OriginalArticle. Critical Parameters for the Isolation of Mesenchymal Stem Cells from Umbilical Cord Blood
|
|
|
- MargaretMargaret McCarthy
- 10 years ago
- Views:
Transcription
1 OriginalArticle Critical Parameters for the Isolation of Mesenchymal Stem Cells from Umbilical Cord Blood KAREN BIEBACK, SUSANNE KERN, HARALD KLÜTER, HERMANN EICHLER Institute of Transfusion Medicine and Immunology, German Red Cross Blood Service of Baden-Württemberg-Hessen, Germany; University of Heidelberg, Faculty of Clinical Medicine Mannheim, Germany Key Words. Mesenchymal stem cells Umbilical cord blood Isolation Multilineage differentiation ABSTRACT Evidence has emerged that mesenchymal stem cells (MSCs) represent a promising population for supporting new clinical concepts in cellular therapy. However, attempts to isolate MSCs from umbilical cord blood (UCB) of full-term deliveries have previously either failed or been characterized by a low yield. We investigated whether cells with MSC characteristics and multilineage differentiation potential can be cultivated from UCB of healthy newborns and whether yields might be maximized by optimal culture conditions or by defining UCB quality criteria. Using optimized isolation and culture conditions, in up to 63% of 59 low-volume UCB units, cells showing a characteristic mesenchymal morphology and immune phenotype (MSC-like cells) were isolated. These were similar to control MSCs from adult bone marrow (BM). The frequency of MSC-like cells ranged from 0 to 2.3 clones per mononuclear cells (MNCs). The cell clones proliferated extensively with at least 20 population doublings within eight passages. In addition, osteogenic and chondrogenic differentiation demonstrated a multilineage capacity comparable with BM MSCs. However, in contrast to MSCs, MSC-like cells showed a reduced sensitivity to undergo adipogenic differentiation. Crucial points to isolate MSC-like cells from UCB were a time from collection to isolation of less than 15 hours, a net volume of more than 33 ml, and an MNC count of more than MNCs. Because MSC-like cells can be isolated at high efficacy from full-term UCB donations, we regard UCB as an additional stem cell source for experimental and potentially clinical purposes. Stem Cells 2004;22: INTRODUCTION Mesenchymal stem cells (MSCs) comprise a rare population of multipotent progenitors capable of both supporting hematopoiesis and differentiating into at least the osteogenic, adipogenic, and chondrogenic lineages [1, 2]. These characteristics make MSCs very promising candidates to develop new cell-based therapeutic strategies, such as the treatment of mesenchymal tissue injuries or the supportive application in the context of hematopoietic stem cell (HSC) transplantation [3, 4]. Currently, bone marrow (BM) represents the main source of MSCs for both experimental and clinical studies [2 4]. As the number of MSCs and their differentiation capacity decline with age [5], their therapeutic potential might be diminished as well. Therefore, it can be argued that cells with Correspondence: Karen Bieback, Ph.D., Institute of Transfusion Medicine and Immunology, German Red Cross Blood Service of Baden-Württemberg-Hessen, Friedrich-Ebert-Strasse 107, D Mannheim, Germany. Telephone: ; Fax: ; [email protected] Received October 29, 2003; accepted for publication February 18, AlphaMed Press /2004/$12.00/0 STEM CELLS 2004;22:
2 626 Critical Parameters for Isolation of MSCs from UCB both extensive potency of proliferation and differentiation would represent an optimal tool for future cell-based therapeutic applications. Umbilical cord blood (UCB) has turned out to be an excellent alternative source of HSCs for clinical-scale allogeneic transplantation [6]. Essential preclinical studies proved a higher percentage of CD34 + CD38 cells in UCB compared with BM, suggesting that more primitive progenitors may be abundant in neonatal blood [7]. The same might apply for the presence of MSCs or progenitor cells. However, previous attempts to isolate MSCs from UCB have either failed [8 10] or have demonstrated a low frequency of mesenchymal progenitors in UCB of full-term deliveries [11, 12]. Therefore, the first goal of the present study was to verify whether cells with MSC traits can be isolated from fullterm UCB units. Second, those cells were to be compared with BM-derived MSCs to assess potential similarities or differences. In addition, we intended to optimize MSC isolation protocols to increase the efficacy towards levels that are applicable for therapeutic approaches by modifying cell culture conditions as well as by defining quality criteria for UCB units. MATERIALS AND METHODS Collection of UCB UCB units from full-term deliveries were collected from the unborn placenta with informed consent of the mothers [13]. Abag system containing 17 ml of citrate phosphate dextrose (CPD) anticoagulant within the collection bag was used (Cord Blood Collection System, Eltest, Bonn, Germany). The units were stored at 22 ± 4 C before processing. Isolation and Culture of Adherent Cells from UCB To isolate mononuclear cells (MNCs), each UCB unit was diluted 1:1 with phosphate-buffered saline (PBS)/2 mm EDTA (Nexell, Baxter, Unterschleißheim, Germany, www. baxter.de, and Merck, Darmstadt, Germany, com) and carefully loaded onto Ficoll-Hypaque solution (Amersham, Freiburg, Germany, amershambiosciences. com). After density gradient centrifugation at 435 g for 30 minutes at room temperature, MNCs were removed from the interphase and washed two to three times with PBS/EDTA. Cell counts were performed using an automated cell analyzer (Cell-Dyn 3200, Abbott, Wiesbaden, Germany, abbott.de). UCB-derived MNCs were set in culture at a density of /cm 2 into six-well culture plates (Falcon, Becton Dickinson, Heidelberg, Germany, in MSCGM medium (MSCGM BulletKit TM, CellSystems, St. Katharinen, Germany, For example, a UCB unit with MNCs was plated in 10 wells. Where specified, MesenCult was used as growth-promoting medium (Stem Cell Technologies, St. Katharinen, Germany, In contrast to MSCGM, which is based on Dulbecco s modified Eagle s medium (DMEM, low glucose) containing 10% fetal calf serum (FCS) from selected lots, MesenCult is based on McCoy s medium, also containing 10% FCS. After overnight incubation at 37 C in humidified atmosphere containing 5% carbon dioxide, nonadherent cells were removed and fresh medium was added to the wells. Cultures were maintained, and remaining nonadherent cells were removed by complete exchange of culture medium every 7 days. Culture wells were screened continuously to get hold of developing colonies of adherent cells. The precursor frequency of UCB-derived adherent fibroblastoid cells was calculated by counting the number of colonies per 10 8 MNCs. UCB units, which displayed no colony formation, were also considered for calculation. Fibroblastoid cells were recovered between days 16 through 20 after initial plating using 0.04% Trypsin/0.03% EDTA (PromoCell, Heidelberg, Germany, promocell.com). Recovered cells were replated at a density of 4,000 to 5,000 cells/cm 2 as passage 1 cells and thereafter. The population-doubling level was calculated for each subcultivation by using the following equation: population doublings = [log 10 (N H ) - log 10 (N 1 )]/log 10 (2), where N 1 is inoculum number and N H is cell harvest number [14]. The calculated population-doubling increase was added to the population-doubling levels of the previous passages to yield the cumulative population-doubling level. Monocyte Removal To prevent stable adherence of monocytic cells, MNC fractions from 48 UCB units were plated in six-well plates precoated with FCS [15]. In detail, culture plates were precoated with FCS (batches S0113/1038E and S0113/892E, Biochrom, Berlin, Germany, for 30 to 60 minutes at room temperature. After removing FCS, the plates were used immediately or stored at 4 C until further use. The MNCs were seeded as indicated above into the FCSprecoated plates. Nonadherent cells were harvested after overnight incubation. To calculate the effect of FCS coating on the adherence of monocytes, we assessed the recovery of monocytes by comparing the percentage of monocytes in the MNC fraction before and after plating by flow cytometry. The cells were labeled with CD14-FITC (Becton Dickinson) and CD45-PerCP (Beckman Coulter). To compare the effect of
3 Bieback, Kern, Klüter et al. 627 FCS coating on the remaining adherent cells, photomicrographs were taken every week and the number of adherent cells was determined. Osteoclast-like cells and fibroblastoid cells were discriminated microscopically, and the number of wells with any of these types of cells was counted. Collection and Isolation of Control MSCs from BM BM served as positive control and was obtained from the femoral shaft of patients undergoing total hip replacement at the Orthopedic Department of the University Hospital Mannheim. Cells were aspirated into a 5-ml syringe containing CPD anticoagulant. In total, six specimens from female patients were obtained, with the donor age ranging from 68 to 84 years. To isolate MSCs from BM, the aspirate was diluted 1:5 and processed as described above. In contrast to MNCs from UCB, BM-derived MNCs were cultured at a density of cells/cm 2 in T75 culture flasks (Nunc, Wiesbaden, Germany, and the first change of medium was performed 3 days after initial plating. Two weeks later, at reaching 80% 90% confluence, MSCs were suspended and replated as described for the UCB-derived adherent cells. Primary Fibroblasts as Controls Primary normal human dermal fibroblasts (PromoCell) served as negative control in the differentiation studies. The cells were maintained in supplemented fibroblast growth medium (PromoCell). Immune Phenotypic Analyses Efforts to define a distinct phenotype characteristic for MSC have been confounded by the fact that these cells can express a range of cell lineage specific antigens [16]. To analyze cell-surface expression of typical marker proteins, UCB- and BM-derived adherent cells, each at fourth passage, were labelled with the following anti-human antibodies: CD14-FITC, CD34-PE, CD73-PE (also referred to as SH3 and SH4 [2, 16]), CD90-Cy5 (Becton Dickinson), CD29-PE, CD44-FITC, CD45-PerCP, HLA class I FITC, HLA class II FITC (Beckman Coulter, Krefeld, Germany, CD133-PE (Miltenyi Biotech, Bergisch Gladbach, Germany, CD105-FITC (SH2 [2, 16]), and CD106-PE (Immunokontakt, AMS Biotechnology, Wiesbaden, Germany, Mouse isotype antibodies served as respective controls (Becton Dickinson and Beckman Coulter). Ten thousand labelled cells were acquired and analyzed using a FACScan flow cytometer running CellQuest software (Becton Dickinson). Differentiation Studies Osteogenic Differentiation To induce osteogenic differentiation, the cells were seeded at a density of cells/cm 2 and cultured in eight-chamber slides (Nunc) until they reached approximately 80% confluence. Additional culture was performed in osteogenic differentiation medium supplemented with hmsc Osteogenic SingleQuots TM containing 0.1 µm dexamethasone, 10 mm β- glycerophosphate, 0.05 mm ascorbate, and 10% FCS (Cell- Systems) [17]. The onset of osteoblast formation was evaluated after 3 weeks by both the expression of alkaline phosphatase and by calcium accumulation. Activity of alkaline