Pathology of hairy-cell leukaemia
|
|
|
- Ferdinand Shelton
- 10 years ago
- Views:
Transcription
1 Best Practice & Research Clinical Haematology Vol. 16, No. 1, pp , 2003 doi: /ybeha , 2 Pathology of hairy-cell leukaemia Kelly J. Bethel* MD Director Robert W. Sharpe MD Department of Pathology, Scripps Clinic, La Jolla, CA 92037, USA Hairy-cell leukaemia (HCL) is a low grade B-cell lymphoproliferative process that presents either with lymphocytosis or neutropenia/monocytopenia. It is a disease predominantly of bone marrow and spleen, although it can also involve other organs and sites. Recent advances including multiparameter flow cytometry and the development of antibodies with high specificity for HCL have permitted precise distinction of typical HCL from other lymphoproliferative diseases that can morphologically mimic the appearance of HCL. Although there is a commonly held belief that HCL is associated with a significant increase in second neoplasms, several recent studies have not supported this notion. The development of extremely effective therapy for HCL results in a high incidence of complete remission. However, a significant percentage of patients continue to harbour minimal residual disease that can be revealed with immunohistochemical and flow cytometric studies. Key words: antigens, CD; immunohistochemistry; immunophenotyping; leukaemia, hairy cell; leukaemia, hairy cell pathology; leukaemia, hairy cell immunology; leukaemia, B-cell; lymphoma; lymphoma, B-cell; neoplasm; residual; receptors, interleukin-2. Hairy-cell leukaemia (HCL) has a characteristic morphological appearance in the peripheral blood, bone marrow and spleen. Coupled with distinctive immunophenotypic and cytochemical characteristics, HCL has features that permit precise classification and distinction from other B-cell lymphoproliferative processes. This unique profile is of critical clinical importance because of the specific treatment protocols utilized in the management of HCL and, in particular, the striking responsiveness of the disease to therapy with cladibrine. A subset of patients with HCL has atypical morphological, immunophenotypic or cytochemical features. Usually designated hairy-cell leukaemia-variant, this lymphoproliferative process poses both diagnostic and management challenges that are distinct from those of typical HCL. This entity, and the differential diagnosis raised by its atypical features, is the subject of a subsequent chapter. This chapter discusses the morphology and ancillary diagnostic findings of typical HCL. Emphasis is placed on the immunophenotypic profile characteristic of HCL and * Corresponding author. Tel.: þ ; Fax: þ address: [email protected] (K.J. Bethel) /03/$ - see front matter Q 2003 Elsevier Science Ltd. All rights reserved.
2 16 K. J. Bethel and R. W. Sharpe the evolving role of immunohistochemical and flow cytometry studies in the diagnosis of HCL. Transformations of HCL to other lymphoproliferative disorders and associated malignancies are also reviewed. Finally, the approach to evaluation for residual disease following treatment is addressed. MORPHOLOGY Peripheral blood The morphological presentation of HCL in the peripheral blood is variable. Some patients present with the appearance of leukaemic peripheral blood, including a moderate lymphocytosis composed of morphologically identifiable hairy cells. In other cases, the peripheral blood smear is notable only for monocytopenia or neutropenia, with scant to absent circulating hairy cells. A hairy cell is a cell of small to intermediate size with an inconspicuous appearance under low-power microscopy consistent with a mature lymphoid cell. At higher power, however, characteristic features can be identified. The sharply delineated nuclear contour is generally oval and sometimes contains an indentation, giving the nucleus a kidney-shaped appearance. Occasional cases may have nuclei that are monocytoid, folded or even bilobed in shape. The chromatin stains medium blue with Romanowskytype stains (e.g. Wright stain), is finely granular, evenly distributed, and may contain a small, centrally located, often inconspicuous nucleolus. The generous cytoplasm appears pale blue on Wright-stained peripheral smears. It has a textured appearance at high magnification not quite foamy, but vaguely flocculent. The edges of the cytoplasmic domain are often indistinct, and the pale blue colour of the cytoplasm merges imperceptibly with the unstained background of the blood smear. Many hairy cells have a cytoplasmic border best described as frayed, although the most typical cells have a visible circumferential halo of long slender villous cytoplasmic projections that produce the classic hairy appearance (see Figure 1). Distinction from the villous projections present in the circulating lymphoma cells of splenic lymphoma with villous lymphocytes (SLVL) can be challenging. One of the most useful distinguishing features is the circumferential distribution of the hairs in a hairy cell, which is different from the longer, often focally clustered projections in SLVL. Bone marrow Because HCL induces bone marrow fibrosis, it is often difficult to get cytological preparations from studies of bone marrow. Dry taps are common when attempting to collect aspirate material, and even touch preparations of the core biopsy may lack evaluable cells. However, careful scrutiny of even paucicellular cytology preparations can be useful because of the fairly characteristic morphology of the cells, which is the same as that described above in the peripheral blood. In histological sections of the core biopsy, HCL occurs as an interstitial, patchy, or diffuse non-paratrabecular lymphoid infiltrate with a well-spaced appearance due to the large cytoplasmic domain of the cells (see Figure 2). There is usually an accompanying background of reticulin fibrosis. It has been demonstrated that hairy cells synthesize and assemble fibronectin, which may contribute to the reticulin fibrosis. 1 Characteristically, the infiltrates are accompanied by extravasated red cells. Occasional blood lakes, similar to those seen in the spleen, may be observed in
3 Pathology of HCL 17 Figure 1. Peripheral blood, 100 (oil). At high power, this circulating hairy cell has a sharply circumscribed oval nucleus, textured -appearing cytoplasm and nearly circumferential villous cytoplasmic extensions, or hairs. Figure 2. Bone marrow, 20. At low power, the hairy cell infiltrate in the bone marrow is interstitial, wellspaced, and accompanied by vascular tenting from the associated reticulin fibrosis.
4 18 K. J. Bethel and R. W. Sharpe Figure 3. Bone marrow, 40. At high power, the oval-to-reniform nuclear shape of the hairy cells can be appreciated, along with the voluminous cytoplasm that yields a fried-egg appearance to the individual cells. the bone marrow. At high power, the individual cells have a fried egg appearance, with the cleared out non-staining cytoplasm surrounding the small, dark, central nucleus (see Figure 3). Minimal involvement of the bone marrow can be extremely subtle, as the cells are small and tend to intercalate interstitially in a nearly invisible manner. Careful highpower inspection of haematoxylin and eosin (H&E) stained sections under oil immersion may reveal small numbers of characteristic cells, which can be further demonstrated by using immunostains (see discussion below). Although not generally obvious in diagnostic bone marrow biopsies, abnormal bone remodelling has been described in areas of hairy-cell infiltrate, including excessive bone resorption, which can result in lytic-appearing lesions on imaging studies. 2 Spleen HCL involves the red pulp of the spleen, with expansion by the well-spaced mononuclear infiltrate, and marked diminution of the white pulp areas (see Figure 4). Within the red pulp, a characteristic feature is the formation of blood lakes, which are blood-filled areas surrounded by hairy cells. These are probably formed by disruption of splenic sinusoidal vascular architecture by the neoplastic hairy cells. The red pulp pattern of involvement in the spleen is in contrast to the pattern displayed by other low-grade B-cell processes and is a useful histopathological distinguishing feature. Lymph node HCL can involve lymph nodes, although clinically significant lymphadenopathy is usually present only in advanced disease. The pattern of involvement is interfollicular.
5 Pathology of HCL 19 Figure 4. Spleen, 10. The splenic infiltrate occupies the red pulp compartment, with atrophy of the white pulp. Here the trabeculum is lacking the normal cuff of white pulp lymphocytes and there is a diffuse red pulp infiltrate of hairy cell leukaemia. Architectural features similar to those seen in spleen and bone marrow are seen. The typical appearance is patchy areas of well-spaced small lymphoid cells with voluminous clear cytoplasm, dotted with extravasated red blood cells, and focal blood lake formation (see Figure 5). Other sites Although predominantly a disease of peripheral blood, bone marrow and spleen, HCL can involve other body sites. Hepatic involvement may be seen in HCL, and is generally portal and sinusoidal in distribution. A distinctive feature of HCL in the liver, in contrast to other low-grade B-cell processes, is the finding of so-called angiomatous lesions, which are areas of focal hairy-cell infiltrate within hepatic lobules. The foci form small cavities, which appear lined by tumour cells and are filled with tumour cells and red blood cells. 3 The features are analogous to the blood lakes described in the spleen. Ultrastructural studies demonstrate disruption of the hepatic sinusoidal lining cells with extravasation of blood cells and other changes similar to those of peliosis hepatis. 4 As noted above, HCL can involve lymph nodes, although generally this is a finding associated with high tumour burden. Abdominal lymphadenopathy has been correlated with a more immature morphological appearance and a more aggressive clinical course. Whether this clinical evolution represents part of the natural history of HCL or is a form of transformation of hairy-cell leukaemia is uncertain. 5,6 Skin involvement has been reported, including an interesting case of transient leukaemia cutis in a patient with HCL who had systemic perturbation by pulmonary
6 20 K. J. Bethel and R. W. Sharpe Figure 5. Lymph node, 10. Hairy cell leukaemia occurs in an interfollicular and paracortical pattern. Here, it nearly entirely occupies the nodal paracortex, with several residual peripheral germinal centres. tuberculosis. 7 Also described in the literature is a case of HCL discovered in a herniorrhaphy specimen. 8 ANCILLARY DIAGNOSTIC STUDIES Acid phosphatase stain (TRAP) A useful cytochemical stain for the identification of HCL is tartrate-resistant acid phosphatase (TRAP). TRAP staining can be performed on air-dried cytological preparations, such as peripheral blood smears or bone marrow aspirate smears. Positivity appears as cytoplasmic granularity, and when such staining is present in cells that are otherwise morphologically suspect as hairy cells the specificity of the cytochemical stain is high. While TRAP positivity has been reported in the cells of several lymphoid malignancies, such as B-CLL, T-CLL (now T-PLL), ATCL/L and HCL 9,it is nevertheless a helpful stain for identifying HCL as the cells in these other processes are usually morphologically distinct from hairy cells. Immunoperoxidase staining of paraffin-embedded tissue As in other lymphoproliferative diseases, immunohistochemical stains have become a mainstay in the diagnosis and monitoring of patients with HCL. Both generic B-cell antibodies and markers with specificity for HCL are of utility. The B-cell markers CD20 and CD79a are useful in the quantification of disease, uncovering a morphologically indistinct infiltrate, and cytological characterization of positive cells (see Figure 6.). DBA.44 and antibodies reactive with TRAP provide a moderate degree of specificity for
