Populations Interventions Comparators Outcomes Interventions of interest are: Mechanochemical ablation
|
|
|
- Ashlynn Brown
- 7 years ago
- Views:
Transcription
1 Protocol Treatment of Varicose Veins/Venous Insufficiency (701124) Medical Benefit Effective Date: 07/01/16 Next Review Date: 05/17 Preauthorization No Review Dates: 11/07, 11/08, 09/09, 09/10, 07/11, 07/12, 05/13, 05/14, 05/15, 05/16 Preauthorization is not required. The following Protocol contains medical necessity criteria that apply for this service. The criteria are also applicable to services provided in the local Medicare Advantage operating area for those members, unless separate Medicare Advantage criteria are indicated. If the criteria are not met, reimbursement will be denied and the patient cannot be billed. Please note that payment for covered services is subject to eligibility and the limitations noted in the patient s contract at the time the services are rendered. Populations Interventions Comparators Outcomes Interventions of interest are: Mechanochemical ablation Individuals: With varicose veins/ venous insufficiency Individuals: With varicose veins/ venous insufficiency Individuals: With varicose veins/ venous insufficiency Interventions of interest are: Cyanoacrylate adhesive Interventions of interest are: Cryoablation Comparators of interest are: Conservative therapy Ligation and stripping Endovenous radiofrequency or laser ablation Microfoam sclerotherapy Comparators of interest are: Conservative therapy Ligation and stripping Endovenous radiofrequency or laser ablation Microfoam sclerotherapy Comparators of interest are: Conservative therapy Ligation and stripping Endovenous radiofrequency or laser ablation Microfoam sclerotherapy Relevant outcomes include: Symptoms Morbid events Functional outcomes Change in disease status Relevant outcomes include: Symptoms Morbid events Functional outcomes Change in disease status Relevant outcomes include: Symptoms Morbid events Functional outcomes Change in disease status Description A variety of treatment modalities are available to treat varicose veins/venous insufficiency, including surgical approaches, thermal ablation, and sclerotherapy. The application of each of these treatment options is influenced by the severity of the symptoms, type of vein, source of venous reflux, and the use of other (prior or concurrent) treatments. Summary of Evidence The evidence on mechanochemical ablation, cyanoacrylate adhesive, and cryoablation in patients with varicose veins/venous insufficiency includes randomized controlled trials (RCTs) and multicenter series. Relevant outcomes are symptoms, morbid events, functional outcomes, and change in disease status. Several series have Page 1 of 13
2 been reported on mechanochemical ablation (MCA), and a large RCT comparing MCA with radiofrequency (RF) ablation is ongoing. Efficacy of cyanoacrylate adhesion at three months has been shown to be noninferior to RF in a multicenter RCT. Longer term follow-up is needed to determine durability of this treatment. Results from a recent RCT of cryoablation indicate that this therapy is inferior to conventional stripping. The evidence is insufficient to determine the effects of the technology on health outcomes. Policy Great or Small Saphenous Veins Treatment of the great or small saphenous veins by surgery (ligation and stripping), endovenous radiofrequency or laser ablation, or microfoam sclerotherapy may be considered medically necessary for symptomatic varicose veins/venous insufficiency when the following criteria have been met: There is demonstrated saphenous reflux and CEAP [Clinical, Etiology, Anatomy, Pathophysiology] class C2 or greater; AND there is documentation of one or more of the following indications: Ulceration secondary to venous stasis; OR Recurrent superficial thrombophlebitis; OR Hemorrhage or recurrent bleeding episodes from a ruptured superficial varicosity; OR Persistent pain, swelling, itching, burning, or other symptoms are associated with saphenous reflux, AND the symptoms significantly interfere with activities of daily living, AND conservative management including compression therapy for at least three months has not improved the symptoms. Treatment of great or small saphenous veins by surgery or endovenous radiofrequency or laser ablation, or microfoam sclerotherapy that do not meet the criteria described above is considered not medically necessary. Accessory Saphenous Veins Treatment of accessory saphenous veins by surgery (ligation and stripping), endovenous radiofrequency or laser ablation, or microfoam sclerotherapy may be considered medically necessary for symptomatic varicose veins/venous insufficiency when the following criteria have been met: Incompetence of the accessory saphenous vein is isolated, OR the great or small saphenous veins had been previously eliminated (at least three months); AND there is demonstrated accessory saphenous reflux; AND there is documentation of one or more of the following indications: Ulceration secondary to venous stasis; OR Recurrent superficial thrombophlebitis; OR Hemorrhage or recurrent bleeding episodes from a ruptured superficial varicosity; OR Persistent pain, swelling, itching, burning, or other symptoms are associated with saphenous reflux, AND the symptoms significantly interfere with activities of daily living, AND conservative management including compression therapy for at least three months has not improved the symptoms. Treatment of accessory saphenous veins by surgery or endovenous radiofrequency or laser ablation, or microfoam sclerotherapy that do not meet the criteria described above is considered not medically necessary. Page 2 of 13
3 Symptomatic Varicose Tributaries The following treatments are considered medically necessary as a component of the treatment of symptomatic varicose tributaries when performed either at the same time or following prior treatment (surgical, radiofrequency or laser) of the saphenous veins (none of these techniques has been shown to be superior to another): Stab avulsion Hook phlebectomy Sclerotherapy Transilluminated powered phlebectomy. Treatment of symptomatic varicose tributaries when performed either at the same time or following prior treatment of saphenous veins using any other techniques than noted above is considered investigational. Perforator Veins Surgical ligation (including subfascial endoscopic perforator surgery) or endovenous radiofrequency or laser ablation of incompetent perforator veins may be considered medically necessary as a treatment of leg ulcers associated with chronic venous insufficiency when the following conditions have been met: There is demonstrated perforator reflux; AND The superficial saphenous veins (great, small, or accessory saphenous and symptomatic varicose tributaries) have been previously eliminated; AND Ulcers have not resolved following combined superficial vein treatment and compression therapy for at least three months; AND The venous insufficiency is not secondary to deep venous thromboembolism. Ligation or ablation of incompetent perforator veins performed concurrently with superficial venous surgery is not medically necessary. Telangiectasia Treatment of telangiectasia such as spider veins, angiomata, and hemangiomata is considered not medically necessary. Other Techniques for conditions not specifically listed above are investigational, including, but not limited to: Sclerotherapy techniques, other than microfoam sclerotherapy, of great, small, or accessory saphenous veins Sclerotherapy of perforator veins Sclerotherapy of isolated tributary veins without prior or concurrent treatment of saphenous veins Stab avulsion, hook phlebectomy, or transilluminated powered phlebectomy of perforator, great or small saphenous, or accessory saphenous veins Endovenous radiofrequency or laser ablation of tributary veins Endovenous cryoablation of any vein Mechanochemical ablation of any vein Cyanoacrylate adhesive of any vein. Page 3 of 13
4 Policy Guidelines The standard classification of venous disease is the CEAP (Clinical, Etiologic, Anatomic, Pathophysiologic) classification system. The following is the Clinical portion of the CEAP. C0 C1 C2 C3 C4a C4b C5 C6 S A Clinical Classification No visible or palpable signs of venous disease Telangiectasies or reticular veins Varicose veins Edema Pigmentation and eczema Lipodermatosclerosis and atrophie blanche Healed venous ulcer Active venous ulcer Symptoms including ache, pain, tightness, skin irritation, heaviness, muscle cramps, as well as other complaints attributable to venous dysfunction Asymptomatic The Etiologic, Anatomic, And Pathophysiologic portions of the classifications are online ( It should be noted that the bulk of the literature discussing the role of ultrasound guidance refers to sclerotherapy of the saphenous vein, as opposed to the varicose tributaries. When ultrasound guidance is used to guide sclerotherapy of the varicose tributaries, it would be considered either not medically necessary or incidental to the injection procedure. Medicare Advantage Medicare Advantage will consider interventional treatment of varicose veins (sclerotherapy, ligation with or without stripping, and endovenous radiofrequency or laser ablation) medically necessary if the member remains symptomatic after a documented six-week trial of conservative therapy. The components of the conservative therapy include, but are not limited to: weight reduction, a daily exercise plan, periodic leg elevation, and the use of graduated compression stockings. The member is considered symptomatic if any of the following signs and symptoms of significantly diseased vessels of the lower extremities are documented: stasis ulcer of the lower leg, as above, significant pain and significant edema that interferes with activities of daily living, bleeding associated with the diseased vessels of the lower extremities, recurrent episodes of superficial phlebitis, stasis dermatitis, or refractory dependent edema. In addition to the requirement for failure of a six-week trial of conservative treatment and the symptoms described above, endovenous ablation therapy is considered medically necessary for members with: Page 4 of 13
