Elderly: in elderly patients there is no need to reduce the daily dosage (see section 5.2).
|
|
|
- Ross Dickerson
- 9 years ago
- Views:
Transcription
1 Mesulid-DL-Feb Doctor Leaflet 1. NAME OF THE MEDICINAL PRODUCT Mesulid 100 mg caplets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each caplet contains 100mg nimesulide. For excipients, see section PHARMACEUTICAL FORM Off-white to light yellow capsule-shaped tablet. 4. CLINICAL PARTICULARS 4.1 Therapeutic indications Treatment of acute pain. Primary dysmenorrhoea. Nimesulide should only be prescribed as second line treatment. The decision to prescribe nimesulide should be based on assessment of the individual patient's overall risks (see section 4.3 and 4.4). 4.2 Posology and method of administration Mesulid should be used for the shortest possible duration, as required by the clinical situation. Moreover, undesirable effects may be minimized by using the minimum effective dose for the shortest duration necessary to control symptoms (see section 4.4). The maximum duration of a treatment course with nimesulide is 15 days. Adults: One 100mg caplet twice a day after meals. Elderly: in elderly patients there is no need to reduce the daily dosage (see section 5.2). Children (< 12 years): Mesulid is contraindicated in these patients (see also section 4.3). Adolescents (from 12 to 18 years): on the basis of the kinetic profile in adults and on the pharmacodynamic characteristics of nimesulide, no dosage adjustment in these patients is necessary. Impaired renal function: on the basis of pharmacokinetics, no dosage adjustment is necessary in patients with mild to moderate renal impairment (creatinine clearance of ml/min), while Mesulid is contraindicated in case of severe renal impairement (creatinine clearance<30 ml/min) (see sections 4.3 and 5.2). Hepatic impairment: the use of Mesulid is contraindicated in patients with hepatic impairment (see sections 4.3 and 5.2). 4.3 Contraindications Hypersensitivity to nimesulide or to any of the excipients. History of hypersensitivity reactions (e.g. bronchospasm, rhinitis, urticaria, nasal polyps)) in response to acetylsalicylic acid or other non-steroidal anti-inflammatory drugs. History of hepatotoxic reactions to nimesulide. Concomitant exposure to other potentially hepatotoxic substances. Alcoholism, drug addiction. History of gastrointestinal bleeding or perforation, related to previous NSAIDs therapy. Active, or history of recurrent peptic ulcer/haemorrhage (two or more distinct episodes of proven ulceration or bleeding). cerebrovascular bleeding or other active bleeding or bleeding disorders. 1
2 Severe coagulation disorders. Severe heart failure. Severe renal impairment. Hepatic impairment. Patients with fever and / or flu-like symptoms. Children under 12 years. The third trimester of pregnancy and breastfeeding (see sections 4.6 and 5.3). 4.4 Special warnings and precautions for use The use of Mesulid with concomitant NSAIDs including cyclooxygenase-2 selective inhibitors should be avoided. In addition, patients should be advised to refrain from other concomitant analgesics. Undesirable effects may be minimised by using the minimum effective dose for the shortest duration necessary to control symptoms (see section 4.2). Treatment should be discontinued if no benefit is seen. Hepatic effects Rarely Mesulid has been reported to be associated with serious hepatic reactions, including very rare fatal cases (see also section 4.8). Patients who experience symptoms compatible with hepatic injury during treatment with Mesulid (e.g. anorexia, nausea, vomiting, abdominal pain, fatigue, dark urine) or patients who develop abnormal liver function tests should have treatment discontinued. These patients should not be rechallenged with nimesulide. Liver damage, in most cases reversible, has been reported following short exposure to the drug. Patients receiving nimesulide who develop fever and / or flu-like symptoms should discontinue treatment. Gastrointestinal effects Gastrointestinal bleeding, ulceration and perforation: GI bleeding, ulceration or perforation, which can be fatal, has been reported with all NSAIDs at anytime during treatment, with or without warning symptoms or a previous history of GI events. The risk of GI bleeding, ulceration or perforation is higher with increasing NSAID doses, in patients with a history of ulcer, particularly if complicated with haemorrhage or perforation (see section 4.3), and in the elderly. These patients should commence treatment on the lowest dose available. Combination therapy with protective agents (e.g. misoprostol or proton pump inhibitors) should be considered for these patients, and also for patients requiring concomitant low dose aspirin, or other drugs likely to increase gastrointestinal risk (see below and 4.5). Patients with a history of GI toxicity, particularly when elderly, should report any unusual abdominal symptoms (especially GI bleeding), particularly in the initial stages of treatment. Gastrointestinal bleeding or ulceration / perforation can occur at any time during treatment with or without warning symptoms or a previous history of gastrointestinal events. If gastrointestinal bleeding or ulceration occurs, nimesulide should be discontinued. Nimesulide should be used with caution in patients with gastrointestinal disorders, including history of peptic ulceration, history of gastrointestinal haemorrhage, ulcerative colitis or Crohn s disease. Caution should be advised in patients receiving concomitant medications which could increase the risk of ulceration or bleeding, such as oral corticosteroids, anticoagulants such as warfarin, selective serotoninreuptake inhibitors or antiplatelet agents such as aspirin (see section 4.5). When GI bleeding or ulceration occurs in patients receiving Mesulid the treatment should be withdrawn. NSAIDs should be given with care to patients with a history of gastrointestinal disease (ulcerative colitis, Crohn s disease) as their condition may be exacerbated (see section 4.8 undesirable effects). Elderly: The elderly have an increased frequency of adverse reactions to NSAIDs, especially gastrointestinal bleeding and perforation which may be fatal (see section 4.2). Therefore, appropriate clinical monitoring is advisable. Cardiovascular and cerebrovascular effects Appropriate monitoring and advice are required for patients with a history of hypertension and/or mild to moderate congestive heart failure as fluid retention and oedema have been reported in association with NSAID therapy. Page 2 of 8
3 Clinical trial and epidemiological data suggest that use of some NSAIDs (particularly at high doses and in long term treatment) may be associated with a small increased risk of arterial thrombotic events (for example, myocardial infarction or stroke). There are insufficient data to exclude such a risk for Mesulid. Patients with uncontrolled hypertension, congestive heart failure, established ischaemic heart disease, peripheral arterial disease, and/or cerebrovascular disease should only be treated with Mesulid after careful consideration. Similar consideration should be made before initiating longer-term treatment of patients with risk factors for cardiovascular disease (e.g. hypertension, hyperlipidaemia, diabetes mellitus, smoking). As nimesulide can interfere with platelet function, it should be used with caution in patients with bleeding diathesis (see also section 4.3). However, Mesulid is not a substitute for acetylsalicylic acid for cardiovascular prophylaxis. Renal effects In patients with renal or cardiac impairment, caution is required since the use of Mesulid may result in deterioration of renal function. In the event of deterioration, the treatment should be discontinued (see also section 4.5). Skin Reactions Serious skin reactions, some of them fatal, including exfoliative dermatitis, Stevens-Johnson syndrome, and toxic epidermal necrolysis, have been reported very rarely in association with the use of NSAIDs (see section 4.8). Patients appear to be at highest risk of these reactions early in the course of therapy, the onset of the reaction occurring in the majority of cases within the first month of treatment. Mesulid should be discontinued at the first appearance of skin rash, mucosal lesions, or any other sign of hypersensitivity. Fertility effects The use of Mesulid may impair female fertility and is not recommended in women attempting to conceive. In women who have difficulties conceiving or who are undergoing investigation of infertility, withdrawal of Mesulid should be considered (see section 4.6). Mesulid contain lactose, therefore patients with rare hereditary problems of galactose intolerance, the Lapp lactase deficiency or glucose-galactose malabsorption should not take this medicine. 