7 Pathology of HCL 21 Figure 6. Bone marrow, CD20 immunostain, 100 (oil). The immunostain demonstrates a strong and coarse membrane pattern staining of the interstitially distributed cells. This technique is very useful to highlight a minimal infiltrate, which can be very subtle and hard to appreciate on an H&E-stained section. HCL and are of use in confirming the diagnosis or in ensuring that a small residual population of B-cells following chemotherapy is indicative of HCL. DBA.44 is an antibody, originally raised against B lymphocytes, that was found to be a sensitive marker of HCL and was expressed in greater than 95% of cases. 10 While this antibody, positive in less than 20% of other low-grade B-cell lymphoproliferative disease, appears to be useful in distinguishing HCL, it marks up to 80% of cases of SLVL, an entity which is often a primary differential diagnostic consideration in suspected HCL. 11 Cytochemical staining for TRAP requires cytological material and cannot be performed on paraffin-embedded tissue. This limitation has been circumvented by the development of an antibody, 9C5, with high specificity for acid phosphatase isoenzyme 5 responsible for the cytochemical TRAP positivity of HCL. 12 This antibody marks greater than 90% of HCL and has a sensitivity slightly less than that of cytochemical TRAP. As with DBA.44, this antibody marks only a subset of the total neoplastic population and is also dimly expressed in some cases of SLVL. 13 Immunohistochemical analysis assists in the assessment of biopsies from patients with HCL in a number of ways. Both DBA.44 and 9C5, because of their moderately high specificity, are of utility in confirming suspected HCL, particularly in the absence of fresh or cytological material for flow cytometric and cytochemical studies. CD20, which shows uniform bright expression in HCL, is an excellent antibody for quantifying the neoplastic infiltrate and almost always reveals a greater degree of marrow involvement than suspected from standard histological stains. This is particularly important following treatment when there is no morphological evidence of residual HCL (see discussion of residual disease that follows). DBA.44 and 9C5 are poor substitutes for quantification because a significant portion of the neoplastic infiltrate is often not marked by these
8 22 K. J. Bethel and R. W. Sharpe antibodies. Finally, and particularly in the setting of minimal residual disease, CD20 serves to highlight small populations of B-lymphocytes that can be examined for cytological features of HCL. In our institution, CD20 is routinely employed in the assessment of all HCL bone marrows, primarily for quantification, while other antibodies are used infrequently to support a morphological impression of HCL in the absence of cytochemical or flow cytometry studies. Immunophenotype Immunophenotypic analysis of HCL has played a pivotal role in ascertaining the lineage and probable normal counterpart of the neoplastic cells in this disorder. Until recently, these studies played a relatively small role in the clinical diagnosis of HCL. However, in the past 10 years the development of (i) antibodies of high specificity and (ii) multiparameter flow cytometry have facilitated the identification of minor populations of abnormal cells and established a utility for immunophenotypic analysis in the diagnosis of HCL comparable to other lymphoproliferative disorders. The emergence of readily available immunophenotypic analysis by flow cytometry for the evaluation of haematolymphoid malignancies in the 1990s significantly altered the approach to the diagnosis of these neoplasms. Immunophenotypic profiles rapidly assumed equal footing with morphological findings in the classification of disease. However, flow cytometry requires fresh tissue. Patients with HCL frequently manifest a dearth of circulating malignant cells. As well, the consistent presence of reticulin fibrosis usually causes an inaspirable bone marrow. For these reasons, large numbers of neoplastic cells for immunophenotypic analysis are rarely available in patients with HCL. Two recent developments have altered this equation and ushered immunophenotypic analysis to the forefront in the evaluation of patients with suspected HCL. First, late generation flow cytometers are capable of simultaneous analysis of four labelled antibodies in addition to assessment of forward and side-angle light scatter. This multiparameter capability enables an approach to flow cytometry known as population analysis that can permit the identification of extremely small subsets of cells. The second and synergistic advance was the identification of antibodies to antigens with a high specificity for HCL, specifically CD103, CD25 and bright CD11c. These two advances permit the reliable identification of HCL even with leukaemic populations of less than 0.5%. Because small populations of circulating hairy cells are present in nearly all untreated patients with HCL, immunophenotypic analysis of the peripheral blood or bone marrow has become a mainstay of diagnosis, relegating acid phosphatase stains to a supporting role. Prior to the identification of cell-surface antigens with high specificity for HCL, it was recognized that the distinctive topology of the neoplastic cells resulted in characteristic light scatter properties that could be exploited as a diagnostic parameter. 14 The irregular cytoplasmic projections that impart the characteristic hairy appearance of the leukaemic cells cause an increase in right-angle light scatter similar to that of monocytes and distinct from other benign and neoplastic lymphocytes. Coupling of a lineage-specific marker with right-angle light scatter permits the rapid identification of an abnormal lymphocytic population suspect for HCL. This characteristic finding is exploited in many laboratories as part of strategies for performing cost-effective immunophenotypic analysis, serving as a trigger for the assessment of antigens specific to hairy cell leukaemia. CD11c is a cell-surface antigen that is expressed on granulocytes, monocytes, natural killer cells and small populations of normal Tand B lymphocytes. This antigen is
9 Pathology of HCL 23 consistently expressed in HCL. 15 CD11c is expressed in other lymphoproliferative disorders, including B-cell chronic lymphocytic leukaemia/small lymphocytic lymphoma and splenic marginal zone lymphoma, seeming to limit the diagnostic utility of this antigen. 16 However, the intensity of expression of CD11c in HCL is 30-fold greater than B-CLL/SLL and SMZL. Similar brightness of CD11c expression is encountered only in prolymphocytic leukaemia. 17 Hence, the bright expression of CD11c retains utility in the diagnosis of HCL. The IL-2 receptor CD25 is expressed in greater than 90% of HCL. 18 However, this marker is also expressed in nearly half of B-CLL and other types of B-cell lymphoproliferative disorders. As with CD11c, CD25 is typically brighter in HCL than in other lymphoproliferative diseases. Unlike CD11c, this difference in intensity is small and does not permit reliable distinction between HCL and other B-cell processes. 17 Nevertheless, CD25 retains utility in the immunophenotypic analysis of HCL. Because of the reliable expression of this antigen in HCL, absence of CD25 can be a pertinent finding in excluding typical HCL. CD103 is a cell surface antigen initially identified as a marker of intraepithelial T lymphocytes. This antigen is expressed in greater than 95% of cases of HCL. 19 Except for in a minority of splenic marginal zone lymphomas, it is rarely expressed in other B-cell lymphoproliferative diseases. 17 Hence, expression of this antigen is the most specific immunophenotypic marker of HCL. The immunophenotypic profile of typical HCL is remarkably consistent. Pan B- cell antigens are expressed, with high intensity of CD20 and CD22. In addition, the cells express CD11c, CD25 and CD103. Surface immunoglobulin is usually brightly positive. This characteristic phenotype will be expressed in greater than 90% of cases of HCL. Occasionally, otherwise typical HCL will manifest immunophenotypic aberrations from the characteristic pattern. CD10 expression is the most common variation, present in 25% of cases in one large series. 17 CD5 is expressed in less than 5% of cases of HCL. Lastly, antigens commonly expressed in HCL, including CD11c, CD25 and CD103, may occasionally be absent. These phenotypic variations, while not excluding a diagnosis of typical HCL, raise additional diagnostic considerations, including variant HCL, splenic marginal zone lymphoma, follicular lymphoma and atypical B-CLL. 20 The evolution of flow cytometry during the past decade has led to an important shift in data analysis. With a limited number of parameters that could be simultaneously assessed, interpretation relied predominantly on the percentage of cells expressing evaluated antigens and co-expression by populations of cells of two antigens. The availability of flow cytometers capable of analysing five or six parameters simultaneously, coupled with the burgeoning knowledge of patterns of antigen expression and intensity in haematolymphoid malignancies, have permitted through the use of sophisticated gating strategies comprehensive analysis of specific abnormal populations of cells. This approach to interpretation of immunophenotypic data has greatly enhanced the sensitivity of flow cytometry in the detection of abnormal cellular populations. The distinctive and consistent immunophenotype observed in HCL has established population analysis by flow cytometry as a primary diagnostic tool. HCL can be reliably detected and characterized in the peripheral blood, bone marrow, and other body tissues even when the neoplastic cells comprise less than 1% of the analysed population. 21 When neoplastic cells are present in large numbers, the distinctive phenotype of typical HCL usually is easily distinguished from other B-cell lymphoproliferative diseases. In the setting of a very small number of leukaemic cells
10 24 K. J. Bethel and R. W. Sharpe it is important to distinguish this population from small subsets of normal lymphocytes with unusual antigen expression, specifically CD103 and CD11c. The bright expression of CD20, CD22 and CD11c, coupled with immunoglobulin light-chain restriction, usually permits discrimination between HCL and small normal populations expressing CD103 and CD11c. A highly sensitive and specific technique, immunophenotypic profiling now occupies a primary role in the diagnosis of HCL. The presence of characteristic morphological and immunophenotypic findings in the peripheral blood and/or bone marrow is sufficient to establish a definitive diagnosis and supplants the need for cytochemical and immunohistochemical studies, although the latter is of considerable utility in quantification, assessment of response to therapy and exclusion of minimal residual disease. Electron microscopy evaluation Hairy cells have a characteristic appearance under the electron microscope. In the past, prior to the wide availability of reliable immunophenotyping, electron microscopy has been useful for diagnosis. The presence of ribosome-lamellar structures in the cytoplasm is the characteristic ultrastructural feature of HCL, although they can also be rarely seen in other neoplastic and non-neoplastic cells. 22 Furthermore, their presence and appearance can be affected by interferon therapy, which can result in a morphologically immature structure. 23 Ultrastructural evaluation of the hairs demonstrates thin microvilli with associated actin-containing intermediate filaments. With TPA induction, the cells sprout long dendritic processes rich in submembranous F-actin, which makes intertwined networks. 24 In practical terms, with the advent of widely available immunostaining and flow cytometry, electron microscopy is rarely useful in the diagnosis of HCL. TRANSFORMATIONS OF HAIRY-CELL LEUKAEMIA Transformation to aggressive B-cell neoplasms Very rarely, HCL transforms to an aggressive lymphoproliferative process with an immature cytological appearance that retains some morphological, cytochemical, immunophenotypic or molecular features of the antecedent HCL. This process is referred to as blastic transformation of HCL. 25,26 There are a few case reports of diffuse large B-cell lymphoma arising in patients with HCL However, none of these reports demonstrate a clonal relationship between the two neoplasms, and whether these cases represent transformation of HCL or an associated but different lymphoid malignancy is uncertain. Transformations to other low-grade B-cell processes There are rare case reports of transformations between HCL and other low-grade B-cell processes. In one case, a patient with HCL transformed after 9 years to a lymphoplasmacytic lymphoma with an accompanying circulating IgG monoclonal protein. 31 A second report describes transformation from B-CLL to HCL characterized by acquisition of CD11c and CD Both reports pre-date
