Use of imaging in the management of malignant pleural mesothelioma
|
|
|
- Bryce Marshall
- 8 years ago
- Views:
Transcription
1 Clinical Radiology (2005) 60, REVIEW Use of imaging in the management of malignant pleural mesothelioma R.E. Benamore a, M.J. O Doherty b, J.J. Entwisle a, * a Department of Radiology, University Hospitals of Leicester, Leicester, UK b Clinical PET Centre, Guy s and St Thomas Hospital, London, UK Received 26 January 2005; received in revised form 23 May 2005; accepted 25 May 2005 KEYWORDS Mesothelioma; Pleura; Computed tomography; CT; Biopsy Malignant pleural mesothelioma (MPM) is an increasingly prevalent tumour. The death rate associated with MPM is predicted to peak in the next 10 years, although radiologists and clinicians will be encountering cases for the next few decades. Contrast-enhanced CT is an established technique for evaluating suspected malignant pleural disease, but MPM can be reliably diagnosed only by histological sampling. However, even with adequate sampling and the use of immunocytochemistry, histological diagnosis is known to be difficult; definitive diagnosis may involve a combination of clinical presentation, radiological and histological appearances. Percutaneous biopsy is a promising technique for sampling the pleura. In view of its pattern of growth, MPM is a challenging disease to image by any method, and it behaves quite differently from lung cancer. This review aims to highlight the practical aspects of assessing malignant pleural mesothelioma. Q 2005 The Royal College of Radiologists. Published by Elsevier Ltd. All rights reserved. Introduction Malignant pleural mesothelioma (MPM) currently accounts for 1700 deaths per year in the UK, but is forecast to peak at 1950 to 2450 deaths per year between 2011 and Although the peak of the epidemic may last for 10 to 15 years, the tail may last until ,2 MPM is predicted to become one of the more common cancers, with a higher mortality than melanoma, uterine or cervical cancer. 1 Men born in the late 1940s are predicted to be at the highest risk of dying from MPM 3 and, although rare in the first 10 to 15 years after exposure to asbestos, the risk of mesothelioma increases thereafter with time. 4,5 MPM has a high association with asbestos exposure, 5 but there is no relevant history in up to 20% of cases. 6 MPM should be considered in all cases of unilateral pleural * Guarantor and correspondent: J.J. Entwisle, Department of Radiology, Glenfield Hospital, Groby Road, Leicester LE3 9QP, UK. Tel.: C ; fax: C address: james.entwisle@uhl-tr.nhs.uk (J.J. Entwisle). abnormalities and chest pain, and in cases of chest pain with a history of asbestos exposure. The biological behaviour of MPM is very different to that seen in lung cancer, and there are many difficulties in diagnosis, staging and assessment of response. There are a number of ongoing or planned trials in the UK. Surgery may have a role in a limited number of cases, and preoperative assessment holds further challenges. We present a review of the radiological techniques involved in the management of MPM. Pleural anatomy and lymph node drainage The anatomy of the pleura is complex and is not always appreciated. 7 The inferior margins of the pleura in the posterior costodiaphragmatic recesses of the hemithorax extend considerably lower than the corresponding border of the lung, to the level of the 12th dorsal vertebra. The diaphragm extends more inferiorly, and the right crus arises from /$ - see front matter Q 2005 The Royal College of Radiologists. Published by Elsevier Ltd. All rights reserved. doi: /j.crad
2 1238 R.E. Benamore et al. the anterolateral surfaces of the bodies and intervertebral discs of the upper three lumbar vertebrae. In cases of established mesothelioma being considered for radical treatment, the entire pleural and diaphragmatic surfaces need to be scanned contiguously from the thoracic inlet to the level of the third lumbar vertebra. This allows coronal and sagittal images to be reconstructed. The lymphatic drainage of the pleura is equally complex. Although the visceral pleural lymphatics follow the same pattern of drainage as the lung, the lymphatic drainage of the parietal pleura is very different. The anterior parietal pleura drains to internal mammary nodes. The posterior parietal pleura drains to extrapleural lymph nodes, which lie in the paraspinal fat adjacent to the heads of the ribs. The diaphragm is commonly involved in MPM; the anterior and lateral diaphragmatic lymphatics drain to internal mammary and anterior peridiaphragmatic nodes. The posterior diaphragm drains to para-aortic and posterior mediastinal nodes. There are free anastomoses between lymphatics on both surfaces of the diaphragm, and coeliac axis or gastrohepatic nodal enlargement may be seen. 7,8 In practice, the lymphatic drainage can be thought of as a circle of nodes in the extrapleural space. Any nodes visualized here should be viewed with suspicion. In addition to inspection of the thorax, a thorough search for enlarged nodes should be made in the retrocrural region and upper abdomen. First-line imaging techniques Chest radiograph A plain chest radiograph is usually the first imaging investigation to be performed, and here the appearances of MPM range from normal in early disease to complete opacification of a hemithorax. These findings depend on the differing amounts of pleural thickening and fluid. A review of 4710 cases 9 showed that a pleural effusion is usually present on the initial radiograph. The mediastinum can be central or it can be displaced towards or away from the affected hemithorax. The pleural thickening can manifest as discrete pleural nodules or can progressively encase the lung. Pleural masses may be visible, often after drainage of the effusion. Rib destruction or pneumothorax may occasionally be seen. 9 Right-sided tumours are more common, accounting for approximately 60% of MPM. 5 The differential for the plain chest radiograph appearances of MPM includes unilateral diffuse pleural thickening; this is more commonly bilateral and relatively symmetrical. An unexplained unilateral pleural effusion or pleural thickening warrants further investigation with contrast-enhanced CT. 10 Ultrasound Ultrasound (US) can be very useful in identifying pathology of the pleura. The presence of a pleural effusion acts as an acoustic window and can enable the detection of intrapleural and intrapulmonary processes. Pleural effusions and thickening can be readily appreciated by US, and discrete malignant nodules may be seen. 11 US-guided biopsy of pleural thickening and drainage of effusions are wellestablished, safe techniques. Computed tomography Contrast-enhanced CT is the primary imaging method for the evaluation of MPM. Malignant or inflammatory pleural disease enhances strongly, and the contrast enables differentiation between thickened pleura, effusion and underlying aerated or collapsed lung. With modern multidetector CT (MDCT), imaging of the entire chest can be completed in under 10 s. The use of narrow collimation of less than 1 mm in thickness allows the final images to be visualized in any plane. Coronal and sagittal reconstructions are particularly useful for assessing chest wall or subdiaphragmatic invasion. In cancer staging, the thorax is normally scanned in the arterial phase. With a delay of 20 to 60 s, pleural thickening can be distinguished from pleural fluid. 12 However, it is our experience that pleural enhancement is more delayed than parenchymal lung enhancement, and with suspected malignant pleural disease there is a case for scanning in a more delayed phase at 45 s. CT makes possible a detailed evaluation of the pleura and differentiation of benign from malignant pleural disease. Leung et al. 13 studied 74 consecutive patients with diffuse pleural disease. CT features used to distinguish malignant from benign pleural disease included circumferential pleural thickening, nodular pleural thickening, parietal pleural thickening greater than 1 cm and mediastinal pleural involvement. The specificities of these findings were 100%, 94%, 94% and 88%, respectively. The sensitivities were 41%, 51%, 36% and 56%, respectively. Whereas the positive predictive value of these signs is high, the absence of the various signs does not exclude a diagnosis of MPM or metastatic pleural adenocarcinoma. Using these criteria, Traill et al. 14 studied 40 consecutive patients; contrast-enhanced CT correctly identified
3 Use of imaging in the management of malignant pleural mesothelioma /32 cases of malignant disease and all 8 cases of benign disease, giving a sensitivity of 84% and a specificity of 100% for the technique. This study found circumferential thickening a less reliable indicator for malignancy, and this was observed in the benign and malignant groups equally. Metintas et al. 15 studied the CT features of benign and malignant pleural disease in 215 cases, and confirmed high specificities for circumferential pleural thickening, nodularity and mediastinal pleural involvement, with a 64% specificity for pleural thickening greater than 1 cm. It is important in cases of suspected pleural malignancy to differentiate between MPM and other pleural malignancy, because the prognosis, treatment and compensation issues are very different. Differentiation would include a full clinical history and examination, and there are certain imaging features which, when used in combination with the clinical findings, are suggestive of MPM. Metinas et al. 15 reviewed the CT findings in 215 cases, 99 of MPM, 39 of metastatic pleural disease and 77 of benign pleural disease. In cases of MPM, the most common CT features were circumferential nodular lung encasement, pleural thickening with irregular pleuropulmonary margins and pleural thickening with superimposed nodules (Fig. 1). In 70% of cases of MPM, there was rind-like extension of tumour on the pleural surfaces. On multivariate analysis, the CT findings of rindlike pleural involvement, mediastinal pleural involvement and pleural thickness O1 cm were independent findings in differentiating MPM from other malignant pleural disease, with sensitivity/ specificity values of 0.7/0.85, 0.85/0.67 and 0.59/0.82, respectively. A definitive diagnosis of MPM normally requires cytological or histological sampling, but CT findings may be important for patients with poor performance status, who do not want or are not fit for any invasive biopsy procedures. There can be contraction of the affected hemithorax with associated ipsilateral mediastinal shift, narrowed intercostal spaces and elevation of the ipsilateral hemidiaphragm. Contralateral mediastinal shift is usually due to large effusions (Table 1) CT evidence of distant metastases is rare at presentation and may indicate metastatic pleural disease. Pleural plaques are found at CT in approximately 20% of cases of MPM (Fig. 1) and there may be other features of asbestos exposure. Asbestosis, however, is rare in the presence of mesothelioma. 17 MPM is locally aggressive; chest wall involvement is commonly seen at the site of previous intervention, including biopsy, drain and surgical sites. 20 There should be careful evaluation of these areas, and they may be marked with a suitable radioopaque marker to aid this. Chest wall involvement may manifest as obliteration of extrapleural fat planes, invasion of intercostal muscles, displacement of ribs or bone destruction (Fig. 2). Direct extension of tumour into the major vessels and mediastinum may occur, and is usually manifested as a soft-tissue mass surrounding more than 50% of the structure. 21,22 MPM can invade the pericardium and may be seen at CT as nodular pericardial thickening or pericardial effusion (Fig. 3). The diaphragm does not form a continuous barrier between the pleural and peritoneal cavities, and difficulties can arise in deciding whether the tumour has actually invaded the peritoneum, particularly around the costophrenic angles. 23 Detection of diaphragmatic invasion has proved difficult on single-slice axial CT; the most reliable sign is a soft-tissue mass encasing the diaphragm. A smooth diaphragmatic contour and a clear fat plane between the inferior surface of the diaphragm and the adjacent abdominal organs suggest that tumour does not extend through the diaphragm. 21 However, axial CT has limited sensitivity and specificity in detecting these features, and at our centre we always reconstruct images in the coronal and sagittal planes (Fig. 4). MRI has hitherto had an advantage in view of its superior tissue contrast and multiplanar capability; however, the latter function is likely to be superseded by MDCT. Figure 1 Axial contrast-enhanced CT. Nodular enhancing pleura on the right extends onto the mediastinal surface (arrow). Note contralateral pleural plaques. Image-guided biopsy Pleural fluid cytology has a low sensitivity (26% to 32%) for diagnosis of MPM, 23,24 and reliable
4 1240 R.E. Benamore et al. Table 1 CT features of malignant pleural mesothelioma. Common features Uncommon features Distinctive features a Irregular/nodular pleural thickening Discrete pleural nodules or masses Rind-like pleural involvement Rind-like pleural thickening Rib destruction Mediastinal pleural involvement Pleural effusion Vertebral body erosion Pleural thickness O1cm Mediastinal pleural thickening Mediastinal invasion Pleural plaques Interlobar fissural involvement Diaphragmatic invasion Volume contraction Chest wall invasion a These features distinguish malignant pleural mesothelioma from other pleural malignancies. pathological diagnosis requires histological sampling. Historically, the standard technique for pleural biopsy has been to use a reverse-bevelled needle such as the Abrams needle. Even in experienced hands, Abrams biopsy has a sensitivity of only 55%, 26 probably because of the patchy distribution of MPM and its predilection for the basal and diaphragmatic pleural surfaces. Image-guided pleural biopsy can be performed as a day-case procedure. Several studies have assessed both CT- and US-guided pleural biopsies, with sensitivities of 86% to 93%, specificities of 100% and a low (3%) complication rate. 27,28 US-guided pleural biopsy has a sensitivity of 77% and a specificity of 88% for the diagnosis of MPM, with minor complications only. It seems that 18G or 14G needle gauge has little effect on diagnostic accuracy. 29 Maskell et al. 26 randomized 50 patients with suspected malignant pleural effusions to have either a standard Abrams needle pleural biopsy or CT-guided biopsy. For diagnosis of mesothelioma, CT-guided biopsy has a sensitivity of 88% versus 55% for Abrams biopsy. CT-biopsy as the initial procedure would have averted a repeat biopsy in 40% of patients. By performing core-needle biopsy along the line of the pleura, samples can be taken from pleural thickening as little as 2 mm thick. However, angled biopsies of the pleura may create confusing histology, as the presence of mesothelial cells deep to the pleural surface, a sign of malignancy, may be a result of the angle at which the biopsy has been taken. Tumour seeding along the track has been reported in up to 22% of cases, and post-procedure pneumothorax rates are as high Figure 2 (a) Axial contrast-enhanced CT, (b) reconstructed in the coronal plane, shows an advanced rightsided malignant pleural mesothelioma. Note multifocal chest wall (arrows) and subdiaphragmatic invasion. Figure 3 Axial contrast-enhanced MDCT. The pericardium is thickened, but there is no deep pericardial invasion (IMIG T4). This was confirmed at operation.
