORMATION WHAT IS IN THIS LEAFLET? Please read this leaflet carefully before
New Zealand Consumer Medicine Information. It does not take the place of talking to your doctor or pharmacist.
 New Zealand Consumer Medicine Information Antabuse Disulfiram 200mg tablets (die-sul-fear-am) What is in this leaflet? This leaflet answers some common questions about Antabuse. It does not contain all
New Zealand Consumer Medicine Information Antabuse Disulfiram 200mg tablets (die-sul-fear-am) What is in this leaflet? This leaflet answers some common questions about Antabuse. It does not contain all
IMPORTANT: PLEASE READ
 PART III: CONSUMER INFORMATION combined hepatitis A (inactivated) and hepatitis B (recombinant) vaccine This leaflet is part III of a three-part "Product Monograph" published when was approved for sale
PART III: CONSUMER INFORMATION combined hepatitis A (inactivated) and hepatitis B (recombinant) vaccine This leaflet is part III of a three-part "Product Monograph" published when was approved for sale
European Medicines Agency recommends restricting use of trimetazidine-containing medicines
 22 June 2012 EMA/CHMP/417861/2012 Press Office Press release European Medicines Agency recommends restricting use of trimetazidine-containing medicines Restricted indication for patients with stable angina
22 June 2012 EMA/CHMP/417861/2012 Press Office Press release European Medicines Agency recommends restricting use of trimetazidine-containing medicines Restricted indication for patients with stable angina
MEDICATION GUIDE WELLBUTRIN (WELL byu-trin) (bupropion hydrochloride) Tablets
 MEDICATION GUIDE WELLBUTRIN (WELL byu-trin) (bupropion hydrochloride) Tablets Read this Medication Guide carefully before you start using WELLBUTRIN and each time you get a refill. There may be new information.
MEDICATION GUIDE WELLBUTRIN (WELL byu-trin) (bupropion hydrochloride) Tablets Read this Medication Guide carefully before you start using WELLBUTRIN and each time you get a refill. There may be new information.
Summary of Product Characteristics
 1 NAME OF THE MEDICINAL PRODUCT Librium 10mg Hard capsule Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each capsule contains 10 mg of chlordiazepoxide hydrochloride. Excipients:
1 NAME OF THE MEDICINAL PRODUCT Librium 10mg Hard capsule Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each capsule contains 10 mg of chlordiazepoxide hydrochloride. Excipients:
Package leaflet: Information for the user. Ondemet 4mg and 8mg Tablets (Ondansetron)
 Package leaflet: Information for the user Ondemet 4mg and 8mg Tablets (Ondansetron) Read all of this leaflet carefully before you start taking this medicine because it contains important information for
Package leaflet: Information for the user Ondemet 4mg and 8mg Tablets (Ondansetron) Read all of this leaflet carefully before you start taking this medicine because it contains important information for
PRESCRIBING INFORMATION
 PRESCRIBING INFORMATION EPIPEN (epinephrine) Auto-Injector 0.3 mg EpiPen = one dose of 0.30 mg epinephrine (USP, 1:1000, 0.3 ml) EPIPEN JR (epinephrine) Auto-Injector 0.15 mg EpiPen Jr = one dose of 0.15
PRESCRIBING INFORMATION EPIPEN (epinephrine) Auto-Injector 0.3 mg EpiPen = one dose of 0.30 mg epinephrine (USP, 1:1000, 0.3 ml) EPIPEN JR (epinephrine) Auto-Injector 0.15 mg EpiPen Jr = one dose of 0.15
SUMMARY OF PRODUCT CHARACTERISTICS. Buprenovet 0.3 mg/ml Solution for Injection for Dogs and Cats (AT, DE)
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Buprecare 0.3 mg/ml Solution for Injection for Dogs and Cats (UK, BE, FR, IE, LU, NL, ES) Buprenovet 0.3 mg/ml Solution for
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Buprecare 0.3 mg/ml Solution for Injection for Dogs and Cats (UK, BE, FR, IE, LU, NL, ES) Buprenovet 0.3 mg/ml Solution for
Nurse Initiated Medications Procedure
 1. Purpose This Procedure is performed as a means of ensuring the safe administration of therapeutic medication to patients in accordance with all legislative and regulatory requirements. 2. Application
1. Purpose This Procedure is performed as a means of ensuring the safe administration of therapeutic medication to patients in accordance with all legislative and regulatory requirements. 2. Application
IMIGRAN injection is also used for treatment of cluster headache. Before you use your IMIGRAN injection. When you must not use it
 Imigran Injection Sumatriptan succinate 6mg/0.5mL Consumer Medicine Information What is in this leaflet? Please read this leaflet carefully before you start using. This leaflet answers some common questions
Imigran Injection Sumatriptan succinate 6mg/0.5mL Consumer Medicine Information What is in this leaflet? Please read this leaflet carefully before you start using. This leaflet answers some common questions
Summary of Product Characteristics
 1 NAME OF THE MEDICINAL PRODUCT Summary of Product Characteristics Anapen Junior 150 micrograms in 0.3ml solution for injection in a pre- filled syringe. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each
1 NAME OF THE MEDICINAL PRODUCT Summary of Product Characteristics Anapen Junior 150 micrograms in 0.3ml solution for injection in a pre- filled syringe. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Trihexyphenidyl 2mg Tablets BP 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 2 mg of trihexyphenidyl hydrochloride BP
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Trihexyphenidyl 2mg Tablets BP 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 2 mg of trihexyphenidyl hydrochloride BP
Lidocaine 2% w/v solution for injection Summary of Product Characteristics
 Lidocaine 2% w/v solution for injection Summary of Product Characteristics 1. NAME OF THE MEDICINAL PRODUCT Lidocaine 2% w/v solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each 1 ml
Lidocaine 2% w/v solution for injection Summary of Product Characteristics 1. NAME OF THE MEDICINAL PRODUCT Lidocaine 2% w/v solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each 1 ml
STRATTERA (Stra-TAIR-a)
 1 PV 5859 AMP MEDICATION GUIDE STRATTERA (Stra-TAIR-a) (atomoxetine) Capsules Read the Medication Guide that comes with STRATTERA before you or your child starts taking it and each time you get a refill.
