Direct Energy Conversion: Chemistry, Physics, Materials Science and Thermoelectrics
|
|
|
- Claud Holmes
- 9 years ago
- Views:
Transcription
1 Direct Energy Conversion: Chemistry, Physics, Materials Science and Thermoelectrics Mercouri G Kanatzidis MURI KANATZIDIS KANATZIDIS MSU, MSU, CHEMISTRY CHEMISTRY Solid Solid State State Chemistry Chemistry Synthesis, Synthesis, Discovery Discovery TIM HOGAN MSU, E. ENGINEERING Measurements, Devices HAROLD SCHOCK MSU, Mechanical Engineering Applications S. D. MAHANTI MSU, PHYSICS Theory CTIRAD UHER Univ of of Michigan Physics Measurements ELDON CASE MSU, Mechanical Engineering Metallurgy American Physical Society Meeting, Baltimore March 2006
2 Heat to Electrical Energy Directly Up to 20% conversion efficiency with right materials Schock group
3 Thermoelectric applications Waste heat recovery Automobiles Over the road trucks Utilities Chemical plants Space power Remote Power Generation Solar energy Geothermal power generation Direct nuclear to electrical
4 U.S. Energy Flow, 1999 web site: Given that ~60% of energy becomes waste heat, even a 10% capture and conversion to useful forms can have huge impact on overall energy utilization
5 How does it work? TE devices have no moving parts, no noise, reliable
6 Figure of Merit δ = 0.0 δ = 0.1 electrical conductivity thermopower η δ = 1.1 ZT = σ S2 T κ total Total thermal conductivity Power factor σ S 2 ZT avg δ=r c /R For T h = 800K T c = 300K
7 Today s situation The most efficient materials today for power generation: PbTe and TAGS (TeSbGeAg alloy) The most efficient material for cooling Bi 2 Te 3 PbTe: ZT~0.8 at 800 K (n-type) TAGS: ZT~ K (p-type) Bi 2 Te 3-x Se x : ZT~1 at 300 K Further improvements are needed. New materials needed Quantum Dot Layers in thin MBE-grown PbSe/PbTe superlattices (Harman et al, ZT~3) PbTe PbSe 20 nm dot Hicks LD, Dresselhaus MS Phys. Rev. B (24): 16631
8 Some promising systems under investigation half-heusler alloys (ZrNiSn) Zn 4 Sb 3 Clathrates Skutterudites (CoSb 3 ) Bulk nanocomposites based on PbTe Bulk nanocomposites based on Si-Ge AgSbTe 2 /PbTe, NaSbTe 2 /PbTe See March 2006 issue of MRS Bulletin
9 ZT and Electronic Structure Isotropic structure Z max T γ 3/ 2 τ ( ) z r+ 1/ 2 κ latt m m x m y e Anisotropic structure For acoustic phonon scattering r=-1/2 m= effective mass τ=scattering time r= scattering parameter κ latt = lattice thermal conductivity T = temperature E E γ= band degeneracy Ef Large γ comes with (a) high symmetry e.g. rhombohedral, cubic (b) off-center band extrema k k k Complex electronic structure
10 Selection criteria for candidate materials Narrow band-gap semiconductors Heavy elements High µ, low κ Large unit cell, complex structure low κ Highly anisotropic or highly symmetric Complex compositions low κ, complex electronic structure
11 Chemistry as a source of materials Investigating the System: A 2 Q + PbQ + M 2 Q 3 > (A 2 Q) n (PbQ) m (M 2 Q 3 ) p Map generates target compounds Cubic materials AB m MQ 2+m Phases shown are promising new TE Materials A 2 Q Rb 0.5 Bi 1.83 Te 3 A=Ag, K, Rb, Cs M=Sb, Bi Q=Se, Te PbQ APb 2 Bi 3 Te 7 A 1+x Pb 4-2x Bi 7+x Se 15 Pb 6 Bi 2 Se 9 Pb 5 Bi 6 Se 14 Pb 5 Bi 12 Se 23 β-k 2 Bi 8 Se 13 CsBi 4 Te 6 Bi 2 Q 3
12 Our first contact with cubic AgPb m SbTe 2+m AgBi 3 S 5, KPbBi 9 Se 13, KPb 4 Sb 7 Se 15 CsPbBi 3 Te 6, CsPb 2 Bi 3 Te 7, CsPb 3 Bi 3 Te 8, RbPbBi 3 Te 6, RbPb 2 Bi 3 Te 7, RbPb 3 Bi 3 Te 8, KPbBiSe 3, K 2 PbBi 2 Se 5 K 2 Pb 3 Bi 2 Te 7, KPb 4 SbTe 6
13 AgPb m SbTe 2+m (LAST-m) AgPb m (Sb,Bi)Te 2+m (BLAST-m) Pb Te Sb Ag 268 Unit cell volume (Å) Vegard's law m value (1) (a) Rodot, H. Compt. Rend. 1959, 249, (2) (a) Rosi, F. D.; Hockings, E. S.; Lindenblad, N. E. Adv. Energy Convers. 1961, 1, 151.
14 (LAST-18) Ag 1-x Pb 18 SbTe 20 : Tunable properties Changing x Thermopower (µv/k) Comparison of Thermopower Ag0.73 (KF2207R2b) Ag0.76 (KF2228R3A2) Ag0.78 (KF2213R1B) Ag0.80 (KF2213R2C) Ag0.82 (KF2229R1A) Ag0.82 (KF2229R1B) Ag0.82 (KF2229R1C) Ag0.82 (KF2229R1D) Temperature (K) Comparison of Power Factors ZT Ag0.73 (KF2207R2b) Ag0.76 (KF2228R3A2) Ag0.78 (KF2213R1B) Ag0.80 (KF2213R2C) Ag0.82 (KF2229R1A) Ag0.82 (KF2229R1B) Ag0.82 (KF2229R1C) Ag0.82 (KF2229R1D) Comparison of ZT Temperature, K Power Factor S 2 σ (µw/cm K 2 ) Ag0.73 (KF2207R2b) Ag0.76 (KF2228R3A2) Ag0.78 (KF2213R1B) Ag0.80 (KF2213R2C) Ag0.82 (KF2229R1A) Ag0.82 (KF2229R1B) Ag0.82 (KF2229R1C) Ag0.82 (KF2229R1D) Temperature (K)
15 Synthesis: Heating cooling profiles rock T, K ºC Cool to 50 ºC in 72 h 13 deg/h Ingot properties very sensitive to cooling profile Gravity induced inhomogeneity LAST-18 time, h T, K 1000 ºC rock Cool to 50 ºC in 24 h time, h Wernick, J. H.. Metallurg. Soc. Conf. Proc. (1960), R. G. Maier Z. Metallkunde 1963, 311
16 Samples cooled slowly from liquid to solid Strongly varying composition from top to m= 8 bottom. m= 14 Strongly varying properties from top to bottom. m= 18 Sweet spot exists with very high ZT. m= 25 Mechanical properties weak. m= 40 σ (S/cm) S (µv/k) Strong composition grading along ingot
17 Properties of Ag 1-x Pb 18 SbTe Lattice thermal conductivity σ (S/cm) Temperature, K Lattice Thermal Conductivity (W/mK) S (µv/k) PbTe LAST W/mK (Harman PbTe/PbSe superlattice) Temperature (K)
18 Ag 1-x Pb 18 SbTe Ag 0.86 Pb 18 SbTe ZT Temperature, K Hsu KF, Loo S, Guo F, Chen W, Dyck JS, Uher C, Hogan T, Polychroniadis EK, Kanatzidis MG Science, 2004, 303, 818
