Production and Characterization of Polyclonal Antibodies Specific for Entamoeba histolytica
|
|
|
- Arthur Parks
- 10 years ago
- Views:
Transcription
1 136 (2): , December 2007 ISSN Production and Characterization of Polyclonal Antibodies Specific Ivan Chester G. Canoy 1 and Windell L. Rivera 1,2* 1 Institute of Biology, College of Science, University of the Philippines, Quezon City 2 Molecular Protozoology Laboratory, Natural Sciences Research Institute University of the Philippines, Diliman, Quezon City Murine polyclonal antibodies (pabs) were produced by immunizing BALB/c mice with trophozoites of Entamoeba histolytica HK9 in Freund s adjuvant. Using indirect fluorescent antibody test (IFAT), we have developed polyclonal IgG antibodies that specifically reacted with formalin-fixed E. histolytica trophozoites, but failed to bind with live trophozoites, suggesting the epitope to be cytoplasmic and not cell surface located. Entamoeba dispar, Entamoeba coli, and Blastocystis hominis showed no reactivity with the pabs. Western immunoblot analysis revealed the reactivity of the pabs with 95 kda and 103 kda major proteins, antigens that we consider as novel as there are no reports to date on these molecules. The pabs developed in the present study are valuable, particularly when used in IFAT in the accurate detection and differentiation of the pathogenic E. histolytica from the non-invasive E. dispar. Key Words: amebiasis, indirect fluorescent antibody test, immunoblot, murine antibodies INTRODUCTION Amebiasis, a disease caused by the enteric protozoan Entamoeba histolytica, is a major public health problem, especially, in developing countries. E. histolytica is responsible for up to 100,000 deaths per year, placing it second only to malaria as a cause of mortality due to protozoan parasites (World Health Organization 1997). Because of this, the detection of E. histolytica is an important goal of the clinical microbiology laboratories throughout the world (Tanyuksel & Petri 2003). Laboratory diagnosis of amebiasis based on direct microscopic observation of stool samples is still by far the most widespread method. This approach, however, is labor-intensive and unreliable because it is unable to differentiate E. histolytica from morphologically *Corresponding author: [email protected] identical species that are nonpathogenic, such as Entamoeba dispar and Entamoeba moshkovskii (Diamond & Clark 1993; Ackers 2002; Tanyuksel & Petri 2003). This may lead to false positives for amebic infection and subsequent wrong treatment of the actual disease. More recent methods for the diagnosis of amebic infection are growth culture, stool antigen and antibody detection through enzyme-linked immunosorbent assay (ELISA), polymerase chain reaction (PCR) detection, indirect hemagglutination assays (IHA) and indirect fluorescent antibody test (IFAT) (Tanyuksel & Petri 2003). Studies on the application of monoclonal antibodies in IFAT to detect E. histolytica have been reported (Tachibana et al. 1990; Gonzales-Ruiz et al. 1992; Walderich et al. 1998; Yau et al. 2001). However, monoclonal antibodies are very expensive to produce and are not readily available in developing countries such as the 131
2 Philippines. In addition, there have been no reports on the development of polyclonal antibodies (pabs) being applied in IFAT to detect E. histolytica. If pabs that have comparable specificity and sensitivity to monoclonal antibodies can be produced, it is possible to generate low-priced detection kits for amebiasis. MATERIALS AND METHODS Parasites and culture conditions Trophozoites of E. histolytica HK9 were axenically grown at 35.5º C in BI-S-33 medium (consists of Biosate [a peptone mixture with 2 parts trypticase and 1 part yeast extract] and a vitamin-tween 80 mixture) supplemented with 10% heat-inactivated bovine serum (Diamond et al. 1978). Cultures of enteric protozoans such as E. dispar, E. coli and B. hominis were also used as controls in the IFAT experiment. Three dayold parasite culture suspensions were washed 3 times with ice-cold phosphate-buffered saline (PBS; ph 7.4) by centrifugation at 400 g for 2 min at 4º C before being used. Production of polyclonal antibodies (pabs) Three 7-week old female BALB/c mice were injected intraperitoneally with live E. histolytica HK-9 trophozoites in complete Freund s adjuvant (CFA) according to the method described by Coligan (1991). Prior to immunization, blood was collected to serve as a control. The serum was prepared as follows: the blood samples, collected from the mice through venous puncture, were allowed to stand for an hour at room temperature and another hour at 4º C; it was centrifuged at 10,000 rpm for 10 minutes. The prepared serum was stored in 0.5 ml polypropylene microcentrifuge tubes at -20º C until use. The antibody titer of the serum was assayed using IFAT as described below. For the priming immunization, 1 ml of PBS with 20 x 106 live E. histolytica trophozoites was added in portions of 0.2 ml to 1 ml of CFA at 4º C. Each mouse was injected intraperitoneally with 0.2 ml of the CFA antigen emulsion. Throughout the process, the mixture was chilled on a bed of ice from time to time to keep the mixture as close to 4º C as possible to prevent denaturation of proteins. Blood was collected and the serum was prepared and assayed, 10 days after the priming immunization. Four weeks after the priming immunization, the mice were boosted with same number and concentration of trophozoites in emulsion but instead of using CFA, incomplete Freund s adjuvant (IFA) was used. The mice were boosted again after two additional weeks. The mice were bled 10 days after each booster immunization and the serum was prepared and assayed using IFAT. Isotyping of polyclonal antibodies The pabs produced were assayed for specific isotype using Rapid Mouse IsoGoldTM (BioAssay Works, L.L.C). The procedure was performed according to the manufacturer s instructions. IFAT using formalin-fixed trophozoites IFAT was done according to the methods previously described by Tachibana et al. (1990) with some modifications. Trophozoites in cultures were fixed with 3% formalin in PBS for 30 min at 4º C and washed three times with PBS. The parasites were suspended in PBS to a concentration of 106 cells/ml. Two microliters of the suspension was added to the wells of multispot glass slides, which were air-dried at room temperature. The prepared slides were wrapped in aluminum foil and kept at -20º C until use. The IFAT was carried out at 23º C in a moist chamber. The antigen slides were allowed to thaw before use. The wells of the multispot slides were treated with 50 μl of 3% skim milk in PBS (PBS-SKM) for 30 minutes. Thereafter, the PBS-SKM solution was aspirated. For the purpose of titration, the purified antiserum was serially diluted starting at a ratio of 1:16, followed by fourfold stepwise dilutions (1:64, 1:256, 1:1024, 1:4096, 1:16384, and 1:65,536). Then, 50 μl of different dilutions of the antiserum were added to each well and were incubated for 1 h. Three wells were used for each antiserum dilution and three trials were done. The pre-immune serum served as a negative control. The slides were washed five times with PBS by immersing the slide in chambers filled with PBS (each washing was done in separate chambers; 1st wash: immediately; 2nd wash: 4 min; 3rd wash: 4 min; 4th wash; 3 min; 5th wash; 4 min). Afterwards, the wells were allowed to dry and were treated with 40 μl of commercially available fluorescein isothiocyante (FITC)-labeled goat anti-mouse IgG (MBL, Japan), diluted 1:40, in PBS-SKM for 30 min. The slides were again washed with PBS and incubated for 30 min. The slides were mounted with 50% glycerol in PBS and were observed under a Zeiss Axiovert fluorescence microscope. 132
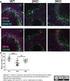
