BONE-GRAFT SUBSTITUTES: FACTS, FICTIONS & APPLICATIONS
|
|
|
- Dwight Harrell
- 7 years ago
- Views:
Transcription
1 BONE-GRAFT SUBSTITUTES: FACTS, FICTIONS & APPLICATIONS AMERICAN ACADEMY OF ORTHOPAEDIC SURGEONS 73rd Annual Meeting March 22-26, 2006 Chicago, Illinois ORTHOPAEDIC DEVICE FORUM Prepared by: A. Seth Greenwald, D.Phil.(Oxon) Scott D. Boden, M.D. Victor M. Goldberg, M.D. Michael Yaszemski, M.D. Christine S. Heim, B.Sc. Acknowledgements: AlloSource Biomet/EBI/Interpore Cross International Exactech, Inc. IsoTis OrthoBiologics, Inc. LifeNet, Inc. Medtronic Sofamor Danek Musculoskeletal Transplant Foundation Orthovita, Inc. Osteotech, Inc. Regeneration Technologies, Inc. Stryker Biotech Synthes USA Wright Medical Technology, Inc. Zimmer, Inc.
2 A REALITY CHECK It is estimated that more than 500,000 bone-grafting procedures are performed annually in the United States, with approximately half of these procedures related to spine fusion. These numbers easily double on a global basis and indicate a shortage in the availability of musculoskeletal donor tissue traditionally used in these reconstructions. (Figure 1) Donors ,433 9,443 14,313 14,120 16,319 15,255 19,250 19, Figure 1: U.S. trends in musculoskeletal tissue donors. Source: AATB Annual Survey U.S. Sales ($billions) Figure 2: U.S. sales of bone graft and bone substitutes Source: Orthopedic Network News BMP Platelet Concentrators Bone Substitutes Machined Bone DBM Products Allograft Bone This reality has stimulated a proliferation of corporate interest in supplying what is seen as a growing market in bone replacement materials. (Figure 2) These graft alternatives are subjected to varying degrees of regulatory scrutiny, and thus their true safety and effectiveness in patients may not be known prior to their use by orthopaedic surgeons. It is thus important to gain insight into this emerging class of bone-substitute alternatives. THE PHYSIOLOGY OF BONE GRAFTING The biology of bone grafts and their substitutes is appreciated from an understanding of the bone formation processes of Osteogenesis, Osteoinduction and Osteoconduction. Graft Osteogenesis: The cellular elements within a donor graft, which survive transplantation and synthesize new bone at the recipient site. Graft Osteoinduction: New bone realized through the active recruitment of host mesenchymal stem cells from the surrounding tissue, which differentiate into bone-forming osteoblasts. This process is facilitated by the presence of growth factors within the graft, principally bone morphogenic proteins (BMPs). Graft Osteoconduction: The facilitation of blood-vessel incursion and new-bone formation into a defined passive trellis structure. All bone graft and bone-graft-substitute materials can be described through these processes. While fresh autologous graft has the capability of supporting new bone growth by all three means, it may not be necessary for a bone graft replacement to have all three properties inherently in order to be clinically efficacious. When inductive molecules are locally delivered on a scaffold, ultimately mesenchymal stem cells are attracted to the site and are capable of reproducibly inducing new bone formation provided minimal concentration and dose thresholds are met. In some clinical studies, osteoinductive agents have been shown to potentially perform superiorly. However, bone marrow aspirate applied to osteoconductive scaffolds are still reliant on the local mechanical and biological signals in order to ultimately form bone. For this reason, these materials are typically used as an adjunct in order to retain efficacy equivalent to autograft. Similarly, osteoconductive materials work well when filling non-critical size defects that would normally heal easily. However, in more challenging critical size defects, either fresh autologous bone graft or osteoinductive agents appear necessary for healing.
3 BONE AUTOGRAFTS Fresh autogenous cancellous and, to a lesser degree, cortical bone are benchmark graft materials that allograft and bone substitutes attempt to match in in vivo performance. They incorporate all of the mentioned properties, are harvested at both primary and secondary surgical sites, and have no associated risk of viral transmission. Furthermore, they offer structural support to implanted devices and, ultimately, become mechanically efficient structures as they are incorporated into surrounding bone through creeping substitution. The availability of autografts is, however, limited and harvest is often associated with donor-site morbidity. BONE ALLOGRAFTS The advantages of bone allograft recovered from deceased donor sources include its ready availability in various shapes and sizes, avoidance of the need to sacrifice host structures and no donor-site morbidity. Bone allografts are distributed through regional tissue banks and by most major orthopaedic and spinal companies. Still, the grafts are not without controversy, particularly regarding their association with the transmission of infectious agents, a concern virtually eliminated through tissue-processing and sterilization. However, uncontrolled and unvalidated processing and irradiation protocols may alter graft biomechanical and biochemical properties. It is critical to know your tissue bank provider to ensure their processing and preservation methods do not negatively alter the biomechanical and biochemical properties of the tissues intended for a particular clinical use. A comparison of Bone Graft Autograft Cancellous Cortical Allograft Cancellous Frozen Freeze-Dry Cortical Frozen Freeze-Dry Demineralized Allogeneic Cancellous Chips Structural Strength properties of allograft and autograft bone is shown in Figure 3. Often, in complex surgical reconstructions, these materials are used in tandem with implants and fixation devices. (Figure 4) Osteo- Conduction Osteo- Induction Figure 3: Comparative properties of bone grafts Osteogenesis (a) (b) (c) (d) Figure 4: (a) A 17-year old patient with osteosarcoma of the distal part of the femur with no extraosseous extension or metastatic disease. Following chemotherapy, (b) limb salvage with wide resection was performed. Femoral reconstruction with the use of an autogenous cortical fibular graft, iliac crest bone chips, morselized cancellous autograft and structural allograft combined with internal fixation. (c) Graft incorporation and remodeling are seen at 3 years. (d) Limb restoration is noted at 10 years following resection. (The intramedullary rod was removed at 5 years.)
4 BONE GRAFT SUBSTITUTES The ideal bone-graft substitute is biocompatible, bioresorbable, osteoconductive, osteoinductive, structurally similar to bone, easy to use, and cost-effective. Within these parameters a growing number of bone alternatives are commercially available for orthopaedic applications, including reconstruction of cavitary bone deficiency and augmentation in situations of segmental bone loss and spine fusion. They are variable in their composition and their claimed mechanisms of action. Those containing growth factors in their composition inclusive of rhbmp-2 (INFUSE Bone Graft) and rhbmp-7 (OP-1 ) demonstrate in clinical application, while the remainder are predominantly osteoconductive in their claims. All offer minimal structural integrity. A series of case examples demonstrate their mechanisms of action through the healing process. (Figures 5-8) (a) (b) (c) Figure 5: (a) A 61-year old male with a comminuted pilon fracture sustained in a motor vehicle accident. (b) After 2 months with an external fixator, definitive fixation of the tibia with a percutaneous injection of IGNITE (Wright Medical Technology, Inc., Arlington, TN) graft to bridge the slow-healing fracture. (c) Two years post-op, the fracture is consolidated and the patient is ambulating pain-free. (a) (b) (c) (d) Figure 6: (a) A 58-year old obese female with a fracture of the right femur after falling from a horse. Treatments included plating with cortical struts and DBM. (b) Nine months following third surgery the plate and several screws are broken. (c) Three months after treatment with IM rod fixation and OP-1 Implant (Stryker Biotech, Hopkinton, MA) she was full weight bearing, with full range of motion and pain free. (d) Nine months postoperative.
5 (a) (b) Figure 7: (a) AP and Lateral radiographs, 67-year old female with depressed fracture of the lateral tibial plateau. (b) AP and Lateral radiographs 12 months after ORIF with filling the defect with rian SRS (Synthes USA, Paoli, PA). loss of reduction of the plateau surface is noted, fracture completely healed. Figure 8: (a) A 29-year old male with a Grade IIIB oblique fracture of the distal tibia from a motorcycle accident. (b) Six weeks after being treated with an unreamed locked nail and INFUSE Bone Graft (Medtronic Sofamor Danek, Memphis, TN). (c) Patient full weight bearing and radiographically healed 20 weeks post-operative. (a) (b) (c) BURDEN OF PROOF It is reasonable to assume that not all bone-substitute products will perform analogously. Thus, a quandary of choice confronts the orthopaedic surgeon. As a first principle, it is important to appreciate that different healing environments (e.g., a metaphyseal defect, a long-bone fracture, an interbody spine fusion, or a posterolateral spine fusion) have different levels of difficulty in forming new bone. For example, a metaphyseal defect will permit the successful use of many purely osteoconductive materials. In contrast, a posterolateral spine fusion will not succeed if purely osteoconductive materials are used as a stand-alone substitute. Thus, validation of any bone-graft substitute in one clinical site may not necessarily predict its performance in another location. A second principle is to seek the highest burden of proof reported from preclinical studies to justify the use of an osteoinductive graft material or the choice of one brand over another. Whether it is more difficult to make bone in humans than it is in cell-culture or rodent models, with a progressive hierarchy of difficulty in more complex species, has not been clearly determined. Only human trials can determine the efficacy of bone-graft substitutes in humans as well as their site-specific effectiveness. In this latter context, surgeons should practice evidence based medicine and tailor treatment for patients based on the published medical literature and the levels of evidence claimed. (Wright JG, Swiontkowski MF, Heckman JD. Introducing levels of evidence to the journal. J Bone Joint Surg Am Jan;85(1):1-3.)
