CLINALFA STERILE PEPTIDES FOR CLINICAL TRIALS
|
|
|
- Madison Griffin
- 9 years ago
- Views:
Transcription
1 CLINALFA STERILE PEPTIDES FOR CLINICAL TRIALS
2 Clinalfa CLINALFA YOUR PARTNER IN CLINICAL RESEARCH Products of highest quality Manufactured under GMP conditions Sterile and pyrogen-free Ready for use in approved clinical studies Released by QP with CofA signed by QP Fast delivery from stock Documentation for ethical committees and regulatory authorities available We invite you to discuss your specific needs with us 2
3 APIs We are the leading independent supplier of peptidic active pharmaceutical ingredients (APIs) for the human and veterinary pharmaceutical markets. With our high capacity and the outstanding quality of our products Bachem holds a unique position. Our expertise is in large-scale solid-phase peptide synthesis (Boc- and Fmoc-strategies) and solutionphase peptide synthesis. Purification procedures include large-scale preparative high performance liquid chromatography (preparative HPLC), conventional chromatography (ion exchange, gel chromatography) and countercurrent (Craig) distribution. Our GMP compliant manufacturing facilities for APIs fulfill the highest standards and are regularly inspected by the FDA and/or local authorities. Additionally, Bachem has a production unit for the custom synthesis of small organic molecules. cgmp PRODUCTION More than 40 Years of Expertise in Peptide Chemistry Fully Independent Company Quality You Can Trust Full Range of Technical and Regulatory Support Excellent customer and technical service 3
4 Clinalfa ST. HELENS (UK) WEIL AM RHEIN (D) VIONNAZ (CH) BUBENDORF (CH) TORRANCE (CA) VISTA (CA) GLOBAL BUSINESS Bachem s facilities are located in Switzerland, Europe and in the USA. All cgmp manufacturing sites are inspected by the US-FDA and national authorities. GMP sites 4
5 OUR PRODUCTS AND SERVICES Clinalfa basic Clinalfa basic peptides are sterile freeze-dried medicinal products for use in clinical trials. The supply of Clinalfa basic peptides is accompanied by Bachem s Certificate of Analysis (CofA), which is signed by Bachem s Qualified Person (QP). The manufacturing process and the analytical methods might not be fully validated and only limited stability data are available. Product Prod. Nr. Pack Size Page Angiotensin II Acetate* U μg/vial 8 (Pyr 1 )-Apelin-13 Acetate* U mg/vial 8 Angiotensin I/II (1-7) (human) Acetate* U mg/vial 9 BQ-123 Sodium salt* U mg/vial 9 BQ-788 Sodium salt U mg/vial 9 Bradykinin Acetate* U μg/vial 10 CCK-4 Acetate U μg/vial 10 CRF (ovine) Acetate U μg/vial 10 Exendin (9-39) Acetate* U mg/vial 11 Ghrelin (human) Acetate (formulated)* U μg/vial 11 (Des-octanoyl)-Ghrelin (human) Acetate* U μg/vial 11 GIP Acetate* U μg/vial 12 GLP-1 (7-36) amide Acetate* U μg/vial 12 Neuropeptide Y (human, rat) Acetate U μg/vial 13 L-NMMA Acetate* U mg/vial 13 Pentagastrin U μg/vial 13 Substance P Acetate U μg/vial 14 *Clinalfa basic peptides available from stock. 5
6 Clinalfa Clinalfa plus Clinalfa plus products are sterile freeze-dried or ready-to-use liquid formulations made according to customer requirements. In many cases, the active pharmaceutical ingredients (APIs) and the resulting Clinalfa products are proprietary products of Bachem s customers. Based on agreements between the customer and Bachem, Bachem s Qualified Person (QP) may release the products for use in a specific clinical study approved by the competent authority, and also for market approval (see page 15). Clinalfa Services Bachem offers related services to Clinalfa basic and Clinalfa plus products, i.e. formulation development, compatibility studies, stress test and post-release stability studies, CMC documentation for IMPD and IND, drug master file and other services (see page 15). 6
7 CLINALFA BASIC PRODUCTS Clinalfa basic products are sterile freeze-dried peptides available from stock. The supply of Clinalfa basic peptides is accompanied by Bachem s Certificate of Analysis (CofA) signed by Bachem s Qualified Person (QP). 7
8 Clinalfa ANGIOTENSIN II 50 μg/vial U-1050, Synonym Ang II H-Asp-Arg-Val-Tyr-Ile-His-Pro- Phe-OH acetate salt C 50 H 71 N 13 O 12 : g/mol CAS No: (net) The octapeptide angiotensin II (Ang II) binds to both the AT1 and the AT2 receptors. Ang II interaction with the AT1 receptor results in strong vasoactive effects, the stimulation of aldosterone synthesis and the elevation of blood pressure via direct vasoconstriction of arteriole smooth muscles. The role of the AT2 receptor is not fully understood. Ang II can be metabolized by aminopeptidase into angiotensin III (Ang 2-8) and angiotensin IV (Ang 3-8). H-1705 Angiotensin II (PYR 1 )-APELIN mg/vial U-1270, Pyr-Arg-Pro-Arg-Leu-Ser-His-Lys-Gly- Pro-Met-Pro-Phe-OH acetate salt C 69 H 108 N 22 O 16 S : g/mol CAS No: (net) Apelin-36 was shown to be the specific endogenous ligand of the orphan G protein-coupled receptor APJ. APJ and the precursor peptide of apelin-36 are expressed in a variety of tissues including the heart, brain, breast, and lung; both are effective in the CNS and in the periphery. The metabolization products of apelin-36, e.g. (Pyr 1 )-Apelin-13, are considerably more active than the native peptide. Apelin-36 and its C- terminal fragments are involved in cell proliferation, vessel formation (regulation of the caliber size of blood vessels), and fluid homeostasis. (Pyr 1 )-Apelin-13, which has been identified as the predominant apelin isoform in the human heart, is a potent regulator of cardiovascular functions and a hypotensive. Apelin is also secreted by mature adipocytes, acting as an adipokinin. Plasma levels of apelin and its metabolites are altered in obese and hyperinsulinemic humans and in case of heart failure. (Pyr 1 )-Apelin-13 was shown to stimulate glucose utilization in normal and obese insulin-resistant mice. First positive results have been obtained in studies applying the apelin fragment in humans with chronic heart failure. (Pyr 1 )-Apelin-13 is a valuable tool in obesity, diabetes and, especially, cardiovascular research. This product is available in research H-4568 (Pyr 1 )-Apelin-13 (human, bovine, mouse, rat) 8
9 ANGIOTENSIN I/II (1-7) (HUMAN) 10 mg/vial U-1280, H-Asp-Arg-Val-Tyr-Ile-His-Pro-OH acetate salt C 41 H 62 N 12 O 11 : g/mol CAS No: (net) Angiotensin I/II (1-7) is a biologically active endogenous angiotensin. It is a product of a pathway independent of the angiotensin I processing by an angiotensin converting enzyme (ACE). Angiotensin I/II (1-7) may play an important role in the regulation of blood pressure as it has possible antihypertensive actions being a specific non-competitive antagonist of angiotensin II. Its vasodilator activity is mediated by stimulation of vasodilator prostaglandins and/ or the release of nitric oxide. Moreover the heptapeptide was shown to inhibit growth of human lung cancer cells in vitro and to reduce the size of human lung cancer xenografts in vivo. This product is available in research H-1715 Angiotensin I/II (1-7) BQ-123 SODIUM SALT 5 mg/vial U-1070, Cyclo(-D-Trp-D-Asp-Pro-D-Val-Leu) sodium salt C 31 H 42 N 6 O 7 : g/mol CAS No: (net) BQ-123 is a highly selective ET-A receptor antagonist. This product is available in research H-1252 BQ-123 BQ-788 SODIUM SALT 1 mg/vial U-1060, N-cis-2,6-Dimethylpiperidino- carbonyl-4-methyl-leu-d-trp(1- methoxycarbonyl)-d-nle-oh sodium salt BQ-788 is a highly selective ET-B receptor antagonist. This product is available in research H-2492 BQ-788 C 34 H 51 N 5 O 7 : g/mol CAS No: (net), (sodium salt) 9
10 Clinalfa BRADYKININ 50 μg/vial U-1080, H-Arg-Pro-Pro-Gly-Phe-Ser-Pro-Phe- Arg-OH acetate salt C 50 H 73 N 15 O 11 : g/mol CAS No: (net), (acetate) The pharmacological effects of bradykinin include smooth muscle contraction, vasodilation and hypotension, increase in capillary permeability with oedema formation and induction of pain. Bradykinin induces bronchoconstriction in asthmatic subjects and symptoms of rhinitis on nasal provocation. H-1970 Bradykinin (BK, Kallidin I, Kallidin-9, BRS-640) CCK-4 50 μg/vial U-1000, Synonyms Gastrin Tetrapeptide, Cholecystokinin Octapeptide (5-8), Cholecystokinin Tetrapeptide, Gastrin (14-17) (human) H-Trp-Met-Asp-Phe-NH 2 acetate salt C 29 H 36 N 6 O 6 S : g/mol CAS No: (net) CCK-4 reliably and dose-dependently induces panic-like symptoms both in healthy volunteers and patients with panic disorders. H-3110 Gastrin Tetrapeptide (Cholecystokinin Tetrapeptide, Cholecystokinin Octapeptide (5-8), CCK-4, Gastrin (14-17) (human)) CRF (OVINE) 100 μg/vial U-1170, H-Ser-Gln-Glu-Pro-Pro-Ile-Ser-Leu- Asp-Leu-Thr-Phe-His-Leu-Leu-Arg-Glu- Val-Leu-Glu-Met-Thr-Lys-Ala-Asp-Gln- Leu-Ala-Gln-Gln-Ala-His-Ser-Asn-Arg- Lys-Leu-Leu-Asp-Ile-Ala-NH 2 acetate salt C 205 H 339 N 59 O 63 S : g/mol CAS No: (net) Corticotropin-releasing factor (CRF) is a hypothalamic peptide which stimulates the release of adrenocorticotropic hormone (ACTH) and β-endorphin from the anterior pituitary. CRF also shows effects on reproductive functioning at local, neuroendocrine and behavioral levels, and depression. Ovine CRF produces a longer lasting ACTH response in humans than human CRF, mainly due to its longer plasma half-life. H-2445 CRF (ovine) (Corticorelin (ovine)) 10
