The MOVIE. Currently VP, Regulatory Affairs, MCRA Former Orthopedic Devices Branch Chief (2006)
|
|
|
- Mitchell Brown
- 8 years ago
- Views:
Transcription
1
2 FDA played by Glenn Stiegman Currently VP, Regulatory Affairs, MCRA Former Orthopedic Devices Branch Chief (2006) Managed Orthopedic and Spine groups (Branch split in 2007) Former Orthopedic Reviewer ( ) Spine Team Leader Author of various Spine and Orthopedic Guidance Documents Various Orthopedic ASTM Standards committees BS, MS in Biomedical Engineering For the Part: The MOVIE Playing 25 year old Biomedical Engineer Courses included Biomechanics, Biomaterials
3 What does the FDA do? Mission: The FDA's mission is to promote and protect the public health by helping safe and effective products reach the market in a timely way, and monitoring products for continued safety after they are in use. Our work is a blending of law and science aimed at protecting consumers. In Not other FDA s words, Mission: FDA The FDA's does mission not consider is to promote cost and protect the public health by helping safe and effective within products their request reach the of market generating in a cost-effective valid way, scientific and monitoring evidence products for proof for continued of safety safety and after they are in use. Our effectiveness. work is a blending of law and science aimed at protecting consumers.
4 Moving on Up Attempting to allow the safest, effective, and innovative devices onto the market. CDRH Medical Device Innovation Initiative New initiatives to better the total product life cycle IOM Evaluation of 510(k) Process 2010 Strategic Goals Met FY 2010 performance target successfully completed 86 percent, 92 of the 107 actions due in FY As of November 30, 2010 successfully completed 91percent, 99 of 109 actions due by the end of November. Priority 1. Fully Implement a Total Product Life Cycle Approach Strategy 1.1. Enhance and Integrate Premarket, Postmarket, and Compliance Information and Functions Priority 2. Enhance Communication and Transparency Priority 3. Strengthen Our Workforce and Workplace Priority 4. Proactively Facilitate Innovation and Address Unmet Public Health Needs FDA Moving on Up to the Government Trickle Down Housing Effect on to the Reviewer: Beltway Minimal
5 The Road Map Center for Devices and Radiological Health (CDRH) Center for Veterinary Medicine (CVM) Center for Drug Evaluation and Research (CDER) Center for Biologics Evaluation and Research (CBER) Center for Food Safety and Applied Nutrition (CFSAN)
6 Setting the Foundation: Spine Branch Branch Chief Ronald Jean, Ph.D. 15 Reviewers and Scientists 3 Medical Officers Review: Pedicle Screw Systems Anterior Cervical Plates Interbody Cages Total Disc Replacements Interspinous devices Posterior Cervical Devices (OCT Systems) Nucleus Replacements Facet Replacements Do Not Review: Biologics Vertebroplasty/Kyphoplasty Cements Vertebral Body Compression Devices Surgical Meshes
7
8 CE Marking versus FDA Process FDA PROCESS Classify devices by complexity and risk (I, II, III) Mandate the implementation of a Quality Management System (QMS) (21 CFR 820) 510(k) paradigm relies on demonstration of substantial equivalence to an existing, FDAcleared medical device. Class II Devices (510(k)): Pedicle Screw Systems Cervical Plates Posterior Cervical Systems Interbody Cages Interspinous Devices (Fusion) Class III (PMA): Total Disc Replacements Interspinous Devices (Non-fusion) Nucleus Replacements Facet Replacements CE MARKING Classify devices by complexity and risk (I, IIa, IIb, III) Mandate the implementation of a Quality Management System (QMS) with the exemption of devices listed under the MDD Class I non-measuring/non-sterile subdivision (MDD outlines compliance) Technical files are prepared in accordance to the MDD for review by a Notified Body. Upon review, a legally-binding Declaration of Conformity is prepared by the manufacturer indicating that the product is in compliance with the MDD. CE Mark confirms the safety of a product CE Mark does not confirm effectiveness of a product CE Mark is not equivalent to FDA approval / clearance Class IIb: All Spinal devices Class III Biologics (Drugs)
9 WHY ARE YOU PICKING ON US???????? Hip Recalls Zimmer, DePuy, Stryker Rengen Biologics MenaFlex ALL ORTHOPEDIC AND SPINE Synthes Spine - Norian MDT Agile DOJ Sen. Grassley Media RELATED
10 PAVING THE HIGHWAY Bound by regulations and precedent Need significant data to demonstrate safety and effectiveness Highest scientific rigor possible Valid Scientific Evidence Valid scientific evidence is evidence from well-controlled investigations, partially controlled studies, studies and objective trials without matched controls, welldocumented case histories conducted by qualified experts, and reports of significant human experience with a marketed device, from which it can fairly and responsibly be concluded by qualified experts that there is reasonable assurance of the safety and effectiveness of a device under its conditions of use. The evidence required may vary according to the characteristics of the device, its conditions of use, the existence and adequacy of warnings and other restrictions, and the extent of experience with its use.. Such information may be considered, however, in identifying a device the safety and effectiveness of which is questionable. Side by Side testing Need to characterize device (lower test parameter) No standard to compare It s what goes into the asphalt that makes the road smoother
11 Class II: Pedicle Based Systems Dynamic Systems Cleared 5 Systems within 2 years FDA getting innovative devices on the market 364 MDRs since 2005 (search criteria NQP) Problem: ASTM F1717 did not predict the failures seen clinically. Agile Recall Breaking in Shear (not part of ASTM F1717) Solution: 522 Studies Rigid Systems Blackstone Medical ICON Recall Unpredicted with mechanical testing ASTM F1717 meant to compare across devices, not mimic clinical environment Lack of evidence validating the loads and parameters from ASTM F1717 tell the clinical story Concept, Prototype, Preclinical Important
12 Bone GRAFT (Extenders) Bone Void Fillers ( Extenders ) Currently cleared posterolaterally with supplemental fixation Can t market BVF intradiscally Still 510(k) Need clinical data Allograft Cages Only indicated with autograft Allograft would need clinical data WHY? Provide valid scientific evidence of substantial equivalence Use of autograft still standard of care Show Us the DATA Bad to the Bone Graft
13 Bumps in the Road? Surgeons? Use a screwdriver as a rod? 5-level total disc replacement? Wear and Tear? Extensive mechanical testing, devices still break Did they compare to older weaker predicate Patients? Endplate failure Extrusion of device Did the patient follow the correct postoperative regiment? FDA Does Not Regulate Practice of Medicine
14 The 510(k) Predicament Planning and Knowledge Predicate may not be a predicate Old devices may not be viable predicate No split predicates Use of one predicate for material, one for mechanical results Clear and thorough risk analysis What are the differences (i.e., risks ) and how risk mitigated Possible side by side testing Early FDA interaction to manage timing and expectations Expect additional data needed Side by Side testing Animal data Clinical data Don t Get Yourself Stuck
15 STOP INNOVATION Just Kidding Barrier to Entry is Substantial Equivalence with a safety and effective device Technology outpaces scientific evidence to demonstrate safety and effectiveness Need that evidence to make decisions May be generated with standardized testing Significant limitations Limited applicability to new designs, materials, and treatment methods Limited precedent, limited ability to extend beyond regulations Industry s burden, not FDA s, to demonstrate substantial equivalence
16 Class III Examples TDRs Heterotopic Ossification Facet disease Out of ODE s hands: Manufacturing, IP Best study design = Right time, right patient Nucleus Replacements Issues with expulsion Feasibility study needed to evaluate safety Stopping rules regarding expulsions Avoided moving possible risky device into pivotal study Biologics INFUSE AMPLIFY OP-1 Off-label use, Study design, Risk/benefit profile
17 US Market versus CE Mark TDRs (Charite) CE Mark approximately 1998 Clinical study 1999, US market 2004 Interspinous Devices (X-STOP) CE Mark 2002 Clinical study 2000, US Approval 2005 BMP (OP-1, Infuse) Market Authorization 2001 (OP-1) Clinical study 1997, US Approval 2002 (INFUSE) EU is increasing level and amount of data for CE marking, while FDA closing the gap
18 PMA Success PMA success correlated to DESIGN and EXECUTION of the IDE study What does that mean?? Need to pick right indication Need to pick right endpoints Need to pick right sites Need to eliminate bias Valid statistical parameters FDA CAN HELP WITH THESE Means FDA is awesome
19 PMA Success Sequel (Execution) Patient Follow-up Adjudication of Adverse >85% events Independent Compliance Binding Surgeon/company Use of CEC/DSMB Site training Mechanism of Action Minimal study Why does the device work violations/deviations Radiographic Per Protocol Understanding failures Strict training program What went wrong Methods of correction (training, labeling, etc) FDA After Highest Scientific Rigor
20 Basic Ingredients Recipe for Success 2 Cup: Demonstration of non-inferiority Adequate statistical parameters 1 Cup: >85% patient accountability 1 Cup: Positive risk/benefit profile 2 Tbsp: Limited unexpected adverse events/observations 1 Tsp: Clear demonstration of mechanism of action 1 Tsp: Elimination of bias Add: Valid Manufacturing Process, Adequate Preclinical testing, IP Result: Nice Piping Hot Ready to Market Approved Device
21 Bullying or Common Sense Goal: Protect the public health Decisions based on science Preclinical testing Side by side testing Animal data Level 1 Clinical Evidence Prospective, randomized controlled study Timing often set by company, not FDA Approval timeliness based on data Address FDA s concerns There are ways not to provide side by side testing, clinical data, etc Justify response! Winning!