5 a maximum vein diameter of 20 mm for laser ablation, absence of thrombosis or vein tortuosity, which would impair catheter advancement, and absence of significant peripheral artery disease. Radiofrequency/laser ablation is medically necessary only for treatment of the lesser or greater saphenous veins to improve symptoms attributable to saphenofemoral or saphenopopliteal reflux, and only for FDA devices specifically approved for these procedures. Non-cosmetic sclerotherapy will also be considered medically necessary if performed in conjunction with surgical ligation or stripping procedures in appropriately selected patients and to medium sized vessels (less than 4 mm in diameter). The following interventional treatments are considered to be cosmetic: Interventional treatment of asymptomatic varicosities, Treatment of telangiectases, Sclerotherapy for cosmetic purposes. The following interventional treatments are considered not medically necessary: Interventional treatment of symptomatic varicosities without documentation of a failed six week trial of conservative therapy, Sclerotherapy for vessels larger than 4 mm in diameter, Reinjection following recanalization or failure of vein closure without recurrent signs or symptoms, Sclerotherapy of the saphenous vein at its junction with the deep system, Noncompressive sclerotherapy, Compressive sclerotherapy for large, extensive or truncal varicosities, Any interventional treatment that uses equipment or sclerosants not approved for such purposes by the FDA, Laser ablation of veins with a diameter greater than 20 mm, Endovenous ablation therapy in the presence of thrombosis or venous tortuosity which would impair catheter advancement. Sclerotherapy, ligation and/or stripping of varicose veins, or endovenous ablation therapy are not appropriate for patients with the inability to tolerate compressive bandages or stockings; severe distal arterial occlusive disease; obliteration of deep venous system; an allergy to the sclerosant; or a hypercoaguable state. Foam sclerotherapy of the saphenous vein at its junction with the deep venous system as an alternative to ligation or saphenectomy is investigational. Refer also to the Cosmetic vs. Reconstructive Services Protocol. Background The venous system of the lower extremities consists of the superficial veins (this includes the great and small saphenous and accessory, or duplicate, veins that travel in parallel with the great and small saphenous veins), the deep system (popliteal and femoral veins), and perforator veins that cross through the fascia and connect the deep and superficial systems. One-way valves are present within all veins to direct the return of blood up Page 5 of 13
6 the lower limb. Because venous pressure in the deep system is generally greater than that of the superficial system, valve incompetence at any level may lead to backflow (venous reflux) with pooling of blood in superficial veins. Varicose veins with visible varicosities may be the only sign of venous reflux, although itching, heaviness, tension, and pain may also occur. Chronic venous insufficiency secondary to venous reflux can lead to thrombophlebitis, leg ulcerations, and hemorrhage. The CEAP classification considers the clinical, etiologic, anatomic, and pathologic (CEAP) characteristics of venous insufficiency, ranging from class 0 (no visible sign of disease) to class 6 (active ulceration). Treatment of venous reflux/venous insufficiency is aimed at reducing abnormal pressure transmission from the deep to the superficial veins. Conservative medical treatment consists of elevation of the extremities, graded compression, and wound care when indicated. Conventional surgical treatment consists of identifying and correcting the site of reflux by ligation of the incompetent junction followed by stripping of the vein to redirect venous flow through veins with intact valves. While most venous reflux is secondary to incompetent valves at the saphenofemoral or saphenopopliteal junctions, reflux may also occur at incompetent valves in the perforator veins or in the deep venous system. The competence of any single valve is not static and may be pressuredependent. For example, accessory saphenous veins may have independent saphenofemoral or saphenopopliteal junctions that become incompetent when the great or small saphenous veins are eliminated and blood flow is diverted through the accessory veins. Saphenous Veins and Tributaries Saphenous veins include the great and small saphenous and accessory saphenous veins that travel in parallel with the great or small saphenous veins. Tributaries are veins that empty into a larger vein. Treatment of venous reflux typically includes the following: 1. Identification by preoperative Doppler ultrasonography of the valvular incompetence 2. Control of the most proximal point of reflux, traditionally by suture ligation of the incompetent saphenofemoral or saphenopopliteal junction 3. Removal of the superficial vein from circulation, for example by stripping of the great and/or small saphenous veins 4. Removal of varicose tributaries (at the time of the initial treatment or subsequently) by stab avulsion (phlebectomy) or injection sclerotherapy. Minimally invasive alternatives to ligation and stripping have been investigated. These include sclerotherapy, TIPP, and thermal ablation using cryotherapy, high-frequency radio waves ( khz), or laser energy. Sclerotherapy The objective of sclerotherapy is to destroy the endothelium of the target vessel by injecting an irritant solution (either a detergent, osmotic solution, or chemical irritant), ultimately resulting in the occlusion of the vessel. The success of the treatment depends on accurate injection of the vessel, an adequate injectate volume and concentration of sclerosant, and compression. Historically, larger veins and very tortuous veins were not considered to be good candidates for sclerotherapy due to technical limitations. Technical improvements in sclerotherapy have included the routine use of Duplex ultrasound to target refluxing vessels, luminal compression of the vein with anesthetics, and a foam/sclerosant injectate in place of liquid sclerosant. Foam sclerosants are commonly produced by forcibly mixing a gas (e.g., air or carbon dioxide) with a liquid sclerosant (e.g., polidocanol or sodium tetradecyl sulfate). The foam is produced at the time of treatment. Varithena (previously known as Varisolve; BTG Plc, London) is a proprietary microfoam sclerosant that is dispersed from a canister with a controlled density and more consistent bubble size. Page 6 of 13
7 Endovenous Mechanochemical Ablation Endovenous mechanochemical ablation (MOCA ) uses both sclerotherapy and mechanical damage to the lumen. Following ultrasound imaging, a disposable catheter with a motor drive is inserted into the distal end of the target vein and advanced to the saphenofemoral junction. As the catheter is pulled back, a wire rotates at 3500 rpm within the lumen of the vein, abrading the lumen. At the same time, a liquid sclerosant (sodium tetradecyl sulfate) is infused near the rotating wire. It is proposed that mechanical ablation allows for better efficacy of the sclerosant, and results in less pain and risk of nerve injury without need for the tumescent anesthesia used with thermal endovenous ablation techniques (radiofrequency ablation [RFA] and endovenous laser treatment [EVLT]). Thermal Ablation RFA is performed by means of a specially designed catheter inserted through a small incision in the distal medial thigh to within one to two cm of the saphenofemoral junction. The catheter is slowly withdrawn, closing the vein. Laser ablation is performed similarly; a laser fiber is introduced into the great saphenous vein under ultrasound guidance; the laser is activated and slowly removed along the course of the saphenous vein. Cryoablation uses extreme cold to cause injury to the vessel. The objective of endovenous techniques is to cause injury to the vessel, causing retraction and subsequent fibrotic occlusion of the vein. Technical developments since thermal ablation procedures were initially introduced include the use of perivenous tumescent anesthesia, which allows successful treatment of veins larger than 12 mm in diameter and helps to protect adjacent tissue from thermal damage during treatment of the small saphenous vein. Cyanoacrylate Adhesive Cyanoacrylate adhesive is a clear, free-flowing liquid that polymerizes in the vessel via an anionic mechanism (i.e., polymerizes into a solid material on contact with body fluids or tissue). The adhesive is gradually injected along the length of the vein in conjunction with ultrasound and manual compression. The acute coaptation halts blood flow through the vein until the implanted adhesive becomes fibrotically encapsulated and establishes chronic occlusion of the treated vein. Cyanoacrylate glue has been used as a surgical adhesive and sealant for a variety of indications, including gastrointestinal bleeding, embolization of brain arteriovenous malformations, and to seal surgical incisions or other skin wounds. Transilluminated Powered Phlebectomy TIPP is an alternative to stab avulsion or hook phlebectomy. This procedure uses two instruments: an illuminator, which also provides irrigation, and a resector, which has an oscillating tip and can perform suction. Following removal of the saphenous vein, the illuminator is introduced via a small incision in the skin and tumescence solution (anesthetic and epinephrine) is infiltrated along the course of the varicosity. The resector is then inserted under the skin from the opposite direction, and the oscillating tip is placed directly beneath the illuminated veins to fragment and loosen the veins from the supporting tissue. Irrigation from the illuminator is used to clear the vein fragments and blood through aspiration and additional drainage holes. The illuminator and resector tips may then be repositioned, thereby reducing the number of incisions needed when compared with stab avulsion or hook phlebectomy. It has been proposed that TIPP might result in decreased operative time, decreased complications such as bruising, and faster recovery compared with the established procedures. Treatment of Perforator Veins Perforator veins cross through the fascia and connect the deep and superficial venous systems. Incompetent perforating veins were originally addressed with an open surgical procedure, called the Linton procedure, which involved a long medial calf incision to expose all posterior, medial, and paramedial perforators. While this procedure was associated with healing of ulcers, it was largely abandoned due to a high incidence of wound complications. The Linton procedure was subsequently modified by using a series of perpendicular skin flaps instead of a longitudinal skin flap to provide access to incompetent perforator veins in the lower part of the leg. The Page 7 of 13
8 modified Linton procedure may occasionally be used for the closure of incompetent perforator veins that cannot be reached by less invasive procedures. Subfascial endoscopic perforator surgery is a less invasive surgical procedure for treatment of incompetent perforators and has been reported since the mid-1980s. Guided by Duplex ultrasound scanning, small incisions are made in the skin, and the perforating veins are clipped or divided by endoscopic scissors. The operation can be performed as an outpatient procedure. Endovenous ablation of incompetent perforator veins with sclerotherapy and RFA has also been reported. Other Deep vein valve replacement is being investigated. Outcomes of interest for venous interventions include healing and recurrence, recannulation of the vein, and neovascularization. Recannulation (recanalization) is the restoration of the lumen of a vein after it has been occluded; this occurs more frequently following treatment with endovenous techniques. Neovascularization is the proliferation of new blood vessels in tissue and occurs more frequently following vein stripping. Direct comparisons of durability for endovenous and surgical procedures are complicated by these different mechanisms of recurrence. Relevant safety outcomes include the incidence of paresthesia, thermal skin injury, thrombus formation, thrombophlebitis, wound infection, and transient neurologic effects. Regulatory Status In 2015, the VenaSeal Closure System (Sapheon, a part of Medtronic) was approved by the U.S. Food and Drug Administration (FDA) through the premarket approval process for the permanent closure of clinically significant venous reflux through endovascular embolization with coaptation. The VenaSeal Closure System