11 EXENDIN (9-39) 10 mg/vial U-1160, H-Asp-Leu-Ser-Lys-Gln-Met-Glu-Glu- Glu-Ala-Val-Arg-Leu-Phe-Ile-Glu-Trp- Leu-Lys-Asn-Gly-Gly-Pro-Ser-Ser-Gly- Ala-Pro-Pro-Pro-Ser-NH 2 acetate salt C 149 H 234 N 40 O 47 S : g/mol CAS No: (net) Exendin (9-39) is a GLP-1 (glucagon-like peptide-1) receptor antagonist and a competitive inhibitor of exendin-3 and exendin-4. H-8740 Exendin (9-39) GHRELIN (HUMAN) 100 μg/vial (FORMULATED) U-1250, H-Gly-Ser-Ser(octanoyl)-Phe-Leu-Ser- Pro-Glu-His-Gln-Arg-Val-Gln-Gln-Arg- Lys-Glu-Ser-Lys-Lys-Pro-Pro-Ala-Lys- Leu-Gln-Pro-Arg-OH acetate salt C 149 H 249 N 47 O 42 : g/mol CAS No: (net) Ghrelin is an endogenous ligand for the growth hormone secretagogue receptor and is involved in regulating growth hormone (GH) release. Apart from GH release ghrelin stimulates appetite and weight gain. H-4864 Ghrelin (human) (DES- OCTANOYL)- GHRELIN (HUMAN) 100 μg/vial U-1300, H-Gly-Ser-Ser-Phe-Leu-Ser-Pro-Glu- His-Gln-Arg-Val-Gln-Gln-Arg-Lys-Glu- Ser-Lys-Lys-Pro-Pro-Ala-Lys-Leu-Gln- Pro-Arg-OH acetate salt C 141 H 235 N 47 O 41 : g/mol CAS No: (net) Ghrelin is an endogenous ligand for the growth hormone secretagogue receptor. Most circulating ghrelin is des-octanoyl ghrelin. The non-acylated ghrelin was considered inactive at first, though biological activities such as stimulation of adipogenesis and control of cell growth could be demonstrated more recently. Both ghrelin and the more abundant endogenous form des-octanoyl ghrelin could play a role in the paracrine regulation of vascular tone in humans. This product is available in research H-5946 Des-octanoyl)-Ghrelin (human) 11
12 Clinalfa GIP 500 μg/vial U-1240, Synonyms Gastric Inhibitory Polypeptide (human), Glucose-Dependent Insulinotropic Polypeptide (human) H-Tyr-Ala-Glu-Gly-Thr-Phe-Ile-Ser-Asp- Tyr-Ser-Ile-Ala-Met-Asp-Lys-Ile-His- Gln-Gln-Asp-Phe-Val-Asn-Trp-Leu-Leu- Ala-Gln-Lys-Gly-Lys-Lys-Asn-Asp-Trp- Lys-His-Asn-Ile-Thr-Gln-OH acetate salt C 226 H 338 N 60 O 66 S : g/mol CAS No: (net) Gastric inhibitory polypeptide (GIP; also, and more adequately, designated as glucose-dependent insulinotropic polypeptide) is related to glucagon-like peptide-1 (GLP-1). Both are incretins, gastrointestinal hormones which regulate glucose homeostasis. GIP and GLP-1 stimulate insulin production after the ingestion of a meal. GIP is generated from a 153-amino acid precursor protein in the K cells of the duodenum mucosa and the jejunum. GIP induces insulin secretion by docking to the GIP receptors on the surface of pancreatic β-cells, though it is (contrary to GLP-1) inactive in type II diabetes. GIP receptors have also been detected in adipose tissue, the guts, the heart, adrenal cortex, and brain, suggesting further functions of the peptide hormone. In adipocytes, GIP enhances the metabolism of fatty acids by stimulating lipase activity. GIP alone and in combination with GLP-1 has gained interest in the secondary prevention of insulin resistance and in the management of obesity. H-5645 Gastric Inhibitory Polypeptide (human) GLP-1 (7-36) AMIDE 100 μg/vial U-1190, H-His-Ala-Glu-Gly-Thr-Phe-Thr-Ser- Asp-Val-Ser-Ser-Tyr-Leu-Glu-Gly-Gln- Ala-Ala-Lys-Glu-Phe-Ile-Ala-Trp-Leu- Val-Lys-Gly-Arg-NH 2 acetate salt C 149 H 226 N 40 O 45 : g/mol CAS No: (net) This GLP-1 fragment is secreted from the lower small intestine and shows a strong insulinotropic effect. It is presently considered as the most important incretin hormone. Its action is mediated by receptors expressed by the endocrine pancreatic B-cells. Recent interest has focused on the development of this peptide as a novel therapeutic strategy for non-insulin-dependent (type 2) diabetes mellitus and associated neuropathy. H-6795 GLP-1 (7-36) amide (human, bovine, guinea pig, mouse, rat) (Glucagon- Like Peptide 1 (7-36) amide (human, bovine, guinea pig, mouse, rat)) 12
13 NEUROPEPTIDE Y (HUMAN, RAT) 200 μg/vial U-1230, H-Tyr-Pro-Ser-Lys-Pro-Asp-Asn-Pro- Gly-Glu-Asp-Ala-Pro-Ala-Glu-Asp-Met- Ala-Arg-Tyr-Tyr-Ser-Ala-Leu-Arg-His- Tyr-Ile-Asn-Leu-Ile-Thr-Arg-Gln-Arg- Tyr-NH 2 acetate salt C 189 H 285 N 55 O 57 S : g/mol CAS No: (net) Neuropeptide Y (NPY) is one of the most potent neurotransmitters in reproductive endocrine functions and endocrine functions regulating stress, metabolism, and eating behavior. H-6375 Neuropeptide Y (human, rat) (NPY (human, rat)) L-NMMA 250 mg/vial U-1090, Synonym L-NMA H-Arg(Me)-OH acetate salt C 7 H 16 N 4 O 2 : g/mol CAS No: (net), (acetate) L-NMMA inhibits the synthesis of nitric oxide (NO) in a dose-dependent and enantio-specific fashion. Intravenous or intra-arterial injections of L-NMMA produce a powerful and prolonged vasoconstrictor effect, which can be reversed by L-Arg in a dosedependent manner. E-3745 H-Arg(Me)-OH (L-NMMA, Tilarginine, Targinine) PENTAGASTRIN 50 μg/vial U-1100, Boc-β-Ala-Trp-Met-Asp-Phe-NH 2 C 37 H 49 N 7 O 9 S : g/mol CAS No: Pentagastrin stimulates the secretion of gastric acid. It is a highly selective CCK- 8 receptor agonist with panicogenic and anxiogenic properties. The peptide elicits symptoms and cardiovascular responses similar to those produced by CCK-4. A-1130 Pentagastrin (AY 6608, ICI 50123, NSC ) 13
14 Clinalfa SUBSTANCE P 50 μg/vial U-1180, H-Arg-Pro-Lys-Pro-Gln-Gln-Phe-Phe- Gly-Leu-Met-NH 2 acetate salt C 63 H 98 N 18 O 13 S : g/mol CAS No: (net) Substance P (SP) is a modulator of pain transmission. As an extremely potent vasodilator, SP produces significant bronchodilation in normal and asthmatic subjects and a maximal increase in forearm blood flow. H-1890 Substance P 14
15 CUSTOM MANUFACTURING: CLINALFA PLUS Clinalfa is Bachem s brand for its clinical trial materials and related services. Clinalfa plus products are sold exclusively to Authorized Persons and Companies. Clinalfa plus products are available as sterile freeze-dried products or as readyto-use liquid formulations. The products are manufactured and released based on the requirements laid down in the European Commission Directives 2001/20/EC and 2003/94/EC and the EU GMP guide including annex 13. The products contain active substances manufactured and released according to ICH Q7. Clinalfa plus products can be used in preclinical or clinical studies (clinical trials) provided that such clinical studies fully comply with the international ethical and scientific standards as stated in the ICH guideline for Good Clinical Practice E6 (R1). Clinalfa plus products are manufactured and released on a customized basis. Based on agreements between the customer and Bachem, Bachem s Qualified Person (QP) may relate the release of the products to the use in a specific clinical trial approved by the competent authority. For Clinalfa plus products the level of validation of the manufacturing process and analytical methods, the design and duration of post-release stability studies and the content of CMC documentation is customized based on the developmental stage and intended use of the products. Clinalfa products are manufactured under cgmp conditions. Please have a look at the respective certificates. For offers and supply of Clinalfa products and Clinalfa services, the General Conditions of Sale of the Bachem Group apply. CLINALFA SERVICES Bachem offers related services to Clinalfa basic and Clinalfa plus products, i.e. formulation development, compatibility studies, stress test and post-release stability studies, CMC documentation for IMPD and IND, drug master file and other services. Please contact us for more information. 15