6 BURDEN OF PROOF (Cont d.) A third principle requiring burden of proof specifically pertains to products that are not subjected to high levels of regulatory scrutiny, such as 100% demineralized bone matrix (DBM) or platelet gels containing autologous growth factors. Such products are considered to involve minimal manipulation of cells or tissue and are thus regulated as tissue rather than as devices, unless they are configured with an additive and then require 510(k) clearance. As a result, there is no standardized level of proof of safety and effectiveness required before these products are marketed and are used in patients. While these products may satisfy the technical definition of minimal manipulation, there is a risk that they will not produce the expected results in humans when there has been little or no testing in relevant animal models. FUTURE Recent FDA approvals include the use of PMA approved rhbmp-2 (INFUSE Bone Graft) as an autograft replacement in spinal fusion and treatment of open tibia fractures; rhbmp-7 (OP-1 ) is HDE approved as an autograft substitute for tibial non-unions; and rhbmp-7 (OP-1 Putty) is HDE approved as an alternative to autograft in compromised patients requiring revision posterolateral (intertransverse) lumbar spinal fusion, for whom autologous bone and bone marrow recovery are not feasible or are not expected to promote fusion. These clinical applications demonstrate impressive osteoinductive capacity and pave the way for broader clinical applications. Their methods of administration include direct placement in the surgical site, but results have been more promising when the growth factors have been administered in combination with substrates to facilitate timed-release delivery and/or provide a material scaffold for bone formation. FDA regulatory imperatives will continue to determine their availability. Their cost/benefit ratio will ultimately influence clinical use. Further advances in tissue-engineering, the integration of the biological, physical and engineering sciences, will create new carrier constructs that regenerate and restore tissue to its functional state. These constructs are likely to encompass additional families of growth factors, evolving biological scaffolds and incorporation of mesenchymal stem cells. Ultimately, the development of ex vivo bioreactors capable of bone manufacture with the appropriate biomechanical cues will provide tissue-engineered constructs for direct use in the skeletal system. TAKE HOME MESSAGE The increasing number of bone-grafting procedures performed annually in the U.S. has created a shortage of cadaver allograft material and a need to increase musculoskeletal tissue donation. This has stimulated corporate interest in developing and supplying a rapidly expanding number of bone substitutes, the makeup of which includes natural, synthetic, human and animal-derived materials. Fresh autogenous cancellous and, to a lesser degree, cortical bone are the benchmark graft materials, which ideally both allograft and bone substitutes should match in in vivo performance. Their shortcomings include limited availability and donor-site morbidity. The advantages of allograft bone include availability in various sizes and shapes as well as avoidance of host structure sacrifice and donor-site morbidity. Transmission of infection, particularly the human immunodeficiency virus (HIV) has been virtually eliminated as a concern. The properties of the allograft should be confirmed with the tissue provider to ensure they correspond with their intended clinical use. The ideal bone-graft substitute is biocompatible, bioresorbable, osteoconductive, osteoinductive, structurally similar to bone, easy to use, and cost-effective. Currently marketed products are variable in their composition and their claimed mechanisms of action. It is reasonable that not all bone-substitute products will perform the same. Recent FDA approvals for specific uses of recombinant human growth factors (rhbmp-2 (INFUSE Bone Graft) and rhbmp-7 (OP-1 and OP-1 Putty)) are based on demonstrated osteoinductive capacity in human trials. Other applications will likely emerge. A quandary of choice confronts the orthopaedic surgeon. Caveat emptor! Selection should be based on reasoned burdens of proof. These include examination of the product claims and whether they are supported by preclinical and human studies in site-specific locations where they are to be utilized in surgery. It is imperative to appreciate the level of evidence claimed in the latter studies.
7 Summary of typical bone-graft substitutes that are commercially available Company Commercially available product Composition Commercially available forms Claimed mechanisms of action Burdens of proof FDA status AlloSource AlloFuse TM Heat sensitive copolymer with DBM Injectable gel and putty Cell culture ProOsteon 500R Coral HA composite Granular or block Biomet/EBI/ Interpore Cross InterGro DBM in a lecithin carrier Paste, putty and mix with coral/ha composite granules Every lot tested for BonePlast Calcium sulfate Powder/fluid mixed with hardenable paste Exactech IsoTis OrthoBiologics LifeNet Opteform Optefil TM Optecure TM OpteMx TM Accell DBM100 DBM and cortical cancellous chips in gelatin carrier DBM suspended in gelatin carrier DBM suspended in a hydrogel carrier HA/TCP biphasic combination DBM in a 100% DBMderived carrier Formable putty, syringeable cylinders and dry powder ready to be hydrated with blood or saline Injectable bone paste, dry powder ready to be hydrated with blood or saline Dry mix kit delivered with diluents and mix with patient s whole blood, autogenous bone Granules, sticks, rounded wedges, wedges and cylinders in several sizes Injectable putty Accell Total Bone Matrix TM Preformed 100% DBM Various sized strips Accell Connexus Optium DBM DBM in a DBM-based carrier with a reverse phase copolymer DBM combined with Glycerol DBM particles and IC Graft Chambers cancellous chips Cellect DBM OraGraft DBM fibers and cancellous chips Demineralized and mineralized cortical powder Injectable putty Formable putty and injectable gel Lyophilized and provided in various sizes Provided in a specialized cartridge Provided in various sized quantities Osteogenesis when mixed with autogenous bone graft Compressive strength of 400psi Osteogenesis and limited when combined with bone marrow aspirate Every lot tested in vivo for Every lot tested in vivo for Every lot tested in vivo for Every DBM lot tested for Every DBM lot tested for Every DBM lot tested for Designed to be used with blood, PRP or bone marrow to enhance DBM activity. Designed for the retention of osteoprogenitor cells Case studies 100% derived from DBM FDA clearance not required 100% derived from DBM FDA clearance not required Regulated under CFR 1270 and 1271 as a human tissue and 510(k) cleared Regulated under CFR 1270 and 1271 as a human tissue and 510(k) cleared 100% DBM FDA clearance not required
8 Company Commercially available product Composition Commercially available forms Claimed mechanisms of action Burdens of proof FDA status Medtronic Sofamor Danek INFUSE TM Bone Graft MasterGraft Granules rhbmp-2 protein on an absorbable collagen sponge Biphasic calcium phosphate Freeze-dried, sterile powder and sponge in several sizes Granules sponge (Level I and Level III data) PMA approved for fusion with spinal cage PMA approved for open tibia fractures with IM nail MasterGraft Matrix Calcium phosphate and collagen Compression resistant block MasterGraft Putty Calcium phosphate and collagen Injectable putty MTF/Synthes DBX DBM in sodium hyaluronate carrier Orthovita Vitoss and 80% b-tcp/20% collagen 100% b-tcp Osteotech Grafton DBM combined with Glycerol Regeneration Technologies Stryker Biotech Synthes BioSet TM OP-1 Implant OP-1 Putty Calstrux DBM combined with non-toxic natural gelatin carrier rhbmp-7 with type 1 bone collagen rhbmp-7 with type 1 bone collagen Tricalcium phosphate with carboxymethelcellulose Paste, putty mix and strip Putty, strip, flow, morsels and shapes Formable putty, injectable gel, putty mixed with chips, flexible sheets and matrix Injectable paste, injectable putty, strips and blocks with cortical cancellous chips Lyophilized powder reconstituted to form wet sand Lyophilized powder reconstituted to form putty Moldable putty rian SRS Calcium phosphate Injectable paste rian SRS Fast Set Putty chronos Calcium phosphate b-tricalcium phosphate Calceon 6 Calcium sulfate Pellets OSTEOSET MIIG X3 Surgical grade calcium sulfate High strength surgical grade calcium sulfate Moldable putty Granules, blocks and wedges Various sized pellets Minimally invasive injectable graft for compression fractures Potential Resorbable collagen scaffold Resorbable collagen scaffold Every lot tested in vivo for (Level I data) (Level I data) HDE approval for long bone nonunions HDE approval for revision posterolateral fusion Wright Medical Technology CELLPLEX Tricalcium phosphate Various sized granules ALLOMATRIX IGNITE DBM with/without CBM in surgical grade calcium sulfate powder DBM in surgical grade calcium sulfate powder to be mixed with bone marrow aspirate Zimmer CopiOS TM phosphate, dibasic and Mixture of calcium type I collagen Various volumes of injectable/ formable putty Percutaneous graft for problem fractures Sponge Osteogenic and limited when combined with bone marrow aspirate Cell culture Cell culture PMA Pre-Market Application; HDE Humanitarian Device Exemption AAOS 2006