22 End of the Road Initiatives in place to add layer of transparency and streamline premarket process Regulatory precedent prevents quicker regulatory pathways Need to streamline reclassification petition process Clinical precedent guides review stringency Dynamic Stabilization Systems Clinical study design and execution is critical Don t forget manufacturing is important Always plan for a worst case timeframe and worst case budget Know your end goal Have the foresight to know what the market and landscape will be in 4-5 years Work with FDA or at least someone who knows what they re doing
23 Thank you
September 25, 2014. Dear Ms. McCoy:
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 September 25, 2014 Biomet
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 September 25, 2014 Biomet
ATLANTIS Anterior Cervical Plate System ATLANTIS Translational Plate. 510(k) Summary. February 2007
 rk,1 10{2 ATLANTIS Anterior Cervical Plate System ATLANTIS Translational Plate 510(k) Summary FEB 2 3 2[007 February 2007 I. Company: Medtronic Sofamor Danek USA 1800 Pyramid Place Memphis, Tennessee 38132
rk,1 10{2 ATLANTIS Anterior Cervical Plate System ATLANTIS Translational Plate 510(k) Summary FEB 2 3 2[007 February 2007 I. Company: Medtronic Sofamor Danek USA 1800 Pyramid Place Memphis, Tennessee 38132
June 9, 2015. Stryker Corporation Garry T. Hayeck, Ph.D. Senior Regulatory Affairs Specialist 2 Pearl Court Allendale, New Jersey 07401
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 Stryker Corporation Garry
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 Stryker Corporation Garry
510(k) Working Group. Preliminary Report and Recommendations
 CDRH PRELIMINARY INTERNAL EVALUATIONS VOLUME I 510(k) Working Group Preliminary Report and Recommendations August 2010 Center for Devices and Radiological Health U.S. Food and Drug Administration TABLE
CDRH PRELIMINARY INTERNAL EVALUATIONS VOLUME I 510(k) Working Group Preliminary Report and Recommendations August 2010 Center for Devices and Radiological Health U.S. Food and Drug Administration TABLE
Patient Information. Lateral Lumbar Interbody Fusion Surgery (LLIF).
 Patient Information. Lateral Lumbar Interbody Fusion Surgery (LLIF). Understanding your spine Disc Between each pair of vertebrae there is a disc that acts as a cushion to protect the vertebra, allows
Patient Information. Lateral Lumbar Interbody Fusion Surgery (LLIF). Understanding your spine Disc Between each pair of vertebrae there is a disc that acts as a cushion to protect the vertebra, allows
Task Force on the Utilization of Science in Regulatory Decision Making. Preliminary Report and Recommendations
 CDRH PRELIMINARY INTERNAL EVALUATIONS VOLUME II Task Force on the Utilization of Science in Regulatory Decision Making Preliminary Report and Recommendations August 2010 Center for Devices and Radiological
CDRH PRELIMINARY INTERNAL EVALUATIONS VOLUME II Task Force on the Utilization of Science in Regulatory Decision Making Preliminary Report and Recommendations August 2010 Center for Devices and Radiological
coligne treatment technology
 coligne treatment technology coligne a strategy in spine Leadership begins before the patient enters the surgical theater, among the spine care professionals forming the coalliance. This may be a small
coligne treatment technology coligne a strategy in spine Leadership begins before the patient enters the surgical theater, among the spine care professionals forming the coalliance. This may be a small
FDA STAFF MANUAL GUIDES, VOLUME I - ORGANIZATIONS AND FUNCTIONS FOOD AND DRUG ADMINISTRATION OFFICE OF OPERATIONS
 SMG 1117.2112 FDA STAFF MANUAL GUIDES, VOLUME I - ORGANIZATIONS AND FUNCTIONS FOOD AND DRUG ADMINISTRATION OFFICE OF OPERATIONS OFFICE OF INFORMATION MANAGEMENT AND TECHNOLOGY OFFICE OF INFORMATION MANAGEMENT
SMG 1117.2112 FDA STAFF MANUAL GUIDES, VOLUME I - ORGANIZATIONS AND FUNCTIONS FOOD AND DRUG ADMINISTRATION OFFICE OF OPERATIONS OFFICE OF INFORMATION MANAGEMENT AND TECHNOLOGY OFFICE OF INFORMATION MANAGEMENT
Information Sheet Guidance For IRBs, Clinical Investigators, and Sponsors
 Information Sheet Guidance For IRBs, Clinical Investigators, and Sponsors Frequently Asked Questions About Medical Devices Additional copies are available from: Office of Good Clinical Practice Office
Information Sheet Guidance For IRBs, Clinical Investigators, and Sponsors Frequently Asked Questions About Medical Devices Additional copies are available from: Office of Good Clinical Practice Office
Guidance for Industry and Food and Drug Administration Staff
 Requests for Feedback on Medical Device Submissions: The Pre-Submission Program and Meetings with Food and Drug Administration Staff Guidance for Industry and Food and Drug Administration Staff Document
Requests for Feedback on Medical Device Submissions: The Pre-Submission Program and Meetings with Food and Drug Administration Staff Guidance for Industry and Food and Drug Administration Staff Document
CLINICAL RESEARCH ORGANIZATION
 CLINICAL RESEARCH ORGANIZATION SERVICES Project Management Study Monitoring Data Management Statistics Regulatory Reimbursement Quality Assurance & Manufacturing Healthcare Compliance 1 A FULLY INTEGRATED
CLINICAL RESEARCH ORGANIZATION SERVICES Project Management Study Monitoring Data Management Statistics Regulatory Reimbursement Quality Assurance & Manufacturing Healthcare Compliance 1 A FULLY INTEGRATED
Patient Information. Anterior Cervical Discectomy and Fusion Surgery (ACDF).
