CRISP1 Data Archive. The NIDDK Repository offers the following three data packages from the CRISP1 study:
|
|
|
- Rodger Warner
- 8 years ago
- Views:
Transcription
1 CRISP1 Data Archive The NIDDK Repository offers the following three data packages from the CRISP1 study: 1. The standard data archive, which contains a dataset compiled by the Data Center, Center for Research on Health Care at the University of Pittsburgh. The original study protocol and other descriptive documentation and the study forms are also provided in the standard data archive for reference. 2. The raw data archive, which contains the original raw data collected by the study, analysis files associated with study publications, and reports of the analyses performed by statisticians and quality control specialists at the Repository to ensure the completeness and accuracy of the analysis datasets in the Repository. 3. The genetic data archive, which contains raw and summary data collected in an ancillary study analyzing mutation-based molecular diagnostics of autosomal dominant polycystic kidney disease (ADPKD) in the CRISP population. The NIDDK Repository recommends that only the standard data archive be used. Requestors must ask for the genetics and/or raw data archives separately from the standard data archive. The CRISP standard data archive files are organized into the following directories: 1. Documentation 2. Data 1. Documentation Directory The Documentation directory contains the documentation from the original study Documents The Documents directory contains the following three files: CrispMOP2003.pdf - Manual of Operations, which also contains all of the study forms, 257 pages CRISP Datasets.doc - forms and key variables, 5 pages CRISP_Metadata.doc description of the study design, goals, and outcomes, 5 pages Forms The Forms subdirectory contains the following 20 pdf files, which are the forms used in the collection of the raw data by the original study. The forms are annotated with the variable names: Registration Form (Crisp2var_Registration.pdf) Initial Screening Form (Crisp3var_InitialScreening.pdf)
2 Baseline Visit Form (Crisp4var_BaselineVisit.pdf) OTC/Prescribed/Other Drugs (Crisp5var_OTCPrescripDrug.pdf) Reproductive History Form (Crisp6var_ReproductiveHistory.pdf) MR Session Information (Crisp7var_MRSession.pdf) GFR Collection Form (Crisp9var_GFRCollection.pdf) GFR Reporting Form (Crisp10var_GFRMayoReport.pdf) Physical Findings Form (Crisp11var_PhysicalFindings.pdf) Symptoms Form (Crisp12var_Symptoms.pdf) Follow-Up Study and Events Form (Crisp13var_FollowupStudyVisit.pdf) Blood Pressure Form (Crisp14var_BloodPressure.pdf) Family History Form (Crisp16var_FamilyHistory.pdf) Renal Blood Flow (Crisp17var_RenalBloodFlow.pdf) Study Withdrawal/Lost to Followup Form (Crisp19var_StudyWithdrawal.pdf) Missed Visit Form (Crisp24var_MissedVisit.pdf) Annual Visit - Labs (Crisp27var_AnnualVisit.pdf) Annual Clinic Visit (Meds and Events Crisp28var_AnnualClinicVisit.pdf ) Current Reproductive History (Crisp29var_CurrentReproductiveHist.pdf) Current Symptoms (Crisp30var_CurrentSymptom.pdf)
3 2. Data Directory The Data directory contains the dataset compiled by the Data Center at Center for Research on Health Care at the University of Pittsburgh. It contains the updated CRISP I analytic dataset and its data dictionary: repository sas7bdat (11.6 MB) This dataset was compiled by the Data Center, Center for Research on Health Care at the University of Pittsburgh. CRISP1_DataDictionary_Alpha_April2009.xls (18 pages) This document serves as the data dictionary for the updated CRISP I analytic dataset (repository sas7bdat). It also defines missing data codes and includes definitions for all derived variables.
4 CRISP2 Data Archive 11/3/2014 The NIDDK Repository offers the following data package from the CRISP2 study: The CRISP2 standard data archive files are organized into the following directories: 1. Documentation 2. Data 3. DSIC 3. Documentation Directory The Documentation directory contains the documentation from the original study Documents The Documents directory contains the following three files: CRISP_2_MOP.pdf - Manual of Operations CRISP_2_Protocol.pdf - Protocol CRISP_2_Publications.doc - List of Publications CRISP_2_Visit_Definitions.xlsx - description of the visits in the CRISP2 study Codebooks: CRISP_2_Data_Dictionary.xlsx CRISP_2_Long_Files_Var_Names.xlsx Analysis_File_Documentation: CJASN differences documentation.docx CJASN without HALT follow-up_for NIDDK.doc CJASN-Data Dictionary_derived vars.docx 3.2. Forms The Forms subdirectory contains the following 28 pdf files, which are the forms used in the collection of the raw data by the original study. Form_2_Registration.pdf Form_7_MR_Session_Renal_Blood_Flow.pdf Form_9_GFR_Collection.pdf Form_10_GFR_Report.pdf Form_11_Physical_Find.pdf Form_12_Symptoms.pdf Form_13_Follow_UP_Study_and_Events.pdf Form_15_Death_Notification.pdf Form_17_Biannual_Medand_Events.pdf Form_18_Transfer_Form.pdf
5 Form_19_Study_Withdrawl.pdf Form_24_Missed_Visit.pdf Form_27_Biannual_Clinic_Visit_Labs.pdf Form_33_Lab_Visit_7_and_9.pdf Form_34_Scan_Evaluation.pdf Form_40_ObGyn_History.pdf Form_41_QualityofLifeQuesSF36.pdf Form_42_Pain_Questionnaire.pdf Form_45_Manifest_Plasma.pdf Form_47_UrineCollection.pdf Form_49_Manifest_Urine.pdf Form_50_Manifest_Cleve.pdf Form_53_Blood_Collection.pdf Form_54_Missing_Data.pdf Form_55_MRI_Status_Verification.pdf Form_56_Manifest_Rutgers.pdf Form_63_Transitional_Symptoms.pdf Form_64_Endpoints.pdf 11/3/ DSIC Directory The DSIC directory contains the dataset integrity check that was performed on the CRISP2 data. CRISP2_DSIC.pdf Dataset integrity check of the CRISP2 data using the Kidney Volume and Functional Outcomes in Autosomal Dominant Polycystic Kidney Disease publication
HALT-PKD Trials
 Building a Homegrown Data Management System: Perspectives from the HALT-PKD Trials Assistant Professor of Medicine, Biostatistics, and Clinical & Translational Sciences Center for Research on Health Care
Building a Homegrown Data Management System: Perspectives from the HALT-PKD Trials Assistant Professor of Medicine, Biostatistics, and Clinical & Translational Sciences Center for Research on Health Care
Problems and Measures Regarding Waste 1 Management and 3R Era of public health improvement Situation subsequent to the Meiji Restoration

Chapter 14 COORDINATING CENTER PROCEDURES
 Chapter 14 COORDINATING CENTER PROCEDURES 14.1 STAFFING AND ORGANIZATION The Coordinating Center for AREDS is located at The EMMES Corporation in Potomac, Maryland. The staff at the Coordinating Center
Chapter 14 COORDINATING CENTER PROCEDURES 14.1 STAFFING AND ORGANIZATION The Coordinating Center for AREDS is located at The EMMES Corporation in Potomac, Maryland. The staff at the Coordinating Center
What is Clinical Data Management
