Sponsored Projects Training: Submitting Contracts & Other Research Agreements to ORA
|
|
|
- Isaac Neal
- 8 years ago
- Views:
Transcription
1 The Johns Hopkins University School of Medicine Office of Research Administration Sponsored Projects Training: Submitting Contracts & Other Research Agreements to ORA March/April 2013
2 We perform our work as a team: SPECIALISTS receive and pre-review contracts, proposals and other research agreements from commercial and federal sponsors. They collect required signatures and prepare the awards for workflow to Sponsored Projects Shared Services. DIRECTORS continue the review, negotiate and ultimately approve the contracts/ agreements. The Directors and Associate Dean for Research execute (sign) the agreements. 2
3 Types of agreements processed by Specialists and Directors at ORA (MRB) Funded Agreements Contracts & Subcontracts Commercial Grants & Subgrants Complex Federal Grants Non-Commercial Clinical Trials Non-profit Sponsored Contracts Non-Monetary Agreements Confidentiality Non- Disclosure Agreements (CDA or NDA) Data Use Agreements Collaborations Memorandum of Understanding (MOU) Equipment/Software Loan Agreement 3
4 1. Specialist receives COEUS PD in ORA mailbox, per Assigned Department 2. Specialist reviews COEUS PD Summary for accuracy and completion; performs initial review (pre-review) for the Required 6-Pack info 3. Specialist creates a MyRAP record; continues pre-review Approves the COEUS PD, except for grants 7. Specialist is identified as new owner; circulates agreement for required signatures 6. Director conducts review, evaluates content, negotiates the agreement, transfers ownership of folder 5. Ownership of Folder and MyRAP record transfer to a Director 4. Specialist starts a Folder : Agreement PD Summary IRB/IACUC 8. Specialist uploads fully executed agreement to MyRAP and processes the records for scanning to Oculus 9. Specialist transfers MyRAP ownership to ORA Operations Analyst, who completes the record when the documents have been scanned to Oculus 10. ORA Operations Analyst workflows funded agreements to SPSS via Oculus 4
5 Who do I contact in ORA? The Department Assignments for the Miller Research Building (MRB) Sponsored Projects Specialists are posted on the ORA website: However, one Specialist may be assigned to a particular Sponsor because of the volume of agreements, funded by that Sponsor, all across the School of Medicine. The MRB ORA Specialists are: Jenifer DeLeaver Mary Deloatch Celeste Hartman Angela Mellerson 5
6 How do we determine priorities? Hard sponsored deadlines in COEUS are noted and confirmed. If there is no hard deadline, please leave the field BLANK on the PD Grants and Contracts w/ hard deadlines are processed first; then CDA s and Data Use Agreements Urgent proposals, as noted on the Supplemental Information for Commercial Agreements form All others are processed in the order received 6
7 What information is necessary? 6-Pack For an initial or original agreement: Supplemental Information form Editable version of the Agreement (preferably in Word) Budget, when applicable Statement of work IRB/IACUC approval COEUS PD, with all documents uploaded For amendments: Supplemental Information form Editable version of the Amendment Budget Statement of work (but only if it has changed) Updated IRB/IACUC approval A COEUS PD incl Master Proposal #, with all documents uploaded 7
8 Who obtains the information? Departmental contacts are responsible for obtaining the required information for all contracts, agreements, and complex grants. ORA can help with the process by clarifying what is necessary, but we cannot get these materials for you. For commercial sponsors, there is a required Supplemental Information for Commercial Agreements form which must be completed and uploaded in order for your proposal to be reviewed. Please note that although information about the proposal was submitted with the application/coeus PD, the department must obtain all follow-up information requested by ORA staff to assure that the proposal continues to move through the process as quickly as possible. While most information is electronic, if you are delivering any related paper (hard copy) materials to ORA, please make sure that you indicate PD # on the envelope or folder. 8
9 Preparing Your 6-Pack for Commercial Agreements For an initial or original agreement: Supplemental Information for Commercial Agreements form website: Editable version of the Agreement (preferably in Word) Budget, when applicable Statement of work IRB/IACUC approval COEUS PD, with all documents uploaded For amendments: Supplemental Information for Commercial Agreements form Editable version of the Amendment Budget Statement of work (but only if it has changed) Updated IRB/IACUC approval A COEUS PD incl Master Proposal #, with all documents uploaded The most important areas to complete on the Supplemental Information form are isolated and displayed on the next 3 slides. Remember to upload the completed form to your COEUS PD. 9
10 Supplemental Information for Commercial Agreements PI: SPONSOR: PROJECT TITLE: I. Type of Study: (CHECK ONLY ONE) Clinical Research Defined as: All commercial clinical research that involves patients or PHI, or clinical testing or procedures, or drug/device diagnostic testing in humans or any planning/lab/clinical service in support of such clinical research. Please send directly to Clinical Research Contracting, 1629 Thames Street, Suite 200, Baltimore, MD or Other Research (anything other than clinical research as defined above) Includes (Check ALL that apply) Materials provided by sponsor or other (with a commitment to conduct specific research) Funding by sponsor or other Equipment provided by sponsor or otherother collaborations Other (e.g., services, lab analysis, etc.)please specify 10
11 Supplemental Information for Commercial Agreements II. Documents (REQUIRED) for Review of Agreements A. Contract documents (items a-c) a. Copy of proposed contract paper and/or electronic (preferred) b. Statement of work (protocol abstract, description) c. Other relevant documents (e.g. invention disclosure, investigator agreement, etc) B. Sponsor contact information for contract negotiation a copy of correspondence or is acceptable. Name Fax Phone Address 11
12 Supplemental Information for Commercial Agreements VI. Priority (select A or B below) A. Urgent -- The following are eligible for urgent review: compassionate use, limited site selection, capped accrual, or other emergent need. To be eligible for urgent review, an explanation of the reason for urgent review MUST be provided below. Without an explanation the agreement will be treated as Routine. (EXPLAIN): Note: Protocol must be submitted to the IRB before or simultaneously with ORA receipt of contract to be considered Priority A and eligible for urgent review. Please provide a copy of the submission (minimally the first page of the form). (ORA initial review within five business days) B. Routine -- All other corporate agreements, investigator initiated clinical trials, basic research agreements. (ORA initial review within ten business days) 12 C. Master or Model Agreements - - Studies that are being done under a Master Agreement, Model Agreement, or where the company has used a JHU template.