16 Clinalfa SITE CERTIFICATIONS BACHEM AG Development and manufacturing of active pharmaceutical ingredients (APIs) Registered with and inspected by the FDA, Swissmedic, and other national authorities Licensed for the manufacture of APIs for the Japanese market Peptides and small organic molecules Manufacturing of catalog products Custom synthesis Production of peptides by solid-phase peptide synthesis (SPPS) and solution-phase chemistry BACHEM DISTRIBUTION SERVICES GMBH Production and release of sterile products for use in approved clinical trials (Clinalfa brand) Intra-Community tax supply Registered with and inspected by the Regierungspräsidium Tübingen 16
17 PRODUCT DISCLAIMER AGREEMENT Bachem Distribution Services GmbH, Hegenheimer Straße 5, Weil am Rhein, Germany Product Disclaimer Agreement between Bachem Distribution Service GmbH, Hegenheimer Strasse 5, Weil am Rhein, Germany (the Supplier) and ( full name and address ) (the Investigator). Whereas the Investigator wishes to buy from Supplier and Supplier wishes to sell to Investigator the following Clinalfa product (the Product): Product Category : Clinalfa basic Product Name : Product Number : To be used in the following the following study (the Study): Study Number/Name : EUDRACT Number : (for Europe only) IND Number : (for USA only) The Investigator acknowledges having been informed by the Supplier about general terms and conditions of sale, in particular that Clinalfa basic products - are not authorized for marketing and - must be used exclusively in human studies (clinical trials) that fully comply with the international ethical and scientific quality standards as stated in the ICH Guideline for Good Clinical Practice E6 (R1). The Investigator has also been informed and is aware that an equivalent medicinal product authorised for marketing may be on the market. The Investigator acknowledges that experimental conditions of the clinical trial (study) are to be established by, and are the sole and exclusive responsibility of the Investigator. The product is for use in trials where the product is not taken home by subjects. The supplier assumes no liability whatsoever for damage and/or loss resulting from the use of the product, except where such damage and/or loss arise from the failure of the Product to meet the specifications. The Investigator shall ensure that the documentation taken into account by Bachem s qualified person (QP) when certifying the batch is consistent with the information submitted to the competent authorities for clinical trial approval (study). Place and Date : Signature : (of the Principal Investigator or Sponsor) Name and Position: BW Bank Lörrach Steuer-Nr /09863 Konto-Nr UST.-Id.-Nr: DE BLZ Handelsregister Freiburg i. Br., HRB IBAN DE Geschäftsführer: Dr. Wolfgang Fritzen, BIC: SOLADEST Dr. Thomas Früh, Christian Schütz 17
18 Clinalfa GENERAL INFORMATION General Conditions of Sale and Delivery The general terms and conditions of sale and delivery currently in force at the Bachem Group of Companies in the USA and in Europe can be obtained from the following sources: 1. Bachem website The information is contained at our website and a pdf file can be downloaded via the link General Conditions of Sale at the footer of the web page or under Service & Support. 2. Bachem Customer Service on request For Europe, Asia, Australia, Africa Tel [email protected] For North and South America Tel Tel [email protected] 3. Quotations and invoices (on the reverse side of the page) Qualified Person All Clinalfa products are released by a qualified person (QP) in full compliance with the EU GMP requirements (Eudralex Vol. 4 - Guidelines for good manufacturing practices for medicinal products for human and veterinary use) for investigational medicinal products. Storing and Handling of Clinalfa Products The required storage temperature is clearly indicated on the label of each vial, on the Certificate of Analysis, and on the box in which the product is shipped. The reconstituted product can be kept for a few days if stored in a freezer at -18 C (please check with us). The freeze/thaw cycle may be repeated only once. The unused portion must be discarded. The reconstituted solution can only be maintained sterile and pyrogen-free if kept in the original Clinalfa vial. Use-by Date The use-by dates stated on the vials, boxes and Certificates of Analysis are the retest dates in accordance with our scheduled stability tests. Beyond this date a revised Certificate of Analysis is available upon request. Product Disclaimer Agreement By purchasing a Clinalfa product, the buyer acknowledges having been informed by the producer that the product sold has been designed and manufactured exclusively for and therefore may only be used in clinical research under conditions to be established by buyer under his sole and exclusive responsibility. Producer and supplier assumes no liability whatsoever for injury and/or loss resulting from the use of the products. Calculations (g versus mol) amount (g) / MW (g/mol) = mol Example: compound with MW=1 000 g/mol g 1 mol 1 g 1 mmol 1 mg 1 μmol 1 μg 1 nmol 1 ng 1 pmol References The publications where Clinalfa products have been used are constantly updated on our website. Please use these references to support your ethical committee applications. 18
19 1 1 PRODUCT BROCHURES AMYLOID PEPTIDES ANTIMICROBIAL PEPTIDES CALCITONIN GENE-RELATED PEPTIDES CASPASE SUBSTRATES INHIBITORS CYSTEINE DERIVATIVES DAP AND DAB DERIVATIVES DIABETES PEPTIDES ENDOTHELINS FRET SUBSTRATES GHRELIN, LEPTIN AND OBESTATIN LHRH AGONISTS AND ANTAGONISTS MATRIX METALLO- PROTEINASES MELANOMA PEPTIDES N-METHYLATED AMINO ACID DERIVATIVES NEUROPEPTIDE Y NON-IONIC DETERGENTS ORTHOGONALITY OF PROTECTING GROUPS PAR ACTIVATING PEPTIDES PEPTIDE YY PEPTIDES IN COSMETICS PRION PEPTIDES PSEUDOPROLINE DIPEPTIDES SECRETASE SUBSTRATES INHIBITORS VETERINARY PEPTIDES VIP/PACAP 19
20 Marketing & Sales Contact Europe, Africa, Middle East and Asia Pacific Bachem AG Tel Americas Bachem Americas, Inc. Tel (toll free in USA & Canada) Visit our website or shop online shop.bachem.com All information is compiled to the best of our knowledge. We cannot be made liable for any possible errors or misprints. Some products may be restricted in certain countries. shop.bachem.com published by Global Marketing, Bachem Group,November 2015
NEW CHEMICAL ENTITIES
 NEW CHEMICAL ENTITIES PIONEERING PARTNER FOR PEPTIDES With more than 40 years of expertise in peptide synthesis, a track record in process development, large-scale manufacturing and outstanding product
NEW CHEMICAL ENTITIES PIONEERING PARTNER FOR PEPTIDES With more than 40 years of expertise in peptide synthesis, a track record in process development, large-scale manufacturing and outstanding product
CUSTOM PEPTIDE SYNTHESIS AT BACHEM
 CUSTOM SYNTHESIS CUSTOM PEPTIDE SYNTHESIS AT BACHEM A strong commitment to quality is the basis of our long-standing market leadership We have the capability to synthesize peptides in mg to kg-scale which
CUSTOM SYNTHESIS CUSTOM PEPTIDE SYNTHESIS AT BACHEM A strong commitment to quality is the basis of our long-standing market leadership We have the capability to synthesize peptides in mg to kg-scale which
Describe how these hormones exert control quickly by changes in phosphorylation state of enzyme, and more slowly by changes of gene expression
 Section VIII. Section VIII. Tissue metabolism Many tissues carry out specialized functions: Ch. 43 look at different hormones affect metabolism of fuels, especially counter-insulin Ch. 44 Proteins and
Section VIII. Section VIII. Tissue metabolism Many tissues carry out specialized functions: Ch. 43 look at different hormones affect metabolism of fuels, especially counter-insulin Ch. 44 Proteins and
H-5118.0010 ([125I]-Tyr)-ACTH (18-39) (human) CE-marked Liquid 10.0 µci on request -20 C DI
![H-5118.0010 ([125I]-Tyr)-ACTH (18-39) (human) CE-marked Liquid 10.0 µci on request -20 C DI H-5118.0010 ([125I]-Tyr)-ACTH (18-39) (human) CE-marked Liquid 10.0 µci on request -20 C DI](/thumbs/24/3463252.jpg) Bachem Immunology Product Prices from MoBiTec (Rev. 05/28/2013) Please note: Prices subject to change without prior notice -- Not responsible for typographical errors -- all prices are in EURO excl. VAT
Bachem Immunology Product Prices from MoBiTec (Rev. 05/28/2013) Please note: Prices subject to change without prior notice -- Not responsible for typographical errors -- all prices are in EURO excl. VAT
Sweet-taste receptors, glucose absorption and insulin release: Are LCS nutritionally active?
