Grand Challenge: Innovations in Vaccine Manufacturing for Global Markets FAQ
|
|
|
- Jennifer Warren
- 3 years ago
- Views:
From this document you will learn the answers to the following questions:
What is the cost of a per - vaccine target?
What type of structure is the foundation open to in this challenge?
What is the main focus of the Grand Challenge?
Transcription
1 Grand Challenge: Innovations in Vaccine Manufacturing for Global Markets FAQ Grant Objectives Is $0.15/dose cost a per-vaccine target or per-disease target? In other words, can pentavalent vaccine come in at a total cost of $0.75/dose because it targets 5 diseases? The cost of $0.15/dose is a per-vaccine target. Does $0.15/dose cost include packaging? Yes, the cost includes packaging and labelling, ready to move the product into a distribution channel. Can you provide unifying assumptions about any of the cost categories encompassed in the target $0.15/dose (consumables, direct and indirect labor, facility, equipment and related costs, QA/QC costs, fill-finish, and licensing costs)? This Grand Challenge values innovative thinking. Any attempt on our part to offer cost assumptions has the potential to constrain thinking. Proposers should develop sound arguments to support any assumptions made in parsing out cost categories. Can I develop a new vaccine to manufacture? No. This Grand Challenge is focused on development of innovative platform technologies for the production of existing vaccines and not discovery of novel untested vaccines. The vaccine(s) selected must be one(s) for which there are established quality standards and clinical endpoints so that a quality comparison may be made between the existing and novel platform technologies. What team structure are you looking for? Am I required to designate an integrator or contact PI? The foundation requires that one lead principal investigator be designated for the project. Beyond that, the foundation is open to whatever team and leadership structure the proposer deems best suited to accomplish the objectives stated within the Challenge. The makeup and strength of the team will be evaluated in the initial letter of intent review phase, after which the foundation may offer comments or suggestions. Should the 5 year plan include human studies? 1 P a g e
2 No. While it is the foundation s expectation that this platform may require a clinical study, this 5 year project is not expected to include that study. By the end of year 5, an IND submission must be made to the appropriate national regulatory agency to begin clinical trial testing of vaccine produced using the new process Can the foundation assist me in getting access to bulk product? No. For work that requires access to virus strains, proposers should consider with whom to engage as well as how that access requirement affects the critical path to development. What steps might the foundation take in support of Global Access? To ensure Global Access, the foundation may: Conduct due diligence and/or request an IP Report to determine the ability of the Applicant to effectively manage the IP Rights associated with the Grand Challenge in such a way that will achieve Global Access. Require a Global Access Strategy or Global Access Commitment Agreement from the Applicant, explaining their plans to achieve their goals and further the foundation s charitable objectives of Global Access, including the identification of third party IP Rights and relationships arising in connection with the research, development, use, manufacturing, marketing, commercialization and/or distribution of the Funded Developments, and the related IP management strategies, licensing structures, data management plans, and pricing frameworks. Require periodic updates of the Global Access Strategy to detail progress and ongoing efforts to achieve Global Access. Reserve IP Rights in Funded Developments. In those cases where the foundation does reserve IP Rights, it does so as necessary to ensure that either the foundation (and/or its grantees, partners or collaborators) has the IP Rights necessary for the research, development, use, manufacturing, marketing, commercialization and/or distribution of Funded Developments (as applicable) to achieve Global Access. More information about the foundation s global access approach can be found at: Application How do I submit a Letter of Intent? Please submit your LOI through the Unison application portal, found here (applicants must first create a log-in for Unison.) For reference, the RFP number is SOL Please see the Grand Challenge for specific guidance on what information your LOI should include. Who is eligible to submit a Letter of Intent? Please refer to page 8 of the Grand Challenge for eligibility information. 2 P a g e
3 Does the 3 page limit include (e.g., graphs, tables, references, contact information)? All information submitted must be contained within 3 pages for the Letter of Intent. LOIs should communicate the proposer s core objectives and plan. Please refer to page 9 of the Grand Challenge for specific elements to address. I submitted my response but did not receive a confirmation. How do I know if my LOI/Proposal went through? You will receive an confirmation when your application is submitted. If you do not receive this confirmation, please contact us at vaccineinnovations@gatesfoundation.org to confirm your application was received. Please first check your junk/spam folder to ensure the proposal confirmation was not accidentally sent there. Review and Selection What qualities are common to successful Grand Challenges proposals? Compelling proposals typically feature the following attributes: Tell a good story Focus on the problem at hand and how to solve it, rather than the past accomplishments of the team members Give an honest and thorough assessment of the risks involved and provide a plan to mitigate those risks State assumptions and provide rationale Identify success metrics, particularly those that indicate the project is on track far in advance of achieving the end goal Assemble the best team possible, which may mean reaching out beyond the typical circle of collaborators Will you give preference to proposals that plan to produce more than one of the priority diseases: human papillomavirus vaccine, inactivated poliovirus vaccine, measles-rubella vaccine, pentavalent vaccine (diphtheria, tetanus, whole-cell pertussis, hepatitis B, Hib), pneumococcal vaccine, and rotavirus vaccine? Priority will be given to those proposals that either validate with multiple priority vaccines or show clear rationale for extension to additional vaccines. Work with non-priority vaccines will also be considered for funding, but only in conjunction with work that includes a priority vaccine. 3 P a g e
4 How many applicants will be invited to submit a full proposal? How many will be awarded funding? The number of applicants invited to submit a full proposal and the number awarded funding will depend on the quality and diversity of proposals received. Who will be reviewing the responses? LOIs and full proposals will be reviewed by a broad range of experts, including but not limited to foundation staff and independent subject matter experts. Award How many grants are you willing to fund, and for how much? What budget range are you expecting to see in the proposal? The foundation will not provide specific funding limits or expectations. The number of awards and quantity of each award amount will depend upon the quality of the proposals received. Proposers are expected to develop a budget commensurate with the scope and scale of the work necessary to achieve the objectives laid out therein. What milestones or criteria must be met to trigger Phase 2 funding, and when will that decision be made? Applicants who are selected for funding will work closely with their foundation program officer to carefully define and agree upon milestones and timelines that must be met for Phase 2 funding. These milestones and timelines will be entirely customized to the objectives and workplan of the project(s). When will applicants be notified? Please see page 8 of the Grand Challenge for the planned review and announcement schedule When will funding commence? Should the foundation choose to move forward with an applicant after the full proposal review, due diligence and grant negotiations would begin in the second quarter of 2016 and will take approximately 1-3 months, after which final award decisions and payments would be made. Other Who can I contact if I have a question about a particular topic? 4 P a g e
