Health Informatics Standardization: Relevance and Indian Initiatives
|
|
|
- Nancy Greene
- 10 years ago
- Views:
Transcription
1 Health Informatics Standardization: Relevance and Indian Initiatives Baljit Singh Bedi Advisor, CDAC (Sc. Society of MCIT, Govt. of India ) Past President, Telemedicine Society of India(TSI) & Member, National EMR Standardization Committee, MoH&FW & Chair, Sub-Group, Interoperability; Chair, FICCI EHR Subcommittee & Ex. Sr. Director & Head, Telemedicine Div., Dept. of IT, MCIT, Govt. of India) Regional Workshop on Establishment of NMCN. MoH&FW, February,2014, Lucknow
2 AGENDA Background - Role of ICT in Healthcare Indian Initiatives in proposing Standards Case for EHR Standardization - Benefits/Relevance of EMR/EHR Ministry of Health & Family Welfare Expert Committee for Standards Some Policy Directions
3 Background-Understanding Information and Communication Technologies (ICTs) Healthcare area is highly Data dependent. Power of ICTs lies in its capacity to effectively Access data Store data Analyze data Transmit data Predictive Analytics Utilizing this capability of ICTs has the potential to significantly contribute in preventive care, improving delivery, disease control, medical education and training, health management and health research
4 Standardisation- Indian Initiative Initiative in suggesting a Framework for Information Technology Infrastructure for Health(ITIH) in India (First effort for suggesting a Standard EHR for India) National efforts in proposing Recommendations on Guidelines, Standards & Practices For Telemedicine In India National Knowledge Commission s India Health Information Network Development (I-HIND) EHR Standardization Initiative of MoH&FW
5 GOVERNMENT PLAYS A LEADERSHIP ROLE IN CREATING HEALTH INFORMATION STANDARDISATION ENVIRONMENT As a PROVIDER, the public healthcare system is the largest integrated delivery network in India Health Information Exchange Standards As a REGULATOR, Govt. will have to frame and enact the required legal and education frameworks As a PAYER, Govt. will have to follow the standard protocols with healthcare providers to honor its clients insurance claims As a CONSUMER, Govt. will require standardised health information from various healthcare providers for policy decisions
6 Defining the standards- Key Objectives A clear vision for the objective of defining standards To Increase accessibility to quality medical care to all To promote growth of e-health and HMIS To identify and support mechanisms for protecting privacy & confidentiality of health data and security and legal issues. To broaden international cooperation in the scientific, legal and ethical aspects of the use of e-health To provide a framework for interoperability and scalability within and outside world To bring profitability to stakeholders
7 Framework in defining the guidelines and standards Interoperability Compatibility Scalability Portability The guiding framework need ensure: Inclusion of all stakeholders Making recommendations vendor neutral Standards also needs to look at clinical protocols and guidelines. Clinical protocols for telemedicine practice include scheduling procedures, consult procedures and equipment operation procedures
8 The Need for Standards EMRs and almost any other information-oriented system in a clinical environment cannot be used without well-defined standards for representing and communicating information Data need to be exchanged between multiple, heterogeneous systems and might be used by very different applications Standards are needed for several different uses: Identifying patients, providers, health-care plans, employers Transferring patient data across different systems
9 Categories of Standards required for health information Identifiers Codes & Terminology Content & Formats Messaging Security & Access Control Patient Id Provider Id Payer Id Health Plan Id Pharmacy Id Disease Codes Procedure Codes Observation Codes Drug Codes Surgical Consumables Patient Enrollment Registration Patient Medical Records Billing Formats Minimum Data Sets HL7, EDI, EDIFACT Authentication Access Control Non Repudiation Privacy Protection Lab Formats
10 EHR (Electronic Health Record) The ISO definition of Integrated Care EHR (Electronic Health Record) is a repository of information regarding the health of a subject of care in computer processable form, stored and transmitted securely, and accessible by multiple authorised users. (interoperability, security, integrated). It has a commonly agreed logical information model which is independent of EHR systems. (interoperability). Its primary purpose is the support of continuing, efficient and quality integrated health care and it contains information which is retrospective, concurrent and prospective (time span)
11 EHR Characteristics A life-long medical record in digital format Clear, well-organized, legible documentation Allows data access in real-time from anywhere, anytime Shared amongst all stakeholders so, able to seamlessly exchange data with related solutions Grows in size from birth till death Requires addressing issues ranging from patient privacy and security to interoperability, and errorfree transmission of information
12 Role of electronic health records (EHRs) in supporting basic medical research and clinical trials Collection of research data for clinical trials can become a by-product of the routine care of the patients Research data may be analyzed directly from the clinical data repository Representing medical knowledge that can be reused through the use of clinical data analytics, Evidence Based Medicine, etc. for improving patient care
13 National Expert Committee for Recommending Standard EMR/EHR set up by Ministry of Health & Family Welfare Expert Committee
14 EMR Standards Committee Ministry of Health & Family Welfare (MoH&FW), Govt. of India constituted a Committee of Experts for Development of Standards on EMR in Sept 2010 under Chairmanship of Additional Secretary &DG,CGHS, MoH&FW Objective of above Committee to recommend a set of EMR Standards for India to be followed by both public & private healthcare provider Procedure for continuous upgradation
15 Activities Undertaken EMR Standardisation Committee set up following Sub-Groups in October 2010: Sub-Group Task I : Standards- Terminology, coding standards Sub-Group Task II : Data connectivity- including H/W,S/W & Interoperability Sub-Group Task III: Data ownership-data protection& including security and legal aspects The current EHR standards are a result of the deliberations of the EMR Standards Committee & Subgroups and consolidation by the Sub-committee under FICCI of the public comments on the draft report put up by MoHFW, GoI in their website in May 2013 and deliberations thereof, finalized in August, Notified in September, 2013 on MoH&FW Website
16 Review of Key Technical Standards For any developing country embarking standardizations for e-health and Hospital Management Information systems (HMIS) need study : Existing International status Organizations working in field and current adoption of standards Standard Development Organizations (SDOs) and Special Interest Groups (SIGs) active in standardization process
17 Healthcare Informatics Organisations / Standards considered Organization Standards National Recommendations for Health Information Infrastructure in India International Organization for Standardization (ISO) European Committee for Standardization (CEN) Code of Federal Regulations (CFR) Information Technology Infrastructure for Health (ITIH) framework Recommendations on Guidelines, Standards & Practices for Telemedicine in India Indian health information network development (ihind) recommendations from the National Knowledge Requirements for Electronic Health Record Architecture (ISO / TS 18308) CEN / TC 251 EN Health Information Technology Standards, Implementation Specifications, and Certification Criteria and Certification Programs for Health Information Technology (Title 45, Part 170)
18 Healthcare Informatics Organisations/ Standards.. Contd. Organization Standards American Society for Testing & Materials (ASTM) Health Level 7 (HL7) HL7 & ASTM Collaboration National Electrical Manufacturer s Association (NEMA) Continuity of Care Record (CCR) HL7 v2.x HL7 v3 HL7 Clinical Document Architecture (CDA) EHR - System Functional Model Continuity of Care Document (CCD) Digital Imaging and Communications in Medicine (DICOM PS onwards) Office of National Coordinator for Health Information Technology (ONCHIT) United States EHR Meaningful Use
19 Approved Standards: Codes Diseases (Diagnosis) WHO s ICD 10 Procedures WHO s PCS Disability WHO s ICF Clinical Terminology (for clinical observations) IHTSDO s SNOMED CT Laboratory Observations Regenstrief Inst s LOINC
20 Messaging, Imaging, Clinical Document Format Messaging HL7 V3.0 RIM (Reference Information Model) HL7 V2.5 (for backward compatibility) Imaging NEMA s Digital Imaging & Communication in Medicine (DICOM) PS Later revisions can be included as evolved Clinical Data Format HL7 CDA 2.0 (Clinical Document Architecture) ASTM CCR (Continuity of Care Record)
21 Minimum Data Set (MDS) Minimum amount of health information required about a patient to profile a disease in a standard format. Ensure that the health information is precise, unambiguous and acceptable to all stakeholders. Represented in such a manner that they can be easily analysed and conclusions drawn from the data.
