AUTOMATED HPLC SCREENING AND METHOD DEVELOPMENT SYSTEM
|
|
|
- Angelina Walsh
- 7 years ago
- Views:
Transcription
1 WA2277 AUTOMATED HPLC SCREENING AND METHOD DEVELOPMENT SYSTEM Rudy Sneyers, Willy Janssens, Achille Pluym and Ivan Somers Johnson & Johnson Pharmaceutical Research & Development a division of Janssen Pharmaceutica N.V. DRUG EVALUATION - ANALYTICAL DEVELOPMENT 1
2 INTRODUCTION For the development of selective HPLC methods to be used for quantitative deteration of impurities, degradants, excipients and active in the drug substance as well as in the drug product a screening concept is currently used. The concept is based on a matrix screening between different ph and stationary phases. The screening module consist of 4 HPLC systems each operating with a different MS compatible mobile phase at a different ph. Each HPLC system is connected with a valve switcher to permit automated selection between 8 columns with different selectivity. After screening 32 chromatograms for each sample have to be evaluated. The best separation is selected based on ph and column selectivity. 2
3 Screening Experiments A B ph = Zorbax Extend C18 2. Zorbax Bonus RP 3. Waters XTerra MS C18 4. Waters XTerra RP18 5. Waters Xterra Phenyl 6. Waters Symmetry Shield 7. YMC Pro C18 8. YMC Pack C4 A B ph = Zorbax Extend C18 2. Zorbax Bonus RP 3. Waters XTerra MS C18 4. Waters XTerra RP18 5. Waters Xterra Phenyl 6. Waters Symmetry Shield 7. YMC Pro C18 8. YMC Pack C4 A : 1 mm NH 4 OAc in water - CH 3 CN (95/5 v/v) +.1%, v/v TFA B : 1 mm NH 4 OAc in water - CH 3 CN (/9 v/v) +.1%, v/v TFA A : 1 mm NH 4 OAc in water - CH 3 CN (95/5 v/v) +.5%, v/v CH 3 COOH B : 1 mm NH 4 OAc in water - CH 3 CN (/9 v/v) +.5%, v/v CH 3 COOH A B ph = 7 1. Zorbax Extend C18 2. Zorbax Bonus RP 3. Waters XTerra MS C18 4. Waters XTerra RP18 5. Waters Xterra Phenyl 6. Waters Symmetry Shield 7. YMC Pro C18 8. YMC Pack C4 A B ph = 9 1. Zorbax Extend C18 2. Zorbax Bonus RP 3. Waters XTerra MS C18 4. Waters XTerra RP18 5. Waters Xterra Phenyl 6. Waters Symmetry Shield 7. YMC Pro C18 8. YMC Pack C4 A : 1 mm NH 4 OAc in water - CH 3 CN (95/5, v/v) B : 1 mm NH 4 OAc in water - CH 3 CN (/9, v/v) A : 1 mm NH 4 OAc in water - CH 3 CN (95/5 v/v) +.5%, v/v DEA B : 1 mm NH 4 OAc in water - CH 3 CN (/9 v/v) +.5%, v/v DEA 3
4 32 chromatograms for each sample 1. Zorbax Extend C 18 DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-3.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-4.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-5.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-6.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-7.D) 5. YMC Pack C 4 DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-35.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-36.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-37.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-38.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-39.D) stress, ph=6, 7 C, OS, 2 dagen 2 stress, ph=6, 7 C, OS, 2 dagen stress, ph=4, 8h daglicht stress, ph=4, 8h daglicht stress, ph=2, 8h daglicht stress, ph=2, 8h daglicht R28176: JFHE_15_77_1 R28176: JFHE_15_77_1 5 T T T T Zorbax Bonus RP DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-11.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-12.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-13.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-14.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-15.D) Waters Symmetry Shield RP 18 DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-43.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-44.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-45.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-46.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-47.D) stress, ph=6, 7 C, OS, 2 dagen stress, ph=6, 7 C, OS, 2 dagen stress, ph=4, 8h daglicht stress, ph=4, 8h daglicht stress, ph=2, 8h daglicht 5 stress, ph=2, 8h daglicht R28176: JFHE_15_77_1 5 R28176: JFHE_15_77_1 T T2475 T T Waters XTerra MS C 18 DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-19.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-2.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-21.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-22.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-23.D) 7. YMC Pro C 18 DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-51.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-52.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-53.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-54.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-55.D) stress, ph=6, 7 C, OS, 2 dagen stress, ph=6, 7 C, OS, 2 dagen stress, ph=4, 8h daglicht stress, ph=4, 8h daglicht stress, ph=2, 8h daglicht 5 stress, ph=2, 8h daglicht R28176: JFHE_15_77_1 T T R28176: JFHE_15_77_1 T T Waters XTerra RP 18 DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-27.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-28.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-29.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-3.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-31.D) 8. Alltech Platinum EPS C 18 DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-59.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-6.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-61.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-62.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-63.D) stress, ph=6, 7 C, OS, 2 dagen 5 stress, ph=4, 8h daglicht stress, ph=2, 8h daglicht R28176: JFHE_15_77_1 T T stress, ph=6, 7 C, OS, 2 dagen stress, ph=4, 8h daglicht stress, ph=2, 8h daglicht R28176: JFHE_15_77_1 T T
5 Selected ph and column DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-3.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-4.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-5.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-6.D) *DAD1 A, Sig=24,4 Ref=55, (G:\CQC-LC\DATA\MO\R28176\PH2.5\PH25-7.D) 25 2 stress, ph=6, 7 C, OS, 2 dagen stress, ph=4, 8h daglicht stress, ph=2, 8h daglicht R28176: JFHE_15_77_1 5 T T
6 DAD1 A, Sig=23,4 Ref=55, (G:\CQC-LC\DATA\MO\R184798\ D) VWD1 A, Wavelength=23 nm (G:\CQC-LC\DATA\MO\R184798\ D) VWD1 A, Wavelength=23 nm (G:\CQC-LC\DATA\MO\R184798\ D) VWD1 A, Wavelength=23 nm (G:\CQC-LC\DATA\MO\R184798\ D) VWD1 A, Wavelength=23 nm (G:\CQC-LC\DATA\MO\R184798\ D) VWD1 A, Wavelength=23 nm (G:\CQC-LC\DATA\MO\R184798\ D) Optimization A : 1 mm NH 4OAc +.5% DEA B : CH 3 CN A : 1 mm NH 4 OAc +.5% DEA B : CH 3CN - IPA (7/3) 95/5 to 4/6 in 2 1 ml/ 23 nm 95/5 to 4/6 in 2 1 ml/ 23 nm Once a suitable ph and column is selected generally other appropriate chromatographic parameters (gradient, organic modifiers, temperature, ionic strength.. ) are optimized step by step. A : 1 mm NH 4OAc +.5% DEA B : CH 3 CN - CH 3 OH (7/3) 95/5 to 4/6 in 2 1 ml/ 23 nm A : 1 mm NH 4 OAc +.5% DEA B : CH 3 CN - CH 3 OH - IPA (5/3/2) 95/5 to 5/5 in 2 1 ml/ 23 nm A : 1 mm NH 4 OAc +.5% DEA B : CH 3OH - IPA (7/3) 95/5 to 4/6 in 2 1 ml/ 23 nm 45 C 4 C 35 C A : 25 mm NH 4 OAc +.5% DEA B : CH 3CN - CH 3OH - IPA (5/3/2) 95/5 to 5/5 in 2 1 ml/ 23 nm LO CK ED METHOD Column W ate rs X T erra M S C 18; 3.5 µm; x 4.6 mm; (ph range: 1-12) Mobile Phase A : 1 mm NH 4 Ac in water +.5 % DEA B : acetonitrile/methanol/2-propanol: 5/3/2 v/v/v% E lution m ode linear gradient Time % A % B Flow 1. m l/m in. Temperature 35 C Detection 23 nm Injection volum e 2 µl Dilution solvent water 6
7 Major Roadblocks with currently used process Many chromatograms to evaluate manually - very complex - very time consug The main problem with this manual selection is the peak tracking between the obtained chromatograms. Currently this peak tracking is performed based on peak heights, peak shapes and PDA spectra YMC Pro C18 ph XTerra RP 18 C18 ph 7 Optimization is done step by step
8 New Approach The new approach is the on-line combination of four HPLC systems and one mass spectrometer with a computer modelling and simulation system. The mass spectrometer is installed with a MUX interface. In this way the related compounds can be immediately identified based on molecular mass. Summary reports will be automatically generated and transfered to a chromatographic method development software (DryLab or ACD). All important chromatographic parameters can be optimized with the assistance of the above described Method Development System. 8
9 Automated Screening and Method Optimization System 9
10 Installation MUX Source on Waters ZQ scan time:.13 sec inter - scan delay:.1 sec 1