19 What is the origin of the TE properties of AgPb m SbTe m+2 systems?
20 HRTEM Coherently embedded nanocrystals Pb- rich region 8-10 nm Ag-Sb rich region Polychroniadis, Frangis, 2004 LAST-18 κ latt =1.2 W/m-K at 300 K PbTe κ latt =2.2 W/m-K at 300 K
21 Coherent compositional fluctuations in AgPb m SbTe m+2 FCC lattice 10 nm Ag, Sb, Pb ordering
22 Driving force for segregation Ag + /Sb 3+ pair: stable Ag Te Pb Te Pb Te Pb Te Pb Te Pb Te Sb Te Pb Te Pb Te Pb Te Pb Te Ag Te Pb Te Pb Te Pb Te Pb Te Pb Te Pb Te Pb Te Pb Te Pb Te Pb Te Sb Te Pb Te Pb Te Ag Te Ag Te Pb Te Pb Te Pb Te Sb Te Pb Pb Te Sb Te Pb Te Pb Te Pb Te Pb Te Pb Te Te Pb Te Pb Te Pb Te Dissociated state..unstable Associated state..stable Any +1/+3 pair
23 LAST-m Solid solutions in (AgSbTe 2 ) 1-x (PbTe) x Lattice Lattice Thermal Thermal Conductivity (mw/cm K) (W/cmK) Klemmens-Drabble theory m=65 m=62 m~62 Random alloy m~30 m=18 m=10 m=18 Experimental Points below theoretical AgSbTe2 PbTe Mol % PbTe T. Irie Jap. J. Appl. Phys. 1966, 5, 854
24
25 P-type materials, LASTT1000 (LASTT-m) Ag(Pb 1-x Sn x ) m SbTe 2+m Sn atoms act as acceptors Ag atoms act as acceptors Sb atoms act as donors e.g AgPb 10 Sn 8 SbTe 20, Ag x Pb 7 Sn 3 Sb y Te 12, Very low lattice thermal conductivity Good homogeneity Temperature ( o C) t (hours) 2 hours rocking Ag 0.3 Pb 7 Sn 3 Sb 0.1 Te 12 Intensity (arb. units) Theta
26 LASTT-16 Very low lattice thermal conductivity κ latt =0.5 W/m K at 650 K Androulakis, Hsu, Hogan, Uher, Kanatzidis Advanced Mater. 2006, in press
27 LASTT-16: AgPb 12 Sn 4 Sb 0.4 Te Seebeck (uv/k) Temperature, K Androulakis, Hsu, Hogan, Uher, Kanatzidis Advanced Mater. 2006, in press
28 Figure of Merit LASTT (p-type) ZT Ag 0.5 Pb 6 Sn 2 Sb 0.2 Te 10 Ag 0.9 Pb 5 Sn 3 Sb 0.7 Te 10 Ag 0.6 Pb 7 Sn 3 Sb 0.2 Te 12 (b) LASTT TAGS PbTe Temperature (K) Androulakis, Hsu, Hogan, Uher, Kanatzidis Advanced Mater. 2006, in press
29 NaPb 20 SbTe 22 (SALT-20) (A) Lattice Thermal conductivity (W/mK) 2 (B) 1.5 Pb Sn Te not-nano 1 PbTe Se Na Pb SbTe 1-x nano Temperature, K 50 nm Poudeu, Hogan, Kanatzidis Angew. Chemie, 2006,
30 SALT-20 Poudeu, Hogan, Kanatzidis Angew. Chemie, 2006,
31 State of the art - bulk LAST ZT 1.5 Tl 9 BiTe 6 NaPb SbTe LASTT TAGS 1 CsBi 4 Te 6 Bi 2 Te 3 Ce 0.9 Fe 3 CoSb 12 SiGe PbTe Temperature, K
32 Conclusions New approaches are succeeding in raising ZT The (A 2 Q) n (PbQ) m (Bi 2 Q 3 ) p (Q=Se, Te) system is a rich source of new materials Several new promising compounds identified strongly anisotropic cubic nanostructured LAST, LASTT and SALT family of materials nanostructured superior ZT Strong thermal conductivity reduction achieved through nanostructuring Doping studies and processing conditions are important in ZT optimization
33 Outlook Further progress is expected on the TE figure of merit Fundamental challenge: Translate current theoretical physical predictions on how to enhance Power Factor (σ S 2 ) into actual chemistry in the laboratory Achieve minimum thermal conductivity (~0.3 W/mK) in a bulk (nano)crystalline TE material Research in new materials should focus on Understanding and controlling carrier scattering Controlling nanostructuring to manipulate phonon propagation Discovering new compounds Long term: Waste heat recovery and conversion could be impacted on a massive scale with low cost materials if ZT>2-3. Thermoelectrics could help utilize existing depletable energy resources more effectively Thermoelectrics could also play role in renewable energy (e.g. solar, etc)
34 Collaborators Tim Hogan, Dept of Electrical Engineering, MSU S. D. (Bhanu) Mahanti, Dept. of Physics, MSU Ctirad Uher, Dept. of Physics, U of Michigan Simon Billinge, Physics, MSU Eldon Case, MSU Harold Schock, MSU Bruce Cook, Iowa State Terry Tritt, Clemson U Art Schultz, Argonne NL
35 TE Research group Dr Duck young Chung Joseph Sootsman Dr Kuei fang Hsu Dr Eric Quarez Aurelie Guegen Ferdinand Poudeu Jun-Ho Kim Dr John Androulakis
On-Chip Sensing of Thermoelectric Thin Film s Merit
 Sensors 2015, 15, 17232-17240; doi:10.3390/s150717232 Article OPEN ACCESS sensors ISSN 1424-8220 www.mdpi.com/journal/sensors On-Chip Sensing of Thermoelectric Thin Film s Merit Zhigang Xiao 1, * and Xiaoshan
Sensors 2015, 15, 17232-17240; doi:10.3390/s150717232 Article OPEN ACCESS sensors ISSN 1424-8220 www.mdpi.com/journal/sensors On-Chip Sensing of Thermoelectric Thin Film s Merit Zhigang Xiao 1, * and Xiaoshan
Solid State Detectors = Semi-Conductor based Detectors
 Solid State Detectors = Semi-Conductor based Detectors Materials and their properties Energy bands and electronic structure Charge transport and conductivity Boundaries: the p-n junction Charge collection
Solid State Detectors = Semi-Conductor based Detectors Materials and their properties Energy bands and electronic structure Charge transport and conductivity Boundaries: the p-n junction Charge collection
Thermoelectric Conversion of Waste Heat to Electricity in an IC Engine Powered Vehicle Presented by:
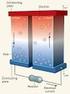 Thermoelectric Conversion of Waste Heat to Electricity in an IC Engine Powered Vehicle Presented by: IOWA STATE UNIVERSITY Harold Schock Dept. of Mechanical Engineering Michigan State University 8/25/2005
Thermoelectric Conversion of Waste Heat to Electricity in an IC Engine Powered Vehicle Presented by: IOWA STATE UNIVERSITY Harold Schock Dept. of Mechanical Engineering Michigan State University 8/25/2005
CVD SILICON CARBIDE. CVD SILICON CARBIDE s attributes include:
 CVD SILICON CARBIDE CVD SILICON CARBIDE is the ideal performance material for design engineers. It outperforms conventional forms of silicon carbide, as well as other ceramics, quartz, and metals in chemical
CVD SILICON CARBIDE CVD SILICON CARBIDE is the ideal performance material for design engineers. It outperforms conventional forms of silicon carbide, as well as other ceramics, quartz, and metals in chemical
Chapter 5. Second Edition ( 2001 McGraw-Hill) 5.6 Doped GaAs. Solution
 Chapter 5 5.6 Doped GaAs Consider the GaAs crystal at 300 K. a. Calculate the intrinsic conductivity and resistivity. Second Edition ( 2001 McGraw-Hill) b. In a sample containing only 10 15 cm -3 ionized
Chapter 5 5.6 Doped GaAs Consider the GaAs crystal at 300 K. a. Calculate the intrinsic conductivity and resistivity. Second Edition ( 2001 McGraw-Hill) b. In a sample containing only 10 15 cm -3 ionized
Exciton dissociation in solar cells:
 Exciton dissociation in solar cells: Xiaoyang Zhu Department of Chemistry University of Minnesota, Minneapolis t (fs) 3h! E, k h! Pc Bi e - 1 Acknowledgement Organic semiconductors: Mutthias Muntwiler,
Exciton dissociation in solar cells: Xiaoyang Zhu Department of Chemistry University of Minnesota, Minneapolis t (fs) 3h! E, k h! Pc Bi e - 1 Acknowledgement Organic semiconductors: Mutthias Muntwiler,
Matter, Materials, Crystal Structure and Bonding. Chris J. Pickard
 Matter, Materials, Crystal Structure and Bonding Chris J. Pickard Why should a theorist care? Where the atoms are determines what they do Where the atoms can be determines what we can do Overview of Structure
Matter, Materials, Crystal Structure and Bonding Chris J. Pickard Why should a theorist care? Where the atoms are determines what they do Where the atoms can be determines what we can do Overview of Structure