13 Information necessary to fill out the COEUS PD Summary Principal Investigator and Co-Investigators Responsible Cost Center number Sponsor Type of Proposal Sponsor grant/contract number, if known Master IPN, if not a new proposal Animal and/or human subjects protocol approvals, if applicable Period of performance dates Amount of the award request, if applicable 13
14 Budgets In all budgets, consider whether the costs are allowed by JHU and the Sponsor. Specifically, costs must be reasonable/necessary, allocable, consistently applied, and in conformance with JHU policy and the terms of the award. Consider whether the costs are allowed under the terms of the RFA or RFP Determine the appropriate Facilities and Administration (F&A) rates, also known as IDC rates. Refer to the following website for current rates: 14
15 Statement of Work Also known as a work scope, the SOW describes what the JHU PI or staff will do. The SOW is not always a summary of the whole proposal. Based on the statement of work, you and the ORA reviewers should be able to determine whether the budget is appropriate. 15
16 IRB and IACUC approvals Please be aware that ORA can review a proposal without current IRB or IACUC approval, but we must have the approval in order to fully execute the contract for account set-up. 16
17 Budget and Payment Information Intellectual Property Technical and Financial Reporting Subject Protection Indemnification What s in a contract? Contact Information Confidentiality Period of Performance Termination Statement of Work 17
18 Things to be aware of 18 If the proposal has a direct commercial sponsor or 3 rd party commercial sponsor AND is clinical research, then it will be processed at Fells Point. If the project has a commercial sponsor BUT the funding is from a federal sponsor, then it will be processed at the MRB. (e.g. SBIR) The PI and each Co-Investigator on the project needs to be listed as a team member on the IRB and/or IACUC approved protocols. Please be very careful when using copy paste features from a prior submission to complete subsequent documents. It is easy to submit documents with outdated information if this is done too quickly and not carefully proof-read. Please read your documents and update ALL information, as needed. The PI must be registered in MyRAP. Go to this link to login and register your MyRAP account:
19 Why is it taking so long?? Can t I just get my SAP update now?? Complicating factors that can delay a fully executed agreement: Negotiation issues w/ contract terms (e.g. Intellectual property) PI has not obtained IRB or IACUC approval Delays in Resubmission of corrected COEUS PD Communication gaps: Please cc Specialist on any follow-up information being provided to the Director Negotiator requires more information from Sponsor or PI Incorrect contact information for the Sponsor Late or no response when Partial Ex is sent to PI or Sponsor for signatures 19
20 So now we have a fully executed agreement, but what s next?? Preparing the File for SPSS Workflow What Specialists do to get you closer to an SAP Update: Purge: Specialists shred paperwork no longer needed to document an official agreement Upload Document to ORA Shared Drive & MyRAP: A complete record of the fully executed agreement and supporting documents is stored on ORA s server Fully executed agreement is uploaded to MyRAP Transfer File to ORA Operations Analyst for Final Review and Scanning: To Oculus To SPSS (via Oculus) for funded agreements 20
21 Please do NOT: - Submit a proposal directly to a Sponsor WITHOUT routing it thru ORA - Provide incomplete protocol approvals - Begin your project before both the ORA and Sponsor have executed the agreement. Only a fully executed agreement is recognized as authorization to begin the work. 21
22 What Departments can do to help us: Provide requested information in a timely manner Send complete documents Ensure consistency in documentation and accuracy when referencing COEUS PD, MyRAP, PI s, Project titles, etc. in communications Be kind and patient 22
23 Contact the Specialists If you have questions about what you need to submit, please call or us. Jenifer DeLeaver Mary Deloatch Celeste Hartman Angela Mellerson
24 Other team members Dezire Chaney, Sponsored Projects Asst Rhanota Edwards, Operations Analyst Rosalind Palmer, Sponsored Agreements Mgr Office of Research Administration Suite 117, Miller Research Bldg 733 N. Broadway Baltimore, MD
Office of Research Administration: Clinical Research Contracting
 Office of Research Administration: Clinical Research Contracting Presented to: Research Administration Professionals Group Presented by: Patricia Travis and Mont Brownlee October 22, 2013 1 Training Agenda
Office of Research Administration: Clinical Research Contracting Presented to: Research Administration Professionals Group Presented by: Patricia Travis and Mont Brownlee October 22, 2013 1 Training Agenda
Research Administration Retreat Research Administration 101. Tracy Wayman twayman@jhu.edu
 Research Administration Retreat Research Administration 101 Tracy Wayman twayman@jhu.edu Sponsored Projects Shared Services (SPSS) Roles SPSS is responsible for setting up each grant s master data elements
Research Administration Retreat Research Administration 101 Tracy Wayman twayman@jhu.edu Sponsored Projects Shared Services (SPSS) Roles SPSS is responsible for setting up each grant s master data elements
SCHOOL OF MEDICINE OFFICE OF RESEARCH ADMINISTRATION
 SCHOOL OF MEDICINE OFFICE OF RESEARCH ADMINISTRATION Step By Step Guide for: Coeus Proposal Development (Coeus PD) (in place of eis) for Paper (Hard) Copy and Electronic (non Grants.gov) submission Effective
SCHOOL OF MEDICINE OFFICE OF RESEARCH ADMINISTRATION Step By Step Guide for: Coeus Proposal Development (Coeus PD) (in place of eis) for Paper (Hard) Copy and Electronic (non Grants.gov) submission Effective
Johns Hopkins University School of Medicine Office of Research Administration
 Checklist for T Applications (Institutional Training Grants) Please read RFA and Individual NIH Institute Guidelines This document is designed only to serve as a project management tool. It does NOT replace
Checklist for T Applications (Institutional Training Grants) Please read RFA and Individual NIH Institute Guidelines This document is designed only to serve as a project management tool. It does NOT replace
Proposal Development Guide
 Proposal Development Guide Contents The Proposal Tab... 3 Document Overview Panel... 3 Required Fields for Saving Document Panel... 3 Institutional Fields (Conditionally Required)... 9 Sponsor & Program
Proposal Development Guide Contents The Proposal Tab... 3 Document Overview Panel... 3 Required Fields for Saving Document Panel... 3 Institutional Fields (Conditionally Required)... 9 Sponsor & Program
KRIEGER SCHOOL OF ARTS & SCIENCES GRANT PROPOSAL GUIDE
 KRIEGER SCHOOL OF ARTS & SCIENCES GRANT PROPOSAL GUIDE Presented by the Grant Proposal Guide Workgroup Fall, 2011 GRANT PROPOSAL GUIDE TABLE OF CONTENTS INTRODUCTION... 4 NEW TO DOING PROPOSALS... 5 NEW
KRIEGER SCHOOL OF ARTS & SCIENCES GRANT PROPOSAL GUIDE Presented by the Grant Proposal Guide Workgroup Fall, 2011 GRANT PROPOSAL GUIDE TABLE OF CONTENTS INTRODUCTION... 4 NEW TO DOING PROPOSALS... 5 NEW
ROLES, RESPONSIBILITIES AND DELEGATION OF DUTIES IN CLINICAL TRIALS OF MEDICINAL PRODUCTS
 ROLES, RESPONSIBILITIES AND DELEGATION OF DUTIES IN CLINICAL TRIALS OF MEDICINAL PRODUCTS STANDARD OPERATING PROCEDURE NO SOP 09 DATE RATIFIED 4/7/13 NEXT REVIEW DATE 4/7/14 POLICY STATEMENT/KEY OBJECTIVES:
ROLES, RESPONSIBILITIES AND DELEGATION OF DUTIES IN CLINICAL TRIALS OF MEDICINAL PRODUCTS STANDARD OPERATING PROCEDURE NO SOP 09 DATE RATIFIED 4/7/13 NEXT REVIEW DATE 4/7/14 POLICY STATEMENT/KEY OBJECTIVES:
Procedures and Guidelines for External Grants and Sponsored Programs
 Definitions: Procedures and Guidelines for External Grants and Sponsored Programs 1. Grant and Contract Financial Administration Office (Grants Office): The office within the Accounting Operations Area
Definitions: Procedures and Guidelines for External Grants and Sponsored Programs 1. Grant and Contract Financial Administration Office (Grants Office): The office within the Accounting Operations Area
Howard University. version 1 created: 11/2014. R e s e a r c h A d m i n i s t r a t i v e S e r v i c e s
 Howard University version 1 created: 11/2014 R e s e a r c h A d m i n i s t r a t i v e S e r v i c e s CONTENTS DocuSign Overview... 2 Account Setup... 2 Getting Started... 2 The Submission Routing Structure...