 What is Clinical Data Management Clinical Data Management is involved in all aspects of processing the clinical data, working with a range of computer applications, database systems to support collection,
What is Clinical Data Management Clinical Data Management is involved in all aspects of processing the clinical data, working with a range of computer applications, database systems to support collection,
Clinical Data Management is involved in all aspects of processing the clinical data, working with a range of computer applications / database systems
 Clinical Data Management is involved in all aspects of processing the clinical data, working with a range of computer applications / database systems to support collection, cleaning and management of subject
Clinical Data Management is involved in all aspects of processing the clinical data, working with a range of computer applications / database systems to support collection, cleaning and management of subject
Definition, Prevalence, Pathophysiology and Complications of CKD. JM Krzesinski CHU Liège-ULg Core curriculum Nephrology September 28 th 2013
 Definition, Prevalence, Pathophysiology and Complications of CKD JM Krzesinski CHU Liège-ULg Core curriculum Nephrology September 28 th 2013 KI supplements January 2013 Objectives of the course on CKD:
Definition, Prevalence, Pathophysiology and Complications of CKD JM Krzesinski CHU Liège-ULg Core curriculum Nephrology September 28 th 2013 KI supplements January 2013 Objectives of the course on CKD:
Site-Specific Data Collection and Management Protocol
 Site-Specific Data Collection and Management Protocol Client Interview and Chart Data- Baseline 1 Study explained 2 Consent obtained Responsible party(ies) Location Protocol Staff NIH certified? Where
Site-Specific Data Collection and Management Protocol Client Interview and Chart Data- Baseline 1 Study explained 2 Consent obtained Responsible party(ies) Location Protocol Staff NIH certified? Where
THE FUTURE OF CLINICAL TRIALS: NEW MODEL TO ENHANCE EFFICIENCY. Jeff Allen, PhD Executive Director Friends of Cancer Research
 THE FUTURE OF CLINICAL TRIALS: NEW MODEL TO ENHANCE EFFICIENCY Jeff Allen, PhD Executive Director Friends of Cancer Research Current Challenges Each potential new therapy is typically tested independently
THE FUTURE OF CLINICAL TRIALS: NEW MODEL TO ENHANCE EFFICIENCY Jeff Allen, PhD Executive Director Friends of Cancer Research Current Challenges Each potential new therapy is typically tested independently
Bench to Bedside Clinical Decision Support:
 Bench to Bedside Clinical Decision Support: The Role of Semantic Web Technologies in Clinical and Translational Medicine Tonya Hongsermeier, MD, MBA Corporate Manager, Clinical Knowledge Management and
Bench to Bedside Clinical Decision Support: The Role of Semantic Web Technologies in Clinical and Translational Medicine Tonya Hongsermeier, MD, MBA Corporate Manager, Clinical Knowledge Management and
Systolic Blood Pressure Intervention Trial (SPRINT) Principal Results
 Systolic Blood Pressure Intervention Trial (SPRINT) Principal Results Paul K. Whelton, MB, MD, MSc Chair, SPRINT Steering Committee Tulane University School of Public Health and Tropical Medicine, and
Systolic Blood Pressure Intervention Trial (SPRINT) Principal Results Paul K. Whelton, MB, MD, MSc Chair, SPRINT Steering Committee Tulane University School of Public Health and Tropical Medicine, and
FSH Society s 2014 Biennial FSHD Connect Meeting: Natural History Studies
 FSH Society s 2014 Biennial FSHD Connect Meeting: Natural History Studies Raymond A. Huml, MS, DVM, RAC Executive Director, Head, Global Biosimilars Business Development and Strategic Planning, Quintiles
FSH Society s 2014 Biennial FSHD Connect Meeting: Natural History Studies Raymond A. Huml, MS, DVM, RAC Executive Director, Head, Global Biosimilars Business Development and Strategic Planning, Quintiles
Guidelines for Telemedicine Scheduling and Patient Care Co-ordination
 Guidelines for Telemedicine Scheduling and Patient Care Co-ordination Content Overview Introduction to Scheduling and Patient Care Coordination 2 2 Model 1: Healthcare Provider Managed Scheduling and Patient
Guidelines for Telemedicine Scheduling and Patient Care Co-ordination Content Overview Introduction to Scheduling and Patient Care Coordination 2 2 Model 1: Healthcare Provider Managed Scheduling and Patient
Digital Health: Catapulting Personalised Medicine Forward STRATIFIED MEDICINE
 Digital Health: Catapulting Personalised Medicine Forward STRATIFIED MEDICINE CRUK Stratified Medicine Initiative Somatic mutation testing for prediction of treatment response in patients with solid tumours:
Digital Health: Catapulting Personalised Medicine Forward STRATIFIED MEDICINE CRUK Stratified Medicine Initiative Somatic mutation testing for prediction of treatment response in patients with solid tumours:
Calculating the stage of Renal Disease
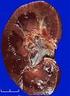 Calculating the stage of Renal Disease When the Refresh Template/Check Labs button is depressed, the box next to MDRD, will be automatically checked. In order to use this in the calculation of the stage
Calculating the stage of Renal Disease When the Refresh Template/Check Labs button is depressed, the box next to MDRD, will be automatically checked. In order to use this in the calculation of the stage
Overview of edx Analytics
 Overview of edx Analytics I. Data Available from edx EdX provides researchers with data about your institution's classes running on edx.org and edge.edx.org. This includes: Course data Student information
Overview of edx Analytics I. Data Available from edx EdX provides researchers with data about your institution's classes running on edx.org and edge.edx.org. This includes: Course data Student information
Bunker Hill Superfund Site 2014 Blood Lead Levels
 Bunker Hill Superfund Site 2014 Blood Lead Levels Panhandle Health District Idaho Department of Environmental Quality United States Environmental Protection Agency 2014 Lead Health Intervention Program
Bunker Hill Superfund Site 2014 Blood Lead Levels Panhandle Health District Idaho Department of Environmental Quality United States Environmental Protection Agency 2014 Lead Health Intervention Program
Design of Data Entry Systems Pam Kellogg, Research Informatics, Family Health International
 Design of Data Entry Systems Pam Kellogg,, Family Health International 1 Data Entry Design Topics CRFs Annotated CRFs Record Layouts Data Dictionaries Code lists Data entry screens Testing QA Documentation
Design of Data Entry Systems Pam Kellogg,, Family Health International 1 Data Entry Design Topics CRFs Annotated CRFs Record Layouts Data Dictionaries Code lists Data entry screens Testing QA Documentation
Understanding. Your Medical Record
 Understanding Your Medical Record Table of Contents Choose topics at right by selecting the phrases. To go to the next or previous page, click on the arrows. To return to the table of contents, please
