EURO DIAGNOSTICA PRODUCT CATALOG
|
|
|
- Oswald Casey
- 9 years ago
- Views:
Transcription
1 EURO DIAGNOSTICA PRODUCT CATALOG
2 Copyright: 2012, Euro Diagnostica AB Version: Graphic design: Oddville AB Printed by: Bildcenter i Lund AB
3 Introduction Welcome to Euro Diagnostica We are proud to introduce the 2012/2013 Euro Diagnostica Product Catalog Today, Euro Diagnostica is an international diagnostic solutions provider in autoimmunity testing with a long history and commitment to quality. The company celebrates 20 years this year and was created following a merger between Ferring Diagnostica, Balticor Diagnostica, Euro Diagnostics and Medscand Diagnostics (MILAB). In 2004, Euro Diagnostica expanded its services by acquiring Wieslab AB, Sweden, a leading autoimmunity testing laboratory. From its facilities in Malmö, Sweden, Euro Diagnostica develops, manufactures and markets an extensive product range of the highest possible quality in over 100 countries. Our complete diagnostic kits and reagents are used daily in medical assessments both manually and on the majority of instrument platforms available worldwide. Our tests are developed for clinical auto immunology, microbiology and immunochemistry. Thanks to the long experience and continuous development of our personnel and production processes, Euro Diagnostica offers top quality cost-effective customized coating services. Our Laboratory Testing Unit, Wieslab, offers clinical testing and disease assessment within autoimmunity by qualified diagnostic specialists every day of the year. Wieslab Laboratory Services offers a comprehensive range of individual and panel tests, and ensures accurate and fast results. Testing is available for most autoimmune diseases. Comprehensive Product Portfolio As you will see, we offer test systems for diagnostics and treatment-associated monitoring in the following major focus areas: Early diagnosis of rheumatoid arthritis the anti-ccp 2 assay is one of the main focus products for our company Serological detection of autoimmune vasculitis we offer the most complete product range for ANCA testing Complement deficiency diagnosis our Complement product line provides a unique functional assessment of the Complement system Neuro-endocrine tumors diagnosis Chromogranin A is available both as radioimmunoassay and ELISA. Our ELISA tests are harmonised for use on Dynex instruments Our Mission - improving patient care Our aim is to constantly improve and expand our offering to contribute to better patient care. To achieve this, we have made a clear shift in emphasis. Euro Diagnostica now has a clear focus on diagnosis, prognosis and theranostics of autoimmune and related diseases. We have a global approach and strategy, are expanding our current business plus our collaboration with research partners, and are building a worldwide sales and distribution network. By having the laboratory services as an integrated unit of our company, we know and understand the challenges laboratories and clinicians face every day in the autoimmunity testing. To help solve these challenges, we continuously validate innovative tests at Wieslab Laboratory Services to develop and bring new solutions for you and for patients. product catalog I 3
4 INTRODUCTION Introduction Welcome to Euro Diagnostica Quality is our main value Quality is a key element at Euro Diagnostica, codified in a quality system that has EN ISO 13485:2003 certification (Sweden). Euro Diagnostica does not regard these standards as a maximum to be achieved, but as a basis for the further improvement of our expertise and know-how. The advanced and extensive range of diagnostic products is the most tangible confirmation of our quality. Furthermore, we ensure that our buyers receive excellent customer service and technical support. We combine our expertise with great commitment in solving the practical issues confronting our clients. Our aim is to constantly develop solutions to aid clinicians in the diagnosis, prognosis, monitoring and treatment of autoimmune and related diseases. With so much at stake, our focus is on helping you to get the correct diagnosis to the patient every time. Euro Diagnostica s sights are set firmly on being part of the personalised medicine development. We thank you for your interest in our products and we commit ourselves to continually developing and providing you with the highest quality innovative clinical solutions. Welcome to Euro Diagnostica! In this edition, you will find short descriptions of all of our products. For a more comprehensive overview of our company, products and services, please visit us at our website: Visit our website 4 I product catalog
5 Euro Diagnostica ANSWERS TO YOU product catalog I 5
6 TABLE OF CONTENTS Product Catalog Table of contents Rheumatoid Arthritis About Rheumatoid Arthritis 09 CCPlus Immunoscan 10 Anti-CCP EDIA 11 CCPoint 12 CCPoint US 13 hcomp quantitative kit Wieslab 14 sgag quantitative kit Wieslab 15 Vasculitis About Vasculitis 17 PR3 ANCA Wieslab /DIASTAT 18 Capture PR3-ANCA Wieslab 18 MPO ANCA Wieslab /DIASTAT 19 Capture MPO-ANCA Wieslab 19 Anti-GMB, ANCA screen kit Wieslab 20 ANCA panel kit Wieslab 20 Goodpasture s syndrome About Goodpasture s syndrome 22 Anti-GBM semi quantitative kit Wieslab 23 Complement About Complement 25 Complement system Screen Wieslab 26 Complement system AP Wieslab 27 Complement system CP Wieslab 27 Complement system MBL Wieslab 28 Complement system Ficolin 3 Wieslab 28 Connective tissue disorders About Connective tissue disorder 29 ANA DIASTAT 30 Anti-Centromere DIASTAT 31 Anti-La (SS-B) DIASTAT 31 Anti-dsDNA DIASTAT 32 Anti-Jo-1 DIASTAT 33 Anti-Mitochondria DIASTAT 33 Anti-Scl-70 DIASTAT 33 Anti-Ro (SS-A) DIASTAT 34 Anti-Sm DIASTAT 34 Anti-Sm/RNP DIASTAT 35 ENA single well screen DIASTAT 35 ENA profile DIASTAT 35 Congenital heart block About Congenital heart block 36 Anti-SS-A p200 Wieslab 37 Anti-phospholipid syndrome About Antiphospholipid syndrome 39 Anti-beta-2-glycoprotein 1 IgG DIASTAT 40 Anti-beta-2-glycoprotein 1 IgM DIASTAT 40 Anti-Cardiolipin IgG DIASTAT 41 Anti-Cardiolipin IgM DIASTAT 41 Anti-Cardiolipin Total DIASTAT 41 Visit our website 6 I product catalog
7 Autoimmune Gastroenterology About Autoimmune Gastroenterology 42 ASCA IgA Wieslab 43 ASCA IgG Wieslab 43 Thyroid diagnostics About Thyroid diagnostics 44 Anti-Tg DIASTAT 45 Anti-TPO DIASTAT 45 Endocrine markers About Endocrine markers 46 Angiotensin II EURIA 47 ANP EURIA 47 CCK EURIA 48 Chromogranin A NeoLisa 48 Chromogranin A and B EURIA 49 Beta-Endorphin EURIA 50 Endothelin EURIA 50 Gastrin EURIA 51 Glucagon EURIA 51 Alpha-MSH EURIA 52 NPY EURIA 52 PP EURIA 53 Somatostatin EURIA 53 Vasopressin EURIA 54 VIP EURIA 54 Pertussis About Pertussis 55 Pertusscan TRO 56 Pertusscan PT IgG 56 Instrumentation 58 Wieslab Laboratory Services 60 Product Overview 62 Product List 68 Ordering Information 70 product catalog I 7
8 THE PAST THE PRESENT THE FUTURE Immunoscan CCPlus Ultimate Anti-CCP ELISA for the serological diagnosis of Rheumatoid Arthritis The CCP2 test is recognized as the gold standard of testing for anti-ccp CCPoint Unique CCP2 rapid test Only 10 minutes to result Visit our website 8 I product catalog
9 Rheumatoid arthritis Disease information Rheumatoid arthritis (RA) is an autoimmune disease characterized by chronic inflammation of the synovial joints The inflammation leads to joint swelling, progressive joint erosion and ultimately to cartilage and bone destruction. The disease is often disabling and significantly affects quality of life. Rheumatoid arthritis is prevalent in all ethnic groups, affecting approximately % of the population of the western world. As is often the case with autoimmune diseases, there is a female over-representation. Onset is most frequent between the ages of 40 and 50, but people of any age can be affected. There is clearly a genetic disposition to the disease suggested by the fact that individuals with the HLA-DR1 or HLA-DR4 serotypes have an increased risk for developing RA. Symptoms The clinical picture of RA involves signs of inflammation, with the affected joints being swollen, warm, painful and stiff, particularly early in the morning on waking or following prolonged inactivity. The swelling of the joint is caused by the excessive production of joint fluid (synovial fluid) as part of the inflammation process (synovitis). While rheumatoid arthritis primarily affects joints, problems involving other organs of the body are known to occur. Extra-articular manifestations are clinically evident in about 15 25% of individuals with rheumatoid arthritis. The rheumatoid nodule, which is sometimes cutaneous, is the feature most characteristic of rheumatoid arthritis. This is a type of inflammatory reaction known to pathologists as a necrotizing granuloma. Fibrosis of the lungs is a recognized extra-articular manifestation of rheumatoid disease. The lung tissue itself can also become inflamed, scarred, and sometimes inflammatory nodules (rheumatoid nodules) develop within the lungs. Inflammation of the pericardium surrounding the heart, called pericarditis, can cause a chest pain. People with rheumatoid arthritis are more prone to atherosclerosis, and risk of myocardial infarction (heart attack) and stroke is markedly increased. Disease outcome may vary from mild clinical symptoms to severe systemic disease when joint destruction is accompanied by these extra-articular manifestations. Diagnosis So far, no therapy has been developed that cures the disease. Current therapies may, however, slow down the extent of swelling and erosive damage. To achieve the greatest therapeutic potential and effect it is essential to initiate treatment at an early stage of the disease. Early and accurate diagnosis is therefore of outmost importance and the presence of autoantibodies to citrullinated proteins/peptide (anti-ccp) has an important prognostic value for the disease. These antibodies can be detected even before the onset of clinical symptoms. The anti-ccp2 test has a very high specificity and sensitivity and is a reliable and accurate toolset for the early diagnosis and follow-up of RA. Due to its wide clinical use, anti-ccp is included in the ACR/EULAR criteria for diagnosing RA. The Rheumatoid Arthritis Classification Criteria, jointly published by the American College of Rheumatology (ACR) and the European League Against Rheumatism (EULAR) established a point value between 0 and 10. Every patient with a point total of 6 or higher is unequivocally classified as an RA patient. Of these six points, three may be acquired by presence of anti-ccp2 autoantibodies. Code Name Description Page RA-96PLUS 1 CCPlus Immunoscan ELISA 10 FCCP Anti-CCP EDIA ELISA 11 CCPoint 20 2 CCPoint Lateral flow/rapid test 12 CCPoint 20 US 1 CCPoint US Lateral flow/rapid test 13 COMP hcomp quantitative kit Wieslab ELISA 14 GAG sgag quantitative kit Wieslab Dye binding assay 15 1) Please contact your local representative for availability in your country, available in US! 2) Please contact your local representative for availability in your country, not available in US! 3) For research use only, contact your local representative for research use availability in your country. product catalog I 9
10 RHEUMATOID ARTHRITIS Rheumatoid arthritis Products CCPlus Immunoscan The Immunoscan CCPlus assay is based on highly purified synthetic peptides containing citrulline residues selected on the basis of their superior performance in detecting Rheumatoid arthritis RA autoantibodies. Euro Diagnostica anti-ccp testing and the anti-ccp2 test is recognized as the gold standard of testing for anti-ccp. The majority of anti-ccp2 in the world are supplied by Euro Diagnostica. Intended use Immunoscan CCPlus test kit is an enzyme-linked immunosorbent assay (ELISA) for qualitative and semi-quantitative determination of IgG antibodies to Cyclic Citrullinated Peptides (CCP) in human sera. The assay is used to detect antibodies in a single serum specimen. The results of the assay are to be used as an aid to the diagnosis of Rheumatoid Arthritis (RA), in conjunction with other laboratory and clinical findings. The analysis should be performed by trained laboratory professionals. For in vitro diagnostic use. Background Since the first report in 1998 that antibodies reacting with synthetic peptides containing the amino acid citrulline are highly specific for RA, the measurement of anti-ccp antibodies has become the method of choice in the accurate diagnosis of this disease. The anti-ccp2 assay is as sensitive as the rheumatoid factor test but with much higher specificity. Anti- CCP antibodies have a high positive predictive value and can be detected years before the development of clinical symptoms. A diagnostic value for the measurement of anti-ccp antibodies has been found in relation to joint involvement and radiological damage in early RA. The presence of anti-citrullinated protein antibodies is now one of the ACR-criteria for classification of RA as well as part of EULAR s recommendations. Sensitivity at stratified specificity (98.5%) for available RA diagnostic markers (100 RA patients and 102 healthy controls) Immunoscan CCPlus 73 CCP3 67 MCV 62 RF 17 Ref: Bizzaro N, Tonutti E, Tozzoli R, Villalta D.Clin Chem Aug;53(8): Epub 2007 Jun 22 RA-96PLUS 1 CCPlus Immunoscan 96 wells (12x8 microtitre strips) 1) Please contact your local representative for availability in your country, available in US! Visit our website 10 I product catalog
11 Anti-CCP EDIA Antibodies against cyclic citrullinated peptide (anti-ccp) are highly specific for rheumatoid arthritis (RA), one of the most common systemic autoimmune diseases affecting up to 0.4-1% of the world population. Euro Diagnostica, in collaboration with world-leading scientists, invented anti-ccp testing and the anti-ccp2 test is recognized as the gold standard of testing for anti-ccp. Today, Euro Diagnostica manufactures and supplies the majority of anti-ccp2 in the world. The presence of anti-citrullinated protein antibodies is now one of the ACR-criteria for classification of RA. The EDIA TM anti-ccp kit contains improved synthetic peptides selected on the basis of their superior performance in the detection of RA autoantibodies. Intended use EDIA anti-ccp test kit is an enzyme-linked immunosorbent assay (ELISA) for detection and semi-quantitation of IgG antibodies to Cyclic Citrullinated Peptides (CCP) in human serum or plasma. The assay is used to detect antibodies in a single specimen. Assay results are used to aid the diagnosis of Rheumatoid Arthritis (RA), in conjunction with other laboratory and clinical findings. The analysis should be performed by trained laboratory professionals. For in vitro diagnostic use. Background Rheumatoid Arthritis (RA) is one of the most common systemic autoimmune diseases. FCCP Anti-CCP EDIA ELISA (96 wells) 1) Please contact your local representative for availability in your country, available in US! The 2010 American College of Rheumatology/European League Against Rheumatism classification criteria for rheumatoid arthritis Target population (Who should be tested?): Patients who 1) have at least 1 joint with definite clinical synovitis (swelling) 2) with the synovitis not better explained by another disease Classification criteria for RA (score-based algorithm: add score of categories A-D; a score of 6/10 is needed for classification of a patient as having definite RA). A. Joint involvement Score 1 large joint large joints small joints (with or without involvement of large joints) 4 10 small joints (with or without involvement of large joints) >10 joints (at least 1 small joint) 5 B. Serology Score Negative RF and negative ACPA 0 Low-positive RF or low-positive ACPA 2 High-positive RF or high-positive ACPA 3 C. Acute-phase reactants Score Normal CRP and normal ESR 0 Abnormal CRP or abnormal ESR 1 D. Duration of symptoms Score 2 3 <6 weeks 0 6 weeks 1 Reference: Arthritis & Rheumatism Vol 62(9), pages , September 2010 product catalog I 11
12 RHEMATOID ARTHRITIS Rheumatoid arthritis Products CCPoint The first RAPID test for anti-ccp that enables fast, near-patient diagnosis, thus making quick treatment start possible. Percent agreement of the CCPoint assay compared to alternative anti-ccp ELISA Intended use Euro Diagnostica CCPoint test is a visually read, qualitative rapid lateral flow test for the detection of IgG antibodies to Cyclic Citrullinated Peptides (CCP) in human capillary whole blood, plasma or serum. Test results are used to aid the diagnosis of Rheumatoid Arthritis (RA), in conjunction with other laboratory and clinical findings. For use by healthcare professionals. For in vitro diagnostic use. CCPoint Alternative ELISA Positive Negative Total Positive Negative Total Background CCPoint test is a colloidal gold-based lateral flow immunoassay. Reactive cyclic citrullinated peptides are immobilised as a discrete line on a porous membrane located in the test zone. The detection reagent, consisting of colloidal gold particles conjugated to anti-human IgG, is deposited within the device onto the conjugate pad. In the assay procedure, a sample of blood is added to the sample port. A blood cell separation membrane transfers the sample fluid onto the porous membrane. After short incubation, running buffer is added to the buffer port. This buffer mobilizes the colloidal gold particles from the conjugate pad. The gold particles and the sample move by capillary force across the membrane. If the sample contains anti-ccp antibodies, they will bind to the peptide-antigens and a red line will appear in the test zone (marked T). If the sample does not contain anti-ccp antibodies, no line will appear. With any sample, a red control line should appear in the control zone (marked C). The control ensures that the coated colloidal gold is still active. Kit components (Contents of CCPoint 20) 20 Foil pouches containing the CCPoint test device 22 Disposable pipettes (20 µl) 2 Droplet bottles, each containing 3 ml of running buffer 1 CCPoint Instructions for Use 1 Quick Reference Instruction CCPoint 20 2 CCPoint Lateral flow/rapid test 2) Please contact your local representative for availability in your country, not available in US! Visit our website 12 I product catalog
13 CCPoint US The first anti-ccp test that enables rapid diagnosis, which makes an early initiation of treatment possible. Percent agreement of the CCPoint assay compared to alternative anti-ccp ELISA Intended use EuroDiagnostica CCPoint test is a visually read, qualitative rapid lateral flow test for the detection of IgG antibodies to Cyclic Citrullinated Peptides (CCP) in human serum or plasma. Test results are used to aid to the diagnosis of Rheumatoid Arthritis (RA), in conjunction with other laboratory and clinical findings. For use by trained laboratory professionals. For in vitro diagnostic use. CCPoint US Alternative ELISA Positive Negative Total Positive Negative Total Background CCPoint test is a colloidal gold-based lateral flow immunoassay. Reactive cyclic citrullinated peptides are immobilised as a discrete line on a porous membrane located in the test zone. The detection reagent, consisting of colloidal gold particles conjugated to anti-human IgG, is deposited within the device onto the conjugate pad. In the assay procedure, a sample of serum or plasma is added to the sample port. A blood cell separation membrane transfers the sample fluid onto the porous membrane. After a short incubation, running buffer is added to the buffer port. This buffer mobilizes the colloidal gold particles from the conjugate pad. The gold particles and the sample move by capillary force across the membrane. If the sample contains anti-ccp antibodies, they will bind to the peptide-antigens and a red line will appear in the test zone (marked T). If the sample does not contain anti-ccp antibodies, no line will appear. With any sample, a red control line should appear in the control zone (marked C). The control ensures that the coated colloidal gold is still active. Kit components (Contents of CCPoint US 20) 20 Foil pouches containing the CCPoint test device 2 Droplet bottles, each containing 3 ml of running buffer 1 CCPoint Instructions for Use CCPoint 20 US 1 CCPoint US Lateral flow/rapid test 1) Please contact your local representative for availability in your country, available in US! product catalog I 13
14 RHEMATOID ARTHRITIS Rheumatoid arthritis Products hcomp quantitative kit Wieslab Serum levels of COMP can provide important information about metabolic changes occurring in the cartilage matrix in joint disease. Serum COMP levels have been shown to correlate with cartilage degradation and are thus a potential prognostic marker in inflammatory joint diseases such as osteoarthritis (OA) and rheumatoid arthritis (RA). Intended use Wieslab hcomp is an enzyme immunoassay for quantitation of intact or fragmented human Cartilage Oligomeric Matrix Protein (COMP) in serum or synovial fluid. The assay serves as an indicator of cartilage turnover. The analysis should be performed by trained laboratory professionals. For research use only. Background COMP is the major non-collagenous protein in cartilage. It is important for the formatation and stability of cartilage. Measurement of intact COMP and fragments thereof in synovial fluid or serum has been shown to correlate with cartilage destruction in rheumatoid arthritis and osteoarthritis patient studies. In synovial fluid, COMP is present to some extent as an apparently intact protein but the majority is found as several different fragments, as in serum. COMP hcomp quantitative kit Wieslab 96 wells 3) For research use only, contact your local representative for research use availability in your country. Visit our website 14 I product catalog
15 sgag quantitative kit Wieslab The Alcian Blue reagent in Wieslab s sgag assay has been carefully selected and optimised for proteoglycan assays. The most abundant heteropolysaccharides in the body are the glycosaminoglycans (GAGs). They are located primarily on the surface of cells or in the extracellular matrix. As an example, cartilage is composed to a large extent of GAGs, which are the dominant part of the proteo - glycan Aggrecan. COOH O H H 4 1 OH H H H OH O HO S O O β O H CH 2 OH O O 3 H H H NHCOCH 3 GAGs are negatively-charged long unbranched polymeric polysaccharides composed of repeating units of disaccharides containing one uronic acid or galactose and one amino sugar, either N-acetyl-glucosamine or N-acetylgalactosamine. The variation in charge may be very large since each disaccharide is more or less sulphated. Based on GAG s high negative charge, a number of dye-binding procedures for their measurement have been developed. In most cases, how ever, these are not applicable to biological material without different forms of pre-treatment such as protease digestion. The present assay makes use of the dye Alcian blue, which has a long history as a histological tissue staining reagent. Alcian blue is a tetravalent cation with a hydrophobic core. The four charges allow the dye to bind to negativelycharged polymers such as GAGs at high ionic strength, in contrast to other cationic dyes which are all monovalent. The molecular structure of Alcian blue, i.e. the plane tetragonal hydrophobic core with positive charges at its corners, may facilitate formation of aggregates of several molecules side-by-side rather than micelle formation. The ionic strength, ph and presence of detergents will affect the size of these aggregates in solution. GAG sgag quantitative kit Wieslab 200 3) For research use only, contact your local representative for research use availability in your country. product catalog I 15