11 Pathology of HCL 25 the recognition of CD103 as a specific marker of HCL and the description of SLVL, an entity that can closely mimic HCL. Hence, in the absence of recent confirmatory reports, the existence of transformation between HCL and other low-grade B-cell processes is speculative. ASSOCIATED MALIGNANCIES IN HAIRY-CELL LEUKAEMIA There is a widely held belief that patients with HCL have an increased risk of developing a variety of second malignancies. 33 This notion has been supported in several recent and relatively small series, one suggesting a link to therapy with alpha-interferon. 34,35 However, several recent large series, including one with over 1000 patients, failed to show a significant increase in second malignancies in patients with HCL or a relationship between secondary neoplasia and interferon therapy While it is likely that HCL confers, at most, only a very modest increase in the overall incidence of second neoplasms, there are several intriguing disease associations that warrant discussion, specifically multiple myeloma (MM) and large granular lymphocytic (LGL) leukaemia. Multiple myeloma There have been case reports of coexistent HCL and MM 39,40 and a small series of three patients with synchronous HCL and MM. 41 However, no studies document evidence of a clonal linkage between the neoplasms in these patients. Additionally, the large series of HCL patients described above document no increase in the incidence of MM. It can be concluded that patients with HCL are at no increased risk of MM. Whether HCL rarely transforms to MM is an unanswered question awaiting molecular evidence of clonal relationship. LGL (T/NK cell processes) There appears to be an association between HCL and lymphoproliferative disorders of large granular lymphocytes Two recent studies have investigated this apparent association. In one series of 32 HCL patients, 10 were found to have populations of clonal cytotoxic T cells. 45 A second study demonstrated in HCL patients oligoclonal T- cell populations with restricted and skewed TCRBV repertoires. 46 These findings suggest that aberrant T-cell proliferations are common in HCL and may reflect a response to immunological aberrations induced by HCL, or may be due to the same stem cell defect as the co-existent HCL. ASSESSMENT OF RESIDUAL DISEASE The assessment of patients with HCL for evidence of residual disease and the clinical significance of minimal residual leukaemia are vexed issues. While the sequential development of treatment strategies employing alpha-interferon, deoxycoformycin, and 2-chlorodeoxyadenosine (2-CdA) have proven remarkable therapeutic advances in the treatment of HCL 47, it has become evident that a significant percentage of patients in apparent remission have demonstrable residual disease using moderately sensitive techniques, including immunohistochemistry and flow cytometry. 48 Often, the degree
12 26 K. J. Bethel and R. W. Sharpe of persistent marrow infiltration is significant, comprising up to 10% of marrow cellularity. Hence, the term minimal residual disease in the context of HCL often reflects the difficulty in morphological assessment of bone marrow using routine histological and cytological stains rather than the utilization of extraordinarily sensitive techniques to ferret out rare residual neoplastic cells. Whether minimal residual disease in HCL predicts clinical relapse is uncertain. Existing studies, although difficult to compare because of varying criteria of residual HCL, suggest that the identification of MRD in HCL has no or only modest positive predictive value for relapse and is therefore of uncertain clinical utility. The counter-intuitive notion that a significant percentage of HCL patients in complete remission (CR) have readily demonstrable residual disease requires that the terms complete remission and minimal residual disease be clarified. In the context of HCL, CR following therapy is generally defined as: 1. absence of organomegaly; 2. normalization of peripheral blood counts; 3. absence of peripheral blood and bone marrow evidence of HCL using routine histological and cytochemical stains. Minimal residual disease, although specific criteria vary, is characterized as identification of residual HCL using either immunohistochemical or flow cytometric techniques in the absence of recognizable leukaemic infiltration of the marrow using routine histological and cytochemical stains. The vast majority of these patients with MRD will meet the other criteria of CR. Dramatic therapeutic advances in the treatment of HCL, culminating in the development of 2-CdA, have resulted in CR rates of 75 91%. 52 Although long-term follow-up studies have confirmed the durability of clinical response, the utilization of immunohistochemical stains and flow cytometry in the assessment of post-treatment peripheral blood and bone marrow specimens has demonstrated that many patients in CR have evidence of MRD. In contrast to the highly sensitive techniques utilizing polymerase chain reaction (PCR) and sophisticated cytometric analysis to detect minute populations of neoplastic cells ( %) in acute leukaemia and chronic myeloid leukaemia, relatively crude techniques will identify residual HCL in up to 50% of patients in CR. This reflects the insensitivity and inadequacy of routine morphological assessment in the evaluation of post-treatment bone marrows in HCL patients. The interstitial and single-cell infiltration pattern characteristic of HCL and the cytolog appearance of the cells that can mimic the appearance of granulocytic and myeloid precursors in histological sections stained with haematoxylin and eosin (H&E) combine to mask minor populations of residual hairy cells comprising up to 10% of marrow cellularity. Immunostains for CD20 and DBA.44 readily demonstrate residual HCL. However, there are several important caveats in the interpretation of immunostains. Both CD20 and DBA.44 are non-specific, and they stain up to 6 and 2% of non-neoplastic marrow lymphocytes. 53 Additionally, a subset of marrow mononuclear cells possesses an abundant cytoplasmic rim that can mimic the histological appearance of HCL. Hence, caution must be exercised in the interpretation of immunostains with careful evaluation of both the percentage and cytological appearance of the cells expressing CD20 and DBA.44. If the population of cells is less than 5%, a definitive diagnosis of residual HCL should be reserved for cases with additional supportive evidence such
13 as TRAP (cytochemical or immunohistochemical) positive cells or flow-cytometric evidence of HCL. Immunophenotypic analysis of the peripheral blood or bone marrow can also be employed in the assessment for MRD in HCL. Only a small number of neoplastic cells are typically present in the post-therapy peripheral blood and marrow aspirate specimens, the latter presumably because of the pericellular fibrosis induced by the HCL infiltrates. Because of the highly specific immunophenotypic profile and identification of light-chain restriction by population analysis, even very small populations (, 0.5%) can be definitively identified. Finally, while there has been a decline in utilization of cytochemical stains in the assessment of haematolymphoid malignancies caused by the higher specificity and general availability of immunohistochemical and flow cytometry studies, acid phosphatase stains for evaluating TRAP-positive cells retain their utility for evaluating MRD in HCL. The presence of even a few brightly TRAP-positive cells that are cytologically appropriate is strong presumptive evidence of MRD. It is valid to question the clinical relevance of performing additional studies to reveal MRD in bone marrow specimens with no morphological evidence of HCL. Since recent studies have demonstrated that MRD has positive predictive value for eventual clinical relapse, these studies are probably justifiable, although institution and provider-specific relevance should be established. If immunohistochemical studies are not performed, the pathological interpretation should specify that MRD cannot be excluded. Other important criteria when considering immunohistochemical studies are the experience of the interpreting pathologist with HCL, the quality of histological sections, and the adequacy of the core biopsy. In reviewing numerous post-treatment bone marrows from referring institutions, we have observed that a remarkable degree of immunohistochemically demonstrable HCL can be masked by poor-quality histological preparation. Additionally, even with well-prepared H&E sections, we are occasionally surprised at the degree of residual marrow infiltration by HCL revealed with immunostains. Hence, in our institution, immunostains for CD20 are routinely performed in all post-treatment marrows. SUMMARY Pathology of HCL 27 HCL is a B-cell lymphoproliferative disease with distinctive morphological, cytochemical and immunophenotypic features. An accurate diagnosis is of critical importance because of the unique approach to the management of HCL patients. The neoplastic cell in HCL is an abnormal lymphocyte with morphological features that permit distinction from other lymphoproliferations. The cells are small with round, oval and reniform nuclei that have smooth nuclear margins, granular chromatin and a single small nucleolus. The ample cytoplasm is grey blue and textured with margins that are often frayed with fine projections imparting a hairy appearance. While the appearance of the neoplastic cells in HCL is distinctive, there are similarities with other lymphoproliferative diseases, especially SLVL. Hence, the diagnosis should be confirmed with ancillary studies. The cytochemical demonstration of TRAP, the first such study developed, has retained a high degree of specificity and utility. Immunophenotypic analysis by flow cytometry has moved to the forefront of confirmatory studies in HCL owing to the recognition of an antigen expression profile that is nearly unique and the development of multi-parameter flow cytometers that can precisely characterize small cellular populations. The leukaemic cells in HCL express bright CD20, bright CD22, bright CD11c, CD25 and CD103.
14 28 K. J. Bethel and R. W. Sharpe HCL almost nevermorphologically transforms to a clinically more aggressive disease a feature unique among low-grade B-cell lymphoproliferative diseases. Additionally, although thereis a commonly held belief that HCL is associated with a significant increasein second neoplasms, several recent studies have not supported this notion. The subtle marrow infiltration pattern of HCL is a challenge to the interpretation of post-treatment bone marrow biopsies. Residual disease of less than 5% of marrow cellularity is generally not recognizable in routinely stained preparations. Hence, immunostains for CD20 or immunophenotypic analysis by flow cytometry are necessary to exclude minimal residual disease. Practice points HCL can present with abnormalities of the peripheral blood, including cytopenias or lymphocytosis the cytological and histological appearance of HCL is highly characteristic. However, because other lymphoproliferative processes can mimic this appearance, confirmatory studies should be performed traditional confirmatory testing for HCL consists of cytochemical staining for TRAP within the cytoplasm of the neoplastic cells immunohistochemical stains, including TRAP, CD20 and DBA.44, are useful both in confirming the diagnosis and quantifying the neoplastic infiltrate flow cytometry is the most specific confirmatory methodology, with typical cases showing expression of CD20, CD11c, CD25 and CD103 HCL rarely undergoes transformation to a morphologically and clinically more aggressive disease. Second malignancies are probably not increased ancillary diagnostic techniques, including immunohistochemical and flow cytometry studies, are important post-therapy to evaluate minimal residual disease that is often inapparent in routinely stained histological sections Research agenda the pathology of typical HCL is well-established. However, there is a need to focus attention on utilizing advances in immunophenotypic and molecular techniques to further clarify the characteristics of other lymphoproliferative disorders that mimic the appearance of HCL, and thus further delineate the distinctions between typical HCL, variant HCL, splenic marginal zone lymphoma and prolymphocytic leukaemia the nature and pathogenesis of the peculiar and consistent myelofibrosis that occurs in association with HCL needs additional characterization molecular evaluation of the occasional synchronous and metachronous association between HCL and MM will elucidate the relationship in individual patients, between these two terminally differentiated B-cell proliferations whether patients with no demonstrable post-therapy disease by routine and immunohistochemical evaluation of the bone marrow are cured of HCL or have minimal residual disease below the threshold of detection remains a vexed question. Sensitive molecular studies on unfixed bone marrow specimens from post-therapy HCL patients will assist in resolving this issue