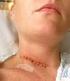 Patient Information. Anterior Cervical Discectomy and Fusion Surgery (ACDF). Understanding your spine Disc Between each pair of vertebrae there is a disc that acts as a cushion to protect the vertebra,
Patient Information. Anterior Cervical Discectomy and Fusion Surgery (ACDF). Understanding your spine Disc Between each pair of vertebrae there is a disc that acts as a cushion to protect the vertebra,
February 22, 2015. Life Spine, Incorporated Mr. Randy Lewis General Manager 2401 West Hassell Road, Suite 1535 Hoffman Estates, Illinois 60169
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 February 22, 2015 Life Spine, Incorporated Mr. Randy
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 February 22, 2015 Life Spine, Incorporated Mr. Randy
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Interspinous Fixation (Fusion) Devices Page 1 of 6 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: See Also: Interspinous Fixation (Fusion) Devices Lumbar Spine
Interspinous Fixation (Fusion) Devices Page 1 of 6 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: See Also: Interspinous Fixation (Fusion) Devices Lumbar Spine
Combination Products Regulation in the United States
 Combination Products Regulation in the United States Presenter: Scott Sardeson RAC US/EU 3M Health Care St. Paul, MN USA 1 Presentation Outline Combination products Definitions and Regulations Jurisdiction
Combination Products Regulation in the United States Presenter: Scott Sardeson RAC US/EU 3M Health Care St. Paul, MN USA 1 Presentation Outline Combination products Definitions and Regulations Jurisdiction
Total Product Lifecycle Solutions from NSF Health Sciences Medical Devices
 Total Product Lifecycle Solutions from NSF Health Sciences Medical Devices Experts in medical device quality systems, compliance, regulatory affairs, auditing and training www.nsf.org The Right People.
Total Product Lifecycle Solutions from NSF Health Sciences Medical Devices Experts in medical device quality systems, compliance, regulatory affairs, auditing and training www.nsf.org The Right People.
ORACLE CONSULTING GROUP
 ORACLE CONSULTING GROUP An Official United States Agent Firm for Foreign Establishments CONSULTING MEMORANDUM: DEALING WITH A MEDICAL DEVICE IN THE U.S. 5398 Golder Ranch Rd., Ste. 1 Tucson, Arizona 85739
ORACLE CONSULTING GROUP An Official United States Agent Firm for Foreign Establishments CONSULTING MEMORANDUM: DEALING WITH A MEDICAL DEVICE IN THE U.S. 5398 Golder Ranch Rd., Ste. 1 Tucson, Arizona 85739
Patient Guide to Lower Back Surgery
 The following is a sampling of products offered by Zimmer Spine for use in Open Lumbar Fusion procedures. Patient Guide to Lower Back Surgery Open Lumbar Fusion Dynesys The Dynesys Dynamic Stabilization
The following is a sampling of products offered by Zimmer Spine for use in Open Lumbar Fusion procedures. Patient Guide to Lower Back Surgery Open Lumbar Fusion Dynesys The Dynesys Dynamic Stabilization
Information Sheet Guidance For IRBs, Clinical Investigators, and Sponsors
 Information Sheet Guidance For IRBs, Clinical Investigators, and Sponsors FDA Institutional Review Board Inspections Additional copies are available from: Office of Good Clinical Practice Office of Special
Information Sheet Guidance For IRBs, Clinical Investigators, and Sponsors FDA Institutional Review Board Inspections Additional copies are available from: Office of Good Clinical Practice Office of Special
Clinical trials for medical devices: FDA and the IDE process
 Clinical trials for medical devices: FDA and the IDE process Owen Faris, Ph.D. Deputy Director Division of Cardiovascular Devices Office of Device Evaluation Center for Devices and Radiological Health,
Clinical trials for medical devices: FDA and the IDE process Owen Faris, Ph.D. Deputy Director Division of Cardiovascular Devices Office of Device Evaluation Center for Devices and Radiological Health,
Guidance for Industry. 21 CFR Part 11; Electronic. Records; Electronic Signatures. Time Stamps
 Guidance for Industry 21 CFR Part 11; Electronic Records; Electronic Signatures Time Stamps Draft Guidance This guidance document is being distributed for comment purposes only. Comments and suggestions
Guidance for Industry 21 CFR Part 11; Electronic Records; Electronic Signatures Time Stamps Draft Guidance This guidance document is being distributed for comment purposes only. Comments and suggestions
Overview of Drug Development: the Regulatory Process
 Overview of Drug Development: the Regulatory Process Roger D. Nolan, PhD Director, Project Operations Calvert Research Institute November, 2006 Adapted from course taught by Cato Research Background: Roger
Overview of Drug Development: the Regulatory Process Roger D. Nolan, PhD Director, Project Operations Calvert Research Institute November, 2006 Adapted from course taught by Cato Research Background: Roger
TABLE OF CONTENTS. Surgical Technique 2. Indications 4. Product Information 5. 1. Patient Positioning and Approach 2
 Surgical Technique TABLE OF CONTENTS Surgical Technique 2 1. Patient Positioning and Approach 2 2. Intervertebral Device Implanted 2 3. Buttress Plate Selection 2 4. Awl Insertion 2 5. Screw Insertion
Surgical Technique TABLE OF CONTENTS Surgical Technique 2 1. Patient Positioning and Approach 2 2. Intervertebral Device Implanted 2 3. Buttress Plate Selection 2 4. Awl Insertion 2 5. Screw Insertion
Reimbursement Overview Supporting Adjunctive Use of the coflex-f Implant During Lumbar Fusion Procedures
 Interlaminar Technology Reimbursement Overview Supporting Adjunctive Use of the coflex-f Implant During Lumbar Fusion Procedures Coding Recommendations Overview Implant Description & Device Type Differentiation
Interlaminar Technology Reimbursement Overview Supporting Adjunctive Use of the coflex-f Implant During Lumbar Fusion Procedures Coding Recommendations Overview Implant Description & Device Type Differentiation
FAST TRACK DEVELOPMENT OF EBOLA VACCINES: FDA REGULATORY PERSPECTIVE
 FAST TRACK DEVELOPMENT OF EBOLA VACCINES: FDA REGULATORY PERSPECTIVE Marion Gruber, Ph.D. Director Office of Vaccines Research & Review Center for Biologics Evaluation and Research US Food and Drug Administration
FAST TRACK DEVELOPMENT OF EBOLA VACCINES: FDA REGULATORY PERSPECTIVE Marion Gruber, Ph.D. Director Office of Vaccines Research & Review Center for Biologics Evaluation and Research US Food and Drug Administration
July 24, 2015. Dear Ms. Rhodes:
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 InnoVision, Incorporated
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center WO66-G609 Silver Spring, MD 20993-0002 InnoVision, Incorporated
Guidance for Industry
 Guidance for Industry Formal Dispute Resolution: Scientific and Technical Issues Related to Pharmaceutical CGMP U.S. Department of Health and Human Services Food and Drug Administration Center for Drug
Guidance for Industry Formal Dispute Resolution: Scientific and Technical Issues Related to Pharmaceutical CGMP U.S. Department of Health and Human Services Food and Drug Administration Center for Drug
Combination Products. Presented by: Karen S. Ginsbury For: IFF March 2014. PCI Pharma
 Combination Products Presented by: Karen S. Ginsbury For: IFF March 2014 Types of products Biological and medical device (freeze dried + syringe dual volume) Medical device and plasma devised product (syringe)
Combination Products Presented by: Karen S. Ginsbury For: IFF March 2014 Types of products Biological and medical device (freeze dried + syringe dual volume) Medical device and plasma devised product (syringe)
August 21, 2015. TCM Associates Ltd Mr. Iain Alligan Technical Director 3 Hillgrove Business Park Nazeing Road Essex EN9 2HB United Kingdom
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center - WO66-G609 Silver Spring, MD 20993-0002 Mr. Iain Alligan Technical
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center - WO66-G609 Silver Spring, MD 20993-0002 Mr. Iain Alligan Technical
Bone Morphogenetic Proteins & Spinal Surgery for Degenarative Disc Disease