16 ULTIMATE SEROLOGICAL TOOL FOR VASCULITIS DIAGNOSIS Wieslab Capture ANCA Higher clinical sensitivity and clinically proven prognostic performance Visit our website 16 I product catalog
17 Vasculitis Disease information Vasculitis is a general term referring to a heterogeneous group of diseases that are characterised by inflammatory destruction of blood vessels The inflammation cause changes in the vessel wall, which can lead to serious complications damaging the body s organs. The cause of many forms of vasculitis is not understood. Often there is a presence of autoantibodies that are sometimes used in classification, as in ANCAassociated vasculitides (Anti-neutrophil cytoplasmic antibodies). Examples of ANCA-associated vasculitides are granulomatosis with polyangiitis (GPA) (previously named Wegener s granulomatosis), microscopic polyangiitis (MPA), and Churg-Strauss syndrome. GPA is a vasculitis that affects the nose, lungs, kidneys and other organs. The incidence of GPA is about 10 cases per million per year. It is a life-threatening disease and requires life-long immunosuppression. Initial signs are extremely variable, and diagnosis can be delayed due to the vague nature of the symptoms. Often the kidneys and lungs are affected and their functions impaired. A serious complication of GPA is rapidly progressive glomerulonephritis (RPGN). Microscopic polyangiitis (MPA) is characterized by small vessel vasculitis without evidence of granulomatous inflammation. The annual incidence in the US of MPA is 3.6 cases per million persons. MPA and GPA comprise a category of small vessel vasculitis related to the presence of ANCAs. MPA and GPA seem to be part of a clinical spectrum and share a number of clinical symptoms. However, the absence of granuloma formation and sparing of the upper respiratory tract are features of MPA that help to distinguish it from GPA. Occasionally, however, the two conditions are difficult to distinguish. Churg-Strauss syndrome mainly involves the small to medium blood vessels of the lungs but eventually also affects the gastrointestinal tract, heart, skin and kidneys. It may also affect the peripheral nerves. Churg-Strauss syndrome is often referred to as allergic granulomatosis due to its clinical course often starting as a severe form of asthma. Code Name Description Page Cap PR3 IU 2 Wieslab Capture PR3-ANCA PR3 ANCA (Capture) 18 PR3 IU 2 Wieslab PR3-ANCA PR3 ANCA 18 FPRO DIASTAT PR3-ANCA PR3 ANCA 18 Cap MPO IU 2 Wieslab Capture MPO-ANCA MPO ANCA (Capture) 19 MPO IU 2 Wieslab MPO-ANCA MPO ANCA 19 FMPO DIASTAT MPO-ANCA MPO ANCA 19 GCP Anti-GBM, ANCA screen kit Wieslab GBM + MPO ANCA + PR3 ANCA 20 PAN ANCA panel kit Wieslab Azurocidin,BPI, Cathepsin G, Elastase, Lactoferrin, Lysozyme ANCA CP Wieslab ANCA screen kit MPO ANCA + PR3 ANCA 20 GCP-CAP 2 Vasculitis screen Wieslab GBM + MPO ANCA (Capture)+ PR3 ANCA Capture GP 104X 1 Wieslab Anti-GBM, semi quantitative kit GBM 23 FGBM DIASTAT Anti-GBM, ANCA screen kit GBM ) Please contact your local representative for availability in your country, available in US! 2) Please contact your local representative for availability in your country, not available in US! product catalog I 17
18 VASCULITIS Vasculitis Products PR3-ANCA Capture PR3-ANCA Wieslab Wieslab Capture PR3-ANCA test kit is an enzyme-linked immunosorbent assay (ELISA) for detection and quantitation of IgG antibodies to proteinase 3 (PR3) in human sera. The assay is used to detect antibodies in a single serum specimen. Assay results are used to aid the diagnosis of granulomatosis with polyangiitis. Wieslab Capture PR3-ANCA is standardized against the AF-CDC standard, code IS2721 (Human Reference Serum 16). Overview ANCAs (anti-neutrophil cytoplasmic antibodies) are a family of autoantibodies related to vasculitis and inflammatory disorders. Since 1985, when c-anca was shown to be related to granulomatosis with polyangiitis, interest in ANCAs has steadily increased. Today these antibodies are considered to be major diagnostic tools for the diagnosis and follow up of systemic vasculitis. Comparison of Wieslab Capture ANCA PR3 assay with other methods n= 120 Wieslab Capture PR3 Linker bound PR3 Mixed PR3 antigen Direct PR3 coating Sensitivity 40/40* 37/40 29/40 34/40 % 100% 93% 73% 85% Specificity 80/80** 79/80 79/80 37/42 % 100% 99% 99% 88% *Selected material, confirmed vasculitis **Blood donors, disease controls Reference: In-house data Clinical evaluation of capture assays for determining PR3 ANCA shows that this format has a higher diagnostic sensitivity for detecting patients with GPA compared with the direct-coating method or Indirect Immunofluorescence. A follow-up study of systemic vasculitis demonstrated that a high level of PR3-ANCA detected with the capture method was associated with significantly poorer renal survival plus higher mortality. No such correlation could be seen with the direct assay. Moreover, studies also show that the capture assay displays higher sensitivity for detecting relapses, which further suggests a biological relation between disease progression and epitope specificity. Principle of the Wieslab Capture PR3 ANCA assay Microtitre plate wells are coated with anti-pr3 monoclonal antibody in complex with PR3. During the first incubation, specific autoantibodies in diluted serum bind to the antigen coating. Optical density is measured following subsequent washing and incubation steps that involve adding conjugate and substrate. Cap PR3 IU 2 Wieslab Capture PR3-ANCA 96 wells PR3 IU 2 Wieslab PR3-ANCA 96 wells FPRO DIASTAT PR3-ANCA 96 wells 1) Please contact your local representative for availability in your country, available in US! 2) Please contact your local representative for availability in your country, not available in US! Visit our website 18 I product catalog
19 MPO-ANCA Capture MPO-ANCA Wieslab Wieslab Capture MPO-ANCA test kit is an enzyme-linked immunosorbent assay (ELISA) for detection and quantitation of IgG antibodies to Myeloperoxidase (MPO) in human sera. The assay is used to detect antibodies in a single serum specimen. Results are used to aid the of Vasculitis, in particular Microscopic polyangitiis. Background ANCAs (anti-neutrophil cytoplasmic antibodies) are a family of autoantibodies related to vasculitis and inflammatory disorders. When c-anca was shown to relate to granulomatosis with poly-angiitis (Wegener s granulomatosis (WG)), interest in ANCAs has increased steadily. Today these antibodies are considered to be major diagnostic tools for investigating systemic vasculitis. Measurement of MPO-specific ANCA is an important adjunct to clinical findings in the evaluation of clinical sub-types within the systemic vasculitides spectrum. Principle of the Wieslab Capture MPO ANCA assay Microtitre plate wells are coated with anti-mpo monoclonal antibody in complex with MPO. During the first incubation, specific autoantibodies in diluted serum bind to the antigen coating. Optical density is measured following subsequent washing and incubation steps that involve adding conjugate and substrate. The Wieslab Capture MPO-ANCA is standardized against the AF-CDC standard (Reference Human Serum 15, code IS2720). Cap MPO IU 2 Wieslab Capture MPO-ANCA 96 wells MPO IU 2 Wieslab MPO-ANCA 96 wells FMPO DIASTAT MPO-ANCA 96 wells 1) Please contact your local representative for availability in your country, available in US! 2) Please contact your local representative for availability in your country, not available in US! product catalog I 19
20 VASCULITIS Multi-parameter kits Products Anti-GBM, ANCA screen kit Wieslab A prompt diagnosis is essential for patients with medical conditions such as Goodpasture s Syndrome, granulomatosis with polyangiitis (Wegener s granulomatosis) or microscopic polyangiitis. Fast and simultaneous detection of autoantibodies against GBM, Proteinase 3 (PR3-ANCA) and Myeloperoxidase (MPO-ANCA) is possible with Wieslab Anti-GBM, ANCA Screening Test Kit. Wieslab Anti-GBM, ANCA Screening Test Kit is an enzyme-linked immunosorbent assay (ELISA) for the qualitative detection of antibodies to glomerular basement membrane (GBM), Proteinase-3 (PR3) and Myeloperoxidase (MPO) in human sera. The assay is used to detect antibodies in a single serum specimen. GCP Anti-GBM, ANCA screen kit Wieslab Break-apart microtitration strips (6x16) 96 wells CP Wieslab ANCA screen kit Break-apart microtitration strips (12x8) 96 wells GCP-CAP Vasculitis screen Wieslab Microtitration 96 wells 1) Please contact your local representative for availability in your country, available in US! ANCA panel kit Wieslab For ANCA-positive samples that are negative for PR3-ANCA and MPO-ANCA, an extended analysis is necessary. In addition, a prompt diagnosis is essential for patients vasculitis or other inflammatory disorders. Wieslab ANCA panel kit allows screening of six other factors that have been proposed to be involved in relevant disorders. Wieslab ANCA panel test kit is an enzyme-linked immunosorbent assay (ELISA) for qualitative detection of IgG antibodies to azurocidin, BPI, cathepsin G, elastase, lactoferrin, and lysozyme in human sera. The assay is used to detect antibodies in a single serum specimen. PAN ANCA panel kit Wieslab Microtitration strips (12x8) 96 wells 2) Please contact your local representative for availability in your country, not available in US! Visit our website 20 I product catalog
21 Euro Diagnostica ANSWERS TO YOU product catalog I 21
22 GOODPASTURE S SYNDROME Goodpasture s syndrome Disease information Goodpasture s syndrome Goodpasture s syndrome is a rare autoimmune disease characterised by lung haemorrhage, rapid destruction of the kidney and the presence of antibodies to glomerular basement membrane (GBM). The GBM antigen responsible for this disease is a component of the non-collagenous domain (NC1) of the alpha-3 chain of collagen type IV. Patients with Goodpasture s syndrome develop antibodies to GBM that attack collagen in the alveoli of the lungs and the glomeruli of the kidney. As there are several other autoimmune diseases that may present with similar symptoms, detection of anti-gbm antibodies is a useful aid in differentiating between these diseases. Deposits of antibodies in the basement membrane break down collagen and interrupt membrane integrity. This causes leakage of blood and a rapid inflammatory response leading to lung haemorrhage and kidney damage. Patients with Goodpasture s syndrome experience a rapid progression to renal failure and death if the disease is not recognised and treated early. Because of the vagueness of early symptoms and its rapid progression, diagnosis is often not reached until very late in the course of the disease. Like many autoimmune diseases, Goodpasture s syndrome responds well to treatment with corticosteroids and immunosuppressants. The concentration of anti-gbm antibodies in the blood may be reduced by plasmapheresis to remove blood plasma and replace a portion of the plasma with an isotonic salt and protein solution. As with many autoimmune conditions, the precise cause of Goodpasture s Syndrome is not yet known. It is believed to be a type II hypersensitivity reaction to Goodpasture s antigens on the basement membrane of the glomerulus of the kidneys and the pulmonary alveolus. Goodpasture s syndrome is a very rare condition and its incidence in Europe is between 1 in 1,000,000 and 1 in 2,000,000. Code Name Description Page GCP Anti-GBM, ANCA screen kit Wieslab Qualitative ELISA 20 GCP-CAP 2 Vasculitis screen Wieslab Qualitative ELISA 20 GP 104X 1 Wieslab Anti-GBM semiquantitative kit FGBM DIASTAT Anti-GBM, ANCA screen kit Quantitative and qualitative ELISA Semi-quantitative ELISA ) Please contact your local representative for availability in your country, available in US! 2) Please contact your local representative for availability in your country, not available in US! Visit our website 22 I product catalog
23 Goodpasture s syndrome Products Anti-GBM semi quantitative kit Wieslab Goodpasture s syndrome is characterised by lung haemorrhage, renal failure and the presence of anti-gbm antibodies. As several other autoimmune diseases may present with similar symptoms, this kit is a useful aid in differentiating between these diseases. Intended use Wieslab anti-gbm test kit is an enzyme-linked immunosorbent assay (ELISA) for detection and semi-quantitation of IgG antibodies to glomerular basement membrane (GBM) in human sera. The assay is used to detect antibodies in a single serum specimen. Its results aid the diagnosis of Goodpasture s syndrome. The analysis should be performed by trained laboratory professionals. Background Goodpasture s syndrome is characterised by lung haemorrhage, renal failure and the presence of anti-gbm antibodies. Diagnosis is based on clinical signs of lung haemorrhage and rapidly progressive glomerulonephritis combined with findings of anti-gbm antibodies. As several other autoimmune diseases may present with similar symptoms, this kit is a useful aid in differentiating between these diseases. Less than one third of patients with reno-pulmonary syndromes have antibodies against the Goodpasture antigen, the majority having either Proteinase 3-ANCA or Myeloperoxidase-ANCA. Historically, indirect immunofluorescence was used to detect anti-gbm antibodies. When the first ELISA based on a collagenase digest was published in 1981, assays using crude extracts were the only alternative. In 1984 the specific antigen was shown to derive from the C-terminal domain of type IV collagen (alpha 3 chain) and sensitive and specific assays were subsequently developed. The antigen is characterised by a restricted tissue distribution occurring mainly in kidney and lungs. GP 104X 1 Wieslab Anti-GBM semi quantitative kit 96 wells FGBM DIASTAT Anti-GBM, ANCA screen kit 96 wells 1) Please contact your local representative for availability in your country, available in US! product catalog I 23
24 COMPLEMENT COMPLEMENT SYSTEM FUNCTIONAL ASSESSMENT A unique diagnostic solution Simple and accurate ELISA All three pathways Lectin pathway with both MBL and Ficolin-3 Visit our website 24 I product catalog
25 Complement Disease information Complement deficiency The immune system is divided into innate immunity and adaptive immunity. Adaptive immunity is specific in its action and is constantly developing throughout life in response to and as an adaption to different pathogenic organisms. In contrast, innate immunity is inherited and acts as a first line of defense. Innate immunity includes cells and mechanisms that defend against a variety of pathogens in a non-specific mode. The complement system is an important part of the innate immune system. The complement system consists of about 30 different proteins, generally synthesized by the liver and normally circulating as inactive precursors. Stimulation by one of several trigger molecules will initiate an amplifying cascade of proteolytic cleavages. Complement can be activated by three pathways, the classical pathway, the lectin pathway and the alternative pathway. The end-result of this activation cascade is massive amplification of the response and activation of the cell-killing membrane attack complex (MAC). Complement may also act through other important mechanisms such as opsonization and chemotaxis. Complement deficiency and disease Deficiencies in complement predispose patients to infection basically via two mechanisms: (1) ineffective opsonization and (2) defects in lytic activity (defects in MAC formation). Individuals with complement deficiencies that hinder opsonization present with frequent recurrent infections and a high rate of morbidity and mortality. Patients with a defect in formation of MAC have a somewhat lesser degree of morbidity and mortality than, for example, patients with a defect in C3. Patients deficient in a lytic component of the complement cascade are at high risk for recurrent infection with Neisseria gonorrhoeae or Neisseria meningitidis. In addition to susceptibility to infections, a number of diseases are suggested to be affected by the complement system. Deficiency of the classical pathway is associated with an increased risk of developing Systemic Lupus Erythematosus (SLE). The complement system has been suggested to be involved in the pathogenesis of ANCA-associated vasculitides (AAV). Rheumatoid arthritis (RA) is affected by complement and although activation is potentially related to the occurrence of inflammation, complement deficiency may induce RA. Deficiency in the lectin pathway (MBL) seems to be associated with disease severity. Over activity of the alternative pathway, due to deficient complement inhibitor activity, has been implicated as a main cause of the seemingly unrelated pathologies of hemolytic uremic syndrome (HUS) and age-related macular degeneration (AMD). In addition, complement activity may have significant effect on the appearance and development/course of a variety of different diseases such as glomerulonephritis, atherosclerosis, Crohn s disease, Alzheimer s disease, and cardiac disease. Interest in complement and complement activity has grown exponentially over the last few years as a result of greater awareness of its important implications on a large variety of diseases and clinical conditions. Accordingly, interest in simple and accurate test methods for assessing complement function has grown significantly. Code Name Page COMPL Complement system screen Wieslab 26 COMPL AP Complement system AP Wieslab 27 COMPL CP Complement system CP Wieslab 27 COMPL MP Complement system MBL Wieslab 28 COMPLF 3 3 Complement system Ficolin 3 Wieslab 28 2) Please contact your local representative for availability in your country, not available in US! 3) For research use only, contact your local representative for research use availability in your country. product catalog I 25
26 FICOLIN 3 MASP-2 COMPLEMENT Complement Products Complement system Screen Wieslab Increased awareness and interest in the complement system has augmented the need for a simple and objective method to assess the function of complement activity. The Euro Diagnostica complement assay solution provides a complete picture of the complement function. Intended use Wieslab Complement system screen kit is an enzyme immunoassay for the qualitative determination of functional classical, MBL and alternative complement pathways in human serum. For in vitro diagnostic use. Background The complement system plays an essential role in chronic, autoimmune and infectious disease. There are three pathways of complement activation, namely the classical, the alternative and the MBL pathway. Universal detection Specific blocking Specific coating Classical Pathway Lectin Pathway C3 Membrane attack complex Alternative Pathway C1q MBL Ficolin 3 C3b/Factor b Impaired complement activity causes humans to become susceptible to repetitive fulminant or severe infections and may contribute to development of autoimmune disease. Inappropriate activation of complement contributes to chronic inflammation and tissue injury. Classical Pathway C9 Lectin Pathway MBL C9 Lectin Pathway Ficolin-3 C9 Alternative Pathway C9 Technical information Wieslab Complement assay combines principles of the hemolytic assay for complement activation with the use of labeled antibodies specific for neoantigen produced as a result of complement activation. The amount of neoantigen generated is proportional to the functional activity of complement pathways. C7 C6 C5 C3 C4 C2 C1 qrs C7 C6 C5 C3 C4 C2 MBL MASP-2 C7 C6 C5 C3 C4 C2 C7 C6 C5 C3 P FB FD IgM Mannan AcBSA LPS The wells of the microtitre strips are coated with specific activators of the classical, the MBL, or the alternative pathway. Patient serum is diluted in diluent containing specific blocker to ensure that only the respective pathway is activated. During the incubation of the diluted patient serum in the wells, complement is activated by the specific coating. The wells are then washed and C5b-9 is detected with a specific alkaline phosphatase-labelled antibody to the neoantigen expressed during MAC formation. COMPL Complement system Screen Wieslab Break-apart microtitration strips (4x8x3) 96 wells 2) Please contact your local representative for availability in your country, not available in US! Visit our website 26 I product catalog
27 Complement system Alternative Pathway Wieslab Increased awareness and interest in the complement system has augmented the need for a simple and objective method to assess the function of complement activity. The Euro Diagnostica complement assay solution provides a complete picture of the complement function. The complement system plays an essential role in chronic, autoimmune and infectious disease. There are three pathways of complement activation, namely the classical, the alternative and the MBL pathway. Wieslab COMPL AP 330 kit is an enzyme immunoassay for the qualitative determination of the functional alternative complement pathway in human serum. COMPL AP Complement system Alternative Pathway Wieslab Break-apart microtitration strips (12x8) 96 wells 2) Please contact your local representative for availability in your country, not available in US! Complement system Classical Pathway Wieslab Increased awareness and interest in the complement system has augmented the need for a simple and objective method to assess the function of complement activity. The Euro Diagnostica complement assay solution provides a complete picture of the complement function. Wieslab COMPL CP 310 is an enzyme immunoassay for the qualita tive determination of the functional classical complement pathway in human serum. COMPL CP Complement system Classical Pathway Wieslab Break-apart microtitration strips (12x8) 96 wells 2) Please contact your local representative for availability in your country, not available in US! product catalog I 27