15 Pathology of HCL 29 the clinical significance of detectable minimal residual disease in HCL is uncertain because of conflicting reports. Studies that stratify patients according to the quantity of residual disease are needed to establish a threshold that identifies patients at high risk of clinical relapse REFERENCES 1. Burthem J & Cawley JC. Specific tissue invasion, localization and matrix modification in hairy cell leukemia. Leukemia & Lymphoma 1994; 14(supplement 1): Marcelli C, Chappard D, Rossi JF et al. Histologic evidence of an abnormal bone remodeling in B cell malignancies other than multiple myeloma. Cancer 1988; 62: Roquet JL, Zafrani ES, Farcet JP et al. Histopathologicalal lesions of the liver in hairy cell leukemia: a report of 14 cases. Hepatology 1985; 5: Zafrani ES, Degos F, Guigui B et al. The hepatic sinusoid in hairy cell leukemia: an ultrastructural study of 12 cases. Human Pathology 1987; 18: Mercieca J, Puga M, Matutes E et al. Incidence and significance of abdominal lymphadenopathy in hairy cell leukaemia. Leukemia & Lymphoma 1994; 14 (supplement 1): Mercieca J, Matutes E, Moskovic E et al. Massive abdominal lymphadenopathy in hairy cell leukemia: a report of 12 cases. British Journal of Haematology 1992; 82: Bilsland D, Shahriari S, Douglas WS et al. Transient leukaemia cutis in hairy-cell leukemia. Clinical and Experimental Dermatology 1991; 16: Melaragno MJ, Theil KS, Marsh WL et al. Hairy cell leukemia involving an inguinal hernial sac. Acta Haematologica 1990; 84: Usui T, Konishi H, Sawada H & Uchino H. Existence of tartrate-resistant acid phosphatase activity in differentiated lymphoid leukemic cells. American Journal of Hematology 1982; 12: * 10. Hounieu H, Chittal SM, Al Saati T et al. Hairy cell leukemia: diagnosis of bone marrow involvement in paraffin-embedded sections with monoclonal antibody DBA.44. American Journal of Clinical Pathology 1992; 98: Salomon-Nguyen F, Valensi F, Troussard X & Flandin G. The value of the monoclonal antibody DBA44 in the diagnosis of B-lymphoid disorders. Leukemia Research 1996; 20: Janckila AJ, Cardwell EM, Yam LT & Li C-Y. Hairy cell identification by immunohistochemistry of tartrateresistant acid phosphatase. Blood 1995; 85: Hoyer JD, Li C-Y, Yam LT et al. Immunohistochemical demonstration of acid phosphatase isoenzyme 5 (tartrate-resistant) in paraffin sections of hairy cell leukemia and other hematologic disorders. American Journal of Clinical Pathology 1997; 108: van Bockstaele DR, Berneman ZN & Peetermans ME. Flow cytometric analysis of hairy cell leukemia using right angle light scatter. Cytometry 1986; 7: Schwarting R, Stein H & Wang CY. The monoclonal antibodies as-hcl1 (aleu-14) and as-hcl3 (aleu- M5) allow the diagnosis of hairy cell leukemia. Blood 1985; 65: Marotta G, Raspadori D, Sestigiani C et al. Expression of CD11c antigen in B-cell lymphoproliferative disorders. Leukemia & Lymphoma 2000; 37: * 17. Robbins BA, Ellison DJ, Spinosa JC et al. Diagnostic application of two-color flow cytometry in 161 cases of hairy cell leukemia. Blood 1993; 82: Korsmeyer SL, Greene WC, Cossman J et al. Rearrangement and expression of immuoglobulin genes and expression of Tac antigen in hairy cell leukemia. Proceedings of the National Academy of Sciences of the USA 1983; 80: * 19. Mulligan SP, Travade P, Matutes E et al. B-ly-7, a monoclonal antibody reactive with hairy cell leukemia, also defines an activation antigen on normal CD8 þ T cells. Blood 1990; 77: * 20. Matutes E, Morilla R, Owusu-Ankomah K et al. The immunophenotype of hairy cell leukemia (HCL). Proposal for a scoring system to distinguish HCL from B-cell disorders with hairy or villous projections. Leukemia & Lymphoma 1994; 14 (supplement 1): * 21. Cornfield DB, Mitchell Nelson DM, Rimsza LM et al. The diagnosis of hairy cell leukemia can be established by flow cytometric analysis of peripheral blood, even in patients with low levels of circulating malignant cells. American Journal of Hematology 2001; 67: Brunning RD & Parkin J. Ribosome-lamella complexes in neoplastic hairy cells. American Journal of Pathology 1975; 79:
16 30 K. J. Bethel and R. W. Sharpe 23. Morroni M & Cinti S. Hairy cell leukemia: an ultrastructural study of hairy cells before and after interferon therapy. Tumori 1995; 81: Caligaris-Cappio F, Bergui L, Tesio L et al. Cytoskeleton organization is aberrantly rearranged in the cells of B chronic lymphocytic leukemia and hairy cell leukemia. Blood 1986; 67: Friedline JA, Crisan D & Chen J. Blastic transformation in a case of hairy cell leukemia. Molecular Diagnosis 1998; 3: Nazeer T, Burkart P, Dunn H & Jennings TA. Blastic transformation of hairy cell leukemia. Archives of Pathology and Laboratory Medicine 1997; 121: Arnalich F, Camacho J, Jimenez C et al. Occurrence of immunoblastic B-cell lymphoma in hairy cell leukemia. Cancer 1987; 59: Downing JR, Grossi CE, Smedberg CT & Burrows PD. Diffuse large cell lymphoma in a patient with hairy cell leukemia: immunoglobulin gene analysis reveals separate clonal origins. Blood 1986; 67: Lopera GA, Alvarez OA & Lee M. Large cell lymphoma of the colon presenting as a second malignancy in a patient with hairy cell leukemia. Journal of Clinical Gastroenterology 1995; 21: Franssila KO. Coincidental hairy cell leukemia and large cell malignant lymphoma. Archives of Pathology and Laboratory Medicine 1979; 103: Huang AT, Silverstein L, Gonia SL et al. Transformation of hairy cell leukemia to EBV genome-containing aggressive B cell lymphoma. Leukemia 1987; 1: Duchayne E, Delsol G, Kuhlein E et al. Hairy cell transformation of a B-cell chronic lymphocytic leukemia; a morphological, cytochemical, phenotypic and molecular study. Leukemia 1991; 5: Jacobs RH, Vokes EE & Golomb HM. Second malignancies in hairy cell leukemia. Cancer 1985; 56: Au WY, Klasa RJ, Gallagher R & Le N. Second malignancies in patients with hairy cell leukemia in British Columbia: a 20-year experience. Blood 1998; 92: Kampmeier P, Spielberger R, Dickstein J et al. Increased incidence of second neoplasms in patients treated with interferon alpha 2b for hairy cell leukemia: a clinicopathologic assessment. Blood 1994; 83: Pawson R, A Hern R & Catovsky D. Second malignancy in hairy cell leukemia: no evidence of increased incidence after treatment with interferon alpha. Leukemia & Lymphoma 1996; 22: * 37. Federico M, Zinzani PL, Frassoldati A & Vinceti M. Risk of second cancer in patients with hairy cell leukemia: long-term follow-up. Journal of Clinical Oncology 2002; 20: Kurzrock R, Strom SS, Estey E & O Brien S. Second cancer risk in hairy cell leukemia: analysis of 350 patients. Journal of Clinical Oncology 1997; 15: Saif MW & Greenberg BR. Multiple myeloma and hairy cell leukemia: a rare association or coincidence? Leukemia & Lymphoma 2001; 42: Aronowitz J, Baral E & Dalal BI. Late transition of hairy cell leukemia to multiple myeloma. American Journal of Hematology 1993; 44: Catovsky D, Costello C, Loukopoulos D & Fessas PR. Hairy cell leukemia and myelomatosis: chance association or clinical manifestations of the same B-cell disease spectrum. Blood 1981; 57: Bassan R, Rambaldi A, Allavena P & Abbate M. Association of large granular lymphocyte/natural killer cell proliferative disease and second hematologic malignancy. American Journal of Hematology 1988; 29: Airo P, Rossi G, Facchetti F & Marocolo D. Monoclonal expansion of large granular lymphocytes with a CD4 þ CD8dim þ/2 phenotype associated with hairy cell leukemia. Haematologica 1995; 80: Marolleau FP, Henni T, Gaulard P et al. Hairy cell leukemia associated with large granular lymphocyte leukemia: immunologic and genomic study, effect of interferon treatment. Blood 1988; 72: * 45. Xie XY, Sorbara L, Kreitman RJ & Fukushima PI. Development of lymphoproliferative disorder of granular lymphocytes in association with hairy cell leukemia. Leukemia & Lymphoma 2000; 37: Kluin-Nelemans JC, Kester MG, Melenhorst JJ & Landegent JE. Persistent clonal excess and skewed T-cell repertoire in T cells from patients with hairy cell leukemia. Blood 1996; 87: Andrey J & Saven A. Therapeutic advances in the treatment of hairy cell leukemia. Leukemia Research 2001; 25: * 48. Ellison DJ, Sharpe RW, Robbins BA et al. Immunomorphologic analysis of bone marrow biopsies after treatment with 2-chlorodeoxyadenosine for hairy cell leukemia. Blood 1994; 84: * 49. Matutes E, Meeus K, McLennen K & Catovsky D. The significance of minimal residual disease in hairy cell leukemia treated with deoxycoformycin: a long-term follow-up study. British Journal of Haematology 1997; 98: Bastie JN, Cazals-Hatem D, Daniel MTet al. Five years follow-up after 2-chlorodeoxyadensine treatment in 30 patients with hairy cell leukemia: evaluation of minimal residual disease and CD4 þ lymphocytopenia after treatment. Leukemia & Lymphoma 1999; 35:
17 Pathology of HCL 31 * 51. Tallman MS, Hakimian D, Kopecky KJ et al. Minimal residual disease in patients with hairy cell leukemia in complete remission treated with 2-chlorodeoxyadenosine or 2 0 -deoxycoformycin and prediction of early relapse. Clinical Cancer Research 1999; 5: Saven A, Burian C, Koziol JA & Piro LD. Long-term follow-up of patients with hairy cell leukemia after cladibrine treatment. Blood 1998; 92: O Donnell LR, Alder SL, Balis UJ et al. Immunohistochemical reference ranges for B lymphocytes in bone marrow biopsy paraffin sections. American Journal of Clinical Pathology 1995; 104:
LYMPHOMA. BACHIR ALOBEID, M.D. HEMATOPATHOLOGY DIVISION PATHOLOGY DEPARTMENT Columbia University/ College of Physicians & Surgeons
 LYMPHOMA BACHIR ALOBEID, M.D. HEMATOPATHOLOGY DIVISION PATHOLOGY DEPARTMENT Columbia University/ College of Physicians & Surgeons Normal development of lymphocytes Lymphocyte proliferation and differentiation:
LYMPHOMA BACHIR ALOBEID, M.D. HEMATOPATHOLOGY DIVISION PATHOLOGY DEPARTMENT Columbia University/ College of Physicians & Surgeons Normal development of lymphocytes Lymphocyte proliferation and differentiation:
Interesting Case Review. Renuka Agrawal, MD Dept. of Pathology City of Hope National Medical Center Duarte, CA
 Interesting Case Review Renuka Agrawal, MD Dept. of Pathology City of Hope National Medical Center Duarte, CA History 63 y/o male with h/o CLL for 10 years Presents with worsening renal function and hypercalcemia
Interesting Case Review Renuka Agrawal, MD Dept. of Pathology City of Hope National Medical Center Duarte, CA History 63 y/o male with h/o CLL for 10 years Presents with worsening renal function and hypercalcemia
Bone Marrow Evaluation for Lymphoma. Faizi Ali, MD Hematopathology Fellow William Beaumont Hospital
 Bone Marrow Evaluation for Lymphoma Faizi Ali, MD Hematopathology Fellow William Beaumont Hospital Indications One of the most common indications for a bone marrow biopsy is to evaluate for malignant lymphoma.
Bone Marrow Evaluation for Lymphoma Faizi Ali, MD Hematopathology Fellow William Beaumont Hospital Indications One of the most common indications for a bone marrow biopsy is to evaluate for malignant lymphoma.
B-cell Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma
 2008 WHO Classification of Lymphoid Neoplasms: Small B-Cell Neoplasms Chronic lymphocytic leukemia/small lymphocytic lymphoma B-cell prolymphocytic leukemia Splenic marginal zone B-cell lymphoma Hairy
2008 WHO Classification of Lymphoid Neoplasms: Small B-Cell Neoplasms Chronic lymphocytic leukemia/small lymphocytic lymphoma B-cell prolymphocytic leukemia Splenic marginal zone B-cell lymphoma Hairy
Malignant Lymphomas and Plasma Cell Myeloma
 Malignant Lymphomas and Plasma Cell Myeloma Dr. Bruce F. Burns Dept. of Pathology and Lab Medicine Overview definitions - lymphoma lymphoproliferative disorder plasma cell myeloma pathogenesis - translocations
Malignant Lymphomas and Plasma Cell Myeloma Dr. Bruce F. Burns Dept. of Pathology and Lab Medicine Overview definitions - lymphoma lymphoproliferative disorder plasma cell myeloma pathogenesis - translocations
Flow Cytometric Diagnosis of Low Grade B-cell Leukemia/Lymphoma
 Flow Cytometric Diagnosis of Low Grade B-cell Leukemia/Lymphoma Maryalice Stetler-Stevenson, M.D., Ph.D. Flow Cytometry Unit, Laboratory of Pathology, DCS, NCI,NIH DEPARTMENT OF HEALTH & HUMAN SERVICES
Flow Cytometric Diagnosis of Low Grade B-cell Leukemia/Lymphoma Maryalice Stetler-Stevenson, M.D., Ph.D. Flow Cytometry Unit, Laboratory of Pathology, DCS, NCI,NIH DEPARTMENT OF HEALTH & HUMAN SERVICES
Flow Cytometric Evaluation of B-cell Lymphoid Neoplasms
 Clin Lab Med 27 (2007) 487 512 Flow Cytometric Evaluation of B-cell Lymphoid Neoplasms Fiona E. Craig, MD Division of Hematopathology, Department of Pathology, University of Pittsburgh School of Medicine,
Clin Lab Med 27 (2007) 487 512 Flow Cytometric Evaluation of B-cell Lymphoid Neoplasms Fiona E. Craig, MD Division of Hematopathology, Department of Pathology, University of Pittsburgh School of Medicine,
Subtypes of AML follow branches of myeloid development, making the FAB classificaoon relaovely simple to understand.
