PHAR 7633 Chapter 3 Biopharmaceutics and Pharmacokinetic Introduction
|
|
|
- Barry McCarthy
- 7 years ago
- Views:
Transcription
1 Student Objectives for this Chapter After completing the material in this chapter each student should:- have an basic understanding of the scope and impact of biopharmaceutics and pharmacokinetics have a general knowledge of the factors affecting the rate and extent of drug absorption, distribution, metabolism and excretion This page was last modified: Tuesday 25 May 2010 at 09:01 PM Page 1 of 30
2 An Introduction to Biopharmaceutics and Pharmacokinetics A Family Circle cartoon show a young child given an im injection who might ask "How will that 'ouch' get from there to my sore throat"? The answer to this question is the basis of biopharmaceutics and pharmacokinetics. That is, how drugs move around the body and how quickly this movement occurs. During this course many of the processes which control the absorption, distribution, metabolism, and excretion of drugs will be discussed, qualitatively (biopharmaceutics) and quantitatively (pharmacokinetics). This page was last modified: Tuesday 25 May 2010 at 09:01 PM Page 2 of 30
3 Complex Interactions Complex picture of drug interactions in the body. This slide gives an idea of the complexity of drug disposition. Shown in this slide are many of the steps to getting drug from one site in the body to another. Many of these processes are enzyme induced. However the overall picture often is much simpler. Many of these processes maybe fast or not significant for any given drug. Creasey, W.A Drug Disposition in Humans, The Basis of Clinical Pharmacology, Oxford University Press, New York p89 This page ( was last modified: Tuesday 25 May 2010 at 09:01 PM Page 3 of 30
4 Drug Transport Fig Slide of processes involved in drug transport. Here the major processes are represented in a less physiological fashion. It is with this approach that we will look at much of the course material this semester. The headings Absorption, Distribution, Metabolism, and Excretion are important. This page ( was last modified: Tuesday 25 May 2010 at 09:01 PM Page 4 of 30
5 One, Two and Three Compartment Models Fig Slide of one, two, and three compartment pharmacokinetic models. Fortunately many of the processes involved in drug movement around the body are not saturated at normal therapeutic dose levels. The pharmacokinetic - mathematical models that can be used to describe plasma concentration as a function of time can then be much simplified. The body may even be represented as a single compartment or container for some drugs. For other drugs a two or three compartment model is found to be necessary. This page ( was last modified: Tuesday 25 May 2010 at 09:01 PM Page 5 of 30
6 IV Bolus Dose - One Compartment redrawn from Niazi, 1979 Fig Slide of body before and after a rapid IV bolus injection, considering the body to behave as a single compartment. In order to simplify the mathematics it is often possible to assume that a drug given by rapid intravenous injection, a bolus, is rapidly mixed. This slide represents the uniformly mixed drug very shortly after administration. Niazi, S Textbook of Biopharmaceutics and Clinical Pharmacokinetics, Appleton-Century-Crofts, New York, p142 This page ( was last modified: Tuesday 25 May 2010 at 09:01 PM Page 6 of 30
7 Absorption and Elimination Fig Slide of oral curve and beakers. We can picture oral administration as water flowing from one bucket (representing the GI tract) into a second beaker (representing the body). At first drug flows into the 'body' beaker and the level rises, as drug concentration rises, then after peaking the levels start to fall as elimination overtakes absorption. This can be represented as an experiment in lab where the body is represented as a beaker. In the lab experiment a dye concentration represents drug concentration in the body. This page ( was last modified: Tuesday 25 May 2010 at 09:01 PM Page 7 of 30
8 IV Bolus Two Compartment Model redrawn from Niazi, 1979 Fig Slide of an intravenous bolus injection with a two compartment model. Often a one compartment model is not sufficient to represent the pharmacokinetics of a drug. A two compartment model often has wider application. Here we consider the body is a central compartment with rapid mixing and a peripheral compartment with slower distribution. The central compartment is uniformly mixed very shortly after drug administration, whereas it takes some time for the peripheral compartment to reach a pseudo equilibrium. Niazi, S Textbook of Biopharmaceutics and Clinical Pharmacokinetics, Appleton-Century-Crofts, New York, p175 This page ( was last modified: Tuesday 25 May 2010 at 09:01 PM Page 8 of 30
9 Two Compartment Model Fig A diagram of a two compartment model showing the parameters to be measured. The processes of distribution and excretion can be represented by the rate constants k12, k21, and k10. The rate constant k0 representing an infusion or absorption process. The drug appears to be dissolved in the body volume. This volume is the 'apparent' volume of distribution. This page ( was last modified: Tuesday 25 May 2010 at 09:01 PM Page 9 of 30
10 Drug Effect versus Time redrawn from Barr, 1968 Fig Slide showing effect as a function of time. From a graph such as this we can see the relationship between drug concentration and drug effect. If a drug has to reach an effective concentration at a receptor site this will be reflected as a required blood concentration. Barr, W.H Principles of biopharmaceutics, Amer. J. Pharm. Ed., 32, 958 This page ( was last modified: Tuesday 25 May 2010 at 09:01 PM Page 10 of 30
11 Drug Product Performance Parameters redrawn from Dittert and DiSanto, 1974 Fig Slide of biopharmaceutic parameters over a blood time curve. This slide shows some of the biopharmaceutic parameters which can be used to measure drug product performance. Later in the semester we will use the trapezoidal method of calculating AUC. Dittert, L.W. and DiSanto, A.R The bioavailability of drug products, J. Amer. Pharm. Assoc., NS13, This page ( was last modified: Tuesday 25 May 2010 at 09:01 PM Page 11 of 30
12 A(bsorption) D(istribution) M(etabolism) E(xcretion) Fig Slide listing the four pharmacokinetic processes. We will briefly consider each of these processes in turn. A(bsorption) D(istribution) M(etabolism) E(xcretion). This page ( was last modified: Tuesday 25 May 2010 at 09:01 PM Page 12 of 30
13 Routes of Drug Administration information from Benet, 1974 Fig Slide showing a model with the various routes of administration. Later we will discuss different routes of administration and see how these effect the kinetics of drug disposition. For example absorption through the lining of the mouth, buccal absorption, often results in rapid absorption without the drug passing through the liver where it can be broken down. However, only low dose drugs can be accommodated by this ROA. Orally absorbed drugs are absorbed in the stomach or intestines and enter the portal blood supply and go to the liver before getting into the central blood supply. This can result in extensive metabolism of the drug before it can take any action. For this reason some drugs are very inefficient when given orally. Benet, L.Z Input factors as determinants of drug activity: route, dose, dosage regimens and the drug delivery system, in Principles and Techniques of Human Research and Therapeutics, Vol 3, McMahon, F.G. ed., Futura, New York, pp 9-23 through Oie, S and Benet, L.Z. Chapter 5 The Effect of Route of Administration and Distribution on Drug Action in Modern Pharmaceutics, 4th ed., ed. Banker, G.S. and Rhodes, C.T. Marcel Dekker, New York, NY, Fig 2, p 121 This page ( was last modified: Tuesday 25 May 2010 at 09:01 PM Page 13 of 30
14 Tablet Dissolution redrawn from Cadwallader, 1983 Fig Slide showing the processes involved in the dissolution of a tablet before absorption. A drug cannot be absorbed across the intestinal wall as a solid. It must first dissolve in the fluid of the G-I tract. Tablets are carefully formulated, designed, to stay together in the bottle during transport but break up quickly once they are in an aqueous environment. This can be an easy or difficult job depending on the drug and the dose required. There are therefore dissolution tests specified to ensure that tablet formulations work. Also it may be appropriate to conduct experiments with human volunteers to ensure that the tablet does release the drug as it should. Cadwallader, D.E Biopharmaceutics and Drug Interactions, 3rd ed., Raven Press, New York, p72 This page ( was last modified: Tuesday 25 May 2010 at 09:01 PM Page 14 of 30