Howard University version 1 created: 11/2014 R e s e a r c h A d m i n i s t r a t i v e S e r v i c e s CONTENTS DocuSign Overview... 2 Account Setup... 2 Getting Started... 2 The Submission Routing Structure...
Crystal Miller, MPA Assistant Director Sponsored Programs Administration. March 26 & 27, 2009 April 3, 2009
 Crystal Miller, MPA Assistant Director Sponsored Programs Administration March 26 & 27, 2009 April 3, 2009 NIH Funding Current Announcements Timelines and Process Challenge Grants Other NIH Stimulus Opportunities
Crystal Miller, MPA Assistant Director Sponsored Programs Administration March 26 & 27, 2009 April 3, 2009 NIH Funding Current Announcements Timelines and Process Challenge Grants Other NIH Stimulus Opportunities
Division of Nutritional Sciences Grant Proposal Preparation and Submission Guidelines
 Division of Nutritional Sciences Grant Proposal Preparation and Submission Guidelines A. DNS Grant and Contract Administrator and DNS Office of Administrative Support (OAS) and what they will or can do
Division of Nutritional Sciences Grant Proposal Preparation and Submission Guidelines A. DNS Grant and Contract Administrator and DNS Office of Administrative Support (OAS) and what they will or can do
Proposal and Award Management Information System. Users Guide
 Proposal and Award Management Information System Users Guide PAMIS, version 1 April 2004 Table of Contents INTRODUCTION......1 PAMIS NOTIFICATION... 2 ACCESS TO AWARD INFORMATION......2 DEPARTMENT ROLES
Proposal and Award Management Information System Users Guide PAMIS, version 1 April 2004 Table of Contents INTRODUCTION......1 PAMIS NOTIFICATION... 2 ACCESS TO AWARD INFORMATION......2 DEPARTMENT ROLES
GRANT MANAGER S HANDBOOK
 GRANT MANAGER S HANDBOOK Office of Research & Sponsored Programs Foust 251 T: (989) 774-ORSP F: (989) 774-3439 E: orsp@cmich.edu The Purpose of the Grant Manager s Handbook is to provide answers to commonly
GRANT MANAGER S HANDBOOK Office of Research & Sponsored Programs Foust 251 T: (989) 774-ORSP F: (989) 774-3439 E: orsp@cmich.edu The Purpose of the Grant Manager s Handbook is to provide answers to commonly
Getting Started ONLINE APPLICATION. Access the online certification application system
 Online Application This instruction guide is for currently certified firms seeking renewal and firms applying for the first time. The information presented is drawn from example scenarios and may not exactly
Online Application This instruction guide is for currently certified firms seeking renewal and firms applying for the first time. The information presented is drawn from example scenarios and may not exactly
Contract Compliance and the Federal Acquisition Regulation (FAR)
 Contract Compliance and the Federal Acquisition Regulation (FAR) ORA CERTIFICATE PROGRAM (MODULE 11) 27 MAY 2015 Learning Objectives Participants will learn about the history of the Federal Acquisition
Contract Compliance and the Federal Acquisition Regulation (FAR) ORA CERTIFICATE PROGRAM (MODULE 11) 27 MAY 2015 Learning Objectives Participants will learn about the history of the Federal Acquisition
Electronic Signature and Approval Process
 Howard University Electronic Signature and Approval Process Submitting a Proposal Package Electronically version 3 created: 05/2013 R e s e a r c h A d m i n i s t r a t i v e S e r v i c e s CONTENTS
Howard University Electronic Signature and Approval Process Submitting a Proposal Package Electronically version 3 created: 05/2013 R e s e a r c h A d m i n i s t r a t i v e S e r v i c e s CONTENTS
POLICY ON SUPPLEMENTAL PAY FROM SPONSORED PROJECTS FACULTY
 POLICY ON SUPPLEMENTAL PAY FROM SPONSORED PROJECTS FACULTY Grant and contract funded work is considered to be a part of routine faculty assignments at Bowling Green State University, and in most circumstances
POLICY ON SUPPLEMENTAL PAY FROM SPONSORED PROJECTS FACULTY Grant and contract funded work is considered to be a part of routine faculty assignments at Bowling Green State University, and in most circumstances
Sub-award Risk Management Operational and Audit Perspectives. 2013 Georgia Conference for College and University Auditors 20 May, 2013
 Sub-award Risk Management Operational and Audit Perspectives 2013 Georgia Conference for College and University Auditors 20 May, 2013 Presentation Goals Look at familiar grants management components with
Sub-award Risk Management Operational and Audit Perspectives 2013 Georgia Conference for College and University Auditors 20 May, 2013 Presentation Goals Look at familiar grants management components with
Kuali Coeus and InfoEd Reporting Rev. 0 RFP #32772360. Scope of Work
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 Scope of Work I. Scope of Solicitation II. Instructions to Offerors III. Scope of Work / Specifications
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 Scope of Work I. Scope of Solicitation II. Instructions to Offerors III. Scope of Work / Specifications
Research Administration Training Program for Existing Staff Johns Hopkins University
 Research Administration Training Program for Existing Staff Johns Hopkins University 2014 Johns Hopkins University Page 1 Research Administration Training Program for Existing JHU Staff Introduction: Several
Research Administration Training Program for Existing Staff Johns Hopkins University 2014 Johns Hopkins University Page 1 Research Administration Training Program for Existing JHU Staff Introduction: Several
HACKENSACK UNIVERSITY MEDICAL CENTER Research Department Policies and Procedures Manual
 Policy Name: Research Grant Submissions Process HACKENSACK UNIVERSITY MEDICAL CENTER Research Department Policies and Procedures Manual Policy #: SPO_003 Effective Date: 01/01/2009 Page 1 of 9 GENERAL
Policy Name: Research Grant Submissions Process HACKENSACK UNIVERSITY MEDICAL CENTER Research Department Policies and Procedures Manual Policy #: SPO_003 Effective Date: 01/01/2009 Page 1 of 9 GENERAL
Research Administrators Meeting Mollie Sykes Associate Dean, Research Administration Office of Research Administration http://research.som.duke.