CLASS TEST GRADE 11. PHYSICAL SCIENCES: CHEMISTRY Test 6: Chemical change
 CLASS TEST GRADE PHYSICAL SCIENCES: CHEMISTRY Test 6: Chemical change MARKS: 45 TIME: hour INSTRUCTIONS AND INFORMATION. Answer ALL the questions. 2. You may use non-programmable calculators. 3. You may
CLASS TEST GRADE PHYSICAL SCIENCES: CHEMISTRY Test 6: Chemical change MARKS: 45 TIME: hour INSTRUCTIONS AND INFORMATION. Answer ALL the questions. 2. You may use non-programmable calculators. 3. You may
Lecture 2 - Semiconductor Physics (I) September 13, 2005
 6.012 - Microelectronic Devices and Circuits - Fall 2005 Lecture 2-1 Lecture 2 - Semiconductor Physics (I) September 13, 2005 Contents: 1. Silicon bond model: electrons and holes 2. Generation and recombination
6.012 - Microelectronic Devices and Circuits - Fall 2005 Lecture 2-1 Lecture 2 - Semiconductor Physics (I) September 13, 2005 Contents: 1. Silicon bond model: electrons and holes 2. Generation and recombination
Thermal unobtainiums? The perfect thermal conductor and the perfect thermal insulator
 Thermal unobtainiums? The perfect thermal conductor and the perfect thermal insulator David G. Cahill Materials Research Lab and Department of Materials Science and Engineering, U. of Illinois Gratefully
Thermal unobtainiums? The perfect thermal conductor and the perfect thermal insulator David G. Cahill Materials Research Lab and Department of Materials Science and Engineering, U. of Illinois Gratefully
Lecture 4: Thermodynamics of Diffusion: Spinodals
 Materials Science & Metallurgy Master of Philosophy, Materials Modelling, Course MP6, Kinetics and Microstructure Modelling, H. K. D. H. Bhadeshia Lecture 4: Thermodynamics of Diffusion: Spinodals Fick
Materials Science & Metallurgy Master of Philosophy, Materials Modelling, Course MP6, Kinetics and Microstructure Modelling, H. K. D. H. Bhadeshia Lecture 4: Thermodynamics of Diffusion: Spinodals Fick
The Fundamentals of Thermoelectrics
 The Fundamentals of Thermoelectrics A bachelor s laboratory practical Contents 1 An introduction to thermoelectrics 1 2 The thermocouple 4 3 The Peltier device 5 3.1 n- and p-type Peltier elements..................
The Fundamentals of Thermoelectrics A bachelor s laboratory practical Contents 1 An introduction to thermoelectrics 1 2 The thermocouple 4 3 The Peltier device 5 3.1 n- and p-type Peltier elements..................
Chemical Synthesis. Overview. Chemical Synthesis of Nanocrystals. Self-Assembly of Nanocrystals. Example: Cu 146 Se 73 (PPh 3 ) 30
 Chemical Synthesis Spontaneous organization of molecules into stable, structurally well-defined aggregates at the nanometer length scale. Overview The 1-100 nm nanoscale length is in between traditional
Chemical Synthesis Spontaneous organization of molecules into stable, structurally well-defined aggregates at the nanometer length scale. Overview The 1-100 nm nanoscale length is in between traditional
2012 DOE 3 rd Thermoelectrics Applications Worshop March 22, 2012
 Nanostructured High Temperature Bulk Thermoelectric Energy Conversion for Efficient Waste Heat Recovery PI - Chris Caylor, GMZ Director of Thermoelectric Systems GMZ Team: Bed Poudel, Giri Joshi, Jonathan
Nanostructured High Temperature Bulk Thermoelectric Energy Conversion for Efficient Waste Heat Recovery PI - Chris Caylor, GMZ Director of Thermoelectric Systems GMZ Team: Bed Poudel, Giri Joshi, Jonathan
Chapter Outline. Diffusion - how do atoms move through solids?
 Chapter Outline iffusion - how do atoms move through solids? iffusion mechanisms Vacancy diffusion Interstitial diffusion Impurities The mathematics of diffusion Steady-state diffusion (Fick s first law)
Chapter Outline iffusion - how do atoms move through solids? iffusion mechanisms Vacancy diffusion Interstitial diffusion Impurities The mathematics of diffusion Steady-state diffusion (Fick s first law)
THERMAL TO ELECTRIC ENERGY CONVERSION
 THERMAL TO ELECTRIC ENERGY CONVERSION PETER L. HAGELSTEIN Research Laboratory of Electronics, Massachusetts Institute of Technology, Cambridge, MA 02139,USA E-mail: [email protected] As research in the area
THERMAL TO ELECTRIC ENERGY CONVERSION PETER L. HAGELSTEIN Research Laboratory of Electronics, Massachusetts Institute of Technology, Cambridge, MA 02139,USA E-mail: [email protected] As research in the area
Hard Condensed Matter WZI
 Hard Condensed Matter WZI Tom Gregorkiewicz University of Amsterdam VU-LaserLab Dec 10, 2015 Hard Condensed Matter Cluster Quantum Matter Optoelectronic Materials Quantum Matter Amsterdam Mark Golden Anne
Hard Condensed Matter WZI Tom Gregorkiewicz University of Amsterdam VU-LaserLab Dec 10, 2015 Hard Condensed Matter Cluster Quantum Matter Optoelectronic Materials Quantum Matter Amsterdam Mark Golden Anne
Chemistry B11 Chapter 4 Chemical reactions
 Chemistry B11 Chapter 4 Chemical reactions Chemical reactions are classified into five groups: A + B AB Synthesis reactions (Combination) H + O H O AB A + B Decomposition reactions (Analysis) NaCl Na +Cl
Chemistry B11 Chapter 4 Chemical reactions Chemical reactions are classified into five groups: A + B AB Synthesis reactions (Combination) H + O H O AB A + B Decomposition reactions (Analysis) NaCl Na +Cl
Graphene a material for the future
 Graphene a material for the future by Olav Thorsen What is graphene? What is graphene? Simply put, it is a thin layer of pure carbon What is graphene? Simply put, it is a thin layer of pure carbon It has
Graphene a material for the future by Olav Thorsen What is graphene? What is graphene? Simply put, it is a thin layer of pure carbon What is graphene? Simply put, it is a thin layer of pure carbon It has
CHAPTER 9 Part 1. = 5 wt% Sn-95 wt% Pb C β. = 98 wt% Sn-2 wt% Pb. = 77 wt% Ag-23 wt% Cu. = 51 wt% Zn-49 wt% Cu C γ. = 58 wt% Zn-42 wt% Cu
 CHAPTER 9 Part 1 9.5 This problem asks that we cite the phase or phases present for several alloys at specified temperatures. (a) For an alloy composed of 15 wt% Sn-85 wt% Pb and at 100 C, from Figure
CHAPTER 9 Part 1 9.5 This problem asks that we cite the phase or phases present for several alloys at specified temperatures. (a) For an alloy composed of 15 wt% Sn-85 wt% Pb and at 100 C, from Figure
The Physics of Energy sources Renewable sources of energy. Solar Energy
 The Physics of Energy sources Renewable sources of energy Solar Energy B. Maffei [email protected] Renewable sources 1 Solar power! There are basically two ways of using directly the radiative
The Physics of Energy sources Renewable sources of energy Solar Energy B. Maffei [email protected] Renewable sources 1 Solar power! There are basically two ways of using directly the radiative
6.772/SMA5111 - Compound Semiconductors Lecture 1 - The Compound Semiconductor Palette - Outline Announcements
 6.772/SMA5111 - Compound Semiconductors Lecture 1 - The Compound Semiconductor Palette - Outline Announcements Handouts - General Information; Syllabus; Lecture 1 Notes Why are semiconductors useful to
6.772/SMA5111 - Compound Semiconductors Lecture 1 - The Compound Semiconductor Palette - Outline Announcements Handouts - General Information; Syllabus; Lecture 1 Notes Why are semiconductors useful to
PERIODIC TABLE OF GROUPS OF ELEMENTS Elements can be classified using two different schemes.