seals the vein using a cyanoacrylate adhesive agent. FDA product code: PJQ. In 2013, Varithena (formerly known as Varisolve ; BTG Plc, London), a sclerosant microfoam made with a proprietary gas mix, was approved by FDA under a new drug application for the treatment of incompetent great saphenous veins, accessory saphenous veins and visible varicosities of the great saphenous vein system above and below the knee. The following devices were cleared for marketing by FDA through the 501(k) process for the endovenous treatment of superficial vein reflux: In 1999, the VNUS Closure System (a radiofrequency device) received FDA clearance through the 510(k) process for endovascular coagulation of blood vessels in patients with superficial vein reflux. The VNUS RFS and RFSFlex devices received FDA clearance in 2005 for use in vessel and tissue coagulation including: treatment of incompetent (i.e., refluxing) perforator and tributary veins. The modified VNUS ClosureFast Intravascular Catheter received FDA clearance through the 510(k) process in FDA product code: GEI. In 2002, the Diomed 810 nm surgical laser and EVLT (endovenous laser therapy) procedure kit received FDA clearance through the 510(k) process, for use in the endovascular coagulation of the great saphenous vein of the thigh in patients with superficial vein reflux. FDA product code: GEX. A modified Erbe Erbokryo cryosurgical unit (Erbe USA) received FDA clearance for marketing in A variety of clinical indications are listed, including cryostripping of varicose veins of the lower limbs. FDA product code: GEH. The Trivex system (InaVein LLC) is a device for transilluminated powered phlebectomy that received FDA clearance through the 510(k) process in October According to the label, the intended use is for ambulatory phlebectomy procedures for the resection and ablation of varicose veins. FDA product code: DNQ. Page 8 of 13
9 The ClariVein Infusion Catheter (Vascular Insights) received marketing clearance through the 510(k) process in 2008 (K071468). It is used for mechanochemical ablation. Predicate devices were listed as the Trellis Infusion System (K013635) and the Slip-Cath Infusion Catheter (K882796). The system includes an infusion catheter, motor drive, stopcock and syringe and is intended for the infusion of physician-specified agents in the peripheral vasculature. FDA product code: KRA. Services that are the subject of a clinical trial do not meet our Technology Assessment Protocol criteria and are considered investigational. For explanation of experimental and investigational, please refer to the Technology Assessment Protocol. It is expected that only appropriate and medically necessary services will be rendered. We reserve the right to conduct prepayment and postpayment reviews to assess the medical appropriateness of the above-referenced procedures. Some of this Protocol may not pertain to the patients you provide care to, as it may relate to products that are not available in your geographic area. References We are not responsible for the continuing viability of web site addresses that may be listed in any references below. 1. O Meara S, Cullum NA, Nelson EA. Compression for venous leg ulcers. Cochrane Database Syst Rev. 2009(1):CD PMID O Meara S, Cullum N, Nelson EA, et al. Compression for venous leg ulcers. Cochrane Database Syst Rev. 2012; 11:CD PMID Shingler S, Robertson L, Boghossian S, et al. Compression stockings for the initial treatment of varicose veins in patients without venous ulceration. Cochrane Database Syst Rev. 2011; 11:CD PMID Howard DP, Howard A, Kothari A, et al. The role of superficial venous surgery in the management of venous ulcers: a systematic review. Eur J Vasc Endovasc Surg. Oct 2008; 36(4): PMID O Donnell TF, Jr. The present status of surgery of the superficial venous system in the management of venous ulcer and the evidence for the role of perforator interruption. J Vasc Surg. Oct 2008; 48(4): PMID Jones L, Braithwaite BD, Selwyn D, et al. Neovascularisation is the principal cause of varicose vein recurrence: results of a randomised trial of stripping the long saphenous vein. Eur J Vasc Endovasc Surg. Nov 1996; 12(4): PMID Rutgers PH, Kitslaar PJ. Randomized trial of stripping versus high ligation combined with sclerotherapy in the treatment of the incompetent greater saphenous vein. Am J Surg. Oct 1994; 168(4): PMID Nesbitt C, Bedenis R, Bhattacharya V, et al. Endovenous ablation (radiofrequency and laser) and foam sclerotherapy versus open surgery for great saphenous vein varices. Cochrane Database Syst Rev. 2014; 7:CD PMID Brittenden J, Cotton SC, Elders A, et al. A randomized trial comparing treatments for varicose veins. N Engl J Med. Sep ; 371(13): PMID Luebke T, Gawenda M, Heckenkamp J, et al. Meta-analysis of endovenous radiofrequency obliteration of the great saphenous vein in primary varicosis. J Endovasc Ther. Apr 2008; 15(2): PMID Page 9 of 13
10 11. Merchant RF, Pichot O. Long-term outcomes of endovenous radiofrequency obliteration of saphenous reflux as a treatment for superficial venous insufficiency. J Vasc Surg. Sep 2005; 42(3): ; discussion 509. PMID Rass K, Frings N, Glowacki P, et al. Comparable effectiveness of endovenous laser ablation and high ligation with stripping of the great saphenous vein: two-year results of a randomized clinical trial (RELACS study). Arch Dermatol. Jan 2012; 148(1): PMID Christenson JT, Gueddi S, Gemayel G, et al. Prospective randomized trial comparing endovenous laser ablation and surgery for treatment of primary great saphenous varicose veins with a 2-year follow-up. J Vasc Surg. Nov 2010; 52(5): PMID Biemans AA, Kockaert M, Akkersdijk GP, et al. Comparing endovenous laser ablation, foam sclerotherapy, and conventional surgery for great saphenous varicose veins. J Vasc Surg. Sep 2013; 58(3): e721. PMID van der Velden SK, Biemans AA, De Maeseneer MG, et al. Five-year results of a randomized clinical trial of conventional surgery, endovenous laser ablation and ultrasound-guided foam sclerotherapy in patients with great saphenous varicose veins. Br J Surg. Sep 2015; 102(10): PMID Theivacumar NS, Darwood RJ, Gough MJ. Endovenous laser ablation (EVLA) of the anterior accessory great saphenous vein (AAGSV): abolition of sapheno-femoral reflux with preservation of the great saphenous vein. Eur J Vasc Endovasc Surg. Apr 2009; 37(4): PMID Shadid N, Ceulen R, Nelemans P, et al. Randomized clinical trial of ultrasound-guided foam sclerotherapy versus surgery for the incompetent great saphenous vein. Br J Surg. Aug 2012; 99(8): PMID U.S. Food and Drug Administration Center for Drug Evaluation and Research Varithena Summary Review. 2013; Accessed April, Todd KL, 3rd, Wright D, for the V-IG. The VANISH-2 study: a randomized, blinded, multicenter study to evaluate the efficacy and safety of polidocanol endovenous microfoam 0.5% and 1.0% compared with placebo for the treatment of saphenofemoral junction incompetence. Phlebology. Oct 2014; 29(9): PMID Bishawi M, Bernstein R, Boter M, et al. Mechanochemical ablation in patients with chronic venous disease: A prospective multicenter report. Phlebology. Jul PMID Elias S, Raines JK. Mechanochemical tumescentless endovenous ablation: final results of the initial clinical trial. Phlebology. Mar 2012; 27(2): PMID Boersma D, van Eekeren RR, Werson DA, et al. Mechanochemical endovenous ablation of small saphenous vein insufficiency using the ClariVein ((R)) device: one-year results of a prospective series. Eur J Vasc Endovasc Surg. Mar 2013; 45(3): PMID Mueller RL, Raines JK. ClariVein mechanochemical ablation: background and procedural details. Vasc Endovascular Surg. Apr 2013; 47(3): PMID Bootun R, Lane T, Dharmarajah B, et al. Intra-procedural pain score in a randomised controlled trial comparing mechanochemical ablation to radiofrequency ablation: The Multicentre Venefit versus ClariVein(R) for varicose veins trial. Phlebology. Sep PMID Page 10 of 13
11 25. U.S. Food and Drug Administration. VenaSeal Closure System - P ; Accessed May 18, Morrison N, Gibson K, McEnroe S, et al. Randomized trial comparing cyanoacrylate embolization and radiofrequency ablation for incompetent great saphenous veins (VeClose). J Vasc Surg. Apr 2015; 61(4): PMID Almeida JI, Javier JJ, Mackay EG, et al. Two-year follow-up of first human use of cyanoacrylate adhesive for treatment of saphenous vein incompetence. Phlebology. Jul 2015; 30(6): PMID Zierau U. Sealing Veins with the VenaSeal Sapheon Closure System: Results for 795 Treated Truncal Veins after 1000 Days. Vasomed. 2015; 27: PMID 29. Klem TM, Schnater JM, Schutte PR, et al. A randomized trial of cryo stripping versus conventional stripping of the great saphenous vein. J Vasc Surg. Feb 2009; 49(2): PMID Disselhoff BC, der Kinderen DJ, Kelder JC, et al. Randomized clinical trial comparing endovenous laser with cryostripping for great saphenous varicose veins. Br J Surg. Oct 2008; 95(10): PMID Disselhoff BC, der Kinderen DJ, Kelder JC, et al. Five-year results of a randomized clinical trial comparing endovenous laser ablation with cryostripping for great saphenous varicose veins. Br J Surg. Aug 2011; 98(8): PMID Milleret R, Huot L, Nicolini P, et al. Great saphenous vein ablation with steam injection: results of a multicenter study. Eur J Vasc Endovasc Surg. Apr 2013; 45(4): PMID Yang L, Wang XP, Su WJ, et al. Randomized clinical trial of endovenous microwave ablation combined with high ligation versus conventional surgery for varicose veins. Eur J Vasc Endovasc Surg. Oct 2013; 46(4): PMID van den Bos RR, Malskat WS, De Maeseneer MG, et al. Randomized clinical trial of endovenous laser ablation versus steam ablation (LAST trial) for great saphenous varicose veins. Br J Surg. Aug 2014; 101(9): PMID Tisi PV, Beverley C, Rees A. Injection sclerotherapy for varicose veins. Cochrane Database Syst Rev. 2006(4):CD PMID Leopardi D, Hoggan BL, Fitridge RA, et al. Systematic review of treatments for varicose veins. Ann Vasc Surg. Mar 2009; 23(2): PMID El-Sheikha J, Nandhra S, Carradice D, et al. Clinical outcomes and quality of life 5 years after a randomized trial of concomitant or sequential phlebectomy following endovenous laser ablation for varicose veins. Br J Surg. Aug 2014; 101(9): PMID Michaels JA, Campbell WB, Brazier JE, et al. Randomised clinical trial, observational study and assessment of cost-effectiveness of the treatment of varicose veins (REACTIV trial). Health Technol Assess. Apr 2006; 10(13):1-196, iii-iv. PMID Yamaki T, Hamahata A, Soejima K, et al. Prospective Randomised Comparative Study of Visual Foam Sclerotherapy Alone or in Combination with Ultrasound-guided Foam Sclerotherapy for Treatment of Superficial Venous Insufficiency: Preliminary Report. Eur J Vasc Endovasc Surg. Jan PMID Luebke T, Brunkwall J. Meta-analysis of transilluminated powered phlebectomy for superficial varicosities. J Cardiovasc Surg (Torino). Dec 2008; 49(6): PMID Page 11 of 13