 Research Administrators Meeting Mollie Sykes Associate Dean, Research Administration Office of Research Administration http://research.som.duke.edu Next Meeting: December 9, 2009 Agenda MyResearch Todd
Research Administrators Meeting Mollie Sykes Associate Dean, Research Administration Office of Research Administration http://research.som.duke.edu Next Meeting: December 9, 2009 Agenda MyResearch Todd
Consolidated I&T Data Centre - P3 Canada Fund
 STAFF REPORT ACTION REQUIRED Consolidated I&T Data Centre - P3 Canada Fund Date: January 16, 2012 To: From: Wards: Reference Number: Government Management Committee Deputy City Manager & Chief Financial
STAFF REPORT ACTION REQUIRED Consolidated I&T Data Centre - P3 Canada Fund Date: January 16, 2012 To: From: Wards: Reference Number: Government Management Committee Deputy City Manager & Chief Financial
Comprehensive Study Documents List (Biomedical Studies)
 Comprehensive Study Documents List (Biomedical Studies) Investigators conducting human subjects research must maintain study documents in adherence to federal and state regulations, USC policies, and good
Comprehensive Study Documents List (Biomedical Studies) Investigators conducting human subjects research must maintain study documents in adherence to federal and state regulations, USC policies, and good
Draft Service Level Agreement between ECDC and Contractor
 Draft Service Level Agreement between ECDC and Contractor The Contractor provides the consultancy services to ECDC in accordance with specifications that form part of Specific Contracts implementing framework
Draft Service Level Agreement between ECDC and Contractor The Contractor provides the consultancy services to ECDC in accordance with specifications that form part of Specific Contracts implementing framework
UT Health Science Center at San Antonio Research/Grant Records
 UT Health Science Center at San Antonio Research/Grant Records Agency 1.1 Administrative Records - General 400 DE Experiments and Tests. AV O False SEE section on Medical/Dental - Patient Records, record
UT Health Science Center at San Antonio Research/Grant Records Agency 1.1 Administrative Records - General 400 DE Experiments and Tests. AV O False SEE section on Medical/Dental - Patient Records, record
esnap Progress Reports: Submission via the era Commons UC Irvine Office of Research Sponsored Projects Administration
 esnap Progress Reports: Submission via the era Commons UC Irvine Office of Research Sponsored Projects Administration Overview esnap: Electronic Streamlined Non-competing Award Process Progress Reports
esnap Progress Reports: Submission via the era Commons UC Irvine Office of Research Sponsored Projects Administration Overview esnap: Electronic Streamlined Non-competing Award Process Progress Reports
Complex Proposal Development Best Practices
 Introduction Complex Proposal Development Best Practices As grant application requirements become increasingly complex, and teams pursuing funding become increasingly diverse, the need for thoroughly planning
Introduction Complex Proposal Development Best Practices As grant application requirements become increasingly complex, and teams pursuing funding become increasingly diverse, the need for thoroughly planning
AURA Meeting 05/28/2014
 AURA Meeting 05/28/2014 Welcome Karen Niemeier Contracts Director, OSP Website Spotlight Cherrelle Duncan, MPSA Assigning a Delegate Sometimes, faculty members are unavailable. Delegates can be assigned
AURA Meeting 05/28/2014 Welcome Karen Niemeier Contracts Director, OSP Website Spotlight Cherrelle Duncan, MPSA Assigning a Delegate Sometimes, faculty members are unavailable. Delegates can be assigned
Determining who the investigators are and who the principal investigator is:
 Responsibilities of the IRBs of the Health System and University for Research Conducted by Investigators from a Combination of the Following Institutions: the Health System, University, and Medical School
Responsibilities of the IRBs of the Health System and University for Research Conducted by Investigators from a Combination of the Following Institutions: the Health System, University, and Medical School
Moving to the Top: Establishing Your Research Program at BWH MODULE 5: Money: How to Spend it January 28, 2010
 Moving to the Top: Establishing Your Research Program at BWH MODULE 5: Money: How to Spend it January 28, 2010 Recommended Reading: Chapter 4, Academic Scientists at Work TOPICS: 1) Get to know your research
Moving to the Top: Establishing Your Research Program at BWH MODULE 5: Money: How to Spend it January 28, 2010 Recommended Reading: Chapter 4, Academic Scientists at Work TOPICS: 1) Get to know your research
The University of Texas at Tyler Office of Sponsored Research (OSR) Time and Effort Reporting Policy
 The University of Texas at Tyler Office of Sponsored Research (OSR) Time and Effort Reporting Policy Applicability This Time and Effort Reporting Policy applies to all individuals receiving funding in
The University of Texas at Tyler Office of Sponsored Research (OSR) Time and Effort Reporting Policy Applicability This Time and Effort Reporting Policy applies to all individuals receiving funding in
IRB Policy for Security and Integrity of Human Research Data
 IRB Policy for Security and Integrity of Human Research Data Kathleen Hay Human Subjects Protection Office Terri Shkuda Research Informatics & Computing, Information Technology Overview of Presentation
IRB Policy for Security and Integrity of Human Research Data Kathleen Hay Human Subjects Protection Office Terri Shkuda Research Informatics & Computing, Information Technology Overview of Presentation
Advisory User Group Meeting. October 18, 2011
 Advisory User Group Meeting October 18, 2011 1 Agenda 11:30 Welcome Christine Lavoie 11:35 Session Objectives Christine Lavoie 11:40 USC s Research Administration System Christine Lavoie 11:50 Project
Advisory User Group Meeting October 18, 2011 1 Agenda 11:30 Welcome Christine Lavoie 11:35 Session Objectives Christine Lavoie 11:40 USC s Research Administration System Christine Lavoie 11:50 Project
Please submit your signed application by the deadline date to: Faculty Research and Sponsored Programs Box 495006 CAMPUS MAIL
 Save Form Print Form Berry College 2014-2015 Faculy Development Grant Application To: Through: Through: Kathy Brittain Richardson, Provost Chair, Department of Dean, School of Applicant Name: Project Title:
Save Form Print Form Berry College 2014-2015 Faculy Development Grant Application To: Through: Through: Kathy Brittain Richardson, Provost Chair, Department of Dean, School of Applicant Name: Project Title:
The George Washington University Hospital
 The George Washington University Hospital Information Access Management to support clinical Research Protocol Specification Effective January 1, 2012 1 Introduction The George Washington University Hospital
The George Washington University Hospital Information Access Management to support clinical Research Protocol Specification Effective January 1, 2012 1 Introduction The George Washington University Hospital
NATIONAL SCIENCE FOUNDATION 4201 Wilson Boulevard ARLINGTON, VIRGINIA 22230
 NATIONAL SCIENCE FOUNDATION 4201 Wilson Boulevard ARLINGTON, VIRGINIA 22230 OFFICE OF INSPECTOR GENERAL Date: January 31, 2012 To: Mary F. Santonastasso, Director Division of Institution and Award Support
NATIONAL SCIENCE FOUNDATION 4201 Wilson Boulevard ARLINGTON, VIRGINIA 22230 OFFICE OF INSPECTOR GENERAL Date: January 31, 2012 To: Mary F. Santonastasso, Director Division of Institution and Award Support
WELCOME. to the Thesis and Dissertation Formatting Tutorial
 WELCOME to the Thesis and Dissertation Formatting Tutorial This tutorial is also available on the Graduate School s website at: http://grad.msu.edu/etd/ along with the formatting guide, sample pages, and
WELCOME to the Thesis and Dissertation Formatting Tutorial This tutorial is also available on the Graduate School s website at: http://grad.msu.edu/etd/ along with the formatting guide, sample pages, and
CONSIDERATION OF SUGGESTIONS
 CONSIDERATION OF SUGGESTIONS INTRODUCTION Every year we receive many letters proposing ideas for the Company s use. We appreciate this interest in BD and want to thank each person who has taken the time
