Botulinum toxins in the treatment of migraine and tension-type headaches
|
|
|
- Adam Brown
- 8 years ago
- Views:
Transcription
1 Phys Med Rehabil Clin N Am 14 (2003) Botulinum toxins in the treatment of migraine and tension-type headaches Paul Winner, DO, FAAN a,b,* a Palm Beach Headache Center, Premiere Research Institute, 5205 Greeenwood Avenue, Suite 200, West Palm Beach, FL 33407, USA b Department of Neurology, Nova Southeastern University, 3301 College Avenue, Ft. Lauderdale, FL 33314, USA Patients who do not respond completely to the acute treatment of migraine or tension-type headaches both episodic and chronic with associated disability should be considered for preventive therapy. Preventive therapy must be tailored to the individual patient s expectations and medical situation. Two recent additions to the armamentarium of preventive medications are botulinum toxin type A (Botox) and botulium toxin B (Moybloc). This article discusses the mechanism of action of both botulinum toxin type A (BTX-A) and botulium toxin type B (BTX-B). It is felt by some that these medications have an antinociceptive mechanism that acts on the muscle spindles or through some direct central nervous system effect [1]; the role played by muscle relaxation is unclear. BTX-A has been used for more than a decade for the treatment of cervical dystonia and spasticity, and has recently received approval for cosmetic use from the US Food and Drug Administration (FDA) [2]. BTX-B has recently become available for clinical use. Botulinum toxins are administered for the treatment of headache in a variety of doses and locations; however, they have a key advantage as a single administration, reportedly resulting in a reduction of the impact of headache in some individuals for 3 to 4 months [3]. At present, three methods of administration of botulinum toxins are used: (1) a fixed-site approach; (2) follow the pain injecting directly into tender muscles; and (3) a combination of these methods. Pertinent literature on the use of BTX-A and BTX-B as a preventive therapy for migraine, tension-type headaches, and chronic headaches is reviewed. * Palm Beach Headache Center, Premiere Research Institute, 5205 Greenwood Avenue, Suite 200, West Palm Beach, FL /03/$ see front matter Ó 2003 Elsevier Inc. All rights reserved. doi: /s (03)
2 886 P. Winner / Phys Med Rehabil Clin N Am 14 (2003) Introduction Millions of lives are disrupted by disability associated with episodic migraine, chronic migraine, and chronic tension-type headaches [4]. The economic burden of missed work days and impaired function due to migraine alone is estimated at $13 billion, and the additional direct and indirect medical costs are estimated to be over $1 billion [5]. Patients who do not respond completely to the acute treatment of their episodic attacks, as well as those with chronic migraine, chronic tension-type headaches, and mixed daily headache, should be placed on preventive therapy. At present, only a small percentage of patients with migraine are receiving preventive therapy [6]. Many of the preventive (prophylactic) therapies used today have limited scientific evidence to support their effectiveness. FDA-approved medications for the preventive treatment of migraine include divalproex sodium (Depakote/Depakote ER), timolol (Blocadren), propranolol (Inderal), and methysergide (Sansert) (Table 1) [7]. Scientific evidence that will help guide treatment choices in the future is accumulating; however, clinicians must draw on present experience with many classes of medications used in the preventive treatment of headache. Selecting the most appropriate class of preventive headache medication (eg, anticonvulsants, antidepressants, beta blockers, calcium channel blockers, nonsteroidal anti-inflammatories, and other agents, including botulinum toxins) may require addressing the presence of comorbid or coexisting medical issues in an individual patient. The preventive therapeutic needs of patients must be tailored to their expectations and medical status; in addition, potential medication overuse must not be overlooked. BTX-A purified neurotoxin complex is produced from fermentation of Hall strain Clostridium botulinum type A. BTX-A blocks neuromuscular transmission by binding to acceptor sites on motor neuron terminals and inhibiting the release of acetylcholine. This results in a partial chemical denervation of the muscle, which in turn results in a localized reduction in muscle activity, and may have additional, direct antinociceptive effects [8]. The exact mechanism of action in the relief of moderate to severe headache is not fully understood. BTX-A has been approved by the FDA for: treatment of patients with cervical dystonia to reduce the severity of abnormal head position and neck pain associated with this disorder; treatment of strabismus and blepharo- Table 1 FDA-approved medication for preventive treatment of migraine Drug Year of approval Divalproex sodium (Depakote ER) 2000 Divalproex sodium (Depakote) 1996 Timolol (Blocadren) 1990 Propranolol (Inderal) 1979 Methysergide (Sansert) 1962
3 P. Winner / Phys Med Rehabil Clin N Am 14 (2003) spasm; and cosmetic use for temporary improvement in the appearance of moderate to severe glabellar lines associated with corrugator or procerus muscle activity [8]. Many other potential clinical uses for botulinum toxins have and are being explored (Box 1). BTX-A is supplied in a single vial containing 100 U of vacuum-dried Clostridium botulinum type A neurotoxin complex. BTX-A should be stored in a freezer before use and should be administered within 4 hours after the vial is removed from the freezer and reconstituted. The product diluent does not contain a preservative; thus, once reconstructed, it should be placed in a refrigerator until administered during a 4-hour period. BTX- A is reconstituted with sterile, nonpreserved saline before intramuscular injection. A 25-, 27-, or 30-gauge needle may be used for superficial muscles. Clinical improvement generally begins within the first 2 weeks after injection with maximum clinical benefit at approximately 6 weeks and a 3-month duration of effect [8]. BTX-B is a sterile, injectable liquid formulation produced by fermentation of the bacterium Clostridium botulinum type B. BTX-B has been demonstrated to cleave synaptic vesicle-associated membrane protein, a component of the protein complex responsible for docking and fusion of the vesicle with a synaptic membrane. It also inhibits the release of acetylcholine into the synapse [9]. BTX-B has been approved by the FDA for the treatment of patients with cervical dystonia to reduce the severity of abnormal head position and neck pain associated with this disorder. It is a sterile, injectable solution supplied in individual 3.5-mm glass vials, and it is available in three presentations: 2500 U, 5000 U, and 10,000 U. BTX-B should be stored in a refrigerator for up to 21 months; it should not be frozen or shaken. It may be diluted with normal saline, but once diluted it is to be used within 4 hours. The formulation does not contain a preservative. BTX-B can be diluted up to sixfold and still maintain its structural integrity. Patients who use doses of 5,000 U to 10,000 U report a duration of effect of 12 to 16 weeks [9]. Common adverse events associated with BTX-B use include dry mouth, dysphagia, and injection site pain. Botulinum toxins for the treatment of migraine Binder et al [10] noted that patients who were treated for glabellar lines with pericranial BTX-A injections reported improvement in their migraine. A randomized, double-blind, placebo-controlled study of 123 patients with a history of two to eight moderate to severe attacks of migraine per month was conducted [3]. The study arm, using 25 U of BTX-A injected pericranially, reported a significant reduction in the frequency and severity of attacks compared with placebo; curiously, the 75-U arm did not. Brin et al [11] reported a randomized, double-blind, placebo-controlled trial of 53
4 888 P. Winner / Phys Med Rehabil Clin N Am 14 (2003) Box 1. Potential uses for botulinum toxins The clinical applications of botulinum toxins are varied and vast. Much of the available data are anecdotal; however, it is important to consider the potential role of botulinum toxins in many disorders related to overactive muscle conditions. Some of the potential uses are shown. Movement disorders Idiopathic focal dystonias (blepharospasm) Craniocervical disorders (torticollis and isolated head tremor, blepharospasm, oromandibular dystonia, lingual dystonia, laryngeal dystonia) Other focal dystonias (writer s cramp, occupational cramps such as musician s cramp) Tardive dystonia Hemifacial spasm/postfacial nerve palsy synkinesis Tic disorders (simple tics, Tourette s syndrome, dystonic tics) Tremor (essential, primary writing, palatal, cerebellar), jaw Painful spinal myoclonus Parkinson s disease (freezing of gait, off period dystonia, severe constipation) Cephalic tetanus, stiff man syndrome, neuromyotonia Muscle stiffness, cramps, spasms Apralela of eyelid opening Spasticity Multiple sclerosis Stroke Traumatic brain injury Cerebral palsy (drooling) Spinal cord injury Neuromuscular disorders Myokymia Neurogenic tibialis anterior hypertrophy with myalgia Benign cramp-fasciculation syndrome Pain Headache (tension type, migraine) Backache (neck, lower back) Myofascial pain Tennis elbow Trigeminal neuralgia Nerve compression syndromes (piriformis syndrome, pronator syndrome)
5 P. Winner / Phys Med Rehabil Clin N Am 14 (2003) Ear, nose, and throat disorders Oromandibular disorders (bruxism, masseter hypertrophy, temporomandibular joint dysfunction) Pharyngeal disorders (cricopharyngeal dysphagia, closure of larynx in chronic aspiration) Larynegeal disorders (vocal fold granuloma, ventricular dysphonia, mutational dysphonia) Stuttering with glottal blocks Vocal tics (coprolalia, sialorrhea) Disorders of pelvic floor Anismus Vaginismus Anal fissures Detrusor-sphincter dyssynergia Overactive bladder Cosmetic applications Wrinkles, frown lines (hyperfunctional facial lines) Rejuvenation of aging neck Hyperhidrosis Opthalmological disorders Concomitant or noncomitant misalignment Primary or secondary esotropia or exotropia Paralytic strabismus (III, IV, VI nerve palsy, internuclear ophthalmoplegia, skew deviation) Duane s syndrome Restrictive or myogenic strabismus Disorders of ocular motility (nystagmus and oscillopsia) Thyroid disease (upper eyelid retraction, glabellar furrowing) Therapeutic ptosis for corneal protection patients who were randomized to one of four injection groups. Only the subjects receiving 45 U in the frontal region and 30 U in the temporal region demonstrated a significant reduction in migraine severity at 12 weeks compared with placebo. Barrientos and Chana [12] reported a double-blind, placebo-controlled study for migraine prophylaxis (two to eight migraines per month at baseline) in which patients were given a total dose of 50 U BTX-A at six injection sites: the temporalis, frontalis, trapezius, and splenius regions (10 U at each site), the glabella (8 U), and the procerus (2 U) (Table 2). The patients were followed for 90 days and demonstrated significant reduction in the frequency of migraine attacks per month at 30, 60, and 90 days (P \ 0.02, P \ 0.02, and P \ 0.01, respectively) compared