12 41. Chetter IC, Mylankal KJ, Hughes H, et al. Randomized clinical trial comparing multiple stab incision phlebectomy and transilluminated powered phlebectomy for varicose veins. Br J Surg. Feb 2006; 93(2): PMID Barwell JR, Davies CE, Deacon J, et al. Comparison of surgery and compression with compression alone in chronic venous ulceration (ESCHAR study): randomised controlled trial. Lancet. Jun ; 363(9424): PMID Gohel MS, Barwell JR, Taylor M, et al. Long term results of compression therapy alone versus compression plus surgery in chronic venous ulceration (ESCHAR): randomised controlled trial. BMJ. Jul ; 335(7610):83. PMID Nelzen O, Fransson I. Early results from a randomized trial of saphenous surgery with or without subfascial endoscopic perforator surgery in patients with a venous ulcer. Br J Surg. Apr 2011; 98(4): PMID Blomgren L, Johansson G, Dahlberg-Akerman A, et al. Changes in superficial and perforating vein reflux after varicose vein surgery. J Vasc Surg. Aug 2005; 42(2): PMID Tenbrook JA, Jr., Iafrati MD, O Donnell T F, Jr., et al. Systematic review of outcomes after surgical management of venous disease incorporating subfascial endoscopic perforator surgery. J Vasc Surg. Mar 2004; 39(3): PMID van Gent WB, Catarinella FS, Lam YL, et al. Conservative versus surgical treatment of venous leg ulcers: 10- year follow up of a randomized, multicenter trial. Phlebology. Mar 2015; 30(1 Suppl): PMID Luebke T, Brunkwall J. Meta-analysis of subfascial endoscopic perforator vein surgery (SEPS) for chronic venous insufficiency. Phlebology. Feb 2009; 24(1):8-16. PMID Hirsch SA, Dillavou E. Options in the management of varicose veins, J Cardiovasc Surg (Torino). Feb 2008; 49(1): PMID Hissink RJ, Bruins RM, Erkens R, et al. Innovative treatments in chronic venous insufficiency: endovenous laser ablation of perforating veins: a prospective short-term analysis of 58 cases. Eur J Vasc Endovasc Surg. Sep 2010; 40(3): PMID Myers KA, Jolley D. Factors affecting the risk of deep venous occlusion after ultrasound-guided sclerotherapy for varicose veins. Eur J Vasc Endovasc Surg. Nov 2008; 36(5): PMID Gloviczki P, Comerota AJ, Dalsing MC, et al. The care of patients with varicose veins and associated chronic venous diseases: clinical practice guidelines of the Society for Vascular Surgery and the American Venous Forum. J Vasc Surg. May 2011; 53(5 Suppl):2S-48S. PMID Silberzweig JE, Funaki BS, Ray CE, Jr., et al. ACR appropriateness criteria treatment of lower-extremity venous insufficiency. 2012; Accessed April, Society of Interventional Radiology. Position Statement on Endovenous Ablation. 2003; Accessed April, Kundu S, Lurie F, Millward SF, et al. Recommended reporting standards for endovenous ablation for the treatment of venous insufficiency: joint statement of the American Venous Forum and the Society of Interventional Radiology. J Vasc Interv Radiol. Sep 2007; 18(9): PMID National Institute for Health and Care Excellence (NICE). Radiofrequency Ablation of Varicose Veins; Interventional Procedure Guidance IPG8 2003; Accessed April, Page 12 of 13
13 57. National Institute for Health and Care Excellence (NICE). Endovenous Laser Treatment of the Long Saphenous Vein. Interventional Procedure Guidance IPG ; Accessed April, National Institute for Health and Care Excellence (NICE). Ultrasound-guided foam sclerotherapy for varicose veins; IPG ; Accessed April, National Institute for Health and Care Excellence (NICE). IPG435 Endovenous mechanochemical ablation for varicose veins: guidance. 2013; Accessed April, National Institute for Health and Care Excellence (NICE). NICE Clinical Guideline 168: Varicose veins in the legs. 2013; Accessed April, Brittenden J, Cotton SC, Elders A, et al. Clinical effectiveness and cost-effectiveness of foam sclerotherapy, endovenous laser ablation and surgery for varicose veins: results from the Comparison of LAser, Surgery and foam Sclerotherapy (CLASS) randomised controlled trial. Health Technol Assess. Apr 2015; 19(27): PMID NGS Local Coverage Determination (LCD): VARICOSE VEINS of the Lower Extremity, Treatment of (L33575), Original Effective Date for services performed on or after 10/01/2015. Page 13 of 13
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: varicose_veins_treatment_for 12/2000 11/2014 11/2015 11/2014 Description of Procedure or Service A variety
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: varicose_veins_treatment_for 12/2000 11/2014 11/2015 11/2014 Description of Procedure or Service A variety
Effective Date: March 2, 2016
 Medical Review Criteria Varicose Vein Procedures Effective Date: March 2, 2016 Treatmetn Subject: Varicose Vein Procedures VeinTreatment of Varicose Policy: Veins HPHC covers specific non-experimental
Medical Review Criteria Varicose Vein Procedures Effective Date: March 2, 2016 Treatmetn Subject: Varicose Vein Procedures VeinTreatment of Varicose Policy: Veins HPHC covers specific non-experimental
POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS DISCLAIMER CODING INFORMATION REFERENCES POLICY HISTORY
 Original Issue Date (Created): 7/12/2003 Most Recent Review Date (Revised): 3/29/2016 Effective Date: 8/1/2016 I. POLICY Great or Small Saphenous Veins Treatment of the greater small saphenous veins by
Original Issue Date (Created): 7/12/2003 Most Recent Review Date (Revised): 3/29/2016 Effective Date: 8/1/2016 I. POLICY Great or Small Saphenous Veins Treatment of the greater small saphenous veins by
MEDICAL COVERAGE POLICY. SERVICE: Varicose Veins of the Lower Extremities. PRIOR AUTHORIZATION: Required.
 Page 1 of 5 MEDICAL COVERAGE POLICY Important note Even though this policy may indicate that a particular service or supply may be considered covered, this conclusion is not based upon the terms of your
Page 1 of 5 MEDICAL COVERAGE POLICY Important note Even though this policy may indicate that a particular service or supply may be considered covered, this conclusion is not based upon the terms of your
Treatment of Varicose Veins/Venous Insufficiency
 MEDICAL POLICY POLICY RELATED POLICIES POLICY GUIDELINES DESCRIPTION SCOPE BENEFIT APPLICATION RATIONALE REFERENCES CODING APPENDIX HISTORY Treatment of Varicose Veins/Venous Insufficiency Number 7.01.519
MEDICAL POLICY POLICY RELATED POLICIES POLICY GUIDELINES DESCRIPTION SCOPE BENEFIT APPLICATION RATIONALE REFERENCES CODING APPENDIX HISTORY Treatment of Varicose Veins/Venous Insufficiency Number 7.01.519
Medicare C/D Medical Coverage Policy
 Varicose Vein Treatment Origination Date: June 1, 1993 Review Date: July 20, 2016 Next Review: July, 2018 Medicare C/D Medical Coverage Policy DESCRIPTION OF PROCEDURE OR SERVICE Varicose veins of the
Varicose Vein Treatment Origination Date: June 1, 1993 Review Date: July 20, 2016 Next Review: July, 2018 Medicare C/D Medical Coverage Policy DESCRIPTION OF PROCEDURE OR SERVICE Varicose veins of the
Medicare C/D Medical Coverage Policy
 Varicose Vein Treatment Medicare C/D Medical Coverage Policy Origination Date: June 1, 1993 Review Date: September 16, 2015 Next Review: September, 2017 DESCRIPTION OF PROCEDURE OR SERVICE Varicose veins
Varicose Vein Treatment Medicare C/D Medical Coverage Policy Origination Date: June 1, 1993 Review Date: September 16, 2015 Next Review: September, 2017 DESCRIPTION OF PROCEDURE OR SERVICE Varicose veins
VARICOSE VEINS AND VENOUS INSUFFICIENCY
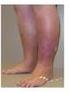 VARICOSE VEINS AND VENOUS INSUFFICIENCY SURGICAL AND ABLATION TREATMENTS Policy Number: 2014M0064A Effective Date: September 1, 2014 Table of Contents: Page: Cross Reference Policy: POLICY DESCRIPTION
VARICOSE VEINS AND VENOUS INSUFFICIENCY SURGICAL AND ABLATION TREATMENTS Policy Number: 2014M0064A Effective Date: September 1, 2014 Table of Contents: Page: Cross Reference Policy: POLICY DESCRIPTION
Clinical Review Criteria
 Clinical Review Criteria Treatment of Varicose Veins Radiofrequency Catheter Closure Sclerotherapy Surgical Stripping Trivex System for Outpatient Varicose Vein Surgery VenaSeal Closure System VNUS Closure
Clinical Review Criteria Treatment of Varicose Veins Radiofrequency Catheter Closure Sclerotherapy Surgical Stripping Trivex System for Outpatient Varicose Vein Surgery VenaSeal Closure System VNUS Closure
Corporate Medical Policy Treatment of Varicose Veins/Venous Insufficiency. Medical Policy
 Corporate Medical Policy Treatment of Varicose Veins/Venous Insufficiency File name: Treatment of Varicose Veins/ Venous Insufficiency File code: UM.SURG.03 Origination: 9/01/2010 Last Review: 07/15/2010,
Corporate Medical Policy Treatment of Varicose Veins/Venous Insufficiency File name: Treatment of Varicose Veins/ Venous Insufficiency File code: UM.SURG.03 Origination: 9/01/2010 Last Review: 07/15/2010,
Yes when meets criteria below
 Vein Disease Treatment MP9241 Covered Service: Prior Authorization Required: Additional Information: Medicare Policy: BadgerCare Plus Policy: Yes when meets criteria below Yes None Dean Health Plan covers
Vein Disease Treatment MP9241 Covered Service: Prior Authorization Required: Additional Information: Medicare Policy: BadgerCare Plus Policy: Yes when meets criteria below Yes None Dean Health Plan covers
Clinical Medical Policy Varicose Vein Treatment
 Benefit Coverage Covered Benefit for lines of business including: Health Benefits Exchange (HBE), Rite Care (MED), Children with Special Needs (CSN), Substitute Care (SUB), Rhody Health Partners (RHP),
Benefit Coverage Covered Benefit for lines of business including: Health Benefits Exchange (HBE), Rite Care (MED), Children with Special Needs (CSN), Substitute Care (SUB), Rhody Health Partners (RHP),
MEDICAL POLICY SUBJECT: VARICOSITIES, TREATMENT ALTERNATIVES TO VEIN STRIPPING AND LIGATION
 MEDICAL POLICY SUBJECT: VARICOSITIES, TREATMENT PAGE: 1 OF: 10 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: VARICOSITIES, TREATMENT PAGE: 1 OF: 10 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
Medical Coverage Policy Treatment for Varicose Veins-PREAUTH
 Medical Coverage Policy Treatment for Varicose Veins-PREAUTH Device/Equipment Drug Medical Surgery Test Other Effective Date: 9/1/2001 Policy Last Updated: 12/20/2011 Prospective review is recommended/required.
Medical Coverage Policy Treatment for Varicose Veins-PREAUTH Device/Equipment Drug Medical Surgery Test Other Effective Date: 9/1/2001 Policy Last Updated: 12/20/2011 Prospective review is recommended/required.
Surgical Options for Venous Disease. Sandra C Carr MD Vascular Surgery Meriter Wisconsin Heart
 Surgical Options for Venous Disease Sandra C Carr MD Vascular Surgery Meriter Wisconsin Heart Chronic Venous Disease Approximately 23% of adults in the US have varicose veins Estimated 22 million women
Surgical Options for Venous Disease Sandra C Carr MD Vascular Surgery Meriter Wisconsin Heart Chronic Venous Disease Approximately 23% of adults in the US have varicose veins Estimated 22 million women
PROVIDER POLICIES & PROCEDURES
 PROVIDER POLICIES & PROCEDURES SCLEROTHERAPY TREATMENT OF SUPERFICIAL VARICOSE VEINS OF THE LEGS The purpose of this document is to provide guidance to providers enrolled in the Connecticut Medical Assistance
PROVIDER POLICIES & PROCEDURES SCLEROTHERAPY TREATMENT OF SUPERFICIAL VARICOSE VEINS OF THE LEGS The purpose of this document is to provide guidance to providers enrolled in the Connecticut Medical Assistance
MEDICAL COVERAGE POLICY SERVICE: Varicose Veins of the Lower Extremities. SERVICE: Treatment of Varicose Veins of the Lower Extremities
 Important note Even though this policy may indicate that a particular service or supply may be considered covered, this conclusion is not based upon the terms of your particular benefit plan. Each benefit
Important note Even though this policy may indicate that a particular service or supply may be considered covered, this conclusion is not based upon the terms of your particular benefit plan. Each benefit
Medical Policy Manual. Date of Origin: October 11, 1999. Topic: Varicose Vein Treatment. Last Reviewed Date: May 2015.