5 Use of imaging in the management of malignant pleural mesothelioma 1241 provide information about metabolically active areas and, when findings are correlated with anatomical imaging information, PET may be used to help determine the most appropriate biopsy site for obtaining positive results. 31 This is particularly useful in cases with an initial suspected falsenegative result. A prospective multicentre trial comparing cutting-needle percutaneous image-guided biopsy and thoracoscopic biopsy would help clarify the potential advantages and cost-effectiveness of each sampling technique. Additional imaging techniques Positron emission tomography Figure 4 Contrast-enhanced CT shows infradiaphragmatic invasion. (a) Axial view: The diaphragm has been invaded by tumour, with loss of the fat plane between diaphragm and liver and flattening of the superior surface of the liver. This is much more clearly appreciated on (b) the coronal reconstruction. as 9.5%, 30 but it seems rates are lower in the presence of a pleural effusion. Thoracoscopic biopsy has the advantage of direct visualization of the pleura but, for this technique, the visceral and parietal pleura must not be adherent. In a prospective study of 188 patients with suspected MPM, thoracoscopic biopsy was compared with pleural fluid cytology and Abram s needle biopsy. 25 Thoracoscopic biopsy yielded sensitivities of 98.4% versus 38.7% for combined pleural fluid cytology and Abrams biopsy. Seeding along the biopsy track can occur in up to 50% of such cases. FDG positron emission tomography (PET) can F-18 fluorodeoxyglucose positron emission tomography (FDG PET) may be a useful problem-solving tool in differentiating benign from malignant pleural disease, with a sensitivity for detecting malignancy of 96.8% and a specificity of 88.5% in 63 cases 32 (Fig. 5), and appears to confirm malignant pleural disease which cannot be identified at CT. In a prospective study, Kramer et al. 33 studied 32 patients, 19 with malignant and 13 with benign disease, and found that FDG PET had a high negative predictive value of 92%. FDG PET imaging showed an absence of FDG uptake, and correctly classified 31/35 benign lesions. False-positive results were seen in cases of parapneumonic effusion and tuberculous and uraemic pleural disease. The standardized uptake value (SUV) is used as a semi-quantitative measure of the metabolic activity of a lesion. However, there is no consensus as to the time point at which to image MPM. The SUV is significantly higher in MPM than in benign pleural diseases such as inflammatory pleuritis and Figure 5 CT-PET showing (from left to right) coronal CT, PET and fused images of a left-sided malignant pleural mesothelioma. No evidence of contralateral disease or metastasis is seen.
6 1242 R.E. Benamore et al. asbestos-related pleural plaques. 34 However, some MPMs are low-grade tumours and may not be intensely hypermetabolic on FDG PET. There is dispute as to whether the SUV correlates with T stage, 35 but the SUV appears to be higher in cases of nodal and metastatic disease and is associated with an unfavourable prognosis The SUV does not appear to be related to histological grade. Caution should be taken with patients who have previously undergone talc pleurodesis, as the inflammatory process caused by this procedure can cause a falsepositive result. Correlation with CT is needed to demonstrate the high-attenuation pleural thickening due to talc administration. 38 Areas of pleural thickening may not necessarily correspond to areas of high metabolic activity, and the most appropriate biopsy site may not be apparent from CT results. Metastases are a common finding at post-mortem, 5 and FDG PET may have a role in detecting occult metastases before consideration for surgery. 39 The combination of PET and CT in PET-CT scanners opens up the possibility of performing diagnostic CT and FDG PET in order to assess the patient at one visit, or of undertaking a low-quality CT and using this to register to an earlier diagnostic CT. Potential problems relate to image registration, which may not be accurate because of respiratory motion and differences between the two imaging methods. It is also possible that FDG is not the optimum tracer, and that one of the other tracers may be more suitable for imaging. This, however, remains to be seen. patients and compared CT with MRI. High signal intensity in relation to intercostal muscles on T2- weighted or contrast-enhanced T1-weighted images or both was significantly suggestive of malignant disease. Using morphological features in combination with the signal intensity features, MRI showed a sensitivity of 100% and a specificity of 93% in the detection of pleural malignancy and was thought superior to CT, particularly in detecting chest wall and diaphragmatic invasion (Fig. 6). However, MDCT can now provide 3D images. MRI is most useful in evaluating patients with questionable areas of local tumour extension at CT, or patients for whom intravenous administration of iodinated contrast material is contraindicated. Magnetic resonance imaging Relative to adjacent chest wall muscle, MPM is typically isotense or slightly hyperintense on T1- weighted images and moderately hyperintense on proton density and T2-weighted images. MPM enhances avidly with use of gadolinium-based contrast material. Anatomical and morphological magnetic resonance imaging (MRI) features similar to those seen at CT are used to establish local invasion of MPM. For patients being assessed for surgery, MRI can provide additional staging information. In a comparative study of MRI and CT, Knuuttila et al. 40 found that contrast-enhanced MRI was superior in demonstrating focal thickening and enhancement of interlobar fissures, which are useful signs in the detection of early malignant pleural disease. On the other hand, enhancement may be seen following pleurodesis of a benign effusion. Hierholzer et al. 41 retrospectively studied 42 Figure 6 Same case as Fig. 2. (a) Axial and (b) sagittal contrast-enhanced T1-weighted MRI shows an advanced right-sided malignant pleural mesothelioma. The chest wall invasion is clearly appreciated on MRI by means of the tissue contrast (asterisks). There is also subdiaphragmatic invasion.