22 Minimum Data Set Patient Demographics Observer Details Reasons for visit Allergies & Immunization Clinical Observations Investigation Orders Laboratory Radiology Investigation Results Treatment Plan Medication Active Discontinued Restarted Procedures Diet Life-style Outcome Date- time Stamp
23 Preservation & Security Purpose To protect the confidentiality, integrity, and availability of information Different From Other Industries Regulated (US: HIPAA and other regulations) Community interest Legal issues Threat Sources Accidental Acts Incidental disclosures, Errors and omissions, Proximity to risk areas, Equipment malfunction Deliberate Acts Misuse/abuse of privileges, Fraud, Theft, Extortion, Crime Environmental threats Fire, Flood, Weather, Power EHR Security Mechanisms Authentication Role-Based Access Control Data Verification Transport Level Security Encryption Mechanisms Data/Storage Audit/Log Anonymization Ethical and Legal Considerations Data Retention Policy Patient Policy/Confidentiality Patient Consent Quality of Service (QOS) Data Ownership Non-repudiation Dispute Resolution
24 Some Policy Directions: Report of the Steering Committee on Health for 12 th Plan (Incorporating Reports of the Working Groups and deliberations of the Steering Committee Meetings) Health Division Planning Commission Feb. 2012
25 Major emphasis on use of Telemedicine from PHC level and use of EMR A composite HIS should incorporate the following: Out-patient and in-patient information through Electronic Medical Records (EMR).This will help provide the best care based on Standard Treatment Guidelines, reduce response time in emergencies, support the organ retrieval and transplantation programme and improve general hospital administration. It would also help estimate burden of disease and facilitate policy decisions at State and national levels.
26 Major emphasis on use of Telemedicine from PHC level and use of EMR A composite HIS should incorporate the following: Out-patient and in-patient information through Electronic Medical Records (EMR).This will help provide the best care based on Standard Treatment Guidelines, reduce response time in emergencies, support the organ retrieval and transplantation programme and improve general hospital administration. It would also help estimate burden of disease and facilitate policy decisions at State and national levels.
27 MINISTRY OF HEALTH & FAMILY WELFARE (Department of Health & Family Welfare) NOTIFICATION regarding Clinical Establishments (Registration & Regulations) Act 2010 (23 of 2010) New Delhi, the 23 rd May 2012
28 G.S.R.387 (E).-In the exercise of powers conferred by Section 52 of the Clinical Establishments (Registration & Regulations) Act 2010 (23 of 2010),the Central Government makes the following Rules, namely:- 9. Other conditions for registration and continuation of clinical establishments.- For registration and continuation every clinical establishment shall fulfill the following conditions, namely.-. (iv) Clinical establishments shall provide and maintain Electronic Medical Record or Electronic Health Records of every patients as may be determined and issued by the Central Government or State Government as the case may be from time to time.
