Prevention of Animal Model of Multiple Sclerosis by oral genistein, extracted from soy bean.
|
|
|
- Flora Lucas
- 8 years ago
- Views:
Transcription
1 Prevention of Animal Model of Multiple Sclerosis by oral genistein, extracted from soy bean. Soodeh Razeghi 1, Seyed rafi Arefhosseini 2, Mehrangiz Ebrahimi Mameghani 3, Mansoureh Togha 4,Leyla Roshangar 5, Taki Tarihi 6 Iranian Journal of Neurology, Vol.7, No.26 & 27, Summer & Autumn 2009, Abstract Introduction: Multiple sclerosis (MS) is a chronic and often debilitating demyelinating disease of the central nervous system (CNS). Many of the current treatments are costly, limited in efficacy, and possess unpleasant side effects. These drawbacks bring the protective strategies into the center of attention. Genistein is one of the components of soy bean which happen to have anti-inflammatory, antioxidant and neuroprotective properties. We conducted this study to assess the potential role of genistein in protection against MS. Methods and Materials: We used Experimental Autoimmune Encephalomyelitis (EAE) -an inducible CD4 + T cell-mediated autoimmune disease with pathological features similar to MS. Oral administration of 20 mg/ kg genistein to MOGimmunized mice was started 21 days before the induction of disease and continued for 42 days. Results: It significantly prevented mice from prolonged neurological sequel by postponing the disease onset, attenuating EAE course and alleviating the severity of clinical and histological signs of EAE. Genistein impose its beneficial role by modulating T-helper1/T-helper2 balance by decreasing CD4 + T-helper 1 cells. Furthermore it inhibited encephalogenic and systemic release of IFN-γ and IL-12 and encephalogenic secretion of TNF-α. On the contrarily it increase the production of IL-10 in central nervous system and spleen. Conclusion: Taking together, these results suggest that genistein may represent a new protective approach for MS by anti-inflammatory and immune-modulating properties. Keywords: Genistein, Experimental Autoimmune Encephalomylitis(EAE), Anti-inflamatory 1 Assistant Professor of nutrition biochemistry, Nutrition and Health Faculty, Tabriz University of Medical Sciences.Arefhosseinir@tbzmed.ac.ir 2 Assistant Professor of nutrition epidemiology, Nutrition and Health Faculty, Tabriz University of Medical Sciences 3 Associate Professor of neurology, Iranian Neurological Research Center, Tehran University of Medical Sciences 4 Assistant Professor of pathology, Medicine Faculty, Tabriz University of Medical Sciences 5 Professor of Anatomy, Medicine Faculty, Tarbiat Modaress University 6 Student of PhD in nutrition. Nutrition and Health Faculty, Tabriz University of Medical Sciences
2 506 / Iran. J. Neurol. Vol.8; No. 26 & 27, Summer & Autumn 2009 Introduction Multiple sclerosis (MS) is a complex demyelinating disease of the central nervous system that afflicts approximately two-and-a-half million individuals worldwide3. (1) It identified as the most debilitating neurological disease in young adults that can lead to irreversible clinical disability. (2) The exact etiology and pathology of MS remains unknown. Different genetic and environmental factors have been implicated in the etiology of MS. Regardless of etiology, most experts agree that MS is an immune-mediated disease. (3,4) This immune attack is mediated by unregulated autoreactive T cells that enter CNS through a disrupted blood-brain-barrier (5) and attack oligodendrocytes as well as the myelin surrounding axons. (6) CD4 + T cells are considered essential in the initial stages of CNS inflammation; however, both adaptive (CD8 + T cells and antibodies) and innate (microglia/macrophages) immunity also play crucial roles to inflammation and tissue injury. (7,6) Cytokines are involved in many of the key pathological feature of MS. Many Th1 cytokines are increased while Th2 cytokines are decreased. Most of the currently approved medications are only partially effective, given parenterally, and often associated with adverse effects and potential toxicities. They also tend to be relatively expensive. Furthermore no approved medication is available for progressive form of MS. (8) These drawbacks bring the protective strategies into the center of attention. There are a number of experimental and clinical evidences to support a protective role for estrogen in MS. The protective effects of estrogen may be mediated by its immunomodulatory mechanism. (9) Phytostrogens comprise a family of biologically active plant agents, possessing estrogenic effect. (10) Phytosterogens have the potential to protect against other disease of immune origin such as diabetes and cancer. (11,12) The most widely distributed phytosterogens are isoflavons, which found in high-concentration in soy bean. Gensitein is the most potent isoflavone of soy bean which happen to have anti-inflammatory, antioxidant and neuroprotective properties that may potentiate it in protection against MS. (12-14) Experimental autoimmune encephalomyelitis (EAE) is an inducible T cell-mediated autoimmune disease. The pathological features of EAE are reminiscent of MS. (15) We conducted this study to assess the ability of genistein to protect against EAE. Methods and Materials Induction of EAE EAE was performed on weeks female C57Bl/6 mice (22.4 gr; Pasteur Institute of Iran). Animals were housed in Shefa Neuroscience research center, with access to food and water ad libitum on 12 h light-dark cycle. All protocols used in this experiment were conducted in accordance with approval by the Tabriz University of medical
3 507 / Prevention of Animal Model of Multiple Sclerosis sciences committee on laboratory animals. Mice were immunized with a 1:1 ratio of MOG (Alexis, Switzerland) dissolved in Complete Freund s adjuvant (CFA) containing 0.4 mg of mycobactererium tuberculosis (Sigma-Aldrich, USA). For this purpose 300 μg of MOG dissolved in 100 μl PBS and mixed with equal volume of CFA. On day 0, the MOG-CFA emulsion was injected subcutaneously into two sites of the upper flunks (200 μg/ mouse). The additional immune adjuvant, pertussis toxin (PTX), (Sigma-Aldrich, USA) was injected i.p (400 ng/mouse) on day 0 and two. Genistein administration Genistein (20 mg/kg) (LC laboratories, USA) dissolved in DMSO 4% (Sigma Aldrich, USA) or DMSO 4% as the vehicle control, was administered daily by oral gavage beginning on 21 days before immunization (n= 6 mice/group) and continued for 42 days. A second control group was not induced with MOG (n= 6 mice/group). Clinical evaluation of EAE The clinical score of each mouse were assessed daily over the duration of 21 days. The following grading scheme was used to clinically score the animals: 0, no clinical signs; 0.5, hook tail; 1, flaccid/floppy tail; 2, walking deficits; 2.5, unilateral hind limb paralysis 3, bilateral hind limb paralysis; 3.5, paraplegia with forelimb weakness; 4 quadriplegia and 5, moribund. Histological evaluation To examine for the presence of cellular infiltrate and demyelination, hematoxylin (MERK, Germany) and eosin (MERK, Germany) and Luxol Fast Blue (Merk, Germany)/Crystal violet (BDH, England) stain was performed respectively. Mice were sacrificed 22 days after immunization. The brains and spinal cords of the animals were removed and post-fixed in 10% paraformaldehid and stored at 4 O C for 48 hours. Prior to sectioning, tissues were put in phosphate buffer saline 10% for 24 h and imbedded in paraffin. Sagittal lumbar spinal cord sections (8 microns) and cross sectional forebrain sections (8 microns) were cut on the cryostat and stained with hematoxylin and eosin and Luxol Fast Blue/Crystal violet. The number of leukocytes and demyelinated foci per mm 2 were determined by assessing at least 10 sections for each mouse. Determination CNS concentration of cytokines For determining the cytokine concentration of CNS, 100 mg of brain tissue of each mouse were homogenized in 10 ml of extraction solution contained: 50 μm tris (Sigma- Aldrich, USA), 2 mm EDTA (MERK, Germany), 0.1 M NaCl (Sigma- Aldrich, USA), 1 mm dithiotheritol (MERK, Germany), and 200 μm phenylmethylsulfonyl fluride (MERK, Germany), and sonicated for 60 seconds at hertz on ice. Brain homogenate was centrifuged (10000*10 min at 4 o C) and supernatant was analyzed for TNF-γ, TNF-α, IL-10 and IL-12 concentration
4 508 / Iran. J. Neurol. Vol.8; No. 26 & 27, Summer & Autumn 2009 using sandwich-based ELISA kits (ELISA Ready-SET-Go! (ebioscience, USA) following manufacturer s instruction. All tests were performed in triplicate for each mouse. Determination systemic concentration of cytokines 24 hours after the last oral administration, mice were sacrificed and their splenocytes were isolated. Mononuclear cells from spleens of immunized mice at a concentration of cells/well in 24-well plates (Nunc, Denmark) were incubated for 2 days in a total volume of 1.5 ml of RPMI-1640 supplemented with 10% FCS, 1% L- glutamine, 1% HEPES, 0.1% 2ME, 0.1% penicillin/streptomycin and pulsed with 10 µg genistein. The cell supernatants were collected and assayed for the presence of cytokines using commercially available sandwich-based ELISA kits (ELISA Ready-SET-Go! (ebioscience, USA) following manufacturer s instruction. All tests were performed in triplicate for each mouse. Lymphocyte proliferation assay (LPA) 24 hours after the last oral administration, single cell suspension of mononuclear cells obtained from immunized mice was used for lymphocyte proliferation assay. Briefly, the suspension of isolated spleen cells was treated with lyses buffer 0.15 M NH4Cl, 1 mm KHCO3, 0.1 mm Na2EDTA, (ph 7.2) in order to clear the red blood cells. In 96-well flat-bottom culture plates (Nunc, Denmark) 2x10 5 cells/well were cultured in 96-well flat-bottom culture plates (Nunc, Denmark). The preparations were cultured with RPMI supplemented with 10% fetal calf serum, 1% L-glutamine, 1% HEPES, 0.1% 2ME, 0.1% penicillin/streptomycin and incubated in the presence of 10 µg genistein. T cell mitogen PHA (phytohemaglotinin, Sigma chemicals) at a concentration of 5 µg/ml, was used in as positive control. After 3 days, MTT (3-(4,5- dimethyl tetrazolyl-2) 2,5 diphenyl) tetrazolyumbromide (Sigma chemicals) in concentration of 5 µg/ml was added per well and incubated for 5 h at 37 0 C in 5% CO2. DMSO (dimethyl sulfoxide) (100 µl) was added to dissolve produced formazan crystals. Plates were read at 540 nm, and the results were expressed as stimulation index (SI). The SI was determined as follows: OD values of stimulated cells (Cs) minus relative cell numbers of unstimulated cells (Cu) by relative OD values of unstimulated cells. SI = (Cs _ Cu)/Cu All tests were performed in triplicate for each mouse. Data Analysis The effect of genistein on clinical scores and histological characteristics (inflammation and demyelination) was analyzed with independent-student-ttest between genistein and vehicle treated groups. One way analysis of variance (ANOVA) with scheff post hok comparison was performed to compare cytokine concentration