3 IFAT using live trophozoites The method was done according to the procedure previously described by Tachibana et al. (1991) with modifications. Harvested trophozoites from cultures were washed three times with cold PBS. The parasites were suspended in PBS to a concentration of 106 cells/ ml. One milliliter of the suspension was placed in 1.5 ml centrifuge tubes. The suspension was centrifuged for 2 min at 400 x g. It was then blocked with 500 μl 3 % PBS-SKM for 15 min. It was centrifuged for 2 min at 400 x g. The cells were incubated with 300 μl antiserum for 15 min. The cells were washed twice with cold PBS and centrifuged for 2 min at 400 x g. The cells were incubated with the 300 μl FITC-labeled goat anti-mouse IgG (MBL, Japan), diluted 1:50, in PBS-SKM for 30 min. The cells were again washed twice with cold PBS and viewed under Zeiss Axiovert fluorescence microscope. The incubation steps were carried out in 1.5 ml centrifuge tubes on ice. We have produced pabs that are capable of detecting and distinguishing pathogenic E. histolytica from noninvasive E. dispar and other enteric protozoans like E. coli and B. hominis. The pabs isotype was identified as IgG. From the results of the IFAT, the sera and ascitic fluids rich in pabs showed a strong positive fluorescence on formalin-fixed trophozoites of E. histolytica (Figure 1). The titer of the produced Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and Western blotting Trophozoites of E. histolytica were solubilized with an equal volume of sample buffer (0.25 mm Tris-HCl [ph 6.8], 4% SDS, 10% β-mercaptoethanol, 20% glycerol, 0.1% bromophenol blue, and 2 mm EDTA) containing 2 mm phenylmethylsulfonyl fluoride, 2 mm pepstatin, and 4 μm leupeptin for 5 min at 95º C. The supernatant that was obtained after centrifugation at 10,000 g for 3 min was subjected to denaturing SDS-PAGE, using 10% polyacrylamide separating gel, according to the method of Laemmli (1970). Santa Cruz Biotechnology stained broad range molecular mass markers (sc-2361) were used. Electrophoresed proteins were transferred onto a sheet of 0.45-μm nitrocellulose membrane using the procedure of Towbin et al. (1979). Electrophoretic transfer was carried out at 200 ma for 1 h. Some lanes of the sheet were stained with Coomassie brilliant blue, while the others were incubated first with PBS-SKM for 2 h and then with antiserum (1:200 dilution in PBS- SKM) for an additional 2 h at 23º C. The membrane was washed with PBS containing 0.05% Tween 20 and afterwards, it was incubated with 1:250 commercially available horseradish peroxidase-labeled goat antimouse IgG (Sigma) for 1 h. The sheet was washed and developed in a solution of 0.05% diaminobenzidine (DAB) plus 0.03% hydrogen peroxide in PBS for 1h. RESULTS AND DISCUSSION Figure 1. (a) Phase-contrast and (b) Immunofluorescence photomicrographs of formalin-fixed trophozoites of Entamoeba histolytica probed with mouse antiserum (diluted 1:64) containing the pabs followed by FITC-labeled goat anti-mouse IgG. Fluorescence was observed (localized at cytoplasm). Bar, 20 μm. 133
4 A B Figure 2. (a) Phase-contrast and (b) Immunofluorescence photomicrographs of formalin-fixed trophozoites of Entamoeba dispar probed with mouse antiserum (diluted 1:64) containing the polyclonal antibodies followed by FITC-labeled goat anti-mouse IgG. No fluorescence was observed. Bar, 20 μm. A B Figure 3. (a) Phase-contrast and (b) Immunofluorescence photomicrographs of formalin-fixed trophozoites of Blastocystis hominis probed with mouse antiserum (diluted 1:64) containing the polyclonal antibodies followed by FITC-labeled goat anti-mouse IgG. No fluorescence was observed. Bar, 20 μm. 134
5 A B Figure 4. (a) Phase-contrast and (b) Immunofluorescence photomicrographs of a live trophozoite of Entamoeba histolytica (inside the circles) probed with mouse antiserum (diluted 1:64) containing the polyclonal antibodies followed by FITC-labeled goat anti-mouse IgG. No fluorescence of live trophozoite was observed (white arrow). Note that cells outside the circle that fluoresced were dead trophozoites of E. histolytica. Bar, 20 μm. antibodies was observed at the 16,384 dilution. In contrast, the pabs did not react with formalin-fixed trophozoites of E. dispar, E. coli (data not shown) and B. hominis as shown by the absence of fluorescence even at the 1:64 dilution of sera and ascitic fluids (Figures 2 and 3). Absence of fluorescence was also observed at the minimum 1:16 dilution. These suggest that the pabs are very sensitive and specific to E. histolytica. Live trophozoites of E. histolytica were also used to localize the epitopes recognized by the antibodies, i.e. if they are surface immunogens or not. From the results, no fluorescence was observed in IFAT of live trophozoites (Figure 4) suggesting that the epitopes recognized by the pabs are not exposed on the surface of the parasite. This was supported by the observed fluorescence localized in the cytoplasm in IFAT using formalin-fixed trophozoites (Figure 1). To characterize the antigenic molecules recognized by the pabs, SDS-PAGE under reducing conditions and Western immunoblot analysis were used. The pabs recognized two major protein bands with estimated molecular masses of 95 kda and 103 kda, respectively (Figure 5). The production of a number of monoclonal antibodies that are specific to E. histolytica have already been reported (Petri et al. 1990; Tachibana et al. 1990, 1991, 1997; Gonzalez-Ruiz et al. 1992, 1994; Sengupta et al. 1993; Leon et al. 1994; Yau et al. 2001) but this is the first report concerning the production of antibodies specific for the 95-kDa and 103-kDa antigenic molecules of E. histolytica. In the review made by Espinosa-Cantellano and Martinez-Palomo (1991), they have reported that most research groups have observed 11 principal antigenic proteins of E. histolytica having molecular weights of 220, 170, 150, 125, 96, 80, 60, 45, 20 and 9 kda. It is notable that the 95 kda molecule that was observed in the study has almost the same molecular mass as the 96 kda molecule reported by Torian et al. (1990). This 96 kda molecule is reported to have two forms in E. histolytica: an integral plasma membrane protein and a soluble form in cytosol. It is possible that the 95 kda molecule described here is the same as the 96 kda molecule reported by Torian et al. (1990) although it remains to be established. The results indicate that there are immunological differences between E. histolytica and E. dispar, and the produced pabs applied in IFAT can be useful in the 135
6 accurate detection and differentiation of E. histolytica from E. dispar. Since the method used can detect formalin-fixed trophozoites, this will be an important tool in epidemiological studies especially in far-flung areas where the samples cannot be kept fresh or frozen and are rather fixed with formalin or methanol. This will be useful in the establishment of an accurate prevalence data of E. histolytica infections in many parts of the world. Furthermore, these pabs may be used commercially in immunodiagnostic kits for E. histolytica infections. These future diagnostic kits can provide a fast, easy, cheap and accurate detection of E. histolytica. The pabs and the epitopes that they recognize may also contribute to the development of a potential vaccine for amebiasis. Lastly, the produced antibodies can be sold commercially as a positive control for immunological tests in laboratories. In summary, we have produced murine polyclonal antibodies that are specific to Entamoeba histolytica. These antibodies can detect and distinguish E. histolytica and E. dispar. The major epitopes that are detected by the produced pabs are located in the cytoplasm and have molecular masses of 95 kda and 103 kda, respectively. These data demonstrate that IFAT with the produced pabs can be useful in the accurate detection and differentiation of the pathogenic E. histolytica from non-invasive E. dispar. In this study, the produced pabs were only used in the detection of E. histolytica trophozoites from axenic cultures. In order to assess its applicability in field studies, the evaluation of the effectiveness of the produced pabs in the detection of E. histolytica in stool samples is highly recommended. The molecular analysis of the 95 kda and 103 kda major antigens and determination of their biological functions deserves further study. Other strains of E. histolytica and more enteric protozoans (e.g. Entamoeba moshkovskii and Cryptosporidium parvum) should be tested for crossreactivity with the produced pabs using IFAT. The application of the produced pabs in the detection of E. histolytica using ELISA should also be evaluated. ACKNOWLEDGEMENT We thank the Natural Sciences Research Institute (NSRI) of the University of the Philippines Diliman for partially funding this research. Figure 5. Western immunoblot analysis of the reactivity of pabs to trophozoites of Entamoeba histolytica HK9. Cell lysates were subjected to SDS-PAGE under reduced conditions and transferred to nitrocellulose membrane. Protein bands in lane b were stained with Coomassie brilliant blue. Lane c was treated with polyclonal antibodies, followed by horseradish peroxidase-labeled goat anti-mouse IgG. Lane a shows molecular mass standards. REFERENCES ACKERS JP The diagnostic implications of the separation of Entamoeba histolytica and Entamoeba dispar. J Biosci 27: COLIGAN JE Current Protocols in Immunology. Greene Pub. Associates and Wiley-Interscience. New York. 136