1 PV 5859 AMP MEDICATION GUIDE STRATTERA (Stra-TAIR-a) (atomoxetine) Capsules Read the Medication Guide that comes with STRATTERA before you or your child starts taking it and each time you get a refill.
DEXTROMETHORPHAN HYDROBROMIDE 10MG/5ML ORAL SOLUTION PL 17907/0314
 DEXTROMETHORPHAN HYDROBROMIDE 10MG/5ML ORAL SOLUTION PL 17907/0314 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 10 Summary of Product Characteristics
DEXTROMETHORPHAN HYDROBROMIDE 10MG/5ML ORAL SOLUTION PL 17907/0314 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 10 Summary of Product Characteristics
VALIUM PRODUCT MONOGRAPH. diazepam. 5 mg Tablets. Anxiolytic-sedative. Date of Revision: February 7, 2014
 PRODUCT MONOGRAPH VALIUM diazepam 5 mg Tablets Anxiolytic-sedative Hoffmann-La Roche Ltd. 7070 Mississauga Road Mississauga, Ontario L5N 5M8 Date of Revision: February 7, 2014 www.rochecanada.com Submission
PRODUCT MONOGRAPH VALIUM diazepam 5 mg Tablets Anxiolytic-sedative Hoffmann-La Roche Ltd. 7070 Mississauga Road Mississauga, Ontario L5N 5M8 Date of Revision: February 7, 2014 www.rochecanada.com Submission
Trileptal (Oxcarbazepine)
 Brand and Generic Names: Trileptal Tablets: 150mg, 300mg, 600mg Liquid Suspension: 300mg/5mL Generic name: oxcarbazepine What is Trileptal and what does it treat? Trileptal (Oxcarbazepine) Oxcarbazepine
Brand and Generic Names: Trileptal Tablets: 150mg, 300mg, 600mg Liquid Suspension: 300mg/5mL Generic name: oxcarbazepine What is Trileptal and what does it treat? Trileptal (Oxcarbazepine) Oxcarbazepine
Adrenaline (epinephrine) 1:1000 Injection BP Summary of Product Characteristics
 Adrenaline (epinephrine) 1:1000 Injection BP Summary of Product Characteristics 1. NAME OF THE MEDICINAL PRODUCT Adrenaline (epinephrine) 1:1000 Injection BP 2. QUALITATIVE AND QUANTITATIVE COMPOSITION
Adrenaline (epinephrine) 1:1000 Injection BP Summary of Product Characteristics 1. NAME OF THE MEDICINAL PRODUCT Adrenaline (epinephrine) 1:1000 Injection BP 2. QUALITATIVE AND QUANTITATIVE COMPOSITION
Levophed Norepinephrine Bitartrate Injection, USP
 Levophed Norepinephrine Bitartrate Injection, USP R x only DESCRIPTION Norepinephrine (sometimes referred to as l-arterenol/levarterenol or l-norepinephrine) is a sympathomimetic amine which differs from
Levophed Norepinephrine Bitartrate Injection, USP R x only DESCRIPTION Norepinephrine (sometimes referred to as l-arterenol/levarterenol or l-norepinephrine) is a sympathomimetic amine which differs from
PACKAGE LEAFLET: INFORMATION FOR THE USER Omeprazol XXX 40 mg powder for solution for infusion omeprazole
 PACKAGE LEAFLET: INFORMATION FOR THE USER Omeprazol XXX 40 mg powder for solution for infusion omeprazole Read all of this leaflet carefully before you start using this medicine. - Keep this leaflet. You
PACKAGE LEAFLET: INFORMATION FOR THE USER Omeprazol XXX 40 mg powder for solution for infusion omeprazole Read all of this leaflet carefully before you start using this medicine. - Keep this leaflet. You
DATA SHEET ADRENALINE ACID TARTRATE INJECTION
 DATA SHEET ADRENALINE ACID TARTRATE INJECTION 1:1 000 (1 mg/ml) Presentation Adrenaline injection contains no antimicrobial agent. It should be used only once and any residue discarded. It is a clear,
DATA SHEET ADRENALINE ACID TARTRATE INJECTION 1:1 000 (1 mg/ml) Presentation Adrenaline injection contains no antimicrobial agent. It should be used only once and any residue discarded. It is a clear,