5 509 / Prevention of Animal Model of Multiple Sclerosis between three groups. Data were analyzed with SPSS 15. Results Clinical score assay To evaluate the protective effect of genistein in EAE, C57Bl/6 mice were put on oral therapy with genistein form 21 days before immunization with MOG to day 21 following induction. Genistein prevented mice from prolonged neurological sequel as shown by a delay in disease onset (on day 14±2.7 vs 9.17±1.72 post induction) and EAE course attenuation (5.2±3.49 vs 11.17±1.47 days) (fig.1). It also alleviates the severity of the disease. The mean daily score of genistein group was always lower than vehicle group and they have significantly lower maximum (1.9±0.81 vs 3.17±1.03) and mean disease score (fig.1). Incidence Onset Peak Course of disease (days)- Mean clinical score mean± Std. Error DMSO 6/6 9.17± ± ± ±0.26 Genistein 5/6 14±2.74* 1.9±0.81* 5.2±3.49** 0.42±0.2** Figure 1: Effects of orally-administered genistein (20mg/kg) on clinical scores of EAE-induced mice after the onset of clinical signs. All mice received genistein or vehicle (6 mice/ group) from 21 days before induction, were induced with EAE on day 0 and continued treatment for 21 days post immunization on daily bases. Significant differences in clinical scores between DMSO and genistein treated group were observed from day 11 to 21. Genistein treated mice had significant difference in mean clinical score, peak score, and mean duration of EAE compared to vehicle. All date, except for disease course, were expressed as mean±sd. P. values are in comparison with vehicle-treated group. *p<0.05, **p<0.005
6
7 510 / Iran. J. Neurol. Vol.8; No. 26 & 27, Summer & Autumn 2009 Histological assay Histological examination of brain and spinal cord of mice demonstrated significant lower level of inflammation and demyelination in genistein-treated mice (fig.2). Demyelinated lesions scattered widely throughout spinal cord and brain in DMSO-treated mice which covers 39.84±8.09 % and 45.98±5.6% of spinal cord and brain white matter respectively compared to 11.62±5.16% and 7.19±2.96% in genistein-treated animals (fig.2). A-1 A-2 B-1 B-2 C Number of Percent of infiltrates/mm 2 (Mean± SD) demyelination (mean± SE) DMSO Spinal cord 66.7±33.02** 39.84±8.09 ** group Brain 42.16±12.14** 45.98±5.67** Genistein Spinal cord 4.93± ±5.16 group Brain 2.77± ±2.96 Figure 2: Cellular infiltration and demylination in the spinal cords of treated (with DMSO or genistein) mice. Spinal cord sections were stained from vehicle-treated and genistein(20mg/kg)-treated EAE mice and stained using H&E (A-1 and B-1) and LFB& crystal violet(a-2 and B-2). Massive demyelination and Infiltration were noticed in vehicle treated mice(a), while cellular infiltrates and demyelination were noticeably absent in genistein treated mice (B). Number of infiltrates in each mm2 and percent of demyelination were significantly lower in genistein-treated group.. P. values are in comparison with vehicletreated group. *p<0.05, **p<0.005
8 511 / Prevention of Animal Model of Multiple Sclerosis Encaphalogenic and systemic cytokine assay To determine the underling mechanism of genistein protective effect, we evaluated the cytokine pattern of splenocyte. After genistein exposure, lower levels of INF-γ and IL-12 and higher level of IL-10 were detected in spelenocytes that isolated from genistein-treated mice compared to DMSO-treated ones. No significant difference in TNF-α level was noted IFN-γ between genistein and DMSO treated group (fig.3). To evaluate whether genistein has a comparable effect on the CNS cytokine profile, we measured IL-10, IL-12, TNF-α and IFN-γ in brain supernatant. As presented in figure 4, genistein reduced the production of IL-12, TNF-α and IFN-γ. It further increased IL-10 level. These data revealed that genistein imposes its beneficial role by decreasing Th1 and Figure3: Spleen concentration of cytokines (IFN-γ, TNF-α, IL-10 and IL-12). The concentration of cytokines was measured in three groups of mice after restimulating with genistein. These groups were included of control group (not immunized with MOG) (6 mice/ group), immunized with MOG and treated either with DMSO (6 mice/ group) or with genistein (6 mice/ group) from 21 days before to 21 days after immunization. All data 512 / Iran. J. Neurol. Vol.8; expressed No. 26 as & the 27, Summer mean±sd. & Autumn *p<0.05, 2009 **p<0.005
9 IFN-γ Figure 4: Brain concentration of cytokines (IFN-γ, TNF-α, IL-10 and IL-12). The concentration of cytokines was measured in three groups of mice including control group (not immunized with MOG) (6 mice/ group), immunized with MOG and treated either with DMSO (6 mice/ group) or with genistein (6 mice/ group) from 21 days before to 21 days after immunization. All data expressed as the mean±sd. *p<0.05, **p<0.005 Lymphocyte proliferation assay Since lymphocyte proliferative responses are generally considered as measure of cell-mediated immunity, genistein specific lymphocyte proliferation was evaluated using MTT assay. As presented in Fig.5, immunized mice with genistein, induced significantly less specific proliferation response, in comparison with control groups which received DMSO. This data indicate the immunomodulatorory capability of genistein Control DMSO Genistein 0 Control DMSO Genistein Figure 5: Lymphocyte proliferation evaluation, using MTT assay. Lymphocyte proliferation response was measured in non-immunize, DMSO-treated and genistein-treated group. Genistein-treated mice, induced less specific proliferation response, in comparison with control groups which received DMSO (p<0.05). All data expressed as the mean±sd.
10 513 / Prevention of Animal Model of Multiple Sclerosis Discussion We have demonstrated that although oral administration of genistein, the major isoflavone of soy bean, didn t prevent from EAE, it significantly delayed the onset, decreased the severity and shorten the course of disease (Fig. 1). More ever it ameliorates inflammation and subsequent demylination in CNS (Fig. 2). Cellular infiltrate, the hallmark characteristics of EAE, and subsequent demyelination is noticeably absent in genistein-treated group (Fig. 4). According to our results, genistein protective properties can be ascribed to its immmunomodulatory effects. As mentioned in results genistein suppressed the systemic and CNS production of inflammatory cytokines (IL-12 and IFN-γ) production (Fig. 3 and 4). Furthermore it lowered TNF-α secretion in CNS (Fig. 3). In contrast it evidently raised the production of IL-10, an immune-suppressing cytokine, in spleen and CNS (fig.3and4). As shown in MTT test, it also decreased CD4+ T. helper 1 cells proliferation which means genistein can modulate the balance of Th1/Th2. In the majority of cases, many Th1 cytokines are increased in MS and EAE, while Th2 cytokines are decreased. Several Th2 cytokines have been implicated in survival of neurons. In contrast one of the key factors which involved in the process of demyelination is Th1 cytokines. (15) Th1 proinflammatory cytokines, such as TNF-α, IL-1, IL-6, and cellular immunity enhancers such as IFN-γ have been shown to enhance demyelination, oligodendrocyte apoptosis, and axonal loss. (16-20) In MS and EAE IL-12 is believed to promote Th1 polarization by stimulating IFN- γ secretion, (21) anti-il- 12 can augment EAE signs. (22) On the other hand anti-inflammatory (Th2) cytokines such as IL-10 has been identified to contribute in EAE remission. (23) It may therefore explain why therapies that promote a Th1 to Th2 cytokine-shift are beneficial in MS. In Ms, all approved therapies, in addition to many of those under investigation appear to possess immunomodulatory and antiinflammatory roles as the main mechanism of action. Beta-interferons and Glatiramer acetate are on top of this list. (24, 26) The results of our study showed that genistein targets this key mechanism of disease and works like approved treatments. IFN-γ enhances cellular immunity. In our study genistein decrease the systemic and encephalogenic secretion of IFN-γ. This result was confirmed by MTT test. As MTT test showed, genistein decreased the number of Th-1 lymphocyte in the periphery. Genistein also decreased the production of TNF-α in the brain, without influencing systemic mononuclear cell activity which differentiates it from other protective and therapeutic agents. The ability of genistein to raise IL-10 level and reduce
11 514 / Iran. J. Neurol. Vol.8; No. 26 & 27, Summer & Autumn 2009 the IL-12 production, strengthen its effect. Previously parenteral admission of 200 mg/kg genistein, two weeks after immunization, was reported to decrease the severity of clinical signs of EAE. (27) In their study genistein injection decreased IFN-γ, TNF-α, and IL-12 level in brain and elevated IL-10 secretion in brain and spleen. Splenocyte from genistein-treated group has also produced lesser amount of IFN-γ. Our results clarified that oral genistein in much smaller dose (20 mg/kg) can have protective effect against the prolonged sequel of the disease by having a similar effect on cytokine pattern. Genistein anti-inflammatory properties were also observed in collagen-induced rheumatoid arthritis (CIA). Genistein modulate Th1/Th2 balance in CIA by inhibiting IFN-γ secretion and heightening IL-4 production. (28) More ever genistein ability to induce apoptosis, suppress cell proliferation, attenuate osteoclast and lymphocyte activities, and impose antioxidant effects, was previously documented. (14,29) The results presented in this article demonstrate that genistein can be effective in protecting against prolonged and severe neurological sequel of MS by imposing anti-inflammatory and immune modulating effects. Considering that soy bean is commonly consumed in Japan, one can hypothesize that the beneficial characteristics of soy bean presented here contribute to the lower prevalence of MS in Japanese population. Acknowledgment This study was supported by research vice-chancellor, Tabriz University of Medical Ssciences. The authors thank the staff of Shefa Neuroscience Research Center especially Mrs. Alizadeh and Mrs.Eshaghabadi for their kind cooperation. We also thank Dr.Ghaemi for his enthusiastic technical supports and Prof.Gorji for providing the materials. None of the authors had any financial or personal conflicts of the interest.