11 Waves Method Variables 1 mm 25 mm Buffer Concentration WAVE 1 (screening) Optimum ph Best column Optimum buffer concentration 25 (35 ) 55 (65 ) Gradient WAVE 2-3 (optimization) ph ph 2.5 (NH 4 OAc + TFA) ph 4.8 (NH 4 OAc + CH 3 COOH) ph 7. (NH 4 OAc) ph 9. (NH 4 OAc + DEA) 64 experiments Columns Zorbax Extend C 18 Zorbax Bonus RP XTerra MS XTerra RP 18 XTerra Phenyl Symmetry Shield RP 18 YMC Pro C 18 YMC Buthyl C 4 Temperature 15 C 6 C 12 experiments Organic Modifier CH 3 CN CH 3 OH IPA THF with and without additive 11
12 Process Raw Data Yellow Box Integrate PDA BPI (MaxPlot) Define correlating MS base peak for each integrated PDA peak Generate a list of all masses found Reject false positives based on missing 13 C-isotope Tracking of all remaining masses with corresponding RT - Area - Peak Width Generate summary table 12
13 Integrate MaxPlot, define BPI and generate list masses found List masses found Example 1 Condition: 1mM ph 2.5 Retention Time: 9.79 Example 2 Condition: 1mM ph 2.5 Retention Time: 12.5 Example 1: MH (BPI) added to list Example 2: co-elution MH (BPI) added to list MH not added to list M.H
14 Integrate MaxPlot, define BPI and generate list masses found List masses found Example 3 Condition: 1mM ph 7 Retention Time: Example 3: MH added to list It is statistically significant that within 64 experiments all peaks are once separated and the corresponding masses are added to the list. M.H
15 OpenLynx : Define All Masses 15
16 Reject false positives Ghost-peaks, baseline fluctuations, blank peaks will also add a mass to the list but will be rejected afterwards based on the missing 13 C-isotope (marked yellow in the list). List masses found Example 4 Condition: 1mM ph 2.5 Retention Time: 3.97 Coctail 1 DE-AD 13-Dec-21 AO-395 1:38:45 COL1_1_25_S1 241 (4.43) Cm (238:243-(226: :258)) 1: Scan ES % m/z Example 4: MH (BPI) will be rejected M.H
17 Reprocess found masses The list of all masses is reprocessed by OpenLynx to obtain for each mass the corresponding RT, area and peak width in each experiment. Co-elutors can now easily be tracked. 17
18 Tracking of all masses found with corresponding RT - Area - Peak Width List masses found ph 2.5 ph 4.8 ph 7 ph 9 1 mm 1 mm 1 mm 1 mm M.H + RT Area Width RT Area Width RT Area Width RT Area Width The list of masses is now extended to a Summary Table 18
19 Summary Table List masses found ph 2.5 ph 4.8 ph 7 ph 9 1 mm 1 mm 1 mm 1 mm M.H + RT Area Width RT Area Width RT Area Width RT Area Width If a particular mass is only found in a few experiments (number set by user), that mass is statistically not relevant and will be rejected. (marked green in the Summary Table) If in the Summary Table two masses consistently co-elute over the whole number of experiments (retention time in two rows of the Summary Table match exactly), then the highest mass may be dropped. e.g. 13 C-isotope, 81 Brisotope, 37 Cl-isotope etc (marked blue in the Summary Table) 19
20 Optimization Software List masses found ph 2.5 ph 4.8 ph 7 ph 9 1 mm 1 mm 1 mm 1 mm M.H + RT Area Width RT Area Width RT Area Width RT Area Width The lean Summary Table will be transferred to a chromatographic method development software (DryLab or ACD) for prediction of the optimum in wave 1, 2 and 3. 2
21 Conclusions and Remarks Conclusions This screening and method development system will improve significantly the quality (reliability, robustness and cost reduction) of the methods and reduce the development time. Remarks Integrated peaks with no mass response are not covered. Compounds with no UV response but with mass response are not covered. Interference of non UV additives (cyclodextrines) can make the process of mass tracking more difficult. ZQ-MUX is exclusively used for peak tracking. The peaks not covered by the system can be investigated afterwards with high resolution LC-MS. 21
22 CONTACT INFORMATION Rudy Sneyers
Automated Method Development Utilizing Software-Based Optimization and Direct Instrument Control
 Automated Method Development Utilizing Software-Based Optimization and Direct Instrument Control Dr. Frank Steiner, 1 Andreas Brunner, 1 Fraser McLeod, 1 Dr. Sergey Galushko 2 1 Dionex Corporation, Germering,
Automated Method Development Utilizing Software-Based Optimization and Direct Instrument Control Dr. Frank Steiner, 1 Andreas Brunner, 1 Fraser McLeod, 1 Dr. Sergey Galushko 2 1 Dionex Corporation, Germering,
ANALYTICAL METHODS INTERNATIONAL QUALITY SYSTEMS
 VALIDATION OF ANALYTICAL METHODS 1 GERT BEUVING INTERNATIONAL PHARMACEUTICAL OPERATIONS TASKS: - Internal auditing - Auditing of suppliers and contract manufacturers - Preparing for and guiding of external
VALIDATION OF ANALYTICAL METHODS 1 GERT BEUVING INTERNATIONAL PHARMACEUTICAL OPERATIONS TASKS: - Internal auditing - Auditing of suppliers and contract manufacturers - Preparing for and guiding of external
# LCMS-35 esquire series. Application of LC/APCI Ion Trap Tandem Mass Spectrometry for the Multiresidue Analysis of Pesticides in Water
 Application Notes # LCMS-35 esquire series Application of LC/APCI Ion Trap Tandem Mass Spectrometry for the Multiresidue Analysis of Pesticides in Water An LC-APCI-MS/MS method using an ion trap system
Application Notes # LCMS-35 esquire series Application of LC/APCI Ion Trap Tandem Mass Spectrometry for the Multiresidue Analysis of Pesticides in Water An LC-APCI-MS/MS method using an ion trap system
Appendix 5 Overview of requirements in English
 Appendix 5 Overview of requirements in English This document is a translation of Appendix 4 (Bilag 4) section 2. This translation is meant as a service for the bidder and in case of any differences between
Appendix 5 Overview of requirements in English This document is a translation of Appendix 4 (Bilag 4) section 2. This translation is meant as a service for the bidder and in case of any differences between
Simultaneous Metabolite Identification and Quantitation with UV Data Integration Using LightSight Software Version 2.2
 Technical ote Simultaneous Metabolite Identification and Quantitation with UV Data Integration Using LightSight Software Version 2.2 Alek. Dooley, Carmai Seto, esham Ghobarah, and Elliott B. Jones verview:
Technical ote Simultaneous Metabolite Identification and Quantitation with UV Data Integration Using LightSight Software Version 2.2 Alek. Dooley, Carmai Seto, esham Ghobarah, and Elliott B. Jones verview:
Step-by-Step Analytical Methods Validation and Protocol in the Quality System Compliance Industry
 Step-by-Step Analytical Methods Validation and Protocol in the Quality System Compliance Industry BY GHULAM A. SHABIR Introduction Methods Validation: Establishing documented evidence that provides a high
Step-by-Step Analytical Methods Validation and Protocol in the Quality System Compliance Industry BY GHULAM A. SHABIR Introduction Methods Validation: Establishing documented evidence that provides a high
Reversed Phase High Presssure Liquid Chromatograhphic Technique for Determination of Sodium Alginate from Oral Suspension
 International Journal of PharmTech Research CODEN (USA): IJPRIF ISSN : 0974-4304 Vol.2, No.2, pp 1634-1638, April-June 2010 Reversed Phase High Presssure Liquid Chromatograhphic Technique for Determination
International Journal of PharmTech Research CODEN (USA): IJPRIF ISSN : 0974-4304 Vol.2, No.2, pp 1634-1638, April-June 2010 Reversed Phase High Presssure Liquid Chromatograhphic Technique for Determination
SIMULTANEOUS DETERMINATION OF TELMISARTAN AND HYDROCHLOROTHIAZIDE IN TABLET DOSAGE FORM USING REVERSE PHASE HIGH PERFORMANCE LIQUID CHROMATOGRAPHY
 CHAPTER 5 SIMULTANEOUS DETERMINATION OF TELMISARTAN AND HYDROCHLOROTHIAZIDE IN TABLET DOSAGE FORM USING REVERSE PHASE HIGH PERFORMANCE LIQUID CHROMATOGRAPHY CHAPTER 5 Simultaneous determination of Telmisartan
CHAPTER 5 SIMULTANEOUS DETERMINATION OF TELMISARTAN AND HYDROCHLOROTHIAZIDE IN TABLET DOSAGE FORM USING REVERSE PHASE HIGH PERFORMANCE LIQUID CHROMATOGRAPHY CHAPTER 5 Simultaneous determination of Telmisartan
GENERAL UNKNOWN SCREENING FOR DRUGS IN BIOLOGICAL SAMPLES BY LC/MS Luc Humbert1, Michel Lhermitte 1, Frederic Grisel 2 1
 GENERAL UNKNOWN SCREENING FOR DRUGS IN BIOLOGICAL SAMPLES BY LC/MS Luc Humbert, Michel Lhermitte, Frederic Grisel Laboratoire de Toxicologie & Génopathologie, CHRU Lille, France Waters Corporation, Guyancourt,
GENERAL UNKNOWN SCREENING FOR DRUGS IN BIOLOGICAL SAMPLES BY LC/MS Luc Humbert, Michel Lhermitte, Frederic Grisel Laboratoire de Toxicologie & Génopathologie, CHRU Lille, France Waters Corporation, Guyancourt,
LUMEFANTRINE Draft proposal for The International Pharmacopoeia (October 2006)
 October 2006 RESTRICTED LUMEFANTRINE Draft proposal for The International Pharmacopoeia (October 2006) DRAFT FOR DISCUSSION World Health Organization 2006 All rights reserved. This draft is intended for
October 2006 RESTRICTED LUMEFANTRINE Draft proposal for The International Pharmacopoeia (October 2006) DRAFT FOR DISCUSSION World Health Organization 2006 All rights reserved. This draft is intended for
ERDOSTEINE - MONOGRAPH.