 Medical Policy Manual Topic: Varicose Vein Treatment Section: Surgery Policy No: 104 Date of Origin: October 11, 1999 Last Reviewed Date: May 2015 Effective Date: September 1, 2015 IMPORTANT REMINDER Medical
Medical Policy Manual Topic: Varicose Vein Treatment Section: Surgery Policy No: 104 Date of Origin: October 11, 1999 Last Reviewed Date: May 2015 Effective Date: September 1, 2015 IMPORTANT REMINDER Medical
RANDOMIZED CONTROL TRIALS (RCT s) on VARICOSE VEIN ENDOVENOUS TREATMENT
 RANDOMIZED CONTROL TRIALS (RCT s) on VARICOSE VEIN ENDOVENOUS TREATMENT M. PERRIN Lyon - France CONTROVERSIES AND UPDATES IN VASCULAR SURGERY JANUARY 19-21, 2012 Paris B. Eklof Helsingborg, Sweden Thirty
RANDOMIZED CONTROL TRIALS (RCT s) on VARICOSE VEIN ENDOVENOUS TREATMENT M. PERRIN Lyon - France CONTROVERSIES AND UPDATES IN VASCULAR SURGERY JANUARY 19-21, 2012 Paris B. Eklof Helsingborg, Sweden Thirty
Medical Coverage Policy Varicose Vein Treatment sad
 Medical Coverage Policy Varicose Vein Treatment sad EFFECTIVE DATE: 10 28 2001 POLICY LAST UPDATED: 05 07 2013 OVERVIEW Varicose veins are large superficial veins that have become swollen. These veins
Medical Coverage Policy Varicose Vein Treatment sad EFFECTIVE DATE: 10 28 2001 POLICY LAST UPDATED: 05 07 2013 OVERVIEW Varicose veins are large superficial veins that have become swollen. These veins
Non-surgical treatment of severe varicose veins
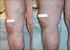 Non-surgical treatment of severe varicose veins Yasu Harasaki UCHSC Department of Surgery General Surgery Grand Rounds March 19, 2007 Definition Dilated, palpable, subcutaneous veins generally >3mm in
Non-surgical treatment of severe varicose veins Yasu Harasaki UCHSC Department of Surgery General Surgery Grand Rounds March 19, 2007 Definition Dilated, palpable, subcutaneous veins generally >3mm in
DEBATE ON Management of primary Varicose Veins SURGERY. Adel Husseiny, Kamhawy Vascular Surgery Unit Tanta University
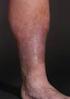 DEBATE ON Management of primary Varicose Veins SURGERY Adel Husseiny, Kamhawy Vascular Surgery Unit Tanta University Efficacy of treatment alternatives Durability of results Cosmetically acceptable results
DEBATE ON Management of primary Varicose Veins SURGERY Adel Husseiny, Kamhawy Vascular Surgery Unit Tanta University Efficacy of treatment alternatives Durability of results Cosmetically acceptable results
Medical Affairs Policy
 Service: Varicose Vein Treatments PUM 250-0032 Medical Affairs Policy Implemented 04/04/14, 04/01/15, 4/1/16 Reviewed 12/12/14 Revised 12/12/14, 12/11/15 Developed Arise/WPS Policy 12/12/14, 12/11/15 Committee
Service: Varicose Vein Treatments PUM 250-0032 Medical Affairs Policy Implemented 04/04/14, 04/01/15, 4/1/16 Reviewed 12/12/14 Revised 12/12/14, 12/11/15 Developed Arise/WPS Policy 12/12/14, 12/11/15 Committee
TREATMENT OF VARICOSE VEINS: CAN IT BE IMPROVED BY MECHANOCHEMICAL ABLATION USING THE CLARIVEIN DEVICE?
 TREATMENT OF VARICOSE VEINS: CAN IT BE IMPROVED BY MECHANOCHEMICAL ABLATION USING THE CLARIVEIN DEVICE? Michel MJP Reijnen Rijnstate Hospital, Arnhem The Netherlands mmpj.reijnen@gmail.com Disclosures
TREATMENT OF VARICOSE VEINS: CAN IT BE IMPROVED BY MECHANOCHEMICAL ABLATION USING THE CLARIVEIN DEVICE? Michel MJP Reijnen Rijnstate Hospital, Arnhem The Netherlands mmpj.reijnen@gmail.com Disclosures
Venous Reflux Disease and Current Treatments VN20-87-A 01/06
 Venous Reflux Disease and Current Treatments Leg Vein Anatomy Your legs are made up of a network of veins and vessels that carry blood back to the heart The venous system is comprised of: Deep veins Veins
Venous Reflux Disease and Current Treatments Leg Vein Anatomy Your legs are made up of a network of veins and vessels that carry blood back to the heart The venous system is comprised of: Deep veins Veins
Local Coverage Determination (LCD): Varicose Veins of the Lower Extremities (L31796)
 Local Coverage Determination (LCD): Varicose Veins of the Lower Extremities (L31796) Contractor Information Contractor Name Palmetto GBA LCD Information Document Information LCD ID L31796 LCD Title Varicose
Local Coverage Determination (LCD): Varicose Veins of the Lower Extremities (L31796) Contractor Information Contractor Name Palmetto GBA LCD Information Document Information LCD ID L31796 LCD Title Varicose
Venous leg ulcers. Venous Ulcers. Treat the source, not just the symptom
 Venous leg ulcers Venous Ulcers Treat the source, not just the symptom Topics What causes venous ulcers (VU) Treating the source, not just the symptom Diagnosis with venous duplex ultrasound scan Treatment
Venous leg ulcers Venous Ulcers Treat the source, not just the symptom Topics What causes venous ulcers (VU) Treating the source, not just the symptom Diagnosis with venous duplex ultrasound scan Treatment
Spotlight Series: Interventional Radiology. Varicose Veins and Venous Insufficiency
 Spotlight Series: Interventional Radiology Varicose Veins and Venous Insufficiency What is venous insufficiency? Spectrum of Disease Spider veins and telangiectasias Small reddish and purple veins near
Spotlight Series: Interventional Radiology Varicose Veins and Venous Insufficiency What is venous insufficiency? Spectrum of Disease Spider veins and telangiectasias Small reddish and purple veins near
SURGICAL AND ABLATIVE PROCEDURES FOR VENOUS INSUFFICIENCY AND VARICOSE VEINS
 SURGICAL AND ABLATIVE PROCEDURES FOR VENOUS INSUFFICIENCY AND VARICOSE VEINS Protocol: SUR037 Effective Date: 5/1/2015 Table of Contents COMMERCIAL INDICATIONS FOR COVERAGE DEFINITIONS MEDICARE AND MEDICAID
SURGICAL AND ABLATIVE PROCEDURES FOR VENOUS INSUFFICIENCY AND VARICOSE VEINS Protocol: SUR037 Effective Date: 5/1/2015 Table of Contents COMMERCIAL INDICATIONS FOR COVERAGE DEFINITIONS MEDICARE AND MEDICAID
Modern Management of Varicose Veins
 Modern Management of Varicose Veins GPCME 2008 David Ferrar Vascular and Endovascular Surgeon Vascular Ultrasound Specialist Multimodality treatment Compression stockings Sclerotherapy Ultrasound guided
Modern Management of Varicose Veins GPCME 2008 David Ferrar Vascular and Endovascular Surgeon Vascular Ultrasound Specialist Multimodality treatment Compression stockings Sclerotherapy Ultrasound guided
Recurrent Varicose Veins. Vineet Mishra, MD Director of Mohs Surgery and Procedural Dermatology University of Texas Health Science Center San Antonio
 Recurrent Varicose Veins Vineet Mishra, MD Director of Mohs Surgery and Procedural Dermatology University of Texas Health Science Center San Antonio Disclosures None Possible Causes of Recurrence DNA:
Recurrent Varicose Veins Vineet Mishra, MD Director of Mohs Surgery and Procedural Dermatology University of Texas Health Science Center San Antonio Disclosures None Possible Causes of Recurrence DNA:
Microfoam Ultrasound-Guided Sclerotherapy of Varicose Veins in 100 Legs
 Microfoam Ultrasound-Guided Sclerotherapy of Varicose Veins in 1 Legs JOHN M. BARRETT, FRNZCGP, n BRUCE ALLEN, FRACR, n ANNE OCKELFORD, FRNZCGP, n AND MITCHEL P. GOLDMAN, MD w n Palm Clinic, Auckland,
Microfoam Ultrasound-Guided Sclerotherapy of Varicose Veins in 1 Legs JOHN M. BARRETT, FRNZCGP, n BRUCE ALLEN, FRACR, n ANNE OCKELFORD, FRNZCGP, n AND MITCHEL P. GOLDMAN, MD w n Palm Clinic, Auckland,
Varicose Vein Therapy: An Introduction to Surgical and Endovascular Treatment
 Varicose Vein Therapy: An Introduction to Surgical and Endovascular Treatment Lower Extremity Venous Anatomy Deep, Superficial, Perforating, Reticular veins Located in two separate compartments (deep and
Varicose Vein Therapy: An Introduction to Surgical and Endovascular Treatment Lower Extremity Venous Anatomy Deep, Superficial, Perforating, Reticular veins Located in two separate compartments (deep and
LOWER EXTREMITY VENOUS DUPLEX ULTRASOUND:
 LOWER EXTREMITY VENOUS DUPLEX ULTRASOUND: Chronic Venous Insufficiency Phillip J Bendick, PhD William Beaumont Hospital Royal Oak, Michigan Prevalence: Carotid ASO ~ 3M Peripheral Arterial Dz ~ 5M + CAD
LOWER EXTREMITY VENOUS DUPLEX ULTRASOUND: Chronic Venous Insufficiency Phillip J Bendick, PhD William Beaumont Hospital Royal Oak, Michigan Prevalence: Carotid ASO ~ 3M Peripheral Arterial Dz ~ 5M + CAD
Modern Varicose Vein Treatments: What Every Patient Should Know
 The Skin and Vein Center Oneonta Laser Derm & Day Spa Natural Good Looks and Leg Veins Our Specialty Dr Eric Dohner, MD 41-45 Dietz St Oneonta, NY 13820 607/431-2525 www.oneontalaserderm.com Modern Varicose
The Skin and Vein Center Oneonta Laser Derm & Day Spa Natural Good Looks and Leg Veins Our Specialty Dr Eric Dohner, MD 41-45 Dietz St Oneonta, NY 13820 607/431-2525 www.oneontalaserderm.com Modern Varicose
Peninsula Commissioning Priorities Group. Commissioning Policy Varicose Vein Referral
 NHS Devon NHS Plymouth Torbay Care Trust Peninsula Commissioning Priorities Group Commissioning Policy Varicose Vein Referral Varicose Vein Referral Guidelines 1. Description of service/treatment Most
NHS Devon NHS Plymouth Torbay Care Trust Peninsula Commissioning Priorities Group Commissioning Policy Varicose Vein Referral Varicose Vein Referral Guidelines 1. Description of service/treatment Most
SURGICAL AND ABLATIVE PROCEDURES FOR VENOUS INSUFFICIENCY AND VARICOSE VEINS
 CLINICAL POLICY SURGICAL AND ABLATIVE PROCEDURES FOR VENOUS INSUFFICIENCY AND VARICOSE VEINS Policy Number: OUTPATIENT 013.24 T2 Effective Date: July 1, 2014 Table of Contents CONDITIONS OF COVERAGE...