6 890 P. Winner / Phys Med Rehabil Clin N Am 14 (2003) Table 2 Dose distribution Injection site Number of sites BTX-A or placebo (total units) Temporalis 2 10 Frontalis 4 10 Glabellar 4 8 Procerus 1 2 Trapezius 2 10 Splenium capitis 2 10 with baseline. Also of note is that the number of severe migraine attacks was significantly reduced for the BTX-A group at days 30, 60, and 90 compared with baseline. The duration of migraine attacks was significantly reduced for the BTX-A group when compared with the baseline at days 30, 60, and 90 (P \ 0.05, P \ 0.02, and P \ 0.01, respectively). The patients taking BTX- A demonstrated a reduction in migraine frequency, severity, and duration compared with placebo after 90 days of treatment; thus the duration of effect observed with BTX-A treatment is approximately 3 months. Acute migraine treated with nonsteroidals and triptans also demonstrated significantly reduced frequency for the BTX-A group at all assessment points after 60 and 90 days. This may ultimately help to decrease the risk of analgesic overuse. BTX-A therapy was reported as safe and well tolerated. Eross and Dodick [13] examined the effect of BTX-A on reducing disability associated with episodic and chronic migraine. Patients with migraine in an open label study received BTX-A and were followed for 3 months. Migraine disability assessment scale (MIDAS) scores were recorded at baseline and 3 months after BTX-A administration. Information regarding the number of headache days and the average pain of each headache was obtained. All treated patients were monitored for change in disability, number of headache days, and severity; subtypes of patients with episodic migraine and chronic migraine were analyzed. Twenty-five patients with migraine, 12 patients with episodic migraine, and 36 patients with chronic migraine received BTX-A. All patients received at least 25 U of BTX-A divided into the frontalis (1.2 U at six sites), temporalis (6.25 U at two sites), procerus (2.5 U at one site), and corrugator muscles (1.25 at two sites). An additional 25 U to 75 U were injected into the cervical paraspinals based on clinical discretion (Table 3). Table 3 Responses stratified by amount of BTX-A injected BTX-A (units) Patients (n) Responders % % % %
7 P. Winner / Phys Med Rehabil Clin N Am 14 (2003) BTX-A decreased migraine disability in Eross and Dodick s study [13]. Of those patients reporting a greater than 50% improvement in disability, the average decrease in disability was 79%; average decreases in headache frequency and severity were 61% and 27%, respectively. Disability responses based on the amount of BTX-A administered did not appear to differ among all patients. Response to BTX-A treatment did not differ significantly based on underlying muscle tenderness; 62% of those with prominent muscle tenderness responded to therapy, compared with 56% who reported no muscle tenderness. Age was noted to have an effect on response. BTX-A was less effective in those patients over 61 years of age; only 20% of the eight patients in this age group responded, compared with 71% of eight patients younger than age 30 and 75% of patients aged 31 to 40 years. For the 12 patients aged 41 to 50 years, 50% responded, as did 63% of the 12 patients aged 51 to 60 years. Further studies addressing subsets of various age groups are needed to further address this issue. Relja et al [14] set out to measure the effect of BTX-A on the use of triptans in migraine patients. Thirty-one patients with moderate to severe migraine were randomized to BTX-A or placebo for the treatment of their migraine in a double-blind, placebo-controlled trial. Disability was measured using a MIDAS with entrance criteria requiring grades III or IV (moderate to severe disability). Patients received a total of 100 U of BTX-A in the active group in the frontalis, corrugator, temporalis, semispinalis, and trapezius muscles. Two treatments were administered at 3-month intervals. Clinical evaluation for efficacy and medication usage for 6 months before and 6 months after the first treatment were analyzed. Rates of headache response and rates of pain-free periods were assessed 2 to 4 hours after acute medication usage in all patients. In addition, medication use and headache-free days were calculated during the study. The impact of migraine on normal daily activities 2 to 4 hours after an attack was reduced in a significantly higher proportion of patients treated with BTX-A compared with those receiving placebo. Requirement for additional medication within 24 hours was also significantly reduced in patients receiving BTX-A. The total triptan usage of the BTX-A treatment group demonstrated a statistically significant reduction in usage compared with the placebo group. Although the total number of headache-free days did not change significantly, the pain characteristics of the BTX-A treated patients had changed to nonpulsating, moderate pain that was not aggravated by movement; this resulted in the patient having more tolerable headache symptoms. The BTX-A treatment group demonstrated a decrease in acute medication usage and an improved quality of life. Lake and Saper [15] evaluated the efficacy of BTX-B for the prevention of migraine in an open-label study. Twenty-one adult patients who met the International Headache Society (IHS) criteria for migraine reporting from at least 4 to 10 migraines per month were treated using 5000 U of BTX-B. The injection sites included five in the frontalis, two in the temporal region,
8 892 P. Winner / Phys Med Rehabil Clin N Am 14 (2003) two in the occipital region, two in the suboccipital region, and two in the trapezius. Patients were evaluated at baseline, monthly postbaseline, or 4 months posttreatment. The primary end point was the headache index (frequency average intensity duration days). The mean headache index dropped from 6.7 preinjection to 3.7 at 1 month, 3.3 at 2 months, 3.3 at 3 months, and 3.4 at 4 months following treatment with BTX-B (P \ ). The mean percentage improvement over 4 months was 47.5%. The mean headache frequency declined from 7.7 preinjection to 5.2 at 1 month, 5.0 at 2 months, 4.9 at 3 months, and 4.6 at 4 months (P \ ). Individuals differed significantly in onset, magnitude, and duration of improvement. The presence of cervical pain with migraine was not associated with differential improvement. Significant improvement also occurred in the visual analog scale of headache, sleep, and treatment satisfaction. MIDAS scores dropped from a mean of 29 days with lost or significantly reduced functioning during the 3 months preinjection to 12 lost or impaired days during the 3 months postinjection. Temporary adverse events included eyelid weakness, dry eyes, neck pain, tingling, facial bruise, neck weakness, difficulty controlling facial muscles, and dizziness. BTX-B proved to be generally well tolerated and efficacious in the study population. Mathew [16] reported long-term experience with the use of BTX-A, multitreatment cycles in chronic migraine patients noting significant impact on migraine, reduced disability, and decreased acute medication usage. Patients who met the criteria for chronic (transformed) migraine with or without analgesic overuse were studied. Indications for BTX-A were: lack of improvement with preventive pharmacotherapy; severe and intolerable adverse events from preventive therapy; patient refusal to use daily medications; contraindications for acute migraine therapy; and elderly patient with chronic migraine. One hundred twelve patients were treated in this open label study with BTX-A using 50 to 100 U in multiple scalp and neck sites using a combination of follow the pain and fixed-site approaches, thus individualizing the approach. Approximately half of the patients received a single treatment, while some patients received up to six treatments. The outcome measures were: MIDAS scale, frequency of headache, and the amount of acute medications used. The mean MIDAS score before the first treatment was ; after 3 months this score had improved to (P \ 0.001). After multiple treatments, continued improvement in disability was reported. Severe episodes of disabling migraine were reduced dramatically compared with low-grade headaches. The mean number of headache days decreased significantly after treatment cycles one, two, and three. Acute medication intake showed significant reduction, and emergency room visits and hospitalizations were dramatically reduced. No serious symptoms secondary to treatment with BTX-A were reported. Those patients who received 100 U of BTX-A responded better than those who received 50 U. Patients who received anterior and posterior injections appeared to have done better than those receiving only
9 P. Winner / Phys Med Rehabil Clin N Am 14 (2003) anterior injections. This study supports BTX-A at 100 U divided between anterior and posterior injection sites as well as consideration of multiple injection treatments to help achieve the best response. It also reports that repeated injections did not seem to produce tachyphylaxis. Botulinum toxins for the treatment of chronic tension type headache A randomized placebo-controlled study of 60 chronic tension-type patients with BTX-A administered in the frontal and temporal muscles did not demonstrate a significant reduction in the pain intensity, number of pain-free days, or medication usages compared with the placebo group [17]. However, only 20 U of BTX-A was used in the study, so this may have been a subtherapeutic dose. Rollnik et al [18] reported a placebo-controlled study of chronic tensiontype headache patients that also failed to demonstrate any clinically significant changes between the two groups. It has been noted that reduction in muscle activity has not resulted in relief of chronic tension-type symptoms. Further study of the efficacy of botulinum toxins in the treatment of chronic tension-type headache using a double-blind placebo-controlled study of 40 patients fulfilled the IHS criteria for chronic tension-type headache did not demonstrate significance [19]. These patients were randomized to receive 1 U of botulinum toxin per kilogram to a maximum of 100 U versus placebo (saline). All patients with analgesic or caffeine abuse or more than one migraine per month were excluded. No reported important differences in baseline characteristics were noted. The primary outcome measure was average headache intensity at 12 weeks compared with baseline. Secondary end points included average number of headache days, headache hours per day, number of days in which symptomatic treatment was taken, and patients global assessment of improvement at 4, 8, and 12 weeks. The botulinum toxin treatment group did not statistically differ itself from the placebo group in the primary or secondary end points. The most commonly reported adverse event was pain on injection site [19]. Further study is needed to address the effect of BTX-A in the treatment of episodic tension and chronic tension-type headaches. The role of botulinum toxins in headache treatment As data continue to accumulate, the role of BTX-A and BTX-B as a preventive treatment for migraine and chronic daily headache (chronic migraine/transformed migraine) is gaining strong support [3,12 16]. Blumenfeld [20] evaluated the effectiveness of BTX-A for the preventive therapy in patients with varied headache types. This retrospective, openlabel study of 271 patients with varying types of headaches (episodic