12 515 / Prevention of Animal Model of Multiple Sclerosis References 1 National Multiple Sclerosis Society. About MS: Who gets MS? 2008a. Ref Type: Report 2 Pryse-Phillips W and Sloka S. Etiopathogenesis and Epidemiology: Clues to Etiology, in Handbook of Multiple Sclerosis Taylor & Francis Group, New York, NY. 2006: Compston A, Coles A: Multiple sclerosis. Lancet 2002;359: Bjartmar C, Trapp BD: Axonal degeneration and progressive neurologic disability in multiple sclerosis. Neurotox Res 2003;5: Minagar A, Alexander JS: Blood-brain barrier disruption in multiple sclerosis. Mult Scler 2003;9: Prat A, Antel J: Pathogenesis of multiple sclerosis. Curr Opin Neurol 2005;18: Becher B, Bechmann I, Greter M: Antigen presentation in autoimmunity and cns inflammation: How t lymphocytes recognize the brain. J Mol Med 2006;84: Bradley W.G, Daroff R.B, Fenichel GM, Jankovic J. N. Neurology in clinical practices, 5th ed. USA. Elsevier 2008: & Offner H: Neuroimmunoprotective effects of estrogen and derivatives in experimental autoimmune encephalomyelitis: Therapeutic implications for multiple sclerosis. J Neurosci Res 2004;78: Kuiper GG, Lemmen JG, Carlsson B, Corton JC, Safe SH, van der Saag PT, van der Burg B, Gustafsson JA: Interaction of estrogenic chemicals and phytoestrogens with estrogen receptor beta. Endocrinology 1998;139: Bhathena SJ, Velasquez MT: Beneficial role of dietary phytoestrogens in obesity and diabetes. Am J Clin Nutr 2002;76: Chen WF, Huang MH, Tzang CH, Yang M, Wong MS: Inhibitory actions of genistein in human breast cancer (mcf-7) cells. Biochim Biophys Acta 2003;1638: Wei H, Bowen R, Cai Q, Barnes S, Wang Y: Antioxidant and antipromotional effects of the soybean isoflavone genistein. Proc Soc Exp Biol Med 1995;208: Polkowski K, Mazurek AP: Biological properties of genistein. A review of in vitro and in vivo data. Acta Pol Pharm 2000;57: Hendriks JJ, Teunissen CE, de Vries HE, Dijkstra CD: Macrophages and neurodegeneration. Brain Res Brain Res Rev 2005;48: Taupin V, Renno T, Bourbonniere L, Peterson AC, Rodriguez M, Owens T: Increased severity of experimental autoimmune encephalomyelitis, chronic macrophage/microglial reactivity, and demyelination in transgenic mice producing tumor necrosis factor-alpha in the central nervous system. Eur J Immunol 1997;27: Jacobs CA, Baker PE, Roux ER, Picha KS, Toivola B, Waugh S, Kennedy MK: Experimental autoimmune encephalomyelitis is exacerbated by il-1 alpha and suppressed by soluble il-1 receptor. J Immunol 1991;146: Okuda Y, Sakoda S, Fujimura H, Saeki Y, Kishimoto T, Yanagihara T: Il-6 plays a crucial role in the induction phase of myelin oligodendrocyte glucoprotein induced experimental autoimmune encephalomyelitis. J Neuroimmunol 1999;101: Tran EH, Prince EN, Owens T: Ifn-gamma shapes immune invasion of the central nervous system via regulation of chemokines. J Immunol 2000;164: Xiao BG, Ma CG, Xu LY, Link H, Lu CZ: Il-12/ifn-gamma/no axis plays critical role in development of th1-mediated experimental autoimmune encephalomyelitis. Mol Immunol 2008;45: Leonard JP, Waldburger KE, Goldman SJ: Prevention of experimental autoimmune encephalomyelitis by antibodies against interleukin 12. J Exp Med 1995;181: Segal BM, Dwyer BK, Shevach EM: An interleukin il-10/il-12 immunoregulatory circuit controls susceptibility to autoimmune disease. J Exp Med 1998;187:
13 516 / Iran. J. Neurol. Vol.8; No. 26 & 27, Summer & Autumn Chen JQ, Brown TR, Russo J: Regulation of energy metabolism pathways by estrogens and estrogenic chemicals and potential implications in obesity associated with increased exposure to endocrine disruptors. Biochim Biophys Acta 2009;1793: Yong VW, Chabot S, Stuve O, Williams G: Interferon beta in the treatment of multiple sclerosis: Mechanisms of action. Neurology 1998;51: Rieks M, Hoffmann V, Aktas O, Juschka M, Spitzer I, Brune N, Schimrigk S, Przuntek H, Pohlau D: Induction of apoptosis of cd4+ t cells by immunomodulatory therapy of multiple sclerosis with glatiramer acetate. Eur Neurol 2003;50: Lindsey JW. EAE: Histology, Clinical Signs, and Disease Course, in Experimental Models of Multiple Sclerosis. Springer Science+ Business Media, Inc., New York, NK. 2005: De Paula ML, Rodrigues DH, Teixeira HC, Barsante MM, Souza MA, Ferreira AP: Genistein down-modulates pro-inflammatory cytokines and reverses clinical signs of experimental autoimmune encephalomyelitis. Int Immunopharmacol 2008;8: Wang J, Zhang Q, Jin S, He D, Zhao S, Liu S: Genistein modulate immune responses in collagen-induced rheumatoid arthritis model. Maturitas 2008;59: Chen X, Garner SC, Quarles LD, Anderson JJ: Effects of genistein on expression of bone markers during mc3t3-e1 osteoblastic cell differentiation. J Nutr Biochem 2003;14:
14 اثرات جنیستي ین خوراکی استخراج شده از سویا در پیشگیري از مدل حیوانیMS سوده رازقی سید رفیع عارف حسینی مهرانگیز ابراهیمی ممقانی منصوره تقاء لیلا روشنگر تقی الطریحی چکیده سابقه و هدف: مرکزي است فصلنامه علوم مغزواعصاب ایران سال هفتم شماره 26 و 27 تابستان و پاییز بیماري اسکلروز منتشر( MS ) شایعترین بیماري مزمن دمیلینه کننده دستگاه عصبی که با کاهش برگشت ناپذیر توان عملکردي همراه است. تا کنون دارویی کاملا اثربخش براي این بیماري عرضه نگردیده است و روشهاي درمانی حاضر پر هزینه داراي عوارض جانبی و نیازمند تزریق هاي متعدد هستند. مشکلات موجود در درمان بیماري MS اهمیت پیشگیري از بیماري را بیشتر کرده است. جنیستي ین یکی از ترکیبات سویا است داراي خواص ضد التهابی آنتی اکسیدانی و نروپروتکتیو می باشد. این مطالعه با هدف بررسی اثر احتمالی جنیستي ین در پیشگیري از بیماري MS طرح ریزي گردید. روش بررسی: در این بررسی از انسفالومیلیت آلرژیک آزمایشی (EAE) در موشهاي C57BL/6 استفاده شد. عوارض پاتولوژیک این بیماري اتوایمیون مشابه MS است و به عنوان مدل حیوانی MS استفاده می شود. براي انجام این بررسی موشها در گروههاي 6 تایی قرار گرفتند. به یک گروه از موشهاي تحت بررسی از 21 روز قبل از ایجاد بیماري تا 21 روز پس از آن 20 میلی گرم به ازاء هر کیلوگرم وزن بدن جنیستي ین به صورت خوراکی داده شد. از DMSO به عنوان حلال جنیستي ین استفاده شد و به یک گروه از موشها به عنوان گروه کنترل بیمار از 21 روز قبل تا 21 روز بعد از ایجاد DMSO EAE داده شد. در گروه سوم بیماري ایجاد نگردید. در طول دوره بررسی شدت علاي م بالینی در دو گروه ثبت گردید. در پایان بررسی هاي هیستولوژي (التهاب و دمیلیناسیون) و ایمونولوژي (میزان TNF-α IFN-γ مغز و طهال) انجام شد IL-10 و IL-12 در یافته ها: دریافت خوراکی جنیستي ین با کاهش معنی دار شدت و مدت بیماري همراه بود. همچنین به طور معنیدار بروز بیماري را به هیستولوژي تعویق انداخت. (التهاب و دملیناسیون در برشهاي مغز و نخاع) نشان داد جنیستي ین اثر خود را با کاهش سیستمیک اثر جنیستي ین در کاهش شدت بیماري با بررسی هاي نیز تا یید شد. (بررسی طحال ( بررسی وضعیت سایتوکین ها و انسفالوژنیک سیتوکین هاي پیش التهابی IFN-γ) TNF-α و (IL-12 و افزایش سیتوکین هاي ضد التهابی (IL-10) اعمال می کند. نتیجه گیري: نتایج این بررسی نشان داد جنیستي ین با دارا بودن خواص ضد التهابی می تواند به پیشگیري از بیماري واژگان کلیدي: MS کمک کند. جنیستي ین انسفالومیلیت آلرژیک آزمایشی ضد التهابی
Genistein Induces a Protective Immunomodulatory Effect in a Mouse Model of Cervical Cancer
 Genistein Induces a Protective Immunomodulatory Effect in a Mouse Model of Cervical Cancer Amir Ghaemi 1,2*, Hoorieh Soleimanjahi 3, Soodeh Razeghi 4, Ali Gorji 2,5, Alijan Tabaraei 1, Abdolvahab Moradi
Genistein Induces a Protective Immunomodulatory Effect in a Mouse Model of Cervical Cancer Amir Ghaemi 1,2*, Hoorieh Soleimanjahi 3, Soodeh Razeghi 4, Ali Gorji 2,5, Alijan Tabaraei 1, Abdolvahab Moradi
Macrophage Migration Inhibitory Factor: A Key Mediator of Inflammation. Aaron Kithcart, B.S.