15 Absorption versus Particle Size redrawn from Prescott et al., 1970 Fig Slide showing plasma phenacetin versus time curve after oral administration of 1.5 g in various suspension dosage forms. The effect of particle size on drug absorption is shown in this slide. Generally speaking the smaller the particle size the quicker the absorption. By increasing the particle surface area it is possible to increase the dissolution rate. However very small particle can clump together. Therefore a wetting agent such as Tween 80 can have a beneficial effect on the overall absorption. The intestinal fluids usually contain some materials which can act as wetting agents, however drug dissolution testing in vitro may neglect this effect. Prescott, L.F., Steel, R.F. and Ferrier, W.R The effects of particle size on the absorption of phenacetin in man, Pharmacol. Therap., 11, This page ( was last modified: Tuesday 25 May 2010 at 09:01 PM Page 15 of 30
16 Absorption of Different Salt Forms redrawn from Juncher and Raaschou, 1957 Fig Slide showing plasma penicillin concentration versus time curve after oral administration of various salts. The rate at which a drug dissolves is dependent on the solubility of the drug. A common occurrence is that different salts will have quite different solubility characteristics, and again somewhat different to the free acid (or base) form. In the case of penicillin V it appears that only drug which is dissolved quickly can be absorbed. Juncher, H. and Raaschou, F The solubility of oral preparations of penicillin V, Antibiot. Med. Clin. Therap., 4, 497 This page ( was last modified: Tuesday 25 May 2010 at 09:01 PM Page 16 of 30
17 Drug Effect and Absorption redrawn from Wagner, 1961 Fig Slide showing blood sugar levels after tolbutamide acid and salt. Rapid dissolution and absorption is not always the objective. Sometimes a slower release is required. In the case of tolbutamide, used to lower blood sugar concentrations, a more sustained release is better causing a more gradual reduction in blood sugar. Wagner, J.G Biopharmaceutics: absorption aspects, J. Pharm. Sci., 50, 359 This page ( was last modified: Tuesday 25 May 2010 at 09:01 PM Page 17 of 30
18 Effect of Absorption Rate Constant Fig Slide showing plasma concentration time curves for three theoretical formulations with different ka values. The plasma concentrations achieved can be controlled by the rate of drug absorption. As shown product I would be toxic. A smaller dose, or a slower formulation should be considered. Formulation II appears to be effective in the dose and formulation used. The dosage form III appears to be too slow and thereby ineffective. If it is a drug requiring a sustained effect this formulation may be more useful at a higher dose or after repeated doses. This page ( was last modified: Tuesday 25 May 2010 at 09:01 PM Page 18 of 30
19 Disposition (DME: Distribution, Metabolism, Excretion) In a Family circle cartoon a young patient might asks "How does the medicine know where to go". Drug moves throughout the body directed by the physicochemical properties of the drug and the physiology of the body. Once the drug is absorbed what happens to it? The processes of excretion, metabolism, and distribution take over to move the drug through and out of the body. Drug is distributed throughout the body depending on the physicochemical properties of the drug and the physiological properties of the body membranes and fluids. The chemistry of the compound and the enzymes available determine the metabolism of the drug. The molecular size, polarity and binding influence the drug excretion. This page ( was last modified: Tuesday 25 May 2010 at 09:01 PM Page 19 of 30
20 ADME Fig Slide showing the four processes schematically. Let's consider elimination. This page ( was last modified: Tuesday 25 May 2010 at 09:01 PM Page 20 of 30
21 Drug Elimination Fig Slide listing methods of drug elimination, metabolism and excretion. The next topic is excretion. Drugs are elimination by a number of processes. Polar drugs are easily filtered from the blood in the kidneys and removed into urine. However non polar material is generally reabsorbed from the kidney or distributes into fat tissue. The liver however is capable of metabolizing many drugs, generally producing compounds which are more polar and more easily excreted. Enzymes in the liver are able to form conjugates with drugs or hydrolyze and oxidize drugs. Drug kinetics can be markedly altered by changes in metabolism or excretion of drugs. These processes together result in the elimination of a drug. This page ( was last modified: Tuesday 25 May 2010 at 09:01 PM Page 21 of 30
22 Elimination and Multiple Dose Fig Slide showing multiple dose curves with slow, medium, and fast excretion. Alteration of drug elimination is particularly important when drugs are given repeatedly. If it is assumed that the drug is eliminated normally when in fact it is slowly eliminated, drug accumulation may occur with toxic concentrations reached. Alternately faster elimination than expected may cause sub therapeutic concentrations to be reached. Dittert, L.W Pharmacokinetic Considerations in Clinical Drug Trials, Drug Intelligence and Clin. Pharmacy, 8, This page ( was last modified: Friday 02 Mar 2012 at 11:24 AM Page 22 of 30
23 Dosing Kanamycin Fig Slide showing a clinical example with kanamycin, showing theoretical curve after multiple dose and a better curve after dose adjustment. Kanamycin is a useful drug but it can cause some serious side effects. By controlling the blood concentration of this drug it is possible to use it effectively. In the case of patients with impaired renal, or kidney, function it is possible to determine the kidney function ahead of time and adjust the kanamycin dosing schedule accordingly. Dittert, L.W Pharmacokinetic Considerations in Clinical Drug Trials, Drug Intelligence and Clin. Pharmacy, 8, This page ( was last modified: Friday 02 Mar 2012 at 11:24 AM Page 23 of 30
24 Renal Disease and Drug Elimination Fig Slide showing relationship between kanamycin half-life and serum creatinine level. As kidney function is reduced the serum creatinine level increases and the also the kanamycin takes longer to be eliminated from the body. Cutler, R.E. and Orme, B.M Correlation of Serum Creatinine Concentration and Kanamycin Half-Life, J. Amer. Med. Assoc., 209 (4), This page ( was last modified: Friday 02 Mar 2012 at 11:43 AM Page 24 of 30
25 Absorption and First-Pass Effect redrawn from Barr, 1969 Figure Slide showing extra absorption of salicylamide at higher doses. The green dots represent results after a 1 g dose and the red dots represent results after a 2 g dose. The original figure presented results after 0.3 and 0.5 g doses with resulting concentrations below those determined after the 1 g dose. Normally the amount of drug present in the body is no more than the enzymes can handle easily. Metabolism then proceeds in apparent first order fashion. However for some drugs the enzymes can not keep up. Illustrated here is one example, saturation of first pass metabolism allowing more or a higher fraction of drug to be absorbed. Barr, W.H Factors involved in the assessment of systemic or biologic availability of drug products, Drug Inform. Bull., 3, This page was last modified: Tuesday 25 May 2010 at 09:01 PM Page 25 of 30
26 Aspirin Elimination redrawn from Levy, 1965 Figure Slide of aspirin plasma concentration as a function of time after 0.25, 1 or 2g. At higher doses the metabolism of aspirin is reduced by saturation. The elimination rate constant (as represented by the slope) is reduced. Levy, G J. Pharm. Sci., 54, p959 This page ( was last modified: Tuesday 25 May 2010 at 09:01 PM Page 26 of 30
27 Multiphasic Drug Elimination? redrawn from Smith et al., 1973 Figure Slide of pentobarbital plasma concentration versus time illustrating the multiphasic distribution pattern. From a plasma concentration time curve it may appear that this drug is rapidly eliminated from the body. Actually little of the drug is eliminated with much of it being distributed in various tissues of the body. Here distribution is a major factor affecting the overall kinetics of this drug. By looking further we can see the real picture. Smith, R.B., Dittert, L.W., Griffen Jr., W.O., and Doluisio, J.T Pharmacokinetics of Pentobarbital after Intravenous and Oral Administration, J. Pharmacokin. Biopharm., 1, p5 This page ( was last modified: Tuesday 25 May 2010 at 09:01 PM Page 27 of 30