 1 PERIODIC TABLE OF GROUPS OF ELEMENTS Elements can be classified using two different schemes. Metal Nonmetal Scheme (based on physical properties) Metals - most elements are metals - elements on left
1 PERIODIC TABLE OF GROUPS OF ELEMENTS Elements can be classified using two different schemes. Metal Nonmetal Scheme (based on physical properties) Metals - most elements are metals - elements on left
SUPERCONDUCTIVITY. PH 318- Introduction to superconductors 1
 SUPERCONDUCTIVITY property of complete disappearance of electrical resistance in solids when they are cooled below a characteristic temperature. This temperature is called transition temperature or critical
SUPERCONDUCTIVITY property of complete disappearance of electrical resistance in solids when they are cooled below a characteristic temperature. This temperature is called transition temperature or critical
Introduction to Quantum Dot Nanocrystals and Nanocrystal Solids. Nuri Yazdani, 10.03.15
 Introduction to Quantum Dot Nanocrystals and Nanocrystal Solids Nuri Yazdani, 10.03.15 What is a QD Nanocrystal Time: ~15m What is a QD nanocrystal? Bulk Crystal Periodic lattice of atoms which extends
Introduction to Quantum Dot Nanocrystals and Nanocrystal Solids Nuri Yazdani, 10.03.15 What is a QD Nanocrystal Time: ~15m What is a QD nanocrystal? Bulk Crystal Periodic lattice of atoms which extends
Applied Physics of solar energy conversion
 Applied Physics of solar energy conversion Conventional solar cells, and how lazy thinking can slow you down Some new ideas *************************************************************** Our work on semiconductor
Applied Physics of solar energy conversion Conventional solar cells, and how lazy thinking can slow you down Some new ideas *************************************************************** Our work on semiconductor
Introduction OLEDs OTFTs OPVC Summary. Organic Electronics. Felix Buth. Walter Schottky Institut, TU München. Joint Advanced Student School 2008
 Felix Buth Joint Advanced Student School 2008 Outline 1 Introduction Difference organic/inorganic semiconductors From molecular orbitals to the molecular crystal 2 Organic Light Emitting Diodes Basic Principals
Felix Buth Joint Advanced Student School 2008 Outline 1 Introduction Difference organic/inorganic semiconductors From molecular orbitals to the molecular crystal 2 Organic Light Emitting Diodes Basic Principals
SEMICONDUCTOR I: Doping, semiconductor statistics (REF: Sze, McKelvey, and Kittel)
 SEMICONDUCTOR I: Doping, semiconductor statistics (REF: Sze, McKelvey, and Kittel) Introduction Based on known band structures of Si, Ge, and GaAs, we will begin to focus on specific properties of semiconductors,
SEMICONDUCTOR I: Doping, semiconductor statistics (REF: Sze, McKelvey, and Kittel) Introduction Based on known band structures of Si, Ge, and GaAs, we will begin to focus on specific properties of semiconductors,
Find a pair of elements in the periodic table with atomic numbers less than 20 that are an exception to the original periodic law.
 Example Exercise 6.1 Periodic Law Find the two elements in the fifth row of the periodic table that violate the original periodic law proposed by Mendeleev. Mendeleev proposed that elements be arranged
Example Exercise 6.1 Periodic Law Find the two elements in the fifth row of the periodic table that violate the original periodic law proposed by Mendeleev. Mendeleev proposed that elements be arranged
3. What would you predict for the intensity and binding energy for the 3p orbital for that of sulfur?
 PSI AP Chemistry Periodic Trends MC Review Name Periodic Law and the Quantum Model Use the PES spectrum of Phosphorus below to answer questions 1-3. 1. Which peak corresponds to the 1s orbital? (A) 1.06
PSI AP Chemistry Periodic Trends MC Review Name Periodic Law and the Quantum Model Use the PES spectrum of Phosphorus below to answer questions 1-3. 1. Which peak corresponds to the 1s orbital? (A) 1.06
Chem 115 POGIL Worksheet - Week 4 Moles & Stoichiometry
 Chem 115 POGIL Worksheet - Week 4 Moles & Stoichiometry Why? Chemists are concerned with mass relationships in chemical reactions, usually run on a macroscopic scale (grams, kilograms, etc.). To deal with
Chem 115 POGIL Worksheet - Week 4 Moles & Stoichiometry Why? Chemists are concerned with mass relationships in chemical reactions, usually run on a macroscopic scale (grams, kilograms, etc.). To deal with
Crystal Structure of Aluminum, Zinc, and their Alloys By: Omar Fajardo Sebastian Henao Devin Baines ENGR45, F2014, SRJC
 Crystal Structure of Aluminum, Zinc, and their Alloys By: Omar Fajardo Sebastian Henao Devin Baines ENGR45, F2014, SRJC Purpose The purpose of this experiment was to examine and observe the microstructure
Crystal Structure of Aluminum, Zinc, and their Alloys By: Omar Fajardo Sebastian Henao Devin Baines ENGR45, F2014, SRJC Purpose The purpose of this experiment was to examine and observe the microstructure
Introduction to Materials Science, Chapter 9, Phase Diagrams. Phase Diagrams. University of Tennessee, Dept. of Materials Science and Engineering 1
 Phase Diagrams University of Tennessee, Dept. of Materials Science and Engineering 1 Chapter Outline: Phase Diagrams Microstructure and Phase Transformations in Multicomponent Systems Definitions and basic
Phase Diagrams University of Tennessee, Dept. of Materials Science and Engineering 1 Chapter Outline: Phase Diagrams Microstructure and Phase Transformations in Multicomponent Systems Definitions and basic
Plate waves in phononic crystals slabs
 Acoustics 8 Paris Plate waves in phononic crystals slabs J.-J. Chen and B. Bonello CNRS and Paris VI University, INSP - 14 rue de Lourmel, 7515 Paris, France [email protected] 41 Acoustics 8 Paris We
Acoustics 8 Paris Plate waves in phononic crystals slabs J.-J. Chen and B. Bonello CNRS and Paris VI University, INSP - 14 rue de Lourmel, 7515 Paris, France [email protected] 41 Acoustics 8 Paris We
Solid-State Physics: The Theory of Semiconductors (Ch. 10.6-10.8) SteveSekula, 30 March 2010 (created 29 March 2010)
 Modern Physics (PHY 3305) Lecture Notes Modern Physics (PHY 3305) Lecture Notes Solid-State Physics: The Theory of Semiconductors (Ch. 10.6-10.8) SteveSekula, 30 March 2010 (created 29 March 2010) Review
Modern Physics (PHY 3305) Lecture Notes Modern Physics (PHY 3305) Lecture Notes Solid-State Physics: The Theory of Semiconductors (Ch. 10.6-10.8) SteveSekula, 30 March 2010 (created 29 March 2010) Review
Solar Cell Parameters and Equivalent Circuit
 9 Solar Cell Parameters and Equivalent Circuit 9.1 External solar cell parameters The main parameters that are used to characterise the performance of solar cells are the peak power P max, the short-circuit
9 Solar Cell Parameters and Equivalent Circuit 9.1 External solar cell parameters The main parameters that are used to characterise the performance of solar cells are the peak power P max, the short-circuit
Electronegativity and Polarity