5 Please note that we are unable to provide specific feedback on draft proposals, given the volume of applicants. All other questions should be directed to 5 P a g e
Grand Challenge: Innovations in Vaccine Manufacturing for Global Markets
 Grand Challenge: Innovations in Vaccine Manufacturing for Global Markets Contents The Grand Challenge... 2 Executive Summary... 2 Background... 2 Objectives... 3 Requirements... 5 Award... 6 A note on
Grand Challenge: Innovations in Vaccine Manufacturing for Global Markets Contents The Grand Challenge... 2 Executive Summary... 2 Background... 2 Objectives... 3 Requirements... 5 Award... 6 A note on
Grand Challenges Explorations Round 10 Rules & Guidelines Proposals due Wednesday, November 7, 2012, 11:30 a.m. U.S. Pacific Daylight Time
 Grand Challenges Explorations Round 10 Rules & Guidelines Proposals due Wednesday, November 7, 2012, 11:30 a.m. U.S. Pacific Daylight Time I. Overview Grand Challenges Explorations (GCE) supports hundreds
Grand Challenges Explorations Round 10 Rules & Guidelines Proposals due Wednesday, November 7, 2012, 11:30 a.m. U.S. Pacific Daylight Time I. Overview Grand Challenges Explorations (GCE) supports hundreds
Addition of vaccination against hepatitis B infection and change of the HPV vaccination programme
 Change to DENMARK S CHILDHOOD VACCINATION PROGRAMME 2014 Addition of vaccination against hepatitis B infection and change of the HPV vaccination programme 2014 Addition to the Danish Health and Medicines
Change to DENMARK S CHILDHOOD VACCINATION PROGRAMME 2014 Addition of vaccination against hepatitis B infection and change of the HPV vaccination programme 2014 Addition to the Danish Health and Medicines
SOLAR TRAINING AND EDUCATION FOR PROFESSIONALS FOA STEP@ee.doe.gov. FOA Webinar DE-FOA-0001329 6/04/2015
 SOLAR TRAINING AND EDUCATION FOR PROFESSIONALS FOA STEP@ee.doe.gov FOA Webinar DE-FOA-0001329 6/04/2015 1 Notice All applicants are strongly encouraged to carefully read the Funding Opportunity Announcement
SOLAR TRAINING AND EDUCATION FOR PROFESSIONALS FOA STEP@ee.doe.gov FOA Webinar DE-FOA-0001329 6/04/2015 1 Notice All applicants are strongly encouraged to carefully read the Funding Opportunity Announcement
Main Changes to the Vaccination Schedule Recommended by the Japan Pediatric Society January 12, 2014
 Main Changes to the Vaccination Schedule Recommended by the Japan Pediatric Society January, 04 ) 3-valent Pneumococcal conjugate vaccine (PCV3) The description of catch-up schedule for PCV3 has been revised
Main Changes to the Vaccination Schedule Recommended by the Japan Pediatric Society January, 04 ) 3-valent Pneumococcal conjugate vaccine (PCV3) The description of catch-up schedule for PCV3 has been revised
Substituting Thimerosal Preservative used in Vaccines: FDA perspective
 Substituting Thimerosal Preservative used in Vaccines: FDA perspective Robert Ball, MD, MPH, ScM FDA/CBER/OBE April 4, 2012 1 Objectives Requirement for preservative in multi-dose vaccine formulations
Substituting Thimerosal Preservative used in Vaccines: FDA perspective Robert Ball, MD, MPH, ScM FDA/CBER/OBE April 4, 2012 1 Objectives Requirement for preservative in multi-dose vaccine formulations
Immunisation. Immunisation
 Immunisation Immunisation Immunisation What is immunisation and why does my child need it? Immunisation is a simple, safe and effective way to protect children (and adults) from serious diseases. Immunisation
Immunisation Immunisation Immunisation What is immunisation and why does my child need it? Immunisation is a simple, safe and effective way to protect children (and adults) from serious diseases. Immunisation
Cultivating Healthy Communities 2016 Grant Program
 Cultivating Healthy Communities 2016 Grant Program www.aetnafoundation.org Aetna Foundation Request for Proposal The Aetna Foundation is the independent charitable and philanthropic arm of Aetna, Inc.
Cultivating Healthy Communities 2016 Grant Program www.aetnafoundation.org Aetna Foundation Request for Proposal The Aetna Foundation is the independent charitable and philanthropic arm of Aetna, Inc.
Discovery Grants. 2015-2016 Request for Applications
 The mission of the American Brain Tumor Association is to advance the understanding and treatment of brain tumors with the goals of improving, extending and, ultimately, saving the lives of those impacted
The mission of the American Brain Tumor Association is to advance the understanding and treatment of brain tumors with the goals of improving, extending and, ultimately, saving the lives of those impacted
BASIC ELEMENTS OF GRANT WRITING
 BASIC ELEMENTS OF GRANT WRITING The Corporation for Public Broadcasting evaluates hundreds of proposals each year for a variety of funding purposes. This publication is an easy guide to the basic elements
BASIC ELEMENTS OF GRANT WRITING The Corporation for Public Broadcasting evaluates hundreds of proposals each year for a variety of funding purposes. This publication is an easy guide to the basic elements
Grant Writing Dictionary
 Grant Writing Dictionary Abstract: A one-page description of your project: the purpose, the number of participants, the amount requested, and how the project is unique. Administrative Costs: Grant funds
Grant Writing Dictionary Abstract: A one-page description of your project: the purpose, the number of participants, the amount requested, and how the project is unique. Administrative Costs: Grant funds
REQUEST FOR PROPOSALS FOR A PROGRAM EVALUATOR TO EVALUATE LSC S MIDWEST LEGAL DISASTER COORDINATION PROJECT OCTOBER 28, 2015
 LEGAL SERVICES CORPORATION REQUEST FOR PROPOSALS FOR A PROGRAM EVALUATOR TO EVALUATE LSC S MIDWEST LEGAL DISASTER COORDINATION PROJECT OCTOBER 28, 2015 Page 1 of 8 INTRODUCTION The Legal Services Corporation
LEGAL SERVICES CORPORATION REQUEST FOR PROPOSALS FOR A PROGRAM EVALUATOR TO EVALUATE LSC S MIDWEST LEGAL DISASTER COORDINATION PROJECT OCTOBER 28, 2015 Page 1 of 8 INTRODUCTION The Legal Services Corporation
Re: Themed Inspections: Sale of Payment Protection Insurance ( PPI )
 27 June 2012 Re: Themed Inspections: Sale of Payment Protection Insurance ( PPI ) Dear I refer to previous correspondence and our recent meeting in relation to the above matter. As you are aware, the Central
27 June 2012 Re: Themed Inspections: Sale of Payment Protection Insurance ( PPI ) Dear I refer to previous correspondence and our recent meeting in relation to the above matter. As you are aware, the Central
Pertussis Vaccination Schedules Summary and recommendations
 Pertussis Vaccination Schedules Summary and recommendations Claire-Anne Siegrist Chair of the Pertussis Working Group SAGE Meeting 14-16 April 2015 FOR DECISION Review the evidence in support/against different
Pertussis Vaccination Schedules Summary and recommendations Claire-Anne Siegrist Chair of the Pertussis Working Group SAGE Meeting 14-16 April 2015 FOR DECISION Review the evidence in support/against different
Project Tracking and Cost Estimation Tool Member Impacting Project Overview
 Project Tracking and Cost Estimation Tool Member Impacting Project Overview Page 1 of 5 Table of Contents PR20120007 Project Tracking and Cost Estimation Table of Contents... 2 Version Control... 2 Executive
Project Tracking and Cost Estimation Tool Member Impacting Project Overview Page 1 of 5 Table of Contents PR20120007 Project Tracking and Cost Estimation Table of Contents... 2 Version Control... 2 Executive
http://www.ilga.gov/commission/jcar/admincode/077/077006650b0240...