7 Use of imaging in the management of malignant pleural mesothelioma 1243 Stewart et al. 42 found that 17/76 patients who were potential surgical candidates by CT criteria had unresectable disease on MRI. Staging and resectability The TNM staging system proposed by the International Mesothelioma Interest Group is used for patients with potentially resectable disease. 43 Emphasis is placed on criteria for determining the extent of local tumour and lymph node involvement, both of which affect overall survival. Patients with T1 to T3 disease are potential surgical candidates but may not necessarily be cured. The staging system was designed as a surgical tool, and may not be completely applicable to imaging. In practical terms, detection on CT of deep myocardial invasion, multifocal chest wall invasion or subdiaphragmatic extension is a contraindication to surgery. Limitations of cross-sectional and functional imaging in staging and determining resectability Imaging has a tendency to understage the extent of MPM, which is crucial in determining which cases are potentially suitable for resection (i.e., in distinguishing T3 from T4 disease). Rusch et al. 44 followed 20 patients who underwent thoracotomy. CT missed chest wall invasion in 7/18 cases, was falsely positive in 2 cases and was also poor at detecting involvement of the diaphragm. Heelan et al. 45 studied 65 patients who underwent CT, MR and resection of MPM. They found that CT and MR overall showed fairly similar accuracies for staging MPM, i.e. between 50% and 65%. Both techniques had limited accuracies for detecting T1 and nodal disease, but MR was superior to CT for detecting T2 disease (invasion of diaphragm and endothoracic fascia). A more recent study by Patz et al. 21 suggested that CT and MR were sensitive in predicting resectability, but the statistics may have been skewed by the study population. This problem may be solved with multidetector CT. In a study comparing MRI and surgical stage, MRI understaged 50% of tumours, primarily because of understaging pericardial involvement, although this may not have a significant effect on prognosis. 42 Because of its poor spatial resolution, FDG PET is unsuitable 35 for defining T status; the data take a significant time to acquire and images are prone to movement artefact, which may be problematic when pleura measuring only a few millimetres in thickness is being imaged. PET should therefore not be used as a tool for determining resectability. Mediastinal nodes are commonly involved with tumour 46 and, as with staging lung cancer, CT has limited accuracy for detecting this; sensitivity is 60% and specificity 70%. Involved nodes may not be enlarged, and ipsilateral mediastinal nodes are often obscured by irregular pleural thickening along the mediastinal border. FDG PET appears to be relatively poor at defining mediastinal nodal metastasis (N2 disease) from adjacent mediastinal pleural involvement. In a study by Flores et al., 35 FDG PET missed mediastinal nodal metastases in 8/9 patients, although a high SUV seemed to correlate with the presence of N2 disease. Reliable detection of nodes by PET may be particularly difficult if there is very nodular pleural disease adjacent to the potentially involved nodes in the internal mammary chain and mediastinal groups. Sugarbaker et al. 47 found that patients with negative extrapleural (i.e., N2 or N3) nodes had significantly better survival (42% at 2 years) than those whose nodes were involved (23% at 2 years). Routine mediastinoscopy may have a role, but how this is performed and which stations are sampled has not been fully studied. 46,48 The procedure targets only limited nodes, which may not be those involved in MPM. Even surgical dissection at operation does not evaluate all N3 nodes (or occult metastases). However, FDG PET may have a role in detecting N3 or M1 disease, and PET-CT appears to be a promising technique. However, there are no published studies of its use in MPM at present. Treatment options Radical surgery Extrapleural pneumonectomy entails removal of the lung and all the parietal pleura, pericardium and diaphragm on the affected side. It usually forms part of trimodality treatment, combined with preoperative and postoperative chemotherapy and radiotherapy. In selected patients, 5-year survival can reach nearly 50%. 49 A large prospective trial over the next 8 years, the Mesothelioma and Radical Surgery (MARS) trial, aims to determine whether radical surgery alters prognosis. Results will therefore be available in time for the peak of the epidemic. Postoperative appearances following radical surgery The chest radiograph findings after extrapleural
8 1244 R.E. Benamore et al. pneumonectomy are often indistinguishable from those of a standard pneumonectomy, although the fluid accumulates more rapidly within the postpneumonectomy space, i.e. over several days rather than weeks. 50 CT or MRI shows a rim of fibrous tissue lining the pneumonectomy space (Fig. 7). This should not be confused with recurrent disease; it is a few millimetres thick and uniform. Complications, which are similar to those of a standard pneumonectomy, may be first suspected radiologically and include bronchial stump fistula, where there is failure of the pneumonectomy space to fill with fluid. The diaphragm is normally removed and replaced by a synthetic patch 51 which is susceptible to rupture, particularly on the left. This may manifest on the postoperative chest radiograph as sudden elevation of the neodiaphragm, with findings confirmed easily by CT. This is important to recognize and indicates immediate surgery. As part of the extrapleural pneumonectomy, some of the pericardium is removed, which can potentially lead to the rare complication of cardiac volvulus. A pericardial patch is normally inserted to prevent this complication. It is likely, as with non-small-cell lung cancer, that if FDG PET were positive preoperatively in this patient group, recurrence could be identified early within the hemithorax from which the lung had been resected. However, this has not been demonstrated in any studies to date. Figure 7 Contrast-enhanced MDCT reconstructed in the coronal plane. There has been a left extrapleural pneumonectomy. Note the smooth enhancing rind and synthetic diaphragmatic patch. Assessing response to chemotherapy Reduction in tumour size is now widely accepted as evidence of tumour response, and the effectiveness of treatment cannot be assessed without robust mechanisms of tumour measurement. The World Health Organization (WHO) introduced measurement criteria in However, it became apparent that tumour measurement using WHO criteria was very laborious. The response evaluation criteria in solid tumours (RECIST) were proposed and validated in RECIST is based on the assumption that tumours are generally spherical, and that a maximal unidimensional measurement correlates well with an overall reduction in tumour size. MPM tends to grow as a rind around the chest wall, and changes in tumour bulk tend to manifest as changes in tumour thickness perpendicular to the chest wall, rather than the maximal tumour dimension. RECIST therefore cannot be applied easily, and MPM was not included in the initial studies. Studies have shown discrepancies between tumour measurement in MPM using the WHO and RECIST criteria. 54 These can occur in up to 47% of cases, in which the majority of cases were underscored by RECIST. Disease progression also was underscored by RECIST in 24% of cases. 55 Tumour bulk may in some cases consist of mainly fibrous tissue, which often shows little immediate change in size, even if the sparse tumour cells within it have responded to treatment. Fibrosis can lead to shrinkage of the affected hemithorax, making it difficult to find the same measurement points on subsequent examination. 23 Difficulties also arise for patients who have undergone pleurodesis, as they have residual pleural thickening even in the absence of active disease (Fig. 8). Byrne et al. 56 have validated modified RECIST criteria for assessing response of MPM to chemotherapy in 73 cases. They made two measurements of tumour thickness perpendicular to the chest wall at three separate reproducible anatomical levels within the chest. The sum of the six measurements provides a pleural unidimensional measurement. Other bidimensionally measurable lesions were measured as per RECIST and added to the pleural unidimensional measurement. The percentage change in measurements between scans was as for RECIST. Changes in forced vital capacity, a surrogate marker for patient well-being, also correlated well with changes in tumour measurement. In the future, computer-assisted techniques for
9 Use of imaging in the management of malignant pleural mesothelioma 1245 change in tumour bulk in malignancies such as mesothelioma. Conclusions MPM is a disease with an increasing incidence, and radiologists and clinicians will be seeing patients with the disease well into the 21st century. MPM behaves quite differently from lung cancer, and there are many difficulties in TNM staging and assessing response to chemotherapy. There is a need for a more practical, radiology-friendly staging system, as the majority of patients do not undergo radical surgery. CT-PET may hold the key to more accurate assessment, by providing further information on anatomy and metabolic function. References Figure 8 Malignant pleural mesothelioma before and after pleurodesis. (a) Contrast-enhanced axial MDCT shows minor right-sided pleural thickening. (b) Following pleurodesis, the pleura is thicker and there has been volume loss within the right hemithorax. These appearances pose challenges when trying to assess response to chemotherapy. measurement of MPM may play a role in assessing tumour response. 57 The RECIST criteria are limited in all tumour groups, since size change is slow and metabolic change should be rapid. Carretta et al. 58 observed a reduction in FDG uptake in a small number of cases with MPM following chemotherapy. Choosing when to scan following chemotherapy, and the timing of the scan following the injection of FDG, are likely to be crucial to this assessment. It is, however, likely that this change in metabolic activity would precede any change in volume, and would also be easier to assess than a 1. National Statistics. Mesothelioma mortality in Great Britain: Estimating the future burden. London: Health and Safety Executive; From cancer research UK statistics, mortality; mortality? versionz1, accessed May Peto J, Decarli A, la Vecchia C, et al. The European mesothelioma epidemic. Br J Cancer 1999;79: Peto J, Seidman H, Selikoff IJ. Mesothelioma mortality in asbestos workers: Implications for models of carcinogenesis and risk assessment. Br J Cancer 1982;45: Yates DH, Corrin B, Stidolph PN, et al. Malignant mesothelioma in south east England: Clinicopathological experience in 272 cases. Thorax 1997;52: Gibbs AR. Role of asbestos and other fibres in the development of diffuse malignant mesothelioma. Thorax 1990;45: Williams PL, editor. Gray s anatomy. 38th ed. London: Churchill Livingstone; 1995; p , , Sharma A, Fidias P, Hayman LA, et al. Patterns of lymphadenopathy in thoracic malignancies. RadioGraphics 2004;24: Hillerdal G. Malignant mesothelioma 1982: Review of 4710 published cases. Br J Dis Chest 1983;77: British Thoracic Society Standards of Care Committee. Statement on malignant mesothelioma in the United Kingdom. Thorax 2001;56: Herth F. Diagnosis and staging of mesothelioma transthoracic ultrasound. Lung Cancer 2000;45:S63 S Evans AL, Gleeson FV. Radiology in pleural disease: State of the art. Respirology 2004;9: Leung AN, Muller NL, Miller RR. CT in differential diagnosis of diffuse pleural disease. AJR Am J Roentgenol 1990;154: Traill ZC, Davies RJ, Gleeson FV. Thoracic computed tomography in patients with suspected malignant pleural effusions. Clin Radiol 2001;56: Metintas M, Ucgun I, Elbek O, et al. Computed tomography features in malignant pleural mesothelioma and other commonly seen pleural diseases. Eur J Radiol 2002;41:1 9.