 Sweet-taste receptors, glucose absorption and insulin release: Are LCS nutritionally active? Samuel V. Molinary, Ph.D. Consultant, Scientific & Regulatory Affairs ILSI/NA April 6, 2011 Washington, DC Why
Sweet-taste receptors, glucose absorption and insulin release: Are LCS nutritionally active? Samuel V. Molinary, Ph.D. Consultant, Scientific & Regulatory Affairs ILSI/NA April 6, 2011 Washington, DC Why
INTRODUCTION TO HORMONES
 INTRODUCTION TO HORMONES UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY PBL MBBS II SEMINAR VJ Temple What are hormones? Cells in multi-cellular
INTRODUCTION TO HORMONES UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY PBL MBBS II SEMINAR VJ Temple What are hormones? Cells in multi-cellular
Endocrine Glands and the General Principles of Hormone Action
 Endocrine Glands and the General Principles of Hormone Action Cai Li, Ph.D. Assistant professor Touchstone Center for Diabetes Research Departments of Physiology and Internal Medicine The University of
Endocrine Glands and the General Principles of Hormone Action Cai Li, Ph.D. Assistant professor Touchstone Center for Diabetes Research Departments of Physiology and Internal Medicine The University of
ICH Topic S 1 A The Need for Carcinogenicity Studies of Pharmaceuticals. Step 5
 European Medicines Agency July 1996 CPMP/ICH/140/95 ICH Topic S 1 A The Need for Carcinogenicity Studies of Pharmaceuticals Step 5 NOTE FOR GUIDANCE ON THE NEED FOR CARCINOGENICITY STUDIES OF PHARMACEUTICALS
European Medicines Agency July 1996 CPMP/ICH/140/95 ICH Topic S 1 A The Need for Carcinogenicity Studies of Pharmaceuticals Step 5 NOTE FOR GUIDANCE ON THE NEED FOR CARCINOGENICITY STUDIES OF PHARMACEUTICALS
glucose and fatty acids to raise your blood sugar levels.
 Endocrine & Cell Communication Part IV: Maintaining Balance (Homeostasis) TEACHER NOTES needs coding 1 Endocrine & Cell Communication Part IV: Maintaining Balance (Homeostasis) 2 AP Biology Curriculum
Endocrine & Cell Communication Part IV: Maintaining Balance (Homeostasis) TEACHER NOTES needs coding 1 Endocrine & Cell Communication Part IV: Maintaining Balance (Homeostasis) 2 AP Biology Curriculum
Hormonal Cycles. 1. Briefly describe each component of an endocrine feedback loop: Stimulus. Production Cell. Hormone. Target Cell. Target Cell Action
 Hormonal Cycles Directions: a. Click the Contents button. b. Open the Endocrine System File. c. Click Animations. d. Click Hormonal Cycles. 1. Briefly describe each component of an endocrine feedback loop:
Hormonal Cycles Directions: a. Click the Contents button. b. Open the Endocrine System File. c. Click Animations. d. Click Hormonal Cycles. 1. Briefly describe each component of an endocrine feedback loop:
Regulation of Metabolism. By Dr. Carmen Rexach Physiology Mt San Antonio College
 Regulation of Metabolism By Dr. Carmen Rexach Physiology Mt San Antonio College Energy Constant need in living cells Measured in kcal carbohydrates and proteins = 4kcal/g Fats = 9kcal/g Most diets are
Regulation of Metabolism By Dr. Carmen Rexach Physiology Mt San Antonio College Energy Constant need in living cells Measured in kcal carbohydrates and proteins = 4kcal/g Fats = 9kcal/g Most diets are
Nursing 113. Pharmacology Principles
 Nursing 113 Pharmacology Principles 1. The study of how drugs enter the body, reach the site of action, and are removed from the body is called a. pharmacotherapeutics b. pharmacology c. pharmacodynamics
Nursing 113 Pharmacology Principles 1. The study of how drugs enter the body, reach the site of action, and are removed from the body is called a. pharmacotherapeutics b. pharmacology c. pharmacodynamics
Digestion, Absorption. How & where?
 Digestion, Absorption How & where? What happens to food? Three processes Digestion Absorption Elimination Where do they occur? GI tract Overview of Digestion GI tract Gastrointestinal (GI) tract: series
Digestion, Absorption How & where? What happens to food? Three processes Digestion Absorption Elimination Where do they occur? GI tract Overview of Digestion GI tract Gastrointestinal (GI) tract: series
DRUGS FOR GLUCOSE MANAGEMENT AND DIABETES
 Page 1 DRUGS FOR GLUCOSE MANAGEMENT AND DIABETES Drugs to know are: Actrapid HM Humulin R, L, U Penmix SUNALI MEHTA The three principal hormones produced by the pancreas are: Insulin: nutrient metabolism:
Page 1 DRUGS FOR GLUCOSE MANAGEMENT AND DIABETES Drugs to know are: Actrapid HM Humulin R, L, U Penmix SUNALI MEHTA The three principal hormones produced by the pancreas are: Insulin: nutrient metabolism:
Blood Pressure Regulation
 Blood Pressure Regulation Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.com) Page 1. Introduction There are two basic mechanisms for regulating
Blood Pressure Regulation Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.com) Page 1. Introduction There are two basic mechanisms for regulating
C 5. chemical development contract research custom synthesis cgmp API manufacturing commercial production. Welcome to
 C 5 chemical development contract research custom synthesis cgmp API manufacturing commercial production Welcome to ChemCon Company profile Company profile ChemCon offers outstanding chemical services
C 5 chemical development contract research custom synthesis cgmp API manufacturing commercial production Welcome to ChemCon Company profile Company profile ChemCon offers outstanding chemical services
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE S1A. Current Step 4 version
 INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE GUIDELINE ON THE NEED FOR CARCINOGENICITY STUDIES
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE GUIDELINE ON THE NEED FOR CARCINOGENICITY STUDIES
Digestion, Absorption. How & where?
 Digestion, Absorption How & where? What happens to food? Three processes Digestion Absorption Elimination Where do they occur? GI tract Overview of Digestion GI tract Gastrointestinal (GI) tract: series
Digestion, Absorption How & where? What happens to food? Three processes Digestion Absorption Elimination Where do they occur? GI tract Overview of Digestion GI tract Gastrointestinal (GI) tract: series
FACT SHEET TESTETROL, A NOVEL ORALLY BIOACTIVE ANDROGEN
 FACT SHEET TESTETROL, A NOVEL ORALLY BIOACTIVE ANDROGEN General Pantarhei Bioscience B.V. is an emerging specialty pharmaceutical company with a creative approach towards drug development. The Company
FACT SHEET TESTETROL, A NOVEL ORALLY BIOACTIVE ANDROGEN General Pantarhei Bioscience B.V. is an emerging specialty pharmaceutical company with a creative approach towards drug development. The Company
Managing Joint Venture Arrangements Between Two Biotech Companies: A Case Report. Dr. Jan Mous Midatech / PharMida May 21 st 2014
 Managing Joint Venture Arrangements Between Two Biotech Companies: A Case Report Dr. Jan Mous Midatech / PharMida May 21 st 2014 1 Midatech Ltd. Corporate verview Founded 2000 in xford, England Drug development
Managing Joint Venture Arrangements Between Two Biotech Companies: A Case Report Dr. Jan Mous Midatech / PharMida May 21 st 2014 1 Midatech Ltd. Corporate verview Founded 2000 in xford, England Drug development
Water Homeostasis. Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.