10 894 P. Winner / Phys Med Rehabil Clin N Am 14 (2003) migraine, chronic daily headaches, episodic tension-type and mixed headaches), all of whom received an average of 63 U of BTX-A, is somewhat representative of clinical practice. Patients were injected using a fixed-site approach (for patients with migraine or migrainous features), a follow the pain approach (for patients with tension-type features), or a combination of the two (for patients with mixed features). Seventy-seven percent of patients were refractory to oral preventive therapy; 48% overused oral abortive medications. The average treatment period was 8.6 months with an average of 3.4 treatments, each given at 3-month intervals. Approximately 80% of patients reported a response to treatment defined as a reduction of headache frequency or intensity; 60.5% reported a good to excellent response of greater than 50%, 19.5% reported a response of 1% to 50%, and 19.9% reported no response. Overall BTX-A treatment resulted in a significant reduction in headache days per month from baseline in 56% of patients regardless of headache type. Headache intensity decreased 25% from baseline. There was no correlation of effect or lack of effect as related to reason for treatment, duration or number of treatments, mean or total dose, age, gender, comorbidities, and pre- or posttreatment headache frequency or intensity. Transient adverse events included: eyelid ptosis, eyebrow ptosis, and neck muscle weakness. BTX-A is efficacious for the preventive treatment of a variety of patients with headache refractory to oral preventive medicines as noted by the study. No patient type for success or failure was established in the study. Gwynn et al [21] evaluated the efficacy and safety of BTX-B in the treatment of chronic headache with tension or migrainous qualities. This randomized, double-blind, placebo-controlled study evaluated adults who had at least 3 months of near daily headaches of migraine, transformed migraine or tension- type history before enrollment. Patients were randomized to receive either 5000 U or 7500 U of BTX-B, or placebo (saline). Study medication was distributed evenly and bilaterally into 20 total injection sites, including frontal, temporal, and occipital-nuchal regions. The preliminary analysis reports a benefit of BTX-B treatment versus placebo. Tepper et al [22] evaluated the effect of BTX-A as a preventive treatment for patients with refractory headache. He studied patients who had failed at least four preventive medications. These patients were followed for at least 6 months after injection of BTX-A. The muscles injected included some or all of the following: the frontalis, temporalis, corrugator, procerus, occipitalis, semispinalis, splenius capitus, trapezius, paraspinals, and sternocleidomastoid. Patients were assessed for frequency of pain, intensity of pain, number of days with severe headache, headache index, number of pain-free days per month, and the MIDAS score. These end points were measured in the month before BTX-A injection, at baseline and monthly for 3 months following injection. One hundred patients were assessed with diagnostic criteria reported as chronic daily headache with analgesic rebound (65%), chronic
11 P. Winner / Phys Med Rehabil Clin N Am 14 (2003) daily headache without analgesic rebound (15%), episodic migraine (12%), and chronic posttraumatic headache (8%). The average number of hours with pain per month was 24. A significant reduction in headache index was obtained in 1 month (22 versus 40 at baseline), 2 months (21), and 3 months (21). A significant reduction in the number of days with severe pain was also obtained at 1 month (2.6 versus 4.9 at baseline), 2 months (2.7), and 3 months (2.6). A significant increase in the number of pain-free days per month was reached at 1 month (15 versus 8 at baseline), 2 months (15), and 3 months (15). The MIDAS score was reduced to 19 at 3 months versus 33 at baseline. BTX-A proved to be effective in the prevention of refractory headache patients. Troost and Rosenberg [23] evaluated the effectiveness of repeated BTX-A treatment in 436 refractory patients. This retrospective evaluation of patients with migraine and episodic tension-type headache including chronic daily headache was evaluated in an open-label study paradigm over a 3-year period. Patients were treated with intramuscular injection of BTX-A in the forehead, temples, neck, and shoulders according to their individual needs. The patients reporting frontal headaches were injected with 30 to 50 U of BTX-A in the procerus, frontalis, and temporalis muscles. For posterior headaches, the occipitalis and trapezius were injected on each side with 20 U and 35 U, respectively; when muscle tenderness or spasm was present, 40 U were injected into the rhomboids. At least one treatment of BTX-A was administered to each patient over a 3-year period. The majority of patients reported previously failing three or more preventive pharmacologic therapies. The average number of treatments administered to patients was 2.4 (range: 1 8). The overall mean dose of BTX-A was 127 U with a range of 25 to 300 U. Overall, 91% of patients reported improvement. No adverse events were reported across treatments other than minor, transient injection pain. This study demonstrated that patients with migraine, chronic daily headache, and episodic tension-type headache treated with BTX-A showed improvement that appeared to be cumulative through multiple treatments (mean: 2.4) and maintained for up to eight treatments. Each headache type was reportedly associated with similar improvements overall. Ondo et al [24] studied the effects of BTX-A in chronic daily headache patients using a randomized placebo-controlled parallel study design. This study is unique in that the BTX-A dosage was 200 U versus placebo. Patients with chronic daily headache who commonly suffer from tensiontype headaches and chronic migraine were included. Sixty patients, 19 with chronic migraine, 22 with chronic tension-type headaches, and 19 with a mixed chronic daily headache pattern were evaluated. Thirty patients were randomized to receive 200 U of BTX-A, which was administered according to the physician s discretion using a follow the pain protocol, and 30 received placebo (saline). Fifty-eight patients completed the trial at 12 weeks; 51 proceeded to the open-label BTX-A, completing the trial at 25 weeks. Patients treated with BTX-A had significantly fewer headache days from
12 896 P. Winner / Phys Med Rehabil Clin N Am 14 (2003) weeks 8 to 12 compared with placebo (P \ 0.05). Three times as many patients had a marked, dramatic improvement in global headache symptoms compared with placebo in the study. Patients who received two 200-U injections did better than those who received only one, suggesting a dose response. Miller and Denny [25] evaluated the effect of BTX-A in chronic headache patients who had failed prior therapy. In this retrospective chart analysis, 48 chronic headache patients received BTX-A at six sites: the frontalis, corrugator, temporalis, splenius, and capitus, with follow the pain injections administered to the cervical paraspinals, trapezius, and levator scapulae muscles as needed. Twenty-one patients received a single treatment of BTX-A, while 27 received two to eight treatments. The mean dose was 139 U, with a median being 100 U, for a single treatment ranging from 50 U to 300 U. For multiple treatments, the mean dose was 141 U, with the median being 100 U, with a range of 75 U to 400 U. The majority of patients with chronic tension-type headaches or migraine who had been unresponsive to prior preventive therapies had good, very good, or complete resolution of their headaches when treated with BTX-A under a combined fixed-site and follow the pain protocol. Those patients who received more than one treatment appeared to have a greater response with treatments lasting at least 3 months, regardless of the number of treatments. BTX-A treatments were found to be safe and effective in patients who had failed prior therapy. Many questions about the specific use of BTX-A and BTX-B for the treatment of headaches remain. For one, what types of patients should be considered for treatment? It would seem that patients with episodic and chronic migraine have demonstrated the most significant reduction in migraine frequency, severity, duration, and intake of acute medications over approximately a 3-month period. Furthermore, those with the most severe migraine seem to report the most prominent reductions. Additional research is needed to further determine the optimum dosage and location of injection for the various headache types (Fig. 1). The use of BTX-A and BTX-B in other primary headache types, such as cluster headache, also requires continued research. Large-scale, double-blind, placebo-controlled studies are needed to further advance our knowledge of the clinical usefulness of BTX-A and BTX-B as well as other botulinum toxin preparations in the management of headache patients. For headache practitioners, BTX-A and BTX-B are becoming valuable treatment additions for moderate to severe episodic migraine, chronic migraine, and chronic daily headache treatment patients. Summary Botulinum toxins are promising preventive treatments for patients with moderate to severe episodic and chronic migraine and chronic daily headache.
13 P. Winner / Phys Med Rehabil Clin N Am 14 (2003) Fig. 1. Potential botulinum toxin injection sites.
14 898 P. Winner / Phys Med Rehabil Clin N Am 14 (2003) The recommended indications for botulinum toxins as preventive therapy lend themselves to the following patient types: those who demonstrate a lack of improvement from preventive (prophylactic) pharmacotherapy; those who experience severe and intolerable adverse events from preventive medications; those who refuse to use daily medications; those who have contraindications to acute migraine therapy, and elderly patients with chronic migraine [16]. Both open-label and double-blind placebo-controlled studies using fixed-site, follow the pain, or a combination approach have demonstrated significant reduction in migraine frequency, severity, and duration, as well as decreased use of acute medications [12 16]. The most prominent reductions have been noted in those with reportedly the most severe migraine headaches [16]. Large, well-designed, double-blind, placebo-controlled studies are recommended to further clarify optimum dosage and location of injection, reduce treatment frequency and duration, and address other primary headache disorders that may benefit from this therapy. References [1] Silberstein SD. Review of botulinum toxin type A and its clinical applications in migraine headache. Expert Opin Pharmacother 2001;2: [2] Allergan s Botox cosmetic approval by the FDA for the treatment of glabellar lines. Available at: Accessed July 16, [3] Silberstein SD, Mathew N, Saper JS, et al. Botulinum toxin type A as a migraine preventive treatment. Headache 2000;40: [4] Stewart W, Lipton RB, Celentano DD, Reed ML. Prevalence of migraine headache in the United States: relation to age, income, race, and other sociodemographic factors. JAMA 1992;267:64 9. [5] Lipton RB, Stewart WF, Diamond S, Diamond M, Reed M. Prevalence and burden of migraine in the United States. Data from the American Migraine Study II. Headache 2001;41: [6] Stewart W, Shechter A, Rasmussen RK. Migraine prevalence. A review of populationbased studies. Neurology 1994;44(Suppl 14): [7] Winner P, Linder S, Wasiewski W. Pharmacologic treatment of headache: headache in children and adolescents. Hamilton (Ontario, Canada): BC Decker; [8] Thomson PDR. Botox (botulinum toxin type A) purified neurotoxin complex. In: Physicians desk reference. 57th edition. Montvale (NJ); p [9] Thomson PDR. Myobloc (botulinum toxin type B) injectable solution. In: PhysiciansÕ desk reference. 57th edition. Montvale (NJ); p [10] Binder WJ, Brin MF, Blitzer A, Pogoda JM. Botulinum toxin type A for treatment of migraine. Semin Cutan Med Surg 2001;20: [11] Brin MF, Swope DM, Abassi S, et al. Botulinum toxin type A for migraine: double-blind, placebo controlled region specific evaluation. Poster presented at the Headache World 2000, London, September 2000 (poster 1196). [12] Barrientos N, Chana P. Efficacy and safety of botulinum toxin type A in the prophylactic treatment of migraine. Presented at the American Headache Society 44th Annual Scientific Meeting, Seattle, Washington, June 21 23, [13] Eross EG, Dodick DW. The effects of botulinum toxin type A on disability in episodic and chronic miraine. Presented at the American Headache Society 44th Annual Scientific Meeting, Seattle, Washington, June 21 23, 2002 [abstract S108].
