Experimental Brain Research 9 Springer-Vertag 1990
|
|
|
- Kelly Bishop
- 5 years ago
- Views:
Transcription
1 Exp Brain Res (1990) 82: Experimental Brain Research 9 Springer-Vertag 1990 Saccade-related responses of centrifugal neurons projecting to the chicken retina G. Marin, J.C. Letelier, and J. Wallman Biology Department, City College, City University of New York, New York, NY 10031, USA Received September 18, 1989 / Accepted March 30, 1990 Summary. Centrifugal projections to several sensory systems modulate the afferent activity during active behaviors. To see whether such modulation occurred in the visual system, we recorded the activity of isthmo-optic neurons in awake chickens during eye movements. We find that the discharge of all isthmo-optic neurons tends to stop during saccades, although every neuron does not pause for every saccade. The pause begins at approximately the same time as the saccade, and pause duration is correlated with saccade duration. Pausing during saccades occurs in both dark and light suggesting that it is motoric rather that visual in origin. In addition, we find that the spontaneous activity of isthmo-optic neurons increases in darkness. We discuss the significance of the saccadic modulation of isthmo-optic activity in terms of possible functions of the centrifugal projection in modulation of ganglion cell activity. Key words: Isthmo-optic nucleus Saccades Retinal ganglion cells - Chickens Introduction Despite thirty years of study, the functions of the centrifugal projection to the avian retina are not well understood. Anatomical studies carried out in chickens and pigeons have demonstrated that the only source of centrifugal fibers to the birds' retina are the isthmo-optic nucleus (ION) of the caudal midbrain (McGill et al. 1966b; Cowan 1970) and the so-called ectopic cells surrounding the nucleus (Wolf-Oberhollenzer 1987). The ION receives a topographically organized projection from the ipsilateral optic tectum (McGill et al. 1966a). Neurons in the ION project back to the retinal quadrant that is the source of their visual input (McGill et al. 1966b). As a result of this anatomical arrangement, each part of the Offprint requests to. G. Marin (address see above) retina projects via the tectum to that part of the nucleus from which it receives its centrifugal afferents. The centrifugal fibers terminate in the inner plexiform layer of the retina, where they make large synapses on the primary processes of amacrine cells (Maturana and Frenk 1965; Dowling and Cowan 1966), and perhaps upon displaced ganglion cells (Maturana and Frenk 1965). Experiments in which the isthmo-optic tract was either electrically stimulated (Miles 1972c; Holden 1978) or reversibly cooled (Miles 1972d; Pearlman and Hughes 1976b) all show that the centrifugal activity enhances the response of retinal ganglion cells to visual stimulation. In chickens and pigeons this effect is produced mainly by centrifugal suppression of the ganglion cells' inhibitory surround (Miles 1972c; Holden 1978), presumably via amacrine cells. Despite these influences, only subtle visual deficiencies result from lesioning the ION or the isthtoo-optic tract (Rogers and Miles 1972; Shortess and Klose 1977; Knipling 1978). In other sensory systems, centrifugal fibers modulate the sensitivity of sensory cells during active movements of the animal. For example, the efferent fibers to the lateral line organs of fishes and amphibia fire during movements and prevent self excitation by inhibiting afferent discharge (Russell and Roberts 1974). On the other hand, efferents to the muscle spindles increase the responsiveness of the afferents while the muscle is shortening (Matthews 1982). The centrifugal fibers of the vestibular system in monkeys may function to extend the dynamic range of the afferents during large accelerations accompanying voluntary head movements (Goldberg and Fernandez 1980). To explore the possibility that the centrifugal activity in the visual system is also modulated by active movements, we recorded from ION neurons of awake chickens. We investigated the relation between the firing pattern of ION cells and eye movements. We found that ION cells, while discharging at a variable rate during fixational intervals, tend to pause during saccades. A preliminary report of these findings has been presented (Letelier et al. 1985).
2 264 Methods The experiments were carried out on 14 four-week-old White Leghorn chicks (Gallus gallus domesticus) hatched in the laboratory from eggs obtained from a commercial supplier. In each animal recordings were obtained from both IO nuclei in two sessions two or three days apart. To permit stabilizing the head during the experiment without obscuring vision, two days before the first recording session the animals were deeply anesthetized with Chloropent (0.35 ml per 100 g body weight), positioned in a stereotaxic apparatus, and an acrylic plate was attached with dental cement to the skull and to a pair of anchoring screws fixed to the frontal bones on each side of the head. Then, a bilateral craniotomy over both IONs was made without opening the underlying dura. A 4 x 8 mm acrylic chamber was cemented to the skull, filled with agar saline and sealed with low melting point wax. Animals recovered overnight and fed normally the following day. Eye movements were recorded using the magnetic field search coil technique. In most experiments only the horizontal component of eye movements was measured, but on some occasions the vertical or the torsional component was simultaneously measured by a second coil placed perpendicular to the first. On the day of the experiment the bird was anesthetized with halothane, and a search coil was attached to the eye contralateral to the side of the ION to be recorded from (for details of this method, see Wallman et al. 1982). After a recovery period of two hours, the animal was placed in an alternating magnetic field, the lower lid was retracted with a fine thread and the plastic chamber was opened, exposing the brain for electrode penetrations. The chamber was kept filled with saline solution. The animal, loosely wrapped in a towel, lay calmly throughout the experiment except for occasional episodes of vocalization and neck movement. Treatment of the animals was in accord with the Helsinki/APS agreement. Extracellular recordings were made using tungsten-in-glass microelectrodes (Merrill and Ainsworth 1972) with five to twenty Ixm of exposed tip. Microelectrodes were passed through the dura and advanced into the brain using an hydraulic micromanipulator. The recording system was conventional. Visual stimuli consisted of dark bars and squares moved on a tangent screen located about 50 cm from the bird's eye. Stimuli were moved by hand or reflected from a mirror mounted on a galvanometer driven by a sinusoidal or trapezoidal voltage function. Optokinetic stimulation was produced with a vertical striped drum that surrounded the animal, and was rotated at a constant velocity by a servo motor. For recordings in darkness, the animal was surrounded by black curtains and the room lights turned off. Single- or multi-unit activity from the IO nucleus together with the eye position signal were recorded on a FM tape recorder for subsequent analysis. In order to localize the recording sites, small lesions were produced by passing anodal current (usually 5 pa for 5 s) through the microelectrode. At the end of the first session, the birds were anesthetized with halothane, the incision over the eye sutured and the plastic chamber resealed. To reconstruct recording locations, the chicks were deeply anesthetized with pentobarbital and perfused ir~tracardially with 0.85% saline followed by 10% formalin. Brains were cut in 40 gm sections on a freezing microtome and stained with cresyl violet for histological analysis. Spike data and eye position were sampled by a DEC 11/73 computer at 1000 samples/s for 600 ms around each saccade, thereby sampling the unit activity at least 200 ms before and 200 ms after the saccade. These data were used to make peri-stimulus histograms synchronized with the beginning and the end of each saccade using one of two methods. The first method electronically detected the saccade beginning and end using a special purpose device that relied on the characteristic 25 Hz oscillation of the eye during saccades. This method had an uncertainity of +/- 5 ms for the beginning, and +/- 15 ms for the end of the saccades. To confirm our results, in a subset of the units the eye position and spike train were digitized, displayed on an oscilloscope, and the beginning and end of each saccade was marked by the user (uncertainty = +/- 1 ms). To express the rate of firing before, during and after saccades, we counted spikes during 100 ms intervals beginning 150 ms before saccade onset, at saccade onset, and at saccade offset, respectively. Results Fifty-one single-unit and multi-unit recordings were obtained from the ION. Twenty-two well resolved single- Unit and nine multi-unit recordings were used for quantitative analysis. Histological reconstruction of electrode penetrations showed that all the recordings were located within the ION (Fig. 1). In general, spikes were biphasic, with a positive-negative shape and durations longer than 700 gsec, as commonly recorded near cell bodies. These facts, along with the fact that all the cells except two presented clear visual responses, make us confident that our data are not significantly contaminated with recordings either from terminals in the nucleus or fibers passing through it. Visual responses To the extent that we could assess, given the presence of the frequent spontaneous eye movements, the cells showed most of the visual response properties previously reported in paralyzed decerebrate chicks (Miles 1972b): cells produced a weak or phasic discharge at the "on" and "off" of light, they responded more vigorously to movements of targets within the receptive fields (Fig. 2, top), and rapid movements were better stimuli than slower ones. Receptive fields were relatively small, with an average diameter of approximately 10 deg. Some units showed habituation to repetitive stimulation, but this was neither so strong nor so frequent a characteristic as reported by Miles in paralyzed decerebrate chicks. A dark square, 10 deg in size, quickly moved across the receptive field, produced a strong response and was used as a search stimulus as we advanced the electrode. Fig. 1. Photomicrograph of a transverse section through the isthmooptic nucleus, stained with cresyl violet, showing a marking lesion (arrow) made 200 lain below the region in which visually responsive neurons were encountered. Abbreviations: ION isthmo-optic nucleus; TeO rectum opticum; Ce cerebellum; Ipc nucleus isthmi, pars magnocellularis. Calibration bar= 500 gm