BONE-GRAFT SUBSTITUTES: FACTS, FICTIONS & APPLICATIONS
 BONE-GRAFT SUBSTITUTES: FACTS, FICTIONS & APPLICATIONS AMERICAN ACADEMY OF ORTHOPAEDIC SURGEONS 70th Annual Meeting February 5-9, 2003 New Orleans, Louisiana COMMITTEE ON BIOLOGICAL IMPLANTS Prepared by:
BONE-GRAFT SUBSTITUTES: FACTS, FICTIONS & APPLICATIONS AMERICAN ACADEMY OF ORTHOPAEDIC SURGEONS 70th Annual Meeting February 5-9, 2003 New Orleans, Louisiana COMMITTEE ON BIOLOGICAL IMPLANTS Prepared by:
THE EVOLVING ROLE OF BONE-GRAFT SUBSTITUTES
 THE EVOLVING ROLE OF BONE-GRAFT SUBSTITUTES AMERICAN ACADEMY OF ORTHOPAEDIC SURGEONS 77TH ANNUAL MEETING MARCH 9-13, 2010 NEW ORLEANS, LOUISIANA ORTHOPAEDIC DEVICE FORUM PREPARED BY: A. SETH GREENWALD,
THE EVOLVING ROLE OF BONE-GRAFT SUBSTITUTES AMERICAN ACADEMY OF ORTHOPAEDIC SURGEONS 77TH ANNUAL MEETING MARCH 9-13, 2010 NEW ORLEANS, LOUISIANA ORTHOPAEDIC DEVICE FORUM PREPARED BY: A. SETH GREENWALD,
Summary of typical bone-graft substitutes that are commercially available - 2010
 Summary of typical bone-graft substitutes that are commercially available - 2010 Company AlloSource AlloFuse TM Heat sensitive copolymer with DBM Injectable gel and putty Cell culture Biomet Osteobiologics
Summary of typical bone-graft substitutes that are commercially available - 2010 Company AlloSource AlloFuse TM Heat sensitive copolymer with DBM Injectable gel and putty Cell culture Biomet Osteobiologics
Choosing Bone Graft Substitutes: Filling the Gaps on Evidence and Pricing. September 24, 2014
 Choosing Bone Graft Substitutes: Filling the Gaps on Evidence and Pricing September 24, 2014 Choosing Bone Graft Substitutes: Filling the Gaps on Evidence and Pricing ECRI Institute Webinar Presented by:
Choosing Bone Graft Substitutes: Filling the Gaps on Evidence and Pricing September 24, 2014 Choosing Bone Graft Substitutes: Filling the Gaps on Evidence and Pricing ECRI Institute Webinar Presented by:
Dental Bone Grafting Options. A review of bone grafting options for patients needing more bone to place dental implants
 Dental Bone Grafting Options A review of bone grafting options for patients needing more bone to place dental implants Dental Bone Grafting Options What is bone grafting? Bone grafting options Bone from
Dental Bone Grafting Options A review of bone grafting options for patients needing more bone to place dental implants Dental Bone Grafting Options What is bone grafting? Bone grafting options Bone from
chronos BOne VOid Filler Beta-Tricalcium Phosphate (b-tcp) bone graft substitute
 chronos BOne VOid Filler Beta-Tricalcium Phosphate (b-tcp) bone graft substitute chronos Bone Void Filler Osteoconductive Resorbable Synthetic chronos Granules and Preforms are synthetic, porous, osteoconductive,
chronos BOne VOid Filler Beta-Tricalcium Phosphate (b-tcp) bone graft substitute chronos Bone Void Filler Osteoconductive Resorbable Synthetic chronos Granules and Preforms are synthetic, porous, osteoconductive,
S YN T H E T I C C A N C E L LO U S B O N E
 S YN T H E T I C C A N C E L LO U S B O N E CELLPLEX TCP Graft is a synthetic cancellous scaffold that provides an optimum environment for cellular infiltration. Composed of tricalcium phosphate, the scaffold
S YN T H E T I C C A N C E L LO U S B O N E CELLPLEX TCP Graft is a synthetic cancellous scaffold that provides an optimum environment for cellular infiltration. Composed of tricalcium phosphate, the scaffold
ORTHOPEDICS BONE GRAFT & SYNTHETIC BONE. Gabelli & Company, Inc. January 27, 2004. Companies Highlighted
 One Corporate Center Rye, NY 10580-1422 Tel (914) 921-3700 Fax (914) 921-5098 www.gabelli.com January 27, 2004 Gabelli & Company, Inc. ORTHOPEDICS BONE GRAFT & SYNTHETIC BONE MD Series Threaded Bone Dowels
One Corporate Center Rye, NY 10580-1422 Tel (914) 921-3700 Fax (914) 921-5098 www.gabelli.com January 27, 2004 Gabelli & Company, Inc. ORTHOPEDICS BONE GRAFT & SYNTHETIC BONE MD Series Threaded Bone Dowels
DBX. Bone Graft Substitute. The osteoinductive power.
 DBX. Bone Graft Substitute. The osteoinductive power. Osteoinductivity for challenging bony fusion Excellent handling properties Certified safety The answer to bone voids Brochure for use outside of the
DBX. Bone Graft Substitute. The osteoinductive power. Osteoinductivity for challenging bony fusion Excellent handling properties Certified safety The answer to bone voids Brochure for use outside of the
INFUSE Bone Graft. Patient Information Brochure
 INFUSE Bone Graft Patient Information Brochure This Patient Guide is designed to help you decide whether or not to have surgery using INFUSE Bone Graft to treat your broken tibia (lower leg). There are
INFUSE Bone Graft Patient Information Brochure This Patient Guide is designed to help you decide whether or not to have surgery using INFUSE Bone Graft to treat your broken tibia (lower leg). There are
it will set at body temperature, and it will grow. UNIQUE FORMULATIONS
 BETTER BONE GRAFT FORMULATION. SUPERIOR BONE GRAFT HANDLING. When choosing a Demineralized Bone Matrix, remember that not all products are created equal. For excellent clinical performance and patient
BETTER BONE GRAFT FORMULATION. SUPERIOR BONE GRAFT HANDLING. When choosing a Demineralized Bone Matrix, remember that not all products are created equal. For excellent clinical performance and patient
BONE OR SOFT TISSUE HEALING AND FUSION ENHANCEMENT PRODUCTS
 #1 MEDICAL POLICY BONE OR SOFT TISSUE HEALING AND FUSION ENHANCEMENT PRODUCTS Policy Number: 2016T0410P Effective Date: February 1, 2016 Table of Contents BENEFIT CONSIDERATIONS COVERAGE RATIONALE DEFINITIONS...
#1 MEDICAL POLICY BONE OR SOFT TISSUE HEALING AND FUSION ENHANCEMENT PRODUCTS Policy Number: 2016T0410P Effective Date: February 1, 2016 Table of Contents BENEFIT CONSIDERATIONS COVERAGE RATIONALE DEFINITIONS...
INFUSE Bone Graft (rhbmp-2/acs)
 1 INFUSE Bone Graft (rhbmp-2/acs) For patients who need more bone to place dental implants Enjoy living with INFUSE Bone Graft. www.medtronic.com Medtronic Spinal and Biologics Business Worldwide Headquarters
1 INFUSE Bone Graft (rhbmp-2/acs) For patients who need more bone to place dental implants Enjoy living with INFUSE Bone Graft. www.medtronic.com Medtronic Spinal and Biologics Business Worldwide Headquarters
Bone Graft Substitutes
 Medical Coverage Policy Page: 1 of 21 Change Summary: Updated Description, Coverage Limitations, Background, Provider Claims Codes, Medical Terms, References Disclaimer Description Coverage Determination
Medical Coverage Policy Page: 1 of 21 Change Summary: Updated Description, Coverage Limitations, Background, Provider Claims Codes, Medical Terms, References Disclaimer Description Coverage Determination
LifeNet Health Presented by James Clagett, PhD., Chief Science Officer LifeNet Health - The Best Keep Secret in Regenerative Medicine
 Presented by James Clagett, PhD., Chief Science Officer - The Best Keep Secret in Regenerative Medicine Corporate Headquarters Virginia Beach, VA Who is? helps save lives and restore health for thousands
Presented by James Clagett, PhD., Chief Science Officer - The Best Keep Secret in Regenerative Medicine Corporate Headquarters Virginia Beach, VA Who is? helps save lives and restore health for thousands
CORNERSTONE TISSUE PARALLEL TISSUE OFFERINGS
 CORNERSTONE TISSUE PARALLEL TISSUE OFFERINGS Precision Machined Tissue Medtronic Sofamor Danek established itself as a leader in the machined allograft tissue market with the introduction of the CORNERSTONE-SR
CORNERSTONE TISSUE PARALLEL TISSUE OFFERINGS Precision Machined Tissue Medtronic Sofamor Danek established itself as a leader in the machined allograft tissue market with the introduction of the CORNERSTONE-SR
Technology Breakthrough in Spinal Implants (Technical Insights)
 Technology Breakthrough in Spinal Implants (Technical Insights) Biomaterial innovations is a growth factor for spinal implant market June 2014 Table of Contents Section Page Number Executive Summary 4
Technology Breakthrough in Spinal Implants (Technical Insights) Biomaterial innovations is a growth factor for spinal implant market June 2014 Table of Contents Section Page Number Executive Summary 4
Technique Guide. Norian SRS Fast Set Putty. Calcium phosphate bone void filler.