phosphatase was detected histologically, as described recently (Leukocyte Alkaline Phosphatase-Kit, Sigma Diagnostics, Taufkirchen, Germany, [2]. The accumulation of mineralized calcium phosphate was assessed by von Kossa staining after the protocol from Cheng et al. [18] with few modifications. The cells were fixed for 15 minutes in 10% formalin (Sigma), and after washing they were incubated with 5% silver nitrate (Sigma) for 15 to 30 minutes. Pyrogallol 1% (Merck) and sodium thiosulfate 5% (Sigma) were used to develop and fix the signal. Adipogenic Differentiation The adipogenic differentiation was induced by cyclic changes of adipogenic induction and maintenance medium (CellSystems) in cells grown postconfluently in eight-chamber slides, following the protocol of Pittenger et al. [2]. The induction medium, containing 1 µm dexamethasone, 0.5 mm 3-isobutyl-1-methyl-xanthine, 10 µg/ml recombinant human (rh) insulin, 0.2 mm indomethacin, and 10% FCS, was added for 2 to 3 days to the culture chambers. It was then replaced by maintenance medium containing only rh insulin and 10% FCS. Control cells were kept in adipogenic maintenance medium. After three cycles of changing the two media, cultures were maintained for 1 additional week. The induction of adipogenic differentiation was apparent by intracellular accumulation of lipid-rich vacuoles that stained with Oil Red O (Fluka, Taufkirchen, Germany, The cells were fixed with 10% formalin, washed, and stained with a working solution of 0.18% Oil Red O for 5 minutes. Chondrogenic Differentiation To promote chondrogenic differentiation, cells were gently centrifuged ( 150g, 5 minutes) in a 15-ml polypropylene tube (Greiner) to form a pellet according to the protocol of Mackay et al. [19]. Without disturbing the pellet, the cells were cultured for 4 weeks in complete chondrogenic differentiation medium (CellSystems) including 10
4 628 Critical Parameters for Isolation of MSCs from UCB ng/ml TGFβ3 (Strathmann Biotec AG, Hamburg, Germany, by feeding twice a week. After the culture period, cryosections were analyzed by Safranin O staining. The sections were fixed with ice-cold acetone (Sigma) and stained with 0.1% aqueous Safranin O solution (Sigma). Cell nuclei were counterstained with Weigert s iron hematoxylin (Sigma). Statistical Analysis Data were presented as mean ± standard deviation (SD). Two-sided paired Student s t-test was used to compare the mean values of cell percentages and numbers in wells coated with FCS and in wells left uncoated. Two-sided nonpaired t- test was used to analyze the flow cytometry data. Chi-square (χ 2 ) tests were used to compare the frequency distributions according to differing quality parameters of UCB units. Differences were considered significant at p <.05. The SPSS software package was used for all statistical analyses (Chicago, RESULTS Generation of Primary Cultures from Adherent UCB Cells In total, 59 UCB units entered this study and were analyzed regarding their capacity to generate cultures of MSCs. After plating the MNCs, only a few cells attached to the plastic culture dishes and formed adherent cells within 3 weeks. Most of those cells were monocytes, which fused to form osteoclast-like cells [20]. These appeared in 80% 90% of the wells uncoated with FCS and reached a subconfluent condition within 3 to 4 weeks, as shown in Figure 1A. Rare events displayed fibroblastoid cells, which expanded rapidly (MSC-like cells). In total, we obtained 29 colonies in 17 of the 59 processed UCB units. The onset of colony formation could be observed at first after 14 to 16 days. Cell confluence was reached after approximately 20 days of culture. In contrast, BM-derived MNCs formed clusters of spindle-shaped MSCs within 5 to 10 days and cell confluence after 2 to 3 weeks. But the UCB-derived MSC-like cells were of clonal origin, because at most one colony developed per well. Therefore, the mean (± SD) frequency of MSClike cells was calculated from all tested UCB units to be 0.7 ± 0.2 clone-forming units per MNCs of full-term UCB, ranging from 0 to 2.3 per MNCs. In units that gave rise to MSC-like cells, the frequency was variable and ranged from 0.2 to 2.3 clones per MNCs (mean, 1.1 ± 0.2). Twenty-two UCB-derived clonal cell populations were spindle-shaped, but the other seven were more spherical (Figs. 1B 1D). However, they maintained this phenotype even in between subsequent passages (Figs. 1D 1F). Most clones could be cultured for at least eight passages and approximately 20 population doublings (Fig. 1G) (maximum up to now passage 12, with approximately 27 population doublings, n = 3), showing neither changed morphology nor reduced proliferation (n = 21). In contrast, all BM-derived MSCs displayed a change in morphology and markedly reduced proliferation already after approximately five population doublings (passages 3 through 4), as shown in Figures 1G 1I. Characterization of MSC-Like Cells from UCB Immune Phenotype The expression of cell-surface antigens by flow cytometry was evaluated on 12 clones of MSC-like cells and 4 BMderived MSC preparations, each at passage 4. Neither the cells derived from UCB nor from BM expressed the hematopoietic markers CD14, CD34 and CD45, and CD133 (Fig. 2). Similar to MSCs from BM, UCB-derived clones were strongly positive for CD29, CD44, and CD73. In addition, cells from both sources stained positive for HLA-class I and negative for HLA-class II. However, only 53.6 ± 4.3% of the UCB-derived cells expressed CD105 (also known as SH2) compared with 84.4 ± 2.8% of BM MSC (p =.02). In contrast, 72.4 ± 3.2% of the UCB cells stained positive for CD106 compared with 42.4 ± 6.5% of BM MSC (p =.04). In addition, although most UCB-derived and BM-derived cells expressed CD90 (95.4% and 99.9%, respectively), there was a significant difference in the intensity of antigen expression. The MSC-like cells displayed a mean fluorescence intensity of ± 186.5, whereas BM MSCs showed a significantly higher expression with ± (p =.03). Differentiation Potential The differentiation potential of all 29 UCB-derived clones was compared with BM MSCs. Cells from passage 1 or 2 were cultured under conditions that are favorable for an osteogenic, adipogenic, or chondrogenic differentiation, respectively. First, osteogenic differentiation was induced in all BM MSC preparations and all MSC-like cell clones but not in fibroblasts (not shown), as shown by increased activity of alkaline phosphatase (Figs. 3A 3D) and enhanced mineralization defined by von Kossa staining (Figs. 3E 3H). This capacity was retained in all analyzed UCB clones (n = 8, passages 5 and 7). Second, the adipogenic induction was apparent in MSCs from BM by the cellular accumulation of lipid-rich vacuoles that stained with Oil Red O (Fig. 3L). However, the fibroblasts (not shown) and, most strikingly, all 29 MSC-like cell clones never generated adipocytes under standard differenti-
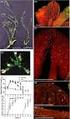
5 Bieback, Kern, Klüter et al. 629 A B C D E F Cumulative population doublings G H I Figure 1. Morphology of UCB-derived adherent cells displaying an osteoclast-like or fibroblastoid morphology. (A): Morphology of UCB-derived osteoclast-like cells in primary culture at day 16. UCB-derived fibroblastoid cells were identified as distinct colonies at days 14 through 16 after initial plating. They displayed either a consistent spindle-shaped (B, D) or a more spherical, less spindleshaped morphology (C). The clone shown in (D) maintained a homogenous phenotype until passage 8 or 19.3 population doublings. (D): The colony at passage 0, day 16 before split. (F, G): The same clone at passage 3, day 37 (4 days after split, approximately eight population doublings) and passage 8, day 90 (4 days after split, 19.3 population doublings). BM-derived MSCs are shown as controls in (H) and (I), with (H) representing cells at passage 0, reaching a confluent stage at day 14. The MSCs depicted in (I) present an altered morphology and a stop in proliferation already at passage 4 (day 31, 7 days after split, 4.5 population doublings). These are representative examples of 29 clones of UCB-derived fibroblastoid cells and six processed BM aspirates, shown at 100 magnification. (G): Mean values of the cumulative population doublings, determined at each subcultivation, with UCB-derived cells shown in black (n = 12) and BM-derived MSCs shown in gray (n = 5). Abbreviations: BM, bone marrow; MSC, mesenchymal stem cell; UCB, umbilical cord blood. ation conditions. Adipogenic differentiation could solely be induced in MSC-like cells cultured continuously in adipogenic induction medium for at least 5 weeks (Fig. 3J). Finally, in chondrogenic differentiation assays, MSClike cells, BM MSC, and fibroblasts consolidated within 1 day, forming aggregates that dislodged to float freely in the suspension culture. Cryosections of the aggregates stained with Safranin O showed a condensed structure with chondrocyte-like lacunae (Figs. 3M and 3N, fibroblasts not shown). Also, UCB-derived MSC-like cells at later passages maintained the ability to form cartilage (n = 8). Optimization of Culture Conditions for the Isolation of MSC-Like Cells Initially, the frequency of MSC-like cells and the percentage of UCB units generating MSC-like cells was quite low, with one of eight processed UCB units (12.5%). Thus, we intended to improve the efficacy at first by modulating the culture con-
6 630 Critical Parameters for Isolation of MSCs from UCB Figure 2. Immune phenotype of MSC-like cells from UCB and BM MSCs. Fibroblastoid cells from UCB and BM at passage 4 were trypsinized, labeled with antibodies against the indicated antigens, and analyzed by flow cytometry. The respective isotype control is shown as a dotted line. Arepresentative example of 12 UCB clones ( ) and 4 BM harvests ( ) is shown.values represent the mean percentage of all assessed cells positively stained by the respective antibodies in the flow cytometry analyses. Abbreviations: BM, bone marrow; MSC, mesenchymal stem cell; UCB, umbilical cord blood. ditions. Because no fibroblastoid cells could be visualized in wells showing osteoclast-like cells, our first approach was to prevent the adherence of monocytes as osteoclast precursors. To dispose the monocytes, we cultured the MNC in FCScoated wells and removed the nonadherent cells after overnight adherence. Analyzing six UCB units in parallel by plating one half of the MNC fraction in FCS-precoated wells and the other half in uncoated wells, we observed a significantly higher percentage of monocytes harvested in the nonadherent fraction after seeding MNCs in FCS-coated wells (Table 1). This indicated that fewer monocytes adhered to the coated plates than to the uncoated plates. In addition, significantly fewer adherent cells and osteoclast-like cells were achieved in the wells precoated with FCS. In consequence, we received four colonies of MSC-like cells from three units plated in FCS-precoated wells compared with two colonies from two units seeded in uncoated wells (Table 1). Therefore, although the impact of FCS coating on displaying MSCs was not significant in those six units, we proceeded using precoated wells for the isolation and gained 12 units with 22 MSC-like cell clones out of 48 UCB units. In a second approach, we tested different commercially available media known to promote the growth of MSCs from BM. Using MSCGM, based on DMEM, 29 colonies of MSC-like cells could be generated from 53 UCB units. Using MesenCult, based on McCoys, we succeeded in isolating MSCs from BM (n = 4) but not from UCB (n = 6; p =.04). Criteria for UCB Units for Efficient Isolation of MSC-Like Cells Even after adjusting the culture conditions to optimal levels, we were only able to isolate MSC-like cells from one third of the processed UCB units (Table 2). To establish criteria to predict an efficient isolation, we analyzed various quality aspects of the processed UCB units. Because it has been shown that cord blood from preterm