 Water Homeostasis Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.com) 1. Water Homeostasis The body maintains a balance of water intake
Water Homeostasis Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.com) 1. Water Homeostasis The body maintains a balance of water intake
Hormones & Chemical Signaling
 Hormones & Chemical Signaling Part 2 modulation of signal pathways and hormone classification & function How are these pathways controlled? Receptors are proteins! Subject to Specificity of binding Competition
Hormones & Chemical Signaling Part 2 modulation of signal pathways and hormone classification & function How are these pathways controlled? Receptors are proteins! Subject to Specificity of binding Competition
SMALL AND LARGE INTESTINE SECRETIONS
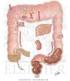 SMALL AND LARGE INTESTINE SECRETIONS Objectives At the end of lecture student should be able to know, Digestive system Digestive system secretions Small intestine Component of small intestine Intestinal
SMALL AND LARGE INTESTINE SECRETIONS Objectives At the end of lecture student should be able to know, Digestive system Digestive system secretions Small intestine Component of small intestine Intestinal
Endocrine Responses to Resistance Exercise
 chapter 3 Endocrine Responses to Resistance Exercise Chapter Objectives Understand basic concepts of endocrinology. Explain the physiological roles of anabolic hormones. Describe hormonal responses to
chapter 3 Endocrine Responses to Resistance Exercise Chapter Objectives Understand basic concepts of endocrinology. Explain the physiological roles of anabolic hormones. Describe hormonal responses to
Response to Stress Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.
 Response to Stress Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.com) Page 1. Introduction When there is an overwhelming threat to the
Response to Stress Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.com) Page 1. Introduction When there is an overwhelming threat to the
Neural Systems Involved in Food Intake: An Integrative Overview
 Neural Systems Involved in Food Intake: An Integrative Overview Stephen C. Woods* Department of Psychiatry and Behavioral Neuroscience University of Cincinnati * No conflicts to report ILSI North America
Neural Systems Involved in Food Intake: An Integrative Overview Stephen C. Woods* Department of Psychiatry and Behavioral Neuroscience University of Cincinnati * No conflicts to report ILSI North America
Weight Loss Surgery and Bariatric Nutrition. Jeanine Giordano, MS, RD, CDN
 Weight Loss urgery and Bariatric Nutrition Jeanine Giordano, M, RD, CDN UA: Mean BMI trends (age standardized) Prevalence of Obesity Among Adults United tates 68% Australia 59% Russia 54% United Kingdom
Weight Loss urgery and Bariatric Nutrition Jeanine Giordano, M, RD, CDN UA: Mean BMI trends (age standardized) Prevalence of Obesity Among Adults United tates 68% Australia 59% Russia 54% United Kingdom
The Endocrine System
 Essentials of Human Anatomy & Physiology Elaine N. Marieb Seventh Edition Chapter 9 The Endocrine System Slides 9.1 9.48 Lecture Slides in PowerPoint by Jerry L. Cook The Endocrine System Second messenger
Essentials of Human Anatomy & Physiology Elaine N. Marieb Seventh Edition Chapter 9 The Endocrine System Slides 9.1 9.48 Lecture Slides in PowerPoint by Jerry L. Cook The Endocrine System Second messenger
New Insights and New Therapies for Insulin Resistance
 New Insights and New Therapies for Insulin Resistance by: Johan H Koeslag Medical Physiology University of Stellenbosch PO Box 19063 Tygerberg, 7505. South Africa and: Peter T Saunders Department of Mathematics
New Insights and New Therapies for Insulin Resistance by: Johan H Koeslag Medical Physiology University of Stellenbosch PO Box 19063 Tygerberg, 7505. South Africa and: Peter T Saunders Department of Mathematics
Extemporaneously Prepared Early Phase Clinical Trial Materials
 Extemporaneously Prepared Early Phase Clinical Trial Materials Richard Hoffman, MS, RAC Eli Lilly & Co. Regulatory Advisor International Consortium for Innovation & Quality in Pharmaceutical Development
Extemporaneously Prepared Early Phase Clinical Trial Materials Richard Hoffman, MS, RAC Eli Lilly & Co. Regulatory Advisor International Consortium for Innovation & Quality in Pharmaceutical Development
SAFETY PHARMACOLOGY STUDIES FOR HUMAN PHARMACEUTICALS S7A
 INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE SAFETY PHARMACOLOGY STUDIES FOR HUMAN PHARMACEUTICALS
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE SAFETY PHARMACOLOGY STUDIES FOR HUMAN PHARMACEUTICALS
BioBusiness Biopharmaceutical Ingredients Recombinant albumins & albumin fusion Dermot Pearson, Strategic Product Director, NZ Biopharma
 BioBusiness Biopharmaceutical Ingredients Recombinant albumins & albumin fusion Dermot Pearson, Strategic Product Director, NZ Biopharma 2 Novozymes solutions dedicated to the biopharmaceutical industry
BioBusiness Biopharmaceutical Ingredients Recombinant albumins & albumin fusion Dermot Pearson, Strategic Product Director, NZ Biopharma 2 Novozymes solutions dedicated to the biopharmaceutical industry
regulation of ECF composition and volume regulation of metabolism thyroid hormones, epinephrine, growth hormone, insulin and glucagon
 Hormonal Effects regulation of ECF composition and volume ADH, aldosterone, ANF regulation of metabolism thyroid hormones, epinephrine, growth hormone, insulin and glucagon regulation of muscle contraction
Hormonal Effects regulation of ECF composition and volume ADH, aldosterone, ANF regulation of metabolism thyroid hormones, epinephrine, growth hormone, insulin and glucagon regulation of muscle contraction
The digestive system eliminated waste from the digestive tract. But we also need a way to eliminate waste from the rest of the body.
 Outline Urinary System Urinary System and Excretion Bio105 Lecture 20 Chapter 16 I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure III. Disorders of the urinary system 1
Outline Urinary System Urinary System and Excretion Bio105 Lecture 20 Chapter 16 I. Function II. Organs of the urinary system A. Kidneys 1. Function 2. Structure III. Disorders of the urinary system 1
EuroPeptides 2014: Workshop Considerations for Peptide Contract Manufacturing: Case Study on Scale-up Considerations
 EuroPeptides 2014: Workshop Considerations for Peptide Contract Manufacturing: Case Study on Scale-up Considerations Bruce H Morimoto, PhD Executive Director, Applied Translational Medicine Disclaimer
EuroPeptides 2014: Workshop Considerations for Peptide Contract Manufacturing: Case Study on Scale-up Considerations Bruce H Morimoto, PhD Executive Director, Applied Translational Medicine Disclaimer
The diagram below summarizes the effects of the compounds that cells use to regulate their own metabolism.
 Regulation of carbohydrate metabolism Intracellular metabolic regulators Each of the control point steps in the carbohydrate metabolic pathways in effect regulates itself by responding to molecules that
Regulation of carbohydrate metabolism Intracellular metabolic regulators Each of the control point steps in the carbohydrate metabolic pathways in effect regulates itself by responding to molecules that
Endocrine System Review Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.
 Endocrine System Review Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.com) Page 1. Endocrine System Review Consists of discrete individual
Endocrine System Review Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.com) Page 1. Endocrine System Review Consists of discrete individual
GLUCOSE HOMEOSTASIS-II: An Overview
 GLUCOSE HOMEOSTASIS-II: An Overview University of Papua New Guinea School of Medicine & Health Sciences, Division of Basic Medical Sciences Discipline of Biochemistry & Molecular Biology, M Med Part I
GLUCOSE HOMEOSTASIS-II: An Overview University of Papua New Guinea School of Medicine & Health Sciences, Division of Basic Medical Sciences Discipline of Biochemistry & Molecular Biology, M Med Part I
Introduction to Enteris BioPharma
 Introduction to Enteris BioPharma Enteris BioPharma Intelligent Solutions for Oral Drug Delivery Privately held, New Jersey based biotech company Owned solely by Victory Park Capital, a large Chicago based
Introduction to Enteris BioPharma Enteris BioPharma Intelligent Solutions for Oral Drug Delivery Privately held, New Jersey based biotech company Owned solely by Victory Park Capital, a large Chicago based
Chapter-21b: Hormones and Receptors
 1 hapter-21b: Hormones and Receptors Hormone classes Hormones are classified according to the distance over which they act. 1. Autocrine hormones --- act on the same cell that released them. Interleukin-2
1 hapter-21b: Hormones and Receptors Hormone classes Hormones are classified according to the distance over which they act. 1. Autocrine hormones --- act on the same cell that released them. Interleukin-2
Chapter 45: Hormones and the Endocrine System
 Name Period Overview 1. What is a hormone? 2. Why does a hormone elicit a response only with target cells? 3. The body has two long-distance regulating systems. Which involves chemical signals by hormones?
Name Period Overview 1. What is a hormone? 2. Why does a hormone elicit a response only with target cells? 3. The body has two long-distance regulating systems. Which involves chemical signals by hormones?