CLINICAL POLICY SURGICAL AND ABLATIVE PROCEDURES FOR VENOUS INSUFFICIENCY AND VARICOSE VEINS Policy Number: OUTPATIENT 013.24 T2 Effective Date: July 1, 2014 Table of Contents CONDITIONS OF COVERAGE...
SURGICAL AND ABLATIVE PROCEDURES FOR VENOUS INSUFFICIENCY AND VARICOSE VEINS
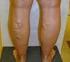 COVERAGE POLICY SURGICAL AND ABLATIVE PROCEDURES FOR VENOUS INSUFFICIENCY AND VARICOSE VEINS Policy Number: 237.E Effective Date: March 1, 2015 Table of Contents BENEFIT CONSIDERATIONS. COVERAGE RATIONALE...
COVERAGE POLICY SURGICAL AND ABLATIVE PROCEDURES FOR VENOUS INSUFFICIENCY AND VARICOSE VEINS Policy Number: 237.E Effective Date: March 1, 2015 Table of Contents BENEFIT CONSIDERATIONS. COVERAGE RATIONALE...
Suffering from varicose veins? Patient Information. ELVeS Radial Minimally invasive laser therapy of venous insufficiency
 Suffering from varicose veins? Patient Information ELVeS Radial Minimally invasive laser therapy of venous insufficiency Do you suffer from heavy legs or visible veins? This makes diseases of the veins
Suffering from varicose veins? Patient Information ELVeS Radial Minimally invasive laser therapy of venous insufficiency Do you suffer from heavy legs or visible veins? This makes diseases of the veins
Patient Information Understanding Varicose Veins
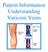 Patient Information Understanding Varicose Veins The Circulatory System Arteries carry oxygenated blood to your legs and the veins carry de-oxygenated blood away from your legs. The blood returns to the
Patient Information Understanding Varicose Veins The Circulatory System Arteries carry oxygenated blood to your legs and the veins carry de-oxygenated blood away from your legs. The blood returns to the
SURGICAL AND ABLATIVE PROCEDURES FOR VENOUS INSUFFICIENCY AND VARICOSE VEINS
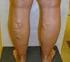 MEDICAL POLICY SURGICAL AND ABLATIVE PROCEDURES FOR VENOUS INSUFFICIENCY AND VARICOSE VEINS Policy Number: 2015T0447O Effective Date: March 1, 2015 Table of Contents BENEFIT CONSIDERATIONS COVERAGE RATIONALE
MEDICAL POLICY SURGICAL AND ABLATIVE PROCEDURES FOR VENOUS INSUFFICIENCY AND VARICOSE VEINS Policy Number: 2015T0447O Effective Date: March 1, 2015 Table of Contents BENEFIT CONSIDERATIONS COVERAGE RATIONALE
treatment of varicose and spider veins patient information SAMPLE a publication by advancing vein care
 treatment of varicose and spider veins patient information a publication by advancing vein care Since most veins lie deep to the skin s surface, vein disorders are not always visible to the naked eye.
treatment of varicose and spider veins patient information a publication by advancing vein care Since most veins lie deep to the skin s surface, vein disorders are not always visible to the naked eye.
VARICOSE VEINS. Information Leaflet. Your Health. Our Priority. VTE Ambulatory Clinic Stepping Hill Hospital
 VARICOSE VEINS Information Leaflet Your Health. Our Priority. Page 2 of 7 Varicose Veins There are no accurate figures for the number of people with varicose veins. Some studies suggest that 3 in 100 people
VARICOSE VEINS Information Leaflet Your Health. Our Priority. Page 2 of 7 Varicose Veins There are no accurate figures for the number of people with varicose veins. Some studies suggest that 3 in 100 people
Varicose Veins: Causes, Symptoms and Management. Andrew C. Stanley MD Section of Vascular Surgery
 Varicose Veins: Causes, Symptoms and Management Andrew C. Stanley MD Section of Vascular Surgery Circulation Heart (Pump)-MI, Sudden Death Arteries (Stroke, Aneurysms, Walking dysfunction, Limb loss) Veins
Varicose Veins: Causes, Symptoms and Management Andrew C. Stanley MD Section of Vascular Surgery Circulation Heart (Pump)-MI, Sudden Death Arteries (Stroke, Aneurysms, Walking dysfunction, Limb loss) Veins
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of endovenous mechanochemical ablation for varicose veins Treating varicose veins
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of endovenous mechanochemical ablation for varicose veins Treating varicose veins
X-Plain Varicose Veins Reference Summary
 X-Plain Varicose Veins Reference Summary Introduction Varicose veins are very common, in both women and men. Varicose veins can be painful and unattractive. Vein doctors use non-invasive ultrasound imaging
X-Plain Varicose Veins Reference Summary Introduction Varicose veins are very common, in both women and men. Varicose veins can be painful and unattractive. Vein doctors use non-invasive ultrasound imaging
Varicose veins - 1 -
 Varicose veins - 1 - Varicose Veins About 3 in 10 adults develop varicose veins at some time in their life. Most people with varicose veins do not have an underlying disease and they usually occur for
Varicose veins - 1 - Varicose Veins About 3 in 10 adults develop varicose veins at some time in their life. Most people with varicose veins do not have an underlying disease and they usually occur for
Health Technology Assessment of Scheduled Surgical Procedures
 HTA of Scheduled Surgical Procedures April 2013 Health Technology Assessment of Scheduled Surgical Procedures Varicose Vein Surgery April 2013 Safer Better Care About the The (HIQA) is the independent
HTA of Scheduled Surgical Procedures April 2013 Health Technology Assessment of Scheduled Surgical Procedures Varicose Vein Surgery April 2013 Safer Better Care About the The (HIQA) is the independent
The Treatment of Varicose Veins and Spider Veins
 The Treatment of Varicose Veins and Spider Veins St. Joseph Vein Center Surgical Specialists of St. Joseph, P.C. Glen H. Hastings, M.D., F.A.C.S. 820 Lester Ave., Suite 105 St. Joseph, MI 49085 (269) 983-3368
The Treatment of Varicose Veins and Spider Veins St. Joseph Vein Center Surgical Specialists of St. Joseph, P.C. Glen H. Hastings, M.D., F.A.C.S. 820 Lester Ave., Suite 105 St. Joseph, MI 49085 (269) 983-3368
CHAPTER 15 SCLEROTHERAPY FOR VENOUS DISEASE
 Introduction CHAPTER 15 SCLEROTHERAPY FOR VENOUS DISEASE Original authors: Niren Angle, John J. Bergan, Joshua I. Greenberg, and J. Leonel Villavicencio Abstracted by Teresa L. Carman New technology has
Introduction CHAPTER 15 SCLEROTHERAPY FOR VENOUS DISEASE Original authors: Niren Angle, John J. Bergan, Joshua I. Greenberg, and J. Leonel Villavicencio Abstracted by Teresa L. Carman New technology has
TREATMENT OF VARICOSE AND SPIDER VEINS Patient Info
 TREATMENT OF VARICOSE AND SPIDER VEINS Patient Info www.heartofthevillages.com TIRED & ACHING LEGS? If you suffer from varicose and spider veins, you are not alone. It is estimated that there are more
TREATMENT OF VARICOSE AND SPIDER VEINS Patient Info www.heartofthevillages.com TIRED & ACHING LEGS? If you suffer from varicose and spider veins, you are not alone. It is estimated that there are more
Recurrent Varicose Veins
 Information for patients Recurrent Varicose Veins Sheffield Vascular Institute Northern General Hospital You have been diagnosed as having Varicose Veins that have recurred (come back). This leaflet explains
Information for patients Recurrent Varicose Veins Sheffield Vascular Institute Northern General Hospital You have been diagnosed as having Varicose Veins that have recurred (come back). This leaflet explains
Greater Manchester EUR Policy Statement. Title/Topic: Varicose Veins Reference: GM003 Date: January 2015
 Greater Manchester EUR Policy Statement Title/Topic: Varicose Veins Reference: GM003 Date: January 2015 VERSION CONTROL Version Date Details Page number 0.1 23/10/2013 Initial Draft for consideration by
Greater Manchester EUR Policy Statement Title/Topic: Varicose Veins Reference: GM003 Date: January 2015 VERSION CONTROL Version Date Details Page number 0.1 23/10/2013 Initial Draft for consideration by
Varicose veins: Surgery can still be considered as an option in the treatment
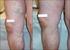 IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-0853, p-issn: 2279-0861.Volume 13, Issue 12 Ver. I (Dec. 2014), PP 72-77 Varicose veins: Surgery can still be considered as an option
IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-0853, p-issn: 2279-0861.Volume 13, Issue 12 Ver. I (Dec. 2014), PP 72-77 Varicose veins: Surgery can still be considered as an option
Varicose Vein Treatment (Endovenous Ablation of Varicose Veins)
 Scan for mobile link. Varicose Vein Treatment (Endovenous Ablation of Varicose Veins) Varicose vein treatment, also known as endovenous ablation, uses radiofrequency or laser energy to cauterize and close
Scan for mobile link. Varicose Vein Treatment (Endovenous Ablation of Varicose Veins) Varicose vein treatment, also known as endovenous ablation, uses radiofrequency or laser energy to cauterize and close
Treatment by LASER. Understanding Varicose veins and. Dr. Rajesh Mundhada (MD. DNB) Dr. Atul Rewatkar (MD)
 Understanding Varicose veins and Treatment by LASER Dr. Rajesh Mundhada (MD. DNB) Dr. Atul Rewatkar (MD) Orange city Hospital & Research Institute 19, Pandey Lay out, Veer Sawarkar Square, Nagpur 440015
Understanding Varicose veins and Treatment by LASER Dr. Rajesh Mundhada (MD. DNB) Dr. Atul Rewatkar (MD) Orange city Hospital & Research Institute 19, Pandey Lay out, Veer Sawarkar Square, Nagpur 440015
National Medical Policy
 National Medical Policy Subject: Policy Number: Varicose Veins Surgical Interventions NMP148 Effective Date*: September 2003 Updated: July 2016 Please contact the Medical Policy Department for an archive
National Medical Policy Subject: Policy Number: Varicose Veins Surgical Interventions NMP148 Effective Date*: September 2003 Updated: July 2016 Please contact the Medical Policy Department for an archive
SEDICO Newsletter Issue 8. Varicose veins
 SEDICO Newsletter Issue 8 Definition: Varicose veins Varicose veins are veins that have become enlarged and twisted. The term commonly refers to the veins on the leg, although varicose veins occur elsewhere.