10 1246 R.E. Benamore et al. 16. Senyigit A, Bayram H, Babayigit C, et al. Malignant pleural mesothelioma caused by environmental exposure to asbestos in the southeast of Turkey: CT findings in 117 patients. Respiration 2000;67: Sahin AA, Coplu L, Selcuk ZT, et al. Malignant pleural mesothelioma caused by environmental exposure to asbestos and erionite in rural Turkey: CT findings in 84 patients. AJR Am J Roentgenol 1993;161: Ng CS, Munden RF, Libshitz HI. Malignant pleural mesothelioma: The spectrum of manifestations on CT in 70 cases. Clin Radiol 1999;54: Kawashima A, Libshitz HI. Malignant pleural mesothelioma: CT manifestations in 50 cases. AJR Am J Roentgenol 1990; 155: Boutin C, Rey F, Viallat J-R. Prevention of malignant seeding after invasive diagnostic procedures in patients with pleural mesothelioma. Chest 1995;108: Patz EF, Shaffer K, Piwnica-Worms DR, et al. Malignant pleural mesothelioma: Value of CT and MR imaging in predicting resectability. AJR Am J Roentgenol 1992;159: Marom EM, Erasmus JJ, Pass HI, et al. The role of imaging in malignant pleural mesothelioma. Semin Oncol 2002;29: Hillerdal G. Staging and evaluating responses in malignant pleural mesothelioma. Lung Cancer 2004;43: Renshaw AA, Dean BR, Antman KH, et al. The role of cytologic evaluation of pleural fluid in the diagnosis of malignant mesothelioma. Chest 1997;111: Boutin C, Rey F. Thoracoscopy in pleural malignant mesothelioma: A prospective study of 188 consecutive patients. Part 1. Diagnosis. Cancer 1993;72: Maskell NA, Gleeson FV, Davies RJO. Standard pleural biopsy versus CT-guided cutting needle biopsy for diagnosis of malignant disease in pleural effusions: A randomised controlled trial. Lancet 2003;361: Adams RF, Gray W, Davies RJO, et al. Percutaneous imageguided cutting needle biopsy of the pleura in the diagnosis of malignant mesothelioma. Chest 2001;120: Adams RF, Gleeson FV. Percutaneous image-guided cutting needle biopsy of the pleura in the presence of a suspected malignant pleural effusion. Radiology 2001;219: Heilo A, Stenwig AE, Solheim OP. Malignant pleural mesothelioma: US-guided histologic core-needle biopsy. Radiology 1999;211: Metintas M, Ozdemir N, Isiksoy S, et al. CT-guided pleural needle biopsy in the diagnosis of malignant mesothelioma. J Comput Assist Tomogr 1995;19: Ng DC, Hain SF, O Doherty MJ, et al. Prognostic value of FDG PET imaging in malignant pleural mesothelioma. J Nucl Med 2000;41: Duysinx B, Nguyen D, Louis R, et al. Evaluation of pleural disease with 18-fluorodeoxyglucose positron emission tomography imaging. Chest 2004;125: Kramer H, Pieterman RM, Slebos D-J, et al. PET for the evaluation of pleural thickening observed on CT. J Nucl Med 2004;45: Benard F, Sterman D, Smith RJ, et al. Metabolic imaging of malignant pleural mesothelioma with fluorodeoxyglucose positron emission tomography. Chest 1998;114: Flores RM, Akhurst T, Gonen M, et al. Positron emission tomography defines metastatic disease but not locoregional disease in patients with malignant pleural mesothelioma. J Thorac Cardiovasc Surg 2003;126: Gerbaudo VH, Britz-Cunningham S, Sugarbaker DJ, et al. Metabolic significance of the pattern, intensity and kinetics of 18F-FDG uptake in malignant pleural mesothelioma. Thorax 2003;58: Benard F, Sterman D, Smith RJ, et al. Prognostic value of FDG PET imaging in malignant pleural mesothelioma. J Nucl Med 1999;40: Kwek BH, Aquino SL, Fischman AJ. Fluorodeoxyglucose positron emission tomography and CT after talc pleurodesis. Chest 2004;125: Gerbaudo VH, Sugarbaker DJ, Britz-Cunningham S, et al. Assessment of malignant pleural mesothelioma with 18F- FDG dual-head gamma-camera coincidence imaging: Comparison with histopathology. J Nucl Med 2002;43: Knuuttila A, Kivisaari L, Kivisaari A, et al. Evaluation of pleural disease using MR and CT with special reference to malignant pleural mesothelioma. Acta Radiol 2001;42: Hierholzer J, Luo L, Bittner RC, et al. MRI and CT in the differential diagnosis of pleural disease. Chest 2000;118: Stewart D, Waller D, Edwards J, et al. Is there a role for preoperative contrast-enhanced magnetic resonance imaging for radical surgery in malignant pleural mesothelioma? Eur J Cardiothorac Surg 2003;24: Rusch VW. A proposed new international TNM staging system for malignant pleural mesothelioma: From the International Mesothelioma Interest Group. Chest 1995;108: Rusch VW, Godwin JD, Shuman WP. The role of computed tomography scanning in the initial assessment and follow-up of malignant pleural mesothelioma. J Thorac Cardiovasc Surg 1988;96: Heelan RT, Rusch VW, Begg CB, et al. Staging of malignant pleural mesothelioma: Comparison of CT and MR imaging. AJR Am J Roentgenol 1999;172: Schouwink JH, Kool LS, Rutgers EJ, et al. The value of chest computer tomography and cervical mediastinoscopy in the preoperative assessment of patients with malignant pleural mesothelioma. Ann Thorac Surg 2003;75: Sugarbaker DJ, Flores RM, Kaklitsch MT, et al. Resection margins, extrapleural nodal status and cell type determine postoperative long-term survival in trimodality therapy of malignant pleural mesothelioma: Results in 183 patients. J Thorac Cardiovasc Surg 1999;117: Pilling J, Stewart D, Martin-Ucar A, et al. The case for routine mediastinoscopy prior to radical resection of malignant pleural mesothelioma. Chest 2003;124:S Treasure T, Waller D, Swift S, et al. Radical surgery for mesothelioma. BMJ 2004;328: Goodman LR. Imaging FTER thoracotomy. In: Goodman LR, Putman CE, editors. Critical care imaging. Philadelphia, PA: Saunders; p Butchart EG, Ashcroft T, Barnsley WC, et al. Pleuropneumonectomy in the management of diffuse malignant mesothelioma of the pleura. Experience in 29 patients. Thorax 1976; 31: WHO: Handbook for reporting results of cancer treatment. Geneva, Switzerland: World Health Organization; 1979 [Offset publication no. 48]. 53. Therasse P, Arbuck SG, Eisenhauer EA, et al. New guidelines to evaluate the response to treatment in solid tumours. J Natl Cancer Inst 2000;92: Monetti F, Casanova S, Grasso A, et al. Inadequacy of the new response evaluation criteria in solid tumours (RECIST) in patients with malignant pleural mesothelioma: Report of four cases. Lung Cancer 2004;43: Van Klaveren RJ, Aerts JGJV, De Bruin H, et al. Inadequacy of
11 Use of imaging in the management of malignant pleural mesothelioma 1247 the RECIST criteria for response evaluation in patients with malignant pleural mesothelioma. Lung Cancer 2004;43: Byrne MJ, Nowak AK. Modified RECIST criteria for assessment of response in malignant pleural mesothelioma. Ann Oncol 2004;15: Armato III SG, Oxnard GR, MacMahon H, et al. Measurement of mesothelioma on thoracic CT scans: A comparison of manual and computer-assisted techniques. Med Phys 2004; 31: Carretta A, Landoni C, Melloni G, et al. 18-FDG positron emission tomography in the evaluation of malignant pleural diseases a pilot study. Eur J Cardiothorac Surg 2002;17:
Objectives. Mylene T. Truong, MD. Malignant Pleural Mesothelioma Background
 Imaging of Pleural Tumors Mylene T. Truong, MD Imaging of Pleural Tumours Mylene T. Truong, M. D. University of Texas M.D. Anderson Cancer Center, Houston, TX Objectives To review tumors involving the
Imaging of Pleural Tumors Mylene T. Truong, MD Imaging of Pleural Tumours Mylene T. Truong, M. D. University of Texas M.D. Anderson Cancer Center, Houston, TX Objectives To review tumors involving the
Malignant Pleural Diseases Advances Clinicians Should Know F Gleeson
 Malignant Pleural Diseases Advances Clinicians Should Know F Gleeson The following relevant disclosures, conflicts of interest and/ or financial relationships exist related to this presentation: Consultant
Malignant Pleural Diseases Advances Clinicians Should Know F Gleeson The following relevant disclosures, conflicts of interest and/ or financial relationships exist related to this presentation: Consultant
Extrapleural Pneumonectomy for Malignant Mesothelioma: Pro. Joon H. Lee 9/17/2012
 Extrapleural Pneumonectomy for Malignant Mesothelioma: Pro Joon H. Lee 9/17/2012 Malignant Pleural Mesothelioma (Epidemiology) Incidence: 7/mil (Japan) to 40/mil (Australia) Attributed secondary to asbestos
Extrapleural Pneumonectomy for Malignant Mesothelioma: Pro Joon H. Lee 9/17/2012 Malignant Pleural Mesothelioma (Epidemiology) Incidence: 7/mil (Japan) to 40/mil (Australia) Attributed secondary to asbestos
The Proposed New International TNM Staging System for Malignant Pleural Mesothelioma: Application to Imaging
 323.....:. #{149} :. #{149}..: #{149}. #{149}. :- : Received June 22, 1995; accepted after revision September 12, 1995. 1 Department of Radiology, Duke University Medical Center, Box 3808, Dunham, NC 2771
323.....:. #{149} :. #{149}..: #{149}. #{149}. :- : Received June 22, 1995; accepted after revision September 12, 1995. 1 Department of Radiology, Duke University Medical Center, Box 3808, Dunham, NC 2771
PET POSITIVE PLEURAL PLAQUES DECADES AFTER PLEURODESIS: MESOLTHELIOMA? Ellen A. Middleton 1. Jonathan C. Daniel 2. Kenneth S.
 PET POSITIVE PLEURAL PLAQUES DECADES AFTER PLEURODESIS: MESOLTHELIOMA? Ellen A. Middleton 1 Jonathan C. Daniel 2 Kenneth S. Knox 1 Kathleen Williams 1 Departments of Medicine 1 and Surgery 2, University
PET POSITIVE PLEURAL PLAQUES DECADES AFTER PLEURODESIS: MESOLTHELIOMA? Ellen A. Middleton 1 Jonathan C. Daniel 2 Kenneth S. Knox 1 Kathleen Williams 1 Departments of Medicine 1 and Surgery 2, University
Radiologic Assessment of Mesothelioma
 28 Radiologic Assessment of Mesothelioma Samuel G. Armato III, Heber MacMahon, Geoffrey R. Oxnard, Charles L. Croteau, and Nicholas J. Vogelzang Imaging plays an essential role in the diagnosis, staging,
28 Radiologic Assessment of Mesothelioma Samuel G. Armato III, Heber MacMahon, Geoffrey R. Oxnard, Charles L. Croteau, and Nicholas J. Vogelzang Imaging plays an essential role in the diagnosis, staging,
Mesothelioma. 1. Introduction. 1.1 General Information and Aetiology
 Mesothelioma 1. Introduction 1.1 General Information and Aetiology Mesotheliomas are tumours that arise from the mesothelial cells of the pleura, peritoneum, pericardium or tunica vaginalis [1]. Most are
Mesothelioma 1. Introduction 1.1 General Information and Aetiology Mesotheliomas are tumours that arise from the mesothelial cells of the pleura, peritoneum, pericardium or tunica vaginalis [1]. Most are
Recommendations for cross-sectional imaging in cancer management, Second edition
 www.rcr.ac.uk Recommendations for cross-sectional imaging in cancer management, Second edition Breast cancer Faculty of Clinical Radiology www.rcr.ac.uk Contents Breast cancer 2 Clinical background 2 Who
www.rcr.ac.uk Recommendations for cross-sectional imaging in cancer management, Second edition Breast cancer Faculty of Clinical Radiology www.rcr.ac.uk Contents Breast cancer 2 Clinical background 2 Who
Malignant Mesothelioma Versus Metastatic Carcinoma of the Pleura: A CT Challenge
 Iran J Radiol. 2016 January; 13(1): e10949. Published online 2015 November 14. CHEST IMAGING DOI: 10.5812/iranjradiol.10949 Research Article Malignant Mesothelioma Versus Metastatic Carcinoma of the Pleura:
Iran J Radiol. 2016 January; 13(1): e10949. Published online 2015 November 14. CHEST IMAGING DOI: 10.5812/iranjradiol.10949 Research Article Malignant Mesothelioma Versus Metastatic Carcinoma of the Pleura:
PET/CT in Lung Cancer
 PET/CT in Lung Cancer Rodolfo Núñez Miller, M.D. Nuclear Medicine and Diagnostic Imaging Section Division of Human Health International Atomic Energy Agency Vienna, Austria GLOBOCAN 2012 #1 #3 FDG-PET/CT
PET/CT in Lung Cancer Rodolfo Núñez Miller, M.D. Nuclear Medicine and Diagnostic Imaging Section Division of Human Health International Atomic Energy Agency Vienna, Austria GLOBOCAN 2012 #1 #3 FDG-PET/CT
What is the best way to diagnose and stage malignant pleural mesothelioma?
 doi:10.1510/icvts.2010.255893 Interactive CardioVascular and Thoracic Surgery 12 (2011) 254 259 Best evidence topic - Thoracic oncologic What is the best way to diagnose and stage malignant pleural mesothelioma?
doi:10.1510/icvts.2010.255893 Interactive CardioVascular and Thoracic Surgery 12 (2011) 254 259 Best evidence topic - Thoracic oncologic What is the best way to diagnose and stage malignant pleural mesothelioma?