28 Tissue Concentration versus Time redrawn from Bischoff and Dedrick, 1968 Figure Slide showing pentobarbital concentrations in various tissues. This illustrates the slow distribution of some drugs into and out of various tissues. Brain and other rapidly perfused tissues rapidly lose drug while other tissues are building up drug concentrations. Eventually significant amounts of drug will be collected in fat tissue from which it may only slowly be removed. The green line represents concentrations in adipose tissue (poorly perfused), the red line represents concentrations in viscera (highly perfused lean tissue), the orange line represents concentrations in lean tissue (poorly perfused) and the blue line represents concentrations in blood. Bischoff, K.B. and Dedrick, R.L Thiopental Pharmacokinetics, J. Pharm. Sci., 57, This page ( was last modified: Tuesday 25 May 2010 at 09:01 PM Page 28 of 30
29 Distribution Fig Slide showing other distribution mechanisms. Methods by which drug is kept in the body for extended action. ph, protein binding and fat storage are three major processes by which drug may be kept within the body. These are mechanisms by which drugs can be distributed throughout the body, protected from elimination, and cause a more prolonged activity. This page ( was last modified: Tuesday 25 May 2010 at 09:01 PM Page 29 of 30
30 Individualized Dosage Regimen "One for man. Two for a horse". There are many processes controlling drug disposition. Therefore dose adjustment is often necessary to produce the desired response from the patient. During this course we will look at the equations needed to calculate appropriate doses and the factors which can influence the plasma concentration of various drugs. In the next session we will start to look at the mathematics associated with a one compartment pharmacokinetic model... Student Objectives for this Chapter This page was last modified: Tuesday 25 May 2010 at 09:01 PM Page 30 of 30
PHAR 7633 Chapter 21 Non-Linear Pharmacokinetic Models
 Student Objectives for this Chapter PHAR 7633 Chapter 21 Non-Linear Pharmacokinetic Models To draw the scheme and write the differential equations for compartmental pharmacokinetic models with non-linear
Student Objectives for this Chapter PHAR 7633 Chapter 21 Non-Linear Pharmacokinetic Models To draw the scheme and write the differential equations for compartmental pharmacokinetic models with non-linear
Absorption of Drugs. Transport of a drug from the GI tract
 Absorption of Drugs Absorption is the transfer of a drug from its site of administration to the bloodstream. The rate and efficiency of absorption depend on the route of administration. For IV delivery,
Absorption of Drugs Absorption is the transfer of a drug from its site of administration to the bloodstream. The rate and efficiency of absorption depend on the route of administration. For IV delivery,
PHAR 7633 Chapter 19 Multi-Compartment Pharmacokinetic Models
 Student Objectives for this Chapter PHAR 7633 Chapter 19 Multi-Compartment Pharmacokinetic Models To draw the scheme and write the differential equations appropriate to a multi-compartment pharmacokinetic
Student Objectives for this Chapter PHAR 7633 Chapter 19 Multi-Compartment Pharmacokinetic Models To draw the scheme and write the differential equations appropriate to a multi-compartment pharmacokinetic
Nursing 113. Pharmacology Principles
 Nursing 113 Pharmacology Principles 1. The study of how drugs enter the body, reach the site of action, and are removed from the body is called a. pharmacotherapeutics b. pharmacology c. pharmacodynamics
Nursing 113 Pharmacology Principles 1. The study of how drugs enter the body, reach the site of action, and are removed from the body is called a. pharmacotherapeutics b. pharmacology c. pharmacodynamics
IV solutions may be given either as a bolus dose or infused slowly through a vein into the plasma at a constant or zero-order rate.
 د.شيماء Biopharmaceutics INTRAVENOUS INFUSION: IV solutions may be given either as a bolus dose or infused slowly through a vein into the plasma at a constant or zero-order rate. The main advantage for
د.شيماء Biopharmaceutics INTRAVENOUS INFUSION: IV solutions may be given either as a bolus dose or infused slowly through a vein into the plasma at a constant or zero-order rate. The main advantage for
Medication Utilization. Understanding Potential Medication Problems of the Elderly
 Medication Utilization Understanding Potential Medication Problems of the Elderly NICE - National Initiative for the Care of the Elderly WHAT ARE MEDICATION UTILIZATION PROBLEMS AMONG THE ELDERLY? A useful
Medication Utilization Understanding Potential Medication Problems of the Elderly NICE - National Initiative for the Care of the Elderly WHAT ARE MEDICATION UTILIZATION PROBLEMS AMONG THE ELDERLY? A useful
Drug Excretion. Renal Drug Clearance. Drug Clearance and Half-Life. Glomerular Filtration II. Glomerular Filtration I. Drug Excretion and Clearance
 t/.drugexcretion AINTRAVENOUSDOSE 36848765430TIME(hours) t/ Drug Excretion Dr. Robert G. Lamb Professor Pharmacology & Toxicology Drug Excretion and Clearance Drug Excretion: is the movement of drug from
t/.drugexcretion AINTRAVENOUSDOSE 36848765430TIME(hours) t/ Drug Excretion Dr. Robert G. Lamb Professor Pharmacology & Toxicology Drug Excretion and Clearance Drug Excretion: is the movement of drug from
Course Curriculum for Master Degree in Clinical Pharmacy
 Course Curriculum for Master Degree in Clinical Pharmacy The Master Degree in Clinical Pharmacy is awarded by the Faculty of Graduate studies at Jordan University of Science and Technology (JUST) upon
Course Curriculum for Master Degree in Clinical Pharmacy The Master Degree in Clinical Pharmacy is awarded by the Faculty of Graduate studies at Jordan University of Science and Technology (JUST) upon
QSAR. The following lecture has drawn many examples from the online lectures by H. Kubinyi
 QSAR The following lecture has drawn many examples from the online lectures by H. Kubinyi LMU Institut für Informatik, LFE Bioinformatik, Cheminformatics, Structure independent methods J. Apostolakis 1
QSAR The following lecture has drawn many examples from the online lectures by H. Kubinyi LMU Institut für Informatik, LFE Bioinformatik, Cheminformatics, Structure independent methods J. Apostolakis 1
Study (s) Degree Center Acad. Period. 1201 - Grado de Farmacia FACULTY OF PHARMACY 3 Annual 1211 - PDG Farmacia-Nutrición Humana y Dietética
 COURSE DATA Data Subject Código 34081 Name Biopharmacy and Pharmacokinetics Cycle Grade ECTS Credits 10.5 Curso académico 2014-2015 Study (s) Degree Center Acad. Period year 1201 - Grado de Farmacia FACULTY
COURSE DATA Data Subject Código 34081 Name Biopharmacy and Pharmacokinetics Cycle Grade ECTS Credits 10.5 Curso académico 2014-2015 Study (s) Degree Center Acad. Period year 1201 - Grado de Farmacia FACULTY
1: CLINICAL PHARMACOKINETICS
 : CLINICAL PHARMACOKINETICS General overview: clinical pharmacokinetics, 2 Pharmacokinetics, 4 Drug clearance (CL), 6 Volume of distribution (Vd), 8 The half-life (t½), 0 Oral availability (F), 2 Protein
: CLINICAL PHARMACOKINETICS General overview: clinical pharmacokinetics, 2 Pharmacokinetics, 4 Drug clearance (CL), 6 Volume of distribution (Vd), 8 The half-life (t½), 0 Oral availability (F), 2 Protein
Introduction to Enteris BioPharma
 Introduction to Enteris BioPharma Enteris BioPharma Intelligent Solutions for Oral Drug Delivery Privately held, New Jersey based biotech company Owned solely by Victory Park Capital, a large Chicago based
Introduction to Enteris BioPharma Enteris BioPharma Intelligent Solutions for Oral Drug Delivery Privately held, New Jersey based biotech company Owned solely by Victory Park Capital, a large Chicago based
A Peak at PK An Introduction to Pharmacokinetics
 Paper IS05 A Peak at PK An Introduction to Pharmacokinetics Hannah Twitchett, Roche Products Ltd, Welwyn Garden City, UK Paul Grimsey, Roche Products Ltd, Welwyn Garden City, UK ABSTRACT The aim of this
Paper IS05 A Peak at PK An Introduction to Pharmacokinetics Hannah Twitchett, Roche Products Ltd, Welwyn Garden City, UK Paul Grimsey, Roche Products Ltd, Welwyn Garden City, UK ABSTRACT The aim of this
Overview of Dissolution for BA/BE
 Biopharmaceutics Classification System based on Solubility/Permeability Biowaivers for BCS I Drugs Discussion of BCS III Drugs Models establishing in vivo-in vitro Correlations (IVIVC Levels A-C) 1 Biopharmaceutics
Biopharmaceutics Classification System based on Solubility/Permeability Biowaivers for BCS I Drugs Discussion of BCS III Drugs Models establishing in vivo-in vitro Correlations (IVIVC Levels A-C) 1 Biopharmaceutics
Guidance for Industry