28 COMPLEMENT Complement Products Complement system MBL Pathway Wieslab Increased awareness and interest in the complement system has augmented the need for a simple and objective method to assess the function of complement activity. The Euro Diagnostica complement assay solution provides a complete picture of the complement function. The complement system plays an essential role in chronic, autoimmune and infectious disease. There are three pathways of complement activation, namely the classical, the alternative and the MBL pathway. The Wieslab COMPL MP 320 kit is an enzyme immunoassay for the qualitative determination of the MBL complement pathway in human serum. COMPL MP Complement system MBL Pathway Wieslab Break-apart microtitration strips (12x8) 96 wells 2) Please contact your local representative for availability in your country, not available in US! Complement system Ficolin 3 Wieslab Increased awareness and interest in the complement system has augmented the need for a simple and objective method to assess the function of complement activity. The Euro Diagnostica complement assay solution provides a complete picture of the complement function. The Complement Ficolin 3 Wieslab kit is an enzyme immunoassay for the qualitative determination of Ficolin 3 in human serum. COMPLF 3 3 Complement system Ficolin 3 Wieslab 96 wells 3) For research use only, contact your local representative for research use availability in your country. Visit our website 28 I product catalog
29 Connective tissue disorders Disease information A number of systemic autoimmune diseases affect connective tissues Autoimmune connective tissue disorders may affect all connective tissues, i.e. joints, muscles, skin and blood vessels, and they therefore have multiple effects on many different organs throughout the body. The causes of autoimmune connective tissue diseases (CTD) are not known. It may involve genetic factors predisposing for the disease, although in several diseases the causal link is rather low. There is a significant female over-representation among autoimmune CTD patients. Autoimmune CTD are characterized as a group by the presence of spontaneous over-activity of the immune system that results in the introduction of extra antibodies into the circulation autoantibodies. The different diseases may develop slowly and eventually show distinct clinical symptoms, characteristic for a specific disease. However, the systemic nature of these diseases makes a differential diagnosis based on clinical signs only sometimes difficult. Different diseases often present with autoanti bodies with characteristics specific for a certain defined disease. Serological testing for these autoantibodies is thus a valuable tool in the diagnosis of autoimmune CTD. Code Name Page FANA ANA DIASTAT 30 FCEN Anti-Centromere DIASTAT 31 FALA Anti-La (SS-B) DIASTAT 31 FDNA Anti-dsDNA DIASTAT 32 FAJO Anti-Jo-1 DIASTAT 33 FMIT Anti-Mitochondria DIASTAT 33 FSCL Anti-Scl-70 DIASTAT 33 FARO Anti-Ro (SS-A) DIASTAT 34 FASM Anti-Sm DIASTAT 34 FRNP Anti-Sm/RNP DIASTAT 35 FSWS ENA-single well screen DIASTAT 35 FAID ENA profile DIASTAT 35 1) Please contact your local representative for availability in your country, available in US! product catalog I 29
30 CONNECTIVE TISSUE DISORDERS Connective tissue disorders Products ANA DIASTAT DIASTAT ELISA is an appropriate first-line screen for total anti-nuclear antibody. A positive ANA test result provides presumptive evidence for systemic rheumatic disease; further definition of specific antibody profiles is a valuable aid in the diagnostic process. DIASTAT anti-nuclear antibody (ANA) test is a qualitative enzymelinked immunosorbent assay (ELISA) for the detection of ANAs in human serum or EDTA plasma. It detects ANAs against Sm, Sm/RNP, Ro (SS-A), La (SS-B), Scl-70, Jo-1, dsdna, histone and centromere antigens. The test may be used for screening to eliminate samples negative for all ANAs. Samples that give a positive result should be further tested to identify the antigen-specific antibody or antibodies present. Background Detection and serological characterisation of specific autoantibodies plays an important role in the differential diagnosis of systemic rheumatic diseases. Autoantibodies to nuclear antigens (ANAs) are a group of antibodies specific for nuclear antigens including Sm, Sm/RNP, Ro (SS A), La (SS-B), Scl-70, Jo-1, dsdna, histone and centromere antigens. A positive ANA test result provides presumptive evidence for systemic rheumatic disease and further definition of specific antibody profiles is a valuable aid in the diagnostic process. A negative result reduces, but does not exclude, the likelihood of systemic rheumatic disease. Technical information The wells of the microtitre strips are coated with highly-purified HEp-2 cell extract. During the first incubation, specific ANAs in diluted serum or EDTA plasma bind to the antigen-coated surface. The wells are then washed to remove unbound components. In the second incubation, the Conjugate, enzyme-labelled antibodies to human IgG and IgM, binds any surface-bound autoantibodies. After further washing, specific autoantibodies are traced by incubation with the Substrate. Addition of Stop Solution terminates the reaction, resulting in a coloured end-product. FANA ANA DIASTAT ELISA (96 wells) Comparison with immunofluorescence 363 serum samples from patients with connective tissue disease (CTD) were compared with immunofluorescence on samples from patients with connective tissue disease at a number of European centres. Positivity or negativity on IFA was assigned according to the trial centre criteria. IFA DIASTAT +ve -ve +ve ve Sensitivity = 86% Specificity = 94% Overall agreement = 89% Comparison with immunofluorescence by individual autoantibodies The assay was compared with immunofluorescence with individual autoantibody specificities. These were determin ed using the specific DIASTAT ELISA kits. Antibody specificity Positive (ANA) Positive (IFA) Ro (SS-A) La (SS-B) Sm Sm/RNP Scl dsdna Centromere Histone % Agreement 28 samples identified as Jo-1 positive were tested, using the DIASTAT ANA kit had the following results: 24/28 (85.7%) were positive in the ANA test. 3/28 (10.7%) fell within the borderline area. Their ratios were 0.99, 0.86 and /28 (3.6%) gave a ratio of ) Please contact your local representative for availability in your country, available in US! Visit our website 30 I product catalog
31 Anti-Centromere DIASTAT Anti-centromere antibodies (ACAs) are detected in 49-96% of patients with CREST syndrome, with a particular association with Raynaud s phenomenon. The presence of ACAs may predate the clinical diagnosis of other CREST* syndrome abnormalities by up to several years. They have also been re ported in primary biliary cirrhosis in association with scleroderma, infrequently in rheumatoid arthritis and systemic lupus erythematosus, and occasionally in other immune disorders. Centromere CENP-B antigen is thought to be the primary autoantigen. An ELISA using the C-terminal CENP-B antigen is capable of detecting more ACA-positive sera than immunofluorescence and thus offers increased sensitivity. FCEN Anti-Centromere DIASTAT ELISA (96 wells) 1) Please contact your local representative for availability in your country, available in US! Anti-La (SS-B) DIASTAT The La (SS-B) antigen is an RNA-protein complex and antibodies against it are found in about 40% of patients with primary Sjögren s syndrome and in 15% of patients with systemic lupus erythematosus (SLE). Anti-La (SS-B) antibodies have also been detected in patients with systemic sclerosis and neonatal lupus erythematosus. In patients with Sjögrens s syndrome and SLE, the antibodies commonly occur together with Ro (SS-A). DIASTAT anti-la (SS-B or La) test is a quantitative/qualitative enzymelinked immunosorbent assay (ELISA) for the detection of autoantibodies specific for La (SS-B) antigen in human serum or EDTA, lithium heparin, citrated plasma. It is intended to aid in the diagnosis of systemic rheumatic diseases, particularly Sjögren s syndrome, and is not definitive in isolation. Autoantibody levels represent one parameter in a multicriterion diagnostic process. FALA Anti-La (SS-B) DIASTAT ELISA (96 wells) *CREST (calcinosis, Raynaud phenomenon, esophageal dysmotility, sclerodactyly, and telangiectasia) syndrome. 1) Please contact your local representative for availability in your country, available in US! product catalog I 31
32 CONNECTIVE TISSUE DISORDERS Connective tissue disorders Products Anti-dsDNA DIASTAT Antibodies against dsdna are characteristic for systemic lupus erythematosus (SLE) and are one of the criteria for disease classification by the American College of Rheumatology. The presence of these antibodies has high diagnostic specificity for SLE (90%) and can be used to monitor the treatment effect in patients. An increase in the level of anti-dsdna often indicates a relapse, while the level might become normal during remission. The detection of dsdna auto antibodies by the ELISA technique is a simple, sensitive, and reproducible method and can be standardised by reference to the World Health Organisation anti-dsdna standard, Wo/80. DIASTAT anti-dsdna test is a quantitative/qualitative enzyme-linked immunosorbent assay (ELISA) for the detection of IgG and IgM autoantibodies specific for double-stranded DNA (dsdna) in human serum or plasma. It is intended to aid in the diagnosis of systemic lupus erythematosus, and to monitor dsdna antibody levels during treatment and remission. Autoantibody levels represent one parameter in a multi-criterion diagnostic process. Background Systemic rheumatic diseases are autoimmune disorders such as systemic lupus erythematosus (SLE), rheumatoid arthritis and mixed connective tissue disease. A general feature of systemic rheumatic diseases is the presence of circulating antibodies to a variety of cellular antigens. The detection and serological characterisation of specific autoantibodies plays an important role in the differential diagnosis of these diseases. Technical information The wells of the microtitre strips are coated with a preparation of calf thymus selected for its high dsdna content. During the first incubation, specific autoantibodies in diluted serum or plasma bind to the antigencoated surface. The wells are then washed to remove unbound components. In the second incubation, the Conjugate, enzyme-labelled antibodies to human IgG and IgM, binds any surface-bound autoantibodies. After further washing, specific autoantibodies are traced by incubation with the Substrate. Addition of Stop Solution terminates the reaction, resulting in a coloured end-product. FDNA Anti-dsDNA DIASTAT ELISA (96 wells) 1) Please contact your local representative for availability in your country, available in US! Visit our website 32 I product catalog
33 Anti-Jo-1 DIASTAT Antibodies to the Jo-1 autoantigen, also recognised as the enzyme histidyl-trna synthetase, are found in approximately 25% of polymyositis patients. A high proportion of patients with interstitial lung disease, associated with polymyositis, are also positive for this autoantibody. DIASTAT anti-jo-1 test is a qualitative enzyme-linked immunosorbent assay (ELISA) for the detection of autoantibodies specific for Jo-1 antigen in human serum or plasma. FAJO Anti-Jo-1 DIASTAT ELISA (96 wells) 1) Please contact your local representative for availability in your country, available in US! Anti-Mitochondria DIASTAT Autoantibodies against the M2 antigen of the mitochondrial membrane are associated with primary biliary cirrhosis (PBC). PBC is a chronic and often fatal cholestatic autoimmune liver disease, primarily affecting young to middle-aged women. It is characterised by slow, progressive destruction of intrahepatic bile ducts, portal inflammation and scarring, eventually leading to cirrhosis and liver failure. FMIT Anti-Mitochondria DIASTAT ELISA (96 wells) 1) Please contact your local representative for availability in your country, available in US! Anti-Scl-70 DIASTAT Scl 70 antibody is a specific marker of systemic sclerosis. Antibodies to the Scl-70 autoantigen, also recognised as the enzyme topoisomerase 1, are found in approximately 20-28% of scleroderma patients and are highly specific for this disease. In the diffuse form of the disease, the frequency may be as high as 70%. DIASTAT anti-scl-70 test is a qualitative enzyme-linked immunosorbent assay (ELISA) for the detection of autoantibodies specific for Scl-70 antigen in human serum or plasma. FSCL Anti-Scl-70 DIASTAT ELISA (96 wells) 1) Please contact your local representative for availability in your country, available in US! product catalog I 33
34 CONNECTIVE TISSUE DISORDERS Connective tissue disorders Products Anti-Ro (SS-A) DIASTAT Autoantibodies to the Ro (SS-A) antigen are one of the most frequent serological markers of autoimmunity in rheumatic diseases. Ro (SS-A) ELISA is a sensitive and suitable method for detecting low titer antibodies. DIASTAT anti-ro (SS-A) test is a quantitative/qualitative enzyme-linked immunosorbent assay (ELISA) for the detection of autoantibodies specific for Ro (SS-A) antigen in human serum or EDTA, lithium heparin, citrated plasma. It is intended to aid the diagnosis of systemic rheumatic diseases, particularly Sjögren s syndrome, and is not definitive in isolation. Autoantibody levels represent one parameter in a multi-criterion diagnostic process. Systemic rheumatic diseases are autoimmune disorders such as systemic lupus erythematosus (SLE), polymyositis, Sjögren s syndrome, scleroderma and mixed connective tissue disease. A general feature of such diseases is the presence of circulating antibodies to a variety of cellular antigens. The detection and serological characterisation of specific autoantibodies plays an important role in the differential diagnosis of these diseases. FARO Anti-Ro (SS-A) DIASTAT ELISA (96 wells) 1) Please contact your local representative for availability in your country, available in US! Anti-Sm DIASTAT Sm is a non-histone glycoprotein and antibodies against this extractable nuclear antigen are specific serological markers of systemic lupus erythematosus (SLE). Antibodies against Sm are often positive in SLE-patients in remission. The need for quantitative analysis for monitoring clearly exists. DIASTAT anti-sm test is a quantitative/qualitative enzyme-linked immunosorbent assay (ELISA) for the detection of autoantibodies specific for Sm antigen in human serum or EDTA, lithium heparin, citrated plasma. Autoantibody levels represent one parameter in a multi-criterion diagnostic process. FASM Anti-Sm DIASTAT ELISA (96 wells) 1) Please contact your local representative for availability in your country, available in US! Visit our website 34 I product catalog
35 Anti-Sm/RNP DIASTAT Antibodies against the U1-RNP antigen are found in several connective tissue diseases, principally systemic lupus erythematosus (SLE) and mixed connective tissue disease (MCTD). High titres of anti-rnp antibodies are considered diagnostic of MCTD. In addition, a number of reports have noted the variation in anti-rnp titre within the course of SLE. FRNP Anti-Sm/RNP DIASTAT ELISA (96 wells) 1) Please contact your local representative for availability in your country, available in US! ENA single-well screen DIASTAT Detection and serological characterisation of specific auto-antibodies plays an important role in the differential diagnosis of systemic rheumatic diseases. DIASTAT ENA single-well screen kit allows the detection of six different autoantibodies, namely Sm, Sm/RNP, Ro (SS-A), La (SS-B), Scl-70 and Jo-1. DIASTAT ENA single-well screen is a qualitative enzyme-linked immunosorbent assay (ELISA) for the simultaneous detection of the IgG class of autoantibodies specific for Sm, RNP, Ro (SS-A), La (SS-B), Scl-70 and Jo-1 in human serum or plasma. FSWS ENA single-well screen DIASTAT ELISA (96 wells) 1) Please contact your local representative for availability in your country, available in US! ENA profile DIASTAT DIASTAT ENA Profile kit allows the detection of four different auto antibodies, namely Sm, Sm/RNP, Ro (SS-A) and La (SS-B). Antibodies against Sm are specific serological markers of systemic lupus erythematosus (SLE), while antibodies against Sm/RNP are found in several connective tissue diseases, principally SLE and MCTD. Ro (SS-A) and La (SS-B) are RNA-protein complexes and antibodies against them are found in Sjögren s syndrome, SLE, scleroderma, and neonatal lupus erythematosus. FAID ENA profile DIASTAT ELISA (96 wells) 1) Please contact your local representative for availability in your country, available in US! product catalog I 35
36 Congenital heart block Congenital heart block Disease information CHB a rare but serious autoimmune cardiac disease Autoantibodies directed against SS-A/Ro proteins occur in 60-70% of patients with primary Sjögrens syndrome (SS) and 40-50% of systemic lupus erythematosus (SLE). Neonatal lupus erythematosus develops in fetuses of mothers positive for the autoantibodies Ro/SS-A and La/ SS-B. These mothers are usually diagnosed with Sjögren s or SLE but may be clinically asymptomatic. It has been observed that mothers with rheumatic diseases and with antibodies to SS-A/Ro are at risk of having children with neonatal lupus erythematosus. Transfer of maternal autoantibodies to the fetus has been implicated in causing the disease. Neonatal lupus syndrome includes dermatological, liver and hematopoetic manifestations that are usually transient and disappear when the maternal autoantibodies are cleared from the circulation of the child. A severe, potentially fatal complication is complete congenital heart block (CHB). This third degree atrio-ventricular (AV) block is a rare disorder with an incidence of 1/ / pregnancies. In women with SS-A/ Ro autoantibodies, the incidence is considerably higher, up to 2-5%. The mortality is high, 15-30% of fetuses with CHB die. Children that survive most often need lifelong pacemaker support. Although CHB is a rare condition, it is important to identify mothers that have an increased risk. The heart block usually develops during gestation week An established third-degree heart block is irreversible. It has been suggested that steroid treatment initiated before week 16 may prevent the progression of a first degree heart block to second or third degree block. Studies have shown that maternal autoantibodies directed against the SS-A/Ro52-protein, rather than the SS-A/Ro60-protein, serve as markers for increased risk of developing CHB. More specifically, there is a strong link between autoantibodies directed against a subregion of the SS-A/Ro52- protein, residues called SS-A p200, and the disruption in heart rate. Experimental studies show that antibodies to SS-A p200 can mediate CHB. Code Name Page SSA p200 2 Anti-SS-A p200 Wieslab 37 2) Please contact your local representative for availability in your country, not available in US! Visit our website 36 I product catalog
37 Congenital heart block Products Anti-SS-A p200 Wieslab Mothers with rheumatic diseases and with antibodies against SS-A/Ro have a heightened risk of bearing children with neonatal lupus erythematosus. More specifically, it has been observed that maternal antibodies against a subregion of SS-A/Ro52, amino acids (p200), are linked to an increased risk. Congenital complete atrio-ventricular heart block, third degree, is a serious and potentially fatal complication. Positive findings in this test therefore support further investigations of the heart status of the foetus. Wieslab SS-A p200 test kit is an enzyme-linked immunosorbent assay (ELISA) for the qualitative detection of autoantibodies against SS-A p200 in human serum. The assay is used to detect antibodies in a single serum specimen. Results are used in combination with other diagnostic methods to aid finding pregnant women at risk of having children with Congenital Heart Block. The analysis should be performed by trained laboratory professionals. SSA p200 2 Anti-SS-A p200 Wieslab ELISA (96 wells) 2) Please contact your local representative for availability in your country, not available in US! product catalog I 37
38 LEADER IN AUTOIMMUNITY TESTING The 1st to develop, manufacture and market top quality ELISA ANCA, anti-ccp and Complement kits Visit our website 38 I product catalog
39 Anti-phospholipid syndrome Disease information Anti-phospholipid syndrome An autoimmune disorder Anti-phospholipid syndrome (APS) is an autoimmune disorder that is characterised by thrombosis in arteries and veins as well as pregnancy-related/ fetal loss complications and the presence of anti -phospholipid antibodies. Like many autoimmune diseases, APS is more common in women than in men. The disease is most common in young to middle-aged adults; however, it also manifests in children and elderly people. The actual frequency of APS in the general population is unknown. One to 5% of healthy individuals have apl antibodies. They are also found in approximately 30-40% of patients with SLE, but only about 10% have APS. Approximately half of APS cases are not associated with another rheumatic disease. Three primary classes of antibodies The exact cause of APS is not known, but activation of the coagulation system is evident. There are three primary classes of antibodies associated with the disease, including anti-cardiolipin antibodies, the lupus anticoagulant, and antibodies directed against beta-2-glycoprotein 1. Anti-phospholipid syndrome - symptoms The most common clinical manifestation of anti-phospholipid syndrome is thrombosis, which can affect the vessels of any organ. Damage depends on the extent and location of the clot. There are three distinct APS disease entities: primary (the absence of any comorbidity), secondary (when there is a pre-existing autoimmune condition, most frequently systemic lupus erythematosus, SLE), and catastrophic (when there is simultaneous multi-organ failure with small vessel occlusion). Catastrophic APS (CAPS) is often a fatal manifestation with a mortality rate of approximately 50%. Code Name Page FBGP Anti-beta-2-glycoprotein 1 IgG DIASTAT 40 FBGP 100M 1 Anti-beta-2-glycoprotein 1 IgM DIASTAT 40 FCAR 100G 1 Anti-Cardiolipin IgG DIASTAT 41 FCAR 100M 1 Anti-Cardiolipin IgM DIASTAT 41 FCAR 100T 1 Anti-Cardiolipin Total DIASTAT 41 1) Please contact your local representative for availability in your country, available in US! product catalog I 39