7 DIAMOND LS, HARLOW DR and CUNNICK CC A new medium for the axenic cultivation of Entamoeba histolytica and other Entamoeba. Trans R Soc Trop Med Hyg 72: DIAMOND LS and CLARK CG A redescription of Entamoeba histolytica Schaudinn, 1903 (Emended Walker, 1911) separating it from Entamoeba dispar Brumpt, J Euk Microbiol 40: ESPINOSA-CANTELLANO M and MARTINEZ- PALOMO A The plasma membrane of Entamoeba histolytica: structure and dynamics. Biol Cell 72: GONZALES-RUIZ A, HAQUE R, REHMAN T, AGUIRRE A, JARAMILLO C, CASTAÑON G, HALL A, GUHL F, RUIZ-PALACIOS G, WARHUST DC and MILES MA A monoclonal antibody for distinction of invasive and noninvasive clinical isolates of Entamoeba histolytica. J Clin Microbiol 30: GONZALES-RUIZ A, HAQUE R, REHMAN T, AGUIRRE A, HALL A, GUHL F, WARHUST DC and MILES MA Diagnosis of amebic dysentery by detection of Entamoeba histolytica fecal antigen by an invasive strain-specific, monoclonal antibody-based enzyme-linked immunosorbent assay. J Clin Microbiol 32: LAEMMLI UK Cleavage of structural proteins during the assembly of the head T4. Nature 227: LEON G, TOVAR R and MUÑOZ MDL A monoclonal antibody specific for a 40 kda polypeptide recognizes pathogenic strains of Entamoeba histolytica. Arch Med Res 25: PETRI WA Jr, JACKSON TFHG, GATHIRAM V, KRESS K, SAFFER LD, SNODGRASS TL, CHAPMAN MD, KEREN Z and MIRELMAN D Pathogenic and nonpathogenic strains of Entamoeba histolytica can be differentiated by monoclonal antibodies to the galactose-specific adherence lectin. Infect Immun 58: SENGUPTA K, DAS P, JOHNSON TM, CHAUDHURI PP, DAS D and NAIR GB Production and characterization of monoclonal antibodies against a highly immunogenic fraction of Entamoeba histolytica (NIH:200) and their application in the detection of current amoebic infection. J Euk Microbiol 40: TACHIBANA H, KOBAYASHI S, KATO Y, NAGAKURA K, KANEDA Y and TAKEUCHI T Identification of a pathogenic isolatespecific 30,000-Mr antigen of Entamoeba histolytica by using a monoclonal antibody. Infect Immun 51: TACHIBANA H, KOBAYASHI S, NAGAKURA K, KANEDA Y and TAKEUCHI T Reactivity of monoclonal antibodies to species-specific antigens of Entamoeba histolytica. J Protozool 38: TACHIBANA H, KOBAYASHI S, CHENG XJ and HIWATASHI E Differentiation of Entamoeba histolytica from E. dispar facilitated by monoclonal antibodies against a 150-kDa surface antigen. Parasitol Res 83: TACHIBANA H, KOBAYASHI S, CHENG XJ and HIWATASHI E Differentiation of Entamoeba histolytica from E. dispar facilitated by monoclonal antibodies against a 150-kDa surface antigen. Parasitol Res 83: TANYUKSEL M and PETRI WA Jr Laboratory Diagnosis of Amebiasis. Clin Microbiol Rev 16: TORIAN BE, REED SL, FLORES BM, CREELY CM, COWARD JE, VIAL K and STAMM WE The 96-kilodalton antigen as an integral membrane protein in pathogenic Entamoeba histolytica: potential differences in pathogenic and nonpathogenic isolates. Infect Immun 58: TOWBIN H, STAEHELIN T and GORDON J Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci 76:
8 WALDERICH B, BURCHARD G, KNOBLOCH J and MULLER L Development of monoclonal antibodies specifically recognizing the cyst stage of Entamoeba histolytica. Am J Trop Med Hyg 59: [WHO] WORLD HEALTH ORGANIZATION Entamoeba taxonomy. Bull WHO 75: YAU YC, CRANDALL I and KAIN KC Development of monoclonal antibodies which specifically recognize Entamoeba histolytica in preserved stool samples. J Clin Microbiol 39:
Protocol for Western Blotting
 Protocol for Western Blotting Materials Materials used on Day 3 Protease inhibitor stock: 1 μg/μl pepstatin A in DMSO 200 μm leupeptin in OG Buffer 200 mm PMSF: Freshly made. Ex) 34.8 mg PMSF in 1 ml isopropanol
Protocol for Western Blotting Materials Materials used on Day 3 Protease inhibitor stock: 1 μg/μl pepstatin A in DMSO 200 μm leupeptin in OG Buffer 200 mm PMSF: Freshly made. Ex) 34.8 mg PMSF in 1 ml isopropanol
Custom Antibodies Services. GeneCust Europe. GeneCust Europe
 GeneCust Europe Laboratoire de Biotechnologie du Luxembourg S.A. 6 rue Dominique Lang L-3505 Dudelange Luxembourg Tél. : +352 27620411 Fax : +352 27620412 Email : [email protected] Web : www.genecust.com
GeneCust Europe Laboratoire de Biotechnologie du Luxembourg S.A. 6 rue Dominique Lang L-3505 Dudelange Luxembourg Tél. : +352 27620411 Fax : +352 27620412 Email : [email protected] Web : www.genecust.com
Anti-ATF6 α antibody, mouse monoclonal (1-7)
 Anti-ATF6 α antibody, mouse monoclonal (1-7) 73-500 50 ug ATF6 (activating transcription factor 6) is an endoplasmic reticulum (ER) membrane-bound transcription factor activated in response to ER stress.
Anti-ATF6 α antibody, mouse monoclonal (1-7) 73-500 50 ug ATF6 (activating transcription factor 6) is an endoplasmic reticulum (ER) membrane-bound transcription factor activated in response to ER stress.
Western Blot Analysis with Cell Samples Grown in Channel-µ-Slides
 Western Blot Analysis with Cell Samples Grown in Channel-µ-Slides Polyacrylamide gel electrophoresis (PAGE) and subsequent analyses are common tools in biochemistry and molecular biology. This Application
Western Blot Analysis with Cell Samples Grown in Channel-µ-Slides Polyacrylamide gel electrophoresis (PAGE) and subsequent analyses are common tools in biochemistry and molecular biology. This Application
Anti-V5 Antibody Anti-V5-HRP Antibody
 Anti-V5 Antibody Anti-V5-HRP Antibody Catalog nos. R960-25, R961-25 Version F 073001 28-0140 www.invitrogen.com [email protected] ii Table of Contents Table of Contents...iii Overview...1 Western
Anti-V5 Antibody Anti-V5-HRP Antibody Catalog nos. R960-25, R961-25 Version F 073001 28-0140 www.invitrogen.com [email protected] ii Table of Contents Table of Contents...iii Overview...1 Western
Optimal Conditions for F(ab ) 2 Antibody Fragment Production from Mouse IgG2a
 Optimal Conditions for F(ab ) 2 Antibody Fragment Production from Mouse IgG2a Ryan S. Stowers, 1 Jacqueline A. Callihan, 2 James D. Bryers 2 1 Department of Bioengineering, Clemson University, Clemson,
Optimal Conditions for F(ab ) 2 Antibody Fragment Production from Mouse IgG2a Ryan S. Stowers, 1 Jacqueline A. Callihan, 2 James D. Bryers 2 1 Department of Bioengineering, Clemson University, Clemson,
Serology: Fluorescent antibody tests and other tests employing conjugated antibodies
 Serology: Fluorescent antibody tests and other tests employing conjugated antibodies Authors: Adapted by Prof M van Vuuren. Originally compiled by Dr RW Worthington. (Retired) Licensed under a Creative
Serology: Fluorescent antibody tests and other tests employing conjugated antibodies Authors: Adapted by Prof M van Vuuren. Originally compiled by Dr RW Worthington. (Retired) Licensed under a Creative
Protein extraction from Tissues and Cultured Cells using Bioruptor Standard & Plus
 Protein extraction from Tissues and Cultured Cells using Bioruptor Standard & Plus Introduction Protein extraction from tissues and cultured cells is the first step for many biochemical and analytical
Protein extraction from Tissues and Cultured Cells using Bioruptor Standard & Plus Introduction Protein extraction from tissues and cultured cells is the first step for many biochemical and analytical
THE His Tag Antibody, mab, Mouse
 THE His Tag Antibody, mab, Mouse Cat. No. A00186 Technical Manual No. TM0243 Update date 01052011 I Description.... 1 II Key Features. 2 III Storage 2 IV Applications.... 2 V Examples - ELISA..... 2 VI
THE His Tag Antibody, mab, Mouse Cat. No. A00186 Technical Manual No. TM0243 Update date 01052011 I Description.... 1 II Key Features. 2 III Storage 2 IV Applications.... 2 V Examples - ELISA..... 2 VI
Chapter 6: Antigen-Antibody Interactions
 Chapter 6: Antigen-Antibody Interactions I. Strength of Ag-Ab interactions A. Antibody Affinity - strength of total noncovalent interactions between single Ag-binding site on an Ab and a single epitope
Chapter 6: Antigen-Antibody Interactions I. Strength of Ag-Ab interactions A. Antibody Affinity - strength of total noncovalent interactions between single Ag-binding site on an Ab and a single epitope
Running protein gels and detection of proteins
 Running protein gels and detection of proteins 1. Protein concentration determination using the BIO RAD reagent This assay uses a colour change reaction to give a direct measurement of protein concentration.
Running protein gels and detection of proteins 1. Protein concentration determination using the BIO RAD reagent This assay uses a colour change reaction to give a direct measurement of protein concentration.