 Guidance for Industry Food-Effect Bioavailability and Fed Bioequivalence Studies U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER)
Guidance for Industry Food-Effect Bioavailability and Fed Bioequivalence Studies U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER)
Session 3 Topics. Argatroban. Argatroban. Drug Use and Adverse Effects. Laboratory Monitoring of Anticoagulant Therapy
 ~~Marshfield Labs Presents~~ Laboratory Monitoring of Anticoagulant Therapy Session 3 of 4 Michael J. Sanfelippo, M.S. Technical Director, Coagulation Services Session 3 Topics Direct Thrombin Inhibitors:
~~Marshfield Labs Presents~~ Laboratory Monitoring of Anticoagulant Therapy Session 3 of 4 Michael J. Sanfelippo, M.S. Technical Director, Coagulation Services Session 3 Topics Direct Thrombin Inhibitors:
Volume of Distribution
 1 Volume of Distribution Nick Holford Dept Pharmacology & Clinical Pharmacology University of Auckland, New Zealand 2 Objectives Learn the definition of volume of distribution Understand the physiological
1 Volume of Distribution Nick Holford Dept Pharmacology & Clinical Pharmacology University of Auckland, New Zealand 2 Objectives Learn the definition of volume of distribution Understand the physiological
Basic Pharmacology. 2009 by Paul Nicolazzo the Wilderness Medicine Training Center wildmedcenter.com
 Basic Pharmacology 2009 by Paul Nicolazzo the Wilderness Medicine Training Center wildmedcenter.com Basic Pharmacology Introduction Pharmacology is the study of drugs. Drugs are defined as chemical substances
Basic Pharmacology 2009 by Paul Nicolazzo the Wilderness Medicine Training Center wildmedcenter.com Basic Pharmacology Introduction Pharmacology is the study of drugs. Drugs are defined as chemical substances
EFFIMET 1000 XR Metformin Hydrochloride extended release tablet
 BRAND NAME: Effimet XR. THERAPEUTIC CATEGORY: Anti-Diabetic PHARMACOLOGIC CLASS: Biguanides EFFIMET 1000 XR Metformin Hydrochloride extended release tablet COMPOSITION AND PRESENTATION Composition Each
BRAND NAME: Effimet XR. THERAPEUTIC CATEGORY: Anti-Diabetic PHARMACOLOGIC CLASS: Biguanides EFFIMET 1000 XR Metformin Hydrochloride extended release tablet COMPOSITION AND PRESENTATION Composition Each
Asian Journal of Research in Biological and Pharmaceutical Sciences Journal home page: www.ajrbps.com
 Research Article ISSN: 2349 4492 Asian Journal of Research in Biological and Pharmaceutical Sciences Journal home page: www.ajrbps.com IMPROVEMENT OF SOLUBILITY OF OMEPRAZOLE MAGNESIUM BY SOLID DISPERSION
Research Article ISSN: 2349 4492 Asian Journal of Research in Biological and Pharmaceutical Sciences Journal home page: www.ajrbps.com IMPROVEMENT OF SOLUBILITY OF OMEPRAZOLE MAGNESIUM BY SOLID DISPERSION
Bundesinstitut für Arzneimittel und Medizinprodukte. Dissolution Testing. Analytik,Methodenentwicklung, Bioäquivalenz SAQ. Olten, 25.
 Dissolution Testing Analytik,Methodenentwicklung, Bioäquivalenz SAQ Olten, 25. Januar 2006 Dr. H. Potthast (h.potthast@bfarm.de) 1 2 Basis for Biowaiver Applications/Decisions Note for Guidance on the
Dissolution Testing Analytik,Methodenentwicklung, Bioäquivalenz SAQ Olten, 25. Januar 2006 Dr. H. Potthast (h.potthast@bfarm.de) 1 2 Basis for Biowaiver Applications/Decisions Note for Guidance on the
The role of CO 2 in pool water
 The role of CO 2 in pool water A series of e-mail articles from the research group onbalance, January 2006 The role of CO 2 in pool water #1 While some service techs go about their business taking care
The role of CO 2 in pool water A series of e-mail articles from the research group onbalance, January 2006 The role of CO 2 in pool water #1 While some service techs go about their business taking care
6023-1 - Page 1. Name: 4) The diagram below represents a beaker containing a solution of various molecules involved in digestion.
 Name: 6023-1 - Page 1 1) Which one of the following situations indicates a serious organ system malfunction? A) Mitochondria stop functioning in a unicellular organism exposed to pollutants. B) White blood
Name: 6023-1 - Page 1 1) Which one of the following situations indicates a serious organ system malfunction? A) Mitochondria stop functioning in a unicellular organism exposed to pollutants. B) White blood
DIVISION OF HUMAN NUTRITION
 DIVISION OF HUMAN NUTRITION Example EXAM 2011 - HNE-23306 Nutrition & Pharmacology Date : Place : Explanation : This exam consists of: **** open problems on pharmacokinetics (normally 2-3) *****series
DIVISION OF HUMAN NUTRITION Example EXAM 2011 - HNE-23306 Nutrition & Pharmacology Date : Place : Explanation : This exam consists of: **** open problems on pharmacokinetics (normally 2-3) *****series
Guidance for Industry Safety Testing of Drug Metabolites
 Guidance for Industry Safety Testing of Drug Metabolites U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2008 Pharmacology
Guidance for Industry Safety Testing of Drug Metabolites U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2008 Pharmacology
COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP)
 The European Agency for the Evaluation of Medicinal Products Human Medicines Evaluation Unit London, 29 July 1999 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) NOTE FOR GUIDANCE ON QUALITY OF MODIFIED
The European Agency for the Evaluation of Medicinal Products Human Medicines Evaluation Unit London, 29 July 1999 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) NOTE FOR GUIDANCE ON QUALITY OF MODIFIED
EXIGENCIA DE ESTUDIOS DE BIOEQUIVALENCIA A TRAVÉS DE METODOS IN VITRO
 EXIGENCIA DE ESTUDIOS DE BIOEQUIVALENCIA A TRAVÉS DE METODOS IN VITRO Q.F. ALEXIS ACEITUNO, PhD Jefe Subdepto. Biofarmacia & Bioequivalencia Agencia Nacional de Medicamentos Instituto de Salud Pública
EXIGENCIA DE ESTUDIOS DE BIOEQUIVALENCIA A TRAVÉS DE METODOS IN VITRO Q.F. ALEXIS ACEITUNO, PhD Jefe Subdepto. Biofarmacia & Bioequivalencia Agencia Nacional de Medicamentos Instituto de Salud Pública
Pharmacology skills for drug discovery. Why is pharmacology important?
 skills for drug discovery Why is pharmacology important?, the science underlying the interaction between chemicals and living systems, emerged as a distinct discipline allied to medicine in the mid-19th
skills for drug discovery Why is pharmacology important?, the science underlying the interaction between chemicals and living systems, emerged as a distinct discipline allied to medicine in the mid-19th
Name Date Period. Keystone Review Enzymes
 Name Date Period Keystone Review Enzymes 1. In order for cells to function properly, the enzymes that they contain must also function properly. What can be inferred using the above information? A. Cells
Name Date Period Keystone Review Enzymes 1. In order for cells to function properly, the enzymes that they contain must also function properly. What can be inferred using the above information? A. Cells
PRINCIPLES OF PHARMACOKINETICS
 Harvard-MIT Division of Health Sciences and Technology HST.151: Principles of Pharmocology Instructor: Prof. Carol Walsh HST-151 1 PRINCIPLES OF PHARMACOKINETICS Learning Objectives: 1. Describe the physicochemical
Harvard-MIT Division of Health Sciences and Technology HST.151: Principles of Pharmocology Instructor: Prof. Carol Walsh HST-151 1 PRINCIPLES OF PHARMACOKINETICS Learning Objectives: 1. Describe the physicochemical
Understanding Our Curriculum
 Understanding Our Curriculum One question that comes up quiet frequently when talking with preceptors is what are we teaching our students and when are they exposed to certain classes. Below you will find
Understanding Our Curriculum One question that comes up quiet frequently when talking with preceptors is what are we teaching our students and when are they exposed to certain classes. Below you will find
TOTAL PROTEIN FIBRINOGEN
 UNIT: Proteins 16tproteins.wpd Task Determination of Total Protein, Albumin and Globulins Objectives Upon completion of this exercise, the student will be able to: 1. Explain the ratio of albumin and globulin
UNIT: Proteins 16tproteins.wpd Task Determination of Total Protein, Albumin and Globulins Objectives Upon completion of this exercise, the student will be able to: 1. Explain the ratio of albumin and globulin
PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement)
 1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
Public Assessment Report Scientific discussion. Tenofovir disoproxil Teva (tenofovir disoproxil) SE/H/1432/01/DC
 Public Assessment Report Scientific discussion Tenofovir disoproxil Teva (tenofovir disoproxil) SE/H/1432/01/DC This module reflects the scientific discussion for the approval of Tenofovir disoproxil Teva.