 Macrophage Migration Inhibitory Factor: A Key Mediator of Inflammation Aaron Kithcart, B.S. Introduction Jean-Martin Charcot first distinguished multiple sclerosis (MS) from other neurological diseases
Macrophage Migration Inhibitory Factor: A Key Mediator of Inflammation Aaron Kithcart, B.S. Introduction Jean-Martin Charcot first distinguished multiple sclerosis (MS) from other neurological diseases
The Prospect of Stem Cell Therapy in Multiple Sclerosis. Multiple sclerosis is a multifocal inflammatory disease of the central
 The Prospect of Stem Cell Therapy in Multiple Sclerosis Multiple sclerosis is a multifocal inflammatory disease of the central nervous system that generally affects young individuals, causing paralysis
The Prospect of Stem Cell Therapy in Multiple Sclerosis Multiple sclerosis is a multifocal inflammatory disease of the central nervous system that generally affects young individuals, causing paralysis
Autoimmunity. Autoimmunity. Genetic Contributions to Autoimmunity. Targets of Autoimmunity
 Autoimmunity Factors predisposing an individual to autoimmune disease Mechanisms of initiation of autoimmunity Pathogenesis of particular autoimmune disease Animal models of autoimmune disease Treatment
Autoimmunity Factors predisposing an individual to autoimmune disease Mechanisms of initiation of autoimmunity Pathogenesis of particular autoimmune disease Animal models of autoimmune disease Treatment
The role of IBV proteins in protection: cellular immune responses. COST meeting WG2 + WG3 Budapest, Hungary, 2015
 The role of IBV proteins in protection: cellular immune responses COST meeting WG2 + WG3 Budapest, Hungary, 2015 1 Presentation include: Laboratory results Literature summary Role of T cells in response
The role of IBV proteins in protection: cellular immune responses COST meeting WG2 + WG3 Budapest, Hungary, 2015 1 Presentation include: Laboratory results Literature summary Role of T cells in response
Multiple Sclerosis (MS) Aprile Royal, Novartis Pharma Canada Inc. September 21, 2011 Toronto, ON
 Multiple Sclerosis (MS) Aprile Royal, Novartis Pharma Canada Inc. September 21, 2011 Toronto, ON First-line DMTs Reduce Relapse Frequency by ~30% vs. Placebo Frequency of relapse with various DMTs, based
Multiple Sclerosis (MS) Aprile Royal, Novartis Pharma Canada Inc. September 21, 2011 Toronto, ON First-line DMTs Reduce Relapse Frequency by ~30% vs. Placebo Frequency of relapse with various DMTs, based
Mouse IFN-gamma ELISpot Kit
 Page 1 of 8 Mouse IFN-gamma ELISpot Kit Without Plates With Plates With Sterile Plates Quantity Catalog Nos. 862.031.001 862.031.001P 862.031.001S 1 x 96 tests 862.031.005 862.031.005P 862.031.005S 5 x
Page 1 of 8 Mouse IFN-gamma ELISpot Kit Without Plates With Plates With Sterile Plates Quantity Catalog Nos. 862.031.001 862.031.001P 862.031.001S 1 x 96 tests 862.031.005 862.031.005P 862.031.005S 5 x
New Treatment Options for MS Patients: Understanding risks versus benefits
 New Treatment Options for MS Patients: Understanding risks versus benefits By Michael A. Meyer, MD Department of Neurology, Sisters Hospital, Buffalo, NY Objectives: 1. to understand fundamentals of MS
New Treatment Options for MS Patients: Understanding risks versus benefits By Michael A. Meyer, MD Department of Neurology, Sisters Hospital, Buffalo, NY Objectives: 1. to understand fundamentals of MS
Programa Cooperación Farma-Biotech Neurociencias NT-KO-003
 Programa Cooperación Farma-Biotech Neurociencias NT-KO-003 A new oral treatment for Multiple Sclerosis based on a novel mechanism of action Barcelona, 15 de febrero 2011 Programa Cooperación Farma-Biotech
Programa Cooperación Farma-Biotech Neurociencias NT-KO-003 A new oral treatment for Multiple Sclerosis based on a novel mechanism of action Barcelona, 15 de febrero 2011 Programa Cooperación Farma-Biotech
The Immunopathogenesis of Relapsing MS
 The Immunopathogenesis of Relapsing MS Olaf Stüve, M.D., Ph.D. Neurology Section VA North Texas Health Care System Dallas VA Medical Center Departments of Neurology and Neurotherapeutics University of
The Immunopathogenesis of Relapsing MS Olaf Stüve, M.D., Ph.D. Neurology Section VA North Texas Health Care System Dallas VA Medical Center Departments of Neurology and Neurotherapeutics University of
Factors that Lead to the Immunotherapy Gap in Multiple Sclerosis Testing by Karthika Solai
 Factors that Lead to the Immunotherapy Gap in Multiple Sclerosis Testing by Karthika Solai Introduction Multiple sclerosis is a disease that affects the central nervous system. Most doctors and scientists
Factors that Lead to the Immunotherapy Gap in Multiple Sclerosis Testing by Karthika Solai Introduction Multiple sclerosis is a disease that affects the central nervous system. Most doctors and scientists
Basic Science in Medicine
 Medical Journal of th e Islamic Republic of Iran Volume 18 Number 3 Fall 1383 November 2004 Basic Science in Medicine ] EXPANSION OF HUMAN CORD BLOOD PRIMITIVE PROGENITORS IN SERUM-FREE MEDIA USING HUMAN
Medical Journal of th e Islamic Republic of Iran Volume 18 Number 3 Fall 1383 November 2004 Basic Science in Medicine ] EXPANSION OF HUMAN CORD BLOOD PRIMITIVE PROGENITORS IN SERUM-FREE MEDIA USING HUMAN
Vitamin D and multiple sclerosis: an update
 Vitamin D and multiple sclerosis: an update Margherita T Cantorna Observational studies document a positive relationship between vitamin D from the environment (sunlight or diet), circulating vitamin D
Vitamin D and multiple sclerosis: an update Margherita T Cantorna Observational studies document a positive relationship between vitamin D from the environment (sunlight or diet), circulating vitamin D
Recent Multiple Sclerosis (MS)
 neuroscience Promoting Remyelination and Preventing Demyelination New Research Goals in Finding a Therapy for Multiple Sclerosis Priya Rajgopal 11 24 Recent Multiple Sclerosis (MS) research has made it
neuroscience Promoting Remyelination and Preventing Demyelination New Research Goals in Finding a Therapy for Multiple Sclerosis Priya Rajgopal 11 24 Recent Multiple Sclerosis (MS) research has made it
Disclosures. Consultant and Speaker for Biogen Idec, TEVA Neuroscience, EMD Serrono, Mallinckrodt, Novartis, Genzyme, Accorda Therapeutics
 Mitzi Joi Williams, MD Neurologist MS Center of Atlanta, Atlanta, GA Disclosures Consultant and Speaker for Biogen Idec, TEVA Neuroscience, EMD Serrono, Mallinckrodt, Novartis, Genzyme, Accorda Therapeutics
Mitzi Joi Williams, MD Neurologist MS Center of Atlanta, Atlanta, GA Disclosures Consultant and Speaker for Biogen Idec, TEVA Neuroscience, EMD Serrono, Mallinckrodt, Novartis, Genzyme, Accorda Therapeutics
PPAR-γ agonist pioglitazone affects rat gouty arthritis by regulating cytokines
 PPAR-γ agonist pioglitazone affects rat gouty arthritis by regulating cytokines R.-C. Wang and D.-M. Jiang Department of Orthopedics, The First Affiliated Hospital of Chongqing Medical University, Chongqing,
PPAR-γ agonist pioglitazone affects rat gouty arthritis by regulating cytokines R.-C. Wang and D.-M. Jiang Department of Orthopedics, The First Affiliated Hospital of Chongqing Medical University, Chongqing,
Multiple Sclerosis. Matt Hulvey BL A - 615
 Multiple Sclerosis Matt Hulvey BL A - 615 Multiple Sclerosis Multiple Sclerosis (MS) is an idiopathic inflammatory disease of the central nervous system (CNS) MS is characterized by demyelination (lesions)
Multiple Sclerosis Matt Hulvey BL A - 615 Multiple Sclerosis Multiple Sclerosis (MS) is an idiopathic inflammatory disease of the central nervous system (CNS) MS is characterized by demyelination (lesions)
OPG-Fc inhibits ovariectomy-induced growth of disseminated breast cancer cells in bone.