Understanding Your Medical Record Table of Contents Choose topics at right by selecting the phrases. To go to the next or previous page, click on the arrows. To return to the table of contents, please
Why is this Job Description being written? New Position Replacement Position Position re-designed Position not previously described
 Why is this Job Description being written? New Position Replacement Position Position re-designed Position not previously described P O SITION DET AI L S: Position Title: C LI NICAL DATA MAN AGER Division:
Why is this Job Description being written? New Position Replacement Position Position re-designed Position not previously described P O SITION DET AI L S: Position Title: C LI NICAL DATA MAN AGER Division:
Identifying and treating long-term kidney problems (chronic kidney disease)
 Understanding NICE guidance Information for people who use NHS services Identifying and treating long-term kidney problems (chronic kidney disease) NICE clinical guidelines advise the NHS on caring for
Understanding NICE guidance Information for people who use NHS services Identifying and treating long-term kidney problems (chronic kidney disease) NICE clinical guidelines advise the NHS on caring for
 A Gay Men s Health Service Audit of: Post Exposure Prophylaxis Robert Obara, BSc MPH MBBChBAO Candidate, TCD Class of 2014 Poster for BASHIV, Dr Dominic Rowley (GMHS) et al. PEP Talk Antiretroviral drugs
A Gay Men s Health Service Audit of: Post Exposure Prophylaxis Robert Obara, BSc MPH MBBChBAO Candidate, TCD Class of 2014 Poster for BASHIV, Dr Dominic Rowley (GMHS) et al. PEP Talk Antiretroviral drugs
Oral Health Care: Dental Insurance Program
 Oral Health Care: Dental Insurance Program Approved: November 2009 Oral Health Care includes diagnostic, preventive, and therapeutic services provided by general dental practitioners, dental specialists,
Oral Health Care: Dental Insurance Program Approved: November 2009 Oral Health Care includes diagnostic, preventive, and therapeutic services provided by general dental practitioners, dental specialists,
EXPLORE LEARN RESEARCH. Quick Guide: Overview. The National Children's Study Archive
 Quick Guide: Overview LEARN From the Home page, learn about when and where the available NCS data were collected From the Data & Documentation tab: View high-level Public Data collected under the IVS,
Quick Guide: Overview LEARN From the Home page, learn about when and where the available NCS data were collected From the Data & Documentation tab: View high-level Public Data collected under the IVS,
BG MEDICINE. Corporate Presentation February 2013
 BG MEDICINE Corporate Presentation February 2013 CONFIDENTIAL Oct 9, 2007 Forward-Looking Statements This presentation contains forward-looking statements regarding future events or the future financial
BG MEDICINE Corporate Presentation February 2013 CONFIDENTIAL Oct 9, 2007 Forward-Looking Statements This presentation contains forward-looking statements regarding future events or the future financial
Meaningful use. Meaningful data. Meaningful care. The 3M Healthcare Data Dictionary: Standardizing lab data to LOINC for meaningful use
 Meaningful use. Meaningful data. Meaningful care. The 3M Healthcare Data Dictionary: Standardizing lab data to LOINC for meaningful use Executive summary By using standard terminologies to report on core
Meaningful use. Meaningful data. Meaningful care. The 3M Healthcare Data Dictionary: Standardizing lab data to LOINC for meaningful use Executive summary By using standard terminologies to report on core
Funded by NIH for~30 Years
 Funded by NIH for~30 Years NIMH NHLBI NHGRI NIAMS ORDR NIH Office of the Director ORIP NCI OD NEI NIDDK NIAID NDRI Rare Disease Biospecimen Program Penn Medicine Center for Orphan Disease Research & Therapy
Funded by NIH for~30 Years NIMH NHLBI NHGRI NIAMS ORDR NIH Office of the Director ORIP NCI OD NEI NIDDK NIAID NDRI Rare Disease Biospecimen Program Penn Medicine Center for Orphan Disease Research & Therapy
Albuminuria versus GFR as markers of diabetic CKD progression
 Albuminuria versus GFR as markers of diabetic CKD progression KDIGO Controversies Conference: Diabetic Kidney Disease New Delhi, March 2012 Richard J MacIsaac PhD FRACP Director of Endocrinology & Diabetes,
Albuminuria versus GFR as markers of diabetic CKD progression KDIGO Controversies Conference: Diabetic Kidney Disease New Delhi, March 2012 Richard J MacIsaac PhD FRACP Director of Endocrinology & Diabetes,
Open & Big Data for Life Imaging Technical aspects : existing solutions, main difficulties. Pierre Mouillard MD
 Open & Big Data for Life Imaging Technical aspects : existing solutions, main difficulties Pierre Mouillard MD What is Big Data? lots of data more than you can process using common database software and
Open & Big Data for Life Imaging Technical aspects : existing solutions, main difficulties Pierre Mouillard MD What is Big Data? lots of data more than you can process using common database software and
DIVISION of SUBSTANCE ABUSE SERVICES. Tuberculosis Control Guidelines for Alcohol and Drug Abuse Treatment Programs
 DIVISION of SUBSTANCE ABUSE SERVICES Tuberculosis Control Guidelines for Alcohol and Drug Abuse Treatment Programs 1. Purpose: This policy provides guidance and instruction to identify and prevent tuberculosis
DIVISION of SUBSTANCE ABUSE SERVICES Tuberculosis Control Guidelines for Alcohol and Drug Abuse Treatment Programs 1. Purpose: This policy provides guidance and instruction to identify and prevent tuberculosis
Clinical Trials: Questions and Answers
 Clinical Trials: Questions and Answers Key Points Clinical trials are research studies that test how well new medical approaches work in people (see Question 1). Every clinical trial has a protocol, which
Clinical Trials: Questions and Answers Key Points Clinical trials are research studies that test how well new medical approaches work in people (see Question 1). Every clinical trial has a protocol, which
LJMU Research Data Policy: information and guidance
 LJMU Research Data Policy: information and guidance Prof. Director of Research April 2013 Aims This document outlines the University policy and provides advice on the treatment, storage and sharing of
LJMU Research Data Policy: information and guidance Prof. Director of Research April 2013 Aims This document outlines the University policy and provides advice on the treatment, storage and sharing of
Electronic Health Record Strategies for Pay for Performance. James O Connor MD Director of Clinical Informatics
 Electronic Health Record Strategies for Pay for Performance This Title is Copy where the title would go. James O Connor MD Director of Clinical Informatics Early Days of EMR... Access to Records E&M Coding