 and Polarity N Goalby Chemrevise.org Definition: is the relative tendency of an atom in a molecule to attract electrons in a covalent bond to itself. is measured on the Pauling scale (ranges from 0 to
and Polarity N Goalby Chemrevise.org Definition: is the relative tendency of an atom in a molecule to attract electrons in a covalent bond to itself. is measured on the Pauling scale (ranges from 0 to
Energy Transport. Focus on heat transfer. Heat Transfer Mechanisms: Conduction Radiation Convection (mass movement of fluids)
 Energy Transport Focus on heat transfer Heat Transfer Mechanisms: Conduction Radiation Convection (mass movement of fluids) Conduction Conduction heat transfer occurs only when there is physical contact
Energy Transport Focus on heat transfer Heat Transfer Mechanisms: Conduction Radiation Convection (mass movement of fluids) Conduction Conduction heat transfer occurs only when there is physical contact
Arizona Institute for Renewable Energy & the Solar Power Laboratories
 Arizona Institute for Renewable Energy & the Solar Power Laboratories International Photovoltaic Reliability Workshop July 29-31, Tempe AZ Christiana Honsberg, Stephen Goodnick, Stuart Bowden Arizona State
Arizona Institute for Renewable Energy & the Solar Power Laboratories International Photovoltaic Reliability Workshop July 29-31, Tempe AZ Christiana Honsberg, Stephen Goodnick, Stuart Bowden Arizona State
9.11 Upon heating a lead-tin alloy of composition 30 wt% Sn-70 wt% Pb from 150 C and utilizing Figure
 9-13 9.8: 9.11 Upon heating a lead-tin alloy of composition 30 wt% Sn-70 wt% Pb from 150 C and utilizing Figure (a) The first liquid forms at the temperature at which a vertical line at this composition
9-13 9.8: 9.11 Upon heating a lead-tin alloy of composition 30 wt% Sn-70 wt% Pb from 150 C and utilizing Figure (a) The first liquid forms at the temperature at which a vertical line at this composition
Electronegativity and Polarity MAIN Idea A chemical bond s character is related to each atom s
 Section 8.5 Objectives Describe how electronegativity is used to determine bond type. Compare and contrast polar and nonpolar covalent bonds and polar and nonpolar molecules. Generalize about the characteristics
Section 8.5 Objectives Describe how electronegativity is used to determine bond type. Compare and contrast polar and nonpolar covalent bonds and polar and nonpolar molecules. Generalize about the characteristics
FYS3410 - Vår 2015 (Kondenserte fasers fysikk) http://www.uio.no/studier/emner/matnat/fys/fys3410/v15/index.html
 FYS3410 - Vår 015 (Kondenserte fasers fysikk) http://www.uio.no/studier/emner/matnat/fys/fys3410/v15/index.html Pensum: Introduction to Solid State Physics by Charles Kittel (Chapters 1-9 and 17, 18, 0,
FYS3410 - Vår 015 (Kondenserte fasers fysikk) http://www.uio.no/studier/emner/matnat/fys/fys3410/v15/index.html Pensum: Introduction to Solid State Physics by Charles Kittel (Chapters 1-9 and 17, 18, 0,
Chem 115 POGIL Worksheet - Week 4 Moles & Stoichiometry Answers
 Key Questions & Exercises Chem 115 POGIL Worksheet - Week 4 Moles & Stoichiometry Answers 1. The atomic weight of carbon is 12.0107 u, so a mole of carbon has a mass of 12.0107 g. Why doesn t a mole of
Key Questions & Exercises Chem 115 POGIL Worksheet - Week 4 Moles & Stoichiometry Answers 1. The atomic weight of carbon is 12.0107 u, so a mole of carbon has a mass of 12.0107 g. Why doesn t a mole of
Experimental Observation of the Quantum Anomalous Hall Effect in a Magnetic Topological Insulator
 Experimental Observation of the Quantum Anomalous Hall Effect in a Magnetic Topological Insulator Chang et al., Science 340, 167 (2013). Joseph Hlevyack, Hu Jin, Mazin Khader, Edward Kim Outline: Introduction:
Experimental Observation of the Quantum Anomalous Hall Effect in a Magnetic Topological Insulator Chang et al., Science 340, 167 (2013). Joseph Hlevyack, Hu Jin, Mazin Khader, Edward Kim Outline: Introduction:
Perfect Fluidity in Cold Atomic Gases?
 Perfect Fluidity in Cold Atomic Gases? Thomas Schaefer North Carolina State University 1 Hydrodynamics Long-wavelength, low-frequency dynamics of conserved or spontaneoulsy broken symmetry variables τ
Perfect Fluidity in Cold Atomic Gases? Thomas Schaefer North Carolina State University 1 Hydrodynamics Long-wavelength, low-frequency dynamics of conserved or spontaneoulsy broken symmetry variables τ
Peltier Application Note
 Peltier Application Note Early 19th century scientists, Thomas Seebeck and Jean Peltier, first discovered the phenomena that are the basis for today s thermoelectric industry. Seebeck found that if you
Peltier Application Note Early 19th century scientists, Thomas Seebeck and Jean Peltier, first discovered the phenomena that are the basis for today s thermoelectric industry. Seebeck found that if you
Chapter 8 - Chemical Equations and Reactions
 Chapter 8 - Chemical Equations and Reactions 8-1 Describing Chemical Reactions I. Introduction A. Reactants 1. Original substances entering into a chemical rxn B. Products 1. The resulting substances from
Chapter 8 - Chemical Equations and Reactions 8-1 Describing Chemical Reactions I. Introduction A. Reactants 1. Original substances entering into a chemical rxn B. Products 1. The resulting substances from
B) atomic number C) both the solid and the liquid phase D) Au C) Sn, Si, C A) metal C) O, S, Se C) In D) tin D) methane D) bismuth B) Group 2 metal
 1. The elements on the Periodic Table are arranged in order of increasing A) atomic mass B) atomic number C) molar mass D) oxidation number 2. Which list of elements consists of a metal, a metalloid, and
1. The elements on the Periodic Table are arranged in order of increasing A) atomic mass B) atomic number C) molar mass D) oxidation number 2. Which list of elements consists of a metal, a metalloid, and
Bonding Practice Problems
 NAME 1. When compared to H 2 S, H 2 O has a higher 8. Given the Lewis electron-dot diagram: boiling point because H 2 O contains stronger metallic bonds covalent bonds ionic bonds hydrogen bonds 2. Which
NAME 1. When compared to H 2 S, H 2 O has a higher 8. Given the Lewis electron-dot diagram: boiling point because H 2 O contains stronger metallic bonds covalent bonds ionic bonds hydrogen bonds 2. Which
1 Wetting your feet. 2 Scaling. 8.298 Lies / Check your understanding: Solutions
 1 Wetting your feet 1.1 Estimate how many liters are in a barrel of oil and how many barrels of oil the United States imports every year. A: A barrel may be a few feet high, so h 1m, and have a diameter
1 Wetting your feet 1.1 Estimate how many liters are in a barrel of oil and how many barrels of oil the United States imports every year. A: A barrel may be a few feet high, so h 1m, and have a diameter
47374_04_p25-32.qxd 2/9/07 7:50 AM Page 25. 4 Atoms and Elements
 47374_04_p25-32.qxd 2/9/07 7:50 AM Page 25 4 Atoms and Elements 4.1 a. Cu b. Si c. K d. N e. Fe f. Ba g. Pb h. Sr 4.2 a. O b. Li c. S d. Al e. H f. Ne g. Sn h. Au 4.3 a. carbon b. chlorine c. iodine d.