3 265 Fig. 2A-D. Isthmo-optic cell activity in relation to visual stimulation and saccades. A Visual response to a dark square, 8 degrees of side, moved sinusoidally up and down (peak velocity, 140 deg/s) through its receptive field. Arrows indicate the direction of the movement. Lower trace shows the position of the stimulus. B Activity of the same cell, recorded in darkness, in relation to spon- taneous saccades. Upper trace, horizontal position of the contralateral eye. (In birds, saccades have a prominent 25 Hz oscillation and a duration of 100 to 300 ms). The unit has a variable discharge rate during fixational intervals and pauses during saccades. C, D Activity of a different ION unit recorded in light. Note pausing during saccades In addition to showing responses to our visual stimuli, ION cells showed very irregular activity as the bird looked around the laboratory, with short periods of silence and bursting periods of spikes/second. Some of this irregular activity was probably due to eye- movement-related visual stimulation; in particular the bursting periods resembled the responses elicited by objects moved in the visual field. However, ION cells also discharged in darkness, even at a higher average rate. To assess the average discharge rate, we counted spikes
4 266 during periods of two to three minutes under light and dark conditions. To avoid contamination by saccades we averaged the number of spikes in periods of 100 ms beginning 150 ms before saccade onset. The discharge frequency for a sample of 9 units recorded in darkness was significantly higher (14.3 spikes/s) than of 11 units recorded in light (7.50 spikes/s) (Mann-Whitney U test, p=0.03). Oculomotor responses The most striking characteristic observed in ION cells recorded was that the activity in every unit tended to pause during saccades (Fig. 2), and during quick phases of optokinetic nystagmus. This was especially evident in more active cells, but could be seen in all cells by using peristimulus time histograms. The pauses cannot be explained merely in terms of an interruption of the visual input during saccades, because they also were observed in total darkness. Figure 3 shows raster displays representing the discharge of a cell, recorded both in light and in darkness, in relation to saccades. Because these data include saccades with different durations, raster displays are aligned both with the beginning of the saccades (left side of the figure) and with the end of saccades (right side of the figure). The histograms at the bottom show the activity displayed in the rasters above in bins of 4 ms. In light (Fig. 3A), the discharge of this cell decreased or paused near the onset of the saccades, the discharge was reduced or absent throughout the duration of the saccades and was resumed between 20 ms before and 20 ms after saccade completion. In complete darkness, the cell showed pauses in activity during saccades similar to those recorded in light (Fig. 3B). Comparison of the histograms in light and dark shows that in light the cell has a lower spontaneous discharge rate and shows an overshoot in firing rate after saccade completion. All ION cells recorded in these experiments decreased or interrupted their firing during most saccades. In order to quantify the depth of modulation of the ION activity by saccadic eye movements, we used a "pause index", defined as the percent decrease in firing during saccades relative to the rate before saccades (Fig. 4 legend). Figure 4A shows the distribution of pause index measured in a sample of units in light and in darkness. The firing rate during saccades showed a median decrease across units of 67% (range: 20% to 100%) in light and 72% (range: 40% to 100%) in darkness. This substantial decrement in the activity of ION cells during saccades presumably produces a significant modulation of the sensitivity of retinal neurons in synchrony with eye movements. To further investigate whether visual factors such as the movement of the retinal image contribute to the pause, we compared the depth of modulation by saccades for units tested in both light and dark (Fig. 4B). The median decrease in firing tends to be slightly higher (Mann-Whitney U test p = 0.03) in darkness, suggesting that the pause is not related to vision. (sp/sec) (.:,i,i:,;: I..,,. ', :.(. '~.:.':.,1'., r.,.'.,,.~.,,.,...., '. :..,.~ :~,~,~,,,,,. LIGHT i.-j I. 9,,.h 4,11L,,.I[L I A (sp/sec) Time (msec) "* ',,,~..'..:',,] " ' 'l ''..,'..,. ;,,,, 9,,, 9 '.,,. '.., DARK :.. ii.'. :.""' '..' "..".~':i ',. '~',,',,,'"v,,. ", ~,~,~,.'..,, ~/.' _,._~. ij..._,,,ijd~ul.. i ,2,,:#,,,,,', Time (msee)....,,,,h_~, i_ ~,J i~.i,i Time (msec) Time (msec) 300 Fig. 3A, B. Discharge of a isthmo-optic neuron in relation to spontaneous saccadic eye movements. The unit was recorded while the animal was in light A and in darkness B. Rasters in the left side of A, B are aligned on the onset of saccades; those in the right side are aligned on the end of the saccades. The vertical line extending through the rasters indicates the event used to align the rasters. Each dot in the raster display represents a single action potential, and the cell discharge during consecutive spontaneous saccades are shown as successive lines on the raster. The histograms at the bottom of A, B show the sum of the above rasters in bins of 4 ms. Because consecutive saccades with different durations were sampled together, the histograms shown directly below the rasters in A, B represent the distribution of saccade durations, as indicated by the percentage of saccades (minimum 0%, maximum 100%) ongoing at the indicated time from the synchronizing event. Thus in A (light), all saccades lasted at least 70 ms and none lasted more than 250 ms
5 r..) 5 r~ 0 4 "r 3 2 z 1 g b" < O O ~ ~176 o ~ o 7 ~6 ~ 4 1 A PAUSE INDEX PAUSE INDEX < I i O,260 SACCADE DURATION (msec) Fig. 5. Relation between pause duration and saccade duration. Each symbol represents a different unit. Only pauses starting within 50 ms of the saccade beginning and lasting more than 75 ms were plotted (see text). The pause duration tends to be overestimated because of the bursty firing of the ION neurons, which includes frequent gaps in firing even between saccades. The line shown is the linear regression of pause duration on saccade duration o "~ o o ,.,,, O 100 [3 Pause index in light Fig. 4A, B. Depth of modulation by saccades of discharge of units in the isthmo-optic nucleus. The firing rates were obtained by counting the number of spikes during two 100 ms intervals, one beginning 150 ms before (B) and the other at the onset (D) of saccades respectively. The pause index (100 x [B-D]/B) represents the percent decrease in average firing during saccades relative to the average firing before saccades. A Distribution of pause index measured in light and in darkness for units recorded in one or both conditions. 21 single-unit (filled bars) and 8 multi-unit (hatched bars) recordings were included in these histograms. B Scatter plot of pause index measured in dark and light, for 11 single-unit (open symbols) and 5 multi-unit (filled symbols) recordings made under both conditions The low and irregular activity of ION cells made it difficult to measure the timing of the pauses with respect to the beginning and end of the saccades. However, from histograms like those of Fig. 3, we estimated that the pauses started between 25 ms before and 25 ms after the beginning of the saccades and ended between 35 ms before and 35 ms after the end of the saccades, the median being 0 ms in both cases. To ascertain whether the saccade determined the duration of the pause or simply triggered it, we assessed the relation between saccade duration and pause duration in 104 well-defined saccade-related pauses of 6 ION units. We measured the pause duration as the longest interspike interval greater than 75 ms that began within 50 ms of the saccade beginning. If the adjacent interspike interval was greater than 50 ms, we included it in the pause to prevent single spikes from causing underestimation of the pause duration. We excluded cases in which fewer than 5 spikes occurred within our 600 ms sampling interval. The results show a moderate effect of saccade duration on pause duration (Fig. 5), with a slope of 0.61 (pooled results; for separated units, mean = 0.63, s.e.m. = 0.08) and an intercept of 101 ms (r=0.44, p<0.001). Thus, saccades may trigger a pause of at least 100 ms, with the duration being loosely determined by the saccade duration. The burstiness of the ION discharges and the variability in depth of modulation prevent us from being definitive on this point. As mentioned above, the discharge rate showed a transient increase just after saccades in light but not in dark. To quantify this effect, the ratio between average frequency after and before saccades was measured in a sample of units. As shown in Fig. 6, after saccades in light, units fired a median of 2.3 times the rate before the saccades; in darkness the median of this ratio was 0.9 (difference of the medians was significant, Mann-Whitney U test p<0.001). This relative increase in the discharge rate at the end of saccades observed only in light may represent a wave of excitation coming from the retina, as the visual scene becomes stable after the saccades. This idea is supported by the fact that the increase in firing was usually a discrete burst, beginning ms after the saccade ended. Because, in birds, saccades are frequently associated with blinks of the nictitating membrane, this transient excitation may also reflect the
6 268 J b E Z u~ b E z 4 2 6] 4-2- O I AFTER DISCHARGE RATIO B DARK o ~ AFTER DISCHARGE RATIO Fig. 6A, B. Increase in the firing of ION units at the end of saccades when the bird is in light A but not in dark B. The after-discharge ratio is the ratio between the average firing after saccades and the average firing before saccades, measured at intervals of 100 ms starting 30 ms after the end of saccades and 150 ms before the beggining of saccades respectively. A value grater than one indicates more spikes after the saccade than before it. 21 single-unit (filled bars) and 9 multi-unit (hatched bars) were included in these histograms reappearance of the visual image. For the same reason, the pause in activity could be correlated with blinks rather than saccades. We found, however, that pauses regularly occurred during saccades that were not associated with blinks. Discussion The results presented in this study show that the centrifugal fibers in the chicken optic nerve discharge at reduced rates during saccadic eye movements. This pause in activity is "oculomotor, rather than visual, in origin because it occurs in dark as well as in light and because it generally begins as soon as the saccade does; if the pause were visual in origin a latency of ms (Miles 1972c) would be expected. Finally, the spontaneous activity (between saccades) of the isthmo-optic neurons is higher in dark than in light. Mechanism of saccadic pause Because anatomical studies have shown no prominent local circuits or intrinsic axon collaterals in the ION (Crossland 1979), the pause is probably produced by extranuclear afferents, either the activation of an inhibit- ory signal or the interruption of an excitatory one during saccades. The most likely source of the pause is the input (predominantly excitatory [Holden 1968]) coming from the optic tectum, which is the main source of afferents to the ION. Because in birds, as in mammals, the tectum plays a role in generating spatially oriented eye and head movements (du Lac and Knudsen 1986; Wise et al. 1986), saccade-related signals may be carried by the tectal afferents to the ION. The tectal neurons that project to the ION have been anatomically identified, corresponding to a single layer of cells