Milwaukee School of Engineering [email protected]. Case Study: Factors that Affect Blood Pressure Instructor Version
 Case Study: Factors that Affect Blood Pressure Instructor Version Goal This activity (case study and its associated questions) is designed to be a student-centered learning activity relating to the factors
Case Study: Factors that Affect Blood Pressure Instructor Version Goal This activity (case study and its associated questions) is designed to be a student-centered learning activity relating to the factors
The Background for the Diabetes Detection Model
 The Background for the Diabetes Detection Model James K. Peterson Department of Biological Sciences and Department of Mathematical Sciences Clemson University November 23, 2014 Outline The Background for
The Background for the Diabetes Detection Model James K. Peterson Department of Biological Sciences and Department of Mathematical Sciences Clemson University November 23, 2014 Outline The Background for
Public Assessment Report. Decentralised Procedure
 Public Assessment Report Decentralised Procedure TELMISARTAN DR REDDY S 20 MG TABLETS TELMISARTAN DR REDDY S 40 MG TABLETS TELMISARTAN DR REDDY S 80 MG TABLETS (telmisartan) Procedure No: UK/H/5034/001-003/DC
Public Assessment Report Decentralised Procedure TELMISARTAN DR REDDY S 20 MG TABLETS TELMISARTAN DR REDDY S 40 MG TABLETS TELMISARTAN DR REDDY S 80 MG TABLETS (telmisartan) Procedure No: UK/H/5034/001-003/DC
Comparative analysis between the possible regulatory approaches to GMP compliance TITOLO PRESENTAZIONE
 Comparative analysis between the possible regulatory approaches to GMP compliance TITOLO PRESENTAZIONE Dr. Fulvio CARLOTTI, GNOSIS SpA, Corporate QA Director September 26, 2014 Scope of GMP GMP compliance
Comparative analysis between the possible regulatory approaches to GMP compliance TITOLO PRESENTAZIONE Dr. Fulvio CARLOTTI, GNOSIS SpA, Corporate QA Director September 26, 2014 Scope of GMP GMP compliance
The Vertebrate (mostly human) Digestive System
 The Vertebrate (mostly human) Digestive System Mouth - mastication, lubrication, digestion Pharynx and Esophagus - swallowing Stomach - some digestion Small intestine - most digestion and absorption Large
The Vertebrate (mostly human) Digestive System Mouth - mastication, lubrication, digestion Pharynx and Esophagus - swallowing Stomach - some digestion Small intestine - most digestion and absorption Large
Comparative Studies and Metabolic Effects of Sleeve Gastrectomy
 Comparative Studies and Metabolic Effects of Sleeve Gastrectomy Alfonso Torquati MD, MSCI Associate Professor of Surgery Discosures NIH-NIDDK: grant support Covidien: consulting agreement, grant support
Comparative Studies and Metabolic Effects of Sleeve Gastrectomy Alfonso Torquati MD, MSCI Associate Professor of Surgery Discosures NIH-NIDDK: grant support Covidien: consulting agreement, grant support
Ch16 Endocrine part 2
 Ch16 Endocrine part 2 several separate organs release hormones into capillaries hormones are transported in the blood Hypothalamus Pituitary glands Pineal gland Thyroid Parathyroid Thymus Adrenal Cortex
Ch16 Endocrine part 2 several separate organs release hormones into capillaries hormones are transported in the blood Hypothalamus Pituitary glands Pineal gland Thyroid Parathyroid Thymus Adrenal Cortex
INSULIN AND INCRETIN THERAPIES: WHAT COMBINATIONS ARE RIGHT FOR YOUR PATIENT?
 INSULIN AND INCRETIN THERAPIES: WHAT COMBINATIONS ARE RIGHT FOR YOUR PATIENT? MARTHA M. BRINSKO, MSN, ANP-BC CHARLOTTE COMMUNITY HEALTH CLINIC CHARLOTTE, NC Diagnosed and undiagnosed diabetes in the United
INSULIN AND INCRETIN THERAPIES: WHAT COMBINATIONS ARE RIGHT FOR YOUR PATIENT? MARTHA M. BRINSKO, MSN, ANP-BC CHARLOTTE COMMUNITY HEALTH CLINIC CHARLOTTE, NC Diagnosed and undiagnosed diabetes in the United
Decentralised Procedure. Public Assessment Report
 Decentralised Procedure Public Assessment Report Articainhydrochlorid mit Epinephrin Pierrel 40 mg/ml + 0.01 mg/ml Injektionslösung Articainhydrochlorid mit Epinephrin Pierrel 40 mg/ml + 0.005 mg/ml Injektionslösung
Decentralised Procedure Public Assessment Report Articainhydrochlorid mit Epinephrin Pierrel 40 mg/ml + 0.01 mg/ml Injektionslösung Articainhydrochlorid mit Epinephrin Pierrel 40 mg/ml + 0.005 mg/ml Injektionslösung
Methods of Grading S/N Style of grading Percentage Score 1 Attendance, class work and assignment 10 2 Test 20 3 Examination 70 Total 100
 COURSE: MIB 303 Microbial Physiology and Metabolism (3 Units- Compulsory) Course Duration: Three hours per week for 15 weeks (45 hours). Lecturer: Jimoh, S.O. B.Sc., M.Sc, Ph.D Microbiology (ABU, Zaria)
COURSE: MIB 303 Microbial Physiology and Metabolism (3 Units- Compulsory) Course Duration: Three hours per week for 15 weeks (45 hours). Lecturer: Jimoh, S.O. B.Sc., M.Sc, Ph.D Microbiology (ABU, Zaria)
INSULIN RESISTANCE, POLYCYSTIC OVARIAN SYNDROME
 1 University of Papua New Guinea School of Medicine and Health Sciences Division of Basic Medical Sciences Discipline of Biochemistry and Molecular Biology PBL SEMINAR INSULIN RESISTANCE, POLYCYSTIC OVARIAN
1 University of Papua New Guinea School of Medicine and Health Sciences Division of Basic Medical Sciences Discipline of Biochemistry and Molecular Biology PBL SEMINAR INSULIN RESISTANCE, POLYCYSTIC OVARIAN
Introduction. Pathogenesis of type 2 diabetes
 Introduction Type 2 diabetes mellitus (t2dm) is the most prevalent form of diabetes worldwide. It is characterised by high fasting and high postprandial blood glucose concentrations (hyperglycemia). Chronic
Introduction Type 2 diabetes mellitus (t2dm) is the most prevalent form of diabetes worldwide. It is characterised by high fasting and high postprandial blood glucose concentrations (hyperglycemia). Chronic
New and Future Treatments for Diabetes. Mary Charlton Specialty Doctor in Diabetes University Hospital Birmingham BARS Oct 2014
 New and Future Treatments for Diabetes Mary Charlton Specialty Doctor in Diabetes University Hospital Birmingham BARS Oct 2014 Conflicts of interest Investigator Carmelina study of Linagliptin (Boehringer
New and Future Treatments for Diabetes Mary Charlton Specialty Doctor in Diabetes University Hospital Birmingham BARS Oct 2014 Conflicts of interest Investigator Carmelina study of Linagliptin (Boehringer
Chapter 18. An Introduction to the Endocrine System. Hormone Chemistry
 Chapter 18 An Introduction to the Endocrine System Hormone Chemistry Endocrine System Components endocrine system - glands, tissues, and cells that secrete hormones Copyright The McGraw-Hill Companies,
Chapter 18 An Introduction to the Endocrine System Hormone Chemistry Endocrine System Components endocrine system - glands, tissues, and cells that secrete hormones Copyright The McGraw-Hill Companies,
Give a NOD to diabetes:
 Give a NOD to diabetes: NOD proteins ti link immunity it and metabolism tbli Jonathan Schertzer McMaster University McMaster University Faculty of Health Sciences Department of Biochemistry and Biomedical
Give a NOD to diabetes: NOD proteins ti link immunity it and metabolism tbli Jonathan Schertzer McMaster University McMaster University Faculty of Health Sciences Department of Biochemistry and Biomedical
QUESTIONS AND ANSWERS ABOUT THE EDQM ACTIVITIES
 QUESTIONS AND ANSWERS ABOUT THE EDQM ACTIVITIES Why are Pharmacopoeias so important in a globalised world? Pharmacopoeias have historically provided collections of medical recipes intended to ensure accurate
QUESTIONS AND ANSWERS ABOUT THE EDQM ACTIVITIES Why are Pharmacopoeias so important in a globalised world? Pharmacopoeias have historically provided collections of medical recipes intended to ensure accurate
Effect of liraglutide on body weight in overweight or obese subjects with type 2 diabetes: SCALE - Diabetes
 Effect of liraglutide on body weight in overweight or obese subjects with type 2 diabetes: SCALE - Diabetes This trial is conducted in Africa, Asia, Europe and the United States of America (USA). The aim
Effect of liraglutide on body weight in overweight or obese subjects with type 2 diabetes: SCALE - Diabetes This trial is conducted in Africa, Asia, Europe and the United States of America (USA). The aim
Endocrine System: Practice Questions #1
 Endocrine System: Practice Questions #1 1. Removing part of gland D would most likely result in A. a decrease in the secretions of other glands B. a decrease in the blood calcium level C. an increase in
