Application of Assisted Reproductive Technologies (ART) to Clinical Practice
|
|
|
- Brook Hall
- 10 years ago
- Views:
Transcription
1 Application of Assisted Reproductive Technologies (ART) to Clinical Practice Katrin Hinrichs, DVM, PhD, Diplomate ACT Author s address: Departments of Large Animal Clinical Sciences and Veterinary Physiology and Pharmacology, College of Veterinary Medicine and Biomedical Sciences, Texas A&M University, College Station, Texas ; [email protected] AAEP. 1. Introduction In the past decade, assisted reproductive technologies (ART) such as oocyte transfer, intracytoplasmic sperm injection (ICSI), and nuclear transfer (cloning) have become effective enough for clinical use. ART such as oocyte transfer and ICSI allow production of foals from mares that cannot provide an embryo by standard embryo-transfer procedures. For the equine practitioner, application of ART can range from simply sending ovaries to a laboratory after a mare s untimely death to setting up complete oocyte and embryo culture systems in a practice. This paper outlines some of the increasingly common ways in which ART may be applied to equine clinical practice, including ongoing developments in embryo biopsy for pre-implantation genetic diagnosis and embryo vitrification. 2. Shipment of Ovaries Postmortem It is possible to recover oocytes from ovaries up to 24 h postmortem and produce foals from those oocytes by either ICSI or oocyte transfer. Although little critical information exists in this area, data suggest that oocytes recovered less than 6 8 h after the mare s death are more likely to produce embryos, and the embryos that are produced yield pregnancies that are more likely to be carried to term. 1,2 When ovaries arrive at the laboratory, oocytes are recovered from all visible follicles. Because these oocytes are from follicles that were not ready to ovulate, the oocytes are immature; they are placed in culture for h to mature in vitro. After culture, about 50% of oocytes are expected to mature, and these can be used for (1) oocyte transfer (surgical transfer to the oviduct of an inseminated recipient mare), (2) ICSI and then surgical oviductal transfer of cleaved embryos 24 h after ICSI, or (3) ICSI and in vitro culture to the blastocyst stage followed by transcervical transfer to a recipient mare (either immediately or after vitrification or freezing 3 ). If multiple oocytes are recovered, the third procedure (ICSI and culture of oocytes to the blastocyst stage) would seem to be the treatment of choice, because this will allow transfer of each developing embryo to a separate recipient mare. In contrast, when surgical transfers are performed, the cost and labor involved necessitate transfer of multiple oocytes into single recipients, thus incurring the risk of multiple pregnancies in one recipient and no pregnancies in another recipient. NOTES
2 Table 1. Media Used for Oocyte and Embryo Manipulation Source Catalog Number Item Invitrogen b Medium 199 with Earle s salts Invitrogen Medium 199 with Hank s salts and Hepes Invitrogen Fetal bovine serum (certified; US origin) Invitrogen Gentamycin (cell-culture tested) SIGMA c H KU Heparin sodium (cell-culture tested) SIGMA D8437 Dulbecco s Modified Eagle s Medium/Ham s F-12 (DMEM/F-12) (500 ml) Removal of Ovaries and Transport to the Laboratory To ship excised ovaries for oocyte transfer or ICSI, the ovaries should be removed from the mare as soon as possible postmortem; however, delays in recovery from the mare of up to 5 h postmortem have been associated with production of foals from shipped ovaries. 4 We have obtained embryos and pregnancies from mares euthanized with barbiturate overdose and from mares from which the ovaries were removed under general anaesthesia (inhalant or xylazine/ketamine) before euthanasia. We suspect that euthanasia by potassium chloride overdose may reduce oocyte viability. After removal, ovaries should not be refrigerated. If transport time is less than 2 h, ovaries should be kept at body temperature (98 F or 37 C). For longer transport times, ovaries should be cooled to room temperature (about F or 20 C). The ovaries should be packaged to minimize temperature fluctuation during shipment. For shipment by car, ovaries may be placed in a styrofoam container with ballast (bottles or bags of water at the appropriate temperature) around them. For shipment by air, the currently recommended method of packaging uses an Equitainer. a The coolant cans should be warmed (for short transport) or at room temperature (for longer transport) for 24 h before shipment. If coolant cans at the appropriate temperature are not available, then the coolant cans should be removed from the Equitainer and a ballast (e.g., a rectal sleeve filled with water at the appropriate temperature) placed in the receptacle in the Equitainer. The ovaries should be bagged separately, either in normal saline or if none is available, in a closed plastic bag and placed on the ballast. Another ballast at the appropriate temperature should be placed on top of the ovaries. Frozen coolant cans, as are typically used with transported semen, should not be used. For best results, ovaries should be received within 6 h of the mare s death or removal of the ovaries from a live mare. Semen from the desired stallion should be shipped to the laboratory to arrive no later than the day after the ovaries are received. If oocyte transfer is being performed, the semen should be of high fertility, because the recipient mare will be inseminated; semen of low quality, including frozen semen, should be avoided. If ICSI is being performed, fresh, cooled-shipped, or frozen semen may be used. Reported rates of production of late pregnancies/ foals with oocytes recovered from clinical cases postmortem per donor mare are 8/25 (32%) by oocyte transfer 1 and 10/16 (63%) by ICSI and in vitro embryo culture to the blastocyst stage followed by transcervical transfer Shipment of Oocytes Postmortem In the case of postmortem production of foals, to avoid the reduction in viability associated with prolonged shipment of ovaries to the laboratory, as noted above, it is possible for the practitioner to recover oocytes from the ovaries and ship these to the laboratory. We have identified a medium, EH medium (Table 1), that allows holding of oocytes overnight at room temperature without a reduction in the rate of oocyte maturation or embryo development. 6 EH medium is a mixture of 40% M199/ Hank s/hepes and 40% M199 with Earle s salts with 25 g/ml gentamycin and 20% fetal calf serum. To recover oocytes from the follicles of excised ovaries, aspiration with a needle and syringe, as used in cattle, should not be used; this results in a low recovery rate and stripping of the majority of the cumulus from the oocytes. 7 This is because of the tighter and stronger attachment of the oocyte to the follicle wall in the horse than in other species. 8 Although there are many ways to recover oocytes from excised ovaries, the following is the procedure that is used in our laboratory, which has been associated with good meiotic and developmental competence of the recovered oocytes. 6,9,10 Method for Recovery of Oocytes From Excised Ovaries The medium used is Medium 199 with Hank s salts and 25 mm Hepes. Gentamycin (25 g/ml) is added to each bottle of medium when it is opened, and thus, every medium containing M199 in this report also contains 25 g/ml gentamycin. Cell culture-tested gentamycin should be used, rather than gentamycin produced for medical use, which may have contaminants. Sterile technique should be used while opening the bottle and when pouring from it, and the bottle should be stored in a refrigerator. Properly handled, the medium should store for a couple of weeks after opening. When we use
3 the medium for follicle scraping, we do not add serum because the follicular fluid provides enough protein to prevent cells from sticking to the dishes, but we do add heparin to about 20 IU/ml; cell culture-tested heparin should be used rather than medical heparin, which typically contains preservatives). The medium is poured into a specimen cup and warmed to room temperature. To recover oocytes, a sterile drape is placed on a table, and the supplies needed for the recovery are set out. Supplies include the following: scalpel blades (#10 or #20), all-plastic 10-ml syringe, 20- gauge needles, sterile pack with scalpel handle, forceps, scissors, and bone curettes (0.5 cm or a variety of sizes, if available), sterile gauze 4 4s, and 35-mm Petri dishes (both tops and bottoms of the Petri dishes may be used for collection). Examination or surgical gloves should be worn by the operator. An ovary is placed on a couple of 4 4s, and any attached tissue is cut off. Visible or palpable follicles should be opened first; using the scalpel blade, the follicle is cut open completely until it is on the half shell the interiors of both halves are entirely visible. Medium is aspirated from the cup into the syringe, and a 20-gauge needle is placed on the syringe. Using the bone curette, one swath is scraped along the follicle wall, and the accumulated tissue is rinsed into a Petri dish using a stream of medium from the syringe. In this manner, one swath at a time, the entire wall of the follicle is scraped until all possible granulosa cells have been collected, and no more tissue comes off the curette. If time permits, this dish may be searched, and if the oocyte is not present, the follicle may be scraped more. All visible follicles are opened and scraped, and then the ovaries are sliced in about 3-mm slices to open follicles within the parenchyma. If a follicle is big enough to see, it should be scraped for oocyte recovery. Handling of Immature Oocytes The recovered tissue is searched using a dissection microscope at magnification to locate the oocytes in the scraped granulosa cells. The oocyte may be entirely enclosed in cumulus or obscured by granulosa cells; needles may be used to move the cells around to locate the oocyte. In contrast to searching for embryos, roundness is not a major help in locating oocytes, which may appear irregular in shape on low power because of segregation of the light and dark areas of the cytoplasm. Oocytes are the same size as early zona-enclosed equine embryos ( 180 m in diameter). Oocytes typically appear as grey spots within the cumulus; closer inspection shows the clear area of the zona pellucida around the grey cytoplasm of the oocyte, which is diagnostic. The granulosa may be present in small shards or sheets (compact granulosa) or may appear puffy and expanded, even having a yellow jelly-like substance IN-DEPTH: (expanded granulosa). All collected oocytes should be sent to the ART lab, regardless of morphology. After it is located, the oocyte may be cut from the enclosing cumulus or granulosa using two needles, leaving a generous amount of cumulus with the oocyte. The oocyte-cumulus complex is then transferred to a holding Petri dish containing clean medium; this can be done with a steady hand using a sterile Pasteur pipette with rubber bulb or with a 25- l or larger pipettor and tip. If using the pipettor, work with this to just bring fluid and the oocyte into the very end of the pipettor tip. For either method, make sure fluid is in the pipette before the oocyte is picked up, or the oocyte may cling to the wall of the pipette when the fluid is ejected. When all oocytes have been collected, they are placed in a small vial ( 1 ml) of EH medium. We use 1-ml borosilicate glass vials for this procedure, but any small non-toxic vial should work. If no trained personnel or microscope are available to search for oocytes, all of the recovered cells can be sent in a larger volume (50 ml) of EH medium. 11 Because one of the EH medium components (M199 with Earle s salts) has bicarbonate in it, there should be very little air space in the vials so that the bicarbonate does not gas off, causing the medium to become alkaline. The oocytes or cells can be shipped overnight in this medium at room temperature (e.g., in an Equitainer with the coolant cans at room temperature, as described above for ovaries, by overnight courier) to arrive at the laboratory the next morning. 4. Oocyte Recovery From Live Mares Oocytes may also be recovered from live mares in practice, and the oocytes can be sent to a laboratory for oocyte transfer or ICSI. These procedures may be performed to obtain foals from mares that are not capable of providing an embryo by standard embryo flush (cannot conceive or maintain the embryo for 7 days because of oviductal or uterine adhesions, chronic uterine infection, or other conditions). ICSI may be used on oocytes recovered from live mares to obtain foals in cases in which there is a limited amount of semen available, such as in deceased stallions with only a few straws of frozen semen in existence. There are two main approaches to collection of oocytes from live mares, which are detailed in the following sections: (1) recovery of the maturing oocyte from the dominant preovulatory follicle after administration of gonadotropins human chorionic gonadotropin d (hcg) or a gonadotropin-releasing hormone e (GnRH) analog) to stimulate final maturation, or 2) recovery of immature oocytes from all follicles on the ovary through transvaginal ultrasound-guided follicle aspiration. Although recovery of the oocyte from the stimulated preovulatory follicle is a much simpler and often more rewarding procedure (the follicle may be aspirated through the flank with simple equipment and the recovery rate is high), shipment of the ma