4.5 Interaction with other medicinal products and other forms of interaction Pharmacodynamic interactions Other non steroidal anti-inflammatory drugs (NSAIDs) : The combined use of Mesulid (see section 4.4) with other non steroidal anti-inflammatory drugs, including acetylsalicylic acid given at anti-inflammatory doses ( 1g as single intake or 3g as total daily amount) is not recommended. Corticosteroids Increased risk of gastrointestinal ulceration or bleeding (see section 4.4). Anti-coagulants: NSAIDs may enhance the effects of anti-coagulants, such as warfarin (see section 4.4). Patients receiving warfarin or similar anticoagulant agents or acetylsalicylic acid have an increased risk of bleeding complications, when treated with Mesulid. Therefore this combination is not recommended (see also section 4.4) and is contraindicated in patients with severe coagulation disorders (see also section 4.3). If the combination cannot be avoided, anticoagulant activity should be monitored closely. Anti-platelet agents and selective serotonin reuptake inhibitors (SSRIs): increased risk of gastrointestinal bleeding (see section 4.4). Diuretics, Angiotensin Conversion Enzyme Inhibitors (ACE inhibitors) and Angiotensin II Antagonists (AIIA): NSAIDs may reduce the efficacy of diuretics and that of other antihypertensive drugs. In some patients with reduced renal function (e.g. dehydrated patients or elderly subjects with impairment of renal function), concomitant administration of an ACE inhibitor and cyclo-oxygenase inhibitors may result in progression of the deterioration of renal function, including the possibility of acute renal insufficiency, which is normally reversible. Page 3 of 8
4 The occurrence of these interactions should be taken into consideration in patients who have to take Mesulid in association with ACE inhibitors or AIIA. Consequently, this drug association should be administered with precaution, especially in elderly patients. Patients should be properly hydrated, and the need for monitoring of renal function after starting the concomitant treatment and periodically after that should be analysed. Pharmacokinetic interactions: effect of nimesulide on the pharmacokinetics of other drugs. Furosemide: In healthy subjects, nimesulide transiently decreases the effect of furosemide on sodium excretion and, to a lesser extent, on potassium excretion and reduces the diuretic response. Co-administration of nimesulide and furosemide results in a decrease (of about 20%) of the AUC and cumulative excretion of furosemide, without affecting its renal clearance. The concomitant use of furosemide and Mesulid requires caution in susceptible renal or cardiac patients, as described under section 4.4. Lithium: Non-steroidal anti-inflammatory drugs have been reported to reduce the clearance of lithium, resulting in elevated plasma levels and lithium toxicity. If Mesulid is prescribed for a patient receiving lithium therapy, lithium levels should be monitored closely. Potential pharmacokinetic interactions with glibenclamide, theophylline, warfarin, digoxin, cimetidine and an antacid preparation (i.e. a combination of aluminium and magnesium hydroxide) were also studied in vivo. No clinically significant interactions were observed. Nimesulide inhibits CYP2C9. The plasma concentrations of drugs that are substrates of this enzyme may be increased when Mesulid is used concomitantly. Caution is required if nimesulide is used less than 24 hours before or after treatment with methotrexate because the serum level of methotrexate might increase and therefore, the toxicity of this drug might increase. Due to their effect on renal prostaglandins, prostaglandin synthetase inhibitors like nimesulide may increase the nephrotoxicity of cyclosporines. Pharmacokinetic Interactions: Effects of other drugs on the pharmacokinetics of nimesulide: In vitro studies have shown displacement of nimesulide from binding sites by tolbutamide, salicylic acid and valproic acid. However, despite a possible effect on plasma levels, these interactions have not demonstrated clinical significance. 4.6 Fertility, Pregnancy and lactation The use of Mesulid is contraindicated in the third trimester of pregnancy (see section 4.3). Like other NSAIDs, Mesulid is not recommended in women attempting to conceive (see section 4.4). Inhibition of prostaglandin synthesis may have a negative impact on pregnancy and/or embryonic/fetal development. Results of epidemiological studies suggest an increased risk of miscarriage and of cardiac malformation and gastroschisis after the use of an inhibitor of prostaglandin synthesis in the first stage of pregnancy. The absolute risk for cardiac malformations was increased from less than 1% to approximately 1.5%. The risk has been considered to increase with the dose and duration of treatment. In animals, administration of inhibitors of prostaglandin synthesis has been shown to provoke an increase in pre- and postimplantation loss and in embryonic-fetal mortality. Furthermore, an increased incidence of various malformations, including the cardiovascular one, has been reported in animals to which inhibitors of prostaglandin synthesis were administered during the period of organogenesis. Studies in rabbits have shown an atypical reproductive toxicity (see section 5.3) and no adequate data from the use of nimesulide-containing medicinal products in pregnant women are available. Therefore, the potential risk for humans is unknown and prescribing the drug during the first two trimesters of pregnancy is not recommended, except in cases where it is strictly necessary. If Mesulid is used by a woman who is trying to conceive, or during the first and second trimesters of pregnancy, the dose and duration of treatment should be kept as low as possible. During the third trimester of pregnancy, all inhibitors of prostaglandin synthesis may expose the fetus to: Page 4 of 8
5 - cardiopulmonary toxicity (with premature closure of the ductus arteriosus and pulmonary hypertension); - renal dysfunction, which may progress to renal insufficiency with oligohydramnios; the mother and the newborn infant, at the end of pregnancy, to: - possible prolongation of bleeding time, and an antiplatelet effect which may occur even at very low doses; - inhibition of uterine contractions resulting in delay or prolongation of labour. Consequently, Mesulid is contraindicated during the third trimester of pregnancy. Lactation: It is not known whether nimesulide is excreted in human milk. Mesulid is contraindicated when breastfeeding (see sections 4.3 and 5.3). 4.7 Effects on ability to drive and use machines No studies on the effect of Mesulid on the ability to drive or use machines have been performed. However, patients who experience dizziness, vertigo or somnolence after receiving Mesulid should refrain from driving or operating machines. 4.8 Undesirable effects a) General Description Clinical trial and epidemiological data suggest that use of some NSAIDs (particularly at high doses and in long term treatment) may be associated with a small increased risk of arterial thrombotic events (for example myocardial infarction or stroke) (see section 4.4). Oedema, hypertension, and cardiac failure have been reported in association with NSAID treatment. Very rare cases of bullous reactions including Stevens Johnson Syndrom and Toxic Epidermal Necrolysis have been reported. The most commonly-observed adverse events are gastrointestinal in nature. Peptic ulcers, perforation or GI bleeding, sometimes fatal, particularly in the elderly, may occur (see section 4.4). Nausea, vomiting, diarrhoea, flatulence, constipation, dyspepsia, abdominal pain, melaena, haematemesis, ulcerative stomatitis, exacerbation of colitis and Crohn s disease (see section Special warnings and precautions for use) have been reported following administration. Less frequently, gastritis has been observed. b) Table of adverse reactions The following listing of undesirable effects is based on data from controlled clinical trials* (approximately 7,800 patients) and from post marketing surveillance with reporting rates classified as: very common (>1/10); common (>1/100, <1/10); uncommon (>1/1,000, <1/100); rare (>1/10,000, <1/1,000); very rare (<1/10,000), including isolated cases. Blood disorders Rare Anemia* Eosinophilia* Very rare Thrombocytopenia Pancytopenia Purpura Immune system disorders Rare Hypersensitivity* Very rare Anaphylaxis Metabolism and nutrition disorders Rare Hyperkalemia* Psychiatric disorders Rare Anxiety* Nervousness* Nightmare* Nervous system disorders Uncommon Dizziness* Very rare Headache Somnolence Encephalopathy (Reye s syndrome) Eye disorders Rare Vision blurred* Very rare Visual disturbance Ear and labyrinth disorders Very rare Vertigo Cardiac disorders Rare Tachycardia* Vascular disorders Uncommon Hypertension* Page 5 of 8