Public Assessment Report Scientific discussion Tenofovir disoproxil Teva (tenofovir disoproxil) SE/H/1432/01/DC This module reflects the scientific discussion for the approval of Tenofovir disoproxil Teva.
Teriflunomide is the active metabolite of Leflunomide, a drug employed since 1994 for the treatment of rheumatoid arthritis (Baselt, 2011).
 Page 1 of 10 ANALYTE NAME AND STRUCTURE TERIFLUNOMIDE Teriflunomide TRADE NAME Aubagio CATEGORY Antimetabolite TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND sclerosis. The
Page 1 of 10 ANALYTE NAME AND STRUCTURE TERIFLUNOMIDE Teriflunomide TRADE NAME Aubagio CATEGORY Antimetabolite TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND sclerosis. The
NSW Higher School Certificate Senior Science 9.2 Lifestyle Chemistry
 NSW Higher School Certificate Senior Science 9.2 Lifestyle Chemistry Section 5 Drug Solubility 9.2 Lifestyle Chemistry Section 5 ::: Drug Solubility 9.2.5 The solubility of drugs has an effect on the way
NSW Higher School Certificate Senior Science 9.2 Lifestyle Chemistry Section 5 Drug Solubility 9.2 Lifestyle Chemistry Section 5 ::: Drug Solubility 9.2.5 The solubility of drugs has an effect on the way
Statistics and Pharmacokinetics in Clinical Pharmacology Studies
 Paper ST03 Statistics and Pharmacokinetics in Clinical Pharmacology Studies ABSTRACT Amy Newlands, GlaxoSmithKline, Greenford UK The aim of this presentation is to show how we use statistics and pharmacokinetics
Paper ST03 Statistics and Pharmacokinetics in Clinical Pharmacology Studies ABSTRACT Amy Newlands, GlaxoSmithKline, Greenford UK The aim of this presentation is to show how we use statistics and pharmacokinetics
Enzymes. A. a lipid B. a protein C. a carbohydrate D. a mineral
 Enzymes 1. All cells in multicellular organisms contain thousands of different kinds of enzymes that are specialized to catalyze different chemical reactions. Given this information, which of the following
Enzymes 1. All cells in multicellular organisms contain thousands of different kinds of enzymes that are specialized to catalyze different chemical reactions. Given this information, which of the following
6 Characterization of Casein and Bovine Serum Albumin
 6 Characterization of Casein and Bovine Serum Albumin (BSA) Objectives: A) To separate a mixture of casein and bovine serum albumin B) to characterize these proteins based on their solubilities as a function
6 Characterization of Casein and Bovine Serum Albumin (BSA) Objectives: A) To separate a mixture of casein and bovine serum albumin B) to characterize these proteins based on their solubilities as a function
ROUTES OF DRUG ADMINISTRATION
 ROUTES OF DRUG ADMINISTRATION Review Article P. Verma (1) *, A.S. Thakur (1), K. Deshmukh (2), Dr. A.K. Jha (1) S. Verma (2) Address for Correspondence 1. Shri Shankaracharya Institute of Pharmaceutical
ROUTES OF DRUG ADMINISTRATION Review Article P. Verma (1) *, A.S. Thakur (1), K. Deshmukh (2), Dr. A.K. Jha (1) S. Verma (2) Address for Correspondence 1. Shri Shankaracharya Institute of Pharmaceutical
Eating, pooping, and peeing THE DIGESTIVE SYSTEM
 THE DIGESTIVE SYSTEM Ingested food is not technically in the body until it is absorbed so it needs to be: Mechanically and chemically reduced Transported by the blood to the cells Large portions are not
THE DIGESTIVE SYSTEM Ingested food is not technically in the body until it is absorbed so it needs to be: Mechanically and chemically reduced Transported by the blood to the cells Large portions are not
Hillsborough Community College - Ybor City Campus 1025C Laboratory Exercise 9: Testing Urine for the Presence of Drugs Introduction
 Hillsborough Community College - Ybor City Campus 1025C Laboratory Exercise 9: Testing Urine for the Presence of Drugs Introduction Drug testing beyond the health care and criminal justice systems has
Hillsborough Community College - Ybor City Campus 1025C Laboratory Exercise 9: Testing Urine for the Presence of Drugs Introduction Drug testing beyond the health care and criminal justice systems has
DRUGS FOR GLUCOSE MANAGEMENT AND DIABETES
 Page 1 DRUGS FOR GLUCOSE MANAGEMENT AND DIABETES Drugs to know are: Actrapid HM Humulin R, L, U Penmix SUNALI MEHTA The three principal hormones produced by the pancreas are: Insulin: nutrient metabolism:
Page 1 DRUGS FOR GLUCOSE MANAGEMENT AND DIABETES Drugs to know are: Actrapid HM Humulin R, L, U Penmix SUNALI MEHTA The three principal hormones produced by the pancreas are: Insulin: nutrient metabolism:
serum protein and A/ G ratio
 serum protein and A/ G ratio Blood plasma contains at least 125 individual proteins. Serum ( as contrasted with plasma) is deficient in those coagulation protein which are consumed during the process of
serum protein and A/ G ratio Blood plasma contains at least 125 individual proteins. Serum ( as contrasted with plasma) is deficient in those coagulation protein which are consumed during the process of
Carl Rosow, M.D., Ph.D. 1 HST-151. Lecture 1 - Principles of Pharmacology: Introduction
 Harvard-MIT Division of Health Sciences and Technology HST.151: Principles of Pharmocology Instructor: Dr. Carl Rosow Carl Rosow, M.D., Ph.D. 1 HST-151 Lecture 1 - Principles of Pharmacology: Introduction
Harvard-MIT Division of Health Sciences and Technology HST.151: Principles of Pharmocology Instructor: Dr. Carl Rosow Carl Rosow, M.D., Ph.D. 1 HST-151 Lecture 1 - Principles of Pharmacology: Introduction
Guidance for Industry
 Guidance for Industry Bioavailability and Bioequivalence Studies for Orally Administered Drug Products General Considerations U.S. Department of Health and Human Services Food and Drug Administration Center
Guidance for Industry Bioavailability and Bioequivalence Studies for Orally Administered Drug Products General Considerations U.S. Department of Health and Human Services Food and Drug Administration Center
Chapter 14 Urinalysis, Body Fluids and Other Specimens. Objectives:
 EXERCISE 15: CHEMICAL EXAMINATION OF URINE Textbook: Skill: Chapter 14 Urinalysis, Body Fluids and Other Specimens 15 points Objectives: 1. Name 10 routine chemical tests performed on urine and list a
EXERCISE 15: CHEMICAL EXAMINATION OF URINE Textbook: Skill: Chapter 14 Urinalysis, Body Fluids and Other Specimens 15 points Objectives: 1. Name 10 routine chemical tests performed on urine and list a
INSULIN PRODUCTS. Jack DeRuiter
 INSULIN PRODUCTS Jack DeRuiter The number and types of insulin preparations available in the United States is constantly changing, thus students should refer to recent drug resources for a current list
INSULIN PRODUCTS Jack DeRuiter The number and types of insulin preparations available in the United States is constantly changing, thus students should refer to recent drug resources for a current list
Digestive System Why is digestion important? How is food digested? Physical Digestion and Movement
 Digestive System The digestive system is made up of the digestive tract a series of hollow organs joined in a long, twisting tube from the mouth to the anus and other organs that help the body break down
Digestive System The digestive system is made up of the digestive tract a series of hollow organs joined in a long, twisting tube from the mouth to the anus and other organs that help the body break down
Drug passage across the cell membrane
 SECTION I Basic principles 1 Drug passage across the cell membrane Many drugs need to pass through one or more cell membranes to reach their site of action. A common feature of all cell membranes is a
SECTION I Basic principles 1 Drug passage across the cell membrane Many drugs need to pass through one or more cell membranes to reach their site of action. A common feature of all cell membranes is a
THE DIGESTIVE SYSTEM