Malignant pleural mesothelioma is a rare primary neoplasm,
 Assessment of Malignant Pleural Mesothelioma with F-FDG Dual-Head Gamma-Camera Coincidence Imaging: Comparison with Histopathology Victor H. Gerbaudo, PhD 1 ; David J. Sugarbaker, MD 2 ; Scott Britz-Cunningham,
Assessment of Malignant Pleural Mesothelioma with F-FDG Dual-Head Gamma-Camera Coincidence Imaging: Comparison with Histopathology Victor H. Gerbaudo, PhD 1 ; David J. Sugarbaker, MD 2 ; Scott Britz-Cunningham,
ARTICLE IN PRESS. European Journal of Radiology xxx (2009) xxx xxx. Contents lists available at ScienceDirect. European Journal of Radiology
 European Journal of Radiology xxx (2009) xxx xxx Contents lists available at ScienceDirect European Journal of Radiology journal homepage: www.elsevier.com/locate/ejrad Three-dimensional evaluation of
European Journal of Radiology xxx (2009) xxx xxx Contents lists available at ScienceDirect European Journal of Radiology journal homepage: www.elsevier.com/locate/ejrad Three-dimensional evaluation of
Sternotomy and removal of the tumor
 Sternotomy and removal of the tumor All thymomas originate from epithelial thymic cells 4% of them consist of a pure population of epithelial cells Most have mixed populations of lymphoid cells to a
Sternotomy and removal of the tumor All thymomas originate from epithelial thymic cells 4% of them consist of a pure population of epithelial cells Most have mixed populations of lymphoid cells to a
International Journal of Case Reports in Medicine
 International Journal of Case Reports in Medicine Vol. 2013 (2013), Article ID 409830, 15 minipages. DOI:10.5171/2013.409830 www.ibimapublishing.com Copyright 2013 Andrew Thomas Low, Iain Smith and Simon
International Journal of Case Reports in Medicine Vol. 2013 (2013), Article ID 409830, 15 minipages. DOI:10.5171/2013.409830 www.ibimapublishing.com Copyright 2013 Andrew Thomas Low, Iain Smith and Simon
Recommendations for the Reporting of Pleural Mesothelioma
 Recommendations for the Reporting of Pleural Mesothelioma Association of Directors of Anatomic and Surgical Pathology * DOI: 10.1309/6A30YQHBMTHEJTEM It has been evident for decades that pathology reports
Recommendations for the Reporting of Pleural Mesothelioma Association of Directors of Anatomic and Surgical Pathology * DOI: 10.1309/6A30YQHBMTHEJTEM It has been evident for decades that pathology reports
Clinical cases in Malignant Pleural Mesothelioma: Adherence to the ESMO Clinical Practice Guidelines
 Clinical cases in Malignant Pleural Mesothelioma: Adherence to the ESMO Clinical Practice Guidelines Wieneke Buikhuisen The Netherlands Cancer Institute Amsterdam The Netherlands Case (1) Male, 56 year
Clinical cases in Malignant Pleural Mesothelioma: Adherence to the ESMO Clinical Practice Guidelines Wieneke Buikhuisen The Netherlands Cancer Institute Amsterdam The Netherlands Case (1) Male, 56 year
Malignant Mesothelioma Current Approaches to a Difficult Problem. Raja M Flores, MD Thoracic Surgery Memorial Sloan-Kettering Cancer Center
 Malignant Mesothelioma Current Approaches to a Difficult Problem Raja M Flores, MD Thoracic Surgery Memorial Sloan-Kettering Cancer Center Malignant Pleural Mesothelioma Clinical Presentation Insidious
Malignant Mesothelioma Current Approaches to a Difficult Problem Raja M Flores, MD Thoracic Surgery Memorial Sloan-Kettering Cancer Center Malignant Pleural Mesothelioma Clinical Presentation Insidious
بسم هللا الرحمن الرحيم
 بسم هللا الرحمن الرحيم Updates in Mesothelioma By Samieh Amer, MD Professor of Cardiothoracic Surgery Faculty of Medicine, Cairo University History Wagner and his colleagues (1960) 33 cases of mesothelioma
بسم هللا الرحمن الرحيم Updates in Mesothelioma By Samieh Amer, MD Professor of Cardiothoracic Surgery Faculty of Medicine, Cairo University History Wagner and his colleagues (1960) 33 cases of mesothelioma
Primary -Benign - Malignant Secondary
 TUMOURS OF THE LUNG Primary -Benign - Malignant Secondary The incidence of lung cancer has been increasing almost logarithmically and is now reaching epidemic levels. The overall cure rate is very low
TUMOURS OF THE LUNG Primary -Benign - Malignant Secondary The incidence of lung cancer has been increasing almost logarithmically and is now reaching epidemic levels. The overall cure rate is very low
INTERNATIONAL ASSOCIATION FOR THE STUDY OF LUNG CANCER Prospective Mesothelioma Staging Project
 INTERNATIONAL ASSOCIATION FOR THE STUDY OF LUNG CANCER Prospective Mesothelioma Staging Project Data Forms and Fields in CRAB Electronic Data Capture System - Reduced Set - Pivotal data elements for developing
INTERNATIONAL ASSOCIATION FOR THE STUDY OF LUNG CANCER Prospective Mesothelioma Staging Project Data Forms and Fields in CRAB Electronic Data Capture System - Reduced Set - Pivotal data elements for developing
Disease/Illness GUIDE TO ASBESTOS LUNG CANCER. What Is Asbestos Lung Cancer? www.simpsonmillar.co.uk Telephone 0844 858 3200
 GUIDE TO ASBESTOS LUNG CANCER What Is Asbestos Lung Cancer? Like tobacco smoking, exposure to asbestos can result in the development of lung cancer. Similarly, the risk of developing asbestos induced lung
GUIDE TO ASBESTOS LUNG CANCER What Is Asbestos Lung Cancer? Like tobacco smoking, exposure to asbestos can result in the development of lung cancer. Similarly, the risk of developing asbestos induced lung
MALIGNANT PLEURAL MESOTHELIOMA TREATED IN CLINIC FOR PULMONARY DISEASES AND TUBERCULOSIS PODHRASTOVI IN TEN-YEAR PERIOD ( FROM 1998 TO 2007 )
 & MALIGNANT PLEURAL MESOTHELIOMA TREATED IN CLINIC FOR PULMONARY DISEASES AND TUBERCULOSIS PODHRASTOVI IN TEN-YEAR PERIOD ( FROM 1998 TO 2007 ) Vesna Čukić*, Aida Ustamujić, Vladimir Lovre, Hasan Žutić,
& MALIGNANT PLEURAL MESOTHELIOMA TREATED IN CLINIC FOR PULMONARY DISEASES AND TUBERCULOSIS PODHRASTOVI IN TEN-YEAR PERIOD ( FROM 1998 TO 2007 ) Vesna Čukić*, Aida Ustamujić, Vladimir Lovre, Hasan Žutić,
Introduction. Case Report
 DOI: 10.4046/trd.2009.67.4.369 ISSN: 1738-3536(Print)/2005-6184(Online) Tuberc Respir Dis 2009;67:369-373 CopyrightC2009. The Korean Academy of Tuberculosis and Respiratory Diseases. All rights reserved.
DOI: 10.4046/trd.2009.67.4.369 ISSN: 1738-3536(Print)/2005-6184(Online) Tuberc Respir Dis 2009;67:369-373 CopyrightC2009. The Korean Academy of Tuberculosis and Respiratory Diseases. All rights reserved.
Mesothelioma. Malignant Pleural Mesothelioma
 Mesothelioma William G. Richards, PhD Brigham and Women s Hospital Malignant Pleural Mesothelioma 2,000-3,000 cases per year (USA) Increasing incidence Asbestos (50-80%, decreasing) 30-40 year latency
Mesothelioma William G. Richards, PhD Brigham and Women s Hospital Malignant Pleural Mesothelioma 2,000-3,000 cases per year (USA) Increasing incidence Asbestos (50-80%, decreasing) 30-40 year latency
Accelerated hemithoracic radiation followed by extrapleural pneumonectomy for malignant pleural mesothelioma
 Accelerated hemithoracic radiation followed by extrapleural pneumonectomy for malignant pleural mesothelioma Marc de Perrot, Ronald Feld, Natasha B Leighl, Andrew Hope, Thomas K Waddell, Shaf Keshavjee,
Accelerated hemithoracic radiation followed by extrapleural pneumonectomy for malignant pleural mesothelioma Marc de Perrot, Ronald Feld, Natasha B Leighl, Andrew Hope, Thomas K Waddell, Shaf Keshavjee,
Diagnostic Imaging and workup of Malignant Pleural Mesothelioma.