in the stratum griseum et fibrosum superficialis near the junction of sublaminae h and i (Crossland and Hughes 1978). In the chicken and the quail, their dendritic arbors have long ramifying branches oriented toward deeper layers (Uchiyama and Watanabe 1985; Marin et al. 1988) and shorter branches extending upward, toward lamina g. (In the pigeon, Woodson et al. [1988] reported that dendrites extend to the superficial laminae where the fibers from the retina terminate.) Therefore, tecto-ion neurons might well integrate the input from the retina with oculomotor signals from deeper tectal layers. Another possibility is that the pause results from non-tectal inputs acting upon the ION. In an electron microscopy study, Crossland (1979) has shown that a distinct class of synaptic endings in the ION, constituting more than 30% of the total, remains after removal of the optic tectum. Earlier studies have suggested that afferents may come from the motor nuclei of the extraocular muscles (Huber and Crosby 1929; Agnaut and Reperant 1978). More recent studies, using the HRP tracing technique, have not confirmed this projection (Uchiyama and Watanabe 1985; Marin et al. 1988). More likely sources of these afferents are the contralateral ION, the hyperstriatum accessorium and the nucleus raphe (Woodson et al. 1989). Effect of saccadic pause As mentioned above, the centrifugal system modulates the sensitivity of retinal ganglion cells: electrical stimulation of the isthmo-optic tract increases the responsiveness of retinal ganglion cells to visual stimuli, while cooling it has the opposite effect. Activation of the centrifugal pathway reduces the effectiveness of the inhibitory surround of the ganglion cell receptive fields, and, in some cases, can directly increase the sensitivity of the center (Miles 1972c). The decreased activity of isthmooptic cells during saccades probably induces a decrease in the sensitivity of retinal ganglion cells during that period, just as the cooling of the IO tract has been shown to do. In mammals, a decrease in the transmission of visual information through the geniculo-striate system during saccades has been reported by some workers (Bartlett et al. 1976; Duffy and Burchfiel 1975) but not by others (Buettner and Fuchs 1973; Adey and Noda 1973). In humans, a decrease in visual sensitivity during saccades-saccadic suppression-has been shown psychophysically (Stevenson et al. 1986). Our results suggest that, during saccades, the centrifugal system of birds may
7 269 reduce visual sensitivity at the level of the retina, as suggested by Holden (1966). It may, however, be an oversimplification to describe the action of the centrifugal fibers as retinal saccadic suppression, in view of the evidence suggesting that the centrifugal activity specifically affects the potency of the inhibitory surround of the retinal ganglion cells (Miles 1972c), thereby altering their transfer function. Two types of alterations are plausible: In classical cat or monkey retinal ganglion cells with a concentric centersurround organization, the inhibitory effect of the surround confers contrast sensitivity, but the stronger influence of the center means that the neuron is also sensitive to the overall luminance of its receptive field. In such a neuron, lower centrifugal activity during saccades would increase the influence of the surround, shifting the neuron to being more contrast sensitive and less luminance sensitive. Alternatively, in motion-sensitive ganglion cells, commonly found in the avian retina (Miles 1972a; Pearlman and Hughes 1976a), interactions among receptive-field subunits may confer directional selectivity if activity at one location is delayed and multiplied by that at another, with inhibition among subunits (Reichardt 1987). Analogous to the case just discussed, centrifugal activity might alter the balance between motion- and luminance-selectivity, although electrophysiological studies of retinal ganglion cells have failed to show an effect of ION activity on motion sensitivity (Miles 1972c; Pearlman and Hughes 1976b). In summary, it is possible that decreased centrifugal activity during saccades may make retinal ganglion cells less sensitive but more selective. Function of centrifugal fibers Despite the strong influence that saccades have on the activity of the neurons projecting to the retina, we are not inclined to view the principal function of this projection as being simply saccadic modulation of retinal output, because the anatomical and physiological properties seem too elaborate for so simple a function. First, there is the two-fold retinotopy, whereby individual ION neurons project to the same retinal region from which they receive their visual afference. Second, although we found that the majority of the ION units consistently paused for most saccades, which is compatible with a global modulation mechanism acting upon the entire retina, the activity between saccades was very irregular, and multi-unit recordings showed that the activity of nearby cells was not tightly correlated. Therefore the modulation of retinal ganglion cells between saccades is presumably constantly changing. We speculate that the saccadic modulation of the isthmo-optic nucleus is one of several ways that centrifugal influences on the retinal ganglion cells are exerted. Others may be: (1) Monocular enhancement-in many birds the two eyes generally view rather different scenes. A differential activation of the two IONs might provide an attentional function by changing the relative activity sent to the brain by the two eyes. (A similar hypothesis was advanced by Holden 1966.) For example, a visual feature on the right side of the visual field might lead to a head movement to the right and a simultaneous enhancement of the responsiveness of the right eye. Part of the mechanism of this differential activation of the two IONs might be the mutual inhibiton between the two tecta (Robert and Cuenod 1969) or the commissural projection between the two IONs (Woodson et al. 1989). (2) Local enhancement-alternatively, the centrifugal projections might change the responsiveness of local retinal regions as the bird attends to different parts of the visual field (Holden 1966; Uchiyama 1989). This ability to enhance local retinal regions may be especially useful to birds like chickens, which have a relatively uniform ganglion cell spacing across the retina (Ehrlich 1981). (3) Peripheral enhancement-several lines of evidence suggest that ION activity might bias the visual system toward panoramic vision at the expense of focal or foveal vision. In ground-feeding avian species, which continually alternate between hunting for seeds and scanning for predators (Powell 1974), the ION is larger than in raptors (Shortess and Klose 1975; Weidner et al. 1987), and projects mostly to extrafoveate retina (Hayes and Holden 1983). ION-lesioned chicks are less responsive to moving peripheral targets while feeding (Rogers and Miles 1972). In addition, ION neurons adapt strongly to repetitive stimulation (Miles 1972b), a property appropiate to a warning function. Finally, ION neurons in chicks accumulate androgens (Meyer et al. 1976) and androgens cause chicks to perseverate at pecking (Andrew 1972); perhaps they do so by modulating ION function. (4) Dark-adaptation-The higher spontaneous ION activity observed in darkness presumably increases the size of receptive fields by suppressing the inhibitory surrounds (Miles 1972c; Pearlman and Hughes 1976b), similar to what has been observed in cat ganglion cells during dark adaptation (Barlow et al. 1957); this may increase the retinal sensitivity. Thus, the increase in ION activity in darkness may represent a centrifugal contribution to dark adaptation. This role for the centrifugal projection to the retina is well established in arthropods (Fleissner and Fleissner 1988). In conclusion, the decrease in firing of centrifugal neurons during saccades may be one example of the ways in which the ION modulates the sensitivity of retinal ganglion cells. This simple coupling between the centrifugal and the oculomotor systems may give insight into other more complex interactions, such as dynamic changes in visual attention coordinated with changes in gaze. Acknowledgements. We are grateful to F.A. Miles for reading this manuscript. This work was supported by NSF grant BNS and NEI grant 1 R15 EY References Adey WR, Noda H (1973) Influence of eye movements on geniculostriate excitability in the cat. J Physiol (Lond) 235: Agnaut P, Reperant J (1978) A light and electron microscopic study of the nucleus isthmo-optieus in the pigeon. Arch Anat Microsc Morphol 67:63-78 Andrew RJ (1972) Recognition processes and behavior, with special reference to effects of testosterone on persistence. In: Lehrman
8 270 DS, Hinde RA, Shaw E (eds) Advances in the study of behavior, Vol 4. Academic Press, New York London, pp Barlow H B, Fitzhugh R, Kuffier SW (1957) Change of organization in the receptive fields of the cat's retina during dark adaptation. J Physiol (Lond) 137: Bartlett JR, Doty RW, Lee BB, Sakakura H (1976) Influence of saccadic eye movements on geniculostriate excitability in normal monkeys. Exp Brain Res 25: Buettner U, Fuchs AF (1973) Influence of saccadic eye movements on unit activity in simian lateral geniculate and pregeniculate nuclei. J Neurophysiol 36: Cowan WM (1970) Centrifugal fibers to the avian retina. Br Med Bull 26: Crossland WJ (1979) Identification of the tectal synaptic terminals in the avian isthmo-optic nucleus. In: Granda AM, Maxwell JW (eds) Neural mechanisms of behavior in the pigeon. Plenum Press, New York, pp Crossland WJ, Hughes CP (1978) Observations on the afferent and efferent connections of the avian isthmo-optic nucleus. Brain Res 145: Dowling JE, Cowan WM (1966) An electron microscopic study of normal and degenerating centrifugal fiber terminals in the pigeon retina. Z Zellforsch 71 : Duffy FH, Burchfiel JL (1975) Eye movement-related inhibition of primate visual neurons. Brain Res 89 : du Lac S, Knudsen El (1986) A map of head movements in barn owls is revealed by electrical stimulation of the optic tectum. Soc Neurosci Abstr 12(2) : 1185 Ehrlich D (1981) Regional specialization of the chick retina as revealed by the size and density of neurons in the ganglion cell layer. J Comp Neurol 195: Fleissner G, Fleissner G (1988) Efferent control of visual sensitivity in arthropod eyes: with emphasis on circadian rythms. In: Information processing in animals, Vol 5. Gustav Fischer Verlag, Stuttgart New York Goldberg JM, Fernandez C (1980) Efferent vestibular system in the squirrel monkey: anatomical location and influence on afferent activity. J Neurophysiol 43: Hayes BP, Holden AL (1983) The distribution of centrifugal terminals in the pigeon retina. Exp Brain Res 49: Holden AL (1966) Two possible visual functions for centrifugal fibers to the retina. Nature 212: Holden AL (1968) The centrifugal system running to the pigeon retina. J Physiol (Lond) 197: Holden AL (1978) Centrifugal actions on pigeon retinal ganglion cells. J Physiol (Lond) 282:8P-9P Huber GC, Crosby EC (1929) The nuclei and fiber paths of the avian diencephalon, with consideration of telenchephalic and certain mesencephalic centers and connections. J Comp Neurol 48 : Knipling RR, (1978) No deficit in