Electronic Health Record Strategies for Pay for Performance This Title is Copy where the title would go. James O Connor MD Director of Clinical Informatics Early Days of EMR... Access to Records E&M Coding
Distributed Networking
 Distributed Networking Millions of people. Strong collaborations. Privacy first. Jeffrey Brown, Lesley Curtis, Richard Platt Harvard Pilgrim Health Care Institute and Harvard Medical School Duke Medical
Distributed Networking Millions of people. Strong collaborations. Privacy first. Jeffrey Brown, Lesley Curtis, Richard Platt Harvard Pilgrim Health Care Institute and Harvard Medical School Duke Medical
TRACKS GENETIC EPIDEMIOLOGY
 Dr. Priya Duggal, Director In the post-genomic era where larger amounts of genetic data are now readily available, it has become increasingly important to design studies and use analytical techniques that
Dr. Priya Duggal, Director In the post-genomic era where larger amounts of genetic data are now readily available, it has become increasingly important to design studies and use analytical techniques that
GE Healthcare. Centricity PACS and PACS-IW with Universal Viewer* Where it all comes together
 GE Healthcare Centricity PACS and PACS-IW with Universal Viewer* Where it all comes together The healthcare industry is going through an unprecedented period of change with providers being called upon
GE Healthcare Centricity PACS and PACS-IW with Universal Viewer* Where it all comes together The healthcare industry is going through an unprecedented period of change with providers being called upon
The Landscape of Clinical Trial Data Sharing Initiatives
 2 T R I A L D ES I G N & R EG I S TRAT I O N P A R T I C I P ANT EN R O L L M EN T M E T A D ATA R EG I STRAT I O N EL EM EN TS DATA S H A R I N G P L A N I N D I V I D U A L P A R T I C I PA NT D A T
2 T R I A L D ES I G N & R EG I S TRAT I O N P A R T I C I P ANT EN R O L L M EN T M E T A D ATA R EG I STRAT I O N EL EM EN TS DATA S H A R I N G P L A N I N D I V I D U A L P A R T I C I PA NT D A T
Using Big Data to Advance Healthcare Gregory J. Moore MD, PhD February 4, 2014
 Using Big Data to Advance Healthcare Gregory J. Moore MD, PhD February 4, 2014 Sequencing Technology - Hype Cycle (Gartner) Gartner - Hype Cycle for Healthcare Provider Applications, Analytics and Systems,
Using Big Data to Advance Healthcare Gregory J. Moore MD, PhD February 4, 2014 Sequencing Technology - Hype Cycle (Gartner) Gartner - Hype Cycle for Healthcare Provider Applications, Analytics and Systems,
3. Provide the capacity to analyse and report on priority business questions within the scope of the master datasets;
 Business Intelligence Policy Version Information A. Introduction Purpose Business Intelligence refers to the practice of connecting facts, objects, people and processes of interest to an organisation in
Business Intelligence Policy Version Information A. Introduction Purpose Business Intelligence refers to the practice of connecting facts, objects, people and processes of interest to an organisation in
CHRONIC KIDNEY DISEASE MANAGEMENT GUIDE
 CHRONIC KIDNEY DISEASE MANAGEMENT GUIDE Outline I. Introduction II. Identifying Members with Kidney Disease III. Clinical Guidelines for Kidney Disease A. Chronic Kidney Disease B. End Stage Renal Disease
CHRONIC KIDNEY DISEASE MANAGEMENT GUIDE Outline I. Introduction II. Identifying Members with Kidney Disease III. Clinical Guidelines for Kidney Disease A. Chronic Kidney Disease B. End Stage Renal Disease
Latest advice for medicines users The monthly newsletter from the MHRA and its independent advisor the Commission on Human Medicines
 Latest advice for medicines users The monthly newsletter from the MHRA and its independent advisor the Commission on Human Medicines Volume 6, Issue 10, May 2013 Drug safety advice Yellow card scheme Stop
Latest advice for medicines users The monthly newsletter from the MHRA and its independent advisor the Commission on Human Medicines Volume 6, Issue 10, May 2013 Drug safety advice Yellow card scheme Stop
Chronic Kidney Disease (CKD) Program
 Chronic Kidney Disease (CKD) Program Graphic courtesy of the Kidney Foundation of Canada Chronic Kidney Disease Program Kidney Health Centre 235 Albert Street North Regina, Saskatchewan, S4R 3C2 Ph: (306)
Chronic Kidney Disease (CKD) Program Graphic courtesy of the Kidney Foundation of Canada Chronic Kidney Disease Program Kidney Health Centre 235 Albert Street North Regina, Saskatchewan, S4R 3C2 Ph: (306)
Xerox Mobile Link App Benefits Examples
 App Benefits Examples Contents Case 1: Healthcare Forms ing Case 2: Auto Lease Case 3: Sales Contract ing Case 4: Enrollment and Registrations Case 5: Insurance Claims ing Case 6: Editorial Markups/ Collaboration
App Benefits Examples Contents Case 1: Healthcare Forms ing Case 2: Auto Lease Case 3: Sales Contract ing Case 4: Enrollment and Registrations Case 5: Insurance Claims ing Case 6: Editorial Markups/ Collaboration
Innovation for Safety: Achievements and Challenges
 Innovation for Safety: Achievements and Challenges Nicole Denjoy DITTA Chair COCIR Secretary General IMDRF Open Stakeholders Forum Brussels, Belgium November 13, 2013 DITTA - the Global Diagnostic Imaging,
Innovation for Safety: Achievements and Challenges Nicole Denjoy DITTA Chair COCIR Secretary General IMDRF Open Stakeholders Forum Brussels, Belgium November 13, 2013 DITTA - the Global Diagnostic Imaging,
Data Management and Analysis for Successful Clinical Research. Lily Wang, PhD Department of Biostatistics Vanderbilt University
 Data Management and Analysis for Successful Clinical Research Lily Wang, PhD Department of Biostatistics Vanderbilt University Goals of This Presentation Provide an overview on data management and analysis
Data Management and Analysis for Successful Clinical Research Lily Wang, PhD Department of Biostatistics Vanderbilt University Goals of This Presentation Provide an overview on data management and analysis
Research Skills for Non-Researchers: Using Electronic Health Data and Other Existing Data Resources
 Research Skills for Non-Researchers: Using Electronic Health Data and Other Existing Data Resources James Floyd, MD, MS Sep 17, 2015 UW Hospital Medicine Faculty Development Program Objectives Become more
Research Skills for Non-Researchers: Using Electronic Health Data and Other Existing Data Resources James Floyd, MD, MS Sep 17, 2015 UW Hospital Medicine Faculty Development Program Objectives Become more
Data Management Implementation Plan
 Appendix 8.H Data Management Implementation Plan Prepared by Vikram Vyas CRESP-Amchitka Data Management Component 1. INTRODUCTION... 2 1.1. OBJECTIVES AND SCOPE... 2 2. DATA REPORTING CONVENTIONS... 2
Appendix 8.H Data Management Implementation Plan Prepared by Vikram Vyas CRESP-Amchitka Data Management Component 1. INTRODUCTION... 2 1.1. OBJECTIVES AND SCOPE... 2 2. DATA REPORTING CONVENTIONS... 2
MASTERS IN PUBLIC HEALTH (M.A.)