6 Rare Respiratory disorders Uncommon Dyspnoea* Very rare Haemorrhage* Blood pressure fluctuation* Hot flushes* Asthma Bronchospasm Gastrointestinal disorders Common Diarrhoea* Nausea* Vomiting* Hepato-biliary disorders (see section 4.4. Special warnings and special precautions for use ) Uncommon Very rare Common Very rare Constipation* Flatulence* Gastrointestinal bleeding Duodenal ulcer and perforation Gastric ulcer and perforation Gastritis* Abdominal pain Dyspepsia Stomatitis Melaena Skin and subcutaneous tissue disorders Uncommon Pruritus* Rash* Rare Very rare Renal and urinary disorders Rare Dysuria* Haematuria* Hepatic enzymes increased* Hepatitis Fulminant hepatitis (including fatal cases) Jaundice Cholestasis Sweating increased* Erythema* Dermatitis* Urticaria Angioneurotic oedema Face oedema Erythema multiforme Stevens Johnson syndrome Toxic epidermal necrolysis Very rare Urinary retention* Renal failure Oliguria Interstitial nephritis General disorders Uncommon Oedema* Rare Malaise* Asthenia* Very rare Hypothermia *frequency based on clinical trial 4.9 Overdose Symptoms following acute NSAID overdoses are usually limited to lethargy, drowsiness, nausea, vomiting and epigastric pain, which are generally reversible with supportive care. Gastrointestinal bleeding can occur. Hypertension, acute renal failure, respiratory depression and coma may occur, but are rare. Anaphylactoid reactions have been reported with therapeutic ingestion of NSAIDs, and may occur following an overdose. Patients should be managed by symptomatic and supportive care following an NSAID overdose. There are no specific antidotes. No information is available regarding the removal of nimesulide by haemodialysis, but based on its high degree of plasma protein binding (up to 97.5%) dialysis is unlikely to be useful in overdose. Emesis and/or activated charcoal (60 to 100 g in adults) and/or osmotic cathartic may be indicated in patients seen within 4 hours of ingestion with symptoms or following a large overdose. Forced diuresis, alkalinization of Page 6 of 8
7 urine, haemodialysis, or haemoperfusion may not be useful due to high protein binding. Renal and hepatic function should be monitored. 5. PHARMACOLOGICAL PROPERTIES 5.1 Pharmacodynamic properties Pharmacotherapeutic group: ATC code: M01AX17 Nimesulide is a non-steroidal anti-inflammatory drug with analgesic and antipyretic properties which acts as an inhibitor of prostaglandin synthesis enzyme cyclo-oxygenase. 5.2 Pharmacokinetic properties Nimesulide is well absorbed when given per os. After a single dose of 100mg nimesulide a peak plasma level of 3-4 mg/l is reached in adults after 2-3 hours. AUC = mg h/l. No statistically significant difference has been found between these figures and those seen after 100mg given twice daily for 7 days. Up to 97.5% binds to plasma proteins. Nimesulide is extensively metabolised in the liver following multiple pathways, including cytochrome P450 (CYP) 2C9 isoenzymes. Therefore, there is a potential for a drug interaction with concomitant administration of drugs which are metabolised by CYP2C9 (see under section 4.5). The main metabolite is the para-hydroxy derivative which is also pharmacologically active. The lag time before the appearance of this metabolite in the circulation is short (about 0.8 hour) but its formation constant is not high and is considerably lower than the absorption constant of nimesulide. Hydroxynimesulide is the only metabolite found in plasma and it is almost completely conjugated. T ½ is between 3.2 and 6 hours. Nimesulide is excreted mainly in the urine (approximately 50% of the administered dose). Only 1-3% is excreted as the unmodified compound. Hydroxynimesulide, the main metabolite, is found only as a glucuronate. Approximately 29% of the dose is excreted after metabolism in the faeces. The kinetic profile of nimesulide was unchanged in the elderly after acute and repeated doses. In an acute experimental study carried out in patients with mild to moderate renal impairment (creatinine clearance ml/min) versus healthy volunteers, peak plasma levels of nimesulide and its main metabolite were not higher than in healthy volunteers. AUC and t 1/2 beta were 50% higher, but were always within the range of kinetic values observed with nimesulide in healthy volunteers. Repeated administration did not cause accumulation. Nimesulide is contra-indicated in patients with hepatic impairment (see section 4.3). 5.3 Preclinical safety data Preclinical data reveal no special hazards for humans based on conventional studies of safety pharmacology, repeated dose toxicity, genotoxicity and carcinogenic potential. In repeated dose toxicity studies, nimesulide showed gastrointestinal, renal and hepatic toxicity. In reproductive toxicity studies, embryotoxic and teratogenic effects (skeletal malformations, dilatation of cerebral ventricles) were observed in rabbits, but not in rats, at maternally non-toxic dose levels. In rats, increased mortality of offspring was observed in the early postnatal period and nimesulide showed adverse effects on fertility. 6. PHARMACEUTICAL PARTICULARS 6.1 List of excipients Cellulose microcrystalline, docusate sodium, hydrogenated castor oil, hydroxypropyl cellulose, lactose, magnesium stearate, sodium starch glycolate. 6.2 Incompatibilities Not applicable. 6.3 Special precautions for storage Store below 25 C. 6.4 Nature and contents of the container Page 7 of 8
8 Blister packs (10, 20, 30 caplets). 6.5 Special precautions for disposal Not applicable. 7. MARKETING AUTHORISATION HOLDER RAFA LABORATORIES LTD., P. O. Box 450, Jerusalem Registration number: Manufactured under license from Helsinn Healthcare SA, Switzerland. The format of this leaflet was determined by the Ministry of Health that checked and approved its content in February Page 8 of 8
SUMMARY OF PRODUCT CHARACTERISTICS BREXIN. 3. Pharmaceutical Form Tablet Pale yellow, hexagonal tablet with a median score line on one side
 פורמט עלון זה נקבע ע"י משרד הבריאות ותוכנו נבדק ואושר SUMMARY OF PRODUCT CHARACTERISTICS 1. Name of the Medicinal Product BREXIN 20 mg TABLETS BREXIN 2. Qualitative and Quantitative Composition Each tablet
פורמט עלון זה נקבע ע"י משרד הבריאות ותוכנו נבדק ואושר SUMMARY OF PRODUCT CHARACTERISTICS 1. Name of the Medicinal Product BREXIN 20 mg TABLETS BREXIN 2. Qualitative and Quantitative Composition Each tablet
PACKAGE INSERT TEMPLATE FOR ACETYLSALICYLIC ACID/ASPIRIN TABLET
 PACKAGE INSERT TEMPLATE FOR ACETYLSALICYLIC ACID/ASPIRIN TABLET Brand or Product Name [Product name] tablet 500mg Name and Strength of Active Substance(s) Acetylsalicylic acid.500mg Product Description
PACKAGE INSERT TEMPLATE FOR ACETYLSALICYLIC ACID/ASPIRIN TABLET Brand or Product Name [Product name] tablet 500mg Name and Strength of Active Substance(s) Acetylsalicylic acid.500mg Product Description
patient group direction
 DICLOFENAC v01 1/8 DICLOFENAC PGD Details Version 1.0 Legal category Staff grades Approved by POM Paramedic (Non-ECP) Nurse (Non-ECP) Emergency Care Practitioner (Paramedic) Emergency Care Practitioner
DICLOFENAC v01 1/8 DICLOFENAC PGD Details Version 1.0 Legal category Staff grades Approved by POM Paramedic (Non-ECP) Nurse (Non-ECP) Emergency Care Practitioner (Paramedic) Emergency Care Practitioner
There is a risk of renal impairment in dehydrated children and adolescents.
 PACKAGE LEAFLET: INFORMATION FOR THE USER MELFEN 200mg FILM-COATED TABLETS MELFEN 400mg FILM-COATED TABLETS Ibuprofen Read all of this leaflet carefully before you start taking this medicine because it
PACKAGE LEAFLET: INFORMATION FOR THE USER MELFEN 200mg FILM-COATED TABLETS MELFEN 400mg FILM-COATED TABLETS Ibuprofen Read all of this leaflet carefully before you start taking this medicine because it
PARACETAMOL REXIDOL. 600 mg Tablet. Analgesic-Antipyretic. Paracetamol 600 mg
 (Insert Text) UL Consumer Health PARACETAMOL REXIDOL 600 mg Tablet Analgesic-Antipyretic FORMULATION Each tablet contains: Paracetamol 600 mg PRODUCT DESCRIPTION Rexidol is a round, yellow, flat, bevel-edged
(Insert Text) UL Consumer Health PARACETAMOL REXIDOL 600 mg Tablet Analgesic-Antipyretic FORMULATION Each tablet contains: Paracetamol 600 mg PRODUCT DESCRIPTION Rexidol is a round, yellow, flat, bevel-edged
UpRight Aceclofenac 100 mg and Paracetamol 500 mg fixed dose combination
 For the use of a Registered Medical Practitioner or a Hospital or a Laboratory only UpRight Aceclofenac 100 mg and Paracetamol 500 mg fixed dose combination DESCRIPTION UPRIGHT is a fixed dose combination
For the use of a Registered Medical Practitioner or a Hospital or a Laboratory only UpRight Aceclofenac 100 mg and Paracetamol 500 mg fixed dose combination DESCRIPTION UPRIGHT is a fixed dose combination
Essential Shared Care Agreement Drugs for Dementia
 Ref No. E040 Essential Shared Care Agreement Drugs for Dementia Please complete the following details: Patient s name, address, date of birth Consultant s contact details (p.3) And send One copy to: 1.