near field visual acuity after transection of the isthmo-optic tract. Physiol Behav 21: Letelier JC, Wallman J, Matin G (1985) Centrifugal fibers of the optic nerve carry eye movement signals. Soc Neurosci Abstr 11(2): 1043 Marin G, Bodnarenko S, McKenna O, Wallman J (1988) Neurons projecting to the retina: afferents and possible functions. Soc Neurosci Abstr 14(2): 992 Matthews, PBC (1982) Evolving views on the muscle spindle. J Physiol (Lond) 320:1-30 Maturana HR, Frenk S (1965) Synaptic connections of the centrifugal fibers in the pigeon retina. Science 100: McGill JI, Powell TPS, Cowan WM (1966a) The retinal representation upon the optic tectum and the isthmo-optic nucleus in the pigeon. J Anat 100:5-33 McGill JI, Powell TPS, Cowan WM (1966b) The organization of the projection of the centrifugal fibers to the retina of the pigeon. J Anat 100:35 49 Merrill EG, Ainsworth A (1972) Glass-coated platinum plated tungsten microelectrodes. Med Biol Eng 10: Meyer CC, Parker DM, Salsen EA (1976) Androgen-sensitive midbrain sites and visual attention in chicks. Nature 259: Miles FA (1972a) Centrifugal control of avian retina. I. Receptive field properties of retinal ganglion cells. Brain Res 48 : Miles FA (1972b) Centrifugal control of the avian retina. II. Receptive field properties of the cells in the isthmo-optic nucleus. Brain Res 48 : Miles FA (1972c) Centrifugal control of the avian retina. III. Effects of electrical stimulation of the isthmo-optic tract on the receptive field properties of retinal ganglion cells. Brain Res 48: Miles FA (1972d) Centrifugal control of the avian retina. IV. Effects of reversible cold block of the isthmo-optic tract on the receptive field properties of cells in the retina and isthmo-optic nucleus. Brain Res 48: Pearlman AL, Hughes CP (1976a) Functional role of efferents to the avian retina. I. Analysis of retinal ganglion cell receptive fields. J Comp Neurol 166: Pearlman AL, Hughes CP (1976b) Functional role of efferents to the avian retina. II. Effects of reversible cooling of the isthmooptic nucleus. J Comp Neurol 166: Powell GVN (1974) Experimental analysis of the social value of the flocking by starlings (Sturnus vulgar&) in relation to predation and foraging. Anim Behav 22: Reichardt W (1987) Evaluation of optical motion information by movement detectors. J Comp Physiol (A) 161: Robert F, Cuenod M (1969) Electrophysiology of the intertectal commissures in the pigeon. II. Inhibitory interaction. Exp Brain Res 9: Rogers L J, Miles FA (1972) Centrifugal control of the avian retina. V. Effects of lesions of the isthmo-optic nucleus on visual behavior. Brain Res 48: Russell IJ, Roberts BL (1974) Active reduction of lateral-line sen- 9 sitivity in swimming dogfish. J Comp Physiol 94:7-15 Shortess GK, Klose EF (1975) The area of the nucleus isthmoopticus in the american kestrel (Falco sparverius) and the redtailed hawk (Buteo jamaicensis). Brain Res 88 : Shortess GK, Klose EF (1977) Effects of lesions involving efferent fibers to the retina in pigeons (Columba livia). Physiol Behav 18: Stevenson SB, Volkmann FC, Kelly JP, Riggs LA (1986) Dependence of visual suppression on the amplitude of saccades and blinks. Vision Res 26: Uchiyama H (1989) Centrifugal pathways to the retina: influence of the optic tectum. Visual Neurosci 3: Uchiyama H, Watanabe M (1985) Tectal neurons projecting to the isthmo-optic nucleus in the japanese quail. Neurosci Lett 58: Wallman J, Velez J, Weinstein B, Green AE (1982) Avian vestibuloocular reflex: adaptative plasticity and developmental changes. J Neurophysiol 48 : Weidner C, Reperant J, Desroches AM, Miceli D, Vesselkin NP (1987) Nuclear origin of the centrifugal visual pathway in birds of prey. Brain Res 436: Wise LZ, Letelier JC, Wallman J (1986) Saccadic eye movements evoked by microstimulation of optic tectum of the chicken. Soc Neurosci Abstr 12(2): 1185 Wolf-Oberhollenzer F (1987) A study of the centrifugal projections to the pigeon retina using two fluorescent markers. Neurosci Lett 73:16-20 Woodson W, Shimizu T, Karten HJ (1988) Afferent and efferent connections of the isthmo-optic nucleus in the pigeon (Columba livia). Soc Neurosci Abstr 14(2): 1121 Woodson W, Shimizu T, Karten HJ (1989) Transmitter and peptide content of the isthmo-optic nucleus in the pigeon (Colurnba livia). Soc Neurosci Abstr 15(2): 459
GAZE STABILIZATION SYSTEMS Vestibular Ocular Reflex (VOR) Purpose of VOR Chief function is to stabilize gaze during locomotion. Acuity declines if
 GAZE STABILIZATION SYSTEMS Vestibular Ocular Reflex (VOR) Purpose of VOR Chief function is to stabilize gaze during locomotion. Acuity declines if slip exceeds 3-5 deg/sec. Ex: Head bobbing and heel strike
GAZE STABILIZATION SYSTEMS Vestibular Ocular Reflex (VOR) Purpose of VOR Chief function is to stabilize gaze during locomotion. Acuity declines if slip exceeds 3-5 deg/sec. Ex: Head bobbing and heel strike
Origin of Electrical Membrane Potential
 Origin of Electrical Membrane Potential parti This book is about the physiological characteristics of nerve and muscle cells. As we shall see, the ability of these cells to generate and conduct electricity
Origin of Electrical Membrane Potential parti This book is about the physiological characteristics of nerve and muscle cells. As we shall see, the ability of these cells to generate and conduct electricity
Laboratory Guide. Anatomy and Physiology
 Laboratory Guide Anatomy and Physiology TBME04, Fall 2010 Name: Passed: Last updated 2010-08-13 Department of Biomedical Engineering Linköpings Universitet Introduction This laboratory session is intended
Laboratory Guide Anatomy and Physiology TBME04, Fall 2010 Name: Passed: Last updated 2010-08-13 Department of Biomedical Engineering Linköpings Universitet Introduction This laboratory session is intended
The Visual Cortex 0 http://www.tutis.ca/neuromd/index.htm 20 February 2013
 T he Visual Cortex 0 Chapter contents Contents Chapter 2... 0 T he Visual Cortex... 0 Chapter Contents... 1 Introduction... 2 Optic Chiasm... 2 Where do the eye's ganglion cells project to?... 3 To where
T he Visual Cortex 0 Chapter contents Contents Chapter 2... 0 T he Visual Cortex... 0 Chapter Contents... 1 Introduction... 2 Optic Chiasm... 2 Where do the eye's ganglion cells project to?... 3 To where
Chapter 13. The Nature of Somatic Reflexes
 Chapter 13 The Nature of Somatic Reflexes Nature of Reflexes (1 of 3) A reflex is an involuntary responses initiated by a sensory input resulting in a change in a gland or muscle tissue occur without our
Chapter 13 The Nature of Somatic Reflexes Nature of Reflexes (1 of 3) A reflex is an involuntary responses initiated by a sensory input resulting in a change in a gland or muscle tissue occur without our
Chapter 7: The Nervous System
 Chapter 7: The Nervous System Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways involved in a withdraw reflex Define
Chapter 7: The Nervous System Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways involved in a withdraw reflex Define
Processing the Image or Can you Believe what you see? Light and Color for Nonscientists PHYS 1230
 Processing the Image or Can you Believe what you see? Light and Color for Nonscientists PHYS 1230 Optical Illusions http://www.michaelbach.de/ot/mot_mib/index.html Vision We construct images unconsciously
Processing the Image or Can you Believe what you see? Light and Color for Nonscientists PHYS 1230 Optical Illusions http://www.michaelbach.de/ot/mot_mib/index.html Vision We construct images unconsciously
Directions for construction used with permission from Pacific Science Center - Brain Power
 Directions for construction used with permission from Pacific Science Center - Brain Power The Vestibular System The vestibular system within the inner ear detects both the position and motion of the head
Directions for construction used with permission from Pacific Science Center - Brain Power The Vestibular System The vestibular system within the inner ear detects both the position and motion of the head
Activity 5: The Action Potential: Measuring Its Absolute and Relative Refractory Periods. 250 20 Yes. 125 20 Yes. 60 20 No. 60 25 No.
 3: Neurophysiology of Nerve Impulses (Part 2) Activity 5: The Action Potential: Measuring Its Absolute and Relative Refractory Periods Interval between stimuli Stimulus voltage (mv) Second action potential?
3: Neurophysiology of Nerve Impulses (Part 2) Activity 5: The Action Potential: Measuring Its Absolute and Relative Refractory Periods Interval between stimuli Stimulus voltage (mv) Second action potential?
Masters research projects. 1. Adapting Granger causality for use on EEG data.
 Masters research projects 1. Adapting Granger causality for use on EEG data. Background. Granger causality is a concept introduced in the field of economy to determine which variables influence, or cause,
Masters research projects 1. Adapting Granger causality for use on EEG data. Background. Granger causality is a concept introduced in the field of economy to determine which variables influence, or cause,
CHAPTER 6 PRINCIPLES OF NEURAL CIRCUITS.
 CHAPTER 6 PRINCIPLES OF NEURAL CIRCUITS. 6.1. CONNECTIONS AMONG NEURONS Neurons are interconnected with one another to form circuits, much as electronic components are wired together to form a functional
CHAPTER 6 PRINCIPLES OF NEURAL CIRCUITS. 6.1. CONNECTIONS AMONG NEURONS Neurons are interconnected with one another to form circuits, much as electronic components are wired together to form a functional
Video-Based Eye Tracking
 Video-Based Eye Tracking Our Experience with Advanced Stimuli Design for Eye Tracking Software A. RUFA, a G.L. MARIOTTINI, b D. PRATTICHIZZO, b D. ALESSANDRINI, b A. VICINO, b AND A. FEDERICO a a Department
Video-Based Eye Tracking Our Experience with Advanced Stimuli Design for Eye Tracking Software A. RUFA, a G.L. MARIOTTINI, b D. PRATTICHIZZO, b D. ALESSANDRINI, b A. VICINO, b AND A. FEDERICO a a Department
Nerves and Nerve Impulse
 Nerves and Nerve Impulse Terms Absolute refractory period: Period following stimulation during which no additional action potential can be evoked. Acetylcholine: Chemical transmitter substance released
Nerves and Nerve Impulse Terms Absolute refractory period: Period following stimulation during which no additional action potential can be evoked. Acetylcholine: Chemical transmitter substance released
Standards Alignment Minnesota Science Standards Alignment Matrix www.brainu.org/resources/mnstds
 Lesson Summary: Neurons transfer information by releasing neurotransmitters across the synapse or space between neurons. Students model the chemical communication between pre-synaptic and post-synaptic
Lesson Summary: Neurons transfer information by releasing neurotransmitters across the synapse or space between neurons. Students model the chemical communication between pre-synaptic and post-synaptic
The Physiology of the Senses Lecture 11 - Eye Movements www.tutis.ca/senses/
 The Physiology of the Senses Lecture 11 - Eye Movements www.tutis.ca/senses/ Contents Objectives... 2 Introduction... 2 The 5 Types of Eye Movements... 2 The eyes are rotated by 6 extraocular muscles....
The Physiology of the Senses Lecture 11 - Eye Movements www.tutis.ca/senses/ Contents Objectives... 2 Introduction... 2 The 5 Types of Eye Movements... 2 The eyes are rotated by 6 extraocular muscles....