Chapter 3 Contd. Western blotting & SDS PAGE
 Chapter 3 Contd. Western blotting & SDS PAGE Western Blot Western blots allow investigators to determine the molecular weight of a protein and to measure relative amounts of the protein present in different
Chapter 3 Contd. Western blotting & SDS PAGE Western Blot Western blots allow investigators to determine the molecular weight of a protein and to measure relative amounts of the protein present in different
Western Blotting. USA: [email protected] UK & Europe: [email protected] China: [email protected]. www.ptglab.com
 Western Blotting All steps are carried out at room temperature unless otherwise indicated. Recipes for all solutions highlighted bold are included at the end of the protocol. SDS-PAGE 1. Construct an SDS-PAGE
Western Blotting All steps are carried out at room temperature unless otherwise indicated. Recipes for all solutions highlighted bold are included at the end of the protocol. SDS-PAGE 1. Construct an SDS-PAGE
Chapter 2 Antibodies. Contents. Introduction
 Chapter 2 Antibodies Keywords Immunohistochemistry Antibody labeling Fluorescence microscopy Fluorescent immunocytochemistry Fluorescent immunohistochemistry Indirect immunocytochemistry Immunostaining
Chapter 2 Antibodies Keywords Immunohistochemistry Antibody labeling Fluorescence microscopy Fluorescent immunocytochemistry Fluorescent immunohistochemistry Indirect immunocytochemistry Immunostaining
Classic Immunoprecipitation
 292PR 01 G-Biosciences 1-800-628-7730 1-314-991-6034 [email protected] A Geno Technology, Inc. (USA) brand name Classic Immunoprecipitation Utilizes Protein A/G Agarose for Antibody Binding (Cat.
292PR 01 G-Biosciences 1-800-628-7730 1-314-991-6034 [email protected] A Geno Technology, Inc. (USA) brand name Classic Immunoprecipitation Utilizes Protein A/G Agarose for Antibody Binding (Cat.
Chapter 6. Antigen-Antibody Properties 10/3/2012. Antigen-Antibody Interactions: Principles and Applications. Precipitin reactions
 Chapter 6 Antigen-Antibody Interactions: Principles and Applications Antigen-Antibody Properties You must remember antibody affinity (single) VS avidity (multiple) High affinity: bound tightly and longer!
Chapter 6 Antigen-Antibody Interactions: Principles and Applications Antigen-Antibody Properties You must remember antibody affinity (single) VS avidity (multiple) High affinity: bound tightly and longer!
Dot Blot Analysis. Teacher s Guidebook. (Cat. # BE 502) think proteins! think G-Biosciences www.gbiosciences.com
 PR110 G-Biosciences 1-800-628-7730 1-314-991-6034 [email protected] A Geno Technology, Inc. (USA) brand name Dot Blot Analysis Teacher s Guidebook (Cat. # BE 502) think proteins! think G-Biosciences
PR110 G-Biosciences 1-800-628-7730 1-314-991-6034 [email protected] A Geno Technology, Inc. (USA) brand name Dot Blot Analysis Teacher s Guidebook (Cat. # BE 502) think proteins! think G-Biosciences
Pure-IP Western Blot Detection Kit
 Product Manual Pure-IP Western Blot Detection Kit Catalog Number PRB-5002 20 blots FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction The technique of immunoprecipitation (IP) is used
Product Manual Pure-IP Western Blot Detection Kit Catalog Number PRB-5002 20 blots FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction The technique of immunoprecipitation (IP) is used
STANDARD OPERATING PROCEDURE
 STANDARD OPERATING PROCEDURE Title: Evaluation using Western Blot SOP#: M-103 Version #: 1 Author: R. Saul Date Approved: Feb. 5, 2009 Date Modified: 1. PURPOSE The purpose of this document is to describe
STANDARD OPERATING PROCEDURE Title: Evaluation using Western Blot SOP#: M-103 Version #: 1 Author: R. Saul Date Approved: Feb. 5, 2009 Date Modified: 1. PURPOSE The purpose of this document is to describe
STANDARD OPERATING PROCEDURE
 STANDARD OPERATING PROCEDURE Title: Antibody Production at Strategic Diagnostics Inc. SOP#: M-119 Version #: 1 Date Approved: August 6, 2009 Author: Strategic Diagnostic Inc. Date Modified: 1. PURPOSE
STANDARD OPERATING PROCEDURE Title: Antibody Production at Strategic Diagnostics Inc. SOP#: M-119 Version #: 1 Date Approved: August 6, 2009 Author: Strategic Diagnostic Inc. Date Modified: 1. PURPOSE
WESTERN BLOT DETECTION KIT Buffers and detection reagents for up to ten 10 x 10 cm 2 blots. Fluorescent detection via: Goat anti-mouse SureLight P3
 WESTERN BLOT DETECTION KIT Buffers and detection reagents for up to ten 10 x 10 cm 2 blots Fluorescent detection via: Goat anti-mouse SureLight P3 Cat. #: WK-P112 6440 Dobbin Road, Suite D Phone (443)
WESTERN BLOT DETECTION KIT Buffers and detection reagents for up to ten 10 x 10 cm 2 blots Fluorescent detection via: Goat anti-mouse SureLight P3 Cat. #: WK-P112 6440 Dobbin Road, Suite D Phone (443)
Topic: Serological reactions: the purpose and a principle of reactions. Agglutination test. Precipitation test. CFT, IFT, ELISA, RIA.
 Topic: Serological reactions: the purpose and a principle of reactions. Agglutination test. Precipitation test. CFT, IFT, ELISA, RIA. Serology is the study and use of immunological tests to diagnose and
Topic: Serological reactions: the purpose and a principle of reactions. Agglutination test. Precipitation test. CFT, IFT, ELISA, RIA. Serology is the study and use of immunological tests to diagnose and
Western Blotting: Mini-gels
 Western Blotting: Mini-gels Materials a Protein Extraction Buffer (for callus or kernel), Solution Stock Final Volume Tris-HCl ph 80 1 M 200 mm 20 ml NaCl 4 M 100 mm 25 ml Sucrose 2 M 400 mm 20 ml EDTA
Western Blotting: Mini-gels Materials a Protein Extraction Buffer (for callus or kernel), Solution Stock Final Volume Tris-HCl ph 80 1 M 200 mm 20 ml NaCl 4 M 100 mm 25 ml Sucrose 2 M 400 mm 20 ml EDTA
Protein transfer from SDS-PAGE to nitrocellulose membrane using the Trans-Blot SD cell (Western).
 Western Blot SOP Protein transfer from SDS-PAGE to nitrocellulose membrane using the Trans-Blot SD cell (Western). Date: 8/16/05, 10/31/05, 2/6/06 Author: N.Oganesyan, R. Kim Edited by: R. Kim Summary:
Western Blot SOP Protein transfer from SDS-PAGE to nitrocellulose membrane using the Trans-Blot SD cell (Western). Date: 8/16/05, 10/31/05, 2/6/06 Author: N.Oganesyan, R. Kim Edited by: R. Kim Summary:
竞 争 性 分 析 Epitope Mapping 实 验 方 法
 竞 争 性 分 析 Epitope Mapping 实 验 方 法 ABSTRACT The simplest way to determine whether two monoclonal antibodies bind to distinct sites on a protein antigen is to carry out a competition assay. The assay can
竞 争 性 分 析 Epitope Mapping 实 验 方 法 ABSTRACT The simplest way to determine whether two monoclonal antibodies bind to distinct sites on a protein antigen is to carry out a competition assay. The assay can
PROTOCOL 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 www.mitosciences.com
 PROTOCOL Western Blotting Transfer and Detection Procedure 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 02-11 DESCRIPTION Western Blotting Transfer and Detection Procedure ADDITIONAL MATERIALS REQUIRED
PROTOCOL Western Blotting Transfer and Detection Procedure 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 02-11 DESCRIPTION Western Blotting Transfer and Detection Procedure ADDITIONAL MATERIALS REQUIRED
Methionine Sulfoxide Immunoblotting Kit
 Methionine Sulfoxide Immunoblotting Kit Item No. 600160 Customer Service 800.364.9897 * Technical Support 888.526.5351 www.caymanchem.com TABLE OF CONTENTS GENERAL INFORMATION 3 Materials Supplied 4 Precautions
Methionine Sulfoxide Immunoblotting Kit Item No. 600160 Customer Service 800.364.9897 * Technical Support 888.526.5351 www.caymanchem.com TABLE OF CONTENTS GENERAL INFORMATION 3 Materials Supplied 4 Precautions
Amersham High Molecular Weight Calibration Kit for native electrophoresis
 Amersham High Molecular Weight Calibration Kit for native electrophoresis A lyophilized mixture of five highly purified well-characterized proteins for use in molecular weight estimation under non-denaturing
Amersham High Molecular Weight Calibration Kit for native electrophoresis A lyophilized mixture of five highly purified well-characterized proteins for use in molecular weight estimation under non-denaturing
LAB 14 ENZYME LINKED IMMUNOSORBENT ASSAY (ELISA)
 STUDENT GUIDE LAB 14 ENZYME LINKED IMMUNOSORBENT ASSAY (ELISA) GOAL The goal of this laboratory lesson is to explain the concepts and technique of enzyme linked immunosorbent assay (ELISA). OBJECTIVES
STUDENT GUIDE LAB 14 ENZYME LINKED IMMUNOSORBENT ASSAY (ELISA) GOAL The goal of this laboratory lesson is to explain the concepts and technique of enzyme linked immunosorbent assay (ELISA). OBJECTIVES
Product name Company Cat # PowerPac Basic Power supply Bio Rad 165-6019 Mini Protean electrophoresis system Mini trans blot cell Bio Rad 170-3930
 SDS-PAGE and western blot for low molecular weight proteins (2-20kDa) Merav Marom Shamur, Smart Assays Aim: Analysis of low molecular weight proteins by SDS-PAGE and western blot under reducing conditions.