 1 2 3 4 The FAB assigns a cut off of 30% blasts to define AML and relies predominantly on morphology and cytochemical stains (MPO, Sudan Black, and NSE which will be discussed later). Subtypes of AML follow
1 2 3 4 The FAB assigns a cut off of 30% blasts to define AML and relies predominantly on morphology and cytochemical stains (MPO, Sudan Black, and NSE which will be discussed later). Subtypes of AML follow
Mature Lymphoproliferative disorders (2): Mature B-cell Neoplasms. Dr. Douaa Mohammed Sayed
 Mature Lymphoproliferative disorders (2): Mature B-cell Neoplasms Dr. Douaa Mohammed Sayed Small lymphocytic lymphoma/b-cell chronic lymphocytic leukemia BMB: nodular, interstitial, diffuse or a combination
Mature Lymphoproliferative disorders (2): Mature B-cell Neoplasms Dr. Douaa Mohammed Sayed Small lymphocytic lymphoma/b-cell chronic lymphocytic leukemia BMB: nodular, interstitial, diffuse or a combination
Pathology No: SHS-CASE No. Date of Procedure: Client Name Address
 TEL #: (650) 725-5604 FAX #: (650) 725-7409 Med. Rec. No.: Date of Procedure: Sex: A ge: Date Received: Date of Birth: Account No.: Physician(s): Client Name Address SPECIMEN SUBMITTED: LEFT PIC BONE MARROW,
TEL #: (650) 725-5604 FAX #: (650) 725-7409 Med. Rec. No.: Date of Procedure: Sex: A ge: Date Received: Date of Birth: Account No.: Physician(s): Client Name Address SPECIMEN SUBMITTED: LEFT PIC BONE MARROW,
Hematopathology VII Acute Lymphoblastic Leukemia, Chronic Lymphocytic Leukemia, And Hairy Cell Leukemia
 John L. Kennedy, M.D. UIC College of Medicine Associate Professor of Clinical Pathology M2 Pathology Course Lead Pathologist, VA Chicago Health Care System Lecture #43 Phone: (312) 569-6690 Thursday, November
John L. Kennedy, M.D. UIC College of Medicine Associate Professor of Clinical Pathology M2 Pathology Course Lead Pathologist, VA Chicago Health Care System Lecture #43 Phone: (312) 569-6690 Thursday, November
Uses of Flow Cytometry
 Uses of Flow Cytometry 1. Multicolour analysis... 2 2. Cell Cycle and Proliferation... 3 a. Analysis of Cellular DNA Content... 4 b. Cell Proliferation Assays... 5 3. Immunology... 6 4. Apoptosis... 7
Uses of Flow Cytometry 1. Multicolour analysis... 2 2. Cell Cycle and Proliferation... 3 a. Analysis of Cellular DNA Content... 4 b. Cell Proliferation Assays... 5 3. Immunology... 6 4. Apoptosis... 7
Oncology Best Practice Documentation
 Oncology Best Practice Documentation Click on the desired Diagnoses link or press Enter to view all information. Diagnoses: Solid Tumors Lymphomas Leukemias Myelodysplastic Syndrome Pathology Findings
Oncology Best Practice Documentation Click on the desired Diagnoses link or press Enter to view all information. Diagnoses: Solid Tumors Lymphomas Leukemias Myelodysplastic Syndrome Pathology Findings
SWOG ONCOLOGY RESEARCH PROFESSIONAL (ORP) MANUAL VOLUME I RESPONSE ASSESSMENT LEUKEMIA CHAPTER 11A REVISED: OCTOBER 2015
 LEUKEMIA Response in Acute Myeloid Leukemia (AML) Response criteria in Acute Myeloid Leukemia for SWOG protocols is based on the review article Diagnosis and management of acute myeloid leukemia in adults:
LEUKEMIA Response in Acute Myeloid Leukemia (AML) Response criteria in Acute Myeloid Leukemia for SWOG protocols is based on the review article Diagnosis and management of acute myeloid leukemia in adults:
Lymphoma Diagnosis and Classification
 Lymphoma Diagnosis and Classification By Atef Shrit, MD, Pathology B- and T/NK-cell lymphomas are clonal neoplasms of immature and mature B-lymphocytes, T-lymphocytes or natural killer cells at various
Lymphoma Diagnosis and Classification By Atef Shrit, MD, Pathology B- and T/NK-cell lymphomas are clonal neoplasms of immature and mature B-lymphocytes, T-lymphocytes or natural killer cells at various
Hematology Morphology Critique
 Survey Slide: History: 60-year-old female presenting with pneumonia Further Laboratory Data: Hgb : 90 g/l RBC : 2.92 10 12 /L Hct : 0.25 L/L MCV : 87 fl MCH : 30.8 pg MCHC : 355 g/l RDW : 17.7 % WBC :
Survey Slide: History: 60-year-old female presenting with pneumonia Further Laboratory Data: Hgb : 90 g/l RBC : 2.92 10 12 /L Hct : 0.25 L/L MCV : 87 fl MCH : 30.8 pg MCHC : 355 g/l RDW : 17.7 % WBC :
Introduction. About 10,500 new cases of acute myelogenous leukemia are diagnosed each
 Introduction 1.1 Introduction: About 10,500 new cases of acute myelogenous leukemia are diagnosed each year in the United States (Hope et al., 2003). Acute myelogenous leukemia has several names, including
Introduction 1.1 Introduction: About 10,500 new cases of acute myelogenous leukemia are diagnosed each year in the United States (Hope et al., 2003). Acute myelogenous leukemia has several names, including
Leukemias and Lymphomas: A primer
 Leukemias and Lymphomas: A primer Normal blood contains circulating white blood cells, red blood cells and platelets 700 red cells (oxygen) 1 white cell Neutrophils (60%) bacterial infection Lymphocytes
Leukemias and Lymphomas: A primer Normal blood contains circulating white blood cells, red blood cells and platelets 700 red cells (oxygen) 1 white cell Neutrophils (60%) bacterial infection Lymphocytes
EDUCATIONAL COMMENTARY - GRANULOCYTE FORMATION AND CHRONIC MYELOCYTIC LEUKEMIA
 LEUKEMIA Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE credits click on Earn CE Credits under Continuing Education
LEUKEMIA Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE credits click on Earn CE Credits under Continuing Education
for Leucocyte Immunophenotyping Leukaemia Diagnosis Interpretation All Participants Date Issued: 08-September-2014 Closing Date: 26-September-2014
 for Leucocyte Immunophenotyping Leukaemia Interpretation All Participants Participant: 4xxxx Trial No: 141502 Date Issued: 08-September-2014 Closing Date: 26-September-2014 Trial Comments This was an electronic
for Leucocyte Immunophenotyping Leukaemia Interpretation All Participants Participant: 4xxxx Trial No: 141502 Date Issued: 08-September-2014 Closing Date: 26-September-2014 Trial Comments This was an electronic
Diagnostic Challenge. Department of Pathology,
 Cytology of Pleural Fluid as a Diagnostic Challenge Paavo Pääkkö,, MD, PhD Chief Physician and Head of the Department Department of Pathology, Oulu University Hospital,, Finland Oulu University Hospital
Cytology of Pleural Fluid as a Diagnostic Challenge Paavo Pääkkö,, MD, PhD Chief Physician and Head of the Department Department of Pathology, Oulu University Hospital,, Finland Oulu University Hospital
WHICH SAMPLES SHOULD BE SUBMITTED WHEN LYMPHOID NEOPLASIA IS SUSPECTED?
 WHICH SAMPLES SHOULD BE SUBMITTED WHEN LYMPHOID NEOPLASIA IS SUSPECTED? Which test should be submitted? The answer to this depends on the clinical signs, and the diagnostic question you are asking. If
WHICH SAMPLES SHOULD BE SUBMITTED WHEN LYMPHOID NEOPLASIA IS SUSPECTED? Which test should be submitted? The answer to this depends on the clinical signs, and the diagnostic question you are asking. If
Exercise 9: Blood. Readings: Silverthorn 5 th ed, 547 558, 804 805; 6 th ed, 545 557, 825 826.
 Exercise 9: Blood Readings: Silverthorn 5 th ed, 547 558, 804 805; 6 th ed, 545 557, 825 826. Blood Typing The membranes of human red blood cells (RBCs) contain a variety of cell surface proteins called
Exercise 9: Blood Readings: Silverthorn 5 th ed, 547 558, 804 805; 6 th ed, 545 557, 825 826. Blood Typing The membranes of human red blood cells (RBCs) contain a variety of cell surface proteins called
chronic leukemia lymphoma myeloma differentiated 14 September 1999 Pre- Transformed Ig Surface Surface Secreted Myeloma Major malignant counterpart
 Disease Usual phenotype acute leukemia precursor chronic leukemia lymphoma myeloma differentiated Pre- B-cell B-cell Transformed B-cell Plasma cell Ig Surface Surface Secreted Major malignant counterpart
Disease Usual phenotype acute leukemia precursor chronic leukemia lymphoma myeloma differentiated Pre- B-cell B-cell Transformed B-cell Plasma cell Ig Surface Surface Secreted Major malignant counterpart
An overview of CLL care and treatment. Dr Dean Smith Haematology Consultant City Hospital Nottingham
 An overview of CLL care and treatment Dr Dean Smith Haematology Consultant City Hospital Nottingham What is CLL? CLL (Chronic Lymphocytic Leukaemia) is a type of cancer in which the bone marrow makes too
An overview of CLL care and treatment Dr Dean Smith Haematology Consultant City Hospital Nottingham What is CLL? CLL (Chronic Lymphocytic Leukaemia) is a type of cancer in which the bone marrow makes too
Hairy Cell Leukemia Facts
 No. 16 in a series providing the latest information for patients, caregivers and healthcare professionals www.lls.org Information Specialist: 800.955.4572 Highlights l Hairy cell leukemia (HCL) is a chronic
No. 16 in a series providing the latest information for patients, caregivers and healthcare professionals www.lls.org Information Specialist: 800.955.4572 Highlights l Hairy cell leukemia (HCL) is a chronic
Supplementary appendix
 Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: Farooqui MZH, Valdez J, Martyr S, et al. Ibrutinib
Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: Farooqui MZH, Valdez J, Martyr S, et al. Ibrutinib
White Blood Cells (WBCs) or Leukocytes
 Lec.5 Z.H.Al-Zubaydi Medical Physiology White Blood Cells (WBCs) or Leukocytes Although leukocytes are far less numerous than red blood cells, they are important to body defense against disease. On average,
Lec.5 Z.H.Al-Zubaydi Medical Physiology White Blood Cells (WBCs) or Leukocytes Although leukocytes are far less numerous than red blood cells, they are important to body defense against disease. On average,
Aggressive lymphomas. Michael Crump Princess Margaret Hospital
 Aggressive lymphomas Michael Crump Princess Margaret Hospital What are the aggressive lymphomas? Diffuse large B cell Mediastinal large B cell Anaplastic large cell Burkitt lymphoma (transformed lymphoma:
Aggressive lymphomas Michael Crump Princess Margaret Hospital What are the aggressive lymphomas? Diffuse large B cell Mediastinal large B cell Anaplastic large cell Burkitt lymphoma (transformed lymphoma:
specific B cells Humoral immunity lymphocytes antibodies B cells bone marrow Cell-mediated immunity: T cells antibodies proteins
 Adaptive Immunity Chapter 17: Adaptive (specific) Immunity Bio 139 Dr. Amy Rogers Host defenses that are specific to a particular infectious agent Can be innate or genetic for humans as a group: most microbes
Adaptive Immunity Chapter 17: Adaptive (specific) Immunity Bio 139 Dr. Amy Rogers Host defenses that are specific to a particular infectious agent Can be innate or genetic for humans as a group: most microbes
ACUTE MYELOID LEUKEMIA (AML),
 1 ACUTE MYELOID LEUKEMIA (AML), ALSO KNOWN AS ACUTE MYELOGENOUS LEUKEMIA WHAT IS CANCER? The body is made up of hundreds of millions of living cells. Normal body cells grow, divide, and die in an orderly
1 ACUTE MYELOID LEUKEMIA (AML), ALSO KNOWN AS ACUTE MYELOGENOUS LEUKEMIA WHAT IS CANCER? The body is made up of hundreds of millions of living cells. Normal body cells grow, divide, and die in an orderly
MALIGNANT LYMPHOMAS. Dr. Olga Vujovic (Updated August 2010)
 MALIGNANT LYMPHOMAS Dr. Olga Vujovic (Updated August 2010) Malignant lymphomas consist of Hodgkin and non-hodgkin lymphomas. The current management of these diseases involves a multi-disciplinary approach.