 Technique Guide Norian SRS Fast Set Putty. Calcium phosphate bone void filler. TableofContents Introduction Norian SRS Fast Set Putty 2 Indications and Contraindications 3 Basic Science 4 Surgical Technique
Technique Guide Norian SRS Fast Set Putty. Calcium phosphate bone void filler. TableofContents Introduction Norian SRS Fast Set Putty 2 Indications and Contraindications 3 Basic Science 4 Surgical Technique
BONE OR SOFT TISSUE HEALING AND FUSION ENHANCEMENT PRODUCTS
 CLINICAL POLICY BONE OR SOFT TISSUE HEALING AND FUSION ENHANCEMENT PRODUCTS Policy Number: SURGERY 056.10 T2 Effective Date: May 1, 2013 Table of Contents CONDITIONS OF COVERAGE... COVERAGE RATIONALE BACKGROUND...
CLINICAL POLICY BONE OR SOFT TISSUE HEALING AND FUSION ENHANCEMENT PRODUCTS Policy Number: SURGERY 056.10 T2 Effective Date: May 1, 2013 Table of Contents CONDITIONS OF COVERAGE... COVERAGE RATIONALE BACKGROUND...
Bone Morphogenetic Proteins & Spinal Surgery for Degenarative Disc Disease
 Ontario Health Technology Assessment Series 2004; Vol. 4, No. 4 Bone Morphogenetic Proteins & Spinal Surgery for Degenarative Disc Disease An Evidence Based Analysis March 2004 Medical Advisory Secretariat
Ontario Health Technology Assessment Series 2004; Vol. 4, No. 4 Bone Morphogenetic Proteins & Spinal Surgery for Degenarative Disc Disease An Evidence Based Analysis March 2004 Medical Advisory Secretariat
Bone Grafts, Bone Morphogenetic Proteins, and Bone Substitutes
 Chapter 6 Bone Grafts, Bone Morphogenetic Proteins, and Bone Substitutes Hyun Bae, MD Neil Bhamb, MD Linda E.A. Kanim, MA Justin S. Field, MD I. Bone Healing A. Primary bone healing Occurs with constructs
Chapter 6 Bone Grafts, Bone Morphogenetic Proteins, and Bone Substitutes Hyun Bae, MD Neil Bhamb, MD Linda E.A. Kanim, MA Justin S. Field, MD I. Bone Healing A. Primary bone healing Occurs with constructs
Patient Information. Lateral Lumbar Interbody Fusion Surgery (LLIF).
 Patient Information. Lateral Lumbar Interbody Fusion Surgery (LLIF). Understanding your spine Disc Between each pair of vertebrae there is a disc that acts as a cushion to protect the vertebra, allows
Patient Information. Lateral Lumbar Interbody Fusion Surgery (LLIF). Understanding your spine Disc Between each pair of vertebrae there is a disc that acts as a cushion to protect the vertebra, allows
SUPERIOR COURT OF THE STATE OF CALIFORNIA FOR THE COUNTY OF LOS ANGELES. 21 COMES NOW Plaintiff APRIL CHRISTINE CABANA who alleges as follows:
 RonaldL.M. Goldman, Esq., Bar Number Bijan Esfandiari, Esq., Bar Number 1 A. Ilyas Akbari, Esq., Bar Number 1 BAUM HEDLUND ARISTEI & GOLDMAN, P.C. 1 Wilshire Blvd., Suite 0 Los Angeles, CA 00. Tel: ()-
RonaldL.M. Goldman, Esq., Bar Number Bijan Esfandiari, Esq., Bar Number 1 A. Ilyas Akbari, Esq., Bar Number 1 BAUM HEDLUND ARISTEI & GOLDMAN, P.C. 1 Wilshire Blvd., Suite 0 Los Angeles, CA 00. Tel: ()-
WRIGHT MEDICAL GROUP INC
 WRIGHT MEDICAL GROUP INC FORM 10-K (Annual Report) Filed 02/27/14 for the Period Ending 12/31/13 Address 5677 AIRLINE ROAD ARLINGTON, TN 38002 Telephone 9018679971 CIK 0001137861 SIC Code 3842 - Orthopedic,
WRIGHT MEDICAL GROUP INC FORM 10-K (Annual Report) Filed 02/27/14 for the Period Ending 12/31/13 Address 5677 AIRLINE ROAD ARLINGTON, TN 38002 Telephone 9018679971 CIK 0001137861 SIC Code 3842 - Orthopedic,
Bone graft substitutes currently available in orthopaedic practice
 INSTRUCTIONAL REVIEW: TRAUMA Bone graft substitutes currently available in orthopaedic practice THE EVIDENCE FOR THEIR USE T. Kurien, R. G. Pearson, B. E. Scammell From Queen s Medical Centre, University
INSTRUCTIONAL REVIEW: TRAUMA Bone graft substitutes currently available in orthopaedic practice THE EVIDENCE FOR THEIR USE T. Kurien, R. G. Pearson, B. E. Scammell From Queen s Medical Centre, University
Patellofemoral Chondrosis
 Patellofemoral Chondrosis What is PF chondrosis? PF chondrosis (cartilage deterioration) is the softening or loss of smooth cartilage, most frequently that which covers the back of the kneecap, but the
Patellofemoral Chondrosis What is PF chondrosis? PF chondrosis (cartilage deterioration) is the softening or loss of smooth cartilage, most frequently that which covers the back of the kneecap, but the
Mitchell S. Fourman M.Phil Eugene Borst BS Eric Bogner, MD S. Robert Rozbruch MD Austin T. Fragomen, MD
 Post-Operative CT Measurements Accurately Predict the Need for Clinical Intervention Retrospective Findings to Support Prospective Clinical Trial Protocol Mitchell S. Fourman M.Phil Eugene Borst BS Eric
Post-Operative CT Measurements Accurately Predict the Need for Clinical Intervention Retrospective Findings to Support Prospective Clinical Trial Protocol Mitchell S. Fourman M.Phil Eugene Borst BS Eric
Patient Information. Anterior Cervical Discectomy and Fusion Surgery (ACDF).
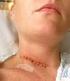 Patient Information. Anterior Cervical Discectomy and Fusion Surgery (ACDF). Understanding your spine Disc Between each pair of vertebrae there is a disc that acts as a cushion to protect the vertebra,
Patient Information. Anterior Cervical Discectomy and Fusion Surgery (ACDF). Understanding your spine Disc Between each pair of vertebrae there is a disc that acts as a cushion to protect the vertebra,
Alternatives to Autologous Bone Harvest in Spine Surgery
 The University of Pennsylvania Orthopaedic Journal 12: 77 88, 1999 1999 The University of Pennsylvania Orthopaedic Journal Alternatives to Autologous Bone Harvest in Spine Surgery EERIC TRUUMEES, M.D.
The University of Pennsylvania Orthopaedic Journal 12: 77 88, 1999 1999 The University of Pennsylvania Orthopaedic Journal Alternatives to Autologous Bone Harvest in Spine Surgery EERIC TRUUMEES, M.D.