EXCITABILITY & ACTION POTENTIALS page 1
 page 1 INTRODUCTION A. Excitable Tissue: able to generate Action Potentials (APs) (e.g. neurons, muscle cells) B. Neurons (nerve cells) a. components 1) soma (cell body): metabolic center (vital, always
page 1 INTRODUCTION A. Excitable Tissue: able to generate Action Potentials (APs) (e.g. neurons, muscle cells) B. Neurons (nerve cells) a. components 1) soma (cell body): metabolic center (vital, always
Action Potentials I Generation. Reading: BCP Chapter 4
 Action Potentials I Generation Reading: BCP Chapter 4 Action Potentials Action potentials (AP s) aka Spikes (because of how they look in an electrical recording of Vm over time). Discharges (descriptive
Action Potentials I Generation Reading: BCP Chapter 4 Action Potentials Action potentials (AP s) aka Spikes (because of how they look in an electrical recording of Vm over time). Discharges (descriptive
Name Date Hour. Nerve Histology Microscope Lab
 Name Date Hour Nerve Histology Microscope Lab PRE-LAB: Answer the following questions using your reading and class notes before starting the microscope lab. 1. What is the difference between the functions
Name Date Hour Nerve Histology Microscope Lab PRE-LAB: Answer the following questions using your reading and class notes before starting the microscope lab. 1. What is the difference between the functions
E. K. A. ADVANCED PHYSICS LABORATORY PHYSICS 3081, 4051 NUCLEAR MAGNETIC RESONANCE
 E. K. A. ADVANCED PHYSICS LABORATORY PHYSICS 3081, 4051 NUCLEAR MAGNETIC RESONANCE References for Nuclear Magnetic Resonance 1. Slichter, Principles of Magnetic Resonance, Harper and Row, 1963. chapter
E. K. A. ADVANCED PHYSICS LABORATORY PHYSICS 3081, 4051 NUCLEAR MAGNETIC RESONANCE References for Nuclear Magnetic Resonance 1. Slichter, Principles of Magnetic Resonance, Harper and Row, 1963. chapter
Resting membrane potential ~ -70mV - Membrane is polarized
 Resting membrane potential ~ -70mV - Membrane is polarized (ie) Electrical charge on the outside of the membrane is positive while the electrical charge on the inside of the membrane is negative Changes
Resting membrane potential ~ -70mV - Membrane is polarized (ie) Electrical charge on the outside of the membrane is positive while the electrical charge on the inside of the membrane is negative Changes
REVIEW SHEET EXERCISE 3 Neurophysiology of Nerve Impulses Name Lab Time/Date. The Resting Membrane Potential
 REVIEW SHEET EXERCISE 3 Neurophysiology of Nerve Impulses Name Lab Time/Date ACTIVITY 1 The Resting Membrane Potential 1. Explain why increasing extracellular K + reduces the net diffusion of K + out of
REVIEW SHEET EXERCISE 3 Neurophysiology of Nerve Impulses Name Lab Time/Date ACTIVITY 1 The Resting Membrane Potential 1. Explain why increasing extracellular K + reduces the net diffusion of K + out of
What role does the nucleolus have in cell functioning? Glial cells
 Nervous System Lab The nervous system of vertebrates can be divided into the central nervous system, which consists of the brain and spinal cord, and the peripheral nervous system, which contains nerves,
Nervous System Lab The nervous system of vertebrates can be divided into the central nervous system, which consists of the brain and spinal cord, and the peripheral nervous system, which contains nerves,
Biology Slide 1 of 38
 Biology 1 of 38 2 of 38 35-2 The Nervous System What are the functions of the nervous system? 3 of 38 35-2 The Nervous System 1. Nervous system: a. controls and coordinates functions throughout the body
Biology 1 of 38 2 of 38 35-2 The Nervous System What are the functions of the nervous system? 3 of 38 35-2 The Nervous System 1. Nervous system: a. controls and coordinates functions throughout the body
The Physiology of the Senses Lecture 10 - Balance www.tutis.ca/senses/
 The Physiology of the Senses Lecture 10 - Balance www.tutis.ca/senses/ Contents Objectives... 1 The sense of balance originates in the labyrinth.... 2 The auditory and vestibular systems have a common
The Physiology of the Senses Lecture 10 - Balance www.tutis.ca/senses/ Contents Objectives... 1 The sense of balance originates in the labyrinth.... 2 The auditory and vestibular systems have a common
Cerebellum and Basal Ganglia
 Cerebellum and Basal Ganglia 1 Contents Cerebellum and Basal Ganglia... 1 Introduction... 3 A brief review of cerebellar anatomy... 4 Basic Circuit... 4 Parallel and climbing fiber input has a very different
Cerebellum and Basal Ganglia 1 Contents Cerebellum and Basal Ganglia... 1 Introduction... 3 A brief review of cerebellar anatomy... 4 Basic Circuit... 4 Parallel and climbing fiber input has a very different
Visual area MT responds to local motion. Visual area MST responds to optic flow. Visual area STS responds to biological motion. Macaque visual areas
 Visual area responds to local motion MST a Visual area MST responds to optic flow MST a Visual area STS responds to biological motion STS Macaque visual areas Flattening the brain What is a visual area?
Visual area responds to local motion MST a Visual area MST responds to optic flow MST a Visual area STS responds to biological motion STS Macaque visual areas Flattening the brain What is a visual area?
Auditory neuroanatomy: the Spanish heritage. Santiago Ramón y Cajal, 1852 1934
 Auditory neuroanatomy: the Spanish heritage Santiago Ramón y Cajal, 1852 1934 Rafael Lorente de Nó, 1902 1990 3 The nervous system is made up of cells. Estimates of the number of cells vary from
Auditory neuroanatomy: the Spanish heritage Santiago Ramón y Cajal, 1852 1934 Rafael Lorente de Nó, 1902 1990 3 The nervous system is made up of cells. Estimates of the number of cells vary from
Physiological Basis of the BOLD Signal. Kerstin Preuschoff Social and Neural systems Lab University of Zurich
 Physiological Basis of the BOLD Signal Kerstin Preuschoff Social and Neural systems Lab University of Zurich Source: Arthurs & Boniface, 2002 From Stimulus to Bold Overview Physics of BOLD signal - Magnetic
Physiological Basis of the BOLD Signal Kerstin Preuschoff Social and Neural systems Lab University of Zurich Source: Arthurs & Boniface, 2002 From Stimulus to Bold Overview Physics of BOLD signal - Magnetic
Bi 360: Midterm Review
 Bi 360: Midterm Review Basic Neurobiology 1) Many axons are surrounded by a fatty insulating sheath called myelin, which is interrupted at regular intervals at the Nodes of Ranvier, where the action potential
Bi 360: Midterm Review Basic Neurobiology 1) Many axons are surrounded by a fatty insulating sheath called myelin, which is interrupted at regular intervals at the Nodes of Ranvier, where the action potential
EXPERIMENT NUMBER 5 BASIC OSCILLOSCOPE OPERATIONS
 1 EXPERIMENT NUMBER 5 BASIC OSCILLOSCOPE OPERATIONS The oscilloscope is the most versatile and most important tool in this lab and is probably the best tool an electrical engineer uses. This outline guides
1 EXPERIMENT NUMBER 5 BASIC OSCILLOSCOPE OPERATIONS The oscilloscope is the most versatile and most important tool in this lab and is probably the best tool an electrical engineer uses. This outline guides
Parts of the Nerve Cell and Their Functions
 Parts of the Nerve Cell and Their Functions Silvia Helena Cardoso, PhD [ 1. Cell body] [2. Neuronal membrane] [3. Dendrites] [4. Axon] [5. Nerve ending] 1. Cell body The cell body (soma) is the factory
Parts of the Nerve Cell and Their Functions Silvia Helena Cardoso, PhD [ 1. Cell body] [2. Neuronal membrane] [3. Dendrites] [4. Axon] [5. Nerve ending] 1. Cell body The cell body (soma) is the factory
Programs for diagnosis and therapy of visual field deficits in vision rehabilitation
 Spatial Vision, vol. 10, No.4, pp. 499-503 (1997) Programs for diagnosis and therapy of visual field deficits in vision rehabilitation Erich Kasten*, Hans Strasburger & Bernhard A. Sabel Institut für Medizinische
Spatial Vision, vol. 10, No.4, pp. 499-503 (1997) Programs for diagnosis and therapy of visual field deficits in vision rehabilitation Erich Kasten*, Hans Strasburger & Bernhard A. Sabel Institut für Medizinische
How to test ocular movements in PSP Jan Kassubek
 How to test ocular movements in PSP Jan Kassubek Universitätsklinik für Neurologie, Ulm Bedside Screening: PSP initially slowing of vertical saccades slowing of downward saccades is considered the hallmark
How to test ocular movements in PSP Jan Kassubek Universitätsklinik für Neurologie, Ulm Bedside Screening: PSP initially slowing of vertical saccades slowing of downward saccades is considered the hallmark
Anatomy Review Graphics are used with permission of: adam.com (http://www.adam.com/) Benjamin Cummings Publishing Co (http://www.awl.com/bc).
 Page 1. Introduction The structure of neurons reflects their function. One part of the cell receives incoming signals. Another part generates outgoing signals. Anatomy Review Graphics are used with permission
Page 1. Introduction The structure of neurons reflects their function. One part of the cell receives incoming signals. Another part generates outgoing signals. Anatomy Review Graphics are used with permission
Functional neuroimaging. Imaging brain function in real time (not just the structure of the brain).
 Functional neuroimaging Imaging brain function in real time (not just the structure of the brain). The brain is bloody & electric Blood increase in neuronal activity increase in metabolic demand for glucose
Functional neuroimaging Imaging brain function in real time (not just the structure of the brain). The brain is bloody & electric Blood increase in neuronal activity increase in metabolic demand for glucose
CHAPTER 5 SIGNALLING IN NEURONS
 5.1. SYNAPTIC TRANSMISSION CHAPTER 5 SIGNALLING IN NEURONS One of the main functions of neurons is to communicate with other neurons. An individual neuron may receive information from many different sources.
5.1. SYNAPTIC TRANSMISSION CHAPTER 5 SIGNALLING IN NEURONS One of the main functions of neurons is to communicate with other neurons. An individual neuron may receive information from many different sources.
Introduction to Psychology, 7th Edition, Rod Plotnik Module 3: Brain s Building Blocks. Module 3. Brain s Building Blocks
 Module 3 Brain s Building Blocks Structure of the Brain Genes chains of chemicals that are arranged like rungs on a twisting ladder there are about 100,000 genes that contain chemical instructions that
Module 3 Brain s Building Blocks Structure of the Brain Genes chains of chemicals that are arranged like rungs on a twisting ladder there are about 100,000 genes that contain chemical instructions that
207-2. Selective Saccadic Palsy
 207-2 Selective Saccadic Palsy Selective Saccadic Palsy after Cardiac Surgery Selective loss of all forms of saccades (voluntary and reflexive quick phases of nystagmus) with sparing of other eye movements.