15 P. Winner / Phys Med Rehabil Clin N Am 14 (2003) [14] Relja M, Zagreb KN, et al. Botulinum toxin type A reduces acute medication (triptans) use in migraine patients. Presented at the American Academy of Neurology 55th Annual Meeting, Honolulu, Hawaii, April 2, 2003 (abstract PO4.147). [15] Lake A, Saper J. Botulinum toxin type B for migraine prophylaxis: a 4-month open-label prospective outcome study. Presented at the American Academy of Neurology 55th Annual Meeting, Honolulu, Hawaii, April 2, 2003 (abstract PO4.150). [16] Mathew N, Kallasam J, Kaupp A, et al. Disease modification in chronic migraine with botulinum toxin type A: long-term experience. Presented at the American Headache Society 44th Annual Scientific Meeting, Seattle, Washington, June 21 23, [17] Schmitt WJ, Fravin SE, et al. Effect of botulinum toxin A injections in the treatment of chronic tension-type headache: a double-blind, placebo-controlled trial. Headache 2001;41: [18] Rollnik JD, Karst M, Fink M, et al. Botulinum Toxin type A and EMG: a Key to the Understanding of Chronic Tension type headache? Headache 2001;41: [19] de Brujin SFTM, Padberg M, Tavy DLJ. Treatment of chronic tension-type headache with botulinum toxin: a double blind placebo-controlled clinical trial. Presented at the American Academy of Neurology 55th Annual Meeting, Honolulu, Hawaii, April 2, 2003 (abstract PO4.152). [20] Blumenfeld A. Botulinum toxin type A as an effective preventive treatment in headache. Presented at the American Academy of Neurology 55th Annual Meeting, Honolulu, Hawaii, April 2, 2003 (abstract PO4.152). [21] Gwynn MW, Baker T, English J. Botulinum toxin type A as an effective preventive treatment in headache. Presented at the American Academy of Neurology 55th Annual Meeting, Honolulu, Hawaii, April 2, 2003 (abstract PO4.149). [22] Tepper SJ, Bigal ME, Sheftell FD, Rapoport AM. Botulinum toxin A in the preventive treatment of refractory headache. Presented at the American Academy of Neurology 55th Annual Meeting, Honolulu, Hawaii, April 2, 2003 (abstract PO4.151). [23] Troost T, Rosenberg JR. Improvement in intractable headache with repeated botulinum toxin type A treatment. Presented at the American Academy of Neurology 55th Annual Meeting, Honolulu, Hawaii, April 2, 2003 (abstract PO4.153). [24] Ondo WG, Vuong KD, Derman HS. Botulinum toxin A (Botox) in a combination fixedinjection-site and follow the pain protocol. Presented at the American Headache Society 44th Annual Scientific Meeting, Seattle, Washington, June 21 23, 2002 (abstract S138). [25] Miller T, Denny L. Retrospective cohort analysis of 48 chronic headache patients treated with botulinum toxin type A (Botox) in a combination fixed injection site and follow the pain protocol. Presented at the American Headache Society 44th Annual Scientific Meeting, Seattle, Washington, June 21 23, 2002 (abstract S138).
PROCEEDINGS BOTULINUM TOXIN: PARADIGMS FOR TREATMENT * Benjamin M. Frishberg, MD ABSTRACT
 BOTULINUM TOIN: PARADIGMS FOR TREATMENT Benjamin M. Frishberg, MD ABSTRACT Botulinum toxin type A has been studied for the treatment of headache disorders since the early 1990s when its usefulness for
BOTULINUM TOIN: PARADIGMS FOR TREATMENT Benjamin M. Frishberg, MD ABSTRACT Botulinum toxin type A has been studied for the treatment of headache disorders since the early 1990s when its usefulness for
Botulinum neurotoxin for the treatment of migraine and other primary headache disorders
 Dermatol Clin 22 (2004) 167 175 Botulinum neurotoxin for the treatment of migraine and other primary headache disorders Andrew M. Blumenfeld, MD a, *, David W. Dodick, MD, FRCP(C), FACP b,c, Stephen D.
Dermatol Clin 22 (2004) 167 175 Botulinum neurotoxin for the treatment of migraine and other primary headache disorders Andrew M. Blumenfeld, MD a, *, David W. Dodick, MD, FRCP(C), FACP b,c, Stephen D.
BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required]
![BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required] BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required]](/thumbs/29/13417512.jpg) BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required] Medical Policy: MP-RX-01-11 Original Effective Date: March 24, 2011 Reviewed: Revised: This policy applies to products
BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required] Medical Policy: MP-RX-01-11 Original Effective Date: March 24, 2011 Reviewed: Revised: This policy applies to products
Treatment of Tension-Type Headache with Botox:
 Treatment of Tension-Type Headache with Botox: A Review of the Literature DIMA ROZEN, M.D. 1, AND JOYTI SHARMA, M.D. 2 Abstract Botulinum toxin has been shown to effectively treat several types of neurological
Treatment of Tension-Type Headache with Botox: A Review of the Literature DIMA ROZEN, M.D. 1, AND JOYTI SHARMA, M.D. 2 Abstract Botulinum toxin has been shown to effectively treat several types of neurological
Neurotoxins and headache: A review of the evidence. Alan G Finkel MD FAHS FAAN Carolina Headache Institute University of North Carolina
 Neurotoxins and headache: A review of the evidence Alan G Finkel MD FAHS FAAN Carolina Headache Institute University of North Carolina Disclosures Allergan: Trainer 2010 present Glaxo Smith Kline: Speaker
Neurotoxins and headache: A review of the evidence Alan G Finkel MD FAHS FAAN Carolina Headache Institute University of North Carolina Disclosures Allergan: Trainer 2010 present Glaxo Smith Kline: Speaker
The use of botulinum toxin is considered medically necessary for the FDA-labeled indications of:
 COVERAGE: The use of botulinum toxin is considered medically necessary for the FDA-labeled indications of: Strabismus, Blepharospasm, Facial nerve (VII) disorders, or Cervical dystonia. The use of botulinum
COVERAGE: The use of botulinum toxin is considered medically necessary for the FDA-labeled indications of: Strabismus, Blepharospasm, Facial nerve (VII) disorders, or Cervical dystonia. The use of botulinum
Botulinum toxin in the treatment of chronic migraine. Gregory P. Hanes, MD Neuroscience Summit 5/14/15
 Botulinum toxin in the treatment of chronic migraine Gregory P. Hanes, MD Neuroscience Summit 5/14/15 Primary Headache Disorders: Frequency Classification After Secondary Causes Are Ruled Out Short-Duration
Botulinum toxin in the treatment of chronic migraine Gregory P. Hanes, MD Neuroscience Summit 5/14/15 Primary Headache Disorders: Frequency Classification After Secondary Causes Are Ruled Out Short-Duration
Onabotulinumtoxin in the Preventive Treatment of Chronic Migraine and Other Headaches
 Onabotulinumtoxin in the Preventive Treatment of Chronic Migraine and Other Headaches While BoNTA shows promise in the management of chronic migraine, optimal outcomes will be acheived when clinicians
Onabotulinumtoxin in the Preventive Treatment of Chronic Migraine and Other Headaches While BoNTA shows promise in the management of chronic migraine, optimal outcomes will be acheived when clinicians
Botulinum Toxins A and B
 DRUG POLICY Botulinum Toxins A and B Policy Number: 2015D0017P Effective Date: 11/1/2015 Table of Contents Page Related Medical or Drug Policies: Temporomandibular Joint Disorders COVERAGE RATIONALE...
DRUG POLICY Botulinum Toxins A and B Policy Number: 2015D0017P Effective Date: 11/1/2015 Table of Contents Page Related Medical or Drug Policies: Temporomandibular Joint Disorders COVERAGE RATIONALE...
BOTOX Treatment. for Chronic Migraine. Information for patients and their families. Botulinum Toxin Type A
 BOTOX Treatment Botulinum Toxin Type A for Chronic Migraine Information for patients and their families. Is Chronic Migraine the same as Migraine? Chronic Migraine is similar to migraine as sufferers experience
BOTOX Treatment Botulinum Toxin Type A for Chronic Migraine Information for patients and their families. Is Chronic Migraine the same as Migraine? Chronic Migraine is similar to migraine as sufferers experience
Coverage Criteria: Express Scripts, Inc. monograph dated 03/19/2010
 BENEFIT DESCRIPTION AND LIMITATIONS OF COVERAGE ITEM: PRODUCT LINES: COVERED UNDER: DESCRIPTION: CPT/HCPCS Code: Company Supplying: Setting: Botox (onabotulinumtoxina, injection) Commercial HMO/PPO/CDHP
BENEFIT DESCRIPTION AND LIMITATIONS OF COVERAGE ITEM: PRODUCT LINES: COVERED UNDER: DESCRIPTION: CPT/HCPCS Code: Company Supplying: Setting: Botox (onabotulinumtoxina, injection) Commercial HMO/PPO/CDHP
CONSUMER MEDICINE INFORMATION
 CONSUMER MEDICINE INFORMATION BOTOX (botulinum toxin, type A) purified neurotoxin complex The information in this leaflet is ONLY a summary and is not a complete statement about BOTOX injection. Your doctor
CONSUMER MEDICINE INFORMATION BOTOX (botulinum toxin, type A) purified neurotoxin complex The information in this leaflet is ONLY a summary and is not a complete statement about BOTOX injection. Your doctor
Will Botox help your migraines?
 Will Botox help your migraines? In case you hadn t heard, Botox can now be used and potentially claimed on insurance for migraines. Surprising I know. You re probably wondering how will an injection of
Will Botox help your migraines? In case you hadn t heard, Botox can now be used and potentially claimed on insurance for migraines. Surprising I know. You re probably wondering how will an injection of
WAT ALS HOOFDPIJN CHRONISCH WORDT? Prof Jan Versijpt, MD PhD Kliniek voor Hoofd- en Aangezichtspijn Neurologie, UZ Brussel
 WAT ALS HOOFDPIJN CHRONISCH WORDT? Prof Jan Versijpt, MD PhD Kliniek voor Hoofd- en Aangezichtspijn Neurologie, UZ Brussel Stovner, IHC 2009 New daily persistent headache Primair = dagelijkse and continue
WAT ALS HOOFDPIJN CHRONISCH WORDT? Prof Jan Versijpt, MD PhD Kliniek voor Hoofd- en Aangezichtspijn Neurologie, UZ Brussel Stovner, IHC 2009 New daily persistent headache Primair = dagelijkse and continue
Botulinum Toxins. Policy # 00012 Original Effective Date: 01/28/2003 Current Effective Date: 02/19/2014
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
How Is OnabotulinumtoxinA Reimbursed For Chronic Migraine? Impact Of FDA Approval And The New CPT Code
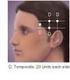 How Is OnabotulinumtoxinA Reimbursed For Chronic Migraine? Impact Of FDA Approval And The New CPT Code Effective January 1, 2013, physicians will be able to report the new CPT code 64615 when performing
How Is OnabotulinumtoxinA Reimbursed For Chronic Migraine? Impact Of FDA Approval And The New CPT Code Effective January 1, 2013, physicians will be able to report the new CPT code 64615 when performing
Address all correspondence to A. Blumenfeld, Headache Center, The Headache Center of Southern CA, 320 Santa Fe Drive, Encinitas, CA 92024, USA.