REPUTATION MANAGEMENT: SPINAL FUSION SURGERY
 REPUTATION MANAGEMENT: SPINAL FUSION SURGERY Meaningful and Positive Progress in Spinal Fusion Results: A Journey towards Excellence Paul J. Slosar, M.D. SpineCare Medical Group As a practicing spine surgeon,
REPUTATION MANAGEMENT: SPINAL FUSION SURGERY Meaningful and Positive Progress in Spinal Fusion Results: A Journey towards Excellence Paul J. Slosar, M.D. SpineCare Medical Group As a practicing spine surgeon,
CPT Codes for Bone Marrow Transplant January 2015 James L. Gajewski, MD
 The blood and marrow transplant field has 15 dedicated CPT codes. These CPT codes can be categorized into three groups: 1. Collection Codes 2. Cell Processing Codes 3. Cell Infusion Codes Collection Codes
The blood and marrow transplant field has 15 dedicated CPT codes. These CPT codes can be categorized into three groups: 1. Collection Codes 2. Cell Processing Codes 3. Cell Infusion Codes Collection Codes
August 21, 2015. TCM Associates Ltd Mr. Iain Alligan Technical Director 3 Hillgrove Business Park Nazeing Road Essex EN9 2HB United Kingdom
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center - WO66-G609 Silver Spring, MD 20993-0002 Mr. Iain Alligan Technical
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center - WO66-G609 Silver Spring, MD 20993-0002 Mr. Iain Alligan Technical
HyStem. Hydrogels CELLULAR MATRICES FOR TRANSLATIONAL RESEARCH. esibio.com
 HyStem Hydrogels CELLULAR MATRICES FOR TRANSLATIONAL RESEARCH HyStem Hydrogel Extracellular Matrices Chemically-defined For use in 2D and 3D formats in vitro and in vivo applications Customizable for many
HyStem Hydrogels CELLULAR MATRICES FOR TRANSLATIONAL RESEARCH HyStem Hydrogel Extracellular Matrices Chemically-defined For use in 2D and 3D formats in vitro and in vivo applications Customizable for many
Don t Let Life Pass You By Because Of Oral Bone Loss
 Don t Let Life Pass You By Because Of Oral Bone Loss Ask For Dental Implant Solutions From BIOMET 3i Scan With Your Smartphone! In order to scan QR codes, your mobile device must have a QR code reader
Don t Let Life Pass You By Because Of Oral Bone Loss Ask For Dental Implant Solutions From BIOMET 3i Scan With Your Smartphone! In order to scan QR codes, your mobile device must have a QR code reader
coligne treatment technology
 coligne treatment technology coligne a strategy in spine Leadership begins before the patient enters the surgical theater, among the spine care professionals forming the coalliance. This may be a small
coligne treatment technology coligne a strategy in spine Leadership begins before the patient enters the surgical theater, among the spine care professionals forming the coalliance. This may be a small
Healthcare Industry: US Orthopedic Implants and Devices Market
 Healthcare Industry: US Orthopedic Implants and Devices Market by KenResearch - Tuesday, March 11, 2014 http://www.kenresearch.com/blog/2014/03/us-orthopedic-implants-and-devices-market/ The Replacement
Healthcare Industry: US Orthopedic Implants and Devices Market by KenResearch - Tuesday, March 11, 2014 http://www.kenresearch.com/blog/2014/03/us-orthopedic-implants-and-devices-market/ The Replacement
chronos. Bone Graft Substitute. Osteoconductive, resorbable, synthetic.
 chronos. Bone Graft Substitute. Osteoconductive, resorbable, synthetic. Optimized osteoconductive matrix Enhancement with biological factors Fast remodeling within 6 to 18 months The answer to bone voids
chronos. Bone Graft Substitute. Osteoconductive, resorbable, synthetic. Optimized osteoconductive matrix Enhancement with biological factors Fast remodeling within 6 to 18 months The answer to bone voids
INFUSE Bone Graft Oral Maxillofacial Bone Grafting Procedures
 INFUSE Bone Graft Oral Maxillofacial Bone Grafting Procedures Dental Products Advisory Panel Committee Ed Chin, DPh Group Director, Regulatory Affairs Medtronic Spinal and Biologics 1 INFUSE Bone Graft
INFUSE Bone Graft Oral Maxillofacial Bone Grafting Procedures Dental Products Advisory Panel Committee Ed Chin, DPh Group Director, Regulatory Affairs Medtronic Spinal and Biologics 1 INFUSE Bone Graft
University of Colorado Hospital Policy and Procedure Transplant and Implant Tissue Storage and Issuance
 University of Colorado Hospital Policy and Procedure Transplant and Implant Tissue Storage and Issuance Related Policies and Procedures: Temperature Controlled Storage Unit Monitoring Hospital Infection
University of Colorado Hospital Policy and Procedure Transplant and Implant Tissue Storage and Issuance Related Policies and Procedures: Temperature Controlled Storage Unit Monitoring Hospital Infection
SPINAL FUSION. North American Spine Society Public Education Series
 SPINAL FUSION North American Spine Society Public Education Series WHAT IS SPINAL FUSION? The spine is made up of a series of bones called vertebrae ; between each vertebra are strong connective tissues
SPINAL FUSION North American Spine Society Public Education Series WHAT IS SPINAL FUSION? The spine is made up of a series of bones called vertebrae ; between each vertebra are strong connective tissues
March 6-7, 2015. (Friday & Saturday) COURSE DIRECTORS. Course will be held at the BIOMET Skills Academy Miami, Florida
 NEW THREE DIMENSIONAL RIDGE AUGMENTATION & ADVANCED DENTAL IMPLANT COURSE Robert E. Marx, DDS Professor of Surgery and Chief March 6-7, 2015 (Friday & Saturday) COURSE DIRECTORS Course will be held at
NEW THREE DIMENSIONAL RIDGE AUGMENTATION & ADVANCED DENTAL IMPLANT COURSE Robert E. Marx, DDS Professor of Surgery and Chief March 6-7, 2015 (Friday & Saturday) COURSE DIRECTORS Course will be held at
Advances In Spine Care. James D. Bruffey M.D. Scripps Clinic Division of Orthopaedic Surgery Section of Spinal Surgery
 Advances In Spine Care James D. Bruffey M.D. Scripps Clinic Division of Orthopaedic Surgery Section of Spinal Surgery Introduction The Spine - A common source of problems Back pain is the #2 presenting
Advances In Spine Care James D. Bruffey M.D. Scripps Clinic Division of Orthopaedic Surgery Section of Spinal Surgery Introduction The Spine - A common source of problems Back pain is the #2 presenting
Spinal Arthrodesis Group Exercises
 Spinal Arthrodesis Group Exercises 1. Two surgeons work together to perform an arthrodesis. Dr. Bonet, a general surgeon, makes the anterior incision to gain access to the spine for the arthrodesis procedure.
Spinal Arthrodesis Group Exercises 1. Two surgeons work together to perform an arthrodesis. Dr. Bonet, a general surgeon, makes the anterior incision to gain access to the spine for the arthrodesis procedure.
Innovation Across Borders Forum VBO-FEB Innovation Case Preparation Form
 Innovation Across Borders Forum VBO-FEB Innovation Case Preparation Form WHO Welke onderneming(en) werd(en) hierbij betrokken? (grootte, bedrijfssector, )? Met welke partner(s) (clusters, O&O-centrum,
Innovation Across Borders Forum VBO-FEB Innovation Case Preparation Form WHO Welke onderneming(en) werd(en) hierbij betrokken? (grootte, bedrijfssector, )? Met welke partner(s) (clusters, O&O-centrum,
ANTERIOR LUMBAR INTERBODY FUSION (ALIF) Basic Anatomical Landmarks: Anterior Lumbar Spine
 (ALIF) Anterior In human anatomy, referring to the front surface of the body or the position of one structure relative to another Lumbar Relating to the loins or the section of the back and sides between
(ALIF) Anterior In human anatomy, referring to the front surface of the body or the position of one structure relative to another Lumbar Relating to the loins or the section of the back and sides between
The Smart Dentin Grinder. Business Opportunity 2014
 The Smart Dentin Grinder Business Opportunity 2014 2 The Market Grafting Materials for Dental Reconstruction Currently a $550M global market An upcoming market, with 5.7% annual growth Tissue and Bone
The Smart Dentin Grinder Business Opportunity 2014 2 The Market Grafting Materials for Dental Reconstruction Currently a $550M global market An upcoming market, with 5.7% annual growth Tissue and Bone
Fracture Care Coding September 28, 2011
 Fracture Care Coding September 28, 2011 Julie Edens Leu, CPC, CPCO, CPMA, CPC-I 1 Disclaimer Every reasonable effort has been made to ensure that the educational material provided today is accurate and
Fracture Care Coding September 28, 2011 Julie Edens Leu, CPC, CPCO, CPMA, CPC-I 1 Disclaimer Every reasonable effort has been made to ensure that the educational material provided today is accurate and
The MOVIE. Currently VP, Regulatory Affairs, MCRA Former Orthopedic Devices Branch Chief (2006)
 FDA played by Glenn Stiegman Currently VP, Regulatory Affairs, MCRA Former Orthopedic Devices Branch Chief (2006) Managed Orthopedic and Spine groups (Branch split in 2007) Former Orthopedic Reviewer (2000-2005)
FDA played by Glenn Stiegman Currently VP, Regulatory Affairs, MCRA Former Orthopedic Devices Branch Chief (2006) Managed Orthopedic and Spine groups (Branch split in 2007) Former Orthopedic Reviewer (2000-2005)
UNITED STATES SECURITIES AND EXCHANGE COMMISSION. Washington, DC 20549 FORM 10-K
 È UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, DC 20549 FORM 10-K ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(D) OF THE SECURITIES EXCHANGE ACT OF 1934 For the fiscal year ended December
È UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, DC 20549 FORM 10-K ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(D) OF THE SECURITIES EXCHANGE ACT OF 1934 For the fiscal year ended December
Low Back Pain (LBP) Prevalence. Low Back Pain (LBP) Prevalence. Lumbar Fusion: Where is the Evidence?