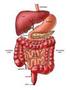 THE DIGESTIVE SYSTEM What is digestion? Digestion is the process of breaking down food so that it's small enough to be absorbed and used by the body for energy or in other bodily functions. Digestion involves
THE DIGESTIVE SYSTEM What is digestion? Digestion is the process of breaking down food so that it's small enough to be absorbed and used by the body for energy or in other bodily functions. Digestion involves
LAB 3: DIGESTION OF ORGANIC MACROMOLECULES
 LAB 3: DIGESTION OF ORGANIC MACROMOLECULES INTRODUCTION Enzymes are a special class of proteins that lower the activation energy of biological reactions. These biological catalysts change the rate of chemical
LAB 3: DIGESTION OF ORGANIC MACROMOLECULES INTRODUCTION Enzymes are a special class of proteins that lower the activation energy of biological reactions. These biological catalysts change the rate of chemical
CHAPTER 13: SOLUTIONS
 CHAPTER 13: SOLUTIONS Problems: 1-8, 11-15, 20-30, 37-88, 107-110, 131-132 13.2 SOLUTIONS: HOMOGENEOUS MIXTURES solution: homogeneous mixture of substances present as atoms, ions, and/or molecules solute:
CHAPTER 13: SOLUTIONS Problems: 1-8, 11-15, 20-30, 37-88, 107-110, 131-132 13.2 SOLUTIONS: HOMOGENEOUS MIXTURES solution: homogeneous mixture of substances present as atoms, ions, and/or molecules solute:
Diabetes mellitus 1 عبد هللا الزعبي. pharmacology. Shatha Khalil Shahwan. 1 P a g e
 Diabetes mellitus 1 pharmacology عبد هللا الزعبي 1 P a g e 4 Shatha Khalil Shahwan Diabetes mellitus The goals of the treatment of diabetes 1. Treating symptoms 2. Treating and Preventing acute complications
Diabetes mellitus 1 pharmacology عبد هللا الزعبي 1 P a g e 4 Shatha Khalil Shahwan Diabetes mellitus The goals of the treatment of diabetes 1. Treating symptoms 2. Treating and Preventing acute complications
1. Essay: The Digestive and Absorption Processes of Macronutrients
 Jenny Kim Professor Rosario Nutrition: Macronutrients Project June 26, 2014 1. Essay: The Digestive and Absorption Processes of Macronutrients Whenever we eat, the foods we ingest in our bodies undergo
Jenny Kim Professor Rosario Nutrition: Macronutrients Project June 26, 2014 1. Essay: The Digestive and Absorption Processes of Macronutrients Whenever we eat, the foods we ingest in our bodies undergo
European Continuing Education College
 Design and Development of Conventional and Modified Release Oral Drug Delivery Systems Three Day Intensive Course for Managers, Scientists and Technicians with the Emphasis on the Principles of Oral Drug
Design and Development of Conventional and Modified Release Oral Drug Delivery Systems Three Day Intensive Course for Managers, Scientists and Technicians with the Emphasis on the Principles of Oral Drug
Analytical Specifications RIVAROXABAN
 Page 1 of 9 ANALYTE NAME AND STRUCTURE - RIVAROXABAN SYNONYMS Xarelto CATEGORY Anticoagulant TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND Xarelto (rivaroxaban) is an orally
Page 1 of 9 ANALYTE NAME AND STRUCTURE - RIVAROXABAN SYNONYMS Xarelto CATEGORY Anticoagulant TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND Xarelto (rivaroxaban) is an orally
10.2 The Human Digestive System pg. 411
 10.2 The Human Digestive System pg. 411 The human digestive system is made up of a group of organs working together. The digestive tract is made up of the mouth, esophagus, stomach, small intestine, and
10.2 The Human Digestive System pg. 411 The human digestive system is made up of a group of organs working together. The digestive tract is made up of the mouth, esophagus, stomach, small intestine, and
Activity Sheets Enzymes and Their Functions
 Name: Date: Activity Sheets Enzymes and Their Functions amylase What are Enzymes? starch glucose Enzymes are compounds that assist chemical reactions by increasing the rate at which they occur. For example,
Name: Date: Activity Sheets Enzymes and Their Functions amylase What are Enzymes? starch glucose Enzymes are compounds that assist chemical reactions by increasing the rate at which they occur. For example,
William Shaw, Ph.D. The Great Plains Laboratory, Inc., Lenexa, Kansas, USA
 Inhibition of dopamine conversion to norepinephrine by Clostridia metabolites appears to be a (the) major cause of autism, schizophrenia, and other neuropsychiatric disorders. All these factors can now
Inhibition of dopamine conversion to norepinephrine by Clostridia metabolites appears to be a (the) major cause of autism, schizophrenia, and other neuropsychiatric disorders. All these factors can now
Acids & Bases. BRONSTED LOWRY ACIDS & BASES A Bronsted Lowry Acid DONATES A PROTON A Bronsted Lowry Base ACCEPTS A PROTON.
 rganic Chemistry Lecture utline BRSTED LWRY ACIDS & BASES A Bronsted Lowry DATES A PRT A Bronsted Lowry ACCEPTS A PRT Cl Cl CJUGATE ACIDS AD BASES A conjugate acid results after a Bronsted Lowry base has
rganic Chemistry Lecture utline BRSTED LWRY ACIDS & BASES A Bronsted Lowry DATES A PRT A Bronsted Lowry ACCEPTS A PRT Cl Cl CJUGATE ACIDS AD BASES A conjugate acid results after a Bronsted Lowry base has
BIOAVAILABILITY & BIOEQUIVALENCE TRIALS
 BIOAVAILABILITY & BIOEQUIVALENCE TRIALS Shubha Rani,, Ph.D. Technical Director & Head-Biometrics and Data Management Synchron Research Services Pvt. Ltd. Ahmedabad 380 054 drshubha@synchronresearch.com
BIOAVAILABILITY & BIOEQUIVALENCE TRIALS Shubha Rani,, Ph.D. Technical Director & Head-Biometrics and Data Management Synchron Research Services Pvt. Ltd. Ahmedabad 380 054 drshubha@synchronresearch.com
NONCLINICAL EVALUATION FOR ANTICANCER PHARMACEUTICALS
 INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE NONCLINICAL EVALUATION FOR ANTICANCER PHARMACEUTICALS
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE NONCLINICAL EVALUATION FOR ANTICANCER PHARMACEUTICALS
Decentralised Procedure. Public Assessment Report
 Bundesinstitut für Arzneimittel und Medizinprodukte Decentralised Procedure Public Assessment Report ben-u-ron direkt Erdbeer/Vanille 250/500 mg Granulat in Beuteln ben-u-ron direkt Cappuccino 500/1000
Bundesinstitut für Arzneimittel und Medizinprodukte Decentralised Procedure Public Assessment Report ben-u-ron direkt Erdbeer/Vanille 250/500 mg Granulat in Beuteln ben-u-ron direkt Cappuccino 500/1000
Introduction to pharmaceutical technology
 Introduction to pharmaceutical technology Marie Wahlgren Chapter 1 What is the topics of today Introduction to the course Introduction to the project assignment How to choose a new drug formulation 1 Contacts
Introduction to pharmaceutical technology Marie Wahlgren Chapter 1 What is the topics of today Introduction to the course Introduction to the project assignment How to choose a new drug formulation 1 Contacts
MOLECULAR PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS
 MOLECULAR PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS R. M. Weinshilboum, M.D., Program Director L. Wang, M.D., Ph.D., Program Co-Director D. C. Mays, Ph.D., Associate Program Director Ph.D. Degree Course
MOLECULAR PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS R. M. Weinshilboum, M.D., Program Director L. Wang, M.D., Ph.D., Program Co-Director D. C. Mays, Ph.D., Associate Program Director Ph.D. Degree Course
BIOL 305L Laboratory Two
 Please print Full name clearly: Introduction BIOL 305L Laboratory Two Osmosis, because it is different in plants! Osmosis is the movement of solvent molecules through a selectively permeable membrane into
Please print Full name clearly: Introduction BIOL 305L Laboratory Two Osmosis, because it is different in plants! Osmosis is the movement of solvent molecules through a selectively permeable membrane into
1. Give the name and functions of the structure labeled A on the diagram. 2. Give the name and functions of the structure labeled B on the diagram.