7 Bieback, Kern, Klüter et al. 631 UCB BM A B C D E F G H I J K L M N Figure 3. Differentiation capacity of MSC-like cells from UCB and MSCs from BM. Cultured cells from UCB-derived clones and BM harvests were exposed in vitro to differentiation medium to induce osteogenic, adipogenic, and chondrogenic differentiation, respectively. UCB-derived fibroblastoid cells were shown to differentiate appropriately to the osteogenic lineage by enhancement of alkaline phosphatase activity (B) and calcium mineralization detected by von Kossa stain (F) [(A) and (E) show noninduced controls]. Correspondingly, the BM MSCs used as a control are shown in (C), (D), (G), and (H) (magnification 100). Adipocytes could solely be detected by Oil Red O stain in the adipogenic cultures of UCB MSC-like cells after intense induction for at least 5 weeks [(I): not induced; (J): induced]. In contrast, BM MSC-derived adipocytes could easily be induced by cyclic changes of adipogenic induction and maintenance medium for 3 weeks. [(K): not induced; (L): induced; magnification 200). Chondrogenesis was shown by Safranin O staining in cryosections from both the UCB-derived clonal cells in (M) and the BM MSC in (N) (magnification 100). Arepresentative example of 29 UCB clones (five for adipogenic differentiation) and six BM harvests at passage 1 is shown. Abbreviations: BM, bone marrow; MSC, mesenchymal stem cell; UCB, umbilical cord blood. deliveries can contain MSCs [11], we tried to correlate the presence of MSCs with either the gestational age or the mode of delivery. The gestational age ranged from 37 to 42 weeks, with a mean of 39.6 ± 1.5 weeks. Forty out of 59 processed units (67.8%) were derived from vaginal deliveries, and 19 were from cesarean sections. However, the presence of MSC-like cell clones correlated neither with the gestational age (p =.50) nor with the kind of delivery (p =.38). However, both the time from collection to isolation and the volume of the UCB units were crucial for MSC-like cell generation (Table 2). Units stored for more than 15 hours failed to establish any MSC-like cells (n = 15). The mean
8 632 Critical Parameters for Isolation of MSCs from UCB Table 1. Effect of FCS coating on the percentage of monocytes in the nonadherent fraction after plating and on the amount of adherent osteoclast-like cells and mesenchymal stem cell (MSC) like cells by plating one half of the mononuclear cells in coated and the other half in uncoated plates Fetal calf Uncoated serum coated plates plates p value Monocytes (%) a 81.9 ± ± Adherent cells b Wk ± 9.7 (2 23) 8.5 ± 8.4 (1 20).86 Wk ± 1.4 (0 4) 9.0 ± 7.4 (0 22).03 Wk ± 0.5 (0 1) 13.8 ± 8.9 (3 24).05 Osteoclast-like cells c (U/U) 1/6 3/6.12 MSC-like cells (U/U) 3/6 (4 colonies) 2/6 (2 colonies).56 n = 6; mean storage time of umbilical cord blood units, 10.7 ± 3.3 hours; range, 6 to 18 hours. p values were calculated using paired Student s t-test and c2 analysis. a Mean percentage of monocytes recovered in the supernatant containing the nonadherent cells after overnight adherence. b Mean value of cells, counted on three photomicrographs at a time. Photographs were taken at the respective time intervals. Range is indicated in parentheses. c Osteoclast-like cells appeared in 70% 80% of the culture wells. storage time of all processed UCB units was 10.2 ± 5.7 hours, ranging from 3 to 22 hours. Furthermore, a net volume of more than 33 ml placental blood and a MNC count greater than were critical for isolation. Mean net volume was 42.8 ± 17.2 ml (range, 13 to 108 ml), and mean MNC count was 1.5 ± (range, 0.3 to 4.5). Three of the processed UCB units showed signs of coagulation, and six units showed signs of hemolysis. These units failed to generate MSC-like cells. By taking into account only those units having optimal quality, we were able to enhance the success rate from 29% to 63% (Table 2). DISCUSSION The expected potential of mesenchymal progenitors is a matter of paramount importance for upcoming therapeutic strategies in the context of cellular therapy. However, although the biological characteristics of MSCs have been subject to intense investigation over the past few years, there is presently no universally accepted definition of this type of stem cell. Despite the lack of a definition, BM-derived MSCs have already been used in various preclinical and clinical studies [1 4]. However, the use of adult tissue-derived mesenchymal cells might not always be acceptable because of a significant drop in cell number and proliferative/differentiation capacity with age [5]. Therefore, UCB might result in an alternative for MSCs that is as good as has been proven for HSCs. Although the evidence for the isolation of fibroblastoid cells with MSC characteristics from UCB is conflicting, our data reveal that MSC-like cells can be isolated from full-term UCB units with an efficiency of greater than 60%. Crucial parameters for achieving this yield were a storage time of less than 15 hours, a net volume of more than 33 ml, an MNC count greater than MNCs, and no coagula or signs of hemolysis. It is tempting to speculate that using UCB units in line with blood-banking requirements will result in even higher yield in efficiency, which may intensify their use in therapeutic approaches. The identification of MSC-like cells in UCB is in line with previously published results [11, 12]. On the other hand, other authors have denied the presence of MSC in UCB and claimed that only hematopoietic cells can be isolated from UCB [8 10]. Indeed, even in our experiments, a high percentage of UCB-derived cultures gave rise to monocytederived osteoclast-like cells. However, using FCS-coated culture plates enabled us to remove myeloid cells and osteoclast-like cells as their progeny. Besides, because it is known that FCS is crucial for the growth of MSCs [2], coated FCS might provide additional growth and adherence factors, favoring the generation of MSC-like cells from UCB. The frequency of UCB-derived MSC-like cells was extremely low compared with BM. On the other hand, the clonal-derived MSC-like cells were able to generate much more progeny than the BM-derived cultures. MSC-like cells were fibroblastoid and expressed a typical set of MSC marker proteins, namely CD29, CD44, CD73, CD105, CD106, and HLA-class I, while lacking hematopoietic markers similar to BM-derived MSC. However, the expression of CD90, CD105, and CD106 differed between the
9 Bieback, Kern, Klüter et al. 633 Table 2. Success rate in isolating mesenchymal stem cell (MSC) like cells after recalculating umbilical cord blood (UCB) units fulfilling special criteria Critical parameters Success rate p value Total UCB units 17/59 (29 clones) 28.8% UCB units 15 h 17/44 (29 clones) 38.9%.03 UCB units 33 ml 17/47 (29 clones) 36.2%.03 UCB units 10 8 MNC 16/40 (27 clones) 40.0%.01 UCB units 15 h, 33 ml, /38 (29 clones) 44.7%.01 UCB units of optimal quality a 17/27 (29 clones) 63.0%.01 Data represent the ratio of units that gave rise to MSC-like cells to the total number of units exploiting special characteristics. p values were calculated by χ 2 analysis comparing the frequency distributions of units according to critical quality parameters. a 15 hours, 33 ml, and 10 8 MNC, fetal calf serum coat, no coagula, and no hemolysis, respectively. two. Because at least CD106 expressed on MSC is functionally associated with hematopoiesis [21], the expression intensities may be related to the tissue the cells are derived from. Just like BM MSCs, the MSC-like cells were also capable of differentiating towards the osteogenic and chondrogenic lineages. However, in contrast to MSCs, the MSC-like cells needed specific culture conditions to differentiate into the adipogenic lineage. The clonal cell isolation and the greater number of cell doublings may have caused an altered responsiveness in the MSC-like cells towards adipogenic induction, as indicated by Campagnoli et al. [22] and Goodwin et al. [12]. Furthermore, various publications have shown that BM-derived MSC populations are heterogeneous and that in multipotential clones the adipogenic potential is the first to be repressed [23]. On the other hand, the differing sensitivity to undergo adipogenic differentiation may also represent an inherent difference between the two cell populations. Because adipocytes, which are characteristic for adult human BM, are absent in fetal BM, the potential of MSCs to become adipocytes may be gained with aging to either occupy excess space or support hematopoiesis [24]. Therefore, the reduced sensitivity of MSC-like cells compared with BM-derived cells may be related to the ontogenetic age of the donors. An open question is whether the MSC-like cells circulate or originate from the placenta. MSCs were isolated from first-trimester fetal blood and liver [22] and second-trimester fetal lung samples [25]. In addition, UCB of preterm deliveries seems to contain MSCs in relevant concentrations [11, 12]. However, we isolated MSC-like cells from more than 60% of the full-term UCB units. Because of the preparative regime of the blood harvest, those might be derived from the subendothelial layer of the cord, which has been shown to be an alternative source for MSCs [26]. However, if the presence of MSC-like cells was the result of lesions of the cord, contaminating endothelial cells within the cultures also should have been observed in high frequency in our experiments. But this was not the case. To sum up, UCB can be used not only as an alternative source for hematopoietic cells but also for MSCs. MSC-like cells herein are characterized by their ability to proliferate in culture with an attached spindle-shaped morphology, by the presence of a consistent set of surface markers, and by their differentiation into at least three mesenchymal lineages under controlled in vitro conditions. The capacity of MSCs to support hematopoiesis [1] and their potential to regulate immune responses [27] would commend their use in cotransplants to enhance the engraftment rate and quality of hematopoietic stem cells. Because hematopoietic stem cells as well as MSCs can be isolated from the same UCB, autologous cotransplantation may prove to be optimal for UCB transplantation approaches. ACKNOWLEDGMENTS We gratefully acknowledge the collaborators in the obstetric departments in Mannheim, especially Dudi Vuthaj, Dr. J. Stöve from the Orthopedic Department of the University Hospital Mannheim for supplying BM harvests, and Daniela Griffiths for editing the manuscript. This work was supported by Forschungsfonds der Fakultät für Klinische Medizin Mannheim. Karen Bieback and Susanne Kern contributed equally to the results.