 Ontario Health Technology Assessment Series 2004; Vol. 4, No. 4 Bone Morphogenetic Proteins & Spinal Surgery for Degenarative Disc Disease An Evidence Based Analysis March 2004 Medical Advisory Secretariat
Ontario Health Technology Assessment Series 2004; Vol. 4, No. 4 Bone Morphogenetic Proteins & Spinal Surgery for Degenarative Disc Disease An Evidence Based Analysis March 2004 Medical Advisory Secretariat
Patient Guide to Neck Surgery
 The following is a sampling of products offered by Zimmer Spine for use in Anterior Cervical Fusion procedures. Patient Guide to Neck Surgery Anterior Cervical Fusion Trinica Select With the Trinica and
The following is a sampling of products offered by Zimmer Spine for use in Anterior Cervical Fusion procedures. Patient Guide to Neck Surgery Anterior Cervical Fusion Trinica Select With the Trinica and
Introduction to Non-fusion Technologies in the Lumbar and Cervical Spine
 Introduction to Non-fusion Technologies in the Lumbar and Cervical Spine Introduction The term motion-sparing technology has been used to describe newer surgical techniques designed to preserve spinal
Introduction to Non-fusion Technologies in the Lumbar and Cervical Spine Introduction The term motion-sparing technology has been used to describe newer surgical techniques designed to preserve spinal
Boost the Success of Medical Device Development With Systematic Literature Reviews
 FOR PHARMA & LIFE SCIENCES WHITEPAPER Boost the Success of Medical Device Development With Systematic Literature Reviews Biomedical literature supports medical Device development Before the ideation and
FOR PHARMA & LIFE SCIENCES WHITEPAPER Boost the Success of Medical Device Development With Systematic Literature Reviews Biomedical literature supports medical Device development Before the ideation and
Biomechanics of the Lumbar Spine
 Biomechanics of the Lumbar Spine Biomechanics of the Spine 6 degrees of freedom Extension & Flexion Translation Rotation Compression & Distraction The disc/annulus/all/pll complex is the major restraint
Biomechanics of the Lumbar Spine Biomechanics of the Spine 6 degrees of freedom Extension & Flexion Translation Rotation Compression & Distraction The disc/annulus/all/pll complex is the major restraint
Guidance for Industry
 Guidance for Industry FDA Export Certificates Submit comments and suggestions regarding this document at anytime to Dockets Management Branch (HFA-305), Food and Drug Administration, 5630 Fishers Lane,
Guidance for Industry FDA Export Certificates Submit comments and suggestions regarding this document at anytime to Dockets Management Branch (HFA-305), Food and Drug Administration, 5630 Fishers Lane,
Technology Breakthrough in Spinal Implants (Technical Insights)
 Technology Breakthrough in Spinal Implants (Technical Insights) Biomaterial innovations is a growth factor for spinal implant market June 2014 Table of Contents Section Page Number Executive Summary 4
Technology Breakthrough in Spinal Implants (Technical Insights) Biomaterial innovations is a growth factor for spinal implant market June 2014 Table of Contents Section Page Number Executive Summary 4
LUMBAR SPINAL STENOSIS OBSERVATIONS, EVIDENCE, AND TRENDS FULILLING THE UNMET CLINICAL NEED WRITTEN BY: HALLETT MATHEWS, MD, MBA
 LUMBAR SPINAL STENOSIS OBSERVATIONS, EVIDENCE, AND TRENDS FULILLING THE UNMET CLINICAL NEED WRITTEN BY: HALLETT MATHEWS, MD, MBA Overview of Lumbar Spinal Stenosis Spine stabilization, which has equated
LUMBAR SPINAL STENOSIS OBSERVATIONS, EVIDENCE, AND TRENDS FULILLING THE UNMET CLINICAL NEED WRITTEN BY: HALLETT MATHEWS, MD, MBA Overview of Lumbar Spinal Stenosis Spine stabilization, which has equated
What is a medical device? Medical Devices: Roadmap to Market. Kathryn Klaus, Esq.
 Medical Devices: Roadmap to Market Kathryn Klaus, Esq. The last installment of Regulatory 360 discussed the FDA organization in general where it came from and a broad overview of how it operates, as well
Medical Devices: Roadmap to Market Kathryn Klaus, Esq. The last installment of Regulatory 360 discussed the FDA organization in general where it came from and a broad overview of how it operates, as well
Curriculum Vitae (Updated 06/09)
 Curriculum Vitae (Updated 06/09) JOHN M. SMALL, M.D. Business Address Florida Orthopaedic Institute 13020 N. Telecom Parkway Temple Terrace, FL 33637 Business Phone 813-978-9700 : March 27, 1958 Medical
Curriculum Vitae (Updated 06/09) JOHN M. SMALL, M.D. Business Address Florida Orthopaedic Institute 13020 N. Telecom Parkway Temple Terrace, FL 33637 Business Phone 813-978-9700 : March 27, 1958 Medical
ANTERIOR LUMBAR INTERBODY FUSION (ALIF) Basic Anatomical Landmarks: Anterior Lumbar Spine
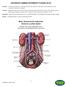 (ALIF) Anterior In human anatomy, referring to the front surface of the body or the position of one structure relative to another Lumbar Relating to the loins or the section of the back and sides between
(ALIF) Anterior In human anatomy, referring to the front surface of the body or the position of one structure relative to another Lumbar Relating to the loins or the section of the back and sides between
Diagnostic Tests. Brad Spring Director, Regulatory Affairs
 Regulatory Challenges in the Development of Diagnostic Tests Industry Perspective Brad Spring Director, Regulatory Affairs September 28, 2011 Balancing Evidence of Safety & Effectiveness and Time to Market
Regulatory Challenges in the Development of Diagnostic Tests Industry Perspective Brad Spring Director, Regulatory Affairs September 28, 2011 Balancing Evidence of Safety & Effectiveness and Time to Market
Artificial Intervertebral Disc: Cervical Spine
 dapplies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
dapplies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Custom Device Exemption. Draft Guidance for Industry and Food and Drug Administration Staff
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 Custom Device Exemption Draft Guidance for Industry and Food and Drug Administration Staff DRAFT GUIDANCE This guidance
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 Custom Device Exemption Draft Guidance for Industry and Food and Drug Administration Staff DRAFT GUIDANCE This guidance
March 3, 2015. Dear Ms. Alice Gong,
 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center - WO66-G609 Silver Spring, MD 20993-0002 Jiangsu Ideal Medical
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control Center - WO66-G609 Silver Spring, MD 20993-0002 Jiangsu Ideal Medical
IVD Regulation Overview. Requirements to Assure Quality & Effectiveness
 IVD Regulation Overview Requirements to Assure Quality & Effectiveness CLIAC Jan. 2002 Statutory and Regulatory Requirements Statute: Food, Drug, and Cosmetic Act Food and Drugs Act of 1906 Food and Drug
IVD Regulation Overview Requirements to Assure Quality & Effectiveness CLIAC Jan. 2002 Statutory and Regulatory Requirements Statute: Food, Drug, and Cosmetic Act Food and Drugs Act of 1906 Food and Drug
FDA Regulation of Hearing Aids. Eric A. Mann, MD, PhD Clinical Deputy Director Division of Ophthalmic and ENT Devices ODE/CDRH/FDA
 FDA Regulation of Hearing Aids Eric A. Mann, MD, PhD Clinical Deputy Director Division of Ophthalmic ENT ODE/CDRH/FDA U.S. Food Drug Administration Presentation Outline Overview of device regulations riskbased
FDA Regulation of Hearing Aids Eric A. Mann, MD, PhD Clinical Deputy Director Division of Ophthalmic ENT ODE/CDRH/FDA U.S. Food Drug Administration Presentation Outline Overview of device regulations riskbased
Guidance for Industry. 21 CFR Part 11; Electronic Records; Electronic Signatures. Maintenance of Electronic Records
 Draft Guidance for Industry -- Not For Implementation Guidance for Industry 21 CFR Part 11; Electronic Records; Electronic Signatures Maintenance of Electronic Records Draft Guidance This guidance document
Draft Guidance for Industry -- Not For Implementation Guidance for Industry 21 CFR Part 11; Electronic Records; Electronic Signatures Maintenance of Electronic Records Draft Guidance This guidance document
Does the pain radiating down your legs, buttocks or lower back prevent you from walking long distances?