 STRUCTURAL FORMULA (±1S-(2-[N-3-(2-oxotetrahydro thienyl)]acetamido)-thioglycolic acid) C 8 H 11 NO 4 S 2 M.W. = 249.307 DESCRIPTION Color : White to ivory white Appearance : Microcrystalline powder SOLUBILITY
STRUCTURAL FORMULA (±1S-(2-[N-3-(2-oxotetrahydro thienyl)]acetamido)-thioglycolic acid) C 8 H 11 NO 4 S 2 M.W. = 249.307 DESCRIPTION Color : White to ivory white Appearance : Microcrystalline powder SOLUBILITY
Overview. Purpose. Methods. Results
 A ovel Approach to Quantify Unbound Cisplatin, Carboplatin, and xaliplatin in Human Plasma Ultrafiltrate by Measuring Platinum-DDTC Complex Using LC/M/M Min Meng, Ryan Kuntz, Al Fontanet, and Patrick K.
A ovel Approach to Quantify Unbound Cisplatin, Carboplatin, and xaliplatin in Human Plasma Ultrafiltrate by Measuring Platinum-DDTC Complex Using LC/M/M Min Meng, Ryan Kuntz, Al Fontanet, and Patrick K.
How To Use An Acquity Qda Detector
 Mass-Directed Isolation of a Synthetic Peptide Using the ACQUITY QDa Detector Jo-Ann M. Jablonski and Andrew J. Aubin Waters Corporation, Milford, MA, USA APPLICATION BENEFITS The ACQUITY QDa Detector
Mass-Directed Isolation of a Synthetic Peptide Using the ACQUITY QDa Detector Jo-Ann M. Jablonski and Andrew J. Aubin Waters Corporation, Milford, MA, USA APPLICATION BENEFITS The ACQUITY QDa Detector
Journal of Chemical and Pharmaceutical Research
 Available on line www.jocpr.com Journal of Chemical and Pharmaceutical Research J. Chem. Pharm. Res., 2010, 2(1): 91-99 ISSN No: 0975-7384 A Stability-Indicating RP-LC method for the Determination of Related
Available on line www.jocpr.com Journal of Chemical and Pharmaceutical Research J. Chem. Pharm. Res., 2010, 2(1): 91-99 ISSN No: 0975-7384 A Stability-Indicating RP-LC method for the Determination of Related
UHPLC/MS: An Efficient Tool for Determination of Illicit Drugs
 Application Note: 439 UHPLC/MS: An Efficient Tool for Determination of Illicit Drugs Guifeng Jiang, Thermo Fisher Scientific, San Jose, CA, USA Key Words Accela UHPLC System MSQ Plus MS Detector Drugs
Application Note: 439 UHPLC/MS: An Efficient Tool for Determination of Illicit Drugs Guifeng Jiang, Thermo Fisher Scientific, San Jose, CA, USA Key Words Accela UHPLC System MSQ Plus MS Detector Drugs
Application of Structure-Based LC/MS Database Management for Forensic Analysis
 Cozette M. Cuppett and Michael P. Balogh Waters Corporation, Milford, MA USA Antony Williams, Vitaly Lashin and Ilya Troisky Advanced Chemistry Development, Toronto, ntario, Canada verview Application
Cozette M. Cuppett and Michael P. Balogh Waters Corporation, Milford, MA USA Antony Williams, Vitaly Lashin and Ilya Troisky Advanced Chemistry Development, Toronto, ntario, Canada verview Application
Daniel M. Mueller, Katharina M. Rentsch Institut für Klinische Chemie, Universitätsspital Zürich, CH-8091 Zürich, Schweiz
 Toxichem Krimtech 211;78(Special Issue):324 Online extraction LC-MS n method for the detection of drugs in urine, serum and heparinized plasma Daniel M. Mueller, Katharina M. Rentsch Institut für Klinische
Toxichem Krimtech 211;78(Special Issue):324 Online extraction LC-MS n method for the detection of drugs in urine, serum and heparinized plasma Daniel M. Mueller, Katharina M. Rentsch Institut für Klinische
Technical Report. Automatic Identification and Semi-quantitative Analysis of Psychotropic Drugs in Serum Using GC/MS Forensic Toxicological Database
 C146-E175A Technical Report Automatic Identification and Semi-quantitative Analysis of Psychotropic Drugs in Serum Using GC/MS Forensic Toxicological Database Hitoshi Tsuchihashi 1 Abstract: A sample consisting
C146-E175A Technical Report Automatic Identification and Semi-quantitative Analysis of Psychotropic Drugs in Serum Using GC/MS Forensic Toxicological Database Hitoshi Tsuchihashi 1 Abstract: A sample consisting
The Theory of HPLC. Gradient HPLC
 The Theory of HPLC Gradient HPLC i Wherever you see this symbol, it is important to access the on-line course as there is interactive material that cannot be fully shown in this reference manual. Aims
The Theory of HPLC Gradient HPLC i Wherever you see this symbol, it is important to access the on-line course as there is interactive material that cannot be fully shown in this reference manual. Aims
Overview. Introduction. AB SCIEX MPX -2 High Throughput TripleTOF 4600 LC/MS/MS System
 Investigating the use of the AB SCIEX TripleTOF 4600 LC/MS/MS System for High Throughput Screening of Synthetic Cannabinoids/Metabolites in Human Urine AB SCIEX MPX -2 High Throughput TripleTOF 4600 LC/MS/MS
Investigating the use of the AB SCIEX TripleTOF 4600 LC/MS/MS System for High Throughput Screening of Synthetic Cannabinoids/Metabolites in Human Urine AB SCIEX MPX -2 High Throughput TripleTOF 4600 LC/MS/MS
Application of a New Immobilization H/D Exchange Protocol: A Calmodulin Study
 Application of a New Immobilization H/D Exchange Protocol: A Calmodulin Study Jiang Zhao; Mei Zhu; Daryl E. Gilblin; Michael L. Gross Washington University Center for Biomrdical and Bioorganic Mass Spectrometry:
Application of a New Immobilization H/D Exchange Protocol: A Calmodulin Study Jiang Zhao; Mei Zhu; Daryl E. Gilblin; Michael L. Gross Washington University Center for Biomrdical and Bioorganic Mass Spectrometry:
Guide to Reverse Phase SpinColumns Chromatography for Sample Prep
 Guide to Reverse Phase SpinColumns Chromatography for Sample Prep www.harvardapparatus.com Contents Introduction...2-3 Modes of Separation...4-6 Spin Column Efficiency...7-8 Fast Protein Analysis...9 Specifications...10
Guide to Reverse Phase SpinColumns Chromatography for Sample Prep www.harvardapparatus.com Contents Introduction...2-3 Modes of Separation...4-6 Spin Column Efficiency...7-8 Fast Protein Analysis...9 Specifications...10
Henry Shion, Robert Birdsall, Steve Cubbedge, and Weibin Chen Waters Corporation, Milford, MA, USA APPLICATION BENEFITS INTRODUCTION WATERS SOLUTIONS
 Development of Integrated Informatics Workflows for the Automated Assessment of Comparability for Antibody Drug Conjugates (ADCs) Using LC-UV and LC-UV/MS Henry Shion, Robert Birdsall, Steve Cubbedge,
Development of Integrated Informatics Workflows for the Automated Assessment of Comparability for Antibody Drug Conjugates (ADCs) Using LC-UV and LC-UV/MS Henry Shion, Robert Birdsall, Steve Cubbedge,
Method development for analysis of formaldehyde in foodsimulant. melamine-ware by GC-MS and LC-MS/MS. Internal Technical Report
 of melamine-ware by GC-MS and LC-MS/MS Page 1 of 15 Method development for analysis of formaldehyde in foodsimulant extracts of melamine-ware by GC-MS and LC-MS/MS December 2012 Contact Point: Chris Hopley
of melamine-ware by GC-MS and LC-MS/MS Page 1 of 15 Method development for analysis of formaldehyde in foodsimulant extracts of melamine-ware by GC-MS and LC-MS/MS December 2012 Contact Point: Chris Hopley
Analytical Test Report
 Analytical Test Report Customer: Address (City, State): Purchase Order: Report Number: Project Number: Date Received: Date of Report: Test Location: Boulder, CO. Assay: Part Number: Amino Acids by HPLC
Analytical Test Report Customer: Address (City, State): Purchase Order: Report Number: Project Number: Date Received: Date of Report: Test Location: Boulder, CO. Assay: Part Number: Amino Acids by HPLC
Chemical analysis service, Turner s Green Technology Group
 Chemical analysis service, Turner s Green Technology Group Hourly costs, Academic collaborations leading to co-publications: 627 SEK/hr (excl. VAT) Hourly costs, Industry/Academic collaborations, no publications:
Chemical analysis service, Turner s Green Technology Group Hourly costs, Academic collaborations leading to co-publications: 627 SEK/hr (excl. VAT) Hourly costs, Industry/Academic collaborations, no publications:
HPLC Column Comparison Screening Study for Reversed Phase Columns
 HPLC Column Comparison Screening Study for Reversed Phase Columns Carmen T. Santasania, Eric M. Snyder, Matilal Sarker, and Tracy L. Ascah Supelco, 595 North Harrison Road, Bellefonte, PA 16823, USA T403058
HPLC Column Comparison Screening Study for Reversed Phase Columns Carmen T. Santasania, Eric M. Snyder, Matilal Sarker, and Tracy L. Ascah Supelco, 595 North Harrison Road, Bellefonte, PA 16823, USA T403058
Ultra Performance LC with Photo Diode Array Detection for the Development of Methods: Impurity Profiling and Long Term Stability Studies
 Ultra Performance LC with Photo Diode Array Detection for the Development of Methods: Impurity Profiling and Long Term Stability Studies Pittsburgh Conference Orlando, FL 14MAR06 Observations The pressure
Ultra Performance LC with Photo Diode Array Detection for the Development of Methods: Impurity Profiling and Long Term Stability Studies Pittsburgh Conference Orlando, FL 14MAR06 Observations The pressure
Alignment and Preprocessing for Data Analysis
 Alignment and Preprocessing for Data Analysis Preprocessing tools for chromatography Basics of alignment GC FID (D) data and issues PCA F Ratios GC MS (D) data and issues PCA F Ratios PARAFAC Piecewise
Alignment and Preprocessing for Data Analysis Preprocessing tools for chromatography Basics of alignment GC FID (D) data and issues PCA F Ratios GC MS (D) data and issues PCA F Ratios PARAFAC Piecewise
STANFORD UNIVERSITY MASS SPECTROMETRY 333 CAMPUS DR., MUDD 175 STANFORD, CA 94305-5080
 Training on the ZQ Open access MS Questions? Contact Dr. Allis Chien allis@stanford.edu 650-723-0710 0710 STANFORD UNIVERSITY MASS SPECTROMETRY STANFORD UNIVERSITY MASS SPECTROMETRY 333 CAMPUS DR., MUDD
Training on the ZQ Open access MS Questions? Contact Dr. Allis Chien allis@stanford.edu 650-723-0710 0710 STANFORD UNIVERSITY MASS SPECTROMETRY STANFORD UNIVERSITY MASS SPECTROMETRY 333 CAMPUS DR., MUDD
Electrospray Ion Trap Mass Spectrometry. Introduction
 Electrospray Ion Source Electrospray Ion Trap Mass Spectrometry Introduction The key to using MS for solutions is the ability to transfer your analytes into the vacuum of the mass spectrometer as ionic
Electrospray Ion Source Electrospray Ion Trap Mass Spectrometry Introduction The key to using MS for solutions is the ability to transfer your analytes into the vacuum of the mass spectrometer as ionic
Q-TOF User s Booklet. Enter your username and password to login. After you login, the data acquisition program will automatically start.
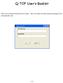 Q-TOF User s Booklet Enter your username and password to login. After you login, the data acquisition program will automatically start. ~ 1 ~ This data acquisition window will come up after the program
Q-TOF User s Booklet Enter your username and password to login. After you login, the data acquisition program will automatically start. ~ 1 ~ This data acquisition window will come up after the program
CONFIRMATION OF ZOLPIDEM BY LIQUID CHROMATOGRAPHY MASS SPECTROMETRY
 CONFIRMATION OF ZOLPIDEM BY LIQUID CHROMATOGRAPHY MASS SPECTROMETRY 9.1 POLICY This test method may be used to confirm the presence of zolpidem (ZOL), with diazepam-d 5 (DZP-d 5 ) internal standard, in
CONFIRMATION OF ZOLPIDEM BY LIQUID CHROMATOGRAPHY MASS SPECTROMETRY 9.1 POLICY This test method may be used to confirm the presence of zolpidem (ZOL), with diazepam-d 5 (DZP-d 5 ) internal standard, in
PVV Satyanaryana et al., IJSID, 2012, 2 (1), 226-231. International Journal of Science Innovations and Discoveries
 ISSN:2249-5347 IJSID International Journal of Science Innovations and Discoveries An International peer Review Journal for Science Research Article Available online through www.ijsidonline.info RP-HPLC
ISSN:2249-5347 IJSID International Journal of Science Innovations and Discoveries An International peer Review Journal for Science Research Article Available online through www.ijsidonline.info RP-HPLC
Advantages of Polar, Reversed- Phase HPLC Columns for the Analysis of Drug Metabolites
 Advantages of Polar, Reversed- Phase HPLC Columns for the Analysis of Drug Metabolites Carmen T. Santasania and David S. Bell SUPELCO, 595 North Harrison Road, Bellefonte, PA 16823 T404045 GUS Abstract
Advantages of Polar, Reversed- Phase HPLC Columns for the Analysis of Drug Metabolites Carmen T. Santasania and David S. Bell SUPELCO, 595 North Harrison Road, Bellefonte, PA 16823 T404045 GUS Abstract
Quality by Design Approach for the Separation of Naproxcinod and its Related Substances by Fused Core Particle Technology Column
 Journal of Chromatographic Science Advance Access published October 11, 2012 Journal of Chromatographic Science 2012;00:1 7 doi:10.1093/chromsci/bms162 Article Quality by Design Approach for the Separation
Journal of Chromatographic Science Advance Access published October 11, 2012 Journal of Chromatographic Science 2012;00:1 7 doi:10.1093/chromsci/bms162 Article Quality by Design Approach for the Separation
The Essential CHROMacademy Guide. Mobile Phase Optimization Strategies for Reversed Phase HPLC
 The Essential CHROMacademy Guide Mobile Phase Optimization Strategies for Reversed Phase HPLC Webcast Notes Type your questions in the Submit Question box, located below the slide window You can enlarge
The Essential CHROMacademy Guide Mobile Phase Optimization Strategies for Reversed Phase HPLC Webcast Notes Type your questions in the Submit Question box, located below the slide window You can enlarge
Chemistry 321, Experiment 8: Quantitation of caffeine from a beverage using gas chromatography
 Chemistry 321, Experiment 8: Quantitation of caffeine from a beverage using gas chromatography INTRODUCTION The analysis of soft drinks for caffeine was able to be performed using UV-Vis. The complex sample
Chemistry 321, Experiment 8: Quantitation of caffeine from a beverage using gas chromatography INTRODUCTION The analysis of soft drinks for caffeine was able to be performed using UV-Vis. The complex sample
HPLC Basics, Equipment, and Troubleshooting
 HPLC Basics, Equipment, and Troubleshooting HPLC Basics, Equipment, and Troubleshooting explains chromatography in practical terms from the ground up HPLC Basics, Equipment, and Troubleshooting HPLC Basics,
HPLC Basics, Equipment, and Troubleshooting HPLC Basics, Equipment, and Troubleshooting explains chromatography in practical terms from the ground up HPLC Basics, Equipment, and Troubleshooting HPLC Basics,
GC METHODS FOR QUANTITATIVE DETERMINATION OF BENZENE IN GASOLINE
 ACTA CHROMATOGRAPHICA, NO. 13, 2003 GC METHODS FOR QUANTITATIVE DETERMINATION OF BENZENE IN GASOLINE A. Pavlova and R. Ivanova Refining and Petrochemistry Institute, Analytical Department, Lukoil-Neftochim-Bourgas
ACTA CHROMATOGRAPHICA, NO. 13, 2003 GC METHODS FOR QUANTITATIVE DETERMINATION OF BENZENE IN GASOLINE A. Pavlova and R. Ivanova Refining and Petrochemistry Institute, Analytical Department, Lukoil-Neftochim-Bourgas
Development of validated RP- HPLC method for estimation of rivaroxaban in pharmaceutical formulation
 IJPAR Vol.4 Issue 4 Oct Dec - 2015 Journal Home page: ISSN: 2320-2831 Research Article Open Access Development of validated RP- HPLC method for estimation of rivaroxaban in pharmaceutical formulation V.