4 turing oocyte requires careful handling and a portable incubator. However, recovery of the preovulatory oocyte works well if oocyte transfer to the oviduct of recipient mares is to be performed at the location at which the oocyte is recovered (see section on oocyte transfer). Recovery of immature oocytes from small follicles requires transvaginal ultrasound-guided follicle aspiration and its accompanying need for equipment and extra hands, and the recovery rate may be dauntingly low. However, immature oocytes recovered in this manner are still in meiotic arrest and thus, are fairly hardy and can be handled as described above for oocytes recovered from excised ovaries. Recovery of Oocytes From the Preovulatory Follicle of Live Mares Recovery of oocytes by aspiration of the gonadotropin-stimulated dominant preovulatory follicle is commonly performed to obtain oocytes for oocyte transfer. Recovery rates from these follicles are typically high (65 80%), because in these follicles, the oocyte-cumulus complex is expanding and loosening from the follicle wall in preparation for ovulation. One major drawback of aspiration of the stimulated preovulatory follicle is that it affords the chance of recovery of only one, or sometimes two, oocytes per cycle. Superstimulatory treatment is not clearly helpful, because the large size of the ovary (because of the presence of multiple preovulatory follicles) may cause aspiration of the follicles to be difficult when more than one or two are present. 16 The second major drawback to collection of the preovulatory oocyte at an equine practice for transport to a laboratory, as noted above, is that handling of the maturing oocyte is problematic. The oocyte recovered from the stimulated follicle is in the process of meiosis at the time that it is recovered and is exquisitely sensitive to temperature and other environmental influences, making it a challenge to ship it without reducing its viability. Method for Aspiration of the Preovulatory Follicle To recover oocytes from the stimulated preovulatory follicle, the mare s ovarian activity is monitored, and the preovulatory follicle is assessed for size and morphology, if data is available, in relation to the known follicle size before ovulation for that mare. Timing of gonadotropin administration can be a challenge, because the gonadotropins must be administered after the follicle becomes receptive to luteinizing hormone (LH) but before the endogenous LH surge. Only follicles that have responded to the gonadotropin treatment will provide high oocyte-recovery rates 14 ; however, if the mare has already responded to her own gonadotropin surge, then the age of the oocyte at the time of recovery (based on the expected time to ovulation from the administration of gonadotropins) will be inaccurate, and oocyte transfer or ICSI may be performed at the incorrect time. The follicle is typically aspirated h after gonadotropin administration; 24 h allows time for the cumulus to expand in response to gonadotropins, and 35 h is the limit at which it may be assumed that the follicle will not ovulate before aspiration. The oocyte is then incubated for the period of time needed so that it is fertilized (by oocyte transfer or ICSI) at the time that ovulation would have occurred (i.e., ovulation would be anticipated to be about 40 h after hcg administration or 44 h after deslorelin administration). Thus, if an oocyte is collected 24 h after gonadotropin stimulation, it is incubated h before manipulation; if it is collected 35 h after gonadotropin stimulation, it may be incubated 1 7 h before manipulation. There is no difference in pregnancy rate after oocyte transfer for oocytes handled under these different schedules. 17 The preovulatory oocyte may be simply recovered using a needle placed through a cannula in the flank while the ovary is manipulated per rectum. 14,18 To do this, an aspiration apparatus is prepared using a 20-cm, 13- to 15-gauge needle attached by extension tubing to an all-plastic 50-ml syringe. The needle is rinsed with heparin-containing medium (modified M199 with Hank s salts and Hepes with 25 g/ml gentamycin, 2% added fetal bovine serum, and 20 IU heparin/ml); by placing the needle in the container of heparinized medium and aspirating the medium through the tube into the syringe, pushing it back. Approximately 1 ml of medium remains in the tubing. Aspiration is performed with the mare standing in stocks, initially sedated with detomidine. A 4-4-cm area on the flank adjacent to the ovary containing the dominant follicle is shaved and scrubbed as for surgery. On palpation per rectum, the ovary is swung to the body wall to determine the location on the flank at which the needle should be inserted, and the skin in this area is blocked using 2 ml of 2% lidocaine. Immediately before the procedure, butorphanol f (10 mg) and N-butylscopolammonium bromide g ( mg) are given IV for analgesia and rectal relaxation. A stab incision is made through the blocked skin with a scalpel blade. Through palpation per rectum, viscera are cleared away from the internal abdominal wall. An equine trochar cannula ( 10 gauge in inner diameter) is placed horizontally through the skin incision and through the abdominal musculature into the peritoneal cavity. The trochar is removed, and the needle of the aspiration apparatus is inserted through the cannula. One hand manipulates the ovary per rectum, while the other hand directs the cannula, so that the cannula is placed against the dominant pre-ovulatory follicle. The needle is then advanced through the cannula into the follicle and then gently advanced so that it is well-seated in the follicle lumen. Aspiration is performed by a second person applying slow, gentle vacuum with the syringe
5 It is important that this be slow, because rapid aspiration may result in breakage of the oocyte. As the follicle empties, the area of the follicle wall overlying the needle is gently massaged per rectum. When the follicle appears empty, the needle is rotated and directed at different angles to pick up any pockets of fluid while very gentle vacuum is applied. At this point, if the follicle has responded to the gonadotropin stimulus (is truly preovulatory), blood and clear masses of granulosa cells should be visible in the tubing. Aspiration, massage of the follicle, and movement of the needle are continued until it is clear that no more material can be aspirated. The needle is withdrawn; the follicle is not flushed after aspiration. The needle is then placed in a container of medium, and this is aspirated into the syringe to recover any fluid or cells from the tubing. There are many possible methods for aspiration of the preovulatory follicle; the above method has proven both simple and consistently effective in our hands. 14,17,19 This flank approach is easily performed by veterinarians with experience in transrectal palpation. Alternatively, the preovulatory follicle may be aspirated by transvaginal ultrasound-guided follicle aspiration (TVA), as described below, for immature follicle aspiration; 13,20 however, this involves increased labor and expense, with no apparent increase in oocyte recovery rates. Handling of Maturing Oocytes After the follicular fluid has been collected, it should be maintained at 37 C during handling. A microscope with a warm stage is preferable. The syringe containing the aspirated fluid is held tip down for about 15 s to allow the cellular material to settle near the tip, and then, a small amount of fluid, containing cells, is expressed into a prewarmed 100-mm Petri dish. The preovulatory oocyte is located in the bloody follicular fluid based on its surrounding clear cumulus; this displaces the blood and forms a clear spot in the Petri dish that is often visible with the naked eye. Other clear areas represent expanded granulosa; the cumulus is typically the clearest area visible. All clear areas are examined under magnification and rolled with a needle to detect the grey, round oocyte in the center of the mass. If the oocyte is not found in the masses in the fluid and clots are present, the clots are torn apart with a needle to determine if the cumulus and oocyte are caught in them. If the oocyte is not in the first fluid expressed from the syringe, the process is repeated until all aspirated fluid has been searched. After it is located, the oocyte and surrounding cumulus should be placed in culture medium (M199/ Earle s salts/gentamycin with 10% fetal bone serum (FBS) in a CO 2 incubator) at C as soon as possible. Note that medium containing Earle s salts is bicarbonate-buffered and therefore, requires a5%co 2 atmosphere to maintain the appropriate ph. If shipped, the oocyte should be shipped in IN-DEPTH: equilibrated culture medium in a tightly capped vial in a portable incubator at 38 C. WhenaCO 2 incubator was not available, we have obtained pregnancies after transfer of oocytes recovered 35 h after hcg and maintained at 38 C in M199/Hank s/hepes/gentamycin with 10% FBS in a tube in a water bath while the recipient was prepared for transfer ( 1 h); this offers an effective way to perform oocyte transfer in practice without investing in cell-culture equipment. However, we do not have any data on the effectiveness of this Hank s salts medium in maintaining preovulatory oocyte viability for longer periods. Hank s salts are a phosphate buffer system that maintains ph at room atmosphere, and the Hepes is a zwitterionic buffer that aids ph maintenance, which may be used either at room atmosphere or in 5% CO 2. Recovery of Immature Oocytes From Live Mares Aspiration of immature oocytes from all follicles present on the ovaries is an alternative option to obtain oocytes from mares in practice for shipment to a laboratory for ART. The pool of small follicles contains immature oocytes that are not in a preovulatory state; oocytes recovered from these follicles will need to be matured in vitro. Unfortunately, recovery rates on aspiration of immature follicles in the mare may be low, often 25% or less Again, this is because of the strong attachment of the equine oocyte to the follicle wall. 8 However, some laboratories have achieved 50 60% recovery rates when implementing aspiration techniques that include manipulation of the ovary during aspiration and flushing of each follicle up to eight times through a double-lumen aspiration needle with separate in- and outflow channels. 19,25,26 We attempted this procedure in our laboratory at Texas A&M for the first time in 2008; research Quarter Horse-type mares were subjected to TVA of all follicles once every 14 days throughout the breeding season, without monitoring of follicle growth between aspirations. We achieved an oocyte recovery rate of 54% (average of five oocytes per mare per aspiration session). 19 Of the recovered oocytes, 57% matured in vitro. After subjecting the oocytes to ICSI and embryo culture, 33% developed to the blastocyst stage. This work supports the report of Colleoni et al. 26 on results from a clinical TVA/ICSI program, indicating that collection of immature follicles by TVA, followed by in vitro maturation and ICSI, may be effective enough to be offered clinically. Recovery of oocytes from immature follicles by TVA may be performed by the equine clinician in practice. However, it involves investment in specialized equipment (transvaginal probe, needles, and vacuum pump) and has the associated costs involved in the cleaning and sterilization of the equipment and increased labor associated with its use. Most importantly, there seems to be a learning curve and an effect of technician on the oocyte
6 recovery rates achieved, regarding both manipulation of the probe and ovaries and manipulation of the aspiration needle. Nevertheless, a major advantage of the procedure in clinical practice is that the oocytes recovered will be immature and thus may be easily sent to the ART laboratory by overnight shipment in EH medium at room temperature, as for oocytes collected postmortem, above. Method for Transvaginal Aspiration of Immature Follicles The first report of TVA in the mare was that of Bruck et al., 20 and many others have reported on methods to do this procedure. 22,23,26 28 We based the method used in our laboratory on these previous reports and personal communications with experienced operators. hi The system used in our laboratory requires three people. One manipulates the ovary per rectum and directs the ultrasound probe, the second advances and manipulates the needle and controls the vacuum pump, and the third controls the inflow of medium and records information. Other systems in which one person can control three functions using a trigger-operated pump and vacuum on the handle of the ultrasound probe have been developed. j The mare is restrained in stocks with the tail bagged and tied and is initially sedated with detomidine. k The perineal region is cleaned using a povidone-iodine scrub. A 3-mHz sector ultrasound probe, fitted within a transvaginal probe handle and needle guide, is treated lightly with a sterile gel (we use gel, l which has been tested to be non-embryo toxic) and covered with a plastic sheath. We cut a small area from the tip of the sheath so that the face of the ultrasound probe is not covered. Butorphanol (10 mg) and N-butylscopolammonium bromide ( mg, IV) are administered immediately before beginning the procedure. The external surface of the sheath covering the transvaginal probe is lubricated lightly with the sterile gel, and the probe is placed into the vagina. A 12- or 15-gauge double-lumen oocyte aspiration needle m is inserted into the needle guide. For inflow, in our laboratory, we use an all-plastic syringe under manual pressure, which is connected by a one-way valve (#CFS36, Mila) to a container of medium. The medium that we employ for flushing is M199/Hank s/ Hepes/gentamycin with 20 IU/ml heparin and 0.4% FBS; others use modified phosphate buffered saline (PBS) or embryo-flushing medium. The outflow of the needle flows into a collection vessel (we use a 500-ml flask) that is connected to a vacuum pump. The vacuum should be set so that 20 ml of medium may be aspirated per minute. n The operator holds the ovary by transrectal palpation and directs the ultrasound probe; by manipulating the ovary and the probe, follicles are visualized on the ovary through the vaginal wall. A follicle is lined up on the needle guide on the ultrasound screen, and the needle is advanced into the follicle by the needle handler. The follicle is aspirated and then flushed with medium. An attempt is made to flush and aspirate each follicle six times. The needle is rotated during each aspiration phase to try to scrape granulosa from the follicle, and the ovary is gently moved back and forth during this time. We have noted that oocytes may be recovered even if the filling and emptying of the follicle is not easily visualized on the screen, and therefore, we continue for six flushes and aspirations whether or not we see the follicle during this time. We struggled with the appropriate placement of the hands, probe, and ultrasound screen during TVA. The method that we developed was to aspirate the right ovary using the left hand per rectum and the right hand on the probe handle, with the ultrasound machine, vacuum, etc. on the left side of the mare. This allows the probe handle to be on the left side of the operator, so it may be pushed away to the left to allow the tip of the probe to move to the right to visualize the ovary. The equipment is reversed for the opposite ovary. Others are able to aspirate both ovaries with one setup. Placement of a urinary catheter is helpful to avoid having to manipulate the ultrasound probe over a full bladder. After aspiration of all follicles is completed, mares are treated with flunixin meglumine (500 mg, IV). We have found that there is little detrimental effect of a single aspiration session on the ovary; however, repeated aspiration of all immature follicles using a 12-gauge needle over the entire season was associated with thickening of the serosa over the ovary in some mares, possibly because of organization of hemorrhage in the mesovarium. o Fertility seems to be normal in mares subjected to repeated TVA, 24 although formation of ovarian abscesses was found in a mare after repeated follicle aspiration; 29 at least one death after TVA of the preovulatory follicle, related to hemorrhage from the uterine artery, has been reported. 30 After the completion of the aspiration session, the bottles containing the aspirated fluid are filtered through a cup embryo filter, p and oocytes are located using a dissection microscope at magnification. Identified oocytes are handled as for immature oocytes recovered from excised ovaries. 5. Oocyte Transfer Oocyte transfer (i.e., surgical transfer of a mature oocyte to the oviduct of an inseminated recipient mare) is a method of ART that can be performed in practice. An effective method of oocyte transfer was first reported by Carnevale and Ginther in In that study, a pregnancy rate of 83% was achieved, and Carnevale et al have shown that this high pregnancy rate is repeatable when fertile mares and stallions are used We achieved a 75% pregnancy rate in our first attempt with this procedure. 14 Therefore, it is puzzling that, despite the relative simplicity of the oocyte-transfer procedure, few reports are available on this technique