 1 of 5 7/30/2014 9:47 AM TITLE 77: PUBLIC HEALTH CHAPTER I: DEPARTMENT OF PUBLIC HEALTH SUBCHAPTER i: MATERNAL AND CHILD HEALTH PART 665 CHILD HEALTH EXAMINATION CODE SECTION 665.240 BASIC IMMUNIZATION
1 of 5 7/30/2014 9:47 AM TITLE 77: PUBLIC HEALTH CHAPTER I: DEPARTMENT OF PUBLIC HEALTH SUBCHAPTER i: MATERNAL AND CHILD HEALTH PART 665 CHILD HEALTH EXAMINATION CODE SECTION 665.240 BASIC IMMUNIZATION
J D R F R E Q U E S T S E X P R E S S I O N S O F I N T E R E S T F O R : C O M B I N AT I O N T H E R AP I E S I N T Y P E 1 D I A B E T E S
 J D R F R E Q U E S T S E X P R E S S I O N S O F I N T E R E S T F O R : C O M B I N AT I O N T H E R AP I E S I N T Y P E 1 D I A B E T E S PURPOSE JDRF, the world s leading non-profit organization with
J D R F R E Q U E S T S E X P R E S S I O N S O F I N T E R E S T F O R : C O M B I N AT I O N T H E R AP I E S I N T Y P E 1 D I A B E T E S PURPOSE JDRF, the world s leading non-profit organization with
7. Website Information Architecture (IA)
 7. Website Information Architecture (IA) 7.1 Section Overview This section of the plan defines Website Information Architecture (IA) at EOEEA, as well as the key rationale and benefits of consolidating
7. Website Information Architecture (IA) 7.1 Section Overview This section of the plan defines Website Information Architecture (IA) at EOEEA, as well as the key rationale and benefits of consolidating
Frequently Asked Questions PROPOSED REVISIONS TO SCHOOL IMMUNIZATION REGULATIONS IN PENNSYLVANIA
 Frequently Asked Questions PROPOSED REVISIONS TO SCHOOL IMMUNIZATION REGULATIONS IN PENNSYLVANIA What is the current school vaccination rate in Pennsylvania? Given the way in which data is collected, we
Frequently Asked Questions PROPOSED REVISIONS TO SCHOOL IMMUNIZATION REGULATIONS IN PENNSYLVANIA What is the current school vaccination rate in Pennsylvania? Given the way in which data is collected, we
Master Service Agreement Extended Level of Support for Journals
 Master Service Agreement Extended Level of Support for Journals This document represents a Master Service Agreement ( MSA ) between the Center for Digital Research and Scholarship ( CDRS ) and [JOURNAL
Master Service Agreement Extended Level of Support for Journals This document represents a Master Service Agreement ( MSA ) between the Center for Digital Research and Scholarship ( CDRS ) and [JOURNAL
Free to Breathe 2015 Research Grant To Prevent or Stop Lung Cancer Metastasis. Request for Proposals
 Free to Breathe 2015 Research Grant To Prevent or Stop Lung Cancer Metastasis Request for Proposals Award Overview Free to Breathe has a new funding opportunity solely focused on research to prevent or
Free to Breathe 2015 Research Grant To Prevent or Stop Lung Cancer Metastasis Request for Proposals Award Overview Free to Breathe has a new funding opportunity solely focused on research to prevent or
Template K Implementation Requirements Instructions for RFP Response RFP #
 Template K Implementation Requirements Instructions for RFP Response Table of Contents 1.0 Project Management Approach... 3 1.1 Program and Project Management... 3 1.2 Change Management Plan... 3 1.3 Relationship
Template K Implementation Requirements Instructions for RFP Response Table of Contents 1.0 Project Management Approach... 3 1.1 Program and Project Management... 3 1.2 Change Management Plan... 3 1.3 Relationship
Childhood Immunization Status (CIS)
 Childhood Immunization Status (CIS) Description The percentage of children 2 years of age who had four diphtheria, tetanus and acellular pertussis (DTaP); three polio (IPV); one measles, mumps and rubella
Childhood Immunization Status (CIS) Description The percentage of children 2 years of age who had four diphtheria, tetanus and acellular pertussis (DTaP); three polio (IPV); one measles, mumps and rubella
Vaccine Development for Developing Countries Regulatory Approach in the European Union
 Vaccine Development for Developing Countries Regulatory Approach in the European Union Dr. Manfred G. Haase, Langen, Germany ECI Vaccine Technology II, June 1-6, 2008, Albufeira, Portugal 1 Regulation
Vaccine Development for Developing Countries Regulatory Approach in the European Union Dr. Manfred G. Haase, Langen, Germany ECI Vaccine Technology II, June 1-6, 2008, Albufeira, Portugal 1 Regulation
Overview of Grant Terms for The Leona M. and Harry B. Helmsley Charitable Trust
 Grant Terms Overview Last Overview of Grant Terms for The Leona M. and Harry B. Helmsley Charitable Trust First, congratulations on being invited to submit a grant application to the Helmsley Trust. As
Grant Terms Overview Last Overview of Grant Terms for The Leona M. and Harry B. Helmsley Charitable Trust First, congratulations on being invited to submit a grant application to the Helmsley Trust. As
Chapter 16. Competitive Negotiation: Negotiations
 Chapter 16. Competitive Negotiation: Negotiations Summary This chapter establishes the procedure for conducting negotiations with vendors selected by the selection committee following evaluation of the
Chapter 16. Competitive Negotiation: Negotiations Summary This chapter establishes the procedure for conducting negotiations with vendors selected by the selection committee following evaluation of the
Small Business Investment Companies Early Stage SBICs. SUMMARY: This call for proposals ( Call ) invites experienced early stage fund managers to
 This document is scheduled to be published in the Federal Register on 01/12/2015 and available online at http://federalregister.gov/a/2015-00247, and on FDsys.gov Billing Code: 8025-01 SMALL BUSINESS ADMINISTRATION
This document is scheduled to be published in the Federal Register on 01/12/2015 and available online at http://federalregister.gov/a/2015-00247, and on FDsys.gov Billing Code: 8025-01 SMALL BUSINESS ADMINISTRATION
Fiscal Year 2013 (FY13) Prostate Cancer Research Program (PCRP) Reference Table of Award Mechanisms and Submission Requirements
 Fiscal Year 2013 (FY13) Prostate Cancer Research Program (PCRP) Reference Table of Award Mechanisms and Submission Requirements PCRP AWARD MECHANISMS WITH EMPHASIS ON RESOURCES Clinical Consortium Award
Fiscal Year 2013 (FY13) Prostate Cancer Research Program (PCRP) Reference Table of Award Mechanisms and Submission Requirements PCRP AWARD MECHANISMS WITH EMPHASIS ON RESOURCES Clinical Consortium Award
Mrs. H.L. (Ruth Ray Hunt Memorial Fund Grant Cycle Spring 2014 Online Application Tips and FAQs
 ABOUT THE COMMON GRANT APPLICATION In an effort to respond to requests from local nonprofit organizations, Dallas Women s Foundation has joined a group of funders in North Texas and developed the Common
ABOUT THE COMMON GRANT APPLICATION In an effort to respond to requests from local nonprofit organizations, Dallas Women s Foundation has joined a group of funders in North Texas and developed the Common