10 634 Critical Parameters for Isolation of MSCs from UCB REFERENCES 1 Majumdar MK, Thiede MA, Mosca JD et al. Phenotypic and functional comparison of cultures of marrow-derived mesenchymal stem cells (MSCs) and stromal cells. J Cell Physiol 1998;176: Pittenger MF, Mackay AM, Beck CB et al. Multilineage potential of adult human mesenchymal stem cells. Science 1999;284: Horwitz EM, Prockop DJ, Fitzpatrick LA et al. Transplantability and therapeutic effects of bone marrow-derived mesenchymal cells in children with osteogenesis imperfecta. Nat Med 1999;5: Koc ON, Gerson SL, Cooper BW et al. Rapid hematopoietic recovery after coinfusion of autologous-blood stem cells and culture-expanded marrow mesenchymal stem cells in advanced breast cancer patients receiving high-dose chemotherapy. J Clin Oncol 2000;18: Mueller SM, Glowacki J. Age-related decline in the osteogenic potential of human bone marrow cells cultured in three-dimensional collagen sponges. J Cell Biochem 2001;82: Grewal SS, Barker JN, Davies SM et al. Unrelated donor hematopoietic cell transplantation: marrow or umbilical cord blood? Blood 2003;101: Broxmeyer HE, Douglas GW, Hangoc G et al. Human umbilical cord blood as a potential source of transplantable hematopoietic stem/progenitor cells. Proc Natl Acad Sci U S A1989;86: Gutiérrez-Rodríguez M, Reyes-Maldonado E, Mayani H. Characterization of the adherent cells developed in Dextertype long-term cultures from human umbilical cord blood. STEM CELLS 2000;18: Mareschi K, Biasin E, Piacibello W et al. Isolation of human mesenchymal stem cells: bone marrow versus umbilical cord blood. Haematologica 2001;86: Wexler SA, Donaldson C, Denning-Kendall P et al. Adult bone marrow is a rich source of human mesenchymal stem cells but umbilical cord and mobilized adult blood are not. Br J Haematol 2003;121: Erices A, Conget P, Minguell JJ. Mesenchymal progenitor cells in human umbilical cord blood. Br J Haematol 2000; 109: Goodwin HS, Bicknese AR, Chien SN et al. Multilineage differentiation activity by cells isolated from umbilical cord blood: expression of bone, fat, and neural markers. Biol Blood Marrow Transplant 2001;7: Eichler H, Richter E, Leveringhaus Aet al. The Mannheim cord blood project: experience in collection and processing of the first 880 banked unrelated cord blood transplants. Infusionsther Transfusionsmed 1999;26: Cristofalo VJ, Allen RG, Pignolo RJ et al. Relationship between donor age and the replicative lifespan of human cells in culture: a reevaluation. Proc Natl Acad Sci U S A 1998;95: Nielsen H. Isolation and functional activity of human blood monocytes at adherence to plastic surfaces: comparison of different detachment methods. Acta Pathol Microbiol Immunol Scand 1987;95: Haynesworth SE, Baber MA, Caplan AI. Cell surface antigens on human marrow-derived mesenchymal cells are detected by monoclonal antibodies. Bone 1992;13: Jaiswal N, Haynesworth SE, Caplan AI et al. Osteogenic differentiation of purified, culture-expanded human mesenchymal stem cells in vitro. J Cell Biochem 1997;64: Cheng SL, Yang JW, Rifas L et al. Differentiation of human bone marrow osteogenic stromal cells in vitro: induction of the osteoblast phenotype by dexamethasone. Endocrinology 1994;134: Mackay AM, Beck SC, Murphy JM et al. Chondrogenic differentiation of cultured human mesenchymal stem cells from marrow. Tissue Eng 1998;4: Roux S, Quinn J, Pichaud F et al. Human cord blood monocytes undergo terminal osteoclast differentiation in vitro in the presence of culture medium conditioned by giant cell tumor of bone. J Cell Physiol 1996;168: Levesque JP, Takamatsu Y, Nilsson SK et al. Vascular cell adhesion molecule-1 (CD106) is cleaved by neutrophil proteases in the bone marrow following hematopoietic progenitor cell mobilization by granulocyte colony-stimulating factor. Blood 2001;98: Campagnoli C, Roberts IA, Kumar S et al. Identification of mesenchymal stem/progenitor cells in human firsttrimester fetal blood, liver, and bone marrow. Blood 2001; 98: Muraglia A, Cancedda R, Quarto R. Clonal mesenchymal progenitors from human bone marrow differentiate in vitro according to a hierarchical model. J Cell Sci 2000;113: Gimble JM, Robinson CE, Wu X et al. The function of adipocytes in the bone marrow stroma: an update. Bone 1996; 19: Noort WA, Kruisselbrink AB, in't Anker PS et al. Mesenchymal stem cells promote engraftment of human umbilical cord blood-derived CD34 + cells in NOD/SCID mice. Exp Hematol 2002;30: Romanov YA, Svintsitskaya VA, Smirnov VN. Searching for alternative sources of postnatal human mesenchymal stem cells: candidate MSC-like cells from umbilical cord. STEM CELLS 2003;21: Bartholomew A, Sturgeon C, Siatskas M et al. Mesenchymal stem cells suppress lymphocyte proliferation in vitro and prolong skin graft survival in vivo. Exp Hematol 2002;30:42 48.
Basic Science in Medicine
 Medical Journal of th e Islamic Republic of Iran Volume 18 Number 3 Fall 1383 November 2004 Basic Science in Medicine ] EXPANSION OF HUMAN CORD BLOOD PRIMITIVE PROGENITORS IN SERUM-FREE MEDIA USING HUMAN
Medical Journal of th e Islamic Republic of Iran Volume 18 Number 3 Fall 1383 November 2004 Basic Science in Medicine ] EXPANSION OF HUMAN CORD BLOOD PRIMITIVE PROGENITORS IN SERUM-FREE MEDIA USING HUMAN
Human Umbilical Cord-derived Multipotent Mesenchymal Stromal Cells (huc-msc) Handling Instructions
 Human Umbilical Cord-derived Multipotent Mesenchymal Stromal Cells (huc-msc) Order No.: 19401-005, 19401-010 Handling Instructions For preclinical ex vivo use. Not intended for therapeutic use. Table of
Human Umbilical Cord-derived Multipotent Mesenchymal Stromal Cells (huc-msc) Order No.: 19401-005, 19401-010 Handling Instructions For preclinical ex vivo use. Not intended for therapeutic use. Table of
HUMAN MESENCHYMAL STEM CELLS AS BASIC TOOLS FOR TISSUE ENGINEERING: ISOLATION AND CULTURE
 HUMAN MESENCHYMAL STEM CELLS AS BASIC TOOLS FOR TISSUE ENGINEERING: ISOLATION AND CULTURE MONICA NEAGU*, ERIKA SUCIU**, VALENTIN ORDODI**, VIRGIL PĂUNESCU** *Department of Biophysics and Medical Informatics,
HUMAN MESENCHYMAL STEM CELLS AS BASIC TOOLS FOR TISSUE ENGINEERING: ISOLATION AND CULTURE MONICA NEAGU*, ERIKA SUCIU**, VALENTIN ORDODI**, VIRGIL PĂUNESCU** *Department of Biophysics and Medical Informatics,
BioResearch. Hematopoietic and Immune Cell Products Essential Tools for Hematopoietic Research
 BioResearch Hematopoietic and Immune Cell Products Essential Tools for Hematopoietic Research BioResearch Hematopoietic and Immune Cell Products Essential Tools for Hematopoietic Research Working with
BioResearch Hematopoietic and Immune Cell Products Essential Tools for Hematopoietic Research BioResearch Hematopoietic and Immune Cell Products Essential Tools for Hematopoietic Research Working with
Equine Stem Cells. Sources, Processing, Expansion, Storage and Shipping
 Equine Stem Cells Sources, Processing, Expansion, Storage and Shipping Sean Owens, DVM, Diplomate ACVP Medical Director, UC Davis Veterinary Regenerative Medicine Laboratory What is a Stem Cell and What
Equine Stem Cells Sources, Processing, Expansion, Storage and Shipping Sean Owens, DVM, Diplomate ACVP Medical Director, UC Davis Veterinary Regenerative Medicine Laboratory What is a Stem Cell and What
3D Cell Culture mimsys G
 3D Cell Culture mimsys G xeno-free & nutrient permeable hydrogel for 3D cell culture mimsys G is a xeno-free and non-immunogenic, easy to handle hydrogel for cell encapsulation in 3D experiments in vitro
3D Cell Culture mimsys G xeno-free & nutrient permeable hydrogel for 3D cell culture mimsys G is a xeno-free and non-immunogenic, easy to handle hydrogel for cell encapsulation in 3D experiments in vitro
Comparison of Mesenchymal Stem Cells derived from Amniotic Fluid and Umbilical Cord
 Academy of Sustainable Development and Technology. Comparison of Mesenchymal Stem Cells derived from Amniotic Fluid and Umbilical Cord Master of Science Thesis (30hp) Flemmingsberg, Sweden 2011 Åsa Ekblad
Academy of Sustainable Development and Technology. Comparison of Mesenchymal Stem Cells derived from Amniotic Fluid and Umbilical Cord Master of Science Thesis (30hp) Flemmingsberg, Sweden 2011 Åsa Ekblad
Chondrogenical Differentiation of Umbilical Cord Lining. Stem Cells
 Chondrogenical Differentiation of Umbilical Cord Lining Stem Cells He Xi, Masilamani Jeyakumar, Phan Toan Thang Bioengineering, National University of Bioengineering Abstract Cartilage defects, such as
Chondrogenical Differentiation of Umbilical Cord Lining Stem Cells He Xi, Masilamani Jeyakumar, Phan Toan Thang Bioengineering, National University of Bioengineering Abstract Cartilage defects, such as
Poietics human mesenchymal stem cells Instructions for use
 Poietics human mesenchymal stem cells Instructions for use www.lonza.com U.S. Scientific Support: 800-521-0390 [email protected] EU/ROW Scientific Support: +49-221-99199-400 [email protected]