Endocrine System: Practice Questions #1 1. Removing part of gland D would most likely result in A. a decrease in the secretions of other glands B. a decrease in the blood calcium level C. an increase in
Biology 12 June 2003 Provincial Examination
 Biology 12 June 2003 rovincial Examination ANWER KEY / CORING GUIDE CURRICULUM: Organizers 1. Cell Biology 2. Cell rocesses and Applications 3. Human Biology ub-organizers A, B, C, D E, F, G, H I, J, K,
Biology 12 June 2003 rovincial Examination ANWER KEY / CORING GUIDE CURRICULUM: Organizers 1. Cell Biology 2. Cell rocesses and Applications 3. Human Biology ub-organizers A, B, C, D E, F, G, H I, J, K,
Abdulaziz Al-Subaie. Anfal Al-Shalwi
 Abdulaziz Al-Subaie Anfal Al-Shalwi Introduction what is diabetes mellitus? A chronic metabolic disorder characterized by high blood glucose level caused by insulin deficiency and sometimes accompanied
Abdulaziz Al-Subaie Anfal Al-Shalwi Introduction what is diabetes mellitus? A chronic metabolic disorder characterized by high blood glucose level caused by insulin deficiency and sometimes accompanied
NO More Heart Disease
 NO More Heart Disease Nitric Oxide Information NO is one of the simplest molecules in biology, comprised of just two atoms one atom of nitrogen (N) and one of oxygen (O). Through NO s structure is simple,
NO More Heart Disease Nitric Oxide Information NO is one of the simplest molecules in biology, comprised of just two atoms one atom of nitrogen (N) and one of oxygen (O). Through NO s structure is simple,
Urinary System. And Adrenal Function
 Urinary System And Adrenal Function Overview Kidney anatomy and physiology Urine Ureters, Bladder and Urethra Adrenal Function Functions of the Kidney Filter fluids from the blood Regulate volume and composition
Urinary System And Adrenal Function Overview Kidney anatomy and physiology Urine Ureters, Bladder and Urethra Adrenal Function Functions of the Kidney Filter fluids from the blood Regulate volume and composition
BSc in Medical Sciences with PHARMACOLOGY
 BSc in Medical Sciences with PHARMACOLOGY Course Director Dr Christopher John Module Leaders Dr Robert Dickinson (Module 1) Dr Anabel Varela Carver (Module 2) Dr Sohag Saleh (Module 3) Course Administrator
BSc in Medical Sciences with PHARMACOLOGY Course Director Dr Christopher John Module Leaders Dr Robert Dickinson (Module 1) Dr Anabel Varela Carver (Module 2) Dr Sohag Saleh (Module 3) Course Administrator
EURO DIAGNOSTICA PRODUCT CATALOG RADIOIMMUNOASSAY (RIA)
 EURO DIAGNOSTICA PRODUCT CATALOG RADIOIMMUNOASSAY (RIA) Radioimmunoassay Catalog Euro Diagnostica We are proud to introduce the 2013/2014 RIA Catalog Euro Diagnostica is an international diagnostic solutions
EURO DIAGNOSTICA PRODUCT CATALOG RADIOIMMUNOASSAY (RIA) Radioimmunoassay Catalog Euro Diagnostica We are proud to introduce the 2013/2014 RIA Catalog Euro Diagnostica is an international diagnostic solutions
EXPLORING THE INTERACTION BETWEEN EXERCISE AND MEDICATION FOR CHRONIC DISEASE: CONSIDERATIONS FOR FITNESS PROFESSIONALS
 EXPLORING THE INTERACTION BETWEEN EXERCISE AND MEDICATION FOR CHRONIC DISEASE: CONSIDERATIONS FOR FITNESS PROFESSIONALS Steven T. Johnson, PhD 1 Introduction The prevalence of chronic diseases like cardiovascular
EXPLORING THE INTERACTION BETWEEN EXERCISE AND MEDICATION FOR CHRONIC DISEASE: CONSIDERATIONS FOR FITNESS PROFESSIONALS Steven T. Johnson, PhD 1 Introduction The prevalence of chronic diseases like cardiovascular
Diabetes? Does Metabolic Surgery. Experts disagree about how surgery treats diabetes but agree more research needs to be done.
 Does Metabolic Surgery The combination of type 2 diabetes and being significantly overweight is a huge burden. Doctors tell you to lose weight, in essence, to save your life. Weight loss, in addition to
Does Metabolic Surgery The combination of type 2 diabetes and being significantly overweight is a huge burden. Doctors tell you to lose weight, in essence, to save your life. Weight loss, in addition to
Chapter 4. The Adrenal Medulla
 Chapter 4 The Adrenal Medulla Introduction Induction of cortisol production requires several minutes, and full elaboration of cortisol action requires several hours. In contrast, the adrenal medullary
Chapter 4 The Adrenal Medulla Introduction Induction of cortisol production requires several minutes, and full elaboration of cortisol action requires several hours. In contrast, the adrenal medullary
Catalent Biologics & Clinical Supplies The SMART Solution
 Catalent Biologics & Clinical Supplies The SMART Solution Advanced Technology and Integrated Solutions From DNA to Clinical Supply & Cold Chain Distribution Dr. Florian Schwaak Account Manager Germany
Catalent Biologics & Clinical Supplies The SMART Solution Advanced Technology and Integrated Solutions From DNA to Clinical Supply & Cold Chain Distribution Dr. Florian Schwaak Account Manager Germany
Actions of Hormones on Target Cells Page 1. Actions of Hormones on Target Cells Page 2. Goals/ What You Need to Know Goals What You Need to Know
 Actions of Hormones on Target Cells Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.com) Page 1. Actions of Hormones on Target Cells Hormones
Actions of Hormones on Target Cells Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.com) Page 1. Actions of Hormones on Target Cells Hormones
Integration and Coordination of the Human Body. Nervous System
 I. General Info Integration and Coordination of the Human Body A. Both the and system are responsible for maintaining 1. Homeostasis is the process by which organisms keep internal conditions despite changes
I. General Info Integration and Coordination of the Human Body A. Both the and system are responsible for maintaining 1. Homeostasis is the process by which organisms keep internal conditions despite changes
US Regulations for Import and Export of Cell Therapy Products
 US Regulations for Import and Export of Cell Therapy Products Kurt Gunter, MD U Minnesota UCB Transplants (2001-2007)* Total units: 790 Number US banks: 19 Number non-us banks: 12 % units from non-us banks:
US Regulations for Import and Export of Cell Therapy Products Kurt Gunter, MD U Minnesota UCB Transplants (2001-2007)* Total units: 790 Number US banks: 19 Number non-us banks: 12 % units from non-us banks:
Cardiovascular disease physiology. Linda Lowe-Krentz Bioscience in the 21 st Century October 14, 2011
 Cardiovascular disease physiology Linda Lowe-Krentz Bioscience in the 21 st Century October 14, 2011 Content Introduction The number 1 killer in America Some statistics Recommendations The disease process
Cardiovascular disease physiology Linda Lowe-Krentz Bioscience in the 21 st Century October 14, 2011 Content Introduction The number 1 killer in America Some statistics Recommendations The disease process
D.U.C. Assist. Lec. Faculty of Dentistry General Physiology Ihsan Dhari. The Autonomic Nervous System
 The Autonomic Nervous System The portion of the nervous system that controls most visceral functions of the body is called the autonomic nervous system. This system helps to control arterial pressure,
The Autonomic Nervous System The portion of the nervous system that controls most visceral functions of the body is called the autonomic nervous system. This system helps to control arterial pressure,
Valentina Gualato, Ph.D. Process Development Scientist
 COMPANY PRESENTATION Quality and Innovation Valentina Gualato, Ph.D. Process Development Scientist MISSION areta international is a biotech company dedicated to the contract development and manufacturing
COMPANY PRESENTATION Quality and Innovation Valentina Gualato, Ph.D. Process Development Scientist MISSION areta international is a biotech company dedicated to the contract development and manufacturing
VPM 152. INFLAMMATION: Chemical Mediators
 General Pathology VPM 152 INFLAMMATION: Chemical Mediators CHEMICAL MEDIATORS OF INFLAMMATION Definition: any messenger that acts on blood vessels, inflammatory cells or other cells to contribute to an
General Pathology VPM 152 INFLAMMATION: Chemical Mediators CHEMICAL MEDIATORS OF INFLAMMATION Definition: any messenger that acts on blood vessels, inflammatory cells or other cells to contribute to an
COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) NOTE FOR GUIDANCE ON THE PRE-CLINICAL EVALUATION OF ANTICANCER MEDICINAL PRODUCTS