29 Thank You B.S. Bedi:
Dr. SB Bhattacharyya MBBS, MBA Member, EHR Standards Committee, MoH&FW, Govt. of India Honorary State Secretary (2015), IMA Haryana President (2010
 Dr. SB Bhattacharyya MBBS, MBA Member, EHR Standards Committee, MoH&FW, Govt. of India Honorary State Secretary (2015), IMA Haryana President (2010 2011), IAMI Standards are a basis for comparison and
Dr. SB Bhattacharyya MBBS, MBA Member, EHR Standards Committee, MoH&FW, Govt. of India Honorary State Secretary (2015), IMA Haryana President (2010 2011), IAMI Standards are a basis for comparison and
Health Care Information System Standards
 Health Care Information System Standards 1 Standards Development Process Four Methods (Hammond & Cimino, 2001) Ad hoc no formal adoption process De facto vendor or other has a very large segment of the
Health Care Information System Standards 1 Standards Development Process Four Methods (Hammond & Cimino, 2001) Ad hoc no formal adoption process De facto vendor or other has a very large segment of the
Electronic Health Record. Standards, Coding Systems, Frameworks, and Infrastructures
 Brochure More information from http://www.researchandmarkets.com/reports/2178436/ Electronic Health Record. Standards, Coding Systems, Frameworks, and Infrastructures Description: Discover How Electronic
Brochure More information from http://www.researchandmarkets.com/reports/2178436/ Electronic Health Record. Standards, Coding Systems, Frameworks, and Infrastructures Description: Discover How Electronic
EHR Standards Landscape
 EHR Standards Landscape Dr Dipak Kalra Centre for Health Informatics and Multiprofessional Education (CHIME) University College London [email protected] A trans-national ehealth Infostructure Wellness
EHR Standards Landscape Dr Dipak Kalra Centre for Health Informatics and Multiprofessional Education (CHIME) University College London [email protected] A trans-national ehealth Infostructure Wellness
Health Informatics in India : Vision and Activities
 Health Informatics in India : Vision and Activities September 5, 2008 S. Ramakrishnan [email protected] 1 Health Informatics : Multidisciplinary in Nature Medicine Biology Computer Science Health Informatics
Health Informatics in India : Vision and Activities September 5, 2008 S. Ramakrishnan [email protected] 1 Health Informatics : Multidisciplinary in Nature Medicine Biology Computer Science Health Informatics
EHR Systems: an Introduction
 ehealth Week 2007 EuroRec Institute / ProRec Germany Workshop EHR Systems: an Introduction Bernd Blobel 1 & Dipak Kalra 2 1 ehealth Competence Center University of Regensburg Medical Center Regensburg,
ehealth Week 2007 EuroRec Institute / ProRec Germany Workshop EHR Systems: an Introduction Bernd Blobel 1 & Dipak Kalra 2 1 ehealth Competence Center University of Regensburg Medical Center Regensburg,
The Big Picture: IDNT in Electronic Records Glossary
 TERM DEFINITION CCI Canada Health Infoway Canadian Institute for Health Information EHR EMR EPR H L 7 (HL7) Canadian Classification of Interventions is the Canadian standard for classifying health care
TERM DEFINITION CCI Canada Health Infoway Canadian Institute for Health Information EHR EMR EPR H L 7 (HL7) Canadian Classification of Interventions is the Canadian standard for classifying health care
New York ehealth Collaborative. Health Information Exchange and Interoperability April 2012
 New York ehealth Collaborative Health Information Exchange and Interoperability April 2012 1 Introductions Information exchange patient, information, care team How is Health information exchanged Value
New York ehealth Collaborative Health Information Exchange and Interoperability April 2012 1 Introductions Information exchange patient, information, care team How is Health information exchanged Value
Standards and their role in Healthcare ICT Strategy. 10th Annual Public Sector IT Conference
 Standards and their role in Healthcare ICT Strategy 10th Annual Public Sector IT Conference Peter Connolly Oct 2014 What is the Direction of Travel? 1 Understanding the Why- The Data Context 2 Stakeholder
Standards and their role in Healthcare ICT Strategy 10th Annual Public Sector IT Conference Peter Connolly Oct 2014 What is the Direction of Travel? 1 Understanding the Why- The Data Context 2 Stakeholder
EHR STRATEGY FINLAND. Kari Harno Helsinki University Central Hospital
 EHR STRATEGY FINLAND Kari Harno Helsinki University Central Hospital The Nordic Welfare Model In Finland this model includes: universal coverage of services universal social security scheme health insurance
EHR STRATEGY FINLAND Kari Harno Helsinki University Central Hospital The Nordic Welfare Model In Finland this model includes: universal coverage of services universal social security scheme health insurance
International HL7 Interoperability Conference - IHIC 2010
 International HL7 Interoperability Conference - IHIC 2010 National ehealth Initiatives: Global Health Information Technology Standards Serving Local Needs Building Interoperability across many localities
International HL7 Interoperability Conference - IHIC 2010 National ehealth Initiatives: Global Health Information Technology Standards Serving Local Needs Building Interoperability across many localities
Certification Guidance for EHR Technology Developers Serving Health Care Providers Ineligible for Medicare and Medicaid EHR Incentive Payments
 I. Background Certification Guidance for EHR Technology Developers Serving Health Care Providers Ineligible for Medicare and Medicaid EHR Incentive Payments The Medicare and Medicaid EHR Incentive Programs,
I. Background Certification Guidance for EHR Technology Developers Serving Health Care Providers Ineligible for Medicare and Medicaid EHR Incentive Payments The Medicare and Medicaid EHR Incentive Programs,
Global Health Informatics Standards for Patient Safety
 Global Health Informatics Standards for Patient Safety Yun Sik Kwak, MD, PhD Dept Med Info.,Kyungpook Nat l Univ Sch Med Daegu, Korea HL7 Korea ISO/TC 215 APAMI ehscg October 28, 2006 APAMI MIC2006 - Taipei
Global Health Informatics Standards for Patient Safety Yun Sik Kwak, MD, PhD Dept Med Info.,Kyungpook Nat l Univ Sch Med Daegu, Korea HL7 Korea ISO/TC 215 APAMI ehscg October 28, 2006 APAMI MIC2006 - Taipei
ehr Sharable Data Vicky Fung Senior Health Informatician ehr Information Standards Office
 ehr Sharable Data Vicky Fung Senior Health Informatician ehr Information Standards Office ehr - Vision DH HA epr Private Private Hospit als Hospitals EHR Repository Access Portal CMS onramp PPP Clinics
ehr Sharable Data Vicky Fung Senior Health Informatician ehr Information Standards Office ehr - Vision DH HA epr Private Private Hospit als Hospitals EHR Repository Access Portal CMS onramp PPP Clinics
Standardization of the Australian Medical Data Exchange Model. Michael Legg PhD
 Standardization of the Australian Medical Data Exchange Model Michael Legg PhD Agenda The National ehealth Program Pathology in Australia Standardisation in Australia Some projects Communication Any meaningful