7 from other laboratories, and few veterinarians offer the service to clients. Oocyte transfer is best used when only one oocyte of high viability is available for transfer (e.g., when an oocyte has been recovered from the dominant preovulatory follicle of the donor mare). If multiple oocytes are available, the option exists to perform oocyte transfer with them, but the problem of multiple pregnancies within the recipient and associated loss of potential foals then occurs. Transfer of multiple oocytes to the oviduct of a recipient mare and then uterine flush to recover embryos followed by standard embryo transfer of the recovered embryos singly to secondary recipient mares could be considered, but these techniques carry the potential for inefficiencies at each step and again, loss of potential pregnancies. When multiple oocytes are available, the use of ICSI and in vitro embryo culture to the blastocyst stage seems to be the method of choice. Donor/Recipient Considerations in Oocyte Transfer For oocyte transfer, typically the oocyte is retrieved from the preovulatory follicle of the donor mare between 24 and 35 h after hcg or deslorelin administration. As described above, the oocyte is placed into culture until it is mature (e.g., until about 40 h after the time of hcg administration or 44 h after deslorelin administration). If a CO 2 incubator is available, the oocyte may be cultured in M199 with Earle s salts and 10% FBS in an atmosphere of 5% CO 2 in air at 38.2 C. If a CO 2 incubator is not available, we have achieved pregnancies after oviductal transfer of oocytes collected from the follicle 35 h after hcg and held in M199 with Hank s salts and 10% FBS at 38 C for the time needed to prepare the recipient for transfer ( 1 h). The recipient should be immediately periovulatory at the time that the oocyte is transferred. This can be achieved in the cyclic recipient by synchronizing follicle growth and gonadotropin administration with that of the donor mare and aspirating the oocyte from the recipient follicle before the recipient is used for transfer. The question arises on what to do if the recipient s oocyte is not recovered on aspiration. If the follicle is completely aspirated (until it has collapsed and blood is recovered in the aspiration tubing), there is very little chance of the recipient conceiving. 17 However, if the follicle is incompletely aspirated (e.g., because the needle slipped from the follicle), there is about a 30% chance of the recipient conceiving on her own, and she should not be used. 17,34 Alternatively, nonovulatory recipients may be used. These may be mares in very early estrus or anestrus, with follicles less than 25 mm in diameter and no luteal function. Such recipients serve well when treated with estrogen (e.g., estradiol 17ß, 3 mg daily) while the donor mare s follicle is approaching ovulatory size and then progesterone after transfer. 32,35,36 Oocyte recipients are inseminated 6 h before transfer or both before and after transfer. Persistent post-breeding endometritis is a common complication of this procedure, probably related to the drugs used for surgery and post-surgical analgesia. 17 Therefore, the recipient mare should be treated as a mare that is susceptible to endometritis, and if possible, time should be allowed between insemination and oocyte recovery or surgery to allow elimination of debris from the uterus; ancillary treatments such as oxytocin, uterine flush, or intrauterine antibiotic administration should be used as indicated by clinical signs (e.g., accumulation of intrauterine fluid). Transfer Technique IN-DEPTH: The oocyte transfer itself is performed by flank laparotomy in the standing mare under tranquilization and analgesia and local anaesthesia with an inverted L-block. 14 An incision is made immediately cranial to the tuber coxae, extending 15 cm ventrally. The incision is extended by direct or blunt dissection until the peritoneal cavity is reached. After the incision has been made and exposure of the ovary has been shown, the oocyte is taken from the incubator and transferred into warmed M199/ Hank s/hepes with 10% FBS. Under a microscope at magnification, the oocyte is loaded into a sterile, cut, fire-polished Pasteur pipette that is attached with silastic tubing to a 1-ml syringe with a minimum ( 0.5 ml) of medium. The loading is done with a small amount of fluid, then an air pocket, and finally, the fluid with the oocyte, as for when loading an embryo in a straw for embryo transfer; approximately 0.5 ml of air should be drawn into the syringe before loading so that it can be used to express the fluid at the time of transfer. Alternatively, a Tomcat catheter, cut to approximately one-half the original length, may be attached to a 1-ml syringe and used to load and transfer the oocyte. The ovary is exteriorized, and the infundibulum of the oviduct is grasped with a forcep. The oviduct is cannulated while being visualized from the serosal surface this is more effective than trying to find the opening of the oviduct on the fimbrial surface. A hemostat or mosquito forceps is inserted under the flap of the infundibulum and into the ampulla and the pipette or catheter is threaded beside it. The guide is withdrawn, and the contents of the pipette are expressed into the ampulla. The ovary is replaced in the abdomen, and the incision is closed. Pregnancy rates after oocyte transfer with oocytes from mature preovulatory follicles are commonly over 70% when fertile donor mares and stallions are used 13,14,31 but may be much lower clinically when mares may be old and/or subfertile, and stallion semen quality or handling during transport may be compromised
8 6. In Vitro Fertilization Standard in vitro fertilization (IVF; i.e., placing sperm and oocytes together in culture, resulting in fertilization) has historically not been successful in the horse. The failure of equine IVF seems to be related to the failure of the sperm to penetrate the zona pellucida in vitro. In 2009, a 60% rate of equine fertilization in vitro was reported after chemical treatment of sperm to induce hyperactivated motility, 37 thus opening up an exciting area for further investigation in equine IVF. Currently, embryo development has not been established after this new procedure, but work in this area is ongoing. Establishment of effective methods for standard IVF would allow the use of this ART in many equine practices, because it alleviates the need for micromanipulation equipment and associated expertise. 7. ICSI Establishment of a program for ICSI requires setting up a laboratory equipped with cell-culture facilities (CO 2 and mixed-gas incubators) and a micromanipulator microscope, and ensuring the presence of personnel with the expertise and dedicated time to perform the procedures. Although this is possible to do in practice, most practitioners will instead send mares or oocytes to central facilities that are already equipped to perform ICSI and in vitro embryo production. Production of embryos through ICSI is applicable to mares from which multiple oocytes are recovered, such as those that had all follicles aspirated by TVA or oocytes recovered postmortem. ICSI and in vitro culture to the blastocyst stage may also be used when the dominant preovulatory follicle is aspirated, to avoid the need for surgery for oocyte transfer. However, with the one preovulatory oocyte, the chance of a resulting pregnancy is lower by ICSI than it is by oocyte transfer. For example, for preovulatory oocytes from normal mares, there may be an approximately 40% chance of pregnancy after ICSI/in vitro culture and transfer of the resulting blastocyst versus a 75% chance of pregnancy after oocyte transfer. 13,19 In contrast, the likelihood of pregnancy when in vitro-matured oocytes are used is similar with both techniques. The poor performance of preovulatory oocytes after ICSI/in vitro culture may represent a mismatch between the requirements of the in vivo-matured oocyte and the current system, which was developed using in vitromatured oocytes. ICSI is also applicable as a means to produce foals from stallions for which there is only a limited quantity of sperm, such as for limited stores of frozen semen or an older stallion that can no longer produce enough normal, motile sperm for a standard insemination dose. In ICSI, only one viable sperm is needed to fertilize each oocyte. Sperm are typically prepared by swim-up selection of the most highly motile sperm; however, immotile sperm can also result in blastocyst production after ICSI, although blastocyst rates are lower. 38 Fresh and frozen sperm provide similar fertilization rates. 39 Stores of frozen sperm may be thawed, diluted 1:100, and refrozen with no decrease in embryo development; 38 thus, ICSI allows the production of multiple embryos and foals from stores of semen that might not yield a pregnancy when used for standard insemination. To perform ICSI, the sperm and mature oocytes are placed in droplets of medium on a Petri dish and viewed on an inverted microscope. Using micromanipulation, one sperm is picked up into a small-bore (8 m in diameter) pipette and is injected into the oocyte. Use of the Piezo drill during sperm injection has been associated with the greatest efficiency in embryo development after ICSI. 3,9 After sperm injection, the presumptive zygotes may be cultured in a modified Dublecco s Modified Eagle s Medium/ Ham s F-12 (DMEM/F-12) cell-culture medium for 7 10 days until they form blastocysts. Equine embryos do not form expanded blastocysts in vitro, because the zona pellucida does not thin normally. In addition, the embryonic capsule does not form in vitro. Thus, equine in vitro-produced blastocysts are recognized by the formation of an organized layer of cells, presumably the trophoblast, adjacent to the zona pellucida. In vitro blastocyst production rates after ICSI seem to vary among laboratories. Colleoni et al. 26 reported an in vitro blastocyst production rate of 13% in a commercial program using oocytes obtained by TVA. Galli et al. 3 reported up to 26% blastocyst development in vitro in a research study using one stallion, and similar rates have been reported by others. 40 Altermatt et al. 41 reported a 29% blastocyst rate after ICSI of oocytes matured in vivo (recovered from the stimulated preovulatory follicle). 41 Rates of in vitro blastocyst production per injected oocyte in our laboratory at Texas A&M are typically 25 35% and have been up to 42% for oocytes from normal mares. 2,6,9,10 In cattle, oocytes recovered from preovulatory follicles in vivo provide much higher blastocyst rates than do oocytes that are matured in vitro, but as noted above, this does not seem to be the case in horses, perhaps because of inefficiencies in our embryo culture systems. In a recent report, we achieved a 33% blastocyst formation after ICSI from oocytes recovered from immature follicles of mares by TVA and matured in vitro versus a 41% rate of blastocyst formation after ICSI of oocytes recovered from stimulated preovulatory follicles. 19 Initially, there seemed to be problems with low pregnancy rates and formation of trophoblast-only pregnancies after transfer of embryos produced by ICSI. 42,43 However, with improved embryo culture conditions, these problems seem to have abated. In our most recent study on transfer of ICSI-produced embryos, 13/17 (76%) established pregnancy, and all but one showed a normal heartbeat at 25 days. 43 Colleoni et al, 26 after commercial TVA, in