207-2 Selective Saccadic Palsy Selective Saccadic Palsy after Cardiac Surgery Selective loss of all forms of saccades (voluntary and reflexive quick phases of nystagmus) with sparing of other eye movements.
Brain Computer Interfaces (BCI) Communication Training of brain activity
 Brain Computer Interfaces (BCI) Communication Training of brain activity Brain Computer Interfaces (BCI) picture rights: Gerwin Schalk, Wadsworth Center, NY Components of a Brain Computer Interface Applications
Brain Computer Interfaces (BCI) Communication Training of brain activity Brain Computer Interfaces (BCI) picture rights: Gerwin Schalk, Wadsworth Center, NY Components of a Brain Computer Interface Applications
Second Quarterly Progress Report NO1-DC-6-2111 The Neurophysiological Effects of Simulated Auditory Prosthesis Stimulation
 Second Quarterly Progress Report NO1-DC-6-2111 The Neurophysiological Effects of Simulated Auditory Prosthesis Stimulation J.T. Rubinstein, A.J. Matsuoka, P.J. Abbas, and C.A. Miller Department of Otolaryngology
Second Quarterly Progress Report NO1-DC-6-2111 The Neurophysiological Effects of Simulated Auditory Prosthesis Stimulation J.T. Rubinstein, A.J. Matsuoka, P.J. Abbas, and C.A. Miller Department of Otolaryngology
DIODE CIRCUITS LABORATORY. Fig. 8.1a Fig 8.1b
 DIODE CIRCUITS LABORATORY A solid state diode consists of a junction of either dissimilar semiconductors (pn junction diode) or a metal and a semiconductor (Schottky barrier diode). Regardless of the type,
DIODE CIRCUITS LABORATORY A solid state diode consists of a junction of either dissimilar semiconductors (pn junction diode) or a metal and a semiconductor (Schottky barrier diode). Regardless of the type,
Reflex Physiology. Dr. Ali Ebneshahidi. 2009 Ebneshahidi
 Reflex Physiology Dr. Ali Ebneshahidi Reflex Physiology Reflexes are automatic, subconscious response to changes within or outside the body. a. Reflexes maintain homeostasis (autonomic reflexes) heart
Reflex Physiology Dr. Ali Ebneshahidi Reflex Physiology Reflexes are automatic, subconscious response to changes within or outside the body. a. Reflexes maintain homeostasis (autonomic reflexes) heart
Internuclear ophthalmoplegia: recovery and plasticity
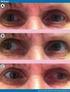 Internuclear ophthalmoplegia: recovery and plasticity M. J. Doslak, L. B. Kline, L. F. Dell'Osso, and R. B. Daroff We studied refixational eye movements of a patient during the gradual resolution of an
Internuclear ophthalmoplegia: recovery and plasticity M. J. Doslak, L. B. Kline, L. F. Dell'Osso, and R. B. Daroff We studied refixational eye movements of a patient during the gradual resolution of an
Electroneuromyographic studies
 Electroneuromyographic studies in the diagnosis of Pudendal Entrapment Syndrome BY NAGLAA ALI GADALLAH PROFESSOR OF PHYSICAL MEDICINE, RHEUMATOLOGY& REHABILITATION AIN SHAMS UNIVERSITY Pudendal neuralgia
Electroneuromyographic studies in the diagnosis of Pudendal Entrapment Syndrome BY NAGLAA ALI GADALLAH PROFESSOR OF PHYSICAL MEDICINE, RHEUMATOLOGY& REHABILITATION AIN SHAMS UNIVERSITY Pudendal neuralgia
ELECTRICAL ACTIVITY IN THE RADIAL NERVE CORD AND AMPULLAE OF SEA URCHINS*
 J. Exp. Biol. (1965), 43, 247-256 247 With 6 text-figures Printed in Great Britain ELECTRICAL ACTIVITY IN THE RADIAL NERVE CORD AND AMPULLAE OF SEA URCHINS* BY D. C. SANDEMANf Department of Zoology, University
J. Exp. Biol. (1965), 43, 247-256 247 With 6 text-figures Printed in Great Britain ELECTRICAL ACTIVITY IN THE RADIAL NERVE CORD AND AMPULLAE OF SEA URCHINS* BY D. C. SANDEMANf Department of Zoology, University
BIOPHYSICS OF NERVE CELLS & NETWORKS
 UNIVERSITY OF LONDON MSci EXAMINATION May 2007 for Internal Students of Imperial College of Science, Technology and Medicine This paper is also taken for the relevant Examination for the Associateship
UNIVERSITY OF LONDON MSci EXAMINATION May 2007 for Internal Students of Imperial College of Science, Technology and Medicine This paper is also taken for the relevant Examination for the Associateship
Neurophysiology. 2.1 Equilibrium Potential
 2 Neurophysiology 2.1 Equilibrium Potential An understanding of the concepts of electrical and chemical forces that act on ions, electrochemical equilibrium, and equilibrium potential is a powerful tool
2 Neurophysiology 2.1 Equilibrium Potential An understanding of the concepts of electrical and chemical forces that act on ions, electrochemical equilibrium, and equilibrium potential is a powerful tool
E/M Experiment: Electrons in a Magnetic Field.
 E/M Experiment: Electrons in a Magnetic Field. PRE-LAB You will be doing this experiment before we cover the relevant material in class. But there are only two fundamental concepts that you need to understand.
E/M Experiment: Electrons in a Magnetic Field. PRE-LAB You will be doing this experiment before we cover the relevant material in class. But there are only two fundamental concepts that you need to understand.
An Introduction to ERP Studies of Attention
 An Introduction to ERP Studies of Attention Logan Trujillo, Ph.D. Post-Doctoral Fellow University of Texas at Austin Cognitive Science Course, Fall 2008 What is Attention? Everyone knows what attention
An Introduction to ERP Studies of Attention Logan Trujillo, Ph.D. Post-Doctoral Fellow University of Texas at Austin Cognitive Science Course, Fall 2008 What is Attention? Everyone knows what attention
Nerve Cell Communication
 Nerve Cell Communication Core Concept: Nerve cells communicate using electrical and chemical signals. Class time required: Approximately 2 forty minute class periods Teacher Provides: For each student
Nerve Cell Communication Core Concept: Nerve cells communicate using electrical and chemical signals. Class time required: Approximately 2 forty minute class periods Teacher Provides: For each student
Reduction of the flash-lag effect in terms of active observation
 Attention, Perception, & Psychophysics 2010, 72 (4), 1032-1044 doi:10.3758/app.72.4.1032 Reduction of the flash-lag effect in terms of active observation MAKOTO ICHIKAWA Chiba University, Chiba, Japan
Attention, Perception, & Psychophysics 2010, 72 (4), 1032-1044 doi:10.3758/app.72.4.1032 Reduction of the flash-lag effect in terms of active observation MAKOTO ICHIKAWA Chiba University, Chiba, Japan
Axonal degeneration as a result of in vitro Mercury exposure
 Axonal degeneration as a result of in vitro Mercury exposure Jenifer DeNormandie Neurobiology Short Report Bio 324 / Neurobiology Wheaton College, Norton, Massachusetts, USA 11/30/11 Introduction: Neural
Axonal degeneration as a result of in vitro Mercury exposure Jenifer DeNormandie Neurobiology Short Report Bio 324 / Neurobiology Wheaton College, Norton, Massachusetts, USA 11/30/11 Introduction: Neural
6/2016 E&M forces-1/8 ELECTRIC AND MAGNETIC FORCES. PURPOSE: To study the deflection of a beam of electrons by electric and magnetic fields.
 6/016 E&M forces-1/8 ELECTRIC AND MAGNETIC FORCES PURPOSE: To study the deflection of a beam of electrons by electric and magnetic fields. APPARATUS: Electron beam tube, stand with coils, power supply,
6/016 E&M forces-1/8 ELECTRIC AND MAGNETIC FORCES PURPOSE: To study the deflection of a beam of electrons by electric and magnetic fields. APPARATUS: Electron beam tube, stand with coils, power supply,
Module 13 : Measurements on Fiber Optic Systems
 Module 13 : Measurements on Fiber Optic Systems Lecture : Measurements on Fiber Optic Systems Objectives In this lecture you will learn the following Measurements on Fiber Optic Systems Attenuation (Loss)
Module 13 : Measurements on Fiber Optic Systems Lecture : Measurements on Fiber Optic Systems Objectives In this lecture you will learn the following Measurements on Fiber Optic Systems Attenuation (Loss)
NUCLEAR MAGNETIC RESONANCE. Advanced Laboratory, Physics 407, University of Wisconsin Madison, Wisconsin 53706
 (revised 4/21/03) NUCLEAR MAGNETIC RESONANCE Advanced Laboratory, Physics 407, University of Wisconsin Madison, Wisconsin 53706 Abstract This experiment studies the Nuclear Magnetic Resonance of protons
(revised 4/21/03) NUCLEAR MAGNETIC RESONANCE Advanced Laboratory, Physics 407, University of Wisconsin Madison, Wisconsin 53706 Abstract This experiment studies the Nuclear Magnetic Resonance of protons
Correlated Neuronal Response: Time Scales and Mechanisms
 Correlated Neuronal Response: Time Scales and Mechanisms Wyeth Bair Howard Hughes Medical nst. NYU Center for Neural Science 4 Washington P., Room 809 New York, NY 10003 Ehud Zohary Dept. of Neurobiology
Correlated Neuronal Response: Time Scales and Mechanisms Wyeth Bair Howard Hughes Medical nst. NYU Center for Neural Science 4 Washington P., Room 809 New York, NY 10003 Ehud Zohary Dept. of Neurobiology
THE HUMAN BRAIN. observations and foundations
 THE HUMAN BRAIN observations and foundations brains versus computers a typical brain contains something like 100 billion miniscule cells called neurons estimates go from about 50 billion to as many as
THE HUMAN BRAIN observations and foundations brains versus computers a typical brain contains something like 100 billion miniscule cells called neurons estimates go from about 50 billion to as many as
How To Understand The Effects Of Saccades On The Visual Cortex In Macaque Monkeys
 Exp Brain Res (1998) 123:341 345 Springer-Verlag 1998 RESEARCH NOTE David A. Leopold Nikos K. Logothetis Microsaccades differentially modulate neural activity in the striate and extrastriate visual cortex
Exp Brain Res (1998) 123:341 345 Springer-Verlag 1998 RESEARCH NOTE David A. Leopold Nikos K. Logothetis Microsaccades differentially modulate neural activity in the striate and extrastriate visual cortex
QUANTAL ANALYSIS AT THE NEUROMUSCULAR JUNCTION
 Hons Neuroscience Professor R.R. Ribchester QUANTAL ANALYSIS AT THE NEUROMUSCULAR JUNCTION Our present understanding of the fundamental physiological mechanism of transmitter release at synapses is mainly
Hons Neuroscience Professor R.R. Ribchester QUANTAL ANALYSIS AT THE NEUROMUSCULAR JUNCTION Our present understanding of the fundamental physiological mechanism of transmitter release at synapses is mainly
Anatomy Review. Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.