 Does the pain radiating down your legs, buttocks or lower back prevent you from walking long distances? Do you experience weakness, tingling, numbness, stiffness, or cramping in your legs, buttocks or
Does the pain radiating down your legs, buttocks or lower back prevent you from walking long distances? Do you experience weakness, tingling, numbness, stiffness, or cramping in your legs, buttocks or
ANTERIOR CERVICAL DISCECTOMY AND FUSION. Basic Anatomical Landmarks: Anterior Cervical Spine
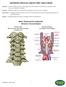 Anterior In the human anatomy, referring to the front surface of the body or position of one structure relative to another Cervical Relating to the neck, in the spine relating to the first seven vertebrae
Anterior In the human anatomy, referring to the front surface of the body or position of one structure relative to another Cervical Relating to the neck, in the spine relating to the first seven vertebrae
Motion Preservation. Hansen Yuan, MD President, Spine Arthroplasty Society
 Motion Preservation Procedure Codes Hansen Yuan, MD President, Spine Arthroplasty Society Who are we? The Spine Arthroplasty Society (SAS) is a group of medical and associated specialists devoted to the
Motion Preservation Procedure Codes Hansen Yuan, MD President, Spine Arthroplasty Society Who are we? The Spine Arthroplasty Society (SAS) is a group of medical and associated specialists devoted to the
Enabling Faster, Better Medical Device Development and Evaluation with Modeling and Simulation Tina Morrison PhD
 Enabling Faster, Better Medical Device Development and Evaluation with Modeling and Simulation Tina Morrison PhD Office of Device Evaluation Center for Devices and Radiological Health U.S. Food and Drug
Enabling Faster, Better Medical Device Development and Evaluation with Modeling and Simulation Tina Morrison PhD Office of Device Evaluation Center for Devices and Radiological Health U.S. Food and Drug
Guidance for Industry. 21 CFR Part 11; Electronic Records; Electronic Signatures. Electronic Copies of Electronic Records
 Guidance for Industry 21 CFR Part 11; Electronic Records; Electronic Signatures Electronic Copies of Electronic Records Draft Guidance This guidance document is being distributed for comment purposes only.
Guidance for Industry 21 CFR Part 11; Electronic Records; Electronic Signatures Electronic Copies of Electronic Records Draft Guidance This guidance document is being distributed for comment purposes only.
How to Use the Design Process to Manage Risk: Elements of Design Controls and Why It Matters
 environmental failure analysis & prevention health technology development How to Use the Design Process to Manage Risk: Elements of Design Controls and Why It Matters Kevin L. Ong, Ph.D., P.E. Managing
environmental failure analysis & prevention health technology development How to Use the Design Process to Manage Risk: Elements of Design Controls and Why It Matters Kevin L. Ong, Ph.D., P.E. Managing
MEDICAL DEVICE & DIAGNOSTICS
 to Guide Strategic Medical Device and Introduction Medical device and diagnostic makers face myriad challenges when developing a new product, including working within resource and financial constraints,
to Guide Strategic Medical Device and Introduction Medical device and diagnostic makers face myriad challenges when developing a new product, including working within resource and financial constraints,
Clinical evaluation Latest development in expectations EU and USA
 Clinical evaluation Latest development in expectations EU and USA Medical Devices: staying ahead of regulatory developments Gert Bos BSI Israel 22 April - Herzliya Copyright 2012 BSI. All rights reserved.
Clinical evaluation Latest development in expectations EU and USA Medical Devices: staying ahead of regulatory developments Gert Bos BSI Israel 22 April - Herzliya Copyright 2012 BSI. All rights reserved.
ISPI Newsletter Archive Lumbar Spine Surgery
 ISPI Newsletter Archive Lumbar Spine Surgery January 2005 Effects of Charite Artificial Disc on the Implanted and Adjacent Spinal Segments Mechanics Using a Hybrid Testing Protocol Spine. 30(24):2755-2764,
ISPI Newsletter Archive Lumbar Spine Surgery January 2005 Effects of Charite Artificial Disc on the Implanted and Adjacent Spinal Segments Mechanics Using a Hybrid Testing Protocol Spine. 30(24):2755-2764,
What Does Having a FDA Cleared Pregnancy Test Mean?