SDS-PAGE and western blot for low molecular weight proteins (2-20kDa) Merav Marom Shamur, Smart Assays Aim: Analysis of low molecular weight proteins by SDS-PAGE and western blot under reducing conditions.
Pharmaceutical Biotechnology. Recombinant DNA technology Western blotting and SDS-PAGE
 Pharmaceutical Biotechnology Recombinant DNA technology Western blotting and SDS-PAGE Recombinant DNA Technology Protein Synthesis Western Blot Western blots allow investigators to determine the molecular
Pharmaceutical Biotechnology Recombinant DNA technology Western blotting and SDS-PAGE Recombinant DNA Technology Protein Synthesis Western Blot Western blots allow investigators to determine the molecular
Custom Antibody Services
 prosci-inc.com Custom Antibody Services High Performance Antibodies and More Broad Antibody Catalog Extensive Antibody Services CUSTOM ANTIBODY SERVICES Established in 1998, ProSci Incorporated is a leading
prosci-inc.com Custom Antibody Services High Performance Antibodies and More Broad Antibody Catalog Extensive Antibody Services CUSTOM ANTIBODY SERVICES Established in 1998, ProSci Incorporated is a leading
Page finder. 1. Legal 3. 2. Handling 4 2.1. Safety warnings and precautions 4 2.2. Storage 4 2.3. Expiry 4
 GE Healthcare Amersham Low Molecular Weight Calibration Kit for SDS Electrophoresis A lyophilized mixture of six highly purified well-characterized proteins for use in molecular weight determination in
GE Healthcare Amersham Low Molecular Weight Calibration Kit for SDS Electrophoresis A lyophilized mixture of six highly purified well-characterized proteins for use in molecular weight determination in
POLYCLONAL ANTIBODY DEVELOPMENT
 POLYCLONAL ANTIBODY DEVELOPMENT Abcore is committed to excellence, and to responding to the changing needs of our customers. We have developed polyclonal antibodies for biotechnology and pharmaceutical
POLYCLONAL ANTIBODY DEVELOPMENT Abcore is committed to excellence, and to responding to the changing needs of our customers. We have developed polyclonal antibodies for biotechnology and pharmaceutical
Protein immunoblotting
 Protein immunoblotting (Western blotting) Dr. Serageldeen A. A. Sultan Lecturer of virology Dept. of Microbiology SVU, Qena, Egypt [email protected] Western blotting -It is an analytical technique used to
Protein immunoblotting (Western blotting) Dr. Serageldeen A. A. Sultan Lecturer of virology Dept. of Microbiology SVU, Qena, Egypt [email protected] Western blotting -It is an analytical technique used to
Chapter 18: Applications of Immunology
 Chapter 18: Applications of Immunology 1. Vaccinations 2. Monoclonal vs Polyclonal Ab 3. Diagnostic Immunology 1. Vaccinations What is Vaccination? A method of inducing artificial immunity by exposing
Chapter 18: Applications of Immunology 1. Vaccinations 2. Monoclonal vs Polyclonal Ab 3. Diagnostic Immunology 1. Vaccinations What is Vaccination? A method of inducing artificial immunity by exposing
EZ-Run Protein Gel Solution. EZ-Run Protein Standards. EZ-Run Gel Staining Solution. Traditional SDS-Page Reagents. Protein Electrophoresis
 EZ-Run Protein Gel Solution EZ-Run Protein Standards EZ-Run Gel Staining Solution Traditional SDS-Page Reagents Protein Electrophoresis protein electrophoresis Introduction Sodium dodecyl sulfate polyacrylamide
EZ-Run Protein Gel Solution EZ-Run Protein Standards EZ-Run Gel Staining Solution Traditional SDS-Page Reagents Protein Electrophoresis protein electrophoresis Introduction Sodium dodecyl sulfate polyacrylamide
ECL Western Blotting Substrate INSTRUCTIONS FOR USE OF PRODUCTS W1001 AND W1015.
 Technical Manual ECL Western Blotting Substrate INSTRUCTIONS FOR USE OF PRODUCTS W1001 AND W1015. PRINTED IN USA. 6/09 ECL Western Blotting Substrate All technical literature is available on the Internet
Technical Manual ECL Western Blotting Substrate INSTRUCTIONS FOR USE OF PRODUCTS W1001 AND W1015. PRINTED IN USA. 6/09 ECL Western Blotting Substrate All technical literature is available on the Internet
Nitrotyrosine Western blot starter pack
 Nitrotyrosine Western blot starter pack Cat no. A010-513 A convenient reagent pack containing Badrilla s high integrity nitrotyrosine monoclonal antibody (clone 2E11) and associated positive and negative
Nitrotyrosine Western blot starter pack Cat no. A010-513 A convenient reagent pack containing Badrilla s high integrity nitrotyrosine monoclonal antibody (clone 2E11) and associated positive and negative
CUSTOM ANTIBODIES. Fully customised services: rat and murine monoclonals, rat and rabbit polyclonals, antibody characterisation, antigen preparation
 CUSTOM ANTIBODIES Highly competitive pricing without compromising quality. Rat monoclonal antibodies for the study of gene expression and proteomics in mice and in mouse models of human diseases available.
CUSTOM ANTIBODIES Highly competitive pricing without compromising quality. Rat monoclonal antibodies for the study of gene expression and proteomics in mice and in mouse models of human diseases available.
2. Cut 6 sheets of Whatman 3MM paper and 1 sheet of blotting membrane to the size of the gel, or slightly smaller.
 Method for Western Blotting Western Blotting ELECTROPHORESIS Prepare and run an SDS PAGE gel. Select a gel percent which will give the best resolution for the size of antigen being analyzed (if known).
Method for Western Blotting Western Blotting ELECTROPHORESIS Prepare and run an SDS PAGE gel. Select a gel percent which will give the best resolution for the size of antigen being analyzed (if known).
ArC Amine Reactive Compensation Bead Kit
 ArC Amine Reactive Compensation Bead Kit Catalog no. A1346 Table 1. Contents and storage information. Material Amount Composition Storage Stability ArC reactive beads (Component A) ArC negative beads (Component
ArC Amine Reactive Compensation Bead Kit Catalog no. A1346 Table 1. Contents and storage information. Material Amount Composition Storage Stability ArC reactive beads (Component A) ArC negative beads (Component
Fluorescein Isothiocyanate (FITC)- conjugated Antibodies
 USER GUIDE Fluorescein Isothiocyanate (FITC)- conjugated Antibodies Catalog Numbers R933-25, R953-25, R963-25 Document Part Number 25-0376 Publication Number MAN0000194 Revision 2.0 For Research Use Only.
USER GUIDE Fluorescein Isothiocyanate (FITC)- conjugated Antibodies Catalog Numbers R933-25, R953-25, R963-25 Document Part Number 25-0376 Publication Number MAN0000194 Revision 2.0 For Research Use Only.