 Title of the poster Authors Sheffield Cancer Research Centre University of Sheffield OPG-Fc inhibits ovariectomy-induced growth of disseminated breast cancer cells in bone. Dr Penelope Ottewell Background
Title of the poster Authors Sheffield Cancer Research Centre University of Sheffield OPG-Fc inhibits ovariectomy-induced growth of disseminated breast cancer cells in bone. Dr Penelope Ottewell Background
Personalised Medicine in MS
 Personalised Medicine in MS Supportive Evidence from Therapeutic Trials Ludwig Kappos Neurology and Department of Biomedicine University Hospital CH-4031 Basel LKappos@uhbs.ch Established partially effective
Personalised Medicine in MS Supportive Evidence from Therapeutic Trials Ludwig Kappos Neurology and Department of Biomedicine University Hospital CH-4031 Basel LKappos@uhbs.ch Established partially effective
FastTest. You ve read the book... ... now test yourself
 FastTest You ve read the book...... now test yourself To ensure you have learned the key points that will improve your patient care, read the authors questions below. The answers will refer you back to
FastTest You ve read the book...... now test yourself To ensure you have learned the key points that will improve your patient care, read the authors questions below. The answers will refer you back to
Focus on CNS Drug Development Treating secondary progressive multiple sclerosis. Simon Wilkinson, CEO
 Focus on CNS Drug Development Treating secondary progressive multiple sclerosis Simon Wilkinson, CEO Forward Looking Statements 2 This Presentation (and any financial information that may be provided by
Focus on CNS Drug Development Treating secondary progressive multiple sclerosis Simon Wilkinson, CEO Forward Looking Statements 2 This Presentation (and any financial information that may be provided by
PRODUCT INFORMATION SHEET Monoclonal antibodies detecting human antigens
 www.iqproducts.nl PRODUCT INFORMATION SHEET Monoclonal antibodies detecting human antigens IFN- γ PURE [RUO] [REF] IQP-160P s 50 tests FITC [RUO] [REF] IQP-160F s 50 tests R-PE [RUO] [REF] IQP-160R s 50
www.iqproducts.nl PRODUCT INFORMATION SHEET Monoclonal antibodies detecting human antigens IFN- γ PURE [RUO] [REF] IQP-160P s 50 tests FITC [RUO] [REF] IQP-160F s 50 tests R-PE [RUO] [REF] IQP-160R s 50
specific B cells Humoral immunity lymphocytes antibodies B cells bone marrow Cell-mediated immunity: T cells antibodies proteins
 Adaptive Immunity Chapter 17: Adaptive (specific) Immunity Bio 139 Dr. Amy Rogers Host defenses that are specific to a particular infectious agent Can be innate or genetic for humans as a group: most microbes
Adaptive Immunity Chapter 17: Adaptive (specific) Immunity Bio 139 Dr. Amy Rogers Host defenses that are specific to a particular infectious agent Can be innate or genetic for humans as a group: most microbes
Effects of Two Proprietary Compounds in Multiple Sclerosis DATE
 Effects of Two Proprietary Compounds in a Model of Multiple Sclerosis DATE Authentication This study was conducted under the terms of a Services Agreement between Inc. and CLIENT dated DATE. Sponsor XXX
Effects of Two Proprietary Compounds in a Model of Multiple Sclerosis DATE Authentication This study was conducted under the terms of a Services Agreement between Inc. and CLIENT dated DATE. Sponsor XXX
A method for histopathological study of the multifocal nature of spinal cord lesions in murine experimental autoimmune encephalomyelitis
 A method for histopathological study of the multifocal nature of spinal cord lesions in murine experimental autoimmune encephalomyelitis Katherine N. Gibson-Corley, Alexander W. Boyden, Mariah R. Leidinger,
A method for histopathological study of the multifocal nature of spinal cord lesions in murine experimental autoimmune encephalomyelitis Katherine N. Gibson-Corley, Alexander W. Boyden, Mariah R. Leidinger,
An introduction to modern MS treatments
 BETAFERON is a Prescription Medicine. Use strictly as directed. Consult your pharmacist or other health professional in case of side effects. BETAFERON is reimbursed for some patients. See your neurologist
BETAFERON is a Prescription Medicine. Use strictly as directed. Consult your pharmacist or other health professional in case of side effects. BETAFERON is reimbursed for some patients. See your neurologist
Mariusz Stasiołek Neurology Department Polish Mother s Memorial Hospital Research Institute, Lodz
 Therapeutic Plasma Exchange in Multiple Sclerosis Relapses Mariusz Stasiołek Neurology Department Polish Mother s Memorial Hospital Research Institute, Lodz MS heterogeneity Multiple Sclerosis differences
Therapeutic Plasma Exchange in Multiple Sclerosis Relapses Mariusz Stasiołek Neurology Department Polish Mother s Memorial Hospital Research Institute, Lodz MS heterogeneity Multiple Sclerosis differences
ANIMALS FORM & FUNCTION BODY DEFENSES NONSPECIFIC DEFENSES PHYSICAL BARRIERS PHAGOCYTES. Animals Form & Function Activity #4 page 1
 AP BIOLOGY ANIMALS FORM & FUNCTION ACTIVITY #4 NAME DATE HOUR BODY DEFENSES NONSPECIFIC DEFENSES PHYSICAL BARRIERS PHAGOCYTES Animals Form & Function Activity #4 page 1 INFLAMMATORY RESPONSE ANTIMICROBIAL
AP BIOLOGY ANIMALS FORM & FUNCTION ACTIVITY #4 NAME DATE HOUR BODY DEFENSES NONSPECIFIC DEFENSES PHYSICAL BARRIERS PHAGOCYTES Animals Form & Function Activity #4 page 1 INFLAMMATORY RESPONSE ANTIMICROBIAL
AUBMC Multiple Sclerosis Center
 AUBMC Multiple Sclerosis Center 1 AUBMC Multiple Sclerosis Center The vision of the American University of Beirut Medical Center (AUBMC) is to be the leading academic medical center in Lebanon and the
AUBMC Multiple Sclerosis Center 1 AUBMC Multiple Sclerosis Center The vision of the American University of Beirut Medical Center (AUBMC) is to be the leading academic medical center in Lebanon and the
The Most Common Autoimmune Disease: Rheumatoid Arthritis. Bonita S. Libman, M.D.
 The Most Common Autoimmune Disease: Rheumatoid Arthritis Bonita S. Libman, M.D. Disclosures Two googled comics The Normal Immune System Network of cells and proteins that work together Goal: protect against
The Most Common Autoimmune Disease: Rheumatoid Arthritis Bonita S. Libman, M.D. Disclosures Two googled comics The Normal Immune System Network of cells and proteins that work together Goal: protect against
Understanding How Existing and Emerging MS Therapies Work
 Understanding How Existing and Emerging MS Therapies Work This is a promising and hopeful time in the field of multiple sclerosis (MS). Many new and different therapies are nearing the final stages of
Understanding How Existing and Emerging MS Therapies Work This is a promising and hopeful time in the field of multiple sclerosis (MS). Many new and different therapies are nearing the final stages of
2.1.2 Characterization of antiviral effect of cytokine expression on HBV replication in transduced mouse hepatocytes line
 i 1 INTRODUCTION 1.1 Human Hepatitis B virus (HBV) 1 1.1.1 Pathogenesis of Hepatitis B 1 1.1.2 Genome organization of HBV 3 1.1.3 Structure of HBV virion 5 1.1.4 HBV life cycle 5 1.1.5 Experimental models
i 1 INTRODUCTION 1.1 Human Hepatitis B virus (HBV) 1 1.1.1 Pathogenesis of Hepatitis B 1 1.1.2 Genome organization of HBV 3 1.1.3 Structure of HBV virion 5 1.1.4 HBV life cycle 5 1.1.5 Experimental models
Medical Therapies Limited EGM Presentation
 Medical Therapies Limited EGM Presentation Maria Halasz Chief Executive Officer 5 May 2009 1 Agenda 1. Company information 2. Recent developments 3. Business strategy 4. Key value inflection points for
Medical Therapies Limited EGM Presentation Maria Halasz Chief Executive Officer 5 May 2009 1 Agenda 1. Company information 2. Recent developments 3. Business strategy 4. Key value inflection points for
9/16/2014. Anti-Immunoglobulin E (IgE) Omalizumab (Xolair ) Dosing Guidance
 Disclosure Statement of Financial Interest New Therapies for Asthma Including Omalizumab and Anti-Cytokine Therapies Marsha Dangler, PharmD, BCACP Clinical Pharmacy Specialist James H. Quillen VA Medical
Disclosure Statement of Financial Interest New Therapies for Asthma Including Omalizumab and Anti-Cytokine Therapies Marsha Dangler, PharmD, BCACP Clinical Pharmacy Specialist James H. Quillen VA Medical
Vitamin D and its role in immunology: Multiple sclerosis, and inflammatory bowel disease
 Progress in Biophysics and Molecular Biology 92 (2006) 60 64 Review Vitamin D and its role in immunology: Multiple sclerosis, and inflammatory bowel disease Margherita T. Cantorna Department of Veterinary
Progress in Biophysics and Molecular Biology 92 (2006) 60 64 Review Vitamin D and its role in immunology: Multiple sclerosis, and inflammatory bowel disease Margherita T. Cantorna Department of Veterinary
EFFECTS OF IRON ON THE IMMUNE SYSTEM
 EFFECTS OF IRON ON THE IMMUNE SYSTEM Gert Mayer Department of Internal Medicine IV (Nephrology and Hypertension) Medical University Innsbruck Austria Conflicts of interest Research Support: Amgen, Roche,
EFFECTS OF IRON ON THE IMMUNE SYSTEM Gert Mayer Department of Internal Medicine IV (Nephrology and Hypertension) Medical University Innsbruck Austria Conflicts of interest Research Support: Amgen, Roche,
Identification of T-cell epitopes of SARS-coronavirus for development of peptide-based vaccines and cellular immunity assessment methods
 RESEARCH FUND FOR THE CONTROL OF INFECTIOUS DISEASES PKS Chan 陳 基 湘 S Ma 文 子 光 SM Ngai 倪 世 明 Key Messages 1. Subjects recovered from SARS-CoV infection retain memory of cellular immune response to epitopes
RESEARCH FUND FOR THE CONTROL OF INFECTIOUS DISEASES PKS Chan 陳 基 湘 S Ma 文 子 光 SM Ngai 倪 世 明 Key Messages 1. Subjects recovered from SARS-CoV infection retain memory of cellular immune response to epitopes
TABLE OF CONTENT. Page ACKNOWLEDGEMENTS. iii ENGLISH ABSTRACT THAI ABSTRACT. vii LIST OF TABLES LIST OF FIGURES. xvi ABBREVIATIONS.