 The European Agency for the Evaluation of Medicinal Products Human Medicines Evaluation Unit London, 23 July 1998 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) NOTE FOR GUIDANCE ON THE PRE-CLINICAL
The European Agency for the Evaluation of Medicinal Products Human Medicines Evaluation Unit London, 23 July 1998 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) NOTE FOR GUIDANCE ON THE PRE-CLINICAL
Chapter 25: Metabolism and Nutrition
 Chapter 25: Metabolism and Nutrition Chapter Objectives INTRODUCTION 1. Generalize the way in which nutrients are processed through the three major metabolic fates in order to perform various energetic
Chapter 25: Metabolism and Nutrition Chapter Objectives INTRODUCTION 1. Generalize the way in which nutrients are processed through the three major metabolic fates in order to perform various energetic
High Potent Drugs & Containment Technology
 High Potent Drugs & Containment Technology Technology Overview & Quality Risk-Based Design Selection Holger Fabritz Head of Quality & Validation Assurance Part 1 Definition High Potent Drugs, OEL & hazardous
High Potent Drugs & Containment Technology Technology Overview & Quality Risk-Based Design Selection Holger Fabritz Head of Quality & Validation Assurance Part 1 Definition High Potent Drugs, OEL & hazardous
Yamaguchi University, Japan
 Yamaguchi University, Japan The United Graduate School of Veterinary Science The Stress Related Neuroendocrine and Metabolic Effects of Alpha-2 Adrenergic Agents and Their Combinations with Injectable
Yamaguchi University, Japan The United Graduate School of Veterinary Science The Stress Related Neuroendocrine and Metabolic Effects of Alpha-2 Adrenergic Agents and Their Combinations with Injectable
Dr. RAJENDRAN S INSTITUTE OF MEDICAL EDUCATION
 Page 1 of 7 Dr. RAJENDRAN S INSTITUTE OF MEDICAL EDUCATION AIIMS NOVEMBER 2012 - QUESTIONS AND ANSWERS PHYSIOLOGY This contains only 3 out of 7 questions. For complete questions with explanatory answers,
Page 1 of 7 Dr. RAJENDRAN S INSTITUTE OF MEDICAL EDUCATION AIIMS NOVEMBER 2012 - QUESTIONS AND ANSWERS PHYSIOLOGY This contains only 3 out of 7 questions. For complete questions with explanatory answers,
7 Answers to end-of-chapter questions
 7 Answers to end-of-chapter questions Multiple choice questions 1 B 2 B 3 A 4 B 5 A 6 D 7 C 8 C 9 B 10 B Structured questions 11 a i Maintenance of a constant internal environment within set limits i Concentration
7 Answers to end-of-chapter questions Multiple choice questions 1 B 2 B 3 A 4 B 5 A 6 D 7 C 8 C 9 B 10 B Structured questions 11 a i Maintenance of a constant internal environment within set limits i Concentration
GI TRACT ORGANS ACCESSORY ORGANS
 Digestive System GI TRACT ORGANS Oral cavity Oropharynx Esophagus Stomach Small intestine Large Intestine Anus ACCESSORY ORGANS Salivary glands Pancreas Liver Gall bladder GI TRACT LAYERS Mucosa Submucosa
Digestive System GI TRACT ORGANS Oral cavity Oropharynx Esophagus Stomach Small intestine Large Intestine Anus ACCESSORY ORGANS Salivary glands Pancreas Liver Gall bladder GI TRACT LAYERS Mucosa Submucosa
Adrenergic agonists:-
 Adrenergic agonists:- A.) Catecholamines They are called catecholamines because they contain a catechol group, they are water-soluble and are 50% bound to plasma proteins, so they circulate in the bloodstream.
Adrenergic agonists:- A.) Catecholamines They are called catecholamines because they contain a catechol group, they are water-soluble and are 50% bound to plasma proteins, so they circulate in the bloodstream.
OVERVIEW OF LIPID METABOLISM
 VERVIEW F LIPID METABLISM Date: September 20, 2005 * Time: 8:00 am 8:50 am * Room: G202 Biomolecular Building Lecturer: Steve Chaney 515A Mary Ellen Jones Building [email protected] 9663286 *Please
VERVIEW F LIPID METABLISM Date: September 20, 2005 * Time: 8:00 am 8:50 am * Room: G202 Biomolecular Building Lecturer: Steve Chaney 515A Mary Ellen Jones Building [email protected] 9663286 *Please
Non-clinical development of biologics
 Aurigon Life Science GmbH Non-clinical development of biologics Requirements, challenges and case studies Committed to Life. Sigrid Messemer vet. med. M4 Seminar March 10 th 2014 Aurigon - your full service
Aurigon Life Science GmbH Non-clinical development of biologics Requirements, challenges and case studies Committed to Life. Sigrid Messemer vet. med. M4 Seminar March 10 th 2014 Aurigon - your full service
Disability Evaluation Under Social Security
 Disability Evaluation Under Social Security Revised Medical Criteria for Evaluating Endocrine Disorders Effective June 7, 2011 Why a Revision? Social Security revisions reflect: SSA s adjudicative experience.
Disability Evaluation Under Social Security Revised Medical Criteria for Evaluating Endocrine Disorders Effective June 7, 2011 Why a Revision? Social Security revisions reflect: SSA s adjudicative experience.
Lacidipine 2 mg Film-Coated Tablets PL 08553/0502. Lacidipine 4 mg Film-Coated Tablets PL 08553/0503. UK Public Assessment Report
 Lacidipine 2 mg Film-Coated Tablets PL 08553/0502 Lacidipine 4 mg Film-Coated Tablets PL 08553/0503 UK Public Assessment Report TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 4 Steps taken
Lacidipine 2 mg Film-Coated Tablets PL 08553/0502 Lacidipine 4 mg Film-Coated Tablets PL 08553/0503 UK Public Assessment Report TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 4 Steps taken
Pharmacy Policy Bulletin
 Pharmacy Policy Bulletin Title: Policy #: Pulmonary Arterial Hypertensive (PAH) agents Rx.01.83 Application of pharmacy policy is determined by benefits and contracts. Benefits may vary based on product
Pharmacy Policy Bulletin Title: Policy #: Pulmonary Arterial Hypertensive (PAH) agents Rx.01.83 Application of pharmacy policy is determined by benefits and contracts. Benefits may vary based on product
Diabetes and Drug Development
 Diabetes and Drug Development Metabolic Disfunction Leads to Multiple Diseases Hypertension ( blood pressure) Metabolic Syndrome (Syndrome X) LDL HDL Lipoproteins Triglycerides FFA Hyperinsulinemia Insulin
Diabetes and Drug Development Metabolic Disfunction Leads to Multiple Diseases Hypertension ( blood pressure) Metabolic Syndrome (Syndrome X) LDL HDL Lipoproteins Triglycerides FFA Hyperinsulinemia Insulin
Expectations for Data to Support Clinical Trial Drugs
 Expectations for Data to Support Clinical Trial Drugs Presentation to: APEC Advanced Workshop on Review of Drug Development in Clinical Trials Bangkok Thailand Feb 2-6 2009 Willem Stevens Ph.D., Chief
Expectations for Data to Support Clinical Trial Drugs Presentation to: APEC Advanced Workshop on Review of Drug Development in Clinical Trials Bangkok Thailand Feb 2-6 2009 Willem Stevens Ph.D., Chief
Your Life Your Health Cariodmetabolic Risk Syndrome Part VII Inflammation chronic, low-grade By James L. Holly, MD The Examiner January 25, 2007
 Your Life Your Health Cariodmetabolic Risk Syndrome Part VII Inflammation chronic, low-grade By James L. Holly, MD The Examiner January 25, 2007 The cardiometabolic risk syndrome is increasingly recognized
Your Life Your Health Cariodmetabolic Risk Syndrome Part VII Inflammation chronic, low-grade By James L. Holly, MD The Examiner January 25, 2007 The cardiometabolic risk syndrome is increasingly recognized
Chapter 5. Endocrine Regulation of Glucose Metabolism
 Chapter 5 Endocrine Regulation of Glucose Metabolism Overview of Glucose Homeostasis Glucose metabolism is critical to normal physiological functioning. Glucose acts both as a source of energy and as a
Chapter 5 Endocrine Regulation of Glucose Metabolism Overview of Glucose Homeostasis Glucose metabolism is critical to normal physiological functioning. Glucose acts both as a source of energy and as a
Absorption of Drugs. Transport of a drug from the GI tract
 Absorption of Drugs Absorption is the transfer of a drug from its site of administration to the bloodstream. The rate and efficiency of absorption depend on the route of administration. For IV delivery,
Absorption of Drugs Absorption is the transfer of a drug from its site of administration to the bloodstream. The rate and efficiency of absorption depend on the route of administration. For IV delivery,
Guidance for Industry Safety Testing of Drug Metabolites
 Guidance for Industry Safety Testing of Drug Metabolites U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2008 Pharmacology
Guidance for Industry Safety Testing of Drug Metabolites U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2008 Pharmacology