47374_04_p25-32.qxd 2/9/07 7:50 AM Page 25 4 Atoms and Elements 4.1 a. Cu b. Si c. K d. N e. Fe f. Ba g. Pb h. Sr 4.2 a. O b. Li c. S d. Al e. H f. Ne g. Sn h. Au 4.3 a. carbon b. chlorine c. iodine d.
DIFFUSION IN SOLIDS. Materials often heat treated to improve properties. Atomic diffusion occurs during heat treatment
 DIFFUSION IN SOLIDS WHY STUDY DIFFUSION? Materials often heat treated to improve properties Atomic diffusion occurs during heat treatment Depending on situation higher or lower diffusion rates desired
DIFFUSION IN SOLIDS WHY STUDY DIFFUSION? Materials often heat treated to improve properties Atomic diffusion occurs during heat treatment Depending on situation higher or lower diffusion rates desired
From Quantum to Matter 2006
 From Quantum to Matter 006 Why such a course? Ronald Griessen Vrije Universiteit, Amsterdam AMOLF, May 4, 004 vrije Universiteit amsterdam Why study quantum mechanics? From Quantum to Matter: The main
From Quantum to Matter 006 Why such a course? Ronald Griessen Vrije Universiteit, Amsterdam AMOLF, May 4, 004 vrije Universiteit amsterdam Why study quantum mechanics? From Quantum to Matter: The main
Crystal Structure of High Temperature Superconductors. Marie Nelson East Orange Campus High School NJIT Professor: Trevor Tyson
 Crystal Structure of High Temperature Superconductors Marie Nelson East Orange Campus High School NJIT Professor: Trevor Tyson Introduction History of Superconductors Superconductors are material which
Crystal Structure of High Temperature Superconductors Marie Nelson East Orange Campus High School NJIT Professor: Trevor Tyson Introduction History of Superconductors Superconductors are material which
CRYSTALLINE SOLIDS IN 3D
 CRYSTALLINE SOLIDS IN 3D Andrew Baczewski PHY 491, October 7th, 2011 OVERVIEW First - are there any questions from the previous lecture? Today, we will answer the following questions: Why should we care
CRYSTALLINE SOLIDS IN 3D Andrew Baczewski PHY 491, October 7th, 2011 OVERVIEW First - are there any questions from the previous lecture? Today, we will answer the following questions: Why should we care
Work and Energy. Work = Force Distance. Work increases the energy of an object. Energy can be converted back to work.
 Work and Energy Ch. 6 Work = Force Distance Work increases the energy of an object. Energy can be converted back to work. Therefore, energy and work have the same unit: Newton meter = Nm Energy per gram,
Work and Energy Ch. 6 Work = Force Distance Work increases the energy of an object. Energy can be converted back to work. Therefore, energy and work have the same unit: Newton meter = Nm Energy per gram,
Organic semiconductors
 Plastic (Organic) Solar Cells: Accomplishments, Challenges, and Strategies Sumit Chaudhary Assistant Professor Department of Electrical and Computer Engineering Materials Science and Engineering Iowa State
Plastic (Organic) Solar Cells: Accomplishments, Challenges, and Strategies Sumit Chaudhary Assistant Professor Department of Electrical and Computer Engineering Materials Science and Engineering Iowa State
NANO SILICON DOTS EMBEDDED SIO 2 /SIO 2 MULTILAYERS FOR PV HIGH EFFICIENCY APPLICATION
 NANO SILICON DOTS EMBEDDED SIO 2 /SIO 2 MULTILAYERS FOR PV HIGH EFFICIENCY APPLICATION Olivier Palais, Damien Barakel, David Maestre, Fabrice Gourbilleau and Marcel Pasquinelli 1 Outline Photovoltaic today
NANO SILICON DOTS EMBEDDED SIO 2 /SIO 2 MULTILAYERS FOR PV HIGH EFFICIENCY APPLICATION Olivier Palais, Damien Barakel, David Maestre, Fabrice Gourbilleau and Marcel Pasquinelli 1 Outline Photovoltaic today
High Open Circuit Voltage of MQW Amorphous Silicon Photovoltaic Structures
 High Open Circuit Voltage of MQW Amorphous Silicon Photovoltaic Structures ARGYRIOS C. VARONIDES Physics and EE Department University of Scranton 800 Linden Street, Scranton PA, 18510 United States Abstract:
High Open Circuit Voltage of MQW Amorphous Silicon Photovoltaic Structures ARGYRIOS C. VARONIDES Physics and EE Department University of Scranton 800 Linden Street, Scranton PA, 18510 United States Abstract:
Interface Reaction and Mechanical Properties of Lead-free Sn Zn Alloy/Cu Joints
 Materials Transactions, Vol. 43, No. 8 (2002) pp. 1797 to 1801 Special Issue on Lead-Free Electronics Packaging c 2002 The Japan Institute of Metals Interface Reaction and Mechanical Properties of Lead-free
Materials Transactions, Vol. 43, No. 8 (2002) pp. 1797 to 1801 Special Issue on Lead-Free Electronics Packaging c 2002 The Japan Institute of Metals Interface Reaction and Mechanical Properties of Lead-free
Types of Epitaxy. Homoepitaxy. Heteroepitaxy
 Epitaxy Epitaxial Growth Epitaxy means the growth of a single crystal film on top of a crystalline substrate. For most thin film applications (hard and soft coatings, optical coatings, protective coatings)
Epitaxy Epitaxial Growth Epitaxy means the growth of a single crystal film on top of a crystalline substrate. For most thin film applications (hard and soft coatings, optical coatings, protective coatings)
Thermal diffusivity and conductivity - an introduction to theory and practice
 Thermal diffusivity and conductivity - an introduction to theory and practice Utrecht, 02 October 2014 Dr. Hans-W. Marx Linseis Messgeräte GmbH Vielitzer Str. 43 D-95100 Selb / GERMANY www.linseis.com
Thermal diffusivity and conductivity - an introduction to theory and practice Utrecht, 02 October 2014 Dr. Hans-W. Marx Linseis Messgeräte GmbH Vielitzer Str. 43 D-95100 Selb / GERMANY www.linseis.com
Moles. Balanced chemical equations Molar ratios Mass Composition Empirical and Molecular Mass Predicting Quantities Equations
 Moles Balanced chemical equations Molar ratios Mass Composition Empirical and Molecular Mass Predicting Quantities Equations Micro World atoms & molecules Macro World grams Atomic mass is the mass of an
Moles Balanced chemical equations Molar ratios Mass Composition Empirical and Molecular Mass Predicting Quantities Equations Micro World atoms & molecules Macro World grams Atomic mass is the mass of an
Fall 2004 Ali Shakouri
 University of California at Santa Cruz Jack Baskin School of Engineering Electrical Engineering Department EE-145L: Properties of Materials Laboratory Lab 5b: Temperature Dependence of Semiconductor Conductivity
University of California at Santa Cruz Jack Baskin School of Engineering Electrical Engineering Department EE-145L: Properties of Materials Laboratory Lab 5b: Temperature Dependence of Semiconductor Conductivity
Lecture 24 - Surface tension, viscous flow, thermodynamics
 Lecture 24 - Surface tension, viscous flow, thermodynamics Surface tension, surface energy The atoms at the surface of a solid or liquid are not happy. Their bonding is less ideal than the bonding of atoms
Lecture 24 - Surface tension, viscous flow, thermodynamics Surface tension, surface energy The atoms at the surface of a solid or liquid are not happy. Their bonding is less ideal than the bonding of atoms
Magnetic Data Storage and Nanoparticles Ernie Chang
 Magnetic Data Storage and Nanoparticles Ernie Chang Introduction Magnetic storage, specifically in hard drives has advanced significantly since the first disk drive built in 1956 [1]. Interestingly enough,
Magnetic Data Storage and Nanoparticles Ernie Chang Introduction Magnetic storage, specifically in hard drives has advanced significantly since the first disk drive built in 1956 [1]. Interestingly enough,