Standardization of the Australian Medical Data Exchange Model Michael Legg PhD Agenda The National ehealth Program Pathology in Australia Standardisation in Australia Some projects Communication Any meaningful
Health Informatics Development in the Hospital Authority
 Health Informatics Development in the Hospital Authority Dr CP Wong Chairman, Clinical Informatics Program Executive Group Co-Chairman, Clinical Informatics Program Steering Group Begin to take off in
Health Informatics Development in the Hospital Authority Dr CP Wong Chairman, Clinical Informatics Program Executive Group Co-Chairman, Clinical Informatics Program Steering Group Begin to take off in
Overview of ehr Development. Slide - 1
 Overview of ehr Development Slide - 1 Where are we today? Hospital Authority 8 million patient records 800 million laboratory results 340 million prescribed drugs 34 million Xray images 33 million transactions
Overview of ehr Development Slide - 1 Where are we today? Hospital Authority 8 million patient records 800 million laboratory results 340 million prescribed drugs 34 million Xray images 33 million transactions
Electronic Health Record (EHR) Standards Survey
 Electronic Health Record (EHR) Standards Survey Compiled by: Simona Cohen, Amnon Shabo Date: August 1st, 2001 This report is a short survey about the main emerging standards that relate to EHR - Electronic
Electronic Health Record (EHR) Standards Survey Compiled by: Simona Cohen, Amnon Shabo Date: August 1st, 2001 This report is a short survey about the main emerging standards that relate to EHR - Electronic
The Continuity of Care Document. Changing the Landscape of Healthcare Information Exchange
 The Continuity of Care Document Changing the Landscape of Healthcare Information Exchange 1 Electronic Clinical Document Exchange Prior to the approval of the Continuity of Care Document (CCD) as an ANSI
The Continuity of Care Document Changing the Landscape of Healthcare Information Exchange 1 Electronic Clinical Document Exchange Prior to the approval of the Continuity of Care Document (CCD) as an ANSI
Overview of the national laws on electronic health records in the EU Member States National Report for Lithuania
 Overview of the national laws on electronic health records in the EU Member States and their interaction with the provision of cross-border ehealth services Contract 2013 63 02 Overview of the national
Overview of the national laws on electronic health records in the EU Member States and their interaction with the provision of cross-border ehealth services Contract 2013 63 02 Overview of the national
Presentation in Steering Group meeting Planning Commission, Govt of India. Public Health Informatics Group Discussion Inputs to Steering Committee
 P i i Presentation in Steering Group meeting Planning Commission, Govt of India Public Health Informatics Group Discussion Inputs to Steering Committee References to Informatics Key Inputs to the 12 th
P i i Presentation in Steering Group meeting Planning Commission, Govt of India Public Health Informatics Group Discussion Inputs to Steering Committee References to Informatics Key Inputs to the 12 th
HEALTH INFORMATION TECHNOLOGY*
 GLOSSARY of COMMON TERMS and ACRONYMS In HEALTH INFORMATION TECHNOLOGY* (April 2011) AHIC American Health Information Community The AHIC was a federal advisory panel created by HHS to make recommendations
GLOSSARY of COMMON TERMS and ACRONYMS In HEALTH INFORMATION TECHNOLOGY* (April 2011) AHIC American Health Information Community The AHIC was a federal advisory panel created by HHS to make recommendations
HL7 and Meaningful Use
 HL7 and Meaningful Use Grant M. Wood HL7 Ambassador HIMSS14 2012 Health Level Seven International. All Rights Reserved. HL7 and Health Level Seven are registered trademarks of Health Level Seven International.
HL7 and Meaningful Use Grant M. Wood HL7 Ambassador HIMSS14 2012 Health Level Seven International. All Rights Reserved. HL7 and Health Level Seven are registered trademarks of Health Level Seven International.
TEST INSTRUCTIONS FOR CROSS VENDOR EXCHANGE TABLE OF CONTENTS
 TEST INSTRUCTIONS FOR CROSS VENDOR EXCHANGE TABLE OF CONTENTS Introduction...2 Meaningful Use Objectives and Measures... 3 Eligible Professionals 495.6(j)(14)... 3 Eligible Hospitals and Critical Access
TEST INSTRUCTIONS FOR CROSS VENDOR EXCHANGE TABLE OF CONTENTS Introduction...2 Meaningful Use Objectives and Measures... 3 Eligible Professionals 495.6(j)(14)... 3 Eligible Hospitals and Critical Access
Health Smart Cards and NHIP A Taiwan Perspective
 Health Smart Cards and NHIP A Taiwan Perspective Yu-Chuan (Jack) Li, M.D., Ph.D. Vice President, Asia Pacific Assoc. for Medical Informatics (APAMI) President Emeritus, Taiwan Assoc. for Medical Informatics
Health Smart Cards and NHIP A Taiwan Perspective Yu-Chuan (Jack) Li, M.D., Ph.D. Vice President, Asia Pacific Assoc. for Medical Informatics (APAMI) President Emeritus, Taiwan Assoc. for Medical Informatics
Health Information and Quality Authority. To drive continuous improvements in the quality and safety of health and social care in Ireland
 Introduction Health Information and Quality Authority To drive continuous improvements in the quality and safety of health and social care in Ireland Background Health Act 2007, establishment 15 May 2007
Introduction Health Information and Quality Authority To drive continuous improvements in the quality and safety of health and social care in Ireland Background Health Act 2007, establishment 15 May 2007
Accelerating Clinical Trials Through Shared Access to Patient Records
 INTERSYSTEMS WHITE PAPER Accelerating Clinical Trials Through Shared Access to Patient Records Improved Access to Clinical Data Across Hospitals and Systems Helps Pharmaceutical Companies Reduce Delays
INTERSYSTEMS WHITE PAPER Accelerating Clinical Trials Through Shared Access to Patient Records Improved Access to Clinical Data Across Hospitals and Systems Helps Pharmaceutical Companies Reduce Delays
Summary of the Proposed Rule for the Medicare and Medicaid Electronic Health Records (EHR) Incentive Program (Eligible Professionals only)
 Summary of the Proposed Rule for the Medicare and Medicaid Electronic Health Records (EHR) Incentive Program (Eligible Professionals only) Background Enacted on February 17, 2009, the American Recovery
Summary of the Proposed Rule for the Medicare and Medicaid Electronic Health Records (EHR) Incentive Program (Eligible Professionals only) Background Enacted on February 17, 2009, the American Recovery
HL7 & Meaningful Use. Charles Jaffe, MD, PhD CEO Health Level Seven International. HIMSS 11 Orlando February 23, 2011
 HL7 & Meaningful Use Charles Jaffe, MD, PhD CEO Health Level Seven International HIMSS 11 Orlando February 23, 2011 Overview Overview of Meaningful Use HIT Standards and Meaningful Use Overview HL7 Standards
HL7 & Meaningful Use Charles Jaffe, MD, PhD CEO Health Level Seven International HIMSS 11 Orlando February 23, 2011 Overview Overview of Meaningful Use HIT Standards and Meaningful Use Overview HL7 Standards
Table of Contents. Preface... 1. 1 CPSA Position... 2. 1.1 How EMRs and Alberta Netcare are Changing Practice... 2. 2 Evolving Standards of Care...