40 ANTI-PHOSPHOLIPID SYNDROME Anti-phospholipid syndrome Products Anti-beta-2-glycoprotein 1 IgG and IgM DIASTAT A number of studies have demonstrated that detection of Beta-2GP1 antibodies, in conjunction with anti-cardiolipin measurement, is important in defining the thrombotic risk associated with APAs. DIASTAT anti-beta-2-glycoprotein 1 IgG and IgM tests are quantitative/ qualitative enzyme-linked immunosorbent assays (ELISA) for the detection of the IgG or IgM class of autoantibodies respectively, specific for beta-2gp1 antigen in human serum or plasma. Both are intended to aid the assessment of thrombotic risk in patients with autoimmune disease associated with thrombotic disorders such as primary anti-phospholipid syndrome or secondary to systemic lupus erythematosus. Neither are definitive in isolation. Autoantibody levels represent one parameter in a multi-criterion diagnostic process. Beta-2-glycoprotein 1 (Beta-2GP1) is a plasma glycoprotein and has been shown to be the dominant antigen for anti-phospholipid antibodies (APAs) in patients with anti-phospholipid syndrome. DIASTAT Anti-b2-glycoprotein 1 IgM (b2-gp1 IgM) test is a semi-quantitative enzyme-linked immunosorbent assay (ELISA) for the detection of the IgM class of autoantibodies specific for b2-gp1 antigen in human serum or plasma. It is intended to aid the assessment of thrombotic risk in patients with autoimmune disease associated with thrombotic disorders such as primary anti-phospholipid syndrome or secondary to systemic lupus erythematosus. It is not definitive in isolation. FBGP Anti-beta-2-glycoprotein 1 IgG DIASTAT ELISA (96 wells) FBGP 100M 1 Anti-beta-2-glycoprotein 1 IgM DIASTAT ELISA (96 wells) 1) Please contact your local representative for availability in your country, available in US! Visit our website 40 I product catalog
41 Anti-Cardiolipin IgG and IgM DIASTAT Anti-cardiolipin antibodies are detected in 80% of patients with anti-phospholipid syndrome (APS). They are also associated with a diverse set of clinical manifestations including neurological dys function, venous or arterial thrombosis, thrombocytopaenia and recurrent foetal loss. DIASTAT anti-cardiolipin tests are a quantitative/qualitative ELISA for the detection of IgG and IgM autoantibodies specific for the phospholipid cardiolipin in human serum and EDTA, lithium heparin or citrated plasma. FCAR 100G 1 Anti-Cardiolipin IgG DIASTAT ELISA (96 wells) FCAR 100M 1 Anti-Cardiolipin IgM DIASTAT ELISA (96 wells) FCAR 100T 1 Anti-Cardiolipin Total DIASTAT ELISA (96 wells) 1) Please contact your local representative for availability in your country, available in US! Consequences for treatment & prognosis Thrombosis Diagnosis and/or PLUS Persistently positive test for LAC and/or acl and/or anti-beta-2-gpi APS Recurrent pregnancy loss Diagnosis product catalog I 41
42 GASTROENTEROLOGY Autoimmune Gastroenterology Disease information Inflammatory Bowel Disease and Irritable Bowel Syndrome Gastrointestinal problems are common in a number of different systemic autoimmune diseases such as SLE. The common clinical signs may also be attributed to a primary gastrointestinal autoimmune disease. When discussing autoimmune gastrointestinal diseases, they are often divided in two general groups, IBD (Inflammatory Bowel Disease) and IBS (Irritable Bowel Syndrome). Celiac disease is often included as well. Although IBD and IBS are two very different conditions, they have many clinical signs in common so a differential diagnosis is important. Irritable bowel syndrome (IBS) is one of the most common ailments of the bowel (intestines). IBS is not an autoimmune disease but is best described as a functional disease and is believed to be due to the abnormal function (dysfunction) of the muscles in the organs of the gastrointestinal tract or of the nerves controlling the organs. The most commonly affected function in these diseases is transportation. In the stomach and small intestine, the symptoms of slowed transportation are nausea, vomiting, abdominal bloating, and abdominal distention (enlargement). The symptom of rapid transportation is usually diarrhea. Inflammatory Bowel Diseases (IBD) is a broad term that describes autoimmune conditions with chronic or recurring immune response and inflammation of the gastrointestinal tract. The two most common inflammatory bowel diseases are Crohn s disease and ulcerative colitis. Crohn s disease is a condition of chronic inflammation potentially involving any location of the gastrointestinal tract, but frequently affecting the end of the small bowel and the beginning of the large. Symptoms include persistent diarrhea, cramping abdominal pain, fever, and sometimes rectal bleeding. Loss of appetite and weight loss also may occur. However, the disease is not always limited to the gastrointestinal tract; it can also affect the joints, eyes, skin, and liver. Fatigue is a common complaint. Ulcerative colitis is a chronic gastrointestinal disorder that is limited to the large bowel (colon). It does not affect all layers of the bowel, only the top layers of the colon in an even and continuous distribution. The first symptom of ulcerative colitis is a progressive loosening of the stool. The stool is generally bloody and may be associated with cramping abdominal pain. Loss of appetite and subsequent weight loss are common, as is fatigue. Celiac disease is a chronic autoimmune digestive disorder in which damage to the lining of the small intestine leads to the malabsorption of minerals and nutrients. The destruction of the inner lining of the small intestine in celiac disease is caused by an immunological reaction to gluten present in wheat, barley and rye. Individuals with celiac disease may develop diarrhea, steatorrhea, weight loss, flatulence, iron deficiency anemia, abnormal bleeding, or weakened bones. However, many adults with celiac disease may have only vague abdominal discomfort such as bloating, abdominal distension, and excess gas. Adults with celiac disease have a several-fold higher than normal risk of developing lymphomas. Code Name Page ASCA ASCA IgA Wieslab 43 ASCA ASCA IgG Wieslab 43 2) Please contact your local representative for availability in your country, not available in US! Visit our website 42 I product catalog
43 Autoimmune Gastroenterology Products ASCA IgA/ASCA IgG Wieslab Wieslab ASCA tests are enzyme-linked immunosorbent assays (ELISA) for semi-quantitative/qualitative detection of IgA or IgG autoantibodies against the mannan structure of Saccharomyces cerevisiae in human sera. The assay is used to detect antibodies in a single serum specimen. Its results aid the diagnosis of Crohn s disease as well as its differentiation from ulcerative colitis. Forty patient samples can be tested in duplicate quantitatively or 45 patient samples qualitatively. The test is completed within two hours. The Wieslab ASCA test kit is an enzyme-linked immunosorbent assay (ELISA) for detection and semi-quantitation of IgA and/or IgG antibodies to mannan of Saccharomyces cerevisiae in human sera. The assay is used to detect antibodies in a single serum specimen. Results are used as an aid to diagnosing Crohn s disease. The combination of positive ASCA with a negative panca has a high positive predictive value and specificity for Chron s disease. ASCA ASCA IgA Wieslab Break-apart microtitration strips (12x8) 96 wells ASCA ASCA IgG Wieslab Break-apart microtitration strips (12x8) 96 wells 2) Please contact your local representative for availability in your country, not available in US! Ulcerative colitis: Multifactorial cause Diet Drugs Infections Stress Allergens Genes ALTERED IMMUNITY Ulcerative colitis product catalog I 43
44 THYROID DIAGNOSTICS Thyroid diagnostics Disease information The two most prevalent autoimmune thyroid diseases are Grave s disease and Hashimoto s disease The thyroid gland, one of the largest endocrine glands, is found in the neck below the thyroid cartilage which forms the Adam s apple. It is involved in a number of processes such as energy metabolism, protein synthesis and endocrine processes. It participates in these processes by producing thyroid hormones, the principal ones being triiodothyronine (T3) and thyroxine, sometimes referred to as tetraiodothyronine (T4). These hormones regulate the rate of metabolism and affect the growth and rate of function of many other systems in the body. The thyroid also produces calcitonin, which plays a role in calcium homeostasis. Diseases of the thyroid include hyperthyroidism (abnormally increased activity), hypothyroidism (abnormally decreased activity) and thyroid nodules, which are generally benign thyroid neoplasms, but may be thyroid cancers. All these disorders may give rise to goiter, i.e. an enlarged thyroid. Autoimmune thyroid diseases are generally hereditary in origin, although environmental factors, such as infection, certain drugs, and iodine consumption, can play a role in their progression. Autoimmune thyroiditis primarily affects people between 40 and 60 years of age. Like many autoimmune disorders, an individual s chances of contracting autoimmune thyroiditis are increased if they have other autoimmune disorders. The two most prevalent autoimmune thyroid diseases are Hashimoto s disease and Graves disease. Hashimoto s thyroiditis is the most common cause of hypothyroidism and is caused by inflammation of the thyroid gland. The disease tends to run in families, and is associated with a clustering of other autoimmune conditions such as Type 1 diabetes, and celiac disease. Hashimoto s thyroiditis is 5-10 times more common in women than in men and most often starts in adulthood. Graves disease, the most common cause of hyperthyroidism, is an autoimmune thyroid condition that results from abnormal stimulation of the thyroid gland. The cause of Graves disease is thought to be related to many factors including genes, gender, stress, pregnancy, and possibly infections. Graves disease affects both men and women; however, women are affected about 8-10 times more often than men. Risk factors for Graves disease are associated with other auto immune diseases such as rheumatoid arthritis, Addison s disease, type 1 diabetes, pernicious anemia, and SLE. Code Name Page FATG Anti-Tg DIASTAT 45 FTPO Anti-TPO DIASTAT 45 1) Please contact your local representative for availability in your country, available in US! Visit our website 44 I product catalog
45 Thyroid diagnostics Products Anti-Tg DIASTAT Thyroglobulin (Tg) is, together with thyroid peroxidase (TPO), considered the major autoantigen associated with chronic autoimmune thyroiditis. DIASTAT anti-thyroglobulin (anti-tg) test is a quantitative/qualitative enzyme-linked immunosorbent assay (ELISA) for the detection of autoantibodies specific for thyroglobulin in human serum or EDTA, Li-Heparin or citrated plasma. It aids the diagnosis of autoimmune thyroid disorders but is not definitive in isolation. Autoantibody levels represent one parameter in a multi-criterion diagnostic process. Thyroglobulin (Tg) and thyroid peroxidase (TPO) are considered the major autoantigens associated with chronic autoimmune thyroiditis. Clinical diagnosis is usually based on the presence of serum or plasma autoantibodies to Tg and TPO. Thyroglobulin, a 660 kd water-soluble glycoprotein, is the precursor of the thyroid hormones tri iodothyronine and thyroxine. The presence of anti-tg autoantibodies in patients with Hashimoto s thyroiditis was first demonstrated in FATG Anti-Tg DIASTAT ELISA (96 wells) 1) Please contact your local representative for availability in your country, available in US! Anti-TPO DIASTAT Thyroid peroxidase (TPO) is a membrane-bound glycoprotein enzyme and TPO autoantibodies may play a pathogenic role in destructive autoimmune thyroid diseases. Anti-TPO antibodies are found, often in conjunction with anti-thyroglobulin autoantibodies, in the majority of Hashimoto s thyroiditis and primary myxedema. DIASTAT Anti-Thyroid Peroxidase (anti-tpo) test is a quantitative/ qualitative enzyme-linked immunosorbent assay (ELISA) for detecting the IgG class of autoantibody specific for thyroid peroxidase in human serum or EDTA, lithium heparin, and sodium citrate plasma. It aids the diagnosis of autoimmune thyroid disorders but is not definitive in isolation. Autoanti body levels represent one parameter in a multi-criterion diagnostic process. FTPO Anti-TPO DIASTAT ELISA (96 wells) 1) Please contact your local representative for availability in your country, available in US! product catalog I 45
46 ENDOCRINE MARKERS Endocrine markers Disease information Peptide hormones The amount of hormones produced by each gland is carefully balanced to generate needed effects. Too much or too little of a certain hormone can have effects throughout the body and cause various endocrine disorders. In addition to the classical endocrine glands, a number of tissues also secrete hormones. Examples are the intestines, kidneys, liver, stomach and the heart. Peptide hormones are involved in a great number of different physiological functions. Each hormone is often involved in several different physiological processes. An example is Angiotensin II, which is part of the renin-angiotensin system, a system involved regulating blood pressure through vasoconstriction, but which also effects renal functions, inducing thirst sensation. Another example is Vasointestinal peptide (VIP) which is produced in several tissues and has a multitude of functions, e.g. affecting the digestive system. It is a key component of the mammalian circadian timekeeping machinery, causes vasodilation, increases glycogenolysis, and lowers arterial blood pressure. Some forms of tumors, called VIPomas, secrete excessive amounts of VIP that cause Verner Morrison syndrome, also called the WDHA-syndrome, characterized by profuse and chronic watery diarrhea. Like VIPoma, many peptide hormones may be excessively secreted by a form of tumors categorized as neuroendocrine tumors (NETs). One important category of NETs is the neuroendocrine tumors of the gastro-entero-pancreatic (GEP) system. These tumors originate from the diffuse endocrine system located in the gastrointestinal tract (carcinoids) and in the pancreas (insular tumors). In most cases of NETs, in particular carcinoids, Chromogranin A is cosecreted by these tumors. Code Name Page RB Angiotensin II EURIA 47 BANI 1 3 ANP EURIA 47 RB CCK EURIA 48 CGA 2 Chromogranin A NEOLISA 48 RB Chromogranin A EURIA 49 RB Chromogranin B EURIA 49 RB Beta-Endorphin EURIA 50 RB Endothelin EURIA 50 MD /MD 302 US 1 Gastrin EURIA 51 RB /RB 310 US 1 Glucagon EURIA 51 RB Alpha-MSH EURIA 52 RB NPY EURIA 52 RB PP EURIA 53 RB Somatostatin EURIA 53 RB Vasopressin EURIA 54 RB VIP EURIA 54 1) Please contact your local representative for availability in your country, available in US! 2) Please contact your local representative for availability in your country, not available in US! 3) For research use only, contact your local representative for research use availability in your country. Visit our website 46 I product catalog
47 Endocrine markers Products Angiotensin II EURIA Angiotensin II is a biologically active key compound of the reninangiotensin system. The octapeptide angiotensin II is the strongest physiological vasoconstrictor known. Euro Diagnostica angiotensin II kit contains reagents and instructions for measuring angiotensin II in plasma. After ethanol extraction of the plasma samples, angiotensin II is assayed by a competitive radioimmunoassay. The assay is specific and the cross-reactivity with Angiotensin I is very low (<0.1%). Sensitivity determined as 3 standard deviations change from zero calibrator is 2.0 pmol/l. RB Angiotensin II EURIA RIA (100 tubes) 2) Please contact your local representative for availability in your country, not available in US! ANP EURIA It has been shown that alpha-anp causes diuretic and natriuretic activities and relaxes smooth muscle. These effects are thought to be mediated by circulation in peripheral blood. EURIA-ANP is a simple, sensitive and reliable radioimmunoassay for ANP in human plasma. Euro Diagnostica ANP kit contains reagents and instructions for quantitative measurement in plasma samples. After extraction with Sep-pak C18, concentrations are measured by non-equilibrium radioimmunoassay with delayed addition of the iodinated tracer. The determined concentration of ANP is expressed in picograms/ml (pg/ml). The sensitivity of the assay, judged as 3 SD change from zero calibrator is 3.5 pg/ml. BANI 1 3 ANP EURIA RIA (100 tubes) 3) For research use only, contact your local representative for research use availability in your country. product catalog I 47
48 ENDOCRINE MARKERS Endocrine markers Products CCK EURIA Cholecystokinin (CCK) is one of the classical gut hormones. It is believed to be a major regulator of gall bladder contraction and pancreatic enzyme secretion. CCK occurs in many different molecular forms. The intended use of these reagents is for determination of CCK in plasma. CCK is extracted from plasma by an ethanol extraction method. The C-terminal sulphation and the C-terminal amidation are important for the biological activity of CCK. CCK shares an identical sequence with gastrin in the five C-terminal amino acids, which requires an antiserum with very low cross-reactivity to gastrin. Plasma concentrations of CCK are very low, which makes it necessary to have a highly sensitive assay system. EURIA CCK radioimmunoassay is based on an antiserum specific for the sulphated form of CCK, with very low cross-reactivity to gastrin-17, sulphated gastrin. The assay system has been optimized to a very high sensitivity of 0.3 pmol/l Specificity The following cross-reactions have been found: Peptide Cholecystokinin 26-33, sulphate 100.0% Cholecystokinin-33, sulphate 134.0% Cross-reaction Cholecystokinin 26-33, non-sulphated < 0.01% Cholecystokinin < 0.01% Gastrin-17, sulphate 0.5% Gastrin-17, non-sulphated < 0.01% RB CCK EURIA RIA (100 tubes) 2) Please contact your local representative for availability in your country, not available in US! Chromogranin A NEOLISA Tumours of neuroendocrine origin such as carcinoids usually present with increased levels of chromogranin A in serum and plasma. Chromogranin A has been demonstrated to be a good circulating marker for these tumours. NEOLISA Chromogranin A is an enzyme-linked immunosorbent assay (ELISA) for accurate detection and quantitation of chromogranin A in human plasma or serum. The calibrator in the kit is a synthetic peptide corresponding to chromogranin A. The levels of the calibrator are fixed from lot to lot and the limit of detection is 0.54 nmol/l. The assay is divided into three incubation steps with an assay time of minutes. Two controls are included in the kit and the microtitre plate is in break-apart format, allowing for maximum test flexibility. Parameter Neolisa Provider X Provider Y Provider Z Incubation (steps) 105 (3) 135 (2) 250 (3) 200 (3) Shaking No Yes Yes Yes Controls H+L 1 H+L 1 Cal Dilution Lyo RTU Lyo Lyo Calibrators (value) Fixed Lot Lot Lot Strips/plate Break Strips Strips Strips Samples P+S P P+S S Washes CGA 2 Chromogranin A NEOLISA ELISA (96 wells) 2) Please contact your local representative for availability in your country, not available in US! Visit our website 48 I product catalog
49 Chromogranin A EURIA Tumours of neuroendocrine origin such as carcinoids usually present with increased levels of chromogranin A in serum and plasma. Neuroendocrine tumours are derived from the neuroendocrine cells and typically include carcinoid tumours, pheochromocytomas, neuroblastomas, small cell lung cancers, hyperparathyroid adenomas, pituitary tumours, prostate cancers and pancreatic islet tumours, including MEN1 and MEN2 syndromes. The present chromogranin A RIA is a competitive method based on polyclonal rabbit antibodies. The antibodies were raised against a purified fragment containing amino acid sequence of the chromogranin A molecule. The sensitivity of the assay, judged as 2 SD change from zero calibrator, is 0.15 nmol/l. The assay kit includes two controls (high and low). Tumour Blood tests chromogranins Tumour secretes chromogranins into the blood RB Chromogranin A EURIA RIA (100 tubes) 2) Please contact your local representative for availability in your country, not available in US! Chromogranin B EURIA Chromogranins are soluble secretory proteins which are stored in the majority of different neuroendocrine cells. Increased levels in the circulation are often seen in conjunction with various neuroendocrine tumours. However, chromogranin A levels are often elevated in response to decreased renal function, atrophic gastritis and treatment with proton pump inhibitors. Chromogranin B levels are not affected by either proton pump inhibitor treatment or decreased renal function and have been shown to have similar use as chromogranin A. With its lower sensitivity but very high specificity, chromgranin B measurements can serve as a valuable complement to chromogranin A. EURIA Chromogranin B kit is a competitive radioimunoassay (RIA) for determination of chromogranin B in human plasma and serum. The antibodies were raised against a synthetic peptide containing amino acid sequence in the chromogranin B molecule. RB Chromogranin B EURIA RIA (100 tubes) 2) Please contact your local representative for availability in your country, not available in US! product catalog I 49
50 ENDOCRINE MARKERS Endocrine markers Products Beta-Endorphin EURIA ß-endorphin is found in neurons of the hypothalamus, as well as the pituitary gland. The behavioural effects of ß-endorphin are exerted by its actions in the brain and spinal cord, and it is presumed that the hypothalamic neurons are the major source of ß-endorphin at these sites. In situations where the level of ACTH is increased (e.g., Cushing s Syndrome), the level of endorphins also increases. ß-endorphin in sample (plasma, cerebrospinal fluid or tissue) is extracted using Sep-pak C18 cartridges. The extracts are analysed by a competitive radioimmunoassay using antibodies against synthetic human ß-endorphin. Sensitivity calculated from a decrease in binding of 2 SD in the zero standard is 3 pmol/l. RB Beta-Endorphin EURIA RIA (100 tubes) 3) For research use only, contact your local representative for research use availability in your country. Endothelin EURIA Endothelin is synthetized in and released from endothelial cells. Endothelin occurs in three different molecular forms: Endothelin 1, 2 and 3. Endothelin 1 is a potent vasoconstrictor peptide. Endothelin occurs in the circulation in very low concentrations and may be an important factor in the regulation of local blood circulation, blood pressure and contractility of the heart. Endothelin is extracted from plasma using Sep-pak C-18 columns or similar. The extracts are assayed by a competitive radioimmunoassay using a rabbit antiserum raised against an endothelin-1 albumin conjugate. The antiserum used in this assay cross-reacts with endothelin-1 (100%), endothelin-2 (48%) and endothelin-3 (109%). The lowest detectable concentration is 4 pmol/l corresponding to 0.4 pmol/l in the sample. RB Endothelin EURIA RIA (100 tubes) 3) For research use only, contact your local representative for research use availability in your country. Visit our website 50 I product catalog