 x TABLE OF CONTENT ACKNOWLEDGEMENTS ENGLISH ABSTRACT THAI ABSTRACT LIST OF TABLES LIST OF FIGURES ABBREVIATIONS iii iv vii xv xvi xviii CHAPTER I: INTRODUCTION 1.1 Statement of problems 1 1.2 Literature
x TABLE OF CONTENT ACKNOWLEDGEMENTS ENGLISH ABSTRACT THAI ABSTRACT LIST OF TABLES LIST OF FIGURES ABBREVIATIONS iii iv vii xv xvi xviii CHAPTER I: INTRODUCTION 1.1 Statement of problems 1 1.2 Literature
EFFECT OF PREDATED VACCINATION AND CHINESE HERBAL ADJUVANT RABBIT HEMORRHAGIC DISEASE VACCINE ON IMMUNE RESPONSE IN YOUNG RABBITS
 Pathology and Hygiene EFFECT OF PREDATED VACCINATION AND CHINESE HERBAL ADJUVANT RABBIT HEMORRHAGIC DISEASE VACCINE ON IMMUNE RESPONSE IN YOUNG RABBITS Longsheng Yang*, Jiabin Xue, Fang Wang, Yuanliang
Pathology and Hygiene EFFECT OF PREDATED VACCINATION AND CHINESE HERBAL ADJUVANT RABBIT HEMORRHAGIC DISEASE VACCINE ON IMMUNE RESPONSE IN YOUNG RABBITS Longsheng Yang*, Jiabin Xue, Fang Wang, Yuanliang
Vitamin D and MS: Implications for Clinical Practice
 733 Third Avenue New York, NY 10017-3288 Clinical Bulletin Information for Health Professionals Vitamin D and MS: Implications for Clinical Practice Allen C. Bowling, MD, PhD Introduction Recent studies
733 Third Avenue New York, NY 10017-3288 Clinical Bulletin Information for Health Professionals Vitamin D and MS: Implications for Clinical Practice Allen C. Bowling, MD, PhD Introduction Recent studies
Fighting the Battles: Conducting a Clinical Assay
 Fighting the Battles: Conducting a Clinical Assay 6 Vocabulary: In Vitro: studies in biology that are conducted using components of an organism that have been isolated from their usual biological surroundings
Fighting the Battles: Conducting a Clinical Assay 6 Vocabulary: In Vitro: studies in biology that are conducted using components of an organism that have been isolated from their usual biological surroundings
placebo-controlledcontrolled double-blind, blind,
 Clinical Potential of Minocycline for Depression with Psychotic Features Tsuyoshi Miyaoka Department of Psychiatry Shimane University School of Medicine Minocycline 1. Second-generation tetracycline which
Clinical Potential of Minocycline for Depression with Psychotic Features Tsuyoshi Miyaoka Department of Psychiatry Shimane University School of Medicine Minocycline 1. Second-generation tetracycline which
CONTENT. Chapter 1 Review of Literature. List of figures. List of tables
 Abstract Abbreviations List of figures CONTENT I-VI VII-VIII IX-XII List of tables XIII Chapter 1 Review of Literature 1. Vaccination against intracellular pathogens 1-34 1.1 Role of different immune responses
Abstract Abbreviations List of figures CONTENT I-VI VII-VIII IX-XII List of tables XIII Chapter 1 Review of Literature 1. Vaccination against intracellular pathogens 1-34 1.1 Role of different immune responses
Current and future options of MS treatment Prof. Dr. Karl Vass, AKH Wien
 Current and future options of MS treatment Prof. Dr. Karl Vass, AKH Wien European Health Forum, Gastein 6 th October 2010 Multiple Sclerosis is the most common neurological disorder in young Caucasian
Current and future options of MS treatment Prof. Dr. Karl Vass, AKH Wien European Health Forum, Gastein 6 th October 2010 Multiple Sclerosis is the most common neurological disorder in young Caucasian
Immunoablative therapy with autologous hematopoietic stem cell transplantation in the treatment of poor risk multiple sclerosis
 Immunoablative therapy with autologous hematopoietic stem cell transplantation in the treatment of poor risk multiple sclerosis T Kozák, P Lhotáková Department of Clinical Haematology, 3r d School of Medicine,
Immunoablative therapy with autologous hematopoietic stem cell transplantation in the treatment of poor risk multiple sclerosis T Kozák, P Lhotáková Department of Clinical Haematology, 3r d School of Medicine,
A Genetic Analysis of Rheumatoid Arthritis
 A Genetic Analysis of Rheumatoid Arthritis Introduction to Rheumatoid Arthritis: Classification and Diagnosis Rheumatoid arthritis is a chronic inflammatory disorder that affects mainly synovial joints.
A Genetic Analysis of Rheumatoid Arthritis Introduction to Rheumatoid Arthritis: Classification and Diagnosis Rheumatoid arthritis is a chronic inflammatory disorder that affects mainly synovial joints.
Case Report Early Bilateral Cystoid Macular Oedema Secondary to Fingolimod in Multiple Sclerosis
 Case Reports in Medicine Volume 2012, Article ID 134636, 4 pages doi:10.1155/2012/134636 Case Report Early Bilateral Cystoid Macular Oedema Secondary to Fingolimod in Multiple Sclerosis Lei Liu and Fiona
Case Reports in Medicine Volume 2012, Article ID 134636, 4 pages doi:10.1155/2012/134636 Case Report Early Bilateral Cystoid Macular Oedema Secondary to Fingolimod in Multiple Sclerosis Lei Liu and Fiona
MALIGNANT PLEURAL FLUIDS AID MESOTHELIOMA CELL GROWTH AND PROPAGATION
 MALIGNANT PLEURAL FLUIDS AID MESOTHELIOMA CELL GROWTH AND PROPAGATION HUI MIN CHEAH PhD Candidate School of Medicine and Pharmacology University of Western Australia Lung Institute of Western Australia
MALIGNANT PLEURAL FLUIDS AID MESOTHELIOMA CELL GROWTH AND PROPAGATION HUI MIN CHEAH PhD Candidate School of Medicine and Pharmacology University of Western Australia Lung Institute of Western Australia
Targeting Specific Cell Signaling Pathways for the Treatment of Malignant Peritoneal Mesothelioma
 The Use of Kinase Inhibitors: Translational Lab Results Targeting Specific Cell Signaling Pathways for the Treatment of Malignant Peritoneal Mesothelioma Sheelu Varghese, Ph.D. H. Richard Alexander, M.D.
The Use of Kinase Inhibitors: Translational Lab Results Targeting Specific Cell Signaling Pathways for the Treatment of Malignant Peritoneal Mesothelioma Sheelu Varghese, Ph.D. H. Richard Alexander, M.D.
Cytokines pattern of Multiple Sclerosis patients treated with Apitherapy
 Cytokines pattern of Multiple Sclerosis patients treated with Apitherapy Ahmad G. Hegazi, Khaled Al- Menabbawy, Eman H. Abd El- Rahman and Suzette I. Helal National Research Center, Dokki, Giza, Egypt
Cytokines pattern of Multiple Sclerosis patients treated with Apitherapy Ahmad G. Hegazi, Khaled Al- Menabbawy, Eman H. Abd El- Rahman and Suzette I. Helal National Research Center, Dokki, Giza, Egypt
 CAMBRIDGE UNIVERSITY CENTRE FOR BRAIN REPAIR A layman's account of our scientific objectives What is Brain Damage? Many forms of trauma and disease affect the nervous system to produce permanent neurological
CAMBRIDGE UNIVERSITY CENTRE FOR BRAIN REPAIR A layman's account of our scientific objectives What is Brain Damage? Many forms of trauma and disease affect the nervous system to produce permanent neurological
CytoSelect LDH Cytotoxicity Assay Kit
 Product Manual CytoSelect LDH Cytotoxicity Assay Kit Catalog Number CBA-241 960 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction The measurement and monitoring of cell cytotoxicity
Product Manual CytoSelect LDH Cytotoxicity Assay Kit Catalog Number CBA-241 960 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction The measurement and monitoring of cell cytotoxicity
Vitamin D deficiency exacerbates ischemic cell loss and sensory motor dysfunction in an experimental stroke model
 Vitamin D deficiency exacerbates ischemic cell loss and sensory motor dysfunction in an experimental stroke model Robyn Balden & Farida Sohrabji Texas A&M Health Science Center- College of Medicine ISC
Vitamin D deficiency exacerbates ischemic cell loss and sensory motor dysfunction in an experimental stroke model Robyn Balden & Farida Sohrabji Texas A&M Health Science Center- College of Medicine ISC
Life Science Journal 2015;12(8) http://www.lifesciencesite.com. Role of Vitamin D in the Pathogenesis of Multiple Sclerosis. ammalsallam@hotmail.
 Role of Vitamin D in the Pathogenesis of Multiple Sclerosis 1 Ammal A.W. Sallam and 2 Ayman M.H. El-Sharawy 1 Department of Clinical & Chemical Pathology, Research Institute of Ophthalmology, Giza, Egypt.
Role of Vitamin D in the Pathogenesis of Multiple Sclerosis 1 Ammal A.W. Sallam and 2 Ayman M.H. El-Sharawy 1 Department of Clinical & Chemical Pathology, Research Institute of Ophthalmology, Giza, Egypt.
Chapter 43: The Immune System
 Name Period Our students consider this chapter to be a particularly challenging and important one. Expect to work your way slowly through the first three concepts. Take particular care with Concepts 43.2
Name Period Our students consider this chapter to be a particularly challenging and important one. Expect to work your way slowly through the first three concepts. Take particular care with Concepts 43.2
RayBio Creatine Kinase (CK) Activity Colorimetric Assay Kit
 RayBio Creatine Kinase (CK) Activity Colorimetric Assay Kit User Manual Version 1.0 May 28, 2014 RayBio Creatine Kinase Activity Colorimetric Assay (Cat#: 68CL-CK-S100) RayBiotech, Inc. We Provide You
RayBio Creatine Kinase (CK) Activity Colorimetric Assay Kit User Manual Version 1.0 May 28, 2014 RayBio Creatine Kinase Activity Colorimetric Assay (Cat#: 68CL-CK-S100) RayBiotech, Inc. We Provide You
Title: Mapping T cell epitopes in PCV2 capsid protein - NPB #08-159. Date Submitted: 12-11-09
 Title: Mapping T cell epitopes in PCV2 capsid protein - NPB #08-159 Investigator: Institution: Carol Wyatt Kansas State University Date Submitted: 12-11-09 Industry summary: Effective circovirus vaccines
Title: Mapping T cell epitopes in PCV2 capsid protein - NPB #08-159 Investigator: Institution: Carol Wyatt Kansas State University Date Submitted: 12-11-09 Industry summary: Effective circovirus vaccines
NCL Method ITA-6. Leukocyte Proliferation Assay
 NCL Method ITA-6 Leukocyte Proliferation Assay Nanotechnology Characterization Laboratory National Cancer Institute-Frederick SAIC-Frederick Frederick, MD 21702 (301) 846-6939 ncl@mail.nih.gov December
NCL Method ITA-6 Leukocyte Proliferation Assay Nanotechnology Characterization Laboratory National Cancer Institute-Frederick SAIC-Frederick Frederick, MD 21702 (301) 846-6939 ncl@mail.nih.gov December
CFSE Cell Division Assay Kit
 CFSE Cell Division Assay Kit Item No. 10009853 Customer Service 800.364.9897 * Technical Support 888.526.5351 www.caymanchem.com TABLE OF CONTENTS GENERAL INFORMATION 3 Materials Supplied 4 Precautions
CFSE Cell Division Assay Kit Item No. 10009853 Customer Service 800.364.9897 * Technical Support 888.526.5351 www.caymanchem.com TABLE OF CONTENTS GENERAL INFORMATION 3 Materials Supplied 4 Precautions
NEW DIRECTION IN THE IMPROVEMENT OF CLINICAL CONDITIONS IN MULTIPLE SCLEROSIS PATIENTS By F. De Silvestri, E. Romani, A. Grasso
 NEW DIRECTION IN THE IMPROVEMENT OF CLINICAL CONDITIONS IN MULTIPLE SCLEROSIS PATIENTS By F. De Silvestri, E. Romani, A. Grasso Introduction Multiple sclerosis (MS), in its many and varied clinical forms,
NEW DIRECTION IN THE IMPROVEMENT OF CLINICAL CONDITIONS IN MULTIPLE SCLEROSIS PATIENTS By F. De Silvestri, E. Romani, A. Grasso Introduction Multiple sclerosis (MS), in its many and varied clinical forms,
Immunology and immunotherapy in allergic disease
 Immunology and immunotherapy in allergic disease Jing Shen, MD Faculty Advisor: Matthew Ryan, MD The University of Texas Medical Branch Department of Otolaryngology Grand Rounds Presentation February 2005
Immunology and immunotherapy in allergic disease Jing Shen, MD Faculty Advisor: Matthew Ryan, MD The University of Texas Medical Branch Department of Otolaryngology Grand Rounds Presentation February 2005
Autoimmunity and immunemediated. FOCiS. Lecture outline
 1 Autoimmunity and immunemediated inflammatory diseases Abul K. Abbas, MD UCSF FOCiS 2 Lecture outline Pathogenesis of autoimmunity: why selftolerance fails Genetics of autoimmune diseases Therapeutic
1 Autoimmunity and immunemediated inflammatory diseases Abul K. Abbas, MD UCSF FOCiS 2 Lecture outline Pathogenesis of autoimmunity: why selftolerance fails Genetics of autoimmune diseases Therapeutic
Interferon- (1a and 1b) and glatiramer acetate (GA
 The Journal of Immunology Cutting Edge: Oral Type I IFN- Promotes a Th2 Bias and Enhances Suppression of Autoimmune Encephalomyelitis by Oral Glatiramer Acetate 1 Jeanne M. Soos, 2 * Olaf Stüve, Sawsan
The Journal of Immunology Cutting Edge: Oral Type I IFN- Promotes a Th2 Bias and Enhances Suppression of Autoimmune Encephalomyelitis by Oral Glatiramer Acetate 1 Jeanne M. Soos, 2 * Olaf Stüve, Sawsan
Summary and conclusion 2013
 The work presented in the thesis is focused on the problems related to the prostate gland. Benign prostatic hyperplasia (BPH) and prostate cancer (PCa) are the two major problems associated with prostate.