 Diagnostic Imaging and workup of Malignant Pleural Mesothelioma. A pictorial essay Luciano Cardinale, Francesco Ardissone*, Dario Gned, Nicola Sverzellati**, Edoardo Piacibello, Andrea Veltri Department
Diagnostic Imaging and workup of Malignant Pleural Mesothelioma. A pictorial essay Luciano Cardinale, Francesco Ardissone*, Dario Gned, Nicola Sverzellati**, Edoardo Piacibello, Andrea Veltri Department
The Need for Accurate Lung Cancer Staging
 The Need for Accurate Lung Cancer Staging Peter Baik, DO Thoracic Surgery Cancer Treatment Centers of America Oklahoma Osteopathic Association 115th Annual Convention Financial Disclosures: None 2 Objectives
The Need for Accurate Lung Cancer Staging Peter Baik, DO Thoracic Surgery Cancer Treatment Centers of America Oklahoma Osteopathic Association 115th Annual Convention Financial Disclosures: None 2 Objectives
Plueral Malignancy: Radiologic-pathologic
 Plueral Malignancy: Radiologic-pathologic Correlation Ritu R. Gill, MD Pleural Malignancies: Radiologic-Pathologic Correlation Ritu R Gill MD Brigham and Women s Hospital Boston, Massachusetts Pleural
Plueral Malignancy: Radiologic-pathologic Correlation Ritu R. Gill, MD Pleural Malignancies: Radiologic-Pathologic Correlation Ritu R Gill MD Brigham and Women s Hospital Boston, Massachusetts Pleural
Malignant Pleural Mesothelioma in Singapore
 RESEARCH COMMUNICATION C SP Yip 1, HN Koong 2, CM Loo 3, KW Fong 1* Abstract Aim: To examine the clinical characteristics and outcomes of malignant pleural mesothelioma (MPM) in Singapore. Methods and
RESEARCH COMMUNICATION C SP Yip 1, HN Koong 2, CM Loo 3, KW Fong 1* Abstract Aim: To examine the clinical characteristics and outcomes of malignant pleural mesothelioma (MPM) in Singapore. Methods and
GUIDELINES FOR THE MANAGEMENT OF LUNG CANCER
 GUIDELINES FOR THE MANAGEMENT OF LUNG CANCER BY Ali Shamseddine, MD (Coordinator); as04@aub.edu.lb Fady Geara, MD Bassem Shabb, MD Ghassan Jamaleddine, MD CLINICAL PRACTICE GUIDELINES FOR THE TREATMENT
GUIDELINES FOR THE MANAGEMENT OF LUNG CANCER BY Ali Shamseddine, MD (Coordinator); as04@aub.edu.lb Fady Geara, MD Bassem Shabb, MD Ghassan Jamaleddine, MD CLINICAL PRACTICE GUIDELINES FOR THE TREATMENT
Mesothelioma: Questions and Answers
 CANCER FACTS N a t i o n a l C a n c e r I n s t i t u t e N a t i o n a l I n s t i t u t e s o f H e a l t h D e p a r t m e n t o f H e a l t h a n d H u m a n S e r v i c e s Mesothelioma: Questions
CANCER FACTS N a t i o n a l C a n c e r I n s t i t u t e N a t i o n a l I n s t i t u t e s o f H e a l t h D e p a r t m e n t o f H e a l t h a n d H u m a n S e r v i c e s Mesothelioma: Questions
Treatment of mesothelioma in Bloemfontein, South Africa
 European Journal of Cardio-thoracic Surgery 24 (2003) 434 440 www.elsevier.com/locate/ejcts Treatment of mesothelioma in Bloemfontein, South Africa W.J. de Vries*, M.A. Long Cardiothoracic Department,
European Journal of Cardio-thoracic Surgery 24 (2003) 434 440 www.elsevier.com/locate/ejcts Treatment of mesothelioma in Bloemfontein, South Africa W.J. de Vries*, M.A. Long Cardiothoracic Department,
Thoracic 18F-FDG PETCT
 Thoracic 18F-FDG PETCT RAD Magazine, 41, 482, 13-16 Dr Allanah arker Specialist registrar radiology Dr Nagmi Qureshi Consultant cardiothoracic radiologist Papworth Hospital, Cambridge email: allanahbarker@nhs.net
Thoracic 18F-FDG PETCT RAD Magazine, 41, 482, 13-16 Dr Allanah arker Specialist registrar radiology Dr Nagmi Qureshi Consultant cardiothoracic radiologist Papworth Hospital, Cambridge email: allanahbarker@nhs.net
False positive PET in lymphoma
 False positive PET in lymphoma Thomas Krause Introduction and conclusion 2 3 Introduction 4 FDG-PET in staging of lymphoma 34 studies with 2227 Patients CT FDG-PET Sensitivity 63 % 89 % (58%-100%) (63%-100%)
False positive PET in lymphoma Thomas Krause Introduction and conclusion 2 3 Introduction 4 FDG-PET in staging of lymphoma 34 studies with 2227 Patients CT FDG-PET Sensitivity 63 % 89 % (58%-100%) (63%-100%)
Most cases of mesothelioma are caused by occupational
 Percutaneous Image-Guided Cutting Needle Biopsy of the Pleura in the Diagnosis of Malignant Mesothelioma* Rosie F. Adams, BM BCh; Winifred Gray, MB BS; Robert J. O. Davies, DM; and Fergus V. Gleeson, MB
Percutaneous Image-Guided Cutting Needle Biopsy of the Pleura in the Diagnosis of Malignant Mesothelioma* Rosie F. Adams, BM BCh; Winifred Gray, MB BS; Robert J. O. Davies, DM; and Fergus V. Gleeson, MB
A Practical Guide to Advances in Staging and Treatment of NSCLC
 A Practical Guide to Advances in Staging and Treatment of NSCLC Robert J. Korst, M.D. Director, Thoracic Surgery Medical Director, The Blumenthal Cancer Center The Valley Hospital Objectives Revised staging
A Practical Guide to Advances in Staging and Treatment of NSCLC Robert J. Korst, M.D. Director, Thoracic Surgery Medical Director, The Blumenthal Cancer Center The Valley Hospital Objectives Revised staging
Malignant Mesothelioma
 Malignant Malignant mesothelioma is a tumour originating from mesothelial cells. 85 95% of mesotheliomas are caused by asbestos exposure. It occurs much more commonly in the chest (malignant pleural mesothelioma)
Malignant Malignant mesothelioma is a tumour originating from mesothelial cells. 85 95% of mesotheliomas are caused by asbestos exposure. It occurs much more commonly in the chest (malignant pleural mesothelioma)
Malignant Mesothelioma
 Malignant mesothelioma is a tumour originating from mesothelial cells. 85 95% of mesotheliomas are caused by asbestos exposure. It occurs much more commonly in the chest (malignant pleural mesothelioma)
Malignant mesothelioma is a tumour originating from mesothelial cells. 85 95% of mesotheliomas are caused by asbestos exposure. It occurs much more commonly in the chest (malignant pleural mesothelioma)
Detection and staging of recurrent prostate cancer is still one of the important clinical problems in prostate cancer. A rise in PSA or biochemical
 Summary. 111 Detection and staging of recurrent prostate cancer is still one of the important clinical problems in prostate cancer. A rise in PSA or biochemical recurrence (BCR) is the first sign of recurrent
Summary. 111 Detection and staging of recurrent prostate cancer is still one of the important clinical problems in prostate cancer. A rise in PSA or biochemical recurrence (BCR) is the first sign of recurrent
Malignant pleural mesothelioma: outcome of limited surgical management
 Interactive Cardiovascular and Thoracic Surgery 2 (2003) 30 34 Institutional review Thoracic general Malignant pleural mesothelioma: outcome of limited surgical management Peter G. Phillips a, George Asimakopoulos
Interactive Cardiovascular and Thoracic Surgery 2 (2003) 30 34 Institutional review Thoracic general Malignant pleural mesothelioma: outcome of limited surgical management Peter G. Phillips a, George Asimakopoulos
Morphologic and functional imaging of malignant pleural mesothelioma
 European Journal of Radiology 64 (2007) 356 366 Review Morphologic and functional imaging of malignant pleural mesothelioma Masaki Yamamuro, Victor H. Gerbaudo, Ritu R. Gill, Francine L. Jacobson, David
European Journal of Radiology 64 (2007) 356 366 Review Morphologic and functional imaging of malignant pleural mesothelioma Masaki Yamamuro, Victor H. Gerbaudo, Ritu R. Gill, Francine L. Jacobson, David
Mesothelioma. 1995-2013, The Patient Education Institute, Inc. www.x-plain.com ocft0101 Last reviewed: 03/21/2013 1
 Mesothelioma Introduction Mesothelioma is a type of cancer. It starts in the tissue that lines your lungs, stomach, heart, and other organs. This tissue is called mesothelium. Most people who get this
Mesothelioma Introduction Mesothelioma is a type of cancer. It starts in the tissue that lines your lungs, stomach, heart, and other organs. This tissue is called mesothelium. Most people who get this
L Lang-Lazdunski, A Bille, S Marshall, R Lal, D Landau, J Spicer
 Pleurectomy/decortication, hyperthermic pleural lavage with povidone-iodine and systemic chemotherapy in malignant pleural mesothelioma. A 10-year experience. L Lang-Lazdunski, A Bille, S Marshall, R Lal,
Pleurectomy/decortication, hyperthermic pleural lavage with povidone-iodine and systemic chemotherapy in malignant pleural mesothelioma. A 10-year experience. L Lang-Lazdunski, A Bille, S Marshall, R Lal,
Assessment of 18 F-fluorodeoxyglucose dual-head gamma camera in asbestos lung diseases
 Eur Respir J 2004; 24: 814 821 DOI: 10.1183/09031936.04.00004504 Printed in UK all rights reserved Copyright #ERS Journals Ltd 2004 European Respiratory Journal ISSN 0903-1936 Assessment of 18 F-fluorodeoxyglucose
Eur Respir J 2004; 24: 814 821 DOI: 10.1183/09031936.04.00004504 Printed in UK all rights reserved Copyright #ERS Journals Ltd 2004 European Respiratory Journal ISSN 0903-1936 Assessment of 18 F-fluorodeoxyglucose
What is Mesothelioma?
 What is Mesothelioma? Mesothelioma is a rare type of cancer that develops in the mesothelial cells found in one s body. These cells form membranous linings that surround and protect the body s organs and
What is Mesothelioma? Mesothelioma is a rare type of cancer that develops in the mesothelial cells found in one s body. These cells form membranous linings that surround and protect the body s organs and
Diseases. Inflammations Non-inflammatory pleural effusions Pneumothorax Tumours
 Pleura Visceral pleura covers lungs and extends into fissures Parietal pleura limits mediastinum and covers dome of diaphragm and inner aspect of chest wall. Two layers between them (pleural cavity) contains
Pleura Visceral pleura covers lungs and extends into fissures Parietal pleura limits mediastinum and covers dome of diaphragm and inner aspect of chest wall. Two layers between them (pleural cavity) contains
SMALL CELL LUNG CANCER
 Protocol for Planning and Treatment The process to be followed in the management of: SMALL CELL LUNG CANCER Patient information given at each stage following agreed information pathway 1. DIAGNOSIS New
Protocol for Planning and Treatment The process to be followed in the management of: SMALL CELL LUNG CANCER Patient information given at each stage following agreed information pathway 1. DIAGNOSIS New
Case Report A Cause of Bilateral Chylothorax: A Case of Mesothelioma without Pleural Involvement during Initial Diagnosis
 Case Reports in Pulmonology Volume 2015, Article ID 962504, 4 pages http://dx.doi.org/10.1155/2015/962504 Case Report A Cause of Bilateral Chylothorax: A Case of Mesothelioma without Pleural Involvement
Case Reports in Pulmonology Volume 2015, Article ID 962504, 4 pages http://dx.doi.org/10.1155/2015/962504 Case Report A Cause of Bilateral Chylothorax: A Case of Mesothelioma without Pleural Involvement
Patterns of nodal spread in thoracic malignancies
 Patterns of nodal spread in thoracic malignancies Poster No.: C-0977 Congress: ECR 2010 Type: Educational Exhibit Topic: Chest Authors: R. dos Santos, M. Duarte, J. Alpendre, J. Castaño, Z. Seabra, Â.
Patterns of nodal spread in thoracic malignancies Poster No.: C-0977 Congress: ECR 2010 Type: Educational Exhibit Topic: Chest Authors: R. dos Santos, M. Duarte, J. Alpendre, J. Castaño, Z. Seabra, Â.
ASBESTOS EXPOSURE AND SARCOMATOID MALIGNANT PLEURAL MESOTHELIOMA Gorantla Sambasivarao 1, Namballa Usharani 2, Tupakula Suresh Babu 3
 ASBESTOS EXPOSURE AND SARCOMATOID MALIGNANT PLEURAL MESOTHELIOMA Gorantla Sambasivarao 1, Namballa Usharani 2, Tupakula Suresh Babu 3 HOW TO CITE THIS ARTICLE: Gorantla Sambasivarao, Namballa Usharani,
ASBESTOS EXPOSURE AND SARCOMATOID MALIGNANT PLEURAL MESOTHELIOMA Gorantla Sambasivarao 1, Namballa Usharani 2, Tupakula Suresh Babu 3 HOW TO CITE THIS ARTICLE: Gorantla Sambasivarao, Namballa Usharani,
Proposed Adjustments to Pathologic Staging of Epithelial Malignant Pleural Mesothelioma Based on Analysis of 354 Cases
 Original Article Proposed Adjustments to Pathologic Staging of Epithelial Malignant Pleural Mesothelioma Based on Analysis of 354 Cases William G. Richards, PhD 1,2 ; John J. Godleski, MD 2,3 ; Beow Y.
Original Article Proposed Adjustments to Pathologic Staging of Epithelial Malignant Pleural Mesothelioma Based on Analysis of 354 Cases William G. Richards, PhD 1,2 ; John J. Godleski, MD 2,3 ; Beow Y.