 March 2015 Table of Contents Preface... 1 1 CPSA Position... 2 1.1 How EMRs and Alberta Netcare are Changing Practice... 2 2 Evolving Standards of Care... 4 2.1 The Medical Record... 4 2.2 Shared Medical
March 2015 Table of Contents Preface... 1 1 CPSA Position... 2 1.1 How EMRs and Alberta Netcare are Changing Practice... 2 2 Evolving Standards of Care... 4 2.1 The Medical Record... 4 2.2 Shared Medical
National Integrated Services Framework The Foundation for Future e-health Connectivity. Peter Connolly HSE May 2013
 National Integrated Framework The Foundation for Future e-health Connectivity Peter Connolly HSE May 2013 The Context Introduction A national approach to interoperability is essential for Ireland s E-Health
National Integrated Framework The Foundation for Future e-health Connectivity Peter Connolly HSE May 2013 The Context Introduction A national approach to interoperability is essential for Ireland s E-Health
Meaningful Use Stage 2 Certification: A Guide for EHR Product Managers
 Meaningful Use Stage 2 Certification: A Guide for EHR Product Managers Terminology Management is a foundational element to satisfying the Meaningful Use Stage 2 criteria and due to its complexity, and
Meaningful Use Stage 2 Certification: A Guide for EHR Product Managers Terminology Management is a foundational element to satisfying the Meaningful Use Stage 2 criteria and due to its complexity, and
Relationship of HL7 EHR System Draft Standard to X12N
 Relationship of HL7 EHR System Draft Standard to X12N EHR Technical Committee Co-Chairs: Gary Dickinson Linda Fischetti Sam Heard Excerpt of EHR-S DSTU Class Overview of Discussion Background Where We
Relationship of HL7 EHR System Draft Standard to X12N EHR Technical Committee Co-Chairs: Gary Dickinson Linda Fischetti Sam Heard Excerpt of EHR-S DSTU Class Overview of Discussion Background Where We
IHE Australia Workshops July 2011. Prepared by: Heather Grain Chair: Standards Australia IT14 Health Informatics and Ehealth Education
 IHE Australia Workshops July 2011 Prepared by: Heather Grain Chair: Standards Australia IT14 Health Informatics and Ehealth Education Standards are key to healthcare at all levels service, fiscal, administrative
IHE Australia Workshops July 2011 Prepared by: Heather Grain Chair: Standards Australia IT14 Health Informatics and Ehealth Education Standards are key to healthcare at all levels service, fiscal, administrative
THE E-HEALTH JOURNEY. Ministry of Health Jamaica. Optimizing the use of ICT Applications in Health and Patient Care
 THE E-HEALTH JOURNEY Ministry of Health Jamaica Optimizing the use of ICT Applications in Health and Patient Care 8 th Caribbean Conference on Health Financing Initiatives Presenter: Mr. Arnold Cooper
THE E-HEALTH JOURNEY Ministry of Health Jamaica Optimizing the use of ICT Applications in Health and Patient Care 8 th Caribbean Conference on Health Financing Initiatives Presenter: Mr. Arnold Cooper
Healthcare Data: Secondary Use through Interoperability
 Healthcare Data: Secondary Use through Interoperability Floyd Eisenberg MD MPH July 18, 2007 NCVHS Agenda Policies, Enablers, Restrictions Date Re-Use Landscape Sources of Data for Quality Measurement,
Healthcare Data: Secondary Use through Interoperability Floyd Eisenberg MD MPH July 18, 2007 NCVHS Agenda Policies, Enablers, Restrictions Date Re-Use Landscape Sources of Data for Quality Measurement,
Singapore s National Electronic Health Record
 Singapore s National Electronic Health Record The Roadmap to 2010 Dr Sarah Christine Muttitt Chief Information Officer Information Systems Division 17 th July, 2009 Taking the Next Step (MSM April 2008)
Singapore s National Electronic Health Record The Roadmap to 2010 Dr Sarah Christine Muttitt Chief Information Officer Information Systems Division 17 th July, 2009 Taking the Next Step (MSM April 2008)
Version: January 2008 ASTM E-31: EHR and Informatics Standards Education For Health Professional Disciplines. Background
 Version: January 2008 ASTM E-31: EHR and Informatics Standards Education For Health Professional Disciplines Background Work on standards for the EHR in the context of all such standards for the Health
Version: January 2008 ASTM E-31: EHR and Informatics Standards Education For Health Professional Disciplines Background Work on standards for the EHR in the context of all such standards for the Health
HIE Services & Pricing
 Services Available at No Cost Health Information Exchange Services & Pricing Package Effective: December 11, 2015 0 Interface Connection Details Services Available at No Cost HealthlinkNY Web Portal The
Services Available at No Cost Health Information Exchange Services & Pricing Package Effective: December 11, 2015 0 Interface Connection Details Services Available at No Cost HealthlinkNY Web Portal The
ehealth, HIS, etc ehealth All information about health HMIS mhealth HIS Statistical IS Credited: Karl Brown, Rockefeller Foundation
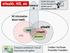 ehealth, HIS, etc Statistical IS Health Informatics: Formal discipline that studies use of information in health All information about health HIS HMIS ehealth mhealth Tele-medicine Enterprise architecture:
ehealth, HIS, etc Statistical IS Health Informatics: Formal discipline that studies use of information in health All information about health HIS HMIS ehealth mhealth Tele-medicine Enterprise architecture:
HIE Services & Pricing
 Services Available at No Cost Health Information Exchange Services & Pricing Package Services Available at No Cost Services Available at No Cost HealthlinkNY Web Portal The HealthlinkNY Web Portal is available
Services Available at No Cost Health Information Exchange Services & Pricing Package Services Available at No Cost Services Available at No Cost HealthlinkNY Web Portal The HealthlinkNY Web Portal is available
Health Information Exchange. Scalable and Affordable
 Integration is Everything Health Information Exchange Scalable and Affordable Today s healthcare organizations are transforming the quality of patient care by electronically exchanging patient data at
Integration is Everything Health Information Exchange Scalable and Affordable Today s healthcare organizations are transforming the quality of patient care by electronically exchanging patient data at
Medical Information Systems
 Medical Information Systems Introduction The introduction of information systems in hospitals and other medical facilities is not only driven by the wish to improve management of patient-related data for
Medical Information Systems Introduction The introduction of information systems in hospitals and other medical facilities is not only driven by the wish to improve management of patient-related data for
Recommendations On Electronic Medical Records Standards In India
 Report of Sub-Group Task II (data connectivity) with reports of Sub-Group Task I (standards) and Sub-Group Task III (data ownership) incorporated Recommendations On Electronic Medical Records Standards
Report of Sub-Group Task II (data connectivity) with reports of Sub-Group Task I (standards) and Sub-Group Task III (data ownership) incorporated Recommendations On Electronic Medical Records Standards
MED 2400 MEDICAL INFORMATICS FUNDAMENTALS
 MED 2400 MEDICAL INFORMATICS FUNDAMENTALS NEW YORK CITY COLLEGE OF TECHNOLOGY The City University Of New York School of Arts and Sciences Biological Sciences Department Course title: Course code: MED 2400
MED 2400 MEDICAL INFORMATICS FUNDAMENTALS NEW YORK CITY COLLEGE OF TECHNOLOGY The City University Of New York School of Arts and Sciences Biological Sciences Department Course title: Course code: MED 2400
HL7 & HL7 CDA: The Implementation of Thailand s Healthcare Messaging Exchange Standards Nawanan Theera-Ampornpunt, M.D., Ph.D.