Poietics human mesenchymal stem cells Instructions for use www.lonza.com U.S. Scientific Support: 800-521-0390 [email protected] EU/ROW Scientific Support: +49-221-99199-400 [email protected]
Minimal residual disease detection in Acute Myeloid Leukaemia on a Becton Dickinson flow cytometer
 Minimal residual disease detection in Acute Myeloid Leukaemia on a Becton Dickinson flow cytometer Purpose This procedure gives instruction on minimal residual disease (MRD) detection in patients with
Minimal residual disease detection in Acute Myeloid Leukaemia on a Becton Dickinson flow cytometer Purpose This procedure gives instruction on minimal residual disease (MRD) detection in patients with
Mesenchymal Stem Cells
 Mesenchymal Stem Cells Instruction Manual Product Size Catalog Number from Bone Marrow (hmsc-bm) from Umbilical Cord Matrix (hmsc-uc) from Adipose Tissue (hmsc-at) 500,000 cryopreserved cells 500,000 proliferating
Mesenchymal Stem Cells Instruction Manual Product Size Catalog Number from Bone Marrow (hmsc-bm) from Umbilical Cord Matrix (hmsc-uc) from Adipose Tissue (hmsc-at) 500,000 cryopreserved cells 500,000 proliferating
How To Expand Hematopoietic Stem Cells
 Purification and Expansion of Hematopoietic Stem Cells Based on Proteins Expressed by a Novel Stromal Cell Population Our bodies are constantly killing old, nonfunctional, and unneeded cells and making
Purification and Expansion of Hematopoietic Stem Cells Based on Proteins Expressed by a Novel Stromal Cell Population Our bodies are constantly killing old, nonfunctional, and unneeded cells and making
Mesenchymal progenitor cells in human umbilical cord blood
 British Journal of Haematology 2000, 109, 235±242 Mesenchymal progenitor cells in human umbilical cord blood ALEJANDRO ERICES, 1 PAULETTE CONGET 1 and JOSEÂ J. MINGUELL 1,2 1 Unidad de BiologõÂa Celular,
British Journal of Haematology 2000, 109, 235±242 Mesenchymal progenitor cells in human umbilical cord blood ALEJANDRO ERICES, 1 PAULETTE CONGET 1 and JOSEÂ J. MINGUELL 1,2 1 Unidad de BiologõÂa Celular,
Cord derived MSC-Like Placenta derived Membranes- Matrix- Cells- MSC-like,
 Bank Public/Pvt Bank other cells Ship and distribute Collect and process other samples Support development of novel uses One cord Blood Unit Non stem cells RBC s Plateletes T B Monocytes NK cells Stem
Bank Public/Pvt Bank other cells Ship and distribute Collect and process other samples Support development of novel uses One cord Blood Unit Non stem cells RBC s Plateletes T B Monocytes NK cells Stem
Human Umbilical Cord Blood CD34 + Progenitor Cell Care Manual
 Human Umbilical Cord Blood CD34 + Progenitor Cell Care Manual INSTRUCTION MANUAL ZBM0065.03 SHIPPING CONDITIONS Human Umbilical Cord Blood CD34+ Progenitor Cells, cryopreserved Cryopreserved human umbilical
Human Umbilical Cord Blood CD34 + Progenitor Cell Care Manual INSTRUCTION MANUAL ZBM0065.03 SHIPPING CONDITIONS Human Umbilical Cord Blood CD34+ Progenitor Cells, cryopreserved Cryopreserved human umbilical
Processing & Utilization of Cord Blood for Transplant
 Processing & Utilization of Cord Blood for Transplant 2010-Jan Jan-15 Nicole L. Prokopishyn, PhD HPC Processing Laboratory Director Calgary Laboratory Services Overview Cord Blood Processing Pre-Freeze
Processing & Utilization of Cord Blood for Transplant 2010-Jan Jan-15 Nicole L. Prokopishyn, PhD HPC Processing Laboratory Director Calgary Laboratory Services Overview Cord Blood Processing Pre-Freeze
The Therapeutic Potential of Human Umbilical Cord Blood Transplantation for Neonatal Hypoxic-Ischemic Brain Injury and Ischemic Stroke
 The Therapeutic Potential of Human Umbilical Cord Blood Transplantation for Neonatal Hypoxic-Ischemic Brain Injury and Ischemic Stroke a,b* b,c a a b b b b a b a b c 430 Wang et al. Acta Med. Okayama Vol.
The Therapeutic Potential of Human Umbilical Cord Blood Transplantation for Neonatal Hypoxic-Ischemic Brain Injury and Ischemic Stroke a,b* b,c a a b b b b a b a b c 430 Wang et al. Acta Med. Okayama Vol.
低 剂 量 辐 射 对 脐 血 T 淋 巴 细 胞 膜 分 子 表 达 的 影 响 EFFECT OF LOW DOSE IRRADIATION ON EXPRESSION OF MEMBRANE MOLECULES OF T LYMPHOCYTES IN CORD BLOOD
 CNIC-01513 PUTH-0001 低 剂 量 辐 射 对 脐 血 T 淋 巴 细 胞 膜 分 子 表 达 的 影 响 EFFECT OF LOW DOSE IRRADIATION ON EXPRESSION OF MEMBRANE MOLECULES OF T LYMPHOCYTES IN CORD BLOOD 中 国 核 情 报 中 心 China Nuclear Information
CNIC-01513 PUTH-0001 低 剂 量 辐 射 对 脐 血 T 淋 巴 细 胞 膜 分 子 表 达 的 影 响 EFFECT OF LOW DOSE IRRADIATION ON EXPRESSION OF MEMBRANE MOLECULES OF T LYMPHOCYTES IN CORD BLOOD 中 国 核 情 报 中 心 China Nuclear Information
PROTOCOL. Immunostaining for Flow Cytometry. Background. Materials and equipment required.
 PROTOCOL Immunostaining for Flow Cytometry 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 Rev.0 Background The combination of single cell analysis using flow cytometry and the specificity of antibody-based
PROTOCOL Immunostaining for Flow Cytometry 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 Rev.0 Background The combination of single cell analysis using flow cytometry and the specificity of antibody-based
Cord Blood Re-imagined The use of cord blood stem cells for clinical applications and medical innovations
 GE Healthcare Proceedings of the Symposium Cord Blood Re-imagined The use of cord blood stem cells for clinical applications and medical innovations 12 th Annual Meeting of the International Society for
GE Healthcare Proceedings of the Symposium Cord Blood Re-imagined The use of cord blood stem cells for clinical applications and medical innovations 12 th Annual Meeting of the International Society for
PROTOCOL. Immunocytochemistry (ICC) MATERIALS AND EQUIPMENT REQUIRED
 PROTOCOL Immunocytochemistry (ICC) 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 11-07 MATERIALS AND EQUIPMENT REQUIRED Materials: MitoSciences primary monoclonal antibody/antibodies Fluorophore-conjugated
PROTOCOL Immunocytochemistry (ICC) 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 11-07 MATERIALS AND EQUIPMENT REQUIRED Materials: MitoSciences primary monoclonal antibody/antibodies Fluorophore-conjugated
A Public Cord Blood Bank for South Africa? i
 No. 42/2007 A Public Cord Blood Bank for South Africa? i By Dr Robert Crookes MBChB (Wits), Dip. Internal Medicine (American Board of Internal Medicine, USA) Transfusion Medicine Consultant. South African
No. 42/2007 A Public Cord Blood Bank for South Africa? i By Dr Robert Crookes MBChB (Wits), Dip. Internal Medicine (American Board of Internal Medicine, USA) Transfusion Medicine Consultant. South African
Osteoblast Differentiation and Mineralization
 Osteoblast Differentiation and Mineralization Application Note Background Osteoblasts are specialized fibroblasts that secrete and mineralize the bone matrix. They develop from mesenchymal precursors.
Osteoblast Differentiation and Mineralization Application Note Background Osteoblasts are specialized fibroblasts that secrete and mineralize the bone matrix. They develop from mesenchymal precursors.
CORD BLOOD BANKING FAQ
 CORD BLOOD BANKING FAQ Cord Blood & Stem Cells Q: What is umbilical cord blood (UCB)? A: Bone marrow, peripheral blood and UCB constitute the three primary sources of stem cells. Cord blood, which, until
CORD BLOOD BANKING FAQ Cord Blood & Stem Cells Q: What is umbilical cord blood (UCB)? A: Bone marrow, peripheral blood and UCB constitute the three primary sources of stem cells. Cord blood, which, until
HARVESTING AND CRYOPRESERVATION OF HUMAN EMBRYONIC STEM CELLS (hescs)
 HARVESTING AND CRYOPRESERVATION OF HUMAN EMBRYONIC STEM CELLS (hescs) OBJECTIVE: can be cryopreserved in a liquid nitrogen (LN 2 ) freezer for long-term storage. This Standard Operating Procedure (SOP)
HARVESTING AND CRYOPRESERVATION OF HUMAN EMBRYONIC STEM CELLS (hescs) OBJECTIVE: can be cryopreserved in a liquid nitrogen (LN 2 ) freezer for long-term storage. This Standard Operating Procedure (SOP)
The immunomodulatory activity of human umbilical cord bloodderived mesenchymal stem cells in vitro
 IMMUNOLOGY ORIGINAL ARTICLE The immunomodulatory activity of human umbilical cord bloodderived mesenchymal stem cells in vitro Meng Wang, 1 Yuan Yang, 2 Dongming Yang, 1 Fei Luo, 1 Wenjie Liang, 1 Shuquan
IMMUNOLOGY ORIGINAL ARTICLE The immunomodulatory activity of human umbilical cord bloodderived mesenchymal stem cells in vitro Meng Wang, 1 Yuan Yang, 2 Dongming Yang, 1 Fei Luo, 1 Wenjie Liang, 1 Shuquan
Review. Cord blood as an alternative source of haemopoietic stem cells. J. Hows, A. Nicol, P. Denning-Kendall, C. Donaldson, M. Nieda & B.
 Annals of Oncology 7 (Suppi 2): 47-51, 1996. O 1996 Kluwer Academic Publishers. Printed in the Netherlands. Review Cord blood as an alternative source of haemopoietic stem cells J. Hows, A. Nicol, P. Denning-Kendall,
Annals of Oncology 7 (Suppi 2): 47-51, 1996. O 1996 Kluwer Academic Publishers. Printed in the Netherlands. Review Cord blood as an alternative source of haemopoietic stem cells J. Hows, A. Nicol, P. Denning-Kendall,
Current Issues in Stem Cell Technologies. Lance D. Trainor, MD OneBlood, Inc.