 MASTERS IN PUBLIC HEALTH (M.A.) Department of Social Sciences Course Director : Prof. J.K. Das Coordinators : Prof. A.M. Khan Prof. T. Bir Co- coordinator : Dr. A.M. Elizabeth Course Associate : Mr. Bacchu
MASTERS IN PUBLIC HEALTH (M.A.) Department of Social Sciences Course Director : Prof. J.K. Das Coordinators : Prof. A.M. Khan Prof. T. Bir Co- coordinator : Dr. A.M. Elizabeth Course Associate : Mr. Bacchu
VICTORIAN CARDIAC OUTCOMES REGISTRY. Data Management Policy
 VICTORIAN CARDIAC OUTCOMES REGISTRY Data Management Policy Version 1.0 26 February 2014 Table of Contents 1. Document Version Control... 1 1. Preface... 2 2. Project Information... 2 2.1 Purpose of VCOR...
VICTORIAN CARDIAC OUTCOMES REGISTRY Data Management Policy Version 1.0 26 February 2014 Table of Contents 1. Document Version Control... 1 1. Preface... 2 2. Project Information... 2 2.1 Purpose of VCOR...
EDRN s Validation Study Information Management System
 EDRN s Validation Study Information Management System Developed for EDRN by the DMCC Jet Propulsion Laboratory California Institute of Technology Cancer Biomarkers Group Division of Cancer Prevention VSIMS
EDRN s Validation Study Information Management System Developed for EDRN by the DMCC Jet Propulsion Laboratory California Institute of Technology Cancer Biomarkers Group Division of Cancer Prevention VSIMS
How To Be An Fht Nurse Practitioner
 JOB DESCRIPTION Position Title: Program: Service: Accountable to: FHT NURSE PRACTITIONER Family Health Team Health Family Health Team Manager Issued By: Date Revised: June 18, 2007 Date Issued: Classification:
JOB DESCRIPTION Position Title: Program: Service: Accountable to: FHT NURSE PRACTITIONER Family Health Team Health Family Health Team Manager Issued By: Date Revised: June 18, 2007 Date Issued: Classification:
Glossary of Methodologic Terms
 Glossary of Methodologic Terms Before-After Trial: Investigation of therapeutic alternatives in which individuals of 1 period and under a single treatment are compared with individuals at a subsequent
Glossary of Methodologic Terms Before-After Trial: Investigation of therapeutic alternatives in which individuals of 1 period and under a single treatment are compared with individuals at a subsequent
A SERENDIPITOUS EVALUATION OF MANDATORY HEALTH INSURANCE IN MASSACHUSETTS
 A SERENDIPITOUS EVALUATION OF MANDATORY HEALTH INSURANCE IN MASSACHUSETTS New England Research Institutes, (NERI) Institute for Health Services and Disparities Research E: jmckinlay@neriscience.com 1.
A SERENDIPITOUS EVALUATION OF MANDATORY HEALTH INSURANCE IN MASSACHUSETTS New England Research Institutes, (NERI) Institute for Health Services and Disparities Research E: jmckinlay@neriscience.com 1.
Corporate Medical Policy Genetic Testing for Fanconi Anemia
 Corporate Medical Policy Genetic Testing for Fanconi Anemia File Name: Origination: Last CAP Review: Next CAP Review: Last Review: genetic_testing_for_fanconi_anemia 03/2015 3/2016 3/2017 3/2016 Description
Corporate Medical Policy Genetic Testing for Fanconi Anemia File Name: Origination: Last CAP Review: Next CAP Review: Last Review: genetic_testing_for_fanconi_anemia 03/2015 3/2016 3/2017 3/2016 Description
Active Clinical Trials
 Active Clinical Trials 1. Genentech Efalizumab Protocol No. ACD4230g Title: A Phase II/III, Randomized, Open-Label, Active-Controlled, Multicenter Trial to Evaluate the Safety and Efficacy of Efalizumab
Active Clinical Trials 1. Genentech Efalizumab Protocol No. ACD4230g Title: A Phase II/III, Randomized, Open-Label, Active-Controlled, Multicenter Trial to Evaluate the Safety and Efficacy of Efalizumab
An Introduction to Genomics and SAS Scientific Discovery Solutions
 An Introduction to Genomics and SAS Scientific Discovery Solutions Dr Karen M Miller Product Manager Bioinformatics SAS EMEA 16.06.03 Copyright 2003, SAS Institute Inc. All rights reserved. 1 Overview!
An Introduction to Genomics and SAS Scientific Discovery Solutions Dr Karen M Miller Product Manager Bioinformatics SAS EMEA 16.06.03 Copyright 2003, SAS Institute Inc. All rights reserved. 1 Overview!