 HL7 & HL7 CDA: The Implementation of Thailand s Healthcare Messaging Exchange Standards Nawanan Theera-Ampornpunt, M.D., Ph.D. Deputy Executive Director for Informatics, Chakri Naruebodindra Medical Institute,
HL7 & HL7 CDA: The Implementation of Thailand s Healthcare Messaging Exchange Standards Nawanan Theera-Ampornpunt, M.D., Ph.D. Deputy Executive Director for Informatics, Chakri Naruebodindra Medical Institute,
Data Integrity in an Era of EHRs, HIEs, and HIPAA: A Health Information Management Perspective
 Office for Civil Rights, HHS National Institute for Standards and Technology Conference Safeguarding Health Information: Building Assurance through HIPAA Security Data Integrity in an Era of EHRs, HIEs,
Office for Civil Rights, HHS National Institute for Standards and Technology Conference Safeguarding Health Information: Building Assurance through HIPAA Security Data Integrity in an Era of EHRs, HIEs,
GLOBAL ICT TRENDS IN HEALTH PRESENTED BY: GERRIT HENNING CEO: AMETHST (PTY) LTD
 This document is protected by copyright. No part of this document may be reproduced manual or otherwise, in whole or in part, without the prior, written consent of Health System Technologies (Pty) Ltd.
This document is protected by copyright. No part of this document may be reproduced manual or otherwise, in whole or in part, without the prior, written consent of Health System Technologies (Pty) Ltd.
Table of Contents. Page 1
 Table of Contents Executive Summary... 2 1 CPSA Interests and Roles in ehealth... 4 1.1 CPSA Endorsement of ehealth... 4 1.2 CPSA Vision for ehealth... 5 1.3 Dependencies... 5 2 ehealth Policies and Trends...
Table of Contents Executive Summary... 2 1 CPSA Interests and Roles in ehealth... 4 1.1 CPSA Endorsement of ehealth... 4 1.2 CPSA Vision for ehealth... 5 1.3 Dependencies... 5 2 ehealth Policies and Trends...
HIE Ready 2.0 SPECIFICATIONS MATRIX. Product Name: Version Number: Preferred Message and Trigger
 HIE Ready 2.0 SPECIFICATIONS MATRIX Entity Name: Street Address: City, State, Zip: Point of Contact Name: E-mail & Phone: Alternate Contact Name: Alternate E-mail & Phone: Product Name: Version Number:
HIE Ready 2.0 SPECIFICATIONS MATRIX Entity Name: Street Address: City, State, Zip: Point of Contact Name: E-mail & Phone: Alternate Contact Name: Alternate E-mail & Phone: Product Name: Version Number:
Meaningful use. Meaningful data. Meaningful care. The 3M Healthcare Data Dictionary: Enabling effective health information exchange
 Meaningful use. Meaningful data. Meaningful care. The 3M Healthcare Data Dictionary: Enabling effective health information exchange Understanding health information exchanges (HIEs) What is the goal of
Meaningful use. Meaningful data. Meaningful care. The 3M Healthcare Data Dictionary: Enabling effective health information exchange Understanding health information exchanges (HIEs) What is the goal of
Demonstrating Meaningful Use of EHRs: The top 10 compliance challenges for Stage 1 and what s new with 2
 Demonstrating Meaningful Use of EHRs: The top 10 compliance challenges for Stage 1 and what s new with 2 Today s discussion A three-stage approach to achieving Meaningful Use Top 10 compliance challenges
Demonstrating Meaningful Use of EHRs: The top 10 compliance challenges for Stage 1 and what s new with 2 Today s discussion A three-stage approach to achieving Meaningful Use Top 10 compliance challenges
Creating a national electronic health record: The Canada Health Infoway experience
 Creating a national electronic health record: The Canada Health Infoway experience Presentation by Dennis Giokas Chief Technology Officer, Canada Health Infoway October 11, 2007 Overview The need for EHR
Creating a national electronic health record: The Canada Health Infoway experience Presentation by Dennis Giokas Chief Technology Officer, Canada Health Infoway October 11, 2007 Overview The need for EHR
Health Information Exchange in Minnesota & North Dakota
 Health Information Exchange in Minnesota & North Dakota April 16, 2014 Objectives Learn basic HIE concepts Understand key success factors for HIE Gain an understanding of Minnesota and North Dakota s approach
Health Information Exchange in Minnesota & North Dakota April 16, 2014 Objectives Learn basic HIE concepts Understand key success factors for HIE Gain an understanding of Minnesota and North Dakota s approach
I n t e r S y S t e m S W h I t e P a P e r F O R H E A L T H C A R E IT E X E C U T I V E S. In accountable care
 I n t e r S y S t e m S W h I t e P a P e r F O R H E A L T H C A R E IT E X E C U T I V E S The Role of healthcare InfoRmaTIcs In accountable care I n t e r S y S t e m S W h I t e P a P e r F OR H E
I n t e r S y S t e m S W h I t e P a P e r F O R H E A L T H C A R E IT E X E C U T I V E S The Role of healthcare InfoRmaTIcs In accountable care I n t e r S y S t e m S W h I t e P a P e r F OR H E
Meaningful Use Stage 2 as it Relates to the Lab Implementing the Public Health Agency Interface. Interface
 Meaningful Use Stage 2 as it Relates to the Lab Implementing the Public Health Agency Interface Interface Topics Covered Meaningful Use Stage 2 (MU2) Electronic Lab Results (ELR) interface Lab Results
Meaningful Use Stage 2 as it Relates to the Lab Implementing the Public Health Agency Interface Interface Topics Covered Meaningful Use Stage 2 (MU2) Electronic Lab Results (ELR) interface Lab Results
Carolyn P. Hartley, MLA President, CEO Physicians EHR, Inc
 Carolyn P. Hartley, MLA President, CEO Physicians EHR, Inc Health Information Management Professional SME to Regional Extension Centers President, CEO: Physicians EHR, Inc 140+ complex to risky EHR installations
Carolyn P. Hartley, MLA President, CEO Physicians EHR, Inc Health Information Management Professional SME to Regional Extension Centers President, CEO: Physicians EHR, Inc 140+ complex to risky EHR installations
Canada Health Infoway