 Headache 2010 American Headache Society ISSN 0017-8748 doi: 10.1111/j.1526-4610.2010.01766.x Published by Wiley Periodicals, Inc. Research Submissions Method of Injection of OnabotulinumtoxinA for Chronic
Headache 2010 American Headache Society ISSN 0017-8748 doi: 10.1111/j.1526-4610.2010.01766.x Published by Wiley Periodicals, Inc. Research Submissions Method of Injection of OnabotulinumtoxinA for Chronic
Botulinum Toxins (BOTOX, DYSPORT, MYOBLOC and XEOMIN)
 Botulinum Toxins (BOTOX, DYSPORT, MYOBLOC and XEOMIN) Policy Number: Original Effective Date: MM.04.004 03/14/2006 Line(s) of Business: Current Effective Date: HMO; PPO 10/01/2012 Section: Prescription
Botulinum Toxins (BOTOX, DYSPORT, MYOBLOC and XEOMIN) Policy Number: Original Effective Date: MM.04.004 03/14/2006 Line(s) of Business: Current Effective Date: HMO; PPO 10/01/2012 Section: Prescription
Treatment of migraine headache with botulinum toxin type A
 Facial Plast Surg Clin N Am 11 (2003) 465 475 Treatment of migraine headache with botulinum toxin type A William J. Binder, MD, FACS a,b, *, Andrew Blitzer, DDS, MD, FACS c a Department of Head and Neck
Facial Plast Surg Clin N Am 11 (2003) 465 475 Treatment of migraine headache with botulinum toxin type A William J. Binder, MD, FACS a,b, *, Andrew Blitzer, DDS, MD, FACS c a Department of Head and Neck
Billing and Coding Guidelines
 Billing and Coding Guidelines Contractor Name Wisconsin Physicians Service Insurance Corporation Title INJ- 018 Botulinum Toxin Type A & Type B Revision Effective Date 10/01/2011 AMA CPT/ ADA CDT Copyright
Billing and Coding Guidelines Contractor Name Wisconsin Physicians Service Insurance Corporation Title INJ- 018 Botulinum Toxin Type A & Type B Revision Effective Date 10/01/2011 AMA CPT/ ADA CDT Copyright
Botulinum Toxin in the Treatment of Chronic Migraine
 Botulinum Toin in the Treatment of Chronic Migraine ERIC L. FRY, MD ASSOCIATE PROFESSOR OF OPHTHALMOLOGY UNIVERSITY OF KANSAS FRY EYE ASSOCIATES, GARDEN CITY, KS History of Botulinum Toin Justinus Kerner,
Botulinum Toin in the Treatment of Chronic Migraine ERIC L. FRY, MD ASSOCIATE PROFESSOR OF OPHTHALMOLOGY UNIVERSITY OF KANSAS FRY EYE ASSOCIATES, GARDEN CITY, KS History of Botulinum Toin Justinus Kerner,
MEDICAL POLICY. I. POLICY Top BOTULINUM TOXIN MP-2.006 POLICY TITLE POLICY NUMBER
 Original Issue Date (Created): July 1, 2002 Most Recent Review Date (Revised): November 25, 2014 Effective Date: April 1, 2015 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT
Original Issue Date (Created): July 1, 2002 Most Recent Review Date (Revised): November 25, 2014 Effective Date: April 1, 2015 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT
Review for NHS Surrey Prescribing Clinical Network. Treatment: Botox for chronic migraine. Prepared by: Victoria Overland
 Review for NHS Surrey Prescribing Clinical Network Treatment: Botox for chronic migraine Prepared by: Victoria Overland Date: To be considered at September 2012 PCN meeting 1. Purpose of the Review NICE
Review for NHS Surrey Prescribing Clinical Network Treatment: Botox for chronic migraine Prepared by: Victoria Overland Date: To be considered at September 2012 PCN meeting 1. Purpose of the Review NICE
How To Use A Botox
 BTFR-04(17-28) 7/23/03 5:43 PM Page 17 4 Preparation,storage and injection technique NEVER handle botulinum toxin without surgical gloves. PRESENTATION Botox Botox is delivered in an insulated container.
BTFR-04(17-28) 7/23/03 5:43 PM Page 17 4 Preparation,storage and injection technique NEVER handle botulinum toxin without surgical gloves. PRESENTATION Botox Botox is delivered in an insulated container.
botulinum toxin type A, 50 unit, 100 unit and 200 unit powder for solution for injection (Botox ) SMC No. (692/11) Allergan Ltd
 Resubmission botulinum toxin type A, 50 unit, 100 unit and 200 unit powder for solution for injection (Botox ) SMC No. (692/11) Allergan Ltd 08 March 2013 The Scottish Medicines Consortium (SMC) has completed
Resubmission botulinum toxin type A, 50 unit, 100 unit and 200 unit powder for solution for injection (Botox ) SMC No. (692/11) Allergan Ltd 08 March 2013 The Scottish Medicines Consortium (SMC) has completed
Post Traumatic and other Headache Syndromes. Danielle L. Erb, MD Brain Rehabilitation Medicine, LLC Brain Injury Rehab Center, PRA
 Post Traumatic and other Headache Syndromes Danielle L. Erb, MD Brain Rehabilitation Medicine, LLC Brain Injury Rehab Center, PRA Over 45 million Americans have chronic, recurring headaches 62% of these
Post Traumatic and other Headache Syndromes Danielle L. Erb, MD Brain Rehabilitation Medicine, LLC Brain Injury Rehab Center, PRA Over 45 million Americans have chronic, recurring headaches 62% of these
HEADACHE. as. MUDr. Rudolf Černý, CSc. doc. MUDr. Petr Marusič, Ph.D.
 HEADACHE as. MUDr. Rudolf Černý, CSc. doc. MUDr. Petr Marusič, Ph.D. Dpt. of Neurology Charles University in Prague, 2nd Faculty of Medicine Motol University Hospital History of headache 1200 years B.C.
HEADACHE as. MUDr. Rudolf Černý, CSc. doc. MUDr. Petr Marusič, Ph.D. Dpt. of Neurology Charles University in Prague, 2nd Faculty of Medicine Motol University Hospital History of headache 1200 years B.C.
Chronic daily headache
 Chronic daily headache Chronic daily headache (CDH) is defined as any headache syndrome affecting more than half of the month, i.e. a headache on more than 15 days per month. It is also known as daily
Chronic daily headache Chronic daily headache (CDH) is defined as any headache syndrome affecting more than half of the month, i.e. a headache on more than 15 days per month. It is also known as daily
Tension Type Headaches
 Tension Type Headaches Research Review by : Dr. Ian MacIntyre Physiotherapy for tension-type Headache: A Controlled Study P. Torelli, R. Jenson, J. Olsen: Cephalalgia, 2004, 24, 29-36 Tension-type headache
Tension Type Headaches Research Review by : Dr. Ian MacIntyre Physiotherapy for tension-type Headache: A Controlled Study P. Torelli, R. Jenson, J. Olsen: Cephalalgia, 2004, 24, 29-36 Tension-type headache
BOTOX (onabotulinumtoxina) for injection, for intramuscular, intradetrusor, or intradermal use Initial U.S. Approval: 1989
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use safely and effectively. See full prescribing information for. (onabotulinumtoxina) for injection,
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use safely and effectively. See full prescribing information for. (onabotulinumtoxina) for injection,
Botulinum Toxin. Policy MEDICAL POLICY. Focal dystonias
 MEDICAL POLICY POLICY RELATED POLICIES POLICY GUIDELINES DESCRIPTION SCOPE BENEFIT APPLICATION RATIONALE REFERENCES CODING APPENDIX HISTORY Botulinum Toxin Number 5.01.512 Effective Date July 14, 2015
MEDICAL POLICY POLICY RELATED POLICIES POLICY GUIDELINES DESCRIPTION SCOPE BENEFIT APPLICATION RATIONALE REFERENCES CODING APPENDIX HISTORY Botulinum Toxin Number 5.01.512 Effective Date July 14, 2015
Chronic Headaches. David R. Greeley, MD, FAAN Northwest Neurological, PLLC October 23, 2015
 s David R. Greeley, MD, FAAN Northwest Neurological, PLLC October 23, 2015 Why is Headache Important? 36,000,000 people have migraine - more than asthma and diabetes combined 13,000,000 have chronic daily
s David R. Greeley, MD, FAAN Northwest Neurological, PLLC October 23, 2015 Why is Headache Important? 36,000,000 people have migraine - more than asthma and diabetes combined 13,000,000 have chronic daily
TITLE: Botulinum Toxin A for Headaches in Adults: A Review of Clinical-Effectiveness and Safety
 TITLE: Botulinum Toxin A for Headaches in Adults: A Review of Clinical-Effectiveness and Safety DATE: 28 July 2009 CONTEXT AND POLICY ISSUES: Botulinum toxin is a neurotoxin that causes muscle paralysis
TITLE: Botulinum Toxin A for Headaches in Adults: A Review of Clinical-Effectiveness and Safety DATE: 28 July 2009 CONTEXT AND POLICY ISSUES: Botulinum toxin is a neurotoxin that causes muscle paralysis
Botox treatment for chronic migraine
 University Teaching Trust Botox treatment for chronic migraine Humphrey Booth Building Neurosciences 0161 206 2563 0161 206 2427 All Rights Reserved 2016. Document for issue as handout.. This information
University Teaching Trust Botox treatment for chronic migraine Humphrey Booth Building Neurosciences 0161 206 2563 0161 206 2427 All Rights Reserved 2016. Document for issue as handout.. This information
Natural Modality in the Treatment of Primary Headaches. William S. Mihin, D.C. Catharine Helms, M.S. Michelle M. Anderson, M.S.N., F.N.P.
 Natural Modality in the Treatment of Primary Headaches William S. Mihin, D.C. Catharine Helms, M.S. Michelle M. Anderson, M.S.N., F.N.P. Abstract Headaches are both a prevalent and disabling condition.
Natural Modality in the Treatment of Primary Headaches William S. Mihin, D.C. Catharine Helms, M.S. Michelle M. Anderson, M.S.N., F.N.P. Abstract Headaches are both a prevalent and disabling condition.
33 % of whiplash patients develop. headaches originating from the upper. cervical spine
 33 % of whiplash patients develop headaches originating from the upper cervical spine - Dr Nikolai Bogduk Spine, 1995 1 Physical Treatments for Headache: A Structured Review Headache: The Journal of Head
33 % of whiplash patients develop headaches originating from the upper cervical spine - Dr Nikolai Bogduk Spine, 1995 1 Physical Treatments for Headache: A Structured Review Headache: The Journal of Head
Guidance for Industry Migraine: Developing Drugs for Acute Treatment
 Guidance for Industry Migraine: Developing Drugs for Acute Treatment DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft
Guidance for Industry Migraine: Developing Drugs for Acute Treatment DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft
Guidelines for Insurance Approval and the Appeal Process in the Treatment of Spasmodic Dysphonia with Botulinum Toxin Injections
 NATIONAL SPASMODIC DYSPHONIA ASSOCIATION Guidelines for Insurance Approval and the Appeal Process in the Treatment of Spasmodic Dysphonia with Botulinum Toxin Injections Published by National Spasmodic
NATIONAL SPASMODIC DYSPHONIA ASSOCIATION Guidelines for Insurance Approval and the Appeal Process in the Treatment of Spasmodic Dysphonia with Botulinum Toxin Injections Published by National Spasmodic
Sporadic attacks of severe tension-type headaches may respond to analgesics.