The work presented in the thesis is focused on the problems related to the prostate gland. Benign prostatic hyperplasia (BPH) and prostate cancer (PCa) are the two major problems associated with prostate.
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE. Proposed Health Technology Appraisal
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Proposed Health Technology Appraisal Daclizumab for treating relapsing-remitting multiple Draft scope (pre-referral) Draft remit/appraisal objective To
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Proposed Health Technology Appraisal Daclizumab for treating relapsing-remitting multiple Draft scope (pre-referral) Draft remit/appraisal objective To
TREATING AUTOIMMUNE DISEASES WITH HOMEOPATHY. Dr. Stephen A. Messer, MSEd, ND, DHANP Professor and Chair of Homeopathic Medicine
 TREATING AUTOIMMUNE DISEASES WITH HOMEOPATHY Dr. Stephen A. Messer, MSEd, ND, DHANP Professor and Chair of Homeopathic Medicine AUTOIMMUNE DISEASES An autoimmune disorder occurs when the body s immune
TREATING AUTOIMMUNE DISEASES WITH HOMEOPATHY Dr. Stephen A. Messer, MSEd, ND, DHANP Professor and Chair of Homeopathic Medicine AUTOIMMUNE DISEASES An autoimmune disorder occurs when the body s immune
RELAPSE MANAGEMENT. Pauline Shaw MS Nurse Specialist 25 th June 2010
 RELAPSE MANAGEMENT Pauline Shaw MS Nurse Specialist 25 th June 2010 AIMS OF SESSION Relapsing/Remitting MS Definition of relapse/relapse rate Relapse Management NICE Guidelines Regional Clinical Guidelines
RELAPSE MANAGEMENT Pauline Shaw MS Nurse Specialist 25 th June 2010 AIMS OF SESSION Relapsing/Remitting MS Definition of relapse/relapse rate Relapse Management NICE Guidelines Regional Clinical Guidelines
Sex Hormone Treatments for Multiple Sclerosis
 JOM Volume 27, Number 2, 2012 Synthesis Article 87 Sex Hormone Treatments for Multiple Sclerosis Jessa Haldane, BSc 1 1.ND Candidate, Clinical Intern, Canadian College of Naturopathic Medicine, 1255 Sheppard
JOM Volume 27, Number 2, 2012 Synthesis Article 87 Sex Hormone Treatments for Multiple Sclerosis Jessa Haldane, BSc 1 1.ND Candidate, Clinical Intern, Canadian College of Naturopathic Medicine, 1255 Sheppard
Multiple Sclerosis Update. Bridget A. Bagert, MD, MPH Director, Ochsner Multiple Sclerosis Center
 Multiple Sclerosis Update Bridget A. Bagert, MD, MPH Director, Ochsner Multiple Sclerosis Center None Disclosures First of All. Why is my talk in the Neurodegenerative hour? I respectfully object! Case
Multiple Sclerosis Update Bridget A. Bagert, MD, MPH Director, Ochsner Multiple Sclerosis Center None Disclosures First of All. Why is my talk in the Neurodegenerative hour? I respectfully object! Case
Neurotrophic factors and Their receptors
 Neurotrophic factors and Their receptors Huang Shu-Hong Institute of neurobiology 1 For decades, scientists believed that brain cells of the central nervous system could not regrow following damage due
Neurotrophic factors and Their receptors Huang Shu-Hong Institute of neurobiology 1 For decades, scientists believed that brain cells of the central nervous system could not regrow following damage due
Protein extraction from Tissues and Cultured Cells using Bioruptor Standard & Plus
 Protein extraction from Tissues and Cultured Cells using Bioruptor Standard & Plus Introduction Protein extraction from tissues and cultured cells is the first step for many biochemical and analytical
Protein extraction from Tissues and Cultured Cells using Bioruptor Standard & Plus Introduction Protein extraction from tissues and cultured cells is the first step for many biochemical and analytical
National MS Society Information Sourcebook www.nationalmssociety.org/sourcebook
 National MS Society Information Sourcebook www.nationalmssociety.org/sourcebook Chemotherapy The literal meaning of the term chemotherapy is to treat with a chemical agent, but the term generally refers
National MS Society Information Sourcebook www.nationalmssociety.org/sourcebook Chemotherapy The literal meaning of the term chemotherapy is to treat with a chemical agent, but the term generally refers
Rheumatoid arthritis: an overview. Christine Pham MD
 Rheumatoid arthritis: an overview Christine Pham MD RA prevalence Chronic inflammatory disease affecting approximately 0.5 1% of the general population Prevalence is higher in North America (approaching
Rheumatoid arthritis: an overview Christine Pham MD RA prevalence Chronic inflammatory disease affecting approximately 0.5 1% of the general population Prevalence is higher in North America (approaching
Integrating New Treatments: A Case Based Approach
 Integrating New Treatments: A Case Based Approach JILL CONWAY, MD, MA, MSCE DIRECTOR, MS CENTER DIRECTOR, NEUROLOGY CLERKSHIP AT UNCSOM- CHARLOTTE CAMPUS CAROLINAS HEALTHCARE CENTER Objectives Provide
Integrating New Treatments: A Case Based Approach JILL CONWAY, MD, MA, MSCE DIRECTOR, MS CENTER DIRECTOR, NEUROLOGY CLERKSHIP AT UNCSOM- CHARLOTTE CAMPUS CAROLINAS HEALTHCARE CENTER Objectives Provide
Standard Operating Procedure
 1.0 Purpose: 1.1 The characterisation of of main leukocyte subsets in peripheral blood cells from mice by flow cytometry. Reliable values of frequencies of leukocyte clusters are very much dependent on
1.0 Purpose: 1.1 The characterisation of of main leukocyte subsets in peripheral blood cells from mice by flow cytometry. Reliable values of frequencies of leukocyte clusters are very much dependent on
Conflict of Interest Declaration. Overview of New Medications for Multiple Sclerosis. Assessment Question. Objectives 4/1/2011
 Conflict of Interest Declaration Overview of New Medications for Multiple Sclerosis I or my spouse have no actual or potential conflict of interest in relation to this activity. Crystal Obering, Pharm.D.,
Conflict of Interest Declaration Overview of New Medications for Multiple Sclerosis I or my spouse have no actual or potential conflict of interest in relation to this activity. Crystal Obering, Pharm.D.,
Newborn Stem Cells from Cord Blood and the Brain: Repairing Injury and Improving Function
 BACKROUNDER: Newborn Stem Cells from Cord Blood and the Brain: Repairing Injury and Improving Function Introduction WITH ONGOING DEVELOPMENTS IN RESEARCH, it is estimated that 1 in every 3 people may benefit
BACKROUNDER: Newborn Stem Cells from Cord Blood and the Brain: Repairing Injury and Improving Function Introduction WITH ONGOING DEVELOPMENTS IN RESEARCH, it is estimated that 1 in every 3 people may benefit
25-hydroxyvitamin D: from bone and mineral to general health marker
 DIABETES 25 OH Vitamin D TOTAL Assay 25-hydroxyvitamin D: from bone and mineral to general health marker FOR OUTSIDE THE US AND CANADA ONLY Vitamin D Receptors Brain Heart Breast Colon Pancreas Prostate
DIABETES 25 OH Vitamin D TOTAL Assay 25-hydroxyvitamin D: from bone and mineral to general health marker FOR OUTSIDE THE US AND CANADA ONLY Vitamin D Receptors Brain Heart Breast Colon Pancreas Prostate
STANDARD OPERATING PROCEDURE
 Title: Lymphocyte Proliferation Assay (LPA) Using 3 H- Thymidine Incorporation Assay Core Name: Lloyd Mayer, Mount Sinai Medical Center Effective Date: 02/16/2012 Trial Number: ITN047AI SOP # ITN2800 SOP
Title: Lymphocyte Proliferation Assay (LPA) Using 3 H- Thymidine Incorporation Assay Core Name: Lloyd Mayer, Mount Sinai Medical Center Effective Date: 02/16/2012 Trial Number: ITN047AI SOP # ITN2800 SOP
Laquinimod Polman, C. et al. Neurology 2005;64:987-991
 Laquinimod Polman, C. et al. Neurology 2005;64:987-991 Multicenter, double-blind, randomized trial, patients with RR MS received 0.1 mg or 0.3 mg laquinimod or placebo as three daily tablets for 24 weeks
Laquinimod Polman, C. et al. Neurology 2005;64:987-991 Multicenter, double-blind, randomized trial, patients with RR MS received 0.1 mg or 0.3 mg laquinimod or placebo as three daily tablets for 24 weeks
A disease and antibody biology approach to antibody drug discovery
 A disease and antibody biology approach to antibody drug discovery Björn Frendéus, PhD VP, Preclinical research Presenter: Björn Frendéus Date: 2011-11-08 1 Antibodies have revolutionized Cancer Treatment!