MALIGNANT LYMPHOMAS Dr. Olga Vujovic (Updated August 2010) Malignant lymphomas consist of Hodgkin and non-hodgkin lymphomas. The current management of these diseases involves a multi-disciplinary approach.
Immunophenotyping in the differential diagnosis of histologically low grade B cell lymphomas
 Current Diagnostic Pathology (2000) 6, 55 63 2000 Harcourt Publishers Ltd doi:10.1054/cdip.2000.0015, available online at http://www.idealibrary.com on REVIEW Immunophenotyping in the differential diagnosis
Current Diagnostic Pathology (2000) 6, 55 63 2000 Harcourt Publishers Ltd doi:10.1054/cdip.2000.0015, available online at http://www.idealibrary.com on REVIEW Immunophenotyping in the differential diagnosis
Collaboration to collect Autologous transplant outcomes in Lymphoma and Myeloma (CALM) Additional Questionnaire (MED C) INCLUSION CRITERIA CALM STUDY
 Additional Questionnaire (MED C) CALM study Inclusion period: 01/01/2008 to 31/12/2011 PATIENT REGISTRATION FORM Disease Diagnosis Lymphoma S Non Hodgkin Lymphoma (NHL) Mature B-cell neoplasm Follicular
Additional Questionnaire (MED C) CALM study Inclusion period: 01/01/2008 to 31/12/2011 PATIENT REGISTRATION FORM Disease Diagnosis Lymphoma S Non Hodgkin Lymphoma (NHL) Mature B-cell neoplasm Follicular
Adult Medical-Surgical Nursing H A E M A T O L O G Y M O D U L E : L E U K A E M I A 2
 Adult Medical-Surgical Nursing H A E M A T O L O G Y M O D U L E : L E U K A E M I A 2 Leukaemia: Description A group of malignant disorders affecting: White blood cells (lymphocytes or leucocytes) Bone
Adult Medical-Surgical Nursing H A E M A T O L O G Y M O D U L E : L E U K A E M I A 2 Leukaemia: Description A group of malignant disorders affecting: White blood cells (lymphocytes or leucocytes) Bone
DELPHI 27 V 2016 CYTOMETRY STRATEGIES IN THE DIAGNOSIS OF HEMATOLOGICAL DISEASES
 DELPHI 27 V 2016 CYTOMETRY STRATEGIES IN THE DIAGNOSIS OF HEMATOLOGICAL DISEASES CLAUDIO ORTOLANI UNIVERSITY OF URBINO - ITALY SUN TZU (544 b.c. 496 b.c) SUN TZU (544 b.c. 496 b.c.) THE ART OF CYTOMETRY
DELPHI 27 V 2016 CYTOMETRY STRATEGIES IN THE DIAGNOSIS OF HEMATOLOGICAL DISEASES CLAUDIO ORTOLANI UNIVERSITY OF URBINO - ITALY SUN TZU (544 b.c. 496 b.c) SUN TZU (544 b.c. 496 b.c.) THE ART OF CYTOMETRY
Why discuss CLL? Common: 40% of US leukaemia. approx 100 pa in SJH / MWHB 3 inpatients in SJH at any time
 Why discuss CLL? Common: 40% of US leukaemia approx 100 pa in SJH / MWHB 3 inpatients in SJH at any time Median age of dx is 65 (30s. Incurable, survival 2-202 20 years Require ongoing supportive care
Why discuss CLL? Common: 40% of US leukaemia approx 100 pa in SJH / MWHB 3 inpatients in SJH at any time Median age of dx is 65 (30s. Incurable, survival 2-202 20 years Require ongoing supportive care
Estimated New Cases of Leukemia, Lymphoma, Myeloma 2014
 ABOUT BLOOD CANCERS Leukemia, Hodgkin lymphoma (HL), non-hodgkin lymphoma (NHL), myeloma, myelodysplastic syndromes (MDS) and myeloproliferative neoplasms (MPNs) are types of cancer that can affect the
ABOUT BLOOD CANCERS Leukemia, Hodgkin lymphoma (HL), non-hodgkin lymphoma (NHL), myeloma, myelodysplastic syndromes (MDS) and myeloproliferative neoplasms (MPNs) are types of cancer that can affect the
Stem Cell Transplantation
 Harmony Behavioral Health, Inc. Harmony Behavioral Health of Florida, Inc. Harmony Health Plan of Illinois, Inc. HealthEase of Florida, Inc. Ohana Health Plan, a plan offered by WellCare Health Insurance
Harmony Behavioral Health, Inc. Harmony Behavioral Health of Florida, Inc. Harmony Health Plan of Illinois, Inc. HealthEase of Florida, Inc. Ohana Health Plan, a plan offered by WellCare Health Insurance
Non-Hodgkin s Lymphoma
 Non-Hodgkin s Lymphoma Luis Fayad, MD Assistant Professor Clinical Medical Director Lymphoma/Myeloma Department Non-Hodgkin s Lymphoma Non-Hodgkin s lymphomas (NHL) are a heterogeneous group of malignant
Non-Hodgkin s Lymphoma Luis Fayad, MD Assistant Professor Clinical Medical Director Lymphoma/Myeloma Department Non-Hodgkin s Lymphoma Non-Hodgkin s lymphomas (NHL) are a heterogeneous group of malignant
The Diagnosis of Cancer in the Pathology Laboratory
 The Diagnosis of Cancer in the Pathology Laboratory Dr Edward Sheffield Christmas Select 74 Meeting, Queen s Hotel Cheltenham, 3 rd December 2014 Agenda Overview of the pathology of cancer How specimens
The Diagnosis of Cancer in the Pathology Laboratory Dr Edward Sheffield Christmas Select 74 Meeting, Queen s Hotel Cheltenham, 3 rd December 2014 Agenda Overview of the pathology of cancer How specimens
Outline of lecture. Disclosures. Current Issues in Practical Hematopathology: Diagnosis of Bone Marrow Lymphomas
 Current Issues in Practical Hematopathology: Diagnosis of Bone Marrow Lymphomas Disclosures I have no disclosures relevant to the content of this lecture Robert P Hasserjian, MD Associate Professor Massachusetts
Current Issues in Practical Hematopathology: Diagnosis of Bone Marrow Lymphomas Disclosures I have no disclosures relevant to the content of this lecture Robert P Hasserjian, MD Associate Professor Massachusetts
A 32 year old woman comes to your clinic with neck masses for the last several weeks. Masses are discrete, non matted, firm and rubbery on
 A 32 year old woman comes to your clinic with neck masses for the last several weeks. Masses are discrete, non matted, firm and rubbery on examination. She also has fever, weight loss, and sweats. What
A 32 year old woman comes to your clinic with neck masses for the last several weeks. Masses are discrete, non matted, firm and rubbery on examination. She also has fever, weight loss, and sweats. What
Two Retroperitoneal Low-Grade B-Cell Lymphoma Successfully Treated With a Combination of Chimeric Anti-CD20 Monoclonal Antibody and CHOP Chemotherapy
 Two Retroperitoneal Low-Grade B-Cell Lymphoma Successfully Treated With a Combination of Chimeric Anti-CD20 Monoclonal Antibody and CHOP Chemotherapy Yoichi Kitamura, MD Kazuhiko Hayashi, MD Kazumi Uchida,
Two Retroperitoneal Low-Grade B-Cell Lymphoma Successfully Treated With a Combination of Chimeric Anti-CD20 Monoclonal Antibody and CHOP Chemotherapy Yoichi Kitamura, MD Kazuhiko Hayashi, MD Kazumi Uchida,
Acute myeloid leukemia (AML)
 Acute myeloid leukemia (AML) Adult acute myeloid leukemia (AML) is a type of cancer in which the bone marrow makes abnormal myeloblasts (a type of white blood cell), red blood cells, or platelets. Adult
Acute myeloid leukemia (AML) Adult acute myeloid leukemia (AML) is a type of cancer in which the bone marrow makes abnormal myeloblasts (a type of white blood cell), red blood cells, or platelets. Adult
Pulling the Plug on Cancer Cell Communication. Stephen M. Ansell, MD, PhD Mayo Clinic
 Pulling the Plug on Cancer Cell Communication Stephen M. Ansell, MD, PhD Mayo Clinic Why do Waldenstrom s cells need to communicate? Waldenstrom s cells need activating signals to stay alive. WM cells
Pulling the Plug on Cancer Cell Communication Stephen M. Ansell, MD, PhD Mayo Clinic Why do Waldenstrom s cells need to communicate? Waldenstrom s cells need activating signals to stay alive. WM cells
Utility of flow cytometric κ and λ light chain analysis of peripheral blood
 JBUON 2015; 20(5): 1322-1326 ISSN: 1107-0625, online ISSN: 2241-6293 www.jbuon.com E-mail: [email protected] ORIGINAL ARTICLE Utility of flow cytometric κ and λ light chain analysis of peripheral
JBUON 2015; 20(5): 1322-1326 ISSN: 1107-0625, online ISSN: 2241-6293 www.jbuon.com E-mail: [email protected] ORIGINAL ARTICLE Utility of flow cytometric κ and λ light chain analysis of peripheral
Granulocytes vs. Agranulocytes
 Leukocytes are white blood cells (AKA colorless (non-pigmented) blood cells). (Much) smaller in number than RBCs. Unlike RBCs, there are several different types of WBCs. All contain a visible nucleus.
Leukocytes are white blood cells (AKA colorless (non-pigmented) blood cells). (Much) smaller in number than RBCs. Unlike RBCs, there are several different types of WBCs. All contain a visible nucleus.
PERIPHERAL SMEARS: THE PRIMARY DIAGNOSTIC TOOL PART II IRMA PEREIRA, MT(ASCP)SH
 PERIPHERAL SMEARS: THE PRIMARY DIAGNOSTIC TOOL PART II IRMA PEREIRA, MT(ASCP)SH WORKSHOPS FOR LABORATORY PROFESSIONALS As a service to the medical profession and to the general public, the American Society
PERIPHERAL SMEARS: THE PRIMARY DIAGNOSTIC TOOL PART II IRMA PEREIRA, MT(ASCP)SH WORKSHOPS FOR LABORATORY PROFESSIONALS As a service to the medical profession and to the general public, the American Society
Corporate Medical Policy
 Corporate Medical Policy Hematopoietic Stem-Cell Transplantation for CLL and SLL File Name: Origination: Last CAP Review: Next CAP Review: Last Review: hematopoietic_stem-cell_transplantation_for_cll_and_sll
Corporate Medical Policy Hematopoietic Stem-Cell Transplantation for CLL and SLL File Name: Origination: Last CAP Review: Next CAP Review: Last Review: hematopoietic_stem-cell_transplantation_for_cll_and_sll
Activation and effector functions of HMI
 Activation and effector functions of HMI Hathairat Thananchai, DPhil Department of Microbiology Faculty of Medicine Chiang Mai University 25 August 2015 ว ตถ ประสงค หล งจากช วโมงบรรยายน แล วน กศ กษาสามารถ
Activation and effector functions of HMI Hathairat Thananchai, DPhil Department of Microbiology Faculty of Medicine Chiang Mai University 25 August 2015 ว ตถ ประสงค หล งจากช วโมงบรรยายน แล วน กศ กษาสามารถ
Tumour Markers. What are Tumour Markers? How Are Tumour Markers Used?