9 vitro oocyte maturation, ICSI, in vitro embryo culture, and transcervical blastocyst transfer, reported an initial pregnancy rate of 15/18 (83%), with 3 of 15 pregnancies lost before 30 days and 2 more pregnancies lost before 60 days. 26 To our knowledge, there have been no reports on the health of foals produced by ICSI. Colleoni et al. 26 reported that of six foals resulting from embryos produced by ICSI, one died at birth and one died from septicemia at 10 days postpartum. The foals that we have produced by ICSI at Texas A&M all have been normal from birth. There is no indication that foals produced by ICSI suffer from a large offspring syndrome, as may be seen after transfer of cattle and sheep embryos produced in vitro, or from problems at birth, including neonatal maladjustment, as do cloned foals Nuclear Transfer (Cloning) In nuclear transfer, a cell from the donor animal is transferred to an enucleated oocyte, and the oocyte is stimulated to begin embryonic development. Performance of nuclear transfer requires essentially the same equipment as is required for ICSI; however, the procedures are more involved, and the expertise required to perform them is greatly increased. Currently, only three laboratories have reported birth of live foals after nuclear transfer with adult somatic cells. Although cloning is performed only at a few specialized laboratories, equine practitioners may be involved in cloning through consultation with clients on the possible benefits and drawbacks of the procedure, collection of tissue from the donor horse, and possibly, care of the pregnant mare and neonate. Cloning as an ART Production of foals by nuclear transfer (NT) seems to be a repeatable procedure in the horse. The first cloned equid, a mule produced from the cells of a 45-day gestation fetus, was produced in Foals from nuclear transfer with adult horse somatic cells have been reported from the laboratory of Cesare Galli in Italy 46,47 and our laboratory at Texas A&M A commercial laboratory, ViaGen, has announced birth of cloned foals in the popular press. At the time of writing, the only reports on the efficiency of cloning using cells from adult horses are from our laboratory and the laboratory in Italy. In two studies, the Italian laboratory produced a total of 3 live foals after transfer of over 100 embryos. One foal died of septicemia, and the other two foals were healthy. 46,47 Texas A&M has produced 14 live foals after transfer of 54 embryos, of which 2 have died 1 from pneumonia and 1 after a hypotensive episode while being induced for anesthesia to explore a possible ruptured bladder This live foal rate per embryo transferred is notably higher than the 5 10% rate of viable offspring per transferred embryo reported in other species. 51 In our laboratory, the pregnancy rate after transfer of cloned embryos approaches normal ( 60%). IN-DEPTH: Approximately 50% of established cloned pregnancies are lost, and these losses are distributed throughout gestation. At birth, cloned foals have an increased incidence of maladjustment, enlarged umbilical remnant, and contracted tendons and valgus deformity of the front legs. 44 However, these conditions respond to treatment, and the health of cloned foals, after the first 1 2 wks post-partum, seems to be normal. Selection of Horses for Cloning Cloning should be regarded as a method to preserve genetics for breeding rather than as a method to produce individuals of a given type. As noted above, at this time, cloned foals have a higher incidence than do normally conceived foals of some problems at birth that could affect their performance as adults. Although cloned foals have the same genetics as the original donor horse, they may use these genes differently (have different epigenetic patterns) and thus, may be phenotypically slightly different from the original animal. However, when cloned horses are used for breeding, their offspring should be completely normal. The selection of a horse for cloning is largely a decision of the owner, similar to other breeding decisions. Cloning is an option when a horse of a valued bloodline cannot produce offspring or when an owner desires more offspring from this horse s genetics. A major application of cloning is to rescue the genetics of geldings that have proven themselves to be outstanding individuals the resulting cloned colt, left intact, will sire foals that should be identical to the foals that the original animal would have produced. Cloning also has the potential to narrow the gene pool; for instance, if a popular stallion that has already sired a lifetime worth of foals is cloned, then that clone is used at stud. It should be noted that this narrowing of the gene pool is not specific to cloning: artificial insemination, semen transport, and frozen semen are current ARTs that allow production of many times more offspring from a given stallion than would be possible by natural cover alone. Currently, major breed associations in the United States do not recognize foals produced by NT. The American Quarter Horse Association voted in 2010 to uphold its ban on registration of foals produced by NT. Cloned horses are allowed to compete in National Cutting Horse Association competitions in the United States as well as other competitions that require only identification of competitors rather than breed registration. There are sport horse registries in Europe that will register cloned foals. The effect of registration status on the value of the cloned animal and its offspring must be weighed in the decision to perform cloning. Clones will have the chromosomal DNA of the donor horse. However, the cells of the clone will have the mitochondria and mitochondrial DNA of the oocyte used as a host for the NT. This has no
10 effect on the offspring of a male clone, because the mitochondria of the sperm are eliminated after fertilization. However, because the eggs of the female clone will contain the clone s mitochondria (originating from the host oocyte), a female clone will pass these mitochondria on to her offspring. Thus, the foals of a cloned mare will not be 100% genetically identical to the foals of the original mare unless the oocytes used for cloning have been selected for mitochondrial identity. The impact of this mitochondrial heterogeneity on the performance or phenotype of the offspring is currently unknown in the horse; however, in cattle, there is no difference in growth rates or milk composition or production between donor cattle and clones of those cattle produced using slaughterhouse-derived oocytes More information on mitochondrial identity among horse breeds and effects of mitochondria on horse phenotype should be available in the near future. Tissue Collection Procedures for cloning start with collection of a tissue sample from the donor animal. This is typically a small (0.5 cm 2 ) piece of subcutaneous connective tissue, collected by everting the skin after making a small incision. The tissue is placed immediately into chilled culture medium (embryoholding medium may be used). Care should be taken to place the tissue immediately into cold medium and not expose the tissue to sunlight or other adverse environments, to keep it cold, and to ship the tissue to the laboratory for culture as quickly as possible. Contamination is the major reason that cells may not grow in culture, and therefore, careful sterile technique is essential during tissue collection. A tissue may produce cells well in culture, but the ability of the cells to produce viable offspring through nuclear transfer is not clear until this has occurred Biopsy of Embryos for Pre-Implantation Genetic Diagnosis Our laboratory has recently developed a method for biopsy of embryos, both in vitro produced and those recovered in vivo, for pre-implantation genetic diagnosis. 55 Cells recovered by embryo biopsy may be analyzed for genetic diseases including hyperkalemic periodic paralysis (HYPP), hereditary equine regional dermal asthenia (HERDA), and glycogen branching enzyme deficiency (GBED); work is currently ongoing to improve the accuracy of these genetic evaluations given the small number of cells obtained. The embryos have a normal pregnancy rate (83%) after biopsy, and nine foals produced from biopsied embryos were all normal. q In the near future, this technique should allow horse breeders to avoid the production of individuals affected with these devastating genetic diseases; we anticipate that this technique will allow for the eventual elimination of these diseases from the industry altogether. We have established that embryos may be collected from mares on day 6 or 7 after ovulation and shipped to the laboratory overnight in standard commercial embryo-holding medium with no detrimental effect on pregnancy rates after biopsy. At the laboratory, the embryo is biopsied, and the collected cells are saved for genetic analysis. After the biopsy, the embryo may be shipped back for immediate transfer; if the genetic analysis shows the presence of the defective gene, the pregnancy may be terminated. Alternatively, the embryo may be vitrified after biopsy. We hope to be able to offer embryo biopsy as a clinical service in the near future. 10. Embryo Vitrification Freezing and vitrification of early equine embryos (less than 300 m in diameter) are effective; however, this necessitates recovery of embryos on day 6 after ovulation, which has been reported to be associated with lower rates of embryo recovery. 56 Vitrification is a relatively simple technique that may be performed in practice without extensive equipment and has been well-described previously by Carnevale et al. 57 However, freezing or vitrification of embryos 300 m in diameter has resulted in low pregnancy rates after transfer. In 2008 and 2009, while working on embryo biopsy, our laboratory discovered that embryos biopsied by micromanipulation could be cryopreserved effectively. 58 This extended to even expanded day 7 blastocysts, cryopreservation of which had not been previously effective. Embryos up to 700 min diameter were collapsed by micromanipulation, and then, were vitrified in a small-diameter pipette. r The pregnancy rate after biopsy and vitrification using this technique was 50%. 58 At the time of writing, we have not attempted other methods of cryopreservation combined with blastocyst puncture, but it is possible that blastocyst collapse may allow even standard cryopreservation procedures to be performed; this is a currently developing area. Because blastocyst puncture currently requires micromanipulation capabilities, for clinical use, embryos may be recovered in the field, shipped to the laboratory for vitrification, and then, shipped back to the practitioner for eventual transfer. We are working to develop a method for blastocyst collapse that may be used in the field. 11. Summary The year 2010 brings to an end a decade of amazing progress in equine ART. These advanced techniques contribute greatly to the tools available to practitioners working in equine reproduction. The next decade should show a notable increase in efficiency and accessibility of many equine ART procedures. Acknowledgments Work in the author s laboratory was supported in part by the Link Equine Research Endowment Fund
11 at Texas A&M University, the American Quarter Horse Foundation, and Ms. Kit Knotts. References and Footnotes 1. Carnevale EM, Coutinho da Silva MA, Preis KA, et al. Establishment of pregnancies from oocytes collected from the ovaries of euthanized mares, in Proceedings. 50th Annual American Association of Equine Practitioners Convention 2004; Ribeiro BI, Love LB, Choi YH, et al. Transport of equine ovaries for assisted reproduction. Anim Reprod Sci 2008; 108: Galli C, Colleoni S, Duchi R, et al. Developmental competence of equine oocytes and embryos obtained by in vitro procedures ranging from in vitro maturation and ICSI to embryo culture, cryopreservation and somatic cell nuclear transfer. Anim Reprod Sci 2007;98: Carnevale EM, Maclellan LJ, Coutinho da Silva MA, et al. Pregnancies attained after collection and transfer of oocytes from ovaries of five euthanatized mares. J Am Vet Med Assoc 2003;222: Hinrichs K, Choi YH, Norris JD, et al. Use of intracytoplasmic sperm injection and in vitro culture to the blastocyst stage for clinical production of foals post mortem. Anim Reprod Sci 2010;121(Suppl 1 2): Choi YH, Love LB, Varner DD, et al. Holding immature equine oocytes in the absence of meiotic inhibitors: effect on germinal vesicle chromatin and blastocyst development after intracytoplasmic sperm injection. Theriogenology 2006;66: Hinrichs K. The relationship of follicle atresia to follicle size, oocyte recovery rate on aspiration, and oocyte morphology in the mare. Theriogenology 1991;36: Hawley LR, Enders AC, Hinrichs K. Comparison of equine and bovine oocyte-cumulus morphology within the ovarian follicle. Biol Reprod 1995;1: Hinrichs K, Choi YH, Love LB, et al. Chromatin configuration within the germinal vesicle of horse oocytes: changes post mortem and relationship to meiotic and developmental competence. Biol Reprod 2005;72: Choi YH, Love LB, Varner DD, et al. Effect of holding technique and culture drop size in individual or group culture on blastocyst development after ICSI of equine oocytes with low meiotic competence. Anim Reprod Sci 2007;102: Velez IC, Norris JD, Ballard K, et al. Effect of method of overnight shipment of oocytes or ovaries on blastocyst formation after intracytoplasmic sperm injection. Anim Reprod Sci 2010;121(Suppl 1 2): Carnevale EM, Coutinho da Silva MA, Panzani D, et al. Factors affecting the success of oocyte transfer in a clinical program for subfertile mares. Theriogenology 2005;64: Carnevale EM, Ginther OJ. Defective oocytes as a cause of subfertility in old mares. Biol Reprod 1995;1: Hinrichs K, Matthews GL, Freeman DA, et al. Oocyte transfer in mares. J Am Vet Med Assoc 1998;212: Hinrichs K, Kenney DF, Kenney RM. Aspiration of oocytes from mature and immature preovulatory follicles in the mare. Theriogenology 1990;34: Maclellan LJ, Carnevale EM, Coutinho da Silva MA, et al. Pregnancies from vitrified equine oocytes collected from super-stimulated and non-stimulated mares. Theriogenology 2002;58: Hinrichs K, Betschart RW, McCue PM, et al. Effect of timing of follicle aspiration on pregnancy rate after oocyte transfer in the mares. J Reprod Fertil 2000;56(Suppl): Vogelsang MM, Kreider JL, Bowen MJ, et al. Methods for collecting follicular oocytes from mares. Theriogenology 1988;29: Jacobson CC, Choi YH, Hayden SS, et al. Recovery of mare oocytes on a fixed biweekly schedule, and resulting blastocyst formation after intracytoplasmic sperm injection. Theriogenology 2010;73: IN-DEPTH: 20. Brück I, Raun K, Synnestvedt B, et al. Follicle aspiration in the mare using a transvaginal ultrasound-guided technique. Equine Vet J 1992;24: Cook NL, Squires EL, Jasko DJ. Repeated transvaginal follicular aspiration in cyclic mares. Theriogenology 1993; 39: Duchamp G, Bézard J, Palmer E. Oocyte yield and the consequences of puncture of all follicles larger than 8 millimetres in mares. Biol Reprod 1995;1: Kanitz W, Becker F, Alm H, et al. Ultrasound-guided follicular aspiration in mares. Biol Reprod 1995;1: Mari G, Merlo B, Iacono E, et al. Fertility in the mare after repeated transvaginal ultrasound-guided aspirations. Anim Reprod Sci 2005;88: Bøgh IB, Bézard J, Duchamp G, et al. Pure preovulatory follicular fluid promotes in vitro maturation of in vivo aspirated equine oocytes. Theriogenology 2002;57: Colleoni S, Barbacini S, Necci D, et al. Application of ovum pick-up, intracytoplasmic sperm injection and embryo culture in equine practice, in Proceedings. 53rd Annual American Association of Equine Practitioners Convention 2007: Meintjes M, Bellow MS, Broussard JR, et al. Repeated transvaginal ultrasound-guided oocyte retrieval from pregnant mares. Theriogenology 1994;41: Franz LC, Squires EL, O Donovan MK, et al. Collection and in vitro maturation of equine oocytes from estrus, diestrus and pregnant mares. J Equine Vet Sci 2001;21: Bøgh IB, Brink NS, Jensen HE, et al. Ovarian function and morphology in the mare after multiple follicular punctures. Equine Vet J 2003;35: Vanderwall DK, Woods GL. Severe internal hemorrhage resulting from transvaginal ultrasound-guided follicle aspiration in a mare. Equine Vet J 2002;22: Carnevale EM, Coutinho da Silva MA, Maclellan LJ, et al. Use of parentage testing to determine optimum insemination time and culture media for oocyte transfer in mares. Reproduction 2004;128: Carnevale EM, Maclellan LJ, Coutinho da Silva MA, et al. Equine sperm-oocyte interaction: results after intraoviductal and intrauterine inseminations of recipients for oocyte transfer. Anim Reprod Sci 2001;68: Carnevale EM, Maclellan LJ, Coutinho da Silva MA, et al. Comparison of culture and insemination techniques for equine oocyte transfer. Theriogenology 2000;54: Palmer E, Duchamp G, Cribiu EP, et al. Follicular fluid is not a compulsory carrier of the oocyte at ovulation in the mare. Equine Vet J 1997;25(Suppl): Hinrichs K, Provost PJ, Torello EM. Birth of a foal after oocyte transfer to a nonovulating, hormone-treated mare. Theriogenology 1999;51: Hinrichs K, Provost PJ, Torello EM. Treatments resulting in pregnancy in nonovulating, hormone-treated oocyte recipient mares. Theriogenology 2000;54: McPartlin LA, Suarez SS, Czaya CA, et al. Hyperactivation of stallion sperm is required for successful in vitro fertilization of equine oocytes. Biol Reprod 2009;81: Choi YH, Love CC, Varner DD, et al. Equine blastocyst development after intracytoplasmic injection of sperm subjected to two freeze-thaw cycles. Theriogenology 2006;65: Choi YH, Love CC, Love LB, et al. Developmental competence in vivo and in vitro of in vitro-matured equine oocytes fertilized by intracytoplasmic sperm injection with fresh or frozen-thawed sperm. Reproduction 2002;123: Matsukawa K, Akagi S, Adachi N, et al. In vitro development of equine oocytes from preserved ovaries after intracytoplasmic sperm injection. J Reprod Dev 2007;53: Altermatt JL, Suh TK, Stokes JE, et al. Effects of age and equine follicle-stimulating hormone (efsh) on collection and viability of equine oocytes assessed by morphology and developmental competency after intracytoplasmic sperm injection (ICSI). Reprod Fertil Dev 2009;21:
12 42. Li X, Morris LHA, Allen WR. Influence of co-culture during maturation on the developmental potential of equine oocytes fertilized by intracytoplasmic sperm injection (ICSI). Reproduction 2001;121: Hinrichs K, Choi YH, Walckenaer BE, et al. In vitro-produced equine embryos: production of foals after transfer, assessment by differential staining, and effect of medium calcium concentrations during culture. Theriogenology 2007;68: Johnson AK, Clark-Price S, Choi YH, et al. Physical and clinicopathologic findings in foals produced by somatic cell nuclear transfer 14 cases ( ). J Am Vet Med Assoc 2010;236: Woods GL, White KL, Vanderwall DK, et al. A mule cloned from fetal cells by nuclear transfer. Science 2003;301: Galli C, Lagutina I, Crotti G, et al. A cloned horse born to its dam twin. Nature 2003;424: Lagutina I, Lazzari G, Duchi R, et al. Somatic cell nuclear transfer in horses: effect of oocyte morphology, embryo reconstruction method and donor cell type. Reproduction 2005;130: Hinrichs K, Choi YH, Love CC, et al. Production of horse foals via direct injection of roscovitine-treated donor cells and activation by injection of sperm extract. Reproduction 2006; 131: Hinrichs K, Choi YH, Varner DD, et al. Production of cloned horse foals using roscovitine-treated donor cells and activation with sperm extract and/or ionomycin. Reproduction 2007;134: Choi YH, Hartman DL, Fissore RA, et al. Effect of sperm extract injection volume, injection of PLC crna, and tissue cell line on efficiency of equine nuclear transfer. Cloning Stem Cells 2009;11: Wells DN. Animal cloning: problems and prospects. Rev Sci Tech 2005;24: Wells DN, Forsyth JT, McMillan V, et al. The health of somatic cell cloned cattle and their offspring. Cloning Stem Cells 2004;6: Norman HD, Walsh MK. Performance of dairy cattle clones and evaluation of their milk composition. Cloning Stem Cells 2004;6: Watanabe S, Nagai T. Health status and productive performance of somatic cell cloned cattle and their offspring produced in Japan. J Reprod Dev 2008;54: Choi YH, Gustafson-Seabury A, Velez IC, et al. Viability of equine embryos after puncture of the capsule and biopsy for pre-implantation genetic diagnosis. Reproduction, in press. 56. Squires EL, Imel KJ, Iuliano MF, et al. Factors affecting reproductive efficiency in an equine embryo transfer programme. J Reprod Fertil 1982;32(Suppl): Carnevale EM, Eldridge-Panuska WD, Caracciolo di Brienza V. How to collect and vitrify equine embryos for direct transfer, in Proceedings. 50th Annual American Association of Equine Practitioners Convention 2004: Choi YH, Hartman DL, Bliss SB, et al. High pregnancy rates after transfer of large equine blastocysts collapsed via micromanipulation before vitrification. Reprod Fertil Dev 2010;22:203. a Equitainer, Hamilton Research, Inc., South Hamilton, MA. b Invitrogen, Carlsbad, CA, USA c Sigma-Aldrich Corp., St. Louis, MO d Chorulon, Intervet, Millsboro, DE, USA e Biorelease deslorelin, BETPharn, Lexington, KY f Torbugesic, Fort Dodge Animal Health, Ford Dodge, IA, USA g Buscopan, Boehringer Ingelheim, Ingelheim, Germany. h Franz L. Personal communication, i Galli C. Personal communication, j Kanitz W. Personal communication, k Dormosedan, Pfizer, Exton, PA l PreSeed EQ, INGfertility, Valleyford, WA, USA m Mila International, Erlanger, KY or Casmed International Ltd., Epsom, Surrey, England KT19 9AP. n Galli C. Personal communication, o Jacobson C. Personal communication, p EmCon filter, Immuno Systems, Spring Valley, WI q Choi et al. Submitted for publication, r Femtotip II, Eppendorf North America, Westbury, NY
Assisted Reproductive Technologies at IGO
 9339 Genesee Avenue, Suite 220 San Diego, CA 92121 858 455 7520 Assisted Reproductive Technologies at IGO Although IGO no longer operates an IVF laboratory or program as such, we work closely with area
9339 Genesee Avenue, Suite 220 San Diego, CA 92121 858 455 7520 Assisted Reproductive Technologies at IGO Although IGO no longer operates an IVF laboratory or program as such, we work closely with area
A POWERFUL IN VITRO FERTILIZATION
 A POWERFUL During the past 50 years technological advances in the field of bovine reproduction have led to some dramatic changes in the way cattle look, reproduce, perform, and even taste. Artificial Insemination
A POWERFUL During the past 50 years technological advances in the field of bovine reproduction have led to some dramatic changes in the way cattle look, reproduce, perform, and even taste. Artificial Insemination
In - Vitro Fertilization Handbook
 In - Vitro Fertilization Handbook William F. Ziegler, D.O. Medical Director Scott Kratka, ELD, TS Embryology Laboratory Director Lauren F. Lucas, P.A.-C, M.S. Physician Assistant Frances Cerniak, R.N.
In - Vitro Fertilization Handbook William F. Ziegler, D.O. Medical Director Scott Kratka, ELD, TS Embryology Laboratory Director Lauren F. Lucas, P.A.-C, M.S. Physician Assistant Frances Cerniak, R.N.
Artificial Insemination in Cattle
 Artificial Insemination in Cattle Introduction This slide show is designed to introduce students to artificial insemination in cattle. However, it is only a brief overview and further training is necessary
Artificial Insemination in Cattle Introduction This slide show is designed to introduce students to artificial insemination in cattle. However, it is only a brief overview and further training is necessary
Day -1 RETRIEVAL OF OVARIES AND OOCYTE COLLECTION AND MATURATION
 Day -1 RETRIEVAL OF OVARIES AND OOCYTE COLLECTION AND MATURATION OVARY COLLECTION Materials and Equipment Needed Thermos with 2 containers with 0.5 L of transport saline in each. Appropriate attire as
Day -1 RETRIEVAL OF OVARIES AND OOCYTE COLLECTION AND MATURATION OVARY COLLECTION Materials and Equipment Needed Thermos with 2 containers with 0.5 L of transport saline in each. Appropriate attire as
Welcome to chapter 8. The following chapter is called "Monitoring IVF Cycle & Oocyte Retrieval". The author is Professor Jie Qiao.
 Welcome to chapter 8. The following chapter is called "Monitoring IVF Cycle & Oocyte Retrieval". The author is Professor Jie Qiao. The learning objectives of this chapter are 2 fold. The first section
Welcome to chapter 8. The following chapter is called "Monitoring IVF Cycle & Oocyte Retrieval". The author is Professor Jie Qiao. The learning objectives of this chapter are 2 fold. The first section
Symposium on RECENT ADVANCES IN ASSISTED REPRODUCTIVE TECHNOLOGY
 Symposium on RECENT ADVANCES IN ASSISTED REPRODUCTIVE TECHNOLOGY Dr Niel Senewirathne Senior Consultant of Obstetrician & Gynaecologist De zoyza Maternity Hospita 1 ART - IVF & ICSI 2 Infertility No pregnancy
Symposium on RECENT ADVANCES IN ASSISTED REPRODUCTIVE TECHNOLOGY Dr Niel Senewirathne Senior Consultant of Obstetrician & Gynaecologist De zoyza Maternity Hospita 1 ART - IVF & ICSI 2 Infertility No pregnancy
ANS 3319C Reproductive Physiology and Endocrinology Techniques for In-Vitro Embryo Production
 ANS 3319C Reproductive Physiology and Endocrinology Techniques for In-Vitro Embryo Production Objectives 1) To gain an understanding of the process of in-vitro embryo production in cattle and other domestic
ANS 3319C Reproductive Physiology and Endocrinology Techniques for In-Vitro Embryo Production Objectives 1) To gain an understanding of the process of in-vitro embryo production in cattle and other domestic
In Vitro Fertilization
 Patient Education In Vitro Fertilization What to expect This handout describes how to prepare for and what to expect when you have in vitro fertilization. It provides written information about this process,
Patient Education In Vitro Fertilization What to expect This handout describes how to prepare for and what to expect when you have in vitro fertilization. It provides written information about this process,
In Vitro Fertilization (IVF) Page 1 of 11
 In Vitro Fertilization (IVF) Page 1 of 11 This document is a part of your informed consent process. Both partners should read the entire document carefully. In vitro fertilization (IVF) is a treatment
In Vitro Fertilization (IVF) Page 1 of 11 This document is a part of your informed consent process. Both partners should read the entire document carefully. In vitro fertilization (IVF) is a treatment
Artificial insemination
 Artificial insemination What is involved? Artificial insemination is an assisted reproduction technique that consists of inserting laboratory-treated spermatozoa into the woman s uterus or cervical canal.
Artificial insemination What is involved? Artificial insemination is an assisted reproduction technique that consists of inserting laboratory-treated spermatozoa into the woman s uterus or cervical canal.
ART - - - - - IVF - NO.1 IVF - IVF
 The success of any ART laboratory depends on its IVF laboratory. The primary function of an ART laboratory is to provide an optimal environment for gametes and embryos. To set up an ART laboratory, the
The success of any ART laboratory depends on its IVF laboratory. The primary function of an ART laboratory is to provide an optimal environment for gametes and embryos. To set up an ART laboratory, the
Authorized By: Holly C. Bakke, Commissioner, Department of Banking and Insurance.
 INSURANCE DIVISION OF INSURANCE Actuarial Services Benefit Standards for Infertility Coverage Proposed New Rules: N.J.A.C. 11:4-54 Authorized By: Holly C. Bakke, Commissioner, Department of Banking and
INSURANCE DIVISION OF INSURANCE Actuarial Services Benefit Standards for Infertility Coverage Proposed New Rules: N.J.A.C. 11:4-54 Authorized By: Holly C. Bakke, Commissioner, Department of Banking and
Understanding Animal Reproduction Technology
 Lesson 251c Understanding Animal Reproduction Technology Core Area. Animal Science Unit 250. Genetics and Breeding Topic 251. Fertilization California Academic Standard. Science Grades 9 through 12 Biology/Life
Lesson 251c Understanding Animal Reproduction Technology Core Area. Animal Science Unit 250. Genetics and Breeding Topic 251. Fertilization California Academic Standard. Science Grades 9 through 12 Biology/Life
Standard Operating Procedure IVF and ICSI procedures
 Pages: 1 of 6 PRINCIPLES Oocytes are aspirated from mature Graafian follicles after the administration of ovarian hyperstimulation pharmacological regimes designed to produce multiple follicular development.
Pages: 1 of 6 PRINCIPLES Oocytes are aspirated from mature Graafian follicles after the administration of ovarian hyperstimulation pharmacological regimes designed to produce multiple follicular development.
Reproductive Technology. Chapter 21
 Reproductive Technology Chapter 21 Assisted Reproduction When a couple is sub-fertile or infertile they may need Assisted Reproduction to become pregnant: Replace source of gametes Sperm, oocyte or zygote
Reproductive Technology Chapter 21 Assisted Reproduction When a couple is sub-fertile or infertile they may need Assisted Reproduction to become pregnant: Replace source of gametes Sperm, oocyte or zygote
Informed Consent Packet - In Vitro Fertilization (IVF)
 Center for Reproductive Medicine (CRM) Informed Consent Packet - In Vitro Fertilization (IVF) This packet contains the required IVF treatment consent documents. Please read, consider and, if you agree,
Center for Reproductive Medicine (CRM) Informed Consent Packet - In Vitro Fertilization (IVF) This packet contains the required IVF treatment consent documents. Please read, consider and, if you agree,
Headquarters in Sioux Center, IA 1
 Nicholas Lemmel Cornfields, soybeans, and cows, I had finally arrived at Trans Ova Genetics Headquarters in Sioux Center, Iowa. I pulled into the drive of the intern house located on the corner of the
Nicholas Lemmel Cornfields, soybeans, and cows, I had finally arrived at Trans Ova Genetics Headquarters in Sioux Center, Iowa. I pulled into the drive of the intern house located on the corner of the
Ovarian Cysts in Dairy Cattle
 AS-451-W Reviewed 2001 Purdue University Cooperative Extension Service West Lafayette, IN 47907 Ovarian Cysts in Dairy Cattle R. D. Allrich, Department of Animal Sciences Purdue University, West Lafayette,
AS-451-W Reviewed 2001 Purdue University Cooperative Extension Service West Lafayette, IN 47907 Ovarian Cysts in Dairy Cattle R. D. Allrich, Department of Animal Sciences Purdue University, West Lafayette,
CONSENT TO PARTICIPATE IN THE IN VITRO FERTILIZATION-EMBRYO TRANSFER PROGRAM
 CONSENT TO PARTICIPATE IN THE IN VITRO FERTILIZATION-EMBRYO TRANSFER PROGRAM I, after consultation with my physician, request to participate in the In Vitro Fertilization (IVF)-Embryo Transfer (ET) procedures
CONSENT TO PARTICIPATE IN THE IN VITRO FERTILIZATION-EMBRYO TRANSFER PROGRAM I, after consultation with my physician, request to participate in the In Vitro Fertilization (IVF)-Embryo Transfer (ET) procedures
ANS 3319C Reproductive Physiology and Endocrinology Artificial Insemination in Cattle. Objectives. What are the advantages and disadvantages of AI?