51 Gastrin EURIA Gastrin is involved in a number of physiological processes in the GI tract such as stimulating secretion of gastric acid (HCl) by the parietal cells of the stomach and inducing pancreatic secretions and gallbladder emptying. Determining gastrin is useful when diagnosing gastrin-producing tumours and achylia with or without pernicious anemia. In Zollinger- Ellison syndrome, gastrin is produced at excessive levels, often by a gastrinoma. Treatment with powerful anti-secretagogues may cause a rise in the serum gastrin concentration, and measurement of serum gastrin can thus be used to monitor the treatment efficacy. Gastrin occurs in the circulation in several different forms, among those gastrin-34 and gastrin-17, sulphated and non-sulphated. The five C-terminal amino acids of gastrin and cholecystokinin are identical, which explains their overlapping biological effects. The antiserum used in this assay completely cross-reacts with gastrin-34 and the sulphated forms of gastrin-17 and gastrin-34, but much less with CCK-8. Specificity The following cross-reactions have been found: Compound Gastrin % Gastrin-17, sulphated 83% Gastrin-34 61% CCK-8 36% Gastrin 1-14 < 0.1% Cross-reaction Gastric releasing peptide < 0.01% Vascoactive intestinal peptide < 0.01% Motilin < 0.01% Glucagon < 0.01% Somatostatin 14 < 0.01% C-peptide < 0.01% MD /MD 302 US 1 Gastrin EURIA RIA (100 tubes) 1) Please contact your local representative for availability in your country, available in US! 2) Please contact your local representative for availability in your country, not available in US! Glucagon EURIA Glucagon is involved in carbohydrate, fat and protein metabolism. Basal amounts of glucagon are essential for the maintenance of normoglycemia and one physiological role for glucagon is to prevent hypoglycemia. Since glucagon in diabetics has been found elevated absolutely or relatively to insulin, it has been proposed that glucagon contributes essentially to the development of the hyperglycemia and keto acidosis found in diabetes. Elevated levels of glucagon in plasma are found in patients with A-cell tumors. (Glucagon is a 29-amino acid straight-chain peptide produced in the pancreatic alpha cells.) EURIA radioimmuno assay is specific for pancreatic glucagon with no cross-reactivity with gut GLI (Glucagon-Like-Immunoreactivity). The sensitivity of the assay, judged as 2 SD change from zero calibrator, is 3 pmol/l. Specificity The following cross-reactions have been found: Peptide Glucagon, pancreatic, human 100.0% Gut GLI < 0.1% Cross-reaction Secretin < 0.02% Cholecystokinin -39 < 0.02% Vasoactive intestinal peptide < 0.02% Gastric inhibitory peptide < 0.02% RB /RB 310 US 1 Glucagon EURIA RIA (100 tubes) 1) Please contact your local representative for availability in your country, available in US! 2) Please contact your local representative for availability in your country, not available in US! product catalog I 51
52 ENDOCRINE MARKERS Endocrine markers Products Alpha-MSH EURIA Alpha-Melanocyte Stimulating Hormone (Alpha-MSH) is a potent modulator of fever and inflammation. The average plasma Alpha- MSH level has been found higher in subjects with AIDS than in control subjects. Alpha-MSH is derived from pro-opiomelanocortin, a precursor protein that contains, within its structure, the sequences of other melanotropic peptides. Alpha-MSH is a 13-amino acid peptide with identical sequence to adrenocorticotropic hormone (ACTH 1-13). The intended use of the EURIA radioimmuno assay kit is the determination of Alpha-MSH in human plasma or cerebrospinal fluid. The antiserum used in this kit is directed to the amidated C-terminal part of the Alpha-MSH molecule and shows no cross-reactivity to ACTH (Adreno-Corticotropic-Hormone). Sensitivity calculated from a decrease in binding of 2 SD in the zero standard is 3 pmol/l. RB Alpha-MSH EURIA RIA (100 tubes) 3) For research use only, contact your local representative for research use availability in your country. NPY EURIA Neuropeptide Y (NPY) is a 36-amino acid peptide hormone found in the brain and autonomic nervous system. The hormone has been shown to be involved in several physiological/clinical processes including food intake, obesity, and anorexia nervosa. Increased serum/plasma concentrations of NPY have been found in patients with neuroblastoma and phaeochromocytoma. Increased serum/ plasma concentrations of NPY have also been found in pediatric B-cell precursor leukemia. The intended use of this EURIA kit is determination of NPY in human serum/plasma by direct assay without extraction. The antiserum used is very specific for NPY with no cross-reactivity with either pancreatic polypeptide (PP) or peptide YY (PYY). Sensitivity calculated from a decrease in binding of 2 SD in the zero standard is 3 pmol/l. RB NPY EURIA RIA (100 tubes) 2) Please contact your local representative for availability in your country, not available in US! Visit our website 52 I product catalog
53 PP EURIA Pancreatic polypeptide (PP) is secreted by the F-cells of the islets of Langerhans in the pancreas. PP is localized almost entirely in the pancreas, although detectable levels throughout gastrointestinal tract have been reported. The secretion of PP is stimulated by intake of food, especially protein and fat. Its function is to self-regulate pancreatic secretion activities. PP also effects hepatic glycogen levels and gastrointestinal secretions. It is also produced by endocrine-active tumours in the pancreas and the gastrointestinal tract. These tumours often produce several peptide hormones in the combinations PP-VIP, PP-glucagon or PP-gastrin. Tumours with only PP-secretion have been reported. These tumours may occur at the WDHA or Verner- Morrison syndrome. EURIA PP kit is intended for determining PP in human serum. Serum PP is assayed without extraction by a competitive radio immunoassay using a rabbit antiserum raised against bovine PP. The assay is specific for PP with very low cross-reactivity to a number of other functionally-related peptide hormones. Sensitivity calculated from a decrease in binding of 2 SD in the zero standard is 3 pmol/l. RB PP EURIA RIA (100 tubes) 2) Please contact your local representative for availability in your country, not available in US! Somatostatin EURIA Somatostatin is a cyclic peptide hormone secreted in several locations of the digestive system such as the stomach, intestine and delta cells of the pancreas. It is also secreted in the brain (e.g. hypothalamus). Functionally, somatostatin is an inhibitor hormone, inhibiting release of other hormones such as growth hormone, TSH, glucagon and gastrin. Somatostatin in plasma is extracted with Sep-pak C18 cartridges. The extracts are analysed by a competitive radioimmunoassay using an antiserum to synthetic cyclic somatostatin 14. The sensitivity calculated from a decrease in binding of 2 SD in the zero standard is 6 pmol/l. RB Somatostatin EURIA RIA (100 tubes) 3) For research use only, contact your local representative for research use availability in your country. product catalog I 53
54 ENDOCRINE MARKERS Endocrine markers Products Vasopressin EURIA One of the most important roles of vasopressin or antidiuretic hormone (ADH) is to regulate the body s retention of water. In addition, vasopressin has been implicated in a variety of physiological functions and has clinical applications in diabetes, hyponatraemia, and hypertension studies. Vasopressin, or Antidiuretic Hormone (ADH), is a cyclic nanopeptide with a structure very similar to that of oxytocin, differing in only two amino acids. Euro Diagnostica vasopressin kit contains reagents and instructions for the quantitative measurement of vasopressin in plasma or urine. After solid-phase extraction (SPE) or ethanol extraction, plasma vasopressin concentrations are measured by radioimmunoassay (RIA). Urine vasopressin concentrations can be measured directly. The assay is specific for vasopressin with no cross-reactivity to oxytocin. It has been calibrated against WHO standard 77/501. Sensitivity calculated from a decrease in binding of 3 SD in the zero standard is 0.5 pmol/l RB Vasopressin EURIA RIA (100 tubes) 2) Please contact your local representative for availability in your country, not available in US! VIP EURIA Vasoactive intestinal peptide (VIP) is produced in many tissues including the gut, pancreas and hypothalamus. It is a linear polypeptide structurally related to secretin and other members of the secretin family. VIP is believed to play crucial roles in the regulation of intestinal motility and intestinal epithelial ion and water transport. Increased plasma immunoreactive VIP concentrations have been reported in patients with WDHA syndrome (water, diarrhea, hypokalemia and achlor hydria). Increased plasma levels have also been reported in patients with cirrhosis. EURIA VIP kit is used to determine VIP in human plasma by a competitive radioimmunoassay without extraction. The assay is specific for VIP with very low cross-reactivity to other members of the secretin family. Its sensitivity calculated from a decrease in binding of 2 SD in the zero standard is 3 pmol/l. RB VIP EURIA RIA (100 tubes) 2) Please contact your local representative for availability in your country, not available in US! Specificity The following cross-reactions have been found: Parameter VIP 1-28 (whole sequence) 100% VIP 1-6 < 2.5% VIP 1-18 < 2.5% VIP 1-22 < 2.5% VIP % VIP % VIP % Cross-reaction Secretin, porcine < 0.01% Gastric inhibitory peptide, porcine < 0.01% Pancreatic gluacon, porcine < 0.01% Enteroglucagon, porcine < 0.01% Pancreatic polypeptide, human < 0.01% Substance P < 0.01% Somatostatine, ovine < 0.01% Visit our website 54 I product catalog
55 Pertussis Disease information Pertussis or whooping cough is a serious disease caused by the Gram-negative bacteria Bordetella pertussis Pertussis is distributed all over the world with millions of new cases annually and a mortality of almost 300,000 per year. The disease is especially serious in small children and infants where the mortality rate may be rather high. Moreover, pertussis is prevalent in developing countries and mortality is high. The classic symptoms of pertussis are a paroxysmal cough, inspiratory whoop, and vomiting after coughing. The incubation period is between seven and ten days in infants or young children, followed by a period of usually mild respiratory symptoms such as mild coughing, sneezing, or runny nose. After one to two weeks, the coughing classically develops into uncontrollable fits, each with several forceful coughs, followed by a high-pitched whoop sound in younger children, or a gasping sound in older children and adults, as the patient struggles to breathe. This stage usually lasts for two to eight weeks, sometimes even longer. A gradual transition subsequently occurs to the convalescent stage, which usually lasts one to two weeks. This stage is marked by a decrease in paroxysms of coughing, both in frequency and severity, and a cessation of vomiting. Common complications of the disease include pneumonia, encephalopathy, and seizures. Prevention via vaccination is of primary importance as treatment is of little clinical benefit to the person infected. Antibiotics, however, may decrease the duration of infectiousness and are thus recommended in some countries. Pertussis toxin (PT) is of significant importance for the pathogenesis of whooping cough. It is a real exotoxin responsible for many physiological, immunological and pharmacological effects. In addition, the toxin is highly specific for B. pertussis, in contrast to some other exotoxins of the species Bordetella, including Bordetella parapertussis. Filamentous haemagglutinin adhesin (FHA) is an important virulence factor from B. pertussis related to the adhesion and spread by the bacteria through the respiratory tract. Data from the literature suggest that systemic immunization with FHA can provide significant protection against B. pertussis infection in both the lower and upper respiratory tract. Code Name Description Page PERT-TRO 2 Pertusscan TRO Qualitative ELISA 56 PERT-PTG2 1 Pertusscan PT IgG Qualitative ELISA 56 1) Please contact your local representative for availability in your country, available in US! 2) Please contact your local representative for availability in your country, not available in US! Optimal Timing for Diagnostic Testing (weeks) Cough Onset Culture PCR Serology product catalog I 55
56 PERTUSSIS Pertussis Products Pertusscan TRO Pertusscan TRO is an ELISA for the detection of IgA and IgG antibodies against Bordetella pertussis filamentous hemagglutinin (FHA) and pertussis toxin (PT) in serum or plasma specimens. The test is intended for determining current/recent infection and for immunity. Pertusscan TRO is for professional use within a laboratory. IgG antibodies and also often IgA antibodies to FHA and/or PT develop during the active stage of the infection. The IgG will stay at increased levels for years after the infection. IgA antibodies, however, will disappear within four months. Unimmunized children, especially infants, produce more antibodies to PT than to FHA. Elderly people may develop only an IgA response to FHA. An antibody titer against FHA may be seen in individuals subjected to vaccination with whole cell preparations. PERT-TRO 2 Pertusscan TRO 2x 96 wells (FHA and PT) 2) Please contact your local representative for availability in your country, not available in US! Pertusscan PT IgG Pertusscan PT IgG is an enzyme-linked immunosorbent assay (ELISA) for detecting IgG antibodies against Bordetella pertussis toxin (PT) in serum specimens. It is used to determine current/recent infection. Pertusscan PT IgG is intended for professional use within a laboratory. Pertusscan PT IgG is an enzyme linked immunosorbent assay (ELISA) for the detection of IgG antibodies against Bordetella pertussis toxin (PT) in serum specimens. The test is intended for determination of current/ recent infection. Pertussis toxin (PT) is of significant importance for the patho-genesis of whooping cough. It is a real exotoxin responsible for many physiological, immunological and pharmacological effects. In addition, Pertussis toxin is highly specific for B. pertussis in contrast to some other exotoxins of the species Bordetella. Serology with determination of IgG against PT is a valuable diagnostic tool and has proven especially useful for later diagnosis of older children and adults where traditional diagnostic methods such as culture and direct immunofluorescence have limited value. PERT-PTG2 1 Pertusscan PT IgG 96 wells (12x8) 1) Please contact your local representative for availability in your country, available in US! Visit our website 56 I product catalog
57 ALL-IN-ONE DIAGNOSTIC SOLUTION Introducing a total diagnostic solution from a single trusted source product catalog I 57
58 ALL-IN-ONE DIAGNOSTIC SOLUTION All-in-one diagnostic solution A total diagnostic solution - from a single trusted source Empowering you to achieve the correct diagnosis Euro Diagnostica s world renowned kits offer a full range of ELISA tests, included in this Product Catalog and comprising anti-ccp, capture ANCA, and Complement that are harmonised for use on Dynex instruments. Dynex processing systems are widely known for their open and flexible automation, ease of use and accuracy. Dynex processing systems Perfecting accuracy in open and flexible automation Euro Diagnostica total system solution comes with a choice of two Dynex processing systems; Dynex DS2 and Dynex DSX. Dynex DS2 DS2 is a 2-plate ELISA processing system designed for walk-away usage. The result of major progress in robotics and software design, DS2 is reliable, cost-effective, and easy to use and maintain. Impressive automation power in the smallest available footprint. Dynex DSX Dynex DSX offers flexible and reliable sample-in/result-out processing and is designed for true walk-away automation. Its fully-automated 4-plate processing system performs multiple assays per plate. DSX s modular design provides flexible configuration and ease of use. It incorporates many features to ensure the quality and accuracy of results, and has the performance to handle a wide variety of assays. Visit our website 58 I product catalog
59 DS2 Specifications DSX Specifications Dimensions Metric Non-Metric Width: < 540 mm 21 Depth: < 680 mm 27 Height: < 660 mm 26 Footprint: < 540 x 560 mm 21 x 27 General specifications Dimensions Metric Non-Metric Width: < 1060 mm 42 Depth: < 910 mm 36 Height: < 800 mm 32 Footprint: < 1060 x 610 mm 42 x 24 Reader specifications Number of plates: 2 Photometric range: OD Sample capacity: 100 per load Spectral range: nm Continuous load: Yes Assays per plate: Up to 12 Reader specifications Washer specifications Manifold configurations: Programmable volumes: 8-way wash head μl Photometric range: OD Residual volume: < 3 μl / per well Spectral range: nm Incubator specifications Washer specifications Manifold configurations: Programmable volumes: 8-way wash head μl Number of incubators: Up to 4 Temperature range: RT + 7 C to 50 C Temperature accuracy: ± 1 C Incubator specifications Temperature range: RT +4 C to 40 C Shaking: Pipetting specifications > 15 Hz periodic or continuous Temperature uniformity: ±1 C across 37 C Number of plates: 4 Shaking: Hz periodic or continuous Number of assays: 1 assay per strip or up to 12 assays per plate Pipetting specifications Type: Disposable tips (2 types) Maximum dilution: Serial dilutions: Yes Replicates: Up to 96 samples, standards and controls Precision, sample tip: <3% CV ( μl) Precision, reagent tip: <3% CV ( μl) Single-shot sample pipetting precisions: Single-shot sample pipetting accuracy: Number of sample tips loaded: 4 racks of 108 Reagent pipetting precision: Reagent pipetting accuarcy: Process security 3% CV at any operating volume above 10 μl ± 2% of target volume at any operating volume above 10 μl 3% CV at 10 shots at any volume in operating range above 25 μl ± 2% of target volume in operating range (single-shot mode) Process security Liquid-level sensing: Yes (reagents, controls and samples) Liquid-level sensing: Yes (reagents, controls and samples) Level-sensor system: Pressure differential Level-sensor system: Pressure differential Clot detection: Yes Clot detection: Yes Foam detection: Yes Foam detection: Yes Tip detection: Yes Tip detection: Yes Well-fill verification: Yes Well-fill verification: Yes Alarms: Yes Alarms: Yes Software Software Computer(not included): Current model desktop or laptop PC running MS Windows XP (contact Euro Diagnostica for current specs prior to purchase) Computer(not included): Controlling software: Current model desktop or laptop PC running MS Windows XP (contact Euro Diagnostica for current specs prior to purchase) Revelation Controlling software: DS-Matrix Work protocols (assays): Unlimited Work protocols (assays): Unlimited Data processing: Quantitative and qualitative Data processing: Quantitative and qualitative Levey-Jennings: Yes Levey-Jennings: Yes Westgard rules: Yes Westgard rules: Yes Process reporting: Event log and error log Process reporting: Event log and error log Automatic / learn error recovery: Yes Automatic / learn error recovery: Yes Password access control: Yes Password access control: Yes Connection to LIS -system: Yes, by Lis-link application Connection to LIS -system: Yes, by Lis-link application product catalog I 59
60 WIESLAB ANALYTICAL SERVICE LABORATORY Wieslab Analytical Service Laboratory Your Autoimmunity testing partner Your one-stop Autoimmunity resource Wieslab Laboratory Services has been a trusted partner in autoimmunity testing for more than 20 years. We offer a comprehensive range of individual and panel tests, and pride ourselves on always ensuring accurate results. Wieslab Laboratory Services offers one of the widest ranging autoimmune testing services available. We also lead the development of panel tests containing key parameters for several diseases within the same test package. And we continue to invest in research and development of new tests, collaborating with leading scientists and institutions worldwide to find new possibilities - not only for diagnosis, but prognosis and theranostics as well. The breadth, speed and accuracy of our testing services is just one of the reasons our client list includes many of the leading laboratories and health care organizations in Scandinavia. Testing is available for most autoimmune diseases, including: Systemic vasculitis Anti-phospholipid syndrome Autoimmune liver disease Autoimmune thyroiditis Celiac disease Congenital heart block Glomerulonephritis Guillan-Barré syndrome Inflammatory bowel disease Mixed connective tissue disease Myasthenia gravis Multiple sclerosis Paraneoplastic neurological syndromes Pemphigus/Pemphigoid Polymyositis/Dermatomyositis Rheumatoid arthritis Sjögren s syndrome Scleroderma Systemic lupus erythematosus (SLE) Autoimmunity support tools The field of autoimmunity and testing can be confusing and information about it difficult to find. And patients diagnosed with a serious disease such as systemic vasculitis need all the support they can get. That s why we have produced a range of reference materials that are recognised and appreciated worldwide. In fact, the Laboratory Handbook - Guide to Autoimmune Testing - is used as a text book in many medical schools. Recognising the need for a reference resource for health care professionals, Wieslab Laboratory Services founder Jörgen Wieslander penned the Laboratory Handbook in This comprehensive resource includes descriptions of autoimmunity processes and diseases, as well as testing interpretation guidance. An invaluable, one-of-a-kind reference only available from Wieslab. A shorter Quick Guide with a simpler overview of analyses and diseases is also available. Wieslab Request form Autoimmune diagnostics Send to Wieslab AB, Box 50117, Malmö, Sweden Contact Information / Billing Information Street Address Lundavägen 151, Contact Patient Identification S Malmö, Sweden T +46 (0) F +46 (0) Requesting Doctor Sample Date Type of specimen Telephone: Serum EDTA-plasma Liquor (CSF)... Specimen Collection Information Take a venous or capillary blood sample in a tube without additive, let the sample clot and centrifuge it. The sample can be kept in a refrigerator while waiting for transport. Send 1-3 ml serum by ordinary mail. This will be enough for one or several analyses. Visit our website 60 I product catalog Select test package ANCA for Systemic Vasculitis (GPA (formerly WG), MPA) PR3-ANCA, Capture PR3-ANCA, MPO-ANCA, Capture MPO-ANCA Additional testing for positive ANCA C-ANCA, P-ANCA (IIF) IgG-subclass determination of anti-pr3 or anti-mpo PiZ determination of alpha-1-antitrypsin ANCA follow up testing of Systemic vasculitis PR3-ANCA, Capture PR3-ANCA 571 Hereditary angioedema (HAE) Individual analyses on the reverse side C1-inactivator (C1-esterase inhibitor or C1-INH), C3, C4, C1q, Complement function (Classical and alternative patways and lectin pathway (MBL- and Ficolin 3 patways), Expanded analysis is performed at decreased levels of C1-INH or the complement function. 505 MPO-ANCA, Capture MPO-ANCA 513 Inflammatory bowel diseases ASCA IgA+IgG, IIF ANCA ANCA expand 503 If IIF-ANCA is positive, a follow