 What Does Having a FDA Cleared Pregnancy Test Mean? Clinical Trials Transformation Initiative (CTTI) July 15-16, 2013 Denise N. Johnson-Lyles, Ph.D. Toxicology Branch Chief Division of Chemistry and Toxicology
What Does Having a FDA Cleared Pregnancy Test Mean? Clinical Trials Transformation Initiative (CTTI) July 15-16, 2013 Denise N. Johnson-Lyles, Ph.D. Toxicology Branch Chief Division of Chemistry and Toxicology
Guidance for Industry and FDA Staff Tonometers - Premarket Notification [510(k)] Submissions
![Guidance for Industry and FDA Staff Tonometers - Premarket Notification [510(k)] Submissions Guidance for Industry and FDA Staff Tonometers - Premarket Notification [510(k)] Submissions](/thumbs/39/18581577.jpg) Guidance for Industry and FDA Staff Tonometers - Premarket Notification [510(k)] Submissions Document issued on: March 27, 2006 For questions regarding this document, contact Everette Beers, Ph.D. at 240-276-4200
Guidance for Industry and FDA Staff Tonometers - Premarket Notification [510(k)] Submissions Document issued on: March 27, 2006 For questions regarding this document, contact Everette Beers, Ph.D. at 240-276-4200
Office of Acquisitions & Grants Services (OAGS) FDA Small Business Outreach Vendor Fair. February 19, 2014
 Office of Acquisitions & Grants Services (OAGS) FDA Small Business Outreach Vendor Fair February 19, 2014 FDA Mission FDA is responsible for: Protecting the public health by assuring that foods are safe,
Office of Acquisitions & Grants Services (OAGS) FDA Small Business Outreach Vendor Fair February 19, 2014 FDA Mission FDA is responsible for: Protecting the public health by assuring that foods are safe,
visualized. The correct level is then identified again. With the use of a microscope and
 SURGERY FOR SPINAL STENOSIS Laminectomy A one inch (or longer for extensive stenosis) incision is made in the middle of the back over the effected region of the spine. The muscles over the bone are moved
SURGERY FOR SPINAL STENOSIS Laminectomy A one inch (or longer for extensive stenosis) incision is made in the middle of the back over the effected region of the spine. The muscles over the bone are moved
CTC Technology Readiness Levels
 CTC Technology Readiness Levels Readiness: Software Development (Adapted from CECOM s Software Technology Readiness Levels) Level 1: Basic principles observed and reported. Lowest level of software readiness.
CTC Technology Readiness Levels Readiness: Software Development (Adapted from CECOM s Software Technology Readiness Levels) Level 1: Basic principles observed and reported. Lowest level of software readiness.
SPINAL FUSION. North American Spine Society Public Education Series
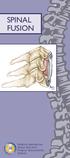 SPINAL FUSION North American Spine Society Public Education Series WHAT IS SPINAL FUSION? The spine is made up of a series of bones called vertebrae ; between each vertebra are strong connective tissues
SPINAL FUSION North American Spine Society Public Education Series WHAT IS SPINAL FUSION? The spine is made up of a series of bones called vertebrae ; between each vertebra are strong connective tissues
Plamena Entcheva-Dimitrov, PhD, RAC On-line Course. www.preferredregulatoryconsulting.com 1
 Plamena Entcheva-Dimitrov, PhD, RAC On-line Course 1 The Preferred Regulatory Consulting content in this presentation is copyright protected under United States law and applicable international copyright
Plamena Entcheva-Dimitrov, PhD, RAC On-line Course 1 The Preferred Regulatory Consulting content in this presentation is copyright protected under United States law and applicable international copyright
Regulatory Pathways for Licensure and Use of Ebola Virus Vaccines During the Current Outbreak FDA Perspective
 Regulatory Pathways for Licensure and Use of Ebola Virus Vaccines During the Current Outbreak FDA Perspective Office of Vaccines Research and Review Center for Biologics Evaluation and Research U.S. Food
Regulatory Pathways for Licensure and Use of Ebola Virus Vaccines During the Current Outbreak FDA Perspective Office of Vaccines Research and Review Center for Biologics Evaluation and Research U.S. Food
Guidance for Clinical Investigators, Sponsors, and IRBs
 Guidance for Clinical Investigators, Sponsors, and IRBs Adverse Event Reporting to IRBs Improving Human Subject Protection U.S. Department of Health and Human Services Office of the Commissioner (OC) Center
Guidance for Clinical Investigators, Sponsors, and IRBs Adverse Event Reporting to IRBs Improving Human Subject Protection U.S. Department of Health and Human Services Office of the Commissioner (OC) Center
Introduction to Post marketing Drug Safety Surveillance: Pharmacovigilance in FDA/CDER
 Introduction to Post marketing Drug Safety Surveillance: Pharmacovigilance in FDA/CDER LT Andrew Fine, Pharm.D., BCPS Safety Evaluator Division of Pharmacovigilance Office of Pharmacovigilance and Epidemiology
Introduction to Post marketing Drug Safety Surveillance: Pharmacovigilance in FDA/CDER LT Andrew Fine, Pharm.D., BCPS Safety Evaluator Division of Pharmacovigilance Office of Pharmacovigilance and Epidemiology
DUKE ORTHOPAEDIC SURGERY GOALS AND OBJECTIVES SPINE SERVICE
 GOALS AND OBJECTIVES PATIENT CARE Able to perform a complete musculoskeletal and neurologic examination on the patient including cervical spine, thoracic spine, and lumbar spine. The neurologic examination
GOALS AND OBJECTIVES PATIENT CARE Able to perform a complete musculoskeletal and neurologic examination on the patient including cervical spine, thoracic spine, and lumbar spine. The neurologic examination
Guidance for Industry
 Guidance for Industry Container and Closure System Integrity Testing in Lieu of Sterility Testing as a Component of the Stability Protocol for Sterile Products For questions on the content of the guidance,
Guidance for Industry Container and Closure System Integrity Testing in Lieu of Sterility Testing as a Component of the Stability Protocol for Sterile Products For questions on the content of the guidance,
Regulatory Science Support of Device Innovation Eugene Civillico, Ph.D. Cristin Welle, Ph.D.
 Regulatory Science Support of Device Innovation Eugene Civillico, Ph.D. Cristin Welle, Ph.D. Center for Devices and Radiological Health Office of Science and Engineering Laboratories Division of Biomedical
Regulatory Science Support of Device Innovation Eugene Civillico, Ph.D. Cristin Welle, Ph.D. Center for Devices and Radiological Health Office of Science and Engineering Laboratories Division of Biomedical
AC 2007-668: UNDERSTANDING THE MEDICAL PRODUCT DEVELOPMENT PROCESS: CONTINUING PROFESSIONAL DEVELOPMENT FOR LIFE SCIENCE PROFESSIONALS
 AC 2007-668: UNDERSTANDING THE MEDICAL PRODUCT DEVELOPMENT PROCESS: CONTINUING PROFESSIONAL DEVELOPMENT FOR LIFE SCIENCE PROFESSIONALS Rogelio Rodriguez, UC Irvine Rogelio C. Rodriguez, M.S. Director,
AC 2007-668: UNDERSTANDING THE MEDICAL PRODUCT DEVELOPMENT PROCESS: CONTINUING PROFESSIONAL DEVELOPMENT FOR LIFE SCIENCE PROFESSIONALS Rogelio Rodriguez, UC Irvine Rogelio C. Rodriguez, M.S. Director,
Metal-on-Metal Hip Systems
 Meeting of the Orthopaedic and Rehabilitation Devices Advisory Panel June 27-28, 2012 Metal-on-Metal Hip Systems Center for Devices and Radiological Health U.S. Food and Drug Administration Meeting of
Meeting of the Orthopaedic and Rehabilitation Devices Advisory Panel June 27-28, 2012 Metal-on-Metal Hip Systems Center for Devices and Radiological Health U.S. Food and Drug Administration Meeting of
510(k) Summary. Dentsply Implants. April 30, 2013
 5 1 0(k) Summary AtlantisTM Straumann Bone Level Abutment 510(k) Summary Dentsply Implants MAY 1 2013 ADMINISTRATIVE INFORMATION AtlantisTM Straumann Bone Level Abutment K130216 April 30, 2013 Manufacturer
5 1 0(k) Summary AtlantisTM Straumann Bone Level Abutment 510(k) Summary Dentsply Implants MAY 1 2013 ADMINISTRATIVE INFORMATION AtlantisTM Straumann Bone Level Abutment K130216 April 30, 2013 Manufacturer
FDA TRANSPARENCY INITIATIVE: IMPROVING TRANSPARENCY TO REGULATED INDUSTRY
 FDA TRANSPARENCY INITIATIVE: IMPROVING TRANSPARENCY TO REGULATED INDUSTRY TRANSPARENCY TASK FORCE U.S. DEPARTMENT OF HEALTH AND HUMAN SERVICES FOOD AND DRUG ADMINISTRATION JANUARY 2011 TABLE OF CONTENTS
FDA TRANSPARENCY INITIATIVE: IMPROVING TRANSPARENCY TO REGULATED INDUSTRY TRANSPARENCY TASK FORCE U.S. DEPARTMENT OF HEALTH AND HUMAN SERVICES FOOD AND DRUG ADMINISTRATION JANUARY 2011 TABLE OF CONTENTS
Custom Device Exemption Guidance for Industry and Food and Drug Administration Staff
 Custom Device Exemption Guidance for Industry and Food and Drug Administration Staff Document issued on: September 24, 2014 The draft of this document was issued on January 14, 2014. An agency may not
Custom Device Exemption Guidance for Industry and Food and Drug Administration Staff Document issued on: September 24, 2014 The draft of this document was issued on January 14, 2014. An agency may not
Low Back Pain (LBP) Prevalence. Low Back Pain (LBP) Prevalence. Lumbar Fusion: Where is the Evidence?