 Current Issues in Stem Cell Technologies Lance D. Trainor, MD OneBlood, Inc. Objective: The big picture of stem cell therapy Outline: Relevant definitions History of Stem Cell Therapy Hematopoietic Stem
Current Issues in Stem Cell Technologies Lance D. Trainor, MD OneBlood, Inc. Objective: The big picture of stem cell therapy Outline: Relevant definitions History of Stem Cell Therapy Hematopoietic Stem
STEM CELL FELLOWSHIP
 Module I: The Basic Principles of Stem Cells 1. Basics of Stem Cells a. Understanding the development of embryonic stem cells i. Embryonic stem cells ii. Embryonic germ cells iii. Differentiated stem cell
Module I: The Basic Principles of Stem Cells 1. Basics of Stem Cells a. Understanding the development of embryonic stem cells i. Embryonic stem cells ii. Embryonic germ cells iii. Differentiated stem cell
Ficoll-Paque PREMIUM Ficoll-Paque PREMIUM 1.084 Ficoll-Paque PREMIUM 1.073
 Instructions 28-4039-56 AE Cell Preparation Media Ficoll-Paque PREMIUM Ficoll-Paque PREMIUM 1.084 Ficoll-Paque PREMIUM 1.073 Intended use For in vitro isolation of mononuclear cells and/or granulocytes
Instructions 28-4039-56 AE Cell Preparation Media Ficoll-Paque PREMIUM Ficoll-Paque PREMIUM 1.084 Ficoll-Paque PREMIUM 1.073 Intended use For in vitro isolation of mononuclear cells and/or granulocytes
Human Placenta MSC in Placental Analysis
 Chang ARTICLE Dong LI et al Mesenchymal stem cells derived from human placenta suppress allogeneic umbilical cord blood lymphocyte proliferation Chang Dong LI 1, Wei Yuan ZHANG 1,*, He Lian LI 2, XiaoXia
Chang ARTICLE Dong LI et al Mesenchymal stem cells derived from human placenta suppress allogeneic umbilical cord blood lymphocyte proliferation Chang Dong LI 1, Wei Yuan ZHANG 1,*, He Lian LI 2, XiaoXia
Flow cytometric analysis of stem cells derived from umbilical cord blood CD34+/CD45+ enumeration and viability
 Flow cytometric analysis of stem cells derived from umbilical cord blood CD34+/CD45+ enumeration and viability Doretha Thompson Dissertation in fulfillment of the requirements for the degree Magister Scientiae
Flow cytometric analysis of stem cells derived from umbilical cord blood CD34+/CD45+ enumeration and viability Doretha Thompson Dissertation in fulfillment of the requirements for the degree Magister Scientiae
BioArchive Freezing Curve for Cord Blood
 BioArchive Freezing Curve for Cord Blood Determination of the Specific Freezing in the BioArchive System to Provide the Highest Viability of Cord Blood MNC as Measured by -C Assays Introduction Freezing
BioArchive Freezing Curve for Cord Blood Determination of the Specific Freezing in the BioArchive System to Provide the Highest Viability of Cord Blood MNC as Measured by -C Assays Introduction Freezing
WHOLE BLOOD LYSING SOLUTION FOR FLOW CYTOMETRIC APPLICATIONS
 FOR IN-VITRO DIAGNOSTIC USE INVITROGEN CAL-LYSE TM Lysing Solution WHOLE BLOOD LYSING SOLUTION FOR FLOW CYTOMETRIC APPLICATIONS CAL-LYSE TM CATALOG No. GAS-010 250 tests 25 ml CATALOG No. GAS-010S-100
FOR IN-VITRO DIAGNOSTIC USE INVITROGEN CAL-LYSE TM Lysing Solution WHOLE BLOOD LYSING SOLUTION FOR FLOW CYTOMETRIC APPLICATIONS CAL-LYSE TM CATALOG No. GAS-010 250 tests 25 ml CATALOG No. GAS-010S-100
Do you have anything to add? If so, I d love to hear from you! Jessica Robinson Conference Manager Life Sciences @jessbiopharma
 1 Who is the most influential figure in cord blood around the world? What is the biggest challenge to overcome in the use of cord blood as a source of stem cells? We asked 10 leading experts in the cord
1 Who is the most influential figure in cord blood around the world? What is the biggest challenge to overcome in the use of cord blood as a source of stem cells? We asked 10 leading experts in the cord
RPCI 004 v.002 Staining Procedure For all Directly Conjugated Reagents (Whole Blood Method)
 Immune Tolerance Network RPCI 004 v.002 Staining Procedure For all Directly Conjugated Reagents (Whole Blood Method) Author: Paul Wallace, Director, RPCI Laboratory of Flow Cytometry Approved by: Paul
Immune Tolerance Network RPCI 004 v.002 Staining Procedure For all Directly Conjugated Reagents (Whole Blood Method) Author: Paul Wallace, Director, RPCI Laboratory of Flow Cytometry Approved by: Paul
5 Frequently Asked Questions About Adult Stem Cell Research
 5 Frequently Asked Questions About Adult Stem Cell Research Stem cells are often referred to in the sociopolitical realm with some level of controversy and beyond that, some level of confusion. Many researchers
5 Frequently Asked Questions About Adult Stem Cell Research Stem cells are often referred to in the sociopolitical realm with some level of controversy and beyond that, some level of confusion. Many researchers
Enumeration and Viability of Nucleated Cells from Bone Marrow, Cord Blood, and Mobilized Peripheral Blood
 Application Note Primary Cell Analysis Enumeration and Viability of Nucleated Cells from Bone Marrow, Cord Blood, and Mobilized Peripheral Blood Cell Viability Introduction: Bone Marrow, Cord Blood, Stem
Application Note Primary Cell Analysis Enumeration and Viability of Nucleated Cells from Bone Marrow, Cord Blood, and Mobilized Peripheral Blood Cell Viability Introduction: Bone Marrow, Cord Blood, Stem
CFSE Cell Division Assay Kit
 CFSE Cell Division Assay Kit Item No. 10009853 Customer Service 800.364.9897 * Technical Support 888.526.5351 www.caymanchem.com TABLE OF CONTENTS GENERAL INFORMATION 3 Materials Supplied 4 Precautions
CFSE Cell Division Assay Kit Item No. 10009853 Customer Service 800.364.9897 * Technical Support 888.526.5351 www.caymanchem.com TABLE OF CONTENTS GENERAL INFORMATION 3 Materials Supplied 4 Precautions
Proliferation/Potency Assays: Is there more to CD34 and CFU?
 Proliferation/Potency Assays: Is there more to CD34 and CFU? CBMTG 2012 Laboratory Committee Meeting April 11, 2012 Toronto, ON, Canada Leah Marquez-Curtis CBS-Edmonton OUTLINE I. Rationale and Definitions
Proliferation/Potency Assays: Is there more to CD34 and CFU? CBMTG 2012 Laboratory Committee Meeting April 11, 2012 Toronto, ON, Canada Leah Marquez-Curtis CBS-Edmonton OUTLINE I. Rationale and Definitions
Transfection reagent for visualizing lipofection with DNA. For ordering information, MSDS, publications and application notes see www.biontex.
 METAFECTENE FluoR Transfection reagent for visualizing lipofection with DNA For ordering information, MSDS, publications and application notes see www.biontex.com Description Cat. No. Size METAFECTENE
METAFECTENE FluoR Transfection reagent for visualizing lipofection with DNA For ordering information, MSDS, publications and application notes see www.biontex.com Description Cat. No. Size METAFECTENE
INFORMATION ON STEM CELLS/BONE MARROW AND REINFUSION/TRANSPLANTATION SUR703.002
 INFORMATION ON STEM CELLS/BONE MARROW AND REINFUSION/TRANSPLANTATION SUR703.002 COVERAGE: SPECIAL COMMENT ON POLICY REVIEW: Due to the complexity of the Peripheral and Bone Marrow Stem Cell Transplantation
INFORMATION ON STEM CELLS/BONE MARROW AND REINFUSION/TRANSPLANTATION SUR703.002 COVERAGE: SPECIAL COMMENT ON POLICY REVIEW: Due to the complexity of the Peripheral and Bone Marrow Stem Cell Transplantation
Improved Ex Vivo Expansion of Functional CD34 + Cells Using Stemline II Hematopoietic Stem Cell Expansion Medium
 Improved Ex Vivo Expansion of Functional CD34 + Cells Using Stemline II Hematopoietic Stem Cell Expansion Medium Daniel W. Allison, 1 Stacy L. Leugers, 1 Barry J. Pronold, 2 Gary Van Zant, 2 Jenny A. Harrington,
Improved Ex Vivo Expansion of Functional CD34 + Cells Using Stemline II Hematopoietic Stem Cell Expansion Medium Daniel W. Allison, 1 Stacy L. Leugers, 1 Barry J. Pronold, 2 Gary Van Zant, 2 Jenny A. Harrington,
Human Adult Mesothelial Cell Manual
 Human Adult Mesothelial Cell Manual INSTRUCTION MANUAL ZBM0025.01 SHIPPING CONDITIONS Human Adult Mesothelial Cells Orders are delivered via Federal Express courier. All US and Canada orders are shipped
Human Adult Mesothelial Cell Manual INSTRUCTION MANUAL ZBM0025.01 SHIPPING CONDITIONS Human Adult Mesothelial Cells Orders are delivered via Federal Express courier. All US and Canada orders are shipped
Direct Antiglobulin Test (DAT)
 Exercise 8 Exercise 9 Direct Antiglobulin Test (DAT) Elution Study Task Aim Introduction To perform the DAT and elution procedure with correct interpretation of results. To perform with 100% accuracy the
Exercise 8 Exercise 9 Direct Antiglobulin Test (DAT) Elution Study Task Aim Introduction To perform the DAT and elution procedure with correct interpretation of results. To perform with 100% accuracy the
STEM CELLS FROM THE UMBLICAL CORD BLOOD AND UMBLICAL CORD TISSUE
 STEM CELLS FROM THE UMBLICAL CORD BLOOD AND UMBLICAL CORD TISSUE What are Stem Cells? Stem cells are the basic building blocks of all the cells, tissues and organs in the human body. The role of the stem
STEM CELLS FROM THE UMBLICAL CORD BLOOD AND UMBLICAL CORD TISSUE What are Stem Cells? Stem cells are the basic building blocks of all the cells, tissues and organs in the human body. The role of the stem
UMBILICAL CORD BLOOD TRANSPLANTATION: KFSH EXPERIENCE
 UMBILICAL CORD BLOOD TRANSPLANTATION: KFSH EXPERIENCE HIND AL HUMAIDAN, MD,FRCPA Director, Blood Bank (Donor & Transfusion Services) and Stem Cell Cord Blood Bank Consultant Hematopathologist INTRODUCTION
UMBILICAL CORD BLOOD TRANSPLANTATION: KFSH EXPERIENCE HIND AL HUMAIDAN, MD,FRCPA Director, Blood Bank (Donor & Transfusion Services) and Stem Cell Cord Blood Bank Consultant Hematopathologist INTRODUCTION
CD3/TCR stimulation and surface detection Determination of specificity of intracellular detection of IL-7Rα by flow cytometry
 CD3/TCR stimulation and surface detection Stimulation of HPB-ALL cells with the anti-cd3 monoclonal antibody OKT3 was performed as described 3. In brief, antibody-coated plates were prepared by incubating
CD3/TCR stimulation and surface detection Stimulation of HPB-ALL cells with the anti-cd3 monoclonal antibody OKT3 was performed as described 3. In brief, antibody-coated plates were prepared by incubating
Generation of EBV-immortalized B cell lines
 UCANU 0014 Version November 01, 2011 Page 1 van 6 Generation of EBVimmortalized B cell lines CMCI (Center for molecular and Cellular Intervention) University Medical Center Utrecht Written by Name Function
UCANU 0014 Version November 01, 2011 Page 1 van 6 Generation of EBVimmortalized B cell lines CMCI (Center for molecular and Cellular Intervention) University Medical Center Utrecht Written by Name Function
Corporate Medical Policy Cord Blood as a Source of Stem Cells
 Corporate Medical Policy Cord Blood as a Source of Stem Cells File Name: Origination: Last CAP Review: Next CAP Review: Last Review cord_blood_as_a_source_of_stem_cells 2/2001 3/2015 3/2016 3/2015 Description
Corporate Medical Policy Cord Blood as a Source of Stem Cells File Name: Origination: Last CAP Review: Next CAP Review: Last Review cord_blood_as_a_source_of_stem_cells 2/2001 3/2015 3/2016 3/2015 Description
Stem Cell Transplantation
 Harmony Behavioral Health, Inc. Harmony Behavioral Health of Florida, Inc. Harmony Health Plan of Illinois, Inc. HealthEase of Florida, Inc. Ohana Health Plan, a plan offered by WellCare Health Insurance
Harmony Behavioral Health, Inc. Harmony Behavioral Health of Florida, Inc. Harmony Health Plan of Illinois, Inc. HealthEase of Florida, Inc. Ohana Health Plan, a plan offered by WellCare Health Insurance
Role Of Mitochondria In Mesenchymal Stem Cells
 Role Of Mitochondria In Mesenchymal Stem Cells Roman Eliseev, MD, PhD, Tamara Raymond, Regis O'Keefe, MD, PhD. University of Rochester, Rochester, NY, USA. Disclosures: R. Eliseev: None. T. Raymond: None.