WESTERN BLOTTING TIPS AND TROUBLESHOOTING GUIDE TROUBLESHOOTING GUIDE
 WESTERN BLOTTING TIPS AND TROUBLESHOOTING GUIDE TIPS FOR SUCCESSFUL WESTERB BLOTS TROUBLESHOOTING GUIDE 1. Suboptimal protein transfer. This is the most common complaint with western blotting and could
WESTERN BLOTTING TIPS AND TROUBLESHOOTING GUIDE TIPS FOR SUCCESSFUL WESTERB BLOTS TROUBLESHOOTING GUIDE 1. Suboptimal protein transfer. This is the most common complaint with western blotting and could
Investigating the role of a Cryptosporidium parum apyrase in infection
 Investigating the role of a Cryptosporidium parum apyrase in infection David Riccardi and Patricio Manque Abstract This project attempted to characterize the function of a Cryptosporidium parvum apyrase
Investigating the role of a Cryptosporidium parum apyrase in infection David Riccardi and Patricio Manque Abstract This project attempted to characterize the function of a Cryptosporidium parvum apyrase
MagExtractor -Genome-
 Instruction manual MagExtractor-Genome-0810 F0981K MagExtractor -Genome- NPK-101 100 preparations Store at 4 C Contents [1] Introduction [2] Components [3] Materials required [4] Protocol 1. Purification
Instruction manual MagExtractor-Genome-0810 F0981K MagExtractor -Genome- NPK-101 100 preparations Store at 4 C Contents [1] Introduction [2] Components [3] Materials required [4] Protocol 1. Purification
4A. Types of Laboratory Tests Available and Specimens Required. Three main types of laboratory tests are used for diagnosing CHIK: virus
 4. LABORATORY 4A. Types of Laboratory Tests Available and Specimens Required Three main types of laboratory tests are used for diagnosing CHIK: virus isolation, reverse transcriptase-polymerase chain reaction
4. LABORATORY 4A. Types of Laboratory Tests Available and Specimens Required Three main types of laboratory tests are used for diagnosing CHIK: virus isolation, reverse transcriptase-polymerase chain reaction
Section XIII: Protein Separation in Agarose Gels
 Section XIII: In This Section Introduction 196 Buffers for in Agarose 197 Casting Agarose Gels for 198 Preparation and Loading of Protein Samples 198 Optimal Voltage and Electrophoretic Times 199 Detection
Section XIII: In This Section Introduction 196 Buffers for in Agarose 197 Casting Agarose Gels for 198 Preparation and Loading of Protein Samples 198 Optimal Voltage and Electrophoretic Times 199 Detection
Elabscience Biotechnology Co.,Ltd
 Biotechnology Co.,Ltd Your one-stop custom antibody service Antibody is a kind of protein that is the most widely used in the fields of biology and medicine. Antibodies, as the preparations for disease
Biotechnology Co.,Ltd Your one-stop custom antibody service Antibody is a kind of protein that is the most widely used in the fields of biology and medicine. Antibodies, as the preparations for disease
Compromise Elsewhere Protocols. Western Blotting Methods. 800-656-ROCK www.rockland-inc.com [email protected] 1 of 11
 Compromise Elsewhere Protocols Western Blotting Methods [email protected] 1 of 11 Odyssey Western Blotting Protocols Odyssey reagents: Additional reagents needed: IR-labeled secondary antibodies Odyssey
Compromise Elsewhere Protocols Western Blotting Methods [email protected] 1 of 11 Odyssey Western Blotting Protocols Odyssey reagents: Additional reagents needed: IR-labeled secondary antibodies Odyssey
1.Gene Synthesis. 2.Peptide & Phospho-P. Assembly PCR. Design & Synthesis. Advantages. Specifications. Advantages
 1.Gene Synthesis Assembly PCR Looking for a cdna for your research but could not fish out the gene through traditional cloning methods or a supplier? Abnova provides a gene synthesis service via assembly
1.Gene Synthesis Assembly PCR Looking for a cdna for your research but could not fish out the gene through traditional cloning methods or a supplier? Abnova provides a gene synthesis service via assembly
RPCI 004 v.002 Staining Procedure For all Directly Conjugated Reagents (Whole Blood Method)
 Immune Tolerance Network RPCI 004 v.002 Staining Procedure For all Directly Conjugated Reagents (Whole Blood Method) Author: Paul Wallace, Director, RPCI Laboratory of Flow Cytometry Approved by: Paul
Immune Tolerance Network RPCI 004 v.002 Staining Procedure For all Directly Conjugated Reagents (Whole Blood Method) Author: Paul Wallace, Director, RPCI Laboratory of Flow Cytometry Approved by: Paul
Antibody Services. Best Guarantees in the Industry! Monoclonal Antibody Services. Polyclonal Antibody Services. Express Antibody TM Services
 Antibody Services Best Guarantees in the Industry! Monoclonal Antibody Services Polyclonal Antibody Services Express Antibody TM Services Guaranteed Antibody Services Antibody Purification Services Your
Antibody Services Best Guarantees in the Industry! Monoclonal Antibody Services Polyclonal Antibody Services Express Antibody TM Services Guaranteed Antibody Services Antibody Purification Services Your
Comparing the Tube and Gel Techniques for ABO Antibody Titration, as Performed in Three European Centers 林 良 豐 100.07.18
 Comparing the Tube and Gel Techniques for ABO Antibody Titration, as Performed in Three European Centers 林 良 豐 100.07.18 Introduction Data from 60 consecutive ABO-incompatible kidney transplantations performed
Comparing the Tube and Gel Techniques for ABO Antibody Titration, as Performed in Three European Centers 林 良 豐 100.07.18 Introduction Data from 60 consecutive ABO-incompatible kidney transplantations performed
TECHNICAL BULLETIN. FluoroTag FITC Conjugation Kit. Product Number FITC1 Storage Temperature 2 8 C
 FluoroTag FITC Conjugation Kit Product Number FITC1 Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description The FluoroTag FITC Conjugation Kit is suitable for the conjugation of polyclonal and
FluoroTag FITC Conjugation Kit Product Number FITC1 Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description The FluoroTag FITC Conjugation Kit is suitable for the conjugation of polyclonal and
Methods for Protein Analysis
 Methods for Protein Analysis 1. Protein Separation Methods The following is a quick review of some common methods used for protein separation: SDS-PAGE (SDS-polyacrylamide gel electrophoresis) separates
Methods for Protein Analysis 1. Protein Separation Methods The following is a quick review of some common methods used for protein separation: SDS-PAGE (SDS-polyacrylamide gel electrophoresis) separates
How To Use An Antibody
 GUIDELINES FOR THE PRODUCTION OF ANTIBODIES IN LABORATORY ANIMALS Table of Contents 1. Purpose 2. Choice of Species and Strain 3. Immunizing Antigen 4. Procedures for Polyclonal Antibody Production 5.
GUIDELINES FOR THE PRODUCTION OF ANTIBODIES IN LABORATORY ANIMALS Table of Contents 1. Purpose 2. Choice of Species and Strain 3. Immunizing Antigen 4. Procedures for Polyclonal Antibody Production 5.
Western Blotting. Prepare samples:
 Western Blotting Sive Lab Protocol March 2007 Prepare samples: For zebrafish embryos: Option 1: Take live embryos and put into 1.5 ml tube with E3. Centrifuge gently for 1-2 minutes -yolk lipids will rise
Western Blotting Sive Lab Protocol March 2007 Prepare samples: For zebrafish embryos: Option 1: Take live embryos and put into 1.5 ml tube with E3. Centrifuge gently for 1-2 minutes -yolk lipids will rise
TECHNICAL BULLETIN. HIS-Select Nickel Affinity Gel. Catalog Number P6611 Storage Temperature 2 8 C
 HIS-Select Nickel Affinity Gel Catalog Number P6611 Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description HIS-Select Nickel Affinity Gel is an immobilized metalion affinity chromatography (IMAC)
HIS-Select Nickel Affinity Gel Catalog Number P6611 Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description HIS-Select Nickel Affinity Gel is an immobilized metalion affinity chromatography (IMAC)
Chromatin Immunoprecipitation (ChIP)
 Chromatin Immunoprecipitation (ChIP) Day 1 A) DNA shearing 1. Samples Dissect tissue (One Mouse OBs) of interest and transfer to an eppendorf containing 0.5 ml of dissecting media (on ice) or PBS but without
Chromatin Immunoprecipitation (ChIP) Day 1 A) DNA shearing 1. Samples Dissect tissue (One Mouse OBs) of interest and transfer to an eppendorf containing 0.5 ml of dissecting media (on ice) or PBS but without
Chromatin Immunoprecipitation
 Chromatin Immunoprecipitation A) Prepare a yeast culture (see the Galactose Induction Protocol for details). 1) Start a small culture (e.g. 2 ml) in YEPD or selective media from a single colony. 2) Spin
Chromatin Immunoprecipitation A) Prepare a yeast culture (see the Galactose Induction Protocol for details). 1) Start a small culture (e.g. 2 ml) in YEPD or selective media from a single colony. 2) Spin
How To Make A Tri Reagent
 TRI Reagent For processing tissues, cells cultured in monolayer or cell pellets Catalog Number T9424 Store at room temperature. TECHNICAL BULLETIN Product Description TRI Reagent is a quick and convenient
TRI Reagent For processing tissues, cells cultured in monolayer or cell pellets Catalog Number T9424 Store at room temperature. TECHNICAL BULLETIN Product Description TRI Reagent is a quick and convenient
INSTRUCTION Probemaker
 INSTRUCTION Probemaker Instructions for Duolink In Situ Probemaker PLUS (Art. no. 92009-0020) and Duolink In Situ Probemaker MINUS (Art. no. 92010-0020) Table of content 1. Introduction 4 2. Applications
INSTRUCTION Probemaker Instructions for Duolink In Situ Probemaker PLUS (Art. no. 92009-0020) and Duolink In Situ Probemaker MINUS (Art. no. 92010-0020) Table of content 1. Introduction 4 2. Applications
WESTERN BLOT PROTOCOL FOR LICOR ODYSSEY SCANNER (HAKE S LAB)
 WESTERN BLOT PROTOCOL FOR LICOR ODYSSEY SCANNER (HAKE S LAB) WESTERN BLOT FOR ANALYSIS ON LICOR ODYSSEY SCANNER. 1) The Licor Odyssey protein marker is optimal as it is visible on channel 700 (2ul is enough
WESTERN BLOT PROTOCOL FOR LICOR ODYSSEY SCANNER (HAKE S LAB) WESTERN BLOT FOR ANALYSIS ON LICOR ODYSSEY SCANNER. 1) The Licor Odyssey protein marker is optimal as it is visible on channel 700 (2ul is enough
From Biology to Discovery
 POLYCLONAL ANTIBODY PRODUCTION SERVICES Abbiotec offers custom polyclonal antibody production services to the scientific community to accelerate a project From Biology to Discovery. When the antibody for
POLYCLONAL ANTIBODY PRODUCTION SERVICES Abbiotec offers custom polyclonal antibody production services to the scientific community to accelerate a project From Biology to Discovery. When the antibody for
Aviva Systems Biology
 Aviva Custom Antibody Service and Price Mouse Monoclonal Antibody Service Package Number Description Package Contents Time Price Customer provides antigen protein $6,174 Monoclonal package1 (From protein
Aviva Custom Antibody Service and Price Mouse Monoclonal Antibody Service Package Number Description Package Contents Time Price Customer provides antigen protein $6,174 Monoclonal package1 (From protein
TABLE OF CONTENT. Page ACKNOWLEDGEMENTS. iii ENGLISH ABSTRACT THAI ABSTRACT. vii LIST OF TABLES LIST OF FIGURES. xvi ABBREVIATIONS.