CONSIDERATION OF SUGGESTIONS INTRODUCTION Every year we receive many letters proposing ideas for the Company s use. We appreciate this interest in BD and want to thank each person who has taken the time
Records Management and Retention for Sponsored Programs
 Records Management and Retention for Sponsored Programs Camilla Curnow, Outreach and Compliance Manager Angela Sherman, Research Administration Manager, School of Medicine University of Virginia Office
Records Management and Retention for Sponsored Programs Camilla Curnow, Outreach and Compliance Manager Angela Sherman, Research Administration Manager, School of Medicine University of Virginia Office
AURA Clinical Trial FP Guidelines
 AURA Clinical Trial FP Guidelines Table of Contents Overview 2 What is a Clinical Trial? Guidelines Acronym/ Abbreviations/ Key Codes FP Workflow.. Step 1: PC-O create CT.. 3 Step 2: OCR/ URA-M advance
AURA Clinical Trial FP Guidelines Table of Contents Overview 2 What is a Clinical Trial? Guidelines Acronym/ Abbreviations/ Key Codes FP Workflow.. Step 1: PC-O create CT.. 3 Step 2: OCR/ URA-M advance
How To Manage Money At The Purdue Plant
 Account Management Guidelines for SPS Funds (4XXXXXXX) Summary In order to achieve maximum utilization of Purdue resources, the Account Management Guidelines are used as a working document to provide more
Account Management Guidelines for SPS Funds (4XXXXXXX) Summary In order to achieve maximum utilization of Purdue resources, the Account Management Guidelines are used as a working document to provide more
HOUSE BILL 1311 A BILL ENTITLED. 2 Cigarette Restitution Fund - Statewide Academic Health Center Grants - 3 Clarifications
 HOUSE BILL 1311 Unofficial Copy J1 2004 Regular Session 4lr2556 By: Delegate Rosenberg Introduced and read first time: February 13, 2004 Assigned to: Health and Government Operations and Appropriations
HOUSE BILL 1311 Unofficial Copy J1 2004 Regular Session 4lr2556 By: Delegate Rosenberg Introduced and read first time: February 13, 2004 Assigned to: Health and Government Operations and Appropriations
Electronic Signature and Routing Process
 Howard University Electronic Signature and Routing Process Submitting a Proposal Package Electronically 2012 R e s e a r c h A d m i n i s t r a t i v e S e r v i c e s CONTENTS DocuSign Overview... 2
Howard University Electronic Signature and Routing Process Submitting a Proposal Package Electronically 2012 R e s e a r c h A d m i n i s t r a t i v e S e r v i c e s CONTENTS DocuSign Overview... 2
Office of Research Administration Clinical Research Support Services
 Office of Research Administration Clinical Research Support Services 1629 S. Thames Street, Suite 200 Baltimore, Maryland 21213 410-502-6001/office 410-502-6004/fax CRSS@jhmi.edu 1 Office Organization
Office of Research Administration Clinical Research Support Services 1629 S. Thames Street, Suite 200 Baltimore, Maryland 21213 410-502-6001/office 410-502-6004/fax CRSS@jhmi.edu 1 Office Organization
Small Business Electronic Applications: Annotated SF424 (R&R) Form Set
 Small Business Electronic Applications: Annotated SF424 (R&R) Form Set FORMS Included in SBIR and STTR applications: Federal-wide Forms SF424 (R&R) Cover Component [Page 2] Project/Performance Site Location(s)
Small Business Electronic Applications: Annotated SF424 (R&R) Form Set FORMS Included in SBIR and STTR applications: Federal-wide Forms SF424 (R&R) Cover Component [Page 2] Project/Performance Site Location(s)
Commons Email Messages
 Commons Email Messages 1 of 18 Table of Contents 1. NIH era Commons Email Specification... 4 1.1 From Clause... 4 1.2 Standard Subject... 4 1.3 Standard Footer... 4 1.4 Detailed email notification events
Commons Email Messages 1 of 18 Table of Contents 1. NIH era Commons Email Specification... 4 1.1 From Clause... 4 1.2 Standard Subject... 4 1.3 Standard Footer... 4 1.4 Detailed email notification events
Parent and Community- Funded Contributions for Programs at School Sites
 Parent and Community- Funded Contributions for Programs at School Sites A guide to: ü How School Trust Accounts Work ü Paying Consultants or SFUSD Employees ü Contracts, MOUs and Facilities Use Permits
Parent and Community- Funded Contributions for Programs at School Sites A guide to: ü How School Trust Accounts Work ü Paying Consultants or SFUSD Employees ü Contracts, MOUs and Facilities Use Permits
Christopher Kratochvil, MD, Assistant Vice Chancellor for Clinical Research Deborah Vetter, MS, Director, Sponsored Programs Administration
 Christopher Kratochvil, MD, Assistant Vice Chancellor for Clinical Research Deborah Vetter, MS, Director, Sponsored Programs Administration Clinical Research Center Learn-At-Lunch Series October 13, 2011
Christopher Kratochvil, MD, Assistant Vice Chancellor for Clinical Research Deborah Vetter, MS, Director, Sponsored Programs Administration Clinical Research Center Learn-At-Lunch Series October 13, 2011
Creating Proposals and Managing Grants
 Creating Proposals and Managing Grants Page 1 of 41 Table of Contents Lesson 1 Grants Overview... 1 Grants Overview... 3 Pre-Award... 3 Post-Award... 4 Proposal Creation... 4 Proposal Projects... 4 Lesson
Creating Proposals and Managing Grants Page 1 of 41 Table of Contents Lesson 1 Grants Overview... 1 Grants Overview... 3 Pre-Award... 3 Post-Award... 4 Proposal Creation... 4 Proposal Projects... 4 Lesson
Children s Research Management System (CRMS) Version 3.0. Children s Hospital Colorado Research Institute Training Guide April 2015
 Children s Research Management System (CRMS) Version 3.0 Children s Hospital Colorado Research Institute Training Guide April 2015 Table of Contents Operational Needs Assessment (ONA) 3 Visit Schedules
Children s Research Management System (CRMS) Version 3.0 Children s Hospital Colorado Research Institute Training Guide April 2015 Table of Contents Operational Needs Assessment (ONA) 3 Visit Schedules
Fundamentals of Clinical Trial Financial Management
 Fundamentals of Clinical Trial Financial Management Lindsey Spangler Michelle Smith COURSE OBJECTIVES Understand the life cycle of a clinical study from a financial perspective Understand the purpose of
Fundamentals of Clinical Trial Financial Management Lindsey Spangler Michelle Smith COURSE OBJECTIVES Understand the life cycle of a clinical study from a financial perspective Understand the purpose of
Session 1 Introduction to Research Administration
 Session 1 Introduction to Research Administration Thursday, November 19, 2015 1 Becky Bound, Office of the Vice Chancellor for Research and Graduate Education Dawn Herrick, College of Engineering Tammy
Session 1 Introduction to Research Administration Thursday, November 19, 2015 1 Becky Bound, Office of the Vice Chancellor for Research and Graduate Education Dawn Herrick, College of Engineering Tammy
CONTROLLED DOCUMENT. Uncontrolled Copy. RDS014 Research Related Archiving. University Hospitals Birmingham NHS Foundation Trust
 University Hospitals Birmingham NHS Foundation Trust CONTROLLED DOCUMENT RDS014 Research Related Archiving CATEGORY: CLASSIFICATION: PURPOSE Controlled Document Number: Version Number: 1 Controlled Document
University Hospitals Birmingham NHS Foundation Trust CONTROLLED DOCUMENT RDS014 Research Related Archiving CATEGORY: CLASSIFICATION: PURPOSE Controlled Document Number: Version Number: 1 Controlled Document
Clinical Trials Reporting Program (CTRP) and Clinicaltrials.gov (CT.gov) Helpful tips and guidance
 Clinical Trials Reporting Program (CTRP) and Clinicaltrials.gov (CT.gov) Helpful tips and guidance Objectives NCI s Clinical Trials Reporting Program (CTRP) Overview CTRP What is it? CTRP is an NCI mandated
Clinical Trials Reporting Program (CTRP) and Clinicaltrials.gov (CT.gov) Helpful tips and guidance Objectives NCI s Clinical Trials Reporting Program (CTRP) Overview CTRP What is it? CTRP is an NCI mandated
Lymphoma Clinical Research Mentoring Program
 Lymphoma Clinical Research Mentoring Program A Training Program for Hematology/Oncology Fellows and Junior Faculty with a focus on Clinical Research in Lymphoma KEY DATES 2016-2018 Program Overview, Guidelines
Lymphoma Clinical Research Mentoring Program A Training Program for Hematology/Oncology Fellows and Junior Faculty with a focus on Clinical Research in Lymphoma KEY DATES 2016-2018 Program Overview, Guidelines