Workforce Information Technology Procurement Project Executive Steering Committee Conference Call Minutes
 Workforce Information Technology Procurement Project Executive Steering Committee Conference Call Minutes Date: January 24, 2014 Steering Committee Members Attending: Members Absent from Meeting: Other
Workforce Information Technology Procurement Project Executive Steering Committee Conference Call Minutes Date: January 24, 2014 Steering Committee Members Attending: Members Absent from Meeting: Other
Strategic Wealth 360 Student FAQ
 Strategic Wealth 360 Student FAQ Key Features and Introduction to the SW360 Can you provide me with a snapshot of the various components of the SW360? Once enrolled in the SW360 - please complete the following
Strategic Wealth 360 Student FAQ Key Features and Introduction to the SW360 Can you provide me with a snapshot of the various components of the SW360? Once enrolled in the SW360 - please complete the following
Modifying the childhood immunisation schedule
 Modifying the childhood immunisation schedule With thanks to Dr. Marilyn Lansley for allowing adaptation and use of her presentations Vaccine Advice for CliniCians Service Objectives Understand the flexibility
Modifying the childhood immunisation schedule With thanks to Dr. Marilyn Lansley for allowing adaptation and use of her presentations Vaccine Advice for CliniCians Service Objectives Understand the flexibility
10 Steps for Managing Sub-Grants
 New Partners Initiative 10 Steps for Managing Sub-Grants DRAFT Table of Contents ACRONYMS AED PEPFAR NPI USG Introduction T he Academy for Educational Development (AED) provides technical support for grantees
New Partners Initiative 10 Steps for Managing Sub-Grants DRAFT Table of Contents ACRONYMS AED PEPFAR NPI USG Introduction T he Academy for Educational Development (AED) provides technical support for grantees
PA 14-01: CIRM Accelerated Development Pathway
 PA 14-01: CIRM Accelerated Development Pathway I. Purpose Through the Disease Team (Disease Team Research or Therapy Development) and Strategic Partnership Award programs, the California Institute for
PA 14-01: CIRM Accelerated Development Pathway I. Purpose Through the Disease Team (Disease Team Research or Therapy Development) and Strategic Partnership Award programs, the California Institute for
A Blue Horse Initiative
 A Blue Horse Initiative THE HEALTHY MINDS COOPERATIVE REQUEST FOR PROPOSALS (RFP) ACCOUNTING SOFTWARE AND SERVICES ISSUE DATE: December 2, 2014 CLOSING DATE: December 18, 2014, 4:00 pm (A.S.T.) CONTACT
A Blue Horse Initiative THE HEALTHY MINDS COOPERATIVE REQUEST FOR PROPOSALS (RFP) ACCOUNTING SOFTWARE AND SERVICES ISSUE DATE: December 2, 2014 CLOSING DATE: December 18, 2014, 4:00 pm (A.S.T.) CONTACT
LEGAL SERVICES CORPORATION REQUEST FOR PROPOSAL FOR DEVELOPMENT OF E-LEARNING TRAINING COURSE
 LEGAL SERVICES CORPORATION REQUEST FOR PROPOSAL FOR DEVELOPMENT OF E-LEARNING TRAINING COURSE OCTOBER 14, 2015 INTRODUCTION Legal Services Corporation (LSC) invites your company to submit a proposal for
LEGAL SERVICES CORPORATION REQUEST FOR PROPOSAL FOR DEVELOPMENT OF E-LEARNING TRAINING COURSE OCTOBER 14, 2015 INTRODUCTION Legal Services Corporation (LSC) invites your company to submit a proposal for
APPENDIX EE VACCINE STATUS AND DATE
 VACCINE STATUS AND DATE Vaccine Status, Date is a ten-character field which presents information about each of the vaccines required for children. For most cases, the first character tells the vaccine
VACCINE STATUS AND DATE Vaccine Status, Date is a ten-character field which presents information about each of the vaccines required for children. For most cases, the first character tells the vaccine
Vertex Investigator-Initiated Studies Program Overview
 Vertex Investigator-Initiated Studies Program Overview Our Goal To support independent, investigator-initiated research designed to advance scientific knowledge of disease states, patient populations,
Vertex Investigator-Initiated Studies Program Overview Our Goal To support independent, investigator-initiated research designed to advance scientific knowledge of disease states, patient populations,
Proposed Vaccine Information Materials for HPV (Human. AGENCY: Centers for Disease Control and Prevention (CDC),
 This document is scheduled to be published in the Federal Register on 10/22/2015 and available online at http://federalregister.gov/a/2015-26868, and on FDsys.gov BILLING CODE: 4163-18-P DEPARTMENT OF
This document is scheduled to be published in the Federal Register on 10/22/2015 and available online at http://federalregister.gov/a/2015-26868, and on FDsys.gov BILLING CODE: 4163-18-P DEPARTMENT OF
IMMUNISATION & EXCLUSIONS FOR INFECTIOUS DISEASES & ACUTELY ILL CHILDREN & STAFF POLICY
 IMMUNISATION & EXCLUSIONS FOR INFECTIOUS DISEASES & ACUTELY ILL CHILDREN & STAFF POLICY POLICY SILVERDALE CHILD CARE CENTRE has a duty of care to ensure that all persons are provided with a high level
IMMUNISATION & EXCLUSIONS FOR INFECTIOUS DISEASES & ACUTELY ILL CHILDREN & STAFF POLICY POLICY SILVERDALE CHILD CARE CENTRE has a duty of care to ensure that all persons are provided with a high level
Guided Pathways to Success in STEM Careers. Request for Proposals
 Guided Pathways to Success in STEM Careers Request for Proposals June 2013 Table of Contents Table of Contents... 2 Introduction... 3 Principles and Practices of Guided Pathways to Success... 4 Complete
Guided Pathways to Success in STEM Careers Request for Proposals June 2013 Table of Contents Table of Contents... 2 Introduction... 3 Principles and Practices of Guided Pathways to Success... 4 Complete
Solarize Dummerston RFP
 Solarize Dummerston is requesting proposals from Installer(s) willing to provide pricing for a group purchase rate of photovoltaic (PV) systems in conjunction with the Solarize Dummerston Program in Dummerston
Solarize Dummerston is requesting proposals from Installer(s) willing to provide pricing for a group purchase rate of photovoltaic (PV) systems in conjunction with the Solarize Dummerston Program in Dummerston
Duke University Medical Center Library and Archives. Project Planning Template. Initial Date of Assignment/Charge to Work Group:
 Duke University Medical Center Library and Archives Project Planning Template Project Title Initial Date of Assignment/Charge to Work Group: Last Major Revision to this Plan : Part 1: General Scope of
Duke University Medical Center Library and Archives Project Planning Template Project Title Initial Date of Assignment/Charge to Work Group: Last Major Revision to this Plan : Part 1: General Scope of
The Importance of Following the PROTOCOL in Clinical Trials