 15 th Annual Cleveland Clinic Pain Management Symposium Sarasota, Florida Lumbar Fusion: Where is the Evidence? Gordon R. Bell, M.D. Director, Cleveland Clinic Low Back Pain (LBP) Prevalence Lifetime prevalence:
15 th Annual Cleveland Clinic Pain Management Symposium Sarasota, Florida Lumbar Fusion: Where is the Evidence? Gordon R. Bell, M.D. Director, Cleveland Clinic Low Back Pain (LBP) Prevalence Lifetime prevalence:
Moderator and Panelists
 Panelists Moderator and Panelists Moderator Dr. Hallett Mathews Executive Vice President Chief Medical Officer Paradigm Spine, LLC Panelist Dr. John Peloza Orthopedic Surgeon The Center for Spine Care
Panelists Moderator and Panelists Moderator Dr. Hallett Mathews Executive Vice President Chief Medical Officer Paradigm Spine, LLC Panelist Dr. John Peloza Orthopedic Surgeon The Center for Spine Care
Simpler Implant Solutions Inc. 2930 Arbutus St. Vancouver, BC V6i 3Y9. haroldbergmanl~ogmail.com 604 736 9890 Telephone 604 736 9747 Fax K080115
 APR 5 2013 Summary: K121281 Submitter: Company Address Contact Person: Simpler Implant Solutions Inc. 2930 Arbutus St. Vancouver, BC V6i 3Y9 Dr. Harold Bergman haroldbergmanl~ogmail.com 604 736 9890 Telephone
APR 5 2013 Summary: K121281 Submitter: Company Address Contact Person: Simpler Implant Solutions Inc. 2930 Arbutus St. Vancouver, BC V6i 3Y9 Dr. Harold Bergman haroldbergmanl~ogmail.com 604 736 9890 Telephone
Title: Department: Approved by:
 Title: Department: Emergency Use of an Investigational Drug or Biological Product, or Unapproved Medical Device Human Research Affairs Policy Type: Partners System-wide Partners System-wide Template Partners
Title: Department: Emergency Use of an Investigational Drug or Biological Product, or Unapproved Medical Device Human Research Affairs Policy Type: Partners System-wide Partners System-wide Template Partners
Guidance Medication Guides Distribution Requirements and Inclusion in Risk Evaluation and Mitigation Strategies (REMS)
 Guidance s Distribution Requirements and Inclusion in Risk Evaluation and Mitigation Strategies (REMS) U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation
Guidance s Distribution Requirements and Inclusion in Risk Evaluation and Mitigation Strategies (REMS) U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation
2014 Annual Report on Inspections of Establishments
 2014 Annual Report on Inspections of Establishments Table of Contents Introduction... 2 Data Collection and Definitions... 3 Section 510(h)(6)(A)(i) Number of Domestic and Foreign Establishments Registered
2014 Annual Report on Inspections of Establishments Table of Contents Introduction... 2 Data Collection and Definitions... 3 Section 510(h)(6)(A)(i) Number of Domestic and Foreign Establishments Registered
Minimally Invasive Lumbar Fusion
 Minimally Invasive Lumbar Fusion Biomechanical Evaluation (1) coflex-f screw Biomechanical Evaluation (1) coflex-f intact Primary Stability intact Primary Stability Extension Neutral Position Flexion Coflex
Minimally Invasive Lumbar Fusion Biomechanical Evaluation (1) coflex-f screw Biomechanical Evaluation (1) coflex-f intact Primary Stability intact Primary Stability Extension Neutral Position Flexion Coflex
POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS DISCLAIMER CODING INFORMATION REFERENCES POLICY HISTORY
 Original Issue Date (Created): 6/14/2004 Most Recent Review Date (Revised): 9/29/2015 Effective Date: 12/1/2015 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS
Original Issue Date (Created): 6/14/2004 Most Recent Review Date (Revised): 9/29/2015 Effective Date: 12/1/2015 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS
U.S. Food and Drug Administration Center for Devices and Radiological Health REGULATORY SCIENCE PRIORITIES (FY2016)
 U.S. Food and Drug Administration Center for Devices and Radiological Health REGULATORY SCIENCE PRIORITIES (FY2016) Executive Summary October 20, 2015 The Regulatory Science Subcommittee (RSS) of the CDRH
U.S. Food and Drug Administration Center for Devices and Radiological Health REGULATORY SCIENCE PRIORITIES (FY2016) Executive Summary October 20, 2015 The Regulatory Science Subcommittee (RSS) of the CDRH
The Product Review Life Cycle A Brief Overview
 Stat & Quant Mthds Pharm Reg (Spring 2, 2014) Lecture 2,Week 1 1 The review process developed over a 40 year period and has been influenced by 5 Prescription User Fee Act renewals Time frames for review
Stat & Quant Mthds Pharm Reg (Spring 2, 2014) Lecture 2,Week 1 1 The review process developed over a 40 year period and has been influenced by 5 Prescription User Fee Act renewals Time frames for review
Presented by Zoran Maric, M.D. Orthopaedic Spine Surgeon May 22, 2010
 Presented by Zoran Maric, M.D. Orthopaedic Spine Surgeon May 22, 2010 1 cervical area thoracic area lumbar area sacrum coccyx Mayfield Clinic 2 3 4 5 Zoran Maric, MD Spine Surgery Procedures How to Document
Presented by Zoran Maric, M.D. Orthopaedic Spine Surgeon May 22, 2010 1 cervical area thoracic area lumbar area sacrum coccyx Mayfield Clinic 2 3 4 5 Zoran Maric, MD Spine Surgery Procedures How to Document
A Cost Effective Way to De Risk Biomarker Clinical Trials: Early Development Considerations
 A Cost Effective Way to De Risk Biomarker Clinical Trials: Early Development Considerations Ce3, Inc. and Insight Genetics, Inc. Oncology Forum July 15, 2015 Agenda Introductions Definitions Regulations
A Cost Effective Way to De Risk Biomarker Clinical Trials: Early Development Considerations Ce3, Inc. and Insight Genetics, Inc. Oncology Forum July 15, 2015 Agenda Introductions Definitions Regulations
kok1 UQ 510(k)J Device {Applicant 510(k) Summary (per 21 CFR 807.92(c)) OCT 2 7 2008 1. Applicant
 kok1 UQ 510(k) Summary (per 21 CFR 807.92(c)) OCT 2 7 2008 1. Applicant Tech Avenue Ventures d/b/a MPowRx Health and Wellness Products Inc. #510 3553-31 St. NW Calgary, Alberta T2L 2K7 Canada Contact Person:
kok1 UQ 510(k) Summary (per 21 CFR 807.92(c)) OCT 2 7 2008 1. Applicant Tech Avenue Ventures d/b/a MPowRx Health and Wellness Products Inc. #510 3553-31 St. NW Calgary, Alberta T2L 2K7 Canada Contact Person:
PREP Course #27: Medical Device Clinical Trial Management
 PREP Course #27: Medical Device Clinical Trial Management Presented by: Evelyn Huang Jeffrey Revello Office of Research Compliance North Shore-LIJ Health System CME Disclosure Statement The North Shore
PREP Course #27: Medical Device Clinical Trial Management Presented by: Evelyn Huang Jeffrey Revello Office of Research Compliance North Shore-LIJ Health System CME Disclosure Statement The North Shore
Guidance for Industry Part 11, Electronic Records; Electronic Signatures Scope and Application
 Guidance for Industry Part 11, Electronic Records; Electronic Signatures Scope and Application U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research
Guidance for Industry Part 11, Electronic Records; Electronic Signatures Scope and Application U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research
How To Write A Cer
 2014 CBER Update: Advertising & Promotional Labeling Compliance Alpita Popat, PharmD, MBA Consumer Safety Officer Advertising & Promotional Labeling Branch Division of Case Management Office of Compliance
2014 CBER Update: Advertising & Promotional Labeling Compliance Alpita Popat, PharmD, MBA Consumer Safety Officer Advertising & Promotional Labeling Branch Division of Case Management Office of Compliance
Minimally Invasive Spine Surgery What is it and how will it benefit patients?