 Canada Health Infoway EHR s in the Canadian Context June 7, 2005 Mike Sheridan, COO Canada Health Infoway Healthcare Renewal In Canada National Healthcare Priorities A 10-year Plan to Strengthen Healthcare
Canada Health Infoway EHR s in the Canadian Context June 7, 2005 Mike Sheridan, COO Canada Health Infoway Healthcare Renewal In Canada National Healthcare Priorities A 10-year Plan to Strengthen Healthcare
Custom Report Data Elements: 2012 IT Database Fields. Source: American Hospital Association IT Survey
 Custom Report Data Elements: 2012 IT Database Fields Source: American Hospital Association IT Survey COMPUTERIZED SYSTEM IMPLEMENTATION 3 Bar Coding 3 Computerized Provider Order Entry 3 Decision Support
Custom Report Data Elements: 2012 IT Database Fields Source: American Hospital Association IT Survey COMPUTERIZED SYSTEM IMPLEMENTATION 3 Bar Coding 3 Computerized Provider Order Entry 3 Decision Support
MEDICAL ASSISTANCE BULLETIN
 ISSUE DATE April 8, 2011 EFFECTIVE DATE April 8, 2011 MEDICAL ASSISTANCE BULLETIN NUMBER 03-11-01, 09-11-02, 14-11-01, 18-11-01 24-11-03, 27-11-02, 31-11-02, 33-11-02 SUBJECT Electronic Prescribing Internet-based
ISSUE DATE April 8, 2011 EFFECTIVE DATE April 8, 2011 MEDICAL ASSISTANCE BULLETIN NUMBER 03-11-01, 09-11-02, 14-11-01, 18-11-01 24-11-03, 27-11-02, 31-11-02, 33-11-02 SUBJECT Electronic Prescribing Internet-based
Trends in Healthcare Information Standardization
 TANJI Natsuki Abstract Standardization of medical information systems by industry associations such as ISO/TC 215 and CEN/TC 251 is currently underway internationally. In Japan, too, participation in and
TANJI Natsuki Abstract Standardization of medical information systems by industry associations such as ISO/TC 215 and CEN/TC 251 is currently underway internationally. In Japan, too, participation in and
Use of Electronic Health Record Data in Clinical Investigations
 Use of Electronic Health Record Data in Clinical Investigations Guidance for Industry DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding
Use of Electronic Health Record Data in Clinical Investigations Guidance for Industry DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding
Stage 2 Eligible Professional Meaningful Use Core Measures Measure 15 of 17 Last Updated: August, 2015
 Summary of Care Objective Measures Exclusion Table of Contents Definition of Terms Attestation Requirements Additional Information Certification and Standards Criteria Stage 2 Eligible Professional Meaningful
Summary of Care Objective Measures Exclusion Table of Contents Definition of Terms Attestation Requirements Additional Information Certification and Standards Criteria Stage 2 Eligible Professional Meaningful
HL7 and SOA Based Distributed Electronic Patient Record Architecture Using Open EMR
 HL7 and SOA Based Distributed Electronic Patient Record Architecture Using Open EMR Priti Kalode 1, Dr Onkar S Kemkar 2, Dr P R Gundalwar 3 Research Student, Dept of Comp Sci &Elec, RTM Nagpur University
HL7 and SOA Based Distributed Electronic Patient Record Architecture Using Open EMR Priti Kalode 1, Dr Onkar S Kemkar 2, Dr P R Gundalwar 3 Research Student, Dept of Comp Sci &Elec, RTM Nagpur University
A Semantic Foundation for Achieving HIE Interoperability
 A Semantic Foundation for Achieving HIE Interoperability Introduction Interoperability of health IT systems within and across organizational boundaries has long been the holy grail of healthcare technologists.
A Semantic Foundation for Achieving HIE Interoperability Introduction Interoperability of health IT systems within and across organizational boundaries has long been the holy grail of healthcare technologists.
Eligible Professionals please see the document: MEDITECH Prepares You for Stage 2 of Meaningful Use: Eligible Professionals.
 s Preparing for Meaningful Use in 2014 MEDITECH (Updated December 2013) Professionals please see the document: MEDITECH Prepares You for Stage 2 of Meaningful Use: Professionals. Congratulations to our
s Preparing for Meaningful Use in 2014 MEDITECH (Updated December 2013) Professionals please see the document: MEDITECH Prepares You for Stage 2 of Meaningful Use: Professionals. Congratulations to our
The Do s and Don ts of Medical Device integration
 Advances in Wireless Technologies for Healthcare The Do s and Don ts of Medical Device integration Shahid N. Shah, CEO Visit Dräger and Shahid at HIMSS 2012 Dräger Booth on the main floor: Booth #5734
Advances in Wireless Technologies for Healthcare The Do s and Don ts of Medical Device integration Shahid N. Shah, CEO Visit Dräger and Shahid at HIMSS 2012 Dräger Booth on the main floor: Booth #5734
Advancing Laboratory Interoperability in Health IT
 Advancing Laboratory Interoperability in Health IT Introduction & CDC LabHIT Update Megan E. Sawchuk, MT(ASCP) Health Scientist Clinical Laboratory Improvement Advisory Committee (CLIAC) 8-22-2013 Center
Advancing Laboratory Interoperability in Health IT Introduction & CDC LabHIT Update Megan E. Sawchuk, MT(ASCP) Health Scientist Clinical Laboratory Improvement Advisory Committee (CLIAC) 8-22-2013 Center
THE STIMULUS AND STANDARDS. John D. Halamka MD
 THE STIMULUS AND STANDARDS John D. Halamka MD THE ONC STRATEGY Grants - Accelerating Adoption Standards - Interim Final Rule Meaningful Use - Notice of Proposed Rulemaking Certification - Notice of Proposed
THE STIMULUS AND STANDARDS John D. Halamka MD THE ONC STRATEGY Grants - Accelerating Adoption Standards - Interim Final Rule Meaningful Use - Notice of Proposed Rulemaking Certification - Notice of Proposed
HIMSS Electronic Health Record Definitional Model Version 1.0
 HIMSS Electronic Health Record Definitional Model Version 1.0 Prepared by HIMSS Electronic Health Record Committee Thomas Handler, MD. Research Director, Gartner Rick Holtmeier, President, Berdy Systems
HIMSS Electronic Health Record Definitional Model Version 1.0 Prepared by HIMSS Electronic Health Record Committee Thomas Handler, MD. Research Director, Gartner Rick Holtmeier, President, Berdy Systems