 The Importance of Following the PROTOCOL in Clinical Trials Presentation Objectives: Upon completion of this presentation, participants will be able to: Describe the following terms: Protocol, Protocol
The Importance of Following the PROTOCOL in Clinical Trials Presentation Objectives: Upon completion of this presentation, participants will be able to: Describe the following terms: Protocol, Protocol
RE AP QUE PLIC FOR IONS. Compa CPRIT RFA C 12 FORM 2. Company. p.1/17
 RE AP QUE ST PLIC AT RFA C 12 FORM 2 FOR IONS Compa any Formation Awards FY 2012 Fiscal Year Award Period September 1, 2011 Augustt 31, 2012 CPRIT RFA C 12 FORM 2 (Rev 6/ /30/11) Company Formation Awards
RE AP QUE ST PLIC AT RFA C 12 FORM 2 FOR IONS Compa any Formation Awards FY 2012 Fiscal Year Award Period September 1, 2011 Augustt 31, 2012 CPRIT RFA C 12 FORM 2 (Rev 6/ /30/11) Company Formation Awards
Proposal Negotiation. a guide. Constantine Vaitsas, vaitsas@help-forward.gr
 Proposal Negotiation a guide Constantine Vaitsas, vaitsas@help-forward.gr Presentation Outline Proposal Time Scale Negotiation Process Legal Negotiations Financial Negotiations Technical Negotiations NEF
Proposal Negotiation a guide Constantine Vaitsas, vaitsas@help-forward.gr Presentation Outline Proposal Time Scale Negotiation Process Legal Negotiations Financial Negotiations Technical Negotiations NEF
WellCare Clinical Practice Guidance - A Review
 Copyright 2008 by the American Academy of Pediatrics. Please see the following link for additional information concerning the AAP recommendations. http://brightfutures.aap.org/ Reference: American Academy
Copyright 2008 by the American Academy of Pediatrics. Please see the following link for additional information concerning the AAP recommendations. http://brightfutures.aap.org/ Reference: American Academy
https://www.fbo.gov/index?s=opportunity&mode=form&id=682b18787a8886f6ce36a118...
 Page 1 of 6 Clinical Studies Monitoring Service (CSMS) National Center for Complementary and Integrative Health (NCCIH) Solicitation Number: HHSNI-NCISBSSTSB5701304 Agency: Department of Health and Human
Page 1 of 6 Clinical Studies Monitoring Service (CSMS) National Center for Complementary and Integrative Health (NCCIH) Solicitation Number: HHSNI-NCISBSSTSB5701304 Agency: Department of Health and Human
Step by Step Project Planning
 Step by Step Project Planning Contents Introduction The Planning Process 1 Create a Project Plan...1 Create a Resource Plan...1 Create a Financial Plan...1 Create a Quality Plan...2 Create a Risk Plan...2
Step by Step Project Planning Contents Introduction The Planning Process 1 Create a Project Plan...1 Create a Resource Plan...1 Create a Financial Plan...1 Create a Quality Plan...2 Create a Risk Plan...2
Vaccine Information Statements
 Vaccine Information Statements IMPORTANT: By Federal law, all vaccine providers must give patients, or their parents or legal representatives, the appropriate Vaccine Information Statement (VIS) whenever
Vaccine Information Statements IMPORTANT: By Federal law, all vaccine providers must give patients, or their parents or legal representatives, the appropriate Vaccine Information Statement (VIS) whenever
Keiretsu 2 s K 2 Velocity Program Program Overview
 Great The Association Keiretsu with Acceleration CFOsGo Management Industry Phone: 415-860- 33 K@keiretsuforum.com Keiretsu s K Velocity Program Mission: Keiretsu s mission is To Radically Accelerate A
Great The Association Keiretsu with Acceleration CFOsGo Management Industry Phone: 415-860- 33 K@keiretsuforum.com Keiretsu s K Velocity Program Mission: Keiretsu s mission is To Radically Accelerate A
Regulations of Connecticut State Agencies. R.C.S.A. 10-204a-1 10-204a-4 CONTENTS. Procedures for reporting immunization data
 Agency Subject School Immunizations Requirements Inclusive Sections 10-204a-1 10-204a-4 Sec. 10-204a-1. Sec. 10-204a-2. Sec. 10-204a-2a. Sec. 10-204a-3. Sec. 10-204a-3a. Sec. 10-204a-4. CONTENTS Definitions
Agency Subject School Immunizations Requirements Inclusive Sections 10-204a-1 10-204a-4 Sec. 10-204a-1. Sec. 10-204a-2. Sec. 10-204a-2a. Sec. 10-204a-3. Sec. 10-204a-3a. Sec. 10-204a-4. CONTENTS Definitions
Successful Grant Writing
 Mathematical Association of America Successful Grant Writing www.maa.org/programs/grantwriting Revised February 2009 Preliminaries Determine your reasons for writing a proposal. Identify the problem. Research
Mathematical Association of America Successful Grant Writing www.maa.org/programs/grantwriting Revised February 2009 Preliminaries Determine your reasons for writing a proposal. Identify the problem. Research
Controlling Our Critical Path: A CDOT Guide to Better Project Management Practices
 : A CDOT Guide to Better Project Management Practices Project Management practices have been a part of the CDOT culture for many years. However, with a transitioning workforce and increasing demands, it
: A CDOT Guide to Better Project Management Practices Project Management practices have been a part of the CDOT culture for many years. However, with a transitioning workforce and increasing demands, it
Table of Contents. Grant Types. Who Can Apply? Timeline Application - Section by Section. Recipient Commitments. Review Process. Best Practices & FAQ
 2016 GRANT WORKSHOP Table of Contents Grant Types Who Can Apply? Timeline Application - Section by Section Recipient Commitments Review Process Best Practices & FAQ Types of Grants Available Kateri Brow
2016 GRANT WORKSHOP Table of Contents Grant Types Who Can Apply? Timeline Application - Section by Section Recipient Commitments Review Process Best Practices & FAQ Types of Grants Available Kateri Brow
Computing Services Network Project Methodology
 Computing Services Network Project Prepared By: Todd Brindley, CSN Project Version # 1.0 Updated on 09/15/2008 Version 1.0 Page 1 MANAGEMENT PLANNING Project : Version Control Version Date Author Change
Computing Services Network Project Prepared By: Todd Brindley, CSN Project Version # 1.0 Updated on 09/15/2008 Version 1.0 Page 1 MANAGEMENT PLANNING Project : Version Control Version Date Author Change
CLINICAL TRIALS SHOULD YOU PARTICIPATE? by Gwen L. Nichols, MD
 CLINICAL TRIALS SHOULD YOU PARTICIPATE? by Gwen L. Nichols, MD Gwen L. Nichols, M.D., is currently the Oncology Site Head of the Roche Translational Clinical Research Center at Hoffman- LaRoche. In this
CLINICAL TRIALS SHOULD YOU PARTICIPATE? by Gwen L. Nichols, MD Gwen L. Nichols, M.D., is currently the Oncology Site Head of the Roche Translational Clinical Research Center at Hoffman- LaRoche. In this
Project Scorecard Template
 Project Scorecard Template 1. Identify criteria for success: Review the objectives and deliverables in the Project Definition, as well as any other existing information that is relevant to the project.