 15 th Annual Cleveland Clinic Pain Management Symposium Sarasota, Florida Lumbar Fusion: Where is the Evidence? Gordon R. Bell, M.D. Director, Cleveland Clinic Low Back Pain (LBP) Prevalence Lifetime prevalence:
15 th Annual Cleveland Clinic Pain Management Symposium Sarasota, Florida Lumbar Fusion: Where is the Evidence? Gordon R. Bell, M.D. Director, Cleveland Clinic Low Back Pain (LBP) Prevalence Lifetime prevalence:
vitality meets bone volume around dental implants Straumann BoneCeramic
 vitality meets bone volume around dental implants Straumann BoneCeramic The vitality you need around your implants Bone volume restoration and preservation for the desired esthetic outcome Image by PD
vitality meets bone volume around dental implants Straumann BoneCeramic The vitality you need around your implants Bone volume restoration and preservation for the desired esthetic outcome Image by PD
Demineralized Bone Matrix: Development of a Process for Optimal Performance. the better approach
 Demineralized Bone Matrix: Development of a Process for Optimal Performance the better approach the better approach Introduction MTF is a non-profit organization founded in 1987 by academic orthopaedic
Demineralized Bone Matrix: Development of a Process for Optimal Performance the better approach the better approach Introduction MTF is a non-profit organization founded in 1987 by academic orthopaedic
PRESENTATIONS/PAPERS INTERNATIONAL. RAISING STANDARDS IN JOINT ARTHROPLASTY Course Co-Chairman, The Great Debate - London, June 2013
 PRESENTATIONS/PAPERS INTERNATIONAL 1998 2013 RAISING STANDARDS IN JOINT ARTHROPLASTY Course Co-Chairman, The Great Debate - London, June 2013 HOW TO CHOOSE THE RIGHT STEM FOR THE RIGHT PATIENT Corin Symposium
PRESENTATIONS/PAPERS INTERNATIONAL 1998 2013 RAISING STANDARDS IN JOINT ARTHROPLASTY Course Co-Chairman, The Great Debate - London, June 2013 HOW TO CHOOSE THE RIGHT STEM FOR THE RIGHT PATIENT Corin Symposium
.org. Metastatic Bone Disease. Description
 Metastatic Bone Disease Page ( 1 ) Cancer that begins in an organ, such as the lungs, breast, or prostate, and then spreads to bone is called metastatic bone disease (MBD). More than 1.2 million new cancer
Metastatic Bone Disease Page ( 1 ) Cancer that begins in an organ, such as the lungs, breast, or prostate, and then spreads to bone is called metastatic bone disease (MBD). More than 1.2 million new cancer
Clinical Commissioning Policy Proposition: Bone morphogenetic protein-2 in spinal fusion
 DRAFT FOR PUBLIC CONSULTATION Clinical Commissioning Policy Proposition: Bone morphogenetic protein-2 in spinal fusion Reference: NHS England D14X01/01 DRAFT FOR PUBLIC CONSULTATION Information Reader
DRAFT FOR PUBLIC CONSULTATION Clinical Commissioning Policy Proposition: Bone morphogenetic protein-2 in spinal fusion Reference: NHS England D14X01/01 DRAFT FOR PUBLIC CONSULTATION Information Reader
Bone graft materials in fixation of orthopaedic implants
 PHD THESIS DANISH MEDICAL JOURNAL Bone graft materials in fixation of orthopaedic implants in sheep Hassan Babiker This review has been accepted as a thesis together with 3 previously published papers
PHD THESIS DANISH MEDICAL JOURNAL Bone graft materials in fixation of orthopaedic implants in sheep Hassan Babiker This review has been accepted as a thesis together with 3 previously published papers
WRIGHT MEDICAL GROUP, INC. (Exact name of registrant as specified in its charter)
 UNITED STATES SECURITIES AND EXCHANGE COMMISSION WASHINGTON, DC 20549 FORM 10-K (Mark One) þ ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 For the fiscal year ended
UNITED STATES SECURITIES AND EXCHANGE COMMISSION WASHINGTON, DC 20549 FORM 10-K (Mark One) þ ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 For the fiscal year ended
ICD-10-PCS Documentation and Coding for Spinal Procedures October 22, 2015
 Questions and Answers 1. We have a question regarding a spinal surgical procedure. The diagnosis was bilateral lateral recess stenosis and central stenosis from L2-L5. The procedure was an open lumbar
Questions and Answers 1. We have a question regarding a spinal surgical procedure. The diagnosis was bilateral lateral recess stenosis and central stenosis from L2-L5. The procedure was an open lumbar
Do you have anything to add? If so, I d love to hear from you! Jessica Robinson Conference Manager Life Sciences @jessbiopharma
 1 Who is the most influential figure in cord blood around the world? What is the biggest challenge to overcome in the use of cord blood as a source of stem cells? We asked 10 leading experts in the cord
1 Who is the most influential figure in cord blood around the world? What is the biggest challenge to overcome in the use of cord blood as a source of stem cells? We asked 10 leading experts in the cord
This is a licensed product of Ken Research and should not be copied
 TABLE OF CONTENTS 1. The US Orthopedic Implants and Devices Market Introduction 2. The US Orthopedic Implants and Devices Market Value Chain 3. The US Orthopedic Implants and Devices Market Size, 2008-2013
TABLE OF CONTENTS 1. The US Orthopedic Implants and Devices Market Introduction 2. The US Orthopedic Implants and Devices Market Value Chain 3. The US Orthopedic Implants and Devices Market Size, 2008-2013
Spine Clinic Neurospine Specialists, Orthopaedics and Neurosurgery
 Spine Clinic Neurospine Specialists, Orthopaedics and Neurosurgery REVISION SPINE SURGERY Revision surgery is a very complex field which requires experience, training and evaluation in a very individual
Spine Clinic Neurospine Specialists, Orthopaedics and Neurosurgery REVISION SPINE SURGERY Revision surgery is a very complex field which requires experience, training and evaluation in a very individual
San Diego Stem Cell Treatment Center Frequently Asked Questions
 San Diego Stem Cell Treatment Center Frequently Asked Questions What is a Stem Cell? A stem cell is basically any cell that can replicate and differentiate. This means the cell can not only multiply, but
San Diego Stem Cell Treatment Center Frequently Asked Questions What is a Stem Cell? A stem cell is basically any cell that can replicate and differentiate. This means the cell can not only multiply, but
Anterior Lumbar Interbody Fusion (ALIF). Instrument set supports placement of ALIF spacers using anterior or anterolateral approach.