SEDICO Newsletter Issue 8 Definition: Varicose veins Varicose veins are veins that have become enlarged and twisted. The term commonly refers to the veins on the leg, although varicose veins occur elsewhere.
THE VEIN CENTER. State-of-the-Art Treatment for Varicose Veins and Spider Veins
 THE VEIN CENTER State-of-the-Art Treatment for Varicose Veins and Spider Veins Vein Disorders Nearly 50 percent of the adult population suffers from undesirable, sometimes painful vein disease. The most
THE VEIN CENTER State-of-the-Art Treatment for Varicose Veins and Spider Veins Vein Disorders Nearly 50 percent of the adult population suffers from undesirable, sometimes painful vein disease. The most
Varicose Veins Backgrounder
 What Are Varicose Veins? Varicose veins are a clinical presentation of superficial venous insufficiency a condition in which veins are inefficient in returning blood to the heart due to venous hypertension.
What Are Varicose Veins? Varicose veins are a clinical presentation of superficial venous insufficiency a condition in which veins are inefficient in returning blood to the heart due to venous hypertension.
Treatment of Varicose Veins
 Varicose Vein Treatment of Varicose Veins JMAJ 47(3): 146 151, 2004 Osamu SATO Associate Professor, Department of Surgery, Saitama Medical Center Abstract: Varicose vein is a condition that was first described
Varicose Vein Treatment of Varicose Veins JMAJ 47(3): 146 151, 2004 Osamu SATO Associate Professor, Department of Surgery, Saitama Medical Center Abstract: Varicose vein is a condition that was first described
Provided by the American Venous Forum: veinforum.org
 CHAPTER 17 SURGICAL THERAPY FOR DEEP VALVE INCOMPETENCE Original author: Seshadri Raju Abstracted by Gary W. Lemmon Introduction Deep vein valvular incompetence happens when the valves in the veins (tubes
CHAPTER 17 SURGICAL THERAPY FOR DEEP VALVE INCOMPETENCE Original author: Seshadri Raju Abstracted by Gary W. Lemmon Introduction Deep vein valvular incompetence happens when the valves in the veins (tubes
Beaumont Hospital. Varicose Veins. and their TREATMENT. Professor Austin Leahy, MCh, FRCS, FRCSI WWW.VEINCLINICSOFIRELAND.COM
 Beaumont Hospital Varicose Veins and their TREATMENT Professor Austin Leahy, MCh, FRCS, FRCSI WWW.VEINCLINICSOFIRELAND.COM Department of Surgery Beaumont Hospital and Royal College of Surgeons in Ireland
Beaumont Hospital Varicose Veins and their TREATMENT Professor Austin Leahy, MCh, FRCS, FRCSI WWW.VEINCLINICSOFIRELAND.COM Department of Surgery Beaumont Hospital and Royal College of Surgeons in Ireland
CHINOOK VASCULAR - ENDOVENOUS LASER ABLATION PROCEDURE
 CHINOOK VASCULAR - ENDOVENOUS LASER ABLATION PROCEDURE Endovenous Laser Ablation is a minimally invasive option for treating great saphenous vein incompetence (leaky valves). The first stage of your surgery
CHINOOK VASCULAR - ENDOVENOUS LASER ABLATION PROCEDURE Endovenous Laser Ablation is a minimally invasive option for treating great saphenous vein incompetence (leaky valves). The first stage of your surgery
Varicose Veins Operation. Patient information Leaflet
 Varicose Veins Operation Patient information Leaflet 22 nd August 2014 WHAT IS VARICOSE VEIN SURGERY (HIGH LIGATION AND MULTIPLE AVULSIONS) The operation varies from case to case, depending on where the
Varicose Veins Operation Patient information Leaflet 22 nd August 2014 WHAT IS VARICOSE VEIN SURGERY (HIGH LIGATION AND MULTIPLE AVULSIONS) The operation varies from case to case, depending on where the
Effectiveness of foam sclerotherapy for the treatment of varicose veins
 Effectiveness of foam sclerotherapy for the treatment of varicose veins Vascular Medicine 15(1) 27 32 The Author(s) 2009 Reprints and permission: http://www. sagepub.co.uk/journalspermission.nav DOI: 10.1177/1358863X09106325
Effectiveness of foam sclerotherapy for the treatment of varicose veins Vascular Medicine 15(1) 27 32 The Author(s) 2009 Reprints and permission: http://www. sagepub.co.uk/journalspermission.nav DOI: 10.1177/1358863X09106325
Prevalence and surgical outcomes of varicose veins at Regional Institute of Medical Sciences, Imphal
 ORIGINAL ARTICLE JIACM 2013; 14(3-4): 209-13 Prevalence and surgical outcomes of varicose veins at Regional Institute of Medical Sciences, Imphal Ksh Kala Singh*, A Surjyalal Sharma**, L Sunil Singh***,
ORIGINAL ARTICLE JIACM 2013; 14(3-4): 209-13 Prevalence and surgical outcomes of varicose veins at Regional Institute of Medical Sciences, Imphal Ksh Kala Singh*, A Surjyalal Sharma**, L Sunil Singh***,
LONG TERM FOLLOW-UP AFTER ENDOVENOUS LASER ABLATION
 LONG TERM FOLLOW-UP AFTER ENDOVENOUS LASER ABLATION Giorgio Spreafico Centro Multidisciplinare Day Surgery Azienda Ospedaliera Università Padova - Italy Faculty Disclosure I have no financial relationships
LONG TERM FOLLOW-UP AFTER ENDOVENOUS LASER ABLATION Giorgio Spreafico Centro Multidisciplinare Day Surgery Azienda Ospedaliera Università Padova - Italy Faculty Disclosure I have no financial relationships
Developments in Endovascular and Endoscopic Surgery
 Developments in Endovascular and Endoscopic Surgery Radiofrequency Ablation and Laser Ablation in the Treatment of Varicose Veins Jose I. Almeida, MD, FACS, RVT, 1,2 and Jeffrey K. Raines, PhD, RVT, 1,2
Developments in Endovascular and Endoscopic Surgery Radiofrequency Ablation and Laser Ablation in the Treatment of Varicose Veins Jose I. Almeida, MD, FACS, RVT, 1,2 and Jeffrey K. Raines, PhD, RVT, 1,2
V03 Varicose Veins Surgery
 V03 Varicose Veins Surgery What are varicose veins? Varicose veins are enlarged and twisted veins in the leg. They are common and affect up to 3 in 10 people. More women than men ask for treatment, with
V03 Varicose Veins Surgery What are varicose veins? Varicose veins are enlarged and twisted veins in the leg. They are common and affect up to 3 in 10 people. More women than men ask for treatment, with
Varicose veins are generally identified
 Management of Varicose Veins Richard H. Jones, MD, MSPH, Naval Health Clinic, Quantico, Virginia Peter J. Carek, MD, MS, Medical University of South Carolina, Charleston, South Carolina Varicose veins
Management of Varicose Veins Richard H. Jones, MD, MSPH, Naval Health Clinic, Quantico, Virginia Peter J. Carek, MD, MS, Medical University of South Carolina, Charleston, South Carolina Varicose veins
PRIMARY VARICOSE VEIN management has undergone a
 Treatment of Primary Varicose Veins Has Changed with the Introduction of New Techniques Eric Mowatt-Larssen* and Cynthia K. Shortell* New technologies have produced a revolution in primary varicose vein
Treatment of Primary Varicose Veins Has Changed with the Introduction of New Techniques Eric Mowatt-Larssen* and Cynthia K. Shortell* New technologies have produced a revolution in primary varicose vein
Endovenous Laser Therapy
 Information for patients Endovenous Laser Therapy Northern General Hospital You have been given this leaflet because you will be having Endovenous Laser Therapy (EVLT). This leaflet explains more about
Information for patients Endovenous Laser Therapy Northern General Hospital You have been given this leaflet because you will be having Endovenous Laser Therapy (EVLT). This leaflet explains more about
Lower extremity venous insufficiency is a very common medical
 Diagn Interv Radiol 2012; 18:417 422 Turkish Society of Radiology 2012 INTERVENTIONAL RADIOLOGY REVIEW Endovenous laser ablation for the treatment of varicose veins Levent Oğuzkurt ABSTRACT Lower extremity
Diagn Interv Radiol 2012; 18:417 422 Turkish Society of Radiology 2012 INTERVENTIONAL RADIOLOGY REVIEW Endovenous laser ablation for the treatment of varicose veins Levent Oğuzkurt ABSTRACT Lower extremity
Diagnosis and Treatment of Venous Insufficiency and Varicose Veins
 Diagnosis and Treatment of Venous Insufficiency and Varicose Veins Joseph Conley, MD Scott and White Memorial Hospital Temple, TX September 22, 2012 Objectives Anatomy and Pathophysiology of venous insufficiency
Diagnosis and Treatment of Venous Insufficiency and Varicose Veins Joseph Conley, MD Scott and White Memorial Hospital Temple, TX September 22, 2012 Objectives Anatomy and Pathophysiology of venous insufficiency
New approaches for the treatment of varicose veins
 Surg Clin N Am 84 (2004) 1397 1417 New approaches for the treatment of varicose veins Theodore H. Teruya, MD, FACS a, Jeffrey L. Ballard, MD, FACS b,c, * a Division of Vascular Surgery, Loma Linda University,
Surg Clin N Am 84 (2004) 1397 1417 New approaches for the treatment of varicose veins Theodore H. Teruya, MD, FACS a, Jeffrey L. Ballard, MD, FACS b,c, * a Division of Vascular Surgery, Loma Linda University,
Institute of Health Economics ENDOVENOUS ABLATION INTERVENTIONS FOR SYMPTOMATIC VARICOSE VEINS OF THE LEGS
 Institute of Health Economics ENDOVENOUS ABLATION INTERVENTIONS FOR SYMPTOMATIC VARICOSE VEINS OF THE LEGS September 2014 INSTITUTE OF HEALTH ECONOMICS The Institute of Health Economics (IHE) is an independent,
Institute of Health Economics ENDOVENOUS ABLATION INTERVENTIONS FOR SYMPTOMATIC VARICOSE VEINS OF THE LEGS September 2014 INSTITUTE OF HEALTH ECONOMICS The Institute of Health Economics (IHE) is an independent,
AMERICAN VENOUS FORUM
 Revised Venous Clinical Severity Score AMERICAN VENOUS FORUM Pain : 0 Mild: 1 or other discomfort (ie, aching, heaviness, fatigue, soreness, burning) origin Occasional pain or other discomfort (ie, not
Revised Venous Clinical Severity Score AMERICAN VENOUS FORUM Pain : 0 Mild: 1 or other discomfort (ie, aching, heaviness, fatigue, soreness, burning) origin Occasional pain or other discomfort (ie, not
Varicose veins and spider veins
 Varicose veins and spider veins Summary Varicose veins are knobbly, twisted and darkish-blue in appearance, and are most commonly found on people s legs. Varicose veins are caused by faulty valves within
Varicose veins and spider veins Summary Varicose veins are knobbly, twisted and darkish-blue in appearance, and are most commonly found on people s legs. Varicose veins are caused by faulty valves within
Endovenous Treatment of Varicose Veins
 Supplement to Produced under an unrestricted educational grant from Diomed, Inc. November/December 2004 Endovenous A comprehensive guide to the techniques and technology necessary to establish and maintain
Supplement to Produced under an unrestricted educational grant from Diomed, Inc. November/December 2004 Endovenous A comprehensive guide to the techniques and technology necessary to establish and maintain
Australian Safety and Efficacy Register of New Interventional Procedures Surgical. Rapid review ASERNIP-S REPORT NO. 66
 ASERNIP S Australian Safety and Efficacy Register of New Interventional Procedures Surgical Rapid review Treatments for varicose veins ASERNIP-S REPORT NO. 66 Australian Safety & Efficacy Register of New
ASERNIP S Australian Safety and Efficacy Register of New Interventional Procedures Surgical Rapid review Treatments for varicose veins ASERNIP-S REPORT NO. 66 Australian Safety & Efficacy Register of New
Randomized clinical trial comparing endovenous laser ablation and stripping of the great saphenous vein with clinical and duplex outcome after 5 years
 Randomized clinical trial comparing endovenous laser ablation and stripping of the great saphenous vein with clinical and duplex outcome after 5 years Lars Rasmussen, MD, Martin Lawaetz, MB, Lars Bjoern,
Randomized clinical trial comparing endovenous laser ablation and stripping of the great saphenous vein with clinical and duplex outcome after 5 years Lars Rasmussen, MD, Martin Lawaetz, MB, Lars Bjoern,
Jose Almeida MD, FACS, RVT Jeff Raines PhD, RVT. Oliver Göckeritz MD Christian Wentzel MD. Tim Richards MD Daniel F. Hoheim, MD.