Project Scorecard Template 1. Identify criteria for success: Review the objectives and deliverables in the Project Definition, as well as any other existing information that is relevant to the project.
GAVI FULL COUNTRY EVALUATION 2013 PROCESS EVALUATION OF PNEUMOCOCCAL VACCINE INTRODUCTION
 GAVI FULL COUNTRY EVALUATION 2013 PROCESS EVALUATION OF PNEUMOCOCCAL VACCINE INTRODUCTION ALLIANCE RESPONSE JULY 2014 The GAVI Alliance is a learning organisation; we take the need for independent evaluation
GAVI FULL COUNTRY EVALUATION 2013 PROCESS EVALUATION OF PNEUMOCOCCAL VACCINE INTRODUCTION ALLIANCE RESPONSE JULY 2014 The GAVI Alliance is a learning organisation; we take the need for independent evaluation
The Ireland Funds Guidance on Preparing a Grant Proposal
 The Ireland Funds Guidance on Preparing a Grant Proposal Funding by The Ireland Funds The Ireland Funds give to charitable organisations in two ways: the annual Small Grants Round and donor advised gifts.
The Ireland Funds Guidance on Preparing a Grant Proposal Funding by The Ireland Funds The Ireland Funds give to charitable organisations in two ways: the annual Small Grants Round and donor advised gifts.
TERM SHEET EXAMPLE. 1 P age
 1 P age TERM SHEET EXAMPLE BIOTECHCO Overview & Business Strategy BIOTECHCO (the licensor), located in North Dakota, has a proprietary technology called ZIP that can generate fully human antibodies with
1 P age TERM SHEET EXAMPLE BIOTECHCO Overview & Business Strategy BIOTECHCO (the licensor), located in North Dakota, has a proprietary technology called ZIP that can generate fully human antibodies with
Pricing for Profit with Project Management & Process Improvement
 Pricing for Profit with Project Management & Process Improvement Presented by Timothy B. Corcoran FM03 10/2/2015 10:30 AM - 12:00 PM The handouts and presentations attached are copyright and trademark
Pricing for Profit with Project Management & Process Improvement Presented by Timothy B. Corcoran FM03 10/2/2015 10:30 AM - 12:00 PM The handouts and presentations attached are copyright and trademark
Missouri Technology Corporation Early-Stage Business Grant Program Fiscal Year 2015 Request for Proposals
 Missouri Technology Corporation Early-Stage Business Grant Program Fiscal Year 2015 Request for Proposals Key Dates: RFP Release Date June 4, 2015 Written Questions (Preliminary Submissions) July 7, 2015
Missouri Technology Corporation Early-Stage Business Grant Program Fiscal Year 2015 Request for Proposals Key Dates: RFP Release Date June 4, 2015 Written Questions (Preliminary Submissions) July 7, 2015
DARPA-BAA-16-42. Frequently Asked Questions
 Last Updated: 7/5/2016 REMINDER GENERAL INFORMATION DARPA-BAA-16-42 Frequently Asked Questions Q: If my research is relevant in this field, but is not geared specifically to meet these goals, is there
Last Updated: 7/5/2016 REMINDER GENERAL INFORMATION DARPA-BAA-16-42 Frequently Asked Questions Q: If my research is relevant in this field, but is not geared specifically to meet these goals, is there
Securing Polio s Legacy. Transition Planning. Frequently Asked Questions
 Securing Polio s Legacy Transition Planning March 2015 Frequently Asked Questions Context In May 2013, the 66 th World Health Assembly endorsed the Polio Eradication and Endgame Strategic Plan 2013-2018.
Securing Polio s Legacy Transition Planning March 2015 Frequently Asked Questions Context In May 2013, the 66 th World Health Assembly endorsed the Polio Eradication and Endgame Strategic Plan 2013-2018.
Cancer Clinical Trials. Canadian Dream Team Supplementary Funding
 SU2C Canada-OICR Cancer Clinical Trials Canadian Dream Team Supplementary Funding Call for Ideas and Full Application Guidelines 9861 Leslie St Richmond Hill, ON L4B 3Y4 416-797-5366; 905-883-6800 (FAX)
SU2C Canada-OICR Cancer Clinical Trials Canadian Dream Team Supplementary Funding Call for Ideas and Full Application Guidelines 9861 Leslie St Richmond Hill, ON L4B 3Y4 416-797-5366; 905-883-6800 (FAX)
OPERATING PRINCIPLES. Strengthening Nonprofit Organizations. Approaching Grants as Investments. Leveraging Resources
 OPERATING PRINCIPLES Fidelity Investments Chairman Edward C. Johnson 3d and his father, the founder of the company, established the Fidelity Foundation, a private, non-operating foundation, in 1965 with
OPERATING PRINCIPLES Fidelity Investments Chairman Edward C. Johnson 3d and his father, the founder of the company, established the Fidelity Foundation, a private, non-operating foundation, in 1965 with
Guidance for Industry
 Guidance for Industry Expanded Access to Investigational Drugs for Treatment Use Qs & As DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding
Guidance for Industry Expanded Access to Investigational Drugs for Treatment Use Qs & As DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding
RFP for Case Writing 2013
 RFP for Case Writing 2013 PMI India is pleased to announce its 2013 Request for Proposals (RFP) for Case Writing funding on topics related to project, program, and portfolio management (PPPM). PMI India
RFP for Case Writing 2013 PMI India is pleased to announce its 2013 Request for Proposals (RFP) for Case Writing funding on topics related to project, program, and portfolio management (PPPM). PMI India
How To Apply For A Cybersecurity Investment Fund
 REQUEST FOR APPLICATIONS (RFA) Cybersecurity Investment Fund (CIF) Purpose: The Cybersecurity Investment Fund (CIF) exists to support seed/early-stage Maryland companies in their effort to develop and