 x TABLE OF CONTENT ACKNOWLEDGEMENTS ENGLISH ABSTRACT THAI ABSTRACT LIST OF TABLES LIST OF FIGURES ABBREVIATIONS iii iv vii xv xvi xviii CHAPTER I: INTRODUCTION 1.1 Statement of problems 1 1.2 Literature
x TABLE OF CONTENT ACKNOWLEDGEMENTS ENGLISH ABSTRACT THAI ABSTRACT LIST OF TABLES LIST OF FIGURES ABBREVIATIONS iii iv vii xv xvi xviii CHAPTER I: INTRODUCTION 1.1 Statement of problems 1 1.2 Literature
CHAPTER 2 ANTIGEN/ANTIBODY INTERACTIONS
 CHAPTER 2 ANTIGEN/ANTIBODY INTERACTIONS See APPENDIX (1) THE PRECIPITIN CURVE; (2) LABELING OF ANTIBODIES The defining characteristic of HUMORAL immune responses (which distinguishes them from CELL-MEDIATED
CHAPTER 2 ANTIGEN/ANTIBODY INTERACTIONS See APPENDIX (1) THE PRECIPITIN CURVE; (2) LABELING OF ANTIBODIES The defining characteristic of HUMORAL immune responses (which distinguishes them from CELL-MEDIATED
SDS-PAGE Protocol Mutated from the SDS-PAGE protocol written by the Lord of the Flies
 SDS-PAGE Protocol Mutated from the SDS-PAGE protocol written by the Lord of the Flies Pouring the resolving gel 1. Clean glass plates with soap and water, then with ethanol. Assemble the glass plates and
SDS-PAGE Protocol Mutated from the SDS-PAGE protocol written by the Lord of the Flies Pouring the resolving gel 1. Clean glass plates with soap and water, then with ethanol. Assemble the glass plates and
Approaches that can be used to study expression of specific proteins
 Approaches that can be used to study expression of specific proteins Receptors and transporters Homogenate binding studies Receptor autoradiography Radiochemical Western blotting Immunohistochemistry/cytochemistry
Approaches that can be used to study expression of specific proteins Receptors and transporters Homogenate binding studies Receptor autoradiography Radiochemical Western blotting Immunohistochemistry/cytochemistry
Covalent Conjugation to Cytodiagnostics Carboxylated Gold Nanoparticles Tech Note #105
 Covalent Conjugation to Cytodiagnostics Carboxylated Gold Nanoparticles Tech Note #105 Background Gold nanoparticle conjugates have been widely used in biological research and biosensing applications.
Covalent Conjugation to Cytodiagnostics Carboxylated Gold Nanoparticles Tech Note #105 Background Gold nanoparticle conjugates have been widely used in biological research and biosensing applications.
WHO Prequalification of Diagnostics Programme PUBLIC REPORT. Product: Genscreen ULTRA HIV Ag-Ab Number: PQDx 0096-031-00. Abstract
 WHO Prequalification of Diagnostics Programme PUBLIC REPORT Product: Genscreen ULTRA HIV Ag-Ab Number: PQDx 0096-031-00 Abstract Genscreen ULTRA HIV Ag-Ab with product codes 72386 and 72388, manufactured
WHO Prequalification of Diagnostics Programme PUBLIC REPORT Product: Genscreen ULTRA HIV Ag-Ab Number: PQDx 0096-031-00 Abstract Genscreen ULTRA HIV Ag-Ab with product codes 72386 and 72388, manufactured
The immune response Antibodies Antigens Epitopes (antigenic determinants) the part of a protein antigen recognized by an antibody Haptens small
 The immune response Antibodies Antigens Epitopes (antigenic determinants) the part of a protein antigen recognized by an antibody Haptens small molecules that can elicit an immune response when linked
The immune response Antibodies Antigens Epitopes (antigenic determinants) the part of a protein antigen recognized by an antibody Haptens small molecules that can elicit an immune response when linked
INTERPRETATION INFORMATION SHEET
 Creative Testing Solutions 2424 West Erie Dr. 2205 Highway 121 10100 Martin Luther King Jr. St. No. Tempe, AZ 85282 Bedford, TX 76021 St. Petersburg, FL 33716 INTERPRETATION INFORMATION SHEET Human Immunodeficiency
Creative Testing Solutions 2424 West Erie Dr. 2205 Highway 121 10100 Martin Luther King Jr. St. No. Tempe, AZ 85282 Bedford, TX 76021 St. Petersburg, FL 33716 INTERPRETATION INFORMATION SHEET Human Immunodeficiency
Fast Semi-Dry Transfer System with Yrdimes
 Fast Semi-Dry Transfer System with Yrdimes INTRODUCTION Yrdimes is a semi-dry transfer system for Western blotting mainly used for fast and efficient blotting by using less transfer buffer. It is used
Fast Semi-Dry Transfer System with Yrdimes INTRODUCTION Yrdimes is a semi-dry transfer system for Western blotting mainly used for fast and efficient blotting by using less transfer buffer. It is used
Outline. 1. Experiment. 2. Sample analysis and storage. 3. Image analysis and presenting data. 4. Probemaker
 Tips and tricks Note: this is just an informative document with general recommendations. Please contact [email protected] should you have any queries. Document last reviewed 2011-11-17 Outline 1. Experiment
Tips and tricks Note: this is just an informative document with general recommendations. Please contact [email protected] should you have any queries. Document last reviewed 2011-11-17 Outline 1. Experiment
Electrophoresis and Electroblotting of Proteins
 Electrophoresis and Electroblotting of Proteins The purpose of the next lab exercises will be to study the relative amounts of β-actin in cells of the B-16 melanoma, liver and muscle of mice. Electrophoresis
Electrophoresis and Electroblotting of Proteins The purpose of the next lab exercises will be to study the relative amounts of β-actin in cells of the B-16 melanoma, liver and muscle of mice. Electrophoresis
Biology 309 Lab Notebook
 Name: Biology 309 Lab Notebook This is a guided lab notebook for you to keep well-organized notes about procedures and record experimental data for experiments as they are performed. It is guided because,
Name: Biology 309 Lab Notebook This is a guided lab notebook for you to keep well-organized notes about procedures and record experimental data for experiments as they are performed. It is guided because,
Hypoxyprobe -1 Plus Kit Kit contents:
 Updated 2015 1 PRODUCT INSERT Hypoxyprobe, Inc 121 Middlesex Turnpike Burlington, MA 01803 USA www.hypoxyprobe.com Hypoxyprobe -1 Plus Kit Kit contents: Solid pimonidazole HCl (Hypoxyprobe -1) FITC conjugated
Updated 2015 1 PRODUCT INSERT Hypoxyprobe, Inc 121 Middlesex Turnpike Burlington, MA 01803 USA www.hypoxyprobe.com Hypoxyprobe -1 Plus Kit Kit contents: Solid pimonidazole HCl (Hypoxyprobe -1) FITC conjugated
Virological Methods. Flint et al. Principles of Virology (ASM), Chapter 2
 Virological Methods Flint et al. Principles of Virology (ASM), Chapter 2 Overview The most commonly used laboratory methods for the detection of viruses and virus components in biological samples can be
Virological Methods Flint et al. Principles of Virology (ASM), Chapter 2 Overview The most commonly used laboratory methods for the detection of viruses and virus components in biological samples can be
Mouse krebs von den lungen 6 (KL-6) ELISA
 KAMIYA BIOMEDICAL COMPANY Mouse krebs von den lungen 6 (KL-6) ELISA For the quantitative determination of mouse KL-6 in serum, plasma, cell culture supernatants, body fluid and tissue homogenate Cat. No.