Medical Billing & Coding Catalog Course Description
 Medical Billing & Coding Catalog Course Description Effective 6/10/2011 Medical Billing & Coding Course Description Educational Objectives: American School of Technology s Medical Billing and Coding program
Medical Billing & Coding Catalog Course Description Effective 6/10/2011 Medical Billing & Coding Course Description Educational Objectives: American School of Technology s Medical Billing and Coding program
Professional Certificate in Primary Care Psychology
 Professional Certificate in Primary Care Psychology The NAPPP Primary Care Psychology Certificate training program requires the completion of 10 courses. Each course awards 15 CE credit hours. NAPPP is
Professional Certificate in Primary Care Psychology The NAPPP Primary Care Psychology Certificate training program requires the completion of 10 courses. Each course awards 15 CE credit hours. NAPPP is
CLINICAL DATA MONITORING PLAN (CDMoP) PROTOCOL # [0000] [TITLE]
![CLINICAL DATA MONITORING PLAN (CDMoP) PROTOCOL # [0000] [TITLE] CLINICAL DATA MONITORING PLAN (CDMoP) PROTOCOL # [0000] [TITLE]](/thumbs/39/18344673.jpg) CLINICAL DATA MONITORING PLAN (CDMoP) PROTOCOL # [0000] [TITLE] CONTRACT RESEARCH ORGANIZATION SPONSOR [NAME] [ADDRESS] 1 TABLE OF CONTENTS 1. Purpose 3 2. References 3 3. Study Roles and Responsibilities
CLINICAL DATA MONITORING PLAN (CDMoP) PROTOCOL # [0000] [TITLE] CONTRACT RESEARCH ORGANIZATION SPONSOR [NAME] [ADDRESS] 1 TABLE OF CONTENTS 1. Purpose 3 2. References 3 3. Study Roles and Responsibilities
TAKING PART IN CANCER TREATMENT RESEARCH STUDIES
 For more infomation about Cancer Clinical Trials at Upstate Cancer Center please call Upstate Connect 1.800.464.8668 TAKING PART IN CANCER TREATMENT RESEARCH STUDIES Information provided by: National Cancer
For more infomation about Cancer Clinical Trials at Upstate Cancer Center please call Upstate Connect 1.800.464.8668 TAKING PART IN CANCER TREATMENT RESEARCH STUDIES Information provided by: National Cancer
Unit Number Unit Name Person Responsible 0121302 Accounting Teheitha Murray. Outcome Number: Year: 2014 2015 Outcome Type: Program Operational Outcome
 Unit Number Unit Name Person Responsible 0121302 Accounting Teheitha Murray Number: Year: 2014 2015 Type: : Institutional Goal: Initial Assessment: Scheduled Completion Date: Budget Amount: Action Plan:
Unit Number Unit Name Person Responsible 0121302 Accounting Teheitha Murray Number: Year: 2014 2015 Type: : Institutional Goal: Initial Assessment: Scheduled Completion Date: Budget Amount: Action Plan:
Application for Research
 Application for Research All research conducted within the Brooks Rehabilitation system or any entity or facility bearing the Brooks name, including the Brooks Rehabilitation Clinical Research Center,
Application for Research All research conducted within the Brooks Rehabilitation system or any entity or facility bearing the Brooks name, including the Brooks Rehabilitation Clinical Research Center,
Big Data Analytics Predicting Risk of Readmissions of Diabetic Patients
 Big Data Analytics Predicting Risk of Readmissions of Diabetic Patients Saumya Salian 1, Dr. G. Harisekaran 2 1 SRM University, Department of Information and Technology, SRM Nagar, Chennai 603203, India
Big Data Analytics Predicting Risk of Readmissions of Diabetic Patients Saumya Salian 1, Dr. G. Harisekaran 2 1 SRM University, Department of Information and Technology, SRM Nagar, Chennai 603203, India
Renal Cysts What should I do now?
 Renal Cysts What should I do now? Dr Edmund Chiong Asst. Professor & Consultant Department of Urology National University Hospital What are renal cysts? Fluid-filled structures in the kidney that are not
Renal Cysts What should I do now? Dr Edmund Chiong Asst. Professor & Consultant Department of Urology National University Hospital What are renal cysts? Fluid-filled structures in the kidney that are not
Introduction to openehr Archetypes & Templates. Dr Ian McNicoll Dr Heather Leslie
 Introduction to openehr Archetypes & Templates Dr Ian McNicoll Dr Heather Leslie Traditional Application Development Clinical Knowledge Data Model Ocean Informatics 2010 Tradi&onal Informa&on model 2 level
Introduction to openehr Archetypes & Templates Dr Ian McNicoll Dr Heather Leslie Traditional Application Development Clinical Knowledge Data Model Ocean Informatics 2010 Tradi&onal Informa&on model 2 level
Best Practices in Opioid Dependence Treatment
 Best Practices in Opioid Dependence Treatment Anthony L. Jordan Health Center Linda Clark, MD, MS Medical Director Alana Ramos, BS Suboxone Clinic Manager Case Studies Nicole White female 27 years of age
Best Practices in Opioid Dependence Treatment Anthony L. Jordan Health Center Linda Clark, MD, MS Medical Director Alana Ramos, BS Suboxone Clinic Manager Case Studies Nicole White female 27 years of age
Clinical Trials and YOU
 Clinical Trials and YOU What are clinical trials? Controlled studies to determine how a particular anti-cancer strategy ( i.e. treatment ) affects the people who receive it Involve rigorous scientific
Clinical Trials and YOU What are clinical trials? Controlled studies to determine how a particular anti-cancer strategy ( i.e. treatment ) affects the people who receive it Involve rigorous scientific
European Statistical Workshop (EFSPI/PSI): EMA Clinical Trial Data Transparency
 European Statistical Workshop (EFSPI/PSI): EMA Clinical Trial Data Transparency Dr. Christoph Gerlinger 2013-08-22 Overview How it all began Current status EFSPI s Position EMA s draft Policy Aim of this
European Statistical Workshop (EFSPI/PSI): EMA Clinical Trial Data Transparency Dr. Christoph Gerlinger 2013-08-22 Overview How it all began Current status EFSPI s Position EMA s draft Policy Aim of this
Section 1 Project Management, Project Communication/Process Design, Mgmt, Documentation, Definition & Scope /CRO-Sponsor Partnership
 Section 1 Project Management, Project Communication/Process Design, Mgmt, Documentation, Definition & Scope /CRO-Sponsor Partnership PROJECT MANAGEMENT - SCOPE DEFINITION AND MANAGEMENT Understands the
Section 1 Project Management, Project Communication/Process Design, Mgmt, Documentation, Definition & Scope /CRO-Sponsor Partnership PROJECT MANAGEMENT - SCOPE DEFINITION AND MANAGEMENT Understands the
Importance of Statistics in creating high dimensional data
 Importance of Statistics in creating high dimensional data Hemant K. Tiwari, PhD Section on Statistical Genetics Department of Biostatistics University of Alabama at Birmingham History of Genomic Data
Importance of Statistics in creating high dimensional data Hemant K. Tiwari, PhD Section on Statistical Genetics Department of Biostatistics University of Alabama at Birmingham History of Genomic Data
Twente Grants Week: Data management. Maarten van Bentum (Library & Archive)
 Twente Grants Week: Data management Maarten van Bentum (Library & Archive) Overview 1. Questions to be answered (storage, description and sharing/archiving) 2. Scientific integrity 3. Why data management
Twente Grants Week: Data management Maarten van Bentum (Library & Archive) Overview 1. Questions to be answered (storage, description and sharing/archiving) 2. Scientific integrity 3. Why data management
Exploring the Challenges and Opportunities of Leveraging EMRs for Data-Driven Clinical Research
 White Paper Exploring the Challenges and Opportunities of Leveraging EMRs for Data-Driven Clinical Research Because some knowledge is too important not to share. Exploring the Challenges and Opportunities
White Paper Exploring the Challenges and Opportunities of Leveraging EMRs for Data-Driven Clinical Research Because some knowledge is too important not to share. Exploring the Challenges and Opportunities
Documentation and Medical Records Paper-based and Computer-based
 Documentation and Medical Records Paper-based and Computer-based Bengt Göransson bengt.goransson@it.uu.se Medical Informatics 1MD012, Fall 2013 Division of Visual Information and Interaction, HCI Brainstorming
Documentation and Medical Records Paper-based and Computer-based Bengt Göransson bengt.goransson@it.uu.se Medical Informatics 1MD012, Fall 2013 Division of Visual Information and Interaction, HCI Brainstorming
GE Healthcare. Centricity * PACS with Universal Viewer. Universal Viewer. Where it all comes together.