Creating and Routing a New Clinical Trial Project
 Creating and Routing a New Clinical Trial Project To create a new clinical trial project in InfoEd, click My Proposals in the left sidebar and click. Step 0: Confirm the Intended Principal Investigator
Creating and Routing a New Clinical Trial Project To create a new clinical trial project in InfoEd, click My Proposals in the left sidebar and click. Step 0: Confirm the Intended Principal Investigator
PROFESSIONAL SERVICES AGREEMENT
 PROFESSIONAL SERVICES AGREEMENT This Agreement is made and entered into by and between EMMA PENDLETON BRADLEY HOSPITAL, a not-for-profit corporation, organized and existing under the laws of the State
PROFESSIONAL SERVICES AGREEMENT This Agreement is made and entered into by and between EMMA PENDLETON BRADLEY HOSPITAL, a not-for-profit corporation, organized and existing under the laws of the State
Clinical Trial Office Investigator Handbook. Table of Contents
 Clinical Trial Office Investigator Handbook Table of Contents Section 1: Clinical Trial Office Mission Section 2: List of Services Provided Section 3: Contact Information for the CTO Section 4: Flow chart
Clinical Trial Office Investigator Handbook Table of Contents Section 1: Clinical Trial Office Mission Section 2: List of Services Provided Section 3: Contact Information for the CTO Section 4: Flow chart
APPLICATION FOR SABBATICAL LEAVE UNIVERSITY OF ILLINOIS. UIN: RANK: Date:
 UIN: RANK: Date: DEPT: SCHOOL/COLLEGE: List all joint tenure-track or tenured appointments including department, college/school (approvals must be obtained). Date of Appointment to Faculty Date and Duration
UIN: RANK: Date: DEPT: SCHOOL/COLLEGE: List all joint tenure-track or tenured appointments including department, college/school (approvals must be obtained). Date of Appointment to Faculty Date and Duration
National Institute for Health Research Coordinated System for gaining NHS Permission (NIHR CSP)
 National Institute for Health Research Coordinated System for gaining NHS Permission (NIHR CSP) Operating Manual Please check the CRN Website for the latest version. Version: 6.0 Status: Consultation in
National Institute for Health Research Coordinated System for gaining NHS Permission (NIHR CSP) Operating Manual Please check the CRN Website for the latest version. Version: 6.0 Status: Consultation in
CROHN S & COLITIS FOUNDATION OF AMERICA. Senior Research Award INSTRUCTIONS. Effective September 2013
 CROHN S & COLITIS FOUNDATION OF AMERICA Senior Research Award INSTRUCTIONS Effective September 2013 Crohn s & Colitis Foundation of America National Office Research & Scientific Programs Department 733
CROHN S & COLITIS FOUNDATION OF AMERICA Senior Research Award INSTRUCTIONS Effective September 2013 Crohn s & Colitis Foundation of America National Office Research & Scientific Programs Department 733
CIP s Open Data & Data Management Guidelines and Procedures
 CIP s Open Data & Data Management Guidelines and Procedures 1.1 Scope The CIP Data Management Guidelines and Procedures aim to provide guidance and support throughout the Data Management Cycle to facilitate
CIP s Open Data & Data Management Guidelines and Procedures 1.1 Scope The CIP Data Management Guidelines and Procedures aim to provide guidance and support throughout the Data Management Cycle to facilitate
Kennedy Krieger Institute. Policy and Procedure for Research Involving Nursing Resources. April 2011
 Kennedy Krieger Institute Policy and Procedure for Research Involving Nursing Resources April 2011 Policy: Human research projects conducted within the Kennedy Krieger Institute (KKI) that require the
Kennedy Krieger Institute Policy and Procedure for Research Involving Nursing Resources April 2011 Policy: Human research projects conducted within the Kennedy Krieger Institute (KKI) that require the
PI - Request for Medical Records
 PI - Request for Medical Records Purpose: Describe the initiation of the PI medical record request process. Identification of Roles: IME-Program Integrity (PI) is responsible for the initiation and completion
PI - Request for Medical Records Purpose: Describe the initiation of the PI medical record request process. Identification of Roles: IME-Program Integrity (PI) is responsible for the initiation and completion
Guidance Document for HRA Statement of Activities for Participating NHS Organisations in England (version 4.1)
 Guidance Document for HRA Statement of Activities for Participating NHS Organisations in England (version 4.1) What is the HRA Statement of Activities? The Statement of Activities is part of the application
Guidance Document for HRA Statement of Activities for Participating NHS Organisations in England (version 4.1) What is the HRA Statement of Activities? The Statement of Activities is part of the application
CONTRACTORS: GL RATER TRAINING GUIDE
 Getting Started: AUI is providing custom direct access rating and servicing solutions to III and Nationwide agencies. This unique end to end platform affords agents one of the quickest self-serve rating
Getting Started: AUI is providing custom direct access rating and servicing solutions to III and Nationwide agencies. This unique end to end platform affords agents one of the quickest self-serve rating
The Ohio State University. Office of Research. Five-Year Business Plan for Research Administrative Units
 The Ohio State University Office of Research Five-Year Business Plan for Research Administrative Units March 16, 2007 Table of Contents Executive Summary 3 Overview of OSU Research Enterprise 3 Strategic
The Ohio State University Office of Research Five-Year Business Plan for Research Administrative Units March 16, 2007 Table of Contents Executive Summary 3 Overview of OSU Research Enterprise 3 Strategic
Fiscal Year End Procedures Workshop. May 12, 2016
 Fiscal Year End Procedures Workshop FISCAL YEAR END The paperless AP office! Departments Presenting Today Student Account Services Purchasing Accounting Services Asset Management Grants & Contracts Payroll
Fiscal Year End Procedures Workshop FISCAL YEAR END The paperless AP office! Departments Presenting Today Student Account Services Purchasing Accounting Services Asset Management Grants & Contracts Payroll
Concur FAQ (Frequently Asked Questions)
 Concur Basics What is my login name? Your employee number (found on your paystub). ######@precisiondrilling.com What if I forget my password? Click the Forgot Password on the Concur page. You will be prompted
Concur Basics What is my login name? Your employee number (found on your paystub). ######@precisiondrilling.com What if I forget my password? Click the Forgot Password on the Concur page. You will be prompted
The Johns Hopkins Technology Ventures Inventor Portal How to Submit an Invention Disclosure Table of Contents
 The Johns Hopkins Technology Ventures Inventor Portal How to Submit an Invention Disclosure Table of Contents 1. Requesting an Account Page 2. 2. Logging In to the Inventor Portal Page 4. 3. Administrative
The Johns Hopkins Technology Ventures Inventor Portal How to Submit an Invention Disclosure Table of Contents 1. Requesting an Account Page 2. 2. Logging In to the Inventor Portal Page 4. 3. Administrative
GRANT & RESEARCH MANAGEMENT REPORT BOOK
 GRANT & RESEARCH MANAGEMENT REPORT BOOK Increase Oversight with Essential Sponsored Program Reports The KualiCo Grant & Research Management suite provides built-in reports to improve oversight and visibility
GRANT & RESEARCH MANAGEMENT REPORT BOOK Increase Oversight with Essential Sponsored Program Reports The KualiCo Grant & Research Management suite provides built-in reports to improve oversight and visibility
Eligibility to Serve as a Principal Investigator for Research Involving Human Subjects
 Eligibility to Serve as a Principal Investigator for Research Involving Human Subjects Statement of Policy Tenured or tenure track Purdue faculty, including emeriti, who are certified by Purdue University
Eligibility to Serve as a Principal Investigator for Research Involving Human Subjects Statement of Policy Tenured or tenure track Purdue faculty, including emeriti, who are certified by Purdue University
THIS ADDENDUM MUST BE ACKNOWLEDGED. THIS ADDENDUM SHALL BECOME A PART OF THIS SOLICITATION.