Role Of Mitochondria In Mesenchymal Stem Cells Roman Eliseev, MD, PhD, Tamara Raymond, Regis O'Keefe, MD, PhD. University of Rochester, Rochester, NY, USA. Disclosures: R. Eliseev: None. T. Raymond: None.
For in vitro preparation of human mononuclear cells from peripheral blood, bone marrow, and umbilical cord blood. Not for in vitro diagnostic use.
 GE Healthcare Instructions 28-4039-56 AA Cell Preparation Media Ficoll-Paque PREMIUM Intended use For in vitro preparation of human mononuclear cells from peripheral blood, bone marrow, and umbilical cord
GE Healthcare Instructions 28-4039-56 AA Cell Preparation Media Ficoll-Paque PREMIUM Intended use For in vitro preparation of human mononuclear cells from peripheral blood, bone marrow, and umbilical cord
guides BIOLOGY OF AGING STEM CELLS An introduction to aging science brought to you by the American Federation for Aging Research
 infoaging guides BIOLOGY OF AGING STEM CELLS An introduction to aging science brought to you by the American Federation for Aging Research WHAT ARE STEM CELLS? Stem cells are cells that, in cell cultures
infoaging guides BIOLOGY OF AGING STEM CELLS An introduction to aging science brought to you by the American Federation for Aging Research WHAT ARE STEM CELLS? Stem cells are cells that, in cell cultures
Stem Cell Quick Guide: Stem Cell Basics
 Stem Cell Quick Guide: Stem Cell Basics What is a Stem Cell? Stem cells are the starting point from which the rest of the body grows. The adult human body is made up of hundreds of millions of different
Stem Cell Quick Guide: Stem Cell Basics What is a Stem Cell? Stem cells are the starting point from which the rest of the body grows. The adult human body is made up of hundreds of millions of different
The Prospect of Stem Cell Therapy in Multiple Sclerosis. Multiple sclerosis is a multifocal inflammatory disease of the central
 The Prospect of Stem Cell Therapy in Multiple Sclerosis Multiple sclerosis is a multifocal inflammatory disease of the central nervous system that generally affects young individuals, causing paralysis
The Prospect of Stem Cell Therapy in Multiple Sclerosis Multiple sclerosis is a multifocal inflammatory disease of the central nervous system that generally affects young individuals, causing paralysis
CHARACTERISTIC FEATURES OF STEM CELLS. CLONING TECHNOLOGIES
 CHARACTERISTIC FEATURES OF STEM CELLS. CLONING TECHNOLOGIES 1 Science is discovering the unknown Stem cell field is still in its infancy Human embryonic stem cell research is a decade old, adult stem cell
CHARACTERISTIC FEATURES OF STEM CELLS. CLONING TECHNOLOGIES 1 Science is discovering the unknown Stem cell field is still in its infancy Human embryonic stem cell research is a decade old, adult stem cell
UMBILICAL CORD BLOOD HARVESTING & STORAGE
 Protocol: TRP009 Effective Date: October 14, 2013 UMBILICAL CORD BLOOD HARVESTING & STORAGE Table of Contents Page COMMERCIAL, MEDICARE & MEDICAID COVERAGE RATIONALE... 1 BACKGROUND... 2 CLINICAL EVIDENCE...
Protocol: TRP009 Effective Date: October 14, 2013 UMBILICAL CORD BLOOD HARVESTING & STORAGE Table of Contents Page COMMERCIAL, MEDICARE & MEDICAID COVERAGE RATIONALE... 1 BACKGROUND... 2 CLINICAL EVIDENCE...
Stem Cell Research: Adult or Somatic Stem Cells
 Chiang 1 Stem Cell Research: Adult or Somatic Stem Cells Abstract Kelly Chiang Cluster 7 Dr. LeFebvre 07/26/10 Over the past few decades, stem cells have been a controversial topic in the scientific field.
Chiang 1 Stem Cell Research: Adult or Somatic Stem Cells Abstract Kelly Chiang Cluster 7 Dr. LeFebvre 07/26/10 Over the past few decades, stem cells have been a controversial topic in the scientific field.
Prostaglandin E2 Enhances Human Cord Blood Stem. Cell Xenotransplants and Shows Long-Term Safety. in Preclinical Nonhuman Primate Transplant Models
 Cell Stem Cell, Volume 8 Supplemental Information Prostaglandin E2 Enhances Human Cord Blood Stem Cell Xenotransplants and Shows Long-Term Safety in Preclinical Nonhuman Primate Transplant Models Wolfram
Cell Stem Cell, Volume 8 Supplemental Information Prostaglandin E2 Enhances Human Cord Blood Stem Cell Xenotransplants and Shows Long-Term Safety in Preclinical Nonhuman Primate Transplant Models Wolfram
No-wash, no-lyse detection of leukocytes in human whole blood on the Attune NxT Flow Cytometer
 APPLICATION NOTE Attune NxT Flow Cytometer No-wash, no-lyse detection of leukocytes in human whole blood on the Attune NxT Flow Cytometer Introduction Standard methods for isolating and detecting leukocytes
APPLICATION NOTE Attune NxT Flow Cytometer No-wash, no-lyse detection of leukocytes in human whole blood on the Attune NxT Flow Cytometer Introduction Standard methods for isolating and detecting leukocytes
Automation of Cell Staining Technical Information Bulletin
 Automation of Cell Staining Technical Information Bulletin Automation of Cell Staining Using the Biomek 4000 Laboratory Automation Workstation Amy N. Yoder, Sr. Development Scientist and Li Liu, Sr. Development
Automation of Cell Staining Technical Information Bulletin Automation of Cell Staining Using the Biomek 4000 Laboratory Automation Workstation Amy N. Yoder, Sr. Development Scientist and Li Liu, Sr. Development
Follicle Dermal Papilla Cell
 Follicle Dermal Papilla Cell Instruction Manual Product Size Catalog Number Human Follicle Dermal Papilla Cells (HFDPC) 500,000 cryopreserved cells 500,000 proliferating cells C-12071 C-12072 Product Description
Follicle Dermal Papilla Cell Instruction Manual Product Size Catalog Number Human Follicle Dermal Papilla Cells (HFDPC) 500,000 cryopreserved cells 500,000 proliferating cells C-12071 C-12072 Product Description
Therapeutic Potential of Cells Derived from Gestational Tissues: A coming of age. Dr Roisin Deane Cell Care Australia Pty Ltd
 Therapeutic Potential of Cells Derived from Gestational Tissues: A coming of age Dr Roisin Deane Cell Care Australia Pty Ltd What are Gestational Stem Cells? Stem cells isolated from the gestational tissues
Therapeutic Potential of Cells Derived from Gestational Tissues: A coming of age Dr Roisin Deane Cell Care Australia Pty Ltd What are Gestational Stem Cells? Stem cells isolated from the gestational tissues
SAVE A LIFE... BY GIVING LIFE!
 SAVE A LIFE... BY GIVING LIFE! FOLLOW US ON: HÉMA-QUÉBEC PUBLIC CORD BLOOD BANK www.hema-quebec.qc.ca Scan this code with your smart phone to access the page Register to the Public Cord Blood Bank on the
SAVE A LIFE... BY GIVING LIFE! FOLLOW US ON: HÉMA-QUÉBEC PUBLIC CORD BLOOD BANK www.hema-quebec.qc.ca Scan this code with your smart phone to access the page Register to the Public Cord Blood Bank on the
EdU Flow Cytometry Kit. User Manual
 User Manual Ordering information: (for detailed kit content see Table 2) EdU Flow Cytometry Kits for 50 assays: Product number EdU Used fluorescent dye BCK-FC488-50 10 mg 6-FAM Azide BCK-FC555-50 10 mg
User Manual Ordering information: (for detailed kit content see Table 2) EdU Flow Cytometry Kits for 50 assays: Product number EdU Used fluorescent dye BCK-FC488-50 10 mg 6-FAM Azide BCK-FC555-50 10 mg
APPLICATION INFORMATION
 DRAFT: Rev. D A-2045A APPLICATION INFORMATION Flow Cytometry 3-COLOR COMPENSATION Raquel Cabana,* Mark Cheetham, Jay Enten, Yong Song, Michael Thomas,* and Brendan S. Yee Beckman Coulter, Inc., Miami FL
DRAFT: Rev. D A-2045A APPLICATION INFORMATION Flow Cytometry 3-COLOR COMPENSATION Raquel Cabana,* Mark Cheetham, Jay Enten, Yong Song, Michael Thomas,* and Brendan S. Yee Beckman Coulter, Inc., Miami FL
Direct Antiglobulin Test (DAT)
 Exercise 8 Direct Antiglobulin Test (DAT) Objectives: 1. State the purpose for performing the DAT. 2. State what a positive DAT indicates. 3. List the reagents which are used for performing the DAT. 4.
Exercise 8 Direct Antiglobulin Test (DAT) Objectives: 1. State the purpose for performing the DAT. 2. State what a positive DAT indicates. 3. List the reagents which are used for performing the DAT. 4.