Ref No. E040 Essential Shared Care Agreement Drugs for Dementia Please complete the following details: Patient s name, address, date of birth Consultant s contact details (p.3) And send One copy to: 1.
2. QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Ibuprofen Tablets BP 200mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 200mg of Ibuprofen. Excipients with known
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Ibuprofen Tablets BP 200mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 200mg of Ibuprofen. Excipients with known
Doxylamine succinate belongs to the ethanolamine class of antihistamines with sedative properties.
 Data Sheet MERSYNDOL Tablet Paracetamol 450mg per tablet Codeine Phosphate 9.75mg per tablet Doxylamine Succinate 5mg per tablet MERSYNDOL FORTE Tablet Paracetamol 450mg per tablet Codeine Phosphate 30mg
Data Sheet MERSYNDOL Tablet Paracetamol 450mg per tablet Codeine Phosphate 9.75mg per tablet Doxylamine Succinate 5mg per tablet MERSYNDOL FORTE Tablet Paracetamol 450mg per tablet Codeine Phosphate 30mg
White, circular, biconvex, uncoated tablets with a score line on one side, plain on the other.
 Nausicalm Cyclizine hydrochloride Ph. Eur. 50 mg Presentation White, circular, biconvex, uncoated tablets with a score line on one side, plain on the other. Uses Actions The active ingredient-cyclizine
Nausicalm Cyclizine hydrochloride Ph. Eur. 50 mg Presentation White, circular, biconvex, uncoated tablets with a score line on one side, plain on the other. Uses Actions The active ingredient-cyclizine
IBUPROFEN 200 MG COATED TABLETS. (Ibuprofen) PL 03105/0102-3 UK PAR TABLE OF CONTENTS
 IBUPROFEN 200 MG COATED TABLETS (Ibuprofen) PL 03105/0102-3 UK PAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 11 Summary of Product Characteristics
IBUPROFEN 200 MG COATED TABLETS (Ibuprofen) PL 03105/0102-3 UK PAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 11 Summary of Product Characteristics
See 17 for PATIENT COUNSELING INFORMATION. Revised: 3/2016 FULL PRESCRIBING INFORMATION: CONTENTS* WARNING: ADRENAL CRISIS IN THE SETTING OF SHOCK OR
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LYSODREN safely and effectively. See full prescribing information for LYSODREN. LYSODREN (mitotane)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LYSODREN safely and effectively. See full prescribing information for LYSODREN. LYSODREN (mitotane)
ZOVIRAX Cold Sore Cream
 Data Sheet ZOVIRAX Cold Sore Cream Aciclovir 5% w/w Presentation Topical cream Indications ZOVIRAX Cold Sore Cream is indicated for the treatment of Herpes simplex virus infections of the lips and face
Data Sheet ZOVIRAX Cold Sore Cream Aciclovir 5% w/w Presentation Topical cream Indications ZOVIRAX Cold Sore Cream is indicated for the treatment of Herpes simplex virus infections of the lips and face
NSAID PREPARATIONS. COMPOSITION : Each enteric coated tablet contains Diclofenac Sodium BP 25 mg. Clofenac 50. Clofenac SR. Sodium BP 50 mg.
 Diclofenac Analgesic & Anti-inflammatory COMPOSITION Clofenac 25 : Each enteric coated tablet contains Diclofenac Sodium BP 25 mg. Clofenac 50 : Each enteric coated tablet contains Diclofenac Sodium BP
Diclofenac Analgesic & Anti-inflammatory COMPOSITION Clofenac 25 : Each enteric coated tablet contains Diclofenac Sodium BP 25 mg. Clofenac 50 : Each enteric coated tablet contains Diclofenac Sodium BP
Public Assessment Report
 Public Assessment Report IBUPROFEN 200 MG TABLETS EXTRA STRENGTH IBUPROFEN 400 MG TABLETS (IBUPROFEN) PL 17907/0159-61 MHRA PAR Ibuprofen Tablets PL 17907/0159-61 - 1 - IBUPROFEN TABLETS (IBUPROFEN) PL
Public Assessment Report IBUPROFEN 200 MG TABLETS EXTRA STRENGTH IBUPROFEN 400 MG TABLETS (IBUPROFEN) PL 17907/0159-61 MHRA PAR Ibuprofen Tablets PL 17907/0159-61 - 1 - IBUPROFEN TABLETS (IBUPROFEN) PL
INITIATING ORAL AUBAGIO (teriflunomide) THERAPY
 FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
PRESCRIBING INFORMATION Refer to Summary of Product Characteristics (SmPC) before Prescribing
 EZETROL (ezetimibe) PRESCRIBING INFORMATION Refer to Summary of Product Characteristics (SmPC) before Prescribing Adverse events should be reported. Reporting forms and information can be found at www.mhra.gov.uk/yellowcard.
EZETROL (ezetimibe) PRESCRIBING INFORMATION Refer to Summary of Product Characteristics (SmPC) before Prescribing Adverse events should be reported. Reporting forms and information can be found at www.mhra.gov.uk/yellowcard.
SUMMARY OF PRODUCT CHARACTERISTICS. Paracetamol...10.00 mg for 1 ml of solution for infusion
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Paracetamol...10.00 mg for 1 ml of
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Paracetamol...10.00 mg for 1 ml of
NEUROTONE THR 00904/0005 UKPAR
 NEUROTONE THR 00904/0005 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 10 Summary of Product Characteristics Page 11 Product Information Leaflet
NEUROTONE THR 00904/0005 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 10 Summary of Product Characteristics Page 11 Product Information Leaflet
UK Public Assessment Report
 IBUPROFEN 200MG TABLETS (ibuprofen) PL 31308/0013-17 UK Public Assessment Report TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 11 Summary of Product
IBUPROFEN 200MG TABLETS (ibuprofen) PL 31308/0013-17 UK Public Assessment Report TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 11 Summary of Product
IBUPROFEN 400MG TABLETS PL 20395/0080 UKPAR TABLE OF CONTENTS
 IBUPROFEN 400MG TABLETS PL 20395/0080 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 11 Steps taken after authorisation summary Summary of Product
IBUPROFEN 400MG TABLETS PL 20395/0080 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 11 Steps taken after authorisation summary Summary of Product
CODEINE PHOSPHATE 15 MG TABLETS CODEINE PHOSPHATE 30 MG TABLETS CODEINE PHOSPHATE 60 MG TABLETS PL 24837/0023-5 UK PAR TABLE OF CONTENTS
 CODEINE PHOSPHATE 15 MG TABLETS CODEINE PHOSPHATE 30 MG TABLETS CODEINE PHOSPHATE 60 MG TABLETS PL 24837/0023-5 UK PAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for
CODEINE PHOSPHATE 15 MG TABLETS CODEINE PHOSPHATE 30 MG TABLETS CODEINE PHOSPHATE 60 MG TABLETS PL 24837/0023-5 UK PAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for
The format of this leaflet was determined by the Ministry of Health and its content was checked and approved in May 2013.
 The format of this leaflet was determined by the Ministry of Health and its content was checked and approved in May 2013 Havrix 720 Junior 1 NAME OF THE MEDICINAL PRODUCT Havrix 720 Junior 2 QUALITATIVE
The format of this leaflet was determined by the Ministry of Health and its content was checked and approved in May 2013 Havrix 720 Junior 1 NAME OF THE MEDICINAL PRODUCT Havrix 720 Junior 2 QUALITATIVE
MIGRAINE RELIEF 342 MG TABLETS PL 12063/0071 UKPAR TABLE OF CONTENTS
 MIGRAINE RELIEF 342 MG TABLETS PL 12063/0071 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 12 Steps taken after authorisation summary Page 13 Summary
MIGRAINE RELIEF 342 MG TABLETS PL 12063/0071 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 12 Steps taken after authorisation summary Page 13 Summary
PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement)
 1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
Package leaflet: Information for the user. < ASPROFLASH and associated names > 500 mg coated tablet Acetylsalicylic acid
 PACKAGE LEAFLET 1 Package leaflet: Information for the user < ASPROFLASH and associated names > 500 mg coated tablet Acetylsalicylic acid Read all of this leaflet carefully before you start using this
PACKAGE LEAFLET 1 Package leaflet: Information for the user < ASPROFLASH and associated names > 500 mg coated tablet Acetylsalicylic acid Read all of this leaflet carefully before you start using this
It can therefore be bought without a prescription but should be used correctly to ensure its efficacy and to reduce side effects.