 MEDICATIONS While we are big advocates of non-drug treatments, many people do require the use of medications to control headaches. Headache medications are divided into two categories. Abortive drugs are
MEDICATIONS While we are big advocates of non-drug treatments, many people do require the use of medications to control headaches. Headache medications are divided into two categories. Abortive drugs are
CHRONIC MIGRAINE TACKLED HEAD-ON
 MEDIA RELEASE [Insert embargo time and date] March 2014 CHRONIC MIGRAINE TACKLED HEAD-ON BOTOX (botulinum toxin type A) receives government subsidy for debilitating condition Sydney, Australia, [insert
MEDIA RELEASE [Insert embargo time and date] March 2014 CHRONIC MIGRAINE TACKLED HEAD-ON BOTOX (botulinum toxin type A) receives government subsidy for debilitating condition Sydney, Australia, [insert
BOTOX is the first and only preventive treatment proven to reduce headache days every month.
 For adults with Chronic Migraine, 15 or more headache days a month, each lasting 4 hours or more, is the first and only preventive treatment proven to reduce headache days every month. prevents up to 9
For adults with Chronic Migraine, 15 or more headache days a month, each lasting 4 hours or more, is the first and only preventive treatment proven to reduce headache days every month. prevents up to 9
Webinar title: Know Your Options for Treating Severe Spasticity
 Webinar title: Know Your Options for Treating Severe Spasticity Presented by: Dr. Gerald Bilsky, Physiatrist Medical Director of Outpatient Services and Associate Medical Director of Acquired Brain Injury
Webinar title: Know Your Options for Treating Severe Spasticity Presented by: Dr. Gerald Bilsky, Physiatrist Medical Director of Outpatient Services and Associate Medical Director of Acquired Brain Injury
Ask your doctor about BOTOX and Chronic Migraine. Learn more at BOTOXChronicMigraine.com
 Track your headache days and see if you have Chronic Migraine Indication (onabotulinumtoxina) is a prescription medicine that is injected to prevent headaches in adults with chronic migraine who have 15
Track your headache days and see if you have Chronic Migraine Indication (onabotulinumtoxina) is a prescription medicine that is injected to prevent headaches in adults with chronic migraine who have 15
Botulinum Toxin: Could It Be An Effective Treatment For Chronic Tension Type Headache?
 Sherif M. Hamdy et al. Botulinum Toxin: Could It Be An Effective Treatment For Chronic Tension Type Headache? Sherif M. Hamdy 1, Hatem Samir 1, M. El-Sayed 1, Nermin Adel 1, Rasha Hassan 2 Department of
Sherif M. Hamdy et al. Botulinum Toxin: Could It Be An Effective Treatment For Chronic Tension Type Headache? Sherif M. Hamdy 1, Hatem Samir 1, M. El-Sayed 1, Nermin Adel 1, Rasha Hassan 2 Department of
Blepharospasm: 1.25 Units-2.5 Units into each of 3 sites per affected eye (2.6) Strabismus: 1.25 Units-2.5 Units initially in any one muscle (2.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use safely and effectively. See full prescribing information for. (onabotulinumtoxina) Initial U.S. Approval:
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use safely and effectively. See full prescribing information for. (onabotulinumtoxina) Initial U.S. Approval:
BOTOX (onabotulinumtoxina) for injection, for intramuscular, intradetrusor, or intradermal use Initial U.S. Approval: 1989
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use safely and effectively. See full prescribing information for. (onabotulinumtoxina) for injection,
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use safely and effectively. See full prescribing information for. (onabotulinumtoxina) for injection,
Informed Consent BOTOX / DYSPORT / XEOMIN Injection for
 Fresno Laser Aesthetics 1105 E. Spruce Ph (559) 432-3492 Suite 203 Fax (559) 256-7711 Fresno, CA 93720 Informed Consent BOTOX / DYSPORT / XEOMIN Injection for INSTRUCTIONS This is an informed-consent document
Fresno Laser Aesthetics 1105 E. Spruce Ph (559) 432-3492 Suite 203 Fax (559) 256-7711 Fresno, CA 93720 Informed Consent BOTOX / DYSPORT / XEOMIN Injection for INSTRUCTIONS This is an informed-consent document
15 or more headache days a month, each headache lasting 4 hours or more. Isn t it time to reduce those headache days?
 15 or more headache days a month, each headache lasting 4 hours or more (onabotulinumtoxina) is a prescription medicine that is injected to prevent headaches in adults with chronic migraine who have 15
15 or more headache days a month, each headache lasting 4 hours or more (onabotulinumtoxina) is a prescription medicine that is injected to prevent headaches in adults with chronic migraine who have 15
Botulinum Neurotoxin for Chronic Migraine and Trigeminal Neuralgia
 Botulinum Neurotoxin for Chronic Migraine and Trigeminal Neuralgia David W. Dodick Professor Department of Neurology, Mayo Clinic Phoenix Arizona, USA Onabotulinumtoxin A is the only botulinum toxin serotype
Botulinum Neurotoxin for Chronic Migraine and Trigeminal Neuralgia David W. Dodick Professor Department of Neurology, Mayo Clinic Phoenix Arizona, USA Onabotulinumtoxin A is the only botulinum toxin serotype
Classification of Chronic Headache
 Chronic Headache Classification of Chronic Headache JMAJ 47(3): 112 117, 2004 Mitsunori MORIMATSU Professor, Department of Neurology and Clinical Neuroscience, Yamaguchi University School of Medicine Abstract:
Chronic Headache Classification of Chronic Headache JMAJ 47(3): 112 117, 2004 Mitsunori MORIMATSU Professor, Department of Neurology and Clinical Neuroscience, Yamaguchi University School of Medicine Abstract:
Guidelines for Medical Necessity Determination for Speech and Language Therapy
 Guidelines for Medical Necessity Determination for Speech and Language Therapy These Guidelines for Medical Necessity Determination (Guidelines) identify the clinical information MassHealth needs to determine
Guidelines for Medical Necessity Determination for Speech and Language Therapy These Guidelines for Medical Necessity Determination (Guidelines) identify the clinical information MassHealth needs to determine
Behavioral and Physical Treatments for Tension-type and Cervicogenic Headache
 Evidence Report: Behavioral and Physical Treatments for Tension-type and Cervicogenic Headache Douglas C. McCrory, MD, MHSc Donald B. Penzien, PhD Vic Hasselblad, PhD Rebecca N. Gray, DPhil Duke University
Evidence Report: Behavioral and Physical Treatments for Tension-type and Cervicogenic Headache Douglas C. McCrory, MD, MHSc Donald B. Penzien, PhD Vic Hasselblad, PhD Rebecca N. Gray, DPhil Duke University
Is Botox-A Effective For Headaches and Neck Pain?
 0008-3194/2006/263 270/$2.00/ JCCA 2006 The use of botulinum neurotoxin type A (Botox) for headaches: a case review Mia Oliver, BA, DC* Joanna MacDonald, BHK, DC* Moez Rajwani, DC** Chiropractic care is
0008-3194/2006/263 270/$2.00/ JCCA 2006 The use of botulinum neurotoxin type A (Botox) for headaches: a case review Mia Oliver, BA, DC* Joanna MacDonald, BHK, DC* Moez Rajwani, DC** Chiropractic care is
Appendix 1 (as supplied by the authors): Framework and content of the intervention of the LIMIT study
 Appendix 1 (as supplied by the authors): Framework and content of the intervention of the LIMIT study Background The training was based on the learning cycle of Kolb 1. According to this cycle effective
Appendix 1 (as supplied by the authors): Framework and content of the intervention of the LIMIT study Background The training was based on the learning cycle of Kolb 1. According to this cycle effective
Prior Authorization Guideline
 Prior Authorization Guideline Guideline: MEDPD SPAP Inj - Botox Therapeutic Class: Musculoskeletal Agents Therapeutic Sub-Class: Musculoskeletal Relaxant Agents Client: PDP SPAP Inj Approval Date: 3/17/2000
Prior Authorization Guideline Guideline: MEDPD SPAP Inj - Botox Therapeutic Class: Musculoskeletal Agents Therapeutic Sub-Class: Musculoskeletal Relaxant Agents Client: PDP SPAP Inj Approval Date: 3/17/2000
Frequent headache is defined as headaches 15 days/month and daily. Course of Frequent/Daily Headache in the General Population and in Medical Practice
 DISEASE STATE REVIEW Course of Frequent/Daily Headache in the General Population and in Medical Practice Egilius L.H. Spierings, MD, PhD, Willem K.P. Mutsaerts, MSc Department of Neurology, Brigham and
DISEASE STATE REVIEW Course of Frequent/Daily Headache in the General Population and in Medical Practice Egilius L.H. Spierings, MD, PhD, Willem K.P. Mutsaerts, MSc Department of Neurology, Brigham and
DEPRESSION CARE PROCESS STEP EXPECTATIONS RATIONALE
 1 DEPRESSION CARE PROCESS STEP EXPECTATIONS RATIONALE ASSESSMENT/PROBLEM RECOGNITION 1. Did the staff and physician seek and document risk factors for depression and any history of depression? 2. Did staff
1 DEPRESSION CARE PROCESS STEP EXPECTATIONS RATIONALE ASSESSMENT/PROBLEM RECOGNITION 1. Did the staff and physician seek and document risk factors for depression and any history of depression? 2. Did staff
Clinical and Therapeutic Cannabis Information. Written by Cannabis Training University (CTU) All rights reserved
 Clinical and Therapeutic Cannabis Information Written by Cannabis Training University (CTU) All rights reserved Contents Introduction... 3 Chronic Pain... 6 Neuropathic Pain... 8 Movement Disorders...
Clinical and Therapeutic Cannabis Information Written by Cannabis Training University (CTU) All rights reserved Contents Introduction... 3 Chronic Pain... 6 Neuropathic Pain... 8 Movement Disorders...