HL7 Clinical Document Architecture: Overview and Applications
 HL7 Clinical Document Architecture: Overview and Applications Nawanan Theera-Ampornpunt, M.D., Ph.D. Department of Community Medicine Faculty of Medicine Ramathibodi Hospital Certified HL7 CDA Specialist
HL7 Clinical Document Architecture: Overview and Applications Nawanan Theera-Ampornpunt, M.D., Ph.D. Department of Community Medicine Faculty of Medicine Ramathibodi Hospital Certified HL7 CDA Specialist
International Organization for Standardization TC 215 Health Informatics. Audrey Dickerson, RN MS ISO/TC 215 Secretary
 International Organization for Standardization TC 215 Health Informatics Audrey Dickerson, RN MS ISO/TC 215 Secretary 1 Topics Introduction to ISO TC 215, Health Informatics Definitions Structure Membership
International Organization for Standardization TC 215 Health Informatics Audrey Dickerson, RN MS ISO/TC 215 Secretary 1 Topics Introduction to ISO TC 215, Health Informatics Definitions Structure Membership
Privacy and Security within an Interoperable EHR
 1 Privacy and Security within an Interoperable EHR Stan Ratajczak Director Privacy and Security Solutions Architecture Group November 30, 2005 Electronic Health Information and Privacy Conference Ottawa
1 Privacy and Security within an Interoperable EHR Stan Ratajczak Director Privacy and Security Solutions Architecture Group November 30, 2005 Electronic Health Information and Privacy Conference Ottawa
HIPAA: AN OVERVIEW September 2013
 HIPAA: AN OVERVIEW September 2013 Introduction The Health Insurance Portability and Accountability Act of 1996, known as HIPAA, was enacted on August 21, 1996. The overall goal was to simplify and streamline
HIPAA: AN OVERVIEW September 2013 Introduction The Health Insurance Portability and Accountability Act of 1996, known as HIPAA, was enacted on August 21, 1996. The overall goal was to simplify and streamline
Custom Report Data Elements: IT Database Fields. Source: American Hospital Association IT Survey
 Custom Report Data Elements: IT Database Fields Source: American Hospital Association IT Survey TABLE OF CONTENTS COMPUTERIZED SYSTEM IMPLEMENTATION... 4 Bar Coding... 4 Computerized Provider Order Entry...
Custom Report Data Elements: IT Database Fields Source: American Hospital Association IT Survey TABLE OF CONTENTS COMPUTERIZED SYSTEM IMPLEMENTATION... 4 Bar Coding... 4 Computerized Provider Order Entry...
Agenda. What is Meaningful Use? Stage 2 - Meaningful Use Core Set. Stage 2 - Menu Set. Clinical Quality Measures (CQM) Clinical Considerations
 AQAF Health Information Technology Forum Meaningful Use Stage 2 Clinical Considerations Marla Clinkscales & Mike Bice Alabama Regional Extension Center (ALREC) August 13, 2013 0 Agenda What is Meaningful
AQAF Health Information Technology Forum Meaningful Use Stage 2 Clinical Considerations Marla Clinkscales & Mike Bice Alabama Regional Extension Center (ALREC) August 13, 2013 0 Agenda What is Meaningful
Title Draft Pan-Canadian Primary Health Care Electronic Medical Record Content Standard, Version 2.0 Data Extract Specifi cation Business View
 pic Title Draft Pan-Canadian Primary Health Care Electronic Medical Record Content Standard, Version 2.0 Data Extract Specifi cation Business View Primary Health Care Who We Are Established in 1994, CIHI
pic Title Draft Pan-Canadian Primary Health Care Electronic Medical Record Content Standard, Version 2.0 Data Extract Specifi cation Business View Primary Health Care Who We Are Established in 1994, CIHI
Measuring the Interoperability Degree of Interconnected Healthcare Information Systems Using the LISI Model
 Measuring the Interoperability Degree of Interconnected Healthcare Information Systems Using the LISI Model Mihaela Vida*, Lăcrămioara Stoicu-Tivadar*, Elena Bernad**, *Faculty of Automatics and Computers,
Measuring the Interoperability Degree of Interconnected Healthcare Information Systems Using the LISI Model Mihaela Vida*, Lăcrămioara Stoicu-Tivadar*, Elena Bernad**, *Faculty of Automatics and Computers,
Stage 1 measures. The EP/eligible hospital has enabled this functionality
 EMR Name/Model Ingenix CareTracker - version 7 EMR Vendor Ingenix CareTracker Stage 1 objectives Use CPOE Use of CPOE for orders (any type) directly entered by authorizing provider (for example, MD, DO,
EMR Name/Model Ingenix CareTracker - version 7 EMR Vendor Ingenix CareTracker Stage 1 objectives Use CPOE Use of CPOE for orders (any type) directly entered by authorizing provider (for example, MD, DO,
JA to support the ehealth Network
 JA to support the ehealth Network ehealth Network & ehgi Directive 2011/24/EU of the European Parliament and of the Council on the application of patients` rights in cross-border healtcare 08.05.2015 2
JA to support the ehealth Network ehealth Network & ehgi Directive 2011/24/EU of the European Parliament and of the Council on the application of patients` rights in cross-border healtcare 08.05.2015 2
Leveraging Health Information Exchange to Improve Quality and Efficiency
 Leveraging Health Information Exchange to Improve Quality and Efficiency Presentation for the TIGER Initiative November 1, 2007 Joyce Sensmeier MS, RN-BC, CPHIMS, FHIMSS Objectives Examine the role of
Leveraging Health Information Exchange to Improve Quality and Efficiency Presentation for the TIGER Initiative November 1, 2007 Joyce Sensmeier MS, RN-BC, CPHIMS, FHIMSS Objectives Examine the role of
HIMSS Interoperability Showcase 2011
 Interoperability will bind together a wide network of real-time life critical data that not only transform but become healthcare. Health Information Interoperability Challenges and Integrating Healthcare
Interoperability will bind together a wide network of real-time life critical data that not only transform but become healthcare. Health Information Interoperability Challenges and Integrating Healthcare