 Denver Public Schools Purchasing Department 900 Grant Street, Room 301 Denver, Colorado 80203 Request for Proposal BD1354 ADDENDUM NUMBER ONE THIS ADDENDUM MUST BE ACKNOWLEDGED. THIS ADDENDUM SHALL BECOME
Denver Public Schools Purchasing Department 900 Grant Street, Room 301 Denver, Colorado 80203 Request for Proposal BD1354 ADDENDUM NUMBER ONE THIS ADDENDUM MUST BE ACKNOWLEDGED. THIS ADDENDUM SHALL BECOME
Grand Challenges Explorations Round 10 Rules & Guidelines Proposals due Wednesday, November 7, 2012, 11:30 a.m. U.S. Pacific Daylight Time
 Grand Challenges Explorations Round 10 Rules & Guidelines Proposals due Wednesday, November 7, 2012, 11:30 a.m. U.S. Pacific Daylight Time I. Overview Grand Challenges Explorations (GCE) supports hundreds
Grand Challenges Explorations Round 10 Rules & Guidelines Proposals due Wednesday, November 7, 2012, 11:30 a.m. U.S. Pacific Daylight Time I. Overview Grand Challenges Explorations (GCE) supports hundreds
IND Process and Review Procedures (Including Clinical Holds) CONTENTS
 MANUAL OF POLICIES AND PROCEDURES CENTER FOR DRUG EVALUATION AND RESEARCH MAPP 6030.1 REVIEW MANAGEMENT IND Process and Review Procedures (Including Clinical Holds) CONTENTS PURPOSE REFERENCES DEFINITIONS
MANUAL OF POLICIES AND PROCEDURES CENTER FOR DRUG EVALUATION AND RESEARCH MAPP 6030.1 REVIEW MANAGEMENT IND Process and Review Procedures (Including Clinical Holds) CONTENTS PURPOSE REFERENCES DEFINITIONS
Millersville University Graduate Studies and Adult Learning. Master s Thesis Guidelines and Requirements Updated November 2015
 Millersville University Graduate Studies and Adult Learning Master s Thesis Guidelines and Requirements Updated November 2015 I. Definition of a Thesis & Why to Complete One A master s thesis is an approved
Millersville University Graduate Studies and Adult Learning Master s Thesis Guidelines and Requirements Updated November 2015 I. Definition of a Thesis & Why to Complete One A master s thesis is an approved
Grant Application Package
 Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Grant Application Package Reducing
Opportunity Title: Offering Agency: CFDA Number: CFDA Description: Opportunity Number: Competition ID: Opportunity Open Date: Opportunity Close Date: Agency Contact: Grant Application Package Reducing
ACCOUNTS PAYABLE POLICIES AND PROCEDURES
 ACCOUNTS PAYABLE POLICIES AND PROCEDURES...... Accounts Payable Payment Processing General Information Overview of the Disbursement Voucher Disbursement Voucher Approval Payments Against Purchase Orders
ACCOUNTS PAYABLE POLICIES AND PROCEDURES...... Accounts Payable Payment Processing General Information Overview of the Disbursement Voucher Disbursement Voucher Approval Payments Against Purchase Orders
GRANT AND CONTRACT ACCOUNTING GRANT REVIEW TOOLKIT POST AWARD PROCESS
 The below information is a brief overview of the Post Award Process that Grant and Contract Accounting follows in its post award administration of awarded sponsored projects. Please note that this overview
The below information is a brief overview of the Post Award Process that Grant and Contract Accounting follows in its post award administration of awarded sponsored projects. Please note that this overview
Service Level Agreement
 Template SDSU-TPL-11085 v2 20/1/11 IT Services Service Level Agreement HR Management System -SAP (HRMS) Document Ref: DSLAHRMS Version: 0.3 24/03/2011 Contents 1 INTRODUCTION... 4 1.1 Service Outline...
Template SDSU-TPL-11085 v2 20/1/11 IT Services Service Level Agreement HR Management System -SAP (HRMS) Document Ref: DSLAHRMS Version: 0.3 24/03/2011 Contents 1 INTRODUCTION... 4 1.1 Service Outline...