A disease and antibody biology approach to antibody drug discovery Björn Frendéus, PhD VP, Preclinical research Presenter: Björn Frendéus Date: 2011-11-08 1 Antibodies have revolutionized Cancer Treatment!
Original Policy Date
 MP 5.01.20 Tysabri (natalizumab) Medical Policy Section Prescription Drug Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Local Policy/12:2013 Return to Medical Policy Index Disclaimer
MP 5.01.20 Tysabri (natalizumab) Medical Policy Section Prescription Drug Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Local Policy/12:2013 Return to Medical Policy Index Disclaimer
THE SPINAL CORD AND THE INFLUENCE OF ITS DAMAGE ON THE HUMAN BODY
 THE SPINAL CORD AND THE INFLUENCE OF ITS DAMAGE ON THE HUMAN BODY THE SPINAL CORD. A part of the Central Nervous System The nervous system is a vast network of cells, which carry information in the form
THE SPINAL CORD AND THE INFLUENCE OF ITS DAMAGE ON THE HUMAN BODY THE SPINAL CORD. A part of the Central Nervous System The nervous system is a vast network of cells, which carry information in the form
If you were diagnosed with cancer today, what would your chances of survival be?
 Q.1 If you were diagnosed with cancer today, what would your chances of survival be? Ongoing medical research from the last two decades has seen the cancer survival rate increase by more than 40%. However
Q.1 If you were diagnosed with cancer today, what would your chances of survival be? Ongoing medical research from the last two decades has seen the cancer survival rate increase by more than 40%. However
Gene Therapy. The use of DNA as a drug. Edited by Gavin Brooks. BPharm, PhD, MRPharmS (PP) Pharmaceutical Press
 Gene Therapy The use of DNA as a drug Edited by Gavin Brooks BPharm, PhD, MRPharmS (PP) Pharmaceutical Press Contents Preface xiii Acknowledgements xv About the editor xvi Contributors xvii An introduction
Gene Therapy The use of DNA as a drug Edited by Gavin Brooks BPharm, PhD, MRPharmS (PP) Pharmaceutical Press Contents Preface xiii Acknowledgements xv About the editor xvi Contributors xvii An introduction
Which injectable medication should I take for relapsing-remitting multiple sclerosis?
 Which injectable medication should I take for relapsing-remitting multiple sclerosis? A decision aid to discuss options with your doctor This decision aid is for you if you: Have multiple sclerosis Have
Which injectable medication should I take for relapsing-remitting multiple sclerosis? A decision aid to discuss options with your doctor This decision aid is for you if you: Have multiple sclerosis Have
Calming microglia: a future method for treating multiple sclerosis Jarred Younger, PhD University of Alabama at Birmingham
 Calming microglia: a future method for treating multiple sclerosis Jarred Younger, PhD University of Alabama at Birmingham Sonja Paetau, University of Helsinki Disclosures Will be discussing off-label
Calming microglia: a future method for treating multiple sclerosis Jarred Younger, PhD University of Alabama at Birmingham Sonja Paetau, University of Helsinki Disclosures Will be discussing off-label
Natalia Taborda Vanegas. Doc. Sci. Student Immunovirology Group Universidad de Antioquia
 Pathogenesis of Dengue Natalia Taborda Vanegas Doc. Sci. Student Immunovirology Group Universidad de Antioquia Infection process Epidermis keratinocytes Dermis Archives of Medical Research 36 (2005) 425
Pathogenesis of Dengue Natalia Taborda Vanegas Doc. Sci. Student Immunovirology Group Universidad de Antioquia Infection process Epidermis keratinocytes Dermis Archives of Medical Research 36 (2005) 425
Chapter 10. Summary & Future perspectives
 Summary & Future perspectives 123 Multiple sclerosis is a chronic disorder of the central nervous system, characterized by inflammation and axonal degeneration. All current therapies modulate the peripheral
Summary & Future perspectives 123 Multiple sclerosis is a chronic disorder of the central nervous system, characterized by inflammation and axonal degeneration. All current therapies modulate the peripheral
Microbiology AN INTRODUCTION EIGHTH EDITION
 TORTORA FUNKE CASE Microbiology AN INTRODUCTION EIGHTH EDITION Differentiate between innate and acquired immunity. Chapter 17 Specific Defenses of the Host: The Immune Response B.E Pruitt & Jane J. Stein
TORTORA FUNKE CASE Microbiology AN INTRODUCTION EIGHTH EDITION Differentiate between innate and acquired immunity. Chapter 17 Specific Defenses of the Host: The Immune Response B.E Pruitt & Jane J. Stein
Chapter 3. Immunity and how vaccines work
 Chapter 3 Immunity and how vaccines work 3.1 Objectives: To understand and describe the immune system and how vaccines produce immunity To understand the differences between Passive and Active immunity
Chapter 3 Immunity and how vaccines work 3.1 Objectives: To understand and describe the immune system and how vaccines produce immunity To understand the differences between Passive and Active immunity
MULTIPLE MYELOMA. Dr Malkit S Riyat. MBChB, FRCPath(UK) Consultant Haematologist
 MULTIPLE MYELOMA Dr Malkit S Riyat MBChB, FRCPath(UK) Consultant Haematologist Multiple myeloma is an incurable malignancy that arises from postgerminal centre, somatically hypermutated B cells.
MULTIPLE MYELOMA Dr Malkit S Riyat MBChB, FRCPath(UK) Consultant Haematologist Multiple myeloma is an incurable malignancy that arises from postgerminal centre, somatically hypermutated B cells.
CHAPTER 2 ANTIGEN/ANTIBODY INTERACTIONS
 CHAPTER 2 ANTIGEN/ANTIBODY INTERACTIONS See APPENDIX (1) THE PRECIPITIN CURVE; (2) LABELING OF ANTIBODIES The defining characteristic of HUMORAL immune responses (which distinguishes them from CELL-MEDIATED
CHAPTER 2 ANTIGEN/ANTIBODY INTERACTIONS See APPENDIX (1) THE PRECIPITIN CURVE; (2) LABELING OF ANTIBODIES The defining characteristic of HUMORAL immune responses (which distinguishes them from CELL-MEDIATED
6 rubin international edition 2013. Psoriasis drug combats multiple sclerosis
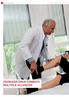 6 rubin international edition 2013 Psoriasis drug combats multiple sclerosis Fig. 1: Prof Dr Ralf Gold examines a patient in the St. Josef Hospital. During an episode, the patients can experience, for
6 rubin international edition 2013 Psoriasis drug combats multiple sclerosis Fig. 1: Prof Dr Ralf Gold examines a patient in the St. Josef Hospital. During an episode, the patients can experience, for
A Definition of Multiple Sclerosis
 English 182 READING PRACTICE by Alyx Meltzer, Spring 2009 Vocabulary Preview (see bolded, underlined words) gait: (n) a particular way of walking transient: (adj) temporary; synonym = transitory remission:
English 182 READING PRACTICE by Alyx Meltzer, Spring 2009 Vocabulary Preview (see bolded, underlined words) gait: (n) a particular way of walking transient: (adj) temporary; synonym = transitory remission:
New research in primary and secondary progressive multiple sclerosis. Dr Claire McCarthy MRCP PhD Neurology SpR Addenbrooke s Hospital
 New research in primary and secondary progressive multiple sclerosis Huntingdon MS Society Annual Meeting Sat 22nd March 2014 Dr Claire McCarthy MRCP PhD Neurology SpR Addenbrooke s Hospital 1 Overview
New research in primary and secondary progressive multiple sclerosis Huntingdon MS Society Annual Meeting Sat 22nd March 2014 Dr Claire McCarthy MRCP PhD Neurology SpR Addenbrooke s Hospital 1 Overview
A link between gut microbiota, mucosal immunity and autoimmune arthritis. Hsin-Jung Joyce Wu "Microbiota and man: the story about us
 A link between gut microbiota, mucosal immunity and autoimmune arthritis Microbes and Man The majority of microbes we encounter are gut microbiota that live mostly in harmony with their host Duodenum 10
A link between gut microbiota, mucosal immunity and autoimmune arthritis Microbes and Man The majority of microbes we encounter are gut microbiota that live mostly in harmony with their host Duodenum 10
Graduate and Postdoctoral Affairs School of Biomedical Sciences College of Medicine. Graduate Certificate. Metabolic & Nutritional Medicine
 Graduate and Postdoctoral Affairs School of Biomedical Sciences College of Medicine Graduate Certificate in Metabolic & Nutritional Medicine Graduate Certificate Metabolic & Nutritional Medicine Purpose
Graduate and Postdoctoral Affairs School of Biomedical Sciences College of Medicine Graduate Certificate in Metabolic & Nutritional Medicine Graduate Certificate Metabolic & Nutritional Medicine Purpose
ACUPUNCTURE AND MULTIPLE SCLEROSIS
 ACUPUNCTURE AND MULTIPLE SCLEROSIS About multiple sclerosis Around 100,000 people in the UK have multiple sclerosis (MS) (MS Society 2012), an autoimmune disorder of the central nervous system (Compston
ACUPUNCTURE AND MULTIPLE SCLEROSIS About multiple sclerosis Around 100,000 people in the UK have multiple sclerosis (MS) (MS Society 2012), an autoimmune disorder of the central nervous system (Compston
Research Article Persistent Inflammation in the CNS during Chronic EAE Despite Local Absence of IL-17 Production
 Mediators of Inflammation, Article ID 519627, 1 pages http://dx.doi.org/1.1155/213/519627 Research Article Persistent Inflammation in the CNS during EAE Despite Local Absence of IL-17 Production Sofia
Mediators of Inflammation, Article ID 519627, 1 pages http://dx.doi.org/1.1155/213/519627 Research Article Persistent Inflammation in the CNS during EAE Despite Local Absence of IL-17 Production Sofia