 GE Healthcare Centricity * PACS with Universal Viewer Universal Viewer. Where it all comes together. Where it all comes together Centricity PACS with Universal Viewer introduces an intuitive imaging application
GE Healthcare Centricity * PACS with Universal Viewer Universal Viewer. Where it all comes together. Where it all comes together Centricity PACS with Universal Viewer introduces an intuitive imaging application
Lessons on the Metadata Approach. Dave Iberson- Hurst 9 th April 2014 CDISC Euro Interchange 2014
 1 Lessons on the Metadata Approach Dave Iberson- Hurst 9 th April 2014 CDISC Euro Interchange 2014 2 Experience Human beings, who are almost unique in having the ability to learn from the experience of
1 Lessons on the Metadata Approach Dave Iberson- Hurst 9 th April 2014 CDISC Euro Interchange 2014 2 Experience Human beings, who are almost unique in having the ability to learn from the experience of
Renal Cell Carcinoma (Event Driven)
 Brochure More information from http://www.researchandmarkets.com/reports/2365661/ Renal Cell Carcinoma (Event Driven) Description: The Renal Cell Carcinoma market is highly lucrative. We anticipate that
Brochure More information from http://www.researchandmarkets.com/reports/2365661/ Renal Cell Carcinoma (Event Driven) Description: The Renal Cell Carcinoma market is highly lucrative. We anticipate that
Release of Data from EORTC Studies for Use in External Research Projects
 Release of Data from Studies for Use in External Research Projects POL008 Version 2.03 ALWAYS REFER TO THE INTERNET WEBSITE TO CHECK THE VALIDITY OF THIS DOCUMENT Author: Associate Head of Statistics Department
Release of Data from Studies for Use in External Research Projects POL008 Version 2.03 ALWAYS REFER TO THE INTERNET WEBSITE TO CHECK THE VALIDITY OF THIS DOCUMENT Author: Associate Head of Statistics Department
Newborn Screening Interoperability Specification
 Newborn Screening Interoperability Specification Alan E Zuckerman MD Department of Pediatrics, Georgetown University National Library of Medicine, Lister Hill Center Washington DC aez@georgetown.edu 1
Newborn Screening Interoperability Specification Alan E Zuckerman MD Department of Pediatrics, Georgetown University National Library of Medicine, Lister Hill Center Washington DC aez@georgetown.edu 1
Management and Treatment Guidelines for Cornelia de Lange Syndrome
 Management and Treatment Guidelines for Cornelia de Lange Syndrome These cards highlight routine care for people with CdLS needed by specific age groups, including infancy, early and late childhood, adolescence
Management and Treatment Guidelines for Cornelia de Lange Syndrome These cards highlight routine care for people with CdLS needed by specific age groups, including infancy, early and late childhood, adolescence
Big Data and Data Analysis for Personalized Medicine
 Big Data and Data Analysis for Personalized Medicine Dr. Paul Terry Ambassador Agenda Information and Data The Technology The Promise Personalized Medicine 2 CEO/CTO of PHEMI Board of Life Sciences BC
Big Data and Data Analysis for Personalized Medicine Dr. Paul Terry Ambassador Agenda Information and Data The Technology The Promise Personalized Medicine 2 CEO/CTO of PHEMI Board of Life Sciences BC
THE INTRICATE ORGANIZATION AND MANAGEMENT OF CLINICAL RESEARCH LABORATORY SAMPLES USING SAS/AF"
 THE INTRICATE ORGANIZATION AND MANAGEMENT OF CLINICAL RESEARCH LABORATORY SAMPLES USING SAS/AF" Jacqueline A. Wendel, M.S., University of Rochester Daniel A. Nosek, University of Rochester ABSTRACT: The
THE INTRICATE ORGANIZATION AND MANAGEMENT OF CLINICAL RESEARCH LABORATORY SAMPLES USING SAS/AF" Jacqueline A. Wendel, M.S., University of Rochester Daniel A. Nosek, University of Rochester ABSTRACT: The
INTERACTIVE MANIPULATION, VISUALIZATION AND ANALYSIS OF LARGE SETS OF MULTIDIMENSIONAL TIME SERIES IN HEALTH INFORMATICS
 Proceedings of the 3 rd INFORMS Workshop on Data Mining and Health Informatics (DM-HI 2008) J. Li, D. Aleman, R. Sikora, eds. INTERACTIVE MANIPULATION, VISUALIZATION AND ANALYSIS OF LARGE SETS OF MULTIDIMENSIONAL
Proceedings of the 3 rd INFORMS Workshop on Data Mining and Health Informatics (DM-HI 2008) J. Li, D. Aleman, R. Sikora, eds. INTERACTIVE MANIPULATION, VISUALIZATION AND ANALYSIS OF LARGE SETS OF MULTIDIMENSIONAL
Assessment of Uranium, Manganese, Fluoride, Nitrate, and Nitrite in Domestic Well Water in Maine, 2005-2009. Michael Foley and Robert Marvinney
 Assessment of Uranium, Manganese, Fluoride, Nitrate, and Nitrite in Domestic Well Water in Maine, 2005-2009 Michael Foley and Robert Marvinney 2011 Northeast Private Well Water Symposium November 14 &
Assessment of Uranium, Manganese, Fluoride, Nitrate, and Nitrite in Domestic Well Water in Maine, 2005-2009 Michael Foley and Robert Marvinney 2011 Northeast Private Well Water Symposium November 14 &
Clinical Trials and Screening: What You Need to Know
 Scan for mobile link. Clinical Trials and Screening: What You Need to Know What is a Clinical Trial? At A Glance A clinical trial is a research study that tests how well new medical techniques work in
Scan for mobile link. Clinical Trials and Screening: What You Need to Know What is a Clinical Trial? At A Glance A clinical trial is a research study that tests how well new medical techniques work in
Pharmaceutical Applications
 Integrated Summary of Safety and Efficacy Programming for Studies Using Electronic Data Capture Changhong Shi, Merck & Co., Inc., Rahway, NJ Qing Xue, Merck & Co., Inc., Rahway, NJ ABSTRACT The Integrated
Integrated Summary of Safety and Efficacy Programming for Studies Using Electronic Data Capture Changhong Shi, Merck & Co., Inc., Rahway, NJ Qing Xue, Merck & Co., Inc., Rahway, NJ ABSTRACT The Integrated
Understanding CDISC Basics
 Trends in Bio/Pharmaceutical Industry Understanding CDISC Basics Jane Ma Abstract Data standards can make data and its associated program more portable. The CDISC (Clinical Data Interchange Standards Consortium)
Trends in Bio/Pharmaceutical Industry Understanding CDISC Basics Jane Ma Abstract Data standards can make data and its associated program more portable. The CDISC (Clinical Data Interchange Standards Consortium)
SJÖGREN S INTERNATIONAL COLLABORATIVE CLINICAL ALLIANCE (SICCA) BIOREPOSITORY AND DATA REGISTRY DATA MANAGEMENT PLAN
 1 SJÖGREN S INTERNATIONAL COLLABORATIVE CLINICAL ALLIANCE (SICCA) BIOREPOSITORY AND DATA REGISTRY DATA MANAGEMENT PLAN 1. Database Overview The SICCA database includes the Clinical Database, the Biorepository
1 SJÖGREN S INTERNATIONAL COLLABORATIVE CLINICAL ALLIANCE (SICCA) BIOREPOSITORY AND DATA REGISTRY DATA MANAGEMENT PLAN 1. Database Overview The SICCA database includes the Clinical Database, the Biorepository
Building the Foundation for Clinical Research Nursing
 Building the Foundation for Clinical Research Nursing A CLINICAL RESEARCH NURSING MODEL OF CARE Updated: 7/6/2011 If you wish to quote information from this document, please use the following citation
Building the Foundation for Clinical Research Nursing A CLINICAL RESEARCH NURSING MODEL OF CARE Updated: 7/6/2011 If you wish to quote information from this document, please use the following citation
Tuberculosis (TB) Screening Guidelines for Substance Use Disorder Treatment Programs in California
 Tuberculosis (TB) Screening Guidelines for Substance Use Disorder Treatment Programs in California 1 of 7 Table of Contents Preface 2 TB Symptoms and TB History 2 Initial Screening 2 Follow-Up Screening
Tuberculosis (TB) Screening Guidelines for Substance Use Disorder Treatment Programs in California 1 of 7 Table of Contents Preface 2 TB Symptoms and TB History 2 Initial Screening 2 Follow-Up Screening
Science Highlights. To PSA or not to PSA: That is the Question.