 2013 ANATOMY & PHYSIOLOGY Sample Tournament Station A: Use the diagram in answering Questions 1-5. 1. Give the name and functions of the structure labeled A on the diagram. 2. Give the name and functions
2013 ANATOMY & PHYSIOLOGY Sample Tournament Station A: Use the diagram in answering Questions 1-5. 1. Give the name and functions of the structure labeled A on the diagram. 2. Give the name and functions
Guidance for Industry
 Guidance for Industry Dissolution Testing of Immediate Release Solid Oral Dosage Forms U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research
Guidance for Industry Dissolution Testing of Immediate Release Solid Oral Dosage Forms U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research
Chemistry 20 Chapters 15 Enzymes
 Chemistry 20 Chapters 15 Enzymes Enzymes: as a catalyst, an enzyme increases the rate of a reaction by changing the way a reaction takes place, but is itself not changed at the end of the reaction. An
Chemistry 20 Chapters 15 Enzymes Enzymes: as a catalyst, an enzyme increases the rate of a reaction by changing the way a reaction takes place, but is itself not changed at the end of the reaction. An
Acid/Base Homeostasis (Part 4)
 Acid/Base Homeostasis (Part 4) Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.com) 5. The newly formed bicarbonate moves into the plasma.
Acid/Base Homeostasis (Part 4) Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.com) 5. The newly formed bicarbonate moves into the plasma.
CLOSED LOOP MODEL FOR GLUCOSE INSULIN REGULATION SYSTEM USING LABVIEW
 CLOSED LOOP MODEL FOR GLUCOSE INSULIN REGULATION SYSTEM USING LABVIEW 1 P Srinivas 2 P.Durga Prasada Rao 1 Associate Professor, Department of EIE, VR Siddhartha Engineering College, Vijayawada, India Email:
CLOSED LOOP MODEL FOR GLUCOSE INSULIN REGULATION SYSTEM USING LABVIEW 1 P Srinivas 2 P.Durga Prasada Rao 1 Associate Professor, Department of EIE, VR Siddhartha Engineering College, Vijayawada, India Email:
Oral Bioavailability of Creatine Supplements: Is There Room for Improvement?
 Oral Bioavailability of Creatine Supplements: Is There Room for Improvement? Donald W. Miller Associate Professor Department of Pharmacology and Therapeutics University of Manitoba Acknowledgements ISSN
Oral Bioavailability of Creatine Supplements: Is There Room for Improvement? Donald W. Miller Associate Professor Department of Pharmacology and Therapeutics University of Manitoba Acknowledgements ISSN
FIGURE 2.18. A. The phosphate end of the molecule is polar (charged) and hydrophilic (attracted to water).
 PLASMA MEMBRANE 1. The plasma membrane is the outermost part of a cell. 2. The main component of the plasma membrane is phospholipids. FIGURE 2.18 A. The phosphate end of the molecule is polar (charged)
PLASMA MEMBRANE 1. The plasma membrane is the outermost part of a cell. 2. The main component of the plasma membrane is phospholipids. FIGURE 2.18 A. The phosphate end of the molecule is polar (charged)
Generic drugs are copies of innovator drug products
 dx.doi.org/10.14227/dt190412p51 In Vitro Equivalence Studies of Generic Metformin Hydrochloride Tablets and Propranolol Hydrochloride Tablets Under Biowaiver Conditions in Lagos State, Nigeria e-mail:
dx.doi.org/10.14227/dt190412p51 In Vitro Equivalence Studies of Generic Metformin Hydrochloride Tablets and Propranolol Hydrochloride Tablets Under Biowaiver Conditions in Lagos State, Nigeria e-mail:
Lead optimization services
 Lead optimization services The WIL Research Company (WRC) has extensive experience in fast track tailor-made screening strategies to help you with the challenging task of selecting your best candidate
Lead optimization services The WIL Research Company (WRC) has extensive experience in fast track tailor-made screening strategies to help you with the challenging task of selecting your best candidate
Guidance for Industry
 Guidance for Industry Bioavailability and Bioequivalence Studies Submitted in NDAs or INDs General Considerations DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments
Guidance for Industry Bioavailability and Bioequivalence Studies Submitted in NDAs or INDs General Considerations DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments
Abdulaziz Al-Subaie. Anfal Al-Shalwi
 Abdulaziz Al-Subaie Anfal Al-Shalwi Introduction what is diabetes mellitus? A chronic metabolic disorder characterized by high blood glucose level caused by insulin deficiency and sometimes accompanied
Abdulaziz Al-Subaie Anfal Al-Shalwi Introduction what is diabetes mellitus? A chronic metabolic disorder characterized by high blood glucose level caused by insulin deficiency and sometimes accompanied
Biological importance of metabolites. Safety and efficacy aspects
 Biological importance of metabolites Safety and efficacy aspects Bernard Walther Technologie Servier Biological importance of metabolites Safety testing of drug metabolites Bioanalytical strategy Structural
Biological importance of metabolites Safety and efficacy aspects Bernard Walther Technologie Servier Biological importance of metabolites Safety testing of drug metabolites Bioanalytical strategy Structural
5.111 Principles of Chemical Science
 MIT OpenCourseWare http://ocw.mit.edu 5.111 Principles of Chemical Science Fall 2008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 5.111 Principles
MIT OpenCourseWare http://ocw.mit.edu 5.111 Principles of Chemical Science Fall 2008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 5.111 Principles
DRAFT GUIDANCE. This guidance document is being distributed for comment purposes only.
 Waiver of In Vivo Bioavailability and Bioequivalence Studies for Immediate-Release Solid Oral Dosage Forms Based on a Biopharmaceutics Classification System Guidance for Industry DRAFT GUIDANCE This guidance
Waiver of In Vivo Bioavailability and Bioequivalence Studies for Immediate-Release Solid Oral Dosage Forms Based on a Biopharmaceutics Classification System Guidance for Industry DRAFT GUIDANCE This guidance
MANAGING ANEMIA. When You Have Kidney Disease or Kidney Failure. www.kidney.org
 MANAGING ANEMIA When You Have Kidney Disease or Kidney Failure www.kidney.org About the Information in this Booklet Did you know that the National Kidney Foundation (NKF) offers guidelines and commentaries
MANAGING ANEMIA When You Have Kidney Disease or Kidney Failure www.kidney.org About the Information in this Booklet Did you know that the National Kidney Foundation (NKF) offers guidelines and commentaries
4. Biology of the Cell
 4. Biology of the Cell Our primary focus in this chapter will be the plasma membrane and movement of materials across the plasma membrane. You should already be familiar with the basic structures and roles
4. Biology of the Cell Our primary focus in this chapter will be the plasma membrane and movement of materials across the plasma membrane. You should already be familiar with the basic structures and roles
Table of Content. Enzymes and Their Functions Teacher Version 1
 Enzymes and Their Functions Jeisa Pelet, Cornell University Carolyn Wilczynski, Binghamton High School Cornell Learning Initiative in Medicine and Bioengineering (CLIMB) Table of Content Title Page Abstract..