61 Our partners are always welcome to order these materials free of charge, through our website or by contacting a Wieslab Laboratory Services representative. You also have complete access to our in-house immunologists, whose experience and knowledge is built on the thousands of individual cases they see each year. Our partners are always welcome to call and discuss results, patient cases and options for further testing strategies free of charge. Quality matters High quality standards and processes are absolutely essential when testing autoimmune samples. Our autoimmunity experts have established strict internal practices that ensure dependable results every time. Naturally, our laboratories are also certified according to the highest regulatory standards: Our Scandinavia lab is certified SS-EN ISO/IEC 17025:2005 and accredited by SWEDAC since Wieslab Laboratory Services at a glance Pioneers in ANCA and CCP testing Wide portfolio of single-test and multi-test panels, latest tests and methods Critical Rapid Testing for ANCA and anti-gbm answers within 1 hour, fully confirmed within 24 hours Fast 2-day service on all standard tests 80% of tests have turn-around-time of 1 day Ongoing access to experienced in-house immunologists Support tools for patients as well as professionals in medicine Fully-accredited laboratory Experience in supporting clinical trials as partner to CROs Ongoing research and development product catalog I 61
62 TECHNICAL / PRODUCT OVERVIEW Technical/Product overview Alphabetical Order Technical / Product overview Product Code Antigen(s)/Material Technique ANA (IgG, IgM) FANA 200 Cell extract ELISA Qualitative ANCA GBM screen (IgG) GCP 100 Human Myeloperoxidase (MPO), Proteinase 3 (PR3), bovine GBM ANCA screen (IgG) CP 111 Human Myeloperoxidase (MPO), Proteinase 3 (PR3) ANCA panel (IgG) PAN 106 Human Azurozidin, Bactericidal Permeability Increasing Protein (BPI), Cathepsin G, Elastase, Elastase, Laktoferrin, (+ higg, HSA) ELISA Qualitative ELISA Qualitative ELISA Qualitative Angiotensin II RB 320 Synthetic peptide, polyclonal antiserum Competitive RIA ANP BAN I Synthetic peptide, polyclonal antiserum Competitive RIA ASCA IgA ASCA 150 Mannan (S. cerevisiae) ELISA ASCA IgG ASCA 151 Mannan (S. cerevisiae) ELISA ß2-Glycoprotein-1 IgG FBGP 200 Human ß2-Glycoprotein-1 ELISA ß2-Glycoprotein-1 IgM FBGP 100M Human ß2-Glycoprotein-1 ELISA Anti-Cardiolipin IgG FCAR 100G Human Cardiolipin + ß2-GP1 ELISA Cardiolipin IgM FCAR 100M Human Cardiolipin + ß2-GP1 ELISA Cardiolipin ( IgM, IgG, IgA) FCAR 100T Human Cardiolipin + ß2-GP1 ELISA CCK RB 302 Synthetic sulphated peptide (CCK-8), polyclonal antiserum Competitive RIA Anti-CCP (IgG) RA-96Plus Synthetic cyclic citrullinated peptides ELISA Immunoscan CCPlus Anti-CCP (IgG) EDIA anti-ccp FCCP 100 Synthetic cyclic citrullinated peptides ELISA CCPoint CCPoint20 US Synthetic cyclic citrullinated peptides Lateral flow, rapid test CCPoint CCPoint20 Synthetic cyclic citrullinated peptides Lateral flow, rapid test Anti-Centromer (IgG) FCEN 200 Recombinant c-terminal CENP-B ELISA Chromogranin A NEOLISA CGA Synthetic CgA fragments ELISA Chromogranin A RB 321 Purified human chromogranin A fragment Competitive RIA Chromogranin B RB 322 Synthetic peptide Competitive RIA hcomp COMP 200 Human COMP (cartilage oligomeric matrix protein), polyclonal antiserum Complement, Alternative pathwway COMPL AP 330 Lipopolysaccharide LPS, monoclonal anti-c5b-9 conjugate ELISA ELISA Complement Classical pathway COMPL CP 310 Human IgM, monoclonal anti-c5b-9 conjugate ELISA Complement Lectin/MBL pathway COMPL MP 320 Mannan, monoclonal anti-c5b-9 conjugate ELISA Complement Lectin/Ficolin3 pathway COMPLF3 Acetylated BSA, monoclonal anti-c5b-9 conjugate ELISA Complement, Total functional screen COMPL 300 Mannan, IgM, Lipopolysaccharide LPS, monoclonal anti-c5b-9 conjugate ELISA ds DNA (IgG, IgM) FDNA 100 Calf thymus dsdna ELISA Anti-ENA Profile FAID 200 Purified antigens, Sm, Sm/RNP, Ro, La, individual antigens each well Anti-ENA (IgG), single well FSWS 200 Purified antigens, Sm, Sm/RNP, Ro, La, Scl 70, Jo-1 mix of antigens in each well ELISA, Qualitative ELISA Qualitative Visit our website 62 I product catalog
63 Regulatory status Standards, Controls Specimen, Sample volume Incubation times (min) Wavelength Test per Kit CE, 510 (k) RC, PC, NC Serum, plasma, 10 μl nm 96 CE, 510 (k) NC, PC Serum, plasma, 25 μl nm 6x(2x8) CE, 510 (k) NC, PC Serum, 25 µl nm 96 CE NC, PC Serum, 10 µl nm 12 x 8 CE 7, low C, high C Plasma, extraction 24 h N/A 100 RUO 9, low C, high C Plasma, extraction 48 h N/A 100 CE 5, NC, PC Serum, 10 µl nm 96 CE 5, NC, PC Serum, 5 µl nm 96 CE, 510 (k) 5, NC, PC, RC Serum, plasma 10 µl nm 96 CE, 510 (k) 5, NC, PC, RC Serum, plasma 10 µl nm 96 CE, 510 (k) 5, NC, PC, RC Serum, plasma 10 µl nm 96 CE, 510 (k) 5, NC, PC, RC Serum, plasma 10 µl nm 96 CE, 510 (k) NC, PC Serum, plasma 10 µl nm 96 CE High C, low C Plasma, extraction 6 days N/A 100 CE, 510 (k) 5, NC, PC, RC Serum, 10 µl nm 96 CE, 510 (k) 6, NC, PC, RC Serum, plasma 10 µl nm (k) N/A Serum, plasma 10 N/A 20x1 CE N/A Whole blood, serum, plasma 10 N/A 20x1 CE, 510 (k) 5, NC, PC, RC Serum, plasma 10 µl nm 96 CE 6, low C, high C Serum, plasma 50 µl nm 96 CE 7, low C, high C Serum, plasma 100 µl 24 h N/A 100 CE 7, low C, high C Serum, plasma 100 µl 24 h N/A 100 RUO 6, 2 controls Serum, synovial fluid h (4 C) nm 96 CE NC, RC Serum, 20 µl nm 96 CE NC, RC Serum, 5 µl nm 96 CE NC, RC Serum, 5 µl nm 96 RUO NC, RC Serum, 10 µl nm 96 CE NC, RC Serum 5/5/20 µl (15) nm 3x32 CE, 510 (k) 5, NC, PC, RC Serum, plasma 10 µl nm 96 CE, 510 (k) NC, PC, RC Serum, plasma 10 µl nm 96 CE, 510 (k) NC, PC, RC Serum, plasma 10 µl nm 96 product catalog I 63
64 TECHNICAL / PRODUCT OVERVIEW Technical/Product overview Alphabetical Order Technical / Product overview Product Code Antigen(s)/Material Technique Beta-Endorphin RB 301 Synthetic peptide, polyclonal antiserum Competitive RIA Endothelin RB 304 Synthetic peptide, polyclonal antiserum Competitive RIA sgag (glycosaminoglycan)) GAG 201 Alcian blue, chondroitin sulphate-6 Dye-binding assay Gastrin MD 302 Synthetic sulphated peptide, polyclonal antiserum Competitive RIA Gastrin MD 302 US Synthetic sulphated peptide, polyclonal antiserum Competitive RIA Anti-GBM (IgG) GP 104X Purified bovine GBM (NC1 domain) ELISA Anti-GBM (IgG) FGBM 200 Purified bovine GBM (NC1 domain) ELISA Glucagon RB 310 Synthetic peptide, polyclonal antiserum Competitive RIA Glucagon RB 310 US Synthetic peptide, polyclonal antiserum Competitive RIA Jo-1 (IgG) FAJO 200 Purified Jo-1 ELISA Qualitative La (SS-B) (IgG) FALA 200 Purified La (SS-B) ELISA Anti-Mitochondria (IgG, IgM) FMIT 200 Purified M2 protein ELISA, Qualitative Capture MPO-ANCA (IgG) Cap MPO US Human myeloperoxidase Capture ELISA Capture MPO-ANCA (IgG) Cap MPO IU Human myeloperoxidase Capture ELISA MPO ANCA (IgG) MPO IU Human myeloperoxidase ELISA MPO ANCA (IgG) FMPO 200 Human myeloperoxidase ELISA Alpha-MSH RB 303 Synthetic peptide, polyclonal antiserum Competitive RIA Pertussis IgG/IgA, PT/FHA PERT-TRO Purified Pertussis toxin, Purified Filamentous Hemagglutinin ELISA Pertussis PT IgG PERT-PTG2 Purified Pertussis toxin ELISA PP (pancreatic polypeptide) RB 316 Synthetic peptide, polyclonal antiserum Competitive RIA Capture PR3-ANCA (IgG) Cap PR3 US Human proteinase 3 ELISA, capture Capture PR3-ANCA (IgG) Cap PR3 IU Human proteinase 3 ELISA, capture PR3 ANCA (IgG) PR3 IU Human proteinase 3 ELISA PR3 ANCA (IgG) FPRO 200 Human proteinase 3 ELISA Ro (SS-A) (IgG) FARO 300 Purified Ro 60 (SS-A) ELISA Scl 70 (IgG) FSCL 200 Purified Scl 70 ELISA Qualitative Anti-Sm IgG) FASM 200 Purified Sm (Smith antigen) ELISA Anti-Sm/RNP FRNP 200 Purified Sm/RNP ELISA Somatostatin RB 306 Synthetic peptide, polyclonal antiserum Competitive RIA SS-A p200 (IgG) SSA-p200 Synthetic peptide Subregion SS-A/Ro 52 ( ) ELISA, Qualitative Anti-Thyroglobulin (IgG) FATG 200 Purified thyroglobulin ELISA Anti- Thyroid peroxidase (IgG) FTPO 300 Recombinant human thyroid peroxidase ELISA Vasculitis screen (IgG) GCP-CAP Purified bovine GBM, human PR3, human MPO (+ HAMA, HSA) Qualitative Direct GBM, Capture PR3/MPO ANCA Vasopressin RB 319 Synthetic peptide, polyclonal antiserum Competitive RIA VIP (Vasointestinal peptide) RB 311 Synthetic peptide, polyclonal antiserum Competitive RIA Visit our website 64 I product catalog
65 Regulatory status Standards, Controls Specimen, Sample volume Incubation times (min) Wavelength Test per Kit RUO 7, low C, high C Plasma, extraction 48 h N/A 100 RUO 7, low C, high C Plasma, extraction 48 h N/A 100 RUO 6 Tissue, blood, culture medium 7 x nm Ca 200 CE 7, low C, high C Serum, 100 µl 2 h N/A 100 CE, Class 1 Exempt 7, low C, high C Serum, 100 µl 2 h N/A 100 CE, 510 (k) 5, NC, PC Serum, 5 µl nm 96 CE, 510 (k) 5, NC, PC, RC Serum, plasma 10 µl nm 96 CE 7, low C, high C Plasma 48 h N/A 100 CE, Class 1 Exempt 7, low C, high C Plasma 48 h N/A 100 CE, 510 (k) NC, PC, RC Serum, plasma 10 µl nm 96 CE, 510 (k) 5, NC, PC, RC Serum, plasma 10 µl nm 96 CE, 510 (k) NC, PC, RC Serum, plasma 10 µl nm (k) 6, NC, PC Serum, 10 µl nm 96 CE 6, NC, PC Serum, 10 µl nm 96 CE 6, NC, PC Serum, 10 µl nm 96 CE, 510 (k) 5, NC, PC RC Serum, plasma 10 µl nm 96 RUO 7, low C, high C Plasma, Liqvor, 100 µl 48 h N/A 100 CE NC, PC: FHA/PT IgG NC, PC: FHA/PT IgA Serum, plasma 10 µl nm 2x96 CE, Class 1 Exempt PC, NC Serum, 10 µl nm 96 CE 7, low C, high C Serum, 100 µl 48 h N/A (k) 6, NC, PC Serum, 10 µl nm 96 CE 6, NC, PC Serum, 10 µl nm 96 CE 6, NC, PC Serum, 10 µl nm 96 CE, 510 (k) 5, NC, PC, RC Serum, plasma 10 µl nm 96 CE, 510 (k) 5, NC, PC, RC Serum, plasma 10 µl nm 96 CE, 510 (k) NC, PC, RC Serum, plasma 10 µl nm 96 CE, 510 (k) 5, NC, PC, RC Serum, plasma 10 µl nm 96 CE, 510 (k) 5, NC, PC, RC Serum, plasma 10 µl nm 96 RUO 7, low C, high C Plasma, extraction 48 h N/A 100 CE NC, PC, RC Serum, 5 µl nm 96 CE, 510 (k) 5 NC, PC, RC Serum, plasma 10 µl nm 96 CE, 510 (k) 5, NC, PC, RC Serum, plasma 10 µl nm 96 CE NC, PC Serum, 50 µl nm 96 CE 7, low C, high C Plasma extraction, urine 48 h N/A 100 CE 7, low C, high C Plasma, 200 µl 48 h N/A 100 product catalog I 65
66 Neolisa Chromogranin a An excellent marker for neuroendocrine tumours Visit our website 66 I product catalog
67 Pertusscan Pt IgG Accurate diagnosis of Pertussis infection product catalog I 67
68 ProDUCT LIST Euro Diagnostica Product List Alphabetical Order Code Product name Page COMPL AP Alternative Pathway (Complement system) Wieslab 27 RB Alpha-MSH EURIA 52 FANA ANA DIASTAT 30 PAN ANCA panel kit Wieslab 20 CP ANCA screen kit Wieslab 20 RB Angiotensin II EURIA 47 BANI 1 3 ANP EURIA 47 FBGP Anti-beta-2-glycoprotein 1 IgG DIASTAT 40 FBGP 100M 1 Anti-beta-2-glycoprotein 1 IgM DIASTAT 40 FCAR 100G 1 Anti-Cardiolipin IgG DIASTAT 41 FCAR 100M 1 Anti-Cardiolipin IgM DIASTAT 41 FCAR 100T 1 Anti-Cardiolipin Total DIASTAT 41 FCCP Anti-CCP EDIA 11 FCEN Anti-Centromere DIASTAT 31 FDNA Anti-dsDNA DIASTAT 32 FGBM Anti-GBM DIASTAT 23 GP 104X 1 Anti-GBM semi quantitative kit Wieslab 23 GCP Anti-GBM, ANCA screen kit Wieslab 20 FAJO Anti-Jo-1 DIASTAT 33 FALA Anti-La (SS-B) DIASTAT 31 FMIT Anti-Mitochondria DIASTAT 33 FARO Anti-Ro (SS-A) DIASTAT 34 FSCL Anti-Scl-70 DIASTAT 33 FASM Anti-Sm DIASTAT 34 FRNP Anti-Sm/RNP DIASTAT 35 SSA p200 2 Anti-SS-A p200 Wieslab 37 FATG Anti-Tg DIASTAT 45 FTPO Anti-TPO DIASTAT 45 ASCA ASCA IgG semi quantitative kit Wieslab 43 ASCA ASCA IgA semi quantitative kit Wieslab 43 RB Beta-Endorphin EURIA 50 FBGP Beta-2-glycoprotein 1 IgG DIASTAT 40 FBGP 100M 1 Beta-2-glycoprotein 1 IgM DIASTAT 40 Cap MPO IU 2 Capture MPO-ANCA Wieslab 19 Cap PR3 IU 2 Capture PR3-ANCA Wieslab 18 FCAR 100G 1 Cardiolipin IgG DIASTAT 41 FCAR 100M 1 Cardiolipin IgM DIASTAT 41 FCAR 100T 1 Cardiolipin Total DIASTAT 41 RB CCK EURIA 48 FCCP100 1 CCP EDIA 11 RA-96PLUS 1 CCPlus Immunoscan 10 CCPoint 20 2 CCPoint 12 CCPoint 20 US 1 CCPoint US 13 FCEN Centromere DIASTAT 31 RB Chromogranin A EURIA 49 CGA 2 Chromogranin A Neolisa 48 RB Chromogranin B EURIA 49 COMPL CP Classical Pathway (Complement system) Wieslab 28 Visit our website 68 I product catalog
69 Code Product name Page COMPL Complement system Screen Wieslab 26 COMPL AP Complement system Alternative Pathway Wieslab 27 COMPL CP Complement system Classical Pathway Wieslab 27 COMPLF 3 3 Complement system Ficolin 3 Wieslab 28 COMPL MP Complement system MBL pathway Wieslab 28 FDNA100 1 dsdna DIASTAT 30 FAID ENA profile DIASTAT 35 FSWS ENA single well screen DIASTAT 35 RB Endothelin EURIA 50 COMPLF 3 3 Ficolin 3 (Complement system) Wieslab 28 MD /MD 302 US 1 Gastrin EURIA 51 GCP GBM, ANCA screen kit Wieslab 20 FGBM GBM DIASTAT 23 GP 104X 1 GBM semi quantitative kit Wieslab 23 RB /RB 310 US 1 Glucagon EURIA 51 COMP hcomp quantitative kit Wieslab 14 FAJO Jo-1 DIASTAT 33 FALA La (SS-B) DIASTAT 31 COMPL MP MBL pathway (Complement system) Wieslab 27 FMIT Mitochondria DIASTAT 33 Cap MPO IU 2 MPO-ANCA (Capture) Wieslab 19 FMPO MPO ANCA DIASTAT 19 MPO IU 2 MPO-ANCA Wieslab 19 RB NPY EURIA 52 PERT-PTG2 1 Pertusscan PT IgG 56 PERT-TRO 2 Pertusscan TRO 56 RB PP EURIA 53 FPRO PR3-ANCA DIASTAT 18 PR3 IU 2 PR3-ANCA Wieslab 18 Cap PR3 IU 2 PR3-ANCA (Capture) Wieslab 18 FARO Ro (SS-A) DIASTAT 34 COMPL Screen (Complement system) Wieslab 26 FSCL Scl-70 DIASTAT 33 GAG sgag quantitative kit Wieslab 15 FASM Sm DIASTAT 34 FRNP Sm/RNP DIASTAT 35 RB Somatostatin EURIA 53 SSA p200 2 SS-A p200 Wieslab 37 FATG Tg DIASTAT 45 FTPO TPO DIASTAT 45 GCP-CAP 2 Vasculitis screen Wieslab 20 RB Vasopressin EURIA 54 RB VIP EURIA 54 1) Please contact your local representative for availability in your country, available in US! 2) Please contact your local representative for availability in your country, not available in US! 3) For research use only, contact your local representative for research use availability in your country. product catalog I 69
70 ORDERING INFORMATION Ordering Information Ordering and Shipping Contact Information Address: Euro Diagnostica P. O. Box SE Malmö Sweden Visiting address: Lundavägen 151, Malmö, Sweden Phone: Fax: [email protected] Pricing Contact your local Euro Diagnostica sales representative or your distributor to obtain up-to-date price information. Ordering Our professional Customer Service team is dedicated to providing you with swift and comprehensive service. Your satisfaction is our goal. You can reach us by mail, phone, and fax. Orders received before noon (Swedish time) will be shipped within 24 hours if not agreed otherwise. Standing Orders Products can be shipped on a periodic basis as requested with an accompanying purchase order. Please contact your local Euro Diagnostica sales representative or your distributor to schedule standing orders. Product Returns Our products meet rigid production and quality control standards before they are released for sale. Proper storage and handling by the customer is necessary to assure satisfactory performance. Goods may be returned only with prior authorization of Euro Diagnostica. Warranty Statement Products are warranted to perform according to the procedures described in the product insert accompanying the product. Results may be altered by any changes or modifications to the procedures. Euro Diagnostica disclaims any implied warranty, merchantability or fitness for any other purpose. In no event shall Euro Diagnostica be liable for any consequential damages arising out of the above express warranty. Euro Diagnostica Online For up-to-date information about Euro Diagnostica, our products, services and subsidiaries, please visit us at Product support literature, product inserts and material safety data sheets are all accessible as downloads. An online version of our most updated Catalog is also available on our website. Visit our website 70 I product catalog
71 For a comprehensive overview of the main autoimmune diseases and their diagnostics please visit Euro Diagnostica s new website: product catalog I 71
72 Diagnostic solutions leader in autoimmunity testing We thank you for your interest in our products and we commit ourselves to continually developing and providing you with the highest quality innovative clinical solutions. Euro Diagnostica AB Mail address: P.O. Box SE Malmö Sweden Visiting address: Lundavägen 151 Malmö Sweden T F E W [email protected] Doc No: E-063-GB00, September 2012
Rheumatology Labs for Primary Care Providers. Robert Monger, M.D., F.A.C.P. 2015 Frontiers in Medicine
 Rheumatology Labs for Primary Care Providers Robert Monger, M.D., F.A.C.P. 2015 Frontiers in Medicine Objectives Review the Indications for and Interpretation of lab testing for the following diseases:
Rheumatology Labs for Primary Care Providers Robert Monger, M.D., F.A.C.P. 2015 Frontiers in Medicine Objectives Review the Indications for and Interpretation of lab testing for the following diseases:
Rheumatoid arthritis: an overview. Christine Pham MD
 Rheumatoid arthritis: an overview Christine Pham MD RA prevalence Chronic inflammatory disease affecting approximately 0.5 1% of the general population Prevalence is higher in North America (approaching
Rheumatoid arthritis: an overview Christine Pham MD RA prevalence Chronic inflammatory disease affecting approximately 0.5 1% of the general population Prevalence is higher in North America (approaching
ANTIBODIES AGAINST CITRULLINATED PEPTIDES IN EARLY RHEUMATOID ARTHRITIS: DIAGNOSTIC AND PROGNOSTIC SIGNIFICANCE
 ANTIBODIES AGAINST CITRULLINATED PEPTIDES IN EARLY RHEUMATOID ARTHRITIS: DIAGNOSTIC AND PROGNOSTIC SIGNIFICANCE Principal investigators: Dr Raimon Sanmartí Sala Hospital Clínic i Provincial de Barcelona
ANTIBODIES AGAINST CITRULLINATED PEPTIDES IN EARLY RHEUMATOID ARTHRITIS: DIAGNOSTIC AND PROGNOSTIC SIGNIFICANCE Principal investigators: Dr Raimon Sanmartí Sala Hospital Clínic i Provincial de Barcelona
Testing for RA. The Ideal Lab Test. William M. Wason, MD, PhD 9/24/2010. Confusion Abounds
 Confusion Abounds Rheumatoid arthritis: ulnar deviation and muscle artrophy, hands Poor sensitivity and specificity Hepatitis C causes lots of false + tests Changing technology in how tests are done Historic
Confusion Abounds Rheumatoid arthritis: ulnar deviation and muscle artrophy, hands Poor sensitivity and specificity Hepatitis C causes lots of false + tests Changing technology in how tests are done Historic
Systemic Lupus Erythematosus
 Harvard-MIT Division of Health Sciences and Technology HST.021: Musculoskeletal Pathophysiology, IAP 2006 Course Director: Dr. Dwight R. Robinson Systemic Lupus Erythematosus A multi-system autoimmune
Harvard-MIT Division of Health Sciences and Technology HST.021: Musculoskeletal Pathophysiology, IAP 2006 Course Director: Dr. Dwight R. Robinson Systemic Lupus Erythematosus A multi-system autoimmune
ANA testing can now be ordered in several ways, depending on the clinical circumstances:
 LAB TEST CONNECT Multiplex ANA Screen Dr. Joseph Schappert, M.D.,Medical Director Chief Medical Offi cer ANA has been the primary screening test for connective tissue diseases (CTD s) for many years. While
LAB TEST CONNECT Multiplex ANA Screen Dr. Joseph Schappert, M.D.,Medical Director Chief Medical Offi cer ANA has been the primary screening test for connective tissue diseases (CTD s) for many years. While
LAB 1 - Direct agglutination. Serology-the study of the in vitro reactions between antibody and antigen
 LAB 1 - Direct agglutination Serology-the study of the in vitro reactions between antibody and antigen Serological reaction: - quantitative (weight/volume) - qualitative Agglutination - the aggregation
LAB 1 - Direct agglutination Serology-the study of the in vitro reactions between antibody and antigen Serological reaction: - quantitative (weight/volume) - qualitative Agglutination - the aggregation
Topic: Serological reactions: the purpose and a principle of reactions. Agglutination test. Precipitation test. CFT, IFT, ELISA, RIA.
 Topic: Serological reactions: the purpose and a principle of reactions. Agglutination test. Precipitation test. CFT, IFT, ELISA, RIA. Serology is the study and use of immunological tests to diagnose and
Topic: Serological reactions: the purpose and a principle of reactions. Agglutination test. Precipitation test. CFT, IFT, ELISA, RIA. Serology is the study and use of immunological tests to diagnose and
The ANA Test: All You Need to Know Department of Family and Community Medicine Family Medicine Update April 25, 2014
 The ANA Test: All You Need to Know Department of Family and Community Medicine Family Medicine Update April 25, 2014 Celso R. Velázquez MD Division of Rheumatology University of Missouri [email protected]
The ANA Test: All You Need to Know Department of Family and Community Medicine Family Medicine Update April 25, 2014 Celso R. Velázquez MD Division of Rheumatology University of Missouri [email protected]
Dr Sarah Levy Consultant Rheumatology Croydon University Hospital
 Dr Sarah Levy Consultant Rheumatology Croydon University Hospital Contents Definition/ epidemiology Diagnosis Importance of early diagnosis/ treatment Guidelines Evidence based treatment protocol Current
Dr Sarah Levy Consultant Rheumatology Croydon University Hospital Contents Definition/ epidemiology Diagnosis Importance of early diagnosis/ treatment Guidelines Evidence based treatment protocol Current
NURS 821 Alterations in the Musculoskeletal System. Rheumatoid Arthritis. Type III Hypersensitivity Response
 NURS 821 Alterations in the Musculoskeletal System Margaret H. Birney PhD, RN Lecture 12 Part 2 Joint Disorders (cont d) Rheumatoid Arthritis Definition: Autoimmune disorder occurring in genetically sensitive
NURS 821 Alterations in the Musculoskeletal System Margaret H. Birney PhD, RN Lecture 12 Part 2 Joint Disorders (cont d) Rheumatoid Arthritis Definition: Autoimmune disorder occurring in genetically sensitive
WIESLAB Complement system
 Instruction WIESLAB Complement system Alternative pathway Enzyme immunoassay for assessment of Complement functional activity Break apart microtitration strips (12x8) 96 wells Store the kit at +2-8 C Store
Instruction WIESLAB Complement system Alternative pathway Enzyme immunoassay for assessment of Complement functional activity Break apart microtitration strips (12x8) 96 wells Store the kit at +2-8 C Store
Differential Diagnosis
 Differential Diagnosis Limited Scleroderma and Scleroderma-Like Disorders Morphea, or localized Scleroderma, usually begins between the ages of 20 to 50 years as patches of yellowish or ivory-colored rigid,
Differential Diagnosis Limited Scleroderma and Scleroderma-Like Disorders Morphea, or localized Scleroderma, usually begins between the ages of 20 to 50 years as patches of yellowish or ivory-colored rigid,
Autoimmune Diseases More common than you think Randall Stevens, MD
 Autoimmune Diseases More common than you think Randall Stevens, MD picture placeholder Autoimmune Diseases More than 60 different disorders Autoimmune disorders (AID) diseases caused by the immune system
Autoimmune Diseases More common than you think Randall Stevens, MD picture placeholder Autoimmune Diseases More than 60 different disorders Autoimmune disorders (AID) diseases caused by the immune system
The Most Common Autoimmune Disease: Rheumatoid Arthritis. Bonita S. Libman, M.D.