Characterization and Qualitative Assessment of Silicon Wafers with Photoluminescence Imaging at Room Temperature
 Characterization and Qualitative Assessment of Silicon Wafers with Photoluminescence Imaging at Room Temperature SSW: B. Birkmann, A. Seidl, Chr. Lemke, M. Müller Business Unit/Department 2 Agenda - Motivation:
Characterization and Qualitative Assessment of Silicon Wafers with Photoluminescence Imaging at Room Temperature SSW: B. Birkmann, A. Seidl, Chr. Lemke, M. Müller Business Unit/Department 2 Agenda - Motivation:
Preparation of ZnS and SnS Nanopowders by Modified SILAR Technique
 Journal of Physical Sciences, Vol. 13, 009, 9-34 ISSN: 097-8791 : www.vidyasagar.ac.in/journal Preparation of ZnS and SnS Nanopowders by Modified SILAR Technique Department of Physics The University of
Journal of Physical Sciences, Vol. 13, 009, 9-34 ISSN: 097-8791 : www.vidyasagar.ac.in/journal Preparation of ZnS and SnS Nanopowders by Modified SILAR Technique Department of Physics The University of
BINARY SYSTEMS. Definition of Composition: Atomic (molar) fraction. Atomic percent. Mass fraction. Mass percent (weight percent)
 BINARY SYSTEMS Definition of Composition: Atomic (molar) fraction Atomic percent Mass fraction Mass percent (weight percent) na =, x i n = A i i i Weight percent mainly in industry! x at % A = x 100 A
BINARY SYSTEMS Definition of Composition: Atomic (molar) fraction Atomic percent Mass fraction Mass percent (weight percent) na =, x i n = A i i i Weight percent mainly in industry! x at % A = x 100 A
This experiment involves the separation and identification of ions using
 Chemistry 112: Reactions Involving Complex Ions Page 27 COMPLEX IONS AND AMPHOTERISM This experiment involves the separation and identification of ions using two important reaction types: (i) the formation
Chemistry 112: Reactions Involving Complex Ions Page 27 COMPLEX IONS AND AMPHOTERISM This experiment involves the separation and identification of ions using two important reaction types: (i) the formation
Name Date Class CHAPTER 1 REVIEW. Answer the following questions in the space provided.
 CHAPTER 1 REVIEW Matter and Change SECTION 1 SHORT ANSWER Answer the following questions in the space provided. 1. a Technological development of a chemical product often (a) lags behind basic research
CHAPTER 1 REVIEW Matter and Change SECTION 1 SHORT ANSWER Answer the following questions in the space provided. 1. a Technological development of a chemical product often (a) lags behind basic research
Chapter Outline. How do atoms arrange themselves to form solids?
 Chapter Outline How do atoms arrange themselves to form solids? Fundamental concepts and language Unit cells Crystal structures Simple cubic Face-centered cubic Body-centered cubic Hexagonal close-packed
Chapter Outline How do atoms arrange themselves to form solids? Fundamental concepts and language Unit cells Crystal structures Simple cubic Face-centered cubic Body-centered cubic Hexagonal close-packed
100% ionic compounds do not exist but predominantly ionic compounds are formed when metals combine with non-metals.
 2.21 Ionic Bonding 100% ionic compounds do not exist but predominantly ionic compounds are formed when metals combine with non-metals. Forming ions Metal atoms lose electrons to form +ve ions. Non-metal
2.21 Ionic Bonding 100% ionic compounds do not exist but predominantly ionic compounds are formed when metals combine with non-metals. Forming ions Metal atoms lose electrons to form +ve ions. Non-metal
Chem 106 Thursday Feb. 3, 2011
 Chem 106 Thursday Feb. 3, 2011 Chapter 13: -The Chemistry of Solids -Phase Diagrams - (no Born-Haber cycle) 2/3/2011 1 Approx surface area (Å 2 ) 253 258 Which C 5 H 12 alkane do you think has the highest
Chem 106 Thursday Feb. 3, 2011 Chapter 13: -The Chemistry of Solids -Phase Diagrams - (no Born-Haber cycle) 2/3/2011 1 Approx surface area (Å 2 ) 253 258 Which C 5 H 12 alkane do you think has the highest
It takes four quantum numbers to describe an electron. Additionally, every electron has a unique set of quantum numbers.
 So, quantum mechanics does not define the path that the electron follows; rather, quantum mechanics works by determining the energy of the electron. Once the energy of an electron is known, the probability
So, quantum mechanics does not define the path that the electron follows; rather, quantum mechanics works by determining the energy of the electron. Once the energy of an electron is known, the probability
Semiconductors, diodes, transistors
 Semiconductors, diodes, transistors (Horst Wahl, QuarkNet presentation, June 2001) Electrical conductivity! Energy bands in solids! Band structure and conductivity Semiconductors! Intrinsic semiconductors!
Semiconductors, diodes, transistors (Horst Wahl, QuarkNet presentation, June 2001) Electrical conductivity! Energy bands in solids! Band structure and conductivity Semiconductors! Intrinsic semiconductors!
POLYMER BASED PHOTOVOLTAICS
 PLYMER BASED PHTVLTAICS Novel concepts, materials and state-of-the-art performances Jan Kroon Semiconducting polymers Nobel Prize Chemistry 2000 (Alan J. Heeger, Alan G. MacDiarmid, Hideki Shirakawa) Conducting
PLYMER BASED PHTVLTAICS Novel concepts, materials and state-of-the-art performances Jan Kroon Semiconducting polymers Nobel Prize Chemistry 2000 (Alan J. Heeger, Alan G. MacDiarmid, Hideki Shirakawa) Conducting
Free Electron Fermi Gas (Kittel Ch. 6)
 Free Electron Fermi Gas (Kittel Ch. 6) Role of Electrons in Solids Electrons are responsible for binding of crystals -- they are the glue that hold the nuclei together Types of binding (see next slide)
Free Electron Fermi Gas (Kittel Ch. 6) Role of Electrons in Solids Electrons are responsible for binding of crystals -- they are the glue that hold the nuclei together Types of binding (see next slide)
KS3 Science: Chemistry Contents
 summary KS3 Science MyWorks Guide Chemistry KS3 Science: Chemistry Mini zes: 40 Super zes: 5 Extension zes: 4 Skills zes: 6 TOTAL 54 What are MyWorks zes? MyWorks zes are short individual learning tasks
summary KS3 Science MyWorks Guide Chemistry KS3 Science: Chemistry Mini zes: 40 Super zes: 5 Extension zes: 4 Skills zes: 6 TOTAL 54 What are MyWorks zes? MyWorks zes are short individual learning tasks
REVIEW QUESTIONS Chapter 8
 Chemistry 101 ANSWER KEY REVIEW QUESTIONS Chapter 8 Use only a periodic table to answer the following questions. 1. Write complete electron configuration for each of the following elements: a) Aluminum
Chemistry 101 ANSWER KEY REVIEW QUESTIONS Chapter 8 Use only a periodic table to answer the following questions. 1. Write complete electron configuration for each of the following elements: a) Aluminum
Perfect Fluids: From Nano to Tera
 Perfect Fluids: From Nano to Tera Thomas Schaefer North Carolina State University 1 2 Perfect Fluids sqgp (T=180 MeV) Neutron Matter (T=1 MeV) Trapped Atoms (T=0.1 nev) 3 Hydrodynamics Long-wavelength,
Perfect Fluids: From Nano to Tera Thomas Schaefer North Carolina State University 1 2 Perfect Fluids sqgp (T=180 MeV) Neutron Matter (T=1 MeV) Trapped Atoms (T=0.1 nev) 3 Hydrodynamics Long-wavelength,
The Current status of Korean silicon photovoltaic industry and market. 2011. 3.17 Sangwook Park LG Electronics Inc.