IJPAR Vol.4 Issue 4 Oct Dec - 2015 Journal Home page: ISSN: 2320-2831 Research Article Open Access Development of validated RP- HPLC method for estimation of rivaroxaban in pharmaceutical formulation V.
Guidance for Industry
 Guidance for Industry Q2B Validation of Analytical Procedures: Methodology November 1996 ICH Guidance for Industry Q2B Validation of Analytical Procedures: Methodology Additional copies are available from:
Guidance for Industry Q2B Validation of Analytical Procedures: Methodology November 1996 ICH Guidance for Industry Q2B Validation of Analytical Procedures: Methodology Additional copies are available from:
Gas Chromatography. Let s begin with an example problem: SPME head space analysis of pesticides in tea and follow-up analysis by high speed GC.
 Gas Chromatography Let s begin with an example problem: SPME head space analysis of pesticides in tea and follow-up analysis by high speed GC. Samples in 10mL sealed glass vials were placed in the MPS-2
Gas Chromatography Let s begin with an example problem: SPME head space analysis of pesticides in tea and follow-up analysis by high speed GC. Samples in 10mL sealed glass vials were placed in the MPS-2
Simultaneous qualitative and quantitative analysis using the Agilent 6540 Accurate-Mass Q-TOF
 Simultaneous qualitative and quantitative analysis using the Agilent 654 Accurate-Mass Q-TOF Technical Overview Authors Pat Perkins Anabel Fandino Lester Taylor Agilent Technologies, Inc. Santa Clara,
Simultaneous qualitative and quantitative analysis using the Agilent 654 Accurate-Mass Q-TOF Technical Overview Authors Pat Perkins Anabel Fandino Lester Taylor Agilent Technologies, Inc. Santa Clara,
Implementing New USP Chapters for Analytical Method Validation
 Implementing New USP Chapters for Analytical Method Validation March 2010 Ludwig Huber Fax.: +49 7243 602 501 E-mail: Ludwig_Huber@labcompliance.com Today s Agenda Handling Method Changes vs. Adjustments
Implementing New USP Chapters for Analytical Method Validation March 2010 Ludwig Huber Fax.: +49 7243 602 501 E-mail: Ludwig_Huber@labcompliance.com Today s Agenda Handling Method Changes vs. Adjustments
Standard Analytical Methods of Bioactive Metabolitesfrom Lonicera japonica Flower Buds by HPLC-DAD and HPLC-MS/MS
 Standard Analytical Methods of Bioactive Metabolitesfrom Lonicera japonica Flower Buds by HPLCDAD and HPLCMS/MS Jongheon Shin College of Pharmacy Seoul National University Summary The standard analytical
Standard Analytical Methods of Bioactive Metabolitesfrom Lonicera japonica Flower Buds by HPLCDAD and HPLCMS/MS Jongheon Shin College of Pharmacy Seoul National University Summary The standard analytical
A Complete Solution for Method Linearity in HPLC and UHPLC
 Now sold under the Thermo Scientific brand A Complete Solution for Method Linearity in HPLC and UHPLC Frank Steiner, Fraser McLeod, Tobias Fehrenbach, and Andreas Brunner Dionex Corporation, Germering,
Now sold under the Thermo Scientific brand A Complete Solution for Method Linearity in HPLC and UHPLC Frank Steiner, Fraser McLeod, Tobias Fehrenbach, and Andreas Brunner Dionex Corporation, Germering,
Application Note. Determination of Nitrite and Nitrate in Fruit Juices by UV Detection. Summary. Introduction. Experimental Sample Preparation
 Application Note Determination of Nitrite and Nitrate in Fruit Juices by UV Detection Category Food Matrix Fruit Juice Method HPLC Keywords Ion pair chromatography, fruit juice, inorganic anions AZURA
Application Note Determination of Nitrite and Nitrate in Fruit Juices by UV Detection Category Food Matrix Fruit Juice Method HPLC Keywords Ion pair chromatography, fruit juice, inorganic anions AZURA
Method Development for Size-Exclusion Chromatography of Monoclonal Antibodies and Higher Order Aggregates
 Method Development for Size-Exclusion Chromatography of Monoclonal Antibodies and Higher Order Aggregates Paula Hong and Kenneth J. Fountain Waters Corporation, 34 Maple St., Milford, MA, USA APPLICATION
Method Development for Size-Exclusion Chromatography of Monoclonal Antibodies and Higher Order Aggregates Paula Hong and Kenneth J. Fountain Waters Corporation, 34 Maple St., Milford, MA, USA APPLICATION
QUANTITATIVE AMINO ACID ANALYSIS. Aurélie Lolia Applications Manager, Biochrom Ltd
 QUANTITATIVE AMINO ACID ANALYSIS Aurélie Lolia Applications Manager, Biochrom Ltd QUANTITATIVE AMINO ACID ANALYSIS Principles of amino acid analysis Ion exchange chromatography The Biochrom 30 physiological
QUANTITATIVE AMINO ACID ANALYSIS Aurélie Lolia Applications Manager, Biochrom Ltd QUANTITATIVE AMINO ACID ANALYSIS Principles of amino acid analysis Ion exchange chromatography The Biochrom 30 physiological
Application Note # LCMS-92 Interlaboratory Tests Demonstrate the Robustness and Transferability of the Toxtyper Workflow
 Application Note # LCMS-92 Interlaboratory Tests Demonstrate the Robustness and Transferability of the Toxtyper Workflow Abstract There is high demand in clinical research and forensic toxicology for comprehensive,
Application Note # LCMS-92 Interlaboratory Tests Demonstrate the Robustness and Transferability of the Toxtyper Workflow Abstract There is high demand in clinical research and forensic toxicology for comprehensive,
LC-MS/MS Method for the Determination of Docetaxel in Human Serum for Clinical Research
 LC-MS/MS Method for the Determination of Docetaxel in Human Serum for Clinical Research J. Jones, J. Denbigh, Thermo Fisher Scientific, Runcorn, Cheshire, UK Application Note 20581 Key Words SPE, SOLA,
LC-MS/MS Method for the Determination of Docetaxel in Human Serum for Clinical Research J. Jones, J. Denbigh, Thermo Fisher Scientific, Runcorn, Cheshire, UK Application Note 20581 Key Words SPE, SOLA,
Turning HTE Agilent LC/MS Data to Knowledge in 1hr
 With permission Reaction Analytics Inc. 2011 Page 1 Turning HTE Agilent LC/MS Data to Knowledge in 1hr Neal Sach With permission Reaction Analytics Inc. 2011 Page 2 Contents Acquiring Data Fast demo method
With permission Reaction Analytics Inc. 2011 Page 1 Turning HTE Agilent LC/MS Data to Knowledge in 1hr Neal Sach With permission Reaction Analytics Inc. 2011 Page 2 Contents Acquiring Data Fast demo method
Supplementary Materials and Methods (Metabolomics analysis)
 Supplementary Materials and Methods (Metabolomics analysis) Metabolite extraction and mass spectrometry analysis. 12 Vldlr-/- knock out and 12 wild type (WT) retinas were separately frozen at -80 ºC in
Supplementary Materials and Methods (Metabolomics analysis) Metabolite extraction and mass spectrometry analysis. 12 Vldlr-/- knock out and 12 wild type (WT) retinas were separately frozen at -80 ºC in
Monoclonal Antibody Fragment Separation and Characterization Using Size Exclusion Chromatography Coupled with Mass Spectrometry
 Monoclonal ntibody Fragment Separation and haracterization Using Size Exclusion hromatography oupled with Mass Spectrometry uthors Haiying hen Katherine McLaughlin Sepax Technologies, Inc. 5 Innovation
Monoclonal ntibody Fragment Separation and haracterization Using Size Exclusion hromatography oupled with Mass Spectrometry uthors Haiying hen Katherine McLaughlin Sepax Technologies, Inc. 5 Innovation