 The Most Common Autoimmune Disease: Rheumatoid Arthritis Bonita S. Libman, M.D. Disclosures Two googled comics The Normal Immune System Network of cells and proteins that work together Goal: protect against
The Most Common Autoimmune Disease: Rheumatoid Arthritis Bonita S. Libman, M.D. Disclosures Two googled comics The Normal Immune System Network of cells and proteins that work together Goal: protect against
Rheumatoid Arthritis
 Rheumatoid Arthritis While rheumatoid arthritis (RA) has long been feared as one of the most disabling types of arthritis, the outlook has dramatically improved for many newly diagnosed patients. Certainly
Rheumatoid Arthritis While rheumatoid arthritis (RA) has long been feared as one of the most disabling types of arthritis, the outlook has dramatically improved for many newly diagnosed patients. Certainly
Rheumatoid Arthritis www.arthritis.org.nz
 Rheumatoid Arthritis www.arthritis.org.nz Did you know? RA is the second most common form of arthritis Approximately 40,000 New Zealanders have RA RA can occur at any age, but most often appears between
Rheumatoid Arthritis www.arthritis.org.nz Did you know? RA is the second most common form of arthritis Approximately 40,000 New Zealanders have RA RA can occur at any age, but most often appears between
How Does a Doctor Test for AIDS?
 Edvo-Kit #S-70 How Does a Doctor Test for AIDS? S-70 Experiment Objective: The Human Immunodefi ciency Virus (HIV) is an infectious agent that causes Acquired Immunodefi ciency Syndrome (AIDS) in humans.
Edvo-Kit #S-70 How Does a Doctor Test for AIDS? S-70 Experiment Objective: The Human Immunodefi ciency Virus (HIV) is an infectious agent that causes Acquired Immunodefi ciency Syndrome (AIDS) in humans.
Early Diagnosis of Rheumatoid Arthritis & Axial Spondyloarthritis
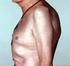 Early Diagnosis of Rheumatoid Arthritis & Axial Spondyloarthritis 奇 美 醫 院 過 敏 免 疫 風 濕 科 陳 宏 安 Rheumatoid arthritis Most common chronic inflammatory joint disease Multisystem autoimmune disease of unknown
Early Diagnosis of Rheumatoid Arthritis & Axial Spondyloarthritis 奇 美 醫 院 過 敏 免 疫 風 濕 科 陳 宏 安 Rheumatoid arthritis Most common chronic inflammatory joint disease Multisystem autoimmune disease of unknown
1.5 Function of analyte For albumin, see separate entry. The immunoglobulins are components of the humoral arm of the immune system.
 Total protein (serum, plasma) 1 Name and description of analyte 1.1 Name of analyte Total protein 1.2 Alternative names None 1.3 NMLC code 1.4 Description of analyte This is a quantitative measurement
Total protein (serum, plasma) 1 Name and description of analyte 1.1 Name of analyte Total protein 1.2 Alternative names None 1.3 NMLC code 1.4 Description of analyte This is a quantitative measurement
The EliA System Time for the essentials Cost efficient and flexible A boost in service for your laboratory and your clinicians
 CCP The EliA System Time for the essentials completely automated (true walk-away, overnight runs) easy instrument management by Phadia Data Manager (IDM) software barcode-reader protocols, QC and raw data
CCP The EliA System Time for the essentials completely automated (true walk-away, overnight runs) easy instrument management by Phadia Data Manager (IDM) software barcode-reader protocols, QC and raw data
Basic Immunologic Procedures. Complex Serological Tests
 Basic Immunologic Procedures Complex Serological Tests Amal Alghamdi 2014-2015 1 Classification of antigen-antibody interactions: 1. Primary serological tests: (Marker techniques) e.g. Enzyme linked immuonosorben
Basic Immunologic Procedures Complex Serological Tests Amal Alghamdi 2014-2015 1 Classification of antigen-antibody interactions: 1. Primary serological tests: (Marker techniques) e.g. Enzyme linked immuonosorben
Corporate Medical Policy
 Corporate Medical Policy Serum Biomarker Panel Testing for Systemic Lupus Erythematosus File Name: Origination: Last CAP Review: Next CAP Review: Last Review: serum_biomarker_panel_testing_for_systemic_lupus_erythematosus
Corporate Medical Policy Serum Biomarker Panel Testing for Systemic Lupus Erythematosus File Name: Origination: Last CAP Review: Next CAP Review: Last Review: serum_biomarker_panel_testing_for_systemic_lupus_erythematosus
LAB 14 ENZYME LINKED IMMUNOSORBENT ASSAY (ELISA)
 STUDENT GUIDE LAB 14 ENZYME LINKED IMMUNOSORBENT ASSAY (ELISA) GOAL The goal of this laboratory lesson is to explain the concepts and technique of enzyme linked immunosorbent assay (ELISA). OBJECTIVES
STUDENT GUIDE LAB 14 ENZYME LINKED IMMUNOSORBENT ASSAY (ELISA) GOAL The goal of this laboratory lesson is to explain the concepts and technique of enzyme linked immunosorbent assay (ELISA). OBJECTIVES
Lyme (IgG and IgM) Antibody Confirmation
 Pathology & Laboratory Medicine Lyme (IgG and IgM) Antibody Confirmation TEST UPDATE: New Test Notification Date: 1/9/2013 Effective Date: 1/7/2013 CONTACT INFO Call 802-847-5121 800-991-2799 email [email protected]
Pathology & Laboratory Medicine Lyme (IgG and IgM) Antibody Confirmation TEST UPDATE: New Test Notification Date: 1/9/2013 Effective Date: 1/7/2013 CONTACT INFO Call 802-847-5121 800-991-2799 email [email protected]
IgM ELISA. For the quantitative determination of IgM in human serum and plasma. For Research Use Only. Not For Use In Diagnostic Procedures.
 IgM ELISA For the quantitative determination of IgM in human serum and plasma For Research Use Only. Not For Use In Diagnostic Procedures. Please read carefully due to Critical Changes, e.g., Calibrator
IgM ELISA For the quantitative determination of IgM in human serum and plasma For Research Use Only. Not For Use In Diagnostic Procedures. Please read carefully due to Critical Changes, e.g., Calibrator
Arthritis in Children: Juvenile Rheumatoid Arthritis By Kerry V. Cooke
 Reading Comprehension Read the following essay on juvenile rheumatoid arthritis. Then use the information in the text to answer the questions that follow. Arthritis in Children: Juvenile Rheumatoid Arthritis
Reading Comprehension Read the following essay on juvenile rheumatoid arthritis. Then use the information in the text to answer the questions that follow. Arthritis in Children: Juvenile Rheumatoid Arthritis
WHO Prequalification of Diagnostics Programme PUBLIC REPORT. Product: Genscreen ULTRA HIV Ag-Ab Number: PQDx 0096-031-00. Abstract
 WHO Prequalification of Diagnostics Programme PUBLIC REPORT Product: Genscreen ULTRA HIV Ag-Ab Number: PQDx 0096-031-00 Abstract Genscreen ULTRA HIV Ag-Ab with product codes 72386 and 72388, manufactured
WHO Prequalification of Diagnostics Programme PUBLIC REPORT Product: Genscreen ULTRA HIV Ag-Ab Number: PQDx 0096-031-00 Abstract Genscreen ULTRA HIV Ag-Ab with product codes 72386 and 72388, manufactured
INTERPRETATION INFORMATION SHEET
 Creative Testing Solutions 2424 West Erie Dr. 2205 Highway 121 10100 Martin Luther King Jr. St. No. Tempe, AZ 85282 Bedford, TX 76021 St. Petersburg, FL 33716 INTERPRETATION INFORMATION SHEET Human Immunodeficiency
Creative Testing Solutions 2424 West Erie Dr. 2205 Highway 121 10100 Martin Luther King Jr. St. No. Tempe, AZ 85282 Bedford, TX 76021 St. Petersburg, FL 33716 INTERPRETATION INFORMATION SHEET Human Immunodeficiency
Selective IgA deficiency (slgad) [email protected] 0800 987 8986 www.piduk.org
 Selective IgA deficiency (slgad) [email protected] 0800 987 8986 www.piduk.org About this booklet This booklet provides information on selective IgA deficiency (sigad). It has been produced by the PID UK
Selective IgA deficiency (slgad) [email protected] 0800 987 8986 www.piduk.org About this booklet This booklet provides information on selective IgA deficiency (sigad). It has been produced by the PID UK
Proteins. Protein Trivia. Optimizing electrophoresis
 Proteins ELECTROPHORESIS Separation of a charged particle in an electric field Michael A. Pesce, Ph.D Department of Pathology New York-Presbyterian Hospital Columbia University Medical Center Rate of migration
Proteins ELECTROPHORESIS Separation of a charged particle in an electric field Michael A. Pesce, Ph.D Department of Pathology New York-Presbyterian Hospital Columbia University Medical Center Rate of migration
Understanding Rheumatoid Arthritis
 Understanding Rheumatoid Arthritis Understanding Rheumatoid Arthritis What Is Rheumatoid Arthritis? 1,2 Rheumatoid arthritis (RA) is a chronic autoimmune disease. It causes joints to swell and can result
Understanding Rheumatoid Arthritis Understanding Rheumatoid Arthritis What Is Rheumatoid Arthritis? 1,2 Rheumatoid arthritis (RA) is a chronic autoimmune disease. It causes joints to swell and can result
A Genetic Analysis of Rheumatoid Arthritis
 A Genetic Analysis of Rheumatoid Arthritis Introduction to Rheumatoid Arthritis: Classification and Diagnosis Rheumatoid arthritis is a chronic inflammatory disorder that affects mainly synovial joints.
A Genetic Analysis of Rheumatoid Arthritis Introduction to Rheumatoid Arthritis: Classification and Diagnosis Rheumatoid arthritis is a chronic inflammatory disorder that affects mainly synovial joints.
Chapter 43: The Immune System
 Name Period Our students consider this chapter to be a particularly challenging and important one. Expect to work your way slowly through the first three concepts. Take particular care with Concepts 43.2
Name Period Our students consider this chapter to be a particularly challenging and important one. Expect to work your way slowly through the first three concepts. Take particular care with Concepts 43.2
Fast Facts. Rheumatoid Arthritis. Fast Facts: John D Isaacs and Larry W Moreland. Second edition. 2011 Health Press Ltd www.fastfacts.
 Fast Facts Fast Facts: Rheumatoid Arthritis John D Isaacs and Larry W Moreland Second edition Fast Facts Fast Facts: Rheumatoid Arthritis Second edition John D Isaacs PhD FRCP Professor of Clinical Rheumatology
Fast Facts Fast Facts: Rheumatoid Arthritis John D Isaacs and Larry W Moreland Second edition Fast Facts Fast Facts: Rheumatoid Arthritis Second edition John D Isaacs PhD FRCP Professor of Clinical Rheumatology
510(k) SUBSTANTIAL EQUIVALENCE DETERMINATION DECISION SUMMARY
 A. 510(k) Number: K092353 510(k) SUBSTANTIAL EQUIVALENCE DETERMINATION DECISION SUMMARY B. Purpose for Submission: This is a new 510k application for a new indication for the MONOLISA Anti-HAV IgM EIA
A. 510(k) Number: K092353 510(k) SUBSTANTIAL EQUIVALENCE DETERMINATION DECISION SUMMARY B. Purpose for Submission: This is a new 510k application for a new indication for the MONOLISA Anti-HAV IgM EIA
ELISA BIO 110 Lab 1. Immunity and Disease
 ELISA BIO 110 Lab 1 Immunity and Disease Introduction The principal role of the mammalian immune response is to contain infectious disease agents. This response is mediated by several cellular and molecular
ELISA BIO 110 Lab 1 Immunity and Disease Introduction The principal role of the mammalian immune response is to contain infectious disease agents. This response is mediated by several cellular and molecular
Autoimmunity and immunemediated. FOCiS. Lecture outline
 1 Autoimmunity and immunemediated inflammatory diseases Abul K. Abbas, MD UCSF FOCiS 2 Lecture outline Pathogenesis of autoimmunity: why selftolerance fails Genetics of autoimmune diseases Therapeutic
1 Autoimmunity and immunemediated inflammatory diseases Abul K. Abbas, MD UCSF FOCiS 2 Lecture outline Pathogenesis of autoimmunity: why selftolerance fails Genetics of autoimmune diseases Therapeutic
Infl ectra for rheumatoid arthritis
 Infl ectra for rheumatoid arthritis Some important information to get you started with your treatment This booklet is intended only for use by patients who have been prescribed Inflectra. Introduction
Infl ectra for rheumatoid arthritis Some important information to get you started with your treatment This booklet is intended only for use by patients who have been prescribed Inflectra. Introduction
Basics of Immunology
 Basics of Immunology 2 Basics of Immunology What is the immune system? Biological mechanism for identifying and destroying pathogens within a larger organism. Pathogens: agents that cause disease Bacteria,
Basics of Immunology 2 Basics of Immunology What is the immune system? Biological mechanism for identifying and destroying pathogens within a larger organism. Pathogens: agents that cause disease Bacteria,
ANIMALS FORM & FUNCTION BODY DEFENSES NONSPECIFIC DEFENSES PHYSICAL BARRIERS PHAGOCYTES. Animals Form & Function Activity #4 page 1
 AP BIOLOGY ANIMALS FORM & FUNCTION ACTIVITY #4 NAME DATE HOUR BODY DEFENSES NONSPECIFIC DEFENSES PHYSICAL BARRIERS PHAGOCYTES Animals Form & Function Activity #4 page 1 INFLAMMATORY RESPONSE ANTIMICROBIAL
AP BIOLOGY ANIMALS FORM & FUNCTION ACTIVITY #4 NAME DATE HOUR BODY DEFENSES NONSPECIFIC DEFENSES PHYSICAL BARRIERS PHAGOCYTES Animals Form & Function Activity #4 page 1 INFLAMMATORY RESPONSE ANTIMICROBIAL
Clinically Actionable Biomarkers in Rheumatoid Arthritis
 Clinically Actionable Biomarkers in Rheumatoid Arthritis PepTalk January 6, 2009 William Robinson, MD, PhD Stanford University School of Medicine VA Palo Alto Health Care System T cell mediated Autoimmune
Clinically Actionable Biomarkers in Rheumatoid Arthritis PepTalk January 6, 2009 William Robinson, MD, PhD Stanford University School of Medicine VA Palo Alto Health Care System T cell mediated Autoimmune
Diagnosis of HIV-1 Infection. Estelle Piwowar-Manning HPTN Central Laboratory The Johns Hopkins University
 Diagnosis of HIV-1 Infection Estelle Piwowar-Manning HPTN Central Laboratory The Johns Hopkins University Tests Used to Diagnose HIV-1 Infection HIV antibody (today s topic) HIV p24 antigen HIV DNA HIV
Diagnosis of HIV-1 Infection Estelle Piwowar-Manning HPTN Central Laboratory The Johns Hopkins University Tests Used to Diagnose HIV-1 Infection HIV antibody (today s topic) HIV p24 antigen HIV DNA HIV
Rheumatoid Arthritis. Nicole Klett,, M.D.
 Rheumatoid Arthritis Nicole Klett,, M.D. Rheumatoid Arthritis Systemic Chronic Inflammatory Primarily targets the synovium of diarthrodial joints Etiology likely combination genetic and environmental Diarthrodial
Rheumatoid Arthritis Nicole Klett,, M.D. Rheumatoid Arthritis Systemic Chronic Inflammatory Primarily targets the synovium of diarthrodial joints Etiology likely combination genetic and environmental Diarthrodial
Rheumatoid Arthritis www.arthritis.org.nz
 Rheumatoid Arthritis www.arthritis.org.nz Did you know? Rheumatoid arthritis (RA) is the third most common form of arthritis Approximately 40,000 New Zealanders have RA RA can occur at any age, but most
Rheumatoid Arthritis www.arthritis.org.nz Did you know? Rheumatoid arthritis (RA) is the third most common form of arthritis Approximately 40,000 New Zealanders have RA RA can occur at any age, but most
Chapter 18: Applications of Immunology
 Chapter 18: Applications of Immunology 1. Vaccinations 2. Monoclonal vs Polyclonal Ab 3. Diagnostic Immunology 1. Vaccinations What is Vaccination? A method of inducing artificial immunity by exposing
Chapter 18: Applications of Immunology 1. Vaccinations 2. Monoclonal vs Polyclonal Ab 3. Diagnostic Immunology 1. Vaccinations What is Vaccination? A method of inducing artificial immunity by exposing
In non-hodgkin s lymphoma, MabThera is used to treat two types of the disease, both of which affect B-lymphocytes:
 EMA/614203/2010 EMEA/H/C/000165 EPAR summary for the public rituximab This is a summary of the European public assessment report (EPAR) for. It explains how the Committee for Medicinal Products for Human
EMA/614203/2010 EMEA/H/C/000165 EPAR summary for the public rituximab This is a summary of the European public assessment report (EPAR) for. It explains how the Committee for Medicinal Products for Human
Chapter 6: Antigen-Antibody Interactions
 Chapter 6: Antigen-Antibody Interactions I. Strength of Ag-Ab interactions A. Antibody Affinity - strength of total noncovalent interactions between single Ag-binding site on an Ab and a single epitope
Chapter 6: Antigen-Antibody Interactions I. Strength of Ag-Ab interactions A. Antibody Affinity - strength of total noncovalent interactions between single Ag-binding site on an Ab and a single epitope
Identifying Celiac Disease and Gluten Sensitivity with Minimally Invasive Testing
 Identifying Celiac Disease and Gluten Sensitivity with Minimally Invasive Testing Enjoy this white paper by Jack A. Maggiore, PhD, MT(ASCP), Assistant Director, Chemistry Laboratory and R&D at Doctor s
Identifying Celiac Disease and Gluten Sensitivity with Minimally Invasive Testing Enjoy this white paper by Jack A. Maggiore, PhD, MT(ASCP), Assistant Director, Chemistry Laboratory and R&D at Doctor s
Rheumatoid arthritis
 Rheumatoid arthritis Rheumatoid arthritis Chronic multisystem disease Unknown cause Characteristic feature persistent inflammation of synovia in symmetric peripheral joints Synovial inflammation cartilage
Rheumatoid arthritis Rheumatoid arthritis Chronic multisystem disease Unknown cause Characteristic feature persistent inflammation of synovia in symmetric peripheral joints Synovial inflammation cartilage
Pulmonary-Renal Syndromes. Spyros A Papiris MD, FCCP Athens Medical School National and Kapodistrian University of Athens, Greece
 Pulmonary-Renal Syndromes Spyros A Papiris MD, FCCP Athens Medical School National and Kapodistrian University of Athens, Greece Pulmonary-Renal Syndromes are a combination of Diffuse Alveolar Hemorrhage
Pulmonary-Renal Syndromes Spyros A Papiris MD, FCCP Athens Medical School National and Kapodistrian University of Athens, Greece Pulmonary-Renal Syndromes are a combination of Diffuse Alveolar Hemorrhage
Serology: Fluorescent antibody tests and other tests employing conjugated antibodies
 Serology: Fluorescent antibody tests and other tests employing conjugated antibodies Authors: Adapted by Prof M van Vuuren. Originally compiled by Dr RW Worthington. (Retired) Licensed under a Creative
Serology: Fluorescent antibody tests and other tests employing conjugated antibodies Authors: Adapted by Prof M van Vuuren. Originally compiled by Dr RW Worthington. (Retired) Licensed under a Creative
Rheumatoid Arthritis. Disease RA Final.indd 2 15. 6. 10. 11:23
 Rheumatoid Arthritis Disease RA Final.indd 2 15. 6. 10. 11:23 Understanding what to expect can help you prepare for your transition into treatment. Rheumatoid Arthritis What You Need To Know About Rheumatoid
Rheumatoid Arthritis Disease RA Final.indd 2 15. 6. 10. 11:23 Understanding what to expect can help you prepare for your transition into treatment. Rheumatoid Arthritis What You Need To Know About Rheumatoid
1) Siderophores are bacterial proteins that compete with animal A) Antibodies. B) Red blood cells. C) Transferrin. D) White blood cells. E) Receptors.
 Prof. Lester s BIOL 210 Practice Exam 4 (There is no answer key. Please do not email or ask me for answers.) Chapters 15, 16, 17, 19, HIV/AIDS, TB, Quorum Sensing 1) Siderophores are bacterial proteins
Prof. Lester s BIOL 210 Practice Exam 4 (There is no answer key. Please do not email or ask me for answers.) Chapters 15, 16, 17, 19, HIV/AIDS, TB, Quorum Sensing 1) Siderophores are bacterial proteins
Rheumatoid Arthritis
 Rheumatoid Arthritis Rheumatoid arthritis (RA) is an autoimmune disease that causes chronic inflammation of the joints. Autoimmune diseases are illnesses that occur when the body's tissues are mistakenly
Rheumatoid Arthritis Rheumatoid arthritis (RA) is an autoimmune disease that causes chronic inflammation of the joints. Autoimmune diseases are illnesses that occur when the body's tissues are mistakenly
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE. Health Technology Appraisal
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Health Technology Appraisal Adalimumab, etanercept, infliximab, rituximab and abatacept for the treatment of rheumatoid arthritis after the failure
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Health Technology Appraisal Adalimumab, etanercept, infliximab, rituximab and abatacept for the treatment of rheumatoid arthritis after the failure
Severe rheumatoid arthritis (a disease that causes inflammation of the joints),where MabThera is given intravenously together with methotrexate.