Workshop 14-16 February 2006
 Theoretical and practical approaches of Hepatocyte primary culture Workshop 14-16 February 2006 Lecture (2) Disaggregation & purification of target cells Coarse organizer Dr. Abo bakr Mohamed Eltayeb General
Theoretical and practical approaches of Hepatocyte primary culture Workshop 14-16 February 2006 Lecture (2) Disaggregation & purification of target cells Coarse organizer Dr. Abo bakr Mohamed Eltayeb General
Cultivation and Cryopreservation of Cord Tissue MSCs with Cord Blood AB Plasma
 Cultivation and Cryopreservation of Cord Tissue MSCs with Cord Blood AB Plasma Manasi Talwadekar, Darshana Kadekar, Sonal Rangole, Nikhat Firdaus Khan, Vaijayanti Kale and Lalita Limaye* Stem Cell Laboratory,
Cultivation and Cryopreservation of Cord Tissue MSCs with Cord Blood AB Plasma Manasi Talwadekar, Darshana Kadekar, Sonal Rangole, Nikhat Firdaus Khan, Vaijayanti Kale and Lalita Limaye* Stem Cell Laboratory,
STEM CELL FACTS. The ISSCR is an independent, nonproft organization providing a global forum for stem cell research and regenerative medicine.
 STEM CELL FACTS The ISSCR is an independent, nonproft organization providing a global forum for stem cell research and regenerative medicine. WHAT ARE STEM CELLS? Stem cells are the foundation cells for
STEM CELL FACTS The ISSCR is an independent, nonproft organization providing a global forum for stem cell research and regenerative medicine. WHAT ARE STEM CELLS? Stem cells are the foundation cells for
Hematopoiesis i starts from. HPC differentiates t to sequentially more mature. HPC circulate in very low
 Upstate Cord Blood Bank Hematopoietic Progenitor Cells Hematopoiesis i starts from self renewing HPC HPC differentiates t to sequentially more mature blood cells HPC circulate in very low numbers in adults
Upstate Cord Blood Bank Hematopoietic Progenitor Cells Hematopoiesis i starts from self renewing HPC HPC differentiates t to sequentially more mature blood cells HPC circulate in very low numbers in adults
Stem cells possess 2 main characteristics: Sources of pluripotent stem cells: -Long-term self renewal. -The inner cell mass of the blastocyst.
 Stem cells possess 2 main characteristics: -Long-term self renewal. - They give rise to all types of differentiate cells. Sources of pluripotent stem cells: -The inner cell mass of the blastocyst. - Fetal
Stem cells possess 2 main characteristics: -Long-term self renewal. - They give rise to all types of differentiate cells. Sources of pluripotent stem cells: -The inner cell mass of the blastocyst. - Fetal
Cord Blood: Thawing & Infusion Practical Methods
 Cord Blood: Thawing & Infusion Practical Methods Donna M Regan, MT(ASCP)SBB St. Louis Cord Blood Bank @ SSM Cardinal Glennon Children s s Medical Center Objectives Present variety of product configurations
Cord Blood: Thawing & Infusion Practical Methods Donna M Regan, MT(ASCP)SBB St. Louis Cord Blood Bank @ SSM Cardinal Glennon Children s s Medical Center Objectives Present variety of product configurations
Technical report High-efficiency volume reduction of cord blood using pentastarch
 (2001) 27, 457 461 2001 Nature Publishing Group All rights reserved 0268 3369/01 $15.00 www.nature.com/bmt Technical report High-efficiency volume reduction of cord blood using pentastarch H Yang 1,2,
(2001) 27, 457 461 2001 Nature Publishing Group All rights reserved 0268 3369/01 $15.00 www.nature.com/bmt Technical report High-efficiency volume reduction of cord blood using pentastarch H Yang 1,2,
Unit 1 Higher Human Biology Summary Notes
 Unit 1 Higher Human Biology Summary Notes a. Cells tissues organs body systems Division of labour occurs in multicellular organisms (rather than each cell carrying out every function) Most cells become
Unit 1 Higher Human Biology Summary Notes a. Cells tissues organs body systems Division of labour occurs in multicellular organisms (rather than each cell carrying out every function) Most cells become
Application Note 10. Measurement of Cell Recovery. After Sorting with a Catcher-Tube-Based. Cell Sorter. Introduction
 Application Note 10 Measurement of Cell Recovery After Sorting with a Catcher-Tube-Based Cell Sorter Introduction In many experiments using sorted cells, it is important to be able to count the number
Application Note 10 Measurement of Cell Recovery After Sorting with a Catcher-Tube-Based Cell Sorter Introduction In many experiments using sorted cells, it is important to be able to count the number
Overview. Transcriptional cascades. Amazing aspects of lineage plasticity. Conventional (B2) B cell development
 Overview B cell development Transcriptional cascades Amazing aspects of lineage plasticity Conventional (B2) B cell development What happens to an autoreactive B cell? B1 vs B2 cells Key anatomical sites
Overview B cell development Transcriptional cascades Amazing aspects of lineage plasticity Conventional (B2) B cell development What happens to an autoreactive B cell? B1 vs B2 cells Key anatomical sites
Protocols for Adipogenic Differentiation Assays for Characterization of Adipose Stromal Cells (ASC)
 Protocols for Adipogenic Differentiation Assays for Characterization of Adipose Stromal Cells (ASC) Purpose: To characterize the adipogenic differentiation capabilities of adipose stromal cells. Overview:
Protocols for Adipogenic Differentiation Assays for Characterization of Adipose Stromal Cells (ASC) Purpose: To characterize the adipogenic differentiation capabilities of adipose stromal cells. Overview:
AAGPs TM Anti-Aging Glyco Peptides. Enhancing Cell, Tissue and Organ Integrity Molecular and biological attributes of lead AAGP molecule
 AAGPs TM Anti-Aging Glyco Peptides Enhancing Cell, Tissue and Organ Integrity Molecular and biological attributes of lead AAGP molecule 1 Acknowledgements This presentation was prepared by Dr. Samer Hussein
AAGPs TM Anti-Aging Glyco Peptides Enhancing Cell, Tissue and Organ Integrity Molecular and biological attributes of lead AAGP molecule 1 Acknowledgements This presentation was prepared by Dr. Samer Hussein
tem ells /background /information Stem cell research Copyright 2007 MRC Centre for Regenerative Medicine, Institute for Stem Cell Research
 tem ells /background /information Stem cell research Copyright 2007 MRC Centre for Regenerative Medicine, Institute for Stem Cell Research /02 /information Table of contents Page 01. What are stem cells?
tem ells /background /information Stem cell research Copyright 2007 MRC Centre for Regenerative Medicine, Institute for Stem Cell Research /02 /information Table of contents Page 01. What are stem cells?
Corporate Medical Policy Cord Blood as a Source of Stem Cells
 Corporate Medical Policy Cord Blood as a Source of Stem Cells File Name: Origination: Last CAP Review: Next CAP Review: Last Review cord_blood_as_a_source_of_stem_cells 2/2001 3/2015 3/2016 3/2015 Description
Corporate Medical Policy Cord Blood as a Source of Stem Cells File Name: Origination: Last CAP Review: Next CAP Review: Last Review cord_blood_as_a_source_of_stem_cells 2/2001 3/2015 3/2016 3/2015 Description
BD PuraMatrix Peptide Hydrogel
 BD PuraMatrix Peptide Hydrogel Catalog No. 354250 Guidelines for Use FOR RESEARCH USE ONLY NOT FOR CLINICAL, DIAGNOSTIC OR THERAPEUTIC USE SPC-354250-G rev 2.0 1 TABLE OF CONTENTS Intended Use... 2 Materials
BD PuraMatrix Peptide Hydrogel Catalog No. 354250 Guidelines for Use FOR RESEARCH USE ONLY NOT FOR CLINICAL, DIAGNOSTIC OR THERAPEUTIC USE SPC-354250-G rev 2.0 1 TABLE OF CONTENTS Intended Use... 2 Materials
CD34+ CD45- Cells Isolated from Human Blood Have Molecular and Functional Characteristics of Vascular Endothelial Progenitors
 CD34+ CD45- Cells Isolated from Human Blood Have Molecular and Functional Characteristics of Vascular Endothelial Progenitors Yajuan Jiang 1, Alexander M. Tsankov 2, Alexander Meissner 2 and David W. O
CD34+ CD45- Cells Isolated from Human Blood Have Molecular and Functional Characteristics of Vascular Endothelial Progenitors Yajuan Jiang 1, Alexander M. Tsankov 2, Alexander Meissner 2 and David W. O
STEMCELL Quality Control Kits
 STEMCELL Quality Control Kits i Table of Contents 1.0 Introduction...1 2.0 Thawing Cells, Plating and Colony Enumeration...2 2.1 Supplies and reagents included in the QC kit...2 2.2 Additional reagents
STEMCELL Quality Control Kits i Table of Contents 1.0 Introduction...1 2.0 Thawing Cells, Plating and Colony Enumeration...2 2.1 Supplies and reagents included in the QC kit...2 2.2 Additional reagents
The Types of stem cells: Totipotent Pluripotent Multipotent
 Stem Cells is the main material for building and regeneration of the body Stem cells are not differentiated and can transform to any cell of organism Stem cells are capable of indefinite renewal through
Stem Cells is the main material for building and regeneration of the body Stem cells are not differentiated and can transform to any cell of organism Stem cells are capable of indefinite renewal through
Stem Cells in Umbilical Cord Blood: A Review
 Stem Cells in Umbilical Cord Blood: A Review December 13, 2006 Cellular and Molecular Biotechnology of Mammalian Systems Undergraduate Students: Andrew Fu Allen Fung Graduate Student: Aizhan Tastanova
Stem Cells in Umbilical Cord Blood: A Review December 13, 2006 Cellular and Molecular Biotechnology of Mammalian Systems Undergraduate Students: Andrew Fu Allen Fung Graduate Student: Aizhan Tastanova
How To Make A Tri Reagent
 TRI Reagent For processing tissues, cells cultured in monolayer or cell pellets Catalog Number T9424 Store at room temperature. TECHNICAL BULLETIN Product Description TRI Reagent is a quick and convenient
TRI Reagent For processing tissues, cells cultured in monolayer or cell pellets Catalog Number T9424 Store at room temperature. TECHNICAL BULLETIN Product Description TRI Reagent is a quick and convenient
MEF Nucleofector Kit 1 and 2
 page 1 of 7 MEF Nucleofector Kit 1 and 2 for Mouse Embryonic Fibroblasts (MEF) MEF display significant phenotypic variations which depend on the strain, the genetic background of the they are isolated
page 1 of 7 MEF Nucleofector Kit 1 and 2 for Mouse Embryonic Fibroblasts (MEF) MEF display significant phenotypic variations which depend on the strain, the genetic background of the they are isolated