 Science Highlights June 2012 by Ann A. Kiessling, PhD at the To PSA or not to PSA: That is the Question. The current raucous debate over the commonly used PSA blood test to screen for prostate cancer,
Science Highlights June 2012 by Ann A. Kiessling, PhD at the To PSA or not to PSA: That is the Question. The current raucous debate over the commonly used PSA blood test to screen for prostate cancer,
QsarDB first 100 DOIs for predictive models
 QsarDB first 100 DOIs for predictive models Uko Maran Institute of chemistry, University of Tartu, Estonia LOD: Content Data Predictive (and descriptive) models? Goal Components Persistent digital identifiers
QsarDB first 100 DOIs for predictive models Uko Maran Institute of chemistry, University of Tartu, Estonia LOD: Content Data Predictive (and descriptive) models? Goal Components Persistent digital identifiers
Implementing the ImPACT Program
 Implementing the ImPACT Program Doug Tauchen Director, Customer Support ImPACT Applications Headquarter: Pittsburgh, PA Subsidiary: Iowa City, IA Phone: (877) 646-7991 Ext. 3 doug@impacttest.com Copyright
Implementing the ImPACT Program Doug Tauchen Director, Customer Support ImPACT Applications Headquarter: Pittsburgh, PA Subsidiary: Iowa City, IA Phone: (877) 646-7991 Ext. 3 doug@impacttest.com Copyright
Delivering the power of the world s most successful genomics platform
 Delivering the power of the world s most successful genomics platform NextCODE Health is bringing the full power of the world s largest and most successful genomics platform to everyday clinical care NextCODE
Delivering the power of the world s most successful genomics platform NextCODE Health is bringing the full power of the world s largest and most successful genomics platform to everyday clinical care NextCODE
A grant number provides unique identification for the grant.
 Data Management Plan template Name of student/researcher(s) Name of group/project Description of your research Briefly summarise the type of your research to help others understand the purposes for which
Data Management Plan template Name of student/researcher(s) Name of group/project Description of your research Briefly summarise the type of your research to help others understand the purposes for which
Version 12. Module 6: Medicare for People with End-Stage Renal Disease - Alaska
 Version 12 Module 6: Medicare for People with End-Stage Renal Disease - Alaska Session Objectives This session will help you understand Medicare for people with End-stage Renal Disease Medicare eligibility
Version 12 Module 6: Medicare for People with End-Stage Renal Disease - Alaska Session Objectives This session will help you understand Medicare for people with End-stage Renal Disease Medicare eligibility
Preimplantation Genetic Diagnosis. Evaluation for single gene disorders
 Preimplantation Genetic Diagnosis Evaluation for single gene disorders What is Preimplantation Genetic Diagnosis? Preimplantation genetic diagnosis or PGD is a technology that allows genetic testing of
Preimplantation Genetic Diagnosis Evaluation for single gene disorders What is Preimplantation Genetic Diagnosis? Preimplantation genetic diagnosis or PGD is a technology that allows genetic testing of
UK Data Archive Data Dictionary
 UK Data Archive Study Number 7182 - Understanding Society: Waves 1-5, 2009-2014: Special Licence Access, School Codes UK Data Archive Data Dictionary a_schcode_protect Number of cases = 7570 Pos. = 2 Variable
UK Data Archive Study Number 7182 - Understanding Society: Waves 1-5, 2009-2014: Special Licence Access, School Codes UK Data Archive Data Dictionary a_schcode_protect Number of cases = 7570 Pos. = 2 Variable
General Services Administration Federal Supply Service Authorized Federal Supply Schedule Price List
 General Services Administration Federal Supply Service Authorized Federal Supply Schedule Price List GSA Schedule 66 Scientific Equipment and Services SIN 66-1000 Professional Scientific Services IHRC,
General Services Administration Federal Supply Service Authorized Federal Supply Schedule Price List GSA Schedule 66 Scientific Equipment and Services SIN 66-1000 Professional Scientific Services IHRC,
Guidance for the format and content of the protocol of non-interventional post-authorisation safety studies
 26 September 2012 EMA/623947/2012 Patient Health Protection Guidance for the format and content of the protocol of non-interventional post-authorisation Introduction From 10 January 2013, marketing authorisation
26 September 2012 EMA/623947/2012 Patient Health Protection Guidance for the format and content of the protocol of non-interventional post-authorisation Introduction From 10 January 2013, marketing authorisation
The Cell Therapy Catapult UK Clinical Trials Database
 May 2015 The UK Clinical Trials Database covers cell therapy clinical trial activity that the Cell Therapy Catapult believes to be ongoing in the UK as of May 2015. It supersedes the database of April
May 2015 The UK Clinical Trials Database covers cell therapy clinical trial activity that the Cell Therapy Catapult believes to be ongoing in the UK as of May 2015. It supersedes the database of April
case management controlled
 The effecte ffects s of a nurse-led case management programme on patients undergoing peritoneal dialysis: a randomized controlled trial Susan Chow RN, PhD The HK Polytechnic University Frances Wong RN,
The effecte ffects s of a nurse-led case management programme on patients undergoing peritoneal dialysis: a randomized controlled trial Susan Chow RN, PhD The HK Polytechnic University Frances Wong RN,