Views and Perspectives
 Headache 2010 American Headache Society ISSN 0017-8748 doi: 10.1111/j.1526-4610.2010.01764.x Published by Wiley Periodicals, Inc. Views and Perspectives Defining the Pharmacologically Intractable Headache
Headache 2010 American Headache Society ISSN 0017-8748 doi: 10.1111/j.1526-4610.2010.01764.x Published by Wiley Periodicals, Inc. Views and Perspectives Defining the Pharmacologically Intractable Headache
A Guide to Urinary Incontinence (UI) in Adults With Overactive Bladder (OAB) Due to a Neurologic Condition
 A Guide to Urinary Incontinence (UI) in Adults With Overactive Bladder (OAB) Due to a Neurologic Condition Information and treatment options BOTOX is a prescription medicine that is injected into muscles
A Guide to Urinary Incontinence (UI) in Adults With Overactive Bladder (OAB) Due to a Neurologic Condition Information and treatment options BOTOX is a prescription medicine that is injected into muscles
Update on Therapeutics in Neurology Evaluation Tabulation
 Update on Therapeutics in Neurology Evaluation Tabulation June 23, 2011 Georgetown, Washington, D.C. I am a: What was your overall rating of the faculty Jeffrey Frank, MD? The learning objectives designated
Update on Therapeutics in Neurology Evaluation Tabulation June 23, 2011 Georgetown, Washington, D.C. I am a: What was your overall rating of the faculty Jeffrey Frank, MD? The learning objectives designated
Tension-type headache Non-pharmacological and pharmacological treatment
 Danish Headache Center Tension-type headache Non-pharmacological and pharmacological treatment Lars Bendtsen Associate professor, MD, PhD, Dr Med Sci Danish Headache Center, Department of Neurology Glostrup
Danish Headache Center Tension-type headache Non-pharmacological and pharmacological treatment Lars Bendtsen Associate professor, MD, PhD, Dr Med Sci Danish Headache Center, Department of Neurology Glostrup
POSTER PRESENTATIONS
 POSTER PRESENTATIONS POSTER PRESENTATIONS The following are based on posters presented at the 44th Annual Meeting of the American Headache Society, held June 21-23, 2002, in Seattle, Washington. TOPIRAMATE
POSTER PRESENTATIONS POSTER PRESENTATIONS The following are based on posters presented at the 44th Annual Meeting of the American Headache Society, held June 21-23, 2002, in Seattle, Washington. TOPIRAMATE
New appendix criteria open for a broader concept of chronic migraine
 Blackwell Publishing LtdOxford, UKCHACephalalgia0333-1024Blackwell Science, 20062006266742746Original ArticleA broader concept of chronic migrainej Olesen et al. BRIEF REPORT New appendix criteria open
Blackwell Publishing LtdOxford, UKCHACephalalgia0333-1024Blackwell Science, 20062006266742746Original ArticleA broader concept of chronic migrainej Olesen et al. BRIEF REPORT New appendix criteria open
PATIENT PACKAGE INSERT IN ACCORDANCE WITH THE PHARMACISTS' REGULATIONS (PREPARATIONS) - 1986
 PATIENT PACKAGE INSERT IN ACCORDANCE WITH THE PHARMACISTS' REGULATIONS (PREPARATIONS) - 1986 The dispensing of this medicine requires a doctor s prescription BOTOX 50 Powder for solution for injection
PATIENT PACKAGE INSERT IN ACCORDANCE WITH THE PHARMACISTS' REGULATIONS (PREPARATIONS) - 1986 The dispensing of this medicine requires a doctor s prescription BOTOX 50 Powder for solution for injection
OnabotulinumtoxinA 155 U in medication overuse headache: a two years prospective study
 DOI 10.1186/s40064-015-1636-9 RESEARCH Open Access OnabotulinumtoxinA 155 U in medication overuse headache: a two years prospective study Andrea Negro 1,2,6*, Martina Curto 2,3,4, Luana Lionetto 5, Dorotea
DOI 10.1186/s40064-015-1636-9 RESEARCH Open Access OnabotulinumtoxinA 155 U in medication overuse headache: a two years prospective study Andrea Negro 1,2,6*, Martina Curto 2,3,4, Luana Lionetto 5, Dorotea
Ultrasound guided onabotulinum injection in chronic migraine: a case series
 Ultrasound guided onabotulinum injection in chronic migraine: a case series L. Di Lorenzo 1, F. Di Stani 2, D. Santopadre 3, C. Foti 4 1 Department of Rehabilitation, Hospital G. Rummo, Benevento, Italy
Ultrasound guided onabotulinum injection in chronic migraine: a case series L. Di Lorenzo 1, F. Di Stani 2, D. Santopadre 3, C. Foti 4 1 Department of Rehabilitation, Hospital G. Rummo, Benevento, Italy
Headaches and Kids. Jennifer Bickel, MD Assistant Professor of Neurology Co-Director of Headache Clinic Children s Mercy Hospital
 Headaches and Kids Jennifer Bickel, MD Assistant Professor of Neurology Co-Director of Headache Clinic Children s Mercy Hospital Overview Headache classifications and diagnosis Address common headache
Headaches and Kids Jennifer Bickel, MD Assistant Professor of Neurology Co-Director of Headache Clinic Children s Mercy Hospital Overview Headache classifications and diagnosis Address common headache
An impressive arsenal of medications and
 COMMON PROPHYLACTIC PRACTICES FOR CHRONIC DAILY HEADACHE * John F. Rothrock, MD ABSTRACT A variety of medications and treatment interventions, including antiepileptic drugs, tricyclic antidepressants,
COMMON PROPHYLACTIC PRACTICES FOR CHRONIC DAILY HEADACHE * John F. Rothrock, MD ABSTRACT A variety of medications and treatment interventions, including antiepileptic drugs, tricyclic antidepressants,
Headache - What is Your Migraine Size?
 Headache The Pharmacist s Role in Assessment & Peter Loewen, B.Sc.(Pharm), Pharm.D. Vancouver Hospital & Health Sciences Centre University of British Columbia ETC, Headache. Nan Quintin www.vhpharmsci.com
Headache The Pharmacist s Role in Assessment & Peter Loewen, B.Sc.(Pharm), Pharm.D. Vancouver Hospital & Health Sciences Centre University of British Columbia ETC, Headache. Nan Quintin www.vhpharmsci.com
Tension-type headache Non-pharmacological and pharmacological treatment
 Danish Headache Center Tension-type headache Non-pharmacological and pharmacological treatment Lars Bendtsen Associate professor, MD, PhD, Dr Med Sci Danish Headache Center, Department of Neurology Glostrup
Danish Headache Center Tension-type headache Non-pharmacological and pharmacological treatment Lars Bendtsen Associate professor, MD, PhD, Dr Med Sci Danish Headache Center, Department of Neurology Glostrup
Headaches in Children How to Manage Difficult Headaches
 Headaches in Children How to Manage Difficult Headaches Peter Procopis Childhood headaches Differential diagnosis Migraine Psychological Raised Pressure Childhood headaches Other causes: Constitutional
Headaches in Children How to Manage Difficult Headaches Peter Procopis Childhood headaches Differential diagnosis Migraine Psychological Raised Pressure Childhood headaches Other causes: Constitutional
Clinical & Quality Management BOTULINUM TOXIN. Table of Contents: Page: Cross Reference Policy:
 BOTULINUM TOXIN Policy Number: 2014D0070A Effective Date: December 1, 2014 Table of Contents: Page: Cross Reference Policy: POLICY DESCRIPTION 2 Not Available. COVERAGE RATIONALE/CLINICAL CONSIDERATIONS
BOTULINUM TOXIN Policy Number: 2014D0070A Effective Date: December 1, 2014 Table of Contents: Page: Cross Reference Policy: POLICY DESCRIPTION 2 Not Available. COVERAGE RATIONALE/CLINICAL CONSIDERATIONS
Vision Health: Conditions, Disorders & Treatments NEUROOPTHALMOLOGY
 Vision Health: Conditions, Disorders & Treatments NEUROOPTHALMOLOGY Neuroophthalmology focuses on conditions caused by brain or systemic abnormalities that result in visual disturbances, among other symptoms.
Vision Health: Conditions, Disorders & Treatments NEUROOPTHALMOLOGY Neuroophthalmology focuses on conditions caused by brain or systemic abnormalities that result in visual disturbances, among other symptoms.
Pharmacy Medical Necessity Guidelines: Botulinum Toxins
 Pharmacy Medical Necessity Guidelines: Effective: June 14, 2016 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy (RX) or Medical (MED) Benefit
Pharmacy Medical Necessity Guidelines: Effective: June 14, 2016 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy (RX) or Medical (MED) Benefit
Michelle H. Cameron, M.D., P.T., M.C.R. Portland VA MS Center of Excellence- West, and Oregon Health & Science University
 Michelle H. Cameron, M.D., P.T., M.C.R. Portland VA MS Center of Excellence- West, and Oregon Health & Science University Ileana Howard, M.D. VA Puget Sound, Seattle, WA and University of Washington PVA
Michelle H. Cameron, M.D., P.T., M.C.R. Portland VA MS Center of Excellence- West, and Oregon Health & Science University Ileana Howard, M.D. VA Puget Sound, Seattle, WA and University of Washington PVA
Drugs for MS.Drug fact box cannabis extract (Sativex) Version 1.0 Author
 Version History Policy Title Drugs for MS.Drug fact box cannabis extract (Sativex) Version 1.0 Author West Midlands Commissioning Support Unit Publication Date Jan 2013 Review Date Supersedes/New (Further
Version History Policy Title Drugs for MS.Drug fact box cannabis extract (Sativex) Version 1.0 Author West Midlands Commissioning Support Unit Publication Date Jan 2013 Review Date Supersedes/New (Further
BOTOX (onabotulinumtoxina) for injection, for intramuscular, intradetrusor, or intradermal use Initial U.S. Approval: 1989
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use safely and effectively. See full prescribing information for. (onabotulinumtoxina) for injection,
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use safely and effectively. See full prescribing information for. (onabotulinumtoxina) for injection,
Efficacy, safety, and predictors of response to botulinum toxin type A in refractory chronic migraine: A retrospective study
 Available online at www.sciencedirect.com ScienceDirect Journal of the Chinese Medical Association 77 (2014) 10e15 Original Article Efficacy, safety, and predictors of response to botulinum toxin type
Available online at www.sciencedirect.com ScienceDirect Journal of the Chinese Medical Association 77 (2014) 10e15 Original Article Efficacy, safety, and predictors of response to botulinum toxin type
Botulinum toxin for the treatment of headache: a promising path on a dead end road?
 Acta Neurol. Belg., 2010, 110, 221-229 Review article Botulinum toxin for the treatment of headache: a promising path on a dead end road? Stephanie DELSTANCHE and Jean SCHOENEN Headache Research Unit.