 140 mg Medicated plaster Diclofenac Sodium BEFORE USE, CAREFULLY READ ALL THE INFORMATION GIVEN ON THE INFORMATIVE LEAFLET This medicinal product is intended for SELF-MEDICATION. It can be used to treat
140 mg Medicated plaster Diclofenac Sodium BEFORE USE, CAREFULLY READ ALL THE INFORMATION GIVEN ON THE INFORMATIVE LEAFLET This medicinal product is intended for SELF-MEDICATION. It can be used to treat
PRODUCT INFORMATION PANAMAX
 NAME OF THE MEDICINE Non-proprietary Name Paracetamol PRODUCT INFORMATION PANAMAX DESCRIPTION Each tablet contains paracetamol 500 mg. The inactive ingredients are: maize starch, purified talc, pregelatinised
NAME OF THE MEDICINE Non-proprietary Name Paracetamol PRODUCT INFORMATION PANAMAX DESCRIPTION Each tablet contains paracetamol 500 mg. The inactive ingredients are: maize starch, purified talc, pregelatinised
Public Assessment Report. Decentralised Procedure
 Public Assessment Report Decentralised Procedure Nurofen Express Soluble 400mg Oral Powder UK/H/3534 & 3996/001/DC UK licence no: PL 00063/0611 & 0616 Reckitt Benckiser Healthcare (UK) Limited 1 LAY SUMMARY
Public Assessment Report Decentralised Procedure Nurofen Express Soluble 400mg Oral Powder UK/H/3534 & 3996/001/DC UK licence no: PL 00063/0611 & 0616 Reckitt Benckiser Healthcare (UK) Limited 1 LAY SUMMARY
Core Safety Profile. Pharmaceutical form(s)/strength: 50mg tablets IE/H/PSUR/0028/002 Date of FAR: 17.07.2013
 Core Safety Profile Active substance: Naltrexone Hydrochloride Pharmaceutical form(s)/strength: 50mg tablets P-RMS: IE/H/PSUR/0028/002 Date of FAR: 17.07.2013 4.2 Posology and method of administration
Core Safety Profile Active substance: Naltrexone Hydrochloride Pharmaceutical form(s)/strength: 50mg tablets P-RMS: IE/H/PSUR/0028/002 Date of FAR: 17.07.2013 4.2 Posology and method of administration
PACKAGE LEAFLET: INFORMATION FOR THE USER. PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion. Paracetamol
 PACKAGE LEAFLET: INFORMATION FOR THE USER PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion Paracetamol Read all of this leaflet carefully before you start using this medicine. Keep this leaflet.
PACKAGE LEAFLET: INFORMATION FOR THE USER PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion Paracetamol Read all of this leaflet carefully before you start using this medicine. Keep this leaflet.
SR 5. W2364A_NT.XATRAL SR5 PAK/SA 5/12/05 9:47 Page 1
 W2364A_NT.XATRAL SR5 PAK/SA 5/12/05 9:47 Page 1 alfuzosin SR 5 mg (Alfuzosin HCI) COMPOSITION Each sustained-release, film-coated tablet contains: Alfuzosin hydrochloride......... 5 mg Excipients... q.s.
W2364A_NT.XATRAL SR5 PAK/SA 5/12/05 9:47 Page 1 alfuzosin SR 5 mg (Alfuzosin HCI) COMPOSITION Each sustained-release, film-coated tablet contains: Alfuzosin hydrochloride......... 5 mg Excipients... q.s.
RELONCHEM HAYFEVER AND ALLERGY 10 MG TABLETS PL 20395/0088 UKPAR TABLE OF CONTENTS
 RELONCHEM HAYFEVER AND ALLERGY 10 MG TABLETS PL 20395/0088 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 11 Steps taken after authorisation summary
RELONCHEM HAYFEVER AND ALLERGY 10 MG TABLETS PL 20395/0088 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 11 Steps taken after authorisation summary
NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate.
 NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate. PRESENTATION Eye Drops: NAPHCON-A Eye Drops are a combination of an antihistamine (pheniramine maleate) and a decongestant
NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate. PRESENTATION Eye Drops: NAPHCON-A Eye Drops are a combination of an antihistamine (pheniramine maleate) and a decongestant
Patient Group Direction Hospital: Bristol Royal Infirmary Department: UHBristol Thrombosis Service University Hospitals Bristol NHS Foundation Trust.
 Patient Group Direction Hospital: Bristol Royal Infirmary Department: UHBristol Thrombosis Service University Hospitals Bristol NHS Foundation Trust. This Patient Group Direction (PGD) has been written
Patient Group Direction Hospital: Bristol Royal Infirmary Department: UHBristol Thrombosis Service University Hospitals Bristol NHS Foundation Trust. This Patient Group Direction (PGD) has been written
P AC K AG E L E AF L E T: INFORMAT I ON FO R THE USER. 500 mg, film-coated tablet Active substance: metformin hydrochloride
 P AC K AG E L E AF L E T: INFORMAT I ON FO R THE USER Siofor 500 500 mg, film-coated tablet Active substance: metformin hydrochloride For use in children above 10 years and adults Read all of this leaflet
P AC K AG E L E AF L E T: INFORMAT I ON FO R THE USER Siofor 500 500 mg, film-coated tablet Active substance: metformin hydrochloride For use in children above 10 years and adults Read all of this leaflet
Rivaroxaban: Amber Drug Guidance for the prevention of stroke and systemic embolism in patients with non-valvular AF
 Leeds Rivaroxaban: Amber Drug Guidance for the prevention of stroke and systemic embolism in patients with non-valvular AF Amber Drug Level 3 (amber drug with monitoring requirements) We have started your
Leeds Rivaroxaban: Amber Drug Guidance for the prevention of stroke and systemic embolism in patients with non-valvular AF Amber Drug Level 3 (amber drug with monitoring requirements) We have started your
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Progit 50 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each film-coated tablet contains 50 mg of itopride hydrochloride.
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Progit 50 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each film-coated tablet contains 50 mg of itopride hydrochloride.
Summary of Product Characteristics
 1 NAME OF THE MEDICINAL PRODUCT Librium 10mg Hard capsule Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each capsule contains 10 mg of chlordiazepoxide hydrochloride. Excipients:
1 NAME OF THE MEDICINAL PRODUCT Librium 10mg Hard capsule Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each capsule contains 10 mg of chlordiazepoxide hydrochloride. Excipients:
LEFLUNOMIDE (Adults)
 Shared Care Guideline DRUG: Introduction: LEFLUNOMIDE (Adults) Indication: Disease modifying drug for rheumatoid arthritis and psoriatic arthritis Licensing Information: Disease modifying drug for active
Shared Care Guideline DRUG: Introduction: LEFLUNOMIDE (Adults) Indication: Disease modifying drug for rheumatoid arthritis and psoriatic arthritis Licensing Information: Disease modifying drug for active
EFFIMET 1000 XR Metformin Hydrochloride extended release tablet
 BRAND NAME: Effimet XR. THERAPEUTIC CATEGORY: Anti-Diabetic PHARMACOLOGIC CLASS: Biguanides EFFIMET 1000 XR Metformin Hydrochloride extended release tablet COMPOSITION AND PRESENTATION Composition Each
BRAND NAME: Effimet XR. THERAPEUTIC CATEGORY: Anti-Diabetic PHARMACOLOGIC CLASS: Biguanides EFFIMET 1000 XR Metformin Hydrochloride extended release tablet COMPOSITION AND PRESENTATION Composition Each
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Prednicare Tablets 5mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains: Active Substance(s) Prednisolone
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Prednicare Tablets 5mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains: Active Substance(s) Prednisolone
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Eskazole Tablets 400 mg 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Albendazole 400 mg Excipients with known effect: Lactose, sunset yellow
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Eskazole Tablets 400 mg 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Albendazole 400 mg Excipients with known effect: Lactose, sunset yellow
Adjunctive psychosocial intervention. Conditions requiring dose reduction. Immediate, peak plasma concentration is reached within 1 hour.