 Dr. Anthony C.H. YING What are? Tumour markers are substances that can be found in the body when cancer is present. They are usually found in the blood or urine. They can be products of cancer cells or
Dr. Anthony C.H. YING What are? Tumour markers are substances that can be found in the body when cancer is present. They are usually found in the blood or urine. They can be products of cancer cells or
亞 東 紀 念 醫 院 Follicular Lymphoma 臨 床 指 引
 前 言 : 惡 性 淋 巴 瘤 ( 或 簡 稱 淋 巴 癌 ) 乃 由 體 內 淋 巴 系 統 包 括 淋 巴 細 胞 淋 巴 管 淋 巴 腺 及 一 些 淋 巴 器 官 或 組 織 如 脾 臟 胸 腺 及 扁 桃 腺 等 所 長 出 的 惡 性 腫 瘤 依 腫 瘤 病 理 組 織 型 態 的 不 同 可 分 為 何 杰 金 氏 淋 巴 瘤 (Hodgkin s disease) 與 非 何 杰 金
前 言 : 惡 性 淋 巴 瘤 ( 或 簡 稱 淋 巴 癌 ) 乃 由 體 內 淋 巴 系 統 包 括 淋 巴 細 胞 淋 巴 管 淋 巴 腺 及 一 些 淋 巴 器 官 或 組 織 如 脾 臟 胸 腺 及 扁 桃 腺 等 所 長 出 的 惡 性 腫 瘤 依 腫 瘤 病 理 組 織 型 態 的 不 同 可 分 為 何 杰 金 氏 淋 巴 瘤 (Hodgkin s disease) 與 非 何 杰 金
TUBERCULOSIS PLEURAL EFFUSION - MANAGEMENT
 TUBERCULOSIS PLEURAL EFFUSION - MANAGEMENT Introduction : ETB 15-20% Pleural effusion 20% in non HIV Under reporting because of AFB negative in fluid In HIV patients: EPTB 20% PTB + EPTB 50% Pleural Effusion
TUBERCULOSIS PLEURAL EFFUSION - MANAGEMENT Introduction : ETB 15-20% Pleural effusion 20% in non HIV Under reporting because of AFB negative in fluid In HIV patients: EPTB 20% PTB + EPTB 50% Pleural Effusion
Platelet Transmission Electron Microscopy and Flow Cytometry 11/15/2015
 Welcome to Mayo Medical Laboratories Hot Topics. These presentations provide short discussion of current topics and may be helpful to you in your practice. Today our topic looks at hereditary platelet
Welcome to Mayo Medical Laboratories Hot Topics. These presentations provide short discussion of current topics and may be helpful to you in your practice. Today our topic looks at hereditary platelet
HOVON Staging and Response Criteria for Non-Hodgkin s Lymphomas Page 1
 HOVON Staging and Response Criteria for Non-Hodgkin s Lymphomas Page 1 This document describes the minimally required staging and evaluation procedures and response criteria that will be applied in all
HOVON Staging and Response Criteria for Non-Hodgkin s Lymphomas Page 1 This document describes the minimally required staging and evaluation procedures and response criteria that will be applied in all
2011 Update on the ECIL-3 guidelines for EBV management in patients with leukemia and other hematological disorders
 UPDATE ECIL-4 2011 2011 Update on the ECIL-3 guidelines for EBV management in patients with leukemia and other hematological disorders Jan Styczynski, Hermann Einsele, Rafael de la Camara, Catherine Cordonnier,
UPDATE ECIL-4 2011 2011 Update on the ECIL-3 guidelines for EBV management in patients with leukemia and other hematological disorders Jan Styczynski, Hermann Einsele, Rafael de la Camara, Catherine Cordonnier,
Cytology of Lymph Nodes
 Indications Cytology of Lymph Nodes Lymph node enlargement That was easy Mary Anna Thrall Don Meuten Indications Lymph node enlargement Suspect metastasis Normal sized lymph nodes are Normal Do NOT aspirate
Indications Cytology of Lymph Nodes Lymph node enlargement That was easy Mary Anna Thrall Don Meuten Indications Lymph node enlargement Suspect metastasis Normal sized lymph nodes are Normal Do NOT aspirate
Multiple Myeloma Workshop- Tandem 2014
 Multiple Myeloma Workshop- Tandem 2014 1) Review of Plasma Cell Disorders Asymptomatic (smoldering) myeloma M-protein in serum at myeloma levels (>3g/dL); and/or 10% or more clonal plasma cells in bone
Multiple Myeloma Workshop- Tandem 2014 1) Review of Plasma Cell Disorders Asymptomatic (smoldering) myeloma M-protein in serum at myeloma levels (>3g/dL); and/or 10% or more clonal plasma cells in bone
6/20/2014. PART I: Plasma Cell Myeloma. Plasma Cells
 MULTIPLE MYELOMA: THE TESTING, VALIDATION AND IMPLEMENTATION OF CELL SEPARATION TECHNOLOGY FOR IMPROVED PATIENT CARE Elizabeth Harper CG(ASCP), Binh Vo CG(ASCP), Joey Pena CG(ASCP), Denise Lovshe CG(ASCP),
MULTIPLE MYELOMA: THE TESTING, VALIDATION AND IMPLEMENTATION OF CELL SEPARATION TECHNOLOGY FOR IMPROVED PATIENT CARE Elizabeth Harper CG(ASCP), Binh Vo CG(ASCP), Joey Pena CG(ASCP), Denise Lovshe CG(ASCP),
The Immune System: A Tutorial
 The Immune System: A Tutorial Modeling and Simulation of Biological Systems 21-366B Shlomo Ta asan Images taken from http://rex.nci.nih.gov/behindthenews/uis/uisframe.htm http://copewithcytokines.de/ The
The Immune System: A Tutorial Modeling and Simulation of Biological Systems 21-366B Shlomo Ta asan Images taken from http://rex.nci.nih.gov/behindthenews/uis/uisframe.htm http://copewithcytokines.de/ The
LYMPHOMA IN DOGS. Diagnosis/Initial evaluation. Treatment and Prognosis
 LYMPHOMA IN DOGS Lymphoma is a relatively common cancer in dogs. It is a cancer of lymphocytes (a type of white blood cell) and lymphoid tissues. Lymphoid tissue is normally present in many places in the
LYMPHOMA IN DOGS Lymphoma is a relatively common cancer in dogs. It is a cancer of lymphocytes (a type of white blood cell) and lymphoid tissues. Lymphoid tissue is normally present in many places in the
LEUKEMIA LYMPHOMA MYELOMA Advances in Clinical Trials
 LEUKEMIA LYMPHOMA MYELOMA Advances in Clinical Trials OUR FOCUS ABOUT emerging treatments Presentation for: Judith E. Karp, MD Advancements for Acute Myelogenous Leukemia Supported by an unrestricted educational
LEUKEMIA LYMPHOMA MYELOMA Advances in Clinical Trials OUR FOCUS ABOUT emerging treatments Presentation for: Judith E. Karp, MD Advancements for Acute Myelogenous Leukemia Supported by an unrestricted educational
Lauren Berger: Why is it so important for patients to get an accurate diagnosis of their blood cancer subtype?
 Hello, I m Lauren Berger and I m the Senior Director of Patient Services Programs at The Leukemia & Lymphoma Society. I m pleased to welcome Dr. Rebecca Elstrom. Dr. Elstrom is an Assistant Professor in
Hello, I m Lauren Berger and I m the Senior Director of Patient Services Programs at The Leukemia & Lymphoma Society. I m pleased to welcome Dr. Rebecca Elstrom. Dr. Elstrom is an Assistant Professor in
Minimal residual disease detection in Acute Myeloid Leukaemia on a Becton Dickinson flow cytometer
 Minimal residual disease detection in Acute Myeloid Leukaemia on a Becton Dickinson flow cytometer Purpose This procedure gives instruction on minimal residual disease (MRD) detection in patients with
Minimal residual disease detection in Acute Myeloid Leukaemia on a Becton Dickinson flow cytometer Purpose This procedure gives instruction on minimal residual disease (MRD) detection in patients with
Recognition of T cell epitopes (Abbas Chapter 6)
 Recognition of T cell epitopes (Abbas Chapter 6) Functions of different APCs (Abbas Chapter 6)!!! Directon Routes of antigen entry (Abbas Chapter 6) Flow of Information Barrier APCs LNs Sequence of Events
Recognition of T cell epitopes (Abbas Chapter 6) Functions of different APCs (Abbas Chapter 6)!!! Directon Routes of antigen entry (Abbas Chapter 6) Flow of Information Barrier APCs LNs Sequence of Events
Cytology : first alert of mesothelioma? Professor B. Weynand, UCL Yvoir, Belgium
 Cytology : first alert of mesothelioma? Professor B. Weynand, UCL Yvoir, Belgium Introduction 3 cavities with the same embryologic origin the mesoderme Pleura Exudates Pleura Peritoneum Pericardium 22%
Cytology : first alert of mesothelioma? Professor B. Weynand, UCL Yvoir, Belgium Introduction 3 cavities with the same embryologic origin the mesoderme Pleura Exudates Pleura Peritoneum Pericardium 22%
UNDERSTANDING MULTIPLE MYELOMA AND LABORATORY VALUES Benjamin Parsons, DO [email protected] Gundersen Health System Center for Cancer and
 UNDERSTANDING MULTIPLE MYELOMA AND LABORATORY VALUES Benjamin Parsons, DO [email protected] Gundersen Health System Center for Cancer and Blood Disorders La Crosse, WI UNDERSTANDING MULTIPLE
UNDERSTANDING MULTIPLE MYELOMA AND LABORATORY VALUES Benjamin Parsons, DO [email protected] Gundersen Health System Center for Cancer and Blood Disorders La Crosse, WI UNDERSTANDING MULTIPLE
Lymphoplasmacytic Lymphoma. Hematology fellows conference 4/12/2013 Christina Fitzmaurice, MD, MPH
 Lymphoplasmacytic Lymphoma versus IGM Multiple Myeloma Hematology fellows conference 4/12/2013 Christina Fitzmaurice, MD, MPH Hematology consult patient 48 yo woman presents to ER with nonspecific complaints:
Lymphoplasmacytic Lymphoma versus IGM Multiple Myeloma Hematology fellows conference 4/12/2013 Christina Fitzmaurice, MD, MPH Hematology consult patient 48 yo woman presents to ER with nonspecific complaints:
Response Criteria for Malignant Lymphoma 2007. Cheson Criteria. Quick Reference Guide
 Response Criteria for Malignant Lymphoma 2007 Cheson Criteria Quick Reference Guide Table of Contents Summary of Assessments...3 Baseline Lesion Burden...4 What isameasurable Lesion?...5 Choosing Target
Response Criteria for Malignant Lymphoma 2007 Cheson Criteria Quick Reference Guide Table of Contents Summary of Assessments...3 Baseline Lesion Burden...4 What isameasurable Lesion?...5 Choosing Target
CHAPTER 2. Neoplasms (C00-D49) March 2014. 2014 MVP Health Care, Inc.