Extraction of Epinephrine, Norepinephrine and Dopamine from Human Plasma Using EVOLUTE EXPRESS WCX Prior to LC-MS/MS Analysis
 Application Note AN844 Extraction of, and from Human Plasma Using EVOLUTE EXPRESS WCX Page 1 Extraction of, and from Human Plasma Using EVOLUTE EXPRESS WCX Prior to LC-MS/MS Analysis Introduction Catecholamines
Application Note AN844 Extraction of, and from Human Plasma Using EVOLUTE EXPRESS WCX Page 1 Extraction of, and from Human Plasma Using EVOLUTE EXPRESS WCX Prior to LC-MS/MS Analysis Introduction Catecholamines
DRAFT MONOGRAPH FOR THE INTERNATIONAL PHARMACOPOEIA PARACETAMOL ORAL SUSPENSION (September 2010)
 September 2010 RESTRICTED DRAFT MONOGRAPH FOR THE INTERNATIONAL PHARMACOPOEIA PARACETAMOL ORAL SUSPENSION (September 2010) DRAFT FOR COMMENTS This document was provided by a quality control expert and
September 2010 RESTRICTED DRAFT MONOGRAPH FOR THE INTERNATIONAL PHARMACOPOEIA PARACETAMOL ORAL SUSPENSION (September 2010) DRAFT FOR COMMENTS This document was provided by a quality control expert and
Materials for Pharmaceutical Manufacturing
 Materials for Pharmaceutical Manufacturing GRACE WHITEPAPER Vydac 150HC Purification Media Greater Loading Capacity and Resolution for Improved Productivity in Peptide Purification Technical Development:
Materials for Pharmaceutical Manufacturing GRACE WHITEPAPER Vydac 150HC Purification Media Greater Loading Capacity and Resolution for Improved Productivity in Peptide Purification Technical Development:
Thermo Scientific HyperSep Solid Phase Extraction Method Development Guide
 chromatography Thermo Scientific HyperSep Solid Phase Extraction Method Development Guide The following guide provides considerations, tips and general guidelines for developing SPE methods using the Thermo
chromatography Thermo Scientific HyperSep Solid Phase Extraction Method Development Guide The following guide provides considerations, tips and general guidelines for developing SPE methods using the Thermo
How To Test For Contamination In Large Volume Water
 Automated Solid Phase Extraction (SPE) of EPA Method 1694 for Pharmaceuticals and Personal Care Products in Large Volume Water Samples Keywords Application Note ENV0212 This collaboration study was performed
Automated Solid Phase Extraction (SPE) of EPA Method 1694 for Pharmaceuticals and Personal Care Products in Large Volume Water Samples Keywords Application Note ENV0212 This collaboration study was performed
Quantitative analysis of anabolic steroids in control samples from food-producing animals using a column-switching LC-HESI-MS/MS assay
 PO-CON1484E Quantitative analysis of anabolic steroids in control samples from food-producing animals using a column-switching ASMS 014 TP85 David R. Baker 1, John Warrander 1, Neil Loftus 1, Simon Hird
PO-CON1484E Quantitative analysis of anabolic steroids in control samples from food-producing animals using a column-switching ASMS 014 TP85 David R. Baker 1, John Warrander 1, Neil Loftus 1, Simon Hird
A High Throughput Automated Sample Preparation and Analysis Workflow for Comprehensive Forensic Toxicology Screening using LC/MS/MS
 A High Throughput Automated Sample Preparation and Analysis Workflow for Comprehensive Forensic Toxicology Screening using LC/MS/MS AB SCIEX QTRAP 4500 LC/MS/MS System and Gerstel, Inc. MultiPurpose Sampler
A High Throughput Automated Sample Preparation and Analysis Workflow for Comprehensive Forensic Toxicology Screening using LC/MS/MS AB SCIEX QTRAP 4500 LC/MS/MS System and Gerstel, Inc. MultiPurpose Sampler
Analyzing Small Molecules by EI and GC-MS. July 2014
 Analyzing Small Molecules by EI and GC-MS July 2014 Samples Appropriate for GC-MS Volatile and semi-volatile organic compounds Rule of thumb,
Analyzing Small Molecules by EI and GC-MS July 2014 Samples Appropriate for GC-MS Volatile and semi-volatile organic compounds Rule of thumb,
International GMP Requirements for Quality Control Laboratories and Recomendations for Implementation
 International GMP Requirements for Quality Control Laboratories and Recomendations for Implementation Ludwig Huber, Ph.D. ludwig_huber@labcompliance.com Overview GMP requirements for Quality Control laboratories
International GMP Requirements for Quality Control Laboratories and Recomendations for Implementation Ludwig Huber, Ph.D. ludwig_huber@labcompliance.com Overview GMP requirements for Quality Control laboratories
Improving the Metabolite Identification Process with Efficiency and Speed: LightSight Software for Metabolite Identification
 Improving the Metabolite Identification Process with Efficiency and Speed: LightSight Software for Metabolite Identification Overview LightSight Software for Metabolite Identification is a complete environment
Improving the Metabolite Identification Process with Efficiency and Speed: LightSight Software for Metabolite Identification Overview LightSight Software for Metabolite Identification is a complete environment
Ultra Fast UHPLC-LCMSMS Method Development in Clinical Drug Monitoring
 PO-CON1359E Ultra Fast UHPLC-LCMSMS Method Development in HPLC 2013 MASS-09 Anja Grüning 1 ; Brigitte Richrath 1 ; Klaus Bollig 2 ; Sven Vedder 1 ; Robert Ludwig 1 1 Shimadzu Europa GmbH, Duisburg, Germany;
PO-CON1359E Ultra Fast UHPLC-LCMSMS Method Development in HPLC 2013 MASS-09 Anja Grüning 1 ; Brigitte Richrath 1 ; Klaus Bollig 2 ; Sven Vedder 1 ; Robert Ludwig 1 1 Shimadzu Europa GmbH, Duisburg, Germany;
Transfer of a USP method for prednisolone from normal phase HPLC to SFC using the Agilent 1260 Infinity Hybrid SFC/UHPLC System Saving time and costs
 Agilent Application Solution Transfer of a USP method for prednisolone from normal phase PLC to SFC using the Agilent 126 Infinity ybrid SFC/UPLC System Saving time and costs Application Note Pharmaceutical
Agilent Application Solution Transfer of a USP method for prednisolone from normal phase PLC to SFC using the Agilent 126 Infinity ybrid SFC/UPLC System Saving time and costs Application Note Pharmaceutical
S olid Ph ase Extraction (SP E) Method:
 Solid Phase Extraction of US EPA 535 for Chloroacetanilide and Acetamide Degradates in Drinking Water Samples Prior to Liquid Chromatography/ Tandem Mass Spectrometry Toni Hofhine, Horizon Technology,
Solid Phase Extraction of US EPA 535 for Chloroacetanilide and Acetamide Degradates in Drinking Water Samples Prior to Liquid Chromatography/ Tandem Mass Spectrometry Toni Hofhine, Horizon Technology,
HPLC Analysis of Acetaminophen Tablets with Waters Alliance and Agilent Supplies
 HPLC Analysis of Acetaminophen Tablets with Waters Alliance and Agilent Supplies Application Note Small Molecule Pharmaceuticals Authors Jignesh Shah, Tiantian Li, and Anil Sharma Agilent Technologies,
HPLC Analysis of Acetaminophen Tablets with Waters Alliance and Agilent Supplies Application Note Small Molecule Pharmaceuticals Authors Jignesh Shah, Tiantian Li, and Anil Sharma Agilent Technologies,
AMD Analysis & Technology AG
 AMD Analysis & Technology AG Application Note 120419 Author: Karl-Heinz Maurer APCI-MS Trace Analysis of volatile organic compounds in ambient air A) Introduction Trace analysis of volatile organic compounds