 Shared Care Guideline for Prescription and monitoring of Naltrexone Hydrochloride in alcohol dependence Author(s)/Originator(s): (please state author name and department) Dr Daly - Consultant Psychiatrist,
Shared Care Guideline for Prescription and monitoring of Naltrexone Hydrochloride in alcohol dependence Author(s)/Originator(s): (please state author name and department) Dr Daly - Consultant Psychiatrist,
SUMMARY OF PRODUCT CHARACTERISTICS. Albuman 200 g/l is a solution containing 200 g/l (20%) of total protein of which at least 95% is human albumin.
 Albuman 200 g/l SPC 01 December 2015 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Albuman 200 g/l solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Albuman 200 g/l
Albuman 200 g/l SPC 01 December 2015 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Albuman 200 g/l solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Albuman 200 g/l
Nōdia DESCRIPTION PHARMACOLOGY. Loperamide hydrochloride USP 2 mg Tablets. Pharmacological classification - antidiarrhoeal.
 Nōdia Loperamide hydrochloride USP 2 mg Tablets DESCRIPTION Nōdia 2 mg tablets are green, capsule-shaped tablets with a break-line on one side. Each tablet contains 2 mg loperamide hydrochloride and typically
Nōdia Loperamide hydrochloride USP 2 mg Tablets DESCRIPTION Nōdia 2 mg tablets are green, capsule-shaped tablets with a break-line on one side. Each tablet contains 2 mg loperamide hydrochloride and typically
1. NAME OF THE MEDICINAL PRODUCT. Impavido 10 mg capsules Impavido 50 mg capsules 2. QUALITATIVE AND QUANTITATIVE COMPOSITION. 1 capsule contains:
 1. NAME OF THE MEDICINAL PRODUCT Impavido 10 mg capsules Impavido 50 mg capsules 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 capsule contains: Impavido 10 mg capsules 10 mg Miltefosine. Impavido 50 mg
1. NAME OF THE MEDICINAL PRODUCT Impavido 10 mg capsules Impavido 50 mg capsules 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 capsule contains: Impavido 10 mg capsules 10 mg Miltefosine. Impavido 50 mg
EMEA PUBLIC STATEMENT ON LEFLUNOMIDE (ARAVA) - SEVERE AND SERIOUS HEPATIC REACTIONS -
 The European Agency for the Evaluation of Medicinal Products Post-authorisation evaluation of medicines for human use London, 12 March 2001 Doc. Ref: EMEA/H/5611/01/en EMEA PUBLIC STATEMENT ON LEFLUNOMIDE
The European Agency for the Evaluation of Medicinal Products Post-authorisation evaluation of medicines for human use London, 12 March 2001 Doc. Ref: EMEA/H/5611/01/en EMEA PUBLIC STATEMENT ON LEFLUNOMIDE
European Medicines Agency recommends restricting use of trimetazidine-containing medicines
 22 June 2012 EMA/CHMP/417861/2012 Press Office Press release European Medicines Agency recommends restricting use of trimetazidine-containing medicines Restricted indication for patients with stable angina
22 June 2012 EMA/CHMP/417861/2012 Press Office Press release European Medicines Agency recommends restricting use of trimetazidine-containing medicines Restricted indication for patients with stable angina
Dabigatran: Amber Drug Guidance for the prevention of stroke and systemic embolism in patients with non-valvular AF
 Leeds Dabigatran: Amber Drug Guidance for the prevention of stroke and systemic embolism in patients with non-valvular AF Amber Drug Level 3 (amber drug with monitoring requirements) We have started your
Leeds Dabigatran: Amber Drug Guidance for the prevention of stroke and systemic embolism in patients with non-valvular AF Amber Drug Level 3 (amber drug with monitoring requirements) We have started your
DATA SHEET. This product is may not be interchangeable with other products containing this ingredient in the New Zealand Market.
 DATA SHEET METFORMIN GENERIC HEALTH Metformin hydrochloride 500mg and 850mg tablets This product is may not be interchangeable with other products containing this ingredient in the New Zealand Market.
DATA SHEET METFORMIN GENERIC HEALTH Metformin hydrochloride 500mg and 850mg tablets This product is may not be interchangeable with other products containing this ingredient in the New Zealand Market.
HYPERTENSION ASSOCIATED WITH RENAL DISEASES
 RENAL DISEASE v Patients with renal insufficiency should be encouraged to reduce dietary salt and protein intake. v Target blood pressure is less than 135-130/85 mmhg. If patients have urinary protein
RENAL DISEASE v Patients with renal insufficiency should be encouraged to reduce dietary salt and protein intake. v Target blood pressure is less than 135-130/85 mmhg. If patients have urinary protein
PATIENT INFORMATION LEAFLET. CEFALEXIN 250 mg AND 500 mg CAPSULES CEFALEXIN
 PATIENT INFORMATION LEAFLET CEFALEXIN 250 mg AND 500 mg CAPSULES CEFALEXIN Read all of this leaflet carefully before you start taking this medicine. - Keep this leaflet. You may need to read it again.
PATIENT INFORMATION LEAFLET CEFALEXIN 250 mg AND 500 mg CAPSULES CEFALEXIN Read all of this leaflet carefully before you start taking this medicine. - Keep this leaflet. You may need to read it again.
5.07.09. Aubagio. Aubagio (teriflunomide) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.07.09 Subject: Aubagio Page: 1 of 6 Last Review Date: December 5, 2014 Aubagio Description Aubagio (teriflunomide)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.07.09 Subject: Aubagio Page: 1 of 6 Last Review Date: December 5, 2014 Aubagio Description Aubagio (teriflunomide)
Thioctacid 600 T Solution for Injection contains 600 mg alpha-lipoic acid
 Package Leaflet: Information for the User Thioctacid 600 T Solution for Injection contains 600 mg alpha-lipoic acid For use in adults Active substance: Alpha-lipoic acid, Trometamol salt (1:1) Read all
Package Leaflet: Information for the User Thioctacid 600 T Solution for Injection contains 600 mg alpha-lipoic acid For use in adults Active substance: Alpha-lipoic acid, Trometamol salt (1:1) Read all
OMNIC OCAS Film coated Tablets SAJA PHARMA
 08-15 OMNIC OCAS Film coated Tablets SAJA PHARMA Tamsulosin Hydrochloride 0.4 mg film-coated prolonged release tablets 1. NAME OF THE MEDICINAL PRODUCT OMNIC OCAS, 0.4 mg prolonged release tablets, film-coated.
08-15 OMNIC OCAS Film coated Tablets SAJA PHARMA Tamsulosin Hydrochloride 0.4 mg film-coated prolonged release tablets 1. NAME OF THE MEDICINAL PRODUCT OMNIC OCAS, 0.4 mg prolonged release tablets, film-coated.
Shared Care Guideline for the use of Leflunomide for Rheumatoid Arthritis
 Shared Care Guideline for the use of Leflunomide for Rheumatoid Section 1: Agreement for transfer of prescribing to GP Please sign this form and return it to the named consultant if you are willing to
Shared Care Guideline for the use of Leflunomide for Rheumatoid Section 1: Agreement for transfer of prescribing to GP Please sign this form and return it to the named consultant if you are willing to
Ibuprofen 10% w/w gel Nurofen Maximum Strength 10 % Gel
 Ibuprofen 10% w/w gel Nurofen Maximum Strength 10 % Gel PL 10972/0089 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 11 Summary of Product Characteristics
Ibuprofen 10% w/w gel Nurofen Maximum Strength 10 % Gel PL 10972/0089 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 11 Summary of Product Characteristics
sodium [0-(2,6-dicloroanilino)phenyl] acetate Molecular formula:
![sodium [0-(2,6-dicloroanilino)phenyl] acetate Molecular formula: sodium [0-(2,6-dicloroanilino)phenyl] acetate Molecular formula:](/thumbs/26/8144647.jpg) PRODUCT INFORMATION ARTHROTEC 50 (Diclofenac sodium/misoprostol) NAME OF THE MEDICINE ARTHROTEC 50 Diclofenac sodium 50 mg/misoprostol 200 µg tablets. Diclofenac sodium The structural formula of diclofenac
PRODUCT INFORMATION ARTHROTEC 50 (Diclofenac sodium/misoprostol) NAME OF THE MEDICINE ARTHROTEC 50 Diclofenac sodium 50 mg/misoprostol 200 µg tablets. Diclofenac sodium The structural formula of diclofenac
Teriflunomide is the active metabolite of Leflunomide, a drug employed since 1994 for the treatment of rheumatoid arthritis (Baselt, 2011).