Enzymes and Their Functions Jeisa Pelet, Cornell University Carolyn Wilczynski, Binghamton High School Cornell Learning Initiative in Medicine and Bioengineering (CLIMB) Table of Content Title Page Abstract..
I. ACID-BASE NEUTRALIZATION, TITRATION
 LABORATORY 3 I. ACID-BASE NEUTRALIZATION, TITRATION Acid-base neutralization is a process in which acid reacts with base to produce water and salt. The driving force of this reaction is formation of a
LABORATORY 3 I. ACID-BASE NEUTRALIZATION, TITRATION Acid-base neutralization is a process in which acid reacts with base to produce water and salt. The driving force of this reaction is formation of a
FACT SHEET TESTETROL, A NOVEL ORALLY BIOACTIVE ANDROGEN
 FACT SHEET TESTETROL, A NOVEL ORALLY BIOACTIVE ANDROGEN General Pantarhei Bioscience B.V. is an emerging specialty pharmaceutical company with a creative approach towards drug development. The Company
FACT SHEET TESTETROL, A NOVEL ORALLY BIOACTIVE ANDROGEN General Pantarhei Bioscience B.V. is an emerging specialty pharmaceutical company with a creative approach towards drug development. The Company
HYPERTENSION ASSOCIATED WITH RENAL DISEASES
 RENAL DISEASE v Patients with renal insufficiency should be encouraged to reduce dietary salt and protein intake. v Target blood pressure is less than 135-130/85 mmhg. If patients have urinary protein
RENAL DISEASE v Patients with renal insufficiency should be encouraged to reduce dietary salt and protein intake. v Target blood pressure is less than 135-130/85 mmhg. If patients have urinary protein
BSc in Medical Sciences with PHARMACOLOGY
 BSc in Medical Sciences with PHARMACOLOGY Course Director Dr Christopher John Module Leaders Dr Robert Dickinson (Module 1) Dr Anabel Varela Carver (Module 2) Dr Sohag Saleh (Module 3) Course Administrator
BSc in Medical Sciences with PHARMACOLOGY Course Director Dr Christopher John Module Leaders Dr Robert Dickinson (Module 1) Dr Anabel Varela Carver (Module 2) Dr Sohag Saleh (Module 3) Course Administrator
The Clinical Trials Process an educated patient s guide
 The Clinical Trials Process an educated patient s guide Gwen L. Nichols, MD Site Head, Oncology Roche TCRC, Translational and Clinical Research Center New York DISCLAIMER I am an employee of Hoffmann-
The Clinical Trials Process an educated patient s guide Gwen L. Nichols, MD Site Head, Oncology Roche TCRC, Translational and Clinical Research Center New York DISCLAIMER I am an employee of Hoffmann-
VITAMIN B2. By : Dania Adel & Hanin Fareed
 VITAMIN B2 By : Dania Adel & Hanin Fareed VITAMIN B2 Vitamin B2, which is also known as Riboflavin, is an easily absorbed colored micronutrient with a key role in maintaining health in humans and other
VITAMIN B2 By : Dania Adel & Hanin Fareed VITAMIN B2 Vitamin B2, which is also known as Riboflavin, is an easily absorbed colored micronutrient with a key role in maintaining health in humans and other
Chapter 48. Nutrients in Food. Carbohydrates, Proteins, and Lipids. Carbohydrates, Proteins, and Lipids, continued
 Carbohydrates, Proteins, and Lipids The three nutrients needed by the body in the greatest amounts are carbohydrates, proteins, and lipids. Nutrients in Food All of these nutrients are called organic compounds,
Carbohydrates, Proteins, and Lipids The three nutrients needed by the body in the greatest amounts are carbohydrates, proteins, and lipids. Nutrients in Food All of these nutrients are called organic compounds,
Chapter 25: Metabolism and Nutrition
 Chapter 25: Metabolism and Nutrition Chapter Objectives INTRODUCTION 1. Generalize the way in which nutrients are processed through the three major metabolic fates in order to perform various energetic
Chapter 25: Metabolism and Nutrition Chapter Objectives INTRODUCTION 1. Generalize the way in which nutrients are processed through the three major metabolic fates in order to perform various energetic
The diagram below summarizes the effects of the compounds that cells use to regulate their own metabolism.
 Regulation of carbohydrate metabolism Intracellular metabolic regulators Each of the control point steps in the carbohydrate metabolic pathways in effect regulates itself by responding to molecules that
Regulation of carbohydrate metabolism Intracellular metabolic regulators Each of the control point steps in the carbohydrate metabolic pathways in effect regulates itself by responding to molecules that
Biopharmaceuticals and Biotechnology Unit 2 Student Handout. DNA Biotechnology and Enzymes
 DNA Biotechnology and Enzymes 35 Background Unit 2~ Lesson 1 The Biotechnology Industry Biotechnology is a process (or a technology) that is used to create products like medicines by using micro-organisms,
DNA Biotechnology and Enzymes 35 Background Unit 2~ Lesson 1 The Biotechnology Industry Biotechnology is a process (or a technology) that is used to create products like medicines by using micro-organisms,
Unit 2: Cells, Membranes and Signaling CELL MEMBRANE. Chapter 5 Hillis Textbook
 Unit 2: Cells, Membranes and Signaling CELL MEMBRANE Chapter 5 Hillis Textbook HOW DOES THE LAB RELATE TO THE NEXT CHAPTER? SURFACE AREA: the entire outer covering of a cell that enables materials pass.
Unit 2: Cells, Membranes and Signaling CELL MEMBRANE Chapter 5 Hillis Textbook HOW DOES THE LAB RELATE TO THE NEXT CHAPTER? SURFACE AREA: the entire outer covering of a cell that enables materials pass.
Digestion, Absorption. How & where?
 Digestion, Absorption How & where? What happens to food? Three processes Digestion Absorption Elimination Where do they occur? GI tract Overview of Digestion GI tract Gastrointestinal (GI) tract: series
Digestion, Absorption How & where? What happens to food? Three processes Digestion Absorption Elimination Where do they occur? GI tract Overview of Digestion GI tract Gastrointestinal (GI) tract: series
MANAGEMENT OF DIABETIC PATIENTS IN RADIOLGY. Edition 2
 MANAGEMENT OF DIABETIC PATIENTS IN RADIOLGY Edition 2 Author, Authority Clinical Director Date of Origin December 2007 Date of Review November 2009 Scope of Authority, (Trust, other) Trust Total pages
MANAGEMENT OF DIABETIC PATIENTS IN RADIOLGY Edition 2 Author, Authority Clinical Director Date of Origin December 2007 Date of Review November 2009 Scope of Authority, (Trust, other) Trust Total pages
Revision of The Dissolution Procedure: Development and Validation 1092
 Page 1 of 5 STIMULI TO THE REVISION PROCESS Stimuli articles do not necessarily reflect the policies of the USPC or the USP Council of Experts Revision of The Dissolution Procedure: Development and Validation
Page 1 of 5 STIMULI TO THE REVISION PROCESS Stimuli articles do not necessarily reflect the policies of the USPC or the USP Council of Experts Revision of The Dissolution Procedure: Development and Validation
Lecture 11 Enzymes: Kinetics
 Lecture 11 Enzymes: Kinetics Reading: Berg, Tymoczko & Stryer, 6th ed., Chapter 8, pp. 216-225 Key Concepts Kinetics is the study of reaction rates (velocities). Study of enzyme kinetics is useful for
Lecture 11 Enzymes: Kinetics Reading: Berg, Tymoczko & Stryer, 6th ed., Chapter 8, pp. 216-225 Key Concepts Kinetics is the study of reaction rates (velocities). Study of enzyme kinetics is useful for
Laboratory 5: Properties of Enzymes
 Laboratory 5: Properties of Enzymes Technical Objectives 1. Accurately measure and transfer solutions with pipettes 2. Use a Spectrophotometer to study enzyme action. 3. Properly graph a set of data. Knowledge
Laboratory 5: Properties of Enzymes Technical Objectives 1. Accurately measure and transfer solutions with pipettes 2. Use a Spectrophotometer to study enzyme action. 3. Properly graph a set of data. Knowledge