REQUEST FOR APPLICATIONS (RFA) Cybersecurity Investment Fund (CIF) Purpose: The Cybersecurity Investment Fund (CIF) exists to support seed/early-stage Maryland companies in their effort to develop and
Crystal Miller, MPA Assistant Director Sponsored Programs Administration. March 26 & 27, 2009 April 3, 2009
 Crystal Miller, MPA Assistant Director Sponsored Programs Administration March 26 & 27, 2009 April 3, 2009 NIH Funding Current Announcements Timelines and Process Challenge Grants Other NIH Stimulus Opportunities
Crystal Miller, MPA Assistant Director Sponsored Programs Administration March 26 & 27, 2009 April 3, 2009 NIH Funding Current Announcements Timelines and Process Challenge Grants Other NIH Stimulus Opportunities
DL Course Development Process (New Course)
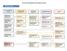 DL Course Development Process (New Course) Preliminary Stage Design Work with Deans to Determine Course Development Needs Work with Deans to select SME and establish a Contact the Design regarding development
DL Course Development Process (New Course) Preliminary Stage Design Work with Deans to Determine Course Development Needs Work with Deans to select SME and establish a Contact the Design regarding development
BCS HIGHER EDUCATION QUALIFICATIONS - GUIDANCE NOTES FOR THE PROFESSIONAL PROJECT IN IT
 BCS HIGHER EDUCATION QUALIFICATIONS - GUIDANCE NOTES FOR THE PROFESSIONAL PROJECT IN IT These guidance notes should be read in conjunction with the General Notes for Guidance and the Professional Project
BCS HIGHER EDUCATION QUALIFICATIONS - GUIDANCE NOTES FOR THE PROFESSIONAL PROJECT IN IT These guidance notes should be read in conjunction with the General Notes for Guidance and the Professional Project
Expanded Programme on Immunization
 Expanded Programme on Immunization Expanded Programme on Immunization has been delivered the immunization services to the targeted children of under one year old child and pregnant women. Currently total
Expanded Programme on Immunization Expanded Programme on Immunization has been delivered the immunization services to the targeted children of under one year old child and pregnant women. Currently total
VISION 2017 Coding and Robotics Grants 2016. Grant Guidelines
 VISION 2017 Coding and Robotics Grants 2016 Overview of Coding and Robotics Grants program Reflecting the themes outlined in The Next Horizon: Vision 2017 for Public Libraries, funding of $200,000 is now
VISION 2017 Coding and Robotics Grants 2016 Overview of Coding and Robotics Grants program Reflecting the themes outlined in The Next Horizon: Vision 2017 for Public Libraries, funding of $200,000 is now
PERFORMANCE IN INITIATING CLINICAL RESEARCH (PI) CLINICAL TRIALS DATA RECORD ADJUSTMENT PROCESS
 PERFORMANCE IN INITIATING CLINICAL RESEARCH (PI) CLINICAL TRIALS DATA RECORD ADJUSTMENT PROCESS Version Control Amended small error in Acronyms table (page 2) 1 Acronyms CTP FPR MHRA REC SOP TMB VRA Clinical
PERFORMANCE IN INITIATING CLINICAL RESEARCH (PI) CLINICAL TRIALS DATA RECORD ADJUSTMENT PROCESS Version Control Amended small error in Acronyms table (page 2) 1 Acronyms CTP FPR MHRA REC SOP TMB VRA Clinical
Frequently Asked Questions for the Pediatric Immunization Administration Codes
 Frequently Asked Questions for the Pediatric Immunization Administration Codes Component Definition Old versus New IA Codes Counseling and OQHCP Documentation Requirements When to still report 90471-90474
Frequently Asked Questions for the Pediatric Immunization Administration Codes Component Definition Old versus New IA Codes Counseling and OQHCP Documentation Requirements When to still report 90471-90474
Guidance for Outline Stage BMC: DPFS Applicants
 Guidance for Outline Stage BMC: DPFS Applicants Contents Page Oversight and Reporting... 2 Project Management... 2 Intellectual Property Management and Exploitation... 2 How to apply... 3 Outline Applications...
Guidance for Outline Stage BMC: DPFS Applicants Contents Page Oversight and Reporting... 2 Project Management... 2 Intellectual Property Management and Exploitation... 2 How to apply... 3 Outline Applications...
California Networks for EHR Adoption
 California Networks for EHR Adoption Request for Proposals: Phase I - Planning A program jointly sponsored by: Community Clinics Initiative A joint program of Tides and The California Endowment California
California Networks for EHR Adoption Request for Proposals: Phase I - Planning A program jointly sponsored by: Community Clinics Initiative A joint program of Tides and The California Endowment California
In those instances, the National Vaccine Injury Compensation Program (VICP) allows individuals to file a petition for compensation.
 Data & Statistics The United States has the safest, most effective vaccine supply in history. In the majority of cases, vaccines cause no side effects, however they can occur, as with any medication but
Data & Statistics The United States has the safest, most effective vaccine supply in history. In the majority of cases, vaccines cause no side effects, however they can occur, as with any medication but
Community Shares Practitioner Training and Licensing
 Community Shares Practitioner Training and Licensing January 2016 Contents Introduction... 4 Becoming a licensed practitioner... 4 Other materials... 5 Skills Specification... 6 Functional analysis...
Community Shares Practitioner Training and Licensing January 2016 Contents Introduction... 4 Becoming a licensed practitioner... 4 Other materials... 5 Skills Specification... 6 Functional analysis...
https://syracuseuniversity.qualtrics.com/controlpanel/ajax.php?action=getsurveyprintpre...