 Page 1 of 10 ANALYTE NAME AND STRUCTURE TERIFLUNOMIDE Teriflunomide TRADE NAME Aubagio CATEGORY Antimetabolite TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND sclerosis. The
Page 1 of 10 ANALYTE NAME AND STRUCTURE TERIFLUNOMIDE Teriflunomide TRADE NAME Aubagio CATEGORY Antimetabolite TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND sclerosis. The
PACKAGE LEAFLET: INFORMATION FOR THE USER Paracetamol 500 mg Effervescent Tablets Paracetamol
 PACKAGE LEAFLET: INFORMATION FOR THE USER Paracetamol 500 mg Effervescent Tablets Paracetamol Read all of this leaflet carefully because it contains important information for you. This medicine is available
PACKAGE LEAFLET: INFORMATION FOR THE USER Paracetamol 500 mg Effervescent Tablets Paracetamol Read all of this leaflet carefully because it contains important information for you. This medicine is available
SUMMARY OF PRODUCT CHARACTERISTICS. Each tablet contains aspirin 75mg. Also contains lactose.
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Dispersible Aspirin 75mg Tablets BP 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains aspirin 75mg. Also contains lactose.
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Dispersible Aspirin 75mg Tablets BP 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains aspirin 75mg. Also contains lactose.
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Diazepam Tablets BP 2mg 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Diazepam BP 2.00mg 3 PHARMACEUTICAL FORM Tablet 4. CLINICAL PARTICULARS
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Diazepam Tablets BP 2mg 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Diazepam BP 2.00mg 3 PHARMACEUTICAL FORM Tablet 4. CLINICAL PARTICULARS
Diabetic nephropathy is detected clinically by the presence of persistent microalbuminuria or proteinuria.
 Kidney Complications Diabetic Nephropathy Diabetic nephropathy is detected clinically by the presence of persistent microalbuminuria or proteinuria. The peak incidence of nephropathy is usually 15-25 years
Kidney Complications Diabetic Nephropathy Diabetic nephropathy is detected clinically by the presence of persistent microalbuminuria or proteinuria. The peak incidence of nephropathy is usually 15-25 years
Medication Guide Plavix (PLAV-iks) (clopidogrel bisulfate) tablets
 Medication Guide Plavix (PLAV-iks) (clopidogrel bisulfate) tablets Read this Medication Guide before you start taking Plavix and each time you get a refill. There may be new information. This Medication
Medication Guide Plavix (PLAV-iks) (clopidogrel bisulfate) tablets Read this Medication Guide before you start taking Plavix and each time you get a refill. There may be new information. This Medication
This leaflet answers some common questions about ARROW - MELOXICAM.
 Arrow - Meloxicam Meloxicam Tablets 7.5mg What is in this leaflet This leaflet answers some common questions about ARROW - MELOXICAM. It does not contain all of the available information. It does not take
Arrow - Meloxicam Meloxicam Tablets 7.5mg What is in this leaflet This leaflet answers some common questions about ARROW - MELOXICAM. It does not contain all of the available information. It does not take
PACKAGE LEAFLET: INFORMATION FOR THE USER. ADRENALINE (TARTRATE) STEROP 1 mg/1 ml Solution for injection. Adrenaline (Levorenine, Epinephrine)
 PACKAGE LEAFLET: INFORMATION FOR THE USER ADRENALINE (TARTRATE) STEROP 1 mg/1 ml Solution for injection Adrenaline (Levorenine, Epinephrine) Read all of this leaflet carefully before you start using this
PACKAGE LEAFLET: INFORMATION FOR THE USER ADRENALINE (TARTRATE) STEROP 1 mg/1 ml Solution for injection Adrenaline (Levorenine, Epinephrine) Read all of this leaflet carefully before you start using this
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Aknemycin Solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 10 g of solution contains 0.2 g of erythromycin. Structural formula of
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Aknemycin Solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 10 g of solution contains 0.2 g of erythromycin. Structural formula of
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 Temporary Core Product Information Inde- (13May2013) Page 1 / 7 1 NAME OF THE MEDICINAL PRODUCT XYLONOR SPRAY, oromucosal spray, solution. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each gram contains:
Temporary Core Product Information Inde- (13May2013) Page 1 / 7 1 NAME OF THE MEDICINAL PRODUCT XYLONOR SPRAY, oromucosal spray, solution. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each gram contains:
Summary of Product Characteristics
 1 NAME OF THE MEDICINAL PRODUCT Diamox SR 250mg Modified Release Hard Capsules. Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each capsule contains Acetazolamide 250 mg.
1 NAME OF THE MEDICINAL PRODUCT Diamox SR 250mg Modified Release Hard Capsules. Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each capsule contains Acetazolamide 250 mg.
Novartis Gilenya FDO Program Clinical Protocol and Highlights from Prescribing Information (PI)
 Novartis Gilenya FDO Program Clinical Protocol and Highlights from Prescribing Information (PI) Highlights from Prescribing Information - the link to the full text PI is as follows: http://www.pharma.us.novartis.com/product/pi/pdf/gilenya.pdf
Novartis Gilenya FDO Program Clinical Protocol and Highlights from Prescribing Information (PI) Highlights from Prescribing Information - the link to the full text PI is as follows: http://www.pharma.us.novartis.com/product/pi/pdf/gilenya.pdf
Safety Information Card for Xarelto Patients
 Safety Information Card for Xarelto Patients 15mg Simply Protecting More Patients 20mg Simply Protecting More Patients Keep this card with you at all times Present this card to every physician or dentist
Safety Information Card for Xarelto Patients 15mg Simply Protecting More Patients 20mg Simply Protecting More Patients Keep this card with you at all times Present this card to every physician or dentist
DVT/PE Management with Rivaroxaban (Xarelto)
 DVT/PE Management with Rivaroxaban (Xarelto) Rivaroxaban is FDA approved for the acute treatment of DVT and PE and reduction in risk of recurrence of DVT and PE. FDA approved indications: Non valvular
DVT/PE Management with Rivaroxaban (Xarelto) Rivaroxaban is FDA approved for the acute treatment of DVT and PE and reduction in risk of recurrence of DVT and PE. FDA approved indications: Non valvular
Always take this medicine exactly as described in this leaflet or as your doctor, pharmacist or nurse have told you.