 Neoplasms (C00-D49) March 2014 2014 MVP Health Care, Inc. CHAPTER SPECIFIC CATEGORY CODE BLOCKS C00-C14 Malignant neoplasms of lip, oral cavity and pharynx C15-C26 Malignant neoplasms of digestive organs
Neoplasms (C00-D49) March 2014 2014 MVP Health Care, Inc. CHAPTER SPECIFIC CATEGORY CODE BLOCKS C00-C14 Malignant neoplasms of lip, oral cavity and pharynx C15-C26 Malignant neoplasms of digestive organs
Immunohistochemical classification of malignant lymphomas
 Immunohistochemical classification of malignant lymphomas Prof. Mogens Vyberg NordiQC Institute of Pathology Aalborg, Denmark Thanks to Jan Klos Important issues Distinction from non-hematopoietic neoplasms
Immunohistochemical classification of malignant lymphomas Prof. Mogens Vyberg NordiQC Institute of Pathology Aalborg, Denmark Thanks to Jan Klos Important issues Distinction from non-hematopoietic neoplasms
Hodgkin Lymphoma Disease Specific Biology and Treatment Options. John Kuruvilla
 Hodgkin Lymphoma Disease Specific Biology and Treatment Options John Kuruvilla My Disclaimer This is where I work Objectives Pathobiology what makes HL different Diagnosis Staging Treatment Philosophy
Hodgkin Lymphoma Disease Specific Biology and Treatment Options John Kuruvilla My Disclaimer This is where I work Objectives Pathobiology what makes HL different Diagnosis Staging Treatment Philosophy
Explanation of your PAP smear
 Explanation of your PAP smear Approximately 5-10% of PAP smears in the United States are judged to be abnormal. Too often, the woman who receives this news worries that she already has, or will develop,
Explanation of your PAP smear Approximately 5-10% of PAP smears in the United States are judged to be abnormal. Too often, the woman who receives this news worries that she already has, or will develop,
MRI of Bone Marrow Radiologic-Pathologic Correlation
 MRI of Bone Marrow Radiologic-Pathologic Correlation Marilyn J. Siegel, M.D. Mallinckrodt Institute of Radiology Washington University School of Medicine St. Louis, MO and Visiting Scientist, AFIP, Washington,
MRI of Bone Marrow Radiologic-Pathologic Correlation Marilyn J. Siegel, M.D. Mallinckrodt Institute of Radiology Washington University School of Medicine St. Louis, MO and Visiting Scientist, AFIP, Washington,
Polyps. Hyperplasias. CAP 2011: Course AP104. The High Risk Benign Endometrium. Mutter and Nucci 1
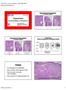 Course AP104 Endometrial Hyperplasia A morphologic Definition Hyperplasias Hormonal Effect or Precancer? George L. Mutter, MD Harvard Medical School and Brigham and Women s Hospital Boston, MA Endometrial
Course AP104 Endometrial Hyperplasia A morphologic Definition Hyperplasias Hormonal Effect or Precancer? George L. Mutter, MD Harvard Medical School and Brigham and Women s Hospital Boston, MA Endometrial
Understanding Metastatic Disease
 Supported by an unrestricted educational grant from Pfizer Understanding Metastatic Disease Metastatic disease or metastases are phrases that mean the same as Secondary cancer. This means that the cancer
Supported by an unrestricted educational grant from Pfizer Understanding Metastatic Disease Metastatic disease or metastases are phrases that mean the same as Secondary cancer. This means that the cancer
Best Practice in Lymphoma Diagnosis and Reporting
 Best Practice in Lymphoma Diagnosis and Reporting British Committee for Standards in Haematology Royal College of Pathologists Address for correspondence: BCSH Administrator British Society for Haematology
Best Practice in Lymphoma Diagnosis and Reporting British Committee for Standards in Haematology Royal College of Pathologists Address for correspondence: BCSH Administrator British Society for Haematology
Carcinosarcoma of the Ovary
 Carcinosarcoma of the Ovary A Rare Finding Presented By: Kathryn Kiely Anisa I. Kanbour School of Cytotechnology of the University of Pittsburgh Medical Center Pittsburgh, PA Patient History 55 year old
Carcinosarcoma of the Ovary A Rare Finding Presented By: Kathryn Kiely Anisa I. Kanbour School of Cytotechnology of the University of Pittsburgh Medical Center Pittsburgh, PA Patient History 55 year old
Immuno-Oncology Therapies to Treat Lung Cancer
 Immuno-Oncology Therapies to Treat Lung Cancer What you need to know ONCHQ14NP07519 Introduction: Immuno-oncology represents an innovative approach to cancer research that seeks to harness the body s own
Immuno-Oncology Therapies to Treat Lung Cancer What you need to know ONCHQ14NP07519 Introduction: Immuno-oncology represents an innovative approach to cancer research that seeks to harness the body s own
Practical Effusion Cytology
 Practical Effusion Cytology A Community Pathologist s Approach to Immunocytochemistry in Body Fluid Cytology Emily E. Volk, MD William Beaumont Hospital Troy, MI College of American Pathologists 2004.
Practical Effusion Cytology A Community Pathologist s Approach to Immunocytochemistry in Body Fluid Cytology Emily E. Volk, MD William Beaumont Hospital Troy, MI College of American Pathologists 2004.
treatments) worked by killing cancerous cells using chemo or radiotherapy. While these techniques can
 Shristi Pandey Genomics and Medicine Winter 2011 Prof. Doug Brutlag Chronic Myeloid Leukemia: A look into how genomics is changing the way we treat Cancer. Until the late 1990s, nearly all treatment methods
Shristi Pandey Genomics and Medicine Winter 2011 Prof. Doug Brutlag Chronic Myeloid Leukemia: A look into how genomics is changing the way we treat Cancer. Until the late 1990s, nearly all treatment methods
Emerging New Prognostic Scoring Systems in Myelodysplastic Syndromes 2012
 Emerging New Prognostic Scoring Systems in Myelodysplastic Syndromes 2012 Arjan A. van de Loosdrecht, MD, PhD Department of Hematology VU University Medical Center VU-Institute of Cancer and Immunology
Emerging New Prognostic Scoring Systems in Myelodysplastic Syndromes 2012 Arjan A. van de Loosdrecht, MD, PhD Department of Hematology VU University Medical Center VU-Institute of Cancer and Immunology
Things You Don t Want to Miss in Multiple Myeloma
 Things You Don t Want to Miss in Multiple Myeloma Sreenivasa Chandana, MD, PhD Attending Hematologist and Medical Oncologist West Michigan Cancer Center Assistant Professor, Western Michigan University
Things You Don t Want to Miss in Multiple Myeloma Sreenivasa Chandana, MD, PhD Attending Hematologist and Medical Oncologist West Michigan Cancer Center Assistant Professor, Western Michigan University
Report series: General cancer information
 Fighting cancer with information Report series: General cancer information Eastern Cancer Registration and Information Centre ECRIC report series: General cancer information Cancer is a general term for
Fighting cancer with information Report series: General cancer information Eastern Cancer Registration and Information Centre ECRIC report series: General cancer information Cancer is a general term for
Interesting Case Series. Periorbital Richter Syndrome
 Interesting Case Series Periorbital Richter Syndrome MarkGorman,MRCS,MSc, a Julia Ruston, MRCS, b and Sarath Vennam, BMBS a a Division of Plastic Surgery, Royal Devon and Exeter Hospital, Exeter, Devon,
Interesting Case Series Periorbital Richter Syndrome MarkGorman,MRCS,MSc, a Julia Ruston, MRCS, b and Sarath Vennam, BMBS a a Division of Plastic Surgery, Royal Devon and Exeter Hospital, Exeter, Devon,
Unit 1 Higher Human Biology Summary Notes
 Unit 1 Higher Human Biology Summary Notes a. Cells tissues organs body systems Division of labour occurs in multicellular organisms (rather than each cell carrying out every function) Most cells become
Unit 1 Higher Human Biology Summary Notes a. Cells tissues organs body systems Division of labour occurs in multicellular organisms (rather than each cell carrying out every function) Most cells become
The Treatment of Leukemia
 The Treatment of Leukemia Guest Expert: Peter, MD Associate Professor of Hematology Director, Yale Cancer Center Leukemia Program www.wnpr.org www.yalecancercenter.org Hi, I am Bruce Barber and welcome
The Treatment of Leukemia Guest Expert: Peter, MD Associate Professor of Hematology Director, Yale Cancer Center Leukemia Program www.wnpr.org www.yalecancercenter.org Hi, I am Bruce Barber and welcome
Outline. Workup for metastatic breast cancer. Metastatic breast cancer
 Metastatic breast cancer Immunostain Update: Diagnosis of metastatic breast carcinoma, emphasizing distinction from GYN primary 1/3 of breast cancer patients will show metastasis 1 st presentation or 20-30
Metastatic breast cancer Immunostain Update: Diagnosis of metastatic breast carcinoma, emphasizing distinction from GYN primary 1/3 of breast cancer patients will show metastasis 1 st presentation or 20-30
http://www.springer.com/3-540-22006-2
 http://www.springer.com/3-540-22006-2 1 Molecular Pathology Laboratory of the Future Christopher A. Moskaluk 1.1 The Past The integration of laboratory analysis with human medicine has traditionally been
http://www.springer.com/3-540-22006-2 1 Molecular Pathology Laboratory of the Future Christopher A. Moskaluk 1.1 The Past The integration of laboratory analysis with human medicine has traditionally been
Standardization, Calibration and Quality Control
 Standardization, Calibration and Quality Control Ian Storie Flow cytometry has become an essential tool in the research and clinical diagnostic laboratory. The range of available flow-based diagnostic
Standardization, Calibration and Quality Control Ian Storie Flow cytometry has become an essential tool in the research and clinical diagnostic laboratory. The range of available flow-based diagnostic
Chronic lymphocytic EBMT Slideleukemia. University of Heidelberg, Germany March 22, 2010. The European Group for Blood and Marrow Transplantation
 Chronic lymphocytic EBMT Slideleukemia template Peter Barcelona Dreger Chairman, CLL 7 February subcommittee 2008 University of Heidelberg, Germany March 22, 2010 The European Group for Blood and Marrow
Chronic lymphocytic EBMT Slideleukemia template Peter Barcelona Dreger Chairman, CLL 7 February subcommittee 2008 University of Heidelberg, Germany March 22, 2010 The European Group for Blood and Marrow
Lymphoma in Dogs: Diagnosis & Treatment
 c o n s u l t a n t o n c a l l O N C O L O G Y David M.Vail, DVM, Diplomate ACVIM (Oncology), University of Wisconsin Madison Lymphoma in Dogs: Diagnosis & Treatment Profile Definitions Lymphoma can be
c o n s u l t a n t o n c a l l O N C O L O G Y David M.Vail, DVM, Diplomate ACVIM (Oncology), University of Wisconsin Madison Lymphoma in Dogs: Diagnosis & Treatment Profile Definitions Lymphoma can be
Immunohistochemical differentiation of metastatic tumours
 Immunohistochemical differentiation of metastatic tumours Dr Abi Wheal ST1. TERA 3/2/14 Key points from a review article written by Daisuke Nonaka Intro Metastatic disease is the initial presentation in
Immunohistochemical differentiation of metastatic tumours Dr Abi Wheal ST1. TERA 3/2/14 Key points from a review article written by Daisuke Nonaka Intro Metastatic disease is the initial presentation in
Update in Hematology Oncology Targeted Therapies. Mark Holguin
 Update in Hematology Oncology Targeted Therapies Mark Holguin 25 years ago Why I chose oncology People How to help people with possibly the most difficult thing they may have to deal with Science Turning
Update in Hematology Oncology Targeted Therapies Mark Holguin 25 years ago Why I chose oncology People How to help people with possibly the most difficult thing they may have to deal with Science Turning