 Anatomy Review Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.com) Page 1. Introduction The structure of neurons reflects their function.
Anatomy Review Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.com) Page 1. Introduction The structure of neurons reflects their function.
Neu. al Network Analysis of Distributed Representations of Dynamical Sensory-Motor rrransformations in the Leech
 28 Lockery t Fang and Sejnowski Neu. al Network Analysis of Distributed Representations of Dynamical Sensory-Motor rrransformations in the Leech Shawn R. LockerYt Van Fangt and Terrence J. Sejnowski Computational
28 Lockery t Fang and Sejnowski Neu. al Network Analysis of Distributed Representations of Dynamical Sensory-Motor rrransformations in the Leech Shawn R. LockerYt Van Fangt and Terrence J. Sejnowski Computational
12. Nervous System: Nervous Tissue
 12. Nervous System: Nervous Tissue I. Introduction to the Nervous System General functions of the nervous system The nervous system has three basic functions: 1. Gather sensory input from the environment
12. Nervous System: Nervous Tissue I. Introduction to the Nervous System General functions of the nervous system The nervous system has three basic functions: 1. Gather sensory input from the environment
2 Neurons. 4 The Brain: Cortex
 1 Neuroscience 2 Neurons output integration axon cell body, membrane potential Frontal planning control auditory episodes soma motor Temporal Parietal action language objects space vision Occipital inputs
1 Neuroscience 2 Neurons output integration axon cell body, membrane potential Frontal planning control auditory episodes soma motor Temporal Parietal action language objects space vision Occipital inputs
The Sonometer The Resonant String and Timbre Change after plucking
 The Sonometer The Resonant String and Timbre Change after plucking EQUIPMENT Pasco sonometers (pick up 5 from teaching lab) and 5 kits to go with them BK Precision function generators and Tenma oscilloscopes
The Sonometer The Resonant String and Timbre Change after plucking EQUIPMENT Pasco sonometers (pick up 5 from teaching lab) and 5 kits to go with them BK Precision function generators and Tenma oscilloscopes
The Action Potential Graphics are used with permission of: adam.com (http://www.adam.com/) Benjamin Cummings Publishing Co (http://www.awl.
 The Action Potential Graphics are used with permission of: adam.com (http://www.adam.com/) Benjamin Cummings Publishing Co (http://www.awl.com/bc) ** If this is not printed in color, it is suggested you
The Action Potential Graphics are used with permission of: adam.com (http://www.adam.com/) Benjamin Cummings Publishing Co (http://www.awl.com/bc) ** If this is not printed in color, it is suggested you
Nervous Tissue Dr. Archana Rani Associate Professor Department of Anatomy KGMU UP, Lucknow
 13.01.2015 Nervous Tissue Dr. Archana Rani Associate Professor Department of Anatomy KGMU UP, Lucknow Introduction Property of irritability and conductivity Respond to various types of stimuli Distributed
13.01.2015 Nervous Tissue Dr. Archana Rani Associate Professor Department of Anatomy KGMU UP, Lucknow Introduction Property of irritability and conductivity Respond to various types of stimuli Distributed
Nervous System Organization. PNS and CNS. Nerves. Peripheral Nervous System. Peripheral Nervous System. Motor Component.
 Nervous System Organization PNS and CNS Chapters 8 and 9 Peripheral Nervous System (PNS) connects CNS to sensory receptors, muscles and glands Central Nervous System (CNS) control/integrating center brain
Nervous System Organization PNS and CNS Chapters 8 and 9 Peripheral Nervous System (PNS) connects CNS to sensory receptors, muscles and glands Central Nervous System (CNS) control/integrating center brain
What is the basic component of the brain and spinal cord communication system?
 EXPLORING PSYCHOLOGY David Myers The Biology of Mind Chapter 2 Neural Communication Neurons How Neurons Communicate How Neurotransmitters Influence Us The Nervous System The Peripheral Nervous System The
EXPLORING PSYCHOLOGY David Myers The Biology of Mind Chapter 2 Neural Communication Neurons How Neurons Communicate How Neurotransmitters Influence Us The Nervous System The Peripheral Nervous System The
is refractory for some period of time following its occurval, of the intensity of the stimuli at a given interrence
 J.Comp. Physiological Psychology 1973, Vol. 83, No. 3, 492-498 Refractory Period and Habituation of Acoustic Startle Response in Rats Charles J. Wilson and Philip M. Groves Department of Psychology, University
J.Comp. Physiological Psychology 1973, Vol. 83, No. 3, 492-498 Refractory Period and Habituation of Acoustic Startle Response in Rats Charles J. Wilson and Philip M. Groves Department of Psychology, University
The intermedius nucleus of the medulla: A potential site for the integration of cervical information and the generation of autonomic responses
 The intermedius nucleus of the medulla: A potential site for the integration of cervical information and the generation of autonomic responses 1 Journal of Chemical Neuroanatomy November 2009, 38, pp.
The intermedius nucleus of the medulla: A potential site for the integration of cervical information and the generation of autonomic responses 1 Journal of Chemical Neuroanatomy November 2009, 38, pp.
A Segmentation Algorithm for Zebra Finch Song at the Note Level. Ping Du and Todd W. Troyer
 A Segmentation Algorithm for Zebra Finch Song at the Note Level Ping Du and Todd W. Troyer Neuroscience and Cognitive Science Program, Dept. of Psychology University of Maryland, College Park, MD 20742
A Segmentation Algorithm for Zebra Finch Song at the Note Level Ping Du and Todd W. Troyer Neuroscience and Cognitive Science Program, Dept. of Psychology University of Maryland, College Park, MD 20742
Encoders for Linear Motors in the Electronics Industry
 Technical Information Encoders for Linear Motors in the Electronics Industry The semiconductor industry and automation technology increasingly require more precise and faster machines in order to satisfy
Technical Information Encoders for Linear Motors in the Electronics Industry The semiconductor industry and automation technology increasingly require more precise and faster machines in order to satisfy
NEUROLOCALIZATION MADE EASY
 NEUROLOCALIZATION MADE EASY Jared B. Galle, DVM, Diplomate ACVIM (Neurology) Dogwood Veterinary Referral Center 4920 Ann Arbor-Saline Road Ann Arbor, MI 48103 Localizing a neurologic problem to an anatomical
NEUROLOCALIZATION MADE EASY Jared B. Galle, DVM, Diplomate ACVIM (Neurology) Dogwood Veterinary Referral Center 4920 Ann Arbor-Saline Road Ann Arbor, MI 48103 Localizing a neurologic problem to an anatomical
Designing eye tracking experiments to measure human behavior
 Designing eye tracking experiments to measure human behavior Eindhoven, The Netherlands August, 2010 Ricardo Matos Tobii Technology Steps involved in measuring behaviour 1. Formulate and initial question
Designing eye tracking experiments to measure human behavior Eindhoven, The Netherlands August, 2010 Ricardo Matos Tobii Technology Steps involved in measuring behaviour 1. Formulate and initial question
Primary Motor Pathway
 Understanding Eye Movements Abdullah Moh. El-Menaisy, MD, FRCS Chief, Neuro-ophthalmology ophthalmology & Investigation Units, Dhahran Eye Specialist Hospital, Dhahran, Saudi Arabia Primary Motor Pathway
Understanding Eye Movements Abdullah Moh. El-Menaisy, MD, FRCS Chief, Neuro-ophthalmology ophthalmology & Investigation Units, Dhahran Eye Specialist Hospital, Dhahran, Saudi Arabia Primary Motor Pathway
PUMPED Nd:YAG LASER. Last Revision: August 21, 2007
 PUMPED Nd:YAG LASER Last Revision: August 21, 2007 QUESTION TO BE INVESTIGATED: How can an efficient atomic transition laser be constructed and characterized? INTRODUCTION: This lab exercise will allow
PUMPED Nd:YAG LASER Last Revision: August 21, 2007 QUESTION TO BE INVESTIGATED: How can an efficient atomic transition laser be constructed and characterized? INTRODUCTION: This lab exercise will allow
The Physiology of the Senses Lecture 1 - The Eye www.tutis.ca/senses/
 The Physiology of the Senses Lecture 1 - The Eye www.tutis.ca/senses/ Contents Objectives... 2 Introduction... 2 Accommodation... 3 The Iris... 4 The Cells in the Retina... 5 Receptive Fields... 8 The
The Physiology of the Senses Lecture 1 - The Eye www.tutis.ca/senses/ Contents Objectives... 2 Introduction... 2 Accommodation... 3 The Iris... 4 The Cells in the Retina... 5 Receptive Fields... 8 The
Appendix 4 Simulation software for neuronal network models
 Appendix 4 Simulation software for neuronal network models D.1 Introduction This Appendix describes the Matlab software that has been made available with Cerebral Cortex: Principles of Operation (Rolls
Appendix 4 Simulation software for neuronal network models D.1 Introduction This Appendix describes the Matlab software that has been made available with Cerebral Cortex: Principles of Operation (Rolls
Hearing and Deafness 1. Anatomy & physiology
 Hearing and Deafness 1. Anatomy & physiology Chris Darwin Web site for lectures, lecture notes and filtering lab: http://www.lifesci.susx.ac.uk/home/chris_darwin/ safari 1 Outer, middle & inner ear Capture;
Hearing and Deafness 1. Anatomy & physiology Chris Darwin Web site for lectures, lecture notes and filtering lab: http://www.lifesci.susx.ac.uk/home/chris_darwin/ safari 1 Outer, middle & inner ear Capture;
Biological Neurons and Neural Networks, Artificial Neurons
 Biological Neurons and Neural Networks, Artificial Neurons Neural Computation : Lecture 2 John A. Bullinaria, 2015 1. Organization of the Nervous System and Brain 2. Brains versus Computers: Some Numbers
Biological Neurons and Neural Networks, Artificial Neurons Neural Computation : Lecture 2 John A. Bullinaria, 2015 1. Organization of the Nervous System and Brain 2. Brains versus Computers: Some Numbers
The Number of Cortical Neurons Used to See
 The Number of Cortical Neurons Used to See Krisha Mehta Bronx High School of Science Mentor: Dr.Denis Pelli Teacher: Mr.Lee K. K. Mehta (2013) The number of cortical neurons used to see. Intel Science
The Number of Cortical Neurons Used to See Krisha Mehta Bronx High School of Science Mentor: Dr.Denis Pelli Teacher: Mr.Lee K. K. Mehta (2013) The number of cortical neurons used to see. Intel Science
CHAPTER XV PDL 101 HUMAN ANATOMY & PHYSIOLOGY. Ms. K. GOWRI. M.Pharm., Lecturer.