 ANS 3319C Reproductive Physiology and Endocrinology Artificial Insemination in Cattle Objectives 1) To provide an overview of the process of artificial insemination (AI) in cattle. 2) To gain an understanding
ANS 3319C Reproductive Physiology and Endocrinology Artificial Insemination in Cattle Objectives 1) To provide an overview of the process of artificial insemination (AI) in cattle. 2) To gain an understanding
Proceedings, Applied Reproductive Strategies in Beef Cattle September 11 and 12, 2007, Billings, Montana NEW TECHNOLOGIES FOR REPRODUCTION IN CATTLE
 Proceedings, Applied Reproductive Strategies in Beef Cattle September 11 and 12, 2007, Billings, Montana NEW TECHNOLOGIES FOR REPRODUCTION IN CATTLE George E. Seidel, Jr. Animal Reproduction and Biotechnology
Proceedings, Applied Reproductive Strategies in Beef Cattle September 11 and 12, 2007, Billings, Montana NEW TECHNOLOGIES FOR REPRODUCTION IN CATTLE George E. Seidel, Jr. Animal Reproduction and Biotechnology
THE CENTER FOR ADVANCED REPRODUCTIVE SERVICES (CARS) (The Center) CONSENT FOR IN VITRO FERTILIZATION AND EMBRYO TRANSFER
 THE CENTER FOR ADVANCED REPRODUCTIVE SERVICES (CARS) (The Center) CONSENT FOR IN VITRO FERTILIZATION AND EMBRYO TRANSFER Partner #1 Last Name (Surname): Partner #1 First Name: Partner #1 Last 5 Digits
THE CENTER FOR ADVANCED REPRODUCTIVE SERVICES (CARS) (The Center) CONSENT FOR IN VITRO FERTILIZATION AND EMBRYO TRANSFER Partner #1 Last Name (Surname): Partner #1 First Name: Partner #1 Last 5 Digits
Fertility care for women diagnosed with cancer
 Saint Mary s Hospital Department of Reproductive Medicine Fertility care for women diagnosed with cancer Information For Patients INF/DRM/NUR/16 V1/01/11/2013 1 2 Contents Page Overview 4 Our Service 4
Saint Mary s Hospital Department of Reproductive Medicine Fertility care for women diagnosed with cancer Information For Patients INF/DRM/NUR/16 V1/01/11/2013 1 2 Contents Page Overview 4 Our Service 4
Consent for In Vitro Fertilization
 Consent for In Vitro Fertilization Print Patient s Name Print Partner s Name We (I), the undersigned, request, authorize and consent to the procedure of In Vitro Fertilization (IVF) and Embryo Transfer
Consent for In Vitro Fertilization Print Patient s Name Print Partner s Name We (I), the undersigned, request, authorize and consent to the procedure of In Vitro Fertilization (IVF) and Embryo Transfer
DARTMOUTH-HITCHCOCK MEDICAL CENTER Lebanon, New Hampshire IN VITRO FERTILIZATION PROCEDURE DESCRIPTION
 DARTMOUTH-HITCHCOCK MEDICAL CENTER Lebanon, New Hampshire IN VITRO FERTILIZATION PROCEDURE DESCRIPTION I. INTRODUCTION A. The Assisted Reproductive Technology (ART) Program. The ART Program is operated
DARTMOUTH-HITCHCOCK MEDICAL CENTER Lebanon, New Hampshire IN VITRO FERTILIZATION PROCEDURE DESCRIPTION I. INTRODUCTION A. The Assisted Reproductive Technology (ART) Program. The ART Program is operated
Illinois Insurance Facts Illinois Department of Financial and Professional Regulation Division of Insurance
 Illinois Insurance Facts Illinois Department of Financial and Professional Regulation Division of Insurance Insurance Coverage for Infertility Treatment Revised November 2004 Infertility is a condition
Illinois Insurance Facts Illinois Department of Financial and Professional Regulation Division of Insurance Insurance Coverage for Infertility Treatment Revised November 2004 Infertility is a condition
Consent for Frozen Donor Oocyte In Vitro Fertilization and Embryo Transfer (Recipient)
 Name of Patient: Name of Partner: We, the Patient and Partner (if applicable) named above, are each over the age of twenty-one (21) years. By our signatures below, I/we request and authorize the performance
Name of Patient: Name of Partner: We, the Patient and Partner (if applicable) named above, are each over the age of twenty-one (21) years. By our signatures below, I/we request and authorize the performance
Infertility Services Medical Policy For University of Vermont Medical Center and Central Vermont Medical Center employer groups
 Infertility Services Medical Policy For University of Vermont Medical Center and Central Vermont Medical Center employer groups File name: Infertility Services File code: UM.REPRO.01 Last Review: 02/2016
Infertility Services Medical Policy For University of Vermont Medical Center and Central Vermont Medical Center employer groups File name: Infertility Services File code: UM.REPRO.01 Last Review: 02/2016
EVERY LIVING THING has a number of
 Anatomy and Physiology of Animal Reproductive Systems EVERY LIVING THING has a number of organ systems operating to perform specific functions. If you were to examine one of these systems, you would observe
Anatomy and Physiology of Animal Reproductive Systems EVERY LIVING THING has a number of organ systems operating to perform specific functions. If you were to examine one of these systems, you would observe
Artificial Insemination. Advanced Reproductive Techniques in Small Ruminants. Success of AI Programs 9/20/2013
 Artificial Insemination Advanced Reproductive Techniques in Small Ruminants Jason W. Johnson, DVM, MS, Dip ACT Medical Director Large Animal Teaching and Research Center Assistant Professor of Theriogenology
Artificial Insemination Advanced Reproductive Techniques in Small Ruminants Jason W. Johnson, DVM, MS, Dip ACT Medical Director Large Animal Teaching and Research Center Assistant Professor of Theriogenology
Guide to IVF Laboratory Results
 Guide to IVF Laboratory Results PACIFIC CENTRE FOR REPRODUCTIVE MEDICINE 500-4601 Canada Way, Burnaby BC V5G 4X7 LAB-416-20140122-1 The following information will guide you through what results to expect
Guide to IVF Laboratory Results PACIFIC CENTRE FOR REPRODUCTIVE MEDICINE 500-4601 Canada Way, Burnaby BC V5G 4X7 LAB-416-20140122-1 The following information will guide you through what results to expect
Risks and complications of assisted conception
 Risks and complications of assisted conception August 005 Richard Kennedy British Fertility Society Factsheet www.fertility.org.uk No medical treatment is entirely free from risk and infertility treatment
Risks and complications of assisted conception August 005 Richard Kennedy British Fertility Society Factsheet www.fertility.org.uk No medical treatment is entirely free from risk and infertility treatment
REPRODUCTIVE MEDICINE AND INFERTILITY ASSOCIATES Woodbury Medical Arts Building 2101 Woodwinds Drive Woodbury, MN 55125 (651) 222-6050
 REPRODUCTIVE MEDICINE AND INFERTILITY ASSOCIATES Woodbury Medical Arts Building 2101 Woodwinds Drive Woodbury, MN 55125 (651) 222-6050 RECIPIENT COUPLE INFORMED CONSENT AND AUTHORIZATION FOR IN VITRO FERTILIZATION
REPRODUCTIVE MEDICINE AND INFERTILITY ASSOCIATES Woodbury Medical Arts Building 2101 Woodwinds Drive Woodbury, MN 55125 (651) 222-6050 RECIPIENT COUPLE INFORMED CONSENT AND AUTHORIZATION FOR IN VITRO FERTILIZATION
AGE & FERTILITY: Effective Evaluation & Treatment I. LANE WONG, MD, FACOG. www.hopefertilitycenter.com www.hopeivf.com
 Page 1 of 6 AGE & FERTILITY: Effective Evaluation & Treatment I. LANE WONG, MD, FACOG. www.hopefertilitycenter.com www.hopeivf.com Age has a profound effect on female fertility. This is common knowledge,
Page 1 of 6 AGE & FERTILITY: Effective Evaluation & Treatment I. LANE WONG, MD, FACOG. www.hopefertilitycenter.com www.hopeivf.com Age has a profound effect on female fertility. This is common knowledge,
Artificial Insemination Technique. Dairy Integrated Reproductive Management. Dr. M.L. O Connor The Pennsylvania State University. Reproductive Anatomy
 Artificial Insemination Technique IRM-12 Dairy Integrated Reproductive Management Dr. M.L. O Connor The Pennsylvania State University Many dairy producers are artificially breeding their own cattle. A
Artificial Insemination Technique IRM-12 Dairy Integrated Reproductive Management Dr. M.L. O Connor The Pennsylvania State University Many dairy producers are artificially breeding their own cattle. A
Technological & Ethical Issues In Laboratory-Assisted Reproduction A Short History to Accompany the Lecture
 1 Technological & Ethical Issues In Laboratory-Assisted Reproduction A Short History to Accompany the Lecture Richard Bronson, M.D. Professor of Obstetrics & Gynecology and Pathology The treatment of infertility
1 Technological & Ethical Issues In Laboratory-Assisted Reproduction A Short History to Accompany the Lecture Richard Bronson, M.D. Professor of Obstetrics & Gynecology and Pathology The treatment of infertility
REPRODUCTION IN DONKEYS
 REPRODUCTION IN DONKEYS Stephen R. Purdy, DVM Department of Veterinary and Animal Science University of Massachusetts, Amherst, MA INTRODUCTION This is an overview of the practical aspects of male and
REPRODUCTION IN DONKEYS Stephen R. Purdy, DVM Department of Veterinary and Animal Science University of Massachusetts, Amherst, MA INTRODUCTION This is an overview of the practical aspects of male and
PRODUCERS can choose to use natural
 Artificial Insemination PRODUCERS can choose to use natural or artificial means of breeding their animals. Technology has advanced in the last 30 to 40 years to allow animal producers to use means other
Artificial Insemination PRODUCERS can choose to use natural or artificial means of breeding their animals. Technology has advanced in the last 30 to 40 years to allow animal producers to use means other
BOER GOAT EMBRYO TRANSFER
 BOER GOAT EMBRYO TRANSFER Good management No shortcuts PLAN AHEAD AVOID STRESS Some examples of how stress is induced are: Mixing groups or individual animals together that have not previously been together.
BOER GOAT EMBRYO TRANSFER Good management No shortcuts PLAN AHEAD AVOID STRESS Some examples of how stress is induced are: Mixing groups or individual animals together that have not previously been together.