Update on Mesothelioma
 November 8, 2012 Update on Mesothelioma Intro incidence and nomenclature Update on Classification Diagnostic specimens Morphologic features Epithelioid Histology Biphasic Histology Immunohistochemical
November 8, 2012 Update on Mesothelioma Intro incidence and nomenclature Update on Classification Diagnostic specimens Morphologic features Epithelioid Histology Biphasic Histology Immunohistochemical
Malignant Pleural Mesothelioma Caused by Environmental Exposure to Asbestos in the Southeast of Turkey: CT Findings in 117 Patients
 Clinical Investigations Respiration 2000;67:615 622 Received: August 6, 1999 Accepted after revision: June 27, 2000 Malignant Pleural Mesothelioma Caused by Environmental Exposure to Asbestos in the Southeast
Clinical Investigations Respiration 2000;67:615 622 Received: August 6, 1999 Accepted after revision: June 27, 2000 Malignant Pleural Mesothelioma Caused by Environmental Exposure to Asbestos in the Southeast
Research Article Frequency of Surgery in Black Patients with Malignant Pleural Mesothelioma
 Disease Markers Volume 2015, Article ID 282145, 5 pages http://dx.doi.org/10.1155/2015/282145 Research Article Frequency of Surgery in Black Patients with Malignant Pleural Mesothelioma Emanuela Taioli,
Disease Markers Volume 2015, Article ID 282145, 5 pages http://dx.doi.org/10.1155/2015/282145 Research Article Frequency of Surgery in Black Patients with Malignant Pleural Mesothelioma Emanuela Taioli,
Metastatic Renal Cell Carcinoma: Staging and Prognosis of Three Separate Cases.
 Metastatic Renal Cell Carcinoma: Staging and Prognosis of Three Separate Cases. Abstract This paper describes the staging, imaging, treatment, and prognosis of renal cell carcinoma. Three case studies
Metastatic Renal Cell Carcinoma: Staging and Prognosis of Three Separate Cases. Abstract This paper describes the staging, imaging, treatment, and prognosis of renal cell carcinoma. Three case studies
Case Report: Whole-body Oncologic Imaging with syngo TimCT
 Case Report: Whole-body Oncologic Imaging with syngo TimCT Eric Hatfield, M.D. 1 ; Agus Priatna, Ph.D. 2 ; John Kotyk, Ph.D. 1 ; Benjamin Tan, M.D. 1 ; Alto Stemmer 3 ; Stephan Kannengiesser, Ph.D. 3 ;
Case Report: Whole-body Oncologic Imaging with syngo TimCT Eric Hatfield, M.D. 1 ; Agus Priatna, Ph.D. 2 ; John Kotyk, Ph.D. 1 ; Benjamin Tan, M.D. 1 ; Alto Stemmer 3 ; Stephan Kannengiesser, Ph.D. 3 ;
Small Cell Lung Cancer
 Small Cell Lung Cancer Types of Lung Cancer Non-small cell carcinoma (NSCC) (87%) Adenocarcinoma (38%) Squamous cell (20%) Large cell (5%) Small cell carcinoma (13%) Small cell lung cancer is virtually
Small Cell Lung Cancer Types of Lung Cancer Non-small cell carcinoma (NSCC) (87%) Adenocarcinoma (38%) Squamous cell (20%) Large cell (5%) Small cell carcinoma (13%) Small cell lung cancer is virtually
Malignant Mesothelioma State of the Art
 Malignant Mesothelioma State of the Art Paul Baas The Netherlands Cancer Institute August 12, 2011, Carlsbad, CA Summary Diagnosis; epithelial type subdivided Pleiomorphic vs other Staging: IASLC-IMIG
Malignant Mesothelioma State of the Art Paul Baas The Netherlands Cancer Institute August 12, 2011, Carlsbad, CA Summary Diagnosis; epithelial type subdivided Pleiomorphic vs other Staging: IASLC-IMIG
Imaging of pleural and chest wall tumors
 Thorac Surg Clin 14 (2004) 15 23 Imaging of pleural and chest wall tumors Michael J. Weyant, MD a, Raja M. Flores, MD a,b, * a Cardiothoracic Surgery, Memorial Sloan-Kettering Cancer Center, 1275 York
Thorac Surg Clin 14 (2004) 15 23 Imaging of pleural and chest wall tumors Michael J. Weyant, MD a, Raja M. Flores, MD a,b, * a Cardiothoracic Surgery, Memorial Sloan-Kettering Cancer Center, 1275 York
Understanding Pleural Mesothelioma
 Understanding Pleural Mesothelioma UHN Information for patients and families Read this booklet to learn about: What is pleural mesothelioma? What causes it? What are the symptoms? What tests are done to
Understanding Pleural Mesothelioma UHN Information for patients and families Read this booklet to learn about: What is pleural mesothelioma? What causes it? What are the symptoms? What tests are done to
How to measure effect of treatment? chest radiography CT scan MRI scan PET scan ultrasound physical examination bone scintygraphy
 respons meting Bonne Biesma Franz Schramel Terschelling 2012 How to measure effect of treatment chest radiography CT scan MRI scan PET scan ultrasound physical examination bone scintygraphy guidelines
respons meting Bonne Biesma Franz Schramel Terschelling 2012 How to measure effect of treatment chest radiography CT scan MRI scan PET scan ultrasound physical examination bone scintygraphy guidelines
Screening, early referral and treatment for asbestos related cancer
 Screening, early referral and treatment for asbestos related cancer Marc de Perrot, MD, MSc, FRCSC Toronto Mesothelioma Research Program University of Toronto Asbestos related diseases Mesothelioma Lung
Screening, early referral and treatment for asbestos related cancer Marc de Perrot, MD, MSc, FRCSC Toronto Mesothelioma Research Program University of Toronto Asbestos related diseases Mesothelioma Lung
Evaluation of asbestos-exposure, clinical diagnosis and treatment of MPM
 Evaluation of asbestos-exposure, clinical diagnosis and treatment of MPM Aija Knuuttila, MD, PhD Dept. of Pulmonary Medicine Helsinki University Central Hospital, Finland 15.11.2007 Malignant pleural mesothelioma
Evaluation of asbestos-exposure, clinical diagnosis and treatment of MPM Aija Knuuttila, MD, PhD Dept. of Pulmonary Medicine Helsinki University Central Hospital, Finland 15.11.2007 Malignant pleural mesothelioma
How To Understand How Cancer Works
 Mesothelioma Understanding your diagnosis Mesothelioma Understanding your diagnosis When you first hear that you have cancer, you may feel alone and afraid. You may be overwhelmed by the large amount of
Mesothelioma Understanding your diagnosis Mesothelioma Understanding your diagnosis When you first hear that you have cancer, you may feel alone and afraid. You may be overwhelmed by the large amount of
Discussion. AJR:159, November1992 MALIGNANT PLEURAL MESOTHELIOMA 965
 AJR:159, November1992 MALIGNANT PLEURAL MESOTHELIOMA 965 Fig. 7.-45-year-old woman with unresectable malignant mesothelioma. CT scan shows lobular tumor rind with direct extension into azygoesophageal
AJR:159, November1992 MALIGNANT PLEURAL MESOTHELIOMA 965 Fig. 7.-45-year-old woman with unresectable malignant mesothelioma. CT scan shows lobular tumor rind with direct extension into azygoesophageal
General Information About Non-Small Cell Lung Cancer
 General Information About Non-Small Cell Lung Cancer Non-small cell lung cancer is a disease in which malignant (cancer) cells form in the tissues of the lung. The lungs are a pair of cone-shaped breathing
General Information About Non-Small Cell Lung Cancer Non-small cell lung cancer is a disease in which malignant (cancer) cells form in the tissues of the lung. The lungs are a pair of cone-shaped breathing
9. Discuss guidelines for follow-up post-thyroidectomy for cancer (labs/tests) HH
 9. Discuss guidelines for follow-up post-thyroidectomy for cancer (labs/tests) HH Differentiated thyroid cancer expresses the TSH receptor on the cell membrane and responds to TSH stimulation by increasing
9. Discuss guidelines for follow-up post-thyroidectomy for cancer (labs/tests) HH Differentiated thyroid cancer expresses the TSH receptor on the cell membrane and responds to TSH stimulation by increasing
Surgeons Role in Symptom Management. A/Prof Cliff K. C. Choong Consultant Thoracic Surgeon Latrobe Regional Hospital GIPPSLAND
 Surgeons Role in Symptom Management A/Prof Cliff K. C. Choong Consultant Thoracic Surgeon Latrobe Regional Hospital GIPPSLAND Conditions PLEURAL Pleural effusion Pneumothorax ENDOBRONCHIAL Haemoptysis
Surgeons Role in Symptom Management A/Prof Cliff K. C. Choong Consultant Thoracic Surgeon Latrobe Regional Hospital GIPPSLAND Conditions PLEURAL Pleural effusion Pneumothorax ENDOBRONCHIAL Haemoptysis
Asbestos and your lungs
 This information describes what asbestos is and the lung conditions that are caused by exposure to it. It also includes information about what to do if you have been exposed to asbestos, and the benefits
This information describes what asbestos is and the lung conditions that are caused by exposure to it. It also includes information about what to do if you have been exposed to asbestos, and the benefits
Lung cancer forms in tissues of the lung, usually in the cells lining air passages.
 Scan for mobile link. Lung Cancer Lung cancer usually forms in the tissue cells lining the air passages within the lungs. The two main types are small-cell lung cancer (usually found in cigarette smokers)
Scan for mobile link. Lung Cancer Lung cancer usually forms in the tissue cells lining the air passages within the lungs. The two main types are small-cell lung cancer (usually found in cigarette smokers)
Round Table with Drs. Anne Tsao and Alex Farivar, Case 2: Mesothelioma
 Round Table with Drs. Anne Tsao and Alex Farivar, Case 2: Mesothelioma I d like to welcome everyone, thanks for coming out to our lunch with experts. The faculty today are great people in the thoracic
Round Table with Drs. Anne Tsao and Alex Farivar, Case 2: Mesothelioma I d like to welcome everyone, thanks for coming out to our lunch with experts. The faculty today are great people in the thoracic
PRINCESS MARGARET CANCER CENTRE CLINICAL PRACTICE GUIDELINES
 PRINCESS MARGARET CANCER CENTRE CLINICAL PRACTICE GUIDELINES LUNG SITE MESOTHELIOMA Lung Site Group Mesothelioma Date Guideline Created: April 2013 Authors: Dr. Meredith Giuliani, Dr. Andrea Bezjak 1.
PRINCESS MARGARET CANCER CENTRE CLINICAL PRACTICE GUIDELINES LUNG SITE MESOTHELIOMA Lung Site Group Mesothelioma Date Guideline Created: April 2013 Authors: Dr. Meredith Giuliani, Dr. Andrea Bezjak 1.