AMD Analysis & Technology AG Application Note 120419 Author: Karl-Heinz Maurer APCI-MS Trace Analysis of volatile organic compounds in ambient air A) Introduction Trace analysis of volatile organic compounds
A feasibility study of the determination of B-group vitamins in food by HPLC with Mass Spectrometric detection. LGC/R/2011/181 LGC Limited 2011
 A feasibility study of the determination of B-group vitamins in food by HPLC with Mass Spectrometric detection LGC/R/2011/181 LGC Limited 2011 GC11/0005A A feasibility study of the determination of B-group
A feasibility study of the determination of B-group vitamins in food by HPLC with Mass Spectrometric detection LGC/R/2011/181 LGC Limited 2011 GC11/0005A A feasibility study of the determination of B-group
A NEW METHOD DEVELOPMENT AND VALIDATION FOR ANALYSIS OF RIVAROXABAN IN FORMULATION BY RP HPLC
 A NEW METHOD DEVELOPMENT AND VALIDATION FOR ANALYSIS OF RIVAROXABAN IN FORMULATION BY RP HPLC K.Chandra sekhar, P. Satya vani, A. Dhana lakshmi, Ch.LL.Devi, Anupama Barik, Narendra Devanaboyina * Department
A NEW METHOD DEVELOPMENT AND VALIDATION FOR ANALYSIS OF RIVAROXABAN IN FORMULATION BY RP HPLC K.Chandra sekhar, P. Satya vani, A. Dhana lakshmi, Ch.LL.Devi, Anupama Barik, Narendra Devanaboyina * Department
The Use of Micro Flow LC Coupled to MS/MS in Veterinary Drug Residue Analysis
 The Use of Micro Flow LC Coupled to MS/MS in Veterinary Drug Residue Analysis Stephen Lock AB SCIEX Warrington (UK) Overview A rapid, robust, sensitive and specific LC-MS/MS method has been developed for
The Use of Micro Flow LC Coupled to MS/MS in Veterinary Drug Residue Analysis Stephen Lock AB SCIEX Warrington (UK) Overview A rapid, robust, sensitive and specific LC-MS/MS method has been developed for
Pesticide Analysis by Mass Spectrometry
 Pesticide Analysis by Mass Spectrometry Purpose: The purpose of this assignment is to introduce concepts of mass spectrometry (MS) as they pertain to the qualitative and quantitative analysis of organochlorine
Pesticide Analysis by Mass Spectrometry Purpose: The purpose of this assignment is to introduce concepts of mass spectrometry (MS) as they pertain to the qualitative and quantitative analysis of organochlorine
Using Natural Products Application Solution with UNIFI for the Identification of Chemical Ingredients of Green Tea Extract
 Using Natural Products Application Solution with UNIFI for the Identification of Chemical Ingredients of Green Tea Extract Lirui Qiao, 1 Rob Lewis, 2 Alex Hooper, 2 James Morphet, 2 Xiaojie Tan, 1 Kate
Using Natural Products Application Solution with UNIFI for the Identification of Chemical Ingredients of Green Tea Extract Lirui Qiao, 1 Rob Lewis, 2 Alex Hooper, 2 James Morphet, 2 Xiaojie Tan, 1 Kate
Strength in Technology HPLC COLUMNS. Product Guide
 Strength in Technology HPLC COLUMNS Product Guide [ m V ] Cefadroxil 3 5 0 3 0 0 UniverSil C18 250x4.6mm 5µm Cefachlor Surface Area - 175m 2 /g 2 5 0 Pore Size - 140Å Voltage 2 0 0 Cefradine % Carbon -
Strength in Technology HPLC COLUMNS Product Guide [ m V ] Cefadroxil 3 5 0 3 0 0 UniverSil C18 250x4.6mm 5µm Cefachlor Surface Area - 175m 2 /g 2 5 0 Pore Size - 140Å Voltage 2 0 0 Cefradine % Carbon -
Overview. Triple quadrupole (MS/MS) systems provide in comparison to single quadrupole (MS) systems: Introduction
 Advantages of Using Triple Quadrupole over Single Quadrupole Mass Spectrometry to Quantify and Identify the Presence of Pesticides in Water and Soil Samples André Schreiber AB SCIEX Concord, Ontario (Canada)
Advantages of Using Triple Quadrupole over Single Quadrupole Mass Spectrometry to Quantify and Identify the Presence of Pesticides in Water and Soil Samples André Schreiber AB SCIEX Concord, Ontario (Canada)
VALIDATION OF ANALYTICAL PROCEDURES: TEXT AND METHODOLOGY Q2(R1)
 INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE VALIDATION OF ANALYTICAL PROCEDURES: TEXT AND METHODOLOGY
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE VALIDATION OF ANALYTICAL PROCEDURES: TEXT AND METHODOLOGY
Analysis of Phthalate Esters in Children's Toys Using GC-MS
 C146-E152 Analysis of Phthalate Esters in Children's Toys Using GC-MS GC/MS Technical Report No.4 Yuki Sakamoto, Katsuhiro Nakagawa, Haruhiko Miyagawa Abstract As of February 29, the US Consumer Product
C146-E152 Analysis of Phthalate Esters in Children's Toys Using GC-MS GC/MS Technical Report No.4 Yuki Sakamoto, Katsuhiro Nakagawa, Haruhiko Miyagawa Abstract As of February 29, the US Consumer Product
Thermo Scientific Vanquish UHPLC System. Revolutionize Your. UHPLC Experience
 Thermo Scientific Vanquish UHPLC System Revolutionize Your UHPLC Experience We feel that achieving better performance is not the same as spending more effort. We know that every breakthrough starts with
Thermo Scientific Vanquish UHPLC System Revolutionize Your UHPLC Experience We feel that achieving better performance is not the same as spending more effort. We know that every breakthrough starts with
SIMULTANEOUS DETERMINATION OF NALTREXONE AND 6- -NALTREXOL IN SERUM BY HPLC
 SIMULTANEOUS DETERMINATION OF NALTREXONE AND 6- -NALTREXOL IN SERUM BY HPLC Katja SÄRKKÄ, Kari ARINIEMI, Pirjo LILLSUNDE Laboratory of Substance Abuse, National Public Health Institute Manerheimintie,
SIMULTANEOUS DETERMINATION OF NALTREXONE AND 6- -NALTREXOL IN SERUM BY HPLC Katja SÄRKKÄ, Kari ARINIEMI, Pirjo LILLSUNDE Laboratory of Substance Abuse, National Public Health Institute Manerheimintie,
Introduction to HPLC. Basic Module in Bioorganic Chemistry January 16, 2007 Johannes Ranke. Introduction to HPLC p.1/17
 Introduction to HPLC Basic Module in Bioorganic Chemistry January 16, 2007 Johannes Ranke Introduction to HPLC p.1/17 Sources R. Kellner, J.-M. Mermet, M. Otto, H. M. Widmer (Eds.), Analytical Chemistry,
Introduction to HPLC Basic Module in Bioorganic Chemistry January 16, 2007 Johannes Ranke Introduction to HPLC p.1/17 Sources R. Kellner, J.-M. Mermet, M. Otto, H. M. Widmer (Eds.), Analytical Chemistry,
A Generic LC-MS Method for the Analysis of Multiple of Drug of Abuse Classes with the Thermo Scientific Exactive TM System
 A Generic LC-MS Method for the Analysis of Multiple of Drug of Abuse Classes with the Thermo Scientific Exactive TM System Kent Johnson Fortes lab, Wilsonville Oregon List of drug of abuse candidates for
A Generic LC-MS Method for the Analysis of Multiple of Drug of Abuse Classes with the Thermo Scientific Exactive TM System Kent Johnson Fortes lab, Wilsonville Oregon List of drug of abuse candidates for
GUIDELINES ON THE USE OF MASS SPECTROMETRY (MS) FOR IDENTIFICATION, CONFIRMATION AND QUANTITATIVE DETERMINATION OF RESIDUES CAC/GL 56-2005
 CAC/GL 56-2005 Page 1 of 6 GUIDELINES ON THE USE OF MASS SPECTROMETRY (MS) FOR IDENTIFICATION, CONFIRMATION AND QUANTITATIVE DETERMINATION OF RESIDUES CAC/GL 56-2005 CONFIRMATORY TESTS When analyses are
CAC/GL 56-2005 Page 1 of 6 GUIDELINES ON THE USE OF MASS SPECTROMETRY (MS) FOR IDENTIFICATION, CONFIRMATION AND QUANTITATIVE DETERMINATION OF RESIDUES CAC/GL 56-2005 CONFIRMATORY TESTS When analyses are
Strategies for Developing Optimal Synchronous SIM-Scan Acquisition Methods AutoSIM/Scan Setup and Rapid SIM. Technical Overview.