USING DOCUWARE TO MANAGE WORKFORCE SOLUTIONS FILES CAREER OFFICE
 USING DOCUWARE TO MANAGE WORKFORCE SOLUTIONS FILES CAREER OFFICE Using DocuWare to Manage Workforce Solutions Files Workforce Solutions 2015 Version 1.1, 3/17/15 Workforce Solutions is an equal opportunity
USING DOCUWARE TO MANAGE WORKFORCE SOLUTIONS FILES CAREER OFFICE Using DocuWare to Manage Workforce Solutions Files Workforce Solutions 2015 Version 1.1, 3/17/15 Workforce Solutions is an equal opportunity
Loan Site Management - The Bank of Africa
 Challenge ID: Published: Customised, flexible workflow and status reporting solution for managing a loan application process, and online interface between clients and the bank s lending department HELPDESK:
Challenge ID: Published: Customised, flexible workflow and status reporting solution for managing a loan application process, and online interface between clients and the bank s lending department HELPDESK:
National Institutes of Health/Office of Extramural Research. esnap User Guide
 National Institutes of Health/Office of Extramural Research esnap User Guide Version 2.2.3.0 August 01, 2003 Table of Contents Introduction...4 Logging In to esnap...4 Logging In...4 Logging Out...5 Username
National Institutes of Health/Office of Extramural Research esnap User Guide Version 2.2.3.0 August 01, 2003 Table of Contents Introduction...4 Logging In to esnap...4 Logging In...4 Logging Out...5 Username
Xerox Mobile Link App Benefits Examples
 App Benefits Examples Contents Case 1: Healthcare Forms ing Case 2: Auto Lease Case 3: Sales Contract ing Case 4: Enrollment and Registrations Case 5: Insurance Claims ing Case 6: Editorial Markups/ Collaboration
App Benefits Examples Contents Case 1: Healthcare Forms ing Case 2: Auto Lease Case 3: Sales Contract ing Case 4: Enrollment and Registrations Case 5: Insurance Claims ing Case 6: Editorial Markups/ Collaboration
Clinical Trials (Post Award and CRMS) Dorothy Damron, Oncology Research Manager and CRMS Lead Scott Streibich, Clinical Research Revenue Cycle
 Clinical Trials (Post Award and CRMS) Dorothy Damron, Oncology Research Manager and CRMS Lead Scott Streibich, Clinical Research Revenue Cycle 1 Objectives Key CRMS Functionality for Financial RAPs Centralizing
Clinical Trials (Post Award and CRMS) Dorothy Damron, Oncology Research Manager and CRMS Lead Scott Streibich, Clinical Research Revenue Cycle 1 Objectives Key CRMS Functionality for Financial RAPs Centralizing
Transferring Grants. between Institutions. November 2014. You will learn: Grant Transfers
 Transferring Grants between Institutions November 2014 You will learn: Grant Transfers General principles Who is involved Specifics on transferring in and out How an administrator can help Resources available
Transferring Grants between Institutions November 2014 You will learn: Grant Transfers General principles Who is involved Specifics on transferring in and out How an administrator can help Resources available
REQUEST FOR PROPOSALS For. Kelowna and Vernon Hospitals Project
 REQUEST FOR PROPOSALS For The Kelowna and Vernon Hospitals Project VOLUME 2 of 4 Instructions to Proponents Closing Time: Delivery Address: 3:00 pm (local time) Thursday, March 6,2008 Kelowna and Vernon
REQUEST FOR PROPOSALS For The Kelowna and Vernon Hospitals Project VOLUME 2 of 4 Instructions to Proponents Closing Time: Delivery Address: 3:00 pm (local time) Thursday, March 6,2008 Kelowna and Vernon
Wound Healing Society Foundation 3M Fellowship Award. Application and Instructions Deadline for Submission November 21, 2014
 Wound Healing Society Foundation 3M Fellowship Award Application and Instructions Deadline for Submission November 21, 2014 ANNOUNCEMENT The Wound Healing Society Foundation, through the support of 3M
Wound Healing Society Foundation 3M Fellowship Award Application and Instructions Deadline for Submission November 21, 2014 ANNOUNCEMENT The Wound Healing Society Foundation, through the support of 3M
Subject: No. Page PROTOCOL AND CASE REPORT FORM DEVELOPMENT AND REVIEW Standard Operating Procedure
 703 1 of 11 POLICY The Beaumont Research Coordinating Center (BRCC) will provide advice to clinical trial investigators on protocol development, content and format. Upon request, the BRCC will review a
703 1 of 11 POLICY The Beaumont Research Coordinating Center (BRCC) will provide advice to clinical trial investigators on protocol development, content and format. Upon request, the BRCC will review a
Boston Medical Center Policy and Procedure Manual
 Policy #: 39.03.489 Issued: October 2001 Reviewed: Revised: March 2015 Section: 39 Research Subcontract (Subrecipient) Monitoring Policy and Procedure Purpose: To provide guidance to Research Administrators,
Policy #: 39.03.489 Issued: October 2001 Reviewed: Revised: March 2015 Section: 39 Research Subcontract (Subrecipient) Monitoring Policy and Procedure Purpose: To provide guidance to Research Administrators,
ONLINE APPLICATION GUIDELINES Last Updated: December 9, 2015
 endms Scholar Program for Researchers IN Training (SPRINT) ONLINE APPLICATION GUIDELINES Last Updated: December 9, 2015 Multiple Sclerosis Society of Canada Research Department 500-250 Dundas Street West
endms Scholar Program for Researchers IN Training (SPRINT) ONLINE APPLICATION GUIDELINES Last Updated: December 9, 2015 Multiple Sclerosis Society of Canada Research Department 500-250 Dundas Street West
Digital Measures UTSA Faculty Annual Report User s Manual
 The University of Texas at San Antonio Office of the Vice Provost for Institutional Effectiveness Digital Measures UTSA Faculty Annual Report User s Manual Revised: 11/12/2015 UTSA Faculty Annual Report
The University of Texas at San Antonio Office of the Vice Provost for Institutional Effectiveness Digital Measures UTSA Faculty Annual Report User s Manual Revised: 11/12/2015 UTSA Faculty Annual Report
DISCLOSURE OF INVENTION
 DISCLOSURE OF INVENTION All information requested in this document must be completed in order to expeditiously process this Invention Disclosure. Any missing or incomplete information may delay processing
DISCLOSURE OF INVENTION All information requested in this document must be completed in order to expeditiously process this Invention Disclosure. Any missing or incomplete information may delay processing
EAST CAROLINA UNIVERSITY FACULTY MANUAL PART VII. Faculty Research and Scholarship
 EAST CAROLINA UNIVERSITY FACULTY MANUAL PART VII Faculty Research and Scholarship PART VII FACULTY RESEARCH AND SCHOLARSHIP SECTION II Scholarship/Research/Creative Activity Guidelines CONTENTS I. Funded
EAST CAROLINA UNIVERSITY FACULTY MANUAL PART VII Faculty Research and Scholarship PART VII FACULTY RESEARCH AND SCHOLARSHIP SECTION II Scholarship/Research/Creative Activity Guidelines CONTENTS I. Funded
The Freelance Bookkeeper s Business Blueprint
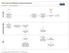 Transition to Transition BK101 Ongoing Set-Up Mgr Notify Ongoing new ready for transition. Set up time to meet Review client s folder and formulates questions prior to meeting Meet with Set-Up Mgr and
Transition to Transition BK101 Ongoing Set-Up Mgr Notify Ongoing new ready for transition. Set up time to meet Review client s folder and formulates questions prior to meeting Meet with Set-Up Mgr and
JOHNS HOPKINS NOTICE OF PRIVACY PRACTICES FOR HOPKINS ELDERPLUS/PROGRAM OF ALL-INCLUSIVE CARE FOR THE ELDERLY (PACE)
 JOHNS HOPKINS NOTICE OF PRIVACY PRACTICES FOR HOPKINS ELDERPLUS/PROGRAM OF ALL-INCLUSIVE CARE FOR THE ELDERLY (PACE) Effective Date: September 1, 2013 THIS NOTICE DESCRIBES HOW MEDICAL INFORMATION ABOUT
JOHNS HOPKINS NOTICE OF PRIVACY PRACTICES FOR HOPKINS ELDERPLUS/PROGRAM OF ALL-INCLUSIVE CARE FOR THE ELDERLY (PACE) Effective Date: September 1, 2013 THIS NOTICE DESCRIBES HOW MEDICAL INFORMATION ABOUT
Seeking Grant Funding 101
 Seeking Grant Funding 101 Jennifer Calzada, MA Director, Tulane Sim Center FIND US : www.simcenter.tulane.edu Why are you here? Medical simulation is an expensive undertaking that requires significant
Seeking Grant Funding 101 Jennifer Calzada, MA Director, Tulane Sim Center FIND US : www.simcenter.tulane.edu Why are you here? Medical simulation is an expensive undertaking that requires significant
Hollie Goddard Sr. IRB Coordinator McKesson Specialty Health
 Hollie Goddard Sr. IRB Coordinator McKesson Specialty Health We are responsible for acquiring, analyzing, and protecting medical information vital to providing quality patient care HIM professionals ensure
Hollie Goddard Sr. IRB Coordinator McKesson Specialty Health We are responsible for acquiring, analyzing, and protecting medical information vital to providing quality patient care HIM professionals ensure