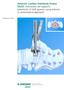 Anterior Lumbar Interbody Fusion (ALIF). Instrument set supports placement of ALIF spacers using anterior or anterolateral approach. Technique Guide Instruments and implants approved by the AO Foundation
Anterior Lumbar Interbody Fusion (ALIF). Instrument set supports placement of ALIF spacers using anterior or anterolateral approach. Technique Guide Instruments and implants approved by the AO Foundation
Stem Cell Background Paper
 Stem Cell Background Paper Introduction...2 Stem Cell Basics...3 Stem Cell Process Flow...9 Comparison of Blood, Stem Cells, Tissues and Organs Processes...10 Responsibilities for the Blood, Stem Cells,
Stem Cell Background Paper Introduction...2 Stem Cell Basics...3 Stem Cell Process Flow...9 Comparison of Blood, Stem Cells, Tissues and Organs Processes...10 Responsibilities for the Blood, Stem Cells,
STEM CELL TRANSPLANTS
 UAMS Information on STEM CELL TRANSPLANTS What is a Stem Cell Transplant? A stem cell transplant is an infusion of stem cells following high-dose chemotherapy. The infused cells effectively rescue the
UAMS Information on STEM CELL TRANSPLANTS What is a Stem Cell Transplant? A stem cell transplant is an infusion of stem cells following high-dose chemotherapy. The infused cells effectively rescue the
THE CARTILAGE IMPLANT FOR BIOLOGICAL CARTILAGE REPAIR
 THE CARTILAGE IMPLANT FOR BIOLOGICAL CARTILAGE REPAIR 02/13 Content chondrotissue (p.4) Scientific Background (p.5) Preclinical Outcomes (p.6) Clinical Outcomes: MRI Evaluation (p.7) Clinical Outcomes:
THE CARTILAGE IMPLANT FOR BIOLOGICAL CARTILAGE REPAIR 02/13 Content chondrotissue (p.4) Scientific Background (p.5) Preclinical Outcomes (p.6) Clinical Outcomes: MRI Evaluation (p.7) Clinical Outcomes:
Osseous Tissue & Structure. The skeletal system includes: Storage of minerals: calcium salts
 Chapter 15 Lecture The Skeletal System: Osseous Tissue & Skeletal Structure The Skeletal System The skeletal system includes: Bones, cartilages, ligaments Bone tissue = osseous tissue Includes living cells
Chapter 15 Lecture The Skeletal System: Osseous Tissue & Skeletal Structure The Skeletal System The skeletal system includes: Bones, cartilages, ligaments Bone tissue = osseous tissue Includes living cells
Zimmer Periarticular Elbow Locking Plate System
 Zimmer Periarticular Elbow Locking Plate System Surgical Technique The Right Solutions for Fractures Around the Elbow Disclaimer This document is intended exclusively for physicians and is not intended
Zimmer Periarticular Elbow Locking Plate System Surgical Technique The Right Solutions for Fractures Around the Elbow Disclaimer This document is intended exclusively for physicians and is not intended
Wrist and Hand. Patient Information Guide to Bone Fracture, Bone Reconstruction and Bone Fusion: Fractures of the Wrist and Hand: Carpal bones
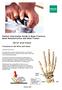 Patient Information Guide to Bone Fracture, Bone Reconstruction and Bone Fusion: Wrist and Hand Fractures of the Wrist and Hand: Fractures of the wrist The wrist joint is made up of the two bones in your
Patient Information Guide to Bone Fracture, Bone Reconstruction and Bone Fusion: Wrist and Hand Fractures of the Wrist and Hand: Fractures of the wrist The wrist joint is made up of the two bones in your
ATLANTIS Anterior Cervical Plate System ATLANTIS Translational Plate. 510(k) Summary. February 2007
 rk,1 10{2 ATLANTIS Anterior Cervical Plate System ATLANTIS Translational Plate 510(k) Summary FEB 2 3 2[007 February 2007 I. Company: Medtronic Sofamor Danek USA 1800 Pyramid Place Memphis, Tennessee 38132
rk,1 10{2 ATLANTIS Anterior Cervical Plate System ATLANTIS Translational Plate 510(k) Summary FEB 2 3 2[007 February 2007 I. Company: Medtronic Sofamor Danek USA 1800 Pyramid Place Memphis, Tennessee 38132
PART B - Human Tissue List February 2015
 PART B - Human Tissue List February 2015 Billing Code CARDIO-THORACIC Conduit - Aortic TBV56 Donor Tissue Bank of Victoria $1,505.00 Conduit - Aortic + Valve QHV09 The Prince Charles Hospital - Queensland
PART B - Human Tissue List February 2015 Billing Code CARDIO-THORACIC Conduit - Aortic TBV56 Donor Tissue Bank of Victoria $1,505.00 Conduit - Aortic + Valve QHV09 The Prince Charles Hospital - Queensland
The therapies are focused in cardiovascular, orthopedics and neurological indications.
 1 2 CescaTherapeutics is (to our knowledge) the first company to integrate all of the essential: 1. Cell friendly devices, 2. Proprietary cell formulations, and Strict clinical steps to ensure the autologous
1 2 CescaTherapeutics is (to our knowledge) the first company to integrate all of the essential: 1. Cell friendly devices, 2. Proprietary cell formulations, and Strict clinical steps to ensure the autologous
. Soft Tissue Regeneration... Consultant. RTI Surgical...Consultant. OSSUR...Consultant. Arthrex...Consultant
 sl23lls 1 E ACL Graft Choice in the Elite Athlete and When Do I Let Them Return to Sport: Graft Ghoice: Allografts : @ Peter A. lndelicato MD Emeritus Professor Sports Medicine Department of OrthoPaedic
sl23lls 1 E ACL Graft Choice in the Elite Athlete and When Do I Let Them Return to Sport: Graft Ghoice: Allografts : @ Peter A. lndelicato MD Emeritus Professor Sports Medicine Department of OrthoPaedic
US Regulations for Import and Export of Cell Therapy Products
 US Regulations for Import and Export of Cell Therapy Products Kurt Gunter, MD U Minnesota UCB Transplants (2001-2007)* Total units: 790 Number US banks: 19 Number non-us banks: 12 % units from non-us banks:
US Regulations for Import and Export of Cell Therapy Products Kurt Gunter, MD U Minnesota UCB Transplants (2001-2007)* Total units: 790 Number US banks: 19 Number non-us banks: 12 % units from non-us banks:
Implant materials. Learning outcomes. Implant materials in trauma. How to use this handout? Functions of implants. Types of materials
 Implant materials How to use this handout? The left column is the information as given during the lecture. The column at the right gives you space to make personal notes. Learning outcomes At the end of
Implant materials How to use this handout? The left column is the information as given during the lecture. The column at the right gives you space to make personal notes. Learning outcomes At the end of
.org. Arthritis of the Hand. Description
 Arthritis of the Hand Page ( 1 ) The hand and wrist have multiple small joints that work together to produce motion, including the fine motion needed to thread a needle or tie a shoelace. When the joints
Arthritis of the Hand Page ( 1 ) The hand and wrist have multiple small joints that work together to produce motion, including the fine motion needed to thread a needle or tie a shoelace. When the joints
Curriculum Vitae (Updated 06/09)
 Curriculum Vitae (Updated 06/09) JOHN M. SMALL, M.D. Business Address Florida Orthopaedic Institute 13020 N. Telecom Parkway Temple Terrace, FL 33637 Business Phone 813-978-9700 : March 27, 1958 Medical
Curriculum Vitae (Updated 06/09) JOHN M. SMALL, M.D. Business Address Florida Orthopaedic Institute 13020 N. Telecom Parkway Temple Terrace, FL 33637 Business Phone 813-978-9700 : March 27, 1958 Medical
Update on Bone Morphogenetic Proteins and Their Application in Spine Surgery
 SPECIALTY ARTICLE: ORTHOPAEDIC SURGERY Update on Bone Morphogenetic Proteins and Their Application in Spine Surgery Dino Samartzis, BS, Dip EBHC, Nitin Khanna, MD, Francis H Shen, MD, Howard S An, MD No
SPECIALTY ARTICLE: ORTHOPAEDIC SURGERY Update on Bone Morphogenetic Proteins and Their Application in Spine Surgery Dino Samartzis, BS, Dip EBHC, Nitin Khanna, MD, Francis H Shen, MD, Howard S An, MD No
Degenerative Spine Solutions
 Degenerative Spine Solutions The Backbone for Your Surgical Needs Aesculap Spine Backbone for Your Degenerative Spine Needs Comprehensive operative solutions, unique product technology and world-class
Degenerative Spine Solutions The Backbone for Your Surgical Needs Aesculap Spine Backbone for Your Degenerative Spine Needs Comprehensive operative solutions, unique product technology and world-class
Orthopedic Applications of Stem Cell Therapy
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
ICUC One-Page Paper. Mixing a lag screw with a splinting protection plate Alberto Fernández Dell Oca, Pietro Regazzoni, Stephan Perren May 2016
 Mixing a lag screw with a splinting protection plate Alberto Fernández Dell Oca, Pietro Regazzoni, Stephan Perren May 2016 The 20th century has seen three essential improvements of surgical fracture fixation:
Mixing a lag screw with a splinting protection plate Alberto Fernández Dell Oca, Pietro Regazzoni, Stephan Perren May 2016 The 20th century has seen three essential improvements of surgical fracture fixation:
Anatomy and Physiology 101 for Attorneys
 Knee Injuries Anatomy and Physiology 101 for Attorneys Phil Davidson, MD Heiden-Davidson Orthopedics Salt Lake City, UT May 2011 Introduction Dr. Phil Davidson Park City and SLC clinics Education: Harvard,
Knee Injuries Anatomy and Physiology 101 for Attorneys Phil Davidson, MD Heiden-Davidson Orthopedics Salt Lake City, UT May 2011 Introduction Dr. Phil Davidson Park City and SLC clinics Education: Harvard,
CHS 06-07 BONES AND SKELETAL TISSUES
 CHS 06-07 BONES AND SKELETAL TISSUES This chapter provides a review of bone and skeletal tissue. The human skeleton is composed primarily of two connective tissues: (1) cartilage and (2) bone. CHARACTERISTICS
CHS 06-07 BONES AND SKELETAL TISSUES This chapter provides a review of bone and skeletal tissue. The human skeleton is composed primarily of two connective tissues: (1) cartilage and (2) bone. CHARACTERISTICS
PERIPROSTHETIC IMPLANTS
 PERIPROSTHETIC IMPLANTS PRODUCT OVERVIEW CLINICAL SOLUTIONS Periprosthetic fractures present unique challenges, such as how to gain fixation when the medullary canal is occupied. Special techniques and
PERIPROSTHETIC IMPLANTS PRODUCT OVERVIEW CLINICAL SOLUTIONS Periprosthetic fractures present unique challenges, such as how to gain fixation when the medullary canal is occupied. Special techniques and
Aesculap Veterinary Orthopaedics. Targon VET Interlocking Nail
 Aesculap Veterinary Orthopaedics Targon VET Interlocking Nail Flexibility Stability Dynamics The Targon VET is based on new, technology which reverses the known principles of interlocking nails. The rotation-stabilising
Aesculap Veterinary Orthopaedics Targon VET Interlocking Nail Flexibility Stability Dynamics The Targon VET is based on new, technology which reverses the known principles of interlocking nails. The rotation-stabilising
Norbert Weber, Lilly Ostrovsky. Musculoskeletal Transplant Foundation (MTF), Edison, NJ, USA. Disclosures: N. Weber: None. L. Ostrovsky: None.