KAMIYA BIOMEDICAL COMPANY Mouse krebs von den lungen 6 (KL-6) ELISA For the quantitative determination of mouse KL-6 in serum, plasma, cell culture supernatants, body fluid and tissue homogenate Cat. No.
Geniron. Custom Antibody Services. Serum services Antibody Monoclonal. Purification Antibody Mono Y Genetic Immunization Genbody Polyclonal Antibody
 Geniron Custom Antibody Services Serum services Antibody Monoclonal Purification Antibody Mono Y Genetic Immunization Genbody Polyclonal Antibody Geniron Poly Y WE PROVIDE OUR SERVICES TO With Expertise
Geniron Custom Antibody Services Serum services Antibody Monoclonal Purification Antibody Mono Y Genetic Immunization Genbody Polyclonal Antibody Geniron Poly Y WE PROVIDE OUR SERVICES TO With Expertise
Antibody Production Price List
 Antibody Production Price List Presenting Insight Biotechnology s price list for custom polyclonal and monoclonal antibody production services. We are happy to tailor individual packages towards the specific
Antibody Production Price List Presenting Insight Biotechnology s price list for custom polyclonal and monoclonal antibody production services. We are happy to tailor individual packages towards the specific
SDS-PAGE. (June 23, 2005)
 SDS-PAGE (June 23, 2005) GATHER REAGENTS & MATERIALS 30% acrylamide/0.8% bisacrylamide (30:1) 4X Tris. Cl/SDS, ph 8.8 4X Tris. Cl/SDS, ph 6.8 Ammonium persulfate, 10% (Make fresh each time.) SDS electrophoresis
SDS-PAGE (June 23, 2005) GATHER REAGENTS & MATERIALS 30% acrylamide/0.8% bisacrylamide (30:1) 4X Tris. Cl/SDS, ph 8.8 4X Tris. Cl/SDS, ph 6.8 Ammonium persulfate, 10% (Make fresh each time.) SDS electrophoresis
METHOD USED TO EXTRACT TOTAL MUSCLE PROTEIN FOR WESTERN BLOT USING TRIS-EDTA BUFFER*
 METHOD USED TO EXTRACT TOTAL MUSCLE PROTEIN FOR WESTERN BLOT USING TRIS-EDTA BUFFER* SOLUTIONS FOR SAMPLE EXTRACTION 1. Tris-EDTA Buffer, ph 8.3 1 liter 50 mm Tris 6.06 g 10 mm EDTA 3.72 g Adjust ph to
METHOD USED TO EXTRACT TOTAL MUSCLE PROTEIN FOR WESTERN BLOT USING TRIS-EDTA BUFFER* SOLUTIONS FOR SAMPLE EXTRACTION 1. Tris-EDTA Buffer, ph 8.3 1 liter 50 mm Tris 6.06 g 10 mm EDTA 3.72 g Adjust ph to
Western Blot Protocol (updated on 05/20/14)
 Western Blot Protocol (updated on 05/20/14) Required Solutions 10x PBS (1L) 80 g NaCl 2 g KCl 14.4 g Na 2 HPO 4 or 22 g Na 2 HPO 4 7H 2 O 2.4 g KH 2 PO 4 or 2 g KH 2 PO4 Adjust ph to 7.4 Autoclave PBST
Western Blot Protocol (updated on 05/20/14) Required Solutions 10x PBS (1L) 80 g NaCl 2 g KCl 14.4 g Na 2 HPO 4 or 22 g Na 2 HPO 4 7H 2 O 2.4 g KH 2 PO 4 or 2 g KH 2 PO4 Adjust ph to 7.4 Autoclave PBST
KMS-Specialist & Customized Biosimilar Service
 KMS-Specialist & Customized Biosimilar Service 1. Polyclonal Antibody Development Service KMS offering a variety of Polyclonal Antibody Services to fit your research and production needs. we develop polyclonal
KMS-Specialist & Customized Biosimilar Service 1. Polyclonal Antibody Development Service KMS offering a variety of Polyclonal Antibody Services to fit your research and production needs. we develop polyclonal
Lyme (IgG and IgM) Antibody Confirmation
 Pathology & Laboratory Medicine Lyme (IgG and IgM) Antibody Confirmation TEST UPDATE: New Test Notification Date: 1/9/2013 Effective Date: 1/7/2013 CONTACT INFO Call 802-847-5121 800-991-2799 email [email protected]
Pathology & Laboratory Medicine Lyme (IgG and IgM) Antibody Confirmation TEST UPDATE: New Test Notification Date: 1/9/2013 Effective Date: 1/7/2013 CONTACT INFO Call 802-847-5121 800-991-2799 email [email protected]
VETERINARY SERVICES MEMORANDUM NO. 800.90
 August 5, 1998 VETERINARY SERVICES MEMORANDUM NO. 800.90 Subject: To: Guidelines for Veterinary Biological Relative Potency Assays and Reference Preparations Based on ELISA Antigen Quantification Veterinary
August 5, 1998 VETERINARY SERVICES MEMORANDUM NO. 800.90 Subject: To: Guidelines for Veterinary Biological Relative Potency Assays and Reference Preparations Based on ELISA Antigen Quantification Veterinary
PROTOCOL. Immunocytochemistry (ICC) MATERIALS AND EQUIPMENT REQUIRED
 PROTOCOL Immunocytochemistry (ICC) 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 11-07 MATERIALS AND EQUIPMENT REQUIRED Materials: MitoSciences primary monoclonal antibody/antibodies Fluorophore-conjugated
PROTOCOL Immunocytochemistry (ICC) 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 11-07 MATERIALS AND EQUIPMENT REQUIRED Materials: MitoSciences primary monoclonal antibody/antibodies Fluorophore-conjugated
Antibody Purification and Labeling
 5 Antibody Purification and Labeling 5.1 Antibody Purification 65 Magne Protein A Beads and Magne Protein G Beads 67 5.2 Antibody Labeling 69 phab Amine and Thiol Reactive Dyes 71 63 Discover Reliable
5 Antibody Purification and Labeling 5.1 Antibody Purification 65 Magne Protein A Beads and Magne Protein G Beads 67 5.2 Antibody Labeling 69 phab Amine and Thiol Reactive Dyes 71 63 Discover Reliable
PROTOCOL. Immunostaining for Flow Cytometry. Background. Materials and equipment required.
 PROTOCOL Immunostaining for Flow Cytometry 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 Rev.0 Background The combination of single cell analysis using flow cytometry and the specificity of antibody-based
PROTOCOL Immunostaining for Flow Cytometry 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 Rev.0 Background The combination of single cell analysis using flow cytometry and the specificity of antibody-based
Western Blot Protocol Protein isolation
 Western Blot Protocol Protein isolation A. Preparation of cell lysates. - Preparation of materials: -Dial the microcentrifuge temperature control setting to 4 C -Prepare a bucket of ice -Prepare lysis
Western Blot Protocol Protein isolation A. Preparation of cell lysates. - Preparation of materials: -Dial the microcentrifuge temperature control setting to 4 C -Prepare a bucket of ice -Prepare lysis
Diagnosis of HIV-1 Infection. Estelle Piwowar-Manning HPTN Central Laboratory The Johns Hopkins University
 Diagnosis of HIV-1 Infection Estelle Piwowar-Manning HPTN Central Laboratory The Johns Hopkins University Tests Used to Diagnose HIV-1 Infection HIV antibody (today s topic) HIV p24 antigen HIV DNA HIV
Diagnosis of HIV-1 Infection Estelle Piwowar-Manning HPTN Central Laboratory The Johns Hopkins University Tests Used to Diagnose HIV-1 Infection HIV antibody (today s topic) HIV p24 antigen HIV DNA HIV
Mouse Typer Sub-Isotyping Kit Instruction Manual
 Mouse Typer Sub-Isotyping Kit Instruction Manual Protocol for Mouse Typer Sub-Isotyping Kit (Catalog Number 172-2051) and Mouse Typer Sub Isotyping Panel (Catalog Number 172-2055) For Technical Service
Mouse Typer Sub-Isotyping Kit Instruction Manual Protocol for Mouse Typer Sub-Isotyping Kit (Catalog Number 172-2051) and Mouse Typer Sub Isotyping Panel (Catalog Number 172-2055) For Technical Service