Assignment Discovery Online Curriculum
 Assignment Discovery Online Curriculum Lesson title: In Vitro Fertilization Grade level: 9-12, with adaptation for younger students Subject area: Life Science Duration: Two class periods Objectives: Students
Assignment Discovery Online Curriculum Lesson title: In Vitro Fertilization Grade level: 9-12, with adaptation for younger students Subject area: Life Science Duration: Two class periods Objectives: Students
Artificial Reproductive Technologies I: insemination
 Artificial Reproductive Technologies I: insemination Cinzia Allegrucci LO s List the main advantages and disadvantages of artificial insemination (AI) Explain methods for evaluating volume and concentration,
Artificial Reproductive Technologies I: insemination Cinzia Allegrucci LO s List the main advantages and disadvantages of artificial insemination (AI) Explain methods for evaluating volume and concentration,
Artificial insemination with donor sperm
 Artificial insemination with donor sperm Ref. 123 / 2009 Reproductive Medicine Unit Servicio de Medicina de la Reproducción Gran Vía Carlos III 71-75 08028 Barcelona Tel. (+34) 93 227 47 00 Fax. (+34)
Artificial insemination with donor sperm Ref. 123 / 2009 Reproductive Medicine Unit Servicio de Medicina de la Reproducción Gran Vía Carlos III 71-75 08028 Barcelona Tel. (+34) 93 227 47 00 Fax. (+34)
LEUKODYSTROPHY GENETICS AND REPRODUCTIVE OPTIONS FOR AFFECTED FAMILIES. Leila Jamal, ScM Kennedy Krieger Institute, Baltimore MD
 LEUKODYSTROPHY GENETICS AND REPRODUCTIVE OPTIONS FOR AFFECTED FAMILIES Leila Jamal, ScM Kennedy Krieger Institute, Baltimore MD 2 Outline Genetics 101: Basic Concepts and Myth Busting Inheritance Patterns
LEUKODYSTROPHY GENETICS AND REPRODUCTIVE OPTIONS FOR AFFECTED FAMILIES Leila Jamal, ScM Kennedy Krieger Institute, Baltimore MD 2 Outline Genetics 101: Basic Concepts and Myth Busting Inheritance Patterns
X-Plain Subclavian Inserted Central Catheter (SICC Line) Reference Summary
 X-Plain Subclavian Inserted Central Catheter (SICC Line) Reference Summary Introduction A Subclavian Inserted Central Catheter, or subclavian line, is a long thin hollow tube inserted in a vein under the
X-Plain Subclavian Inserted Central Catheter (SICC Line) Reference Summary Introduction A Subclavian Inserted Central Catheter, or subclavian line, is a long thin hollow tube inserted in a vein under the
How To Get A Refund On An Ivf Cycle
 100% IVF Refund Program Community Hospital North Clearvista Dr. N Dr. David Carnovale and Dr. Jeffrey Boldt, along with everyone at Community Reproductive Endocrinology, are committed to providing you
100% IVF Refund Program Community Hospital North Clearvista Dr. N Dr. David Carnovale and Dr. Jeffrey Boldt, along with everyone at Community Reproductive Endocrinology, are committed to providing you
Quick guide: using CTE security straws for cryopreservation of pronuclear stages, embryos and sperm
 Quick guide: using CTE security straws for cryopreservation of pronuclear stages, embryos and sperm 1. How to prepare straws for pronuclear stages and embryos (use straws with plug, 66.5 mm) Straws are
Quick guide: using CTE security straws for cryopreservation of pronuclear stages, embryos and sperm 1. How to prepare straws for pronuclear stages and embryos (use straws with plug, 66.5 mm) Straws are
COVENTRY HEALTH CARE OF ILLINOIS, INC. COVENTRY HEALTH CARE OF MISSOURI, INC. Medical Management Policy and Procedure PROPRIETARY
 COVENTRY HEALTH CARE OF ILLINOIS, INC. COVENTRY HEALTH CARE OF MISSOURI, INC. Medical Management Policy and Procedure PROPRIETARY Policy: Infertility Evaluation and Treatment Number: MM 1306 Date Effective:
COVENTRY HEALTH CARE OF ILLINOIS, INC. COVENTRY HEALTH CARE OF MISSOURI, INC. Medical Management Policy and Procedure PROPRIETARY Policy: Infertility Evaluation and Treatment Number: MM 1306 Date Effective:
LONDON REGIONAL TRANSGENIC AND GENE TARGETING FACILITY
 LONDON REGIONAL TRANSGENIC AND GENE TARGETING FACILITY SPERM CRYOPRESERVATION AND IN VITRO FERTILIZATION RESOURCE PACKAGE For information, contact: Dr. Christopher Pin, PhD, Scientific Director London
LONDON REGIONAL TRANSGENIC AND GENE TARGETING FACILITY SPERM CRYOPRESERVATION AND IN VITRO FERTILIZATION RESOURCE PACKAGE For information, contact: Dr. Christopher Pin, PhD, Scientific Director London
טופס הסכמה לטיפולי הפרייה חוץ גופית
 טופס הסכמה לטיפולי הפרייה חוץ גופית CONSENT FORM: IN-VITRO FERTILIZATION (IVF) 1. General In-vitro fertilization is performed in cases of impaired fertility, which may be caused by the following: Obstruction
טופס הסכמה לטיפולי הפרייה חוץ גופית CONSENT FORM: IN-VITRO FERTILIZATION (IVF) 1. General In-vitro fertilization is performed in cases of impaired fertility, which may be caused by the following: Obstruction
Revised minimum standards for in vitro fertilization, gamete intrafallopian transfer, and related procedures
 Revised minimum standards for in vitro fertilization, gamete intrafallopian transfer, and related procedures A PRACTICE COMMITTEE REPORT Guidelines and Minimum Standards I. Introduction Treatment of the
Revised minimum standards for in vitro fertilization, gamete intrafallopian transfer, and related procedures A PRACTICE COMMITTEE REPORT Guidelines and Minimum Standards I. Introduction Treatment of the
Abnormal Uterine Bleeding FAQ Sheet
 Abnormal Uterine Bleeding FAQ Sheet What is abnormal uterine bleeding? Under normal circumstances, a woman's uterus sheds a limited amount of blood during each menstrual period. Bleeding that occurs between
Abnormal Uterine Bleeding FAQ Sheet What is abnormal uterine bleeding? Under normal circumstances, a woman's uterus sheds a limited amount of blood during each menstrual period. Bleeding that occurs between
MINISTRY OF HEALTH Quality and Service Administration. Fe r t i l i z at i o n. to I n - V i t r o. G u i d e. i n I s r a e l
 MINISTRY OF HEALTH Quality and Service Administration G u i d e to I n - V i t r o Fe r t i l i z at i o n i n I s r a e l Contents Introduction 3 The Natural Fertilization Process 4 In Vitro Fertilization
MINISTRY OF HEALTH Quality and Service Administration G u i d e to I n - V i t r o Fe r t i l i z at i o n i n I s r a e l Contents Introduction 3 The Natural Fertilization Process 4 In Vitro Fertilization
Recommended Resources: The following resources may be useful in teaching this
 Unit B: Anatomy and Physiology of Poultry Lesson 4: Artificial Poultry Reproduction Student Learning Objectives: Instruction in this lesson should result in students achieving the following objectives:
Unit B: Anatomy and Physiology of Poultry Lesson 4: Artificial Poultry Reproduction Student Learning Objectives: Instruction in this lesson should result in students achieving the following objectives:
Age and Fertility. A Guide for Patients PATIENT INFORMATION SERIES
 Age and Fertility A Guide for Patients PATIENT INFORMATION SERIES Published by the American Society for Reproductive Medicine under the direction of the Patient Education Committee and the Publications
Age and Fertility A Guide for Patients PATIENT INFORMATION SERIES Published by the American Society for Reproductive Medicine under the direction of the Patient Education Committee and the Publications
Egg Donation Process, Risk, Consent and Agreement
 Department of Obstetrics and Gynecology Strong Fertility Center Kathleen Hoeger, MD, MPH Director Bala Bhagavath, MD Vivian Lewis, MD John T. Queenan, Jr., MD Wendy Vitek, MD Egg Donation Process, Risk,
Department of Obstetrics and Gynecology Strong Fertility Center Kathleen Hoeger, MD, MPH Director Bala Bhagavath, MD Vivian Lewis, MD John T. Queenan, Jr., MD Wendy Vitek, MD Egg Donation Process, Risk,
Frequently Asked Questions About Ovarian Cancer
 Media Contact: Gerri Gomez Howard Cell: 303-748-3933 [email protected] Frequently Asked Questions About Ovarian Cancer What is ovarian cancer? Ovarian cancer is a cancer that forms in tissues
Media Contact: Gerri Gomez Howard Cell: 303-748-3933 [email protected] Frequently Asked Questions About Ovarian Cancer What is ovarian cancer? Ovarian cancer is a cancer that forms in tissues
Lesbian Pregnancy: Donor Insemination
 Lesbian Pregnancy: Donor Insemination (Based on an article originally published in the American Fertility Association 2010 National Fertility and Adoption Directory. Much of this information will also
Lesbian Pregnancy: Donor Insemination (Based on an article originally published in the American Fertility Association 2010 National Fertility and Adoption Directory. Much of this information will also
The IUI procedure Who should consider an IUI IUI success rates IUI cost What to consider if IUI is unsuccessful. The IUI procedure:
 A Complete Guide to understanding IUI (intrauterine insemination) and artificial insemination (Eric Daiter, MD Board Certified in Reproductive Endocrinology and Infertility) The IUI procedure Who should
A Complete Guide to understanding IUI (intrauterine insemination) and artificial insemination (Eric Daiter, MD Board Certified in Reproductive Endocrinology and Infertility) The IUI procedure Who should
AN OVERVIEW OF EMBRYO TRANSFER IN HORSES
 AN OVERVIEW OF EMBRYO TRANSFER IN HORSES BACKGROUND Embryo transfer refers to the procedure for collecting a fertilized ocyte (embryo) from a donor mare and transferring it to the synchronized reproductive
AN OVERVIEW OF EMBRYO TRANSFER IN HORSES BACKGROUND Embryo transfer refers to the procedure for collecting a fertilized ocyte (embryo) from a donor mare and transferring it to the synchronized reproductive
Stem Cell Quick Guide: Stem Cell Basics
 Stem Cell Quick Guide: Stem Cell Basics What is a Stem Cell? Stem cells are the starting point from which the rest of the body grows. The adult human body is made up of hundreds of millions of different
Stem Cell Quick Guide: Stem Cell Basics What is a Stem Cell? Stem cells are the starting point from which the rest of the body grows. The adult human body is made up of hundreds of millions of different
Director, IVF Program, Division of Reproductive Endocrinology & Infertility
 Director, IVF Program, Division of Reproductive Endocrinology & Infertility Date: January 17, 2006 To: From: RE: All IVF candidates Chief, Reproductive Endocrinology & Infertility Criteria for IVF program
Director, IVF Program, Division of Reproductive Endocrinology & Infertility Date: January 17, 2006 To: From: RE: All IVF candidates Chief, Reproductive Endocrinology & Infertility Criteria for IVF program
PICSI Sperm Selection Device Instructions for Use
 PICSI Sperm Selection Device Instructions for Use Manufacturer: Biocoat, Inc., 211 Witmer Rd., Horsham, PA 19044 USA Distributor: MidAtlantic Diagnostics, Inc., Telephone: 856-762-2000, www.midatlanticdiagnostics.com
PICSI Sperm Selection Device Instructions for Use Manufacturer: Biocoat, Inc., 211 Witmer Rd., Horsham, PA 19044 USA Distributor: MidAtlantic Diagnostics, Inc., Telephone: 856-762-2000, www.midatlanticdiagnostics.com
Summary of Product Characteristics
 Summary of Product Characteristics 1 NAME OF THE VETERINARY MEDICINAL PRODUCT ACEGON, 50 microgram/ml, solution for injection for cattle. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains: Active
Summary of Product Characteristics 1 NAME OF THE VETERINARY MEDICINAL PRODUCT ACEGON, 50 microgram/ml, solution for injection for cattle. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains: Active
INFORMED CONSENT AND AUTHORIZATION FOR IN VITRO FERTILIZATION OF PREVIOUSLY CRYOPRESERVED OOCYTES
 INFORMED CONSENT AND AUTHORIZATION FOR IN VITRO FERTILIZATION OF PREVIOUSLY CRYOPRESERVED OOCYTES We, the undersigned, as patient and partner understand that we will be undergoing one or more procedures
INFORMED CONSENT AND AUTHORIZATION FOR IN VITRO FERTILIZATION OF PREVIOUSLY CRYOPRESERVED OOCYTES We, the undersigned, as patient and partner understand that we will be undergoing one or more procedures
Center for Women s Reproductive Care at Columbia University
 Center for Women s Reproductive Care at Columbia University Oocyte Recipients Greetings, Thank you for your interest in the Center for Women s Reproductive Care at Columbia University. We hope that the
Center for Women s Reproductive Care at Columbia University Oocyte Recipients Greetings, Thank you for your interest in the Center for Women s Reproductive Care at Columbia University. We hope that the
Preimplantation Genetic Diagnosis (PGD) in Western Australia
 Preimplantation Genetic Diagnosis (PGD) in Western Australia Human somatic cells have 46 chromosomes each, made up of the 23 chromosomes provided by the egg and the sperm cell from each parent. Each chromosome
Preimplantation Genetic Diagnosis (PGD) in Western Australia Human somatic cells have 46 chromosomes each, made up of the 23 chromosomes provided by the egg and the sperm cell from each parent. Each chromosome
Recent Progress in In Vitro Fertilization and Intracytoplasmic Sperm Injection Technologies in Japan
 Research and Reviews Recent Progress in In Vitro Fertilization and Intracytoplasmic Sperm Injection Technologies in Japan JMAJ 52(1): 29 33, 2009 Kaoru YANAGIDA* 1 Abstract The three basic pillars of fertility
Research and Reviews Recent Progress in In Vitro Fertilization and Intracytoplasmic Sperm Injection Technologies in Japan JMAJ 52(1): 29 33, 2009 Kaoru YANAGIDA* 1 Abstract The three basic pillars of fertility
WOMENCARE A Healthy Woman is a Powerful Woman (407) 898-1500. Endometriosis
 Endometriosis WOMENCARE A Healthy Woman is a Powerful Woman (407) 898-1500 The lining of the uterus is called the endometrium. Sometimes, endometrial tissue grows elsewhere in the body. When this happens
Endometriosis WOMENCARE A Healthy Woman is a Powerful Woman (407) 898-1500 The lining of the uterus is called the endometrium. Sometimes, endometrial tissue grows elsewhere in the body. When this happens
Human Adult Mesothelial Cell Manual
 Human Adult Mesothelial Cell Manual INSTRUCTION MANUAL ZBM0025.01 SHIPPING CONDITIONS Human Adult Mesothelial Cells Orders are delivered via Federal Express courier. All US and Canada orders are shipped
Human Adult Mesothelial Cell Manual INSTRUCTION MANUAL ZBM0025.01 SHIPPING CONDITIONS Human Adult Mesothelial Cells Orders are delivered via Federal Express courier. All US and Canada orders are shipped
IVF OVERVIEW. Tracy Telles, M.D.