 CHAPTER XV PDL 101 HUMAN ANATOMY & PHYSIOLOGY Ms. K. GOWRI. M.Pharm., Lecturer. Types of Muscle Tissue Classified by location, appearance, and by the type of nervous system control or innervation. Skeletal
CHAPTER XV PDL 101 HUMAN ANATOMY & PHYSIOLOGY Ms. K. GOWRI. M.Pharm., Lecturer. Types of Muscle Tissue Classified by location, appearance, and by the type of nervous system control or innervation. Skeletal
Measurement of Charge-to-Mass (e/m) Ratio for the Electron
 Measurement of Charge-to-Mass (e/m) Ratio for the Electron Experiment objectives: measure the ratio of the electron charge-to-mass ratio e/m by studying the electron trajectories in a uniform magnetic
Measurement of Charge-to-Mass (e/m) Ratio for the Electron Experiment objectives: measure the ratio of the electron charge-to-mass ratio e/m by studying the electron trajectories in a uniform magnetic
Vestibular Assessment
 Oculomotor Examination A. Tests performed in room light Vestibular Assessment 1. Spontaneous nystagmus 2. Gaze holding nystagmus 3. Skew deviation 4. Vergence 5. Decreased vestibular ocular reflex i. Head
Oculomotor Examination A. Tests performed in room light Vestibular Assessment 1. Spontaneous nystagmus 2. Gaze holding nystagmus 3. Skew deviation 4. Vergence 5. Decreased vestibular ocular reflex i. Head
Exploring Creation with Advanced Biology ~ Schedule for 2015-2016 Apologia ~ Exploring Creation with Biology ~ Module 1 (Week 1) Date:
 Apologia ~ Exploring Creation with Biology ~ Module 1 (Week 1) Mon., Aug. 31 Tue., Sep. 1 Wed., Sept. 2 Thurs., Sept. 3 Fri., Sept. 4 Reading Pgs. 1-4; Introduction, Heart of the matter, Pgs. 5-8; Organization
Apologia ~ Exploring Creation with Biology ~ Module 1 (Week 1) Mon., Aug. 31 Tue., Sep. 1 Wed., Sept. 2 Thurs., Sept. 3 Fri., Sept. 4 Reading Pgs. 1-4; Introduction, Heart of the matter, Pgs. 5-8; Organization
Ampere's Law. Introduction. times the current enclosed in that loop: Ampere's Law states that the line integral of B and dl over a closed path is 0
 1 Ampere's Law Purpose: To investigate Ampere's Law by measuring how magnetic field varies over a closed path; to examine how magnetic field depends upon current. Apparatus: Solenoid and path integral
1 Ampere's Law Purpose: To investigate Ampere's Law by measuring how magnetic field varies over a closed path; to examine how magnetic field depends upon current. Apparatus: Solenoid and path integral
INTERFERENCE OF SOUND WAVES
 1/2016 Sound 1/8 INTERFERENCE OF SOUND WAVES PURPOSE: To measure the wavelength, frequency, and propagation speed of ultrasonic sound waves and to observe interference phenomena with ultrasonic sound waves.
1/2016 Sound 1/8 INTERFERENCE OF SOUND WAVES PURPOSE: To measure the wavelength, frequency, and propagation speed of ultrasonic sound waves and to observe interference phenomena with ultrasonic sound waves.
Human Neuroanatomy. Grades 9-12. Driving Question: How did the evolution of the human brain impact the structure and function it has today?
 Human Neuroanatomy Grades 9-12 Driving Question: How did the evolution of the human brain impact the structure and function it has today? Objectives: Students will be able to Describe the basic parts and
Human Neuroanatomy Grades 9-12 Driving Question: How did the evolution of the human brain impact the structure and function it has today? Objectives: Students will be able to Describe the basic parts and
Doppler. Doppler. Doppler shift. Doppler Frequency. Doppler shift. Doppler shift. Chapter 19
 Doppler Doppler Chapter 19 A moving train with a trumpet player holding the same tone for a very long time travels from your left to your right. The tone changes relative the motion of you (receiver) and
Doppler Doppler Chapter 19 A moving train with a trumpet player holding the same tone for a very long time travels from your left to your right. The tone changes relative the motion of you (receiver) and
Reflection and Refraction
 Equipment Reflection and Refraction Acrylic block set, plane-concave-convex universal mirror, cork board, cork board stand, pins, flashlight, protractor, ruler, mirror worksheet, rectangular block worksheet,
Equipment Reflection and Refraction Acrylic block set, plane-concave-convex universal mirror, cork board, cork board stand, pins, flashlight, protractor, ruler, mirror worksheet, rectangular block worksheet,
Adding Heart to Your Technology
 RMCM-01 Heart Rate Receiver Component Product code #: 39025074 KEY FEATURES High Filtering Unit Designed to work well on constant noise fields SMD component: To be installed as a standard component to
RMCM-01 Heart Rate Receiver Component Product code #: 39025074 KEY FEATURES High Filtering Unit Designed to work well on constant noise fields SMD component: To be installed as a standard component to
Andrew Rosen - Chapter 3: The Brain and Nervous System Intro:
 Intro: Brain is made up of numerous, complex parts Frontal lobes by forehead are the brain s executive center Parietal lobes wave sensory information together (maps feeling on body) Temporal lobes interpret
Intro: Brain is made up of numerous, complex parts Frontal lobes by forehead are the brain s executive center Parietal lobes wave sensory information together (maps feeling on body) Temporal lobes interpret
http://abcnews.go.com/politics/video/obama-says-brain-initiative-will-be-transformative-18861944
 http://abcnews.go.com/politics/video/obama-says-brain-initiative-will-be-transformative-18861944 What are the nervous system s functions? The nervous system organizes and controls an individual s appropriate
http://abcnews.go.com/politics/video/obama-says-brain-initiative-will-be-transformative-18861944 What are the nervous system s functions? The nervous system organizes and controls an individual s appropriate
Anatomy and Physiology of Hearing (added 09/06)
 Anatomy and Physiology of Hearing (added 09/06) 1. Briefly review the anatomy of the cochlea. What is the cochlear blood supply? SW 2. Discuss the effects of the pinna, head and ear canal on the transmission
Anatomy and Physiology of Hearing (added 09/06) 1. Briefly review the anatomy of the cochlea. What is the cochlear blood supply? SW 2. Discuss the effects of the pinna, head and ear canal on the transmission
Lab Exercise 9. Nervous Tissue. Brain. Cranial Nerves. Spinal Cord. Spinal Nerves
 Lab Exercise 9 Nervous Tissue Brain Cranial Nerves Spinal Cord Spinal Nerves Textbook Reference: See Chapter 11 for histology of nerve tissue and spinal cord See Chapter 12 for brain and spinal cord anatomy
Lab Exercise 9 Nervous Tissue Brain Cranial Nerves Spinal Cord Spinal Nerves Textbook Reference: See Chapter 11 for histology of nerve tissue and spinal cord See Chapter 12 for brain and spinal cord anatomy
GE Healthcare. Quick Guide PDF PROOF. Neuromuscular Transmission (NMT)
 GE Healthcare Quick Guide Neuromuscular Transmission (NMT) WHAT is Neuromuscular Transmission (NMT)? Neuromuscular Transmission (NMT) is the transfer of an impulse between a nerve and a muscle in the neuromuscular
GE Healthcare Quick Guide Neuromuscular Transmission (NMT) WHAT is Neuromuscular Transmission (NMT)? Neuromuscular Transmission (NMT) is the transfer of an impulse between a nerve and a muscle in the neuromuscular
Muscle Physiology. Lab 5. Human Muscle Physiology
 Lab 5 Human At the beginning of lab you will have the opportunity for 2 bonus points! You must guess which person in the class will have: 1) Maximum Grip Force 2) Longest time to half-max Force (longest
Lab 5 Human At the beginning of lab you will have the opportunity for 2 bonus points! You must guess which person in the class will have: 1) Maximum Grip Force 2) Longest time to half-max Force (longest
3nd Biennial Contemporary Clinical Neurophysiological Symposium October 12, 2013 Fundamentals of NCS and NMJ Testing
 3nd Biennial Contemporary Clinical Neurophysiological Symposium October 12, 2013 Fundamentals of NCS and NMJ Testing Peter D. Donofrio, M.D. Professor of Neurology Vanderbilt University Medical Center
3nd Biennial Contemporary Clinical Neurophysiological Symposium October 12, 2013 Fundamentals of NCS and NMJ Testing Peter D. Donofrio, M.D. Professor of Neurology Vanderbilt University Medical Center
ZEBRAFISH SYSTEMS SYSTEMS ZEBRAFISH. Fast, automatic analyses
 ZEBRAFISH SYSTEMS ZEBRAFISH SYSTEMS Fast, automatic analyses Wide-scale evaluation possibilities ( averaged ECG, QT, QTv, HRV) Distortion-free ECG signal transfer Social interaction examinations Measurements
ZEBRAFISH SYSTEMS ZEBRAFISH SYSTEMS Fast, automatic analyses Wide-scale evaluation possibilities ( averaged ECG, QT, QTv, HRV) Distortion-free ECG signal transfer Social interaction examinations Measurements
Vision: Receptors. Modes of Perception. Vision: Summary 9/28/2012. How do we perceive our environment? Sensation and Perception Terminology
 How do we perceive our environment? Complex stimuli are broken into individual features, relayed to the CNS, then reassembled as our perception Sensation and Perception Terminology Stimulus: physical agent
How do we perceive our environment? Complex stimuli are broken into individual features, relayed to the CNS, then reassembled as our perception Sensation and Perception Terminology Stimulus: physical agent