 Strategies for Developing Optimal Synchronous SIM-Scan Acquisition Methods AutoSIM/Scan Setup and Rapid SIM Technical Overview Introduction The 5975A and B series mass selective detectors (MSDs) provide
Strategies for Developing Optimal Synchronous SIM-Scan Acquisition Methods AutoSIM/Scan Setup and Rapid SIM Technical Overview Introduction The 5975A and B series mass selective detectors (MSDs) provide
The Scheduled MRM Algorithm Enables Intelligent Use of Retention Time During Multiple Reaction Monitoring
 The Scheduled MRM Algorithm Enables Intelligent Use of Retention Time During Multiple Reaction Monitoring Delivering up to 2500 MRM Transitions per LC Run Christie Hunter 1, Brigitte Simons 2 1 AB SCIEX,
The Scheduled MRM Algorithm Enables Intelligent Use of Retention Time During Multiple Reaction Monitoring Delivering up to 2500 MRM Transitions per LC Run Christie Hunter 1, Brigitte Simons 2 1 AB SCIEX,
Chapter 28: High-Performance Liquid Chromatography (HPLC)
 Chapter 28: High-Performance Liquid Chromatography (HPLC) Scope Instrumentation eluants, injectors, columns Modes of HPLC Partition chromatography Adsorption chromatography Ion chromatography Size exclusion
Chapter 28: High-Performance Liquid Chromatography (HPLC) Scope Instrumentation eluants, injectors, columns Modes of HPLC Partition chromatography Adsorption chromatography Ion chromatography Size exclusion
Online SPE-LC-APCI-MS/MS for the Determination of Steroidal Hormones in Drinking Water
 Online SPE-LC-APCI-MS/MS for the Determination of Steroidal ormones in Drinking Water presented by Paul Fayad, Ph.D. Candidate under the supervision of Sébastien Sauvé, Ph.D. Department of Chemistry paul.fayad@umontreal.ca
Online SPE-LC-APCI-MS/MS for the Determination of Steroidal ormones in Drinking Water presented by Paul Fayad, Ph.D. Candidate under the supervision of Sébastien Sauvé, Ph.D. Department of Chemistry paul.fayad@umontreal.ca
Determination of Food Dye Concentrations in an Unknown Aqueous Sample Using HPLC
 Determination of Food Dye Concentrations in an Unknown Aqueous Sample Using HPLC Abstract: High performance liquid chromatography (HPLC) was used to determine the identity and concentrations of food dyes
Determination of Food Dye Concentrations in an Unknown Aqueous Sample Using HPLC Abstract: High performance liquid chromatography (HPLC) was used to determine the identity and concentrations of food dyes
What Do We Learn about Hepatotoxicity Using Coumarin-Treated Rat Model?
 What Do We Learn about Hepatotoxicity Using Coumarin-Treated Rat Model? authors M. David Ho 1, Bob Xiong 1, S. Stellar 2, J. Proctor 2, J. Silva 2, H.K. Lim 2, Patrick Bennett 1, and Lily Li 1 Tandem Labs,
What Do We Learn about Hepatotoxicity Using Coumarin-Treated Rat Model? authors M. David Ho 1, Bob Xiong 1, S. Stellar 2, J. Proctor 2, J. Silva 2, H.K. Lim 2, Patrick Bennett 1, and Lily Li 1 Tandem Labs,
PURIFICATION. Preparative Chromatography Modular Solutions from Manual Systems to Full Automation. Scale Up. Reverse. Phase. Impurity Isolation
 PURIFICATION Preparative Chromatography Modular Solutions from Manual Systems to Full Automation PREPARATIVE CHROMATOGRAPHY SOLUTIONS Lab scale Scale Up Reverse Phase Medicinal Chemistry Natural Products
PURIFICATION Preparative Chromatography Modular Solutions from Manual Systems to Full Automation PREPARATIVE CHROMATOGRAPHY SOLUTIONS Lab scale Scale Up Reverse Phase Medicinal Chemistry Natural Products
Purification of reaction mixtures using flash chromatography.
 Purification of reaction mixtures using flash chromatography. This technical note details the use of ISOLUTE Flash chromatography columns for the purification of reaction mixtures. What is flash chromatography?
Purification of reaction mixtures using flash chromatography. This technical note details the use of ISOLUTE Flash chromatography columns for the purification of reaction mixtures. What is flash chromatography?
Analysis of Polyphenols in Fruit Juices Using ACQUITY UPLC H-Class with UV and MS Detection
 Analysis of Polyphenols in Fruit Juices Using ACQUITY UPLC H-Class with UV and MS Detection Evelyn Goh, Antonietta Gledhill Waters Pacific, Singapore, Waters Corporation, Manchester, UK A P P L I C AT
Analysis of Polyphenols in Fruit Juices Using ACQUITY UPLC H-Class with UV and MS Detection Evelyn Goh, Antonietta Gledhill Waters Pacific, Singapore, Waters Corporation, Manchester, UK A P P L I C AT
Hydrophilic-Interaction Chromatography (HILIC) for LC-MS/MS Analysis of Monoamine Neurotransmitters using XBridge BEH Amide XP Columns
 Hydrophilic-Interaction Chromatography (HILIC) for LC-MS/MS Analysis of Monoamine Neurotransmitters using XBridge BEH Amide XP Columns Jonathan P. Danaceau, Kenneth J. Fountain, and Erin E. Chambers Waters
Hydrophilic-Interaction Chromatography (HILIC) for LC-MS/MS Analysis of Monoamine Neurotransmitters using XBridge BEH Amide XP Columns Jonathan P. Danaceau, Kenneth J. Fountain, and Erin E. Chambers Waters
Smartline HPLC by design
 HPLC SMB Osmometry Smartline Systems Smartline HPLC by design Smartline high performance liquid chromatography instruments are designed to provide reliable separation solutions for routine tasks such as
HPLC SMB Osmometry Smartline Systems Smartline HPLC by design Smartline high performance liquid chromatography instruments are designed to provide reliable separation solutions for routine tasks such as
Develop a Quantitative Analytical Method for low (» 1 ppm) levels of Sulfate
 Cantaurus, Vol. 7, 5-8, May 1999 McPherson College Division of Science and Technology Develop a Quantitative Analytical Method for low (» 1 ppm) levels of Sulfate Janet Bowen ABSTRACT Sulfate is used in
Cantaurus, Vol. 7, 5-8, May 1999 McPherson College Division of Science and Technology Develop a Quantitative Analytical Method for low (» 1 ppm) levels of Sulfate Janet Bowen ABSTRACT Sulfate is used in
Waters Corporation. Waters 2690/5 USER & TROUBLESHOOTING GUIDE
 Waters Corporation Waters 2690/5 USER & TROUBLESHOOTING GUIDE Contents 2690/5 Theory Setup procedures. Troubleshooting the 2690/5 User maintenance of the 2690/5 Spare Parts 2 2690/5 Theory 2690/5 Solvent
Waters Corporation Waters 2690/5 USER & TROUBLESHOOTING GUIDE Contents 2690/5 Theory Setup procedures. Troubleshooting the 2690/5 User maintenance of the 2690/5 Spare Parts 2 2690/5 Theory 2690/5 Solvent