 IVF OVERVIEW By Tracy Telles, M.D. Dr. Hendler: Hello and welcome to KP Healthcast. I m your host Dr. Peter Hendler and today our guest is Dr. Tracy Telles. Dr. Telles is an IVF physician in Kaiser Walnut
IVF OVERVIEW By Tracy Telles, M.D. Dr. Hendler: Hello and welcome to KP Healthcast. I m your host Dr. Peter Hendler and today our guest is Dr. Tracy Telles. Dr. Telles is an IVF physician in Kaiser Walnut
Unit B: Understanding Animal Reproduction. Lesson 3: Understanding Animal Reproduction Technology
 Unit B: Understanding Animal Reproduction Lesson 3: Understanding Animal Reproduction Technology Student Learning Objectives: Instruction in this lesson should result in students achieving the following
Unit B: Understanding Animal Reproduction Lesson 3: Understanding Animal Reproduction Technology Student Learning Objectives: Instruction in this lesson should result in students achieving the following
WHAT YOU SHOULD KNOW ABOUT ABORTION
 WHAT YOU SHOULD KNOW ABOUT ABORTION It is the public policy of the state of Idaho to prefer live childbirth over abortion: "The Supreme Court of the United States having held that the states have a "profound
WHAT YOU SHOULD KNOW ABOUT ABORTION It is the public policy of the state of Idaho to prefer live childbirth over abortion: "The Supreme Court of the United States having held that the states have a "profound
WHAT YOU SHOULD KNOW ABOUT ABORTION
 WHAT YOU SHOULD KNOW ABOUT ABORTION It is the public policy of the state of Idaho to prefer live childbirth over abortion: "The Supreme Court of the United States having held that the states have a "profound
WHAT YOU SHOULD KNOW ABOUT ABORTION It is the public policy of the state of Idaho to prefer live childbirth over abortion: "The Supreme Court of the United States having held that the states have a "profound
Introduction Ovarian cysts are a very common female condition. An ovarian cyst is a fluid-filled sac on an ovary in the female reproductive system.
 Ovarian Cysts Introduction Ovarian cysts are a very common female condition. An ovarian cyst is a fluid-filled sac on an ovary in the female reproductive system. Most women have ovarian cysts sometime
Ovarian Cysts Introduction Ovarian cysts are a very common female condition. An ovarian cyst is a fluid-filled sac on an ovary in the female reproductive system. Most women have ovarian cysts sometime
Sexuality after your Spinal Cord Injury
 Sexuality after your Spinal Cord Injury Introduction Spinal Cord Injury (SCI) affects to varying degrees the mechanics and sensations of sex and sexuality. Some people think that spinal injury means an
Sexuality after your Spinal Cord Injury Introduction Spinal Cord Injury (SCI) affects to varying degrees the mechanics and sensations of sex and sexuality. Some people think that spinal injury means an
Placement of an indwelling urinary catheter in female dogs
 Female Dog Urinary Catheterization 1 of 6 Placement of an indwelling urinary catheter in female dogs Bernie Hansen DVM MS North Carolina State University College of Veterinary Medicine Materials Needed
Female Dog Urinary Catheterization 1 of 6 Placement of an indwelling urinary catheter in female dogs Bernie Hansen DVM MS North Carolina State University College of Veterinary Medicine Materials Needed
ASSISTED REPRODUCTIVE TECHNOLOGIES (ART)
 ASSISTED REPRODUCTIVE TECHNOLOGIES (ART) Dr. Herve Lucas, MD, PhD, Biologist, Andrologist Dr. Taher Elbarbary, MD Gynecologist-Obstetrician Definitions of Assisted Reproductive Technologies Techniques
ASSISTED REPRODUCTIVE TECHNOLOGIES (ART) Dr. Herve Lucas, MD, PhD, Biologist, Andrologist Dr. Taher Elbarbary, MD Gynecologist-Obstetrician Definitions of Assisted Reproductive Technologies Techniques
Consent to Perform Preimplantation Genetic Screening (PGS) using. Comparative Genomic Hybridization (acgh) or Next Generation Sequencing (NGS)
 Consent to Perform Preimplantation Genetic Screening (PGS) using Array Comparative Genomic Hybridization (acgh ) or Next Generation Sequencing (NGS) Purpose The purpose of Preimplantation Genetic Screening
Consent to Perform Preimplantation Genetic Screening (PGS) using Array Comparative Genomic Hybridization (acgh ) or Next Generation Sequencing (NGS) Purpose The purpose of Preimplantation Genetic Screening
Patient Information: Endometriosis Disease Process and Treatment
 1 William N. Burns, M. D. Associate Professor Department of Obstetrics & Gynecology Joan C. Edwards School of Medicine Marshall University Huntington, West Virginia, USA Patient Information: Endometriosis
1 William N. Burns, M. D. Associate Professor Department of Obstetrics & Gynecology Joan C. Edwards School of Medicine Marshall University Huntington, West Virginia, USA Patient Information: Endometriosis
Preimplantation genetic diagnosis new method of screening of 24 chromosomes with the Array CGH method...2
 August 2012 content 8 Preimplantation genetic diagnosis new method of screening of 24 chromosomes with the Array CGH method...2 Maintaining fertility new opportunities in GENNET...3 Hysteroscopy without
August 2012 content 8 Preimplantation genetic diagnosis new method of screening of 24 chromosomes with the Array CGH method...2 Maintaining fertility new opportunities in GENNET...3 Hysteroscopy without
Course: AG 534 Zoology - Science of Animal Reproduction
 Course: AG 53 Zoology - Science of Animal Reproduction Unit Objective CAERT Lesson Plan Library Unit Problem Area Les son Animal. Plant & Soil Science 1 1,2, 3 Introduction to Animal Science Match terms
Course: AG 53 Zoology - Science of Animal Reproduction Unit Objective CAERT Lesson Plan Library Unit Problem Area Les son Animal. Plant & Soil Science 1 1,2, 3 Introduction to Animal Science Match terms
30% Off Cycle 1. Possible Preliminary Discussions With Contract Negotiations
 Specialists In Reproductive Medicine & Surgery, P.A. www.dreamababy.com [email protected] Excellence, Experience & Ethics Gestational Surrogacy Price List (2015) We here at Specialists in Reproductive
Specialists In Reproductive Medicine & Surgery, P.A. www.dreamababy.com [email protected] Excellence, Experience & Ethics Gestational Surrogacy Price List (2015) We here at Specialists in Reproductive
INTRODUCTION TO ARTIFICIAL INSEMINATION APICHAI POONCHAI D.V.M.
 INTRODUCTION TO ARTIFICIAL INSEMINATION APICHAI POONCHAI D.V.M. In natural mating a bull mounts a cow & ejuculates his semen in her vagina INTRODUCTION In A.I. Semen is collected from a bull in an artificial
INTRODUCTION TO ARTIFICIAL INSEMINATION APICHAI POONCHAI D.V.M. In natural mating a bull mounts a cow & ejuculates his semen in her vagina INTRODUCTION In A.I. Semen is collected from a bull in an artificial
Midland Fertility Services Blastocyst Transfer
 Midland Fertility Services Blastocyst Transfer Building futures, transforming lives What is a blastocyst? When a sperm fertilises an egg the resulting embryo develops from the original single cell into
Midland Fertility Services Blastocyst Transfer Building futures, transforming lives What is a blastocyst? When a sperm fertilises an egg the resulting embryo develops from the original single cell into
Reproduction and its Hormonal Control
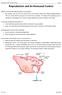 Reproduction and its Hormonal Control Page 1 Reproduction and its Hormonal Control Different mammals have different patterns of reproduction Eg mammals, rats and mice can breed all year round, whereas
Reproduction and its Hormonal Control Page 1 Reproduction and its Hormonal Control Different mammals have different patterns of reproduction Eg mammals, rats and mice can breed all year round, whereas
Clinical Policy Committee
 Northern, Eastern and Western Devon Clinical Commissioning Group South Devon and Torbay Clinical Commissioning Group Clinical Policy Committee Commissioning policy: Assisted Conception Fertility assessment
Northern, Eastern and Western Devon Clinical Commissioning Group South Devon and Torbay Clinical Commissioning Group Clinical Policy Committee Commissioning policy: Assisted Conception Fertility assessment
DNA Fingerprinting. Unless they are identical twins, individuals have unique DNA
 DNA Fingerprinting Unless they are identical twins, individuals have unique DNA DNA fingerprinting The name used for the unambiguous identifying technique that takes advantage of differences in DNA sequence
DNA Fingerprinting Unless they are identical twins, individuals have unique DNA DNA fingerprinting The name used for the unambiguous identifying technique that takes advantage of differences in DNA sequence
Gestational Carrier / Directed Donor In-Vitro Fertilization Handbook
 Gestational Carrier / Directed Donor In-Vitro Fertilization Handbook William F. Ziegler, D.O. Medical Director Scott Kratka, ELD, TS Embryology Laboratory Director Lauren F. Lucas, PA-C, M.S Physician
Gestational Carrier / Directed Donor In-Vitro Fertilization Handbook William F. Ziegler, D.O. Medical Director Scott Kratka, ELD, TS Embryology Laboratory Director Lauren F. Lucas, PA-C, M.S Physician
FERTILITY AND AGE. Introduction. Fertility in the later 30's and 40's. Am I fertile?
 FERTILITY AND AGE Introduction Delaying pregnancy is a common choice for women in today's society. The number of women in their late 30s and 40s attempting pregnancy and having babies has increased in
FERTILITY AND AGE Introduction Delaying pregnancy is a common choice for women in today's society. The number of women in their late 30s and 40s attempting pregnancy and having babies has increased in
, hereby agree to a form of treatment known
 Patient Consent for Therapy Human In Vitro Fertilization and Embryo Transfer This is to certify that I, as In Vitro Fertilization and Embryo Transfer., hereby agree to a form of treatment known I have
Patient Consent for Therapy Human In Vitro Fertilization and Embryo Transfer This is to certify that I, as In Vitro Fertilization and Embryo Transfer., hereby agree to a form of treatment known I have
Consent for Treatment
 IVF_Consent_Booklet_Web_5.2015_IVFNE:IVFConsentBook.v2 5/28/2015 11:21 AM Page 1 Assisted Reproduction In Vitro Fertilization Intracytoplasmic Sperm Injection Assisted Hatching Embryo Cryopreservation
IVF_Consent_Booklet_Web_5.2015_IVFNE:IVFConsentBook.v2 5/28/2015 11:21 AM Page 1 Assisted Reproduction In Vitro Fertilization Intracytoplasmic Sperm Injection Assisted Hatching Embryo Cryopreservation
Artificial Insemination in Dairy Cattle 1
 Whole Document Navigator (Click Here) Artificial Insemination in Dairy Cattle 1 D. W. Webb 2 Artificial insemination (AI) is a process by which sperm are collected from the male, processed, stored and
Whole Document Navigator (Click Here) Artificial Insemination in Dairy Cattle 1 D. W. Webb 2 Artificial insemination (AI) is a process by which sperm are collected from the male, processed, stored and
Arizona State University Institutional Animal Care and Use Committee STANDARD INSTITUTIONAL GUIDELINE RODENT SURGERY
 Arizona State University Institutional Animal Care and Use Committee STANDARD INSTITUTIONAL GUIDELINE RODENT SURGERY It is the policy of the IACUC that all survival surgeries involving rodents be conducted
Arizona State University Institutional Animal Care and Use Committee STANDARD INSTITUTIONAL GUIDELINE RODENT SURGERY It is the policy of the IACUC that all survival surgeries involving rodents be conducted
Uterine fibroids (Leiomyoma)
 Uterine fibroids (Leiomyoma) What are uterine fibroids? Uterine fibroids are fairly common benign (not cancer) growths in the uterus. They occur in about 25 50% of all women. Many women who have fibroids
Uterine fibroids (Leiomyoma) What are uterine fibroids? Uterine fibroids are fairly common benign (not cancer) growths in the uterus. They occur in about 25 50% of all women. Many women who have fibroids
A Guide to Hysteroscopy. Patient Education
 A Guide to Hysteroscopy Patient Education QUESTIONS AND ANSWERS ABOUT HYSTEROSCOPY Your doctor has recommended that you have a procedure called a hysteroscopy. Naturally, you may have questions about
A Guide to Hysteroscopy Patient Education QUESTIONS AND ANSWERS ABOUT HYSTEROSCOPY Your doctor has recommended that you have a procedure called a hysteroscopy. Naturally, you may have questions about
HARVESTING AND CRYOPRESERVATION OF HUMAN EMBRYONIC STEM CELLS (hescs)
 HARVESTING AND CRYOPRESERVATION OF HUMAN EMBRYONIC STEM CELLS (hescs) OBJECTIVE: can be cryopreserved in a liquid nitrogen (LN 2 ) freezer for long-term storage. This Standard Operating Procedure (SOP)
HARVESTING AND CRYOPRESERVATION OF HUMAN EMBRYONIC STEM CELLS (hescs) OBJECTIVE: can be cryopreserved in a liquid nitrogen (LN 2 ) freezer for long-term storage. This Standard Operating Procedure (SOP)
London Fertility Centre Price List
 London Fertility Centre Price List Fertility Testing Packages Standard Female fertility testing package AMH Ultrasound scan 15 minute doctor consultation to discuss your results Premium Female fertility
London Fertility Centre Price List Fertility Testing Packages Standard Female fertility testing package AMH Ultrasound scan 15 minute doctor consultation to discuss your results Premium Female fertility
ARTIFICIAL INSEMINATION
 ARTIFICIAL INSEMINATION Artificial insemination is the deposition of male reproductive cells (spermatozoa) in the female reproductive tract by mechanical means rather than by natural mating. The success
ARTIFICIAL INSEMINATION Artificial insemination is the deposition of male reproductive cells (spermatozoa) in the female reproductive tract by mechanical means rather than by natural mating. The success