 Final Results 1 Jose Almeida MD, FACS, RVT Jeff Raines PhD, RVT Miami Vein Center Miami, Florida Oliver Göckeritz MD Christian Wentzel MD Venenzentrum Elserpark Leipzig, Germany John Kaufman MD Dotter
Final Results 1 Jose Almeida MD, FACS, RVT Jeff Raines PhD, RVT Miami Vein Center Miami, Florida Oliver Göckeritz MD Christian Wentzel MD Venenzentrum Elserpark Leipzig, Germany John Kaufman MD Dotter
Treating varicose veins with foam injections using ultrasound guidance
 Issue date February 2013 Information for the public NICE interventional procedures guidance advises the NHS on when and how new procedures can be used in clinical practice. Treating varicose veins with
Issue date February 2013 Information for the public NICE interventional procedures guidance advises the NHS on when and how new procedures can be used in clinical practice. Treating varicose veins with
Understanding Varicose Veins
 Understanding Varicose Veins Professor Bruce Campbell Published by Family Doctor Publications Limited in association with the British Medical Association IMPORTANT NOTICE This book is intended not as a
Understanding Varicose Veins Professor Bruce Campbell Published by Family Doctor Publications Limited in association with the British Medical Association IMPORTANT NOTICE This book is intended not as a
LASER TREATMENT FOR VARICOSE VEINS
 LASER TREATMENT FOR VARICOSE VEINS How can varicose veins be treated by laser? Laser treatment of varicose veins, Endovascular Laser Ablation (known as EVLA), is a minimally invasive procedure for treating
LASER TREATMENT FOR VARICOSE VEINS How can varicose veins be treated by laser? Laser treatment of varicose veins, Endovascular Laser Ablation (known as EVLA), is a minimally invasive procedure for treating
ENTITLEMENT ELIGIBILITY GUIDELINES VARICOSE VEINS AND SUPERFICIAL THROMBOPHLEBITIS
 ENTITLEMENT ELIGIBILITY GUIDELINES VARICOSE VEINS AND SUPERFICIAL THROMBOPHLEBITIS 1. VARICOSE VEINS MPC 00727 ICD-9 454 DEFINITION Varicose Veins of the lower extremities are a dilatation, lengthening
ENTITLEMENT ELIGIBILITY GUIDELINES VARICOSE VEINS AND SUPERFICIAL THROMBOPHLEBITIS 1. VARICOSE VEINS MPC 00727 ICD-9 454 DEFINITION Varicose Veins of the lower extremities are a dilatation, lengthening
Part Two: Against the Motion. Venous Perforator Surgery is Unproven and Does not Reduce Recurrences
 242 Trans-Atlantic Debate 17 Kianifard B, Browning L, Holdstock JM, Whiteley MS. Surgical technique and preliminary results of perforator vein closure e TRLOPS (Transluminal Occlusion of Perforators).
242 Trans-Atlantic Debate 17 Kianifard B, Browning L, Holdstock JM, Whiteley MS. Surgical technique and preliminary results of perforator vein closure e TRLOPS (Transluminal Occlusion of Perforators).
Australian Safety and Efficacy Register of New Interventional Procedures Surgical. Systematic review ASERNIP-S REPORT NO. 69
 ASERNIP S Australian Safety and Efficacy Register of New Interventional Procedures Surgical Systematic review Treatments for varicose veins ASERNIP-S REPORT NO. 69 Australian Safety & Efficacy Register
ASERNIP S Australian Safety and Efficacy Register of New Interventional Procedures Surgical Systematic review Treatments for varicose veins ASERNIP-S REPORT NO. 69 Australian Safety & Efficacy Register
Dr. Linde is an international beauty expert and an acclaimed keynote speaker at conferences all over the world. He is specialized in the treatment of
 VEINS Dr. Linde is an international beauty expert and an acclaimed keynote speaker at conferences all over the world. He is specialized in the treatment of fat pads, wrinkles and transformations of the
VEINS Dr. Linde is an international beauty expert and an acclaimed keynote speaker at conferences all over the world. He is specialized in the treatment of fat pads, wrinkles and transformations of the
EndoVenous Laser Therapy (EVLT) Information Booklet. Dr. Dueck. Varicose Veins
 EndoVenous Laser Therapy (EVLT) Information Booklet Dr. Dueck Varicose Veins Varicose veins are abnormally large veins that protrude from under the skin in the legs. They can be associated with a brown
EndoVenous Laser Therapy (EVLT) Information Booklet Dr. Dueck Varicose Veins Varicose veins are abnormally large veins that protrude from under the skin in the legs. They can be associated with a brown
Successful Endovenous Ablation
 Successful Endovenous Ablation Is it the technology or the technique? PANEL Moderator: Steve Elias, MD, is Director, Center for Vein Disease, Englewood Hospital and Medical Center, New Jersey; and Assistant
Successful Endovenous Ablation Is it the technology or the technique? PANEL Moderator: Steve Elias, MD, is Director, Center for Vein Disease, Englewood Hospital and Medical Center, New Jersey; and Assistant
Varicose Veins and Spider Veins
 page 1 Varicose Veins and Spider Veins Q: What are varicose veins and spider A: Varicose (VAR-i-kos) veins are enlarged veins that can be blue, red, or fleshcolored. They often look like cords and appear
page 1 Varicose Veins and Spider Veins Q: What are varicose veins and spider A: Varicose (VAR-i-kos) veins are enlarged veins that can be blue, red, or fleshcolored. They often look like cords and appear
Emory Healthcare s model for a PA based vein clinic
 Emory Healthcare s model for a PA based vein clinic Stephen Konigsberg PA-C No relationships to disclose Society for Vascular Surgery 2011 Vascular Annual Meeting Chicago, IL June 17, 2011 Division of
Emory Healthcare s model for a PA based vein clinic Stephen Konigsberg PA-C No relationships to disclose Society for Vascular Surgery 2011 Vascular Annual Meeting Chicago, IL June 17, 2011 Division of
Veins. Zapping Varicose. Venous diseases affect many people and represent a major cost to the health-care system.
 Zapping Varicose Veins Improving the Quality of Life for Sufferers By Jeannette Soriano, MD Venous diseases affect many people and represent a major cost to the health-care system. Hippocrates used compression
Zapping Varicose Veins Improving the Quality of Life for Sufferers By Jeannette Soriano, MD Venous diseases affect many people and represent a major cost to the health-care system. Hippocrates used compression
Causes of varicose vein recurrence: Late results of a randomized controlled trial of stripping the long saphenous vein
 Causes of varicose vein recurrence: Late results of a randomized controlled trial of stripping the long saphenous vein Rebecca J. Winterborn, AFRCSI, a Chris Foy, b and Jonothan J. Earnshaw, DM FRCS, a
Causes of varicose vein recurrence: Late results of a randomized controlled trial of stripping the long saphenous vein Rebecca J. Winterborn, AFRCSI, a Chris Foy, b and Jonothan J. Earnshaw, DM FRCS, a
A Randomized Trial Comparing Treatments for Varicose Veins
 The new england journal of medicine Original Article A Randomized Trial Comparing Treatments for Varicose Veins Julie Brittenden, M.D., Seonaidh C. Cotton, Ph.D., Andrew Elders, M.Sc., Craig R. Ramsay,
The new england journal of medicine Original Article A Randomized Trial Comparing Treatments for Varicose Veins Julie Brittenden, M.D., Seonaidh C. Cotton, Ph.D., Andrew Elders, M.Sc., Craig R. Ramsay,
Provided by the American Venous Forum: veinforum.org
 CHAPTER 1 NORMAL VENOUS CIRCULATION Original author: Frank Padberg Abstracted by Teresa L.Carman Introduction The circulatory system is responsible for circulating (moving) blood throughout the body. The
CHAPTER 1 NORMAL VENOUS CIRCULATION Original author: Frank Padberg Abstracted by Teresa L.Carman Introduction The circulatory system is responsible for circulating (moving) blood throughout the body. The
Varicose Vein Information Pack
 Varicose Vein Information Pack What are Varicose Veins? Varicose veins are veins under the skin of the legs which have become widened, bulging and tortuous. They are very common and do not cause medical
Varicose Vein Information Pack What are Varicose Veins? Varicose veins are veins under the skin of the legs which have become widened, bulging and tortuous. They are very common and do not cause medical
Venous and Lymphatic Disorders
 Venous and Lymphatic Disorders. ก. Venous and Lymphatic Disorders Varicose Veins Deep Vein Thrombosis (DVT) Lymphedema What Is Varicose Veins? Latin: Varicose = Varix = twisted Abnormal venous dilatation
Venous and Lymphatic Disorders. ก. Venous and Lymphatic Disorders Varicose Veins Deep Vein Thrombosis (DVT) Lymphedema What Is Varicose Veins? Latin: Varicose = Varix = twisted Abnormal venous dilatation