Neoplasms of the LUNG and PLEURA
 Neoplasms of the LUNG and PLEURA 2015-2016 FCDS Educational Webcast Series Steven Peace, BS, CTR September 19, 2015 2015 Focus o Anatomy o SSS 2000 o MPH Rules o AJCC TNM 1 Case 1 Case Vignette HISTORY:
Neoplasms of the LUNG and PLEURA 2015-2016 FCDS Educational Webcast Series Steven Peace, BS, CTR September 19, 2015 2015 Focus o Anatomy o SSS 2000 o MPH Rules o AJCC TNM 1 Case 1 Case Vignette HISTORY:
OBJECTIVES By the end of this segment, the community participant will be able to:
 Cancer 101: Cancer Diagnosis and Staging Linda U. Krebs, RN, PhD, AOCN, FAAN OCEAN Native Navigators and the Cancer Continuum (NNACC) (NCMHD R24MD002811) Cancer 101: Diagnosis & Staging (Watanabe-Galloway
Cancer 101: Cancer Diagnosis and Staging Linda U. Krebs, RN, PhD, AOCN, FAAN OCEAN Native Navigators and the Cancer Continuum (NNACC) (NCMHD R24MD002811) Cancer 101: Diagnosis & Staging (Watanabe-Galloway
Surgical therapy of. who should be operated
 SAMO Interdisciplinary Workshop on Chest Tumors Lucerne, 13th and 14th January 2012 Surgical therapy of mesothelioma, who should be operated Walter Weder MD Professor of Surgery University Hospital Zurich
SAMO Interdisciplinary Workshop on Chest Tumors Lucerne, 13th and 14th January 2012 Surgical therapy of mesothelioma, who should be operated Walter Weder MD Professor of Surgery University Hospital Zurich
Cardiac Masses and Tumors
 Cardiac Masses and Tumors Question: What is the diagnosis? A. Aortic valve myxoma B. Papillary fibroelastoma C. Vegetation from Infective endocarditis D. Thrombus in transit E. None of the above Answer:
Cardiac Masses and Tumors Question: What is the diagnosis? A. Aortic valve myxoma B. Papillary fibroelastoma C. Vegetation from Infective endocarditis D. Thrombus in transit E. None of the above Answer:
In Practice Whole Body MR for Visualizing Metastatic Prostate Cancer
 In Practice Whole Body MR for Visualizing Metastatic Prostate Cancer Prostate cancer is the second most common cancer in men worldwide, accounting for 15% of all new cancer cases. 1 Great strides have
In Practice Whole Body MR for Visualizing Metastatic Prostate Cancer Prostate cancer is the second most common cancer in men worldwide, accounting for 15% of all new cancer cases. 1 Great strides have
ASBESTOS MANAGEMENT REVIEW A CRITICAL REVIEW OF MEDICAL MANAGEMENT IN MALIGNANT PLEURAL MESOTHELIOMA - AUSTRALIA, 2011
 ASBESTOS MANAGEMENT REVIEW A CRITICAL REVIEW OF MEDICAL MANAGEMENT IN MALIGNANT PLEURAL MESOTHELIOMA - AUSTRALIA, 2011 Dr Malcolm Feigen Senior Radiation Oncologist Radiation Oncology Centre Austin Health
ASBESTOS MANAGEMENT REVIEW A CRITICAL REVIEW OF MEDICAL MANAGEMENT IN MALIGNANT PLEURAL MESOTHELIOMA - AUSTRALIA, 2011 Dr Malcolm Feigen Senior Radiation Oncologist Radiation Oncology Centre Austin Health
Asbestos Related Diseases
 Asbestos Related Diseases Asbestosis Mesothelioma Lung Cancer Pleural Disease Asbestosis and Mesothelioma (LUNG CANCER) Support Group 1800 017 758 www.amsg.com.au ii Helping you and your family through
Asbestos Related Diseases Asbestosis Mesothelioma Lung Cancer Pleural Disease Asbestosis and Mesothelioma (LUNG CANCER) Support Group 1800 017 758 www.amsg.com.au ii Helping you and your family through
MESOTHELIOMA. Not Just a Late Night Commercial. Graciela Hoal, RN, MSN, ACNP-BC
 MESOTHELIOMA Not Just a Late Night Commercial Graciela Hoal, RN, MSN, ACNP-BC Saturday Session Thoracic Surgery Nurse Practitioner Greater Los Angeles Veteran Affairs Objectives Course Objectives: Discuss
MESOTHELIOMA Not Just a Late Night Commercial Graciela Hoal, RN, MSN, ACNP-BC Saturday Session Thoracic Surgery Nurse Practitioner Greater Los Angeles Veteran Affairs Objectives Course Objectives: Discuss
Is pleurectomy and decortication superior to palliative care in the treatment of malignant pleural mesothelioma?
 doi:10.1510/icvts.2010.256271 Interactive CardioVascular and Thoracic Surgery 12 (2011) 812 817 www.icvts.org Best evidence topic - Thoracic oncologic Is pleurectomy and decortication superior to palliative
doi:10.1510/icvts.2010.256271 Interactive CardioVascular and Thoracic Surgery 12 (2011) 812 817 www.icvts.org Best evidence topic - Thoracic oncologic Is pleurectomy and decortication superior to palliative
Laparoscopic Ultrasonography Assisted Retroperitoneal Lymph Node Sampling in Patients Evaluated for Stomach Cancer Recurrence
 Case Series Laparoscopic Ultrasonography Assisted Retroperitoneal Lymph Node Sampling in Patients Evaluated for Stomach Cancer Recurrence Honsoul Kim, MD, Woo Jin Hyung, MD, Joon Seok Lim, MD, Mi-Suk Park,
Case Series Laparoscopic Ultrasonography Assisted Retroperitoneal Lymph Node Sampling in Patients Evaluated for Stomach Cancer Recurrence Honsoul Kim, MD, Woo Jin Hyung, MD, Joon Seok Lim, MD, Mi-Suk Park,
CT findings in Differential Diagnosis between Tuberculous Pleurisy and Malignant Effusion
 CT findings in Differential Diagnosis between Tuberculous Pleurisy and Malignant Effusion Poster No.: E-0084 Congress: ESTI 2012 Type: Scientific Exhibit Authors: S. S. Shim, Y. Kim; Seoul/KR Keywords:
CT findings in Differential Diagnosis between Tuberculous Pleurisy and Malignant Effusion Poster No.: E-0084 Congress: ESTI 2012 Type: Scientific Exhibit Authors: S. S. Shim, Y. Kim; Seoul/KR Keywords:
Basic Data. 鍾 XX, female Age:59 y/o
 Basic Data 鍾 XX, female Age:59 y/o Chief complain for evaluation of the left pleural mass Clinical course This 59 years old lady Hypertension for 3 years, under herbal control. An episode of high BP up
Basic Data 鍾 XX, female Age:59 y/o Chief complain for evaluation of the left pleural mass Clinical course This 59 years old lady Hypertension for 3 years, under herbal control. An episode of high BP up
Mesothelioma and Asbestos
 CANCER INFORMATION FACTSHEET Mesothelioma and Asbestos The information in this factsheet will help you to understand more about mesothelioma. It is an agreed view on this cancer by medical experts. We
CANCER INFORMATION FACTSHEET Mesothelioma and Asbestos The information in this factsheet will help you to understand more about mesothelioma. It is an agreed view on this cancer by medical experts. We
How To Treat Mesothelioma
 Mesothelioma in the UK Liz Darlison Mesothelioma UK The National Macmillan Mesothelioma Resource Centre June 2010 Mesothelioma Applied Research Foundation - 2010 www.curemeso.org Contents Size of Mesothelioma
Mesothelioma in the UK Liz Darlison Mesothelioma UK The National Macmillan Mesothelioma Resource Centre June 2010 Mesothelioma Applied Research Foundation - 2010 www.curemeso.org Contents Size of Mesothelioma
British Thoracic Society Standards of Care Committee
 250 Thorax 2001;56:250 265 BTS statement Statement on malignant mesothelioma in the United Kingdom British Thoracic Society Standards of Care Committee Correspondence to: Dr J Wiggins, Wexham Park Hospital,
250 Thorax 2001;56:250 265 BTS statement Statement on malignant mesothelioma in the United Kingdom British Thoracic Society Standards of Care Committee Correspondence to: Dr J Wiggins, Wexham Park Hospital,
Small cell lung cancer
 Small cell lung cancer Small cell lung cancer is a disease in which malignant (cancer) cells form in the tissues of the lung. The lungs are a pair of cone-shaped breathing organs that are found within
Small cell lung cancer Small cell lung cancer is a disease in which malignant (cancer) cells form in the tissues of the lung. The lungs are a pair of cone-shaped breathing organs that are found within
Lung cancer LUNG CANCER. Box 1 Clinical signs
 22 LUNG CANCER Lung cancer Bronchial carcinoma refers to two distinct clinical entities small cell and non-small cell carcinoma. Although these conditions have much in common, with broadly similar presenting
22 LUNG CANCER Lung cancer Bronchial carcinoma refers to two distinct clinical entities small cell and non-small cell carcinoma. Although these conditions have much in common, with broadly similar presenting
Malignant Mesothelioma: an Update
 Malignant Mesothelioma: an Update Nico van Zandwijk Asbestos Diseases Research Institute Bernie Banton Centre University of Sydney Australia Physicians Week RACP 19-5-2009 Health Risks of Asbestos Fibers
Malignant Mesothelioma: an Update Nico van Zandwijk Asbestos Diseases Research Institute Bernie Banton Centre University of Sydney Australia Physicians Week RACP 19-5-2009 Health Risks of Asbestos Fibers
Radiologic Diagnosis of Spinal Metastases
 September 2002 Radiologic Diagnosis of Spinal Metastases Natalie J. M. Dailey, Harvard Medical Student Year III Our Patient s Presenting Story 70 year old male Presents to the hospital for laparascopic
September 2002 Radiologic Diagnosis of Spinal Metastases Natalie J. M. Dailey, Harvard Medical Student Year III Our Patient s Presenting Story 70 year old male Presents to the hospital for laparascopic
Post-recurrence survival in completely resected stage I non-small cell lung cancer with local recurrence
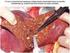 Post- survival in completely resected stage I non-small cell lung cancer with local J-J Hung, 1,2,3 W-H Hsu, 3 C-C Hsieh, 3 B-S Huang, 3 M-H Huang, 3 J-S Liu, 2 Y-C Wu 3 See Editorial, p 185 c A supplementary
Post- survival in completely resected stage I non-small cell lung cancer with local J-J Hung, 1,2,3 W-H Hsu, 3 C-C Hsieh, 3 B-S Huang, 3 M-H Huang, 3 J-S Liu, 2 Y-C Wu 3 See Editorial, p 185 c A supplementary
Lung Cancer. This reference summary will help you better understand lung cancer and the treatment options that are available.
 Lung Cancer Introduction Lung cancer is the number one cancer killer of men and women. Over 165,000 people die of lung cancer every year in the United States. Most cases of lung cancer are related to cigarette
Lung Cancer Introduction Lung cancer is the number one cancer killer of men and women. Over 165,000 people die of lung cancer every year in the United States. Most cases of lung cancer are related to cigarette
General Rules SEER Summary Stage 2000. Objectives. What is Staging? 5/8/2014
 General Rules SEER Summary Stage 2000 Linda Mulvihill Public Health Advisor NCRA Annual Meeting May 2014 National Center for Chronic Disease Prevention and Health Promotion Division of Cancer Prevention
General Rules SEER Summary Stage 2000 Linda Mulvihill Public Health Advisor NCRA Annual Meeting May 2014 National Center for Chronic Disease Prevention and Health Promotion Division of Cancer Prevention