 leaflet: Information for the user Macrogol 4000 10 g powder for oral solution in sachet Macrogol 4000
leaflet: Information for the user Macrogol 4000 10 g powder for oral solution in sachet Macrogol 4000
4 Clinical Particulars
 SUMMARY OF PRODUCT CHARACTERISTICS 1 Name of the Medicinal Product Procyclidine Syrup 5mg/5ml 2. Qualitative and Quantitative Composition Each 5ml dose contains 5mg Procyclidine Hydrochloride BP. 3. Pharmaceutical
SUMMARY OF PRODUCT CHARACTERISTICS 1 Name of the Medicinal Product Procyclidine Syrup 5mg/5ml 2. Qualitative and Quantitative Composition Each 5ml dose contains 5mg Procyclidine Hydrochloride BP. 3. Pharmaceutical
1.3.1.3 PACKAGE LEAFLET
 Sandoz Business use only Page 1 of 8 1.3.1.3 PACKAGE LEAFLET Sandoz Business use only Page 2 of 8 Package leaflet: Information for the patient Cefuhexal 250 mg film-coated tablets Cefuhexal 500 mg film-coated
Sandoz Business use only Page 1 of 8 1.3.1.3 PACKAGE LEAFLET Sandoz Business use only Page 2 of 8 Package leaflet: Information for the patient Cefuhexal 250 mg film-coated tablets Cefuhexal 500 mg film-coated
Nursing 113. Pharmacology Principles
 Nursing 113 Pharmacology Principles 1. The study of how drugs enter the body, reach the site of action, and are removed from the body is called a. pharmacotherapeutics b. pharmacology c. pharmacodynamics
Nursing 113 Pharmacology Principles 1. The study of how drugs enter the body, reach the site of action, and are removed from the body is called a. pharmacotherapeutics b. pharmacology c. pharmacodynamics
Metformin 500 & 850 mg Film-coated Tablets SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Metformine HCl 500 mg ratiopharm, filmomhulde tabletten Metformine HCl 850 mg ratiopharm, filmomhulde tabletten 2. QUALITATIVE AND QUANTITATIVE
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Metformine HCl 500 mg ratiopharm, filmomhulde tabletten Metformine HCl 850 mg ratiopharm, filmomhulde tabletten 2. QUALITATIVE AND QUANTITATIVE
Medication for the Treatment of Alcohol Use Disorder. Pocket Guide
 Medication for the Treatment of Alcohol Use Disorder Pocket Guide Medications are underused in the treatment of alcohol use disorder. According to the National Survey on Drug Use and Health, of the estimated
Medication for the Treatment of Alcohol Use Disorder Pocket Guide Medications are underused in the treatment of alcohol use disorder. According to the National Survey on Drug Use and Health, of the estimated
Public Assessment Report. Meloxicam 7.5mg Orodispersible Tablets Meloxicam 15mg Orodispersible Tablets. Meloxicam PL 31388/0003 PL 31388/0004
 Public Assessment Report Meloxicam 7.5mg Orodispersible Tablets Meloxicam 15mg Orodispersible Tablets Meloxicam PL 31388/0003 PL 31388/0004 Alpex Pharma (UK) Ltd Table of Contents Page Lay Summary 2 Scientific
Public Assessment Report Meloxicam 7.5mg Orodispersible Tablets Meloxicam 15mg Orodispersible Tablets Meloxicam PL 31388/0003 PL 31388/0004 Alpex Pharma (UK) Ltd Table of Contents Page Lay Summary 2 Scientific
**Form 1: - Consultant Copy** Telephone Number: Fax Number: Email: Author: Dr Bernard Udeze Pharmacist: Claire Ault Date of issue July 2011
 Effective Shared Care Agreement for the treatment of Dementia in Alzheimer s Disease Donepezil tablets / orodispersible tablets (Aricept / Aricept Evess ) These forms (1 and 2) are to be completed by both
Effective Shared Care Agreement for the treatment of Dementia in Alzheimer s Disease Donepezil tablets / orodispersible tablets (Aricept / Aricept Evess ) These forms (1 and 2) are to be completed by both
TORADOL ORAL (ketorolac tromethamine tablets)
 1 2 3 4 R x only TORADOL ORAL (ketorolac tromethamine tablets) 5 WARNING 6 TORADOL ORAL (ketorolac tromethamine), a nonsteroidal anti-inflammatory drug 7 (NSAID), is indicated for the short-term (up to
1 2 3 4 R x only TORADOL ORAL (ketorolac tromethamine tablets) 5 WARNING 6 TORADOL ORAL (ketorolac tromethamine), a nonsteroidal anti-inflammatory drug 7 (NSAID), is indicated for the short-term (up to
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 5 mg of amlodipine (as amlodipine maleate).
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 5 mg of amlodipine (as amlodipine maleate).
Xarelto (rivaroxaban) Prescriber Guide
 Xarelto (rivaroxaban) Prescriber Guide Simple Protection For More Patients 2 Xarelto Prescriber Guide Patient Alert Card 4 Dosing Recommendations 4 Dosing in patients with atrial fibrillation 4 Patients
Xarelto (rivaroxaban) Prescriber Guide Simple Protection For More Patients 2 Xarelto Prescriber Guide Patient Alert Card 4 Dosing Recommendations 4 Dosing in patients with atrial fibrillation 4 Patients
Over the Counter Drugs (OTCs): Considerations for Physical Therapy Practice in Canada
 Background Over the Counter Drugs (OTCs): Considerations for Physical Therapy Practice in Canada The use of medications or drugs by non-physician health professionals is evolving and is linked to collaboration
Background Over the Counter Drugs (OTCs): Considerations for Physical Therapy Practice in Canada The use of medications or drugs by non-physician health professionals is evolving and is linked to collaboration
Prescriber Guide. 20mg. 15mg. Simply Protecting More Patients. Simply Protecting More Patients
 Prescriber Guide 20mg Simply Protecting More Patients 15mg Simply Protecting More Patients 1 Dear Doctor, This prescriber guide was produced by Bayer Israel in cooperation with the Ministry of Health as
Prescriber Guide 20mg Simply Protecting More Patients 15mg Simply Protecting More Patients 1 Dear Doctor, This prescriber guide was produced by Bayer Israel in cooperation with the Ministry of Health as
COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON HYPERICUM PERFORATUM L., HERBA (WELL-ESTABLISHED MEDICINAL USE)
 European Medicines Agency Evaluation of Medicines for Human Use London, 12 November 2009 Doc. Ref.: EMA/HMPC/101304/2008 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON
European Medicines Agency Evaluation of Medicines for Human Use London, 12 November 2009 Doc. Ref.: EMA/HMPC/101304/2008 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON
Naloxone Hydrochloride Injection PRODUCT INFORMATION
 Naloxone Hydrochloride Injection PRODUCT INFORMATION DESCRIPTION Naloxone hydrochloride is 17-allyl-4,5α-epoxy-3,14-dihydroxymorphinan-6-one hydrochloride; C 19 H 21 NO 4.HCl. It is an off-white powder
Naloxone Hydrochloride Injection PRODUCT INFORMATION DESCRIPTION Naloxone hydrochloride is 17-allyl-4,5α-epoxy-3,14-dihydroxymorphinan-6-one hydrochloride; C 19 H 21 NO 4.HCl. It is an off-white powder
A guide to the accelerated elimination procedure
 A guide to the accelerated elimination procedure Prescribing information and information on adverse event reporting can be found on the last page. What is the accelerated elimination procedure? An accelerated
A guide to the accelerated elimination procedure Prescribing information and information on adverse event reporting can be found on the last page. What is the accelerated elimination procedure? An accelerated
PACKAGE LEAFLET: INFORMATION FOR THE USER Omeprazol XXX 40 mg powder for solution for infusion omeprazole
 PACKAGE LEAFLET: INFORMATION FOR THE USER Omeprazol XXX 40 mg powder for solution for infusion omeprazole Read all of this leaflet carefully before you start using this medicine. - Keep this leaflet. You
PACKAGE LEAFLET: INFORMATION FOR THE USER Omeprazol XXX 40 mg powder for solution for infusion omeprazole Read all of this leaflet carefully before you start using this medicine. - Keep this leaflet. You
VOLTAREN OPHTHA EYE DROPS
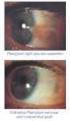 2013 במאי משרד הבריאות ותוכנו נבדק ואושר על ידו ע"י עלון זה נקבע ע פורמט PRESCRIBING INFORMATION VOLTAREN OPHTHA EYE DROPS 1 Name of the medicinal product Voltaren Ophtha Eye Drops. 2 Qualitative and quantitative
2013 במאי משרד הבריאות ותוכנו נבדק ואושר על ידו ע"י עלון זה נקבע ע פורמט PRESCRIBING INFORMATION VOLTAREN OPHTHA EYE DROPS 1 Name of the medicinal product Voltaren Ophtha Eye Drops. 2 Qualitative and quantitative
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Methadone 10mg/ml Injection / Physeptone 10mg/ml Injection 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Contains: Methadone Hydrochloride
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Methadone 10mg/ml Injection / Physeptone 10mg/ml Injection 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Contains: Methadone Hydrochloride
Paxil/Paxil-CR (paroxetine)
 Generic name: Paroxetine Available strengths: 10 mg, 20 mg, 30 mg, 40 mg tablets; 10 mg/5 ml oral suspension; 12.5 mg, 25 mg, 37.5 mg controlled-release tablets (Paxil-CR) Available in generic: Yes, except
Generic name: Paroxetine Available strengths: 10 mg, 20 mg, 30 mg, 40 mg tablets; 10 mg/5 ml oral suspension; 12.5 mg, 25 mg, 37.5 mg controlled-release tablets (Paxil-CR) Available in generic: Yes, except
Summary of the risk management plan (RMP) for Ofev (nintedanib)
 EMA/738120/2014 Summary of the risk management plan (RMP) for Ofev (nintedanib) This is a summary of the risk management plan (RMP) for Ofev, which details the measures to be taken in order to ensure that
EMA/738120/2014 Summary of the risk management plan (RMP) for Ofev (nintedanib) This is a summary of the risk management plan (RMP) for Ofev, which details the measures to be taken in order to ensure that
Kalms Tablets THR 01074/0235 UKPAR
 Kalms Tablets THR 01074/0235 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 10 Summary of Product Characteristics Page 11 Patient Information Leaflet
Kalms Tablets THR 01074/0235 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 10 Summary of Product Characteristics Page 11 Patient Information Leaflet