Acta Neurol. Belg., 2010, 110, 221-229 Review article Botulinum toxin for the treatment of headache: a promising path on a dead end road? Stephanie DELSTANCHE and Jean SCHOENEN Headache Research Unit.
Handicap after acute whiplash injury A 1-year prospective study of risk factors
 1 Handicap after acute whiplash injury A 1-year prospective study of risk factors Neurology 2001;56:1637-1643 (June 26, 2001) Helge Kasch, MD, PhD; Flemming W Bach, MD, PhD; Troels S Jensen, MD, PhD From
1 Handicap after acute whiplash injury A 1-year prospective study of risk factors Neurology 2001;56:1637-1643 (June 26, 2001) Helge Kasch, MD, PhD; Flemming W Bach, MD, PhD; Troels S Jensen, MD, PhD From
Sativex. Spirella Building, Letchworth, SG6 4ET 01462 476700 www.mstrust.org.uk reg charity no. 1088353
 Sativex Spirella Building, Letchworth, SG6 4ET 01462 476700 www.mstrust.org.uk reg charity no. 1088353 Sativex Date of issue: July 2010 Review date: July 2011 Contents 1. Introduction 1 2. What is Sativex?
Sativex Spirella Building, Letchworth, SG6 4ET 01462 476700 www.mstrust.org.uk reg charity no. 1088353 Sativex Date of issue: July 2010 Review date: July 2011 Contents 1. Introduction 1 2. What is Sativex?
PROTOCOL SYNOPSIS Evaluation of long-term opioid efficacy for chronic pain
 P a g e 1 PROTOCOL SYNOPSIS Evaluation of long-term opioid efficacy for chronic pain Clinical Phase 4 Study Centers Study Period 25 U.S. sites identified and reviewed by the Steering Committee and Contract
P a g e 1 PROTOCOL SYNOPSIS Evaluation of long-term opioid efficacy for chronic pain Clinical Phase 4 Study Centers Study Period 25 U.S. sites identified and reviewed by the Steering Committee and Contract
BOTOX Injection (Onabotulinumtoxin A) for Chronic Migraine Headaches
 BOTOX Injection (Onabotulinumtoxin A) for Chronic Migraine Headaches [For the list of services and procedures that need preauthorization, please refer to www.mcs.com.pr go to Comunicados a Proveedores,
BOTOX Injection (Onabotulinumtoxin A) for Chronic Migraine Headaches [For the list of services and procedures that need preauthorization, please refer to www.mcs.com.pr go to Comunicados a Proveedores,
Breaking the cycle of medication overuse headache
 REVIEW CME CREDIT EDUCATIONAL OBJECTIVE: Readers will recognize the risk of developing medication overuse headache with increasing use of medications to treat acute migraine Stewart J. Tepper, MD * Center
REVIEW CME CREDIT EDUCATIONAL OBJECTIVE: Readers will recognize the risk of developing medication overuse headache with increasing use of medications to treat acute migraine Stewart J. Tepper, MD * Center
Migraine is one of the most prevalent
 Migraine is a common, incapacitating disorder that is underdiagnosed in clinical practice. Early and correct diagnosis of migraine is essential and can lead to significant improvements in a patient s quality
Migraine is a common, incapacitating disorder that is underdiagnosed in clinical practice. Early and correct diagnosis of migraine is essential and can lead to significant improvements in a patient s quality
None related to the presentation Grants to conduct clinical trials from:
 Chronic Daily Headache Bassel F. Shneker, MD, MBA Associate Professor Vice Chair, OSU Neurology The Ohio State University Wexner Medical Center Financial Disclosures None related to the presentation Grants
Chronic Daily Headache Bassel F. Shneker, MD, MBA Associate Professor Vice Chair, OSU Neurology The Ohio State University Wexner Medical Center Financial Disclosures None related to the presentation Grants
Migraine headaches affect over 35 million RECONSTRUCTIVE
 RECONSTRUCTIVE A Comparison of Outcome of Surgical Treatment of Migraine Headaches Using a Constellation of Symptoms versus Botulinum Toxin Type A to Identify the Trigger Sites Mengyuan T. Liu, B.S. Bryan
RECONSTRUCTIVE A Comparison of Outcome of Surgical Treatment of Migraine Headaches Using a Constellation of Symptoms versus Botulinum Toxin Type A to Identify the Trigger Sites Mengyuan T. Liu, B.S. Bryan
The Prevalence of Neck Pain in Migrainehead_1608
 1..5 Headache 2010 the Authors Journal compilation 2010 American Headache Society ISSN 0017-8748 doi: 10.1111/j.1526-4610.2009.01608.x Published by Wiley Periodicals, Inc. Research Submission The Prevalence
1..5 Headache 2010 the Authors Journal compilation 2010 American Headache Society ISSN 0017-8748 doi: 10.1111/j.1526-4610.2009.01608.x Published by Wiley Periodicals, Inc. Research Submission The Prevalence
One Day at a Time: When Headaches Become Chronic. Robert Shapiro, MD, PhD
 One Day at a Time: When Headaches Become Chronic Robert Shapiro, MD, PhD Disclosures Scientific/Medical Advisory Boards (since 10/12) Transcept Pharmaceuticals Chronic Headaches: Overview What is a chronic
One Day at a Time: When Headaches Become Chronic Robert Shapiro, MD, PhD Disclosures Scientific/Medical Advisory Boards (since 10/12) Transcept Pharmaceuticals Chronic Headaches: Overview What is a chronic
New daily persistent headache in the paediatric population
 doi:10.1111/j.1468-2982.2008.01647.x New daily persistent headache in the paediatric population E Kung 1, SJ Tepper 2, AM Rapoport 3, FD Sheftell 4 & ME Bigal 1,5 1 Department of Neurology, Albert Einstein
doi:10.1111/j.1468-2982.2008.01647.x New daily persistent headache in the paediatric population E Kung 1, SJ Tepper 2, AM Rapoport 3, FD Sheftell 4 & ME Bigal 1,5 1 Department of Neurology, Albert Einstein
Guideline for the Management of Acute Peripheral Facial nerve palsy. Bells Palsy in Children
 Guideline for the Management of Acute Peripheral Facial nerve palsy Definition Bells Palsy in Children Bell palsy is an acute, idiopathic unilateral lower motor neurone facial nerve palsy that is not associated
Guideline for the Management of Acute Peripheral Facial nerve palsy Definition Bells Palsy in Children Bell palsy is an acute, idiopathic unilateral lower motor neurone facial nerve palsy that is not associated
HEADACHES IN CHILDREN AND ADOLESCENTS. Brian D. Ryals, M.D.
 HEADACHES IN CHILDREN AND ADOLESCENTS Brian D. Ryals, M.D. Frequency and Type of Headaches in Schoolchildren 8993 children age 7-15 in Sweden Migraine in 4% Frequent Nonmigrainous in 7% Infrequent Nonmigrainous
HEADACHES IN CHILDREN AND ADOLESCENTS Brian D. Ryals, M.D. Frequency and Type of Headaches in Schoolchildren 8993 children age 7-15 in Sweden Migraine in 4% Frequent Nonmigrainous in 7% Infrequent Nonmigrainous
Botulinum toxin A for the treatment of chronic neck pain
 Pain 94 (2001) 255 260 www.elsevier.com/locate/pain Botulinum toxin A for the treatment of chronic neck pain Anthony H. Wheeler a, *, Paula Goolkasian b, Stephanie S. Gretz c a Charlotte Spine Center,
Pain 94 (2001) 255 260 www.elsevier.com/locate/pain Botulinum toxin A for the treatment of chronic neck pain Anthony H. Wheeler a, *, Paula Goolkasian b, Stephanie S. Gretz c a Charlotte Spine Center,
Oh, 14 C O M M U N I T Y M A G A Z I N E S M A Y / J U N E 2 0 0 8
 14 C O M M U N I T Y M A G A Z I N E S M A Y / J U N E 2 0 0 8 Oh, my aching head NEW DOCTOR TEAM ADDRESSES CHRONIC HEADACHE PROBLEMS SEVERE HEADACHES IN THIS COUNTRY ARE A LEADING CAUSE OF DISRUP- TION
14 C O M M U N I T Y M A G A Z I N E S M A Y / J U N E 2 0 0 8 Oh, my aching head NEW DOCTOR TEAM ADDRESSES CHRONIC HEADACHE PROBLEMS SEVERE HEADACHES IN THIS COUNTRY ARE A LEADING CAUSE OF DISRUP- TION
Which injectable medication should I take for relapsing-remitting multiple sclerosis?
 Which injectable medication should I take for relapsing-remitting multiple sclerosis? A decision aid to discuss options with your doctor This decision aid is for you if you: Have multiple sclerosis Have
Which injectable medication should I take for relapsing-remitting multiple sclerosis? A decision aid to discuss options with your doctor This decision aid is for you if you: Have multiple sclerosis Have
Alan Rosenberg, MD VP Medical Policy, Technology Assessment and Credentialing WellPoint, Inc. 233 S. Wacker Drive, Suite 3900 Chicago, IL 60606
 October 5, 2010 Alan Rosenberg, MD VP Medical Policy, Technology Assessment and Credentialing WellPoint, Inc. 233 S. Wacker Drive, Suite 3900 Chicago, IL 60606 Dear Dr. Rosenberg, The American Gastroenterological
October 5, 2010 Alan Rosenberg, MD VP Medical Policy, Technology Assessment and Credentialing WellPoint, Inc. 233 S. Wacker Drive, Suite 3900 Chicago, IL 60606 Dear Dr. Rosenberg, The American Gastroenterological
Pain Syndromes Acute Neck and Back Pain with or without arm or leg pain Chronic Neck and Back Pain with or without arm or leg pain
 Pain Syndromes The most common and best understood type of pain is acute pain. Acute pain will result when tissue is injured by trauma, surgery, illness, or infection. This type of pain is generally understood
Pain Syndromes The most common and best understood type of pain is acute pain. Acute pain will result when tissue is injured by trauma, surgery, illness, or infection. This type of pain is generally understood
Oculopharyngeal muscular dystrophy (OPMD)
 Oculopharyngeal muscular dystrophy (OPMD) The term muscular dystrophy is used to cover a wide range of conditions which have in common progressive muscle weakness due to an inherited genetic defect (mutation).
Oculopharyngeal muscular dystrophy (OPMD) The term muscular dystrophy is used to cover a wide range of conditions which have in common progressive muscle weakness due to an inherited genetic defect (mutation).

![Bell s Palsy ]]> <![CDATA[Bell's Palsy]]> Bell s Palsy ]]> <![CDATA[Bell's Palsy]]>](/thumbs/27/10105461.jpg)