 Minimally Invasive Spine Surgery What is it and how will it benefit patients? Dr Raoul Pope MBChB, FRACS, Neurosurgeon and Minimally Invasive Spine Surgeon Concord Hospital and Mater Private Hospital Sydney
Minimally Invasive Spine Surgery What is it and how will it benefit patients? Dr Raoul Pope MBChB, FRACS, Neurosurgeon and Minimally Invasive Spine Surgeon Concord Hospital and Mater Private Hospital Sydney
L~l t l V~! MAY 23 200?
 L~l t l V~! MAY 23 200? 510 (k) Summay (As required by 21 CFR 807.92 and 21 CFR 807.93) NAME OF SPONSOR: 510(K) CONTACT: DePuy Orthopaedics, Inc. 700 Orthopaedic Drive Warsaw, Indiana 46582 Establishment
L~l t l V~! MAY 23 200? 510 (k) Summay (As required by 21 CFR 807.92 and 21 CFR 807.93) NAME OF SPONSOR: 510(K) CONTACT: DePuy Orthopaedics, Inc. 700 Orthopaedic Drive Warsaw, Indiana 46582 Establishment
Clinical Data for Medical Devices
 White Paper Clinical Data for Medical Devices Preparing for increased requirements in the EU Table of Contents 1. Regulation of medical devices in the EU: on the cusp of change... 3 2. Traditional differences
White Paper Clinical Data for Medical Devices Preparing for increased requirements in the EU Table of Contents 1. Regulation of medical devices in the EU: on the cusp of change... 3 2. Traditional differences
[DOCKET NO.96N-0002] DRAFT
![[DOCKET NO.96N-0002] DRAFT [DOCKET NO.96N-0002] DRAFT](/thumbs/26/7603707.jpg) [DOCKET NO.96N-0002] DRAFT DRAFT DOCUMENT CONCERNING THE REGULATION OF PLACENTAL/UMBILICAL CORD BLOOD STEM CELL PRODUCTS INTENDED FOR TRANSPLANTATION OR FURTHER MANUFACTURE INTO INJECTABLE PRODUCTS DECEMBER,
[DOCKET NO.96N-0002] DRAFT DRAFT DOCUMENT CONCERNING THE REGULATION OF PLACENTAL/UMBILICAL CORD BLOOD STEM CELL PRODUCTS INTENDED FOR TRANSPLANTATION OR FURTHER MANUFACTURE INTO INJECTABLE PRODUCTS DECEMBER,
Subject: Implanted Devices for Spinal Stenosis Policy #: SURG.00092 Current Effective Date: 07/13/2011 Status: Reviewed Last Review Date: 05/19/2011
 1 of 5 6/18/2012 11:08 AM Medical Policy Subject: Implanted Devices for Spinal Stenosis Policy #: SURG.00092 Current Effective Date: 07/13/2011 Status: Reviewed Last Review Date: 05/19/2011 Description/Scope
1 of 5 6/18/2012 11:08 AM Medical Policy Subject: Implanted Devices for Spinal Stenosis Policy #: SURG.00092 Current Effective Date: 07/13/2011 Status: Reviewed Last Review Date: 05/19/2011 Description/Scope
2013 North American SSL Certificate
 2013 2014 INSERT COMPANY LOGO HERE 2014 North American 2013 North American SSL Certificate Minimally Invasive Spinal Surgical Solutions Product Product Line Leadership Strategy Leadership Award Award Background
2013 2014 INSERT COMPANY LOGO HERE 2014 North American 2013 North American SSL Certificate Minimally Invasive Spinal Surgical Solutions Product Product Line Leadership Strategy Leadership Award Award Background
510(k) Summary. Atlanti STM Abutment and AtlantisTM Crown Abutment in Zirconia for Dentsply Ankylos Implant
 /p(so 3 Premarket Notification SectionS5: Page - 4 JUL 5 2012 510(k) Summary Astra Tech Inc. Atlantisim Abutment and Atlantis~m Crown Abutment in Zirconia for Dentsply Ankylos Implant ADMINISTRATIVE INFORMATION
/p(so 3 Premarket Notification SectionS5: Page - 4 JUL 5 2012 510(k) Summary Astra Tech Inc. Atlantisim Abutment and Atlantis~m Crown Abutment in Zirconia for Dentsply Ankylos Implant ADMINISTRATIVE INFORMATION
Guidance for Sponsors, Institutional Review Boards, Clinical Investigators and FDA Staff
 Guidance for Sponsors, Institutional Review Boards, Clinical Investigators and FDA Staff Guidance on Informed Consent for In Vitro Diagnostic Device Studies Using Leftover Human Specimens that are Not
Guidance for Sponsors, Institutional Review Boards, Clinical Investigators and FDA Staff Guidance on Informed Consent for In Vitro Diagnostic Device Studies Using Leftover Human Specimens that are Not
REPUTATION MANAGEMENT: SPINAL FUSION SURGERY
 REPUTATION MANAGEMENT: SPINAL FUSION SURGERY Meaningful and Positive Progress in Spinal Fusion Results: A Journey towards Excellence Paul J. Slosar, M.D. SpineCare Medical Group As a practicing spine surgeon,
REPUTATION MANAGEMENT: SPINAL FUSION SURGERY Meaningful and Positive Progress in Spinal Fusion Results: A Journey towards Excellence Paul J. Slosar, M.D. SpineCare Medical Group As a practicing spine surgeon,