 EMA/614203/2010 EMEA/H/C/000165 EPAR summary for the public rituximab This is a summary of the European public assessment report (EPAR) for. It explains how the Committee for Medicinal Products for Human
EMA/614203/2010 EMEA/H/C/000165 EPAR summary for the public rituximab This is a summary of the European public assessment report (EPAR) for. It explains how the Committee for Medicinal Products for Human
(Intro to Arthritis with a. Arthritis) Manager of Education & Services for the Vancouver Island Region of The Arthritis Society
 Arthritis 101 (Intro to Arthritis with a Focus on Rheumatoid Arthritis) by Cari Taylor by Cari Taylor Manager of Education & Services for the Vancouver Island Region of The Arthritis Society What You Will
Arthritis 101 (Intro to Arthritis with a Focus on Rheumatoid Arthritis) by Cari Taylor by Cari Taylor Manager of Education & Services for the Vancouver Island Region of The Arthritis Society What You Will
Urinalysis and Body Fluids CRg. Synovial Fluid. Synovial Fluid. Unit 4. Composition and formation. Functions. Reasons for analysis.
 Urinalysis and Body Fluids CRg Unit 4 Synovial Fluid Synovial Fluid Composition and formation Secreted by cells of synovial membrane Very viscous, clear ultrafiltrate of plasma Contains Hyaluronic acid
Urinalysis and Body Fluids CRg Unit 4 Synovial Fluid Synovial Fluid Composition and formation Secreted by cells of synovial membrane Very viscous, clear ultrafiltrate of plasma Contains Hyaluronic acid
chronic leukemia lymphoma myeloma differentiated 14 September 1999 Pre- Transformed Ig Surface Surface Secreted Myeloma Major malignant counterpart
 Disease Usual phenotype acute leukemia precursor chronic leukemia lymphoma myeloma differentiated Pre- B-cell B-cell Transformed B-cell Plasma cell Ig Surface Surface Secreted Major malignant counterpart
Disease Usual phenotype acute leukemia precursor chronic leukemia lymphoma myeloma differentiated Pre- B-cell B-cell Transformed B-cell Plasma cell Ig Surface Surface Secreted Major malignant counterpart
EXERCISE 5: ERYTHROCYTES SEDIMENTATION RATE - ESR, SED RATE
 EXERCISE 5: ERYTHROCYTES SEDIMENTATION RATE - ESR, SED RATE Textbook: Skills: None 10 points Objectives: 1. State the principle of the Erythrocytes Sedimentation Rate - ESR. 2. List two factors which may
EXERCISE 5: ERYTHROCYTES SEDIMENTATION RATE - ESR, SED RATE Textbook: Skills: None 10 points Objectives: 1. State the principle of the Erythrocytes Sedimentation Rate - ESR. 2. List two factors which may
INTRODUCTION Thrombophilia deep vein thrombosis DVT pulmonary embolism PE inherited thrombophilia
 INTRODUCTION Thrombophilia (Hypercoagulability) is a condition in which a person forms blood clots more than normal. Blood clots may occur in the arms or legs (e.g., deep vein thrombosis DVT), the lungs
INTRODUCTION Thrombophilia (Hypercoagulability) is a condition in which a person forms blood clots more than normal. Blood clots may occur in the arms or legs (e.g., deep vein thrombosis DVT), the lungs
Measles (Rubeola) IgM ELISA Catalog No. CB40-101-325099 (96 Tests)
 For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE Measles test is an enzyme linked immunosorbent assay (ELISA) for the detection of IgG class antibodies to Measles (Rubeola) in
For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE Measles test is an enzyme linked immunosorbent assay (ELISA) for the detection of IgG class antibodies to Measles (Rubeola) in
Anti-nuclear (ANA), anti-double stranded DNA (dsdna) and anti-extractable nuclear antigen (ENA) antibodies
 Autoimmune testing The interpretation of all autoantibody tests is highly dependent on the likelihood of disease in the patient. The results should always be interpreted with the clinical features of the
Autoimmune testing The interpretation of all autoantibody tests is highly dependent on the likelihood of disease in the patient. The results should always be interpreted with the clinical features of the
www.gbo.com/bioscience Tissue Culture 1 Cell/ Microplates 2 HTS- 3 Immunology/ HLA 4 Microbiology/ Bacteriology Purpose Beakers 5 Tubes/Multi-
 11 Cryo 5 Tubes/Multi 2 HTS 3 Immunology / Immunology Technical Information 3 I 2 96 Well ELISA 3 I 4 96 Well ELISA Strip Plates 3 I 6 8 Well Strip Plates 3 I 7 12 Well Strip Plates 3 I 8 16 Well Strip
11 Cryo 5 Tubes/Multi 2 HTS 3 Immunology / Immunology Technical Information 3 I 2 96 Well ELISA 3 I 4 96 Well ELISA Strip Plates 3 I 6 8 Well Strip Plates 3 I 7 12 Well Strip Plates 3 I 8 16 Well Strip
HuCAL Custom Monoclonal Antibodies
 HuCAL Custom Monoclonal Antibodies Highly Specific Monoclonal Antibodies in just 8 Weeks PROVEN, HIGHLY SPECIFIC, HIGH AFFINITY ANTIBODIES IN 8 WEEKS WITHOUT HuCAL PLATINUM IMMUNIZATION (Human Combinatorial
HuCAL Custom Monoclonal Antibodies Highly Specific Monoclonal Antibodies in just 8 Weeks PROVEN, HIGHLY SPECIFIC, HIGH AFFINITY ANTIBODIES IN 8 WEEKS WITHOUT HuCAL PLATINUM IMMUNIZATION (Human Combinatorial
Interpreting Rheumatologic Lab Tests
 Interpreting Rheumatologic Lab Tests Jonathan Graf, M.D. Associate Professor of Clinical Medicine University of California, San Francisco Division of Rheumatology San Francisco General Hospital The black
Interpreting Rheumatologic Lab Tests Jonathan Graf, M.D. Associate Professor of Clinical Medicine University of California, San Francisco Division of Rheumatology San Francisco General Hospital The black
Management in the pre-hospital setting
 Management in the pre-hospital setting Inflammation of the joints Two main types: Osteoarthritis - cartilage loss from wear and tear Rheumatoid arthritis - autoimmune disorder Affects all age groups,
Management in the pre-hospital setting Inflammation of the joints Two main types: Osteoarthritis - cartilage loss from wear and tear Rheumatoid arthritis - autoimmune disorder Affects all age groups,
IgE (Human) ELISA Kit
 IgE (Human) ELISA Kit Catalog Number KA0216 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
IgE (Human) ELISA Kit Catalog Number KA0216 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
2.0 Rationale, Purpose and Scope... 5. 4.0 Definitions... 6. 5.0 General Principles... 7
 Table of Contents Principles of IVD Medical Devices Classification 1.0 Introduction... 4 2.0 Rationale, Purpose and Scope... 5 2.1 Rationale... 5 2.2 Purpose... 5 2.3 Scope... 5 3.0 References... 5 4.0
Table of Contents Principles of IVD Medical Devices Classification 1.0 Introduction... 4 2.0 Rationale, Purpose and Scope... 5 2.1 Rationale... 5 2.2 Purpose... 5 2.3 Scope... 5 3.0 References... 5 4.0
CANADIAN RULE BASED CLASSIFICATION SYSTEM (IVDD) Life Sciences British Columbia NRC-Industry Research Assistance Program
 CANADIAN RULE BASED CLASSIFICATION SYSTEM (IVDD) Life Sciences British Columbia NRC-Industry Research Assistance Program Health Canada Regulations on Medical Devices Vancouver, B.C. October 29, 2007 Nancy
CANADIAN RULE BASED CLASSIFICATION SYSTEM (IVDD) Life Sciences British Columbia NRC-Industry Research Assistance Program Health Canada Regulations on Medical Devices Vancouver, B.C. October 29, 2007 Nancy
serum protein and A/ G ratio
 serum protein and A/ G ratio Blood plasma contains at least 125 individual proteins. Serum ( as contrasted with plasma) is deficient in those coagulation protein which are consumed during the process of
serum protein and A/ G ratio Blood plasma contains at least 125 individual proteins. Serum ( as contrasted with plasma) is deficient in those coagulation protein which are consumed during the process of
Pharmacology of the Respiratory Tract: COPD and Steroids
 Pharmacology of the Respiratory Tract: COPD and Steroids Dr. Tillie-Louise Hackett Department of Anesthesiology, Pharmacology and Therapeutics University of British Columbia Associate Head, Centre of Heart
Pharmacology of the Respiratory Tract: COPD and Steroids Dr. Tillie-Louise Hackett Department of Anesthesiology, Pharmacology and Therapeutics University of British Columbia Associate Head, Centre of Heart
QUANTA Lite TM CCP 3.1 IgG/IgA ELISA 704550 For In Vitro Diagnostic Use CLIA Complexity: High
 QUANTA Lite TM CCP 3.1 IgG/IgA ELISA 704550 For In Vitro Diagnostic Use CLIA Complexity: High Intended Use The QUANTA Lite TM CCP3.1 IgG/IgA ELISA is a semiquantitative enzyme-linked immunosorbent assay
QUANTA Lite TM CCP 3.1 IgG/IgA ELISA 704550 For In Vitro Diagnostic Use CLIA Complexity: High Intended Use The QUANTA Lite TM CCP3.1 IgG/IgA ELISA is a semiquantitative enzyme-linked immunosorbent assay
specific B cells Humoral immunity lymphocytes antibodies B cells bone marrow Cell-mediated immunity: T cells antibodies proteins
 Adaptive Immunity Chapter 17: Adaptive (specific) Immunity Bio 139 Dr. Amy Rogers Host defenses that are specific to a particular infectious agent Can be innate or genetic for humans as a group: most microbes
Adaptive Immunity Chapter 17: Adaptive (specific) Immunity Bio 139 Dr. Amy Rogers Host defenses that are specific to a particular infectious agent Can be innate or genetic for humans as a group: most microbes
Blood Transfusion. There are three types of blood cells: Red blood cells. White blood cells. Platelets.
 Blood Transfusion Introduction Blood transfusions can save lives. Every second, someone in the world needs a blood transfusion. Blood transfusions can replace the blood lost from a serious injury or surgery.
Blood Transfusion Introduction Blood transfusions can save lives. Every second, someone in the world needs a blood transfusion. Blood transfusions can replace the blood lost from a serious injury or surgery.
SARCOIDOSIS. Signs and symptoms associated with specific organ involvement can include the following:
 SARCOIDOSIS Sarcoidosis is a disease that occurs when areas of inflammation develop in different organs of the body. Very small clusters of inflammation, called granulomas, are seen with sarcoidosis. They
SARCOIDOSIS Sarcoidosis is a disease that occurs when areas of inflammation develop in different organs of the body. Very small clusters of inflammation, called granulomas, are seen with sarcoidosis. They
INTRODUCTION Thrombophilia deep vein thrombosis DVT pulmonary embolism PE inherited thrombophilia
 INTRODUCTION Thrombophilia (Hypercoagulability) is a condition in which a person forms blood clots more than normal. Blood clots may occur in the arms or legs (e.g., deep vein thrombosis DVT), the lungs
INTRODUCTION Thrombophilia (Hypercoagulability) is a condition in which a person forms blood clots more than normal. Blood clots may occur in the arms or legs (e.g., deep vein thrombosis DVT), the lungs
Immunoglobulin E (IgE) concentrations in Human. Immunoglobulin E (IgE) Human ELISA Kit
 ab108650 Immunoglobulin E (IgE) Human ELISA Kit Instructions for Use For the quantitative measurement of Immunoglobulin E (IgE) concentrations in Human serum. This product is for research use only and
ab108650 Immunoglobulin E (IgE) Human ELISA Kit Instructions for Use For the quantitative measurement of Immunoglobulin E (IgE) concentrations in Human serum. This product is for research use only and
PROPOSED DOCUMENT. Global Harmonization Task Force. Title: Principles of In Vitro Diagnostic (IVD) Medical Devices Classification
 SG1(PD)/N045R12 PROPOSED DOCUMENT Global Harmonization Task Force Title: Principles of In Vitro Diagnostic (IVD) Medical Devices Classification Authoring Group: Study Group 1 of the Global Harmonization
SG1(PD)/N045R12 PROPOSED DOCUMENT Global Harmonization Task Force Title: Principles of In Vitro Diagnostic (IVD) Medical Devices Classification Authoring Group: Study Group 1 of the Global Harmonization
Rheumatoid Arthritis. Treating Inflammation. Sequoia Education Systems, Inc http://www.functionalmedicineuniversity.com 1. How is RA Diagnosed?
 Rheumatoid Arthritis Causes, Assessment and Treatment How is RA Diagnosed? The following labs make up the basics of ruling in or out RA. Citrullinated Peptide (anti-ccp) Antibodies Rheumatoid factor Erythrocyte
Rheumatoid Arthritis Causes, Assessment and Treatment How is RA Diagnosed? The following labs make up the basics of ruling in or out RA. Citrullinated Peptide (anti-ccp) Antibodies Rheumatoid factor Erythrocyte
Some Immunological Test. Presented by Alaa Faeiz Ashwaaq Dyaa Aseel Abd AL-Razaq Supervised by D.Feras
 Some Immunological Test Presented by Alaa Faeiz Ashwaaq Dyaa Aseel Abd AL-Razaq Supervised by D.Feras Alaa Faeiz Antigen -Antibody Reactions. Antigen antibody reactions are performed to determine the presence
Some Immunological Test Presented by Alaa Faeiz Ashwaaq Dyaa Aseel Abd AL-Razaq Supervised by D.Feras Alaa Faeiz Antigen -Antibody Reactions. Antigen antibody reactions are performed to determine the presence
P R O D U C T S CATALOG 2011-2012
 P R O D U C T S CATALOG 2011-2012 INSTITUTE FOR THE APPLICATION OF NUCLEAR ENERGY - INEP Banatska 31b 11080 Zemun Belgrade Serbia Tel: (+381 11) 2619 252, 2618 696, 2199 949 Fax: (+381 11) 2618 724 www.inep.co.rs
P R O D U C T S CATALOG 2011-2012 INSTITUTE FOR THE APPLICATION OF NUCLEAR ENERGY - INEP Banatska 31b 11080 Zemun Belgrade Serbia Tel: (+381 11) 2619 252, 2618 696, 2199 949 Fax: (+381 11) 2618 724 www.inep.co.rs
Arthritis and Rheumatology. Antoni Chan MBChB, FRCP, PhD Consultant Rheumatologist Royal Berkshire NHS Foundation Trust
 Antoni Chan MBChB, FRCP, PhD Consultant Rheumatologist Royal Berkshire NHS Foundation Trust Rheumatology Investigation, Diagnosis, Treatment The challenge 8 billion a year in cost 700,000 people suffering
Antoni Chan MBChB, FRCP, PhD Consultant Rheumatologist Royal Berkshire NHS Foundation Trust Rheumatology Investigation, Diagnosis, Treatment The challenge 8 billion a year in cost 700,000 people suffering
Dengue IgM ELISA. For the quantitative determination of IgM-class antibodies to Dengue Virus in serum.
 Dengue IgM ELISA For the quantitative determination of IgM-class antibodies to Dengue Virus in serum. For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number: 20-DEMHU-E01 Size: 96
Dengue IgM ELISA For the quantitative determination of IgM-class antibodies to Dengue Virus in serum. For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number: 20-DEMHU-E01 Size: 96
The Immune System: A Tutorial
 The Immune System: A Tutorial Modeling and Simulation of Biological Systems 21-366B Shlomo Ta asan Images taken from http://rex.nci.nih.gov/behindthenews/uis/uisframe.htm http://copewithcytokines.de/ The
The Immune System: A Tutorial Modeling and Simulation of Biological Systems 21-366B Shlomo Ta asan Images taken from http://rex.nci.nih.gov/behindthenews/uis/uisframe.htm http://copewithcytokines.de/ The
Once the immune system is triggered, cells migrate from the blood into the joints and produce substances that cause inflammation.
 HealthExchange Points For Your Joints An Arthritis Talk Howard Epstein, MD Orthopaedic & Rheumatologic Institute Rheumatic & Immunologic Disease Cleveland Clinic Beachwood Family Health & Surgery Center
HealthExchange Points For Your Joints An Arthritis Talk Howard Epstein, MD Orthopaedic & Rheumatologic Institute Rheumatic & Immunologic Disease Cleveland Clinic Beachwood Family Health & Surgery Center
Medical Policy Anti-CCP Testing for Rheumatoid Arthritis
 Medical Policy Anti-CCP Testing for Rheumatoid Arthritis Table of Contents Policy: Commercial Coding Information Information Pertaining to All Policies Policy: Medicare Description References Authorization
Medical Policy Anti-CCP Testing for Rheumatoid Arthritis Table of Contents Policy: Commercial Coding Information Information Pertaining to All Policies Policy: Medicare Description References Authorization
High Resolution Epitope Mapping of Human Autoimmune Sera against Antigens CENPA and KDM6B. PEPperPRINT GmbH Heidelberg, 06/2014
 High Resolution Epitope Mapping of Human Autoimmune Sera against Antigens CENPA and KDM6B PEPperPRINT GmbH Heidelberg, 06/2014 Introduction This report describes the epitope mapping of autoimmune sera
High Resolution Epitope Mapping of Human Autoimmune Sera against Antigens CENPA and KDM6B PEPperPRINT GmbH Heidelberg, 06/2014 Introduction This report describes the epitope mapping of autoimmune sera
Summary of the risk management plan (RMP) for Accofil (filgrastim)
 EMA/475472/2014 Summary of the risk management plan (RMP) for Accofil (filgrastim) This is a summary of the risk management plan (RMP) for Accofil, which details the measures to be taken in order to ensure
EMA/475472/2014 Summary of the risk management plan (RMP) for Accofil (filgrastim) This is a summary of the risk management plan (RMP) for Accofil, which details the measures to be taken in order to ensure
Media Release. Basel, 11 June 2009. RA patients with enhanced response identified
 Media Release Basel, 11 June 2009 New data demonstrate the ability of MabThera to reduce the progression of joint damage when used as a first-line biologic treatment in rheumatoid arthritis RA patients
Media Release Basel, 11 June 2009 New data demonstrate the ability of MabThera to reduce the progression of joint damage when used as a first-line biologic treatment in rheumatoid arthritis RA patients
Your partner in immunology
 Your partner in immunology Expertise Expertise Reactivity Reactivity Quality Quality Advice Advice Who are we? Specialist of antibody engineering Covalab is a French biotechnology company, specialised
Your partner in immunology Expertise Expertise Reactivity Reactivity Quality Quality Advice Advice Who are we? Specialist of antibody engineering Covalab is a French biotechnology company, specialised
Thymus Cancer. This reference summary will help you better understand what thymus cancer is and what treatment options are available.
 Thymus Cancer Introduction Thymus cancer is a rare cancer. It starts in the small organ that lies in the upper chest under the breastbone. The thymus makes white blood cells that protect the body against
Thymus Cancer Introduction Thymus cancer is a rare cancer. It starts in the small organ that lies in the upper chest under the breastbone. The thymus makes white blood cells that protect the body against
Name (print) Name (signature) Period. (Total 30 points)
 AP Biology Worksheet Chapter 43 The Immune System Lambdin April 4, 2011 Due Date: Thurs. April 7, 2011 You may use the following: Text Notes Power point Internet One other person in class "On my honor,
AP Biology Worksheet Chapter 43 The Immune System Lambdin April 4, 2011 Due Date: Thurs. April 7, 2011 You may use the following: Text Notes Power point Internet One other person in class "On my honor,
Information on Rheumatoid Arthritis
 Information on Rheumatoid Arthritis Table of Contents About Rheumatoid Arthritis 1 Definition 1 Signs and symptoms 1 Causes 1 Risk factors 1 Test and diagnosis 2 Treatment options 2 Lifestyle 3 References
Information on Rheumatoid Arthritis Table of Contents About Rheumatoid Arthritis 1 Definition 1 Signs and symptoms 1 Causes 1 Risk factors 1 Test and diagnosis 2 Treatment options 2 Lifestyle 3 References
Anti-ds-DNA-A ELISA. For the quantitative and qualitative determination of IgA antibodies against double-stranded DNA (dsdna) in human serum
 Anti-ds-DNA-A ELISA For the quantitative and qualitative determination of IgA antibodies against double-stranded DNA (dsdna) in human serum For Research Use Only. Not For Use In Diagnostic Procedures.
Anti-ds-DNA-A ELISA For the quantitative and qualitative determination of IgA antibodies against double-stranded DNA (dsdna) in human serum For Research Use Only. Not For Use In Diagnostic Procedures.
TREATING AUTOIMMUNE DISEASES WITH HOMEOPATHY. Dr. Stephen A. Messer, MSEd, ND, DHANP Professor and Chair of Homeopathic Medicine
 TREATING AUTOIMMUNE DISEASES WITH HOMEOPATHY Dr. Stephen A. Messer, MSEd, ND, DHANP Professor and Chair of Homeopathic Medicine AUTOIMMUNE DISEASES An autoimmune disorder occurs when the body s immune
TREATING AUTOIMMUNE DISEASES WITH HOMEOPATHY Dr. Stephen A. Messer, MSEd, ND, DHANP Professor and Chair of Homeopathic Medicine AUTOIMMUNE DISEASES An autoimmune disorder occurs when the body s immune
The Immune System and Disease
 Chapter 40 The Immune System and Disease Section 40 1 Infectious Disease (pages 1029 1033) This section describes the causes of disease and explains how infectious diseases are transmitted Introduction
Chapter 40 The Immune System and Disease Section 40 1 Infectious Disease (pages 1029 1033) This section describes the causes of disease and explains how infectious diseases are transmitted Introduction