 Immunogenicity of Biopharmaceuticals in Large Animal Models: Timedependent Formation of Antibodies and Detection of TNF-alpha in Serum and Synovial Fluid Norbert Weber, Lilly Ostrovsky. Musculoskeletal
Immunogenicity of Biopharmaceuticals in Large Animal Models: Timedependent Formation of Antibodies and Detection of TNF-alpha in Serum and Synovial Fluid Norbert Weber, Lilly Ostrovsky. Musculoskeletal
Corporate Medical Policy Electrical Bone Growth Stimulation
 Corporate Medical Policy Electrical Bone Growth Stimulation File Name: Origination: Last CAP Review: Next CAP Review: Last Review: electrical_bone_growth_stimulation 4/1981 6/2016 6/2017 6/2016 Description
Corporate Medical Policy Electrical Bone Growth Stimulation File Name: Origination: Last CAP Review: Next CAP Review: Last Review: electrical_bone_growth_stimulation 4/1981 6/2016 6/2017 6/2016 Description
now virtually as commonplace as getting a getting a flu shot. As the industry continues to grow
 Over the past decade, the number of stem cell therapy providers has dramatically increased. From what was once a rare and medically advanced procedure, stem cell therapy is now virtually as commonplace
Over the past decade, the number of stem cell therapy providers has dramatically increased. From what was once a rare and medically advanced procedure, stem cell therapy is now virtually as commonplace
The goals of modern spinal surgery are to maximize
 SPRING 2013 Robot-Guided Spine Surgery Christopher R. Good, M.D., F.A.C.S. and Blair K. Snyder, P.A.-C. The goals of modern spinal surgery are to maximize patient function and accelerate a return to a
SPRING 2013 Robot-Guided Spine Surgery Christopher R. Good, M.D., F.A.C.S. and Blair K. Snyder, P.A.-C. The goals of modern spinal surgery are to maximize patient function and accelerate a return to a
LUMBAR SPINAL STENOSIS OBSERVATIONS, EVIDENCE, AND TRENDS FULILLING THE UNMET CLINICAL NEED WRITTEN BY: HALLETT MATHEWS, MD, MBA
 LUMBAR SPINAL STENOSIS OBSERVATIONS, EVIDENCE, AND TRENDS FULILLING THE UNMET CLINICAL NEED WRITTEN BY: HALLETT MATHEWS, MD, MBA Overview of Lumbar Spinal Stenosis Spine stabilization, which has equated
LUMBAR SPINAL STENOSIS OBSERVATIONS, EVIDENCE, AND TRENDS FULILLING THE UNMET CLINICAL NEED WRITTEN BY: HALLETT MATHEWS, MD, MBA Overview of Lumbar Spinal Stenosis Spine stabilization, which has equated
14.0 Stem Cell Laboratory Services
 Laboratory Services Contact Information: To inquire about assisting with surgical harvesting of bone marrow, cellular therapy (CT) product processing, cryopreservation, storage, or any other lab services,
Laboratory Services Contact Information: To inquire about assisting with surgical harvesting of bone marrow, cellular therapy (CT) product processing, cryopreservation, storage, or any other lab services,
Rodding Surgery. 804 W. Diamond Ave., Ste. 210 Gaithersburg, MD 20878 (800) 981-2663 (301) 947-0083
 Rodding Surgery 804 W. Diamond Ave., Ste. 210 Gaithersburg, MD 20878 (800) 981-2663 (301) 947-0083 Fax: (301) 947-0456 Internet: www.oif.org Email: bonelink@oif.org The Osteogenesis Imperfecta Foundation,
Rodding Surgery 804 W. Diamond Ave., Ste. 210 Gaithersburg, MD 20878 (800) 981-2663 (301) 947-0083 Fax: (301) 947-0456 Internet: www.oif.org Email: bonelink@oif.org The Osteogenesis Imperfecta Foundation,
High-Flex Solutions for the MIS Era. Zimmer Unicompartmental High Flex Knee System
 High-Flex Solutions for the MIS Era Zimmer Unicompartmental High Flex Knee System Zimmer Unicompartmental High Flex Knee Built On Success In today s health care environment, meeting patient demands means
High-Flex Solutions for the MIS Era Zimmer Unicompartmental High Flex Knee System Zimmer Unicompartmental High Flex Knee Built On Success In today s health care environment, meeting patient demands means
Minimally Invasive Lumbar Fusion
 Minimally Invasive Lumbar Fusion Biomechanical Evaluation (1) coflex-f screw Biomechanical Evaluation (1) coflex-f intact Primary Stability intact Primary Stability Extension Neutral Position Flexion Coflex
Minimally Invasive Lumbar Fusion Biomechanical Evaluation (1) coflex-f screw Biomechanical Evaluation (1) coflex-f intact Primary Stability intact Primary Stability Extension Neutral Position Flexion Coflex
ORTHOPAEDIC RESEARCH AND INNOVATION DAY, April 6, 2015
 ORTHOPAEDIC RESEARCH AND INNOVATION DAY, April 6, 2015 SAFETY STUDY OF INTRAVENOUSLY ADMINISTERED HUMAN CORD BLOOD STEM CELLS IN THE TREATMENT OF SYMPTOMS RELATED TO CHRONIC INFLAMATION Brian Mehling,
ORTHOPAEDIC RESEARCH AND INNOVATION DAY, April 6, 2015 SAFETY STUDY OF INTRAVENOUSLY ADMINISTERED HUMAN CORD BLOOD STEM CELLS IN THE TREATMENT OF SYMPTOMS RELATED TO CHRONIC INFLAMATION Brian Mehling,
CURRICULUM VITAE Jefferson Christopher Eyke, M.D.
 CURRICULUM VITAE Jefferson Christopher Eyke, M.D. OFFICE: Orthopaedic Associates of Muskegon 1400 Mercy Drive, Suite 100 Muskegon, MI 49444 Telephone: 231.733.1326 Fax: 231.733.5212 Joined Orthopaedic
CURRICULUM VITAE Jefferson Christopher Eyke, M.D. OFFICE: Orthopaedic Associates of Muskegon 1400 Mercy Drive, Suite 100 Muskegon, MI 49444 Telephone: 231.733.1326 Fax: 231.733.5212 Joined Orthopaedic
How To Make An Implant From A Metal That Is Like Bone
 Trabecular Metal Technology The Best Thing Next to Bone Trabecular Metal Technology The Best Thing Next to Bone When you need a hip, knee, shoulder or spine implant, you want assurance that your implant
Trabecular Metal Technology The Best Thing Next to Bone Trabecular Metal Technology The Best Thing Next to Bone When you need a hip, knee, shoulder or spine implant, you want assurance that your implant
One of the most important recent developments in cancer treatment has been the ability to harvest stem
 BIOTECHNOLOGY A Patient-Friendly Approach to Human Cell Transplantation One of the most important recent developments in cancer treatment has been the ability to harvest stem cells from bone marrow or
BIOTECHNOLOGY A Patient-Friendly Approach to Human Cell Transplantation One of the most important recent developments in cancer treatment has been the ability to harvest stem cells from bone marrow or
Direct Lateral Interbody Fusion A Minimally Invasive Approach to Spinal Stabilization
 APPROVED IRN10389-1.1-04 Direct Lateral Interbody Fusion A Minimally Invasive Approach to Spinal Stabilization Because it involves accessing the spine through the patient s side, the Direct Lateral approach
APPROVED IRN10389-1.1-04 Direct Lateral Interbody Fusion A Minimally Invasive Approach to Spinal Stabilization Because it involves accessing the spine through the patient s side, the Direct Lateral approach
TERUMO Corporation Business Strategy Conference
 TERUMO Corporation Business Strategy Conference THERAPEUTIC SYSTEMS & CELL PROCESSING MARK FLOWER GLOBAL MARKETING December 5, 2011 Regenerative Medicine, Cell Therapy, & Cell Processing Regenerative Medicine
TERUMO Corporation Business Strategy Conference THERAPEUTIC SYSTEMS & CELL PROCESSING MARK FLOWER GLOBAL MARKETING December 5, 2011 Regenerative Medicine, Cell Therapy, & Cell Processing Regenerative Medicine