 The Current status of Korean silicon photovoltaic industry and market 2011. 3.17 Sangwook Park LG Electronics Inc. contents 1.Introduction (World PV Market) 2.Korean PV market 3.Photovoltaics in LG Electronics
The Current status of Korean silicon photovoltaic industry and market 2011. 3.17 Sangwook Park LG Electronics Inc. contents 1.Introduction (World PV Market) 2.Korean PV market 3.Photovoltaics in LG Electronics
Effects of interface morphology and geometry on the thermoelectric properties of artificially structured ZnO-based thin-films
 Effects of interface morphology and geometry on the thermoelectric properties of artificially structured ZnO-based thin-films Dissertation zur Erlangung des Doktorgrades der Naturwissenschaften (Dr. rer.
Effects of interface morphology and geometry on the thermoelectric properties of artificially structured ZnO-based thin-films Dissertation zur Erlangung des Doktorgrades der Naturwissenschaften (Dr. rer.
Chapter Outline Dislocations and Strengthening Mechanisms
 Chapter Outline Dislocations and Strengthening Mechanisms What is happening in material during plastic deformation? Dislocations and Plastic Deformation Motion of dislocations in response to stress Slip
Chapter Outline Dislocations and Strengthening Mechanisms What is happening in material during plastic deformation? Dislocations and Plastic Deformation Motion of dislocations in response to stress Slip
A metal-free polymeric photocatalyst for hydrogen production from water under visible light
 A metal-free polymeric photocatalyst for hydrogen production from water under visible light Xinchen Wang, Kazuhiko Maeda, Arne Thomas, Kazuhiro Takanabe, Gang Xin, Johan M. Carlsson, Kazunari Domen, Markus
A metal-free polymeric photocatalyst for hydrogen production from water under visible light Xinchen Wang, Kazuhiko Maeda, Arne Thomas, Kazuhiro Takanabe, Gang Xin, Johan M. Carlsson, Kazunari Domen, Markus
ORIENTATION CHARACTERISTICS OF THE MICROSTRUCTURE OF MATERIALS
 ORIENTATION CHARACTERISTICS OF THE MICROSTRUCTURE OF MATERIALS K. Sztwiertnia Polish Academy of Sciences, Institute of Metallurgy and Materials Science, 25 Reymonta St., 30-059 Krakow, Poland MMN 2009
ORIENTATION CHARACTERISTICS OF THE MICROSTRUCTURE OF MATERIALS K. Sztwiertnia Polish Academy of Sciences, Institute of Metallurgy and Materials Science, 25 Reymonta St., 30-059 Krakow, Poland MMN 2009
From Nano-Electronics and Photonics to Renewable Energy
 From Nano-Electronics and Photonics to Renewable Energy Tom Smy Department of Electronics, Carleton University Questions are welcome! OUTLINE Introduction: to EE and Engineering Physics Renewable Energy
From Nano-Electronics and Photonics to Renewable Energy Tom Smy Department of Electronics, Carleton University Questions are welcome! OUTLINE Introduction: to EE and Engineering Physics Renewable Energy
FUNDAMENTAL PROPERTIES OF SOLAR CELLS
 FUNDAMENTAL PROPERTIES OF SOLAR CELLS January 31, 2012 The University of Toledo, Department of Physics and Astronomy SSARE, PVIC Principles and Varieties of Solar Energy (PHYS 4400) and Fundamentals of
FUNDAMENTAL PROPERTIES OF SOLAR CELLS January 31, 2012 The University of Toledo, Department of Physics and Astronomy SSARE, PVIC Principles and Varieties of Solar Energy (PHYS 4400) and Fundamentals of
Effect of the oxide film formed on the electrical properties of Cu-Zn alloy electric contact material
 Effect of the oxide film formed on the electrical properties of Cu-Zn alloy electric contact material Hao-Long Chen *, Ke-Cheng Tseng and Yao-Sheng Yang Department of Electronic Engineering, Kao Yuan University,
Effect of the oxide film formed on the electrical properties of Cu-Zn alloy electric contact material Hao-Long Chen *, Ke-Cheng Tseng and Yao-Sheng Yang Department of Electronic Engineering, Kao Yuan University,
Development of certified reference material of thin film for thermal diffusivity
 Development of certified reference material of thin film for thermal diffusivity Takashi Yagi, Thermophysical properties section, NMIJ/AIST Joshua Martin MML, National Institute of Standards and Technology
Development of certified reference material of thin film for thermal diffusivity Takashi Yagi, Thermophysical properties section, NMIJ/AIST Joshua Martin MML, National Institute of Standards and Technology
Phase. Gibbs Phase rule
 Phase diagrams Phase A phase can be defined as a physically distinct and chemically homogeneous portion of a system that has a particular chemical composition and structure. Water in liquid or vapor state
Phase diagrams Phase A phase can be defined as a physically distinct and chemically homogeneous portion of a system that has a particular chemical composition and structure. Water in liquid or vapor state
Lecture 19: Eutectoid Transformation in Steels: a typical case of Cellular
 Lecture 19: Eutectoid Transformation in Steels: a typical case of Cellular Precipitation Today s topics Understanding of Cellular transformation (or precipitation): when applied to phase transformation
Lecture 19: Eutectoid Transformation in Steels: a typical case of Cellular Precipitation Today s topics Understanding of Cellular transformation (or precipitation): when applied to phase transformation
The Lewis structure is a model that gives a description of where the atoms, charges, bonds, and lone pairs of electrons, may be found.
 CEM110 Week 12 Notes (Chemical Bonding) Page 1 of 8 To help understand molecules (or radicals or ions), VSEPR shapes, and properties (such as polarity and bond length), we will draw the Lewis (or electron
CEM110 Week 12 Notes (Chemical Bonding) Page 1 of 8 To help understand molecules (or radicals or ions), VSEPR shapes, and properties (such as polarity and bond length), we will draw the Lewis (or electron
Chapter 5: Diffusion. 5.1 Steady-State Diffusion
 : Diffusion Diffusion: the movement of particles in a solid from an area of high concentration to an area of low concentration, resulting in the uniform distribution of the substance Diffusion is process
: Diffusion Diffusion: the movement of particles in a solid from an area of high concentration to an area of low concentration, resulting in the uniform distribution of the substance Diffusion is process
NORGES TEKNISK- NATURVITENSKAPELIGE UNIVERSITET INSTITUTT FOR FYSIKK. Eksamen i Emne TFY4220 Faste Stoffers Fysikk
 Page of 5 NORGES TEKNISK- NATURVITENSKAPELIGE UNIVERSITET INSTITUTT FOR FYSIKK Fagleg kontakt under eksamen: Institutt for fysikk, Gløshaugen Professor Steinar Raaen, 4896758 Eksamen i Emne TFY40 Faste
Page of 5 NORGES TEKNISK- NATURVITENSKAPELIGE UNIVERSITET INSTITUTT FOR FYSIKK Fagleg kontakt under eksamen: Institutt for fysikk, Gløshaugen Professor Steinar Raaen, 4896758 Eksamen i Emne TFY40 Faste
Anharmonicity and Weak Mode Assignment in La 2 x Sr x CuO 4 with Oxygen Isotopic Substitution
 Vol. 111 (2007) ACTA PHYSICA POLONICA A No. 1 Proceedings of the Symposium K: Complex Oxide Materials for New Technologies of E-MRS Fall Meeting 2006, Warsaw, September 4 8, 2006 Anharmonicity and Weak
Vol. 111 (2007) ACTA PHYSICA POLONICA A No. 1 Proceedings of the Symposium K: Complex Oxide Materials for New Technologies of E-MRS Fall Meeting 2006, Warsaw, September 4 8, 2006 Anharmonicity and Weak