 Page 1 of 7 SU - UMU Pilot Grant Program Driving Inspiration and Innovation through Collaboration Syracuse University- Upstate Medical University 2015 Pilot Grant Application Responding to society s most
Page 1 of 7 SU - UMU Pilot Grant Program Driving Inspiration and Innovation through Collaboration Syracuse University- Upstate Medical University 2015 Pilot Grant Application Responding to society s most
The Technology Transfer Process
 The Technology Transfer Process The Offices of Technology Management The Offices of Technology Management are responsible for managing the intellectual property generated by research and educational activities
The Technology Transfer Process The Offices of Technology Management The Offices of Technology Management are responsible for managing the intellectual property generated by research and educational activities
TIPS & TRICKS FOR USA JOBS. By Junell L. Norris Human Resource Office U.S. Geological Survey
 TIPS & TRICKS FOR USA JOBS By Junell L. Norris Human Resource Office U.S. Geological Survey OVERVIEW Creating a User Account Creating Your Resume Creating Job Search Agents & Saving Jobs Reviewing Application
TIPS & TRICKS FOR USA JOBS By Junell L. Norris Human Resource Office U.S. Geological Survey OVERVIEW Creating a User Account Creating Your Resume Creating Job Search Agents & Saving Jobs Reviewing Application
Contract Notification
 Department of Education and Training Higher Education and Skills Contract Notification CN No. 2016-02 Date: 18 January 2016 To: Purpose: Scope: Subject: Chief Executive Officer or equivalent Information
Department of Education and Training Higher Education and Skills Contract Notification CN No. 2016-02 Date: 18 January 2016 To: Purpose: Scope: Subject: Chief Executive Officer or equivalent Information
Request for Proposals. Site C Clean Energy Project. Worker Accommodation RFP #1807
 RFP #1807 Issued SUMMARY OF KEY INFORMATION Page i RFP TITLE CONTACT PERSON The title of this RFP is: RFP #1807 Worker Accommodation Proponents should use this title on all correspondence. The Contact
RFP #1807 Issued SUMMARY OF KEY INFORMATION Page i RFP TITLE CONTACT PERSON The title of this RFP is: RFP #1807 Worker Accommodation Proponents should use this title on all correspondence. The Contact
Request for Proposals
 OICR Health Services Research Program Knowledge Translation Research Network Request for Proposals Funding Partners: OICR Knowledge Translation Research Network (KT Net) OnePage Letter of Intent (LOI)
OICR Health Services Research Program Knowledge Translation Research Network Request for Proposals Funding Partners: OICR Knowledge Translation Research Network (KT Net) OnePage Letter of Intent (LOI)
HEALTHCARE DEVELOPMENT CENTER
 HEALTHCARE DEVELOPMENT CENTER Partnership Guide Hanover Research s Health Care Development Center (HDC) was founded in response to overwhelming demand from Hanover Research clients for assistance identifying
HEALTHCARE DEVELOPMENT CENTER Partnership Guide Hanover Research s Health Care Development Center (HDC) was founded in response to overwhelming demand from Hanover Research clients for assistance identifying
An Introduction to Sustainability Reporting. What Is Sustainability Reporting. White Paper: An Introduction to Sustainability Reporting
 An Introduction to Sustainability Reporting There is a growing movement, worldwide, to not only be a more responsible corporate citizen, but to trade on that fact and Sustainability Reporting is the lynchpin
An Introduction to Sustainability Reporting There is a growing movement, worldwide, to not only be a more responsible corporate citizen, but to trade on that fact and Sustainability Reporting is the lynchpin
Contents. Project Management: August 2005 Page 2 Clinical Research Operational Standards for Professional Practice for Professional Practice
 Project Management: August 2005 Contents Introduction........................................................... 4 About this standard: what is the importance of project management?................. 4
Project Management: August 2005 Contents Introduction........................................................... 4 About this standard: what is the importance of project management?................. 4
A Framework for Project Metrics
 A Framework for Project Metrics Deciding what to measure and how to measure it August 13, 2007 Dr. Gary J. Evans, PMP CVR/IT Consulting LLC www.cvr-it.com Welcome! Focus of this workshop: Project Metrics
A Framework for Project Metrics Deciding what to measure and how to measure it August 13, 2007 Dr. Gary J. Evans, PMP CVR/IT Consulting LLC www.cvr-it.com Welcome! Focus of this workshop: Project Metrics
The PCORI Application Process
 The PCORI Application Process James Hulbert Pre- Award Manager February 19, 2015 Welcome! James Hulbert Pre-Award Manager 2 Session Objectives List and define each sec>on of the PCORI applica>on State the
The PCORI Application Process James Hulbert Pre- Award Manager February 19, 2015 Welcome! James Hulbert Pre-Award Manager 2 Session Objectives List and define each sec>on of the PCORI applica>on State the
Illinois IKE Disaster Recovery Program Housing Rehabilitation and Reconstruction Program (IKE-HRR) FAQS
 Illinois IKE Disaster Recovery Program Housing Rehabilitation and Reconstruction Program (IKE-HRR) FAQS Existing Damage In reference to the requirement for $10,000 in damages to the home, does that mean
Illinois IKE Disaster Recovery Program Housing Rehabilitation and Reconstruction Program (IKE-HRR) FAQS Existing Damage In reference to the requirement for $10,000 in damages to the home, does that mean
Business Retention, Expansion & Investment Attraction Project. Request for Proposal (RFP) FDI Strategy, Marketing Plan & Collateral
 Business Retention, Expansion & Investment Attraction Project Request for Proposal (RFP) FDI Strategy, Marketing Plan & Collateral December 16, 2013 Town of Sylvan Lake Administration 5012-48 th Avenue
Business Retention, Expansion & Investment Attraction Project Request for Proposal (RFP) FDI Strategy, Marketing Plan & Collateral December 16, 2013 Town of Sylvan Lake Administration 5012-48 th Avenue
Health Insurance Rate Review Grant Program Cycle I Quarterly Report
 Submission Date: February 25, 2011 State: Project Title: Mississippi MS Health Insurance Rate Review Program Project Quarter Reporting Period: Quarter 1 (08/09/2010-12/31/2010) Grant Project Director:
Submission Date: February 25, 2011 State: Project Title: Mississippi MS Health Insurance Rate Review Program Project Quarter Reporting Period: Quarter 1 (08/09/2010-12/31/2010) Grant Project Director:
Reporting Service Performance Information
 AASB Exposure Draft ED 270 August 2015 Reporting Service Performance Information Comments to the AASB by 12 February 2016 PLEASE NOTE THIS DATE HAS BEEN EXTENDED TO 29 APRIL 2016 How to comment on this
AASB Exposure Draft ED 270 August 2015 Reporting Service Performance Information Comments to the AASB by 12 February 2016 PLEASE NOTE THIS DATE HAS BEEN EXTENDED TO 29 APRIL 2016 How to comment on this
iillli United States Department of Agriculture
 USDA iillli United States Department of Agriculture Food and Nutrition Service Park Office Center 3101 Park Center Drive Alexandria VA 22302 MAY 1 3 2014 SUBJECT: To: Supplemental Nutrition Assistance
USDA iillli United States Department of Agriculture Food and Nutrition Service Park Office Center 3101 Park Center Drive Alexandria VA 22302 MAY 1 3 2014 SUBJECT: To: Supplemental Nutrition Assistance
POLICY AND PROCEDURES OFFICE OF NEW DRUGS. Good Review Practice: Management of Breakthrough Therapy-Designated Drugs and Biologics.
 POLICY AND PROCEDURES OFFICE OF NEW DRUGS Good Review Practice: Management of Breakthrough Therapy-Designated Drugs and Biologics Table of Contents PURPOSE...1 BACKGROUND...2 POLICY...3 ROLES AND RESPONSIBILITIES...4
POLICY AND PROCEDURES OFFICE OF NEW DRUGS Good Review Practice: Management of Breakthrough Therapy-Designated Drugs and Biologics Table of Contents PURPOSE...1 BACKGROUND...2 POLICY...3 ROLES AND RESPONSIBILITIES...4
Crucell Operational Excellence: Healthy Ambition
 Crucell Operational Excellence: Healthy Ambition Cees de Jong Chief Operating Officer March 12 th, 2008 London Agenda Introduction COO s perspective on Crucell Marketed products Excellent scope for growth
Crucell Operational Excellence: Healthy Ambition Cees de Jong Chief Operating Officer March 12 th, 2008 London Agenda Introduction COO s perspective on Crucell Marketed products Excellent scope for growth
